Strategy advancements in placental pharmacokinetics: from in vitro experiments to in silico prediction
Abstract
Background:
The placental barrier is a critical interface that regulates drug transport between maternal and fetal circulation and is an important component in assessing fetal drug-exposure risk. Since pregnant women are often excluded from clinical trials, pharmacokinetic (PK) analysis data on placental drug transport remain limited. Currently, in vitro experiments and in silico simulation strategies are the primary and effective means for understanding drug transport across the placenta.
Method:
Various in vitro experimental methods, including cell monolayer models, ex vivo placental perfusion, and organ-on-a-chip platforms, along with model-based computational simulations, were systematically reviewed. The advantages, limitations, and potential future applications of these methods were evaluated.
Result:
A total of seven studies using cell models, 28 employing ex vivo perfusion, six utilizing placenta-on-a-chip technology, and 39 focusing on in silico simulations, were identified, involving 8, 34, 5, and 42 drugs, respectively. Antiviral agents, antibiotics, and opioids were the most frequently investigated drug types. Overall, in silico simulations informed by in vitro data as baseline parameters and constraints demonstrated higher predictive accuracy. Integrating multi-model data was shown to be a reliable strategy for improving the precision of placental PK studies.
Conclusion:
This review highlights the current strategies in placental PK research and supports safer drug use during pregnancy. Multi-model data integration is essential for developing reliable and quantitative fetal drug-exposure assessment frameworks, thus addressing data gaps caused by the exclusion of pregnant women from clinical trials.
Article type: Review Article
Keywords: pharmacokinetics, placental drug transfer
Affiliations: Department of Pharmacy, Personalized Drug Research and Therapy Key Laboratory of Sichuan Province, Sichuan Provincial People’s Hospital, School of Medicine, University of Electronic Science and Technology of China, Chengdu, China; Department of Pharmacy, Chongqing Red Cross Hospital (Jiangbei District People’s Hospital), Chongqing, China; Department of Pharmacy, Affiliated Hospital of Southwest Jiaotong University, The Third People’s Hospital of Chengdu, Chengdu, China
License: Copyright © 2025 Li, Wu, Zeng, Wang, Zhang, Li, Yang and Yang. CC BY 4.0 This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
Article links: DOI: 10.3389/fphar.2025.1694886 | PubMed: 41221050 | PMC: PMC12597936
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.2 MB)
Highlights
- This review systematically examined state-of-the-art in vitro modeling methods, including cell monolayer models, ex vivo placental perfusion experiments, organ-on-a-chip platforms, and model-guided computer simulations, and it critically appraised their respective strengths and limitations in quantifying transplacental drug transfer.
- Physiologically based pharmacokinetic (PBPK) models simulate the absorption, distribution, metabolism, and excretion (ADME) of drugs and are a powerful tool for predicting fetal drug exposure during pregnancy.
- The implementation of an integrated multi-model approach significantly enhances the reliability of maternal–fetal pharmacokinetic (PK) assessments, providing robust evidence to support guideline updates and clinical decision-making during pregnancy.
1 Introduction
Many pregnant women require one or more medications for health problems (ref. Be et al., 2025; ref. Lupattelli et al., 2014). Drug use during pregnancy is on the rise (ref. Subramanian et al., 2023; ref. Phillips et al., 2024), and recent studies have shown an increase in the use of at least one prescribed medication from 56.9% in 1998 to 63.3% in 2018 and an increase in the concurrent use of multiple medicines from 24.8% to 35.2% in the Danish population (ref. Thunbo et al., 2024). Although drugs have therapeutic significance in clinical practice, their safety in mothers and fetuses remains unclear. Maternal drug use may affect the fetus, leading to developmental abnormalities (ref. Foster et al., 2022; ref. De Felice and Kane, 2021) or long-term health issues (ref. Abolhassani et al., 2023; ref. Owen et al., 2021). Therefore, fetal drug exposure and safety assessments have gradually become important topics in clinical pharmacology.
The effects of maternal drug use on the fetus are complex and sensitive and require a multifaceted assessment. The syncytiotrophoblast (STB) and its two polarized plasma membranes, the maternal-facing microvillous membrane (MVM) and fetal-facing basal membrane (BM), represent the primary barrier in the human placenta, controlling transplacental transfer of small solutes. Key factors, including the physicochemical properties of the drug (MW, logP, pKa, protein binding, transporters, and metabolism) and placental function at different time points, affect the ability of the drug to cross the placental barrier during pregnancy. Due to the complexity of the placental structure, the assessment of transplacental drug transfer is a long-standing and difficult issue. Understanding and quantifying drug transport through the placenta can inform the assessment of fetal drug exposure. However, pregnant women are often excluded from clinical and post-marketing studies. This has led to a lack of PK data regarding drug use during pregnancy, particularly data related to placental drug transfer (ref. Pariente et al., 2016). Therefore, considering the mismatch between the available evidence from existing studies and real-world medication needs during pregnancy (ref. Haas et al., 2018), PK research on placental drug transfer remains a challenging and long-term endeavor.
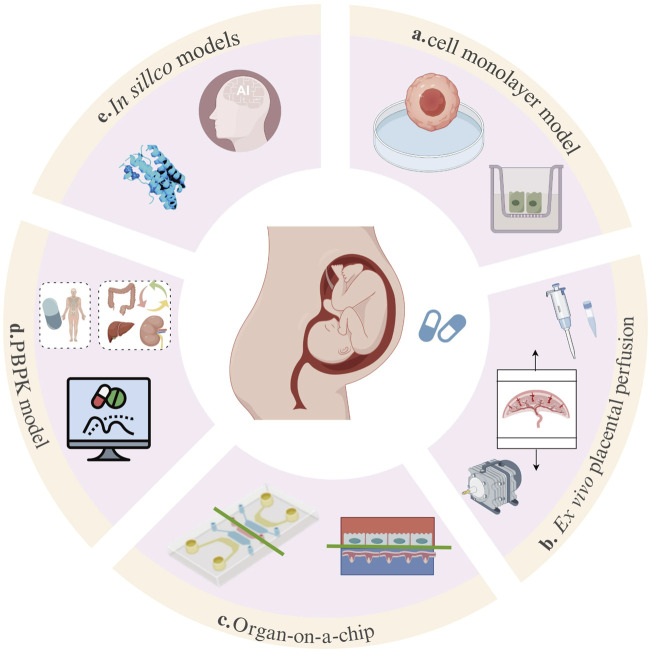
In recent years, with the advancement of computer and bioengineering technologies, methods for placental PK studies have developed considerably. In addition to the previously established in vitro methods (e.g., cell monolayer model, Figure 1a; ex vivo placental perfusion experiment; Figure 1b), several new attempts have been made (for example, organ-on-a-chip Figure 1c; PBPK model, Figure 1d; in silico models, Figure 1e). Multiple approaches have been attempted, including the combination of PBPK models with in vitro and ex vivo data. However, systematic evaluation and synergistic integration of these methods have not yet been fully explored. This review systematically discusses the evolution, application scenarios, and limitations of various placental drug transfer methods in the context of drug studies. We have also attempted to explore the future directions of these research methods. This study ultimately aims to contribute to the methodological advancement of placental pharmacokinetics and risk assessment for medication use during pregnancy.
2 Literature review procedure
A systematic literature search was performed in PubMed (https://pubmed.ncbi.nlm.nih.gov/) from inception to February 2025 using the keywords “placental drug transfer,” “placental pharmacokinetics,” and “foetal drug exposure.” Two team members independently screened the titles and abstracts, and any controversial finding was resolved through discussion with a third researcher. Studies were included if they investigated drug transfer across the human placenta using cell monolayer models, ex vivo placental perfusion, organ-on-a-chip platforms, or in silico simulations and reported relevant methodological outcomes. Studies not related to the human placenta, not addressing drug transfer across the placenta, lacking original data, or with insufficient methodological details were excluded. Figure 1 summarizes the in vitro models applied in transplacental drug transfer research. Detailed information on the progress, advantages, and limitations of each method was reviewed and summarized to highlight the future research directions for in vitro human placental models.
3 Cell experimental models for placental drug-transfer
Current placental transport models include primary trophoblasts, trophoblast cell lines (BeWo b30, JEG-3, JAR, and HTR-8/SVneo), stem cell-derived systems, and intestinal Caco-2 (cancer coli-2) cells, with BeWo b30 and primary trophoblasts remaining the gold standards according to the FDA/ICH guidelines. We mainly focus on placental barrier studies, with studies on BeWo cells focusing on the mechanism of drug transport across the placenta and the role of transport proteins, while Caco-2 cells are mainly used to assess drug permeability across the placental barrier, and therefore, the two cellular studies will be described in detail below.
3.1 Trophoblast cell lines
The BeWo cell line serves as a valuable in vitro model for placental transport studies. Derived from human choriocarcinoma, these cells form functional syncytialized monolayers when properly differentiated upon cAMP induction (ref. Drwal et al., 2018), exhibiting key characteristics of the placental barrier (ref. Moore et al., 2021). Syncytialized BeWo cells were employed as an in vitro model of the placental barrier to investigate drug-transport kinetics, following verification of barrier integrity through transepithelial electrical resistance (TEER) measurements (≥80 Ω · cm2).
The transporters expressed in the STB, including the ABC family members (P-gp, BCRP, and MRP) and SLC family members (OCT and OAT), bidirectionally regulate placental substance exchange through efflux (e.g., P-gp/BCRP/MRP-mediated excretion into maternal circulation) and uptake (e.g., OCT/OAT-mediated substance intake) mechanisms (ref. Bai et al., 2016). BeWo b30 cells, a subclone of the BeWo cell line, expresses important placental transporters (ref. Han et al., 2018), particularly BCRP and P-gp (ref. Ceckova et al., 2006), and it demonstrates compound permeability patterns that correlate well with ex vivo results (ref. Correia Carreira et al., 2011; ref. Li et al., 2013; ref. Poulsen et al., 2009). Researchers have successfully utilized this model to initially elucidate the transplacental transport of clinically relevant compounds spanning antivirals, opioids, and fluoroquinolones (Table 1).
TABLE 1: Details of placental drug-transfer study by the cell experimental models.
| Cell line | Origin | Drug | Type | Key findings | Ref. |
|---|---|---|---|---|---|
| Caco-2 cell | Colon cancer cells | Acetaminophen | Quantitative | CLp: 4354 | ref. Mian et al. (2020) |
| Colon cancer cells | Nifedipine | Quantitative | CLp: 3779 | ref. Werdan Romão et al. (2024) | |
| Colon cancer cells | Vancomycin | Quantitative | CLp: 27 | ref. Yan et al. (2024) | |
| BeWo cell | Choriocarcinoma cells | Opioids | Quantitative | P (heroin): 0.21; P (oxycodone): 2.46 | ref. Mortensen et al. (2019) |
| Choriocarcinoma cells | Abacavir | Descriptive | TTPTs: ENT1 | ref. Cerveny et al. (2018) | |
| Choriocarcinoma cells | ZidovudineEmtricitabine | Descriptive | TPTs: without ENTs | ref. Karbanova et al. (2017) | |
| Choriocarcinoma cells | Fluoroquinolone | Descriptive | TPTs (LVFX): BCRP、OAT、 MCT | ref. Po et al. (2018) |
Abbreviations: CLp (mL/min), transplacental clearance; P (×10−5 cm/s): the permeability across the cells; TPTs, placental transporter proteins; ENT, equilibrative nucleoside transporter; LVFX, levofloxacin; BCRP, breast cancer resistance protein; OAT, organic anion transporter; MCT, monocarboxylate transporter.
However, this model has notable limitations. First, a comparative transcriptomic study of human placental in vitro models revealed substantial numbers of differentially expressed genes across all in vitro placental models. To date, no placental cell line has been able to accurately mimic human placental tissue (ref. Lapehn et al., 2024). Although steroidogenic enzymes such as 3β-hydroxysteroid dehydrogenase type 1 (3β-HSD1) and 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) are highly expressed and functionally active in the in vivo placenta, their expression levels in trophoblast cell lines differ from those in the in vivo placenta (ref. Karahoda et al., 2021). Similarly, cytochrome P450 enzymes, including CYP2C8, CYP2C9, and CYP2J2, are poorly expressed (ref. Kammala et al., 2022). Second, the expression or localization of placental cellular transporters may also vary with temporal placental development; thus, the cell model cannot fully replicate the dynamic changes in transporter expression that occur during different gestational stages (ref. Veiga-Lopez et al., 2025). Future research should focus on three key directions to enhance BeWo-based placental model characteristics (ref. Be et al., 2025): developing microfluidic co-culture systems integrating BeWo cells with human umbilical vein endothelial cells (HUVECs) (ref. Fuenzalida et al., 2024) under physiological shear stress; (ref. Lupattelli et al., 2014) applying CRISPR editing to introduce disease-specific mutations; (ref. Subramanian et al., 2023) establishing 3D organoid models through BeWo-fibroblast co-culture. These advanced models require standardized validation, including cAMP-induced syncytialization, TEER monitoring, and transporter functionality assays, to ensure regulatory acceptance and enhance the predictive accuracy in placental drug-transport studies.
