Interactions Between Variation in Candidate Genes and Environmental Factors in the Etiology of Schizophrenia and Bipolar Disorder: a Systematic Review
Abstract
Schizophrenia and bipolar disorder (BD) are complex and multidimensional disorders with high heritability rates. The contribution of genetic factors to the etiology of these disorders is increasingly being recognized as the action of multiple risk variants with small effect sizes, which might explain only a minor part of susceptibility. On the other site, numerous environmental factors have been found to play an important role in their causality. Therefore, in recent years, several studies focused on gene × environment interactions that are believed to bridge the gap between genetic underpinnings and environmental insults. In this article, we performed a systematic review of studies investigating gene × environment interactions in BD and schizophrenia spectrum phenotypes. In the majority of studies from this field, interacting effects of variation in genes encoding catechol-O-methyltransferase (COMT), brain-derived neurotrophic factor (BDNF), and FK506-binding protein 5 (FKBP5) have been explored. Almost consistently, these studies revealed that polymorphisms in COMT, BDNF, and FKBP5 genes might interact with early life stress and cannabis abuse or dependence, influencing various outcomes of schizophrenia spectrum disorders and BD. Other interactions still require further replication in larger clinical and non-clinical samples. In addition, future studies should address the direction of causality and potential mechanisms of the relationship between gene × environment interactions and various categories of outcomes in schizophrenia and BD.
Article type: Review Article
Keywords: Psychosis, Bipolarity, Gene polymorphism, Gene × environment interaction
Affiliations: 0001 1090 049Xgrid.4495.cDepartment of Genetics, Wroclaw Medical University, 1 Marcinkowski Street, 50-368 Wroclaw, Poland; 0001 1090 049Xgrid.4495.cDepartment of Psychiatry, Wroclaw Medical University, 10 Pasteur Street, 50-367 Wroclaw, Poland; 0001 2180 3484grid.13648.38Department of Psychiatry and Psychotherapy, University Medical Center Hamburg-Eppendorf, Hamburg, Germany; grid.13339.3bII Department of Psychiatry, Medical University of Warsaw, Warsaw, Poland; 0001 2162 9631grid.5522.0Institute of Psychology, Jagiellonian University, Krakow, Poland; 0000 9939 5719grid.1029.aSchool of Social Sciences and Psychology, Marcs Institute of Brain and Behaviour, Western Sydney University, Penrith, NSW Australia
License: © The Author(s) 2017 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Article links: DOI: 10.1007/s12035-017-0708-y | PubMed: 28822116 | PMC: PMC5948257
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (855 KB)
Introduction
Schizophrenia and bipolar disorder (BD) represent complex and multidimensional phenotypes with high heritability rates, exceeding 80% in twin studies [ref. 1–ref. 3]. However, the concept of schizophrenia and BD as monogenic disorders was abandoned many years ago, since genome-wide association studies (GWASs) have revealed that the risk of schizophrenia and BD is conferred by a large number of alleles with small effect sizes, all of them explaining only some part of susceptibility [ref. 4, ref. 5]. In addition, GWASs have not confirmed several findings from studies based on a candidate gene approach [ref. 4, ref. 5]. The polygenic risk score that was developed based on GWASs has been found to mediate nearly 20% of familial liability for schizophrenia, suggesting an important role of non-genetic factors [ref. 6]. It has been also reported that some cases of schizophrenia might be attributed to rare structural aberrations that are characterized by moderate or large effects and include copy number variations, deletions, duplications, and translocations [ref. 7]. To make things more complex, it has been shown that schizophrenia and BD might share common genetic underpinnings [ref. 8]. Indeed, there are studies showing familial co-aggregation of schizophrenia and BD [ref. 9], common cognitive and neurostructural endophenotypes [ref. 10–ref. 12], and genetic variability [ref. 13].
Apart from genetic risk factors, it has been demonstrated that a number of environmental exposures including urban upbringing, stressful life events and early life stress, prenatal infections and obstetric complications interfering with brain development, and substance abuse or dependence may underlie the development of schizophrenia and BD [ref. 14, ref. 15]. In light of several genetic and environmental factors involved in the etiology of these disorders, gene × environment (G × E) interactions have emerged as a novel research paradigm that might serve as a missing link in trajectories leading to schizophrenia or BD. These interactions refer to various scenarios, where genotype expression depends on exposure to a particular environment or in other words—the effects of environmental exposure depend on a particular genotype [ref. 16–ref. 18]. This approach has also indicated that the effects of some candidate genes might be significant only when certain environmental factors are taken into account.
To date, several G × E interactions have been reported in schizophrenia and BD; however, a recent systematic review of studies in this field was performed in 2013 and was limited to studies on patients with schizophrenia spectrum phenotypes [ref. 19]. Importantly, this systematic review focused on studies that examined psychotic disorders or various subgroups of clinically relevant or subthreshold psychotic symptoms as outcomes of G × E interactions. Therefore, studies investigating other outcome variables, such as cognitive performance or structural and functional brain alterations were not included. In turn, a systematic review of studies addressing G × E interactions in BD has not been performed so far. Therefore, the aim of this study was to perform an updated systematic and comprehensive review of studies investigating interactions between genetic variation in candidate genes and environmental factors in patients with schizophrenia spectrum phenotypes and BD.
Materials and Methods
Search strategy followed PRISMA guidelines [ref. 20], although our systematic review was not registered. Three people (B.M., F.S., and D.F.) independently performed an online search for relevant publications in the PubMed, MEDLINE, ERIC (Education Resource Information Center), CINAHL, and Complete, Academic Search Complete and Health Source – Consumer Edition databases, using the following combination of keywords: (1) “schizophrenia” or “psychosis” or “bipolar disorder,” (2) “gene,” and (3) “environment.” In addition, our search strategy was supplemented by reference lists of relevant publications. After that, search results were compared and studies investigating interactions between candidate gene polymorphisms and environmental exposures in patients with schizophrenia spectrum phenotypes and BD were included in further analysis. There were following exclusion criteria: (1) publications written in non-English language; (2) non-original articles (commentaries, editorials, hypotheses, study protocols, methodological articles, reviews) and meta-analyses; (3) conference proceedings; (4) publications from studies with proxy measures of genetic liability, e.g., studies on relatives of patients with schizophrenia or BD and twin studies; (5) studies without genetic and/or environmental measures; (6) studies investigating DNA methylation patterns without genetic and/or environmental measures; (7) publications from GWASs; and (8) publications from studies on animal models and/or cell lines. We did not include studies investigating subclinical symptoms of BD in non-clinical samples, because these symptoms might be also closely related to major depressive disorder. Our systematic review covered publication records from database inception until 13 November 2016.
Results
We identified 11 eligible studies performed on patients with BD and 50 studies on schizophrenia spectrum phenotypes as well as 1 study from both diagnostic groups (Fig. 1). These studies were grouped into five distinct clusters, based on environmental factors: (1) gene × cannabis interactions, (2) gene × stress and childhood trauma interactions, (3) gene × season of birth interactions, (4) gene × infectious factors interactions, and (5) gene × obstetric complications interactions (for overview of studies, see Tables 1 and 2).
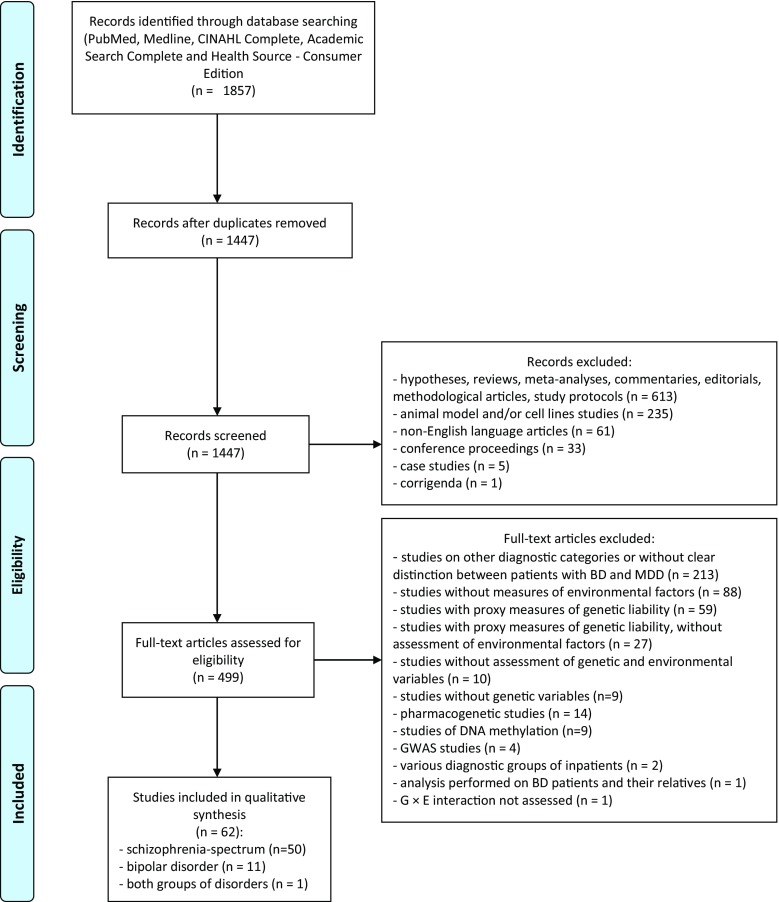
Table 1: Studies investigating interactions between candidate genes and environmental factors in schizophrenia-spectrum phenotypes
| Study (year) | Sample | Candidate gene polymorphisms | Candidate environmental factors | Outcome | Main results |
|---|---|---|---|---|---|
| De Castro-Catala et al. 2016 [ref. 21] | Two independent non-clinical samples of students (n = 808) and female twins (n = 621) | BDNF Val66Met (rs6265) | Childhood trauma (CTQ) | Subclinical psychotic experiences (CAPE) | A history of childhood trauma was associated with both positive and negative psychotic experiences. In the sample of students, the Val allele, especially in males, was associated with higher vulnerability of the effects of childhood trauma on psychotic experiences, while in the group of female twins this association was driven by the Met allele. |
| Cristobal-Narvaez et al. 2016 [ref. 22] | 206 non-clinical young adults | FKBP5 (rs3800373, rs9296158, and rs1360780) | Childhood trauma (CECA.Q) and social stress appraisal (assessed by ESM [ref. 23]) | Negative affect, social contact, and psychotic-like experiences assessed by ESM [ref. 23] | There was a significant effect of interaction between the FKBP5 risk haplotype and childhood bullying on positive psychotic-like experiences, paranoia, and negative affect. The childhood bullying × the FKBP5 haplotype interaction moderated the association of social stress appraisal with psychotic-like experiences and negative affect in daily life. Specifically, the associations were significantly increased in those with the risk haplotype, but not in individuals without the risk haplotype. |
| Gattere et al. 2016 [ref. 24] | 124 individuals with early PD, 36 ARMS subjects, and 62 HCs | BDNF Val66Met (rs6265) | Stressful life events during previous 6 months (SRRS), perceived stress (PSS), and physical activity (IPAQ-SF) | Dietary patterns (FCQ-S) | Perceived stress was not associated with calorie intake in HCs. ARMS subjects with the Met allele and low perceived stress presented with increased caloric intake, while those with high perceived stress presented with decreased caloric intake. In patients with early psychosis, perceived stress was not associated with calorie intake. Perceived stress was associated with food craving in patients with psychosis. A similar association was present in ARMS subjects and HCs who were Val/Val homozygotes. |
| Mandelli et al. 2016 [ref. 25] | 94 patients with SZ176 HCs | ST8SIA2(rs3759917, rs11632521, rs3784722, rs4777989, rs2290492, rs8035760, rs11853992, and rs17522085) | Stressful life events (authors’ own questionnaire) | Age of psychosis onset | No significant interactions between the ST8SIA2 gene polymorphisms and stressful life events on age of psychosis onset. |
| Nieman et al. 2016 [ref. 26] | 147 ARMS subjects | COMT Val158Met (rs4680) | Cannabis use (CIDI) | Severity of ARMS psychopathology (CAARMS) | Weekly cannabis users at some point before the study had higher levels of positive symptomatology. This effect was stronger in the Val allele carriers and even more stronger in the Val/Val homozygotes. |
| Ursini et al. 2016 [ref. 27] | 244 HCs, 162 patients with SZ, 140 siblings of patients with SZ and 214 parents of SZ patients | BDNF Val66Met (rs6265)—genotype, DNA methylation and expression | Obstetric complications—McNeil-Sjöström Scale [ref. 28] | Working memory—n-back task, dorsolateral prefrontal cortex activity, and schizophrenia risk | In Val/Val HCs, increased methylation at the rs6265 polymorphic site was associated with exposure to hypoxia-related early life events and working memory impairment (opposite effect was observed in Val/Met heterozygotes). The same effect was found for working memory-related prefrontal activity. Higher methylation levels in Val/Val homozygotes was associated with schizophrenia. |
| Colizzi et al. 2015 [ref. 29] | 272 FEP patients and 234 HCs (case-control study), and 252 healthy subjects | DRD2 (rs1076560) | Cannabis use (CEQ) in all participants | Psychosis risk, schizotypy (SPQ), and cognition (n-back working memory task) in healthy subjects | In cannabis users, T allele carriers had a threefold increase in psychosis risk compared to GG homozygotes. In daily users, T allele carriers had a fivefold increase in psychosis risk compared to GG homozygotes. In healthy subjects, daily users with T allele had higher schizotypy compared to cannabis-naïve T allele carriers, cannabis users with GG genotype and cannabis-naïve GG homozygotes. Cannabis users with T allele had lower working memory performance in comparison with other groups. |
| Ermis et al. 2015 [ref. 30] | 80 male patients with SZ | COMT Val158Met (rs4680) | Cannabis use (disease history, family interviews and medical records) | Schizophrenia psychopathology (PANSS) | The Val/Val genotype was significantly more frequent in patients with premorbid cannabis use compared to those without cannabis use. There were higher levels of positive and negative symptoms in Val/Val homozygotes compared to the Met allele carriers. |
| Green et al. 2015 [ref. 31] | 444 patients with SZ and 292 HCs | FKBP5 (rs1360780, rs9470080, rs4713902, and rs9394309) | Childhood trauma (CAQ) | Cognitive performance (RBANS, premorbid IQ—WTAR, the letter number sequencing—WAIS, COWAT) | There were significant main effects of the rs1360870 genotype and childhood trauma and a significant interaction between these variables affecting attention in both groups (CC homozygotes performed worse in the context of childhood trauma). Additionally, there were significant main effects of this polymorphism on global cognition in SZ patients (TT homozygotes performed worse). |
