Inhibitory effects of Δ8-tetrahydrocannabinol on major hepatic cytochrome P450 enzymes and implications for drug disposition
Abstract
The increased use of cannabis in many parts of the United States and other countries has led to a need for a more comprehensive understanding of cannabis constituents and their potential for drug-drug interactions. Δ8-Tetrahydrocannabinol (Δ8-THC) is a psychoactive cannabinoid that is found at low concentrations in cannabis but is growing in popularity, especially where the use of Δ9-THC is restricted. Although certain cannabinoids including cannabidiol (CBD) are known to inhibit several metabolizing enzymes including many in the cytochrome P450 family, the effects of Δ8-THC remain poorly characterized. This study evaluated the inhibitory potential of Δ8-THC and its metabolites, 11-hydroxy-Δ8-THC and 11-nor-Δ8-THC-9-carboxylic acid, on major hepatic cytochrome P450 enzymes using in vitro assays with recombinant P450-overexpressing microsomes and pooled human liver microsomes. Δ8-THC and 11-hydroxy-Δ8-THC significantly inhibited CYP2C9- and CYP3A4-mediated metabolism in a dose-dependent, reversible manner. Lineweaver–Burk analysis indicated competitive inhibition for CYP2C9-mediated warfarin hydroxylation and noncompetitive inhibition for CYP2C9- and CYP3A4-mediated metabolism of diclofenac and midazolam, respectively. In contrast, 11-nor-Δ8-THC-9-carboxylic acid showed no significant inhibition of P450 enzymes. Static modeling predicted clinically relevant drug interactions, particularly with oral Δ8-THC. These findings underscore the potential for Δ8-THC to impact the pharmacokinetics of coadministered drugs and highlight the need for further clinical studies.
Significance Statement:
This study is the first to assess how Δ8-tetrahydrocannabinol and its active metabolites inhibit key hepatic P450 enzymes. Results suggest a high risk of Δ8-tetrahydrocannabinol–related drug interactions, especially with oral use, underscoring the need for clinical caution and further research.
Article type: Research Article
Keywords: Cannabinoid, Cannabis, CYP450, Drug-drug interactions, Tetrahydrocannabinol
Affiliations: Division of Molecular Biosciences, Department of Pharmaceutical Sciences, School of Pharmacy and Pharmaceutical Sciences, University at Buffalo, Buffalo, New York; Department of Pharmaceutical Sciences, College of Pharmacy and Pharmaceutical Sciences, Washington State University, Spokane, Washington
License: © 2025 The Author(s) CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.dmd.2025.100122 | PubMed: 40829519 | PMC: PMC12597552
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (229 KB)
Introduction
Cannabis, one of the most widely used natural products worldwide, has been valued for centuries for its medicinal and recreational properties. More than 100 different cannabinoids have been identified in cannabis, with cannabidiol (CBD) and Δ9-tetrahydrocannabinol (Δ9-THC) among the most extensively studied. CBD is known for its low psychoactive properties and therapeutic benefits, including anxiety relief and anti-inflammatory effects.ref. bib1 In contrast, Δ9-THC is the main psychoactive component responsible for the "high" associated with cannabis use.ref. bib2 Their popularity stems from a growing interest in natural health products, shifting legal landscapes, and strong evidence supporting their potential to treat a range of conditions—from chronic pain to epilepsy.ref. bib3 As research expands, cannabinoids continue to drive public attention and scientific exploration, shaping how cannabis is understood and used worldwide.
Although not usually abundant in cannabis, Δ8-THC exhibits similar but milder psychoactive properties than Δ9-THC with reduced anxiety and sedation,ref. bib4 and it has surged in popularity especially in regions where Δ9-THC is restricted.ref. bib5 Structurally similar to Δ9-THC, Δ8-THC differs only in the location of a double bond, lying on the eighth carbon of the THC molecule as compared to the ninth carbon on Δ9-THC.ref. bib4 Its increasing availability is largely due to legal ambiguities in cannabis regulation. Δ8-THC can be derived from hemp-derived CBD and it is less legally restricted than Δ9-THC.ref. bib6 This has led to a flood of Δ8-THC products including edibles, tinctures, and vape cartridges into the consumer market.ref. bib7 A March 2024 survey found that approximately 11% of US 12th graders had used Δ8-THC in the past year.ref. bib8 Some states, including Alaska, Arizona, Arkansas, and Colorado, have banned Δ8-THC, whereas others lack specific legislation. The rapid rise in use raises pressing concerns about its public health implications, particularly its potential to interact with other medications.ref. bib9
The liver plays a central role in drug metabolism, largely through the action of the cytochrome P450 (P450) enzyme system.ref. bib10 P450 enzymes are responsible for the metabolism of most Food and Drug Administration-approved drugs, with enzymes like CYPs 1A2, 2C9, 2C19, 2D6, and 3A4 being especially significant.ref. bib11 Any substance that inhibits or induces these enzymes can affect how other drugs are processed, potentially altering their efficacy or toxicity.ref. bib12 Both Δ9-THC and CBD are metabolized by CYPs 2C9, 2C19, and 3A4, and studies show that they can inhibit multiple P450 enzymes. For example, CBD is a potent inhibitor of CYPs 2D6, 2C9, 3A4, and 2C19, which affects the metabolism of medications like dextromethorphan, warfarin, midazolam, and omeprazole.ref. bib13 Δ9-THC also inhibits CYPs 1A2 and 2C9, affecting the metabolism of drugs such as phenacetin and diclofenac.ref. bib14 The degree of interaction depends on dosage, frequency, and individual metabolic differences, highlighting the clinical importance of cannabinoid-drug interactions.ref. bib3
Given its structural similarity to Δ9-THC, Δ8-THC likely possesses comparable pharmacological properties. For example, it acts as a partial agonist of CB1 and CB2 receptors, with approximately half the potency of Δ9-THC.ref. bib15 Similar to that observed for Δ9-THC, once consumed, Δ8-THC is primarily metabolized to its active metabolite 11-hydroxy-Δ-8-THC (11-OH-Δ8-THC) by CYP2C9,ref. bib16 then oxidized to 11-nor-Δ8-tetrahydrocannabinol-9-carboxylic acid (Δ8-THC-COOH) and excreted in the urine.ref. bib17 Inhalation leads to rapid absorption, with peak plasma concentrations within 10 – 30 minutes. Oral consumption results in slower absorption, with peak effects occurring in 1 – 2 hours.ref. bib18
Despite these biochemical insights, direct studies on the impact of Δ8-THC on P450 enzymes have not yet been performed. In the present study, the effects of Δ8-THC on the activities of several major hepatic P450 enzymes were examined. Results from the present studies indicate a strong potential for drug-drug interactions (DDIs), underscoring the need for caution among individuals using Δ8-THC alongside prescription medications and calling for further research into its clinical interactions.
