Association Between Prenatal Cannabis Exposure and Child Health Care Use: A Retrospective Cohort Study in Ontario, Canada
Abstract
Objective:
Cannabis use among expectant mothers has increased steadily over the past two decades. We compared the long-term health services use of offspring prenatally exposed to cannabis to that of matched, unexposed offspring.
Study design:
We conducted a retrospective cohort study using linked perinatal and health administrative databases of all live, singleton births in Ontario, Canada hospitals between April 1, 2007 and March 31, 2012. Infants were followed until March 31, 2017, with a primary outcome of primary care visits up to age 10. Secondary outcomes included rates of outpatient psychiatrist visits, emergency department visits and hospitalizations. We used adjusted Poisson regression to assess differences in rates of health service use between children with and without exposure to prenatal cannabis.
Results:
We included 508 025 infants, 3248 (0.6%) had cannabis exposure. Prenatal cannabis use was associated with a decreased rate of primary care physician visits (adjusted rate ratio [aRR]: 0.86, 95% confidence interval [CI]: 0.84-0.87) and an increase in the rate of outpatient psychiatrist visits (aRR: 1.29, 95% CI: 1.00-1.66), emergency department visits (aRR: 1.05, 95% CI: 1.03-1.08), and hospitalizations (aRR: 1.12, 95% CI: 1.04-1.20). Among preterm offspring, cannabis was associated with a decrease in primary care visits but no difference in other visits. Among those in the highest income quintiles, cannabis use was associated with a two-fold increase in the rate of outpatient psychiatrist visits.
Conclusions:
Offspring exposed to prenatal cannabis receive fewer primary care visits but have increased rates of visiting health care specialists past the neonatal period.
Article type: Research Article
Keywords: cannabis, marijuana, pregnancy, health services, health administrative databases, retrospective, substance use, BORN
Affiliations: School of Epidemiology and Public Health, Faculty of Medicine, University of Ottawa, Ottawa, ON, Canada; Children’s Hospital of Eastern Ontario Research Institute, Ottawa, ON, Canada; Faculty of Medicine, University of Ottawa, Ottawa, ON, Canada; ICES uOttawa, Ottawa, ON, Canada; Kingston Health Sciences Centre, Kingston, ON, Canada
License: © 2025 The Author(s) CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.jpedcp.2025.200151 | PubMed: 40584565 | PMC: PMC12206123
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (270 KB)
Cannabis is one of the most widely used drugs in Canada, and its use has steadily increased across many subpopulations, including pregnant women.ref. bib1,ref. bib2 Among Canadians, between 2% and 5% of women self-report cannabis use in pregnancy.ref. bib3 However, rates are substantially higher among women in certain age groups,ref. bib4 and self-reports may underestimate use due to the stigma around disclosure in pregnancy.ref. bib5 Available evidence suggests that pregnant individuals may use cannabis as a way to address health concerns, including pain, nausea, anxiety, depression, and other problems.ref. 6, ref. 7, ref. 8, ref. 9 In our previous qualitative interviews with patients, we noted that individuals who regularly consume cannabis before conception may be more likely to continue their use, either out of habit or to maintain a sense of normalcy throughout their pregnancy.ref. bib10
Cannabinoids can reach the fetal bloodstream via the placenta.ref. bib11,ref. bib12 These cannabinoids can disrupt the fetal endocannabinoid system, which is active from early stages of embryonic development and plays a critical role in neurodevelopment into adulthood. Due to its lipophilic nature, the principal psychoactive component of cannabis, delta-9 tetrahydrocannabinol, has a long half-life, which results in extended exposure to fetal tissues even after the mother stops consuming cannabis.ref. bib13,ref. bib14 Accumulating evidence suggests that cannabis use during pregnancy may be associated with adverse perinatal and neonatal outcomes, including stillbirth,ref. bib15,ref. bib16 preterm birth,ref. bib15,ref. 17, ref. 18, ref. 19, ref. 20 growth restrictionsref. bib17,ref. 21, ref. 22, ref. 23, and increased admission to the neonatal intensive care unit.ref. bib17,ref. bib18,ref. bib20,ref. bib24 These outcomes are often associated with more extended hospital stays after delivery and more complex health and social needs throughout early childhood.ref. bib25 However, little is known about the patterns of long-term use of health services of children prenatally exposed to cannabis in Canada. Using Ontario’s population-based birth registry and linked health administrative databases, we aimed to compare the rates of long-term health services use among offspring associated with prenatal cannabis use vs no use.
Methods
Study Design
We conducted a population-based retrospective cohort study of all live, singleton births to Ontario, Canada residents, between April 1, 2007, and March 31, 2012. An analysis examining child neurodevelopmental outcomes in a cohort with follow-up from 18 months was previously published.ref. bib26 This paper describes the use of health services among children with and without prenatal cannabis exposure using the entire birth cohort from birth to age 10. We report the study according to the REporting of studies Conducted using Observational Routinely collected Data reporting guidelines.ref. bib27
Data Sources
The study was conducted at Institute for Clinical Evaluative Sciences (ICES), an independent, nonprofit research institute whose legal status under Ontario’s health information privacy law allows it to collect and analyze health care and demographic data without consent for health system evaluation and improvement. We used the Better Outcomes Registry & Network (BORN, www.bornontario.ca) Niday Perinatal Database to define the birth cohort. The BORN Niday Perinatal Database includes a historical subset of the BORN registry, covering perinatal data from hospital births in Ontario from 2006 to 2012. Routine data collected in BORN includes information on maternal demographics and health behaviors, pre-existing health problems, obstetric complications, and birth outcomes. Data were collected from medical records, clinical forms, and patient interviews. In addition to the cohort, prenatal cannabis exposure and covariates related to pregnancy and birth outcomes were ascertained from this database. The birth cohort was subsequently linked to health administrative databases using unique encoded identifiers and analyzed at ICES. The study was approved by the research ethics boards of the Ottawa Health Science Network and the Children’s Hospital of Eastern Ontario.
