Cannabidiol attenuates epileptic phenotype and increases survival in a mouse model of developmental and epileptic encephalopathy type 1
Abstract
Objective:
Developmental and epileptic encephalopathy type 1 (DEE1) is a rare drug‐resistant pediatric epilepsy caused by trinucleotide repeat expansions in the X‐linked ARX gene, leading to elongation of the first polyalanine tract. It presents with early onset tonic seizures or spasms, developmental and cognition delay, and high risk of premature mortality. We evaluated the therapeutic potential of highly purified cannabidiol (CBD) in Arx (GCG)7/Y mice, a genetic DEE1 model that replicates key features of the human condition.
Methods:
Arx(GCG)7/Y mice received daily intraperitoneal CBD (100 mg/kg) for 7 days. The epileptic phenotype was evaluated via video monitoring and a scoring matrix. In Arx‐DEE1 male cortex, real‐time polymerase chain reaction and Western blotting assessed CBD effects on proinflammatory and neuronal markers. Microglial morphology was analyzed by Iba1 immunostaining and Sholl analysis. In vitro patch‐clamp recordings tested CBD activity on Arx (GCG)7/Y cortical neurons.
Results:
CBD reduced the severity and frequency of spontaneous recurrent seizures and significantly extended the lifespan of epileptic mice. In mutant symptomatic mice, CBD activated peroxisome Pparg expression and the concurrent desensitization/inactivation of TRPV1 channels. Additionally, CBD counteracted the dysregulated expression of the proinflammatory genes Ptgs2, Mmp9, Il12, Cd68, Ccl2, and Irf3, while also restoring normal microglial morphology. Further molecular analysis demonstrated that CBD effectively offsets normal alternative splicing for the presynaptic receptor genes Nrnx1 and Nrnx3. Consistent with this, CBD rescued proper Nrnx1 splicing in mutant cortical neurons after K+‐induced depolarization. Finally, we found that CBD reduced neuronal excitability by inducing hyperpolarization, raising the action potential threshold, and reducing the frequency and mean charge of inhibitory postsynaptic currents and the mean charge of excitatory postsynaptic currents.
Significance:
These findings represent the first preclinical evidence of CBD efficacy in a murine model of genetic DEE1, identifying CBD‐sensitive downstream targets and paving the way to further exploration of CBD effects in this disease for future clinical consideration.
Article type: Research Article
Keywords: Arx polyalanine mice, cannabidiol, neurexin splicing switches, pharmacoresistant seizure, TRPV1
Affiliations: Institute of Genetics and Biophysics “Adriano Buzzati‐Traverso,” National Research Council of Italy (CNR) Naples Italy; Institute of Biomolecular Chemistry, National Research Council of Italy Pozzuoli, Naples Italy; Istituto Neurologico Mediterraneo Pozzilli (Neuromed) ‐ Scientific Institute for Research , Hospitalization and Healthcare (IRCCS) Pozzilli (Isernia) Italy; Department of Human Sciences Society and Health University of Cassino and Southern Lazio Cassino (Frosinone) Italy; Department of Translational Medicine, Child Neurology, and Psychiatry University of Naples “Federico II” Naples Italy; Department of Physiology and Pharmacology Sapienza University of Rome Rome Italy; Joint International Unit Between the CNR of Italy and Université Laval (Quebec, Canada) on Chemical and Biomolecular Research on the Microbiome and Its Impact on Metabolic Health and Nutrition Quebec Canada
License: © 2025 The Author(s). Epilepsia published by Wiley Periodicals LLC on behalf of International League Against Epilepsy. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/epi.18522 | PubMed: 40608247 | PMC: PMC12605674
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (968 KB)
Key points
- Mice with GCG trinucleotide expansion in Arx gene (Arx (GCG)7/Y) present severe seizures and high risk of mortality.
- Treatment with CBD in young Arx‐mutated mice reduces frequency and severity of spontaneous recurrent seizures and increases overall survival.
- CBD treatment dampens neuroinflammation, reducing the expression of proinflammatory genes and correcting microglia morphology.
- The anti‐ictogenic effects of CBD may be mediated by decreasing neuronal excitability.
INTRODUCTION
Developmental and epileptic encephalopathies (DEEs) represent a clinically and genetically heterogeneous group of rare neurodevelopmental disorders (NDDs) characterized by early onset of drug‐resistant epilepsy, typical electroencephalographic (EEG) patterns, developmental delay or regression, particularly after the onset of refractory seizures, and a complex range of comorbidities, ranging from movement disorders to autismlike symptoms.ref. epi18522-bib-0001 To date, many DEE genes have been identified, including those encoding transcription factors, ion channels, or proteins involved in neurotransmission.ref. epi18522-bib-0002
Expanded runs of consecutive mixed (GCN)n repeats in the first and second polyalanine tract of the Aristaless‐related homeobox gene (ARX; Mendelian Inheritance in Man [MIM] 300382) have been identified in male children with a severe form of DEE, termed DEE type 1 (DEE1; MIM 308350), also known as infantile spasm syndrome X‐linked type 1(ISSX1).ref. epi18522-bib-0003, ref. epi18522-bib-0004 ARX is an X‐chromosome gene that encodes a bifunctional high‐hierarchy homeotic transcription factor with a pivotal role in cortex development.ref. epi18522-bib-0005 DEE1 is part of a phenotypic spectrum of ARX‐related diseases—generally affecting only male children—including lissencephaly (MIM 300215), Proud syndrome (MIM 300004), DEE1 (MIM 308350), and syndromic (MIM 309510) and nonsyndromic (MIM 300419) intellectual disability.ref. epi18522-bib-0005, ref. epi18522-bib-0006 ARX‐DEE1 patients typically develop pharmacoresistant infantile epileptic spasms (ES), associated with a characteristic pattern on EEG called hypsarrhythmia, development arrest, and high risk for premature death.ref. epi18522-bib-0007, ref. epi18522-bib-0008 Mechanistically, expanded ARX proteins show reduced transactivation activity and impaired DNA binding at specific gene‐regulatory regions, altering the broad ARX‐dependent transcriptional program.ref. epi18522-bib-0004, ref. epi18522-bib-0009, ref. epi18522-bib-0010 Given the broad spectrum of ARX functions, which are finely tuned both spatially and temporally with cellular specificity, a complete delineation of the multiple molecular and cellular processes damaged by polyalanine elongations remains a challenging and only partially accomplished task.ref. epi18522-bib-0011, ref. epi18522-bib-0012 In this context, our previous research revealed abnormalities in neuronal network formation, translation efficiency, and RNA splicing in the neonatal brains of Arx transgenic mice.ref. epi18522-bib-0012
Patients with ARX‐related DEE may display different epileptic phenotypes, ranging from early infantile epileptic encephalopathy with a suppression–burst pattern on EEG and myoclonic seizures to infantile ES, all characterized by severe pharmacoresistance.ref. epi18522-bib-0013, ref. epi18522-bib-0014 The limited effectiveness of current antiseizure medications (ASMs) highlights the persistent demand from clinicians and families for the identification of new therapeutic strategies for this early onset DEE. A promising pharmacological therapy with 17β‐estradiol (E2) has been tested in Arx (GCG)10+7/Y mice harboring the insertion of eight GCG alanine codons in the first Arx polyalanine stretch.ref. epi18522-bib-0015, ref. epi18522-bib-0016 This is a vital knockin Arx‐epileptic male model characterized by spontaneous spasmlike myoclonus in pups and seizures in young animals, in which presymptomatic E2 treatment halts spasmlike myoclonus.ref. epi18522-bib-0015, ref. epi18522-bib-0016 Beneficial effects of early E2 treatment were also observed in Arx (GCG)7/Y mice.ref. epi18522-bib-0017 This is a different knockin Arx‐ epileptic male model harboring the insertion of seven GCG alanine codons in the first polyalanine stretch.ref. epi18522-bib-0011, ref. epi18522-bib-0015, ref. epi18522-bib-0017, ref. epi18522-bib-0018, ref. epi18522-bib-0019 Arx (GCG)7/Y young mice exhibit severe spontaneous tonic–clonic seizures—which generally start at postnatal day (PND) 30—and present a high mortality incidence.ref. epi18522-bib-0011, ref. epi18522-bib-0015, ref. epi18522-bib-0017, ref. epi18522-bib-0018, ref. epi18522-bib-0019 In this Arx‐polyalanine model, E2 treatment ameliorates seizure frequency but has no effect on mortality rate.ref. epi18522-bib-0017 All this evidence highlights the urgent need to explore the potential efficacy of new ASMs suitable for these disorders.
