An integrated approach to identify bimodal genes associated with prognosis in câncer
Abstract
Bimodal gene expression (where a gene expression distribution has two maxima) is associated with phenotypic diversity in different biological systems. A critical issue, thus, is the integration of expression and phenotype data to identify genuine associations. Here, we developed tools that allow both: i) the identification of genes with bimodal gene expression and ii) their association with prognosis in cancer patients from The Cancer Genome Atlas (TCGA). Bimodality was observed for 554 genes in expression data from 25 tumor types. Furthermore, 96 of these genes presented different prognosis when patients belonging to the two expression peaks were compared. The software to execute the method and the corresponding documentation are available at the Data access section.
Article type: Research Article
Keywords: Cancer, gene expression, bimodal distribution, Gaussian Mixture Model, survival analysis
Affiliations: Universidade Federal do Rio Grande do Norte (UFRN), Metrópole Digital, Centro Multiusuário de Bioinformática, Natal, RN, Brazil.; Universidade Federal de Rondônia, Departamento de Matemática e Estatística, Ji-Parana, RO, Brazil.; Universidade de São Paulo, Departamento de Genética e Biologia Evolutiva, São Paulo, SP, Brazil.; Universidade Federal do Rio Grande do Norte (UFRN), Instituto do Cérebro, Natal, RN, Brazil.; Sichuan University, West China Hospital, Institutes for Systems Genetics, Chengdu, China.; Universidade Federal do Rio Grande do Norte (UFRN), Centro de Tecnologia, Departamento de Engenharia Biomédica, Natal, RN, Brazil.
License: CC BY 4.0 This is an open-access article distributed under the terms of the Creative Commons Attribution License
Article links: DOI: 10.1590/1678-4685-GMB-2021-0109 | PubMed: 34617951 | PMC: PMC8495773
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.5 MB)
Introduction
Studies on gene expression and regulation have been directed towards a better understanding of a diverse range of biological processes, including initial differentiation in the embryonic stage and changes in health and disease that occur during life. These patterns of gene expression have been extensively used to establish associations between phenotypes and genetic/epigenetic information (ref. Boyle et al., 2017; ref. Young et al., 2019). The challenges for such studies are significant, however, and the identification of expression signatures enriched with genuine phenotypic associations is particularly welcome. In that aspect, bimodal gene expression is an interesting pattern since their identification capitalizes on the availability of genetic and phenotypic data from large cohorts of samples and each mode can, in theory, correspond to a phenotypic state of the system.
Few previous studies have searched for bimodality in large-scale gene expression data (ref. Bessarabova et al., 2010; ref. Mason et al., 2011; ref. Shalek et al., 2013) and causes for such bimodality have been discussed, including: i) differential action of transcription factors (ref. Ochab-Marcinek and Tabaka; 2010), ii) regulation by microRNAs (ref. Bosia et al., 2017; ref. Del Giudice et al., 2018); iii) regulation by circular RNA (ref. Hu and Zhou; 2018) and even iv) stochastic events (ref. Samoilov et al.,
2005). For an extensive review of the different methods developed for detection of bimodality, please see ref. Moody et al.
(2019).
The identification of genes with a bimodal expression pattern, together with sample stratification, can be used to identify important clinical and therapeutic targets in different cancer types (ref. Floristan et al.,
2020). Furthermore, this process can reveal molecular signatures that distinguish tumor subtypes, which would contribute to a better clinical understanding of the biological characteristics of cancer. To be clinically useful, a bimodal pattern must exhibit a clear separation between the two groups and have significant sample sizes (ref. Han et al., 2013).
The term “bimodal expression” is related in biology to two distinct groups of continuous values of gene expression for the same gene. As discussed by ref. Moody et al. (2019), genes presenting a bimodal pattern present two modes of expression in the same population. Statistically, the set of continuous values of lower and higher expression has a more consistent definition as a mixture of Gaussian distributions.
Here, a computational protocol was developed to identify, in a genome-wide context, genes with bimodal expression patterns associated with prognosis in cancer samples. To prove the applicability and robustness of our method, we used this new tool to identify genes with bimodal expression in 25 tumor types whose expression data is available from The Cancer Genome Atlas (TCGA). Finally, we made use of the availability of clinical data from TCGA to find 96 genes, among the ones with bimodal gene expression, in which patients in the two expression peaks showed different prognosis. The software to execute the method and the corresponding documentation are available at the Data access section.