| Wegelius et al. 2015 [ref. 32] | 457 subjects from the Finnish Schizophrenia Birth Cohort | NDE1 (rs4781678, rs2242549, rs881803, and rs2075512), PDE4B (rs7412571) PDE4D (haplotype) | Birth weight | A diagnosis of SZ (medical records, SCID) | High birth weight was associated with schizophrenia risk in subjects homozygous for risk alleles (a four-SNP haplotype spanning NDE1 gene and one of its constituent SNPs—rs4781678). |
| Aas et al. 2014 [ref. 33] | 182 patients with SZ spectrum diagnosis, 130 patients with BD, 11 patients with MDD with psychotic features | BDNF Val66Met (rs6265) | Childhood trauma (CTQ) | Hippocampal subfield measures and BDNF mRNA levels | Met allele carriers with high levels of childhood trauma had significantly lower levels of BDNF mRNA and reduced CA2/3 and CA4 subfields of dentate gyrus. |
| Ajnakina et al. 2014 [ref. 34] | 291 FEP patients and 218 HCs | FKBP5 (rs1360780) | Cannabis use (CEQ)Childhood trauma (CECA.Q) | FEP—risk of psychosis (ICD-10) | The FKBP5 rs1360780 polymorphism was associated with the risk of psychosis only after adjustment for environmental factors. There was a significant effect of the interaction between the FKBP5 rs1360780 polymorphism and parental separation on psychosis risk. |
| Hernaus et al. 2014 [ref. 35] | 89 patients with PD and 95 healthy siblings | FKBP5 (rs9296158, rs4713916, rs992105, and rs38003), BDNF Val66Met (rs6265) | Childhood trauma (CTQ) | Hippocampal volume and cognitive performance (auditory verbal learning task and block design task from WAIS) | There were no significant effects of interactions between studied polymorphism and childhood trauma on hippocampal volumes and cognition. |
| McCarthy-Jones et al. 2014 [ref. 36] | 333 SZ spectrum patients | FOXP2 (rs1456031, rs2396753, and rs2253478) | Childhood trauma (CTQ) | Lifetime history of AVHs (DIP) | There was a significant effect of the interaction between the rs1456031 polymorphism and parental emotional abuse. Emotional abuse was only associated with significantly higher levels of AVHs in patients with CC genotype. However, in the absence of emotional abuse TT homozygotes had significantly higher levels of AVHs than those with CC genotype. |
| Pishva et al. 2014 [ref. 37] | Sample I: 112 HCsSample II: 434 general population twinsSample III: 85 siblings of patients with PDSample IV: 110 patients with PDSample V: 126 patients with at least one episode of MDD | 31 SNPs in epigenetic-regulatory genes: MTHFR, DNMT1, DNMT3A, and DNMT3B | Daily life stressors (ESM) | Emotional responses (ESM) | Three SNPs in the DNMT3A gene (rs11683424, rs1465764, rs1465825) and the MTHFR rs1801131 moderated the effect of stressful events on negative affect. Effects of the DNMT3A rs11683424 polymorphism were consistent in the majority of samples. |
| Collip et al. 2013 [ref. 38] | 401 general population twins, 195 patients with psychosis, 200 unaffected siblings and 175 HCs | FKBP5 (rs9296158, rs4713916, rs1043805, and rs1360780) | Childhood trauma (CTQ) | Salivary free cortisol levelsPsychotic experiences (CAPE, SIS-R) | There was a significant interaction between the rs9296158/rs4713916 polymorphisms and childhood trauma on psychotic symptoms and cortisol levels in the twin sample. Similar findings were obtained for the rs4713916 polymorphism in siblings and for the rs9296158 polymorphism in patients. Specifically, the A allele carriers at both polymorphisms were most vulnerable to childhood trauma. |
| De Sousa et al. 2013 [ref. 39] | 403 patients with SZ and schizoaffective disorder | COMT polymorphisms (rs4680, rs4633, rs4818, and rs6269) | Cannabis use (DSM-IV, clinical assessment) | Age of psychosis onset | There were no significant interactions between the COMT gene polymorphisms and age of psychosis onset. |
| Onwuaemeze et al. 2013 [ref. 40] | 235 SZ patients | MAPK14 polymorphisms (rs3804454, rs2237094, rs12199654, rs851007, rs851006, rs3804452, rs8510, rs7757672, and rs916346) and CNR1 rs12720071 polymorphism | Substance use (CASH, DSM-IV criteria) | White matter brain volumes | The rs12199654 AA homozygotes with cannabis abuse/dependence had significantly smaller total cerebral and lobar white matter volumes. This effect remained significant after controlling for the CNR1 genotype. There were significant main effects of the MAPK14/CNR1 diplotype and the interaction between this diplotype and cannabis abuse/dependence on white matter brain volumes. The effects of these two polymorphisms were additive. |
| Ramsay et al. 2013 [ref. 41] | 237 general population individuals | COMT Val158Met (rs4680) BDNF Val66Met (rs6265) | Childhood trauma (K-SADS) | Psychotic experiences (K-SADS) | Individuals with the COMT Val/Val genotype exposed to childhood trauma were more likely to report psychotic experiences than those with other COMT genotypes (trend level significance). No significant interactions with childhood trauma were found for the BDNF Val66Met polymorphism. |
| Vinkers et al. 2013 [ref. 42] | Discovery sample: 918 (general population)Replication sample: 339 healthy controls and healthy siblings of patients with NAPD | COMT Val158Met (rs4680) | Cannabis use (CIDI, the monetary amount spent on cannabis)Childhood trauma (CTQ) | Psychotic experiences (CAPE) | Val/Val homozygotes from the discovery sample exposed to cannabis use and childhood maltreatment had significantly higher levels of psychotic experiences compared to Met allele carriers (Val/Met and Met/Met genotypes). These results did not reach statistical significance in the replication sample. |
| Bhattacharyya et al. 2012 [ref. 43] | 35 HCs | DAT1 3′UTR VNTR, AKT1 rs1130233 | Cannabis (delta-9-THC) intake | Delta-9-THC-induced psychotic experiences (PANSS) | The GG homozygotes for the AKT1 rs1130233 polymorphism and also carriers of the 9-repeat allele of the DAT1 3′UTR VNTR had greater increase in psychotic symptoms induced by delta-9-THC compared to subjects with other genotypes. |
| Di Forti et al. 2012 [ref. 44] | 489 FEP patients and 278 HCs | AKT1 rs2494732 | Cannabis use (lifetime use and frequency of use) | FEP—risk of psychosis (ICD-10) | The CC homozygotes with a history of cannabis use showed a greater than twofold increase in the likelihood of PD in comparison with the TT homozygotes.Among daily cannabis users, individuals with the CC genotype demonstrated a sevenfold increase in the odds of psychosis compared to the TT homozygotes. |
| Husted et al. 2012 [ref. 45] | 162 patients with SZ spectrum disorders and 75 HCs | CAPON (NOS1AP) rs12742393 | Cannabis use, childhood trauma (SCID-I) | SZ risk (SCID-I) | No significant effects of interactions between the NOS1AP genotype and environmental factors on SZ risk were found. |
| Peerboms et al. 2012 [ref. 46] | 84 patients with NAPD and 107 HCs | COMT Val158Met (rs4680), MTHFR C677T (rs1801133), MTHFR A1298C (rs1801131) | Daily life stressors (ESM) | Psychotic experiences (ESM self-report) in NAPD (OPCRIT or CASH) | Patients with the MTHFR T allele, COMT Met/Met homozygotes showed the largest increases in psychotic experiences in response to stress. In patients who were the MTHFR CC homozygotes, there was no interaction between the COMT Val158Met polymorphism and stress. There was no moderating effect of MTHFR A1298C on the interaction between the COMT Val158Met polymorphism and stress. |
| Alemany et al. 2011 [ref. 47] | 533 HCs | BDNF Val66Met (rs6265) | Stress-childhood adversity defined as childhood neglect and childhood abuse (CTQ) | Psychotic-like experiences (CAPE) | Individuals carrying the Met allele had higher scores on adult positive psychotic-like experiences when childhood abuse was present, when compared to Val/Val homozygotes.No significant gene × environment interaction was detected with respect to childhood neglect. |
| Collip et al. 2011 [ref. 48] | 86 patients with NAPD and 109 HCs | COMT Val158Met (rs4680) | Daily life stressors (ESM) | Psychotic experiences (ESM self-report) in NAPD (OPCRIT or CASH) | Patients being the COMT Met/Met homozygotes showed increased psychotic reactivity to stress compared to the Val allele carriers. |
| Costas et al. 2011 [ref. 49] | 748 patients with SZ recruited in two independent samples | COMT (rs737865, rs6269, rs4633, rs4818, and rs4680) | Lifetime cannabis use according to DSM-IV criteria or medical records | The risk of cannabis use in patients with schizophrenia | Patients with low activity variants were significantly more prone to use cannabis (joint analysis, results were consistent between two independent samples). In the joint analysis, the probability of lifetime cannabis use was twofold higher in the rs4680 Met/Met homozygotes compared to Val/Val homozygotes. |
| Decoster et al. 2011 [ref. 50] | 585 patients with SZ | BDNF Val66Met (rs6265) | Substance use (CIDI) | Age of psychosis onset | In female patients, cannabis use was associated with earlier age of psychosis onset in the Met allele carriers, but not in Val/Val homozygotes. In male patients, cannabis use was associated with earlier age of psychosis onset, regardless of the BDNF genotype. The main effect of the BDNF genotype on age of psychosis onset was not significant in the whole group as well as in males and females separately. |
| Demontis et al. 2011 [ref. 51] | Three independent samples of SZ patients and HCs (Denmark I: 385 patients and 780 HCs, Denmark II: 365 patients and 434 HCs, Denmark III: 234 patients and 286 HCs) | 81 SNPs in GRIN2A and GRIN2B genes | Maternal HSV-2 seropositivity | The risk of schizophrenia (ICD-10) | The GRIN2B rs1806194 polymorphism was significantly associated with schizophrenia risk after Bonferroni correction. There were significant effects of interactions between two SNPs in the GRIN2B gene (rs1805539 and rs1806205) and maternal HSV-2 seropositivity on schizophrenia risk after Bonferroni correction. |
| Estrada et al. 2011 [ref. 52] | 80 patients with SZ spectrum disorders and 77 patients with non-psychotic disorders (conduct and affective disorders) | COMT Val158Met (rs4680) | Cannabis use (DIGS and urine drug scrrening) | Age at onset of psychiatric disorders | There were no significant differences in genotype distributions between diagnostic groups or between cannabis users or non-users. However, the Val/Val homozygotes had earlier age of psychosis onset compared to the Met allele carriers. This effect was not significant in patients with non-psychotic disorders. |
| Ho et al. 2011 [ref. 53] | 235 patients with SZ | 12 SNPs in the CNR1 gene | Substance use (CASH) | Brain volumes and cognitive performance (WAIS-R, WCST, TMT-A, and Shipley Institute of Living Scale abstractions subtest) | There were significant main effects of the CNR1 gene SNPs (rs7766029, rs12720071, and rs9450898) on white matter volumes. Patients with cannabis abuse/dependence had smaller frontotemporal white matter volumes. There were significant effects of the interaction between the rs12720071 genotype and cannabis abuse/dependence on parietal white matter volumes and cognition (problem solving). |
| Muntjewerff et al. 2011 [ref. 54] | 742 patients with SZ | MTHFR C677T (rs1801133) | Seasonality of birth (winter birth) | SZ (CASH) | There was no evidence for the interaction between MTHFR 677TT genotype and winter birth in the development of SZ. |
| Van Winkel et al. 2011 [ref. 55] | 801 patients with NAPD, 740 unaffected siblings and 419 HCs | 152 SNPs in 42 genes | Cannabis (recent use —urinary result, lifetime pattern of use—CIDI) | Non-affective psychotic disorderPositive schizotypy (factor structure of SIS-R) | Case-only design: the AKT1 rs2494732 polymorphism showed association with lifetime use, restricted to use preceding onset of psychosis.Case-siblings design: AKT1 rs2494732, AKT1 rs1130233, and LRRTM1 rs673871 showed significant interactions with recent cannabis use on positive schizotypy.In follow-up, patients with the AKT1 rs2494732 CC genotype displayed approximately twofold higher odds of being diagnosed with PD when having used cannabis in comparison with TT homozygotes.Case-control design: no significant interaction was shown. |
| Haukvik et al. 2010 [ref. 56] | 54 schizophrenia patients and 53 HCs | BDNF, DTNBP1, GRM3, and NRG1 (32 SNPs) | Obstetric complications assessed with McNeil-Sjöström Scale [ref. 28] | Hippocampal volume | Severe obstetric complications were associated with reduced hippocampal volumes in both groups. There were no significant main effects of studied SNPs on hippocampal volumes. There was a significant effect of the interaction between the GRM3 rs13242038 polymorphism and severe obstetric complications on hippocampal volumes in patients with schizophrenia and HCs. |
| Pelayo-Teran et al. 2010 [ref. 57] | 169 FEP patients | COMT Val158Met (rs4680) | Cannabis use in the previous year (clinical assessment) | Age of onset, DUP | Cannabis users had significantly earlier age of psychosis onset. There was significant interaction between COMT genotype and cannabis use on age of psychosis onset and DUP. Post hoc analyses revealed that the effect of COMT genotype on age of psychosis onset was significant only in cannabis non-users (longer DUP and earlier age of psychosis onset in Val/Val homozygotes. |
| Zammit et al. 2010 [ref. 58] | 2630 HCs | COMT Val158Met (rs4680), rs4818, rs6269, rs737865, rs2097603, rs165599 | Cannabis (self-report postal questionnaires completed at age 14) | Psychotic experiences (incident psychotic experiences at age 16) | There was no evidence of an interaction between COMT SNPs and cumulative use of cannabis on the development of psychotic experiences. |
| Gutierrez et al. 2009 [ref. 59] | 91 patients with SZ and 192 HCs | COMT Val158Met (rs4680) | Cannabis use (frequency of taking in the previous month) | Schizophrenia risk (DSM-IV) | Cannabis consumption was significantly more frequent in the group of patients. In the Val allele carriers, cannabis consumption rates were higher in female schizophrenia patients compared to healthy women, while in the Met/Met homozygotes, cannabis consumption rates were higher in healthy women compared to female schizophrenia patients (non-significant interaction). |
