Multicomponent Interventions for Adults With Cancer Cachexia: A Systematic Review
Abstract
Background:
Cancer cachexia has substantial impacts on people’s quality of life. There is no current gold standard treatment, but the complex pathophysiology of cachexia suggests that a multitargeted and individualised treatment approach is needed. We aimed to evaluate the extent to which multicomponent interventions have targeted the key features of cachexia and been tailored to individuals, and differential effects on quality of life.
Methods:
We conducted a systematic review of multicomponent interventions for adults with cancer cachexia. We searched four databases, two clinical trial registers and MedRxiv on 20 June 2024. Intervention components were classified by intervention category (nutritional, exercise/physical activity, pharmacological and psychosocial), cachexia feature(s) targeted (reduced energy intake, altered metabolism, involuntary weight loss and decline in physical function) and level of tailoring. Within‐arm standardised mean changes in quality of life over time, as well as standardised mean differences between study arms, were calculated.
Results:
Sixty‐two multicomponent interventions were included, of which two combined components from all four intervention categories, and nine targeted all four key features of cachexia. Eighteen multicomponent interventions were fully tailored and 30 were partly tailored to individuals. Within‐arm standardised mean changes in quality of life were calculated for thirteen studies; all had a high risk of bias or raised concerns. In eleven studies, quality of life scores improved following the intervention, whereas in two studies they declined. Standardised mean differences between study arms were calculated for four studies; in three, the intervention arm showed a greater improvement in quality of life scores than the usual care arm. Amongst these data, there was no indication that the number of cachexia features targeted, or the extent of tailoring, was associated with a greater improvement in quality of life scores; however, the heterogeneity prevented us from concluding on our hypothesis.
Conclusions:
This review mapped out in detail the combinations of intervention categories used, the key features of cachexia targeted, and the extent of tailoring across multicomponent interventions for adults with cancer cachexia. Only a small proportion of the multicomponent interventions targeted all four key features of cachexia, but most were either partly or fully tailored to individuals. Despite sixty‐two multicomponent interventions being investigated, only four studies compared these to usual care and reported quality of life outcomes. High risk of bias, low sample sizes and variable outcome data remain challenges to the interpretability of results in this field.
Article type: Review Article
Keywords: cachexia, cancer, multimodal, quality of life, treatment, weight loss
Affiliations: Cicely Saunders Institute of Palliative Care, Policy & Rehabilitation, Florence Nightingale Faculty of Nursing, Midwifery & Palliative Care King’s College London London UK; Edinburgh Palliative Care and Supportive Care Group The University of Edinburgh Edinburgh UK; St Columba’s Hospice Edinburgh UK
License: © 2025 The Author(s). Journal of Cachexia, Sarcopenia and Muscle published by Wiley Periodicals LLC. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/jcsm.13716 | PubMed: 40012451 | PMC: PMC11865637
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.6 MB)
Introduction
Cancer cachexia is ‘a multi‐factorial syndrome characterised by an ongoing loss of skeletal muscle mass (with or without loss of fat mass) that cannot be fully reversed by conventional nutritional support and leads to progressive functional impairment’ [ref. 1]. Key pathophysiological features of the syndrome include a negative protein and energy balance, which results from reduced food intake in combination with abnormal metabolism [ref. 1]. Cachexia is generally progressive, especially if the underlying disease cannot be cured [ref. 2]. The prevalence of cachexia amongst people with cancer varies from around 20%–80%, depending on the primary tumour site, stage and other patient factors [ref. 3, ref. 4]. Cancer cachexia has physical, psychological, social and existential consequences for patients and carers [ref. 5]. It impacts the quality of life, with eating‐related issues and worries about physical decline contributing to negative body image, altered sense of identity, feelings of loss of control and negative emotions [ref. 6].
Although there is no current gold standard treatment for cancer cachexia, trials of interventions over the past decades have targeted different aspects of the syndrome, such as inflammation [ref. 7], appetite [ref. 8], muscle mass and function [ref. 9] and psychosocial wellbeing [ref. 10], often in isolation. The complex pathophysiology of cancer cachexia suggests the need for a multitargeted intervention. Pharmaceutical trials have developed and tested various drugs with the aim of alleviating cachexia with one single pill, some of which have shown positive effects on certain outcomes like body weight [ref. 11]. However, few drugs have shown positive effects on functional outcomes like handgrip strength or on patient‐reported outcomes like quality of life. Some trials have tested combinations of pharmacological agents, and although data on this are limited, there is some indication that these may be more effective than single drugs, highlighting the need for a multitargeted approach [ref. 11].
Therefore, the current dominant view is that a combination of interventions is required to alleviate cachexia [ref. 12, ref. 13], a concept that was pioneered by Fearon, who proposed combining exercise, nutrition support and anti‐inflammatory drugs [ref. 14]. Since then, many trials have embraced a multimodal approach, investigating combinations of pharmacological, nutritional, exercise and/or psychosocial intervention components [ref. 15]. However, it is not known how many of these multicomponent interventions have targeted all the key features of cachexia or whether interventions that target more key features of cachexia are more effective at improving patient‐centred outcomes including quality of life. Indeed, the diversity of multicomponent interventions brings the challenge of pooling study results in meta‐analyses to evaluate which combinations of intervention components are effective at improving patient outcomes.
Furthermore, interview studies have highlighted that patients and carers value interventions that are individualised and flexible [ref. 16, ref. 17, ref. 18, ref. 19, ref. 20]. Although some trials have incorporated this approach (e.g., individualised exercise or nutritional counselling [ref. 20, ref. 21]), usually all intervention components are provided, in one way or another, to all participants. In contrast, in nonexperimental settings like clinical services, it is more common for intervention components to only be provided if there is an indicated need (e.g., prescriptions and recommendations for present symptoms, e.g., [ref. 22]). This approach aligns with the progressive nature of cachexia; individuals with refractory cachexia will likely benefit from different intervention components than individuals with pre‐cachexia [ref. 23]. It also supports the large variability in cachexia presentation, where appetite may be the primary concern for one person, whereas muscle weakness may be the primary concern for another.
Systematic reviews of cancer cachexia interventions have often included only randomised trials with published results. Usually, nonrandomised studies of interventions (NRSIs), such as single‐arm feasibility trials and studies evaluating the effects of clinical services, and ongoing studies that have not yet published results, are excluded. However, both NRSIs and ongoing studies can contribute valuable information about the types of interventions that have been and are currently being investigated for people with cancer cachexia. In addition to this, although methodological limitations and potential biases must be considered, such as confounding [ref. 24], NRSIs with results can provide data on important patient‐centred outcomes like quality of life. If positive, results from NRSIs can encourage the design of randomised trials to test those interventions [ref. 25], so should not be overlooked.
We therefore synthesised studies of multicomponent interventions for adults with cancer cachexia. Our aims were to evaluate the extent to which multicomponent interventions have targeted the key features of cachexia and been tailored to individuals and whether this is associated with differential effects on quality of life outcomes.
Methods
Design
We conducted a systematic review of studies of multicomponent interventions for people with cancer cachexia. The protocol was published on PROSPERO on 25 July 2023 (CRD42023412551). We have reported this review according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis (PRISMA) criteria.
Eligibility Criteria
Population
We considered studies with evidence that (a) all participants were adults (aged ≥ 18 years) and had a diagnosis of cancer at baseline and (b) ≥ 75% of participants had or were at risk of cachexia at baseline. We applied the international consensus definition of cancer cachexia: weight loss of > 5% in the previous 6 months or BMI < 20 kg/m2 and ongoing weight loss of > 2% [ref. 1]. We considered participants with advanced (Stage 3 or 4) lung, upper gastrointestinal or head and neck cancers to be at risk of cachexia [ref. 3, ref. 26, ref. 27]. We excluded studies if any participant was < 18 years at baseline, if any participant did not have a cancer diagnosis at baseline or if there was no evidence that ≥ 75% of participants had or were at risk of cachexia at baseline based on our criteria.
Interventions
We considered studies that investigated a multicomponent intervention, which we defined as an intervention that included at least two components (that could be individual interventions), either concurrently or sequentially. We excluded studies which investigated single‐component interventions. We did not consider anticancer treatments (e.g., chemotherapy) as intervention components; thus, if a study investigated a combination of an anticancer treatment and one additional intervention component, it was excluded. Examples of eligible multicomponent interventions included two or more separate drugs (e.g., megestrol acetate and celecoxib), one or more drugs plus a nutritional supplement (e.g., ibuprofen and fish oil), two or more nutritional interventions (e.g., fish oil and dietary counselling) or two or more nonpharmacological interventions (e.g., aerobic exercise and psychosocial support). We did not set any restrictions for comparators or outcome measures reported by studies.
Study Design
We considered primary research studies that were randomised studies or NRSIs of interventions, in either an experimental (e.g., clinical trial) or nonexperimental setting (e.g., evaluation of a clinical service). We excluded secondary research studies (e.g., reviews) and records that did not present original data (e.g., expert opinions, letters to the editor and commentaries). We also excluded studies with fewer than five participants enrolled at baseline.
Record Type
We considered published journal papers, pre‐prints and clinical trial register records. We considered ongoing studies without published results, if adequate information about the intervention was provided (e.g., if there was a published protocol or clinical trial register record available with a description of the intervention). We did not set any restrictions for the publication year. We considered records written in English or records written in other languages if they could be successfully translated by Google Translate.
Information Sources and Search Strategy
We conducted a comprehensive search strategy, which included databases, clinical trial registers and pre‐prints. We searched four databases (MEDLINE via Ovid, EMBASE via Ovid, Cochrane Central Register of Controlled Trials and CINAHL via EBSCO) from inception to 26 July 2023 and then conducted an updated search on 20 June 2024. In line with Cochrane [ref. 28] and BMJ guidance [ref. 29], we searched two clinical trial registers (ClinicalTrials.gov and World Health Organisation International Clinical Trials Registry Platform [WHO ICTRP]) on 26 July 2023 and again on 20 June 2024. We also searched MedRxiv on 26 July 2023 and again on 20 June 2024. Full search strategies can be found in Supporting Information S1.
Study Records
Data Management
We imported all records from the database searches and MedRxiv into a systematic review manager (Rayyan) for screening. We imported records from clinical trial registers into an Excel spreadsheet for screening.
Selection Process
We used Rayyan’s automatic duplicate detection function to identify possible duplicate records amongst database records, and then, the primary reviewer (M.B.) manually resolved each one. The primary reviewer (M.B.) screened each record based on its title and abstract, and a second reviewer (A.M.) independently screened 10% of the records to check for consistency in decisions. The primary reviewer (M.B.) and at least one other reviewer (A.M./C.P./J.B.) independently screened each full text. In the case of conflict between the reviewers, the article was discussed between the primary reviewer (M.B.) and another independent reviewer (M.M.). To achieve consistency in the selection of articles, the final decision was ultimately made by the primary reviewer (M.B.).
Data Collection
The primary reviewer (M.B.) extracted data from all included records, on report characteristics (e.g., publication type and year), study design, population characteristics (e.g., cancer site and stage, weight loss and cachexia status), interventions provided to each study arm, sample size and attrition. A second reviewer (C.P./A.M./R.D.) then checked all extracted data.
For each multicomponent intervention, the primary reviewer (M.B.) listed each individual component individually, alongside its intervention category (pharmacological, nutritional, exercise/physical activity or psychosocial), type (e.g., appetite stimulant, exercise and nutrition support), subtype where applicable (e.g., anamorelin, resistance training and enteral nutrition), key feature(s) of cachexia targeted and level of tailoring.
Two reviewers (M.B. and C.P./A.M./R.D.) independently extracted measures of quality of life, in addition to any stated intention to measure quality of life and outcome data (preferentially as mean and standard deviation or as reported by the study).
Risk of Bias in Individual Studies
Two reviewers (M.B. and C.P./A.M.) independently assessed the risk of bias for each study that provided data for the quality of life analysis. We used Cochrane tools for these assessments: For randomised trials, we used the revised tool for risk of bias in randomised trials (RoB 2), and for NRSIs, we used the Risk Of Bias in Non‐randomised Studies—of Interventions (ROBINS‐I). We were interested in the effect of assignment on the intervention (intention‐to‐treat analysis).
Data Synthesis and Analysis
Summary Statistics
Firstly, we categorised studies as either randomised trials or NRSIs. Subsequently, we subcategorised NRSI study designs based on guidance from Cochrane [ref. 25] and Reeves and colleagues [ref. 30].
We calculated the median (range) sample size at baseline, the median (range) average age of participants and the median (range) average percentage weight loss of participants at baseline from all studies that reported these data.
Additionally, we calculated how many participants had lung, gastrointestinal and head and neck cancers and how many participants had Stage 3 or 4 cancer at baseline. These analyses included all participants from all studies that reported these data.
Finally, we calculated how many participants had cachexia at baseline. If the inclusion criteria of the study indicated that all enrolled participants had cachexia; then, we assumed that the number of participants with cachexia was equal to the sample size. If the study reported the number of participants with cachexia at baseline, then we used this number.
