Effect of Quantitative Structural Properties and Drug Formulation in Four Cannabinoids (Cannabidiol, Cannabigerol, Cannabichromene, and Cannabinol) on Their Lymphatic Transport after Enteral Administration in Rats
Abstract
The effect of quantitative structural properties of drugs on the extent of lymphatic transport is not well understood. Our study aimed to describe these principles in four cannabinoids, cannabidiol (CBD), cannabigerol (CBG), cannabichromene (CBC), and cannabinol (CBN) administered as oil solutions and nanoemulsions. A series of studies in jugular vein cannulated rats and anesthetized mesenteric lymph duct cannulated rats was conducted to measure drug oral bioavailability and lymphatic transport. Log P was measured, and quantitative structural properties were correlated to the extent of lymphatic absorption. Nanoemulsion did not increase the absolute bioavailability via lymph in CBD but led to an 8-fold increase in CBG and a 3-fold increase in CBC and CBN. There was an even higher increase in the absolute bioavailability via portal vein (11-fold for CBD, 71-fold for CBG, 8-fold for CBC, and 13-fold for CBN). Relative bioavailability via lymph increased with decreasing smallest orthogonal molecular size and topological polar surface area. Nanoemulsion did not affect the total oral bioavailability but led to an increased absorption into portal blood. Intestinal lymphatic transport plays a major role in the absorption of CBD, CBG, CBC, and CBN. Planarity of the molecule and low surface polarity could be crucial structural features facilitating lymphatic transport.
Article type: Research Article
Keywords: cannabinoids, pharmacokinetics, oral bioavailability, lymphatic transport, lymph-targeting, nanoemulsion, quantitative structure−activity
relationship (QSAR)
Affiliations: a Institute of Pharmacology, 112302First Faculty of Medicine and General University Hospital in Prague, Charles University, Albertov 4, 128 00 Prague, Czech Republic; b Department of Chemical Engineering, Faculty of Chemical Engineering, University of Chemistry and Technology, Technická 3, 166 28 Prague, Czech Republic; c Department of Analytical Chemistry, Faculty of Science, Charles University, Hlavova 2030/8, 128 43, Prague, Czech Republic; d Institute of Physics of the Czech Academy of Sciences, Na Slovance 2, 182 00 Prague, Czech Republic
License: © 2025 The Authors. Published by American Chemical Society CC BY 4.0 This article is licensed under CC-BY 4.0
Article links: DOI: 10.1021/acs.molpharmaceut.4c01357 | PubMed: 40611782 | PMC: PMC12326358
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (208 KB)
Introduction
Lymphatic transport of drugs after oral administration plays an important role in the absorption of highly lipophilic compounds with log P > 5.ref1−ref2ref3ref4 After solubilization in the gastrointestinal tract, these compounds are absorbed into enterocytes, the intestinal epithelial cells, where they are incorporated into chylomicrons. Chylomicrons are large lipoprotein particles produced specifically in the intestines that are naturally involved in the transport of highly lipophilic molecules. Their size/diameter is too large to pass into the intestinal blood capillaries. Therefore, they take an alternative route via the intestinal lymphatic system. Chylomicrons enter the lacteals (lymph capillaries) and are transported through a network of lymphatic vessels and mesenteric lymph nodes until they reach the thoracic duct, where intestinal lymph mixes with lymph from the lower parts of the body. All the lymph eventually enters the systemic blood circulation at the confluence of the thoracic duct with the jugular and subclavian veins.
The transport through the intestinal lymphatic system can have a major impact on the drug pharmacokinetics. It can increase the absolute oral bioavailability because the lymph represents an additional gateway into the systemic circulation besides the standard transport through the portal vein. Another mechanism for increasing oral bioavailability is by avoiding first-pass metabolism in the liver. Mesenteric lymph is a dominant (sometimes even exclusive) source of systemically available drug for compounds with a high extraction ratio in the liver.ref5,ref6 Lymphatic transport can improve the efficacy of drugs that act against components of the lymphatic system, such as immunosuppressants, anti-HIV and anticancer drugs by reaching high exposure in the mesenteric lymphatic system.ref7,ref8
Given the possible advantages of targeting drugs into the mesenteric lymph, various approaches have been tested to increase lymphatic transport. However, summarizing all the available literature on in vivo lymphatic transport data, it seems that different strategies must be implemented based on the drug’s lipophilicity. In highly lipophilic compounds (log P > 5), it may be sufficient to use a lipid-based drug formulation (oil solution, o/w emulsion, self-nanoemulsifying system, etc.), because lipids (especially long-chain triglycerides) promote the physiological assembly of chylomicrons in which the drugs are dissolved and transported through the lymph.ref9,ref10 Targeting drugs with lower lipophilicity (log P < 5) into the mesenteric lymph is much more challenging. The only functional technique discovered so far is a synthesis of a lipophilic prodrug by covalently binding a lipid residue to the original drug molecule. Such complex molecule has log P > 5 and is effectively transported into the lymph. Examples of drugs successfully targeted into the lymph despite their insufficient lipophilicity are e.g. mycophenolate, valproate, and paracetamol.ref11−ref12ref13 There are few reports on targeting low-lipophilicity drugs into the lymph without lipophilic prodrug synthesis. However, the extent of the lymphatic transport for these compounds has been investigated using a noninvasive lymphatic transport measurement, which has been shown to significantly overestimate the results.ref14−ref15ref16
Cannabinoids are an important group of compounds found naturally in cannabis or produced synthetically and frequently used as registered medicines, nutritional supplements or abused as recreational drugs. They are lipophilic, mostly with log P > 5, and are therefore suitable model drugs for lymphatic transport driven absorption. The most frequently investigated cannabinoids are THC (tetrahydrocannabinol), the main psychoactive compound in cannabis, and CBD (cannabidiol), the second most abundant but nonpsychoactive cannabinoid. Besides these two, there is a variety of “minor” cannabinoids with much less pharmacological data available. In this work, we focus on four nonpsychoactive compounds: the “major” cannabinoid CBD, and three “minor” cannabinoids CBG (cannabigerol), CBC (cannabichromene), and CBN (cannabinol). Their structures are shown in Figure .
CBD has been registered for the treatment of specific forms of childhood epilepsy. It is administered orally in a formulation based on sesame oil solution (Epidyolex). Besides that, CBD, together with THC, are the active components of authorized cannabis extract registered for the treatment of spasticity in patients with multiple sclerosis (Sativex). CBD has a very limited oral bioavailability, estimated at <10% in man.ref. ref17 When taken with food, the bioavailability increases up to 4-fold.ref. ref18 Preclinical pharmacokinetic data are consistent with the human observations: CBD had a bioavailability of 8% and 22% in rats when administered in a lipid-free formulation and in a long-chain triglycerides-based formulation, respectively.ref. ref19
CBG is a much less known cannabinoid compared to CBD. There are no registered drug products containing this compound worldwide. Nevertheless, it is marketed as a nutritional supplement and various pharmacological effects have been reported in preclinical studies. CBG possesses antioxidant, anti-inflammatory, and antitumoral activities, and has antianxiety, neuroprotective and appetite-stimulating effects.ref. ref20 Absolute oral bioavailability is not known. When administered to dogs, CBG was rapidly absorbed with an average Tmax of 0.75 h.ref. ref21 When coadministered with food, the speed and extent of absorption did not change significantly.
