New insights into the crosstalk between endocannabinoids and sphingosine-1-phosphate
Abstract
This review aims at highlighting the interplay between the endocannabinoids (eCBs) anandamide and 2-arachidonoylglycerol and sphingosine-1-phosphate (S1P) signaling. The eCBs and S1P are bioactive compounds that exemplify a paradigm of crosstalk among lipid signals, with profound implications for physiological processes and disease pathogenesis. Crosscommunication between eCBs and S1P occurs through multiple mechanisms: (i) receptor heterodimerization and coregulation, (ii) mutual metabolic modulation, and (iii) integrated regulation of downstream effectors. The latter emerged as a key mechanism underlying the bidirectional interactions between eCBs and S1P, with functional overlaps that regulate several processes, including inflammation, vascular function, and neuronal activity. In addition, cannabis-derived compounds (such as cannabidiol) can influence eCBs and S1P signaling, calling for further research into their therapeutic exploitation. Overall, the dynamic interplay between endogenous eCBs and S1P—as well as with exogenous cannabidiol—described here offers a compelling example of the complexity of interactions among bioactive lipids. A deeper mechanistic understanding of these relationships could pave the way to novel strategies in drug design and development, emphasizing the importance of integrated approaches in the study of bioactive lipid biochemistry.
Article type: Review Article
Keywords: endocannabinoids, anandamide, 2-arachidonoylglycerol, sphingosine-1-phosphate, cannabidiol, crosstalk, GPCRs, TRPV1, PPARγ
Affiliations: Department of Veterinary Medicine, University of Teramo, Teramo, Italy; Department of Experimental and Clinical Biomedical Sciences “Mario Serio”, University of Florence, Florence, Italy; Laboratory of Lipid Neurochemistry, European Center for Brain Research, IRCCS Santa Lucia Foundation of Rome, Rome, Italy; Department of Biotechnological and Applied Clinical Sciences, University of L’Aquila, L’Aquila, Italy
License: © 2025 The Authors CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.jbc.2025.110781 | PubMed: 41033556 | PMC: PMC12597274
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (7.0 MB)
Bioactive lipids: Endocannabinoids versus sphingosine-1-phosphate
Bioactive lipids—particularly endocannabinoids (eCBs) and sphingosine-1-phosphate (S1P)—act as cellular messengers with highly dynamic transport and trafficking mechanisms that drive their signaling activity (ref. 1, ref. 2). The major eCBs are anandamide (N-arachidonoylethanolamine [AEA]) and 2-arachidonoylglycerol (2-AG), which possess multiple biological functions (ref. 1, ref. 3, ref. 4). AEA and 2-AG are, respectively, an N-acylethanolamine (NAE) and a glycerol ester formed through condensation with the carboxylic group of arachidonic acid (AA) (ref. 5, ref. 6). Both eCBs serve as key homeostatic regulators throughout the body and play vital roles in numerous physiological functions, such as brain development, cell survival, neurotransmission, pain sensation, immune response, energy homeostasis, bone metabolism, and vascular tone, just to list a few (ref. 1, ref. 7).
S1P is an important bioactive sphingolipid with a distinctive molecular structure typically characterized by an 18-carbon sphingosine backbone and a phosphate group esterified to the first hydroxyl position (ref. 8, ref. 9). However, the sphingoid base chain length can vary significantly, ranging from 12 to 22 carbons (ref. 10). S1P is present in biological fluids and is locally produced by most cell types and tissues, with particular relevance in the central nervous system (CNS), immune system, and vascular system (ref. 11).
Despite their different origins, eCBs and S1P share key molecular features. Indeed, they are both primarily produced “on demand” upon specific cellular stimuli (ref. 12, ref. 13); in addition, eCBs—but not S1P—can also be stored in cytosolic organelles such as adiposomes (ref. 14, ref. 15). Both eCBs and S1P require specific carriers for transmembrane transport (in and out) and trafficking (ref. 1, ref. 16, ref. 17) and usually act as local mediators with short-range paracrine action because of rapid degradation by specific enzymes (ref. 1, ref. 16, ref. 17). Furthermore, eCBs and S1P are ligands at different G-protein–coupled receptors (GPCRs) (ref. 18, ref. 19) with putative crosstalk capabilities (ref. 20, ref. 21, ref. 22).
In this review, we will critically discuss the points of contact and intersection between eCBs and S1P signaling, at the molecular and functional levels.
A common strategy of metabolic enzymes, receptors, and transporters
The metabolic routes of eCBs and S1P reveal intriguing commonalities, despite the distinct chemical nature of these lipids. The endogenous levels of both classes are tightly controlled by distinct biosynthetic and catabolic enzymes, which in the case of AEA and 2-AG lead to different in vivo concentrations (higher for 2-AG than for AEA), and hence to distinct receptor activation and signaling thereof (ref. 23).
The biosynthesis of AEA and 2-AG occurs mainly through the action of N-acyl-phosphatidylethanolamine–specific phospholipase D (NAPE-PLD) (ref. 24) and diacylglycerol lipase α and β isoforms (ref. 25, ref. 26, ref. 27), respectively. However, studies performed in NAPE-PLD knockout mice have revealed additional pathways (ref. 28), with an apparent redundancy that warrants AEA production even when the primary NAPE-PLD–dependent pathway is compromised.
AEA is primarily degraded by fatty acid amide hydrolase (FAAH) into AA and ethanolamine (EtNH2) (ref. 29), whereas 2-AG is metabolized by monoacylglycerol lipase (MAGL) into AA and glycerol (ref. 30). Alternatively, AEA can undergo oxygenation via cyclooxygenase-2 (COX-2), forming prostaglandin-H2-EtNH2 (ref. 31, ref. 32), or it can be transformed by lipoxygenase (LOX) isozymes—5-LOX, 12-LOX, and 15-LOX—into hosphor derivatives like 11-HETE-EA, 12-HETE-EA, and 15-HETE-EA, respectively (ref. 33, ref. 34). Finally, cytochrome P450 (CYP) enzymes can epoxygenate AEA to form epoxyeicosatrienoyl-ethanolamide (ref. 35, ref. 36).
Much alike AEA, 2-AG can be oxygenated by COX-2, leading to PG-glycerol (ref. 37, ref. 38) and by LOX isozymes that form hosphor derivatives such as 12-HETE-G, 15-HETE-G, and eoxin derivatives (ref. 39, ref. 40, ref. 41). Epoxygenation of 2-AG by CYP at various positions results in different epoxyeicosatrienoyl-glycerols (ref. 42, ref. 43). These oxidative pathways represent critical control points for eCB signaling duration and can lead to new metabolites endowed with their own biological activity (ref. 44).
As for S1P, it is synthesized by ATP-dependent phosphorylation of sphingosine, catalyzed by sphingosine kinases 1 (SphK1) and 2 (SphK2), which have distinct subcellular localizations: SphK1 is primarily in the cytoplasm and cell membranes, whereas SphK2 is present mainly in mitochondria, nucleus, and endoplasmic reticulum (ref. 45, ref. 46, ref. 47). The SphK substrate sphingosine is, in turn, produced by ceramide hydrolysis driven by ceramidases (ref. 48). The catabolic pathways of cellular S1P involve S1P phosphatase–dependent dephosphorylation and irreversible cleavage into hexadecenal and EtNH2 phosphate via S1P lyase, which represents the only exit point for sphingolipid degradation (ref. 49). Extracellular S1P can also be degraded by lysophospholipid phosphatase 3 into sphingosine, which can then be taken up by cells for further metabolism (ref. 50).
Both eCBs and S1P primarily signal through distinct families of GPCRs. In particular, AEA and 2-AG target cannabinoid receptors 1 and 2 (CB1 and CB2), whereas S1P activates five distinct receptors (S1PR1–5) with both systems exhibiting overlapping anatomical distributions in the CNS and peripheral tissues (ref. 51, ref. 52, ref. 53, ref. 54). In addition to canonical CB1 and CB2 receptors, eCBs can bind to other GPCRs like GPR55, as well as non-GPCRs such as transient receptor potential vanilloid-1 (TRPV1) channel and nuclear peroxisome proliferator–activated receptors (PPARs) α, γ, and δ (ref. 20, ref. 55, ref. 56).
The biological activity of eCBs and S1P—both acting as intracellular messengers and extracellular agonists—is subjected to spatiotemporal regulation, which is tightly dependent on metabolic enzymes and transport/trafficking mechanisms (ref. 14, ref. 57, ref. 58, ref. 59). For instance, in the case of S1P, its metabolism is finely regulated by diverse stimuli, including growth factors, cytokines, hormones, and neurotransmitters (ref. 57, ref. 59), and the tissue-specific expression of S1P1–5 receptors contributes to the selectivity of intracellular signaling pathways triggered thereof (ref. 60). Much alike eCBs (ref. 61, ref. 62), the rapid metabolism of S1P is critical to maintain its endogenous tone and hence signaling pathways.
Of note, S1P is not the only bioactive sphingolipid. Indeed, ceramide—considered the central hub of the sphingolipid pathway—has opposite effects compared with S1P: it induces apoptosis, whereas S1P promotes cell survival and proliferation (ref. 63). Remarkably, ceramide metabolism is closely interconnected with that of S1P and is regarded as the “sphingolipid rheostat” that determines cell fate (ref. 64). Due to its polar nature, intracellularly produced S1P must flip to the outer layer of plasma membrane in order to interact with its receptors (the so-called inside–out signaling) or to be released into the bloodstream (ref. 65). In this context, it should be recalled that S1P levels significantly vary between different body districts and may be altered under disease conditions (ref. 11).
Although eCBs and S1P are primarily produced inside the cell and then exported outside in response to various stimuli, emerging evidence demonstrates that both lipids can also be packaged into and transported by extracellular vesicles (Evs) (ref. 66, ref. 67). Indeed, Evs serve as carriers that protect these hydrophobic signals from degradation and facilitate their transport across aqueous environments to distant cellular targets. The packaging of eCBs in microvesicles has been demonstrated in microglial cells, where AEA localizes on vesicle surfaces and remains biologically active, that is, capable of binding to and activating presynaptic CB1 receptors to modulate synaptic transmission (ref. 66, ref. 68). Similarly, S1P—and in some cases even its biosynthetic enzymes—have been detected within Evs released from various cell types, such as hepatocarcinoma cells, endothelial cells, and pancreatic β-cells (ref. 67, ref. 69, ref. 70, ref. 71). The vesicular transport of eCBs and S1P not only extends their signaling range beyond the production sites but also provides a mechanism that enables navigation of these lipids through aqueous extracellular environments to reach their ultimate targets.
Further details of eCB and S1P systems—including metabolic enzymes, receptors, transporters, and trafficking mechanisms—are summarized in Box 1 and Box 2, respectively (Figs. 1 and 2).
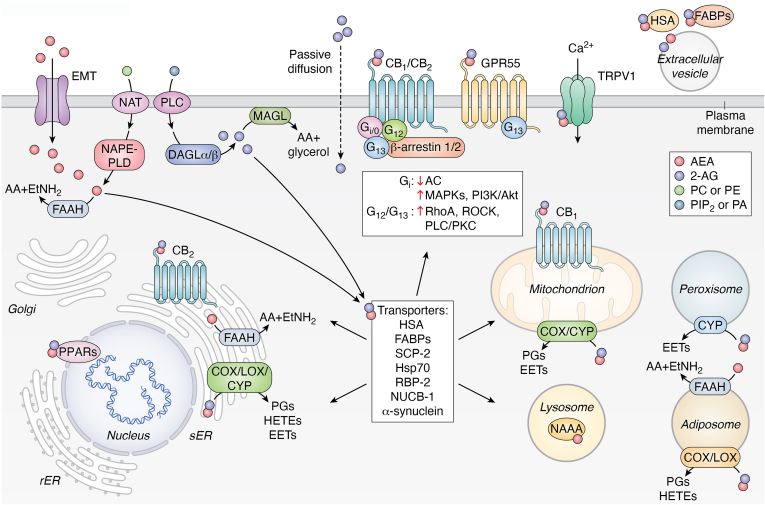
Box 1
The eCB system
The eCB system represents a complex network of signaling pathways primarily mediated by endogenous ligands known as eCBs, including AEA and 2-AG (ref. 1). AEA biosynthesis begins with the metabolism of AA-containing NAPEs, which are derived from the acylation of the amine group of phosphatidylethanolamines by both Ca2+-dependent and –independent N-acyltransferases, including cytosolic phospholipase A2ε (ref. 113).
