Diagnosis and treatment of autonomic failure, pain and sleep disturbances in Parkinson’s disease: guideline “Parkinson’s disease” of the German Society of Neurology
Abstract
Background and objective:
Non-motor symptoms frequently develop throughout the disease course of Parkinson’s disease (PD), and pose affected individuals at risk of complications, more rapid disease progression and poorer quality of life. Addressing such symptom burden, the 2023 revised “Parkinson’s disease” guideline of the German Society of Neurology aimed at providing evidence-based recommendations for managing PD non-motor symptoms, including autonomic failure, pain and sleep disturbances.
Methods:
Key PICO (Patient, Intervention, Comparison, Outcome) questions were formulated by the steering committee and refined by the assigned authors. Recommendations were drafted based on relevant studies, systematic reviews, meta-analyses and high-quality guidelines identified by the literature search. They were subsequently reviewed, revised, and voted by the Guideline Group in online consensus conferences. Consensus was achieved in case of > 75% agreement among the group members. The consensus was considered strong, if agreement was > 95%.
Results:
The guideline entails: (i) 10 PICOs and 23 recommendations on the diagnosis and treatment of urogenital, cardiovascular and gastrointestinal autonomic failure; (ii) four PICOs and four recommendations on the possible types of pain in PD individuals, their diagnosis and treatment; (iii) 11 PICOs and 11 recommendations on the screening, diagnosis and treatment of sleep disturbances and excessive daytime sleepiness in PD individuals, as well as on their prognostic implications. Thirty-one out of 38 recommendations achieved a strong consensus.
Conclusion:
The current German PD guideline provides a practice-oriented and etiology-driven stepwise approach to the diagnosis and treatment of autonomic failure, pain and sleep disturbances in PD individuals.
Article type: Review Article
Keywords: Parkinson’s disease, Orthostatic hypotension, Supine hypertension, Urinary incontinence, Nocturia, Erectile dysfunction, Constipation, Pain, Sleep, Daytime sleepiness, REM-sleep behavior disorder
Affiliations: https://ror.org/03pt86f80grid.5361.10000 0000 8853 2677Department of Neurology, Innsbruck Medical University, Anichstrasse 35, 6020 Innsbruck, Austria; https://ror.org/0270sxy44grid.440220.0Paracelsus-Elena-Klinik, Kassel, Germany; https://ror.org/01rdrb571grid.10253.350000 0004 1936 9756Department of Neurology, Philipps-Universität, Marburg, Germany; https://ror.org/03wjwyj98grid.480123.c0000 0004 0553 3068Department of Neurology, University Clinic Eppendorf, Hamburg, Germany; https://ror.org/03zdwsf69grid.10493.3f0000 0001 2185 8338Department of Neurology, Rostock University Medical Center, Rostock, Germany; Deutsches Zentrum für Neurodegenerative Erkrankungen (DZNE) Rostock/Greifswald, Rostock, Germany; Department of Neurology, Lindenbrunn Hospital, Coppenbrügge, Germany; Kathol. Kliniken der Ruhrhalbinsel, Essen, Germany; https://ror.org/055w00q26grid.492054.eParkinson-Klinik Ortenau, Wolfach, Germany; https://ror.org/021ft0n22grid.411984.10000 0001 0482 5331Department of Neurosurgery, University Medical Center Göttingen, Göttingen, Germany; https://ror.org/05591te55grid.5252.00000 0004 1936 973XDepartment of Neurology, LMU University Hospital, Ludwig-Maximilians-Universität (LMU) München, Munich, Germany; https://ror.org/043j0f473grid.424247.30000 0004 0438 0426Deutsches Zentrum für Neurodegenerative Erkrankungen (DZNE), Munich, Germany; https://ror.org/025z3z560grid.452617.3Munich Cluster for Systems Neurology (SyNergy), Munich, Germany
License: © The Author(s) 2024 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s00415-024-12730-5 | PubMed: 39751950 | PMC: PMC11698777
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.4 MB)
Introduction
Non-motor symptoms, including REM-sleep behavior disorders (RBD), insomnia, autonomic failure, and sometimes pain, may precede the development of Parkinson’s disease (PD) motor symptoms by years and their recognition often contributes to an early identification of affected PD individuals [ref. 19, ref. 67]. The frequency and severity of PD non-motor symptoms however typically increases with disease duration, and has been associated with morbidity, mortality, more rapid disease progression and overall poorer quality of life of PD individuals and their caregivers [ref. 15].
Lower urinary tract symptoms are a common non-motor symptom in individuals with PD and atypical parkinsonian disorders and are divided into disturbances of the storage phase, mostly due to bladder detrusor hyperactivity, and disturbances of the voiding phase due to detrusor-sphincter dyssynergia or hypocontractile detrusor [ref. 1, ref. 88, ref. 167]. Neurogenic bladder dysfunction refers to any disturbance of bladder function due to impaired neural control and needs to be distinguished from non-neurogenic bladder disorders, especially in elderly individuals.
Male erectile dysfunction is defined as the inability to achieve and maintain a penile erection sufficient for a satisfactory sexual intercourse [ref. 44]. Erectile dysfunction has a major impact on the well-being of affected individuals and may be accompanied by decreased libido and ejaculation difficulties. The incidence of erectile dysfunction increases exponentially with age [ref. 26] due to concomitant vascular and other contributing factors. Up to 80% of male PD individuals have been indeed found to suffer from erectile dysfunction [ref. 191], highlighting a major, though often unspoken, therapeutic need in these individuals.
Neurogenic orthostatic hypotension (OH) is another non-motor feature affecting one-third of PD individuals [ref. 166, ref. 241]. In most cases, OH manifests with non-specific symptoms and complaints, such as orthostatic dizziness, light-headedness or blurred vision, but it may cause orthostatic syncope, increasing risk of injurious falls and their complications, often leading to hospitalization. Neurogenic OH in PD develops after a degeneration of the post-ganglionic sympathetic neurons constituting the efferent branch of the baroreflex arch, but concomitant non-neurogenic factors including infections, dehydration and cardiac diseases, may unmask or worsen OH, especially in elderly individuals [ref. 112]. Parkinsonian medications and other frequently used CNS-active drugs (i.e. hypnotics, antidepressants, neuroleptics, opioids) may also aggravate orthostatic intolerance [ref. 187]. Due to progressive baroreflex dysfunction and other, to date not fully understood, additional mechanisms, about 50% of individuals with neurogenic OH paradoxically develop hypertensive blood pressure (BP) levels in the supine position [ref. 59, ref. 66] that can be severe and last for several hours during sleep (nocturnal hypertension). Drugs to treat OH can unmask or aggravate supine hypertension, but this often develops independently of any anti-hypotensive treatment [ref. 62]. Possible complications of supine and nocturnal hypertension include hypertensive emergencies, chronic end-organ damage and increased pressure natriuresis overnight, this in turn aggravating nocturia and early morning OH symptoms—a phenomenon that often remains unrecognized by affected individuals and their caregivers.
Constipation is the most frequent autonomic feature affecting PD individuals and is determined by both neurogenic and non-neurogenic factors, such as reduced mobility and abdominal muscle tone, a diet poor in fibers and insufficient fluid intake, all contributing to a prolonged gastrointestinal transit time and sometimes megacolon [ref. 251]. Antiparkinsonian medications, especially anticholinergics [ref. 102, ref. 116], as well as several other frequently prescribed medications, such as opioids, tricyclic antidepressants, calcium-containing antacids, antihypertensives, spasmolytics, sympathomimetics, and diuretics, may also worsen constipation. Anismus is defined by the lack of relaxation or involuntary contraction of the anal sphincter during defecation and may rarely cause an ‘outlet constipation’ in PD individuals [ref. 250].
Pain is another common non-motor symptom in PD. Different pain phenotypes have been described in PD individuals with complex and often multifactorial etiologies. Some painful conditions, such as arthrosis of the spine or joints, may be aggravated by PD-related motor impairment and may further limit the mobility of affected individuals in everyday life. A bidirectional influence in fact exists between PD-related and PD-unrelated pain determinants [ref. 29], ultimately preventing an exact etiological pain classification.
Sleep disturbances develop in up to 90% of PD individuals, significantly impacting on their quality of life [ref. 15, ref. 17, ref. 118, ref. 196, ref. 264], caregivers’ burden [ref. 25, ref. 202, ref. 220, ref. 266] and disease progression, especially in case of RBD [ref. 157, ref. 176]. Daytime sleepiness, due to sleep impairment, medications or as a separate entity caused by PD itself, further restricts social interaction, participation, mobility, professional and leisure activities of PD individuals.
Acknowledging the symptom burden and health hazard derived by PD non-motor symptoms, the 2023 edition of the “Parkinson’s disease” guideline of the German Society of Neurology (Deutsche Gesellschaft für Neurologie, DGN) for the first time entailed recommendations for the diagnosis and treatment of non-motor PD symptoms, including autonomic failure, pain and sleep disturbances.
This article is an abridged translation of the original guideline in German language and focuses on the diagnosis and treatment of autonomic failure, pain and sleep in individuals with PD. The full German version of the guideline is available under www.dgn.org/leitlinien and the AWMF (Arbeitsgemeinschaft wissenschaftlicher Medizinischer Gesellschaften, AWMF version number: 8.0. Level of guideline: S2k. Date of last update: 10/2023. Valid until: 24.10.2028. Edited by: Guidelines Committee of the German Society of Neurology) [ref. 93].
Guideline methodology
During the purposing phase, key PICO (Patient, Intervention, Comparison, Outcome) questions were formulated by the guideline steering committee. The respective chapter authors subsequently refined the PICO wording and performed a comprehensive literature search, identifying pertinent studies, systematic reviews, meta-analyses and high-quality guidelines. The identified literature was further enriched with additional evidence sources provided by the assigned authors. The chapter authors subsequently drafted the background and recommendation texts, which were reviewed, and put to an online vote by all members of the German Parkinson Guideline Group. Recommendations that achieved < 75% consensus required group discussion, revision and re-voting. Recommendations with > 75% agreement among the group members were considered consented. Consensus was “strong", if agreement outreached a 95% quote.
For the purposes of the present work, the dosage ranges of the recommended medications have been added based on the available manufacturer’s information and clinical experience of the authors. Studies assessing the efficacy and safety of different drug schedules in the specific setting of PD were, however, oftentimes missing. Treating physicians are therefore requested to consult the applicable prescribing information in their respective country and to perform a case-by-case risk–benefit assessment when considering prescribing any of the therapeutic options mentioned hereafter.
The most important recommendations
Bladder disturbances
- Non-pharmacological measures to treat neurogenic bladder dysfunction in PD include bladder and pelvic floor training, adjusting fluid intake according to the time of day and avoiding caffeine, alcohol and carbonated drinks.
- Antimuscarinics (preferably solifenacin 5 mg o.i.d, trospium 15–30 mg b.i.d or darifenacin 7.5–15 mg o.i.d., due to their lower risk of cognitive side effects) should be considered for the pharmacological treatment of urinary urge incontinence due to bladder detrusor overactivity in PD.
- β3 adrenoceptor agonists (e.g. mirabegron 50 mg o.i.d.) can be considered for treating urinary urge due to bladder detrusor overactivity in PD individuals, who have responded inadequately to antimuscarinics, have not tolerated them or in whom antimuscarinics are contraindicated.
- In patients who have responded inadequately to oral therapy, botulinum toxin A intravesical injection (200 U or customized) may be considered for treating severe urinary urge incontinence, if the individual motor and cognitive performance enables the subsequently likely necessary intermittent catheterization.
- For the non-pharmacological treatment of nocturia, affected individuals should be advised to limit their fluid intake in the second half of the day, avoid alcohol consumption in the evening and sleep in a 10°–20° head-up tilted position.
- For the treatment of nocturnal polyuria, an evening desmopressin administration (5–40 µg o.i.d. nasal spray or 100–800 µg o.i.d. per os) can be considered under close monitoring of blood pressure, serum electrolytes and body weight.
- To treat nocturia due to reduced bladder capacity, treatment with antimuscarinics (preferably solifenacin, trospium or darifenacin due to their lower risk of cognitive side effects) should be considered instead.
Orthostatic hypotension
The following stepwise approach should be applied for treating OH in PD:
- Eliminate or treat aggravating factors (e.g., infectious diseases, dehydration).
- Review concomitant medications (if antihypertensives are being used, a dose reduction or discontinuation should be sought).
- Apply non-pharmacological strategies.
- Start anti-hypotensive medications.
Constipation
- For treating constipation in individuals with PD, the DGN recommends following the AWMF guideline on ‘Chronic Constipation’.
Pain
- The first therapeutic step is an optimization of antiparkinsonian medications.
- Nociceptive pain should be treated according to the 3-step WHO regimen.
- Neuropathic pain should be treated with anticonvulsants and/or antidepressants as indicated in the guidelines for the treatment of neuropathic pain, with preference given to gabapentin 300–1800 mg (rarely up to 3600 mg), especially in case of concomitant restless legs syndrome, and/or duloxetine 60–120 mg, in case of concomitant depression. Gabapentin is preferred over pregabalin due to its proven tolerability in PD and potentially positive influence on motor symptoms [ref. 163, ref. 238].
- For severe pain, treatment with prolonged-release oxycodone/naloxone 5/2.5–20/10 mg (rarely up to 40/20 mg) may be considered.
- In the experience of the authors of the present publication, local botulinum toxin application might be considered in single individuals with pain refractory to maximized oral or transdermal treatment, especially in case of on- and off dystonia, muscular pain trigger points, painful myotonus, spasms in the esophagus, pylorus, anal sphincter or hyperactive bladder [ref. 29]. In such cases, the botulinum toxin application should be endoscopically guided.
Sleep
- Primary sleep disorders such as restless leg syndrome or sleep disordered breathing should be treated according to the corresponding guidelines.
- If motor or non-motor fluctuations determine sleep disturbances, the dopaminergic pharmacotherapy should be adjusted accordingly.
- RBD should be firstly treated by creating a safe sleep environment. Clonazepam (0.125–3 mg) and/or melatonin (2–9 mg) can be considered, bearing in mind possible side effects.
- In case of insomnia or circadian rhythm disorders, underlying causes such as medication side effects and/or primary sleep disorders should be ruled out. If treatable causes have been ruled out, circadian rhythm disorders should be treated with sleep hygiene measures, intensive physical training and light therapy. Starting with the lowest possible dosage, eszopiclone (1 mg), doxepin (25 mg), zolpidem (5 mg), trazodone (50 mg), melatonin (2 mg), venlafaxine (37.5 mg, in case of comorbid depression), nortriptyline (25 mg) or mirtazapine (7.5 mg) can be used for treating insomnia (low level of evidence). Further dose adjustments may be required on an individual basis. Side effects should be carefully monitored at follow-up.
The guidelines in detail
What kind of bladder dysfunction can occur in individuals with PD?
Neurogenic bladder dysfunction is found (depending on the stage of the disease) in around 60% of all individuals with PD [ref. 122]. The bladder filling or storage phase is most frequently affected, leading to urinary frequency, nocturia, urinary urge and urge incontinence. Such symptoms impact significantly on the quality of life of affected individuals and often require dedicated, multidisciplinary management. Non-neurogenic bladder disorders (e.g. prostate hyperplasia in men or pelvic floor prolapse in women) must be carefully differentiated from neurogenic bladder disorders in close collaboration with allied specialties such as urology and gynecology. In addition to urological/gynecological consultations, urinalysis is also essential to rule out reversible causes of bladder disturbances such as urinary tract infections, and to screen for proteinuria, diabetes mellitus or space-occupying lesions, in case of persistent hematuria. Keeping a bladder diary for 48–72 h helps to quantify the micturition frequency, urine volume, incontinence episodes, as well as to identify nocturnal polyuria [ref. 117], ultimately enabling an accurate estimation of the individual symptomatic burden. Additional examinations include the measurement of post-void residual urine volume by means of bladder ultrasound or in-and-out catheterization, uroflowmetry, and urodynamics. The latter is considered the gold standard in the diagnostic workup of neurogenic bladder disorders.
| Recommendation | |
|---|---|
| Individuals with PD frequently develop disturbances of the bladder storage phase, including nocturia, urinary frequency, urgency and/or urge incontinence | |
| Level of consensus: 100%, strong consensus |
How to treat bladder dysfunction in PD?
The treatment of bladder dysfunction aims to improve the quality of life of affected individuals by reducing incontinence episodes and to prevent secondary upper urinary tract damage by improving micturition control [ref. 167, ref. 193]. Non-pharmacological and behavioral measures such as bladder training, adjusting the amount of fluid intake, avoiding caffeine, alcohol and carbonated drinks can alleviate symptoms associated with neurogenic bladder dysfunction [ref. 45, ref. 223, ref. 249]. To avoid social stigmatization due to bladder incontinence, appliances (e.g. condom urinals, pads) can be of help [ref. 88]. Antimuscarinics, including oxybutinin, tolterodine, solifenacin, darifenacin and trospium, are the first pharmacological option for treating urinary urgency and urge incontinence due to bladder detrusor overactivity. Possible side effects of antimuscarinics include: increased post-void residual urine volume, xerostomia, constipation and blurred vision [ref. 255]. Delirium, confusion and worsening of pre-existing cognitive impairment are further side effects due to the central anticholinergic effect and limit the use of anticholinergics in PD individuals with dementia. Trospium (15–30 mg b.i.d.), which does not cross the blood–brain barrier, as well as solifenacin (5 mg o.i.d), and darifenacin (7.5–15 mg o.i.d.), that selectively stimulate peripheral M3 muscarinic receptors, are less likely to cause cognitive side effects and may be therefore preferred [ref. 114, ref. 232]. The post-void residual urine volume should be monitored during the titration phase and after dose changes of antimuscarinics, as this may increase due to drug-induced bladder detrusor weakness [ref. 4].
Mirabegron, a β3 adrenoceptor agonist administered 50 mg o.i.d., was found to reduce urinary urge incontinence due to detrusor overactivity in different neurological disorders, including PD [ref. 43, ref. 150], with an acceptable safety profile [ref. 42]. Compared to antimuscarinic drugs, mirabegron was less frequently associated with urinary retention, constipation, cognitive impairment and dry mouth and may be therefore considered as an alternative for patients in whom antimuscarinics are contraindicated, not tolerated [ref. 81, ref. 115, ref. 223] or did not sufficiently improve incontinence symptoms [ref. 55].
In refractory cases, more invasive options may also be considered. Intravesical botulinum toxin A injection (200 U, or customized) reduces incontinence episodes due to detrusor overactivity [ref. 83], but impairs voluntary voiding and leads to urinary retention. Such procedure is therefore only indicated in patients with sufficient hand-dexterity to perform clean intermittent self-catheterizations and not in patients with severe cognitive or motor disorders, or without available caregiver [ref. 47, ref. 200]. Percutaneous [ref. 33] and transcutaneous [ref. 7, ref. 171] tibial nerve stimulation can also improve symptoms of overactive bladder in PD.
In patients reporting frequent nocturnal toilet visits, a bladder diary should be used to differentiate between nocturnal polyuria (i.e. more than 1/3 of the 24-h total urine volume is excreted overnight, e.g. in patients with nocturnal hypertension), a generally increased urine output (> 40 ml/kg body weight, e.g. in uncontrolled diabetes) and a reduced bladder capacity (e.g. in overactive bladder). A combination of more than one cause is common and additional factors that may contribute to nocturia should also be considered as part of the initial assessment. These include nocturnal hypertension, which is found in approximately 50% of PD individuals with OH, evening use of diuretics, sleep apnoea, psychogenic polydipsia and insufficient secretion of antidiuretic hormone (either idiopathic, drug-induced or due to pituitary lesions).
