Cannabidiol suppresses proliferation and induces cell death, autophagy and senescence in human cholangiocarcinoma cells via the PI3K/AKT/mTOR pathway
Abstract
Background and aim:
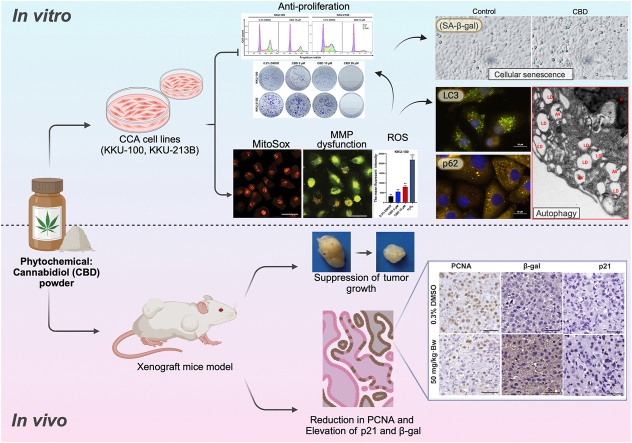
Cholangiocarcinoma (CCA) is usually diagnosed at a late stage, leading to treatment failure. Cannabidiol (CBD), exhibits diverse anti-cancer effects in various cancers, offering avenues for improving CCA treatment. This study investigated the effects of CBD on human CCA cells and the underlying mechanisms in vitro and in vivo.
Experimental procedure:
The effects of CBD on three CCA cell lines (KKU-213B, KKU-100, KKU-055) were assessed using the SRB assay, clonogenic assay, cell cycle arrest, and 3D holotomography. Morphological changes were examined using transmission electron microscopy, while mitochondrial ROS levels and mitochondrial membrane potential were studied using MitoSOX, JC-1, and DCFH-DA. Cellular senescence induction was evaluated via SA-β-gal staining. Protein associatedwith autophagy and cellular senescence were analyzed using Western blot and/or immunofluorescent assays. A xenograft model demonstrated the anti-tumor activity of CBD and the induction of cellular senescence through immunohistochemistry targeting PCNA, β-gal, and p21.
Results and conclusion:
CBD effectively inhibited CCA cell proliferation, suppressed colony formation and induced G0/G1 phase cell cycle arrest. Morphological examination revealed lipid droplets/vesicles in CCA cell lines. CBD induced autophagy by upregulating LC3BII, downregulating p62, and inhibiting the p-PI3K, p-AKT, and p-mTOR pathways. Additionally, CBD disrupted mitochondrial homeostasis by elevating ROS, reducing membrane potential, and induced cellular senescence by increasing the expression of p53 and p21. In-vitro results were confirmed by xenograft models. Overall, CBD suppresses proliferation and induces cell death, autophagy and senescence in CCA cells via the PI3K/AKT/mTOR pathway, which indicates a therapeutic option for CCA treatment.
Article type: Research Article
Keywords: Cholangiocarcinoma, Cannabidiol, Cannabis, Autophagy, Cellular senescence, Cell cycle arrest
Affiliations: Biomedical Sciences Program, Graduate School, Khon Kaen University, Khon Kaen 40002, Thailand; Cholangiocarcinoma Research Institute, Faculty of Medicine, Khon Kaen University, Khon Kaen 40002, Thailand; Department of Pathology, Faculty of Medicine, Khon Kaen University, Khon Kaen 40002, Thailand; Department of Parasitology, Faculty of Medicine, Khon Kaen University, Khon Kaen 40002, Thailand; Department of Biochemistry, Faculty of Medicine, Khon Kaen University, Khon Kaen 40002, Thailand; Centre for Research and Development of Medical Diagnostic Laboratories, Faculty of Associated Medical Sciences, Khon Kaen University, Khon Kaen 40002, Thailand; Department of Chemistry, Faculty of Science, Khon Kaen University, Khon Kaen 40002, Thailand
License: © 2024 Center for Food and Biomolecules, National Taiwan University. Production and hosting by Elsevier Taiwan LLC. CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.jtcme.2024.04.007 | PubMed: 39850601 | PMC: PMC11752120
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.0 MB)
Introduction
The incidence of cholangiocarcinoma (CCA), commonly known as bile-duct cancer, is increasing worldwide. There is a high prevalence of CCA in the Greater Mekong subregion, primarily due to infections with liver flukes, notably Opisthorchis viverrini and Clonorchis sinensis. The reported incidence rates of CCA in the region exceed 6 per 100,000 population.ref. bib1 Unfortunately, most CCA patients are diagnosed at an advanced stage, resulting in a poor prognosis, unsatisfactory surgical outcomes, and low survival rates.ref. bib1 Chemotherapy plays a central role in CCA treatment, typically involving drugs such as gemcitabine (GEM), 5-fluorouracil, and cisplatin.ref. bib2 However, the effectiveness of chemotherapy decreases over time due to cancer adaptation.ref. bib3,ref. bib4 Although the combination of GEM with other anti-cancer drugs has shown improved response rates in CCA, it is associated with the risk of adverse effects such as hepatotoxicity, nausea, decreased immunity, diarrhea, and sensory neuropathy.ref. bib5 Therefore, there is an urgent need for alternative therapeutic approaches to improve efficacy of CCA treatment.ref. bib1
Cannabis is derived from a plant scientifically known as Cannabis sativa L.ref. bib6 Throughout history, cannabis has been used for the treatment of various diseases, including multiple sclerosis,ref. bib7 and end-stage cancer.ref. bib8 Numerous cannabis-derived phytochemicals, such as cannabidiol (CBD), delta-9-tetrahydrocannabinol (Δ9-THC), cannabinol (CBN), cannabigerol (CBG), and cannabichromene (CBC), have shown potential therapeutic effects in clinical trials for a variety of diseases, including cancer.ref. bib9 Compelling evidence suggests that CBD can induce cancer-cell death through apoptosis and autophagy in various cancer types.ref. 10, ref. 11, ref. 12
Previous studies have highlighted the critical role of the PI3K/AKT/mTOR pathway in cancer progression, particularly in O. viverrini-associated CCA.ref. bib13 Activation of this pathway elicits several dynamic changes, including cell cycle arrest, apoptosis, autophagy, reduction in mitochondrial membrane potential, and an excessive generation of reactive oxygen species (ROS). These changes contribute to lipid droplet accumulation or vesicle formation.ref. bib14 In addition, it has been shown that inhibiting the PI3K/AKT/mTOR pathway induces cellular senescence in various cancers.ref. bib15 Hence, the use of cannabis-derived phytochemicals, particularly CBD, presents a promising approach to suppressing CCA cells through the modulation of multiple signaling pathways, thereby potentially minimizing undesired side effects.
This study aimed to gain a better understanding of the effects of CBD on CCA and its molecular mechanisms, using three human CCA cell lines (KKU-055, KKU-100, and KKU-213B). It is hypothesized that CBD exerts its anti-CCA effects through a variety of mechanisms, with some involvement of the PI3K/AKT/mTOR. The efficacy of CBD in combating CCA was also validated through in vivo experiments. The results obtained from this study provide compelling evidence supporting the potential inclusion of CBD in clinical trials as a therapeutic option for CCA treatment.
Materials and methods
CCA cell culture and cannabidiol (CBD) preparation
The CCA cell lines (KKU-055, KKU-100, and KKU-213B) were originally established from patients with opisthorchiasis-associated CCA by Sripa et al.ref. bib16 These CCA cell lines were cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 4.5 g/L glucose (Gibco, Grand Island, NY, USA), 10 % fetal bovine serum (FBS; Corning, Woodland, CA,USA), 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco) at 37 °C with 5 % CO2 in a humidified incubator.
