Comprehensive pan-cancer analysis of 33 human cancers reveals immunotherapeutic value of focal adhesion tyrosine kinase
Abstract
The immune environment in tumors is the key factor affecting the survival and immunotherapeutic response of patients. This research aimed to explore the underlying association between focal adhesion tyrosine kinase (FAK/PTK2) and cancer immunotherapy in 33 human cancers. Gene expression data and clinical features of 33 cancers were retrieved from the Cancer Genome Atlas Database. The immunotherapy cohorts included GSE67501, GSE78220, and IMVIGOR210, which were derived from the comprehensive gene expression database or from previous studies. Clinical parameters including patient age, gender, survival rate, and tumor stage were analyzed to evaluate the prognostic value of FAK/PTK2. FAK/PTK2 activity was detected by single-sample gene set enrichment analysis and used to compare the difference between FAK/PTK2 transcriptome and protein expression levels. To better understand the role of FAK/PTK2 in cancer immunotherapy, we analyzed its correlations with tumor microenvironment and with immune processes/elements (e.g., immune cell infiltration, immunosuppressants, and stimulants) and major histocompatible complexes. Potential pathways associated with FAK/PTK2 signaling in cancers were also explored. Correlations between FAK/PTK2 and 2 immunotherapeutic biomarkers (tumor mutation load and microsatellite instability) were studied. Finally, the 3 independent immunotherapy cohorts were used to study the relationship between FAK/PTK2 and immunotherapeutic response. Although FAK/PTK2 is not closely associated with age (13/33), gender (5/33), or tumor stage (5/33) in any of the studied human cancers, it has potential prognostic value for predicting patient survival. Consistency between FAK/PTK2 activity and expression exists in some cancers (3/33). Generally, FAK/PTK2 is robustly correlated with immune cell infiltration, immune modulators, and immunotherapeutic markers. Moreover, high FAK/PTK2 expression is significantly related to immune-relevant pathways. However, FAK/PTK2 is not significantly correlated with the immunotherapeutic response. Research on the immunotherapeutic value of FAK/PTK2 in 33 human cancers provides evidence regarding the function of FAK/PTK2 and its role in clinical treatment. However, given the use of a bioinformatics approach, our results are preliminary and require further validation.
Article type: Research Article
Keywords: FAK/PTK2, immune checkpoint inhibitors, immunotherapeutic response, tumor microenvironment
Affiliations: Department of Oncology, The People’s Hospital of Jurong City, Jurong Hospital Affiliated to Jiangsu University, Zhenjiang, China; Department of Radiation Oncology, Osaka University Graduate School of Medicine, Osaka, Japan; Department of Radiation Center, Shanghai First Maternity and Infant Hospital. School of Medicine, Tongji University, Shanghai, China; Department of Radiotherapy, Jiangsu Provincial People’s Hospital, Nanjing, China.
License: Copyright © 2024 the Author(s). Published by Wolters Kluwer Health, Inc. CC BY 4.0 This is an open access article distributed under the Creative Commons Attribution License 4.0 (CCBY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1097/MD.0000000000037362 | PubMed: 38518034 | PMC: PMC10956984
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.5 MB)
1. Introduction
Although immune checkpoint inhibitors (ICI) targeting programmed cell death protein 1, its ligand 1 or cytotoxic T lymphocyte antigen 4 significantly prolong the overall survival of patients with different types of cancers, only a limited subset of patients are beneficiary.[1] Therefore, finding out reasonable and effective strategies to improve the efficacy of ICIs in cancerous patients is urgent. Understanding the genomic relevance of the response of ICIs will help to develop new biomarkers and therapies to enhance clinical response and expand the beneficiary population.[2,3]
Focal adhesions are important mediators in the interaction between the cytoskeleton and intracellular signaling molecules into the extracellular matrix through transmembrane receptors.[4,5] This communication is mediated by the nonreceptor cytoplasmic protein tyrosine kinase focal adhesion tyrosine kinase (FAK/PTK2).[6] This FAK/PTK2, located at the tip of chromosome 8q24.3,[7] is defined as the phosphorylated substrate of viral Src oncogene and anchored at the integrin-rich cell adhesion site, which was first discovered by Hanks et al[8] and Schaller et al[9] in the early 1990s. Initially, Weiner et al[10] found that FAK/PTK2 may be related to tumors. Moreover, FAK/PTK2 is overexpressed and activated in many types of advanced solid cancers, including colorectal,[11] ovarian,[12] esophageal,[13] and hepatocellular[14] carcinomas, and is associated with poor overall survival.[7,15] FAK/PTK2 reportedly plays an important role in tumor adhesion, diffusion, motility, invasion, metastasis, survival, angiogenesis, epithelial to mesenchymal transformation, cancer stem cells, and the tumor microenvironment (TME).[16–18]
The TME consists of various immune cells, mesenchymal-origin cells, and extracellular matrix,[19] which influence tumorigenesis at all stages by directly interacting with tumor cells.[20] FAK/PTK2 promotes antitumor immune evasion by increasing the count of regulatory T cells in tumors.[21] In myeloid-specific FAK/PTK2 knock-out mice, FAK/PTK2 can reduce the killing ability of neutrophils against pathogens.[22] However, after FAK/PTK2 was silenced in tumor cells or became less metastasitic through FAK/PTK2 pharmacological inhibition,[23,24] hematopoietic FAK/PTK2 in mice lacked a metastasis-promoting microenvironment.[25] Also, FAK/PTK2 is pivotal in macrophage invasion and movement.[26] These reports suggest that matrix FAK/PTK2 plays a regulatory role in tumor growth.
The advent of next-generation sequencing and large-scale genomics has shifted oncology research from single-gene analysis to pan-cancer analysis. The Cancer Genome Atlas (TCGA) based on epigenome, genomic, proteome, and transcriptome data from different cancers can be used to identify similarities and differences in important bioprocesses among different cancers.[27] Therefore, with the large-scale RNA sequencing data from TCGA, we performed a pan-cancer genome analysis of FAK/PTK2 from 33 cancer types. This study was aimed to evaluate the expression of FAK/PTK2 in different cancer types; its prognostic value in different tumors; its relationship with tumor immune characteristics, and its association with drug response. Specifically, the tumor immune characteristics include intraturmoral immune invasion, checkpoint markers, tumor mutation load (TMB), and microsatellite instability (MSI), which have been identified as potential biomarkers for predicting therapeutic response with ICIs.