In case of nocturia, patients should be advised to limit their fluid intake in the late afternoon/evening, avoid any evening alcohol consumption and sleep in a 10°–20° head-up tilted position [ref. 205]. Bedtime desmopressin (5–40 µg o.i.d. nasal spray or 100–800 µg o.i.d. per os) can be considered in patients with nocturnal polyuria under close BP, serum electrolytes and body weight monitoring [ref. 192]. Bedtime antimuscarinics (i.e., trospium 15–30 mg) can be considered in patients with nocturia due to reduced bladder capacity.
| Recommendation | |
|---|---|
| Non-pharmacological measures for treating neurogenic bladder dysfunction in PD include bladder training, adjusting fluid intake to the daily routine, as well as avoiding caffeine, alcohol and carbonated drinks | |
| To avoid social stigmatisation due to bladder incontinence, continence appliances should be recommended | |
| Antimuscarinics (preferably solifenacin, trospium or darifenacin, due to their lower risk of cognitive side effects) should be considered for the pharmacological treatment of urinary urge incontinence due to bladder detrusor overactivity in PD | |
| β3 adrenoceptor agonists (e.g. mirabegron) can be considered for the treatment of urinary urge and urge incontinence due to bladder detrusor overactivity in those PD individuals who have responded inadequately or have not tolerated antimuscarinics, or for whom antimuscarinics are contraindicated | |
| Intravesical injection of botulinum toxin A can be considered for the treatment of detrusor overactivity in individuals, who have responded inadequately to oral therapy, provided that the patient’s motor and cognitive performance enables intermittent self-catheterizations that are likely required after the procedure | |
| PD individuals with nocturia should be advised to limit fluid intake in the late afternoon/evening, avoid alcohol consumption in the evening, and sleep in a 10°–20° head-up tilted position | |
| To treat nocturnal polyuria, bedtime desmopressin can be considered under close BP, serum electrolytes and body weight monitoring | |
| To treat nocturia due to reduced bladder capacity, bedtime antimuscarinics should be considered | |
| Level of consensus: 100%, strong consensus |
How to diagnose erectile dysfunction in PD?
The diagnosis of erectile dysfunction is primarily in the hands of urologists. A detailed medical history is essential to detect modifiable causes and influencing factors (e.g. concomitant SSRIs or β-blockers treatment). A neurological and urological examination should be performed, and signs of peripheral arterial disease assessed. A laboratory examination should include cardiovascular risk factors (fasting glucose, HbA1c, lipid status), common causes of polyneuropathy (renal and liver function parameters, serum protein electrophoresis, vitamin B1, B6, B12 status) and, in patients with gynaecomastia, testosterone levels. If a vascular origin is suspected, a provocation test with intracavernous application of alprostadil (± vascular sonography) may be necessary. Additional electrophysiological examinations (e.g. pudendal nerve SSEP) can help to confirm a neurogenic cause of erectile dysfunction [ref. 12, ref. 87].
| Recommendation | |
|---|---|
| A multidisciplinary approach with colleagues from the urology service is necessary in PD individuals complaining of erectile dysfunction. The basic diagnostic work-up includes a detailed medical history, clinical neurological and urological examination, as well as laboratory testing. Additional electrophysiological examinations may be necessary in single cases | |
| Level of consensus: 100%, strong consensus |
How to treat male erectile dysfunction in PD?
The efficacy of oral phophodiesterase 5 (PDE-5) inhibitors (sildenafil, tadalafil, vardenafil), and in particular of sildenafil (sildenafil 50–100 mg on demand), for treating erectile dysfunction in male individuals PD was sufficiently proven [ref. 127, ref. 204] [ref. 20, ref. 96, ref. 180, ref. 267]. Indirect comparisons between different PDE-5 inhibitors also suggest that they are equally effective [ref. 148]. The occurrence of acute cardiovascular events during intercourse and worsening of OH must however be considered. For this reason, PDE-5 inhibitors with long half-life (tadalafil) should be used with caution in patients with proven OH, as there is a risk of long-lasting arterial hypotension. Intraurethral or intracavernous application of vasodilatory prostaglandins (alprostadil 10–20 µg intra-cavernous injection on demand) may be considered, although there have been no studies in PD individuals and there is an increased risk of priapism and bleeding [ref. 12, ref. 149]. Case series and a multicentre double-blind study suggest a positive therapeutic effect of apomorphine (sublingual or subcutaneous) on erectile dysfunction [ref. 54, ref. 160]. Penile prosthesis can be considered in single cases [ref. 12].
| Recommendation | |
|---|---|
| Treatment with PDE-5 inhibitors should be considered for the pharmacological treatment of erectile dysfunction in individuals with PD with a low cardiovascular risk during sexual intercourse. A possible unmasking/aggravation of OH should be considered | |
| Level of consensus: 100%, strong consensus |
How to diagnose OH in PD?
OH is defined by a fall in systolic BP ≥ 20 mmHg or diastolic ≥ 10 mmHg within 3 min of head-up tilt test or active standing [ref. 77]. Sustained systolic BP values < 90 mmHg upon standing are also considered suggestive of OH [ref. 27]. In the expert consensus, two methods are approved for OH detection and should be considered for its diagnosis in PD: the active standing test (also known as Schellong test, with BP and heart rate measurements after at least 5 min in the supine position and at least after 1 and 3 min after changing to the upright position [ref. 58]) and the head-up tilt test examination (with tilting by at least 60°) [ref. 229]. Neurogenic OH is characterized by a very modest or completely abolished heart rate increase despite severe orthostatic BP fall, indicating baroreflex dysfunction [ref. 112]. This is identified by a neurogenic OH ratio < 0.5, which is calculated by dividing the supine-to-upright change in heart rate per the supine-to-upright fall in systolic BP [ref. 63, ref. 158].
| Recommendation | |
|---|---|
| In individuals with PD, a Schellong test should be performed to diagnose OH. A tilt table examination can be considered if available, as it provides more comprehensive and precise information | |
| Level of consensus: 100%, strong consensus |
How to treat OH in PD?
OH in PD should be treated with a stepwise approach [ref. 27, ref. 57, ref. 254]. First, exacerbating factors such as concomitant infections, anemia, dehydration, and the combination of multiple drugs with BP lowering effect should be excluded or treated.
In a second step, non-pharmacological therapeutic measures should be considered. Rapid drinking of cold water has a significant, though transient, pressor effect lasting about 1 h [ref. 101, ref. 140, ref. 203, ref. 206, ref. 263]. Studies showed that salt supplements had no additive effect with respect to increased water intake [ref. 125, ref. 181, ref. 265], but no dietary salt restriction should be recommended either. Large meals can produce significant falls in BP (e.g. post-prandial hypotension) [ref. 179], and it is therefore advisable to divide the daily caloric intake into several smaller meals. Sleeping in a 10°–20° head up tilted position can also improve orthostatic tolerance the day after [ref. 228, ref. 239]. A steeper head-up tilt sleeping position is likely more effective and can be pursued, if well tolerated. Physical counterpressure maneuvers (squatting or crossing the legs and tensing the leg, buttock, abdominal and arm muscles) can be recommended to stabilize the BP in case of dizziness [ref. 212, ref. 227, ref. 236, ref. 240]. Wearing an elastic abdominal binder is an additional non-pharmacological option to increase orthostatic BP levels, and more effective than compression stockings [ref. 61, ref. 71, ref. 156, ref. 169, ref. 212, ref. 236].
Midodrine (2.5–10 mg b.i.d./t.i.d) is a vascular adrenoceptor agonist and its efficacy to treat OH has been demonstrated in several randomized control trials [ref. 75, ref. 98, ref. 110, ref. 129, ref. 183, ref. 256], with safety concerns regarding the exacerbation of supine hypertension and urinary retention. Paresthesia, pruritus, goose bumps and shivering are relatively common, and sometimes unpleasant side effects of midodrine to monitor for.
Fludrocortisone (0.1–0.3 µg o.i.d.) is a synthetic mineralocorticoid agent and represents an additional pharmacological option to combat OH, although with a lower level of evidence regarding its efficacy and safety [ref. 110, ref. 198], mostly concerning the exacerbation of supine (and nocturnal) hypertension, and electrolyte disturbances.
Randomized controlled trials, case series and long-term observational studies reported on the efficacy of droxidopa (l-Threo-DOPS, 100–300 mg b.i.d. or t.i.d., rarely up to 600 mg t.i.d.), a synthetic noradrenalin precursor, to treat OH in individuals with PD and other primary autonomic [ref. 22, ref. 23, ref. 35, ref. 76, ref. 89, ref. 90, ref. 97, ref. 104, ref. 111, ref. 113, ref. 133, ref. 141, ref. 142, ref. 194] disorders. Based on these studies, Droxidopa was approved by the Food and Drug Administration for the treatment of OH, but is not available in Europe. Like other pressor agents, droxidopa may also exacerbate or worsen supine and nocturnal hypertension.
Smaller case series reported on the efficacy of atomoxetine, yohimbine and pyridostigmine to treat OH in different disorders, including PD [ref. 57]. However, the clinical use of the latter drugs is often limited to specialized dysautonomia centers.
| Recommendation | |
|---|---|
| To treat OH in individuals with PD, a stepwise approach should be followed | |
| 1. Eliminate or treat aggravating and triggering factors (infections, dehydration, etc.) | |
| 2. Review the medication schedule (if anti-hypertensive drugs are being used, a dose reduction or discontinuation should be sought) | |
| 3. Apply non-pharmacological therapeutic measures (see recommendations below) | |
| 4. Start pressor agents (see recommendations below) | |
| Level of consensus: 100%, strong consensus | |
| The following non-pharmacological therapeutic measures should be recommended for treating OH in PD | |
| 1. Adequate fluid and salt intake, provided there are no contraindications for this (e.g. cardiac, liver, renal failure) | |
| 2. Abstain from large meals or excessive alcohol consumption | |
| 3. Minimize heat exposure | |
| 4. Sleep in a 10°–20° head-up tilt position | |
| 5. Perform counterpressure manoeuvres in case of orthostatic intolerance (e.g. tensing the legs, buttock, abdominal and arm muscles) | |
| 6. Wear an elastic abdominal binder during daytime (more effective than compression stockings) | |
| Level of consensus: 95%, consensus | |
| Midodrine should be considered as pharmacological measure to treat OH in PD | |
| Level of consensus: 100%, strong consensus | |
| Fludrocortisone can be considered as pharmacological measure to treat OH in PD | |
| Level of consensus: 100%, strong consensus | |
| Droxidopa can be considered as pharmacological measure to treat orthostatic hypotension in individuals with PD (off-label use, not licensed in Europe) | |
| Level of consensus: 95%, consensus |
How to diagnose supine and nocturnal hypertension in PD?
PD individuals newly diagnosed with neurogenic OH, should be screened for the presence of supine and nocturnal hypertension, and such screening should be repeated at regular intervals during the disease course, especially if treatment with pressor agents is started, its dosage is increased, or if individuals with known neurogenic OH develop multiple episodes of nocturia or ankle oedema.
Measuring the BP during the supine phase of the Schellong test already provides initial insights into the presence of supine hypertension. In addition, patients should consider keeping blood pressure diaries with measurements at different times of the day [i.e., in the early morning, at midday and before going to bed, each time with measurements in the supine, (sitting) and standing position] to gain insights into circadian and activity-dependent BP fluctuations. In a second step, a 24-h ambulatory BP monitoring can be considered to determine the presence of nocturnal hypertension and to document the absolute BP values reached over the 24-h. To ensure an accurate interpretation of the exam, patients should be instructed to complete an activity diary on the day of the examination, indicating the times of medication intake (especially antihypertensive and pressor agents), meals, physical activities and any phase in the upright position overnight, e.g. to use the bathroom.
The diagnosis of supine hypertension is defined by a systolic BP ≥ 140 mmHg and/or diastolic BP ≥ 90 mmHg, measured after at least 5 min of rest in the supine position in patients with known neurogenic OH [ref. 62].
A distinction is made between the following severity degrees:
- Mild supine hypertension: systolic BP values of 140–159 mmHg or diastolic of 90–99 mmHg;
- Moderate supine hypertension: systolic BP values of 160–179 mmHg or diastolic of 100–109 mmHg;
- Severe supine hypertension: if systolic BP ≥ 180 mmHg or diastolic BP ≥ 110 mmHg.
In addition, patients with cardiovascular autonomic failure often have nocturnal hypertension, which is associated with a loss of the physiological fall in blood pressure (> 10%) during sleep. A distinction is made between two nocturnal blood pressure profiles:
- reduced dipping, characterized by an average nocturnal BP reduction < 10% with respect to daytime values.
- non-dipping OR nocturnal BP rise: when the average BP does not fall or even increases overnight.
| Recommendation | |
|---|---|
| In individuals with PD and neurogenic OH, a Schellong test with at least a 5-min in the supine position, keeping home BP diaries and 24-h ambulatory BP monitoring should be sought to diagnose supine or nocturnal hypertension | |
| Level of consensus: 95%, consensus |
How to treat nocturnal (supine) hypertension in PD?
For the treatment of nocturnal (supine) hypertension in PD, experts recommend the use of a stepwise approach [ref. 100]. Non-pharmacological preventive and behavioral measures should be sought for, including avoidance of the supine position during daytime, patient education with regard to the timing of anti-hypotensive medication intake in case of OH [e.g., avoidance of any pressor agent intake after 4 p.m.], reducing fluid intake in the evening, eating a small meal before bedtime, and sleeping in a 10°–20° head-up tilted position, or steeper if tolerated. If the concomitant OH requires pharmacological treatment, short-acting pressor agents (e.g. midodrine, droxidopa) should be preferred over long-acting ones (e.g. fludrocortisone).
Small-sized studies reported on the efficacy of bedtime administration of transdermal nitroglycerin (0.1–0.2 mg/h) [ref. 242], losartan (25–100 mg) [ref. 11], eplerenone (50 mg) [ref. 10], sildenafil (25 mg), nebivolol (5 mg) [ref. 161] and clonidine (100–150 µg) [ref. 208] in improving nocturnal hypertension in individuals with primary autonomic failure, including PD. A bedtime administration of short-acting antihypertensive agents (e.g. losartan 25–100 mg, nitroglycerine 0.1–0.2 mg/h transdermal patch overnight, carefully dosed on the individual hemodynamic profile at 24 h ambulatory BP monitoring) can be, therefore, considered in the opinion of the authors of the current manuscript in case of severe nocturnal hypertension, bearing in mind the risk of worsening OH.
| Recommendation | |
|---|---|
| For the treatment of (nocturnal) supine hypertension in PD, the following stepwise approach should be followed | |
| 1. Non-pharmacological preventive and behavioral measures (see recommendation below) | |
| 2. Switching from long-acting to short-acting pressor agents | |
| 3. Short-acting anti-hypertensive agents at bedtime (see recommendation below) | |
| Level of consensus: 95%, consensus | |
| For the prevention and treatment of (nocturnal) supine hypertension in PD, the following non-pharmacological preventive and behavioral measures should be sought | |
| Avoidance of the supine position during daytime | |
| Refraining from taking any pressor agent after 4 p.m. | |
| Reducing fluid intake in the second half of the day (after 18:00) | |
| Eating a small meal before bedtime | |
| Sleep in a 10°–20° head-up tilted position | |
| Level of consensus: 95%, consensus | |
| For the pharmacological treatment of nocturnal supine hypertension, bedtime administration of clonidine, eplerenone, losartan, nebivolol, nitroglycerin or sildenafil (alphabetically ordered) can be considered | |
| Level of consensus: 95%, consensus |
How to diagnose constipation in PD?
Lower gastrointestinal dysfunction in PD often manifests with problems with passing stools, hard stools, several days without bowel movements, feeling of fullness, and sometimes abdominal pain. Such symptoms may be already present in the prodromal stage of PD [ref. 259]. At the time of first diagnosis, constipation is present in 24–63% of PD individuals, depending on the diagnostic criteria used for constipation [ref. 219], and its prevalence raises up to 90% with disease progression [ref. 251], ultimately impacting on the quality of life of PD individuals, who may notwithstanding tend to underreport it [ref. 251].
The guideline on ‘Chronic constipation’ of the German Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF) addresses the difficulty of defining constipation in clinical practice, including the setting of PD [ref. 3]: already in 1994, Probert et al. pointed out the discrepancy between subjectively reported constipation and consensus definitions, ultimately leading to the wide prevalence ranges found in the literature. One of the main difficulties is that a purely objective definition of constipation, e.g. solely based on stool frequency, does not capture the complexity of symptoms experienced by a large proportion of individuals with chronic constipation. Many patients are, for example, only able to pass stools if pushing hard and applying a major strain, but manage in this way to have daily bowel movements. Likewise, gastrointestinal transit time measurements correlate with stool consistency rather than stool frequency. For this reason, the most recent and internationally acknowledged definition of constipation, i.e. the Rome IV criteria, combines subjective (e.g. heavy straining or incomplete evacuation) and objective parameters (e.g. stool frequency, stool consistency) [ref. 146] and was taken over in the German guideline for the diagnosis and treatment of constipation [ref. 3].
The diagnostic work-up of individuals with chronic constipation should include a detailed medical history concerning the stool behavior, medication intake including those that may worsen constipation (i.e. opioids, tricyclic antidepressants, calcium-containing antiacids, antihypertensives, antispasmodics, sympathomimetics, and diuretics), concomitant symptoms and diseases that may contribute to constipation. The physical examination should encompass an inspection of the perineal region, a digital rectal examination with evaluation of the sphincter tone at rest and upon contraction, as well as a defecation attempt [ref. 3].
Further examinations are not usually recommended unless there are severe symptoms, warning signs and/or pain warranting immediate additional examinations. These are also to be considered in case constipation does not respond to probatory laxative therapy. Additional examinations aim firstly at excluding an obstructive cause and, secondly, at clarifying the pathophysiological substrate of constipation by means of functional examinations. A stool culture is generally not recommended [ref. 3].
| Recommendation | |
|---|---|
| The AWMF criteria should be used to diagnose constipation in individuals with PD | |
| Chronic constipation is present if the following three criteria have been met for at least 3 months | |
| (1) ≥ 2 of the following symptoms should be present | |
| Lumpy or hard stools (Bristol Stool Form Scale 1–2) in > 25% of defecations | |
| Strong straining in > 25% of defecations | |
| Subjective incomplete evacuation in > 25% of defecations | |
| Subjective obstruction in > 25% of defecations | |
| Manual manoeuvres to facilitate stool expulsion in > 25% of defecations (digital manipulation, pelvic floor support) | |
| < 3 spontaneous bowel movements per week | |
| (2) Soft stools rarely occur without the use of laxatives | |
| (3) The criteria for irritable bowel syndrome are not met | |
| Level of consensus: 100%, strong consensus |
How to treat constipation in PD?
These recommendations largely reflect the German AWMF guideline for treating chronic constipation in the general population [ref. 3]. Non-pharmacological measures include a diet rich in fibers and a fluid intake of 1.5–2 L per day. Any additional fluid intake has no further effect on constipation [ref. 3]. Regular exercise should be also aimed for, while physical activity beyond the appropriate level for the respective age does not result in an increased therapeutic effect [ref. 3]. On the other hand, the relationship between constipation and physical inactivity is well acknowledged. A diet rich in fibers can result in abdominal disturbances and even pain at the beginning, but these usually diminish with prolonged administration. Especially in elderly individuals, the individual tolerance of natural dietary fibers should be taken into account and attention should be paid to sufficient fluid intake. Soluble fibers can be tried as an alternative, but it should be born in mind that the prolonged transit time typically observed in PD individuals with constipation may lower the overall tolerance to an increased dietary fibers supply [ref. 3].
Small studies showed that abdominal massages may have positive effects on constipation on PD individuals [ref. 145], especially if following a tensegrity technique, which is based on balancing the tension of muscles, fascia and ligaments [ref. 108].
Macrogol (i.e., polyethylene glycol 3350 or 4000, PEG, 13–26 g o.i.d.) is an osmotic laxative and the pharmacological option of first choice against constipation in PD. A meta-analysis [ref. 119] also showed that PEG is superior to lactulose in terms of efficacy (i.e., stool frequency, stool form, relief of abdominal pain) and safety profile. For an improved effect, macrogol should be administered regularly, not on demand, with sufficient fluids and the dosage should be adjusted individually, informing patients that even at increased dosages, macrogol remains barely absorbable [ref. 3]. Other pharmacological options include prucalopride (1–2 mg o.i.d.) or a combination of bisacodyl (5–10 mg o.i.d.) and sodium picosulphate (5–10 mg o.i.d.), administered in the evening, these latter also barely absorbed from the gastrointestinal tract. Comparative studies between macrogol and bisacodyl/sodium picosulphate are not available, but, according to a network meta-analysis, bisacodyl/sodium picosulphate appeared to be more effective than PEG [ref. 131]. Such substances have been indeed often used as rescue medications in interventional trials for constipation [ref. 3].