The CBD (#02A64217) used in this study was obtained from the Department of Medical Sciences Reference Standards, Bureau of Drug and Narcotic, Department of Medical Sciences, Ministry of Public Health, Nonthaburi, Bangkok, Thailand. The CBD stock solution was prepared by dissolving it in dimethyl sulfoxide (DMSO; PanReac AppliChem ITW Reagents, Darmstadt, Germany) and diluted with complete medium to make 2–20 μM final concentration.
Cell proliferation detection by sulforhodamine B (SRB) assay
The SRB assay was slightly modified as previously described.ref. bib17 Cells (2 × 103 cells/well) were seeded into flat-bottom 96-well plates and treated with various concentrations of CBD for 24, 48 and 72 h. Subsequently, cells were fixed with 40 % cold trichloroacetic acid for 1 h and stained with 0.4 % (w/v) SRB solution in 1 % acetic acid for 1 h. SRB dye was dissolved in 10 mM Tris buffer pH 10.5. Absorbance was measured at 492 nm using an ELISA reader (Tecan group Ltd., Männedorf, Switzerland).
Clonogenicity assay
CCA cells (1 × 103 cells/well) were grown in 6-well plates and treated with various concentrations of CBD. The culture medium was changed every 2 days, and cells were grown for approximately 2 weeks. Subsequently, cells were stained, and absorbance was measured as previously described.ref. bib17
Cell cycle analysis
CCA cells (2 × 105 cells/well) were seeded into 6-well plates and treated with 10 μM CBD for 48 h. Subsequently, cells were fixed with 70 % ethanol and stained with FxCycle™ PI/RNase staining solution as described.ref. bib17 The stained cells were then sorted and analyzed using a BD FACSCanto II flow cytometer (BD Biosciences, San Jose, CA, USA) and FlowJo™ software version 10.8.1 (Becton, Dickinson & Company, OR, USA).
Lipid droplet/vesicle formation
Lipid droplet/vesicle formation was assessed as previously mentioned.ref. bib18 CCA cells (2 × 105 cells/well) were seeded into 6-well plates overnight. Then, cells were treated with 10 μM CBD for 48 h. A Nikon Eclipse TS 100 inverted light microscope (Nikon, Tokyo, Japan) was used to visualize cell morphology.
Optical diffraction tomography (ODT) and fluorescence imaging
To localize the lipid droplet accumulation and vesicle formation, ODT and fluorescence imaging were performed as previously described.ref. bib19 CCA cells were exposed to 10 μM CBD for 48 h and cultured in a TomoDish at a density of 2 × 104 cells per dish. Live cells were imaged using a Mach-Zehnder interferometric microscope HT-2H (Tomocube, Inc., Daejeon, Republic of Korea). Fluorescence imaging was used for additional 3D visualization, reconstructed via deconvolution using TomoStudio software (Tomocube, Inc.).
Transmission electron microscopy (TEM)
CCA cells (2 × 105 cells/well) were seeded in a 6-well plates and treated with 10 μM CBD for 48 h. Cells were processed according to the methods outlined in a previous article.ref. bib20 TEM photographs were obtained using a JEOL JEM 1011 microscope, equipped with a Morada camera (JEOL Ltd., Tokyo, Japan).
Mitochondrial ROS assay
CCA cells (2.5 × 104 cells/well) were seeded onto 8-well chamber slides. After overnight incubation, cells were treated with 10 μM CBD for 48 h. Then, cells were washed with 1X PBS, exposed to 5 μM MitoSOX Red for 30 min at RT, followed by Hoechst 33,342 (#H3570, Invitrogen).ref. bib21 Imaging was conducted using Zeiss LSM880 confocal scanning microscope, capturing ten fields of view/group (20 × magnification). Fluorescence intensity was analyzed using ImageJ software as previously described.ref. bib22
Mitochondrial membrane potential (MMP; ΔΨm) assay
The MMP assay was performed as previously described.ref. bib23 CCA cells were seeded in 96-well plates (1 × 104 cells/well) and treated with 5 and 10 μM CBD for 48 h. A positive control was exposed to 50 μM carbonyl cyanide m-chlorophenyl hydrazone (CCCP) for 10 min. JC-1 dye (200 μM) was added to each well and incubated for 20 min. Confocal images were captured using a Zeiss LSM880 microscope with ten fields per experimental group at 20 × magnification. Fluorescence intensity was quantified using ImageJ software, and mean values were calculated with GraphPad Prism 9.0 (GraphPad Software, La Jolla, CA, USA).
Reactive oxygen species (ROS) detection
CCA cells (2 × 105 cells/well) were seeded into 6-well plates. After overnight incubation, cells were treated with 10 μM CBD for 12–24 h and 1 mM hydrogen peroxide (H2O2) as a positive control for 3 h. Following treatment, cells were incubated with 25 μM 2′,7′-dichlorofluorescein diacetate (DCFH-DA) for 30 min in the dark,ref. bib24 and washed twice with 1X PBS. Subsequently, cells were transferred to a FACS tube, washed twice with 1X PBS, and analyzed for ROS production using a flow cytometer (BD FACSCanto™ II) with data analyzed using BDFACSDiva™ software.
Senescence-associated β-galactosidase (SA- β-gal) staining
The effect of CBD on cellular senescence in CCA cells was investigated by SA-β-gal staining as previously described.ref. bib25 Cells were seeded into 6-well plates and treated with CBD for 48 h, and then stained with senescence β-galactosidase staining kit (#9860, Cell Signaling Technology, Danvers, MA, USA) following the manufacturer’s guidelines. Stained cells were imaged using a light microscope with ten fields per well captured at 20× magnification. Scoring of stained cells was done with the experiment replicated twice independently, each with triplicate technical replicates. Percentage of SA-β-gal-positive cells relative to total cells was computed for quantification.
Immunofluorescence assay (IFA)
CCA cells (2.5 × 104 cells/well) were systematically seeded onto sterile cover slides within 24-well plates and treated with 10 μM CBD for 48 h. Additionally, a subset of cells was pre-treated with chloroquine (CQ) at a concentration of 10 μM, administered 8 h prior to CBD exposure.ref. bib26,ref. bib27 After fixing with 4 % paraformaldehyde and permeabilizing with 0.2 % Triton X-100 (Amresco, Solon, OH, USA), cells were blocked with PBS-5% BSA with 0.1 % Triton X-100 and incubated overnight with primary antibodies against LC3BI/II (1:250, #A7198, ABclonal, Wuhan, China) and p62 (1:250, #A7758, ABclonal). Subsequently, cells underwent incubation with secondary antibodies (1:10, #5230-0298, KPL Affinity Purified Antibody Fluorescein-Labeled Goat Anti-Rabbit IgG (H + L), SeraCare Life Sciences Inc., MA, USA). Concurrently, CellMask Deep Red, a specialized plasma membrane stain (#C10046, Invitrogen), was employed to highlight the cellular plasma membranes. Additionally, Hoechst 33,342 was applied for nuclear counterstaining purposes. Ten fields from images of each experimental group were acquired at a magnification of 100 × using a fluorescence imaging microscope (ECLIPSE Ni–U, Nikon Instruments Inc, Japan). Subsequently, fluorescence-intensity images were analyzed using ImageJ softwareref. bib22 (National Institutes of Health, Bethesda, MD, USA).
Western blotting
Protein extraction was performed using 1X RIPA reagent (#9806S, Cell Signaling Technology) and protein concentrations were determined using Pierce BCA Protein Assay Kit (Thermo Fisher Scientific Inc., Rockford, IL, USA). SDS-PAGE technique was used to separate proteins (20–25 μg).ref. bib27 PVDF membranes were incubated with primary antibodies (dilution 1:1000) for: p62 (#A7758), LC3BI/II (#A7198), p21 (#A19094), and phospho-PI3K (#AP0854), all purchased from Abclonal; Bcl-2 (#ab7973) and β-actin (#ab3280), both purchased from Abcam; phospho-mTOR (Ser2448) (#5536), phospho-AKT (Ser473) (#2983), PI3K (#42925), AKT (#92725) and mTOR (#2983), all purchased from Cell Signaling Technology, and p53 (#10442-1-AP, from Proteintech, IL, USA). Then, membranes were incubated with secondary antibodies HRP-conjugated goat anti-rabbit IgG (1:2,000, #111035003, Jackson Immuno Research Inc., West Grove, PA, USA).