Study Population
The study cohort was derived by accessing multiple databases that record the provision of health care in Ontario (Supplemental Appendix). We included all hospital births from 20 weeks of gestation weighing at least 500g and occurring in the province between April 1, 2007 and March 31, 2012. Infants were followed through linked databases until March 31, 2017, death or migration out of province using the ICES Registered Persons Database. We excluded mothers older than 50, those ineligible for the Ontario Health Insurance Plan (OHIP) during pregnancy, individuals who did not reside in Ontario for 2 years before pregnancy or had multifetal pregnancies (Supplemental Table S1; available at www.jpeds.com). Infants were excluded if they were stillborn, did not have a valid health card number at birth or within 60 days, were born to non-Ontario residents, or due to missing gestational age or incomplete database linkage.
Exposure
Maternal cannabis use in pregnancy included either therapeutic or recreational use. Cannabis use in the current pregnancy was reported to providers during 1st trimester visits, which usually occur by the 11th gestational weekref. bib26 but could also have been identified from clinical histories at admission for labor and delivery. At the first prenatal visit, providers complete a standardized perinatal record, which includes information about cannabis and substance use in the current pregnancy. Data from the perinatal and labor and delivery records were abstracted into the BORN Niday Perinatal Database. Cannabis use reporting in the perinatal database was well validated against clinical records.ref. bib28 We found that BORN had a sensitivity of 97% (95% confidence interval [CI]: 93%-99%) and a specificity of 94% (95% CI: 91%-96%) compared to clinical records, and the positive predictive value was 90% (95% CI: 85% – 94%).ref. bib18
Outcomes
The primary outcome was the number of primary care visits during the follow-up period, which was identified using the OHIP claims database. Primary care visits were defined as any encounter with a family physician or pediatrician based on provider codes (Supplemental Table S2; available at www.jpeds.com). The secondary outcomes included the number of outpatient psychiatrist visits, identified using provider codes in the OHIP database, emergency department (ED) visits and inpatient hospitalizations, determined from the NACRS and DAD, respectively during the follow-up period (Supplemental Appendix; available at www.jpeds.com). If an ED visit led to hospitalization, it was counted as one ED visit and one hospitalization to reflect the frequency of services used.
Covariates
Covariates of interest included maternal age, education level, prenatal substance use (including alcohol, cocaine, hallucinogens, methadone, opioids, and prescribed medication), rurality, household income, parity, antenatal care provider status, smoking, diagnosis of mood/anxiety disorder, diabetes, hypertension, asthma, and heart conditions. Maternal age was categorized into: <20, 20-24, 25-29, 30-34 and 35 and older. Education was categorized into quartiles, with one representing the lowest and four representing the highest level. Prenatal consumption of alcohol, cocaine, hallucinogens, methadone, opioids, or prescribed medication (eg, oxycodone, codeine) were each dichotomized (yes/no) and restricted to any consumption during the gestational period. Household income was categorized into quantiles, with one representing the lowest and five representing the highest level. Parity was categorized into nulliparous, primiparous, or multiparous. Antenatal care provider status, maternal smoking status and pre-existing maternal conditions, including mood/anxiety disorder, diabetes, hypertension, asthma, and heart conditions (eg, coronary heart disease, arrythmia), were each dichotomized (yes/no). Mood/anxiety disorder was defined as any psychiatric diagnosis recorded in the last two years before the last menstrual period.
Statistical Analyses
We conducted descriptive analyses to describe the differences in demographic and clinical characteristics between mothers who consumed cannabis during pregnancy and those who abstained (Table I). This analysis was performed again after applying coarsened exact matching (CEM). We used CEM to reduce bias and imbalance in covariates across cannabis use.ref. bib29 To achieve a balanced dataset, cannabis users were 1:k matched to nonusers within categories of the covariates mentioned above. Weights were applied to equalize the number of cannabis-using and non-using subjects in each stratum. Standardized mean differences (SMDs) were used to assess the balance in the distribution of baseline covariates between exposed and unexposed subjects before and after matching.ref. bib30 An absolute SMD above 0.10 was considered a meaningful imbalance in the covariate distribution between the groups.
