Abnormalities of the endocannabinoid system produce piercing nuclear hernias in migrating cerebral neurons
Abstract
We are reporting powerful streams of chromatin rupturing the nuclear envelope (NE) and the plasma membrane of migrating cerebral neurons in mouse embryos, which we suggest naming “piercing nuclear hernia” (PNH). About 40% of migrating neurons in cannabinoid type 1 receptor knock-out (CB1R−/−) mouse embryos and in wildtype embryos exposed to CB1R agonists show NE rupture and/or PNH. This indicates that deviations from optimal functioning of the endocannabinoid system in under- or over-activity may trigger analogous mechanisms increasing intranuclear pressure and chromatin herniation. The cells from CB1R−/− embryos showed pronounced ultrastructural disorders, such as high volume of herniated chromatin, mitochondrial fission, and negative correlation of the mitochondrial length with the volume of herniated chromatin. Catastrophic rupture of the nuclear and plasma membranes may provoke accidental cell death. At the same time, a fraction of neurons with PNH showed generally normal ultrastructure, which could indicate a mechanism of cell body repair.
Article type: Research Article
Keywords: Biological sciences, Cellular neuroscience, Developmental neuroscience, Natural sciences, Neuroscience
Affiliations: Department of Neuroscience, Yale University School of Medicine and Kavli Institute for Neuroscience, New Haven, CT 06510, USA
License: CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.isci.2025.113188 | PubMed: 40799395 | PMC: PMC12341634
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.8 MB)
Introduction
The endocannabinoid system has been identified in numerous phylogenetically distant animal clades, indicating it as an evolutionarily conserved trait.ref. bib1,ref. bib2,ref. bib3,ref. bib4,ref. bib5,ref. bib6 It is involved in several cognitive and physiological processes, including cardiovascular regulation, fertility, pregnancy, prenatal and postnatal development, activity of the immune system, food consumption, and energy metabolism.ref. bib7,ref. bib8,ref. bib9,ref. bib10,ref. bib11,ref. bib12,ref. bib13,ref. bib14,ref. bib15 Endocannabinoid signaling via cannabinoid type 1 receptor (CB1R) often participates in retrograde synaptic modulation but also involves non-synaptic pathways.ref. bib16,ref. bib17,ref. bib18 In addition to abundant expression in the axonal plasma membrane, CB1R accumulates in the membranes of intracellular vesicles in the cell bodies of certain types of adult and immature neurons.ref. bib19,ref. bib20,ref. bib21 One demonstrated role of CB1R, which putatively involves the intracellular vesicles, is self-inhibition of cholecystokinin-expressing interneurons and a subpopulation of cortical pyramidal neurons through the cell’s autonomous Ca2+-dependent production of endocannabinoids and K+ channel activation.ref. bib22,ref. bib23 The endocannabinoid system influences synapse target selection by pyramidal neurons and GABAergic interneurons through CB1R internalization from axonal filopodia and chemorepulsion of the growth cones.ref. bib24,ref. bib25 We recently detected initial expression of CB1R in immature projection neurons vertically migrating through the cerebral intermediate zone and cortical plate, as well as in interneurons tangentially migrating through the marginal zone. We also demonstrated that, in CB1R−/− embryos, migrating cerebral projection neurons emitted an increased number of processes and deviated from the vertical orientation, implicating CB1R’s role in cellular migration.ref. bib21 However, molecular mechanisms of the endocannabinoid system in the cytoarchitecture of the developing brain are still enigmatic.
Therapeutic benefits of cannabis as an analgesic and antiemetic agent are well-known and applied for centuries.ref. bib26,ref. bib27 Inhibition of MAGL (monoacylglycerol lipase – enzyme degrading endocannabinoid 2-arachidonyl glycerol) has been proposed as a potential therapeutic approach for treatment of diverse neurological and neurodegenerative diseases, such as multiple sclerosis, Alzheimer’s disease, Parkinson’s disease, amyotrophic lateral sclerosis, and traumatic brain injury.ref. bib28,ref. bib29,ref. bib30,ref. bib31,ref. bib32,ref. bib33,ref. bib34 Cannabinoid-based anti-epileptic and anti-spasticity medications such as Epidiolex and nabiximols (Sativex) have demonstrated efficacy in randomized controlled trials.ref. bib35,ref. bib36 Two cannabinoid drugs (dronabinol and nabilone) are approved by the US Food and Drug Administration for the prevention or treatment of cancer-related side effects, such as nausea and vomiting. In vitro and in vivo experiments on cancer models show that cannabinoids can kill cancer cells, making cannabis-based medicine a promising option to effectively modulate growth of certain tumor types. However, suspected anticancer benefits of cannabinoids have yet to be confirmed in clinical trials.ref. bib37,ref. bib38,ref. bib39,ref. bib40,ref. bib41,ref. bib42,ref. bib43 Medical cannabis products are approved or tolerated in many countries and, increasingly, self-administered by pregnant individuals seeking relief from pain, nausea, or depression. The use of marijuana and synthetic cannabinoids during pregnancy and lactation poses potential risks for intrauterine and postnatal brain development, particularly as cannabinoids can appear in breast milk. In the absence of exhaustive biomedical studies, the extent to which marijuana and medicinal cannabinoids affect immature brains is still the subject of debate [Reviewed inref. bib14,ref. bib26,ref. bib44,ref. bib45].
The nuclear envelope (NE) consists of the inner and outer nuclear membranes (INM and ONM, respectively) and the nuclear lamina – a composite matrix assembled on the inner nuclear membrane. The main function of the NE is to allow molecular exchange between the nucleoplasm and the cytoplasm through specified nuclear pores. Recent works highlighted the dynamic properties of the nuclear membranes and demonstrated that dysregulation of their functions has significant consequences for the cell.ref. bib46,ref. bib47,ref. bib48,ref. bib49,ref. bib50 In addition to biochemical inputs, nuclei of the cells may be exposed to intrinsic and extrinsic mechanical forces transmitted by the cytoskeleton and nucleoskeleton that trigger dynamic changes in nuclear morphology.ref. bib46,ref. bib48,ref. bib49,ref. bib51 Cytoskeleton components (such as centrosome, microtubules, actin, myosin II, dynein, etc.) fulfill a crucial role in nucleus translocation, as demonstrated in migrating neurons.ref. bib52,ref. bib53,ref. bib54,ref. bib55 Emerging data suggests that extensive deformation of the cell and its nucleus during constrained migration transmits substantial physical stress on the NE and may result in its rupture, which in turn leads to chromatin herniation, uncontrolled exchange of nucleo-cytoplasmic content, DNA damage, and cell death.ref. bib46,ref. bib56,ref. bib57,ref. bib58,ref. bib59 Distribution of a diffusible fluorescent marker from ruptured nucleus through the cytoplasm was observed as early as minutes after NE rupture, indicating the high speed of the reaction.ref. bib57,ref. bib60,ref. bib61,ref. bib62,ref. bib63 The full impact of nucleo-cytoplasmic mixing is likely to be extensive yet poorly understood. Ruptures of the NE have been observed in several species in vitro and in vivo indicating that it is a consequence of the environmental conditions rather than a species-specific phenomenon [Reviewed inref. bib64]. For example, NE rupture was observed in neurons migrating during brain development.ref. bib56,ref. bib65 Further, NE ruptures were extensively studied in many cancer cell lines during cell migration through tightly constricted areas.ref. bib47,ref. bib57,ref. bib58,ref. bib60 NE ruptures can be induced in non-tumorigenic cell lines by generating a lamin B1 deficiency or depletion of two major tumor suppressors, p53 and retinoblastoma (Rb1).ref. bib56,ref. bib66,ref. bib67 Cultivated cells also can be provoked to experience NE rupture by external mechanical force,ref. bib51,ref. bib58,ref. bib68 human immunodeficiency virus infection,ref. bib62 and activation of apoptosis, inflammation, or autophagy.ref. bib66,ref. bib69,ref. bib70,ref. bib71 Depletion, or mutation of nuclear lamin proteins as well as conditions that impair the connections between the INM and the chromatin increase the probability of NE ruptures.ref. bib47,ref. bib48,ref. bib56,ref. bib57,ref. bib59,ref. bib60,ref. bib68,ref. bib72,ref. bib73,ref. bib74,ref. bib75 Thus, NE ruptures may be caused by defects in lamina organization, or increased intranuclear pressure transmitted by actin cytoskeleton and/or nucleoskeleton.ref. bib72 Several NE repair mechanisms have been proposed, including attachment of endoplasmic reticulum sheets to the exposed chromatin, spreading of the preserved ONM, plugging using membrane fragments, and resealing by protein complexes [Reviewed inref. bib64,ref. bib76]. Migrating cells reseal their ruptured NE using ESCRT-III (the endosomal sorting complexes required for transport III) and by doing so reduce DNA damage and cell death.ref. bib57,ref. bib58 An individual NE rupture may persist for minutes to hours before repair, however unrepaired NE ruptures eventually lead the cells to death.ref. bib57,ref. bib58,ref. bib60,ref. bib61,ref. bib77
Here, extending our previous study of the role of endocannabinoid signaling in the developing brain,ref. bib20,ref. bib21,ref. bib24,ref. bib78,ref. bib79 we performed extensive electron microscopy analysis with 3D reconstruction of migrating neurons in CB1R−/− embryos, as well as after in utero application of CB1R agonists in wildtype mice. Morpho-functional conditions of mitochondria correlate with general cellular functionality, while swelling of the mitochondrial matrix and predominance of fission over fusion may serve as evidence of disordered cellular energetics or other malfunctions.ref. bib80,ref. bib81,ref. bib82 This inspired our choice of ultrastructural and morphometric analyses of mitochondria as an indicator for potential cell disorder. We unexpectedly discovered a correlation between the frequency of chromatin herniation and disorder of the endocannabinoid system, which reveals a possible cellular mechanism for known effects of cannabinoids in the developing brain.ref. bib14 Our study of in vivo experimental conditions for increasing frequency and power of chromatin herniation may be instrumental for further investigation of the mechanisms of membrane rupture and repair, as well as in prospective medicinal applications of the obtained knowledge.