Material and Methods
Data samples
Expression and clinical data from 25 different tumor types were obtained from The Cancer Genome Atlas (TCGA) project through the ref. Genomic Data Commons Data Portal. Expression data for 24,456 genes were evaluated to identify genes with a bimodal distribution, using Fragments by Exon Kilobase per Millions of Mapped Fragments (FPKM) values. For survival analysis, clinical information was extracted from cBioPortal for Cancer Genomics.
Detection of bimodality
The detection of bimodality involves a three-step process, configured by seven parameters, listed below:
minExpression – defines the minimum expression value in the analysis. It prevents noise in readings of low expression value from influencing the correct detection of peaks, particularly at values close to zero. This parameter must be appropriate to the type of measurement unit of expression to be used. Its default value is 0.02 FPKM;
minSampleSize – defines the minimum sample size in the analysis. Datasets with a number of samples smaller than this value do not undergo any processing, being immediately discarded. Its default value is 50 samples.
MinClusterSize – defines the minimum size, in relation to the total number of samples, that a cluster must have to be considered as one of the bimodal clusters. This aims to discard groups of relatively small sample populations composed of outliers, capable of altering the density profile to the point of being mistaken as a peak, especially when they occur in the upper tail of the distribution. Its default value is 10% of the total samples considered.
Threshold Up – defines the minimum difference between the points detected as adjacent peaks and valleys on the density curve. If the difference between them is less than the parameter value, this oscillation in the density graph will be disregarded in the detection process. Its default value is 10% of the maximum density value.
Threshold Down – peaks whose density values are below this threshold will be discarded. This aims to rule out small fluctuations in the expression values that normally occur in the upper tail of the distribution, which cause the density to fluctuate widely in this region. Its default value is 20% of the maximum density value.
Smoothing factor – this parameter mitigates the variations in the derivatives curve to make detection less sensitive. Its default value is ‘true’.
useLog – this parameter defines whether the expression values will be considered in their original form or whether they should be transformed into a base 2 or base 10 logarithm before analysis. This helps to improve the sensitivity of the algorithm, particularly when the range of expression values is quite wide, which causes the upper tail of the density curve to flatten, making the peak detection process more difficult. An example of this difference in the density profile can be seen in Figure S1 where the same dataset has its density curve plotted with and without the log10 transformation of the expression values. Its default value is “none”.
The three steps are:
a) Peak detection:
In this step, the initial screening of candidate genes for bimodality is performed using the density derivative. First, the density of the expression distribution of each gene is calculated using the density function of the R stats package ref. R Core Team (2021), with the “nrd0” method to calculate the smoothing bandwidth. This method was chosen specifically because it is less precise than methods like the Sheater Jones bandwidth, guaranteeing only the detection of large fluctuations in density.
Next, the first density derivative is calculated, which undergoes a smoothing process designed to decrease the sensitivity of peak detection. For this purpose, the smooth.spline function of the R stats package was used (ref. R Core Team, 2021), with the parameter defined by the smoothing factor. Derivative values tending to zero indicate a peak or valley. The threshold Up and Down parameters are then applied, which will define which peaks are relevant. As a result, this process returns the estimated number of peaks, which will become variable k in the subsequent step.
b) Clustering:
A data model that presents a characteristic of bimodality can be considered as the overlap of probabilistic models that represent two distinct subpopulations. In this way, we can consider bimodal distributions as a model of mixing Gaussian data (Gaussian Mixture Models – GMM) and use their specific algorithms to perform the identification and separation of these subpopulations (ref. Titterington et al., 1986).
To perform the classification based on GMM, we used the Mclust function of the R mclust package (ref. Scrucca et al., 2016), which performs a clustering of data using the expectation maximization (EM) technique, performing successive grouping operations and comparing groups with a Gaussian distribution (ref. Hasselblad, 1966; ref. Gelman et al., 2013). This process can either infer the number of clusters expected in the distribution or start from a ‘k’ parameter that will designate the number of desired clusters. In our case, we already have such information, the number of clusters will be equal to the number of peaks, estimated in the previous step, plus one. Consequently, the algorithm will group the data in k effective clusters, plus an additional cluster (k + 1), that will contain all data points with low affinity to the main clusters.
