Cannabinol modulates the endocannabinoid system and shows TRPV1‐mediated anti‐inflammatory properties in human keratinocytes
Abstract
Cannabinol (CBN) is a secondary metabolite of cannabis whose beneficial activity on inflammatory diseases of human skin has attracted increasing attention. Here, we sought to investigate the possible modulation by CBN of the major elements of the endocannabinoid system (ECS), in both normal and lipopolysaccharide‐inflamed human keratinocytes (HaCaT cells). CBN was found to increase the expression of cannabinoid receptor 1 (CB1) at gene level and that of vanilloid receptor 1 (TRPV1) at protein level, as well as their functional activity. In addition, CBN modulated the metabolism of anandamide (AEA) and 2‐arachidonoylglicerol (2‐AG), by increasing the activities of N‐acyl phosphatidylethanolamines‐specific phospholipase D (NAPE‐PLD) and fatty acid amide hydrolase (FAAH)—the biosynthetic and degradative enzyme of AEA—and that of monoacylglycerol lipase (MAGL), the hydrolytic enzyme of 2‐AG. CBN also affected keratinocyte inflammation by reducing the release of pro‐inflammatory interleukin (IL)‐8, IL‐12, and IL‐31 and increasing the release of anti‐inflammatory IL‐10. Of note, the release of IL‐31 was mediated by TRPV1. Finally, the mitogen‐activated protein kinases (MAPK) signaling pathway was investigated in inflamed keratinocytes, demonstrating a specific modulation of glycogen synthase kinase 3β (GSK3β) upon treatment with CBN, in the presence or not of distinct ECS‐directed drugs. Overall, these results demonstrate that CBN modulates distinct ECS elements and exerts anti‐inflammatory effects—remarkably via TRPV1—in human keratinocytes, thus holding potential for both therapeutic and cosmetic purposes.
Article type: Research Article
Keywords: cannabinol, cytokines, endocannabinoid system, keratinocytes, skin inflammation
Affiliations: Department of Veterinary Medicine University of Teramo Teramo Italy; Department of Biotechnological and Applied Clinical Sciences University of L’Aquila L’Aquila Italy; European Center for Brain Research (CERC) Santa Lucia Foundation IRCCS Rome Italy; InMed Pharmaceuticals Inc. Vancouver BC Canada
License: © 2024 The Author(s). BioFactors published by Wiley Periodicals LLC on behalf of International Union of Biochemistry and Molecular Biology. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/biof.2122 | PubMed: 39275884 | PMC: PMC11681214
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (2.9 MB)
INTRODUCTION
Phytocannabinoids (pCBs) are a group of more than 150 different lipophilic compounds derived from cannabis (Cannabis sativa), a flowering plant native to Central Asia.ref. biof2122-bib-0001, ref. biof2122-bib-0002 Cannabis has been cultivated for millennia with different aims such as nutrition, recreation, generation of seed oil and fibers for industrial purposes, as well as for religious and spiritual practices and, most interestingly, for medical applications.ref. biof2122-bib-0003 The best studied pCBs are Δ9‐tetrahydrocannabinol (THC) and cannabidiol (CBD),ref. biof2122-bib-0002, ref. biof2122-bib-0004 although cannabis produces many other “minor” pCBs whose biological profile remains poorly investigated.ref. biof2122-bib-0005, ref. biof2122-bib-0006 Recently, improved extraction and (bio)synthetic techniques have led to amounts of less abundant pCBs that are enough to investigate their biological activity.ref. biof2122-bib-0007 Among them, cannabinol (CBN) has recently attracted particular attention,ref. biof2122-bib-0007, ref. biof2122-bib-0008 as a non‐psychoactive compound whose levels in cannabis flowers increase over time via the first conversion of THC acid (THCA) into THC, and the subsequent oxidation of THC into CBN.ref. biof2122-bib-0008, ref. biof2122-bib-0009 Recent studies have suggested that CBN may have anti‐inflammatory, analgesic and antibacterial properties,ref. biof2122-bib-0006, ref. biof2122-bib-0007, ref. biof2122-bib-0010 although the mechanisms of action have still to be clarified.
In general, pCBs can interact with the endocannabinoid system (ECS),ref. biof2122-bib-0007, ref. biof2122-bib-0009, ref. biof2122-bib-0011 a complex network composed of endocannabinoids (eCBs), their receptors, metabolic enzymes and transporters, which altogether regulate various physiological processes and maintain homeostasis in the human body,ref. biof2122-bib-0012, ref. biof2122-bib-0013 skin included.ref. biof2122-bib-0014, ref. biof2122-bib-0015 Anandamide (N‐arachidonoylethanolamine, AEA) and 2‐arachidonoylglycerol (2‐AG) are the best studied eCBs, that are both synthesized from membrane phospholipid precursors via different biosynthetic pathways.ref. biof2122-bib-0012, ref. biof2122-bib-0016, ref. biof2122-bib-0017, ref. biof2122-bib-0018, ref. biof2122-bib-0019, ref. biof2122-bib-0020 In particular, the cleavage of N‐arachidonoyl phosphatidylethanolamine (NArPE) by N‐acyl phosphatidylethanolamines‐specific phospholipase D (NAPE‐PLD) produces AEA,ref. biof2122-bib-0021, ref. biof2122-bib-0022 that is hydrolyzed by fatty acid amide hydrolase (FAAH).ref. biof2122-bib-0023 2‐AG is generated from the cleavage of diacylglycerol (DAG) by the two isoforms of diacylglycerol lipase (DAGL) α and β,ref. biof2122-bib-0024 which exhibit distinct spatial and cellular distributions. DAGLα is predominantly localized in the brain,ref. biof2122-bib-0025, ref. biof2122-bib-0026 while DAGLβ is primarily expressed in immune cells including microglia, macrophages, and dendritic cells, being also involved in inflammatory processes.ref. biof2122-bib-0027, ref. biof2122-bib-0028, ref. biof2122-bib-0029 Then, 2‐AG is cleaved by monoacylglycerol lipase (MAGL).ref. biof2122-bib-0023, ref. biof2122-bib-0030 The two main receptors responsible for the biological activity of eCBs are the cannabinoid receptors 1 (CB1) and 2 (CB2), though other orphan G‐coupled receptors like the G protein‐coupled receptor 55 (GPR55), the transient receptor potential (TRP) vanilloid receptor 1 (TRPV1), and peroxisome proliferator‐activated nuclear receptors (PPARs) α, γ, δ can be activated by eCBs.ref. biof2122-bib-0011 Remarkably, the main components of the ECS have been found in human keratinocytes, which are the predominant cells of the epidermis.ref. biof2122-bib-0015, ref. biof2122-bib-0031, ref. biof2122-bib-0032, ref. biof2122-bib-0033, ref. biof2122-bib-0034 ECS activation in keratinocytes has been shown to impact on various processes engaged in the regulation of skin physiology, while the dysregulation of the same system is linked to different skin diseases.ref. biof2122-bib-0014, ref. biof2122-bib-0015, ref. biof2122-bib-0031 In this context, we have recently shown that minor pCBs [i.e., cannabigerol (CBG), cannabichromene (CBC), Δ9‐tetrahydrocannabivarin (THCV) and cannabigerolic acid (CBGA)] modulate the expression and functional activity of distinct ECS elements in HaCaT cells,ref. biof2122-bib-0035 and block skin inflammation.ref. biof2122-bib-0036 Here, we sought to extend these observations to CBN, by using lipopolysaccharide (LPS)‐inflamed HaCaT cells. Taken together, our data reveal different CBN‐induced modulations of the ECS, as well as its anti‐inflammatory effect by reducing IL‐31 release through a TRPV1‐dependent mechanism.
