Unravelling herbicide stress and its impact on metabolite profiling in Cannabis sativa: an investigative study
Abstract
Background:
Cannabis sativa L., renowned for its versatility in pharmaceutical, textile, and cosmetic industries, is highly susceptible to several agronomic and environmental factors, particularly herbicides. These chemical agents, while commonly used for weed control, can adversely affect plant growth, physiology, and secondary metabolite production. Understanding the plant’s response to such external stressors is essential for optimizing its cultivation and ensuring the quality of its bioactive compounds.
Methods:
In our current work, we studied the impact of two herbicides- glyphosate and metribuzin on the morpho-physiological and biochemical characteristics of cannabis plants. The secondary metabolite production analysis was carried out using Gas Chromatography-Mass S pectrometry (GC-MS). Furthermore, in silico studies using molecular modelling and optimization via Density Functional Theory (DFT) were performed, followed by molecular docking.
Results:
It was observed that both herbicides greatly impact overall plant productivity including primary and secondary metabolite production. Further, glyphosate treatment caused an increase in fatty acid synthesis while the contrary was observed in case of metribuzin. Also, herbicide stress leads to the synthesis of cannabidivarol and cannabidiol although they were absent in the untreated group. These findings provide crucial insights for optimizing agricultural practices in cannabis cultivation. Moreover, molecular simulation results showed that both metribuzin and glyphosate bind at the active pocket of Tetrahydrocannabinolic acid synthase (THCA synthase) and offer a mechanistic explanation for the observed variations in Δ9 -tetrahydocannabinol (THC) levels by suggesting that both herbicides inhibit THCA synthase activity, contributing to a deeper understanding of herbicide-plant interactions at the molecular level.
Conclusions:
Our findings indicate that herbicide stress impacts overall cannabis productivity and alters biosynthesis. The stress notably stimulates the production of cannabidivarol and cannabidiol. In addition, molecular docking studies revealed that metribuzin binds to the same active channel as Cannabigerolic acid (CBGA)- the THC precursor, while glyphosate binds at the entrance, thereby hindering THC production. This multifaceted approach guides sustainable farming strategies and has implications for manipulating cannabinoid profiles in pharmaceutical and other industrial applications.
Supplementary Information:
The online version contains supplementary material available at 10.1186/s42238-025-00300-z.
Article type: Research Article
Keywords: Phytocannabinoids, Herbicide stress, DFT, Molecular Docking, THC
Affiliations: https://ror.org/00et6q107grid.449005.c0000 0004 1756 737XSchool of Bioengineering and Biosciences, Lovely Professional University, Phagwara, 144411 Punjab India; https://ror.org/00et6q107grid.449005.c0000 0004 1756 737XDivision of Research and Development, Lovely Professional University, Phagwara, 144411 Punjab India; https://ror.org/05eer8g02grid.411903.e0000 0001 2034 9160Department of Biomedical Sciences, Institute of Health, Jimma University, Jimma, 00000 Ethiopia; https://ror.org/04fhee747grid.19100.390000 0001 2176 7428Gene Regulation Laboratory, National Institute of Immunology, New Delhi, 110067 India
License: © This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s42238-025-00300-z | PubMed: 40624563 | PMC: PMC12236017
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (3.0 MB)
Introduction
Over the last decade, the prevailing agricultural practice of growing a limited number of crops in monoculture has faced substantial criticism due to its adverse impact on the environment. Both the recent European Union policy and the common worldwide trend emphasize the need for diversified cropping and the revival of numerous abandoned crops. Among such crops, Cannabis sativa L. which belongs to the family Cannabaceae, has received heightened attention from farmers (Durak et al. ref. 2021). It is one of the earliest domesticated plants with a cultivation history spanning over 6000 years, substantiated by archeological evidence. It is a multipurpose crop with applications ranging from textile fiber (Réquilé et al. ref. 2021), food (Clarke and Merlin ref. 2016), paper production (Karche and Singh ref. 2019), pharmaceutical products (Legare et al. ref. 2022), cosmetics (de Andrade et al. ref. 2023), biofuel production (Bharath Viswanathan et al. ref. 2020), biomass (energy) (Rehman et al. ref. 2021), and much more(Abdollahi et al. ref. 2020) and can thus be considered a fundamental cash crop.
Cannabis plants contribute significantly to environmental well-being by absorbing up to 10 metric tons of carbon dioxide per vegetation cycle and promoting air quality, thermal balance, and ecological stability. Thus, the agricultural sector is increasingly drawn to cannabis cultivation for its environmental advantages and growing cannabis product market, aligning with sustainable practices and climate change mitigation throughout the entire value chain (Raihan and Bijoy ref. 2023). Cannabis (Cannabis sativa L.) is also gaining prominence as an industrial crop due to its large number of applications (Peng and Shahidi ref. 2021). Market liberalization and UN reclassification have driven global expansion, increasing demand for cannabis fiber and derivatives (Guedes et al. ref. 2024; Simiyu et al. ref. 2022). The global cannabis market is projected to grow rapidly, reaching over USD 444 billion by 2030 (Gallien and Occhiali ref. 2023). Cannabis is estimated to possess more than 550 molecules of bioactive compounds (Lowe et al. ref. 2021), belonging to alkaloids, flavonoids, terpenoids, carotenoids, and cannabinoid classes (Andre et al. ref. 2016). Currently, research on Cannabis sativa L. is reviving in several disciplines, including microbiology and cancer (Giupponi et al. ref. 2020; Pollastro et al. ref. 2018). These compounds can solve therapeutic issues such as antibiotic resistance and the toxicity that results from ingestion and metabolism while treating cancer (Hourfane et al. ref. 2023). However, various agronomic and environmental factors can influence the cannabis productivity. As the plant’s cultivation expands, it becomes important to understand the effects of commonly applied agrochemicals on its morphological, physiological, and biochemical aspects.
Today’s agronomy is dependent on herbicide application, which although is greatly advantageous but also poses threats to the biosphere. With the legalization of cannabis cultivation, herbicides are one of the stressors that have the potential to affect its production (Kaur et al. ref. 2025). Among the herbicides which are used globally, glyphosate and metribuzin are widely applied (Kostopoulou et al. ref. 2020). Glyphosate is a broad-spectrum non-specific herbicide with a half-life of 2 to 215 days in soil and 2 to 91 days in water (Singh et al. ref. 2020). Glyphosate inhibits 5-enolpruvylshikimate-3-phosphate synthase (EPSPS) enzyme in the shikimic acid pathway, thereby preventing the synthesis of three aromatic amino acids- phenylalanine, tyrosine, and tryptophan in plants (Liu et al. ref. 2023). These are vital precursors for numerous plant metabolites involved in defense, growth, and stress response (Fuchs et al. ref. 2021). This disruption induces oxidative stress, alters phytohormonal balance, reduces photosynthetic efficiency, and impairs plant-microbe interactions (Eceiza et al.ref. 2022a; Gomes et al. ref. 2016). Glyphosate exposure can also lead to lipid peroxidation and degradation of chlorophyll, severely affecting the plant’s biochemical homeostasis (Gomes et al. ref. 2016). On the other hand, metribuzin is a selective triazinone herbicide, used pre- and post-emergence to control broadleaf weeds. It competes with plastoquinone on D1 protein for QB-binding site, therefore blocking the transfer of electrons from QA to QB leading to photooxidation and ultimately causing plant death (Atef Abdel Fatah et al. ref. 2023). Metribuzin is known for its moderate persistence in soil with a 30 to 120-day half-life under field conditions (Maqueda et al. ref. 2009). Its water solubility and leaching properties make it a potential hazard to non-target organisms by contaminating surface and groundwater (Karimmojeni et al. ref. 2022). Metribuzin application has been associated with increased production of reactive oxygen species (ROS), lipid peroxidation, and compromised antioxidant defense systems in several plant species (Kumar et al. ref. 2022). Metabolomic studies further suggest that metribuzin alters amino acid pools and activates salicylate signaling pathways, reflecting substantial physiological shifts under herbicide-induced stress (Kostopoulou et al. ref. 2020).
Increasing evidence supports the notion that environmental and agronomic stress conditions significantly influence cannabinoid biosynthesis in Cannabis sativa (Gorelick and Bernstein ref. 2014). Salinity stress has been shown to reduce shoot and bud weight and suppress total THC and CBD yields (Baas and Wijnenref. 2023a). Similarly, nutritional stress such as phosphorus (P) and potassium (K) deficiencies has led to elevated THC concentrations (Song et al. ref. 2023), despite overall lower plant biomass (Saloner and Bernsteinref. 2022a; Shiponi and Bernstein ref. 2021a). Conversely, high P levels tend to dilute cannabinoid content, likely due to a yield-dilution effect (Shiponi and Bernstein ref. 2021a). Nitrogen stress also plays a critical role: high ammonium/nitrate (NH₄⁺/NO₃⁻) ratios reduce cannabinoid biosynthesis, whereas low nitrogen conditions were found to increase it (Saloner and Bernstein ref. 2022b). Moreover, abiotic stresses such as drought, UV radiation, and salinity are known to modulate secondary metabolite pathways, providing further insights into the adaptive strategies of cannabis under stress (Gorelick and Bernstein ref. 2017). In addition to environmental stresses, agronomic practices such as planting density (Danziger and Bernstein ref. 2022), and plant shape (Danziger and Bernstein ref. 2021) which affect microclimate conditions in the shoot; and low root salinity levels affected cannabinoid yield (Baas and Wijnen ref. 2023a).
The understanding of cannabis-herbicide interaction is limited, impeding the development of optimized crop protection strategies (Kaur et al. ref. 2025). Glyphosate and metribuzin herbicides are frequently applied during the cannabis season and unintentional exposure through drift is a documented concern for non-target crops (McNaughton et al. ref. 2012). These findings underscore the importance of understanding herbicide-induced stress responses within the broader context of plant-environment interactions. Thus, in the present investigation, these herbicides were sprayed on the groups of cannabis plants chosen for the study to evaluate the potential impact of herbicide drift from surrounding fields on non-target Cannabis sativa plants. Further critical analysis was carried out through biochemical studies. Moreover, molecular docking analysis of THCA synthase with glyphosate and metribuzin was performed to validate the results obtained through Gas chromatography-mass spectrometry (GC-MS) analysis for ∆9-Tetrahydrocannabinol (THC) for variation in its synthesis across treatments with different herbicide concentrations. THC is the most powerful psychoactive cannabinoid, and thus, it becomes one of the primary focuses of our investigations (Citti et al. ref. 2019).