Intervention Analysis
We used cancer cachexia guidelines from the European Society of Medical Oncology (ESMO [ref. 12]) and the American Society of Clinical Oncology (ASCO [ref. 31]), in addition to the international consensus definition of cancer cachexia [ref. 1], to define four key features of cancer cachexia for use in this review. These key features were as follows: (1) reduced energy intake (including causal factors like appetite loss, food aversion and nutrition impact symptoms), (2) altered metabolism (including systemic inflammation), (3) involuntary weight loss (including loss of lean and/or fat mass) and (4) decline in physical function (including causal factors like fatigue and loss of muscle strength).
We classified each individual intervention component into one of four ‘intervention categories’: nutritional, exercise/physical activity, pharmacological or psychosocial. We also classified each component based on which of the four key features of cancer cachexia it targeted. To illustrate intervention categories and targets, we created Sunburst diagrams using the Plotly package in Python. To visualise which intervention categories were combined and which key features of cachexia were targeted together, we created UpSet plots [ref. 32].
To assess the level of intervention tailoring, we first classified each component based on its provision; a component had ‘targeted provision’ if there was evidence that it was only given to study participants who needed it; otherwise, it had ‘total provision’. We also classified each component based on its delivery; a component was ‘individualised’ if there was evidence that it was modified for each participant; otherwise, it was ‘standardised’. Subsequently, we classified components with evidence of targeted provision and/or individualised delivery as ‘tailored’ and components with total provision and standardised delivery as ‘not tailored’. Then, if a multicomponent intervention consisted of some components that were tailored and some components that were not tailored, we classified it as ‘partly tailored’. If all the components in a multicomponent intervention were tailored, we classified it as ‘fully tailored’; if none of the components were tailored, we classified it as ‘not tailored’.
Effects on Quality of Life
We extracted data from all studies that reported any validated measure of overall quality of life. We did not consider measures of symptom burden (e.g., FAACT anorexia–cachexia subscale) to be measures of quality of life. We developed a hierarchy to choose which measure of quality of life to include in the analysis, in the case that a study reported data for more than one measure of quality of life. The hierarchy placed composite/comprehensive measures (e.g., EORTC QLQ‐C30 overall score) higher than subscales or single‐item measures (e.g., EORTC QLQ‐C30 global health status subscale and ESAS quality of life item) and placed disease/condition‐specific measures higher than nonspecific measures.
We extracted follow‐up data from the time point which was closest to the end of the intervention. If there was no defined end to the intervention (e.g., in an ongoing clinical service), we extracted data from the final follow‐up time point. If there was a defined end to the intervention, but follow‐up data were not reported at the end of the intervention, we extracted data from the final follow‐up time point, even if this was part‐way through the intervention.
To explore the effect of multicomponent interventions on quality of life, we were interested in the mean change in quality of life score over the intervention period. If the mean change in quality of life score was reported in the study, we used these data. If the mean change in quality of life score was not reported in the study, but the mean baseline quality of life score and the mean postintervention quality of life score were reported, then we calculated the mean change in quality of life score, based on the Cochrane guidance [ref. 33]. We used a standardised version of the mean change in quality of life score (from now on referred to as the standardised mean change [SMC]) so that we could analyse data from different quality of life measures together. Additional information on the calculations we conducted can be found in Supporting Information S2.
To compare the effect of multicomponent interventions on quality of life versus usual care, we were interested in the difference in the mean change in quality of life scores (from now on referred to as the mean difference) between the study arms. If the mean difference was reported in the study, we used these data; if it was not reported in the study, we calculated it (Supporting Information S2). We used a standardised version of the mean difference (from now on referred to as standardised mean difference [SMD]) so that we could analyse data from different quality of life measures together. In line with the Cochrane guidance [ref. 33], we chose to use the adjusted Hedges g version of SMD. For our analysis, SPSS calculated the adjusted Hedges g and its 95% confidence intervals based on mean change in quality of life score and standard error.
Standard deviation is required to calculate SMCs and SMDs, but it is not always reported. If a study did not report a standard deviation, then we calculated or estimated it, where possible, using methods suggested by Cochrane [ref. 33] (Supporting Information S2).
Results
Results of the Search
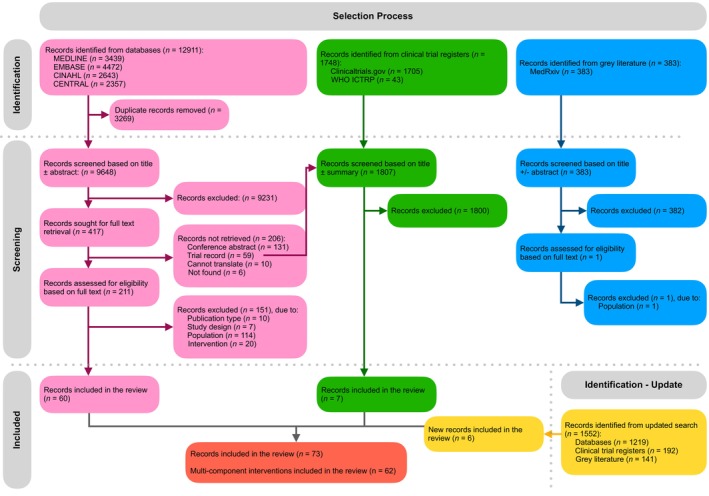
The PRISMA flow diagram detailing the selection process is shown in Figure 1. In the original search, 12 911 records from databases, 1748 clinical trial register records, and 383 pre‐prints were identified. After the removal of 3269 duplicates, 11 413 records were excluded based on titles and abstracts/summaries (Figure 1). Full texts in English were successfully retrieved for 212 records. Most studies excluded at the full‐text stage were excluded because of the population; in the majority of cases, this was because there was no evidence that at least 75% of participants had or were at risk of cachexia. In the updated search, an additional 1552 records were identified and screened following the same process as the original search. Ultimately, 73 records of 62 multicomponent interventions were included in the review.
Included Studies
For each multicomponent intervention, we identified a main record [ref. 20, ref. 21, ref. 22, ref. 34, ref. 35, ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41, ref. 42, ref. 43, ref. 44, ref. 45, ref. 46, ref. 47, ref. 48, ref. 49, ref. 50, ref. 51, ref. 52, ref. 53, ref. 54, ref. 55, ref. 56, ref. 57, ref. 58, ref. 59, ref. 60, ref. 61, ref. 62, ref. 63, ref. 64, ref. 65, ref. 66, ref. 67, ref. 68, ref. 69, ref. 70, ref. 71, ref. 72, ref. 73, ref. 74, ref. 75, ref. 76, ref. 77, ref. 78, ref. 79, ref. 80, ref. 81, ref. 82, ref. 83, ref. 84, ref. 85, ref. 86, ref. 87, ref. 88, ref. 89, ref. 90, ref. 91, ref. 92] alongside, in some cases, supplementary records (e.g., protocol and secondary data analysis). Tables A1 and A2 in the appendix provide overviews of the main study records for each of the 62 multicomponent interventions included in the review. The main studies were published between 1984 and 2024 and were conducted across 21 countries; 23 studies were conducted in Europe, 18 in North America, 14 in Asia, seven in Oceania and one in South America (Table A1). Of the 62 multicomponent interventions, we found published outcomes for 44 of them (Table A1); for the other 18, only a protocol or clinical trial register record was available (Table A2).
TABLE A1: Characteristics of included multicomponent interventions (with published results).
| Main record | Supplementary records | Study design (n of arms) | Country (n of centres) | n | Average age* | Males, n (%) | Primary cancer sites, n | Cancer stages, n | Cachexia or weight loss screening/classification criteria | % Weight change (time) | Cachexia prevalence, n | Intervention components | Comparator |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Naito et al. [ref. 21] | Mouri et al. [ref. 93] | NRSI—before–after study (1 arm) | Japan (4 centres) | 30 | 75 (70–85) | 20 (67%) | Lung n = 24, pancreatic n = 6 | III n = 3 and IV or postoperative recurrence n = 27 | Unintentional WL > 5% in last 6 months or WL > 2% and BMI < 20 or presence of muscle depletion according to consensus criteria | −3.0 ± 6.8 (6 months) | Cachexia n = 12 | Nutritional counselling + ONS + resistance exercise + PA counselling | None (uncontrolled) |
| Solheim et al. [ref. 48] | Balstad et al. [ref. 94] | Randomised trial (2 arms) | Norway and United Kingdom (3 centres) | 46 | Intervention: 63.0 (54.5–68.0) and control: 59.0 (52.5–67.0)jcsm13716-note-0006 | 26 (57%) | Lung n = 26, pancreatic n = 20 | III n = 16 and IV n = 30 | BMI < 30 kg/m2 and < 20% WL in last 6 months | Intervention arm: −5.7 and control arm: −5.4 (6 months)jcsm13716-note-0003 | Cachexia n = 46 | Nutritional counselling + ONS + omega 3 FA + celecoxib + resistance exercise + aerobic exercise | Standard care |
| Maccio et al. [ref. 42] | NA | Randomised trial (2 arms) | Italy (4 centres) | 144 | 60.5 ± 12.74 | Not specified | Ovary n = 50, endometrium n = 49 and cervix n = 25 | IIIC n = 5 and IV n = 119 | Loss of at least 5% of ideal or pre‐illness BW in the last 3 months | < −5% n = 2, −5% to −10% n = 69, > −10 n = 53 (3–6 months) | Cachexia n = 144 | Lipoic acid + L‐carnitine + carbocysteine + Celecoxib + heparin + psychosocial counselling | MA + heparin + psychosocial counselling |
| Buonaccorso et al. [ref. 20] | Buonaccorso et al. [ref. 95] | NRSI—before–after study (1 arm) | Italy (1 centre) | 24 | 65.96 ± 10.61 | 15 (63%) | Pancreatic n = 6, lung n = 5, renal n = 3, upper GI n = 3, bladder n = 2 and other n = 5 | Not specified | Refractory cachexia and cachexia based on ESPEN guidelines and MUST | Not specified | Reversible cachexia n = 20 and refractory cachexia n = 4 | Mapping changing eating habits + resistance exercise | None (uncontrolled) |
| Mikkelsen et al. [ref. 34] | Mikkelsen et al. [ref. 96] | Randomised trial (2 arms) | Denmark (1 centre) | 84 | Intervention 72.1 (67.3–74.5), control arm 71.5 (68.5–75.3)jcsm13716-note-0006 | 36 (43%) | Lung n = 39, pancreatic n = 36 and biliary tract n = 9 | Locally advanced n = 12 and metastatic n = 72 | None specified | Not specified | Unknown | ONS + resistance exercise + PA counselling + psychosocial counselling | Standard oncological treatment |
| Bland et al. [ref. 22] | NA | NRSI—before–after study—retrospective (1 arm) | Australia (1 centre) | 162 | 67.2 ± 12.0 | 94 (58%) | Upper GI n = 49, lung n = 38, colorectal n = 24, prostate n = 17, H&N n = 8 and other n = 26 | Metastatic n = 120 and locally advanced n = 42 | Pre‐cachexia: WL > 1 kg but < 5%. Cachexia: WL > 5% or WL > 2% and BMI < 20 kg/m2. Refractory cachexia: WL > 15% and BMI < 23 kg/m2 or WL > 20% and BMI < 27 kg/m2 | −10.4 ± 9.4 (6 months) | Pre‐cachexia n = 7, cachexia n = 83 and refractory cachexia n = 29 | Nutritional counselling + ONS + pharmacological management of NIS, malabsorption and inflammation if indicated + resistance exercise + PA counselling + fatigue/breathlessness management + safety advice | None (uncontrolled) |
| Chang [ref. 50] | NA | NRSI—before–after study—retrospective (1 arm) | Taiwan (1 centre) | 126 | Under 65 group: 48 [ref. 3, ref. 26, ref. 27, ref. 28, ref. 29, ref. 30, ref. 31, ref. 32, ref. 33, ref. 34, ref. 35, ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41, ref. 42, ref. 43, ref. 44, ref. 45, ref. 46, ref. 47, ref. 48, ref. 49, ref. 50, ref. 51, ref. 52, ref. 53, ref. 54, ref. 55, ref. 56, ref. 57, ref. 58, ref. 59, ref. 60, ref. 61, ref. 62, ref. 63], 65 + group 68 [ref. 64, ref. 65, ref. 66, ref. 67, ref. 68, ref. 69, ref. 70, ref. 71, ref. 72, ref. 73, ref. 74, ref. 75] | 116 (92%) | H&N n = 126 | III n = 24 and IV n = 102 | None specified | Not specified | Not specified | EN + NIS management + vitamins | None (uncontrolled) |
