Omega-3 Recovers Cannabinoid 1 Receptor Expression in the Adult Mouse Brain after Adolescent Binge Drinking
Abstract
Adolescent binge drinking is a social problem with a long-lasting impact on cognitive functions. The cannabinoid type-1 (CB1) receptor of the endocannabinoid system (ECS) is involved in brain synaptic plasticity, cognition and behavior via receptor localization at specific subcellular compartments of the cortical, limbic and motor regions. Alcohol (EtOH) intake affects the ECS, CB1 and their functions. Evidence indicates that binge drinking during adolescence impairs memory via the abrogation of CB1-dependent synaptic plasticity in the hippocampus. However, the impact of EtOH consumption on global CB1 receptor expression in the adult brain is unknown. We studied this using optical density analysis throughout brain regions processed for light microscopy (LM) immunohistotochemistry. CB1 staining decreased significantly in the secondary motor cortex, cerebellum, cingulate cortex, amygdala and nucleus accumbens. Next, as omega-3 (n-3) polyunsaturated fatty acids (PUFAs) rescue synaptic plasticity and improve EtOH-impaired cognition, we investigated whether docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) had any effect on CB1 receptors. N-3 intake during EtOH abstinence restored CB1 immunostaining in the secondary motor cortex, cerebellum and amygdala, and ameliorated receptor density in the cingulate cortex. These results show that n-3 supplementation recovers CB1 receptor expression disrupted by EtOH in distinct brain regions involved in motor functions and cognition.
Article type: Research Article
Keywords: ethanol, polyunsaturated fatty acids, endocannabinoid system, immunohistochemistry, motor system, cognition, rodent
Affiliations: Laboratory of Ultrastructural and Functional Neuroanatomy of the Synapse, Department of Neurosciences, Faculty of Medicine and Nursing, University of the Basque Country UPV/EHU, 48940 Leioa, Spain; anemartinllorente@gmail.com (A.M.-L.); maitane.serrano@ehu.eus (M.S.); itziar.bonilla@ehu.eus (I.B.-D.R.); leire.lecumberri@ehu.eus (L.L.); garaziocerin97@gmail.com (G.O.); nagore.puente@ehu.eus (N.P.); almudena.ramos@ehu.eus (A.R.); irantzu.rico@ehu.eus (I.R.-B.); Achucarro Basque Center for Neuroscience, Science Park of the UPV/EHU, 48940 Leioa, Spain
License: © 2023 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/ijms242417316 | PubMed: 38139145 | PMC: PMC10744058
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.7 MB)
1. Introduction
The ECS system is mainly composed of endocannabinoids (eCBs), the enzymes involved in their synthesis and degradation, as well as cannabinoid receptors [ref. 1]. The cannabinoid CB1, the most abundant G-protein-coupled receptor in the brain, is highly expressed in the basal ganglia, cerebral cortex, cerebellum (Cb) and hippocampus, which is consistent with its role in learning and memory, motor control, motivation, reward and emotional processing, among many others [ref. 1,ref. 2]. Furthermore, CB1 increases and changes its distribution from adolescence to adulthood, is predominantly localized in presynaptic terminals and noticeably present in glial cells and mitochondria, modulates neurotransmitter release and also participates in synaptic plasticity [ref. 1,ref. 2,ref. 3,ref. 4,ref. 5].
Binge drinking during adolescence has a long-lasting impact on cognition and behavior via interference with brain neurochemical systems, including the ECS which participates in the regulation of motivation and EtOH intake [ref. 6,ref. 7,ref. 8,ref. 9]. EtOH exposure alters both CB1 mRNA and protein [ref. 10,ref. 11], changing the availability and binding of the receptor [ref. 12,ref. 13]. Conversely, gene deletion and CB1 receptor antagonism affect drinking behavior [ref. 14]. In addition, a CB1 mRNA decrease in the adult hippocampus after adolescent binge drinking correlates with CB1 receptor reduction in excitatory terminals of the hippocampal dentate molecular layer. These changes, together with the rise in monoacylglycerol lipase (MAGL) mRNA, the main degrading enzyme of the eCB 2-arachidonoylglycerol (2-AG), disrupt endocannabinoid-dependent long-term depression (LTD) and set off memory impairment [ref. 15]. Remarkably, the 2-AG increase elicited by MAGL inhibition recovers synaptic plasticity and cognition [ref. 15]. Although pharmacological manipulations are potential therapeutic strategies for treating alcohol-induced long-term cognitive deficits, nutritional supplementation might also stand up as a suitable alternative.
DHA and EPA are n-3 PUFAs that accumulate in brain cell membranes and maintain their structure and fluidity, restore glutathione levels, are anti-inflammatory, reduce oxidative stress and apoptosis, and overcome synaptic plasticity deficits, improving the cognitive impairment caused by prenatal EtOH exposure [ref. 16,ref. 17,ref. 18,ref. 19]. DHA is unevenly distributed among brain regions and between neurons and glial cells, and participates in membrane-associated protein functions, cellular signaling, gene expression, lipogenesis, neurogenesis and synaptic pruning [ref. 16]. EtOH decreases n-3 via different mechanisms [ref. 20,ref. 21]. The negative impact of EtOH on DHA alters synaptic plasticity in the hippocampus and medial prefrontal cortex, both enriched in DHA under normal conditions [ref. 16]. Noticeably, the enhancement of eCB signaling recovers emotional and cognitive functions as well as reverses the abrogated eCB-dependent synaptic plasticity caused by n-3 PUFA deficiency in brain regions processing mood and cognition [ref. 22,ref. 23,ref. 24]. Moreover, DHA modulates CB1 mRNA and protein expression [ref. 25] as well as eCBs [ref. 26,ref. 27]. In fact, a new class of n-3-derived eCBs has been identified in addition to the most-known n-6-derived 2-AG and anandamide (AEA) [ref. 28].
However, despite the close relationship between EtOH, n-3 and CB1 receptors, the direct impact of n-3 PUFAs on brain CB1 receptor expression after EtOH intake remains unknown. In this investigation, we studied the effects of an n-3-enriched diet on CB1 receptor expression in the adult brain after binge drinking during adolescence. In particular, we analyzed CB1 receptor optical density in fourteen brain regions that are sensitive to EtOH damage and are known to express CB1 receptors.
2. Results
N-3 and Brain CB1 Receptor Expression
The brain and cerebellar CB1 immunostaining patterns in H2O, EtOH, n-3-EtOH and n-3-H2O mice corresponded well with previous observations of the CB1 receptor distribution in the mouse brain and cerebellum. CB1 immunoreactive punctate appeared concentratedin certain brain regions as well as in some cerebral and cerebellar cortical layers. Thus, strong CB1 receptor immunoreactivity was observed in the granule cell layer of the olfactory bulb (OB) [ref. 29] and remarkable staining was also revealed in the striatum, cerebral cortex (layers II–III and V–VI), olfactory tubercle, substantia nigra pars reticulata (SN), amygdala (Amg) and hippocampus [ref. 30], as well as the cerebellar molecular and Purkinje cell layers. More moderate immunoreactivity was detected in the nucleus accumbens (Acb) [ref. 31].
