Characterization of the Volatile Compounds of the Hardwood Portion of Betula papyrifera Marshall From Quebec, Canada
Abstract
Betula papyrifera Marshall (paper birch) hardwood is an abundant yet underutilized resource for Quebec’s forestry industry. We investigated the volatile compounds of the hardwood extracted using hydrodistillation (HD) and headspace solid‐phase microextraction (HS‐SPME) and analyzed them by gas chromatography–mass spectrometry (GC–MS) and GC‐flame ionization detection. HD produced an essential oil with a low average yield (0.010% ± 0.001%), from which we identified 51 compounds, dominated by linoleic acid and its oxidation products. HS‐SPME provided a complementary profile, with 50 compounds identified, including aromatics and sesquiterpenes absent from the essential oil. The findings suggest that direct valorization of B. papyrifera hardwood for its volatile secondary metabolites is limited due to low yields and the prevalence of common compounds. Nevertheless, the study provides novel insights into the volatile chemical composition of B. papyrifera, contributing to the fundamental understanding of its extractives profile.
Article type: Research Article
Keywords: fatty acids, hardwood composition, natural products, volatile extract
Affiliations: Département De Chimie PROTEO and Centre d’études nordiques Université Laval Québec Canada
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202501398 | PubMed: 40680211 | PMC: PMC12629156
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.9 MB)
Introduction
The paper birch or white birch, scientifically known as Betula papyrifera Marshall, is easily identifiable by its distinctive white bark, which gradually exfoliates into thin lateral strips over time [ref. 1]. The peeling bark is the main characteristic that sets it apart from poplars and yellow birch, whose bark does not peel off. Birch’s white bark contains lenticels, identifiable by horizontal black streaks, and is embellished with black whisker‐like markings along the trunk. It is often found in mixed forests but also sometimes as a pioneer after fires or tree removal. B. papyrifera is distributed across North America and is particularly abundant in Canada and the northern United States. Geographically, it ranges from Alaska in the west to Newfoundland and Labrador in the east, and from throughout all southern regions of Canada to high latitudes in the north [ref. 1]. In Quebec, birch grows at least as far north as 55° N, with anecdotal observations suggesting occurrences as far north as 58° N [ref. 2]. Birch thrives in well‐drained, sandy, and cold soils. Upon reaching maturity, the tree typically attains a height ranging from 15 to 21 m, with an average diameter spanning 0.3–0.6 m [ref. 3, ref. 4]. Throughout history, indigenous communities in North America have utilized the tree’s bark to craft items such as canoes, baskets, and food containers [ref. 5].
Although the B. papyrifera is already an abundant species in Quebec, recent observations have revealed significant population changes. For example, in 2011 in the region of Saguenay Lac‐Saint‐Jean, a study noted that coniferous populations were decreasing in favor of deciduous trees [ref. 6]. This shift has resulted in a predominant presence of B. papyrifera and trembling aspen, Populus tremuloides Michx, in the region. Unfortunately, these two species, often undervalued by the forest industry, are frequently cut down but left unused on logging sites, representing a significant waste [ref. 7]. The surge in these tree species gives rise to multiple concerns as it creates changes in plant composition of ecosystems, thus creating challenges in terms of biodiversity and forest economics. In 2023, Canadian forests witnessed an unprecedented surge in wildfires, covering approximately 15 million hectares, representing about 4% of the total forest area, and greater than seven times the average annual burned area recorded over the past 40 years [ref. 8]. These fires continued to engulf large areas of the country in 2024 [ref. 9], further compounding their environmental impact. This raises concerns about the B. papyrifera’s tendency to thrive as a pioneer species after wildfires [ref. 10]. This highlights the importance of developing new strategies to better integrate B. papyrifera into the forestry industry. Doing so would help justify the challenges related to its exploitation and prevent the underutilization of this abundant and expanding forest resource.
New and innovative ways are being developed to add value to forest residues. A number of valorization projects have already highlighted potential new uses [ref. 11]. There are numerous studies on extractables from B. papyrifera, almost exclusively focused on the bark, which is particularly rich in pentacyclic triterpenes, interesting for their bioactive properties. Betulin, a common pentacyclic triterpene, is believed to be present in more than 200 plants and is responsible for the characteristic white color of the bark of B. papyrifera, in which it is particularly abundant [ref. 12, ref. 13]. The bark mainly contains betulin (20%–30%, or even up to 45%) that can easily be purified from the mixture by recrystallization [ref. 14, ref. 15]. The bark also contains, to a lesser extent but in significant proportions, lupeol, betulinic acid, and betulin 3‐caffeate. Other betulin‐derived triterpenoids, such as betulinic aldehyde, oleanolic acid, oleanolic acid‐3‐acetate, and other compounds, have been identified in various species of the genus Betula [ref. 16]. Betulin and its numerous natural and synthetic derivatives are particularly bioactive and have been studied, among other things, for their antiviral, antitumor, antibacterial, anti‐inflammatory, and anti‐HIV properties, and have also been the subject of studies to evaluate their potential in the treatment of metabolic disorders, infectious diseases, and cardiovascular and neurological disorders [ref. 12, ref. 13, ref. 16, ref. 17, ref. 18, ref. 19].
Although B. papyrifera extractive compounds have been the focus of increased research interest over the past decade, the volatile compounds in its hardwood remain mostly unexplored, presenting new opportunities for investigation into its unused potential. Wood from other trees, such as sandalwood (Santalum album L.), camphorwood (Cinnamomum camphora L.), or rosewood (Aniba duckei Ducke), are economically important sources of interesting volatile compounds known as extractives [ref. 11]. Moreover, solvent extractives of the wood from other birch species led to interesting lipophilic extracts containing volatile compounds such as those that can be extracted by processes like hydrodistillation (HD) [ref. 20, ref. 21]. The hardwood of trees contains many organic compounds: Polysaccharides, such as cellulose, hemicellulose, and lignin, give the wood its distinct structural properties, whereas other extractive compounds are found in much smaller quantities. Within the intricate physiology of plants, extractives have three distinct roles: serving as nutrient reserves, acting as plant hormones, and protecting the wood [ref. 22]. They contribute to many characteristic properties of wood, such as its odor, color, toxicity, stability to light, flammability, hygroscopicity, degradation, insect resistance, and permeability [ref. 23]. These compounds, often of low molecular weight, encompass different classes, including lipophilic compounds like fats, fatty acids, sterols, terpenoids, terpenes, and fatty alcohols or hydrosoluble compounds such as polyphenols, sugars, and lignans [ref. 24]. The organic compound composition in wood exhibits variability influenced by factors such as genetics, season, and age and significantly differs depending on the tree species.
HD is a traditional method of volatile compound extraction that has been employed for centuries and maintains economic significance, especially in the perfume and aromatherapy industries [ref. 25, ref. 26, ref. 27]. The extraction of volatile compounds by HD does not alter the wood other than dampening it; the wood could still be used in other valuable ways after extraction. Headspace solid‐phase microextraction (HS‐SPME) is a highly efficient and non‐destructive method for collecting volatile compounds while minimizing compound degradation, thus allowing analyzed volatilome to be closer to the real composition of the volatile compounds produced by the organism itself [ref. 28, ref. 29]. Although being an interesting extraction method for volatilome characterization, it is not suited for large‐scale recovery of extractives for industrial applications and is used as a complementary scientific investigation technique. Given that HD is more sensitive to high molecular weight compounds and favors degradation, the two methods are often viewed as complementary extraction methods for the analysis of volatile compounds [ref. 30].
Herein, we report the detailed characterization of the volatile profile of the hardwood of B. papyrifera. Two extraction methods were employed: HD, yielding the essential oil, and HS‐SPME. The overarching goals include acquiring fundamental knowledge about the volatilome of B. papyrifera hardwood, identifying commercially valuable or scientifically interesting molecules, and finding potential applications for B. papyrifera for the forestry industry.