In recent years, highly purified cannabidiol (CBD) derived from Cannabis sativa was approved by the US Food and Drug Administration and European Medicines Agency for the treatment of seizures associated with Dravet syndrome (DS; MIM 607208)ref. epi18522-bib-0020 Lennox–Gastaut syndrome,ref. epi18522-bib-0021 and tuberous sclerosis complexref. epi18522-bib-0022 in patients 2 years of age and older. Regarding its mechanism of action, CBD exerts a broad range of effects at the molecular and cellular level, influencing inflammation, pain, excitability, and other physiological and pathophysiological processes including peroxisome proliferator‐activated receptor gamma (PPARγ) receptors and transient receptor potential vanilloid subtype 1 (TRPV1) channels, among others.ref. epi18522-bib-0023, ref. epi18522-bib-0024 Although the anticonvulsant action of CBD has been observed in other DEEs, including infantile epileptic spasms syndrome patients, the lack of sufficient studies demonstrating its efficacy has hindered its approval for the treatment of additional DEEs.ref. epi18522-bib-0025, ref. epi18522-bib-0026
This study is the first to explore postnatal CBD treatment in epileptic Arx (GCG)7/Y mice, a preclinical model of early onset DEE1. We demonstrate that daily administration of CBD significantly improves the epileptic behavior and extends lifespan in Arx (GCG)7/Y mice. We further reveal unanticipated secondary molecular and cellular changes driven by Arx‐polyalanine elongation that CBD effectively reverses. Overall, these findings establish a preclinical framework for the antiseizure potential of CBD in DEE1 and provide scientific rationale for further exploration of the effects of CBD in this disease toward future clinical consideration.
MATERIALS AND METHODS
All materials and methods are described in Supporting Information.
RESULTS
CBD reduces seizure frequency in Arx‐DEE1 mice
To determine the effect of CBD on spontaneous recurrent seizures (SRS) in symptomatic Arx (GCG)7/Y young mice, we assessed seizure frequency and duration before, during, and after 1 week of daily intraperitoneal administration of CBD (100 mg/kg) or vehicle (Veh). The experimental workflow is depicted in Figure 1A, summarizing the treatment strategy including the baseline, treatment, and washout phases. Arx (GCG)7/Y young mice (PND28–30) were assigned in a random and blinded manner to receive Veh (study group 1 [sg1]: Veh‐treated Arx (GCG)7/Y, n = 10) or CBD (study group 2 [sg2]: CBD‐treated Arx (GCG)7/Y, n = 12).
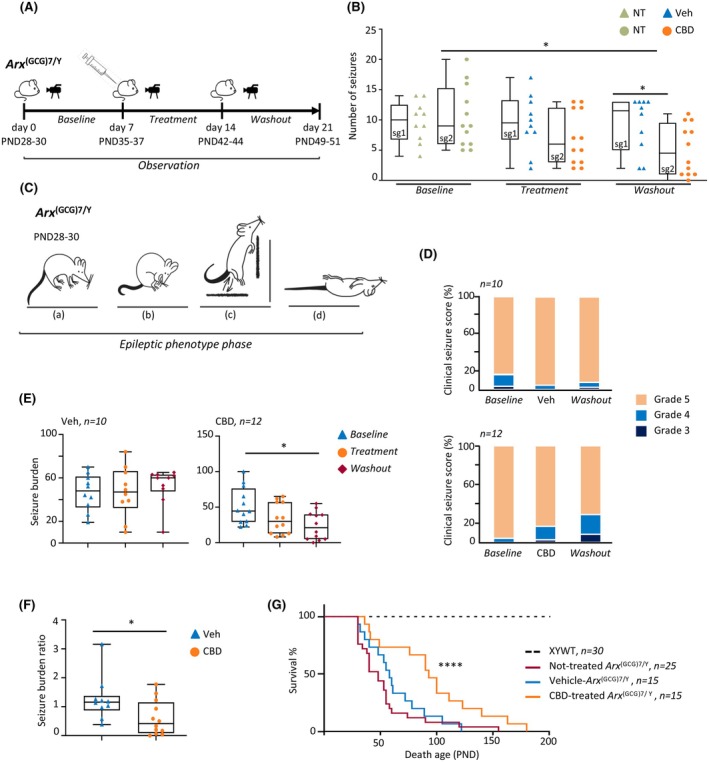
Before initiating treatments, the epileptic phenotype was analyzed in both study groups sg1 and sg2 (baseline; Figure 1A). All seizures started with trembling of the limbs and progressed to tonic–clonic convulsions, running fits, and then complete loss of postural control and movement, as previously described.ref. epi18522-bib-0019 Seven days of video recording showed that Arx (GCG)7/Y (n = 5) experienced frequent SRS (approximately 2 episodes per day) with a median duration of 30 s (Figure S1A and Video S1).
The two experimental groups sg1 Veh‐treated Arx (GCG)7/Y and sg2 CBD‐treated Arx (GCG)7/Y received once‐daily intraperitoneal injections respectively of Veh or CBD for 7 days (PND35–37) and were recorded (treatment); treatments were then discontinued, and mice (PND42–44) were recorded for the next 7 days (washout; Figure 1A). This additional recording time enabled us to assess seizure frequency after drug washout and to address any potential disease‐modifying effects of CBD (Figure 1A). No differences in SRS frequency were detected in Veh‐treated Arx (GCG)7/Y during treatment and after washout phases. On the contrary, we observed a significant reduction in seizure frequency in CBD‐treated Arx (GCG)7/Y during the washout phase in comparison to the baseline and treatment phases (Figure 1B). Specifically, median seizures were 9 (interquartile range [IQR] = 9.25–6, n = 125 total seizures) during the baseline period, 6 (IQR = 12–3, n = 84 total seizures) during CBD daily treatment, and finally, 4.25 (IQR = 9.5–1, n = 61 seizures) during the posttreatment period. Concerning the seizure duration, we found a similar distribution during baseline (29 s, IQR = 40–20), treatment (35 s, IQR = 45–25), and washout phases (35 s, IQR = 40–23) in the two experimental groups CBD‐treated Arx (GCG)7/Y and Veh‐treated Arx (GCG)7/Y (Figure S1B). Given these results, we conclude that CBD reduces the frequency of SRS in Arx (GCG)7/Y mice in a time‐dependent manner, without affecting seizure duration. Importantly, its effects persist, although briefly (for 7 days), after the treatment is discontinued.
CBD reduces seizure severity and extends lifespan of Arx‐DEE1 mice
Next, we assessed whether CBD alleviates disease severity in addition to seizure frequency. As summarized in Figure 1C, Arx (GCG)7/Y mice show very severe SRS, with head nodding, forelimb clonus, loss of postural tone, rearing, falling, wild jumping, and finally sudden death, mainly scored as 4 and 5 according to the Racine scale score of severity (Table S1).ref. epi18522-bib-0027 Clinical seizure score was evaluated upon behavioral analysis in both experimental groups CBD‐treated Arx (GCG)7/Y and Veh‐treated Arx (GCG)7/Y. In the baseline phase, seizures were mostly graded 5 as expected in both experimental groups, and during and after the Veh treatments (Figure 1D, upper panel). During CBD treatments, 3.6% and 15.5% of CBD‐treated Arx (GCG)7/Y were grade 3 and 4, respectively; during post‐CBD treatments, 11.5% and 18% of CBD‐treated Arx (GCG)7/Y were grade 3 and 4, respectively (Figure 1D, bottom panel). Given that both seizure severity and frequency contribute significantly to disease manifestation, we also measured the seizure burden, a comprehensive parameter that accounts for both the severity of the epilepsy phenotype and the frequency of seizures.ref. epi18522-bib-0028 We found that the Veh‐treated group showed stable seizure burden median values with no significant differences at baseline (median value = 48), treatment (median value = 47), and washout (median value = 60) phases (Figure 1E). On the contrary, a significantly lower seizure burden was observed in the CBD group in the washout (median value = 21) compared to the baseline (median value = 44.5; Figure 1E). In line with the beneficial effects of CBD, we also observed a significantly lower seizure burden ratio in CBD‐treated Arx (GCG)7/Y mice with respect to Veh‐treated Arx (GCG)7/Y mice (Figure 1F).