| Greig et al. [ref. 51] | NA | NRSI—before–after study (1 arm) | United Kingdom (1 centre) | 13 | 65.46 ± 9.84 | 5 (38%) | Pancreatic n = 5, gastric n = 2, cholangiocarcinoma n = 2, oesophageal n = 2, rectal n = 1 and mesothelioma n = 1 | IV n = 13 | WL ≥ 2% in 2 months or ≥ 5% in 6 months | −10.95 ± 4.42 (mean of 4.72 months) | Cachexia n = 13 | MA + formoterol fumarate | None (uncontrolled) |
| Parmar et al. [ref. 35] | Chasen et al. [ref. 97], Gagnon et al. [ref. 98], Parmar et al. [ref. 99], Eades et al. [ref. 100] | NRSI—before–after study—retrospective (1 arm) | Canada (1 centre) | 405 | 65.2 ± 13.3 | 208 (51%) | Lung n = 137, GI n = 104, haematological n = 48, breast n = 24 and miscellaneous n = 61 | Not specified | WL, anorexia or generalised functional decline; cachexia classification of participants based on international consensus definition | −10.2 ± 9.8 (6 months) | Pre‐cachexia n = 64, cachexia n = 257 | Nutritional counselling + pharmacological management of symptoms if indicated + resistance exercise + aerobic exercise | None (uncontrolled) |
| Del Fabbro et al. [ref. 52] | NA | NRSI—before–after study—retrospective (1 arm) | United States (1 centre) | 151 | 60 (19–86) | 95 (63%) | GI n = 58, respiratory n = 33, genitourinary n = 16, H&N n = 15 and other n = 29 | IV n = 147 | History of WL ≥ 5% and/or complained of poor appetite | −9 (100 days)jcsm13716-note-0003 | Cachexia n = 151 | Nutritional counselling + exercise recommendations | None (uncontrolled) |
| Wen et al. [ref. 36] | NA | Randomised trial (2 arms) | China (1 centre) | 108 | 61.95jcsm13716-note-0005 | 55 (51%) | Lung n = 34, gastric n = 13, breast n = 13, colorectal n = 11, liver n = 8, pancreas n = 7 and oesophagus n = 7 | III n = 10 and IV n = 83 | Loss of > 5% of pre‐illness or ideal BW in the last 3 months | −5% to −10% n = 75 | Cachexia n = 108 | MA + thalidomide | MA |
| Bibby et al. [ref. 53] | NA | NRSI—before–after study—retrospective (1 arm) | United Kingdom (1 centre) | 137 | 69 (35–86) | 70 (51%) | Pancreas n = 137 | Not specified | None specified | −6.3 (−27.7 to +10.4) (time unspecified) | Unknown | Nutritional counselling + ONS + EN or PN if indicated + PA counselling | None (uncontrolled) |
| Kufeldt et al. [ref. 54] | NA | NRSI—historically controlled cohort study (2 arms) | Germany (1 centre) | 2368 | 62.99 ± 12.31 | 555 (23%) | Bronchial, breast, prostate, oesophageal, cervical, ENT, lymphoma, glioblastoma and other (n not specified) | Not specified | None specified | Up to −39.2% n = 569 (of n = 666) | Unknown | Nutritional counselling + EN or PN if indicated | Historical control |
| Hajdu et al. [ref. 56] | NA | Randomised trial (2 arms) | Denmark (2 centres) | 69 | 62 (39–80)jcsm13716-note-0004 | 57 (83%) | H&N n = 69 | I n = 9, II n = 7, III n = 10 and IV n = 43 | None specified | Not specified | Unknown | Swallowing exercises + ONS + resistance exercise | Standard cancer care |
| Kouchaki et al. [ref. 58] | NA | Randomised trial (2 arms) | Iran (1 centre) | 90 | 60.92jcsm13716-note-0005 | 56 (62%) | Gastric n = 47, colorectal n = 26, oesophageal n = 14 and pancreatic n = 3 | Metastasis n = 30 and no metastasis n = 60 | At least 5% unintentional WL in the last 6 months or relative to premorbid stable weight or BMI < 20 | −17.45 (relative to premorbid historical stable weight)jcsm13716-note-0005 | Cachexia n = 90 | MA + Celecoxib | MA + placebo |
| Percival et al. [ref. 59] | NA | NRSI—before–after study (1 arm) | United Kingdom (1 centre) | 243 | 70 ± 10 | 139 (57%) | Lung n = 219, clinical diagnosis n = 14 and mesothelioma n = 10 (all thoracic) | I n = 30, II n = 26, III n = 52, IV n = 93. local n = 8 and extensive n = 34 | None specified | 5 (a). > −10% n = 59 | Cachexia n = 59 | Nutritional counselling + ONS | None (uncontrolled) |
| Sykes et al. [ref. 60] | NA | Randomised trial (2 arms) | United States (1 centre) | 55 | 63.2 ± 11.8 | 30 (55%) | H&N n = 55 | I or II n = 5 and III or IV n = 43 | None specified | Not specified | Unknown | Nutritional counselling + nutritional information + ONS + omega 3 FA | Standard care |
| Capozzi et al. [ref. 37] | NA | Randomised trial (2 arms) | Canada (1 centre) | 60 | 56.1 ± 9.2 | 49 (81.7%) | H&N n = 60 | I n = 1, II n = 7, III n = 3 and IV n = 48 | None specified | Not specified | Unknown | Nutritional information + resistance exercise + health education + behaviour change | Waitlist control |
| Daly et al. [ref. 62] | NA | Randomised trial (2 arms) | United States (2 centres) | 40 | 54jcsm13716-note-0005 | 32 (80%) | H&N n = 40 | II or nonstaged recurrence n = 3, III n = 9 and IV n = 28 | None specified | −7.15 (time unspecified)jcsm13716-note-0005 | Unknown | Nutritional counselling + ONS if indicated + pharmacological management of NIS if indicated | EN + nutritional counselling |
| Orell et al. [ref. 63] | NA | Randomised trial (2 arms) | Finland (1 centre) | 65 | Intervention 57 (52–64), control 61 (56–64)jcsm13716-note-0006 | 46 (79%) | H&N n = 65 | I n = 2, II n = 6, III n = 12, IV n = 37 and unknown n = 1 | None specified | Not specified | Unknown | Nutritional counselling + EN | On‐demand nutritional counselling |
| Luo et al. [ref. 43] | NA | NRSI—before–after study (1 arm) | Australia (1 centre) | 6 | 68.17 ± 6.05 | 5 (83%) | Pancreas n = 6 | Metastatic n = 4 and locally advanced n = 2 | None specified | Not specified | Unknown | Resistance exercise + aerobic exercise | None (uncontrolled) |
| Zhao et al. [ref. 38] | NA | Randomised trial (2 arms) | United States (2 centres) | 20 | 57jcsm13716-note-0005 | 17 (94%) | H&N n = 20 | III n = 4 and IV n = 14 | None specified | Not specified | Unknown | Resistance exercise + aerobic exercise | Standard treatment |
| Yennurajalingam et al. [ref. 64] | NA | NRSI—before–after study (1 arm) | United States (1 centre) | 45 | 64jcsm13716-note-0003 | 18 (64.3%) | Lung n = 17, bile duct n = 2, colon n = 2, pancreas n = 2, breast n = 2, gynaecological n = 2 and H&N n = 1 | Advanced n = 45 | Unintentional WL from ≥ 2% to ≤ 15% at any time in the last 12 months | Not specified | Unknown | Nutritional counselling + ONS + anamorelin + zinc + resistance exercise + aerobic exercise | None (uncontrolled) |
| Lundholm et al. [ref. 65] | NA | Randomised trial (2 arms) | Sweden (1 centre) | 138 | 70jcsm13716-note-0005 | Not specified | Oesophageal/gastric n = 53, pancreatic n = 44, colorectal n = 15, liver/bile duct n = 12, miscellaneous n = 9 and unknown n = 5 | Not specified | Manifest WL due to generalised malignant disease | −10 (1)jcsm13716-note-0007 | Unknown | Nutritional counselling + ONS if indicated + PN if indicated + insulin if indicated + indomethacin + erythropoietin | Best available palliative support |
| Persson et al. [ref. 67] | NA | Randomised trial (2 arms) | Sweden (1 centre) | 24 | 67.5jcsm13716-note-0005 | 10 (42%) | Pancreatic n = 8, colorectal n = 8, biliary n = 4, gastric n = 2 and adenocarcinoma or unknown primary origin n = 2 (all GI) | Not specified | > 10% WL in last 6 months | −12 (6 months)jcsm13716-note-0005 | Cachexia n = 24 | Nutritional counselling + melatonin + omega 3 FA | Omega 3 FA only OR melatonin only |
| Tobberup et al. [ref. 68] | NA | NRSI—historically controlled cohort study (2 arms) | Denmark (1 centre) | 120 | 67.75jcsm13716-note-0005 | 71 (59%) | Lung n = 120 | I–IIb n = 5, IIIa–IIIb n = 31 and IV n = 84 | Pre‐cachexia: any WL < 5% and presence of anorexia; cachexia: WL > 5% in last 6 months or ongoing WL > 2% and BMI < 20 or low skeletal muscle mass, < 55 cm2/m2 for men and < 39 cm2/m2 for women | −2.2 (compared to pretreatment weight)c | Cachexia n = 40, pre‐cachexia n = 14, no cachexia n = 66 | Nutritional counselling + omega 3 FA + resistance exercise + aerobic exercise | Historical control |
| Matsumoto et al. [ref. 69] | NA | NRSI—before–after study (1 arm) | Japan (1 centre) | 50 | 66 (40–78) | 42 (84%) | Lung n = 50 | IV or extensive disease n = 50 | None specified | Not specified | Unknown | Nutritionist referral if indicated + information + physical, psychological or socio‐economic intervention if indicated | None (uncontrolled) |
| Glare et al. [ref. 71] | NA | NRSI—before–after study (1 arm) | Australia (1 centre) | 54 | 62 (24–85) | 36 (68%) | Lung n = 18, colorectal n = 9, upper GI (oesophageal and gastric) n = 9, hepatopancreatobiliary n = 8, breast n = 4, prostate n = 2 and other n = 4 | Not specified | None specified | −10.2 (0–27) (6 months) | Unknown | Nutritional intervention + resistance exercise + aerobic exercise | None (uncontrolled) |
| Vaughan et al. [ref. 72] | NA | NRSI—before–after study—retrospective (1 arm) | Australia (1 centre) | 175 | 71 (33–91) | 107 (61%) | Lung n = 57, colorectal n = 17, pancreas n = 13, H&N n = 11, other GI n = 22, prostate n = 13, breast n = 9, other urogenital n = 10 and other n = 23 | I n = 2, II n = 3, III and loco‐regional disease not resectable n = 15, IV n = 130 and advanced disease stage not determined n = 25 | Concern of WL | Not specified | Unknown | Nutritional counselling + ONS + nutritional management of NIS if indicated (e.g., zinc) + education on cachexia impacts + omega 3 FA if indicated + pharmacological management of symptoms if indicated + exercise + vitamins | None (uncontrolled) |
| Mantovani et al. [ref. 73] | Mantovani et al. [ref. 101] | Randomised trial (5 arms) | Italy (not specified) | 332 | 61.94jcsm13716-note-0005 | 181 (55%) | Lung n = 72, breast n = 55, colorectum n = 45, pancreas n = 32, H&N n = 28, ovary n = 26, stomach n = 17, uterus n = 10, kidney n = 10, biliary ducts n = 8, bladder n = 7, prostate n = 7, oesophagus n = 8 and liver n = 7 | III n = 16 and IV n = 317 | Loss of > 5% of ideal or pre‐illness BW in last 3 months with or without abnormal values of pro‐inflammatory cytokines predictive of onset of clinical cachexia | < −5%: n = 76, −5 to −10%: n = 188 and > −10%: n = 68 (3–6 months) | Cachexia n = 332 | MA/MPA + thalidomide + polyphenols + lipoic acid + carbocysteine + omega 3 FA + L‐carnitine + vitamins | Polyphenols + lipoic acid + carbocysteine + vitamins + one of MA/MPA OR omega 3 FA OR L‐carnitine OR thalidomide |
| Jatoi et al. [ref. 74] | NA | Randomised trial (3 arms) | United States and Canada (26 centres) | 421 | 65.67jcsm13716-note-0005 | 293 (70%) | Lung n = 166, GI n = 141 and other n = 114 | Not specified | Self‐reported, 2‐month WL ≥ 5 lb (2.3 kg) and/or physician‐estimated caloric intake of < 20 cal/kg of BW per day | Not specified | Unknown | ONS + MA + omega 3 FA | MA + placebo OR omega 3 FA + placebo |
| Madeddu et al. [ref. 44] | NA | Randomised trial (2 arms) | Italy (1 centre) | 60 | 64.45jcsm13716-note-0005 | 33 (55%) | H&N n = 13, lung n = 12, colorectal n = 7, stomach n = 7, ovary n = 7, pancreas n = 6, oesophagus n = 2, biliary ducts n = 1 and liver n = 1 | IIIB n = 3 and IV n = 53 | Loss of ≥5% of ideal or pre‐illness BW in last 6 months (associated with abnormal values of pro‐inflammatory cytokines and/or CRP protein) | < −5%: n = 3, −5% to −10%: n = 29 and > −10%: n = 24 (time unspecified) | Cachexia n = 60 | MA + L‐carnitine + celecoxib | L‐carnitine + celecoxib |
| Odelli et al. [ref. 75] | NA | NRSI—historically controlled cohort study (2 arms) | Australia (1 centre) | 48 | 69.15jcsm13716-note-0005 | 33 (69%) | Oesophagus n = 48 | Not specified | None specified | −7.35 (compared with usual weight)jcsm13716-note-0005 | Unknown | Nutritional counselling + EN if indicated | Historical control |
