Ethnomedicinal Uses, Phytochemistry, Pharmacological Activities, and Toxicology of the Subfamily Gomphrenoideae (Amaranthaceae): A Comprehensive Review
Abstract
The subfamily Gomphrenoideae is composed of about 480 accepted species, many of which have been historically used as medicinal plants, reason why they have been studied in terms of chemical profile, biological activity, and safety. This review consolidates the advances in research on this subfamily over the past 47 years, emphasizing its promising biotechnological potential and justifying the development of research in species that remain unstudied; additionally, it presents new perspectives based on the current knowledge, including the study of in vitro cultures and co‐cultures of the members of this subfamily as a sustainable approach to standardizing their chemical profiles and, consequently, enhancing their biotechnological potential. The information was collected from scientific databases such as Wiley Online Library, PubMed, Springer Link, Scielo, and Nature Research for 4 years. Verification of the scientific names and affiliations of the plants was carried out using the databases Global Biodiversity Information Facility (www.gbif.org), Plants of the World Online (www.plantsoftheworldonline.org), and The Plant List (www.theplantlist.org). To date, 512 chemical compounds have been reported for this subfamily, evidencing a wide diversity of chemical structures. It was also shown that the extracts, fractions, isolated pure compounds, and nanoparticles of this subfamily present antimicrobial, antioxidant, anticancer, anti‐inflammatory, antidiabetic, and antihyperglycemic activity, among others. Likewise, it is evident that the members of this subfamily do not present toxicity.
Article type: Review Article
Keywords: bioactivity, gomphrenoideae, phytochemistry, toxicology, traditional use
Affiliations: Department of Plant Biology University of Campinas (UNICAMP) São Paulo Brazil; Department of Industrial Microbiology Universidad de Santander (UDES) Bucaramanga Colombia
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202500530 | PubMed: 40273036 | PMC: PMC12435438
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (6.3 MB)
Introduction
Amaranthaceae is an important family of plants and includes species of economic interest; many are marketed as ornamental plants or to be used as food or healthcare based on traditional medicinal knowledge. However, several species are also known as invasive or parasitic plants and are even listed among the worst weeds. Amaranthaceae is placed in the order Caryophyllales Juss. ex Bercht. & J. Presl. and comprises about 163–195 genera and approximately 2215–3805 species, according to The Plant List database (www.theplantlist.org), including those formerly treated as the family Chenopodiaceae [ref. 1, ref. 2, ref. 3, ref. 4, ref. 5, ref. 6, ref. 7, ref. 8, ref. 9].
The Amaranthaceae family has recently become the subject of intensive systematics research. Results of the molecular genetic studies suggest that the traditional classification based on morphological and anatomical characters often did not reflect phylogenetic relationships. The family Amaranthaceae (in their narrow circumscription) is classified into two subfamilies, Amaranthoideae and Gomphrenoideae, and contains about 65 genera and 900 species [ref. 1, ref. 7].
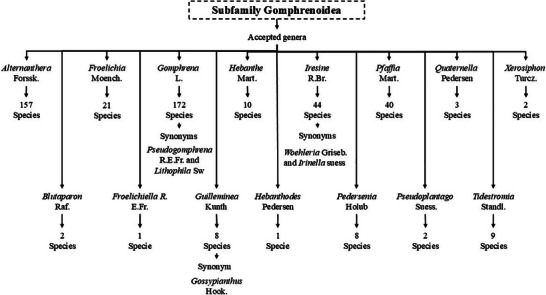
The subfamily Gomphrenoideae comprises about 480 accepted species distributed in 15 genera (Scheme 1) (GBIF), with the majority of its members being annual and perennial herbs, with some shrubs or small trees and climbing plants that have adapted to salty soils, arid environments, and human settlements [ref. 4, ref. 6, ref. 8].
Members of the Gomphrenoideae subfamily are widely used in traditional medicine in Asia, America, and Africa, making them a focus of interest for researchers. Scientists seek to verify their medicinal properties through studies of the chemical and pharmacological profile, with the goal of finding new chemical compounds that could lead to the development of new, more efficient, and safer drugs.
This article provides a comprehensive review that consolidates all available information on members of this subfamily. A wide range of topics are covered, including traditional uses, reported phytochemical profiles, biological activities of interest, and safety and/or toxicity of the extracts studied up until April 2024. Additionally, it concludes with a comparison of traditional uses and activities verified under laboratory conditions and perspectives and research directions.
This review aims to highlight the biotechnological potential of the members of this subfamily, proposing them as a promising source of bioactive molecules. It also emphasizes the importance of studying the relationship among traditional uses, chemical profiles, biological activities, and safety; additionally, it seeks to demonstrate the effect of biotic and abiotic factors on the chemical profile, among which it can mention location, climatic conditions, available nutrients, exposure to UV light, interaction with other living beings, and even the plant genotype; this highlights the need for new research strategies that allow for controlled growth conditions, enabling the optimization and standardization of metabolite production in plants. As a sustainable alternative, the use of in vitro plant tissue cultures is suggested.
Methodology
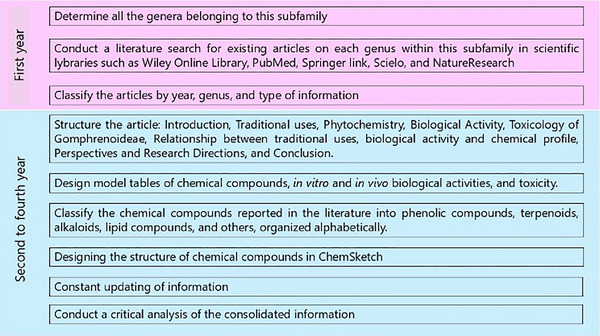
To gather relevant literature, a comprehensive search was conducted using widely recognized scientific libraries. The search focused on keywords such as names of accepted genera or their synonyms, and the literature search was limited to sources in English. Chemical structures were drawn using ChemSketch, and their names, structures, and classifications were confirmed via the PubChem and ChemSpider websites. The information was summarized in different sections in the form of tables and figures for a better understanding. Scheme 2 outlines the methodology and work plan followed to develop this review. All plant names were consulted in “Global Biodiversity Information Facility” (www.gbif.org), “Plants of the World Online” (www.plantsoftheworldonline.org), “The Plant List” (www.theplantlist.org), “The World Flora Online” (http://www.worldfloraonline.org), MPNS (http://mpns.kew.org), on July 14, 2023 and May 20, 2024.
Traditional Medicinal Uses
Ethnomedicine plays a significant role in both research and society, with 80% of the population relying on traditional medicine for healthcare. The evidence of the pharmaceutical potential of commonly used plants has increased since 2013. By 2020, the WHO indicated that over 20 000 species of plants are utilized in medicine, with 13 000 plants having been studied. Furthermore, various sources indicate that 25%–50% of modern medicine is based on compounds derived from plants [ref. 10, ref. 11, ref. 12, ref. 13, ref. 14, ref. 15].
Currently, many drug development studies are based on traditional medicine, among which can be cited aspirin, atropine, curare alkaloids, ephedrine, cortisone, digoxin, morphine, penicillin, and tubocurarine [ref. 11, ref. 15, ref. 16].
In Africa, America, Asia, Europe, and Antarctica, different members of this subfamily have been used to treat a wide variety of conditions, including chronic diseases, infectious diseases, skin diseases, respiratory issues, gastrointestinal disorders and sexually transmitted diseases, urinary disorders, malaria, diabetes, cancer, hypertension, burns, wounds, snake bites, and scorpion stings, among others. The main uses of some of the members of this subfamily are summarized in Table 1. It is important to note that some plants listed in Table 1 are included in Ayurveda, Unani, Siddha, Homeopathy, Chinese Pharmacopoeia, and “Zhonghua Bencao.”
TABLE 1: Uses in traditional medicine for major species of the Gomphrenoideae subfamily.
| Accepted names | Vernacular names and/or synonyms | Geographical location | Medicinal part | Medicinal condition treated | References |
|---|---|---|---|---|---|
| Alternanthera bettzickiana | Baptist plant, border plant, joyweed, Matiti ya ba temoins de Jéhovah, nanthara, and red calico plant | Pakistan, South America, Thailand | Whole plant, leaves | Treatment of arthritis, gastrointestinal discomfort, menstrual pain, prevention of dementia, and its use as a mild laxative. Additionally, it is characterized by having anti‐Alzheimer’s, anti‐inflammatory, antimicrobial, antioxidant, antipyretic, blood purifying, cytotoxic, diuretic, healing, hemolytic, and mutagenic properties. It is also used to promote lactation (as a galactagogue) and to provide nourishment | [ref. 17, ref. 18, ref. 19, ref. 20, ref. 21] |
| Alternanthera brasiliana | Brazilian joyweed, Carrapichinho, Doril, Novalgina, Lancetilla macho, Penicillin, Perpétua, Perpétua do mato, perpetuate of the bush, Tetracycline, Terramycin | Australia, Central America (e.g., Belize, Guatemala, Honduras, Nicaragua) French Guiana French Guiana, India, North America (Mexico, United States), South America (e.g., Argentina, Bolivia, Brazil, Colombia, Ecuador, Guyana, Peru, Suriname, Venezuela) | Leaves, whole plant | Treatment of asthma, bronchitis, cancer, cough, cold, diarrhea, discharge, fever, flu, headache, infections, inflammation, influenza, skin injuries, and wound healing. It is also used as an abortifacient, analgesic, antinociceptive, anticonvulsant, antitumoral, antiviral, anxiolytic, cholagogue, diuretic, galactagogue, and immunomodulator | [ref. 22, ref. 23, ref. 24, ref. 25, ref. 26, ref. 27, ref. 28, ref. 29, ref. 30, ref. 31, ref. 32] |
| Alternanthera caracasana HBK | Tianguis, tianguistumina, tianguispepetla, tlalpetate | Mexico | Stems, leaves, flowers, and roots | Treatment of dysentery, diarrhea, fever, and other conditions | [ref. 33] |
| Alternanthera flavescens | Lancetilla hembra | Treatment of fever and wound healing | [ref. 31] | ||
| Alternanthera littoralis P. Beauv. | Alternanthera maritima (Mart.) St. Hil. | Brazil | Treatment of infectious and inflammatory diseases | [ref. 8, ref. 34] | |
| Alternanthera paronychioides | Central and South America | Treatment of hyperuricemia, gout, rheumatic arthritis, nephritis, cystitis, uremia, diabetes, and systemic neuralgia | [ref. 35] | ||
| Alternanthera philoxeroides | Alligator weed, haicha shak, Phak Pet | Australia, Asia (e.g., Bangladesh, China, India), South America | Treatment of acute brain fever, anemia, diabetes, diarrhea, dysentery, encephalitis, hazy vision, herpes zoster, inflammation, influenza, malaria, measles, night blindness, pain, postnatal complaints, postnatal depression, puerperal fever, and viral infectious diseases. It is also used as an antipyretic, diuretic, and dressing for wounds and ulcers | [ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41, ref. 42] | |
| Alternanthera porrigens | Sanguinaria, Moradilla, Lancetilla | Peru | Whole plant | Cleansing the womb after childbirth | [ref. 43] |
| Alternanthera pungens | Kakishak and Motsweetswe | Bangladesh, Limpopo Province | Tuber and whole plant | Treatment of mouth ulcers, cough, fever, gonorrhea (drop), kidney problems, and malaria | [ref. 16, ref. 44, ref. 45] |
| Alternanthera repens | Tianquis, tianquiz, or tianguispepetla | Mexico | Treatment of gastrointestinal ailments, such as diarrhea, inflammation, and stomach ache, as well as for the treatment of typhus fever. It is also used as diaphoretic, diuretic, and astringent agent | [ref. 46, ref. 47] | |
| Alternanthera sessilis | Alternanthera triandra, Alternanthera repens, Abisrana, amaranth, Angelica, Bhiringi jhar, Brede chevrette, bunga‐bunga, Carpet weed, Chanchi, Chanchi shak, Daun tolod, Dwarf copperleaf, Gandal, Gudrisag, Hong Tian Wu, Haicha, Honagone, Honugonesoppu, Hong Tian Wu, Horng‐tyan‐wu, Kachari, keremek, keremak merah, kermak putih, Lian zi cao, Lilonchi, Lupo, Matyakshika, Matikanduri, Matsyaksi, Minannani, Mukunuwenna, Phak ped khao, Phak pet daeng, Ponnagantikura, ponnankannikkirai, ponnandan, ponnanganni, ponnannani, pudoh, rumput aoh, red sessile joyweed, Sachi‐shak, serapat, Sessile Joy weed, water Dwarf Copperleaf | Africa, Argentina, Australia, Bangladesh, Bhutan, Brazil, Cameroon, Chad, China (e.g., Huanjiang), Ecuador, Egypt, Gambia, India, Indonesia, Iran, Kenya, Malaysia, Micronesia, Nepal, New Zealand, Nigeria, Pakistan, Philippines, Saudi Arabia, Singapore, Soloman Islands, Sri Lanka, Taiwan, Uganda, United States, Zambia, and Zaire | Whole plant, leaves, roots, and shoots | Treatment of anemia, aphthous ulcer, asthma, blood dysentery, bone fractures, bronchitis, burning sensations, chickenpox, cough, cuts, diabetes, diarrhea, dysentery, dyspepsia, eczema, eye diseases, fever, flatulence, gonorrhea, hemorrhoids, headache, helminthiasis, hepatitis, hernia, hypertension, indigestion, kidney diseases, leucorrhea, liver and spleen diseases, low sperm count, lung diseases, malaria, measles, menstrual disorder, nausea, neuralgia, night blindness, ophthalmia, post‐natal depression, pruritis, rheumatism, severe pain, skin diseases, splenomegaly, sprains, tight chest, ulcers, venereal disease, vertigo, vomiting, vomiting blood, and wound healing. It is also used as an abortifacient, analgesic, anti‐inflammatory, antioxidant, antidote to snakebite and scorpion sting, antimicrobial, for bleeding control, as a cholagogue, diuretic, galactagogue, and for refreshing of eyes and body. Additionally, it is used as a poultice for boils, to relieve neuritis, and to remove tiredness, laziness, and sleepiness | [ref. 38, ref. 48, ref. 49, ref. 50, ref. 51, ref. 52, ref. 53, ref. 54, ref. 55, ref. 56, ref. 57, ref. 58, ref. 59, ref. 60, ref. 61, ref. 62, ref. 63, ref. 64, ref. 65] |
| Alternanthera tenella | Anador, Enxuga, Joyweed, melhoral, Meracilina, pérpetua do mato, and quebra panela | Australia, India, and South America (e.g., Brazil) | Leaves and roots | Treatment of bronchitis, bruises, cough, diabetes, diarrhea, dysentery, fevers, flatulence, genital inflammation, headache, inflammation, infections, itches, nausea, pain, swelling, vomiting, and wounds. It is also used as a diuretic | [ref. 29, ref. 66, ref. 67, ref. 68, ref. 69, ref. 70] |
| Blutaparon portulacoides | Capotiraguá, pirrixiu, or bredo‐de‐praia | Brazil | Treatment of leukorrhea and vulvovaginitis | [ref. 71] | |
| Froelichia | Cottonweed, snake‐cotton, and roadside weed | From the southern extremes of Canada to Northern Argentina and Uruguay | NA | To date, no traditional uses have been reported | [ref. 72] |
| Froelichia floridana (Nuttall) | Florida snake‐cotton and plains snake‐cotton | North America, West Indies of the Caribbean, and Australia | NA | To date, no traditional uses have been reported | [ref. 72] |
| Gomphrena | Bachelor Button, Globe Amaranth | Americas (particularly in South America), Antarctica, and Indo‐Malaysia | Treatment of asthma, infant flu, body wounds, bronchial disorders, cooling, cough, diarrhea, fever, gastrointestinal and respiratory disorders, high cholesterol, infectious diseases, jaundice, kidney disorders, liver disease, malaria, oliguria, throat disorders, and urinary problems. It is also utilized as an analgesic, tonic, and carminative | [ref. 2, ref. 3, ref. 73] | |
| Gomphrena arborescens L. | Paratudo, Paratudinho, Perpétua raiz do padre | Brazil | Leaves, flowers, and tuberous roots | Treatment of colitis, fevers, intermittent fevers, malaria, mental fatigue, and weakness. It is also utilized as an antidiarrheal, antithermal, antitoxic, aromatic, emmenagogue, eupeptic, protector, and tonic | [ref. 74] |
| Gomphrena boliviana | Argentina | Leaves and roots | Treatment of gastrointestinal disorders, infections, stomachache, and traumatic injuries | [ref. 75] | |
| Gomphrena celosioides | Gomphrena serrata, Gomphrena decumbens, adukowé, amegantaxe, bachelor’s button, brava, perdudilla, perpétua, pkaa Toum Hou, prostrate globe‐amaranth, soft khaki weed, and white‑eye | Americas (Argentina, Benin, Brazil, Paraguay, and Uruguay), Africa, Australia, Cambodia, East and West Vietnam, India, Indo‐Malaysia, Nigeria, Togo, and Zimbabwe | Leaves, roots, whole plant | Treatment of asthma, bronchitis, wound healing, coughs, cold, dermatological problems, diabetes, diarrhea, dysmenorrhea, fever, gastrointestinal diseases, hay fever, hypertension, kidney infections, jaundice, kidney stones, lithiasic problems, liver diseases (e.g., viral hepatitis A and C, liver damage), malaria, renal disorders, respiratory diseases, sexually transmitted diseases, skin infections/diseases/problems, infectious diseases, urinary tract disorders, vulvovaginitis, and worms. It is also used as an abortive, analgesic, antifungal, antibiotic, diuretic, immunostimulant, and tonic/carminative | [ref. 3, ref. 76, ref. 77, ref. 78, ref. 79, ref. 80, ref. 81, ref. 82, ref. 83, ref. 84, ref. 85, ref. 86, ref. 87, ref. 88, ref. 89, ref. 90, ref. 91, ref. 92] |
| Gomphrena globosa | Bachelor button, Botamphul, Globe amaranth, Meilingper, Perpétua, Perpétuas‐roxas, Qianrihong, Trochiek Toun Say, and White bachelor button | Argentina, Bangladesh, Belize, Bolivia, Brazil, Cambodia, Canada, China (Huanjiang), Colombia, Costa Rica, Ecuador, French Guiana, Guatemala, Guyana, Honduras, India, Mexico, Panama, Peru, Portugal, South Africa, Suriname, United States, Trinidad, Tobago, Venezuela | Leaves, inflorescence, flowers, rhizome, and whole plant | Treatment of bronchial asthma, bronchitis, cough, diabetes, diarrhea, gallstones, gangrenous wounds, giddiness, hemorrhage, headache, heat and indigestion, hemoptysis, hoarseness, hypertension, indigestion, jaundice, kidney and prostate problems, oliguria, reproductive problems, respiratory diseases, urinary retention, tuberculosis, urinary system conditions, uterine infection, and whooping cough. It is also used as an antimicrobial, antioxidant, and expectorant | [ref. 3, ref. 25, ref. 29, ref. 44, ref. 79, ref. 93, ref. 94, ref. 95, ref. 96, ref. 97, ref. 98] |
| Gomphrena macrocephala | Brazil | Roots | It is used as a stimulant and a tonic | [ref. 99] | |
| Gomphrena martiana | Solo and yerba de pollo | South America (e.g., Argentina) | Leaves and roots | Treatment of liver, kidney, urinary tract, and gastrointestinal disorders; infections; stomachache; and traumatic injuries. It is also used as a diuretic and blood purifier | [ref. 75, ref. 100] |
| Gomphrena virgata | Cangussu‐branco | Brazil | Treatment of pain, inflammation, and infection. It is also used as an anti‐lethargic | [ref. 101] | |
| Guilleminea densa | Sanguinaria | Treatment of gastric ulcers and menstrual cramps. It is also used as an antihemorrhagic | [ref. 102] | ||
| Iresine angustifolia | Hierba del arlomo | Mexico | Treatment of insect bites | [ref. 103] | |
| Iresine diffusa | Iresine celosia, Iresine celosioides, herb of the Mayas, Paja Blanca, and Sangrinaria | Central and South America (e.g., Mexico, Peru), the West Indies, and the Southeastern United States | Whole plant | Treatment of anorexia, cancer, fever, inflammation, malaria, menstrual symptoms in adolescents, mouth sores, oral infections, prostate and urethra ailments, rash, skin problems, swelling, and typhoid fever | [ref. 43, ref. 104, ref. 105] |
| Iresine herbstii Hook | Bloodleaf, cimora senorita, chicken gizzard, beefsteak plant, herbst’s bloodleaf, Mussurú, and Phak phaeo daeng | The entire world | Whole plant, aerial part, leaves, and stem | Treatment of anemia, broken bones, cancer, candidiasis, burns, eczema, wound healing, inflammatory bowel diseases, peptic ulcer, pimples, and sores. It is also used as antipyretic, skin depurative, and tonic | [ref. 106, ref. 107, ref. 58, ref. 59, ref. 108, ref. 109, ref. 110, ref. 111] |
| Pfaffia glomerata | Acônito, Brazil ginseng, corango sempre‐viva, dipyrone, fáfia, paratudo, and novalgina | Brazil and Ecuador | Roots | Treatment of cancer, cholesterol, diabetes, flu, gastritis, impotence, inflammatory disorders, memory lapses, local pain, palpitations, rheumatism, stomach problems, and stress. It is also used as antioxidants, aphrodisiac, stimulant, tonic, and for wound healing. As well as it is utilized for restoring vital functions, increasing physical strength and mental equilibrium, and protecting the gastric mucosa from injury | [ref. 25, ref. 112, ref. 113, ref. 114, ref. 115, ref. 116, ref. 117, ref. 118, ref. 119] |
| Pfaffia paniculata | Hebanthe eriantha, Hebanthe paniculata, Gomphrena paniculata, Gomphrena eriantha, Iresine erianthos, Iresine paniculata, Iresine tenuis, Pfaffia eriantha, Pfaffia virgata, Xeraea paniculata, Brazilian ginseng, paratudo, suma | Brazil, Ecuador, Panama, Peru, and Venezuela | Roots | Treatment of arthritis, diabetes, cancer, rheumatism, and ulcers. It is also used as an analgesic, anti‐inflammatory, antistress, antitumor, aphrodisiac, invigorating, memory booster, and tonic | [ref. 120, ref. 121, ref. 122, ref. 123, ref. 124, ref. 125, ref. 126] |
| Pfaffia townsendii | Brazilian ginseng | Brazil | It is used as an anti‐inflammatory, tonic, analgesic, and antidiabetic agent | [ref. 127] | |
| Tidestromia oblongifolia | United States, Mexico | Treatment of headache and foot pain | [ref. 128] |
It is noteworthy that among the 15 genera of this subfamily, the traditional use of only eight (8) has been documented in the literature, accounting for 53.33%. Most of these plants have been used to treat infections, inflammation, pain and gastrointestinal, respiratory, and skin diseases. In this regard, conducting multidisciplinary research to verify whether these plants really have the potential to treat these conditions, as shown in Scheme 3. To date, only some species belonging to the subfamily have been studied in terms of chemical profile and biotechnological potential, as described in detail in the following sections.
Phytochemistry
Medicinal plants produce secondary metabolites or phytochemicals, which are responsible for their biological and pharmacological activity [ref. 12, ref. 85, ref. 88].
The production, quality, and quantity of phytochemical compounds are influenced by the biotic and abiotic factors present in the environment. Consequently, the phytochemical profile of a plant can vary significantly based on the location and growing conditions.
Furthermore, the concentration of secondary metabolites differs among the different parts of the plant, with the leaves typically exhibiting the highest concentration of phytochemicals [ref. 16, ref. 129, ref. 130].
Different natural products and their derivatives have been studied, revealing therapeutic potential for various diseases with fewer side effects than synthetic drugs [ref. 14, ref. 131]. In this context, 109 compounds identified within this subfamily have been evaluated for various biological activities, demonstrating significant activity in most instances. Notably, phenolic compounds are the most widely studied, with antimicrobial activity being the most assessed.
In the case of this subfamily until 2024, 512 compounds have been reported, including phenolic compounds, terpenoids, alkaloids, lipid compounds, and other minor compounds, demonstrating the wide chemical diversity present in the members of this subfamily.
A comprehensive review of the bioactive secondary metabolites isolated from Gomphrenoideae, including their sources, structures, and biological properties, is presented below and summarized in Tables 2, 3, 4, 5, 6 and Figure 1. The structures of compounds 63, 286, 287, and 334 are not provided, as there is no available literature presenting them, nor sufficient information to determine them.
TABLE 2: Phenolic compounds isolated from the Gomphrenoideae subfamily.
| No. | Compound | Species | Parts of plant | References |
|---|---|---|---|---|
| Flavonoids | ||||
| Flavan3‐ols | ||||
| 1 | Catechin | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Alternanthera philoxeroides | Whole plant | [ref. 132] | ||
| Alternanthera sessilis | Whole plant | [ref. 57, ref. 133] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 81] | ||
| Gomphrena perennis | Aerial parts | [ref. 134] | ||
| 2 | Epigallocatechin | Alternanthera sessilis | — | [ref. 57] |
| Flavones | ||||
| 3 | Apigenin | Alternanthera brasiliana | Leaves | [ref. 26] |
| Alternanthera sessilis | Leaves | [ref. 55, ref. 57] | ||
| 4 | Demethyltorosaflavone B | Alternanthera philoxeroides | Aerial parts | [ref. 38] |
| 5 | Demethyltorosaflavone D | Alternanthera philoxeroides | Aerial parts and whole plant | [ref. 38, ref. 135, ref. 136] |
| 6 | 5,7‐Dihydroxy‐3,6‐dimethoxyflavone | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75, ref. 100, ref. 137] | ||
| 7 | 5,7‐Dihydroxy‐6‐methoxyflavone (oroxilin A) | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75, ref. 137] | ||
| 8 | Dimethoxy‐flavone | Gomphrena celosioides Mart. | Aerial parts | [ref. 81] |
| 9 | 5,6‐Dimethoxy‐7‐hydroxyflavone (baicalein 5,6‐dimethyl ether) | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75, ref. 137] | ||
| 10 | 3,5‐Dimethoxy‐6,7‐methylenedioxyflavone | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75, ref. 137] | ||
| 11 | Diosmetin | Froelichia floridana | Whole plants | [ref. 72] |
| 12 | Isoorientin | Alternanthera sessilis | Leaves | [ref. 137] |
| 13 | Isorhamnetin | Alternanthera maritima | Aerial parts | [ref. 138] |
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena claussenii | Whole plant | [ref. 137] | ||
| Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] | ||
| 14 | Isovitexin | Alternanthera maritima | Aerial parts | [ref. 137] |
| Alternanthera sessilis | Whole plant | [ref. 140] | ||
| Gomphrena perennis | Aerial parts | [ref. 134] | ||
| 15 | Luteolin | Alternanthera brasiliana | Leaves | [ref. 26] |
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| 16 | Orientin | Alternanthera brasiliana | Leaves | [ref. 26] |
| Alternanthera sessilis | Leaves | [ref. 137] | ||
| 17 | 3,5,6,7‐Tetramethoxyflavone | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75, ref. 137] | ||
| Flavonol | ||||
| 18 | Galangin triOMe | Gomphrena martiana | Whole plant | [ref. 137] |
| 19 | Gomphrenol | Blutaparon portulacoides | Stems | [ref. 71] |
| Gomphrena celosioides Mart. | — | [ref. 84] | ||
| Gomphrena claussenii | Whole plant | [ref. 137] | ||
| Gomphrena globosa | Leaves | [ref. 137] | ||
| 20 | Kaempferol | Alternanthera brasiliana | Leaves, stems, and whole plant | [ref. 23, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 34, ref. 137] | ||
| Alternanthera philoxeroides | Leaves | [ref. 38, ref. 141] | ||
| Alternanthera tenella Colla | Leaves, stems, and whole plant | [ref. 28, ref. 142, ref. 143] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena claussenii | Whole plant | [ref. 137] | ||
| Gomphrena globosa | Inflorescence and leaves | [ref. 94, ref. 98, ref. 136, ref. 144] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 21 | Kaempferol monosulfate | Alternanthera sessilis | Stems | [ref. 56] |
| 22 | Myricetin | Alternanthera sessilis | — | [ref. 55] |
| 23 | Patuletin | Gomphrena claussenii | Whole plant | [ref. 137] |
| 24 | Quercetin | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Alternanthera brasiliana | Leaves, stems, and whole plant | [ref. 23, ref. 26, ref. 28] | ||
| Alternanthera maritima | Aerial parts | [ref. 34, ref. 137, ref. 138, ref. 145] | ||
| Alternanthera paronychioides | — | [ref. 35] | ||
| Alternanthera philoxeroides | Leaves | [ref. 42, ref. 136] | ||
| Alternanthera sessilis | Leaves and whole plant | [ref. 55, ref. 133, ref. 137] | ||
| Alternanthera tenella Colla | Leaves, stems, and whole plant | [ref. 28, ref. 142, ref. 143] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides | Roots | [ref. 146] | ||
| Gomphrena claussenii | Whole plant | [ref. 137] | ||
| Gomphrena globosa | Inflorescence and leaves | [ref. 94, ref. 98, ref. 137] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 25 | Quercetin 3‐methyl ether (3‐methoxy quercetin) | Alternanthera maritima | Aerial parts | [ref. 138, ref. 145] |
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| 26 | Quercetin‐3‐O‐methyl ester | Alternanthera maritima | Aerial parts | [ref. 34] |
| 27 | 3,5,3′,4′‐Tetrahydroxy‐6,7‐methylenedioxy flavone | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| Isoflavone | ||||
| 28 | Daidzein | Alternanthera sessilis | Stem | [ref. 56, ref. 57] |
| 29 | Daidzin | Froelichia floridana | Whole plants | [ref. 72] |
| 30 | 2′,5‐Dimethoxy‐6,7‐methylenedioxyisoflavon (tlatlancuayin) | Iresine celosioides | Whole plant | [ref. 137] |
| Iresine herbstii | Aerial parts | [ref. 111, ref. 147] | ||
| 31 | Irisone B | Blutaparon portulacoides | Aerial parts | [ref. 148] |
| Gomphrena celosioides Mart. | Aerial parts | [ref. 81] | ||
| 32 | 2′,2,5‐Trimethoxy‐6,7‐methylenedioxyisoflavanone | Iresine herbstii | Aerial parts | [ref. 111] |
| Aurone | ||||
| 33 | (E)‐3′‐O‐β‐d‐glucopyranosyl‐4,5,6,4′‐tetrahydroxy‐7,2′‐dimethoxyaurone | Gomphrena agrestis | Whole plant | [ref. 2] |
| Flavonoid glycosides | ||||
| Flavone glycosides | ||||
| 34 | Acacetin 8‐C‐[α‐l‐rhamnopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Alternanthera maritima | Aerial parts | [ref. 34, ref. 138, ref. 145] |
| Alternanthera tenella Colla | Whole plant | [ref. 142, ref. 143] | ||
| 35 | Alternanthin | Alternanthera philoxeroides | Aerial parts, stems, leaves, and whole plant | [ref. 36, ref. 38, ref. 135, ref. 137] |
| 36 | Alternanthin B | Alternanthera philoxeroides | Aerial parts and whole plant | [ref. 36, ref. 38, ref. 135, ref. 136] |
| 37 | Apigenin‐6,8‐di‐C‐β‐d‐glucopyranoside | Alternanthera sessilis | Stems and whole plant | [ref. 56, ref. 149] |
| 38 | Chrysin 7‐O‐glucuronide | Gomphrena martiana | Whole plant | [ref. 137] |
| 39 | Chrysoeriol‐6‐C‐β‐d‐boivinopyranoside | Alternanthera philoxeroides | — | [ref. 40] |
| 40 | Chrysoeriol‐6‐C‐β‐d‐Boivinopyranosyl‐4′‐O‐β‐d‐glucopyranoside | Alternanthera philoxeroides | — | [ref. 40] |
| 41 | Chrysoeriol 7‐O‐rhamnoside or chrysoeriol 7‐rhamnoside | Alternanthera philoxeroides | Whole plant and aerial parts | [ref. 38, ref. 135, ref. 136] |
| 42 | Glucopyranosyl‐vitexin | Alternanthera tenella Colla | Leaves and stems | [ref. 28] |
| 43 | 2″‐O‐β‐d‐glucopyranosyl‐vitexin | Alternanthera maritima | Aerial parts | [ref. 34, ref. 138, ref. 145] |
| Alternanthera tenella Colla | Whole plant | [ref. 142, ref. 143] | ||
| 44 | Isorhamnetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐galactopyranoside | Alternanthera maritima | Aerial parts | [ref. 34, ref. 138] |
| 45 | Isorhamnetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside | Alternanthera maritima | Aerial parts | [ref. 34] |
| 46 | Isorhamnetin‐3‐hexoside | Gomphrena globosa | Inflorescence | [ref. 73, ref. 94, ref. 98] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 47 | Isorhamnetin‐3‐(pentosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 48 | Isorhamnetin‐3‐(6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 49 | Isorhamnetin 3‐O‐[α‐rhamnopyranosyl‐(1 → 6)‐β‐d‐glucopyranoside] | Alternanthera maritima | Aerial parts | [ref. 138] |
| Gomphrena celosioides | Aerial parts | [ref. 92] | ||
| 50 | Isorhamnetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside | Alternanthera maritima | Aerial parts | [ref. 145] |
| Gomphrena globosa L. | Flower | [ref. 144] | ||
| 51 | Isorhamnetin‐3‐O‐β‐d‐apiofuranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 52 | Isorhamnetin 3‐O‐glucoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 53 | Isorhamnetin 3‐O‐robinobioside | Alternanthera maritima | Aerial parts | [ref. 137] |
| Gomphrena martiana | Whole plant | [ref. 137] | ||
| 54 | Isorhamnetin 3‐O‐β‐robinobioside | Gomphrena boliviana | Whole plant | [ref. 75] |
| Gomphrena martiana | Whole plant | [ref. 75] | ||
| 55 | Isorhamnetin 3‐O‐rutinoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 137] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena globosa | Flower | [ref. 150] | ||
| 56 | Isorhamnetin‐O‐glucuronyl‐deoxyhexosyl‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 57 | Isorhamnetin‐O‐glucuronyl‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 58 | Luteolin‐6‐C‐β‐d‐boivinopyranoside | Alternanthera philoxeroides | — | [ref. 40] |
| 59 | Luteolin‐6‐C‐β‐d‐boivinopyranosyl‐3′‐O‐β‐d‐glucopyranoside | Alternanthera philoxeroides | — | [ref. 40] |
| 60 | Luteolin‐6‐C‐β‐d‐boivinopyranosyl‐4′‐O‐β‐d‐glucopyranoside | Alternanthera philoxeroides | — | [ref. 40] |
| 61 | Luteolin 8‐C‐E‐propenoic acid | Alternanthera philoxeroides | Aerial parts | [ref. 38] |
| 62 | Luteolin‐8‐C‐rhamnosylglucoside | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 63 | Methoxy‐trihydroxymethylenedioxyflavone O‐glucuronyl‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 64 | Nepetin 3‐O‐rhamnoside | Alternanthera philoxeroides | Leaves | [ref. 137] |
| 65 | Patuletin O‐deoxyhexosyl‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 66 | Patuletin 3‐O‐glucoside | Gomphrena claussenii | Whole plant | [ref. 137] |
| 67 | Patuletin 3‐O‐β‐d‐glucopyranoside | Pfaffia townsendii | Whole plant | [ref. 127] |
| 68 | Patuletin O‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 69 | 2″‐O‐pentosyl‐6‐C‐hexosyl‐apigenin (2″‐O‐pentosyl‐isovitexin) | Alternanthera brasiliana | Leaves | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 70 | 2″‐O‐pentosyl‐8‐C‐hexosyl‐apigenin (2″‐O‐pentosyl‐vitexin) | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Stems | [ref. 28] | ||
| 71 | Potentilin A | Gomphrena globosa L. | Flower | [ref. 144] |
| 72. | 2″‐O‐Rhamnopyranosyl‐vitexin | Alternanthera brasiliana | Leaves | [ref. 28] |
| Alternanthera maritima | Aerial part | [ref. 34, ref. 138, ref. 145] | ||
| Alternanthera tenella Colla | Leaves and whole plant | [ref. 28, ref. 143] | ||
| 73 | 2″‐O‐rhamnosyl‐6‐C‐glucosil methylluteolin | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 74 | 2″‐O‐rhamnosylvitexin | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Alternanthera sessilis | Stems | [ref. 56] | ||
| 75 | 2″‐O‐Rhamnosylswertisin | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 76 | 3,5,3′,4′‐Tetrahydroxy‐6,7‐methylenedioxyflavone‐3‐O‐deoxyhexosyl‐hexoside | Gomphrena haageana K. | Flower | [ref. 73] |
| 77 | 3,5,3′,4′‐tetrahydroxy‐6,7‐methylenedioxyflavone‐3‐hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| Gomphrena haageana K. | Flower | [ref. 73] | ||
| 78 | 7,3′,4′,5′‐Tetrahydroxy‐flavanone 7‐O‐glucoside | Alternanthera sessilis | Leaves | [ref. 137] |
| 79 | Torosaflavone E | Alternanthera philoxeroides | Aerial parts and whole plant | [ref. 38, ref. 145] |
| 80 | 3,5,3′‐Trihydroxy‐4′‐methoxy‐6,7‐methylenedioxyflavone | Blutaparon portulacoides | Aerial parts, stem, and whole plant | [ref. 71, ref. 148, ref. 151, ref. 152] |
| 81 | 3,5,3′‐Trihydroxy‐4′‐methoxy‐6,7‐methylenedioxy‐favone‐glucosilated | Blutaparon portulacoides | Stems and whole plant | [ref. 71, ref. 152] |
| 82 | 3′,4′,7‐Trihydroxy‐6‐methoxyflavone | Iresine herbstii | — | [ref. 111] |
| 83 | 3,5,4′‐Trihydroxy‐6,7‐methylenedioxyflavone‐3‐(6‐acetyl)hexoside or Gomphrenol 3‐O‐(6‐acetyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 73, ref. 94, ref. 98, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 84 | 3,5,4′‐Trihydroxy‐6,7‐methylenedioxyflavone‐3‐hexoside or gomphrenol 3‐O‐hexoside | Gomphrena globosa | Inflorescence | [ref. 73, ref. 94, ref. 98, ref. 150] |
| Gomphrena sp | Flower | [ref. 73] | ||
| 85 | 3,5,4′‐trihydroxy‐6,7‐methylenedioxyflavone‐3‐(2‐pentosyl) hexoside or gomphrenol 3‐O‐(2‐pentosyl)‐hexoside | Gomphrena globosa | Inflorescence | [ref. 73, ref. 94, ref. 98] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 86 | 3,5,4′‐Trihydroxy‐6,7‐methylenedioxyflavone‐3‐(2‐pentosyl, 6‐acetyl)hexoside or gomphrenol 3‐O‐(2‐pentosyl, 6‐acetyl)‐hexoside | Gomphrena globosa | Inflorescence | [ref. 73, ref. 94, ref. 98] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 87 | 3,5,4′‐Trihydroxy‐6,7‐methylenedioxyflavone‐3‐(6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 88 | Vitexin | Alternanthera brasiliana | Leaves and whole plant | [ref. 23, ref. 26, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 34, ref. 137, ref. 138] | ||
| Alternanthera tenella Colla | Leaves, stems, and whole plant | [ref. 28, ref. 142, ref. 143] | ||
| 89 | 2″ Vitexin‐O‐glucoside | Alternanthera maritima | Aerial parts | [ref. 137] |
| 90 | 2″ Vitexin‐O‐rhamnoside | Alternanthera maritima | Aerial parts | [ref. 137] |
| Flavonol glycosides | ||||
| 91 | Gomphrenol‐3‐glucoside | Blutaparon portulacoides | Stems and whole plant | [ref. 71, ref. 152] |
| 92 | Gomphrenol‐3‐O‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 93 | Gomphrenol‐3‐O‐β‐d‐glucopyranosyl‐(1 → 6)‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 94 | Gomphrenol‐3‐O‐β‐d‐xylopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 95 | 3′‐Hydroxygomphrenol‐3‐O‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 96 | Kaempferol glucoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 97 | Kaempferol‐3‐(2‐pentosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 98 | Kaempferol‐3‐(2‐pentosyl, 6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 99 | Kaempferol‐3‐(6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 100 | Kaempferol‐3‐O‐α‐l‐rhamnopyranosyl‐(1 → 6)‐β‐dglucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 101 | Kaempferol‐3‐O‐β‐dglucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 102 | Kaempferol O‐acetylhexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 103 | Kaempferol 3‐O‐glucoside | Alternanthera philoxeroides | Whole plant | [ref. 136] |
| Gomphrena claussenii | Whole plant | [ref. 137] | ||
| Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 94, ref. 98, ref. 150, ref. 153] | ||
| Gomphrena sp. | Flower | [ref. 73] | ||
| Pfaffia glomerata | Inflorescences | [ref. 154] | ||
| 104 | Kaempferol O‐glucuronide‐O‐hexoside | Gomphrena globosa | Flower | [ref. 150] |
| 105 | Kaempferol 3‐O‐(2‐pentosyl)‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 106 | Kaempferol 3‐O‐β‐d‐(6‐O‐p‐E‐coumaroyl)‐glucopyranoside (tiliroside) or kaempferol‐3‐O‐(6‐p‐coumaroyl)‐glucoside | Froelichia floridana | Whole plants | [ref. 72] |
| Gomphrena agrestis | Whole plant | [ref. 2] | ||
| Pfaffia glomerata | Inflorescences | [ref. 154] | ||
| Pfaffia townsendii | Whole plant | [ref. 127] | ||
| 107 | Kaempferol‐3‐O‐(6″‐O‐(E)‐p‐coumaroyl)‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 108 | Kaempferol‐3‐O‐(6″‐O‐(Z)‐p‐coumaroyl)‐β‐d‐glucopyranoside | Gomphrena globosa L. | Flower | [ref. 144] |
| 109 | Kaempferol 3‐O‐β‐d‐(6″‐feruloylglucopyranoside) | Gomphrena globosa L. | Flower | [ref. 144] |
| 110 | Kaempferol 3‐O‐(2‐pentosyl, 6‐O‐rhamnosyl)‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 111 | Kaempferol‐rhamnosyl‐rhamnosyl‐glycoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Stems | [ref. 28] | ||
| 112 | Kaempferol 3‐O‐(2‐rhamnosyl)‐hexoside | Gomphrena globosa | Flower | [ref. 150] |
| 113 | Kaempferol 3‐O‐(6‐rhamnosyl)‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 114 | Kaempferol O‐rhamnosyl‐pentoside | Gomphrena globosa var. albiflora | Flower | [ref. 73] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 115 | Kaempferol 3‐O‐robinobioside | Alternanthera brasiliana | — | [ref. 28] |
| 116 | Kaempferol rutinoside or kaempferol 3‐O‐rutinoside or kaempferol 3‐rutinoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides | Aerial parts | [ref. 92] | ||
| Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150, ref. 153] | ||
| Gomphrena sp. | Flower | [ref. 73] | ||
| 117 | Laricitin 3‐O‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 118 | 8,8‴‐methylene bis(spinacetin 3‐O‐robinobioside) | Blutaparon portulacoides | Leaves | [ref. 155] |
| 119 | Quercetin‐glucoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 120 | Quercetin‐3‐hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 121 | Quercetin‐3‐(pentosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 122 | Quercetin‐3‐(2‐pentosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 123 | Quercetin‐3‐(2‐pentosyl, 6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 124 | Quercetin‐3‐(6‐rhamnosyl)hexoside | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98] |
| 125 | Quercetin 3‐O‐(2‐pentosyl, 6‐rhamnosyl)‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 126 | Quercetin 3‐O‐glucoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena haageana K. | Flower | [ref. 73] | ||
| Gomphrena sp. | Flower | [ref. 73] | ||
| Pfaffia glomerata | Inflorescences | [ref. 154] | ||
| 127 | Quercetin 3‐OMe | Alternanthera maritima | Aerial parts | [ref. 137] |
| 128 | Quercetin 3‐O‐(6‐pentosyl)‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 129 | Quercetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside | Alternanthera maritima | Aerial parts | [ref. 145] |
| 130 | Quercetin 3‐O‐rutinoside (rutin) | Alternanthera brasiliana | Aerial parts and stems | [ref. 22, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 34, ref. 137, ref. 138] | ||
| Alternanthera sessilis | Leaves and whole plant | [ref. 55, ref. 133] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150, ref. 153] | ||
| Gomphrena haageana K. | Flower | [ref. 73] | ||
| Gomphrena sp. | Flower | [ref. 73] | ||
| 131 | Quercetin‐O‐acetylhexoside | Gomphrena sp. | Flower | [ref. 73] |
| 132 | Quercetin O‐glucuronide‐O‐hexoside | Gomphrena globosa var. albiflora | Flower | [ref. 73, ref. 150] |
| Gomphrena sp. | Flower | [ref. 73] | ||
| 133 | Spinacetin 3‐O‐robinobioside | Blutaparon portulacoides | Leave and whole plant | [ref. 152, ref. 155] |
| Non‐flavonoid phenolic compounds | ||||
| Benzoic acids | ||||
| 134 | Dihydroxybenzoic acid glucoside | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 135 | 3,4‐Dimethoxybenzoic acid | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 136 | 3,4‐Dimethylbenzoic acid | Gomphrena globosa | — | [ref. 156] |
| 137 | Ethyl gallate | Alternanthera sessilis | — | [ref. 57] |
| 138 | Gallic acid | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Alternanthera brasiliana | Leaves | [ref. 26] | ||
| Alternanthera philoxeroides | Whole plant | [ref. 132] | ||
| Alternanthera sessilis | — | [ref. 55] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena globosa | Flowers | [ref. 153] | ||
| Gomphrena perennis | Aerial parts | [ref. 134] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 139 | Gentisic acid | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 140 | 4‐Hydroxibenzoic acid or p‐hydroxybenzoic acid | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera philoxeroides | Aerial parts | [ref. 38] | ||
| Alternanthera sessilis | Stems | [ref. 55, ref. 56, ref. 57] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 78, ref. 157] | ||
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| 141 | p‐Methoxybenzoic acid | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 142 | Protocatechuic acid | Alternanthera sessilis | Stems | [ref. 56] |
| Hydroxycinnamic acids–phenolic acids | ||||
| 143 | Caffeic acid | Alternanthera brasiliana | Leaves | [ref. 26] |
| Blutaparon portulacoides | Stems and whole plant | [ref. 71, ref. 152] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 81] | ||
| Gomphrena globosa | Flowers | [ref. 153] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 144 | Caffeoyl‐glucose | Gomphrena celosioides Mart. | Aerial parts | [ref. 81] |
| 145 | Chlorogenic acid | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Alternanthera brasiliana | Leaves and stems | [ref. 26, ref. 28] | ||
| Alternanthera philoxeroides | Leaves | [ref. 38, ref. 141] | ||
| Alternanthera sessilis | — | [ref. 57] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 146 | Cinnamic acid | Iresine angustifolia | Whole plant | [ref. 103] |
| 147 | Coumaric acid | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 148 | cis‐p‐Coumaric acid | Gomphrena globosa | Flowers | |
| 149 | Ferulic acid or trans‐ferulic acid | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera paronychioides | — | [ref. 35] | ||
| Alternanthera philoxeroides | Leaves | [ref. 38, ref. 141] | ||
| Alternanthera sessilis | Leaves | [ref. 55, ref. 57] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Blutaparon portulacoides | Stems and whole plant | [ref. 71, ref. 152] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 81] | ||
| Gomphrena globosa | Inflorescence | [ref. 94, ref. 98, ref. 150] | ||
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 150 | cis‐Ferulic acid | Gomphrena globosa | Flowers | [ref. 150] |
| 151 | cis‐Ferulic acid hexoside | Gomphrena globosa | Flowers | [ref. 150] |
| 152 | trans‐Ferulic acid hexoside | Gomphrena globosa | Flowers | [ref. 150] |
| 153 | Isoferulic acid | Gomphrena elegans Mart. | Leaves | |
| 154 | p‐Coumaric acid or trans‐p‐coumaric acid | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena globosa | Inflorescence | [ref. 94, ref. 98, ref. 150] | ||
| Gomphrena perennis | Aerial parts | [ref. 134] | ||
| Gomphrena haageana K. | Flower | [ref. 73] | ||
| 155 | Sinapic acid | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Iresine angustifolia | Whole plant | [ref. 103] | ||
| 156 | Vanillic acid | Alternanthera philoxeroides | Aerial parts and whole plant | [ref. 38, ref. 132] |
| Alternanthera sessilis | — | [ref. 57] | ||
| Blutaparon portulacoides | Stems, roots, and whole plant | [ref. 71, ref. 148, ref. 152] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 78, ref. 81, ref. 157] | ||
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| Gallic acid derivatives | ||||
| 157 | Ellagic acid | Alternanthera sessilis | Whole plant | [ref. 133] |
| 158 | Syringic acid | Alternanthera philoxeroides | Leaves | [ref. 38, ref. 141] |
| Coumarins | ||||
| 159 | 7‐Methoxycoumarin | Alternanthera caracasana | Aerial parts | [ref. 33] |
| Lignans | ||||
| 160 | Pinoresinol‐4″‐O‐β‐d‐glucopyranoside | Gomphrena celosioides | Aerial parts | [ref. 92] |
| 161 | Tortoside A | Gomphrena celosioides | Aerial parts | [ref. 92] |
| Coumarinolignoids | ||||
| 162 | Cleomiscosin A | Gomphrena celosioides Mart. | Leaves | [ref. 84] |
| Phenylpropanoid | ||||
| 163 | 3,4‐Dihydroxyphenyl caffeate | Froelichia floridana | Whole plants | [ref. 72] |
| 164 | Safrole | Alternanthera philoxeroides | Leaves | [ref. 141] |
| Phenylpropanoid glycosides | ||||
| 165 | β‐d‐(1‐O‐acetyl‐3,6‐O‐p‐E‐dicoumaroyl)‐fructofuranosyl‐α‐d‐(4′‐O‐acetyl‐2′‐O‐p‐E‐coumaroyl)‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| Sesquiterpene phenol | ||||
| 166 | Dictyoceratin C | Gomphrena celosioides Mart. | Leaves | [ref. 84] |
| Other phenolic compounds | ||||
| 167 | 2‐Ethyl‐4,5‐dimethylphenol | Alternanthera sessilis | Stems | [ref. 57] |
| 168 | Hydrangeifolin I | Gomphrena celosioides | Aerial parts | [ref. 92] |
| 169 | Hydroxytyrosol | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| 170 | 2‐Phenylethyl β‐primeveroside | Gomphrena celosioides | Aerial parts | [ref. 92] |
| 171 | 2‐Phenylethyl β‐rutinoside | Gomphrena celosioides | Aerial parts | [ref. 92] |
| 172 | Salicylic acid | Alternanthera philoxeroides | Leaves | [ref. 38, ref. 141] |
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| 173 | Tannic acid | Alternanthera philoxeroides | Whole plant | [ref. 132] |
TABLE 3: Terpenoids compounds isolated from the Gomphrenoideae subfamily.