Results
Piercing nuclear hernia is an ultrastructural pathology of migrating neurons
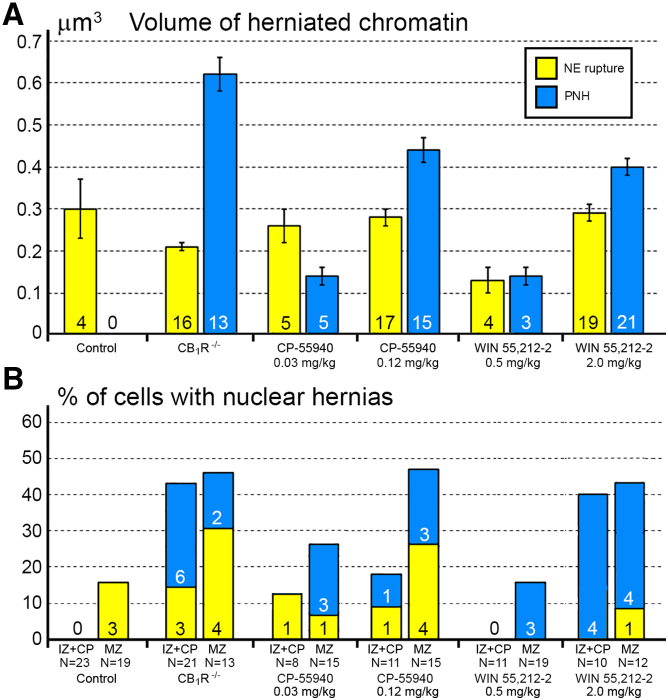
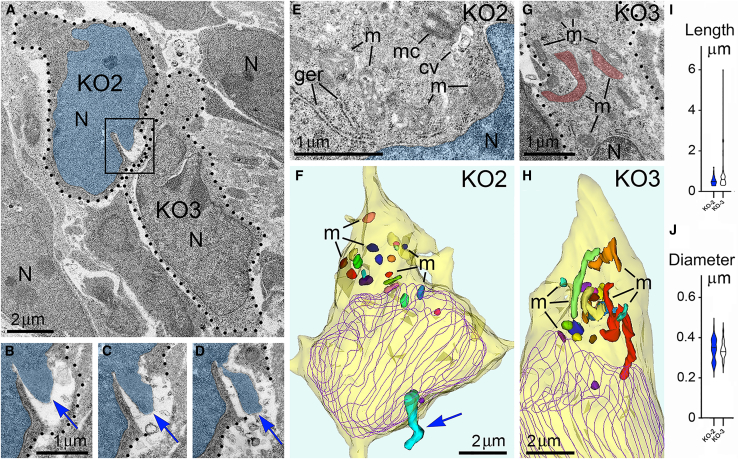
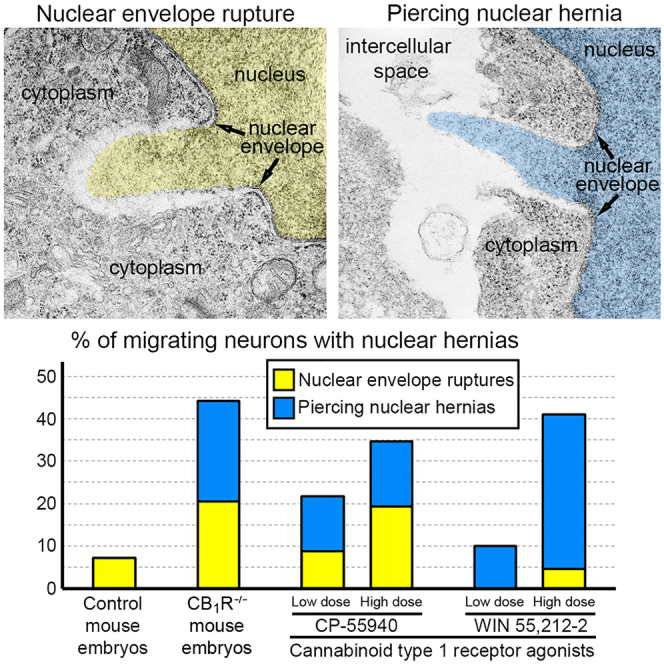
Here, we report ultrastructural pathology in mouse embryo cerebral neurons related to CB1R knock-out and in utero application of synthetic cannabinoids. Particularly, we documented numerous local breakups of the NE accompanied by herniation of nuclear chromatin into the cytoplasm (referred as NE rupture hereafter; Figures 1A–1D ). Moreover, in a fraction of the cells, streams of herniated chromatin ruptured not only the NE, but also the plasma membrane, exposing cytoplasm and nucleoplasm to the intercellular space (Figures 1E–1H). To our knowledge, this is the first published description of simultaneous rupture of the NE and the plasma membrane, and we suggest naming this cellular pathology PNH. For detailed characterization of this phenomenon, we performed extensive analyses of electron microscopy imagery of migrating neurons in CB1R−/− embryos, wildtype control mouse embryos and embryos after in utero application of CB1R agonists CP-55940 and WIN 55,212-2 at different doses. Using three-dimensional (3D) reconstruction, we identified 122 nuclear hernias (including 65 NE ruptures and 57 PNHs). Morphometric analyses showed that volume of the streams of herniated chromatin varied from 0.02 to 1.63 μm3 (Table S1). Average volumes of herniated chromatin differed between the experimental groups showing a maximum (0.62 ± 0.04 μm3) for neurons with PNH from CB1R−/− embryos (Figure 2A). In some analyzed cells, the PNH stream was narrow and reached the length of several microns (Figure S1). Although the molecular mechanism of PNH remains unclear, the large volume and length of the herniated chromatin streams indicate that at least one detail involved in the event is the presence of high intranuclear pressure during nuclear translocation in a tightly packed environment, such as a developing cerebral cortex.ref. bib72
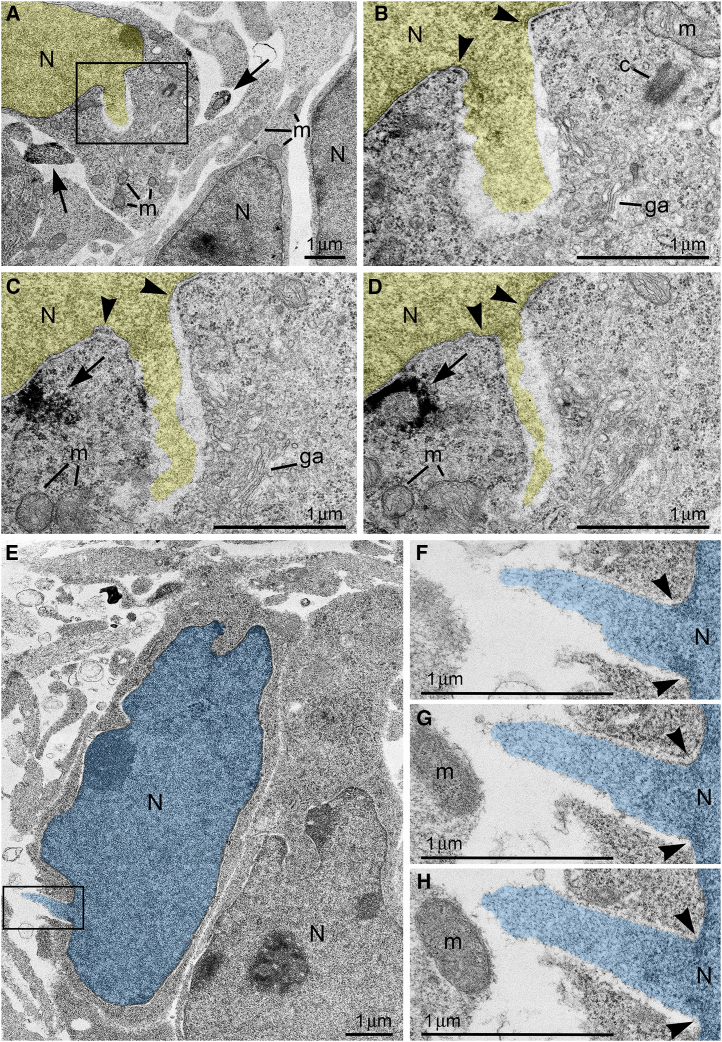
The vast majority of analyzed cells demonstrated an elongated shape, indicating active nuclear translocation in the moment of fixation. Another important piece of evidence for active migration is the intracellular position of the centrosome —the main center of cytoskeleton reformation— which periodically relocates in the direction of migration.ref. bib21,ref. bib83,ref. bib84 By contrast, the anchoring of the mother centriole to plasma membrane and elongation of the cilia outside of the cell correlates with termination of migration.ref. bib85 Accordingly, the majority of analyzed cells [total of 124 cells; of them, 37 cells from control group (Table S2), 23 cells from CB1R−/− mice (Table S3), 32 cells from CP-55940 exposed embryos (Table S4); 32 cells from WIN 55,212-2 exposed embryos (Table S5)] contained free centrosomes in cytoplasm or showed initial steps of cilium development, such as mother centrioles connected to the cilial vesicles in cytoplasm or anchored to the plasma membrane on occasion producing short procilium. Moderately developed cilia (length of the axoneme ∼1 μm) were identified only in 4 cells from CP-55940 group (Table S4) and 6 cells from WIN 55,212-2 group (Table S5). Therefore, most if not all analyzed cells were in process of active migration in the moment of fixation, and their nuclei, likely exposed to high pressure, were predisposed to NE rupture.