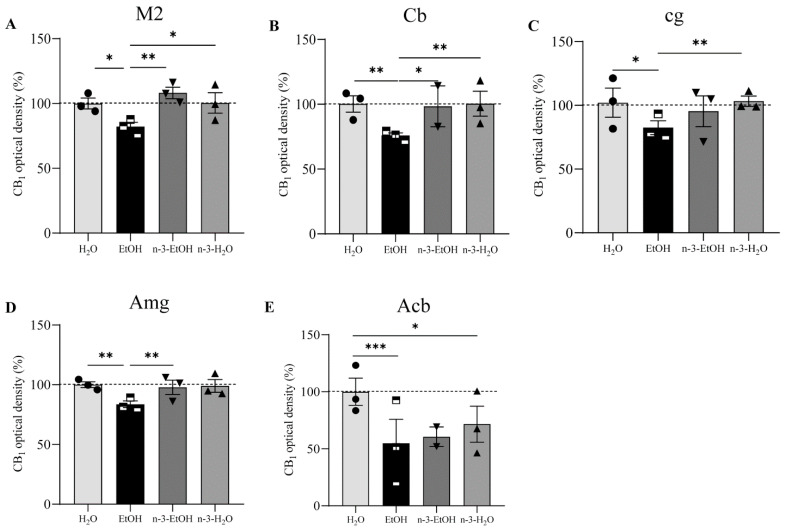
The analysis of the CB1 receptor optical density showed that the receptor distribution pattern varied in some regions depending on EtOH and/or n-3 intake. However, no changes were found in the OB, primary motor cortex (M1), frontal cortex (Fr3), cingular cortex area (Cg1), caudate putamen (CPu), dentate gyrus (DG), hippocampal CA1, SN and entorhinal cortex (Ent) in the four conditions (p > 0.05; Figure 1, Table 1). In contrast, the secondary motor cortex (M2), Cb, cingulum (cg), Amg and Acb were affected by EtOH and n-3 intake (p < 0.05; Figure 2). Thus, a significant reduction in CB1 receptor staining (~20%) was detected in the adult M2 after adolescent EtOH intake relative to the control (EtOH: 82.20 ± 3.732%; H2O: 100.00 ± 3.507% * p < 0.05; Figure 2 and Figure 3 A; Table 1). Remarkably, n-3 supplementation during withdrawal recovered CB1 receptor expression in the M2 to control levels (n-3-EtOH: 106.30 ± 4.604% ** p < 0.01), without the enriched diet having any effect on H2O mice (n-3-H2O: 100.50 ± 5.218% * p < 0.05; Figure 2 and Figure 3A; Table 1). The decrease in CB1 optical density following EtOH exposure during adolescence was more conspicuous in the Cb (EtOH: 76.40 ± 4.445% versus H2O: 100.00 ± 5.267%, ** p < 0.01), particularly in the molecular layer (Figure 2 and Figure 3B; Table 1). The n-3 diet normalized the detrimental effect of EtOH on CB1 values (n-3-EtOH: 98.43 ± 5.627%, * p < 0.05; Figure 2 and Figure 3B; Table 1). Again, n-3 under standard H2O conditions did not modify receptor expression in this region (n-3-H2O: 100.40 ± 4.620%, ** p < 0.01; Figure 2 and Figure 3B; Table 1). A similar effect of EtOH was observed in the cg, where CB1 optical density decreased significantly in adult mice after binge drinking during adolescence (EtOH: 80.77 ± 4.864% versus H2O: 100.00 ± 4.868% * p < 0.05; Figure 2 and Figure 3C; Table 1). However, n-3 was unable to revert significantly the receptor deficit (n-3-EtOH: 97.86 ± 5.274%), in contrast to the dietary effect on H2O (n-3-H2O: 105.20 ± 4.610%) relative to the CB1 decrease in EtOH mice (** p < 0.001; Figure 2 and Figure 3C; Table 1). The values in the Amg were also affected (Figure 2 and Figure 3D; Table 1): the significant CB1 reduction in EtOH mice (EtOH: 82.80 ± 3.468% versus H2O: 100.00 ± 3.594%, ** p < 0.01) returned to normal with n-3 intake (n-3-EtOH: 102.30 ± 3.977%, ** p < 0.01), with no effect of the nutritional supplementation when mice only drank H2O (97.69 ± 5.744%). Finally, CB1 optical density was drastically reduced in the Acb of the mature brain exposed to EtOH during adolescence (EtOH: 54.18 ± 10.81% versus H2O: 100.00 ± 6.028, *** p < 0.001; Figure 2 and Figure 3E; Table 1). However, n-3 intake could not restore CB1 receptor expression in EtOH mice (n-3-EtOH: 60.61 ± 8.153%). In addition, and contrary to the other studied brain regions, n-3 downregulated CB1 staining in H2O mice (n-3-H2O: 66.15 ± 11.04%, * p < 0.05; Figure 2 and Figure 3E; Table 1).
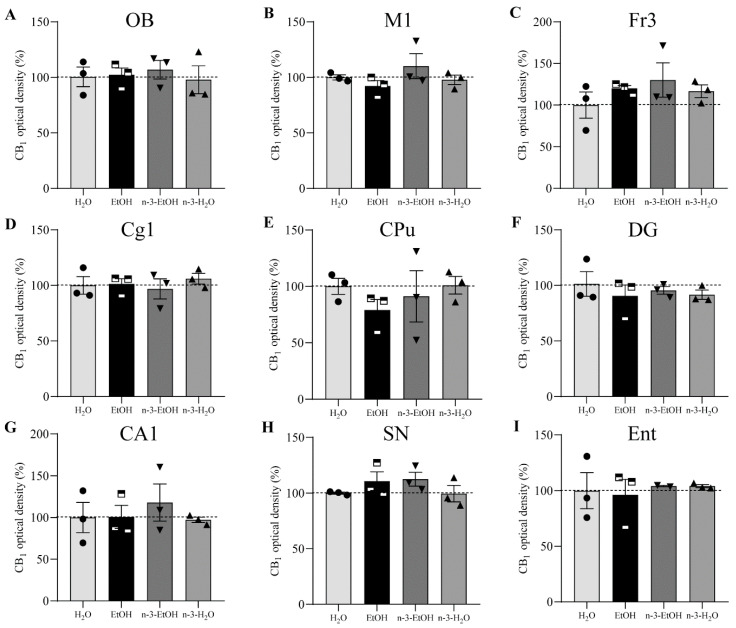
Table 1: Normalized values (% mean ± SEM) of CB1 receptor optical density in olfactory bulb (OB), primary and secondary motor cortex (M1, M2), frontal cortex (Fr3), cingular cortex area 1 (Cg1), cingulum (cg), caudate putamen (CPu), nucleus accumbens (Acb), amygdala (Amg), dentate gyrus (DG), hippocampal CA1, substantia nigra (SN), entorhinal cortex (Ent) and cerebellum (Cb) for each experimental condition (n = 3 mice/group).