This process returns, in addition to the clustering of samples in k + 1 clusters, an uncertainty index related to such classification. Arbitrarily, only the samples whose reliability in the classification received an index higher than 46% are maintained. This low rigor in the use of uncertainty values is justified because a distribution of expression indexes tends to be closer to a Poisson than to a Gaussian (ref. Marioni et al., 2008; ref. Langmead et al., 2010; ref. Wang et al., 2010), and an excessive rigor in the use of such reliability would cause large disposal of samples. The result of the process is illustrated in Figure S2. After discarding clusters smaller than MinClusterSize (m) and samples contained in cluster k + 1, the remaining samples are passed on to the third phase of the process.
c) Peak confirmation
The samples contained in the largest k-m clusters are subjected to a new peak detection process, identical to step 1, to confirm the initial screening. If the Peak Detection process continues to identify a bimodality, as is the case shown in Figure S3A, that gene is classified as bimodal. Otherwise, the gene is discarded from the bimodal gene pool. Such a situation can be seen in Figure S3B, where the bimodality existing in the original dataset no longer can be identified when the filtered samples are used.
Survival Analysis
To verify if individuals belonging to the two different peaks of expression in a bimodal gene presented a significant difference in survival curves, we performed an analysis using the clinical data from ref. CBioPortal, obtained as indicated above, and the Survival package in R (ref. Therneau, 2021).
The samples identified as peak 1 and peak 2 from 554 genes with bimodal distribution were selected and Kaplan-Meier curves were evaluated with a significance level of 5% and 1% using the log-rank test. Kaplan-Meier curves were plotted using the ggplot2 package (ref. Wickham,
2016). All computacional analysis were done using ref. RStudio IDE.
Data availability
The computational pipeline to execute the method is freely available at https://github.com/LabBiosystemUFRN/Bimodality_Genes.
Results
Development of a method to identify genes with bimodality in gene expression data
In our method, described in Figure 1A, the identification of gene expression bimodality involves a density function, which can be used to analyse the expression values (in FPKM) for all human genes in any set of samples. Using a computational algorithm in R, the maximum and minimum points in the expression density curve of each gene is defined by identifying where the values of its derivative curve change its value signal (Figure 1B). To avoid possible noises in the stratification of samples, data points below 0.02 FPKM were excluded. To identify robust distributions concerning the difference in bimodality peaks, two thresholds were established: (i) a maximum value of 10% of density, used specifically to eliminate small ripples in the upper tail of the distribution curve, which could indicate irrelevant peaks; (ii) a 5% difference between the peak and the density valley, guaranteeing significant differences in the bimodality peaks. All these parameters are shown in Figure 1B in a schematic bimodal distribution of a hypothetical gene.
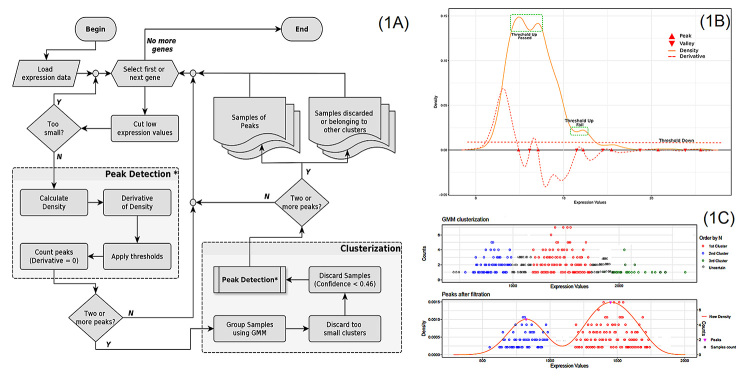
After the identification of genes with a bimodal expression pattern, we next performed sample stratification to identify samples belonging to the first and second peaks. Assuming that each peak of the density curve represents the fashion of a subpopulation with different expression features, we can consider that the distribution of expression values, of genes with bimodality characteristics, constitutes a mixture model. Therefore, to ensure the reliable selection of samples according to the selected bimodal genes, an analysis based on the Gaussian Mixture Models (GMM) probabilistic model was introduced in our computational protocol. A schematic view of the sample stratification step is shown in Figure 1C. Finally, stratified samples were submitted to the step of bimodality detection again. Only genes that remained with a bimodal pattern after sample stratification are listed in our final results.
