Cannabidiol attenuates precuneus activation during appetitive cue exposure in individuals with alcohol use disorder
Abstract
Supplementary Information:
The online version contains supplementary material available at 10.1007/s00406-025-01983-4.
Article type: Research Article
Keywords: Cannabidiol, Neuroimaging, Cue reactivity, Alcohol use disorder, Pharmacotherapy
Affiliations: https://ror.org/0384j8v12grid.1013.30000 0004 1936 834XSpecialty of Addiction Medicine, Sydney Medical School, Faculty of Medicine and Health, University of Sydney, Sydney, NSW Australia; https://ror.org/05gpvde20grid.413249.90000 0004 0385 0051Edith Collins Centre for Translational Research (Alcohol, Drugs & Toxicology), Royal Prince Alfred Hospital, Sydney Local Health District, Sydney, NSW Australia; https://ror.org/0384j8v12grid.1013.30000 0004 1936 834XLambert Initiative for Cannabinoid Therapeutics, University of Sydney, Sydney, NSW Australia; https://ror.org/0384j8v12grid.1013.30000 0004 1936 834XFaculty of Science, School of Psychology, University of Sydney, Sydney, NSW Australia
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s00406-025-01983-4 | PubMed: 40102270 | PMC: PMC12589260
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (810 KB)
Introduction
Alcohol use disorder (AUD) is characterised by compulsive, chronic alcohol use despite adverse social, occupational, or health consequences [ref. 1]. Heavy, chronic alcohol use perturbs functioning within regions of the meso-corticolimbic [ref. 2, ref. 3], salience networks [ref. 4], limbic networks [ref. 5] as well as fronto-striatal functional connectivity [ref. 6] which are thought to underpin the compulsive seeking behaviour and cognitive processes which maintain AUD [ref. 7]. Pharmacotherapies targeting these dysregulated neurobiological systems aim to manage the core symptoms of AUD and improve clinical outcomes by facilitating behavioural change. However, TGA approved medication for the management of AUD (naltrexone, disulfiram (Antabuse), and acamprosate [ref. 8–ref. 10]), are only modestly effective and there is a large degree of heterogeneity in treatment response [ref. 11] wherein new pharmacotherapeutic options are required [ref. 12].
Cannabidiol (CBD), a non-intoxicating component of Cannabis sativa, has emerged in recent years as a potential treatment target for AUD [ref. 13]. CBD is a non-intoxicating phytocannabinoid with known anti-seizure properties [ref. 14] and a favourable side effect profile[ref. 15–ref. 17] . It is a CB1R non-competitive allosteric modulator [ref. 18], mediating anandamide (AEA) transportation by targeting fatty acid-binding proteins (FABP). CBD inhibits FABP catabolism of AEA and reduces cellular uptake of endocannabinoids [ref. 19] and modulates dopaminergic release [ref. 20] in regions of networks involved in cue induced craving and regulatory behaviours [ref. 21] including mesocorticolimbic, salience, and fronto-striatal networks in healthy brains. CBD may therefore normalise functions characteristic of AUD such as reward anticipation, emotion regulation, salience processing, and executive functioning.
CBD has shown promise for the management of cue-induced craving in both preclinical models and clinical samples with substance use disorders. Preclinical sutides have demonstrate that acute doses of CBD have been shown to reduce stress and alcohol cue reinstatement in preclinical models [ref. 22, ref. 23]. The only previous clinical study of CBD in a substance use disorder to-date demonstrated that CBD, compared to placebo, reduced opiate cue-induced craving and anxiety for those with opioid use disorder [ref. 24]. Further, CBD induced decreases in attentional bias to cigarettes for those with nicotine dependence [ref. 25] suggests a capacity to normalise the appraisal of drug associated cues as hyper-salient. While THC disrupts salience network activation, neuroimaging studies have found that CBD restores salience network functionality [ref. 26]. CBD also normalises insular dysfunction during motivational salience processing for those with clinical high risk of psychosis [ref. 27]. CBD may thus have potential to manage cue induced craving in AUD through the regulation of hyper-sensitivity to alcohol associated cues, however, the effect of CBD on alcohol cue reactivity and neurocircuitry in the AUD population has not been studied to-date.
Cue-reactivity neuroimaging paradigms are a well-validated paradigm for investigating craving associated neuronal activity [ref. 28] and provide a method for examining the mechanism of action of alcohol treatments and to identify preliminary signals of efficacy. Previous research demonstrates that the dorsolateral PFC, left and right caudate, and bilateral ventromedial PFC, among others, have been shown to be relevant in alcohol cue studies in which participants are exposed to alcohol cues [ref. 29, ref. 30]. Using a similar fMRI cue reactivity paradigm, we have previously shown that, compared to high dose baclofen, placebo-treated participants demonstrated hyperactivation in the bilateral caudate nucleus and dorsal anterior cingulate cortex and that this activation was positively correlated with heavy drinking days (HDD) [ref. 31]. Similarly, naltrexone has been shown to attenuate fronto-striatal regions associated with alcohol-cue induced craving [ref. 32] and that the degree of this modulation predicts time to relapse [ref. 33].
As previously outliend [ref. 34], we aimed to conduct a double-blind, within-subject cross-over, randomised trial in individuals with AUD to determine the effect of CBD versus placebo on blood-oxygen-level-dependent functional magenetic resonance imaging (BOLD fMRI) and self-reported alcohol craving, mood and cognitive functioning. We hypothesised that mesocorticolimbic brain activation (e.g., dorsolateral PFC, left and right caudate, and bilateral ventromedial PFC), as measured using the blood oxygen level dependent (BOLD) signal when exposed to an alcohol cue, will be significantly attenuated in CBD-treated AUD individuals compared to those on placebo.