| No. | Compound | Species | Parts of plant | References |
|---|---|---|---|---|
| Monoterpenes | ||||
| 174 | Linalool | Gomphrena virgata | Whole plant | [ref. 101] |
| 175 | (−)‐Loliolide | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 176 | Myrcene | Alternanthera philoxeroides | Leaves | [ref. 141] |
| 177 | Neryl acetone | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Monoterpene glycoside | ||||
| 178 | (+)‐Angelicoidenol‐2‐O‐β‐d‐glucopyranoside | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| Sesquiterpenes | ||||
| 179 | 11,12 Acetonide of 11,12,13‐trihydroxydrimene | Tidestromia oblongifolia | Aerial parts | [ref. 128] |
| 180 | α‐Amorphene | Gomphrena virgata | Whole plant | [ref. 101] |
| 181 | Aromadendrene | Gomphrena virgata | Whole plant | [ref. 101] |
| 182 | δ‐Cadinene | Gomphrena virgata | Whole plant | [ref. 101] |
| 183 | α‐Cadinol | Gomphrena virgata | Whole plant | [ref. 101] |
| 184 | cis‐Calamenene | Gomphrena virgata | Whole plant | [ref. 101] |
| 185 | β‐Caryophillene | Gomphrena virgata | Whole plant | [ref. 101] |
| 186 | β‐Elemene | Gomphrena virgata | Whole plant | [ref. 101] |
| 187 | Ilimaquinone | Gomphrena celosioides Mart. | Leaves | [ref. 84] |
| 188 | α‐Ionone | Alternanthera sessilis | Leaves | [ref. 63] |
| 189 | α‐Muurolene | Gomphrena virgata | Whole plant | [ref. 101] |
| 190 | Neodactyloquinone | Gomphrena celosioides Mart. | Leaves | [ref. 84] |
| 191 | Nerolidol | Gomphrena virgata | Whole plant | [ref. 101] |
| 192 | Polygodial | Tidestromia oblongifolia | Aerial parts | [ref. 128] |
| 193 | β‐Selinene | Gomphrena virgata | Whole plant | [ref. 101] |
| 194 | 11,12,13‐Trihydroxydrimene | Tidestromia oblongifolia | Aerial parts | [ref. 128] |
| 195 | 3β,7α,14‐Trihydroxy‐Δ8,9‐drimen‐11,12‐olide | Iresine diffusa | Aerial parts | [ref. 105] |
| 196 | 3β,7β,14‐Trihidroxy‐Δ8,9‐drimen‐11,12‐olide | Iresine diffusa | Aerial parts | [ref. 105] |
| 197 | 6,10,14‐Trimetil‐2‐pentadecanone or phytone | Alternanthera sessilis | Stems | [ref. 57] |
| Gomphrena virgata | Whole plant | [ref. 101] | ||
| Diterpenoids | ||||
| 198 | Fitone | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Alternanthera sessilis | Leaf | [ref. 158] | ||
| 199 | Gibberellin | Alternanthera sessilis | Stems | [ref. 56] |
| 200 | Jatropone | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 201 | Neophytadiene | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Alternanthera sessilis | Stems | [ref. 57, ref. 158] | ||
| 202 | Phytol | Alternanthera bettzickiana | — | [ref. 19] |
| Alternanthera brasiliana | Aerial parts | [ref. 22] | ||
| Alternanthera philoxeroides | — | [ref. 41] | ||
| Alternanthera sessilis | Stems and leaves | [ref. 57, ref. 158] | ||
| Triterpenes | ||||
| 203 | α‐Amyrin | Alternanthera brasiliana | Aerial parts and whole plant | [ref. 22, ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 204 | α‐Amyrin acetate | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 205 | α‐Amyrin‐3‐O‐β‐d‐glucopyranoside | Iresine diffusa | Aerial parts | [ref. 105] |
| 206 | β‐Amyrin | Alternanthera brasiliana | Aerial parts and whole plant | [ref. 22, ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 207 | β‐Amyrin‐3‐O‐β‐d‐glucopyranoside | Iresine diffusa | Aerial parts | [ref. 105] |
| 208 | Azadirachtin | Alternanthera sessilis | Whole plant | [ref. 149] |
| 209 | Calenduloside E 6′‐methyl ester | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| 210 | Epitaraxerol | Gomphrena globosa | — | [ref. 156] |
| 211 | Friedelin | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 212 | Glomeric acid | Pfaffia glomerata | Roots | [ref. 154] |
| Pfaffia paniculata | — | [ref. 159] | ||
| 213 | Gypsogenic acid | Pfaffia glomerata | Roots | [ref. 115] |
| 214 | Handianol | Alternanthera sessilis | — | [ref. 64] |
| 215 | 16β‐Hydroxyl‐3‐oxo‐akebonoic acid | Pfaffia glomerata | Roots | [ref. 115] |
| 216 | 16β‐Hydroxyl‐3‐oxo‐akebonoic acid 28‐O‐β‐d‐glucopyranoside | Pfaffia glomerata | Roots | [ref. 115] |
| 217 | Lupeol | Alternanthera sessilis | Leaves | [ref. 62, ref. 158] |
| 218 | Lupeol acetate | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 219 | Mesembryanthemoidigenic acid | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| 220 | 24‐Methylenecycloartanol | Alternanthera philoxeroides | — | [ref. 41] |
| Alternanthera sessilis | — | [ref. 64] | ||
| 221 | Oleanolic acid | Alternanthera philoxeroides | — | [ref. 41] |
| Alternanthera sessilis | — | [ref. 64] | ||
| Pfaffia glomerata | Roots | [ref. 115, ref. 160] | ||
| Pfaffia paniculata | — | [ref. 159] | ||
| 222 | Oleanolic acid 28‐O‐β‐d‐glucopyranoside | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| 223 | Oleanonic acid | Alternanthera philoxeroides | Aerial parts | [ref. 38] |
| Pfaffia glomerata | Inflorescences | [ref. 154] | ||
| 224 | 3‐Oxo‐akebonoic acid | Pfaffia glomerata | Roots | [ref. 115] |
| 225 | Pfaffianol A | Pfaffia glomerata | Roots | [ref. 161] |
| 226 | Pfaffic acid | Hebanthe eriantha | Roots | [ref. 122] |
| Hebanthe paniculata | — | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata Kuntze | Roots | [ref. 123, ref. 125] | ||
| 227 | Pfaffine A | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| Pfaffia glomerata | Roots and aerial parts | [ref. 162] | ||
| 228 | Pfaffine B | Pfaffia paniculata Kuntze | Roots and aerial parts | [ref. 4, ref. 123] |
| 229 | Pfameric acid | Pfaffia glomerata | Roots | [ref. 154] |
| Pfaffia paniculata Kuntze | Roots | [ref. 123] | ||
| 230 | Serratagenic acid | Pfaffia glomerata | Roots | [ref. 115] |
| 231 | Squalene | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena elegans Mart. | Leaves and stem | [ref. 156] | ||
| 232 | Taraxerone | Gomphrena globosa | — | [ref. 156] |
| 233 | Taraxerol | Gomphrena globosa | — | [ref. 156] |
| Triterpenoid saponins | ||||
| 234 | Akebonoic acid | Pfaffia glomerata | Roots | [ref. 161] |
| 235 | Boussingoside A2 | Pfaffia glomerata | Aerial parts and roots | [ref. 4 ] |
| 236 | Calenduloside E | Alternanthera philoxeroides | Whole plant | [ref. 38, ref. 41] |
| Pfaffia glomerata | Roots | |||
| 237 | Chikusetsusaponin IV | Pfaffia glomerata | Inflorescences | [ref. 154] |
| 238 | Chikusetsusaponin IVa | Alternanthera philoxeroides | Aerial parts and whole plant | [ref. 4, ref. 41] |
| Pfaffia glomerata | Roots | [ref. 115, ref. 163] | ||
| 239 | 2α,3β‐Dihydroxyurs‐12,20(30)‐diene‐28‐oic acid‐3‐α‐l‐arabinopyranosyl‐(1 → 2)‐[β‐d‐xylopyranosyl‐(1 → 3)]‐β‐d‐glucopyranosyl | Alternanthera repens | Aerial parts | [ref. 4] |
| 240 | 2α,3β‐Dihydroxyurs‐12,20(30)‐diene‐28‐oic acid‐3‐β‐d‐glucopyranosyl‐(1 → 2)‐α‐l‐arabinopyranosyl‐(1 → 2)‐[β‐d‐xylopyranosyl‐(1 → 3)]‐β‐d‐glucuronopuranoside | Alternanthera repens | Aerial parts | [ref. 4] |
| 241 | 2α,3β‐Dihydroxyurs‐12,20(30)‐diene‐28‐oic acid‐3‐β‐d‐quinovopyranosyl‐(1 → 2)‐α‐l‐arabinopyranosyl‐(1 → 2)‐[β‐d‐xylopyranosyl‐(1 → 3)]‐β‐d‐glucopyranosyl | Alternanthera repens | Aerial parts | [ref. 4] |
| 242 | 2α,3β‐Dihydroxyurs‐12,20(30)‐diene‐28‐oic acid‐3‐β‐d‐xylopyranosyl‐(1 → 3)‐β‐d‐glucopyranosyl | Alternanthera repens | Aerial parts | [ref. 4] |
| 243 | 11α,12α‐epoxy‐3β‐[(O‐β‐d‐galactopyranosyl‐(1 → 3)‐O‐[β‐d‐glucopyranosyl‐(1 → 2)]‐β‐d‐glucuronopyranosyl)‐oxy]olean‐28,13‐olide | Gomphrena macrocephala | Roots | [ref. 99] |
| 244 | 11α,‐12α‐Epoxy‐3β‐[(O‐β‐d‐glucuronopyranosyl)oxy]olean‐28,13‐olide | Gomphrena macrocephala | Roots | [ref. 99] |
| 245 | 11α,‐12α‐Epoxy‐3β‐[(O‐β‐d‐glucuronopyranosyl)oxy]taraxer‐14‐en‐28‐oic acid β‐d‐glucopyranosyl ester | Gomphrena macrocephala | Roots | [ref. 99] |
| 246 | 11α,12α‐Epoxy‐3β‐hydroxyolean‐28,13‐olide | Gomphrena macrocephala | Roots | [ref. 99] |
| 247 | 11α,12α‐Epoxy‐3β‐hydroxytaraxer‐14‐en‐28‐oic acid | Gomphrena macrocephala | Roots | [ref. 99] |
| 248 | Ginsenoside R0 | Pfaffia glomerata | Inflorescences | [ref. 154] |
| 249 | 3β‐O‐(β‐d glucopyranosyluronic acid) 28‐O‐β‐d‐Glucopyranosyl oleanolic acid | Alternanthera sessilis | — | [ref. 64] |
| 250 | Gomphrenoside | Gomphrena globosa | Aerial parts | [ref. 4] |
| 251 | Oleanolic acid‐3‐β‐d‐glucopyranosyl | Alternanthera philoxeroides | Aerial parts | [ref. 4] |
| 252 | Oleanolic acid 3‐O‐β‐d‐glucuronopyranoside | Alternanthera philoxeroides | — | [ref. 39] |
| 253 | Pfaffiaglycoside A | Pfaffia glomerata | Aerial parts and roots | [ref. 4, ref. 161] |
| 254 | Pfaffiaglycosides B | Pfaffia glomerata | Aerial parts and roots | [ref. 4, ref. 161, ref. 163] |
| 255 | Pfaffoside A | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata | Roots | [ref. 125] | ||
| 256 | Pfaffoside B | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata | Roots | [ref. 125] | ||
| 257 | Pfaffoside C | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 161] | ||
| Pfaffia paniculata | Roots | [ref. 125] | ||
| 258 | Pfaffoside D | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata | Roots | [ref. 164] | ||
| 259 | Pfaffoside E | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata | Roots | [ref. 164] | ||
| 260 | Pfaffoside F | Hebanthe eriantha | Roots | [ref. 126] |
| Hebanthe paniculata | Roots | [ref. 121] | ||
| Pfaffia glomerata | Roots | [ref. 112] | ||
| Pfaffia paniculata | Roots | [ref. 164] | ||
| 261 | Philoxeroideside A | Alternanthera philoxeroides | Aerial parts | [ref. 37] |
| 262 | Philoxeroideside B | Alternanthera philoxeroides | Aerial parts | [ref. 37] |
| 263 | Philoxeroideside C | Alternanthera philoxeroides | Aerial parts | [ref. 37] |
| 264 | Philoxeroideside D | Alternanthera philoxeroides | Aerial parts | [ref. 37] |
| Carotenoids | ||||
| 265 | Astaxanthin | Alternanthera sessilis | Whole plant | [ref. 149] |
| 266 | β‐Carotene | Alternanthera sessilis | — | [ref. 64] |
| 267 | Dihydroactinidiolide | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Alternanthera sessilis | Leaf | [ref. 158] | ||
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| Drimene | ||||
| 268 | 3β,14‐Dihydroxy‐Δ7,8‐drimen‐11,12‐acetonide | Iresine diffusa | Aerial parts | [ref. 105] |
TABLE 4: Alkaloid compounds isolated from the Gomphrenoideae subfamily.
| No. | Compound | Species | Parts of plant | References |
|---|---|---|---|---|
| Guanidine alkaloids | ||||
| 269 | Celosiadine A | Iresine diffusa | Aerial parts | [ref. 165] |
| 270 | Celosiadine B | Iresine diffusa | Aerial parts | [ref. 165] |
| Indole alkaloid | ||||
| 271 | Bruceolline F | Gomphrena celosioides Mart. | Leaves | [ref. 84] |
| 272 | β‐Carboline | Alternanthera philoxeroides | Leaves | [ref. 42] |
| Pyridine alkaloids | ||||
| 273 | Trigonelline | Iresine herbstii | — | [ref. 111] |
| Alkaloids with phenethylamine nucleus | ||||
| 274 | Alternamide A (7,8‐dihydroxy‐1,2,4,5‐tetrahydro‐3H‐1,5‐ethano[c]azepin‐3‐one) | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| 275 | Alternamide B (6,7‐dihydroxy‐3,4‐dihydroquinoline‐1‐one) | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| Betalains | ||||
| Amaranthin group (betacyanins) | ||||
| 276 | Amaranthine (previously named amaranthin) | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Alternanthera brasiliana | Leaves | [ref. 27, ref. 28] | ||
| Alternanthera ficoidea | Leaves | [ref. 166] | ||
| Alternanthera tenella | Leaves and stems | [ref. 28] | ||
| Colla | ||||
| Gomphrena globosa | Petals and inflorescences | [ref. 29, ref. 166, ref. 167, ref. 168] | ||
| Iresine herbstii | Leaves | [ref. 110, ref. 111] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 277 | Celosianin I | Alternanthera bettzickiana | Leaves | [ref. 166] |
| 278 | Celosianin II or celosianin | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Gomphrena globosa | Red petals and red flowers | [ref. 29, ref. 167, ref. 170] | ||
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 279 | 17‐Decarboxy‐amaranthin | Gomphrena globosa | Red petals and red flowers | [ref. 29, ref. 150, ref. 167, ref. 171] |
| 280 | 17‐Decarboxy‐isoamaranthine | Gomphrena globosa | Red inflorescences | [ref. 29, ref. 170] |
| 281 | 2‴‐O‐E‐Feruloyl‐iresinin or (2‴‐O‐E‐feruloyl)‐iresinin I | Iresine herbstii | Leaves | [ref. 110, ref. 169] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 282 | 2‴‐O‐E‐Feruloyl‐isoiresinin or (2‴‐O‐E‐Feruloyl)‐isoiresinin I | Iresine herbstii | Leaves | [ref. 110] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 283 | Iresin | Iresine celosia | Aerial parts | [ref. 104] |
| Iresine diffusa | Aerial parts | [ref. 105] | ||
| 284 | Iresinin previously named iresinin I | Alternanthera brasiliana | Leaves | [ref. 27] |
| Iresine herbstii | Leaves | [ref. 110, ref. 111, ref. 166, ref. 172] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 285 | Iresinin II (=isoiresinine I) | Iresine herbstii | Leaves | [ref. 110, ref. 111, ref. 166] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 286 | Iresinin III | Iresine herbstii | — | [ref. 111] |
| 287 | Iresinin IV | Iresine herbstii | — | [ref. 111] |
| 288 | Isoamaranthine (Isoamaranthin) | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Alternanthera brasiliana | Leaves | [ref. 27, ref. 28] | ||
| Alternanthera ficoidea | Leaves | [ref. 166] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena globosa | Red and orange petals | [ref. 29, ref. 167] | ||
| Iresine herbstii | Leaves | [ref. 110, ref. 111, ref. 166] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 289 | Isocelosianin or isocelosianin II or (2″‐O‐E‐sinapoyl)‐amaranthine or lindenin | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Gomphrena globosa | Red petals and red flowers | [ref. 29, ref. 170 ] | ||
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 290 | Sinapoyl‐amaranthin | Gomphrena globosa | Red petals and red flowers | [ref. 29, ref. 167, ref. 170] |
| 291 | 2‴‐O‐E‐sinapoyl‐iresinin or (2‴‐O‐E‐sinapoyl)‐iresinin I | Iresine herbstii | Leaves | [ref. 110] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 292 | (2″‐O‐E‐Sinapoyl)‐isoamaranthine or Isolindenin | Iresine herbstii | Leaves | [ref. 110] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 293 | 2‴‐O‐E‐sinapoyl‐isoiresinin or (2‴‐O‐E‐sinapoyl)‐isoiresinin I | Iresine herbstii | Leaves | [ref. 110] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| Betanin group (betacyanins) | ||||
| 294 | Betanidin | Gomphrena globosa | Red and purple petals | [ref. 29] |
| 295 | Betanin | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Alternanthera brasiliana | Leaves and stems | [ref. 27, ref. 28] | ||
| Alternanthera ficoidea | Leaves | [ref. 166] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena globosa | Red petals | [ref. 29, ref. 167] | ||
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 296 | Isobetanidin | Gomphrena globosa | Purple petals | [ref. 29, ref. 167] |
| 297 | Isobetanin | Alternanthera bettzickiana | Leaves | [ref. 166] |
| Alternanthera brasiliana | Leaves and stems | [ref. 28] | ||
| Alternanthera ficoidea | Leaves | [ref. 166] | ||
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| Gomphrena globosa | Red petals | [ref. 29, ref. 167] | ||
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| Gomphrenin group (betacyanins) | ||||
| 298 | cis‐Isomer of gomphrenin II | Gomphrena globosa | Purple flower | [ref. 167, ref. 168, ref. 173] |
| 299 | cis‐Isomer of gomphrenin III | Gomphrena globosa | Pigmented floral parts from the inflorescences | [ref. 167, ref. 168, ref. 173, ref. 174] |
| 300 | cis‐Isomer of isogomphrenin II | Gomphrena globosa | Purple petals | [ref. 167] |
| 301 | cis‐Isomer of isogomphrenin III | Gomphrena globosa | Purple petals | [ref. 167] |
| 302 | Gomphrenin | Gomphrena celosioides | — | [ref. 84] |
| Gomphrena globosa | Flowers, bract, and bracteoles | [ref. 29, ref. 175] | ||
| 303 | Gomphrenin I | Gomphrena globosa | Pigmented floral parts from the inflorescences | [ref. 94, ref. 98, ref. 166, ref. 168, ref. 173, ref. 174, ref. 176] |
| 304 | Gomphrenin II or globosin | Gomphrena globosa | Pigmented floral parts from the inflorescences | [ref. 94, ref. 98, ref. 150, ref. 166, ref. 167, ref. 168, ref. 173, ref. 174, ref. 176] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| 305 | Gomphrenin III or basellin | Gomphrena globosa | Pigmented floral parts from the inflorescences | [ref. 94, ref. 98 ref. 150, ref. 166, ref. 167, ref. 168, ref. 173, ref. 174, ref. 176] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 306 | Gomphrenin IV | Gomphrena globosa | Inflorescences | [ref. 175] |
| 307 | Isogomphrenin | Gomphrena globosa | Flowers | [ref. 175] |
| 308 | Isogomphrenin I | Gomphrena globosa | Pigmented floral parts from the inflorescences | [ref. 94, ref. 98, ref. 166, ref. 168, ref. 173, ref. 174, ref. 176] |
| 309 | Isogomphrenin II or Isoglobosin | Gomphrena globosa | Inflorescence | [ref. 29, ref. 94, ref. 98, ref. 150, ref. 166, ref. 167, ref. 168, ref. 176] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| 310 | Isogomphrenin III or isobasellin | Gomphrena globosa | Pigmented floral parts from the inflorescences, bract, and bracteoles | [ref. 29, ref. 94, ref. 98, ref. 150, ref. 166, ref. 167, ref. 168, ref. 173, ref. 174, ref. 176–] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| Iresine lindenii | Leaves | [ref. 169] | ||
| 311 | Isosinapoyl‐gomphrenin I or isogandolin | Gomphrena globosa | Purple flowers | [ref. 173] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| 312 | Sinapoyl‐gomphrenin I or gandolin | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98, ref. 167, ref. 168, ref. 173] |
| Iresine herbstii | Leaves | [ref. 110] | ||
| 313 | Sinapoyl‐isogomphrenin I | Gomphrena globosa | Inflorescence | [ref. 94, ref. 98, ref. 167, ref. 168] |
| Other betalains | ||||
| 314 | Hylocerenin | Iresine herbstii | Leaves | [ref. 110] |
| Iresine lindenii | Leaves | [ref. 169] | ||
| 315 | Isohylocerenin | Iresine lindenii | Leaves | [ref. 169] |
| Betaxanthins | ||||
| 316 | Arginine‐betaxanthin | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| 317 | Dopamine‐betaxanthine | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 318 | Glutamine‐betaxanthin (vulgaxanthin I) | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| 319 | Histidine‐betaxanthin (muscaarin VII) | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| 320 | Isoleucine‐betaxanthin | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| 321 | Lysine‐betaxanthin | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| 322 | 3‐Methoxytyramine‐betaxanthin | Alternanthera brasiliana | Leaves and stems | [ref. 28] |
| Alternanthera tenella Colla | Leaves and stems | [ref. 28] | ||
| 323 | Tryptophan‐betaxanthin | Gomphrena globosa | Red petals | [ref. 29, ref. 167] |
| Other alkaloids | ||||
| 324 | Alternamine A ((R)‐1‐(3,4‐dihydroxyphenyl)‐1,2,3,4‐tetrahydroisoquinoline‐6,7‐diol) | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| 325 | Alternamine B (4‐(2‐aminoethyl)benzene‐1,2‐diol‐4‐(2‐aminoethyl)benzene‐1,2‐diol‐b‐d‐glucopyranose) | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| 326 | Aurantiamide | Gomphrena celosioides Mart. | Whole plant | [ref. 83–ref. 85, ref. 89] |
| 327 | Aurantiamide acetate | Gomphrena agrestis | Whole plant | [ref. 2] |
| Gomphrena celosioides Mart. | Whole plant | [ref. 83, ref. 85, ref. 89] | ||
| 328 | N‐(3,4‐dihydroxyphenethyl)formamide | Alternanthera littoralis P. Beauv. | Aerial parts | [ref. 8] |
| 329 | N‐feruloyl‐tyramine | Iresine herbstii | — | [ref. 111] |
| 330 | Pyrimidine‐2,4 (1H, 3H)‐dione (uracil) | Gomphrena elegans Mart. | Leaves, roots, and stem | [ref. 156] |
TABLE 5: Lipid compounds isolated from Gomphrenoideae subfamily.
| No. | Compound | Species | Parts of plant | References |
|---|---|---|---|---|
| Fatty acids | ||||
| 331 | Arachidonic acid | Alternanthera sessilis | Whole plant | [ref. 149] |
| 332 | Butyl hexadecanoate | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 333 | Ethyl linolenate | Gomphrena elegans Mart. | Leaves, root, and stem | [ref. 156] |
| 334 | Ethyl linoleolate | Gomphrena elegans Mart. | Leaves, root, and stem | [ref. 156] |
| 335 | Ethyl palmitate or ethyl hexadecanoate | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera sessilis | Stems | [ref. 56] | ||
| Gomphrena elegans Mart. | Leaves, root, and stem | [ref. 156] | ||
| Pfaffia glomerata | Roots | [ref. 177] | ||
| 336 | Hexadecanoate | Alternanthera sessilis | Stems | [ref. 56] |
| 337 | (8E)‐10‐Hydroxy‐8‐octadecenoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 338 | (10E)‐9‐Hydroxy‐10‐octadecenoic | Alternanthera brasiliana | Stems | [ref. 32] |
| 339 | (8E,12Z)‐10‐Hydroxy 8,12‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 340 | (9Z,11E)‐13‐Hydroxy‐9,11‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 341 | (9Z,11E,15Z)‐13‐Hydroxy‐9,11,15‐octadecatrienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 342 | (9Z,12Z,14E)‐16‐Hydroxy‐9,12,14‐octadecatrienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 343 | (9Z,13E)‐12‐Hydroxy‐9,13‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 344 | (9Z,13E,15Z)‐12‐Hydroxy‐9,13,15‐octadecatrienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 345 | (10E,12E)‐9‐Hydroxy‐10,12‐Octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 346 | (10E,12Z)‐9‐Hydroxy‐10,12‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 347 | Linoleic acid | Alternanthera bettzickiana | — | [ref. 19] |
| Alternanthera brasiliana | Aerial parts | [ref. 22] | ||
| 348 | Linoleic acid ethyl ester | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 349 | Methyl linoleate or linoleic acid methyl ester | Alternanthera sessilis | Stems | [ref. 56] |
| Gomphrena elegans Mart. | Leaves and stem | [ref. 156] | ||
| 350 | Methyl linolenate | Gomphrena elegans Mart. | Leaves and stem | [ref. 156] |
| 351 | Methyl palmitate or methyl hexadecanoate | Gomphrena celosioides | Roots | [ref. 157] |
| Gomphrena elegans Mart. | Leaves, stem | [ref. 156] | ||
| Alternanthera sessilis | Leaves | [ref. 158] | ||
| 352 | Methyl stearate | Gomphrena elegans Mart. | — | [ref. 156] |
| 353 | Miristic acid | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| 354 | (9Z)‐9‐octadecenedioic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 355 | Oleic acid | Alternanthera brasiliana | Whole plant | [ref. 23] |
| 356 | (7E)‐9‐oxo‐7‐octadecenoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 357 | (8E)‐10‐oxo‐8‐octadecenoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 358 | (9E,11E)‐13‐oxo‐9,11‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 359 | (9Z,11E)‐13‐oxo‐9,11‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 360 | (10E,12E)‐9‐oxo‐10,12‐octadecadienoic acid | Alternanthera brasiliana | Stems | [ref. 32] |
| 361 | (10E,12Z)‐9‐oxo‐10,12‐octadecadienoic | Alternanthera brasiliana | Stems | [ref. 32] |
| 362 | Palmitic acid | Alternanthera brasiliana | Aerial parts and whole plant | [ref. 22, ref. 23] |
| Alternanthera sessilis | Stems, leaves, and whole plant | [ref. 57, ref. 149, ref. 158] | ||
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| 363 | Stearic acid | Alternanthera brasiliana | Aerial parts and whole plant | [ref. 22, ref. 23] |
| Fatty alcohol | ||||
| 364 | 1‐Hexadecanol | Gomphrena elegans Mart. | Leaves and stem | [ref. 156] |
| Fatty amide | ||||
| 365 | Erucamide | Alternanthera brasiliana | Aerial parts | [ref. 22] |
TABLE 6: Other compounds isolated from the Gomphrenoideae subfamily.
| No. | Compound | Species | Parts of plant | References |
|---|---|---|---|---|
| Phytoecdysones | ||||
| 366 | Ecdysone | Pfaffia glomerata | Inflorescences, roots, and aerial parts | [ref. 154, ref. 162, ref. 163] |
| Pfaffia paniculata | Roots | [ref. 123] | ||
| 367 | β‐Ecdysone or 1α,20R‐dihydroxyecdysone or ecdysterone or 20‐hydroxyecdysone | Froelichia floridana | Seeds and whole plant | [ref. 72] |
| Gomphrena celosioides | Roots and aerial parts | [ref. 133 ] | ||
| Gomphrena virgata | Roots | [ref. 101] | ||
| Pfaffia glomerata | Inflorescences, stems, roots, and aerial parts | [ref. 154, ref. 159, ref. 161, ref. 162, ref. 179, ref. 180, ref. 181, ref. 182, ref. 183, ref. 184] | ||
| Pfaffia paniculata Kuntze | Roots | [ref. 123] | ||
| Phytoecdysteroids | ||||
| 368 | Achyranthesterone A | Froelichia floridana | Whole plants | [ref. 72] |
| 369 | Blechnoside B | Froelichia floridana | Whole plants | [ref. 72] |
| 370. | 2‐Dehydro‐3‐epi‐20‐hydroxyecdysone | Froelichia floridana | Seeds | [ref. 182] |
| 371 | 2,22‐Dideoxyecdysone 25‐O‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 372 | 2,22‐Dideoxyecdysone 25‐O‐β‐d‐glucopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 373 | (5α)‐2,22‐Dideoxyecdysone 25‐O‐β‐d‐glucopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 374 | 2,22‐Dideoxy‐20‐hydroxyecdysone 25‐O‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 375 | 2,22‐dideoxy‐5β‐hydroxyecdysone 25‐O‐β‐d‐glucopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside | Froelichia floridana | Whole plants | [ref. 72] |
| 376 | β‐Glucopyranosil oleanolate | Pfaffia paniculata | — | [ref. 159] |
| 377 | 22‐Oxo‐20‐hydroxyecdysone | Pfaffia glomerata | Roots | [ref. 161] |
| 378 | Pfaffiaglycoside C | Pfaffia glomerata | Roots | [ref. 161] |
| 379 | Pfaffiaglycoside D | Pfaffia glomerata | Roots | [ref. 161] |
| 380 | Pfaffiaglycoside E | Pfaffia glomerata | Roots | [ref. 161] |
| 381 | Pterosterone | Pfaffia glomerata | Roots | [ref. 161] |
| Pfaffia paniculata Kuntze | Roots | [ref. 123] | ||
| 382 | Rapisterone | Pfaffia paniculata Kuntze | Roots | [ref. 123] |
| 383 | Rubrosterone | Pfaffia glomerata | Roots, aerial parts, and roots | [ref. 154, ref. 162] |
| Pfaffia paniculata | — | [ref. 159] | ||
| 384 | Taxisterone | Pfaffia glomerata | Roots | [ref. 161] |
| 385 | 2β,3β,14α,17β‐Tetrahydroxy‐5β‐androst‐7‐en‐6‐one | Pfaffia glomerata | Roots | [ref. 161] |
| Phytosterols | ||||
| 386 | Campesterol | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| Alternanthera sessilis | — | [ref. 62] | ||
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| Blutaparon portulacoides | Aerial parts | [ref. 148] | ||
| Gomphrena celosioides Mart. | Aerial Parts | [ref. 78, ref. 157] | ||
| Gomphrena globosa | — | [ref. 156] | ||
| Pfaffia glomerata | Roots | [ref. 177] | ||
| 387 | Campestrol | Gomphrena celosioides Mart. | Aerial parts | [ref. 81] |
| 388 | Cycloeucalenol | Alternanthera philoxeroides | — | [ref. 41] |
| Alternanthera sessilis | — | [ref. 64] | ||
| 389 | 3‐β‐Hydroxystigmast‐5‐en‐7‐one | Alternanthera philoxeroides | — | [ref. 41] |
| 390 | Sitostenone or β‐sitostenone | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| 391 | Sitosterol or β‐sitosterol | Alternanthera brasiliana | Whole plant and leaves | [ref. 23, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| Alternanthera philoxeroides | — | [ref. 41] | ||
| Alternanthera sessilis | — | [ref. 62] | ||
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| Blutaparon portulacoides | Aerial parts | [ref. 148] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides Mart. | Aerial parts | [ref. 78, ref. 81, ref. 157] | ||
| Gomphrena elegans Mart. | Leaves, roots, and stem | [ref. 156] | ||
| Gomphrena globosa | — | [ref. 156] | ||
| Hebanthe paniculata | — | [ref. 121] | ||
| Iresine diffusa | Aerial parts | [ref. 105] | ||
| Pfaffia glomerata | Roots | [ref. 112, ref. 177] | ||
| Pfaffia paniculata | Roots | [ref. 183] | ||
| Tidestromia oblongifolia | Aerial parts | [ref. 128] | ||
| 392 | β‐Sitosteryl‐β‐O‐d‐glucopyranoside | Iresine diffusa | Aerial parts | [ref. 105] |
| 393 | γ‐Sitosterol | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 394. | Sitosterol glycoside or 3‐O‐β‐d‐glucopyranosyl β‐sitosterol | Alternanthera brasiliana | — | [ref. 32] |
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| 395 | Sitosteryl | Blutaparon portulacoides | Roots | [ref. 148, ref. 151] |
| 396 | Spinasterol or α‐spinasterol | Alternanthera brasiliana | Leaves and whole plant | [ref. 23, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| Alternanthera philoxeroides | — | [ref. 41] | ||
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| Alternanthera sessilis | — | [ref. 64] | ||
| Pfaffia glomerata | Roots | [ref. 177] | ||
| 397 | Δ7‐Spinasterol | Pfaffia glomerata | Roots | [ref. 177] |
| 398 | β‐Spinasterol | Alternanthera sessilis | — | [ref. 64] |
| 399 | Spinasteryl β‐d‐glucopyranoside | Blutaparon portulacoides | Roots | [ref. 148] |
| 400 | 5α‐Stigmasta‐7,22‐dien‐3β‐ol | Gomphrena elegans Mart. | Leaves, roots, and stem | [ref. 156] |
| 401 | 5α‐Stigmasta‐enol | Alternanthera sessilis | — | [ref. 64] |
| 402 | 5α‐Stigmasta‐7‐enol | Alternanthera sessilis | — | [ref. 184] |
| 403 | Stigmasta 4,6,22 trien‐3‐α‐ol | Alternanthera brasiliana | Whole plant | [ref. 23] |
| 404 | Stigmasta 4,7,22 trien‐3‐β‐ol | Alternanthera brasiliana | Whole plant | [ref. 23] |
| 405 | Δ7‐Stigmastenol | Pfaffia glomerata | Roots | [ref. 177] |
| 406 | Stigmast‐7‐en‐3‐ol | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 407 | Stigmast‐7en‐3β‐ol | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| 408 | Stigmast‐22‐en‐3β‐ol | Tidestromia oblongifolia | Aerial parts | [ref. 128] |
| 409 | Stigmast‐7‐enyl | Blutaparon portulacoides | Roots | [ref. 148] |
| 410 | Stigmasterol | Alternanthera brasiliana | Leaves and whole plant | [ref. 23, ref. 28] |
| Alternanthera maritima | Aerial parts | [ref. 138] | ||
| Alternanthera sessilis | — | [ref. 62] | ||
| Alternanthera tenella Colla | Whole plant | [ref. 142] | ||
| Blutaparon portulacoides | Aerial parts | [ref. 148] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena celosioides Mart. | Aerial parts and roots | [ref. 78, ref. 81, ref. 157] | ||
| Gomphrena globosa | — | [ref. 156] | ||
| Hebanthe paniculata | — | [ref. 121] | ||
| Iresine diffusa | Aerial parts | [ref. 105] | ||
| Pfaffia glomerata | Roots | [ref. 177] | ||
| Pfaffia paniculata | Roots | [ref. 183] | ||
| Tidestromia oblongifolia | Aerial parts | [ref. 128] | ||
| 411 | Δ7‐Stigmasterol | Alternanthera tenella Colla | Whole plant | [ref. 142] |
| 412 | Stigmasteryl 3‐β‐O‐glucoside 6′‐O‐palmitate | Blutaparon portulacoides | Roots | [ref. 151] |
| Phytosteroids | ||||
| 413 | 4,6 Cholestadien‐3‐beta‐ol | Alternanthera brasiliana | Whole plant | [ref. 23] |
| Saponins | ||||
| 414 | 3‐O‐β‐d‐glucopyranosyl spinasterol | Alternanthera tenella Colla | Whole plant | [ref. 142] |
| 415 | 3‐O‐β‐d‐glucopyranosyl stigmasterol | Alternanthera tenella Colla | Whole plant | [ref. 142] |
| 416 | 3‐O‐β‐d‐glucopyranosyl Δ7‐stigmasterol | Alternanthera tenella Colla | Whole plant | [ref. 142] |
| 417 | Stigmast‐6‐en‐3‐O‐β‐(d‐glicopiranoside) | Gomphrena celosioides | Roots | [ref. 157] |
| Aliphatic alcohols | ||||
| 418 | Triacontanol | Iresine diffusa | Aerial parts | [ref. 105] |
| Aliphatic hydrocarbons | ||||
| 419 | cis‐Jasmone | Gomphrena virgata | Whole plant | [ref. 101] |
| 420 | Docosane | Pfaffia glomerata | Roots | [ref. 177] |
| 421 | Docosano | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 422 | Docoseno | Gomphrena elegans Mart. | Stem, root and leaves | [ref. 156] |
| 423 | Eicosene | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 424 | 3‐Eicosene | Gomphrena elegans Mart. | — | [ref. 156] |
| 425 | 9‐Eicosene | Gomphrena elegans Mart. | — | [ref. 156] |
| 426 | Heptacosane | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 427 | Heptadecane | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 428 | Hexacosane | Pfaffia glomerata | Roots | [ref. 177] |
| 429 | 1‐Hexacosene | Gomphrena elegans Mart. | Roots | [ref. 156] |
| 430 | Hexacosano | Gomphrena elegans Mart. | Roots | [ref. 156] |
| 431 | Hexadecane | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 432 | Nonadecane | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| Pfaffia glomerata | Roots | [ref. 177] | ||
| 433 | Octadecane | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| Pfaffia glomerata | Roots | [ref. 177] | ||
| 434 | 1‐Octadecene | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 435 | Pentacosane | Pfaffia glomerata | Roots | [ref. 177] |
| 436 | Pentacosano | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 437 | Pentadecane | Gomphrena elegans Mart. | Leaves and stem | [ref. 156] |
| 438 | Tetracosane | Pfaffia glomerata | Roots | [ref. 177] |
| 439 | Tetracosano | Gomphrena elegans Mart. | Roots and stem | [ref. 156] |
| 440 | 7,11,15‐Trimethyl‐3‐methylenehexadec‐1‐ene | Gomphrena elegans Mart. | Leaves and stem | [ref. 156] |
| Alkane | ||||
| 441 | Eicosane | Pfaffia glomerata | Roots | [ref. 177] |
| 442 | 16‐Hentriacontane | Alternanthera sessilis | — | [ref. 64] |
| 443 | Heptadecane | Gomphrena virgata | Whole plant | [ref. 101] |
| 444 | Pentadecane | Gomphrena virgata | Whole plant | [ref. 101] |
| 445 | Tetradecane | Gomphrena virgata | Whole plant | [ref. 101] |
| 446 | 2,6,10‐Trimethyldodecane | Gomphrena virgata | Whole plant | [ref. 101] |
| Carboxylic acid | ||||
| 447 | Citric acid | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena globosa | — | [ref. 97] | ||
| 448 | Fumaric acid | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena globosa | — | [ref. 97] | ||
| 449 | 3‐(4‐Hydroxyphenyl) methylpropenoate | Gomphrena celosioides | Whole plant | [ref. 82] |
| 450 | Malic acid | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena globosa | — | [ref. 97] | ||
| 451 | Methylsalicylate | Gomphrena virgata | Whole plant | [ref. 101] |
| 452 | Oxalic acid | Gomphrena globosa | — | [ref. 97] |
| 453 | Quinic acid | Gomphrena globosa | Flowers | [ref. 153] |
| Feruloyl tyramine | ||||
| 454 | N‐cis‐feruloyl tyramine | Alternanthera philoxeroides | Aerial parts | [ref. 36] |
| 455 | N‐trans‐feruloyl tyramine | Alternanthera philoxeroides | Aerial parts | [ref. 36] |
| 456 | N‐trans‐feruloyl‐3,5‐dimethoxytyramine | Alternanthera philoxeroides | Aerial parts | [ref. 36] |
| 457 | N‐trans‐feruloyl‐3‐methyldopamine | Alternanthera philoxeroides | Aerial parts | [ref. 36] |
| Heterocyclic compounds | ||||
| 458 | 4H‐Pyran‐4‐one,2,3‐dihydro‐3,5‐dihydroxy‐6‐methyl | Alternanthera sessilis | Whole plant | [ref. 149] |
| 459 | 2‐Methoxy‐3‐isopropylpyrazine | Gomphrena virgata | Whole plant | [ref. 101] |
| Hydrocarbon | ||||
| 460 | α‐Copaene | Gomphrena virgata | Whole plant | [ref. 101] |
| 461 | Nonacosane | Alternanthera sessilis | — | [ref. 64] |
| Gomphrena elegans Mart. | Leaves | [ref. 156] | ||
| 462 | Tricosano | Gomphrena elegans Mart. | Roots | [ref. 156] |
| Organic acid | ||||
| 463 | Gluconic acid | Pfaffia glomerata | Inflorescences and aerial parts | [ref. 154, ref. 162] |
| Tocopherols | ||||
| 464 | α‐Tocopherol | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Alternanthera brasiliana | Aerial parts | [ref. 22] | ||
| Gomphrena agrestis | Roots and leaves | [ref. 139] | ||
| Gomphrena globosa | — | [ref. 97] | ||
| 465 | α‐Tocopherol acetate | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 466 | γ‐Tocopherol | Alternanthera bettzickiana | Aerial parts | [ref. 19] |
| Gomphrena globosa | — | [ref. 97] | ||
| 467 | δ‐Tocopherol | Gomphrena agrestis | Roots and leaves | [ref. 139] |
| Gomphrena globosa | — | [ref. 97] | ||
| Vitamins | ||||
| 468 | Riboflavin | Alternanthera brasiliana | — | [ref. 185] |
| 469 | Niacin | Alternanthera brasiliana | — | [ref. 185] |
| Other types of compounds | ||||
| 470 | Allantoin | Hebanthe paniculata | — | [ref. 121] |
| Pfaffia paniculata | Roots | [ref. 183] | ||
| 471 | Benzophenone‐4 | Alternanthera sessilis | Stem | [ref. 56] |
| 472 | Butyrolactone | Alternanthera sessilis | Stems | [ref. 57] |
| 473 | Choline | Alternanthera sessilis | — | [ref. 64] |
| 474 | 2‐Decenal | Alternanthera brasiliana | Whole plant | [ref. 23] |
| 475 | Diisobutil Phthalate | Gomphrena elegans Mart. | Stem | [ref. 156] |
| 476 | 7,9‐di‐ter‐butyl‐oxaspiro [ref. 4, ref. 5] deca‐6,9‐dien‐2,8‐dione | Gomphrena elegans Mart. | Roots | [ref. 156] |
| 477 | 2,4‐Dihydroxy‐2,5‐dimethyl‐3(2H)‐furan‐3‐one | Alternanthera sessilis | Stems | [ref. 57] |
| 478 | 11‐(3‐Ethenylcyclopentyl)undec‐10‐enoic acid, ethyl ester | Alternanthera sessilis | Stems | [ref. 57] |
| 479 | 3‐Ethyl‐5‐(2‐ethylbutyl)‐octadecane | Gomphrena elegans Mart. | Roots | [ref. 156] |
| 480 | Ethyl 9‐octadecenoate | Gomphrena elegans Mart. | Leaves, root, and stem | [ref. 156] |
| 481 | Formic acid, 2‐propenyl ester | Alternanthera sessilis | Stems | [ref. 57] |
| 482 | Furfural | Alternanthera sessilis | Stems | [ref. 57] |
| 483 | β‐Glucopyranosyl oleanolate | Pfaffia glomerata | Roots | [ref. 154] |
| 484 | l‐Glutamic acid | Alternanthera sessilis | Stems | [ref. 57] |
| 485 | Glutamine, L‐ | Alternanthera sessilis | Stems | [ref. 57] |
| 486 | Glycinebetaine | Iresine herbstii | — | [ref. 111] |
| 487 | Heneicosane | Pfaffia glomerata | Roots | [ref. 177] |
| 488 | 1‐Hexadecene | Gomphrena elegans Mart. | — | [ref. 156] |
| 489 | (E)‐hexyl 2‐methylbut‐2‐enoate | Gomphrena virgata | Whole plant | [ref. 101] |
| 490 | p‐Hydroxycinnamoyl moiety | Alternanthera sessilis | Stems | [ref. 56] |
| 491 | Indole‐3‐carbaldehyde | Alternanthera philoxeroides | Aerial parts | [ref. 38] |
| 492. | Inulin | Pfaffia glomerata | — | [ref. 186] |
| 493 | Laurenan‐2‐ona | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 494 | Levan | Gomphrena marginata | — | [ref. 186] |
| 495 | 8‐Methyl‐1‐decene | Gomphrena elegans Mart. | Stem | [ref. 156] |
| 496 | Methyl octadecanoate | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| Alternanthera sessilis | Leaves | [ref. 158] | ||
| 497 | Methyl 6‐octadecenoate | Gomphrena elegans Mart. | Stem | [ref. 156] |
| 498 | Methyl 8‐octadecenoate | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 499 | 3‐Methyl‐5‐propylnonane | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 500 | Nonanal | Gomphrena virgata | Whole plant | [ref. 101] |
| 501 | Z‐3,17‐Octadecadien‐1‐ol acetate | Alternanthera sessilis | Stems | [ref. 57] |
| 502 | Palmitic acid ethyl ester | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 503 | Pentanal | Alternanthera sessilis | Stems | [ref. 57] |
| 504 | Phaeophytin a | Alternanthera philoxeroides | — | [ref. 41] |
| 505 | Phenylacetaldehyde | Alternanthera sessilis | Stems | [ref. 57] |
| 506 | Pheophytin a′ | Alternanthera philoxeroides | — | [ref. 41] |
| 507 | Sebacic acid, bis(2‐ethylhexyl) ester | Alternanthera sessilis | Stems | [ref. 57] |
| 508 | 6,10,14‐Trimethyl‐2‐pentadecanone | Gomphrena elegans Mart. | Leaves and roots | [ref. 156] |
| 509 | (5Z)‐2,6,10‐Trimethyl‐1,5,9‐undecatriene | Alternanthera brasiliana | Aerial parts | [ref. 22] |
| 510 | 1,2,4‐Trioxolane,3‐phenyl‐ | Alternanthera sessilis | Stems | [ref. 57] |
| 511 | Uridine | Gomphrena elegans Mart. | Leaves | [ref. 156] |
| 512 | Umbellatosides B | Gomphrena celosioides | Aerial parts | [ref. 178] |
FIGURE 1Chemical compounds identified in the subfamily Gomphrenoideae.
Currently, only 8.22% of the members of Gomphrenoideae have a documented phytochemical profile. Notably, Gomphrena globosa L. has been found to contain 107 compounds, representing 20.94% of the total compounds reported in Gomphrenoideae, including phenolic compounds, terpenoids, alkaloids, lipid compounds, carboxylic acids, and tocopherols. Similarly, Alternanthera brasiliana (L.) Kuntze exhibits a remarkable phytochemical profile with 91 secondary metabolites, including phenolic compounds, terpenoids, alkaloids, lipid compounds, tocopherols, vitamins, among other compounds. In contrast, for other species, fewer than 10 chemical compounds have been reported, examples include Alternanthera paronychioides A.St.‐Hil. (24, 149), Alternanthera repens (Synm. Alternanthera caracasana Kunth) (159, 239–242), Gomphrena boliviana Moq. (6, 7, 9, 10, 17, 54), Gomphrena claussenii Moq. (13, 19, 20, 23–24, 66, 96), Gomphrena macrocephala A.St.‐Hil. (243–247), Gomphrena marginata Seub. (494), Gomphrena martiana Gillies ex Moq. (6–7, 9–10, 17–18, 38, 53–54), Pfaffia townsendii Pedersen (67, 106), and Tidestromia oblongifolia (S. Watson) Standl. (Synm. Tidestromia suffruticosa var. oblongifolia (S. Watson) Sánch.Pino & Flores Olv.) (179, 192, 194, 391, 408, 410).