Several biosynthetic routes contribute to AEA and other NAE formation. In the classical pathway, NAPE-PLD hydrolyzes NAPEs (ref. 24, ref. 114), whereas alternative pathways involve phospholipase C (PLC)-hydrolyzing NAPEs to generate hosphor-AEA (ref. 115), which is subsequently dephosphorylated by phosphatases, including PTPN22 and SHIP1 (ref. 115, ref. 116, ref. 117). In addition, PLA2 generates lyso-NAPE, which is further hydrolyzed by lysophospholipase D (ref. 117, ref. 118).
Once biosynthesized, AEA can undergo degradation or be transformed by oxygenation. Known enzymes of AEA hydrolysis include (1) FAAH, which hydrolyzes AEA (but also other NAEs) into AA and EtNH2, controlling the endogenous AEA tone (ref. 29, ref. 119); (2) the lysosomal N-acylethanolamine acid amidase, which preferentially processes saturated NAEs, such as N-palmitoyl-EtNH2 (ref. 120, ref. 121).
2-AG biosynthesis originates from AA-containing diacylglycerols (DAGs), typically derived from PLC-β hydrolysis of phosphatidylinositol 4,5-bisphosphate or from phosphatidic acid hydrolysis by phosphatidic acid phosphohydrolase (ref. 122). Subsequently, Ca2+-dependent PLC or DAG lipases (α and β) further hydrolyze DAG to release 2-AG (ref. 25, ref. 26, ref. 27). 2-AG is degraded mainly by MAGL (ref. 30) or by α,β-hydrolase domain–containing proteins, ABHD12 and ABHD6, responsible for approximately 9% and 4%, respectively, or by carboxylesterases (CES1 and CES2), producing fatty acids and glycerol (ref. 123, ref. 124).
The biological activity of AEA and 2-AG is primarily mediated through interactions with cannabinoid receptors type 1 and 2 (CB1 and CB2), which belong to the GPCR superfamily (ref. 125). Upon activation, CB1/2 receptors modulate diverse intracellular signaling cascades, including those involving cAMP, PI3K, and MAPK. This results in varied cellular responses, such as alterations in cell motility, survival, proliferation, and neurotransmitter release (ref. 126, ref. 127).
Specifically, CB1 is predominantly expressed in the CNS, including regions such as the neocortex, hippocampus, basal ganglia, cerebellum, and brainstem, whereas CB2 is primarily found in immune cells and peripheral tissues, with lower levels present in the CNS, especially in microglial cells and neurons of the brainstem and spinal cord (ref. 22, ref. 128).
In addition to CB1 and CB2, AEA and 2-AG can also activate nonclassical cannabinoid receptors, such as GPR55 (ref. 55, ref. 129). GPR55 is implicated in signaling pathways related to calcium release and Ras homolog gene family member A activation, affecting cell proliferation and migration (ref. 55, ref. 127, ref. 130). Furthermore, eCBs, especially AEA and 2-AG, interact with the TRPV1 receptor, a cation channel involved in nociception and thermoregulation. eCBs bind to the same vanilloid-binding site as capsaicin but with distinct configurations that reduce their partial agonist efficacy compared with capsaicin (ref. 20, ref. 21, ref. 56, ref. 131).
Despite their preference for GPCRs, both AEA and 2-AG can also activate PPARs, which are nuclear receptors that modulate gene expression by binding to DNA peroxisome proliferator response elements. These receptors have three isoforms: PPARα, PPARγ, and PPARδ (ref. 74, ref. 132, ref. 133). AEA and 2-AG primarily activate PPARα and PPARγ, thereby influencing lipid metabolism, energy homeostasis, and inflammatory responses (ref. 74). Notably, PPARα activation by eCBs plays a significant role in hepatic lipid metabolism, whereas PPARγ modulates adipogenesis and glucose metabolism (ref. 133).
As uncharged lipids, eCBs can cross the membrane bilayer through passive diffusion (ref. 134) or facilitated diffusion via a putative transmembrane carrier termed eCB membrane transport (ref. 135, ref. 136), which acts bidirectionally (ref. 135). Once inside the cell, intracellular binding proteins play crucial roles in not only eCB trafficking, particularly fatty acid–binding proteins, but also retinol-binding protein 2, sterol carrier protein 2, nucleobindin-1, human serum albumin, and heat shock protein 70 for breakdown by the hydrolase FAAH, thereby terminating signal transduction and receptor signaling (ref. 137, ref. 138, ref. 139, ref. 140, ref. 141, ref. 142, ref. 143, ref. 144).
Alternatively, eCBs can be transported to other cellular compartments, such as (i) mitochondria for oxidation by COX-2 or CYP (ref. 14, ref. 36, ref. 145) and potential activation of CB1 (ref. 146, ref. 147); (ii) lysosomes for degradation by N-acylethanolamine acid amidase (ref. 121, ref. 146); or (iii) the nucleus for PPAR activation (ref. 14, ref. 74).
In addition to accumulation and sequestration in specific organelles, extracellular transport of eCBs is mediated not only by fatty acid–binding proteins but also by Evs and proteins, such as human serum albumin, contributing to the fine regulation of eCB activity (ref. 15, ref. 68, ref. 137, ref. 142, ref. 148). Furthermore, synucleins (α-Syn) can facilitate postsynaptical eCB release, with synuclein deletion blocking eCB-mediated synaptic plasticity (ref. 149). These intricate regulations allow eCBs to exert precise and context-dependent effects in maintaining homeostasis throughout the body, and when disrupted, can lead to pathological conditions. The different receptors (GPCR, nuclear receptors, and TRPV1), metabolic enzymes, trafficking elements, and transmembrane transport of eCBs are schematically depicted in Figure 1.
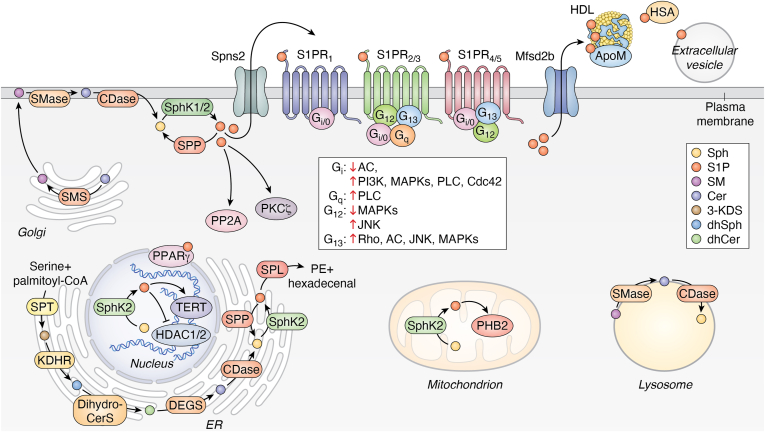
Box 2
The S1P system
S1P is a potent bioactive sphingolipid generated by the ATP-dependent phosphorylation of sphingosine catalyzed by two distinct enzymes, SphK1 and 2, which differ in subcellular localization, regulatory properties, and often exert divergent functions (ref. 8, ref. 150). This sphingolipid was regarded for a long time as a mere metabolite in the catabolic pathway of sphingolipids, where the only exit route is represented by the cleavage of S1P into phosphoethanolamine and hexadecenal, catalyzed by S1P lyase (ref. 151). However, it is now well established that S1P formation is intimately connected to ceramide metabolism, which is considered the central hub of sphingolipid metabolism, and is also biologically active.
Ceramide can be synthesized de novo in the endoplasmic reticulum through a pathway initiated by serine palmitoyltransferase, which catalyzes the condensation of serine and palmitoyl-CoA to form 3-keto-dihydrosphingosine. This product is then reduced to dihydrosphingosine, acylated by a family of six (dihydro)ceramide synthases to dihydroceramide and finally converted into ceramide by ceramide desaturase (ref. 152). Ceramide serves as a precursor of glycosphingolipids and sphingomyelin, which are key components of biological membranes. In the synthesis of sphingomyelin, a phosphorylcholine group is transferred from phosphatidylcholine to ceramide by sphingomyelin synthase. Alternatively, ceramide coupling with a glucose or a galactose residue initiates glycosphingolipid synthesis, eventually followed by further glycosyltransferase reactions (ref. 152, ref. 153). Notably, sphingosine, which serves as a product of sphingomyelin catabolism, is largely salvaged through reacylation by (dihydro)ceramide synthase, thus producing ceramide, playing a prominent role in maintaining the cellular ceramide levels (ref. 154). The main pathway responsible for S1P biosynthesis relies on sphingomyelin hydrolysis driven by sphingomyelinase, which releases ceramide that is further cleaved by ceramidase to produce sphingosine, which is then phosphorylated by SphK1/2 (ref. 8, ref. 150, ref. 152).
Besides the S1P lyase–directed irreversible breakdown, S1P can also be dephosphorylated to sphingosine by two specific S1P phosphatases, whereas lysophospholipid phosphatase 3 has a prominent role in its dephosphorylation in extracellular compartments (ref. 50, ref. 155). Consistent with its function as a cellular mediator, S1P metabolism is strictly controlled: SphK1 and SphK2 are highly regulated at transcriptional, translational, and post-translational levels (ref. 45, ref. 46, ref. 156).
Once formed, S1P can either act as an intracellular signaling molecule or be transported extracellularly to exert its effects. Two specific transporters, Mfsd2b and Spns2, in red blood cells, platelets, and endothelial cells, respectively (ref. 17, ref. 157, ref. 158), ensure the maintenance of plasma levels of S1P, whereas Spns2 alone participates in S1P export in other cellular contexts where S1P acts through the so-called inside–out signaling mechanism (ref. 65).
While S1P levels in most animal tissues are typically in the nanomolar range because of its rapid turnover, its concentration in plasma is approximately 100 times higher. Notably, the significant concentration gradient of S1P between plasma and interstitial fluids is important for immune cell homing to lymphoid organs and regulating their egress into blood and lymph (ref. 49, ref. 159, ref. 160).
The amphipathic nature of S1P hinders its free state in the aqueous phase; indeed, more than 50% of plasma S1P is associated with high-density lipoprotein, whereas approximately 35% is bound to albumin, and the remainder is likely bound to other lipoproteins. ApoM, a component of high-density lipoprotein, is the unique high-affinity S1P-binding protein that preserves S1P from hydrolysis and regulates its function by facilitating interaction with its receptors, thus enhancing its biological functions (ref. 17, ref. 161, ref. 162, ref. 163, ref. 164, ref. 165).
S1P, acting as S1P1–5 ligand, has emerged as a potent bioactive lipid mediator, orchestrating numerous cellular functions, including cell proliferation, survival, motility, and differentiation. S1P receptors transmit diverse intracellular signals depending on the coupled Gα subunits of heterotrimeric G proteins and the expression pattern of each receptor in a given cell (ref. 8, ref. 166, ref. 167).
S1P1 is ubiquitously expressed, with a prominent role in controlling endothelial integrity and immune cell trafficking. It couples exclusively with Gi proteins, activating PLC, Ras, PI3K, and inhibiting adenylyl cyclase (ref. 168). S1P2, which is abundantly expressed, interacts with Gi, Gq, and G13 proteins and negatively regulates cell motility by activating Rho and inhibiting Rac (ref. 73, ref. 169). S1P3, sharing structural homology with cannabinoid receptors and also abundantly expressed, interacts with Gi, Gq, and G13 proteins (ref. 54). It regulates cytoskeletal reorganization and promotes tissue fibrosis. S1P4 and S1P5 expression is more restricted, with a role in the immune and nervous systems, respectively (ref. 13, ref. 54, ref. 170).