| H2O | EtOH | n-3-EtOH | n-3-H2O | |
|---|---|---|---|---|
| OB | 100.00 ± 3.982 | 102.30 ± 5.493 | 108.50 ± 4.499 | 100.40 ± 6.144 |
| M1 | 100.00 ± 4.074 | 89.57 ± 4.427 | 106.80 ± 6.384 | 92.25 ± 5.785 |
| M2 | 100.00 ± 3.507 | 82.20 ± 3.732 | 106.30 ± 4.604 | 100.50 ± 5.218 |
| Fr3 | 100.00 ± 15.57 | 114.10 ± 14.25 | 130.20 ± 12.08 | 116.60 ± 13.00 |
| Cg1 | 100.00 ± 6.694 | 77.99 ± 9.688 | 77.06 ± 14.08 | 90.67 ± 10.22 |
| cg | 100.00 ± 4.868 | 80.77 ± 4.864 | 97.86 ± 5.274 | 105.20 ± 4.610 |
| CPu | 100.00 ± 11.31 | 79.01 ± 11.61 | 83.25 ± 12.03 | 90.02 ± 15.31 |
| Acb | 100.00 ± 6.028 | 54.18 ± 10.81 | 60.61 ± 8.153 | 66.15 ± 11.04 |
| Amg | 100.00 ± 3.594 | 82.80 ± 3.468 | 102.30 ± 3.977 | 97.69 ± 5.744 |
| DG | 100.00 ± 7.651 | 92.42 ± 4.135 | 96.72 ± 4.383 | 93.04 ± 3.291 |
| CA1 | 100.00 ± 10.62 | 100.80 ± 10.23 | 103.80 ± 5.839 | 98.02 ± 3.491 |
| SN | 100.00 ± 4.788 | 111.20 ± 3.457 | 109.10 ± 3.957 | 99.57 ± 3.918 |
| Ent | 100.00 ± 8.555 | 96.17 ± 4.879 | 104.20 ± 7.367 | 103.70 ± 3.471 |
| Cb | 100.00 ± 5.267 | 76.40 ± 4.445 | 98.43 ± 5.627 | 100.40 ± 4.620 |
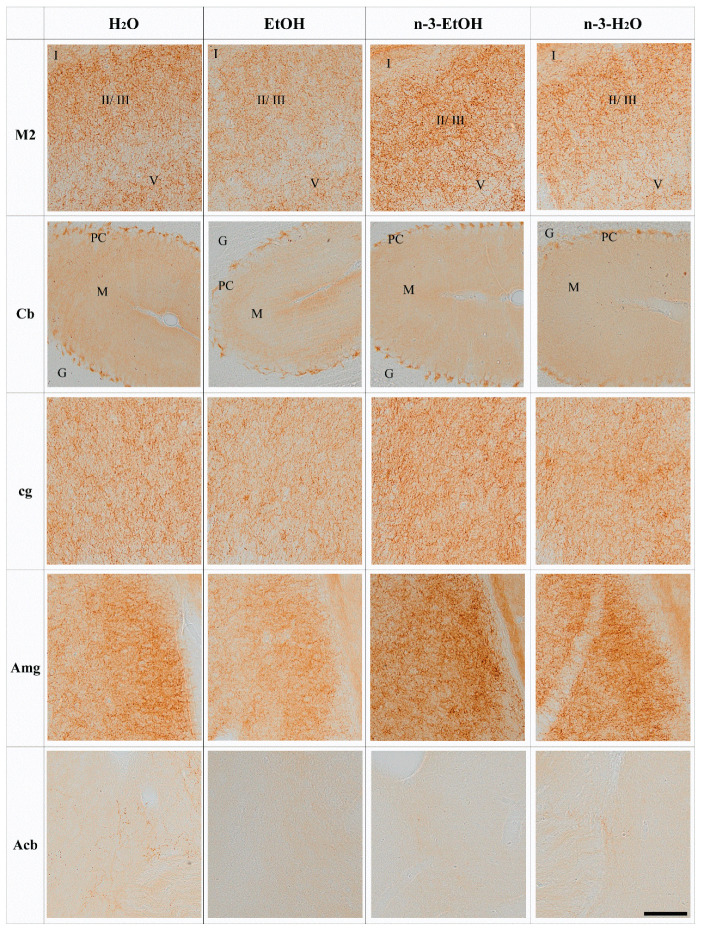
3. Discussion
3.1. Long-Lasting Effect of Adolescent Binge Drinking on CB1 Receptor Expression
We have shown that binge drinking during adolescence reduces CB1 receptor immunostaining in certain regions of the mature mouse brain, in particular, the M2, Cb, cg, Amg and Acb. Interestingly, n-3 supplementation during abstinence restores CB1 receptor expression measured using optical density in the M2, Cb and Amg and ameliorates density levels in the cg.
The CB1 receptor expression pattern matched the brain receptor distribution in the cortical, limbic and motor regions [ref. 2,ref. 30]. However, long-term changes in CB1 immunostaining after adolescent alcohol intake were restricted to some brain regions and the Cb that seem to correlate with the impact of EtOH intake on brain structure and function [ref. 6,ref. 32,ref. 33]. EtOH alters grey matter throughout the cortex, including the olfactory areas, Amg and Cb [ref. 33,ref. 34,ref. 35]. Also, the mesocorticolimbic system is affected. We observed that the long-lasting decrease in CB1 receptors normally expressed in the Acb [ref. 36] did not recover over time after adolescent binge drinking, remaining low even when the animals were under n-3 supplementation. As CB1 receptors intervene in brain maturation, it is plausible that the CB1 receptor expression deficits revealed in the Acb negatively contribute to the shape of the mesocorticolimbic system during the adolescent period, ultimately promoting brain vulnerability and alcohol addiction [ref. 35].
This study was conducted using male mice. Though males might be more vulnerable to withdrawal [ref. 37], females are more sensitive to EtOH [ref. 38,ref. 39]. Also, the effectiveness of treatments differs between males and females [ref. 40]. It is plausible that the EtOH impact on CB1 receptor expression and the effects of the n-3-enriched diet may vary between males and females, a possibility that will be explored in our future investigations.