MATERIALS AND METHODS
Materials
Dulbecco’s modified Eagle medium‐high glucose (DMEM‐HG) and fetal bovine serum (FBS) were purchased from Corning Incorporated (NY, USA); antibiotics (penicillin/streptomycin, pen/strep), Dulbecco’s phosphate buffer saline (D‐PBS without calcium and magnesium), and trypsin (2.5%) were from Gibco by Life Technologies (Thermo Fisher Scientific Company, Waltham, MA, USA); ethylenediamine tetraacetic acid (EDTA, 0.5 M) was from Invitrogen (Thermo Fisher Scientific Company, Waltham, MA, USA); Opti‐MEM was from Thermo Fisher Scientific (Waltham, MA, USA). CBN was purchased from Cerilliant Corporation (Sigma Aldrich Company, St. Louis, MO, USA). LPS from Escherichia coli O111:B4 (suitable for cell culture) and hydrocortisone (HC) were purchased from Sigma Aldrich (St. Louis, MO, USA). The agonist capsaicin, the antagonists SR141716A and CPZ were obtained from Sigma Aldrich (St. Louis, MO, USA). 3‐(4,5‐Dimethylthiazol‐2‐yl)‐2,5‐diphenyltetrazolium bromide (MTT) reagent was purchased from Sigma‐Aldrich (St. Louis, MO, USA) and dimethyl sulfoxide (DMSO) was from PanReac AppliChem (Glenview, IL, USA). ELISA kit to detect apoptosis was purchased from Roche Diagnostic (Basilea, Switzerland), while ELISA selective kits for each interleukin were purchased from R&D System DuoSet (Bio‐Techne, Minneapolis, MN, USA). Supplies for the RT‐qPCR, including the RevertAid H Minus First Strand cDNA Synthesis Kit and SensiFAST SYBR Lo‐ROX Kit, were obtained from Thermo Fisher Scientific (Waltham, MA, USA) and Bioline (Meridian Bioscience Inc. Company, Cincinnati, OH, USA), respectively, whereas supplies for Western blotting were purchased from Bio‐Rad Laboratories (Hercules, California, USA). [3H]CP55.940, CP55.940, and [3H]AEA were from PerkinElmer Life Sciences (Waltham, MA, USA); Fluo‐3 AM and Pluronic F‐127 were from Molecular Probes (Thermo Fisher Scientific, Waltham, MA, USA); N‐((6‐(2,4 dinitrophenyl)amino)hexanoyl)‐2(4,4‐difluoro‐5,7‐dimethyl‐4‐bora‐3a,4a‐diaza‐s indacene‐3‐pentanoyl)‐1‐hexadecanoyl‐sn‐glycero‐3 phosphoethanolamine, triethylammonium salt (PED6) was from Thermo Fisher Scientific (Waltham, MA, USA); p‐nitrophenyl butyrate (pNPB) was from Sigma Aldrich (St. Louis, MO, USA) and KT172 and arachidonoyl‐1‐thio‐glycerol were from Cayman Chemical (Ann Arbor, MI, USA). Human/mouse MAPK phosphorylation array was obtained from RayBiotech (RayBiotech Inc., Peachtree Corners, GA, USA).
Cell line and treatments
Immortalized HaCaT cells of Caucasian skin type (phototype) from the original depositor (DKFZ, Heidelberg) were purchased from Cell Lines Service (CLS Cell Lines Service, 300493). In all the experiments, HaCaT cells were used at the 5th–6th passage after thawing, cultured at 37°C in a humidified 5% CO2 atmosphere in DMEM‐HG and supplemented with 10% FBS and 1% antibiotic–antimycotic (pen/strep) solution. For MTT and apoptosis assays, RT‐qPCR, western blotting, receptors binding, and enzymes activity assays, cells were seeded in specific plates and, after 24 h, the cell medium was replaced with DMEM‐HG supplemented with 1% FBS and 1% pen/strep (starvation medium) and incubated for 6, 12, 24, and/or 48 h in the presence of CBN at specific concentrations (μM) according to each analysis. For the ELISA assay and MAPK array, performed in inflammatory conditions, cells were starved with DMEM‐HG with 1% FBS and 1% pen/strep for 24 h and then incubated as the following conditions: control (DMEM‐HG + 5% FBS + 1% pen/strep); LPS at the concentration of 5.0 μg/mL, positive control HC at 10 μM; CBN at 2.5 μM alone and in combination with the selective CB1 antagonist (SR141716A) at 0.1 μMref. biof2122-bib-0037 and the selective TRPV1 antagonist CPZ at 5.0 μM.ref. biof2122-bib-0038 After 24 and 48 h, according to specific analysis, cell pellets and supernatants (medium) were collected from each condition, and then centrifuged at 300g for 5 min and stored at −20 and at −80°C, respectively.
MTT cytotoxicity assay
HaCaT cells were seeded into 96‐well plates at a density of 1 × 104 per well and the day after were treated with vehicle (0.8% methanol) and CBN at different concentrations (0.5, 1.0, 2.5, 5.0, 10, and 25 μM), for the timepoints of 6, 12, and 24 h. After each timepoint, cells were incubated with MTT reagent at the final concentration of 0.5 mg/mL for 4 h. Then, the MTT solution was discarded, and DMSO was added to dissolve the formazan crystals. Cell viability was assessed by the reduction of the MTT reagent to purple formazan, by measuring the optical densities (OD) values at the EnSpire Multimode Plate Reader (Perkin Elmer, Waltham, MA, USA). Cell viability was expressed as percentage of control (100%), calculated by subtracting the 630 nm OD background from the 570 nm OD total signal for each sample, followed by the subtraction of cell‐free blank wells, as reported.ref. biof2122-bib-0035
Determination of apoptosis
HaCaT cells were seeded into 96‐well plates at a density of 1 × 104 per well and the day after were treated with CBN at different concentrations (0.5, 1.0, 2.5, 5.0, 10, and 25 μM) for 24 h. Apoptotic cell death was quantified by an ELISA assay (Cell Death Detection ELISAPLUS, #11774425001, Roche Diagnostic, Basilea, Switzerland), based on the evaluation of DNA fragmentation through an immunoassay for histone‐associated DNA fragments in the cell cytoplasm. The ELISA kit also provides a positive control, represented by a DNA–histone‐complex. Specific determination of mono‐ and oligonucleosomes in the cytoplasmic fraction of cell lysates was performed by measuring the absorbance values at 405 nm, as reported.ref. biof2122-bib-0039
Quantitative real‐time polymerase chain reaction
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then exposed to CBN at 2.5 μM for 24 h. After treatments, cell pellets were collected and stored at −20°C. Total RNA was extracted from the cell pellets by using QIAzol Lysis Reagent (Qiagen, Hilden, Germany), according to manufacturer’s instructions, and then quantified with a NanoDrop™ 2000/2000c Spectrophotometer (Thermo Fisher Scientific Company, Waltham, MA, USA). Five hundred nanograms of isolated mRNA was reverse transcribed in a final volume of 20 μL, later diluted 1:3, following incubation in a thermocycler for 5 min at 65°C, 5 min at 25°C, 60 min at 42°C, and 5 min at 70°C, by using the RevertAid H Minus First Strand cDNA Synthesis (Thermo Fisher Scientific Company, Waltham, MA, USA). Quantitative real‐time polymerase chain reaction (RT‐qPCR) was performed in a final volume of 10 μL, consisting of 1 μL of cDNA, 1 μL of primers Forward + Reverse (10 μM mix) and 5 μL of Sybr Green 1× (SensiFAST SYBR Lo‐ROX Kit, Bioline by Meridian Bioscience Inc., Cincinnati, OH, USA), in the Applied Biosystems 7500 Fast Real‐Time PCR System (Life Technologies, Carlsbad, CA, USA). The qPCR reaction was performed with the following conditions: holding at 50°C for 2 min and 95°C for 10 min, cycling at 95°C for 15 s and 60°C for 30 s (for 40 cycles) and a dissociation curve (melting curve) in the range of 60–95°C,ref. biof2122-bib-0040 to evaluate the specificity of the amplification products. After assigning a fluorescence threshold above background (threshold cycle number or C t), differences in threshold cycle number were used to quantify the relative amount of each PCR target. The relative expression of different amplicons was calculated by the delta–delta C t (ΔΔC t) method and was converted to relative expression ratio (2−ΔΔCt) for statistical analysis.ref. biof2122-bib-0041 All data were normalized to the endogenous reference genes β‐actin and glyceraldehyde‐3‐phosphate dehydrogenase (GAPDH). The specific primers used were designed with Primer3 and ordered from Integrated DNA Technologies (IDT; Coralville, IA, USA), and their specific sequences are indicated in Table S2.
Western blotting
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then treated with CBN at 2.5 μM for 24 h. Cell pellets were collected and then lyzed in ice‐cold RIPA lysis buffer (Thermo Fisher Scientific Company, Waltham, MA, USA) in combination with the protease inhibitor cocktail 100× (Sigma Aldrich, St. Louis, MO, USA). The amount of protein was determined by the Bio‐Rad Protein assay (Bio‐Rad Laboratories, Hemel Hempstead, UK). An equal amount of protein (60 μg) for each sample was loaded onto 10% sodium dodecyl sulfate (SDS)–polyacrylamide gels and blotted onto polyvinylidene fluoride (PVDF) sheets (Amersham Biosciences, Piscataway, NJ, USA). Membranes were blocked with 5% non‐fat dried milk for 1 h at room temperature, and then incubated with suitable primary antibodies. Detection was performed using Azure Biosystems c400 (Dublin, CA, USA). The primary antibodies used were obtained from various sources, as detailed in Table S3.
