Cytotoxic Activity of Opopanax hispidus (Friv.) Griseb.: Characterization of a New Chalcone‐type Compound
Abstract
The present study aimed to investigate cytotoxic compounds of Opopanax hispidus (Friv.) Griseb. through in vitro, in silico, and phytochemical analyses. The cytotoxic activity of O. hispidus was evaluated against four different cancer cell lines – lung (A549), breast (MCF‐7), liver (HepG2), and cervix (HeLa) – as well as one healthy cell line (Beas‐2B), using the 3‐(4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyltetrazolium bromide colorimetric assay. n‐Hexane and dichloromethane sub‐extracts with the IC50 values of 9.77 ± 0.57 and 7.10 ± 0.78 mg/mL on A549, respectively, were fractionated through chromatographic techniques, and the structures of the isolated compounds were elucidated using various spectroscopic methods. A novel compound, 2′,6’‐dihydroxy‐4’‐methoxy‐3”‐(ɤ,ɤ‐dimethylallyl)‐4”‐hydroxy β‐hydroxydihydrochalcone (3), along with three known coumarins – umbelliferone 6‐carboxylic acid (1), umbelliferone (2), and nodakenetin (4) – were isolated from the active fractions. Compound 3, which possesses a chalcone structure, exhibited notable cytotoxicity against lung cancer cells with an IC50 value of 13.72 ± 0.80 µM.
Article type: Research Article
Keywords: Apiaceae, chalcone, cytotoxic activity, lung cancer
Affiliations: Department of Pharmacognosy, Faculty of Pharmacy Sivas Cumhuriyet University Sivas Türkiye; Department of Pharmacognosy, Faculty of Pharmacy Gazi University Ankara Türkiye; Department of Pharmacognosy, Faculty of Pharmacy Erciyes University Kayseri Türkiye; Department of Pharmaceutical Toxicology, Faculty of Pharmacy Gazi University Ankara Türkiye; Department of Pharmaceutical Botany, Faculty of Pharmacy Selcuk University Konya Türkiye; Department of Basic Sciences, Faculty of Pharmacy Erciyes University, Kayseri Türkiye; Department of Chemistry, Faculty of Basic Sciences Gebze Technical University Gebze, Kocaeli Türkiye; Troyasil HPLC Column Technologies Doruk Analitik, Mehmet Akif Mah. Yumurcak Sok. No:43 Ümraniye İstanbul Türkiye
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202501077 | PubMed: 40694628 | PMC: PMC12629158
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (627 KB)
Introduction
Cancer, characterized by uncontrolled cell growth and abnormal signaling processes, is one of the most life‐threatening diseases. Its global incidence is on the rise, emphasizing the urgent need for the discovery of novel compounds and therapeutic strategies to combat cancer. One of the primary challenges in cancer treatment is the toxicity of anticancer drugs to healthy cells, which results in numerous adverse effects. The structural diversity and bioactivity potential of natural compounds present promising opportunities for developing new anticancer agents targeting key cancer types [ref. 1]. Cytotoxicity‐guided studies serve as an essential initial step in identifying compounds that can target tumors through various mechanisms, such as alkylating agents, mitotic inhibitors, and topoisomerase inhibitors.
The Apiaceae family, which includes a wide array of economically and medicinally valuable species, comprises approximately 450 genera and over 3500 species worldwide [ref. 2]. Numerous species within this family, particularly those from the genera Ferula L., Smyrnium L., Prangos Lindl., Ferulago W. Koch, Pimpinella L., Heracleum L., Angelica L., Eryngium L., and Peucedanum L., have been studied for their anticancer properties. The active compounds responsible for these effects are predominantly flavonoids, terpenoids, and coumarins [ref. 3, ref. 4, ref. 5, ref. 6, ref. 7]. Based on the literature, it was hypothesized that plants from the Apiaceae family could serve as potential sources for anticancer research. Therefore, this study focused on Opopanax hispidus (Friv.) Griseb., from the Apiaceae family, a species for which detailed phytochemical and cytotoxicity studies have yet to be conducted.
The term Opopanax is a source of confusion, as it refers to distinct substances that share the same name. Similar to olibanum and myrrh, Opopanax designates a resin obtained from specific Commiphora shrub species, which are unrelated to the genus Opopanax W.D.J. Koch and are distributed across regions from the Middle East to Africa [ref. 8]. In contrast, the genus Opopanax W.D.J. Koch is primarily found in temperate climates. The precise origin of the so‐called “true Opopanax” remains uncertain due to the ambiguous descriptions provided by ancient authors [ref. 9].
The genus Opopanax is represented by four species in Türkiye, namely Opopanax hispidus (Friv.) Griseb., Opopanax chironium (L.) W. Koch, Opopanax persicus Boiss. and Opopanax siifolius (Boiss. & Heldr.) Menemen [ref. 10]. These species have traditionally been used for their various medicinal properties, including cancer. For instance, ointment with a mixture of essential oil, gum, and resin obtained from O. chironium has been used to treat different cancers [ref. 11]. Moreover, heraclenin and imperatorin from O. chironium were determined to induce apoptosis in leukemia cells and show selective cytotoxicity toward cancer cells [ref. 12].
O. hispidus, known locally as “kekire, kaymecik, gaymecik” [ref. 13, ref. 14, ref. 15], has a historical use as an antidote in Dynameron [ref. 16]. The stems, leaves, and flowers of O. hispidus were used for antiseptic purposes in Iran. In Türkiye, fresh stems are used to treat female infertility; leaves are used against hemorrhoids [ref. 15, ref. 17, ref. 18]. Moreover, fresh stems and basal leaves are consumed as food in some parts of Türkiye [ref. 17].
Several bioactive compounds from O. hispidus have been described in the literature, demonstrating anti‐inflammatory, antibacterial, antioxidant, and enzyme‐inhibitory properties, particularly in relation to infertility [ref. 8, ref. 10, ref. 19, ref. 20, ref. 21]. The essential oil of O. hispidus contains labdane‐type diterpenes and sesquiterpenes, with torulosol, geranyl geraniol acetate, and germacrene D as major components [ref. 22]. Additionally, the flowers and leaves of O. hispidus are rich in phenolics and flavonoids [ref. 8]. However, isolation studies on this plant remain limited. Notably, seven coumarins—3’‐isobutyryl‐3’‐hydroxymarmesin, oreoselon, peucedanin, officinalin, smirniorin, 4’‐acetyl‐3’‐isobutyryl‐3’‐hydroxymarmesin, and 3’‐hydroxypranthimgin—were isolated from the chloroform extract of the aerial parts of O. hispidus [ref. 23].
In the present study, an in vitro activity‐guided fractionation assay was conducted to isolate cytotoxic secondary metabolites from the n‐hexane and dichloromethane sub‐extracts of the whole plant of O. hispidus. The cytotoxicity of these compounds was evaluated against lung (A549), breast (MCF7), liver (HepG2), and cervix (HeLa) cancer cells. Additionally, in silico predictions were performed to assess the biological activities of the isolated compounds and to support cytotoxicity results.
