Crop steering through osmotic stress can reduce height but reduced yield in medical Cannabis
Abstract
Background:
Precision water stress, achieved via osmotic stress, has the potential to control plant size and improve crop quality without reducing yield, but results across species and cultivars have been inconsistent.
Methods:
This study examined the effects of elevated salinity on two diverse Cannabis sativa cultivars, Trump and Cherry. One group (control group) was grown at 4 mS cm⁻¹ (-0.14 MPa), one group at 8 mS cm⁻¹ (-0.28 MPa), and a third (hybrid) group at 8 mS cm⁻¹ (to reduce vegetative growth) until flowering and then reduced to 4 mS cm⁻¹ to minimize effect on flower yield.
Results:
Plant height was reduced 15% in both the high and hybrid treatments. Flower yield decreased by 20% in cv. ‘Trump’ in the hybrid treatment, but the decrease in yield in the high salinity treatment was not statistically significant. In cv. ‘Cherry’, flower yield declined by approximately 20% in both the constant high salinity and hybrid treatments. There was no difference in cannabinoid concentrations among treatments in either cultivar.
Conclusions:
These findings indicate that Cannabis sativa is highly tolerant to osmotic stress, but the response varies by cultivar. It is difficult to reduce plant size without reducing yield. There is no evidence that increased salinity altered cannabinoid concentration.
Article type: Research Article
Keywords: Abiotic stress, Flower yield, Cannabinoids, Salinity tolerance, Harvest index
Affiliations: https://ror.org/00h6set76grid.53857.3c0000 0001 2185 8768Crop Physiology Laboratory, Utah State University, Logan, UT 84322-4820 USA
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s42238-025-00351-2 | PubMed: 41254801 | PMC: PMC12625080
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.5 MB)
Introduction
Precision water stress is a holy grail of crop production because it can reduce plant size and increase quality. In a classic study, Boyer (ref. 1970) reported the potential of mild water stress to reduce leaf expansion without reducing photosynthesis in soybeans, sunflowers, and corn. Multiple studies over the past 50 years have reported mixed results when trying to replicate this study (Berni et al. ref. 2024).
Studies of precision water stress (often called “crop steering”) in Cannabis are limited but have been well studied in tomatoes. The results have been mixed. Wu and Kubota (ref. 2008) found that some tomato cultivars exhibited an increase in photosynthesis under moderate salinity (4.8 mS cm⁻¹), while higher salinity (8.4 mS cm⁻¹) reduced photosynthesis. Schwarz et al. (ref. 2002) reported reductions in both dry mass and photosynthesis as salinity increased. Romero-Aranda et al. (ref. 2000) reported a decrease in growth as salinity increased. Quality improvements under salinity stress have also been reported: increased sugar content (Agius et al. ref. 2022), improved fruit flavor (Dorai et al. ref. 2001; Chretien et al. ref. 2000; Wu et al. ref. 2004), with only slightly reduced yield. However, some studies noted reductions in fruit size (Dorai et al. ref. 2001) or fruit weight, as seen in strawberries (Awang et al., ref. 1995) and eggplant (Savvas et al., ref. 1999) under elevated salinity conditions.
While drought and salinity are distinct abiotic plant stressors, both reduce water availability by lowering water potential. Drought can occur either through matric potential or salinity through osmotic potential. As such, salinity can be used to create precise levels of osmotic stress. Salinity is often associated with sodium chloride salt (NaCl), but multiple types of salts and nutrients can be used. Salinity is typically measured using electrical conductivity (EC), which is a measure of dissolved ions in solution (Richards ref. 1954; Langenfeld et al. ref. 2022).
Yield responses of Cannabis to salinity have been limited and variable. Caplan et al. (ref. 2019) reported increased yield under elevated salinity, while Yep et al. (ref. 2020) observed yield reductions in hydroponic culture. Gill et al. (ref. 2022) found increased levels of proline, a known osmoprotectant, under drought conditions and noted that Cannabis can survive at extremely low soil moisture levels. Growth and yield responses can be variable among genetic lines. Cannabis has a wide genetic diversity that is poorly characterized. Both osmotic and hormonal changes can both contribute to yield responses to crop steering. The high commercial value of Cannabis has led to growers to use osmotic stress to increase cannabinoid content (Maravaneh et al., ref. 2022). Some studies have noted increases in cannabinoid content under moderate water stress (Caplan et al. ref. 2019), whereas others have reported no effect or reductions under severe water stress (Morgan et al. ref. 2024; Yep et al. ref. 2020; Duong et al. ref. 2023; Yuan et al. ref. 2024). These findings suggest a threshold, where moderate stress may enhance secondary metabolite production, but exceeding a threshold could diminish both biomass and cannabinoid content (Rezghiyan et al. ref. 2024; Morgan et al. ref. 2024). Similar patterns have been reported in other high-value crops. Peppermint and lemon verbena both showed reduced essential oil content under drought conditions, largely due to reduced plant size (Tabatabaie et al., ref. 2007).