Material and methods
Chemicals and reagents
Δ8-THC, 11-OH-Δ8-THC, and Δ8-THC-COOH were obtained from Cayman Chemical. Pooled human liver microsomes (HLMs; mixed gender, n = 50 donors) were purchased from Sekisui Xenotech. An NADPH-regenerating system was acquired from Corning. P450-specific probe substrates (phenacetin, bupropion, warfarin sodium, amodiaquine, diclofenac, omeprazole, dextromethorphan, chlorzoxazone, testosterone, and midazolam) of tested P450 enzymes were acquired from LGC Standards, as were their corresponding metabolite standards and internal standards (alpha-hydroxy midazolam-d4, 4-hydroxydiclofenac-d5, and (S)-7-hydroxywarfarin-d5). Liquid chromatography-mass spectrometry (LC-MS)-grade solvents, microcentrifuge tubes, and BCA assay kits were obtained from Fisher Scientific. All other chemicals were analytical grade or higher. The anti-V5 tag monoclonal antibody horseradish peroxidase (HRP) (Catalog No. R96125) was purchased from Novex, Fisher Scientific, whereas the anti-calnexin polyclonal rabbit antibody (Catalog No. 2433) and the anti-rabbit IgG, HRP-linked antibody (Catalog No. 7044S) were obtained from Cell Signaling Technology.
Cytochrome P450 enzyme inhibition assays
V5-tagged human P450 enzymes (CYPs 1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4) were cloned and overexpressed in HEK293 cells and microsomal fractions were isolated by differential centrifugation as previously described,ref. bib19 with expression monitored by western blot analysis. An anti-V5-tagged antibody (1:1000) and an anti-calnexin polyclonal rabbit antibody (1:1000) together with an anti-rabbit IgG, HRP-linked antibody (1:5000) were used to probe V5 and calnexin, respectively (Supplemental Fig. 1). Briefly, microsomes (25 μg) were electrophoresed on a 10% SDS–polyacrylamide gel and proteins were transferred to polyvinylidene difluoride membranes using a iBlot Gel Transfer Stacks polyvinylidene difluoride, Regular kit (Catalog No. IB401001). Membranes were blocked with 5% nonfat milk and incubated overnight at 4°C with a polyclonal rabbit anti-calnexin antibody (1:1000 dilution). They were then incubated for 1 hour at room temperature with an HRP-linked anti-rabbit IgG antibody, followed by incubation with a monoclonal anti-V5 tag antibody. Protein bands were visualized using the SuperSignal Femto Maximum Sensitivity Substrate (Bio-Rad). Calnexin bands served as the loading control for microsomal protein samples, whereas V5-HRP bands indicated the expression of each P450 enzyme, which was not detected in microsomes from parental HEK293 cells. Microsomal protein quantified using the BCA protein assay following the manufacturer’s instructions. To assess P450 enzyme inhibition, Δ8-THC, 11-OH-Δ8-THC, and Δ8-THC-COOH (1 or 10 μM) were incubated with probe substrates in a reaction mixture containing 3 mM MgCl2, potassium phosphate buffer (pH 7.4), and either 20 – 50 μg of recombinant CYP microsomes or 20 μg of pooled HLM. Reactions were preincubated at 37°C for 3 minutes before initiating with an NADPH-regenerating system. Incubation times were 5 – 30 minutes for recombinant P450 microsomes (Supplemental Table 1) and 10 minutes for HLM. Reactions were terminated by the addition of 30 μL of ice-cold acetonitrile, followed by vortexing and centrifugation at 17,000g for 15 minutes. Approximately 30 μL of the supernatant was transferred to an ultraperformance liquid chromatography (UPLC) vial for analysis. To reduce nonspecific binding of cannabinoids, all incubations were performed using low-binding 0.6-mL tubes. Probe substrates were used at concentrations near their Michaelis-Menten constants () to minimize off-target interactions.ref. bib13,ref. 20, ref. 21, ref. 22, ref. 23, ref. 24, ref. 25
Liquid chromatography-tandem mass spectrometry analysis
Metabolites were quantified using a Waters Acquity UPLC system coupled to a Xevo TQD triple-quadrupole mass spectrometer (Waters). Chromatographic separation was achieved on an Acquity UPLC BEH C18 column (2.1 × 100 mm, 1.7 μm) at 40°C. The mobile phase consisted of water with 0.1% formic acid (A) and methanol (B) with an 8-minute gradient as follows: 2 minutes at 95% A, a linear increase to 95% B over 4 minutes, a 1-minute hold at 95% B, and a 1-minute re-equilibration with 95% A. The flow rate was 0.3–0.4 mL/min. Detection was performed in positive electrospray ionization mode using Multiple Reaction Monitoring (Supplemental Table 2). Capillary voltage, cone voltage, and collision energy were set at 0.6 kV, 20 V, and 15 eV, respectively.
Determination of IC50 and Ki values
The percent activity for a given reaction was calculated by comparing metabolite formation between inhibitor-treated incubations and vehicle controls, as detailed below.
To improve cannabinoid solubility without significantly impacting enzyme activity, 3% methanol (MeOH) was used as the vehicle for Δ8-THC, 11-OH-Δ8-THC, and Δ8-THC-COOH in all incubations. Initial screening studies were performed using recombinant P450-overexpressing microsomes and 1 or 10 μM cannabinoid concentrations and validated using HLM. Cannabinoids that reduced activity by ≥ 50% at 1 μM or 10 μM in reactions containing either recombinant enzyme or HLM were further evaluated for in recombinant P450-overexpressing microsomes as well as HLM. assays were conducted across inhibitor concentrations of 0.1 – 100 μM under consistent incubation conditions. All determinations were performed in triplicate for reproducibility. values, defined as the concentration reducing enzyme activity by 50%, were calculated by nonlinear regression (see below) using GraphPad Prism 10.0 (GraphPad):
where represents the logarithm of inhibitor concentration, is the highest % activity (set to 100%, assuming no inhibition without inhibitor), and is the lowest % activity (set to 0%, assuming full inhibition at high concentrations).
IC50 shift assay
To characterize inhibition type, shift studies were performed using recombinant P450-overexpressing microsomes. Assays were preincubated at 37°C for 30 minutes with cannabinoid with or without NADPH in the absence of a probe substrate. After preincubation, substrate was added and incubated for 30 minutes. values were calculated as mentioned above using GraphPad Prism 10.0.
According to Food and Drug Administration guidance, a shift > 1.5 indicates time-dependent inhibition.ref. bib11
To determine Ki values for each P450 enzyme, substrate and inhibitor concentrations were selected based on known Km and values. Substrate concentrations were 2.5, 10, 30, and 60 μM for diclofenacref. bib26 and warfarin,ref. bib27 and 1, 5, 10, and 25 μM for midazolamref. bib28 for CYPs 2C9 and 3A4, respectively. Internal standards (deuterated metabolites) were used to normalize metabolite peak areas. Lineweaver–Burk plots ( vs ) were used to characterize inhibition type. Based on their inhibition type, reversible inhibition data were fitted to multiple models using nonlinear regression in GraphPad Prism 10.0 to estimate values.
where is inhibitor concentration, is the inhibition constant, is the substrate concentration, and the Michaelis-Menten constant. Reaction velocity () refers to the rate of metabolite formation, calculated by dividing the metabolite peak area by the internal standard peak area for a given reaction.
and values were corrected for nonspecific binding using the unbound fraction term (). The of 0.051 in HLM and 0.043 in recombinant P450-overexpressing microsomes for Δ9-THC, and 0.094 in HLM and 0.078 in recombinant P450-overexpressing microsomes for 11-OH-Δ9-THC were used to estimate the corrected inhibitory potency for Δ8-THC and 11-OH-Δ8-THC, respectively.ref. bib13
with and the mean unbound and , respectively.
Prediction of in vivo DDI by static modeling
To predict the risk of clinical DDI, we applied static models of reversible inhibition.ref. bib29 The area under the concentration-time curve ratio () was used to estimate the impact of Δ8-THC and 11-OH-Δ8-THC on probe substrates.