S1P, initially discovered as an intracellular messenger, also acts by binding to intracellular protein targets. Indeed, S1P can function as a ligand for the nuclear receptor PPARγ (ref. 75), as detailed in the text. Moreover, S1P produced within the nucleus specifically binds to histone deacetylase-1 and -2, thereby inhibiting their enzymatic activity and consequently affecting gene expression, thus linking S1P to epigenetic regulation of gene expression (ref. 171). The mitochondrial protein prohibitin-2 is another intracellular target of S1P, important for oxidative phosphorylation (ref. 172). In addition, S1P has been shown to bind to human telomerase reverse transcriptase by allosterically mimicking protein phosphorylation, suggesting a role in regulating telomere integrity, which may contribute to delaying cellular senescence and promoting longevity (ref. 173). Furthermore, it has been recently reported that in red blood cells, S1P regulates glucose transport by binding and activating of the catalytic subunit of protein phosphatase 2A to reduce GLUT1 phosphorylation, cell surface localization, and glucose uptake (ref. 174). Finally, cellular S1P has been shown to directly activate PKCζ by relieving autoinhibitory constraints (ref. 175), thereby driving lipolysis in adipocytes (ref. 176).
Collectively, this comprehensive picture underscores the complexity of sphingolipid metabolism and signaling, with special emphasis on S1P, as summarized in Figure 2.
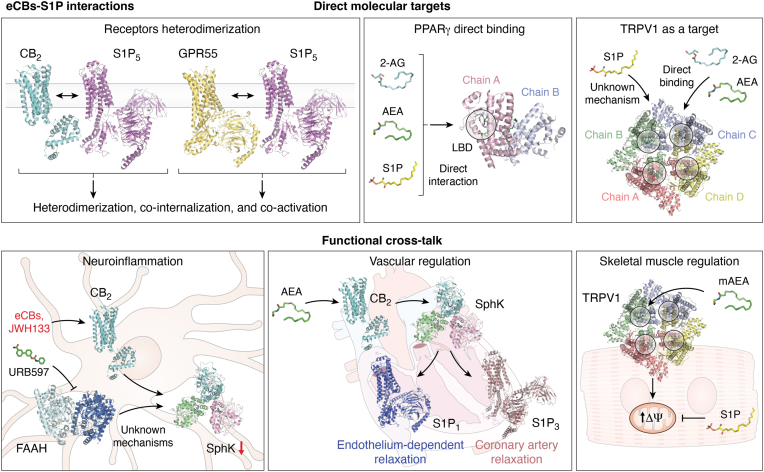
Crosstalk between eCBs and S1P
Crosscommunication between eCB and S1P systems occurs through multiple mechanisms: (i) receptor heterodimerization and coregulation, (ii) mutual metabolic modulation, and (iii) integrated regulation of downstream effectors.
Heterodimerization and coregulation
Heterodimerization (i.e., the formation of a complex between two different receptor partners) and coregulation of distinct receptors play crucial roles in intracellular signaling (ref. 72, ref. 73), with a particular impact on the interaction between S1P-binding and eCB-binding receptors, and hence on energy homeostasis, cancer progression, and pain modulation.
The interaction between CB2 and S1P5 receptors forms a regulatory checkpoint that limits tumor progression in glioblastoma cells, as revealed by bioluminescence resonance energy transfer analysis (ref. 73). Confocal microscopy confirmed colocalization and cointernalization into the cytoplasm upon agonist stimulation. Moreover, CB2 upregulated tumor progression genes like Ki67 and p21, but S1P5 costimulation attenuated these effects by 60% to 70%. Such a CB2–S1P5 receptor heterodimerization expands the functional diversity of both lipid signaling systems, enabling context-dependent modulation of cellular responses (ref. 72, ref. 73). The heterodimerization of S1P5 with GPR55 represents another regulatory checkpoint that balances protumorigenic and antitumorigenic signals. Indeed, GPR55 and S1P5 form a heterodimer in colon cancer cells, which involves the transmembrane segments of S1P5 with a minimal contribution from its C-terminal region, as demonstrated by bioluminescence resonance energy transfer experiments (ref. 72). When activated independently, each receptor promotes extracellular signal–regulated kinase phosphorylation, upregulation of protumorigenic genes ATF3, HIF-1α, and Sp1, and thus colon cancer cell proliferation. Instead, simultaneous stimulation of both receptors triggers cointernalization of the heterodimer, ultimately suppressing tumor progression (ref. 72).
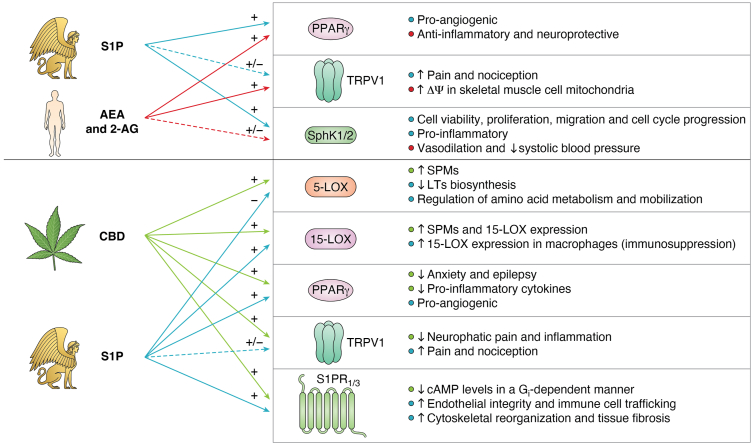
An example of receptor coregulation is the ability of both S1P and eCBs to interact with PPARγ through distinct molecular mechanisms, thus influencing gene regulation and therapeutic outcomes (ref. 74, ref. 75). Parham et al. (ref. 75) showed that S1P directly interacts with PPARγ through its ligand-binding domain that involves histidine 323, as revealed by in silico docking and in vitro binding assays. At physiological concentrations, S1P stimulates recruitment of the PPARγ coactivator 1-β, leading to the formation of a transcriptional complex within endothelial cells (ref. 75). Accordingly, S1P activates PPARγ-dependent gene reporters, selectively modulating target genes by upregulating PPARγ coactivator 1-β and plasminogen activator inhibitor-1, while suppressing the expression of the scavenger receptor CD36. Importantly, the proangiogenic action of S1P, leading to in vitro tube formation, is PPARγ dependent and is blocked by PPARγ antagonists (ref. 75).
Notably, AEA and 2-AG can also bind directly to the ligand-binding domain of PPARγ, contributing to anti-inflammatory and neuroprotective effects (ref. 74). Moreover, 2-AG also interacts indirectly with PPARγ via its COX-2-generated metabolites (ref. 76). Further indirect mechanisms of eCB–S1P intersection include (i) PPARγ ligand (e.g., ciglitazone)-dependent inhibition of FAAH and subsequent increase of AEA levels that potentiate PPARγ activation (ref. 77) and (ii) synergistic effect of CB1–CB2 signaling with PPARγ pathways, to mediate neuroprotection and related metabolic effects (ref. 74, ref. 78, ref. 79).
Mutual metabolic modulation
Communication between the eCB and S1P systems also occurs through regulation of their metabolic enzymes. In rat carotid arteries, AEA induces relaxation through a mechanism that involves CB2, which subsequently activates SphK1 to produce S1P. The latter, in turn, acts via S1P3 receptor in the vascular endothelium to promote vasodilation (ref. 80). Similarly, the activation of SphK1, but not of SphK2, and the engagement of S1P1 that is expressed at the level of vascular smooth muscle, is required for the reduction of systolic blood pressure exerted by AEA (ref. 81).
Moreover, in lipopolysaccharide (LPS)-stimulated BV2 microglia cells, transcriptional upregulation of both SphK1 and SphK2 is induced, contributing to increased production of proinflammatory cytokines like tumor necrosis factor α and interleukin-1β (ref. 22). In the same cells, inhibition of FAAH prevents LPS-induced SphK1 and SphK2 upregulation as well as cytokine production. In addition, activation of CB2 by the synthetic agonist JWH133 mimics the anti-inflammatory effects of FAAH inhibition, positioning SphK1 and SphK2 at the intersection between proinflammatory LPS and anti-inflammatory eCB signaling pathways in microglia (ref. 22).
Also, LOX isozymes, which metabolize eCBs into oxylipin derivatives like hydroxyeicosatetraenoic acids (ref. 82), are modulated by S1P (ref. 83, ref. 84). For instance, S1P can reduce 5-LOX activity—particularly in immune cells like neutrophils and macrophages—through S1P4 engagement (ref. 83). In general, it should be noted that regulation of AA metabolism by S1P likely affects eCB tone and biological activity. Although the eCB–S1P crosstalk via AA is complex and still under investigation, several mechanistic links support its potential in vivo relevance. S1P can influence intracellular AA mobilization by activating calcium-independent and calcium-dependent phospholipases A2 (iPLA2 and cPLA2) (ref. 85). By inducing the expression of the AA dioxygenase COX-2, S1P triggers a metabolic competition that favors the fueling of prostaglandin production at the expense of eCB synthesis (ref. 86). Depending on the target tissue, such a switch of AA metabolism toward prostaglandins or eCBs may tip the balance between proinflammatory and anti-inflammatory effects or pronociceptive and antinociceptive effects (ref. 87). While direct experimental evidence linking S1P-mediated regulation of AA metabolism to changes in eCB levels is limited, the biochemical overlaps between S1P and eCB systems speak in favor of this hypothesis. Ongoing research in lipidomics and cell signaling has begun to uncover this intricate crosstalk, especially in contexts such as neurodegenerative diseases, cancer, chronic pain, and immune function (ref. 88, ref. 89, ref. 90).
A third mechanism of intersection between S1P and eCB signal transduction pathways involves the eCB-binding receptor TRPV1, which is also a key effector of S1P in several cellular contexts (ref. 91, ref. 92, ref. 93). In fact, S1P transactivates TRPV1 in primary sensory neurons to regulate nociception and pain. In particular, TRPV1 is coexpressed with S1P3 receptors in subsets of nociceptors, where S1P3 stimulation potentiates TRPV1 activity, overall leading to heat hypersensitivity and pain (ref. 91). Likewise, S1P-induced calcium influx in dorsal root ganglia neurons is partially abolished by TRPV1 antagonists, whereas in vivo studies show that TRPV1-knockout mice exhibit significantly reduced pain responses (e.g., wiping and thermal hyperalgesia) after S1P injection (ref. 92). At the mechanistic level, S1P enhances TRPV1-mediated thermal pain sensitivity via a rapid Gαi-coupled signaling pathway that involves PI3K, PKC, and p38 mitogen-activated protein kinase (MAPK) l (ref. 93).
Finally, in a recent study performed in murine C2C12 myoblasts, S1P treatment for 24 h significantly increased TRPV1 expression at both mRNA and protein levels, whereas reducing CB2 protein levels. The opposite regulation of TRPV1 and CB2 by S1P suggests a mechanism whereby S1P channels eCB signaling toward TRPV1-dependent pathways, which are critical for calcium influx and mitochondrial function (ref. 21). In the same C2C12 cells, TRPV1 activation by the stable AEA analog methanandamide (mAEA) increases mitochondrial membrane potential (ΔΨm) and upregulates PPARγ coactivator 1-α, a key regulator of mitochondrial biogenesis. Of note, treatment with S1P counteracts these effects of mAEA, likely through TRPV1 desensitization or altered calcium flux (ref. 21). The interplay between mAEA and S1P in C2C12 cells appears critical during myogenesis, where balanced mitochondrial activity is needed to ensure energy supply for cell differentiation.
A summary of the interactions between eCB and S1P at the structural and functional levels is shown in Figure 3.
Integrated regulation of downstream effectors
Crosscommunication between eCBs and S1P can also occur through the integrated regulation of downstream effectors triggered by the binding of eCBs and S1P to their receptor targets. Indeed, both lipid signaling systems modulate a variety of physiological processes via binding to Gi-, Gq-, and G12/13-coupled receptors, which are coexpressed in specific body districts (i.e., CNS, immune, and vascular systems), thus allowing context-specific integration of signals. As a consequence, the cooperative modulation of effectors, like ion channels, protein kinases, and nuclear transcription factors, may have important implications in physiological and pathological processes.