Xenograft model
KKU-100 cells (1 × 106 cells) were injected subcutaneously into female knockout BALB/c Rag-2.Jak3 double-knockout mice.ref. bib28 After 21 days, mice were treated with either DMSO or 50 mg/kg·Bw of CBD (n = 5) daily for 4 days. Tumor sizes were measured at days 0, 3 and 5 following this, and the tumor volume was calculated using the formula: tumor volume = (length × widthref. bib2)/2.
Immunohistochemistry study
Tumor tissues from mice were immunohistochemically analyzed by antigen retrieval with citrate buffer and Tris-EDTA buffer, blocking with 5 % FBS, and incubation with anti-proliferating cell nuclear antigen (PCNA) antibody (1:750, #Ab2426, Abcam), p21 antibody (1:50, #A19094, Abclonal), and GBL1 antibody (1:40, #A1863, Abclonal) overnight. The signal was detected with diaminobenzidine substrate and counterstained with Mayer’s hematoxylin.ref. bib27 Brown staining indicated positive cells. Ten fields (20 × magnification) were analyzed using ImageJ software.
Statistical analysis
The experiments were conducted in biological triplicate with results presented as mean ± SD. Statistical comparisons involved one-way ANOVA followed by Tukey’s test for multiple group comparisons and Student’s t-test for in vivo investigations with two cohorts.ref. bib29 A significance threshold of p < 0.05 was used. All analyses were performed using GraphPad Prism 9.0 for Mac.
Results
CBD treatment suppresses CCA cell proliferation, induces cell cycle arrest and inhibits colony formation
The SRB assay showed that CCA cell proliferation in all three cell lines decreased following CBD treatment in a dose- and time-dependentmanners (Fig. 1A). The half-maximal inhibitory concentration (IC50) values at 48 h of treatment were 11.34 ± 1.46, 10.62 ± 1.23, and 10.59 ± 1.29 μM for KKU-055, KKU-100 and KKU-213B, respectively. After 48 h of treatment, there were no differences in the IC50 values among the three distinct CCA cell lines. Cell line KKU-055 was not used in the remaining investigations reported below. While, IC50 for MMNK 1 was 12.73 ± 1.09, indicating that CBD has low toxic effect (Supplementary figure 1).
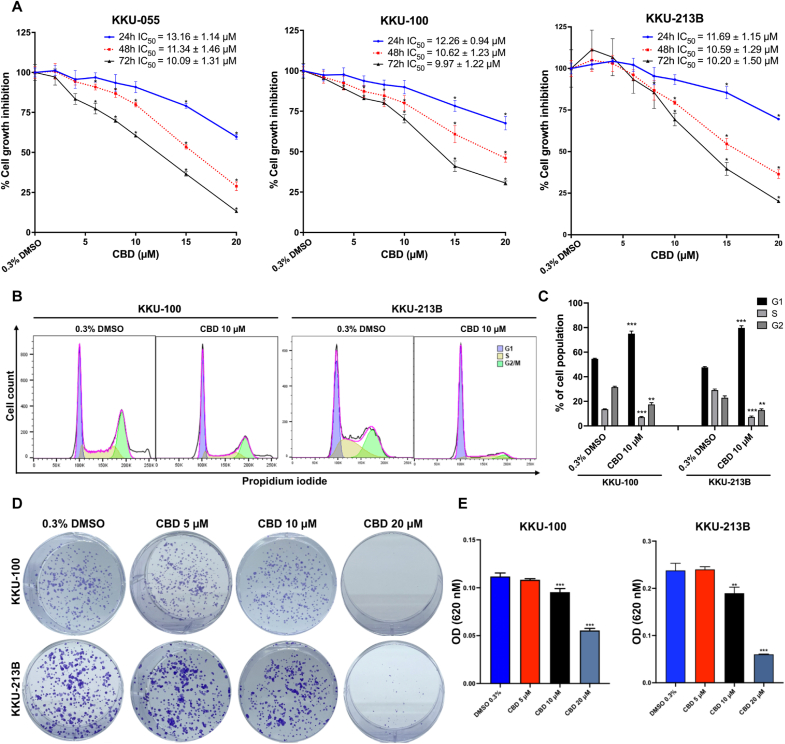
To further investigate the inhibitory effects of CBD on CCA cell proliferation, KKU-100 and KKU-213B cells were treated with 10 μM CBD for 48 h. Flow cytometric analysis revealed a significant increase in the proportion of KKU-100 and KKU-213B cells in the G1 phase (KKU-100; p < 0.001, KKU-213B; p < 0.001) after treatment with 10 μM CBD compared to the 0.3 % DMSO treatment (Fig. 1B and C).
To investigate the inhibitory effect of CBD on the proliferative capacity of a single CCA cell, a clonogenic assay was performed (Fig. 1D). CBD treatment inhibited colony formation in both CCA cell lines (KKU-100 and KKU-213B) in a dose-dependent manner. Treatment with CBD at 10 and 20 μM significantly inhibited colony formation of KKU-100 cells (p < 0.001 and p < 0.001, respectively) and KKU-213 (p < 0.01 and < 0.001, respectively) compared to vehicle control (0.3 % DMSO treatment) (Fig. 1E). Notably, 20 μM CBD exhibited greater efficacy than 10 μM CBD in inhibiting colony formation in both cell lines (Fig. 1D and E).
CBD induces lipid droplet and vesicle formation in CCA cells
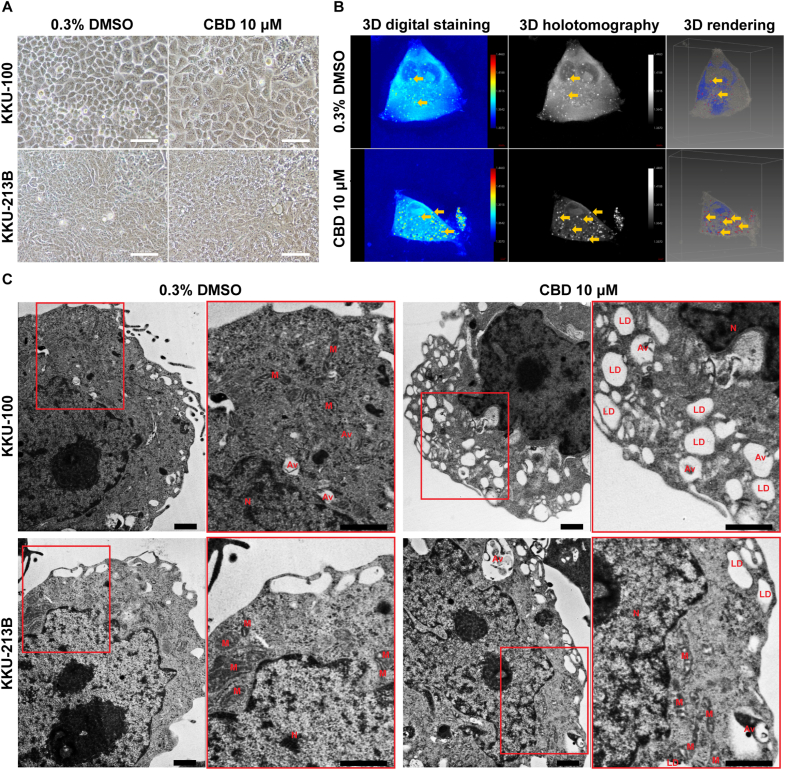
Following a 48-h treatment with 10 μM CBD, KKU-100 CCA cells enlarged and contained various vesicles in the cytoplasm when observed using an inverted light microscope and 3D holotomography (Fig. 2A and B). This was confirmed by the observations obtained from TEM, which showed extensive cytoplasmic vacuolation, nucleolus enlargement, and the presence of autophagosomes (Fig. 2C). In addition, both CCA cell lines exhibited the presence of single- and double-membrane vacuoles.