Materials and methods
Plant material
The Cannabis sativa L. seeds were collected from a roadside growing wild plant which was dioecious (female) and initial identification was carried out by Botany Faculty, LPU. Licenses are not required for collection and research working on a wild variety of Cannabis sativa in India. The herbarium was prepared and sent to a national referral facility: Janaki Ammal Herbarium CSIR-IIIM. The plant identification was performed by a taxonomist Dr. Sumeet Gairola, at the Council of Scientific and Industrial Research- Indian Institute of Integrative Medicine (CSIR-IIIM) Jammu [accession number = 26 832]. The healthy and uniform seeds were surface sterilized for 15 min using 0.1% Sodium hypochloride. Further, the seeds were rinsed and soaked with distilled water for 20 min. The seeds were germinated uptill 4 leaf stage and plants exhibiting uniform developmental characteristics were transferred in plastic pots containing soil that was not previously treated with any type of chemical. The pots were kept in a greenhouse under controlled conditions at a mean temperature of 24–28℃ in an agricultural field at Lovely Professional University, Phagwara, Punjab. Five weeks later, different concentrations of glyphosate and metribuzin were used to treat the plants. Both glyphosate (active ingredients 40.6% w/w, Agri Venture) and metribuzin (active ingredients 88% w/w, TATA) herbicides used in the current experiment were of industrial grade. The experiment comprised of following treatments: T0 (control), T1 (X/8), T2 (X/4), T3 (X/2), T4(X), where X (2.47 L/ha for glyphosate and 742 g/ha for metribuzin) is the recommended herbicide dose. Each treatment was replicated three times. Glyphosate samples were collected after three days (de Freitas-Silva et al. ref. 2020) of treatment and metribuzin samples after seven days (Kumar et al. ref. 2022) of treatment for morphological analysis and were stored at -20℃ for biochemical analysis.
Fresh weight, dry weight, and leaf water relations
After harvesting, the fresh weight (FW) of the plants was measured immediately; however, for dry biomass, the plants were placed in an oven set at 80 ± 1.5°C and weighed after five days [30]. Leaf relative water content (RWC) was calculated according to (Barrs and Weatherley ref. 1962), RWC%= (FW − DW)/(TW − DW) x 100, where FW is fresh leaf weight, DW is dry weight and TW is turgid weight after 24 h floating in distilled water at 4 °C in darkness.
Photosynthetic pigments
Leaf pigments i.e., chlorophyll, carotenoids were extracted with 80% acetone and anthocyanins were extracted with acidified methanol (methanol: water: HCl, 79:20:1). Chlorophyll, carotenoids, and anthocyanins were estimated by methods given by (Arnon ref. 1949), (Maclachlan and Zalik ref. 1963) and (Mancinelli ref. 1984) respectively. The absorbance of supernatant was noted at 645 nm, 663 nm, 480 nm, 510 nm, 530 nm, and 657 nm using UV-vis spectrophotometer (LI-2800 Ex) and calculated through the following formulas: chlorophyll a (mg g− 1 FW)= (Abs663 × 12.7) −(Abs645 × 2.69) × V/(1000 × W); chlorophyll b (mg g− 1 FW)= (Abs645 × 22.9) − (Abs663 × 4.68) × V/(1000 × W); total chlorophyll content (mg g− 1 FW)= (Abs645 × 22.2) + (Abs663 × 8.03) × V/(1000 × W); total carotenoid content (mg g− 1 FW) = 1000 (Abs480) − 1.8 (Chl a)– 85.02 (Chl b)/ 198; anthocyanin content = Abs530– 0.25(Abs657).
Protein and sugar Estimation
Protein content was determined using supernatant obtained through the method by (Lowry et al. ref. 1951) by plotting its absorbance against the BSA standard. Subsequently, Total Soluble Carbohydrate was estimated according to (Islam et al. ref. 2021) and sucrose content was determined according to (van Handel ref. 1968). 25 mg of leaf sample was homogenized in 95% of 5 mL ethanol, followed by centrifugation (3500 rpm), and the supernatant was used for sugar estimation. Glucose and sucrose were used to plot the standard curve by taking absorbance at 625 nm and 620 nm.
Antioxidant enzyme activity
In a pre-chilled pestle and mortar, 0.1 g of plant material was crushed using 3 mL of 100 mM potassium phosphate buffer (PPB) at a pH of 7.0. Then the crushed material was centrifuged for 20 min at 13,000 rpm and 4°C. The supernatant was then collected to calculate various antioxidant activities. Peroxidase (POD) activity, catalase activity (CAT), and Malondialdehyde (MDA) contents were determined by the methods given by (Pütter ref. 1974), (Aebi et al. ref. 1983), and (Heath and Packer ref. 1968) respectively.
Secondary metabolite Estimation
The conventional method of maceration extraction was utilized to examine the bioactive compounds found in cannabis leaves (Pande and Chanda ref. 2020). Fresh cannabis leaves were air-dried at room temperature (below 24°C) in a shaded and ventilated area before being ground into fine powder for extraction. 1 g of dried leaf powder was then extracted at room temperature for 24 h in 100 mL of methanol by placing it in shaking incubator. The mixture was then paper-filtered and kept at room temperature until the excess solvent evaporated. After getting the extract, it was kept at 4°C in an airtight container for secondary metabolite estimation. The extract was then reconstituted in methanol and filtered through 0.22 micron nylon syringe filter (Moxcare labware) before injecting into the GC. The analysis was conducted at Lovely Professional University’s Central Instrumentation Facility using a Shimadzu gas chromatograph instrument model, GCMS-TQ8040 NX, equipped with a mass detector. 1µL of sample was injected into GC via autosampler. The injector was used in splitless mode. Additionally, the injector was set at a temperature of 250℃. The compounds were separated using an SH-RXi-5Sil MS capillary column and crosslinked to a 30 m × 0.25 mmID, 0.25 μm df (similar to 5% diphenyl/95%dimethyl polysiloxane). The column temperature was initially set at 40°C and then it was raised to 300°C at a rate of 7°C per minute and maintained for three minutes. The carrier gas utilized was helium (flow rate = 1 mL min− 1). The National Institute of Standards and Technology’s spectrum database was used to find and identify the components in the extract. Within the m/z range of 40 to 1000, detection was carried out using the Q3 scan acquisition mode. The proportion of an individual component is presented as a relative peak area percentage of the total peak percentage (Vadhel et al. ref. 2023).
Molecular Docking studies
Protein Preparation
The protein 3D structure of the Cannabis sativa L. tetrahydrocannabinolic acid synthase (THCA synthase) was acquired from the Protein Data Bank (PDB ID 3VTE), with a resolution of 2.75 Å having no mutation (Shoyama et al. ref. 2012). The flavine adenine dinucleotide (FAD) ligand was kept as it is responsible for the enzyme’s catalytic activity, and the protein was cleaned by removing other ligands and water molecules using AutoDockTools 1.5.6. Polar hydrogens and Kollmann charges were added, and Gasteiger charges were computed. The atomic type was set to assign AD4 type (Verma et al. ref. 2023a).
Ligand Preparation
The 3D canonical structures of ligands- Cannabigerolic acid (CBGA), metribuzin, and glyphosate, were drawn using Marvin sketch software. 2D and 3D cleaning was performed each time, and structures were checked for 3D geometrical conformation by visualizing them in Marvin’s view. Further, the structures were optimized to transition state, and the energy was minimized sequentially using Chem3D Pro 12.0.2, Avogadro 1.2, and Chem3D 22. The ligand with the most minimized energy was finally optimized using DFT via Gaussian 09 software. Here, we utilized RB3LYP functionals and a 6-311G basis set. These optimized structures were further used for docking purposes to determine the interactions of ligands with the enzyme THCA Synthase (Gulati et al. ref. 2023).
Molecular Docking
The Rigid molecular docking experiment was performed with optimized ligands using Autodock 4.2. Grid box dimensions were set to cover the whole protein at a distance of 0.714 Å (Supplementary Table S1). The Genetic Algorithm simulation program was used with a 300 population size, 100 genetic algorithm runs, and a medium number of evaluations for metribuzin and glyphosate due to a medium number of torsions, whereas a long number of evaluations were used for Cannabigerolic acid (CBGA) due to a higher number of torsions in CBGA, and the remaining parameters were set to their default values. The output file was saved as Lamarckian GA. Each ligand’s top 10 conformations were saved based on the negative binding energy (∆G). Conformations binding to the same active site and the most negative binding energy in all three were further analyzed in Discovery Studio Visualizer v21.1 and ChimeraX 1.7 (Verma et al. ref. 2022, ref. 2023a).
Statistical analysis
The collected data was analyzed using one-way Analysis of Variance (ANOVA) with a significance level of ≤ 0.05. Duncan multiple comparison test was conducted using IBM SPSS version 22 software. The different letters on each error bar are statistically significant. Graphs were plotted using OriginLab software. The chemical structures were drawn with ChemAxon Marvin Sketch software.