| Ester et al. [ref. 61] | NA | NRSI—before–after study (1 arm) | Canada (1 centre) | 10 | 64.4 ± 10.3 | 4 (40%) | Lung n = 10 | IV n = 10 | None specified | Not specified | Unknown | Nutritional counselling + health behaviour change + resistance exercise + aerobic exercise + flexibility exercise | None (uncontrolled) |
| Cerchietti et al. [ref. 76] | NA | NRSI—before–after study (1 arm) | Argentina (1 centre) | 15 | 58.8 (40–76)jcsm13716-note-0004 | 11 (73%) | Lung n = 15 | IIIb n = 2 and IV n = 13 | Presence of cancer cachexia (i.e., ≥ 10% WL plus anorexia), fatigue, WHO PS ≥ 2 and evidence of acute‐phase response assessed by a level of CRP > 10 μg/mL | −17.8 (10–36.5) (compared to historical stable weight) | Cachexia n = 15 | MA + EN + celecoxib + exercise + psychological/psychiatric intervention if indicated | None (uncontrolled) |
| Wang et al. [ref. 77] | NA | NRSI—historically controlled cohort study (2 arms) | Taiwan (1 centre) | 58 | 53.5jcsm13716-note-0005 | 54 (93%) | H&N n = 58 | IVA n = 37 and IVB n = 21 | None specified | Not specified | Unknown | Nutritional counselling + ONS + EN | No nutrition support (historical control) |
| Vanderbyl et al. [ref. 39] | NA | Randomised trial—crossover (2 arms) | Canada (1 centre) | 36 | 64.9jcsm13716-note-0005 | 14 (38%) | Lung n = 12 and GI n = 12 | III n = 8 and IV n = 16 | None specified | Not specified | Unknown | Resistance exercise + aerobic exercise | Qigong |
| Chen et al. [ref. 45] | NA | Randomised trial (2 arms) | China (1 centre) | 120 | 63.02jcsm13716-note-0005 | 81 (68%) | Lung n = 120 | IIIB n = 14, IIIC n = 18 and IV n = 88 | None specified | Not specified | Unknown | Nutritional intervention as indicated + psychotherapy + pain management if indicated + psychotropic intervention if indicated | Standard care |
| Jatoi et al. [ref. 40] | NA | Randomised trial (3 arms) | United States (20 centres) | 485 | 66.33jcsm13716-note-0005 | 308 (66%) | Lung n = 208, GI n = 139 and other n = 122 | Not specified | Self‐reported WL ≥ 5 pounds (2.3 kg) in the last 2 months and/or physician‐estimated caloric intake of < 20 cal/kg of BW per day | Not specified | Unknown | MA + dronabinol | MA only OR dronabinol only |
| Xie et al. [ref. 41] | NA | Randomised trial (2 arms) | China (1 centre) | 54 | 56.84 (46–66)jcsm13716-note-0004 | 42 (75%) | Lung n = 54 | IV n = 54 | WL > 5% or BMI < 20 kg/m2 in 6 months and WL > 2% or appendicular SMI matched with sarcopenia (male < 7.26 kg/m2, female < 5.45 kg/m2) having lost > 2% weight | Not specified | Cachexia n = 54 | Thalidomide + cinobufagin | Cinobufagin |
| Bakitas et al. [ref. 79] | Bakitas et al. [ref. 102] | Randomised trial (2 arms) | United States (1 centre) | 332 | 65.3jcsm13716-note-0005 | 168 (60%) | GI tract n = 119, lung = 93, genitourinary tract n = 37 and breast n = 30 | Advanced n = 332 | None | Not specified | Unknown | Education on cancer + problem‐solving + communication and social support + symptom management + advance care planning | Care as usual |
| Kapoor et al. [ref. 81] | NA | Randomised trial (2 arms) | India (1 centre) | 63 | 45.9jcsm13716-note-0005 | 0 (0%) | Anorectum n = 5, bone n = 3, brain n = 1, breast n = 14, buccal cavity n = 2, chest wall n = 2, eyelid n = 1, genitourinary tract n = 22, lung n = 6, olfactory n = 1, spine n = 1, suprarenal mass n = 1 and thyroid n = 4 | Advanced n = 63 | WL > 5% from pretreatment weight, BMI < 20 and haemoglobin < 12 g/dL and energy intake < 1500 kcal/day | Not specified | Cachexia n = 63 | Nutritional counselling + atta flour | Nutritional counselling |
| Obling et al. [ref. 82] | NA | Randomised trial (2 arms) | Denmark (1 centre) | 47 | 66.9 (41.5–88.2) | 30 (64%) | Oesophagus n = 2, stomach n = 9, duodenum n = 2, pancreas n = 27, bile duct n = 2 and colorectum n = 5 | Advanced n = 47 | None specified | < 5% n = 8, 5%–10% n = 10 and 10% n = 29 | Cachexia n = 47 | Nutritional counselling + PN | Nutritional counselling |
| Milla et al. [ref. 92] | NA | Randomised trial (2 arms) | Spain (4 centres) | 57 | 64.03jcsm13716-note-0005 | 37 (65%) | H&N n = 4, upper GI (oesophagus, stomach) n = 8, lower GI (small and large intestines) n = 9, pancreas n = 5, liver n = 1, lung n = 15, gynaecological n = 2, urological n = 6 and other n = 7 | I n = 1, II n = 9, III n = 10 and IV n = 25 | WL > 5% in last 6 months | Not specified | Cachexia n = 57 | Nutritional counselling + ONS (enriched) | Nutritional counselling + ONS (standard) |
Note: Studies were prospective designs unless otherwise stated. Randomised trials were parallel design unless otherwise stated. Age and baseline weight loss of participants were reported as mean ± standard deviation or median (range) unless indicated by the following:
Median only,
Mean (range),
Mean only,
Median (interquartile range),
Mean (standard error).
Abbreviations: BMI, body mass index; BW, body weight; CRP, C‐reactive protein; EN, enteral nutrition; ENT, ear, nose and throat; ESPEN, European Society for Clinical Nutrition and Metabolism; FA, fatty acids; GI, gastrointestinal; H&N, head and neck; MA, megestrol acetate; MPA, medroxyprogesterone acetate; MUST, Malnutrition Universal Screening Tool; NIS, nutrition impact symptoms; NRSI, nonrandomised study of intervention; ONS, oral nutritional supplement; PA, physical activity; PN, parenteral nutrition; PS, performance status; SMI, skeletal muscle index; WHO, World Health Organisation; WL, weight loss.
TABLE A2: Characteristics of included multicomponent interventions (without published results).
| Study | Trial registration number | Study design (n of arms) | Country (n of centres) | Primary cancer sites | Cancer stages | Cachexia or weight loss screening/classification criteria | Intervention components | Comparator |
|---|---|---|---|---|---|---|---|---|
| Protocol papers | ||||||||
| Miura et al. [ref. 46] | UMIN000028801 | Randomised trial (2 arms) | Japan (15 centres) | Lung and pancreas | Advanced (lung) or inoperable (pancreas) | None specified | Nutritional counselling + ONS + resistance exercise + PA counselling | Standard care |
| Maeng et al. [ref. 47] | NCT04907864 | Randomised trial (2 arms) | South Korea (1 centre) | Stomach, colorectum, pancreas, biliary tract and lung | Recurrent or metastatic | Pre‐cachexia: WL ≤ 5% in last 6 months, anorexia and metabolic changes, e.g., glucose intolerance; cachexia: WL > 5% in the last 6 months or BMI < 20 and WL > 2% in the last 6 months | Nutritional counselling + ONS + omega 3 FA + ibuprofen + Bojungikki‐tang + resistance exercise + aerobic exercise + flexibility exercise | Conventional palliative care |
| Waldman et al. [ref. 55] | NCT04629300 | Randomised trial (2 arms) | United States (6 centres) | Lung | III or IV | None specified | Managing energy + symptom self‐management + social support + coping skills + reflection | Usual oncology care |
| Neuzillet et al. [ref. 57] | NCT02184663 | Randomised trial (2 arms) | France (16 centres) | Pancreas | Locally advanced or metastatic | None specified | Nutritional intervention (oral or EN or PN) as indicated + resistance exercise + aerobic exercise | Usual care |
| Rogers et al. [ref. 66] | ACTRN12611000870954 | Randomised trial (2 arms) | New Zealand (1 centre) | Lung | Not specified | Loss of 5% of oedema‐free BW in the last 3–6 months; if no documented WL, BMI < 20.0 kg/m2; at least three out of (a) patient‐reported decreased muscle strength, (b) fatigue, (c) patient‐reported anorexia,(d) low fat‐free mass index and (e) abnormal biochemistry | Celecoxib + omega 3 FA + amino acids + resistance exercise | Omega 3 FA + celecoxib |
| Quist et al. [ref. 70] | NCT01881906 | Randomised trial (2 arms) | Denmark (1 centre) | Lung | IIIb or IV | None specified | Resistance exercise + aerobic exercise | Usual care (wait‐list control) |
| Solheim et al. [ref. 49] | NCT02330926 | Randomised trial (2 arms) | Europe and Canada (13 centres) | Lung and pancreas | Not specified | None specified | Nutritional counselling + ONS + ibuprofen + omega 3 FA + resistance exercise + aerobic exercise | Standard care (wait‐list control) |
| Moinuddin et al. [ref. 78] | Not specified | Randomised trial (4 arms) | United States (1 centre) | Any | Any | WL ≥ 5% in the last 12 months, plus any three of the following criteria: decreased muscle strength, fatigue, anorexia, low fat‐free mass index and abnormal biochemistry | Citrulline + resistance exercise | Control OR resistance exercise only OR citrulline only |
| Trial records | ||||||||
| Li et al. [ref. 86] | NCT02681601 | Randomised trial (2 arms) | United States | Pancreas | Unresectable | WL > 5% in last 6 months | Diet prescription + ONS + omega 3 FA | Diet prescription |
| Waqar et al. [ref. 85] | NCT01501396 | Randomised trial (2 arms) | United States | Not specified | Not specified | WL ≥ 5% in the last 6 weeks or WL ≥ 10% in the last 6 months and poor appetite | MA + mirtazapine | MA |
| Strasser et al. [ref. 84] | NCT01127386 | Randomised trial (3 arms) | Switzerland | Any | Advanced (locally recurrent or metastatic) and incurable | Cachexia: WL ≥ 2% in last 2 months or ≥ 5% in last 6 months and ongoing in last 4 weeks and CRP ≥ 30 mg/L | Lenalidomide + prokinetics + nutritional counselling + PA counselling | Prokinetics + nutritional counselling + PA counselling |
| Dev et al. [ref. 83] | NCT00625742 | NRSI—before–after study (1 arm) | United States | Not specified | Not specified | WL ≥ 5% in last 6 months | Nutritional counselling + ONS + melatonin + ibuprofen + atenolol if indicated + resistance exercise + aerobic exercise | None (uncontrolled) |
| Naito et al. [ref. 80] | PRN‐jRCTs041210053 | Randomised trial (2 arms) | Japan | Lung and pancreas | Locally advanced or metastatic | Cancer cachexia with an indication of anamorelin hydrochloride | Nutritional counselling + anamorelin + resistance exercise + PA counselling | Active control |
| Croagh et al. [ref. 91] | ACTRN12624000084583 | Randomised trial (2 arms) | Australia | Pancreas | Borderline resectable, locally advanced or metastatic | None specified | Nutritional counselling + education + ONS + EN if indicated + pharmacological NIS management | Usual nutrition care |
| Yen et al. [ref. 90] | NCT05915325 | NRSI—before–after study (1 arm) | Taiwan | Not specified | Not specified | Pre‐cachexia: WL 1%–5% in last 6 months or WL 1%–2% and BMI < 20 and one of: elevated CRP, impaired fasting glucose or diabetes, corticosteroid use, hypogonadism and insufficient calorie intake (< 20 kcal/kg/day); cachexia: WL > 5% in last 6 months or WL > 2% and BMI < 20 | Nutritional counselling + aerobic exercise + resistance exercise | None (uncontrolled) |
| van Laarhoven et al. [ref. 89] | NCT06138223 | Randomised trial (2 arms) | Netherlands | Oesophagus and stomach | Incurable | None specified | Nutritional counselling + aerobic exercise + resistance exercise + PA counselling | Usual care |
| Peyrachon et al. [ref. 88] | NCT06323733 | NRSI—before–after study (1 arm) | France | Not specified | Not specified | Cachexia according to Martin et al. classification | Aerobic exercise + resistance exercise | None (uncontrolled) |
| Unspecified et al. [ref. 87] | NCT06338683 | Randomised trial (2 arms) | China | Stomach, oesophagus, hepato‐pancreaticobiliary and lung | III or IV inoperable or metastatic | None specified | Nutritional counselling + olanzapine | Nutritional counselling |
Note: Studies were all prospective design. Randomised trials were parallel design unless otherwise stated. For trial records, the name is the principal investigator and the year is when the record was first posted on the trial register.