Results and Discussion
Essential Oil Yields
Three trees were sampled in total, and three essential oils were obtained per tree from fresh wood using a 5‐h HD process. The extract obtained from birch was colorless to pale yellow, with an aroma reminiscent of grilled chicken and freshly cut green leaves. Previous studies on Betula pubescens Ehrh. and B. pendula Roth showed that the upper part of the tree may be richer in extractive compounds [ref. 31]. Therefore, to obtain a more representative extract of the entire tree, we selected pieces from the bottom, middle, and upper sections, blended them together, and performed three distillations on the resulting mix. These were then used to calculate the mean and standard deviation. The ages of the trees selected for the study were 54, 36, and 83 years. The youngest tree (Tree 2) exhibited a slightly lower volatile extract yield (calculated from dry mass) compared to the 2 older trees. They exhibited an average of triplicate yields (% w/w on dry mass) of 0.0114% ± 0.0008%, 0.0087% ± 0.0006%, and 0.010% ± 0.001% for Trees 1, 2, and 3, respectively. These yields can be considered particularly low, as HD of the same quantity of S. album L. hardwood (sandalwood) by Bisht et al. [ref. 32] resulted in yields of 1.6%–3.6%, more than 300 times higher than those obtained from B. papyrifera. Lower yields of volatile compounds are generally expected from the wood than other parts of the tree. For instance, Roitto et al. [ref. 31] observed that extractives from B. pendula and B. pubescens were almost double in branch biomass and 5–10 fold in bark than the from the stemwood.
Although the yield is low, extracting phenols from wood by hot‐water extraction could easily be combined with the recovery of an essential oil with the same process. Indeed, other research projects on the hot‐water extraction of wood phenols have demonstrated that it is an interesting way to valorize industrial woodwork residues [ref. 33]. It may be possible to improve the low yields obtained and add more value to B. papyrifera wood by HD.
Essential Oils Composition
A total of 51 compounds, constituting 96.6% (Tree 2) and 97.6% (Trees 1 and 3) of the total integrated area, were identified in the essential oils of B. papyrifera. A typical chromatogram of the volatile compounds in B. papyrifera is shown in Figure 1, and the peak assignments are presented in Table 1. Other chromatograms were very similar and are presented in the Supporting Information section. The compounds were separated and identified using gas chromatography–mass spectrometry (GC–MS), with semi‐quantitative proportions determined via GC‐flame ionization detection (FID).
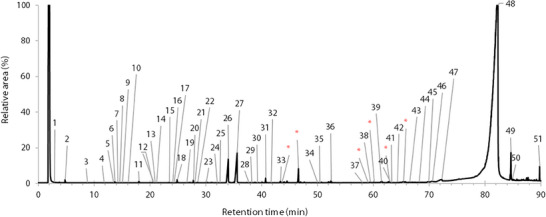
TABLE 1: Volatile organic compounds (VOCs) identified in the essential oils of the three B. papyrifera trees using mass spectrometry and linear retention index comparisons.
| No. | LRI | Compound name | No. CAS | Relative area (% ± SD)cbdv70230-tbl1-note-0002 | |||
|---|---|---|---|---|---|---|---|
| Exp.cbdv70230-tbl1-note-0003 | Lit. | Tree 1 | Tree 2 | Tree 3 | |||
| 1 | 703 | 700cbdv70230-tbl1-note-0004 | Heptane | 142‐82‐5 | Tr | Tr | Tr |
| 2 | 802 | 800cbdv70230-tbl1-note-0004 | Hexanal | 66‐25‐1 | 0.1 ± 0.0 | 0.1 ± 0.0 | Tr |
| 3 | 902 | 906cbdv70230-tbl1-note-0005 | Heptanal | 111‐71‐7 | Tr | Tr | Tr |
| 4 | 954 | 958 or 956cbdv70230-tbl1-note-0004 | (E or Z)‐2‐heptenal | 57266‐86‐1 or 18829‐55‐5 | Tr | Tr | Tr |
| 5 | 985 | 986cbdv70230-tbl1-note-0004 | 6‐Methyl‐5‐hepten‐2‐one | 110‐93‐0 | Tr | Tr | Tr |
| 6 | 989 | 991cbdv70230-tbl1-note-0005 | 2‐Pentylfuran | 3777‐69‐3 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 7 | 998 | 1002cbdv70230-tbl1-note-0004 | (E or Z)‐2‐(2‐pentenyl)furan | 70424‐14‐5 or 70424‐13‐4 | Tr | Tr | Tr |
| 8 | 1003 | 1003cbdv70230-tbl1-note-0004 | Octanal | 124‐13‐0 | Tr | Tr | Tr |
| 9 | 1010 | 1013cbdv70230-tbl1-note-0005 | (E,E)‐2,4‐heptadienal | 881395 | Tr | Tr | Tr |
| 10 | 1026 | 1031cbdv70230-tbl1-note-0004 | 3‐Ethyl‐2‐methyl‐1,3‐hexadiene | 61142‐36‐7 | Tr | Tr | Tr |
| 11 | 1055 | 1059cbdv70230-tbl1-note-0005 | (E)‐2‐Octenal | 2363‐89‐5 | 0.1 ± 0.0 | 0.1 ± 0.1 | 0.1 ± 0.0 |
| 12 | 1095 | 1105cbdv70230-tbl1-note-0004 | (E)‐4‐Nonenal | 2277‐16‐9 | Tr | Tr | Tr |
| 13 | 1099 | 1101cbdv70230-tbl1-note-0005 | Linalool | 78‐70‐6 | Tr | Tr | Tr |
| 14 | 1103 | 1107cbdv70230-tbl1-note-0005 | Nonanal | 124‐19‐6 | Tr | Tr | Tr |
| 15 | 1136 | 1134cbdv70230-tbl1-note-0005 | Non‐3‐en‐2‐one | 14309‐57‐0 | Tr | Tr | Tr |
| 16 | 1144 | 1148cbdv70230-tbl1-note-0004 | (Z)‐2‐nonenal | 60784‐31‐8 | Tr | Tr | Tr |
| 17 | 1149 | 1155 or 1152cbdv70230-tbl1-note-0004 | (E,E) or (E,Z)‐2,6‐nonadienal | 17587‐33‐6 or 557‐48‐2 | Tr | Tr | Tr |
| 18 | 1157 | 1163cbdv70230-tbl1-note-0005 | (E)‐2‐nonenal | 18829‐56‐6 | 0.2 ± 0.0 | 0.1 Tr 0.0 | 0.1 ± 0.0 |
| 19 | 1182 | 1192cbdv70230-tbl1-note-0005 | Octanoic acid | 124‐07‐2 | Tr | Tr | Tr |
| 20 | 1200 | 1200cbdv70230-tbl1-note-0004 | Dodecane | 112‐40‐3 | 0.1 ± 0.0 | 0.2 ± 0.1 | 0.1 ± 0.0 |
| 21 | 1204 | 1208cbdv70230-tbl1-note-0005 | Decanal | 112‐31‐2 | Tr | Tr | Tr |
| 22 | 1211 | 1218cbdv70230-tbl1-note-0005 | (E,E)‐2,4‐nonadienal | 5910‐87‐2 | Tr | Tr | Tr |
| 23 | 1234 | 1238cbdv70230-tbl1-note-0005 | Neral | 106‐26‐3 | Tr | Tr | Tr |
| 24 | 1264 | 1268cbdv70230-tbl1-note-0005 | Geranial | 141‐27‐5 | Tr | Tr | Tr |
| 25 | 1272 | 1273cbdv70230-tbl1-note-0004 | Nonanoic acid | 112‐05‐0 | Tr | Tr | Tr |
| 26 | 1294 | 1292cbdv70230-tbl1-note-0004 | (E,Z)‐2,4‐decadienal | 25152‐83‐4 | 2.2 ± 0.3 | 2.8 ± 0.2 | 1.1 ± 0.2 |