Result
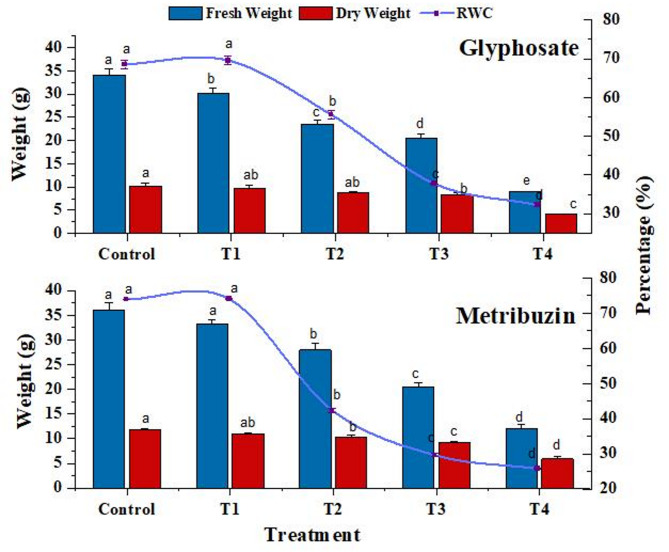
Fresh weight, dry weight, and relative water content
Increased herbicide stress led to a significant reduction in fresh and dry weights, with glyphosate and metribuzin exposure compared to the control at the highest concentration (T4). The RWC of leaves markedly decreased in both herbicide treatments, reaching its lowest value at T4 (30.3% for glyphosate and 28.6% for metribuzin) (Fig. 1).
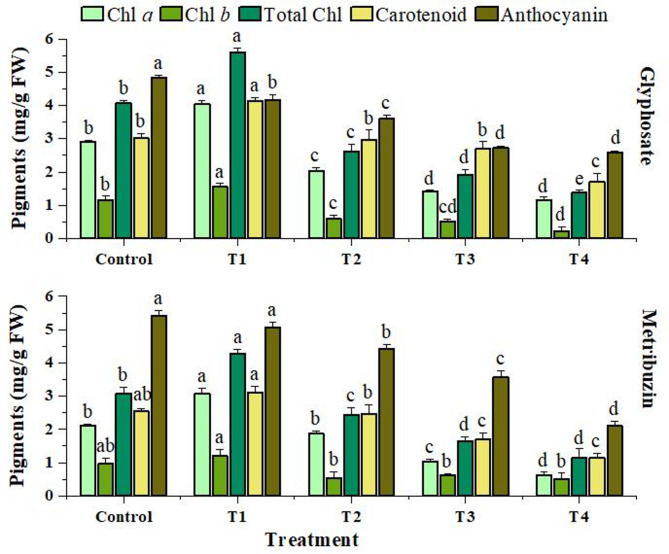
Photosynthetic pigments
The photosynthetic pigments showed a significant fold reduction in levels of Chlorophyll a (2.51, 3.34), Chlorophyll b (5.2, 1.89), total Chlorophyll (2.95, 2.69), and carotenoids (1.77, 1.64) under highest glyphosate and metribuzin herbicide stress (T4) as compared to the control group. However, at the lowest concentration (T1), a notable increase was observed in the levels of pigments. Anthocyanin content also showed a decline with increasing stress of 1.8-fold in glyphosate and 1.12-fold in metribuzin at T4 (Fig. 2).
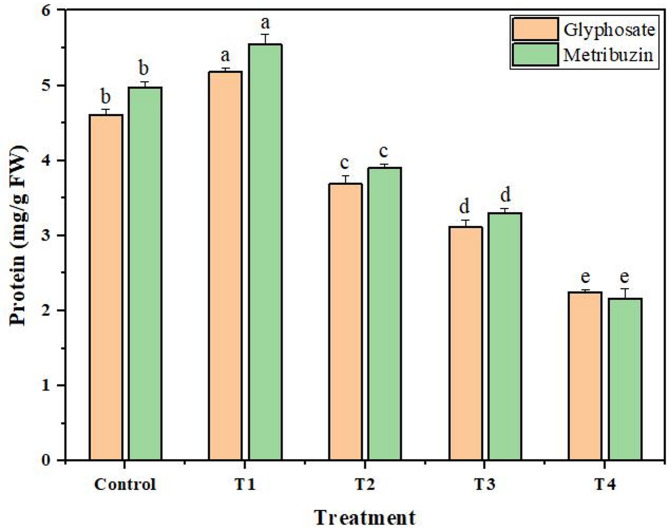
Protein content
The protein content declined in correlation with an increase in concentrations of both glyphosate and metribuzin. The maximum reduction in protein content reaches 2.04-fold at T4 − the highest glyphosate stress level. Similarly, with the increase in metribuzin concentration, the protein content was observed to decrease, culminating in a maximum reduction of 2.29-fold at the most intensive metribuzin stress point − T4. This indicates a consistent pattern of decreasing protein content with heightened herbicide concentrations except at the lowest doses where a 1.12-fold and 1.11-fold increase in protein content was observed for glyphosate and metribuzin respectively in comparison to the untreated group (Fig. 3).
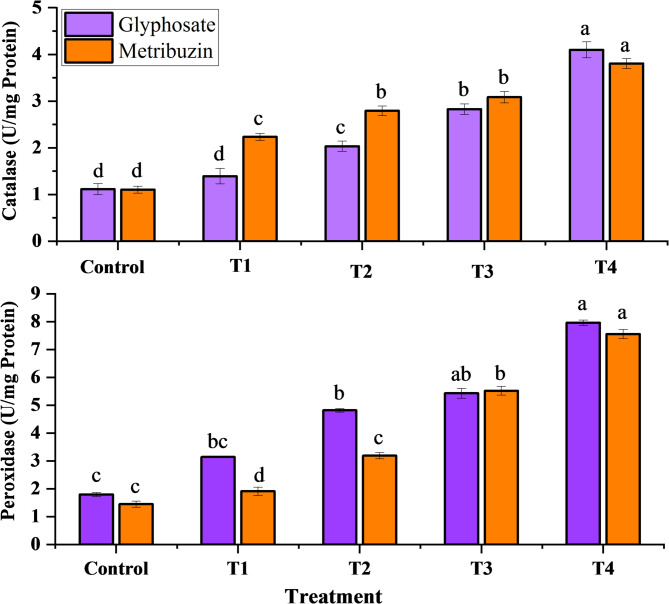
Antioxidant enzymes
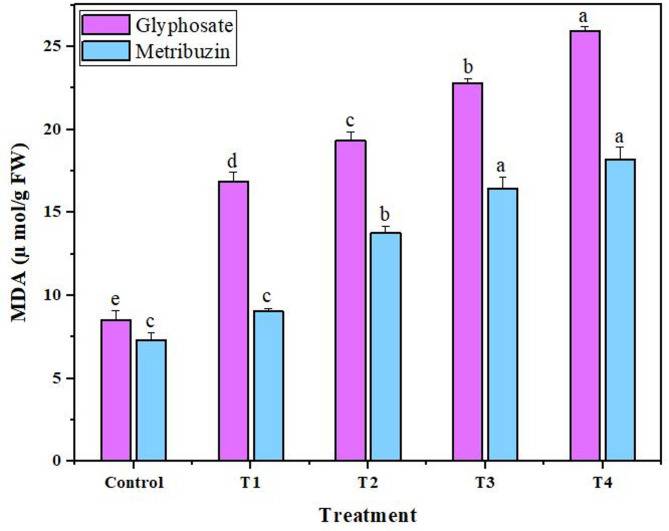
The observed data indicated a dose-dependent escalation in peroxidase and catalase activities for both glyphosate and metribuzin treatments. The most substantial enhancement in catalase and peroxidase activities, reaching 3.6-fold and 3.7-fold for glyphosate, and 3.4-fold and 5.2-fold for metribuzin compared to the control, was notably evident at T4, signifying the highest response to oxidative stress (Fig. 4). Simultaneously, the Malondialdehyde (MDA) levels, serving as an index of oxidative damage, consistently rose with increasing concentrations of the herbicides. At T4, MDA also exhibited its highest content, displaying an increase of 3.0-fold and 2.4-fold compared to the control, underlining the culmination of oxidative stress at the highest concentration levels of both glyphosate and metribuzin respectively (Fig. 5).
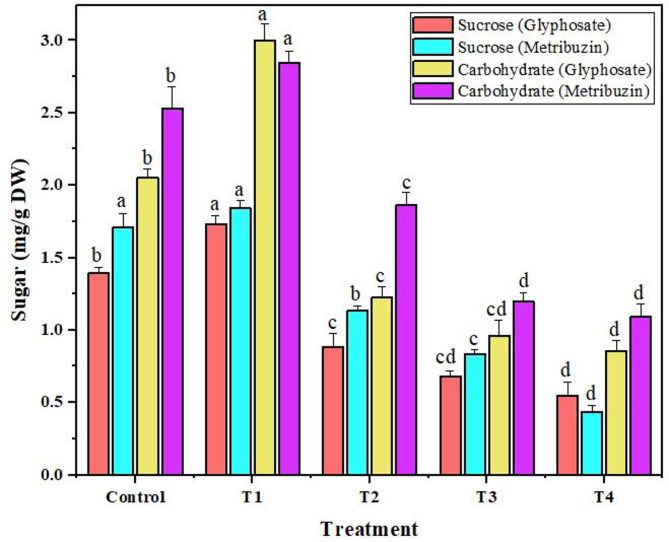
Carbohydrate and sucrose
In accordance with the behavior of photosynthetic pigments, the levels of carbohydrate and sucrose mirrored a similar trend when subjected to varying herbicide concentrations. Both compounds displayed a consistent decrease as the herbicide concentration increased, aligning with the anticipated impact on these plant sugars. An interesting deviation from this trend emerged at the lowest concentration (T1), where a slight increase of 1.4-fold and 1.2-fold (glyphosate) and 1.12-fold and 1.07-fold (metribuzin) was observed in both carbohydrate and sucrose levels respectively. Remarkably, when exposed to glyphosate, the most significant reduction percentages were noted at the T4, measuring 2.3-fold for carbohydrates and 2.5-fold for sucrose. Comparatively, with metribuzin application, the reduction percentages were recorded at 2.3-fold for carbohydrates and 3.9-fold for sucrose at the same T4 concentration (Fig. 6).