2. Materials and methods
2.1. Data collection and statistical processing
The genomic and clinicopathological information of 33 cancers was obtained from TCGA (https://portal.gdc.cancer.gov/) and the University of California Santa Cruz Xena Explorer (cohort: TCGA pan-cancer). Somatic mutation data were acquired from TCGA. For the therapeutic cohort, a systematic search was performed to identify the immune checkpoint blockade cohorts, which can be publicly retrieved and reported with complete clinical information. Three immunotherapeutic cohorts were finally employed: advanced urothelial cancer with atezolizumab intervention (IMvigor210 cohort from Mariathasan et al[28]), metastatic melanoma with pembrolizumab treatment (GSE78220 cohort from the Gene Expression Omnibus), and renal cell carcinoma with nivolumab treatment (GSE67501cohort from Gene Expression Omnibus). Statistical Analysis R software (version 4.1.2) was used for all statistical analysis and graphical visualization. Wilcoxon test was performed based on the FAK/PTK2 expression of nonresponder and responder groups. The Student t test or 1-way one-way analysis of variance test was used to compare 2 or more groups of continuous variables with a normal distribution (including expression and activation of FAK/PTK2 in 33 cancers).
2.2. Clinical correlation between FAK/PTK2 expression and various cancers
With package Limma in R Studio, differential gene expressions were analyzed to determine whether expression varied between tumor and normal groups. The correlation between FAK/PTK2 expression and other clinical parameters (age, gender, and tumor stage) was also investigated. To explore the time-dependent prognostic value of FAK/PTK2 in 33 cancers, we performed univariate Cox regression analysis on package Survival in R. The studied survival outcomes included overall survival (OS; period from the start of treatment to death from any cause), disease-free survival (DFS; period from the start of treatment to recurrence or death from any cause), disease-specific survival (DSS: cancer survival in the absence of other causes of death), and progression-free survival (PFS: period from the start of treatment to progression or death from any cause). The hazard ratio >1 indicates that the exposure factor FAK/PTK2 expression is a promoting factor of positive events (death). Variations with P < .05 are considered significant.
2.3. Gene set enrichment analyses
Gene set enrichment analyses (GSEA) provide a robust way to analyze molecular profiling data. To further investigate the protein level of FAK/PTK2 in pan-cancers, we used 69 relevant genes that were significantly up-regulated after 3-MC agonist treatment and down-regulated after GNF-351 antagonist treatment in 8 cell lines.[29] FAK/PTK2 activity was detected by single-sample GSEA. After that, the difference in FAK/PTK2 activity between the normal and tumor groups was investigated. Then the mean values of expression and activity were calculated and arranged in 33 types of cancers, aiming to explore the potential features of FAK/PTK2 expression and activity.
2.4. Potential association between FAK/PTK2 expression and immune-related factors
The stromal score and immune score of each case were first calculated using the Estimation of Stromal and Immune cells in Malignant Tumour tissues using Expression data package, a tool for predicting tumor purity and the presence of infiltrating stromal/immune cells in tumor tissues.[30] The Estimation of Stromal and Immune cells in Malignant Tumour tissues using Expression data is based on single-sample GSEA and generates 3 final scores: the stromal score (indicating the presence of stromal cells in tumor tissues), the immune score (representing the infiltration of immune cells in tumor tissues), and tumor purity. Then the abundance of immune cell infiltration in low FAK/PTK2-expressing and high FAK/PTK2-expressing groups was estimated using CIBERSORT, a deconvolution algorithm that evaluates the proportions of 22 tumor-infiltrating lymphocyte subsets.[31] In short, the number of permutations was set to be 1000 and the samples with P < .05 in the cohort were eligible for further investigation. Correlations of FAK/PTK2 expression with TMB and MSI were also investigated, since these 2 indicators are closely associated with the immune response. TMB was defined as the total number of errors in somatic gene coding, base substitution, gene insertions, or deletions detected in every million bases. To calculate the TMB of each case, the total number of mutations counted was divided by the exome size (38 Mb). The MSI score of each TCGA cancer case was obtained from a previous study.[32] In addition, the underlying relationship between FAK/PTK2 expression and immunological modulators (immune inhibitors, immune stimulators, and MHC molecules) was explored via TISIDB (http://cis.hku.hk/TISIDB/index.php). The 4 most relevant results were then highlighted and presented in plots. Finally, to further investigate the relevant signaling pathways, we performed GSEA to identify differential pathways between the low FAK/PTK2-expressing and high FAK/PTK2-expressing groups, which were obtained from the Kyoto Encyclopedia of Genes and Genomes. The relevant signaling pathways were presented in plots if they fulfilled certain criterion (P < .05) and the pathways with the top 5 normalized enrichment scores were considered.
2.5. Analysis of immunotherapeutic response
As mentioned above, 3 relevant independent immunotherapeutic cohorts were included and analyzed. In general, immunotherapeutic approaches yielded 4 outcomes: complete response, partial response, progressive disease, and stable disease. Patients who achieved complete response or partial response were categorized as responders and compared to nonresponders, who showed signs of stable disease or progressive disease. Then the differences in FAK/PTK2 expression between the responders and nonresponders were identified by the Wilcoxon test.
3. Results
3.1. Clinical landscape of FAK/PTK2 expression in 33 cancers
The analysis details are summarized and presented for a more comprehensive outlook Figure 1. The abbreviations and full names of the 33 cancers considered here are available in Table 1. FAK/PTK2 is differentially expressed in 15 of the 33 cancers (BLCA, BRCA, CHOL, COAD, ESCA, GBM, HNSC, KICH, KIRP, LIHC, LUAD, LUSC, RWAD, STAD, THCA) (Fig. 2A). FAK/PTK2 is highly differentially expressed among elder patients with LAML, STAD or THYM, whereas it is weakly expressed in COAD, ESCA KIRC, KIPR, LGG, MESO, PCPG, PRAD, and READ (Fig. 2B). Meanwhile, FAK/PTK2 expression is significantly correlated with tumor stage of some cancers, including BLCA, COAD, KIRC, KIRP, and TGCT (Fig. 2C). Besides, the results indicate significant gender-based differences in FAK/PTK2 expression of the DLBC, KIRC, KIRP, LIHC, and SKCM cases (Fig. 2D).