| Henquet et al. 2009 [ref. 60] | 31 patients with PD and 25 HCs | COMT Val158Met (rs4680) | Cannabis use (ESM self-report) | Psychosis liability (CAPE)Psychotic experiences (ESM self-report) | Cannabis significantly increased hallucinatory experiences only in individuals who were carriers of the Val allele and also had high levels of psychometric psychosis liability. No such associations were observed for delusional experiences. |
| Kantrowitz et al. 2009 [ref. 61] | 92 patients with PD | COMT Val158Met (rs4680) | Adolescent cannabis use prior to age 18 (SCID) | Psychotic disorder (SCID) | No differences in COMT Val58Met genotype distribution with respect to adolescent cannabis use neither in Caucasian nor in African-American patients with psychotic disorder. |
| Keri et al. 2009 [ref. 62] | 200 SZ | NRG1 (rs6994992, rs10954867, and rs7005288) | Psychosocial stress (conflict-related family interactions—PSP) | Odd and unusual thought content during neutral and conflict-related family interactions over the two 10-min interactions with one of the family members: mothers, fathers, wives, husbands, and siblings in SZ (MINI) | Patients with the NRG1 TT genotype showed more unusual thoughts during conflict-related interactions than patients with CT and CC genotypes (rs6994992). There were no significant differences between the NRG1 CT and CC patients. There were no significant differences among patients with different NRG1 genotypes during neutral interactions. |
| Simons et al. 2009 [ref. 63] | 579 HC female twins | COMT Val158Met (rs4680) BDNF Val66Met (rs6265) | Event stress and social stress in daily life (ESM) | Feelings of paranoia (ESM self-report) | Carriers of the COMT 158 Val allele displayed more feelings of paranoia in response to event stress compared to Met carriers.Carriers of the BDNF 66Met allele showed more social stress-induced paranoia than individuals with the Val/Val genotype. |
| Nicodemus et al. 2008 [ref. 64] | 116 SZ spectrum disorders and 134 HCs | AKT1, BDNF, CAPON (NOS1AP), CHRNA7, COMT, DTNBP1, GAD1, GRM3, NOTCH4, NRG1, PRODH, RGS4, TNF-α | Obstetric complications (questionnaires were completed by parents of affected individuals and of control subjects) | SZ spectrum disorders (SCID-I, SCID-II) | Probands with obstetric complications were more likely to have minor allele at the AKT1 rs2494735 and rs1130233 polymorphisms, major allele at the BDNF rs2049046 polymorphism and the minor allele at the rs76882600 polymorphism, minor allele at the DTNBP1 rs875462, minor allele at the GRM3 rs7808623. |
| Van Winkel et al. 2008 [ref. 65] | 31 patients with PD and cannabis use, 25 non-psychotic cannabis users | COMT Val158Met (rs4680) | Daily life stressors (ESM) | Psychotic experiences (ESM self-report) | Subjects with the COMT 158 Met/Met genotype had greater increase in overall psychotic experiences in response to daily stressors in comparison to Val/Met and Val/Val carriers both among patients and healthy controls. |
| Shirts et al. 2007 [ref. 66] | Three independent samples of SZ patients (primary sample: 236 patients and 240 HCs, Baltimore: 272 cases and 108 HC, Pittsburgh: 221 case-parent trios) | 26 SNPs from the locus 6p21 | CMV and HSV1 seropositivity | Schizophrenia risk (DSM-IV) | In Baltimore controls, the MICB rs1051788 polymorphism was associated with HSV1 seropositivity, while the MICB rs2523651 polymorphism was associated with CMV seropositivity. The former association was also observed in Pittsburg parents. None of them was observed in patients with schizophrenia. There was a significant transmission distortion of the MICB SNPs (rs1051788 and rs1055569) in case-parent trios regardless of antibody status. The association between the MICB rs1051788 polymorphism and schizophrenia risk was not significant. |
| Stefanis et al. 2007 [ref. 67] | 306 male HCs | COMT Val158Met (rs4680) | Stress (recruitment in the army) | Psychotic symptoms (SCL-90-R) | Carriers of the COMT 158Val allele were more sensitive to psychosis inducing effects of stress exposure at army in comparison with Met/Met homozygotes. |
| Zammit et al. 2007 [ref. 68] | 750 patients with SZ and 688 HCs | CNR1 rs1049353, COMT Val158Met (rs4680), rs737865, and rs165599 CHRNA7-86C/T | Cannabis use (interview and case-note records) | Schizophrenia (SCAN, OPCRIT) | No evidence of interaction between cannabis use and selected polymorphisms. |
| Henquet et al. 2006 [ref. 69] | 30 patients with PD, 12 first and second-degree relatives (3 with BPD and 1 with MDD) and 32 HCs | COMT Val158Met (rs4680) | Cannabis (delta-9-THC) intake | Psychosis liability (CAPE) Delta-9-THC-induced psychotic experiences (PANSS) | The COMT Val/Val homozygotes had largest increase in delta-9-THC -induced psychotic experiences varying as a function of psychometric psychosis liability. |
| Caspi et al. 2005 [ref. 70] | 803 HCs | COMT Val158Met (rs4680) | Cannabis (follow-ups carried out at ages 3, 5, 7, 9, 11, 13, 15, 18, 21, 26) | Psychosis outcomes assessed at age 26 (DIS): schizophrenia and schizophreniform disorder. A 60-item questionnaire was mailed to persons nominated by each study member at age 26 as “someone who knows you well” | Adolescent cannabis use was associated with increased risk of schizophrenia and schizophreniform disorder in adulthood in the COMT 158Val allele carriers, but not in Met/Met homozygotes. |
| Chotai et al. 2003 [ref. 71] | 147 patients with SZ and 395 HCs | TPH1 A218C (rs1800532) 5-HTTLPR DRD4 | Seasonality of birth | SZ (OPCRIT) | The frequency of the DRD4 7-repeat allele showed one-cyclic season of birth variation in women with SZ. |
| Tochigi et al. 2002 [ref. 72] | 110 patients with SZ and 493 HCs | HLA (HLA-A24, HLA-A26) | Seasonality of birth (winter birth) | SZ (DSM-IV) | No association between HLA (-A24 or -A26) and winter birth (December–March) in patients with schizophrenia. |
| Narita et al. 2000 [ref. 73] | 60 SZ patients with HLA-DR1 and 307 SZ patients without HLA-DR1 | HLA (HLA-DR1) | Seasonality of birth (winter birth) | SZ (DSM-IV) | Increased incidence of winter births (February–March) in patients with HLA-DR1 than in patients without HLA-DR1. |
AKT1 RAC-α serine/threonine-protein kinase, ARMS At-Risk Mental State, AVHs auditory verbal hallucinations, BD bipolar disorder, BDNF brain-derived neurotrophic factor, CAARMS Comprehensive Assessment of At-Risk Mental State [ref. 74], CAPE Community Assessment of Psychic Experiences [ref. 75], CAPON (NOS1AP) nitric oxide synthase 1 adaptor protein, CAQ Childhood Adversity Questionnaire [ref. 76], CASH Comprehensive Assessment of Symptoms and History [ref. 77], CECA.Q the Childhood Experience and Abuse Questionnaire [ref. 78], CEQ Cannabis Experience Questionnaire [ref. 79], CHRNA7 neuronal acetylcholine receptor subunit alpha-7, CIDI the Composite International Diagnostic Interview [ref. 80], CMV cytomegalovirus, CNR1 cannabinoid receptor 1, COMT catechol-O-methyltransferase, COWAT the Controlled Oral Word Association Test [ref. 81], CTQ Childhood Trauma Questionnaire [ref. 82], DAT1 dopamine active transporter 1, DIP Diagnostic Interview for Psychoses [ref. 83], DIS Diagnostic Interview Schedule for DSM-IV [ref. 84], DNMT DNA methyltransferase, DRD4 dopamine D4 receptor, DTNBP1 dystrobrevin binding protein 1, ESM experience sampling methodology, FCQ–S Food Craving Questionnaire-State [ref. 85], FEP first-episode psychosis, FKBP5 FK506 binding protein 5, FOXP2 forkhead box protein 2, GAD1 glutamate decarboxylase 1, GRIN2A glutamate ionotropic receptor NMDA type subunit 2A, GRIN2B glutamate ionotropic receptor NMDA type subunit 2B, GRM3 glutamate metabotropic receptor 3, HCs healthy controls, HLA human leukocyte antigen, HSV–1 Herpes Simplex Virus 1, IPAQ–SF International Physical Activity Questionnaire Short Form [ref. 86], K–SADS the Schedule for Affective Disorders and Schizophrenia for School-Aged Children [ref. 87], MAPK14 mitogen-activated protein kinase 14, MDD major depressive disorder, MINI Mini-International Neuropsychiatric Interview [ref. 88], MTHFR methylenetetrahydrofolate reductase, NAPD non-affective psychotic disorder, NDE1 nuclear distribution protein nudE homolog 1, NOS not otherwise specified, NOTCH4 neurogenic locus notch homolog 4, PANSS Positive and Negative Syndrome Scale [ref. 89], NRG1 neuregulin 1, OPCRIT the Operational Criteria for Psychotic Illness Checklist [ref. 90], PANSS the Positive and Negative Syndrome Scale [ref. 89], PD psychotic disorder, PRODH proline dehydrogenase 1, PSP Patient Symptom Profile [ref. 91, ref. 92], PSS Perceived Stress Scale [ref. 93], RBANS Repeatable Battery for the Assessment of Neuropsychological Status [ref. 94], RGS4 regulator of G protein signaling 4, SCID Structured Clinical Interview for DSM-IV [ref. 95], SIS–R the Structured Interview for Schizotypy-Revisited [ref. 96], SNPs single nucleotide polymorphisms, SPQ Schizotypal Personality Questionnaire [ref. 97], SRRS Holmes-Rahe Social Readjustment Rating Scale [ref. 98], ST8SIA2 ST8 alpha-N-acetyl-neuraminide alpha-2,8-sialyltransferase 2, SZ schizophrenia, TNF–α tumor necrosis factor-α, TPH1 tryptophan hydroxylase 1, WAIS Wechsler Adult Intelligence Scale [ref. 99], 5-HTTLPR serotonin-transporter-linked polymorphic region
Table 2: Studies investigating interactions between candidate genes and environmental factors in bipolar disorder
| Study (year) | Sample | Candidate gene polymorphisms | Candidate environmental factors | Outcome | Main results |
|---|---|---|---|---|---|
| Chotai et al. 2003 [ref. 71] | 456 patients with BD351 patients with MDD147 patients with SZ395 HCs | TPH1 A218C (rs1800532), 5-HTTLPR L/S (rs25531), and DRD4 7-repeat allele | Season of birth | The risk of BD, MDD, SZ | The allele frequencies did not show any significant variation with respect to seasons defined as four 3-month periods beginning in January. However, the analysis of one-cyclic month of birth variations showed that the TPH1 allele A had a positive peak around the birth month December and a negative peak around June in men with BD, but not in women with BD. There were more cases of BP among men with TPH1 allele A born in Nov-Jan, and less cases of BP among women with TPH1 allele A born in Feb-Jul in comparison to healthy controls. Moreover, analysis of two cycles per year showed differences in the DRD4 gene variations both among women and men with BD. |
| Dickerson et al. 2006 [ref. 100] | 107 patients with BD95 HCs | COMT Val158Met (rs4680) | Antibodies to HSV-1 | Cognitive performance (RBANSS) | The COMT 158Val/Val genotype and HSV-1 seropositivity were independent predictors of lower global cognitive performance in patients with BD but not in HCs. Patients with both COMT 158Val/Val genotype and HSV-1 seropositivity were 85 times more likely to be in the lowest quintile of global cognitive performance. |
| De Pradier et al. 2010 [ref. 101] | 137 patients with BD | 5-HTTLPR L/S (rs25531) | Cannabis abuse or dependence (DIGS)Childhood trauma (THQ) | Lifetime occurrence of psychotic symptoms (DIGS) | The interaction between the S allele and childhood sexual abuse increased odds of cannabis abuse or dependence. Cannabis abuse or dependence and S allele, but not childhood sexual abuse, were significantly more frequent in those patients with lifetime occurrence of psychotic symptoms. |
| Hosang et al. 2010 [ref. 102] | 487 patients with BD type 1598 HCs | BDNF Val66Met (rs6265) | Stressful life events (LTE-Q) | Worst episodes of depression and mania (SCAN) | The BDNF 66Met allele carriers with higher levels of stressful life events had higher severity of the worst depression ever. |
| Savitz et al. 2010 [ref. 103] | 222 patients with BD type 1 | DRD4 48 bp VNTR, DRD2 Taq1A, DAT1 3′VNTR, and MAOA promoter VNTR | Childhood trauma (CTQ) | Schizotypy (STA) | The COMT 158Val allele was associated with higher levels of schizotypy in patients exposed to higher levels of childhood trauma. There were no main effects of the COMT 158Val/Met polymorphism on the levels of schizotypy. |
| Debnath et al. 2013 [ref. 104] | 561 patients with BD161 HCs | HLA-G 14 bp ins/del (66554220) | Season of birth | The risk of BD | The HLA-G ins/ins genotype was significantly less frequent in patients with BD. The prevalence of this genotype was significantly lower in patients born in the winter season. |
| Miller et al. 2013 [ref. 105] | 80 patients with BD (43 with type I, 33 with type II, 4 with NOS) | BDNF 66Val/Met (rs6265) | Childhood trauma (CTQ) | Severity and chronicity of BD (CGI-BP-OS) | The BDNF 66Met allele carriers with a history of childhood sexual abuse had 21% higher levels of BD severity and chronicity as well as 35% earlier age of BD onset compared to those without this type of childhood trauma. This interaction was not significant in regression analysis |
| Bortolasci et al. 2014 [ref. 106] | 45 patients with BD91 patients with MDD199 HCs | PON1 Q192R (rs662) | Cigarette smoking | The risk of BD and MDD | The interaction between PON1 QQ genotype and cigarette smoking increased the risk of BD and MDD in separate analyses. |
| Breen et al. 2015 [ref. 107] | 631 patients with BD and lifetime history of suicide attempts 657 patients with BD without lifetime history of suicide attempts | 235 HPA axis SNPs | Childhood physical and sexual abuse (ELES) | Suicide attempts (DIGS) | No significant effects of interactions between polymorphisms in HPA axis genes and childhood trauma on lifetime occurrence of suicide attempts. |
| Oliveira et al. 2015 [ref. 108] | 531 patients with BD (391 with type 1, 113 with type 2 and 27 with NOS) | TLR2 (rs4696480 and rs3804099) and TLR4 (rs1927914 and rs11536891) | Childhood trauma (CTQ) | Age of BD onset | Patients with the TLR2 rs3804099 TT genotype and a history of sexual abuse had earlier age of BD onset compared to other patients in Kaplan-Meier survival curve analysis but not in regression analysis. |
| Zeni et al. 2016 [ref. 109] | 29 children and adolescents with BD22 HCs | BDNF Val66Met (rs6265) | Family functioning (FES) | Hippocampal volumes | A significant interaction between Met allele and low scores of cohesion subscale (from Family Environment Scale-Revised) on the left hippocampal volume in patients with BD. Main effects were not significant. There were no significant differences between HCs and BD patients in terms of hippocampal volume. |