Table I: Baseline Characteristics of the Original and Matched Cohorts, Separated by Exposure Status and Compared Using Standardized Differences
| Characteristic (%) | Birth cohort by prenatal cannabis exposure (n = 508 025) | CEM cohort by prenatal cannabis exposure (n = 175 700) | ||||
|---|---|---|---|---|---|---|
| Unexposed n = 504 777 | Exposed n = 3248 | Standardized mean difference | Unexposed n = 173 269 | Exposed n = 2431 | Standardized mean difference | |
| Maternal age, mean (SD) | 30 (27-34) | 23 (19-27) | 1.11 | 23.6 (5.4) | 23.4 (5.5) | 0.02 |
| Maternal age (categories) | ||||||
| <20 | 18 199 (3.6%) | 843 (26.0%) | 0.66 | 47 327 (27.3%) | 664 (27.3%) | 0.00 |
| 20-24 | 63 600 (12.6%) | 1183 (36.4%) | 0.58 | 65 288 (37.7%) | 916 (37.7%) | 0.00 |
| 25-29 | 138 307 (27.4%) | 723 (22.3%) | 0.12 | 36 421 (21.0%) | 511 (21.0%) | 0.00 |
| 30-34 | 173 555 (34.4%) | 327 (10.1%) | 0.61 | 16 108 (9.3%) | 226 (9.3%) | 0.00 |
| 35+ | 111 116 (22.0%) | 172 (5.3%) | 0.50 | 8125 (4.7%) | 114 (4.7%) | 0.00 |
| Maternal education, mean (SD)∗ | 0.51 ± 0.13 | 0.43 ± 0.12 | 0.63 | 0.43 ± 0.12 | 0.43 ± 0.12 | 0.00 |
| Maternal education (quartiles)∗ | ||||||
| 1 (lowest) | 4022 (0.8%) | 73 (2.2%) | 0.12 | 82 465 (47.6%) | 1157 (47.6%) | 0.00 |
| 2 | 119 353 (23.6%) | 1475 (45.4%) | 0.47 | 47 184 (27.2%) | 662 (27.2%) | 0.00 |
| 3 | 130 826 (25.9%) | 895 (27.6%) | 0.04 | 2780 (1.6%) | 39 (1.6%) | 0.00 |
| 4 (highest) | 15 902 (3.2%) | 69 (2.1%) | 0.06 | 38 452 (22.2%) | 536 (22.1%) | 0.00 |
| Missing | 234 674 (46.5%) | 736 (22.7%) | 0.52 | 2388 (1.4%) | 37 (1.5%) | 0.01 |
| Pre-existing maternal conditions | ||||||
| Diabetes | 10 715 (2.1%) | 55 (1.7%) | 0.03 | 1426 (0.8%) | 20 (0.8%) | 0.00 |
| Hypertension | 14 041 (2.8%) | 29 (0.9%) | 0.14 | 927 (0.5%) | 13 (0.5%) | 0.00 |
| Asthma | 23 197 (4.6%) | 405 (12.5%) | 0.28 | 18531 (10.7%) | 260 (10.7%) | 0.00 |
| Heart disease | 2722 (0.5%) | 24 (0.7%) | 0.03 | 642 (0.4%) | 9 (0.4%) | 0.00 |
| Maternal psychiatric disorders | ||||||
| Mood/anxiety disorders | 136 537 (27.0%) | 1449 (44.6%) | 0.37 | 71 560 (41.3%) | 1004 (41.3%) | 0.00 |
| Substance use disorder | 7750 (1.5%) | 604 (18.6%) | 0.59 | 19 957 (11.5%) | 280 (11.5%) | 0.00 |
| Maternal substance use history | ||||||
| Alcohol | 453 (0.1%) | 129 (4.0%) | 0.28 | 1354 (0.8%) | 19 (0.8%) | 0.00 |
| Cocaine | 674 (0.1%) | 450 (13.9%) | 0.56 | 5916 (3.4%) | 83 (3.4%) | 0.00 |
| Hallucinogens | 59 (0.0%) | 78 (2.4%) | 0.22 | 71 (0.0%) | <6† | 0.00 |
| Methadone | 1089 (0.2%) | 149 (4.6%) | 0.29 | 2922 (1.7%) | 41 (1.7%) | 0.00 |
| Opioids | 481 (0.1%) | 169 (5.2%) | 0.32 | 1069 (0.6%) | 15 (0.6%) | 0.00 |
| Prescription medication | 14 953 (3.0%) | 371 (11.4%) | 0.33 | 12 259 (7.1%) | 172 (7.1%) | 0.00 |
| Rurality | ||||||
| Urban | 454 714 (90.1%) | 2667 (82.1%) | 0.23 | 146 256 (84.4%) | 2052 (84.4%) | 0.00 |
| Rural | 50 026 (9.9%) | 580 (17.9%) | 0.23 | 27 013 (15.6%) | 379 (15.6%) | 0.00 |
| Income quintile | ||||||
| 1 (lowest) | 106 072 (21.0%) | 1454 (44.8%) | 0.52 | 80 826 (46.6%) | 1134 (46.6%) | 0.00 |
| 2 | 99 429 (19.7%) | 741 (22.8%) | 0.08 | 40 128 (23.2%) | 563 (23.2%) | 0.00 |
| 3 | 104 029 (20.6%) | 425 (13.1%) | 0.20 | 21 953 (12.7%) | 308 (12.7%) | 0.00 |
| 4 | 108 515 (21.5%) | 342 (10.5%) | 0.30 | 17 035 (9.8%) | 239 (9.8%) | 0.00 |
| 5 (highest) | 84 267 (16.7%) | 231 (7.1%) | 0.30 | 11 404 (6.6%) | 160 (6.6%) | 0.00 |
| Missing | 2465 (0.5%) | 55 (1.7%) | 0.12 | 1924 (1.1%) | 27 (1.1%) | 0.00 |
| Parity | ||||||
| Nulliparous | 214 290 (42.5%) | 1856 (57.1%) | 0.30 | 106 199 (61.3%) | 1490 (61.3%) | 0.00 |
| Primiparous | 182 311 (36.1%) | 727 (22.4%) | 0.31 | 36 921 (21.3%) | 518 (21.3%) | 0.00 |
| Multiparous | 105 010 (20.8%) | 654 (20.1%) | 0.02 | 29 737 (17.2%) | 415 (17.1%) | 0.00 |
| Missing | 3166 (0.6%) | 11 (0.3%) | 0.04 | 412 (0.2%) | 8 (0.3%) | 0.02 |
| Antenatal care provider | ||||||
| No | 2939 (0.6%) | 100 (3.0%) | 0.19 | 1853 (1.1%) | 26 (1.1%) | 0.00 |