Fermented milk containing probiotic bacterial strains proved efficacious in increasing the frequency of defecations, improving the stool consistency and reducing the necessity of laxatives intake in PD individuals [ref. 14]. Bacterial strains contained in probiotic capsules also showed a significant increase in the number of weekly defecations and an improved quality of life in PD individuals in past interventional studies [ref. 224]. It has been postulated that probiotics may also influence PD motor and non-motor symptoms such as cognition, depression and psychosis, but long-term studies are missing at present [ref. 225].
Rectal evacuation aids are available for severe constipation, including bisacodyl-cellulose or CO2-releasing suppositories.
| Recommendation | |
|---|---|
| For the treatment of constipation in PD individuals, including geriatric ones, the AWMF guidelines for "Chronic constipation" should be followed. The most important general recommendations are (see the original guideline for detailed recommendations [ref. 3]) | |
| Secure a fluid intake of 1.5–2 L per day | |
| Avoid physical inactivity | |
| Avoid suppressing the urge to defecate | |
| Increase the amount of dietary fibers. In geriatric patients, the individual tolerance of natural dietary fibers should be taken into account and attention should be paid to sufficient fluid intake. Soluble dietary fibers can be tried as an alternative. If gastrointestinal side effects occur, the intake of dietary fibre should be reduced and other measures to treat constipation prioritized | |
| Macrogol should be used as the drug of first choice to treat constipation in PD and there is no reason to limit the period of use | |
| Sodium picosulphate, bisacodyl, and prucalopride represent alternative pharmacological options in individuals for whom general measures and macrogol were not sufficiently effective or poorly tolerated | |
| Further pharmacological options include antraquinones, linaclotide, plecanatide, sugar and sugar alcohols (lactulose, lactitol, sorbitol and lactose) | |
| Non-pharmacological measures for treating chronic constipation in the general population include biofeedback, pelvic floor training and acupuncture procedures (acupuncture, acupressure, moxibustion, electro-acupuncture), with limited evidence in the specific setting of PD | |
| In PD, probiotics, prebiotics and synbiotics (without strain-specific recommendations), as well as abdominal massage (especially if following the tensegrity technique) were shown to improve constipation and can be recommended as additional measures | |
| Level of consensus: 100%, strong consensus |
Pain
How does pain in individuals with PD differ from pain in individuals without PD in terms of frequency, impairment of quality of life and risk factors?
Pain in PD is common [ref. 197], predominantly chronic [ref. 30] and has a negative impact on quality of life [ref. 130, ref. 137, ref. 210]. The prevalence of pain in PD individuals ranges from 68 to 95%, depending on the characteristics of the examined population, applied study design and assessed pain types [ref. 29]. Pain can occur early in disease course or even precede a PD diagnosis [ref. 124], but with disease progression, the majority of PD individuals will ultimately experience painful sensations [ref. 30, ref. 137, ref. 154, ref. 237]. Female gender, dyskinesias, postural instability, motor complications and depression have been found to predict painful sensations in PD individuals [ref. 30]. The prevalence of pain in PD is likely higher than in the general population, in which 50–80% of individuals over 65 years of age reported occasional or regular pain.
| Recommendation | |
|---|---|
| Patients with PD should be asked about pain, because pain is very common in PD and has a negative impact on quality of life | |
| Level of consensus: 100%, strong consensus |
Which subtypes of pain can occur in PD?
There is no general and unambiguous pain classification system applicable to PD. For a long time, the Ford pain classification was most commonly used in PD settings. This distinguishes between musculoskeletal, radicular/neuropathic, dystonia-related and akathisia-related, as well as central pain [ref. 73, ref. 74]. Wasner and Deuschl subsequently proposed an improved four-tier taxonomy for classifying pain in PD. This taxonomy firstly distinguishes between nociceptive, neuropathic and other pain types into different categories, which are further divided into subcategories [ref. 252]. A recently proposed pain classification system for PD (PD-PCS) aims at differentiating PD-related from PD-unrelated pain using four questions, followed by additional distinctions based on the International Association for the Study of Pain (IASP) criteria. In addition to the well-known categories of neuropathic and nociceptive pain, the IASP criteria introduced the concept of nociplastic pain to describe persistent pain that arises from altered nociception, despite no clear evidence of actual or threatened tissue damage causing the activation of peripheral nociceptors, or evidence for disease or lesion of the somatosensory system causing the pain [ref. 165]. The term nociplastic pain was first used in 2019 to describe central pain and restless legs syndrome in PD [ref. 135], but has not been used in studies on pain in PD yet. The PD-PCS system has been however validated considering the presence of nociplastic pain in PD [ref. 154]. According to the PD-PCS classification, PD-related pain is defined as a pain that occurs in association with motor symptoms, responds to dopaminergic medication, or is exacerbated by PD motor fluctuations, including OFF-phenomena. Such pain can be nociceptive, nociplastic or neuropathic in nature [ref. 153, ref. 154] and is to be distinguished from pain that occurs independently from PD. To this end, pain that increases with increased rigidity and improves with dopaminergic medication intake is to be considered as PD-related or PD-dependent. Pain that develops with motor fluctuations and is often associated with dystonia, as well as visceral pain that PD individuals may describe as internal or localized in the belly, should be likewise considered as PD-related. Unpleasant and sometimes painful sensations in the extremities such as edema or in connection with restlessness, especially overnight, such as in the case of restless legs syndrome, should also be inquired about. Examples of frequent pain causes that are less related to PD include pain due to articular or vertebral osteodegenerative changes, even though these may also occasionally improve with dopaminergic medications.
There is consensus that musculoskeletal pain is the most common type of pain in PD. Several epidemiological studies reported that this nociceptive form of pain occurs in 40–80% of PD individuals [ref. 41, ref. 137, ref. 189, ref. 237]. Neuropathic pain has been reported in 10–30% of PD individuals [ref. 137, ref. 152], while Mylius et al. found a 22% frequency of PD-related nociplastic pain, laying between nociceptive (55%) and neuropathic (16%) pain [ref. 153, ref. 154].
| Recommendation | |
|---|---|
| In PD individuals, a distinction should be made between PD-related and PD-unrelated pain | |
| When it comes to PD-associated pain, a distinction should be applied between nociceptive, nociplastic and neuropathic pain | |
| Level of consensus: 100%, strong consensus |
Which scales and questionnaires are suitable for assessing pain in PD?
Identifying pain in PD is important, because it is frequent and has a major impact on the quality of life of affected individuals. Individuals with PD may not see a connection between painful sensations and their PD diagnosis and therefore often do not report it to their treating neurologist. This could be one of the reasons why neurologists often play a subordinate role in the treatment of pain compared to orthopedists and general practitioners [ref. 30]. PD individuals should be therefore specifically asked about painful sensations during neurological consultations, taking into account the following pain subtypes: musculoskeletal, neuropathic, fluctuation-dependent, dystonic, visceral (often referred to as deep internal) and pain in the extremities accompanied by edema or restless legs syndrome. Pain documentation should be ideally carried out in a structured way using scales or questionnaires.
Most PD scales unfortunately only have a marginal focus on pain. In the Unified Parkinson Disease Rating Scale (UPDRS), item 17 asks about “sensory complaints as a result of parkinsonism” and item 34 asks to what extent dyskinesias are painful. The newer Movement Disorder Society UPDRS (MDS-UPRDS) also asks little about pain (Item 1.9: Pain and other sensations; Item 4.6: Painful dystonia in the off state).
In the Non-Motor Symptom Scale (NMSS), item 27 asks about pain that cannot be explained by other illnesses. In the newer MDS Non-Motor Rating Scale (MDS-NMS), pain is assessed more in detail with a division into muscle/joint/back pain versus deep or dull body pain versus dystonia-related pain or pain from other causes such as nocturnal or facial pain. The MDS-NMS also documents pain severity and to what extent pain severity changes in response to motor fluctuations.
At present, two pain assessment tools have been specifically developed and validated in PD settings: the King’s Parkinson’s disease Pain Questionnaire and related Scale (KPPQ and KPPS) [ref. 41, ref. 103], and the Parkinson’s disease pain classification system (PD-PCS) [ref. 137, ref. 153]. In the KPPS, pain is initially divided into 7 categories (musculoskeletal, pain deep in the body, fluctuation-dependent pain, nocturnal pain, facial pain, pain caused by edema, radicular pain), and subsequently further characterized based on 14 subcategories. In the PD-PCS, an association between pain and PD is inquired before further categorizations are operated. An association between pain and PD is assumed, if at least one of the following criteria applies: pain has a temporal association with the development of parkinsonism, increases if rigidity, bradykinesia or tremor worsen, is associated with motor fluctuations and/or decreases under dopaminergic medication intake.
| Recommendation | |
|---|---|
| The following scales/questionnaires should be used to assess pain in Parkinson’s patients | |
| 1. Pain assessment and pain categorization: Parkison’s disease pain classification system (PD-PCS) | |
| 2. Pain assessment and quantification: King’s Parkinson’s disease Pain Scale (KPPS) | |
| Level of consensus: 100%, strong consensus |
How to treat pain in PD?
In clinical practice, an attempt should be made to classify pain according to its etiology (mechanism-based classification approach) and this should in turn guide the therapeutic strategy.
Optimizing dopaminergic and other anti-parkinsonian medications is essential to improve PD-related pain. This also includes the treatment of painful dystonia and restless legs syndrome that fall under the category of nociplastic PD-related pain. Regardless of whether PD-related or not, nociceptive pain should be treated according to the WHO 3-step pain treatment ladder, while the treatment of neuropathic pain should follow the guidelines for neuropathic pain in other diseases. An algorithm for treating pain in PD individuals is proposed in Fig. 1 [ref. 29].
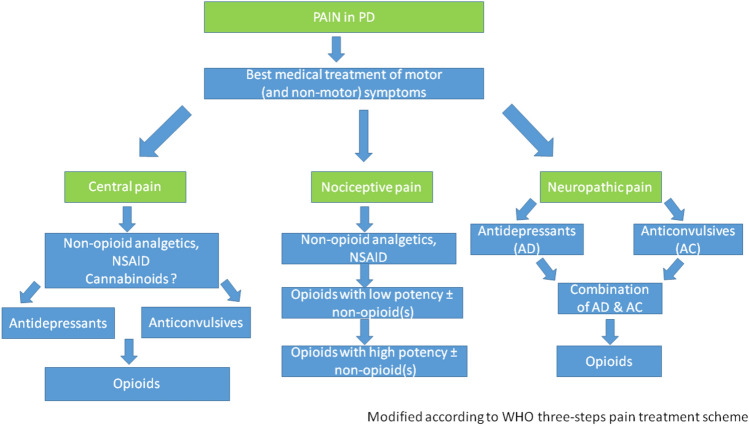
There is no uniform approach to the treatment of pain in PD, because the painful sensation experienced by single individuals can vary substantially in terms of quality and quantity, and because there are no comparative trials between different approaches. In available controlled and uncontrolled trials with different compounds, pain was usually not the primary endpoint, no analysis was performed according to different pain subtypes, and a placebo or a standard of care was usually used as a control. Pain-relieving effects have been demonstrated for some antiparkinsonian medications, including pramipexole, rotigotine and safinamide (summarized in [ref. 29, ref. 106]). Advanced PD therapeutic options such as apomorphine or levodopa pump-therapies or deep brain stimulation of the subthalamic nucleus could also improve specific pain subtypes [ref. 29]. It is conceivable that part of the pain reduction achieved with classic antiparkinsonian drugs is due to an improved dopaminergic stimulation or a reduction in motor fluctuations, so that pain treatment strategies may entail an adjustment of levodopa dosages or add-on of COMT inhibitors.
Table 1 provides an overview of the randomized control trials for the treatment of pain in PD with antiparkinsonian medications and opiates [ref. 29], as well of the studies with other compounds providing lower levels of evidence for pharmacological treatment of pain in PD individuals. Additionally to the consensus guidelines, local botulinum toxin application might be considered in special cases of pain refractory to maximized oral or transdermal drug therapy [ref. 29].
Table 1: Studies investigating the effects of antiparkinsonian medications, opioids and other drugs on pain in individuals with PD
| Treatment | Study design | Applied pain scales | No. of patients versus controls | ResultsMeans with (SD), (range) or (CI) |
|---|---|---|---|---|
| Randomized controlled trials | ||||
| Pramipexole [ref. 16] | RCT | VAS-P | N = 138 versus 148 | − 3,3 (− 8.9 to 0.3) versus − 2.4 (− 8.9 to 2.6)Group comparison − 1.3 (− 3.3 to 0.8) p = 0.19 |
| Safinamide [ref. 36] | RCT (post hoc) | PDQ-39 | N = 440 versus 438 | 100 mg daily dosage: 23.6% reduction of the number of concomitant pain treatments [95% confidence interval (CI): 41.1%, 1.0%; p = 0.0421]PDQ-39: item 37 (− 0.26 vs. − 0.07; p = 0.0009), item 38 (0,19 vs. 0.1; p = 0.1585), item 39 (− 0.18 vs. − 0.03; p = 0.0060) |
| Entacapone [ref. 162] | RCT | PDQ-39 | N = 281 vs. 274 | (0.04) versus 0.1 (0.04); p = 0.9. SMD = 1.81, p < 0.0001 |
| Oxycodone [ref. 233] | RCT | KPPS | N = 88 versus 106 | 5.0 (95% CI 4.5 to 5.5) versus 5.6 (5.1 to 6.0)Difference − 0.6, 95% CI − 1.3–0.0; p = 0.058) |
| Pardoprunox [ref. 185] | RCT (post-hoc) | VAS-P | N = 140 versus 133 | During OFF time − 2.2 (2.7) versus − 1.0 (2.7), no p-values givenDuring ON time − 2.3 (2.8) versus − 0.5 (3.0), no p-values given |
| Rotigotine [ref. 109, ref. 234] | RCTRCT (post-hoc) | Likert pain scaleLikert pain scale | N = 178 versus 89N = 178 versus 89 | − 0.9 (SD 2.2) versus − 0.1 (SD 2.3), p = 0.004Difference − 0.77 [− 1.28 to − 0.25]’Any pain’: − 0.88 [95% CI − 1.56, − 0.19], p = 0.013)’Moderate-to-severe’ pain: (− 1.38 [− 2.44, − 0.31], p = 0.012UPDRS III or PDSS-2 responders showed greater improvement in pain than non-responders |
| Studies with anti-Parkinsonian medications with lower level of evidence (III or IV) | ||||
| Rotigotine [ref. 109, ref. 231] | RECOVER—Post-hoc analysisNEUPAD—Non-interventional multicenter observational study over 4 weeks | Likert pain scaleerman Pain Questionnaire | N = 178 versus 89N = 70 | Predictors for treatment response: higher pain level, improvement in UPDRS III and PDSS-2 with rotigotine treatmentNon-significant reduction in the affective dimension of pain (probably dependent on motor improvement) |
| Safinamide [ref. 36, ref. 37, ref. 82, ref. 195] | Post-hoc analysis of studies 016 and SETTLE (placebo-controlled) over 24 weeksPost-hoc analysis of study 018 (placebo-controlled) over 2 yearsOpen-label prospective single center study over 12 weeksOpen-label prospective multicenter study over 6 months (SAFINOMOTOR; Phase IV) | PDQ-39PDQ-39KPPS, BPIKPPS, PDQ-39 | Study 016: N = 224 versus 222Study SETTLE: N = 274 versus 275N = 180 versus 175N = 13N = 50 | Improvement in 2 out of 3 domains of physical limitations (musculoskeletal and neuropathic pain) in the PDQ-39Reduced frequency of concomitant pain therapies [100mg/d] [36, 37]Significant improvement in PDQ-39 items 37 (painful cramps or spasm) and 39 (unpleasantly hot or cold)Significantly reduced number of concomitant pain medication use versus baseline and versus placeboSignificant improvement in KPPS and BPI Intensity and InterferenceSignificant improvement of PDQ-39, Item 8 (pain and discomfort)-Significant improvement on the KPPS, − 43.6% (p < 0.0001) |
| Levodopa-Carbidopa intestinal gel [ref. 8] | GLORIA; international postmarketing study | UPDRS, NMSS, PDQ-8, EQ-5D | N = 375 | -Less painful dyskinesia (after 12 months of treatment) and muscle cramps (after 6 months) |
| Studies with other pain medications | ||||
| Non-opioid analgesics | No studies available | |||
| Antidepressants | ||||
| Duloxetine [ref. 53] | Open-label, 6 week | VAS, BPI, Mc Gill short form, PDQ-39, UPDRS-III | N = 20 | Reduction of central and peripheral neuropathic pain |
| Antiepileptics | With the exception of gabapentin, no studies addressing pain in PD, data for efficacy on neuropathic pain driven from other populations | |||
| Gabapentine [ref. 163, ref. 238] | Double-blind, placebo-controlled, crossover trialDouble-blind, placebo-controlled, crossover trial, 4 weeks of treatment | UPDRSUPDRS | N = 19N = 20 | Total UPDRS improvement with gabapentine compared with placebo (p = 0.0005)UPDRS III improvement with gabapentine compared with placebo (p < 0.001) |
| Botulinum toxin | ||||
| Incobotulinum toxin A [ref. 186] | Double-blind randomized trial 6 and 18 weeks post-injection | CGI, Dystonia severity and associated pain | N = 45 | Reduced dystonic plantar flection and associated pain in BoNT group versus baseline but not versus placebo |
| Onabotulinumtoxine A [ref. 28] | Double-blind, randomized crossover trial 4 and 12 weeks post-injection | NRS | N = 12 | Reduced global limb pain in BoNT group versus baseline but not versus placebo |
| Cannabinoids | ||||
| Marijuana [ref. 128, ref. 209] | Two open-label, uncontrolled observational studies | UPDRS, present pain intensity scale, PRI, VAS, Mc Gill short-form | N = 22 and N = 20 | Pain reduction measured with different scores 30 min after smoking 0.5–1.0 g Marijuana |
| Nabilone (synthetic THC analogue) [ref. 170] | Double-blind, placebo-controlled, randomized 4-weeks trial | KPPS, VAS | N = 38 (19 nabilone, 19 placebo) | Pain improvement |
| Various cannabinoids [ref. 68, ref. 260] | Two question-naire-based uncontrolled national studies in the PD community | Ad-hoc questionnaires | N = 113 (Germany)N = 261 (USA) | Pain reported in 57 PD individuals with improvement in N = 25 (44%)Pain reported in 88 patients with considerable or moderate improvement of severity and frequency in N = 63 (72%) and N = 56 (64%), respectively |
BoNT Botulinum Neurotoxin, BPI Brief Pain Inventory, CGI Clinical Global Impression, CI confidence interval, EQ-5D European Quality of Life-5 Domains, KPPS King’s Parkinson’s disease Pain Scale, NMSS Non-Motor Symptom Scale, NRS Numeric Rating Scale, PDQ-39/8 Parkinson’s disease questionnaire-39/8 items, PDSS Parkinson’s disease sleep scale, PRI Pain rating Index, RCT randomized-controlled trial, SD standard deviation, VAS-P visual analogic scale-pain, UPDRS Unified Parkinson’s disease rating scale
| Recommendation | |
|---|---|
| Severe chronic pain in PD should be treated in a multimodal, interdisciplinary manner, like other pain disordersPain treatment should be guided by the etiology of painThe basis of treatment is to optimize the antiparkinsonian medication schedule, whereby levodopa, dopamine agonists, COMT inhibitors or safinamide can be used to treat dopamine-dependent painNociceptive pain should be treated according to the 3-step WHO regimenNeuropathic pain should be treated with anticonvulsants and/or antidepressants according to the guidelines for the treatment of neuropathic pain, with preference given to gabapentin and/or duloxetine (particularly in case of concurrent depression)If pain is severe, treatment with oxycodone/naloxone may be considered | |
| Level of consensus: 100%, strong consensus |
Sleep
Which sleep disturbances may occur in PD?
Individuals with PD report insomnia with difficulties falling asleep and maintaining sleep, pain, involuntary movements and restlessness of limbs, worsening of motor symptoms such as nocturnal akinesia, rigidity and/or resting tremor, nocturia, nocturnal behavioral disorders, including REM-sleep behavior disorders (RBD) and non-REM parasomnias, anxiety, non-restorative sleep with fatigue and/or daytime sleepiness, and breathing disorders [ref. 38, ref. 207, ref. 218]. The etiology of these symptoms may be classified into comorbid primary sleep disorders, motor and non-motor symptoms of PD, coexistence and influence of neuropsychiatric disturbances, autonomic dysfunction, side-effects of PD medications and the impact of neurodegeneration itself on cerebral sleep regulating centers [ref. 38, ref. 134, ref. 218, ref. 244, ref. 266, ref. 268]. A bidirectional relationship between sleep disorders and disease progression has been postulated [ref. 266].