To determine whether CBD administration affects mortality risk, we compared the survival rates of CBD‐treated mutant mice with those of untreated and Veh‐treated groups, excluding animals in the untreated group that died before weaning. Notably, Arx (GCG)7/Y mice typically experience early mortality, often as a result of severe epileptic fits.ref. epi18522-bib-0019, ref. epi18522-bib-0029 Consistent with previous studies, we found that approximately 38% of Arx (GCG)7/Y mice died before weaning (PND28), and the majority of those that survived weaning died within 3 months of age (PND90; Figure 1G, Figure S1C). Upon treatments, we observed a significant increase in survival of CBD‐treated Arx (GCG)7/Y with a median survival of 93 days compared to the Veh‐treated Arx (GCG)7/Y with a median survival of 58 days (log‐rank test, p < .0001), suggesting a strong protective CBD effect (Figure 1G). No significant differences in weight changes were observed between the Veh‐treated and CBD‐treated groups (Figure S1D). Collectively, these results demonstrate the antiepileptic effects of CBD in improving epileptic outcomes and reducing mortality in Arx (GCG)7/Y mice, highlighting the potential of CBD as a primary treatment option following a DEE1 diagnosis.
CBD offsets the defective expression levels of the cannabinoid‐related targets PPARγ and TRPV1
Recent investigations have increasingly elucidated the role of cannabinoid‐related targets in mediating the anticonvulsant effects of CBD. Notably, CBD anti‐ictogenic properties have been associated with its interaction with specific molecular pathways, particularly involving PPARγ and the TRPV1 channels. Remarkably, the upregulation of PPARγ was found to correlate with the anticonvulsant action of CBD, suggesting a possible contribution of this receptor in epilepsy management.ref. epi18522-bib-0030 Additionally, TRPV1 channels, which are cation‐permeant, have been identified as contributors to neuronal excitability. These channels are often found to be overexpressed in the brains of various rodent models, ex vivo systems, and postmortem analyses of patients with epilepsy. By desensitizing TRPV1 channels, CBD could reduce neurotransmission and neuronal excitability, thereby exerting its anticonvulsant effects. Similar outcomes have been observed with TRPV1 agonists, such as capsaicin, and antagonists such as capsazepine, or TRPV1 knockout, further supporting this mechanism.ref. epi18522-bib-0031
In light of these findings, we moved to explore whether the effects of CBD we observed in DEE1 mice were dependent on PPARγ and TRPV1. To do this, Arx (GCG)7/Y young mice were treated by daily intraperitoneal administration with CBD (100 mg/kg, n = 5) or Veh (n = 5) for 7 days, at the same age as the behavioral study (PND35–37; Figure 1). We first performed quantitative real‐time polymerase chain reaction (qRT‐PCR) of Pparg mRNA levels in the cortex isolated from Arx (GCG)7/Y‐Veh and Arx (GCG)7/Y‐CBD mice and sex‐ and age‐matched wild type male controls (XYWT). We found a striking reduction in Pparg transcript levels in the epileptic cortex of Veh‐treated Arx (GCG)7/Y mice compared to XYWT mice (Figure 2A). CBD completely restores Pparg transcript levels to match those of control mice, suggesting that Pparg downregulation may be an intrinsic mechanism contributing to the DEE1 pathophysiology (Figure 2A). Notably, the Pparg gene is a confirmed ARX target,ref. epi18522-bib-0032 containing a 5′‐TAATTA‐3′ ARX binding motifref. epi18522-bib-0004 located in a conserved noncoding element of its promoter region (Figure S2).ref. epi18522-bib-0033 Given the established antineuroinflammation properties of PPARG,ref. epi18522-bib-0034 we next assessed whether CBD could also exert a favorable effect on three PPARγ target genes involved in inflammation: Mmp‐9 and Ptgs2, which are negative PPARγ targets and mediate proinflammatory responses,ref. epi18522-bib-0034 and Socs3, a gene transcriptionally activated by PPARγ that suppresses cytokine signaling (Figure 2B,C).ref. epi18522-bib-0035 Consistent with the Pparg levels observed in Veh‐treated Arx (GCG)7/Y and CBD‐treated Arx (GCG)7/Y mice, the transcript levels of Mmp‐9 and Ptgs2 were found respectively increased in Veh‐treated mutant cortex and decreased in CBD‐treated mutant cortex (Figure 2B). In contrast, Socs3 was significantly downregulated in the epileptic cortex of both Veh‐treated Arx (GCG)7/Y and CBD‐treated Arx (GCG)7/Y mice (Figure 2B), indicating that CBD specifically targets the transcriptional axis of Pparg with Mmp‐9 and Ptgs2, while leaving Socs3 expression unchanged.
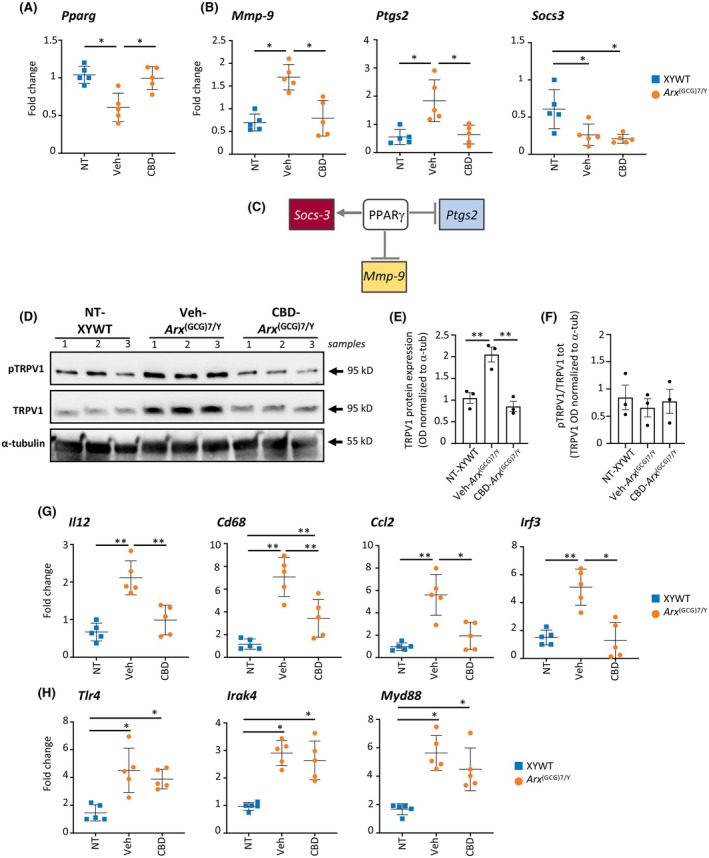
By Western blot analysis, we then measured the protein levels of TRPV1 and phosphoTRPV1 (Ser502 residue) in the cortex isolated from Veh‐treated Arx (GCG)7/Y and CBD‐treated Arx (GCG)7/Y mice and sex‐ and age‐matched controls (XYWT) using specific antibodies (Figure 2D–F). Consistent with previous research showing that Ptgs2 expression increases along with the upregulation of TRPV1,ref. epi18522-bib-0036 we found significantly higher levels of TRPV1 and phosphorylated TRPV1 (pTRPV1) in the cortex of epileptic Veh‐treated Arx (GCG)7/Y mice compared to XYWT mice (Figure 2D–F). In contrast, in the cortex of CBD‐treated Arx (GCG)7/Y mice, the levels of these proteins were comparable to those in control animals (Figure 2D–F). Altogether, these findings suggest that CBD, also in the epileptic model we analyzed, exerts its beneficial effects through the promotion of PPARγ expression and concomitant inactivation of TRPV1 channels. Furthermore, they highlight a newly identified role of these CBD‐sensitive markers as key factors in the onset of the epileptic phenotype in DEE1 mice.