| Yes | 484 800 (96.0%) | 3042 (93.7%) | 0.11 | 168 351 (97.1%) | 2362 (97.1%) | 0.00 |
| Missing | 17 038 (3.4%) | 106 (3.3%) | 0.01 | 3065 (1.8%) | 43 (1.8%) | 0.00 |
| Maternal smoking | ||||||
| No | 421 957 (83.6%) | 594 (18.3%) | 1.73 | 132 072 (76.2%) | 1853 (76.2%) | 0.00 |
| Yes | 58 921 (11.7%) | 2539 (78.2%) | 1.8 | 4918 (2.8%) | 69 (2.8%) | 0.00 |
| Missing | 23 899 (4.7%) | 115 (3.5%) | 0.06 | 36 279 (20.9%) | 509 (20.9%) | 0.00 |
| Obstetric complications‡ | ||||||
| Placental abruption | 2703 (0.5%) | 44 (1.4%) | 0.08 | 1366 (0.8%) | 34 (1.4%) | 0.06 |
| Placenta previa | 3231 (0.6%) | 16 (0.5%) | 0.02 | 676 (0.4%) | 11 (0.5%) | 0.01 |
| Pre-eclampsia | 9633 (1.9%) | 62 (1.9%) | <0.01 | 3209 (1.9%) | 49 (2.0%) | 0.01 |
| Eclampsia | 177 (0.0%) | <6† | 0.01 | 112 (0.1%) | <6† | 0.01 |
| Gestational diabetes | 23 903 (4.7%) | 83 (2.6%) | 0.12 | 4702 (2.7%) | 59 (2.4%) | 0.02 |
| Gestational hypertension | 17 479 (3.5%) | 104 (3.2%) | 0.01 | 6458 (3.7%) | 71 (2.9%) | 0.05 |
| Gestational age at delivery‡ | ||||||
| Preterm birth (<37 week) | 31 066 (6.2%) | 419 (12.9%) | 0.23 | 13 819 (8.0%) | 283 (11.6%) | 0.12 |
| Full-term birth (≥37 weeks) | 473 711 (93.8%) | 2829 (87.1%) | 0.23 | 159 450 (92.0%) | 2148 (88.4%) | 0.12 |
∗ Measured using census data as the neighborhood proportion of people with a postsecondary education degree. Percentages were rounded to the nearest decimal place. Area-level median family income quintiles were extracted from the Canadian Census using patient postal codes mapped to standard geographical units for census tracts and dissemination areas. Gestational age was estimated by first-trimester ultrasound or menstrual dating.
Poisson regression was used to compute rate ratios and 95% CIs to assess the differences in the rates of long-term health service use between children with and without prenatal cannabis exposure. We first fit unadjusted models on the full sample, followed by adjusted analyses using the matched sample, and finally, fully-adjusted models within the matched sample. Using the matched sample, fully adjusted models further accounted for obstetric complications, including placental abruption, placenta previa, pre-eclampsia, eclampsia, gestational diabetes, gestational hypertension, and preterm birth. Subgroup analyses were performed using a priori-specified factors associated with prenatal cannabis use, which may also be intermediate variables affecting health service use.ref. bib4,ref. bib18 These were preterm birth (born <37 or ≥37 weeks gestation), area-level median household income (high vs low income) and substance use (any substance use vs no use).
Results
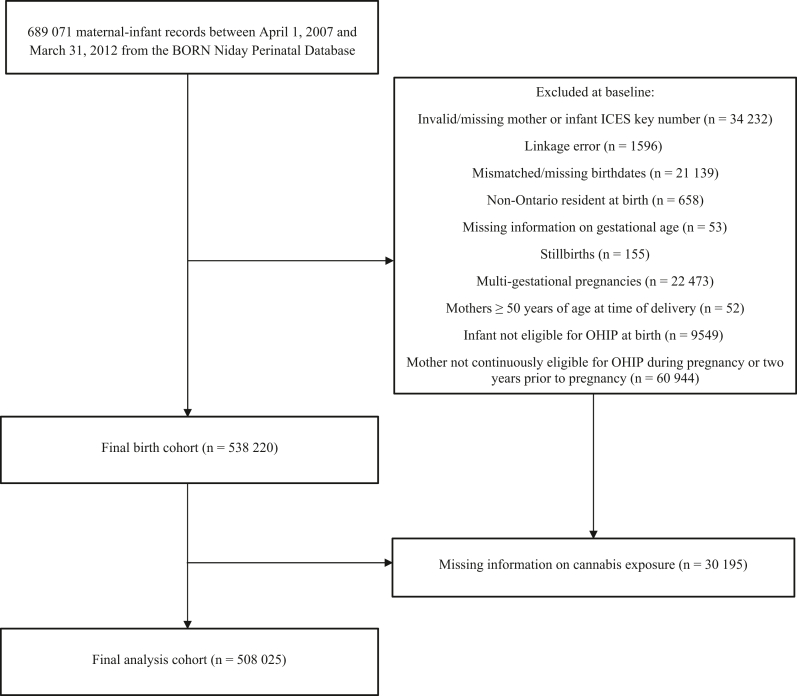
The initial study sample comprised 689 071 pregnancy records. In total, 181 046 records were excluded due to missing information, linkage errors, being a non-Ontario resident at birth, stillbirth, multifetal pregnancies, OHIP ineligibility, maternal age above 50 years at delivery or missing information on cannabis, yielding a final analytic sample of 508 025 (Figure). Children were followed up for a median of 7 years. Total person-years were 3 713 219.88 and 22 606.70 among the unexposed and exposed groups, respectively (Supplemental Table S3; available at www.jpeds.com).
Overall, 3248/508 025 (0.6%) children were exposed to cannabis in utero, and the prevalence was highest among mothers younger than 20 (4.4% = 843/19 042). Before matching, mothers who used cannabis during pregnancy were younger (mean age 23 years vs 30 years; SMD: 1.11), less educated (SMD: 0.63) and of lower income (44.8% vs 21.0%; SMD: 0.52) compared to nonusers (Table I). After applying CEM and removing unmatched observations, our cohort included 175 700 infants (2431 with cannabis use and 173 269 without). All measured baseline covariates used in matching were well-balanced (SMD < 0.1) across exposure groups (Table I). After matching, preterm birth remained more prevalent among mothers with cannabis use compared to nonusers (11.6% vs 8.0%, respectively).