Our objective was to quantify the effects of osmotic stress on Cannabis height, yield, harvest index and cannabinoid concentration in two diverse Cannabis cultivars.
Materials and methods
Propagation
Cuttings were propagated in 2 × 2 cm stone wool blocks (Grodan; Milton, ON). After 4 weeks of root development, they were placed into larger stone wool blocks (15 × 15 cm) and pruned to five nodes (Figs. 1 and 2). Two medical Cannabis cultivars (‘Trump’ and ‘Cherry’) were selected based on their commercial importance. In addition, cv. ‘Trump’ has thicker rigid stems and ‘Cherry’ has thinner, less rigid stems.


Growth conditions and experimental design
Plants were grown in a growth chamber for one week at an 18-hour photoperiod before changing to an inductive 12-hour photoperiod for the eight weeks until harvest. The LED lighting was adjusted weekly by reducing the electrical current, (dimming) to maintain a constant PPFD (photosynthetic photon flux density) at 1000 µmol m− 2 s− 1. The spectral distribution was 0.1% UV, 10.8% Blue, 21.4% Green, 66.2% Red, 1.6% Far red. The temperature was 27 °C/22°C day/night and the vapor pressure deficit was 1 kPa. CO2 was elevated to 1200 ppm.
Plants were divided into three treatment groups with six replicate plants per group. The first group was watered with a constant nutrient solution concentration of 4 mS cm− 1, the second group was watered with 8 mS cm− 1, and the third (hybrid) group was watered with 8 mS cm− 1 solution for the first four weeks of short-days and then watered with 4 mS cm− 1 for the remaining four weeks (Fig. 3). This hybrid treatment was selected because stem elongation occurrs during the first four weeks of growth. The goal was to reduce plant height without reducing yield. Tables 1 and 2 show the concentration of the nutrient solutions.
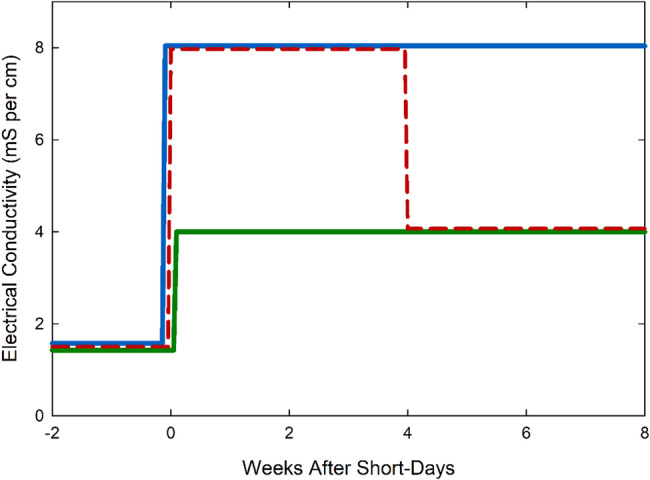
Table 1: Fertilizer salts and molar concentrations used to make the nutrient solutions. K2SiO3 and micronutrients were the same in both treatment solutions
| Macro | 4 mS cm− 1 (mM) | 8 mS cm− 1 (mM) |
|---|---|---|
| Ca(NO3)2 | 9 | 18 |
| KNO3 | 12 | 24 |
| KH2PO4 | 3 | 6 |
| MgSO4 | 2.4 | 4.8 |
| K2SiO3 | 0.6 | 0.6 |
| HNO3 | 3 | 6 |
Table 2: Elemental composition of nutrient solutions
| Macro | 4 mS cm− 1 (ppm) | 8 mS cm− 1 (ppm) |
|---|---|---|
| N | 462 | 925 |
| P | 93 | 186 |
| K | 633 | 1220 |
| Ca | 361 | 721 |
| Mg | 58 | 117 |
| S | 77 | 154 |
| Si | 17 | 17 |
Osmotic stress and water potential
Osmotic potential (Ψs) was calculated for the nutrient solution concentrations using the following equation (Richards ref. 1954; Langenfeld et al. ref. 2022):
\:EC\:\times\:\:-0.036={\Psi\:}s
\]
where EC is expressed in mS cm− 1 and Ψs is in MPa. Osmotic potential values for the treatments were: −0.14 MPa for 4 mS cm− 1, −0.29 MPa for 8 mS cm− 1, and − 0.22 MPa for 8/4 mS cm− 1. The osmotic potential for the 8/4 mS cm− 1 treatment was calculated by taking the mean value for the 8-week growth period.