However, no studies have been conducted on the phytochemical profile of species within the genera Froelichiella, Guilleminea, Hebanthodes, Pedersenia, Pseudoplantago, Quaternella, and Xerosiphon, leaving them chemically unexplored.
Determining the chemical profile of a plant is crucial, as it allows for the deduction of its biological activity, safety, and toxicity. Additionally, it facilitates the study of how internal and external factors influence the production of secondary metabolites and, consequently, biological activity. Research has demonstrated that plants of the same species collected from different locations or at different times, as well as subspecies, can exhibit different phytochemical profiles.
In accordance with the above, it is also worth mentioning that in many cases, the compounds identified in the chemical profile of a plant are produced by its endophytic microorganisms or by the interaction plant‐microorganisms. This association has become a new area of multidisciplinary research of high biotechnological interest.
It is important to emphasize that, to date, there are no studies that evaluate the effect of biotic and abiotic factors on the chemical profile of the members of this subfamily. Additionally, there are no reports on the endophytic communities present in these plants and their effect on the production of secondary metabolites.
Phenolic Compounds
Phenolic compounds are the most numerous and ubiquitously distributed group of secondary plant metabolites [ref. 73]. This group includes all substances that contain phenolic functions linked to aromatic or aliphatic structures [ref. 187].
These compounds exhibit a broad range of biological effects mainly related to their antioxidant capacity due to the presence of hydrogen‐donating hydroxyl groups [ref. 73, ref. 77, ref. 90, ref. 188]. Previous studies have reported that these compounds inhibit cellular DNA mutagenicity and possess antimicrobial and anti‐inflammatory activities [ref. 73, ref. 85, ref. 188].
To date, 173 phenolic compounds have been isolated from the Gomphrenoideae subfamily. In this review, these compounds have been classified into two categories: flavonoids and their glycosides (1–133) and non‐flavonoid phenolic compounds (134–173), as detailed in Sections 4.1.1 and 4.1.2, Table 2, and Figure 1.
Some of these compounds have been subjected to in vivo and in vitro studies to evaluate their biological activity, aiming to propose them as potential therapeutic agents for various diseases.
In terms of antimicrobial activity, compounds 5,7‐dihydroxy‐3,6‐dimethoxyflavone (6), oroxilin A (7), baicalein 5,6‐dimethyl ether (9), 3,5‐dimethoxy‐6,7‐methylenedioxyflavone (10), and 3,5,6,7‐tetramethoxyflavone (17) were evaluated against Mycobacterium phlei, Staphylococcus aureus ATTC 12600, and Enterococcus faecalis ATCC 19433. All were more active against M. phlei, with compounds 6 and 7 presenting the highest activity, with an MIC of 15 µg/mL [ref. 75]. Likewise, these compounds were evaluated against Aspergillus niger, Candida albicans, and Saccharomyces cerevisiae at concentrations of 125–4000 µg/mL and showed MICs ranging from 250 to 1000 µg/mL [ref. 75]. Compounds kaempferol (20), quercetin (24), quercetin 3‐methyl ether (3‐methoxy quercetin) (25), acacetin 8‐C‐[α‐l‐rhamnopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside (34), 2″‐O‐β‐d‐glucopyranosyl‐vitexin (43), 2″‐O‐rhamnopyranosyl‐vitexin (72), and vitexin (88) were assessed against 15 gram‐positive and 4 gram‐negative bacteria, 7 yeast, and 4 dermatophytes at concentrations of 50–500 µg/mL. All these compounds showed activity against at least 3 microorganisms, with compound 24 showing the broadest spectrum, exhibiting activity against 19 of the 30 microorganisms evaluated [ref. 142].
Compounds (E)‐3′‐O‐β‐d‐glucopyranosyl‐4,5,6,4′‐tetrahydroxy‐7,2′‐dimethoxyaurone (33) and kaempferol 3‐O‐β‐d‐(6‐O‐p‐E‐coumaroyl)‐glucopyranoside (tiliroside) or kaempferol‐3‐O‐(6‐p‐coumaroyl)‐glucoside (106) were tested against 19 bacteria but only showed activity against four microorganisms in a concentration range of 0.02–0.5 mg/mL [ref. 2]. Compound 3,5,3′‐trihydroxy‐4′‐methoxy‐6,7‐methylenedioxyflavone (80) was evaluated against 15 microorganisms and showed activity against 7 microorganisms, with the lowest MIC observed against Streptococcus mutans 11.22.1 (20 µg/mL) and the highest in S. aureus ATCC 6538 (1250 µg/mL) [ref. 151]. Compounds kaempferol (20), kaempferol‐3‐O‐(6″‐O‐(E)‐p‐coumaroyl)‐β‐d‐glucopyranoside (107), kaempferol‐3‐O‐(6″‐O‐(Z)‐p‐coumaroyl)‐β‐d‐glucopyranoside (108), and kaempferol 3‐O‐β‐d‐(6″‐feruloylglucopyranoside) (109) were evaluated against Pseudomonas aeruginosa. Among these, compound 20 showed the highest activity with an MIC of 0.008 mg/mL, which was lower than that of ceftriaxone sodium [ref. 144].
The compound vanillic acid (156) was evaluated against five (5) bacteria at a concentration of 0.2 mg/disc and showed activity against two (2) of these microorganisms [ref. 157]. The compound 7‐methoxycoumarin (159), identified in A. caracasana, was evaluated in vitro for antimicrobial activity against Bacillus subtilis, S. aureus ATCC 12398, Staphylococcus epidermidis, Sarcina lutea, and Vibrio cholerae No. 01 ATCC 35971, showing MIC between 0.5 and 0.75 mg/mL, highlighting that this compound demonstrated an MCB of 1 mg/mL against V. cholerae No. 01 ATCC 35971 [ref. 33].
Compounds chrysoeriol‐6‐C‐β‐d‐boivinopyranoside (39), chrysoeriol‐6‐C‐β‐d‐boivinopyranosyl‐4′‐O‐β‐d‐glucopyranoside (40), luteolin‐6‐C‐β‐d‐boivinopyranoside (58), luteolin‐6‐C‐β‐d‐boivinopyranosyl‐3′‐O‐β‐d‐glucopyranoside (59), and luteolin‐6‐C‐β‐d‐boivinopyranosyl‐4′‐O‐β‐d‐glucopyranoside (60) were evaluated for antiviral activity. It was evident that compounds 40, 59, and 60 blocked the secretion of HBsAg, as detailed in subsequent sections [ref. 40]. The compound (80) was evaluated as an antiparasitic against Trypanosoma cruzi and Leishmania amazonensis at concentrations between 4 and 500 µg/mL, but they showed a low reduction in viability [ref. 151].
The results described show that the phenolic compounds exhibit antimicrobial activity at the laboratory level. Most were evaluated against bacteria, followed by yeasts, fungi, viruses, and, to a lesser extent, parasites, highlighting the potential of these compounds as a promising source of antimicrobials.
The isolated phenolic compounds of this subfamily have also been assessed for antioxidant activity. Compounds patuletin 3‐O‐β‐d‐glucopyranoside (67) and kaempferol‐3‐O‐(6″‐O‐(E)‐p‐coumaroyl)‐β‐d‐glucopyranoside (106), as well as a mixture of these two, were evaluated using the DPPH and ORAC assays, both compound 67 and the mixture exhibited relevant activity [ref. 127]. Compounds quercetin (24), quercetin 3‐methyl ether (3‐methoxy quercetin) (25), acacetin 8‐C‐[α‐l‐rhamnopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside (34), 2″‐O‐β‐d‐glucopyranosyl‐vitexin (43), isorhamnetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside (50), 2″‐O‐rhamnopyranosyl‐vitexin (72), and quercetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside (129) were evaluated using chemiluminescent assays, showing activity, with compounds 24 and 25 exhibiting greater activity [ref. 145]. Similarly, compounds 34, 43, 70, and 88 were evaluated using ORAC, demonstrating their antioxidant potential, where compound 88 showed the best activity with 0.96 relative TE [ref. 143]. Although there have been few studies focused on studying the antioxidant potential of the phenolic compounds identified and isolated from this subfamily, the research conducted to date demonstrates that they are a promising source of antioxidant compounds.
Regarding cytotoxic activity against cancer cell lines, the mixture of compounds 5,7‐dihydroxy‐3,6‐dimethoxyflavone (6), oroxilin A (7), 3,5‐dimethoxy‐6,7‐methylenedioxyflavone (10), and 3,5,6,7‐tetramethoxyflavone (17) presented an ED50: 27.5 µg/mL against KB cell [ref. 100]. The compounds alternanthin (35) and alternanthin B (36) were evaluated against HeLa cells at concentrations of 10 and 30 µg/mL, showing inhibition percentages between 8.9% and 55.9% [ref. 36]. Likewise, the mixture of flavonoids (6, 7, 10, and 17) was evaluated in vivo, resulting in an increase in the survival of the mice and a reduction in the size of the tumor [ref. 100].
Furthermore, compounds quercetin (24), quercetin 3‐methyl ether (3‐methoxy quercetin) (25), acacetin 8‐C‐[α‐l‐rhamnopyranosyl‐(1 → 2)‐β‐d‐glucopyranoside (34), 2″‐O‐β‐d‐glucopyranosyl‐vitexin (43), isorhamnetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside (50), 2″‐O‐rhamnopyranosyl‐vitexin (72), and quercetin 3‐O‐α‐l‐rhamnosyl‐(1 → 6)‐β‐d‐glucopyranoside (129) were evaluated as immunomodulators, and it was evidenced that at 50 µmol/L, they do not induce significant release of LDH and are not cytotoxic against PMNL [ref. 145].
For some compounds, the effect on the nervous system has been evaluated. Compounds demethyltorosaflavone D (5), alternanthin (35), alternanthin B (36), chrysoeriol 7‐O‐rhamnoside or chrysoeriol 7‐rhamnoside (41), and torosaflavone E (79) were evaluated as antidepressants and antidementia agents. In the first case, the inhibition of MAO‐A and MAO‐B enzymes was evaluated, whereas in the second, the reduction in β‐amyloid (Aβ) aggregation was assessed. The authors report that the compounds were able to inhibit MAO‐A and MAO‐B, as well as reduce the formation of the Aβ‐aggregation, highlighting that compound 36 presents the greatest inhibition of toxic Aβ plaques [ref. 38, ref. 135]. These results show that the phenolic compounds isolated from this family also have promising activity in the nervous system.
Additionally, the analgesic and anti‐inflammatory activity in vivo was evaluated for compounds 2″‐O‐β‐d‐glucopyranosyl‐vitexin (43) and 2″‐O‐rhamnopyranosyl‐vitexin (72). In both cases, the compounds inhibited hyperalgesia, edema formation, and reduced leukocyte migration [ref. 34, ref. 67]. Likewise, compounds patuletin 3‐O‐β‐d‐glucopyranoside (67) and tiliroside (106) were evaluated for anti‐inflammatory activity in vivo, demonstrating a reduction in the edema formation and leukocyte migration [ref. 127]. These results show that phenolic compounds have potential as analgesic compounds.
Hypoglycemic activity was also evaluated in cleomiscosin A (162) and dictyoceratin C (166), but their activity was low [ref. 84].
Besides the above, it should be noted that some phenolic compounds such as caffeic acid (132) and ferulic acid (137), which have been isolated from members of this subfamily, can become candidates for drugs with dual activity (anti‐inflammatory and antimicrobial activity), which would provide an advantage from the pharmacoeconomic point of view [ref. 71].
A total of 36 out of 173 phenolic compounds identified in this subfamily have been researched in terms of biological activity, showing promising effects such as antimicrobial, antiviral, antioxidant, anticancer, immunomodulatory, antidepressant, antidementia, analgesic, and anti‐inflammatory. Compounds 43 and 72 are particularly noteworthy, as they have demonstrated antimicrobial, immunomodulatory, anti‐inflammatory, and analgesic activity, highlighting their biotechnological potential.
Flavonoids and Their Glycosides
Flavonoids are phenolic compounds commonly found in nature, known for their diverse biological activities. They exhibit antioxidant, analgesic, anti‐inflammatory, anticancer, antipyretic, antiallergic, antidiabetes, antiulcer, antimicrobial, antiprotozoal, antiplatelet, antiatherogenic, antiestrogenic, cardioprotective, neuroprotective, hepatoprotective, and radioprotective activities [ref. 28, ref. 77, ref. 85, ref. 90, ref. 187, ref. 188, ref. 189, ref. 190].
Currently, 133 flavonoids (Table 2) have been reported in members of Gomphrenoideae (Table 2 and Figure 1), consisting of 57 flavone glycosides (34–90), 43 flavonol glycosides (91–133), 15 flavones (3–17), 10 flavonols (18–27), 5 iso‐flavones (28–32), 2 flavan‐3‐ols (1–2), and an aurone (33). It should be noted that quercetin (24), kaempferol (20), and rutin (130) are the most commonly isolated flavonoids from these plants.
Some important aspects of the discovery of flavonoids are described below. In 1992, Pomilio reported the occurrence of an isorhamnetin glycoside (54) for the first time in G. boliviana Moq. [ref. 75]. Later, in 2003, Oliveira first reported the isolation of the symmetrically glycosylated methylene bioflavonoid 8,8‴‐methylene bis(spinacetin 3‐O‐robinobioside) (118) from the ethanol extract of Blutaparon portulacoides (A.St.‐Hil.) Mears leaves. In 2004, Ferreira discovered a new hepta‐substituted (E)‐aurone glucoside (33) in the ethanol extract of Gomphrena agrestis Mart. [ref. 2]. In 2011, Valentová isolated a new isoflavone (32) from the aerial parts of Iresine diffusa f. herbstii (Hook.) Pedersen, characterized by the presence of a methoxy group in position two of the isoflavanone skeleton [ref. 111].
In 2011, Ferreres reported a tetrahydroxymethylenedioxyflavone derivative (77) for the first time in natura [ref. 94]. In 2014, Felipe reported the presence of flavonoids (103, 106, 126) in the inflorescences of Pfaffia glomerata (Spreng.) Pedersen for the first time [ref. 154]. In 2017, Deladino reported the isolation of pentosyl‐vitexin (70) and pentosyl‐isovitexin (69) in A. brasiliana and Alternanthera tenella (Synm. Alternanthera sessilis (L.) R.Br. ex DC. and Alternanthera pungens Kunth) for the first time [ref. 28].
Quercetin (24) has been reported in various species, including Alternanthera bettzickiana (Regel) G. Nicholson, A. brasiliana, Alternanthera maritima (Mart.) A.St.‐Hil. (Synm. Alternanthera littoralis Beauv. ex Moq.), A. paronychioides, Alternanthera philoxeroides (Mart.) Griseb., A. sessilis (L.) R.Br. ex DC., A. tenella Moq. (Synm. A. sessilis (L.) DC. and Alternanthera ficoidea (L.) P. Beauv.), G. agrestis, Gomphrena celosioides Mart., G. claussenii, Gomphrena globosa, and Iresine angustifolia Euphrasén. Quercetin is one of the most abundant flavonoids in the nature and is known for its therapeutic application in allergies, cancer, inflammation, obesity, arthritis, asthma, diabetes, prostate adenocarcinoma, immunity, and infections, as well as its gastroprotective and analgesic properties [ref. 189].
On the other hand, it is noteworthy that G. globosa, with 50 flavonoids (13, 19–20, 24, 27, 46–48, 50–52, 55, 71, 77, 83–87, 92–95, 97–105, 107–110, 112–114, 116, 120–126, 128, 130, 132), is the plant with the highest number of flavonoid‐type compounds reported to date.
Non‐Flavonoid Phenolic Compounds
Currently, only 40 non‐flavonoid phenolic compounds have been reported. These include 14 hydroxycinnamic acids (143–156), 9 benzoic acids (134–142), 2 derivatives of gallic acid (157–158), 2 lignans (160–161), 2 phenylpropanoids (163–164), a coumarin (159), a coumarinolignoid (162), a glycosylated phenylpropionate (165), a sesquiterpene phenol (166), and 7 other phenolic compounds (167–173) (Table 2 and Figure 1).
G. celosioides has the highest number of non‐flavonoid phenolic constituents, with 12 compounds (140, 143–144, 149, 156, 160–162, 166, 168, 170–171). Ferulic acid (149) is the most abundant non‐flavonoid phenolic compound distributed among the species of Gomphrenoideae, having been reported in A. brasiliana, A. paronychioides, A. philoxeroides, A. sessilis, A. tenella, B. portulacoides, G. celosioides Mart, G. globosa, and I. angustifolia.
Terpenoids
Among the terpenes are found monoterpenes, triterpenes, sesquiterpenes, and triterpenoid saponins. These compounds exhibit different biological activities, including antioxidant, antimicrobial, antimalarial, anti‐HIV, anti‐inflammatory, antitumor, antimutagenic, anticancer, antipruritic, antidiabetic, anti‐atherosclerotic, antiallergic, cytotoxic, hemolytic, hypotensive, hepatoprotective, immunomodulatory, and nutraceutical activities, as well as inhibition of cardio‐cerebral vascular diseases, playing an important role in the pharmaceutical industry [ref. 4, ref. 9, ref. 77, ref. 131, ref. 191].
To date, the production of 95 different terpenes in this subfamily has been reported (Table 3 and Figure 1), including 31 triterpenoid saponins (234–264), 31 triterpenes (203–233), 19 sesquiterpenes (179–197), 5 diterpenoids (198–202), 4 monoterpenes (174–177), 3 carotenoids (265–267), a glycoside monoterpene (178), and a drimene (268).
Phytol (202), oleanolic acid (221), pfaffic acid (226), and pfaffosides A–F (255–260) are the most abundant terpenoid compounds distributed among the species of Gomphrenoideae.
In 1984, Nishimoto was the first to isolate the nortriterpene glucuronides called pfaffosides A–C (255–257) from the roots of Pfaffia paniculata (Mart.) Kuntze (Synm. Hebanthe eriantha (Poir.) Pedersen) [ref. 125]. That same year, Nakai reported the isolation of three additional nortriterpenoids, pfaffosides D–F (258–260), from the roots of P. paniculata [ref. 164]. In 2005, Rios identified three new drimenes (268, 195, 196) from the acetone extract of aerial parts of I. diffusa Humb. & Bonpl. ex Willd. [ref. 105]. In 2006, Kuroda reported for the first time the isolation of a taraxane glycoside (245) from a natural source, the roots of G. macrocephala [ref. 99].
In 2008, Chaudhary reported the presence of drimanes in the Amaranthaceae family for the first time, with the isolation of 11,12‐acetonide of 11,12,13‐trihydroxydrimene (179) and 11,12,13‐trihydroxydrimene (194) from the acetone extract of the aerial parts of T. oblongifolia (S. Watson) Standl. (Synm. T. suffruticosa var. oblongifolia (S. Watson) Sánch.Pino & Flores Olv.). Notably, this remains the only phytochemical study conducted on any Tidestromia species [ref. 128]. In 2010, 26 years after Nishimoto’s discovery, Li reported the isolation of two new nortriterpenoids, pfaffine A and B (227–228), from the root of P. paniculata [ref. 123]. In 2018, Han reported the isolation of three new norolean‐type triterpenes (215, 216, 224) from the root of P. glomerata [ref. 115].
With 24 compounds (212–213, 215–216, 221, 223–226, 229–230, 234–235, 237–238, 248, 253–260), P. glomerata has the highest number of terpenoid compounds. It is worth noting that in 2014, Felipe was the first to report the presence of glomeric acid (212), oleanonic acid (223), pfameric acid (229), chikusetsusaponin IV (237), and ginsenoside R0 (248) in this plant [ref. 154].
As mentioned, terpenes exhibit various pharmaceutical activities, which has led to research on compounds isolated from this subfamily. However, their antimicrobial activity (antiparasitic, antibacterial, and antifungal activity) was not extensively studied. To date, only pfaffic acid (226) has been evaluated against T. cruzi, showing an IC50 of 44.78 ± 7.83 µg/mL [ref. 177].
Antiviral activity has been assessed solely for chikusetsusaponin IVa (238) against 11 viruses, demonstrating efficacy against enveloped viruses (Table S1), and this compound was also evaluated in vivo in mice infected with HSV‐2, revealing a reduction in viral titer, symptom alleviation, and increased survival (Table 10) [ref. 41].
TABLE 10: Pharmacological effects of crude extracts and compounds of Gomphrenoideae subfamily.
| Species | Extract (s)/Compounds | Models | Assay method | Dose | Positive control | Effects/Mechanisms | References |
|---|---|---|---|---|---|---|---|
| Antimicrobial activity | |||||||
| Gomphrena globosa | 20 | Male Kunming mice | Murine model of superficial skin infection | 30 mg/kg | Ceftriaxone sodium | ↓ The number of colonies in the woundAntibacterial efficacy was better than positive control | [ref. 136] |
| Activity related to the sexual system | |||||||
| Pfaffia glomerata | HaE of roots | C57BL/6J | Histomorphometric studies | 600 and 1000 mg/kg | — | Does not act as an endocrine disruptor and has no antiandrogenic activity | [ref. 247] |
| Pfaffia glomerata | HaE of roots | Swiss mice (male) | Histological analysis | 100, 200, and 400 mg/kg for 42 days | Sildenafil citrate | 400 mg/kg ↑ the weight of the testes and Leydig somatic index200 mg/kg administered intermittently ↑ the weight of the testes and parenchymaHaE ↑ the proportions of interstitium, Leydig cells, lymphatic vessels, and NO400 mg/kg of HaE causes DNA damage and cell death in Leydig cells, whereas lower concentrations and sildenafil citrate do not show cytotoxicity200 and 400 mg/kg ↓ plasma testosterone and ↑ 17β‐estradiol levelsAt 400 mg/kg, the amount of connective tissue in the penis ↓, whereas type I collagen ↑↓ The amount of type III collagen and the % of smooth muscle | [ref. 113] |
| Volumetric proportions and interstitium analysis | |||||||
| Leydig cell morphometry | |||||||
| Leydig cell viability | |||||||
| Hormone assay | |||||||
| Collagen and smooth muscle quantification | |||||||
| NO assay | |||||||
| Pfaffia glomerata | HaE of roots | SPF ICR mice | Sexual behavior experiment | 150, 750, and 1500 mg/kg for 28 days | Paroxetine | The behavioral study indicated that HaE improved sexual performance in miceHaE helps mitigate the damage caused by paroxetine to the testes and has the potential to enhance sexual functionHaE concentrations ↑ the levels of T, FSH, and E2, restore NO and cGMP content, and significantly reduced PDE5 activity, with results comparable to the control group. HaE effectively reduced PRX‐induced MDA elevationHistologically, testicular lesions were recovered by HaEHaE promoted spermatogenesis | [ref. 168] |
| Organ coefficient | |||||||
| Measurements of hormones and enzymes | |||||||
| Testis histopathological and sperm analysis | |||||||
| Pfaffia glomerata | HaE of roots | Swiss mice (male) | Sexual behavior experiment | 100, 200, and 400 mg/kg for 42 days | Sildenafil citrate | Daily intake of HaE did not affect the BW and caused no morphometric differences in organs such as the uterus, placenta, and ovariesHaE at a concentration of 200 mg/kg ↑ pregnancy rates in females and fertility in malesHaE ↑ ROS levelsMn ↑ at concentrations of 200 and 400 mg/kg of HeEAll concentrations of HaE caused germ cell damage↓ Daily sperm production and elongated spermatids | [ref. 186] |
| Fertility rates | |||||||
| Oxidative stress | |||||||
| Mineral quantification in the testis | |||||||
| Cell viability | |||||||
| Histopathology | |||||||
| Sperm evaluation | |||||||
| Morphometry | |||||||
| Analgesic activity | |||||||
| Alternanthera brasiliana | AqE aerial part | Wistar rats (male) | Acetic acid‐induced abdominal contractions | 25, 50, 100, 200, and 400 mg/kg | Dipyrone | ↓ Of contractions for the different []: 90.35%, 91.73%, 95.17%, 94.45%, and 96.55%All [] evaluated had a higher activity than dipyrone | [ref. 30] |
| Alternanthera maritima | EE of aerial parts | Swiss mice | Carrageenan‐induced paw edema | 30, 100, and 300 mg/kg | — | EE at doses of 300 and 500 mg/kg prevents mechanical hyperalgesiaThe isolated compound (72) at doses of 0.3, 3, and 300 µg/paw prevents a significant ↓ in sensitivity in rats with carrageenan‐induced paw edemaCompound 72 inhibited the hyperalgesic effects of TNF and significantly prevented the ↓ in the threshold sensitivity but did not inhibit the hyperalgesic effects of L‐DOPA | [ref. 34] |
| 72 | Swiss mice | Carrageenan‐induced paw edema | 1, 10, and 20 mg/kg or 3 µg/paw | — | |||
| Swiss mice | TNF‐ or L‐DOPA‐induced hyperalgesia | 3 µg/paw | — | ||||
| Alternanthera philoxeroides | ME of whole plant | Swiss albino mice | Acetic acid‐induced constriction | 50, 100, 200, and 400 mg/kg BW | Aspirin | ↓ The number of constrictions in a dose‐dependent manner (%I 50 mg/kg: 31 and %I 400 g/kg: 44.8) | [ref. 39] |
| Alternanthera sessilis | EE of whole plant | Swiss albino mice | Hot plates test | 250 and 500 mg/kg BW | Morphine | Inhibits the number of contortions by 37.28% (250 mg/kg) and 59.52% (500 mg/kg) the number of contortionsMaximum reaction time 6.87 and 7.28 s at doses of 250 and 500 mg/kgEE ↑ pain threshold | [ref. 139] |
| Swiss albino mice | Writhing test | 250 and 500 mg/kg BW | Diclofenac sodium | ||||
| Alternanthera sessilis | ME of aerial parts | Swiss albino mice (male) | Abdominal writhing test | 50, 100, 200, and 400 mg/kg BW | Aspirin | Dose‐dependent activity. ↓ Writhing for the different []: 27.6%, 37.9%, 41.4%, and 44.8%. The last three doses of ME showed greater activity than the control | [ref. 232] |
| Alternanthera tenella Colla | EE of whole plant | Swiss and C57bl6 | Paw edema, mechanical hyperalgesia, and cold allodynia induced by carrageenan (acetone drop test) | 30, 100, and 300 mg/kg | PRED | Four hours after carrageenan injection, EE and compound 43 inhibited edema by 54% (100 mg/kg) and 56% (10 mg/kg), respectivelyThree hours after carrageenan injection, EE and compound 43 inhibited mechanical hyperalgesia by 99% (300 mg/kg) and 100% (10 mg/kg), respectively | [ref. 67] |
| Swiss | Knee edema and mechanical hyperalgesia induced by zymosan | 100 mg/kg | PRED | Three hours after carrageenan injection, EE inhibited the cold response by 82% at 300 mg/kg, but the isolated compound had no effectA total of 4 and 6 h after zymosan injection, EE and compound 43 blocked mechanical hyperalgesiaFour hours after zymosan injection, EE and compound 43 inhibited edema by 58% and 72%, respectively | |||
| 43 | Swiss and C57bl6 | Paw edema, mechanical hyperalgesia, and cold allodynia induced (acetone drop test) by carrageenan | 0.1, 1, and 10 mg/kg | PRED | |||
| Swiss | Knee edema and mechanical hyperalgesia induced by zymosan | 1 mg/kg | PRED | ||||
| Blutaparon portulacoides | EE of stems | Swiss mice (male) | Paw edema, mechanical hyperalgesia | 30, 100, or 300 mg/kg | Dexamethasone | EE ↓ the sensitivity to mechanical stimuli and reduced carrageenan‐induced mechanical hyperalgesia | [ref. 71] |
| C57BL/6 mice (male) | Mechanical sensitivity and cold sensitivity | 30 and 100 mg/kg | Dexamethasone | In mice treated with CFA, EE at a dose of 30 mg/kg inhibited mechanical sensitivity by 60% (6 days), 90% (16 days), and 77% (22 days) and inhibited cold sensitivity in a manner comparable to dexamethasone | |||
| Gomphrena celosioides | AqE of leaves | Swiss albino mice | Hot plate test | 100, 200, and 400 mg/kg | Morphine | Reaction time: 18.97 ± 0.47, 19.32 ± 1.14, 20.64 ± 0.51. At 400 mg/kg, activity was significant | [ref. 87] |
| Acetic acid‐induced writhing movement test | 100, 200, and 400 mg/kg | Paracetamol | Dose‐dependent activity. Number of writhing movements: 35.78 ± 3.2, 30.16 ± 1.67, 28.27 ± 2.11 | ||||
| Gomphrena celosioides | EE of aerial part | Swiss mice | Paw edema, mechanical hyperalgesia, and cold allodynia induced (acetone drop test) by carrageenan | 300, 700, or 1000 mg/kg | Dexamethasone | All doses ↓ carrageenan‐induced edema formation, with a ↑ inhibition of 61% ± 5%, 53% ± 6%, and 68% ± 5%, respectivelyAll doses ↓ hyperalgesia, exhibited ↑ activity at 300 mg/kg with 91% ± 22%Allodynia ↓ at doses of 700 and 1000 mg/kg, with a ↑ inhibition of 58% ± 14%EE significantly ↓ leukocyte migration (58% ± 14%) but did not reduce protein extravasation into the pleural cavityEE ↓ hyperalgesia and leukocyte migration induced with zymosan, with inhibition rates of 52% ± 3% and 81% ± 4%, respectivelyEE did not significantly alter NO levelsEE inhibits cell adhesion to the endothelium (40% ± 7%) and rolling cells (48% ± 6%)EE ↓ edema (25% ± 18%), and CFA‐induced hyperalgesia | [ref. 78] |
| Swiss mice | Model of carrageenan‐induced pleurisy | 300, 700, or 1000 mg/kg | Dexamethasone | ||||
| Swiss mice | Leukocyte recruitment and mechanical model of zymosan | 300 mg/kg | — | ||||
| Swiss male mice | Zymosan‐induced peritonitis | 300 mg/kg | Dexamethasone | ||||
| Swiss male mice | In situ intravital microscopy analysis | 300 mg/kg | Indomethacin | ||||
| C57BL6 mice (male) | Paw edema and mechanical hyperalgesia induced by CFA | 100 mg/kg | Dexamethasone | ||||
| Pfaffia glomerata | HaE of roots and rhizomes | Swiss albino mice (male) | Writhing test (acetic acid) | 100, 200, and 300 mg/kg | Indomethacin | %I of writhing: 69.1%, 66.4%, and 74.1% for 100, 200, and 300 mg/kg, respectivelyShowed no effect in the hot plate test, indicating no analgesic effect on the central nervous system | [ref. 118] |
| Wistar rats (male) | Hot plate test | 100, 200, and 300 mg/kg | Morphine | ||||
| Pfaffia glomerata | HaE of roots | Swiss mice (female) | Acetic acid‐induced constriction | 10–300 mg/kg | Indomethacin | Inhibits abdominal constriction by 78% ± 3% at 300 mg/kg and has an ID50 of 64.6 mg/kg↓ Glutamate‐induced nociception in a dose‐dependent manner (ID50: 370.8)Antinociceptive activity is not ← by naloxone pre‐treatmentHad no effect against nociceptive responses induced by spinal injections of NMDA, AMPA, and kainite but inhibited (32% ± 8%) nociceptive responses induced by spinal injection of trans‐ACPD↓ The biting response was induced by TNF‐α but did not alter the biting response induced by IL‐1β | [ref. 231] |
| Swiss mice (female) | Glutamate‐induced nociception | 100–600 mg/kg | Morphine | ||||
| Swiss mice (female) injected with acetic acid | Involvement of opioid system | 300 mg/kg | Morphine | ||||
| Swiss mice (female) | Involvement of glutamatergic system was | 300 mg/kg | — | ||||
| Angiogenesis | |||||||
| Alternanthera brasiliana | HaE of leaves | Wistar rats (male‐trichotomy was performed on the dorsum skin and a dermatological punch of 1.5 cm in diameter) | Histomorphometry and Western blotting | 20% HaE of leaves in 2% carbopol gel | — | Favors angiogenesis | [ref. 195] |
| Antiangiogenic activity | |||||||
| Pfaffia paniculata | ME of roots | Adult BALB/c mice (male) with corneal lesion | Histopathological study | 250, 500, or 1000 mg/kg | — | 1000 mg/kg significantly ↓ the number of new blood vessels formed in mouse cornea | [ref. 188] |
| Anti‐arthritic activity | |||||||
| Alternanthera bettzickiana | EE of aerial parts | Wistar rats with CFA‐induced arthritis | Determination of physical parameters | 250, 500, and 1000 mg/kg | Diclofenac sodium | %I of edema: 70.56% and 65.81% for 1000 and 500 mg/kg, respectively1000 and 500 mg/kg reinstated the arthritis index and BWSignificant ↓ in CRP, AST, ALP, ALT, RF, urea, creatinine, and BUN levelsImproved [] of RBCs, Hb, WBCs, and ESR↓ Pannus formation, synovial hyperplasia, inflammatory cell infiltration, bone erosion, bone resorption, joint deformation, soft tissues inflammation, and connective tissue alterations↑ NF‐kB, IL‐4, IL‐10, I‐kB expression↓ COX‐2, IL‐6, TNF‐α, IL‐1β expression, and MDA levelRestores SOD and CAT values | [ref. 19] |
| Arthritic Index | |||||||
| Hematological and biochemical parameters | |||||||
| Histopathological analysis | |||||||
| Radiographic assessment | |||||||
| RT‐PCR | |||||||
| ELISA | |||||||
| Alternanthera bettzickiana | EE of aerial parts | Wistar rats with formaldehyde‐induced arthritis | Hematological and biochemical parameters | 250, 500, 1000 mg/kg | Diclofenac sodium | %I of edema: 72.11%, 65.25%, and 56.62% for 1000, 500, and 250 mg/kg500 and 1000 mg/kg significantly ↓ ALP, ALT, and AST levels↓ Creatinine, urea, CRP, and RF levels500 and 1000 mg/kg: Significant ↓ in TNF‐α and IL‐6500 and 1000 mg/kg: ↑ SOD and CAT, and ↓ MDA and NOEE may be an optimal therapy for the treatment of rheumatoid arthritis | [ref. 20] |
| Enzyme‐linked immunosorbent assay | |||||||
| Oxidative stress biomarkers | |||||||
| Anticancer activity | |||||||
| Alternanthera brasiliana | EaE of leaves | EAC bearing Swiss albino mice | Estimation of hematological parameters | 200 and 400 mg/kg | 5‑FU | ↓ BW; doses of 200 and 400 mg/kg ↓ tumor volume, tumor weight, and viable cell count, whereas ↑ the non‐viable cell countSignificantly ↑ survival rates (%ILS200 mg/kg: 53.33 and %ILS400 mg/kg:78.37)← EAC‐induced changes in Hb, WBCs, and RBCs↓ SGPT, SGOT, TGL, and ALP, restoring them to normal levels↑ GSH, SOD, and CAT levels, while ↓ MDA levelsPrevented the development of steatosis and lymphocyte accumulation in the liver, maintaining an almost normal liver histology | [ref. 190] |
| Biochemical analysis | 200 and 400 mg/kg | 5‑FU | |||||
| Histopathological study | 200 and 400 mg/kg | 5‑FU | |||||
| Tumor growth response analysis | 200 and 400 mg/kg | 5‑FU | |||||
| Determination of %ILS | 200 and 400 mg/kg | 5‑FU | |||||
| Alternanthera sessilis | Paste of leaves | Swiss mice induced with 3,4‐benzo[α]pyrene (male) | BW measurement | 600 mg/g | Uninformed | Had no effect on mice or rats BWDid not significantly inhibit squamous cell carcinoma of the stomach in mice and did not prevent adenocarcinoma development | [ref. 228] |
| Histopathological study | |||||||
| Wistar rats induced with 3′‐methyl‐4‐dimethylaminoazobenzene (male) | BW measurement | 600 mg/g | Uninformed | ||||
| Histopathological study | |||||||
| Alternanthera tenella Colla | AqE of aerial parts | Swiss mice (male) injected with EAC cells | Tumor growth response analysis | 5 or 50 mg/kg | — | ↑ Survival time of mice50 mg/kg inhibited viable tumor cell count by 59%5 mg/kg ↓ BW after 8 days of tumor inoculation | [ref. 66] |
| Determination of %ILS | 5 or 50 mg/kg | — | |||||
| Gomphrena celosioides | Crude powder | DEN/HCB‐induced Albino Wistar rats (male) | Biochemical analysis | 200 mg/kg | — | LPO GOT and GPT levels were significantly ↓, indicating antioxidant activityALP, ACP, and GGT levels significantly ↓ to near normal values, possibly due to the regenerative capacity of liver cellsCrude extract showed remarkable activity on SGOT | [ref. 133] |
| EE of whole plant | DEN/HCB induced Albino Wistar rats (male) | Biochemical analysis | 50 mg/kg | — | |||
| Gomphrena martiana | Mixture of 10, 6, 7, and 17 | S180 bearing BALB/c mice and Swiss mice | BW measurement | 20 and 40 mg/kg | — | The mixture of flavonoids ↑ the survival of S180‐bearing mice by 40%The flavonoid mixture ↓ tumor growth in S180‐bearing mice by 21.6% at 40 mg/kg on Day 18The flavonoid mixture at 20 mg/kg did not ↑ the survival in Ehr Ca‐bearing mice, with only a 10%–20% survival increase at 40 and 60 mg/kgTumor inhibition in Ehr Ca‐bearing mice was 32% at 40 mg/kg on Day 18 | [ref. 100] |
| Determination of the %I of tumor ascites | |||||||
| Mixture of 10, 6, 7, and 17 | Ehr Ca bearing BALB/c mice and Swiss mice | BW measurement | 20, 40, and 60 mg/kg | — | |||
| Determination of the %I of tumor ascites | |||||||
| Pfaffia paniculata | Powdered root | Ehr Ca bearing inbred BALB/cICB mice (male) | Ehrlich ascitic tumor growth | 200 mg/kg | — | Significantly ↓ the Ehrlich ascitic volume but had no significant effect on total tumor cell count | [ref. 224] |
| Pfaffia paniculata | Powdered root | BALB/c mice treated with N‐nitrosodiethylamine | Liver macroscopic analysis | 0.5%, 2%, or 10% by weight | — | Male mice treated with 10% powder showed a small incidence of macroscopic lesions, while mice treated with 2% did not show any lesions. Female mice treated with 0.5% and 2% showed a ↓ in macroscopic lesions↓ Mean lesion number, mean area of the preneoplastic lesions, the % of area with lesions, lesions number by cm2, and total preneoplastic lesions↓ Adenoma incidence in male mice, and adenomas disappeared in female mice | [ref. 226] |
| Morphologic and morphometric analysis | |||||||
| Pfaffia paniculata | EE of rootsAF of roots | Ehr Ca bearing SWISS mice (male) | Survival time | 50, 100, or 200 mg/kg/day | — | BuF 50 and 200 mg/kg ↑ survival, but AF and EE had no effect on survivalBuFF had no effect on total ascitic volume, tumor cell count per mL, or total tumor cells collected | [ref. 248] |
| BuF of roots | Ehr Ca bearing SWISS mice (male) | Survival time | 50, 100, or 200 mg/kg/day | — | |||
| Ehrlich ascitic tumor growth | 50 or 200 mg/kg/day | — | |||||
| Pfaffia paniculata | ME of roots | Ehr Ca bearing BALB/c mice | Macrophage activity | 100, 250, and 500 mg/kg | — | 500 mg/kg ↑ the spreading index of peritoneal macrophages and phagocytosis indexHad no effect on H2O2 and NO production | [ref. 225] |
| Pfaffia paniculata | Powdered root | BALB/c mice (male) treated with N‐nitrosodiethylamine | Histopathological study | 0%, 2%, and 10% of weight | — | 0% and 10% ↑ relative liver weightHistopathological examination showed diffuse mononuclear inflammatory infiltrates and coagulation necrosis0% and 10% ↑ cellular proliferation, but 2% ↓ cellular proliferation and PCNA‐positive nuclei2% and 10% ↑ apoptosisDid not alter intercellular hepatocyte communication via gap junctions | [ref. 227] |
| Immunohistochemical staining | |||||||
| Fluorescence microscopy | |||||||
| Alkaline comet assay | |||||||
| Western blot | |||||||
| Real‐time PCR | |||||||
| Antidiabetic and antihyperglycemic activity | |||||||
| Alternanthera philoxeroides | ME of whole plant | Swiss albino mice | OGTT | 50, 100, 200, and 400 mg/kg BW | GLB | ↓ Serum glucose levels in a dose‐dependent manner (%I 100 mg/kg: 58.6 and %I 400 mg/kg: 65.6)Doses equal to or greater than 100 mg/kg of ME showed greater activity than GLB | [ref. 39] |
| Alternanthera sessilis (red) | FH of EE of aerial parts | Male Sprague Dawley rats induced with STZ | OGTT | 500 mg/kg | GLB | FH and AF did not show significant antihyperglycemic effectFEA showed a more significant hypoglycemic effect than GLBFEA ↓ blood glucose levels in the rats over 15 days but did not affect serum insulin levelsFEA ↓ HOMA index and ↑ QUICKI indexFEA ↓ triglyceride levels (↓ 42.04%) and free fatty acid levels (↓ 34.38%) in plasma but did not alter the triglyceride contentFEA ↑ insulin levels and SOD activity in the pancreas | [ref. 236] |
| AF of EE of aerial parts | OGTT | 500 mg/kg | GLB | ||||
| FEA of EE of aerial parts | Male Sprague Dawley rats induced with STZ | OGTT | 500 mg/kg | GLB | |||
| Biochemical assay | 250 mg/kg | Pioglitazone | |||||
| Liver triglyceride assay | 250 mg/kg | Pioglitazone | |||||
| Pancreatic insulin and SOD assay | 250 mg/kg | Pioglitazone | |||||
| Insulin sensitivity indexes | 250 mg/kg | Pioglitazone | |||||
| Alternanthera sessilis | Green leaf juice | Wistar rats (male) | OGTT | NA | — | Did not reduce starch‐ or glucose‐induced postprandial glycemic load | [ref. 211] |
| OSTT | NA | — | |||||
| Alternanthera sessilis | ME of aerial parts | Swiss albino mice (male) | OGTT | 50, 100, 200, and 400 mg/kg BW | GLB | Dose‐dependent activity. ↓ blood glucose levels by 22.9%, 30.7%, 45.4%, and 46.1%. The highest [] showed activity comparable to that of GLB | [ref. 232] |
| Antihypertensive effect | |||||||
| Alternanthera sessilis | EE of whole plant | Sprague–Dawley albino rats | Measures of SDB, DBP, MABP | 1–10 mg/kg | Verapamil | ↓ SBP, DBP, and MABPDose‐dependent hypotensive activity | [ref. 64] |
