Overexpressed MET drives aggressive thyroid cancer phenotypes and serves as a precision therapeutic target
Abstract
Supplementary Information:
The online version contains supplementary material available at 10.1038/s41598-025-23587-7.
Article type: Research Article
Keywords: MET, Thyroid cancer, Migration, EMT, Cancer, Biomarkers
Affiliations: https://ror.org/03xb04968grid.186775.a0000 0000 9490 772XDepartment of Biochemistry and Molecular Biology, Anhui Medical University, Hefei, 230032 China; https://ror.org/03t1yn780grid.412679.f0000 0004 1771 3402The Clinical Laboratory, the First Affiliated Hospital of Anhui Medical University, Hefei, 230011 Anhui China
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1038/s41598-025-23587-7 | PubMed: 41233563 | PMC: PMC12615610
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (4.5 MB)
Introduction
Thyroid carcinoma (THCA), which arises from thyroid follicular epithelial cells, represents the predominant endocrine malignancy globally1–3, and its incidence has increased steadily in recent decades1. Over 95% of THCA cases originate from follicular cells and typically represent indolent tumors amenable to cure through combined surgical resection and radioactive iodine ablation4,5. The 5-year survival rates are 93% and 88% for well-differentiated thyroid cancer in female and male patients, respectively6. Nevertheless, clinical challenges persist, particularly since a subset of patients is diagnosed with metastatic disease upon initial presentation. Although targeted therapies and immunotherapy are effective in some advanced cases7,8, many patients still lack effective treatment options9. This unmet clinical need underscores the urgent need for developing innovative therapeutic approaches to improve outcomes in thyroid cancer management.
The hepatocyte growth factor (HGF)/cellular mesenchymal-epithelial transition factor (c-MET) axis plays a pivotal role in diverse physiological processes, such as embryonic development, angiogenesis, and wound healing10. In cancer, this signaling cascade may become dysregulated through multiple mechanisms, including c-MET overexpression or gain-of-function mutations in its kinase domain. These aberrations result in constitutive activation of downstream pathways, such as mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK), phosphoinositide 3-kinase/protein kinase B (PI3K/AKT), signal transducer and activator of transcription (STAT), and inhibitor of kappa B alpha/nuclear factor kappa-light-chain-enhancer of activated B cells (IκBα/NF-κB). Ultimately, these dysregulated pathways collectively drive tumor proliferation, invasion, migration, survival, and metastasis11–14. Notably, both THCA cell lines and clinical specimens display significant c-MET overexpression. Although c-MET is largely absent in normal thyroid tissues, it is markedly upregulated in approximately 70–90% of papillary THCA samples15–17. These findings establish c-MET as a highly promising therapeutic target in THCA.
In summary, our pan-cancer analysis of TCGA data identified MET as exhibiting the highest expression level in THCA among all cancer types. Notably, elevated MET expression drives metastatic progression in thyroid cancer, a finding corroborated by clinical specimen analysis revealing a significant correlation between high MET expression and metastatic potential (P = 0.0002). Genetic silencing of MET markedly attenuated cancer cell migration capacity, concomitant with downregulation of epithelial–mesenchymal transition (EMT)-related signaling molecules and diminished activation of both STAT3 and ERK pathways. Moreover, we uncovered significant associations between MET expression and immune cell infiltration within the tumor microenvironment. Collectively, these multidimensional findings establish MET as a pivotal regulator of THCA progression, orchestrating metastasis, modulating oncogenic signaling pathways, and shaping the immune landscape.
Results
MET is upregulated in THCA
Utilizing the UALCAN database, we assessed MET expression across multiple cancer types. THCA exhibited the highest MET expression levels (Median = 7.38, n = 505) among all evaluated tumors (Fig. 1A). Notably, MET expression was significantly higher in THCA samples (n = 505) than in normal thyroid tissues (n = 59) (P < 1 × 10−12, Fig. 1B). Although Kaplan-Meier survival analysis indicated a non-significant trend (P = 0.11) toward worse prognosis in patients with high MET expression (n = 255) compared to the low-expression group (n = 255) (Fig. 1C), immunohistochemical analysis of our independent cohort (31 matched THCA-normal tissue pairs) confirmed consistent MET upregulation in tumors (Fig. 1D). These findings suggest that MET overexpression may contribute to THCA progression.