| 27 | 1318 | 1322cbdv70230-tbl1-note-0005 | (E,E)‐2,4‐decadienal | 25152‐84‐5 | 3.3 ± 0.3 | 4.3 ± 0.1 | 1.9 ± 0.3 |
| 28 | 1353 | 1363cbdv70230-tbl1-note-0004 | γ‐Nonalactone | 104‐61‐0 | Tr | Tr | Tr |
| 29 | 1361 | 1365cbdv70230-tbl1-note-0005 | (Z)‐8‐undecenal | 147159‐49‐7 | Tr | Tr | Tr |
| 30 | 1375 | 1386cbdv70230-tbl1-note-0005 | 9‐Decenoic acid | 14436‐32‐9 | — | Tr | Tr |
| 31 | 1400 | 1400cbdv70230-tbl1-note-0004 | Tetradecane | 629‐59‐4 | 0.1 ± 0.1 | 0.3 ± 0.1 | 0.2 ± 0.0 |
| 32 | 1416 | 1425cbdv70230-tbl1-note-0005 | (E,E)‐2,4‐undecadienal | 30361‐29‐6 | Tr | Tr | Tr |
| 33 | 1444 | 1453cbdv70230-tbl1-note-0004 | (E)‐geranyl acetone | 3796‐70‐1 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 34 | 1557 | 1561cbdv70230-tbl1-note-0005 | (E)‐nerolidol | 40716‐66‐3 | Tr | Tr | Tr |
| 35 | 1563 | 1568cbdv70230-tbl1-note-0004 | Dodecanoic acid | 143‐07‐7 | Tr | Tr | Tr |
| 36 | 1599 | 1600cbdv70230-tbl1-note-0005 | Hexadecane | 544‐76‐3 | 0.1 ± 0.0 | 0.1 ± 0.1 | 0.1 ± 0.0 |
| 37 | 1702 | 1714cbdv70230-tbl1-note-0005 | (E,Z)‐2,6‐farnesal | 3790‐67‐8 | Tr | Tr | Tr |
| 38 | 1730 | 1735cbdv70230-tbl1-note-0004 | (E,E)‐2,6‐farnesal | 502‐67‐0 | Tr | Tr | Tr |
| 39 | 1767 | 1768cbdv70230-tbl1-note-0004 | Tetradecanoic acid | 544‐63‐8 | 0.1 ± 0.1 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 40 | 1799 | 1800cbdv70230-tbl1-note-0005 | Octadecane | 593‐45‐3 | Tr | Tr | Tr |
| 41 | 1804 | 1818cbdv70230-tbl1-note-0005 | (E,E)‐2,6‐farnesoic acid | 462‐11‐3 | Tr | Tr | Tr |
| 42 | 1825 | 1832cbdv70230-tbl1-note-0005 | Farnesyl acetate | 29548‐30‐9 | Tr | Tr | Tr |
| 43 | 1866 | 1867cbdv70230-tbl1-note-0004 | Pentadecanoic acid | 1002‐84‐2 | Tr | — | — |
| 44 | 1903 | — | Farnesyl acetone isomer | — | Tr | Tr | Tr |
| 45 | 1935 | 1929cbdv70230-tbl1-note-0005 | Ambrettolide | 123‐69‐3 | 0.2 ± 0.1 | — | — |
| 46 | 1944 | 1951cbdv70230-tbl1-note-0004 | Palmitoleic acid | 373‐49‐9 | 0.2 ± 0.1 | 0.1 ± 0.0 | Tr |
| 47 | 1977 | 1968cbdv70230-tbl1-note-0004 | Hexadecanoic acid | 57‐10‐3 | 1.3 ± 0.9 | 0.2 ± 0.1 | 0.1 ± 0.0 |
| 48 | 2126 | 2133cbdv70230-tbl1-note-0004 | Linoleic acid | 60‐33‐3 | 87.7 ± 1.4 | 85.0 ± 1.3 | 91.8 ± 0.6 |
| 49 | 2375 | 2386cbdv70230-tbl1-note-0004 | (Z)‐9‐octadecenamide | 301‐02‐0 | 1.2 ± 0.2 | 1.8 ± 0.3 | 1.3 ± 0.5 |
| 50 | 2391 | 2374cbdv70230-tbl1-note-0004 | Octadecanamide | 124‐26‐5 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 51 | 2815 | 2832cbdv70230-tbl1-note-0004 | Squalene | 111‐02‐4 | 0.4 ± 0.0 | 0.8 ± 0.2 | 0.3 ± 0.1 |
Note: In bold, the main compounds (>1%).
Relative areas are calculated based on the average of nine replicates (three analyses from three separate distillations) for each of the three trees (1, 2, and 3); tr: relative proportion is marked as trace for values <0.1%.
Database linear retention indices (LRIs) shown in this table for non‐polar column DB‐5MS are obtained according to standards of n‐alkanes (C7–C30).
cLRI in NIST 14 database Mainlib and Replib [34].
dLRI in FFNSC 3 database [35].
B. papyrifera hardwood essential oil is rich in unsaturated fatty acids and their oxidation derivatives. Its profile is largely dominated by linoleic acid (85.0% ± 1.3% to 91.8% ± 0.6%), a fatty acid common to many plants and organisms [ref. 36, ref. 37]. Other major components include (E,E)‐2,4‐decadienal (1.9% ± 0.3% to 4.3% ± 0.1%), (E,Z)‐2,4‐decadienal (1.1% ± 0.2% to 2.8% ± 0.2%), (Z)‐9‐octadecenamide (1.2% ± 0.2% to 1.8% ± 0.3%), hexadecanoic acid (0.1% ± 0.0% to 1.3% ± 0.9%), squalene (0.3% ± 0.1% to 0.8% ± 0.2%), tetradecane (0.1% ± 0.1% to 0.3% ± 0.1%), and ambrettolide, identified only in Tree 1 (0.2% ± 0.1%).
Among the identified compounds, aldehydes constitute the most prevalent class in the essential oil, with 21 compounds, of which only 5 are saturated, namely, hexanal, heptanal, octanal, nonanal, and decanal. The remaining 16 are unsaturated compounds, including lipid oxidation products and terpenoids. Among these, the two most abundant compounds in the extract, following linoleic acid, are the isomers (E,Z)‐2,4‐decadienal and (E,E)‐2,4‐decadienal. Specimens with higher linoleic acid content exhibit lower proportions of these isomers, and vice versa. These 2 isomers, along with hexanal, are recognized as primary oxidation products of linoleic acid [ref. 38]. In the food industry, it has been observed that when using linoleic acid‐rich oils for frying, the concentrations of 2,4‐decadienal isomers increase progressively over time [ref. 39]. These compounds are considered significant contributors to the aroma of grilled chicken and frying in general [ref. 38, ref. 40], which may account for the distinctive scent of the essential oil.
Carboxylic acids, specifically fatty acids in this context, represent the second most identified class of compounds in the essential oil, with nine compounds detected. They are the most significant in terms of proportion, largely due to the high abundance of linoleic acid. Fatty acids originate from the saponification of structural lipids constituting the organism [ref. 27]. Hexadecanoic acid, more commonly known as palmitic acid, was identified in varying proportions. Although considerably less abundant, it is the second‐most‐abundant fatty acid in the essential oil of B. papyrifera, after linoleic acid. Palmitic acid is the most common saturated fatty acid in the human body and is very common in many animals and plants [ref. 41]. Trace amounts of other linear saturated fatty acids were identified, including pentadecanoic, tetradecanoic, dodecanoic, nonanoic, and octanoic. Additionally, two unsaturated fatty acids were observed: 9‐decenoic and (E,E)‐2,6‐farnesoic.