For oral administration, the was calculated using eq.1, with the effect of reversible inhibition, and and denoting time-dependent inhibition and induction respectively. Subscripts “h” and “g” denote hepatic and gut, respectively. and were set to 1 (not applicable here, because no time-dependent inhibition was observed). is the fraction of substrate available after intestinal metabolism, and it was set to 0.64 for diclofenac,ref. bib14 0.99 for warfarin,ref. bib30 and 0.51 for midazolam.ref. bib31 is the fraction of hepatic clearance of the substrate mediated by the P450 enzyme that is subject to inhibition/induction, which was set to 0.98 for diclofenac,ref. bib14 0.91 for warfarin,ref. bib32 and 0.93 for midazolam.ref. bib31 An suggests a significant presystemic hepatic DDI after oral administration.
The was calculated using the (the inhibitor drug concentration in the gut) and values determined in recombinant P450-overexpressing microsomes (eq. 2), with the calculated as indicated in eq.3, with the fraction of absorbed inhibitor (Δ8-THC or 11-OH-Δ8-THC) set to 1.0; , the intestinal absorption rate of inhibitor (Δ8-THC or 11-OH-Δ8-THC) set to 0.1 min-1; and , the enterocyte blood flow set to 0.3 L/min.ref. bib29
The was calculated using the (the inhibitor drug concentration in the liver) and values determined in recombinant P450-overexpressing microsomes (see Results section; eq. 4), with the unbound maximum hepatic inlet concentration () calculated as indicated in eq.5. Due to the chemical similarities between Δ8-THC and Δ9-THC, the (the unbound fraction of drug in plasma) was set at 0.03 for both Δ8-THC and 11-OH-Δ8-THC.ref. bib33 , the ratio of drug concentration in blood () to drug concentration in plasma, was set to 0.40, and , the hepatic blood flow, was set to 1.5 L/min.ref. bib29 is the maximum total plasma concentration of inhibitor (Δ8-THC or 11-OH-Δ8-THC).ref. bib34,ref. bib35
For inhalation exposure, was calculated using eqs. 6 and 7, with the equaling the which is the unbound peak plasma concentration. An indicates a significant systemic DDI after inhalation exposure.ref. bib29
Results
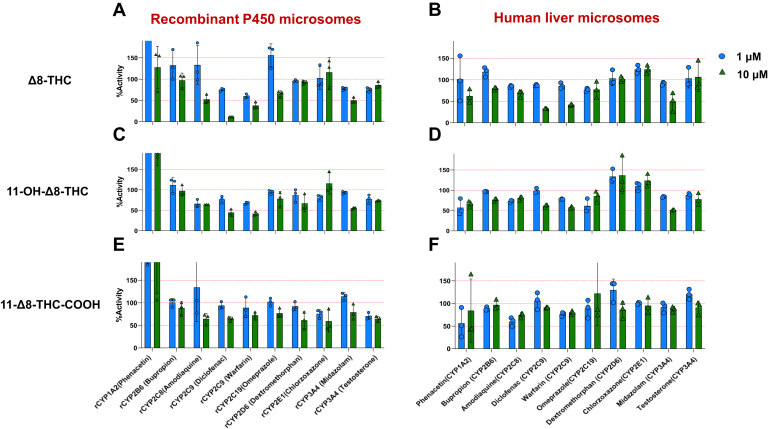
The chemical structures of Δ9-THC, Δ8-THC, and their metabolites are shown in Fig. 1. Initial studies were performed to screen for possible cytochrome P450 inhibition by Δ8-THC, 11-OH-Δ8-THC, and Δ8-THC-COOH. Both Δ8-THC and 11-OH-Δ8-THC exhibited consistent inhibitory effects on both CYP2C9 and CYP3A4 in recombinant P450-overexpressing microsomes as well as HLM using P450-specific probe substrates. At 10 μM, both cannabinoids reduced the formation of 4′-hydroxydiclofenac and 7′-hydroxywarfarin (CYP2C9 substrates) as well as 1′-hydroxymidazolam (CYP3A4 substrate) by > 40% (Fig. 2, A–D). In contrast, Δ8-THC-COOH exhibited minimal or no inhibition against any P450 enzymes tested (Fig. 2, E and F).
curves for Δ8-THC and 11-OH-Δ8-THC inhibition of P450 enzyme activity are shown in Fig. 3. Both compounds demonstrated concentration-dependent inhibition of CYPs 2C9 and 3A4 as indicated by reduced formation of 4′-hydroxydiclofenac (Fig. 3, A and B), 7′-hydroxywarfarin (Fig. 3. C and D), and 1′-hydroxymidazolam (Fig. 3, E and F) in both recombinant P450-overexpressing microsomes and HLM. The unbound () values for Δ8-THC were < 0.5 μM against CYPs 2C9 and 3A4 for both recombinant enzyme microsomes and HLM (Table 1), with the strongest inhibition observed for CYP2C9 in recombinant microsomes ( = 0.07 ± 0.01 μM for the 4′-hydroxylation of diclofenac, and 0.20 ± 0.03 μM for the 7′-hydroxylation of warfarin). Similar inhibition was observed in HLM, with corresponding values of 0.23 ± 0.03 μM and 0.44 ± 0.14 μM, respectively. Strong inhibition of CYP3A4 was also observed for Δ8-THC against 1′-hydroxyl-midazolam formation ( = 0.44 ± 0.14 μM for recombinant CYP3A4-overexpressing microsomes and 0.48 ± 0.11 μM for HLM).
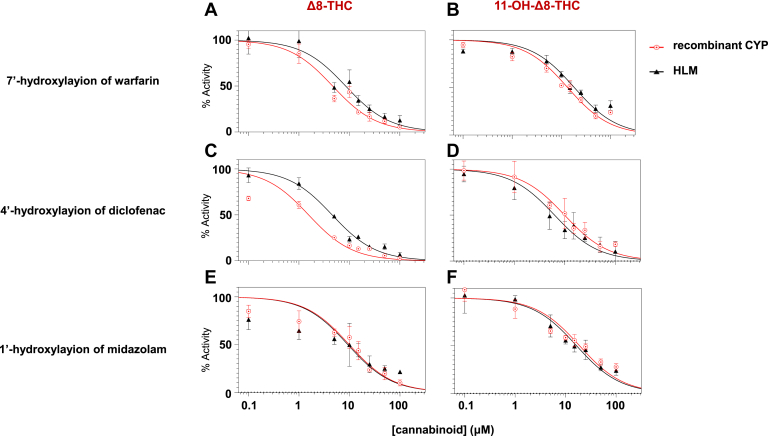
Table 1: Unbound values for Δ8-THC and 11-OH-Δ8-THC against CYP2C9 and CYP3A4
| Cannabinoid | Substrate | Metabolite Formed | Microsomes | a, b (μM) |
|---|---|---|---|---|
| Δ8-THC | Diclofenac | 4′-Hydroxydiclofenac | rec CYP2C9 | 0.07 ± 0.01 |
| HLM | 0.23 ± 0.03 | |||
| Warfarin | 7′-Hydroxywarfarin | rec CYP2C9 | 0.20 ± 0.03 | |
| HLM | 0.44 ± 0.14 | |||
| Midazolam | 1′-Hydroxymidazolam | rec CYP3A4 | 0.44 ± 0.14 | |
| HLM | 0.48 ± 0.11 | |||
| 11-OH- Δ8-THC | Diclofenac | 4′-Hydroxydiclofenac | rec CYP2C9 | 0.79 ± 0.29 |
| HLM | 0.71 ± 0.43 | |||
| Warfarin | 7′-Hydroxywarfarin | rec CYP2C9 | 1.05 ± 0.13 | |
| HLM | 1.72 ± 0.10 | |||
| Midazolam | 1′-Hydroxylmidazolam | rec CYP3A4 | 1.48 ± 0.21 | |
| HLM | 1.57 ± 0.32 |
rec, recombinant.