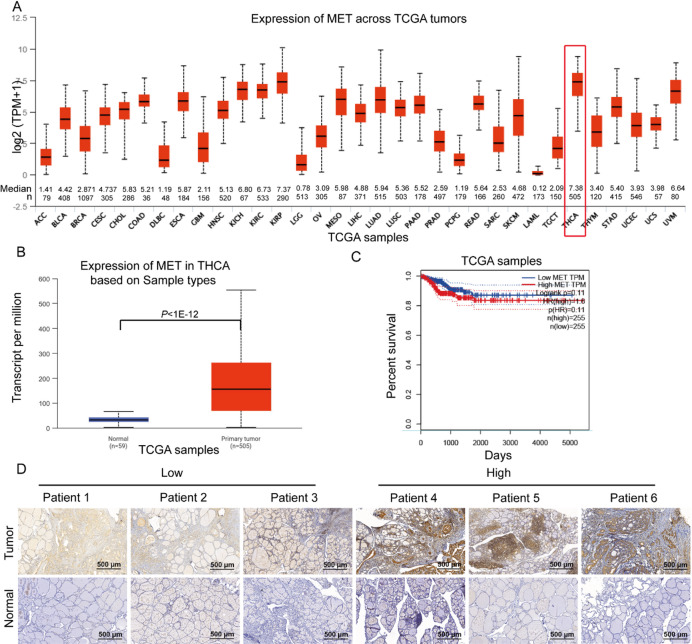
Elevated MET expression correlates with metastatic progression in thyroid cancer
Our findings revealed a positive correlation between MET expression and metastatic potential in THCA. Analysis demonstrated significantly higher MET expression in both male (n = 136) and female (n = 369) THCA patients compared to normal controls (n = 59), with no significant gender-based differences among THCA cases (Fig. 2A). Age-stratified evaluation showed consistent MET upregulation across all age groups (21–40 years: n = 175, P < 1 × 10−12; 41–60 years: n = 200, P < 1 × 10−12; 61–80 years: n = 103, P < 1 × 10−12; 81–100 years: n = 10, P = 3.68 × 10−2) compared to normal tissues, but revealed no significant age-dependent variation (Fig. 2B). Strikingly, MET expression increased progressively from normal tissues (n = 59) to N0 (n = 230, P < 1 × 10−12) and N1 (n = 58, P = 1.59 × 10−12) cases, with significantly elevated levels in N1 compared to N0 patients (P = 9.77 × 10−3) (Fig. 2C).
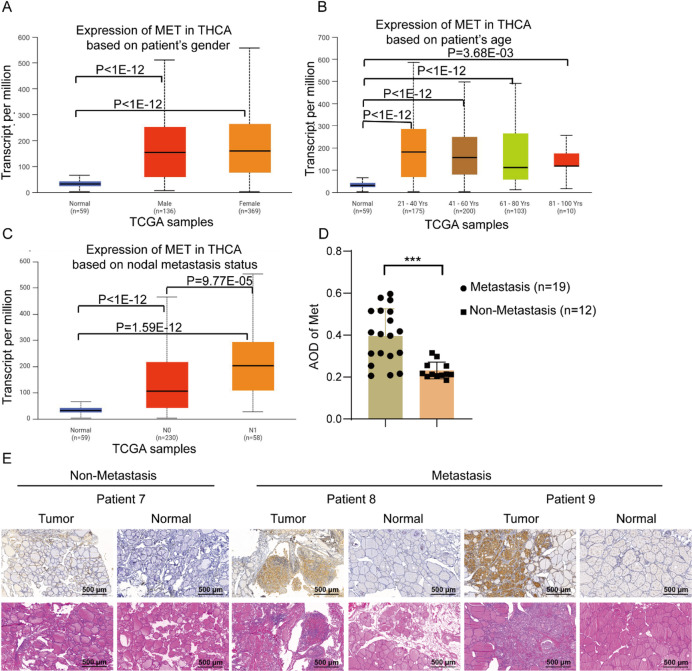
Immunohistochemical validation in 31 clinical specimens (12 non-metastatic vs. 19 metastatic cases) confirmed that high MET expression was strongly associated with metastatic status (P = 0.0002) (Fig. 2D,E). Representative cases illustrated increasing metastatic potential with higher MET expression (Patient 7 with metastasis vs. Patients 8–9 without metastasis). Taken together, these results strongly suggest that MET overexpression drives THCA progression and metastasis.
Knockdown of MET suppresses thyroid cancer cell migration in vitro
Employing RNAi knockout (sgRNA-3165 demonstrated the highest efficiency, Fig. 3A,B), we established MET-deficient thyroid cancer cell lines (BCPAP/MET−/− and TPC1/MET−/−) for functional analysis. Wound-healing assays demonstrated significantly greater scratch width (24 h) and reduced migration capacity in MET-knockout cells compared to wild-type controls (Fig. 3C). Consistent with these findings, Transwell assays revealed markedly decreased cell migration through membranes (Fig. 3D). Together, these results establish that MET is essential for thyroid cancer cell migration.