CBD increases mitochondrial ROS, changes mitochondrial membrane potential (MMP; ΔΨm) and increases ROS production in CCA cells
Fig. 3 demonstrates the effect of CBD on mitochondrial ROS, changes MMP and increases ROS production in CCA cells. Induction of ROS generation in mitochondria was evaluated using MitoSOX Red as a fluorescent probe. Elevated orange-red fluorescence indicated increased mitochondrial ROS levels. Treatment with 10 μM CBD significantly boosted fluorescence intensity in KKU-100 and KKU-213B cell lines (p < 0.0001 in both cases) compared to vehicle control (Fig. 3A–C). Additionally, cells treated with vehicle control (0.3 % DMSO) exhibited a higher proportion of red fluorescence,indicating healthy mitochondria, whereas, cells treated with 50 μM CCCP or CBD displayed a higher proportion of green fluorescence,indicating mitochondrial damage, relative to red fluorescence (Fig. 3D). In CBD treatment, the red/green fluorescence ratio of KKU-100 cells significantly decreased to 1.12 ± 0.07 (p < 0.001) and 0.83 ± 0.08 (p < 0.001) with CBD at concentrations of 5 and 10 μM, respectively, when compared to vehicle control (1.53 ± 0.14). Similarly, the red/green ratio of KKU-213B cells was significantly lower at 1.01 ± 0.12 (p < 0.001) and 0.83 ± 0.06 (p < 0.001) by CBD treatment at concentrations of 5 and 10 μM, respectively, when compared to vehicle control (1.24 ± 0.10) (Fig. 3E).
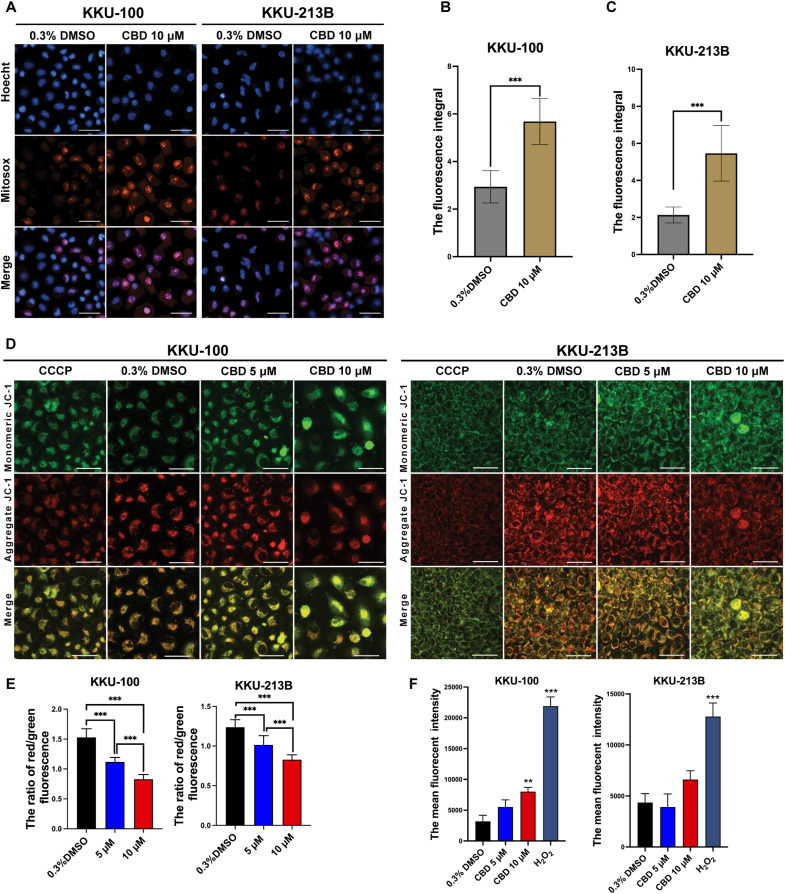
Flow cytometry revealed that KKU-100 CCA cells displayed a slight increase in fluorescence intensity when treated with 5 μM CBD and a significant rise (p < 0.01) with 10 μM CBD after 12 h. This did not occur in KKU-213B cells. As expected, both CCA cell lines gave the highest fluorescence intensity with 1 mM H2O2, the positive ROS control, after 3 h (Fig. 3F).
CBD induces autophagy in CCA cells
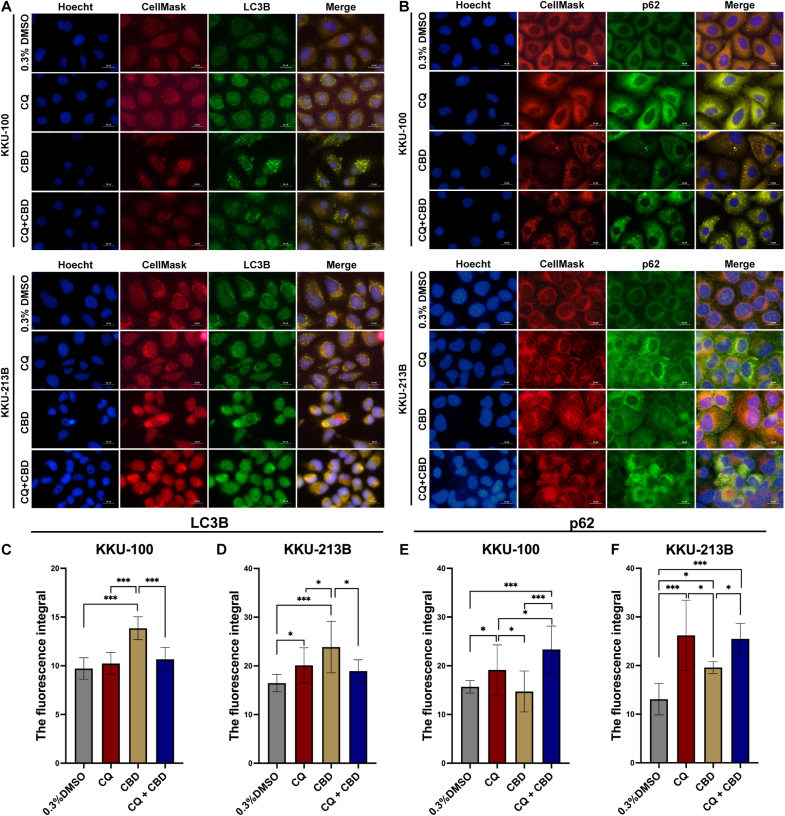
We further examined the impact of co-administration of CBD and CQ, an autophagy inhibitor, on autophagy pathways through IFA (Fig. 4) and Western blot analysis. IFA revealed increased LC3 fluorescence following CBD and CQ co-administration compared to 0.3 % DMSO control (p < 0.0001) (Fig. 4A and 4C-D). CBD treatment also altered p62 fluorescence, with significant effects in KKU-213B (p = 0.025) but not KKU-100 (p = 0.9023) (Fig. 4B and 4E-F). In contrast, CBD treatment led to a significant reduction in fluorescence intensity of p62 compared to CQ alone or the combination of CQ with CBD.