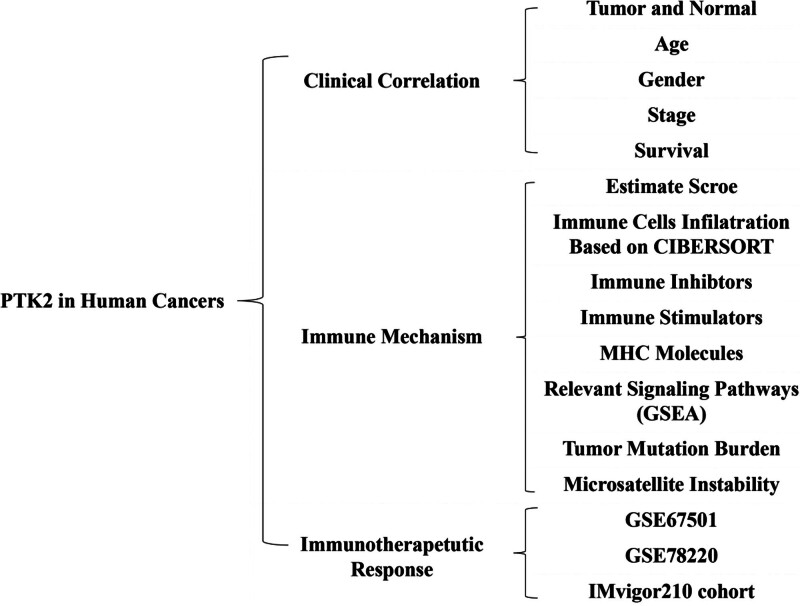
Table 1: Thirty-three types of human cancers employed in our research (abbreviation full name).
| ACC | Adrenocortical carcinoma |
| BLCA | Bladder urothelial carcinoma |
| BRCA | Breast invasive carcinoma |
| CESC | Cervical squamous cell carcinoma and endocervical adenocarcinoma |
| CHOL | Cholangiocarcinoma |
| COAD | Colon adenocarcinoma |
| DLBC | Lymphoid neoplasm diffuse large B-cell lymphoma |
| ESCA | Esophageal carcinoma |
| GBM | Glioblastoma multiforme |
| HNSC | Head and neck squamous cell carcinoma |
| KICH | Kidney chromophobe |
| KIRC | Kidney renal clear cell carcinoma |
| KIRP | Kidney renal papillary cell carcinoma |
| LAML | Acute myeloid leukemia |
| LGG | Brain lower-grade glioma |
| LIHC | Liver hepatocellular carcinoma |
| LUAD | Lung adenocarcinoma |
| LUSC | Lung squamous cell carcinoma |
| MESO | Mesothelioma |
| OV | Ovarian serous cystadenocarcinoma |
| PAAD | Pancreatic adenocarcinoma |
| PCPG | Pheochromocytoma and paraganglioma |
| PRAD | Prostate adenocarcinoma |
| READ | Rectum adenocarcinoma |
| SARC | Sarcoma |
| SKCM | Skin cutaneous melanoma |
| STAD | Stomach adenocarcinoma |
| TGCT | Testicular germ cell tumors |
| THCA | Thyroid carcinoma |
| THYM | Thymoma |
| UCEC | Uterine corpus endometrial carcinoma |
| UCS | Uterine carcinosarcoma |
| UVM | Uveal melanoma |
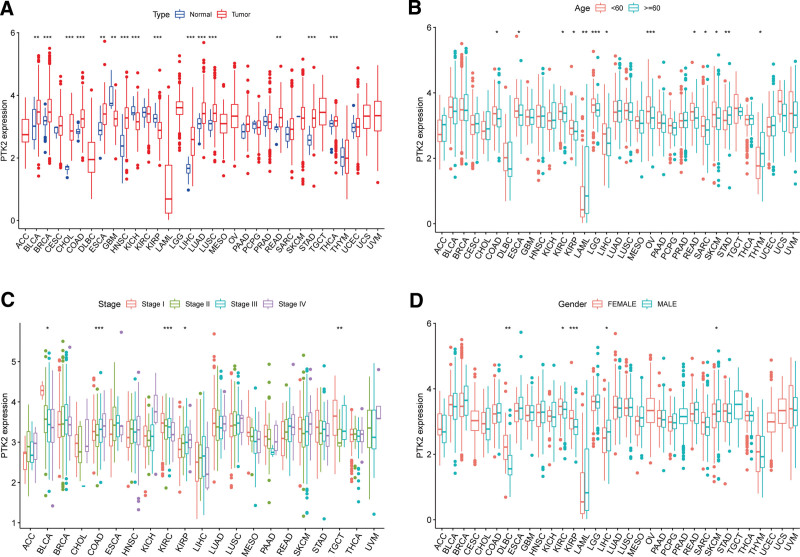
FAK/PTK2 activity is significantly increased in tumor groups of CHOL, COAD, HNSC, LIHC, LUAD, LUSC, and STAD, and is decreased in tumor groups of KIRC, KIRP, THCA, and UCEC (Fig. 3A). FAK/PTK2 expression varies across tumors from high to low (Fig. 3B). The gene activity of FAK/PTK2 differs from high to low among different tumors (Fig. 3C).
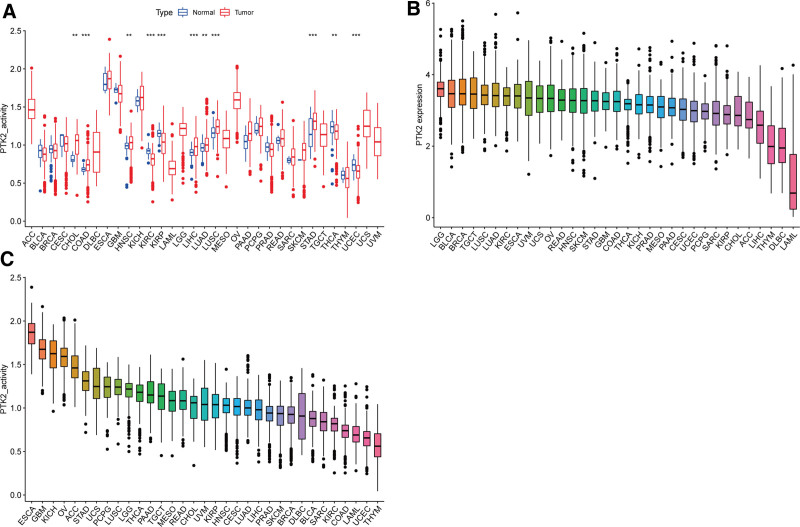