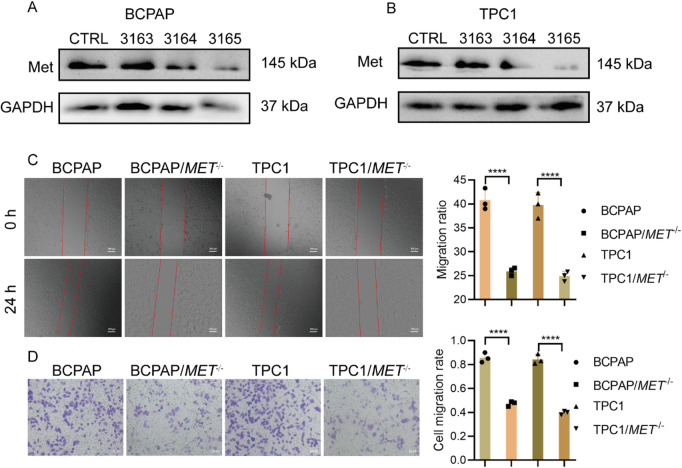
Knockdown of MET inhibits EMT and the ERK and STAT3 signaling pathways
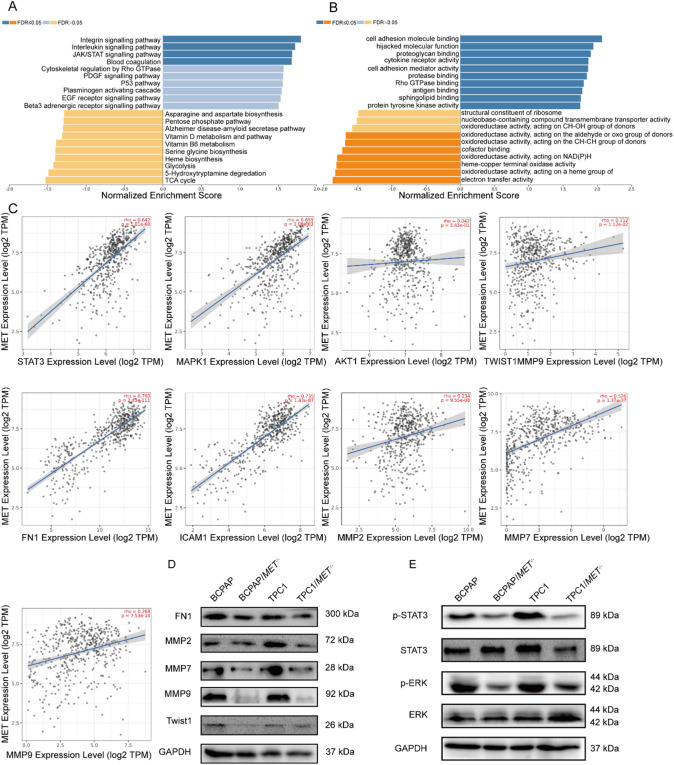
Gene Set Enrichment Analysis (GSEA) of MET-high THCA using LinkedOmics identified significant enrichment in critical oncogenic pathways, particularly JAK/STAT signaling (NES = 1.72, FDR q < 0.001) and cell adhesion molecule binding (NES = 1.58, FDR q < 0.01) (Fig. 4A,B). Subsequent correlation analysis via TIMER 2.0 revealed strong positive associations between MET expression and key metastasis-related molecules: STAT3 (r = 0.642, P = 1.81E−60), MAPK1 (r = 0.655, P = 1.08E−63), AKT1 (r = 0.042, P = 3.43E−01), Twist1 (r = 0.112, P = 1.12E−02), FN1 (r = 0.793, P = 2.43E−111), ICAM1 (r = 0.735, P = 1.43E−87), MMP2 (r = 0.234, P = 9.55E−08), MMP7 (r = 0.526, P = 1.37E−37), MMP9 (r = 0.268, P = 7.53E−10)(Fig. 4C). In MET-knockout cell lines (BCPAP/MET−/− and TPC1/MET−/−), we observed coordinated downregulation of EMT markers (FN1, MMP2/7/9, and Twist1) at the protein level (Fig. 4D). Mechanistic studies through phosphoprotein profiling demonstrated significant reduction in both STAT3 and ERK pathway activation in MET-deficient cells compared to wild-type controls (Fig. 4E). These findings define a MET-mediated oncogenic axis in THCA progression, wherein MET coordinately activates ERK/STAT3 signaling cascades, enhances expression of EMT-transcription factors, upregulates extracellular matrix remodeling proteins, and collectively driving tumor invasion and metastasis.