| Oliveira et al. 2016 [ref. 110] | 138 patients with BD167 HCs | TLR2 (rs4696480 and rs3804099), TLR4 (rs1927914 and rs11536891) and NOD2 (rs2066842) | Toxoplasma gondii seropositivity | The risk of BD | A trend toward statistical significance for the interaction between Toxoplasma gondii seropositivity and the TLR2 rs3804099 polymorphism in conferring BD risk. |
BD bipolar disorder, BDHI Buss-Durkee Hostility Inventory [ref. 111], BDNF brain-derived neurotrophic factor, BGAI Brown-Goodwin Aggression Inventory [ref. 112], BIS Barratt Impulsivity Scale [ref. 113], CGI–BP–OS Clinical Global Impressions Bipolar Version Overall Severity of Illness [ref. 114], COMT catechol-O-methyltransferase, CTQ Childhood Trauma Questionnaire [ref. 82], DAT1 dopamine active transporter 1,DIGS Diagnostic Interview for Genetic Studies [ref. 115], DRD2 dopamine D2 receptor, DRD4 dopamine D4 receptor, ELES Early Life Events Scale unpublished [ref. 107], FES Family Environment Scale [ref. 116], HCs healthy controls, HDRS Hamilton Depression Rating Scale [ref. 117], HLA–G histocompatibility antigen class I,G, HPA hypothalamic-pituitary-adrenal, HSV–1 Herpes Simplex Virus 1, LTE–Q the List of Threatening Experiences Questionnaire [ref. 118], MAOA monoamine oxidase A, MDD major depressive disorder, PBI Parental Bonding Instrument [ref. 119], PON1 paraoxonase 1, SCAN Schedules for Clinical Assessments in Neuropsychiatry [ref. 120], STA the Schizotypal Personality Scale [ref. 121], SZ schizophrenia, THQ Trauma History Questionnaire [ref. 122], TLR2 toll-like receptor 2, TLR–4 toll-like receptor 4, TPH1 tryptophan hydroxylase 1, VNTR variable number of tandem repeats, 5–HTTLPR serotonin-transporter-linked polymorphic region
Gene × Cannabis Interactions
To date, interactions between cannabis use and genetic factors have been tested in 24 studies performed on patients with schizophrenia spectrum phenotypes [ref. 26, ref. 29, ref. 30, ref. 34, ref. 39, ref. 40, ref. 42–ref. 45, ref. 49, ref. 50, ref. 52, ref. 53, ref. 55, ref. 57–ref. 61, ref. 68–ref. 70]. The vast majority of these studies analyzed the impact of dopaminergic genes. In one longitudinal study of a representative cohort followed from adolescence to adulthood, Caspi et al. [ref. 70] found that cannabis use in adolescence increased odds of schizophrenia and schizophreniform disorder in the catechol-O-methyltransferase (COMT) 158Val allele carriers, but not in the COMT 158Met allele homozygotes. Subsequent studies have provided mixed results. Henquet et al. [ref. 69] revealed that exposure to delta-9-tetrahydrocannabinol (THC) leads to the largest increase in psychotic symptoms and memory impairment in patients with psychotic disorders and healthy controls homozygous for the COMT 158Val allele. More specifically, the same group showed that carriers of the COMT 158Val allele, but not subjects with the COMT 158 Met/Met genotype, present an increase in hallucinations after cannabis exposure that is conditional on prior psychosis liability [ref. 60]. Similar results showing higher levels of positive psychopathology in the COMT 158Val allele carriers (or COMT 158 Val/Val homozygotes) have been reported in cannabis users among at-risk mental state (ARMS) individuals [ref. 26] and schizophrenia patients [ref. 30]. Interestingly, in one study [ref. 52], the effects of the interaction between the COMT Val158Met polymorphism and cannabis use on age at onset of schizophrenia spectrum disorders and non-psychotic disorders were tested. Authors found earlier age at onset in patients with schizophrenia spectrum disorders with the COMT 158 Val/Val genotype compared to the COMT 158Met allele carriers. This effect was insignificant in patients with non-psychotic disorders. A similar interaction between cannabis use and the COMT Val158Met polymorphism was reported by Pelayo-Teran et al. [ref. 57]. Authors found that earlier age of psychosis onset and longer duration of untreated psychosis in the COMT Val/Val first-episode psychosis (FEP) patients, who were cannabis non-users. This effect was insignificant in cannabis users with FEP. Vinkers et al. [ref. 42] also found in the discovery sample of general population individuals that the COMT 158Val/Val homozygotes exposed to cannabis use and childhood maltreatment have higher levels of psychotic experiences than carriers of the COMT 158Met allele. However, these findings were not replicated in the confirmation sample. In one study, the opposite results were obtained showing that the probability of lifetime cannabis use was twofold higher in schizophrenia patients with the COMT 158 Met/Met genotype compared to Val/Val homozygotes [ref. 49]. Other studies have not confirmed the interaction between the COMT 158Val/Met polymorphism and cannabis use on the risk of psychosis [ref. 55, ref. 61, ref. 68] or risk of subclinical psychotic experiences [ref. 58] and age of psychosis onset [ref. 39]. In one study, no significant differences in the frequency of cannabis use between schizophrenia patients with distinct COMT genotypes were found [ref. 59]. Additionally, Zammit et al. [ref. 68] did not find any significant interactions between variation in the cannabinoid receptor 1 (CNR1) gene and the cholinergic receptor, nicotinic, alpha 7 (CHRNA7) genes, and cannabis use. However, Ho et al. [ref. 53] found that schizophrenia patients with cannabis abuse/dependence with the CNR1 rs12720071 G allele had lower parietal white matter volumes (trend level significance) and performed significantly worse on problem solving tasks. In agreement with these findings, a more recent study demonstrated an interaction between the CNR1 rs12720071/MAPK14 rs12199654 diplotype and cannabis abuse/dependence on white matter volumes in another sample of patients with schizophrenia [ref. 40].
In three studies [ref. 43, ref. 44, ref. 55], the interaction between genetic variation in the RAC-alpha serine/threonine-protein kinase 1 (AKT1) gene and cannabis use has been tested. Van Winkel et al. [ref. 55] addressed this issue in a study of patients with non-affective psychosis, unaffected siblings of patients with psychosis and healthy controls that initially included a set of 152 single nucleotide polymorphisms (SNPs) in 42 candidate genes. Main effects of three SNPs on positive schizotypy, after recent cannabis exposure, remained significant after correction for multiple testing in the group of unaffected siblings. These variants included two SNPs in the AKT1 gene (rs2494732 and rs1130233) and one SNP in the leucine-rich repeat transmembrane protein 1 (LRRTM1) gene (rs673871). There were significant interactions between the AKT1 rs2494732 polymorphism and lifetime or restricted cannabis use on psychotic symptoms in the subsequent case-only analysis. In the case-sibling design, patients with the AKT1 rs2494732 CC genotype displayed approximately twofold higher odds of being diagnosed with psychotic disorder when having used cannabis in comparison with TT homozygotes. However, no significant interactions were found in the case-control design. Importantly, DiForti et al. [ref. 44] confirmed these findings for the AKT1 rs2494732 gene polymorphism. The AKT1 rs2494732 CC homozygotes with a history of cannabis use showed a greater than twofold increase in the likelihood of having a psychotic disorder in comparison with the AKT1 rs2494732 TT homozygotes. Among daily cannabis users, individuals with the AKT1 rs2494732 CC genotype demonstrated a sevenfold increase in the odds of developing psychosis compared to the AKT1 rs2494732 TT homozygotes. Similarly, in the experimental study by Bhattacharyya et al. [ref. 43], healthy individuals with the AKT1 rs1130233 GG genotype and the dopamine transporter (DAT1) 9-repeat allele had significantly higher increase in the levels of psychotic symptoms after acute exposure to delta-9-THC compared to subjects with other genotypes.
In single studies, the effects of interactions between cannabis use and polymorphisms in the genes encoding dopamine D2 receptor (DRD2) [ref. 29], FK506-binding protein 5 (FKBP5) [ref. 34], and brain-derived neurotrophic factor (BDNF) [ref. 50] were investigated. In a case-control study of FEP patients, Colizzi et al. [ref. 29] found that cannabis users with the DRD2 rs1076560 T allele had a threefold increase in psychosis risk compared to GG homozygotes. In daily users, T allele carriers had a fivefold increase in psychosis risk compared to GG homozygotes. In healthy subjects, daily users with T allele had higher schizotypy compared to cannabis-naïve T allele carriers, cannabis users with GG genotype and cannabis-naïve GG homozygotes. Cannabis users with T allele had lower working memory performance in comparison with other groups. Ajnakina et al. [ref. 34] studied the effects of the rs1360780 polymorphism in the FKBP5 gene in 291 first-episode psychosis patients and 218 controls. They found the association between this polymorphism and psychosis risk after co-varying for environmental factors including parental separation and cannabis use. However, no significant interactions between genetic variation in FKBP5 gene and cannabis use on psychosis risk were found. Finally, in female schizophrenia patients, cannabis use was associated with earlier age of psychosis onset in the BDNF 66Met allele carriers, but not in the BDNF 66Val/Val homozygotes. In male patients, cannabis use was associated with earlier age of psychosis onset, regardless of the BDNF Val66Met genotype. The main effect of the BDNF Val66Met genotype on age of psychosis onset was not significant in the whole group as well as in males and females.
Interactions between genetic factors and cannabis use were tested only in one study on BD patients [ref. 101]. Authors found that the interaction between the serotonin-transporter-linked polymorphic region (5-HTTLPR) S allele and childhood sexual abuse increased odds of cannabis abuse or dependence. Cannabis abuse or dependence and the 5-HTTLPR S allele, but not childhood sexual abuse, were significantly more frequent in those patients with lifetime occurrence of psychotic symptoms.
Gene × Stress Interactions
According to our review, 12 studies have evaluated interactions between exposure to childhood trauma and genetic factors in schizophrenia spectrum disorders [ref. 21, ref. 22, ref. 31, ref. 33–ref. 36, ref. 38, ref. 41, ref. 42, ref. 45, ref. 123]. The vast majority of studies suggest significant interactions between genetic underpinnings and exposure to childhood adversities, stressful life events or recent stressors and genetic factors, with only three studies reporting negative findings [ref. 25, ref. 35, ref. 45].
In three studies [ref. 21, ref. 33, ref. 123], interactions between a history of childhood trauma and the BDNF Val66Met polymorphism were addressed, showing positive findings. Aas et al. [ref. 33] found an additive effect of the BDNF 66Met allele and a history of childhood trauma on reduced levels of BDNF mRNA as well as CA2/3 and CA4 subfield volumes of dentate gyrus in the hippocampus. Similar effects of interactions between the BDNF Val66Met polymorphism and childhood abuse, but not childhood neglect, on positive psychotic-like experiences have been found in a non-clinical study [ref. 123]. Another non-clinical study revealed that the BDNF 66Val allele, especially in male twins, was associated with higher vulnerability of the effects of childhood trauma on psychotic experiences, while in the group of female twins this association was driven by the BDNF 66Met allele [ref. 21]. There are also studies addressing the effects of interaction between variability in the FKBP5 gene and childhood trauma on psychosis phenotypes in clinical and non-clinical populations [ref. 22, ref. 31, ref. 34, ref. 38]. In the study by Cristobal-Narvaez et al. [ref. 22], there was a significant effect of the interaction between the FKBP5 risk haplotype (composed of three SNPs: rs3800373, rs9296158, and rs1360780) and childhood bullying on positive psychotic-like experiences, paranoia, and negative affect. The childhood bullying × FKBP5 haplotype interaction moderated the association of social stress appraisal with psychotic-like experiences and negative affect in daily life. Specifically, this association was significantly increased in those with the risk haplotype, but not in individuals without the risk haplotype. Collip et al. [ref. 38] studied the effects of SNPs in the FKBP5 gene (rs9296158, rs4713916, rs1043805, and rs1360780) in the general population twins, patients with psychosis, unaffected siblings of patients with psychosis and matched controls. The authors found a significant interaction between the rs9296158 and rs4713916 polymorphisms and childhood trauma on psychotic symptoms and cortisol levels in the twin sample. Similar findings were obtained for the rs4713916 polymorphism in siblings and for rs9296158 in patients. Specifically, the A allele carriers at both polymorphisms were most vulnerable to childhood trauma. In a case-control study of FEP patients mentioned above, Ajnakina et al. [ref. 34] demonstrated that the FKBP5 rs1360780 polymorphism was associated with the risk of psychosis only after adjustment for environmental factors. Authors reported a significant effect of the interaction between the FKBP5 rs1360780 polymorphism and parental separation on psychosis risk. In one study [ref. 31], the FKBP5 gene polymorphisms were studied with respect to cognitive performance in patients with schizophrenia and healthy controls. Authors found significant main effects of the rs1360870 genotype and childhood trauma as well as a significant interaction between these variables affecting attention in both groups (CC homozygotes performed worse in the context of childhood trauma). Additionally, there were significant main effects of this polymorphism on global cognition in schizophrenia patients (TT homozygotes performed worse). Furthermore, McCarthy-Jones et al. [ref. 36] found in a large sample of schizophrenia spectrum patients, the interaction between variation in the forkhead box protein 2 (FOXP2) gene and childhood trauma in predicting a lifetime history of auditory verbal hallucinations. Emotional abuse was found to interact with the rs1456031 polymorphism in patients with CC genotype in predicting higher levels of auditory verbal hallucinations.