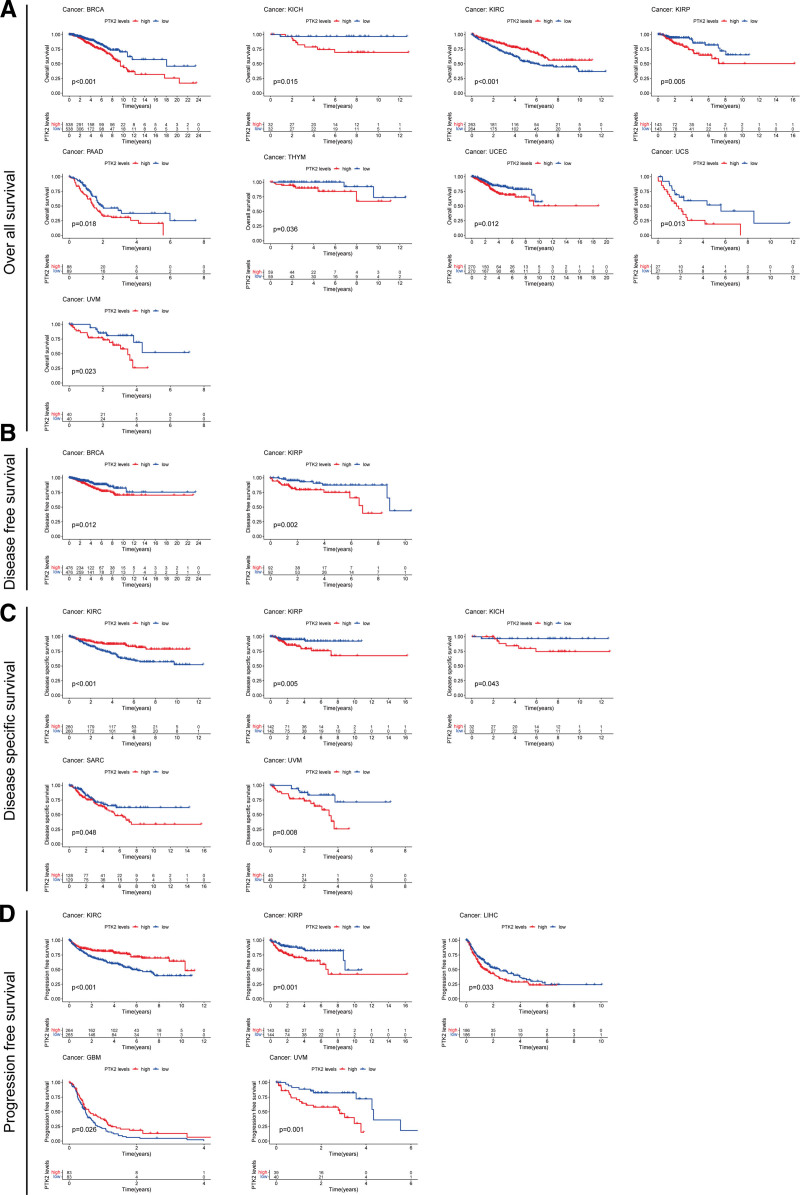
According to the forest plots (Fig. 4A–D), the association between FAK/PTK2 expression and OS is apparently positive in BRCA, KICH, KIRP, LAML, LIHL, SAPC, UCEC, and UVM, but is negative in KIRC (Fig. 4A).
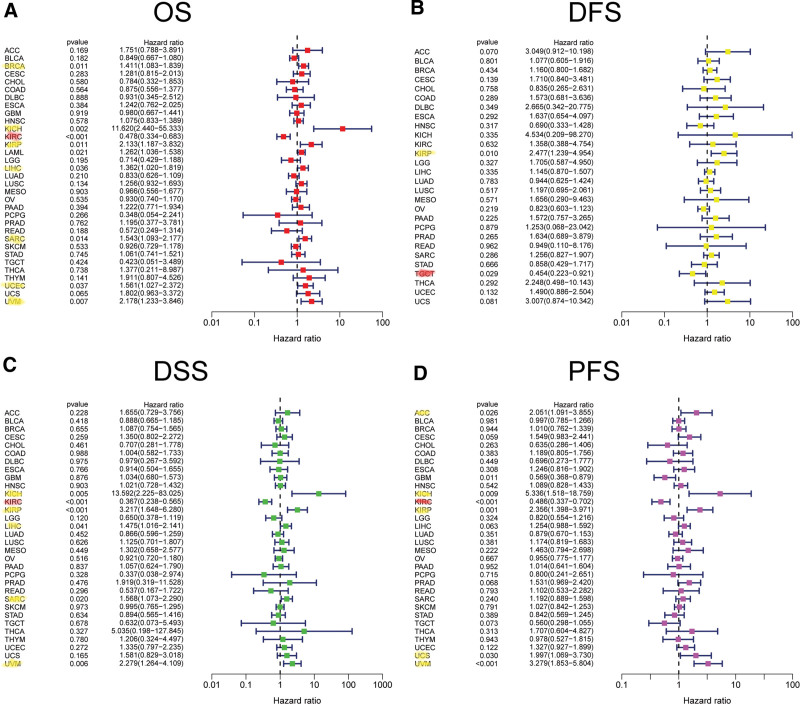
As for FAK/PTK2 and DFS, a significant negative association exists in TGCT, but a positive association is present in KIRP (Fig. 4B). In terms of DSS, FAK/PTK2 expression has a protective effect on KIRC, whereas it seems to be a risk factor in KICH, KIRP, LIHC, SARC, and UVM (Fig. 4C). Moreover, the PFS forest plot confirms the protective role of FAK/PTK2 expression in GBM and KIRC, and its role as a risk factor in ACC, KICH, KIRP, UCS, and UVM (Fig. 4D). The effect of FAK/PTK2 on prognosis in different tumors is shown in Figure 5.
3.2. Underlying association between FAK/PTK2 expression and immune-related factors
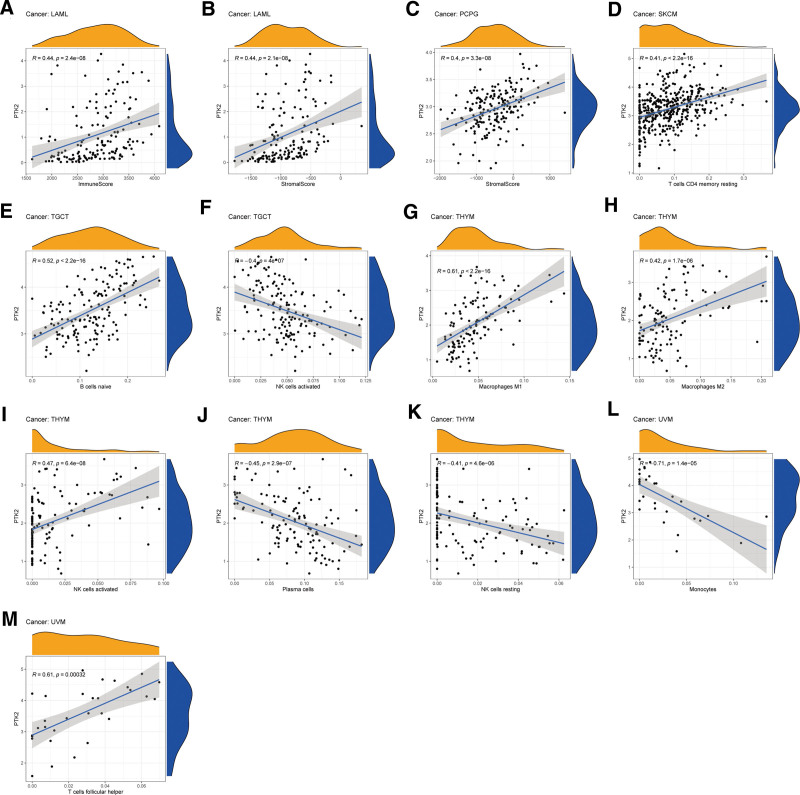
The stromal score, immune score, and immune cell infiltration are summarized in Figure 6 (P < .01 and |R| > 0.4). Notably, FAK/PTK2 expression is positively associated with the stromal scores of both LAML and PCPG, and with the immune score of LAML (all R < 0.5). In terms of immune cell infiltration, the FAK/PTK2 expression is positively associated with the M0/M1/M2 macrophage count in THYM, and with the count of NK cells activated, but R is over 0.5 only in M1 (R = 0.61, P < 2.2e−16). Besides, the FAK/PTK2 expression is negatively associated with the counts of plasma cells and NK cells resting. In UVM, FAK/PTK2 expression is associated negatively with monocyte infiltration (R = −0.71, P = 1.4e−0.5) and positively with T cell follicular helper infiltration (R = 0.61, P < .005) and the count of B cells naive (R = 0.52, P < 2.2e−16).