3.2 Caco-2 cell
The Caco-2 cell model is widely used to study drug permeability (ref. Artursson et al., 2001). Derived from human colon cancer cells, Caco-2 cells differentiate into polarized monolayers that resemble small intestinal epithelial cells and mimic the barrier function of the human intestine (ref. Sambuy et al., 2005). Over the past 2 decades, the Caco-2 monolayer model has become a crucial tool for preclinical drug development, particularly for permeability screening (ref. Panse and Gerk, 2022). The Caco-2 monolayer allows the study of all major absorption pathways, such as passive transcellular and paracellular diffusion and carrier-mediated active and facilitated transport (ref. Volpe, 2020).
Caco-2 cells form a continuous monolayer barrier for intestinal barrier simulation, and similarly, researchers have adapted this well-characterized system as a functional surrogate for placental barrier studies, leveraging its reproducible tight junction formation and transport properties. Mian et al. utilized a confluent Caco-2 monolayer system in Transwell chambers, with pre-experimental TEER measurements confirming barrier integrity (ref. Mian et al., 2020). This established model served as an effective placental barrier surrogate for characterizing acetaminophen transport and determining the apparent permeability coefficient (Papp) across the placental barrier. This experimental approach has been similarly applied to evaluate the transplacental permeability of various therapeutic agents, including nifedipine and vancomycin (ref. Werdan Romão et al., 2024; ref. Yan et al., 2024) (Table 1).
The utility of Caco-2 cells as an in vitro model for placental transport research is constrained by several inherent biological and methodological limitations. For instance, there are biological differences between Caco-2 cells and placental epithelial cells, which may lead to overestimation or underestimation of drug permeability. Caco-2 cells express key efflux transporters found in the STB, including P-glycoprotein (P-gp/ABCB1), BCRP (ABCG2), and certain MRPs (ABCC family) (ref. Bai et al., 2016), but they show limited expression of uptake transporters such as OCTs and OATs (ref. Wenzel et al., 2021). Therefore, Caco-2 cells are suitable for studying efflux-mediated transport but have limitations in modeling placental uptake processes. To bridge physiological gaps between models and human placenta, researchers employ genetic engineering strategies to upregulate key protein expression (ref. Ohta et al., 2020) and apply biosimilar mucus to mimic native mucosal barriers (ref. Panse and Gerk, 2022). Nevertheless, such models provide valuable preliminary data when access to more physiologically relevant systems is limited.
3.3 Primary trophoblasts
Primary cytotrophoblasts (CTBs) isolated from term human placentas that spontaneously fuse and differentiate into STB (ref. Li and Schust, 2015)-like cells in vitro are used to investigate the functions of the STBs (ref. Hawkins et al., 2025) and placentas with multiple modifications (ref. Motomura et al., 2025). These cells maintain native transporter expression profiles (e.g., BCRP and P-gp) and metabolic activity, which are the most physiologically relevant in vitro cell model for studying human placental function, but unfortunately, CTBs are rarely used for placental barrier-related studies. In addition, trophoblast stem cells (hTSCs) were derived from human villous CTBs and human blastocysts (ref. Okae et al., 2018). These long-term stem cell cultures may give rise to CTBs, EVTBs, and STB-like cells that show transcriptomes similar to those of the corresponding primary trophoblasts (ref. Shannon et al., 2024), which have emerged as a powerful tool for modeling the placental cytotrophoblast in vitro (ref. Karakis et al., 2025). Given that the primary focus of our investigation centers on placental barrier function, a comprehensive discussion of this aspect falls beyond the scope of this study.
4 Organ experimental models for placental transfer
Ex vivo placental perfusion experiments, first proposed by Professor Maurice Panigel in 1967, have been continuously refined to address challenges such as revascularization and placental hypoxia resistance (ref. Schneider et al., 2021). As research has expanded in recent years, new methods of studying placental drug-transport have emerged. Organ-on-a-chip technology, which integrates microfluidics and cell culture, replicates the human organ microenvironment on a chip and accurately simulates the blood flow, oxygen exchange, and nutrient delivery.
4.1 Ex vivo placental perfusion experiments
Ex vivo placental perfusion experiments are crucial ex vivo models used to study the transport of exogenous substances and the effects of hormones (ref. Page, 1991). This model simulates the exchange of substances between the mother and fetus by creating specific external conditions and perfusion systems that closely mimic physiological processes in the human body. This model allows researchers to study the mechanisms of exogenous substance transfer, fetal exposure, and associated diseases (ref. Villalobos-Labra et al., 2023). In addition, this model can be used to study the effects of factors such as inflammation and oxidative stress on biochemical and physiological imbalances that are closely linked to conditions such as preeclampsia, fetal growth restriction, diabetes, and microbial infections (ref. Koren and Ornoy, 2018).
Ex vivo placental perfusion experiments comprised maternal and fetal reservoirs, the placenta, and a perfusion pump that simulated blood flow with controlled pressure and flow rate (Figure 2). A well-defined artery–vein pair on the fetal-side cotyledon with minimal branching was chosen for cannulation following the confirmation of intact placental integrity post-delivery (ref. Jovelet et al., 2015). The system only remains patent for 2–6 h, so only short-term assessments can be made, and the circuit design (open, semi-closed, or closed loop) determines the physiological relevance, ranging from first-pass transfer simulation to steady-state PK assessment. The transfer process is mathematically described by Fick’s first law using the transfer equations outlined by ref. Mian et al. (2020). Our review focused on 28 recent studies (2015–2025) employing ex vivo human placental perfusion to investigate transplacental kinetics of various therapeutics, including antivirals, psychotropics, and biologics (Table 2). This 10-year timeframe was selected to account for methodological advancements while ensuring data comparability. Notably, researchers exhibit a distinct preference for double-closed perfusion systems, which consistently operate for significantly longer durations than open-circuit configurations (avg. 3.6 h vs. 1.9 h). This operational divergence substantiates the superior suitability of double-closed systems for steady-state PK investigations. Small molecules dominate the studied drugs (30/34, 88%), exposing a critical evidence gap for biologics/antibodies despite their clinical relevance.
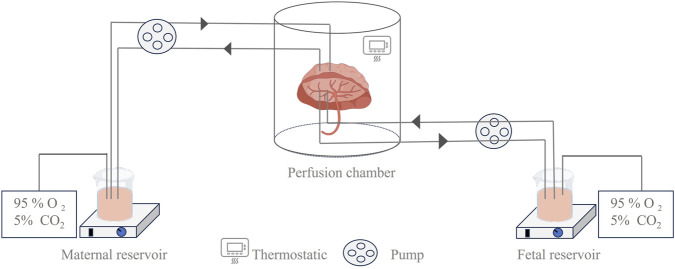
TABLE 2: Overview of studies that reported ex vivo placental perfusion models.
| Drug | Dosage | Perfusion system | Duration | FTR | CLI | CAI | TPTs/SF | Ref. |
|---|---|---|---|---|---|---|---|---|
| Acetaminophen | 1,000 mg | Double-closed | 3.5 h | 0.737 | 36 mL/min | NA | NA | ref. Mian et al. (2020) |
| Aldosterone | 1.5 × 10−9 M; 1.5 × 10−7 M | Open-circuit | 2 h | NA | NA | NA | TNF-α decreased; PlGF increased | ref. Mistry et al. (2022) |
| Bictegravir and cabotegravir | 550 ng/mL; 1,650 ng/mL | Open-circuit | 1.5 h | 7%, 10% | 22% and 21% | NA | NA | ref. Pencolé et al. (2020) |
| Bromocriptine | 10 ng/mL (low); 100 ng/mL (high) | Double-closed | 3 h | 0·37 ± 0·03 (low); 0·37 ± 0·03 (high) | NA | NA | NA | ref. Zheng et al. (2019) |
| Ciprofloxacin | 1.6 μg/mL | Double-closed | 3 h | 0.53 | 0.28 mL/min | 22% | NA | ref. Noergaard et al. (2021) |
| CZP | 200 μg/mL | Double-closed | 6 h | Undetected | NA | NA | CZP: FcRn non-binder | ref. Porter et al. (2016) |
| Doravirine | 0.96 mg/L | Double-closed | 3 h | 82% | 37.2 mL/min | NA | NA | ref. Bukkems et al. (2022) |
| Dolutegravir | 4 μg/mL | closed-open | 3 h | 0.57–1.51 | 1.03 ± 0.06 mL/min | NA | NA | ref. Freriksen et al. (2020) |
| Dolutegravir | 4.2 mg/L | Double-closed | 3 h | 0.6 ± 0.2 | NA | NA | NA | ref. Schalkwijk et al. (2016) |
| EMPA, DAPA, and CANA | 150 μg/L | Double-closed | 6 h | 0.38, 0.67, 0.62 | NA | NA | NA | ref. Kuoni et al. (2024) |
| Etanercept and infliximab | 100 μg/mL | Double-closed | 6 h | 0.04, 1.6 ± 0.4 | NA | NA | NA | ref. Eliesen et al. (2020) |
| Fexofenadine | 75 ng/mL | Double-closed | 1.5 h | 0.17–0.19 | NA | NA | NA | ref. Pinto et al. (2021) |
| Gefitinib, Imatinib, and Erlotinib | 500, 1,000, 1,500 ng/mL | Open-circuit | 1.5 h | 16.8%, 10.6%, and 31.4% | 0.59 ± 0.04, 0.48 ± 0.14, and 0.93 ± 0.03 | NA | NA | ref. Jovelet et al. (2015) |
| Lacosamide | 2.5 μg/mL; 10 μg/mL | Double-closed | 3 h | 0.94, 0.86 | NA | NA | SLC19A1 decreased | ref. Berman et al. (2022) |
| LMV, MBV | 480 mg and 400 mg | Open-circuit | 1.5 h | 0.09 ± 0.01,0.1 ± 0.01 | 0.35 ± 0.06 and 0.5 ± 0.07 | 0.04 ± 0.02, 0.04 ± 0.01 | NA | ref. Faure Bardon et al. (2020) |
| Omalizumab | 28.6 μg/mL; 45.8 μg/mL; 65.7 μg/mL; 107.0 μg/mL | Double-closed | 4 h | 0.02%–0.03% | NA | NA | PEA: doubled transfer; IgE: unchanged | ref. Kothari et al. (2022) |
| OP, OC | 65.2 ng/mL, 348 ng/mL | Double-closed | 3 h | 12.39% ± 3.26% (OC) and 10.17% ± 2.03% (OP) | 0.36 ± 0.11 and 0.29 ± 0.06 | NA | NA | ref. Huang et al. (2017) |
| Oxytocin | 0.8 ng/mL | Double-closed | 3 h | 15% (0 min) → 173% (180 min), equilibrium at 90 min | NA | NA | NA | ref. Nathan et al. (2021) |
| Pentoxifylline | 10 mg/L | Double-closed | 3 h | 0.9–1.0 | NA | NA | NA | ref. Broekhuizen et al. (2022) |
| Pravastatin | 0.2 μmol/L | Double-closed | 5 h | NA | NA | NA | PlGF/sFlt-1/sEng/p-eNOS: unaltered | ref. B et al. (2017) |
| Rivaroxaban | 250 ng/mL | Double-closed | 3 h | 0.69 | NA | NA | NA | ref. Bapat et al. (2015) |
| Rmd, GS | 4,000 ng/mL | Double-closed | 3 h | 8.15% and 9.9% | NA | NA | NA | ref. Louchet et al. (2023) |
| Sildenafil | 500 ng/mL | Double-closed | 3 h | 0.37 ± 0·03 (health); 0.47-0·66 (PE) | NA | NA | Normal: PDE5, eNOS, iNOS; decreased in PE:PDE1 | ref. Hitzerd et al. (2019) |
| Vancomycin | NA | Double-closed | NA | NA | 0.002421L/h/mL | NA | NA | ref. Yan et al. (2024) |
| Venlafaxine | 18.1 ± 2.1 μg/L | Double-closed | 3 h | 0.89 | NA | 22% | NA | ref. Hirschmugl and Wadsack (2022) |
Abbreviations: FTR, fetal–maternal ratio; CLI, clearance index; CAI, cotyledon accumulation index; TPTs, placental transporter proteins; SF, secreted factor; TNF-α, tumor necrosis factor-alpha; PlGF, placental growth factor; EMPA, empagliflozin; DAPA, dapagliflozin; CANA, canagliflozin; SGLT2-i, sodium glucose co-transporter 2 inhibitors; LMV, letermovir; MBV, maribavir; PEA, peanut allergen; EC50, half-maximal effective concentration; Rmd, remdesivir; GS, GS‐441524; PE, preeclampsia; OP, oseltamivir phosphate; OC, oseltamivir carboxylate, active metabolite of oseltamivir phosphate; sFlt-1, soluble fms-like tyrosine kinase-1; sEng, soluble endoglin; p-eNOS, endothelial nitric oxide synthase phosphorylation; CZP, certolizumab pegol; PDE1, phosphodiesterase-1; PDE5, phosphodiesterase-5; iNOS, inducible nitric oxide synthase; LLOQ, lower limit of quantification; NA, not applicable.
In addition, ex vivo placental perfusion models have emerged as pivotal tools for evaluating nanocarrier transplacental behavior, with recent studies demonstrating that targeted lipid nanoparticles (LNPs) remain largely confined to maternal circulation, exhibiting minimal fetal transfer, thereby suggesting their utility in preventing unintended drug passage across the placental barrier (ref. van Kammen et al., 2024). Complementing these findings, a review of 16 perfusion studies further elucidates that nanoparticle (NP) transport efficiency is critically governed by physicochemical determinants, particularly sub-100-nm dimensions and surface modifications such as PEGylation, which collectively modulate placental uptake kinetics (ref. Aengenheister et al., 2021). These insights establish that rational nanocarrier design represents a promising strategy for blocking unintended fetal drug exposure.