The LM immunohistochemistry applied in the present study has some obvious limitations that deserve attention. Immunohistochemical techniques for LM were used in the 1990s to describe the pattern of CB1 receptor-like immunoreactivity in the brain [ref. 41,ref. 42]. Consequently, the strong immunostaining observed in certain brain regions (motor, limbic, reward, cortical) endorsed the advance in the knowledge of CB1 receptor functions in brain circuits. However, low CB1 receptor expression in cell types cannot be visualized using LM immunohistochemistry [ref. 30]. In addition, the tendency to diffusion of the 3-3′diaminobenzidine (DAB) reaction product used as chromogen in LM immunohistochemistry could lead to potential unspecific staining or false positives due to endogenous biotinylated proteins. These pitfalls can only be ruled out by using appropriate controls. In this study, we have used CB1-knockout brain tissue to discard bias, confirming the specificity of the CB1 staining observed throughout the brain in our experimental conditions. Ultimately, high-resolution immunoelectron microscopy that has been shown to be an excellent tool for unveiling the precise subcellular localization of CB1 receptors in the brain, would definitely identify the subcellular compartments and the CB1 receptor pools that were conspicuously reduced by adolescent EtOH intake, and recovered by n-3 in the specific brain regions identified in this study.
Endocannabinoid levels, membrane fluidity and EtOH-degrading enzymatic machinery could contribute to the altered CB1 receptor pattern observed in the adult brain after adolescent binge drinking. In addition, EtOH intake during adolescence causes memory impairment that can last into adulthood [ref. 43,ref. 44]. Our model of binge drinking during adolescence used in this study has previously been shown to be associated with hippocampal memory deficits in adulthood [ref. 15,ref. 45]. Although CB1 receptor optical density was not significantly affected by binge drinking, subtle subcellular changes in receptor expression were detected in the dentate molecular layer that should contribute to the abrogation of cannabinoid-dependent synaptic plasticity at the excitatory medial perforant path synapses and related memory loss [ref. 15]. Remarkably, the deleterious cognitive binge drinking effects were recovered by increasing the endocannabinoid 2-AG or by environmental conditions [ref. 15,ref. 45]. Furthermore, the M2 and Cb, both affected by EtOH, are brain regions involved in motor coordination [ref. 46,ref. 47] and EtOH intake leads to motor incoordination and ataxia [ref. 43]. Our present results show a significant decrease in CB1 staining in the cerebellar molecular layer, where the receptor is mostly localized to the excitatory granule cell parallel fiber terminals [ref. 48]. However, the lack of CB1 receptors does not seem to cause evident cerebellar motor deficits [ref. 49], despite their role in motor learning [ref. 50]. Nevertheless, cannabinoid-dependent motor control is also exerted from the cortex [ref. 51], where we detected deficits in CB1 receptors upon adolescent binge drinking. Interestingly, young adult mice under enriched environment recovered motor coordination and balance after adolescent binge drinking [ref. 45]. It is likely that the ECS participates in this motor improvement, as it is the case in the memory recovery elicited by environmental enrichment via the restoration of endocannabinoid-dependent excitatory synaptic plasticity, in which CB1 receptors, group I metabotropic glutamate receptors and 2-AG were involved [ref. 9].
EtOH modifies synaptic membrane fluidity [ref. 52] and stimulates arachidonic acid (AA) production from membrane phospholipids by increasing phospholipase A2 (PLA2) [ref. 53]. The availability of more AA for AEA synthesis may be responsible for the decrease in CB1 agonist binding and gene expression elicited by chronic EtOH in certain brain regions [ref. 54,ref. 55]). In fact, the drop in CB1 receptor immunostaining in the cerebellar molecular layer correlates with AEA transport inhibition and a 2-AG increase in granule cells after chronic EtOH [ref. 56,ref. 57]. Cannabinoids internalize CB1 receptors and reduce their mobility, having an impact on receptor availability at the synapse [ref. 58]. We have demonstrated previously that Δ-9-tetrahydrocannabinol (THC) causes a selective CB1 receptor labeling decrease in certain subcellular compartments (excitatory and inhibitory terminals, mitochondria, astrocytes) of several brain regions [ref. 59]. This distinct impact of THC could be related to the different THC levels and metabolites detected among brain regions after acute THC administration [ref. 60]. A similar phenomenon could befall our model of binge drinking. In fact, brain EtOH metabolism by class III alcohol dehydrogenase (ADH) generates acetaldehyde that accumulates in the hippocampus, cortex and Cb, where the enzyme is more expressed [ref. 61]. Interestingly, the enzyme distribution coincides in brain regions with high CB1 receptor expression, such as the cortex and Cb, both strikingly affected by adolescent binge drinking. EtOH decreases glutathione in these same regions, thus increasing oxidative processes and brain damage in a model of prenatal EtOH exposure [ref. 17]. As class III ADH is a glutathione-dependent formaldehyde dehydrogenase, it is likely that the glutathione reduction elicited by EtOH and the consequent oxidative state leads to enzyme malfunction, jeopardizing EtOH elimination in regions where the enzyme is more abundant.
3.2. N-3 Recovers CB1 Receptor Expression in the Brain
DHA and AA are major phospholipid components of brain cell membranes [ref. 16,ref. 19]. EtOH reduces DHA in the brain [ref. 20,ref. 21] and its deficit impacts on both cell membranes, altering their biophysical properties, and related membrane proteins, such as enzymes and receptors [ref. 19]. N-3 deficiency lowers CB1 receptors in different brain regions [ref. 62] and impairs endocannabinoid-mediated synaptic plasticity [ref. 22]. The Fr3, OB, Cb, hippocampus, midbrain and striatum rank in high-to-low order among the brain regions with more DHA [ref. 63]. However, the negative effect on CB1 receptor expression in the adult brain after adolescent binge drinking was particularly outstanding in the M2, Cb and Acb (ventral striatum), with no effect in the hippocampus, dorsal striatum, some cortical areas, OB and SN. A DHA-enriched diet counteracts the low brain n-3 PUFA levels due to EtOH intake [ref. 21] and reverses EtOH-induced impairment of synaptic plasticity [ref. 18]. It also restores aquaporin-4, PLA-2 and glutathione affected by EtOH [ref. 17,ref. 21,ref. 64,ref. 65]. The recovery of CB1 receptor immunostaining by n-3 supplementation during the abstinence period points to the normalization of cell membrane homeostasis.
In conclusion, abusive EtOH consumption during adolescence alters CB1 receptor immunostaining optical density in some brain regions of the adult mouse, and an n-3-enriched diet recovers the reduced CB1 expression in limbic and motor structures following binge drinking. Uncovering the PUFA effects and mechanisms by which the n-3-enriched diet can impact on brain cannabinoid receptor expression (as shown in this paper) and function after adolescent binge drinking, could be an appropriate non-pharmacological approach to counteract the EtOH impact on cannabinoid-dependent synaptic plasticity, cognition and behavior.
4. Material & Methods
4.1. Generation of CB1-KO
CB1-knockout (CB1-KO) mice were generated and genotyped as previously described [ref. 66] and formerly collected [ref. 30]. They were obtained by crossing CB1f/f mice with CMV-Cre mice (“Cre deleter”). Mice were of a predominant C57BL/6-N background (9–10 back-crossings) and the breeding strategy used was female CB1+/− × male CB1+/− (Table 2).