Another line of studies focused on the interactions between recent or daily life stressors and genetic factors [ref. 24, ref. 37, ref. 46, ref. 62, ref. 63, ref. 65, ref. 124]. In the majority of these studies, the effects of variation in the COMT gene were addressed [ref. 46, ref. 63, ref. 65, ref. 124]. Van Winkel et al. [ref. 65] found that subjects with the COMT 158 Met/Met genotype had greater increase in overall psychotic experiences in response to daily stressors compared to those with Val/Met and Val/Val genotypes, both among patients with psychosis and healthy controls. Similarly, in the study of patients with non-affective psychosis [ref. 46], which also included the analysis of the methylenetetrahydrofolate reductase (MTHFR) gene polymorphisms (C677T and A1298C variants), patients with the MTHFR T allele, COMT Met/Met homozygotes showed the largest increases in psychotic experiences in response to stress. In patients, who were the MTHFR CC homozygotes, there was no interaction between the COMT Val158Met polymorphism and stress on psychotic experiences. There was also no moderating effect of the MTHFR A1298C polymorphism on the interaction between the COMT Val158Met polymorphism and stress. On the contrary, a study of female twins revealed that carriers of the COMT 158 Val allele displayed more feelings of paranoia in response to event stress compared with Met carriers [ref. 63]. This study also included effects of the BDNF Val66Met polymorphism, showing that carriers of the BDNF 66Met allele presented more social stress-induced paranoia than individuals with the Val/Val genotype. Similarly, Stefanis et al. [ref. 124] revealed that carriers of the COMT 158Val allele were more sensitive to psychosis inducing effects of stress exposure during army recruitment in comparison with the Met/Met homozygotes.
Finally, single studies focused on interacting effects of genes involved in DNA methylation, the BDNF gene and the neuregulin 1 (NRG1) gene. Pishva et al. [ref. 37], who studied the effects of DNA methylation genes in clinical and non-clinical samples, found that three SNPs in the DNA methyltransferase 3A (DNMT3A) gene (rs11683424, rs1465764, rs1465825) and one in the MTHFR (rs1801131) moderated the effect of stressful events on negative affect. The effects of the DNMT3A rs11683424 polymorphism were consistent in the majority of studied samples. An interesting outcome measure has been selected in the study by Gattere et al. [ref. 24], who assessed caloric intake in patients with early psychosis, ARMS individuals and healthy controls with respect to the BDNF Val66Met polymorphism. Authors found that perceived stress was not associated with calorie intake in healthy controls. ARMS subjects with the Met allele and low perceived stress presented with increased caloric intake, while those with high levels of perceived stress presented with decreased caloric intake. In patients with early psychosis, perceived stress was not associated with calorie intake. Perceived stress was associated with food craving in patients with psychosis. A similar association was present in ARMS subjects and healthy controls who were Val/Val homozygotes. Finally, Keri et al. [ref. 62] studied the effects of variation in the NRG1 gene with respect to psychosocial stress in terms of conflict-related family interactions. Authors explored odd and unusual thought content during neutral and conflict-related family interactions with one of the family members: mothers, fathers, wives, husbands, and siblings in patients with schizophrenia. Patients with the NRG1 rs6994992 TT genotype showed more unusual thoughts during conflict-related interactions than patients with CT and CC genotypes. There were no significant differences between the NRG1 CT and CC patients. There were also no significant differences among patients with different NRG1 genotypes during neutral interactions.
Effects of interactions between genetic factors and stressful experiences on clinical characteristics of patients with BD have been tested in six studies [ref. 101–ref. 103, ref. 107, ref. 108, ref. 125]. All of these studies examined the impact of childhood trauma except for the study by Hosang et al. [ref. 102], which assessed traumatic life events. Negative results were published only by Breen et al. [ref. 107], who found no significant interactions between variation in hypothalamus-pituitary-adrenal (HPA) axis genes and childhood sexual or physical abuse on lifetime occurrence of suicide attempts. In this study, effects of the BDNF Val66Met polymorphism with respect to lifetime traumatic experiences and the severity of worst episodes of BD were explored. Authors revealed that the BDNF 66Met allele carriers with higher levels of stressful life events had a higher severity of the worst depression ever. The effects of the BDNF Val66Met polymorphism were also examined in the study by Miller et al. [ref. 125], who demonstrated that the BDNF 66Met allele carriers with a history of childhood sexual abuse had significantly higher BD severity and chronicity as well as earlier age of onset. However, these results appeared to be insignificant after controlling for potential confounders. In three studies [ref. 101, ref. 103, ref. 126], the impact of variation in monoaminergic genes was addressed. De Pradier et al. [ref. 101] found that the interaction between the 5-HTTLPR S allele and childhood sexual abuse increased odds of cannabis abuse or dependence. Another study [ref. 103] revealed that the COMT 158Val allele was associated with higher levels of schizotypy in BD patients exposed to higher levels of childhood trauma. There were no significant main effects of the COMT Val158Met polymorphism on the levels of schizotypy. Finally, in one study [ref. 108], patients with the toll-like receptor 2 (TLR2) rs3804099 TT genotype and a history of sexual abuse had earlier age of BD onset compared to other patients. However, these results were non-significant after taking into account the effects of potential confounders.
Gene × Season of Birth Interactions
Interactions between genetic factors and seasonality of birth were tested in four studies on patients with schizophrenia spectrum disorders [ref. 54, ref. 71–ref. 73]. In the study by Narita et al. [ref. 73], the presence of the HLA-DR1 allele was associated with increased incidence of winter births (February–March) in schizophrenia patients. In turn, Tochigi et al. [ref. 72] revealed no significant association between genetic variation in the HLA-A gene (A24 and A26 variants) and winter birth (December–March) in schizophrenia patients. Chotai et al. [ref. 71] investigating three SNPs—one in the tryptophan hydroxylase 1 (TPH1) gene (A218C), 5-HTTLPR L/S polymorphism, and the dopamine D4 receptor (DRD4) 7-repeat allele polymorphism—demonstrated that the frequency of the DRD4 7-repeat allele showed one-cyclic season of birth variation in women with schizophrenia. However, no significant interactions were found for the TPH1 A218C and 5-HTTLPR L/S polymorphisms. In the study by Muntjewerff et al. [ref. 54], there was no significant interaction between the MTHFR C677T polymorphism and winter birth on schizophrenia susceptibility.
There are two studies looking into the interaction between seasonality of birth and genetic factors in BD [ref. 71, ref. 104]. The abovementioned study by Chotai et al. [ref. 71] also included patients with BD demonstrating that allelic frequencies did not show any significant variation with respect to seasons of birth defined as four 3-month periods beginning in January. However, the analysis of one-cyclic month of birth variations showed that the TPH1 allele A had a positive peak around the birth month December and a negative peak around June in men with BD, but not in women with BD. There were more cases of BD among men with the TPH1 allele A born in between November and January and less cases of BD among women with the TPH1 allele A born between February and July in comparison with healthy controls. Moreover, analysis of two cycles per year showed differences in the DRD4 gene variations both among women and men with BD. Finally, in one study the HLA-G 14 bp ins/del polymorphism was investigated with respect to seasonality of birth in patients with BD [ref. 104]. The authors found that the HLA-G ins/ins genotype was significantly less frequent in patients with BD. The prevalence of this genotype was significantly lower in patients born in the winter season.
Gene × Infectious Factors
Interactions between genetic and infectious factors were tested in two studies on schizophrenia spectrum phenotypes [ref. 51, ref. 66]. The study by Demontis et al. [ref. 51] revealed significant effects of the interactions between two SNPs in the glutamate ionotropic receptor NMDA type subunit 2B (GRIN2B) gene (rs1805539 and rs1806205) and maternal herpes simplex virus type 2 (HSV-2) seropositivity on schizophrenia risk. The latter one [ref. 66] demonstrated that variation in the MHC Class I Polypeptide-Related Sequence B (MICB) gene may interact with cytomegalovirus (CMV) and herpes simplex virus type 1 (HSV-1) seropositivity, influencing schizophrenia susceptibility.
In two studies on BD patients [ref. 100, ref. 110], the presence of antibodies to HSV-1 and Toxoplasma gondii was analyzed. Dickerson et al. [ref. 100] found that the COMT 158Val/Val genotype and HSV-1 seropositivity were independent predictors of lower global cognitive performance in patients with BD. Patients with both the COMT 158Val/Val genotype and HSV-1 seropositivity were 85 times more likely to be in the lowest quintile of global cognitive performance. In turn, Oliveira et al. [ref. 110] revealed a trend toward significant interaction between Toxoplasma gondii seropositivity and the TLR2 (rs3804099) gene polymorphism in conferring the risk of BD.
Gene × Obstetric Complications Interactions
Interactions between genetic factors and obstetric complications were assessed in four studies on schizophrenia patients [ref. 27, ref. 32, ref. 56, ref. 64]. In the study by Nicodemus et al. [ref. 64], patients with schizophrenia spectrum disorders and obstetric complications were more likely to have minor allele at the AKT1 rs2494735 and rs1130233 polymorphisms, major allele at the BDNF rs2049046 polymorphism and minor allele at the BDNF rs76882600, minor allele at the dystrobrevin binding protein 1 (DTNBP1) gene (rs875462), and minor allele at the glutamate metabotropic receptor 3 (GRM3) gene (rs7808623). In turn, Ursini et al. [ref. 27] revealed that the BDNF Val66Met polymorphism, together with DNA methylation within this polymorphic site, might interact with obstetric complications influencing intermediate schizophrenia phenotypes, such as working memory impairment and alterations in dorsolateral prefrontal cortex activity. Haukvik et al. [ref. 56] revealed a significant effect of the interaction between the GRM3 rs13242038 polymorphism and severe obstetric complications on hippocampal volumes in patients with schizophrenia and healthy controls. Finally, in one study [ref. 32], high birth weight was associated with schizophrenia risk in subjects homozygous for risk alleles in a four-SNP haplotype spanning the NudE Neurodevelopment Protein 1 (NDE1) gene and one of its constituent SNPs (rs4781678).
Discussion
The majority of studies addressing G × E interactions in schizophrenia spectrum phenotypes and BD have focused on the effect of variation in the COMT, BDNF, and FKBP5 genes, showing interactions with cannabis use and childhood trauma. Results of studies described in this systematic review should be discussed in frame of distinct G × E interactions models that might explain various scenarios of causality: (1) the genotype gives rise to the phenotype as the consequence of environmental exposure; however, when a risk genotype is not present, the phenotype might be expressed in case of a high-level exposure to environmental factor; (2) the genotype increases effects of environmental risk factor; however, when environmental exposure does not appear, the effects of genotype expression remain silent; (3) the environmental factor increases the effects of the high-risk genotype, but not the effects of the low-risk genotype; (4) both genetic and environmental risk factors are required to trigger the expression of the phenotype; and (5) both environmental and genetic risk factors have some effect on the phenotype; however, if they appear together the risk is higher or lower than in the situation, when they occur alone [ref. 18].
The effects of G × E interactions become even more complicated in psychiatric research, when the impact of genetic variation on personality traits, which conditions risky behaviors, is taken into account. Indeed, it might be hypothesized that some genetic factors might make individuals more prone to engage in high-risk environments [ref. 127]. This scenario might be theoretically relevant for studies investigating interactions between the COMT Val158Met polymorphism and cannabis use. The Val allele at codon 158 is known to increase enzymatic activity and lead to a faster breakdown of dopamine [ref. 128]. It has been hypothesized that the Val allele increases the risk of psychosis via depleting prefrontal dopamine availability that in turn increases mesolimbic dopaminergic activity in a feedback loop [ref. 129]. In turn, Δ9-tetrahydrocannabinol (THC), an active ingredient of Cannabis sativa, is known to enhance mesolimbic dopaminergic activity contributing to the development of psychosis [ref. 130]. Hypodopaminergic prefrontal activity, which is closely related to blunted reward processing and cognitive decline [ref. 131], has been widely observed in subjects at risk of psychosis and might explain high levels of comorbidity between cannabis abuse or dependence and schizophrenia spectrum disorders.
Although a few repeatedly tested G × E interactions can be indicated based on this systematic review, current evidence does not allow to generalize findings due to methodological heterogeneity and limitations as well as a variety of explored outcome variables. One of the main methodological problems of studies addressing G × E is sample size. Roughly speaking, a general approach states that sample sizes required to detect interactions should be at least four times higher compared to sample sizes of studies that aim to detect main effects of comparable magnitude [ref. 132, ref. 133]. However, precise calculations that take into account genotype distribution, frequency of environmental exposure, precision of measurement, and validity of hypothesis behind a potential interaction (for a summary of various approaches see [ref. 134]), might greatly improve statistical power. Interesting simulations have been made by Uher [ref. 135], who demonstrated for instance that an interaction of moderate effect size with the genotype that is present in only 5% of the population would require 5200 participants to achieve the power of 80%. However, such requirements are a function of measurement reliability of environmental exposure. For instance, a decrease of 20% in the reliability of environment measures equates to losing about 50% of the sample. This simulation shows the importance of measurement accuracy in designing studies on G × E interactions.