Results and Discussion
Structure Elucidation and Identification
The crude methanol extract of O. hispidus was suspended with distilled water and partitioned with organic solvents of increasing polarity: n‐hexane, dichloromethane, ethyl acetate, and n‐butanol (see section 2.3). The OHNH and OHDCM subextracts were fractionated by repeated column chromatography (Sephadex LH‐20, silica gel, MPLC, and preparative TLC) to obtain a new β‐hydroxydihydrochalcone (3), and three known coumarins: umbelliferone 6‐carboxylic acid (1), umbelliferone (2), and nodakenetin (4) (Figure 1).
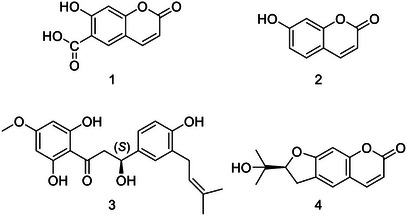
Although coumarin derivatives, umbelliferone, umbelliferone 6‐carboxylic acid, and nodakenetin, were isolated from different genera belonging to the Apiaceae family in the literature, their isolation from the Opopanax genus was reported for the first time in the present study.
Compound 3 was isolated as a yellowish‐white solid. The molecular formula C21H24O6 was established based on the electrospray ionization (negative ion mode) molecular ion obtained at m/z 371.1457 [M─H]‐ (calcd. for C21H23O6, 371.1489). The 1H nuclear magnetic resonance (NMR) showed two doublet of doublets at δ H 3.12 (dd, J = 17.1, 12.6 Hz, H‐2a) and δ H 2.73 (dd, J = 17.1, 3.1 Hz, H‐2b) indicates the two diasterotopic protons of methylene group and 13C NMR δ C 198.3 (C═O), 44.0 (C‐2), and 80.8 (C‐3) data suggested the compound to have a β‐hydroxydihydrochalcone skeleton. The 1H NMR and 1H‐1H correlated spectroscopy (COSY) spectra of 1 revealed two aromatic rings. The first one showed 2,4,6‐trisubstitution ring systems and confirmed from the signals δ H 6.05 (d, J = 2.3 Hz, H‐5ʹ) and δ H 6.03 (d, J = 2.3 Hz, H‐3ʹ). The protons of the second aromatic ring revealed the ABX system at δ H 7.17 (d, J = 2.2 Hz, H‐2ʹʹ), 7.13 (dd, J = 8.2, 2.2 Hz, H‐6ʹʹ), and 6.79 (d, J = 8.2 Hz, H‐5ʹʹ). Besides, the presence of those at δ H 1.72 and 1.74 and at δ C 133.2 and 123.7 indicated the ɤ,ɤ‐dimethylallyl group. The 13C NMR spectrum of the compound exhibited twenty‐one carbon resonances including a carbonyl carbon (δ C 198.3), two methylene carbons (δ C 44.0 and 29.3), a carbon bearing a hydroxyl group (δ C 80.8), carbons belong to the ɤ,ɤ‐dimethylallyl group (δ C 133.2, 123.7, 25.9, 17.9), one methoxyl group (δ C 56.2) and aromatic carbons δ C 169.5, 165.2, 164.7, 156.7, 130.8, 129.6, 129.0, 126.2, 115.7, 104.1, 95.7, and 94.9 (Table 1). The connectivity of the pronated carbons between C‐1ʹʹʹ and C‐2 ʹʹʹ and C‐2 and C‐3 was detected by the COSY spectrum of 3 (Figure 2).
TABLE 1: 1H and 13C nuclear magnetic resonance (NMR) data of compound 3.
| Position | C/H | δC ppm | δH ppm, J (Hz) |
|---|---|---|---|
| 1 | C | 198.3 | — |
| 2 | CH2 | 44.0 | 3.12 (H‐2a, dd, J = 17.1; 12.6 Hz) |
| 2.73 (H‐2b, dd, J = 17.1; 3.1 Hz) | |||
| 3 | CH | 80.8 | 5.30‐5.37cbdv70254-tbl1-note-0001 (1H, m) |
| 1ʹ | C | 104.1 | — |
| 2ʹ | C | 165.2 | — |
| 3ʹ | CH | 95.7 | 6.03 (1H, d, J = 2.4 Hz) |
| 4ʹ | C | 169.5 | — |
| 5ʹ | CH | 94.9 | 6.05 (1H, d, J = 2.3 Hz) |
| 6ʹ | C | 164.7 | — |
| 1ʹʹ | C | 130.8 | — |
| 2ʹʹ | CH | 129.0 | 7.17 (1H, d, J = 2.2 Hz) |
| 3ʹʹ | C | 129.6 | — |
| 4ʹʹ | C | 156.7 | — |
| 5ʹʹ | CH | 115.7 | 6.79 (1H, d, J = 8.2 Hz) |
| 6ʹʹ | CH | 126.2 | 7.13 (1H, dd, J = 8.2 and 2.3‐2.2 Hz) |
| 1ʹʹʹ | CH2 | 29.3 | 3.30z |
| 2ʹʹʹ | CH | 123.7 | 5.30‐5.37cbdv70254-tbl1-note-0001(1H, m) |
| 3ʹʹʹ | C | 133.2 | — |
| 4ʹʹʹ | CH3 | 25.9 | 1.72 (3H, s) |
| 5ʹʹʹ | CH3 | 17.9 | 1.74 (3H, s) |
| 4′‐OCH3 | CH3 | 56.2 | 3.81 (3H, s) |
* Overlapped protons, z Overlapped with solvent signal.
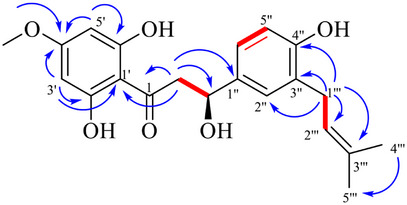
The linkage position of the methoxy and dimethylallyl groups was established by observation of HMBC correlations. The HMBC spectrum showed that the methylene signal at δ H 3.30 (H‐1′′′) was correlated with C‐3′′ (δ C 129.6), C‐4′′ (δ C 156.7), C‐2′′′ (δ C 123.7), and C‐3′′′ (δ C 133.2), indicating that the dimethylallyl group was attached to C‐3′′. Moreover, the methoxy signal at δ H 3.81 (4′‐OCH3) was correlated with C‐4′ (δ C 169.5), suggesting that the methoxy group was attached to C‐4′. From the above discussion, compound 3 was found to be a new β‐hydroxydihydrochalcone and given a trivial name as opopanaxchalcone.
Based on the conformational analysis, the electronic circular dichroism (ECD) spectra of (S)‐compound 3 and (R)‐compound 3 were calculated in chloroform at the B3LYP/6‐311G(d,p) level using time‐dependent density functional theory (TDDFT). As shown in Figure 3, the experimental spectrum shows a strong negative Cotton effect around 220 nm and a positive one near 250 nm. These features are well reproduced by the calculated spectrum of the (S)‐enantiomer. In contrast, the (R)‐enantiomer shows opposite signs at these wavelengths. A weak shoulder around 285 nm is also better matched by the (S)‐compound 3. Overall, the comparison confirms that compound 3 has the (S)‐absolute configuration.