The two nitrogen compounds identified, (Z)‐9‐octadecenamide and octadecanamide, originate most probably from the cleavage of sphingolipids, which are lipids composed of a sphingosine on which a fatty acid is amidated, in this case oleic and stearic acid [ref. 42]. Sphingolipids are essential components of plasma membranes and other endomembranes of plant cells [ref. 43]. The two compounds identified are primary amides. (Z)‐9‐octadecenamide was identified in a particularly high relative proportion, varying on average between 1.2% ± 0.2% and 1.8% ± 0.3%, whereas octadecanamide was in trace amounts.
Terpenes and terpenoids are generally important for the valorization of essential oils due to their diverse bioactive properties and odorous profiles [ref. 26], making their identification and analysis pertinent for understanding the extract’s potential applications. Three monoterpenoids were identified in small proportions: linalool, neral, and geranial. Neral and geranial, both isomers of citral found in many flower and herb species, have a pleasant citrus aroma and are oxidation derivatives of geraniol [ref. 44]. Linalool, with its fresh, flowery scent, is a component of numerous essential oils and is widely used in the fragrance industry [ref. 45].
All the identified sesquiterpenoids were identified in trace amounts and belong to the farnesene family, including compounds such as (E)‐nerolidol, (E,E)‐ and (E,Z)‐2,6‐farnesal, (E,E)‐farnesoic acid, farnesyl acetate, and an unidentified isomer of farnesyl acetone (Compound 44).
These sesquiterpenoids are likely derived from farnesyl pyrophosphate (FPP), which, after cleavage from the terminal phosphate group by a phosphatase, can lead to the formation of various derivatives [ref. 46]. FPP can undergo hydrolysis to form 2,6‐(E,E)‐farnesol or (E)‐nerolidol. The latter, a sesquiterpenoid with a woody odor reminiscent of fresh bark, is found in many plants such as ginger, jasmine, lavender, lemongrass, and cannabis [ref. 47]. Farnesol can then be oxidized to produce aldehydes or carboxylic acids, leading to the formation of (E,E)‐2,6‐farnesal and (E,E)‐2,6‐farnesoic acid. The presence of an unsaturation in the trans configuration of FPP explains why most of the metabolites observed in this study exhibit the same configuration. Given that HD creates oxidative conditions, it is challenging to confirm whether some of these metabolites were formed during the extraction process or whether they are naturally produced by the plant itself.
Squalene, a triterpene, was identified as the molecule with the highest molecular weight in the essential oil, present in proportions ranging from 0.3% ± 0.1% to 0.8% ± 0.2%. Squalene is known to be a precursor for the biosynthesis of other triterpenes, including betulin and betulinic acid, which are major components of birch bark [ref. 16, ref. 48]. Neither of these compounds was detected in the essential oil. Squalene has also been identified as a major compound in B. papyrifera wood extracts obtained via Soxhlet extraction by Lavoie and Stevanovic [ref. 49], where it was found at a concentration of 4.062 mg/g (dry mass).
HS‐SPME/GC–MS
Wood chips from the hardwood of three freshly cut B. papyrifera trees were analyzed in triplicate by HS‐SPME–GC–MS. The typical chromatogram obtained is shown in Figure 2. The chromatograms of the other replicates are available in the Supporting Information section. A total of 50 compounds were identified in the headspace and are presented in Table 2, accounting for 80.8%, 75.1%, and 71.7% of the area of compounds integrated on the chromatogram for Trees 1, 2, and 3, respectively. The high variation in the percentage of identified compounds is due largely to the high variability of unidentified Compound 31 (6%–20%). The chromatogram of the headspace portion showed many signals, sometimes overlapping or with very low intensity. A significant portion of the identified compounds were unsaturated or branched, predominantly alkenes and sesquiterpenes. This made the identification process more difficult, as databases provided either too many possible molecules or no satisfactory options. Due to these factors, the percentages of identified compounds are lower using HS‐SPME, especially in the absence of linoleic acid. Linoleic acid constitutes a significant portion of the essential oil composition but is completely absent from the headspace analysis. This is most probably due to its high molecular weight and low volatility at the temperature used for headspace sampling.
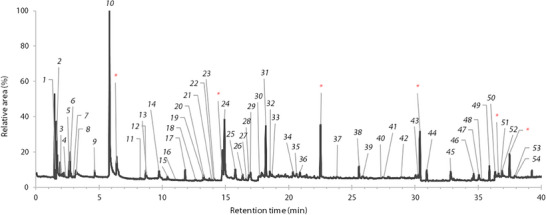
TABLE 2: Volatile organic compounds (VOCs) identified in the headspace of 3 Betula papyrifera trees.
| No. | LRI | Compound name | No. CAS | Relative area (% ± SD)cbdv70230-tbl2-note-0003 | |||
|---|---|---|---|---|---|---|---|
| Exp.cbdv70230-tbl2-note-0004 | Lit. | Tree 1 | Tree 2 | Tree 3 | |||
| 1 | <600 | 401cbdv70230-tbl2-note-0005 | Acetaldehydecbdv70230-tbl2-note-0007 | 75‐07‐0 | 2.4 ± 1.2 | 1.8 ± 0.1 | 1.2 ± 0.4 |
| 2 | <600 | 427cbdv70230-tbl2-note-0005 | Ethanolcbdv70230-tbl2-note-0007 | 64‐17‐5 | 3.7 ± 1.2 | 2.4 ± 0.2 | 0.5 ± 0.1 |
| 3 | <600 | 552cbdv70230-tbl2-note-0005 | 2‐Methylpropanalcbdv70230-tbl2-note-0007 | 78‐84‐2 | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.8 ± 0.1 |
| 4 | 605 | 631cbdv70230-tbl2-note-0005 | Methylcyclopentane | 96‐37‐7 | tr | Tr | 0.1 ± 0.0 |
| 5 | 636 | 676cbdv70230-tbl2-note-0006 | 3‐Methylbutanal | 590‐86‐3 | 1.7 ± 0.6 | 1.4 ± 0.2 | 1.5 ± 0.2 |