CB1
binding assay
HaCaT cells were seeded in 12‐well plates at a density of 2 × 105 cells per well under the following conditions, each in triplicate: total binding (TB), non‐specific binding (NSB), CB1 receptor antagonist (SR141716), and blanks (Bk). Cells were then treated with 2.5 μM CBN for 24 h. After two washes with 1 mL PBS, each well was incubated with 500 μL of incubation buffer (50 mM Tris–HCl, 5 mM MgCl2, 1 mM CaCl2, 0.2% BSA, and pH 7.4) preheated to 37°C for 15 min. The incubation buffer contained either 1 μM non‐radiolabelled CP55.940 for NSB wells, or 0.1 μM SR141716 for SR1 wells.ref. biof2122-bib-0037, ref. biof2122-bib-0042 Subsequently, 2.5 nM [3H]CP55.940 was added to all wells, and the plates were incubated for 1 h at 37°C. This step allowed the radiolabeled ligand to compete with the pre‐incubated ligands for binding sites on the CB1 receptors. Then, the buffer was removed and the cells were washed again with an ice‐cold washing buffer (50 mM Tris–HCl, 500 mM NaCl, 0.1% BSA, and pH 7.4) to remove unbound radioactive material. Finally, 0.5 M NaOH (500 μL) was added to each well to lyse the cells. Cell lysates were transferred to 10 mL scintillation vials containing liquid scintillation cocktail, and radioactivity was measured by using a scintillation β‐counter (Tri‐Carb 2810 TR, Perkin Elmer, Waltham, MA, USA) as previously described.ref. biof2122-bib-0043
TRPV1 receptor activity assay
HaCaT cells (1 × 106 cells) in suspension were incubated with 4 μM of the selective intracellular fluorescent probe Fluo‐3 AM containing 0.02% Pluronic F‐127 in Opti‐MEM medium, for 15 min at 25°C. Then, cells were washed in Tyrode’s buffer (145 mM NaCl; 2.5 mM KCl; 1.5 mMCaCl2; 1.2 mM MgCl; 10 mM D‐glucose, 10 mM HEPES, and pH 7.4), resuspended in 2 mL of Tyrode’s buffer and transferred to the quartz cuvette of the LS50B spectrofluorometer (Perkin Elmer, Waltham, MA, USA). Fluorescence was measured at 25°C (excitation at λ = 488 nm; emission at λ = 516 nm) from HaCaT cells pre‐incubated with CBN at 2.5 μM, and then stimulated with the selective TRPV1 agonist capsaicin (1 μM), as reported.ref. biof2122-bib-0044 TRPV1 activity was expressed as fluorescence intensity (arbitrary units, AU) per 106 cells, as reported.ref. biof2122-bib-0035
Enzyme assays
NAPE‐PLD assay
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then treated with CBN at 2.5 μM for 24 h. Cell pellets were collected and lyzed in a reaction mixture containing 50 mM Tris–HCl, 0.05% Triton X‐100 pH 8.0, and 10 μM of fluorogenic substrate PED6. Cell homogenates were pre‐incubated with the specific NAPE‐PLD inhibitor ARN19874 at 3.8 mMref. biof2122-bib-0045 for 30 min at room temperature, to fully erase enzyme activity as a control. Fluorescence values were measured with excitation at λ = 485–488 nm, and emission at λ = 530 nm at 37°C, by means of continuity kinetics with 30‐s intervals for 30 min in an Enspire multimode plate reader (Perkin Elmer, Waltham, MA, USA), as reported.ref. biof2122-bib-0046 NAPE‐PLD activity was expressed as fluorescence intensity (arbitrary units, AU) per min per mg of protein, as reported.ref. biof2122-bib-0035
FAAH assay
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then treated with CBN at 2.5 μM for 24 h. Cell pellets were collected and FAAH activity was determined in membranes isolated from these cells. Briefly, membranes were incubated with 10 μM [3H]AEA and 50 mM Tris–HCl, 0.05% BSA at pH 7.4 at 37°C in water bath for 30 min. The reaction was blocked with 1 mL chloroform/methanol (1:1; v/v) and centrifuged to 900g for 10 min at 4°C. The aqueous (top) layer was collected and transferred into scintillation vials. The amount of [3H]ethanolamine released was expressed as pmol per min per mg of protein, as reported.ref. biof2122-bib-0047
DAGLα/β assay
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then treated with CBN at 2.5 μM for 24 h. Cell pellets were collected and DAGLα/β activity was determined in membranes isolated from these cells, by using the synthetic substrate pNPB (8 mM) in 50 mM HEPES pH 7.3, at room temperature for 20 min.ref. biof2122-bib-0048 The DAGLα/β inhibitor KT172 (1 μM) was used to full erase enzyme activity as a control, as reported.ref. biof2122-bib-0048
MAGL assay
HaCaT cells were seeded into six‐well plates at a density of 4 × 105 per well, incubated overnight, and then treated with CBN at 2.5 μM for 24 h. Cell pellets were collected and MAGL activity was determined in membranes isolated from these cells, by using the synthetic substrate arachidonoyl‐1‐thio‐glycerol (200 μM) in 10 mM Tris/1 mM EDTA pH 7.2, at 4°C for 15 min, and the MAGL inhibitor JZL184 (4 μM), to full erase enzyme activity, as reported.ref. biof2122-bib-0048
Enzyme‐linked immunosorbent assay
HaCaT cells were seeded into 12‐well plates at a density of 2 × 105 per well, incubated overnight and then treated as follows: untreated (control) cells, 2.5 μM CBN, 5.0 μg/mL LPS, 10 μM HC, and 2.5 μM CBN. In addition, HaCaT cells were exposed to CBN in combination with the selective CB1 and TRPV1 antagonists (SR141716A at 0.1 μM and CPZ at 5.0 μM, respectively) for 24 h in the case of IL‐31 and for 48 h in the case of IL‐12. After each treatment, supernatants (medium) of each condition were collected, centrifuged at 4°C at 300g for 5 min and stored at −80°C until the assay. Cytokine quantification was performed using the DuoSet ELISA kit (#DY201, #DY208, #DY217B, #DY1270, and #DY2824, R&D Systems, Minneapolis, MN, USA), following the manufacturer’s instructions. Briefly, 96‐well plates were coated overnight with the specific capture antibody for each interleukin and then blocked with 1% BSA. Selective standard proteins in duplicate and samples in triplicate were added and incubated for 2 h. The standard curve for each interleukin was generated by using serially diluted standard proteins at known concentrations as provided by the manufacturer, and the calibration curves obtained were used to interpolate sample values. Each well with standards and/or samples was incubated with the specific detection antibody for 2 h. The streptavidin–HRP was added and incubated for 20 min, followed by the substrate solution for another 20 min, and then the reaction was stopped with the specific Stop Solution (1 M H2SO4). Optical densities were determined using an Enspire microplate reader (Perkin Elmer, Waltham, MA, USA) at the specific wavelengths of 450 and 570 nm, as reported.ref. biof2122-bib-0036
MAPK signaling pathway array
HaCaT cells were seeded into 100 mm plates at a density of 8 × 105 per well, incubated overnight and then exposed to 24 h treatment with LPS at 5.0 μg/mL, positive control HC at 10 μM, CBN at 2.5 μM both alone and in combination with the selective CB1 and TRPV1 antagonists (SR141716A at 0.1 μM and CPZ at 5.0 μM, respectively). The signaling pathway was analysed through a C‐Series Human/Mouse MAPK phosphorylation array, according to manufacturer recommendations (#AAH‐MAPK‐1‐8, RayBiotech Inc., Peachtree Corners, GA, USA). Briefly, proteins were extracted from HaCaT cell pellets and quantified using the BCA method. 500 μg of proteins were added into each nitrocellulose membrane coated with the specific antibody and incubated overnight at 4°C. The day after, each well was incubated with the specific detection antibody and with streptavidin–HRP for 2 h at room temperature. Chemiluminescent readings were taken using a C‐DiGit Blot Scanner (LI‐COR Bioscience, Lincoln, NE, USA), and densitometry data were extracted using Image Studio™ software (LI‐COR Bioscience, Lincoln, NE, USA). Readings were normalized to the positive loading controls, and the membrane background signal was subtracted, as reported.ref. biof2122-bib-0036
Statistical analysis
Data were analysed using the GraphPad Prism 8 program (GraphPad Software, La Jolla, CA, USA) and were reported as means ± SEM of three independent experiments. Statistical analysis was performed using the unpaired t‐test, multiple t‐test, and the one‐way analysis of variance (ANOVA), according to the data set, followed by a Bonferroni post‐hoc test. A value of p < 0.05 was considered statistically significant.