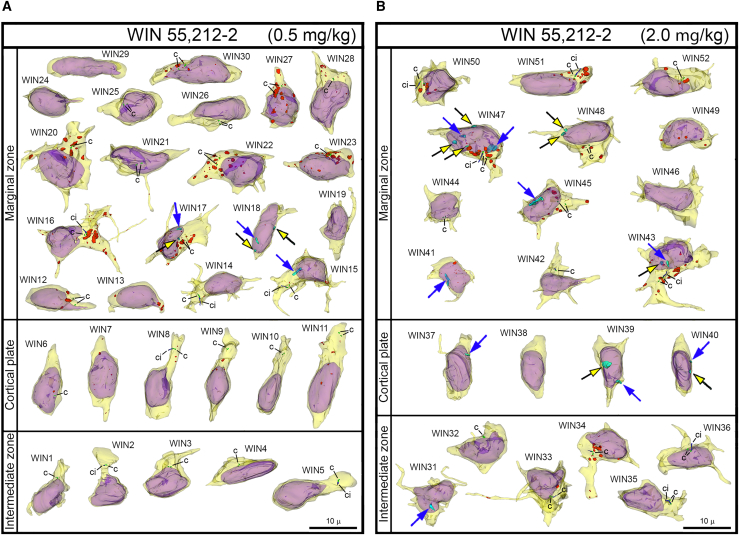
NE ruptures and PNHs are frequent in CB1R−/− embryos and wildtype embryos exposed to CB1R agonists
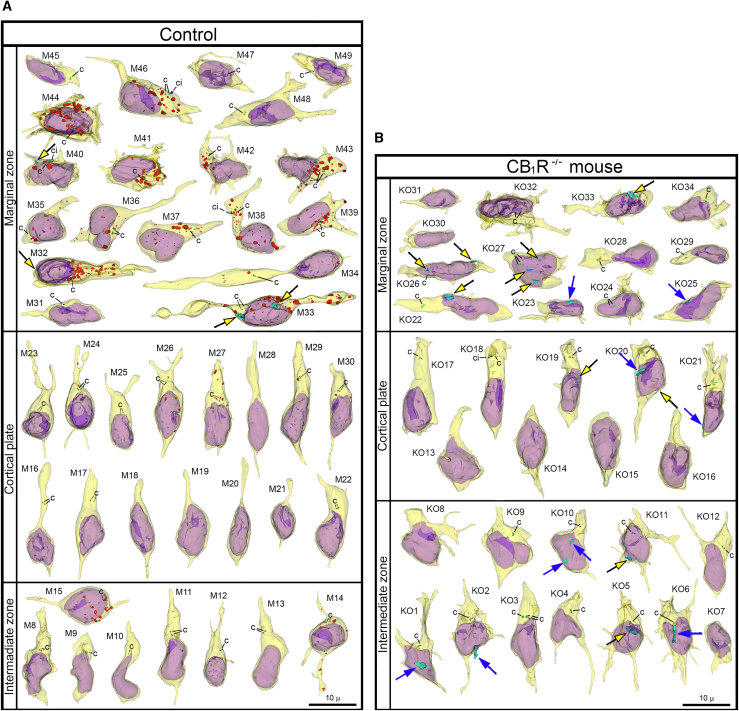
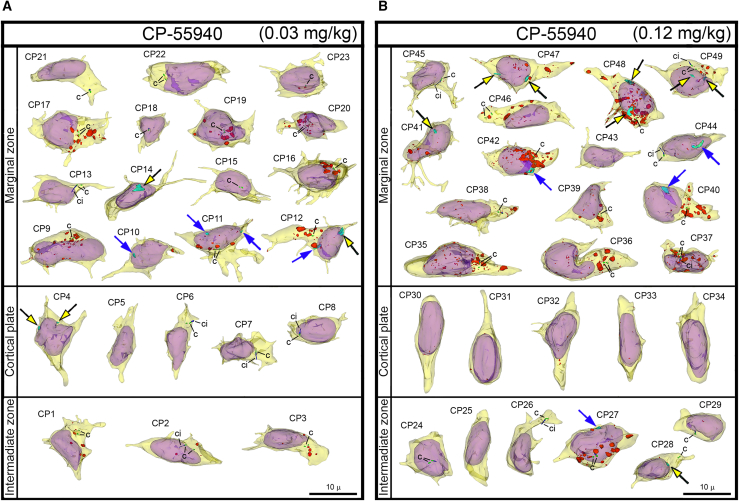
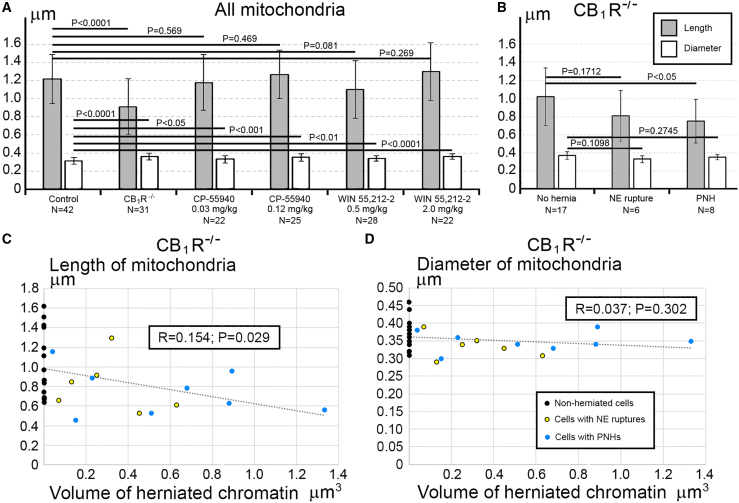
For objective estimation of the frequency of chromatin herniation, we performed complete 3D reconstruction of randomly selected nuclei and cell bodies of migrating neurons from CB1R−/− mouse embryos, wildtype control embryos and embryos exposed to CB1R agonists CP-55940 and WIN 55,212-2 at different doses (Figure 2B). Out of a total of 42 reconstructed cells from control embryos, only 3 cells showed NE ruptures, and none had PNH (Figure 3A; Table S2). In contrast, among 34 analyzed cells from CB1R−/− embryos, 15 cells were herniated (of which 7 showed one or several NE ruptures and 8 cells showed at least one PNH; Figure 3B; Table S3). Application of two different CB1R agonists produced similar chromatin herniation, the extent of which was dose dependent. After injection of CP-55940 at low dose (0.03 mg/kg body weight), we detected 5 herniated cells (2 cells with NE ruptures and 3 cells with PNHs) out of a total of 23 analyzed cells, whereas the high dose (0.12 mg/kg) produced 9 herniated cells (5 cells with NE ruptures and 4 cells with PNHs) out of a total of 26 analyzed cells (Figure 4; Table S4). In the embryos exposed to low dose (0.5 mg/kg) of WIN 55,212-2, only 3 cells out of 30 analyzed cells showed hernias (all 3 cells showed at least one PNH), whereas the high dose (2.0 mg/kg) produced 1 cell with NE ruptures and 8 cells with PNHs out of a total of 22 analyzed cells (Figure 5; Table S5). Elevated frequencies (more than 40%) of herniated cells (NE ruptures and PNHs combined) were observed in the marginal zone of CB1R−/− embryos and wildtype embryos exposed to high doses of both CB1R agonists. High frequencies of herniated cells were also detected in the intermediate zone and the cortical plate of CB1R−/− embryos and wildtype embryos exposed to the high dose of WIN 55,212-2 (Figure 2B). Thus, we observed increased occurrence of NE ruptures and PNHs in all analyzed populations of migrating neurons – in both, the projection neurons migrating vertically through the developing cerebrum and in the horizontally migrating interneurons (Figure 2B). It is notable that profound effects to the frequency of herniated cells were produced by the opposite actions – knock-out of CB1R and its stimulation by agonists. This indicates that deviations from optimal functionality of the endocannabinoid system into either direction may affect the welfare of the nucleus and provoke chromatin herniation.
Relationship of chromatin herniation and expression of CB1R in wildtype embryos
In control, CP-55940 and WIN 55,212-2 groups combined, a total of 29 herniated cells (NE ruptures and PNHs combined) were identified. Of those, 22 were located in the marginal zone (36% out of the 61 analyzed marginal zone cells), and only 7 were located in the intermediate zone or cortical plate (11% out of the 63 analyzed intermediate zone and cortical plate cells). This corresponds with a concentration of developing CB1R-expressing interneurons in the marginal zone and lower accumulation of CB1R in the cell bodies of migrating projection neurons.ref. bib21,ref. bib78 To further investigate if emergence of the chromatin hernia may be related to the level of CB1R accumulation, we estimated the amount of anti-CB1R immunolabeling end-product depositions in the herniated and non-herniated cells from all groups of wildtype embryos. Among 22 herniated cells from the marginal zone, 16 demonstrated visually high or moderate CB1R levels while 6 cells showed little to no CB1R immunolabeling in the reconstructed cell segments. Accordingly, out of 7 herniated cells from the intermediate zone and cortical plate, only 1 contained high levels of CB1R (cell CP27, which showed horizontal phenotype and may be an interneuron tangentially migrating through the intermediate zone), while 6 cells showed little to no CB1R immunolabeling (Table S6). While the applied method of estimation of CB1R content had several limitations (e.g., CB1R immunolabeling may be underestimated because of incomplete reconstruction of the cells), our observations indicate some degree of relationship between chromatin herniation and CB1R accumulation in the cell bodies. At the same time, this relationship may have probabilistic character as many CB1R-expressing neurons do not display hernias (Figures 4 and 5). Thus, our study revealed more frequent chromatin herniation in the CB1R-expressing interneurons rather than in the projection neurons in wildtype mouse embryos. Our immunohistochemical analyses show the probability (rather than unequivocal proof) of a link between chromatin herniation and the amount of CB1R in the cell bodies.
Chromatin herniation correlates with mitochondrial fission in CB1R−/− embryos
Morphology of mitochondria may be useful for quantitative characterization of cellular functionality. For example, prevalence of mitochondrial fission – as evidenced by their shorter length – reflects cellular disorder.ref. bib86,ref. bib87,ref. bib88,ref. bib89 Here, we observed that cells from the same sample may demonstrate dramatic ultrastructural differences – short mitochondria were found in the cells with PNH while adjacent non-herniated cells show normally sized mitochondria (Figure 6). These observations inspired our extensive morphometric analysis of the 3D reconstructed mitochondria, which identified morphologic heterogeneity of mitochondrial length and diameter in migrating neurons from all studied experimental groups (Tables S2, S3, S4, and S5; Figures S2, S3, and S4). Namely, in Control, CP-55940 and WIN 55212-2 groups, we detected similar fluctuations in mitochondrial length from ∼0.3 to ∼8 μm, and fluctuations of diameter from ∼0.2 to ∼0.6 μm, whereas in CB1R−/− group, the length fluctuated between ∼0.3 and ∼6 μm, and diameter fluctuated from ∼0.1 to ∼0.8 μm. Analysis of average mitochondrial length showed a statistically significant decrease of mitochondrial length only in the CB1R−/− group in comparison with the control group—this suggested up-regulation of mitochondrial fission in CB1R−/− embryos (Figure 7A). An unpaired T-test of variances of average mitochondrial diameters showed a minor statistically significant increase in CB1R−/−, CP-55940 and WIN 55212-2 groups in comparison with the control group—this may indicate a slight swelling of a fraction of mitochondria after disruption of the endocannabinoid system (Figure 7A). However, while we saw minimal differences between the averages, certain cells showed a greater range in diameter measurements (Figures S2, S3, and S4). Accordingly, F-tests to compare variances in diameter and length showed no significant differences between these groups. We also analyzed mitochondrial morphometric characteristics in the experimental groups, separating them into three subgroups based on absence of the hernia (No hernia), or presence of a certain type of hernia (NE rupture or PNH). Average mitochondrial length showed statistically significant reduction in T-test between “No hernia” and PNH subgroups in CB1R−/− embryos — this indicated an association of PNH with mitochondrial fission (Figure 7B). Moreover, the mitochondrial length (but not diameter) negatively correlated with the volume of herniated chromatin in the cells with NE ruptures and PNHs in CB1R−/− embryos (Figures 7C and 7D). In CP-55940 and WIN 55,212-2 exposed embryos, variations of mitochondrial morphology between “No hernia”, NE rupture and PNH subgroups, as well as correlations of mitochondrial length and diameter with the volume of herniated chromatin, were not statistically significant (data not shown). Thus, at least in the CB1R−/− embryos, emergence of PNHs correlates with mitochondrial fission — an ultrastructural feature associated with moderate cell disorder.ref. bib86,ref. bib87,ref. bib88,ref. bib89 However, the massive mitochondrial swelling that was documented in previous studies to be irreversible and foreshadowing cell death through a necrotic mechanismref. bib86,ref. bib88 was not identified in this study.