\begin{aligned}&\frac{\left(-0.14\:MPa\:\times\:4\:weeks\right)+\left(-0.29\:MPa\:\times\:4\:weeks\right)}{8\:weeks}\\&=\:-0.22\:MPa \end{aligned}
\]
Based on van Genuchten (ref. 1980) model parameters reported by Chamindu et al. (ref. 2013), matric potential in stone wool at 50% VWC is approximately − 2.05 kPa (−0.002 MPa). Throughout the study, plants were maintained at or above field capacity (VWC > 50%), under which conditions matric potential (Ψm) was effectively near zero.
Irrigation was automated using electromagnetic sensors (model Teros 12, Meter Group; Pullman, WA) connected to a data acquisition system (model CR 1000x, Campbell Scientific, Logan UT). Due to the nature of stone wool blocks to turn hydrophobic if allowed to dry out after the start of irrigation, all blocks were kept above 50% volumetric water content (VWC). Irrigation frequency increased to several times an hour as the plant grew. Irrigation was more frequent in the day than in the night and larger plants used more water. VWC was monitored by moisture sensors, and VWC irrigation setpoints were established and periodically adjusted to ensure plants were not limited by water availability. Irrigation setpoints were used to turn irrigation on and off. For example, to control the setpoint at 55% VWC, irrigation would turn on for 1 s on, 15 s off, until VWC was above the 55% threshold, measured by the sensors. Each stone wool block had a sensor to ensure all plants did not have additional matric potential water stress (Fig. 1).
The stone wool blocks were placed on load cells (model LSP-5, Transducer Techniques; Temecula, CA) to provide a secondary way to monitor water content. Excess nutrient solution from the irrigation system drained free from the stone wool blocks, past the load cells, and into a collection tray. The leachate from three stone wool blocks (and subsequent plants), all from the same treatment, were collected in the same collection tray (n = 2).
Plants were inspected weekly for pests and were occasionally treated for spider mites (neem oil and Avid miticide) and powdery mildew (sulfur powder).
Growth and harvest measurements
Plant height was measured and recorded three times a week from the top of the stone-wool block to the highest point on the plant. Leachate was measured for electrical conductivity (model HI98304, Hanna Instruments; Woonsocket RI) after 10 min of irrigation (about a liter of solution in all treatments).
Plants were harvested 8 weeks after the start of short days. Stems, leaves, and flowers were separated and dried at 80 °C for two days. For each group, subsamples (n = 2 for leaves and flowers for nutrient analysis; n = 2 for flower cannabinoid analysis) were randomly chosen for nutrient and cannabinoid content (Tables 2 and 3).
Table 3: Yield and quality for the three treatments for both Cannabis cultivars ‘Trump’ and ‘Cherry’. Sample size for dry flower, harvest index, and height were: n = 6 (‘Trump’) and n = 4 (‘Cherry’). Sample size for CBD, THC, and CBD/THC were n = 2 (both cultivars). Differences among cannabinoid concentrations were not statistically significant
| Treatment EC(mS cm− 1) | Dry Flower (g m− 2) | Harvest Index | Height (mm) | CBD (%) | THC (%) | CBD/THC | |
|---|---|---|---|---|---|---|---|
| Trump | 4 | 690A | 0.56A | 618A | 11.6 | 0.39 | 30.5 |
| 8/4 | 558B | 0.59B | 522B | 11.1 | 0.39 | 28.8 | |
| 8 | 620AB | 0.59B | 544C | 11.1 | 0.37 | 31.4 | |
| Cherry | 4 | 706a | 0.48a | 498a | 8.0 | 0.20 | 40.0 |
| 8/4 | 560b | 0.52b | 440b | 7.4 | 0.18 | 41.6 | |
| 8 | 557b | 0.52ab | 423b | 8.5 | 0.23 | 38.8 |
Statistical analysis
Statistical analyses were performed in R version 4.4.1 (R Core Team, ref. 2024) within the RStudio environment (Posit Software, ref. 2024). Data management and visualization were carried out using ‘tidyverse’ (Wickham et al. ref. 2019). Analysis of variance (ANOVA) was conducted using the ‘car’ package (Fox & Weisberg, ref. 2019), and Tukey’s Honest Significant Difference (HSD) tests for pairwise post-hoc comparisons were implemented via the ‘multcomp’ package (Hothorn et al. ref. 2008).