Both eCB and S1P systems regulate common effectors that drive distinct signal transduction pathways, such as adenylyl cyclase, MAPK/extracellular signal–regulated kinase, PI3K/Akt, and Rho/ROCK (Rho-associated protein kinase). The crosstalk between eCBs and S1P can reinforce or weaken a specific pathway of signal transduction in both the CNS and peripheral tissues, as schematically summarized in Table 1.
Table 1: Commonalities in downstream effectors triggered by eCBs and S1P in different cell processes
| Process | Bioactive lipid | Receptor | G protein | Effectors | Biological evidence | References |
|---|---|---|---|---|---|---|
| Modulation of neurotransmission | S1P | S1P3 | Gi/0, Gq | ↑PLC, ↑ERK | ↑ Glutamate release in hippocampus, modulating synaptic transmission | (ref. 177, ref. 178, ref. 179) |
| S1P1 | Gi/0, Gβγ | ↓Calcium influx | ↓ Glutamate release | (ref. 180) | ||
| eCB | CB1 | Gi/0 | ↓AC, ↓calcium mobilization, ↑MAPK inwardly rectifying potassium currents | ↓ Neurotransmitter release, modulating synaptic transmission | (ref. 181, ref. 182) | |
| TRPV1 | NA (cation channel) | ↑Calcium mobilization | ↑ Synaptic plasticity and long-term depression | (ref. 183) | ||
| Immune cell trafficking | S1P | S1P1 | Gi/0 | ↑PI3K/Akt, Rac, MAPK | ↑ T-cell egress from lymphoid organs | (ref. 184) |
| S1P2/3 | G12/13 | ↑Rho/ROCK | ↑ Proinflammatory cytokine release in macrophages and endothelial cells | (ref. 169, ref. 185, ref. 186) | ||
| eCB | CB1 | Gi/0 | ↓Cytokine release; induces an anti-inflammatory phenotype | (ref. 187) | ||
| CB2 | Gi/0 | ↓Cytokine release modulates immune cell function and inflammation | (ref. 188) | |||
| GPR55 | G13 | ↑RhoA, Cdc42, and Rac1 | ↑ Cell migration and inflammatory responses | (ref. 189) | ||
| Lipid metabolism and insulin sensitivity | S1P | S1P1-3,4 | Gi/0, Gq | ↑PI3K/Akt, AMPK, calcium mobilization | (ref. 190, ref. 191) | |
| eCB | CB1 | Gi/0 | ↑p38 MAPK; ↑ERK1/2; ↓mTORC2, ↓IRS1-PI3K-Akt ↓AMPK | (ref. 192, ref. 193, ref. 194) | ||
| CB2 | Gi/0, Gβγ | PI3K activation, Akt phosphorylation ERK1/2, and CREB | (ref. 195, ref. 196) | |||
| Cardioprotection and vascular development/integrity | S1P | S1P1 | Gi/0 | ↑PI3K/Akt, ↑eNOS | ↑ Vascular maturation and integrity | (ref. 197) |
| S1P2 | G12/13, Gq | ↑ Vascular permeability and endothelial dysfunction | (ref. 2, ref. 198) | |||
| S1P3 | G13 | ↑Rho/ROCK | ↑Cardioprotection after I/R injury | (ref. 199) | ||
| eCB | CB1 | Gi/0 | ↓AC, ↑MAPK | ↓Cardiac contractility, associated with cirrhosis and heart failure | (ref. 200) | |
| CB2 | Gi/0 | ↑ PI3K/Akt, ERK1/2, STAT-3, eNOS | ↑Cardioprotection after I/R injury | (ref. 201, ref. 202, ref. 203) |
Akt, protein kinase B; AMPK, AMP-activated protein kinase; Cdc42, cell division cycle 42; CREB, cAMP response element–binding protein; eNOS, endothelial nitric oxide synthase; ERK, extracellular signal–regulated kinase; Gβγ, G beta–gamma subunits; Gi/0, inhibitory G protein alpha subunit; Gq, Gq alpha subunit; G12/13, G12 and G13 alpha subunits; I/R, ischemia/reperfusion; IRS1, insulin receptor substrate 1; MAPK, mitogen-activated protein kinase; mTORC2, mechanistic target of rapamycin complex 2; NA, not applicable; PLC, phospholipase C; Rac, Ras-related C3 botulinum toxin substrate; Rac1, Ras-related C3 botulinum toxin substrate 1; Rho, Ras homolog family member; ROCK, Rho-associated protein kinase; STAT-3, signal transducer and activator of transcription 3.
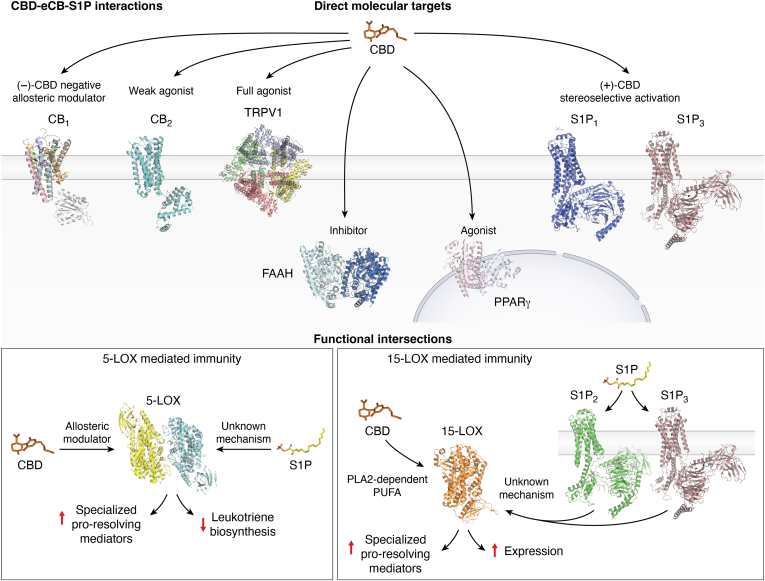
Cannabidiol: A link between eCBs and S1P
Among the numerous constituents of cannabis (Cannabis sativa) preparations that interact with the eCB system, cannabidiol (CBD) has emerged as one of the most intriguing compounds for its therapeutic potential (ref. 94). The (+)-enantiomer of CBD, unlike its (−)-counterpart, has been shown to potently activate S1P1 and S1P3 receptors, thereby reducing cAMP levels in a Gi-dependent manner (ref. 95). This effect was blocked by both dual and selective S1P1 and S1P3 antagonists, confirming the potential of (+)-CBD for the treatment of neurological disorders via S1P signaling (ref. 95). Notably, (+)-CBD also binds to CB1 as an inverse agonist (ref. 96, ref. 97, ref. 98), whereas (−)-CBD acts as a negative allosteric modulator of the same receptor and reduces the efficacy and potency of Δ9-tetrahydrocannabinol—the main psychoactive ingredient of cannabis—and AEA at CB1 (ref. 99). Moreover, CBD acts as a full agonist at TRPV1 channels (ref. 100), and with lower potency at CB2 (ref. 101) and PPARγ (ref. 102), thereby influencing anxiety, epilepsy, neuropathic pain, and inflammation (ref. 103).
In addition to eCBs—and S1P-binding receptors—CBD also modulates the metabolic enzymes of these lipids. Indeed, CBD is a potent inhibitor of FAAH, thus increasing cellular levels and signaling activity of AEA (ref. 104). CBD also modulates 2-AG metabolism by inhibiting MAGL and the other 2-AG hydrolases, ABHD6 and ABHD12 (ref. 105). In parallel, CBD influences sphingolipid metabolism by increasing S1P levels while reducing ceramide accumulation. The latter effect clearly shifts the ceramide–S1P rheostat toward cell survival and improved insulin sensitivity in the CNS (ref. 106, ref. 107), as well as in peripheral tissues like skeletal muscles, where it ameliorates glucose metabolism (ref. 103, ref. 106). Incidentally, another cannabis ingredient, cannabigerol, when administered in a rat model of high-fat–induced insulin resistance, increases ceramide conversion to S1P, potentially improving insulin sensitivity and providing protection from liver steatosis (ref. 108).
CBD has been shown to inhibit several CYP enzymes, particularly CYP2J2, which metabolizes eCBs into bioactive epoxyeicosatrienoic acids (ref. 109). This metabolic interference creates a complex pharmacological profile, whereby CBD not only enhances eCB tone but also redirects eCB metabolism toward alternative pathways that may influence the sphingolipid system, in particular through production of common precursors like AA.
The intersection of CBD and S1P signaling can also occur at the level of LOX isozymes, which represents an emerging and particularly fascinating area of bioactive lipid crossregulation. In fact, allosteric modulation of 5-LOX by CBD can shift its enzymatic activity from producing proinflammatory leukotrienes to generating anti-inflammatory specialized proresolving mediators (ref. 110). In line with this, S1P negatively modulates 5-LOX activity and reduces leukotriene biosynthesis to exert potent anti-inflammatory effects (ref. 83). Moreover, CBD strongly induces the formation of specialized proresolving mediators and 12/15-LOX products in resting cells by stimulating cPLA2-dependent release of polyunsaturated fatty acids as well as through allosteric activation of 15-LOX (ref. 110). In addition, S1P upregulates 15-LOX expression through a complex molecular pathway that engages activation of S1P1 and S1P3 receptors in macrophages, contributing to immunosuppressive tumor microenvironments (ref. 84). Overall, the intersections between CBD, S1P, and eCBs hold a promising therapeutic potential—as yet poorly explored—for the treatment of inflammatory conditions through their convergent effects on the production of bioactive lipids.
A summary of the interactions between CBD, eCBs, and S1P at the structural and functional levels is shown in Figure 4.
Conclusions
This review has highlighted how eCBs and S1P represent two complex signaling networks that, beyond canonical GPCR activation, can mutually influence metabolism, transport, trafficking, and transcription factor activity of each other. Both systems interact through multiple coregulatory mechanisms, such as receptor heterodimerization and coregulation (e.g., S1P5, CB2, GPR55, PPARγ), shared metabolic enzymes (e.g., SphK1–2, LOXs), and integrated downstream effectors, particularly of TRPV1, as summarized in Figure 5. This interplay results in finely tuned, context-dependent modulation of bioactive lipids in cellular responses, with an impact on the regulation of inflammation, vascular function, neuronal activity, and insulin sensitivity.
Furthermore, the cannabis ingredient CBD demonstrates significant potential as a modulator of eCB and S1P endogenous systems. By modulating dioxygenase enzymes that metabolize eCBs and S1P—such as LOX, COX, and CYP isoforms—CBD further underscores the biochemical convergence of their pathways. Therefore, the activity of these enzymes represents a metabolic “checkpoint,” where the outputs of eCB and S1P signaling, along with those of CBD, are integrated and finely tuned.
On a final note, recent studies have revealed novel mechanisms of crosscommunication between bioactive lipid systems. In fact, an intriguing discovery has shown that the nuclear translocation of a truncated form of S1P2 induces PPARα degradation, which, as a consequence, blocks transcription of lipid catabolic genes (ref. 111). Moreover, a novel regulatory role of NAPE-PLD as an unexpected target of thiazide diuretics has been recently reported, pointing at it as a moonlighting enzyme (ref. 112). These findings further support the notion that our current understanding of lipid signaling is still in its infancy, and so far, only the tip of the iceberg has been scratched.
In conclusion, it seems apparent that eCB and S1P signaling pathways operate through interconnected networks of remarkable complexity. As yet, the biochemical crosstalk between these bioactive lipids remains incompletely understood, potentially limiting the therapeutic exploitation of these signals. Future strategies targeting the spatiotemporal dynamics of lipid transport—from intracellular trafficking to extracellular distribution—combined with selective receptor engagement may unlock novel therapeutic opportunities that current approaches have not fully realized.
Conflict of interest
The authors declare that they have no conflicts of interest with the contents of this article.