Abbreviations: BMI, body mass index; BW, body weight; CRP, C‐reactive protein; EN, enteral nutrition; FA, fatty acids; MA, megestrol acetate; NIS, nutrition impact symptoms; NRSI, nonrandomised study of intervention; ONS, oral nutritional supplement; PA, physical activity; PN, parenteral nutrition; WL, weight loss.
Study Designs
Of the 62 main studies included, 39 were randomised trials and 23 were NRSIs (Tables A1 and A2). Of the NRSIs, 19 were before–after studies (uncontrolled), and four were historically controlled cohort studies. Of the 44 studies with published results, the median (range) sample size at baseline was 61.5 (6–2368) (Table A1).
Participants
The median (range) age of participants at baseline was 64.2 (45.9–75) years (Table A1). Of the 15 studies that reported the average percentage weight loss of participants at baseline; the median (range) was 10% (2.2%–17.8%), which in most studies was over the preceding 3–6 months or compared with pre‐illness/pretreatment stable weight. Of the 4654 total participants with cancer site reported, 1591 (34%) had lung cancer, 1509 (32%) had gastrointestinal cancers, 577 (12%) had head and neck cancers and 977 (21%) had other or unspecified cancer sites (Table A1). Of the 2084 total participants with cancer stage reported, 265 (13%) had Stage 3 and 1682 (81%) had Stage 4 disease. Information on cachexia status at baseline was available for 2188 participants; of these, 1708 (78%) had cachexia or refractory cachexia (Table A1).
Risk of Bias in Included Studies
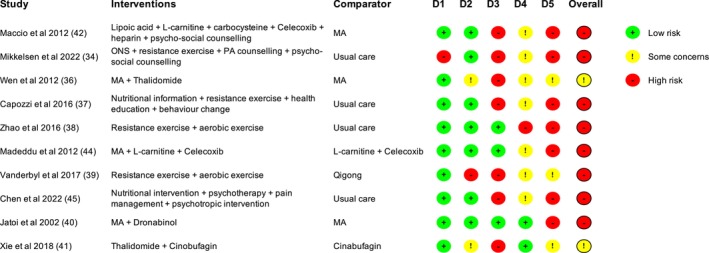
The 13 studies that provided data for the quality of life analysis were assessed for risk of bias [ref. 20, ref. 34, ref. 35, ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41, ref. 42, ref. 43, ref. 44, ref. 45]. Figures 2 and 3 provide summaries of the assessments.

Randomised Trials
We assessed eight randomised trials to be at high risk of bias overall, while the other two raised some concerns (Figure 2). Only one study had a high risk of bias relating to the randomisation process. Six studies were unblinded, in most cases, because of the nature of the intervention (e.g., exercise). Although the appropriate study population for an analysis of the intention to treat effect is all randomised participants, two studies appeared to only analyse participants who completed the trial, and in one, it was unclear who was included in the analysis.
Nonrandomised Studies of Interventions
We assessed all three NRSIs to be at critical risk of bias overall (Figure 3). All studies had a critical risk of bias because of confounding, but all had a low risk of bias relating to the selection of participants for the study, classification of interventions and deviations from intended interventions.
All Studies
Seven of 10 randomised trials had a high risk of bias, and two of three NRSIs had a serious risk of bias because of missing outcome data. In these studies, the ‘complete cases’ (participants for which complete data were available) may not be representative of those with missing data, potentially leading to an overestimate of the value of the intervention (inflating the within‐group quality of life change estimates). As all quality of life measures were subjective, assessment could have been influenced by knowledge of the intervention received. Therefore, given that all measures were participant‐reported, there were some concerns regarding the measurement of the outcome in all studies where participants were unblinded. In relation to the selection of the reported result, seven of 10 randomised trials had a high risk of bias, and two of three NRSIs had a critical or serious risk of bias.
Intervention Analysis
Intervention Categories
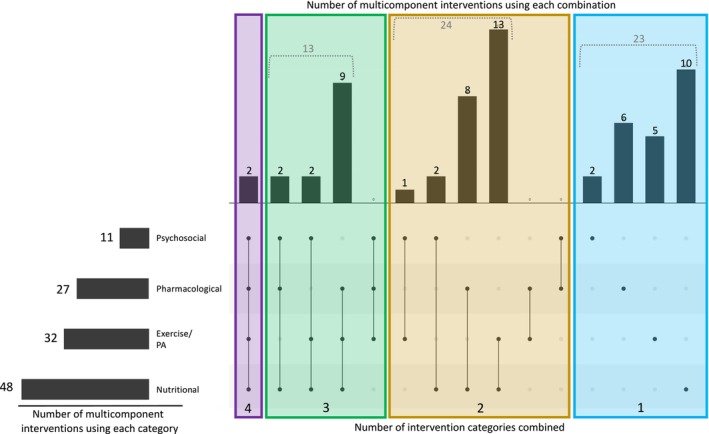
From the 62 multicomponent interventions, we identified 232 individual components. Of the 232 components, 99 (43%) were nutritional, 62 (27%) were exercise/physical activity, 50 (22%) were pharmacological, and 21 (9%) were psychosocial.
Of the 62 multicomponent interventions, 48 (77%) included a nutritional component, 32 (52%) included an exercise/physical activity component, 27 (44%) included a pharmacological component and 11 (18%) included a psychosocial component (Figure 4). Two multicomponent interventions combined components from all four intervention categories [ref. 72, ref. 76]. The most common combination of intervention categories was nutritional + exercise/physical activity, which was used in 13 multicomponent interventions.
Key Features of Cancer Cachexia Targeted
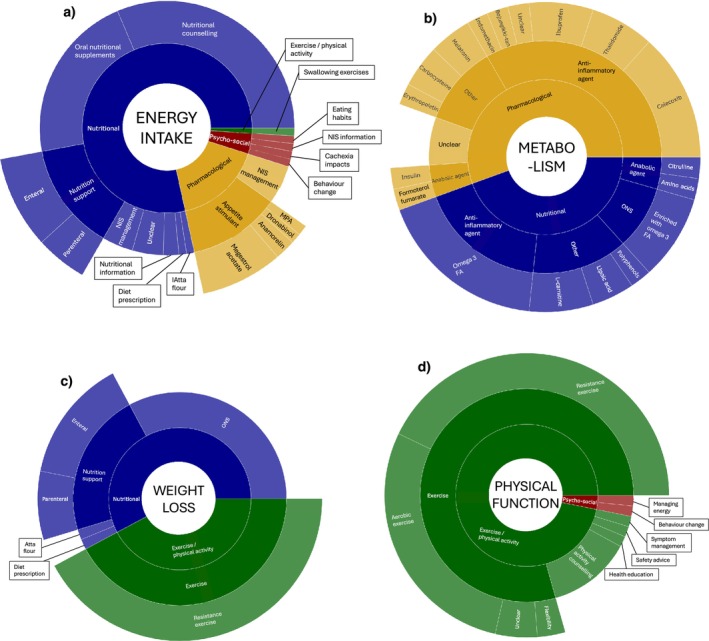
Of the 232 components, 102 (44%) targeted energy intake, 63 (27%) targeted physical function, 64 (28%) targeted weight/muscle loss and 45 (19%) targeted metabolism.
Most components targeting energy intake were nutritional (80/102, 78%), and the most common type of component was nutritional counselling (32/102, 31%; Figure 5a). Components targeting metabolism were either pharmacological (25/45, 56%) or nutritional (20/45, 44%), and over half were anti‐inflammatory agents (26/45, 58%; Figure 5b). Components targeting weight/muscle loss were either nutritional (37/64, 58%) or exercise/physical activity (27/64, 42%; Figure 5c). Most components targeting physical function were exercise/physical activity (61/63, 97%), and the most common type of component was resistance training (27/63, 43%; Figure 5d).
Of the 62 multicomponent interventions, 51 (82%) targeted energy intake, 45 (73%) targeted weight/muscle loss, 33 (53%) targeted physical function and 24 (39%) targeted metabolism (Figure 6). Nine multicomponent interventions targeted all four key features of cachexia [ref. 22, ref. 35, ref. 47, ref. 48, ref. 49, ref. 68, ref. 72, ref. 76, ref. 83]. Fourteen multicomponent interventions targeted energy intake + weight/muscle loss + physical function, which was the most common combination of key features of cachexia targeted together.
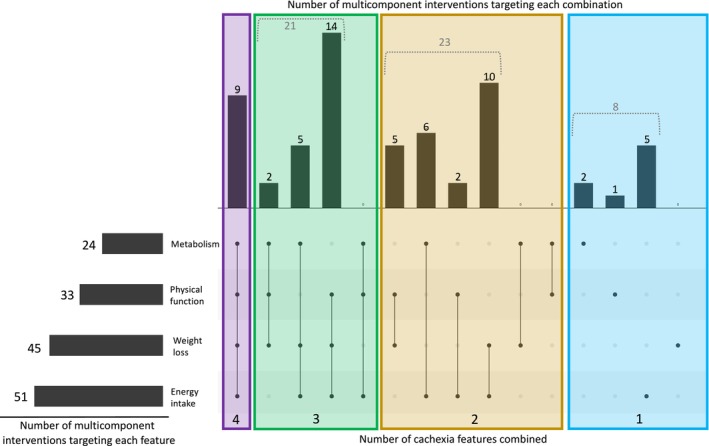
Tailoring
Of the 232 components, 36 (16%) had targeted provision (i.e., were only given to study participants who needed it), of which 23/36 (64%) were nutritional, 11/36 (31%) were pharmacological, and 2/36 (6%) were psychosocial. Of the 232 components, 96 (41%) were individualised (i.e., modified to each participant), of which 48/96 (50%) were exercise/physical activity, 39/96 (41%) were nutritional, and 9/96 (9%) were psychosocial. Based on these classifications, just over half of all components were tailored (126/232; 54%). Overall, 18/62 (29%) multicomponent interventions were fully tailored, 30 (48%) were partly tailored, and 14 (23%) were not tailored.
Effects on Quality of Life
Twenty‐one studies reported data on quality of life at baseline and at least one follow‐up time point and/or as change over time. However, two of these studies reported data as median and range, so we excluded them from our analysis [ref. 74, ref. 81]. Nine studies reported within‐arm mean change in quality of life over time and its associated standard deviation (or standard error, which we converted to standard deviation) [ref. 20, ref. 34, ref. 35, ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41]. We used these data to calculate a within‐arm SMC for each intervention arm. For the remaining 10 studies, we calculated within‐arm mean change in quality of life over time as it was not reported by the study [ref. 22, ref. 42, ref. 43, ref. 44, ref. 45, ref. 58, ref. 61, ref. 69, ref. 73, ref. 82]. However, we were only able to estimate standard deviations and therefore calculate SMCs, for four of these studies [ref. 42, ref. 43, ref. 44, ref. 45]. Therefore, 13 SMCs were analysed (Figure 7).
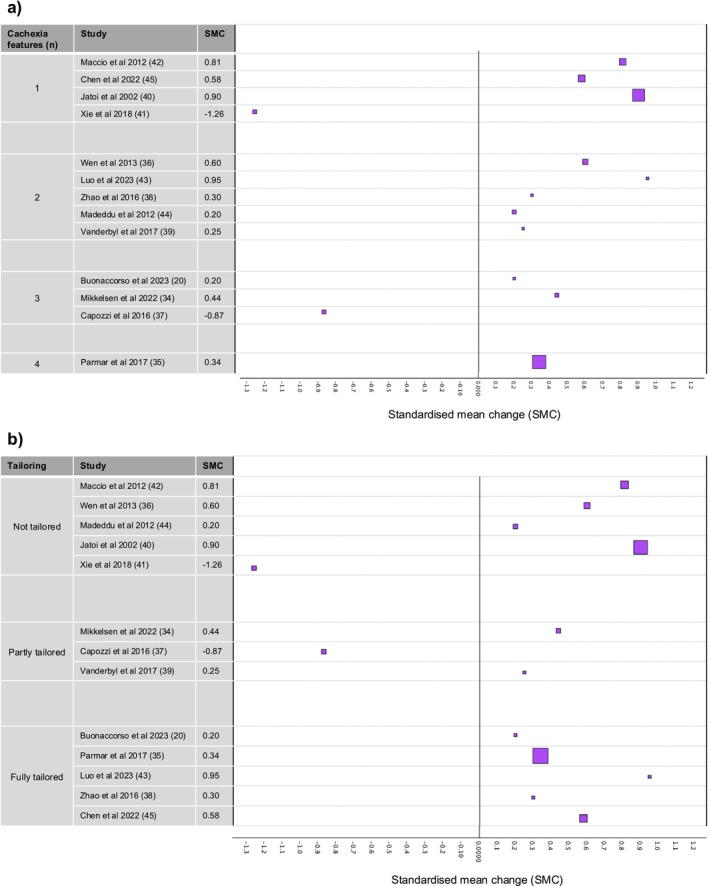
Amongst the 13 studies, the following measures of quality of life were used: EORTC‐QLQ‐C30 standardised overall score [ref. 36, ref. 41, ref. 42, ref. 44], FAACT raw overall score [ref. 20, ref. 35, ref. 40], FACT‐G overall score as percentage [ref. 39]; FACT‐Lung overall raw score [ref. 45], FACT‐Anaemia trial outcome index [ref. 37], MOS‐SF‐36 raw overall score [ref. 38] and EORTC‐QLQ‐C30 standardised global health status subscale score [ref. 34, ref. 43]. For all these quality of life measures, a higher score indicated a better quality of life.