CBC has shown an anti-inflammatory and antihypertensive effect in preclinical studies.ref22,ref23 After oral administration to man, it absorbs with moderate speed (Tmax 2–4 h). Absolute oral bioavailability is not known.ref. ref24
CBN was successfully tested in the treatment of dermatological disease epidermolysis bulosa.ref. ref25 After oral administration to rats, it was absorbed with Tmax of 1.5 to 3 h.ref. ref26 Absolute oral bioavailability was not determined.
One way of increasing the bioavailability of lipophilic compounds with problematic dissolution in the gastrointestinal tract is the development of formulations where the active substance is dispersed in small lipid droplets (microemulsions, nanoemulsions, self-emulsifying systems etc.). This method was successfully used e.g. in ivacaftor, where the bioavailability was increased 7-fold in Beagle dogs and previously present positive food effect was eliminated.ref. ref27 There are discussions, whether this bioavailability increase is solely due to better solubilization of the active substance in the gastrointestinal tract, or if there are some additional mechanisms like e.g. involvement of the intestinal lymphatic transport. Unfortunately, there are only very few studies comparing the lymphatic transport of one particular compound administered in a basic formulation (aqueous or oil solution) and in an advanced formulation containing a solubilized drug.ref. ref2 In our recently published preliminary data, the CBD oral bioavailability did increase mainly due to increased nonlymphatic transport when administered as a microemulsion compared to a simple oil solution.ref. ref28
There are specific physicochemical properties and molecular structure characteristics that help the drugs partition into the lymph after intestinal absorption. The role of log P has been postulated already several decades ago by Charman and Stella.ref. ref1 The same authors also discussed the importance of compound solubility in long-chain triglycerides. However, at least one molecule was later found (penclomedine) with a very limited partitioning into lymph despite high lipophilicity (log P > 5) and high solubility in long-chain triglycerides (>50 mg/g).ref. ref29 Moreover, log P was absent in the mathematical model developed by Holm and Hoest predicting precise lymphatic transport of drugs that was based on in vivo data from 19 molecules.ref. ref30 Instead, factors such as the size of the hydrophilic area in the molecule and hydrophilic–lipophilic ratio did play a role in this model. It is therefore evident that no single physicochemical property or structural pattern can be used to precisely predict the extent of lymphatic transport and the result is formed by an interplay of more variables. Unfortunately, there is a lack of experimental data investigating molecular structure characteristics like molecular size, molecular shape, and surface polarity on the extent of lymphatic transport.
The aim of this study was to evaluate whether an advanced drug formulation (a nanoemulsion) increases bioavailability via portal blood or via lymphatic transport and to assess the importance of quantitative structural properties of the molecule for the extent of lymphatic transport. To achieve these aims, we investigated four similar compounds CBD, CBG, CBC and CBN and their quantitative structure–activity relationship (QSAR) with lymphatic transport after oral dosing in a simple formulation (oil solution) and a nanoemulsion.
Materials and Methods
Drug Formulations
In the pharmacokinetic and lymphatic transport studies, the cannabinoids CBD, CBG, CBC, CBN (purchased from Pharmabinoid, Netherlands) were administered in sunflower oil and nanoemulsion. The required amount of cannabinoid was dissolved in sunflower oil of pharmaceutical quality (Fagron, Czech Republic) to obtain a reference formulation. The composition of the nanoemulsion formulation was optimized. In brief, a three-component system was selectedpropylene glycol monocaprylate (PGMC; Gattefosse’, France), Kolliphor EL (CR-EL; BASF, Germany) and the third component acting as a cosurfactant and solubilizer was subject to optimization. Diethylene glycol monoethyl ether (TRSC; Gattefosse’, France), ethyl alcohol – 99.8% (ETOH; Penta, Czech Republic) or propylene glycol – 99.5% (PRGL; Penta, Czech Republic) were tested as cosurfactant and solubilizer. According to the optimization results and literature research, TRSC was chosen for further in vivo experiments. Further information regarding optimization is shown in the Supporting Information. The nanoemulsion formulation used in the pharmacokinetic study for both the oral and the intravenous administration was prepared as follows. Cannabinoid was dissolved in the oil phase (50 wt % of CR-EL, 30 wt % of TRSC and 20 wt % of PGMC). Next, four parts of water with respect to one part of oil (by weight) were added dropwise to the oil mixture at mild stirring. The final formulation was kept out of light and stored in the fridge before application.
Animals
All animal experiments were performed with approval from the Ministry of Education, Youth and Sports, Czech Republic (MSMT-26838/2021-4). All efforts were made to minimize animal suffering. Male Wistar rats (weight 300–450 g, age 3–5 months) were purchased from Velaz s. r. o., Prague, Czech Republic. They were housed under standard conditions (12-h light/dark cycle, 22 °C temperature, and 50% humidity) in cages with wood shavings bedding (two rats per cage during acclimation, one rat per cage during experiment) and fed on water and granulated diet ad libitum. The acclimation period took at least 1 week. The animals were randomly assigned to experimental groups.
Pharmacokinetic Studies
Absolute oral bioavailability and other pharmacokinetic parameters were determined in a series of two-period, crossover studies, where the cannabinoids were administered intravenously in the first period, and orally in the second. Both jugular veins were cannulated (3 Fr polyurethane catheter, Instech Laboratories, Plymouth Meeting, USA). One catheter was used for intravenous dosing, and the second one was used for repetitive blood sampling in order to avoid sampling cannula contamination with highly concentrated intravenous drug solution. The anesthesia was performed using a rapid inhalational induction with isoflurane (IsoFlo, Zoetis, Czech Republic) followed by intramuscular injection of ketamine 100 mg/kg (Narkamon, Bioveta a.s., Czech Republic) and xylazine 5 mg/kg (Rometar, Bioveta a.s., Czech Republic). After a three-day recovery period, the rats were intravenously dosed with the particular cannabinoid (1 mg of CBD, 1.5 mg of the other three cannabinoids in the form of nanoemulsion, volume 100 μL). Systemic blood (100 μL) was drawn at 5, 15, and 30 min and at 1, 2, 4, 6, 8, 10, and 24 h(s) postdose. After a two-day wash-out period, the dosing via oral gavage followed (10 mg of CBD, 15 mg of the other three cannabinoids in the form of nanoemulsion, volume 1 mL). Systemic blood (100 μL) was drawn at 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8, 10, and 24 h(s). Blood samples were centrifuged (4500 rpm for 10 min) and serum was extracted and stored in −80 °C until analysis. The laboratory was unaware of animal assignment to particular experimental groups (laboratory blinding).