RESULTS
CBN modulates distinct ECS elements at gene and protein levels
First, different concentrations of CBN—from 0.5 to 25 μM—were tested at three timepoints—6, 12, and 24 h—on HaCaT cells, through cell viability and apoptosis assays (Figure S1). A dose–response curve was plotted to determine the half‐maximal inhibitory concentration (IC50) of CBN at each timepoint, as indicated in Table S1. Based on these preliminary data, the non‐cytotoxic dose of 2.5 μM at 24 h (i.e., half of the calculated IC50) was selected to perform all subsequent analyses. Thus, the expression of ECS elements [eCB‐binding receptors (CB1, CB2, GPR55, TRPV1, and PPARα/γ/δ), and the main AEA (NAPE‐PLD, FAAH), and 2‐AG (DAGLα/β, MAGL) metabolic enzymes] was evaluated, at both mRNA—through quantitative real‐time polymerase chain reaction (RT‐qPCR)—and protein level—through Western blotting—in HaCaT cells after treatment with 2.5 μM CBN for 24 h. Notably, no alterations were found at gene level for any of the ECS elements tested, with the exception of CB1 which was significantly increased by CBN compared to control cells (p < 0.01 vs. CTRL) (Figure 1).
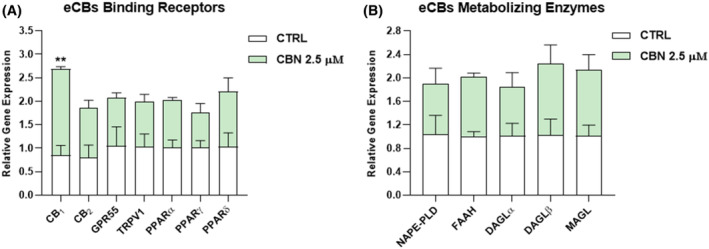
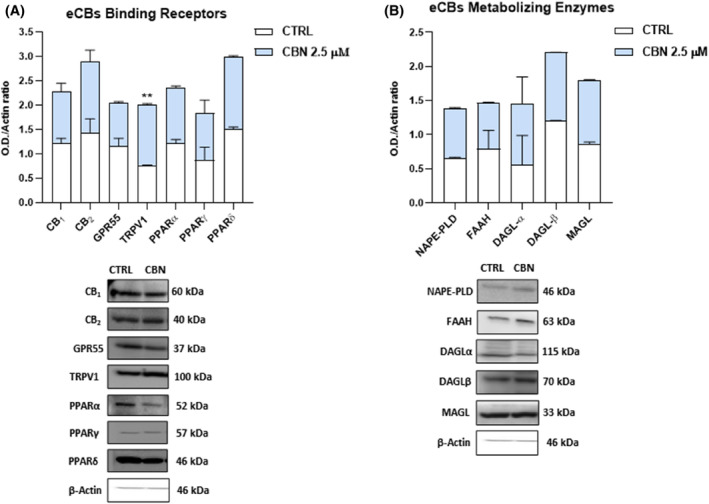
As for the protein expression, only that of TRPV1 was significantly increased (p < 0.01 and p < 0.05, respectively vs. CTRL) in HaCaT cells exposed to 2.5 μM CBN, while the other ECS components remained unaffected under the same experimental conditions (Figure 2).
CBN modulates ECS at the functional level
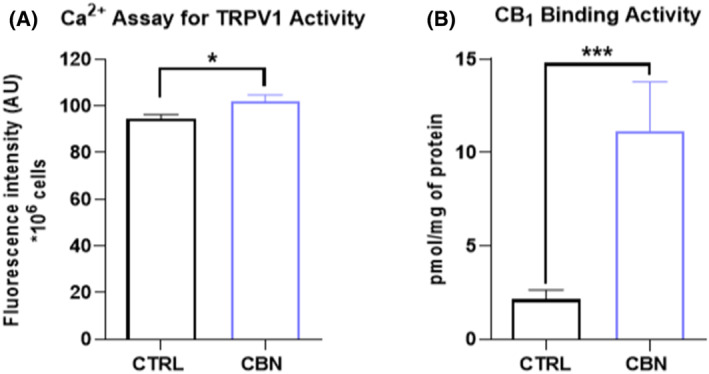
Then, the effect of CBN on functional activity of specific eCBs‐binding receptors and eCBs metabolic enzymes was investigated. In particular, the modulation of TRPV1 channel and CB1 receptor was assessed through specific intracellular Ca2+ release and radioligand binding assays, respectively. TRPV1 was activated by CBN in HaCaT cells, as shown by the significant increase in the calcium indicator (Fluo 3‐AM) fluorescence intensity in CBN‐treated cells (p < 0.05 vs. CTRL) (Figure 3A). Similarly, the CBN‐mediated activation of CB1 receptor was demonstrated by the significant increase (p < 0.001 vs. CTRL) in the binding activity, up to 3‐fold compared to control cells (Figure 3B).
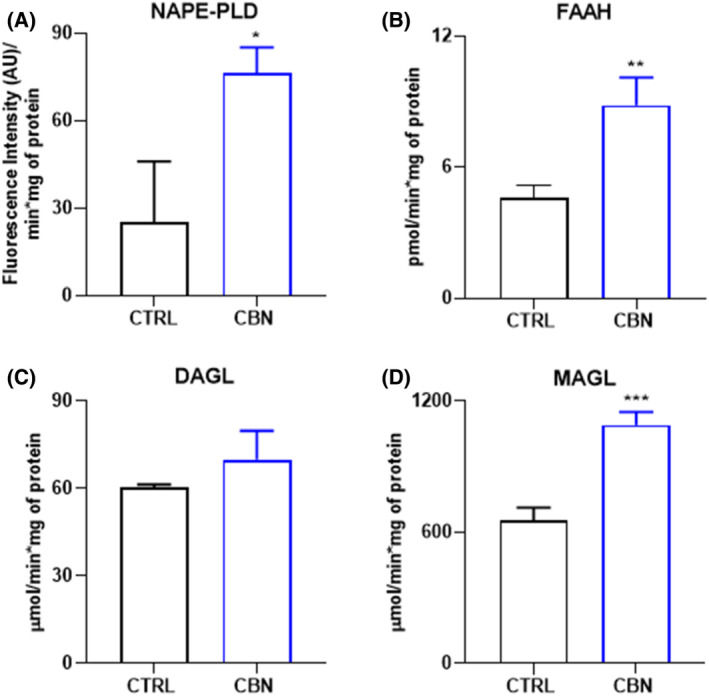
The activity of AEA and 2‐AG metabolic enzymes was evaluated through specific enzymatic assays under the same experimental conditions. At 2.5 μM, CBN stimulated AEA metabolism since both NAPE‐PLD and FAAH activities increased in HaCaT cells compared to controls (p < 0.05 and p < 0.01, respectively vs. CTRL) (Figure 4A,B). As for 2‐AG metabolism, MAGL activity was significantly increased by CBN (p < 0.001 vs. CTRL), while no changes were observed for DAGLα/β activity (Figure 4C,D).