| Gomphrena celosioides | EE of aerial parts | Wistar rats (male)—the 1K1C method | Acute model of direct blood pressure measurement | 30, 100, or 300 mg/kg diluted | — | EE at [] of 100 and 300 mg/kg ↓ MAP in a dose‐dependent manner, reducing PAM by 36.7 and 38.2 mm Hg, respectivelyEE acted as a diureticEE ↑ the Na levels in the urine, while K and Cl levels in urine were similar to the control, and serum remained unchanged in all groupsEE ↓ MAP in the 2K1C model, but the effect was lower than of enalaprilEE inhibited ACE even more than enalapril, as well as ↓ serum aldosterone [] and TBARS, and ↑ serum nitriteThe isolated mesenteric beds in the EE group showed ↓ contractility and reduced pressure ↑ after Phe administration, as well as ↑ relaxation after treatment with ACh and SNPThe left ventricle was thinner in EE‐treated rats than in control, and EE did not affect heart, liver, or kidney weights | [ref. 80] |
| Wistar rats (male)—the 2K1C method | Diuretic assessment | 100 mg/kg | Enalapril | ||||
| Blood pressure assessment | |||||||
| Urine and serum analysis | |||||||
| ACE activity, aldosterone, nitrite, and TBARS | |||||||
| Isolation of the mesenteric bed and assessment of vascular reactivity to Phe, ACh, and SNP | |||||||
| Organ weighing and histopathology | |||||||
| Anti‐inflammatory activity | |||||||
| Alternanthera brasiliana | Infusion of aerial part | Wistar rats (male) | Carrageenan‐induced pleurisy | 200 and 400 mg/kg doses | Indomethacin | 400 mg/kg significantly ↓ the number of lymphocytes↓ Exudate by 19.8% (200 mg/kg) and 23.9% (400 mg/kg)↓ Polymorphonuclear cells (47.5% and 48.02%) and ↑ mononuclear cells (72.13% and 55.74%) | [ref. 30] |
| Alternanthera brasiliana | EE of leaves | Mus musculus mice | Formalin test | 25, 50, and 100 mg/kg | Indomethacin | EE at concentrations of 25, 50, and 100 mg/kg, ↓ the edematogenic process by 35.57%, 64.67%, and 64.17%, respectively | [ref. 26] |
| Alternanthera brasiliana | HaE of leaves | Wistar rats (male‐trichotomy was performed on the dorsum skin and a dermatological punch of 1.5 cm in diameter) | Histomorphometry | 20% HaE of leaves in 2% carbopol gel | — | 20% HaE leaf extract in 2% carbopol gel controls the recruitment of inflammatory cells at the wound site, according to histomorphometry and biochemical analysis20% HaE leaf extract in 2% carbopol gel modulates inflammation by ↑ IL‐1β and ↓ TGF‐β1 levelsHaE at 20% exhibited anti‐inflammatory activity in the acute phase of inflammation | [ref. 195] |
| Biochemical analysis (MPO and NAG) | 20% HaE of leaves in 2% carbopol gel | ||||||
| Western blotting | 20% HaE of leaves in 2% carbopol gel | ||||||
| Alternanthera maritima | EE of aerial parts | Swiss mice | Carrageenan‐induced paw edema | 30, 100, and 300 mg/Kg | Dexamethasone | EE inhibits edema formation at 100 and 300 mg/kg (79%), whereas the isolated compound inhibits it at all [] tested, achieving the ↑ inhibition at 1 mg/kg (76%)EE ↓ leukocyte counts at 100 and 300 mg/kg, with %I of 65% and 68%, respectivelyThe isolated compound ↓ leukocyte migration and protein extravasation at all doses, with the ↑ inhibition of leukocyte migration at a dose of 10 mg/kg (77%) and protein extravasation at 20 mg/kg (56%) | [ref. 34] |
| 72 | Swiss mice | Carrageenan‐induced paw edema | 1, 10, and 20 mg/Kg | Dexamethasone | |||
| EE of aerial parts | Swiss mice | Carrageenan‐induced pleurisy | 30, 100, and 300 mg/Kg | Dexamethasone | |||
| 72 | Swiss mice | Carrageenan‐Induced Pleurisy | 1, 10, and 20 mg/Kg | Dexamethasone | |||
| Alternanthera tenella Colla | AqE of whole plant MHW | BALB/c mice (male) induced with carrageenan | Paw thickness measurement | 200 or 400 mg/kg | Indomethacin | The extracts inhibit edema formation in a dose‐dependent mannerAqE MCW and MHW showed greater inhibition of edema than indomethacin, with a %I 400 mg/kg of 61% and 56% at 3 h, respectively | [ref. 234] |
| AqE of whole plant MCW | Paw thickness measurement | 200 or 400 mg/kg | Indomethacin | ||||
| Alternanthera tenella Colla | EE of whole plant | Swiss mice | Zymosan‐induced articular inflammation | 100 mg/kg | PRED | Four hours after carrageenan injection, EE and 43 inhibited edema by 54% at 100 and 1 mg/kg, respectivelyFour hours after zymosan injection, EE and 43 inhibited edema by 58% and 72%, respectivelyEE and 43 ↓ the total leukocyte count in synovial fluid by 65% and 61%, respectivelyEE and 43 blocked leukocyte migration and inhibited edema proteins by 54% and 72%, respectivelyTwenty‐four hours after CFA‐injection, EE and 43 inhibited MPO activity by 82.86% and 79.15%, respectively, and also inhibited NAG by 67.87% and 68.56% | [ref. 67] |
| Carrageenan‐Induced Pleurisy | 100 mg/kg | PRED | |||||
| C57bl6 mice | MPO and NAG activity assay | 100 mg/kg | |||||
| 43 | Swiss mice | Zymosan‐induced articular inflammation | 1 mg/kg | PRED | |||
| Carrageenan‐induced pleurisy | 1 mg/kg | PRED | |||||
| C57bl6 mice | MPO and NAG activity assay | 100 mg/kg | |||||
| Blutaparon portulacoides | EE of aerial parts | Swiss mice (male) | Venom of Bothrops jararacussu induced paw edema | 100, 250, or 500 mg/kg | Dexamethasone | EE at 250 and 500 mg/kg inhibits venom‐induced edema formation by 28.5% and 39% within 6 h, respectivelyEE also ↓ the edematogenic effect induced by BthTX‐I and BthTX‐II from 30 min to 6 h (end of experiment)EE had no effect on leukocyte migration induced by venom or BthTX‐II, but it significantly inhibited the leukocyte flux induced by BthTX‐I | [ref. 235] |
| BthTX‐I‐ and BthTX‐II‐induced paw edema | 500 mg/kg | Dexamethasone | |||||
| Venom of Bothrops jararacussu induced pleurisy | 500 mg/kg | Dexamethasone | |||||
| BthTX‐I‐ and BthTX‐II‐induced pleurisy | 500 mg/kg | Dexamethasone | |||||
| Blutaparon portulacoides | EE of stems | Swiss mice (female) | Carrageenan‐induced pleurisy | 30, 100, 300, and 1000 mg/kg | Dexamethasone | Only at 1000 mg/kg was a 55% inhibition of leukocyte invasion in the pleura, and protein exudation was ↓ at doses of 300 and 100 mg/kg, as well as a ↓ in IL‐1β levelsEE ↓ edema in a dose‐ and time‐dependent manner, achieving a 67% reduction at 4 h in mice injected with carrageenan, and EE ↓ edema in mice treated with CFA in a manner comparable to that of dexamethasone, reaching an 80% inhibition at Day 22 | [ref. 71] |
| Swiss mice (male) | Carrageenan‐induced paw edema | 30, 100, or 300 mg/kg | Dexamethasone | ||||
| C57BL/6 mice (male) | CFA | 30 and 100 mg/kg | Dexamethasone | ||||
| C57BL/6 mice (female) | BCG‐induced pleurisy | 30 and 100 mg/kg | Isoniazid | ||||
| Gomphrena celosioides | AqE of leaves | Adult Sprague–Dawley rats and Swiss albino mice | Carrageenan‐induced edema | 100, 200, and 400 mg/kg | Indomethacin | Dose‐dependent activity%I of edema: 27.97, 39.62, and 39.62At 100 mg/kg, the effect was not significant | [ref. 87] |
| Gomphrena celosioides | EE of whole plant | Wistar albino rats | Carrageenan induced paw edema | 200 mg/kg of BW | Diclofenac | ↓ CRP levels, possibly due to the inhibition of inflammatory mediatorsInhibition of edema | [ref. 77] |
| Pfaffia glomerata | HaE of roots and rhizomes | Wistar rats (male) | Carrageenan induced paw edema | 100, 200, and 300 mg/kg | Dexamethasone | %I of edema: 46.3, 56.8, and 63.2 for 100, 200, and 300 mg/kg, respectivelyHaE does not inhibit cell migration↓ Granulomatous tissue formation by 29% | [ref. 118] |
| Granulomatous tissue assay | 100 mg/kg | Dexamethasone | |||||
| Pfaffia glomerata | HaE of roots | Swiss mice (male) | Carrageenan induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ↓ Carrageenan‐induced paw edema in a dose‐dependent manner (ID50: 60.5 (dose oral) and 20.4 (dose intraperitoneal))↓ Edema induced by bradykinin and substance P, highlighting that 300 mg/kg of HaE completely ↓ edema at 120 min. These results suggest that HaE has antinociceptive activity↓ Edema induced by histamine, serotonin, and LPS↑ NO levelsWhen the ↑ in NO levels is blocked with L‐NAME, a ↓ in anti‐edema activity is observed | [ref. 119] |
| Swiss mice (male) with carrageenan‐induced edema | Evaluation of the influence of NO synthase and guanylate cyclase inhibition | 300 mg/kg | Indomethacin | ||||
| Swiss mice (male) | Bradykinin induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ||||
| Swiss mice (male) | Substance P induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ||||
| Swiss mice (male) | Serotonin induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ||||
| Swiss mice (male) | Histamine induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ||||
| Swiss mice (male) | LPS induced paw edema | 1, 10, 30, 100, or 300 mg/kg | Indomethacin | ||||
| Pfaffia paniculata | ME of roots | Wistar rats (male) | TNBS induced intestinal inflammation | 25, 50, 100, 200, or 400 mg/kg | Prednisolone | No ↓ macroscopic damage score in the preventive protocol, but at 200 mg/kg in the curative protocol, ↓ in gross damage score was observedDoses of 50 and 100 mg/kg ↓ microscopic damage score (↑ mucosal regeneration)↓ Polymorphonuclear cell infiltration, fewer ulceration sites, ↓ dilated crypts, and depletion of goblet cell200 mg/kg, ↓ MPO activity was observed, and doses of 50 or 200 mg/kg prevented glutathione depletion in the curative protocol↓ IL‐1β, IFN‐γ, and C‐reactive protein levelsAt 200 mg/kg, ↓TNF‐α and IL‐6 levelsIt was not able to prevent TNBS‐induced intestinal inflammation but was able to ↓ colonic inflammation | [ref. 215] |
| Pfaffia paniculata | ME of roots | Male Wistar rats (male) | Intestinal inflammation assessment | 25, 50, 100, and 200 mg/kg | — | At 200 mg/kg ↓ gross damage score, extent of injury, and MPO activity were observedAt 25 mg/kg, ↓ Hsp70; 50 mg/kg, ↓ Mapk3 and ↑ Muc4; 100 mg/kg, ↑ Mapk1, Muc3, Muc4, and ↓ Mapk3; at 200 mg/kg, ↓ Mapk3No effect was observed on the mRNA levels of heparanase, NF‐κB, Mapk6, Mapk9, Muc1, or Muc2Inflammatory activity was related to the differential modulation of MAPKs and the expression and production of mucin | [ref. 120] |
| Inflammatory mediator’s analysis | 25, 50, 100, and 200 mg/kg | — | |||||
| Pfaffia townsendii | EE of whole plant | Swiss mice (male) | Carrageenan‐induced paw edema | 300 mg/kg | Dexamethasone | EE inhibits the formation of edema by 51.00% ± 11.0%, while compound 67 inhibited it by 75.4% ± 4.0% and compound 98 by 73.00% ± 4.0%EE inhibited leukocyte migration to the pleura by 69.2% ± 1.04%, while compounds 67 and 106 by 50.7% ± 1.03% and 59.4% ± 1.25%, respectivelyEE ↑ plasma leakage, whereas flavonoids ↓ plasma leakageThe anti‐inflammatory activity of compound 67 was similar to that of dexamethasone | [ref. 127] |
| 67 | 1 mg/kg | ||||||
| 106 | 1 mg/kg | ||||||
| EE of whole plant | Swiss mice (female) | Carrageenan‐induced pleurisy | 300 mg/kg | Dexamethasone | |||
| 67 | 1 mg/kg | ||||||
| 106 | 1 mg/kg | ||||||
| Antimutagenic activity | |||||||
| Pfaffia glomerata | Commercial root dry extract | Wistar rats (Rattus norvegicus) treated with cyclophosphamide | Chromosomal aberration test | 1.5 mg/mL simultaneous or pretreatment or post‐treatment | — | Significantly ↓ the % of damage induced by cyclophosphamide; damage was ↓ by 87% for simultaneous treatment, 98% for pretreatment, and 99% for post‐treatment | [ref. 112] |
| Antispasmodic activity | |||||||
| Gomphrena perennis | HaE of aerial parts | Sprague–Dawley rats (female) | Carbachol concentration‐response (CCh‐CRCs) | 0.14–28.1 mg extract/mL | — | HaE demonstrated antispasmodic activity through several mechanisms, highlighting the non‐competitive inhibition of Ca2+ influx. Significant values were obtained for the inhibition of CCh‐CRCs in a concentration‐dependent manner | [ref. 140] |
| Calcium concentration‐response (Ca2+‐CRCs) | |||||||
| Relaxation response concentration (RRC) | |||||||
| Antioxidant activity | |||||||
| Alternanthera brasiliana | HaE of leaves | Wistar rats (male‐trichotomy was performed on the dorsum skin and a dermatological punch of 1.5 cm in diameter) | Biochemical analysis: TBARS and antioxidants—SH groups | 20% HaE of leaves in 2% carbopol gel | — | Relevant antioxidant activity was observed on Day 2, probably to control oxidative damage | [ref. 195] |
| Gomphrena celosioides | EE of whole plant | Wistar albino rats | FRAP and TBARS assays | 200 mg/kg of BW | Vitamin C | Significantly ↓ serum TBAR levels↑ Serum levels total antioxidant capacitySignificant ↓ in Fe3+ to ion Fe2+ ion activity | [ref. 77] |
| Pfaffia glomerata | FD of roots | C57BL/6 mice | Tissue oxidative induction | 250, 25, and 2.5 µg/mL | Quercetin | FDs showed inhibitory activity against lipid peroxidation and the formation of ROS. In particular, only the FD of aerial parts, at the highest concentration, showed significant total antioxidant capacity | [ref. 167] |
| FD of aerial part | Total antioxidant capacity | ||||||
| Lipid peroxidation | |||||||
| Sulfhydryl groups content | |||||||
| Reactive oxygen species (ROS) | |||||||
| Antiviral activity | |||||||
| Alternanthera philoxeroides | 238 | BALB/c mice inoculated HSV‐2 strain UW 264 | Determination of clinical signs and viral shedding | 0.3 or 0.6 mg | — | Dose‐dependent protection against HSV‐2In the 3rd day of treatment, it ↓ the mean titers of virus shed by 33.33%Significantly suppressed herpetic lesions↑ Survival by 40 and 60% for doses of 0.3 and 0.6 mg, respectively | [ref. 41] |
| Cardioprotective activity | |||||||
| Gomphrena celosioides | AqE of stems, flowers, and leaves | Wistar albino rats induced with DOX | Biochemical study | 200 and 500 mg/kg for 14 days | Resveratrol | AqE and EE ↓ the [] of ALT, AST, CK‐MB, cholesterol, and triglycerides in serumAqE and EE ↑ the [] of HDL‐CIt ↓ the weight loss of the rat but did not affect the relative weight of the heart | [ref. 76] |
| RW of the body and heart | |||||||
| EE of stems, flowers, and leaves. | Wistar albino rats induced with DOX | Biochemical study | 200 and 500 mg/kg for 14 days | Resveratrol | |||
| RW of the body and heart | |||||||
| Gomphrena perennis | HaE of aerial parts | Sprague–Dawley rats (female) | Langendorff method with control Krebs solution (Krebs‐C), Ischemia/reperfusion (I/R) model | Oral 25 mg HaE/kg/day and HaE at 0.1% v/v were perfused for 15 min prior to I/R | Krebs‐C | HaE has a cardioprotective effect due to its action on NO production, attributed to the presence of flavones in its composition | [ref. 140] |
| Blutaparon portulacoides | AqE of whole plant | SHRs and Wistar‐Kyoto rats (male) | Electrocardiography | 30, 100, and 300 mg/kg | HCTZ | AqE prevents changes in the RW of the heart and left ventricle, as well as changes in the levels of MDA and NTAqE has significant diuretic and cardioprotective effects | [ref. 155] |
| Blood pressure | |||||||
| Biochemical analysis | |||||||
| RW of organs, histopathology, and heart morphometry | |||||||
| Diuretic activity | |||||||
| Gomphrena celosioides | EE of aerial parts | Wistar rats (male) | Single‐dose model of diuretic assessment | 30, 100, and 300 mg/kg | HCTZ | EE ↑ urine volume, comparable to HCTZ, DI100 mg/kg: 1.74 ± 0.25 and DI300 mg/kg: 1.86 ± 0.28EE had a higher UNa value than HCTZEE had no effect on urine K+, Ca, and Cl− [], nor on pH, density, serum electrolytes, urea, and creatinineAfter pretreatment with L‐NAME, indomethacin, or HOE‐140, EE did not significantly promote diuresis or natriuresisDuring the 7 days of treatment, an ↑ in UV and UNa and ↓ aldosterone levels were observed | [ref. 81] |
| Assessment of the involvement of the prostaglandin, bradykinin, and NO pathways | 100 mg/kg | HCTZ | |||||
| Urine and serum analysis Urine | 100 mg/kg | HCTZ | |||||
| Gomphrena perennis | HaE of aerial parts | Sprague–Dawley rats (female) | Quantification of Na+ and K+ content urinary volumetric excretion (UVE %) | 100 and 400 mg/kg | Amiloride | HaE did not show an ↑ in the total urine volume at the evaluated doses and did not cause a differences in ion excretion in the animal’s urine | [ref. 140] |
| Blutaparon portulacoides | AqE of whole plant | SHRs and Wistar‐Kyoto rats (male and SHRs) | Biochemical analysis | 30, 100, and 300 mg/kg | HCTZ | 300 mg/kg of AqE prevents changes in renal sodium and chloride excretion, maintaining urinary volume and electrolyte elimination. AqE does not increase the potassium elimination, preventing the appearance of cramps and arrhythmia300 mg/kg of AqE has diuretic effects in SHRs | [ref. 155] |
| Gastrointestinal activity | |||||||
| Alternanthera repens | ME | CD1 strain mice (male) | Evaluation of the number of feces | 50 and 100 mg/kg | Diphenoxylate | AqE (%I 100 mg kg: 49 ± 6.9) and ME (%I 50 mg kg: 37.9 ± 0.7) significantly ↓ castor oil‐induced diarrheaHeE and ClE did not show antidiarrheal activityME modifies normal defecation (%I 100 mg/kg: 59.6 ± 3.2)ME has dose‐dependent antidiarrheal activity at 12.5, 25, and 50 mg/kg, with highest effect at 50 mg/kg in mice with MgSO4‐induced diarrhea (%I:75.9)ME ↓ intestinal transit by 24% at 60 min, but at 90 min, intestinal transit is returned to 100% | [ref. 197] |
| AqE or ClE or HeE or ME | CD1 strain mice (male) treated with castor oil or MgSO4 | Evaluation of the antidiarrheal activity | 50 and 100 mg/kg | Diphenoxylate | |||
| ME | CD1 strain mice (male) treated with castor oil | Evaluation of the antidiarrheal activity | 12.5, 25, 50, and 100 mg/kg | Diphenoxylate | |||
| ME | Wistar rats (male) mice treated with castor oil | Effect on small intestinal transit | 12.5, 25, 50, and 100 mg/kg | Diphenoxylate | |||
| Alternanthera repens | AqE of aerial parts | Swiss mice (female) | Charcoal meal method | 1–300 mg/kg | Atropine | The extracts ↓ gastrointestinal content and contain metabolites with antidiarrheal activity | [ref. 46] |
| EE of aerial parts | Swiss mice (female) | Charcoal meal method | 1–300 mg/kg | Atropine | |||
| Gastroprotective activity | |||||||
| Gomphrena celosioides | ME of leaves | Wistar rats (male) induced with indomethacin | Determination of gastric volumes, pH, acid outputs, ulcer score, and ulcer index Biochemical analysis | 200, 500, and 800 mg/kg BW | Cimetidine | ↑ pH in a dose‐dependent manner, and at 800 mg/kg this ↑ was more significant than that of cimetidine↓ Acidity, gastric volume, ulcer index, ulcer score, and pepsin activity in a dose‐dependent manner, with these effects being more marked at 800 mg/kg of ME than cimetidine↓ MDA levels and ↑ protein levelsME displays an antiulcerogenic effect related to its gastroprotective activity | [ref. 88] |
| Gomphrena celosioides | ME of leaves | Wistar rats (male) induced with acidified ethanol | Biochemical analysis | 200, 400, and 800 mg/kg BW | Cimetidine | ME ↓ the increase in gastric volume and acid output, and ↑ the mucus content↑ SOD, GSH, and GPX activities↓ LPO and XO levelsME promotes the restoration of the epithelial layer, lamina propria, and submucosal layer in a dose‐dependent manner | [ref. 246] |
| Histopathological study | |||||||
| Guilleminea densa | HaE of leaves | Holtzman rats induced with ethanol (male) | Macroscopic analysis | 200, 400, and 600 mg/kg | Ranitidine and sucralfate | Dose‐dependent gastroprotective effect (%I of gastric lesions400 mg/kg: 58,78 and (%I of gastric lesions600 mg/kg: 82,72)Significantly ← mucus depletion induced by ethanol at HaE [] of 400 and 600 mg/kg but had no effect on NP‐SG levelsAt 600 mg/kg, it inhibited gastric erosions, ulcers, acute inflammation infiltration, and focal bleeding development | [ref. 102] |
| Determination of gastric mucus and NP‐SG | |||||||
| Histopathological study | |||||||
| Pfaffia glomerata | AqE of roots and rhizomes | Wistar rats (female) with ulcers induced by restriction or ethanol or indomethacin | Determination of IMD | 125, 250, 500, and 1000 mg/kg | Ranitidine | ↓ Ethanol‐induced gastric lesion in a dose‐dependent manner, reducing them by over 90%At 250 and 1000 mg/kg, ↓ stress‐induced gastric lesion by 37.8% and 47.8%, respectivelyThe AqE did not show gastric mucosa protective activity against indomethacin‐induced ulcers↑ pHThe ↓ in total acidity and gastric volume in rats with a pylorus ligation is associated with an ↑ in NOx contentAqE has no effect on total acidity and gastric secretion volume in rats treated with bethanechol or pentagastrinAt 2000 mg/kg, ↓ total acidity by 18.4% and gastric secretion volume by 53.2% in histamine‐injected rats | [ref. 164] |
| Wistar rats (female) with pylorus ligation | Determination of gastric acid secretion | 125, 250, 500, and 1000 mg/kg | — | ||||
| Wistar rats (female) with pylorus ligation and administration of bethanechol, histamine or pentagastrin | Determination of gastric acid secretion | 125, 250, 500, and 1000 mg/kg | — | ||||
| Wistar rats (female) with pylorus ligation | Determination of nitric oxide production | 1500 mg/kg | — | ||||
| Hepatic activity | |||||||
| Pfaffia glomerata | HaE of roots | Swiss mice (male) | Biochemical analysis | 100, 200, and 400 mg/kg | — | The HaE showed effects such as mineral content changes, antioxidant enzymes, and morphological modificationsIn general, HaE generated oxidative stress severe enough to induce liver damage | [ref. 185] |
| Liver oxidative stress markers | |||||||
| Morphology | |||||||
| Histology and histopathology | |||||||
| Mineral and hepatic glycogen content | |||||||
| Hepatoprotective activity | |||||||
| Alternanthera brasiliana L. | HaE of leaves | BALB/c mice (male) induced with CCl4 | Body weight, liver weight, and liver morphology | 200 and 400 mg/kg | — | HaE restored the BW↓ Liver index compared to CCl4 aloneRestore ALT, AST, and ALP levelsRestored liver size to normal, decreased the echogenic pattern, recovered nodular edge appearance, and ↓ the CBD dilation400 mg/kg of HaE improvement hepatic architecture, showing minimal mononuclear cell infiltration and a ↓ number of mitotic figuresHaE significantly ↓ MDA levelsRestore GSH, GST, SOD, and vitamin C levels↓ TNF‐α, IL‐1β, and IL‐18 levels400 mg/kg of HaE significantly ↓ phospho–NF–kB (p65) and NLRP3 protein levels400 mg/kg of HaE ↓ MMP‐2 and MMP‐9 levels and restores TIMP‐1↓ TGF‐β, α‐SMA, and p‐Smad2/3 protein levelsHaE leaf extracts may serve as an herbal hepatoprotective agent | [ref. 208] |
| Biochemical analysis (AST, ALT, and ALP) | — | ||||||
| Lipid peroxidation and antioxidant parameters | — | ||||||
| Histopathological study | — | ||||||
| Western blot analysis | — | ||||||
| qRT‐PCR analysis | — | ||||||
| ELISA | — | ||||||
| Alternanthera sessilis | ME of whole plant | Wistar rats (male) induced with CCl4 | Biochemical analysis (TBARS, GSH, CAT) | 50, 200, and 250 mg/kg | Silymarin | At 250 mg/kg, ← the ↑ in serum SGPT, SGOT, and ALP levelsGenerates a ↓ in serum cholesterol and bilirubin levelsIt ↓ lipid oxidation, ↑ GSH levels, and improves CAT levelsAt higher doses, ← body degeneration (↓ necrosis and restores cellular integrity) | [ref. 51] |
| Histopathological study | |||||||
| Gomphrena celosioides | AqE of stems and leaves | Wistar rats induced with CCl4 (PT and TC) | Biochemical analysis (AST, ALT, ALP, BT, and CB) | 500 mg/kg BW for 5 days | Silymarin | AqE ↓ AST, ALT, ALP, BT, and CB values, indicating a preventive (PT) and restorative (TC) effectWith PT and CT, liver lesions are ↓ severe, with PT showing greater hepatoprotective activity | [ref. 90] |
| Histopathological study | |||||||
| Gomphrena globosa L. | AqE | C57BL/6 mice (male) with CCl4 | Biochemical analysis (AST, ALT, ROS, and SOD) | 100, 200, or 300 mg/kg | Bifendatatum | AqE ↓ serum AST, ALT levels, and hepatic MPOAqE improves hepatic total protein contentIt ↓ ROS and MDA levels and ↑ GSH, GSH‐Px, and SOD activitiesAqE improves liver injury in a dose‐dependent mannerIt activates Nrf2 protein expression and regulates Keap1 levelsIt activates GCLC, GCLM, HO‐1, and NQO1 protein expressionIt ↓ PI3K and mTOR phosphorylation, inhibits P62 protein expression, and activate LC3 II protein expressionIt promotes autophagyAqE alleviates CCl4‐induced chronic liver injury in mice by activating antioxidant signaling pathways and promoting autophagy | [ref. 249] |
| Measurement of MPO, TP, MDA, GSH, and GSH‑Px | |||||||
| Histopathological studies | |||||||
| Western blot | |||||||
| Immunomodulatory activity | |||||||
| Alternanthera tenella Colla | AqE of aerial parts | Swiss mice (male) immunized with sheep RBC | Antibody assays | 5 and 50 mg/kg | — | At 5 mg/kg, it ↓ anti‐SRBC IgM secreting cells prior to immunization, but 50 mg/kg has no effect on PFC count50 mg/kg improves the production of IgM and IgG2a antibodies in mice stimulated with LPSIt had no effect on the total number of nucleated spleen cells or spleen weight | [ref. 66] |
| LPS stimulated mice (male) | Antibody assays | 5 and 50 mg/kg | — | ||||
| Alternanthera tenella Colla | AqE of whole plant MHW | BALB/c mice (male) immunized with sheep RBC | Measurement of BW and lymphoid organs | 50, 100, or 200 mg/kg BW | BALB/c mice (male) only immunized with sheep RBC | The extracts at 50, 100, and 200 mg/kg did not show significant differences in BW and lymphoid organ weightAqE MCW ↑ liver weight by 19.5%The extracts did not affect spleen cellularityAqE MHW and AqE MCW maintained cell viability at 88% and 90%, respectivelyOnly AqE MCW at 100 mg/kg significantly ↑ PFC, suggesting the presence of immunomodulatorsAqE MCW ↑ anti‐SRBC IgM and IgG titers, but AqE MHW did not show any significant effect on antibody titers | [ref. 234] |
| Splenic cellularity | |||||||
| PFC assay | |||||||
| Antibody assays | |||||||
| AqE of whole plant MCW | BALB/c mice (male) immunized with sheep RBC | Measurement of BW and lymphoid organs | 50, 100, or 200 mg/kg BW | BALB/c mice (male) only immunized with sheep RBC | |||
| Splenic cellularity | |||||||
| PFC assay | |||||||
| Antibody assays | |||||||
| Neuropharmacological activity | |||||||
| Alternanthera brasiliana | Infusion of aerial part | Wistar rats (male) | Open field test | 100, 200, and 400 mg/kg | — | There was no effect on latency time in the first rectangle or the number of crossings100 mg/kg ↑ the number of rearings, while 200 mg/kg ↓ the number of fecal boluses | [ref. 30] |
| Elevated plus maze | 100, 200, and 400 mg/kg | — | |||||
| Alternanthera brasiliana | ME of leaves | Swiss albino mice (male) | Hole board test | 100, 300, and 600 mg/kg | Diazepam | ME at 300 and 600 mg/kg significantly ↑ the number of times and duration of the mice that poke their headsIt ↑ rearing, assisted rearing, and the number of squares traveled, showing comparable or superior activity to diazepamIt ↑ in the number of entries into the open arm but ↓ entries into the close armIt ↑ time spent in lighted box, crossing numbers, and transfer latency, while ↓ time spent in the dark boxIt ↓ locomotor activity in a dose‐dependent manner (CNS depressant effect)ME has anxiolytic activityIt protected mice from PTZ‐induced seizures in a dose‐dependent manner, achieving maximum protection of 66.66%ME at 600 mg/kg ↓ the latency of maximal electroshock‐induced seizures, but had no effect on seizure incidence | [ref. 24] |
| Open field test | 100, 300, and 600 mg/kg | Diazepam | |||||
| Elevated plus maze test | 100, 300, and 600 mg/kg | Diazepam | |||||
| Light/dark exploration test | 100, 300, and 600 mg/kg | Diazepam | |||||
| Locomotor Activity test | 100, 300, and 600 mg/kg | Diazepam | |||||
| PTZ injected Swiss albino mice (male) | Chemoshock convulsion | 100, 300, and 600 mg/kg | Diazepam | ||||
| Swiss albino mice (male) | Maximal electroshock induced convulsion | 100, 300, and 600 mg/kg | Diazepam | ||||
| Alternanthera philoxeroides | EE of whole plants | ICR‐OVX mice (female) | FST | 250 and 500 mg/kg | 17β‐Estradiol | It ↓ immobility time in FST and TSTIt did not alter locomotor activity in OVX miceIt ↓ serum corticosterone levels in a dose‐dependent mannerIt normalized the expression of CREB and BDNF | [ref. 38] |
| TST | |||||||
| LAT | |||||||
| Corticosterone ELISA | |||||||
| RT‐PCR | |||||||
| Alternanthera philoxeroides | EE of whole plant | ICR‐OVX mice (female) | NORT | 250 and 500 mg/kg/day | 17β‐Estradiol | It significantly improved the acquisition and retrieval of reference memory, ↓ by OVX, in a dose‐dependent mannerIt significantly improves discrimination performance in NORT (enhanced recognition memory)It restored spatial working memory deficits induced by OVXIt significantly ↑ the % of spontaneous alternationIt ↓ significant MDA values, indicating antioxidant activityIt ↓ IL‐1β, IL‐6, and TNF‐α mRNA expressionsIt normalized the expression of the PI3K and AKT genesMetabolomic analysis showed that 500 mg/kg of EE had better effects against OVX‐induced alterations, and the most relevant metabolites in the study were responsible for the galactose metabolic pathway | [ref. 141] |
| Y‐Maze Task | 250 and 500 mg/kg/day | 17β‐Estradiol | |||||
| MWMT | 250 and 500 mg/kg/day | 17β‐Estradiol | |||||
| Locomotor Activity Test | 250 and 500 mg/kg/day | 17β‐Estradiol | |||||
| Lipid peroxidation of the brain | 250 and 500 mg/kg (mice) | 17β‐Estradiol (mice) | |||||
| Bradford’s method (brain) | 250 and 500 mg/kg (mice) | 17β‐Estradiol (mice) and BSA (testes) | |||||
| RT‐PCR | 250 and 500 mg/kg (mice) | 17β‐Estradiol (mice) | |||||
| NMR‐metabolomic analysis | 250 and 500 mg/kg (mice) | 17β‐Estradiol (mice) | |||||
| Alternanthera philoxeroides | EE of whole plant | ICR mice (male) | Behavioral assessment | 250 and 500 mg/kg/day | Vitamin E | EE contributed to the maintenance of short‐term and long‐term memory in behavioral testsThe two EE concentrations evaluated were able to restore SOD and CAT levels to healthy conditionsEE activity regulates the expression of mTERT, mTRF1, and mTRF2, leading to a delay in telomere shortening | [ref. 142] |
| Biochemical assay | |||||||
| Determination of CAT and SOD activities | |||||||
| qPCR | |||||||
| Alternanthera sessilis | EE of whole plant | Swiss Albino mice | Pentobarbital‐induced sleeping time | 250 and 500 mg/kg | Caffeine | It ↑ the time required for the onset of sleep by 188.70% and 377.49% and ↓ its duration by 12.70% and 23.08%, at doses of 250 and 500 mg/kg, respectivelyLocomotion ↑, but after reaching maximum activity, the stimulating effect gradually ↓ with passage of time | [ref. 139] |
| Open field test | 250 and 500 mg/kg | — | |||||
| Hole cross test | 250 and 500 mg/kg | — | |||||
| Iresine celosia | EE of aerial parts | ICR mice (male) treated with LPS | Open field test | 30 or 100 mg/kg | — | 100 mg/kg significantly ↓ LPS‐induced activated microglia and number of S100β‐positive cellsIt ↑ ambulation in open field tests and improved T‐turn and T‐LAInhibition of microglia and astrocytes alleviated behavioral dysfunction | [ref. 104] |
| Pole test | |||||||
| Immunohistochemistry and image analysis | |||||||
| Pfaffia glomerata | EE of roots and rhizomes | Wistar rats (male) | Open field test | 500 mg/kg | Diazepam | Rats in the open‐field test exhibited behavior similar to those treated with diazepam500 mg/kg ↓ sleep latency and ↑ sleeping time, but 1000 mg/kg had no effect on latency and sleep duration500 mg/kg ↑ latency and a ↓ in the duration of the first convulsion, but 1000 mg/kg had no effectIt ↓ entries in enclosed arms500 and 1000 mg/kg ↓ step‐down latency in a []‐dependent manner1500 mg/kg showed a tendency to ↑ the memory retention, but this effect was not statistically significantNo antidepressant effect was observed | [ref. 245] |
| Elevated plus maze test | 500 mg/kg | Diazepam | |||||
| Step‐down inhibitory avoidance task | 100, 500, 1000, and 1500 mg/kg | Diazepam | |||||
| FST | 500 mg/kg | Imipramine hydrochloride | |||||
| Swiss mice (male) | Pentobarbital‐induced sleeping time | 500 and 1000 mg/kg | Diazepam | ||||
| Pentylenetetrazole‐induced convulsions | 500 and 1000 mg/kg | Diazepam | |||||
| Pfaffia glomerata | HaE of roots | Albino mice (male) | Spontaneous movement | 10 and 100 mg/kg | — | It causes abdominal contraction when administered intraperitoneallyIt ↓ motor activity and stereotypyAt 10 mg/kg, it ↓ scaling behavior and grooming100, 200, and 1000 mg/kg caused ruffled fur100 mg/kg ↓ sleep time but did not ← memory retention damage caused by scopolamineOld rats treated with HaE performed similarly to young rats in the discrimination test, demonstrating that HaE improved acquisition and retention of determined behaviorHaE partially reversed age‐associated memory deficit. It ↓ BW | [ref. 116] |
| Rota‐rod | 10 and 100 mg/kg | — | |||||
| Potentiation of sodium pentobarbital sleeping time | 10 and 100 mg/kg | — | |||||
| Albino mice treated with scopolamine | Passive avoidance test | 100 mg/kg | — | ||||
| Young and old Wistar rats (male) | Passive avoidance test | 100 mg/kg | — | ||||
| Old Wistar rats | Right–left discrimination test | 100 mg/kg | — | ||||
| Pfaffia glomerata | HF of ME of roots | C57BL/6J mice (male) with acute stress | Open‐field test | 3, 10, or 30 mg/kg | — | Avoid ↑ motor function of mice (↓ stress‐related behavior)It ↓ depressive‐like behaviorsAt 30 mg/kg, it ↓ the time spent in closed arms after Day 2 and gradually ↓ the time spent in the open arms of the maze. At 10 mg/kg, it ↓ immobility time after Day 2 (protective effect in anxiety development)It did not show any effect in the rotation testIt prevented the ↓ of SOD and GPx activity in the cortex and striatum, but not in the hippocampus. It restores CAT activity in the striatum, but not in the cortex or hippocampus | [ref. 114] |
| Elevated plus maze test | 3, 10, or 30 mg/kg | — | |||||
| FST | 3, 10, or 30 mg/kg | — | |||||
| Rotation test | 3, 10, or 30 mg/kg | Diazepam | |||||
| Biochemical analysis of tissues | 3, 10, or 30 mg/kg | — | |||||
| Wound‐healing activity | |||||||
| Alternanthera brasiliana | Ointment of ME of leaves | Sprague Dawley rats (excision wound model) | Wound area contraction measures | 5% | Himax | ME ointment completely contracted the excision wound (100%), showing greater activity than HimaxMice treated with ME ointment had a granulation tissue completely filled with epidermal cells covered by a thick layer of keratinME ointment generated a tensile strength of the healing tissue of 4.861 ± 0.664, indicating wound‐healing activity | [ref. 244] |
| Histopathological study | |||||||
| Sprague Dawley rats (incision wound model) | Wound area contraction measures | 5% | Himax | ||||
| Alternanthera brasiliana | Ointment of ME of leaves | Sprague Dawley rats (dermal burn wound) | Wound area contraction measures | 5% | Himax | On Day 8, it ↓ the wound area by 92.13%, showing greater activity than Himax (72%)It ↑ protein and hydroxyproline contentIt ↑ CAT, GSH, and SOD levels in granulation tissueIt ↑ Vitamin C levelsME has good wound‐healing activity, as histopathological studies showed collagen fiber deposition and a keratin layer, indicating tissue recovery and regeneration | [ref. 250] |
| Biochemical estimations | |||||||
| Histopathological study | |||||||
| Alternanthera brasiliana | Ointment of ME of leaves | Immunocompromised Sprague Dawley rats with HC | Wound area contraction measures | 2.5%, 5.0%, and 7.5% | Himax | An ointment with 5% ME achieved 77.10% wound contraction on Day 8, outperforming Himax (60%)It ↑ GSH, CAT, and SOD levels, as well as protein content in granulation tissuesAt 5%, the highest levels of hydroxyproline and vitamin C were observed, exceeding those of HimaxMice treated with ME showed abundant collagen fibers, fibroblast proliferation, angiogenesis, and development of basement membrane beneath the necrotic debris | [ref. 251] |
| Biochemical estimations | |||||||
| Histopathological Study | |||||||
| Alternanthera brasiliana | Ointment of ME of leaves | Aged Sprague Dawley rats (aged wound model) | Wound area contraction measures | 5% | — | The % of wound contraction ↑ to 97.62 ± 0.14 on Day 21, indicating wound‐healing activityIt ↑ the content of collagen, elastin, and hydroxyproline by 16.33% ± 0.42%, 6.67 ± 0.42%, and 64.67 ± 3.83 (mg/g) on Day 21, respectivelyGranulation tissues showed abundant collagen fibers and re‐epithelization | [ref. 252] |
| Biochemical estimations | |||||||
| Histopathological Study | |||||||
| Alternanthera brasiliana | HaE of leaves | Wistar rats (male‐trichotomy was performed on the dorsum skin and a dermatological punch of 1.5 cm in diameter) | Re‑epithelialization analysis | 20% HaE of leaves in 2% carbopol gel | — | According to histomorphometry analysis, the extract did not induce a significant fibroblast proliferationAccording to biochemical studies, the collagen formation ↑ on the 2nd day. Additionally, they observed that it stimulated wound healing, as it gradually reduced collagen III levels, and after Day 21, collagen I increased | [ref. 195] |
Abbreviations: ACh, acetylcholine; AF, aqueous fraction; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CAT, catalase; CFA, Freund’s complete adjuvant; DBP, diastolic blood pressure; EE, ethanolic extracts; ELISA, enzyme‐linked immunosorbent assay; FD, fraction of DCM; FEA, fraction of ethyl acetate; FH, fraction of hexane; GLB, glibenclamide; HCTZ, hydrochlorothiazide; MABP, mean arterial blood pressure; MCW, made with cold water; ME, methanolic extracts; MHW, made with hot water; MPO, myeloperoxidase; NA, not applicable; OGTT, oral glucose tolerance test; PFC, plaque forming cells; RBC, red blood cells; ROS, reactive oxygen species; SNP, sodium nitroprusside; SOD, superoxide dismutase.
The cytotoxicity of pfaffoside A (255), pfaffoside C (257), pfaffoside D (258), pfaffoside E (259), and pfaffoside F (260) was evaluated against B‐16 cells, with inhibitory concentrations between 30 and 120 µg/mL as seen in Table S2 [ref. 125, ref. 164]. Philoxeroideside A (261), philoxeroideside B (262), philoxeroideside C (263), and philoxeroideside D (264) were evaluated against HL60 and SK‐N‐SH cell lines, with IC50 values ranging from 37.29 to 271.45 µg/mL. Compound 264 exhibited the highest activity, with IC50 values of 45.93 and 37.29 µg/mL, respectively [ref. 37].
Additionally, compounds 11α,12α‐epoxy‐3β‐[(O‐β‐d‐galactopyranosyl‐(1 → 3)‐O‐[β‐d‐glucopyranosyl‐(1 → 2)]‐β‐d‐glucuronopyranosyl)‐oxy]olean‐28,13‐olide (243), 11α,‐12α‐epoxy‐3β‐[(O‐β‐d‐glucuronopyranosyl)oxy]olean‐28,13‐olide (244), 11α,‐12α‐epoxy‐3β‐[(O‐β‐d‐glucuronopyranosyl)oxy]taraxer‐14‐en‐28‐oic acid β‐d‐glucopyranosyl ester (245), 11α,12α‐epoxy‐3β‐hydroxyolean‐28,13‐olide (246), and 11α,12α‐epoxy‐3β‐hydroxytaraxer‐14‐en‐28‐oic acid (247) were evaluated against HSC‐2, but only compounds 246 and 247 showed activity, with IC50 values of 20 µM [ref. 99]. These results confirm the potential of most terpenoid‐type compounds as anticancer agents.
Hypoglycemic activity was evaluated in ilimaquinone (187) and neodactyloquinone (190), showing different levels of activity, with the compound 187 showing the highest efficacy [ref. 84].
Pfaffianol A (225), boussingoside A2 (235), pfaffiaglycosides B (254), and pfaffoside C (257) were evaluated for their effects on melanogenesis inhibition. The results indicated that only compounds 225 and 257 had a significant effect, even greater than that of arbutin [ref. 161], as shown in Table 9.
TABLE 9: Other activities of the Gomphrenoideae subfamily.