Identification of bimodal genes using data from 25 different tumor types from TCGA
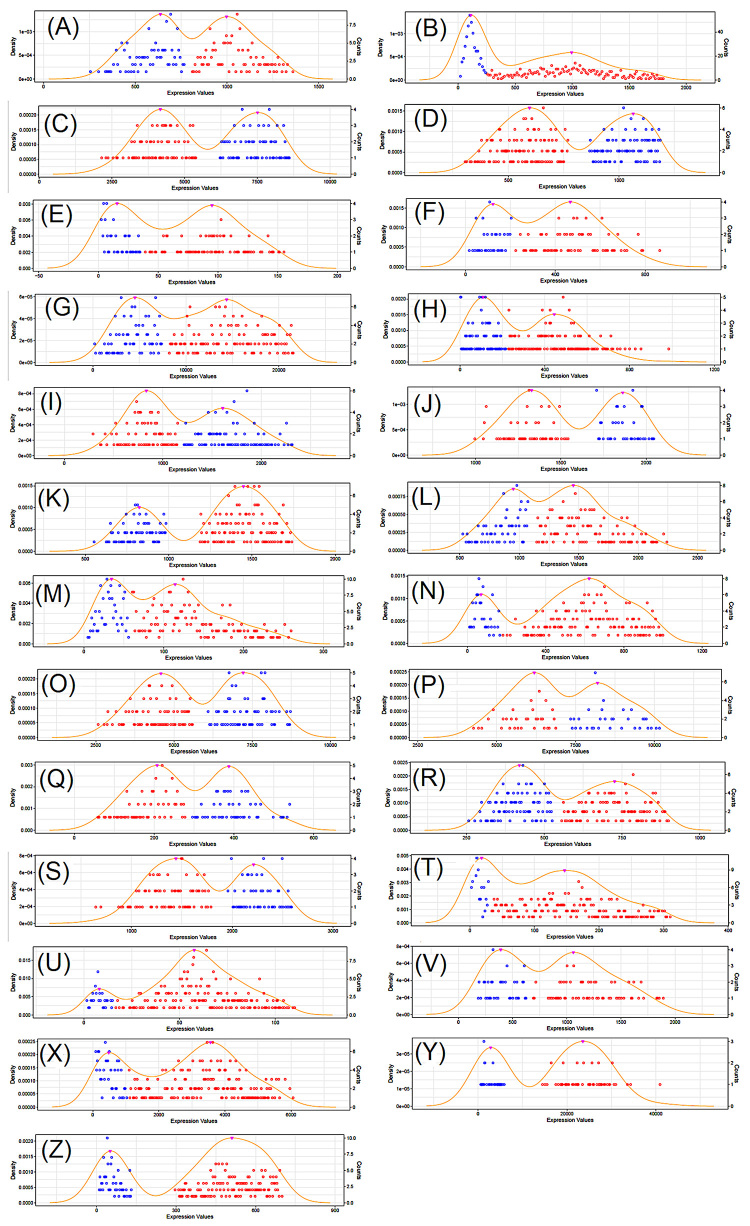
To illustrate the use of our method, we have collected all gene expression and clinical data from TCGA for the following tumors: BLCA, BRCA, CESC, COADREAD, ESCA, GBM, HNSC, KIRC, KIRP, LAML, LGG, LIHC, LUAD, LUSC, OV, PAAD, PCPG, PRAD, SARC, SKCM, STAD, TGCT, THCA, THYM, UCEC. These tumor types were selected because they have a minimum number of 100 patients in their respective cohorts. A total of 554 unique genes was identified as having a bimodal pattern for at least one tumor type. Table 1 shows the numbers of genes identified as bimodal for each tumor type (listed at Table S1) and Figure 2 shows the bimodal pattern of expression for 25 genes, arbitrarily selected, one for each tumor type.
Table 1 –: Number of genes showing bimodality for each tumor type.
| Tumor | Bimodal genes | Tumor | Bimodal genes | Tumor | Bimodal genes |
|---|---|---|---|---|---|
| BLCA | 8 | LAML | 18 | SARC | 6 |
| BRCA | 13 | LGG | 90 | SKCM | 14 |
| CESC | 14 | LIHC | 11 | STAD | 3 |
| COADREAD | 22 | LUAD | 7 | TGCT | 77 |
| ESCA | 8 | LUSC | 6 | THCA | 55 |
| GBM | 29 | OV | 5 | THYM | 181 |
| HNSC | 5 | PAAD | 12 | UCEC | 11 |
| KIRC | 15 | PCPG | 53 | ||
| KIRP | 11 | PRAD | 9 |
We found 46 genes showing bimodal expression in more than one tumor type (Table 2). The ones most frequently found were SLC35E2, EIF1AY and RPS27, which have a bimodal pattern in 19, 10 and 10 tumor types, respectively. In this list of genes, chromosomal distribution is significantly biased toward the Y chromosome (p<10-5), as observed in Table 2.