CBD suppresses the mRNA levels of the proinflammatory genes Il12, Cd68, Ccl2, and Irf3
Given the extensive anti‐inflammatory activity of CBD, we next focused on analyzing potential anti‐inflammatory targets that could underlie the beneficial effects observed on the epileptic phenotype of DEE1 mice. To this end, we analyzed the transcript levels of the inflammasome mediators Il1β, Il6, Il12, Il10, Tnfα, Cd68, Ccl2, and Ccl5, which are upregulated in pilocarpine‐ and kainic acid‐induced epilepsy models.ref. epi18522-bib-0037, ref. epi18522-bib-0038 Furthermore, the mRNA levels of Irf3, Tlr4, Myd88, and Irak4 genes—encoding innate immune system markers essential for maintaining neuronal excitation/inhibition balance and implicated in epilepsy—were examined.ref. epi18522-bib-0039, ref. epi18522-bib-0040 qRT‐PCR analysis was carried out in the cortex isolated from Arx (GCG)7/Y young mice treated by daily intraperitoneal administration with CBD (100 mg/kg, n = 5) or Veh (n = 5) for 7 days, at the same age as the behavioral study (PND35–37; Figure 1). We found that the expression levels of Il12, Cd68, Ccl2, and Irf3 were significantly elevated in the Arx (GCG)7/Y cortex, and these increases were notably reduced with CBD treatment (Figure 2G). In contrast, CBD had no effect on the upregulation of Tlr4, Irak4, or Myd88 (Figure 2H), and no significant differences were observed in the levels of Tnfα, Il10, Il1β, Il6, and Ccl5 between mutant and control mice (Figure S3). Overall, alongside the observed reversal of the Pparg‐ Ptgs2‐Mmp9 axis and TRPV1 protein levels, these results strongly support the hypothesis that CBD exerts its therapeutic effects on the DEE1 phenotype by modulating a specific set of pro‐ and anti‐inflammatory molecules.
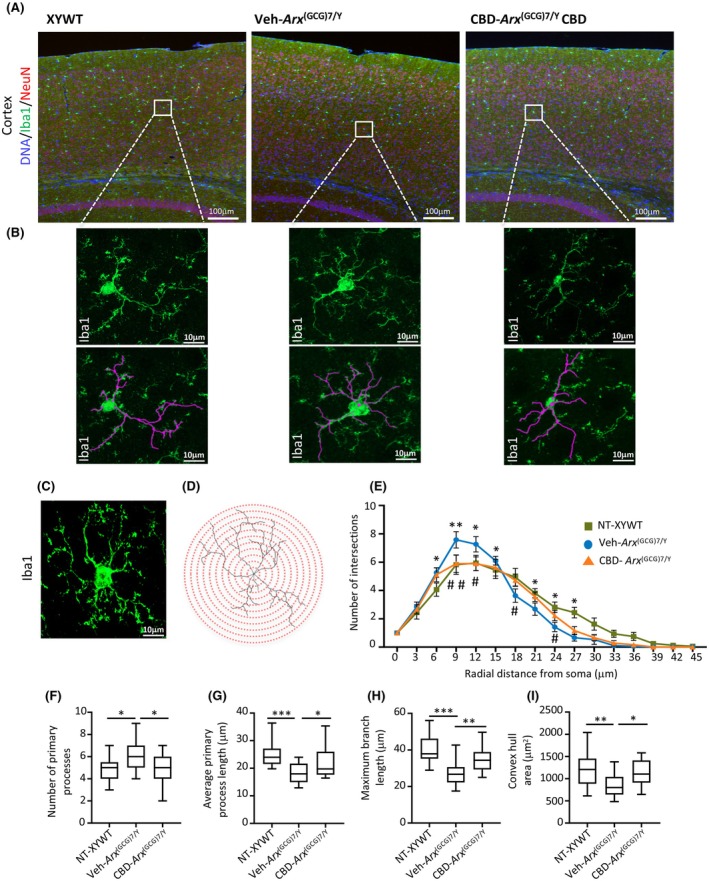
CBD alleviates morphological changes in cortical microglia
Based on the significant beneficial effects of CBD on neuroinflammation markers described above, we analyzed microglial morphology in the cortex of Veh‐treated Arx (GCG)7/Y or of CBD‐treated Arx (GCG)7/Y and age‐matched male controls. Numerous in vivo studies have demonstrated that CBD reduces neuroinflammation by suppressing microglial activation, thereby aiding in the restoration of brain homeostasis.ref. epi18522-bib-0041 Across multiple epileptic conditions, the activation of microglia is marked by observable changes in their morphology and functional properties.ref. epi18522-bib-0042 Building on these considerations, we analyzed the morphological complexity of microglia in the cortex of symptomatic Arx (GCG)7/Y mice, at the end of the Veh and CBD treatments and in untreated controls. For this analysis, we performed anti‐Iba1 immunostaining on cortices of young male mice (Figure 3A,B), followed by Sholl analysis examining microglial morphology and branching patterns (Figure 3C–I, Figure S4A–H). When compared to the XYWT profile, Veh‐treated Arx (GCG)7/Y microglia exhibited a Sholl intersection profile characterized by a significant increase in the number of intersections near the soma, followed by a marked decrease distally from the soma (Figure 3E). Furthermore, Veh‐treated Arx (GCG)7/Y microglia displayed a significant increase in the number of primary processes, along with a notable decrease in the average length of primary processes, maximum branch length, and convex hull area (Figure 3F–I). These morphological changes were significantly reversed by CBD, returning to those observed in the control microglia (Figure 3E–I). No significant differences were observed in the cell density, soma size, number of secondary processes, total process length, or average secondary process length of Iba1+ cells between Arx (GCG)7/Y and control mice, nor did CBD treatment affects these measures (Figure S4I). We therefore conclude that CBD reverses microglial morphology, which is likely influenced by an underlying inflammatory response, and in doing so, may contribute to mitigating the epileptic phenotype in this DEE murine model.
CBD reverses the defective isoform splicing switches of neurexin 1 and neurexin 3
In addition to its effects on neuroinflammation and microglia, CBD may rapidly modulate neuroplasticity by promoting neural network remodeling and potentially restoring normal synaptic function.ref. epi18522-bib-0023, ref. epi18522-bib-0043 Therefore, we investigated whether CBD might counteract the abnormal neuroplasticity features associated with aberrant splicing switches of neurexin 1 (Nrxn1) and neurexin 3 (Nrxn3), which we previously detected in the Arx mutant cortex.ref. epi18522-bib-0012 Splicing of neurexin exon 22 produces two presynaptic NRXN isoforms—AS4(+) and AS4(−)—that differ in their interactions with the canonical postsynaptic neuroligin receptors in excitatory neurons, thereby influencing synaptic remodeling.ref. epi18522-bib-0044 Importantly, Nrxn1 and Nrxn2, along with the Nrxn3, belong to the neurexin gene family, which has been linked to autism spectrum disorder (ASD), and epilepsy.ref. epi18522-bib-0045 Using a semiquantitative PCR assay, we analyzed the levels of AS4(+) and AS4(−) isoforms of Nrxn1, Nrxn2, and Nrxn3 in the cortex isolated from Arx (GCG)7/Y young mice treated by daily intraperitoneal administration with CBD (100 mg/kg, n = 5) or Veh (n = 5) for 7 days, at the same age as the behavioral study (PND35–37; Figure 1A). Consistent with our previous findings in neonatal animals,ref. epi18522-bib-0012 we observed that the cortex of symptomatic untreated and Veh‐treated DEE1 mice exhibits a significant increase in the Nrxn1 AS4(+)/AS4(−) and Nrxn3 AS4(+)/AS4(−) ratios compared to mouse controls, whereas the Nrxn2 AS4(+)/AS4(−) ratio remained unchanged (Figure 4A). Considering the distinct binding affinities of NRXN AS4(+) and AS4(−) isoforms for neuroligin receptors, this altered splicing ratio could compromise the appropriate neurexin–neuroligin interaction at the synaptic cleft, leading to a synaptopathy. Upon treatment, we observed that CBD is capable to rectify the altered neurexin repertoire for both Nrxn1 and Nrxn3 genes with a drastic decrease of AS4(+)/AS4(−) ratio in the cortex isolated from symptomatic Arx (GCG)7/Y mice (Figure 4A).