Results and discussion
Plant growth and development are influenced by multiple abiotic stresses, including temperature, drought, and salinity, which disrupt homeostasis, inhibit photosynthesis, and impair water and nutrient uptake. These stressors can act independently or interact to amplify one another’s effects, ultimately reducing plant performance and compromising crop yield and productivity (Zhang et al. ref. 2022). By using a growth chamber, the effects of osmotic stress could be isolated from other environmental variables. The following results highlight how varying levels of osmotic stress influenced key agronomic traits, including plant height, flower yield, and crop quality.
Effects on plant height
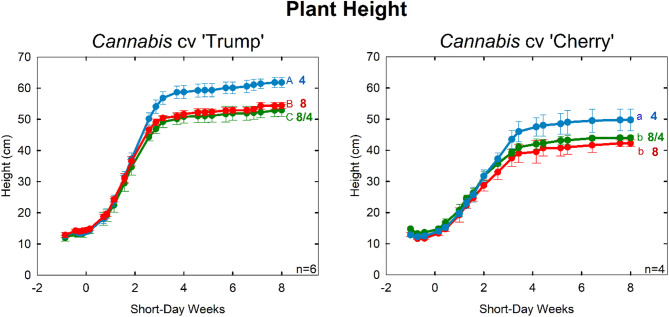
Plant height in the higher EC treatments was reduced by 15% in both cultivars (Table 3; Fig. 4). This height reduction demonstrates the potential of crop steering through osmotic stress. As expected, there was no significant height difference between the 8 mS cm−1 and 8/4 mS cm−1 treatment, indicating that osmotic stress during the flowering phase was not necessary for height reduction.
Effect on yield and quality
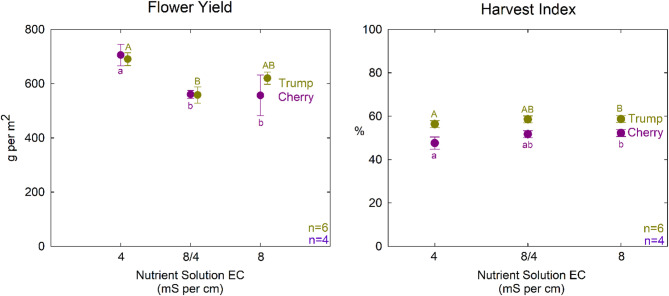
The higher osmotic stress treatments (8/4 mS cm−1 and 8 mS cm−1) resulted in a 20% reduction in flower yield for ‘Cherry’. In contrast, in ‘Trump,’ only the 8/4 mS cm−1 treatment significantly reduced yield, while the 8 mS cm−1 treatment was not different from the other treatments (Table 3; Fig. 5). This suggests that ‘Trump’ may be more tolerant of osmotic stress. Plants respond to osmotic stress in multiple ways. The genetic differences could be the result of a differing ability for osmotic adjustment or altered carbon partitioning to roots. Root size and depth are important considerations in the field, but the small root zone in these studies suggests that root system variability did not contribute to the differences between these cultivars.
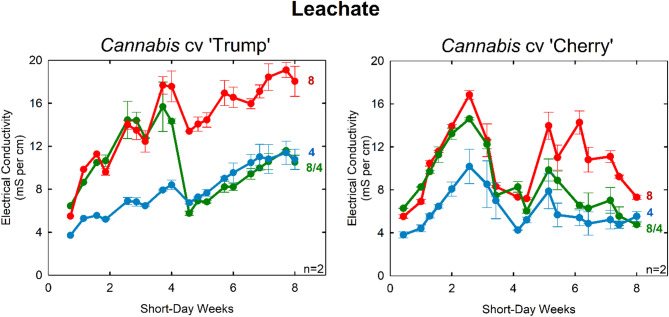
Notably, leachate collected from ‘Trump’ samples reached 20 mS cm⁻¹, corresponding to an osmotic stress of −0.72 MPa. This is nearly halfway to permanent wilting point (θwp = −1.5 MPa) (Fig. 6). This highlights the need for growers using osmotic stress for crop steering to maintain vigilance over irrigation management.