Materials and method
Design
The trial was conducted over a 48-month period at the Royal Prince Alfred Hospital (RPAH) in Sydney, NSW Australia between 2021 and 2023. The study was approved by the Human Ethics Review Committee of the Sydney Local Health District (X19-0416) and sponsored by the Sydney Local Health District. The trial was registered in the Clinical Trials Registry (NCT05387148) and the protocol (including design and analytic plan) was previously published [ref. 34]. Twenty-two non-treatment seeking individuals who met the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-V) [ref. 35], criteria for current AUD were recruited to participate in a randomised, double-blinded, placebo-controlled, crossover trial including 3 days of data collection. Here, we present results from the cue reactivity fMRI paradigm (day 2). Recruitment streams consisted of clinical referral from treating physicians, nurses, and psychologists among RPAH outpatients as well as via flyers/community advertisements at local general practitioners, newspapers, and websites. All participants provided written informed consent prior to randomisation and the commencement of the trial.
Participants
Twenty-two participants were recruited following screening for inclusion and exclusion criteria. Inclusion criteria: a) Male and female patients between the ages of 18 and 65 meeting DSM-V criteria for current AUD; b) Adequate cognition and English language skills to be able and willing to give valid informed consent and complete research interviews; c) Must have a stable housing and be able to nominate a reliable contact person for locating them if necessary.
Exclusion criteria: a) Active major psychological disorder associated with psychosis or significant suicide risk; b) Pregnancy or lactation. Women were advised to use reliable contraception throughout the duration of drug therapy, and a urine pregnancy test was conducted as necessary; c) Dependence on any substance other than nicotine (e.g., methadone); d) Diagnosis of epilepsy and/or current use of anti-epileptic drugs; e) Liver failure with jaundice or prolonged INR above 1.3; f) Medical complications such as liver failure, cardiac ischemia or conduction abnormalities, renal impairment or unstable elevated vital signs (systolic blood pressure > 180, diastolic blood pressure > 120, or heart rate > 150); g) Severe cognitive impairment or insufficient English or literacy to complete study processes; h) Concurrent use of drugs potentially exacerbated by CBD via CYP3A5, including cardiac medication (eg betablockers, calcium channel blockers, and statins), macrolides and recent antihistamine use; i) Claustrophobia; j) Extreme obesity; k) Previous brain surgery; l) Unable to complete an MRI scan (e.g., metal implants, previous emoployment as a machinist, welder or metal worker).
Procedure
After meeting the eligibility criteria through structured interview and medical evaluations, participants provide informed consent. They were then randomly assigned to one of two treatment regimens: either receiving 800 mg CBD (4 × 200 mg gel capsules per day, days 1–3) or matched placebo for two consecutive days. Following an average washout of 29.2 days (SD = 11.25), participants received the alternate treatment allocation at the subsequent session.
On the first testing day (T1), participants presented to the RPAH to receive the first dose of CBD or Placebo and complete a series of questionnaires as detailed above. On the second day (T2), participants received their second dose under supervision then completed a battery of executive functioning tasks, followed by assessments of craving and mood (see below). They were then escorted to an imaging facility for an MRI scan conducted 90 min after their second dose of CBD/matched placebo, aligned with literature suggesting Cmax occurs between 1 and 4 h post-dose [ref. 16, ref. 36, ref. 37]. Prior to imaging, participants underwent a blood alcohol content (BAC) reading (BAC = 0.00 was required), followed by a structural scan and functional cue reactivity task-based acquisition. Craving was assessed after alcohol and control cue blocks, as well as before and after the scan. Following washout, participants subsequently received the alternate capsules and completed the same testing procedure. Following completion of the repeated testing days, participants underwent a follow-up over the phone, with an average of 34 days (SD = 15.7) between final testing day and follow-up.
Interventions
Softgel capsules containing 200 mg CBD in medium chain triglyceride (MCT) oil (manufactured by Linnea SA, Lavertezzo, Switzerland) [ref. 38] and matching placebo (softgel capsules containing only MCT oil) was purchased from BOD Science [ref. 39]. The matching placebo was identical in appearance, taste, and composition except for the active ingredient of pure CBD. A three-day consecutive dose regimen was administered as follows: T1 (Day 1): 4 × 200 mg followed by T2 (Day 2 Experimental Session 1): 4 × 200 mg and then T3 (Day 3 Experimental Session 2): 4 × 200 mg. Participants were administered CBD and placebo orally with water, under supervision, to ensure compliance with the dose regimen.
Randomisation and allocation concealment
This study was conducted under double-blind conditions such that participants and study staff were unaware of medication assignment. A computer-generated random allocation procedure was conducted through REDCap (Research Electronic Data Capture; a secure web application for building and managing online surveys and databases; [ref. 40]) by the allocated RPAH pharmacist.
Measures
Sample characteristics
The following questionnaires were presented on Day 1, and responses collected online using REDCap: (1) Demographics, medical history, personal and family history of AUD, and alcohol treatment history as collected in previous research [ref. 41]; (2) a structured psychiatric diagnostic interview using the Mini-International Neuropsychiatric Interview (MINI) [ref. 42] (3) recent (last 28 days) alcohol consumption (frequency/quantity) assessed by the Timeline Followback Method (TLFB; [ref. 43]); (4) severity of alcohol dependence assessed by the Alcohol Dependence Scale (ADS; [ref. 44]); (5) craving for alcohol measured by the Penn Alcohol Craving Scale (PACS; [ref. 45]; (6) symptoms of depression, anxiety and stress as measured by the Depression Anxiety Symptom Scale (DASS) (Lovibond & Lovibond, 1995); (7) sleep problems as assessed by the Insomnia Severity Index (ISI; Bastien, Vallieres, & Morin, 2001).
Craving and mood
The following self-reported questionnaires were collected on Day 2 using REDCap unless preformed within the scanner for which Inquisit 4 [ref. 46] was used: (1) Positive and Negative Affect Schedule (PANAS; [ref. 47]), with higher scores indicating higher positive and negative mood states pre- and post- scan; (2) Alcohol Urge Questionnaire (AUQ) was used to assess expectancy of alcohol effects and urge to drink alcohol [ref. 48]; (3) 11-point Visual Analogue Scale (VAS) was used to assess alcohol craving, thirst, and anxiety, and was captured before, during, and afte the scan.