| Species | Extract(s)/Compounds | Assay method | Model | Dose | Positive control | Activity | References | ||
|---|---|---|---|---|---|---|---|---|---|
| Values | Analysis | ||||||||
| Activity in blood cells | |||||||||
| Alternanthera bettzickiana | AqE of leaves | Emmel test | Sickle cell blood | 0, 5, 10, 15, and 20 µg/mL | Parahydroxybenzoic acid | % of normalization: 86 for 11 µg/mL | It has an antisickling effect since the RBCs maintain a circular (biconcave) and normal shape | [ref. 18] | |
| Pfaffia paniculata | Commercial capsule powder | Ektacytometric studies | RBCs | 0–0.5 mg/mL | ↑ The deformity index indicates improvement in cellular hydration, which is independent of density. Also ↑ the mean corpuscular volume, Na+, and ↓ the mean cell hemoglobin [] | [ref. 239] | |||
| Pfaffia paniculata | Extract of commercial powder | Hematological and RBC deformability measurement | RBCs | 0.2 or 0.5 mg/mL | 0.5 mg/mL ↑ the deformity of falciform cellsHad no effect on the RBC’s aggregation properties | [ref. 240] | |||
| RBC aggregation | RBCs | 0.2 or 0.5 mg/mL | |||||||
| Activity related to the urinary system | |||||||||
| Gomphrena celosioides | 367 | Xanthine oxidase inhibitory activity | — | Uninformed | Allopurinol | IC50: 81.04 ± 0.49 µM | The inhibitory action of xanthine oxidase was most evident in compound 512 | [ref. 183] | |
| 512 | Uninformed | Allopurinol | IC50: 33.78 ± 0.49 µM | ||||||
| 20‐Hydroxyecdysone‐20,22‐monoacetonide | Uninformed | Allopurinol | IC50: 101.15 ± 0.48 µM | ||||||
| Anti‐aging activities | |||||||||
| Gomphrena globosa | HaE of flowers | Determination of anti‐elastase activity | Neutrophil elastase | 100 and 250 µg/mL | SPCK | NDNS | Elastase and collagenase inhibitory activity were [] dependentHaE has significant anti‐collagenase properties | [ref. 156] | |
| Determination of anti‐collagenase activity | Collagenase | 100 and 250 µg/mL | 1,10‐Phenanthroline | %I 250 µg/mL about 40% | |||||
| Anti‐allergy activity | |||||||||
| Alternanthera sessilis | EE of aerial parts | Measurement of LDH release | RBL‐2H3 cells | 25, 50, and 100 µg/mL | — | NE | ↓ The release of β‐hexosaminidase in a []‐dependent manner↓ Intracellular Ca2+ levelsSuppresses the release of IL‐6, TNF‐α, IL‐4, and IL‐13↓ Degradation of IkBα and nuclear translocation of p65 NF‐kBSuppresses antigen‐induced degranulation of RBL‐2H3 cell | [ref. 241] | |
| β‐Hexosaminidase secretion assay | RBL‐2H3 cells sensitized with DNP‐specific IgE | 25, 50, and 100 µg/mL | — | NDNS | |||||
| Ca2+ measurement | RBL‐2H3 cells sensitized with DNP‐specific IgE | 25, 50, and 100 µg/mL | — | NDNS | |||||
| Measurement of IL‐6, TNF‐α, IL‐13, and IL‐4 (ELISA= | RBL‐2H3 cells sensitized with DNP‐specific IgE | 25, 50, and 100 µg/mL | — | NDNS | |||||
| Western Blot Analysis | RBL‐2H3 cells sensitized with DNP‐specific IgE | 25, 50, and 100 µg/mL | — | NDNS | |||||
| Antiarthritic activity | |||||||||
| Alternanthera bettzickiana | EE of aerial parts | Egg albumin denaturation inhibition | — | 50, 100, 200, 400, 800, 1600, 3200, and 6400 µg/mL | Diclofenac sodium | %I 6400 µg/mL: 94.23% | Dose‐dependent activityThe results of the EE were better than those of the standard drug | [ref. 19] | |
| Protein denaturation using BSA | %I 6400 µg/mL: 97.43 ± 0.70 | ||||||||
| Anticoagulant activity | |||||||||
| Alternanthera philoxeroides | ME of whole plant | PTT test | 250, 500, and 1000 µg/mL | — | To 1000 µg/mL: 13.26 | Showed significant anticoagulant activity of MEAccording to PT, vanillic acid exhibited the highest anticoagulant activity, whereas in aPTT, tannic acid had the highest activity | [ref. 138] | ||
| aPTT tests | — | To 1000 µg/mL: 66.28 | |||||||
| Tannic acid | PTT test | 10 µg/mL | — | 12.62 | |||||
| aPTT tests | 10 µg/mL | — | 57.54 | ||||||
| Gallic acid | PTT test | 10 µg/mL | — | 13.33 | |||||
| aPTT tests | 10 µg/mL | — | 56.26 | ||||||
| Catechin | PTT test | 10 µg/mL | — | 11.71 | |||||
| aPTT tests | 10 µg/mL | — | 56.87 | ||||||
| Vanillic acid | PTT test | 10 µg/mL | — | 15.91 | |||||
| aPTT tests | 10 µg/mL | — | 56.14 | ||||||
| Antidepressant activity | |||||||||
| Alternanthera philoxeroides | EE of whole plant | Estrogenic activity (cell‐based assay) | MCF‐7 | 1–100 µg/mL | 17β‐Estradiol | EqE100 = 1.68 µg/mL | 1.68 µg/mL is equally effective as 100 pM of 17β‐estradiol | [ref. 38] | |
| Anti‐diarrhea and/or anti‐dysentery activity | |||||||||
| Alternanthera repens | AqE of leaves | []–response curves to CaCl2 | Ileum of rats | 0.56, 1.09, or 2.09 mg/mL | — | IC50: 0.25 ± 0.03 mM at a [] of 0.56 mg/mL | AqE, ME, and F2 to F4 have a spasmolytic effect on the CaCl2‐induced contractions, with ME showing the highest activityME, AqE, and F2 to F4 inhibit the stimulatory effect of KCl; therefore, their mechanism of action is CCBAll extracts and F2 to F4 inhibited 5‐HT‐induced ileum contractions, presenting a spasmolytic effect | [ref. 47] | |
| Relaxant effect on K+‐C | Ileum of rats | 0.56, 1.09, or 2.09 mg/mL | — | IC50: 0.82 ± 0.01 mM at a [] of 0.56 mg/mL | |||||
| Inhibition of dose–response curves to 5‐HT | Ileum of rats | 0.56, 1.09, or 2.09 mg/mL | — | IC50: 7.19 ± 0.04 × 10−8 M at a [] of 0.56 mg/mL | |||||
| []–response curves to ACh | Ileum of rats | 2.09 mg/mL | — | %I: 58.6IC50: 72.68 ± 0.08 × 10−6 M | |||||
| HeE of leaves | []–response curves to CaCl2 | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | NE | ||||
| Relaxant effect on K+‐C | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | NE | |||||
| Inhibition of dose–response curves to 5‐HT | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | IC50: 5.44 ± 0.08 × 10−6 M at a [] of 0.24 mg/mL | |||||
| ME of leaves | []–response curves to CaCl2 | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | IC50: 0.18 ± 0.061 mM at a [] of 0.24 mg/mL | ||||
| Relaxant effect on K+‐C | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | IC50: 0.043 ± 0.001 mM at a [] of 0.24 mg/mL | |||||
| Inhibition of dose–response curves to 5‐HT | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | IC50: 2.24 ± 0.06 × 10−7 M at a [] of 0.24 mg/mL | |||||
| Six DF of ME | []–response curves to CaCl2 | Ileum of rats | 0.66 mg/mL | — | NDNS | ||||
| Relaxant effect on K+‐C | Ileum of rats | 0.66 mg/mL | — | NDNS | |||||
| Inhibition of dose–response curves to 5‐HT | Ileum of rats | 0.24, 0.47, or 0.91 mg/mL | — | NDNS | |||||
| Alternanthera sessilis | EE of whole plant | Relaxant and/or contractile | Jejunum of rabbit | Uninformed | Verapamil | EC50: 0.26 mg/mL SRCEC50: 0.08 mg/mL K+‐SC | EE and DF relax SRC and Kþ‐SC, but AF only has a spasmolytic effect on CRSEE acts as CCB and generates the repolarization of the ΔΨmDF showed greater CCB activity than FA | [ref. 64] | |
| Evaluation of CCB | Jejunum of rabbit | 0.1–0.3 mg/mL | Verapamil | ||||||
| DF of EE | Relaxant and/or contractile | Jejunum of rabbit | Uninformed | Verapamil | EC50: 0.02 mg/mL SRCEC50: 0.04 mg/mL K+‐SC | ||||
| Evaluation of CCB | Jejunum of rabbit | Uninformed | Verapamil | ||||||
| AF of EE | Relaxant and/or contractile | Jejunum of rabbit | Uninformed | Verapamil | EC50: 0.36 mg/mL SRCEC50: NE | ||||
| Evaluation of CCB | Jejunum of rabbit | Uninformed | Verapamil | ||||||
| Antihypertensive effect | |||||||||
| Alternanthera sessilis | EE of whole plant | Vasorelaxant activity | Aorta of rabbit constricted by PE or K+ | Uninformed | Verapamil | EC50 (mg/mL): 2.03 and 0.34 for PE and K+ induced contraction, respectively | DF exhibited the highest vasorelaxant effectEE and DF reinforce the presence of the CCB mechanism | [ref. 64] | |
| DF of EE | Vasorelaxant activity | Aorta of rabbit constricted by PE or K+ | Uninformed | Verapamil | EC50 (mg/mL): 0.32 and 0.15 for PE and K+ induced contraction, respectively | ||||
| AF of EE | Vasorelaxant activity | Aorta of rabbit constricted by PE or K+ | Uninformed | Verapamil | NE | ||||
| Antiparasitic activity | |||||||||
| P (min for 10 mg/mL) | D (min for 10 mg/mL) | ||||||||
| Gomphrena celosioides | EaE of whole plant | Anthelmintic assay (P and D) | F. gigantica | 10, 20, 30, 40, 50, 70, 80, and 100 mg/mL | Piperazine citrate | 20 ± 0.8 | 35 ± 0.5 | ME was more potent in causing paralysis of P. posthuma than EaE and the positive control | [ref. 82] |
| P. posthuma | 40 ± 0.2 | >60 | |||||||
| T. solium | 15 ± 0.4 | 40 ± 0.3 | |||||||
| ME of whole plant | F. gigantica | 10, 20, 30, 40, 50, 70, 80, and 100 mg/mL | Piperazine citrate | 40 ± 0.5 | 45 ± 0.1 | ||||
| P. posthuma | 8 ± 0.9 | 60 ± 0.5 | |||||||
| T. solium | 37 ± 0.3 | 42 ± 0.3 | |||||||
| Antitumor activity | |||||||||
| Alternanthera pungens | AqE of whole plant | Potato disc method | A. tumefaciens | 10, 100, 1000, and 10 000 ppm | Vincristine | IC50 (ppm): 1800 | Dose‐dependent activity | [ref. 16] | |
| ME of whole plant | Potato disc method | A. tumefaciens | 10, 100, 1000, and 10 000 ppm | Vincristine | IC50 (ppm): 11 | ||||
| nHE of whole plant | Potato disc method | A. tumefaciens | 10, 100, 1000, and 10 000 ppm | Vincristine | IC50 (ppm): 90 | ||||
| Cardioprotective activity | |||||||||
| Alternanthera philoxeroides | ME of leaves | MTT assay | Rat cardiac H9c2 cells with DOX‐induced apoptosis | 10, 20, 40, 80, and 160 mg/mL | — | %V 10 mg/mL: 38.43 ± 11.5%V 20 mg/mL: 66.33 ± 6.03%V 40 mg/mL: 79.00 ± 3.6%V 80 mg/mL: 84.33 ± 5.5%V 160 mg/mL: 83.16 ± 8.12 | ↑ Cell viability, indicating a protective effect against DOX‐mediated cytotoxicity↓ The cell apoptosis induced by DOX | [ref. 42] | |
| Annexin V‐FITC/PI staining assay | Rat cardiac H9c2 cells with DOX‐induced apoptosis | 10, 20, 40, 80, and 160 mg/mL | — | %A 10 mg/mL: 51.18%A 20 mg/mL: 42.5%A 40 mg/mL: 33.18%A 80 mg/mL: 25.2%A 160 mg/mL: 23.46 | |||||
| Endothelial activation | |||||||||
| Alternanthera sessilis | EE of whole plant | MTT assay | HAECs cells | 25, 50, 100, 200, 400, and 800 µg/mL | — | 6.25–200 µg/mL of EE does not affect the viability of HAECs200 µg/mL of EE ↓ the ↑ permeability stimulated by TNF‐αEE does not alter the secretion of sVCAM‐1 induced by TNF‐α200 µg/mL of EE suppresses the release of intracellular ROS stimulated by TNF‐αEE does not ↓ the elevated production of H2O2 induced by TNF‐αEE ↑ SOD activity in a dose‐dependent mannerEE enhances CAT activity in cells exposed to by H2O2 | [ref. 153] | ||
| FITC‐dextran permeability assay | HAECs cells | 25–200 µg/mL | Simvastatin | ||||||
| sVCAM‐1 production assay | HAECs cells | 25, 50, 100, and 200 µg/mL | NAC | ||||||
| ROS quantitative assay | HAECs cells | 25, 50, 100, and 200 µg/mL | — | ||||||
| H2O2 | HAECs cells | 25, 50, 100, and 200 µg/mL | Simvastatin | ||||||
| SOD activity | HAECs cells | 25, 50, 100, and 200 µg/mL | Simvastatin, NAC, or dexamethasone | ||||||
| CAT activity | HAECs cells | 25, 50, 100, and 200 µg/mL | Simvastatin | ||||||
| Hemolytic activity | |||||||||
| Pfaffia glomerata | FD of roots | Hemolysis assay | Blood from 6‐ to 8‐week‐old male C57BL/6 mice | 250, 25, 2.5, and 0.25 µg/mL | Saponin from Quillaja sp. | NDNS | The two FDs did not cause hemolytic effects | [ref. 167] | |
| FD of aerial part | |||||||||
| Inhibition of melanogenesis | |||||||||
| Pfaffia glomerata | 225 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: 80.0 ± 2.5IC50: 44 µM | Pfaffianol A (225) and pfaffoside C (257) substantially inhibited melanogenesis without cytotoxic effects, showing stronger effects than arbutinThe other compounds showed no effect | [ref. 166] | |
| Viability assay | |||||||||
| 254 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 10 µM: −1.2 ± 1.6 | ||||
| 235 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: 22.1 ± 2.4 | ||||
| 367 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: −6.1 ± 7.6 | ||||
| 384 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 30 µM: 1.9 ± 3.5 | ||||
| 381 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: −3.3 ± 3.0 | ||||
| 377 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 30 µM: 7.4 ± 2.2 | ||||
| 385 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: 2.3 ± 4.2 | ||||
| 257 | Melanogenesis assay | Murine B16 melanoma 4A5 cells RCB0557 | 0, 1, 3, 10, 30, and 100 µM | Arbutin | %I 100 µM: 51.4 ± 1.2IC50: 92 µM | ||||
| Immunomodulatory activity | |||||||||
| Alternanthera brasiliana | AqE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | Aqueous and ethanol extracts inhibit the proliferative response of lymphocytes to PHA10 µg/mL of FEA completely inhibits lymphocyte proliferation | [ref. 242] | ||
| EE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | |||||
| Alternanthera maritima | AqE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | ||||
| EE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | |||||
| Alternanthera tenella | AqE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | ||||
| EE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | |||||
| FEA of AqE of leaves | Proliferation assay | PBMN stimulated with PHA | 0–200 µg/mL | — | |||||
| Alternanthera maritima | EE of aerial parts | Cytotoxicity and LDH assay | PMNLs | 100 µg/mL | Total cell lysis | %V: 78.50 ± 1.50 and LA (IU*1000): 1.39 ð 0.07 | EE, BuF, and the seven isolated compounds do not induce significant LDH release, nor do they exhibit cytotoxicity against human PMNL | [ref. 149] | |
| BuF of EE of aerial parts | Cytotoxicity and LDH assay | PMNLs | 100 µg/mL | Total cell lysis | %V: 82.00 ± 1.00 and LA (IU*1000): 0.76 ± 0.17 | ||||
| 25 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 97.75 ± 0.35 and LA (IU*1000): 5.22 ± 0.27 | ||||
| 24 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 91.25 ± 1.77 and LA (IU*1000): 6.29 ± 3.20 | ||||
| 129 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 94.50 ± 0.71 and LA (IU*1000): 5.63 ± 1.01 | ||||
| 50 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 87.00 ± 2.83 and LA (IU*1000): 4.13 ± 1.46 | ||||
| 43 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 91.50 ± 3.54 and LA (IU*1000): 4.08 ± 1.93 | ||||
| 72 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 87.50 ± 0.71 and LA (IU*1000): 5.81 ± 1.64 | ||||
| 34 | Cytotoxicity and LDH assay | PMNLs | 50 µmol/L | Total cell lysis | %V: 93.50 ± 1.41 and LA (IU*1000): 5.74 ± 2.27 | ||||
| Gomphrena celosioides | Extract | Proliferation assay (MTT) | RAW 264.7 | 0–100 µg/mL | — | IC50: >100 | It has no cytotoxic effect against RAW 264.7 cells, nor does it enhance phagocytosisSignificantly inhibits TNF‐α production | [ref. 213] | |
| Phagocytosis assay | RAW 264.7 | 10 µg/mL | — | NDNS | |||||
| Measurement of TNF‐α | RAW 264.7 | 10 µg/mL | — | %I: 32.5 | |||||
| Gomphrena virgata | AqE of roots | Viability assay (trypan blue) | PBMN | 1, 0.5, and 0.025 mg/mL | Cadmium chloride | %V 1 mg/mL: 95.86 ± 7.12 (24 h) and %V 1 mg/mL: 69.14 ± 26.69 (7 days) | c. | [ref. 101] | |
| Insecticidal activity | |||||||||
| Gomphrena elegans Mart. | AqE, ClE, HeE, and ME of leaves | Insecticidal activity tests | Larvae of Aedes aegypti | 10 mg/L | Bt | None of the extracts exhibited insecticidal activity. | [ref. 159] | ||
| Larvicidal activity | |||||||||
| Alternanthera sessilis | AgNPs of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver nitrate | LC50: 7.2 (24 h), 4.63 (48 h), and 2.93 (72 h) | For the three larval models, both the AgNPs complex and the different extracts exhibited a dose‐dependent effectHeE stands out as having the greatest larvicidal effect compared to the others | [ref. 163] | |
| Culex quinquefasciatus | LC50: 9.28 (24 h), 5.43 (48 h), and 2.82 (72 h) | ||||||||
| Anopheles stephensi | LC50: 12.92 (24 h), 3.29 (48 h), and 2.9 (72 h) | ||||||||
| ME of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver nitrate | LC50: 88.75 (24 h), 82.21 (48 h), and 71.74 (72 h) | ||||
| Culex quinquefasciatus | LC50: 32.32 (24 h), 26.59 (48 h), and 20.63 (72 h) | ||||||||
| Anopheles stephensi | LC50: 40.13 (24 h), 23.98 (48 h), and 17.26 (72 h) | ||||||||
| HeE of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver Nitrate | LC50: 24.41 (24 h), 20.72 (48 h). and 17.03 (72 h) | ||||
| Culex quinquefasciatus | LC50: 26.25 (24 h), 20.63 (48 h). and 16.03 (72 h) | ||||||||
| Anopheles stephensi | LC50: 47.48 (24 h), 39.64 (48 h). and 30.23 (72 h) | ||||||||
| ClE of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver nitrate | LC50: 31.90 (24 h), 26.24 (48 h), and 18.83 (72 h) | ||||
| Culex quinquefasciatus | LC50: 49.19 (24 h), 35.22 (48 h), and 22.47 (72 h) | ||||||||
| Anopheles stephensi | LC50: 45.97 (24 h), 34.16 (48 h), and 26.28 (72 h) | ||||||||
| AcE of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver nitrate | LC50: 85.35 (24 h), 81.67 (48 h), and 67.43 (72 h) | ||||
| Culex quinquefasciatus | LC50: 39.85 (24 h), 28.38 (48 h), and 20.73 (72 h) | ||||||||
| Anopheles stephensi | LC50: 29.75 (24 h), 23.98 (48 h), and 19.02 (72 h) | ||||||||
| PEE of leaves | Mosquito culture and larvicidal bioassays | Aedes aegypti | Uninformed | Silver nitrate | LC50: 156.87 (24 h), 150.22 (48 h), and 119.38 (72 h) | ||||
| Culex quinquefasciatus | LC50: 37.79 (24 h), 30.23 (48 h), and 18.83 (72 h) | ||||||||
| Anopheles stephensi | LC50: 59.0 (24 h), 49.21 (48 h), and 37.93 (72 h) | ||||||||
| Neuroprotective or neurological activity | |||||||||
| Alternanthera philoxeroides | EE of whole plant | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 252.9 ± 0.02 µM | EE and isolated compounds inhibit MAO‐A and MAO‐BEE and isolated compounds, except for compound 35, show partial selectivity for MAO‐B. Alternatin exhibits partial selectivity by MAO‐A | [ref. 38] | |
| MAO‐B | Deprenyl | IC50: 90.69 ± 0.02 µM | |||||||
| 35 | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 0.00046 ± 0.04 µM | ||||
| MAO‐B | Deprenyl | IC50: 0.00060 ± 0.12 µM | |||||||
| 36 | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 0.00206 ± 0.04 µM | ||||
| MAO‐B | Deprenyl | IC50: 0.00022 ± 0.12 µM | |||||||
| 79 | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 18.37 ± 1.47 µM | ||||
| MAO‐B | Deprenyl | IC50: 0.6748 ± 0.46 µM | |||||||
| 5 | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 0.0541 ± 0.01 µM | ||||
| MAO‐B | Deprenyl | IC50: 0.1293 ± 0.42 µM | |||||||
| 41 | Inhibitory assay | MAO‐A | Uninformed | Clorgyline | IC50: 3.051 ± 0.35 µM | ||||
| MAO‐B | Deprenyl | IC50: 0.5441 ± 0.33 µM | |||||||
| Alternanthera philoxeroides | EE of whole plant | Thioflavin‐T assay | Aβ1‐42 solution | 100 µg/mL | Cur | %I: 83.25 ± 4.25 | Among the isolated compounds, compound 36 exhibits the greatest inhibitory activity against the formation of toxic Aβ plaques in the brain, surpassing that of Cur | [ref. 141] | |
| 35 | Thioflavin‐T assay | Aβ1‐42 solution | 100 µM | Cur | NDNS | ||||
| 36 | Thioflavin‐T assay | Aβ1‐42 solution | 100 µM | Cur | %I: 81.96 ± 2.14 | ||||
| 41 | Thioflavin‐T assay | Aβ1‐42 solution | 100 µM | Cur | NDNS | ||||
| 79 | Thioflavin‐T assay | Aβ1‐42 solution | 100 µM | Cur | NDNS | ||||
| 5 | Thioflavin‐T assay | Aβ1‐42 solution | 100 µM | Cur | NDNS | ||||
| Alternanthera philoxeroides | EE of whole plant | Ellman’s method | AChE | Uninformed | Tacrine | IC50:2.06 ± 0.016 mg/mL | The SIs of the EE was 1.60, indicating its partial selectivity toward AChE | [ref. 141] | |
| BChE | IC50: 3.27 ± 0.011 mg/mL | ||||||||
| Gomphrena globosa L. | AqE or EB or commercial preparation of inflorescences | Ellman’s method | AChE | Uninformed | — | NE | None of the extracts is capable of inhibiting AChE | [ref. 98] | |
| Iresine celosia | EE of aerial part | WST‐1 assay | Mouse BV2 microglial cells stimulated with/without LPS | 1, 10, or 100 µg/mL | Quercetin | No cytotoxic activity was observed against BV2 cells. 100 µg/mL ↓ NO production and PGE2 levelsSignificantly ↓ MAPK phosphorylation, without affecting total protein levels of MAPK factors100 µg/mL significantly ↓ transcriptional activity of NF‐κBSignificantly suppresses the phosphorylation of p65 and its translocation to the nucleusCan inhibit LPS‐induced neuroinflammation without cytotoxicity in BV2 cells, as well as suppress the expression of proinflammatory mediators mediated by the MAPKs/NF‐κB signaling pathway30 and 100 µM of Iresin (283) significantly ↓ NO production | [ref. 104] | ||
| Measurement of NO and PGE2 | Mouse BV2 microglial cells stimulated with LPS | 1, 10, or 100 µg/mL | Quercetin | ||||||
| qRT‐PCR | Mouse BV2 microglial cells | 1, 10, or 100 µg/mL | |||||||
| Western blotting | Mouse BV2 microglial cells stimulated with LPS | 1, 10, or 100 µg/mL | |||||||
| Luciferase assay | Mouse BV2 microglial cells stimulated with LPS | 100 µg/mL | |||||||
| 283 | Measurement of NO and PGE2 | Mouse BV2 microglial cells stimulated with LPS | 1, 10, 30, and 100 µM | Quercetin | |||||
| Iresine herbstii | AqE of aerial part | 5‐HT1A serotoninergic assay | Cerebral cortex of rats | 25, 50, 75, 100, and 125 µg/mL | 8‐OH‐DPAT | %I: 13.51 | AqE and ME show no affinity for 5‐HT2A receptor, and additionally, the ME did not show affinity against the D2 receptorAqE and ME have an effect on the CNS similar to that observed with some psychotropic agents | [ref. 108] | |
| 5‐HT2C serotoninergic assay | Frontal cortical regions of rats | 25, 50, 75, 100, and 125 µg/mL | Mesulergine | %I: 22.13 | |||||
| D1 dopaminergic assay | Corpora striata | 25, 50, 75, 100, and 125 µg/mL | Spiroperidol | %I: 48.32 | |||||
| D2 dopaminergic assay | Corpora striata | 25, 50, 75, 100, and 125 µg/mL | Spiroperidol | %I: 88.82IC50: 32.08 ± 0.52 | |||||
| α1‐Adrenergic binding assay | Brain cortex | 25, 50, 75, 100, and 125 µg/mL | Prazosin | %I: 13.51 | |||||
| ME of aerial part | 5‐HT1A serotoninergic assay | Cerebral cortex of rats | 25, 50, 75, 100, and 125 µg/mL | 8‐OH‐DPAT | %I: 22.44 | ||||
| 5‐HT2C serotoninergic assay | Frontal cortical regions of rats | 25, 50, 75, 100, and 125 µg/mL | Mesulergine | %I: 92.46IC50: 34.78 ± 1.80 | |||||
| D1 dopaminergic assay | Corpora striata | 25, 50, 75, 100, and 125 µg/mL | Spiroperidol | %I: 90.52IC50: 19.63 ± 2.10 | |||||
| α1‐Adrenergic binding assay | Brain cortex | 25, 50, 75, 100, and 125 µg/mL | Prazosin | %I: 11.76 | |||||
| Iresine herbstii | AqE of aerial part | 5‐HT1A serotoninergic assay | Cerebral cortex of rats | 7.8–125 µg/mL | — | %I 125 µg/mL: 13.51 | AqE and ME have no affinity for the 5‐HT2A receptor. Additionally, ME did not exhibit affinity for the D2 and α2 receptors, while AqE showed no affinity for the α1 receptorAqE and ME interact with 5‐HT receptors, indicating an effect on the CNS | [ref. 243] | |
| 5‐HT2C serotoninergic assay | Frontal cortical regions of rats | 7.8–125 µg/mL | — | %I 125 µg/mL: 22.13 | |||||
| D1 dopaminergic assay | Corpora striata | 7.8–125 µg/mL | — | %I 125 µg/mL: 48.32 | |||||
| D2 dopaminergic assay | Corpora striata | 7.8–125 µg/mL | — | IC50: 2.99 ± 0.02 µg/mL | |||||
| α2‐Adrenergic binding assay | Brain cortex | 7.8–125 µg/mL | — | %I 125 µg/mL: 25.73 | |||||
| ME of aerial part | 5‐HT1A serotoninergic assay | Cerebral cortex of rats | 7.8–125 µg/mL | — | %I 125 µg/mL: 22.44 | ||||
| 5‐HT2C serotoninergic assay | Frontal cortical regions of rats | 7.8–125 µg/mL | — | IC50: 60.27 ± 5.59 µg/mL | |||||
| D1 dopaminergic assay | Corpora striata | 7.8–125 µg/mL | — | IC50: 1.39 ± 1.70µg/mL | |||||
| α1‐Adrenergic binding assay | Brain cortex | 7.8–125 µg/mL | — | %I 125 µg/mL: 11.76 | |||||
| Pfaffia glomerata | ME of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | Only the FD of aerial part showed activity | [ref. 167] | |
| ME of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FnH of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FnH of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FD of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FD of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | IC50: 2.7 µg/mL | ||||
| FEA of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FEA of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FnB of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| FnB of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| AF of roots | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| AF of aerial part | Sharma and Bhat method | AChE | 10 mg/mL | Galantamine | NE | ||||
| Sun protective effect | |||||||||
| Gomphrena globosa | HaE of flowers | Determination of SPF | — | 50 µg/mL | — | SPF of about 20 | [ref. 156] | ||
| Treatment of respiratory diseases | |||||||||
| Alternanthera sessilis | EE of whole plant | Assessment of bronchodilator activity | Tracheal tissue of rabbit induced with CCh or K+ | Uninformed | Verapamil | EC50: 0.22 and 0.18 mg/mL for CCh and K+ induced contraction, respectively | EE and DF decrease the maximum contractile effect similar to verapamil through CCBIt should be noted that CCBs are beneficial as bronchodilators | [ref. 64] | |
| DF of EE | Assessment of bronchodilator activity | Tracheal tissue of rabbit induced with CCh or K+ | Uninformed | Verapamil | EC50: 0.04 and 0.03 mg/mL for CCh and K+ induced contraction, respectively | ||||
| AF of EE | Assessment of bronchodilator activity | Tracheal tissue of rabbit induced with CCh or K+ | Uninformed | Verapamil | NE | ||||
| Wound‐healing activity | |||||||||
| Alternanthera brasiliana | ME of leaves | CM model | Embryonated chicken eggs | 200 and 400 µg | — | ME has dose‐dependent angiogenetic activity, ranging from mild (200 µg) to marked (400 µg) | [ref. 244] | ||
| Alternanthera sessilis | EE of stem | Scratch assay | NHDF cells | 12.5, 25, and 50 µg/mL | Allantoin | ↑ Migration 50 µg/mL: 86% | Dose‐dependent activity was observed in all cell lines | [ref. 57] | |
| Scratch assay | HDF‐D cells | 12.5, 25, and 50 µg/mL | Allantoin | ↑ Migration 50 µg/mL: 65% | |||||
| Scratch assay | HaCaT cells | 12.5, 25, and 50 µg/mL | Allantoin | ↑ Migration 50 µg/mL: 99% | |||||
| Iresine herbstii | EE of leaves | NF‐κB electrophoretic mobility shift assay | Jurkat T cells ACC No. 282 | 100 µg/mL | Parthenolide | %I: <30 | The extracts exhibited moderate migration and filling in the damaged areaEE inhibits elastase release, while HeE alters elastase activityEE did not show a cytotoxic effect, while HeE exhibited cytotoxic effect in a Jurkat T cellsHeE has moderate caspase activity | [ref. 109] | |
| p38α assay | — | 100 µg/mL | %I: 30.27 ± 0.67 | ||||||
| Elastase assay | PAF‐stimulated neutrophils | 10, 50, and 100 µg/mL | Resveratrol, GW311616A | Release100 µg/mL: 42.62 ± 1.66Inhibition100 µg/mL: 13.02 ± 0.92 | |||||
| Scratch assay | Mouse fibroblasts | 10 µg/mL | PDGF‐BB | %St: 34.33 ± 2.92 | |||||
| MTT assay | Jurkat T cells | 50 and 100 µg/mL | Parthenolide | %I 100 µg/mL: 7 ± 0.7 | |||||
| HeE of leaves | NF‐κB electrophoretic mobility shift assay | Jurkat T cells ACC No. 282 | 100 µg/mL | Parthenolide | %I: <30 | ||||
| p38α assay | — | 100 µg/mL | %I: 74.14 ± 6.33 | ||||||
| Elastase assay | PAF‐stimulated neutrophils | 10, 50, and 100 µg/mL | Resveratrol, GW311616A | Release100 µg/mL: 39.22 ± 0.77Inhibition100 µg/mL: 59.68 ± 0.60 | |||||
| Scratch assay | Mouse fibroblasts | 10 µg/mL | PDGF‐BB | %St: 28.26 ± 2.41 | |||||
| Caspase‐3‐like assay | Jurkat T cells | 50 µg/mL | Actinomycin D | Relative fluorescence unit of 1.07 | |||||
| MTT assay | Jurkat T cells | 50 and 100 µg/mL | Parthenolide | %I 50 µg/mL: 12 ± 1.4 and %I 100 µg/mL: 61 ± 6.4 | |||||
Abbreviations: aPTT, activated partial thromboplastin; CCB, calcium channel blocking; ClE, chloroform extract; DF, dichloromethane fraction; EE, ethanolic extracts; FD, fraction of DCM; FEA, fraction of ethyl acetate; HeE, hexanic extracts; IL‐6, interleukin‐6; LDH, lactate deshydrogenase; ME, methanolic extracts; NDNS, numerical data not shown; NE, no effect; PBMN, peripheral blood mononuclear cells; PMNLs, polymorphonuclear leukocytes; PTT, prothrombin time; RBCs, red blood cells; ROS, reactive oxygen species; TNF‐α, tumor necrosis factor alpha.
The studies described above provide evidence of antiparasitic, antiviral, and cytotoxic activity against carcinogenic cell lines, as well as hypoglycemic activity and antimelanosis properties in 21 out of the 95 compounds identified in this family. This suggests that the members of this subfamily serve as a reservoir of terpenoid compounds with pharmacological activity.
Alkaloids
Alkaloids possess a wide range of biological activities, including inhibition of malignant cell growth and proliferation, as well as antioxidant, anti‐inflammatory, antiviral, antibacterial, and immunomodulatory effects [ref. 77, ref. 84, ref. 151].
To date, 62 alkaloids have been isolated (Table 4 and Figure 1), including 48 betalains (276–323), 2 guanidine alkaloids (269–270), 2 indole alkaloids (271–272), 2 tricyclic alkaloids (274–275), a pyridine alkaloid (273), and other alkaloids (324–330). Among these, the betacyanins amaranthine (276), isoamaranthine (288), betanin (295), and isobetanin (297) are the most abundant alkaloids found in species of Gomphrenoideae, being reported all in A. bettzickiana, A. brasiliana, A. ficoidea Griseb. (Synm. A. littoralis Beauv. ex Moq.), A. tenella, G. globosa, I. herbstii, and Iresine lindenii Van Houtte.
G. globosa has the highest number of alkaloid compounds, with 33 compounds (276, 278–280, 288–290, 294–313, 316, 318–321, 323).
In 2020, Killian reported the isolation of two new, unusual guanidine alkaloids (269–270) from the ethanol extract of aerial parts of I. diffusa [ref. 165], being the first time that this type of compound was isolated from a Gomphrenoideae species.
On the other hand, aurantiamide (326) has been found to possess antioxidant, anti‐inflammatory, antiviral, antibacterial, and immunomodulating properties [ref. 84]. This compound has been reported in the extract of the whole plant G. celosioides [ref. 83–ref. 85, ref. 89]. The presence of betalains in this subfamily is noteworthy, as these compounds are exclusively produced by plants belonging to the order Caryophyllales and have demonstrated significant biological activity.
Of the 62 identified alkaloids, 16 have been studied for their biological activity. For instance, the antimicrobial activity of compound aurantiamide acetate (327) was evaluated against 19 bacteria and 3 yeasts, showing activity against only 5 bacteria, as detailed in Table S1 [ref. 2]. In 2018, Spórna‐Kucab evaluated extracts, seven fractions (mixtures of alkaloids), and individual alkaloid‐type compounds for antimicrobial activity (bacteria and yeast). The study demonstrated that the isolated compounds exhibited better activity than the fractions, whereas the fractions showed better activity than the extracts. The compounds evaluated included cis‐isomer of gomphrenin II (298), cis‐isomer of gomphrenin III (299), cis‐isomer of isogomphrenin II (300), cis‐isomer of isogomphrenin III (301), gomphrenin II (304), gomphrenin III (305), isogomphrenin II (309), isogomphrenin III (310), isosinapoyl‐gomphrenin I (311), and sinapoyl‐isogomphrenin I (313) [ref. 173]. These findings are intriguing, as they suggest that the combination of these compounds may produce an antagonistic effect on antibacterial activity, which is not always the case. In many instances, fractions and extracts exhibit greater activity than isolated compounds due to compound synergism. This research highlights the importance of studying extracts, fractions, and isolated compounds in the search for substances of biotechnological interest.
Additionally, antiparasitic and antioxidant activities were evaluated for compounds alternamide A (7,8‐dihydroxy‐1,2,4,5‐tetrahydro‐3H‐1,5‐ethano[c]azepin‐3‐one) (274), alternamide B (6,7‐dihydroxy‐3,4‐dihydroquinoline‐1‐one) (275), alternamine A ((R)‐1‐(3,4‐dihydroxyphenyl)‐1,2,3,4‐tetrahydroisoquinoline‐6,7‐diol) (324), and alternamine B (4‐(2‐aminoethyl)benzene‐1,2‐diol‐4‐(2‐aminoethyl)benzene‐1,2‐diol‐b‐d‐glucopyranose) (325). These compounds showed different degrees of activity, as detailed in Tables S1 and S2, with compound 324 exhibiting promising antiparasitic activity [ref. 8].
Regarding anticancer activity, only compounds celosiadine A (269) and celosiadine B (270) have been evaluated in vitro, showing activity against LNCaP but not PC3 cells. Hypoglycemic activity was evaluated in bruceolline F (271), but it showed no activity [ref. 84].
The studies evaluating the bioactivity of alkaloid compounds from this subfamily have confirmed their antimicrobial, antioxidant, anticancer, and hypoglycemic properties, demonstrating the potential of this subfamily as a source of biologically active compounds. However, there is still a vast area for investigation, as there have been limited studies to ascertain the biotechnological potential of the betalains identified in this subfamily.
Betalains
Betalains are classified into betacyanins (red‐violet) and betaxanthins (yellow) based on their structure. In plants, they serve protective functions against pathogens and various environmental conditions, aiding in plant propagation. Once isolated, betalains can be utilized as colorants and exhibit a wide range of biological activities, including antioxidants, anticancer, radioprotective, antilipidemic, antihypoglycemic, anti‐inflammatory, and antimicrobial (antibacterial, antifungal, and antiviral) effects. They are also used in the treatment of hypertension, diabetes, anemia, thalassemia, high cholesterol levels, calcium deficiency disorders, and liver‐related issues. Additionally, betalains demonstrate chemopreventive properties and have positive effects on metabolism, cardiovascular health, and gastrointestinal health in humans. Importantly, betalains are non‐toxic and do not exhibit mutagenic or allergic reactions [ref. 29, ref. 96, ref. 130, ref. 174].
To date, 48 types of betalains have been reported in this subfamily. These include 18 belonging to the amaranthin group (276–293), 16 to the gomphrenin group (298–313), 8 types of betaxanthine (316–323), 4 belonging to the betanin group (294–297), and 2 other betalains (314–315). Deladino’s 2017 study indicates that it was the first report of amaranthine (276), isoamaranthine (288), betanin (295), and isobetanin (297) in Alternanthera species, although these compounds are commonly isolated from Amaranthus species [ref. 28]. However, it is important to note that these compounds were previously reported by Cai in A. bettzickiana and A. ficoidea, making Cai’s study the first to report these compounds [ref. 166].
As mentioned above, G. globosa showed the broadest phytochemical profile in terms of betalains. It is also worth noting that 22 betalains (276, 277, 281–282, 284–289, 291–293, 295, 297, 304–305, 309–312, 314) have been reported in I. herbstii and 17 (276, 278, 281–282, 284–285, 288–289, 291–293, 295, 297, 305, 310, 314–315) have been reported in I. lindenii. Among the compounds isolated in this subfamily, Betanin (295) is known to combat oxidative stress and reduce tumors in the lung, skin, colon, liver, and esophageal in various animal models. It also shows activity against tumors in the prostate, breast, and pancreas in humans and inhibits the proliferation of various human cancer cell lines [ref. 130]. Additionally, gomphrenin (302) has been found to have chemopreventive activity, whereas celosianin (278) and iresin (283) exhibit antioxidant potential [ref. 130].
Spórna‐Kucab in 2020 proposed new tribal names for some betalains. Among the proposed changes was renaming gomphrenina II (304) to globosin, gomphrenina III (305) to basellin (due to its presence in Basella alba), 2″‐OE‐sinapoyl‐amaranthin (289) to lindenin (because of its presence in I. lindenii), sinapoyl‐gomphrenin I (312) to gandolin (due to its presence in Gandola nigra), celosianin I (277) to argentianin, and celosianin II (278) to celosianin [ref. 110].
Compounds of Lipid Nature
Currently, 35 compounds have been reported in members of this subfamily. These include 33 fatty acids (331–363), a fatty alcohol (364), and a fatty amide (365) (Table 5 and Figure 1). A. brasiliana has the largest spectrum of lipid compounds (335, 337–348, 353–363, 365).
Regarding the evaluation of biological activity, only the antimicrobial and antimelanosis activities have been evaluated as described below. Compounds (8E)‐10‐hydroxy‐8‐octadecenoic acid (337), (10E)‐9‐hydroxy‐10‐octadecenoic (338), (8E,12Z)‐10‐hydroxy 8,12‐octadecadienoic acid (339), (9Z,11E)‐13‐hydroxy‐9,11‐octadecadienoic acid (340), (9Z,11E,15Z)‐13‐hydroxy‐9,11,15‐octadecatrienoic acid (341), (9Z,12Z,14E)‐16‐hydroxy‐9,12,14‐octadecatrienoic acid (342), (9Z,13E)‐12‐hydroxy‐9,13‐octadecadienoic acid (343), (9Z,13E,15Z)‐12‐hydroxy‐9,13,15‐octadecatrienoic acid (344), (10E,12E)‐9‐hydroxy‐10,12‐octadecadienoic acid (345), (10E,12Z)‐9‐hydroxy‐10,12‐octadecadienoic acid (346), (9Z)‐9‐octadecenedioic acid (354), (7E)‐9‐oxo‐7‐octadecenoic acid (356), (8E)‐10‐oxo‐8‐octadecenoic acid (357), (9E,11E)‐13‐oxo‐9,11‐octadecadienoic acid (358), (9Z,11E)‐13‐oxo‐9,11‐octadecadienoic acid (359), (10E,12E)‐9‐oxo‐10,12‐octadecadienoic acid (360), and (10E,12Z)‐9‐oxo‐10,12‐octadecadienoic (361) were evaluated against three bacteria, but only nine of them showed activity as detailed in Table S1 [ref. 32].
Of the 35 compounds identified, only 17 have been evaluated for biological activity, highlighting a significant research opportunity.
Other Compounds
A total of 146 other compounds were reported, which consisted of 26 phytosterols (386–412), 22 aliphatic hydrocarbons (419–440), 20 phytoecdysteroids (368–385), 7 carboxylic acids (447–443), 6 alkanes (441–446), 4 saponins (414–417), 4 feruloyl tyramine derivatives (454–457), 4 tocopherols (464–467), 3 hydrocarbons (460–462), 2 phytoecdysones (366–367), 2 vitamins (468–469), 2 heterocyclic compounds (458–459), 1 phytosteroid (413), 1 aliphatic alcohol (418), 1 organic acid (463), and 43 compounds that were not classified (470–512) (Table 6 and Figure 1). With 30 compounds, Gomphrena elegans Mart. had the highest number of secondary metabolites of another type (421–427, 429–434, 436–437, 439–440, 461–462, 475–476, 479–480, 488, 493, 495–498, 508, 511), highlighting that 77.27% of aliphatic hydrocarbons were reported in G. elegans Mart. Additionally, it can be seen from Table 6 that feruloyl tyramine‐type compounds were only reported in A. philoxeroides.
In 1998, Sarker was the first to report the phytoecdysteroid 2‐dehydro‐3‐epi‐20‐hydroxyecdysone (370) in seeds of Froelichia floridana (Nutt.) Moq. [ref. 192]. Another important aspect was that Roriz, in 2014, reported tocopherols (464, 466, 467) for the first time in G. globosa [ref. 97].
The biological activity of these compounds has been studied little. Compounds Δ7‐stigmasterol (411) and 3‐O‐β‐d‐glucopyranosyl Δ7‐stigmasterol (416), and the following mixtures 411 and campesterol (386); spinasterol (396) and 411; 3‐O‐β‐d‐glucopyranosyl stigmasterol (415) and 416; sitosterol glycoside (394) and 3‐O‐β‐d‐glucopyranosyl spinasterol (414), were evaluated against bacteria, yeast, and fungi, showing activity to varying degrees, as detailed in Table S1 [ref. 142]. Likewise, the mixture of compounds sitosteryl (395) and stigmasteryl 3‐β‐O‐glucoside 6′‐O‐palmitate (412) was evaluated for antimicrobial activity against 15 microorganisms but showed activity against 6 (Table S1) [ref. 151]. Compound stigmast‐6‐en‐3‐O‐β‐(d‐glicopyranoside) (417) was evaluated against five microorganisms but only had activity against two of them (Table S1) [ref. 157]. On the other hand, the mixture of compounds 395 and 412 was evaluated against T. cruzi and L. amazonensis and showed activity (Table S1) [ref. 151]. Compound (449) was tested against six bacteria and showed activity against all of them, although the activity was lower than that of the extracts tested (Table S1) [ref. 82].
Additionally, compounds β‐ecdysone (367), 22‐oxo‐20‐hydroxyecdysone (377), pterosterone (381), taxisterone (384), and 2β,3β,14α,17β‐tetrahydroxy‐5β‐androst‐7‐en‐6‐one (385) were evaluated as melanogenesis inhibitors, but none showed activity. Notably, terpenoid compounds were also assessed as melanogenesis inhibitors and demonstrated significant activity (see Table 9) [ref. 161]. Compounds (455), (456), and (457) were evaluated against HeLa cells, and two of them showed significant effects, as evidenced in Table S2 [ref. 36].
Pharmacological Activities
To date, 162 articles have evaluated biological activity, both in vitro (108 articles) and in vivo (73 articles), of 30 species within the Gomphrenoideae subfamily. This research shows that only 6.10% of the members of this subfamily have been studied in terms of biological activity. Furthermore, of the 15 genera, only 6 have been studied. This indicates that the remaining genera, Froelichia, Froelichiella, Hebanthodes, Pedersenia, Pseudoplantago, Quaternella, Tidestromia, and Xerosiphon, remain unexplored in terms of biological activity.
On the other hand, it should be noted that the pharmacological investigations of different extracts and isolated compounds from members of this subfamily confirm various biological activities. These include antioxidant, antimicrobial, anticancer, anti‐inflammatory, antidiabetic, antihyperglycemic, antiarthritic, antihypertensive, analgesic, immunomodulatory, neuroprotective, cardioprotective, gastroprotective, hepatoprotective, diuretic, and wound‐healing properties.
It is important to emphasize that when consolidating and analyzing the biological activity data of the isolated compounds, fractions, extracts, and extract‐based nanoparticles, a notable variability becomes evident. In some cases, isolated compounds exhibit greater activity compared to extracts and fractions, whereas in others, the opposite occurs. This variability can be attributed to synergistic and antagonistic interactions between the compounds, as well as the specific concentrations of each compound present in the extract, which can influence the overall activity.
In this sense, the importance of conducting studies focused on examining extracts, fractions, isolated compounds, and compound mixtures is evident, as this could lead to the discovery of phytotherapeutics that are beneficial for the pharmaceutical industry and the community. Additionally, it is crucial to conduct studies that evaluate the potential of free extracts and drug delivery systems, such as nanoparticles, because the latter have shown that they can enhance the biological activity of the extract in many cases.
Another important observation, which is discussed in the following sections, is that extracts from the same species exhibited different results for the same biological activity. This variability can be attributed to the fact that the study plants were sourced from different geographical locations and times, resulting in distinct phytochemical profiles and consequently varying biological activities. These findings underscore the importance of conducting studies that consider different varieties of the same species, plants collected at different times and locations, and various environmental and stress conditions, as well as the use of in vitro systems and greenhouses to assess the stability of the phytochemical profile and biological activity. Such studies can help determine the optimal conditions for producing bioactive substances of interest.
The significance of the studies that lead to the discovery of new phytotherapeutic compounds lies in their lower cost, greater availability, reduced adverse effects, biodegradability, and environmental safety compared to synthetic drugs [ref. 14, ref. 16, ref. 129, ref. 192].
An overview of the modern pharmacological studies conducted on different extracts and isolated compounds is described in the following subsections, Tables 7, 8, 9, 10, and Schemes 4, 5, 6, 7. In general, the reported studies used standardized and/or similar techniques and concentrations, allowing comparisons between the reported results, thus generating a more comprehensive analysis.
TABLE 7: Anti‐inflammatory activity of the Gomphrenoideae subfamily.
| Species | Extract(s)/Compounds | Assay method | Model | Dose | Positive control | Activity | References | |
|---|---|---|---|---|---|---|---|---|
| Values | Analysis | |||||||
| Alternanthera brasiliana | HaE of leaves | ELISA IL‐6 | RAW 264.7 cells | 10.0, 50.0, and 100.0 µg/mL | Gallic acid and quercetin | ↓ Production of IL‐6 | ↓ The production of IL‐6 and TNF‐α in a dose‐dependent mannerSignificantly ↓ free radical (NO and O2•−)The HaE of leaves exhibited anti‐inflammatory activity | [ref. 193] |
| ELISA TNF‐α | RAW 264.7 cells | 10.0, 50.0, and 100.0 µg/mL | Gallic acid and quercetin | ↓ Production of TNF‐α | ||||
| NO releasing | RAW 264.7 cells | 10.0, 50.0, and 100.0 µg/mL | Gallic acid and quercetin | IC50: 79.2 ± 5.7 µg/mL | ||||
| Inhibition of O2 •− production | RAW 264.7 cells | 10.0, 50.0, and 100.0 µg/mL | Gallic acid and quercetin | IC50: 72.1 ± 6.0 µg/mL | ||||
| Alternanthera sessilis | EE of stems | Cell viability (MTT method) | RAW 264.7 cells | 25, 50, 100, 200, 300, 400, and 500 µg/mL | — | 500 µg/mL ↓ cell viability by 80%↓ NO levels in a dose‐dependent mannerSignificantly suppresses the production of PGE2, IL‐6, IL‐1β, and TNF‐αInhibit the translocation of the NF‐κB subunit p65 to the nucleus↓ The expression of iNOS and COX‐2Prevents phosphorylation of IκBα and, consequently, activation of NF‐κB p65 | [ref. 56] | |
| NO releasing | RAW 264.7 cells | 50, 100, and 200 µg/mL | Dexamethasone | |||||
| ELISA | RAW 264.7 cells | 50, 100, and 200 µg/mL | Dexamethasone | |||||
| Immunocytochemistry | RAW 264.7 cells | 50, 100, and 200 µg/mL | — | |||||
| Protein expression analysis | RAW 264.7 cells | 50, 100, and 200 µg/mL | BSA | |||||
| Gomphrena celosioides | Extract | NO releasing | RAW 264.7 cells | 10 mg/mL | — | %I: 31.1 | Anti‐inflammatory effect | [ref. 195] |
| Determination of COX‐2 | RAW 264.7 cells | Uninformed | Celecoxib | %I: 59.4 | ||||
| Gomphrena celosioides | 367 | NO determination | RAW 264.7 cells | Uninformed | L‐NMMA | NDNS | 512 showed the greatest activity in inhibiting nitric oxide activity | [ref. 178] |
| 512 | IC50: 19.55 ± 0.61 µM | |||||||
| 20‐Hydroxyecdysone‐20,22‐monoacetonide | IC50: 97.35 ± 1.14 µM | |||||||
| Gomphrena globosa | AqE of inflorescences | Cell viability (MTT and LDH assays) | RAW 264.7 cells | Uninformed | Dexamethasone | NDNS | Extracts do not significantly affect cell viabilityAll the extracts decrease nitric oxide levels, observing the highest activity in commercial preparation | [ref. 98] |
| NO determination | NDNS | |||||||
| EB of inflorescences | Cell viability (MTT and LDH assays) | RAW 264.7 cells | Uninformed | Dexamethasone | NDNS | |||
| NO determination | NDNS | |||||||
| Commercial preparation of inflorescences | Cell viability (MTT and LDH assays) | RAW 264.7 cells | Uninformed | Dexamethasone | NDNS | |||
| NO determination | NDNS | |||||||
| Gomphrena globosa var. albiflora (white amaranth) | HE of flowers | NO determination | RAW 264.7 cells | Uninformed | Dexamethasone | EC50 (µg/mL): 198 ± 5 | The HE from flowers of the three Gomphrena species exhibited dose‐dependent activity and has potential as an anti‐inflammatory agent | [ref. 73] |
| Gomphrena haageana K. (red amaranth) | HE of flowers | NO determination | RAW 264.7 cells | Uninformed | Dexamethasone | EC50 (µg/mL): 136 ± 4 | ||
| Gomphrena sp. (pink globe amaranth) | HE of flowers | NO determination | RAW 264.7 cells | Uninformed | Dexamethasone | EC50 (µg/mL): 133 ± 7 | ||
| Pfaffia glomerata | FD of roots | NO determination | BMDM (bone marrow‐derived macrophage) | 250, 25, 2.5, and 0.25 µg/mL | — | NDNS | The highest concentrations evaluated (250 and 25 µg/mL) of the two fractions ↓ NO production. Only the FD of roots at a concentration of 0.25 µg/mL was able to ↓ the NO production of cells stimulated with LPS | [ref. 162] |
| FD of aerial part | NDNS | |||||||
Abbreviations: EE, ethanolic extracts; ELISA, enzyme‐linked immunosorbent assay; FD, fraction of DCM; HaE, hydroalcoholic extract; HE, hydromethanolic extracts; IL‐6, interleukin‐6; LDH, lactate deshydrogenase; NDNS, numerical data not shown; TNF‐α, tumor necrosis factor alpha.
TABLE 8: Antidiabetic and antihyperglycemic activity of the Gomphrenoideae subfamily.
| Species | Extract(s)/Compounds | Assay method | Model | Dose | Positive control | Activity | References | |
|---|---|---|---|---|---|---|---|---|
| Alternanthera paronychioides | ME | CAA assay | HepG2 | Uninformed | — | EE presented the highest antioxidant activity (175.8 ± 1.7 µM QE/g)EE did not show a cytotoxic effect, on the contrary, inhibited 90% of the cytotoxicity induced by HG in HIT‐T15 cellsEE inhibits: HG‐induced ROS production, cell accumulation in sub‐G1, cell apoptosis by up to 14%, activation and activity of caspases 3 and 9, cleavage of PARPEE ↑ ΔΨm and attenuates the ↑ in Bax/ Bcl‐2 ratioEE ← PDX1 translocation in RIN‐m5F cells and ↑ the level of insulin secretion | [ref. 35] | |
| EE | CAA assay | HepG2 | Uninformed | — | ||||
| MTT assay | HIT‐T15 | 10–100 µg/mL | Ferulic acid and quercetin | |||||
| RIN‐m5F | ||||||||
| Staining with PI and DAPI | HIT‐T15 | 20 and 50 µg/mL | Quercetin | |||||
| ΔΨm analysis | HIT‐T15 | 50 µg/mL | Quercetin | |||||
| DCFH‐DA | HIT‐T15 | 20 and 50 µg/mL | Quercetin | |||||
| RIN‐m5F | ||||||||
| Western blot analysis | HIT‐T15 | 20 and 50 µg/mL | Quercetin | |||||
| ELISA | RIN‐m5F | 20 and 50 µg/mL | Quercetin | |||||
| AqE | CAA assay | HepG2 | Uninformed | — | ||||
| Alternanthera philoxeroides | Fraction X of ME of leaves | α‐Glucosidase inhibition assay | — | 20, 40, and 60 µg/mL | Luteolin | %I 60 µg/mL: 58.2 and IC50: 52.41 ± 5.22 | [ref. 141] | |
| Alternanthera sessilis | Green leaf juice | Prevention of Hb glycation | — | 200 µL | — | NE | Potent α‐glucosidase inhibitor | [ref. 195] |
| α‐Amylase inhibition assay | Pancreatic α‑amylase | — | — | NE | ||||
| α‐Glucosidase inhibition assay | Rat intestinal α‑glucosidase | 20 µL | — | IC50: 0.22 ± 0.0 mg/mL acarbose equivalent | ||||
| Alternanthera sessilis | FH of ME of leaves | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 6.31 ± 1.70 mg/mL | Callus fractions exhibited higher antiglucosidase activity than leaf fractionsThe FEA of leaves, and FH, CF, and FEA of callus showed an EC50 lower than acarbose. In contrast, the AF of leaves, and BuF and AF of callus exhibited glucosidase‐stimulating activityThe FEA of leaves and callus have been identifiedAs noncompetitive and competitive α‐glucosidase inhibitors, respectively | [ref. 196] |
| CF of ME of leaves | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 4.89 ± 1.67 mg/mL | |||
| FEA of ME of leaves | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 0.55 mg/mL | |||
| Lineweaver–Burk plot analysis | — | 0, 550, and 825 µg/mL | Km : 2.406 ± 0.070 and Vmax: 0.018 ± 0.001 for 550 µg/mL | |||||
| BuF of ME of leaves | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 2.95 ± 0.31 mg/mL | |||
| AF of ME of leaves | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: Nd | |||
| FH of ME of callus | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 0.67 ± 0.05 mg/mL | |||
| CF of ME of callus | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 0.90 ± 0.11 mg/mL | |||
| FEA of ME of callus | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: 0.25 ± 0.01 mg/mL | |||
| Lineweaver–Burk plot analysis | — | 0, 250, and 375 µg/mL | Km : 3.706 ± 0.283 and Vmax: 0.063 ± 0.009 for 375 µg/mL | |||||
| BuF of ME of callus | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: Nd | |||
| AF of ME of callus | α‐Glucosidase inhibition assay | — | Uninformed | Acarbose | EC50: Nd | |||
| Gomphrena celosioides | 271 | PTP1B inhibition assay | — | 50 mM | Ursolic acid | 40.21% ± 1.69% | [ref. 84] | |
| Measuring glucose uptake | 3T3‐L1 adipocytes | 20 µM | Insulin | % 2‐NBDG absorbance: ≅125 | ||||
| 162 | PTP1B inhibition assay | — | 50 mM | Ursolic acid | 17.89% ± 2.76% | |||
| Measuring glucose uptake | 3T3‐L1 adipocytes | 20 µM | Insulin | % 2‐NBDG absorbance: ≅118 | ||||
| 166 | PTP1B inhibition assay | — | 50 mM | Ursolic acid | 8.10% ± 5.19% | |||
| Measuring glucose uptake | 3T3‐L1 adipocytes | 20 µM | Insulin | % 2‐NBDG absorbance: 100 | ||||
| 187 | PTP1B inhibition assay | — | 50 mM | Ursolic acid | 28.64% ± 8.01% | |||
| Measuring glucose uptake | 3T3‐L1 adipocytes | 20 µM | Insulin | % 2‐NBDG absorbance: ≅122 | ||||
| 190 | PTP1B inhibition assay | — | 50 mM | Ursolic acid | 80.39% ± 6.88% | |||
| Measuring glucose uptake | 3T3‐L1 adipocytes | 20 µM | Insulin | % 2‐NBDG absorbance: ≅138 | ||||
Abbreviations: AF, aqueous fraction; CAA, cellular antioxidant activity; CF, chloroform fraction; DCFH‐DA, 6‐carboxy‐2′,7′‐dichlorodihydrofluorescein diacetate; EE, ethanolic extracts; ELISA, enzyme‐linked immunosorbent assay; FEA, fraction of ethyl acetate; FH, fraction of hexane; ME, methanolic extracts.
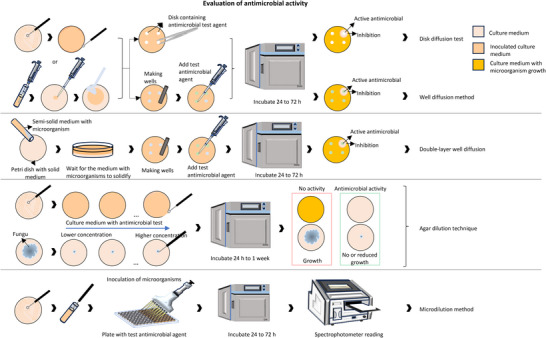
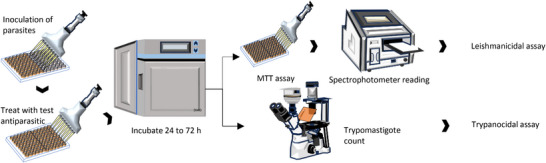
SCHEME 7Anticancer activity assays.