Table 2 –: List of genes showing bimodality for more than one tumor type.
| Gene | Number | Chr | Gene | Number | Chr |
|---|---|---|---|---|---|
| RPS27 | 10 | 1 | TP53 | 3 | 17 |
| GSTM1 | 4 | 1 | PLCD3 | 2 | 17 |
| LQK1 | 3 | 1 | RPS28 | 4 | 19 |
| KHDRBS1 | 2 | 1 | C19orf46 | 2 | 19 |
| RPF1 | 2 | 1 | FKBP1AP1 | 2 | 19 |
| SLC35E2 | 19 | 1 | ZNF304 | 2 | 19 |
| C2orf43 | 2 | 2 | GSTT1 | 2 | 22 |
| SPAG16 | 2 | 2 | ZDHHC15 | 2 | X |
| MLH1 | 2 | 3 | AWAT1 | 2 | X |
| ZNF502 | 2 | 3 | MAGEA6 | 2 | X |
| RPL9 | 4 | 4 | RPS26P11 | 2 | X |
| ERAP2 | 3 | 5 | CYorf15A | 9 | Y |
| PDCD2 | 2 | 6 | EIF1AY | 10 | Y |
| CHMP7 | 2 | 8 | DDX3Y | 8 | Y |
| MTAP | 2 | 9 | UTY | 8 | Y |
| CSNK2A1P | 2 | 11 | KDM5D | 7 | Y |
| XRRA1 | 2 | 11 | RPS4Y1 | 7 | Y |
| SCNN1A | 3 | 12 | ZFY | 8 | Y |
| CEP290 | 2 | 12 | USP9Y | 4 | Y |
| CHFR | 2 | 12 | PRKY | 3 | Y |
| SNRPN | 2 | 15 | TMSB4Y | 3 | Y |
| TUBGCP4 | 2 | 15 | NLGN4Y | 2 | Y |
| ZNF597 | 2 | 16 | TTTY15 | 3 | Y |
Patients in different expression peaks have different prognosis
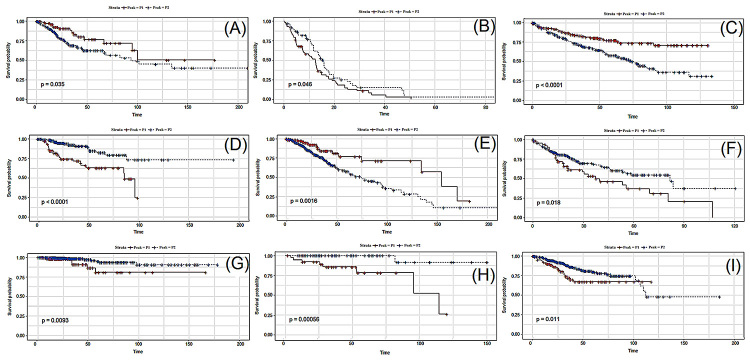
We wondered whether patients belonging to the two different peaks of the bimodal distribution would present different prognosis, as evaluated by survival curves in a Kaplan-Meyer plot. All genes identified as having a bimodal distribution (Table 1) were tested. A total of 96 genes were identified as having their bimodal pattern significantly (p<0.01) associated with prognosis (samples belonging to the first peak having either a better or worse prognosis when compared to samples belonging to the second peak). If a threshold of p<0.05 is used, 176 genes are identified as associated with prognosis. Figure S4 shows the expression plots, reporting the bimodality, for all 96 genes found to have a bimodal expression pattern.
Figure 3 shows the respective Kaplan-Meyer plots for few genes that showed significant differences in survival between patients belonging to peaks 1 and 2. Figure S5 shows the Kaplan-Meyer plot for all 96 genes associated with prognosis.
Discussion
A new genome-wide method is presented to identify genes with bimodal patterns of expression using GMM analysis (ref. Titterington et al.,
1986) for the stratification of samples. GMM has been previously used in the analysis of gene expression data (ref. Ficklin et al.,
2017; ref. Golumbeanu et al., 2019; ref. Mirzal, 2020) but to our knowledge this is the first application of such a method for the identification of genes with bimodal expression patterns.