Bioactive compound identification
The methanolic leaf extract of the cannabis plant comprised various compounds identified (Supplentary Table S6). Among the identified compounds, the most abundant compounds were hexadecenoic acid, methyl stearate, 2,4-Di-tert-butylphenol (2,4-DTBP), and eicosane. The most important phytocannabinoids included delta.9-tetrahydrocannabivarin (THCV), dronabinol (THC), cannabinol (CBN), Cannabidivarol (CBDV), and cannabidiol (CBD). Dronabinol is the synthetic active enantiomer of Δ9 -tetrahydocannabinol (THC) which is a plant-derived cannabinoid (Devinsky et al. ref. 2015). Therefore, in this paper, the word THC has been used in place of dronabinol. Other compounds including heptadecane, hexadecane, heneicosane, docosane, and tetracosane were also found in the extract in trace amounts (Supplementary Figure S1).
Semi-quantitative analysis of bioactive compounds from C. sativa L
2,4-Di-tert-butylphenol (2,4-DTBP), an allelopathic component identified in cannabis, has shown significant variation when exposed to different concentrations of glyphosate and metribuzin herbicides. Interestingly, an increase in area percent of 2,4-DTBP was noted at lower concentrations of both herbicides, which gradually decreased as the herbicide stress increased, indicating a direct link between herbicide stress and 2, 4-DTBP biosynthesis (Fig. 7).
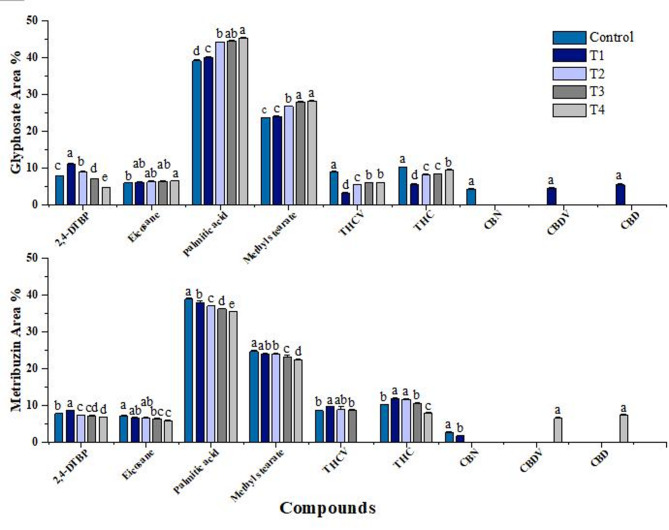
A dynamic profile of fatty acid methyl esters, including methyl stearate, hexadecanoic acid, hexadecane, heptadecane, eicosane, etc. was also observed in GC-MS analysis of leaf extract of cannabis. The glyphosate-induced stress led to increase in fatty acids area percentage (Fig. 7). Consequently, stress induced by metribuzin had an inverse effect, resulting in a decline of the fatty acids.
The presence of cannabinoids including THCV, THC, cannabinol, Cannabidivarol, cannabidiol, etc. were also observed. The present data revealed an increase in area percent of THCV and THC under glyphosate stress while metribuzin stress exhibited a negative correlation (Fig. 7). However, the results of THC show some inconsistencies as in the case of metribuzin treatment, the THC area percent in T1 is higher which can be explained by hormesis. On the other hand, glyphosate resulted in a decrease in THC at the lowest concentration. The cannabinol content was observed exclusively in the control group and the lowest concentration of metribuzin. In contrast, Cannabidivarol and cannabidiol were absent in the control group but were present at the lowest concentration of glyphosate and the highest concentration of metribuzin.
Ligand structural comparison and optimization
This study conducted a comparative analysis of the physicochemical characteristics of CBGA, metribuzin, and glyphosate (Supplementary Table S2). The aromatic ring (AR) is responsible for π − π interactions with amino acid residues, which are present in both CBGA and metribuzin leading to an increase in binding affinity and influencing biological activity. Glyphosate has the highest hydrogen bond acceptors (HBA) and hydrogen bond donors (HBD) which positively influence the number of hydrogen bonds between the ligand and the enzyme THCA Synthase; however, the number of rotatable bonds (RB) in metribuzin is lower than in glyphosate. Furthermore, metribuzin has a slightly greater molecular weight (MW) compared to glyphosate.
After initial energy minimization using several software programs, CBGA, and metribuzin still exhibited positive ∆G energy, while glyphosate demonstrated negative ∆G energy (Supplementary Table S3). Upon Gaussian optimization, all three ligands exhibited minimal negative ∆G energy (Supplementary Figure S2, S3, S4). CBGA had the lowest energy, followed by metribuzin and glyphosate (Supplementary Table S4). Upon optimization, the bond length and angle of CBGA and metribuzin exhibited minor modifications; however, in the case of glyphosate, considerable alterations were observed (Supplementary Table S5).
Molecular Docking
Molecular docking studies were performed to understand the impact of varying glyphosate and metribuzin herbicide concentrations on the biosynthesis of THC by analyzing their possible interactions with the active site of THCA synthase. THCA synthase is the enzyme that converts the CBGA precursor into THC, hence CBGA was also docked with THCA synthase, and the docking model is shown in (Fig. 8). (Fig. 9) shows the docking model of metribuzin and glyphosate at the same site respectively.
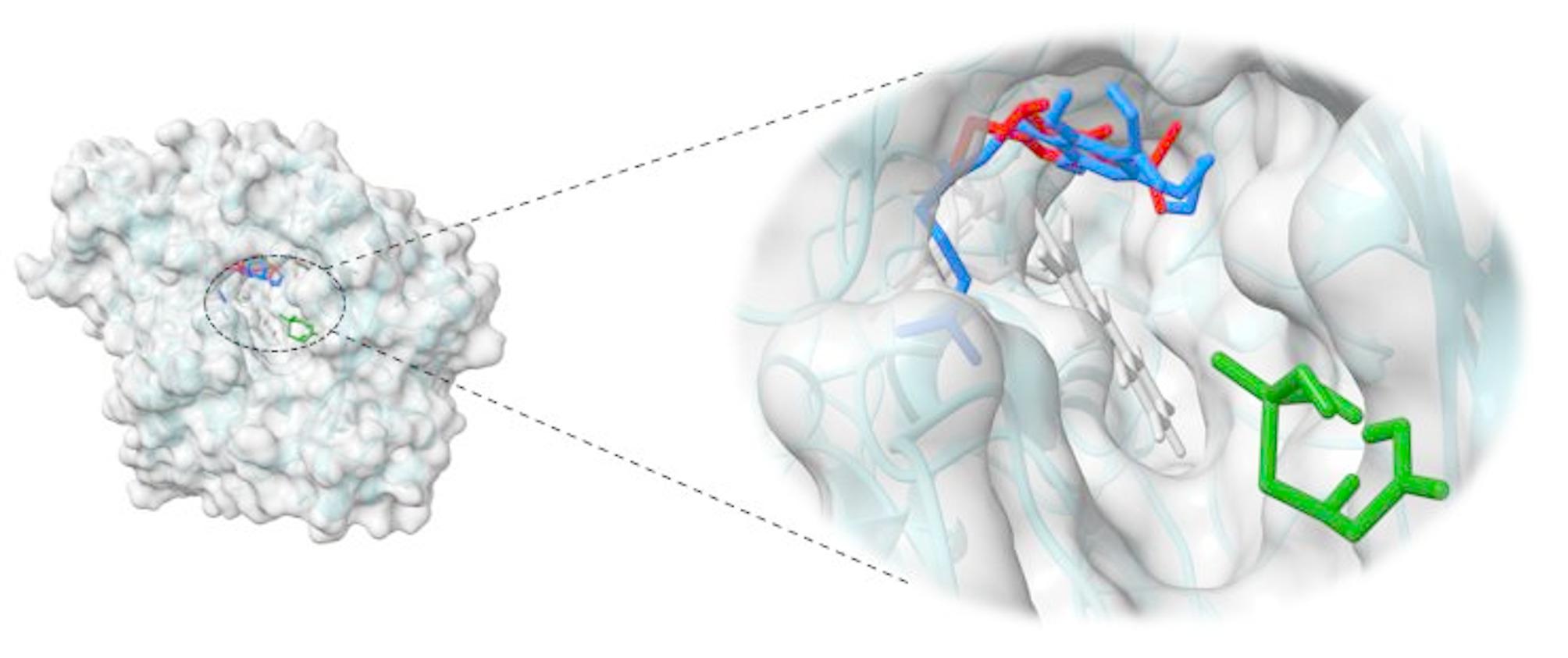
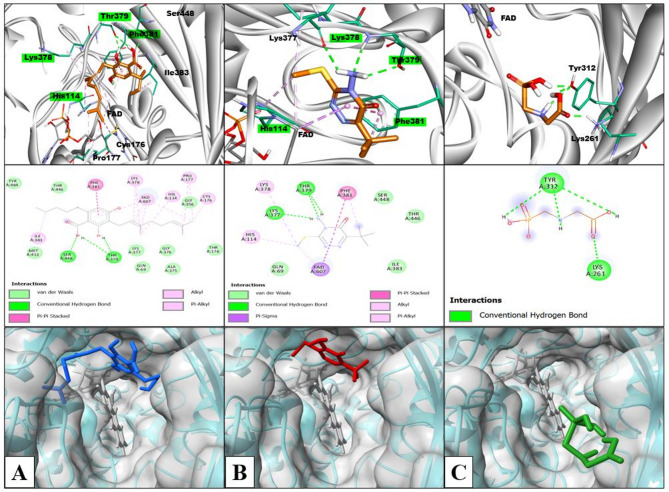
Table 1: Docking results of CBGA, metribuzin, and glyphosate with THCA synthase along with interacting amino acids
| Ligand | Interacting amino acids(H-bond) | Hydrophobic interaction | van der Waal’s interaction | Number of hydrogen bonds | Binding Affinity (Kcal/mol) |
|---|---|---|---|---|---|
| CBGA | Thr 379, Ser 448 | FAD, His 114, Lys 378, Phe 381, Cys 176, Pro 177, Ile 383 | Gln 69, Thr 446, Thr 178, Gly 356, Ala 375, Gly 376, Lys 377, Met 413, Tyr 484, | 3 | -6.06 |
| Metribuzin | Thr 379, Lys 377 | FAD, His 114, Lys 378, Phe 381 | Gln 69, Thr 446, Ser 448, Ile 383 | 3 | -5.79 |
| Glyphosate | Lys 261, Tyr 312 | NA | NA | 4 | -2.87 |
Metribuzin was completely embedded in the active site and showed interactions with the same amino acids as CBGA. Both metribuzin and CBGA formed conventional hydrogen bonds, alkyl, and π-alkyl interactions with Thr 379, Lys 378, and His 114, respectively. Phe 381 interacted with CBGA and metribuzin with π − π stacked interactions and an additional π-Alkyl interaction with metribuzin. FAD, responsible for the core catalytic activity of enzyme THCA synthase, formed alkyl interaction with CBGA and metribuzin. On top of that, it also formed π-alkyl interaction with CBGA and π-π interaction with metribuzin. Moreover, both CBGA and metribuzin interacted with Thr 446, Ile 383, Ser448, Lys 377, and Gln 69 to form alkyl, conventional hydrogen bond, and van der Waal’s interactions. Therefore, metribuzin and CBGA bind at the same active site with similar kind of interactions with binding energies of -5.79Kcal/mol and -6.06Kcal/mol respectively. From the docking result, it is clear that the interaction of metribuzin is slightly lower than the CBGA; but both are bound deep inside the active site as there are slight differences in their binding energies. However, glyphosate was observed to be present at the entrance of the active site as its binding energy is less (-2.87Kcal/mol) as compared to the former two i.e., metribizin and CBGA. Glyphosate interacted with Tyr 312 and Lys 261 amino acid residues of THCA synthase by forming conventional hydrogen bonds as shown in Table 1. The same observation was obtained from the GC-MS analysis that supports our in-silico result.