Lymphatic Transport Studies
Anesthetized mesenteric lymph duct cannulated rat model was used for the lymphatic transport measurement as previously described with slight modifications.ref. ref31 Briefly, rats were left on normal diet and given 1 mL of olive oil 1 h prior to the surgery to visualize the mesenteric lymph (milky white color). They were anesthetized with intramuscular xylazine 5 mg/kg and ketamine 100 mg/kg after a rapid isoflurane induction. A transverse laparotomy was performed. The mesenteric duct was identified cranially to superior mesenteric artery and cannulated with heparin prefilled 0.91 mm O.D., 0.46 mm I.D. polyethylene catheter (Instech Laboratories, Plymouth Meeting, USA). The catheter was fixed in place with two to three drops of tissue adhesive (Histoacryl, B. Braun Surgical, S.A., Spain). A duodenal catheter was also placed (polyethylene, 0.97 mm O.D., 0.58 mm I.D.) via a small duodenotomy and fixed with tissue adhesive. The abdominal wall was sutured in two layers, with both catheters leaving the abdominal cavity through the right flank. At the end of the procedure, the right jugular vein was cannulated for blood sampling.
The rats were then placed on heated pads and covered with blankets to prevent heat loss. Cannabinoids in oil solution or nanoemulsion were then dosed slowly via duodenal catheter over 30 min. The administered dose was 10 mg of CBD and 15 mg of CBG, CBC and CBN (volume 1 mL). Whole lymph was collected in regularly changed Eppendorf tubes from the time the dosing started. When the dosing was finished, continuous hydration with normal saline at a rate of 3 mL/h intraduodenally followed using an infusion pump (Perfusor compactplus, B. Braun Melsungen AG, Germany). Anesthesia was maintained throughout the rest of the experiment and additional ketamine intramuscular boluses were given whenever necessary. Eppendorf tubes were changed hourly and systemic blood was drawn at the same time points. Blood samples were centrifuged (4500 rpm for 10 min) and serum was extracted. Lymph volume was measured gravimetrically and the samples were further processed without additional adjustment. All samples were stored in −80 °C until analysis. Laboratory was unaware of animal assignment to the particular experimental groups (laboratory blinding).
Bioanalysis
The concentrations of the cannabinoids under study, namely CBD, CBG, CBC and CBN were determined in both serum and lymph samples. An ultrahigh performance liquid chromatography-tandem mass spectrometric (UHPLC-MS/MS) method was developed for the quantification of each cannabinoid, employing an isotopically labeled internal standard (IS). The samples underwent identical processing procedures for each cannabinoid: 80 μL of 100% acetonitrile containing 30 ng/mL of an appropriate internal standard was added to 20 μL of the sample. After vortexing and centrifugation at 10000 × g for 8 min, 60 μL of the supernatant was transferred into an LC vial. For the UHPLC-MS/MS analysis, the Shimadzu UHPLC Nexera X3 coupled with a Triple Quad 8045 tandem mass spectrometer (Shimadzu, Kyoto, Japan) was used. Chromatographic analysis was performed on a Poroshell 120 EC-C18 column (50 × 2.1 mm; 1.9 μm; Agilent Technologies, Inc., Santa Clara, CA, USA). The mobile phase consisted of 0.1% formic acid in deionized water (Solvent A) and methanol with 0.1% formic acid (Solvent B). The flow rate of the mobile phase was maintained at 0.4 mL/min, except for the CBC determination method, which had a flow rate of 0.45 mL/min. The injection volume was 2 μL. The temperature of the column was kept at 40 °C and samples were thermostated at 10 °C. The optimized gradient elution proceeded as follows for each cannabinoid: CBD (min/% B): 0/50, 0.5/50, 2.5/90, 3.5/90, 4.0/50, and 5.5/50; CBG (min/% B): 0/50, 0.5/50, 2.5/90, 4.0/90, 4.5/50, and 7.0/50; CBC and CBN (min/% B): 0/40, 0.5/40, 2.5/90, 4.0/90, 4.5/40, and 7.0/40. The MS/MS spectrometer was operated in a positive mode. The applied conditions of the electrospray ion source were: nebulizing gas flow: 3 L/min, heating gas flow: 10 L/min, interface temperature: 300 °C, desolvation line temperature: 250 °C, heat block temperature: 400 °C, and drying gas flow: 10 L/min. The MS/MS measurement was performed in multiple reaction-monitoring mode (MRM). MRM transitions of 315.2 > 193.1 (Q1 prebias −16 V, Q3 prebias −20 V and collision energy −22 V) and 318.2 > 196.1 (Q1 prebias −16 V, Q3 prebias −22 V and collision energy −35 V) were monitored for CBD and CBD-d3, respectively. MRM transitions of 315.5 > 193.2 (Q1 prebias −13 V, Q3 prebias −20 V and collision energy −20 V) and 324.4 > 202.2 (Q1 prebias −17 V, Q3 prebias −21 V and collision energy −20 V) were monitored for CBC and CBC-d9, respectively. MRM transitions of 317.2 > 193.2 (Q1 prebias −15 V, Q3 prebias −20 V and collision energy −19 V) and 326.3 > 202.2 (Q1 prebias −16 V, Q3 prebias −21 V and collision energy −18 V) were monitored for CBG and CBG-d9, respectively. MRM transitions of 311.3 > 223.2 (Q1 prebias −17 V, Q3 prebias −21 V and collision energy −21 V) and 314.3 > 223.2 (Q1 prebias −17 V, Q3 prebias −24 V and collision energy −22 V) were monitored for CBN and CBN-d3, respectively.
The calibration curves were constructed in each blank matrix (serum and lymph) with seven concentrations by plotting the ratio of the peak area of cannabinoid to that of IS against cannabinoid concentration. The weighted least-squares linear regression method was used with a weighting factor of 1/x2, which improved the accuracy in low concentrations. The methods were linear (coefficients of determination (R2) higher than 0.9995) in the concentration ranges of 1–1000 ng/mL for CBD, and 2.5–1250 ng/mL for CBG, CBC, and CBN. The methods were validated according to the requirements of the European Medicines Agency (EMA) Guideline on bioanalytical method validation in terms of linearity, lower limit of quantification (LLOQ), upper limit of quantification (ULOQ), accuracy, precision, selectivity, recovery, carry-over effect, matrix effects, robustness, dilution integrity, and stability of quality control (QC), and have been demonstrated to be suitable for their intended purpose.ref. ref32 All validation parameters met the acceptance criteria defined in the EMA guideline.
Log P Measurement
There are only calculated log P values available for CBD, CBG, CBC and CBN (range 6.1–7.4). Therefore, log P was measured experimentally for all four cannabinoids during this study. The partition coefficient describes the distribution of a tested compound in a two-phase octanol–water system. It is defined as a ratio of concentrations of the un-ionized compound in octanol and water at equilibrium. Cannabinoids were weighed (2 mg) and dissolved in 2.5 mL of octanol, followed by the addition of 2.5 mL of deionized water. The mixture was shaken for 2 h and then allowed to separate for 24 h. Twenty μL of the aqueous (bottom) phase was transferred into an LC vial, diluted with 60 μL of an appropriate IS at a concentration of 30 ng/mL, and analyzed using the aforementioned UHPLC-MS/MS method. The experiment was repeated five times for each cannabinoid.
Quantitative Structure Determination
First, an optimal, energy-minimized conformation was obtained by molecular mechanics modeling in the software Avogadro, version 1.2.0.n (an open-source molecular builder and visualization tool). The force field GAFF, which is optimized for drug molecules, was used. The initial conformations of the cannabinoids were obtained either from available crystal structures (CBD and CBG) or generated using SMILES descriptors (CBC and CBN). The size of the molecules was calculated as the smallest, medium, and largest orthogonal dimensions in Å (10–10 m) using the software rPluto, version 5.26 (The Cambridge Crystallographic Data Centre, Cambridge, UK).