We further confirmed this by Western blot analysis (Fig. 5). CQ alone or with CBD significantly increased LC3B–I and II expression compared to 0.3 % DMSO or CBD treatment alone. CBD at 10 μM notably elevated LC3B-II expression in both cell lines compared to 0.3 % DMSO alone (KKU-100: p = 0.011, KKU-213B: p = 0.049). CBD treatment exhibited the most pronounced LC3B-II/I ratio (p < 0.0001) (Fig. 5A–D). A significantly decreased expression of p62 levels was observed in CCA cell lines with CBD treatment compared to 0.3 % DMSO or CQ alone (Fig. 5A and E). Additionally, CBD treatment reduced Bcl-2 expression levels in a dose-dependent manner. Significant reductions in Bcl-2 expression were observed at various CBD concentrations (KKU-100: 10 μM, p = 0.036; KKU-213B: 5 μM, p = 0.027; 10 μM, p = 0.007) relative to vehicle control (Fig. 5F).
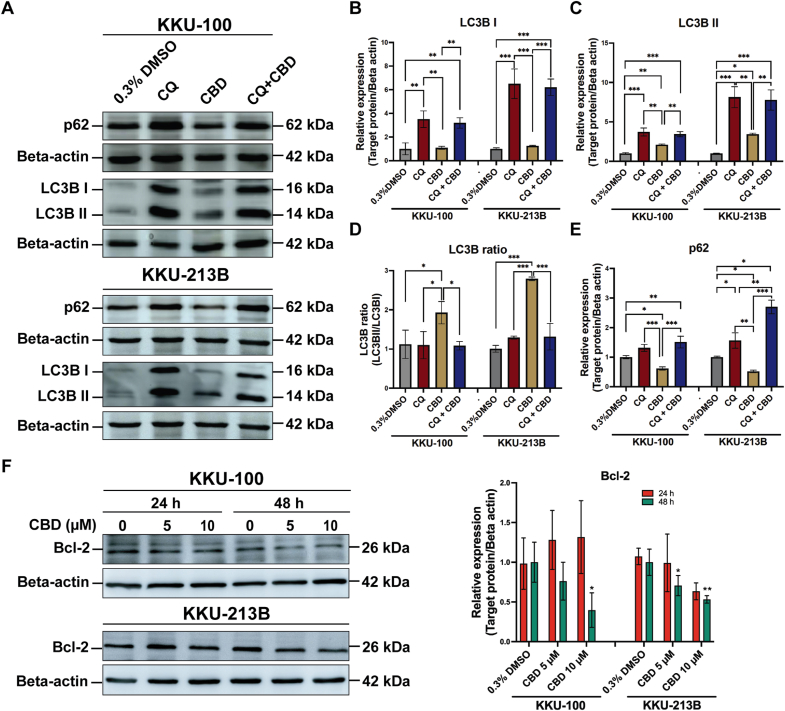
CBD suppresses the expression of the PI3K/Akt/mTOR pathway
To investigate the impact of CBD on the PI3K/Akt/mTOR pathway, Western blot analysis was conducted (Fig. 6). The basal expression of PI3K, Akt and mTOR in KKU-100 and KKU-213B cells did not differ between the control and CBD treatment groups. However, in KKU-100 cells treated with 5 and 10 μM CBD, there was a reduction in the expression of phosphorylated PI3K (p-PI3K), phosphorylated Atk (p-Atk) and phosphorylated mTOR (p-mTOR) at 24 h compared to the control group. Interestingly, relative to controls, KKU-100 cells treated with 10 μM CBD exhibited a significant decrease in the expression of p-PI3K (p = 0.039), p-AKT (p = 0.048), and p-mTOR (p = 0.037) at 48 h. Likewise, KKU-213B cells treated with CBD 10 μM showed a slight decrease in the expression of p-PI3K, p-AKT, and p-mTOR at 24 h, but a significant decrease in these proteins was observed at 48 h (p = 0.019, 0.006 and 0.030, respectively; Fig. 6A and D).
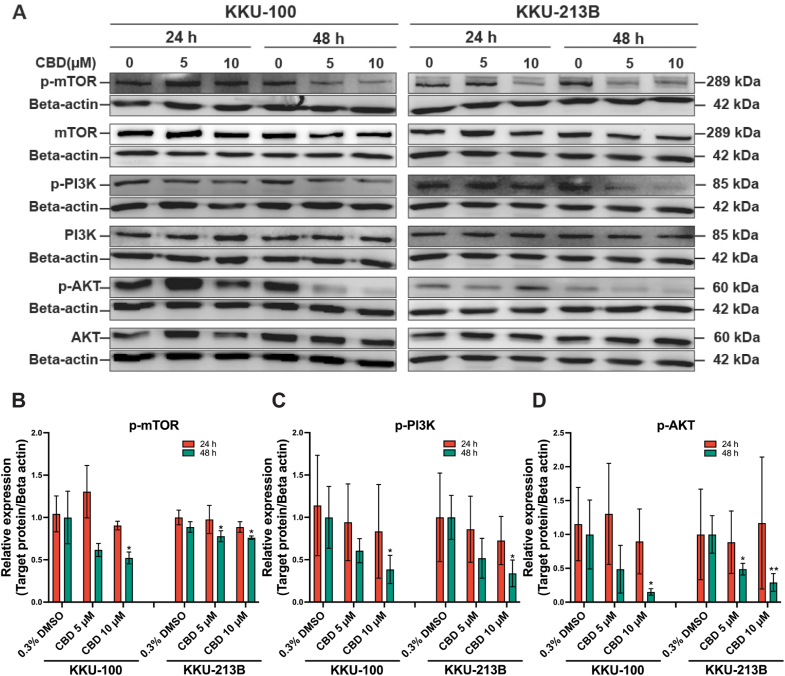
CBD promotes cellular senescence in CCA cells
To evaluate the effect of CBD on induction of cellular senescence in CCA cell lines, SA-β-gal staining was performed. SA-β-gal appeared deep blue as shown in Fig. 7A. Cellular senescence significantly increased in KKU-100 and KKU-213B cells treated with 5 μM and 10 μM (p < 0.0001) compared to the vehicle control (Fig. 7B and C). To confirm cellular senescence, a Western blot analysis was performed to examine the cellular senescence markers p21 and p53 (Fig. 7D). CBD treatment (10 μM) of KKU-100 and KKU-213B for 24 h elicited a significant upregulation in the expression of p21 (KKU-100: p < 0.0001) when contrasted with treatment involving 0.3 % DMSO (Fig. 7E). In addition, p53 expression was found to be significantly elevated in the groups treated with 10 μM CBD (KKU-100: p = 0.042, KKU-213B: p = 0.035; Fig. 7D and F), confirming the activation of cell senescence by CBD in CCA cells.
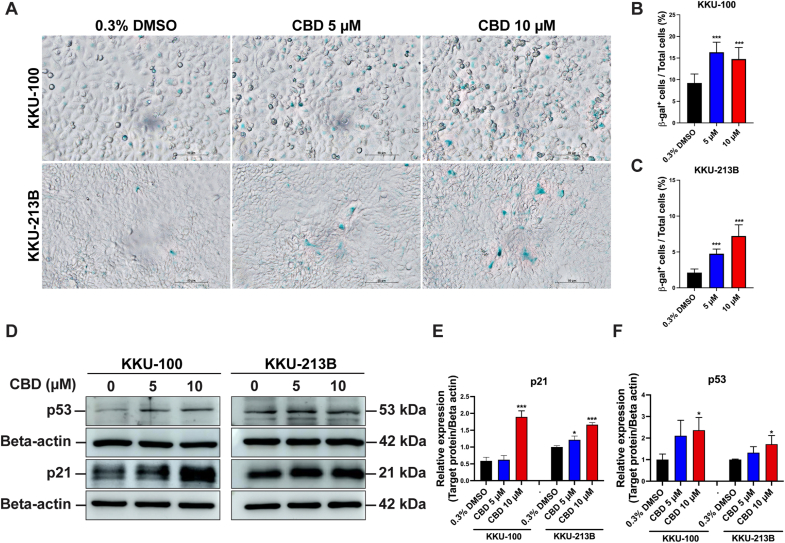
CBD suppresses CCA cell proliferation and promotes cellular senescence in vivo
The tumor volume of xenograft mice treated with 50 mg/kg·Bw was significantly lower upon evaluation on day 5 after treatment (p = 0.0357) when compared to vehicle control treatment (Fig. 8A and B). Additionally, an immunohistochemistry study was performed to confirm the effect of CBD on the suppression of CCA proliferation and induction of cellular senescence at the molecular level (Fig. 8C). The expression of PCNA, β-gal and p21 were assessed and its area and intensity were measured using ImageJ software (Fig. 8D). The expression of PCNA in the 50 mg/kg·Bw group was significantly reduced (p < 0.0001) relative to the vehicle control group. Immunoreactivity indicated a significant increase in β-gal levels in mice treated with 50 mg/kg·Bw of CBD (p < 0.0001). Consistently, a noteworthy elevation in p21 expression was observed in tumor tissues by CBD treatment at the same dosage (p < 0.0001).