Probable recovery of the cells with PNH
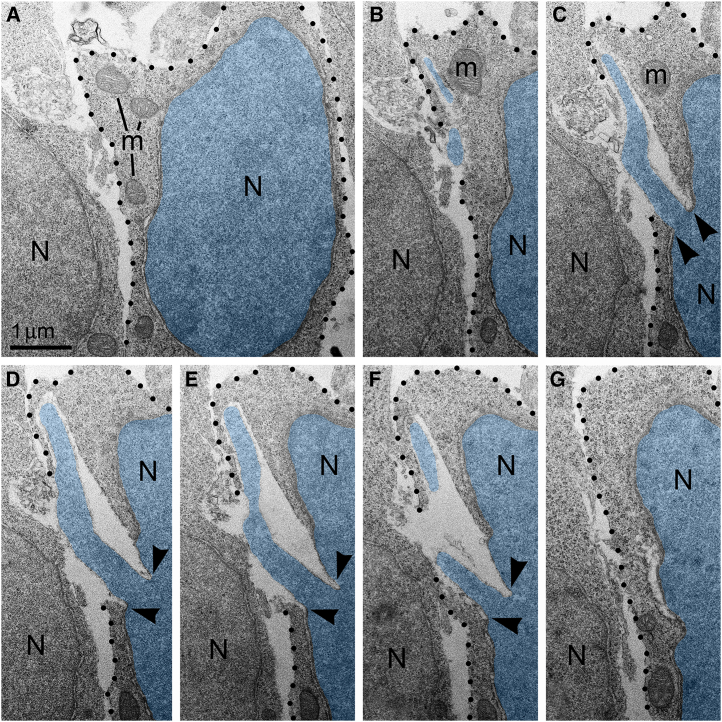
Isolation of the cellular inner content by an intact plasma membrane is among the fundamental criteria of a living cell. High frequency of cells with ruptured plasma membrane in certain experimental conditions (Figure 2) suggests the death of many cells. Surprisingly, we did not observe cellular remnants or cells with ultrastructural features of necrotic, apoptotic or autophagy type degradation in the analyzed embryos. Instead, a fraction of the cells showed reduction of mitochondrial length — evidence of mitochondrial fission and cell disorder (Figures S2, S3, and S4). Different time courses of NE rupture (likely occurring in the span of secondsref. bib57,ref. bib60,ref. bib61,ref. bib62,ref. bib63) and mitochondrial reaction (delay in the span of hoursref. bib88) may partially explain the divergence between emergence of PNH and reaction of organelles in the embryos temporarily exposed to CB1R agonists. Nevertheless, the absence of a considerable number of dead cells in CB1R−/− embryos, which constantly experience disorder of endocannabinoid signaling, raises a supposition that PNH may be repaired rather than always be fatal for the cell. This observation aligns with the fact that CB1R−/− mice generally do not exhibit obvious behavioral phenotypes.ref. bib90 In accordance, partial recovery of cells after NE rupture is known.ref. bib64,ref. bib76 Surviving neurons were reported after transection of the axons, but not after damage of the cell bodies.ref. bib91 Here, we considered morphological features that can provide possible recovery of migrating neurons with PNH. Namely, for a fraction of the cells with PNH, the area of the plasma membrane rupture was closely surrounded by adjacent cell bodies or processes (Figure 8), which may contribute to decreasing the flow of cytoplasm and effectively blocking the leak. We observed blocking of the plasma membrane rupture by adjacent cells at 25 PNHs among a total of 57 examples analyzed. Of them, 4 blocked PNHs out of 13 analyzed (30.8%) were identified in CB1R−/− embryos, 13 blocked out of 20 analyzed (65.0%) — in CP-55940 exposed embryos and 8 blocked out of 24 analyzed (33.3%) — in WIN 55,212-2 exposed embryos (Table S1). Blocking of the plasma membrane rupture by adjacent cells, together with a possible delay of mitochondrial reaction, may explain our observations of normally sized mitochondria in several 3D reconstructed cells with PNH. At the same time, plasma membrane blocking may be ineffective for other cells that showed considerable reduction of mitochondrial length (Figures S2, S3, and S4). Thus, although death of cells with ruptured NE and plasma membrane is probable, PNH in mouse embryo brains may be non-fatal for a fraction of the affected cells. This suggests the possibility of recovery for catastrophically damaged neuronal cell bodies, which has not been demonstrated previously.
Discussion
Here, we report generation of powerful chromatin streams breaking NE together with the plasma membrane, which we suggest naming PNH. About 40% of randomly analyzed cells among migrating projection neurons and interneurons demonstrated NE ruptures or PNHs in CB1R−/− embryos and in wildtype embryos shortly exposed to high doses of synthetic agonists of CB1R – 0.12 mg/kg of CP-55940 or 2.0 mg/kg of WIN 55,212-2. This indicates that deviation from optimal functioning of the endocannabinoid system in either direction —knock-out of CB1R or its temporary overstimulation— increases likelihood of altered cellular nucleus and provoke chromatin herniation in a similar way. Among the embryos exposed to aforementioned experimental conditions, CB1R−/− embryos showed maximal ultrastructural pathology in terms of high frequency of herniated cells, high volume of chromatin streams, reduced average length of mitochondria in all the cells, more pronounced reduction of average length of mitochondria in the cells with PNHs, and negative correlation of the mitochondrial length with the volume of herniated chromatin in the cells with NE ruptures and PNHs. Our finding of experimental conditions increasing the frequency and power of chromatin herniation in migrating cells may be instrumental for further investigation of mechanisms of NE rupture and repair as well as for study of the role of the endocannabinoid system in cytoskeleton functionality.
As described here, PNHs catastrophically break isolation of the inner content of the cell from the environment. Cells exposed to extreme physical, chemical, or mechanical stimuli may immediately lose their structural integrity and die in an uncontrollable manner termed ‘accidental cell death’, which is opposite to mechanisms of programmed cell death such as apoptosis, autophagy, or necrosis-type cell death.ref. bib91,ref. bib92,ref. bib93,ref. bib94 Cases of accidental cell death are virtually undetectable using biochemical or immunochemical methods, for example, TUNEL staining, caspase immunolabeling, or labeling of autophagy markers. Electron microscopy visualizes organelles regardless of their functional conditions and can detect dead cells or cellular remnants. Surprisingly, we did not observe considerable numbers of dead cells in the samples containing numerous cells with herniated nuclei. This suggests an opportunity for repair of NE and plasma membranes in at least a fraction of the affected cells, providing their survival of the initial breach. Indeed, NE ruptures may be repaired without loss of cell viability.ref. bib57,ref. bib60,ref. bib61,ref. bib63 Repair of large ruptures of the plasma membrane was shown in muscle cells, oocytes, epithelial cells, invasive cancer cells and other cell types. Neurons may effectively restore transected axons, but recovery of mechanically damaged cell bodies was not documented.ref. bib95,ref. bib96 The initial repair steps include plugging the membrane rupture with conglomerates of vesicles or membrane whorls that restrict the leak of cytoplasm providing time for reconstruction of intact plasma membrane.ref. bib95,ref. bib96,ref. bib97,ref. bib98 The major trigger for the signaling cascade that precedes membrane sealing is the influx of Ca2+ through the membrane disruption. Subsequent vesicle deposition and fusion involve complex Ca2+-dependent pathways acting through the cAMP signaling cascade, cytoskeleton reconstruction, SNAP receptors, the phospholipase enzymes, calpain proteases and others. The membrane patch normally serves as a temporary barrier and is subsequently remodeled or removed via exocytic or endocytic machinery.ref. bib95,ref. bib97,ref. bib98,ref. bib99 We observed analogous cases when adjacent cells surround the plasma membrane rupture, apparently delaying degeneration of the damaged cell, as identified by a reduction in morphological disorder of mitochondria. Lack of dead cells in samples containing numerous neurons with catastrophically ruptured plasma membranes is the first evidence for survival of severely damaged cell bodies of mammalian neurons. Deeper study of this phenomenon may become instrumental for increasing regenerative capacity of neuronal cell bodies after traumatic brain injury, ischemic conditions and during neurodegenerative diseases.ref. bib91,ref. bib96,ref. bib97,ref. bib98,ref. bib99,ref. bib100
The early expression of CB1R indicates its probable involvement in prenatal and postnatal brain development.ref. bib20,ref. bib78 Numerous studies indicate that prenatal inhibition or overstimulation of the endocannabinoid system may provoke long-term consequences in the offspring, for example, altered breathing, disturbed suckling, and memory deficit linked to disorder of glutamate-ergic neurons,ref. bib15,ref. bib45,ref. bib101,ref. bib102,ref. bib103,ref. bib104 but the underlying mechanisms remain enigmatic in many aspects. Our data indicate that disorder of the endocannabinoid system may provoke NE ruptures and PNHs in migrating neurons, a fraction of which also acquire moderate ultrastructural pathology. Although we did not observe completely destroyed cells, the present study does not assume a complete recovery of all cells experiencing NE ruptures or PNHs. More likely, temporary damage of NE and plasma membrane provokes a delay or disorientation of cell migration. This may be the reason for previously identified brain dysfunction in CB1R−/− mice or cognitive deviations in the animals and humans exposed to cannabis during development that, nevertheless, do not exhibit obvious behavioral phenotypes or evidence of teratogenic effects of the cannabinoids.ref. bib14,ref. bib15,ref. bib45,ref. bib90,ref. bib101,ref. bib102,ref. bib103,ref. bib104 Our finding opens an opportunity for further investigation if temporary disorder of the endocannabinoid system, for example, in cases of recreational cannabis use by pregnant women, may provoke accidental death or malfunction of migrating neurons.
Judging from the large volume and length of the herniated chromatin streams, the NE ruptures and PNHs observed here may be consequences of increased intranuclear pressure, which occurs during nuclear translocation through tightly packed tissue such as the developing mammalian cerebrum. The generally accepted model of NE rupture and chromatin herniation presumes participation of cytoskeleton and nucleoskeleton in the increasing of intranuclear pressure.ref. bib59,ref. bib72 Chronic treatment of adult rats with WIN 55,212-2 increased expression of neurofilaments Nf-160 and Nf-200, and microtubule-associated protein-2 (MAP-2). Meanwhile, CB1R−/− mice demonstrated lower expression of the neurofilaments and MAP-2, along with reduced dendritic arborization in the hippocampus.ref. bib105,ref. bib106,ref. bib107 Such deviated expressions of the cytoskeletal components after stimulation or knock-out of CB1R constitute evidence of its participation in neuronal cytoskeleton consolidation. Link of endocannabinoid system and particularly CB1R with the function of actin filaments was also demonstrated in the context of axonal pathfinding. This mechanism involves CB1R internalization from filopodia and chemorepulsion of the axonal growth cones by activating RhoA GTPases (small guanosine triphosphatases).ref. bib24,ref. bib25 RhoA GTPases are known for their role as molecular switches transducing extracellular stimuli to the actin cytoskeleton.ref. bib108,ref. bib109 Thus, the unknown role of CB1R in the developing brain may include maintaining optimal function of the cytoskeleton, which is crucial for translocation of neuronal cell bodies.
Our findings may also have applications for cancer research. Although NE rupture resulting in genomic instability might promote cancer progression, unstable NE also represents a particular weakness of metastatic cancer cells. Accordingly, NE ruptures detected in many cancer cell lines during cell migration through tightly constricted areas often result in cell death.ref. bib57,ref. bib58,ref. bib60,ref. bib66 Conditions that have potential to increase the probability of NE rupture and death of metastatic cells are considered potent for cancer treatment.ref. bib47,ref. bib57 We suggest investigating the conditions of PNH generation for targeting metastatic cancer cells. Such conditions may include manipulations of the endocannabinoid system and probably other molecular mechanisms, to increase the frequency and power of NE ruptures. Numerous studies have shown capability of cannabinoids as anticancer agents in several disease models in vitro and in vivo. The observed effects of the cannabinoids’ application include inhibiting tumor growth and metastasis, reducing cell viability through promoting apoptosis, and inhibiting angiogenesis; but the molecular mechanisms at play were not identified in detail. Accordingly, no strong clinical trial data exists to confirm the pre-clinical evidence for cannabinoids’ anticancer effects.ref. bib38,ref. bib39,ref. bib40,ref. bib41,ref. bib43,ref. bib110,ref. bib111,ref. bib112 Our findings suggest a mechanism of cannabinoid action on the nuclei of migrating cells and may be instrumental for inducing breaks of the plasma membrane in metastatic tumor cells. This opens an opportunity for a new research direction that may result in development of therapeutic applications of cannabinoids for targeting cancer cells.