11-OH-Δ8-THC exhibited moderate-strong inhibition across all P450 enzyme-substrate pairs, with values ranged from 0.71 to 1.70 μM in HLM and 0.79 to 1.50 μM in recombinant P450-overexpressing microsomes. The strongest inhibition was observed against diclofenac 4′-hydroxylation formation in recombinant CYP2C9 microsomes with an = 0.79 ± 0.29 μM, which was approximately 11-fold higher than that of Δ8-THC.
To assess whether Δ8-THC and 11-OH-Δ8-THC exhibit time- and NADPH-dependent inhibition (hallmarks of irreversible or mechanism-based inhibition), shift assays were conducted. Inhibitory potency was measured after a 30-minute preincubation in recombinant CYPs 2C9 and 3A4 microsomes with and without NADPH in the absence of substrate, respectively. curves for Δ8-THC (Fig. 4, A, C, and E) and 11-OH-Δ8-THC (Fig. 4, B, D, and F) across CYPs 2C9- and 3A4-mediated reactions after 30-minute preincubation with or without NADPH. For both cannabinoids, no appreciable differences were observed between the +NADPH and –NADPH conditions. Table 2 summarizes the shift values of Δ8-THC and 11-OH-Δ8-THC in CYPs 2C9- and 3A4-overexpressing microsomes, with the shift reflecting the fold-change in inhibitory potency following a 30-minute preincubation with versus without NADPH. All of the shift values observed in this study were below the shift threshold of 1.5 that is generally used to distinguish between reversible and time-dependent inhibition.ref. bib11 This suggests that neither Δ8-THC nor 11-OH-Δ8-THC undergoes time-dependent inhibition.
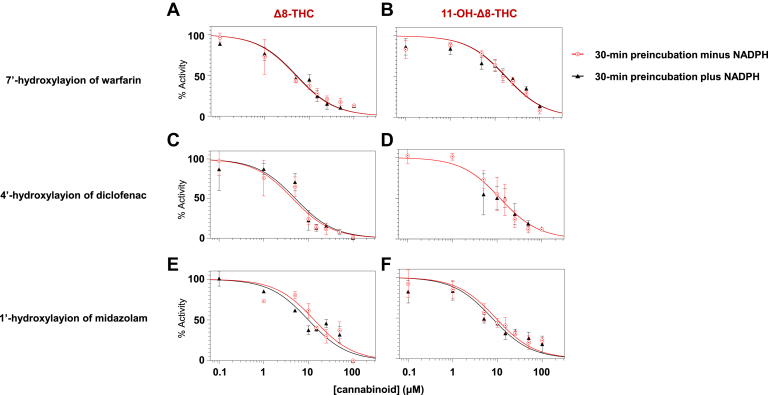
Table 2: The shift of Δ8-THC and 11-OH-Δ8-THC in recombinant P450-overexpressing microsomes
| Cannabinoid | Reaction | Microsomes | Shifta,b |
|---|---|---|---|
| Δ8-THC | 4′-Hydroxylation of diclofenac | rec CYP2C9 | 1.24 ± 0.34 |
| 7′-Hydroxylation of warfarin | rec CYP2C9 | 0.93 ± 0.02 | |
| 1′-Hydroxylation of midazolam | rec CYP3A4 | 0.85 ± 0.09 | |
| 11-OH- Δ8-THC | 4′-Hydroxylation of diclofenac | rec CYP2C9 | 0.95 ± 0.01 |
| 7′-Hydroxylation of warfarin | rec CYP2C9 | 1.11 ± 0.08 | |
| 1′-Hydroxylation of midazolam | rec CYP3A4 | 0.83 ± 0.11 |
rec, recombinant.
To further characterize the reversible inhibition profile of Δ8-THC and 11-OH-Δ8-THC, both cannabinoids were evaluated over a concentration range of 0 – 50 μM in incubations with recombinant P450 microsomes and HLM. Diclofenac and warfarin were used as probe substrates for CYP2C9, and midazolam for CYP3A4, with substrate concentrations ranging from 3 to 60 μM (diclofenac, warfarin) and 1 – 25 μM (midazolam) (see Material and methods for details). Consistent inhibition profiles were observed across both systems. Lineweaver–Burk plots were used to differentiate among inhibition mechanisms (Fig. 5). Δ8-THC and 11-OH-Δ8-THC exhibited noncompetitive inhibition of CYP2C9-mediated diclofenac metabolism and CYP3A4-mediated midazolam metabolism, indicated by shared X-axis intercepts. In contrast, competitive inhibition was observed for CYP2C9-mediated warfarin metabolism, as shown by shared Y-axis intercepts.
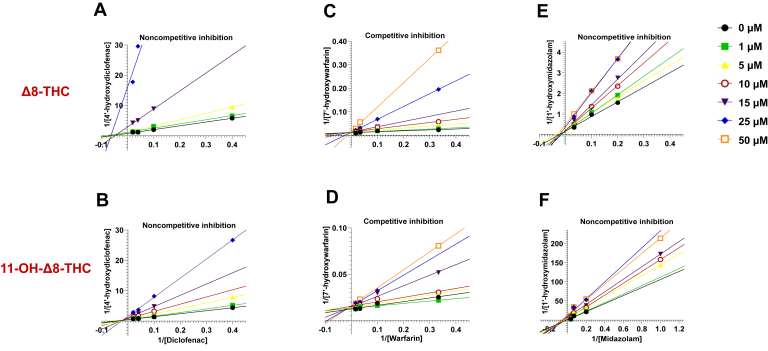
Unbound inhibition constants () were calculated using both recombinant P450-overexpressing microsomes and HLM. Δ8-THC demonstrated potent inhibition of CYP2C9, with values of 0.26 ± 0.01 μM (recombinant CYP2C9) and 0.32 ± 0.06 μM (HLM) for diclofenac 4′-hydroxylation, and even lower values observed for warfarin metabolism (0.16 ± 0.04 μM and 0.23 ± 0.09 μM for recombinant CYP2C9 and HLM, respectively; Table 3). CYP3A4 inhibition was also strong, with a of 0.52 ± 0.02 μM. 11-OH-Δ8-THC exhibited generally weaker inhibition across all reactions, with values ranging from 0.40 ± 0.08 μM to 1.20 ± 0.07 μM. These data indicate that Δ8-THC may pose a significant drug interaction risk with CYP2C9 substrates such as warfarin and diclofenac and possibly with CYP3A4 substrates like midazolam.