In two studies, quality of life scores declined over the intervention period [ref. 37, ref. 41], whereas in 11 studies, quality of life scores suggested an improvement (Figure 7). SMCs varied from −1.26 to 0.95. Amongst these data, there was no indication that the number of key features of cachexia targeted, or the extent of tailoring, was associated with a greater improvement in quality of life scores.
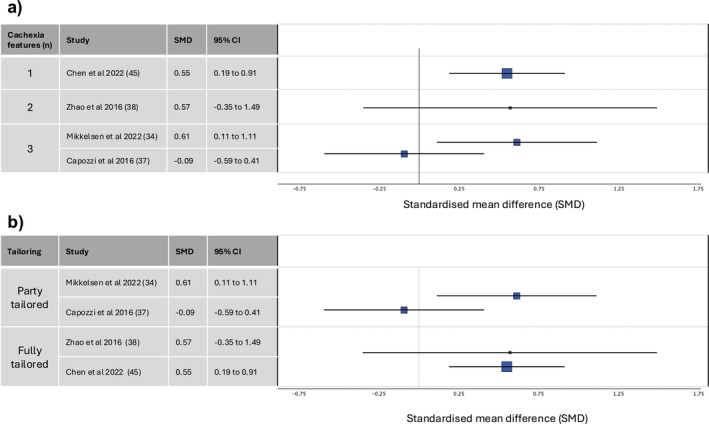
Only four studies with quality of life data compared the multicomponent intervention with usual care [ref. 34, ref. 37, ref. 38, ref. 45] (Figure 8). In three studies, the multicomponent intervention arm showed an improvement in quality of life scores, while the usual care arm showed either no change or a decline in quality of life scores [ref. 34, ref. 38, ref. 45]. Two of these studies reported the results of a statistical test, and in both, the difference between the two arms was statistically significant. In the fourth study, both arms showed a decline in quality of life scores, which was greater in the multicomponent intervention arm but not statistically significant [ref. 37].
Discussion
Summary of Key Results
We identified 62 multicomponent interventions for people with cancer cachexia. Only two interventions combined all four intervention categories (pharmacological, nutritional, exercise/physical activity and psychosocial); most included a nutritional component, whereas a minority included a psychosocial component. Nine interventions targeted all four key features of cachexia (reduced energy intake, altered metabolism, involuntary weight loss and decline in physical function); most targeted energy intake, but fewer than half targeted metabolism. We found that about half of all individual components were tailored towards individuals; in most cases, these were nutritional or exercise/physical activity components delivered in an individualised manner. Eighteen multicomponent interventions were fully tailored, and 30 were partly tailored to individuals.
Although outcomes were available for 44 interventions, only 13 reported quality of life data that could be used to calculate SMCs. In 11 of these studies, quality of life scores suggested an improvement over the intervention period, amongst participants in the multicomponent intervention arm. Four studies compared a multicomponent intervention with usual care; in three, quality of life scores suggested a greater improvement in quality of life in the intervention arm than in the usual care arm. Amongst the limited data, there was no suggestion that the number of key features of cachexia targeted, or the extent of tailoring, was associated with a greater improvement in quality of life.
Results in Context
To the best of our knowledge, this is the first systematic review to map out in detail the combinations of intervention categories used and key features of cachexia targeted by multicomponent interventions for adults with cancer cachexia. An intervention that combines all four intervention categories is likely to require the involvement of multiple healthcare professions, bringing challenges around organisation and funding. This may explain why one of the studies that used all four categories was a retrospective, nonexperimental study of a clinical service and the other was a small‐scale pilot study. On the other hand, more interventions targeted all four key features of cachexia. We would not have been able to determine this if only randomised trials with results had been included (which is often the case with systematic reviews) because four of the studies that targeted all four key features of cachexia were NRSIs (three nonexperimental studies of clinical services and one historically controlled cohort study) and two were protocols of randomised trials without published results. This emphasises the value of including NRSIs and ongoing studies, to increase our understanding of the types of interventions that have been and are being investigated for cancer cachexia. However, it also highlights the challenges of evaluating the effectiveness of these interventions.
As nutritional aspects of cachexia have dominated the literature and interventions have generally taken a biomedical approach, we were not surprised that a high proportion of multicomponent interventions involved a nutritional component and/or targeted energy intake. However, we had expected a higher proportion of the interventions to target metabolism. Meanwhile, only 11 multicomponent interventions involved a psychosocial component, which is likely because the psychosocial impacts of the syndrome have only become a focus of cachexia literature more recently. Interview studies exploring experiences of cancer cachexia have repeatedly reported themes of eating‐related distress and family conflicts, in part because of a lack of understanding about cachexia and the role of food in reversing weight loss [ref. 5, ref. 6]. Participants receiving psychosocial/educational interventions have reported improved health literacy, awareness about nutrition and relationships with food in recent interview studies [ref. 19, ref. 20]. These positive experiences suggest that some of the key quality of life issues around eating may be effectively tackled with psychosocial interventions rather than, or in addition to, nutritional interventions. Therefore, we believe that future interventions for people with cancer cachexia should include a psychosocial component. Going forward, the recently developed patient‐reported outcome measure to assess eating‐related distress [ref. 103] could be used in addition to qualitative data from interview studies, to evaluate the effects of interventions on this key quality of life issue amongst people with cancer cachexia.
We believe this is also the first review to systematically assess tailoring across interventions for people with cancer cachexia. Over a quarter of the multicomponent interventions were fully tailored, and almost half were partly tailored. This, in part, was due to the inclusion of NRSIs (10 out of the 18 fully tailored interventions and 11 out of the 30 partly tailored interventions were NRSIs), particularly nonexperimental studies of clinical services, which often provided intervention components based on indicated need. Interview studies have found that patients want and value interventions that are individualised and flexible [ref. 16, ref. 19, ref. 20], so we believe that future interventions should take a tailored approach. Ideally, this should not be limited to NRSIs and be incorporated into large‐scale clinical trials, so that the effectiveness of tailored interventions can be evaluated with sufficient power to detect effects and without the methodological limitations and potential biases of NRSIs.
We had expected more of the 62 multicomponent interventions to report data on quality of life and were surprised at the number of studies that did not intend to measure it. Assessment of quality of life in interventional studies is an important part of conducting patient‐centred research and whether patients perceive a benefit should be a key consideration towards whether an intervention is deemed to be successful or not [ref. 104]. It is a particularly relevant outcome for the cancer cachexia population, who often have incurable cancers and/or irreversible cachexia and so outcomes like survival are not appropriate measures of intervention success. Furthermore, this population experiences multidimensional quality of life issues related to both the nutritional and functional aspects of cachexia [ref. 104, ref. 105]. Therefore, measures of overall quality of life that encompass these domains should be reported in addition to subscales and specific symptoms that are relevant to the population, like eating‐related distress.
We were encouraged by the fact that 11 out of the 13 studies that we could calculate SMCs for reported that participants in the multicomponent intervention arm experienced an improvement in quality of life over the intervention period. However, we believe that this result comes with a high risk of bias and should be interpreted with caution. There were many studies that measured quality of life but did not report the data (or only reported subscales and not the overall score) and so could not be included in our analysis. Additionally, we assessed 11 out of the 13 studies in our analysis as having a high risk of bias. Much of this risk of bias is difficult to avoid in this field, in part because of the nature of the interventions (blinding is often not possible), the nature of the outcome measure (quality of life being a subjective and participant‐reported measure, where participants are usually aware of the intervention received, leading to risk of bias in measurement of the outcome), and the population (high levels of attrition due to worsening health status and death, leading to risk of bias due to missing outcome data). However, nine out of the 13 studies had a high risk of bias because of the selection of the reported result, which can be controlled by the investigators. In addition, there may be publication bias whereby studies showing improvements in outcomes like quality of life were more likely to be published than studies not showing improvements. As a result, we believe that (a) the SMCs in our analysis are likely to be overestimates and (b) the number of studies of multicomponent interventions in which participants experienced a decline in quality of life over the intervention period is likely to be higher than is shown in this review.
Furthermore, although there was no indication that the number of key features of cachexia targeted, or the extent of tailoring, was associated with a greater improvement in quality of life scores amongst these data, the heterogeneity across the 13 studies prevented us from concluding on our hypothesis. Therefore, to better understand the effects of multicomponent interventions on quality of life for people with cancer cachexia, we need more robust and rigorous randomised trials that measure quality of life and report it appropriately and transparently.
Although this systematic review was being conducted, a new Phase 2 drug trial testing the efficacy and safety of ponsegromab was published [ref. 106]. As pharmaceutical trials such as this one continue their efforts to develop a drug that effectively alleviates cachexia, we believe that nonpharmacological interventions remain an important focus of cachexia research. Ponsegromab has shown promising early results, with participants gaining weight over 12 weeks and reporting reduced symptom burden. However, the inclusion criteria for participants were strict (including GDF‐15 levels of at least 1500 pg/mL), and the list of exclusion criteria was long, which led to over a quarter of the participants initially enrolled on the trial being excluded. This highlights the need for a range of interventions to be available, so that suitable options exist for everybody with cachexia.
Strengths and Limitations of the Review Methods
As there is no agreed framework for the key features of cancer cachexia, we devised four key features for use in this review. We identified these key features based on the consensus definition of cancer cachexia [ref. 1] and guidelines from ASCO [ref. 31] and ESMO [ref. 12], which ensured that they were strongly aligned with current understanding and agreement about what cachexia is. However, the psychosocial impact of cachexia on patients is not represented in these definitions, so we did not include it as a key feature of cachexia in this review. Instead, ‘psychosocial’ was included as an intervention category. However, targeting the psychosocial impact of cachexia in addition to the four key features could lead to greater improvements in quality of life, as indicated by qualitative findings of interventional studies [ref. 19, ref. 20]. As it was not assessed in this review, future work could explore whether interventions targeting the psychosocial impacts of cachexia have greater effects on quality of life than interventions which do not.
We classified each intervention component based on which key feature(s) of cachexia it targeted and not on which feature(s) it impacted. For example, we classified aerobic exercise as targeting physical function, but we acknowledge that it is also likely to impact the other three features (metabolism, energy intake and weight/body composition). This distinction was necessary for the purposes of the review, to avoid classifying all interventions as having an impact on all four features. However, biological connections between the features of cachexia are important to consider, to fully understand which interventions may be effective at improving this multifactorial syndrome.
We included studies where there was evidence that the population either had cachexia or was at risk of cachexia. We took this approach because if the review had been restricted only to studies that assessed potentially eligible participants for cachexia, we would have excluded many relevant studies. We used cancer stage and site as indicators of cachexia risk. Published data show that certain cancers have a much higher prevalence of cachexia than others [ref. 3, ref. 27]. Furthermore, as cachexia is a progressive syndrome, it is generally agreed that cachexia increases with cancer severity [ref. 107, ref. 108, ref. 109]. Although there are some data supporting this [ref. 111, ref. 112], we acknowledge that this is not universal and that cachexia can present in earlier stages of disease. However, no indicator of cachexia risk is perfect, and other clinical characteristics, such as nutritional status or chronic comorbidities, may also indicate the risk of cachexia.
Furthermore, we did not exclude studies based on the intended target of the intervention. For example, if a study met all our eligibility criteria and stated that their multicomponent intervention was aimed at improving fatigue or poor appetite, we included it in the review. We made this decision because the aim of this review was to evaluate multicomponent interventions that have been tested amongst people with (or at risk of) cancer cachexia, rather than only evaluating multicomponent interventions explicitly aimed at cachexia. However, this meant that some studies included in the review investigated interventions that were not necessarily designed with cachexia in mind. This could be one of the reasons that some interventions targeted only one or two of the key features of cachexia, so should be considered when interpreting the results of the review.