Data Analysis and Statistics
Serum and lymph concentrations in all studies were dose-normalized to 1 mg/kg prior to further calculations. AUC values were determined using linear trapezoidal rule. Exact actual sampling times were used for this purpose. Scheduled sampling times were used for mean concentration plotting in the graphs. PK-solver add-on for MS Excel was used for all basic pharmacokinetic calculations.ref. ref33 GraphPad Prism version 10.2.2 (GraphPad Software, San Diego, CA, USA) was used for all statistical analyses and graph plotting. Unpaired Student’s t test was used to compare pharmacokinetic and lymphatic transport parameters between the experimental groups. Level of significance was set to p < 0.05.
Calculation of Lymphatic Transport Parameters
Lymphatic transport parameters were defined and calculated as previously described.ref2,ref14,ref34 Briefly, absolute bioavailability via lymph (FAL) was defined as percentage of administered drug dose absorbed into the lymph. It was determined directly from lymph volume and drug concentration in lymph duct cannulated rats. Absolute bioavailability via portal vein (FAP) was analogically defined as percentage of administered drug dose reaching the systemic circulation after direct absorption into blood. It was calculated using equation
where AUCent is the area under the dose-normalized blood concentration–time curve after enteral dosing in lymph duct cannulated (i.e., lymph deprived) rats and AUCiv is the respective parameter in a separate intravenously dosed group. Total absolute bioavailability (F) in lymph duct cannulated rats was calculated as a sum of FAL and FAP. In normal animals with no lymph duct cannulation, F was calculated using standard formula for oral bioavailability:
Relative bioavailability via lymph (FRL) was defined as the percentage of systemically available drug that was absorbed via lymph. It was calculated using the equation
Results and Discussion
Pharmacokinetics
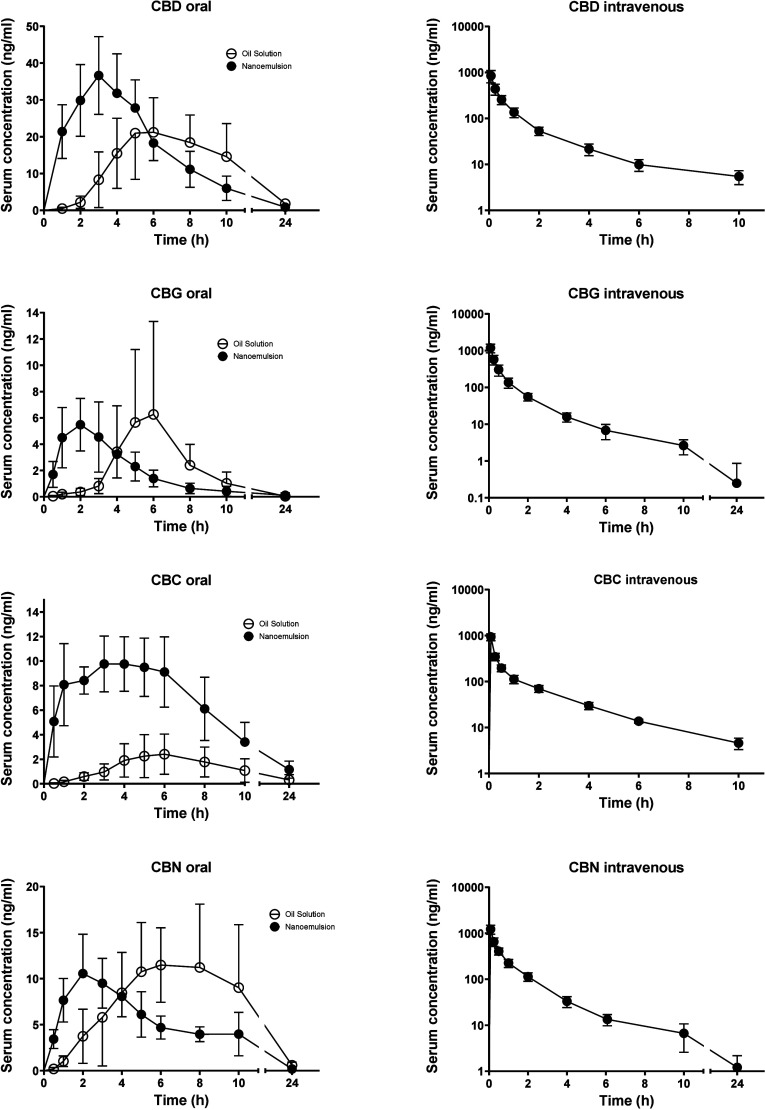
Pharmacokinetic profiles for all four cannabinoids after oral and intravenous administration are shown in Figure . The corresponding pharmacokinetic parameters are summarized in Table . The absolute oral bioavailability varied significantly from 4.4% in CBG nanoemulsion to as much as 52% in CBD oil solution. The bioavailability from the oil solution and nanoemulsion did not differ significantly in the particular cannabinoids with the exception of CBN, where the compound achieved a higher systemic exposure when administered in an oil solution. The drugs were absorbed at a low speed after oil solution administration (Tmax of 4.9 to 6.8 h) and at a moderate speed after nanoemulsion administration (Tmax of 2 to 3.5 h). The elimination was quite fast in CBD, CBG and CBC with T 1/2 of 2 to 2.5 h after intravenous dosing, whereas the CBN elimination was slower with T 1/2 of 5 h.
1: Mean ± SD Serum Pharmacokinetic Parameters of CBD, CBG, CBC, and CBN after Administration in the Form of an Oil Solution (oral) and Nanoemulsion (Oral and Intravenous) to Rats
| CBD | CBG | CBC | CBN | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oil | Emulsion | Oil | Emulsion | Oil | Emulsion | Oil | Emulsion | |||||
| PO | PO | IV | PO | PO | IV | PO | PO | IV | PO | PO | IV | |
| N | 6 | 7 | 7 | 8 | 5 | 6 | 8 | 6 | 6 | 8 | 7 | 7 |
| Cmax (ng/mL) | 27 ± 10* | 39 ± 8 | 845 ± 253 | 15 ± 15 | 6 ± 2 | 1186 ± 286 | 2.6 ± 1.5* | 12 ± 2 | 935 ± 147 | 15.3 ± 4.6 | 12 ± 3 | 1226 ± 252 |
| Tmax (h) | 6.8 ± 1.9* | 3.2 ± 0.9 | – | 4.9 ± 2.0* | 2.0 ± 1.3 | – | 6.0 ± 1.3* | 3.6 ± 1.9 | – | 6.5 ± 1.2* | 2.2 ± 0.6 | – |
| T1/2 (h) | 4.8 ± 0.8 | 4.4 ± 0.8 | 2.5 ± 0.5 | 3.1 ± 1.3 | 5.1 ± 4.1 | 2.5 ± 1.6 | 13 ± 19 | 5.8 ± 1.9 | 2.2 ± 0.4 | 4.2 ± 1.7 | 3.5 ± 1.1 | 5.3 ± 1.8 |
| AUC0‑inf (ng h/mL) | 205 ± 57 | 256 ± 38 | 623 ± 141 | 36 ± 22 | 28 ± 10 | 716 ± 136 | 31 ± 23* | 117 ± 33 | 640 ± 85 | 148 ± 57* | 91 ± 19 | 1041 ± 212 |
| F0‑inf (%) | 52 ± 19 | 45 ± 18 | – | 12 ± 11 | 4.4 ± 2.6 | – | 34 ± 18 | 18 ± 5 | – | 20 ± 7* | 9.2 ± 2.6 | – |
| Vss (L/kg) | – | – | 2.9 ± 1.1 | – | – | 7.3 ± 2.3 | – | – | 11 ± 2.2 | – | – | 6.5 ± 1.6 |
| CL (L/h kg) | – | – | 1.7 ± 0.6 | – | – | 5.9 ± 1.1 | – | – | 6.0 ± 0.7 | – | – | 3.0 ± 0.7 |
a The administered dose was 10 mg of CBD and 15 mg of the other cannabinoids in oral dosing (volume 1 mL). The intravenous doses were 1 mg of CBD and 1.5 mg of the other cannabinoids (volume 0.1 mL). All concentrations and AUCs are dose-normalized to 1 mg/kg. CBD intravenous data have already been published earlier. * marks a significant difference (p < 0.05) between nanoemulsion and the corresponding oil solution.