β‐Hydroxydihydrochalcones are rare natural compounds. Ziganin, a glucosyldihydrochalcone from Pimpinella rhodantha Boiss. (Apiaceae); 3‐(S)‐hydroxy‐3‐phenyl‐1‐(2′,4′,6′‐trihydroxyphenyl)propan‐1‐one from Myristica beddomei subsp. spherocarpa W.J. de Wilde (Myristicaceae); balanochalcone from Balanophora laxiflora Hemsl. (Balanophoraceae); (S)‐elatadihydrochalcone from Tephrosia elata Deflers (Fabaceae); (S)‐elatadihydrochalcone‐2′‐methyl ether from Tephrosia uniflora Persia. (Fabaceae) were reported as new β‐hydroxydihydrochalcone derivatives [ref. 24, ref. 25, ref. 26, ref. 27, ref. 28]. In this study, opopanaxchalcone isolated from the dichloromethane sub‐extract of O. hispidus was reported as a novel β‐hydroxydihydrochalcone derivative.
Cytotoxic Activity
The cytotoxic activity of crude methanol extract was investigated on MCF‐7, HeLa, HepG2, A549 cancer cells, and Beas‐2B cells using the 3‐(4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyltetrazolium bromide (MTT) assay. The extract showed the highest activities on MCF‐7 and A549 with IC50 values of 21.44 µg/mL and 40.78 µg/mL, respectively. Thus, the sub‐extracts obtained from the methanol extract were studied on lung and breast cancer cells (Figures S1 and S2). Among sub‐extracts, OHNH and OHDCM showed significant effects in a dose‐dependent manner with the IC50 values of 9.77 ± 0.57 and 7.10 ± 0.78 on A549, respectively. These two sub‐extracts had IC50 values of less than 20 µg/mL in both breast and lung cancer cells (Table 2). Ethyl acetate, n‐butanol, and water sub‐extracts showed no activity in the tested concentrations.
TABLE 2: Cytotoxic activity results of O. hispidus methanol extract and sub‐extracts against the tested cell lines.
| Extract | IC50 ± SEM (µg/mL) | ||||
|---|---|---|---|---|---|
| MCF‐7 | A549 | HeLa | HepG2 | Beas‐2B | |
| OH MeOH | 21.44 ± 2.01 | 40.78 ± 2.54 | 49.53 ± 2.77 | 77.68 ± 5.84 | 36.82 ± 2.01 |
| OHNH | 10.90 ± 0.41 | 9.77 ± 0.57 | — | — | 6.42 ± 0.32 |
| OHDCM | 8.47 ± 0.95 | 7.10 ± 0.78 | — | — | 2.16 ± 0.19 |
| OHEtOAc | >100 | >100 | — | — | >100 |
| OHBuOH | >100 | >100 | — | — | >100 |
| OHH2O | >100 | >100 | — | >100 | |
| Doxcbdv70254-tbl2-note-0001 | 1.89 ± 0.27 | 0.82 ± 0.13 | 0.55 ± 0.06 | 3.02 ± 0.37 | 0.34 ± 0.02 |
* The results of doxorubicin were given as IC50 ± SEM (µM). Dox: doxorubicin; SEM: standard error of the mean; OHNH: n‐hexane; OHDCM: dichloromethane; OHEtOAc: ethyl acetate; OHBuOH: n‐butanol; OHH2O: water.
A dramatic increase in activity was noted in the OHNH and OHDCM sub‐extracts compared to OH MeOH. In breast cancer cells, the IC50 for OH MeOH was 21.44 µg/mL, and it was calculated as 10.90 and 8.47 µg/mL in OHNH and OHDCM, respectively. On the other hand, in the lung cancer cells, IC50 values were recorded as 40.78 µg/mL for OH MeOH, while for OHNH and OHDCM, it was 9.77 and 7.10 µg/mL, respectively. The cytotoxic activity potential of sub‐extracts against lung cancer was remarkable.
According to the World Health Organisation (WHO) report, lung cancer ranks first in cancer‐related deaths and second among the most diagnosed cancers [ref. 29]. In this context, the need to discover new strategies and new compounds in the treatment remains essential. Lung cancer cells were the most susceptible to the cytotoxicity of the O. hispidus sub‐extracts among the cancer cell lines employed in this study. Therefore, to identify potent cytotoxic compounds against lung cancer, further research was conducted on OHNH and OHDCM.
In activity‐guided fractionation studies, OHNH and OHDCM were fractionated using the Sephadex LH‐20 CC, and OHNH Fr1‐4 and OHDCM Fr1‐2 subfractions were obtained. Each fraction was studied for cytotoxicity against A549 and Beas‐2B cells (Figures S3 and S4). Except for OHNH Fr4, all fractions of OHNH exerted potent activity (IC50 = 6.89‐12.05 µg/mL), and OHNH Fr2 was the most active one (Table 3). The fractions of the dichloromethane subextract, OHDCM Fr1 and OHDCM Fr2, showed potent dose‐dependent cytotoxicity against A549 (IC50 = 4.92 and 3.20 µg/mL, respectively).
TABLE 3: Cytotoxic activity results of the O. hispidus subfraction of OHNH and OHDCM against A549 and Beas‐2B cell lines.
| Fraction | IC50 ± SEM (µg/mL) | SI | |
|---|---|---|---|
| A549 | Beas‐2B | ||
| OHNH Fr1 | 8.20 ± 1.28 | 3.37 ± 0.20 | 0.41 |
| OHNH Fr2 | 6.89 ± 0.63 | 4.23 ± 0.33 | 0.61 |
| OHNH Fr3 | 12.05 ± 1.36 | 10.50 ± 1.24 | 0.87 |
| OHNH Fr4 | >100 | 28.03 ± 11.91 | — |
| OHDCM Fr1 | 4.92 ± 0.52 | 4.49 ± 0.39 | 0.91 |
| OHDCM Fr2 | 3.20 ± 0.41 | 2.28 ± 0.28 | 0.71 |
| Doxcbdv70254-tbl3-note-0001 | 0.59 ± 0.05 | 0.07 ± 0.017 | 0.12 |
* The results of doxorubicin were given as IC50 ± SEM (µM). Dox: doxorubicin; SEM: Standard error of the mean; SI: selectivity index; OHNH: n‐hexane; OHDCM: dichloromethane.
Following sequential chromatographic techniques on active OHNH Fr2 and OHDCM Fr2 sub‐extracts, four compounds were isolated, and their cytotoxic effects were tested on A549 and Beas 2B (Figure S5). Compound 3, whose structure was elucidated for the first time in this study, exerted potent cytotoxicity against lung cancer cells (IC50 = 13.72 µM). The selectivity index of the compound 3 was calculated as 0.87. However, it was found to be 0.12 for the positive control, doxorubicin. The primary goal of finding an ideal anticancer compound is to develop a structure that targets tumors specifically while causing minimal or no harm to healthy cells. Although studies have been conducted with this approach, the drugs currently used in the clinics still face challenges. In the present study, for compound 3, whose selectivity was less than 1, in addition to advanced anticancer research, different approaches may need to be pursued to reduce its toxicity. Today, the efforts are continued that selectivity can be improved by changing the formulation forms of compounds, using smart drug technologies, or varying the physicochemical properties of the compound [ref. 30, ref. 31, ref. 32]. Compounds with coumarin skeleton (1, 2, and 4) did not show an antiproliferative effect at the determined concentrations (Table 4).