Despite being non-invasive, ex vivo placental perfusion experiments are constrained by the limited availability of human placental tissue (ref. Parks, 2015) and the need to maintain strict physiological conditions (37 °C, fetal arterial pressure 30 mmHg–60 mmHg, 95% O2/5% CO2 (maternal), 95% N2/5% CO2 (fetal), oxygen consumption, glucose, lactate, and HCG indicators) (ref. Schneider, 2015). Furthermore, the intricate vascular network of the placenta makes it challenging to accurately replicate blood flow and material exchange ex vivo, resulting in a low success rate and limiting its application (ref. Mathiesen et al., 2010). Unfortunately, full-term placental extracts may not reflect the evolution of the placenta during pregnancy.
Future advancements should refine placental perfusion systems by implementing multi-cannula designs (e.g., 22 maternal cannulas) to replicate physiological oxygen gradients, which are confirmed by direct measurements to maintain the target median oxygen tension (∼50 mmHg) and reduce hypoxic heterogeneity in the intervillous space (ref. Schneider, 2015). Concurrently, preserving endothelial integrity requires real-time monitoring of trophoblastic vacuolization, which is a histopathological marker strongly correlated with fetal circuit leakage, combined with mechanical stress reduction via low-pressure perfusion (<60 mmHg) and albumin-supplemented media (ref. Maroun et al., 2014). Crucially, mitigating oxidative stress demands protocol modifications, such as pre-perfusion hypothermia (4 °C) and HO-1 inducers (e.g., hemin) to activate antioxidant pathways, pulsed reoxygenation to minimize ROS bursts, and antibiotic supplementation to prevent bacterial-induced inflammation (ref. Schneider, 2009). Moreover, the standardization of placental perfusion techniques should be accelerated to ensure data consistency across research centers, facilitate cross-study validation, and promote broader adoption (ref. Schneider et al., 2022). It is foreseeable that with continuous optimization, placental perfusion experiments will hold significant application in placental PK research.
4.2 Organ-on-a-chip
An organ-on-a-chip is an in vitro biomimetic microfluidic device that replicates the structure and function of human organs, typically at 10–6 to 10–4 times the size of the actual organ (ref. Shuler, 2017). It provides a model that closely simulates the in vivo cellular environment, enabling cells and tissues to grow and complete their life cycles. This device uses microchip technology to create microfluidic cell culture chambers, where live cells are organized according to the organ physiology. By reproducing multicellular structures, tissue interfaces, and vascular perfusion, it mimics in vivo organ function and cellular environments (Figure 3) (ref. Cavero et al., 2019). The basic organ-on-a-chip is fabricated using standard soft-lithography techniques, where a 10:1 mixture of polydimethylsiloxane (PDMS) substrate and curing agent is cast onto an SU-8 master mold to form a bilayer or multicavity microfluidic structure with precisely defined channels. A porous polyester track-etched membrane or polycarbonate membrane is then sandwiched between the microchannels as a biological barrier, which is subsequently assembled and selectively coated with extracellular matrix components to form a biomimetic placental barrier through plasma bonding.
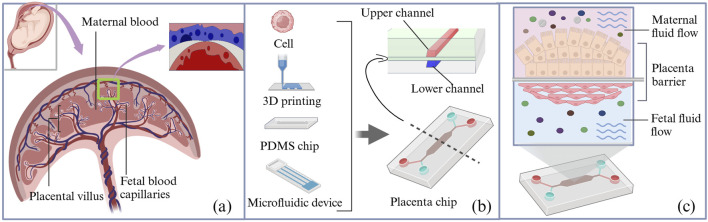
A comprehensive literature search revealed that, to date, placental drug transport has been evaluated and reported for only five pharmacological agents using the placenta-on-a-chip technology under controlled microphysiological conditions (Table 3), involving opioids, hypoglycemic agents, statins, and other drugs. Evidently, placenta-on-a-chip studies commonly use a core co-culture model of paired trophoblast cells (BeWo/STB) and endothelial cells (HUVECs/HPVECs) to replicate the maternal–fetal barrier. For statin (e.g., rosuvastatin)-specific studies, this basis can be extended to complex multicompartmental systems (3 layers–7 layers), which include fetal membrane cells (AECs, AMCs, and CTCs) and decidual cells (DCs) to mimic tissue interactions. Barrier integrity is generally verified by a tripartite protocol: immunostaining, FITC-dextran permeability assay, and morphological analysis, while some studies are supplemented with TEER measurements. Functionally, the simpler two-chamber design is sufficient for passive diffusion studies, whereas the ≥4-chamber configuration becomes crucial for drug metabolism studies, allowing important analyses of statin-derived metabolites and inflammatory cascades.
TABLE 3: Overview of reported organ-on-a-chip studies on placental drug transfer.
| Drug/compound | Dosage | Duration | Cell line (maternal) | Cell line (fetal) | Chambers | Barrier integrity and function verification | Key findings | Ref. |
|---|---|---|---|---|---|---|---|---|
| Naltrexone/6ß-naltrexol | 100 ng/mL | 8 h | BeWo cells | HUVECs | 2 | ImmunostainingFITC-dextran fluxMicrovilli imaging | FTR: 2.5% (naltrexone), 2.2% (6β-naltrexone) | ref. Pemathilaka et al. (2022) |
| Caffeine | 0.25 mg/mL | 7.5 h | BeWo cells | HUVECs | 2 | ImmunostainingFITC-dextran fluxMicrovilli imaging | FTR: 1.3% (at 5 h) | ref. Pemathilaka et al. (2019b) |
| Glyburide | 1 × 10−6 M | 7.5 h | STB | HPVECs | 2 | ImmunostainingTEER measurementFITC-dextran flux | FTR: 5.6%; clearance: BCRP-mediated exocytosis | ref. Blundell et al. (2018) |
| Pravastatin/rosuvastatin | 200 ng/mL | 24 h | DCs | Fetal membrane (AECs, AMCs, and CTCs); Placenta (BeWo, STB, and HUVECs) | 3,4 | ImmunostainingFITC-dextran fluxCell morphology and coverage checkCytotoxicity assayCytokine analysis | Metabolites produced; anti-inflammatory cytokines increased | ref. Richardson et al. (2022) |
| Pravastatin | 200 ng/mL | 24 h | DCs, STB, BeWo | CTCs, AMCs, AECs, and HUVECs | 7 | ImmunostainingFITC-dextran fluxCell morphology and coverage checkCell viability assay | FTR: 18% | ref. Kammala et al. (2023) |
| Rosuvastatin | 200 ng/mL | BeWo cell | AECs, AMCs, and CTCs | 4 | ImmunohistochemistryFlow cytometry | OATP2B1: mediate transplacental transport | ref. Ganguly et al. (2021) |
Abbreviations: HUVECs, human umbilical vein endothelial cell lines; TEER, trans-epithelial electrical resistance; HPVECs, human placental villous endothelial cells; FTR: fetal-to-maternal ratio; BCRP, breast cancer resistance protein; AECs, amnion epithelial cells; AMCs, amnion mesenchymal cells; CTCs, chorionic trophoblast cells; DCs, decidual cells; STB, syncytiotrophoblasts, BeWo + forskolin.
The development of three-dimensional (3D) placenta-on-a-chip models has been accelerated by the availability of microfluidic devices, commercial chips, and 3D bioprinting technologies. For instance, HUVECs and placental BeWo cells were co-cultured (1:3) for the first time to form spheroids (microtissues) on a 3D Petri Dish® mold, which mimicked in vivo responses more closely than 2D systems, in three main ways (ref. Be et al., 2025): spontaneous self-organization into a vascularized histoarchitecture with HUVECs peripherally enveloping a BeWo core, mimicking placental villi (ref. Lupattelli et al., 2014); enhanced intercellular junction formation (E-cadherin/ZO-1) and cytoskeletal reorganization yielding physiologically relevant TEER values (ref. Subramanian et al., 2023); superior functional maturation evidenced by elevated β-hCG secretion and drug metabolic capacity that more accurately predicted in vivo pharmacological responses (ref. Öztürk et al., 2024). Cao et al. developed a biomimetic placental barrier model that incorporated glucose transport and hCG secretion (ref. Cao et al., 2023). Another study developed a second-trimester (14 W–28 W) placental model (ref. Vidal et al., 2024) that offered valuable insights into placental drug transfer. These methodological advancements, when successfully implemented in transplacental drug studies, are anticipated to significantly enhance the quality of evidence regarding fetal drug exposure in future research endeavors.
The placenta-on-a-chip technology offers significant advantages over traditional trophoblast monolayer models by recreating the multilayer structure of the placental barrier and the hemodynamic environment (ref. Zambuto et al., 2024). However, the model has several limitations. First, current microarrays struggle to replicate complex overall physiological environments, which hamper their ability to model systemic diseases. Second, current placenta-on-a-chip models are a poor representation of the human placental villous structure. Another limitation is that as the complexity of 3D co-culture models increases, it becomes more challenging to perform high-resolution screening and accurately localize the tissues (ref. Bhatia and Ingber, 2014). Future placenta-on-a-chip development must, therefore, improve both physiological fidelity and microscale spatial resolution (ref. Pemathilaka et al., 2019a; ref. Esch et al., 2015), while incorporating gestational changes (ref. Blundell et al., 2018). Critical advancements must target the following (ref. Be et al., 2025): multi-lineage co-culture optimization (trophoblast/endothelial/stromal cell interactions) to maintain tissue-relevant cellular diversity (ref. Lupattelli et al., 2014); sub-10 μm spatial resolution achievement through nanopatterned surface functionalization (ref. Subramanian et al., 2023); hierarchical biomaterial development integrating decellularized ECM and precision-bioprinted villous structures; (ref. Phillips et al., 2024) programmable microenvironment platforms with feedback-controlled gas/endocrine factor delivery, enabling phase-transition modeling between gestational periods. Following significant advancements in organ-on-a-chip technology, subsequent research efforts should prioritize the implementation of these systems for longitudinal exposure studies and polypharmacy interaction analyses that are specifically designed to characterize the teratogenic consequences of sustained drug administration during critical periods of fetal development.
5 In silico simulation for placental transfer
Animal studies remain difficult to interpret due to structural and functional inter-species placental differences. The ex vivo perfusion of the human placental cotyledon is the method of reference for studying the human placental transfer of drugs because it is thought to mimic the functional placental tissue. However, it is challenged by the constraints of limited placental tissue and low experimental success rates. Recently, in silico techniques have further been advanced as complementary tools to validate experimental placental transfer data, offering a promising alternative for high-throughput screening of potential fetotoxicity at the early stages of drug design. PBPK models are promising in silico simulation tools that provide valuable insights into drug PK, particularly those that are difficult to assess in vivo. Several other methods have also been used to investigate placental PK.
5.1 Physiologically based pharmacokinetics (PBPK)
The PBPK model uses mathematical simulations to describe the absorption, distribution, metabolism, and excretion (ADME) of drugs by integrating the anatomical, physiological, and drug-specific data with preclinical and clinical information to simulate PK (ref. Dallmann et al., 2018). When applied to special populations, such as pregnant women and neonates, PBPK models incorporate organ development and individual variations. The key components of PBPK model development are shown in Figure 4a. The general workflow for developing a maternal–fetal PBPK model is shown (Figure 4c). First, a non-pregnant PBPK model was established and validated by comparison with the observed in vivo data. Once the non-pregnant PBPK model accurately captured PK, all drug-specific parameters were fixed. Pregnancy-specific changes [e.g., increase in blood volume, changes in metabolism, increased cardiac output, lower hypertension, increased renal blood flow, and variation with gestational week (ref. Zhang et al., 2017; ref. Zhang and Unadkat, 2017)] were then incorporated to extend the model to a pregnancy-specific version (ref. Dallmann et al., 2017), which involved adding pregnancy-specific compartments to the 27-compartment model (Figure 4b). The maternal–fetal PBPK (M–F PBPK) model is linked via a placental permeability-limited model subdivided into maternal blood, placental tissue, and fetal blood compartments (ref. Zhang and Unadkat, 2017). Transplacental clearance (ref. Yan et al., 2024) and other key parameters were assessed using various methods (Figure 4d) and were included in the final formation of the M–F PBPK models. In addition, depending on the purpose of the study, researchers may selectively incorporate placental drug metabolism/efflux transporters into the model.