Executive functioning tasks
A 30-min neurocognitive battery of tasks measuring executive function was administered via Inquisit 4 before the scan on Day 2. These tasks included: (1) The Balloon Analogue Risk Task (BART; [ref. 49]). Participants are instructed to pump a balloon to earn as many points as possible, risking a pop that erases points for that balloon. Participants must complete 50 trials, with trial end signified either by a balloon pop (unsuccessful) or collection of trial earnings (successful). The overall number of pumps for successful trials assess self-regulation and adaptive risk taking; (2) Columbia Card Task, Hot Version with delayed feedback (CCT; [ref. 50]): a task where 32 cards are presented facedown, participants are instructed to select as many cards they would like for points before requesting them to be revealed. All selected cards are then shown to the participants one-by-one with points being awarded for every “win” card until there is none left, or a “loss” card is encountered, whereby points are subtracted from the total gained thus far in the trial. The average number of cards turned over in the task is a score of inhibition, working memory updating, task-set switching, and attention.
Participants were also administered a paper version of the Trail Making Task (TMT), comprising of Part A and B (TMT-A and TMT-B) [ref. 51]. In TMT-A, participants must connect consecutively numbered circles as quickly as possible, while in TMT-B, they alternate between numbers and letters (1-A-2-B-3-C, etc.). Difference scores were derived by subtracting completion times for TMT-A from TMT-B, with shorter completion times indicating better executive functioning.
Cue reactivity task
An adapted cue reactivity task [ref. 52] was presented to measure alcohol cue-elicited brain activity. Stimuli comprised of two types: alcohol-related pictures depicting types of alcohol (lager/wine/spirits) and drinking situations; and a control type comprising of validated, neutral pictures matched for colour and complexity. Images were presented on an MRI-compatible screen for 6.6 s in blocks of three images of the same type. Ten alcohol and six neutral blocks presented throughout the experiment with stimuli and the block order was randomised [16 total blocks, 646 s]. Following each block, an 11-point VAS was presented and participants responded using an MRI-compatible two-button response pad (Cedrus Corporation; San Pedro) within a 10 s window (see supplementary files for task procedure).
MRI acquisition
Participants were scanned using a 3-Tesla GE Discovery scanner and a 32-channel head coil. A structural scan (T1 weighted 1-mm3 voxel resolution) was acquired for use in pre-processing purposes (TR: 7200 ms, TE 2.7 ms, 176 sagittal slices, 1 mm thick, no gap, 256 × 256 × 256 matrix). For functional imaging, a GE Multiecho “HyperMEPI” echoplanar imaging sequence [ref. 53] that employs multiband [3 echo times] fMRI was used, enabling improved signal to inferior regions (e.g., VMPFC etc.) which generally suffer from signal loss [ref. 54]. Multiphase volumes of whole brain, comprising 45 axial slices were collected in an ascending interleaved fashion angled 15 degree from the AC-PC line superior to inferior using a Gradient Echo pulse sequence with 3 echoes (TR: 2000 ms, TE1 /TE2 /TE3 = 10/25/40 ms, Flip Angle:70°, FOV:220 mm, matrix of 64 × 64, slice thickness 3 mm; slice gap 0.4 mm; with a voxel resolution 3.44 × 3.44 × 3.44 mm3). Additionally, A B0 map was acquired (TR = 1000 ms, TE = 4.6 ms, flip angle 30°, FOV: 220 mm, matrix 64 × 64 and bandwidth 62.5). Task acquisition was 646 s. 323 echoplanar image volumes comprising 45 axial slices were acquired from ventral to dorsal (963 images from three diff TEs). Participants’ heads were fixed with foam pads and a set of MRI compatible headphones to minimise head movement and framewise displacement.
Image processing
Cue reactivity relevant functional scans were processed using FMRIPrep ([ref. 55, ref. 56], RRID:SCR_016216); a summary is provided here, with full preprocessing pipeline presented in Supplementary Material (FMRIprep boiler plate in supplementary). Intensity non-uniformity correction was applied to T1-w images which were then skull-stripped. These T1-w images were then used in brain surface reconstruction with volume-bases structural images segmented and normalised into MNI space.
For each participant, and session a reference volume was generated from the shortest echo of the BOLD run and skull-stripped. Head motion was estimated with respect to the BOLD reference. Respective field maps (B0-nonuniformity map) were co-registered to the EPI reference run and then converted to a displacements field map. A corrected EPI reference was calculated using the estimated susceptibility distortion for a more accurate co-registration with the T1-w image. This displacement field map was then used to better co-register the BOLD reference scan to the T1w. BOLD reference volumes were then co-registered to the T1w reference and the timeseries were resampled onto their original, native space by applying a single, composite transform to correct for head-motion and susceptibility distortions resulting in a pre-processed BOLD timeseries (PP BOLD). fMRIPrep was then leveraged to optimally combine each of the three different echo image sets. To do this the monoexponential signal decay model was fitted to a nonlinear regression to create a T2* map (using T2*/S0 estimates from a log-linear regression fit as initial values). Using the method described in [ref. 57], this T2* map was then used to combine PP BOLD across echoes. The remaining were optimally combined PP BOLD time series. These were then distortion-corrected using the B0 field maps, motion correct, co-registered to the T1-w structural data, normalised to MNI space, projected to cortical surface and then resampled to FreeSurfer’s (FreeSurfer 6.0.1, surfer.nmr.mgh.harvard.edu) fsaverage space. fMRI images were then resampled in SPM12 with spatial smoothing using a kernal of 8mm for subsequent fMRI subject modelling. See Supplementary Material for further details (image data preprocessing).
Statistical analysis
Mean, standard deviation, and proportions were calculated for all relevant sample characteristics using R software (Version 4.0.3). Further relevant state change covariates (previous day drinking, session order, and crossover contrasts defined using crosscarry package [ref. 58]) were extracted for use as covariates in subsequent generalised estimation equations (GEE). For all other non-imaging measures including mood, craving and cognitive tasks, GEEs in R software (Version 4.0.3) were used to test for statistically significant differences between treatment (CBD vs placebo) and across time (pre/post scan and alcohol vs control blocks). Interactions were examined for treatment effects across time (ie treatment effects for comparisons between alcohol/control cue exposure blocks or pre/post scan). Implementation of Generalised Estimating Equations (GEE), in addition to the use of the CrossCarry package, accounts for the complexities typical of crossover designs. CrossCarry allows for the incorporation of flexible covariate structures that can explicitly model distinct effects such as carry over effects, order effects, and habituation. In addition, GEE explicitly accounts for within-subject correlation by using a working correlation structure (in this case exchangeable), ensuring accurate estimation of the treatment effects and standard error. Therefore, by leveraging CrossCarry along with GEE our analysis presents a statistically rigorous method for isolating treatment effects while accounting for typical effects such as habituation.