TABLE 4: Cytotoxic activity results of the isolated compounds of O. hispidus against A549 and Beas‐2B cell lines.
| Compound | IC50 ± SEM (µM) | SI | |
|---|---|---|---|
| A549 | Beas‐2B | ||
| 1 | >50 | >50 | — |
| 2 | >50 | >50 | — |
| 3 | 13.72 ± 0.80 | 11.97 ± 1.5 | 0.87 |
| 4 | >50 | >50 | — |
| Dox | 0.59 ± 0.05 | 0.07 ± 0.017 | 0.12 |
Dox: doxorubicin; SEM: standard error of the mean; SI: selectivity index.
Chalcones are known for their simple chemistry, ease of synthesis, and suitability for derivatization. These compounds attracted attention due to a wide range of biological activities such as antidiabetic [ref. 33], antihypertensive [ref. 34], antiviral [ref. 35], anti‐inflammatory [ref. 36], antihistaminic [ref. 37], antioxidant [ref. 38], antimalarial [ref. 39], and anticancer [ref. 40]. Chalcones display notable anticancer activity through both structural features and cellular mechanisms [ref. 41]. 2′,4′‐dihydroxy‐3′‐methoxy‐3,4‐methylenedioxy‐8‐hydroxymethylene dihydrochalcone isolated from Sansevieria cylindrica Bojer ex Hook (Dracaenaceae) showed a moderate cytotoxic effect against MCF‐7 cells (IC50 = 34 µg/mL) [ref. 42]. Erioschalcone A isolated from Eriosema glomeratum (Guill. & Perr.) Hook.f. (Fabaceae) and its semi‐synthetic derivative 4‐(3‐(4‐methoxyphenyl)propanoyl)‐2‐(3‐methylbut‐2‐en‐1‐yl)‐1,3‐phenylenediacetate exhibited strong antiproliferative effect in A549, MCF‐7 and HeLA cells [ref. 43]. Inhibition of different targets such as aromatase, CDC25B, topoisomerase‐II, ABCG2/P‐gp/BCRP, 5α‐reductase, HDAC/Situin‐1, proteasome, VEGF, VEGFR‐2 kinase, 17‐β‐hydroxysteroid dehydrogenase, MMP‐2/9, JAK/STAT signaling pathways, tubulin, cathepsin‐K, Wnt, NF‐κB, B‐Raf, and mTOR played a role in the anticancer activity mechanism. Replacing the aryl ring with electron‐withdrawing/donating groups and/or replacing the aryl with a heteroaryl ring was found to increase the activity in structure‐anticancer activity studies [ref. 40, ref. 41, ref. 42, ref. 43].
Chalcones isolated from Apiaceae species, such as those found in Ferula and related genera, display remarkable anticancer properties through multiple mechanisms of action. Prenylated chalcones, including kayserin A and B from F. caspica roots, have been shown to induce apoptosis in COLO 205, K‐562, and MCF‐7 cells via caspase‐3/8/9 activation and suppression of Bcl‐xL expression [ref. 44]. Similarly, the prenylated chalcone xanthohumol exhibits strong cytotoxic activity across prostate, breast, pancreatic, and leukemia cell lines; it promotes mitochondrial depolarization, upregulates ROS, triggers caspase cleavage, inhibits survival signaling (Akt, NF‐κB, mTOR), and disrupts VEGF‐driven angiogenesis in vitro and in tumor xenografts [ref. 45]. These bioactivities align with broader findings on structure‐activity relationship (SAR) in the Apiaceae family. Diprenylation often enhances lipophilicity and membrane affinity, which strengthens interaction with mitochondrial or signaling targets, while combinations of hydroxyl and methoxy substitutions modulate redox balance and pathway inhibition [ref. 46]. Collectively, chalcones from Apiaceae exert a polypharmacological approach, including apoptosis induction, ROS generation, survival‐pathway blockade, and anti‐angiogenic effects, that underscores their potential as promising leads in anticancer drug development.
The newly isolated chalcone‐type compound, 2′,6’‐dihydroxy‐4’‐methoxy‐3”‐(γ,γ‐dimethylallyl)‐4”‐hydroxy β‐hydroxydihydrochalcone (compound 3), incorporates several structural elements that are closely associated with anticancer activity according to SAR studies. Hydroxyl groups at the 2′ and 6′ positions enhance reactive oxygen species (ROS) formation and support mitochondrial apoptosis, as seen in other chalcone analogues [ref. 47]. The 4′‐methoxy group is known to improve membrane permeability and bioavailability without dramatically raising lipophilicity, consistent with findings in 2,4,5‐trimethoxy chalcone studies, where para electron‐donating substitutions increased cytotoxicity in MCF‐7, SW‐982, and HeLa cells [ref. 48]. Incorporation of a prenyl (γ,γ‐dimethylallyl) group at 3″ likely boosts lipophilicity and membrane localization, which has been shown to enhance selectivity and cell uptake . Yet, maintaining optimal lipophilicity is critical since SAR assessments revealed that excessive lipophilicity can impair cellular permeability in K562 and SH‐SY5Y models [ref. 49]. Although the β‐hydroxydihydrochalcone backbone lacks the usual α,β‐unsaturated carbonyl, the saturated β‐hydroxy form may confer increased metabolic stability while still engaging alternative targets. Methoxy and hydroxyl substitutions are also known to disrupt survival signaling via NF‐κB or PI3K/Akt pathways and to inhibit drug‐efflux transporters like ABCG2 with the 6′‐OH + 2′,4′‐OMe pattern identified as especially effective [ref. 47]. Collectively, these structural features suggest that compound 3 could act through mitochondrial apoptosis activation, redox modulation, signal‐pathway interference, and possible reversal of multidrug resistance, although confirmatory biochemical assays will be essential.
In Silico Analysis
Considering cytotoxic activity, according to the CLC‐Pred software, umbelliferone 6‐carboxylic acid (1) would not show cytotoxicity on any cell line. However, umbelliferone (2) may have cytotoxic activity against oligodendroglioma (Pa = 0.618; Pi = 0.021) and breast carcinoma (Pa = 0.504; Pi = 0.051). Nodakenetin (4) was estimated to be effective against promyeloblast leukemia (Pa = 0.524; Pi = 0.024) and small cell lung carcinoma (Pa = 0.508; Pi = 0.013). It was determined that compound 3 may show activity against small‐cell lung carcinoma (Pa = 0.547; Pi = 0.007), consistent with the results of our experimental study. For the new compound whose cytotoxic activity we identified in lung cancer cells, the first 10 genes listed for mRNA‐based up‐ and down‐regulation were determined (Table 5).