Table 3: The unbound of Δ8-THC and 11-OH-Δ8-THC against CYPs 2C9 and 3A4
| Cannabinoid | Reaction | Microsomes | a, b (μM) | Inhibition Typec |
|---|---|---|---|---|
| Δ8-THC | 4′-Hydroxylation of diclofenac | rec CYP2C9HLM | 0.26 ± 0.010.32 ± 0.06 | N |
| 7′-Hydroxylation of warfarin | rec CYP2C9HLM | 0.09 ± 0.040.23 ± 0.09 | C | |
| 1′-Hydroxylation of midazolam | rec CYP3A4HLM | 0.86 ± 0.280.52 ± 0.02 | N | |
| 11-OH- Δ8-THC | 4′-Hydroxylation of diclofenac | rec CYP2C9HLM | 0.73 ± 0.040.77 ± 0.04 | N |
| 7′-Hydroxylation of warfarin | rec CYP2C9HLM | 0.40 ± 0.160.40 ± 0.08 | C | |
| 1′-Hydroxylation of midazolam | rec CYP3A4HLM | 0.89 ± 0.201.20 ± 0.07 | N |
rec, recombinant.
To evaluate the potential clinical impact of Δ8-THC on DDI, static models were used to predict the AUCR.ref. bib29 Due to their structural and pharmacokinetic similarities, Δ8-THC and 11-OH-Δ8-THC were assumed to share equivalent values with Δ9-THC and 11-OH-Δ9-THC at matched inhalation doses, respectively.ref. bib14,ref. bib36
values were used to calculate AUCR, and for orally administered agents, a predicted AUCR ≥ 1.25 indicates potential for significant presystemic DDIs. In the present model, DDI risk increased in a dose-dependent manner (Table 4). At a low Δ8-THC dose (10 mg), predicted AUCRs were 2.29 (diclofenac), 1.61 (warfarin), and 2.34 (midazolam), whereas at a medium Δ8-THC dose (20 mg), AUCRs rose to 3.04 (diclofenac), 2.14 (warfarin), and 2.92 (midazolam), respectively. At a relatively high dose (40 mg), AUCRs further increased to 4.48 (diclofenac), 3.05 (warfarin), and 3.94 (midazolam). For 11-OH-Δ8-THC, predicted AUCRs were comparatively lower than that of Δ8-THC. They were 1.79 (diclofenac), 1.35 (warfarin), and 1.89 (midazolam) at a low Δ8-THC dose (10 mg), 2.12 (diclofenac), 1.66 (warfarin), and 2.23 (midazolam) at a medium Δ8-THC dose (20 mg), and 2.72 (diclofenac), 2.23 (warfarin), and 2.73 (midazolam) at a relatively high Δ8-THC dose (40 mg).
Table 4: The predicted area-under-the-curve ratio (AUCR) for P450-mediated Δ8-THC drug interactions using static models
| Cannabinoid | Route of Administration | Dose (mg) | a (μM) | Predicted AUCRbc | ||
|---|---|---|---|---|---|---|
| CYP2C9 | CYP3A4 | |||||
| Diclofenac | Warfarin | Midazolam | ||||
| Δ8-THC | Oral | 10 | 0.0079 | 1.61 | 2.29 | 2.34 |
| 20 | 0.0178 | 2.14 | 3.04 | 2.92 | ||
| 40 | 0.0480 | 3.05 | 4.48 | 3.94 | ||
| Inhalation | 25 | 0.2500 | 1.02 | 1.08 | 1.01 | |
| 70 | 0.7000 | 1.07 | 1.23 | 1.04 | ||
| 100 | 0.9900 | 1.09 | 1.32 | 1.06 | ||
| 11-OH-Δ8-THC | Oral | 10 | 0.0150 | 1.35 | 1.79 | 1.89 |
| 20 | 0.1000 | 1.66 | 2.12 | 2.23 | ||
| 40 | 0.1200 | 2.23 | 2.72 | 2.73 | ||
| Inhalation | 25 | 0.0120 | 1.00 | 1.00 | 1.00 | |
| 70 | 0.0240 | 1.00 | 1.00 | 1.00 | ||
| 100 | 0.0360 | 1.00 | 1.00 | 1.00 | ||
a The inhalation doses and Cmax′s used for AUCR prediction are the inhalation doses and Cmax′s described for Δ9-THC.14,34,36 The oral doses and Cmax′s used for AUCR prediction are the oral doses and Cmax′s for Δ8-THC.35
For inhalation exposure, a predicted AUCR ≥ 1.02 indicates potential for systemic DDI.ref. bib29 At low (25 mg) and moderate (54 mg) Δ8-THC inhalation doses, AUCRs for midazolam remained near 1.00 (Table 4).ref. bib14,ref. bib29,ref. 34, ref. 35, ref. 36 At a high inhalation dose (70 mg), AUCRs increased slightly to 1.07 (diclofenac), 1.23 (warfarin), and 1.04 (midazolam). However, at a very high dose (100 mg), values reached 1.09 (diclofenac), 1.32 (warfarin), and 1.06 (midazolam). For 11-OH-Δ8-THC, AUCRs remained at 1.00 across all inhalation doses tested.
Discussion
This is the first study to investigate the potential inhibitory effects of Δ8-THC on hepatic P450-mediated drug metabolism. Results from this study showed that Δ8-THC and its active metabolite, 11-OH-Δ8-THC, inhibited the activities of CYPs 2C9 and 3A4 in both recombinant P450-overexpressing systems and in HLM. Minimal or no inhibition of enzyme activity was observed for other major hepatic P450 enzymes tested in this study. Interestingly, although this inhibition was observed for both substrates tested for CYP2C9 (diclofenac and warfarin), this inhibition appeared to be substrate specific for CYP3A4, with inhibition observed for midazolam but not testosterone. CYP3A4 is known for its large and flexible active site that accommodates a wide variety of substrates. Both midazolam and testosterone bind in the same active site,ref. bib37 but their binding orientations differ, with midazolam having a more flexible binding mode, whereas testosterone’s steroid structure results in a more rigid fit.ref. bib37 Although midazolam binds near the heme group within the CYP3A4 molecule,ref. bib38 interacting with key residues such as Phe108, Phe304, Ile120, and Ala305,ref. bib39 the key interacting residues between testosterone and CYP3A4 are different and include Ser119, Ile301, Ala305, and Leu373.ref. bib40
Interestingly, Δ8-THC and 11-OH-Δ8-THC were competitive inhibitors for CYP2C9-mediated 7′-hydroxylation of warfarin but noncompetitive inhibitors for 4′-hydrxolation of diclofenac suggesting that Δ8-THC and 11-OH-Δ8-THC share the same binding pocket with warfarin but not for diclofenac on the CYP2C9 protein. These differences in inhibition type could potentially be explained by the fact that both warfarin and diclofenac interact with hydrophobic sites within the CYP2C9 enzyme, but this interaction is with different amino acid residues.ref. bib41 Further structural studies examining Δ8-THC binding within the CYP2C9 and CYP3A4 molecules will be necessary to better assess the structural mechanisms underlying these interactions.