Association Between Prenatal Cannabis Exposure and Health Service Use
After adjusting for obstetrical complications in the matched cohort, we found that offspring exposed to cannabis during the gestational period had decreased rates of primary care visits compared to those unexposed (additionally adjusted RR [a2RR]: 0.86, 95% CI: 0.84-0.87). However, they had increased rates of visiting an outpatient psychiatrist (a2RR: 1.29, 95% CI: 1.00-1.66), the ED (a2RR: 1.05, 95% CI: 1.03-1.08) and being hospitalized (a2RR: 1.12, 95% CI: 1.04-1.20) (Table II). The mean frequency of outpatient psychiatrist visits across the cohort was low among both unexposed (mean = 0.07, SMD: 1.07) and exposed (mean = 0.14, SMD: 1.09) infants (Supplemental Table S3; available at www.jpeds.com). Similarly, hospitalizations were infrequent among the cohort’s unexposed (mean = 0.29, SMD: 0.97) and exposed (mean = 0.37, SMD: 1.08) children. In contrast, offspring in the cohort frequently utilized primary care and ED services throughout follow-up.
Table II: Association Between Prenatal Cannabis Exposure and Rate of Childhood Health Services Use by Service
| Outcomes | Crude RR (95% CI) (n = 508 025) | Adjusted RR [a1RR] (95% CI)∗ (n = 175 700) | Additionally adjusted RR [a2RR] (95% CI)∗† (n = 175 700) |
|---|---|---|---|
| Primary care visits (family physicians and pediatricians) | 0.79 (0.77-0.81) | 0.85 (0.84-0.87) | 0.86 (0.84-0.87) |
| Outpatient psychiatrist visits | 1.94 (1.40-2.69) | 1.30 (1.01-1.67) | 1.29 (1.00-1.66) |
| Emergency department visits | 1.46 (1.40-1.51) | 1.06 (1.04-1.09) | 1.05 (1.03-1.08) |
| In-patient hospitalizations | 1.41 (1.30-1.54) | 1.19 (1.11-1.28) | 1.12 (1.04-1.20) |
RR, rate ratio; 95% CI, 95% confidence interval.
Stratified Analysis by Preterm Birth, Other Substance Use, and Area-Level Income
Among children born preterm (n = 31 485), the unadjusted analysis showed a similar pattern across health service outcomes compared to the overall population. However, after matching and adjustment for obstetrical complications, those exposed to prenatal cannabis had lower rates of primary care visits (a2RR: 0.82, 95% CI: 0.78-0.87) compared to no cannabis, while the rates of other service use were not statistically different (Table III). Among full-term infants, the cannabis-outcome associations closely aligned with the overall sample. Further, among offspring of women without additional substance use (eg, no alcohol, tobacco, cocaine, hallucinogens, methadone, or opioids), we found the rates of health service use associated with prenatal cannabis exposure remained similar to that of the overall cohort for all outcomes.
Table III: Association Between Prenatal Cannabis Exposure and Childhood Health Services Outcomes, by Preterm Birth, Substance Use and Income
| Cohort characteristic | Outcomes | Crude RR (95% CI) (n = 508 025) | Adjusted RR [a1RR] (95% CI)∗ (n = 175 700) | Additionally adjusted RR [a2RR] (95% CI)∗† (n = 175 700) |
|---|---|---|---|---|
| Preterm birth | ||||
| Primary care visits (family physicians and pediatricians) | 0.73 (0.68-0.79) | 0.82 (0.78-0.87) | 0.82 (0.78-0.87) | |
| Outpatient psychiatrist visits | 1.24 (0.54-2.82) | 0.96 (0.46-2.01) | 0.96 (0.46-2.02) | |
| Emergency department visits | 1.24 (1.12-1.38) | 0.98 (0.91-1.05) | 0.98 (0.91-1.05) | |
| In-patient hospitalizations | 1.15 (0.96-1.38) | 0.96 (0.81-1.12) | 0.94 (0.80-1.11) | |
| Full-term birth | ||||
| Primary care visits (family physicians and pediatricians) | 0.79 (0.77-0.81) | 0.86 (0.85-0.87) | 0.86 (0.85-0.87) | |
| Outpatient psychiatrist visits | 2.02 (1.42-2.88) | 1.34 (1.02-1.75) | 1.33 (1.01-1.74) | |
| Emergency department visits | 1.47 (1.41-1.53) | 1.06 (1.04-1.09) | 1.06 (1.04-1.09) | |
| In-patient hospitalizations | 1.35 (1.22-1.48) | 1.16 (1.07-1.26) | 1.16 (1.07-1.26) | |
| No use of other substances‡ | ||||
| Primary care visits (family physicians and pediatricians) | 0.78 (0.76-0.81) | 0.84 (0.83-0.86) | 0.84 (0.83-0.86) | |
| Outpatient psychiatrist visits | 1.98 (1.34-2.93) | 1.32 (1.01-1.72) | 1.31 (1.00-1.71) | |
| Emergency department visits | 1.43 (1.37-1.50) | 1.04 (1.02-1.07) | 1.04 (1.01-1.06) | |
| In-patient hospitalizations | 1.34 (1.21-1.49) | 1.16 (1.08-1.26) | 1.09 (1.01-1.18) | |
| Income | ||||
| Quintile: 1,2 (low income)Quintile: 3,4, 5 (higher income) | Primary care visits (family physicians and pediatricians) | 0.78 (0.76-0.81) | 0.87 (0.85-0.89) | 0.87 (0.85-0.89) |
| Outpatient psychiatrist visits | 1.53 (1.04-2.24) | 1.00 (0.68-1.45) | 0.99 (0.68-1.44) | |
| Emergency department visits | 1.30 (1.25-1.37) | 1.01 (0.98-1.04) | 1.00 (0.97-1.04) | |
| In-patient hospitalizations | 1.31 (1.18-1.46) | 1.12 (1.03-1.23) | 1.06 (0.97-1.16) | |
| Primary care visits (family physicians and pediatricians) | 0.83 (0.79-0.86) | 0.85 (0.83-0.87) | 0.85 (0.83-0.87) | |
| Outpatient psychiatrist visits | 2.76 (1.51-5.01) | 2.46 (1.81-3.35) | 2.49 (1.83-3.38) | |
| Emergency department visits | 1.59 (1.49-1.70) | 1.17 (1.13-1.21) | 1.16 (1.12-1.21) | |
| In-patient hospitalizations | 1.37 (1.18-1.60) | 1.29 (1.13-1.46) | 1.21 (1.06-1.37) | |
RR, rate ratio; 95% CI, 95% confidence interval.