Previous studies on G × E interactions have used a variety of measures for assessment of environmental exposure. The majority of them have focused on retrospective and self-report measures. This approach is particularly controversial in light of potential reporting bias driven by self-reports of childhood trauma or substance use. Previous studies with longer test-retest intervals have revealed that inconsistency rates in case of childhood trauma self-reports might reach nearly 40% [ref. 136, ref. 137]. These studies have suggested that such factors as age, educational attainment, depressive symptoms, psychological distress, and chronic stress might impact consistency self-reports of childhood trauma. On the contrary, the Aetiology and Ethnicity of Schizophrenia and Other Psychoses (ÆSOP) study demonstrated that self-reports of childhood trauma in FEP patients remained stable over a 7-year follow-up period and were not influenced by the levels of depressive and psychotic symptoms [ref. 138]. Moreover, there are longitudinal studies that have found exposure to stressful events before the measurement of psychotic experiences or onset of psychotic disorder [ref. 139–ref. 141]. Therefore, caution should be taken on the way childhood adversities are assessed. For instance, variables that have been associated with reporting consistency should be included in statistical models analyzing G × E interactions. It has been suggested that measurement accuracy in case of childhood trauma might be increased by combining different sources of information, such as self-reports, case notes, or court records [ref. 142]. In turn, reporting accuracy of current or recent substance use might be improved by inclusion of urinary screening tests.
Another important point in G × E interactions research is that a number of individual characteristics and environmental factors are closely interrelated. For instance, it has been proposed that known schizophrenia risk factors, such as childhood trauma, urban upbringing, low intelligence quotient, migration and substance use have a common denominator—social defeat, which is defined as individual appraisal of being excluded from the society [ref. 143]. Moreover, it has been shown that childhood adversities combine with subsequent cannabis use and further increase odds of psychotic experiences—the effect described as an environment × environment interaction [ref. 144, ref. 145]. The social defeat paradigm points to the consideration, whether environmental risk factors for schizophrenia should be captured in collective measures. Emerging evidence shows that the development of the so-called polyenviromic risk scores [ref. 146] might hold a great promise in recognizing a missing environmental contribution. This approach has been successfully implemented in the field of GWASs studies, which developed the polygenic risk score, showing that this measure is associated with schizophrenia risk and its clinical characteristics [ref. 147]. A recent study by Padmanabhan et al. [ref. 146], for the first time, demonstrated that an aggregate score of environmental exposures (winter or spring birth, cannabis abuse, advanced paternal age, obstetric and perinatal complications, physical and sexual abuse, neglect, and parental death) predicted conversion from familial high-risk state to psychosis [ref. 146].
It should be also noted that the impact of environmental exposure might be different in distinct time points of brain development, at least theoretically. Indeed, it is widely known that various neurodevelopmental processes, such as neurogenesis, neuronal migration, gliogenesis, synaptogenesis, myelination, and synaptic pruning have specific critical windows in prenatal and postnatal development [ref. 148, ref. 149]. Seasonality of birth, advanced paternal age, and obstetric complications are considered to act as early insults and may share similar mechanisms, contributing to the pathogenesis of schizophrenia [ref. 150]. Seasonality of birth, a proxy measure of prenatal infections, might increase the risk of schizophrenia in offspring via maternal immune activation. It has been shown that elevated maternal levels of cytokines might be associated with increased risk of schizophrenia in the offspring [ref. 151, ref. 152]. Changes in brain regions that are typical for schizophrenia patients, such as reduced cortical thickness, decreased hippocampal, prefrontal cortical and striatal volumes, and enlarged ventricles, together with reduced density of Purkinje neurons, have been also observed in the adult offspring from animal studies of maternal immune activation [ref. 153]. In addition, maternal immune activation has been found to impact dopaminergic and serotoninergic neurotransmission [ref. 153]. Obstetric complications leading to hypoxia might also indirectly act via immune-inflammatory mechanisms [ref. 150]. Another hypothesis is that hypoxia may induce expression of schizophrenia susceptibility genes. Interestingly, a recent systematic analysis of schizophrenia susceptibility genes revealed that 55% of candidate genes might be associated with ischemia-hypoxia response [ref. 154]. Further support for the association with perinatal hypoxia originates from neuroimaging studies, showing reduced gray matter volumes and increased cerebrospinal fluid space in schizophrenia patients and their siblings with a history of obstetric complications [ref. 155]. Similarly, exposure to obstetric complications has been associated with lower amygdala and hippocampal volumes in patients with BD [ref. 156]. Finally, the effect of advanced paternal age on schizophrenia risk can be explained by accumulation of de novo mutations in paternal sperm DNA or epigenetic alterations [ref. 157]. Environmental factors that act in later life, including childhood trauma and cannabis abuse might also affect brain development. A history of childhood trauma has been associated with smaller volumes of distinct brain regions, including, i.e., the corpus callosum, hippocampus, and amygdala as well as distinct reward circuits [ref. 158]. Previous studies have demonstrated that early life stress may impact psychosis risk via various biological mechanisms, such as HPA axis alterations, aberrant immune-inflammatory response, dysregulation of dopaminergic neurotransmission, and epigenetic processes [ref. 159]. Similarly, cannabis abuse might impact the development of various brain regions. The psychoactive ingredient of cannabis, THC, stimulates midbrain dopaminergic activity [ref. 130, ref. 160]. Chronic cannabis abuse has been associated with functional and structural alterations in the hippocampus and amygdala, which are the brain regions implicated in the pathophysiology of psychosis [ref. 161]. However, it is important to note that later life environmental insults have been found to act in a dose-dependent manner and are believed to differentially impact brain development in various age groups [ref. 130, ref. 158]. This point should be considered in analyzing results of studies on G × E interactions in schizophrenia spectrum phenotypes and BD. Given that age of exposure might be important in determining the impact of environmental exposure, a simplistic analysis of traumatic stress or substance use in the childhood or the whole lifespan, using dichotomous or continuous variables, might lead to overlooking more specific G × E interactions.
Moreover, controversy appears in the comparison of relevant G x E interactions or even main effects of environmental exposure between schizophrenia and BD as well as other psychiatric disorders or non-clinical samples. Indeed, it becomes increasingly apparent that similar G × E interactions might contribute to a broad spectrum of phenotypes. Addressing this issue should be posited as one of main directions for future studies. One direction is to implement the concept of endophenotypes, which states that there are a number of intermediate phenotypes, associated with particular disorders in the population, which are observed in non-affected individuals, exert familial co-segregation and heritability, can be observed in related disorders as well as have state-independent manifestation [ref. 162–ref. 164]. Following this broad definition, several biochemical, cognitive, behavioral, neurostructural, and neurofunctional endophenotypes have been proposed for schizophrenia and BD in order to conceptualize overlapping characteristics that fall beyond a categorical distinction [ref. 3].
Overlapping G × E interactions in various psychiatric disorders also raise a question whether G × E interactions operate through single clinical outcomes or more complex mechanisms. Recent theoretical accounts have pointed out to the need of more integrative and dynamic view and propose models that may explain the mechanisms of how the G × E interactions contribute to psychosis. On the one hand, these theories need to take into account the effects of the environment on structural and functional brain characteristics, neuroendocrine functioning, attachment styles, and patterns of affective and cognitive processing that may shape risk for later psychopathology. On the other hand, mechanisms by which genetic variation may increase susceptibility to environmental stressors should also be tested. The underlying pathophysiological pathways of these mechanisms need to be established in order to develop a priori G × E research paradigms and proper methodological designs.
For instance, Howes and Murray [ref. 165] suggested that G × E interactions contribute to dopaminergic dysregulation and lead to psychotic symptoms through a mediating role of biased information processing (i.e., cognitive biases). Indeed, with regard to cannabis use, some findings suggest that cognitive dysfunctions mediate the association between cannabis use and psychotic experiences [ref. 166]. Similarly, social adversities have been suggested to shape the risk of psychosis via biased information processing, which was confirmed in the very recent study showing a full mediation of attention to threat and external attributions biases in the relationship between traumatic life events and psychosis proneness in a non-clinical sample [ref. 167]. Several studies suggest that cognitive impairments in working memory and executive functions are linked to polymorphisms in the COMT gene [ref. 168, ref. 169] and this association might be the mechanism explaining G × E interactions. For instance, a recent study has shown possible epigenetic modulation of the expression of the COMT Val158Met polymorphism and subsequent effects on the relationship between traumatic life events and cognition in schizophrenia [ref. 170]. However, the impact of G × E interactions on biased information processing (cognitive biases) that may be specific for psychotic or BD symptoms has not been investigated so far.
Traumatic life events may also increase the risk of psychosis since they trigger disturbances in emotional and cognitive regulative processes. In this context, an interesting approach of environmental conceptualization using the experimental sampling methodology (ESM) has been adopted in some studies discussed in this systematic review. In brief, ESM can be defined as “a research procedure that aims to provide a systematic set of self-reports obtained on random occasions about what people do and feel during waking hours of a normal week” [ref. 171]. Hence, the ESM provides an excellent tool to investigate environmental factors and its dynamic impact on well-being via cognitive-emotional processes. For instance, it has been found that a history of trauma and daily life stress impact on psychosis risk through the impaired cognitive [ref. 172] and emotional regulative processes [ref. 173] as well as a lack of resilience factors. Studies addressing G × E interactions that utilized the ESM methodology may be then interpreted rather as a dynamic phenomenon, which results in disrupted regulative processes that lead to psychotic experiences, than a simple result of direct influence of G × E on psychosis. Therefore, studies in this field may greatly benefit from combining G × E interactions and potential mediating mechanisms related to cognitive-emotional regulation in predicting psychosis or BD.
Although there is a great methodological heterogeneity in previous studies on G × E interactions in schizophrenia and BD, some findings, especially related to childhood trauma and cannabis use, have been replicated. In light of a rapid progress of GWASs in the field of BD and schizophrenia, it should be recommended and expected that future studies will focus on combining data from GWASs with environmental measures to provide more comprehensive insights into G × E interactions. This approach is currently ongoing under the European Network of National Schizophrenia Networks studying Gene-Environment Interactions (EU-GEI) Initiative [ref. 174]. In addition, future studies should also include assessment of epigenetic processes that bridge a gap between environmental exposure and genetic underpinnings.