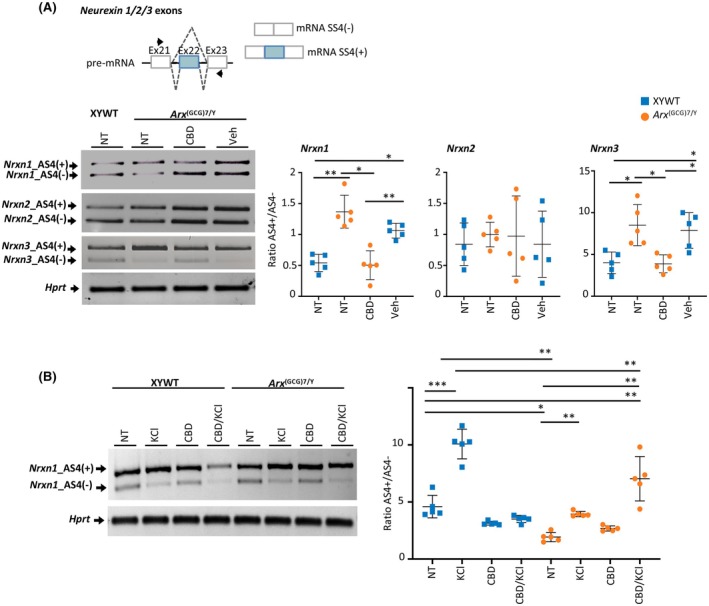
Further confirmation of the beneficial activity of CBD on the neurexin repertoire was found in Arx (GCG)7/Y depolarized cortical neurons. As previously reported, high K+ depolarization of the neuronal membrane alters synaptic plasticity by increasing the Nrxn1 AS4(+)/AS4(−) ratio.ref. epi18522-bib-0012, ref. epi18522-bib-0045, ref. epi18522-bib-0046 By using the cortex of mutant embryos and age‐matched male controls, cortical neuronal cultures were generated (Figure S5A). First, by immunofluorescence with anti‐VGLUT1 and anti‐γ‐aminobutyric acid (GABA), we established that primary cultures of both genotypes had comparable proportions of glutamatergic and GABAergic neurons (respectively 70%:30%; Figure S5B,C). Successively, we carried out single treatment with CBD (10 μmol·L−1) or KCl (51 mmol·L−1), or with CBD and KCl in two sequential steps (CBD/KCl, 10 μmol·L−1 and 51 mmol·L−1, respectively; Figure S5D). Supporting the role of KCl in inducing changes in synaptic plasticity, increased transcript levels of c‐fos were found in KCl‐treated XYWT and KCl‐treated mutant primary neurons compared to the resting (untreated) neurons (Figure S5D). Regarding the Nrxn1 AS4(+)/AS4(−) ratio, a significant increase was observed in KCl‐treated XYWT neurons and CBD/KCl‐treated mutant neurons (Figure 4B). Additionally, we observed that the response in CBD/KCl‐treated mutant neurons closely mirrored KCl‐treated XYWT neurons rather than KCl‐treated Arx (GCG)7/Y neurons, suggesting a synergistic effect of CBD and KCl in promoting a more appropriate synaptic plasticity response, potentially through the restoration of proper neurexin–neuroligin receptor clustering. In summary, these findings underscore the potential of neurexin 1 and neurexin 3 splicing switches as targets for CBD action. Given the involvement of these genes in various forms of ASD and pediatric epilepsy, this represents a significant advancement in exploring the applicability of CBD to other NDDs.
CBD reduces synaptic transmission in cultured primary neurons
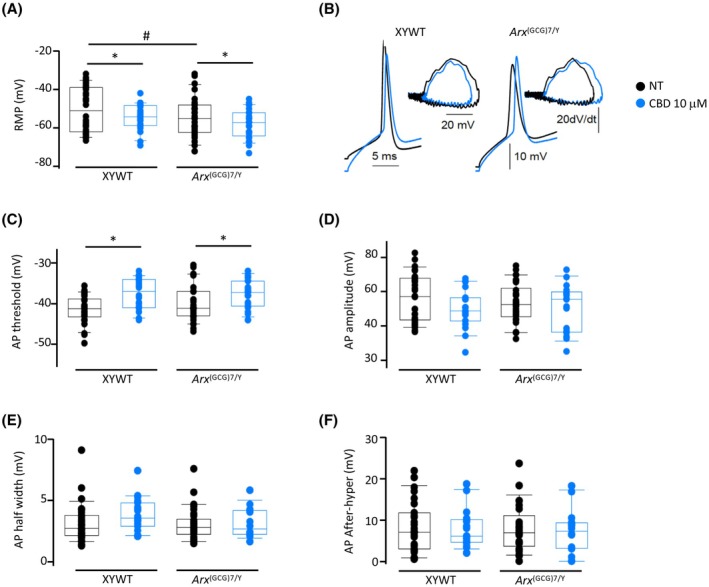
Given the significant beneficial effects of CBD on synaptic plasticity response in Arx (GCG)7/Y primary neurons, we next assessed whether CBD could also exert a favorable effect on the intrinsic excitability of mutant neurons. We then performed in vitro patch‐clamp recordings on cortical neurons from days 13–14 after initiating a primary culture using the cortex of mutant embryos and age‐matched male controls.ref. epi18522-bib-0047 Pretreatments with 10 μmol·L−1 CBD were done on day 12 (for 2 h) in XYWT and Arx (GCG)7/Y neurons (Figure S5E). In 25 XYWT control neurons, we found a mean resting membrane potential (RMP) of −48 ± 2 mV, which decreased to −53 ± 2 mV in 23 Arx (GCG)7/Y cells (p = .042; Figure 5A). In pretreated neuronal cultures, CBD hyperpolarized both XYWT and Arx (GCG)7/Y neurons, lowering the RMP to −55 ± 2 mV and to −58 ± 2 mV, respectively (15 and 15 cells, respectively; two‐way analysis of variance, p = .036; Figure 5A). In the same cells, we elicited depolarizing steps (20 ms) to evoke a single action potential (AP; Figure 5B). Pretreatment with CBD depolarized AP threshold in both XYWT (from −41 ± 1 mV to −37 ± 1 mV) and Arx (GCG)7/Y neurons (from −40 ± 1 mV to −37 ± 1 mV; p = .019; Figure 5B,C). We analyzed other AP kinetic parameters such as amplitude (Figure 5D), half‐width (Figure 5E), and afterhyperpolarization (Figure 5F), and no differences between XYWT, Arx (GCG)7/Y, and CBD treatments were observed.