Table 2: CB1-knockout mice.
| Name | Mouse Line Derived from | Background | Breeding Strategy Used |
|---|---|---|---|
| CB1-KO | CB1-KO (CB1−/−)Originally obtained by crossing CB1f/f mice with CMV-Cre mice (“Cre deleter”) [ref. 66] | Predominant C57BL/6-N(9–10 back-crossings) | Female CB1+/−XMale CB1+/− |
CB1, Type-1 cannabinoid; CB1-KO, Cannabinoid type-1 receptor knockout mouse.
4.2. Animal Treatment
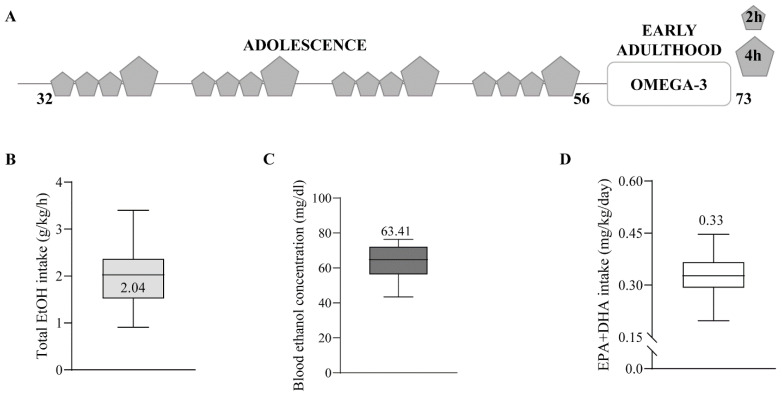
Four-week-old C57BL/6J male mice (Janvier Labs, Le Genest-Saint-Isle, France) were housed in pairs and habituated to a dark cycle (8 a.m.–8 p.m.). Then, they were exposed to water or drinking-in-the-dark (DID) during adolescence, as previously described [ref. 45]. Briefly, the mice were individualized and exposed to a bottle of 10 mL tap water or EtOH 20% (Boter S.L., Barcelona, Spain) four days a week during four weeks (postnatal day (PND) 32 to 56). They had free access to the bottle for 2 h the first three days, and 4 h the fourth day. Mice were resting and kept in pairs with food and water ad libitum for the last three days of the week (Figure 4A–C). On PND 56, a blood sample was collected from the lateral tail vein using a capillary tube 30 min after 4 h of EtOH exposure (Sarstedt, Nümbrecht, Germany). Blood samples were analyzed for EtOH concentration using a commercial OH assay kit (Abcam ab65646, Madrid, Spain), following manufacturer instructions. Half of the mice were fed 2% EPA and DHA (2.2% EPA and 1.5% DHA of total fats; SAFE, Augy, France) during withdrawal (PND 56–73). Twice a week, mice and food were weighed to measure EPA and DHA intake (mg/kg/day) (Figure 4D). Three mice/group were culled on PND 73. They were deeply anesthetized using 4% chloral hydrate (10 mL/kg body weight, i.p.) and perfused through the left ventricle with 30 mL phosphate-buffered saline (PBS, 0.1 M, pH 7.4), followed by 80 mL of the fixative (4% formaldehyde depolymerized from paraformaldehyde, 0.2% picric acid, 0.1% glutaraldehyde) prepared in PBS at room temperature (RT). Brains were removed and post-fixed as described elsewhere in detail [ref. 59].
4.3. Antibody Characterization
The specificity of the CB1 receptor antibody (Nittobo Medical Co., Ltd., Tokyo, Japan); goat polyclonal, CB1-Go-Af450, RRID AB_2571592, Table 3) has been tested thoroughly [ref. 3,ref. 4,ref. 30]. In addition, CB1 receptor staining was not detected in the CB1-KO mouse brain (Figure 5).
Table 3: Primary antibody used for immunohistochemistry.
| Antibody | Immunogen | Manufacturer, Species, Catalog Number, Rrid | Dilution | Characterization |
|---|---|---|---|---|
| ANTI-CB1 | Recognizes the last 31 aminoacids of the C-terminus of the mouse CB1 receptor (NM007726), as provided by the manufacturer: NCBI Reference Sequence: NP_031752.1; 443–473 amino acid residues: MHRAAESCIKSTVKIAKVTMSVSTDTSAEAL | Frontier Institute; Goatpolyclonal; #CB1-Go-Af450, RRID:AB_2571592 | 2 µg/mL | On immunoblot, the antibody detects asingle protein band at 52 kDa |

4.4. Immunohistochemistry for Light Microscopy
This was performed following the protocol previously published [ref. 30]. Briefly, coronal and sagittal sections cut at 50 μm on a vibratome (Leica VT 1000s, Wetzlar, Germany) were taken rostro-caudally from the whole brain and Cb, respectively, and collected in phosphate buffer (PB 0.1 M, pH 7.4) at RT. They were pre-incubated in a blocking solution for 30 min at RT, and then incubated in goat anti-CB1 receptor antibody (2 μg/mL) diluted in 10% horse serum, 0.1% sodium azide, 0.5% Triton in 1× Tris-HCl-buffered saline (TBS) overnight at RT. After washing in 1% horse serum and 0.5% Triton in 1× TBS the next day, tissue sections were incubated with a horse anti-goat IgG biotinylated antibody (H + L) (1:200, Vector Labs, Newark, CA, USA, cat#BA9500; RRID: AB_2336123) for 1 h at RT. Following several washes, they were incubated in avidin-biotin peroxidase complex (1:50, Vector Labs, Newark, CA, USA, Cat#PK-6100, RRID: AB_2336819) for 1 h at RT. Tissue was washed several times with the last two containing 0.5% Triton in PB, and incubated in 0.05% DAB (Sigma-Aldrich, Merck KGaA, Darmstadt, Germany, Cat#D5637; RRID: AB_2336819) with 0.01% hydrogen peroxide prepared in 0.1 M PB for 3 min. Finally, following five washes with 0.5% Triton in PB, the sections were mounted, dehydrated and coverslipped with DPX (Sigma Aldrich, Merck KGaA, Darmstadt, Germany Cat#44581).
4.5. Semiquantitative Analysis of CB1 Receptor Optical Density
Brain and cortical regions known to express CB1 receptors were selected: OB, M1, M2, Fr3, Cg1, cg, CPu, Acb, Amg, DG, hippocampal CA1, SN, Ent and Cb. Micrographs were taken at 10× using a light microscope (Olympus BX61, Hamburg, Germany) and processed using the Olympus cellSens Dimension using consecutive sections containing the brain regions and cortical areas of interest. For each region, three independent optical density measurements were performed at 10×, and two more were taken in a blank zone to rank background level. As some regions were through several slides, they were analyzed repeatedly in each mouse of the four experimental conditions (Table 4). All measurements were then pooled by mouse. Subsequently, data were normalized to 100% of the H2O group. Image J software (1.8.0_322); NIH; RRID:SCR_003070) and a statistical software package were used (GraphPad Prism 8; RRID: SCR_002798). The Shapiro–Wilk normality test was applied before running one-way ANOVA. Parametric data were analyzed using Holm Sidak’s multiple comparison test and non-parametric data using Dunn’s multiple comparison test. All values are given as mean ± SEM.