TABLE 5: Prediction results of gene expression alterations induced by the new compound 3.
| Up regulation | Down regulation | ||||
|---|---|---|---|---|---|
| Pa‐Pi | Genes | cbdv70254-tbl5-note-0001IAP, LOO CV | Pa‐ Pi | Genes | cbdv70254-tbl5-note-0001IAP, LOO CV |
| 0.943‐0.007 | PARP1 | 0.861 | 0.958‐0.003 | HK2 | 0.816 |
| 0.925‐0.006 | BIRC2 | 0.824 | 0.947‐0.004 | PARP1 | 0.807 |
| 0.922‐0.004 | SNORA74A | 0.902 | 0.942‐0.004 | ADD1 | 0.793 |
| 0.920‐0.004 | ZBTB10 | 0.799 | 0.932‐0.004 | IDNK | 0.764 |
| 0.915‐0.004 | SOD2 | 0.904 | 0.926‐0.003 | LRG1 | 0.865 |
| 0.913‐0.005 | HTRA2 | 0.762 | 0.923‐0.004 | COX10 | 0.762 |
| 0.908‐0.002 | EBLN2 | 0.910 | 0.916‐0.004 | CHML | 0.808 |
| 0.903‐0.004 | AP5Z1 | 0.859 | 0.913‐0.003 | SULT1A1 | 0.786 |
| 0.900‐0.009 | MSANTD3 | 0.762 | 0.912‐0.005 | PWP1 | 0.864 |
| 0.899‐0.004 | FZD2 | 0.774 | 0.907‐0.004 | ACE2 | 0.866 |
* IAP: invariant accuracy of prediction; LOO CV: leave‐one‐out cross‐validation.
Recently, the HK2 (hexokinase) gene has been shown to be a novel therapeutic target in lung cancer [ref. 50], and compound 3 was predicted as an inhibitor of HK2 in the current study. Investigating the interaction with this gene in future studies involving compound 3 would be beneficial. For the isolated compounds, the top 10 highest prediction scores among the different activities are given in Table 6.
TABLE 6: In silico biological activity results of isolated compounds.
| Compound | Biological activities | Probability (Pactive‐Pinactive) |
|---|---|---|
| 1 | 4‐Nitrophenol 2‐monooxygenase inhibitor | 0.936‐0.001 |
| CYP2C12 substrate | 0.938‐0.005 | |
| Chlordecone reductase inhibitor | 0.926‐0.004 | |
| Antiuremic | 0.917‐0.002 | |
| CYP2A11 substrate | 0.912‐0.002 | |
| Antiseptic | 0.903‐0.003 | |
| Glucan endo‐1,6‐beta‐glucosidase inhibitor | 0.900‐0.003 | |
| Aryl‐alcohol dehydrogenase (NADP+) inhibitor | 0.896‐0.002 | |
| UGT1A6 substrate | 0.893‐0.003 | |
| Cholestantriol 26‐monooxygenase inhibitor | 0.885‐0.003 | |
| 2 | CYP2C12 substrate | 0.976‐0.002 |
| CYP2A11 substrate | 0.941‐0.001 | |
| Chlordecone reductase inhibitor | 0.936‐0.003 | |
| Membrane integrity agonist | 0.937‐0.004 | |
| Aspulvinone dimethylallyl transferase | 0.933‐0.004 | |
| 4‐Nitrophenol 2‐monooxygenase inhibitor | 0.928‐0.002 | |
| CYP2B5 substrate | 0.915‐0.002 | |
| CYP2A4 substrate | 0.914‐0.001 | |
| Cardiovascular analeptic | 0.908‐0.003 | |
| Aryl‐alcohol dehydrogenase (NADP+) inhibitor | 0.906‐0.002 | |
| 3 | CDP‐glycerol glycerophosphotransferase inhibitor | 0.881‐0.013 |
| Membrane integrity agonist | 0.866‐0.020 | |
| Ubiquinol‐cytochrome‐c reductase inhibitor | 0.836‐0.021 | |
| CYP2D6 substrate | 0.817‐0.005 | |
| UDP‐glucuronosyltransferase substrate | 0.804‐0.006 | |
| UDP‐glucuronosyltransferase substrate | 0.804‐0.006 | |
| Aspulvinone dimethylallyltransferase inhibitor | 0.785‐0.038 | |
| Reductive | 0.740‐0.005 | |
| Lactase inhibitor | 0.733‐0.005 | |
| Choleretic | 0.720‐0.004 | |
| 4 | CYP2C12 substrate | 0.912‐0.009 |
| Antiischemic, cerebral | 0.871‐0.007 | |
| CYP2A11 substrate | 0.855‐0.003 | |
| Spasmolytic | 0.773‐0.005 | |
| Anti‐inflammatory | 0.759‐0.009 | |
| Spasmolytic, urinary | 0.755‐0.006 | |
| 4‐Nitrophenol 2‐monooxygenase inhibitor | 0.751‐0.005 | |
| CYP2A4 substrate | 0.722‐0.005 | |
| CYP2F1 substrate | 0.703‐0.005 | |
| CYP2A6 substrate | 0.694‐0.010 |
On the other hand, Table 7 displayed the compounds with the five highest probability scores for adverse and toxic effects. In further studies, these compounds can be investigated in terms of biological activities that are determined to have a biological effect based on the results of in silico analysis.
TABLE 7: Possible adverse and toxic effects of isolated compounds.
| Compound | Possible adverse and toxic effects | Probability (Pactive‐Pinactive) |
|---|---|---|
| 1 | Hematemesis | 0.891‐0.004 |
| Allergic contact dermatitis | 0.872‐0.004 | |
| Ulcer, aphthous | 0.854‐0.008 | |
| Toxic, vascular | 0.840‐0.010 | |
| Photoallergy dermatitis | 0.814‐0.003 | |
| 2 | Hematemesis | 0.880‐0.005 |
| Shivering | 0.852‐0.018 | |
| Toxic, vascular | 0.835‐0.011 | |
| Allergic contact dermatitis | 0.784‐0.008 | |
| Gastrointestinal hemorrhage | 0.780‐0.009 | |
| 3 | Sensitization | 0.763‐0.005 |
| Hepatotoxic | 0.763‐0.025 | |
| Panic | 0.718‐0.016 | |
| Edema | 0.712‐0.019 | |
| Dyspnea | 0.711‐0.020 | |
| 4 | Sedative | 0.566‐0.011 |
| Hepatitis | 0.586‐0.061 | |
| Hematemesis | 0.535‐0.061 | |
| Inflammation | 0.518‐0.073 | |
| Endocrine disruptor | 0.476‐0.054 |
Conclusions
In the present study, four secondary metabolites, including a previously undescribed β‐hydroxydihydrochalcone (3), were isolated from the whole plant parts of O. hispidus via cytotoxic activity‐guided isolation. At the tested concentrations, umbelliferone (2), umbelliferone 6‐carboxylic acid (1), and nodakenetin (4) did not show antiproliferative activity on the A549, while compound 3 exerted significant activity on the lung cancer cells. Moreover, compound 3 had lower cytotoxicity against normal cells than doxorubicin. Computer‐aided cytotoxicity predictions were also found to be consistent with experimental analysis. Additionally, the prediction analysis indicates that compound 3 affects a gene identified as a new therapeutic target in lung cancer is a positive result for this compound. The relatively high selectivity of compound 3 indicates that this compound deserves further anticancer research. To better understand the therapeutic potential of compound 3, additional studies are needed to clarify its molecular targets and elucidate its mechanism of action for evaluating therapeutic potential and safety.