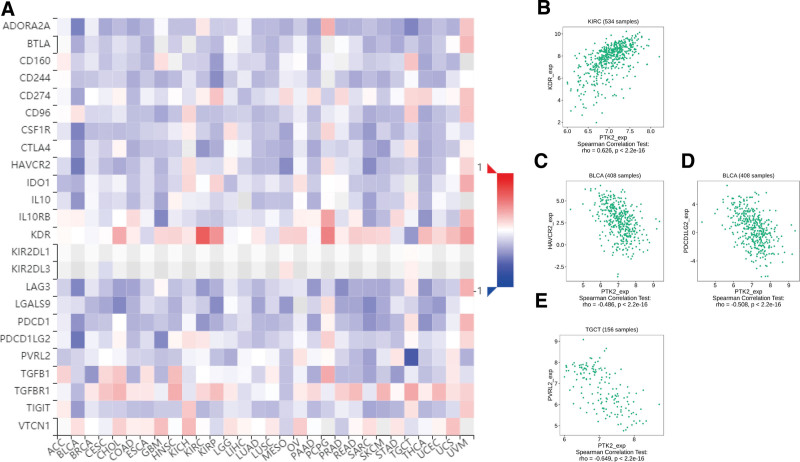
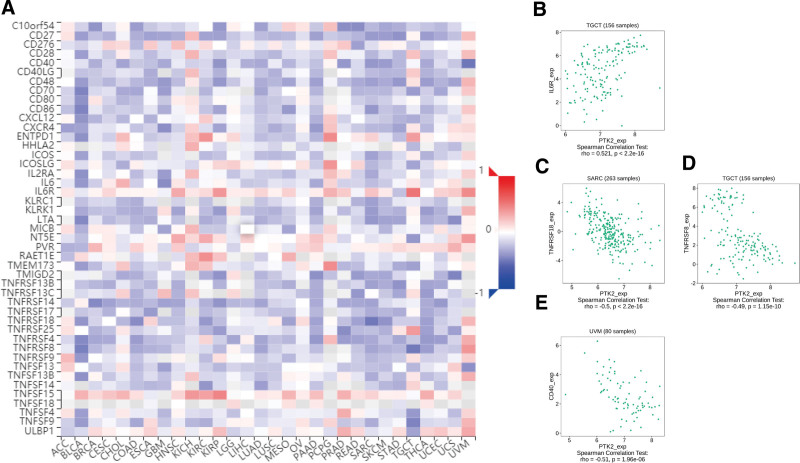
Totally 24 types of immune inhibitors were analyzed. FAK/PTK2 expression is associated positively with KDR in KIRC, and negatively with PVRL2 in TGCT, HAVCR2, and PDCD1LG2 in BLCA (Fig. 7). The correlation analyses of 45 immune stimulators demonstrate that FAK/PTK2 expression is associated positively with IL-6R in TGCT and negatively with TNFRSF18 in SARC, TNFRSF8 in TGTC, and CD40 in UVM (Fig. 8). Moreover, the expression correlation between FAK/PTK2 and MHC molecules demonstrate that FAK/PTK2 expression is negatively associated with HLA-A in CHOL, HLA-B in CHOL, HLA-A in READ, and TAPBP in KIRP (Fig. 9).
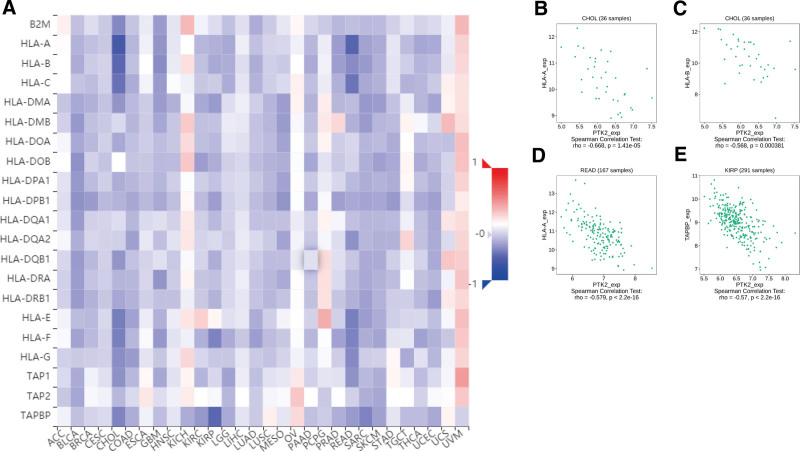
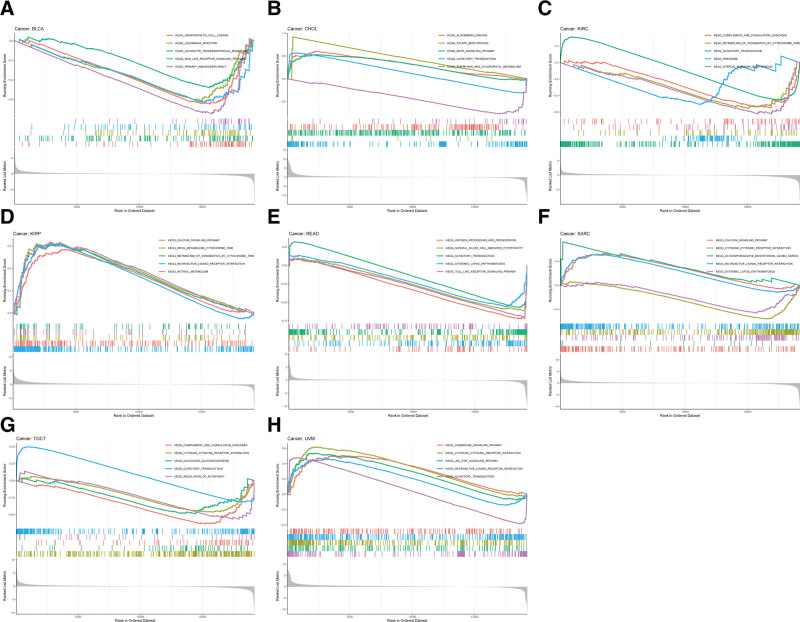
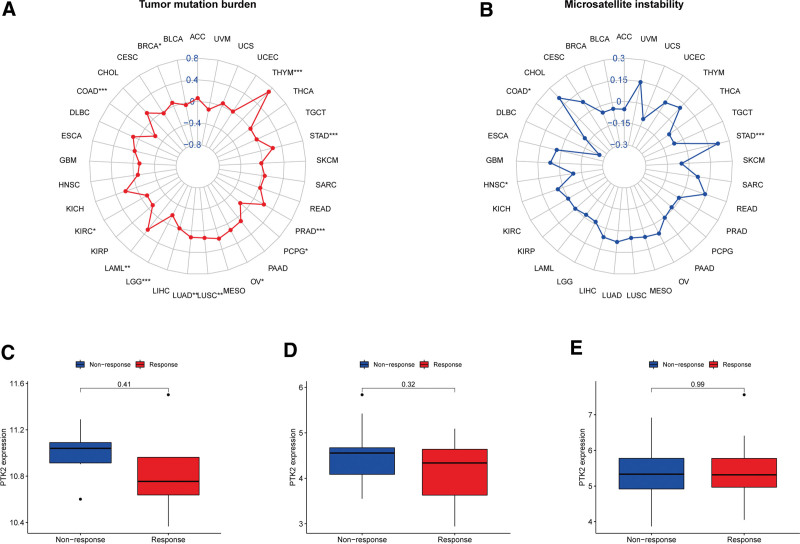
Given the robust correlations of FAK/PTK2 with TGCT, KIRC, CHOL, KIRP, READ, UVM, BLCA, and SARC, we performed GSEA to investigate the potential pathways involved in FAK/PTK2 signaling in these cancers. Genes from immune-relevant pathways (e.g., the drug metabolism cytochrome P450 signaling pathway) tend to be enriched in the high expressing groups of PRAD, but the Cytokine receptor interaction signaling pathway is enriched in the high expressing groups of UVM (Fig. 10). The correlation between FAK/PTK2 and the novel dynamic biomarkers of the immune checkpoint blockade (TMB and MSI) were further explored. FAK/PTK2 expression is related to TMB positively in THYM, LAML, LUAD, LUSC, OV, PRAD, STAD, and BRCA, but negatively in COAD, PCPG, LGG, and KIRC (Fig. 11A). MSI is associated with FAK/PTK2 expression positively in STAD and negatively in COAD and HNSC (Fig. 11B).
3.3. Immunotherapeutic response of FAK/PTK2
No significant difference in FAK/PTK2 expression is evident between the responders and nonresponders in any of the 3 independent cohorts (Fig. 11C–E). A trend observed in the studied cohorts is that patients with low FAK/PTK2 expression are seemingly more responsive to immunotherapy.
4. Discussion
This study extracted potential values of FAK/PTK2 in various cancers, especially in the context of immunotherapy. First, correlation analysis between FAK/PTK2 and clinical parameters shows no significant differences in age, gender, or tumor stage among most cancer types. FAK/PTK2 is overexpressed in BLCA, BRCA, CHOL, COAD, ESCA, GBM, HNSC, KICH, KIRP, LIHC, LUAD, LUSC, RWAD, STAD, and THCA, which is consistent with previous studies that FAK/PTK2 expressions in HNSC,[33] BRCA, and LIHC[34] are significantly higher than in the surrounding normal tissues. In HNSC, strong FAK/PTK2 expression is robustly associated with an increased HNSC risk and more importantly, FAK/PTK2 expression is an independent predictor in multivariate analysis.[35] Moreover, FAK/PTK2 overexpression is associated with local infiltration and distant metastasis.