The absolute oral bioavailability of 52% and 45% in CBD oil solution and nanoemulsion, respectively, (Table ) exceeds significantly reports published so far (8.5% for a lipid-free formulation,ref. ref19 14% for sunflower oil,ref. ref28 22% for a long chain triglyceride-based formulationref. ref19 and 25% for a sunflower oil-based microemulsion).ref. ref28 The reason may lie in the extended time over which the pharmacokinetic profile was measured in this study (24 h and extrapolated to infinity). The older studies usually used a shorter period of measurement which may not have covered the full pharmacokinetic profile and a significant portion of AUC was not captured.
The absolute oral bioavailability was generally very different between the cannabinoids (Oil solution vs nanoemulsion: 52% vs 45% for CBD, 12% vs 4% for CBG, 34% vs 18% for CBC, and 20% vs 9% for CBN) despite their similar chemical properties and despite administration in the same formulations. This indicates that the factors affecting the amount of systemically available drug (intestinal absorption, intestinal and liver first-pass metabolism, extent of lymphatic transport) have to play varying roles in the particular compounds. In the lymphatic transport studies, the extent of portally absorbed drug (FAP) in the nanoemulsions did not differ significantly between the cannabinoids, whereas the relative proportion of lymphatically absorbed drug (FRL) was rather low in CBD and CBG (both 13%) and higher in CBC and CBN (47% and 36%, respectively), see below. The situation was different for the oil solutions, where the FAP in CBG was lower compared to other cannabinoids, but the relative bioavailability via lymph was very high (>50%) similarly in all four compounds.
Lymphatic Transport
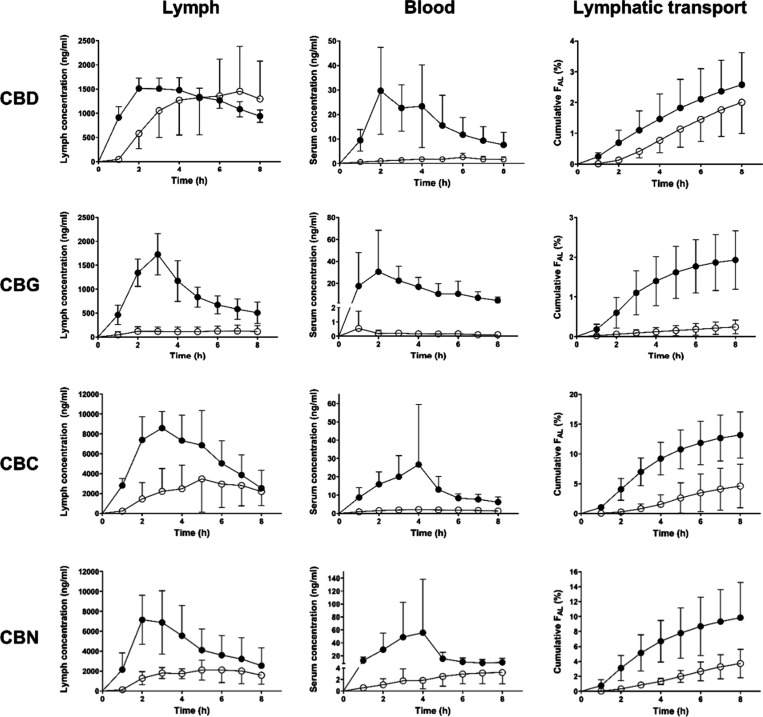
Figure displays CBD, CBG, CBC and CBN serum and lymph profiles and the corresponding cumulative lymphatic transport after administration to anesthetized lymph duct-cannulated rats. Table summarizes all the lymphatic transport parameters. Administration of a nanoemulsion led generally to an increased total absolute bioavailability compared to oil solution. The bioavailability increase was due to both increased direct absorption into portal blood and transport through lymphatic vessels with the exception of CBD, where the lymphatic transport measured as absolute bioavailability via lymph (FAL) did not change significantly. Interestingly, the absolute bioavailability via portal vein increased more than the bioavailability via lymph in all four cannabinoids. Whereas there was no increase, 8-fold, 3-fold, and 3-fold increase in FAL for CBD, CBG, CBC and CBN, respectively, there was an 11-fold, 71-fold, 8-fold and 13-fold increase in absolute bioavailability via portal vein (FAP) (Table ). Consequently, the relative bioavailability via lymph (FRL) dropped by 74% in CBD, 75% in CBG and 48% in CBN in the nanoemulsion compared to oil solution. The FRL decrease in CBC did not reach statistical significance (p = 0.12). The reason for the higher bioavailability via portal vein (FAP) in all cannabinoids and higher bioavailability via lymph (FAL) in all cannabinoids but CBD for the nanoemulsion compared to oil solution lies probably in a poor dispersion and digestion of oil that was administered intraduodenally, skipping the dispersion and digestion process in the stomach.ref. ref3
2: Mean ± SD Lymphatic Transport Parameters of CBD, CBG, CBC, and CBN after Intraduodenal Administration to Anesthetized Lymph Duct-Cannulated Rats in a Form of Nanoemulsion and Oil Solution
| CBD | CBG | CBC | CBN | |||||
|---|---|---|---|---|---|---|---|---|
| Formulation | Emulsion | Oil | Emulsion | Oil | Emulsion | Oil | Emulsion | Oil |
| N | 6 | 6 | 7 | 6 | 7 | 6 | 6 | 7 |
| AUC0–8 lymph (ng h/L) | 9556 ± 996 | 7699 ± 3815 | 7026 ± 1159* | 802 ± 518 | 43,020 ± 11,165* | 16,695 ± 13,034 | 33,935 ± 12,019* | 11,979 ± 4,006 |
| AUC0–8 serum (ng h/L) | 125 ± 54* | 11 ± 5 | 117 ± 71* | 1.6 ± 1.7 | 98 ± 46* | 12 ± 4 | 187 ± 93* | 16 ± 8 |
| Lymph/serum AUC ratio | 100 ± 60* | 699 ± 333 | 77 ± 31* | 802 ± 582 | 530 ± 239* | 1255 ± 676 | 274 ± 231* | 976 ± 519 |
| F (%) | 24 ± 9* | 4.0 ± 1.5 | 19 ± 10* | 0.5 ± 0.3 | 29 ± 7* | 6.6 ± 3.9 | 29 ± 10* | 5.3 ± 2.0 |
| FAP (%) | 21 ± 9* | 1.9 ± 0.9 | 17 ± 10* | 0.24 ± 0.25 | 16 ± 8* | 2.0 ± 0.7 | 20 ± 10* | 1.6 ± 0.9 |
| FAL (%) | 2.6 ± 1.0 | 2.0 ± 0.9 | 1.9 ± 0.7* | 0.24 ± 0.16 | 13 ± 4* | 4.6 ± 3.3 | 9.9 ± 4.3* | 3.7 ± 1.8 |
| FRL (%) | 13 ± 8* | 50 ± 12 | 13 ± 7* | 51 ± 24 | 47 ± 14 | 63 ± 17 | 36 ± 20* | 69 ± 13 |
a The administered dose was 10 mg of CBD and 15 mg of the other cannabinoids. The administered volume was 1 mL. All AUCs are dose-normalized to 1 mg/kg. * marks a significant difference (p < 0.05) between nanoemulsion and the corresponding oil solution. F, total absolute bioavailability; FAP, absolute bioavailability via portal vein; FAL, absolute bioavailability via lymph; FRL, relative bioavailability via lymph.