Antimicrobial Activity
Infections caused by fungi and bacteria are responsible for the development of serious diseases and over fifty thousand deaths per year [ref. 96]. Additionally, many microorganisms have developed resistance to existing drugs, posing a risk to public health and presenting a challenge for the pharmaceutical and healthcare industries, with economic implications [ref. 11, ref. 14, ref. 89, ref. 129, ref. 223].
In addition to the development of drug resistance by microorganisms, developing and underdeveloped countries lack access to medications to treat infections, leading to increased mortality rates. Therefore, the search for new broad‐spectrum antimicrobials with low toxicity derived from natural sources is necessary [ref. 14, ref. 89, ref. 223].
To date, 46 articles have evaluated the antimicrobial activity of members of 5 genera of this subfamily. Twenty‐seven articles report the presence or absence of antibacterial activity, evaluating a total of 98 bacterial strains; 14 articles discuss antifungal potential, evaluating 19 yeast strains and 8 fungal strains; 6 articles report on antiparasitic activity against T. cruzi and L. amazonensis; and 2 articles discuss antiviral potential of A. philoxeroides, evaluating its activity against 13 viruses. These studies examined the potential of approximately 46 extracts, 16 fractions, 6 mixtures of compounds, and 73 isolated compounds (6, 7, 9, 10, 17, 20, 24, 25, 33, 34, 39, 40, 43, 50, 51, 54, 58, 60, 70, 71, 72, 80, 88, 92, 93, 94, 95, 100, 101, 106, 107, 108, 109, 156, 159, 226, 238, 274, 275, 298, 299, 300, 301, 304, 305, 309, 310, 311, 313, 324, 325, 327, 337, 338, 339, 340, 341, 342, 343, 344, 345, 346, 354, 356, 357, 358, 359, 360, 361, 411, 416, 417, 449). These studies cover only 3.54% of the Gomphrenoideae members (A. bettzickiana, A. brasiliana, A. caracasana HBK, A. dentata, A. littoralis P. Beauv., A. philoxeroides, A. pungens, A. repens, A. sessilis (Linn.), A. tenella Colla, B. portulacoides, G. agrestis, G. boliviana, G. celosioides, G. globosa, G. martiana, H. eriantha, I. herbstii, P. glomerata, and P. paniculata). Schemes 4 and 5 summarize the main methodologies, and Table S1 presents the results obtained.
Kumar and his research group are currently the only ones who have studied the antibacterial activity of Alternanthera dentata, demonstrating that AgNPs of AqE of leaves are active against the four microorganisms evaluated [ref. 225]. Research by Zavala and collaborators showed that the ME of A. repens was not active against bacteria and yeasts [ref. 216].
Bhattacherjee and his research group are currently the only ones who have studied the antibacterial activity of A. philoxeroides. They demonstrated that fraction X of leaf ME exhibits remarkable activity, as it was able to inhibit Escherichia coli and Micrococcus luteus with relatively small MICs (11.23 ± 0.11 and 16.23 ± 0.23 µg/mL, respectively) and large ZI, when compared with the results obtained for the AuNPs of AqE from A. bettzickiana, the subfractions of FEaMc and extracts of A. brasiliana, ME of A. sessilis, EE and PEE from G. boliviana, extracts from G. celosioides, AcE from G. globosa, compounds (20, 24, 25, 34, 43, 72, 88, 410, 411), and mixtures of compounds (411 and 386), (396 and 411), (415 and 416), (394 and 414) [ref. 26, ref. 27, ref. 32, ref. 75, ref. 82, ref. 89, ref. 142, ref. 141, ref. 173, ref. 226].
A. sessilis was studied by four research groups, three of which used nanoparticles. Niraimathi et al. and Kabeerdass et al. demonstrated that the AgNPs of AqE of leaves are active against bacteria, especially gram‐negative strains [ref. 60, ref. 227]. Venkatraman and collaborators showed that the ZnONPs of leaves possess antibacterial activity and suggested that the mechanism of action involves membrane destruction, leading to metabolic dysfunction and protein excretion by the bacteria [ref. 228]. Ullah and collaborators reported similar results, demonstrating that the ME of the whole plant presented activity against 10 of the 12 microorganisms evaluated [ref. 226].
G. celosioides was studied by five research groups. Moura’s results for E. coli differed from those obtained by some authors; he reported that the EE of aerial parts and compounds 156 and 417 did not show activity against this strain. However, Dosumu and Omokhua‐Uyi reported that this strain was sensitive to AcE, EaE, and compound 449 [ref. 82, ref. 89, ref. 151]. Another interesting discovery was made by Dosumu, who concludes that the higher activity observed in EaE was due to synergistic relationships between molecules [ref. 82]. However, this information cannot be extrapolated, because extracts sometimes present less activity due to antagonistic relationships between molecules, as observed in the study by Sporna‐Kucab in G. globosa [ref. 173]. Additionally, Dosumu also reported that ME has antifungal activity similar to that of tioconazole [ref. 82]. This finding is particularly relevant because there are currently very few antifungal agents available on the market, highlighting the need for new broad‐spectrum antifungals. In this context, Abalaka et al. also obtained interesting results, demonstrating that AuNPs of leaf extract exhibited activity similar to CAM and streptomycin [ref. 229].
Rahamouz Haghighi and Sharafi in 2024 published the first article of the antibacterial activity of H. eriantha, demonstrating that it was active against the five microorganisms evaluated, having the same effect as gentamicin on S. aureus [ref. 230].
Dipangar and Murugan obtained results similar to Bhattacherjee with AgNPs of AqE from leaves of I. herbstii, which inhibited three gram‐negative bacteria and two gram‐positive with MICs between 6.25 and 50 µg/mL [ref. 231].
A. brasiliana was studied by six different researchers, two of whom studied both antibacterial and antifungal activity. In summary, Johann, Andreaza, and Coutinho reported that the HeE and EE of the whole plant, the HaE of aerial parts, and the EE of leaves did not show relevant activity against the tested microorganisms [ref. 23, ref. 26, ref. 232]. Araújo obtained similar results when evaluating AqE of leaves, showing that the extract did not show activity against four microorganisms, had clinically irrelevant activity against four microorganisms, and showed activity only against Mycobacterium smegmatis (MIC: 15.6 µg/mL) and C. albicans (MIC: 31.2 µg/mL) [ref. 27]. These results are consistent with those obtained by Marchete in 2021, who indicated that the HaE of the leaves has weak activity against P. aeruginosa, S. aureus ATCC 25923, oxacillin‐resistant S. aureus, and E. coli ATCC 25922 [ref. 193]. In contrast, Trapp reported that the FEaMc (1:1) and three sub‐fractions showed activity against B. subtilis and M. luteus, and that compounds 340, 341, 342, 344, 346, 354, 358, 359, and 361 showed activity against B. subtilis, M. luteus, E. coli, and P. aeruginosa [ref. 32]. The difference in results could be attributed to the extraction methods used.
Dominguez et al. in 2022 published the only article that discusses the effect of glycolic extract of the roots of P. paniculata on mixed‐species biofilms of C. albicans and S. mutans, S. aureus, or P. aeruginosa. Their main results reported biofilm inhibition to varying degrees [ref. 233].
Another important discovery was made by the research groups of Nagalingman and Andreazza, who concluded that the effect of the extract on microorganisms can be potentiated when used with drug delivery systems such as nanoparticles or in combination with other methods such as photosensitization [ref. 21, ref. 23].
To date, the trypanocidal and leishmanicidal effects have been evaluated in A. littoralis Beauv. ex Moq., B. portulacoides, G. agrestis, and P. glomerata, all of which showed antiparasitic activity [ref. 2, ref. 8, ref. 117, ref. 151, ref. 177, ref. 234]. The greatest activity was observed in compound 324 isolated from A. littoralis, with IC50 values of 0.23 and 0.16 mM for T. cruzi and L. amazonensis, respectively. Additionally, it should be noted that compounds 226 and FH from the HaE of P. glomerata showed high activity against T. cruzi, with IC50 values of 44.78 and 47.86 µg/mL, respectively [ref. 8, ref. 177].
Antiviral activity has only been evaluated in vitro and in vivo for A. philoxeroides, and the results were promising, as that compound 238 showed in vitro activity against HSV‐1, HSV‐2, human cytomegalovirus, measles virus, and mumps virus. In a genital herpes model in mice (in vivo) caused by HSV‐2, compound 238 was effective, indicating that this compound could be a candidate for an anti‐herpes agent. Additionally, compounds 59, 40, and 60 significantly blocked the secretion of HBsAg in HepG2.2.15 cells [ref. 40, ref. 41].
Antioxidant Activity
Antioxidant compounds play a crucial role in protecting the human body against free radicals, including reactive oxygen species (ROS), which can lead to various pathological conditions. These conditions include Alzheimer’s disease, anemia, arthritis, asthma, atherosclerosis, cancer, cataracts, liver cirrhosis, diabetes, neurological disorders, Parkinson’s disease, cardiovascular diseases, hypertension, hypotension, ischemia, inflammation, Down syndrome, neurodegeneration, and the aging process. Therefore, antioxidant compounds are important in the treatment and prevention of these diseases [ref. 14, ref. 77, ref. 151, ref. 224].
In recent years, there has been a growing interest in the antioxidant potential of plants. This interest persists despite the availability of synthetic drugs on the market, as concerns about their safety and toxicity continue to be significant.
To date, 32 articles have discussed the antioxidant potential of the Gomphrenoideae subfamily members, with 2 studies conducted using in vivo models. These studies described the antioxidant potential of 3.54% of the subfamily members (A. bettzickiana, A. brasiliana, Alternanthera flavescens, A. littoralis P. Beauv., A. paronychioides, A. philoxeroides, A. pungens, A. sessilis (Linn.), A. tenella Colla, G. celosioides, G. globosa, Gomphrena haageana K, I. angustifolia, I. herbstii, P. glomerata, P. paniculata, P. townsendii), evaluating 47 extracts, 22 fractions, and 14 compounds (24, 25, 34, 43, 50, 67, 70, 88, 106, 129, 274–275, 324, 325). Additionally, two extracts contained in nanoparticles, a green leaf juice, a commercial preparation, and a flower infusion were assessed. Scheme 6 summarizes the main methodologies, and Table S2 summarizes the results obtained.
A. brasiliana was evaluated by five different research groups. Pereira et al. evaluated the antioxidant potential of EE, BuF, DF, and FEA using the DPPH assay, but only FEA exhibited radical scavenging activity, which was dose‐dependent [ref. 235]. Marchete et al. showed that the HaE of leaves has antioxidant activity, evidenced by an increase in the scavenging capacity of DPPH, FRAP, and ABTS free radicals [ref. 193]. Paliwal et al. obtained similar results to Marchete et al., showing that HaE of leaves had higher activity than ClE and ME of leaves [ref. 236].
The antioxidant potential of A. philoxeroides was evaluated by five different research groups. In general, fraction X of the ME of leaves showed better activity against DPPH and ABTS radicals compared to the HdE of the tender stem, shoots, and leaves, as well as the EE of the whole plant. The IC50 values were 33.94 ± 3.45 µg/mL for DPPH and 60.76 ± 4.31 µg/mL for ABTS [ref. 132, ref. 135, ref. 136, ref. 141, ref. 237].
Two research groups demonstrated that the antioxidant potential of A. pungens was low [ref. 16, ref. 238].
The antioxidant potential of A. sessilis was evaluated by eight different authors. The best results for DPPH activity were obtained with the FEA of the ME of leaves (EC50: 10.81 ± 0.29 µg/mL), followed by the CF of the ME of callus (EC50: 34.12 ± 0.67 µg/mL), ME of aerial parts (IC50: 35.39 µg/mL), BuF of the ME of leaves (EC50: 35.71 ± 1.24 µg/mL), AF of the ME leaves (EC50: 35.96 ± 1.28 µg/mL), FEA of the ME of callus (EC50: 43.87 ± 0.39 µg/mL), and BuF of the ME of callus (EC50: 57.11 ± 0.13 µg/mL). The other extracts evaluated showed IC5O, EC50, or SC50 values greater than 80 µg/mL [ref. 49, ref. 55, ref. 57, ref. 60, ref. 195, ref. 196, ref. 238]. Additionally, Mohd Hazli and Muniandy evaluated the EE of the stem of A. sessilis, obtaining IC50 values of >1000 µg/mL and 782 ± 29.9 µg/mL, respectively [ref. 55, ref. 57]. Despite the similarity in the results, discrepancies were observed that could be due to differences in the growth conditions of each plant.
The ethanolic extract of G. celosioides has been studied in vitro and in vivo. According to the in vitro study, the EE of leaves eliminates the DPPH radicals more efficiently than Trolox. Additionally, in the in vivo rat model, it is capable of reducing TBARS levels and increasing the total antioxidant ability in serum [ref. 77, ref. 194, ref. 239].
The antioxidant potential of G. globosa has been studied by four research groups. The extracts, commercial preparations, and floral infusions demonstrated antioxidant activity in various assays, including DPPH, FRAP, TBARS, and radical scavenging activities involving O2 − and NO. However, they exhibited high values in EC50 and IC50, in most cases exceeding 500 µg/mL [ref. 73, ref. 98, ref. 150, ref. 153].
Dipankar and Murugan, in their study, evaluated the antioxidant potential of EE of I. herbstii and AgNPs synthesized using the AqE of the leaves, concluding that the NPs potentially enhance the antioxidant activity of the extract [ref. 231].
Regarding H. eriantha (Poir.) Pedersen, it has been reported to have low antioxidant potential [ref. 215]. However, in that publication, the plant is cited as P. paniculata, which is a synonym of H. eriantha according to GBIF (https://www.gbif.org/es/species/101306355).
In 2018, Corrêa et al. conducted an analysis on various extracts and compounds derived from the whole plant of P. townsendii Pedersen. The study encompassed hexanoic and ethanolic extracts, as well as the hexane, dichloromethane, and hydroalcoholic phases. Additionally, the research included an examination of compounds 67 and 106, both individually and in combination. The results demonstrated that the mixture of compounds 67 and 106 exhibited the highest DPPH radical scavenging activity, with an EC50 value of 3.7 µg/mL. Individually, compound 67 showed significant activity with an EC50 of 4.9 µg/mL, whereas compound 106 alone had an EC50 of 83.2 ± 2 µg/mL. Remarkably, when compound 106 was combined with compound 67, there was a 95.55% reduction in the EC50 value, indicating an enhanced activity likely stemming from a synergistic interaction between the two compounds. Notably, the HeE and the hexane phase both displayed the lowest DPPH activity, with EC50 values exceeding 200 µg/mL [ref. 127].
Anticancer Activity
Cancer is one of the main causes of morbidity and mortality worldwide, characterized by irregular cell growth triggered by genetic or environment stimuli. Currently, chemotherapy remains a primary treatment option; however, it often leads to adverse effects, including the development of cancer cell lines resistant to multiple medications, leading to chemotherapy failure. Therefore, one strategy to combat cancer is the search for bioactive compounds with antiproliferative and antitumoral activities [ref. 131, ref. 240].
Currently, 33 articles discuss the in vivo and in vitro anticancer potential of 28 extracts, 16 fractions, 5 NPs, and 22 isolated compounds (35, 36, 243, 244, 245, 246, 247, 255, 256, 257, 258, 259, 260, 261, 262, 263, 264, 269, 270, 455, 456, 457), 3 mixtures of active molecules, and a paste of leaves and plant powder obtained from 3.13% of the members of Gomphrenoideae (A. bettzickiana, A. brasiliana, A. flavescens, A. philoxeroides, A. sessilis, A. tenella, G. celosioides, G. elegans mart., G. globosa, G. macrocephala, G. martiana, H. erianthos (Synm. Hydrangea paniculata, Synm. P. paniculata), I. diffusa, I. herbstii). Ten articles discuss their activity in vivo models, and 26 discuss their activity in vitro models. Scheme 7 summarizes the main methodologies, and Table 10 and Table S3 summarize the results obtained.
In vitro activity was evaluated across a range of cancer cell lines. These included four leukemia cell lines (HL60, MT‐1, MT‐2, MK‐1); four colon cancer cell lines (Caco‐2, HT‐29, HCT‐8, HCT116); three pancreatic cancer cell lines (Panc‐1, MIA PaCa‐2, Capan‐1); two prostate cancer cell lines (PC3, Human LNCaP); two breast cancer cell lines (MCF‐7 cell, MDA‐MB‐435); two cervical cancer lines (HeLa, KB); two cholangiocarcinoma cells (KKU‐100, KKU‐213); two skin cancer cell lines (B16F10, B‐16); one line of lung cancer (A549); one Ehrlich ascites carcinoma cell line (EAC cell); one liver cancer cell line (Hep‐G2); one human neuroblastoma cell line (SK‐N‐SH); a cell line glioblastoma (SF‐295); and one oral cavity carcinoma cell line (HSC‐2 cells).
The ME of aerial parts of A. brasiliana and A. flavescens Kunth. were evaluated against Caco‐2 and HT‐29 cell lines. In both cell lines, the extract of A. brasiliana was more effective, with the lowest IC50 values: 252.9 ± 5.7 µg/mL for Caco‐2 and 160.3 ± 8.5 µg/mL for HT‐29 [ref. 31]. Regarding HCT‐8, it was evaluated only in G. elegans, and it was found that HeE from leaves caused the highest percentage of lethality (101.16%), followed by FnH (%L: 100), AqE, and FnB from leaves with 99.68% lethality, and ClE with %L of 79.08%. The other 12 extracts and 8 fractions showed a lethality percentage lower than 30% [ref. 156, ref. 241]. The HCT11‐6 cell line was evaluated only with the ME of roots of H. paniculata, which generated a decline in cell viability [ref. 230]. These findings suggest that these four plants exhibit cytotoxic activity against colon cancer cell lines.
The ME of the aerial parts of A. brasiliana and A. flavescens, and the pfaffosidic fraction of H. paniculata (Syn. H. eriantha (Poir.) Pedersen), were evaluated against the Hep‐G2 cell line. It was shown that both the extracts and the fraction exhibited activity against this cell line, with the pfaffosidic fraction presenting the highest activity, reducing more than 50% of the cells at a concentration of 100 µg/mL [ref. 31, ref. 121].
Five compounds isolated from A. philoxeroides, AuNPs from the AqE of A. sessilis leaves, the EE from the whole A. sessilis plant, AgNPs of the AqE of I. herbstii leaves, and the ME of the whole G. globosa plant were evaluated against HeLa cells. The results indicated that compounds 455, 456, 457, and 35 isolated from A. philoxeroides exhibited the highest activity against this cell line, followed by AgNPs of I. herbstii. On the other hand, the EE of A. sessilis showed low activity, and G. globosa showed no activity [ref. 37, ref. 62, ref. 231, ref. 242].
The ME of the whole G. globosa plant was evaluated against leukemia cell lines MT‐1, MT‐2, and MK‐1, and the isolated compounds from A. philoxeroides (261–264) were tested against HL60 cell line. The results showed that G. globosa exhibited no cytotoxic activity, whereas all the compounds from A. philoxeroides were active against the HL60 cell line. Compound 264 presented the highest activity with an IC50 value of 45.93 µg/mL, whereas compound 263 had the lowest activity, with an IC50 of 271.45 µg/mL [ref. 37, ref. 243].
Only one study has evaluated the potential of this subfamily against pancreatic cancer cell lines, obtaining remarkable results. Overall, the ME, PEF, and CF of A. sessilis leaves at a concentration of 100 µg/mL reduced survival percentages to less than 2% in Panc‐1 cells. Among these, CF presented the highest activity, and the authors also reported that it obtained IC50 values of 13.08 ± 10.40 µg/mL for MIA PaCa‐2 and 34.92 ± 2.20 µg/mL for Capan‐1 [ref. 244].
The isolated compounds from I. diffuse (269 and 270) were tested against the PC3 and human LNCaP prostate cancer cell lines, and the AgNPs from AqE of the leaves of A. sessilis were tested against the PC3 line. It was found that compounds 269 and 270 showed activity against LNCaP cells. In contrast, the AgNPs of A. sessilis showed remarkable activity inhibiting 94.11% of PC3 cells at 25 µg/mL [ref. 165, ref. 245].
Currently, seven studies have been conducted to find compounds with cytotoxic activity against breast cancer cell lines. In summary, it was found that MCF‐7 cells are sensitive to the AgNPs (IC50: 3.043 µL/mL and 99% of inhibition at 25 µg/mL) and ZnONPs (IC50: 210 µg/mL) of A. sessilis, but not to the AuNPs of this same plant. In this cell line, the AgNPs of A. tenella also showed activity with an IC50 of 42.5 µg/mL. Additionally, the BE of P. paniculata showed cytotoxic activity [ref. 70, ref. 124, ref. 184, ref. 228, ref. 246]. Regarding the MDA‐MB‐435 cell line, different extracts and fractions of G. elegans Mart. were evaluated. It was shown that AqE and FnB at a concentration of 100 µg/mL generated a lethality percentage of 97.39, whereas HeE and FnH reached lethality percentages of 96.35. However, the other 13 extracts and 8 fractions at this same concentration obtained lethality percentages lower than 50% [ref. 155, ref. 241].
Two studies have evaluated the cytotoxic potential against skin cancer cell lines. In this context, the compounds 255, 256, 257, 258, 259, and 260, isolated from P. paniculata, were tested against the B‐16 cell line. Compound 260 presented the highest activity with an IC50 of 30 µg/mL, and the compound with the lowest activity was 259, with an IC50 of 120 µg/mL [ref. 125, ref. 164]. In contrast, the ME of the whole plant of G. globosa was inactive against B16F10 [ref. 247].
A total of 6 of the 10 in vivo studies aimed to find a bioactive substance for treating Ehrlich carcinoma. Remarkably, the EaE of the leaves of A. brasiliana, the AqE of the aerial parts of A. tenella Colla, the BuF from P. paniculata root, and 20 and 60 mg/kg of the mixture of flavonoids (6, 7, 10, 17) isolated from G. martiana increased the survival of mice with EAC. However, only the extract of A. brasiliana and A. tenella reduced viable cells. Additionally, the mixture of flavonoids inhibited tumor formation by 32%, and the root powder of P. paniculata and the EaE of A. brasiliana reduced the volume of EAC [ref. 66, ref. 100, ref. 185, ref. 207]. It should also be noted that Pinello et al. [ref. 210] evaluated the anticancer potential of the ME of P. paniculata root, focusing on macrophage activity. They found that the ME increased peritoneal macrophages and phagocytosis, suggesting that the anticancer activity of P. paniculata may result from the stimulation of macrophages, natural killer cells, and cytotoxic T lymphocytes [ref. 210]. In conclusion, the extracts of A. brasiliana and A. tenella have antitumor activity, the mixture of compounds has moderate cytotoxic activity, and the root powder and BuF of P. paniculata have an antineoplastic effect.
Da Silva et al. [ref. 208, ref. 211] demonstrated that P. paniculata root powder has anti‐hepatocarcinogenic properties, reducing liver lesions and adenoma in mice. The chemopreventive effect is attributed to the inhibition of cell proliferation and an increase in apoptotic processes [ref. 208, ref. 211]. However, A. sessilis exhibited no activity against squamous cell adenocarcinoma of the stomach in mice [ref. 206].
Analgesic and Antinociceptive Activity
Pain, a sensory perception, often represents the primary symptom in the diagnosis of various diseases. Considered a global public health issue, the search for treatments to alleviate or control pain is crucial. Among these treatments, the use of medicinal herbs and their compounds stands out [ref. 190, ref. 191, ref. 248, ref. 249].
It should be noted that many medications commonly used to treat pain have unwanted side effects, including respiratory depression, drowsiness, decreased gastrointestinal motility, nausea, gastric ulcers, hepatotoxicity, and various disorders of the autonomic nervous and endocrine systems. Furthermore, many of them do not reduce pain in all treated individuals [ref. 191, ref. 248, ref. 249]. This highlights the need to search for new bioactive compounds with analgesic activity that lacks side effects.
Currently, 11 articles discuss the analgesic potential of eleven extracts and 2 isolated compounds from 7 members of the Gomphrenoideae subfamily. The results are described in Table 10. In the acetic acid‐induced abdominal contractions assay, extracts from A. brasiliana, A. philoxeroides, A. sessilis, G. celosioides, and P. glomerata all showed a reduction in the number of contractions. Notably, the AqE of A. brasiliana and the ME of A. sessilis exhibited better activity than dipyrone and aspirin, respectively [ref. 30, ref. 39, ref. 87, ref. 118, ref. 133, ref. 204, ref. 205]. In the carrageenan‐induced paw edema assay, the EE of A. maritima (Mart.) A.St.‐Hil. (Synm. A. littoralis Beauv. ex Moq.), A. tenella, B. portulacoides, G. celosioides, and compounds 43 and 72 showed inhibition of mechanical hyperalgesia induced by carrageenan. Compound 43 achieved 100% inhibition of hyperalgesia [ref. 34, ref. 67, ref. 71, ref. 78].
In the hot plate test, the EE of the whole plant of A. sessilis and the AqE of G. celosioides increased the reaction time, indicating an analgesic effect on the central nervous system. However, the HaE of the roots and rhizomes of P. glomerata showed no effect, indicating the absence of analgesic activity [ref. 87, ref. 118, ref. 133].
In the carrageenan‐induced cold allodynia test, the EE of the whole plant of A. tenella Colla and the EE of the aerial parts of G. celosioides inhibited the response to cold, with the EE of the A. tenella showing the highest activity. However, compound 43 did not show any effect in this test. These same extracts and compound 43 were evaluated in the zymosan‐induced articular inflammation assay, where all reduced mechanical hyperalgesia and inhibited edema [ref. 67, ref. 78].
Anti‐Inflammatory Activity
Inflammatory diseases, such as asthma, rheumatoid arthritis, psoriasis, autoimmune diseases, and severe autoinflammatory diseases, develop due to the overproduction of pro‐inflammatory mediators. For this reason, their inhibition has therapeutic value in the development of anti‐inflammatory agents [ref. 73, ref. 250].
Over time, GCs have been used to treat various inflammatory disorders characterized by their effectiveness, but their chronic use causes undesirable adverse effects, such as skin atrophy, inhibition of wound healing, osteoporosis, obesity, hyperglycemia, and glaucoma [ref. 71, ref. 250].
It should be noted that the anti‐inflammatory compounds caffeic acid, ferulic acid, vanillic acid, and catechin have been isolated from G. celosioides [ref. 78].
To date, 21 studies have been carried out to evaluate the anti‐inflammatory activity of extracts, fractions, infusions, isolated compounds, and commercial preparations of 2.30% of the members of the Gomphrenoideae subfamily, with 7 studies conducted using in vitro models and fifteen using in vivo models. Tables 7 and 10 provide detailed information on these studies.
Regarding in vitro activity, extracts of A. sessilis, G. celosioides, G. globosa, and Gomphrena haageana Klotzsch have been shown to reduce NO levels [ref. 56, ref. 73, ref. 98, ref. 194]. Additionally, the extract of G. celosioides and A. sessilis reduces COX‐2 expression levels [ref. 56, ref. 194]. The EE of A. sessilis also reduces the viability of RAW 264.7 cells, proinflammatory cytokines, and PGE2, as well as prevents the activation of the NF‐κB pathway. However, none of the extracts of G. celosioides affect RAW 264.7 cell viability [ref. 194].
The in vivo anti‐inflammatory activity was evaluated in most of the studies through edema or pleuritis induced by carrageenan. The results showed that the extracts of A. maritima [ref. 34], A. tenella Colla [ref. 213], B. portulacoides [ref. 71], G. celosioides [ref. 77, ref. 87], P. glomerata [ref. 118, ref. 119], and P. townsendii [ref. 127], as well as compound 72 isolated from A. maritima [ref. 34], and compounds 67 and 106 isolated from P. townsendii [ref. 127], exhibited inhibition of edema formation. The best activity was observed in compounds 67 and 106, which at a concentration of 1 mg/kg, were able to inhibit edema by 75.4% ± 4.0% and 73.00% ± 4.0%, respectively [ref. 127]. This was followed by the activity of the HaE of P. glomerata, which showed an ID50 of 20.4 mg/kg for the intraperitoneal dose and an ID50 of 60.5 mg/kg for the oral dose [ref. 119]. The least efficient extract was that of G. celosioides, which, at a concentration of 400 mg/kg, only achieved a 39.62% inhibition of edema [ref. 87].
Regarding the anti‐inflammatory activity in the carrageenan‐induced pleuritis model, it was observed that the extracts of A. tenella Colla [ref. 67], B. portulacoides [ref. 71], P. townsendii [ref. 127], compound 72 isolated from A. maritima [ref. 34], and compounds 67 and 106 isolated from P. townsendii [ref. 127] reduced leukocyte migration to the pleura. Compound 72 showed the greatest inhibition at a concentration of 10 mg/kg (%I: 77) [ref. 34]. Compounds 67 and 106 also showed high activity, with an inhibition of 50.7% ± 1.03% and 59.4% ± 1.25%, respectively, at a concentration of 1 mg/kg [ref. 127]. In this test, it was also observed that compound 72 and the extract of B. portulacoides reduced protein extravasation [ref. 34, ref. 71]. The EE of A. maritima [ref. 34] and A. tenella Colla [ref. 67], as well as compound 43 [ref. 67], reduced the number of leukocytes, with the extract of A. maritima showing the best activity (%I: 68) [ref. 34, ref. 67]. Additionally, the AqE of A. brasiliana reduced the number of lymphocytes, polymorphonuclear cells, and exudate [ref. 30]. These results suggest that both the extracts and the isolated compounds have anti‐inflammatory activity.
It should be noted that the EE of leaves from A. brasiliana also reduced the formalin‐induced edematogenic process [ref. 26]. The EE of the whole plant of A. tenella Colla and compound 43 inhibited the formation of zymosan‐induced edema [ref. 67]. The EE of B. portulacoides inhibited Bothrops jararacussu venom, BthTX‐I, and BthTX‐II‐induced edema formation but did not affect leukocyte migration [ref. 214]. HaE from roots of P. glomerata reduced edema induced by bradykinin, substance P, histamine, serotonin, and LPS [ref. 119], and the ME of P. paniculata roots reduced colonic and intestinal inflammation, the latter being related to the modulation of the expression and production of MAPKs and mucin [ref. 120, ref. 215].
Antidiabetic Activity and Antihyperglycemic Activity
Diabetes is a disease that is spreading rapidly throughout the world, and every year the number of people suffering from this disease increases. In 2016, the WHO reported that 400 million people were affected by diabetic disorder, and in 2017, the IDF reported that 425 million people had diabetes mellitus. It is estimated that by 2045, the number of people with diabetes will rise to 629 million. Additionally, diabetes can lead to the development of other diseases, including cardiovascular, kidney, and eye diseases, as well as stroke and lower limb amputation [ref. 15, ref. 204].
Currently, diabetes can be treated with insulin or hypoglycemic agents, but both can cause side effects such as hypoglycemia, weight gain, gastrointestinal upset, nausea, diarrhea, liver dysfunction, jaundice, and heart failure [ref. 15].
It should be noted that around 400 plant species and their metabolites are used to treat diabetes mellitus worldwide. The antioxidant potential of medicinal plants is a key factor in reducing the incidence of diabetic complications [ref. 15].
Currently, eight studies have evaluated the antidiabetic and/or antihyperglycemic potential of five extracts, fourteen fractions, five compounds, and a green juice, representing 0.83% of the members of the Gomphrenoideae subfamily (Tables 8 and 10).
In the in vitro studies, the inhibition of α‐glucosidase was evaluated for fraction X of A. philoxeroides, the green leaf juice of A. sessilis, and eight fractions of the latter plant. The results demonstrated that all the evaluated substances had α‐glucosidase inhibitory activity. The most potent activity was observed in fraction X of A. philoxeroides (IC50: 52.41 ± 5.22 µg/mL), whereas the weakest activity was found in the FH of the ME of A. sessilis leaves (EC50: 6.31 ± 1.70 mg/mL) [ref. 141, ref. 195, ref. 196]. It was also described that A. paronychioides has a preventive action on diabetic glucotoxicity [ref. 35], and among the five compounds isolated from G. celosioides (162, 166, 187, 190, 271), compound 190 showed a significant improvement in glucose uptake and the highest inhibitory effect on PTP1B, indicating its potential for use in the prevention and treatment of Type 2 diabetes [ref. 84].
As for the in vivo studies, they were generally analyzed using the OGTT. Notably, the ME of the entire A. philoxeroides plant [ref. 39], the ME of aerial parts of A. sessilis [ref. 204], and the FEA of the EE from the aerial parts of A. sessilis [ref. 212] reduced glucose levels in a manner comparable to GLB. It should also be noted that neither the green juice nor HF and FA of A. sessilis reduced glucose levels [ref. 195, ref. 212].
Hepatoprotective Activity
The liver is responsible for regulating several important metabolic functions, and injury to this organ can disrupt these functions [ref. 251]. Liver damage typically involves oxidative stress and is characterized by a progressive evolution from steatosis to chronic hepatitis, fibrosis, and cirrhosis [ref. 252].
Currently, liver diseases are treated with corticosteroids, vaccines, and antivirals. However, these treatments often cause side effects, particularly with chronic or subchronic use. This highlights the necessity of finding new phytotherapeutic drugs that are both safe and more effective [ref. 252].
To date, only four in vivo studies on the hepatoprotective activity of members of this subfamily have been conducted (Table 10). The HaE from the leaves of A. brasiliana L. [ref. 236], the ME from the entire A. sessilis plant [ref. 51], the AqE from the stems and leaves of G. celosioides [ref. 90], and AqE from G. globosa L. [ref. 218]. All studies showed an improvement in the antioxidant profile and a reduction in liver damage [ref. 51, ref. 90].
Other Activities
Other activities have been studied in members of the Gomphrenoideae subfamily, as shown in Tables 9 and 10. An analysis of some of these activities will be presented below.
Neurological Activity
To date, 14 investigations have been carried out to evaluate the potential of fourteen extracts, 10 fractions, 6 compounds, and an infusion of aerial parts of 7 members of this subfamily. Among the isolated compounds of A. philoxeroides (5, 35, 36, 41, 79), compound 35 exhibited the highest inhibitory activity against MAO‐A (IC50: 0.00046 ± 0.04 µM), higher than that of Clorgyline. Compound 36 showed the highest inhibitory activity against MAO‐B (IC50: 0.00022 ± 0.12 µM) and inhibited the formation of toxic Aβ plaques (%I: 81.96 ± 2.14), exhibiting greater activity than curcumin in the latter. It should be noted that all these compounds demonstrated greater inhibition of MAO‐A and MAO‐B than the EE of this plant. These results are significant because monoamine oxidases are enzymes related to cognitive dysfunction and depression. Therefore, it can be suggested that these flavonoids have antidepressant activity. Furthermore, these five flavonoids play an important role in the search for anti‐Alzheimer’s compounds, since preventing Aβ aggregation is one of the objectives for the development of therapeutic strategies. In general, these flavone derivatives exhibit an antidementia effect [ref. 38, ref. 135]. Additionally, the EE of the entire A. philoxeroides plant was evaluated in an in vivo model, showing that it improves recognition, spatial working, and reference memory in ovariectomized mice, indicating its potential to prevent senile dementia [ref. 135].
The inhibition of AChE and BChE was also evaluated in the EE of A. philoxeroides, but this extract did not exert a significant effect on these neurotransmitters [ref. 135]. A similar result was previously obtained by Silva and collaborators, who evaluated two extracts (AqE and EB) and a commercial preparation of inflorescences of G. globosa L. and found that none of these substances were capable of inhibiting AChE [ref. 98].
Kim (2019) conducted an in vivo and in vitro study to determine whether the EE of the aerial parts of Iresine celosia L. (Syn. of I. diffusa Humb. & Bonpl. ex Willd.) had an anti‐neuroinflammatory effect. In the in vitro study, the extract reduced cytokine levels and inflammatory mediators in the microglia, partly due to the inhibition of the MAPKs/NF‐κB signaling pathway. In the in vivo study, the researchers concluded that the EE improves behavioral dysfunctions caused by neuroinflammation in mice. These results suggest that the extract is a potential therapeutic agent for treating neuroinflammation associated with neurodegenerative diseases such as Parkinson’s, Alzheimer’s, and Huntington’s [ref. 104].
The aqueous and methanol extracts of I. herbstii were shown to affect the CNS by interacting with dopamine and serotonin receptors, suggesting their potential for treating diseases such as Parkinson’s and schizophrenia [ref. 108, ref. 201]. Regarding A. brasiliana, researchers found that the infusion of its aerial parts increases exploratory activity but has no effect on anxiety [ref. 30]. Meanwhile, the ME exhibited anxiolytic, sedative, and anticonvulsant effects [ref. 24]. In 2014, Mondal reported that the EE of A. sessilis has central stimulant activity [ref. 133]. It is also worth noting that P. glomerata was studied by three different research groups, which found that the EE of its roots and rhizomes did not have robust effects on depression and anxiety [ref. 219]. The HaE of the root exhibited stimulating effects, improved the acquisition and retention of behaviors, and partially reversed age‐related memory deficits [ref. 116]. Finally, the HF of ME of root was found to reduce stress and depressive behaviors and protect against anxiety development, possibly by maintaining antioxidant defenses and reducing oxidative damage [ref. 114, ref. 219].
Gastrointestinal Activity
Eight articles are currently discussed the gastrointestinal activity of 14 extracts and 8 fractions of 5 members of the Gomphrenoideae subfamily. It has been reported that the aqueous, methanolic, and hexanolic extracts of A. repens (Syn. A. sessilis (L.) R.Br. ex DC. and A. pungens Kunth), as well as six fractions of the ME, exhibit spasmolytic activity according to in vitro studies [ref. 216]. The AqE, EE, and ME of this plant also demonstrate antidiarrheal activity in vivo models [ref. 46, ref. 47, ref. 216]. Similarly, Saquib and Janbaz reported that the EE of A. sessilis has spasmolytic activity based on in vitro study results [ref. 64]. Additionally, it was reported that ME from the leaves of G. celosioides has an antiulcerogenic effect (both preventive and curative), which is likely related to its antioxidant activity [ref. 88, ref. 217]. The HaE of Guilleminea densa (Humb. & Bonpl. ex Schult.) Moq. leaves have been shown to have a gastroprotective effect. It can inhibit gastric lesions induced by indomethacin, ethanol, cold immobilization, or stress. Additionally, it increases gastric mucus, demonstrating an enhancing effect on the protective mucosal barrier. Finally, it inhibits the presence of ulcers, erosive gastritis, acute inflammation infiltration, and focal hemorrhage [ref. 102]. Freitas reported in 2004 that the AqE from roots and rhizomes of P. glomerata exerts a protective effect on the gastric mucosa [ref. 159].
Wound‐Healing Activity
To date, there are only six articles discussing the wound‐healing potential of members of the Gomphrenoideae subfamily. Four of these studies focused on the healing potential of the ME of A. brasiliana leaves by the Barua research group. Among the most relevant results obtained by this group is that the ME has a positive effect on wound contraction, fibroblast deposition, and angiogenesis. It also has pro‐healing activity in burn wounds, reducing the wound area more efficiently than Himax. Additionally, it increases endogenous antioxidant activity and angiogenesis. Similarly, it was shown that ME maintains its healing potential in wounds of old or immunocompromised mice [ref. 202, ref. 220, ref. 221, ref. 222]. Additionally, the healing potential of the EE of A. sessilis stems was studied in an in vitro model, revealing that the EE can enhance the progression of wound closure in normal and diabetic fibroblast cells, as well as in keratinocytes. This suggests that it has the potential to be used in late healing stages of diabetic patients [ref. 57].
Toxicology of Gomphrenoideae
Members of the Gomphrenoideae subfamily are widely used in traditional medicine to treat different ailments. To date, only 37 articles have reported the toxicity of 2.35% of the members of this subfamily (Table 11). It has been reported that the nHE and HE of A. bettzickiana possess mutagenic activity and toxicity against the BHK‐21 cell line [ref. 17]. Additionally, the AuNPs of AqE of leaves of this same plant have shown toxicity against zebrafish embryos at concentrations higher than 25 µM [ref. 21]. Regarding A. brasiliana, it was found that the ME and EaE of leaves do not present toxicity in mice at the maximum dose evaluated (5 and 2 g/kg, respectively) [ref. 24, ref. 185, ref. 202, ref. 220, ref. 221, ref. 222]. The HaE of the aerial parts did not present toxicity against murine macrophages at the evaluated concentration (20 µg/mL) [ref. 193], but the EE of leaves showed low toxicity against flies [ref. 26], and the AqE of leaves showed an LC50 of 500 µg/mL for Artemia salina [ref. 27]. The ME of A. philoxeroides did not show toxicity [ref. 39], whereas the AqE and EE extracts of the aerial parts of A. repens showed toxicity against mice (LD50: 3.4782 and 4.0639, respectively) [ref. 46]. On the contrary, the ME of the aerial parts and the EE of the stem of A. sessilis did not show toxicity up to the doses evaluated in an in vivo and in vitro model, respectively [ref. 57, ref. 204].
TABLE 11: Cytotoxicity of the Gomphrenoideae subfamily.
| Species | Extract(s)/Compounds | Assay method | Model | Dose | Positive control | Activity | References |
|---|---|---|---|---|---|---|---|
| Alternanthera bettzickiana | nHE of whole plant | MTT assays | BHK‐21 | Uninformed | — | IC50: 493 µg/mL for HE and IC50: 456 µg/mL for nHE. Cytotoxic activity is dependent on concentration. nHE exhibited greater activity than HEBoth extracts have dose‐dependent mutagenic potential. The enzyme activation system ↑ the mutagenicity in HE, whereas ↓ it in nHEnHE: MIx150 mg/mL: 33.7 and 45.29 for TA100 and TA102, respectivelyHE: MIx15 mg/Ml: 21.78 TA100 and MIx150 mg/mL: 12.30 for TA102 | [ref. 17] |
| Ames reverse mutation assay | Salmonella typhimurium TA‐100 and TA‐102 | 0.015, 0.15, 1.5, 15, and 150 mg/mL | SodiumAzide and H2O2 | ||||
| HE of whole plant | MTT assays | BHK‐21 | Uninformed | — | |||
| Ames reverse mutation assay | S. typhimurium TA‐100 and TA‐102 | 0.015, 0.15, 1.5, 15, and 150 mg/mL | SodiumAzide and H2O2 | ||||
| Alternanthera bettzickiana | AuNPs of AqE of leaves | Toxicity analysis | Zebrafish (Danio rerio) embryo model | 12, 25, and 50 µM | — | No toxicity was observed up to 25 µMAt 50 µM, it strongly inhibited hatching, affected tail formation, and dark material was observed in the intestinal tract | [ref. 21] |
| Alternanthera bettzickiana | EE of aerial parts | Acute oral toxicity study | Wistar rats | 2000 mg/kg | — | No mortality was observed, but slight behavioral changes such as convulsions and tremors occurred, along with ↓ somatomotor activity. It did not affect BW or cause organ injury. No significant changes for hemoglobin, RBCS, ESR, TLC, neutrophils, HCT, MCHC, MCH, MCV, LDL, VLDL, cholesterol, HDL, triglycerides, AST, ALT, proteins, globulin, albumin, and A/G ratio. ↑ platelet count and ↓ alkaline phosphatase and protein levels. It was concluded that the extract was safe | [ref. 20] |
| Alternanthera brasiliana | ME of leaves | Acute toxicity | Albino mice | 2.0 g/kg BW | — | The extract is safe up to 5 g/kg. Showed no changes in motor activity or behavior at a concentration of 2 g/kg | [ref. 202] |
| Determination of LD50 | Albino mice | Different [] up to 5 g/kg | — | ||||
| Alternanthera brasiliana | HaE of aerial parts | Toxicity test | Murine macrophages | 20 µg/mL | — | No apparent cytotoxic effects were observed in murine macrophages | [ref. 232] |
| Alternanthera brasiliana | ME of leaves | Acute toxicity | Swiss albino mice | 100, 200, 400, 800, 100, and 2000 mg/kg | — | The extract is safe up to 2000 mg/kg | [ref. 220] |
| Sprague Dawley rats | |||||||
| Alternanthera brasiliana | ME of leaves | Determination of LD50 | Swiss albino mice | 2.0 g/kg BW | — | The extract is safe up to 2000 mg/kg | [ref. 221, ref. 222] |
| Sprague Dawley rats | 2.0 g/kg BW | — | |||||
| Acute toxicity | Swiss albino mice | 2.0 g/kg BW | — | ||||
| Sprague Dawley rats | 2.0 g/kg BW | — | |||||
| Alternanthera brasiliana | ME of leaves | Determination of LD50 | Swiss albino mice (male) | 2000 mg/kg | — | The extract is safe up to 2000 mg/kg, showed no changes in motor activity or behavior | [ref. 24] |
| Acute toxicity | Swiss albino mice (male) | 2000 mg/kg | — | ||||
| Gross effect | Swiss albino mice (male) | 2000 mg/kg | — | ||||
| Alternanthera brasiliana | EaE of leaves | Acute toxicity | Swiss albino mice | 2000 mg/kg | — | LD50: >2000 mg/kg. Until the evaluated dose, no gross behavior changes or mortality were observed | [ref. 185] |
| Determination of LD50 | Swiss albino mice | Uninformed | — | ||||
| Alternanthera brasiliana | EE of leaves | Viability of flies | Drosophila melanogaster | 10, 20, and 40 µg/mL | — | EE exhibited low toxicity, as mortality significantly differed from the control only after 24 h, without a dose‐dependent response. After 48 h, EE killed more than 50% of the flies | [ref. 26] |
| Locomotor assay | Drosophila melanogaster | 10, 20, and 40 µg/mL | — | ||||
| Alternanthera brasiliana | AqE of leaves | Determination of LC50 | Artemia salina | [] up to 1000 µg/mL | LC50: 500 µg/mL | [ref. 27] | |
| Alternanthera brasiliana | HaE of leaves | MTT assays | RAW 264.7 and L929 | 3.9–500.0 µg/mL | Doxorubicin | IC50: 297.5 ± 22.8 µg/mL for RAW 264.7 and IC50: 340.7 ± 42.4 µg/mL for L929 | [ref. 193] |
| Alternanthera littoralis | EE of aerial parts | Biometric parameters | Swiss mice of both genders | 100 and 1000 mg/kg | — | The extract had no effect on weight, number of implantations, live and dead fetuses, resorptions, fetal viability, resorption rate, post‐implantation loss rate, placental index, weight adequacy for gestational age, head‐to‐tail distance, or urogenital distance in males and femalesMalformations detected: hyperextension of the forelimbs, unilateral hyperflexion of the hindlimb, curly tail, gastroschisis, hydrocephaly, hydronephrosis, femur agenesis, and reduced ossification of skull bonesEE did not change the frequency of micronuclei | [ref. 253] |
| Reproductive parameters and embryofetal development | |||||||
| Micronucleus (MN) levels in peripheral blood | |||||||
| Splenic phagocytosis | |||||||
| Alternanthera philoxeroides | ME of whole plant | Acute toxicity test | Swiss albino mice | 100, 200, 300, 600, 800, 1000, 2000, and 3000 mg/kg BW | — | ME not cause changes in behavior or mortality; therefore, it is considered safe up to the evaluated dose | [ref. 39] |
| Alternanthera philoxeroides | ME extract | Swiss albino mice (female) | Body weight and relative organ weight | 250, 500, and 1000 mg/kg | — | ME did not cause changes in BW or organ weight or cause architectural or degenerative changesNo changes were observed in erythropoiesis, morphology, or osmotic fragility of RBC. No significant changes were observed in leukocyte counts except for neutrophils, which decreased markedly. No changes were observed in platelet count and platelet indicesME is not toxic | [ref. 132] |
| Histopathological analyses | — | ||||||
| Hematological analyses | — | ||||||
| Alternanthera repens | AqE of aerial parts | Determination of LD50 | Swiss mice (female) | 0.25–8 g/kg | — | LD50: 3.4782 and 4.0639 for AqE and EE, respectively. On the basis of LD50 values, the extracts are slightly toxic | [ref. 46] |
| EE of aerial parts | Determination of LD50 | Swiss mice (female) | 0.25–8 g/kg | — | |||
| Alternanthera sessilis | ME of aerial parts | Acute toxicity test | Swiss albino mice (male) | 100, 200, 300, 600, 800, 1000, 2000, and 3000 mg/kg BW | — | Non‐toxic up to the evaluated dose | [ref. 204] |
| Alternanthera sessilis | EE of stem | MTT assays | NHDF cells | 15.62, 31.25, 62.5, 125, 250, and 500 µg/mL | — | Non‐toxic up to the evaluated dose | [ref. 56] |
| HDF‐D cells | |||||||
| HaCaT cells | |||||||
| Alternanthera sessilis | ME of whole plant | Acute toxicity test | Female albino rats | 250, 1000, and 2500 mg/kg BW | — | Results not reported | [ref. 51] |
| Alternanthera sessilis | AgNPs of leaves | Toxic effect of AgNPs against non‐target organisms | Poecilia reticulata | Uninformed | Silver nitrate | The AgNPs complex was not toxic to Poecilia reticulata | [ref. 158] |
| Blutaparon portulacoides | EE of stems | Acute toxicity test | Female Wistar rats | 2000 mg/kg | — | No signs or symptoms of acute and clinical oral pathophysiology were observed with EE use | [ref. 71] |
| Blutaparon portulacoides | AqE of whole plant | Acute toxicity | Female Wistar rats | 2000 mg/kg | — | LD50: >2000 mg/kgNo significant behavioral or physiological changes were induced in female rats | [ref. 152] |
| Gomphrena celosioides | AqE of leaves | Preliminary acute toxicity study | Adult Sprague–Dawley rats | 20, 40, 80, 160, 320, and 640 mg/kg | — | Non‐toxic up to evaluated dose | [ref. 87] |
| Gomphrena celosioides | HeE of whole plant | Brine shrimp toxicity assay | Brine shrimp nauplii | 1000, 100, and 10 ppm | Podophylotoxin | LC50 (µg/mL) of HeE: 52.146LC50 (µg/mL) of 449: 110.654LC50 (µg/mL) of ME: 77.978The LC50 values of HeE and ME suggest the presence of cytotoxic and/or insecticidal compounds. The LC50 indicates weak cytotoxicity | [ref. 82] |
| 449 | |||||||
| ME of whole plant | |||||||
| Gomphrena celosioides | AcE and AqE of flowers, leaves, twigs, and whole plant | MTT assay | Vero monkey kidney cells | Range of 0.03–1 mg/mL | — | All extracts showed low toxicity | [ref. 89] |
| Gomphrena celosioides | EE of aerial parts | Acute oral toxicity | Adult Wistar rats | 2000 mg/kg | — | LD50: >2000 mg/kgEE from aerial parts is safe, as no rats exhibited clinical signs of toxicity. Histopathological studies showed no hepatotoxicity, nephrotoxicity, or hematotoxicity | [ref. 254] |
| Subacute oral toxicity | 75, 150, or 300 mg/kg | — | |||||
| Gomphrena celosioides | EE of aerial parts | Teratogenesis and genotoxicity | Pregnant mice (Mus musculus) | 100, 1000, and 2000 mg/kg | — | Not alter the final weight, weight gain, uterine weight, or net weight gainNot affect the numbers of implantations, live fetuses, dead fetuses, or resorptionsNot change fetal weight, placental weight, or the placental indexThe frequency of malformations (external, visceral) did not differ between the EE‐treated and control groupsEE ↑ the frequency of abnormal sternum and fibula rotation over the tibiaEE did not change the frequency of micronucleiThese results suggest that daily doses up to 2000 mg/kg are not maternotoxic | [ref. 255] |
| Biological tests | Pregnant mice (Mus musculus) | 100, 1000, and 2000 mg/kg | — | ||||
| Biometric parameters | Pregnant mice (Mus musculus) | 100, 1000, and 2000 mg/kg | — | ||||
| Reproductive performance and embryofetal development | Pregnant mice (Mus musculus) | 100, 1000, and 2000 mg/kg | — | ||||
| Micronucleus in peripheral blood | |||||||
| Splenic phagocytosis | |||||||
| Gomphrena globosa var. albiflora (white amaranth) | HE of flowers | SRB assay | PLP2 cells | Uninformed | — | Non‐toxic up to the evaluated doseGI50 (µg/mL): >400 | [ref. 73] |
| Gomphrena haageana K. (red amaranth) | HE of flowers | SRB assay | PLP2 cells | Uninformed | — | Non‐toxic up to the evaluated doseGI50 (µg/mL): >400 | [ref. 73] |
| Gomphrena sp. (pink globe amaranth) | HE of flowers | SRB assay | PLP2 cells | Uninformed | — | Non‐toxic up to the evaluated doseGI50 (µg/mL): >400 | [ref. 73] |
| Gomphrena globosa | HaE of flowers | Alamar blue and neutral red tests | HaCaT cells and BJ cells | 50, 250, and 500 µg/mL | — | It did not affect BJ cell viability but ↓ HaCat cell viability at high concentrations | [ref. 153] |
| Hebanthe eriantha | ME of roots | Determination of LC50 | Artemia salina | Range of 7.81–1000 µg/mL | Potassium dichromate | MEs showed toxicity in A. salina, with lethality ranging from 26.7% to 60% at concentrations of 31.25–1000 µg/mL | [ref. 230] |
| Pfaffia glomerata | HaE of roots | Acute toxicity test | Wistar rats (male) | 3 g/kg | — | No behavioral changes or deaths were observed in rats (LD50: >3 g/kg) | [ref. 116] |
| Determination of LD50 | |||||||
| Pfaffia glomerata | AqE of aerial parts | Cytotoxicity assay | J774 cell line | 1, 10, and 100 µg/mL | — | Non‐toxic up to the evaluated dose | [ref. 234] |
| Pfaffia glomerata | Commercial root dry extract | Cytotoxic assay | Wistar rats | 0.15, 1.5, and 15 mg/mL | — | No cytotoxic effects or mutagenic potentials were observed | [ref. 112] |
| Chromosomal aberration test | Wistar rats | 0.15, 1.5, and 1.5 mg/mL | Cyclophosphamide | ||||
| Pfaffia glomerata | FD of roots | MTT assay | BMDM (bone marrow‐derived macrophage) | 250, 25, 2.5, and 0.25 µg/mL | — | Cytotoxicity was dose‐dependent, with lower cell viability at higher concentrations and higher viability for the concentrations of 2.5 and 0.25 µg/mL, maintaining viability >65% | [ref. 162] |
| FD of aerial part | MTT assay | BMDM (bone marrow‐derived macrophage) | 250, 25, 2.5, and 0.25 µg/mL | — | |||
| Pfaffia paniculata | Powdered roots | ALT activity | Inbred BALB/cICB mice | 200 and 400 mg/kg | — | Histopathological changes were not observed in the liver, kidney, or spleen. No changes in BW and ALT activities were detected | [ref. 207] |
| Histopathological study | |||||||
| Pfaffia paniculata | BuF of roots | BW measurement | SWISS mice (male) | 200 mg/kg | — | ↓ Weight gain on Days 13 and 15. No hepatic or renal toxicity was observed, according to histopathological analysis and levels of ALT, AST, γ‐GT, urea, and creatinine | [ref. 209] |
| Biochemical analysis | |||||||
| Histopathological study | |||||||
| Pfaffia paniculata | ME of roots | Histopathological study | Adult BALB/c mice (male) | 250, 500, and 1000 mg/kg | — | No histopathological alterations were observed in the liver, kidney, lung, brain, eye, or cerebellum at the studied dose. Only a tendency toward ↓ BW was observed at a [] of 1000 mg/kg | [ref. 183] |
Abbreviations: AqE, aqueous extract; EaE, ethyl acetate extract; EE, ethanolic extracts; HaE, hydroalcoholic extract; HE, hydromethanolic extracts; ME, methanolic extracts; nHE, n‐hexane extracts.