Data analysis for the fMRI cue reactivity task was conducted using SPM12 with two levels. Within the first level (session-specific) analysis two conditions were modelled: alcohol cues and control cues (matched for novelty and visual complexity of the alcohol stimuli). These conditions were modelled as a box-car function convolved with the canonical haemodynamic response. Additionally, six motion correction parameters and VAS blocks were included as regressors of no interest while fixation crosses were considered an implicit baseline.
A priori regions of interest (ROIs) were identified for analysis of activity associated with alcohol cue reactivity. These ROIs comprised the dorsolateral PFC, left and right caudate, and bilateral ventromedial PFC which are highlighted during alcohol cue reactivity in studies examining AUD [ref. 59, ref. 60] including treatment effects [ref. 30]. The dorsolateral and bilateral ventromedial PFC ROIs were be defined using probabilistic maps extracted from Brainmap database [ref. 61, ref. 62], binarized with a threshold of ≥ 0.90. While the Harvard–Oxford subcortical probability atlas (http://www.cma.mgh.harvard.edu/fsl_atlas.html) was used here to define the caudate (caudate body) ROIs. ROIs were extracted using the MarsBar toolbox [ref. 63] to obtain unweighted beta estimates within ROIs specific to each task. Relevant betas were collected for both sessions of each participant for each of these regions. As beta weights from Alcohol cue > Control cue contrasts have previously been demonstrated to have low reliability as a contrast when scans repeated [ref. 64], alcohol cue exposure blocks compared to implicit baseline (ALC) and control cue exposure blocks compared to implicit baseline (CON) were applied as contrasts at the second level. Generalised estimating equations (GEEs) were conducted in R software (Version 4.0.3). These equations consisted of beta estimates of both contrasts, including a within-subjects factor of treatment (placebo, CBD) and relevant covariates were included to ensure adequate balancing of potential confounding variables (drinking on the previous day, session order and dummy variables to account for cross over effects). By employing the Simple Interactive Statistical Analysis Bonferroni tool (http:// www.quantitativeskills.com/sisa/calculations/bonfer.htm) to balance type I and type II error associated with multiple comparisons correlation between ROIs, an adjusted bonferoni threshold was set at p < 0.03. For task-based results we report beta mean correlation coefficients.
Post hoc whole brain analysis was then conducted to further investigate difference in BOLD signal between post placebo and CBD dosing imaging session using the Multivariate and Repeated Measures for Neuroimaging (MRM) toolbox (version 1.0). MRM is particularly suited to mixed effects statistical modelling of repeated measures designs. First-level ALC and CON contrasts were implemented using GLM at the second-level with 5000 permutations to account for between subject variances. Family-wise error (p < 0.05) at the cluster was corrected for by using a cluster-forming height threshold of p = 0.001 as recommended by [ref. 65]. x, y, z MNI coordinates are reported and the SPM Wake Forest University (WFU) Pickatlas toolbox (http://www.fmri.wfubmc.edu/cms/software, version 3.0.5) used to identify significant cluster regions.
Results
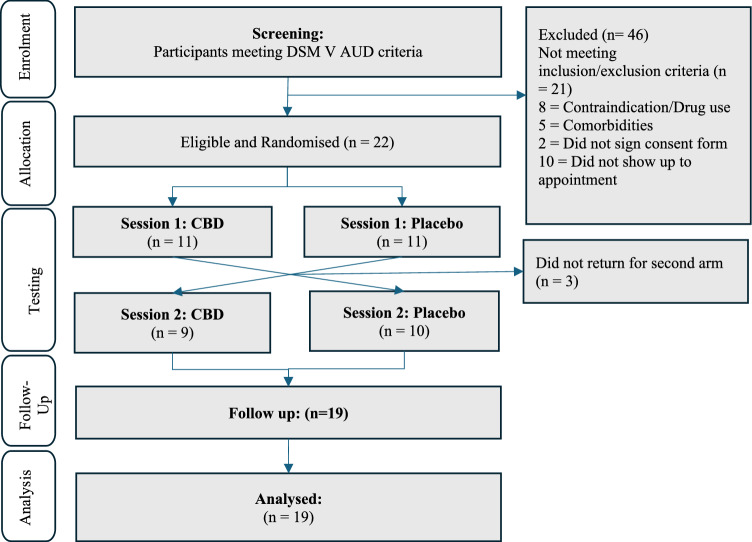
Nineteen participants completed both study sessions of the fMRI study (see Fig. 1: Consort flow diagram) with 18 participants yielding complete and usable neuroimaging data.
Sample characteristics
Baseline sample characteristics are depicted in Table 1. Overall, the mean age of the remaining participants was 29.39 years (SD = 12.83) and participants had received on average 13.95 years (SD = 2.41) of education. Participants, on average, drank 8.41 (SD = 6.02) standard drinks per drinking day, had an ADS score of 15.44 (SD = 7.81) with approximately 8.6 (SD = 9.97) years of problem drinking. Throughout the study, a total of eight side effects were recorded. Among these, four participants experienced side effects during CBD administration while the remaining four reported these following placebo administration. During CBD administration days one participant reported drowsiness, one reported somnolence, and two reported lethargy.