Comorbid primary sleep disorders
Sleep disorders listed as an independent diagnosis in the ICSD-3 (International Classification of Sleep Disorders) are considered primary sleep disorders. These entail Restless Legs Syndrom (RLS), sleep disordered breathing (SDB), and parasomnias, including RBD, which occurs in PD as well as in other α-synucleinopathies:
- RLS: The overlap between nocturnal restlessness in PD, for instance in the context of wearing-off phenomena, and RLS may complicate the diagnostic differentiation [ref. 38]. Several studies investigating the prevalence of RLS in PD came to contradictory results [ref. 21, ref. 85], whereas a meta-analysis concluded on an overall increase in RLS prevalence in PD [ref. 5]. Due to the distinctly different pathophysiology, RLS is interpreted as a comorbidity in PD, with newly diagnosed PD individuals initially showing the same prevalence of RLS as non-PD subjects. The prevalence of RLS increases with PD progression and dopaminergic daily dosage. Whether individuals with RLS have a higher risk of developing PD is a matter of debate [ref. 201]. A prospective longitudinal questionnaire study showed an elevated incidence of PD in individuals with severe RLS [ref. 222].
- SDB: Obstructive and central sleep apneas with recurring oxygen desaturation lead to sleep fragmentation, nocturia, daytime sleepiness and increased cardiovascular risk. Data on the prevalence and severity of SDB in PD patients are controversial [ref. 218]. However, a recent meta-analysis showed that PD patients with obstructive sleep apnea (OSA) suffer more severe motor and cognitive symptoms [ref. 56]. Although the occurrence of excessive daytime sleepiness in PD does not solely depend on the severity of SDB and the risk for a severe form of SDB does not seem to be elevated in PD patients, an individualized diagnostic work-up and therapeutic approach is recommended to minimize the risk for concomitant cardiovascular disease [ref. 218].
PD-related symptoms
- Nocturnal „wearing-off“: Actigraphy studies show a decrease in spontaneous movements towards the morning in PD patients, suggesting a dopaminergic deficit [ref. 121, ref. 215] with recurrence of motor symptoms such as tremor, rigidity and akinesia with patients‘ inability to turn in or get out of bed, increased nocturia, as well as painful dystonic cramps in early-morning OFFs.
- Wearing-off phenomena may include neuropsychiatric symptoms, such as anxiety and depression.
- Autonomic failure: especially at advanced PD stages, autonomic features such as nocturia, OH, constipation, reflux, pain and sweating may cause or worsen sleep fragmentation [ref. 24].
Adverse effects of pharmacotherapy
Dopaminergic medication, especially dopamine agonists are known to induce daytime sleepiness and sleep attacks [ref. 79]. This adverse effect appears dose-dependent: high dosages increase sleep fragmentation and nocturnal wakefulness and reduce the amount of slow-wave sleep, whereas low dosages improve sleep quality and reduce awakenings after sleep onset [ref. 226]. Amantadine is also known to increase nocturnal wakefulness and reduce slow-wave sleep [ref. 230]. In case of sleep disturbances, the beneficial and adverse effect of these medications needs to be balanced. Hallucinations, psychosis and impulse control disorders likewise have a negative impact on objective and subjective sleep quality [ref. 120, ref. 190, ref. 218, ref. 269].
Neurodegenerative alterations in sleep-regulating networks
- RBD: RBD is a recognized possible prodromal feature of PD and other α-synucleinopathies, as well as a progression marker in clinically manifest PD [ref. 19, ref. 157, ref. 164] that may lead to nocturnal disruptive behaviors, stressing both PD individuals and their bed partners. A careful differentiation of other abnormal nighttime behaviors by means of video-polysomnography (VPSG) is essential.
- Changes in sleep micro-and macrostructure: with disease progression, a progressive deconstruction of normal sleep macrostructure with a loss of slow-wave sleep, as well as changes in the sleep microstructure are observed [ref. 134, ref. 177, ref. 266, ref. 268]. The loss of delta waves and sleep spindle reduction has been associated clinically with faster cognitive decline [ref. 52], likely due to a disturbed sleep-dependent restorative function for metabolic homeostasis. The destruction of physiologic sleep architecture and loss of slow-wave sleep also impairs the glymphatic system—a glial-dependent perivascular network with an important role in the clearance of neurotoxic protein waste, such as beta-amyloid and aggregated α-synuclein. These factors may contribute to the malfunction of cerebral sleep regulating centers and possibly enhance the neurodegeneration cascade [ref. 25, ref. 202, ref. 266]. Clinically, patients complain about difficulties with sleep maintenance, early morning awakenings, non-restorative sleep, and in some cases problems falling asleep.
- Impairment of circadian rhythm: changes in the expression of clock genes have been found in PD individuals. These in turns impair the circadian rhythm of kidney function, blood pressure regulation, as well as of ADH, cortisol and melatonin secretion. A progressive loss of orexinergic neurons in the prefrontal cortex and hypothalamus has been found to result in a significant fall of orexin and melatonin levels [ref. 94, ref. 134, ref. 247, ref. 261]. Again, a bidirectional relationship between impaired circadian rhythm and neurodegeneration has been hypothesized [ref. 94].
| Recommendation | |
|---|---|
| Sleep disturbances in PD are frequent, manifest with multiple symptoms and should be considered in all diagnostic and therapeutic decisions. Frequent sleep disturbances in PD include insomnia, pain, RLS, nocturnal akinesia, tremor, rigidity, nocturia, parasomnias, nightmares, SDB and RBD | |
| Level of consensus: 100%, strong consensus |
How to screen for sleep disturbances in PD?
PD individuals seldom report sleep problems spontaneously. Besides specifically addressing individual sleep history and questioning bed partners and caregivers, validated questionnaires help to screen for sleep disturbances and estimate the overall sleep satisfaction. Sleep diaries and morning/evening protocols may also be useful.
Regarding screening questionnaires, an expert consensus conference endorsed by the Movement Disorder Society [ref. 92] recommended to use the Parkinson’s Disease Sleep Scale (PDSS-2), the Pittsburgh Sleep Quality Index (PSQI) and the Scale for Outcomes in Parkinson’s Disease-Sleep (SCOPA-S). The PSQI was not validated in PD [ref. 31]. The SCOPA-S was validated for PD and captures PD-specific nocturnal disturbances as well as daytime sleepiness during the last 4 weeks, but does not ask for potential causes [ref. 136, ref. 138]. The PDSS-2 was validated as a numeric scale [ref. 235] based on the original PDSS as a visual analogous scale [ref. 40]: 15 questions cover overall sleep quality and its restorative effect, as well as PD specific nocturnal problems like nocturnal akinesia, nocturia, nightmares, restlessness, nocturnal pain, and nocturnal breathing problems during the last week, thus creating a profile of specific complaints and potential causes. A cut-off of > 18 points has been validated as indicative of a sleep disturbance and warranting an additional diagnostic work-up with vPSG [ref. 151]. Sleep therapeutic studies usually utilize the PDSS-2, as it enables simplified comparisons and follow-up [ref. 138]. There are no comparative studies between the three scales.
| Recommendation | |
|---|---|
| The PDSS-2 should be used as a screening instrument for sleep disturbances in PD: the numeric scale simplifies comparisons. The evaluation of reported complaints and symptoms may point towards potential causes of the sleep disturbance | |
| Level of consensus: 100%, strong consensus |
How to diagnose sleep disturbances in PD?
If patient and/or caregiver reports, sleep diaries and questionnaires do not clarify the nature of the sleep disturbance, objective measurements are needed to document the course of nocturnal sleep including motor phenomena and behaviors.
- Actigraphy: Around-the-clock activity and resting states are registered in the home environment for a longer period of time, thus enabling an estimation of total sleep time, sleep efficiency, sleep latency and sleep fragmentation, as well as screening for circadian rhythm disorders. The accuracy of actigraphy deteriorates with disease progression [ref. 132, ref. 143].
- Cardiorespiratory polygraphy additionally collects data on breathing, cardiac activity and oxygen levels. It is mainly used to screen for SDB in an outpatient setting.
- vPSG: a laboratory-based vPSG currently represents the diagnostic gold standard. Additional EEG and EMG registrations enable sleep staging and a more in-depth analysis of sleep structure, sleep fragmentation and sleep efficiency. Synchronized video-registration of nocturnal behaviors and motor phenomena helps to clarify nocturnal disturbances that are not satisfactorily explained by patient history, bed partner/caregiver report or the above mentioned screening instruments. According to the International Classification of Sleep Disorders (ICSD-3), vPSG is currently mandatory for a diagnosis of RBD. Future technologies may close the gap between outpatient screening and sleep laboratory settings, as pointed out by recent data on the actigraphy-based detection of RBD [ref. 184].
| Recommendation | |
|---|---|
| In case of sleep disorders that require treatment, objective assessments such as actigraphy, cardiorespiratory polygraphy and vPSG can be recommended, especially if first therapeutic measures, such as optimizing medication, did not prove beneficialActigraphy may be considered to estimate total sleep time, sleep efficiency and wakefulness in PD individuals, facilitating the sleep rhythm evaluation over a longer period of time. The accuracy of actigraphy however deteriorates with disease progressionvPSG is the gold standard for identifying sleep disturbances and a prerequisite for diagnosing RBD. If severe SDB is suspected, polygraphy or vPSG should be recommended | |
| Level of consensus: 100%, strong consensus |
How to treat sleep disturbances in PD?
The diverse etiology and clinical manifestations of sleep disorders in PD imply an individualized therapeutic approach addressing symptoms based on their sleep-related diagnosis.
Comorbid primary sleep disorders
- RLS: there are no randomized controlled trials for the treatment of RLS in the specific setting of PD. Currently valid German guidelines for RLS (without comorbid PD) treatment [ref. 91] recommend a work-up of iron metabolism, aiming at high normal serum levels of ferritine and transferrin saturation. RLS-aggravating medications, such as some antidepressants should be tapered and discontinued if possible. If these measures are insufficient, optimizing dopaminergic medication according to the individual risk profile, as well as alpha-2-delta ligands or opioids may be considered [ref. 38, ref. 201].
- SDB: Treatment for SDB in PD individuals should follow the current guidelines for treating sleep apnea in the general population [ref. 38, ref. 144, ref. 201, ref. 226]: in case of obesity, weight reduction and nocturnal positive pressure ventilation—either as CPAP or BiPAP—are recommended. A randomized controlled study showed that CPAP therapy for PD-associated SDB reduces the apnea–hypopnea index and shortens sleep latency [ref. 155]. However, therapy adherence in PD individuals is often quite low. Both PD individuals and their caregivers may feel encumbered by the ventilation device, so that an individual benefit assessment seems necessary.
PD-related symptoms
- Nocturnal wearing off: treatment should focus on adapting dopaminergic therapy [ref. 201, ref. 226]. Long-acting dopamine agonists or slow-release levodopa or pump therapy may be considered, with re-evaluation of sleep quality. In case of increased daytime sleepiness, dopaminergic therapy might have to be reduced, for example with switch between different dopamine agonists or a change to levodopa. In case of persisting sleep disturbances, further diagnostic work-up by vPSG may be considered to exclude SBD, among others.
Neurodegenerative alterations in sleep-regulating networks
- RBD: therapeutic guidelines based on high evidence levels do not exist due to difficulties in defining adequate assessment methods. Expert consensus calls for a safe sleep environment and recommends the use of clonazepam, melatonin or rivastigmine [ref. 245]. Adverse effects of clonazepam on balance and alertness must be considered, especially in elderly patients. An analysis of all clinical and scientific evidence on the treatment of RBD published in 2022 shows the most positive results for clonazepam (dosages used in comparative studies: 0,125–3 mg) and melatonin (dosages used in comparative studies: 2–9 mg) [ref. 84]; therefore, these substances are regarded as first choice medication.
- Impairment of circadian rhythm: the pathophysiological substrate of insomnia associated with alterations of circadian rhythms provides the rationale for melatonin administration in PD. A multicentre, randomized double-blind, placebo-controlled study indeed demonstrated the safety and efficacy of 2 mg slow-release melatonin [ref. 2], if assessed by means of the PSQI, as a questionnaire non-specific for PD, the Non-Motor-Symptom Scale (NMSS) and PDQ39, while another study had negative results [ref. 50]. Due to the favorable safety profile, the updated MDS evidence-based recommendations [ref. 204] regards the use of melatonin as potentially useful and therefore justified for treating circadian rhythm disorders in PD. Non-pharmaceutical measures include bright light application, which showed positive effects on sleep quality and diurnal physical activity [ref. 139, ref. 246], general advice on sleep hygiene [ref. 201], as well as enforced physical training [ref. 46]. A randomized controlled study showed a significant improvement of sleep quality and efficiency under intensive physical training compared to sleep hygiene measures alone [ref. 46].
- Changes in micro- and macrostructure of sleep: after excluding other sleep disorders like RLS, SDB, nocturnal wearing-off or depression, a pharmacological treatment may be considered following current German guidelines for treating insomnia in neurological disorders [ref. 143]. Eszopiclon (1 mg), doxepin (25 mg), zolpidem (5 mg), trazodone (50 mg), melatonin (2 mg) as well as the antipsychotic drug pimavanserin and the antidepressants venlafaxin (37,5 mg) and nortryptilin (25 mg, both to be considered in case of co-existing depression) may be tried, despite insufficient evidence. Based on a randomized controlled study in non-PD insomniacs [ref. 107], the antidepressant mirtazapin may be also recommended in doses of 7.5–15 mg. In any case, pharmacological treatment should be started with the lowest possible dosage. Further adjustments may be required on an individual basis and side effects should be carefully monitored at follow-up. To date, no sleep-restorative therapies are available.
| Recommendation | |
|---|---|
| Comorbid sleep disorders such as RLS or SDB should be treated according to their specific guidelinesIf motor or non-motor dopaminergic fluctuations are responsible for the sleep disturbance, dopaminergic therapy should be adjusted accordinglyRBD should be treated by creating a safe sleep environment. Clonazepam and/or melatonin may be considered, but potential side effects need to be taken into accountAfter exclusion of medication side effects and/or a primary sleep disorder such as SDB or otherwise treatable causes, insomnia and circadian rhythm disorders can be treated by sleep hygiene, intensive physical training and bright light therapy. Eszopiclon, doxepin, zolpidem, trazodone, melatonin, venlafaxin (in case of comorbid depression), nortryptilin or mirtazapin may be considered for insomnia, despite insufficient evidence (95% consensus) | |
| Level of consensus: 95%, consensus |
How effective are slow-release levodopa preparations or dopamine agonists for treating nocturnal PD motor symptoms?
Whenever PD individuals complain of the nocturnal recurrence of PD symptoms like the inability to turn in or get out of bed, off-dystonic cramps, or the inability to fall asleep again because of tremor worsening, the application of long-acting dopaminergic agents overnight should be considered. To date, the transdermal application of rotigotine is the best investigated dopaminergic treatment option: randomized, controlled trials showed a significant improvement of sleep disturbances [ref. 234], with increased sleep efficiency, shortened sleep latency and reduced nocturnal awakenings under rotigotine treatment [ref. 172]. Pramipexole was also effective, and ropinirole proved non-inferior [ref. 257]. An open-label study showed improvements on PDSS-2 scorings by combining levodopa/carbidopa with entacapon [ref. 168]. PD patients on pump-based continuous delivery of dopaminergic medication will benefit from extending the pump running time through the night. Both an extension of daytime subcutaneous apomorphine infusion [ref. 69] and isolated nocturnal applications [ref. 48] led to a significant improvement of sleep quality. Levodopa/carbidopa intestinal gel instillation via PEG/PEJ through the night also proved beneficial on subjective sleep quality and daytime sleepiness [ref. 39, ref. 49, ref. 216]. Of note, dopamine agonists may cause daytime sleepiness and sleep attacks in a dose-dependent manner. Overnight, dopamine-agonists at high dosages were shown to induce nocturnal wakefulness, increase sleep fragmentation and reduce slow-wave sleep, whereas lower dosages reduced nocturnal wakefulness and improved sleep quality [ref. 226].
A recent overview [ref. 270] described a positive effect of deep brain stimulation on sleep latency and fragmentation. Whether this is due to a direct effect of the stimulation or rather to the postoperatively reduction of dopaminergic medications remains unclear.
It should be kept in mind that many substances successfully used in PD pharmacotherapy have never been tested in trials with sleep disorders as primary endpoint. This is for example the case of slow-release levodopa formulations. The clinical experience, however, underlines its therapeutic benefit, especially when dopamine agonists are contraindicated.
| Recommendation | |
|---|---|
| For sleep disturbances due to a nocturnal dopaminergic deficit, long-acting dopamine agonists such as rotigotine can be recommendedSlow-release levodopa may be offered as an alternativePatients on pump therapy due to PD fluctuations may be offered a nocturnal extension of the pump running time | |
| Level of consensus: 100%, strong consensus |
How to screen for excessive daytime sleepiness in PD?
Daytime sleepiness and sleep attacks show an increased prevalence in PD individuals, representing a major health hazard and impacting on the disease burden, daytime activities, social interactions, ability to drive and, overall, quality of life [ref. 6, ref. 214, ref. 262]. Although the propensity to sleep during the day may be an early symptom of PD [ref. 80], excessive daytime sleepiness increases with disease progression [ref. 6, ref. 211] and may predict mortality [ref. 173]. Fatigue needs to be differentiated from daytime sleepiness. A fatigued person may feel tired, prematurely exhausted, but will not unintentionally fall asleep during resting episodes. PD individuals with excessive daytime sleepiness may, by contrast, frequently fall asleep in monotonous situations and may not be aware of sleep intrusions during the day. Caregivers’ reports are therefore essential for a timely identification [ref. 126].
The SCOPA-S includes questions about daytime sleepiness during the past 4 weeks [ref. 136, ref. 138]. The Epworth Sleepiness Scale (ESS) was purposely designed to capture and grade daytime sleepiness [ref. 99], with a cutoff > 10 points marking excessive daytime sleepiness, and is applicable in PD settings. Many affected patients may however lack an awareness for the problem, with substantial discrepancies between subjectively perceived and objectively measured daytime sleepiness [ref. 13].
| Recommendation | |
|---|---|
| Besides patient history and caregiver reports, the ESS is a validated and recommended screening tool for daytime sleepiness in PD | |
| Level of consensus: 100%, strong consensus |
How to diagnose excessive daytime sleepiness in PD?
The Multiple Sleep Latency Test (MSLT) and the Maintenance of Wakefulness Test (MWT) are considered the gold standard for objectively evaluating daytime sleepiness. Both tests require a sleep laboratory setting. The MWT measures the ability to stay awake in a monotonous situation [ref. 147], whereas the MLST evaluates the time it takes to fall asleep in a monotonous situation [ref. 9, ref. 34]. Due to these opposing concepts, the concordance between both tests is low. Of note, neither the MSLT nor the MWT have been validated in PD. Assessments in individuals with obstructive sleep apnea (OSA) also showed discrepancies between ESS scorings and objective measurements [ref. 18, ref. 32, ref. 72]. MWT and driving simulation tests in OSA patients correlated with regard to driving capability [ref. 174]. However, there is no evidence that MWT results correspond to driving accidents in the real world [ref. 174]. ESS and MSLT did not, but TAP-test and Psychomotor Vigilance Test Reaction Time results did correlate to driving simulator performance in OSA patients [ref. 95]. In PD, studies show that the subjective perception of sleepiness and the objectively measured probability of falling asleep unintentionally may deviate [ref. 13]: every fourth PD individual did not perceive any sleepiness prior to daytime sleep tests, which objectively demonstrated sleep episodes. Evaluation of driving capability or potentially accident-prone daytime sleepiness in the work environment should therefore not rely on subjective perception, although there is no objective gold standard for diagnosing daytime sleepiness in PD individuals at present. VPSG-based assessment of nocturnal sleep quality is an important prerequisite for interpreting daytime vigilance tests properly and identifying potential causes of daytime sleepiness. Several studies demonstrated however an insufficient correlation between nocturnal sleep quality and subjective or objective measurements of daytime sleepiness [ref. 123, ref. 217]. Self-perceived daytime sleepiness was also not consistently reflected in MSLT [ref. 217]. Abovementioned observations altogether indicate that the mechanisms underlying daytime sleepiness in PD are currently poorly understood and that tools for detecting and diagnosing daytime sleepiness in PD are insufficient.
| Recommendation | |
|---|---|
| For a diagnosis of daytime sleepiness, a detailed medical history, caregiver reports, questionnaires such as ESS, as well as an assessment of possible causes of daytime sleepiness are recommended. An assessment of nocturnal sleep quality by means of vPSG, as well as MSLT and other neuropsychological test procedures including PVT or driving simulator test may be considered | |
| Level of consensus: 92.6%, consensus |
How to treat excessive daytime sleepiness in PD?