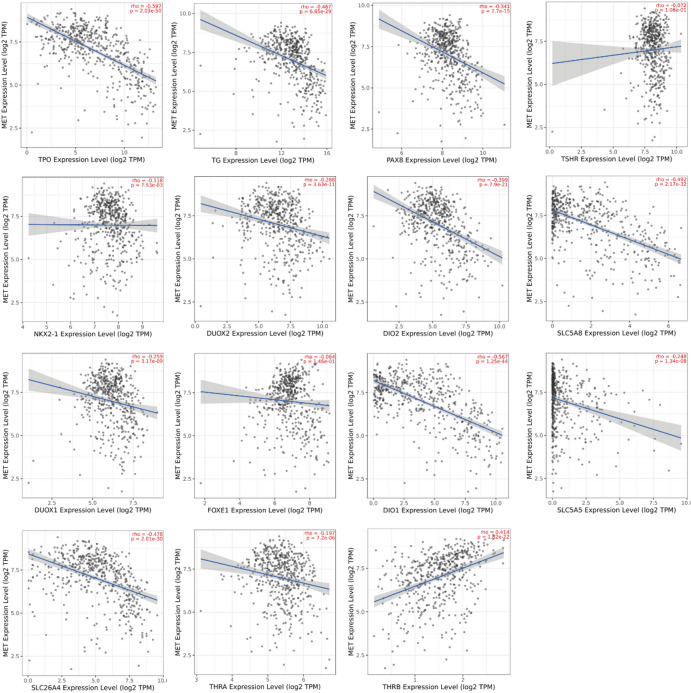
The association between MET expression and thyroid differentiation genes in TCGA
Given the critical role of loss of thyroid differentiation in papillary thyroid carcinoma (PTC) progression, we utilized TIMER 2.0 to systematically analyze the association between MET expression and 15 key thyroid differentiation genes. Our analysis revealed significant negative correlations between MET expression and most differentiation markers (Fig. 5), including thyroperoxidase (TPO, r = − 0.597, P = 2.03E−50), thyroglobulin (TG, r = − 0.467, P = 6.65E−29), paired box 8 (PAX, r = − 0.341, P = 2.7E−15), thyrotropin receptor (TSHR, r = − 0.072, P = 1.06E−01), NK2 homeobox 1 (NKX2-1, r = − 0.118, P = 7.53E−03), dual oxidase 2 (DUOX2, r = − 0.288, P = 3.63E−11), iodothyronine deiodinase 2 (DIO2, r = − 0.399, P = 7.9E−21), apical iodide transporter (SLC5A8, r = − 0.492, P = 2.17E−32), dual oxidase 1 (DUOX1, r = − 0.259, P = 3.17E−09), forkhead box E1 (FOXE1, r = − 0.064, P = 1.46E−01), iodothyronine deiodinase 1 (DIO1, r = − 0.567, P = 1.25E−44), sodium iodide symporter (SLC5A5, r = − 0.2488, P = 1.34E−08), pendrin (SLC26A4, r = − 0.478, P = 2.01E−30), and thyroid hormone receptor alpha (THRA, r = − 0.197, P = 7.2E−06). Notably, MET expression showed a unique positive correlation with thyroid hormone receptor beta (THRB, r = 0.414, P = 1.82E−22). These results demonstrate that elevated MET expression is strongly associated with reduced expression of thyroid differentiation markers, suggesting MET may drive tumor dedifferentiation in THCA. Collectively, our findings support a model wherein MET upregulation promotes THCA progression and metastasis by inducing a dedifferentiated state.
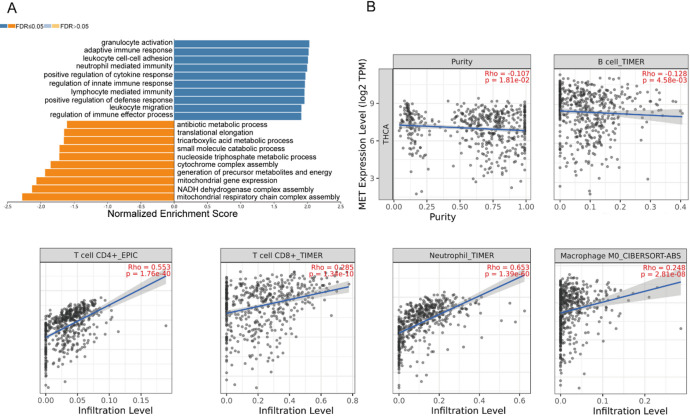
The correlation between MET expression and immune cell infiltration in THCA
GSEA of MET-high-expressing THCA using the LinkedOmics database identified significant enrichment in immune-related processes, most notably regulation of immune effector processes (Fig. 6A). Considering the critical role of the tumor immune microenvironment in cancer progression, we evaluated the correlation between MET expression and immune infiltration levels in THCA via the TIMER database. Remarkably, MET expression exhibited strong positive correlations with the abundance of tumor-infiltrating immune cells, including CD4+ T cells, CD8+ T cells, neutrophils, and macrophages (all P < 0.0001, Fig. 6B). These findings reveal a significant association between MET expression and immune cell infiltration in THCA, suggesting its potential involvement in modulating the tumor immune microenvironment.