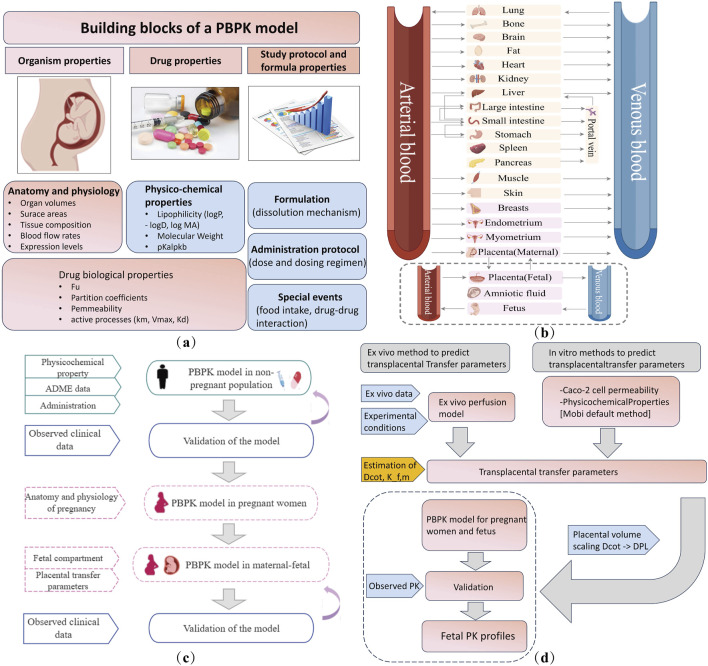
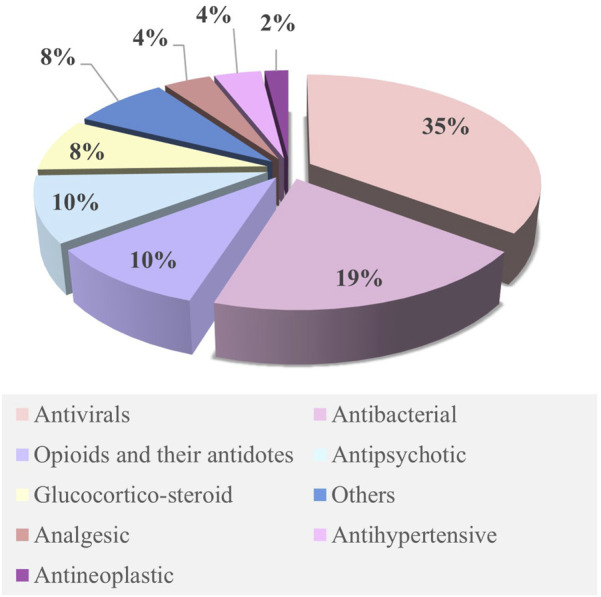
The M–F PBPK model was developed in 2015 (ref. Dallmann et al., 2018) and has rapidly advanced since 2020. Some research groups have extensively worked on PBPK models to predict fetal drug exposure and showed very encouraging results. In Table 4, we summarize the developed M–F PBPK models encompassing 42 pharmacological agents from 39 studies, including antiviral agents, antibacterial, opioids, antipsychotics, hormones, analgesics, anti-hypertensives, and antineoplastic agents. The dominance of antivirals (39%) in all M–F PBPK studies is highlighted in Figure 5, which shows that the use of antiviral drugs in the treatment of HIV/AIDS and hepatitis B is a global concern. During our literature search, we found several interesting research areas, including PBPK models that focused on predicting maternal drug concentrations and fetal environmental toxin exposure. However, these topics were not the focus of this study and have not been discussed further.
TABLE 4: Overview of reported fetal drug exposure studies for M–F PBPK (categorized by the pharmacological effect).
| Compound | Administration | Software | Placental permeability | No. of compartments | Placenta compartments | FetalCompartments | Validation | Clinical translation | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Analgesic | |||||||||
| Acetaminophen | po | PK-Sim/MoBi | Ex vivo perfusion model, Caco-2 assay, and physicochemical properties | 28 | P-m and P-f | AB, VB, AF, fetal liver, and RoB | MP, UCP | No dosage adjustment required | ref. Mian et al. (2020) |
| Indomethacin | po | Simcyp | Published data | 34 | P-m and P-f | Full fetal PBPK model | MP, UCP | Fetal exposure risks elevated in third trimesters | ref. Pillai et al. (2022) |
| Antibacterial | |||||||||
| Cefazolin | iv | GastroPlus | Parameter identification | NA | P-m and P-f | AB, VB, AF, and fetal body | MP, UCP, and AF | NA | ref. Szeto et al. (2021) |
| Cefuroxime | iv. or im | GastroPlus | Parameter identification | NA | P-m and P-f | AB, VB, AF, and fetal body | MP, UCP, AF | NA | ref. Szeto et al. (2021) |
| Cefazolin, Cefuroxime, Amoxicillin | iv. or im. or iv. bolus | Simcyp V 20 | Published data | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP, and AF | No dosage adjustment required | ref. Abduljalil et al. (2022a) |
| Ampicillin | iv | Simcyp V 21 | Physicochemical properties | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP | No dosage adjustment required | ref. Li and Xie (2023) |
| Cefuroxime | iv | PK-Sim/MoBi | Parameter identification | 41 | P-m, P-f | AB, VB, AF, brain, fat tissue, heart, liver, lungs, kidneys, gastrointestinal tract, muscles, and RoB | MP, UCP, AF, heart, liver, and lungs | NA | ref. Liu et al. (2024) |
| Vancomycin | iv | Simcyp | Ex vivo perfusion model, QSPR model, and Caco-2 assay | 35 | P-m, P-t, P-f | Full fetal PBPK model | MP, UCP | NA | ref. Yan et al. (2024) |
| Ceftazidime | iv. or im | Simcyp V 23 | Physicochemical properties | 35 | P-m, P-t, P-f | Full fetal PBPK model | MP, UCP, and AF | NA | ref. Abduljalil et al. (2024) |
| Metronidazole | Iv. or po | GastroPlus | Published data | NA | Not reported | Not reported | MP, UCP | NA | ref. Le Merdy et al. (2024) |
| Antipsychotic | |||||||||
| Oxcarbazepine | po | PK-Sim/MoBi | Ex vivo perfusion model, Caco-2 assay, physicochemical properties | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP | Dose ≥1.5× baseline in 2nd/3rd trimesters | ref. He et al. (2022) |
| Levetiracetam | iv. or po | PK-Sim/MoBi | Physicochemical properties | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP | Dose: 1.2×, 1.6×, 1.5× baseline in 1st/2nd/3rd trimesters | ref. Chen et al. (2023) |
| Sertraline | po | R Studio | Published data | 15 | P-m, Pf | AF, fetal blood, gut, liver, lung, and RoB | MP, UCP | NA | ref. Lartey et al. (2023) |
| Midazolam | iv. or po | GastroPlus | Published data | NA | Not Reported | Not reported | MP, UCP | NA | ref. Le Merdy et al. (2024) |
| Carbamazepine | po | PK-Sim/Mobi | Ex vivo perfusion model, Caco-2 assay, and physicochemical properties | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP | Dose: 1.4×, 1.8×, 2.1× baseline in 1st/2nd/3rd trimesters | ref. Chen et al. (2024) |
| Antivirals | |||||||||
| Darunavir | po | Berkeley Madonna/Simcyp | Ex vivo perfusion model | 13 | Placenta | AF, fetal blood, and fetal body | MP, UCP, AF | No dosage adjustment required | ref. Schalkwijk et al. (2018) |
| Dolutegravir | po | Berkeley Madonna/Simcyp | Ex vivo perfusion model | 13 | Placenta | AF, fetal blood, and fetal body | MP, UCP, AF | No dosage adjustment required | ref. Freriksen et al. (2020) |
| Dolutegravir | po | PK-Sim/MoBi | Published data | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP, NP | NA | ref. Liu et al. (2021),ref. Liu et al. (2020a) |
| Dolutegravir | po | MATLAB | Published data | 19 | Placenta | AB, VB, AF, brain, kidney, liver, and RoB | MP, NP | No dosage adjustment required | ref. Shenkoya et al. (2021) |
| Efavirenz | po | MATLAB | Published data | 19 | Placenta | AB, VB, AF, brain, kidney, liver, and RoB | MP, NP | NA | ref. Shenkoya et al. (2021),ref. Atoyebi et al. (2019) |
| Emtricitabine | iv. or po | Simcyp/R | Ex vivo perfusion model | 20 | P-m, P-f | AF, fetal blood, and fetal body | MP, UCP | NA | ref. De Sousa Mendes et al. (2016) |
| Emtricitabine | po | PK-Sim/MoBi | Ex vivo perfusion model | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP, NP | NA | ref. Liu et al. (2020b) |
| Acyclovir | iv. or po | PK-Sim/MoBi | Parameter identification | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP | NA | ref. Liu et al. (2020b) |
| Nevirapine | iv | R | Ex vivo perfusion model | 20 | P-m, P-f | AF, fetal blood, and fetal body | MP, UCP | No dosage adjustment required | ref. De Sousa Mendes et al. (2017) |
| Raltegravir | po | PK-Sim/MoBi | Ex vivo perfusion model | 27 | P-m, P-f | AB, VB, AF, and fetal body | MP, UCP, NP | NA | ref. Liu et al. (2020a) |
| Tenofovir | iv. or po | Simcyp/R | Ex vivo perfusion model | 20 | P-m, P-f | AF, fetal blood, and fetal body | MP, UCP, AF | NA | ref. De Sousa Mendes et al. (2016) |
| Zidovudine | iv. or po | MATLAB/Simcyp | Published data | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | NA | ref. Zhang et al. (2017),ref. Zhang and Unadkat (2017) |
| Acyclovir, Emtricitabine, Lamivudine | iv. or po | Simcyp V 20 | Published data | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP, and AF | NA | ref. Abduljalil et al. (2022b) |
| Doravirine | iv. or po | Simcyp | Ex vivo perfusion model | 31 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP | Increase in dosage, unspecified | ref. Bukkems et al. (2022) |
| Cabotegravir, Rilpivirine | im | SimBiology/MATLAB | Caco-2 assay, physicochemical properties | 30 | P-m, P-f | AB, VB, and fetal body | MP, UCP | Cabotegravir: no dosage adjustment required; rilpivirine: increase in dosage, unspecified | ref. Atoyebi et al. (2024) |
| Dolutegravir | po | Simcyp | Ex vivo perfusion model | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | No dosage adjustment required | ref. Ning et al. (2024) |
| Antihypertensive | |||||||||
| Nifedipine | oral | Simcyp V 22 | Caco-2 assay | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP, AF | Increase in dosage, unspecified | ref. Werdan Romão et al. (2024) |
| Metoprolol | iv. or po | GastroPlus | Published data | NA | Not Reported | Not Reported | MP, UCP | NA | ref. Le Merdy et al. (2024) |
| Antineoplastic | |||||||||
| Crizotinib | po | Simcyp/MATLAB | PBMP model | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP, and placenta tissue | NA | ref. Roelofsen et al. (2022) |
| Hormones | |||||||||
| BET, DEX | im | Simcyp/MATLAB | Caco-2 assay | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | NA | ref. Anoshchenko et al. (2021) |
| BET, DEX | iv. or im. or po | Simcyp/MATLAB | Ex vivo perfusion model, PBMP model | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | Reduce dose, increased frequency | ref. Van Der Heijden et al. (2024) |
| Opioids | |||||||||
| Fentanyl | iv. or epid | MATLAB/Simulink | Caco-2 assay | 21 | P-m, P-t, and P-f | AB, VB, brain, gut, kidney, liver, and RoB | MP, UCP | NA | ref. Shum et al. (2021) |
| Methadone | po | Simcyp | Ex vivo perfusion model | 35 | P-m, P-t, P-f | Full fetal PBPK model | MP, UCP | Dose adjustment not available | ref. Badhan and Gittins (2021) |
| THC | iv. or inh | Simcyp | Physicochemical properties | 12 | P-m, P-t, P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | Dose adjustment not available | ref. Patilea-Vrana and Unadkat (2021) |
| Buprenorphine | sl | Simcyp V 21 | Physicochemical properties | 35 | P-m, P-t, P-f | Full fetal PBPK model | MP, UCP | NA | ref. van Hoogdalem et al. (2024) |
| Naltrexone | iv. or im | Pumas | Fick’s diffusion equation, Caco-2 assay | 12 | P-m, P-f | Fetal body | MP | Dose adjustment not available | ref. Shenkoya et al. (2024) |
| Others | |||||||||
| Theophylline | iv. or po | MATLAB/Simcyp | Caco-2 assay, published data | 12 | P-m, P-t, and P-f | AB, VB, AF, brain, gut, kidney, liver, and RoB | MP, UCP | NA | ref. Zhang et al. (2017),ref. Zhang and Unadkat (2017) |
| Metformin | po | Simcyp V 20 | Published data | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP, AF | NA | ref. Abduljalil et al. (2022b) |
| Theophylline | iv. or po | Simcyp V 21 | Published data | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP | NA | ref. Abduljalil et al. (2022c) |
| Sildenafil | iv. or po | Simcyp V 21 | Ex vivo perfusion model | 35 | P-m, P-t, and P-f | Full fetal PBPK model | MP, UCP | Dose adjustment not available | ref. Macente et al. (2023) |
Abbreviations: po., oral; iv., intravenous; im., intramuscular; inh., inhalation; epid., epidural; sl., sublingual; P-m, placenta (maternal); P-f, placenta (fetal); P-t, placenta (tissue); AB, arterial blood; VB, venous blood; MP, maternal plasma; UCP, umbilical cord plasma; AF, amniotic fluid; NP, neonatal plasma (around birth); QSPR, quantitative structure–property relationship model; PBMP, physiologically-based semi-mechanistic placenta; RoB, rest of body; THC: cannabinoid (−)-Δ9-tetrahydro-cannabinol; full fetal PBPK mode including adipose, bone, brain, gut, heart, kidney, liver, lungs, pancreas, muscle, skin, spleen, amniotic fluid, blood; BET, betamethasone; DEX, dexamethasone; NA, not applicable.