Limitations of the study
In this article, we applied a method of 3D reconstruction of cells from a long series of ultrathin sections, which provides an opportunity for high resolution in vivo morphologic analysis of cells at certain developmental stages. Nevertheless, the application of electron microscopy as the only research method is among the limitations of this article. Confirmations using different methods of analysis are required. For example, it makes sense to develop equipment and procedures for in vitro study of cell migration through restricted space, suitable for detecting herniated nuclear material with light microscopy or other express methods. Another source of uncertainty in our findings is the fact that the analysis of CB1R was performed with only one immunolabeling method. Different CB1R isoforms, heterodimers, or other receptors, such as cannabinoid type 2 receptor, G protein-coupled receptor 55 and transient receptor potential vanilloid-1 (if present in the developing cerebrum, which awaits conformationref. bib14) may respond to synthetic cannabinoids. Moreover, genetic knockout models such as CB1R−/− mouse preserve the opportunity for developmental compensation that may partially challenge the conclusion. Analysis of conditional knock-out mice can mitigate this problem.
Resource availability
Lead contact
Requests for further information, resources, or reagents should be made to Dr. Yury M. Morozov (yury.morozov@yale.edu).
Materials availability
No novel reagents were generated by this study. CB1R−/− (Cnr1−/−) mice were kindly provided by Dr. Ken Mackie, Indiana University.
Data and code availability
- This study did not generate/analyze any datasets/code.
- Microscopy data reported in this paper will be shared by the lead contact upon request.
- Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request.
Acknowledgments
We are grateful to Ekaterina Morozova and Mikhail Morozov for editorial work on the manuscript. This research was supported by the Kavli Institute for Neuroscience at Yale, and USA NIH NIDA grant DA023999 to P.R.
Author contributions
Y.M.M., conceptualization, data curation, formal analysis, investigation, methodology, validation, visualization, and writing; P.R., funding acquisition, supervision, and validation.
Declaration of interests
The authors declare no competing interests.
STAR★Methods
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Guinea pig CB1R | Frontier Science Co. Ltd, Japan | Cat# CB1-GP-Af530 |
| Chemicals | ||
| WIN 55,212-2 | Biomol International LP, Plymouth, PA, USA | Cat# CR-105 |
| CP-55940 | Sigma-Aldrich | Cat# C1112 |
| 3,3′-Diaminobenzidine | Thomas Scientific | Cat# C963P95 |
| Ultrostain-2 | Leica Biosystems | Cat# 16707235 |
| Experimental models: Organisms/strains | ||
| Mus musculus:C57BL/6 | The Charles River Laboratory | N/A |
| Mus musculus:CD1-Cnr1−/− | Laboratory of Dr. K. Mackie | Ledent et al.ref. bib113MGI: 1857736 |
| Oligonucleotides | ||
| 5′-CATCATCACAGATTTCTATGTAC-3′ | IDTDNA | Parmentier-Batteur et al.ref. bib114 |
| 5′-GAGGTGCCAGGAGGGAACC-3′ | IDTDNA | Parmentier-Batteur et al.ref. bib114 |
| 5′-AAGGAAGGGTGAGAACAGAG-3′ | IDTDNA | Parmentier-Batteur et al.ref. bib114 |
| 5′-GATCCAGAACATCAGGTAGG-3′ | IDTDNA | Parmentier-Batteur et al.ref. bib114 |
| Software and algorithms | ||
| Reconstruct 1.1.0.0. | http://www.bu.edu/neural/Reconstruct.html | Fialaref. bib115; Fiala and Harrsref. bib116 |
| GraphPad Prism 10.1.2 | GraphPad Software, LLC | GraphPad.comRRID:SCR_002798 |
Experimental model and subject details
Animals and genotyping
All animal protocols were compliant with the National Institutes of Health (USA) guidelines for animal care and use and were approved by the Institutional Animal Care and Use Committee of Yale University (Protocol #2018–10750.A3 approved 25 June 2020). Control wild-type mice and mice for prenatal stimulation of CB1R (see below) were obtained from a colony consisting of a stable strain of C57BL/6 mice. CB1R+/+ and CB1R−/− mice were generated by breeding CB1R+/− pairs in CD1 background as described previously.ref. bib113 Breeding pairs were housed together on a 12-12-h light-dark cycle with food and water available ad libitum. Primers used for CB1R PCRs were 5′-CATCATCACAGATTTCTATGTAC-3′ and 5′-GAGGTGCCAGGAGGGAACC-3′, to amplify a 366 bp band from the wild-type allele and 5′-GATCCAGAACATCAGGTAGG-3′ and 5′-AAGGAAGGGTGAGAACAGAG-3′, for a 521 bp band from the mutant CB1R allele.
Method details
Prenatal stimulation of CB1R
For prenatal stimulation of CB1R, we performed two intraperitoneal (IP) injections of wild type pregnant mice with synthetic agonists, WIN 55,212-2, or CP-55940 at low and high doses. First injection was done 24 h before sacrifice, and the second – 1 h before sacrifice the dams, followed by the embryo brains collection. The doses of WIN 55,212-2 – 0.5 mg per kg of body weight (mg/kg) and 2.0 mg/kg – were chosen on the basis of previous publications that showed the low dose to be non-toxic for the embryos and the high dose to have significant affection of reproduction parameters in cases of prolonged prenatal exposure.ref. bib101,ref. bib102 The doses of CP-55940 – 0.03 mg/kg and 0.12 mg/kg – were chosen on the basis of known high range of its effective doses in developing and adult brain.ref. bib12,ref. bib117,ref. bib118,ref. bib119 The drugs were solved in the vehicle containing 5% DMSO, 5% Tween 80 and saline and injected in the volume of 10 mL/kg body weight. Volume-equivalent vehicle was injected in the control pregnant mice. For the morphologic analysis, we used CP-55940 exposed embryos at the low dose at the embryonic day (E) 13 (N = 4); CP-55940 exposed embryos at the high dose at E12 (N = 4); WIN 55,212-2 exposed embryos at the low dose at E12 (N = 1), E13 (N = 3) and E16 (N = 1) and WIN 55,212-2 exposed embryos at the high dose at E13 (N = 4). The control group included embryos from the vehicle-injected pregnant mice at E12 (N = 1) and E16 (N = 3), as well as untreated embryos at E13 (N = 1) and E14 (N = 1). We did not observe systematic differences in the analyzed parameters between the vehicle-exposed and untreated embryos as well as between the embryos at different ages from same experimental group. Totally, 6 control embryos, 4 low dose CP-55940; 4 high dose CP-55940, 5 low dose WIN 55,212-2 and 4 high dose WIN 55,212-2 exposed embryos, as well as 4 CB1R−/− embryos at E14 in CD1 background were used for below morphologic studies.
Electron microscopy and 3D reconstruction
Pregnant mice were anaesthetized with IP injection of sodium pentobarbital (3 mL/kg) or euthasol (0.5 mL/kg) and sacrificed with cervical dislocation. The embryos were removed from the uterus and their age was adjusted based on the anatomical landmarks. Embryos without apparent abnormalities were decapitated, brains were removed and immersed in the fixative containing 4% paraformaldehyde and 0.2% glutaraldehyde in 0.1M phosphate buffer (pH 7.4) for 3–4 days at 4°C. Coronal 100-mm-thick brain slices were cut with a vibratome and prepared for immunohistochemistry and electron microscopy as described below. After incubation with the CB1R antibodies (Frontier Science Co. Ltd; dilution 1:2000), the slices were extensively washed and immersed in a solution of biotinylated anti-guinea pig IgGs (Jackson Immunoresearch; Cat#706-065-148; 1:300) and the Elite ABC kit (Vector Laboratories, Burlingame, CA, USA; Cat# PK-6100; 1:300). Ni-intensified 3,3′-diaminobenzidine–4HCl (DAB-Ni) was used as a chromogen. Brain slices were post-fixed with 1% OsO4, dehydrated and embedded in Durcupan (EMS, Hatfield, PA, USA; Cat# 14610). Electron microscopy and 3D reconstruction from ultrathin sections were performed as described previouslyref. bib21,ref. bib88 with the following minor modifications. Selected segments from the lateral neocortex were re-embedded into Durcupan blocks and cut by a Reichert ultramicrotome into 60-nm-thick sections. Series of 150–200 consecutive sections from occasionally taken segments of the intermediate zone, cortical plate, or marginal zone were collected on one-slot grids covered with Formvar or Butvar B-98 films (both from EMS, Hatfield, PA, USA), stained with Ultrostain-2 (Leica Biosystems, Danvers, MA, USA) and evaluated in JEM 1010 electron microscope (JEOL, Japan) equipped with Multiscan 792 digital camera (Gatan, Pleasanton, CA, USA) or Talos L120C electron microscope equipped with Ceta digital camera (ThermoFisher Scientific, Boston, MA). Serial images of randomly selected interphase cells were made with 12,000× or 15,000× magnification of JEM1010 or with 3,400× or 4,300× of Talos L120C electron microscope. 3D reconstructions of cell bodies, nuclei, centrioles, cilia and mitochondria and measurements of the volumes of the reconstructed objects were performed using Reconstruct 1.1.0.0. software (Boston, MA)ref. bib115,ref. bib116 publicly available at http://www.bu.edu/neural/Reconstruct.html.
Identification of chromatin herniations
Ruptures of the nuclear envelope (NE) and herniations of chromatin streams were identified in serial sections. The hernias that contained chromatin streams in cytoplasm while the plasma membrane stayed intact were identified as NE ruptures; the hernias pairing with local damage of the plasma membrane and extending the chromatin streams in the intercellular space were identified as piercing nuclear hernias (PNH). Cells showing both, NE rupture and PNH, were counted as belonging to the PNH group. PNHs with the plasma membrane rupture closely surrounded by adjacent cells in all serial sections were counted as blocked PNHs. Percentages of the cells with NE rupture and PNH were calculated among the cells randomly selected for serial photography and 3D reconstruction. The quantifications avoided subjectivity as the nuclear hernias were mostly local and identifiable in the analyzed cells during their 3D reconstruction, which was performed after serial photography of the cells was completed. All the serially photographed cells were included in the estimations of the frequencies of herniated cells. The hernias identified during the electron microscopy session were serially photographed for measurements of their volume and were excluded from the estimations of the frequencies of herniated cells. For volume measurement, herniated chromatin streams were traced in the serial sections along the periphery of the stream in cytoplasm and, in case of PNH, in the intercellular space, and along straight lines connecting points of interruption of the nuclear membranes. Values of volume measurements of the herniated chromatin streams for each experimental group were calculated as mean ± SEM.