Finally, when stratifying by lower and higher income levels, both strata had similar rates of primary care visits. The rates of outpatient psychiatrist visits among those prenatally exposed to cannabis and whose families are in the upper-income quintiles were over two times higher (a2RR: 2.49, 95% CI: 1.83-3.38) than those of the same income quintiles who were unexposed. In contrast, the rate of outpatient psychiatrist visits among those of lower quintiles did not differ between those exposed or unexposed (a2RR: 0.99, 95% CI: 0.68-1.44). Similarly, prenatal cannabis use was associated with an increased rate of ED visits (a2RR: 1.16, 95% CI: 1.12-1.21) and hospitalizations (a2RR: 1.21, 95% CI: 1.06 -1.37), only in the higher income strata.
Discussion
In this study, prenatal cannabis use in Ontario was associated with increased rates of ED visits, hospitalizations and psychiatrist visits for offspring within the first ten years since birth. In contrast, the rate of primary care access was roughly 15% lower among these children compared to children with no prenatal cannabis exposure. Prenatal cannabis use was associated with lower rates of primary care visits regardless of income level or prematurity. However, associations with other health services differed by income level and prematurity.
In line with this, Avalos et alref. bib31 found that in-utero exposure to cannabis was associated with a significantly increased risk of missed well-child visits in the first 3 years of life of the offspring, in the United States. Decreased rates of primary care visits in childhood may indicate that, where maternal cannabis use is identified, children and their families face increased barriers to accessing pediatricians or primary care providers due to factors such as fear of stigmatization,ref. bib9,ref. bib32 reduced access to physicians,ref. bib33 inability to take time off work or mental health difficulties.ref. bib34 Not having a primary care physician or having difficulty accessing primary care has been found to increase the likelihood of visiting an emergency room and hospital admissions,ref. bib35,ref. bib36 which was observed in this cohort. Furthermore, unfavorable psychosocial and socioeconomic determinants of health are more prevalent among mothers who use cannabis during pregnancy.ref. bib4,ref. bib26 As a result, these mothers may experience disparities in health services use and quality,ref. 37, ref. 38, ref. 39 and this may persist as they seek appropriate care for their children after birth.
Previous studies have found that prenatal exposure to cannabis through maternal use can cause decreases in cognitive function among offspring.ref. 40, ref. 41, ref. 42 Using a similar cohort, we found that the risk of autism was 50% higher for children exposed to cannabis in utero compared to those unexposed.ref. bib26 A study from Australia reported a 3-fold increase in risk of autism diagnosis among offspring of mothers diagnosed with cannabis use disorder.ref. bib43 Evidence is still emerging, and several cohort studies have been null. However, a 2024 meta-analysis of 4 studies and ∼200 000 participants found a pooled rate ratio of 1.3 (95% CI: 1.03-1.64) for the association between in-utero cannabis exposure and autism in offspring. Psychological perturbations caused by exogenous cannabinoids during development may explain the higher rates of psychiatric visits seen among those in this cohort who were prenatally exposed to cannabis. Consistent with our findings, prenatal cannabis exposure is typically more common among infants born to lower income families.ref. bib4,ref. bib44 Interestingly, outpatient psychiatrist visit rates did not differ by cannabis use among individuals of the lower income quintiles. In contrast, rates were much higher among higher income children with prenatal exposure. This difference might be explained by the difference in socioeconomic position, where parents in higher income quintiles are more aware of the risk of developmental conditions and have more flexibility to take time off work or more resources to access private care and pay for private services through private insurance. Furthermore, children living in neighborhoods of higher median income may be placed in schools that are more aware and possess the resources to monitor for behavioral problems or learning disabilities, which may direct them to seek health care specialists if necessary.
Limitations
In this study, cannabis exposure was a binary variable and did not account for variation in frequency or intensity of exposure during the prenatal period, which could differentially impact the development of exposed children. Data on the use of cannabis in pregnancy are self-reported and collected through disclosure to prenatal care providers using a standardized form during routine perinatal care. Cannabis use data in the BORN perinatal database are highly accurate when validated against clinical records, although similar biases may exist in both sources.ref. bib18 Our cohort was assembled in an early period when the prevalence of cannabis use was lower and the methods for cannabis data collection were less well-established. Misclassification bias remains due to under-reporting of cannabis use in pregnancy, which may arise from social stigma or fear of involvement of child protective services.ref. 45, ref. 46, ref. 47 However, given that the exposure may be under-reported in this cohort, the associations we describe here may underestimate rather than exaggerate the relationship between cannabis use in pregnancy and child health service use. Our cohort also predates the legalization of nonmedical cannabis in Canada (October 2018), although the data are highly relevant for understanding prelegalization patterns of exposure and health service use. Further, data were collected before some increases in potency and product availability. However, the delta-9 tetrahydrocannabinol potency in cannabis products during this time was already beginning to increase compared to prior decades.ref. bib48 Ongoing surveillance and replication using postlegalization data are warranted to assess how changing regulatory and social contexts may influence these associations. Children were followed for a maximum of 10 years, with a median follow-up of 7 years. Since prenatal cannabis use often results in health conditions that require more medical attention immediately after birth rather than later in childhood, health service use patterns may differ by different timing in the life course. Previous studies have found that children exposed to cannabis during gestation are at a higher risk of neonatal intensive care unit admission and transfer.ref. bib17,ref. bib18,ref. bib20,ref. bib24 The number of hospitalizations, for example, is likely disproportionately higher within the first months since birth. Given the long-term follow-up, there is also a possibility of unmeasured confounders that may occur later in life. Future research should focus on assessing service use in different age intervals for a deeper understanding of the pattern of service use throughout childhood.