CBN prevents keratinocyte inflammation via TRPV1
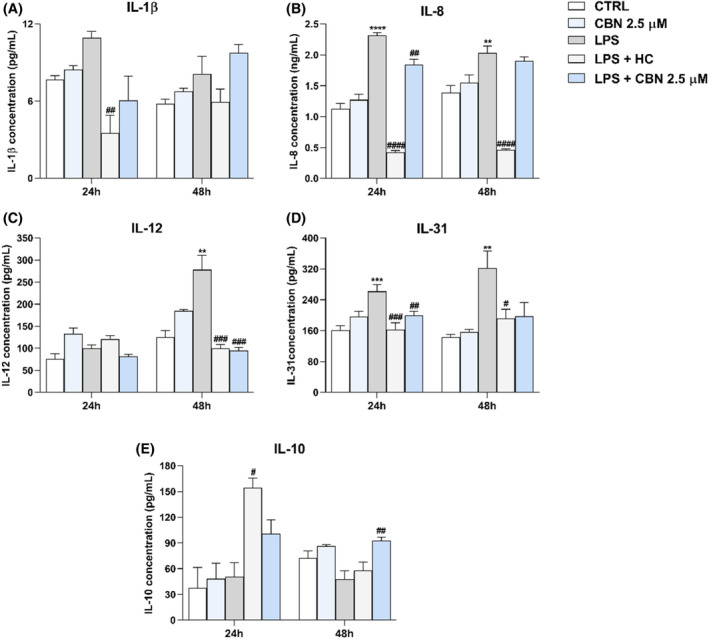
To ascertain whether CBN may have anti‐inflammatory properties in human keratinocytes, as previously shown for other pCBs,ref. biof2122-bib-0036 the release of pro‐inflammatory ILs (IL‐1β, IL‐8, IL‐12, and IL‐31), and of the anti‐inflammatory IL‐10 was analysed through enzyme‐linked immunosorbent (ELISA) assay in inflamed HaCaT cells exposed to 5.0 μg/mL of LPS, with or without 2.5 μM CBN for 24 and 48 h, which represents the two timepoints previously used to set‐up the in vitro inflammatory model of keratinocytes.ref. biof2122-bib-0036 LPS treatment markedly increased the levels of all pro‐inflammatory cytokines, except for IL‐1β and for IL‐12 at 24 h. Notably, 2.5 μM CBN in combination with LPS significantly reverted the increase of IL‐8 (p < 0.01 vs. LPS) at 24 h of treatment, although to a lesser extent than the anti‐inflammatory compound hydrocortisone (HC), used as a positive control (Figure 5A,B). The anti‐inflammatory action of 2.5 μM CBN was more effective on IL‐12 and IL‐31 levels; indeed, 2.5 μM CBN significantly reverted the LPS‐mediated increase in IL‐12 (p < 0.001 vs. LPS) and IL‐31 (p < 0.01 vs. LPS) at 48 and 24 h, respectively, much alike HC (Figure 5C,D). In addition, IL‐31 levels were reduced by CBN at 48 h, yet in a non‐significant manner (Figure 5D). Moreover, the anti‐inflammatory IL‐10 was weakly modulated by LPS alone, whereas 2.5 μM CBN significantly increased its level (p < 0.01 vs. LPS) when cells were exposed to both LPS and CBN for 48 h (Figure 5E). Incidentally, treatment of HaCaT cells with CBN alone did not significantly affect the release of any of the tested cytokines (Figure 5).
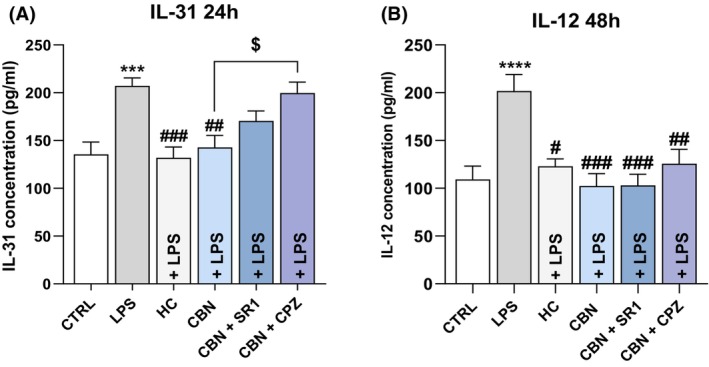
To further investigate whether CBN may regulate IL‐12 and IL‐31 release in an ECS‐dependent manner, HaCaT cells were pretreated also with SR141716A (SR1)—selective antagonist of CB1—and capsazepine (CPZ)—selective TRPV1 antagonist—under inflammatory conditions (Figure 6A,B). Interestingly, CPZ significantly restored the anti‐inflammatory effect of CBN on IL‐31 release (p < 0.05 vs. CBN) in HaCaT cells exposed to LPS for 48 h, suggesting an engagement of TRPV1 (Figure 6A). Instead, the action of CBN on IL‐12 was not mediated by CB1 nor by TRPV1, as the reduction of IL‐12 caused by CBN was not affected by either SR1 or CPZ (Figure 6B).
Expression of mitogen‐activated protein kinases in inflamed keratinocytes
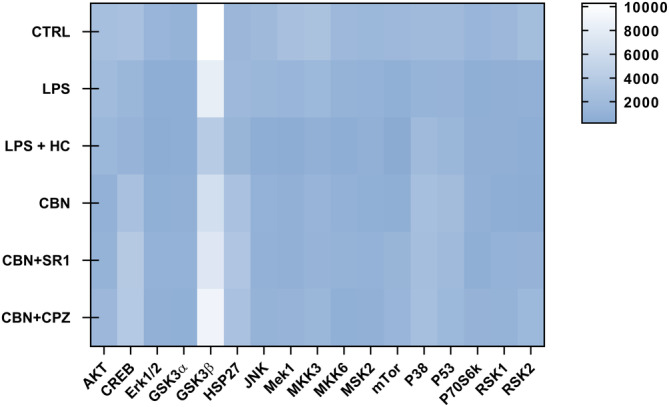
Since mitogen‐activated protein kinases (MAPK) pathway regulates the inflammatory responses of several cell types—keratinocytes included,ref. biof2122-bib-0049 the expression of 17 proteins of the MAPK signaling route was investigated in inflamed HaCaT cells exposed to CBN, alone or in combination with selective CB1 and TRPV1 antagonists, by using a human phosphorylation array (for details see Figure S2). The overall expression pattern of phosphorylated MAPK proteins was found to be low under most experimental conditions, apart from glycogen synthase kinase 3β (GSK3β) that was high in all experimental groups (Figure 7). Of note, GSK3β is the only MAPK analysed to be inactive in the phosphorylated form,ref. biof2122-bib-0050 and its expression was found to be modulated (yet not significantly) under all experimental conditions (Figure 7). As for the other MAPK proteins, only minor alterations in the expression of cyclic adenosine monophosphate response element‐binding protein (CREB), heat shock protein 27 (HSP27), p38 mitogen‐activated protein kinase (p38), and tumor protein p53 (p53) were observed in inflamed HaCaT cells treated with CBN in the presence of SR1 or CPZ, compared to LPS alone (Figure 7).