Discussion
Thyroid cancer remains the most common endocrine malignancy, with its incidence having increased steadily in recent decades. Although improved diagnostic techniques may partly explain this trend, a true increase in both incidence and mortality cannot be ruled out18,19. Metastatic disease represents the main cause of THCA-associated morbidity and mortality20, affecting approximately 10% of papillary and up to 25% of follicular THCA patients21,22. The lungs (80%) and bones (25%) are the most frequent metastatic sites, and patients with multiple-organ metastases experience a sharp decline in 5-year survival rates from 77.6% to 15.3% compared to those with single-organ involvement23. These findings highlight the urgent need to better understand the molecular mechanisms underlying THCA progression, which may facilitate earlier detection and novel therapeutic approaches for advanced cases.
Numerous studies have demonstrated the oncogenic role of MET overexpression in various malignancies, including: mediating chemoresistance in non-small cell lung cancer24, conferring tamoxifen resistance in breast cancer25, promoting gastric cancer progression26, and facilitating hepatocellular carcinoma initiation and metastasis27,28. Our comprehensive pan-cancer analysis identified THCA as exhibiting the highest MET expression levels, with overexpression associated with shorter survival. It should be noted that the survival analysis of MET in the TCGA cohort lacked statistical significance, which may reflect inherent limitations of retrospective datasets, including insufficient follow-up duration, unaccounted treatment heterogeneity, or inadequate statistical power to detect subtle survival differences. Future prospective validation in treatment-standardized cohorts with extended follow-up is warranted to clarify the prognostic value of MET. These collective findings establish MET as a highly promising therapeutic target in THCA, meriting further mechanistic exploration and clinical development.
Metastasis constitutes a major cause of mortality in THCA23,29. Our multidisciplinary investigation elucidates MET’s pivotal role in THCA progression through three complementary lines of evidence: (1) Bioinformatic analysis of TCGA data established a significant association between MET overexpression and lymph node metastatic burden; (2) Histopathological validation in clinical specimens confirmed enhanced metastatic potential in MET-high tumors; (3) MET knockout in THCA cells substantially impaired migratory capacity. Complementary GSEA analysis linked MET to cell adhesion, EMT processes, and MAPK/STAT pathway activation. Notably, while the canonical HGF/c-MET pathway activates PI3K/AKT signaling, our study found no significant correlation between MET and AKT1 expression in thyroid cancer, suggesting tissue-specific pathway rewiring. Instead, MET appears to preferentially signal through STAT3 and ERK/MAPK, as evidenced by: (1) elevated phospho-ERK in MET-high tumors (Fig. 4E); (2) strong MET-STAT3 target gene correlations30 (Fig. 4C,E); and (3) epigenetic silencing of PIK3R1 that may decouple MET from PI3K signaling31. The maintained AKT homeostasis could be attributed to PTEN-mediated feedback or compensation by other AKT isoforms32. Importantly, measuring total AKT1 mRNA levels rather than phosphorylation status may underestimate pathway activity. These findings highlight the context-dependent nature of MET signaling and its alternative pathway utilization in thyroid cancer progression. Furthermore, MET expression showed coordinated regulation with established metastatic mediators (FN1, Twist1, ICAM1, MMP2/7/9), and its genetic suppression not only downregulated these effectors but also inhibited MAPK/STAT3 signaling. These integrated findings collectively establish MET as a key regulator of THCA metastasis, operating through MAPK/STAT3-mediated orchestration of metastatic effector molecules. This work provides a compelling mechanistic rationale for targeting MET in advanced THCA.
Loss of thyroid differentiation markers and impaired iodine avidity represent hallmark features of radioiodine-refractory THCA33,34. Analysis of TCGA data revealed significant co-expression patterns between MET and 14 key thyroid functional genes (including PAX8, TG, TPO, TSHR, NKX2-1, SLC5A5, SLC5A8, SLC26A4, FOXE1, THRA, DUOX1, DUOX2, DIO1, DIO2), while demonstrating a negative correlation with THRB. These genes are involved in iodine metabolism and thyroid-specific transcription, implicating MET in the regulation of thyroid cancer differentiation. The upregulation of THRB may act as a compensatory mechanism against MET-driven dedifferentiation, potentially mediated by MAPK-induced transcriptional activation of THRB or feedback resulting from reduced thyroid hormone production. Accumulating evidence underscores the prognostic significance of the tumor immune microenvironment in multiple cancers34. TIMER database analysis demonstrated significant positive correlations between MET expression and immune cell infiltration, particularly of CD8+ T cells, macrophages, and neutrophils in THCA.
Materials and methods
Data acquisition
Pan-cancer analysis of MET expression was performed using the TCGA database (https://cancergenome.nih.gov/). Differential expression of MET between normal and THCA tissues was assessed via the UALCAN database (https://ualcan.path.uab.edu/). The correlation between MET expression and THCA prognosis was evaluated using the GEPIA2 database (http://gepia2.cancer-pku.cn/#index). GSEA was conducted using the Hallmark and Kyoto Encyclopedia of Genes and Genomes (KEGG) gene sets (available at http://www.genome.jp/kegg/)35–37. Statistical significance was determined using the Benjamini–Hochberg method to adjust P-values, with thresholds set at an adjusted P < 0.05 and a false discovery rate (FDR) < 0.05.