Discussion
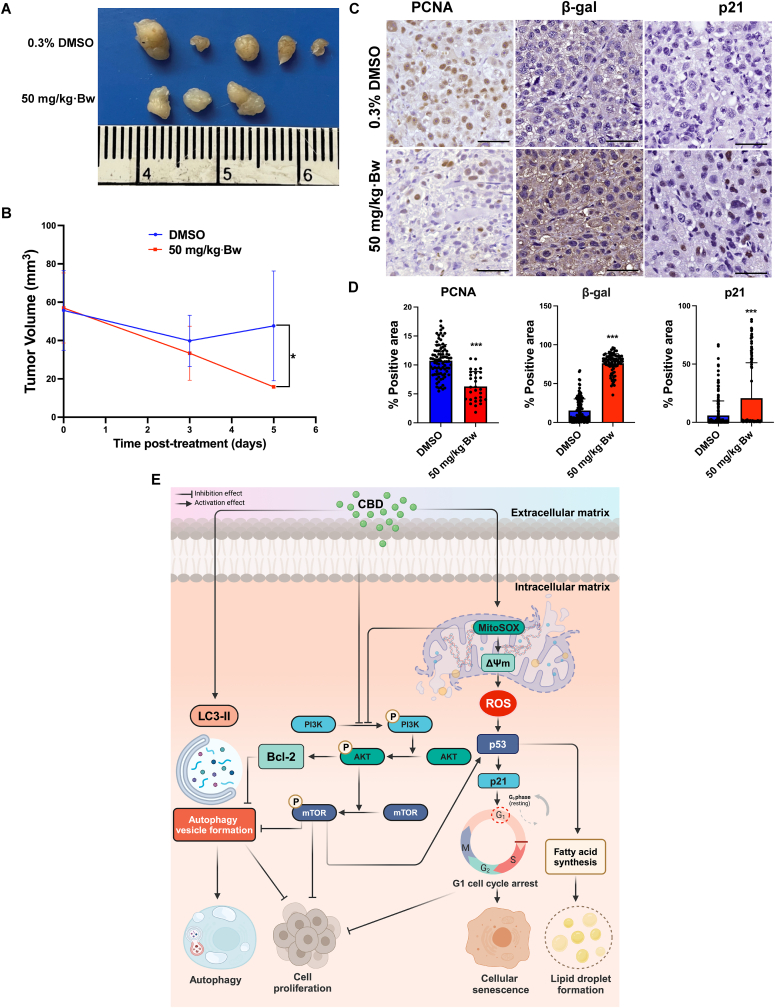
Although CBD has shown anti-tumor activity in various solid tumors, including CCA, its mechanism of action remains poorly understood.ref. bib30,ref. bib31 This study provides, for the first time, an insight into the anti-CCA mechanism of CBD and highlights its effect in vitro and in vivo via multiple mechanisms. Specifically, CBD was found to inhibit cell proliferation, colony formation, induce micro- and ultrastructural changes, disrupt mitochondrial membrane potential and increase mitochondrial ROS generation, and increase ROS production. In addition, CBD induced autophagy by suppression of the PI3K/AKT/mTOR pathway, activated cell cycle arrest and induced cellular senescence in CCA cells. These results suggest that CBD holds potential as an effective treatment for CCA. The proposed anti-CCA mechanism of CBD is depicted in Fig. 8E.
Various herbal agents, including CBD, have shown promise for the treatment of CCA.ref. bib32 Viereckl and colleagues have reported high efficacy of CBD within a dosage range of 50–100 μM.ref. bib31 In this study, we determined lower IC50 values for CBD (10.59–11.43 μM), suggesting heightened sensitivity of CCA to CBD in the context of O. viverrini infection. Our study supports this concept by demonstrating the suppression of tumor cell growth by CBD in both in vitro and in vivo models. Specifically, CBD administration in animal models resulted in a reduction in tumor cell division by downregulating the expression of PCNA, highlighting its potential clinical applications. The anti-CCA activity of CBD can be attributed, at least in part, to its ability to inhibit cancer cell division and prevent colony formation, providing insight into the underlying mechanism of action.
The modulation of cell cycle regulation by phytochemicals and natural products represents an important strategy to combat tumor cell proliferation, metastasis, and recurrence in various cancers.ref. bib33,ref. bib34 Among these agents, CBD has emerged as a notable candidate known for its ability to arrest the cell cycle by upregulating the expression of p53 and p21 in solid and liquid tumors.ref. 34, ref. 35, ref. 36, ref. 37 Consistent with previous research, our study showed that CBD treatment induces G1 cell cycle arrest in CCA cells, mediated in part by upregulation of p53 and p21 expression. This finding is consistent with similar observations in gastric cancer,ref. bib34 chronic myeloid leukemia,ref. bib37 and ovarian cancer,ref. bib36 highlighting the potential of CBD as a promising therapeutic agent for targeting abnormal cell cycle progression in cancer treatment.
Herbal medicines, including CBD, are known for their ability to affect cancer cell morphology and to induce cell deathref. bib30 by induction of mitotically arrested and apoptotic cells, leading to lipid accumulation.ref. bib38 In our study, CBD treatment induced the formation of lipid droplets and vesicles, altering the characteristics of CCA cells. Relevantly, CBD treatment increased lipid accumulation in human mesenchymal stem cells,ref. bib39 and induced vesicle formation in human lung cancer cells,ref. bib40 suggesting CBD treatment may have an effect due to its role in cell cycle arrest, apoptosis, and cell death, possibly through activation of adipogenesis-related metabolic pathways via PPARγ.ref. bib40
The use of natural products, such as CBD, to target autophagy has shown promise for the prevention and treatment of various types of cancerref. bib41 by decreasing the activity of the PI3K/AKT/mTOR pathway.ref. bib30 As we found, CBD inhibited CCA growth by reducing phosphorylation of the PI3K/AKT/mTOR pathway in relation to a decrease in the expression of Bcl-2, a protein involved in cell survival, and an increase in autophagy. Similar effects of CBD on autophagy have been observed in breast cancerref. bib30 and bladder cancer.ref. bib42 Inhibition of autophagosome formation by CQ causes accumulation of LC3-II and p62 while an increase in LC3BII levels and a decrease in p62 levels were demonstrated following CBD treatment in CCA cells. The results suggested that CBD induces autophagy via the conversion of LC3-I to LC3-II, leading to an increase in LC3-II, while the amount of p62 would be decreased by lysosomal degradation. These findings are consistent with prior studies that have utilized plant-based compounds to induce autophagy in CCA.ref. bib27 Essentially, our results indicate that CBD influences the signaling pathways of LC3II and p62, while simultaneously reducing the expression of certain proteins such as p-PI3K, p-AKT, p-mTOR, and Bcl-2 in CCA cells, suggesting that CBD has potential therapeutic application in various cancer types, including CCA.
