Inhibition of cannabinoid degradation enhances hippocampal contextual fear memory and exhibits anxiolytic effects
Abstract
Recent studies have demonstrated the pivotal involvement of endocannabinoids in regulating learning and memory, but the conclusions obtained from different paradigms or contexts are somewhat controversial, and the underlying mechanisms remain largely elusive. Here, we show that JZL195, a dual inhibitor of fatty acid amide hydrolase and monoacylglycerol lipase, can enhance the performance of mice in a contextual fear conditioning task and increase the time spent in open arms in the elevated zero maze (EZM). Although the effect of JZL195 on fear memory could not be inhibited by antagonists of cannabinoid receptors, the effect on the EZM seems to be mediated by CB1R. Simultaneously, hippocampal neurons are hyperactive, and theta oscillation power is significantly increased during the critical period of memory consolidation upon treatment with JZL195. These results suggest the feasibility of targeting the endocannabinoid system for the treatment of various mental disorders.
Article type: Research Article
Keywords: Biological sciences, Neuroscience, Behavioral neuroscience, Cognitive neuroscience
Affiliations: Key Laboratory of Modern Teaching Technology, Ministry of Education, Shaanxi Normal University, Xi’an 710000, China; Department of Histology and Embryology, School of Basic Medical Science, Xi’an Medical University, Xi’an 710000, China; Heze Hospital of Traditional Chinese Medicine, Heze 274000, China; Faculty of Education, Shaanxi Normal University, Xi’an 710000, China
License: © 2024 The Author(s) CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.isci.2024.108919 | PubMed: 38318362 | PMC: PMC10839683
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (216 KB)
Introduction
Memory deficits are prominent features of many mental disorders, such as anxiety,ref. bib1 insomnia,ref. bib2 and even Alzheimer’s disease (AD).ref. bib3 In recent decades, the development of various drugs that were anticipated to alleviate memory deficits but that eventually showed limited clinical benefits has been observed.ref. bib3 As a widely distributed neuromodulatory system, the endocannabinoid system (ECS) and its relative chemical compounds are involved in virtually all brain functions through either traditional cannabinoid receptor type 1 (CB1R) and type 2 (CB2R) or other G protein-coupled receptors (GPRs).ref. bib4 Retrograde control of synaptic transmission and plasticity is a powerful way through which the ECS regulates brain functions;ref. bib5 hence, the ECS may serve as a therapeutic target in neurological disorders. CB1R is the key mediator of endocannabinoid-elicited short- and long-term synaptic plasticity, which is regarded as the neurological basis of learning and memory, in the hippocampus, prefrontal cortex, and amygdala.ref. bib6,ref. bib7 Although sparsely expressed in the central nervous system (CNS), CB2Rs are also indispensable for memory recall.ref. bib8 Traditional cannabinoids (e.g., 9-tetrahydrocannabinol, THC) have been demonstrated to trigger memory impairmentref. bib9 and an increased incidence of mental diseases because of their psychotropic properties.ref. bib10 In this regard, cannabidiol (CBD), a nonpsychotropic Cannabis-derived compound, has been found to improve the performance of mice in memory-retrieval tasksref. bib11,ref. bib12 and has been applied in registered clinical trials for several diseases.ref. bib10,ref. bib13
The concentrations of endocannabinoids are collaboratively controlled by enzymes that catalyze the synthesis and hydrolysis of these metabolites. In particular, the hydrolysis of endocannabinoids, 2-arachidonoyl glycerol (2-AG) and anandamide (AEA), is catalyzed preferably by monoacylglycerol lipase (MAGL) and fatty acid aminohydrolase (FAAH), respectively.ref. bib14 Generally, inhibition of MAGL and FAAH causes an accumulation of 2-AG and AEA within synapses, leading to the reinforcement of cannabinoid-induced intracellular signaling and synaptic plasticity. Global MAGL knockout promotes hippocampus-dependent spatial memory recall in mice in a water maze test.ref. bib15 However, dual ablation of MAGL and FAAH through pharmacological inhibition or genetic manipulation results in a THC-like detrimental effect on memory, as evaluated by the performance of mice in the water maze task.ref. bib16 Recently, several reviews have addressed the role of the ECS as a new target for treating neurodegenerative disorders by alleviating either inflammatory responses or synaptic plasticity in the hippocampus.ref. bib17,ref. bib18,ref. bib19
To pharmacologically inhibit MAGL and FAAH individually or in combination, JZL184 (target to MAGL), URB597 (or PF-3845, target to FAAH), and JZL195 (target to both MAGL and FAAH) are often used. JZL184 typically inhibits MAGL and then results in an increased level of 2-AG, but at a high dose (40 mg/kg) it may also inhibit FAAH.ref. bib16 In contrast to JZL184 and URB597, JZL195 seems to work in a more complex way. JZL195 has also been demonstrated to modulate neuroinflammation,ref. bib20 locomotion,ref. bib21 pain,ref. bib22 and self-groomingref. bib23 through CB1Rs across species under certain circumstances. Although the role of the single drugs JZL184 and URB597 in fear conditioning and extinction has been investigated,ref. bib24 we failed to search the literature in PubMed about JZL195 when using keywords such as “JZL195,” “fear,” and “fear memory,” alone or combined. Therefore, showing the effect and underlying mechanism of JZL195 on fear memory, in terms of its potential as a clinical drug when targeting various mental disorders, is essential.
Due to these inconsistent results, it is of great necessity to probe the role of JZL195 in contextual fear memory formation and anxiety-like behavior. To address these issues, we used fear conditioning and the elevated zero maze (EZM) to assess fear memory and anxiety-like behavior, immunofluorescence and electrophysiology to assess neuronal activity, and pharmacology and trans-genetic mouse lines to assess the role of CB1R and CB2R. Overall, we found that the administration of JZL195, a dual inhibitor of FAAH and MAGL, could promote contextual fear memory consolidation and recall and exhibited anxiolytic effects.