Although PBPK is a physiological PK simulation, it still relies on mathematical and mechanistic modeling. The goal of PBPK model development is to accurately reflect the in vivo physiological conditions. Studies have incorporated the fetal liver as an independent metabolic organ (ref. Mian et al., 2020), considering the scaling of fetal drug-unbound fractions (ref. Roelofsen et al., 2022) and adjusted parameters, such as fetal liver and kidney volumes and cytochrome P450 enzyme levels, to model fetal drug metabolism and clearance (ref. Szeto et al., 2021). These efforts were aimed at improving the precision of fetal drug concentration predictions. Moreover, given the dynamic development of the placenta (ref. Aplin et al., 2020) and the challenges in obtaining early pregnancy samples, future research should prioritize drug safety studies during early and mid-pregnancy to better guide maternal medication use. Further exploration of these approaches is crucial for future research as it will provide valuable evidence for clinical decision-making.
5.2 Computational approaches
Diverting from mechanistic approaches, novel mathematical techniques such as artificial intelligence and machine learning (ML) methods ranging from regression analysis to deep learning and neural networks have recently been utilized for the investigation of placental PK. One study used ML to stratify high-risk pregnant women based on biomarkers and electronic health data, providing insights into maternal vaccination and placental antibody transfer for personalized immunization (ref. Wessel and Dolatshahi, 2024). Another study explored the placental transport of toxic substances using a uterine perfusion model and molecular docking (ref. Zhang et al., 2024). Additionally, a quantitative structure–property relationship (QSPR) model optimized using Monte Carlo methods was developed for high-throughput screening of placental drug permeability (ref. Vukomanović et al., 2024). Similarly, researchers have used eigenvalue analysis, a chiral-sensitive QSPR method combined with ML, to predict placental drug transfer, focusing on chirality and stereochemistry (ref. Gomatam and Coutinho, 2024). These efforts highlight the growing role of computational approaches in advancing our understanding of placental PK and enhancing drug safety during pregnancy. Although the application of these techniques for the prediction of placental drug transfer is in its infancy, they are slowly gaining traction as powerful PK tools. However, it is apparent that novel mathematical techniques such as these have great potential to fill the knowledge gap in underrepresented populations and clinical scenarios, and future research initiatives should encompass these techniques.
6 Discussion
Placental pharmacokinetics refers to the study of how drugs are absorbed, distributed, metabolized, and excreted as they pass from the maternal to the fetal circulation via the placenta. As a selective barrier and active transport interface, the placenta plays a pivotal role in determining fetal drug exposure. Understanding placental pharmacokinetics is essential for optimizing medication use during pregnancy, ensuring maternal efficacy while minimizing fetal risk. To address the complexity of maternal–fetal drug transfer, a range of experimental and computational models have been developed to investigate the placental transport mechanisms and predict fetal exposure. Recent advances in ex vivo placental perfusion, organ-on-a-chip platforms, and in silico simulations offer promising avenues to improve the accuracy of fetal drug-exposure predictions.
6.1 Placental pharmacokinetic models: characteristics and applications
Current methods for placental pharmacokinetics include cell models, organ models, and in silico-guided models, each with its own distinct characteristics (Table 5). Monolayer cell models are convenient and rapid, but they oversimplify placental dynamics (ref. Birch et al., 2018). Ex vivo and organ-on-chip systems offer greater physiological relevance, yet they face practical constraints (ref. Pemathilaka et al., 2019a). In silico models, such as PBPK, allow quantitative predictions but rely on experimental data, highlighting the need for integrated approaches. A review of current studies shows that monolayer cell models are used to evaluate placental drug permeability and transporter-mediated transfer (ref. Mian et al., 2020; ref. Po et al., 2018), ex vivo and organ-on-a-chip systems quantify fetal drug exposure and pharmacokinetics (ref. Freriksen et al., 2020; ref. Zhang and Unadkat, 2017), and PBPK models support clinical dosing during pregnancy while predicting fetal drug exposure (ref. Schneider et al., 2022; ref. Abduljalil et al., 2022b).
TABLE 5: Summary of the characteristics of the various methods.
| Methods | Advantages | Limitations | Applicable boundaries |
|---|---|---|---|
| Caco-2 cell model | Simple, cost-effective, and high-throughput screening | Lacks full placental physiology | Early drug permeability prediction |
| Trophoblast cell lines | Simple, cost-effective, and high-throughput screening | Monolayer cells differ from actual placental barrier | Study of placental transporter proteins |
| Placental cotyledon perfusion experiment | Retains full placental structure and function | Limited donors and demanding experimental conditions | Drug transport kinetics and metabolism studies |
| Organ-on-a-chip | Simulates dynamic placental environment, integrates multiple cell types | Technology lacks standardization, long-term stability issues | Placental drug transport, maternal–fetal barrier function, and nanomedicine evaluation |
| PBPK model | Integrates physiological parameters for PK | Relies on experimental data and needs clinical validation | Prediction of fetal drug-exposure during pregnancy |
Based on the comparative evaluation of each method, model selection in practical research should be guided by the research objective, the required physiological relevance, and the available resources. Simplified in vitro systems, such as cell monolayers or co-cultures, are appropriate for high-throughput screening or preliminary mechanistic studies because of their reproducibility and efficiency. Studies that demand physiologically relevant transport dynamics benefit from ex vivo placental perfusion or organ-on-a-chip models, which preserve tissue architecture and flow conditions absent in conventional in vitro systems. Computational approaches, including PBPK modeling, can supplement experimental data by providing quantitative predictions across different scenarios and species. Careful alignment of model choice with study goals and experimental constraints enhances both the reliability and translational value.
6.2 Multi-model integration: strengthening predictive power and translational relevance
The integration of multiple models holds significant potential in enhancing predictive accuracy and individual risk assessment. For instance, multiple studies demonstrate that incorporating ex vivo placental perfusion parameters into PBPK modeling yields predictions within 2-fold of clinical observations, significantly improving fetal drug-exposure estimates compared to non-integrated models (ref. Mian et al., 2020; ref. Roelofsen et al., 2022; ref. Schalkwijk et al., 2018; ref. Van Der Heijden et al., 2024). Another study showed that incorporating BeWo cell transporter kinetics and placental transporter abundance (particularly efflux transporters) into PBPK models can improve the physiological relevance of fetal drug-exposure predictions, as evidenced by the maternal-to-fetal plasma ratio shifting from approximately 1.0 to 0.1 (ref. Al-Majdoub et al., 2025). In addition, integrating omics data (transcriptomics and proteomics) with dynamic placenta-on-a-chip platforms could enhance our understanding of drug–nutrient interactions, transporter regulation, and placental metabolism. The synergy between these methods facilitates a more accurate representation of the complex physiological processes involved in drug transfer, metabolism, and response, ultimately paving the way for tailored, safer treatments, especially in sensitive populations such as pregnant women. Beyond studies focused solely on placental drug-transfer, modeling approaches developed in this domain may offer significant insights into fetal drug-exposure arising from maternal vaccination, prolonged pharmacotherapy, or environmental toxin exposure during pregnancy.
Interestingly, the integration of data-driven approaches with mechanistic PBPK models also represents a highly promising strategy in PK research, particularly for pregnant populations. ML and QSPR methods can estimate parameters that are difficult to obtain experimentally; these parameters can be incorporated into PBPK models to enhance physiological realism and predictive accuracy. For example, a hybrid deep learning-PBPK model has successfully predicted human PK profiles from chemical structures, generating reliable concentration–time curves (ref. Li et al., 2024). AI-assisted PBPK models have also been applied in the delivery of nanomedicines, producing predictions that closely match experimental PK data (ref. Chou et al., 2023). These studies collectively demonstrate that utilizing ML or QSPR to inform mechanistic models can strengthen model development, optimize predictive performance, and enhance translational relevance. The application of such integrative strategies to placental pharmacokinetics holds considerable promise for advancing the field.
These technologies can help clarify how drugs cross the placenta and guide safer treatment for pregnant women with conditions such as epilepsy, hypertension, or HIV, where treatment is often unavoidable. The valuable results obtained from these methods could not only support drug label modifications but also inform the development of evidence-based treatment guidelines. Placental perfusion studies integrated with PBPK modeling reveal that doravirine exhibits a 55% reduction in maternal AUC during late pregnancy (40 weeks) (ref. Bukkems et al., 2022), elevating treatment failure risk while predicting substantial fetal exposure. These findings have prompted multiple guideline bodies to re-evaluate its gestational use, shifting from “insufficient data, not recommended” toward “use with monitoring.” Similarly, quantified placental transfer kinetics of dexamethasone/betamethasone have enabled the precise prediction of maternal–fetal exposure dynamics (ref. Van Der Heijden et al., 2024), advancing guidelines from fixed-dose to gestational age-adjusted regimens for optimal risk–benefit balance. In addition, a study combining Caco-2 cell models and placental perfusion experiments with PBPK modeling predicted that arterial umbilical concentrations of acetaminophen remain well below the recommended postnatal ductus arteriosus closure threshold (24.47 mg/L) (ref. Mian et al., 2020). Regulatory agencies such as the Food and Drug Administration (FDA) and European Medicines Agency (EMA) increasingly accept PBPK modeling and may require placental transport assessment for drugs used in pregnancy in the future.
6.3 Future perspectives: gestational age and pathophysiology considerations
We have previously emphasized the need to investigate dynamic changes in the placenta during pregnancy, which are mainly driven by alterations in blood volume, blood flow velocity, tissue distribution, and the expression of metabolic enzymes and transporters. Nevertheless, few studies have specifically addressed this aspect, thus requiring greater attention in future research. Reviewing existing methodologies and models, several potential strategies may address this challenge, including (ref. Be et al., 2025) conducting ex vivo placental perfusion using tissue samples from different gestational stages (early, mid, and late pregnancy) under ethically permissible conditions (ref. Goeden and Bonnin, 2013); (ref. Lupattelli et al., 2014) inducing pluripotent stem cells (iPSCs) to differentiate into trophoblasts, simulating developmental states across pregnancy stages (early trophoblasts vs. mature STBs) (ref. Horii et al., 2016); (ref. Subramanian et al., 2023) placenta-on-a-chip platforms, simulating the endocrine environment of different gestational stages by regulating key hormone levels (progesterone, estrogen, and hCG) in culture media (ref. Mathiesen et al., 2025; ref. Shojaei et al., 2021; ref. Phillips et al., 2024). PBPK modeling can incorporate time-dependent parameters such as placental blood flow and transporter expression profiles to construct precise drug-use models for different gestational periods (ref. Zhang et al., 2017) (ref. Zhang and Unadkat, 2017). Furthermore, integrating in vitro data from different gestational stages with PBPK simulations offers a promising strategy to account for pregnancy-related variability.
In addition to gestational age-related dynamic changes, various pathological conditions can also markedly alter the placental structure and function, thereby influencing drug transfer. Preeclampsia [reduced placental perfusion and abnormal spiral artery remodeling (ref. Roberts and Escudero, 2012; ref. Yagel et al., 2022; ref. Harmon et al., 2016)], gestational diabetes mellitus [hyperglycemia-induced upregulation of inflammatory mediators and altered transporter expression (ref. Calvo et al., 2024)], and intrauterine infection [enhanced inflammatory response and increased tissue permeability (ref. Meg et al., 2022)] are among the most common pregnancy complications associated with changes in villous stroma and vascular architecture. These pathological alterations can further modify the expression and activity of transporters and metabolic enzymes, ultimately affecting maternal–fetal drug transfer. To reproduce these pathological phenotypes in vitro or through modeling, several feasible approaches have been explored. For instance, ex vivo placental perfusion using tissues from pathological pregnancies [e.g., preeclampsia or GDM (ref. Villota et al., 2021)] allows direct measurement of permeability differences under disease conditions. In cell-based and organ-on-a-chip models, preeclampsia, infection, or hyperglycemia can be mimicked by inducing hypoxia, adding pro-inflammatory cytokines (such as TNF-α or IL-6), or applying high-glucose culture conditions (ref. Elzinga et al., 2023). Moreover, chip platforms can be adapted to modify shear stress, perfusion rates, or introduce pathological flow patterns to reflect abnormal spiral artery remodeling and reduced perfusion (ref. Mosavati et al., 2020). Together, incorporating both pathophysiological states and gestational dynamics into placental models would greatly enhance their translational relevance for predicting drug disposition during pregnancy.
7 Conclusion
In this review, we describe the cell monolayer model, ex vivo placental perfusion experiments, placental-on-a-chip, PBPK model, and other in vitro research methods and discuss their advantages and disadvantages. The approach of a multi-model data fusion strategy (e.g., integrating a placenta perfusion experiment and PBPK modeling) is promising for the investigation of drug pharmacokinetics during pregnancy early in drug development, but further standardization and extension is needed. As technological fidelity improves and validation expands, placental drug modeling may become an essential, not optional, component of maternal–fetal pharmacokinetics.
