GTSE1: A potential prognostic and diagnostic biomarker in various tumors including lung adenocarcinoma
Abstract
Objective:
This research was aimed to comprehensively investigate the expression levels, diagnostic and prognostic implications, and the relationship with immune infiltration of G2 and S phase‐expressed‐1 (GTSE1) across 33 tumor types, including lung adenocarcinoma (LUAD), through gene expression profiling.
Methods:
GTSE1 mRNA expression data together with clinical information were acquired from Xena database of The Cancer Genome Atlas (TCGA), ArrayExpress, and Gene Expression Omnibus (GEO) database for this study. The Wilcoxon rank‐sum test was used to detect differences in GTSE1 expression between groups. The ability of GTSE1 to accurately predict cancer status was evaluated by calculating the area under the curve (AUC) value for the receiver operating characteristic curve. Additionally, we investigated the predictive value of GTSE1 in individuals diagnosed with neoplasms using univariate Cox regression analysis as well as Kaplan–Meier curves. Furthermore, the correlation between GTSE1 expression and levels of immune infiltration was assessed by utilizing the Tumor Immune Estimate Resource (TIMER) database to calculate the Spearman rank correlation coefficient. Finally, the pan‐cancer analysis findings were validated by examining the association between GTSE1 expression and prognosis among patients with LUAD.
Results:
GTSE1 exhibited significantly increased expression levels in a wide range of tumor tissues in contrast with normal tissues (p < 0.05). The expression of GTSE1 in various tumors was associated with clinical features, overall survival, and disease‐specific survival (p < 0.05). In immune infiltration analyses, a strong correlation of the level of immune infiltration with the expression of GTSE1 was observed. Furthermore, GTSE1 demonstrated good discriminative and diagnostic value for most tumors. Additional experiments confirmed the relationship between elevated GTSE1 expression and unfavorable prognosis in individuals diagnosed with LUAD. These findings indicated the crucial role of GTSE1 expression level in influencing the development and immune infiltration of different types of tumors.
Conclusions:
GTSE1 might be a potential biomarker for the prognosis of pan‐cancer. Meanwhile, it represented a promising target for immunotherapy.
Article type: Research Article
Keywords: G2 and S phase‐expressed‐1, immune infiltration, lung adenocarcinoma, pan‐cancer, prognostic biomarkers
Affiliations: Department of Cardio‐Thoracic Surgery First Affiliated Hospital of Guangxi Medical University Nanning Guangxi People’s Republic of China
License: © 2024 The Authors. The Clinical Respiratory Journal published by John Wiley & Sons Ltd. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/crj.13757 | PubMed: 38715380 | PMC: PMC11077242
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (8.1 MB)
INTRODUCTION
The global incidence of cancer is approximately 20 million cases annually, resulting in nearly 10 million deaths. Breast cancer (BRCA) has surpassed lung cancer as the most prevalent type since 2020, accounting for around 2.3 million new cases (11.7%). Lung cancer (11.4%) and colorectal cancer (10.0%) closely follow in terms of mortality. However, lung cancer remains the leading cause of mortality, with 8 million deaths (18%), significantly higher than colorectal cancer (9.4%), liver cancer (8.3%), stomach cancer (7.7%), and female BRCA (6.9%).ref. crj13757-bib-0001 Currently, a significant number of patients with lung cancer were diagnosed in advanced stages, which poses considerable treatment challenges. Therefore, early diagnosis is of immense significance, especially for screening individuals at higher risk, such as smokers and those exposed to smoke, oil fields, or toxic occupational environments.ref. crj13757-bib-0002 Histological subtypes of lung cancer include adenocarcinoma, squamous cell carcinoma, large cell carcinoma (also known as non‐small cell lung cancer), and small cell lung cancer. The extensive analysis of molecular features in lung adenocarcinoma (LUAD) has broadened our understanding of the cellular foundations and intricate molecular pathways associated with each subtype. Several genetic alterations have been identified as potential therapeutic targets and are currently under continuous development.ref. crj13757-bib-0003 In recent years, there have been notable advancements in the identification, description, and management of LUAD.ref. crj13757-bib-0004 Comprehensive genomic, epigenomic, and cellular analyses of LUAD and the tumor microenvironment have contributed to a better understanding of its development.ref. crj13757-bib-0005 Researchers had continuously discovered pan‐cancer related genes and developed potential targets for tumor diagnosis and treatment through pan‐cancer analysis network.ref. crj13757-bib-0006, ref. crj13757-bib-0007, ref. crj13757-bib-0008 However, the lack of corresponding targeted drug therapies has limited their widespread application. Therefore, further exploration of more effective markers for the diagnosis and treatment of LUAD is of utmost importance to facilitate early detection and reduce patient mortality.
G2 and S phase‐expressed‐1 (GTSE1) is a gene induced by the p53 protein, located on chromosome 22q13.2‐q13.3, which exhibits specific expression during the S and G2 stages of the cell cycle.ref. crj13757-bib-0009 GTSE1 preferentially binds to the most stable microtubules (MT) of the mitotic spindle and promotes their turnover.ref. crj13757-bib-0010 In non‐diploid carcinoma cell lines and tumors, overexpression of GTSE1 regulates MT stability during mitosis by inhibiting the activity of MCAK, an MT depolymerase enzyme.ref. crj13757-bib-0011 GTSE1 has been implicated in various malignant tumors, including osteosarcoma, BRCA, and colon cancer, regarding their occurrence, development, and prognosis.ref. crj13757-bib-0012, ref. crj13757-bib-0013, ref. crj13757-bib-0014 Lai et al.ref. crj13757-bib-0015 found that upregulation of GTSE1 can stimulate the growth of prostate cancer cells via the SP1/FOXM1 signaling pathway, while Zhang et al.ref. crj13757-bib-0016 suggested that GTSE1 induces chromosomal instability in esophageal squamous cell carcinoma cells and inhibits cell apoptosis through the ROS/JNK signaling pathway. Furthermore, enhancing GTSE1 expression facilitates the growth and spread of LUAD cells through the activation of alternative signaling pathways, such as the AKT/mTOR and ERK/MAPK pathways.ref. crj13757-bib-0017, ref. crj13757-bib-0018 GTSE1 also confers radio‐resistance to LUAD by promoting clonogenic generation and inhibiting cell apoptosis. Knocking down GTSE1 expression significantly reduces the proliferation and metastatic potential of LUAD cells while enhancing their sensitivity to radiotherapy.ref. crj13757-bib-0019 Although more and more evidences support the significant involvement of GTSE1 in the development of different carcinoma types, most studies had focused on individual cancers, lacking a systematic pan‐cancer analysis of GTSE1. Therefore, it was crucial to explore the association of GTSE1 expression levels, prognosis of patients, and tumor immune infiltration levels across various tumor types, including LUAD.