DISCUSSION AND CONCLUSION
To date, the effects of cannabis on skin physiology and pathology have focused on the main pCBs CBD and THC,ref. biof2122-bib-0051, ref. biof2122-bib-0052, ref. biof2122-bib-0053 while little is known about minor pCBs like CBN. Recently, we have shown that non‐psychotropic pCBs CBG, CBC, THCV, and CBGA differently modulate gene and protein expression of distinct ECS elements, as well as their functional activity in HaCaT cells.ref. biof2122-bib-0035 In line with this, here we demonstrate that in the same human keratinocytes CBN—used at a non‐cytotoxic half‐IC50 dose—increases gene and protein expression of specific eCBs‐biding receptors (i.e., CB1 and TRPV1, respectively), without modulating gene and protein expression of metabolic enzymes of these endogenous lipids. Of note, differences between gene and protein expression of ECS elements are not unprecedented.ref. biof2122-bib-0032, ref. biof2122-bib-0035, ref. biof2122-bib-0054, ref. biof2122-bib-0055 Such discrepancies may be due to distinct half‐lives of mRNAs and proteins, which are caused by different degrading processesref. biof2122-bib-0056 that lead to independent stability profiles.ref. biof2122-bib-0057, ref. biof2122-bib-0058 At the functional level, CBN upregulates AEA metabolism, by stimulating the activity of NAPE‐PLD and FAAH, and promotes the activity of the degradative enzyme of 2‐AG, MAGL, but not that of its biosynthetic enzymes DAGLα/β. In addition, our functional data show that CB1 binding is significantly increased by CBN, a finding that is in keeping with previous literature data showing CBN activity on CB1 in monkey kidney (COS‐7) transfected cells, as well as in rat brain synaptosomal preparations.ref. biof2122-bib-0059 The engagement of TRPV1 by CBN was also investigated, because CBN was shown to be weakly effective on TRPV1, relatively potent on the transient receptor potential ankyrin 1 (TRPA1), and only weakly active on the TRP vanilloid receptor 2 (TRPV2) in HEK‐293 cells stably overexpressing recombinant rat or human TRP channels.ref. biof2122-bib-0060 Our present data extend previous findings to human keratinocytes, that constitutively express the TRPV1 channel, supporting the concept that CBN may have a promising pharmacological profile for skin‐related conditions by engaging this eCBs‐binding receptor channel. In line with this, we investigated the effects of CBN in an inflamed keratinocyte model consisting of HaCaT cells exposed to LPS.ref. biof2122-bib-0036 It is well known that the process of inflammation is orchestrated by inflammatory mediators, and a typical response evoked by pCBs is the dysregulation of cytokine production by immune cells with an unbalance between those produced by T‐helper subsets Th1 and Th2.ref. biof2122-bib-0061, ref. biof2122-bib-0062 Here, we chose to evaluate the effect of CBN in our inflamed keratinocytes model on the release of some of the main cytokines known to be involved in the pathogenesis of skin diseases.ref. biof2122-bib-0063 Consistently, we found a reduction by CBN in the release of some pro‐inflammatory ILs at different time points of HaCaT stimulation (i.e., IL‐8 and IL‐31 at 24 h and IL‐12 at 48 h), while an increase of anti‐inflammatory IL‐10 release at 48 h. Among these, IL‐12 is a key cytokine involved in the regulation of epidermal functions,ref. biof2122-bib-0064 with possible inflammatoryref. biof2122-bib-0065 and protectiveref. biof2122-bib-0066, ref. biof2122-bib-0067 roles in the skin. The increase in the IL‐12p70 release in psoriasis and during the chronic phase of atopic dermatitis is thought to be responsible for controling the immune and inflammatory response, by promoting the differentiation of Th0 lymphocytes into Th1‐type cells.ref. biof2122-bib-0067, ref. biof2122-bib-0068, ref. biof2122-bib-0069 On the other hand, recent studies suggested a protective effect of IL‐12 in a psoriatic mouse model, due to the activation of a transcriptional programme that limits skin inflammation by engaging IL‐12 receptor signaling.ref. biof2122-bib-0066 Our data showed an increase in IL‐12 levels under inflammatory conditions, thus corroborating its role in inflammation, that was reverted by CBN. Moreover, IL‐31 is one of the most relevant cytokines in the context of the pathogenesis of pruritus.ref. biof2122-bib-0070, ref. biof2122-bib-0071 Indeed, IL‐31 expression was found to be increased in lesional and non‐lesional skin of patients with atopic dermatitis,ref. biof2122-bib-0072, ref. biof2122-bib-0073, ref. biof2122-bib-0074 and it is able to induce severe pruritus in mice where levels of IL‐31 correlated with scratching behaviour.ref. biof2122-bib-0075 In addition to its pruritogenic function, IL‐31 also directly inhibits keratinocyte differentiation by downregulating the expression of proteins involved in barrier and differentiation processes, overall leading to the disruption of the epidermal barrier function.ref. biof2122-bib-0076, ref. biof2122-bib-0077, ref. biof2122-bib-0078 In the light of the preminent role of IL‐31 in skin inflammation, the ability of CBN to decrease its secretion appears noteworthy. More in general, the ability of CBN to decrease or increase the pro‐ and anti‐inflammatory outputs has been previously reported in different paradigms, for example in a murine model of ovalbumin‐sensitized allergic asthma where CBN blocked the production of ILs 2, 4, 5, 13 and decreased allergen mucus production.ref. biof2122-bib-0010 Similarly, CBN was shown to inhibit pro‐inflammatory cytokine production from oral bacteria‐exposed human monocytes and epithelial cells.ref. biof2122-bib-0079 More recently, CBN was also found to exert anti‐inflammatory effects by influencing different stages of gene expression: transcription, post‐transcriptional regulation, translation, and post‐translational regulation in human macrophages.ref. biof2122-bib-0080 It should be recalled that pCBs and pCB‐like compounds have proven potent anti‐inflammatory activity through mechanisms that are not yet fully understood. Under some circumstances, activation of CB1/2 receptors provides anti‐inflammatory responses, and particularly CB2 appears to be the main player in allergic contact dermatitisref. biof2122-bib-0081, ref. biof2122-bib-0082 and immune suppression of oral pathogens, where a CB2/phosphoinositide 3‐kinase (PI3K) axis is activated by CBN (as well as by CBD and THC).ref. biof2122-bib-0079 Conversely, CB1/2 receptors may not even be engaged in these processes, as demonstrated by a study where the topical application of THC effectively improved allergic contact dermatitis in both wild‐type and CB1/CB2‐deficient mice.ref. biof2122-bib-0051 In order to fill this gap knowledge, we decided to focus the attention on IL‐12 and IL‐31 to interrogate a possible correlation with the ECS. In this context, we previously observed beneficial effects induced by other minor pCBs (e.g., cannabigerol, cannabichromene, Δ9‐tetrahydrocannabivarin, and cannabigerolic acid) on IL‐31 via ECS involvement.ref. biof2122-bib-0036 Here, we obtained similar results with CBN, which was able to revert the inflammatory secretion of IL‐31 through the TRPV1 channel, while CB1 did not appear to be involved. Instead, the effects of CBN on the release of IL‐12 were found to be ECS‐independent. The correlation between IL‐31 and TRPV1 channel—in the presence of CBN—seems to call for particular attention, since TRPV1 is known to have pruritic functions and to increase in atopic dermatitis‐like skin lesions, both in humans and in a mouse model of psoriasiform dermatitis.ref. biof2122-bib-0083, ref. biof2122-bib-0084, ref. biof2122-bib-0085 There is also literature evidence that attests beneficial effects of TRPV1‐targeting. Notably, the eCB‐like compound PEA was found to inhibit allergic contact dermatitis in mice, as well as cytokine expression in human HaCaT cells, through TRPV1‐associated mechanisms.ref. biof2122-bib-0086 In addition, some pCBs stimulate TRPV1 channels, as reported by us and others.ref. biof2122-bib-0035, ref. biof2122-bib-0060, ref. biof2122-bib-0087, ref. biof2122-bib-0088 Overall, these results and the key role of IL‐31 in inflammatory skin diseases, support the potential of TRPV1‐targeting (e.g., by CBN) for therapeutic purposes, for instance to treat itch‐associated skin conditions.ref. biof2122-bib-0089 Interestingly, antibodies against IL‐31 have shown to ameliorate the scratching behaviour in a mouse model of atopic dermatitis,ref. biof2122-bib-0090 and patients affected by chronic spontaneous urticaria responded to a treatment able to reduce IL‐31 serum levels.ref. biof2122-bib-0091
In this study, we also sought to elucidate signaling events that could explain the CBN‐mediated suppression of inflammation by analysing the modulation of phosphorylated proteins of the MAPK pathway. The activation of this pathway by LPS involves a series of phosphorylation events leading to the induction of inflammatory mediators, cytokines included.ref. biof2122-bib-0092, ref. biof2122-bib-0093 Among the different MAPKs investigated in our inflamed model, the phosphorylated and inactive form of GSK3β appeared to be the most expressed in HaCaT cells under CBN stimulation and all other experimental conditions. A regulatory role of GSK3β in the modulation of inflammation has been already suggested.ref. biof2122-bib-0094, ref. biof2122-bib-0095 In particular, the knock‐down of GSK3β was found to significantly reduce LPS‐associated pro‐inflammatory cytokines and death of human monocytes. Interestingly, cannabinoids have been reported to suppress neuroinflammation in Alzheimer’s disease with a downregulation of phospho‐GSK3β.ref. biof2122-bib-0096 In contrast, the anti‐inflammatory signal evoked by CBN (as well as by CBD and THC) to suppress oral pathogens does not appear to be executed via the GSK3β‐dependent cholinergic anti‐inflammatory pathway.ref. biof2122-bib-0079 In our study, other phosphorylated active proteins of the MAPK family appear to be involved in the CBN anti‐inflammatory response, yet to a lesser and non‐significant extent.
In summary, in this investigation CBN has been shown to be active on inflamed human keratinocytes via TRPV1 channels, thus supporting a potential therapeutic effect of this minor pCB in the treatment of inflammatory skin diseases. It should be recalled that CBN has been indeed suggested as a potential component of topical applications to be used against psoriasis, where it inhibits keratinocyte proliferation through as yet unclear CB1/2‐independent mechanisms.ref. biof2122-bib-0097 A recent study has also shown anti‐melanoma, anti‐melanogenic, and anti‐tyrosinase properties of CBN (and other pCBs), thus making them useful as novel cosmeceutical products for skin care.ref. biof2122-bib-0098 Moreover, a CBN‐based therapy has been recently shown to be effective in the treatment of epidermolysis bullosa (EB), a group of rare medical conditions characterized by easy blistering of the skin and mucous membranes.ref. biof2122-bib-0099 This therapy was recently completed in a phase 2 clinical trial, aimed at assessing its potential as a topical treatment for EB, where it may help to relieve hallmark symptoms such as itch in a subset of EB Simplex patients.ref. biof2122-bib-0099 On a final note, it can be recalled that the therapeutic potential of CBN goes beyond the skin, because another CBN‐based topical eye drop formulation has been demonstrated to be effective in the treatment of glaucoma, where it may prevent the inflammation leading to elevated intraocular pressure and hence damage to the retinal ganglion cells.ref. biof2122-bib-0100
Taken together, our data suggest that CBN may hold true therapeutic potential to treat different human skin diseases. Such a biological activity of CBN occurs through engagement of selected elements of the endocannabinoid system—in particular TRPV1—a finding that paves the way to the development of distinct formulations of cannabis extracts for selected therapeutic applications.