To further clarify the action mechanism of FAK2 overexpression in tumor growth, in vitro experiments from Ilic et al[36] reveal that endothelial cells lacking FAK/PTK2 expression or function have a severely reduced ability to form tubules in the stromal gel. The global FAK/PTK2 knock in point mutation in the catalytic domain by Lim et al[37] further confirms the role of FAK/PTK2 in the development process, and reveals that the activity of FAK/PTK2 is very important for vascular morphogenesis cell movement and polarity. Furthermore, PTK2 serves as a target for 8q23-q24 amplification events in LIHC, and elevated PTK2 expression is correlated with large tumor volume.[34] FAK/PTK2 can also be translocated to the nucleus of cancer cells, where it regulates the expression program of chemokines and cytokines in inflammatory genes,[38] so as to promote immune escape and immune therapy resistance.[39,40] Besides, the expression and tyrosine phosphorylation of FAK are highly correlated with cell cycle progression by modulating cell cycle-relative molecules, which highlights that FAK functions as a key regulator in promoting cancer proliferation.[41] Tai et al[42] confirm that FAK activation, as determined by phosphospecific antibody recognition of the FAK tyrosine autophosphorylation site, is increased with tumor progression. Hence, the above evidences confirm the usefulness of FAK/PTK2 in cancer prognosis. In a word, these data suggest that FAK/PTK2 is a regulator of tissue growth response and malignant transformation.
To clarify the relationship between FAK/PTK2 overexpression and prognosis, we demonstrate that high FAK/PTK2 expression is associated with worse OS in BRCA, KICH, KIRP, PAAD, THYM, UCEC, UCS, and UVM, worse DFS in BRCA, KIRP, and KICH, worse DSS in KIRP, SARC, and UVM, and worse PFS in KIRP, LIHC, and UVM. These results are consistent with multiple previous studies. For instance, Golubovskaya et al[43] confirm that high FAK expression always predicts poor prognosis in BRCA. Miyazaki et al[13] suggest that FAK is highly expressed in esophageal cancer and is closely associated with poor prognosis. We hypothesize that therapeutic modulation of FAK/PTK2 activity or phosphorylation level in various tumors may be an effective strategy with clinical benefits.