References
- M. Maccarrone, V. Di Marzo, J. Gertsch, U. Grether, A. Howlett, T. Hua. Goods and bads of the endocannabinoid system as a therapeutic target: lessons learned after 30 years. Pharmacol. Rev., 2023. [DOI]
- C. Weigel, J. Bellaci, S. Spiegel. Sphingosine-1-phosphate and its receptors in vascular endothelial and lymphatic barrier function. J. Biol. Chem., 2023
- S. Oddi, M.T. Fiorenza, M. Maccarrone. Endocannabinoid signaling in adult hippocampal neurogenesis: a mechanistic and integrated perspective. Prog. Lipid Res., 2023
- K. Tsuboi, T. Uyama, Y. Okamoto, N. Ueda. Endocannabinoids and related N-acylethanolamines: biological activities and metabolism. Inflamm. Regen., 2018. [DOI]
- W.A. Devane, L. Hanus, A. Breuer, R.G. Pertwee, L.A. Stevenson, G. Griffin. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science, 1992. [PubMed]
- R. Mechoulam, S. Ben-Shabat, L. Hanus, M. Ligumsky, N.E. Kaminski, A.R. Schatz. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol., 1995. [PubMed]
- P. Simankowicz, J. Stępniewska. The role of endocannabinoids in physiological processes and disease pathology: a comprehensive review. J. Clin. Med., 2025. [PubMed]
- S. Spiegel, S. Milstien. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol., 2003. [PubMed]
- N.N. Zhang, Desai, A. Olivera, T. Seki, G. Brooker, S. Spiegel. Sphingosine-1-phosphate, a novel lipid, involved in cellular proliferation. J. Cell Biol., 1991. [PubMed]
- T. Maula, I. Artetxe, P.-M. Grandell, J.P. Slotte. Importance of the sphingoid base length for the membrane properties of ceramides. Biophys. J., 2012. [PubMed]
- A. Cartier, T. Hla. Sphingosine 1-phosphate: lipid signaling in pathology and therapy. Science, 2019
- M. Maccarrone. Missing pieces to the endocannabinoid puzzle. Trends Mol. Med., 2020. [PubMed]
- M. Maceyka, K.B. Harikumar, S. Milstien, S. Spiegel. Sphingosine-1-phosphate signaling and its role in disease. Trends Cell Biol., 2012. [PubMed]
- M. Maccarrone, E. Dainese, S. Oddi. Intracellular trafficking of anandamide: new concepts for signaling. Trends Biochem. Sci., 2010. [PubMed]
- S. Oddi, F. Fezza, N. Pasquariello, C. De Simone, C. Rapino, E. Dainese. Evidence for the intracellular accumulation of anandamide in adiposomes. Cell. Mol. Life Sci. CMLS, 2008. [PubMed]
- M. Kaczocha, S. Haj-Dahmane. Mechanisms of endocannabinoid transport in the brain. Br. J. Pharmacol., 2022. [PubMed]
- S. Spiegel, M.A. Maczis, M. Maceyka, S. Milstien. New insights into functions of the sphingosine-1-phosphate transporter SPNS2. J. Lipid Res., 2019. [PubMed]
- L. Yu, L. He, B. Gan, R. Ti, Q. Xiao, X. Yang. Structural insights into sphingosine-1-phosphate receptor activation. Proc. Natl. Acad. Sci. U. S. A., 2022
- F. Zeng, A. Wade, K. Harbert, S. Patel, J.S. Holley, C.K. Dehghanpuor. Classical cannabinoid receptors as target in cancer-induced bone pain: a systematic review, meta-analysis and bioinformatics validation. Sci. Rep., 2024. [PubMed]
- F.A. Iannotti, C. Silvestri, E. Mazzarella, A. Martella, D. Calvigioni, F. Piscitelli. The endocannabinoid 2-AG controls skeletal muscle cell differentiation via CB1 receptor-dependent inhibition of Kv7 channels. Proc. Natl. Acad. Sci. U. S. A., 2014. [PubMed]
- S. Standoli, S. Pecchioli, D. Tortolani, C. Di Meo, F. Fanti, M. Sergi. The TRPV1 receptor is Up-Regulated by sphingosine 1-Phosphate and is implicated in the anandamide-dependent regulation of mitochondrial activity in C2C12 myoblasts. Int. J. Mol. Sci., 2022
- S. Standoli, C. Rapino, C. Di Meo, A. Rudowski, N. Kämpfer-Kolb, L.M. Volk. Sphingosine kinases at the intersection of pro-inflammatory LPS and anti-inflammatory endocannabinoid signaling in BV2 mouse microglia cells. Int. J. Mol. Sci., 2023. [DOI]
- M. Maccarrone. Need for methods to investigate endocannabinoid signaling. Methods Mol. Biol. Clifton NJ., 2023
- E.D. Mock, M. Mustafa, O. Gunduz-Cinar, R. Cinar, G.N. Petrie, V. Kantae. Discovery of a NAPE-PLD inhibitor that modulates emotional behavior in mice. Nat. Chem. Biol., 2020. [PubMed]
- N. Berger, T. van der Wel, B. Hirschmugl, T. Baernthaler, J. Gindlhuber, N. Fawzy. Inhibition of diacylglycerol lipase β modulates lipid and endocannabinoid levels in the ex vivo human placenta. Front. Endocrinol., 2023. [DOI]
- T. Bisogno, F. Howell, G. Williams, A. Minassi, M.G. Cascio, A. Ligresti. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol., 2003. [PubMed]
- L.D. Schurman, M.C. Carper, L.V. Moncayo, D. Ogasawara, K. Richardson, L. Yu. Diacylglycerol lipase-alpha regulates hippocampal-dependent learning and memory processes in mice. J. Neurosci., 2019. [PubMed]
- D. Leung, A. Saghatelian, G.M. Simon, B.F. Cravatt. Inactivation of N-acyl phosphatidylethanolamine phospholipase D reveals multiple mechanisms for the biosynthesis of endocannabinoids. Biochemistry, 2006. [PubMed]
- L.M. Mayo, E. Gauffin, G.N. Petrie, R. Tansey, R. Mazurka, C.J. Haggarty. The efficacy of elevating anandamide via inhibition of fatty acid amide hydrolase (FAAH) combined with internet-delivered cognitive behavioral therapy in the treatment of post-traumatic stress disorder: a randomized, placebo-controlled clinical trial. Neuropsychopharmacology, 2025. [DOI]
- M. Jiang, M.C.W. Huizenga, J.L. Wirt, J. Paloczi, A. Amedi, R.J.B.H.N. van den Berg. A monoacylglycerol lipase inhibitor showing therapeutic efficacy in mice without central side effects or dependence. Nat. Commun., 2023. [PubMed]
- M. Almada, B.M. Fonseca, C. Amaral, M. Diniz-da-Costa, G. Correia-da-Silva, N. Teixeira. Anandamide oxidative metabolism-induced endoplasmic reticulum stress and apoptosis. Apoptosis Int. J. Program Cell Death, 2017
- D.J. Hermanson, N.D. Hartley, J. Gamble-George, N. Brown, B.C. Shonesy, P.J. Kingsley. Substrate-selective COX-2 inhibition decreases anxiety via endocannabinoid activation. Nat. Neurosci., 2013. [PubMed]
- W.S. Edgemond, C.J. Hillard, J.R. Falck, C.S. Kearn, W.B. Campbell. Human platelets and polymorphonuclear leukocytes synthesize oxygenated derivatives of arachidonylethanolamide (Anandamide): their affinities for cannabinoid receptors and pathways of inactivation. Mol. Pharmacol., 1998. [PubMed]
- A.J. Hampson, W. Hill, M. Zan-Phillips, A. Makriyannis, E. Leung, R.M. Eglen. Anandamide hydroxylation by brain lipoxygenase:metabolite structures and potencies at the cannabinoid receptor. Portal Periód. CAPES, 1995. [DOI]
- N.T. Snider, A.M. Kornilov, U.M. Kent, P.F. Hollenberg. Anandamide metabolism by human liver and kidney microsomal cytochrome p450 enzymes to form hydroxyeicosatetraenoic and epoxyeicosatrienoic acid ethanolamides. J. Pharmacol. Exp. Ther., 2007. [PubMed]
- P. Urquhart, A. Nicolaou, D.F. Woodward. Endocannabinoids and their oxygenation by cyclo-oxygenases, lipoxygenases and other oxygenases. Biochim. Biophys. Acta, 2015. [PubMed]
- S.S.-J. Hu, H.B. Bradshaw, J.S.-C. Chen, B. Tan, J.M. Walker. Prostaglandin E2 glycerol ester, an endogenous COX-2 metabolite of 2-arachidonoylglycerol, induces hyperalgesia and modulates NFκB activity. Br. J. Pharmacol., 2008. [PubMed]
- S. Valdeolivas, M.R. Pazos, T. Bisogno, F. Piscitelli, F.A. Iannotti, M. Allarà. The inhibition of 2-arachidonoyl-glycerol (2-AG) biosynthesis, rather than enhancing striatal damage, protects striatal neurons from malonate-induced death: a potential role of cyclooxygenase-2-dependent metabolism of 2-AG. Cell Death Dis., 2013
- A.-S. Archambault, C. Turcotte, C. Martin, V. Provost, M.-C. Larose, C. Laprise. Comparison of eight 15-lipoxygenase (LO) inhibitors on the biosynthesis of 15-LO metabolites by human neutrophils and eosinophils. PLoS ONE, 2018
- K.R. Kozak, R.A. Gupta, J.S. Moody, C. Ji, W.E. Boeglin, R.N. DuBois. 15-Lipoxygenase metabolism of 2-Arachidonylglycerol: generation of a peroxisome proliferator-activated receptor α agonist. J. Biol. Chem., 2002. [PubMed]
- J.S. Moody, K.R. Kozak, C. Ji, L.J. Marnett. Selective oxygenation of the endocannabinoid 2-Arachidonylglycerol by leukocyte-type 12-Lipoxygenase. Biochemistry, 2000
- J.-K. Chen, J. Chen, J.D. Imig, S. Wei, D.L. Hachey, J.S. Guthi. Identification of novel endogenous cytochrome p450 arachidonate metabolites with high affinity for cannabinoid receptors. J. Biol. Chem., 2008. [PubMed]
- D.R. McDougle, A. Kambalyal, D.D. Meling, A. Das. Endocannabinoids anandamide and 2-Arachidonoylglycerol are substrates for human CYP2J2 epoxygenase. J. Pharmacol. Exp. Ther., 2014. [PubMed]
- C.A. Rouzer, L.J. Marnett. Endocannabinoid oxygenation by cyclooxygenases, lipoxygenases, and cytochromes P450: cross-talk between the eicosanoid and endocannabinoid signaling pathways. Chem. Rev., 2011. [PubMed]
- P.-R. Gan, R.-H. Wang, R. Deng, H. Wu, Y.-H. Bu, F.-Y. Chen. Geniposide inhibits SphK1 membrane targeting to restore macrophage polarization balance in collagen-induced arthritis mice. Eur. J. Pharmacol., 2022
- L. Gong, Y. Shen, S. Wang, X. Wang, H. Ji, X. Wu. Nuclear SPHK2/S1P induces oxidative stress and NLRP3 inflammasome activation via promoting p53 acetylation in lipopolysaccharide-induced acute lung injury. Cell Death Discov., 2023. [PubMed]
- D. Hatoum, N. Haddadi, Y. Lin, N.T. Nassif, E.M. McGowan. Mammalian sphingosine kinase (SphK) isoenzymes and isoform expression: challenges for SphK as an oncotarget. Oncotarget, 2017. [PubMed]
- F. Parveen, D. Bender, S.-H. Law, V.K. Mishra, C.-C. Chen, L.-Y. Ke. Role of ceramidases in sphingolipid metabolism and human diseases. Cells, 2019. [PubMed]
- A. Kuo, T. Hla. Regulation of cellular and systemic sphingolipid homeostasis. Nat. Rev. Mol. Cell Biol., 2024. [PubMed]
- X. Tang, M.G.K. Benesch, D.N. Brindley. Lipid phosphate phosphatases and their roles in mammalian physiology and pathology. J. Lipid Res., 2015. [PubMed]