Estimation of intracellular CB1R
Anti-CB1R DAB-Ni depositions in each cell were traced for 3D reconstruction and counted as “single” depositions if local staining were identified in at least 3 serial sections; merging conglomerates of the immunoreaction end-product around intracellular vesicles were counted as “globule” depositions.ref. bib21 The degree of CB1R accumulation in the 3D reconstructed cell segments was estimated as low, middle or high.
Morphometry of mitochondria
Morphometric characterization of 3D reconstructed mitochondria was performed as we validated in our previous articles.ref. bib86,ref. bib87,ref. bib88,ref. bib120 Namely, the length of mitochondria was estimated in the 3D images. The root-mean-square diameter of the reconstructed mitochondria was calculated based on the volume formula for ellipsoids using their length as the longest diameter of the ellipsoid. To optimize the time-consuming 3D reconstruction and morphometrical analysis of mitochondria, we performed the following pilot test. We reconstructed and measured all available mitochondria (more than 100 in total) in two cells. Then in both cells, we randomly assigned all cellular mitochondria into subgroups containing 20 mitochondria and performed statistical comparison of the group with the subgroups. Unpaired T-test showed absence of statistically significant difference between the groups of all mitochondria and the subgroups containing 20 mitochondria from both analyzed cells. So, this pilot test showed that 20 randomly chosen mitochondria adequately characterize the mitochondrial system of a cell. In this article, we restricted the quantity of analyzed mitochondria to 40 per cell. For the cells containing less than 40 mitochondria in the serial sections, we reconstructed and analyzed all available mitochondria. Cells containing less than 10 completely reconstructed mitochondria were excluded from the quantitative analysis of mitochondria.
Quantification and statistical analysis
Statistical analyses of mitochondrial morphometric parameters and generation of violin estimation plots were performed using GraphPad Prism 10.1.2 software. Values of mitochondrial length and diameter were expressed as mean ± SD for every analyzed cell. Values of mitochondrial length and diameter for each experimental group were calculated as mean ± SD from average length and diameter of each cell. Unpaired T-test and F-test statistical analyses of variances were applied for comparison of mitochondrial lengths and diameters between the experimental groups. The relationships between the quantified mitochondrial parameters and the volume of herniated chromatin were assessed by Pearson’s correlation. All p values were 2-tailed; the value of p < 0.05 was used as the threshold for significance.
References
- M. Salzet, G.B. Stefano. The endocannabinoid system in invertebrates. Prostaglandins Leukot. Essent. Fatty Acids, 2002. [DOI | PubMed]
- S. Fasano, R. Meccariello, G. Cobellis, R. Chianese, G. Cacciola, T. Chioccarelli, R. Pierantoni. The endocannabinoid system: an ancient signaling involved in the control of male fertility. Ann. N. Y. Acad. Sci., 2009. [DOI | PubMed]
- M.R. Elphick. The evolution and comparative neurobiology of endocannabinoid signalling. Philos. Trans. R. Soc. Lond. B Biol. Sci., 2012. [DOI | PubMed]
- R.L. Bailone, H.C.S. Fukushima, L.K. de Aguiar, R.C. Borra. The endocannabinoid system in zebrafish and its potential to study the effects of Cannabis in humans. Lab. Anim. Res., 2022. [DOI]
- A. Mahomed, D. Girn, A. Pattani, B.K. Wells, C.C. King, S. Patel, H. Kaur, C.M. Noravian, J. Sieminski, C. Pham. Cannabinoid receptor type 1 regulates sequential stages of migration and morphogenesis of neural crest cells and derivatives in chicken and frog embryos. J. Morphol., 2023. [DOI]
- G. Tortoriello, J. Beiersdorf, S. Romani, G. Williams, G.A. Cameron, K. Mackie, M.J. Williams, V. Di Marzo, E. Keimpema, P. Doherty, T. Harkany. Genetic Manipulation of sn-1-Diacylglycerol Lipase and CB(1) Cannabinoid Receptor Gain-of-Function Uncover Neuronal 2-Linoleoyl Glycerol Signaling in Drosophila melanogaster. Cannabis Cannabinoid Res., 2021. [DOI | PubMed]
- M. Koch, L. Varela, J.G. Kim, J.D. Kim, F. Hernández-Nuño, S.E. Simonds, C.M. Castorena, C.R. Vianna, J.K. Elmquist, Y.M. Morozov. Hypothalamic POMC neurons promote cannabinoid-induced feeding. Nature, 2015. [DOI | PubMed]
- V. Di Marzo, S.K. Goparaju, L. Wang, J. Liu, S. Bátkai, Z. Járai, F. Fezza, G.I. Miura, R.D. Palmiter, T. Sugiura, G. Kunos. Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature, 2001. [DOI | PubMed]
- M. Maccarrone, I. Bab, T. Bíró, G.A. Cabral, S.K. Dey, V. Di Marzo, J.C. Konje, G. Kunos, R. Mechoulam, P. Pacher. Endocannabinoid signaling at the periphery: 50 years after THC. Trends Pharmacol. Sci., 2015. [DOI | PubMed]
- O. Sagredo, J. Palazuelos, A. Gutierrez-Rodriguez, V. Satta, I. Galve-Roperh, J. Martínez-Orgado. Cannabinoid signalling in the immature brain: Encephalopathies and neurodevelopmental disorders. Biochem. Pharmacol., 2018. [DOI | PubMed]
- B. Bányai, Z. Vass, S. Kiss, A. Balogh, D. Brandhuber, G. Karvaly, K. Kovács, G.L. Nádasy, L. Hunyady, G. Dörnyei. Role of CB1 Cannabinoid Receptors in Vascular Responses and Vascular Remodeling of the Aorta in Female Mice. Int. J. Mol. Sci., 2023. [DOI]
- S.K. Rouzer, A. Sreeram, R.C. Miranda. Reduced fetal cerebral blood flow predicts perinatal mortality in a mouse model of prenatal alcohol and cannabinoid exposure. BMC Pregnancy Childbirth, 2024. [DOI]
- Y.M. Morozov, M. Koch, P. Rakic, T.L. Horvath. Cannabinoid type 1 receptor-containing axons innervate NPY/AgRP neurons in the mouse arcuate nucleus. Mol. Metab., 2017. [DOI | PubMed]
- R.J. Rodrigues, J.M. Marques, A. Köfalvi. Cannabis, Endocannabinoids and Brain Development: From Embryogenesis to Adolescence. Cells, 2024. [DOI]
- M. Maccarrone, M. Guzmán, K. Mackie, P. Doherty, T. Harkany. Programming of neural cells by (endo)cannabinoids: from physiological rules to emerging therapies. Nat. Rev. Neurosci., 2014. [DOI | PubMed]
- I. Katona, T.F. Freund. Multiple functions of endocannabinoid signaling in the brain. Annu. Rev. Neurosci., 2012. [DOI | PubMed]
- D. Piomelli. The molecular logic of endocannabinoid signalling. Nat. Rev. Neurosci., 2003. [DOI | PubMed]
- R.I. Wilson, R.A. Nicoll. Endocannabinoid signaling in the brain. Science, 2002. [DOI | PubMed]
- I. Katona, E.A. Rancz, L. Acsady, C. Ledent, K. Mackie, N. Hajos, T.F. Freund. Distribution of CB1 cannabinoid receptors in the amygdala and their role in the control of GABAergic transmission. J. Neurosci., 2001. [PubMed]
- Y.M. Morozov, T.F. Freund. Post-natal development of type 1 cannabinoid receptor immunoreactivity in the rat hippocampus. Eur. J. Neurosci., 2003. [PubMed]
- Y.M. Morozov, K. Mackie, P. Rakic. Cannabinoid Type 1 Receptor is Undetectable in Rodent and Primate Cerebral Neural Stem Cells but Participates in Radial Neuronal Migration. Int. J. Mol. Sci., 2020. [DOI]
- A. Bacci, J.R. Huguenard, D.A. Prince. Long-lasting self-inhibition of neocortical interneurons mediated by endocannabinoids. Nature, 2004. [DOI | PubMed]
- S. Marinelli, S. Pacioni, A. Cannich, G. Marsicano, A. Bacci. Self-modulation of neocortical pyramidal neurons by endocannabinoids. Nat. Neurosci., 2009. [DOI | PubMed]
- P. Berghuis, A.M. Rajnicek, Y.M. Morozov, R.A. Ross, J. Mulder, G.M. Urbán, K. Monory, G. Marsicano, M. Matteoli, A. Canty. Hardwiring the brain: endocannabinoids shape neuronal connectivity. Science, 2007. [DOI | PubMed]
- E. Keimpema, K. Barabas, Y.M. Morozov, G. Tortoriello, M. Torii, G. Cameron, Y. Yanagawa, M. Watanabe, K. Mackie, T. Harkany. Differential subcellular recruitment of monoacylglycerol lipase generates spatial specificity of 2-arachidonoyl glycerol signaling during axonal pathfinding. J. Neurosci., 2010. [DOI | PubMed]
- M. Maccarrone, V. Di Marzo, J. Gertsch, U. Grether, A.C. Howlett, T. Hua, A. Makriyannis, D. Piomelli, N. Ueda, M. van der Stelt. Goods and Bads of the Endocannabinoid System as a Therapeutic Target: Lessons Learned after 30 Years. Pharmacol. Rev., 2023. [DOI | PubMed]
- A. Sideris, J. Lauzadis, M. Kaczocha. The Basic Science of Cannabinoids. Anesth. Analg., 2024. [DOI | PubMed]
- A. Gil-Ordóñez, M. Martín-Fontecha, S. Ortega-Gutiérrez, M.L. López-Rodríguez. Monoacylglycerol lipase (MAGL) as a promising therapeutic target. Biochem. Pharmacol., 2018. [DOI | PubMed]
- R.W. de Oliveira, C.L. Oliveira, F.S. Guimarães, A.C. Campos. Cannabinoid signalling in embryonic and adult neurogenesis: possible implications for psychiatric and neurological disorders. Acta Neuropsychiatr., 2018. [DOI | PubMed]
- C. Chen. Inhibiting degradation of 2-arachidonoylglycerol as a therapeutic strategy for neurodegenerative diseases. Pharmacol. Ther., 2023. [DOI]