Table 1: Sample characteristics
| Participants (n = 18) | |
| Demographics | |
| Age (years) | 29.39 ± 12.83 [18–62] |
| Education (years) | 13.94 ± 2.41 [11–17] |
| Gender % (F/M) | 72% (13/18) |
| Employment | |
| Employed within last year | 83% (15/18) |
| Months employed within last year | 7.44 ± 4.8 [0–12] |
| Clinical characteristics | |
| Standard drinks per drinking day | 8.41 ± 6.02 [2.71 – 24] |
| Years since alcohol-related problems began | 8.63 ± 9.97 [1–38] |
| ADS score | 15.44 ± 7.81 [9–42] |
| PACs craving score | 11.53 ± 5.50 [4–24] |
| ISI | 8 ± 6.22 [1–19] |
| DASS-21: Anxiety | 3.28 ± 2.72 [0–9] |
| DASS-21: Depression | 5.39 ± 4.98 [0–16] |
| DASS-21: Stress | 7.06 ± 4.87 [0–17] |
| Comorbidities (Mini) % | 33% (6/18) |
| Antidepressants (last 28 days) % | 33% (6/18) |
| Tobacco use (last 28 days) % | 22% (4/18) |
Mean ± SDs [Range] format used to represent data unless otherwise noted
ADS alcohol dependence score, PACS penns alcohol craving score, ISI insomnia severity index, DASS depression anxiety symptom scale
Cognitive functioning
For the BART (p = 0. 4) and CCT (p = 0. 57), there were no significant effects of CBD vs placebo. For the TMT, neither part-A, part-B or B-A times were significantly different between CBD and Pla dosing sessions (p > 0.62).
Mood and subjective craving
There were no statistically significant CBD vs placebo treatment differences on the PANAS either before the scan (negative p = 0.170; positive p = 0.725) or after the scan (negative p = 0.69; positive p = 0.96). Overall, there was a significant increase between pre and post scan PANAS score for positive subscale (p < 0.001), but not negative subscale (p = 0.500), independent of treatment (negative p = 0.804; positive p = 0. 503).
There were no statistically significant differences in AUQ scores pre and post scan (p = 0.058) nor was this mediated by drug. No significant (CBD vs placebo) differences were observed pre or post scan timepoints (p = 0.40 and p = 0.49 respectively) nor was the difference between pre and post scan moderated by drug (p = 0.88). There was a significant increase in VAS scores following alcohol blocks vs control blocks (p < 0.001) but no interaction between treatment and block effect suggesting no differences in-scanner subjective craving (Alc vs Con p = 0.728) (Table 2).
Table 2: Cue Reactivity relevant AUQ and VAS scores
| Craving measures | Placebo | CBD | |
|---|---|---|---|
| AUQ | Pre-scan | 1.54 ± 0.76 | 1.56 ± 0.99 |
| Post-scan | 1.73 ± 1.13 | 1.75 ± 1.03 | |
| VAS craving | Alcohol images | 2.44 ± 2.15 | 3.19 ± 2.84 |
| Control images | 1.68 ± 2.18 | ± 2.53 | |
Mean ± SDs format used to represent data unless otherwise noted
AUQ alcohol urge questionnaire, VAS visual analogue scale
fMRI cue reactivity
ROI analysis
Compared to placebo, CBD non-significantly modulated activation in any a priori selected ROIs compared to placebo during alcohol cue (pFWE > 0.043) or control cue presentations (pFWE > 0.13). GEE results evaluating treatment effects on activation within a priori selected ROIs (left and right dorsolateral PFC, left and right caudate, and bilateral ventromedial) are presented in Supplementary Materials.
Whole brain analysis
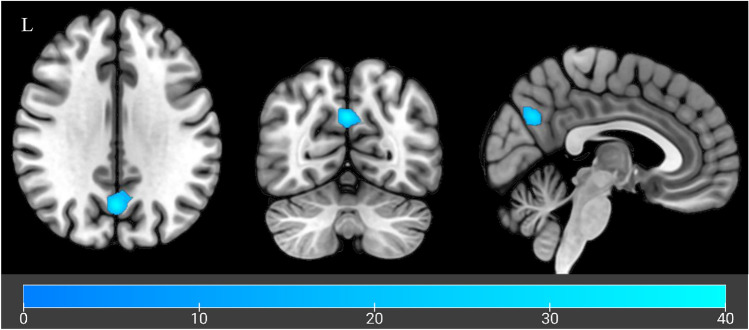
There were clusters demonstrating overall alcohol cue reactivity for the sample, with increased activation to alcohol versus control cues, with one cluster (MNI peak coordinates − 5, − 91, − 9, pFWE = 0.03) spanning the left and right lingual gyrus as well as the cuneus and a second left superior parietal lobule cluster (MNI peak coordinates − 35, − 61, 56, pFWE = 0.042) see Table 3.
Table 3: Whole brain identified brain regions with significant differences
| Side | Area | cluster size | MNI coordinates (max peak) | PFWEc | ||
|---|---|---|---|---|---|---|
| X | Y | Z | ||||
| Condition | ||||||
| Left/Right | Lingual Gyrus, Cuneus, | 329 | − 5 | − 91 | − 9 | 0.03 |
| Left | Superior Parietal Lobule | 276 | − 35 | − 61 | 56 | 0.042 |
| Treatment | ||||||
| Left | Precuneus | 191 | − 1 | − 67 | 32 | 0.038 |
Clusters with statistically significant treatment differences in BOLD signal across Alcohol and Neutral cues compared to fixation
When evaluating treatment effects (CBD vs placebo), CBD sessions were associated with significant hypoactivation for sessions in a cluster within the precuneus (MNI peak coordinates: − 1, − 67, 32, pFWE = 0.038; Fig. 2). This hypoactivation was evident across both control and alcohol cues, suggesting a non cue specific reduction within this cluster following CBD administration.
Discussion
This is the first pharmaco-fMRI crossover design evaluating the neural effects of CBD on alcohol cue reactivity in non-treatment seeking participants with AUD. While there were no treatment effects seen for fMRI cue reactivity in the a priori defined regions (chiefly mesocorticolimbic) implicated in alcohol cue reactivity, whole brain analyses demonstrated CBD mediated reductions in cue reactivity relative to placebo, in a cluster of the precuneus during alcohol as well as control cue exposure blocks.