Glyphosate vs. Metribuzin at the recommended dose
The studied parameters showed detrimental effects in terms of herbicide stress. The comparison at the recommended dose showed glyphosate had a severe impact on RWC (30.3%), photosynthetic pigments (2.95-fold reduction for total chlorophyll, 1.77-fold reduction for carotenoid), anthocyanin (1.8-fold reduction), carbohydrate (2.39-fold reduction), catalase (3.6-fold increase) and MDA (3.0-fold increase). However, an exception in protein (2.29-fold reduction), sucrose (3.9-fold reduction), and peroxidase (5.2-fold increase) were observed. THC, an important pharmaceutical compound was impacted more by metribuzin (7.9% relative peak area) than by glyphosate (9.33% relative peak area) as observed by GC-MS analysis. Thus, we can conclude that comparatively glyphosate impacts the morpho-physiological and primary metabolites more with some exceptions whereas metribuzin impacts THC production at the recommended dose.
Discussion
The RWC indicates the extent of dehydration of leaves. Due to the treated plant’s lower water content, the overall weight shift was less pronounced even though fresh weight and water content both fell, suggesting increased cellular concentration. Another study also recorded similar results (Issaad et al. ref. 2022), where RWC decreased with glyphosate and other herbicide applications with elevation observed at the lowest concentration.
The reduction in chlorophyll content in case of glyphosate can be attributed to an upsurge in chlorophyll degradation, as documented in the study by (Gomes et al. ref. 2016). Similarly, the decline in carotenoids can result from the suppression of the shikimate pathway, which lowers the concentration of plastoquinone (PQ). The decrease in PQ content is significant because it functions as a cofactor for two essential enzymes in the carotenoid biosynthesis pathway: phytoene desaturase and ζ-carotene desaturase. Plastoquinone’s declining concentration therefore exerts a direct effect on carotenoid production (Gomes et al. ref. 2017). Furthermore, blocking EPSPS inhibits chorismate, which in turn is responsible for phenylalanine synthesis and ultimately anthocyanin via the phenylpropanoid pathway. This helps to explain the decrease in anthocyanin concentration with increasing glyphosate stress (Fuchs et al. ref. 2021). The results obtained for metribuzin treatment can be explained because Quinone A (QA) to Quinone B (QB) electron transport is impeded by metribuzin’s binding to the D1 protein on the plastoquinone binding site. Pigment production is hindered by this obstruction of electron transport, which results in electron buildup and ultimately leads to an increase in radiation. As plastoquinone remains unreduced, it causes triplet chlorophyll to build up. The triplet chlorophyll can produce singlet oxygen in the absence of efficient quenchers (Krieger-Liszkay ref. 2004). The accumulation of singlet oxygen and triplet chlorophyll can result in lipid peroxidation, which alters the fluidity of the membrane causing oxidative damage, and consequently declining the production of chlorophyll, carotenoid, and anthocyanin (Kruk and Szymańska ref. 2021). Similar results were observed in a study (Gomes et al. ref. 2016), where different concentrations of glyphosate to willow trees led to a reduction of photosynthetic pigments and ultimately reduction in photosynthesis. Another study (Volova et al. ref. 2020) showed a reduction in photosynthetic pigments while studying the biological effects of metribuzin on various weed species.
Although glyphosate does not directly affect the production of proteins in plants, it indirectly reduces protein levels by blocking the activity of the vital enzyme EPSPS. The production of phenylalanine, tyrosine, and tryptophan- essential aromatic amino acids needed for protein synthesis are curtailed as a result of this suppression (Ahsan et al. ref. 2008). On the contrary, the metribuzin-induced decline in protein content doesn’t stem from ammonia scarcity but rather from a probable insufficiency in its conversion into organic forms. Metribuzin inhibits the enzyme system involving glutamine synthetase (GS) and glutamine-2-oxoglutarate aminotransferase (GOGAT) which are pivotal for ammonia conversion causing accumulation of unconverted ammonia and impacting protein synthesis (Nemat Alla et al. ref. 2008). Reduction in protein content was also observed in a study (Nemat Alla et al. ref. 2008) while studying the effect of field dose application of metribuzin on 10-day-old wheat and maize seedlings. Another study (Radwan and Fayez ref. 2016) showed that glyphosate reduced the protein content and showed other morphological symptoms in Arachis hypogaea leaves.
The principal action mechanism of glyphosate is to inhibit EPSPS, a key component in the shikimate pathway, for the synthesis of aromatic amino acids. The accumulation of shikimate and its derivatives because of the disturbed shikimate pathway puts the plant under oxidative stress (Yokoyama et al. ref. 2021). Metribuzin similarly obstructs photosynthesis by suppressing photosystem II (PSII), preventing electron transfer, and causing an excess of reactive oxygen species (ROS) to be produced in plant cells (Kostopoulou et al. ref. 2020). Antioxidant enzymes like peroxidase and catalase are upregulated as a result of the stress that triggers the plant’s defense mechanisms. The higher amounts of reactive oxygen species (ROS) brought on by the disruption in the metabolic pathways are neutralized and detoxified by these enzymes. Nevertheless, the imbalance brought on by herbicide stress can outweigh the plant’s defense mechanisms despite the antioxidant response, which can lead to the building up of malondialdehyde (MDA), a sign of lipid peroxidation and cellular damage (Khaleghnezhad et al. ref. 2021). Therefore, the simultaneous increase in MDA concentration indicates oxidative damage and disruption by herbicide application, while the increase in antioxidant enzymes demonstrates the plant’s attempt to fight oxidative stress (Karimmojeni et al. ref. 2022). Similar results have been observed by (Eceiza et al. ref. 2022b), where glyphosate-resistant and glyphosate-sensitive Amaranthus palmeri were analyzed to determine glyphosate-induced oxidative stress and found that an increase in antioxidant enzymes is associated with EPSPS inhibition. Another study (Karimmojeni et al. ref. 2022) analyzed that at high metribuzin dose, Echinacea purpurea leaves showed an increase in MDA levels. Similarly, while studying the toxicity assessment of metribuzin on Vigna radiata, it was found that metribuzin-treated plants induced antioxidant enzyme activities (Kumar et al. ref. 2023).
Distinct in their methods, glyphosate and metribuzin cause a fall in the quantities of carbohydrates and sucrose through targeted interference with basic metabolic processes. The inhibition of EPSPS by glyphosate and inhibition of photosystem II by metribuzin results in the reduction of photosynthesis, which explains the decrease of carbohydrate and sucrose levels under herbicide stress (Pline et al. ref. 2003). Change in the photosynthetic process directly impacts the concentration of carbohydrate and sucrose levels (Vital et al. ref. 2017). Analogous results were observed in a study (Razieh Rajabi ref. 2012), where the foliar application of metribuzin resulted in carbohydrate reduction. Another study (Salman et al. ref. 2016) was conducted, where the effect of glyphosate on biochemical features of Oscillatoria limnetica resulted in a marked decrease in carbohydrates. Various other studies (Beker Akbulut et al. ref. 2018; Vital et al. ref. 2017), also noticed a reduction in sugar levels in glyphosate-treated sunflower and maize plants.
The unexpected results observed at the lowest herbicide concentration can be explained through hormesis, a dose-response phenomenon wherein low doses promote biological responses while high doses lead to their inhibition. Various herbicides like glyphosate at low concentrations can induce plant growth regulation through a variety of mechanisms like auxin production, antioxidant defense, and anatomical modifications, affecting rhizosphere cation transporters (Islam et al. ref. 2017). Lower glyphosate levels hinder the shikimic acid pathway, which lowers lignin and promotes better plant growth (Nguyen et al. ref. 2016). In the current study, the hormesis effect due to metribuzin has been documented for the first time. Herbicide sub-doses promote physiological and enzymatic processes as well as plant growth (Brito et al. ref. 2018). Plant growth and function are ultimately enhanced by this adaptive mechanism, which is triggered by exposure to low-stress levels and activation of cellular defense mechanisms (Jalal et al. ref. 2021).
The bioactive compounds identified via GC-MS analysis in our study were similar to compounds observed in other studies (Tayyab and Shahwar ref. 2015; Vadhel et al. ref. 2023; Wishart et al. ref. 2024). 2,4-DTBP, despite being identified in at least 169 species of organisms, there is yet no explanation as to why organisms produce 2,4-DTBP, a toxic lipophilic phenol. A recent study revealed that healthy rice plants show similar levels of 2,4-DTBP as shown by viral infection and insect herbivory samples (Zhao et al. ref. 2020). Nevertheless, more experimentation is required to confirm the correlation between stress and 2,4-DTBP production.