The aim of this study was to comprehensively analyze the degree of GTSE1 expression, along with its diagnostic and prognostic value in 33 types of malignant tumors, as well as its correlation with immune invasion using gene expression profiles. Additionally, we conducted further investigations to validate the specific expression pattern of GTSE1 in LUAD.
MATERIAL AND METHODS
Pan‐cancer expression data collection
The Cancer Genome Atlas (TCGA, https://www.portal.gdc.cancer.gov/) database encompasses a wide range of information on various types of human cancers, including clinical data, genomic variations, mRNA expression levels, miRNA expression patterns, and methylation profiles. To obtain the mRNA expression data for GTSE1 in the TCGA dataset, which includes 33 cancer types, we retrieved it from the Xena database. The mRNA expression values of GTSE1 were processed using the log2 (x + 1) conversion method, aiming to mitigate data skewness and promote a closer adherence to the normal distribution, thereby facilitating subsequent analyses.ref. crj13757-bib-0020, ref. crj13757-bib-0021
Collection of clinical parameters and prognostic data of pan‐cancer
Critical clinical characteristics for cancer patients, including age, gender, and the American Joint Committee on Cancer (AJCC) stage, were obtained from the TCGA dataset for the 33 cancer types. The Xena database was utilized to retrieve this information. Additionally, prognosis data, including overall survival and disease‐specific survival time, were collected from the same database.
Analysis of immune infiltration levels across pan‐cancer
The Tumor Immune Estimation Resource (TIMER, https://cistrome.shinyapps.io/timer/) databaseref. crj13757-bib-0022, ref. crj13757-bib-0023 utilizes high‐throughput sequencing technology (RNA‐Seq expression profiling) to evaluate the infiltration of immune cells within tumor tissues. It primarily provides insights into B cells, CD4+ T cells, CD8+ T cells, neutrophils, macrophages, and dendritic cells. Besides, Estimation of STromal and Immune cells in MAlignant Tumours using Expression data (ESTIMATE)ref. crj13757-bib-0024 is an alternative approach for assessing immune infiltration, enabling the determination of the immune interstitial score, ESTIMATE score, and tumor purity in a given sample. In this study, we employed both TIMER and ESTIMATE techniques to assess the levels of immune infiltration in tumor samples, aiming to explore the potential association of GTSE1 expression with immune infiltration levels.
LUAD expression and prognostic data acquisition
To partly validate the main findings in pan‐cancer, we conducted additional analysis focusing on GTSE1 in LUAD. Public LUAD mRNA expression data were obtained from various databases, including ArrayExpress (https://www.ebi.ac.uk/biostudies/arrayexpress) and Gene Expression Omnibus (GEO, https://www.ncbi.nlm.nih.gov/geo/). The inclusion criteria were as follows: (1) LUAD tissue samples collected from patients with LUAD, (2) paired samples of tissues adjacent to cancer included in the same dataset, and (3) complete mRNA expression data. The exclusion criteria were as follows: (1) non‐human tissue samples and (2) incomplete GTSE1 expression data. Ultimately, nine LUAD datasets, consisting of 485 LUAD samples and 485 samples adjacent to LUAD tissues, were collected for the analysis. The acquisition of prognostic data allowed us to evaluate the prognosis and survival outcomes of LUAD patients, facilitating the development of personalized treatment strategies. For this purpose, four prognostic datasets (GSE11963, GSE13213, GSE14814, and GSE19188), including 377 patients with LUAD, were collected to explore the correlation between GTSE1 expression and the prognosis of LUAD patients.
Statistical analysis
The Wilcoxon rank‐sum test was employed to identify any variations in the expression of GTSE1 between groups.ref. crj13757-bib-0025 Spearman’s rank correlation coefficients were used to assess the association between the expression of GTSE1 and immune infiltration levels. The predictive accuracy of GTSE1 in determining cancer status was evaluated by calculating the area under the curves (AUC) using receiver operating characteristic (ROC) analysis. The prognostic value of GTSE1 on patients with malignancies was evaluated utilizing univariate Cox regression analysis and Kaplan–Meier curves. Statistical analyses were conducted by R software (v4.1.0), except for the generation of the summary ROC curve, which was conducted in Stata (version 15.0). A p value below 0.05 was considered statistically significant.
RESULTS
The differential expression of GTSE1 between cancer tissues and control tissues
This study, comprising 9358 tumor samples and 722 control samples, undertook a comprehensive investigation into the differential expression of GTSE1 between cancerous and control tissues across diverse human tumor types. The findings revealed distinct GTSE1 expression levels among different cancer tissues in contrast with control tissues. Notable disparities were observed in 19 tumor types, such as bladder urothelial carcinoma, where the mRNA expression level of GTSE1 was higher among cancer tissues compared to the control tissues with statistical significance (p < 0.05; Figure 1A). However, no significant differences in GTSE1 expression were detected in cases of pancreatic adenocarcinoma (PAAD) and pheochromocytoma and paraganglioma (PCPG) when comparing cancer tissues to control tissues (p > 0.05; Figure 1A). Therefore, GTSE1 exhibits high expression in most tumors.