Analysis of the thyroid differentiation genes
To explore potential functional links, we examined the relationship between MET expression and 15 key thyroid differentiation genes (PAX8, TG, TPO, TSHR, NKX2-1, SLC5A5, SLC5A8, SLC26A4, FOXE1, THRA, THRB, DUOX1, DUOX2, DIO1, and DIO2) using Spearman’s rank correlation analysis via Timer2.0 (http://timer.cistrome.org/). All correlation analyses were corrected for multiple testing using the Benjamini–Hochberg false discovery rate (FDR) method.
Human tissue samples
Paired THCA specimens (n = 31, including 19 metastatic and 12 non-metastatic cases) along with their matched normal tissues (sampled ≥ 2.5 cm from tumor margins) were collected from treatment-naïve THCA patients during surgical resection at The First Affiliated Hospital of Anhui Medical University. This study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University (Approval No. 20231337) and was conducted in compliance with the Declaration of Helsinki. All participants provided written informed consent.
Immunohistochemistry (IHC)
Formalin-fixed, paraffin-embedded (FFPE) tissue section (5 μm) were adhered to anti-detachment slides, dewaxed with xylene, and rehydrated through an ethanol gradient. Antigen retrieval was performed by heating the sections in citrate buffer (15 min, 95–100 °C) followed by natural cooling. Endogenous peroxidase was inactivated with 3% H2O2 (20 min, PV-9000 kit, reagent 1). Following serum blocking (20 min), slides were incubated with a primary anti-MET antibody (1:100 dilution, CST) overnight at 4 °C. The PV-9000 polymer detection system was employed: reaction-boosting solution (reagent 2) and HRP-conjugated secondary antibody (reagent 3) were applied sequentially (20 min each, room temperature). Chromogenic development was performed using DAB (5 min; Beyotime Biotechnology) with hematoxylin counterstaining and permanent mounting with neutral resin. Whole-slide imaging was performed using a Panoramic MIDI scanner (Jetta JD801 system, Jiangsu Jetta Technology). MET expression was semiquantitatively assessed by combined intensity/distribution scoring, with subsequent stratification into low (lower median) and high (upper median) expression cohorts for comparative analysis.
Cell culture and transfection
The human thyroid cancer cell lines BCPAP and TPC1 were acquired from the American Type Culture Collection (ATCC, Manassas, VA, USA) and routinely screened to maintain mycoplasma-free status. MET knockout lentiviruses were used to generate BCPAP/MET−/−, and TPC1/MET−/− according to the instructions from the manufacturer (Sangon Biotech (Shanghai) Co., Ltd.). The c-MET-targeting shRNA sequences used for lentiviral knockdown were as follows: pLVE3163(CCGGGCTGTGAGAATATACACTTACCTCGAGGTAAGTGTATATTCTCACAGCTTTTTTG), pLVE3164(CCGGCCTTCAGAAGGTTGCTGAGTACTCGAGTACTCAGCAACCTTCTGAAGGTTTTTTG), and pLVE3165(CCGGTCAACTTCTTTGTAGGCAATACTCGAGTATTGCCTACAAAGAAGTTGATTTTTTG). Thyroid cancer cells were infected with the lentiviruses at approximately 50% confluency. After 48 h, transfection efficiency was assessed via microscopy, and MET knockdown efficiency was verified by Western blot analysis. BCPAP and TPC1 cells were maintained in complete RPMI 1640 (HyClone, UT, USA) and DMEM (HyClone, UT, USA) media, respectively, supplemented with 10% fetal bovine serum (FBS; ExCell Bio, Suzhou, China) and 1% penicillin–streptomycin solution (Solarbio, Beijing, China). All cell cultures were incubated at 37 °C in a humidified 5% CO2 incubator.
Western blotting (WB)
Western blotting was performed as previously described38. Total protein lysates were separated on 10% SDS-polyacrylamide gels and transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA, USA) using a wet transfer apparatus. For immunodetection, membranes were incubated with the following primary antibodies: anti-GAPDH (Duoneng Biotechnology; 1:1000, loading control), anti-MET (CST; 1:1000), anti-ERK(CST; 1:1000), anti-p-ERK(CST; 1:1000), anti-STAT3 (CST; 1:1000), anti-p-STAT3 (CST; 1:1000), anti-FN1(CST; 1:1000), anti-MMP2(Abcam; 1:1000), anti-MMP7(Abcam; 1:1000), anti-MMP9(Abcam; 1:1000), and anti-Twist1(CST; 1:1000). After extensive washing, membranes were probed with appropriate HRP-conjugated secondary antibodies (Cell Signaling Technology; 1:2000) for 1 h at room temperature. Immunoreactive bands were detected using ECL substrate and visualized with a chemiluminescence imaging system (Clinx Science Instruments, Shanghai, China). Band intensities were quantified using ImageJ v.1.44p.