- A. Zanfirescu, A. Ungurianu, D.P. Mihai, D. Radulescu, G.M. Nitulescu. Targeting Monoacylglycerol Lipase in Pursuit of Therapies for Neurological and Neurodegenerative Diseases. Molecules, 2021. [DOI]
- N. Pasquarelli, C. Porazik, H. Bayer, E. Buck, S. Schildknecht, P. Weydt, A. Witting, B. Ferger. Contrasting effects of selective MAGL and FAAH inhibition on dopamine depletion and GDNF expression in a chronic MPTP mouse model of Parkinson’s disease. Neurochem. Int., 2017. [DOI | PubMed]
- L.D. Schurman, A.H. Lichtman. Endocannabinoids: A Promising Impact for Traumatic Brain Injury. Front. Pharmacol., 2017. [DOI]
- D. Panikashvili, C. Simeonidou, S. Ben-Shabat, L. Hanus, A. Breuer, R. Mechoulam, E. Shohami. An endogenous cannabinoid (2-AG) is neuroprotective after brain injury. Nature, 2001. [DOI | PubMed]
- R. Sacco, G.C. Riccitelli, G. Disanto, J. Bogousslavsky, A. Cavelti, D. Czell, C.P. Kamm, U. Kliesch, S.P. Ramseier, C. Gobbi, C. Zecca. Effectiveness, Safety and Patients’ Satisfaction of Nabiximols (Sativex(®)) on Multiple Sclerosis Spasticity and Related Symptoms in a Swiss Multicenter Study. J. Clin. Med., 2024. [DOI]
- R.A. Gray, B.J. Whalley. The proposed mechanisms of action of CBD in epilepsy. Epileptic Disord., 2020. [DOI | PubMed]
- M. Guzmán, M.J. Duarte, C. Blázquez, J. Ravina, M.C. Rosa, I. Galve-Roperh, C. Sánchez, G. Velasco, L. González-Feria. A pilot clinical study of Δ9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br. J. Cancer, 2006. [DOI | PubMed]
- M. Zaiachuk, N. Pryimak, O. Kovalchuk, I. Kovalchuk. Cannabinoids, Medical Cannabis, and Colorectal Cancer Immunotherapy. Front. Med., 2021. [DOI]
- P.P. Bathula, M.B. Maciver. Cannabinoids in Treating Chemotherapy-Induced Nausea and Vomiting, Cancer-Associated Pain, and Tumor Growth. Int. J. Mol. Sci., 2023
- C. Pagano, G. Navarra, L. Coppola, G. Avilia, M. Bifulco, C. Laezza. Cannabinoids: Therapeutic Use in Clinical Practice. Int. J. Mol. Sci., 2022
- N.M. Pennant, C.V. Hinton. The evolution of cannabinoid receptors in cancer. WIREs Mech. Dis., 2023. [DOI]
- G. Bononi, M. Di Stefano, G. Poli, G. Ortore, P. Meier, F. Masetto, I. Caligiuri, F. Rizzolio, M. Macchia, A. Chicca. Reversible Monoacylglycerol Lipase Inhibitors: Discovery of a New Class of Benzylpiperidine Derivatives. J. Med. Chem., 2022. [DOI | PubMed]
- G. Nahler. Treatment of malignant diseases with phytocannabinoids: promising observations in animal models and patients. Explor. Med., 2023. [DOI]
- L.C. Ornelas, E.W. Fish, J.C. Dooley, M. Carroll, S.E. Parnell, J. Besheer. The impact of prenatal alcohol, synthetic cannabinoid and co-exposure on behavioral adaptations in adolescent offspring and alcohol self-administration in adulthood. Neurotoxicol. Teratol., 2024. [DOI]
- M.K. Mulligan, K.M. Hamre. Influence of prenatal cannabinoid exposure on early development and beyond. Adv. Drug Alcohol Res., 2023. [DOI]
- Y. Kalukula, A.D. Stephens, J. Lammerding, S. Gabriele. Mechanics and functional consequences of nuclear deformations. Nat. Rev. Mol. Cell Biol., 2022. [DOI | PubMed]
- Y. Jung-Garcia, O. Maiques, J. Monger, I. Rodriguez-Hernandez, B. Fanshawe, M.C. Domart, M.J. Renshaw, R.M. Marti, X. Matias-Guiu, L.M. Collinson. LAP1 supports nuclear adaptability during constrained melanoma cell migration and invasion. Nat. Cell Biol., 2023. [DOI | PubMed]
- G.R. Kidiyoor, Q. Li, G. Bastianello, C. Bruhn, I. Giovannetti, A. Mohamood, G.V. Beznoussenko, A. Mironov, M. Raab, M. Piel. ATR is essential for preservation of cell mechanics and nuclear integrity during interstitial migration. Nat. Commun., 2020. [DOI]
- G. Uzer, G. Bas, B. Sen, Z. Xie, S. Birks, M. Olcum, C. McGrath, M. Styner, J. Rubin. Sun-mediated mechanical LINC between nucleus and cytoskeleton regulates βcatenin nuclear access. J. Biomech., 2018. [DOI | PubMed]
- M. Shokrollahi, M. Stanic, A. Hundal, J.N.Y. Chan, D. Urman, C.A. Jordan, A. Hakem, R. Espin, J. Hao, R. Krishnan. DNA double-strand break-capturing nuclear envelope tubules drive DNA repair. Nat. Struct. Mol. Biol., 2024. [DOI | PubMed]
- B.E. Danielsson, B. George Abraham, E. Mäntylä, J.I. Cabe, C.R. Mayer, A. Rekonen, F. Ek, D.E. Conway, T.O. Ihalainen. Nuclear lamina strain states revealed by intermolecular force biosensor. Nat. Commun., 2023. [DOI]
- J.W. Tsai, K.H. Bremner, R.B. Vallee. Dual subcellular roles for LIS1 and dynein in radial neuronal migration in live brain tissue. Nat. Neurosci., 2007. [DOI | PubMed]
- D.J. Solecki, L. Model, J. Gaetz, T.M. Kapoor, M.E. Hatten. Par6alpha signaling controls glial-guided neuronal migration. Nat. Neurosci., 2004. [DOI | PubMed]
- P. Rakic, E. Knyihar-Csillik, B. Csillik. Polarity of microtubule assemblies during neuronal cell migration. Proc. Natl. Acad. Sci. USA, 1996. [DOI | PubMed]
- A. Bellion, J.P. Baudoin, C. Alvarez, M. Bornens, C. Métin. Nucleokinesis in tangentially migrating neurons comprises two alternating phases: forward migration of the Golgi/centrosome associated with centrosome splitting and myosin contraction at the rear. J. Neurosci., 2005. [DOI | PubMed]
- N.Y. Chen, Y. Yang, T.A. Weston, J.N. Belling, P. Heizer, Y. Tu, P. Kim, L. Edillo, S.J. Jonas, P.S. Weiss. An absence of lamin B1 in migrating neurons causes nuclear membrane ruptures and cell death. Proc. Natl. Acad. Sci. USA, 2019. [DOI | PubMed]
- C.M. Denais, R.M. Gilbert, P. Isermann, A.L. McGregor, M. te Lindert, B. Weigelin, P.M. Davidson, P. Friedl, K. Wolf, J. Lammerding. Nuclear envelope rupture and repair during cancer cell migration. Science, 2016. [DOI | PubMed]
- M. Raab, M. Gentili, H. de Belly, H.R. Thiam, P. Vargas, A.J. Jimenez, F. Lautenschlaeger, R. Voituriez, A.M. Lennon-Duménil, N. Manel, M. Piel. ESCRT III repairs nuclear envelope ruptures during cell migration to limit DNA damage and cell death. Science, 2016. [DOI | PubMed]
- M. Pho, Y. Berrada, A. Gunda, A. Lavallee, K. Chiu, A. Padam, M.L. Currey, A.D. Stephens. Actin contraction controls nuclear blebbing and rupture independent of actin confinement. Mol. Biol. Cell, 2024. [DOI]
- J.D. Vargas, E.M. Hatch, D.J. Anderson, M.W. Hetzer. Transient nuclear envelope rupturing during interphase in human cancer cells. Nucleus, 2012. [DOI | PubMed]
- C.T. Halfmann, K.L. Scott, R.M. Sears, K.J. Roux. Mechanisms by which barrier-to-autointegration factor regulates dynamics of nucleocytoplasmic leakage and membrane repair following nuclear envelope rupture. bioRxiv, 2023. [DOI]
- C.M. de Noronha, M.P. Sherman, H.W. Lin, M.V. Cavrois, R.D. Moir, R.D. Goldman, W.C. Greene. Dynamic disruptions in nuclear envelope architecture and integrity induced by HIV-1 Vpr. Science, 2001. [DOI | PubMed]
- W.H. De Vos, F. Houben, M. Kamps, A. Malhas, F. Verheyen, J. Cox, E.M.M. Manders, V.L.R.M. Verstraeten, M.A.M. van Steensel, C.L.M. Marcelis. Repetitive disruptions of the nuclear envelope invoke temporary loss of cellular compartmentalization in laminopathies. Hum. Mol. Genet., 2011. [DOI | PubMed]
- J. Maciejowski, E.M. Hatch. Nuclear Membrane Rupture and Its Consequences. Annu. Rev. Cell Dev. Biol., 2020. [DOI | PubMed]
- Y. Kamikawa, A. Saito, K. Imaizumi. Impact of Nuclear Envelope Stress on Physiological and Pathological Processes in Central Nervous System. Neurochem. Res., 2022. [DOI | PubMed]
- Z. Dou, C. Xu, G. Donahue, T. Shimi, J.A. Pan, J. Zhu, A. Ivanov, B.C. Capell, A.M. Drake, P.P. Shah. Autophagy mediates degradation of nuclear lamina. Nature, 2015. [DOI | PubMed]
- Z. Yang, J. Maciejowski, T. de Lange. Nuclear Envelope Rupture Is Enhanced by Loss of p53 or Rb. Mol. Cancer Res., 2017. [DOI | PubMed]
- Q. Zhang, A.C. Tamashunas, A. Agrawal, M. Torbati, A. Katiyar, R.B. Dickinson, J. Lammerding, T.P. Lele. Local, transient tensile stress on the nuclear membrane causes membrane rupture. Mol. Biol. Cell, 2019. [DOI | PubMed]
- Z. Dou, K. Ghosh, M.G. Vizioli, J. Zhu, P. Sen, K.J. Wangensteen, J. Simithy, Y. Lan, Y. Lin, Z. Zhou. Cytoplasmic chromatin triggers inflammation in senescence and cancer. Nature, 2017. [DOI | PubMed]
- L. Lindenboim, D. Grozki, A.R. Amsalem-Zafran, A. Peña-Blanco, G.G. Gundersen, C. Borner, D. Hodzic, A.J. Garcia-Sáez, H.J. Worman, R. Stein. Apoptotic stress induces Bax-dependent, caspase-independent redistribution of LINC complex nesprins. Cell Death Discov., 2020. [DOI]
- L. Lindenboim, T. Sasson, H.J. Worman, C. Borner, R. Stein. Cellular stress induces Bax-regulated nuclear bubble budding and rupture followed by nuclear protein release. Nucleus, 2014. [DOI | PubMed]
- E.M. Hatch, M.W. Hetzer. Nuclear envelope rupture is induced by actin-based nucleus confinement. J. Cell Biol., 2016. [DOI | PubMed]