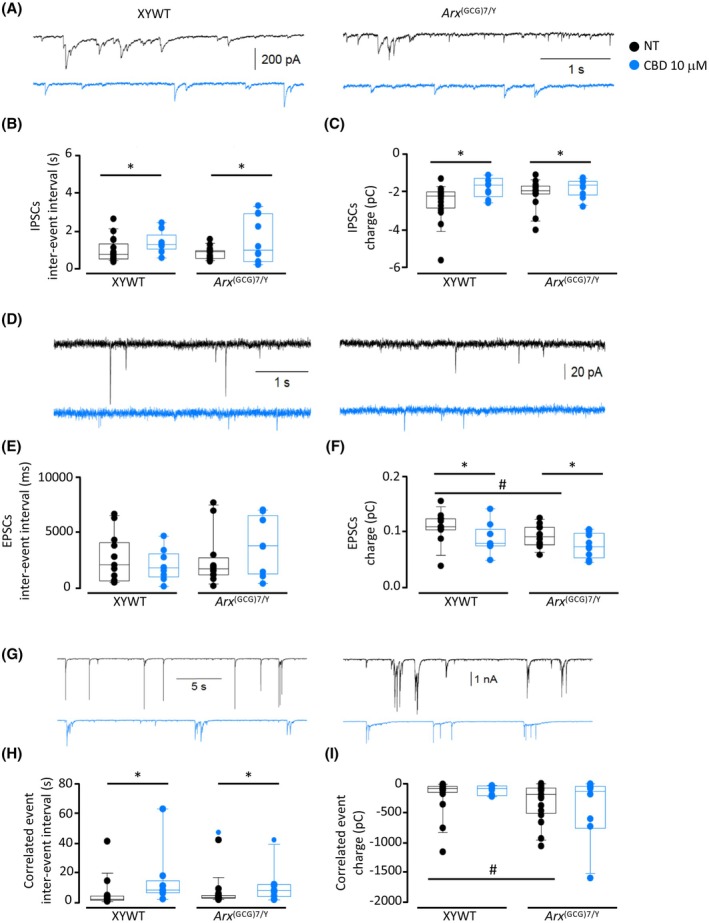
Spontaneous inhibitory postsynaptic currents (IPSCs) were recorded, in control condition or after CBD pretreatment, in XYWT neurons (17 and 9 cells, respectively) and in Arx (GCG)7/Y neurons (17 and 10 cells, respectively; Figure 6A–C). In control condition, no difference was observed in IPSC kinetic parameters measured in both genotypes. Pretreatment with CBD significantly reduced the frequency and the mean charge of IPSCs in both XYWT and Arx (GCG)7/Y neurons (p = .007 and p = .019, respectively; Figure 6B,C). The frequency of glutamatergic excitatory postsynaptic currents (EPSCs) was similar both in XYWT and Arx (GCG)7/Y neurons, whereas the EPSC mean charge was smaller in the mutated neurons (p = .042; Figure 6D,E). Moreover, CBD significantly reduced the EPSC mean charge in both XYWT and Arx (GCG)7/Y neurons (Figure 6F; p = .018). In both XYWT and Arx (GCG)7/Y neurons, we also identified multiple correlated synaptic events (correlated events; Figure 6G–I) due to the summation of several unitary postsynaptic currents. Although the frequency of correlated events was similar for XYWT and Arx (GCG)7/Y neurons (Figure 6H), Arx (GCG)7/Y neurons exhibited a significantly larger mean charge associated with these synaptic events (p = .037; Figure 6I), indicating a higher synchronization of synaptic activity in mutated neurons. Even for correlated events, CBD treatment was able to reduce the frequency in both genotypes (p = .049; Figure 6G,H), confirming the ability of CBD to decrease neuronal signaling in both conditions.
DISCUSSION
We demonstrate for the first time that the administration of CBD improves epileptic phenotype and extends the survival of Arx (GCG)7/Y mice. To our knowledge, CBD is the only drug to date that exerts such effect in this severe DEE mouse model. Remarkably, the Arx (GCG)7/Y replicates one of the most frequently reported polyalanine tract expansion mutations in ARX detected in DEE1/ISSX1 patients, making it a highly suitable system for evaluating promising novel treatments such as CBD.ref. epi18522-bib-0004, ref. epi18522-bib-0006, ref. epi18522-bib-0012, ref. epi18522-bib-0019 Although unraveling the molecular components disrupted by the abnormal transcriptional activity of ARX is highly complex, we showed that CBD has a pleiotropic modifying action targeting simultaneously a range of unanticipated molecular and cellular targets affected in Arx‐DEE1 cortex, such as inflammation, microglia phenotype, synaptic function, and neuronal excitability.
CBD attenuates epileptic phenotype and increases survival
Our overall findings—by using video monitoring and a scoring matrix—show that CBD administration in young symptomatic DEE1 mice (PND35–37 to PND42–44) effectively ameliorates the epileptic phenotype. This was evidenced by a marked reduction in the frequency of SRS and a decrease in seizure burden, with these beneficial effects persisting for up to 7 days after the treatment was discontinued. A long‐term efficacy effect of CBD neuroprotective action has also been observed in preclinical rodent and piglet models of neurodegenerative diseases and newborn hypoxic–ischemic encephalopathy.ref. epi18522-bib-0048, ref. epi18522-bib-0049 Mechanistically, these enduring effects might be caused by modifications in synaptic plasticity leading to lasting changes in global network connectivity that could continue even after the treatment has concluded.
As previously reported, patients with ARX polyalanine elongations have a significantly increased risk of premature death compared to healthy individuals.ref. epi18522-bib-0007 The major causes of death in these patients were not accurately described, but a respiratory illness or sudden unexpected death in epilepsy are reported as frequent death causes in DEEs.ref. epi18522-bib-0050 Remarkably, 30%–38% of Arx (GCG)7/Y mice die before weaning, and the remaining animals succumb within 3 months.ref. epi18522-bib-0017, ref. epi18522-bib-0019 In our study, we showed that CBD significantly extends the lifespan of these mice—a notable finding, especially because other promising drugs like E2 have failed to prolong survival in this model.ref. epi18522-bib-0017 Because there were no differences in body weight between CBD‐treated and Veh‐treated mice, the increased survival is unlikely to be due to enhanced feeding. We therefore speculate that the extension in survival could be primarily driven by the potent anticonvulsant and disease‐modifying effects of CBD. This vital effect was also observed in murine models of DS and Leigh syndrome (LS), both of which exhibit markedly reduced lifespans in patients and animal models.ref. epi18522-bib-0051, ref. epi18522-bib-0052 Importantly, because CBD treatment in Arx (GCG)7/Y mice commenced shortly after seizure onset, our findings strongly indicate that early CBD administration in DEE1/ISSX1 patients could serve as a highly effective therapeutic strategy, both suppressing seizures and reducing premature death risk.
In addition, the CBD dose used in this study is consistent with the effective anticonvulsant doses used in other mouse models of pharmacoresistant epilepsies, including DS, LS, and Angelman syndrome.ref. epi18522-bib-0051, ref. epi18522-bib-0052, ref. epi18522-bib-0053
CBD suppresses inflammatory markers
This study is the first to show that in the cortex of Arx (GCG)7/Y mice, the abnormal expression of a subset of molecules involved in neuroinflammatory response is reversed by CBD treatment. As secondary disease determinants arising from the Arx polyalanine elongation, these molecules could be considered specific targets for the development of targeted therapies for DEE1. We showed that CBD counteracts the transcriptional deficiency of Pparg and suppresses the upregulation of the proinflammatory genes Mmp9 and Ptgs2. Given that Pparg is a positive transcriptional target of ARXref. epi18522-bib-0032 and its protein product acts as a receptor for CBD,ref. epi18522-bib-0023 the stimulation of the Pparg–Mmp9–Ptgs2 transcriptional axis could be one of the functional mechanisms through which CBD impacts on the ARX transcriptional program. Furthermore, we found that CBD reduces the elevated levels of the brain inflammatory marker TRPV1, along with its phosphorylated form, in the cortex of Arx (GCG)7/Y animals. Because TRPV1 levels increase in response to inflammatory signals,ref. epi18522-bib-0054 the excessive TRPV1 levels detected in the mutant cortex further support the notion that a proinflammatory state may contribute to the physiopathology of Arx (GCG)7/Y mice. However, the precise mechanism by which CBD reduces TRPV1—and consequently its phosphorylated form—remains unclear. We speculate that CBD may act on TRPV1 through a double mechanism (1) by downregulating TRPV1 protein via the Pparg–Ptgs2 transcriptional axis contributing to offsetting the inflammation state and (2) by the desensitization of pTRPV1 channels—as observed in other epileptic seizure systemsref. epi18522-bib-0024, ref. epi18522-bib-0055—thus dampening the abnormal neuronal excitability. Notably, we also demonstrated that anti‐inflammatory effects of CBD extend to other key inflammasome mediators that are overexpressed in the Arx (GCG)7/Y cortex. We showed that CBD significantly reduces the transcript levels of the proinflammatory immune biomarkers IL‐12, CD68, CCL2, and IRF3, found previously upregulated in rodent models of induced epilepsy and in pediatric patients with encephalopathy.ref. epi18522-bib-0037, ref. epi18522-bib-0056 The finding that these CBD‐responsive molecules elicit an immune‐mediated inflammatory response in both murine models and clinical epilepsy cases further confirms that the mutant mouse cortex is distinctly marked by a proinflammatory state. The lack of effects in suppressing the activation of the proinflammatory genes Tlr4, Irak4, and Myd88—which are key innate immune molecules implicated in epileptogenesisref. epi18522-bib-0056—highlights a selective anti‐inflammatory action of CBD in this DEE1 model. This discovery points to a previously underestimated role of inflammation in the Arx (GCG)7/Y mouse pathogenesis and underscores the potential of CBD anti‐inflammatory activity in preventing or reducing the severity of DEE1 epilepsy. Future omics studies will help clarify how CBD affects the transcriptional program driven by Arx polyalanine mutations, leading to the identification of primary and secondary disease determinants that are responsive to CBD.