Plants, being sessile organisms, strategically utilize fatty acid production as a crucial part of their adaptive stress response to minimize the negative impact (He and Ding ref. 2020). Many aliphatic chemicals, such as membrane glycerolipids, TAG, cutin/suberin, jasmonates, and nitroalkenes (NO2-FAs), are produced by plants using fatty acids as raw material. All these compounds including fatty acids contribute to plant defense against different biotic and abiotic stressors (He and Ding ref. 2020; de Oliveira et al. ref. 2022). According to a study (Filimonova et al. ref. 2018), there are reports of herbicides having ecotoxicological and biochemical effects on a variety of marine plankton species, leading to an increase in the proportion of fatty acids in response to toxicity. The mechanisms may include alteration in the de novo synthesis of fatty acids through fatty acid synthetase enzyme and/or via modification of existing lipids via desaturase enzyme action. In a study, an increase in ~ 17% lipid content was observed in glyphosate-treated Chlorella sorokiniana as compared to the control (Jaiswal et al. ref. 2020). The effects of metribuzin on fatty acid synthesis are not well documented yet, but fatty acid synthesis is an energy-expensive process that requires ATP and NADPH at different steps; thus, it can be hypothesized that blocking of electron transport chain by metribuzin reduces energy production which in turn affects production of fatty acids. In addition, delay in electron transport from QA to QB results in over-reduction on the PSII acceptor side, which raises the chances of producing reactive oxygen species as explained in the photosynthetic pigments section, leading to lipid peroxidation in thylakoid membranes, resulting in a decrease in fatty acids (Roncel et al. ref. 2007). This complex interaction highlights the diverse ways in which plants react to herbicidal stresses and advances our knowledge of the complex mechanisms underlying plant adaptability.
The impact of herbicides on cannabinoids can be plausibly explained by the observed relationship between herbicide treatments and fatty acid synthesis as the cannabinoids are synthesized from Geranyl pyrophosphate (GPP), a precursor derived from Acetyl-CoA via the fatty acid synthesis pathway (Macherone ref. 2020). On the other hand, the decrease in THC at the lowest concentration of glyphosate is due to the biosynthesis of another compound CBD at the same treatment since THC and CBD are synthesized from the same precursor (CBGA); so even if the precursor were produced in greater concentrations as can be correlated from higher synthesis of fatty acids, the precursor would be utilized by the plant in the production of CBD in addition to THC. According to various studies, the synthesis of cannabinoids in plants varies under different environmental conditions. Drought stress, for example, was associated with an increase in cannabinoid production (Caplan et al. ref. 2019). Similarly, a study reported that the active compound THC increased when the plants were exposed to UVB, whereas identical conditions did not cause any changes in the plant’s cannabinoids (Lydon et al. ref. 1987). Copper was also found to cause a significant temporal increase in the production of THC and CBD (Cahill et al. ref. 2024). Abscisic acid has been implicated in affecting THC concentration in plants, although its effect remains controversial (Mansouri and Asrar ref. 2012). Contrastingly, cannabinoid levels were observed to decline under herbicide stress (Toth et al. ref. 2021). From these observations, it can be concluded that there is a complex relationship between environmental factors and upsurge and decline in cannabinoid levels which has not yet been studied in detail and needs further investigation.
Furthermore, in-silico studies provided valuable insights into the interactions of metribuzin and glyphosate with the target- THCA synthase enzyme. The interaction between the target enzyme and the ligand is influenced by the physicochemical properties and molecular structure of the ligands. The enzymatic activity of THCA synthase occurs in a hydrophobic environment (Rodziewicz et al. ref. 2019), which is consistent with the minimal topological polar surface area (TPSA) of its substrate CBGA. Compared to glyphosate, metribuzin possesses a lower TPSA value, suggesting metribuzin may more freely access and bind to THCA synthase as compared to glyphosate. Based on the physicochemical and optimization analysis, it was observed that metribuzin is more similar to CBGA than glyphosate. Molecular docking studies elucidated the impact of glyphosate and metribuzin on the biosynthesis of THC by analyzing their possible interactions with the active site of THCA synthase (Shoyama et al. ref. 2012). The comparative analysis of the three docking models provides valuable insights into the mechanisms of metribuzin and glyphosate in THC production. The models demonstrate that metribuzin directly competes with CBGA for binding to the active site of THCA synthase. Hence, it might be possible that metribuzin hinders the activity of CBGA by preventing CBGA from binding at the active site of THCA synthase and consequently reduces THC biosynthesis with an increase in metribuzin stress. Moreover, at higher metribuzin concentrations, there might be a reduction in CBGA production through inhibition of the fatty acid synthesis pathway that supplies CBGA. This dual impact of metribuzin, reducing both CBGA production and binding, leads to low activity of THCA synthase and the lowest area percent of THC at the highest concentration (T4) as compared to other treatment groups. In contrast, glyphosate partially blocks the entry to the active channel of THCA synthase, hindering CBGA binding to the active site, and thus resulting in a significant reduction in THC synthesis. The increase in fatty acids from glyphosate treatment produces a slight upward trend in THC levels due to the increased availability of CBGA. From our in-silico investigation, it can be concluded that while glyphosate partially blocks CBGA entry, still causing an increase in THC production due to precursor quantity enhancement, metribuzin leads to a downward trend in THC synthesis by binding to the THCA synthase active site and decreasing CBGA substrate availability through fatty acid pathway inhibition. Thus at the highest herbicide concentration, metribuzin has more impact on the cannabis plant’s THC production as compared to glyphosate as evident from the experimental data and supported by the in-silico data analysis.
The influence of herbicides on cannabinoid biosynthesis aligns with other documented stress responses in Cannabis sativa. For instance, nutrient imbalances such as P and K deficiency have been linked to increased THC production, suggesting that moderate nutrient stress can act as an elicitor (Saloner and Bernstein ref. 2022a). In contrast, salinity and high ammonium supply suppress cannabinoid levels (Baas and Wijnen ref. 2023a; Saloner and Bernstein ref. 2022b), indicating that not all stress types trigger beneficial responses. Such opposing responses highlight the nuanced regulation of secondary metabolism under stress. Our findings are consistent with this complex dynamic, where herbicides, as abiotic stressors, potentially function as elicitors of metabolic reprogramming. Future research should systematically explore the thresholds at which stress transitions from beneficial to detrimental in terms of cannabinoid yield and quality.
Conclusion
Current research work investigated the impact of glyphosate and metribuzin herbicides on Cannabis sativa plants. We observed that both herbicides significantly affected the production of primary and secondary metabolites, including pigments and sugar content. Antioxidant activities also increased with rising herbicide doses. However, at the lowest doses, hormesis occurred with both herbicides. Additionally, glyphosate treatment boosted fatty acid synthesis in cannabis, while metribuzin had the opposite effect. Overall, our findings indicate that herbicide stress impacts overall cannabis productivity and alters biosynthesis. The stress notably stimulates the production of cannabidivarol and cannabidiol, although the reasons behind this require further investigation. In addition, molecular docking studies revealed that metribuzin binds to the same active channel as CBGA- the THC precursor, while glyphosate binds at the entrance, thereby hindering the THC production. This comparative analysis of biochemical and in silico studies elucidates key factors in the disruption of THC biosynthesis by glyphosate and metribuzin.
References
- Abdollahi M, Sefidkon F, Calagari M, Mousavi A, Mahomoodally MF. Impact of four hemp (Cannabis sativa L.) varieties and stage of plant growth on yield and composition of essential oils. Ind Crops Prod. Elsevier B.V.; 2020. p. 155.
- U Aebi, WE Fowler, P Rew, TT Sun. The fibrillar substructure of keratin filaments unraveled. J Cell Biol, 1983. [DOI | PubMed]
- N Ahsan, DG Lee, KW Lee, I Alam, SH Lee, JD Bahk. Glyphosate-induced oxidative stress in rice leaves revealed by proteomic approach. Plant Physiol Biochem, 2008. [DOI | PubMed]
- Andre CM, Hausman J-F, Guerriero G. Cannabis sativa: the plant of the thousand and one molecules. Front Plant Sci. 2016;7.
- Arnon DI. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol [Internet]. 1949;24(1):1–15. Available from: https://academic.oup.com/plphys/article/24/1/1-15/6076157
- Atef Abdel Fatah M, Abd El-Moghny MG, El-Deab MS, Mohamed El Nashar R. Application of molecularly imprinted electrochemical sensor for trace analysis of Metribuzin herbicide in food samples. Food Chem [Internet]. 2023;404:134708. Available from: https://www.sciencedirect.com/science/article/pii/S030881462202670X
- Baas R, Wijnen D. Salinity effects on yield and nutrient uptake in Cannabis sativa L. Acta hortic. International Society for Horticultural Science; 2023a. pp. 785–91.
- H Barrs, P Weatherley. A Re-Examination of the relative turgidity technique for estimating water deficits in leaves. Aust J Biol Sci, 1962. [DOI]
- G Beker Akbulut, E Ylglt, D Bayram. Effect of glyphosate on some protective systems in Zea mays L. Yüzüncü Yıl Üniversitesi Tarım. Bilimleri Dergisi, 2018
- Bharath Viswanathan M, Park K, Cheng M-H, Cahoon EB, Clemente T, Singh V. Variability in structural carbohydrates, lipid composition, and cellulosic sugar production from industrial hemp varieties [Internet]. 2020. Available from: https://www.sciencedirect.com/science/article/abs/pii/S0926669020308232
- IP Brito, L Tropaldi, CA Carbonari, ED Velini. Hormetic effects of glyphosate on plants. Pest Manag Sci, 2018. [DOI | PubMed]
- MS Cahill, T Arsenault, TH Bui, N Zuverza-Mena, A Bharadwaj, K Prapayotin-Riveros. Copper stimulation of tetrahydrocannabinol and Cannabidiol production in hemp ( Cannabis sativa L.) is Copper-Type, dose, and cultivar dependent. J Agric Food Chem, 2024. [DOI | PubMed]
- Caplan D, Dixon M, Zheng Y. Increasing inflorescence dry weight and cannabinoid content in medical cannabis using controlled drought stress. Volume 54. HortScience. American Society for Horticultural Science; 2019. pp. 964–9. 5.