| 6 | 648 | 662cbdv70230-tbl2-note-0005 | 2‐Methylbutanal | 96‐17‐3 | 2.4 ± 0.7 | 1.8 ± 0.1 | 2.2 ± 0.3 |
| 7 | 673 | 680cbdv70230-tbl2-note-0006 | 1‐Penten‐3‐ol | 616‐25‐1 | 0.3 ± 0.1 | 0.2 ± 0.0 | 0.1 ± 0.1 |
| 8 | 695 | 699cbdv70230-tbl2-note-0005 | Pentanal | 110‐62‐3 | 0.4 ± 0.1 | 0.4 ± 0.1 | 0.7 ± 0.2 |
| 9 | 756 | 763cbdv70230-tbl2-note-0005 | Toluene | 108‐88‐3 | 1.5 ± 1.3 | 0.8 ± 0.2 | 0.3 ± 0.2 |
| 10 | 801 | 800cbdv70230-tbl2-note-0005 | Hexanal | 66‐25‐1 | 26.1 ± 2.7 | 25.4 ± 3.4 | 27.4 ± 4.2 |
| 11 | 854 | 855cbdv70230-tbl2-note-0005 | Ethylbenzene | 100‐41‐4 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.0 |
| 12 | 864 | 865cbdv70230-tbl2-note-0005 | p‐Xylene | 106‐42‐3 | 1.1 ± 0.1 | 0.8 ± 0.3 | 0.5 ± 0.1 |
| 13 | 866 | 866cbdv70230-tbl2-note-0005 | m‐Xylene | 108‐38‐3 | 0.8 ± 0.4 | 0.4 ± 0.1 | 0.4 ± 0.1 |
| 14 | 888 | 887cbdv70230-tbl2-note-0005 | o‐Xylene | 95‐47‐6 | 1.7 ± 0.2 | 1.2 ± 0.3 | 0.9 ± 0.3 |
| 15 | 902 | 901cbdv70230-tbl2-note-0005 | Heptanal | 111‐71‐7 | 0.5 ± 0.1 | 0.4 ± 0.1 | 0.7 ± 0.1 |
| 16 | 914 | 920cbdv70230-tbl2-note-0005 | Anisole | 100‐66‐3 | 0.1 ± 0.0 | 0.4 ± 0.1 | 1.2 ± 0.2 |
| 17 | 929 | 929cbdv70230-tbl2-note-0005 | α‐Pinene | 80‐56‐8 | 1.6 ± 0.3 | 0.8 ± 0.1 | 3.0 ± 0.3 |
| 18 | 944 | 952cbdv70230-tbl2-note-0005 or 950cbdv70230-tbl2-note-0005 | Camphene or α‐fenchene | 79‐92‐5 or 471‐84‐1 | 0.3 ± 0.1 | 0.2 ± 0.1 | 0.8 ± 0.1 |
| 19 | 956 | 958cbdv70230-tbl2-note-0005 or 956cbdv70230-tbl2-note-0005 | (E or Z)‐2‐Heptenal | 57266‐86‐1 or 18829‐55‐5 | 0.1 ± 0.1 | 0.2 ± 0.1 | 0.1 ± 0.1 |
| 20 | 957 | 957cbdv70230-tbl2-note-0005 or 954cbdv70230-tbl2-note-0005 | (m or p)‐Methyltoluene | 620‐14‐4 or 622‐96‐8 | 1.0 ± 0.2 | 0.6 ± 0.3 | 0.7 ± 0.1 |
| 21 | 969 | 974cbdv70230-tbl2-note-0005 or 966cbdv70230-tbl2-note-0005 | Sabinene or β‐thujene | 3387‐41‐5 or 28634‐89‐1 | 0.1 ± 0.0 | 0.2 ± 0.1 | 0.7 ± 0.0 |
| 22 | 973 | 979cbdv70230-tbl2-note-0005 or 974cbdv70230-tbl2-note-0005 | β‐Pinene or sabinene | 127‐91‐3 or 3387‐41‐5 | 0.5 ± 0.0 | 0.3 ± 0.1 | 1.1 ± 0.0 |
| 23 | 973 | 970cbdv70230-tbl2-note-0005 | o‐Ethyltoluene | 611‐14‐3 | Tr | Tr | Tr |
| 24 | 988 | 991cbdv70230-tbl2-note-0006 | 2‐Pentylfuran | 3777‐69‐3 | 10.8 ± 1.3 | 7.0 ± 1.0 | 3.1 ± 0.9 |
| 25 | 1005 | 1009cbdv70230-tbl2-note-0006 | o‐Methylanisole | 578‐58‐5 | 1.8 ± 0.1 | 2.8 ± 0.3 | 2.1 ± 0.2 |
| 26 | 1016 | 1013cbdv70230-tbl2-note-0005 | 1,2,3‐Trimethylbenzene | 526‐73‐8 | 1.1 ± 0.2 | 0.6 ± 0.2 | 0.9 ± 0.4 |
| 27 | 1021 | 1025cbdv70230-tbl2-note-0005 | p‐Cymene | 99‐87‐6 | 0.2 ± 0.0 | 0.4 ± 0.2 | 0.5 ± 0.1 |
| 28 | 1025 | 1030cbdv70230-tbl2-note-0006 | d‐Limonene | 138‐86‐3 | 1.0 ± 0.2 | 1.3 ± 0.2 | 1.3 ± 0.2 |
| 29 | 1028 | 1032cbdv70230-tbl2-note-0006 | Eucalyptol | 470‐82‐6 | 1.5 ± 0.4 | 1.9 ± 0.4 | 4.0 ± 0.5 |
| 30 | 1042 | 1045cbdv70230-tbl2-note-0005 | Phenylacetaldehyde | 122‐78‐1 | 0.3 ± 0.2 | Tr | 0.3 ± 0.1 |
| 31 | 1052 | — | Unknowncbdv70230-tbl2-note-0008 | — | 6.0 ± 1.9 | 6.7 ± 1.5 | 19.7 ± 4.3 |
| 32 | 1058 | 1059cbdv70230-tbl2-note-0006 | (E)‐2‐Octenal | 2363‐89‐5 | 0.9 ± 0.3 | 0.8 ± 0.3 | 0.7 ± 0.3 |
| 33 | 1061 | 1058cbdv70230-tbl2-note-0005 | 2,6,7‐Trimethyldecane | 62108‐25‐2 | 0.4 ± 0.1 | 0.6 ± 0.0 | 0.5 ± 0.1 |
| 34 | 1094 | 1105cbdv70230-tbl2-note-0005 | (E)‐4‐Nonenal | 2277‐16‐9 | 0.7 ± 0.1 | 0.4 ± 0.1 | 0.4 ± 0.2 |
| 35 | 1099 | 1100cbdv70230-tbl2-note-0005 | Undecane | 1120‐21‐4 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.4 ± 0.1 |
| 36 | 1104 | 1104cbdv70230-tbl2-note-0005 | Nonanal | 124‐19‐6 | 1.0 ± 0.2 | 0.6 ± 0.1 | 0.8 ± 0.3 |
| 37 | 1164 | 1164cbdv70230-tbl2-note-0005 | 2‐Methylundecane | 7045‐71‐8 | 0.2 ± 0.0 | 0.3 ± 0.1 | 0.2 ± 0.1 |
| 38 | 1199 | 1200cbdv70230-tbl2-note-0006 | Dodecane | 112‐40‐3 | 1.7 ± 0.3 | 1.2 ± 0.2 | 0.4 ± 0.0 |
| 39 | 1206 | 1206cbdv70230-tbl2-note-0005 | Decanal | 112‐31‐2 | 0.3 ± 0.1 | 0.4 ± 0.1 | 0.2 ± 0.0 |
| 40 | 1237 | 1238cbdv70230-tbl2-note-0005 | Hexylcyclohexane | 4292‐75‐5 | 0.1 ± 0.1 | 0.2 ± 0.0 | 0.1 ± 0.0 |
| 41 | 1242 | 1242cbdv70230-tbl2-note-0005 | 4‐Ethylundecane | 17312‐59‐3 | 0.4 ± 0.2 | 0.3 ± 0.1 | 0.2 ± 0.1 |
| 42 | 1271 | 1289cbdv70230-tbl2-note-0006 | Acide nonanoïque | 112‐05‐0 | 0.7 ± 0.4 | 0.5 ± 0.3 | 0.6 ± 0.1 |
| 43 | 1300 | 1300cbdv70230-tbl2-note-0005 | Tridecane | 629‐50‐5 | 0.5 ± 0.1 | 0.4 ± 0.2 | 0.2 ± 0.1 |
| 44 | 1315 | — | Unknowncbdv70230-tbl2-note-0008 | — | 2.4 ± 0.7 | 3.6 ± 2.1 | 1.2 ± 0.3 |
| 45 | 1359 | 1363cbdv70230-tbl2-note-0005 | γ‐Nonalactone | 104‐61‐0 | 1.9 ± 0.2 | 0.5 ± 0.2 | 0.2 ± 0.1 |
| 46 | 1400 | 1400cbdv70230-tbl2-note-0006 | Tetradecane | 629‐59‐4 | 1.1 ± 0.6 | 1.8 ± 0.4 | 0.6 ± 0.0 |
| 47 | 1415 | 1416cbdv70230-tbl2-note-0006 | (Z)‐α‐Bergamotene | 18252‐46‐5 | 1.2 ± 0.2 | 2.0 ± 0.2 | 1.3 ± 0.3 |
| 48 | 1422 | 1418cbdv70230-tbl2-note-0006 | α‐Santalene | 512‐61‐8 | 0.3 ± 0.2 | 0.3 ± 0.1 | 0.5 ± 0.2 |
| 49 | 1444 | 1435cbdv70230-tbl2-note-0005 | (E)‐α‐Bergamotene | 13474‐59‐4 | 2.1 ± 0.2 | 4.8 ± 0.7 | 3.6 ± 0.7 |
| 50 | 1459 | — | C15H24 (Sesquiterpene)cbdv70230-tbl2-note-0008 | — | 0.8 ± 0.2 | 1.5 ± 0.2 | 0.8 ± 0.1 |
| 51 | 1474 | — | C15H24 (Sesquiterpene)cbdv70230-tbl2-note-0008 | — | 0.3 ± 0.3 | 1.1 ± 0.1 | 0.3 ± 0.1 |
| 52 | 1479 | 1483cbdv70230-tbl2-note-0006 | (E)‐β‐Bergamotene | 15438‐94‐5 | 0.7 ± 0.5 | 2.6 ± 0.3 | 1.2 ± 0.1 |
| 53 | 1512 | 1505cbdv70230-tbl2-note-0005 | Cuparene | 16982‐00‐6 | 0.3 ± 0.1 | 0.5 ± 0.1 | 0.3 ± 0.1 |
| 54 | 1534 | 1524cbdv70230-tbl2-note-0005 | β‐Sesquiphellandrene | 20307‐83‐9 | 0.3 ± 0.0 | 0.6 ± 0.2 | 0.3 ± 0.1 |
Note: In bold, the main compounds (>2%).