Table 4: Number of measurements taken in the olfactory bulb (OB), primary and secondary motor cortex (M1, M2), frontal cortex (Fr3), cingular cortex area 1 (Cg1), cingulum (cg), caudate putamen (CPu), nucleus accumbens (Acb), amygdala (Amg), dentate gyrus (DG), CA1 hippocampus, substantia nigra (SN), entorhinal cortex (Ent) and cerebellum (Cb) for each experimental condition (n = 3 mice/group).
| H2O | EtOH | n-3-EtOH | n-3-H2O | |
|---|---|---|---|---|
| OB | 39 | 54 | 45 | 30 |
| M1 | 72f | 69 | 48 | 66 |
| M2 | 72 | 63 | 48 | 66 |
| Fr3 | 18 | 18 | 18 | 18 |
| Cg1 | 18 | 18 | 12 | 18 |
| cg | 102 | 105 | 84 | 70 |
| CPu | 18 | 18 | 15 | 18 |
| Acb | 18 | 18 | 12 | 18 |
| Amg | 90 | 75 | 54 | 60 |
| DG | 51 | 51 | 45 | 36 |
| CA1 | 36 | 36 | 27 | 21 |
| SN | 51 | 51 | 42 | 36 |
| Ent | 36 | 36 | 21 | 24 |
| Cb | 45 | 30 | 24 | 36 |
References
- H.-C. Lu, K. Mackie. An introduction to the endogenous cannabinoid system. Biol. Psychiatry, 2016. [DOI | PubMed]
- R. Mechoulam, L.A. Parker. The endocannabinoid system and the brain. Annu. Rev. Psychol., 2013. [DOI | PubMed]
- A. Gutiérrez-Rodríguez, I. Bonilla-Del Río, N. Puente, S.M. Gómez-Urquijo, C.J. Fontaine, J. Egaña-Huguet, I. Elezgarai, S. Ruehle, B. Lutz, L.M. Robin. Localization of the cannabinoid type-1 receptor in subcellular astrocyte compartments of mutant mouse hippocampus. Glia, 2018. [DOI | PubMed]
- E. Hebert-Chatelain, T. Desprez, R. Serrat, L. Bellocchio, E. Soria-Gomez, A. Busquets-Garcia, A.C. Pagano Zottola, A. Delamarre, A. Cannich, P. Vincent. A cannabinoid link between mitochondria and memory. Nature, 2016. [DOI | PubMed]
- I. Katona, T.F. Freund. Multiple functions of endocannabinoid signaling in the brain. Annu. Rev. Neurosci., 2012. [DOI | PubMed]
- C. Carbia, F. Cadaveira, F. Caamaño-Isorna, S. Rodríguez-Holguín, M. Corral. Binge drinking during adolescence and young adulthood is associated with deficits in verbal episodic memory. PLoS ONE, 2017. [DOI | PubMed]
- B. Lutz, G. Marsicano, R. Maldonado, C.J. Hillard. The endocannabinoid system in guarding against fear, anxiety and stress. Nat. Rev. Neurosci., 2015. [DOI | PubMed]
- M. Pava, J. Woodward. A review of the interactions between alcohol and the endocannabinoid system: Implications for alcohol dependence and future directions for research. Alcohol, 2012. [DOI | PubMed]
- I. Rico-Barrio, S. Peñasco, L. Lekunberri, M. Serrano, J. Egaña-Huguet, A. Mimenza, E. Soria-Gomez, A. Ramos, I. Buceta, I. Gerrikagoitia. Environmental enrichment rescues endocannabinoid-dependent synaptic plasticity lost in young adult male mice after ethanol exposure during adolescence. Biomedicines, 2021. [DOI | PubMed]
- S. Mitrirattanakul, H.E. López-Valdés, J. Liang, Y. Matsuka, K. Mackie, K.F. Faull, I. Spigelman. Bidirectional alterations of hippocampal cannabinoid 1 receptors and their endogenous ligands in a rat model of alcohol withdrawal and dependence. Alcohol. Clin. Exp. Res., 2007. [DOI | PubMed]
- K.Y. Vinod, S.A. Kassir, B.L. Hungund, T.B. Cooper, J.J. Mann, V. Arango. Selective alterations of the CB1 receptors and the fatty acid amide hydrolase in the ventral striatum of alcoholics and suicides. J. Psychiatr. Res., 2010. [DOI | PubMed]
- J. Ceccarini, T. Hompes, A. Verhaeghen, C. Casteels, H. Peuskens, G. Bormans, S. Claes, K. Van Laere. Changes in cerebral CB1 receptor availability after acute and chronic alcohol abuse and monitored abstinence. J. Neurosci., 2014. [DOI | PubMed]
- J. Hirvonen, P. Zanotti-Fregonara, J.C. Umhau, D.T. George, D. Rallis-Frutos, C.H. Lyoo, C.T. Li, C.S. Hines, H. Sun, G.E. Terry. Reduced cannabinoid CB 1 receptor binding in alcohol dependence measured with positron emission tomography. Mol. Psychiatry, 2013. [DOI | PubMed]
- P. Maccioni, G. Colombo, M. Carai. Blockade of the cannabinoid cb1 receptor and alcohol dependence: Preclinical evidence and preliminary clinical data. CNS Neurol. Disord. Drug Targets, 2010. [DOI | PubMed]
- S. Peñasco, I. Rico-Barrio, N. Puente, C.J. Fontaine, A. Ramos, L. Reguero, I. Gerrikagoitia, F. Rodríguez de Fonseca, S. Barrondo, X. Aretxabala. Intermittent ethanol exposure during adolescence impairs cannabinoid type 1 receptor- dependent long-term depression and recognition memory in adult mice. Neuropsychopharmacology, 2020. [DOI | PubMed]
- C. Joffre, C. Rey, S. Layé. N-3 polyunsaturated fatty acids and the resolution of neuroinflammation. Front. Pharmacol., 2019. [DOI | PubMed]
- A.R. Patten, P.S. Brocardo, B.R. Christie. Omega-3 supplementation can restore glutathione levels and prevent oxidative damage caused by prenatal ethanol exposure. J. Nutr. Biochem., 2013. [DOI | PubMed]
- A.R. Patten, H.M. Sickmann, R.A. Dyer, S.M. Innis, B.R. Christie. Omega-3 fatty acids can reverse the long-term deficits in hippocampal synaptic plasticity caused by prenatal ethanol exposure. Neurosci. Lett., 2013. [DOI | PubMed]
- M. Serrano, I. Rico-Barrio, P. Grandes. The effect of omega-3 fatty acids on alcohol-induced damage. Front. Nutr., 2023. [DOI | PubMed]
- G.L. Milne, J.D. Morrow, M.J. Picklo. Elevated oxidation of docosahexaenoic acid, 22:6 (n-3), in brain regions of rats undergoing ethanol withdrawal. Neurosci. Lett., 2006. [DOI | PubMed]