References
- K. Abduljalil, J. Ning, A. Pansari, X. Pan, M. Jamei. Prediction of maternal and fetoplacental concentrations of cefazolin, cefuroxime, and amoxicillin during pregnancy using Bottom-Up physiologically based pharmacokinetic models.. Drug Metab. Dispos., 2022a. [DOI | PubMed]
- K. Abduljalil, A. Pansari, J. Ning, M. Jamei. Prediction of maternal and fetal Acyclovir, emtricitabine, lamivudine, and metformin concentrations during pregnancy using a physiologically based pharmacokinetic modeling approach.. Clin. Pharmacokinet., 2022b. [DOI | PubMed]
- K. Abduljalil, I. Gardner, M. Jamei. Application of a physiologically based pharmacokinetic approach to predict theophylline pharmacokinetics using virtual Non-Pregnant, pregnant, fetal, Breast-Feeding, and neonatal populations.. Front. Pediatr., 2022c. [DOI | PubMed]
- K. Abduljalil, I. Gardner, M. Jamei. An application of a physiologically based pharmacokinetic approach to predict ceftazidime pharmacokinetics in a pregnant population.. Pharmaceutics, 2024. [DOI | PubMed]
- N. Abolhassani, U. Winterfeld, Y. C. Kaplan, C. Jaques, B. Minder Wyssmann, G. C. Del. Major malformations risk following early pregnancy exposure to metformin: a systematic review and meta-analysis.. BMJ Open Diabetes Res. Care, 2023. [DOI | PubMed]
- L. Aengenheister, R. R. Favaro, D. M. Morales-Prieto, L. A. Furer, M. Gruber, C. Wadsack. Research on nanoparticles in human perfused placenta: state of the art and perspectives.. Placenta, 2021. [DOI | PubMed]
- Z. M. Al-Majdoub, J. J. M. Freriksen, A. Colbers, J. van den Heuvel, J. Koenderink, K. Abduljalil. Absolute membrane protein abundance of P-glycoprotein, breast cancer resistance protein, and multidrug resistance proteins in term human placenta tissue and commonly used cell systems: application in physiologically based pharmacokinetic modeling of placental drug disposition.. Drug Metab. Dispos., 2025. [DOI | PubMed]
- O. Anoshchenko, M. A. Milad, J. D. Unadkat. Estimating fetal exposure to the P-gp substrates, corticosteroids, by PBPK modeling to inform prevention of neonatal respiratory distress syndrome.. CPT Pharmacometrics Syst. Pharmacol., 2021. [DOI | PubMed]
- J. D. Aplin, J. E. Myers, K. Timms, M. Westwood. Tracking placental development in health and disease.. Nat. Rev. Endocrinol., 2020. [DOI | PubMed]
- P. Artursson, K. Palm, K. Luthman. Caco-2 monolayers in experimental and theoretical predictions of drug transport.. Adv. Drug Deliv. Rev., 2001. [DOI | PubMed]
- S. A. Atoyebi, R. K. R. Rajoli, E. Adejuyigbe, A. Owen, O. Bolaji, M. Siccardi. Using mechanistic physiologically-based pharmacokinetic models to assess prenatal drug exposure: thalidomide versus efavirenz as case studies.. Eur. J. Pharm. Sci., 2019. [DOI | PubMed]
- S. Atoyebi, F. Bunglawala, N. Cottura, S. Grañana-Castillo, M. C. Montanha, A. Olagunju. Physiologically-based pharmacokinetic modelling of long-acting injectable cabotegravir and rilpivirine in pregnancy.. Br. J. Clin. Pharmacol., 2024. [DOI | PubMed]
- A. Balan, I. Szaingurten-Solodkin, S. S. Swissa, V. Feinshtein, M. Huleihel, G. Holcberg. The effects of pravastatin on the normal human placenta: lessons from ex-vivo models.. PLoS One, 2017. [DOI | PubMed]
- R. K. S. Badhan, R. Gittins. Precision dosing of methadone during pregnancy: a pharmacokinetics virtual clinical trials study.. J. Subst. Abuse Treat., 2021. [DOI | PubMed]
- M. R. Bai, D. L. Sun, H. D. Jiang, C. H. Zheng. Expression and regulation of drug transporters in placenta.. Yao Xue Xue Bao, 2016. [DOI | PubMed]
- P. Bapat, L. S. Pinto, A. Lubetsky, H. Berger, G. Koren. Rivaroxaban transfer across the dually perfused isolated human placental cotyledon.. Am. J. Obstet. Gynecol., 2015. [DOI | PubMed]
- Y. Belsti, A. Mousa, H. Jackson, L. J. Moran, K. R. Palmer, R. R. Dhungana. The use of multiple medications during pregnancy among an ethnically diverse population in South-Eastern Melbourne: a retrospective analysis to explore potential risks and complications.. Drug Saf., 2025. [DOI | PubMed]
- E. Berman, E. Kohn, M. Berkovitch, M. Kovo, S. Eyal. Lacosamide effects on placental carriers of essential compounds in comparison with valproate: studies in perfused human placentas.. Epilepsia, 2022. [DOI | PubMed]
- S. N. Bhatia, D. E. Ingber. Microfluidic organs-on-chips.. Nat. Biotechnol., 2014. [DOI | PubMed]
- D. Birch, R. G. Diedrichsen, P. C. Christophersen, H. Mu, H. M. Nielsen. Evaluation of drug permeation under fed state conditions using mucus-covered Caco-2 cell epithelium.. Eur. J. Pharm. Sci., 2018. [DOI | PubMed]
- C. Blundell, Y. S. Yi, L. Ma, E. R. Tess, M. J. Farrell, A. Georgescu. Placental Drug Transport-on-a-Chip: a microengineered in vitro model of transporter-mediated Drug efflux in the human placental barrier.. Adv. Healthc. Mater, 2018. [DOI | PubMed]
- M. Broekhuizen, R. de Vries, M. A. W. Smits, W. A. Dik, S. Schoenmakers, B. C. P. Koch. Pentoxifylline as a therapeutic option for pre-eclampsia: a study on its placental effects.. Br. J. Pharmacol., 2022. [DOI | PubMed]
- V. E. Bukkems, H. van Hove, D. Roelofsen, J. J. M. Freriksen, E. W. J. van Ewijk-Beneken Kolmer, D. M. Burger. Prediction of maternal and fetal doravirine exposure by integrating physiologically based pharmacokinetic modeling and human placenta perfusion experiments.. Clin. Pharmacokinet., 2022. [DOI | PubMed]
- M. J. Calvo, H. Parra, R. Santeliz, J. Bautista, E. Luzardo, N. Villasmil. The placental role in gestational diabetes mellitus: a molecular perspective.. touchREV Endocrinol., 2024. [DOI | PubMed]
- R. Cao, Y. Wang, J. Liu, L. Rong, J. Qin. Self-assembled human placental model from trophoblast stem cells in a dynamic organ-on-a-chip system.. Cell. Prolif., 2023. [DOI | PubMed]
- I. Cavero, J. M. Guillon, H. H. Holzgrefe. Human organotypic bioconstructs from organ-on-chip devices for human-predictive biological insights on drug candidates.. Expert Opin. Drug Saf., 2019. [DOI | PubMed]
- M. Ceckova, A. Libra, P. Pavek, P. Nachtigal, M. Brabec, R. Fuchs. Expression and functional activity of breast cancer resistance protein (BCRP, ABCG2) transporter in the human choriocarcinoma cell line BeWo.. Clin. Exp. Pharmacol. Physiol., 2006. [DOI | PubMed]
- L. Cerveny, Z. Ptackova, M. Ceckova, R. Karahoda, S. Karbanova, L. Jiraskova. Equilibrative nucleoside transporter 1 (ENT1, SLC29A1) facilitates transfer of the antiretroviral drug abacavir across the placenta.. Drug Metab. Dispos., 2018. [DOI | PubMed]
- J. Chen, X. You, W. Wu, G. Guo, R. Lin, M. Ke. Application of PBPK modeling in predicting maternal and fetal pharmacokinetics of levetiracetam during pregnancy.. Eur. J. Pharm. Sci., 2023. [DOI | PubMed]
- Y. Chen, M. Ke, W. Fang, Y. Jiang, R. Lin, W. Wu. Physiologically based pharmacokinetic modeling to predict maternal pharmacokinetics and fetal carbamazepine exposure during pregnancy.. Eur. J. Pharm. Sci., 2024. [DOI | PubMed]
- W. C. Chou, Q. Chen, L. Yuan, Y. H. Cheng, C. He, N. A. Monteiro-Riviere. An artificial intelligence-assisted physiologically-based pharmacokinetic model to predict nanoparticle delivery to tumors in mice.. J. Control Release, 2023. [DOI | PubMed]
- S. Correia Carreira, L. Cartwright, L. Mathiesen, L. E. Knudsen, M. Saunders. Studying placental transfer of highly purified non-dioxin-like PCBs in two models of the placental barrier.. Placenta, 2011. [DOI | PubMed]
- A. Dallmann, I. Ince, M. Meyer, S. Willmann, T. Eissing, G. Hempel. Gestation-Specific changes in the anatomy and physiology of healthy pregnant women: an extended repository of model parameters for physiologically based pharmacokinetic modeling in pregnancy.. Clin. Pharmacokinet., 2017. [DOI | PubMed]
- A. Dallmann, M. Pfister, J. van den Anker, T. Eissing. Physiologically based pharmacokinetic modeling in pregnancy: a systematic review of published models.. Clin. Pharmacol. Ther., 2018. [DOI | PubMed]
- K. M. De Felice, S. Kane. Safety of anti-TNF agents in pregnancy.. J. Allergy Clin. Immunol., 2021. [DOI | PubMed]
- M. De Sousa Mendes, D. Hirt, C. Vinot, E. Valade, G. Lui, C. Pressiat. Prediction of human fetal pharmacokinetics using ex vivo human placenta perfusion studies and physiologically based models.. Br. J. Clin. Pharmacol., 2016. [DOI | PubMed]
- M. De Sousa Mendes, G. Lui, Y. Zheng, C. Pressiat, D. Hirt, E. Valade. A physiologically-based pharmacokinetic model to predict human fetal exposure for a drug metabolized by several CYP450 pathways.. Clin. Pharmacokinet., 2017. [DOI | PubMed]
- E. Drwal, A. Rak, E. Gregoraszczuk. Co-culture of JEG-3, BeWo and syncBeWo cell lines with adrenal H295R cell line: an alternative model for examining endocrine and metabolic properties of the fetoplacental unit.. Cytotechnology, 2018. [DOI | PubMed]
- G. A. M. Eliesen, J. van Drongelen, H. van Hove, N. I. Kooijman, P. van den Broek, A. de Vries. Assessment of placental disposition of Infliximab and etanercept in women with autoimmune diseases and in the Ex Vivo perfused placenta.. Clin. Pharmacol. Ther., 2020. [DOI | PubMed]
- F. A. Elzinga, B. Khalili, D. J. Touw, J. R. Prins, P. Olinga, H. G. D. Leuvenink. Placenta-on-a-Chip as an in vitro approach to evaluate the physiological and structural characteristics of the human placental barrier upon drug exposure: a systematic review.. J. Clin. Med., 2023. [DOI | PubMed]
- E. W. Esch, A. Bahinski, D. Huh. Organs-on-chips at the frontiers of drug discovery.. Nat. Rev. Drug Discov., 2015. [DOI | PubMed]
- V. Faure Bardon, G. Peytavin, M. P. Lê, T. Guilleminot, E. Elefant, J. Stirnemann. Placental transfer of Letermovir and Maribavir in the ex vivo human cotyledon perfusion model. New perspectives for in utero treatment of congenital cytomegalovirus infection.. PLoS One, 2020. [DOI | PubMed]
- E. G. Foster, H. E. Gendelman, A. N. Bade. HIV-1 integrase strand transfer inhibitors and neurodevelopment.. Pharm. (Basel)., 2022. [DOI | PubMed]
- J. J. M. Freriksen, S. Schalkwijk, A. P. Colbers, K. Abduljalil, F. G. M. Russel, D. M. Burger. Assessment of maternal and fetal Dolutegravir exposure by integrating Ex Vivo placental perfusion data and physiologically-based pharmacokinetic modeling.. Clin. Pharmacol. Ther., 2020. [DOI | PubMed]
- B. Fuenzalida, V. Basler, N. Koechli, N. Yi, F. Staud, C. Albrecht. Modelling the maternal-fetal interface: an in vitro approach to investigate nutrient and drug transport across the human placenta.. J. Cell. Mol. Med., 2024. [DOI | PubMed]
- E. Ganguly, A. K. Kammala, M. Benson, L. S. Richardson, A. Han, R. Menon. Organic anion transporting polypeptide 2B1 in human fetal membranes: a novel gatekeeper for drug transport during pregnancy?. Front. Pharmacol., 2021. [DOI | PubMed]
- N. Goeden, A. Bonnin. Ex vivo perfusion of mid-to-late-gestation mouse placenta for maternal-fetal interaction studies during pregnancy.. Nat. Protoc., 2013. [DOI | PubMed]
- A. Gomatam, E. Coutinho. A chirality-sensitive approach to predict chemical transfer across the human placental barrier.. Toxicol. Lett., 2024. [DOI | PubMed]