Abbreviation: LRIs, linear retention indices.
Relative areas are calculated based on the average of three replicates for each of the three trees; tr: relative proportion is marked as trace for values <0.1%.
Database linear retention indices shown in this table for non‐polar column DB‐5MS are obtained according to standards of n‐alkanes (C6‐C30).
LRI in NIST 14 database Mainlib and Replib [34].
LRI in FFNSC 3 database [35].
Tentative identification made solely from mass spectrum evaluation.
Several possible isomers, mass spectra:[No.31] 41 (15), 42 (16), 43 (23), 55 (21), 56 (14), 84 (88), 86 (21), 87 (100), 97 (28), 112 (24); [No.44] 41 (18), 43 (17), 55 (11), 56 (66), 57 (100), 69 (7), 71 (20), 85 (11), 140 (11), 183 (6); [No.50] 41 (77), 55 (46), 69 (100), 79 (66), 91 (53), 93 (86), 105 (40), 107 (40), 119 (72), 161 (54); [No.51] 41 (38), 44 (31), 55 (31), 69 (100), 79 (34), 91 (42), 93 (62), 105 (30), 120 (37), 161 (47).
The two main volatile compounds obtained by HS‐SPME are hexanal (25.4% ± 3.4% to 27.4% ± 4.2%) and 2‐pentylfuran (3.1% ± 0.9% to 10.8 ± 1.3%), both also identified in the essential oil and derived from linoleic acid. Only 11 compounds were identified in both extracts obtained by the two extraction methods: nonanoic acid, hexanal, heptanal, an isomer of 2‐heptenal, 2‐pentylfuran, (E)‐2‐octenal, nonanal, (E)‐4‐nonenal, dodecane, decanal, nonanoic acid, and γ‐nonalactone. HD methods detected 10 terpenes/terpenoids, whereas 15 were detected through HS‐SPME extraction. Additionally, 13 aromatic compounds were identified in the HS‐SPME extract, whereas none were found in the essential oil. These results highlight the complementary nature of the two extraction methods, as HS‐SPME appears to be more effective in detecting highly volatile compounds that are less efficiently extracted by HD. The two isomers of 2,4‐decadienal were not observed in HS‐SPME. Noteworthy of mention, no compound was detected beyond β‐sesquiphellandrene, which has a molecular weight of 204.19 g/mol. Heavier, less volatile compounds such as farnesenes and oxidation derivatives containing more than 15 carbon atoms were not observed using HS‐SPME.
HD involves harsh conditions, such as high temperatures and water vapor, which can degrade sensitive compounds. In contrast, HS‐SPME operates under milder conditions, providing a more accurate volatile profile that closely reflects the real composition in the organism. HS‐SPME allowed the identification of a greater number of terpenes and aromatic compounds, while HD contains more degradation products; few terpenes, many oxygenated, unsaturated, and fatty acid compounds.
Main compounds identified in each extract are shown in Figure 3. Figure 4 compares the proportions of different classes of compounds depending on the extraction method. Linoleic acid, because it makes up most of the essential oil, has been omitted in Diagram A to facilitate comparison. As in the HD extract (excluding linoleic acid), aldehydes (12 compounds) are the dominant class of volatile compounds identified in HS‐SPME. Followed by unsaturated hydrocarbons (11), aromatic compounds (13), a furan (1), and saturated hydrocarbons (9). However, there are considerable differences in the composition of the two extracts.
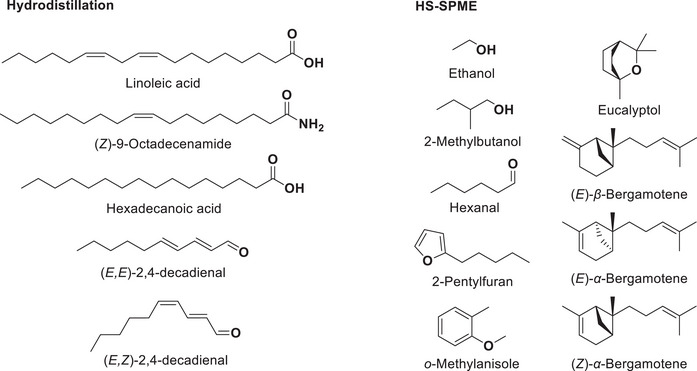
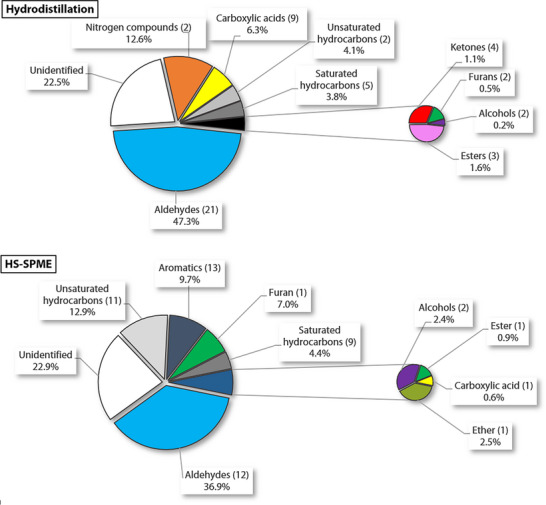
Aromatic compounds (13) include a variety of structures, such as arenes, oxygenated aromatics, and terpenes. The most abundant aromatic compounds are o‐methylanisole (1.8% ± 0.1% to 2.8% ± 0.3%) and o‐xylene (0.9% ± 0.3% to 1.7% ± 0.2%). A total of 12 aldehydes were identified in the headspace, hexanal (25% ± 3% to 27% ± 4%) being the most abundant compound, also identified in the essential oil, followed in smaller proportions by 3‐methylbutanal (1.4 ± 0.2 to 1.7% ± 0.6%) and acetaldehyde (1.2% ± 0.4% to 2% ± 1%). Various saturated hydrocarbons (9) were observed in small proportions, with the major compounds being tetradecane (0.64% ± 0.04% to 1.8% ± 0.4%) and dodecane (0.40% ± 0.04% to 1.7% ± 0.3%). The composition of terpenes in headspace portion differs significantly from what is observed in the essential oil and consists mostly of unsaturated hydrocarbons. A total of seven monoterpene compounds were identified, eucalyptol (1.5% ± 0.4% to 4.0% ± 0.5%) and d‐limonene (1.0 ± 0.2% to 1.3 ± 0.2%) being the most predominant. Additionally, compounds 18, 20, 21, and 22 belong to this class, but each exhibits two equally probable identification possibilities that are shown in Table 2. Sesquiterpenoids are more predominant in the headspace extract. Most are unsaturated hydrocarbons, with (E)‐α‐bergamotene (2.1% ± 0.2% to 4.8% ± 0.7%) being the most abundant, followed by other isomers such as (Z)‐α‐bergamotene (1.2% ± 0.2% to 2.0% ± 0.2%) and (E)‐β‐bergamotene (0.7% ± 0.5% to 2.6% ± 0.3%). Other notable compounds include α‐santalene (0.3% ± 0.1% to 0.5% ± 0.2%) and β‐sesquiphellandrene (0.26% ± 0.02% to 0.6% ± 0.2%). None of these compounds were detected in the essential oil of the hardwood in our study, suggesting that the HD process may cause oxidation and degradation of most terpenes [ref. 50], or that they are not observed because of their low concentration and the very important proportion of linoleic acid. These sesquiterpenes were previously identified in the B. papyrifera inner bark by another group using SPME extraction, with (E)‐α‐bergamotene as the major sesquiterpene accompanied by smaller amounts of (E)‐ and (Z)‐β‐farnesene and (Z)‐α‐bergamotene, which is consistent with our results [ref. 51].