As expected, the lymphatic transport played a major role in the general pharmacokinetics of all four cannabinoids tested. The relative bioavailability via lymph (FRL) of ≥50% seen after administration of oil solutions counts the cannabinoids among compounds with the highest lymphotropic potential, like vitamin D or halofantrine.ref10,ref35
Log P Measurement
Due to the highly lipophilic character of cannabinoids, their concentration in the organic phase is several orders of magnitude higher than the concentration in the aqueous phase. As a result, the difference in the total amount of cannabinoids and the amount present in the organic phase is very small and falls within the experimental error of the analytical method. Therefore, log P values were calculated based on the measured cannabinoid concentration in the aqueous phase, while the concentration in the octanol phase was calculated from the initial amount of the cannabinoid used and its measured concentration in the aqueous phase. Using this approach, log P values were estimated as 6.2 ± 0.2 for CBN and 7.2 ± 0.2 for CBG. Due to the very low concentrations of CBD and CBC in the aqueous phase, which fell below the LOQ of both methods, we were unable to determine the log P for these two cannabinoids. Nevertheless, based on the LOQ values, we can infer that the log P values exceed 6.3 for CBD and 6.7 for CBC.
It is clear that molecules with log P > 5 can be transported through the mesenteric lymph while molecules with log P < 5 cannot.ref. ref2 However, even in molecules with log P > 5, the FRL can still profoundly vary. Reason for a very low FRL (<10%) can be an insufficient amount of long-chain triglycerides present in the drug formulation to stimulate the chylomicron assembly like it was the case in halofantrine (log P 8.9) when administered in an aqueous solution (FRL = 5.3%).ref. ref10 A very high FRL (>90%), on the other hand, is usually seen in drugs with a prominent first-pass metabolism in the liver, like testosterone undecanoate (log P 6.7, FRL = 98.5%).ref. ref6 This high FRL does not mean that no drug is absorbed into the portal vein and all the drug is absorbed into the lymphatics, but rather that the drug absorbed into the portal vein is (almost) completely metabolized during the first pass in the liver and only lymphatically transported portion reaches the systemic circulation. The mechanisms determining the FRL in the range ∼10–90% are not well understood. Important here is the type and amount of lipids administered. Long-chain triglycerides stimulate the lymphatic transport more than medium-chain triglycerides and short-chain triglycerides. The FRL of halofantrine was 70%, 29% and 15% when administered in a long, medium and short-chain triglyceride oil, respectively.ref. ref10 In this study, cannabinoids administered in a sunflower oil, which consists mainly of long chain triglycerides containing oleic and linoleic acid, reached a very high FRL (50–69%). In the nanoemulsion, on the other hand, there were no standard triglycerides. It consisted of polyethoxylated castor oil (Kolliphor EL), diethylene glycol monoethyl ether, and propylene glycol monocaprylate. The lack of long chain triglycerides is the most plausible explanation, why the FRL here was lower (13–47%).
CBD, CBG, CBC, and CBN Quantitative Structure–Activity Relationship with Lymphatic Transport
Table summarizes the basic physicochemical properties and quantitative structural parameters of the four cannabinoids and Figure shows their relation to lymphatic transport expressed as mean relative bioavailability via lymph (FRL). There was a highly significant negative correlation between the smallest orthogonal molecular dimension and FRL marked by the significantly nonzero slope in the linear regression analysis (p < 0.001). Topological polar surface area did also negatively correlate (p = 0.04). Other physicochemical properties (medium and largest orthogonal dimension, molecular volume, rotatable bond count, log P and pK a) did not have a significant effect.
3: Physicochemical Properties and Quantitative Structure Parameters of the Four Tested Cannabinoids
| CBD | CBG | CBC | CBN | |
|---|---|---|---|---|
| Log P | >6.3 | 7.2 | >6.7 | 6.2 |
| pK a | 9.70 | 9.71 | 9.68 | 9.40 |
| Rotatable bond count | 6 | 9 | 7 | 4 |
| Topological polar surface area (Å2) | 40.5 | 40.5 | 29.5 | 29.5 |
| Molecular volume (Å3) | 329 | 339 | 329 | 312 |
| Largest orthogonal dimension (Å) | 17.2 | 15.3 | 16.4 | 17.1 |
| Medium orthogonal dimension (Å) | 9.9 | 10.5 | 11.7 | 10.6 |
| Smallest orthogonal dimension (Å) | 8.31 | 8.12 | 7.10 | 6.58 |
| Medium/largest orthogonal dimension ratio | 0.576 | 0.687 | 0.713 | 0.619 |
| Smallest/largest orthogonal dimension ratio | 0.483 | 0.530 | 0.433 | 0.386 |
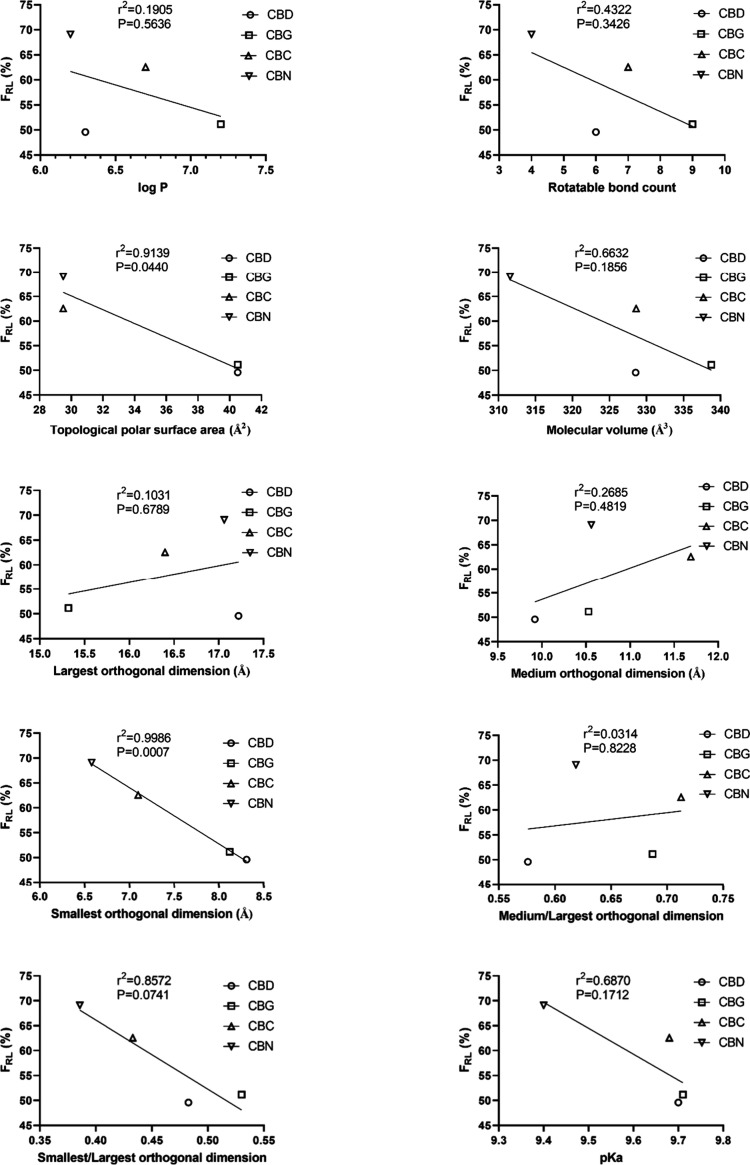
In our study, we compared FRL of four highly lipophilic cannabinoids administered in the very same formulation. Interestingly the FRL did not correlate with the log P value. Based on these results and based on the literature, it seems that log P of 5 represents a threshold beyond which a drug can be lymphatically absorbed but other factors determine to which extent. Our QSAR analysis on a limited number of compounds showed that the overall size of the molecule seems not to be so important for increasing lymphatic uptake as its planarity (FRL increases with decreasing smallest orthogonal dimension, while the largest and medium orthogonal dimensions and volume of the molecule are not significantly associated with FRL). Another structural parameter that (negatively) correlates with the FRL is the topological polar surface area.