AUTHOR CONTRIBUTIONS
C. R. and M. M. contributed to conceptualization, writing‐reviewing and editing. C. D. M. and D. T. worked on validation, draft preparation and visualization. C. D. M, D. T., S. S., F. C., and C. B. A. worked on methodology and investigation. C.D.M, D.T., S.S., F.C, C.B.A. and A.T. worked on formal analysis. C. D. M., D. T., E. H., and S. K. dealt with resources. M. M. was in charge of supervision, project administration, and funding acquisition. All authors reviewed and approved the final version of the manuscript.
FUNDING INFORMATION
This work was supported by InMed Pharmaceuticals Inc. (Vancouver, Canada) under a collaborative research agreement (No. PCI0029) with Professor Mauro Maccarrone. The funder had no role in the design of the study, in the collection, analyses or interpretation of data, nor in the decision to publish the results.
CONFLICT OF INTEREST STATEMENT
The authors declare that the InMed Pharmaceuticals is commercializing cannabinoid‐based therapies. Eric Hsu is the Senior Vice President of this company; Salam Kadhim is a former Senior Scientist of Preclinical R&D, and Mauro Maccarrone is a Scientific Advisory Board member.
References
- Phytocannabinoids: a unified critical inventory.. Nat Prod Rep., 2016. [PubMed]
- Cannabis for medical use: versatile plant rather than a single drug.. Front Pharmacol., 2022. [PubMed]
- Medical Cannabis: a plurimillennial history of an evergreen.. J Cell Physiol., 2019. [PubMed]
- 4 ElSohly MA , Radwan MM , Gul W , Chandra S , Galal A . Phytochemistry of Cannabis sativa L. In: Kinghorn AD , Falk H , Gibbons S , Kobayashi J , editors. Phytocannabinoids, progress in the chemistry of organic natural products. Cham: Springer International Publishing; 2017. p. 1–36.
- Major phytocannabinoids and their related compounds: should we only search for drugs that act on cannabinoid receptors?. Pharmaceutics., 2021. [PubMed]
- Minor cannabinoids: biosynthesis, molecular pharmacology and potential therapeutic uses.. Front Pharmacol., 2021. [PubMed]
- Cannabinol: history, syntheses, and biological profile of the greatest “minor” cannabinoid.. Plants., 2022. [PubMed]
- 8 Huestis MA . Pharmacokinetics and metabolism of the plant cannabinoids, δ 9‐tetrahydrocannibinol, cannabidiol and cannabinol. In: Pertwee RG , editor. Cannabinoids, handbook of experimental pharmacology. Berlin, Heidelberg: Springer; 2005. p. 657–690.
- 9 Morales P , Hurst DP , Reggio PH . Molecular targets of the phytocannabinoids: a complex picture. In: Kinghorn AD , Falk H , Gibbons S , Kobayashi J , editors. Phytocannabinoids, progress in the chemistry of organic natural products. Cham: Springer International Publishing; 2017. p. 103–131.
- Attenuation of the ovalbumin‐induced allergic airway response by cannabinoid treatment in A/J mice.. Toxicol Appl Pharmacol., 2003. [PubMed]
- Goods and bads of the endocannabinoid system as a therapeutic target: lessons learned after 30 years.. Pharmacol Rev., 2023. [PubMed]
- Endocannabinoid signaling at the periphery: 50 years after THC.. Trends Pharmacol Sci., 2015. [PubMed]
- Endocannabinoid signaling and its regulation by nutrients.. BioFactors., 2014. [PubMed]
- Cannabinoid signaling in the skin: therapeutic potential of the “c(ut)annabinoid” system.. Molecules., 2019. [PubMed]
- The endocannabinoid system of the skin. A potential approach for the treatment of skin disorders.. Biochem Pharmacol., 2018. [PubMed]
- Occurrence and biosynthesis of endogenous cannabinoid precursor, N‐arachidonoyl phosphatidylethanolamine, in rat brain.. J Neurosci., 1997. [PubMed]
- Isolation and structure of a brain constituent that binds to the cannabinoid receptor.. Science., 1992. [PubMed]
- Formation and inactivation of endogenous cannabinoid anandamide in central neurons.. Nature., 1994. [PubMed]
- Identification of an endogenous 2‐monoglyceride, present in canine gut, that binds to cannabinoid receptors.. Biochem Pharmacol., 1995. [PubMed]
- 2‐Arachidonoylgylcerol: a possible endogenous cannabinoid receptor ligand in brain.. Biochem Biophys Res Commun., 1995. [PubMed]
- Molecular characterization of a phospholipase D generating anandamide and its congeners.. J Biol Chem., 2004. [PubMed]
- Mammalian enzymes responsible for the biosynthesis of N‐acylethanolamines.. Biochim Biophys Acta (BBA)—Mol Cell Biol Lipids., 2017
- Molecular characterization of an enzyme that degrades neuromodulatory fatty‐acid amides.. Nature., 1996. [PubMed]
- Cloning of the first sn1‐DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain.. J Cell Biol., 2003. [PubMed]
- Chemical proteomics maps brain region specific activity of endocannabinoid hydrolases.. ACS Chem Biol., 2017. [PubMed]
- Loss of retrograde endocannabinoid signaling and reduced adult neurogenesis in diacylglycerol lipase knock‐out mice.. J Neurosci., 2010. [PubMed]
- A chemical proteomic atlas of brain serine hydrolases identifies cell type‐specific pathways regulating neuroinflammation.. eLife., 2016. [PubMed]
- DAGLβ inhibition perturbs a lipid network involved in macrophage inflammatory responses.. Nat Chem Biol., 2012. [PubMed]
- Diacylglycerol lipase‐β is required for TNF‐α response but not CD8+ T cell priming capacity of dendritic cells.. Cell Chem Biol., 2019. [PubMed]
- Brain monoglyceride lipase participating in endocannabinoid inactivation.. Proc Natl Acad Sci., 2002. [PubMed]
- The endocannabinoid system of the skin in health and disease: novel perspectives and therapeutic opportunities.. Trends Pharmacol Sci., 2009. [PubMed]
- Chapter 17 regulation of gene transcription and keratinocyte differentiation by anandamide.. Vitamin Hormone., 2009
- Epigenetic control of skin differentiation genes by phytocannabinoids: epigenesis of skin genes by phytocannabinoids.. Br J Pharmacol., 2013. [PubMed]
- The endocannabinoid system, anandamide and the regulation of mammalian cell apoptosis.. Cell Death Diff., 2003
- Effects of rare phytocannabinoids on the endocannabinoid system of human keratinocytes.. Int J Mol Sci., 2022. [PubMed]
- Rare phytocannabinoids exert anti‐inflammatory effects on human keratinocytes via the endocannabinoid system and MAPK signaling pathway.. Int J Mol Sci., 2023. [PubMed]
- Endocannabinoids stimulate human melanogenesis via Type‐1 cannabinoid receptor.. J Biol Chem., 2012. [PubMed]
- Endocannabinoids modulate human epidermal keratinocyte proliferation and survival via the sequential engagement of cannabinoid receptor‐1 and transient receptor potential vanilloid‐1.. J Invest Dermat., 2011
- Lipid rafts control signaling of type‐1 cannabinoid receptors in neuronal cells.. J Biol Chem., 2005. [PubMed]
- Mutation detection using fluorescent hybridization probes and melting curve analysis.. Exp Rev Mol Diag., 2001
- Analysis of relative gene expression data using real‐time quantitative PCR and the 2−ΔΔCT method.. Methods., 2001. [PubMed]
- Characterization of the endocannabinoid system in human spermatozoa and involvement of transient receptor potential vanilloid 1 receptor in their fertilizing ability.. Endocrinology., 2009. [PubMed]
- Modulation of endocannabinoid‐binding receptors in human neuroblastoma cells by tunicamycin.. Molecules., 2019. [PubMed]
- The anti‐inflammatory mediator palmitoylethanolamide enhances the levels of 2‐arachidonoyl‐glycerol and potentiates its actions at TRPV1 cation channels: novel palmitoylethanolamide ‘entourage effects’.. Br J Pharmacol., 2016. [PubMed]