The EE of B. portulacoides and the AqE of G. celosioides did not show toxicity in rats up to the dose evaluated [ref. 71, ref. 87], but the HeE and ME of G. celosioides showed weak cytotoxicity against brine shrimp or nauplii, and the AcE and AqE extracts of this same plant showed low toxicity against Vero cells of monkey kidneys [ref. 82, ref. 89]. On the other hand, the HE of G. haageana and G. globosa flowers did not show toxicity against PLP2 cells [ref. 73]. Additionally, the HaE of G. globosa flowers did not show toxicity against BJ cells but did against HaCat cells [ref. 153]. Regarding P. glomerata and P. paniculata, no toxic effects were observed up to the doses evaluated in rats (3 g/kg) and mice (1 g/kg), respectively [ref. 112, ref. 116, ref. 183, ref. 207, ref. 209]. It was also reported that the commercial extract of the root of P. glomerata has no cytotoxic effect on the J774 cell line, nor mutagenic effects [ref. 234].
It can be concluded that the extracts, fractions, and commercial preparations of the studied members of this subfamily do not present toxicity, or it is very low. Furthermore, toxicological safety evaluations are essential for the application or use of plants and the development of new drugs. Therefore, it is necessary to evaluate the toxicity of the other members of this subfamily.
Relationship Among Traditional Uses, Biological Activity, and Chemical Profile
Different parts or even the whole plant of the members of Gomphrenoideae have been used in traditional medicine to treat bacterial, fungal, and viral infections, parasitic diseases, diabetes, cancer, hypertension, inflammatory diseases, gastrointestinal diseases, and liver damage. They have also been used as antioxidants, analgesics, and diuretics, as well as to treat other pathologies. Therefore, during the last decades, they have been studied at the laboratory level, mainly to verify their antimicrobial, antioxidant, anticancer, antidiabetic, hepatoprotective, gastroprotective, diuretic, and insecticidal properties. An attempt to compare traditional use with results from the laboratory is described as follows:
Alternanthera bettzickiana: Out of the 17 traditional uses, only 4 have been evaluated at the laboratory level, confirming its antimicrobial, antioxidant, and cytotoxic (against the A549 cell line) activities. Additionally, two in vitro studies showed antiarthritic potential. The results obtained to date under laboratory conditions confirm only 23.53% of the traditional uses; therefore, it is necessary to confirm the other traditional uses.
Alternanthera brasiliana: To date, only 10 of the 25 traditional uses of this plant have been evaluated, confirming its anticancer potential against the A549, CaCo‐2, HT‐29, and Hep‐G2 cell lines, as well as its positive effect in mice bearing EAC. In addition, its use in the treatment of infections was confirmed, because it has been reported to have activity against some bacteria and yeasts. Its anti‐inflammatory, analgesic, anticonvulsant, anxiolytic, and immunomodulatory activities were also confirmed, as well as its great potential in wound healing.
Alternanthera flavescens: None of its traditional uses have been evaluated under laboratory conditions, but its antioxidant and anticancer activities have been evaluated.
Alternanthera littoralis P. Beauv. (Alternanthera maritima (Mart.) St. Hil.): Currently, its two traditional uses have been evaluated under laboratory conditions, confirming that this plant has antifungal activity against yeasts and anti‐inflammatory activity. Although its traditional uses do not include antioxidant, antiparasitic, and immunomodulatory activities, these properties were evidenced through pharmacological studies.
Alternanthera paronychioides: Of the seven uses in traditional medicine, only antidiabetic activity has been evaluated under laboratory conditions through in vitro tests, suggesting that this plant has antidiabetic properties as well as antioxidant activity.
Alternanthera philoxeroides: Of the 21 traditional uses, 4 have been evaluated under laboratory conditions, confirming that this plant has antiviral activity (e.g., anti‐herpes, anti‐measles), potential antidiabetic activity (inhibits α‐glycosidase), analgesic, and antidepressant effects. Additionally, this plant has been reported to have antibiotic, antioxidant, cytotoxic (against HeLa cells), anticoagulant, and cardioprotective activities, as well as an antidementia and memory‐enhancing effect in mice.
Alternanthera porrigens Kuntze: None of its traditional uses have been confirmed under laboratory conditions.
Alternanthera pungens Kunth: None of its traditional uses have been confirmed under laboratory conditions. However, some articles report that this plant has antitumor activity, but it does not have antifungal activity, nor is it a significant antioxidant.
Alternanthera repens (Alternanthera caracasana): Of the nine traditional uses, only its antidiarrheal activity has been evaluated and confirmed under laboratory conditions. It should be noted that this plant has also been reported to have no activity against C. albicans.
Alternanthera sessilis: To date, only 11 of the 63 traditional uses have been evaluated under laboratory conditions, confirming that it has antiasthmatic, antidiarrheal, antidiabetic, hypotensive, hepatoprotective, analgesic, wound healing, anti‐inflammatory, antioxidant, and antimicrobial (against bacteria) activity. It has also been shown to be cytotoxic against HeLa, Panc‐1, MIA, PaCa‐2, Capan‐1, PC3, L929, and MCF‐7 cells and to have antiallergic and CNS‐stimulating properties.
Alternanthera tenella: Of the 18 uses in traditional medicine, only the anti‐inflammatory, analgesic, and antimicrobial activities have been confirmed by pharmacological studies. Additionally, it has been reported to also have antioxidant, anticancer, and immunomodulatory properties.
Blutaparon portulacoides: Of the two traditional uses, studies confirm its potential to treat vulvovaginitis under laboratory conditions. Additionally, it has also been reported to have antibiotic, antiparasitic, anti‐inflammatory, and analgesic properties, as well as diuretic and cardioprotective effects.
Gomphrena agrestis: There are no reports of traditional uses but has been evaluated for antimicrobial, antifungal, and antiparasitic activity.
Gomphrena arborescens L.f.: Under laboratory conditions, none of the traditional uses have been confirmed.
Gomphrena boliviana: Four traditional uses have been reported, of which only antimicrobial activity has been confirmed under laboratory conditions.
Gomphrena celosioides: Of the 22 traditional uses, 9 have been evaluated through pharmacological studies, confirming that G. celosioides has the potential to be used in the prevention and treatment of Type 2 diabetes, as well as in the treatment of gastric lesions, to counteract renovascular hypertension, and to treat infections (antibiotic and antifungal activities). It also has hepatoprotective, analgesic, anti‐inflammatory, and immunomodulatory properties. Additionally, it has been reported to have cardioprotective and anticarcinogenic activity.
Gomphrena elegans: Its use in traditional medicine has not been reported, but pharmacological studies have shown that it has cytotoxicity against the HCT‐8, SF‐295, and MDA‐MB‐435 cell lines, as well as potential as an insecticide against Aedes aegypti.
Gomphrena globosa: To date, only 2 of its 20 traditional uses have been evaluated under laboratory conditions, which report that this plant has antibacterial, antifungal, and antioxidant activities, although these are considered weak. Assays have shown no cytotoxic activity against the MT‐1, MT‐2, B16F10, HeLa, and MK‐1 cell lines, nor any sun protection effect or AChE inhibition. However, it has anti‐inflammatory and anti‐collagenase properties.
Gomphrena haageana K.: No traditional uses have been reported, but it has been found to have antioxidant and anti‐inflammatory properties.
Gomphrena macrocephala: Its two common uses have not been evaluated by pharmacological studies, but two compounds isolated from this plant have been reported to show cytotoxic activity against HSC‐2 cells.
Gomphrena martiana: Of the eight traditional uses, only its activity in the treatment of infections has been confirmed by antimicrobial studies, indicating that it has antibacterial and antifungal activity. Additionally, its activity against the KB cell line has been evaluated, showing moderate cytotoxicity against it. It has also been shown to have a beneficial effect on mice bearing S‐180 cells or EAC.
Gomphrena virgata Mart.: Its five traditional uses have not yet been confirmed by pharmacological studies, but it has been reported that this plant can inhibit the proliferation of lymphocytes.
Guilleminea densa: Only one of the three traditional uses was evaluated by a pharmacological study, which showed that the plant has a gastroprotective effect, inhibiting gastric lesions and preventing the reduction of mucus induced by toxic agents, confirming its use in the treatment of gastric ulcers.
Iresine difusa: Of the 12 traditional uses, only the anticancer potential has been evaluated under laboratory conditions. It was reported to have cytotoxic activity against the human LNCaP cell line, but not against PC3. Additionally, in vitro and in vivo studies have shown that this plant can be used to treat diseases related to neuroinflammation.
Iresine herbstii: Of the 13 traditional uses, only 2 have been investigated pharmacologically. It was found to have anticancer potential, showing cytotoxic activity against the HeLa cell line. But no significant wound‐healing activity. Although its traditional uses do not mention antibiotic, antioxidant, or neurological activity, in vitro studies suggest that this plant has high antibacterial and antioxidant activities as well as has a beneficial effect on the CNS.
Pfaffia glomerata: Of the 22 known traditional uses, only 7 have been evaluated in the laboratory, confirming its anti‐inflammatory, analgesic, and stimulant properties. It also reduces stress and depressive behaviors and protects against the development of anxiety but has shown variable antioxidant activity. Other studies indicate that this plant has antiparasitic activity against Leishmania braziliensis and L. amazonensis, and its fractions have shown activity against T. cruzi. Additionally, this plant has demonstrated inhibition of melanogenesis.
Pfaffia paniculata: Of the 13 traditional uses, only its anticancer activity (in vitro and in vivo) and anti‐inflammatory properties have been confirmed through pharmacological studies. It was also observed that it did not show antitumor activity in EAC‐bearing mice.
Pfaffia townsendii: Of the four traditional uses reported, only the anti‐inflammatory activity has been confirmed through an in vivo study. Although its traditional uses do not include antioxidant activity, an in vitro study showed that it has antioxidant potential.
Tidestromia oblongifolia: Its traditional use as an analgesic has not been confirmed by pharmacological studies, highlighting that, to date, no study has been carried out to evaluate its biological potential.
From the above, it can be concluded that only 21.18% of the traditional uses reported for the members of this subfamily have been evaluated under laboratory conditions, whereas 78.2% remain untested. These data are relevant because they suggest a promising field of study, as most studies have shown that the plants of this subfamily indeed have biological activity consistent with their traditional uses. The traditional uses of these plants justify multidisciplinary research, which may include determination of biological activity, identification of chemical profiles, correlation between phytochemical profiles and biological activity, comparative studies of extracts, fractions, isolated compounds, and drug delivery, as well as comparative studies between varieties, plants collected from different locations under varying environmental conditions, and in vitro systems.
Gomphrenoideae Subfamily: Perspectives and Research Directions
Despite significant progress in understanding the biotechnological and chemical potential of the Gomphrenoideae subfamily, many research opportunities remain. These opportunities can be categorized into the following key areas of focus.
Evaluation of the Chemical Profile and Activity of Species That Remain Unstudied
Over 80% of species have not yet been studied in terms of their chemical profiles and biological activities, presenting a valuable opportunity to discover new chemical structures and significant biological activities that could be of interest to the pharmaceutical industry.
In Vitro Cultures: A Sustainable and Efficient Strategy in Biotechnological Studies
In the sections on pharmacological activity and phytochemical, it is evident that the same species can exhibit multiple chemical profiles, which, in turn, modulate their biological activity. This variation occurs because the phytochemical profile of plants is influenced by various biotic and abiotic factors, including climatic conditions, UV radiation exposure, soil characteristics, nutrient availability, and interactions with other organisms, such as microorganisms.
In this context, a viable alternative is the use of in vitro plant tissue cultures, which allow for the production of genetically identical plants or callus under controlled growth conditions. The use of in vitro cultures not only standardizes growth conditions but also optimizes the production process and helps prevent ecosystem degradation.
Co‐Cultures of Plants and Endophytic Microorganisms
Endophytic microorganisms play a crucial role in the phytochemistry of plants in natura. In this context, three key theories should be considered: (1) Endophytic microorganisms are responsible for producing certain chemical compounds; (2) microorganisms function as elicitors in the plant and stimulate the production of secondary metabolites; and (3) the interaction between the plant and the microorganism is necessary for the synthesis of specific compounds, which cannot be synthesized in their absence. This could be because endophytes act as stimulants, provide essential precursors for synthesis, or vice versa.
In this context, the use of in vitro plant tissue cultures in co‐culture with one or more endophytic microorganisms represents a valuable approach, which also allow for a better understanding of plant‐microorganism interactions.
Endophytic Microorganisms as a Source of Secondary Metabolites
Endophytic microorganisms are sometimes responsible for producing certain plant secondary metabolites, making them a sustainable alternative that minimizes environmental impact, shortens production times, and allows for process optimization.
Additionally, microorganisms are recognized as a promising source of bioactive molecules due to their metabolic plasticity, which enables them to mimic the metabolism of their host.
Conclusion
The members of the Gomphrenoideae subfamily (Amaranthaceae) have been used in traditional medicine around the world since ancient times. Although some members of this subfamily have been studied at the pharmacological and phytochemical levels, many others remain unstudied, presenting opportunities for research. Additionally, the literature review reveals a direct relationship between the traditional uses of these plants and the biological activity exhibited by the extracts, fractions, and compounds studied. There is also a correlation between the chemical profile and the types of compounds present and the observed biological activity. Demonstrating the importance of understanding the pharmacological activity of plants to harness this knowledge for the development of new drugs.
Research focused on discovering bioactive compounds can contribute to the development of drugs with fewer side effects and greater effectiveness than current options. Currently, drugs for treating pain, inflammation, cancer, diabetes, microbial infections, and oxidative stress‐related conditions are known for their side effects and limited effectiveness. This highlights the need to discover new drugs.
In general, it can be concluded that some phytochemicals found in the subfamily Gomphrenoideae can be used in drug development. Studying the members that have not yet been researched could lead to the discovery of new chemical compounds and the potential development of new drug.
Currently, 512 compounds have been isolated, including 173 phenolic compounds, 95 terpenoids, 87 lipid compounds, 62 alkaloids, and 95 other types of compounds, with phenolic compounds being the most abundant. These different extracts, fractions, and isolated compounds have been associated with various biological activities, such as antioxidant, analgesic, antibacterial, antifungal, antiparasitic, anticancer, antitumor, anti‐inflammatory, antidiabetic, antiarthritis, cardioprotective, healing, diuretic, gastroprotective, hepatoprotective, radioprotective, and hypertension and blood pressure management. The most extensively studied biological activities to date are anticancer, antimicrobial, and antioxidant activities. In conclusion, these plants represent a promising source of bioactive molecules with low or no toxicity.
Author Contributions
Dayanna Isabel Araque Gelves: bibliographic survey, writing and revision of text, design of graphical abstract, design of chemical structures and tables. Giulia Cristina Andreoli de Souza: bibliographic survey, writing and revision of text, design of graphical abstract, design of chemical structures and tables. Alvaro Jose Hernandez Tasco: bibliographic survey, writing and revision of text, design of graphical abstract, design of chemical structures and tables. Marcos Jose Salvador: bibliographic survey, writing and revision of text, design of graphical abstract, design of chemical structures and tables.
Conflicts of Interest
The authors declare no conflicts of interest.
Supplementary Materials
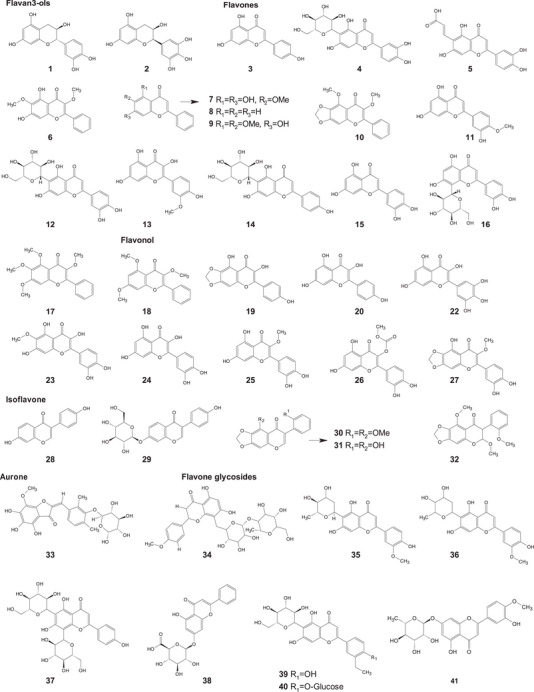
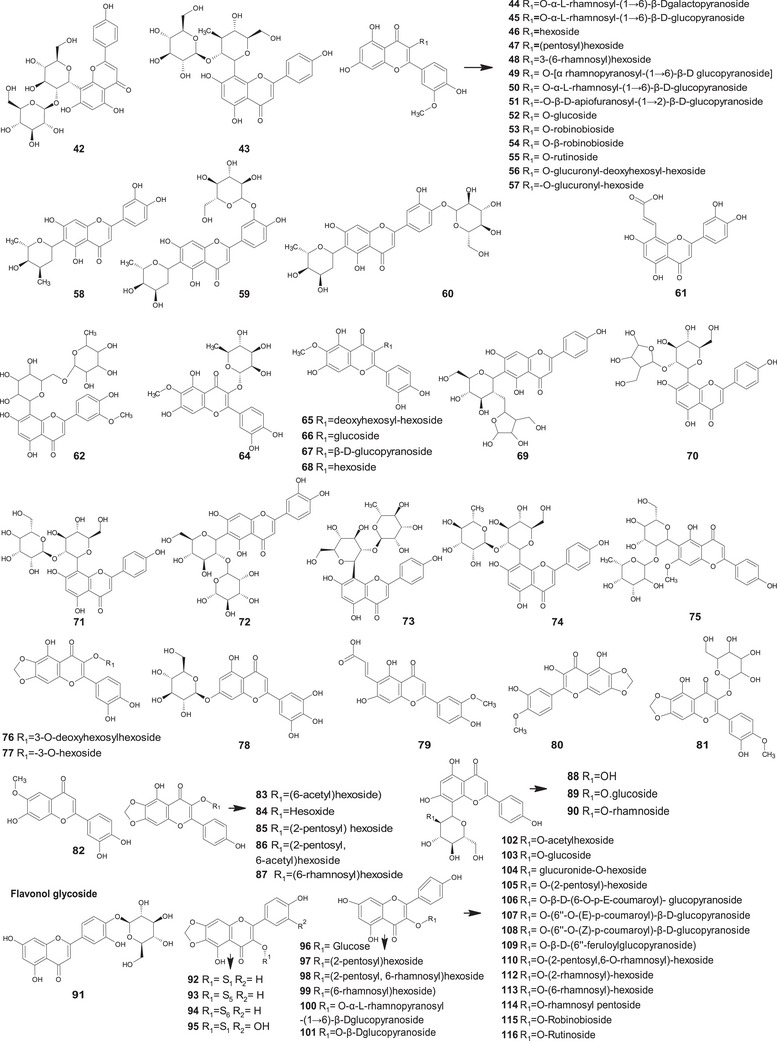
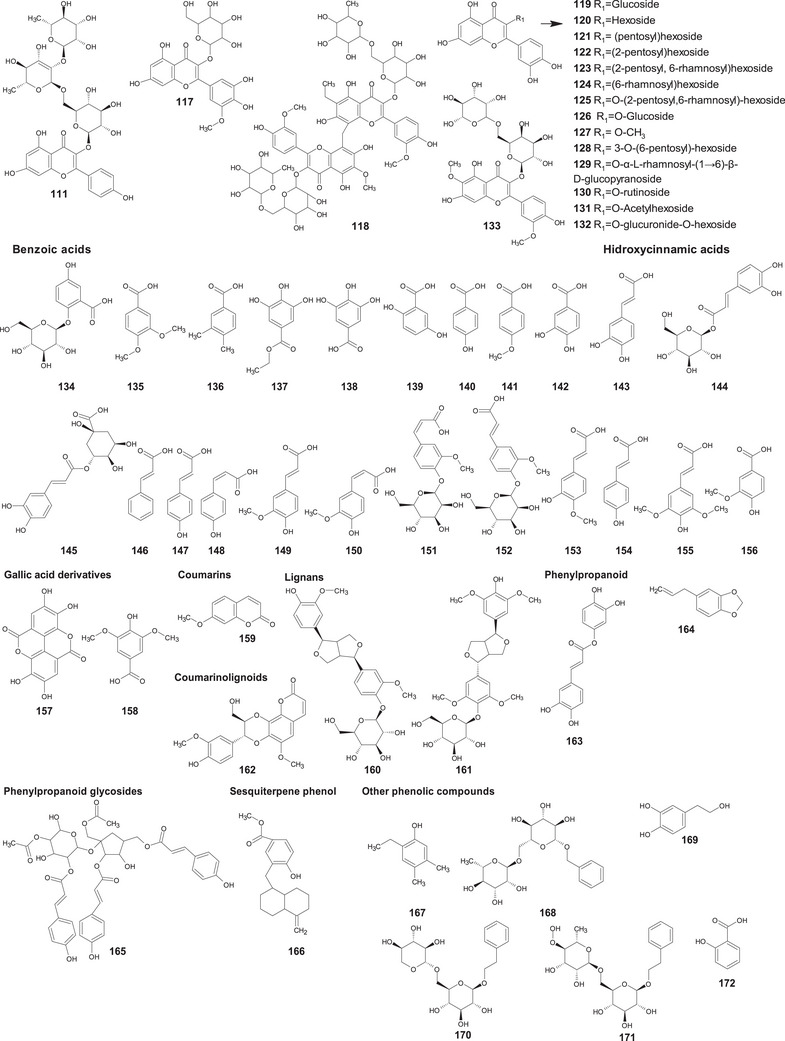
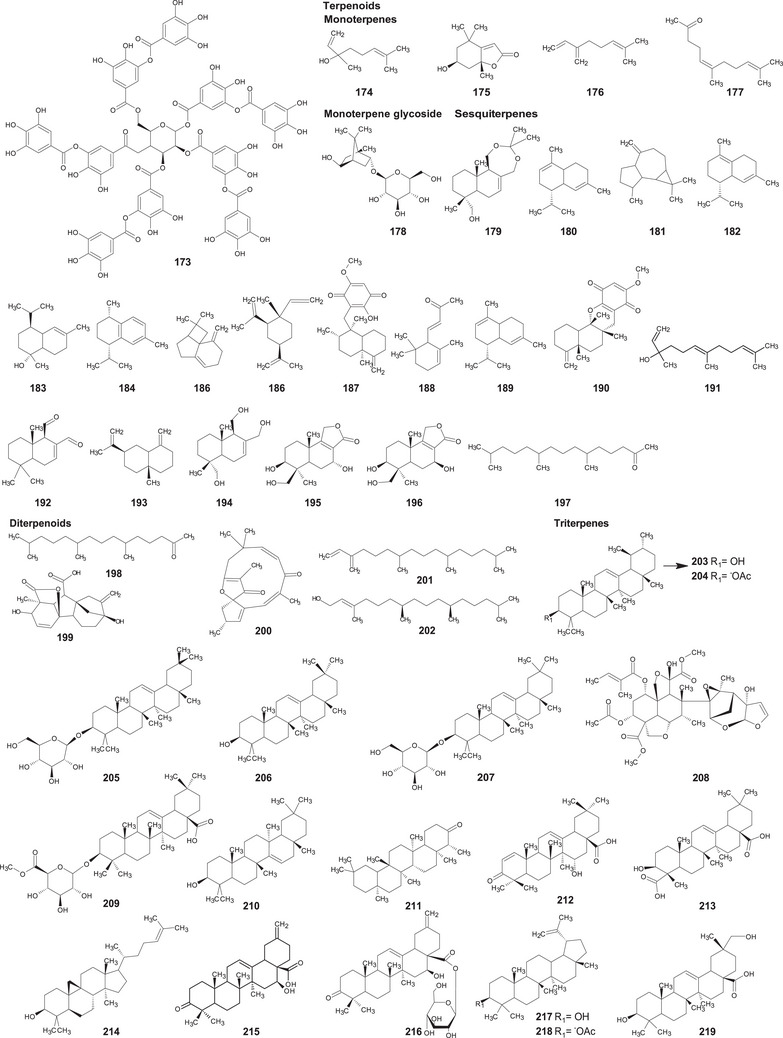
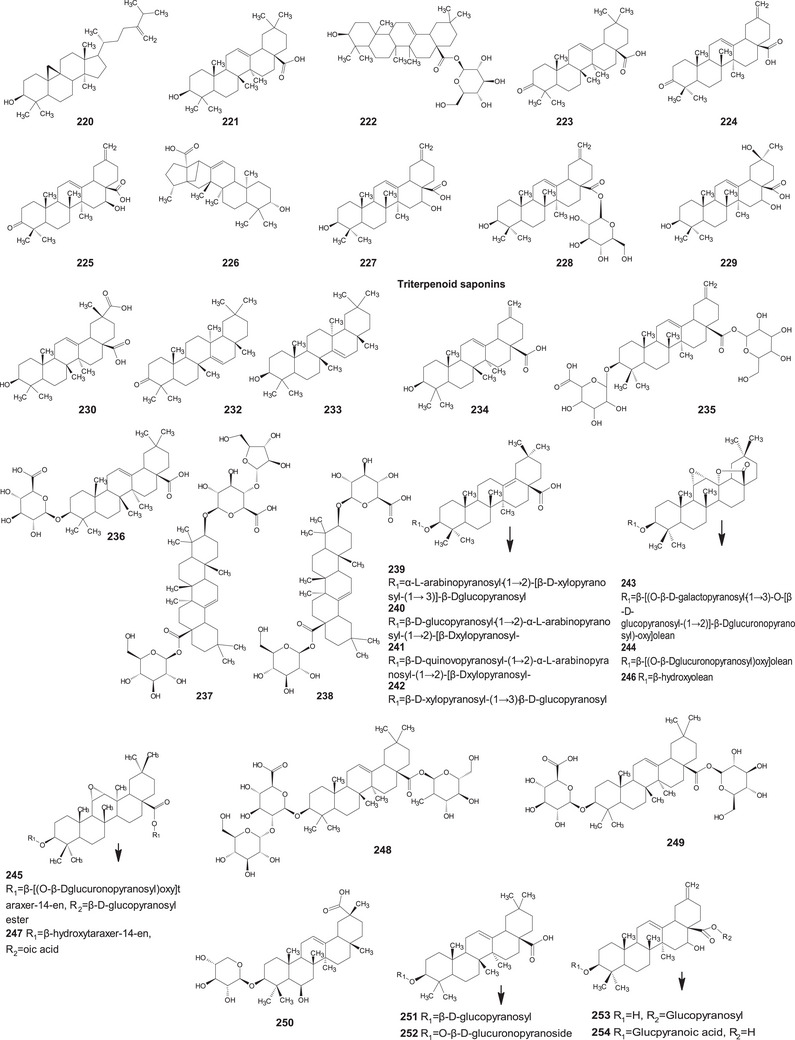
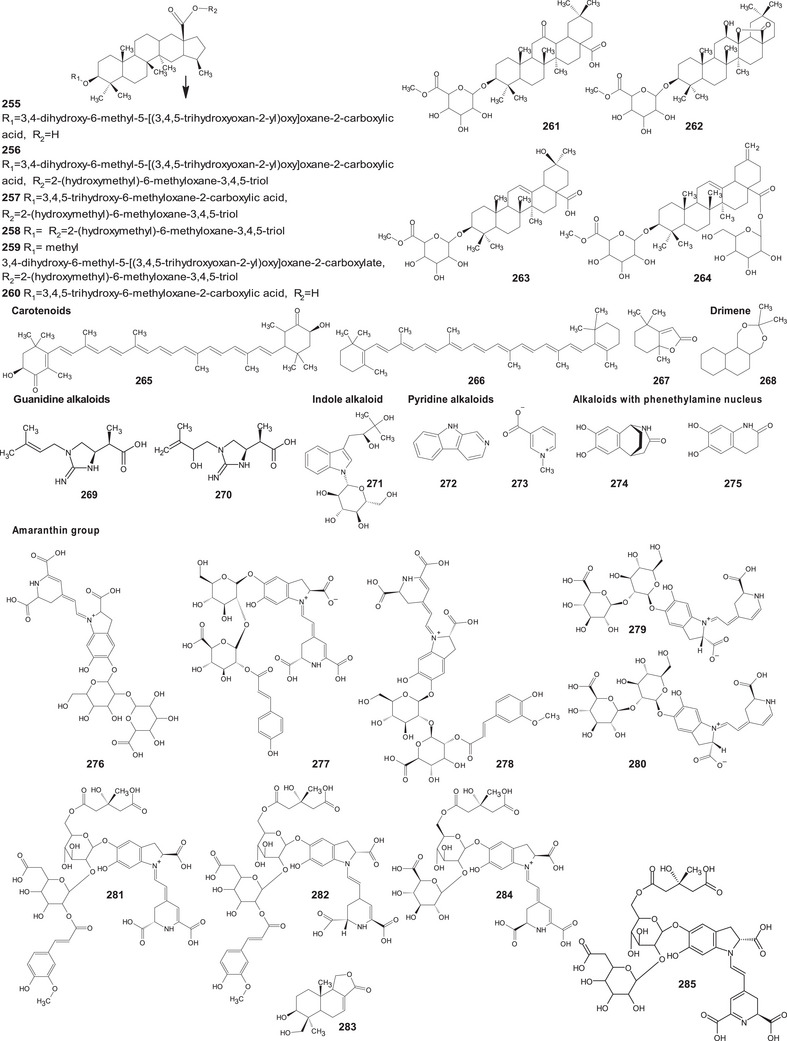
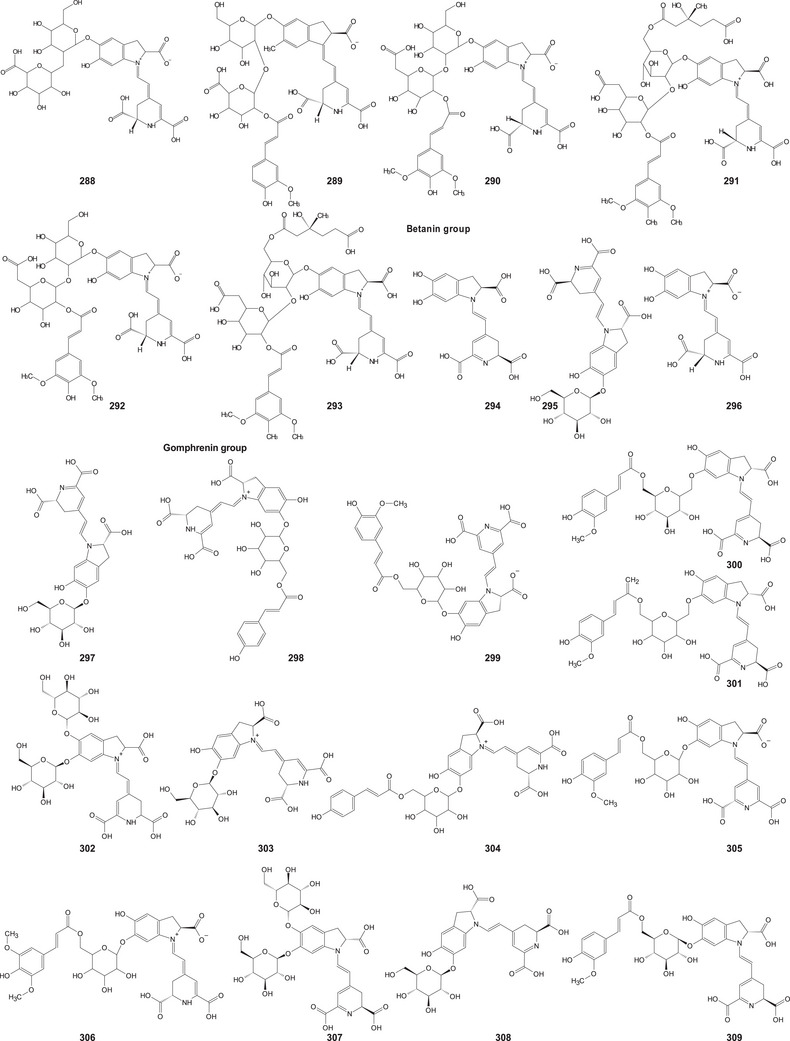
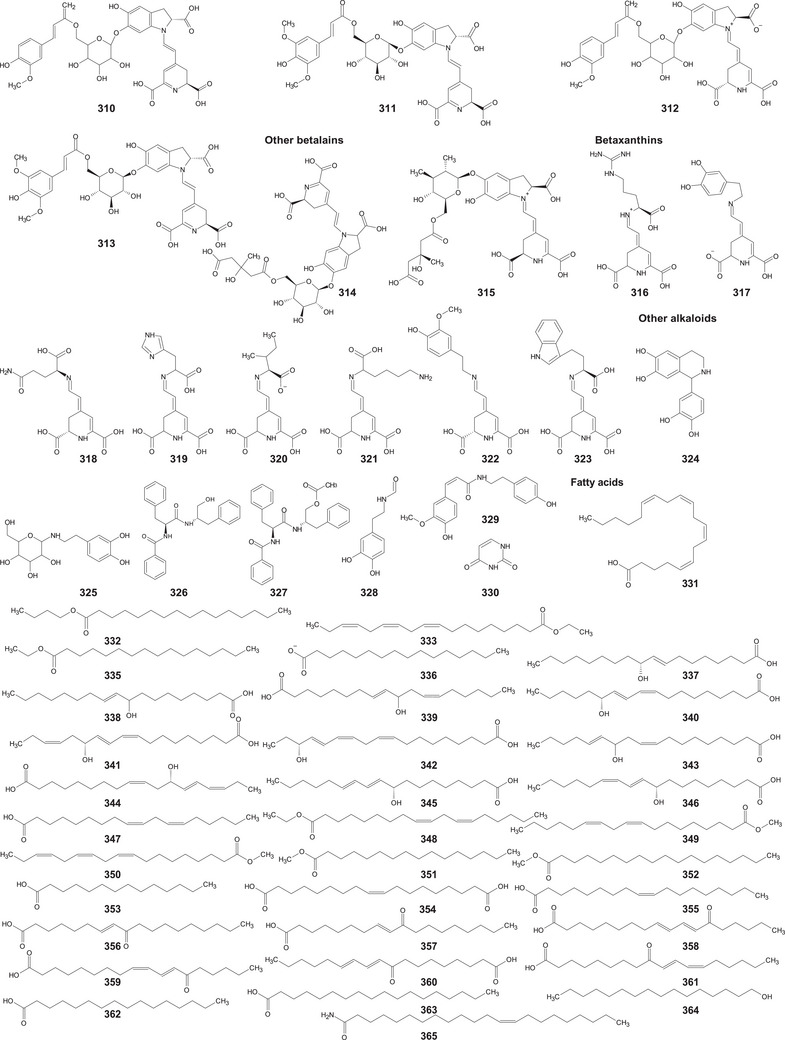
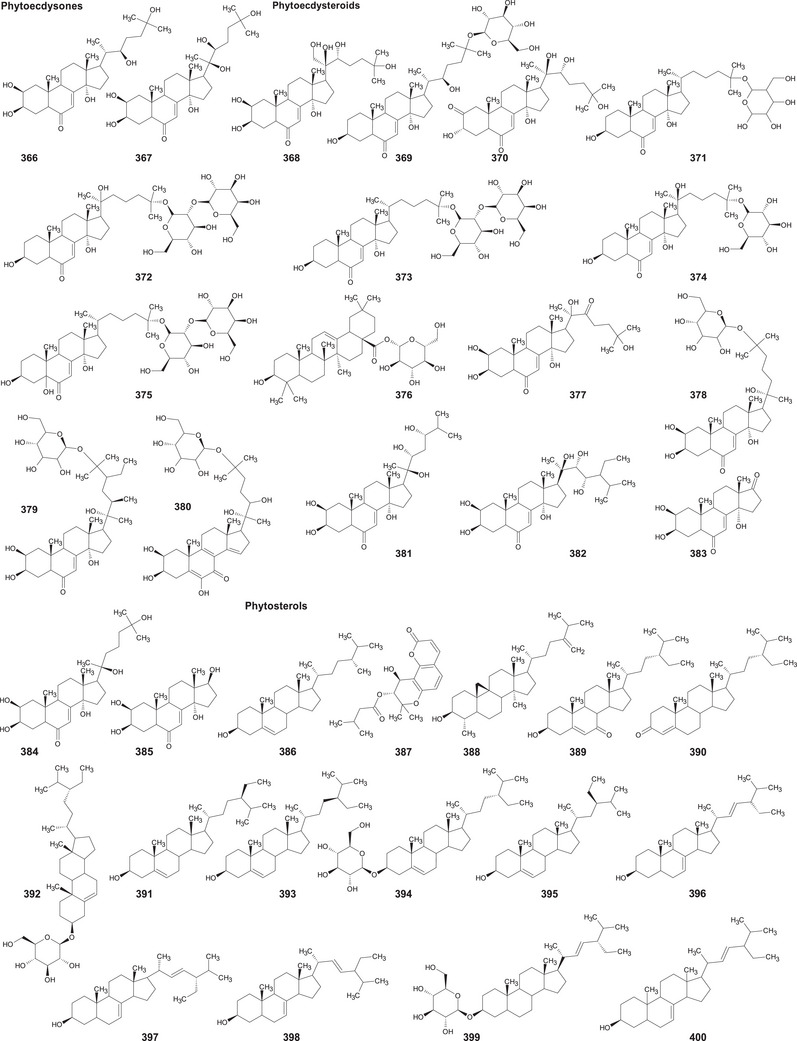
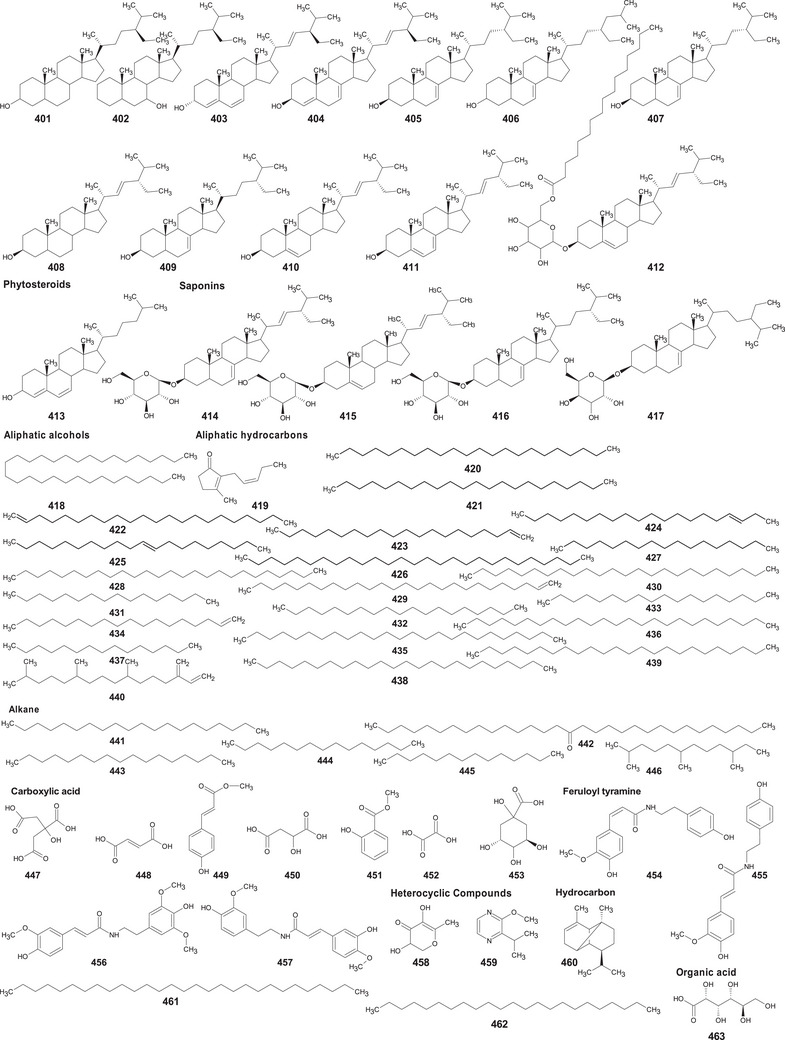
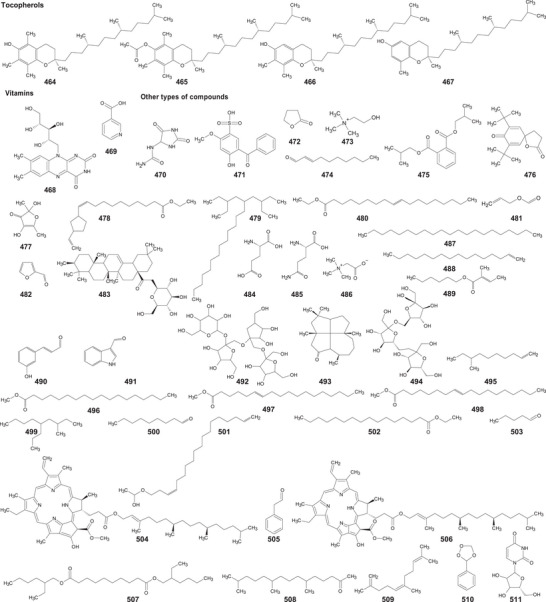
References
- Phylogeny of Amaranthaceae and Chenopodiaceae and the Evolution of C4 Photosynthesis,”. International Journal of Plant Sciences, 2003. [DOI]
- A New Heptasubstituted (E)‐Aurone Glucoside and Other Aromatic Compounds of Gomphrena agrestis With Biological Activity,”. Zeitschrift Fur Naturforschung—Section C Journal of Biosciences, 2004. [DOI]
- Biological Potential and Phytopharmacological Screening of Gomphrena Species,”. International Journal of Pharma Research & Review, 2014. [DOI]
- Phytochemistry and Bioactivity of Triterpene Saponins From Amaranthaceae Family,”. Phytochemistry, 2015. [DOI]
- 5 G. E. Burrows and R. J. Tyrl “Amaranthaceae Juss,” Toxic Plants of North America, 2nd ed. 28–34 (Wiley‐blackwell, 2013), 10.1002/9781118413425.ch5.