- D. M. Haas, D. J. Marsh, D. T. Dang, C. B. Parker, D. A. Wing, H. N. Simhan. Prescription and other medication use in pregnancy.. Obstet. Gynecol., 2018. [DOI | PubMed]
- L. W. Han, C. Gao, Q. Mao. An update on expression and function of P-gp/ABCB1 and BCRP/ABCG2 in the placenta and fetus.. Expert Opin. Drug Metab. Toxicol., 2018. [DOI | PubMed]
- A. C. Harmon, D. C. Cornelius, L. M. Amaral, J. L. Faulkner, M. W. Cunningham, K. Wallace. The role of inflammation in the pathology of preeclampsia.. Clin. Sci. (Lond)., 2016. [DOI | PubMed]
- A. Hawkins, P. Pantazi, L. Yang, C. B. Coyne, V. Bokun, J. M. Lemme-Dumit. Long-term culture and passaging of term trophoblast for the investigation of syncytiotrophoblast function.. Placenta, 2025. [DOI | PubMed]
- L. He, M. Ke, W. Wu, J. Chen, G. Guo, R. Lin. Application of physiologically based pharmacokinetic modeling to predict maternal pharmacokinetics and fetal exposure to oxcarbazepine.. Pharmaceutics, 2022. [DOI | PubMed]
- B. Hirschmugl, C. Wadsack. Transplacental transfer of venlafaxine evaluated by ex vivo perfusion.. Placenta, 2022. [DOI | PubMed]
- E. Hitzerd, M. Broekhuizen, K. M. Mirabito Colafella, M. Glisic, R. de Vries, B. C. P. Koch. Placental effects and transfer of sildenafil in healthy and preeclamptic conditions.. EBioMedicine, 2019. [DOI | PubMed]
- M. Horii, Y. Li, A. K. Wakeland, D. P. Pizzo, K. K. Nelson, K. Sabatini. Human pluripotent stem cells as a model of trophoblast differentiation in both normal development and disease.. Proc. Natl. Acad. Sci. U. S. A., 2016. [DOI | PubMed]
- H. Huang, J. Wang, Q. Li, J. Duan, Q. Yao, Q. Zheng. Transplacental transfer of oseltamivir phosphate and its metabolite oseltamivir carboxylate using the ex vivo human placenta perfusion model in Chinese Hans population.. J. Matern. Fetal Neonatal Med., 2017. [DOI | PubMed]
- C. Jovelet, A. Seck, O. Mir, C. Simasotchi, S. Broutin, F. Goffinet. Variation in transplacental transfer of tyrosine kinase inhibitors in the human perfused cotyledon model.. Ann. Oncol., 2015. [DOI | PubMed]
- A. K. Kammala, R. C. V. Lintao, N. Vora, A. Mosebarger, K. Khanipov, G. Golovko. Expression of CYP450 enzymes in human fetal membranes and its implications in xenobiotic metabolism during pregnancy.. Life Sci., 2022. [DOI | PubMed]
- A. K. Kammala, L. S. Richardson, E. Radnaa, A. Han, R. Menon. Microfluidic technology and simulation models in studying pharmacokinetics during pregnancy.. Front. Pharmacol., 2023. [DOI | PubMed]
- R. Karahoda, S. Kallol, M. Groessl, E. Ontsouka, P. Anderle, C. Fluck. Revisiting steroidogenic pathways in the human placenta and primary human trophoblast cells.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- V. Karakis, J. W. Britt, M. Jabeen, A. San Miguel, B. M. Rao. Derivation of human trophoblast stem cells from placentas at birth.. J. Biol. Chem., 2025. [DOI | PubMed]
- S. Karbanova, L. Cerveny, M. Ceckova, Z. Ptackova, L. Jiraskova, S. Greenwood. Role of nucleoside transporters in transplacental pharmacokinetics of nucleoside reverse transcriptase inhibitors zidovudine and emtricitabine.. Placenta, 2017. [DOI | PubMed]
- G. Koren, A. Ornoy. The role of the placenta in drug transport and fetal drug exposure.. Expert Rev. Clin. Pharmacol., 2018. [DOI | PubMed]
- A. Kothari, B. Hirschmugl, J. S. Lee, B. Pfaller, K. Schmidthaler, Z. Szépfalusi. The impact of maternal-fetal omalizumab transfer on peanut-specific responses in an ex vivo placental perfusion model.. Allergy, 2022. [DOI | PubMed]
- S. Kuoni, R. Steiner, L. Saleh, R. Lehmann, N. Ochsenbein-Kölble, A. P. Simões-Wüst. Safety assessment of the SGLT2 inhibitors empagliflozin, dapagliflozin and canagliflozin during pregnancy: an ex vivo human placenta perfusion and in vitro study.. Biomed. Pharmacother., 2024. [DOI | PubMed]
- S. Lapehn, S. Nair, E. J. Firsick, J. MacDonald, C. Thoreson, J. A. Litch. A transcriptomic comparison of in vitro models of the human placenta.. Placenta, 2024. [DOI | PubMed]
- D. Lartey, D. Jateng, M. Li, C. Nguyen, V. Crentsil, J. Beitz. Quantification of sertraline maternal/fetal ratio and amniotic fluid concentration using a pregnancy physiologically based pharmacokinetic model.. Br. J. Clin. Pharmacol., 2023. [DOI | PubMed]
- M. Le Merdy, K. X. Szeto, J. Perrier, M. B. Bolger, V. Lukacova. PBPK modeling approach to predict the behavior of drugs cleared by metabolism in pregnant subjects and fetuses.. Pharmaceutics, 2024. [DOI | PubMed]
- L. Li, D. J. Schust. Isolation, purification and in vitro differentiation of cytotrophoblast cells from human term placenta.. Reprod. Biol. Endocrinol., 2015. [DOI | PubMed]
- S. Li, F. Xie. Foetal and neonatal exposure prediction and dosing evaluation for ampicillin using a physiologically-based pharmacokinetic modelling approach.. Br. J. Clin. Pharmacol., 2023. [DOI | PubMed]
- H. Li, B. van Ravenzwaay, I. M. Rietjens, J. Louisse. Assessment of an in vitro transport model using BeWo b30 cells to predict placental transfer of compounds.. Arch. Toxicol., 2013. [DOI | PubMed]
- Y. Li, Z. Wang, Y. Li, J. Du, X. Gao, Y. Li. A combination of machine learning and PBPK modeling approach for pharmacokinetics prediction of small molecules in humans.. Pharm. Res., 2024. [DOI | PubMed]
- X. I. Liu, J. D. Momper, N. Y. Rakhmanina, D. J. Green, G. J. Burckart, T. R. Cressey. Prediction of maternal and fetal pharmacokinetics of Dolutegravir and Raltegravir using physiologically based pharmacokinetic modeling.. Clin. Pharmacokinet., 2020a. [DOI | PubMed]
- X. I. Liu, J. D. Momper, N. Rakhmanina, J. N. van den Anker, D. J. Green, G. J. Burckart. Physiologically based pharmacokinetic models to predict maternal pharmacokinetics and fetal exposure to emtricitabine and acyclovir.. J. Clin. Pharmacol., 2020b. [DOI | PubMed]
- X. I. Liu, J. D. Momper, N. Y. Rakhmanina, D. J. Green, G. J. Burckart, T. R. Cressey. Physiologically based pharmacokinetic modeling framework to predict neonatal pharmacokinetics of transplacentally acquired emtricitabine, Dolutegravir, and Raltegravir.. Clin. Pharmacokinet., 2021. [DOI | PubMed]
- X. I. Liu, D. J. Green, J. van den Anker, H. K. Ahmadzia, G. J. Burckart, A. Dallmann. Development of a generic fetal physiologically based pharmacokinetic model and prediction of human maternal and fetal organ concentrations of cefuroxime.. Clin. Pharmacokinet., 2024. [DOI | PubMed]
- M. Louchet, M. Ribot, N. Bouazza, F. Foissac, L. Froelicher, V. Buth. Transplacental transfer of Remdesivir and GS-441524: an ex vivo perfusion study.. Health Sci. Rep., 2023. [DOI | PubMed]
- A. Lupattelli, O. Spigset, M. J. Twigg, K. Zagorodnikova, A. C. Mårdby, M. E. Moretti. Medication use in pregnancy: a cross-sectional, multinational web-based study.. BMJ Open, 2014. [DOI | PubMed]
- J. Macente, N. Nauwelaerts, F. M. Russo, J. Deprest, K. Allegaert, B. Lammens. PBPK-based dose finding for sildenafil in pregnant women for antenatal treatment of congenital diaphragmatic hernia.. Front. Pharmacol., 2023. [DOI | PubMed]
- L. L. Maroun, L. Mathiesen, M. Hedegaard, L. E. Knudsen, L. G. Larsen. Pathologic evaluation of normal and perfused term placental tissue.. Pediatr. Dev. Pathol., 2014. [DOI | PubMed]
- L. Mathiesen, T. Mose, T. J. Mørck, J. K. Nielsen, L. K. Nielsen, L. L. Maroun. Quality assessment of a placental perfusion protocol.. Reprod. Toxicol., 2010. [DOI | PubMed]
- L. Mathiesen, D. Sandal, I. E. M. Hammer, B. Styrishave, L. E. Knudsen. Models of endocrine-disrupting effects: human placental steroidogenesis.. Basic Clin. Pharmacol. Toxicol., 2025. [DOI | PubMed]
- C. J. Megli, C. B. Coyne. Infections at the maternal-fetal interface: an overview of pathogenesis and defence.. Nat. Rev. Microbiol., 2022. [DOI | PubMed]
- P. Mian, K. Allegaert, S. Conings, P. Annaert, D. Tibboel, M. Pfister. Integration of placental transfer in a fetal–maternal physiologically based pharmacokinetic model to characterize acetaminophen exposure and metabolic clearance in the fetus.. Clin. Pharmacokinet., 2020. [DOI | PubMed]
- H. D. Mistry, R. Klossner, S. Kallol, M. P. Lüthi, R. Moser, H. Schneider. Effects of aldosterone on the human placenta: insights from placental perfusion studies.. Placenta, 2022. [DOI | PubMed]
- K. H. Moore, H. A. Murphy, H. Chapman, E. M. George. Syncytialization alters the extracellular matrix and barrier function of placental trophoblasts.. Am. J. Physiol. Cell. Physiol., 2021. [DOI | PubMed]
- N. P. Mortensen, M. M. Caffaro, R. W. Snyder, Y. L. Yueh, T. R. Fennell. Placental trophoblast transfer of opioids following exposures to individual or mixtures of opioids in vitro .. Exp. Biol. Med. (Maywood), 2019. [DOI | PubMed]
- B. Mosavati, A. V. Oleinikov, E. Du. Development of an Organ-on-a-Chip-Device for study of placental pathologies.. Int. J. Mol. Sci., 2020. [DOI | PubMed]
- K. Motomura, H. Morita, H. Yamamoto, S. Wada, H. Sago, H. Takahashi. Isolation of pure primary term human cytotrophoblasts and their differentiation into syncytiotrophoblast-like cells as an ex vivo model of the human placenta.. Placenta, 2025. [DOI | PubMed]
- N. O. Nathan, M. Hedegaard, G. Karlsson, L. E. Knudsen, L. Mathiesen. Intrapartum transfer of oxytocin across the human placenta: an ex vivo perfusion experiment.. Placenta, 2021. [DOI | PubMed]
- J. Ning, A. Pansari, K. Rowland Yeo, A. T. Heikkinen, C. Waitt, L. M. Almond. Using PBPK modeling to supplement clinical data and support the safe and effective use of dolutegravir in pregnant and lactating women.. CPT Pharmacometrics Syst. Pharmacol., 2024. [DOI | PubMed]
- M. Noergaard, P. B. Jensen, D. Resendal Gotfredsen, T. Bergholt, J. Trærup Andersen, L. Mathiesen. Therapeutic concentration of ciprofloxacin and transfer across the human term placenta.. Am. J. Obstet. Gynecol., 2021. [DOI | PubMed]
- Y. Ohta, K. Kazuki, S. Abe, M. Oshimura, K. Kobayashi, Y. Kazuki. Development of Caco-2 cells expressing four CYPs via a Mammalian artificial chromosome.. BMC Biotechnol., 2020. [DOI | PubMed]
- H. Okae, H. Toh, T. Sato, H. Hiura, S. Takahashi, K. Shirane. Derivation of human trophoblast stem cells.. Cell. Stem Cell., 2018. [DOI | PubMed]
- M. D. Owen, B. C. Baker, E. M. Scott, K. Forbes. Interaction between metformin, folate and vitamin B(12) and the potential impact on fetal growth and long-term metabolic health in diabetic pregnancies.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- S. Öztürk, M. Demir, E. A. Koçkaya, C. Karaaslan, A. K. Süloğlu. Establishment of a 3D multicellular placental microtissues for investigating the effect of antidepressant vortioxetine.. Reprod. Toxicol., 2024. [DOI | PubMed]
- K. R. Page. Perfusion of isolated human placenta.. Proc. Nutr. Soc., 1991. [DOI | PubMed]
- N. Panse, P. M. Gerk. The Caco-2 model: modifications and enhancements to improve efficiency and predictive performance.. Int. J. Pharm., 2022. [DOI | PubMed]
- G. Pariente, T. Leibson, A. Carls, T. Adams-Webber, S. Ito, G. Koren. Pregnancy-Associated changes in pharmacokinetics: a systematic review.. PLoS Med., 2016. [DOI | PubMed]
- W. T. Parks. Placental hypoxia: the lesions of maternal malperfusion.. Semin. Perinatol., 2015. [DOI | PubMed]