Wound healing assay
Wound healing assays were performed by plating cells in 12-well plates until reaching 90–100% confluence. Linear wounds were created in the monolayer using a sterile 10 µL pipette tip, maintaining uniform wound widths across replicates. Cell migration was monitored at baseline (0 h) and 24 h post-wounding using an inverted microscope. The wound closure rate was calculated as: Migration rate = [(W0 − W24)/W0] × 100, where W0 and W24 represent wound widths at 0 h and 24 h, respectively.
Transwell assay
Cell migration was evaluated using a 24-well Boyden chamber (Corning, Cat# 3422, NY, USA). Cells were harvested and adjusted to 5 × 104 cells/mL in serum-free medium, and 250 µL aliquots were seeded in the upper chamber. The lower chamber contained 750 µL of complete medium supplemented with 20% FBS as a chemoattractant. After 24 h incubation at 37 °C, non-migratory cells were removed from the upper chamber using cotton swabs. Membrane-adherent cells were fixed with methanol (20 min), stained with crystal violet (15 min), and quantified under an inverted microscope.
Immune cell infiltration analysis
The association between MET expression patterns and immune cell infiltration in THCA was analyzed using the Tumor Immune Estimation Resource (TIMER, https://cistrome.shinyapps.io/timer/). This validated bioinformatics platform enables comprehensive profiling of immune cell abundances and their molecular correlations across diverse malignancies. Specifically, we evaluated infiltration levels of five major immune subsets: B cells, CD4+ T cells, CD8+ T cells, neutrophils, and macrophages.
Statistical analysis
All statistical analyses were performed using GraphPad Prism 8.4.0. Intergroup differences were assessed using unpaired Student’s t-tests for comparisons between two groups or one-way ANOVA for multiple group comparisons. Survival analysis was conducted using the Kaplan–Meier method with log-rank testing. To evaluate associations between variables, we utilized Spearman’s rank correlation analysis. A two-tailed P-value < 0.05 was considered statistically significant.
Conclusions
This study provides a comprehensive elucidation of MET’s oncogenic role in THCA progression through multi-modal investigations. Bioinformatics profiling demonstrated that MET overexpression in THCA is associated with shorter survival and aggressive metastatic behavior, observations corroborated by immunohistochemical analysis of clinical samples. Functional characterization revealed that MET knockdown substantially attenuates THCA cell migration, mediated through ERK/STAT3 signaling-dependent modulation of metastatic effectors. Notably, we established novel associations between MET overexpression and both loss of thyroid differentiation markers and altered immune cell infiltration patterns. Collectively, these findings establish MET as a multifaceted regulator of THCA pathogenesis, offering compelling evidence for pursuing MET-targeted therapies in advanced disease.
Supplementary Materials
References
- DW Chen, BHH Lang, DSA McLeod, K Newbold, MR Haymart. Thyroid cancer. Lancet, 2023. [DOI | PubMed]
- L Boucai, M Zafereo, ME Cabanillas. Thyroid cancer: A review. JAMA, 2024. [DOI | PubMed]
- H Sung. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin., 2021. [DOI | PubMed]
- KA Araque, S Gubbi, J Klubo-Gwiezdzinska. Updates on the management of thyroid cancer. Horm. Metab. Res., 2020. [DOI | PubMed]
- D Laha, N Nilubol, M Boufraqech. New therapies for advanced thyroid cancer. Front. Endocrinol. (Lausanne), 2020. [DOI | PubMed]
- F Nabhan, PH Dedhia, MD Ringel. Thyroid cancer, recent advances in diagnosis and therapy. Int. J. Cancer, 2021. [DOI | PubMed]
- ME Cabanillas, M Ryder, C Jimenez. Targeted therapy for advanced thyroid cancer: kinase inhibitors and beyond. Endocr. Rev., 2019. [DOI | PubMed]
- 8.Agate, L. et al. Nutrition in advanced thyroid cancer patients. Nutrients. 1410.3390/nu14061298 (2022).