Interestingly, the harvest index in both cultivars was increased by the greater osmotic stress (Table 3; Fig. 5). This is due to the reduction in stem mass in the high EC treatments, despite a slight reduction in yield. This also suggests that osmotic stress during vegetative stages can steer biomass allocation toward flower production; a key consideration for optimizing yield in commercial Cannabis cultivation.
Cannabinoid content
There was less than a 1% difference in CBD concentration, and less than a 0.1% difference in THC concentration among treatments. The ratio of CBD/THC was also not significantly different among treatments in either cultivar (Table 3; Fig. 7). No trends were apparent among treatments. The uniformity of the controlled environment facilitated low experimental error among replicate measurements. Although there were only two replicate per cultivar per treatment, the consistent results in each cultivar indicate that osmotic effects on cannabinoids are likely small.
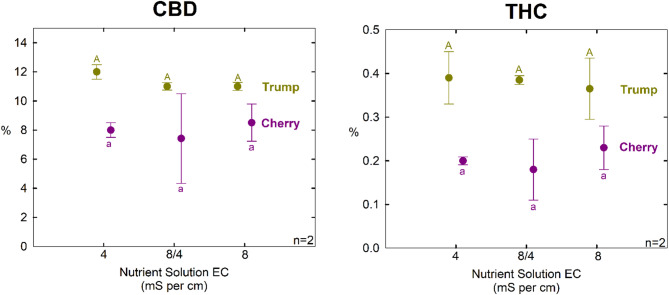
These results indicate the challenge of using crop steering to increase product quality.
Nutrient and media considerations
There were no statistically significant differences in leaf or flower nutrient concentrations among treatments and cultivars (Table 4). The small differences were not biologically important even had they been statistically significant. This indicates that the growth responses were the result of osmotic stress rather than nutrient availability or nutrient toxicity. These leaf tissue nutrient concentrations are within reported sufficiency ranges for Cannabis (Bryson and Mills ref. 2015; Landis et al. ref. 2019; Marschner ref. 2012). These results are consistent with a comprehensive study by Herschowitz et al. (ref. 2025) who also found no difference leaf nutrient concentrations of Cannabis between an EC of 2 and 4 mS cm−1 in hydroponic culture. Cannabis appears to have the ability to regulate nutrient uptake across a wide range of nutrient concentrations in the root-zone. In contrast, Rahnama et al. (ref. 2010) found that growth of wheat was initially inhibited by salt-induced osmotic stress but was affected by the toxicity of sodium chloride.
Stone wool was used as the growing medium in this study due to its widespread use commercial Cannabis cultivation. This has been largely driven by its high water holding capacity and low cation exchange capacity (Bussell et al., ref. 2004; Chamindu et al. ref. 2013) which make it well-suited as a media for precise water stress and crop steering. (Table 4)
Table 4: Effect of electrical conductivity and cultivar on nutrient concentration in leaf and flower tissue (n = 2). There were no significant effects in either cultivar or treatments
| Tissue | Cultivar | Treatment | N | P | K | Ca | Mg | S | Fe | Mn | Cu | Zn | B | Mo | Si |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % | mg kg− 1 | ||||||||||||||
| Flower | Trump | 4 | 3.7 | 0.8 | 3.7 | 2.0 | 0.5 | 0.9 | 137 | 122 | 14 | 80 | 31 | 3.5 | 380 |
| 8/4 | 3.7 | 0.9 | 3.5 | 2.2 | 0.5 | 0.7 | 177 | 108 | 16 | 80 | 33 | 3.7 | 576 | ||
| 8 | 3.7 | 0.8 | 3.3 | 1.9 | 0.4 | 0.7 | 118 | 121 | 15 | 86 | 33 | 3.8 | 464 | ||
| Cherry | 4 | 4.8 | 1.3 | 4.0 | 1.9 | 0.5 | 0.5 | 154 | 77 | 15 | 65 | 34 | 1.9 | 556 | |
| 8/4 | 4.9 | 1.3 | 3.9 | 2.1 | 0.5 | 0.5 | 184 | 113 | 18 | 81 | 38 | 2.2 | 748 | ||