We further investigated the correlation between FAK/PTK2 and immune cell infiltration and observed a robust positive correlation between FAK/PTK2 and M0/M1/M2 macrophages in THYM. Reportedly, FAK/PTK2 affects tumor development as well as immune responses within the tumor environment via tumor-associated macrophages.[44] FAK is important to induce TAMs by recruiting macrophages into tumor tissues, and may perform pretumor function by regulating the expression of downstream genes.[42] T cell infiltration is a reliable predictor of prognosis and has been implemented in the treatment of various cancers.[45] Reportedly, T cells play a positive role in tumor progression and their exclusion from TME leads to immune privilege.[46] In UVM, FAK/PTK2 expression is associated negatively with monocyte infiltration (R = −0.71, P = 1.4e−0.5) and positively with T cell follicular helper infiltration (R = 0.61, P < .005) and B-cell naive count (R = 0.52, P < 2.2e−16). Similarly, previous research shows that the prognosis of UVM is inversely associated with immune cell infiltration.[47]
In addition, 2 immunotherapeutic biomarkers (TMB and MSI) are significantly correlated with FAK/PTK2 in some cancers. Generally, more new antigens may be formed in a tumor with more somatic mutations, and TMB provides a useful tumor neoantigen load.[48] In contrast, MSI is defined as a robust mutant phenotype caused by DNA mismatch repair defects and is a potential predictive marker of immunotherapy.[49] FAK/PTK2 is correlated negatively with TMB and MSI in COAD, but positively with these 2 biomarkers in STAD. It is suggested that FAK/PTK2 may indirectly affect the immunotherapeutic response of STAD and COAD. Subsequently, we studied the correlation between FAK/PTK2 and immunotherapeutic response, but found no significant differences between the 2 study cohorts. We hypothesize that although these 3 cohorts have received anti-death protein 1 treatment and responded, FAK/PTK2 may affect the immunotherapeutic response by targeting other immune checkpoints, such as cytotoxic T lymphocyte antigen 4 or TIGIT. Moreover, we only analyzed 3 related cohorts and can hardly clarify the actual immunotherapeutic response of FAK/PTK2. Hence, more relevant immunotherapy cohorts shall be studied in the future.
To our knowledge, this is the first study to focus on the value of FAK/PTK2 in various cancers (33 types). This study provides valuable insights into the role of FAK/PTK2 in cancer immunotherapy, reveals its relationship with important immune indicators (immune cell infiltration, immune modulators, and immune biomarkers), and may help to understand the potential mechanism between FAK/PTK2 and the immune system. Although not all cancers show an association between tumor immune microenvironment and FAK/PTK2, these findings highlight the immune role of FAK/PTK2 in specific cancers, which will be used as an effective means to target them. However, given the bioinformatics method used here, these findings are preliminary. Therefore, more research on this topic is needed before the association between FAK/PTK2 and cancer immunotherapy is clearly understood and widely accepted.
There are still some limitations in the current study. First of all, this study showed that patients with low FAK/PTK2 expression are more responsive to immunotherapy, but the target molecules of FAK/PTK2 to regulate the immunotherapeutic response of cancer is unknown. Second, We did not conduct clinical trials to further validate our findings, we should go back to the clinical samples to confirm the key conclusions so the results still need further verification.
5. Conclusions
To our knowledge, this is the first study to present a high frequency of FAK/PTK2 expression and the predictive significance of ICI treatment in 33 human cancers. We believe that these findings may lay a groundwork for prospective functional experiments and may eventually have an impact in the clinical setting.
Author contributions
Conceptualization: Yujing Shi, Yumeng Zhang, Liang Liang.
Data curation: Yujing Shi, Yumeng Zhang, Xiaoke Di.
Formal analysis: Yujing Shi, Xiaoke Di.
Investigation: Yujing Shi, Xiaoke Di.
Methodology: Yujing Shi, Mengyang Ju, Yumeng Zhang.
Writing—original draft: Yujing Shi, Xiaoke Di.
Software: Mengyang Ju, Xinchen Sun.
Supervision: Liang Liang, Xinchen Sun.
Validation: Liang Liang, Xinchen Sun, Xiaoke Di.
Writing—review & editing: Xinchen Sun, Xiaoke Di.
Funding acquisition: Xiaoke Di.
Visualization: Xiaoke Di.
References
- AG Sacher, L Gandhi. Biomarkers for the clinical use of PD-1/PD-L1 inhibitors in non-small-cell lung cancer: a review.. JAMA Oncol., 2016. [PubMed]
- TE Keenan, KP Burke, EM Van Allen. Genomic correlates of response to immune checkpoint blockade.. Nat Med., 2019. [PubMed]
- D Miao, CA Margolis, NI Vokes. Genomic correlates of response to immune checkpoint blockade in microsatellite-stable solid tumors.. Nat Genet., 2018. [PubMed]
- RO Hynes. Integrins: bidirectional, allosteric signaling machines.. Cell., 2002. [PubMed]
- JL Guan. Integrin signaling through FAK in the regulation of mammary stem cells and breast cancer.. IUBMB Life., 2010. [PubMed]
- JT Parsons. Focal adhesion kinase: the first ten years.. J Cell Sci., 2003. [PubMed]
- KK Ward, I Tancioni, C Lawson. Inhibition of focal adhesion kinase (FAK) activity prevents anchorage-independent ovarian carcinoma cell growth and tumor progression.. Clin Exp Metastasis., 2013. [PubMed]
- SK Hanks, MB Calalb, MC Harper. Focal adhesion protein-tyrosine kinase phosphorylated in response to cell attachment to fibronectin.. Proc Natl Acad Sci U S A., 1992. [PubMed]
- MD Schaller, CA Borgman, BS Cobb. pp125FAK a structurally distinctive protein-tyrosine kinase associated with focal adhesions.. Proc Natl Acad Sci U S A., 1992. [PubMed]
- TM Weiner, ET Liu, RJ Craven. Expression of focal adhesion kinase gene and invasive cancer.. Lancet., 1993. [PubMed]
- AL Lark, CA Livasy, B Calvo. Overexpression of focal adhesion kinase in primary colorectal carcinomas and colorectal liver metastases: immunohistochemistry and real-time PCR analyses.. Clin Cancer Res., 2003. [PubMed]