Experimental
General Experimental Procedures
Column chromatography (CC) was carried out on silica gel 60 (0.063‐0.200 mm; Merck) and Sephadex LH‐20 (25‐100 µm; General Electric Healthcare). For the flash chromatography, Buchi pump modules (C‐601), control unit (C‐620), and fraction collector (C‐660) were utilized. Analytical and preparative thin layer chromatography (TLC) analyses were carried out on Aluminum TLC plate, silica gel coated with flourescent indicator F254 (Merck); the examination was established by spraying with 1% vanillin/sulphuric acid solution followed by heating at 105°C for 2–3 min, detected with an ultraviolet (UV) at 254 and 365 nm. 1H (500 MHz), 13C (100 MHz), COSY, HSQC, and HMBC NMR spectra were recorded on a Bruker AM in dimethyl sulfoxide (DMSO)‐d6 or CD3OD. Using the peaks of the remaining solvents (DMSO‐d6: 1H, 2.50 ppm; CD3OD: 1H, 3.31 ppm), the chemical shifts are given as δ values in parts per million (ppm). The coupling constants (J) were reported as Hz. The low‐resolution mass spectrometry (LRMS) and high‐resolution MS (HRMS) data were carried out by using an Agilent G6530B TOF/Q‐TOF mass spectrometer (Agilent Technologies, Santa Clara, CA, USA; negative mode; in m/z) and a Thermo Orbitrap Q‐Exactive mass spectrometer (Thermo Fisher Scientific Inc., Waltham, MA, USA), respectively. ECD and UV spectra of the new compound were recorded on a Jasco J‐815 CD spectrophotometer over the range of 200‐400 nm and a Shimadzu UV‐1800 UV‐Vis spectrophotometer, respectively. Chloroform was used as a blank both for ECD and UV spectra. Infrared (IR) was measured on a Perkin Elmer Spectrum 100 FTIR Spectrometer. Optical rotation was measured on KRÜSS Optronic (Germany).
Plant Material
O. hispidus (Friv.) Griseb. was collected from Seydişehir, Konya, in July 2020. The plant was identified by Prof. Dr. Osman Tugay from Selcuk University, Faculty of Pharmacy, Department of Pharmaceutical Botany. A voucher specimen (KNYA‐30.041) was deposited at the Herbarium of the Faculty of Science, Selcuk University, Konya, Türkiye.
Extraction and Isolation
The air‐dried whole plant parts of O. hispidus (1300 g) were powdered and macerated with methanol (5 × 10 L, 24 h). Following filtration, combined extracts were concentrated under reduced pressure. The crude MeOH extract (202.18 g) was suspended with 400 mL distilled water and fractionated with n‐hexane (10 × 600 mL), dichloromethane (8 × 600 mL), ethyl acetate (8 × 600 mL), and n‐butanol saturated with H2O (8 × 600 mL) in a separatory funnel by liquid‐liquid extraction. The crude MeOH extract and sub‐extracts were evaluated for their cytotoxicity by MTT assay. The active sub‐extracts, n‐hexane (OHNH‐O popanax h ispidus N‐Hexane) and dichloromethane (OHDCM‐O popanax h ispidus DiChloroMethane) were purified by successive chromatographic methods.
OHNH (31.27 g) was fractionated over a Sephadex LH‐20 CC (5 × 70 cm) eluted with MeOH to yield four main fractions (OHNH Fr1‐Fr4). The activities of these main fractions were evaluated on A549 and Beas 2B cell lines. The most active fraction, OHNH Fr2 (12.5 g), was separated by SP‐LH 20 CC employing MeOH to give six subfractions: Fr 2a‐2f. Fr 2f (25.7 mg) submitted to SP LH‐20 CC eluted with MeOH and purified via preparative TLC using CHCl3:MeOH:H2O (70:30:3) as mobile phase to yield compound 1 (5.5 mg). The subfraction Fr 2d (340 mg) was applied to the SP‐LH 20 column by eluting with s‐Hex:EtOAc:MeOH (7:4:1) to afford Fr 2d‐1‐ 2d‐5. Fr 2d‐3 (37 mg) was further purified by preparative TLC with s‐Hex:EtOAc:MeOH (7:4:1) to give compound 2 (1.8 mg).
OHDCM (26.32 g) was applied over a Sephadex LH‐20 column eluted with MeOH to yield two main fractions: OHDCM Fr1 and OHDCM Fr2. The most active fraction, OHDCM Fr2 (3.5 g), was separated by Sigel column chromatography eluting with a stepwise n‐Hex:EtOAc:MeOH gradient to obtain Fr2a‐2o subfractions. Fr 2b (940 mg) was subjected to Sephadex LH‐20 and eluted with MeOH to obtain Fr 2b‐1‐2b‐4. Fr 2b‐3 was purified by preparative TLC using n‐Hex:EtOAc (70:40) to afford compound 3 (10 mg). The subfraction Fr 2i (90 mg) was separated by LiChroprep C18‐MPLC using a stepwise H2O:MeOH gradient (65:35→25:75) to yield 2i‐1‐2i‐6. Compound 4 (6.6 mg) was isolated from 2i‐3 using preparative TLC eluting CHCl3:MeOH:H2O (90:10:1).
Structure Elucidation
As a result of sequential chromatographic methods, two simple coumarins (1, 2), a new β‐hydroxydihydrochalcone (3), and a furanocoumarin (4) were isolated from O. hispidus. The structures of the isolated compounds were determined through spectroscopic analysis, including MS, 1D‐/2D‐NMR. The comparison of spectroscopic data with literature led to the characterization of the structures as umbelliferone 6‐carboxylic acid (1) [ref. 51], umbelliferone (2) [ref. 51], and nodakenetin (4) [ref. 52].
Compound 3: Grayish white powder; [α]20D = ‐148 (c = 0.02, MeOH); UV (MeOH) λmax 287 nm; 1H NMR (500 MHz in CD3OD) and 13C NMR (CD3OD, 125 MHz); HRESIMS (m/z) 371.1457 [M‐H]‐ (calcd. for C21H23O6, 371.1489) (Figures S6–S14; Table 1).
Cell Lines and Cell Culture
Adenocarcinomic human alveolar basal epithelial cell line (A549), human cervical cancer cell line (HeLa), human breast cancer cell line (MCF‐7), human hepatocellular carcinoma cell line (HepG2), and human bronchial epithelial cell line (BEAS‐2B) were used for the cytotoxicity test panel of crude extract. O. hispidus sub‐extracts showed high sensitivity in lung cancer cells. Therefore, A549 was used in further activity‐guided fractionation and isolation studies. The cell lines were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 1% L‐glutamine, and 1% penicillin‐streptomycin. Cells were maintained in an incubator at 37°C under 5% CO2.