| 8 | 4.9 | 1.3 | 4.3 | 2.0 | 0.5 | 0.5 | 164 | 125 | 18 | 91 | 36 | 1.9 | 791 | ||
| Leaf | Trump | 4 | 3.5 | 0.6 | 3.9 | 5.6 | 0.4 | 1.1 | 124 | 66 | 10 | 81 | 82 | 3.3 | 630 |
| 8/4 | 3.4 | 0.6 | 3.5 | 6.1 | 0.5 | 1.2 | 82 | 56 | 11 | 78 | 93 | 2.9 | 641 | ||
| 8 | 2.9 | 0.5 | 3.1 | 7.3 | 0.5 | 0.8 | 141 | 55 | 8 | 92 | 103 | 2.9 | 718 | ||
| Cherry | 4 | 2.2 | 0.5 | 1.9 | 9.1 | 1.1 | 0.4 | 172 | 68 | 8 | 23 | 144 | 2.1 | 965 | |
| 8/4 | 2.7 | 0.6 | 2.9 | 8.6 | 0.8 | 0.3 | 183 | 87 | 8 | 38 | 118 | 1.3 | 810 | ||
| 8 | 2.4 | 0.5 | 2.0 | 8.0 | 0.8 | 0.3 | 128 | 145 | 8 | 58 | 167 | 1.1 | 1293 | ||
Conclusion
These findings confirm that Cannabis sativa is highly tolerant to osmotic stress and indicate that osmotic stress during vegetative growth can reduce plant height and increase harvest index. However, yield was reduced 20% in ‘Trump’ in the hybrid treatment and consistently reduced by 20% in ‘Cherry’. Collectively, these results indicate that ‘Trump’ is more tolerant to osmotic stress than ‘Cherry’, suggesting the potential for genetic selection for salinity tolerance in Cannabis. There was no effect on cannabinoid concentration in either cultivar.
Future research should also explore fine-tuning the timing of osmotic stress to mitigate yield loss while preserving the benefits of height control.
References
- C Agius, S von Tucher, W Rozhon. The effect of salinity on fruit quality and yield of Cherry tomatoes. Horticulturae, 2022
- YB Awang, JG Atherton. Growth and fruiting response of strawberry plants grown on Rockwool to shading and salinity. Sci Hortic, 1995. [DOI]
- R Berni, M Thiry, J-F Hausman, S Lutts, G Guerriero. Eustress and plants: a synthesis with prospects for cannabis sativa cultivation. Horticulturae, 2024. [DOI]
- JS Boyer. Leaf enlargement and metabolic rates in corn, soybean, and sunflower at various leaf water potentials. Plant Physiol, 1970. [DOI | PubMed]
- GM Bryson, HA Mills. Plant analysis handbook. A guide to sampling, preparation, analysis, and interpretation for agronomic and horticultural crops, 2015
- WT Bussell, S McKennie. Rockwool in horticulture, and its importance and sustainable use in new Zealand. N Z J Crop Hortic Sci, 2004. [DOI]
- D Caplan, M Dixon, Y Zheng. Increasing inflorescence dry weight and cannabinoid content in medical cannabis using controlled drought stress. HortScience, 2019. [DOI]
- DT Chamindu, P Moldrup, M Tuller, M Pedersen, J Chen, LL Wollesen de Jonge. Gas diffusivity based design and characterization of greenhouse growth substrates. Vadose Zone J, 2013. [DOI]
- S Chretien, A Gosselin, M Dorais. High electrical conductivity and radiation-based water management improve fruit quality of greenhouse tomatoes grown in rockwool. HortScience, 2000. [DOI]
- R Core Team. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing. 2024. https://www.R-project.org/. Accessed 30 Mar 2025.
- M Dorai, A Papadopoulos, A Gosselin. Influence of electric conductivity management on greenhouse tomato yield and fruit quality. Agronomie, 2001. [DOI]
- H Duong, B Pearson, S Anderson, E Berthold, R Kjelgren. Variation in hydric response of two industrial hemp varieties (Cannabis sativa) to induced water stress. Horticulturae, 2023. [DOI]
- Fox J, Weisberg S, An R. Companion to Applied Regression. 3rd ed. Sage; 2019. https://www.john-fox.ca/Companion/. Accessed 30 Mar 2025.