Following oral administration, strongly lipophilic drugs access the mesenteric lymph vessels via absorption across enterocytes.ref. ref4 The substances are drawn into the cell organelles, especially the smooth endoplasmic reticulum, where the formation of chylomicrons takes place. The chylomicrons consist of a phospholipid membrane, proteins and a lipophilic core consisting mainly of triglycerides, in which the highly lipophilic substances, including drugs, are dissolved. Chylomicrons are subsequently transported by the Golgi apparatus toward the basal pole of the cell and secreted into the intercellular space. They are too large to penetrate the wall of blood capillaries and, therefore, enter the lymphatic capillaries, which have a more permeable vessel wall. Thus, the interpretation of our observations may be that higher planarity allows easier diffusion across phospholipid membranes of the cell organelles where the chylomicrons are formed and across the membranes of the chylomicrons themselves. The lower topological polar surface area leads additionally to higher drug solubility in long-chain triglycerides in the chylomicron lipophilic cores. To our knowledge, this is the first report describing these structure–activity relationships that are important for the design and development of modern intestinal lymph-targeting drugs. Further studies are warranted to confirm these preliminary conclusions.
Conclusions
This work shows on the example of four cannabinoids CBD, CBG, CBC, and CBN that for highly lipophilic active compounds, planarity of the molecule and less surface polarity seem to be crucial structural features that facilitate lymphatic transport and could therefore play an important role in this absorption mechanism in the context of absolute oral bioavailability of the drug. The overall size of the molecule was of little significance for the extent of lymphatic absorption. Compared to standard oil solution, administration of a nanoemulsion devoid of standard triglycerides increased the absorption speed and amount of cannabinoids absorbed directly into portal blood, bud did not affect the overall bioavailability. Overall, intestinal lymphatic transport plays a major role in the process of intestinal absorption of the cannabinoids CBD, CBG, CBC, and CBN.
Supplementary Materials
References
- W. N. A. Charman, V. J. Stella. Estimating the Maximal Potential for Intestinal Lymphatic Transport of Lipophilic Drug Molecules. Int. J. Pharm., 1986. [DOI]
- P. Rysanek, T. Grus, M. Sima, O. Slanar. Lymphatic Transport of Drugs after Intestinal Absorption: Impact of Drug Formulation and Physicochemical Properties. Pharm. Res., 2020. [DOI | PubMed]
- C. J. H. Porter, N. L. Trevaskis, W. N. Charman. Lipids and lipid-based formulations: optimizing the oral delivery of lipophilic drugs. Nat. Rev. Drug Discov, 2007. [DOI | PubMed]
- N. L. Trevaskis, L. M. Kaminskas, C. J. H. Porter. From sewer to saviour – targeting the lymphatic system to promote drug exposure and activity. Nat. Rev. Drug Discov, 2015. [DOI | PubMed]
- K. L. White, G. Nguyen, W. N. Charman, G. A. Edwards, W. A. Faassen, C. J. H. Porter. Lymphatic Transport of Methylnortestosterone Undecanoate (MU) and the Bioavailability of Methylnortestosterone Are Highly Sensitive to the Mass of Coadministered Lipid after Oral Administration of MU. J. Pharmacol Exp Ther, 2009. [DOI | PubMed]
- D. M. Shackleford, W. A. Faassen, N. Houwing, H. Lass, G. A. Edwards, C. J. Porter, W. N. Charman. Contribution of lymphatically transported testosterone undecanoate to the systemic exposure of testosterone after oral administration of two andriol formulations in conscious lymph duct-cannulated dogs. J. Pharmacol Exp Ther, 2003. [DOI | PubMed]
- T. Yoshida, K. Nakanishi, T. Yoshioka, Y. Tsutsui, A. Maeda, H. Kondo, K. Sako. Oral tacrolimus oil formulations for enhanced lymphatic delivery and efficient inhibition of T-cell’s interleukin-2 production. Eur. J. Pharm. Biopharm, 2016. [DOI | PubMed]
- S. F. Han, L. J. Hu, Gracia, T. Quach, J. S. Simpson, G. A. Edwards, N. L. Trevaskis, C. J. H. Porter. Lymphatic Transport and Lymphocyte Targeting of a Triglyceride Mimetic Prodrug Is Enhanced in a Large Animal Model: Studies in Greyhound Dogs. Mol. Pharmaceut, 2016. [DOI]
- S. M. Khoo, D. M. Shackleford, C. J. Porter, G. A. Edwards, W. N. Charman. Intestinal lymphatic transport of halofantrine occurs after oral administration of a unit-dose lipid-based formulation to fasted dogs. Pharm. Res., 2003. [DOI | PubMed]
- S. M. Caliph, W. N. Charman, C. J. Porter. Effect of short-, medium-, and long-chain fatty acid-based vehicles on the absolute oral bioavailability and intestinal lymphatic transport of halofantrine and assessment of mass balance in lymph-cannulated and non-cannulated rats. J. Pharm. Sci., 2000. [DOI | PubMed]
- S. F. Han, L. J. Hu, T. Quach, J. S. Simpson, N. L. Trevaskis, C. J. H. Porter. Constitutive Triglyceride Turnover into the Mesenteric Lymph Is Unable to Support Efficient Lymphatic Transport of a Biomimetic Triglyceride Prodrug. J. Pharm. Sci., 2016. [DOI | PubMed]
- A. Dahan, R. Duvdevani, I. Shapiro, A. Elmann, E. Finkelstein, A. Hoffman. The oral absorption of phospholipid prodrugs: In vivo and in vitro mechanistic investigation of trafficking of a lecithin-valproic acid conjugate following oral administration. J. Controlled Release, 2008. [DOI]
- J. Sugihara, S. Furuuchi, K. Nakano, S. Harigaya. Studies on Intestinal Lymphatic Absorption of Drugs.1. Lymphatic Absorption of Alkyl Ester Derivatives and Alpha-Monoglyceride Derivatives of Drugs. J. Pharmacobio-Dynam, 1988. [DOI]
- P. Rysanek, T. Grus, P. Lukac, P. Kozlik, T. Krizek, J. Pozniak, J. Rousarova, J. Kralovicova, N. K. Canova, T. Boleslavska, J. Bosak, F. Stepanek, M. Sima, O. Slanar. Validity of cycloheximide chylomicron flow blocking method for the evaluation of lymphatic transport of drugs. Br. J. Pharmacol., 2021. [DOI | PubMed]
- C. T. Ueda, M. Lemaire, G. Gsell, K. Nussbaumer. Intestinal lymphatic absorption of cyclosporin A following oral administration in an olive oil solution in rats. Biopharm Drug Dispos, 1983. [DOI | PubMed]
- B. T. Griffin, C. M. O’Driscoll. A comparison of intestinal lymphatic transport and systemic bioavailability of saquinavir from three lipid-based formulations in the anaesthetised rat model. J. Pharm. Pharmacol, 2006. [DOI | PubMed]
- WHO . WHO Expert Committee on Drug Dependence (Fourtieth Report); World Health Organization, 2018.