- PL Judson, X He, WG Cance. Overexpression of focal adhesion kinase, a protein tyrosine kinase, in ovarian carcinoma.. Cancer., 1999. [PubMed]
- T Miyazaki, H Kato, M Nakajima. FAK overexpression is correlated with tumour invasiveness and lymph node metastasis in oesophageal squamous cell carcinoma.. Br J Cancer., 2003. [PubMed]
- S Itoh, T Maeda, M Shimada. Role of expression of focal adhesion kinase in progression of hepatocellular carcinoma.. Clin Cancer Res., 2004. [PubMed]
- AK Sood, GN Armaiz-Pena, J Halder. Adrenergic modulation of focal adhesion kinase protects human ovarian cancer cells from anoikis.. J Clin Invest., 2010. [PubMed]
- VM Golubovskaya. Targeting FAK in human cancer: from finding to first clinical trials.. Front Biosci (Landmark Ed)., 2014. [PubMed]
- FJ Sulzmaier, C Jean, DD Schlaepfer. FAK in cancer: mechanistic findings and clinical applications.. Nat Rev Cancer., 2014. [PubMed]
- J Popow, H Arnhof, G Bader. Highly selective PTK2 proteolysis targeting chimeras to probe focal adhesion kinase scaffolding functions.. J Med Chem., 2019. [PubMed]
- D Hanahan, LM Coussens. Accessories to the crime: functions of cells recruited to the tumor microenvironment.. Cancer Cell., 2012. [PubMed]
- X Zheng, Y Hu, C Yao. The paradoxical role of tumor-infiltrating immune cells in lung cancer.. Intractable Rare Dis Res., 2017. [PubMed]
- A Serrels, T Lund, B Serrels. Nuclear FAK controls chemokine transcription, Tregs, and evasion of antitumor immunity.. Cell., 2015. [PubMed]
- A Kasorn, P Alcaide, Y Jia. Focal adhesion kinase regulates pathogen-killing capability and life span of neutrophils via mediating both adhesion-dependent and-independent cellular signals.. J Immunol., 2009. [PubMed]
- SK Mitra, ST Lim, A Chi. Intrinsic focal adhesion kinase activity controls orthotopic breast carcinoma metastasis via the regulation of urokinase plasminogen activator expression in a syngeneic tumor model.. Oncogene., 2006. [PubMed]
- JB Stokes, SJ Adair, JK Slack-Davis. Inhibition of focal adhesion kinase by PF-562,271 inhibits the growth and metastasis of pancreatic cancer concomitant with altering the tumor microenvironment.. Mol Cancer Ther., 2011. [PubMed]
- S Batista, E Maniati, LE Reynolds. Haematopoietic focal adhesion kinase deficiency alters haematopoietic homeostasis to drive tumour metastasis.. Nat Commun., 2014. [PubMed]
- MY Abshire, KS Thomas, KA Owen. Macrophage motility requires distinct alpha5beta1/FAK and alpha4beta1/paxillin signaling events.. J Leukoc Biol., 2011. [PubMed]
- K Tomczak, P Czerwińska, M Wiznerowicz. The Cancer Genome Atlas (TCGA): an immeasurable source of knowledge.. Contemp Oncol (Pozn)., 2015. [PubMed]
- S Mariathasan, SJ Turley, D Nickles. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells.. Nature., 2018. [PubMed]
- DR Neavin, JH Lee, D Liu. Single nucleotide polymorphisms at a distance from aryl hydrocarbon receptor (AHR) binding sites influence AHR ligand-dependent gene expression.. Drug Metab Dispos., 2019. [PubMed]
- K Yoshihara, M Shahmoradgoli, E Martínez. Inferring tumour purity and stromal and immune cell admixture from expression data.. Nat Commun., 2013. [PubMed]
- AM Newman, CL Liu, MR Green. Robust enumeration of cell subsets from tissue expression profiles.. Nat Methods., 2015. [PubMed]
- R Bonneville, MA Krook, EA Kautto. Landscape of microsatellite instability across 39 cancer types.. JCO Precis Oncol., 2017. [PubMed]
- M Canel, P Secades, JP Rodrigo. Overexpression of focal adhesion kinase in head and neck squamous cell carcinoma is independent of fak gene copy number.. Clin Cancer Res., 2006. [PubMed]
- H Okamoto, K Yasui, C Zhao. PTK2 and EIF3S3 genes may be amplification targets at 8q23-q24 and are associated with large hepatocellular carcinomas.. Hepatology., 2003. [PubMed]
- JP Rodrigo, G Álvarez-Alija, ST Menéndez. Cortactin and focal adhesion kinase as predictors of cancer risk in patients with laryngeal premalignancy.. Cancer Prev Res (Phila)., 2011. [PubMed]
- D Ilic, B Kovacic, S McDonagh. Focal adhesion kinase is required for blood vessel morphogenesis.. Circ Res., 2003. [PubMed]
- ST Lim, XL Chen, A Tomar. Knock-in mutation reveals an essential role for focal adhesion kinase activity in blood vessel morphogenesis and cell motilitypolarity but not cell proliferation.. J Biol Chem., 2010. [PubMed]
- ST Lim, NL Miller, XL Chen. Nuclear-localized focal adhesion kinase regulates inflammatory VCAM-1 expression.. J Cell Biol., 2012. [PubMed]
- H Jiang, S Hegde, BL Knolhoff. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy.. Nat Med., 2016. [PubMed]
- B Serrels, N McGivern, M Canel. IL-33 and ST2 mediate FAK-dependent antitumor immune evasion through transcriptional networks.. Sci Signaling., 2017
- KB Dunn, M Heffler, VM Golubovskaya. Evolving therapies and FAK inhibitors for the treatment of cancer.. Anticancer Agents Med Chem., 2010. [PubMed]
- YL Tai, LC Chen, TL Shen. Emerging roles of focal adhesion kinase in cancer.. Biomed Res Int., 2015
- VM Golubovskaya, M Zheng, L Zhang. The direct effect of focal adhesion kinase (FAK), dominant-negative FAK, FAKCD and FAK siRNA on gene expression and human MCF-7 breast cancer cell tumorigenesis.. BMC Cancer., 2009. [PubMed]
- R Noy, JW Pollard. Tumor-associated macrophages: from mechanisms to therapy.. Immunity., 2014. [PubMed]
- WH Fridman, F Pagès, C Sautès-Fridman. The immune contexture in human tumours: impact on clinical outcome.. Nat Rev Cancer., 2012. [PubMed]
- I Atreya, MF Neurath. Immune cells in colorectal cancer: prognostic relevance and therapeutic strategies.. Expert Rev Anticancer Ther., 2008. [PubMed]
- S Zuo, M Wei, S Wang. Pan-cancer analysis of immune cell infiltration identifies a prognostic immune-cell characteristic score (ICCS) in lung adenocarcinoma.. Front Immunol., 2020. [PubMed]
- TA Chan, M Yarchoan, E Jaffee. Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic.. Ann Oncol., 2019. [PubMed]
- H Yamamoto, K Imai. An updated review of microsatellite instability in the era of next-generation sequencing and precision medicine.. Semin Oncol., 2019. [PubMed]