In addition to modulating autophagy, targeting cellular senescence through interruption of ROS production is a promising strategy for cancer treatment. The effect of CBD on ROS production was observed in two CCA cell lines, KKU-100 and KKU-213B. While CBD increased ROS levels in KKU-100 cells, it did not have the same effect in KKU-213B cells, indicating possible differences in glutathione redox status between these cell types.ref. bib43 Our findings revealed that CBD treatment not only induced the production of ROS but also led to an increase in MitoSOX, indicating enhanced mitochondrial ROS generation. CBD treatment stimulated the expression of p53 and its downstream effector, p21, both of which are pivotal regulators of cellular senescence.ref. bib44 Moreover, wide-type p53 inhibits the fatty acid synthesis and lipid accumulation, whereas mutant p53 that found in cancer cells inhibits AMPKα and activates SREBPs, resulting in enhances fatty acid biosynthesis and lipid droplet formations.ref. bib45 Consistent with our observations, the induction of cellular senescence in HepG2 cells using the plant derivative 1,8-cineole increased ROS production, inhibited AKT phosphorylation, and altered mitochondrial membrane potential and induced cellular senescenceref. bib46 in a manner similar to CBD. Notably, our in vitro findings were further supported by tumor xenograft tissues from mice treated with CBD, which revealed elevated levels of p21 and β-gal, indicative of increased cellular senescence. Hence, CBD may function as a therapeutic agent for CCA treatment by targeting mitochondrial ROS and cellular senescence pathways.
In addition, plant-based chemicals such as resveratrol promote autophagy-mediated cancer cell dormancy in ovarian cancerref. bib47 and in CCA.ref. bib48 Likewise, our CBD treatment in CCA alters autophagic markers, mediates cell cycle arrest, induces cellular senescence, and inhibits cell proliferation. CBD-induced autophagy may foster cell dormancy, typified by reduced metabolic activity and proliferation, providing cancer cell resilience under adverse conditions.ref. bib47
It is important to acknowledge certain limitations regarding the anti-tumor effects of CBD observed in our study. We used a xenograft model of CCA in immunocompromised mice, a model which may not reflect the complex interaction between CBD and the tumor environment in humans, suggesting that further studies on an orthotopic xenograft model may better mimic than subcutaneous xenograft.ref. bib49
Conclusions
The study reported here has shown that CBD has a significant anti-tumor effect on CCA cells through various mechanisms, including the inhibition of cell proliferation both in vitro and in vivo, the reduction of colony formation ability and the induction of multiple cellular processes, notably autophagy, cell cycle arrest, cellular senescence, mitochondrial dysfunction, lipid droplet formation, and ROS overproduction. The significant findings from our study strongly suggest that CBD, through its targeting of the PI3K/AKT/mTOR pathway, holds great promise as a therapeutic agent for treating CCA and potentially other cancers.
CRediT authorship contribution statement
Thatsanapong Pongking: Conceptualization, Data curation, Formal analysis, Methodology, Roles/Writing – original draft., Kitti Intuyod, Phonpilas Thongpon and Raynoo Thanan: Conceptualization, Investigation, Data curation, Methodology, Resources, Validation, Writing – review & editing., Chutima Sitthirach, Suppakrit Kongsintaweesuk, and Sirinapha Klungsaeng: Data curation, Investigation, Methodology. Apisit Chaidee, Nuttanan Hongsrichan, and Kulthida Vaeteewoottacharn: Investigation, Methodology, Validation. Chadamas Sakonsinsiri and Somdej Kanokmedhakul: Resources, Validation. Porntip Pinlaor and Somchai Pinlaor: Funding acquisition, Project administration, Resources, Supervision, Validation, Visualization, Writing – review & editing. The final manuscript was reviewed and approved by all authors. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Ethics approval
The protocol of study in CCA cell lines was approved by Khon Kaen University Ethics Committee for Human Research based on the declaration of Helsinki and the ICH good clinical practice guidelines (HE641530). Animal experiments were conducted following the Animal Ethics Committee of Khon Kaen University and the National Research Council of Thailand’s guidelines (No. IACUC-KKU-138/64).
Funding
This research has received funding support from the National Science Research and Innovation Fund (NSRF) via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation [grant number B05F630053/5856] and the Basic Research Fund of Khon Kaen University under Cholangiocarcinoma Research Institute (CARI-BRF64-4). Thatsanapong Pongking and Porntip Pinlaor were supported by National Research Council of Thailand (NRCT) through NRCT5-RGJ63003-055.
References
- J.M. Banales, J.J.G. Marin, A. Lamarca. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol, 2020. [DOI | PubMed]
- B. Doherty, V.E. Nambudiri, W.C. Palmer. Update on the diagnosis and treatment of cholangiocarcinoma. Curr Gastroenterol Rep, 2017. [DOI | PubMed]
- C. Holohan, S. Van Schaeybroeck, D.B. Longley, P.G. Johnston. Cancer drug resistance: an evolving paradigm. Nat Rev Cancer, 2013. [DOI | PubMed]
- J.J.G. Marin, E. Lozano, E. Herraez. Chemoresistance and chemosensitization in cholangiocarcinoma. Biochim Biophys Acta, Mol Basis Dis, 2018. [DOI | PubMed]
- B. Hryciuk, B. Szymanowski, A. Romanowska. Severe acute toxicity following gemcitabine administration: a report of four cases with cytidine deaminase polymorphisms evaluation. Oncol Lett, 2018. [DOI | PubMed]
- M.A. ElSohly, M.M. Radwan, W. Gul, S. Chandra, A. Galal. Phytochemistry of Cannabis sativa L. Prog Chem Org Nat Prod, 2017. [DOI | PubMed]
- T. Rudroff, J.M. Honce. Cannabis and multiple sclerosis-the way forward. Front Neurol, 2017. [DOI | PubMed]
- S.K. Aggarwal. Use of cannabinoids in cancer care: palliative care. Curr Oncol, 2016. [DOI | PubMed]
- J. Guggisberg, M. Schumacher, G. Gilmore, D.M. Zylla. Cannabis as an anticancer agent: a review of clinical data and assessment of case reports. Cannabis Cannabinoid Res, 2022. [DOI | PubMed]
- C. Gross, D.A. Ramirez, S. McGrath, D.L. Gustafson. Cannabidiol induces apoptosis and perturbs mitochondrial function in human and canine glioma cells. Front Pharmacol, 2021. [DOI]
- H. Hamad, B.B. Olsen. Cannabidiol induces cell death in human lung cancer cells and cancer stem cells. Pharmaceuticals, 2021. [DOI]
- T.A.M. Vrechi, A. Leao, I.B.M. Morais. Cannabidiol induces autophagy via ERK1/2 activation in neural cells. Sci Rep, 2021. [DOI | PubMed]
- S. Yothaisong, M. Thanee, N. Namwat. Opisthorchis viverrini infection activates the PI3K/AKT/PTEN and Wnt/beta-catenin signaling pathways in a Cholangiocarcinogenesis model. Asian Pac J Cancer Prev APJCP, 2014. [DOI | PubMed]