- V.A. Blaho, T. Hla. An update on the biology of sphingosine 1-phosphate receptors. J. Lipid Res., 2014. [PubMed]
- D. Haspula, M.A. Clark. Cannabinoid receptors: an update on cell signaling, pathophysiological roles and therapeutic opportunities in neurological, cardiovascular, and inflammatory diseases. Int. J. Mol. Sci., 2020. [PubMed]
- D.A. Kendall, G.A. Yudowski. Cannabinoid receptors in the central nervous System: their signaling and roles in disease. Front. Cell. Neurosci., 2017. [DOI]
- M.P. McGinley, J.A. Cohen. Sphingosine 1-phosphate receptor modulators in multiple sclerosis and other conditions. Lancet, 2021. [PubMed]
- T. Claff, R. Ebenhoch, J.T. Kley, A. Magarkar, H. Nar, D. Weichert. Structural basis for lipid-mediated activation of G protein-coupled receptor GPR55. Nat. Commun., 2025. [PubMed]
- Y. Li, X. Chen, Y. Nie, Y. Tian, X. Xiao, F. Yang. Endocannabinoid activation of the TRPV1 ion channel is distinct from activation by capsaicin. J. Biol. Chem., 2021
- L. Bryan, T. Kordula, S. Spiegel, S. Milstien. Regulation and functions of sphingosine kinases in the brain. Biochim. Biophys. Acta BBA, 2008. [PubMed]
- V. Di Marzo, S. Petrosino. Endocannabinoids and the regulation of their levels in health and disease. Curr. Opin. Lipidol., 2007. [PubMed]
- S.M. Pitson. Regulation of sphingosine kinase and sphingolipid signaling. Trends Biochem. Sci., 2011. [PubMed]
- R.L. Proia, T. Hla. Emerging biology of sphingosine-1-phosphate: its role in pathogenesis and therapy. J. Clin. Invest., 2015. [PubMed]
- C.J. Fowler, P. Doherty, S.P.H. Alexander. Endocannabinoid turnover. Adv. Pharmacol. San Diego Calif., 2017
- Y. Hu, K. Dai. Sphingolipid Metabolism and Metabolic Disease, 2022
- A. Huwiler, J. Pfeilschifter. Altering the sphingosine-1-phosphate/ceramide balance: a promising approach for tumor therapy. Curr. Pharm. Des., 2006. [PubMed]
- J. Armistead, S. Höpfl, P. Goldhausen, A. Müller-Hartmann, E. Fahle, J. Hatzold. A sphingolipid rheostat controls apoptosis versus apical cell extrusion as alternative tumour-suppressive mechanisms. Cell Death Dis., 2024. [PubMed]
- K. Takabe, S.W. Paugh, S. Milstien, S. Spiegel. “Inside-out” signaling of sphingosine-1-phosphate: therapeutic targets. Pharmacol. Rev., 2008. [PubMed]
- M. Lombardi, F. Scaroni, M. Gabrielli, S. Raffaele, E. Bonfanti, F. Filipello. Extracellular vesicles released by microglia and macrophages carry endocannabinoids which foster oligodendrocyte differentiation. Front. Immunol., 2024
- C. Verderio, M. Gabrielli, P. Giussani. Role of sphingolipids in the biogenesis and biological activity of extracellular vesicles. J. Lipid Res., 2018. [PubMed]
- M. Gabrielli, N. Battista, L. Riganti, I. Prada, F. Antonucci, L. Cantone. Active endocannabinoids are secreted on extracellular membrane vesicles. EMBO Rep., 2015. [PubMed]
- C.-Y. Liao, M.J. Song, Y. Gao, A.S. Mauer, A. Revzin, H. Malhi. Hepatocyte-Derived lipotoxic extracellular vesicle sphingosine 1-Phosphate induces macrophage chemotaxis. Front. Immunol., 2018. [DOI]
- S. Rigogliuso, C. Donati, D. Cassarà, S. Taverna, M. Salamone, P. Bruni. An active form of sphingosine Kinase-1 is released in the extracellular medium as component of membrane vesicles shed by two human tumor cell lines. J. Oncol., 2010
- R. Wang, Q. Ding, U. Yaqoob, T.M. de Assuncao, V.K. Verma, P. Hirsova. Exosome adherence and internalization by hepatic stellate cells triggers sphingosine 1-Phosphate-dependent migration. J. Biol. Chem., 2015. [PubMed]
- H. Hong, B. Yoon, S. Ghil. Interactions between lysophosphatidylinositol receptor GPR55 and sphingosine-1-phosphate receptor S1P5 in live cells. Biochem. Biophys. Res. Commun., 2021. [PubMed]
- Y. Kim, S. Ghil. Negative regulation of cannabinoid receptor 2-induced tumorigenic effect by sphingosine-1-phosphate receptor 5 activation. Oncol. Rep., 2025. [PubMed]
- F.A. Iannotti, R.M. Vitale. The endocannabinoid System and PPARs: focus on their signalling crosstalk, action and transcriptional regulation. Cells, 2021. [PubMed]
- K.A. Parham, J.R. Zebol, K.L. Tooley, W.Y. Sun, L.M. Moldenhauer, M.P. Cockshell. Sphingosine 1-phosphate is a ligand for peroxisome proliferator-activated receptor-γ that regulates neoangiogenesis. FASEB J., 2015. [PubMed]
- P. Raman, B.L.F. Kaplan, N.E. Kaminski. 15-Deoxy-Δ12,14-Prostaglandin J2-Glycerol, a putative metabolite of 2-Arachidonyl glycerol and a peroxisome proliferator-activated receptor γ ligand, modulates nuclear factor of activated T cells. J. Pharmacol. Exp. Ther., 2012. [PubMed]
- A. Lenman, C.J. Fowler. Interaction of ligands for the peroxisome proliferator-activated receptor gamma with the endocannabinoid system. Br. J. Pharmacol., 2007. [PubMed]
- B. Hempel, M. Crissman, S. Pari, B. Klein, G.-H. Bi, H. Alton. PPARα and PPARγ are expressed in midbrain dopamine neurons and modulate dopamine- and cannabinoid-mediated behavior in mice. Mol. Psychiatry, 2023. [PubMed]
- J.H. Yu, S.J. Song, A. Kim, Y. Choi, J.W. Seok, H.J. Kim. Suppression of PPARγ-mediated monoacylglycerol O-acyltransferase 1 expression ameliorates alcoholic hepatic steatosis. Sci. Rep., 2016
- K.M. Mair, E. Robinson, K.A. Kane, S. Pyne, R.R. Brett, N.J. Pyne. Interaction between anandamide and sphingosine-1-phosphate in mediating vasorelaxation in rat coronary artery. Br. J. Pharmacol., 2010. [PubMed]
- F.H. Greig, K. Nather, M.D. Ballantyne, Z.H. Kazi, H. Alganga, M.-A. Ewart. Requirement for sphingosine kinase 1 in mediating phase 1 of the hypotensive response to anandamide in the anaesthetised mouse. Eur. J. Pharmacol., 2019. [PubMed]
- B. Wang, L. Wu, J. Chen, L. Dong, C. Chen, Z. Wen. Metabolism pathways of arachidonic acids: mechanisms and potential therapeutic targets | Signal Transduction and Targeted Therapy. Signal. Transduct. Target. Ther., 2021. [DOI]
- J. Fettel, B. Kühn, N.A. Guillen, D. Sürün, M. Peters, R. Bauer. Sphingosine-1-phosphate (S1P) induces potent anti-inflammatory effects in vitro and in vivo by S1P receptor 4-mediated suppression of 5-lipoxygenase activity. FASEB J., 2019. [PubMed]
- X. Wang, W. Guo, X. Shi, Y. Chen, Y. Yu, B. Du. S1PR1/S1PR3-YAP signaling and S1P-ALOX15 signaling contribute to an aggressive behavior in obesity-lymphoma. J. Exp. Clin. Cancer Res., 2023. [PubMed]
- L.-Y. Chen, G. Woszczek, S. Nagineni, C. Logun, J.H. Shelhamer. Cytosolic phospholipase A2α activation induced by S1P is mediated by the S1P3 receptor in lung epithelial cells. Am. J. Physiol., 2008
- A. Völzke, A. Koch, D. Meyer Zu Heringdorf, A. Huwiler, J. Pfeilschifter. Sphingosine 1-phosphate (S1P) induces COX-2 expression and PGE2 formation via S1P receptor 2 in renal mesangial cells. Biochim. Biophys. Acta, 2014. [PubMed]
- N. Undurti Das. Essential fatty acids and their metabolites in the pathobiology of inflammation and its resolution. Biomolecules, 2021. [PubMed]
- N. Brunkhorst-Kanaan, S. Trautmann, Y. Schreiber, D. Thomas, S. Kittel-Schneider, R. Gurke. Sphingolipid and endocannabinoid profiles in adult attention deficit hyperactivity disorder. Biomedicines, 2021. [PubMed]
- C. Fischer, D. Thomas, R. Gurke, I. Tegeder. Brain region specific regulation of anandamide (down) and sphingosine-1-phosphate (up) in association with anxiety (AEA) and resilience (S1P) in a mouse model of chronic unpredictable mild stress. Pflugers Arch., 2024. [PubMed]
- H. Neb, V. Roth, J. Roos, T. Bauer, A. Urbschat, U. Heinicke. Analysis of fatty acid-derived lipids in critically ill patients after cardiac surgery yields novel pathophysiologically relevant mediators with possible relevance for systemic inflammatory reactions. Front. Immunol., 2024
- R.Z. Hill, T. Morita, R.B. Brem, D.M. Bautista. S1PR3 mediates itch and pain via distinct TRP channel-dependent pathways. J. Neurosci., 2018. [PubMed]
- H. Kittaka, J. DeBrecht, S.K. Mishra. Differential contribution of sensory transient receptor potential channels in response to the bioactive lipid sphingosine-1-phosphate. Mol. Pain, 2020. [DOI]
- M. Langeslag, C.E. Constantin, M. Andratsch, S. Quarta, N. Mair, M. Kress. Oncostatin M induces heat hypersensitivity by gp130-Dependent sensitization of TRPV1 in sensory neurons. Mol. Pain, 2011
- D. Friedman, J.A. French, M. Maccarrone. Safety, efficacy, and mechanisms of action of cannabinoids in neurological disorders. Lancet Neurol., 2019. [PubMed]
- T. Bosquez-Berger, S. Wilson, C. Iliopoulos-Tsoutsouvas, S. Jiang, J. Wager-Miller, S.P. Nikas. Differential enantiomer-specific signaling of cannabidiol at CB1 receptors. Mol. Pharmacol., 2022. [PubMed]
- L.O. Hanuš, S. Tchilibon, D.E. Ponde, A. Breuer, E. Fride, R. Mechoulam. Enantiomeric cannabidiol derivatives: synthesis and binding to cannabinoid receptors. Org. Biomol. Chem., 2005. [PubMed]
- L.O. Hanuš, S.M. Meyer, E. Muñoz, O. Taglialatela-Scafati, G. Appendino. Phytocannabinoids: a unified critical inventory. Nat. Prod. Rep., 2016. [PubMed]
- R.G. Pertwee. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. Br. J. Pharmacol., 2008. [PubMed]
- R.B. Laprairie, A.M. Bagher, M.E.M. Kelly, E.M. Denovan-Wright. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol., 2015. [PubMed]
- T. Bisogno, L. Hanus, L. De Petrocellis, S. Tchilibon, D.E. Ponde, I. Brandi. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol., 2001. [PubMed]
- E.J. Carrier, J.A. Auchampach, C.J. Hillard. Inhibition of an equilibrative nucleoside transporter by cannabidiol: a mechanism of cannabinoid immunosuppression. Proc. Natl. Acad. Sci. U. S. A., 2006. [PubMed]
- A.B. Sonego, D.S. Prado, F.S. Guimarães. PPARγ receptors are involved in the effects of cannabidiol on orofacial dyskinesia and cognitive dysfunction induced by typical antipsychotic in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry, 2021