- L. Taylor, B. Gidal, G. Blakey, B. Tayo, G. Morrison. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. Cns Drugs, 2018. [DOI | PubMed]
- A. Zgair, J. C. Wong, J. B. Lee, J. Mistry, O. Sivak, K. M. Wasan, I. M. Hennig, D. A. Barrett, C. S. Constantinescu, P. M. Fischer, P. Gershkovich. Dietary fats and pharmaceutical lipid excipients increase systemic exposure to orally administered cannabis and cannabis-based medicines. Am. J. Transl. Res., 2016. [PubMed]
- F. Calapai, L. Cardia, E. Esposito, I. Ammendolia, C. Mondello, R. Lo Giudice, S. Gangemi, G. Calapai, C. Mannucci. Pharmacological Aspects and Biological Effects of Cannabigerol and Its Synthetic Derivatives. Evidence-Based Complementary Altern. Med., 2022. [DOI]
- K. Amstutz, W. S. Schwark, A. Zakharov, B. Gomez, A. Lyubimov, K. Ellis, K. P. Venator, J. J. Wakshlag. Single dose and chronic oral administration of cannabigerol and cannabigerolic acid-rich hemp extract in fed and fasted dogs: Physiological effect and pharmacokinetic evaluation. J. Vet Pharmacol Ther, 2022. [DOI | PubMed]
- P. W. Wirth, E. S. Watson, M. ElSohly, C. E. Turner, J. C. Murphy. Anti-inflammatory properties of cannabichromene. Life Sci., 1980. [DOI | PubMed]
- J. D. O’Neil, W. S. Dalton, R. B. Forney. The effect of cannabichromene on mean blood pressure, heart rate, and respiration rate responses to tetrahydrocannabinol in the anesthetized rat. Toxicol. Appl. Pharmacol., 1979. [DOI | PubMed]
- E. N. Peters, L. MacNair, I. Mosesova, U. Christians, C. Sempio, J. Klawitter, M. H. Land, M. A. Ware, C. Turcotte, M. O. Bonn-Miller. Pharmacokinetics of cannabichromene in a medical cannabis product also containing cannabidiol and Delta(9)-tetrahydrocannabinol: a pilot study. Eur. J. Clin Pharmacol, 2022. [DOI | PubMed]
- P. B. Sampson. Phytocannabinoid Pharmacology: Medicinal Properties of Cannabis sativa Constituents Aside from the “Big Two”. J. Nat. Prod, 2021. [DOI | PubMed]
- C. F. Moore, E. M. Weerts, J. Kulpa, D. Schwotzer, W. Dye, J. Jantzi, J. D. McDonald, T. W. Lefever, M. O. Bonn-Miller. Pharmacokinetics of Oral Minor Cannabinoids in Blood and Brain. Cannabis Cannabinoid Res., 2023. [DOI | PubMed]
- Y. F. Miao, S. H. Zhao, J. Zuo, J. Q. Sun, J. N. Wang. Reduced the Food Effect and Enhanced the Oral Bioavailability of Ivacaftor by Self-Nanoemulsifying Drug Delivery System (SNEDDS) Using a New Oil Phase. Drug Des Dev Ther, 2022. [DOI]
- P. Jelinek, J. Rousarova, P. Rysanek, M. Jezkova, T. Havlujova, J. Pozniak, P. Kozlik, T. Krizek, T. Kucera, M. Sima, O. Slanar, M. Soos. Application of Oil-in-Water Cannabidiol Emulsion for the Treatment of Rheumatoid Arthritis. Cannabis Cannabinoid Res., 2024. [DOI | PubMed]
- R. A. Myers, V. J. Stella. Factors Affecting the Lymphatic Transport of Penclomedine (Nsc-338720), a Lipophilic Cytotoxic Drug – Comparison to Ddt and Hexachlorobenzene. Int. J. Pharm., 1992. [DOI]
- R. Holm, J. Hoest. Successful in silico predicting of intestinal lymphatic transfer. Int. J. Pharm., 2004. [DOI | PubMed]
- N. L. Trevaskis, L. Hu, S. M. Caliph, S. Han, C. J. Porter. The mesenteric lymph duct cannulated rat model: application to the assessment of intestinal lymphatic drug transport. J. Vis Exp, 2015. [DOI]
- ICH guideline M10 on bioanalytical method validation and study sample analysis.. 2022
- Y. Zhang, M. R. Huo, J. P. Zhou, S. F. Xie. PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comput. Meth Prog. Bio, 2010. [DOI]
- J. Pozniak, P. Rysanek, D. Smrcka, P. Kozlik, T. Krizek, J. Smardova, A. Novakova, D. Das, D. Bobek, M. Arora, J. Hofmann, T. Dousova, M. Sima, O. Slanar. Ivacaftor pharmacokinetics and lymphatic transport after enteral administration in rats. Front. Pharmacol., 2024. [DOI | PubMed]
- A. Dahan, A. Hoffman. Evaluation of a chylomicron flow blocking approach to investigate the intestinal lymphatic transport of lipophilic drugs. Eur. J. Pharm. Sci., 2005. [DOI | PubMed]