Given that Δ8-THC shares high structural similarity with Δ9-THC, similar inhibition profiles were expected for major hepatic P450 enzymes. Previous in vitro studies showed that Δ9-THC inhibited the activities of multiple P450 enzymes including CYPs 1A2 ( = 0.090 ± 0.027 μM for recombinant CYP1A2 and 0.10 ± 0.056 μM for HLM), 2B6 ( = 0.25 ± 0.043 μM for recombinant CYP2B6 and 0.38 ± 0.029 μM for HLM), 2C9 ( = 0.073 ± 0.023 μM for recombinant CYP2C9 and 0.17 ± 0.046 μM for HLM), 2C19 ( = 0.056 ± 0.018 μM for recombinant CYP2C19 and 0.21 ± 0.082 μM for HLM), and 2D6 ( = 0.11 ± 0.015 μM for recombinant CYP2D6 and 0.28 ± 0.030 μM for HLM).ref. bib13 Similarly, the active Δ9-THC metabolite, 11-OH-Δ9-THC, also inhibited CYPs 2B6 ( = 0.086 ± 0.066 μM for recombinant CYP2B6 and 0.26 ± 0.041 μM for HLM), 2C9 ( = 0.057 ± 0.044 μM for recombinant CYP2C9 and 0.21 ± 0.032 μM for HLM), and 2D6 ( = 0.15 ± 0.067 μM for recombinant CYP2D6 and 0.32 ± 0.24 μM for HLM).ref. bib13 In contrast, Δ8-THC and 11-OH-Δ8-THC exhibited a relatively narrow profile of cytochrome P450 inhibition, targeting 2 major hepatic enzymes, CYPs 2C9 and 3A4. Similar to that observed previously for Δ9-THC-COOH,ref. bib13 Δ8-THC-COOH exhibited no inhibition of any major hepatic P450 enzymes. It is important to note that both Δ8-THC and 11-OH-Δ8-THC exhibited an inhibitory effect on CYP3A4-mediated 1′-hydroxylation of midazolam—a result not observed previously with Δ9-THC and 11-OH-Δ9-THC.ref. bib13 In addition, Δ8-THC and Δ9-THC differ in their mechanisms of inhibition. Δ9-THC and 11-OH-Δ9-THC act as competitive inhibitors of CYP2C9-mediated 4′-hydroxylation of diclofenac, whereas Δ8-THC and 11-OH-Δ8-THC inhibit the same pathway noncompetitively. Variations in the inhibition profiles and mechanisms of Δ8-THC and Δ9-THC imply that the slight structural shift between them—namely, the position of the double bond in the cyclohexene ring—may alter their binding affinity for P450 enzymes, and further identification of the key Δ8-THC-enzyme interaction sites will be essential to understanding its inhibitory effects and metabolic behavior.
Diclofenac is a commonly used nonsteroidal anti-inflammatory drug, metabolized primarily by CYP2C9.ref. bib42 Previous in vitro studies suggested that the inhibition of CYP2C9-mediated 4′-hydroxylation of diclofenac by Δ9-THC and 11-OH-Δ9-THC can reduce diclofenac’s clearance, leading to ≥ 44% increase in systematic exposure after a 40 mg oral dose of Δ9-THC based on mechanistic static modeling.ref. bib13 Similar data were observed for Δ8-THC and 11-OH-Δ8-THC in the present study as were observed for Δ9-THC and 11-OH-Δ9-THC in these previous studies. Static modeling performed in the present study predicted strong DDI risks with oral Δ8-THC for diclofenac; inhaled Δ8-THC posed minimal risk at typical usage levels. The inhibition of CYP2C9-mediated 4′-hydroxylation of diclofenac observed with Δ8-THC in the present study were predicted to resulting in a ≥ 119% increase in systematic exposure after a 10 mg oral dose of Δ8-THC, an effect that becomes more dramatic at higher Δ8-THC doses including a 348% increase after a 40 mg oral dose of Δ8-THC. Another study suggested a > 560% increase in systematic exposure after a 130 mg oral dose of Δ9-THC based on mechanistic static modeling,ref. bib43 which is similar to our results. Elevated plasma levels of diclofenac might induce side effects such as gastrointestinal bleeding, renal impairment, and cardiovascular issues.ref. bib44
Warfarin is a commonly prescribed anticoagulant and, like diclofenac, is also predominantly metabolized by CYP2C9.ref. bib45 Previous in vitro studies indicated that both Δ9-THC and 11-OH-Δ9-THC can inhibit CYP2C9-mediated 7′-hydroxylation of warfarin, potentially increasing systemic exposure and elevating the risk of adverse effects.ref. bib46 Similar inhibitory potency ( values) was observed in the present study for Δ8-THC and 11-OH-Δ8-THC, consistent with findings for Δ9-THC and its metabolite, 11-OH-Δ9-THC. Static modeling performed in the current study predicted a strong DDI risk between warfarin and orally administered Δ8-THC. The predicted AUCR suggests at least a 61% increase in systemic warfarin exposure after a 10 mg oral dose of Δ8-THC, with even greater increases at higher doses including a 205% increase after 40 mg oral dose of Δ8-THC. Given warfarin’s narrow therapeutic window, elevated plasma levels may lead to serious adverse outcomes such as excessive bleeding or, paradoxically, clotting due to unstable anticoagulation.ref. bib47 Clinical case reports have documented increased bleeding risks in patients co-using cannabis products, further supporting the predicted interaction between Δ8-THC and warfarin.ref. bib48
Midazolam is a widely used benzodiazepine for sedation and is primarily metabolized by CYP3A4.ref. bib39 Previous in vitro studies reported that CBD inhibits CYP3A4-mediated 1′-hydroxylation of midazolam ( = 0.093 ± 0.037 μM for recombinant CYP3A4 and 0.22 ± 0.044 μM for HLM), resulting in a > 8-fold increase in systemic exposure following an 800 mg oral dose of CBD, as predicted by mechanistic static modeling.ref. bib13 Although Δ9-THC does not inhibit CYP3A4, the present study found that both Δ8-THC and 11-OH-Δ8-THC inhibit CYP3A4-mediated 1′-hydroxylation of midazolam. Static modeling predicted at least a 134% increase in systemic midazolam exposure after a 10 mg oral dose of Δ8-THC, with higher doses leading to more pronounced effects including a 294% increase after a 40 mg oral dose of Δ8-THC. Accumulation of midazolam may result in prolonged sedation and increased risk of respiratory depression. Clinical studies have shown that cannabis use is associated with heightened sedation and greater need for adjunct sedatives, supporting the potential for a clinically significant Δ8-THC–midazolam interaction.ref. bib49
There were several limitations with the present study. Unlike Δ9-THC, information about the disposition and pharmacokinetic profile of Δ8-THC is extremely limited. To predict DDIs, values for Δ9-THC inhalation were used in the Δ8-THC inhalation model, which may cause inaccuracies in predictions for its clinical impact. Although previous studies suggest that Δ8-THC and Δ9-THC have similar metabolism pathways and pharmacokinetic profiles, Δ8-THC is metabolized slower in the liver.ref. bib50 One clinical study found that Δ8-THC has a higher inhalation than that of Δ9-THC.ref. bib35,ref. bib51 Therefore, the AUCRs for inhalation dose calculated in the present study may be underestimating the impact of Δ8-THC inhalation for a given DDI.
Static models for drug-metabolizing enzymes reflect worst-case scenario predictions assuming constant enzyme activity and drug exposure. Predictions using physiologically based pharmacokinetic models, which integrate multiple dynamic processes, such as drug transporter-mediated enterohepatic uptake, renal clearance,ref. bib52 drug transporter-mediated intestinal absorption,ref. bib53,ref. bib54 and intestinal metabolismref. bib55 within a single cohesive system, should be developed to improve predictions for DDI between Δ8-THC and agents metabolized by CYPs 3A4 and 2C9. In addition to DDI predictions in healthy subjects, the ability to predict DDI in pediatric,ref. bib56 geriatric,ref. bib57 pregnancy,ref. bib58 and other special populations is a promising advantage for using physiologically based pharmacokinetic modeling over static DDI models.