We used standardised means to represent change over time and difference between arms (SMCs and SMDs). This approach allowed us to compare the effect on quality of life across studies that used different measurement instruments, which was necessary to explore differential effects on quality of life. However, this approach also introduced some limitations. First, it meant excluding eight studies that reported data on quality of life but for which SMCs could not be calculated. Second, there were small sample sizes in many studies, which can lead to higher standard deviations and therefore lower SMCs. When compared to larger studies with smaller standard deviations, this may skew the results in favour of the larger studies.
Summary of Recommendations
We believe that future interventions for people with cancer cachexia should include a psychosocial component and should take a tailored approach. Although challenging, these approaches should not be limited to NRSIs and small pilot studies, as we need large‐scale, robust randomised trials to properly evaluate the effects of multicomponent interventions. Furthermore, studies should measure overall quality of life in addition to quality of life subscales and specific measures relating to key quality of life issues and symptoms experienced by this population, such as eating‐related distress. These measures need to be reported at multiple time points, appropriately and transparently, to reduce the risk of bias and allow for secondary data analyses to be conducted, such as meta‐analyses.
Authors’ Conclusions
This review mapped out in detail the combinations of intervention categories used, key features of cachexia targeted and the extent of tailoring across multicomponent interventions for adults with cancer cachexia. Only nine of the 62 multicomponent interventions targeted all four key features of cachexia, but 48 were either partly or fully tailored to individuals. Only four studies compared a multicomponent intervention to usual care and reported quality of life outcomes ‐ while three of these showed a positive effect of the intervention, high risk of bias, low sample sizes and variable outcome data remain challenges to the interpretability of these results.
Ethics Statement
The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia and Muscle [ref. 112]. The manuscript does not contain clinical studies or patient data.
Conflicts of Interest
The authors declare no conflicts of interest.
Differences Between Protocol and Review
During the screening process, it became apparent that more specific population eligibility criteria were needed to ensure reproducibility and consistency regarding which studies were included in the review and which were excluded. Therefore, we decided to use a numerical cut‐off, whereby studies with at least 75% of participants at baseline with evidence of cachexia or risk of cachexia were included. In the registered protocol, we stated that only comparative studies would be included in the analysis of clinical effectiveness. However, because of the limited number of studies for which quality of life data were available for an intervention and control arm, we decided to expand our analysis. We therefore analysed the effect of interventions on quality of life over time within study arms, to allow for the inclusion of single‐arm studies, in addition to analysing comparative studies as planned. We had planned to analyse five outcome domains: one primary (quality of life) and four secondary (energy intake, weight/body composition, metabolism and physical function). However, upon data extraction of these outcomes, it became clear that it would be too much to investigate all five of them in this systematic review. Therefore, we decided to focus only on the primary outcome of quality of life in this review.
Supplementary Materials
- Table S1.1 Search strategy for MEDLINE via Ovid. Table S1.2 Search strategy for EMBASE via Ovid. Table S1.3 Search strategy for CINAHL via EBSCO. Table S1.4 Search strategy for Cochrane Central Register of Controlled Trials. Table S1.5 Search strategy for ClinicalTrials.gov. Table S1.6 Search strategy for WHO ICTRP. Table S1.7 Search strategy for MedRxiv. (DOCX)
- Data S1 Supporting Information. (DOCX)
References
- Definition and Classification of Cancer Cachexia: An International Consensus,”. Lancet Oncology, 2011. [PubMed]
- Cancer Cachexia: Definition, Staging, and Emerging Treatments,”. Cancer Management and Research, 2020. [PubMed]
- Prevalence and Prognostic Impact of Cachexia Among Older Patients With Cancer: A Nationwide Cross‐Sectional Survey (NutriAgeCancer),”. Journal of Cachexia, Sarcopenia and Muscle, 2021. [PubMed]
- The Spectrum of Malnutrition/Cachexia/Sarcopenia in Oncology According to Different Cancer Types and Settings: A Narrative Review,”. Nutrients, 2021. [PubMed]
- Understanding and Managing Cancer‐Related Weight Loss and Anorexia: Insights From a Systematic Review of Qualitative Research,”. Journal of Cachexia, Sarcopenia and Muscle, 2015. [DOI | PubMed]
- A Systematic Review to Establish Health‐Related Quality‐Of‐Life Domains for Intervention Targets in Cancer Cachexia,”. BMJ Supportive & Palliative Care, 2016
- Non‐Steroidal Anti‐Inflammatory Drugs for Treatment of Cancer Cachexia: A Systematic Review,”. Journal of Cachexia, Sarcopenia and Muscle, 2023. [PubMed]
- Nutrition Interventions to Improve the Appetite of Adults Undergoing Cancer Treatment: A Systematic Review,”. Supportive Care in Cancer, 2020. [PubMed]
- Exercise‐Based Interventions for Cancer Cachexia: A Systematic Review of Randomised and Non‐Randomised Controlled Trials,”. Asia‐Pacific Journal of Oncology Nursing, 2023. [PubMed]
- The Psychosocial Components of Multimodal Interventions Offered to People With Cancer Cachexia: A Scoping Review,”. Asia‐Pacific Journal of Oncology Nursing, 2021. [PubMed]
- 11 S. M. Advani , P. G. Advani , H. M. VonVille , and S. H. Jafri , “Pharmacological Management of Cachexia in Adult Cancer Patients: A Systematic Review of Clinical Trials,” in BMC Cancer, vol. 18, (Washington DC, 20007, USA: Division of Oncology, Lombardi Comprehensive Cancer Center, Georgetown, University School of Medicine, 2018): 1174, http://europepmc.org/abstract/MED/30482179.30482179
- Cancer Cachexia in Adult Patients: ESMO Clinical Practice Guidelines☆,”. ESMO Open, 2021. [PubMed]
- Cancer Cachexia: Its Mechanism and Clinical Significance,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Cancer Cachexia: Developing Multimodal Therapy for a Multidimensional Problem,”. European Journal of Cancer, 2008. [PubMed]
- A Critical Review of Multimodal Interventions for Cachexia,”. Advances in Nutrition, 2021. [PubMed]
- The Promotion of Homebased Physical Activity for People With Lung Cancer and Cachexia, a Qualitative Study of Healthcare Professionals, Patients and Carers,”. Journal of Cancer Survivorship, 2023. [PubMed]
- “I Want to Get Myself as Fit as I Can and Not Die Just Yet”—Perceptions of Exercise in People With Advanced Cancer and Cachexia: A Qualitative Study,”. BMC Palliative Care, 2022. [PubMed]
- Patient Willingness to Use Digital Health Technologies: A Quantitative and Qualitative Survey in Patients With Cancer Cachexia,”. Patient Preference and Adherence, 2023. [PubMed]
- Evaluation of a Multidisciplinary Cachexia and Nutrition Support Service—The Patient and Carers Perspective,”. Journal of Patient Experience, 2021. [PubMed]
- Psycho‐Educational and Rehabilitative Intervention to Manage Cancer Cachexia (PRICC) for Advanced Patients and Their Caregivers: Lessons Learned From a Single‐Arm Feasibility Trial,”. Cancers (Basel), 2023. [PubMed]
- Feasibility of Early Multimodal Interventions for Elderly Patients With Advanced Pancreatic and Non‐Small‐Cell Lung Cancer,”. Journal of Cachexia, Sarcopenia and Muscle, 2019. [PubMed]
- Quality of Life and Symptom Burden Improve in Patients Attending a Multidisciplinary Clinical Service for Cancer Cachexia: A Retrospective Observational Review,”. Journal of Pain and Symptom Management, 2021. [PubMed]
- Top Ten Tips Palliative Care Clinicians Should Know About Cachexia,”. Journal of Palliative Medicine (, 2023. [PubMed]
- Incorporating NRSIs in Systematic Reviews,” (. 2022
- 25 B. Reeves , J. Deeks , J. Higgins , B. Shea , P. Tugwell , and G. Wells , “Chapter 24: Including Non‐Randomized Studies on Intervention Effects,” in Cochrane Handbook for Systematic Reviews of Interventions Version 6.4 (Updated August 2023), eds. J. P. T. Higgins , J. Thomas , J. Chandler , et al. (Cochrane, 2023), https://training.cochrane.org/handbook/current/chapter‐24.
- Orphan Disease Status of Cancer Cachexia in the USA and in the European Union: A Systematic Review,”. Journal of Cachexia, Sarcopenia and Muscle, 2019. [PubMed]
- An Epidemiological Survey of Cachexia in Advanced Cancer Patients and Analysis on Its Diagnostic and Treatment Status,”. Nutrition and Cancer, 2015. [DOI | PubMed]
- 28 C. Lefebvre , J. Glanville , S. Briscoe , et al., “Chapter 4: Searching for and Selecting Studies,” in Cochrane Handbook for Systematic Reviews of Interventions Version 6.4 (Updated October 2023), eds. J. P. T. Higgins , J. Thomas , J. Chandler , et al. (Cochrane, 2023), https://training.cochrane.org/handbook/current/chapter‐04#section‐4‐3‐3.
- Searching Clinical Trials Registers: Guide for Systematic Reviewers,”. BMJ, 2022. [PubMed]
- Quasi‐Experimental Study Designs Series—Paper 5: A Checklist for Classifying Studies Evaluating the Effects on Health Interventions—A Taxonomy Without Labels,”. Journal of Clinical Epidemiology, 2017. [PubMed]
- Management of Cancer Cachexia: ASCO Guideline,”. Journal of Clinical Oncology, 2020. [PubMed]
- UpSet: Visualization of Intersecting Sets,” (. 2023
- 33 J. Higgins , T. Li , and J. Deeks , “Chapter 6: Choosing Effect Measures and Computing Estimates of Effect,” in Cochrane Handbook for Systematic Reviews of Interventions Version 6.4 (updated August 2023), eds. J. P. T. Higgins , J. Thomas , J. Chandler , et al. (Cochrane, 2023), https://training.cochrane.org/handbook/current/chapter‐06.
- Effects of a 12‐Week Multimodal Exercise Intervention Among Older Patients With Advanced Cancer: Results From a Randomized Controlled Trial,”. Oncologist, 2022. [PubMed]
- A Multidisciplinary Rehabilitation Programme for Cancer Cachexia Improves Quality of Life,”. BMJ Supportive & Palliative Care, 2017
- Clinical Studies on the Treatment of Cancer Cachexia With Megestrol Acetate Plus Thalidomide,”. Chemotherapy, 2013
- Patient‐Reported Outcomes, Body Composition, and Nutrition Status in Patients With Head and Neck Cancer: Results From an Exploratory Randomized Controlled Exercise Trial,”. Cancer, 2016. [PubMed]
- Maintaining Physical Activity During Head and Neck Cancer Treatment: Results of a Pilot Controlled Trial,”. Head & Neck, 2016. [PubMed]
- A Comparison of the Effects of Medical Qigong and Standard Exercise Therapy on Symptoms and Quality of Life in Patients With Advanced Cancer,”. Supportive Care in Cancer, 2017. [PubMed]
- Dronabinol Versus Megestrol Acetate Versus Combination Therapy for Cancer‐Associated Anorexia: A North Central Cancer Treatment Group Study,”. Journal of Clinical Oncology, 2002. [PubMed]
- Clinical Study on Thalidomide Combined With Cinobufagin to Treat Lung Cancer Cachexia,”. Journal of Cancer Research and Therapeutics, 2018. [PubMed]
- A Randomized Phase III Clinical Trial of a Combined Treatment for Cachexia in Patients With Gynecological Cancers: Evaluating the Impact on Metabolic and Inflammatory Profiles and Quality of Life,”. Gynecologic Oncology, 2012. [PubMed]
- Multimodal Exercise in Older Patients With Advanced Pancreatic Cancer Undergoing First‐Line Chemotherapy: A Case Series Examining Feasibility and Preliminary Efficacy,”. European Journal of Cancer Care, 2023
- Randomized Phase III Clinical Trial of a Combined Treatment With Carnitine + Celecoxib ± Megestrol Acetate for Patients With Cancer‐Related Anorexia/Cachexia Syndrome,”. Clinical Nutrition, 2012. [PubMed]
- Early Palliative Care in Patients With Non–Small‐Cell Lung Cancer: A Randomized Controlled Trial in Southwest China,”. American Journal of Hospice & Palliative Medicine, 2022. [PubMed]
- A Randomized Phase II Study of Nutritional and Exercise Treatment for Elderly Patients With Advanced Non‐Small Cell Lung or Pancreatic Cancer: The NEXTAC‐TWO Study Protocol,”. BMC Cancer, 2019. [PubMed]
- Effect of Multimodal Intervention Care on Cachexia in Patients With Advanced Cancer Compared to Conventional Management (MIRACLE): An Open‐Label, Parallel, Randomized, Phase 2 Trial,”. Trials, 2022. [PubMed]
- A Randomized Phase II Feasibility Trial of a Multimodal Intervention for the Management of Cachexia in Lung and Pancreatic Cancer,”. Journal of Cachexia, Sarcopenia and Muscle, 2017. [PubMed]
- Cancer Cachexia: Rationale for the MENAC (Multimodal‐Exercise, Nutrition and Anti‐Inflammatory Medication for Cachexia) Trial,”. BMJ Supportive & Palliative Care, 2018
- Chemoradiotherapy in Elderly Patients With Advanced Head and Neck Cancer Under Intensive Nutritional Support,”. Asia‐Pacific Journal of Clinical Oncology, 2015. [PubMed]
- Phase I/II Trial of Formoterol Fumarate Combined With Megestrol Acetate in Cachectic Patients With Advanced Malignancy,”. Supportive Care in Cancer, 2014. [PubMed]
- Clinical Outcomes and Contributors to Weight Loss in a Cancer Cachexia Clinic,”. Journal of Palliative Medicine, 2011. [PubMed]
- From Prehab to Rehab: Nutritional Support for People Undergoing Pancreatic Cancer Surgery,”. Journal of Human Nutrition and Dietetics, 2023. [PubMed]
- Treatment of Malnutrition Decreases Complication Rates and Shortens the Length of Hospital Stays in a Radiation Oncology Department,”. Strahlentherapie und Onkologie, 2018. [PubMed]
- Study Protocol for a Randomized Trial of a Supportive Care Mobile Application to Improve Symptoms, Coping, and Quality of Life in Patients With Advanced Non‐Small Cell Lung Cancer,”. Frontiers in Psychology, 2023
- Swallowing Therapy and Progressive Resistance Training in Head and Neck Cancer Patients Undergoing Radiotherapy Treatment: Randomized Control Trial Protocol and Preliminary Data,”. Acta Oncology (Madr), 2017
- Rationale and Design of the Adapted Physical Activity in Advanced Pancreatic Cancer Patients (APACaP) GERCOR (Groupe Coopérateur Multidisciplinaire en Oncologie) Trial: Study Protocol for a Randomized Controlled Trial,”. Trials, 2015. [PubMed]
- Randomized Double‐Blind Clinical Trial of Combined Treatment With Megestrol Acetate Plus Celecoxib Versus Megestrol Acetate Alone in Cachexia‐Anorexia Syndrome Induced by GI Cancers,”. Supportive Care in Cancer, 2018. [PubMed]
- Providing Nutritional Support to Patients With Thoracic Cancer: Findings of a Dedicated Rehabilitation Service,”. Respiratory Medicine, 2013. [PubMed]
- Pilot Randomized, Controlled, Preoperative Intervention for Nutrition Trial in Head and Neck Cancer,”. Head & Neck, 2023. [PubMed]
- Feasibility of a Multimodal Exercise, Nutrition, and Palliative Care Intervention in Advanced Lung Cancer,”. BMC Cancer, 2021. [PubMed]
- Nutritional Rehabilitation in Patients With Advanced Head and Neck Cancer Receiving Radiation Therapy.”