The applicability of the method is shown by using gene expression and clinical data for 25 tumor types available from TCGA. We identified 554 unique genes with bimodal gene expression (Table 1). Forty-six of them were identified as bimodal in more than one tumor type. Several of them have been reported previously as having a bimodal expression pattern. One of them, ERAP2, has been found by ref. Mason et. al. (2011) to have bimodal gene expression in human skeletal muscle. The same report (ref. Mason ) found that GSTM1 has a bimodal expression pattern in muscle tissue. Other genes include RPS27, found by ref. Floristan ) to have bimodal expression in several tumor types, and USP9AY, found to show bimodality in endometrium (ref. Bhat et al.,
2019). Interestingly, among the 46 genes with bimodality in more than one tumor type, 12 are mapped to the Y chromosome (p<10-5), an unexpected observation due to the low gene density in this chromosome. As reviewed by ref. Lau (2020), some genes on the Y chromosome have dosage-sensitive functions, which might be related to a bimodal expression pattern. This remains to be further explored.
Ninety six, out of 554 genes with bimodal gene expression in all tumor types analyzed here, were identified as having differential prognosis when patients belonging to the two different modes were compared. Expression of several genes identified by us are known predictors of clinical outcome in different tumor types including ANXA1 (ref. Gibbs and Vishwanatha, 2017), FOXJ3 (ref. Ban et al., 2013; ref. Ma et al., 2016) and CDC25 (ref. Liu
et al., 2019, ref. 2020), among many others. However, the great majority of these reports only associate overall expression with prognosis. Here, on the other hand, we associate the bimodal expression pattern with prognosis. To our knowledge, only ref. Floristan et al. (2020) have associated the bimodal expression pattern of RPS27 with clinical outcome in several tumor types, a gene also observed in our data. This makes our analysis the first one, to our knowledge, to explore the association between the modes of gene expression distribution with prognosis in a genome-wide context.
Several issues should be considered in the interpretation of our results. For example, cellular heterogeneity within samples in a given cohort is a factor that can generate genes with bimodal expression. In our case, this is minimized by the fact that TCGA samples are selected for high tumor cell content but this issue should be critically considered when more heterogeneous cohorts are analyzed. Furthermore, clinical and/or biological features should be considered when interpreting data from our method. For example, in cancer studies one should be careful with cohort heterogeneity regarding staging and progression, among many other clinical features.
We envisage that our method will be a useful tool for the genome-wide identification of genes with bimodal pattern of expression. The software to execute the method and the corresponding documentation are available at the Data access section.
References
- JY Ban, HJ Park, SK Kim, JW Kim, YA Lee, IA Choi, JH Chung, SJ Hong. Association of forkhead box J3 (FOXJ3) polymorphisms with rheumatoid arthritis. Mol Med Rep, 2013. [PubMed]
- M Bessarabova, E Kirillov, W Shi, A Bugrim, Y Nikolsky, T Nikolskaya. Bimodal gene expression patterns in breast cancer. BMC Genomics, 2010
- MA Bhat, JB Sharma, KK Roy, J Sengupta, D Ghosh. Genomic evidence of Y chromosome microchimerism in the endometrium during endometriosis and in cases of infertility. Reprod Biol Endocrinol, 2019
- C Bosia, F Sgrò, L Conti, C Baldassi, D Brusa, F Cavallo, FD Cunto, E Turco, A Pagnani, R Zecchina. RNAs competing for microRNAs mutually influence their fluctuations in a highly non-linear microRNA-dependent manner in single cells. Genome Biol, 2017. [PubMed]
- EA Boyle, YI Li, JK Pritchard. An expanded view of complex traits: From polygenic to omnigenic. Cell, 2017. [PubMed]