- Synthesis and characterization of the first inhibitor of N‐acylphosphatidylethanolamine phospholipase D (NAPE‐PLD).. Chem Commun., 2017
- A novel fluorogenic substrate for the measurement of endothelial lipase activity.. J Lipid Res., 2011. [PubMed]
- Confocal microscopy and biochemical analysis reveal spatial and functional separation between anandamide uptake and hydrolysis in human keratinocytes.. Cell Mol Life Sci., 2005. [PubMed]
- The endocannabinoid system is affected by cholesterol dyshomeostasis: insights from a murine model of Niemann Pick type C disease.. Neurobiol Dis., 2019. [PubMed]
- The role of mitogen‐activated protein kinase‐activated protein kinases (MAPKAPKs) in inflammation.. Genes., 2013. [PubMed]
- Phosphorylation and inactivation of glycogen synthase kinase 3β (GSK3β) by dual‐specificity tyrosine phosphorylation‐regulated kinase 1A (Dyrk1A).. J Biol Chem., 2015. [PubMed]
- Anti‐inflammatory activity of topical THC in DNFB‐mediated mouse allergic contact dermatitis independent of CB1 and CB2 receptors.. Allergy., 2013. [PubMed]
- Topical cannabinoids for the management of psoriasis vulgaris: report of a case and review of the literature.. J Drugs Dermatol., 2020. [PubMed]
- Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes.. J Clin Invest., 2014. [PubMed]
- Transcriptomic and proteomic analyses of mouse cerebellum reveals alterations in RasGRF1 expression following in vivo chronic treatment with delta 9‐tetrahydrocannabinol.. J Mol Neurosci., 2009. [PubMed]
- Characterization of the endocannabinoid system in mouse embryonic stem cells.. Stem Cell Dev., 2011
- Genetic architecture of protein expression and its regulation in the mouse brain.. BMC Genom., 2021
- Delayed correlation of mRNA and protein expression in rapamycin‐treated cells and a role for Ggc1 in cellular sensitivity to rapamycin.. Mol Cell Prot., 2010
- Protein expression regulation under oxidative stress.. Mol Cell Prot., 2011
- Cannabinol derivatives: binding to cannabinoid receptors and inhibition of adenylylcyclase.. J Med Chem., 1997. [PubMed]
- Effects of cannabinoids and cannabinoid‐enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes: novel pharmacology of minor plant cannabinoids.. Br J Pharmacol., 2011. [PubMed]
- The effects of cannabinoids on pro‐ and anti‐inflammatory cytokines: a systematic review of in vivo studies.. Cannabis Cannabinoid Res., 2021. [PubMed]
- Cannabinoids as novel anti‐inflammatory drugs.. Fut Med Chem., 2009
- Secreted immunoregulatory proteins in the skin.. J Dermatol Sci., 2018. [PubMed]
- Keratinocytes under fire of proinflammatory cytokines: bona fide innate immune cells involved in the physiopathology of chronic atopic dermatitis and psoriasis.. J Allergy., 2012
- Interleukin‐22, a TH17 cytokine, mediates IL‐23‐induced dermal inflammation and acanthosis.. Nature., 2007. [PubMed]
- IL‐12 protects from psoriasiform skin inflammation.. Nat Commun., 2016. [PubMed]
- The role of interleukins in the pathogenesis of dermatological immune‐mediated diseases.. Adv Ther., 2022. [PubMed]
- New insights into atopic dermatitis.. J Clin Invest., 2004. [PubMed]
- Interleukin‐12 and the regulation of innate resistance and adaptive immunity.. Nat Rev Immunol., 2003. [PubMed]
- IL‐31: state of the art for an inflammation‐oriented interleukin.. Int J Mol Sci., 2022. [PubMed]
- Cytokinocytes: the diverse contribution of keratinocytes to immune responses in skin.. JCI Insight., 2020. [PubMed]
- IL‐31: a new link between T cells and pruritus in atopic skin inflammation.. J Allergy Clin Immunol., 2006. [PubMed]
- Increased frequencies of IL‐31‐producing T cells are found in chronic atopic dermatitis skin.. Exp Dermat., 2012
- IL‐31 expression by inflammatory cells is preferentially elevated in atopic dermatitis.. Acta Derm Venereol., 2012. [PubMed]
- Interleukin 31, a cytokine produced by activated T cells, induces dermatitis in mice.. Nat Immunol., 2004. [PubMed]
- IL‐31 regulates differentiation and filaggrin expression in human organotypic skin models.. J Allergy Clin Immunol., 2012. [PubMed]
- Reduced filaggrin expression is accompanied by increased Staphylococcus aureus colonization of epidermal skin models.. Clin Exp Allergy., 2014. [PubMed]
- Control of the physical and antimicrobial skin barrier by an IL‐31–IL‐1 signaling network.. J Immunol., 2016. [PubMed]
- Marijuana‐derived cannabinoids trigger a CB2/PI3K axis of suppression of the innate response to oral pathogens.. Front Immunol., 2019. [PubMed]
- Anti‐inflammatory effects of minor cannabinoids CBC, THCV, and CBN in human macrophages.. Molecules (Basel, Switzerland)., 2023. [PubMed]
- Medical cannabis activity against inflammation: active compounds and modes of action.. Front Pharmacol., 2022. [PubMed]
- Anandamide suppresses proinflammatory T cell responses in vitro through type‐1 cannabinoid receptor–mediated mTOR inhibition in human keratinocytes.. J Immunol., 2016. [PubMed]
- The genetics of chronic itch: gene expression in the skin of patients with atopic dermatitis and psoriasis with severe itch.. J Invest Dermatol., 2018. [PubMed]
- TRPV1 mediates inflammation and hyperplasia in imiquimod (IMQ)‐induced psoriasiform dermatitis (PsD) in mice.. J Dermatol Sci., 2018. [PubMed]
- Cannabinoids and their receptors in skin diseases.. Int J Mol Sci., 2023. [PubMed]
- Protective role of palmitoylethanolamide in contact allergic dermatitis: palmitoylethanolamide effects in keratinocytes.. Allergy., 2009. [PubMed]
- Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide: cannabidiol, VR1 receptors and anandamide inactivation.. Br J Pharmacol., 2001. [PubMed]
- Nonpsychotropic plant cannabinoids, cannabidivarin (CBDV) and cannabidiol (CBD), activate and desensitize transient receptor potential vanilloid 1 (TRPV1) channels in vitro: potential for the treatment of neuronal hyperexcitability.. ACS Chem Neurosci., 2014. [PubMed]
- Role of the pruritic cytokine IL‐31 in autoimmune skin diseases.. Front Immunol., 2019. [PubMed]
- Anti‐interleukin‐31‐antibodies ameliorate scratching behaviour in NC/Nga mice: a model of atopic dermatitis.. Exp Dermatol., 2009. [PubMed]
- Successful omalizumab treatment in chronic spontaneous urticaria is associated with lowering of serum il‐31 levels.. J Eur Acad Dermatol Venereol., 2016. [PubMed]
- NF‐κB, inflammation, immunity and cancer: coming of age.. Nat Rev Immunol., 2018. [PubMed]
- Toll‐like receptors and the control of immunity.. Cell., 2020. [PubMed]
- Toll‐like receptor‐mediated cytokine production is differentially regulated by glycogen synthase kinase 3.. Nat Immunol., 2005. [PubMed]
- Tumor necrosis factor induces GSK3 kinase‐mediated cross‐tolerance to endotoxin in macrophages.. Nat Immunol., 2011. [PubMed]
- Effects of cannabidiol interactions with Wnt/beta‐catenin pathway and PPARγ on oxidative stress and neuroinflammation in Alzheimer’s disease.. Acta Biochim Biophys Sin., 2017. [PubMed]
- Cannabinoids inhibit human keratinocyte proliferation through a non‐CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis.. J Dermatol Sci., 2007. [PubMed]
- Cannabidiol and minor phytocannabinoids: a preliminary study to assess their anti‐melanoma, anti‐melanogenic, and anti‐tyrosinase properties.. Pharmaceuticals., 2023. [PubMed]
- INM‐755 Cannabinol (CBN) Cream.
- Cannabinol modulates neuroprotection and intraocular pressure: a potential multi‐target therapeutic intervention for glaucoma.. Biochim Biophys Acta (BBA)—Mol Basis Dis., 2022