- A.L.S. Cruz, E.A. Barreto, N.P.B. Fazolini, J.P.B. Viola, P.T. Bozza. Lipid droplets: platforms with multiple functions in cancer hallmarks. Cell Death Dis, 2020. [DOI | PubMed]
- R. Kumari, P. Jat. Mechanisms of cellular senescence: cell cycle arrest and senescence associated secretory phenotype. Front Cell Dev Biol, 2021. [DOI]
- B. Sripa, W. Seubwai, K. Vaeteewoottacharn. Functional and genetic characterization of three cell lines derived from a single tumor of an Opisthorchis viverrini-associated cholangiocarcinoma patient. Hum Cell, 2020. [DOI | PubMed]
- K. Intuyod, C. Hahnvajanawong, P. Pinlaor, S. Pinlaor. Anti-parasitic drug ivermectin exhibits potent anticancer activity against gemcitabine-resistant cholangiocarcinoma in vitro. Anticancer Res, 2019. [DOI | PubMed]
- C. Li, K. Zhang, G. Pan. Dehydrodiisoeugenol inhibits colorectal cancer growth by endoplasmic reticulum stress-induced autophagic pathways. J Exp Clin Cancer Res, 2021. [DOI | PubMed]
- J.J.Y. Ong, J. Oh, X. Yong Ang. Optical diffraction tomography and image reconstruction to measure host cell alterations caused by divergent Plasmodium species. Spectrochim Acta Mol Biomol Spectrosc, 2023. [DOI]
- S. Chomphoo, H. Kondo, W. Hipkaeo. Electron-translucency and partial defects of synaptic basal lamina in the electrocyte synapse of an electric ray (Narke japonica) in 3D embedment-free section electron microscopy. Microsc Res Tech, 2024. [DOI]
- L. Zhang, H. Zhang, S. Du, X. Song, D. Hu. In Vitro transcriptional response of eimeria tenella to toltrazuril reveals that oxidative stress and autophagy contribute to its anticoccidial effect. Int J Mol Sci, 2023. [DOI]
- J. Beisl, G. Pahlke, M. Ehling-Schulz, G. Del Favero, D. Marko. Cereulide and deoxynivalenol increase LC3 protein levels in HepG2 liver cells. Toxins, 2022. [DOI]
- R. Li, C. Xue, Y. Pan. Research on different compound combinations of Realgar-Indigo naturalis formula to reverse acute promyelocytic leukemia arsenic resistance by regulating autophagy through mTOR pathway. J Ethnopharmacol, 2024. [DOI]
- V. Pasciu, M. Nieddu, F.D. Sotgiu, E. Baralla, F. Berlinguer. An overview on assay methods to quantify ROS and enzymatic antioxidants in erythrocytes and spermatozoa of small domestic ruminants. Animals, 2023. [DOI]
- N. Zhu, H. Wang, B. Wang. A member of the nuclear receptor superfamily, designated as NR2F2, supports the self-renewal capacity and pluripotency of human bone marrow-derived mesenchymal stem cells. Stem Cell Int, 2016. [DOI]
- S. Datta, D. Choudhury, A. Das. Autophagy inhibition with chloroquine reverts paclitaxel resistance and attenuates metastatic potential in human nonsmall lung adenocarcinoma A549 cells via ROS mediated modulation of beta-catenin pathway. Apoptosis, 2019. [DOI | PubMed]
- S. Thongchot, M. Thanee, W. Loilome. Curative effect of xanthohumol supplementation during liver fluke-associated cholangiocarcinogenesis: potential involvement of autophagy. J Tradit Compl Med, 2020. [DOI]
- P. Dana, R. Kariya, K. Vaeteewoottacharn. Upregulation of CD147 promotes metastasis of cholangiocarcinoma by modulating the epithelial-to-mesenchymal transitional process. Oncol Res, 2017. [DOI | PubMed]
- A.-A.R.E. El-Hadary, A.M. Sulieman, G.A. El-Shorbagy. Comparative effects of hibiscus leaves and potato peel extracts on characteristics of fermented orange Juice. J Food Qual Hazards Control, 2023. [DOI]
- E.S. Seltzer, A.K. Watters, D. MacKenzie, L.M. Granat, D. Zhang. Cannabidiol (CBD) as a promising anti-cancer drug. Cancers, 2020. [DOI]
- M.J. Viereckl, K. Krutsinger, A. Apawu. Cannabidiol and cannabigerol inhibit cholangiocarcinoma growth in vitro via divergent cell death pathways. Biomolecules, 2022. [DOI]
- K. Na-Bangchang, T. Plengsuriyakarn, J. Karbwang. The role of herbal medicine in cholangiocarcinoma control: a systematic review. Planta Med, 2023. [DOI | PubMed]
- Y. Sun, Y. Liu, X. Ma, H. Hu. The influence of cell cycle regulation on chemotherapy. Int J Mol Sci, 2021. [DOI]
- X. Zhang, Y. Qin, Z. Pan. Cannabidiol induces cell cycle arrest and cell apoptosis in human gastric cancer SGC-7901 cells. Biomolecules, 2019. [DOI]
- I.L. Hsin, H.P. Shen, H.Y. Chang, J.L. Ko, P.H. Wang. Suppression of PI3K/Akt/mTOR/c-Myc/mtp53 positive feedback loop induces cell cycle arrest by dual PI3K/mTOR inhibitor PQR309 in endometrial cancer cell lines. Cells, 2021. [DOI]
- L. Ma, H. Zhang, C. Liu. A novel mechanism of cannabidiol in suppressing ovarian cancer through LAIR-1 mediated mitochondrial dysfunction and apoptosis. Environ Toxicol, 2023. [DOI]
- F. Maggi, M.B. Morelli, D. Tomassoni. The effects of cannabidiol via TRPV2 channel in chronic myeloid leukemia cells and its combination with imatinib. Cancer Sci, 2022. [DOI | PubMed]
- A. Wong, S. Chen, L.K. Yang, Y. Kanagasundaram, K. Crasta. Lipid accumulation facilitates mitotic slippage-induced adaptation to anti-mitotic drug treatment. Cell Death Dis, 2018. [DOI]
- R.C. Chang, C.S. Thangavelu, E.M. Joloya, A. Kuo, Z. Li, B. Blumberg. Cannabidiol promotes adipogenesis of human and mouse mesenchymal stem cells via PPARgamma by inducing lipogenesis but not lipolysis. Biochem Pharmacol, 2022. [DOI]
- Y.J. Park, H.H. Na, I.S. Kwon. Cannabidiol regulates PPARgamma-Dependent vesicle formation as well as cell death in A549 human lung cancer cells. Pharmaceuticals, 2022. [DOI]
- S. Deng, M.K. Shanmugam, A.P. Kumar, C.T. Yap, G. Sethi, A. Bishayee. Targeting autophagy using natural compounds for cancer prevention and therapy. Cancer, 2019. [DOI | PubMed]
- C. Chen, H. Gao, X. Su. Autophagy-related signaling pathways are involved in cancer (Review). Exp Ther Med, 2021. [DOI | PubMed]
- R.J. Mailloux. An update on methods and approaches for interrogating mitochondrial reactive oxygen species production. Redox Biol, 2021. [DOI]
- L. Bousset, J. Gil. Targeting senescence as an anticancer therapy. Mol Oncol, 2022. [DOI | PubMed]
- A. Parrales, T. Iwakuma. p53 as a regulator of lipid metabolism in cancer. Int J Mol Sci, 2016. [DOI]
- B. Rodenak-Kladniew, A. Castro, P. Starkel, M. Galle, R. Crespo. 1,8-Cineole promotes G0/G1 cell cycle arrest and oxidative stress-induced senescence in HepG2 cells and sensitizes cells to anti-senescence drugs. Life Sci, 2020. [DOI]
- A. Esposito, A. Ferraresi, A. Salwa, C. Vidoni, D.N. Dhanasekaran, C. Isidoro. Resveratrol contrasts IL-6 pro-growth effects and promotes autophagy-mediated cancer cell dormancy in 3D ovarian cancer: role of miR-1305 and of its target ARH-I. Cancers, 2022. [DOI]
- S. Thongchot, A. Ferraresi, C. Vidoni. Preclinical evidence for preventive and curative effects of resveratrol on xenograft cholangiocarcinogenesis. Cancer Lett, 2024. [DOI]
- R.M. Hoffman. Patient-derived orthotopic xenografts: better mimic of metastasis than subcutaneous xenografts. Nat Rev Cancer, 2015. [DOI | PubMed]