- AR Gill, BR Loveys, JM Cowley, T Hall, TR Cavagnaro, RA Burton. Physiological and morphological responses of industrial hemp (Cannabis sativa L.) to water deficit. Ind Crops Prod, 2022. [DOI]
- JA Hershkowitz, FM Westmoreland, B Bugbee. Elevated root-zone P and nutrient concentration do not increase yield or cannabinoids in medical cannabis. Front Plant Sci, 2025. [DOI | PubMed]
- T Hothorn, F Bretz, P Westfall. Simultaneous inference in general parametric models. Biom J, 2008. [DOI | PubMed]
- H Landis, K Hicks, P Cockson, JB Henry, JT Smith, BE Whipker. Expanding leaf tissue nutrient survey ranges for greenhouse cannabidiol-hemp. Crop Forage Turfgrass Manage, 2019. [DOI]
- NJ Langenfeld, DF Pinto, JE Faust, R Heins, B Bugbee. Principles of nutrient and water management for indoor agriculture. Sustainability, 2022. [DOI]
- H Maravaneh, SJ Davarpanah. Study of cannabinoids Biosynthesis-related genes in hemp (Cannabis sativa L.) under drought stress by in vitro and in Silico tools. Appl Biotechnol Rep, 2022
- H Marschner. Mineral nutrition of higher plants, 2012
- W Morgan, J Singh, K Kesheimer, J Davis, A Sanz-Saez. Severe drought significantly reduces floral hemp (Cannabis sativa L.) yield and cannabinoid content but moderate drought does not. Environ Exp Bot, 2024. [DOI]
- Posit Software PBC, RStudio. Mar : Integrated Development Environment for R. Version 2024.12.0.467. Boston, MA. https://posit.co/. Accessed 30 2025.
- A Rahnama, RA James, K Poustini, R Munns. Stomatal conductance as a screen for osmotic stress tolerance in durum wheat growing in saline soil. Funct Plant Biol, 2010. [DOI]
- A Rezghiyan, H Esmaeili, M Farzaneh, H Rezadoost. The interaction effect of water deficit stress and nanosilicon on phytochemical and physiological characteristics of hemp (Cannabis sativa L). Plant Physiol Biochem, 2024. [DOI | PubMed]
- Richards LA, editor. Diagnosis and improvement of saline and alkali soils. USDA Handbook No. 60. 1954.
- R Romero-Aranda, T Soria, J Cuartero. Tomato plant-water uptake and plant-water relationships under saline growth conditions. Plant Sci, 2000. [DOI]
- D Savvas, F Lenz. Effects of NaCl or nutrient-induced salinity on growth, yield, and composition of eggplants grown in rockwool. Sci Hortic, 1999. [DOI]
- D Schwarz, HP Klaring, MW Van Iersel, KT Ingram. Growth and photosynthetic response of tomato to nutrient solution concentration at two light levels. J Am Soc Hortic Sci, 2002. [DOI]
- SJ Tabatabaie, J Nazari. Influence of nutrient concentrations and NaCl salinity on the growth, photosynthesis, and essential oil content of peppermint and lemon verbena. Turk J Agric For, 2007
- MT van Genuchten. A closed-form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci Soc Am J, 1980. [DOI]
- H Wickham, M Averick, J Bryan, W Chang, LD McGowan, R François. Welcome to the tidyverse. J Open Source Softw, 2019. [DOI]
- M Wu, C Kubota. Effects of electrical conductivity of hydroponic nutrient solution on leaf gas exchange of five greenhouse tomato cultivars. HortTechnology, 2008. [DOI]
- M Wu, SB Buck, C Kubota. Effects of nutrient solution EC, plant microclimate and cultivars on fruit quality and yield of hydroponic tomatoes (Lycopersicon esculentum). Acta Hortic, 2004. [DOI]
- B Yep, NV Gale, Y Zheng. Aquaponic and hydroponic solutions modulate NaCl-induced stress in drug-type cannabis sativa L.. Front Plant Sci, 2020. [PubMed]
- H Yuan, H Si, Y Ye, Q Ji, H Wang, Y Zhang. Arbuscular mycorrhizal fungi-mediated modulation of physiological, biochemical, and secondary metabolite responses in hemp (Cannabis sativa L.) under salt and drought stress. J Fungi, 2024. [DOI]
- H Zhang, J Zhu, Z Gong, JK Zhu. Abiotic stress responses in plants. Nat Rev Genet, 2022. [DOI | PubMed]