If the diagnostic workup identifies a sleep disorder as a possible cause for daytime sleepiness, an etiology-driven therapeutic approach according to the guidelines summarized above is recommended. Especially motor or non-motor wearing-off phenomena inducing nocturnal sleeplessness call for the adjustment of dopaminergic medications. Total levodopa-equivalent dose should be critically appraised and the possibility of a dose-reduction or medication redistribution should be considered, since suboptimal medication timing may contribute to sleeplessness and impaired daytime vigilance [ref. 70]. Sleep attacks associated with dopamine agonists [ref. 78] need to be excluded. A switch to amantadine [ref. 207] or invasive therapies may prove beneficial.
Non-pharmacological approaches include improvement of sleep hygiene with regular bedtimes and naps, social activation, bright light application to regulate circadian rhythm and intensified physical activities [ref. 244]. Intensive sport improved subjective sleep quality [ref. 244] and power training has been shown to improve both sleep and cognition [ref. 258].
If all treatable causes of excessive daytime sleepiness have been excluded and non-pharmacological measures prove insufficient, medication may be considered, although evidence is poor. Up to 400 mg of caffeine showed minimal, clinically non-meaningful effects on the ESS [ref. 175]. A meta-analysis of studies investigating modafinil in PD, using dosages of 200–400 mg as per prescribing information, demonstrated a reduction of subjective, but not objectively measured daytime sleepiness [ref. 188]. An open-label study showed preliminary positive effects for methylphenhidate [ref. 51]. Istradefylline, a selective adenosine A2A receptor antagonist, improved daytime sleepiness in an open label study [ref. 221], but is not licensed in Europe. Gamma hydroxybutyric acid improved PSG-based measurements in a double-blind randomized phase II study, but had a severely unfavorable safety profile and cannot be therefore recommended [ref. 51]. The selective noradrenaline-reuptake inhibitor atomoxetine improved daytime sleepiness and cognition in a pilot randomized double-blind study in PD individuals [ref. 253]. Medication for comorbid diseases and other medications, especially hypnotics and sedating antidepressants should be reviewed critically. Ultimately, the MDS recommends to prohibit driving as long as daytime sleepiness persist [ref. 70].
| Recommendation | |
|---|---|
| For treatment of daytime sleepiness in PD, a cause-oriented diagnostic work-up is recommendedDaytime sleepiness due to motor and non-motor complications of PD should be treated by optimizing PD therapyNon-pharmacological therapies such as sleep hygiene, exercise and light therapy should be recommendedPrimary sleep disorders responsible for daytime sleepiness should be diagnosed and treated accordingly. Only in case of persistent daytime sleepiness, pharmacological options (i.e., modafinil) may be considered on an individual basis, though the available evidence is low | |
| Level of consensus: 100%, strong consensus |
What are the effects of nocturnal sleep disturbances on PD individuals and their relatives?
Numerous cross-sectional cohort studies show that insomnia and sleep disorders are associated with a reduced quality of life and an increased disease burden in PD individuals [ref. 105, ref. 118, ref. 243, ref. 264]. An association between sleep disturbances and depression has been found in PD individuals and their partners who may also develop sleep disturbances [ref. 213, ref. 248]. Even mild cases of comorbid SDB are associated with cognitive impairment [ref. 178] and motor performance deteriorates in case of comorbid sleep disorders [ref. 159, ref. 199].
| Recommendation | |
|---|---|
| Sleep disturbances have a pronounced negative impact on PD individuals and their partners, especially regarding motor performance, quality of life, mood and the nocturnal sleep quality of partners. Sleep disorders should be therefore routinely considered in the clinical diagnostic work-up and therapeutic planTherapeutic decisions should not only consider the concerns of PD individuals, but also consequences for their partners and caregivers | |
| Level of consensus: 96,8%, strong consensus |
What are the effects of daytime sleepiness on PD individuals and their relatives?
Increased daytime sleepiness and unintentionally falling asleep impair the ability to drive and thus restrict the individual mobility. Sleep attacks in daily life situations represent a hazard and have a negative impact on quality of life [ref. 6, ref. 38, ref. 214, ref. 262]. A longitudinal cohort study showed that daytime sleepiness increases as the disease progresses [ref. 6]. Although no explicit studies could be found, it is evident that unintended falling asleep, impaired attention and participation due to increased daytime sleepiness inhibit communication and social engangement, implying a bidirectional influence.
| Recommendation | |
|---|---|
| Patients with increased daytime sleepiness are not capable of driving; in addition they suffer impairment in many aspects of communication and social participationThis impacts negatively on their social environment | |
| Level of consensus: 92.6%, consensus |
What is the prognosis of PD individuals with sleep disorders as compared to those without and which sleep disorders impact most?
Apart from the pathophysiological connection between sleep and neurodegeneration, clinical evidence shows that sleep disorders are associated with a faster disease progression [ref. 25, ref. 202, ref. 220, ref. 266]. Especially RBD is associated with a more severe PD course of the disease, earlier motor complications and cognitive decline, indicating that RBD represents a progression marker in PD [ref. 157, ref. 176]. Changes in EEG microstructure during sleep, such as a reduction of delta waves and spindles, are specifically associated with a more rapid cognitive decline [ref. 52]. In the general population, SDB is associated with metabolic complications and cardiovascular disease. In PD individuals, SDB additionally impact on executive function, working memory and motor performance [ref. 56].
| Recommendation | |
|---|---|
| Sleep disorders are associated with more rapid disease progressionRBD in early stages of PD predicts a more severe disease course with earlier cognitive, autonomic and motor complications | |
| Level of consensus: 96.2%, strong consensus |
Discussion
Identifying and treating autonomic failure with a stepwise combination of non-pharmacological and pharmacological options is essential for improving the symptomatic burden of PD individuals and reducing the risk of life-threatening complications such as recurrent urinary tract infections, syncope-related falls, hypertensive emergencies, gastrointestinal occlusions and perforations. The type and time-course of autonomic failure may also contribute to the differential diagnosis of PD from atypical Parkinsonian syndromes like multiple system atrophy, in which an earlier, and more severe autonomic impairment involving multiple domains is frequently observed [ref. 60, ref. 65].
For an effective, etiology-driven pain treatment in PD individuals, it is essential to distinguish between nociceptive, neuropathic and nociplastic pain types by means of detailed history taking and PD-specific scales and questionnaires. It is to be born in mind that multiple pain causes may be present in single PD individuals and that a bidirectional interplay often exists between painful sensations and PD motor symptoms, especially in fluctuating patients. Pain treatment should therefore begin with an adjustment of dopaminergic medications whenever pain is reckoned to depend upon insufficient dopaminergic stimulation and progressively integrate pharmacological measures targeting any identified cause of nociceptive or neuropathic pain, until an adequate symptomatic control is achieved.
The pathophysiology of sleep regulation in PD individuals with its spectrum of clinical manifestations needs to be contextualized in the neurodegenerative process of the disease to deduce diagnostic procedures and appropriate therapeutic steps (Fig. 2). To this end, the interdependence of disturbed nocturnal sleep and diurnal wakefulness with quality of life, disease burden, mood, motor performance, cognition, social interaction, disease progression as well as caregiver burden should be considered. An algorithm for the diagnostic work-up of sleep disturbances and daytime sleepiness in PD is proposed in Fig. 3. The identification of the underlying sleep disorder drives the specific therapeutic approach, which is based on PD-specific measures and also refers to guidelines for other stand-alone disease entities such as RLS, insomnia or SDB.
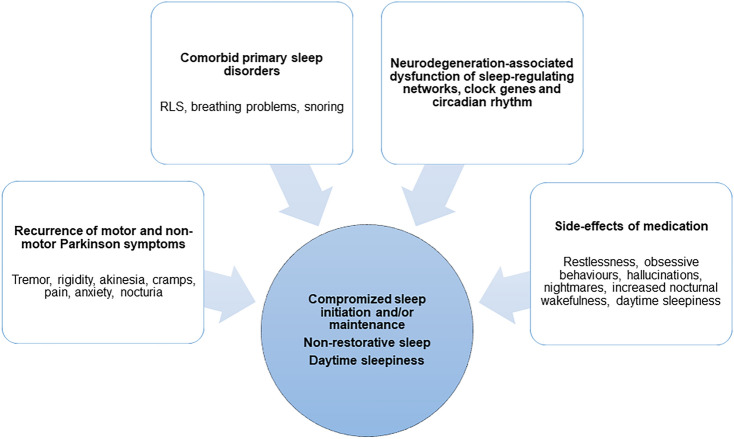
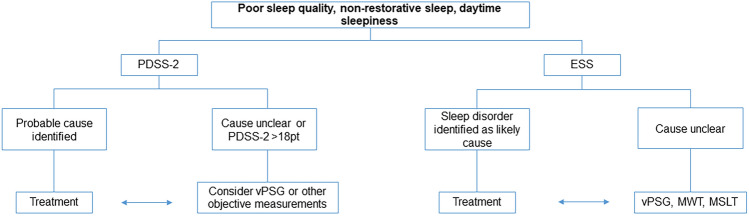
In conclusion, acknowledging the time- and resource constrains [ref. 86] frequently encountered by physicians in everyday practice, especially after COVID-19 pandemic outbreak [ref. 64, ref. 182], yet aiming at maximizing the quality of healthcare provision to individuals living with PD, the 2023 edition of the PD guidelines of the German Society of Neurology provides evidence- and consensus-based recommendations for a practice-oriented and etiology-driven stepwise approach to the diagnosis and treatment of motor and non-motor PD symptoms, including autonomic failure, pain and sleep disturbances. Future trials should ideally close the gaps in evidence for the pharmacological and non-pharmacological treatment of these, still underestimated, disturbances in the specific setting of PD.
References
- P Abrams, L Cardozo, A Wein. The 2nd International Consultation on Incontinence, Research Society (ICI-RS) meeting 2010. Neurourol Urodyn, 2011. [PubMed]
- JH Ahn, M Kim, S Park, W Jang, J Park, E Oh, JW Cho, JS Kim, J Youn. Prolonged-release melatonin in Parkinson’s disease patients with a poor sleep quality: a randomized trial. Parkinsonism Relat Disord, 2020. [PubMed]
- 3.al VAe (2022) Aktualisierte S2k-Leitlinie chronische Obstipation der Deutschen Gesellschaft für Gastroenterologie, Verdauungs- und Stoffwechselkrankheiten (DGVS) und der Deutschen Gesellschaft für Neurogastroenterologie & Motilität (DGNM). In: AWMF (ed)
- 4.Alhasso AA, McKinlay J, Patrick K, Stewart L (2006) Anticholinergic drugs versus non-drug active therapies for overactive bladder syndrome in adults. In: The Cochrane database of systematic reviews, p CD003193
- H Alonso-Navarro, E Garcia-Martin, JAG Agundez, FJ Jimenez-Jimenez. Association between restless legs syndrome and other movement disorders. Neurology, 2019. [PubMed]
- AW Amara, LM Chahine, C Caspell-Garcia, JD Long, C Coffey, B Hogl, A Videnovic, A Iranzo, G Mayer, N Foldvary-Schaefer, R Postuma, W Oertel, S Lasch, K Marek, T Simuni, I Parkinson’s Progression Markers. Longitudinal assessment of excessive daytime sleepiness in early Parkinson’s disease. J Neurol Neurosurg Psychiatry, 2017. [PubMed]
- G Amarenco, SS Ismael, A Even-Schneider, P Raibaut, S Demaille-Wlodyka, B Parratte, J Kerdraon. Urodynamic effect of acute transcutaneous posterior tibial nerve stimulation in overactive bladder. J Urol, 2003. [PubMed]
- A Antonini, A Yegin, C Preda, L Bergmann, W Poewe. Global long-term study on motor and non-motor symptoms and safety of levodopa-carbidopa intestinal gel in routine care of advanced Parkinson’s disease patients; 12-month interim outcomes. Parkinsonism Relat Disord, 2015. [PubMed]
- D Arand, M Bonnet, T Hurwitz, M Mitler, R Rosa, RB Sangal. The clinical use of the MSLT and MWT. Sleep, 2005. [PubMed]
- AC Arnold, LE Okamoto, A Gamboa, BK Black, SR Raj, F Elijovich, D Robertson, CA Shibao, I Biaggioni. Mineralocorticoid receptor activation contributes to the supine hypertension of autonomic failure. Hypertension, 2016. [PubMed]
- AC Arnold, LE Okamoto, A Gamboa, C Shibao, SR Raj, D Robertson, I Biaggioni. Angiotensin II, independent of plasma renin activity, contributes to the hypertension of autonomic failure. Hypertension, 2013. [PubMed]
- 12.Blok DC-DB, Del Popolo G et al (2022) EAU guidelines on neuro-urology
- P Bargiotas, ML Lachenmayer, DR Schreier, J Mathis, CL Bassetti. Sleepiness and sleepiness perception in patients with Parkinson’s disease: a clinical and electrophysiological study. Sleep, 2019. [PubMed]
- M Barichella, C Pacchetti, C Bolliri, E Cassani, L Iorio, C Pusani, G Pinelli, G Privitera, I Cesari, SA Faierman, R Caccialanza, G Pezzoli, E Cereda. Probiotics and prebiotic fiber for constipation associated with Parkinson disease: an RCT. Neurology, 2016. [PubMed]
- P Barone, A Antonini, C Colosimo, R Marconi, L Morgante, TP Avarello, E Bottacchi, A Cannas, G Ceravolo, R Ceravolo, G Cicarelli, RM Gaglio, RM Giglia, F Iemolo, M Manfredi, G Meco, A Nicoletti, M Pederzoli, A Petrone, A Pisani, FE Pontieri, R Quatrale, S Ramat, R Scala, G Volpe, S Zappulla, AR Bentivoglio, F Stocchi, G Trianni, PD Dotto. The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov Disord, 2009. [PubMed]
- P Barone, W Poewe, S Albrecht, C Debieuvre, D Massey, O Rascol, E Tolosa, D Weintraub. Pramipexole for the treatment of depressive symptoms in patients with Parkinson’s disease: a randomised, double-blind, placebo-controlled trial. Lancet Neurol, 2010. [PubMed]
- L Bartolomei, A Pastore, L Meligrana, E Sanson, N Bonetto, GM Minicuci, SZ Marsala, T Mesiano, L Bragagnolo, A Antonini. Relevance of sleep quality on caregiver burden in Parkinson’s disease. Neurol Sci, 2018. [PubMed]
- SR Benbadis, E Mascha, MC Perry, BR Wolgamuth, LA Smolley, DS Dinner. Association between the Epworth sleepiness scale and the multiple sleep latency test in a clinical population. Ann Intern Med, 1999. [PubMed]
- D Berg, RB Postuma, CH Adler, BR Bloem, P Chan, B Dubois, T Gasser, CG Goetz, G Halliday, L Joseph, AE Lang, I Liepelt-Scarfone, I Litvan, K Marek, J Obeso, W Oertel, CW Olanow, W Poewe, M Stern, G Deuschl. MDS research criteria for prodromal Parkinson’s disease. Mov Disord, 2015. [PubMed]
- BA Bernard, LV Metman, L Levine, B Ouyang, S Leurgans, CG Goetz. Sildenafil in the treatment of erectile dysfunction in Parkinson’s disease. Mov Disord Clin Pract, 2017. [PubMed]
- K Bhalsing, K Suresh, UB Muthane, PK Pal. Prevalence and profile of Restless Legs Syndrome in Parkinson’s disease and other neurodegenerative disorders: a case–control study. Parkinsonism Relat Disord, 2013. [PubMed]
- I Biaggioni, R Freeman, CJ Mathias, P Low, LA Hewitt, H Kaufmann, I Droxidopa. Randomized withdrawal study of patients with symptomatic neurogenic orthostatic hypotension responsive to droxidopa. Hypertension, 2015. [PubMed]
- I Biaggioni, D Robertson. Endogenous restoration of noradrenaline by precursor therapy in dopamine-beta-hydroxylase deficiency. Lancet, 1987. [PubMed]