- G. I. Patilea-Vrana, J. D. Unadkat. Development and verification of a linked Δ (9)-THC/11-OH-THC physiologically based pharmacokinetic model in healthy, nonpregnant population and extrapolation to pregnant women.. Drug Metab. Dispos., 2021. [DOI | PubMed]
- R. L. Pemathilaka, D. E. Reynolds, N. N. Hashemi. Drug transport across the human placenta: review of placenta-on-a-chip and previous approaches.. Interface Focus, 2019a. [DOI | PubMed]
- R. L. Pemathilaka, J. D. Caplin, S. S. Aykar, R. Montazami, N. N. Hashemi. Placenta-on-a-Chip: in vitro Study of caffeine transport across placental barrier using liquid chromatography Mass spectrometry.. Glob. Chall., 2019b. [DOI | PubMed]
- R. L. Pemathilaka, N. Alimoradi, D. E. Reynolds, N. N. Hashemi. Transport of maternally administered pharmaceutical agents across the placental barrier in vitro .. ACS Appl. Bio Mater, 2022. [DOI | PubMed]
- L. Pencolé, M. P. Lê, F. Bouchet-Crivat, D. Duro, G. Peytavin, L. Mandelbrot. Placental transfer of the integrase strand inhibitors cabotegravir and bictegravir in the ex-vivo human cotyledon perfusion model.. Aids, 2020. [DOI | PubMed]
- K. Phillips, K. Nirantharakumar, B. R. Wakerley, F. L. Crowe. Trends in the prevalence and pharmacological management of migraine during pregnancy in the UK, 2000-2018.. J. Neurol. Neurosurg. Psychiatry, 2024. [DOI | PubMed]
- V. C. Pillai, M. Shah, E. Rytting, T. N. Nanovskaya, X. Wang, S. M. Clark. Prediction of maternal and fetal pharmacokinetics of indomethacin in pregnancy.. Br. J. Clin. Pharmacol., 2022. [DOI | PubMed]
- L. Pinto, P. Bapat, F. de Lima Moreira, A. Lubetsky, R. de Carvalho Cavalli, H. Berger. Chiral transplacental pharmacokinetics of fexofenadine: impact of P-Glycoprotein inhibitor fluoxetine using the human placental perfusion model.. Pharm. Res., 2021. [DOI | PubMed]
- H. Polachek, N. Debotton, V. Feinshtein, M. Rubin, Z. Ben-Zvi, G. Holcberg. The role of various transporters in the placental uptake of ofloxacin in an in vitro model of human villous trophoblasts.. Drug Des. Devel Ther., 2018. [DOI | PubMed]
- C. Porter, S. Armstrong-Fisher, T. Kopotsha, B. Smith, T. Baker, L. Kevorkian. Certolizumab pegol does not bind the neonatal Fc receptor (FcRn): consequences for FcRn-mediated in vitro transcytosis and ex vivo human placental transfer.. J. Reprod. Immunol., 2016. [DOI | PubMed]
- M. S. Poulsen, E. Rytting, T. Mose, L. E. Knudsen. Modeling placental transport: correlation of in vitro BeWo cell permeability and ex vivo human placental perfusion.. Toxicol Vitro, 2009. [DOI | PubMed]
- L. S. Richardson, A. K. Kammala, M. M. Costantine, S. J. Fortunato, E. Radnaa, S. Kim. Testing of drugs using human feto-maternal interface organ-on-chips provide insights into pharmacokinetics and efficacy.. Lab. Chip, 2022. [DOI | PubMed]
- J. M. Roberts, C. Escudero. The placenta in preeclampsia.. Pregnancy Hypertens., 2012. [DOI | PubMed]
- D. Roelofsen, H. van Hove, V. Bukkems, F. Russel, G. Eliesen, R. Greupink. Predicting fetal exposure of crizotinib during pregnancy: combining human ex vivo placenta perfusion data with physiologically-based pharmacokinetic modeling.. Toxicol Vitro, 2022. [DOI | PubMed]
- Y. Sambuy, I. De Angelis, G. Ranaldi, M. L. Scarino, A. Stammati, F. Zucco. The Caco-2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco-2 cell functional characteristics.. Cell. Biol. Toxicol., 2005. [DOI | PubMed]
- S. Schalkwijk, R. Greupink, A. P. Colbers, A. C. Wouterse, V. G. Verweij, J. van Drongelen. Placental transfer of the HIV integrase inhibitor dolutegravir in an ex Vivo human cotyledon perfusion model.. J. Antimicrob. Chemother., 2016. [DOI | PubMed]
- S. Schalkwijk, A. O. Buaben, J. J. M. Freriksen, A. P. Colbers, D. M. Burger, R. Greupink. Prediction of fetal Darunavir exposure by integrating human Ex-Vivo placental transfer and physiologically based pharmacokinetic modeling.. Clin. Pharmacokinet., 2018. [DOI | PubMed]
- H. Schneider. Tolerance of human placental tissue to severe hypoxia and its relevance for dual ex vivo perfusion.. Placenta, 2009. [DOI | PubMed]
- H. Schneider. IFPA senior award lecture: energy metabolism of human placental tissue studied by ex vivo perfusion of an isolated cotyledon.. Placenta, 2015. [DOI | PubMed]
- H. Schneider, P. Brownbill, C. Albrecht. Ex vivo dual perfusion of an isolated cotyledon of human placenta: history and future challenges.. Placenta, 2021. [DOI | PubMed]
- H. Schneider, C. Albrecht, M. S. Ahmed, M. Broekhuizen, L. Aengenheister, T. Buerki-Thurnherr. Ex vivo dual perfusion of an isolated human placenta cotyledon: towards protocol standardization and improved inter-centre comparability.. Placenta, 2022. [DOI | PubMed]
- M. J. Shannon, G. L. McNeill, B. Koksal, J. Baltayeva, J. Wächter, B. Castellana. Single-cell assessment of primary and stem cell-derived human trophoblast organoids as placenta-modeling platforms.. Dev. Cell., 2024. [DOI | PubMed]
- B. Shenkoya, S. Atoyebi, I. Eniayewu, A. Akinloye, A. Olagunju. Mechanistic modeling of maternal lymphoid and fetal plasma antiretroviral exposure during the third trimester.. Front. Pediatr., 2021. [DOI | PubMed]
- B. Shenkoya, M. Gopalakrishnan, A. C. Eke. Physiologically based pharmacokinetic modeling of long-acting extended-release naltrexone in pregnant women with opioid use disorder.. CPT Pharmacometrics Syst. Pharmacol., 2024. [DOI | PubMed]
- S. Shojaei, M. S. Ali, M. Suresh, T. Upreti, V. Mogourian, M. Helewa. Dynamic placenta-on-a-chip model for fetal risk assessment of nanoparticles intended to treat pregnancy-associated diseases.. Biochim. Biophys. Acta Mol. Basis Dis., 2021. [DOI | PubMed]
- M. L. Shuler. Organ-body- and disease-on-a-chip systems.. Lab. Chip, 2017. [DOI | PubMed]
- S. Shum, D. D. Shen, N. Isoherranen. Predicting maternal-fetal disposition of fentanyl following intravenous and epidural administration using physiologically based pharmacokinetic modeling.. Drug Metab. Dispos., 2021. [DOI | PubMed]
- A. Subramanian, A. Azcoaga-Lorenzo, A. Anand, K. Phillips, S. I. Lee, N. Cockburn. Polypharmacy during pregnancy and associated risk factors: a retrospective analysis of 577 medication exposures among 1.5 million pregnancies in the UK, 2000-2019.. BMC Med., 2023. [DOI | PubMed]
- K. X. Szeto, M. Le Merdy, B. Dupont, M. B. Bolger, V. Lukacova. PBPK modeling approach to predict the behavior of drugs cleared by kidney in pregnant subjects and fetus.. Aaps J., 2021. [DOI | PubMed]
- M. Thunbo, J. H. Vendelbo, D. R. Witte, A. Larsen, L. H. Pedersen. Use of medication in pregnancy on the rise: study on 1.4 million Danish pregnancies from 1998 to 2018.. Acta Obstet. Gynecol. Scand., 2024. [DOI | PubMed]
- J. E. M. Van Der Heijden, H. Van Hove, N. M. Van Elst, P. Van Den Broek, J. Van Drongelen, H. C. J. Scheepers. Optimization of the betamethasone and dexamethasone dosing regimen during pregnancy: a combined placenta perfusion and pregnancy physiologically based pharmacokinetic modeling approach.. Am. J. Obstet. Gynecol., 2024. [DOI | PubMed]
- M. W. van Hoogdalem, R. Tanaka, K. Abduljalil, T. N. Johnson, S. L. Wexelblatt, H. T. Akinbi. Forecasting fetal buprenorphine exposure through maternal-fetal physiologically based pharmacokinetic modeling.. Pharmaceutics, 2024. [DOI | PubMed]
- C. van Kammen, H. van Hove, D. Kapsokalyvas, R. Greupink, R. Schiffelers, T. Lely. Targeted lipid nanoparticles to prevent trans-placental passage in the ex vivo human placental cotyledon perfusion model.. Drug Deliv. Transl. Res., 2024. [DOI | PubMed]
- A. Veiga-Lopez, E. R. Elkin, S. M. Harris, B. E. Blake, A. G. Paquette, L. M. Aleksunes. Current approaches and advances in placental toxicology.. Trends Endocrinol. Metab., 2025. [DOI | PubMed]
- M. S. Vidal, L. S. Richardson, A. Kumar Kammala, S. Kim, P. Y. Lam, R. Cherukuri. Endocrine-disrupting compounds and their impact on human placental function: evidence from placenta organ-on-chip studies.. Lab. Chip, 2024. [DOI | PubMed]
- R. Villalobos-Labra, R. Liu, F. Spaans, T. Sáez, T. Semeria Maitret, A. Quon. Placenta-Derived extracellular vesicles from preeclamptic pregnancies impair vascular endothelial function via lectin-like oxidized LDL Receptor-1.. Hypertension, 2023. [DOI | PubMed]
- S. D. Villota, M. Toledo-Rodriguez, L. Leach. Compromised barrier integrity of human feto-placental vessels from gestational diabetic pregnancies is related to downregulation of occludin expression.. Diabetologia, 2021. [DOI | PubMed]
- D. A. Volpe. Advances in cell-based permeability assays to screen drugs for intestinal absorption.. Expert Opin. Drug Discov., 2020. [DOI | PubMed]
- P. Vukomanović, M. Stefanović, J. M. Stevanović, A. Petrić, M. Trenkić, L. Andrejević. Monte Carlo optimization method based QSAR modeling of placental barrier permeability.. Pharm. Res., 2024. [DOI | PubMed]
- C. Wenzel, M. Drozdzik, S. Oswald. Organic cation transporter 1 an intestinal uptake transporter: fact or fiction?. Front. Pharmacol., 2021. [DOI | PubMed]
- M. A. Werdan Romão, L. Pinto, R. C. Cavalli, G. Duarte, N. V. de Moraes, K. Abduljalil. Mechanistic framework to predict maternal-placental-fetal pharmacokinetics of nifedipine employing physiologically based pharmacokinetic modeling approach.. J. Clin. Pharmacol., 2024. [DOI | PubMed]
- R. E. Wessel, S. Dolatshahi. Regulators of placental antibody transfer through a modeling lens.. Nat. Immunol., 2024. [DOI | PubMed]
- S. Yagel, S. M. Cohen, D. Goldman-Wohl. An integrated model of preeclampsia: a multifaceted syndrome of the maternal cardiovascular-placental-fetal array.. Am. J. Obstet. Gynecol., 2022. [DOI | PubMed]
- Y. Yan, Q. Wang, W. Wu, H. Yi, F. Xie. Evaluation of various approaches to estimate transplacental clearance of Vancomycin for predicting fetal concentrations using a maternal-fetal physiologically based pharmacokinetic model.. Pharm. Res., 2024. [DOI | PubMed]
- S. G. Zambuto, A. K. Scott, M. L. Oyen. Beyond 2D: novel biomaterial approaches for modeling the placenta.. Placenta, 2024. [DOI | PubMed]
- Z. Zhang, J. D. Unadkat. Development of a Novel Maternal-Fetal Physiologically Based Pharmacokinetic Model II: verification of the model for passive placental permeability drugs.. Drug Metab. Dispos., 2017. [DOI | PubMed]
- Z. Zhang, M. Z. Imperial, G. I. Patilea-Vrana, J. Wedagedera, L. Gaohua, J. D. Unadkat. Development of a novel maternal-fetal physiologically based pharmacokinetic model I: insights into factors that determine fetal drug exposure through simulations and sensitivity analyses.. Drug Metab. Dispos., 2017. [DOI | PubMed]
- S. Zhang, Z. Cheng, X. Li, Y. Shi, H. Zhu, T. Zhang. Trans-Placental transfer mechanisms of aromatic amine antioxidants (AAs) and p-Phenylenediamine quinones (PPD-Qs): evidence from human gestation exposure and the Rat Uterine perfusion model.. Environ. Sci. Technol., 2024. [DOI | PubMed]
- Q. Zheng, Q. Zhou, J. Li, Y. Tian, H. Huang, Q. Yao. Placental transfer of bromocriptine in an ex vivo human placental perfusion model.. J. Matern. Fetal Neonatal Med., 2019. [DOI | PubMed]