- C Citti, P Linciano, F Russo, L Luongo, M Iannotta, S Maione. A novel phytocannabinoid isolated from Cannabis sativa L. with an in vivo cannabimimetic activity higher than ∆9-tetrahydrocannabinol: ∆9-Tetrahydrocannabiphorol. Sci Rep, 2019. [DOI | PubMed]
- Clarke RC, Merlin MD. Cannabis Domestication, Breeding History, Present-day Genetic Diversity, and, Prospects F. CRC Crit Rev Plant Sci. Taylor and Francis Inc.; 2016;35(5–6):293–327.
- N Danziger, N Bernstein. Shape matters: plant architecture affects chemical uniformity in Large-Size medical Cannabis plants. Plants, 2021. [DOI | PubMed]
- Danziger N, Bernstein N. Too dense or not too dense: higher planting density reduces cannabinoid uniformity but increases yield/area in Drug-Type medical Cannabis. Front Plant Sci. 2022;13.
- de Andrade CML, Caetano TTV, Campos FK, Gandra VM, Alves FHF, Stein VC. Cannabis sativa L. in the cosmeceutical industry: prospects and biotechnological approaches for metabolite improvement. South African Journal of Botany [Internet]. 2023;161:171–9. Available from: https://www.sciencedirect.com/science/article/pii/S0254629923004593
- L de Freitas-Silva, TO de Araújo, A Nunes-Nesi, C Ribeiro, AC Costa, LC da Silva. Evaluation of morphological and metabolic responses to glyphosate exposure in two Neotropical plant species. Ecol Indic, 2020. [DOI]
- de Oliveira EP, Marchi KE, Emiliano J, Salazar SMCH, Ferri AH, Etto RM, et al. Changes in fatty acid composition as a response to glyphosate toxicity in Pseudomonas fluorescens. Heliyon. Volume 8. Elsevier Ltd; 2022. 8.
- O Devinsky, BJ Whalley, V Di Marzo. Cannabinoids in the treatment of neurological disorders. Neurotherapeutics, 2015. [DOI | PubMed]
- Durak R, Jedryczka M, Czajka B, Dampc J, Wielgusz K, Borowiak-Sobkowiak B. Mild abiotic stress affects development and stimulates hormesis of hemp aphid Phorodon cannabis. Insects MDPI AG; 2021;12(5).
- Eceiza MV, Gil-Monreal M, Barco-Antoñanzas M, Zabalza A, Royuela M. The moderate oxidative stress induced by glyphosate is not detected in Amaranthus palmeri plants overexpressing EPSPS. J plant physiol. Elsevier GmbH; 2022b. Jul 1;274.
- Eceiza MV, Gil-Monreal M, Barco-Antoñanzas M, Zabalza A, Royuela M. The moderate oxidative stress induced by glyphosate is not detected in Amaranthus palmeri plants overexpressing EPSPS. J plant physiol. Elsevier GmbH; 2022a Jul. 1;274.
- Elangovan A, Duc NT, Raju D, Kumar S, Singh B, Vishwakarma C et al. Imaging Sensor-Based High-Throughput measurement of biomass using machine learning models in rice. Agriculture (Switzerland). MDPI; 2023;13(4).
- V Filimonova, M De Troch, F Gonçalves, JC Marques, SM Marques, AMM Gonçalves. Effects of a herbicide and copper mixture on the quality of marine plankton. Ecotoxicol Environ Saf, 2018. [DOI | PubMed]
- Fuchs B, Saikkonen K, Helander M. Glyphosate-Modulated biosynthesis driving plant defense and species interactions. Trends plant sci. Elsevier Ltd; 2021. pp. 312–23.
- Gallien M, Occhiali G. Cannabis Taxation as a Revenue Source for Development: Opportunities and Challenges. 2023 Apr.
- Giupponi L, Leoni V, Carrer M, Ceciliani G, Sala S, Panseri S et al. Overview on Italian hemp production chain, related productive and commercial activities and legislative framework. Italian J Agron. 2020.
- MP Gomes, SG Le Manac’h, S Maccario, M Labrecque, M Lucotte, P Juneau. Differential effects of glyphosate and aminomethylphosphonic acid (AMPA) on photosynthesis and chlorophyll metabolism in Willow plants. Pestic Biochem Physiol, 2016. [DOI | PubMed]
- Gomes MP, Le Manac’h SG, Hénault-Ethier L, Labrecque M, Lucotte M, Juneau P. Glyphosate-dependent Inhibition of photosynthesis in Willow. Front Plant Sci. Frontiers Research Foundation; 2017. p. 8.
- Gorelick J, Bernstein N. Chemical and physical elicitation for enhanced cannabinoid production in cannabis. Cannabis sativa L. – Botany and biotechnology. Springer International Publishing; 2017. pp. 439–56.
- Gorelick J, Bernstein N, Elicitation. An underutilized tool in the development of medicinal plants as a source of therapeutic secondary metabolites. 2014. pp. 201–30.
- G Guedes, R Carvalhas, G Gomes. Hemp fiber: the textile material as a fashion value. Advances in fashion and design research II, 2024
- P Gulati, A Verma, A kumar, Kumar, P Solanki. Para-Cresyl sulfate and BSA conjugation for developing aptasensor: spectroscopic methods and molecular simulation. ECS J Solid State Sci Technol, 2023. [DOI]
- He M, Ding NZ. Plant unsaturated fatty acids: multiple roles in stress response. Front Plant Sci. Frontiers Media S.A.; 2020.
- RL Heath, L Packer. Photoperoxidation in isolated chloroplasts. Arch Biochem Biophys, 1968. [DOI | PubMed]
- S Hourfane, H Mechqoq, AY Bekkali, JM Rocha. El Aouad N. A comprehensive review on Cannabis sativa ethnobotany, phytochemistry, molecular Docking and biological activities. Plants, 2023. [DOI | PubMed]
- F Islam, MA Farooq, RA Gill, J Wang, C Yang, B Ali. 2,4-D attenuates salinity-induced toxicity by mediating anatomical changes, antioxidant capacity and cation transporters in the roots of rice cultivars. Sci Rep, 2017. [DOI | PubMed]
- Islam MJ, Ryu BR, Azad MOK, Rahman MH, Rana MS, Kang CW et al. Comparative growth, photosynthetic pigments, and osmolytes analysis of hemp (Cannabis sativa L.) seedlings under an aeroponics system with different LED light sources. Horticulturae MDPI AG; 2021;7(8).
- G Issaad, H Berrebbah, MR Djebar, R Rouabhi. Cellular investigation on the role of osmotic pressure in limiting the toxic effects of pesticidesin durum wheat (Triticum durum Desf). Ukr J Ecol, 2022
- KK Jaiswal, V Kumar, MS Vlaskin, M Nanda. Impact of glyphosate herbicide stress on metabolic growth and lipid inducement in Chlorella sorokiniana UUIND6 for biodiesel production. Algal Res, 2020. [DOI]
- Jalal A, de Oliveira Junior JC, Ribeiro JS, Fernandes GC, Mariano GG, Trindade VDR, et al. Hormesis in plants: physiological and biochemical responses. Ecotoxicol Environ Saf. Academic; 2021.
- Karche T, Singh MR. The application of hemp Cannabis sativa l. for a green economy: A review. Turk J Bot Turkiye Klinikleri; 2019. pp. 710–23.
- Karimmojeni H, Rezaei M, Tseng TM, Mastinu A. Effects of Metribuzin herbicide on some Morpho-Physiological characteristics of two Echinacea species. Horticulturae MDPI; 2022;8(2).
- N Kaur, A Kumar, T Malik, M Girdhar, M Singh, R Singh. Herbicide use and weed management strategies in hemp cultivation. J Cannabis Res, 2025. [DOI | PubMed]
- V Khaleghnezhad, AR Yousefi, A Tavakoli, B Farajmand, A Mastinu. Concentrations-dependent effect of exogenous abscisic acid on photosynthesis, growth and phenolic content of Dracocephalum moldavica L. under drought stress. Planta, 2021. [DOI | PubMed]
- S Kostopoulou, G Ntatsi, G Arapis, KA Aliferis. Assessment of the effects of metribuzin, glyphosate, and their mixtures on the metabolism of the model plant Lemna minor L. applying metabolomics. Chemosphere, 2020. [DOI | PubMed]
- A Krieger-Liszkay. Singlet oxygen production in photosynthesis. J Exp Bot, 2004. [DOI | PubMed]
- J Kruk, R Szymańska. Singlet oxygen oxidation products of carotenoids, fatty acids and phenolic prenyllipids. J Photochem Photobiol B, 2021. [DOI | PubMed]
- V Kumar, A Khan, A Srivastava, G Saxena. Toxicity assessment of Metribuzin and its amelioration through plant growth regulators in Vigna radiata (L.) R. Wilczek. Environ Sci Pollut Res, 2022. [DOI]
- V Kumar, A Khan, A Srivastava, G Saxena. Toxicity assessment of Metribuzin and its amelioration through plant growth regulators in Vigna radiata (L.) R. Wilczek. Environmental science and pollution research. Springer Sci Bus Media Deutschland GmbH, 2023
- Legare CA, Raup-Konsavage WM, Vrana KE. Therapeutic potential of cannabis, cannabidiol, and Cannabinoid-Based pharmaceuticals. Pharmacology. S. Karger AG; 2022. pp. 131–49.