MTT Assay
MTT assay was used to determine the cytotoxic activity of the crude extract, subextracts, main fractions, and isolated compounds. MTT assay, called the “gold standard” of cell viability tests, is based on measuring metabolic activity. [ref. 53, ref. 54] The cells reaching sufficient confluence were counted and plated in 96‐well plates (6 × 103 cells/well). After 24 h, cells were treated with five concentrations of the extracts (1–100 µg/mL), main fractions (1–100 µg/mL), or pure compounds (0.5–50 µM) in DMSO. Doxorubicin (10/3/1/0.3 µM) was used as a positive control. The plates were incubated with test materials for 48 h. After treatment, the supernatant was removed, and fresh medium and MTT solution (1 g/L in PBS) were added to each well. The cells were incubated for three h at 37°C. The resulting formazan crystals were dissolved in DMSO, and finally, the absorbance was detected at 570 nm using a spectrophotometer (SpectraMax i3x; Molecular Devices, San Jose, CA, USA). All data were collected from three independent experiments, and at least n = 6 data points were obtained. IC50 values were calculated as µg/mL for extracts or fractions and as µM for pure compounds.
Selectivity Index
The selectivity index (SI), which indicates the selectivity between cancer and normal cells, was calculated. The compounds with an SI greater than 1 are more likely to inhibit cancer cells than non‐cancerous cells [ref. 55]. SI values were calculated by the formula of [(IC50 values of non‐cancerous cells)/(IC50 values of cancerous cells)].
In Silico Analysis
The isolated compounds were evaluated in terms of anticancer and different biological activities by in silico analysis. For this purpose, PASS (Prediction of Activity Spectra for Substances) and CLC‐Pred (Cell Line Cytotoxicity Predictor) programs were used [ref. 56]. PASS evaluates the overall biological activity potential of tested molecules. It predicts more than 4000 activities. In this software, Pa and Pi values represent high and low probabilities of being active or inactive, respectively. In silico prediction of the cytotoxicity was performed using CLC‐Pred (Cell Line Cytotoxicity Predictor [2.0]) [ref. 57]. Additionally, the DIGEP‐Pred 2.0 (prediction of drug‐induced changes of gene expression profile) program was utilized to understand the alterations in gene expression related to the identified cytotoxic compound.
ECD Computational Analysis
The initial conformational analysis of compound 3 was performed using the semi‐empirical PM3 method [ref. 58] with the aid of the SPARTAN’04 program package. The resulting minimum‐energy conformers were subsequently optimized using DFT at the B3LYP/6‐31G(d) level, as implemented in the Gaussian 09 software package [ref. 59].
The predominant conformers were then subjected to theoretical ECD calculations using TDDFT at the B3LYP/6‐311G(d,p) level in chloroform [ref. 60, ref. 61], modeled using the conductor‐like polarizable continuum model (CPCM) [ref. 62]. The energies, oscillator strengths, and rotational strengths of each conformer were calculated with the Gaussian 09 software package. The theoretical ECD spectra of each conformer were simulated using Gaussian band‐shape functions.
Statistical Analysis
Data were analyzed using GraphPad Software Prism 8.0 (San Diego, CA, USA; demo version). The significance of differences in means of the cell viability % of each extract, fraction, and isolated compounds on cells was determined by one‐way analysis of variance (ANOVA), followed by Tukey’s multiple comparison tests. Nonlinear regression analysis (dose‐response) was used to determine the IC50 values. p < 0.05 was considered statistically significant.
Author Contributions
Kevser Taban: writing original draft, methodology, investigation, formal analysis, and data curation. İpek Süntar: supervision, methodology, investigation, formal analysis, data curation, conceptualization, writing – review and editing. Perihan Gürbüz: methodology, investigation, data curation, writing – review and editing. Esra Emerce: validation, software, and methodology. Osman Tugay: investigation and resources. Şengül Dilem Doğan: methodology, investigation, and data curation. Beyza Hamur: investigation and formal analysis. Ahmet Ceyhan Gören: investigation and formal analysis.
Conflicts of Interest
The authors declare no conflict of interest.
Supplementary Materials
References
- Natural Products/Bioactive Compounds as a Source of Anticancer Drugs,”. Cancers, 2022. [PubMed]
- The Apiaceae: Ethnomedicinal family as Source for Industrial Uses,”. Industrial Crops and Products, 2017
- Structure Based Multitargeted Molecular Docking Analysis of Selected Furanocoumarins Against Breast Cancer,”. Scientific Reports, 2019. [PubMed]
- Cytotoxic Activity of the Genus Ferula (Apiaceae) and its Bioactive Constituents,”. Avicenna Journal of Phytomedicine, 2018. [PubMed]
- Isolation of Anticancer Compounds From Peucedanum japonicum Thunb. Roots,”. Korean Journal of Plant Resources, 2014
- Assessment of the Antitumor Potential of Umbelliprenin, a Naturally Occurring Sesquiterpene Coumarin,”. Biomedicines, 2020. [PubMed]
- Chemical Profile, Antioxidant, Antimicrobial, Enzyme Inhibitory, and Cytotoxicity of Seven Apiaceae Species From Turkey: A Comparative Study,”. Industrial Crops and Products, 2020
- Antioxidant and Antimicrobial Potential of Opopanax hispidus (Apiaceae) Extracts,”. Lekovite Sirovine, 2015
- Phytochemistry, Traditional Uses and Pharmacological Properties of the Genus Opopanax W. D. J. Koch: A Mini‐Review,”. Pharmaceutical science, 2020
- A preliminary Study on the Antioxidant and Anti‐inflammatory Activities of Opopanax hispidus ,”. Journal of Faculty of Pharmacy of Ankara University, 2021
- 11 B. Javadi , M. Iranshahy , and S. A. Emami , Anticancer Plants in Islamic Traditional Medicine, Complementary Therapies for the Body, Mind and Soul, ed. M. Saad (InTech Open, 2015), 111–144.
- Coumarins From Opopanaxchironium. New Dihydrofuranocoumarins and Differential Induction of Apoptosis by Imperatorin and Heraclenin,”. Journal of Natural Products, 2004. [PubMed]
- Edible Wild Plants and Their Consumption During Winter in a Rural Village on Mount Ida (Kazdağı),”. Bocconea, 2012
- A Review of Edible Plants on the Turkish Apiaceae Species,”. Istanbul Journal of Pharmacy, 2014
- Turkish Folk Medicinal Plants. Part II: Eğirdir (Isparta),”. Fitoterapia, 1999
- Ethnopharmacological Approach to the Herbal Medicines of the “Antidotes” in Nikolaos Myrepsos’ Dynameron,”. Journal of Ethnopharmacology, 2015. [PubMed]
- Ethnobotanical Knowledge of Apiaceae Family in Iran: A Review,”. Avicenna Journal of Phytomedicine, 2016. [PubMed]
- Folk Medicines in the Villages of Ilıca District (Erzurum, Turkey),”. Turkish Journal of Biology, 2012
- Antioxidant Capacities of some Food Plants Wildly Grown in Ayvalik of Turkey,”. Food Science and Technology Research, 2009
- Effect of The Opopanax hispidus Plant’s Aerial Parts Extract on Mice Ovary,”. Selçuk Tıp Dergisi, 2020
- Fatty Acid Composition, Enzyme Inhibitory, and Antioxidant Activities of the Ethanol Extracts of Selected Wild Edible Plants Consumed as Vegetables in the Aegean Region of Turkey,”. International Journal of Food Properties, 2017
- Chemical Composition of the Essential Oil of Opopanax hispidus ,”. Chemistry of Natural Compounds, 2018
- A New Dihydrofuranocoumarin From Opopanax hispidus (Friv.) Griseb.,”. Natural Product Research, 2014. [PubMed]
- A New β‐hydroxydihydrochalcone From Tephrosia uniflora, and the Revision of Three β‐hydroxydihydrochalcones to Flavanones,”. Fitoterapia, 2022. [PubMed]
- Antiplasmodial β‐hydroxydihydrochalcone From Seedpods of Tephrosia elata ,”. Phytochemistry Letters, 2009
- Novel Flavonoids From the Aerial Parts of Unexplored and Endangered Wild Nutmeg Species Myristica beddomei subsp. Spherocarpa W.J. de Wilde,”. Phytochemistry Letters, 2021
- β‐Hydroxydihydrochalcone and Flavonoid Glycosides Along With Triterpene Saponin and Sesquiterpene From the Herbs of Pimpinella rhodantha Boiss.,”. Natural Product Research, 2016. [PubMed]
- Balanochalcone, a New Chalcone From Balanophora laxiflora Hemsl.,”. Natural Product Research, 2018. [PubMed]
- 29 J. Ferlay , M. Ervik , F. Lam , et al., Global Cancer Observatory: Cancer Today (International Agency for Research on Cancer, 2025).