CBD suppresses microglia morphology changes
Our findings reveal that as a corollary of its anti‐inflammatory activity, CBD offsets a defective morphological profile of microglia cells including shortened primary processes, increased intersections, and an overall reduction in branch length and convex hull area. Microglia are highly dynamic cells that continuously extend and retract their processes to survey the tissue microenvironment. As observed in in vivo and in vitro epilepsy models, microglial cells exposed to inflammatory proepileptogenic stimuli become activated, adopting a more amoeboid shape and displaying features similar to those seen in the Arx (GCG)7/Y cortex.ref. epi18522-bib-0041, ref. epi18522-bib-0042 CBD has been shown to reduce microglial activation and lower proinflammatory marker levels in various neurological disease systems, including seizure models.ref. epi18522-bib-0041 On the other hand, the CBD‐responsive targets found deregulated in the mutant cortex—PPARγ, TRPV1, IL‐12, CD68, CCL2, and IRF3—are all mechanistically connected to microglial activation.ref. epi18522-bib-0023, ref. epi18522-bib-0054, ref. epi18522-bib-0055 We therefore speculate that the spontaneous epileptic activity seen in Arx‐DEE1 mice could lead to changes in the microglial state, and that CBD helps to restore microglial morphology by exerting its anti‐inflammatory effects. However, due to the complexity and variability of microglial phenotypes across different functional states, and because the effects on cellular motility are often model‐specific and not easily generalized, the understanding of the precise role of microglia in DEE1 mice is still in its early stages.
CBD suppresses neurexin‐splicing switch abnormalities and changes innate firing properties
Beyond its anti‐inflammatory effects, we uncovered that CBD exerts a neuroprotective activity in Arx (GCG)7/Y cortex and primary neurons. CBD corrects the aberrant splicing switches of neurexin 1 and 3 that we previously identified in the Arx mutant neocortex, contributing to the restoration of the molecular composition of the presynaptic membrane and the proper binding of neuroligins to the postsynaptic membrane as well.ref. epi18522-bib-0012 Given that the neuroligin–neurexin complex has been shown to selectively regulate interactions at both glutamatergic and GABAergic synapses,ref. epi18522-bib-0057, ref. epi18522-bib-0058 we propose that CBD may potentially restore the disrupted balance between excitatory and inhibitory neurotransmission. Consistently with these data, we found a synergistic activity of CBD with KCl at ameliorating Nrxn1 AS4(+)/AS4(−) ratio and thus the depolarization response in primary mutant neurons.
Emerging evidence suggests that CBD may influence splicing mechanisms, although the exact mechanisms remain under investigation. CBD has been shown to directly bind to EFTUD2, a component of the spliceosome complex that processes precursor mRNAs to produce mature mRNAs, indicating a potential role in spliceosome regulation.ref. epi18522-bib-0059 Regarding the direct interactions between CBD and neurexins, or with neuroligin–neurexin complex, no data have been reported to date. This is a highly intriguing field of research due to the association of neurexin genes with ASDref. epi18522-bib-0045 and the potential therapeutic applications of CBD in these disorders.ref. epi18522-bib-0060
Remarkably, in a previous study, an unexpected role for neurexins in the endocannabinoid‐dependent regulation of neural circuits was reported. Presynaptic neurexin splice variants containing the SS4 segment modulate excitatory synaptic strength by regulating the postsynaptic biosynthesis of the endocannabinoid 2‐arachidonoylglycerol, indicating a link between the neurexin family and endocannabinoid signaling.ref. epi18522-bib-0061 Given the complexity of the overlapping pathways regulated by ARX, including alternative splicing control and synaptic plasticity,ref. epi18522-bib-0012 further research is needed to understand how CBD affects alternative splicing switches on a global scale and to pinpoint its precise effects on neurexin genes. Consistent with previous studies,ref. epi18522-bib-0062 we further showed that, in both cultured control and mutant primary neurons, CBD decreases excitability by inducing hyperpolarization and increasing the action potential threshold. This neuroprotective activity of CBD mirrors its ability to influence the intrinsic properties of depolarized neurons, thus dampening hyperexcitability and thereby counteracting recurrent seizures.ref. epi18522-bib-0060 In line with this, we showed that CBD reduces the frequency, the mean charge of IPSCs, and the mean charge of EPSCs in both genotypes. The question of how CBD dampens hyperexcitability in these primary neurons may be answered by the simultaneous interactions of CBD with multiple targets, some of them analyzed in this study.
In summary, this study adds Arx (GCG)7/Y to the range of epilepsy murine models in which CBD treatment is effective. Given the rarity of DEE1 caused by polyalanine elongation mutations in ARX, our findings—obtained using a mouse model that replicates key aspects of the epileptic phenotype—encourage further research that may ultimately lead to clinical assessment of CBD in DEE1 for ameliorating seizure control and survival. This could lead to the development of novel, targeted treatments that address both the genetic and inflammatory aspects of this rare form of DEE exploring the applicability in other ARX‐related disorders. Further studies are needed to better understand how CBD affects ARX‐driven disease mechanisms and its ability to regulate microglia–neuron interactions and neuroligin–neurexin signaling.
AUTHOR CONTRIBUTIONS
Lucia Verrillo and Denise Drongitis contributed to animal behavior experiments and cellular studies and data analysis. Loredana Poeta and Adriano Barra contributed to data acquisition of animal experiments. Katiuscia Martinello and Sergio Fucile contributed to data acquisition and analysis of electrophysiology recording. Fabio Arturo Iannotti and Vincenzo Di Marzo contributed to data acquisition and analysis of CBD treatments. Gaetano Terrone contributed to data analysis of epilepsy behavior. Fabio Arturo Iannotti, Vincenzo Di Marzo, and Maria Giuseppina Miano contributed to the experimental design. Maria Giuseppina Miano contributed to conceptualization, supervision, and writing—review and editing. All authors critically revised the article, commented on the drafts of the manuscript, and approved the final version.