- M. Schouten, S. Dalle, D. Mantini, K. Koppo. Cannabidiol and brain function: current knowledge and future perspectives. Front. Pharmacol., 2023
- P. Massi, M. Valenti, A. Vaccani, V. Gasperi, G. Perletti, E. Marras. 5-Lipoxygenase and anandamide hydrolase (FAAH) mediate the antitumor activity of cannabidiol, a non-psychoactive cannabinoid. J. Neurochem., 2008. [PubMed]
- V. Di Marzo, F. Piscitelli. The endocannabinoid System and its modulation by phytocannabinoids. Neurotherapeutics, 2015. [PubMed]
- P. Bielawiec, E. Harasim-Symbor, A. Chabowski. Phytocannabinoids: useful drugs for the treatment of obesity? Special focus on cannabidiol. Front. Endocrinol., 2020
- T. Charytoniuk, K. Sztolsztener, E. Harasim-Symbor, K. Berk, A. Chabowski, K. Konstantynowicz-Nowicka. Cannabidiol – a phytocannabinoid that widely affects sphingolipid metabolism under conditions of brain insulin resistance. Biomed. Pharmacother. Biomedecine Pharmacother., 2021
- W. Bzdęga, P.F. Kurzyna, E. Harasim-Symbor, A. Hołownia, A. Chabowski, K. Konstantynowicz-Nowicka. How does CBG administration affect sphingolipid deposition in the liver of insulin-resistant rats?. Nutrients, 2023. [PubMed]
- S. Bansal, M.F. Paine, J.D. Unadkat. Comprehensive predictions of cytochrome P450 (P450)-Mediated in vivo cannabinoid-drug interactions based on reversible and time-dependent P450 inhibition in human liver microsomes. Drug Metab. Dispos., 2022. [PubMed]
- L.K. Peltner, L. Gluthmann, F. Börner, S. Pace, R.K. Hoffstetter, C. Kretzer. Cannabidiol acts as molecular switch in innate immune cells to promote the biosynthesis of inflammation-resolving lipid mediators. Cell Chem. Biol., 2023. [PubMed]
- R.-R. Miao, M.-Y. Tan, H.-B. Shao, Z.-M. Sun, S. Zhan, W.-S. Liang. Conjugated bile acids promote metabolic dysfunction-associated steatotic liver disease through inducing nuclear translocation of sphingosine-1-phosphate receptor 2 to disrupt peroxisome proliferator-activated receptor alpha. Cell Commun. Signal. CCS., 2025. [PubMed]
- S. Chiarugi, F. Margheriti, V. De Lorenzi, E. Martino, E.G. Margheritis, A. Moscardini. NAPE-PLD is target of thiazide diuretics. Cell Chem. Biol., 2025. [PubMed]
- S.M.K. Rahman, Z. Hussain, K. Morito, N. Takahashi, M.M. Sikder, T. Tanaka. Formation of N-acyl-phosphatidylethanolamines by cytosolic phospholipase A2ε in an ex vivo murine model of brain ischemia. Biochim. Biophys. Acta Mol. Cell Biol. Lipids, 2022
- E.D. Mock, W.P.F. Driever, M. van der Stelt. Fluorescence-Based NAPE-PLD activity assay. Methods Mol. Biol. Clifton NJ., 2023
- J. Liu, L. Wang, J. Harvey-White, D. Osei-Hyiaman, R. Razdan, Q. Gong. A biosynthetic pathway for anandamide. Proc. Natl. Acad. Sci. U. S. A., 2006. [PubMed]
- J. Liu, L. Wang, J. Harvey-White, B.X. Huang, H.-Y. Kim, S. Luquet. Multiple pathways involved in the biosynthesis of anandamide. Neuropharmacology, 2008. [PubMed]
- Y. Okamoto, J. Morishita, K. Tsuboi, T. Tonai, N. Ueda. Molecular characterization of a phospholipase D generating anandamide and its congeners. J. Biol. Chem., 2004. [PubMed]
- L. Liang, R. Takamiya, Y. Miki, K. Heike, Y. Taketomi, N. Sugimoto. Group IVE cytosolic phospholipase A2 limits psoriatic inflammation by mobilizing the anti-inflammatory lipid N-acylethanolamine. FASEB J., 2022
- J. Fu, G. Bottegoni, O. Sasso, R. Bertorelli, W. Rocchia, M. Masetti. A catalytically silent FAAH-1 variant drives anandamide transport in neurons. Nat. Neurosci., 2011. [PubMed]
- Y. Fotio, A. Mabou Tagne, E. Squire, H. Lee, C.M. Phillips, K. Chang. NAAA-regulated lipid signaling in monocytes controls the induction of hyperalgesic priming in mice. Nat. Commun., 2024. [PubMed]
- D. Piomelli, L. Scalvini, Y. Fotio, A. Lodola, G. Spadoni, G. Tarzia. N-Acylethanolamine acid amidase (NAAA): structure, function, and inhibition. J. Med. Chem., 2020. [PubMed]
- N. Murataeva, A. Straiker, K. Mackie. Parsing the players: 2-arachidonoylglycerol synthesis and degradation in the CNS. Br. J. Pharmacol., 2014. [PubMed]
- G. Bononi, T. Tuccinardi, F. Rizzolio, C. Granchi. α/β-Hydrolase Domain (ABHD) inhibitors as new potential therapeutic options against lipid-related diseases. J. Med. Chem., 2021. [PubMed]
- V. Morozova, D. Pellegata, R.-P. Charles, J. Gertsch. Carboxylesterase 1-mediated endocannabinoid metabolism in skin: role in melanoma progression in BRafV600E/Pten−/− mice. Cancer Metab., 2025. [PubMed]
- B. Ehrlich, M. Patel, X. Zheng, M. Glass. Canonical cannabinoid receptors. Curr. Top. Behav. Neurosci., 2025. [DOI]
- B. Lutz. Neurobiology of cannabinoid receptor signaling. Dialogues Clin. Neurosci., 2020. [PubMed]
- L. Ye, Z. Cao, W. Wang, N. Zhou. New insights in cannabinoid receptor structure and signaling. Curr. Mol. Pharmacol., 2019. [PubMed]
- K. Ghosh, G.-F. Zhang, H. Chen, S.-R. Chen, H.-L. Pan. Cannabinoid CB2 receptors are upregulated via bivalent histone modifications and control primary afferent input to the spinal cord in neuropathic pain. J. Biol. Chem., 2022
- J.D. Hill, V. Zuluaga-Ramirez, S. Gajghate, M. Winfield, Y. Persidsky. Activation of GPR55 increases neural stem cell proliferation and promotes early adult hippocampal neurogenesis. Br. J. Pharmacol., 2018. [PubMed]
- A. Irving, G. Abdulrazzaq, S.L.F. Chan, J. Penman, J. Harvey, S.P.H. Alexander. Cannabinoid receptor-related orphan G protein-coupled receptors. Adv. Pharmacol. San Diego Calif., 2017
- C. Muller, D.L. Lynch, D.P. Hurst, P.H. Reggio. A closer look at anandamide interaction with TRPV1. Front. Mol. Biosci., 2020. [PubMed]
- S.E. O’Sullivan. An update on PPAR activation by cannabinoids. Br. J. Pharmacol., 2016. [PubMed]
- S.E. O’Sullivan, D.A. Kendall. Cannabinoid activation of peroxisome proliferator-activated receptors: potential for modulation of inflammatory disease. Immunobiology., 2010. [PubMed]
- C. Fowler. Transport of endocannabinoids across the plasma membrane and within the cell. FEBS J., 2013. [DOI]
- A. Chicca, J. Marazzi, S. Nicolussi, J. Gertsch. Evidence for bidirectional endocannabinoid transport across cell membranes. J. Biol. Chem., 2012. [PubMed]
- P. Mäder, R. Bartholomäus, S. Nicolussi, A. Baumann, M. Weis, A. Chicca. Synthesis and biological evaluation of endocannabinoid uptake inhibitors derived from WOBE437. ChemMedChem, 2021. [PubMed]
- W.T. Berger, B.P. Ralph, M. Kaczocha, J. Sun, T.E. Balius, R.C. Rizzo. Targeting fatty acid binding protein (FABP) anandamide transporters – a novel strategy for development of anti-inflammatory and anti-nociceptive drugs. PLoS One, 2012
- F. Ciaramellano, F. Fanti, L. Scipioni, M. Maccarrone, S. Oddi. Endocannabinoid metabolism and transport as drug targets. Methods Mol. Biol., 2023. [PubMed]
- M.W. Elmes, M. Kaczocha, W.T. Berger, K. Leung, B.P. Ralph, L. Wang. Fatty acid-binding proteins (FABPs) are intracellular carriers for Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD). J. Biol. Chem., 2015. [PubMed]
- S.-A. Lee, K.J.Z. Yang, P.-J. Brun, J.A. Silvaroli, J.J. Yuen, I. Shmarakov. Retinol-binding protein 2 (RBP2) binds monoacylglycerols and modulates gut endocrine signaling and body weight. Sci. Adv., 2020
- E.S. Liedhegner, C.D. Vogt, D.S. Sem, C.W. Cunningham, C.J. Hillard. Sterol carrier protein-2: binding protein for endocannabinoids. Mol. Neurobiol., 2014. [PubMed]
- S. Oddi, F. Fezza, N. Pasquariello, A. D’Agostino, G. Catanzaro, C. De Simone. Molecular identification of albumin and Hsp70 as cytosolic anandamide-binding proteins. Chem. Biol., 2009. [PubMed]
- J. Plau, M. Golczak, J. Paik, R.M. Calderon, W.S. Blaner. Retinol-binding protein 2 (RBP2): more than just dietary retinoid uptake. Biochim. Biophys. Acta Mol. Cell Biol. Lipids, 2022
- P. Williams, S. Tulke, E. Ilegems, P.-O. Berggren, C. Broberger. Expression of nucleobindin 1 (NUCB1) in pancreatic islets and other endocrine tissues. Cell Tissue Res., 2014. [PubMed]
- N.T. Snider, V.J. Walker, P.F. Hollenberg. Oxidation of the endogenous Cannabinoid Arachidonoyl ethanolamide by the cytochrome P450 monooxygenases: physiological and pharmacological implications. Pharmacol. Rev., 2010. [PubMed]
- G. Bénard, F. Massa, N. Puente, J. Lourenço, L. Bellocchio, E. Soria-Gómez. Mitochondrial CB1 receptors regulate neuronal energy metabolism. Nat. Neurosci., 2012. [PubMed]
- S. Melser, A.C. Pagano Zottola, R. Serrat, N. Puente, P. Grandes, G. Marsicano. Functional analysis of mitochondrial CB1 cannabinoid receptors (mtCB1) in the brain. Methods Enzymol., 2017. [PubMed]
- I.N. Bojesen, H.S. Hansen. Binding of anandamide to bovine serum albumin. J. Lipid Res., 2003. [PubMed]
- E. Albarran, Y. Sun, Y. Liu, K. Raju, A. Dong, Y. Li. Postsynaptic synucleins mediate endocannabinoid signaling. Nat. Neurosci., 2023. [PubMed]
- S. Spiegel, S. Milstien. Functions of the multifaceted family of sphingosine kinases and some close relatives. J. Biol. Chem., 2007. [PubMed]
- F. Cencetti, G. Bruno, C. Bernacchioni, L. Japtok, E. Puliti, C. Donati. Sphingosine 1-phosphate lyase blockade elicits myogenic differentiation of murine myoblasts acting via Spns2/S1P2 receptor axis. Biochim. Biophys. Acta BBA, 2020
- Y.A. Hannun, L.M. Obeid. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol., 2018. [PubMed]
- A.H. Merrill. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem. Rev., 2011. [PubMed]
- K. Kitatani, J. Idkowiak-Baldys, Y.A. Hannun. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell. Signal., 2008. [PubMed]
- K.R. Johnson, K.Y. Johnson, K.P. Becker, J. Bielawski, C. Mao, L.M. Obeid. Role of human Sphingosine-1-phosphate phosphatase 1 in the regulation of Intra- and extracellular Sphingosine-1-phosphate levels and cell viability. J. Biol. Chem., 2003. [PubMed]
- M. Maceyka, H. Sankala, N.C. Hait, H. Le Stunff, H. Liu, R. Toman. SphK1 and SphK2, sphingosine kinase isoenzymes with opposing functions in sphingolipid metabolism. J. Biol. Chem., 2005. [PubMed]