Results
Inhibition of cannabinoid hydrolysis was anxiolytic and enhanced contextual but not cued fear memory
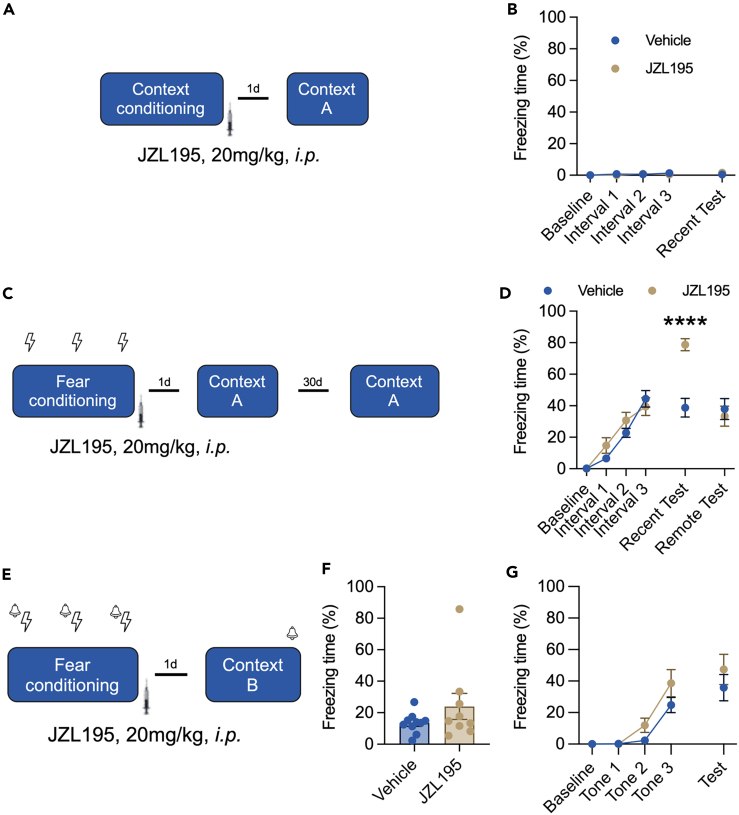
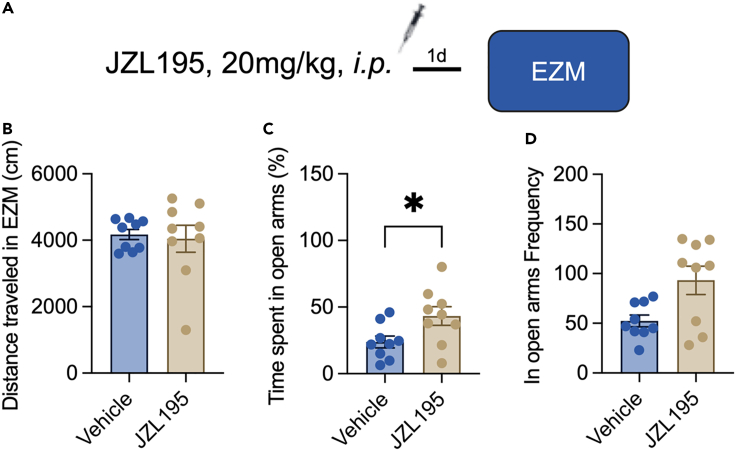
Fear memory has been studied in both cue-dependent and context-dependent fear conditioning paradigms. We found that systemic administration of JZL195 did not change the basal freezing time of mice in the absence of foot shocks in contextual conditioning recall (Figures 1A and 1B) and also the intact short-term memory (Figure S1). Then, we employed a contextual fear conditioning paradigm for the test. Compared with that of untreated mice, those treated with JZL195 displayed significantly prolonged freezing time on Day 2 in the recent recall test, and this increasing effect was abolished in the remote recall test (Figure 1D). Next, we explored the effect of JZL195 on cue fear conditioning paradigms. However, we failed to detect a significant difference between vehicle- and JZL195-treated mice in the cued fear memory recall test (Figures 1E–1G) or basal contextual freezing in Context B (Figure 1F). Mice also spent much more time in the open arms of the EZM (Figure 2), with no modification of traveling distance (Figure 2B). These results showed that JZL195 promotes contextual fear memory consolidation and recall but not cued fear memory.
JZL195 enhanced c-Fos expression induced by the contextual fear memory test
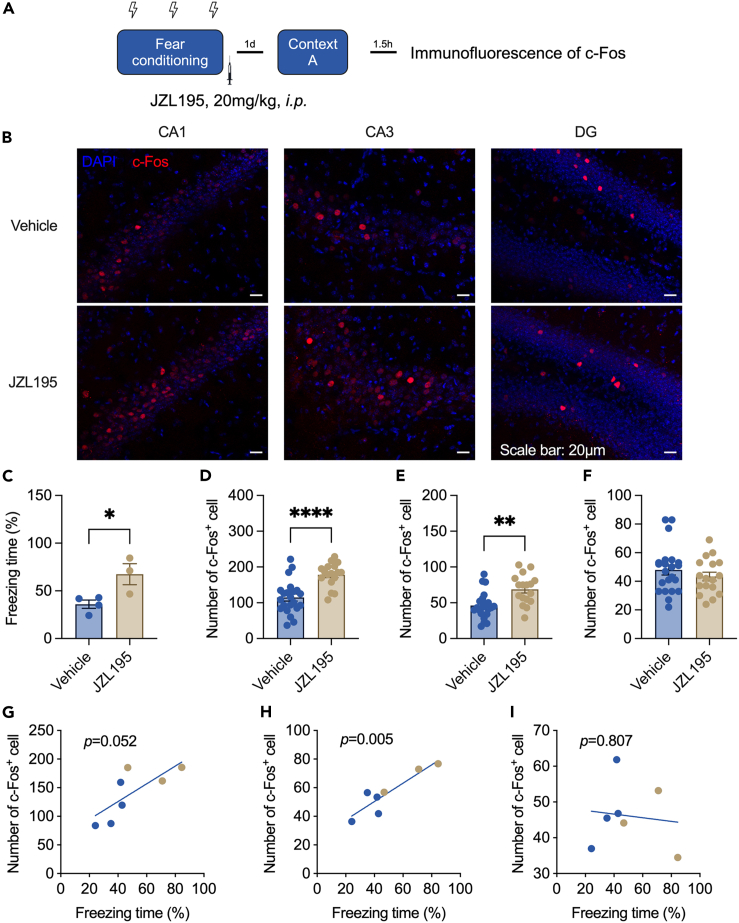
We then tested whether increased freezing could activate the hippocampus, which plays a critical role in contextual fear conditioning. The procedure of the experiment is shown in Figure 3A, and a representative image of c-Fos expression is shown in Figure 3B (Left: Cornu ammonis 1 (CA1), Middle: Cornu ammonis 3 (CA3), Right: Dentate gyrus (DG)). The number of c-Fos-positive cells was higher in the hippocampal CA1 region of JZL195-treated mice than in vehicle-treated mice (Figure 3D). The hyperactivity of neurons was also found in the CA3 (Figure 3E) but not the DG (Figure 3F) region. Correlation analysis also showed that freezing time had a positive correlation with the number of c-Fos+ cells in CA1 (Figure 3G) and CA3 (Figure 3H) but not in the DG (Figure 3I). These data showed that the improvement in contextual fear memory consolidation and recall induced by JZL195 was accompanied by an increase in neuronal activation in the hippocampus.