Taken together, this study provides the first compelling in vitro evidence that Δ8-THC and its active metabolite, 11-OH-Δ8-THC, inhibit the key hepatic enzymes CYP2C9 and CYP3A4, indicating a significant potential for DDI with widely used medications such as diclofenac, warfarin, and midazolam.
Conflict of interest
The authors declare no conflicts of interest. Dr Shelby Coates is currently employed by Pfizer; however, her employment had no influence on the design, conduct, or reporting of this study. The remaining authors have no financial or personal relationships with commercial entities that could have influenced the work presented in this manuscript.
References
- S. Atalay, I. Jarocka-Karpowicz, E. Skrzydlewska. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants (Basel), 2019. [DOI | PubMed]
- Q. Wang, Z. Qin, X. Xing, H. Zhu, Z. Jia. Prevalence of cannabis use around the world: a systematic review and meta-analysis, 2000-2024. China CDC Wkly, 2024. [DOI | PubMed]
- K. Bardhi, S. Coates, C.J.W. Watson, P. Lazarus. Cannabinoids and drug metabolizing enzymes: potential for drug-drug interactions and implications for drug safety and efficacy. Expert Rev Clin Pharmacol, 2022. [DOI | PubMed]
- J.S. Kruger, D.J. Kruger. Delta-8-THC: delta-9-THC’s nicer younger sibling?. J Cannabis Res, 2022. [DOI | PubMed]
- E.C. Leas, A.L. Nobles, Y. Shi, E. Hendrickson. Public interest in Δ(8)-tetrahydrocannabinol (delta-8-THC) increased in US states that restricted Δ(9)-tetrahydrocannabinol (delta-9-THC) use. Int J Drug Policy, 2022
- G.A. Dubrow, R.S. Pawar, C. Srigley. A survey of cannabinoids and toxic elements in hemp-derived products from the United States marketplace. J Food Compost Anal, 2021. [DOI]
- S. Babalonis, W.M. Raup-Konsavage, P.D. Akpunonu, A. Balla, K.E. Vrana. Delta(8)-THC: legal status, widespread availability, and safety concerns. Cannabis Cannabinoid Res, 2021. [DOI | PubMed]
- A.F. Harlow, R.A. Miech, A.M. Leventhal. Adolescent Delta8-THC and marijuana use in the US. JAMA, 2024. [PubMed]
- J.M. Whitehill, K.E. Dunn, R.M. Johnson. The public health challenge of Delta8-THC and derived psychoactive cannabis products. JAMA, 2024. [DOI | PubMed]
- A.M. McDonnell, C.H. Dang. Basic review of the cytochrome P450 system. J Adv Pract Oncol, 2013. [DOI | PubMed]
- K.U. Petersen, W. Schmalix, M. Pesic, T. Stohr. Drug-drug interaction potential of remimazolam: CYP 450, transporters, and protein binding. Curr Drug Metab, 2024. [DOI | PubMed]
- K Ponraj, KA Gaither, D Kumar Singh. Non-additivity of the functional properties of individual P450 species and its manifestation in the effects of alcohol consumption on the metabolism of ketamine and amitriptyline. Biochem Pharmacol, 2024. [PubMed]
- S. Nasrin, C.J.W. Watson, Y.X. Perez-Paramo, P. Lazarus. Cannabinoid metabolites as inhibitors of major hepatic CYP450 enzymes, with implications for cannabis-drug interactions. Drug Metab Dispos, 2021. [DOI | PubMed]
- S. Bansal, N. Maharao, M.F. Paine, J.D. Unadkat. Predicting the potential for cannabinoids to precipitate pharmacokinetic drug interactions via reversible inhibition or inactivation of major cytochromes P450. Drug Metab Dispos, 2020. [DOI | PubMed]
- A.R. Keimowitz, B.R. Martin, R.K. Razdan, P.J. Crocker, S.W. Mascarella, B.F. Thomas. QSAR analysis of Delta(8)-THC analogues: relationship of side-chain conformation to cannabinoid receptor affinity and pharmacological potency. J Med Chem, 2000. [DOI | PubMed]
- S.M. Stout, N.M. Cimino. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev, 2014. [DOI | PubMed]
- S. Valiveti, D.C. Hammell, D.C. Earles, A.L. Stinchcomb. LC-MS method for the estimation of delta8-THC and 11-nor-delta8-THC-9-COOH in plasma. J Pharm Biomed Anal, 2005. [DOI | PubMed]
- K. Watanabe, I. Yamamoto, K. Oguri, H. Yoshimura. Metabolic disposition of delta 8-tetrahydrocannabinol and its active metabolites, 11-hydroxy-delta 8-tetrahydrocannabinol and 11-oxo-delta 8-tetrahydrocannabinol, in mice. Drug Metab Dispos, 1981. [DOI | PubMed]
- K Bardhi, S Coates, M Zhao, MC Anyachebelu, P Lazarus. Identification of human hepatic UDP-glucuronosyltransferases involved in the glucuronidation of temazepam. Drug Metab Dispos, 2025. [PubMed]
- K.C. Patki, L.L. Von Moltke, D.J. Greenblatt. In vitro metabolism of midazolam, triazolam, nifedipine, and testosterone by human liver microsomes and recombinant cytochromes p450: role of cyp3a4 and cyp3a5. Drug Metab Dispos, 2003. [DOI | PubMed]
- S.M. Abdel-Rahman, K. Marcucci, T. Boge, R.R. Gotschall, G.L. Kearns, J.S. Leeder. Potent inhibition of cytochrome P-450 2D6-mediated dextromethorphan O-demethylation by terbinafine. Drug Metab Dispos, 1999. [DOI | PubMed]
- T.J. Yang, Y. Sai, K.W. Krausz, F.J. Gonzalez, H.V. Gelboin. Inhibitory monoclonal antibodies to human cytochrome P450 1A2: analysis of phenacetin O-deethylation in human liver. Pharmacogenetics, 1998. [DOI | PubMed]
- V. Carriere, T. Goasduff, D. Ratanasavanh. Both cytochromes P450 2E1 and 1A1 are involved in the metabolism of chlorzoxazone. Chem Res Toxicol, 1993. [DOI | PubMed]
- T. Andersson, J.O. Miners, M.E. Veronese. Identification of human liver cytochrome P450 isoforms mediating omeprazole metabolism. Br J Clin Pharmacol, 1993. [DOI | PubMed]
- R. Coles, E.D. Kharasch. Stereoselective metabolism of bupropion by cytochrome P4502B6 (CYP2B6) and human liver microsomes. Pharm Res, 2008. [DOI | PubMed]
- R. Bort, K. Mace, A. Boobis, M.J. Gomez-Lechon, A. Pfeifer, J. Castell. Hepatic metabolism of diclofenac: role of human CYP in the minor oxidative pathways. Biochem Pharmacol, 1999. [DOI | PubMed]
- S.Y. Kim, J.Y. Kang, J.H. Hartman. Metabolism of R- and S-warfarin by CYP2C19 into four hydroxywarfarins. Drug Metab Lett, 2012. [DOI | PubMed]
- A. Ghosal, H. Satoh, P.E. Thomas, E. Bush, D. Moore. Inhibition and kinetics of cytochrome P4503A activity in microsomes from rat, human, and cdna-expressed human cytochrome P450. Drug Metab Dispos, 1996. [DOI | PubMed]