- Anticancer Activity of Liposomal Formulation Co‐encapsulated With Coumarin and Phenyl Butyric Acid,”. Journal of Applied Pharmaceutical Science, 2024
- A New Approach for Enhancing Differential Selectivity of Drugs to Cancer Cells,”. ACS Chemical Biology, 2006. [PubMed]
- Achievement of the Selectivity of Cytotoxic Agents against Cancer Cells by Creation of Combined Formulation with Terpenoid Adjuvants as Prospects to Overcome Multidrug Resistance,”. International Journal of Molecular Sciences, 2023. [PubMed]
- Chalcones and Their Therapeutic Targets for the Management of Diabetes: Structural and Pharmacological Perspectives,”. European Journal of Medicinal Chemistry, 2015. [PubMed]
- Synthesis and Evaluation of Chalcone Analogues Based Pyrimidines as Angiotensin Converting Enzyme Inhibitors,”. Pakistan Journal of Biological Sciences, 2013. [PubMed]
- A Comprehensive Review on the Antiviral Activities of Chalcones,”. Journal of Drug Targeting, 2021. [PubMed]
- Chalcone Derivatives: Anti‐inflammatory Potential and Molecular Targets Perspectives,”. Current Topics in Medicinal Chemistry, 2017. [PubMed]
- Chemical and Biological Potentials of Chalcones: A Review,”. Organic & Medicinal Chemistry International Journal, 2015
- Anticancer and Antioxidant Activity of Synthetic Chalcones and Related Compounds,”. Cancer Letters, 1995. [PubMed]
- The Novel Oxygenated Chalcone, 2,4‐Dimethoxy‐4’‐Butoxychalcone, Exhibits Potent Activity Against Human Malaria Parasite Plasmodium falciparum in Vitro and Rodent Parasites Plasmodium berghei and Plasmodium yoelii in Vivo,”. Journal of Infectious Diseases, 1997. [PubMed]
- Anti‐cancer Chalcones: Structural and Molecular Target Perspectives,”. European Journal of Medicinal Chemistry, 2015. [PubMed]
- Bioactive Natural Products: Flavonoids and Anticancer Activity: Structure–Activity Relationship,”. Studies in Natural Products Chemistry, 2022
- New Cytotoxic Dihydrochalcone and Steroidal Saponins From the Aerial Parts of Sansevieria Cylindrica Bojer Ex Hook,”. Phytochemistry Letters, 2017
- Cytotoxic Potential of Dihydrochalcones From Eriosema Glomeratum and Their Semi‐synthetic Derivatives,”. Natural Product Research, 2024. [PubMed]
- Sesquiterpene Coumarins, Chromones, and Acetophenone Derivatives With Selective Cytotoxicities From the Roots of Ferula Caspica M. Bieb. (Apiaceae),”. Pharmaceuticals, 2024. [PubMed]
- Growth Inhibitory and Apoptosis‐inducing Effects of Xanthohumol, a Prenylated Chalcone Present in Hops, in Human Prostate Cancer Cells,”. Anticancer Research, 2010. [PubMed]
- Chalcone Derivatives: Role in Anticancer Therapy,”. Biomolecules, 2021. [PubMed]
- Investigation of Chalcones as Selective Inhibitors of the Breast Cancer Resistance Protein: Critical Role of Methoxylation in both Inhibition Potency and Cytotoxicity,”. Journal of Medicinal Chemistry, 2012. [PubMed]
- Synthesis, Anticancer and Antioxidant Activities of 2,4,5‐trimethoxy Chalcones and Analogues From Asaronaldehyde: Structure–activity Relationship,”. European Journal of Medicinal Chemistry, 2013. [PubMed]
- Halogenated Chalcones With High‐Affinity Binding to P‐Glycoprotein: Potential Modulators of Multidrug Resistance,”. Journal of Medicinal Chemistry, 1998. [PubMed]
- Changes in VGLUT1 and VGLUT2 Expression in Rat Dorsal Root Ganglia and Spinal Cord Following Spared Nerve Injury,”. Neurochemistry International, 2016. [PubMed]
- In Vitro Antioxidant and Anti‐inflammatory Activities of Angelica Decursiva,”. Archives of Pharmacal Research, 2012. [PubMed]
- Isolation and Purification of Coumarin Compounds From the Root of Peucedanum decursivum (Miq.) Maxim by High‐speed Counter‐current Chromatography,”. Journal of Chromatography A, 2005. [PubMed]
- Limitations of the 3‐(4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyl‐2H‐tetrazolium bromide (MTT) assay when compared to three commonly used cell enumeration assays,”. BMC Research Notes, 2015. [PubMed]
- 54 E. Emerce and K. Taban Akça , Cytotoxic Activity Methods, Methods for Preclinical Evaluation of Bioactive Natural Products, ed. I. Suntar , D. Barreca , and L. Milella (Bentham Science Publisher, 2023): 149–176.
- Selective Cytotoxic Activities of Two Novel Synthetic Drugs on Human Breast Carcinoma MCF‐7 Cells,”. Anticancer Research, 2009. [PubMed]
- Prediction of the Biological Activity Spectra of Organic Compounds Using the Pass Online Web Resource,”. Chem. Heterocycl. Compd., 2014
- CLC‐Pred: A Freely Available Web‐service for in silico Prediction of human Cell Line Cytotoxicity for Drug‐Like Compounds,”. PLoS ONE, 2018. [PubMed]
- Optimization of Parameters for Semiempirical Methods V: Modification of NDDO Approximations and Application to 70 Elements,”. Journal of Molecular Modeling, 2007. [PubMed]
- Gaussian 09, Rev. C 01; Gaussian, Inc., Wallingford CT, (. 2009
- Density‐functional Thermochemistry. III. The Role of Exact Exchange,”. Journal of Chemical Physics, 1993
- Density‐Functional Theory for Time‐Dependent Systems,”. Physical Review Letters, 1984
- Quantum Calculation of Molecular Energies and Energy Gradients in Solution by a Conductor Solvent Model,”. Journal of Physical Chemistry A, 1998