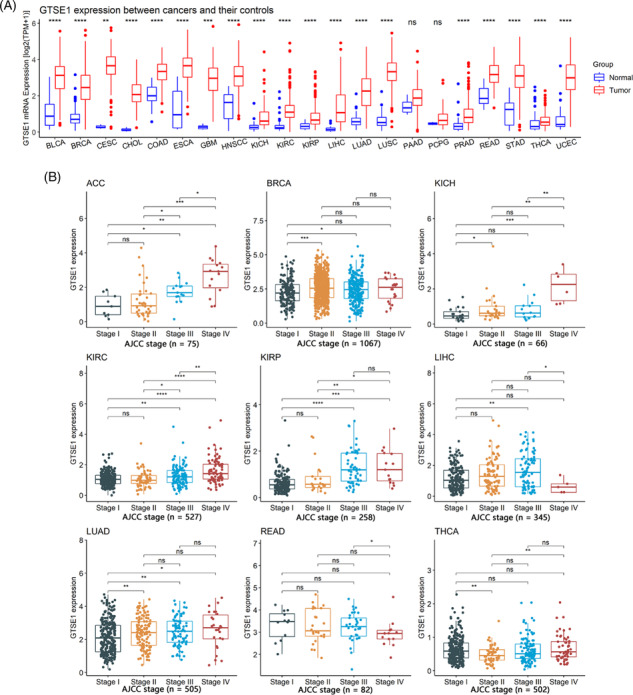
The relationship between GTSE1 and clinical parameters in tumor patients
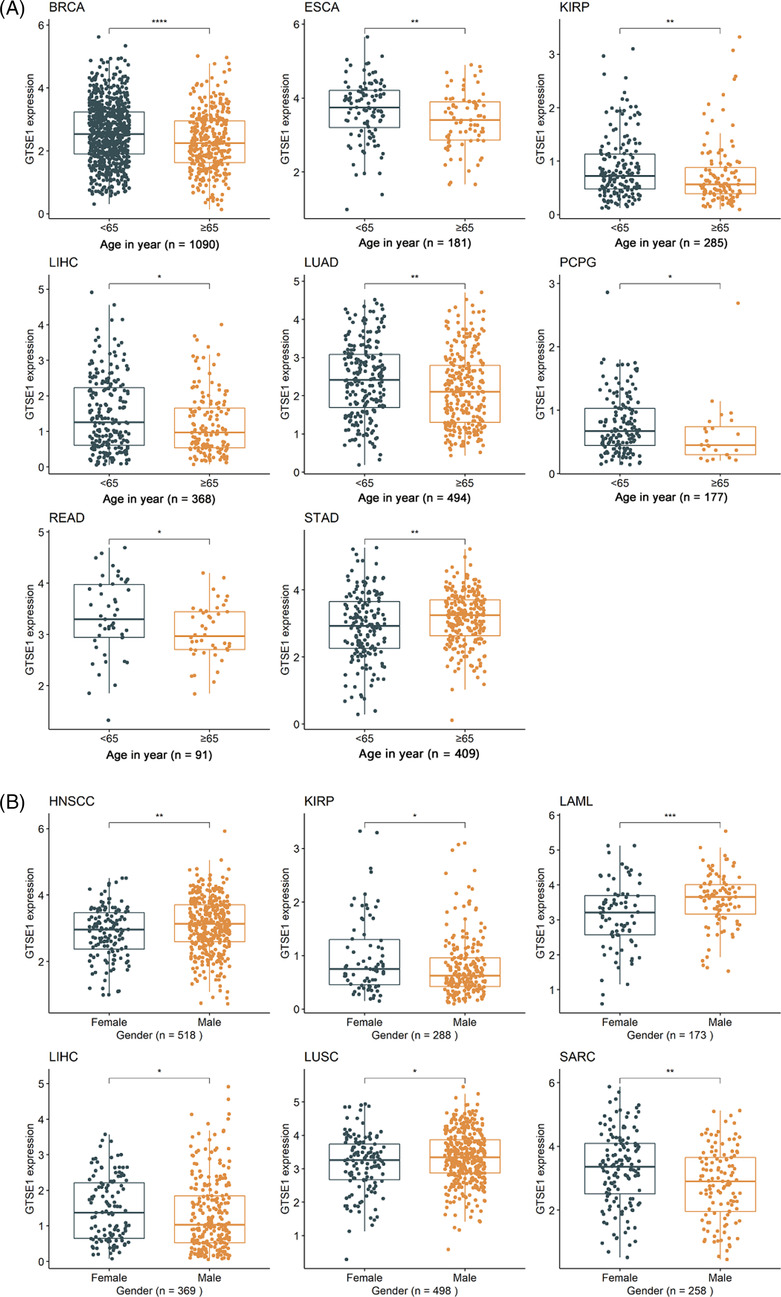
The differential expression of GTSE1 in patients with different clinical parameters was determined using Wilcoxon rank‐sum tests. The results showed that different AJCC stages were associated with varying levels of GTSE1 expression in nine different tumors (p < 0.05; Figure 1B), including adrenocortical carcinoma (ACC), breast invasive carcinoma (BRCA), kidney chromophobe (KICH), kidney renal clear cell carcinoma (KIRC), kidney renal papillary cell carcinoma (KIRP), liver hepatocellular carcinoma (LIHC), LUAD, rectum adenocarcinoma (READ), and thyroid carcinoma (THCA). Higher AJCC stages were consistently associated with higher levels of GTSE1 expression. Moreover, younger patients (<65 years old) with BRCA, esophageal carcinoma (ESCA), KIRP, LIHC, LUAD, PCPG, and READ had higher levels of GTSE1 expression in cancer tissues (p < 0.05; Figure 2A). Conversely, STAD (stomach adenocarcinoma) showed lower levels of GTSE1 in younger patients (<65 years old) (p < 0.05; Figure 2A). In HNSCC (head and neck squamous cell carcinoma), LAML (acute myeloid leukemia), and LUSC (lung squamous cell carcinoma), male patients exhibited higher levels of GTSE1 expression compared to female patients. However, male patients with KIRP, LIHC, and SARC (sarcoma) had reduced levels of GTSE1 expression (p < 0.05; Figure 2B). Baseline information from the TCGA dataset (Supporting Information S1) alongside the results of multivariate Cox regression analysis (Supporting Information S2) aids in investigating the relationship between GTSE1 and overall cancer prognosis, elucidating its prognostic significance across multiple cancer types.