- KA Bjornara, E Dietrichs, M Toft. Clinical features associated with sleep disturbances in Parkinson’s disease. Clin Neurol Neurosurg, 2014. [PubMed]
- NI Bohnen, MTM Hu. Sleep disturbance as potential risk and progression factor for Parkinson’s disease. J Parkinsons Dis, 2019. [PubMed]
- M Braun, G Wassmer, T Klotz, B Reifenrath, M Mathers, U Engelmann. Epidemiology of erectile dysfunction: results of the ‘Cologne Male Survey’. Int J Impot Res, 2000. [PubMed]
- M Brignole, A Moya, FJ de Lange, JC Deharo, PM Elliott, A Fanciulli, A Fedorowski, R Furlan, RA Kenny, A Martin, V Probst, MJ Reed, CP Rice, R Sutton, A Ungar, JG van Dijk. 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J, 2018. [PubMed]
- V Bruno, ME Freitas, D Mancini, JP Lui, J Miyasaki, SH Fox. Botulinum toxin type A for pain in advanced Parkinson’s disease. Can J Neurol Sci, 2018. [PubMed]
- C Buhmann, J Kassubek, WH Jost. Management of pain in Parkinson’s disease. J Parkinsons Dis, 2020. [PubMed]
- C Buhmann, N Wrobel, W Grashorn, O Fruendt, K Wesemann, S Diedrich, U Bingel. Pain in Parkinson disease: a cross-sectional survey of its prevalence, specifics, and therapy. J Neurol, 2017. [PubMed]
- DJ Buysse, CF Reynolds, TH Monk, SR Berman, DJ Kupfer. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res, 1989. [PubMed]
- SJ Cai, R Chen, YL Zhang, KP Xiong, YX Lian, J Li, JC Shen, CF Liu. Correlation of Epworth Sleepiness Scale with multiple sleep latency test and its diagnostic accuracy in assessing excessive daytime sleepiness in patients with obstructive sleep apnea hypopnea syndrome. Chin Med J (Engl), 2013. [PubMed]
- S Canbaz Kabay, S Kabay, E Mestan, M Cetiner, S Ayas, M Sevim, H Ozden, HO Karaman. Long term sustained therapeutic effects of percutaneous posterior tibial nerve stimulation treatment of neurogenic overactive bladder in multiple sclerosis patients: 12-months results. Neurourol Urodyn, 2017. [PubMed]
- MA Carskadon, WC Dement, MM Mitler, T Roth, PR Westbrook, S Keenan. Guidelines for the multiple sleep latency test (MSLT): a standard measure of sleepiness. Sleep, 1986. [PubMed]
- MJ Carvalho, AH van den Meiracker, F Boomsma, AJ Man in’t Veld, J Freitas, O Costa, AF de Freitas. Improved orthostatic tolerance in familial amyloidotic polyneuropathy with unnatural noradrenaline precursor l-threo-3,4-dihydroxyphenylserine. J Auton Nerv Syst, 1997. [PubMed]
- C Cattaneo, P Barone, E Bonizzoni, M Sardina. Effects of safinamide on pain in fluctuating Parkinson’s disease patients: a post-hoc analysis. J Parkinsons Dis, 2017. [PubMed]
- C Cattaneo, J Kulisevsky, V Tubazio, P Castellani. Long-term efficacy of safinamide on Parkinson’s disease chronic pain. Adv Ther, 2018. [PubMed]
- LM Chahine, AW Amara, A Videnovic. A systematic review of the literature on disorders of sleep and wakefulness in Parkinson’s disease from 2005 to 2015. Sleep Med Rev, 2017. [PubMed]
- KR Chaudhuri, A Antonini, R Pahwa, P Odin, N Titova, S Thakkar, SJ Snedecor, S Hegde, A Alobaidi, JC Parra, C Zadikoff, L Bergmann, DG Standaert. Effects of levodopa-carbidopa intestinal gel on dyskinesia and non-motor symptoms including sleep: results from a meta-analysis with 24-month follow-up. J Parkinsons Dis, 2022. [PubMed]
- KR Chaudhuri, S Pal, A DiMarco, C Whately-Smith, K Bridgman, R Mathew, FR Pezzela, A Forbes, B Hogl, C Trenkwalder. The Parkinson’s disease sleep scale: a new instrument for assessing sleep and nocturnal disability in Parkinson’s disease. J Neurol Neurosurg Psychiatry, 2002. [PubMed]
- KR Chaudhuri, A Rizos, C Trenkwalder, O Rascol, S Pal, D Martino, C Carroll, D Paviour, C Falup-Pecurariu, B Kessel, M Silverdale, A Todorova, A Sauerbier, P Odin, A Antonini, P Martinez-Martin. King’s Parkinson’s disease pain scale, the first scale for pain in PD: an international validation. Mov Disord, 2015. [PubMed]
- B Cheng, S Huang, Q Huang, Z Zhou, Y Bao. The efficacy and safety of medication for treating overactive bladder in patients with Parkinson’s disease: a meta-analysis and systematic review of randomized double-blind placebo-controlled trials. Int Urogynecol J, 2023. [PubMed]
- SY Cho, SJ Jeong, S Lee, J Kim, SH Lee, MS Choo, SJ Oh. Mirabegron for treatment of overactive bladder symptoms in patients with Parkinson’s disease: a double-blind, randomized placebo-controlled trial (Parkinson’s Disease Overactive bladder Mirabegron, PaDoMi Study). Neurourol Urodyn, 2021. [PubMed]
- N Consensus Development Conference. Impotence. NIH Consens Statement, 1992
- J Corcos, J Gajewski, D Heritz, A Patrick, I Reid, E Schick, L Stothers, A Canadian Urological. Canadian Urological Association guidelines on urinary incontinence. Can J Urol, 2006. [PubMed]
- J Cristini, M Weiss, HB De Las, A Medina-Rincon, A Dagher, RB Postuma, R Huber, J Doyon, P Rosa-Neto, J Carrier, AW Amara, M Roig. The effects of exercise on sleep quality in persons with Parkinson’s disease: a systematic review with meta-analysis. Sleep Med Rev, 2021. [PubMed]
- F Cruz, S Herschorn, P Aliotta, M Brin, C Thompson, W Lam, G Daniell, J Heesakkers, C Haag-Molkenteller. Efficacy and safety of onabotulinumtoxin A in patients with urinary incontinence due to neurogenic detrusor overactivity: a randomised, double-blind, placebo-controlled trial. Eur Urol, 2011. [PubMed]
- VC De Cock, P Dodet, S Leu-Semenescu, C Aerts, G Castelnovo, B Abril, S Drapier, H Olivet, AG Corbille, L Leclair-Visonneau, M Sallansonnet-Froment, M Lebouteux, M Anheim, E Ruppert, N Vitello, A Eusebio, I Lambert, A Marques, ML Fantini, D Devos, C Monaca, N Benard-Serre, S Lacombe, M Vidailhet, I Arnulf, M Doulazmi, E Roze. Safety and efficacy of subcutaneous night-time only apomorphine infusion to treat insomnia in patients with Parkinson’s disease (APOMORPHEE): a multicentre, randomised, controlled, double-blind crossover study. Lancet Neurol, 2022. [PubMed]
- O De Fabregues, A Ferre, O Romero, M Quintana, J Alvarez-Sabin. Sleep quality and levodopa intestinal gel infusion in Parkinson’s disease: a pilot study. Parkinsons Dis, 2018. [PubMed]
- DL Delgado-Lara, GV Gonzalez-Enriquez, BM Torres-Mendoza, H Gonzalez-Usigli, J Cardenas-Bedoya, MA Macias-Islas, AC de la Rosa, A Jimenez-Delgado, F Pacheco-Moises, JA Cruz-Serrano, GG Ortiz. Effect of melatonin administration on the PER1 and BMAL1 clock genes in patients with Parkinson’s disease. Biomed Pharmacother, 2020. [PubMed]
- D Devos, P Krystkowiak, F Clement, K Dujardin, O Cottencin, N Waucquier, K Ajebbar, B Thielemans, M Kroumova, A Duhamel, A Destee, R Bordet, L Defebvre. Improvement of gait by chronic, high doses of methylphenidate in patients with advanced Parkinson’s disease. J Neurol Neurosurg Psychiatry, 2007. [PubMed]
- F Dijkstra, I de Volder, M Viaene, P Cras, D Crosiers. Polysomnographic predictors of sleep, motor, and cognitive dysfunction progression in Parkinson’s disease. Curr Neurol Neurosci Rep, 2022. [PubMed]
- R Djaldetti, S Yust-Katz, V Kolianov, E Melamed, R Dabby. The effect of duloxetine on primary pain symptoms in Parkinson disease. Clin Neuropharmacol, 2007. [PubMed]
- E Dula, W Keating, PF Siami, A Edmonds, J O’Neil, S Buttler. Efficacy and safety of fixed-dose and dose-optimization regimens of sublingual apomorphine versus placebo in men with erectile dysfunction The Apomorphine Study Group. Urology, 2000. [PubMed]
- E El Helou, C Labaki, R Chebel, J El Helou, G Abi Tayeh, G Jalkh, E Nemr. The use of mirabegron in neurogenic bladder: a systematic review. World J Urol, 2020. [PubMed]
- M Elfil, EI Bahbah, MM Attia, M Eldokmak, BB Koo. Impact of obstructive sleep apnea on cognitive and motor functions in Parkinson’s disease. Mov Disord, 2021. [PubMed]
- S Eschlbock, G Wenning, A Fanciulli. Evidence-based treatment of neurogenic orthostatic hypotension and related symptoms. J Neural Transm (Vienna), 2017. [PubMed]
- A Fanciulli, N Campese, GK Wenning. The Schellong test: detecting orthostatic blood pressure and heart rate changes in German-speaking countries. Clin Auton Res, 2019. [PubMed]
- A Fanciulli, G Gobel, JP Ndayisaba, R Granata, S Duerr, S Strano, C Colosimo, W Poewe, FE Pontieri, GK Wenning. Supine hypertension in Parkinson’s disease and multiple system atrophy. Clin Auton Res, 2016. [PubMed]
- A Fanciulli, G Goebel, G Lazzeri, C Scherfler, ER Gizewski, R Granata, G Kiss, S Strano, C Colosimo, FE Pontieri, H Kaufmann, K Seppi, W Poewe, GK Wenning. Early distinction of Parkinson-variant multiple system atrophy from Parkinson’s disease. Mov Disord, 2019. [PubMed]
- A Fanciulli, G Goebel, B Metzler, F Sprenger, W Poewe, GK Wenning, K Seppi. Elastic abdominal binders attenuate orthostatic hypotension in Parkinson’s disease. Mov Disord Clin Pract, 2016. [PubMed]
- A Fanciulli, J Jordan, I Biaggioni, G Calandra-Buonaura, WP Cheshire, P Cortelli, S Eschlboeck, G Grassi, MJ Hilz, H Kaufmann, H Lahrmann, G Mancia, G Mayer, L Norcliffe-Kaufmann, A Pavy-Le Traon, SR Raj, D Robertson, I Rocha, W Struhal, R Thijs, KP Tsioufis, JG van Dijk, GK Wenning. Consensus statement on the definition of neurogenic supine hypertension in cardiovascular autonomic failure by the American Autonomic Society (AAS) and the European Federation of Autonomic Societies (EFAS): endorsed by the European Academy of Neurology (EAN) and the European Society of Hypertension (ESH). Clin Auton Res, 2018. [PubMed]
- A Fanciulli, K Kerer, F Leys, K Seppi, H Kaufmann, L Norcliffe-Kaufmann, GK Wenning. Validation of the neurogenic orthostatic hypotension ratio with active standing. Ann Neurol, 2020. [PubMed]
- A Fanciulli, F Leys, M Krbot Skoric, DR Carneiro, G Calandra-Buonaura, J Camaradou, G Chiaro, P Cortelli, C Falup-Pecurariu, R Granata, P Guaraldi, R Helbok, MJ Hilz, V Iodice, J Jordan, ECA Kaal, A Kamondi, A Pavy Le Traon, I Rocha, J Sellner, JM Senard, A Terkelsen, GK Wenning, E Moro, T Berger, RD Thijs, W Struhal, M Habek. Impact of the COVID-19 pandemic on clinical autonomic practice in Europe: a survey of the European Academy of Neurology (EAN) and the European Federation of Autonomic Societies (EFAS). Eur J Neurol, 2023
- A Fanciulli, I Stankovic, O Avraham, DR Carneiro, M Jecmenica Lukic, A Ezra, F Leys, G Goebel, F Krismer, I Petrovic, M Svetel, K Seppi, V Kostic, N Giladi, W Poewe, GK Wenning, T Gurevich. Early screening for the Parkinson variant of multiple system atrophy: a 6-item score. Mov Disord Clin Pract, 2024. [PubMed]
- A Fanciulli, S Strano, JP Ndayisaba, G Goebel, L Gioffre, M Rizzo, C Colosimo, C Caltagirone, W Poewe, GK Wenning, FE Pontieri. Detecting nocturnal hypertension in Parkinson’s disease and multiple system atrophy: proposal of a decision-support algorithm. J Neurol, 2014. [PubMed]
- A Fanciulli, GK Wenning. Autonomic failure: a neglected presentation of Parkinson’s disease. Lancet Neurol, 2021. [PubMed]
- MP Feeney, D Bega, BM Kluger, AJ Stoessl, CM Evers, R De Leon, JC Beck. Weeding through the haze: a survey on cannabis use among people living with Parkinson’s disease in the US. NPJ Parkinsons Dis, 2021. [PubMed]
- G Fernandez-Pajarin, A Sesar, B Ares, A Castro. Evaluating the efficacy of nocturnal continuous subcutaneous apomorphine infusion in sleep disorders in advanced Parkinson’s disease: the APO-NIGHT STudy. J Parkinsons Dis, 2016. [PubMed]
- JJ Ferreira, R Katzenschlager, BR Bloem, U Bonuccelli, D Burn, G Deuschl, E Dietrichs, G Fabbrini, A Friedman, P Kanovsky, V Kostic, A Nieuwboer, P Odin, W Poewe, O Rascol, C Sampaio, M Schupbach, E Tolosa, C Trenkwalder, A Schapira, A Berardelli, WH Oertel. Summary of the recommendations of the EFNS/MDS-ES review on therapeutic management of Parkinson’s disease. Eur J Neurol, 2013. [PubMed]
- JJ Figueroa, W Singer, P Sandroni, DM Sletten, TL Gehrking, JA Gehrking, P Low, JR Basford. Effects of patient-controlled abdominal compression on standing systolic blood pressure in adults with orthostatic hypotension. Arch Phys Med Rehabil, 2015. [PubMed]
- SY Fong, CK Ho, YK Wing. Comparing MSLT and ESS in the measurement of excessive daytime sleepiness in obstructive sleep apnoea syndrome. J Psychosom Res, 2005. [PubMed]
- B Ford. Pain in Parkinson’s disease. Mov Disord, 2010. [PubMed]
- B Ford. Pain in Parkinson’s disease. Clin Neurosci, 1998. [PubMed]
- FM Fouad-Tarazi, M Okabe, H Goren. Alpha sympathomimetic treatment of autonomic insufficiency with orthostatic hypotension. Am J Med, 1995. [PubMed]
- R Freeman, L Landsberg, J Young. The treatment of neurogenic orthostatic hypotension with 3,4-DL-threo-dihydroxyphenylserine: a randomized, placebo-controlled, crossover trial. Neurology, 1999. [PubMed]
- R Freeman, W Wieling, FB Axelrod, DG Benditt, E Benarroch, I Biaggioni, WP Cheshire, T Chelimsky, P Cortelli, CH Gibbons, DS Goldstein, R Hainsworth, MJ Hilz, G Jacob, H Kaufmann, J Jordan, LA Lipsitz, BD Levine, PA Low, C Mathias, SR Raj, D Robertson, P Sandroni, I Schatz, R Schondorff, JM Stewart, JG van Dijk. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res, 2011. [PubMed]
- S Frucht, JD Rogers, PE Greene, MF Gordon, S Fahn. Falling asleep at the wheel: motor vehicle mishaps in persons taking pramipexole and ropinirole. Neurology, 1999. [PubMed]
- SJ Frucht, PE Greene, S Fahn. Sleep episodes in Parkinson’s disease: a wake-up call. Mov Disord, 2000. [PubMed]
- J Gao, X Huang, Y Park, A Hollenbeck, A Blair, A Schatzkin, H Chen. Daytime napping, nighttime sleeping, and Parkinson disease. Am J Epidemiol, 2011. [PubMed]
- R Geoffrion. No. 353-treatments for overactive bladder: focus on pharmacotherapy—an addendum. J Obstet Gynaecol Can, 2017. [PubMed]
- C Geroin, IA Di Vico, G Squintani, A Segatti, T Bovi, M Tinazzi. Effects of safinamide on pain in Parkinson’s disease with motor fluctuations: an exploratory study. J Neural Transm (Vienna), 2020. [PubMed]
- A Giannantoni, A Rossi, E Mearini, M Del Zingaro, M Porena, A Berardelli. Botulinum toxin A for overactive bladder and detrusor muscle overactivity in patients with Parkinson’s disease and multiple system atrophy. J Urol, 2009. [PubMed]
- M Gilat, NS Marshall, D Testelmans, B Buyse, SJG Lewis. A critical review of the pharmacological treatment of REM sleep behavior disorder in adults: time for more and larger randomized placebo-controlled trials. J Neurol, 2022. [PubMed]
- MD Gjerstad, OB Tysnes, JP Larsen. Increased risk of leg motor restlessness but not RLS in early Parkinson disease. Neurology, 2011. [PubMed]
- M Habek, F Leys, M Krbot Skoric, D Reis Carneiro, G Calandra-Buonaura, J Camaradou, G Chiaro, P Cortelli, C Falup-Pecurariu, R Granata, P Guaraldi, R Helbok, MJ Hilz, V Iodice, J Jordan, ECA Kaal, A Kamondi, A Pavy Le Traon, I Rocha, J Sellner, JM Senard, A Terkelsen, GK Wenning, T Berger, RD Thijs, W Struhal, A Fanciulli. Clinical autonomic nervous system laboratories in Europe: a joint survey of the European Academy of Neurology and the European Federation of Autonomic Societies. Eur J Neurol, 2022. [PubMed]
- 87.Haensch (2018) Diagnostik und Therapie der erektilen Dysfunktion, S1-Leitlinie. In:Deutsche Gesellschaft für Neurologie (Hrsg.)