JZL195 enhanced hippocampal theta oscillation power in CA1 and neuronal activity
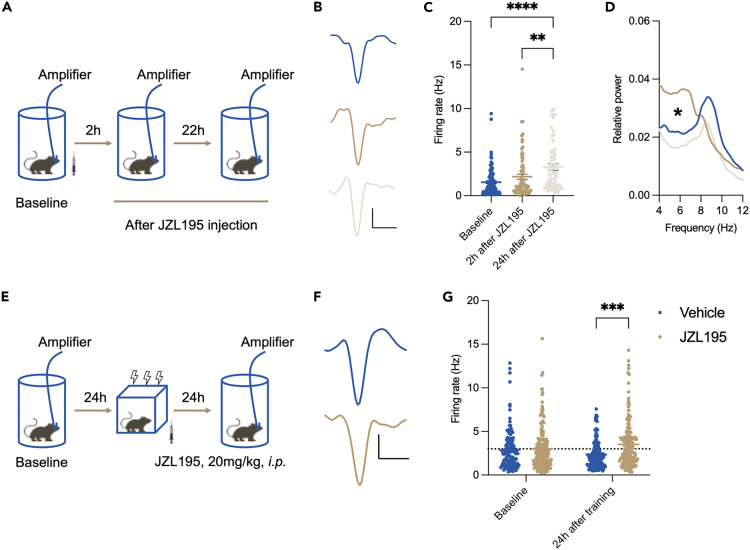
Two hours after drug injection, the local field potential (LFP) data revealed a significant enhancement in the relative power of theta oscillations in CA1 of JZL195-treated mice (Figure 4D). Meanwhile, the single-unit activity measurement did not show any difference compared to the baseline (Figure 4C). Twenty-four hours later, a statistically significant increase in single-unit activity was measured in JZL195-treated mice (Figures 4A–4C). Contextual fear conditioning was then introduced. Although mice from both groups showed similar CA1 neuronal activity, the firing rate of all isolated single units recorded in JZL195-treated mice was significantly increased (Figures 4E‒4G). These data collectively suggested that enhanced activities in the hippocampus were induced by JZL195.
Excitatory synaptic transmission was changed in CA1 pyramidal neurons
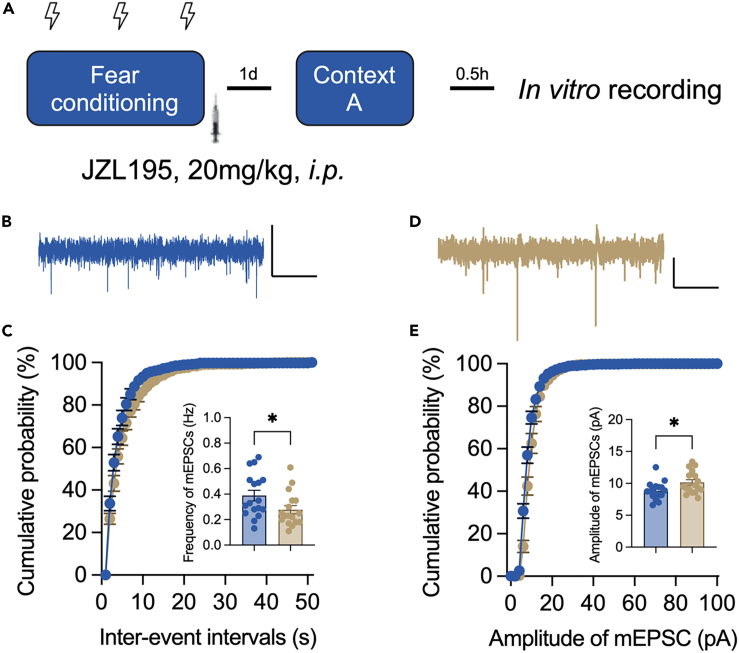
Miniature excitatory postsynaptic current (mEPSC) was thus recorded in the CA1 pyramidal neurons (Figure 5A). Compared to those from vehicle-treated mice, neurons from JZL195-treated mice exhibited a decreased frequency of mEPSCs (Figures 5B and 5C) and an elevated average amplitude (Figures 5D and 5E). These results indicated that the intrahippocampal circuit may contribute to the increased neuronal activity in the CA1 area after JZL195 treatment.
Hippocampus-specific knockout of CB1R blocked the anxiolytic effect of JZL195 but not fear memory recall
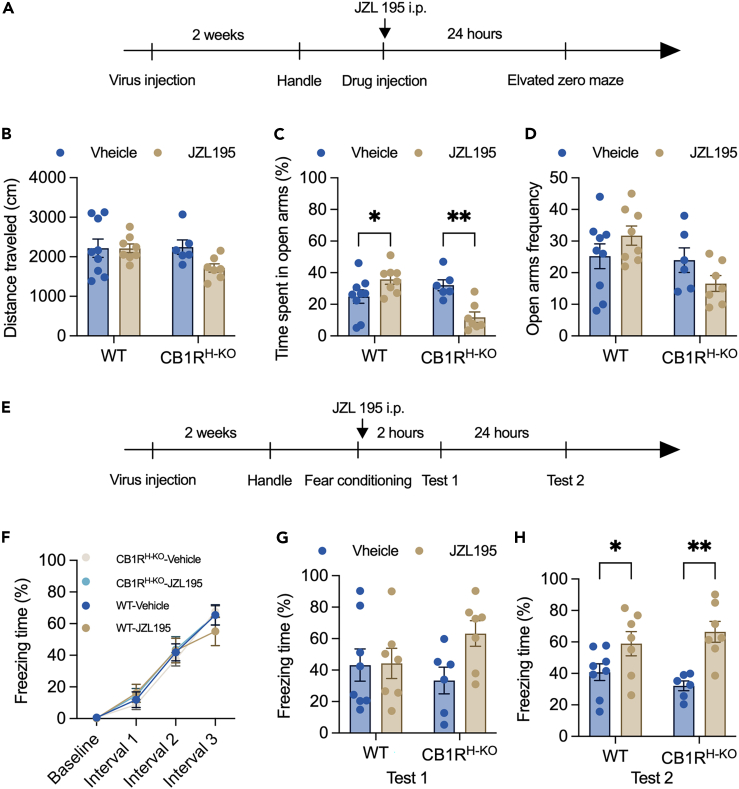
As shown in Figures S2A and S2B, intraperitoneal injection at doses of 0.03 and 3 mg/kg did not influence the performance of mice in the contextual fear conditioning task. Neither AM281 and NESS0327 nor SR144528 could counteract the effect of JZL195 (Figures S3A‒S3D). Although increased freezing time was observed in all of the JZL195-treated mice, it did not tend to be decreased by the coinjection of the antagonist of CB1R or CB2R.