The association between GTSE1 expression and the prognosis of cancer patients
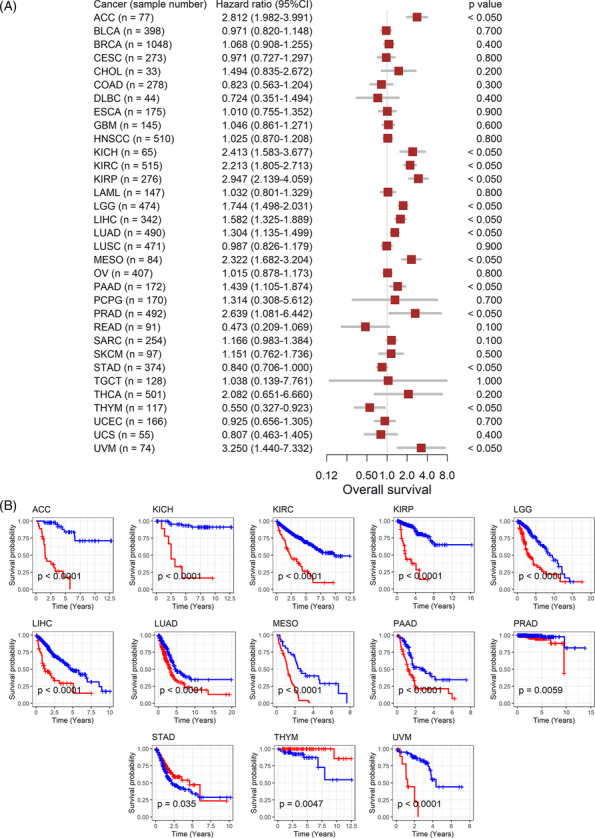
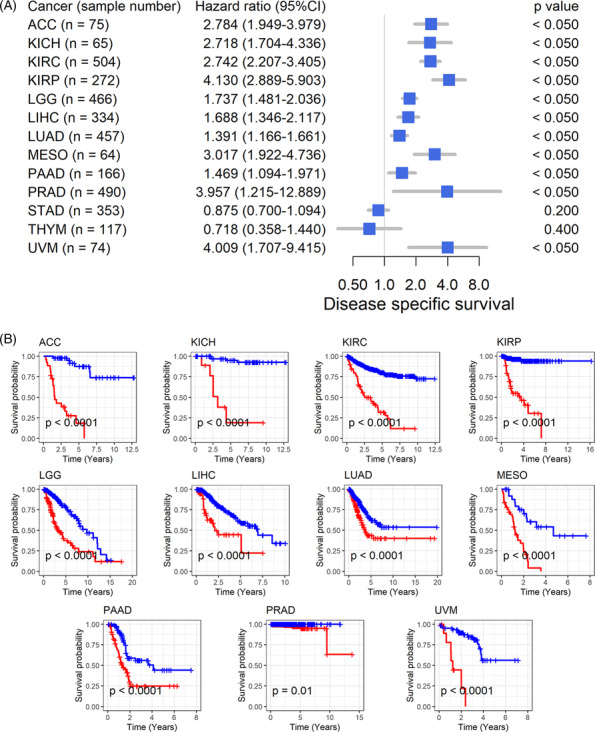
The association between GTSE1 expression and the prognosis of cancer patients suggests further analysis of its correlation with patient prognosis. High expression of GTSE1 is consistently linked to poor overall survival in 11 types of tumors, including ACC, indicating it as a risk factor (HR > 1, p < 0.05; Figure 3A) associated with shorter survival time for patients (p < 0.05; Figure 3B). Conversely, in STAD and THYM patients, high GTSE1 expression acts as a protective factor for overall survival (HR < 1, p < 0.05; Figure 3A), leading to extended survival time (p < 0.05; Figure 3B). Disease‐specific survival data further confirm these results in the overall survival analysis for the remaining 11 cancer types (HR > 1, p < 0.05; Figure 4A,B). Based on these findings, it could be inferred that the elevated GTSE1 expression is a significant prognostic hazard factor.
Potential identification effect of tumor patients using GTSE1
It is crucial for a clinical biomarker to distinguish tumor patients. In this study, results demonstrated that GTSE1 expression effectively distinguished cancer tissues from normal tissues in the 21 tumor types included in this study (sensitivity = 0.89, specificity = 0.92, area under the curve = 0.96; Figure 5A). In the ROC analysis of individual tumor types, AUC > 0.7 was observed for 19 types of cancer, indicating that GTSE1 mRNA expression levels are highly potential to differentiate cancer tissues from control tissues in these tumors (Figure 5B).