- Liu S, Rao J, Zhu J, Li G, Li F, Zhang H et al. Integrated physiological, metabolite and proteomic analysis reveal the glyphosate stress response mechanism in tea plant (Camellia sinensis). J Hazard Mater [Internet]. 2023;454:131419. Available from: https://www.sciencedirect.com/science/article/pii/S0304389423007021
- H Lowe, B Steele, J Bryant, N Toyang, W Ngwa. Non-Cannabinoid metabolites of Cannabis sativa L. with therapeutic potential. Plants, 2021. [DOI | PubMed]
- OH Lowry, NJ Rosebrough, AL Farr, RJ Randall. Protein measurement with the Folin phenol reagent. J Biol Chem, 1951. [DOI | PubMed]
- J Lydon, AH Teramura, CB Coffman. UV-B radiation effects on photosynthesis, growth and cannabinoid production of two Cannabis sativa chemotypes. Photochem Photobiol, 1987. [DOI | PubMed]
- Macherone A. Comprehensive analytical testing of cannabis and hemp. Comprehensive analytical chemistry. Elsevier; 2020. pp. 3–29.
- Maclachlan S, Zalik S, Plastid, Structure. Chlorophyll concentration, and free amino acid composition of a chlorophyll mutant of barley. Can J Bot. 1963;41(7):1053–62.
- AL Mancinelli. Photoregulation of anthocyanin synthesis. Plant Physiol, 1984. [DOI | PubMed]
- H Mansouri, Z Asrar. Effects of abscisic acid on content and biosynthesis of terpenoids in Cannabis sativa at vegetative stage. Biol Plant, 2012. [DOI]
- C Maqueda, P Partal, J Villaverde, JL Perez-Rodriguez. Characterization of sepiolite-gel-based formulations for controlled release of pesticides. Appl Clay Sci, 2009. [DOI]
- KE McNaughton, PH Sikkema, DE Robinson. Response of processing tomato to simulated glyphosate drift followed by In-Crop Metribuzin application. Weed Technol, 2012. [DOI]
- MM Nemat Alla, AM Badawi, NM Hassan, ZM El-Bastawisy, EG Badran. Effect of metribuzin, Butachlor and chlorimuron-ethyl on amino acid and protein formation in wheat and maize seedlings. Pestic Biochem Physiol, 2008. [DOI]
- T-N Nguyen, S Son, MC Jordan, DB Levin, BT Ayele. Lignin biosynthesis in wheat (Triticum aestivum L.): its response to waterlogging and association with hormonal levels. BMC Plant Biol, 2016. [DOI | PubMed]
- J Pande, S Chanda. Determination of phytochemical profile and antioxidant efficacy of lavendula bipinnata leaves collected during Magha Nakshatra days and normal days using LC-QTOF-MS technique. J Pharm Biomed Anal, 2020. [DOI | PubMed]
- H Peng, F Shahidi. Cannabis and Cannabis edibles: A review. J Agric Food Chem, 2021. [DOI | PubMed]
- WA Pline, R Wells, G Little, KL Edmisten, JW Wilcut. Glyphosate and Water-Stress effects on fruiting and carbohydrates in Glyphosate‐Resistant cotton. Crop Sci, 2003. [DOI]
- F Pollastro, A Minassi, LG Fresu. Cannabis phenolics and their bioactivities. Curr Med Chem, 2018. [DOI | PubMed]
- Pütter J. Peroxidases. Methods of enzymatic analysis. Elsevier; 1974. pp. 685–90.
- DEM Radwan, KA Fayez. Photosynthesis, antioxidant status and gas-exchange are altered by glyphosate application in peanut leaves. Photosynthetica, 2016. [DOI]
- A Raihan, TR Bijoy. A review of the industrial use and global sustainability of Cannabis sativa. Global Sustain Res, 2023. [DOI]
- Razieh Rajabi. Effects of stress induced by post-emergence application of Metribuzin herbicide on wheat. Afr J Biotechnol Acad Journals; 2012;11(16).
- M Rehman, S Fahad, G Du, X Cheng, Y Yang, K Tang. Evaluation of hemp (Cannabis sativa L.) as an industrial crop: a review. Environ Sci Pollut Res, 2021. [DOI]
- S Réquilé, B Mazian, M Grégoire, S Musio, M Gautreau, L Nuez. Exploring the dew Retting feasibility of hemp in very contrasting European environments: influence on the tensile mechanical properties of fibres and composites. Ind Crops Prod, 2021. [DOI]
- P Rodziewicz, S Loroch, Ł Marczak, A Sickmann, O Kayser. Cannabinoid synthases and osmoprotective metabolites accumulate in the exudates of Cannabis sativa L. glandular trichomes. Plant Sci, 2019. [DOI | PubMed]
- M Roncel, I Yruela, D Kirilovsky, F Guerrero, M Alfonso, R Picorel. Changes in photosynthetic electron transfer and state transitions in an herbicide-resistant D1 mutant from soybean cell cultures. Biochim Et Biophys Acta (BBA) – Bioenergetics, 2007. [DOI | PubMed]
- J Salman, E Abdul-Adel, AF AlKaim. Effect of pesticide glyphosate on some biochemical features in cyanophyta algae Oscillatoria limnetica. Int J Pharmtech Res, 2016
- Saloner A, Bernstein N. Nitrogen source matters: high NH4/NO3 ratio reduces cannabinoids, terpenoids, and yield in medical Cannabis. Front Plant Sci. Frontiers Media S.A.; 2022b. Jun 1;13.
- Saloner A, Bernstein N. Effect of potassium (K) supply on cannabinoids, terpenoids and plant function in medical Cannabis. Agronomy. MDPI; 2022a May. 1;12(5).
- Shiponi S, Bernstein N. The highs and lows of P supply in medical cannabis: effects on cannabinoids, the ionome, and Morpho-Physiology. Front Plant Sci. Frontiers Media S.A.; 2021a. Jul 15;12.
- Shiponi S, Bernstein N. The highs and lows of P supply in medical cannabis: effects on cannabinoids, the ionome, and Morpho-Physiology. Front Plant Sci. 2021b Jul 15;12.
- Y Shoyama, T Tamada, K Kurihara, A Takeuchi, F Taura, S Arai. Structure and function of ∆1-Tetrahydrocannabinolic acid (THCA) synthase, the enzyme controlling the psychoactivity of Cannabis sativa. J Mol Biol, 2012. [DOI | PubMed]
- DC Simiyu, JH Jang, OR Lee. Understanding Cannabis sativa L.: current status of propagation, use, legalization, and Haploid-Inducer-Mediated genetic engineering. Plants, 2022. [DOI | PubMed]
- Singh S, Kumar V, Datta S, Wani AB, Dhanjal DS, Romero R et al. Glyphosate uptake, translocation, resistance emergence in crops, analytical monitoring, toxicity and degradation: a review. Environ Chem Lett. Springer; 2020. pp. 663–702.
- C Song, A Saloner, A Fait, N Bernstein. Nitrogen deficiency stimulates cannabinoid biosynthesis in medical cannabis plants by inducing a metabolic shift towards production of low-N metabolites. Ind Crops Prod, 2023. [DOI]
- M Tayyab, D Shahwar. GCMS analysis of Cannabis sativa L. from four different areas of Pakistan. Egypt J Forensic Sci Egypt Forensic Med Auth, 2015
- Toth JA, Smart LB, Smart CD, Stack GM, Carlson CH, Philippe G, et al. Limited effect of environmental stress on cannabinoid profiles in high-cannabidiol hemp (Cannabis sativa L). Volume 13. GCB Bioenergy. John Wiley and Sons Inc; 2021. pp. 1666–74. 10.
- A Vadhel, A Kumar, S Bashir, T Malik, A Mohan. Synergistic and non-synergistic impact of HAP-based nano fertilizer and PGPR for improved nutrient utilization and metabolite variation in hemp crops. Environ Sci Nano, 2023. [DOI]
- E van Handel. Direct microdetermination of sucrose. Anal Biochem, 1968. [DOI | PubMed]
- Verma AK, Mishra A, Dhiman TK, Sardar M, Solanki PR. Experimental and In Silico interaction studies of Alpha Amylase-Silver nanoparticle: a nano-bio-conjugate. bioRxiv [Internet]. 2022;2022.06.11.495728. Available from: http://biorxiv.org/content/early/2022/06/13/2022.06.11.495728.abstract
- Verma AK, Gulati P, Lakshmi G, Solanki PR, Kumar A. Interaction studies of Gut metabolite; Trimethylene amine Oxide with Bovine Serum Albumin through Spectroscopic, DFT and Molecular Docking Approach. bioRxiv [Internet]. 2023a Jan 1;2023.04.06.535846. Available from: http://biorxiv.org/content/early/2023/04/06/2023.04.06.535846.abstract.
- Verma AK, Sharma S, Jayaraj A, Deep S. In Silico study of interaction of (ZnO)12 nanocluster to glucose oxidase-FAD in absence and presence of glucose. J Biomol Struct Dyn 2023b Dec 29;41(24):15234–42.
- RG Vital, A Jakelaitis, FB Silva, PF Batista, GM Almeida, AC Costa. Physiological changes and in the carbohydrate content of sunflower plants submitted to sub-doses of glyphosate and trinexapac-ethyl. Bragantia, 2017. [DOI]
- T Volova, S Baranovsky, O Petrovskaya, A Shumilova, A Sukovatyi. Biological effects of the free and embedded Metribuzin and tribenuron-methyl herbicides on various cultivated weed species. J Environ Sci Health Part B, 2020. [DOI]
- Wishart DS, Hiebert-Giesbrecht M, Inchehborouni G, Cao X, Guo AC, LeVatte MA et al. Chemical composition of commercial Cannabis. J Agric Food Chem. 2024.
- R Yokoyama, MVV de Oliveira, B Kleven, HA Maeda. The entry reaction of the plant Shikimate pathway is subjected to highly complex metabolite-mediated regulation. Plant Cell, 2021. [DOI | PubMed]
- Zhao F, Wang P, Lucardi RD, Su Z, Li S. Natural sources and bioactivities of 2,4-di-tert-butylphenol and its analogs. Toxins (Basel). MDPI AG; 2020.