- Y Tao, P Li, C Feng, Y Cao. New insights into immune cells and immunotherapy for thyroid cancer. Immunol. Investig., 2023. [DOI | PubMed]
- L Trusolino, A Bertotti, PM Comoglio. MET signalling: principles and functions in development, organ regeneration and cancer. Nat. Rev. Mol. Cell. Biol., 2010. [DOI | PubMed]
- E Gherardi, W Birchmeier, C Birchmeier, G Vande Woude. Targeting MET in cancer: rationale and progress. Nat. Rev. Cancer, 2012. [DOI | PubMed]
- Y Lan. ITGA3-MET interaction promotes papillary thyroid cancer progression via ERK and PI3K/AKT pathways. Ann. Med., 2025. [DOI | PubMed]
- CD Mohan. c-MET pathway in human malignancies and its targeting by natural compounds for cancer therapy. Phytomedicine, 2024. [DOI | PubMed]
- BH Naguib. Fragment-based design and synthesis of coumarin-based thiazoles as dual c-MET/STAT-3 inhibitors for potential antitumor agents. Bioorg. Chem., 2024. [DOI | PubMed]
- R Mineo. Activation of the hepatocyte growth factor (HGF)-Met system in papillary thyroid cancer: biological effects of HGF in thyroid cancer cells depend on Met expression levels. Endocrinology, 2004. [DOI | PubMed]
- JD Bergstrom, A Hermansson, T Diaz de Stahl, NE Heldin. Non-autocrine, constitutive activation of Met in human anaplastic thyroid carcinoma cells in culture. Br. J. Cancer, 1999. [DOI | PubMed]
- Y Zhou. Off-target effects of c-MET inhibitors on thyroid cancer cells. Mol. Cancer Ther., 2014. [DOI | PubMed]
- CK Jung. The increase in thyroid cancer incidence during the last four decades is accompanied by a high frequency of BRAF mutations and a Sharp increase in RAS mutations. J. Clin. Endocrinol. Metab., 2014. [DOI | PubMed]
- M Zou. Genome-wide transcriptome analysis and drug target discovery reveal key genes and pathways in thyroid cancer metastasis. Front. Endocrinol. (Lausanne), 2025. [DOI | PubMed]
- XY Zhang. Clinical outcomes and prognostic factors in patients with no less than three distant organ system metastases from differentiated thyroid carcinoma. Endocrine, 2019. [DOI | PubMed]
- 21.Toraih, E. A. et al. Site-specific metastasis and survival in papillary thyroid cancer: the importance of brain and multi-organ disease. Cancers (Basel). 1310.3390/cancers13071625 (2021).
- N Hugen. Divergent metastatic patterns between subtypes of thyroid carcinoma results from the nationwide Dutch pathology registry. J. Clin. Endocrinol. Metab., 2020. [DOI | PubMed]
- LY Wang. Multi-organ distant metastases confer worse disease-specific survival in differentiated thyroid cancer. Thyroid, 2014. [DOI | PubMed]
- M Sakamoto, T Patil. MET alterations in advanced non-small cell lung cancer. Lung Cancer, 2023. [DOI | PubMed]
- X Lai. HGF/c-Met promotes breast cancer Tamoxifen resistance through the EZH2/HOTAIR-miR-141/200a feedback signaling pathway. Mol. Carcinog., 2025. [DOI | PubMed]
- H Lin. Characterization of bozitinib as a potential therapeutic agent for MET-amplified gastric cancer. Commun. Biol., 2025. [DOI | PubMed]
- Y Zhou. Loss of TP53 cooperates with c-MET overexpression to drive hepatocarcinogenesis. Cell. Death Dis., 2023. [DOI | PubMed]
- Y Yu. Fis1 phosphorylation by Met promotes mitochondrial fission and hepatocellular carcinoma metastasis. Signal. Transduct. Target. Ther., 2021. [DOI | PubMed]
- Q Liu. Factors involved in cancer metastasis: a better Understanding to seed and soil hypothesis. Mol. Cancer, 2017. [DOI | PubMed]
- WH Lin. STAT3 phosphorylation at Ser727 and Tyr705 differentially regulates the EMT-MET switch and cancer metastasis. Oncogene, 2021. [DOI | PubMed]
- YH Chu. Clinicopathologic features of kinase fusion-related thyroid carcinomas: an integrative analysis with molecular characterization. Mod. Pathol., 2020. [DOI | PubMed]
- L Wei, Q Liang, C Zhou, R Liu, Y Liu. PTEN inhibits epithelial mesenchymal transition of thyroid cancer cells by regulating the Wnt/beta-Catenin signaling pathway. Discov Oncol., 2024. [DOI | PubMed]
- CS Fuziwara, ET Kimura. MicroRNAs in thyroid development, function and tumorigenesis. Mol. Cell. Endocrinol., 2017. [DOI | PubMed]
- 34.Liang, W. et al. High expression of COL8A1 predicts poor prognosis and promotes EMT in papillary thyroid cancer. Endocr. Connect.1310.1530/EC-24-0279 (2024).
- M Kanehisa, Y Sato, M Kawashima, M Furumichi, M Tanabe. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res., 2016. [DOI | PubMed]
- M Kanehisa, S Goto. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res., 2000. [DOI | PubMed]
- M Kanehisa, M Furumichi, Y Sato, Y Matsuura, M Ishiguro-Watanabe. KEGG: biological systems database as a model of the real world. Nucleic Acids Res., 2025. [DOI | PubMed]
- R Xu. c-Met up-regulates the expression of PD-L1 through MAPK/NF-kappaBp65 pathway. J. Mol. Med. (Berl), 2022. [DOI | PubMed]