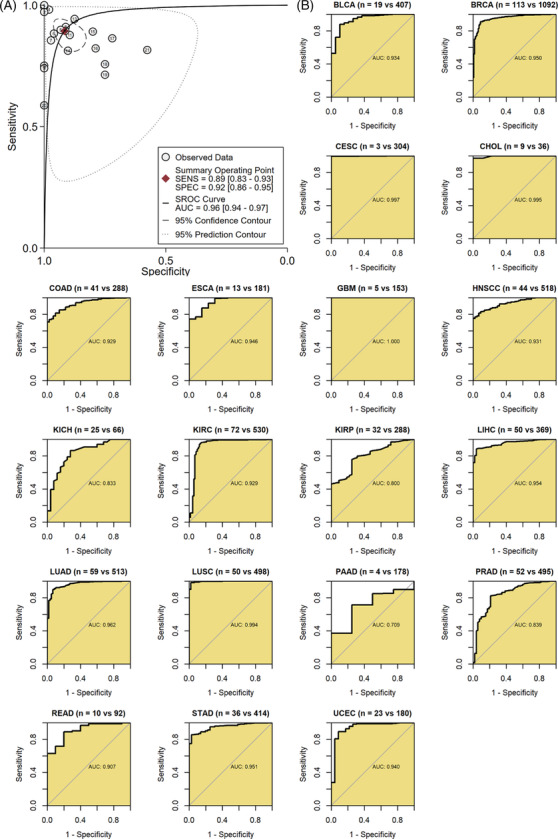
Relationship of GTSE1 expression and immune infiltration in tumors
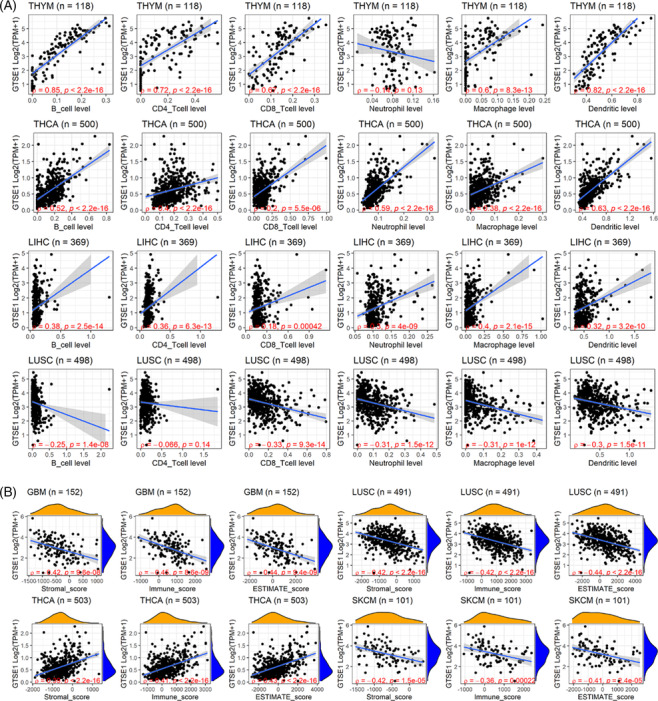
Immune infiltration plays a crucial role in tumor initiation and progression, holding significant clinical significance in various tumors. Thus, this study investigated the relationship of GTSE1 and tumor immune infiltration. Based on the results from the TIMER algorithm, the expression level of GTSE1 was found to be positively correlated with the infiltration levels of six immune cells in THYM, THCA, and LIHC (p < 0.05; Figure 6A). In LUSC, a negative correlation was observed between GTSE1 expression level and the infiltration levels of six immune cells (p < 0.05; Figure 6A). Another algorithm, ESTIMATE, revealed a negative correlation between GTSE1 expression level and three immune scores in GBM, LUSC, and SKCM, while it showed a positive correlation with the immune score in THCA (p < 0.05; Figure 6B).
Expression and clinical implications of GTSE1 in LUAD
Currently, lung cancer has been reported to have the highest mortality rate among tumors. Using LUAD as an example, this study further verified the expression and clinical relevance of GTSE1. Analysis of multi‐center data showed a higher expression level of GTSE1 mRNA in LUAD tissue compared to that in normal lung tissue (p < 0.05; Figure 7). Similar to most cancers, GTSE1 exhibited significant clinical significance in LUAD. Kaplan–Meier curve results suggested an association between a high GTSE1 expression level and a poor prognosis among LUAD patients (Figure 8). Based on ROC analysis, GTSE1 expression level showed a potential to effectively differentiate between LUAD and its control tissue (Table 1). Furthermore, baseline data from selected GEO and ArrayExpress datasets (Supporting Information S3) along with the results of multivariate Cox regression analysis (Supporting Information S4) contribute to the investigation of the relationship between GTSE1 and the prognosis of LUAD, elucidating the prognostic significance of GTSE1 in pulmonary adenocarcinoma.
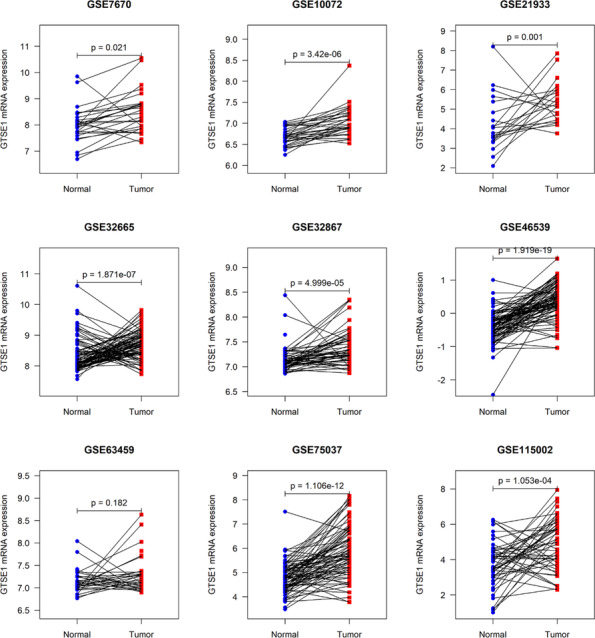
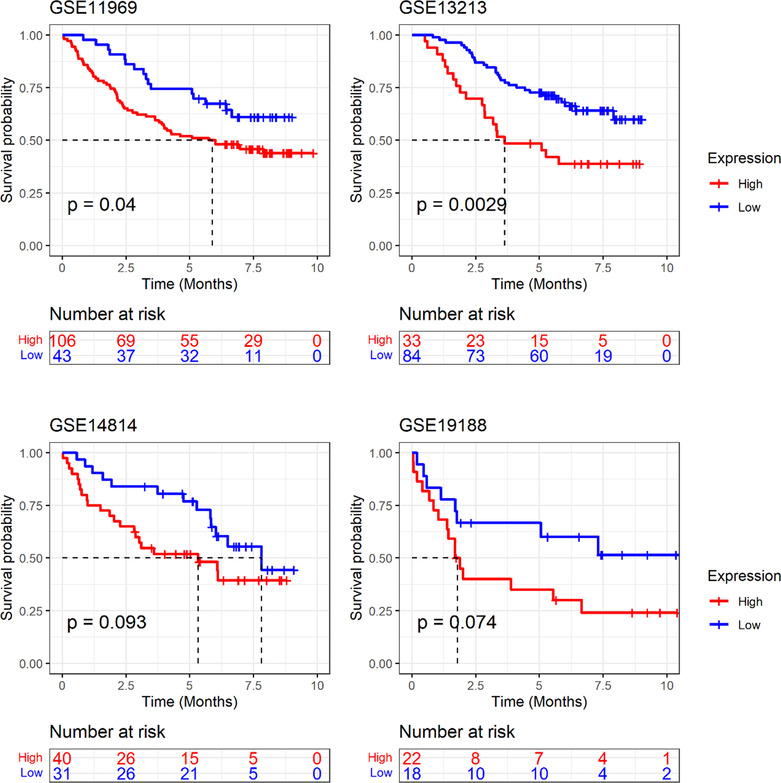
TABLE 1: Receiver operating characteristic curve analysis results of GTSE1 expression to identify normal control and LUAD group.