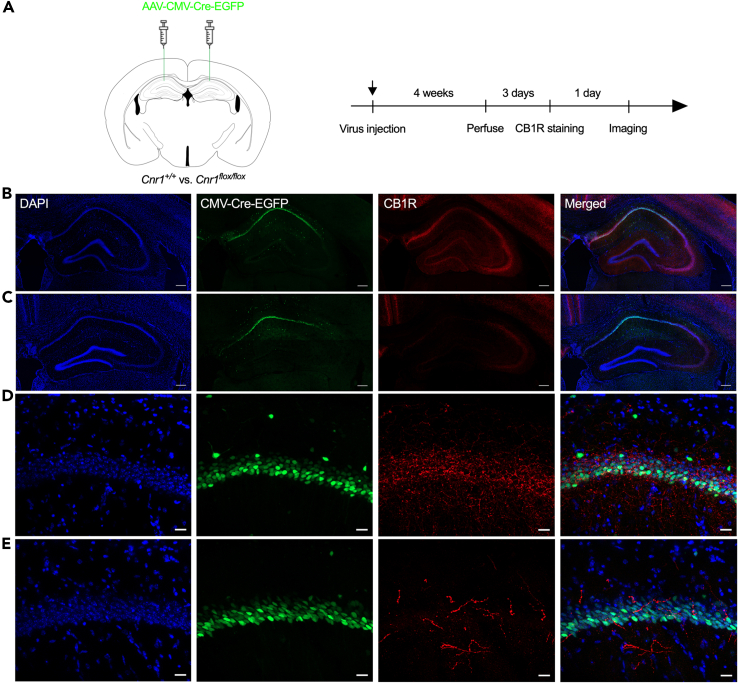
As shown in Figure 6, the CB1R protein could not be detected in the hippocampus only, while CB1R expression in the other brain areas was not affected. The timeline of this experiment is shown in Figure 7A. As shown in Figure 7, wild-type mice spent more time in the open arms of the EZM 24 h after the drug injection test (Figure 7B), while CB1RH−KO mice spent even less time in the open arms. We did not observe any difference in the distance traveled or the frequency of entering the open arms between these two mouse lines (Figures 7A and 7C). CB1RH−KO mice showed freezing time comparable to that of wild-type mice during contextual fear conditioning (Figure 7F) and the short-term memory test (Test 1, Figure 7G). On the other hand, both wild-type mice and CB1RH−KO mice had longer freezing times in the recent memory test (Test 2, Figure 7H). These results suggested that JZL195 may contribute to memory consolidation and recall in a noncannabinoid receptor-dependent manner, but hippocampal CB1R may mediate the anxiolytic effect induced by JZL195.
Discussion
Endocannabinoids have been well documented to play crucial roles in learning and memory.ref. bib14,ref. bib25 In the literature, controversial results regarding the intervention effect of endocannabinoid-degrading enzymes have been collected thus far. Knockout of MAGL, an enzyme that predominantly degrades 2-AG, was reported to promote spatial learning and memory in the water maze task and the long-term plasticity of CA3-CA1 synaptic transmission.ref. bib15 However, in another report, both JZL195 and JZL184 (a selective inhibitor of MAGL) were observed to impair the performance of mice in the water maze.ref. bib16
The influences of MAGL and FAAH deficits on fear conditioning also seem inconsistent across studies. Direct injection of AEA into the nucleus accumbens (NAc) core slightly reduced the freezing time during the contextual fear memory retrieval task,ref. bib26 and systemic administration of URB597, a selective inhibitor of FAAH, impaired fear memory recall.ref. bib27 However, when the time point of drug injection was moved backward to the start of the learning phase, the systemic administration of URB597 enhanced contextual fear memory acquisition, and this effect can be inhibited by JZL185 delivered into the prefrontal cortex or the ventral hippocampus.ref. bib28 If the clue was set as a tone, the systemic administration of JZL184 did not affect fear conditioning, but the fear memory was too strong to be extinguished.ref. bib24 In addition, the enhancement of tone-cued fear memory recall induced by JZL184 but not URB597 was also reported.ref. bib29 Despite these different reports, all the aforementioned positive outcomes achieved from drug administration or genetic manipulation are CB1R dependent. Few studies have been designed to probe the potential role of CB2R, although it was still reported as an important player in contextual fear memoryref. bib8 and a key mediator of JZL184’s effects on fear memory.ref. bib29 In the present study, we found that JZL195, a dual inhibitor of MAGL and FAAH, enhanced the performance of mice in the contextual memory retrieval task. This effect disappeared 30 days later when the mice were returned to Context A as a remote memory test. We also found that the dual inhibition of MAGL and FAAH did not affect the consolidation and recall of tone-cued fear conditioning, as mice did not show much more freezing time in Context B in the presence of training tone. This result suggested that JZL195 may take effect in a context-related brain area such as the hippocampus, which has been identified as a crucial brain area taking part in the regulation of contextual memory and fear memory, as evidenced by significantly increased c-Fos-expressing neurons after contextual fear memory recall. In addition, our data also reveal the change in hippocampal LFPs, which, especially theta (4–7 Hz) oscillation, have been demonstrated, by ample evidence, to be the key neuronal mechanism of learning and memory.ref. bib30,ref. bib31,ref. bib32,ref. bib33 We found here that the theta oscillation power was higher than the basal level 2 h after JZL195 injection. Since the critical period of memory consolidation after acquisition ranges from 2 to 6 h,ref. bib34 the increase in theta oscillation in our study may suggest that JZL195 has the potential to promote memory consolidation.
In this study, a decreased frequency of mEPSCs was observed, which indicated a presynaptic change triggered by JZL195. Conversely, the increased amplitude of mEPSCs is often regarded as a postsynaptic change resulting from upregulated glutamate receptor subunits, mainly α-amino-3-hydroxy-5-methyl-4-isoxazole-propionicacid receptor (AMPAR), and enhanced density of dendritic spines. All these alterations may contribute to high levels of neuronal activity and potential long-term potential (LTP) that underlie efficient memory retrieval. The effect of endocannabinoid (eCB) related drugs is often considered to work through CB1R or CB2R and therefore the following intracellular pathways. However, in the present study, we did not succeed in illustrating the necessity of either CB1R or CB2R for the effect of JZL195 because both the antagonists and knockout mice failed to inhibit the increased freezing. This result suggests that the other newly found receptors (e.g., TRPV1 and GPR55) may take the place of CB1R and CB2R in the contextual fear memory task.