References
- AG Cardno, II Gottesman. Twin studies of schizophrenia: from bow-and-arrow concordances to star wars Mx and functional genomics. Am J Med Genet, 2000. [PubMed]
- P McGuffin, F Rijsdijk, M Andrew, P Sham, R Katz, A Cardno. The heritability of bipolar affective disorder and the genetic relationship to unipolar depression. Arch Gen Psychiatry, 2003. [PubMed]
- B Misiak, D Frydecka, JK Rybakowski. Editorial: endophenotypes for schizophrenia and mood disorders: implications from genetic, biochemical, cognitive, behavioral, and neuroimaging studies. Front Psych, 2016
- 4.Ripke S, Schizophrenia Working Group of the Psychiatric Genomics Consortium (2014) Biological insights from 108 schizophrenia-associated genetic loci. Nature 511(7510):421–427. doi:10.1038/nature13595
- 5.Psychiatric GWAS Consortium Bipolar Disorder Working Group (2011) Large-scale genome-wide association analysis of bipolar disorder identifies a new susceptibility locus near ODZ4. Nat Genet 43 (10):977–983. doi:10.1038/ng.943
- E Agerbo, PF Sullivan, BJ Vilhjalmsson, CB Pedersen, O Mors, AD Borglum, DM Hougaard, MV Hollegaard, S Meier, M Mattheisen, S Ripke, NR Wray, PB Mortensen. Polygenic risk score, parental socioeconomic status, family history of psychiatric disorders, and the risk for schizophrenia: a Danish population-based study and meta-analysis. JAMA Psychiat, 2015
- AV Kotlar, KB Mercer, ME Zwick, JG Mulle. New discoveries in schizophrenia genetics reveal neurobiological pathways: a review of recent findings. Eur J Med Genet, 2015. [PubMed]
- SH Lee, S Ripke, BM Neale, SV Faraone, SM Purcell, RH Perlis, BJ Mowry, A Thapar, ME Goddard, JS Witte, D Absher, I Agartz, H Akil, F Amin, OA Andreassen, A Anjorin, R Anney, V Anttila, DE Arking, P Asherson, MH Azevedo, L Backlund, JA Badner, AJ Bailey, T Banaschewski, JD Barchas, MR Barnes, TB Barrett, N Bass, A Battaglia, M Bauer, M Bayes, F Bellivier, SE Bergen, W Berrettini, C Betancur, T Bettecken, J Biederman, EB Binder, DW Black, DH Blackwood, CS Bloss, M Boehnke, DI Boomsma, G Breen, R Breuer, R Bruggeman, P Cormican, NG Buccola, JK Buitelaar, WE Bunney, JD Buxbaum, WF Byerley, EM Byrne, S Caesar, W Cahn, RM Cantor, M Casas, A Chakravarti, K Chambert, K Choudhury, S Cichon, CR Cloninger, DA Collier, EH Cook, H Coon, B Cormand, A Corvin, WH Coryell, DW Craig, IW Craig, J Crosbie, ML Cuccaro, D Curtis, D Czamara, S Datta, G Dawson, R Day, EJ De Geus, F Degenhardt, S Djurovic, GJ Donohoe, AE Doyle, J Duan, F Dudbridge, E Duketis, RP Ebstein, HJ Edenberg, J Elia, S Ennis, B Etain, A Fanous, AE Farmer, IN Ferrier, M Flickinger, E Fombonne, T Foroud, J Frank, B Franke, C Fraser, R Freedman, NB Freimer, CM Freitag, M Friedl, L Frisen, L Gallagher, PV Gejman, L Georgieva, ES Gershon, DH Geschwind, I Giegling, M Gill, SD Gordon, K Gordon-Smith, EK Green, TA Greenwood, DE Grice, M Gross, D Grozeva, W Guan, H Gurling, L De Haan, JL Haines, H Hakonarson, J Hallmayer, SP Hamilton, ML Hamshere, TF Hansen, AM Hartmann, M Hautzinger, AC Heath, AK Henders, S Herms, IB Hickie, M Hipolito, S Hoefels, PA Holmans, F Holsboer, WJ Hoogendijk, JJ Hottenga, CM Hultman, V Hus, A Ingason, M Ising, S Jamain, EG Jones, I Jones, L Jones, JY Tzeng, AK Kahler, RS Kahn, R Kandaswamy, MC Keller, JL Kennedy, E Kenny, L Kent, Y Kim, GK Kirov, SM Klauck, L Klei, JA Knowles, MA Kohli, DL Koller, B Konte, A Korszun, L Krabbendam, R Krasucki, J Kuntsi, P Kwan, M Landen, N Langstrom, M Lathrop, J Lawrence, WB Lawson, M Leboyer, DH Ledbetter, PH Lee, T Lencz, KP Lesch, DF Levinson, CM Lewis, J Li, P Lichtenstein, JA Lieberman, DY Lin, DH Linszen, C Liu, FW Lohoff, SK Loo, C Lord, JK Lowe, S Lucae, DJ MacIntyre, PA Madden, E Maestrini, PK Magnusson, PB Mahon, W Maier, AK Malhotra, SM Mane, CL Martin, NG Martin, M Mattheisen, K Matthews, M Mattingsdal, SA McCarroll, KA McGhee, JJ McGough, PJ McGrath, P McGuffin, MG McInnis, A McIntosh, R McKinney, AW McLean, FJ McMahon, WM McMahon, A McQuillin, H Medeiros, SE Medland, S Meier, I Melle, F Meng, J Meyer, CM Middeldorp, L Middleton, V Milanova, A Miranda, AP Monaco, GW Montgomery, JL Moran, D Moreno-De-Luca, G Morken, DW Morris, EM Morrow, V Moskvina, P Muglia, TW Muhleisen, WJ Muir, B Muller-Myhsok, M Murtha, RM Myers, I Myin-Germeys, MC Neale, SF Nelson, CM Nievergelt, I Nikolov, V Nimgaonkar, WA Nolen, MM Nothen, JI Nurnberger, EA Nwulia, DR Nyholt, C O’Dushlaine, RD Oades, A Olincy, G Oliveira, L Olsen, RA Ophoff, U Osby, MJ Owen, A Palotie, JR Parr, AD Paterson, CN Pato, MT Pato, BW Penninx, ML Pergadia, MA Pericak-Vance, BS Pickard, J Pimm, J Piven, D Posthuma, JB Potash, F Poustka, P Propping, V Puri, DJ Quested, EM Quinn, JA Ramos-Quiroga, HB Rasmussen, S Raychaudhuri, K Rehnstrom, A Reif, M Ribases, JP Rice, M Rietschel, K Roeder, H Roeyers, L Rossin, A Rothenberger, G Rouleau, D Ruderfer, D Rujescu, AR Sanders, SJ Sanders, SL Santangelo, JA Sergeant, R Schachar, M Schalling, AF Schatzberg, WA Scheftner, GD Schellenberg, SW Scherer, NJ Schork, TG Schulze, J Schumacher, M Schwarz, E Scolnick, LJ Scott, J Shi, PD Shilling, SI Shyn, JM Silverman, SL Slager, SL Smalley, JH Smit, EN Smith, EJ Sonuga-Barke, D St Clair, M State, M Steffens, HC Steinhausen, JS Strauss, J Strohmaier, TS Stroup, JS Sutcliffe, P Szatmari, S Szelinger, S Thirumalai, RC Thompson, AA Todorov, F Tozzi, J Treutlein, M Uhr, EJ van den Oord, G Van Grootheest, J Van Os, AM Vicente, VJ Vieland, JB Vincent, PM Visscher, CA Walsh, TH Wassink, SJ Watson, MM Weissman, T Werge, TF Wienker, EM Wijsman, G Willemsen, N Williams, AJ Willsey, SH Witt, W Xu, AH Young, TW Yu, S Zammit, PP Zandi, P Zhang, FG Zitman, S Zollner, B Devlin, JR Kelsoe, P Sklar, MJ Daly, MC O’Donovan, N Craddock, PF Sullivan, JW Smoller, KS Kendler, NR Wray. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat Genet, 2013. [PubMed]
- JX Van Snellenberg, T de Candia. Meta-analytic evidence for familial coaggregation of schizophrenia and bipolar disorder. Arch Gen Psychiatry, 2009. [PubMed]
- P Skudlarski, DJ Schretlen, GK Thaker, MC Stevens, MS Keshavan, JA Sweeney, CA Tamminga, BA Clementz, K O’Neil, GD Pearlson. Diffusion tensor imaging white matter endophenotypes in patients with schizophrenia or psychotic bipolar disorder and their relatives. Am J Psychiatry, 2013. [PubMed]
- SK Hill, JL Reilly, RS Keefe, JM Gold, JR Bishop, ES Gershon, CA Tamminga, GD Pearlson, MS Keshavan, JA Sweeney. Neuropsychological impairments in schizophrenia and psychotic bipolar disorder: findings from the Bipolar-Schizophrenia Network on Intermediate Phenotypes (B-SNIP) study. Am J Psychiatry, 2013. [PubMed]
- Z Wang, SA Meda, MS Keshavan, CA Tamminga, JA Sweeney, BA Clementz, DJ Schretlen, VD Calhoun, S Lui, GD Pearlson. Large-scale fusion of gray matter and resting-state functional MRI reveals common and distinct biological markers across the psychosis spectrum in the B-SNIP Cohort. Front Psych, 2015
- AG Cardno, MJ Owen. Genetic relationships between schizophrenia, bipolar disorder, and schizoaffective disorder. Schizophr Bull, 2014. [PubMed]
- C Marangoni, M Hernandez, GL Faedda. The role of environmental exposures as risk factors for bipolar disorder: a systematic review of longitudinal studies. J Affect Disord, 2016. [PubMed]
- K Dean, RM Murray. Environmental risk factors for psychosis. Dialogues Clin Neurosci, 2005. [PubMed]
- J van Os, BP Rutten, R Poulton. Gene-environment interactions in schizophrenia: review of epidemiological findings and future directions. Schizophr Bull, 2008. [PubMed]
- Y Ayhan, R McFarland, MV Pletnikov. Animal models of gene-environment interaction in schizophrenia: a dimensional perspective. Prog Neurobiol, 2016. [PubMed]
- R Ottman. Gene-environment interaction: definitions and study designs. Prev Med, 1996. [PubMed]
- G Modinos, C Iyegbe, D Prata, M Rivera, MJ Kempton, LR Valmaggia, PC Sham, J van Os, P McGuire. Molecular genetic gene-environment studies using candidate genes in schizophrenia: a systematic review. Schizophr Res, 2013. [PubMed]
- D Moher, A Liberati, J Tetzlaff, DG Altman. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ, 2009. [PubMed]
- M de Castro-Catala, M van Nierop, N Barrantes-Vidal, P Cristobal-Narvaez, T Sheinbaum, TR Kwapil, E Pena, N Jacobs, C Derom, E Thiery, J van Os, R van Winkel, A Rosa. Childhood trauma, BDNF Val66Met and subclinical psychotic experiences. Attempt at replication in two independent samples. J Psychiatr Res, 2016. [PubMed]
- P Cristobal-Narvaez, T Sheinbaum, A Rosa, S Ballespi, M de Castro-Catala, E Pena, TR Kwapil, N Barrantes-Vidal. The interaction between childhood bullying and the FKBP5 gene on psychotic-like experiences and stress reactivity in real life. PLoS One, 2016. [PubMed]
- N Barrantes-Vidal, CA Chun, I Myin-Germeys, TR Kwapil. Psychometric schizotypy predicts psychotic-like, paranoid, and negative symptoms in daily life. J Abnorm Psychol, 2013. [PubMed]
- 24.Gattere G, Stojanovic-Perez A, Monseny R, Martorell L, Ortega L, Montalvo I, Sole M, Algora MJ et al (2016) Gene-environment interaction between the brain-derived neurotrophic factor Val66Met polymorphism, psychosocial stress and dietary intake in early psychosis. Early Interv Psychiatry. doi:10.1111/eip.12371
- L Mandelli, E Toscano, S Porcelli, C Fabbri, A Serretti. Age of onset in schizophrenia Spectrum disorders: complex interactions between genetic and environmental factors. Psychiatry Investig, 2016. [PubMed]
- DH Nieman, S Dragt, ED van Duin, N Denneman, JM Overbeek, L de Haan, J Rietdijk, HK Ising, RM Klaassen, T van Amelsvoort, L Wunderink, M van der Gaag, DH Linszen. COMT Val(158)Met genotype and cannabis use in people with an at risk mental state for psychosis: exploring gene x environment interactions. Schizophr Res, 2016. [PubMed]
- G Ursini, T Cavalleri, L Fazio, T Angrisano, L Iacovelli, A Porcelli, G Maddalena, G Punzi, M Mancini, B Gelao, R Romano, R Masellis, F Calabrese, A Rampino, P Taurisano, A Di Giorgio, S Keller, L Tarantini, L Sinibaldi, T Quarto, T Popolizio, G Caforio, G Blasi, MA Riva, A De Blasi, L Chiariotti, V Bollati, A Bertolino. BDNF rs6265 methylation and genotype interact on risk for schizophrenia. Epigenetics, 2016. [PubMed]
- TF McNeil, E Cantor-Graae, EF Torrey, K Sjostrom, A Bowler, E Taylor, R Rawlings, ES Higgins. Obstetric complications in histories of monozygotic twins discordant and concordant for schizophrenia. Acta Psychiatr Scand, 1994. [PubMed]
- M Colizzi, C Iyegbe, J Powell, G Ursini, A Porcelli, A Bonvino, P Taurisano, R Romano, R Masellis, G Blasi, C Morgan, K Aitchison, V Mondelli, S Luzi, A Kolliakou, A David, RM Murray, A Bertolino, M Di Forti. Interaction between functional genetic variation of DRD2 and cannabis use on risk of psychosis. Schizophr Bull, 2015. [PubMed]
- A Ermis, M Erkiran, S Dasdemir, AS Turkcan, ME Ceylan, ES Bireller, B Cakmakoglu. The relationship between catechol-O-methyltransferase gene Val158Met (COMT) polymorphism and premorbid cannabis use in Turkish male patients with schizophrenia. In Vivo, 2015. [PubMed]
- MJ Green, A Raudino, MJ Cairns, J Wu, PA Tooney, RJ Scott, VJ Carr. Do common genotypes of FK506 binding protein 5 (FKBP5) moderate the effects of childhood maltreatment on cognition in schizophrenia and healthy controls?. J Psychiatr Res, 2015. [PubMed]
- A Wegelius, M Pankakoski, L Tomppo, U Lehto, J Lonnqvist, J Suvisaari, T Paunio, W Hennah. An interaction between NDE1 and high birth weight increases schizophrenia susceptibility. Psychiatry Res, 2015. [PubMed]
- M Aas, UK Haukvik, S Djurovic, M Tesli, L Athanasiu, T Bjella, L Hansson, A Cattaneo, I Agartz, OA Andreassen, I Melle. Interplay between childhood trauma and BDNF val66met variants on blood BDNF mRNA levels and on hippocampus subfields volumes in schizophrenia spectrum and bipolar disorders. J Psychiatr Res, 2014. [PubMed]
- O Ajnakina, S Borges, M Di Forti, Y Patel, X Xu, P Green, SA Stilo, A Kolliakou, P Sood, TR Marques, AS David, D Prata, P Dazzan, J Powell, C Pariante, V Mondelli, C Morgan, RM Murray, HL Fisher, C Iyegbe. Role of environmental confounding in the association between FKBP5 and first-episode psychosis. Front Psych, 2014
- D Hernaus, R van Winkel, E Gronenschild, P Habets, G Kenis, M Marcelis, J van Os, I Myin-Germeys, D Collip. Brain-derived neurotrophic factor/FK506-binding protein 5 genotype by childhood trauma interactions do not impact on hippocampal volume and cognitive performance. PLoS One, 2014. [PubMed]
- S McCarthy-Jones, MJ Green, RJ Scott, PA Tooney, MJ Cairns, JQ Wu, C Oldmeadow, V Carr. Preliminary evidence of an interaction between the FOXP2 gene and childhood emotional abuse predicting likelihood of auditory verbal hallucinations in schizophrenia. J Psychiatr Res, 2014. [PubMed]
- E Pishva, M Drukker, W Viechtbauer, J Decoster, D Collip, R van Winkel, M Wichers, N Jacobs, E Thiery, C Derom, N Geschwind, D van den Hove, T Lataster, I Myin-Germeys, J van Os, BP Rutten, G Kenis. Epigenetic genes and emotional reactivity to daily life events: a multi-step gene-environment interaction study. PLoS One, 2014. [PubMed]