- 88.Haensch C-A (2020) Diagnostik und Therapie von neurogenen Blasen-störungen, S1-Leitlinie. In:Deutsche Gesellschaft für Neurologie
- RA Hauser, LA Hewitt, S Isaacson. Droxidopa in patients with neurogenic orthostatic hypotension associated with Parkinson’s disease (NOH306A). J Parkinsons Dis, 2014. [PubMed]
- RA Hauser, S Isaacson, JP Lisk, LA Hewitt, G Rowse. Droxidopa for the short-term treatment of symptomatic neurogenic orthostatic hypotension in Parkinson’s disease (nOH306B). Mov Disord, 2015. [PubMed]
- 91.Heidbreder A (2022) Restless Legs Syndrom, S2k Leitlinie. In: Schlafmedizin DGfNuDGfSu (ed) Leitlinien für Diagnostik und Therapie in der Neurologie
- B Hogl, I Arnulf, C Comella, J Ferreira, A Iranzo, B Tilley, C Trenkwalder, W Poewe, O Rascol, C Sampaio, GT Stebbins, A Schrag, CG Goetz. Scales to assess sleep impairment in Parkinson’s disease: critique and recommendations. Mov Disord, 2010. [PubMed]
- 93.Höglinger G TCea (2023) Parkinson-Krankheit, S2k-Leitlinie. In: Neurologie DGf (ed) Leitlinien für Diagnostik und Therapie in der Neurologie
- S Hood, S Amir. Neurodegeneration and the circadian clock. Front Aging Neurosci, 2017. [PubMed]
- Y Huang, S Hennig, I Fietze, T Penzel, C Veauthier. The Psychomotor vigilance test compared to a divided attention steering simulation in patients with moderate or severe obstructive sleep apnea. Nat Sci Sleep, 2020. [PubMed]
- IF Hussain, CM Brady, MJ Swinn, CJ Mathias, CJ Fowler. Treatment of erectile dysfunction with sildenafil citrate (Viagra) in parkinsonism due to Parkinson’s disease or multiple system atrophy with observations on orthostatic hypotension. J Neurol Neurosurg Psychiatry, 2001. [PubMed]
- S Isaacson, S Vernino, A Ziemann, GJ Rowse, U Kalu, WB White. Long-term safety of droxidopa in patients with symptomatic neurogenic orthostatic hypotension. J Am Soc Hypertens (JASH), 2016. [PubMed]
- J Jankovic, JL Gilden, BC Hiner, H Kaufmann, DC Brown, CH Coghlan, M Rubin, FM Fouad-Tarazi. Neurogenic orthostatic hypotension: a double-blind, placebo-controlled study with midodrine. Am J Med, 1993. [PubMed]
- MW Johns. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep, 1991. [PubMed]
- J Jordan, A Fanciulli, J Tank, G Calandra-Buonaura, WP Cheshire, P Cortelli, S Eschlboeck, G Grassi, MJ Hilz, H Kaufmann, H Lahrmann, G Mancia, G Mayer, L Norcliffe-Kaufmann, A Pavy-Le Traon, SR Raj, D Robertson, I Rocha, H Reuter, W Struhal, RD Thijs, KP Tsioufis, J Gert van Dijk, GK Wenning, I Biaggioni. Management of supine hypertension in patients with neurogenic orthostatic hypotension: scientific statement of the American Autonomic Society, European Federation of Autonomic Societies, and the European Society of Hypertension. J Hypertens, 2019. [PubMed]
- J Jordan, JR Shannon, B Pohar, SY Paranjape, D Robertson, RM Robertson, I Biaggioni. Contrasting effects of vasodilators on blood pressure and sodium balance in the hypertension of autonomic failure. J Am Soc Nephrol, 1999. [PubMed]
- WH Jost. Gastrointestinal motility problems in patients with Parkinson’s disease. Effects of antiparkinsonian treatment and guidelines for management. Drugs Aging, 1997. [PubMed]
- WH Jost, A Rizos, P Odin, M Lohle, A Storch. King’s Parkinson’s disease pain scale: intercultural adaptation in the German language. Nervenarzt, 2018. [PubMed]
- T Kachi, S Iwase, T Mano, M Saito, M Kunimoto, I Sobue. Effect of l-threo-3,4-dihydroxyphenylserine on muscle sympathetic nerve activities in Shy-Drager syndrome. Neurology, 1988. [PubMed]
- KH Karlsen, JP Larsen, E Tandberg, JG Maeland. Influence of clinical and demographic variables on quality of life in patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry, 1999. [PubMed]
- V Karnik, N Farcy, C Zamorano, V Bruno. Current status of pain management in Parkinson’s disease. Can J Neurol Sci, 2020. [PubMed]
- J Karsten, LA Hagenauw, J Kamphuis, M Lancel. Low doses of mirtazapine or quetiapine for transient insomnia: a randomised, double-blind, cross-over, placebo-controlled trial. J Psychopharmacol, 2017. [PubMed]
- K Kassolik, W Andrzejewski, I Wilk, M Brzozowski, K Voyce, E Jaworska-Krawiecka, B Nowak, D Kurpas. The effectiveness of massage based on the tensegrity principle compared with classical abdominal massage performed on patients with constipation. Arch Gerontol Geriatr, 2015. [PubMed]
- J Kassubek, KR Chaudhuri, T Zesiewicz, E Surmann, B Boroojerdi, K Moran, L Ghys, C Trenkwalder. Rotigotine transdermal system and evaluation of pain in patients with Parkinson’s disease: a post hoc analysis of the RECOVER study. BMC Neurol, 2014. [PubMed]
- H Kaufmann, T Brannan, L Krakoff, MD Yahr, J Mandeli. Treatment of orthostatic hypotension due to autonomic failure with a peripheral alpha-adrenergic agonist (midodrine). Neurology, 1988. [PubMed]
- H Kaufmann, R Freeman, I Biaggioni, P Low, S Pedder, LA Hewitt, J Mauney, M Feirtag, CJ Mathias. Droxidopa for neurogenic orthostatic hypotension: a randomized, placebo-controlled, phase 3 trial. Neurology, 2014. [PubMed]
- H Kaufmann, L Norcliffe-Kaufmann, JA Palma. Baroreflex dysfunction. N Engl J Med, 2020. [PubMed]
- H Kaufmann, D Saadia, A Voustianiouk, DS Goldstein, C Holmes, MD Yahr, R Nardin, R Freeman. Norepinephrine precursor therapy in neurogenic orthostatic hypotension. Circulation, 2003. [PubMed]
- GG Kay, U Ebinger. Preserving cognitive function for patients with overactive bladder: evidence for a differential effect with darifenacin. Int J Clin Pract, 2008. [PubMed]
- C Kelleher, Z Hakimi, R Zur, E Siddiqui, K Maman, S Aballea, J Nazir, C Chapple. Efficacy and tolerability of mirabegron compared with antimuscarinic monotherapy or combination therapies for overactive bladder: a systematic review and network meta-analysis. Eur Urol, 2018. [PubMed]
- JE Kenna, MC Bakeberg, AM Gorecki, A Chin Yen Tay, S Winter, FL Mastaglia, RS Anderton. Characterization of gastrointestinal symptom type and severity in Parkinson’s disease: a case–control study in an Australian cohort. Mov Disord Clin Pract, 2021. [PubMed]
- C Konstantinidis, Z Kratiras, M Samarinas, K Skriapas. Optimal bladder diary duration for patients with suprapontine neurogenic lower urinary tract dysfunction. Int Braz J Urol, 2016. [PubMed]
- F Lauretani, C Testa, M Salvi, I Zucchini, F Giallauria, M Maggio. Clinical evaluation of sleep disorders in Parkinson’s disease. Brain Sci, 2023. [PubMed]
- H Lee-Robichaud, K Thomas, J Morgan, RL Nelson. Lactulose versus polyethylene glycol for chronic constipation. Cochrane Database Syst Rev, 2010. [DOI | PubMed]
- AH Lee, D Weintraub. Psychosis in Parkinson’s disease without dementia: common and comorbid with other non-motor symptoms. Mov Disord, 2012. [PubMed]
- AJ Lees, NA Blackburn, VL Campbell. The nighttime problems of Parkinson’s disease. Clin Neuropharmacol, 1988. [PubMed]
- FF Li, YS Cui, R Yan, SS Cao, T Feng. Prevalence of lower urinary tract symptoms, urinary incontinence and retention in Parkinson’s disease: a systematic review and meta-analysis. Front Aging Neurosci, 2022. [PubMed]
- C Liguori, NB Mercuri, M Albanese, E Olivola, A Stefani, M Pierantozzi. Daytime sleepiness may be an independent symptom unrelated to sleep quality in Parkinson’s disease. J Neurol, 2019. [PubMed]
- CH Lin, RM Wu, HY Chang, YT Chiang, HH Lin. Preceding pain symptoms and Parkinson’s disease: a nationwide population-based cohort study. Eur J Neurol, 2013. [PubMed]
- A Lipp, J Tank, G Franke, G Arnold, FC Luft, J Jordan. Osmosensitive mechanisms contribute to the water drinking-induced pressor response in humans. Neurology, 2005. [PubMed]
- H Liu, J Li, X Wang, J Huang, T Wang, Z Lin, N Xiong. Excessive daytime sleepiness in Parkinson’s disease. Nat Sci Sleep, 2022. [PubMed]
- G Lombardi, S Musco, TM Kessler, V Li Marzi, M Lanciotti, G Del Popolo. Management of sexual dysfunction due to central nervous system disorders: a systematic review. BJU Int, 2015. [PubMed]
- I Lotan, TA Treves, Y Roditi, R Djaldetti. Cannabis (medical marijuana) treatment for motor and non-motor symptoms of Parkinson disease: an open-label observational study. Clin Neuropharmacol, 2014. [PubMed]
- PA Low, JL Gilden, R Freeman, KN Sheng, MA McElligott. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. a randomized, double-blind multicenter study. Midodrine Study Group. JAMA, 1997. [PubMed]
- M Lubomski, RL Davis, CM Sue. Health-related quality of life for Parkinson’s disease patients and their caregivers. J Mov Disord, 2021. [PubMed]
- P Luthra, M Camilleri, NE Burr, EMM Quigley, CJ Black, AC Ford. Efficacy of drugs in chronic idiopathic constipation: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol, 2019. [PubMed]
- JE Maglione, L Liu, AB Neikrug, T Poon, L Natarajan, J Calderon, JA Avanzino, J Corey-Bloom, BW Palmer, JS Loredo, S Ancoli-Israel. Actigraphy for the assessment of sleep measures in Parkinson’s disease. Sleep, 2013. [PubMed]
- AJ Man in’t Veld, F Boomsma, AH van den Meiracker, MA Schalekamp. Effect of unnatural noradrenaline precursor on sympathetic control and orthostatic hypotension in dopamine-beta-hydroxylase deficiency. Lancet, 1987. [PubMed]
- S Mantovani, SS Smith, R Gordon, JD O’Sullivan. An overview of sleep and circadian dysfunction in Parkinson’s disease. J Sleep Res, 2018. [PubMed]
- A Marques, N Attal, D Bouhassira, X Moisset, N Cantagrel, O Rascol, F Durif, C Brefel-Courbon. How to diagnose parkinsonian central pain?. Parkinsonism Relat Disord, 2019. [PubMed]
- P Martinez-Martin, E Cubo-Delgado, M Aguilar-Barbera, A Bergareche, S Escalante, A Rojo, J Campdelacreu, B Frades-Payo, S Arroyo, E Grupo. A pilot study on a specific measure for sleep disorders in Parkinson’s disease: SCOPA-Sleep. Rev Neurol, 2006. [PubMed]
- P Martinez-Martin, J Manuel Rojo-Abuin, A Rizos, C Rodriguez-Blazquez, C Trenkwalder, L Perkins, A Sauerbier, P Odin, A Antonini, KR Chaudhuri, E Kpps. Distribution and impact on quality of life of the pain modalities assessed by the King’s Parkinson’s disease pain scale. NPJ Parkinsons Dis, 2017. [PubMed]
- P Martinez-Martin, M Visser, C Rodriguez-Blazquez, J Marinus, KR Chaudhuri, JJ van Hilten. SCOPA-sleep and PDSS: two scales for assessment of sleep disorder in Parkinson’s disease. Mov Disord, 2008. [PubMed]
- JK Martino, CB Freelance, GL Willis. The effect of light exposure on insomnia and nocturnal movement in Parkinson’s disease: an open label, retrospective, longitudinal study. Sleep Med, 2018. [PubMed]
- CJ Mathias, E Holly, E Armstrong, M Shareef, R Bannister. The influence of food on postural hypotension in three groups with chronic autonomic failure—clinical and therapeutic implications. J Neurol Neurosurg Psychiatry, 1991. [PubMed]
- CJ Mathias, JM Senard, S Braune, L Watson, A Aragishi, JE Keeling, MD Taylor. L-threo-dihydroxyphenylserine (l-threo-DOPS; droxidopa) in the management of neurogenic orthostatic hypotension: a multi-national, multi-center, dose-ranging study in multiple system atrophy and pure autonomic failure. Clin Auton Res, 2001. [PubMed]
- S Matsubara, Y Sawa, H Yokoji, M Takamori. Shy-Drager syndrome. Effect of fludrocortisone and l-threo-3,4-dihydroxyphenylserine on the blood pressure and regional cerebral blood flow. J Neurol Neurosurg Psychiatry, 1990. [PubMed]
- 143.Mayer G (2020) Insomnie bei neurologischen Erkrankungen, S2k Leitlinie. In: Neurologie DGf (ed) Leitlinien für Diagnostik und Therapie in der Neurologie
- 144.Mayer G (2017) S3-Leitlinie Nicht erholsamer Schlaf/Schlafstörungen—Kapitel „Schlafbezogene Atmungsstörungen“. In: Schlafmedizin DGfSu (ed)
- D McClurg, S Hagen, K Jamieson, L Dickinson, L Paul, A Cunnington. Abdominal massage for the alleviation of symptoms of constipation in people with Parkinson’s: a randomised controlled pilot study. Age Ageing, 2016. [PubMed]
- F Mearin, BE Lacy, L Chang, WD Chey, AJ Lembo, M Simren, R Spiller. Bowel disorders. Gastroenterology, 2016
- MM Mitler, KS Gujavarty, CP Browman. Maintenance of wakefulness test: a polysomnographic technique for evaluation treatment efficacy in patients with excessive somnolence. Electroencephalogr Clin Neurophysiol, 1982. [PubMed]
- RA Moore, S Derry, HJ McQuay. Indirect comparison of interventions using published randomised trials: systematic review of PDE-5 inhibitors for erectile dysfunction. BMC Urol, 2005. [PubMed]
- G Mostile, J Jankovic. Treatment of dysautonomia associated with Parkinson’s disease. Parkinsonism Relat Disord, 2009. [PubMed]
- M Moussa, MA Chakra, B Dabboucy, Y Fares, A Dellis, A Papatsoris. The safety and effectiveness of mirabegron in Parkinson’s disease patients with overactive bladder: a randomized controlled trial. Scand J Urol, 2022. [PubMed]
- ML Muntean, H Benes, F Sixel-Doring, KR Chaudhuri, K Suzuki, K Hirata, J Zimmermann, C Trenkwalder. Clinically relevant cut-off values for the Parkinson’s Disease Sleep Scale-2 (PDSS-2): a validation study. Sleep Med, 2016. [PubMed]
- V Mylius, JC Moller, S Bohlhalter, D Ciampi de Andrade, S Perez Lloret. Diagnosis and management of pain in Parkinson’s disease: a new approach. Drugs Aging, 2021. [PubMed]
- V Mylius, S Perez Lloret, CS Brook, MT Kruger, S Hagele-Link, R Gonzenbach, J Kassubek, S Bohlhalter, JP Lefaucheur, L Timmermann, G Kagi, F Brugger, D Ciampi de Andrade, JC Moller. The new Parkinson’s disease pain classification system (PD-PCS). Nervenarzt, 2022. [PubMed]
- V Mylius, S Perez Lloret, RG Cury, MJ Teixeira, VR Barbosa, ER Barbosa, LI Moreira, C Listik, AM Fernandes, VD de Lacerda, J Barbour, N Hollenstein, M Oechsner, J Walch, F Brugger, S Hagele-Link, S Beer, A Rizos, KR Chaudhuri, D Bouhassira, JP Lefaucheur, L Timmermann, R Gonzenbach, G Kagi, JC Moller, D Ciampi de Andrade. The Parkinson disease pain classification system: results from an international mechanism-based classification approach. Pain, 2021. [PubMed]
- AB Neikrug, L Liu, JA Avanzino, JE Maglione, L Natarajan, L Bradley, A Maugeri, J Corey-Bloom, BW Palmer, JS Loredo, S Ancoli-Israel. Continuous positive airway pressure improves sleep and daytime sleepiness in patients with Parkinson disease and sleep apnea. Sleep, 2014. [PubMed]
- JL Newton, J Frith. The efficacy of nonpharmacologic intervention for orthostatic hypotension associated with aging. Neurology, 2018. [PubMed]
- T Nomura, Y Inoue, T Kagimura, K Nakashima. Clinical significance of REM sleep behavior disorder in Parkinson’s disease. Sleep Med, 2013. [PubMed]
- L Norcliffe-Kaufmann, H Kaufmann, JA Palma, CA Shibao, I Biaggioni, AC Peltier, W Singer, PA Low, DS Goldstein, CH Gibbons, R Freeman, D Robertson. Orthostatic heart rate changes in patients with autonomic failure caused by neurodegenerative synucleinopathies. Ann Neurol, 2018. [PubMed]
- S O’Dowd, B Galna, R Morris, RA Lawson, C McDonald, AJ Yarnall, DJ Burn, L Rochester, KN Anderson. Poor sleep quality and progression of gait impairment in an incident Parkinson’s disease cohort. J Parkinsons Dis, 2017. [PubMed]
- JD O’Sullivan, AJ Hughes. Apomorphine-induced penile erections in Parkinson’s disease. Mov Disord, 1998. [PubMed]
- LE Okamoto, A Gamboa, CA Shibao, AC Arnold, L Choi, BK Black, SR Raj, D Robertson, I Biaggioni. Nebivolol, but not metoprolol, lowers blood pressure in nitric oxide-sensitive human hypertension. Hypertension, 2014. [PubMed]
- CW Olanow, K Kieburtz, M Stern, R Watts, JW Langston, M Guarnieri, J Hubble. Double-blind, placebo-controlled study of entacapone in levodopa-treated patients with stable Parkinson disease. Arch Neurol, 2004. [PubMed]
- WL Olson, M Gruenthal, ME Mueller, WH Olson. Gabapentin for parkinsonism: a double-blind, placebo-controlled, crossover trial. Am J Med, 1997. [PubMed]
- G Pagano, R De Micco, T Yousaf, H Wilson, A Chandra, M Politis. REM behavior disorder predicts motor progression and cognitive decline in Parkinson disease. Neurology, 2018. [PubMed]
- 165.pain IAftso (2024)
- JA Palma, JC Gomez-Esteban, L Norcliffe-Kaufmann, J Martinez, B Tijero, K Berganzo, H Kaufmann. Orthostatic hypotension in Parkinson disease: how much you fall or how low you go?. Mov Disord, 2015. [PubMed]
- JN Panicker, CJ Fowler, TM Kessler. Lower urinary tract dysfunction in the neurological patient: clinical assessment and management. Lancet Neurol, 2015. [PubMed]
- KW Park, S Jo, SH Lee, YS Hwang, D Lee, HS Ryu, SJ Chung. Therapeutic effect of levodopa/carbidopa/entacapone on sleep disturbance in patients with Parkinson’s disease. J Mov Disord, 2020. [PubMed]
- S Paschen, C Hansen, J Welzel, J Albrecht, A Atrsaei, K Aminian, KE Zeuner, R Romijnders, E Warmerdam, PP Urban, D Berg, W Maetzler. Effect of lower limb vs. abdominal compression on mobility in orthostatic hypotension: a single-blinded, randomized, controlled, cross-over pilot study in Parkinson’s disease. J Parkinsons Dis, 2022. [PubMed]
- M Peball, F Krismer, HG Knaus, A Djamshidian, M Werkmann, F Carbone, P Ellmerer, B Heim, K Marini, D Valent, G Goebel, H Ulmer, H Stockner, GK Wenning, R Stolz, K Krejcy, W Poewe, K Seppi. Non-motor symptoms in Parkinson’s disease are reduced by nabilone. Ann Neurol, 2020. [PubMed]
- MC Perissinotto, CA D’Ancona, A Lucio, RM Campos, A Abreu. Transcutaneous tibial nerve stimulation in the treatment of lower urinary tract symptoms and its impact on health-related quality of life in patients with Parkinson disease: a randomized controlled trial. J Wound Ostomy Continence Nurs, 2015. [PubMed]
- M Pierantozzi, F Placidi, C Liguori, M Albanese, P Imbriani, MG Marciani, NB Mercuri, P Stanzione, A Stefani. Rotigotine may improve sleep architecture in Parkinson’s disease: a double-blind, randomized, placebo-controlled polysomnographic study. Sleep Med, 2016. [PubMed]
- RD Piovezan, AD Jadczak, G Tucker, R Visvanathan. Daytime sleepiness predicts mortality in nursing home residents: findings from the Frailty in Residential Aged Care Sector Over Time (FIRST) study. J Am Med Dir Assoc, 2023. [PubMed]
- F Pizza, S Contardi, S Mondini, L Trentin, F Cirignotta. Daytime sleepiness and driving performance in patients with obstructive sleep apnea: comparison of the MSLT, the MWT, and a simulated driving task. Sleep, 2009. [PubMed]
- RB Postuma, J Anang, A Pelletier, L Joseph, M Moscovich, D Grimes, S Furtado, RP Munhoz, S Appel-Cresswell, A Moro, A Borys, D Hobson, AE Lang. Caffeine as symptomatic treatment for Parkinson disease (Cafe-PD): a randomized trial. Neurology, 2017. [PubMed]
- RB Postuma, JA Bertrand, J Montplaisir, C Desjardins, M Vendette, S Rios Romenets, M Panisset, JF Gagnon. Rapid eye movement sleep behavior disorder and risk of dementia in Parkinson’s disease: a prospective study. Mov Disord, 2012. [PubMed]
- L Priano, M Bigoni, G Albani, L Sellitti, E Giacomotti, R Picconi, R Cremascoli, M Zibetti, L Lopiano, A Mauro. Sleep microstructure in Parkinson’s disease: cycling alternating pattern (CAP) as a sensitive marker of early NREM sleep instability. Sleep Med, 2019. [PubMed]
- ME Pushpanathan, AM Loftus, MG Thomas, N Gasson, RS Bucks. The relationship between sleep and cognition in Parkinson’s disease: a meta-analysis. Sleep Med Rev, 2016. [PubMed]