Non-cue-specific reduced activation observed following CBD administration suggests a general attenuation of brain activation in response to external cues. Previous studies have also observed that CBD modulated activation in the precuenous, but in tasks relating to verbal learning [ref. 66]. The precuneus is located between the sensorimotor cortices of the para-central lobule and the parieto-occipital cortex and is linked with subcortical and cortical structures. It is implicated as a hub serving the default mode network and the para-cingulate network, a subnetwork of the central executive network [ref. 67]. The precuneus is involved in episodic memory retrieval, visuospatial processing, and self-mental imagery [ref. 68], which are key processes in alcohol cue reactivity [ref. 69]. The precuneus has also been implicated in higher-order aspects of craving, including the subjective experience of craving [ref. 69]. Futhermore, previous cue reactivity studies have identified a relationship between severity of dependence and activation of the precuneus [ref. 69]. Therefore, given we did not observe any effects of CBD on subjective craving measures, it is plausible that greater treatment differences may emerge in treatment seeking samples and among participants with greater dependence severity.
We found no evidence of performance differences between CBD and placebo across neuropsychological tests, including the BART, CCT, TMT-B or TMT-B-A tasks. These results align with previous literature demonstrating a lack of CBD-mediated modulation of executive functioning in individuals with opioid dependence (as measured using the Digit Symbol Substitution Task, Digit Span Test–Backward, and a Continuous Performance Task) [ref. 24] or during tobacco abstinence [ref. 70]. Taken together, these results suggest that CBD has minimal impact on executive functioning in individuals with substance use disorders. This is relevant to note given that cognitive deficits are common among treatment-seeking individuals [ref. 71] and can hinder medication compliance or response to psychological treatment [ref. 72].
The present study has both strengths and limitations. It is the first study, to our knowledge, to examine the effects of CBD on individuals with AUD with a robust crossover design, which reduces within-participant variability. Our study design included a substantial washout period and used GEE to ensure no carry-over effects of CBD. Additionally, we employed multi-echoplanar imaging which performs better [ref. 73–ref. 75] and recovers more orbito-frontal signal [ref. 76] compared to single band acquisitions. However, limitations include the potentially low generalizability of results due to the recruitment of non-treatment-seeking participants with less severe clinical presentations. The lack of statistically significant CBD-modulated changes in ROIs and clusters in our whole brain analysis may be attributed to reduced treatment effects observed in non-treatment seekers [ref. 77, ref. 78]. Nonetheless, the current sample exhibited a baseline consumption of eight standard drinks per drinking day, which, although is substantially less than the AUD treatment-seeking population in Australian clinical trials [ref. 79, ref. 80], exceeds the national alcohol consumption guidelines [ref. 81] and falls within the high risk category of World Health Organisation risk levels (82).
In conclusion, in non treatment seeking individuals with AUD, high dose CBD (800 mg/day) modulates precuneus activation in response to non-specific cues relative to placebo. CBD did not significantly change subjective craving relative to placebo and did not impair executive function or change self-reports of positive or negative affect. Further investigation in AUD treatment-seeking populations are warranted to determine the potential of CBD for the management of AUD.
Supplementary Materials
References
- PS Haber, BC Riordan, DT Winter. New Australian guidelines for the treatment of alcohol problems: an overview of recommendations. Med J Aust, 2021. [DOI | PubMed]
- VA Cardenas, TC Durazzo, S Gazdzinski. Brain morphology at entry into treatment for alcohol dependence is related to relapse propensity. Biol Psychiat, 2011. [DOI | PubMed]
- A Ilari, L Curti, M Petrella. Moderate ethanol drinking is sufficient to alter Ventral Tegmental area dopamine neurons activity via functional and structural remodeling of GABAergic transmission. Neuropharmacology, 2022. [DOI | PubMed]
- N Makris, M Oscar-Berman, SK Jaffin. Decreased volume of the brain reward system in alcoholism. Biol Psychiatr, 2008. [DOI]
- Y Suzuki, M Oishi, K Ogawa. Atrophy of the parahippocampal gyrus and regional cerebral blood flow in the limbic system in chronic alcoholic patients. Alcohol, 2010. [DOI | PubMed]
- BP Rogers, MH Parks, MK Nickel. Reduced fronto-cerebellar functional connectivity in chronic alcoholic patients. Alcoholism: Clin Exp Res, 2012. [DOI]
- ND Volkow, GF Koob. McLellan ATJNEJoM: neurobiologic advances from the brain disease model of addiction. Evaluat Brain Disease Model Addiction, 2016
- IL Petrakis. A rational approach to the pharmacotherapy of alcohol dependence. J Clin Psychopharmacol, 2006. [DOI | PubMed]
- HM Pettinati, AR Rabinowitz. Choosing the right medication for the treatment of alcoholism. Curr Psychiatry Rep, 2006. [DOI | PubMed]
- LR Zindel, HR Kranzler. Pharmacotherapy of alcohol use disorders: seventy-five years of progress. J Stud Alcohol Drugs Supplement, 2014. [DOI]
- KC Morley, CJ Perry, J Watt. New approved and emerging pharmacological approaches to alcohol use disorder: a review of clinical studies. Exp Opinion Pharmacother, 2021. [DOI]
- EM Burnette, SJ Nieto, EN Grodin. Novel agents for the pharmacological treatment of alcohol use disorder. Drugs, 2022. [DOI | PubMed]
- CN Nona, CS Hendershot, BJE Le Foll. Effects of cannabidiol on alcohol-related outcomes: a review of preclinical and human research. Exp Clin Psychopharmacol, 2019. [DOI | PubMed]