In addition to contextual fear memory, we also found that the injection of JZL195 at a dose of 20 mg/kg produced an anxiolytic effect in the EZM test, which was mediated by CB1R, and this effect was abolished by genetic hippocampus-specific deletion of CB1R. The potential therapeutic effect of eCB has been discussed for decades; however, the dose and the injection time point do matter when applying eCB-related reagents such as JZL184, URB597, PF-3845, and JZL195. For JZL195 specifically, 3 mg/kg administration produced an antidepressant effect in an major drepressive disorder model 24 h after the last injectionref. bib35 but was reported as pro-depression when directly introducing mice into the force-swimming test 3 h after injection,ref. bib36 as was 20 mg/kg in the same report. In an anxiety-like behavioral test, when JZL195 was administered at 10 mg/kg and 2 h before the test, mice showed no such alteration in anxiety state either following acute stress or not.ref. bib37 JZL184 exhibited anxiolytic effects at various doses 3 h after injection.ref. bib38 Consistent with one of the aforementioned reports,ref. bib35 we found in this study that JZL195 produced an anxiolytic effect at a dose of 20 mg/kg when applied 24 h before the EZM test. This result also contradicts some other reports that showed the complex effect and underlying mechanism of JZL195. When CB1H−KO mice were involved, JZL195 even had a pro-anxiety effect. A possible mechanism is that the anxiolytic effect was mediated by CB1Rs expressed on glutamatergic axon terminals,ref. bib39 as we revealed a decrease in the frequency of mEPSCs. Once CB1R is removed, accumulated eCB, particularly AEA, activates TRPV1 receptors, which then increase anxiety-like behavior.ref. bib40
In summary, we established that JZL195 significantly promotes memory consolidation and retrieval in a foot shock-based fear memory task. However, due to the discrepancies among behavioral test paradigms or other unknown reasons, including pharmacological and multi-brain area interaction factors, further investigations are probably needed before the precise and detailed role that the ECS plays in different phases of memory formation can be elucidated. Moreover, we also reported a hippocampal CB1R-dependent anxiolytic effect of JZL195. Nonetheless, our study suggests the feasibility of targeting endocannabinoid-degrading enzymes for the clinical treatment of mental disorders.
Limitations of the study
In this study, we found that the dual inhibitor of FAAH and MAGL could enhance the performance of mice in the contextual fear memory task and the activity of the hippocampus. This is the first time that JZL195 was found to benefit a typical type of memory at a high dose. Although this study suggests the possibility of JZL195 contributing to the treatment of mental disorders, more specific experiments involving disease models should be conducted to identify the potential of JZL195. The subsequent mechanism underlying the anxiolytic effect of JZL195 should be investigated in detail.
STAR★Methods
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Rabbit monoclonal anti-c-Fos | Cell Signaling Technology | Cat#2250; RRID: AB_2247211 |
| Rabbit polyclonal anti-CB1R | Frontier Institute | Cat#CB1-Rb-Af380; RRID: AB_2571591 |
| Donkey anti-rabbit, AlexaFluor 647 conjugated | JacksonImmunoResearch | Cat#711-605-152; RRID: AB_2492288 |
| Bacterial and virus strains | ||
| AAV2/9-CMV-Cre-EGFP | Taitool | Cat#S0231-9 |
| Chemicals, peptides, and recombinant proteins | ||
| JZL195 | MedChemExpress | Cat#HY-15250 |
| AM281 | Sigma-Aldrich | Cat#A0980 |
| NESS0327 | Cayman Chemical | Cat#10004184 |
| SR144528 | MedChemExpress | Cat#HY-13439 |
| Experimental models: Organisms/strains | ||
| Mouse: C57BL/6J | Jax | Cat#000664 |
| Mouse: Cnr1flox/flox | This paper | N/A |
| Software and algorithms | ||
| EthovisionXT | Noldus | https://www.noldus.com.cn/ethovision-xt/ |
| VideoFreeze | Med-associates | https://med-associates.com/product/videofreeze-video-fear-conditioning-software/ |
| pClamp 10.5 | Molecular Devices | https://www.moleculardevices.com/products/axon-patch-clamp-system/acquisition-and-analysis-software/pclamp-software-suite |
| OpenEphys GUI | OpenEphys | https://open-ephys.org/gui |
| Spyking-circus | Yger et al.ref. bib41 | https://github.com/spyking-circus |
Resource availability
Lead contact
Further information and requests for resources and reagents should be directed to and will be fulfilled by the lead contact, Jing Han (jhan2012@snnu.edu.cn).
Materials availability
This study did not generate any new materials.
Data and code availability
- Data reported in this paper will be shared by the lead contact upon request.
- This paper does not report original code.
- Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request.
Experimental model and study participant details
Wild-type and transgenic animals
Male C57BL/6J mice aged 10-16 weeks were used in this study. The number of animals used in each experiment is stated in the figure legends or corresponding results sections. We do not reuse mice among tests unless otherwise stated.
Cnr1flox/flox mice were constructed by Biocytogen (Beijing, China) as previously describedref. bib43. Two loxP sites were inserted at each end of the first exon of the CB1R gene (encoded by the Cnr1 gene). We performed genotyping using PCR with the following primers. Forward: ACTGGACAGCTCATCCCTTG and reverse: AAGTCAATGGTCTTGCATGGATCT (301 bp Cnr1flox/flox, 244 bp wild type).
Hippocampus-specific deletion of CB1R
To confirm whether hippocampal CB1R mediates the effect of JZL195, we introduced a genetically modified mouse line. We constructed a mouse line with a hippocampus-specific deletion of CB1R. We injected Cre-carried AAV into the hippocampal area. Therefore, the Cre/loxP system will work specifically in the hippocampus and result in the deletion of Cnr1, the encoding gene of CB1R (the Cnr1flox/flox::CMVCre mouse line will be described as CB1RH-KO mouse in the manuscript, in which “H-KO” represents hippocampus-specific knockout).
Virus injection
Cnr1flox/flox mice were anesthetized with 1.5∼2% isoflurane and then fixed stereotaxically. AAV2/9-CMV-Cre-EGFP (volume: 300 nl/side, titer: 2.0E+12 vg/ml) was injected into the hippocampus (AP: -1.9, ML: ±1.2, DV: -1.6 mm) at a velocity of 50 nl/min. The syringe was held there for 5 minutes and then dropped off slowly. As the experimental control, wild-type mice were injected with Cre-carried virus as the same as the Cnr1flox/flox mice (so called). To obtain efficient deletion of CB1R, mice were moved to the behavioral procedure 1 month after virus injection.
Ethical statement
Mice were acquired from the laboratory animal center of the Key Laboratory of Modern Teaching Technology, Ministry of Education, Shaanxi Normal University. Mice were reared in a constant temperature and humidity environment (22±1°C, 30-40% RH) on a scale of 4-5 per cage, and the indoor day and night cycle was controlled by a fixed intensity light source (Turn on time 8:00-20:00). Each mouse was acclimated for 1-2 min, and this procedure lasted for three days before the behavioral experiments. Mice were fed ad libitum and euthanized using CO2 after finishing all tests. The experimental protocols described here were approved by the Animal Ethics Committee of Shaanxi Normal University.