- S Puvi-Rajasingham, CJ Mathias. Effect of meal size on post-prandial blood pressure and on postural hypotension in primary autonomic failure. Clin Auton Res, 1996. [PubMed]
- R Raffaele, I Vecchio, B Giammusso, G Morgia, MB Brunetto, L Rampello. Efficacy and safety of fixed-dose oral sildenafil in the treatment of sexual dysfunction in depressed patients with idiopathic Parkinson’s disease. Eur Urol, 2002. [PubMed]
- SR Raj, I Biaggioni, BK Black, A Rali, J Jordan, I Taneja, PA Harris, D Robertson. Sodium paradoxically reduces the gastropressor response in patients with orthostatic hypotension. Hypertension, 2006. [PubMed]
- M Rakusa, E Moro, T Akhvlediani, D Bereczki, B Bodini, F Cavallieri, A Fanciulli, SR Filipovic, A Guekht, R Helbok, S Hochmeister, F Martinelli Boneschi, S Ozturk, A Priori, M Romoli, B Willekens, M Zedde, J Sellner, Neuro C-TFotEAoN,. The COVID-19 pandemic and neurology: a survey on previous and continued restrictions for clinical practice, curricular training, and health economics. Eur J Neurol, 2024. [PubMed]
- CE Ramirez, LE Okamoto, AC Arnold, A Gamboa, A Diedrich, L Choi, SR Raj, D Robertson, I Biaggioni, CA Shibao. Efficacy of atomoxetine versus midodrine for the treatment of orthostatic hypotension in autonomic failure. Hypertension, 2014. [PubMed]
- F Raschella, S Scafa, A Puiatti, E Martin Moraud, PL Ratti. Actigraphy enables home screening of rapid eye movement behavior disorder in Parkinson’s disease. Ann Neurol, 2023. [PubMed]
- O Rascol, J Bronzova, RA Hauser, AE Lang, C Sampaio, A Theeuwes, SV van de Witte. Pardoprunox as adjunct therapy to levodopa in patients with Parkinson’s disease experiencing motor fluctuations: results of a double-blind, randomized, placebo-controlled, trial. Parkinsonism Relat Disord, 2012. [PubMed]
- I Rieu, B Degos, G Castelnovo, C Vial, E Durand, B Pereira, M Simonetta-Moreau, S Sangla, F Fluchere, D Guehl, P Burbaud, C Geny, D Gayraud, F Ory-Magne, F Bouhour, E Llinares, P Derost, A Marques, F Durif. Incobotulinum toxin A in Parkinson’s disease with foot dystonia: a double blind randomized trial. Parkinsonism Relat Disord, 2018. [PubMed]
- G Rivasi, M Rafanelli, E Mossello, M Brignole, A Ungar. Drug-related orthostatic hypotension: beyond anti-hypertensive medications. Drugs Aging, 2020. [PubMed]
- TM Rodrigues, A Castro Caldas, JJ Ferreira. Pharmacological interventions for daytime sleepiness and sleep disorders in Parkinson’s disease: systematic review and meta-analysis. Parkinsonism Relat Disord, 2016. [PubMed]
- C Rodriguez-Blazquez, A Schrag, A Rizos, KR Chaudhuri, P Martinez-Martin, D Weintraub. Prevalence of non-motor symptoms and non-motor fluctuations in Parkinson’s disease using the MDS-NMS. Mov Disord Clin Pract, 2021. [PubMed]
- S Rutten, C Vriend, YD van der Werf, HW Berendse, D Weintraub, OA van den Heuvel. The bidirectional longitudinal relationship between insomnia, depression and anxiety in patients with early-stage, medication-naive Parkinson’s disease. Parkinsonism Relat Disord, 2017. [PubMed]
- R Sakakibara, M Kishi, E Ogawa, F Tateno, T Uchiyama, T Yamamoto, T Yamanishi. Bladder, bowel, and sexual dysfunction in Parkinson’s disease. Parkinsons Dis, 2011. [PubMed]
- R Sakakibara, S Matsuda, T Uchiyama, M Yoshiyama, T Yamanishi, T Hattori. The effect of intranasal desmopressin on nocturnal waking in urination in multiple system atrophy patients with nocturnal polyuria. Clin Auton Res, 2003. [PubMed]
- R Sakakibara, J Panicker, E Finazzi-Agro, V Iacovelli, H Bruschini. A guideline for the management of bladder dysfunction in Parkinson’s disease and other gait disorders. Neurourol Urodyn, 2015. [PubMed]
- S Sakoda, T Suzuki, S Higa, M Ueji, S Kishimoto, M Matsumoto, S Yoneda. Treatment of orthostatic hypotension in Shy-Drager syndrome with dl-threo-3,4-dihydroxyphenylserine: a case report. Eur Neurol, 1985. [PubMed]
- D Santos Garcia, C Labandeira Guerra, R Yanez Bana, MI Cimas Hernando, I Cabo Lopez, JM Paz Gonalez, MG Alonso Losada, MJ Gonzalez Palmas, C Martinez Miro. Safinamide improves non-motor symptoms burden in Parkinson’s disease: an open-label prospective study. Brain Sci, 2021. [PubMed]
- T Scaravilli, E Gasparoli, F Rinaldi, G Polesello, F Bracco. Health-related quality of life and sleep disorders in Parkinson’s disease. Neurol Sci, 2003. [PubMed]
- AHV Schapira, KR Chaudhuri, P Jenner. Non-motor features of Parkinson disease. Nat Rev Neurosci, 2017. [PubMed]
- KL Schoffer, RD Henderson, K O’Maley, JD O’Sullivan. Nonpharmacological treatment, fludrocortisone, and domperidone for orthostatic hypotension in Parkinson’s disease. Mov Disord, 2007. [PubMed]
- SJ Schreiner, LL Imbach, E Werth, R Poryazova, H Baumann-Vogel, PO Valko, T Murer, D Noain, CR Baumann. Slow-wave sleep and motor progression in Parkinson disease. Ann Neurol, 2019. [PubMed]
- B Schurch, M de Seze, P Denys, E Chartier-Kastler, F Haab, K Everaert, P Plante, B Perrouin-Verbe, C Kumar, S Fraczek, MF Brin. Botulinum toxin type a is a safe and effective treatment for neurogenic urinary incontinence: results of a single treatment, randomized, placebo controlled 6-month study. J Urol, 2005. [PubMed]
- L Schutz, F Sixel-Doring, W Hermann. Management of sleep disturbances in Parkinson’s disease. J Parkinsons Dis, 2022. [PubMed]
- A Scott-Massey, MK Boag, A Magnier, D Bispo, TK Khoo, DL Pountney. Glymphatic system dysfunction and sleep disturbance may contribute to the pathogenesis and progression of Parkinson’s disease. Int J Mol Sci, 2022. [PubMed]
- JM Senard, C Brefel, C Carel, MA Tran, JL Montastruc. Water drinking and the heart. Lancet, 1999. [PubMed]
- K Seppi, K Ray Chaudhuri, M Coelho, SH Fox, R Katzenschlager, S Perez Lloret, D Weintraub, C Sampaio. Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov Disord, 2019. [PubMed]
- J Shannon, J Jordan, F Costa, RM Robertson, I Biaggioni. The hypertension of autonomic failure and its treatment. Hypertension, 1997. [PubMed]
- JR Shannon, J Jordan, A Diedrich, B Pohar, BK Black, D Robertson, I Biaggioni. Sympathetically mediated hypertension in autonomic failure. Circulation, 2000. [PubMed]
- Y Shen, JY Huang, J Li, CF Liu. Excessive daytime sleepiness in Parkinson’s disease: clinical implications and management. Chin Med J (Engl), 2018. [PubMed]
- C Shibao, A Gamboa, R Abraham, SR Raj, A Diedrich, B Black, D Robertson, I Biaggioni. Clonidine for the treatment of supine hypertension and pressure natriuresis in autonomic failure. Hypertension, 2006. [PubMed]
- A Shohet, A Khlebtovsky, N Roizen, Y Roditi, R Djaldetti. Effect of medical cannabis on thermal quantitative measurements of pain in patients with Parkinson’s disease. Eur J Pain, 2017. [PubMed]
- MA Silverdale, C Kobylecki, L Kass-Iliyya, P Martinez-Martin, M Lawton, S Cotterill, KR Chaudhuri, H Morris, F Baig, N Williams, L Hubbard, MT Hu, DG Grosset. A detailed clinical study of pain in 1957 participants with early/moderate Parkinson’s disease. Parkinsonism Relat Disord, 2018. [PubMed]
- T Simuni, C Caspell-Garcia, C Coffey, LM Chahine, S Lasch, WH Oertel, G Mayer, B Hogl, R Postuma, A Videnovic, AW Amara, K Marek. Correlates of excessive daytime sleepiness in de novo Parkinson’s disease: a case control study. Mov Disord, 2015. [PubMed]
- AA Smit, W Wieling, J Fujimura, JC Denq, TL Opfer-Gehrking, M Akarriou, JM Karemaker, PA Low. Use of lower abdominal compression to combat orthostatic hypotension in patients with autonomic dysfunction. Clin Auton Res, 2004. [PubMed]
- MC Smith, H Ellgring, WH Oertel. Sleep disturbances in Parkinson’s disease patients and spouses. J Am Geriatr Soc, 1997. [PubMed]
- MA Sobreira-Neto, MA Pena-Pereira, EST Sobreira, MHN Chagas, RMF Fernandes, V Tumas, AL Eckeli. High frequency of sleep disorders in Parkinson’s disease and its relationship with quality of life. Eur Neurol, 2017. [PubMed]
- J Sringean, C Anan, C Thanawattano, R Bhidayasiri. Time for a strategy in night-time dopaminergic therapy? An objective sensor-based analysis of nocturnal hypokinesia and sleeping positions in Parkinson’s disease. J Neurol Sci, 2017. [PubMed]
- DG Standaert, J Aldred, M Anca-Herschkovitsch, P Bourgeois, E Cubo, TL Davis, R Iansek, N Kovacs, FE Pontieri, MS Siddiqui, M Simu, L Bergmann, P Kukreja, WZ Robieson, KR Chaudhuri. DUOGLOBE: one-year outcomes in a real-world study of levodopa carbidopa intestinal gel for Parkinson’s disease. Mov Disord Clin Pract, 2021. [PubMed]
- M Stauder, EB Klerman, W Wang, H Erickson, A Videnovic. Relationships of self-reported and objective measures of sleep, sleepiness, and sleep quality in Parkinson’s disease. Parkinsonism Relat Disord, 2022. [PubMed]
- A Stefani, B Hogl. Sleep in Parkinson’s disease. Neuropsychopharmacology, 2020. [PubMed]
- F Stocchi, M Torti. Constipation in Parkinson’s disease. Int Rev Neurobiol, 2017. [PubMed]
- S Sundaram, RL Hughes, E Peterson, EM Muller-Oehring, HM Bronte-Stewart, KL Poston, A Faerman, C Bhowmick, T Schulte. Establishing a framework for neuropathological correlates and glymphatic system functioning in Parkinson’s disease. Neurosci Biobehav Rev, 2019. [PubMed]
- K Suzuki, M Miyamoto, T Miyamoto, T Uchiyama, Y Watanabe, S Suzuki, T Kadowaki, H Fujita, T Matsubara, H Sakuramoto, K Hirata. Istradefylline improves daytime sleepiness in patients with Parkinson’s disease: an open-label, 3-month study. J Neurol Sci, 2017. [PubMed]
- S Szatmari, D Bereczki, K Fornadi, K Kalantar-Zadeh, CP Kovesdy, MZ Molnar. Association of restless legs syndrome with incident Parkinson’s disease. Sleep, 2017. [PubMed]
- S Takahashi, M Takei, H Asakura, M Gotoh, O Ishizuka, K Kato, M Koyama, M Takeyama, H Tomoe, T Yamanishi, O Yokoyama, M Yoshida, Y Yoshimura, T Yoshizawa. Clinical guidelines for female lower urinary tract symptoms (second edition). Int J Urol, 2021. [PubMed]
- AH Tan, JW Hor, CW Chong, SY Lim. Probiotics for Parkinson’s disease: current evidence and future directions. JGH Open, 2021. [PubMed]
- AH Tan, SY Lim, KK Chong, MAA A Manap, JW Hor, JL Lim, SC Low, CW Chong, S Mahadeva, AE Lang. Probiotics for constipation in Parkinson disease: a randomized placebo-controlled study. Neurology, 2021. [PubMed]
- R Taximaimaiti, X Luo, XP Wang. Pharmacological and non-pharmacological treatments of sleep disorders in Parkinson’s disease. Curr Neuropharmacol, 2021. [PubMed]
- AD Ten Harkel, JJ van Lieshout, W Wieling. Effects of leg muscle pumping and tensing on orthostatic arterial pressure: a study in normal subjects and patients with autonomic failure. Clin Sci (Lond), 1994. [PubMed]
- AD Ten Harkel, JJ Van Lieshout, W Wieling. Treatment of orthostatic hypotension with sleeping in the head-up tilt position, alone and in combination with fludrocortisone. J Intern Med, 1992. [PubMed]
- RD Thijs, M Brignole, C Falup-Pecurariu, A Fanciulli, R Freeman, P Guaraldi, J Jordan, M Habek, M Hilz, AP Traon, I Stankovic, W Struhal, R Sutton, G Wenning, JG Van Dijk. Recommendations for tilt table testing and other provocative cardiovascular autonomic tests in conditions that may cause transient loss of consciousness: consensus statement of the European Federation of Autonomic Societies (EFAS) endorsed by the American Autonomic Society (AAS) and the European Academy of Neurology (EAN). Clin Auton Res, 2021. [PubMed]
- LK Tholfsen, JP Larsen, J Schulz, OB Tysnes, MD Gjerstad. Changes in insomnia subtypes in early Parkinson disease. Neurology, 2017. [PubMed]
- L Timmermann, C Oehlwein, G Ransmayr, H Frohlich, E Will, H Schroeder, T Lauterbach, L Bauer, J Kassubek. Patients’ perception of Parkinson’s disease-associated pain following initiation of rotigotine: a multicenter non-interventional study. Postgrad Med, 2017. [PubMed]
- A Todorova, B Vonderheid-Guth, W Dimpfel. Effects of tolterodine, trospium chloride, and oxybutynin on the central nervous system. J Clin Pharmacol, 2001. [PubMed]
- C Trenkwalder, KR Chaudhuri, P Martinez-Martin, O Rascol, R Ehret, M Valis, M Satori, A Krygowska-Wajs, MJ Marti, K Reimer, A Oksche, M Lomax, J DeCesare, M Hopp. Prolonged-release oxycodone-naloxone for treatment of severe pain in patients with Parkinson’s disease (PANDA): a double-blind, randomised, placebo-controlled trial. Lancet Neurol, 2015. [PubMed]
- C Trenkwalder, B Kies, M Rudzinska, J Fine, J Nikl, K Honczarenko, P Dioszeghy, D Hill, T Anderson, V Myllyla, J Kassubek, M Steiger, M Zucconi, E Tolosa, W Poewe, E Surmann, J Whitesides, B Boroojerdi. Rotigotine effects on early morning motor function and sleep in Parkinson’s disease: a double-blind, randomized, placebo-controlled study (RECOVER). Mov Disord, 2011. [PubMed]
- C Trenkwalder, R Kohnen, B Hogl, V Metta, F Sixel-Doring, B Frauscher, J Hulsmann, P Martinez-Martin, KR Chaudhuri. Parkinson’s disease sleep scale—validation of the revised version PDSS-2. Mov Disord, 2011. [PubMed]
- M Tutaj, H Marthol, D Berlin, CM Brown, FB Axelrod, MJ Hilz. Effect of physical countermaneuvers on orthostatic hypotension in familial dysautonomia. J Neurol, 2006. [PubMed]
- P Valkovic, M Minar, H Singliarova, J Harsany, M Hanakova, J Martinkova, J Benetin. Pain in Parkinson’s disease: a cross-sectional study of its prevalence, types, and relationship to depression and quality of life. PLoS ONE, 2015. [PubMed]
- N Van Blercom, A Lasa, K Verger, X Masramon, VM Sastre, G Linazasoro. Effects of gabapentin on the motor response to levodopa: a double-blind, placebo-controlled, crossover study in patients with complicated Parkinson disease. Clin Neuropharmacol, 2004. [PubMed]
- JJ van Lieshout, AD ten Harkel, W Wieling. Fludrocortisone and sleeping in the head-up position limit the postural decrease in cardiac output in autonomic failure. Clin Auton Res, 2000. [PubMed]
- JJ van Lieshout, AD ten Harkel, W Wieling. Physical manoeuvres for combating orthostatic dizziness in autonomic failure. Lancet, 1992. [PubMed]
- DC Velseboer, RJ de Haan, W Wieling, DS Goldstein, RM de Bie. Prevalence of orthostatic hypotension in Parkinson’s disease: a systematic review and meta-analysis. Parkinsonism Relat Disord, 2011. [PubMed]
- B Verheyden, J Gisolf, F Beckers, JM Karemaker, KH Wesseling, AE Aubert, W Wieling. Impact of age on the vasovagal response provoked by sublingual nitroglycerine in routine tilt testing. Clin Sci (Lond), 2007. [PubMed]
- R Vespalec, P Gebauer, P Bocek. Identification of peaks in capillary zone electrophoresis based on actual mobilities. Electrophoresis, 1992. [PubMed]
- A Videnovic. Management of sleep disorders in Parkinson’s disease and multiple system atrophy. Mov Disord, 2017. [PubMed]
- A Videnovic, YS Ju, I Arnulf, V Cochen-De Cock, B Hogl, D Kunz, F Provini, PL Ratti, MC Schiess, CH Schenck, C Trenkwalder. Clinical trials in REM sleep behavioural disorder: challenges and opportunities. J Neurol Neurosurg Psychiatry, 2020. [PubMed]
- A Videnovic, EB Klerman, W Wang, A Marconi, T Kuhta, PC Zee. Timed light therapy for sleep and daytime sleepiness associated with Parkinson disease: a randomized clinical Trial. JAMA Neurol, 2017. [PubMed]
- A Videnovic, C Noble, KJ Reid, J Peng, FW Turek, A Marconi, AW Rademaker, T Simuni, C Zadikoff, PC Zee. Circadian melatonin rhythm and excessive daytime sleepiness in Parkinson disease. JAMA Neurol, 2014. [PubMed]
- R Wade, NA Pachana, N Dissanayaka. Factors related to sleep disturbances for informal carers of individuals with PD and dyadic relationship: a rural perspective. J Geriatr Psychiatry Neurol, 2021. [PubMed]
- SA Wallace, B Roe, K Williams, M Palmer. Bladder training for urinary incontinence in adults. Cochrane Database Syst Rev, 2004
- T Warnecke, C Lummer, JW Rey, I Claus, D Luttje. Parkinson’s disease. Inn Med (Heidelb), 2023. [PubMed]
- T Warnecke, KH Schafer, I Claus, K Del Tredici, WH Jost. Gastrointestinal involvement in Parkinson’s disease: pathophysiology, diagnosis, and management. NPJ Parkinsons Dis, 2022. [PubMed]
- G Wasner, G Deuschl. Pains in Parkinson disease—many syndromes under one umbrella. Nat Rev Neurol, 2012. [PubMed]
- D Weintraub, S Mavandadi, E Mamikonyan, AD Siderowf, JE Duda, HI Hurtig, A Colcher, SS Horn, S Nazem, TR Ten Have, MB Stern. Atomoxetine for depression and other neuropsychiatric symptoms in Parkinson disease. Neurology, 2010. [PubMed]
- W Wieling, H Kaufmann, VE Claydon, VK van Wijnen, MPM Harms, SP Juraschek, RD Thijs. Diagnosis and treatment of orthostatic hypotension. Lancet Neurol, 2022. [PubMed]
- K Winge, CJ Fowler. Bladder dysfunction in Parkinsonism: mechanisms, prevalence, symptoms, and management. Mov Disord, 2006. [PubMed]
- RA Wright, HC Kaufmann, R Perera, TL Opfer-Gehrking, MA McElligott, KN Sheng, PA Low. A double-blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology, 1998. [PubMed]
- W Xiang, YQ Sun, HC Teoh. Comparison of nocturnal symptoms in advanced Parkinson’s disease patients with sleep disturbances: pramipexole sustained release versus immediate release formulations. Drug Des Devel Ther, 2018. [PubMed]
- Y Yang, G Wang, S Zhang, H Wang, W Zhou, F Ren, H Liang, D Wu, X Ji, M Hashimoto, J Wei. Efficacy and evaluation of therapeutic exercises on adults with Parkinson’s disease: a systematic review and network meta-analysis. BMC Geriatr, 2022. [PubMed]
- L Yao, W Liang, J Chen, Q Wang, X Huang. Constipation in Parkinson’s disease: a systematic review and meta-analysis. Eur Neurol, 2023. [PubMed]
- F Yenilmez, O Frundt, U Hidding, C Buhmann. Cannabis in Parkinson’s disease: the patients’ view. J Parkinsons Dis, 2021. [PubMed]
- S Yin, H Yang, Y Tao, S Wei, L Li, M Liu, J Li. Artesunate ameliorates DSS-induced ulcerative colitis by protecting intestinal barrier and inhibiting inflammatory response. Inflammation, 2020. [PubMed]
- SW Yoo, JS Kim, YS Oh, DW Ryu, KS Lee. Excessive daytime sleepiness and its impact on quality of life in de novo Parkinson’s disease. Neurol Sci, 2019. [PubMed]
- TM Young, CJ Mathias. The effects of water ingestion on orthostatic hypotension in two groups of chronic autonomic failure: multiple system atrophy and pure autonomic failure. J Neurol Neurosurg Psychiatry, 2004. [PubMed]
- RL Yu, CH Tan, RM Wu. The impact of nocturnal disturbances on daily quality of life in patients with Parkinson’s disease. Neuropsychiatr Dis Treat, 2015. [PubMed]
- WJ Z’Graggen, CW Hess, AM Humm. Acute fluid ingestion in the treatment of orthostatic intolerance—important implications for daily practice. Eur J Neurol, 2010. [PubMed]
- H Zahed, JRP Zuzuarregui, R Gilron, T Denison, PA Starr, S Little. The neurophysiology of sleep in Parkinson’s disease. Mov Disord, 2021. [PubMed]
- TA Zesiewicz, M Helal, RA Hauser. Sildenafil citrate (Viagra) for the treatment of erectile dysfunction in men with Parkinson’s disease. Mov Disord, 2000. [PubMed]
- Y Zhang, R Ren, LD Sanford, L Yang, J Zhou, L Tan, T Li, J Zhang, YK Wing, J Shi, L Lu, X Tang. Sleep in Parkinson’s disease: a systematic review and meta-analysis of polysomnographic findings. Sleep Med Rev, 2020. [PubMed]
- K Zhu, JJ van Hilten, J Marinus. The course of insomnia in Parkinson’s disease. Parkinsonism Relat Disord, 2016. [PubMed]
- JRP Zuzuarregui, JL Ostrem. The impact of deep brain stimulation on sleep in Parkinson’s disease: an update. J Parkinsons Dis, 2020. [PubMed]