Conclusions
We characterized the volatile compounds of B. papyrifera hardwood using HD and HS‐SPME/GC–MS. HD yielded an essential oil with a low average yield. Its profile is mainly dominated by linoleic acid and lipid oxidation products, suggesting limited potential for direct valorization of the trees through this method. In contrast, HS‐SPME allowed us to identify a different profile as it is more selective toward low molecular weight compounds than HD. This extraction method provided a more representative composition, most probably reflecting the volatile profile of the hardwood itself, as HD appears to cause the loss and degradation of certain compounds. HS‐SPME method revealed additional volatile compounds, including aromatics absent in the essential oil, as well as various terpenes, particularly sesquiterpenoids. Although these results suggest limited viability for extraction of volatile compounds from hardwood alone, future research could explore incorporating bark or using bark exclusively to enhance yields and compound diversity. Combining volatile compound extraction by HD with hot‐water extraction of phenolic compounds could enable a more integrated and sustainable process for the valorization of B. papyrifera residues in the forestry industry.
Experimental Section
Specimens
Three B. papyrifera trees were cut in St‐Julien, Quebec, Canada, using a chainsaw on August 3, 2022. The trees were alive and a few dozen meters apart in a mixed Eastern Great Lakes Lowland Forest. For each tree, three pieces at least 29 cm long, with diameters of 11 to 23 cm, were cut at different heights on the tree (bottom, middle, and top). The trees selected for the study are designated Trees 1, 2, and 3; the sampled sections are illustrated in the Supporting Information section. Their ages were determined by dendrochronology, and they are approximately 54, 36, and 83 years old, respectively. Branches and leaves were collected for identification purposes and deposited at the Louis‐Marie Herbarium at Université Laval (Entries # QFA0641221, QFA0641222, QFA0641223).
Sample Preparations
The fresh wood samples were immediately transported to the laboratory in plastic bags and stored in the freezer at −20°C. The bark was then removed, and the wood was cut into strips approximately 2.5 cm in diameter. We only kept the pieces in good condition, without visible blemishes (bark, rot, or coloring), for analysis. We then chipped the strips with a Yardworks wood shredder machine. The chips were stored at −20°C in plastic bags until they were analyzed. More details on the process can be found in the Supporting Information section. The wood chips, obtained from the three pieces of each individual tree, were evenly combined and labeled separately for each of the three trees. These steps were all carried within 24 h after cutting the trees.
Essential Oils Extraction by HD
The volatile compounds from the hardwood of B. papyrifera were extracted within 1 month of tree cutting using HD with a Clevenger‐type apparatus. In the process, 500 g of fresh wood chips were added to a 5 L flask containing 2.5 L of deionized water and heated to boiling for 5 h. The essential oil was collected by adding three portions of 5 mL diethyl ether to the Clevenger apparatus. The oil was then dried with Na2SO4, followed by filtration and evaporation under vacuum at room temperature using a rotary evaporator. A yellowish oil with a grass and chicken stock smell was obtained. The samples obtained were stored in glass vials at 4°C and protected from light until analysis. The essential oil yield (% w/w) was calculated from the dry mass.
Analysis of the Volatile Compounds Obtained by HD Using GC–MS and GC–FID
Volatile compounds in the samples were separated and identified using a Thermo Scientific GC–MS system (Trace GC Ultra with DSQ II detector) with a DB‐5MS non‐polar phase column (30 m × 0.25 mm × 0.25 µm). The carrier gas was helium flowed at a constant rate of 1 mL/min, a split ratio of 10, and an injector temperature of 240°C. The mass range was 40–400 Da, and the ionization energy was set at 70 eV, controlled by Xcalibur 4.0 software. The essential oil was diluted to 10% w/w in dichloromethane, then a 1.0 µL volume was injected in the instrument. The temperature program was set as follows: 50°C for 5 min, 2°C/min increase to 200°C, then to 270°C at 10°C/min, held at this temperature for 5 min.
We used Thermo Scientific’s QualBrowser software to process the data and identified compounds by comparing their mass spectra and linear retention indices (LRIs) to the commercial spectral libraries FFNSC 3 from Wiley (Flavor and Fragrance Natural and Synthetic Compounds) [ref. 35] and NIST 14 (National Institute of Standards and Technology) [ref. 34].
The standard solution for the determination of LRIs was prepared from a solution of C7 to C30 alkanes (Millipore Sigma, 49451‐U) diluted to 200 µg/mL in dichloromethane. The LRIs were then calculated using the Van den Dool and Kratz equations [ref. 52]. GC/FID data were used to calculate relative proportions. The percentage of each compound in the total extracts is expressed as the ratio of the area of each individual signal to the total integration area of the chromatogram. We manually integrated the signals without any correction factors. Each of the nine essential oils obtained was subject to a single analysis for identification purposes, and then to triplicate analyses in GC/FID for semi‐quantification. The results are expressed as relative proportions (% ± standard deviation) of volatile compounds grouped for all essential oils from the same tree.
The GC–FID used was a Thermo Trace GC Ultra (Thermo Scientific) with a Triplus autosampler. The column used, the injection conditions, and the temperature ramp are the same as those specified for GC/MS analyses. The FID temperature was 300°C, and gas flow rates were as follows: air = 350 mL/min; H2 = 35 mL/min; N2 = 30 mL/min.
Extraction of Volatile Compounds by Headspace Solid‐Phase Microextraction
We extracted the volatile compounds using an SPME fiber composed of a 50/30 µm layer of DVB/CAR/PDMS (Supelco 57328‐U). The SPME fiber was conditioned before analysis according to the supplier’s instructions. We ran a blank to check the cleanliness of the fiber and observed signals associated with siloxanes in all analyses; we therefore ignored them during integration. Approximately 2 g of fresh wood chips were introduced into a 20 mL SPME vial closed by a cap fitted with a silicone/PTFE septum. The vial was immersed in a water bath at 65°C for 5 min to allow headspace compounds to reach equilibrium. The fiber was then introduced and exposed to the headspace for 30 min. The compounds were finally desorbed manually in the injection port of the GC–MS. Each analysis was completed in triplicate to account for instrumental variability.
Analysis of Headspace Volatile Compounds by GC–MS
The volatile compounds were separated and analyzed using the same instrument, carrier gas, flow rate, ionization energy, and column employed in the analysis of the essential oils obtained by HD with GC–MS.
Thermal desorption of the compounds was carried out in the GC injector at 230°C for 5 min in splitless mode. The temperature program was set as follows: 40°C for 5 min, then an increase of 3°C/min to 140°C, then to 220°C at 12°C/min, and 5 more minutes at the final temperature. The mass range was 40–350 Da. The identification of the compounds was carried out with the same software and using the same method as described above for the essential oil obtained by HD.
The percentage of each compound in the total extraction product is expressed by the ratio of the area of each individual signal to the total integration area of the chromatogram. The signals were integrated manually and without the use of correction factors. Three analyses were conducted per tree for the wood chips of each of the three samples to illuminate aspects of both instrumental and intra‐tree variability. All analyses were completed within a week of the harvest.