- Nutritional Counseling for Head and Neck Cancer Patients Undergoing (Chemo) Radiotherapy—A Prospective Randomized Trial,”. Frontiers in Nutrition, 2019. [PubMed]
- Anamorelin Combined With Physical Activity, and Nutritional Counseling for Cancer‐Related Fatigue: A Preliminary Study,”. Supportive Care in Cancer, 2022. [PubMed]
- Insulin Treatment in Cancer Cachexia: Effects on Survival, Metabolism, and Physical Functioning,”. Clinical Cancer Research, 2007. [PubMed]
- A Randomised Feasibility Study of EPA and Cox‐2 Inhibitor (Celebrex) Versus EPA, Cox‐2 Inhibitor (Celebrex), Resistance Training Followed by Ingestion of Essential Amino Acids High in Leucine in NSCLC Cachectic Patients—ACCeRT Study,”. BMC Cancer, 2011. [PubMed]
- Impact of Fish Oil and Melatonin on Cachexia in Patients With Advanced Gastrointestinal Cancer: A Randomized Pilot Study,”. Nutrition, 2005. [PubMed]
- Feasibility of a Multimodal Intervention on Malnutrition in Patients With Lung Cancer During Primary Anti‐Neoplastic Treatment,”. Clinical Nutrition, 2021. [PubMed]
- Early Specialized Palliative Care for Patients With Metastatic Lung Cancer Receiving Chemotherapy: A Feasibility Study of a Nurse‐Led Screening‐Triggered Programme,”. Japanese Journal of Clinical Oncology, 2022. [PubMed]
- “EXHALE”: Exercise as a Strategy for Rehabilitation in Advanced Stage Lung Cancer Patients: A Randomized Clinical Trial Comparing the Effects of 12 Weeks Supervised Exercise Intervention Versus Usual Care for Advanced Stage Lung Cancer Patients,”. BMC Cancer, 2013. [PubMed]
- Establishing a Cancer Nutrition Rehabilitation Program (CNRP) for Ambulatory Patients Attending an Australian Cancer Center,”. Supportive Care in Cancer, 2011. [PubMed]
- Defining a New Model of Interdisciplinary Cancer Cachexia Care in Regional Victoria, Australia,”. Supportive Care in Cancer, 2020. [PubMed]
- Randomized Phase III Clinical Trial of Five Different Arms of Treatment in 332 Patients With Cancer Cachexia,”. Oncologist, 2010. [PubMed]
- An Eicosapentaenoic Acid Supplement Versus Megestrol Acetate Versus Both for Patients With Cancer‐Associated Wasting: A North Central Cancer Treatment Group and National Cancer Institute of Canada Collaborative Effort,”. Journal of Clinical Oncology, 2004. [PubMed]
- Nutrition Support Improves Patient Outcomes, Treatment Tolerance and Admission Characteristics in Oesophageal Cancer,”. Clinical Oncology, 2005. [PubMed]
- Effects of Celecoxib, Medroxyprogesterone, and Dietary Intervention on Systemic Syndromes in Patients With Advanced Lung Adenocarcinoma: A Pilot Study,”. Journal of Pain and Symptom Management. Elsevier Inc., 2004. [PubMed]
- Early Nutritional Support in Non‐Metastatic Stage IV Oral Cavity Cancer Patients Undergoing Adjuvant Concurrent Chemoradiotherapy: Analysis of Treatment Tolerance and Outcome in an Area Endemic for Betel Quid Chewing,”. Supportive Care in Cancer, 2012. [PubMed]
- Effect of Combined Citrulline Supplementation and Resistance Training Intervention on Muscle Mass, Mitochondrial Respiratory Capacity, and ccf‐mtDNA in Cancer Cachexia Patients: Research Proposal for a Randomized Control Trial,”. National Journal of Physiology, Pharmacy and Pharmacology, 2020
- Effects of a Palliative Care Intervention on Clinical Outcomes in Patients With Advanced Cancer The Project ENABLE II Randomized Controlled Trial,”
- PRN‐jRCTs041210053. 2021 [Cited 2024 Jun 27]. Randomized Phase II Study of Anamorelin Hydrochloride With Nutritional and Exercise Treatment for Cancer Cachexia Among Elderly Patients With Advanced Non‐Small Cell Lung and Pancreatic Cancer (NEXTAC‐III Study),”
- A Prospective Randomized Controlled Trial to Study the Impact of a Nutrition‐Sensitive Intervention on Adult Women With Cancer Cachexia Undergoing Palliative Care in India,”. Integrative Cancer Therapies, 2017. [PubMed]
- Home Parenteral Nutrition Increases Fat Free Mass in Patients With Incurable Gastrointestinal Cancer. Results of a Randomized Controlled Trial,”. Clinical Nutrition, 2019. [PubMed]
- Multimodal Treatment Strategy for Cancer Cachexia,” NCT00625742, (. 2008
- Lenalidomide for Lean Body Mass and Muscle Strength in Inflammatory Cancer Cachexia Syndrome,” NCT01127386, (. 2010
- Megestrol Acetate With or Without Mirtazapine in Treating Cancer Patients With Weight Loss or Loss of Appetite,” NCT01501396, (. 2011
- Nutrition Support to Improve Outcomes in Patients With Unresectable Pancreatic Cancer,” NCT02681601, (. 2016
- Survival With Olanzapine in Patients With Locally Advanced or Metastatic Upper Gastrointestinal and Lung Cancer,” NCT06338683, (. 2024
- Exercise Intervention for Patients With Cancer Cachexia: Effects of a 12‐Week Program and One‐Year Follow‐up,” NCT06323733,
- The Effect of Exercise and Diet on Quality of Life in Patients With Incurable Cancer of Esophagus and Stomach (RADICES),” NCT06138223, (. 2023
- Physical Training for Elderly Cancer Patients With Cachexia,” NCT05915325, (. 2023
- 91 D. Croagh , “Can “Top‐Up” Small Bowel Tube Feeding, With Intensive Dietitian Support Delivered via Telehealth, Improve Quality of Life in Patients With Advanced Pancreatic Cancer?” ACTRN12624000084583 (World Health Organisation International Clinical Trials Registry Platform database, 2024), https://www.cochranelibrary.com/central/doi/10.1002/central/CN‐02658821/full?highlightAbstract=actrn12624000084583.
- Adherence and Impact of an Oral Nutritional Supplement Enriched in Leucine, EVOO, EPA and DHA, and Beta‐Glucans on the Coverage of Energy and Protein Requirements in Patients With Cancer and Malnutrition: Alisenoc Study,”. Nutrition, 2024. [PubMed]
- Promotion of Behavioral Change and the Impact on Quality of Life in Elderly Patients With Advanced Cancer: A Physical Activity Intervention of the Multimodal Nutrition and Exercise Treatment for Advanced Cancer Program,”. Asia‐Pacific Journal of Oncology Nursing, 2018. [PubMed]
- Power Comparisons and Clinical Meaning of Outcome Measures in Assessing Treatment Effect in Cancer Cachexia: Secondary Analysis From a Randomized Pilot Multimodal Intervention Trial,”. Frontiers in Nutrition, 2021. [PubMed]
- Psychoeducational and Rehabilitative Intervention to Manage Cancer Cachexia (PRICC) for Patients and Their Caregivers: Protocol for a Single‐Arm Feasibility Trial,”. BMJ Open, 2021
- Engaging the Older Cancer Patient; Patient Activation Through Counseling, Exercise and Mobilization—Pancreatic, Biliary Tract and Lung Cancer (PACE‐Mobil‐PBL)—Study Protocol of a Randomized Controlled Trial,”. BMC Cancer, 2018. [PubMed]
- Cancer Nutrition and Rehabilitation ‐ Its Time has Come!,”. Current Oncology, 2008. [PubMed]
- A Prospective Evaluation of an Interdisciplinary Nutrition–Rehabilitation Program for Patients With Advanced Cancer,”. Current Oncology, 2013. [PubMed]
- Weight Changes Correlate With Alterations in Subjective Physical Function in Advanced Cancer Patients Referred to a Specialized Nutrition and Rehabilitation Team,”. Supportive Care in Cancer, 2013. [PubMed]
- Effect of an Interdisciplinary Rehabilitation Program on Quality of Life in Patients With Head and Neck Cancer: Review of Clinical Experience,”. Head & Neck, 2013. [PubMed]
- Randomized Phase III Clinical Trial of Five Different Arms of Treatment for Patients With Cancer Cachexia: Interim Results,”. Nutrition, 2008. [PubMed]
- The Project ENABLE II Randomized Controlled Trial to Improve Palliative Care for Rural Patients With Advanced Cancer: Baseline Findings, Methodological Challenges, and Solutions,”. Palliative & Supportive Care, 2009. [PubMed]
- Development and Validation of Questionnaires for Eating‐Related Distress Among Advanced Cancer Patients and Families,”. Journal of Cachexia, Sarcopenia and Muscle, 2023. [PubMed]
- A Systematic Review to Establish Health‐Related Quality‐Of‐Life Domains for Intervention Targets in Cancer Cachexia,”. BMJ Supportive & Palliative Care, 2016
- Development of the EORTC QLQ‐CAX24, a Questionnaire for Cancer Patients With Cachexia,”. Journal of Pain and Symptom Management, 2017. [PubMed]
- Ponsegromab for the Treatment of Cancer Cachexia,”. New England Journal of Medicine, 2024. [PubMed]
- Cancer Cachexia Update in Head and Neck Cancer: Definitions and Diagnostic Features,”. Head & Neck, 2015. [PubMed]
- Understanding Cancer Cachexia and Its Implications in Upper Gastrointestinal Cancers,”. Current Treatment Options in Oncology, 2022. [PubMed]
- Cancer‐Associated Cachexia,”. Nature Reviews Disease Primers, 2018
- Screening the Nutritional Status in Oncology: A Preliminary Report on 1,000 Outpatients,”. Supportive Care in Cancer, 2009. [PubMed]
- Prevalence of Cachexia in Cancer Patients,”. European Journal of Cancer Care, 2023
- Ethical Guidelines for Publishing in the Journal of Cachexia, Sarcopenia and Muscle: Update 2021,”. Journal of Cachexia, Sarcopenia and Muscle, 2021. [PubMed]