- N. Tajuddin, K.H. Moon, S.A. Marshall, K. Nixon, E.J. Neafsey, H.Y. Kim, M.A. Collins. Neuroinflammation and neurodegeneration in adult rat brain from binge ethanol exposure: Abrogation by docosahexaenoic acid. PLoS ONE, 2014. [DOI | PubMed]
- M. Lafourcade, T. Larrieu, S. Mato, A. Duffaud, M. Sepers, I. Matias, V. De Smedt-Peyrusse, V.F. Labrousse, L. Bretillon, C. Matute. Nutritional omega-3 deficiency abolishes endocannabinoid-mediated neuronal functions. Nat. Neurosci., 2011. [DOI | PubMed]
- A. Manduca, A. Bara, T. Larrieu, O. Lassalle, C. Joffre, S. Layé, O.J. Manzoni. Amplification of mGlu5-endocannabinoid signaling rescues behavioral and synaptic deficits in a mouse model of adolescent and adult dietary polyunsaturated fatty acid imbalance. J. Neurosci., 2017. [DOI | PubMed]
- A. Thomazeau, C. Bosch-Bouju, O. Manzoni, S. Layé. Nutritional n-3 PUFA deficiency abolishes endocannabinoid gating of hippocampal long-term potentiation. Cereb. Cortex, 2017. [DOI | PubMed]
- J.P. Pan, H.Q. Zhang, W. Wang, Y.F. Guo, N. Xiao, X.H. Cao, L.J. Liu. Some subtypes of endocannabinoid/endovanilloid receptors mediate docosahexaenoic acid-induced enhanced spatial memory in rats. Brain Res., 2011. [DOI | PubMed]
- A. Artmann, G. Petersen, L.I. Hellgren, J. Boberg, C. Skonberg, C. Nellemann, S.H. Hansen, H.S. Hansen. Influence of dietary fatty acids on endocannabinoid and N-acylethanolamine levels in rat brain, liver and small intestine. Biochim. Biophys. Acta—Mol. Cell Biol. Lipids, 2008. [DOI]
- J.A.T. Wood, J.S. Williams, L. Pandarinathan, D.R. Janero, C.J. Lammi-Keefe, A. Makriyannis. Dietary docosahexaenoic acid supplementation alters select physiological endocannabinoid-system metabolites in brain and plasma. J. Lipid Res., 2010. [DOI | PubMed]
- J.E. Watson, J.S. Kim, A. Das. Emerging class of omega-3 fatty acid endocannabinoids & their derivatives. Prostaglandins Other Lipid Mediat., 2019. [DOI | PubMed]
- E. Soria-Gómez, L. Bellocchio, L. Reguero, G. Lepousez, C. Martin, M. Bendahmane, S. Ruehle, F. Remmers, T. Desprez, I. Matias. The endocannabinoid system controls food intake via olfactory processes. Nat. Neurosci., 2014. [DOI | PubMed]
- A. Gutiérrez-Rodríguez, N. Puente, I. Elezgarai, S. Ruehle, B. Lutz, L. Reguero, I. Gerrikagoitia, G. Marsicano, P. Grandes. Anatomical characterization of the cannabinoid CB1 receptor in cell-type–specific mutant mouse rescue models. J. Comp. Neurol., 2017. [DOI | PubMed]
- B.D. Winters, J.M. Krüger, X. Huang, Z.R. Gallaher, M. Ishikawa, K. Czaja, J.M. Krueger, Y.H. Huang, O.M. Schlüter, Y. Dong. Cannabinoid receptor 1-expressing neurons in the nucleus accumbens. Proc. Natl. Acad. Sci. USA, 2012. [DOI | PubMed]
- K.L. Hanson, K.L. Medina, C.B. Padula, S.F. Tapert, S. Brown. Impact of adolescent alcohol and drug use on neuropsychological functioning in young adulthood: 10-year outcomes. J. Child Adolesc. Subst. Abus., 2011. [DOI]
- B. Lees, J. Debenham, L.M. Squeglia. Alcohol and cannabis use and the developing brain. Alcohol Res. Curr. Rev., 2021. [DOI]
- L.G. Coleman, J. He, J. Lee, M. Styner, F. Crews. Adolescent binge drinking alters adult brain neurotransmitter gene expression, behavior, brain regional volumes, and neurochemistry in mice. Alcohol. Clin. Exp. Res., 2011. [DOI | PubMed]
- C. Guerri, M. Pascual. Mechanisms involved in the neurotoxic, cognitive, and neurobehavioral effects of alcohol consumption during adolescence. Alcohol, 2010. [DOI | PubMed]
- J. Manzanares, D. Cabañero, N. Puente, M.S. García-Gutiérrez, P. Grandes, R. Maldonado. Role of the endocannabinoid system in drug addiction. Biochem. Pharmacol., 2018. [DOI | PubMed]
- A. Flores-Bonilla, H. Richardson. Sex differences in the neurobiology of alcohol use disorder. Alcohol Res., 2020. [DOI]
- S. Alfonso-Loeches, M. Pascual, C. Guerri. Gender differences in alcohol-induced neurotoxicity and brain damage. Toxicology, 2013. [DOI | PubMed]
- M.E. Maynard, E.A. Barton, C.R. Robinson, J.I. Wooden, J.L. Leasure. Sex differences in hippocampal damage, cognitive impairment, and trophic factor expression in an animal model of an alcohol use disorder. Brain Struct. Funct., 2018. [DOI | PubMed]
- C.F. Moore, W.J. Lynch. Alcohol preferring (P) rats as a model for examining sex differences in alcohol use disorder and its treatment. Pharmacol. Biochem. Behav., 2015. [DOI | PubMed]
- M. Egertova, D.K. Giang, B.F. Cravatt, M.R. Elphick. A new perspective on cannabinoid signalling: Complimentary localization of fatty acid amide hydrolase and the CB1 receptor in rat brain. Proc. R. Soc. London Ser. B Biol. Sci., 1998. [DOI]
- K. Tsou, S. Brown, M.C. Sañudo-Peña, K. Mackie, J.M. Walker. Immunohistochemical distribution of cannabinoid CB1 receptors in the rat central nervous system. Neuroscience, 1998. [DOI | PubMed]
- B. Lees, L.R. Meredith, A.E. Kirkland, B.E. Bryant, L.M. Squeglia. Effect of alcohol use on the adolescent brain and behavior. Pharmacol. Biochem. Behav., 2020. [DOI | PubMed]