Method details
Drug preparation
JZL195 (20 mg/kg) was dissolved in DMSO and diluted in sunflower oil. AM281 (3 mg/kg), NESS0327 (0.3 mg/kg), and SR144528 (3 mg/kg) were dissolved in DMSO and diluted in a solution containing 10% Tween-80 and 80% saline. All drugs were delivered through intraperitoneal injection (i.p.). The final concentration of DMSO in the working solution was kept below 10% to avoid potential biotoxicity.
Behavioral tests
Contextual fear conditioning (CFC)
Contextual fear memory was examined using CFC as previously described.ref. bib42 Mice were introduced into the fear conditioning box (MED-VFC-USB-M, Med associates, USA) and allowed to explore the box freely for 4 minutes (cumulative freezing time during this phase was set as the baseline). Then, three foot shocks (0.75 mA, 2 s) were delivered at 4, 6.25, and 8.5 minutes after the beginning of the experiment. Ninety seconds after the last foot shock, the mice were transferred back to the home cage.
Short-term memory test of contextual fear memory
Mice were returned to the conditioned box (Context A) 2 hours after conditioning and were monitored for 5 minutes.
Long-term memory test (also defined as the Recent test in the manuscript)
Mice were returned to the conditioned box (Context A) 24 hours after conditioning and were monitored for 5 minutes.
Remote memory test (also defined as Remote test in the manuscript)
Mice were returned to the conditioned box (Context A) 30 days after conditioning and were monitored for 5 minutes.
Auditory fear conditioning (AFC)
To examine cue-related fear memory, another important type of fear memory that is often thought to be expressed and regulated by the cortex and amygdala, an AFC protocol was performed. Mice were introduced into the fear conditioning box and received foot shocks as described in the Contextual fear conditioning (CFC) section. To build a connection between a specific cue and the foot shock, a 60 s-long tone was presented just before each foot shock. Cued fear memory was tested 24 hours after conditioning. Mice were introduced into a box (Context B) that had a completely different context from the conditioning box (Context A) and were monitored for 5 minutes.
Video was collected and analyzed using VideoFreeze, a commercial software provided by Med-associates.
Elevated zero maze (EZM)
EZM was used to test whether mice were anxious. The EZM used in this study was made of organic glass (at a height of 60 cm), with an inner diameter of 51.8 cm and an outer diameter of 65 cm. The closed arms of the EZM are separated by two 15 cm-high organic glasses, the outer one of which is opaque. After a 15-minute habituation, mice were put into the EZM and allowed to freely move for 10 minutes. The video was collected and analyzed using Ethovision XT. The distance that mice traveled, the time, and the frequency that mice entered into the open arms were compared between groups to evaluate anxiety-like behavior.
Immunofluorescence of c-Fos and CB1R
Immunofluorescence experiments were performed to evaluate the c-Fos expression induced by the contextual memory test and to verify the deletion of CB1R in the hippocampus. Mice were sacrificed and perfused with saline. The brain was taken and immediately immersed in precooled 4% PFA and kept at 4°C overnight. The PFA was then replaced with 30% sucrose solution in 1x PBS and kept at 4°C until the brain sunk into the bottom of the vial. Forty-micron coronal sections were collected using a cryostat microtome. After washing with PBS, the slices were blocked with blocking buffer containing 10% normal donkey serum, 0.3% Triton-X 100, and 89.7% PBS for 2 hours at room temperature. Then, we incubated slices with primary antibodies and secondary antibodies in a 24-well plate. After the slices were pasted, slides were mounted with antifade reagents. Hippocampal images were collected using a Zeiss M2 microscope. c-Fos-positive cells were automatically counted in ImageJ software.
Electrophysiology
In vitro experiment – Slice preparation
Mice were anesthetized with urethane and then decapitated. The brain was quickly removed from the skull and immersed in precooled sucrose-based cutting solution (in mM, 225 sucrose, 2.5 KCl, 1.25 NaH2PO4, 26 NaHCO3, 11 D-Glucose, 5 L-Ascorbic Acid, 3 Sodium Pyruvate, 7 MgSO4·7H2O, 0.5 CaCl2). After being fixed on a metal pallet, the brain was cut into 300-μm slices. Slices were then collected and incubated in artificial cerebrospinal fluid (ACSF) containing (in mM): 122 NaCl, 2.5 KCl, 1.25 NaH2PO4, 26 NaHCO3, 11 D-Glucose, 2 MgSO4·7H2O, 2 CaCl2 equilibrated with 95% O2-5% CO2 at 28°C for at least 1 hour before recording.
Whole-cell recording of miniature excitatory postsynaptic current (mEPSC)
mEPSCs were recorded in CA1 pyramidal neurons to measure excitatory transmission from CA3 to CA1. When performing patch-clamp, ACSF contained 100 μM PTX, and 1 μM TTX was perfused constantly to keep cells healthy. The k-based intracellular solution contained 120 K gluconate, 20 KCl, 10 HEPES, 10 phosphocreatine Na salt, 2 ATP Na salt, 0.4 GTP Na salt and 2 MgCl2, pH 7.35, were used to make a whole-cell recording. Pyramidal neurons in dorsal CA1 were visually identified and clamped. After a stable whole-cell recording was made, the membrane potential was clamped at -70 mV. We then acquired mEPSCs in gap-free mode. Five-minute-long continuous data were saved and analyzed using pClamp.
In vivo experiment – Electrode implantation
Mice were anesthetized with 2% isoflurane and held on a stereotype (RWD, China). Sixteen-channel microwire array electrodes (KD-MWA, KedouBC, China), a 4×4 array consisting of 25 μm NiTi wire spaced 200 μm, were slowly inserted into the mouse brain. Four small nails were first inserted into the skull, with a ground wire presoldered onto one of them. The electrode was left at the dorsal hippocampus (AP: -1.9, ML: -1.2, DV: -1.5 mm), and then dental cement was used to fix it onto the skull.
Data acquisition
Mice were introduced to the recording arena at least one week after surgery. During the day, the electrode on the mouse skull was connected to the OpenEphys acquisition board through an Intel head stage. OpenEphys GUI was used to visualize and save electrical signals. Mice were allowed to freely move inside a home cage-like arena for at least 20 mins. Only data acquired during the last 10 min were saved and then analyzed by Python-based software and a customized MATLAB script (described below).