- A Review on Taxonomic and Use Diversity of the Family Amaranthaceae in Ethiopia,”. Journal of Medicinal Plants Studies, 2016
- An Update of the Angiosperm Phylogeny Group Classification for the Orders and Families of Flowering Plants: APG IV,”. Botanical Journal of the Linnean Society, 2016. [DOI]
- Antiprotozoal and Antioxidant Alkaloids From Alternanthera littoralis ,”. Phytochemistry, 2017. [DOI | PubMed]
- Optimization of Ultrasound‐Assisted Extraction of Bioactive Compounds From Gomphrena celosioides Mart. Using Response Surface Methodology,”. Bulletin of the Chemical Society of Ethiopia, 2020. [DOI]
- Achyranthes Aspera Root Extracts Induce Human Colon Cancer Cell (COLO‐205) Death by Triggering the Mitochondrial Apoptosis Pathway and S Phase Cell Cycle Arrest,”. Scientific World Journal (, 2014. [DOI]
- Evaluation of Antioxidant, Antimicrobial, and Antiurolithiatic Potential of Different Solvent Extracts of Aerva lanata Linn Flowers,”. Pharmacognosy Magazine, 2018. [DOI | PubMed]
- Antimicrobial, Antioxidant, Cytotoxicity and LC‐MS Analyses of Aerva javanica: An Ethnomedicinally Important Plant,”. Journal of Biological Regulators and Homeostatic Agents, 2017. [PubMed]
- A Review on Medicinal Plants With Potential Wound Healing Activity,”. Pharmacy & Pharmacology International Journal, 2012
- Evaluation of Anthelmintic Antimicrobial and Antioxidant Activity of Chenopodium album ,”. Tropical Animal Health and Production, 2017. [DOI | PubMed]
- A Review on Antidiabetic Properties of Indian Mangrove Plants with Reference to Island Ecosystem,”. Evidence‐Based Complementary and Alternative Medicine, 2019. [DOI | PubMed]
- Biological Screening of Three Selected Folklore Medicinal Plants From Pakistan,”. Pakistan Journal of Pharmaceutical Sciences, 2019. [PubMed]
- Genotoxic and Cytotoxic Potential of Alternanthera bettzickiana, an Important Ethno‐Medicinal Plant,”. Cellular and Molecular Biology, 2017. [DOI]
- Ethnopharmacological Survey and Antisickling Activity of Plants Used in the Management of Sickle Cell Disease in Kikwit City, DR Congo,”. Evidence‐Based Complementary and Alternative Medicine, 2020. [DOI | PubMed]
- Antiarthritic Potential of Comprehensively Standardized Extract of Alternanthera bettzickiana: In Vitro and In Vivo Studies,”. ACS Omega, 2020. [DOI | PubMed]
- Anti‐Arthritic and Toxicological Evaluation of Ethanolic Extract of Alternanthera bettzickiana in Rats,”. Frontiers in Pharmacology, 2022. [DOI]
- Biosynthesis, Characterization, and Evaluation of Bioactivities of Leaf Extract‐Mediated Biocompatible Gold Nanoparticles From Alternanthera bettzickiana ,”. Biotechnology Reports, 2018. [DOI]
- Phytochemical Analysis in Alternanthera brasiliana by LC–MS/MS and GC–MS,”. Natural Product Research, 2020. [DOI | PubMed]
- Photodynamic Inactivation of Yeast and Bacteria by Extracts of Alternanthera brasiliana ,”. Current Drug Targets, 2013. [DOI | PubMed]
- Anxiolytic and Anticonvulsant Activity of Methanol Extract of Leaves of Alternanthera brasiliana (L.) Kuntze (Amaranthaceae) in Laboratory Animals,”. Indian Journal of Experimental Biology, 2013. [PubMed]
- Ethnobotanical Study of Medicinal Plants by Population of Valley of Juruena Region, Legal Amazon, Mato Grosso, Brazil,”. Journal of Ethnopharmacology, 2015. [DOI | PubMed]
- Toxicity Against Drosophila melanogaster and Antiedematogenic and Antimicrobial Activities of Alternanthera brasiliana (L.) Kuntze (Amaranthaceae),”. Environmental Science and Pollution Research, 2018. [DOI | PubMed]
- Aqueous Extract of Fresh Leaves from Alternanthera brasiliana (L.) Kuntze: Chemical Evaluation and Antimycobacterial and Anticandidal Activities,”. Advances in Traditional Medicine, 2020. [DOI]
- Betalains and Phenolic Compounds of Leaves and Stems of Alternanthera brasiliana and Alternanthera tenella ,”. Food Research International, 2017. [DOI | PubMed]
- Betalains in Some Species of the Amaranthaceae Family: A Review,”. Antioxidants, 2018. [DOI]
- Evaluation of the Pharmacological Activity of the Alternanthera brasiliana Aqueous Extract,”. Pharmaceutical Biology, 2012. [DOI | PubMed]
- Screening of Medicinal Plants Traditionally Used in Peruvian Amazon for In Vitro Antioxidant and Anticancer Potential,”. Natural Product Research, 2019. [DOI | PubMed]
- Antibiotic Oxylipins from Alternanthera brasiliana and Its Endophytic Bacteria,”. Phytochemistry, 2015. [DOI | PubMed]
- Antimicrobial Activity of Alternanthera caracasana ,”. Pharmaceutical Biology, 2005. [DOI | PubMed]
- Anti‐Hyperalgesic and Anti‐Inflammatory Activity of Alternanthera maritima Extract and 2″‐O‐α‐l‐Rhamnopyranosylvitexin in Mice,”. Inflammation, 2015. [DOI | PubMed]
- Alternanthera paronychioides Protects Pancreatic β‐Cells From Glucotoxicity by Its Antioxidant, Antiapoptotic and Insulin Secretagogue Actions,”. Food Chemistry, 2013. [DOI | PubMed]
- Antitumor Constituents From Alternanthera philoxeroides ,”. Journal of Asian Natural Products Research, 2007. [DOI | PubMed]
- Cytotoxic Triterpene Saponins From Alternanthera philoxeroides ,”. Journal of Asian Natural Products Research, 2009. [DOI | PubMed]
- Chemical Constituents and Antidepressant‐Like Effects in Ovariectomized Mice of the Ethanol Extract of Alternanthera philoxeroides ,”. Molecules (Basel, Switzerland), 2018. [DOI]
- Evaluation of Antinociceptive and Antihyperglycemic Activities in Methanol Extracts of Whole Plants of Alternanthera philoxeroides (Mart.) Griseb. (Amaranthaceae) in Mice,”. Pakistan Journal of Pharmaceutical Sciences, 2012. [PubMed]
- New Anti‐HBV C‐Boivinopyranosyl Flavones From: Alternanthera philoxeroides ,”. Molecules (Basel, Switzerland), 2016. [DOI]
- Evaluation of Chikusetsusaponin IVa Isolated From Alternanthera philoxeroides for Its Potency Against Viral Replication,”. Planta Medica, 2009. [DOI | PubMed]
- Quantitation of β‐Carboline and Quercetin in Alligator Weed (Alternanthera philoxeroides (Mart.) Griseb.) by LC–MS/MS and Evaluation of Cardioprotective Effects of the Methanol Extracts,”. Drug Discoveries & Therapeutics, 2018. [DOI | PubMed]
- Medicinal Plants Used in Northern Peru for Reproductive Problems and Female Health,”. Journal of Ethnobiology and Ethnomedicine, 2010. [DOI | PubMed]
- Qualitative and Quantitative Ethnobotanical Study of the Pangkhua Community in Bilaichari Upazilla, Rangamati District, Bangladesh,”. Journal of Ethnobiology and Ethnomedicine, 2019. [DOI | PubMed]
- Exotic and Indigenous Problem Plants Species Used, by the Bapedi, to Treat Sexually Transmitted Infections in Limpopo Province, South Africa,”. African Health Sciences, 2013. [DOI | PubMed]
- Investigation of Alternanthera repens and Bidens odorata on Gastrointestinal Disease,”. Fitoterapia, 2008. [DOI | PubMed]
- Spasmolytic Effect of Alternanthera repens on Isolated Rat Ileum,”. Pharmaceutical Biology, 2014. [DOI]
- Ethnopharmacological Survey on Medicinal Plants Used in Herbal Drinks Among the Traditional Communities of Pakistan,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- Total Phenolic Content, Flavonoid Content and Antioxidant Potential of Wild Vegetables From Western Nepal,”. Plants, 2019. [DOI | PubMed]
- Effectiveness of Traditional Malaysian Vegetables (Ulam) in Modulating Blood Glucose Levels,”. Asia Pacific Journal of Clinical Nutrition, 2014. [DOI | PubMed]
- Effects of Alternanthera sessilis on Liver Function in Carbon Tetra Chloride Induced Hepatotoxicity in Wister Rat Model,”. Indian Journal of Clinical Biochemistry, 2018. [DOI | PubMed]
- Ethnobotanical Study on Medicinal Plants Used by Maonan People in China,”. Journal of Ethnobiology and Ethnomedicine, 2015. [DOI | PubMed]
- A Preliminary Evaluation of Antihyperglycemic and Analgesic Activity of Alternanthera sessilis Aerial Parts,”. BMC Complementary and Alternative Medicine, 2014. [DOI | PubMed]
- Alternanthera sessilis: Uncovering the Nutritional and Medicinal Values of an Edible Weed,”. Journal of Ethnopharmacology, 2022. [DOI | PubMed]
- Solid‐Liquid Extraction of Bioactive Compounds With Antioxidant Potential From Alternanthera sessilis (red) and Identification of the Polyphenols Using UHPLC‐QqQ‐MS/MS,”. Food Research International, 2019. [DOI | PubMed]
- Suppression of Proinflammatory Cytokines and Mediators in LPS‐Induced RAW 264.7 Macrophages by Stem Extract of Alternanthera sessilis via the Inhibition of the NF‐κB Pathway,”. Journal of Immunology Research, 2018a. [DOI]
- In Vitro Wound Healing Potential of Stem Extract of Alternanthera sessilis ,”. Evidence‐Based Complementary and Alternative Medicine (, 2018b. [DOI]
- A Survey of Herbal Weeds for Treating Skin Disorders From Southern Thailand: Songkhla and Krabi Province,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- A Survey of Herbal Weeds That Are Used to Treat Gastrointestinal Disorders From Southern Thailand: Krabi and Songkhla Provinces,”. Journal of Ethnopharmacology, 2017. [DOI | PubMed]
- Biosynthesis of Silver Nanoparticles Using Alternanthera sessilis (Linn.) Extract and Their Antimicrobial, Antioxidant Activities,”. Colloids Surfaces B Biointerfaces, 2013. [DOI | PubMed]
- Ethnobotanical Perspective of Antimalarial Plants: Traditional Knowledge Based Study,”. BMC Research Notes, 2016. [DOI | PubMed]
- Synthesis and Characterization of Gold Nanoparticles From Aqueous Leaf Extract of Alternanthera sessilis and Its Anticancer Activity on Cervical Cancer Cells (HeLa),”. Artificial Cells Nanomedicine, and Biotechnology, 2019. [DOI | PubMed]
- Ionone Derivatives From Alternanthera sessilis ,”. Journal of Asian Natural Products Research, 2002. [DOI | PubMed]
- Rationalizing Ethnopharmacological Uses of Alternanthera sessilis: A Folk Medicinal Plant of Pakistan to Manage Diarrhea, Asthma and Hypertension,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- A Review of Bioactivity Guided Medicinal Uses and Therapeutic Potentials of Noxious Weed (Alternanthera sessilis),”. International Journal of Chemical and Biochemical Sciences, 2018
- Immunomodulatory Properties of Alternanthera tenella Colla Aqueous Extracts in Mice,”. Brazilian Journal of Medical and Biological Research, 2003. [DOI | PubMed]
- Anti‐Inflammatory Properties of Ethanolic Extract and 2″‐O‐β‐d‐Glucopyranosyl‐Vitexin Obtained From Alternanthera tenella Colla Whole Plant,”. Inflammation, 2021. [DOI | PubMed]
- Ethnobotanical and Ethnopharmacological Study of Medicinal Plants Used by a Traditional Community in Brazil’s Northeastern,”. Brazilian Journal of Biology, 2021. [DOI]
- Local Knowledge About Fodder Plants in the Semi‐Arid Region of Northeastern Brazil,”. Journal of Ethnobiology and Ethnomedicine, 2015. [DOI | PubMed]
- Phyto‐Synthesis of Silver Nanoparticles Using Alternanthera tenella Leaf Extract: An Effective Inhibitor for the Migration of Human Breast Adenocarcinoma (MCF‐7) Cells,”. Bioprocess and Biosystems Engineering, 2016. [DOI | PubMed]
- Blutaparon portulacoides Ethanolic Extract Reduced IL‐1β and Inflammatory Parameters Induced by the Mycobacterium Complex and Carrageenan in Mice,”. Inflammopharmacology, 2021. [DOI | PubMed]
- Ecdysteroids and a Sucrose Phenylpropanoid Ester From Froelichia floridana ,”. Phytochemistry, 2009. [DOI | PubMed]
- A Comparison of the Bioactivity and Phytochemical Profile of Three Different Cultivars of Globe Amaranth: Red, White, and Pink,”. Food & Function, 2016. [DOI | PubMed]
- Plants Used Traditionally to Treat Malaria in Brazil: The Archives of Flora Medicinal,”. Journal of Ethnobiology and Ethnomedicine, 2007. [DOI | PubMed]
- Antimicrobial Constituents of Gomphrena martiana and Gomphrena boliviana ,”. Journal of Ethnopharmacology, 1992. [DOI | PubMed]
- Effects of Aqueous and Ethanolic Extracts of Entandrophragma angolense, Cola nitida and Gomphrena celosioides Against Doxorubicin‐Induced Cardiotoxicity in Rats,”. Journal of Advances in Medical and Pharmaceutical Sciences, 2016. [DOI]
- Anti‐Inflammatory and Antioxidant Effects of Ethanol Extract of Gomphrena celosioides (Amaranthaceae) in Wistar Rats,”. Journal of Pharmaceutical, Chemical and Biological Sciences, 2017
- Antiarthritic and Antihyperalgesic Properties of Ethanolic Extract From Gomphrena celosioides Mart. (Amaranthaceae) Aerial Parts,”. Evidence‐Based Complementary and Alternative Medicine, 2020. [DOI]
- Treatment and Management of Liver Diseases by Khmer Traditional Healers Practicing in Phnom Penh Area, Cambodia,”. Journal of Ethnopharmacology, 2017. [DOI | PubMed]
- Diuretic Herb Gomphrena celosioides Mart. (Amaranthaceae) Promotes Sustained Arterial Pressure Reduction and Protection From Cardiac Remodeling on Rats With Renovascular Hypertension,”. Journal of Ethnopharmacology, 2018. [DOI | PubMed]
- Mechanisms Underlying the Diuretic Effect of Gomphrena celosioides Mart. (Amaranthaceae),”. Journal of Ethnopharmacology, 2017. [DOI | PubMed]
- Isolation of 3‐(4‐Hydroxyphenyl) Methylpropenoate and Bioactivity Evaluation of Gomphrena celosioides Extracts,”. EXCLI Journal, 2010. [PubMed]
- Isolation of Aurantiamides from Gomphrena celosioides C. Mart,”. Iranian Journal of Pharmaceutical Research, 2014
- Hypoglycemic Activity of Isolated Compounds From Gomphrena celosioides Mart,”. Pharmaceutical Chemistry Journal, 2020. [DOI]
- Assessment of Phytochemical and Antibacterial Activities of the Leaf and Stem Extracts of Gomphrena celosioides Mart. (Amaranthaceae),”. Trends in Medical Research, 2020. [DOI]
- An Ethnobotanical Study of Plants Used to Treat Liver Diseases in the Maritime Region of Togo,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- Anti‐Inflammatory and Analgesic Effects of Aqueous Leaf Extracts of Gomphrena celosioides and Momordica Charantia ,”. Journal of Natural Sciences Engineering and Technology, 2009. [DOI]
- Gastroprotective Effect of Methanolic Extract of Gomphrena celosioides on Indomethacin Induced Gastric Ulcer in Wistar Albino Rats,”. International Journal of Applied and Basic Medical Research, 2015. [DOI | PubMed]
- Extracts of Gomphrena celosioides Mart as Potential Treatment for Urinary Tract Infections Against Antibiotic Resistant β‐Lactamase Producing Uropathogens,”. South African Journal of Botany, 2020. [DOI]
- Evaluation of the Hepatoprotective Activity of Gomphrena celosioides (Amaranthaceae) on Wistar Rats Intoxicated With Tetrachloride Carbon,”. International Journal of Current Research, 2012
- Extraction of Essential Oil From Gomphrena celosioides by Green Separation Technology,”. International Journal of Basic and Applied Biology, 2014
- Study on Water Soluble Constituents From Gomphrena celoisiodes ,”. Vietnam Journal of Chemistry, 2019. [DOI]
- An Ethnobotanical Survey of Medicinal Plants in Trinidad,”. Journal of Ethnobiology and Ethnomedicine, 2015. [DOI | PubMed]
- Structural Characterization of Phenolics and Betacyanins in Gomphrena globosa by High‐Performance Liquid Chromatography‐Diode Array Detection/Electrospray Ionization Multi‐Stage Mass Spectrometry,”. Rapid Communications in Mass Spectrometry, 2011. [DOI | PubMed]
- Ethnomedicines Used in Trinidad and Tobago for Urinary Problems and Diabetes Mellitus,”. Journal of Ethnobiology and Ethnomedicine, 2006. [DOI | PubMed]
- Enhancing the Antimicrobial and Antifungal Activities of a Coloring Extract Agent Rich in Betacyanins Obtained From: Gomphrena globosa L. Flowers,”. Royal Society of Chemistry, 2018. [DOI]
- HPLC‐Profiles of Tocopherols, Sugars, and Organic Acids in Three Medicinal Plants Consumed as Infusions,”. International Journal of Food Science, 2014. [DOI | PubMed]
- Phytochemical Investigations and Biological Potential Screening With Cellular and Non‐Cellular Models of Globe Amaranth, (Gomphrena globosa L) Inflorescences,”. Food Chemistry, 2012. [DOI | PubMed]
- Oleanane and Taraxerane Glycosides From the Roots of Gomphrena macrocephala ,”. Journal of Natural Products, 2006. [DOI | PubMed]
- Antitumor and Cytotoxic Screen of 5,6,7‐Trisubstituted Flavones From Gomphrena martiana ,”. Journal of Ethnopharmacology, 1994. [DOI | PubMed]
- Phytochemical Profile and Antiproliferative Activity of Human Lymphocytes of Gomphrena virgata Mart. (Amaranthaceae),”. Natural Product Research, 2022. [DOI | PubMed]
- Evaluación Gastroprotectora Del Extracto Hidroalcohólico De Hojas De Guilleminea densa (Hum. Bonpl. ex Willd.) Moq.(Sanguinaria) En Ulceras Inducidas Con Etanol,”. Revista De La Academia Peruana De Salud, 2010
- Antioxidant Activity, Total Phenolic, Tannin, and Flavonoid Content of Five Plants Used in Traditional Medicine in Penjamo, Guanajuato,”. Chemistry & Biodiversity, 2023. [DOI | PubMed]
- Anti‐Neuroinflammatory Effect of Iresine celosia on Lipopolysaccharide‐Stimulated Microglial Cells and Mouse,”. Biomedicine & Pharmacotherapy, 2019. [DOI | PubMed]
- 1H and 13C Assignments of Three New Drimenes From Iresine diffusa Humb. & Bonpl. Ex Willd,”. Magnetic Resonance in Chemistry, 2005. [DOI | PubMed]
- Ethnobotany of Hill Tribes of northern Thailand. I. Medicinal Plants of Akha,”. Economic Botany, 1986. [DOI]
- Review on Medicinal and Pharmacological Properties of Iresine herbstii, Chrozophora Rottleri and Ecbolium Linneanum,”. African Journal of Traditional, Complementary and Alternative Medicines, 2011. [DOI]
- Affinity of Iresine herbstii and Brugmansia arborea Extracts on Different Cerebral Receptors,”. Journal of Ethnopharmacology, 2006. [DOI | PubMed]
- Biological Studies on Brazilian Plants Used in Wound Healing,”. Journal of Ethnopharmacology, 2009. [DOI | PubMed]
- Separation of Betacyanins From Iresine herbstii Hook. Ex Lindl. Leaves by High‐Speed Countercurrent Chromatography in a Polar Solvent System,”. Journal of Chromatography A, 2020. [DOI | PubMed]
- A New Isoflavanone From Iresine herbstii ,”. Fitoterapia, 2011. [DOI | PubMed]
- In Vivo Antimutagenic Activity of the Medicinal Plants Pfaffia glomerata (Brazilian Ginseng) and Ginkgo biloba ,”. Genetics and Molecular Research, 2017. [DOI]
- Pfaffia glomerata Hydroalcoholic Extract Stimulates Penile Tissue in Adult Swiss Mice,”. Journal of Ethnopharmacology, 2020. [DOI | PubMed]
- A 20‐Hydroxyecdysone‐Enriched Fraction From Pfaffia glomerata (Spreng.) Pedersen Roots Alleviates Stress, Anxiety, and Depression in Mice,”. Journal of Ethnopharmacology, 2021. [DOI | PubMed]
- Three New Noroleanane‐Type Triterpenes From the Roots of Pfaffia glomerata ,”. Journal of Asian Natural Products Research, 2018. [DOI | PubMed]
- Psychopharmacological Assessment of Pfaffia glomerata Roots (Extract BNT‐08) in Rodents,”. Phytotherapy Research, 2004. [DOI | PubMed]
- Evaluation of the Trypanocidal and Leishmanicidal in Vitro Activity of the Crude Hydroalcoholic Extract of Pfaffia glomerata (Amarathanceae) Roots,”. Phytomedicine, 2004. [DOI | PubMed]
- Analgesic and Anti‐Inflammatory Activity of a Crude Root Extract of Pfaffia glomerata (Spreng) Pedersen,”. Journal of Ethnopharmacology, 2005. [DOI | PubMed]
- Involvement of the Nitric Oxide/Soluble Guanylate Cyclase Pathway in the Anti‐Oedematogenic Action of Pfaffia glomerata (Spreng) Pedersen in Mice,”. Journal of Pharmacy and Pharmacology, 2006. [DOI | PubMed]
- Pfaffia paniculata (Brazilian Ginseng) Extract Modulates Mapk and Mucin Pathways in Intestinal Inflammation,”. Journal of Ethnopharmacology, 2018. [DOI | PubMed]
- Pfaffosidic Fraction From Hebanthe paniculata Induces Cell Cycle Arrest and Caspase‐3‐Induced Apoptosis in HepG2 Cells,”. Evidence‐Based Complementary and Alternative Medicine, 2015. [DOI | PubMed]
- Development and Characterization of Microsatellite Markers for Hebanthe eriantha (Amaranthaceae),”. American Journal of Botany, 2011. [DOI]
- Triterpenoids From Brazilian Ginseng, Pfaffia paniculata ,”. Planta Medica, 2010. [DOI | PubMed]
- Cytotoxic Effects of Butanolic Extract From Pfaffia paniculata (Brazilian Ginseng) on Cultured Human Breast Cancer Cell Line MCF‐7,”. Experimental and Toxicologic Pathology, 2009. [DOI | PubMed]
- Pfaffosides and Nortriterpenoid Saponins From Pfaffia paniculata ,”. Phytochemistry, 1984. [DOI]
- Development of an Analytical Method for the Quantification of Pfaffic Acid in Brazilian Ginseng (Hebanthe eriantha),”. Journal of Pharmaceutical and Biomedical Analysis, 2013. [DOI | PubMed]
- Anti‐Inflammatory and Antioxidant Properties of the Extract, Tiliroside, and Patuletin 3‐O‐β‐D‐Glucopyranoside From Pfaffia townsendii (Amaranthaceae),”. Evidence‐Based Complementary and Alternative Medicine, 2018. [DOI | PubMed]
- New Drimane Sesquiterpenoids From Tidestromia oblongifolia ,”. National Institutes of Health, 2008
- Antibacterial Activity Assessment of Petroleum Ether and Methanolic Extracts of Achyranthes aspera Linn (Amaranthaceae),”. Journal of Applied and Natural Science, 2020. [DOI]
- Betalains: Potential Drugs With Versatile Phytochemistry,”. Critical ReviewsTM in Eukaryotic Gene Expression, 2020. [DOI]
- Sesquiterpenes and Their Derivatives‐Natural Anticancer Compounds: An Update,”. Pharmacological Research, 2020. [DOI]
- Subchronic Toxicity Study of Alternanthera philoxeroides in Swiss Albino Mice Having Antioxidant and Anticoagulant Activities,”. Journal of Toxicology, 2022. [DOI | PubMed]
- Central‐Stimulating and Analgesic Activity of the Ethanolic Extract of Alternanthera sessilis in Mice,”. BMC Complementary Medicine and Therapies, 2014. [DOI]
- Antispasmodic, Cardioprotective and Blood‐Pressure Lowering Properties of Gomphrena perennis L. and Its Mechanisms of Action,”. Journal of Traditional and Complementary Medicine, 2024. [DOI | PubMed]
- Antidementia Effects of Alternanthera philoxeroides in Ovariectomized Mice Supported by NMR‐based Metabolomic Analysis,”. Molecules, 2021. [DOI | PubMed]
- Evaluation of the Impact of Alternanthera philoxeroides (Mart.) Griseb. Extract on Memory Impairment in D‐Galactose‐Induced Brain Aging in Mice Through Its Effects on Antioxidant Enzymes, Neuroinflammation, and Telomere Shortening,”. Molecules (Basel, Switzerland), 2024. [DOI | PubMed]
- Flavonoid Properties in Plant Families Synthesizing Betalain Pigments (Review),”. Natural Product Communications, 2015. [DOI | PubMed]
- Extracts From Alternanthera maritima as Natural Photosensitizers in Photodynamic Antimicrobial Chemotherapy (PACT),”. Journal of Photochemistry and Photobiology B: Biology, 2010. [DOI | PubMed]
- A Metabolomic Study of Gomphrena agrestis in Brazilian Cerrado Suggests Drought‐Adaptive Strategies on Metabolism,”. Scientific Reports, 2021. [DOI | PubMed]
- Separation of Antioxidant‐Rich Alternanthera sessilis Red Extracts by Sephadex LH‐20 and Identification of Polyphenols Using HPLC‐QToF‐MS/MS,”. Chemistry and Biodiversity, 2023. [DOI | PubMed]
- Isolation and Characterisation of Methanol‐Soluble Fraction of Alternanthera philoxeroides (Mart.)—Evaluation of Their Antioxidant, α‐Glucosidase Inhibitory and Antimicrobial Activity in In Vitro Systems,”. Natural Product Research, 2014. [DOI | PubMed]
- Bioactive Chemical Constituents and Comparative Antimicrobial Activity of Callus Culture and Adult Plant Extracts From Alternanthera tenella,‖ Zeitschrift Fur Naturforschung”. Section C Journal of Biosciences, 2009. [DOI]
- Isolation and HPLC Quantitative Analysis of Antioxidant Flavonoids From Alternanthera tenella Colla,”. Zeitschrift Fur Naturforschung—Section C Journal of Biosciences, 2006. [DOI]
- Phytochemical and Antibacterial Constituents of Edible Globe Amaranth Flower Against Pseudomonas aeruginosa,”. Chemistry & Biodiversity, 2022. [DOI | PubMed]
- Ethanolic Crude Extract and Flavonoids Isolated From Alternanthera Maritima: Neutrophil Chemiluminescence Inhibition and Free Radical Scavenging Activity,”. Zeitschrift Fur Naturforschung—Section C Journal of Biosciences, 2007. [DOI]
- Isolation and Characterization of Quercetin From Amaranthaceae Family Plants,”. International Journal of Ayurveda and Pharma Research, 2017. [DOI]
- Rapid‐Resolution HPLC With Spectrometric Detection for the Determination and Identification of Isoflavones in Soy Preparations and Plant Extracts,”. Analytical and Bioanalytical Chemistry, 2007. [DOI | PubMed]
- A Methylenedioxyflavonol From Aerial Parts of Blutaparon portulacoides,”. Phytochemistry, 2000. [DOI | PubMed]
- Protective Effects of Alternanthera sessilis Ethanolic Extract Against TNF‐ α or H2O2‐Induced Endothelial Activation in Human Aortic Endothelial Cells,”. Evidence‐Based Complementary and Alternative Medicine, 2022. [DOI | PubMed]
- Scientific Validation of Synergistic Antioxidant Effects in Commercialised Mixtures of Cymbopogon Citratus and Pterospartum Tridentatum or Gomphrena globosa for Infusions Preparation,”. Food Chemistry, 2015. [DOI | PubMed]
- Bioactivity of Crude Extracts and Some Constituents of Blutaparon portulacoides (Amaranthaceae),”. Phytomedicine, 2002. [DOI | PubMed]
- The NO‐cGMP‐K +Channel Pathway Participates in Diuretic and Cardioprotective Effects of Blutaparon portulacoides in Spontaneously Hypertensive Rats,”. Planta Medica, 2022. [DOI | PubMed]
- Antioxidant and Cytoprotective Properties of Plant Extract From Dry Flowers as Functional Dyes for Cosmetic Products,”. Molecules (Basel, Switzerland), 2021. [DOI]
- Phytochemical Analysis of Pfaffia glomerata Inflorescences by LC‐ESI‐MS/MS,”. Molecules (Basel, Switzerland), 2014. [DOI | PubMed]
- First Isolation of a Symmetrical Glycosylated Methylene Bisflavonoid,”. Planta Medica, 2003. [DOI | PubMed]
- Estudo fitoquímico e atividades biológicas de Gomphrena elegans Mart. (amaranthaceae),” Thesis (doctorate),. 2010
- Antimicrobial Screening and Quantitative Determination of Benzoic Acid Derivative of Gomphrena celosioides by TLC‐Densitometry,”. Chemical and Pharmaceutical Bulletin, 2004. [DOI | PubMed]
- Phyto‐Fabrication and Characterization of Alternanthera sessilis Leaf Extract–Mediated Silver Nanoparticles and Evaluation of Larvicidal Potential,”. Biomass Conversion and Biorefinery, 2023. [DOI]
- Involvement of Nitric Oxide in the Gastroprotective Effects of an Aqueous Extract of Pfaffia glomerata (Spreng) Pedersen, Amaranthaceae, in Rats,”. Life Sciences, 2004. [DOI | PubMed]
- Two New Triterpenes From the Roots of Pfaffia glomerata,”. Journal of Asian Natural Products Research, 2019. [DOI | PubMed]
- Brazilian Natural Medicines. IV. New Noroleanane‐Type Triterpene and Ecdysterone‐Type Sterol Glycosides and Melanogenesis Inhibitors From the Roots of Pfaffia glomerata,”. Chemical and Pharmaceutical Bulletin, 2010. [DOI | PubMed]
- Phytochemical Composition of Aerial Parts and Roots of Pfaffia glomerata (Spreng.) Pedersen and Anticholinesterase, Antioxidant, and Antiglycation Activities,”. Protoplasma, 2024. [DOI | PubMed]
- Extract of Pfaffia glomerata Ameliorates Paroxetine‐Induced Sexual Dysfunction in Male Mice and the Characterization of Its Phytoconstituents by UPLC‐MS,”. Foods, 2023. [DOI | PubMed]
- Pfaffosides, Nortriterpenoid Saponins, From Pfaffia paniculata,”. Phytochemistry, 1984. [DOI]
- Celosiadines A and B, Unusual Guanidine Alkaloids From Iresine diffusa,”. Natural Product Research, 2021. [DOI]
- Identification and Distribution of Simple and Acylated Betacyanins in the Amaranthaceae,”. Journal of Agricultural and Food Chemistry, 2001. [DOI | PubMed]
- Characterisation of Betalain Patterns of Differently Coloured Inflorescences From Gomphrena globosa L. and Bougainvillea Sp. by HPLC‐DAD‐ESI‐MSn,”. Analytical and Bioanalytical Chemistry, 2007. [DOI | PubMed]
- Separation of Betacyanins From Purple Flowers of Gomphrena globosa L. by Ion‐Pair High‐Speed Counter‐Current Chromatography,”. Journal of Chromatography A, 2017. [DOI | PubMed]
- Separation of Amaranthine‐Type Betacyanins by Ion‐Pair High‐Speed Countercurrent Chromatography,”. Journal of Chromatography A, 2014. [DOI | PubMed]
- High‐Speed Counter‐Current Chromatography in Separation of Betacyanins From Flowers of Red Gomphrena globosa L. Cultivars,”. Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences, 2016. [DOI]
- Studies on Polar High‐Speed Counter‐Current Chromatographic Systems in Separation of Amaranthine‐Type Betacyanins From Celosia Species,”. Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences, 2018. [DOI | PubMed]
- Floral Parts of Gomphrena globosa L. as a Novel Alternative Source of Betacyanins: Optimization of the Extraction Using Response Surface Methodology,”. Food Chemistry, 2017. [DOI | PubMed]
- Liquid Chromatographic Techniques in Betacyanin Isomers Separation From Gomphrena globosa L. flowers for the Determination of Their Antimicrobial Activities,”. Journal of Pharmaceutical and Biomedical Analysis, 2018. [DOI | PubMed]
- Betacyanins From Gomphrena globosa L. Flowers: Incorporation in Cookies as Natural Colouring Agents,”. Food Chemistry (, 2020. [DOI]
- Phytochemical Molecules From the Decarboxylation of Gomphrenins in Violet Gomphrena globosa L.—Floral Infusions From Functional Food,”. International Journal of Molecular Sciences, 2020. [DOI]
- Rapid Identification of Betacyanins From Amaranthus tricolor, Gomphrena globosa, and Hylocereus polyrhizus by Matrix‐Assisted Laser Desorption/Ionization Quadrupole Ion Trap Time‐of‐Flight Mass Spectrometry (MALDI‐QIT‐TOF MS),”. Journal of Agricultural and Food Chemistry, 2006. [DOI | PubMed]
- In Vitro Activities of Pfaffia Glomerata Root Extract, Its Hydrolyzed Fractions and Pfaffic Acid Against Trypanosoma Cruzi Trypomastigotes,”. Chemistry & Biodiversity, 2017. [DOI]
- Xanthine Oxidase and Nitric Oxide Inhibitory Activities of Compounds Isolated From Gomphrena celosioides,”. Vietnam Journal of Chemistry, 2024. [DOI]
- β‐Ecdysone Content and Antioxidant Capacity in Different Organs of Brazilian Ginsen,”. Ciência Rural (, 2021. [DOI]
- Pfaffia glomerata Polyploid Accession Compromises Male Fertility and Fetal Development,”. Journal of Ethnopharmacology (, 2023. [DOI]
- Exposure to Pfaffia glomerata Causes Oxidative Stress and Triggers Hepatic Changes,”. Brazilian Journal of Biology (, 2023. [DOI]
- 2‐Dehydro‐3‐Epi‐20‐Hydroxyecdysone From Froelichia florida,”. Phytochemistry, 1998. [DOI]
- Pfaffia paniculata (Brazilian ginseng) Methanolic Extract Reduces Angiogenesis in Mice,”. Experimental and Toxicologic Pathology, 2007. [DOI | PubMed]
- Apoptotic Efficacy of Biogenic Silver Nanoparticles on Human Breast Cancer MCF‐7 Cell Lines,”. Progress in Biomaterials, 2015. [DOI | PubMed]
- Evaluation of Antitumor Activity and Antioxidant Status of Alternanthera brasiliana Against Ehrlich Ascites Carcinoma in Swiss Albino Mice,”. Pharmacognosy Research, 2015. [DOI | PubMed]
- Chemical Structure and Localization of LEVAN, the Predominant Fructan Type in Underground Systems of Gomphrena marginata (Amaranthaceae),”. Frontiers in Plant Science, 2018. [DOI | PubMed]
- Compuestos Fenólicos,”. Offarm: farmacia y sociedad, 2004
- Antioxidant/Antidegradative Effects of Trichopus zeylanicus Gaertn and Gomphrena celosioides on Den/Hcb Induced Male Albino Rats,”. Journal of Medical Science And Clinical Research, 2013
- Anti‐Arthritic Properties of Crude Extract From Chenopodium ambrosioides L. Leaves,”. Journal of Pharmacy and Pharmacology, 2018. [PubMed]
- Natural Flavonoids as Promising Analgesic Candidates: A Systematic Review,”. Chemistry & Biodiversity, 2016. [DOI | PubMed]
- Monoterpenes With Analgesic Activity—A Systematic Review,”. Phytotherapy Research, 2013. [DOI | PubMed]
- Effect of Chenopodium Ambrosioides on the Healing Process of the In Vivo Bone Tissue,”. Microscopy Research and Technique, 2017. [DOI | PubMed]
- Anti‐Inflammatory and Antioxidant Properties of Alternanthera brasiliana Improve Cutaneous Wound Healing in Rats,”. Inflammopharmacology, 2021. [DOI | PubMed]
- Immunomodulation of RAW 264.7 Murine Macrophage Functions and Antioxidant Activities of 11 Plant Extracts,”. Cellular & Molecular Immunology, 2015. [DOI]
- Mitigation of Starch and Glucose ‑ Induced Postprandial Glycemic Excursion in Rats by Antioxidant ‑ Rich Green ‑ Leafy Vegetables ‘ Juice,”. Pharmacognosy Magazine, 2013. [DOI | PubMed]
- Alpha‐Glucosidase Inhibitory and Antioxidant Potential of Antidiabetic Herb Alternanthera sessilis: Comparative Analyses of Leaf and Callus Solvent Fractions,”. Pharmacognosy Magazine, 2016. [DOI | PubMed]
- Hydration of Sickle Erythrocytes Using a Herbal Extract (Pfaffia paniculata) In Vitro,”. British Journal of Haematology, 2000. [DOI | PubMed]
- Pfaffia paniculata Extract Improves Red Blood Cell Deformability in Sickle Cell Patients,”. Clinical Hemorheology and Microcirculation, 2016. [DOI]
- Ethanolic Extract of Alternanthera sessilis (AS‐1) Inhibits IgE‐Mediated Allergic Response in RBL‐2H3 Cells,”. Immunological Investigations, 2013. [DOI | PubMed]
- Inhibition of Lymphocyte Activation by Extracts and Fractions of Kalanchoe, Alternanthera, Paullinia and Mikania Species,”. Phytomedicine, 1994. [DOI | PubMed]
- In Vitro Binding Receptors Study by Valeriana Adscendens, Iresine herbstii and Brugmansia Arborea Extracts,”. Journal of Medicinal Chemistry, 2007. [DOI]
- Wound Healing Activity of Methanolic Extract of Leaves of Alternanthera brasiliana Kuntz Using In Vivo and In Vitro Model,”. Indian Journal of Experimental Biology, 2009. [PubMed]
- Evaluation of the Testis Function of Mice Exposed in Utero and During Lactation to Pfaffia glomerata (Brazilian Ginseng),”. Andrologia, 2019. [DOI]
- A Preliminary Evaluation of Antihyperglycemic and Analgesic Activity of Alternanthera sessilis Aerial Parts,”. BMC Complementary Medicine and Therapies, 2014. [DOI]
- Involvement of Glutamate and Cytokine Pathways on Antinociceptive Effect of Pfaffia glomerata in Mice,”. Journal of Ethnopharmacology, 2009. [DOI | PubMed]
- Anticarcinogenic Effects of Some Indian Plant Products,”. Food and Chemical Toxicology, 1992. [DOI | PubMed]
- Effect of Pfaffia paniculata (Brazilian Ginseng) on the Ehrlich Tumor in Its Ascitic Form,”. Life Sciences, 2003. [DOI | PubMed]
- Inhibitory Effects of Pfaffia paniculata (Brazilian Ginseng) on Preneoplastic and Neoplastic Lesions in a Mouse Hepatocarcinogenesis Model,”. Cancer Letters, 2005. [DOI | PubMed]
- Antineoplastic Effects of Butanolic Residue of Pfaffia paniculata,”. Cancer Letters, 2006. [DOI | PubMed]
- Effects of Pfaffia paniculata (Brazilian Ginseng) Extract on Macrophage Activity,”. Life Sciences, 2006. [DOI | PubMed]
- Pfaffia paniculata (Brazilian Ginseng) Roots Decrease Proliferation and Increase Apoptosis but Do Not Affect Cell Communication in Murine Hepatocarcinogenesis,”. Experimental and Toxicologic Pathology, 2010. [DOI | PubMed]
- Alternanthera sessilis Red Ethyl Acetate Fraction Exhibits Antidiabetic Potential on Obese Type 2 Diabetic Rats,”. Evidence‐Based Complementary and Alternative Medicine, 2013. [DOI | PubMed]
- Evaluation of Immunomodulatory and Anti‐Inflammatory Effects and Phytochemical Screening of Alternanthera tenella Colla (Amaranthaceae) Aqueous Extracts,”. Memórias do Instituto Oswaldo Cruz, 2008. [DOI | PubMed]
- Anti‐Inflammatory Activity of Blutaparon portulacoides Ethanolic Extract Against the Inflammatory Reaction Induced by Bothrops jararacussu Venom and Isolated Myotoxins BthTX‐I and II,”. Journal of Venomous Animals and Toxins Including Tropical Diseases, 2009. [DOI]
- Anti‐Inflammatory Effects of Brazilian Ginseng (Pfaffia paniculata) on TNBS‐Induced Intestinal Inflammation: Experimental Evidence,”. International Immunopharmacology, 2015. [DOI | PubMed]
- Antidiarrhoeal Activity of Waltheria americana, Commelina coelestis and Alternanthera repens,”. Journal of Ethnopharmacology, 1998. [DOI | PubMed]
- Protective Role of Methanolic Extract of Gomphrena celosioides Leaves on Acidified Ethanol‐Induced Gastric Ulcer in Male Wistar Rats,”. International Journal of Women in Technical Education and Employment, 2020
- Gomphrena globosa L. Extract Alleviates Carbon Tetrachoride‐Induced Liver Injury in Mice by Activating Antioxidant Signaling Pathways and Promoting Autophagy,”. Molecular Biology Reports, 2023. [DOI | PubMed]
- Psychopharmacological Screening of Pfaffia glomerata Spreng. (Amaranthaceae) In Rodents,”. Journal of Ethnopharmacology, 2000. [DOI | PubMed]
- Effect of Alternanthera brasiliana (L) Kuntze on Healing of Dermal Burn Wound,”. Indian Journal of Experimental Biology, 2012. [PubMed]
- Influence of Alternanthera brasiliana (L.) Kuntze on Altered Antioxidant Enzyme Profile During Cutaneous Wound Healing in Immunocompromised Rats,”. ISRN Pharmacology, 2012. [DOI | PubMed]
- Healing Efficacy of Methanol Extract of Leaves of Alternanthera brasiliana Kuntze in Aged Wound Model,”. Journal of Basic and Clinical Pharmacy, 2012. [DOI | PubMed]
- Antibacterial Activity of a Novel Fatty Acid (14E, 18E, 22E, 26E)‐Methyl Nonacosa‐14, 18, 22, 26 Tetraenoate Isolated From Amaranthus spinosus,”. Pharmaceutical Biology, 2016. [DOI | PubMed]
- Chemical Composition, Antimicrobial, Modulator and Antioxidant Activity of Essential Oil of Dysphania ambrosioides (L.) Mosyakin & Clemants,”. Comparative Immunology, Microbiology and Infectious Diseases, 2019. [DOI | PubMed]
- Green Synthesis of Silver Nanoparticles Using Alternanthera dentata Leaf Extract at Room Temperature and Their Antimicrobial Activity,”. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2014. [DOI | PubMed]
- Anti‐Bacterial Activity and Brine Shrimp Lethality Bioassay of Methanolic Extracts of Fourteen Different Edible Vegetables From Bangladesh,”. Asian Pacific Journal of Tropical Biomedicine, 2013. [DOI | PubMed]
- Biomedical and Textile Applications of Alternanthera sessilis Leaf Extract Mediated Synthesis of Colloidal Silver Nanoparticle,”. Nanomaterials, 2022. [DOI]
- Phyto‐Fabricated ZnO Nanoparticles for Anticancer, Photo‐Antimicrobial Effect on Carbapenem‐Resistant/Sensitive Pseudomonas aeruginosa and Removal of Tetracycline,”. Bioprocess and Biosystems Engineering, 2024. [DOI | PubMed]
- The Antibacterial Efficacy of Gold Nanoparticles Derived From Gomphrena celosioides and Prunus amygdalus (Almond) Leaves on Selected Bacterial Pathogens,”. International Journal of Innovative Research and Scientific Studies, 2014
- Antiproliferative Assay of Suma or Brazilian Ginseng (Hebanthe eriantha) Methanolic Extract on HCT116 and 4T1 Cancer Cell Lines, In Vitro Toxicity on Artemia salina Larvae, and Antibacterial Activity,”. Natural Product Research, 2024. [DOI | PubMed]
- The Green Synthesis, Characterization and Evaluation of the Biological Activities of Silver Nanoparticles Synthesized From Iresine herbstii Leaf Aqueous Extracts,”. Colloids Surfaces B Biointerfaces, 2012. [DOI | PubMed]
- Antifungal Activity of Extracts of Some Plants Used in Brazilian Traditional Medicine Against the Pathogenic Fungus Paracoccidioides brasiliensis,”. Pharmaceutical Biology, 2010. [DOI | PubMed]
- Antimicrobial Action of Four Herbal Plants Over Mixed‐Species Biofilms of Candida albicans With Four Different Microorganisms,”. Australian Endodontic Journal, 2022. [DOI | PubMed]
- Alexandre‐Moreira, M.S. Antileishmanial Activity of Medicinal Plants Used in Endemic Areas in Northeastern Brazil,”. Evidence‐Based Complementary and Alternative Medicine, 2014. [DOI | PubMed]
- Antioxidant Activities and Triterpenoids Isolated From Alternanthera brasiliana (L.) Kuntze Leaves,”. Natural Product Research, 2013. [DOI | PubMed]
- Alternanthera brasiliana L. Extract Alleviates Carbon Tetrachloride‐Induced Liver Injury and Fibrotic Changes in Mice: Role of Matrix Metalloproteinases and TGF‐β/Smad Axis,”. Journal of Ethnopharmacology, 2023. [DOI | PubMed]
- Antioxidant Capacity and Total Phenolic Contents in Hydrophilic Extracts of Selected Bangladeshi Medicinal Plants,”. Asian Pacific Journal of Tropical Medicine, 2014. [DOI]
- Phytochemical Study and In Vitro Biological Activities of Hibiscus panduriformis Burm. F. (Malvaceae), Alternanthera pungens Kunth (Amaranthaceae), and Wissadula Rostrata (Schumach.) Hook. F. (Malvaceae),”. BioMed Research International, 2023. [DOI | PubMed]
- Evaluation of Anticancer Potential of Thai Medicinal Herb Extracts Against Cholangiocarcinoma Cell Lines,”. PLoS ONE, 2019. [DOI]
- Antiproliferative Effect of Amaranth Proteins and Peptides on HT‐29 Human Colon Tumor Cell Line,”. Plant Foods for Human Nutrition, 2019. [DOI | PubMed]
- In Vitro Cytotoxic Activity of Brazilian Middle West Plant Extracts,”. Revista Brasileira De Farmacognosia, 2011. [DOI]
- Cytotoxic Activity of Selected Nigerian Plants,”. African Journal of Traditional, Complementary and Alternative Medicines, 2009. [DOI]
- Screening of Promising Chemotherapeutic Candidates From Plants Against Human Adult T‐Cell Leukemia/Lymphoma (III),”. Journal of Natural Medicines, 2013. [DOI | PubMed]
- Cytotoxicity Screening of Bangladeshi Medicinal Plant Extracts on Pancreatic Cancer Cells,”. BMC Complementary Medicine and Therapies, 2010. [DOI]
- Biosynthesis of Silver Nanoparticles Using the Extract of Alternanthera sessilis‐Antiproliferative Effect Against Prostate Cancer Cells,”. Cancer Nanotechnology, 2013. [DOI | PubMed]
- Facile Synthesis of Anisotropic Gold Nanoparticles and Its Synergistic Effect on Breast Cancer Cell Lines,”. IET Nanobiotechnology, 2020. [DOI | PubMed]
- Screening of Promising Chemotherapeutic Candidates From Plants Extracts,”. Journal of Natural Medicines, 2016. [DOI | PubMed]
- Plants With Central Analgesic Activity,”. Phytomedicine, 2001. [DOI | PubMed]
- Enhanced Analgesic Activity by Cyclodextrins—A Systematic Review and Meta‐Analysis,”. Expert Opinion on Drug Delivery, 2015. [DOI | PubMed]
- Anti‐Inflammatory Effect of Salsola komarovii Extract With Dissociated Glucocorticoid Activity,”. BMC Complementary Medicine and Therapies, 2020. [DOI | PubMed]
- Phytochemistry and Hepatoprotective Activity of Aqueous Extract of Amaranthus tricolor Linn. Roots,”. Journal of Ayurveda and Integrative Medicine, 2013. [DOI | PubMed]
- Protective Effects of Aqueous Extract of Hammada scoparia Against Hepatotoxicity Induced by Ethanol in the Rat,”. Toxicology and Industrial Health, 2012. [DOI]
- Absence of Maternal‐Fetal Adverse Effects of Alternanthera littoralis P. Beauv. Following Treatment During Pregnancy in Mice,”. Journal of Toxicology and Environmental Health, Part A, 2023. [DOI | PubMed]
- Preclinical Safety Evaluation of the Ethanolic Extract From the Aerial Parts of Gomphrena celosioides Mart. in Rodents,”. Regulatory Toxicology and Pharmacology, 2022. [DOI]
- The Ethanolic Extract of Gomphrena celosioides Mart. Does Not Alter Reproductive Performance or Embryo‐Fetal Development, Nor Does It Cause Chromosomal Damage,”. Pharmaceutics, 2022. [DOI]