| Dataset | AUC | Cutoff | Sensitivity | Specificity | TP | FP | FN | TN |
|---|---|---|---|---|---|---|---|---|
| GSE10072 | 0.818 | 6.810 | 0.788 | 0.758 | 26 | 8 | 7 | 25 |
| GSE115002 | 0.721 | 4.864 | 0.538 | 0.865 | 28 | 7 | 24 | 45 |
| GSE21933 | 0.782 | 4.152 | 0.952 | 0.667 | 20 | 7 | 1 | 14 |
| GSE32665 | 0.729 | 8.353 | 0.793 | 0.678 | 69 | 28 | 18 | 59 |
| GSE32867 | 0.718 | 7.211 | 0.672 | 0.759 | 39 | 14 | 19 | 44 |
| GSE46539 | 0.885 | 0.081 | 0.815 | 0.924 | 75 | 7 | 17 | 85 |
| GSE63459 | 0.598 | 7.254 | 0.469 | 0.750 | 15 | 8 | 17 | 24 |
| GSE75037 | 0.820 | 5.443 | 0.663 | 0.916 | 55 | 7 | 28 | 76 |
| GSE7670 | 0.683 | 8.524 | 0.481 | 0.889 | 13 | 3 | 14 | 24 |
DISCUSSION
Up to now, numerous studies have demonstrated that GTSE1 is a potential risk gene for the development and progression of different tumors, including prostate cancer, BRCA, liver cancer, and others.ref. crj13757-bib-0013, ref. crj13757-bib-0015, ref. crj13757-bib-0026 Overexpression of GTSE1 has been found to inhibit apoptosis in esophageal malignant tumor cellsref. crj13757-bib-0016 and promote the growth and metastasis of LUAD cells.ref. crj13757-bib-0017 Despite the extensive research on pan‐cancer in the past 5 years, the association between GTSE1 and various malignant tumors remains unexplored. Hence, this study was aimed to comprehensively analyze gene expression profiles obtained from TCGA and Gene Expression Omnibus (GEO) databases to examine the levels of GTSE1 expression, its potential prognostic significance, and the extent of immune infiltration in neighboring tissues and cancerous tissues across 33 different types of malignant tumors. The results revealed upregulation of GTSE1 in 19 tumor tissues, including cervical cancer, suggesting its potential as a common biomarker for multiple tumors. Furthermore, overexpression of GTSE1 was associated with unfavorable prognosis in various tumor types. Importantly, these conclusions were based on pan‐cancer analysis of large sample data, suggesting g a certain level of reliability.
GTSE1 may exhibit distinct mechanisms of action in the development of different tumors. Analysis by Wu et al.ref. crj13757-bib-0027 demonstrated that GTSE1 levels in most cancer tissues (27/28, 96.4%) were approximately 100 times higher than in non‐cancerous tissues. Immunohistochemical methods revealed that cells with high expression levels of GTSE1 exhibited increased expression of N‐cadherin, β‐catenin, and Snail, indicating that GTSE1 might drive transformation of endothelial cells in hepatocellular carcinoma. Similarly, Liu et al.ref. crj13757-bib-0028 analyzed 95 samples of bladder cancer together with 30 normal samples, finding the increased GTSE1 expression contributed to cell proliferation, migration, and invasion through regulation of the p53/FoxM1/CCNB1 pathway, leading to an unfavorable prognosis in individuals with bladder cancer. To clarify the relationship of elevated expression levels of GTSE1 with overall survival across different tumor types, we conducted an analysis on the clinical features and overall survival of the included samples. The results demonstrated a positive correlation between AJCC stage and GTSE1 expression level. Patients with BRCA and seven other forms of carcinoma under the age of 65 exhibited higher GTSE1 expression levels, whereas young patients with gastric adenocarcinoma had lower levels of GTSE1. Interestingly, head and neck squamous cell carcinoma (HNSCC), acute myeloid leukemia (LAML), and lung squamous cell carcinoma (LUSC) showed higher expression levels of GTSE1 in males compared to females. Additionally, a high GTSE1 expression level was linked to the decreased overall/disease‐specific survival. Lei et al.ref. crj13757-bib-0029 developed a predictive model for clear cell renal cell carcinoma (ccRCC) patients, utilizing age, gender, and GTSE1 expression level, to estimate survival probabilities at 1, 3, and 5 years. They found that increased GTSE1 expression was associated with lower prognostic survival rates. Therefore, GTSE1 is generally considered an important prognostic risk factor.