- O Devinsky, E Marsh, D Friedman. Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. Lancet Neurol, 2016. [DOI | PubMed]
- MM Bergamaschi, RHC Queiroz, MHN Chagas. Cannabidiol reduces the anxiety induced by simulated public speaking in treatment-naive social phobia patients. Neuropsychopharmacology, 2011. [DOI | PubMed]
- M Haney, RJ Malcolm, S Babalonis. Oral cannabidiol does not alter the subjective, reinforcing or cardiovascular effects of smoked Cannabis. Neuropsychopharmacology, 2016. [DOI | PubMed]
- F Leweke, D Piomelli, F Pahlisch. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Translational psychiatr, 2012. [DOI]
- R Laprairie, A Bagher, M Kelly. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br J Pharmacol, 2015. [DOI | PubMed]
- MW Elmes, M Kaczocha, WT Berger. Fatty acid-binding proteins (FABPs) are intracellular carriers for Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD). J Biol Chem, 2015. [DOI | PubMed]
- JF Cheer, KM Wassum, ML Heien. Cannabinoids enhance subsecond dopamine release in the nucleus accumbens of awake rats. J Neurosci, 2004. [DOI | PubMed]
- 21.Hurzeler TW, Joshua; Logge Warren, Towers Ellen, Suraev Anastasia, Lintzeris Nicholas, Haber Paul, Morley Kirsten (2024) Neuroimaging studies of cannabidiol and potential neurobiological mechanisms relevant for alcohol use disorders: A systematic review. J Cannabis Res 6(1):15
- A Viudez-Martinez, MS Garcia-Gutierrez, CM Navarron. Cannabidiol reduces ethanol consumption, motivation and relapse in mice. Addict Biol, 2018. [DOI | PubMed]
- A Viudez-Martínez, MS García-Gutiérrez. Manzanares JJJoP: cannabidiol regulates the expression of hypothalamus-pituitary-adrenal axis-related genes in response to acute restraint stress. J Psychopharmacol, 2018. [DOI | PubMed]
- YL Hurd, S Spriggs, J Alishayev. Cannabidiol for the reduction of cue-induced craving and anxiety in drug-abstinent individuals with heroin use disorder: a double-blind randomized placebo-controlled trial. Am J Psychiatry, 2019. [DOI]
- C Hindocha, TP Freeman, M Grabski. Cannabidiol reverses attentional bias to cigarette cues in a human experimental model of tobacco withdrawal. Addiction, 2018. [DOI | PubMed]
- MB Wall, R Pope, TP Freeman. Dissociable effects of Cannabis with and without cannabidiol on the human brain’s resting-state functional connectivity. J Psychopharmacol, 2019. [DOI | PubMed]
- R Wilson, MG Bossong, E Appiah-Kusi. Cannabidiol attenuates insular dysfunction during motivational salience processing in subjects at clinical high risk for psychosis. Transl Psychiatry, 2019. [DOI | PubMed]
- A Sangchooli, M Zare-Bidoky, AF Jouzdani. Parameter space and potential for biomarker development in 25 years of fMRI drug cue reactivity: a systematic review. JAMA psychiatry, 2024. [DOI | PubMed]
- EN Grodin, LA Ray. The use of functional magnetic resonance imaging to test pharmacotherapies for alcohol use disorder: a systematic review. Alcohol Clin Exp Res, 2019. [DOI | PubMed]
- J Zeng, S Yu, H Cao. Neurobiological correlates of cue-reactivity in alcohol-use disorders: a voxel-wise meta-analysis of fMRI studies. Neurosci Biobehav Rev, 2021. [DOI | PubMed]
- WB Logge, RW Morris, AJ Baillie. Baclofen attenuates fMRI alcohol cue reactivity in treatment-seeking alcohol dependent individuals. Psychopharmacol, 2021. [DOI]
- H Myrick, RF Anton, X Li. Effect of naltrexone and ondansetron on alcohol cue–induced activation of the ventral striatum in alcohol-dependent people. Arch Gen Psychiatry, 2008. [DOI | PubMed]
- K Mann, S Vollstädt-Klein, I Reinhard. Predicting naltrexone response in alcohol-dependent patients: the contribution of functional magnetic resonance imaging. Alcoholism: Clin Exp Res, 2014. [DOI]
- TP Hurzeler, W Logge, J Watt. The neurobehavioural effects of cannabidiol in alcohol use disorder: study protocol for a double-blind, randomised, cross over, placebo-controlled trial. Contemp Clin Trials Commun, 2024. [DOI]
- APA Diagnostic. Statistical Manual of mental disorders: Washington, 1994
- AF Manini, G Yiannoulos, MM Bergamaschi. Safety and pharmacokinetics of oral cannabidiol when administered concomitantly with intravenous fentanyl in humans. J Addict Med, 2015. [DOI | PubMed]
- G Guy, P Robson. A Phase I, open label, four-way crossover study to compare the pharmacokinetic profiles of a single dose of 20 mg of a Cannabis based medicine extract (CBME) administered on 3 different areas of the buccal mucosa and to investigate the pharmacokinetics of CBME per oral in healthy male and female volunteers (GWPK0112). J Cannabis Ther, 2014. [DOI]
- 38.Solutions LNP (2021) Linnea ECS315. Linnea SA, Via Cantonale, CH-6595 Riazzino (TI), Switzerland, 2021
- 39.Pharmaceuticals BA (2021) Cannabidiol (CBD) Isolate (Bod ECS100). Level 1, 377 New South Head Road, Double Bay, NSW 2028, Australia
- PA Harris, R Taylor, R Thielke. A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform, 2009. [DOI | PubMed]
- KC Morley, M Teesson, SC Reid. Naltrexone versus acamprosate in the treatment of alcohol dependence: a multi-centre, randomized, double-blind, placebo-controlled trial. Addiction, 2006. [DOI | PubMed]
- DV Sheehan, Y Lecrubier, KH Sheehan. The mini-international neuropsychiatric interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychitr, 1998
- LC Sobell, MB Sobell, GL Leo. Reliability of a timeline followback method: assessing normal drinkers’ reports of recent drinking and a comparative evaluation across several populations. Br J Addict, 1988. [DOI | PubMed]
- HA Skinner, BA Allen. Alcohol dependence syndrome: measurement and validation. J Abnorm Psychol, 1982. [DOI | PubMed]
- BA Flannery, JR Volpicelli, H Pettinati. Psychometric properties of the penn alcohol craving scale. Alcoholism: Clin Exp Res, 1999. [DOI]
- 46.LLC. MS: Inquisit. Seattle, USA: Millisecond Software LLC., 2009