- M.L. Risher, R.L. Fleming, N. Boutros, S. Semenova, W.A. Wilson, E.D. Levin, A. Markou, H.S. Swartzwelder, S.K. Acheson. Long-term effects of chronic intermittent ethanol exposure in adolescent and adult rats: Radial-arm maze performance and operant food reinforced responding. PLoS ONE, 2013. [DOI | PubMed]
- I. Rico-Barrio, S. Peñasco, N. Puente, A. Ramos, C.J. Fontaine, L. Reguero, M.E. Giordano, I. Buceta, I. Terradillos, L. Lekunberri. Cognitive and neurobehavioral benefits of an enriched environment on young adult mice after chronic ethanol consumption during adolescence. Addict. Biol., 2019. [DOI | PubMed]
- N. Li, T.W. Chen, Z.V. Guo, C.R. Gerfen, K. Svoboda. A motor cortex circuit for motor planning and movement. Nature, 2015. [DOI | PubMed]
- J.D. Schmahmann. The cerebellum and cognition. Neurosci. Lett., 2019. [DOI | PubMed]
- I. Buceta, I. Elezgarai, I. Rico-Barrio, I. Gerrikagoitia, N. Puente, P. Grandes. Deletion of the cannabinoid CB1 receptor impacts on the ultrastructure of the cerebellar parallel fiber-Purkinje cell synapses. J. Comp. Neurol., 2019. [DOI | PubMed]
- C. Albergaria, N.T. Silva, D. Darmohray, M.R. Carey. Cannabinoids modulate associative cerebellar learning via alterations in behavioral state. eLife, 2020. [DOI | PubMed]
- Y. Kishimoto, M. Kano. Endogenous cannabinoid signaling through the CB1 receptor is essential for cerebellum-dependent discrete motor learning. J. Neurosci., 2006. [DOI | PubMed]
- A. El Manira, A. Kyriakatos. The role of endocannabinoid signaling in motor control. Physiology, 2010. [DOI | PubMed]
- V.D. Reddy, P. Padmavathi, S. Bulle, A.V. Hebbani, S.B. Marthadu, N.C. Venugopalacharyulu, P. Maturu, N.C. Varadacharyulu. Association between alcohol-induced oxidative stress and membrane properties in synaptosomes: A protective role of vitamin E. Neurotoxicology Teratol., 2017. [DOI]
- K.H. Moon, N. Tajuddin, J. Brown, E.J. Neafsey, H.Y. Kim, M.A. Collins. Phospholipase A2, Oxidative Stress, and Neurodegeneration in Binge Ethanol-Treated Organotypic Slice Cultures of Developing Rat Brain. Alcohol. Clin. Exp. Res., 2014. [DOI | PubMed]
- B.S. Basavarajappa, N. Nagre, S. Xie, S. Subbanna. Elevation of endogenous anandamide impairs ltp, learning and memory through cb1 receptor signaling in mice. Hippocampus, 2014. [DOI | PubMed]
- S. Ortiz, J.M. Oliva, S. Pérez-Rial, T. Palomo, J. Manzanares. Chronic ethanol consumption regulates cannabinoid CB1 receptor gene expression in selected regions of rat brain. Alcohol Alcohol., 2004. [DOI | PubMed]
- B.S. Basavarajappa, M. Saito, T.B. Cooper, B.L. Hungund. Chronic ethanol inhibits the anandamide transport and increases extracellular anandamide levels in cerebellar granule neurons. Eur. J. Pharmacol., 2003. [DOI | PubMed]
- B. Ferrer, F.J. Bermúdez-Silva, A. Bilbao, L. Alvarez-Jaimes, I. Sanchez-Vera, A. Giuffrida, A. Serrano, E. Baixeras, S. Khaturia, M. Navarro. Regulation of brain anandamide by acute administration of ethanol. Biochem. J., 2007. [DOI | PubMed]
- L. Mikasova, L. Groc, D. Choquet, O.J. Manzoni. Altered surface trafficking of presynaptic cannabinoid type 1 receptor in and out synaptic terminals parallesls receptor desensitization.pdf. Proc. Natl. Acad. Sci. USA, 2008. [DOI | PubMed]
- I. Bonilla-Del Río, N. Puente, A. Mimenza, A. Ramos, M. Serrano, L. Lekunberri, I. Gerrikagoitia, B.R. Christie, P.C. Nahirney, P. Grandes. Acute Δ9-tetrahydrocannabinol prompts rapid changes in cannabinoid CB1 receptor immunolabeling and subcellular structure in CA1 hippocampus of young adult male mice. J. Comp. Neurol., 2020. [DOI]
- E. Leishman, M. Manchanda, R. Thelen, S. Miller, K. Mackie, H.B. Bradshaw. Cannabidiol’s Upregulation of N-acyl Ethanolamines in the Central Nervous System Requires N-acyl Phosphatidyl Ethanolamine-Specific Phospholipase D. Cannabis Cannabinoid Res., 2018. [DOI | PubMed]
- J. Waddell, M.C. McKenna, T. Kristian. Brain ethanol metabolism and mitochondria. Curr. Top. Biochem. Res., 2022. [PubMed]
- A.R. Isaac, P.C. de Velasco, K.Y.D. Fraga, M.d.G. Tavares-do-Carmo, R.M.P. Campos, F.A. Iannotti, R. Verde, D.B.G. Martins, T.A. Santos, B.K. Ferreira. Maternal omega-3 intake differentially affects the endocannabinoid system in the progeny`s neocortex and hippocampus: Impact on synaptic markers. J. Nutr. Biochem., 2021. [DOI | PubMed]
- I. Carrié, M. Clément, D. De Javel, H. Francès, J.M. Bourre. Specific phospholipid fatty acid composition of brain regions in mice: Effects of n-3 polyunsaturated fatty acid deficiency and phospholipid supplementation. J. Lipid Res., 2000. [DOI | PubMed]
- J. Brown, N. Achille, E.J. Neafsey, M.A. Collins. Binge ethanol-induced neurodegeneration in rat organotypic brain slice cultures: Effects of PLA2 inhibitor mepacrine and docosahexaenoic acid (DHA). Neurochem. Res., 2009. [DOI | PubMed]
- M.A. Collins, K. Moon, N. Tajuddin, E.J. Neafsey, Y. Kim. Docosahexaenoic acid (DHA) prevents binge ethanol-dependent aquaporin-4 elevations while inhibiting neurodegeneration: Experiments in rat adult-age entorhino-hippocampal slice cultures. Neurotox. Res., 2013. [DOI | PubMed]
- G. Marsicano, C.T. Wotjak, S.C. Azad. The endogenous cannabinoid system controls extinction of aversive memories. Nature, 2002. [DOI | PubMed]