Interpretation and Generalizability
This study is among the first in Canada to evaluate the relationship between prenatal cannabis exposure and the frequency of health care visits in childhood up to 10 years of age. These findings underscore that prenatal cannabis use may be associated with ongoing changes to health care use patterns in offspring past infancy. Clinical practice should continue to reinforce the importance of safe and respectful dialog between expectant mothers and health care professionals regarding cannabis use during pregnancy. Early screening and identification of prenatal cannabis use, along with appropriate follow-up, may ensure timely assessment of affected children who may be more vulnerable to adverse social and health outcomes. We recommend developing publicly accessible guidelines for the prenatal consumption of cannabis, such as those recently released by the American Society of Clinical Oncology for cancer patients and physicians,ref. bib49 which provides strategies for open and nonjudgmental communication between clinicians and patients around cannabis use. Such guidelines may be used to help reduce potentially harmful cannabis consumption and potentially contribute to reducing the long-term impacts of prenatal cannabis use on health service use.
Data Statement
The dataset used in this research is held securely in coded form at ICES. Requests for data access can be made to ICES at https://www.ices.on.ca/services-for-researchers/submit-a-request/. The complete dataset creation plan is available from the authors upon request.
Disclaimers
This study is based on data from the Better Outcomes Registry & Network (“BORN”), part of the Children’s Hospital of Eastern Ontario. Parts of this material are based on data and information compiled and provided by the Canadian Institute for Health Information and the Ontario Ministry of Health. The analyses, conclusions, opinions, and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred. We use data adapted from the Statistics Canada Postal CodeOM Conversion File, based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from Canada Post Corporation and Statistics Canada. This does not constitute an endorsement by Statistics Canada of this product.
CRediT authorship contribution statement
Gabrielle Pratt Tremblay: Writing – original draft, Investigation. Arum Han: Writing – original draft, Investigation. Ewa Sucha: Writing – review & editing, Formal analysis, Data curation. Helen Hsu: Writing – review & editing, Investigation. Jessy Donelle: Writing – review & editing, Formal analysis, Data curation. Daniel J. Corsi: Writing – review & editing, Supervision, Methodology, Investigation, Funding acquisition, Conceptualization.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. This study was funded by the Canadian Institutes of Health Research.
References
- Health Canada, Cannabis use (non-medical) in Canada. 2024
- G. Pratt Tremblay, S. Dimanlig-Cruz, A. Dion, D.J. Corsi. Trends in prenatal substance use across Ontario, Canada. JAMA Netw Open, 2025
- S. Badowski, G. Smith. Cannabis use during pregnancy and postpartum. Can Fam Physician, 2020. [PubMed]
- D.J. Corsi, H. Hsu, D. Weiss, D.B. Fell, M. Walker. Trends and correlates of cannabis use in pregnancy: a population-based study in Ontario, Canada from 2012 to 2017. Can J Public Health, 2019. [PubMed]
- K.C. Young-Wolff, V. Sarovar, L.Y. Tucker. Validity of self-reported cannabis use among pregnant females in Northern California. J Addict Med, 2020. [PubMed]
- K. Bartlett, K. Kaarid, N. Gervais. Pregnant Canadians’ perceptions about the transmission of cannabis in pregnancy and while breastfeeding and the impact of information from health care providers on discontinuation of use. J Obstet Gynaecol Can, 2020. [PubMed]
- K.R. Skelton, E. Donahue, S.E. Benjamin-Neelon. Measuring cannabis-related knowledge, attitudes, perceptions, motivations, and influences among women of reproductive age: a scoping review. BMC Womens Health, 2022. [PubMed]
- L.B. McKenzie, S.A. Keim, M.A. Klebanoff. Risk perceptions about cannabis use and receipt of health-related information during pregnancy. Am J Health Promot, 2022. [PubMed]
- D.D. King, C.J. Gill, C.S. Cadieux, N. Singh. The role of stigma in cannabis use disclosure: an exploratory study. Harm Reduct J, 2024. [PubMed]
- D.J. Corsi, G. Pratt Tremblay. Cannabis use in Canada: current policies, prevalence trends, socioeconomic and demographic correlates 2024
- K.A. Richardson, A.K. Hester, G.L. McLemore. Prenatal cannabis exposure – the "first hit" to the endocannabinoid system. Neurotoxicol Teratol, 2016. [PubMed]
- J.R. Bailey, H.C. Cunny, M.G. Paule, W. Slikker. Fetal disposition of delta 9-tetrahydrocannabinol (THC) during late pregnancy in the rhesus monkey. Toxicol Appl Pharmacol, 1987. [PubMed]
- J. Friedrich, D. Khatib, K. Parsa, A. Santopietro, G.I. Gallicano. The grass isn’t always greener: the effects of cannabis on embryological development. BMC Pharmacol Toxicol, 2016. [PubMed]
- T. Sithisarn, D.T. Granger, H.S. Bada. Consequences of prenatal substance use. Int J Adolesc Med Health, 2012. [PubMed]