- A. Karoutas, W. Szymanski, T. Rausch, S. Guhathakurta, E.A. Rog-Zielinska, R. Peyronnet, J. Seyfferth, H.R. Chen, R. de Leeuw, B. Herquel. The NSL complex maintains nuclear architecture stability via lamin A/C acetylation. Nat. Cell Biol., 2019. [DOI | PubMed]
- S.M. Schreiner, P.K. Koo, Y. Zhao, S.G.J. Mochrie, M.C. King. The tethering of chromatin to the nuclear envelope supports nuclear mechanics. Nat. Commun., 2015. [DOI]
- A.D. Stephens, E.J. Banigan, J.F. Marko. Chromatin’s physical properties shape the nucleus and its functions. Curr. Opin. Cell Biol., 2019. [DOI | PubMed]
- C.T. Halfmann, K.J. Roux. Barrier-to-autointegration factor: a first responder for repair of nuclear ruptures. Cell Cycle, 2021. [DOI | PubMed]
- Y. Kamikawa, Z. Wu, N. Nakazawa, T. Ito, A. Saito, K. Imaizumi. Impact of cell cycle on repair of ruptured nuclear envelope and sensitivity to nuclear envelope stress in glioblastoma. Cell Death Discov., 2023. [DOI]
- Y.M. Morozov, M. Torii, P. Rakic. Origin, early commitment, migratory routes, and destination of cannabinoid type 1 receptor-containing interneurons. Cereb. Cortex, 2009. [DOI | PubMed]
- C. Bernard, M. Milh, Y.M. Morozov, Y. Ben-Ari, T.F. Freund, H. Gozlan. Altering cannabinoid signaling during development disrupts neuronal activity. Proc. Natl. Acad. Sci. USA, 2005. [DOI | PubMed]
- T. Navaratnarajah, R. Anand, A.S. Reichert, F. Distelmaier. The relevance of mitochondrial morphology for human disease. Int. J. Biochem. Cell Biol., 2021. [DOI]
- J.R. Friedman, J. Nunnari. Mitochondrial form and function. Nature, 2014. [DOI | PubMed]
- R.J. Youle, A.M. van der Bliek. Mitochondrial fission, fusion, and stress. Science, 2012. [DOI | PubMed]
- V. Joukov, A. De Nicolo. The Centrosome and the Primary Cilium: The Yin and Yang of a Hybrid Organelle. Cells, 2019. [DOI]
- Y.M. Morozov, P. Rakic. Lateral expansion of the mammalian cerebral cortex is related to anchorage of centrosomes in apical neural progenitors. Cereb. Cortex, 2024. [DOI]
- J.I. Arellano, S.M. Guadiana, J.J. Breunig, P. Rakic, M.R. Sarkisian. Development and distribution of neuronal cilia in mouse neocortex. J. Comp. Neurol., 2012. [DOI | PubMed]
- Y.M. Morozov, Y.Y. Sun, C.Y. Kuan, P. Rakic. Alteration of SLP2-like immunolabeling in mitochondria signifies early cellular damage in developing and adult mouse brain. Eur. J. Neurosci., 2016. [DOI | PubMed]
- Y.M. Morozov, D. Datta, C.D. Paspalas, A.F.T. Arnsten. Ultrastructural evidence for impaired mitochondrial fission in the aged rhesus monkey dorsolateral prefrontal cortex. Neurobiol. Aging, 2017. [DOI | PubMed]
- Y.M. Morozov, P. Rakic. Disorder of Golgi Apparatus Precedes Anoxia-Induced Pathology of Mitochondria. Int. J. Mol. Sci., 2023. [DOI]
- A.M. Bertholet, T. Delerue, A.M. Millet, M.F. Moulis, C. David, M. Daloyau, L. Arnauné-Pelloquin, N. Davezac, V. Mils, M.C. Miquel. Mitochondrial fusion/fission dynamics in neurodegeneration and neuronal plasticity. Neurobiol. Dis., 2016. [DOI | PubMed]
- A. Zimmer, A.M. Zimmer, A.G. Hohmann, M. Herkenham, T.I. Bonner. Increased mortality, hypoactivity, and hypoalgesia in cannabinoid CB1 receptor knockout mice. Proc. Natl. Acad. Sci. USA, 1999. [DOI | PubMed]
- C. Dias, J. Nylandsted. Neural membrane repair at the core of regeneration. Neural Regen. Res., 2024. [DOI | PubMed]
- J. Yuan, D. Ofengeim. A guide to cell death pathways. Nat. Rev. Mol. Cell Biol., 2024. [DOI | PubMed]
- L. Galluzzi, J.M. Bravo-San Pedro, I. Vitale, S.A. Aaronson, J.M. Abrams, D. Adam, E.S. Alnemri, L. Altucci, D. Andrews, M. Annicchiarico-Petruzzelli. Essential versus accessory aspects of cell death: recommendations of the NCCD 2015. Cell Death Differ., 2015. [DOI | PubMed]
- G. Kroemer, L. Galluzzi, P. Vandenabeele, J. Abrams, E.S. Alnemri, E.H. Baehrecke, M.V. Blagosklonny, W.S. El-Deiry, P. Golstein, D.R. Green. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ., 2009. [DOI | PubMed]
- M.L. Mencel, G.D. Bittner. Repair of traumatic lesions to the plasmalemma of neurons and other cells: Commonalities, conflicts, and controversies. Front. Physiol., 2023. [DOI]
- C. Dias, J. Nylandsted. Plasma membrane integrity in health and disease: significance and therapeutic potential. Cell Discov., 2021. [DOI]
- S.T. Cooper, P.L. McNeil. Membrane Repair: Mechanisms and Pathophysiology. Physiol. Rev., 2015. [DOI | PubMed]
- B.K. Hendricks, R. Shi. Mechanisms of neuronal membrane sealing following mechanical trauma. Neurosci. Bull., 2014. [DOI | PubMed]
- S. Xu, T.J. Yang, S. Xu, Y.N. Gong. Plasma membrane repair empowers the necrotic survivors as innate immune modulators. Semin. Cell Dev. Biol., 2024. [DOI | PubMed]
- H.R. Bulgart, I. Goncalves, N. Weisleder. Leveraging Plasma Membrane Repair Therapeutics for Treating Neurodegenerative Diseases. Cells, 2023. [DOI]
- G. Mereu, M. Fà, L. Ferraro, R. Cagiano, T. Antonelli, M. Tattoli, V. Ghiglieri, S. Tanganelli, G.L. Gessa, V. Cuomo. Prenatal exposure to a cannabinoid agonist produces memory deficits linked to dysfunction in hippocampal long-term potentiation and glutamate release. Proc. Natl. Acad. Sci. USA, 2003. [DOI | PubMed]
- T. Antonelli, M.C. Tomasini, M. Tattoli, T. Cassano, S. Tanganelli, S. Finetti, E. Mazzoni, L. Trabace, L. Steardo, V. Cuomo, L. Ferraro. Prenatal exposure to the CB1 receptor agonist WIN 55,212-2 causes learning disruption associated with impaired cortical NMDA receptor function and emotional reactivity changes in rat offspring. Cereb. Cortex, 2005. [DOI | PubMed]
- K.C. Tree, M. Scotto di Perretolo, J. Peyronnet, F. Cayetanot. In utero cannabinoid exposure alters breathing and the response to hypoxia in newborn mice. Eur. J. Neurosci., 2014. [DOI | PubMed]
- E. Fride. The endocannabinoid-CB(1) receptor system in pre- and postnatal life. Eur. J. Pharmacol., 2004. [DOI | PubMed]
- P. Tagliaferro, A. Javier Ramos, E.S. Onaivi, S.G. Evrard, J. Lujilde, A. Brusco. Neuronal cytoskeleton and synaptic densities are altered after a chronic treatment with the cannabinoid receptor agonist WIN 55,212-2. Brain Res., 2006. [DOI | PubMed]
- S.J. Jackson, G. Pryce, L.T. Diemel, M.L. Cuzner, D. Baker. Cannabinoid-receptor 1 null mice are susceptible to neurofilament damage and caspase 3 activation. Neuroscience, 2005. [DOI | PubMed]
- D. Soriano, M. Vacotto, A. Brusco, L. Caltana. Neuronal and synaptic morphological alterations in the hippocampus of cannabinoid receptor type 1 knockout mice. J. Neurosci. Res., 2020. [DOI | PubMed]
- X.B. Yuan, M. Jin, X. Xu, Y.Q. Song, C.P. Wu, M.M. Poo, S. Duan. Signalling and crosstalk of Rho GTPases in mediating axon guidance. Nat. Cell Biol., 2003. [DOI | PubMed]
- A. Hall. Rho family GTPases. Biochem. Soc. Trans., 2012. [DOI | PubMed]
- J. Guggisberg, M. Schumacher, G. Gilmore, D.M. Zylla. Cannabis as an Anticancer Agent: A Review of Clinical Data and Assessment of Case Reports. Cannabis Cannabinoid Res., 2022. [DOI | PubMed]
- C.J. Fowler. Delta(9) -tetrahydrocannabinol and cannabidiol as potential curative agents for cancer: A critical examination of the preclinical literature. Clin. Pharmacol. Ther., 2015. [DOI | PubMed]
- F. Afrin, M. Chi, A.L. Eamens, R.J. Duchatel, A.M. Douglas, J. Schneider, C. Gedye, A.S. Woldu, M.D. Dun. Can Hemp Help? Low-THC Cannabis and Non-THC Cannabinoids for the Treatment of Cancer. Cancers, 2020. [DOI]
- C. Ledent, O. Valverde, G. Cossu, F. Petitet, J.F. Aubert, F. Beslot, G.A. Böhme, A. Imperato, T. Pedrazzini, B.P. Roques. Unresponsiveness to cannabinoids and reduced addictive effects of opiates in CB1 receptor knockout mice. Science, 1999. [DOI | PubMed]
- S. Parmentier-Batteur, K. Jin, X.O. Mao, L. Xie, D.A. Greenberg. Increased Severity of Stroke in CB1 Cannabinoid Receptor Knock-Out Mice. J. Neurosci., 2002. [DOI | PubMed]
- J.C. Fiala. Reconstruct: a free editor for serial section microscopy. J. Microsc., 2005. [DOI | PubMed]
- J.C. Fiala, K.M. Harris. Extending unbiased stereology of brain ultrastructure to three-dimensional volumes. J. Am. Med. Inform. Assoc., 2001. [PubMed]
- E. Leishman, M.N. Murphy, M.I. Murphy, K. Mackie, H.B. Bradshaw. Broad and Region-Specific Impacts of the Synthetic Cannabinoid CP 55,940 in Adolescent and Adult Female Mouse Brains. Front. Mol. Neurosci., 2018. [DOI]
- S.J. Ward, L.A. Dykstra. The role of CB1 receptors in sweet versus fat reinforcement: effect of CB1 receptor deletion, CB1 receptor antagonism (SR141716A) and CB1 receptor agonism (CP-55940). Behav. Pharmacol., 2005. [DOI | PubMed]
- M. Hajós, W.E. Hoffmann, B. Kocsis. Activation of cannabinoid-1 receptors disrupts sensory gating and neuronal oscillation: relevance to schizophrenia. Biol. Psychiatry, 2008. [DOI | PubMed]
- B.G. Rash, N. Micali, A.J. Huttner, Y.M. Morozov, T.L. Horvath, P. Rakic. Metabolic regulation and glucose sensitivity of cortical radial glial cells. Proc. Natl. Acad. Sci. USA, 2018. [DOI | PubMed]