- M. Chandrakanthan, T.Q. Nguyen, Z. Hasan, S. Muralidharan, T.M. Vu, A.W.L. Li. Deletion of Mfsd2b impairs thrombotic functions of platelets. Nat. Commun., 2021. [PubMed]
- S. Fukuhara, S. Simmons, S. Kawamura, A. Inoue, Y. Orba, T. Tokudome. The sphingosine-1-phosphate transporter Spns2 expressed on endothelial cells regulates lymphocyte trafficking in mice. J. Clin. Invest., 2012. [PubMed]
- Y. Hisano, N. Kobayashi, A. Yamaguchi, T. Nishi. Mouse SPNS2 functions as a sphingosine-1-phosphate transporter in vascular endothelial cells. PLoS One, 2012
- K. Venkataraman, Y.-M. Lee, J. Michaud, S. Thangada, Y. Ai, H.L. Bonkovsky. Vascular endothelium as a contributor of plasma sphingosine 1-phosphate. Circ. Res., 2008. [PubMed]
- K.M. Argraves, P.J. Gazzolo, E.M. Groh, B.A. Wilkerson, B.S. Matsuura, W.O. Twal. High density lipoprotein-associated sphingosine 1-Phosphate promotes endothelial barrier function. J. Biol. Chem., 2008. [PubMed]
- V.A. Blaho, S. Galvani, E. Engelbrecht, C. Liu, S.L. Swendeman, M. Kono. HDL-bound sphingosine-1-phosphate restrains lymphopoiesis and neuroinflammation. Nature, 2015. [PubMed]
- C. Christoffersen, H. Obinata, S.B. Kumaraswamy, S. Galvani, J. Ahnström, M. Sevvana. Endothelium-protective sphingosine-1-phosphate provided by HDL-associated apolipoprotein M. Proc. Natl. Acad. Sci. U. S. A., 2011. [PubMed]
- N. Kobayashi, S. Kawasaki-Nishi, M. Otsuka, Y. Hisano, A. Yamaguchi, T. Nishi. MFSD2B is a sphingosine 1-phosphate transporter in erythroid cells. Sci. Rep., 2018. [PubMed]
- B.A. Wilkerson, G.D. Grass, S.B. Wing, W.S. Argraves, K.M. Argraves. Sphingosine 1-phosphate (S1P) carrier-dependent regulation of endothelial barrier: high density lipoprotein (HDL)-S1P prolongs endothelial barrier enhancement as compared with albumin-S1P via effects on levels, trafficking, and signaling of S1P1. J. Biol. Chem., 2012. [PubMed]
- J. Rivera, R.L. Proia, A. Olivera. The alliance of sphingosine-1-phosphate and its receptors in immunity. Nat. Rev. Immunol., 2008. [PubMed]
- H. Rosen, R.C. Stevens, M. Hanson, E. Roberts, M.B.A. Oldstone. Sphingosine-1-phosphate and its receptors: structure, signaling, and influence. Annu. Rev. Biochem., 2013. [PubMed]
- C. O’Sullivan, K.K. Dev. The structure and function of the S1P1 receptor. Trends Pharmacol. Sci., 2013. [PubMed]
- X. Wang, S. Chen, H. Xiang, X. Wang, J. Xiao, S. Zhao. S1PR2/RhoA/ROCK1 pathway promotes inflammatory bowel disease by inducing intestinal vascular endothelial barrier damage and M1 macrophage polarization. Biochem. Pharmacol., 2022
- A. Bigi, R. Cascella, G. Fani, C. Bernacchioni, F. Cencetti, P. Bruni. Sphingosine 1-phosphate attenuates neuronal dysfunction induced by amyloid-β oligomers through endocytic internalization of NMDA receptors. FEBS J., 2023. [PubMed]
- N.C. Hait, J. Allegood, M. Maceyka, G.M. Strub, K.B. Harikumar, S.K. Singh. Regulation of histone acetylation in the nucleus by sphingosine-1-phosphate. Science, 2009. [PubMed]
- G.M. Strub, M. Paillard, J. Liang, L. Gomez, J.C. Allegood, N.C. Hait. Sphingosine-1-phosphate produced by sphingosine kinase 2 in mitochondria interacts with prohibitin 2 to regulate complex IV assembly and respiration. FASEB J., 2011. [PubMed]
- S. Panneer Selvam, R.M. De Palma, J.J. Oaks, N. Oleinik, Y.K. Peterson, R.V. Stahelin. Binding of the sphingolipid S1P to hTERT stabilizes telomerase at the nuclear periphery by allosterically mimicking protein phosphorylation. Sci. Signal., 2015
- N. Thomas, N.H. Schröder, M.K. Nowak, P. Wollnitzke, S. Ghaderi, K. von Wnuck Lipinski. Sphingosine-1-phosphate suppresses GLUT activity through PP2A and counteracts hyperglycemia in diabetic red blood cells. Nat. Commun., 2023. [PubMed]
- T. Kajimoto, A.D. Caliman, I.S. Tobias, T. Okada, C.A. Pilo, A.-A.N. Van. Activation of atypical protein kinase C by sphingosine 1-phosphate revealed by an aPKC-specific activity reporter. Sci. Signal., 2019
- S. Weske, M.K. Nowak, A. Zaufel, L. Esser, C. Peter, L. Walz. Intracellular Sphingosine-1-Phosphate induces lipolysis through direct activation of protein kinase C zeta. FASEB J., 2025
- T. Kajimoto, T. Okada, H. Yu, S.K. Goparaju, S. Jahangeer, S. Nakamura. Involvement of Sphingosine-1-Phosphate in glutamate secretion in hippocampal neurons. Mol. Cell. Biol., 2007. [PubMed]
- T. Kanno, T. Nishizaki. Endogenous sphingosine 1-phosphate regulates spontaneous glutamate release from mossy fiber terminals via S1P(3) receptors. Life Sci., 2011. [PubMed]
- L. Riganti, F. Antonucci, M. Gabrielli, I. Prada, P. Giussani, P. Viani. Sphingosine-1-Phosphate (S1P) impacts presynaptic functions by regulating synapsin I localization in the presynaptic compartment. J. Neurosci., 2016. [PubMed]
- C.C. Wang, J.R. Kuo, S.J. Wang. Fingolimod inhibits glutamate release through activation of S1P1 receptors and the G protein βγ subunit-dependent pathway in rat cerebrocortical nerve terminals. Neuropharmacology, 2021
- G. Gerdeman, D.M. Lovinger. CB1 Cannabinoid receptor inhibits synaptic release of glutamate in rat dorsolateral striatum. J. Neurophysiol., 2001. [PubMed]
- G.G. Szabó, N. Lenkey, N. Holderith, T. Andrási, Z. Nusser, N. Hájos. Presynaptic calcium channel inhibition underlies CB1 cannabinoid receptor-mediated suppression of GABA release. J. Neurosci., 2014. [PubMed]
- A.E. Chávez, C.Q. Chiu, P.E. Castillo. TRPV1 activation by endogenous anandamide triggers postsynaptic long-term depression in dentate gyrus. Nat. Neurosci., 2010. [PubMed]
- S. Thangada, K.M. Khanna, V.A. Blaho, M.L. Oo, D.-S. Im, C. Guo. Cell-surface residence of sphingosine 1-phosphate receptor 1 on lymphocytes determines lymphocyte egress kinetics. J. Exp. Med., 2010. [PubMed]
- S.S. Dusaban, J. Chun, H. Rosen, N.H. Purcell, J.H. Brown. Sphingosine 1-phosphate receptor 3 and RhoA signaling mediate inflammatory gene expression in astrocytes. J. Neuroinflammation, 2017. [PubMed]
- G. Zhang, L. Yang, G.S. Kim, K. Ryan, S. Lu, R.K. O’Donnell. Critical role of sphingosine-1-phosphate receptor 2 (S1PR2) in acute vascular inflammation. Blood, 2013. [PubMed]
- A.P. Young, E.M. Denovan-Wright. The dynamic role of microglia and the endocannabinoid System in neuroinflammation. Front. Pharmacol., 2022
- C. Turcotte, M.-R. Blanchet, M. Laviolette, N. Flamand. The CB2 receptor and its role as a regulator of inflammation. Cell. Mol. Life Sci. CMLS, 2016. [PubMed]
- H. Sumida, E. Lu, H. Chen, Q. Yang, K. Mackie, J.G. Cyster. GPR55 regulates intraepithelial lymphocyte migration dynamics and susceptibility to intestinal damage. Sci. Immunol., 2017
- C. Bernacchioni, R. Squecco, T. Gamberi, V. Ghini, F. Schumacher, M. Mannelli. S1P signalling axis is necessary for adiponectin-directed regulation of electrophysiological properties and oxidative metabolism in C2C12 myotubes. Cells, 2022. [PubMed]
- A. Botta, Y. Liu, S. Wannaiampikul, R. Tungtrongchitr, K. Dadson, T.-S. Park. An adiponectin-S1P axis protects against lipid induced insulin resistance and cardiomyocyte cell death via reduction of oxidative stress. Nutr. Metab., 2019
- I. Matias, M.-P. Gonthier, P. Orlando, V. Martiadis, L. De Petrocellis, C. Cervino. Regulation, function, and dysregulation of endocannabinoids in models of adipose and beta-pancreatic cells and in obesity and hyperglycemia. J. Clin. Endocrinol. Metab., 2006. [PubMed]
- D. Osei-Hyiaman, M. DePetrillo, P. Pacher, J. Liu, S. Radaeva, S. Bátkai. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J. Clin. Invest., 2005. [PubMed]
- Q. Wei, J.H. Lee, C.-S. Wu, Q.S. Zang, S. Guo, H.-C. Lu. Metabolic and inflammatory functions of cannabinoid receptor type 1 are differentially modulated by adiponectin. World J. Diabetes, 2021. [PubMed]
- H.M. Hashiesh, C. Sharma, S.N. Goyal, N.K. Jha, S. Ojha. Pharmacological properties, therapeutic potential and molecular mechanisms of JWH133, a CB2 receptor-selective agonist. Front. Pharmacol., 2021
- P. Shafiei-Jahani, S. Yan, M.H. Kazemi, X. Li, A. Akbari, K. Sakano. CB2 stimulation of adipose resident ILC2s orchestrates immune balance and ameliorates type 2 diabetes mellitus. Cell Rep., 2024
- B. Jung, H. Obinata, S. Galvani, K. Mendelson, B. Ding, A. Skoura. Flow-regulated endothelial S1P receptor-1 signaling sustains vascular development. Dev. Cell., 2012. [PubMed]
- T. Sanchez, A. Skoura, M.T. Wu, B. Casserly, E.O. Harrington, T. Hla. Induction of vascular permeability by the sphingosine-1-phosphate receptor-2 (S1P2R) and its downstream effectors ROCK and PTEN. Arterioscler. Thromb. Vasc. Biol., 2007. [PubMed]
- B.S. Yung, C.S. Brand, S.Y. Xiang, C.B.B. Gray, C.K. Means, H. Rosen. Selective coupling of the S1P3 receptor subtype to S1P-mediated RhoA activation and cardioprotection. J. Mol. Cell. Cardiol., 2017. [PubMed]
- S. Bátkai, P. Mukhopadhyay, J. Harvey-White, R. Kechrid, P. Pacher, G. Kunos. Endocannabinoids acting at CB1 receptors mediate the cardiac contractile dysfunction in vivo in cirrhotic rats. Am. J. Physiol. Heart Circ. Physiol., 2007. [PubMed]
- C. González, E. Herradón, R. Abalo, G. Vera, B.G. Pérez-Nievas, J.C. Leza. Cannabinoid/agonist WIN 55,212-2 reduces cardiac ischaemia–reperfusion injury in Zucker diabetic fatty rats: role of CB2 receptors and iNOS/eNOS. Diabetes Metab. Res. Rev., 2011. [PubMed]
- Q. Li, F. Wang, Y.-M. Zhang, J.-J. Zhou, Y. Zhang. Activation of cannabinoid type 2 receptor by JWH133 protects heart against ischemia/reperfusion-induced apoptosis. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol., 2013
- S.A. More, R.S. Deore, H.D. Pawar, C. Sharma, K.T. Nakhate, S.S. Rathod. CB2 cannabinoid receptor as a potential target in myocardial infarction: exploration of molecular pathogenesis and therapeutic strategies. Int. J. Mol. Sci., 2024. [PubMed]