The identification of biomarkers with diagnostic and predictive value is crucial for personalized cancer treatment.ref. crj13757-bib-0030 Therefore, we investigated the diagnostic value of GTSE1 as an independent pan‐cancer biomarker. Using ROC curves, we evaluated the diagnostic sensitivity and specificity of GTSE1, which effectively distinguished cancer tissues from normal tissues in 21 types of tumors with high diagnostic efficiency. Immune infiltration exerts a significant impact on tumor development,ref. crj13757-bib-0031 although it is not yet fully understood whether GTSE1 is associated with tumor immune invasion. Some previous studies have explored the link of GTSE1 and immune‐related cells, particularly in the context of cancer immunotherapy involving immune checkpoint inhibitors (ICIs).ref. crj13757-bib-0032 Lei et al.ref. crj13757-bib-0029 discovered a correlation between overexpression of GTSE1 and increased infiltration of immune cells in renal cell carcinoma. Co‐expression analysis revealed a correlation between elevated levels of GTSE1 and the presence of ICIs, including PDCD1 (PD1), LAG3, and CTLA4. This suggests that GTSE1 expression could potentially serve as an indicator to assess the effectiveness of ICIS. Tan et al.ref. crj13757-bib-0033 elucidated the correlation between GTSE1 and stromal score as well as immune score across various cancer types, furnishing pivotal clues for investigating the types and activity levels of immune cells within the tumor microenvironment. Our research results also confirmed a positive connection between the expression level of GTSE1 and infiltration levels of B cells, CD4 T cells, CD8 T cells, macrophage cells, and dendritic cells in THYM, THCA, and LIHC. Identifying tumor biomarkers that impact immune surveillance is crucial for our understanding of tumor evolution and provides an opportunity to study escape mechanisms.ref. crj13757-bib-0034 In this study, we proposed the immunoinfiltration relationship of GTSE1 in pan‐cancer for the first time, suggesting that enhanced expression of GTSE1 might significantly contribute to immune escape of tumor cells, thus affecting tumor progression.
For our research, we obtained a total of 485 LUAD tissue samples and their corresponding adjacent normal tissues from the ArrayExpress and GEO databases. The objective was to investigate the levels of GTSE1 expression in LUAD tissues and assess its potential clinical significance using extensive data obtained from multiple centers. The findings indicated upregulation of GTSE1 in LUAD tissues, showing good discriminatory potential for this type of cancer. However, the mechanism by which high expression of GTSE1 leads to a poorer prognosis in LUAD patients is still controversial. Previous studies have clearly demonstrated upregulation of GTSE1 expression in lung cancer tissues. Nonetheless, Tian et al.ref. crj13757-bib-0035 suggested that differentially expressed GTSE1 was not significantly associated with clinical characteristics and overall survival of patients. Additionally, Cao et al.ref. crj13757-bib-0036 made a significant finding that the GTSE1/p53/NF‐κB pathway can be regulated by miR‐181a‐5p, leading to inhibition of invasion and migration in lung adenoma. Since the expression of miR‐181a‐5p was low in LUAD cells, GTSE1 overexpression could potentially enhance the invasive and migratory abilities of LUAD cells. To clarify the relationship between high GTSE1 expression and the prognosis of LUAD patients, we conducted an analysis on four prognostic datasets from the GEO database, and the results showed that upregulation of GTSE1 expression was related with a shorter survival in LUAD patients.
To our knowledge, this study comprehensively integrated data from the TCGA, ArrayExpress, and GEO databases, resulting in a notably augmented sample size, thereby bolstering the robustness of our findings. Specifically, we elucidate, for the first time, disparities in the expression profiles of GTSE1 across diverse cancer types concerning clinical parameters, thus providing substantive implications for tailored cancer therapeutics. Moreover, our inquiry not only scrutinizes the interplay among GTSE1 expression, prognosis, and immune infiltration in pan‐cancer contexts but also discloses, for the inaugural time, the heightened diagnostic efficacy of GTSE1 across 21 distinct cancer types. Furthermore, we corroborate the expression patterns of GTSE1 and its clinical relevance in LUAD. Expanding upon this groundwork, our investigation delves into the nuanced association between GTSE1 expression levels and the infiltration dynamics of six immune cell subtypes (B cells, CD4 T cells, CD8 T cells, neutrophils, macrophages, and dendritic cells) in pan‐cancer settings, thereby underscoring the correlation between the infiltration levels of disparate immune cell subtypes and GTSE1 expression, thereby furnishing novel insights into immune evasion mechanisms and anti‐tumor modalities. Nevertheless, there were several limitations in our study. First, in our previous analysis of differential gene expression, no obvious disparities in GTSE1 expression levels were observed when compared tumor tissues to normal tissues among patients with PAAD and PCPG. Therefore, additional confirmation with a substantial number of cases is necessary to verify the diagnostic and prognostic significance of GTSE1 in PAAD and PCPG. Second, since our study relied solely on existing data, the bioinformatics findings would need to be confirmed by further experimental investigations. Future research endeavors should focus on unraveling the molecular mechanism of GTSE1 at a microscopic level. Lastly, although pan‐cancer analysis results supported the relationship of GTSE1 expression and immune regulation mechanisms, further exploration is of necessity to thoroughly understand its underlying mechanisms.
CONCLUSIONS
In summary, GTSE1 showed a potential as a diagnostic and prognostic biomarker for various types of cancers. Its expression was linked to the presence of immune cells within tumors and was correlated with immune cell infiltration. These findings provided a bioinformatics foundation for studying the effect of GTSE1 on the treatment and microenvironment of tumors. Further biological experiments will be required to validate its functionality and molecular mechanism in future.
CONFLICT OF INTEREST
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
AUTHOR CONTRIBUTIONS
Guanqiang Yan, Guosheng Li, and Huafu Zhou conceived and designed the study, data analysis, and article writing. Guanqiang Yan provided contributions to data curation and writing—original draft. Guosheng Li made forms. Xiang Gao finished the data validation and paper revision. Jun Liu and Yue Li downloaded data. Jingxiao Li finished visualization and writing—review & editing. Huafu Zhou was the corresponding author. All persons designated as the authors have participated sufficiently in the work to take public responsibility for the content of the manuscript.
ETHICS STATEMENT
The study was conducted in accordance with the Declaration of Helsinki, and granted exemption by the First Affiliated Hospital of Guangxi Medical University Ethical Review Committee.