Single unit isolation
Spikes were detected and divided into single units using the Spyking-circus.ref. bib41 Continuous binary raw data (sampling rate: 30 kHz) were imported and filtered using a bandpass butter filter at a cutoff value of 300 Hz. Movement artifacts were removed by subtracting medians over all channels. The spike detection threshold was set at a 6-fold median absolute deviation (MAD), while spikes beyond 10-fold MAD were abandoned as artifacts. Templates used to detect spikes were automatically generated, and the width of the templates was manually designated as 2 ms. Clustering was also automatically processed using a density-based cluster method, and clusters with a Bhatta distance less than 0.5 were merged into a single cluster. Neurons meeting the following criteria were excluded from the following analysis: (1) spikes with refracting period violations smaller than 1 ms, accounting for more than 1.5% of total spikes, and (2) total frequency lower than 0.2 Hz (probably noise) or larger than 20 Hz (probably fast-spiking interneurons).
Local field potential (LFP) power spectrum
Raw data were loaded into MATLAB and bandpass filtered at 0.5-150 Hz. The power spectrum of LFP was analyzed using the MATLAB function “Signal Analyzer”. The relative power of the specific θ oscillation (4-12 Hz) was calculated per channel.
Quantification and statistical analysis
Data are presented as the mean±SEM in all figures included in this manuscript. To compare mean values between the two groups, in terms of the normally distributed data and equal standard deviation, Student’s t test was performed in GraphPad software. Otherwise, the Mann‒Whitney test was performed instead. In terms of the normally distributed data and equal standard deviation, one-way or two-way ANOVA was performed in GraphPad software followed by Sidak’s or Tukey’s post hoc test to compare mean values among three or more than three groups. Otherwise, the Kruskal‒Wallis test followed by Dunn’s post hoc test was performed instead.
References
- M.M. Shi, K.M. Fan, Y.N. Qiao, J.H. Xu, L.J. Qiu, X. Li, Y. Liu, Z.Q. Qian, C.L. Wei, J. Han. Hippocampal μ-opioid receptors on GABAergic neurons mediate stress-induced impairment of memory retrieval. Mol. Psychiatry, 2020. [PubMed]
- J. Zhang, L. Zhang, Y. Chang, Q. Gu, J. Zhang, Z. Zhu, Z. Qian, C. Wei, Z. Liu, W. Ren, J. Han. The Endocannabinoid System Contributes to Memory Deficits Induced by Rapid-eye-movement Sleep Deprivation in Adolescent Mice. Neuroscience, 2020. [PubMed]
- R. Ossenkoppele, Y.A.L. Pijnenburg, D.C. Perry, B.I. Cohn-Sheehy, N.M.E. Scheltens, J.W. Vogel, J.H. Kramer, A.E. Van Der Vlies, R. La Joie, H.J. Rosen. The behavioural/dysexecutive variant of Alzheimer’s disease: Clinical, neuroimaging and pathological features. Brain, 2015. [PubMed]
- S. Bajaj, S. Jain, P. Vyas, S. Bawa, D. Vohora. The role of endocannabinoid pathway in the neuropathology of Alzheimer’s disease: Can the inhibitors of MAGL and FAAH prove to be potential therapeutic targets against the cognitive impairment associated with Alzheimer’s disease?. Brain Res. Bull., 2021. [PubMed]
- V. Chevaleyre, P.E. Castillo. Endocannabinoid-mediated metaplasticity in the hippocampus. Neuron, 2004. [PubMed]
- V. Chevaleyre, K.A. Takahashi, P.E. Castillo. Endocannabinoid-Mediated Synaptic Plasticity in the Cns. Annu. Rev. Neurosci., 2006. [PubMed]
- S.M. Augustin, D.M. Lovinger. Functional Relevance of Endocannabinoid-Dependent Synaptic Plasticity in the Central Nervous System. ACS Chem. Neurosci., 2018. [PubMed]
- Y. Li, J. Kim. Distinct roles of neuronal and microglial cb2 cannabinoid receptors in the mouse hippocampus. Neuroscience, 2017. [PubMed]
- J. Han, P. Kesner, M. Metna-Laurent, T. Duan, L. Xu, F. Georges, M. Koehl, D.N. Abrous, J. Mendizabal-Zubiaga, P. Grandes. Acute cannabinoids impair working memory through astroglial CB 1 receptor modulation of hippocampal LTD. Cell, 2012. [PubMed]
- N. Stella. THC and CBD : Similarities and differences between siblings. Neuron, 2023. [PubMed]
- C. Song, C.W. Stevenson, F.S. Guimaraes, J.L.C. Lee. Bidirectional effects of cannabidiol on contextual fear memory extinction. Front. Pharmacol., 2016. [PubMed]
- A. Murkar, P. Kent, C. Cayer, J. James, T. Durst, Z. Merali. Cannabidiol and the remainder of the plant extract modulate the effects of Δ9-tetrahydrocannabinol on fear memory reconsolidation. Front. Behav. Neurosci., 2019. [PubMed]
- I. Katz, D. Katz, Y. Shoenfeld, B.S. Porat-Katz. Clinical evidence for utilizing cannabinoids in the elderly. Isr. Med. Assoc. J., 2017. [PubMed]
- H.C. Lu, K. MacKie. An introduction to the endogenous cannabinoid system. Biol. Psychiatry, 2016. [PubMed]
- B. Pan, W. Wang, P. Zhong, J.L. Blankman, B.F. Cravatt, Q.-S. Liu. Alterations of Endocannabinoid Signaling, Synaptic Plasticity, Learning, and Memory in Monoacylglycerol Lipase Knock-out Mice. J. Neurosci., 2011. [PubMed]
- L.E. Wise, K.A. Long, R.A. Abdullah, J.Z. Long, B.F. Cravatt, A.H. Lichtman. Dual fatty acid amide hydrolase and monoacylglycerol lipase blockade produces THC-like morris water maze deficits in mice. ACS Chem. Neurosci., 2012. [PubMed]
- R. Chen, J. Zhang, Y. Wu, D. Wang, G. Feng, Y.P. Tang, Z. Teng, C. Chen. Monoacylglycerol Lipase Is a Therapeutic Target for Alzheimer’s Disease. Cell Rep., 2012. [PubMed]
- J. Zhang, C. Chen. Alleviation of Neuropathology by Inhibition of Monoacylglycerol Lipase in APP Transgenic Mice Lacking CB2 Receptors. Mol. Neurobiol., 2018. [PubMed]