- 29ICHM12 Drug Interaction Studies2024ICH-Multidisciplinary5052https://database.ich.org/sites/default/files/ICH_M12_Step4_Guideline_2024_0521_0.pdfAccessed June 2025
- K.R. Yeo. Quantification of the effects of investigational drugs as victims or perpetrators of cypmediated drug interactions involving inhibition in the Simcyp simulator, 2022
- R.S. Tanna, D.D. Tian, N.B. Cech. Refined prediction of pharmacokinetic kratom-drug interactions: time-dependent inhibition considerations. J Pharmacol Exp Ther, 2021. [DOI | PubMed]
- S. Jin, M.N. Paludetto, M. Kurkela. In vitro assessment of inhibitory effects of kinase inhibitors on CYP2C9, 3A and 1A2: prediction of drug-drug interaction risk with warfarin and direct oral anticoagulants. Eur J Pharm Sci, 2024. [DOI]
- E.R. Garrett, C.A. Hunt. Physiochemical properties, solubility, and protein binding of delta9-tetrahydrocannabinol. J Pharm Sci, 1974. [DOI | PubMed]
- S Coates, K Bardhi, M Zhao, P Lazarus. UGT2B7 mediated drug-drug interaction between cannabinoids and hydromorphone. Drug Metabolism and Disposition, 2025. [DOI]
- C.A. Zamarripa, T.R. Spindle, D. Schriefer. A within-subject cross-over trial comparing the acute effects of oral delta-8-tetrahydrocannabinol and delta-9-tetrahydrocannabinol in healthy adults. Drug Alcohol Depend, 2025. [DOI]
- K. Bardhi, S. Coates, G. Chen, P. Lazarus. Cannabinoid-induced stereoselective inhibition of R-S-oxazepam glucuronidation: cannabinoid-oxazepam drug interactions. Pharmaceutics, 2024. [DOI | PubMed]
- S.E. Kandel, L.W. Han, Q. Mao, J.N. Lampe. Digging deeper into CYP3A testosterone metabolism: kinetic, regioselectivity, and stereoselectivity differences between CYP3A4/5 and CYP3A7. Drug Metab Dispos, 2017. [DOI | PubMed]
- S. Liu, Q. Zheng, F. Bai. Differences of atomic-level interactions between midazolam and two CYP isoforms 3A4 and 3A5. Molecules, 2023. [DOI | PubMed]
- K.K. Khan, Y.Q. He, T.L. Domanski, J.R. Halpert. Midazolam oxidation by cytochrome P450 3A4 and active-site mutants: an evaluation of multiple binding sites and of the metabolic pathway that leads to enzyme inactivation. Mol Pharmacol, 2002. [DOI | PubMed]
- T. Tanaka, T. Okuda, Y. Yamamoto. Characterization of the CYP3A4 active site by homology modeling. Chem Pharm Bull (Tokyo), 2004. [DOI | PubMed]
- G. Tai, L.J. Dickmann, N. Matovic, J.J. DeVoss, E.M. Gillam, A.E. Rettie. Re-engineering of CYP2C9 to probe acid-base substrate selectivity. Drug Metab Dispos, 2008. [DOI | PubMed]
- C.L. Crespi, T.K. Chang, D.J. Waxman. Determination of CYP2C9-catalyzed diclofenac 4′-hydroxylation by high-performance liquid chromatography. Methods Mol Biol, 2006. [DOI | PubMed]
- S. Bansal, M.F. Paine, J.D. Unadkat. Comprehensive predictions of cytochrome P450 (P450)-mediated in vivo cannabinoid-drug interactions based on reversible and time-dependent p450 inhibition in human liver microsomes. Drug Metab Dispos, 2022. [DOI | PubMed]
- M. Rorarius, J. Miralles, G.A. Baer, E. Palomaki. Diclofenac versus indomethacin given as intravenous infusions: their effect on haemodynamics and bleeding time, and side-effects in healthy subjects. Ann Clin Res, 1985. [PubMed]
- T. Inoue, K. Nitta, K. Sugihara, T. Horie, S. Kitamura, S. Ohta. CYP2C9-catalyzed metabolism of S-warfarin to 7-hydroxywarfarin in vivo and in vitro in chimeric mice with humanized liver. Drug Metab Dispos, 2008. [DOI | PubMed]
- S. Yamaori, K. Koeda, M. Kushihara, Y. Hada, I. Yamamoto, K. Watanabe. Comparison in the in vitro inhibitory effects of major phytocannabinoids and polycyclic aromatic hydrocarbons contained in marijuana smoke on cytochrome P450 2C9 activity. Drug Metab Pharmacokinet, 2012. [DOI | PubMed]
- A. Hsu, N.A. Painter. Probable interaction between warfarin and inhaled and oral administration of cannabis. J Pharm Pract, 2020. [DOI | PubMed]
- P. Damkier, D. Lassen, M.M.H. Christensen, K.G. Madsen, M. Hellfritzsch, A. Pottegard. Interaction between warfarin and cannabis. Basic Clin Pharmacol Toxicol, 2019. [DOI | PubMed]
- J. Kosirog, C. Bouvette, J. Pannu, J. Gondal, M. Madhoun. Marijuana and endoscopy: the effects of marijuana on sedation. Gastrointest Endosc, 2024. [DOI | PubMed]
- K. Watanabe, S. Yamaori, T. Funahashi, T. Kimura, I. Yamamoto. Cytochrome P450 enzymes involved in the metabolism of tetrahydrocannabinols and cannabinol by human hepatic microsomes. Life Sci, 2007. [DOI | PubMed]
- M. Tagen, L.E. Klumpers. Review of delta-8-tetrahydrocannabinol (Delta(8) -THC): comparative pharmacology with Delta(9)-THC. Br J Pharmacol, 2022. [DOI | PubMed]
- A.S. Ailabouni, D.K. Singh, A. Thakur. Quantitative contributions of hepatic and renal organic cation transporters to the clinical pharmacokinetic cimetidine-metformin interaction. Clin Pharmacol Ther, 2025. [DOI | PubMed]
- K. Kharve, A.S. Engley, M.F. Paine, J.A. Sprowl. Impact of drug-mediated inhibition of intestinal transporters on nutrient and endogenous substrate disposition…an afterthought?. Pharmaceutics, 2024. [DOI | PubMed]
- J.A. Sprowl, A. Sparreboom. Drug trafficking: recent advances in therapeutics and disease. Clin Pharmacol Ther, 2012. [DOI | PubMed]
- M.F. Paine, M. Khalighi, J.M. Fisher. Characterization of interintestinal and intraintestinal variations in human CYP3A-dependent metabolism. J Pharmacol Exp Ther, 1997. [DOI | PubMed]
- J.S. Leeder, J.C. Dinh, A. Gaedigk, V.S. Staggs, B. Prasad, R.E. Pearce. Ontogeny of scaling factors for pediatric physiology-based pharmacokinetic modeling and simulation: microsomal protein per gram of liver. Drug Metab Dispos, 2022. [DOI | PubMed]
- R.S. Foti. Utility of physiologically based pharmacokinetic modeling in predicting and characterizing clinical drug interactions. Drug Metab Dispos, 2025. [DOI]
- A.R. Kumar, L.S. Benson, E.M. Wymore. Quantification and prediction of human fetal (-)-Δ9-THC/(±)-11-OH-Δ9-THC exposure during pregnancy to inform fetal cannabis toxicity. Nat Commun, 2025. [DOI | PubMed]