Supplementary Materials
- Supplementary material S1. Baseline information of some datasets used to explore the prognosis of GSET1. (DOCX)
- Supplementary material S2. Multivariate Cox regression analysis results of the prognosis of GSET1. (DOCX)
- Supplementary material S3. Baseline information of some datasets used to explore the prognosis of GSET1. (DOCX)
- Supplementary material S4. Multivariate Cox regression analysis results of the prognosis of GSET1. (DOCX)
References
- Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries.. CA Cancer J. Clin.. [DOI]
- Current and future development in lung cancer diagnosis.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- Targeted therapy and checkpoint immunotherapy in lung cancer.. Surg. Pathol. Clin., 2020. [DOI | PubMed]
- Update 2020: management of non‐small cell lung cancer.. Lung.. [DOI | PubMed]
- The biology of lung cancer: development of more effective methods for prevention, diagnosis, and treatment.. Clin. Chest Med., 2020. [DOI | PubMed]
- Moving pan‐cancer studies from basic research toward the clinic.. Nat. Cancer., 2021. [DOI | PubMed]
- GPC2 is a potential diagnostic, immunological, and prognostic biomarker in pan‐cancer.. Front. Immunol., 2022. [DOI | PubMed]
- Systematic pan‐cancer analysis identifies TREM2 as an immunological and prognostic biomarker.. Front. Immunol., 2021. [DOI | PubMed]
- Cell‐cycle regulation of the p53‐inducible gene B99.. FEBS Lett., 2000. [DOI | PubMed]
- GTSE1 regulates spindle microtubule dynamics to control Aurora B kinase and Kif4A chromokinesin on chromosome arms.. J. Cell Biol., 2017. [DOI | PubMed]
- GTSE1 tunes microtubule stability for chromosome alignment and segregation by inhibiting the microtubule depolymerase MCAK.. J. Cell Biol., 2016. [DOI | PubMed]
- GTSE1 is possibly involved in the DNA damage repair and cisplatin resistance in osteosarcoma.. J. Orthop. Surg. Res., 2021. [DOI | PubMed]
- GTSE1 is involved in breast cancer progression in p53 mutation‐dependent manner.. J. Exp. Clin. Cancer Res., 2019. [DOI | PubMed]
- MiR‐509‐3‐5p inhibits colon cancer malignancy by suppressing GTSE1.. Biochem. Biophys. Res. Commun., 2021. [DOI | PubMed]
- GTSE1 promotes prostate cancer cell proliferation via the SP1/FOXM1 signaling pathway.. Lab. Invest., 2021. [DOI | PubMed]
- G2 and S phase‐expressed‐1 induces chromosomal instability in esophageal squamous cell carcinoma cells and inhibits cell apoptosis through ROS/JNK signaling.. Mol. Carcinog., 2023. [DOI | PubMed]
- GTSE1 facilitates the malignant phenotype of lung cancer cells via activating AKT/mTOR signaling.. Anal. Cell. Pathol. (Amst.)., 2021. [PubMed]
- GTSE1 promotes the growth of NSCLC by regulating microtubule‐associated proteins through the ERK/MAPK pathway.. Thorac. Cancer., 2023. [DOI | PubMed]
- Corrigendum.. J. Cell. Mol. Med., 2021. [DOI | PubMed]
- Expression of dendritic cell markers CD11c/BDCA‐1 and CD123/BDCA‐2 in coronary artery disease upon activation in whole blood.. J. Immunol. Methods., 2010. [DOI | PubMed]
- Log transformation: application and interpretation in biomedical research.. Stat. Med., 2013. [DOI | PubMed]
- TIMER: a web server for comprehensive analysis of tumor‐infiltrating immune cells.. Cancer Res., 2017. [DOI | PubMed]
- TIMER2.0 for analysis of tumor‐infiltrating immune cells.. Nucleic Acids Res., 2020. [DOI | PubMed]
- Inferring tumour purity and stromal and immune cell admixture from expression data.. Nat. Commun., 2013. [DOI | PubMed]
- Introduction to biostatistics: part 5, statistical inference techniques for hypothesis testing with nonparametric data.. Ann. Emerg. Med., 1990. [DOI | PubMed]
- GTSE1 promotes SNAIL1 degradation by facilitating its nuclear export in hepatocellular carcinoma cells.. Mol. Med. Rep., 2021. [DOI | PubMed]
- GTSE1 promotes cell migration and invasion by regulating EMT in hepatocellular carcinoma and is associated with poor prognosis.. Sci. Rep., 2017. [DOI | PubMed]
- Overexpression of G2 and S phase‐expressed‐1 contributes to cell proliferation, migration, and invasion via regulating p53/FoxM1/CCNB1 pathway and predicts poor prognosis in bladder cancer.. Int. J. Biol. Macromol., 2019. [DOI | PubMed]
- High GTSE1 expression promotes cell proliferation, metastasis and cisplatin resistance in ccRCC and is associated with immune infiltrates and poor prognosis.. Front. Genet., 2023. [DOI | PubMed]
- HGF/MET pathway aberrations as diagnostic, prognostic, and predictive biomarkers in human cancers.. Crit. Rev. Clin. Lab. Sci., 2019. [DOI | PubMed]
- Immune predictors of cancer progression.. Immunol. Res., 2012. [DOI | PubMed]
- Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance.. Annu. Rev. Pathol., 2021. [DOI | PubMed]
- Pan‐cancer analyses reveal GTSE1 as a biomarker for the immunosuppressive tumor microenvironment.. Medicine (Baltimore)., 2023. [DOI | PubMed]
- The adaptive immune system is a major driver of selection for tumor suppressor gene inactivation.. Science., 2021. [DOI | PubMed]
- Up‐regulation of GTSE1 lacks a relationship with clinical data in lung cancer.. Asian Pac. J. Cancer Prev., 2011. [PubMed]
- miR‐181a‐5p restrains non‐small cell lung cancer cell invasion and migration by regulating the GTSE1/p53/NF‐kappaB axis.. Cells Tissues Organs., 2022