- D Watson, LA Clark, A Tellegen. Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol, 1988. [DOI | PubMed]
- MJ Bohn, DD Krahn, BA Staehler. Development and initial validation of a measure of drinking urges in abstinent alcoholics. Alcoholism: Clin Exp Res, 1995. [DOI]
- CW Lejuez, JP Read, CW Kahler. Evaluation of a behavioral measure of risk taking: the balloon analogue risk task (BART). J Exp Psychol Appl, 2002. [DOI | PubMed]
- B Figner, RJ Mackinlay, F Wilkening. Affective and deliberative processes in risky choice: age differences in risk taking in the columbia card task. J Exp Psychol: Learn Memory Cognition, 2009
- 51.Reitan RM, Wolfson D (1985) The Halstead-Reitan neuropsychological test battery: Theory and clinical interpretation: Neuropsychology Press
- SM Grüsser, J Wrase, S Klein. Cue-induced activation of the striatum and medial prefrontal cortex is associated with subsequent relapse in abstinent alcoholics. Psychopharmacology, 2004. [DOI | PubMed]
- B Fernandez, L Leuchs, PG Sämann. Multi-echo EPI of human fear conditioning reveals improved BOLD detection in ventromedial prefrontal cortex. Neuroimage, 2017. [DOI | PubMed]
- R Deichmann, O Josephs, C Hutton. Compensation of susceptibility-induced BOLD sensitivity losses in echo-planar fMRI imaging. Neuroimage, 2002. [DOI | PubMed]
- O Esteban, R Ciric, K Finc. Analysis of task-based functional MRI data preprocessed with fMRIPrep. Nat Protoc, 2020. [DOI | PubMed]
- O Esteban, CJ Markiewicz, RW Blair. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat Methods, 2019. [DOI | PubMed]
- S Posse, S Wiese, D Gembris. Enhancement of BOLD-contrast sensitivity by single-shot multi-echo functional MR imaging. Magn Reson Med, 1999. [DOI | PubMed]
- 58.Cruz N, Melo O, Martinez CJapa (2023) CrossCarry: An R package for the analysis of data from a crossover design with GEE
- JP Schacht, RF Anton, H Myrick. Functional neuroimaging studies of alcohol cue reactivity: a quantitative meta-analysis and systematic review. Addict Biol, 2013. [DOI | PubMed]
- KE Courtney, JP Schacht, K Hutchison. Neural substrates of cue reactivity: association with treatment outcomes and relapse. Addict Biol, 2016. [DOI | PubMed]
- 61.Brett M, Anton J-L, Valabregue R, et al. (2002) Region of interest analysis using an SPM toolbox; in 8th international conference on functional mapping of the human brain: Sendai
- PT Fox, JL Lancaster. Opinion: mapping context and content: the BrainMap model. Nat Rev Neurosci, 2002. [DOI | PubMed]
- PT Fox, JL Lancaster. Mapping context and content: the BrainMap model. Nat Rev Neurosci, 2002. [DOI | PubMed]
- P Bach, I Reinhard, A Koopmann. Test–retest reliability of neural alcohol cue-reactivity: Is there light at the end of the magnetic resonance imaging tube?. Addict Biol, 2022. [DOI | PubMed]
- A Eklund, TE Nichols, H Knutsson. Cluster failure: Why fMRI inferences for spatial extent have inflated false-positive rates. Proc Natl Acad Sci, 2016. [DOI | PubMed]
- S Bhattacharyya, P Fusar-Poli, S Borgwardt. Modulation of mediotemporal and ventrostriatal function in humans by Delta9-tetrahydrocannabinol: a neural basis for the effects of Cannabis sativa on learning and psychosis. Arch Gen Psychiatry, 2009. [DOI | PubMed]
- NB Dadario, MEJB Sughrue. The functional role of the precuneus. Brain, 2023. [DOI | PubMed]
- AE Cavanna, MR Trimble. The precuneus: a review of its functional anatomy and behavioural correlates. Brain, 2006. [DOI | PubMed]
- KE Courtney, DG Ghahremani, ED London. The association between cue-reactivity in the precuneus and level of dependence on nicotine and alcohol. Drug Alcohol Depend, 2014. [DOI | PubMed]
- C Hindocha, TP Freeman, M Grabski. The effects of cannabidiol on impulsivity and memory during abstinence in cigarette dependent smokers. Sci Rep, 2018. [DOI | PubMed]
- C Bruijnen, S Walvoort, B Dijkstra. The course of cognitive performance during inpatient treatment in patients with alcohol use disorder with no, mild or major neurocognitive disorders. Alcohol Alcohol, 2021. [DOI | PubMed]
- RA Stephan, OM Alhassoon, KE Allen. Meta-analyses of clinical neuropsychological tests of executive dysfunction and impulsivity in alcohol use disorder. Am J Drug Alcohol Abuse, 2017. [DOI | PubMed]
- S Posse, S Wiese, D Gembris. Enhancement of BOLD-contrast sensitivity by single-shot multi-echo functional MR imaging. Magn Reson Med, 1999. [DOI | PubMed]
- N Weiskopf, U Klose, N Birbaumer. Single-shot compensation of image distortions and BOLD contrast optimization using multi-echo EPI for real-time fMRI. Neuroimage, 2005. [DOI | PubMed]
- SJN Posse. Multi-echo acquisition. Neuroimage, 2012. [DOI | PubMed]
- E Kirilina, A Lutti, BA Poser. The quest for the best: The impact of different EPI sequences on the sensitivity of random effect fMRI group analyses. Neuroimage, 2016. [DOI | PubMed]
- BD Kiluk, TL Frankforter, M Cusumano. Change in DSM-5 alcohol use disorder criteria count and severity level as a treatment outcome indicator: results from a randomized trial. Alcoholism: Clin Exp Res, 2018. [DOI]
- LA Ray, S Bujarski, MM Yardley. Differences between treatment-seeking and non-treatment-seeking participants in medication studies for alcoholism: do they matter?. Am J Drug Alcohol Abuse, 2017. [DOI | PubMed]
- KC Morley, A Baillie, I Fraser. Baclofen in the treatment of alcohol dependence with or without liver disease: multisite, randomised, double-blind, placebo-controlled trial. Br J Psychiatry, 2018. [DOI | PubMed]
- KM Conigrave, RL Ali, R Armstrong. Revision of the Australian guidelines to reduce health risks from drinking alcohol. Med J Aust, 2021. [DOI | PubMed]
- D Shmulewitz, E Aharonovich, K Witkiewitz. The world health organization risk drinking levels measure of alcohol consumption: prevalence and health correlates in nationally representative surveys of US adults, 2001–2002 and 2012–2013. Am J Psychiatry, 2021. [DOI | PubMed]