CONFLICT OF INTEREST STATEMENT
G.T. has received speaker honoraria from Jazz Pharmaceuticals, UCB, and Neuraxpharm. None of the other authors has any conflict of interest to disclose. We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Supplementary Materials
References
- Deciphering the concepts behind “epileptic encephalopathy” and “developmental and epileptic encephalopathy”.. Eur J Paediatr Neurol., 2020. [PubMed]
- The genetic landscape of the epileptic encephalopathies of infancy and childhood.. Lancet Neurol., 2016. [PubMed]
- MRX87 family with aristaless X dup24bp mutation and implication for polyAlanine expansions.. BMC Med Genet., 2007. [PubMed]
- A regulatory path associated with X‐linked intellectual disability and epilepsy links KDM5C to the polyalanine expansions in ARX.. Am J Hum Genet., 2013. [PubMed]
- The role of ARX in cortical development.. Eur J Neurosci., 2006. [PubMed]
- ARX: a gene for all seasons.. Curr Opin Genet Dev., 2006. [PubMed]
- A longer polyalanine expansion mutation in the ARX gene causes early infantile epileptic encephalopathy with suppression‐burst pattern (Ohtahara syndrome).. Am J Hum Genet., 2007. [PubMed]
- Long‐term mortality of patients with west syndrome.. Epilepsia Open., 2016. [PubMed]
- Histone demethylase KDM5C is a SAHA‐sensitive central hub at the crossroads of transcriptional axes involved in multiple neurodevelopmental disorders.. Hum Mol Genet., 2019. [PubMed]
- DNA hypermethylation and unstable repeat diseases: a paradigm of transcriptional silencing to decipher the basis of pathogenic mechanisms.. Genes (Basel)., 2020. [PubMed]
- Embryonic forebrain transcriptome of mice with polyalanine expansion mutations in the ARX homeobox gene.. Hum Mol Genet., 2016. [PubMed]
- Deregulation of microtubule organization and RNA metabolism in Arx models for lissencephaly and developmental epileptic encephalopathy.. Hum Mol Genet., 2022. [PubMed]
- A novel ARX phenotype: rapid neurodegeneration with Ohtahara syndrome and a dyskinetic movement disorder.. Dev Med Child Neurol., 2010. [PubMed]
- A novel mutation in the aristaless domain of the ARX gene leads to Ohtahara syndrome, global developmental delay, and ambiguous genitalia in males and neuropsychiatric disorders in females.. Epilepsia., 2011. [PubMed]
- A triplet repeat expansion genetic mouse model of infantile spasms syndrome, Arx(GCG)10+7, with interneuronopathy, spasms in infancy, persistent seizures, and adult cognitive and behavioral impairment.. J Neurosci., 2009. [PubMed]
- Neonatal estradiol stimulation prevents epilepsy in Arx model of X‐linked infantile spasms syndrome.. Sci Transl Med., 2014
- Early 17β‐estradiol treatment reduces seizures but not abnormal behaviour in mice with expanded polyalanine tracts in the aristaless related homeobox gene (ARX).. Neurobiol Dis., 2021. [PubMed]
- Interneuron, interrupted: molecular pathogenesis of ARX mutations and X‐linked infantile spasms.. Curr Opin Neurobiol., 2012. [PubMed]
- Three human ARX mutations cause the lissencephaly‐like and mental retardation with epilepsy‐like pleiotropic phenotypes in mice.. Hum Mol Genet., 2009. [PubMed]
- Trial of cannabidiol for drug‐resistant seizures in the Dravet syndrome.. N Engl J Med., 2017. [PubMed]
- Effect of cannabidiol on drop seizures in the Lennox‐Gastaut syndrome.. N Engl J Med., 2018. [PubMed]
- Add‐on cannabidiol treatment for drug‐resistant seizures in tuberous sclerosis complex: a placebo‐controlled randomized clinical trial.. JAMA Neurol., 2021. [PubMed]
- Cannabinoids and the expanded endocannabinoid system in neurological disorders.. Nat Rev Neurol., 2020. [PubMed]
- Non‐psychotropic plant cannabinoids, cannabidivarin (CBDV) and cannabidiol (CBD) activate and desensitize transient receptor potential vanilloid 1 (TRPV1) channels in vitro: potential for the treatment of neuronal hyperexcitability.. ACS Chem Nerosci., 2014
- Purified cannabidiol as add‐on therapy in children with treatment‐resistant infantile epileptic spasms syndrome.. Seizure., 2024. [PubMed]
- Therapeutic and clinical foundations of cannabidiol therapy for difficult‐to‐treat seizures in children and adults with refractory epilepsies.. Exp Neurol., 2023. [PubMed]
- Retigabine, a Kv7.2/Kv7.3‐channel opener, attenuates drug‐induced seizures in Knock‐in mice harboring Kcnq2 mutations.. PLoS One., 2016. [PubMed]
- Cannabidiol reduces seizures and associated behavioral comorbidities in a range of animal seizure and epilepsy models.. Epilepsia., 2019. [PubMed]
- Extensive phenotyping of two ARX polyalanine expansion mutation mouse models that span clinical spectrum of intellectual disability and epilepsy.. Neurobiol Dis., 2017. [PubMed]
- Antiseizure effects of cannabidiol leading to increased peroxisome proliferator‐activated receptor gamma levels in the hippocampal CA3 subfield of epileptic rats.. Pharmaceuticals (Basel)., 2022. [PubMed]
- Anticonvulsive properties of cannabidiol in a model of generalized seizure are transient receptor potential vanilloid 1 dependent.. Cannabis Cannabinoid Res., 2020. [PubMed]
- High‐throughput analysis of promoter occupancy reveals new targets for Arx, a gene mutated in mental retardation and interneuronopathies.. PLoS One., 2011. [PubMed]
- Structural organization of mouse peroxisome proliferator‐activated receptor gamma (mPPAR gamma) gene: alternative promoter use and different splicing yield two mPPAR gamma isoforms.. Proc Natl Acad Sci U S A., 1995. [PubMed]
- Roles of peroxisome proliferator‐activated receptor gamma on brain and peripheral inflammation.. Cell Mol Neurobiol., 2018. [PubMed]
- SOCS3 transactivation by PPARγ prevents IL‐17‐driven cancer growth.. Cancer Res., 2013. [PubMed]
- TRPV1 feed‐forward sensitisation depends on COX2 upregulation in primary sensory neurons.. Sci Rep., 2021. [PubMed]
- The chemokine CCL2 mediates the seizure‐enhancing effects of systemic inflammation.. J Neurosci., 2016. [PubMed]
- Increased CCL2, CCL3, CCL5, and IL‐1β cytokine concentration in piriform cortex, hippocampus, and neocortex after pilocarpine‐induced seizures.. J Neuroinflammation., 2015. [PubMed]
- Histone deacetylase inhibitor SAHA attenuates post‐seizure hippocampal microglia TLR4/MYD88 signaling and inhibits TLR4 gene expression via histone acetylation.. BMC Neurosci., 2016. [PubMed]
- Toll‐like receptor 4 and high‐mobility group box‐1 are involved in ictogenesis and can be targeted to reduce seizures.. Nat Med., 2010. [PubMed]
- Cannabidiol inhibits microglia activation and mitigates neuronal damage induced by kainate in an in‐vitro seizure model.. Neurobiol Dis., 2022. [PubMed]
- The role of inflammation in epilepsy.. Nat Rev Neurol., 2011. [PubMed]
- Role of the endocannabinoid system in fragile X syndrome: potential mechanisms for benefit from cannabidiol treatment.. J Neurodev Disord., 2023. [PubMed]
- Alternative splicing of presynaptic neurexins differentially controls postsynaptic NMDA and AMPA receptor responses.. Neuron., 2019. [PubMed]
- Neurexins in autism and schizophrenia‐a review of patient mutations, mouse models and potential future directions.. Mol Psychiatry., 2021. [PubMed]
- Activity‐induced histone modifications govern Neurexin‐1 mRNA splicing and memory preservation.. Nat Neurosci., 2017. [PubMed]
- Loss of constitutive functional γ‐aminobutyric acid type A‐B receptor crosstalk in layer 5 pyramidal neurons of human epileptic temporal cortex.. Epilepsia., 2018. [PubMed]
- From Cannabis sativa to cannabidiol: promising therapeutic candidate for the treatment of neurodegenerative diseases.. Front Pharmacol., 2020. [PubMed]
- Mechanisms of cannabidiol neuroprotection in hypoxic‐ischemic newborn pigs: role of 5HT(1A) and CB2 receptors.. Neuropharmacology., 2013. [PubMed]
- Mortality in infantile spasms: a hospital‐based study.. Epilepsia., 2020. [PubMed]
- Cannabidiol attenuates seizures and social deficits in a mouse model of Dravet syndrome.. Proc Natl Acad Sci U S A., 2017. [PubMed]
- Cannabidiol ameliorates mitochondrial disease via PPARγ activation in preclinical models.. Nat Commun., 2024. [PubMed]
- Cannabidiol attenuates seizures and EEG abnormalities in Angelman syndrome model mice.. J Clin Invest., 2019. [PubMed]
- TRPV1 channels are critical brain inflammation detectors and neuropathic pain biomarkers in mice.. Nat Commun., 2017. [PubMed]
- TRPV1 channel in the pathophysiology of epilepsy and its potential as a molecular target for the development of new antiseizure drug candidates.. Prog Neurobiol., 2024. [PubMed]
- Epilepsy, seizures, and inflammation: role of the C‐C motif ligand 2 chemokine.. DNA Cell Biol., 2016. [PubMed]
- Alternative splicing controls selective trans‐synaptic interactions of the neuroligin‐neurexin complex.. Neuron., 2006. [PubMed]
- Neurexins: molecular codes for shaping neuronal synapses.. Nat Rev Neurosci., 2021. [PubMed]
- A label free chemoproteomic‐based platform to disclose cannabidiol molecular mechanism of action on chronic myelogenous leukemia cancer cells.. Heliyon., 2024. [PubMed]
- Cannabinoids for people with ASD: a systematic review of published and ongoing studies.. Brain Sci., 2020. [PubMed]
- Β‐Neurexins control neural circuits by regulating synaptic endocannabinoid signaling.. Cell., 2015. [PubMed]
- Cannabidiol modulates excitatory‐inhibitory ratio to counter hippocampal hyperactivity.. Neuron., 2023. [PubMed]