- J. Hashem, M. Hu, J. Zhang, F. Gao, C. Chen. Inhibition of 2-Arachidonoylglycerol Metabolism Alleviates Neuropathology and Improves Cognitive Function in a Tau Mouse Model of Alzheimer’s Disease. Mol. Neurobiol., 2021. [PubMed]
- S. Bajaj, S. Zameer, S. Jain, V. Yadav, D. Vohora. Effect of the MAGL/FAAH Dual Inhibitor JZL-195 on Streptozotocin-Induced Alzheimer’s Disease-like Sporadic Dementia in Mice with an Emphasis on Aβ, HSP-70, Neuroinflammation, and Oxidative Stress. ACS Chem. Neurosci., 2022. [PubMed]
- M.S. Sufian, M.R. Amin, D.W. Ali. Early suppression of the endocannabinoid degrading enzymes FAAH and MAGL alters locomotor development in zebrafish. J. Exp. Biol., 2021
- R. Greco, C. Demartini, M. Francavilla, A.M. Zanaboni, C. Tassorelli. Dual inhibition of faah and magl counteracts migraine-like pain and behavior in an animal model of migraine. Cells, 2021. [PubMed]
- R. Abohalaka, Y. Karaman, T. Recber, S.C. Onder, E. Nemutlu, T.E. Bozkurt. Endocannabinoid metabolism inhibition ameliorates ovalbumin-induced allergic airway inflammation and hyperreactivity in Guinea pigs. Life Sci., 2022
- N.D. Hartley, O. Gunduz-Cinar, L. Halladay, O. Bukalo, A. Holmes, S. Patel. 2-Arachidonoylglycerol Signaling Impairs Short-Term Fear Extinction. Transl. Psychiatry, 2016. [PubMed]
- I. Mizuno, S. Matsuda. The role of endocannabinoids in consolidation , retrieval , reconsolidation , and extinction of fear memory. Pharmacol. Rep., 2021. [PubMed]
- R. Pedroza-Llinás, M. Méndez-Díaz, A.E. Ruiz-contreras, Ó. Prospéro-García. CB1 receptor activation in the nucleus accumbens core impairs contextual fear learning. Behav. Brain Res., 2013. [PubMed]
- H. Ahmad, K. Rauf, W. Zada, M. McCarthy, G. Abbas, F. Anwar, A.J. Shah. Kaempferol Facilitated Extinction Learning in Contextual Fear Conditioned Rats via Inhibition of. Molecules, 2020. [PubMed]
- Z. Balogh, L. Szente, L. Biro, Z.K. Varga, J. Haller, M. Aliczki. Endocannabinoid interactions in the regulation of acquisition of contextual conditioned fear. Prog. Neuro-Psychopharmacol. Biol. Psychiatry, 2019
- A. Llorente-berzal, A.L.B. Terzian, V. di Marzo, V. Micale, M.P. Viveros, C.T. Wotjak. 2-AG promotes the expression of conditioned fear via cannabinoid receptor type 1 on GABAergic neurons. Psychopharmacology (Berl), 2015. [PubMed]
- I.C. Hutchison, S. Rathore. The role of REM sleep theta activity in emotional memory. Front. Psychol., 2015. [PubMed]
- A. Goyal, J. Miller, S.E. Qasim, A.J. Watrous, H. Zhang, J.M. Stein, C.S. Inman, R.E. Gross, J.T. Willie, B. Lega. Functionally distinct high and low theta oscillations in the human hippocampus. Nat. Commun., 2020. [PubMed]
- G. Etter, S. van der Veldt, J. Choi, S. Williams. Optogenetic frequency scrambling of hippocampal theta oscillations dissociates working memory retrieval from hippocampal spatiotemporal codes. bioRxiv, 2022. [DOI]
- G. Etter, S. van der Veldt, F. Manseau, I. Zarrinkoub, E. Trillaud-Doppia, S. Williams. Optogenetic gamma stimulation rescues memory impairments in an Alzheimer’s disease mouse model. Nat. Commun., 2019. [PubMed]
- T.-M. Prince, M. Wimmer, J. Choi, R. Havekes, S. Aton, T. Abel. Sleep deprivation during a specific 3-hour time window post-training impairs hippocampal synaptic plasticity and memory. Neurobiol. Learn. Mem., 2014. [PubMed]
- B. Dong, B.M. Shilpa, R. Shah, A. Goyal, S. Xie, M.J. Bakalian, R.F. Suckow, T.B. Cooper, J.J. Mann, V. Arango, K.Y. Vinod. Dual pharmacological inhibitor of endocannabinoid degrading enzymes reduces depressive-like behavior in female rats. J. Psychiatr. Res., 2020. [PubMed]
- F.J. Pavón, I.Y. Polis, D.G. Stouffer, B.F. Cravatt, M. Roberto, R. Martin-Fardon, F. Rodríguez de Fonseca, L.H. Parsons, A. Serrano. Selective inhibition of monoacylglycerol lipase is associated with passive coping behavior and attenuation of stress-induced dopamine release in the medial prefrontal cortex. Neurobiol. Stress, 2021
- G. Bedse, R.J. Bluett, T.A. Patrick, N.K. Romness, A.D. Gaulden, P.J. Kingsley, N. Plath, L.J. Marnett, S. Patel. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: Comparative profiling of FAAH, MAGL and dual inhibitors. Transl. Psychiatry, 2018. [PubMed]
- G. Bedse, N.D. Hartley, E. Neale, A.D. Gaulden, T.A. Patrick, P.J. Kingsley, M.J. Uddin, N. Plath, L.J. Marnett, S. Patel. Functional Redundancy Between Canonical Endocannabinoid Signaling Systems in the Modulation of Anxiety. Biol. Psychiatry, 2017. [PubMed]
- A.A. Rey, M. Purrio, M.P. Viveros, B. Lutz. Biphasic effects of cannabinoids in anxiety responses: CB1 and GABA B receptors in the balance of gabaergic and glutamatergic neurotransmission. Neuropsychopharmacology, 2012. [PubMed]
- T. Rubino, N. Realini, C. Castiglioni, C. Guidali, D. Viganó, E. Marras, S. Petrosino, G. Perletti, M. Maccarrone, V. Di Marzo, D. Parolaro. Role in anxiety behavior of the endocannabinoid system in the prefrontal cortex. Cereb. Cortex, 2008. [PubMed]
- P. Yger, G.L. Spampinato, E. Esposito, B. Lefebvre, S. Deny, C. Gardella, M. Stimberg, F. Jetter, G. Zeck, S. Picaud. A spike sorting toolbox for up to thousands of electrodes validated with ground truth recordings in vitro and in vivo. Elife, 2018. [PubMed]
- L. Wang, M. Xing, W. Qu, C. Wang, Z. Liu, J. Han, W. Ren, Y. Qiao. Impaired vessel relaxation response and increased infarct size in smooth muscle cannabinoid receptor 1 knockout mice. Microvascular Research, 2021. [PubMed]
- Y. Chang, J. Zhang, J. Zhang, W. Zhu, Q. Zheng, Z. Qian, C. Wei, Y. Liu, Z. Liu, W. Ren, J. Han. N6-methyladenosine RNA modification of glutamatergic neurons is associated with contextual fear discrimination. Physiol. Behav., 2022