- S. Luke, J. Hutcheon, T. Kendall. Cannabis use in pregnancy in British Columbia and selected birth outcomes. J Obstet Gynaecol Can, 2019. [PubMed]
- M.W. Varner, R.M. Silver, H.C.J. Rowland. Association between stillbirth and illicit drug use and smoking during pregnancy. Obstet Gynecol, 2014. [PubMed]
- P. Koto, V.M. Allen, J. Fahey, S. Kuhle. Maternal cannabis use during pregnancy and maternal and neonatal outcomes: a retrospective cohort study. BJOG, 2022. [PubMed]
- D.J. Corsi, L. Walsh, D. Weiss. Association between self-reported prenatal cannabis use and maternal, perinatal, and neonatal outcomes. JAMA, 2019. [PubMed]
- S.Y. Leemaqz, G.A. Dekker, L.M. McCowan. Maternal marijuana use has independent effects on risk for spontaneous preterm birth but not other common late pregnancy complications. Reprod Toxicol, 2016. [PubMed]
- M.R. Hayatbakhsh, V.J. Flenady, K.S. Gibbons. Birth outcomes associated with cannabis use before and during pregnancy. Pediatr Res, 2012. [PubMed]
- D.M. Fergusson, L.J. Horwood, K. Northstone. Maternal use of cannabis and pregnancy outcome. BJOG, 2002. [PubMed]
- H. El Marroun, H. Tiemeier, E.A. Steegers. Intrauterine cannabis exposure affects fetal growth trajectories: the Generation R Study. J Am Acad Child Adolesc Psychiatry, 2009. [PubMed]
- N. Day, U. Sambamoorthi, P. Taylor. Prenatal marijuana use and neonatal outcome. Neurotoxicol Teratol, 1991. [PubMed]
- J.K. Gunn, C.B. Rosales, K.E. Center. Prenatal exposure to cannabis and maternal and child health outcomes: a systematic review and meta-analysis. BMJ Open, 2016
- D. Moster, R.T. Lie, T. Markestad. Long-term medical and social consequences of preterm birth. New Engl J Med, 2008. [PubMed]
- D.J. Corsi, J. Donelle, E. Sucha. Maternal cannabis use in pregnancy and child neurodevelopmental outcomes. Nat Med, 2020. [PubMed]
- E.I. Benchimol, L. Smeeth, A. Guttmann. The REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement. PLoS Med, 2015
- S. Dunn, A. Lanes, A.E. Sprague. Data accuracy in the Ontario birth Registry: a chart re-abstraction study. BMC Health Serv Res, 2019. [PubMed]
- S.M. Iacus, G. King, G. Porro. Causal inference without balance checking: coarsened exact matching. Polit Anal, 2012
- P.C. Austin. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun Stat Simulation Comput, 2009
- L.A. Avalos, N. Oberman, S.E. Alexeeff. Association between maternal prenatal cannabis use and missed child preventive care visits in an integrated health care delivery system in Northern California. Prev Med, 2023
- S. Daniels, S.P. Michelle, S. Tatiana, Z. Walsh. Physician communication and perceived stigma in prenatal cannabis use. J Psychoactive Drugs, 2023. [PubMed]
- C.M. Flood, B. Thomas, E. McGibbon. Canada’s primary care crisis: federal government response. Healthc Manage Forum, 2023. [PubMed]
- J. Rup, T.P. Freeman, C. Perlman, D. Hammond. Cannabis and mental health: prevalence of use and modes of cannabis administration by mental health status. Addict Behav, 2021
- R.H. Glazier, R. Moineddin, M.M. Agha. 2008
- D. Duong, L. Vogel. National survey highlights worsening primary care access. CMAJ, 2023
- M.R. Lavergne, A. Bodner, S. Allin. Disparities in access to primary care are growing wider in Canada. Healthc Manage Forum, 2023. [PubMed]
- M.E. Olah, G. Gaisano, S.W. Hwang. The effect of socioeconomic status on access to primary care: an audit study. CMAJ, 2013. [PubMed]
- M.P. McCabe, L. Leas. A qualitative study of primary health care access, barriers and satisfaction among people with mental illness. Psychol Health Med, 2008. [PubMed]
- G.L. McLemore, K.A. Richardson. Data from three prospective longitudinal human cohorts of prenatal marijuana exposure and offspring outcomes from the fetal period through young adulthood. Data Brief, 2016. [PubMed]
- P.A. Fried, B. Watkinson, R. Gray. Differential effects on cognitive functioning in 13- to 16-year-olds prenatally exposed to cigarettes and marihuana. Neurotoxicol Teratol, 2003. [PubMed]
- P.A. Fried. Prenatal exposure to marihuana and tobacco during infancy, early and middle childhood: effects and an attempt at synthesis. Arch Toxicol Suppl, 1995. [PubMed]
- A.W. Tadesse, G. Ayano, B.A. Dachew, K. Betts, R. Alati. Exposure to maternal cannabis use disorder and risk of autism spectrum disorder in offspring: a data linkage cohort study. Psychiatry Res, 2024
- A.H. Schempf, D.M. Strobino. Illicit drug use and adverse birth outcomes: is it drugs or context?. J Urban Health, 2008. [PubMed]
- L. Greaves, N. Poole. Victimized or validated? Responses to substance-using pregnant women. Can Woman Stud, 2004
- R. Stone. Pregnant women and substance use: fear, stigma, and barriers to care. Health Justice, 2015
- K.A. Yonkers, H.B. Howell, N. Gotman, B.J. Rounsaville. Self-report of illicit substance use versus urine toxicology results from at-risk pregnant women. J Subst Use, 2011. [PubMed]
- M.A. ElSohly, S. Chandra, M. Radwan, C.G. Majumdar, J.C. Church. A comprehensive review of cannabis potency in the United States in the last decade. Biol Psychiatry Cogn Neurosci Neuroimaging, 2021. [PubMed]
- I.M. Braun, K. Bohlke, D.I. Abrams. Cannabis and cannabinoids in adults with cancer: ASCO guideline. J Clin Oncol, 2024. [PubMed]