Author Contributions
David Fortier did the sample collection, laboratory work, and analyses and wrote the first draft. Jean‐Christophe Séguin performed the preliminary work on the project, contributed to the conception of the project, and reviewed the manuscript. Normand Voyer conceived the project and research plan, supervised the lab work, obtained financial support, and reviewed the manuscript.
Conflicts of Interest
The authors declare no conflict of interest
Supplementary Materials
References
- White Birch (. 2024
- 2 S. Payette , Flore Nordique Du Québec Et Du Labrador (Presses De L’université Laval, 2015).
- 3 M. D. Williams , Identifying Trees of the East: An All‐Season Guide to Eastern North America, 1st ed. (Rowman & Littlefield, 2007).
- 4 L. Safford , J. C. Bjorkbom , and J. C. Zasada , Betula papyrifera Marsh. Paper Birch, in Silvics of North America: Hardwoods, (U.S. Department of Agriculture, Forest Service, 1990).
- Using Traditional Ecological Knowledge as a Basis for Targeted Forest Inventories: Paper Birch (Betula papyrifera) in the US Great Lakes Region,”. Journal of Forestry, 2014
- Portrait de la Ressource Forêt du Saguenay–Lac‐Saint‐Jean (. 2011
- 7 M. Bouchard , V. Vallée , and A. Boudreault , Identification d’Utilisations pour Le Bouleau À Papier et Le Peuplier Faux‐Tremble (Centre De Recherche Industrielle Du Québec (CRIQ), 2013).
- Carbon Emissions From the 2023 Canadian Wildfires,”. Nature, 2024. [PubMed]
- Estimating Air Pollutant Emissions From the 2024 Wildfires in Canada and the Impact on Air Quality,”. Gondwana Research, 2025
- Fire Return Intervals and Tree Species Succession in the North Shore Region of Eastern Quebec,”. Canadian Journal of Forest Research, 2008
- Non‐Wood Forest Products Based on Extractives—A New Opportunity for the Canadian Forest Industry Part 1: Hardwood Forest Species,”. Journal of Food Research, 2012
- Betulin and Its Derivatives as Novel Compounds With Different Pharmacological Effects,”. Biotechnology Advances, 2020. [PubMed]
- Betulin and Its Derivatives. Chemistry and Biological Activity,”. Chemistry for Sustainable Development, 2005
- Effective Method of Purification of Betulin From Birch Bark: The Importance of Its Purity for Scientific and Medicinal Use,”. PLoS ONE, 2016. [PubMed]
- Methods of Betulin Extraction From Birch Bark,”. Molecules (Basel, Switzerland), 2022. [PubMed]
- Birch Bark Research and Development,”. Natural Product Reports, 2006. [PubMed]
- Betulin Derivatives Effectively Suppress Inflammation In Vitro and In Vivo,”. Journal of Natural Products, 2016. [PubMed]
- Synthesis of Betulin Derivatives With New Bioactivities” (Doctoral diss.,. 2015
- Pharmacological Properties of the Ubiquitous Natural Product Betulin,”. European Journal of Pharmaceutical Sciences, 2006. [PubMed]
- Yield and Composition of Lipophylic Extracts of Yellow Birch (Betula alleghaniensis Britton) as a Function of Wood Age and Aging under Industrial Conditions,”. Holzforschung, 2006
- Sensory Characterization and Identification of Odorants in Birch Wood (Betula pendula Roth),”. Wood Science and Technology, 2024
- 22 K. Granström , Emissions of Volatile Organic Compounds From Wood (Karldstad University, 2005).
- Volatile Organic Compounds (VOCs) From Wood and Wood‐Based Panels: Methods for Evaluation, Potential Health Risks, and Mitigation,”. Polymers, 2020. [PubMed]
- 24 M. B. Jansson and N.‐O. Nilvebrant , Pulp and Paper Chemistry and Technology (De Gruyter, 2009).
- Parfum, Chimie et Creation (Actualite Chimique,. 2008
- 26 S. Clarke , Essential Chemistry for Aromatherapy, 2nd ed. (Elsevier Health Sciences, 2009).
- 27 X. Fernandez and F. Chemat , La Chimie Des Huiles Essentielles: Tradition et Innovation, 1st ed. (Vuibert, 2012).
- Stability of Essential Oils: A Review,”. Comprehensive Reviews in Food Science and Food Safety, 2013
- 29 N. F. S. Morsy , Active Ingredients From Aromatic and Medicinal Plants (InTech, 2017).
- Extraction of Natural Fragrance Ingredients: History Overview and Future Trends,”. Chemistry & Biodiversity, 2019
- 5th International Scientific Conference on Hardwood Processing (International Academy of Wood Science,. 2015
- Variability in Yield and Composition of Oil From Indian Sandalwood (Santalum album L.) Trees Grown in Homogeneous Conditions,”. Tropical Plant Research, 2019
- Hot‐Water Extraction Optimization of Sugar Maple (Acer saccharum Marsh.) and Red Maple (Acer rubrum L.) Bark Applying Principal Component Analysis,”. Journal of Wood Chemistry and Technology, 2017
- NIST 14, Mass Spectral Library (NIST/EPA/NIH) (. 2014
- 35 L. Mondello , Mass Spectra of Flavors and Fragrances of Natural and Synthetic Compounds, 3rd ed. (Wiley Blackwell, 2015).
- The Role of Dairy Products in Supplying Conjugated Linoleic Acid to Man’s Diet: A Review,”. Nutrition Research Reviews, 2001. [PubMed]
- Comparative Study of High‐Linoleic Acid Vegetable Oils for the Production of Conjugated Linoleic Acid,”. Journal of Agricultural and Food Chemistry, 2010. [PubMed]
- 38 H. Shi and C.‐T. Ho , Flavor of Meat and Meat Products ed. F. Shahidi (Springer US, 1994).
- Content of Trans,Trans ‐2,4‐Decadienal in Deep‐Fried and Pan‐Fried Potatoes,”. European Journal of Lipid Science and Technology, 2006
- Flavour Chemistry of Chicken Meat: A Review,”. Asian‐Australasian Journal of Animal Sciences, 2013. [PubMed]
- Palmitic Acid: Physiological Role, Metabolism and Nutritional Implications,”. Frontiers in Physiology, 2017. [PubMed]
- An Introduction to Plant Sphingolipids and a Review of Recent Advances in Understanding Their Metabolism and Function,”. New Phytologist, 2004. [PubMed]
- 43 K. D. Luttgeharm , A. N. Kimberlin , and E. B. Cahoon , Lipids in Plant and Algae Development (Springer International Publishing, 2016).
- Geraniol—A Review of a Commercially Important Fragrance Material,”. South African Journal of Botany, 2010
- Fragrance Material Review on Linalool,”. Food and Chemical Toxicology, 2003. [PubMed]
- 46 J. Chappell and R. M. Coates , Comprehensive Natural Products II: Chemistry and Biology (Elsevier, 2010).
- 47 E. M. Thurman , Comprehensive Analytical Chemistry (Elsevier, 2020).
- Betulin and Betulinic Acid: Triterpenoids Derivatives With a Powerful Biological Potential,”. Phytochemistry Reviews, 2019
- Selective Ultrasound‐Assisted Extractions of Lipophilic Constituents From Betula alleghaniensis and B. papyrifera Wood at Low Temperatures,”. Phytochemical Analysis, 2007. [PubMed]
- Thermal Degradation of Terpenes: Camphene, Δ3‐Carene, Limonene, and α‐Terpinene,”. Environmental Science & Technology, 1999
- Sesquiterpenes From the Inner Bark of the Silver Birch and the Paper Birch,”. Natural Products Communications, 2012
- A Generalization of the Retention Index System Including Linear Temperature Programmed Gas—Liquid Partition Chromatography,”. Journal of Chromatography A, 1963