- M Del Giudice, S Bo, S Grigolon, C Bosia. On the role of extrinsic noise in microRNA-mediated bimodal gene expression. PLoS Comput Biol, 2018. [PubMed]
- SP Ficklin, LJ Dunwoodie, WL Poehlman, C Watson, KE Roche, FA Feltus. Discovering condition-specific gene co-expression patterns using Gaussian mixture models: A cancer case study. Sci Rep, 2017. [PubMed]
- A Floristan, L Morales, D Hanniford, C Martinez, E Castellano-Sanz, I Dolgalev, A Ulloa-Morales, ME Vega-Saenz, U Moran, F Darvishian. Functional analysis of RPS27 mutations and expression in melanoma. Pigment Cell Melanoma Res, 2020. [PubMed]
- A Gelman, DB Rubin, JB Carlin, HS Stern. Bayesian Data Analysis, 2013
- LD Gibbs, JK Vishwanatha. Prognostic impact of AnxA1 and AnxA2 gene expression in triple-negative breast cancer. Oncotarget, 2017. [PubMed]
- M Golumbeanu, S Desfarges, C Hernandez, M Quadroni, S Rato, P Mohammadi, A Telenti, N Beerenwinkel, A Ciuffi. Proteo-transcriptomic dynamics of cellular response to HIV-1 infection. Sci Rep, 2019. [PubMed]
- F Han, J Faraco, XS Dong, HM Ollila, L Lin, J Li, P An, S Wang, KW Jiang, ZC Gao. Genome wide analysis of narcolepsy in China implicates novel immune loci and reveals changes in association prior to versus after the 2009 H1N1 influenza pandemic. PLoS Genet, 2013. [PubMed]
- V Hasselblad. Estimation of parameters for a mixture of normal distributions. Technometrics, 1966
- Q Hu, T Zhou. EIciRNA-mediated gene expression: tunability and bimodality. FEBS Lett, 2018. [PubMed]
- B Langmead, KD Hansen, JT Leek. Cloud-scale RNA-sequencing differential expression analysis with Myrna. Genome Biol, 2010. [PubMed]
- YC Lau. Y chromosome in health and diseases. Cell Biosci, 2020. [PubMed]
- K Liu, R Lu, Q Zhao, J Du, Y Li, M Zheng, S Zhang. Association and clinicopathologic significance of p38MAPK-ERK-JNK-CDC25C with polyploid giant cancer cell formation. Med Oncol, 2019
- K Liu, M Zheng, R Lu, J Du, Q Zhao, Z Li, Y Li, S Zhang. The role of CDC25C in cell cycle regulation and clinical cancer therapy: a systematic review. Cancer Cell Int, 2020. [PubMed]
- W Ma, Q Yu, J Jiang, X DU, L Huang, L Zhao, QI Zhou. miR-517a is an independent prognostic marker and contributes to cell migration and invasion in human colorectal cancer. Oncol Lett, 2016. [PubMed]
- JC Marioni, CE Mason, SM Mane, M Stephens, Y Gilad. RNA-seq: an assessment of technical reproducibility and comparison with gene expression arrays. Genome Res, 2008. [PubMed]
- CC Mason, RL Hanson, V Ossowski, L Bian, LJ Baier, J Krakoff, C Bogardus. Bimodal distribution of RNA expression levels in human skeletal muscle tissue. BMC Genomics, 2011
- A Mirzal. Statistical Analysis of Microarray Data Clustering using NMF, Spectral Clustering, Kmeans, and GMM. IEEE/ACM Trans Comput Biol Bioinform, 2020
- L Moody, S Mantha, H Chen, Y Pan. Computational methods to identify bimodal gene expression and facilitate personalized treatment in cancer patients. J Biomed Informatics, 2019
- A Ochab-Marcinek, M Tabaka. Bimodal gene expression in noncooperative regulatory systems. Proc Natl Acad Sci U S A, 2010. [PubMed]
- M Samoilov, S Plyasunov, AP Arkin. Stochastic amplification and signaling in enzymatic futile cycles through noise-induced bistability with oscillations. Proc Natl Acad Sci U S A, 2005. [PubMed]
- L Scrucca, M Fop, TB Murphy, AE Raftery. mclust 5: Clustering, classification and density estimation using gaussian finite mixture models. R Journal, 2016
- AK Shalek, R Satija, X Adiconis, RS Gertner, JT Gaublomme, R Raychowdhury, S Schwartz, N Yosef, C Malboeuf, D Lu. Single-cell transcriptomics reveals bimodality in expression and splicing in immune cells. Nature, 2013. [PubMed]
- DM Titterington, AFM Smith, UE Makov. Statistical Analysis of Finite Mixture Distributions, 1986
- L Wang, Z Feng, X Wang, X Wang, X Zhang. DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics, 2010. [PubMed]
- H Wickham. ggplot2: Elegant Graphics for Data Analysis, 2016
- AI Young, S Benonisdottir, M Przeworski, A Kong. Deconstructing the sources of genotype-phenotype associations in humans. Science, 2019. [PubMed]