- D Collip, I Myin-Germeys, M Wichers, N Jacobs, C Derom, E Thiery, T Lataster, C Simons, P Delespaul, M Marcelis, J van Os, R van Winkel. FKBP5 as a possible moderator of the psychosis-inducing effects of childhood trauma. Br J Psychiatry, 2013. [PubMed]
- KR De Sousa, AK Tiwari, DE Giuffra, B Mackenzie, CC Zai, JL Kennedy. Age at onset of schizophrenia: cannabis, COMT gene, and their interactions. Schizophr Res, 2013. [PubMed]
- OE Onwuameze, KW Nam, EA Epping, TH Wassink, S Ziebell, NC Andreasen, BC Ho. MAPK14 and CNR1 gene variant interactions: effects on brain volume deficits in schizophrenia patients with marijuana misuse. Psychol Med, 2013. [PubMed]
- H Ramsay, I Kelleher, P Flannery, MC Clarke, F Lynch, M Harley, D Connor, C Fitzpatrick, DW Morris, M Cannon. Relationship between the COMT-Val158Met and BDNF-Val66Met polymorphisms, childhood trauma and psychotic experiences in an adolescent general population sample. PLoS One, 2013. [PubMed]
- CH Vinkers, WA Van Gastel, CD Schubart, KR Van Eijk, JJ Luykx, R Van Winkel, M Joels, RA Ophoff, MP Boks, R Bruggeman, W Cahn, L de Haan, RS Kahn, CJ Meijer, I Myin-Germeys, J van Os, D Wiersma. The effect of childhood maltreatment and cannabis use on adult psychotic symptoms is modified by the COMT Val(1)(5)(8)Met polymorphism. Schizophr Res, 2013. [PubMed]
- S Bhattacharyya, Z Atakan, R Martin-Santos, JA Crippa, J Kambeitz, D Prata, S Williams, M Brammer, DA Collier, PK McGuire. Preliminary report of biological basis of sensitivity to the effects of cannabis on psychosis: AKT1 and DAT1 genotype modulates the effects of delta-9-tetrahydrocannabinol on midbrain and striatal function. Mol Psychiatry, 2012. [PubMed]
- M Di Forti, C Iyegbe, H Sallis, A Kolliakou, MA Falcone, A Paparelli, M Sirianni, C La Cascia, SA Stilo, TR Marques, R Handley, V Mondelli, P Dazzan, C Pariante, AS David, C Morgan, J Powell, RM Murray. Confirmation that the AKT1 (rs2494732) genotype influences the risk of psychosis in cannabis users. Biol Psychiatry, 2012. [PubMed]
- JA Husted, R Ahmed, EW Chow, LM Brzustowicz, AS Bassett. Early environmental exposures influence schizophrenia expression even in the presence of strong genetic predisposition. Schizophr Res, 2012. [PubMed]
- O Peerbooms, BP Rutten, D Collip, M Lardinois, T Lataster, V Thewissen, SM Rad, M Drukker, G Kenis, J van Os, I Myin-Germeys, R van Winkel. Evidence that interactive effects of COMT and MTHFR moderate psychotic response to environmental stress. Acta Psychiatr Scand, 2012. [PubMed]
- S Alemany, B Arias, M Aguilera, H Villa, J Moya, MI Ibanez, H Vossen, C Gasto, G Ortet, L Fananas. Childhood abuse, the BDNF-Val66Met polymorphism and adult psychotic-like experiences. Br J Psychiatry, 2011. [PubMed]
- D Collip, R van Winkel, O Peerbooms, T Lataster, V Thewissen, M Lardinois, M Drukker, BP Rutten, J Van Os, I Myin-Germeys. COMT Val158Met-stress interaction in psychosis: role of background psychosis risk. CNS Neurosci Ther, 2011. [PubMed]
- J Costas, J Sanjuan, R Ramos-Rios, E Paz, S Agra, A Tolosa, M Paramo, J Brenlla, M Arrojo. Interaction between COMT haplotypes and cannabis in schizophrenia: a case-only study in two samples from Spain. Schizophr Res, 2011. [PubMed]
- J Decoster, J van Os, G Kenis, C Henquet, J Peuskens, M De Hert, R van Winkel. Age at onset of psychotic disorder: cannabis, BDNF Val66Met, and sex-specific models of gene-environment interaction. Am J Med Genet B Neuropsychiatr Genet, 2011. [PubMed]
- D Demontis, M Nyegaard, HN Buttenschon, A Hedemand, CB Pedersen, J Grove, TJ Flint, M Nordentoft, T Werge, DM Hougaard, KM Sorensen, RH Yolken, O Mors, AD Borglum, PB Mortensen. Association of GRIN1 and GRIN2A-D with schizophrenia and genetic interaction with maternal herpes simplex virus-2 infection affecting disease risk. Am J Med Genet B Neuropsychiatr Genet, 2011. [PubMed]
- G Estrada, M Fatjo-Vilas, MJ Munoz, G Pulido, MJ Minano, E Toledo, JM Illa, M Martin, ML Miralles, S Miret, S Campanera, C Bernabeu, ME Navarro, L Fananas. Cannabis use and age at onset of psychosis: further evidence of interaction with COMT Val158Met polymorphism. Acta Psychiatr Scand, 2011. [PubMed]
- BC Ho, TH Wassink, S Ziebell, NC Andreasen. Cannabinoid receptor 1 gene polymorphisms and marijuana misuse interactions on white matter and cognitive deficits in schizophrenia. Schizophr Res, 2011. [PubMed]
- Effects of season of birth and a common MTHFR gene variant on the risk of schizophrenia. Eur Neuropsychopharmacol J Eur College Neuropsychopharmacol, 2011
- R van Winkel. Family-based analysis of genetic variation underlying psychosis-inducing effects of cannabis: sibling analysis and proband follow-up. Arch Gen Psychiatry, 2011. [PubMed]
- UK Haukvik, P Saetre, T McNeil, PS Bjerkan, OA Andreassen, T Werge, EG Jonsson, I Agartz. An exploratory model for G x E interaction on hippocampal volume in schizophrenia; obstetric complications and hypoxia-related genes. Prog Neuro-Psychopharmacol Biol Psychiatry, 2010
- JM Pelayo-Teran, R Perez-Iglesias, I Mata, E Carrasco-Marin, JL Vazquez-Barquero, B Crespo-Facorro. Catechol-O-methyltransferase (COMT) Val158Met variations and cannabis use in first-episode non-affective psychosis: clinical-onset implications. Psychiatry Res, 2010. [PubMed]
- S Zammit, G Lewis, C Dalman, P Allebeck. Examining interactions between risk factors for psychosis. Br J Psychiatry, 2010. [PubMed]
- B Gutierrez, M Rivera, L Obel, K McKenney, R Martinez-Leal, E Molina, M Dolz, S Ochoa, J Usall, JM Haro, M Ruiz-Veguilla, F Torres, J Cervilla. Variability in the COMT gene and modification of the risk of schizophrenia conferred by cannabis consumption. Rev Psiquiatr Salud Ment, 2009. [PubMed]
- C Henquet, A Rosa, P Delespaul, S Papiol, L Fananas, J van Os, I Myin-Germeys. COMT ValMet moderation of cannabis-induced psychosis: a momentary assessment study of ‘switching on’ hallucinations in the flow of daily life. Acta Psychiatr Scand, 2009. [PubMed]
- JT Kantrowitz, KA Nolan, S Sen, AA Simen, HM Lachman, MB Bowers. Adolescent cannabis use, psychosis and catechol-O-methyltransferase genotype in African Americans and Caucasians. Psychiatry Q, 2009
- S Keri, I Kiss, I Seres, O Kelemen. A polymorphism of the neuregulin 1 gene (SNP8NRG243177/rs6994992) affects reactivity to expressed emotion in schizophrenia. Am J Med Genet B Neuropsychiatr Genet, 2009. [PubMed]
- CJ Simons, M Wichers, C Derom, E Thiery, I Myin-Germeys, L Krabbendam, J van Os. Subtle gene-environment interactions driving paranoia in daily life. Genes Brain Behav, 2009. [PubMed]
- KK Nicodemus, S Marenco, AJ Batten, R Vakkalanka, MF Egan, RE Straub, DR Weinberger. Serious obstetric complications interact with hypoxia-regulated/vascular-expression genes to influence schizophrenia risk. Mol Psychiatry, 2008. [PubMed]
- R van Winkel, C Henquet, A Rosa, S Papiol, L Fananas, M De Hert, J Peuskens, J van Os, I Myin-Germeys. Evidence that the COMT(Val158Met) polymorphism moderates sensitivity to stress in psychosis: an experience-sampling study. Am J Med Genet B Neuropsychiatr Genet, 2008. [PubMed]
- BH Shirts, JJ Kim, S Reich, FB Dickerson, RH Yolken, B Devlin, VL Nimgaonkar. Polymorphisms in MICB are associated with human herpes virus seropositivity and schizophrenia risk. Schizophr Res, 2007. [PubMed]
- NC Stefanis, C Henquet, D Avramopoulos, N Smyrnis, I Evdokimidis, I Myin-Germeys, CN Stefanis, J Van Os. COMT Val158Met moderation of stress-induced psychosis. Psychol Med, 2007. [PubMed]
- S Zammit, G Spurlock, H Williams, N Norton, N Williams, MC O’Donovan, MJ Owen. Genotype effects of CHRNA7, CNR1 and COMT in schizophrenia: interactions with tobacco and cannabis use. Br J Psychiatry, 2007. [PubMed]
- C Henquet, A Rosa, L Krabbendam, S Papiol, L Fananas, M Drukker, JG Ramaekers, J van Os. An experimental study of catechol-o-methyltransferase Val158Met moderation of delta-9-tetrahydrocannabinol-induced effects on psychosis and cognition. Neuropsychopharmacology, 2006. [PubMed]
- A Caspi, TE Moffitt, M Cannon, J McClay, R Murray, H Harrington, A Taylor, L Arseneault, B Williams, A Braithwaite, R Poulton, IW Craig. Moderation of the effect of adolescent-onset cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: longitudinal evidence of a gene X environment interaction. Biol Psychiatry, 2005. [PubMed]
- J Chotai, A Serretti, E Lattuada, C Lorenzi, R Lilli. Gene-environment interaction in psychiatric disorders as indicated by season of birth variations in tryptophan hydroxylase (TPH), serotonin transporter (5-HTTLPR) and dopamine receptor (DRD4) gene polymorphisms. Psychiatry Res, 2003. [PubMed]
- M Tochigi, J Ohashi, T Umekage, K Kohda, H Hibino, T Otowa, T Marui, K Masui, Y Sugahara, R Kanamori, T Juji, N Kato, K Tokunaga, T Sasaki. Human leukocyte antigen-A specificities and its relation with season of birth in Japanese patients with schizophrenia. Neurosci Lett, 2002. [PubMed]
- K Narita, T Sasaki, R Akaho, Y Okazaki, I Kusumi, T Kato, O Hashimoto, R Fukuda, T Koyama, K Matsuo, Y Okabe, S Nanko, H Hohjoh, K Tokunaga. Human leukocyte antigen and season of birth in Japanese patients with schizophrenia. Am J Psychiatry, 2000. [PubMed]
- AR Yung, HP Yuen, PD McGorry, LJ Phillips, D Kelly, M Dell’Olio, SM Francey, EM Cosgrave, E Killackey, C Stanford, K Godfrey, J Buckby. Mapping the onset of psychosis: the comprehensive assessment of at-risk mental states. Aust N Z J Psychiatry, 2005. [PubMed]
- M Konings, M Bak, M Hanssen, J van Os, L Krabbendam. Validity and reliability of the CAPE: a self-report instrument for the measurement of psychotic experiences in the general population. Acta Psychiatr Scand, 2006. [PubMed]
- S Rosenman, B Rodgers. Childhood adversity in an Australian population. Soc Psychiatry Psychiatr Epidemiol, 2004. [PubMed]
- NC Andreasen, M Flaum, S Arndt. The Comprehensive Assessment of Symptoms and History (CASH). An instrument for assessing diagnosis and psychopathology. Arch Gen Psychiatry, 1992. [PubMed]
- A Bifulco, O Bernazzani, PM Moran, C Jacobs. The childhood experience of care and abuse questionnaire (CECA.Q): validation in a community series. Br J Clin Psychol, 2005. [PubMed]
- M Di Forti, C Morgan, P Dazzan, C Pariante, V Mondelli, TR Marques, R Handley, S Luzi, M Russo, A Paparelli, A Butt, SA Stilo, B Wiffen, J Powell, RM Murray. High-potency cannabis and the risk of psychosis. Br J Psychiatry, 2009. [PubMed]
- G Andrews, L Peters. The psychometric properties of the Composite International Diagnostic Interview. Soc Psychiatry Psychiatr Epidemiol, 1998. [PubMed]
- AS Loonstra, AR Tarlow, AH Sellers. COWAT metanorms across age, education, and gender. Appl Neuropsychol, 2001. [PubMed]
- DP Bernstein, L Fink, L Handelsman, J Foote, M Lovejoy, K Wenzel, E Sapareto, J Ruggiero. Initial reliability and validity of a new retrospective measure of child abuse and neglect. Am J Psychiatry, 1994. [PubMed]
- DJ Castle, A Jablensky, JJ McGrath, V Carr, V Morgan, A Waterreus, G Valuri, H Stain, P McGuffin, A Farmer. The diagnostic interview for psychoses (DIP): Development, reliability and applications. Psychol Med, 2006. [PubMed]
- 84.Robins LN, Cottler L, Bucholtz K, Compton W (1995) Diagnostic Interview Schedule for DSM-IV. St. Louis, MO: Department of Psychiatry, Washington University School of Medicine
- S Moreno, S Rodriguez, MC Fernandez, J Tamez, A Cepeda-Benito. Clinical validation of the trait and state versions of the Food Craving Questionnaire. Assessment, 2008. [PubMed]
- CL Craig, AL Marshall, M Sjostrom, AE Bauman, ML Booth, BE Ainsworth, M Pratt, U Ekelund, A Yngve, JF Sallis, P Oja. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc, 2003. [PubMed]
- J Kaufman, B Birmaher, D Brent, U Rao, N Ryan. The Schedule for Affective Disorders and Schizophrenia for School Aged Children: present and lifetime version, 1996
- DV Sheehan, Y Lecrubier, KH Sheehan, P Amorim, J Janavs, E Weiller, T Hergueta, R Baker, GC Dunbar. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry, 1998. [PubMed]
- SR Kay, A Fiszbein, LA Opler. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull, 1987. [PubMed]
- P McGuffin, A Farmer, I Harvey. A polydiagnostic application of operational criteria in studies of psychotic illness. Development and reliability of the OPCRIT system. Arch Gen Psychiatry, 1991. [PubMed]
- IS Rosenfarb, MJ Goldstein, J Mintz, KH Nuechterlein. Expressed emotion and subclinical psychopathology observable within the transactions between schizophrenic patients and their family members. J Abnorm Psychol, 1995. [PubMed]
- IS Rosenfarb, KH Nuechterlein, MJ Goldstein, KL Subotnik. Neurocognitive vulnerability, interpersonal criticism, and the emergence of unusual thinking by schizophrenic patients during family transactions. Arch Gen Psychiatry, 2000. [PubMed]
- S Cohen, T Kamarck, R Mermelstein. A global measure of perceived stress. J Health Soc Behav, 1983. [PubMed]
- C Randolph, MC Tierney, E Mohr, TN Chase. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): preliminary clinical validity. J Clin Exp Neuropsychol, 1998. [PubMed]
- MB First, RL Spitzer, M Gibbon, JBW Williams. Structured Clinical Interview for DSM-IV Axis I Disorders, Clinician Version (SCID-CV), 1996
- KS Kendler, JA Lieberman, D Walsh. The Structured Interview for Schizotypy (SIS): a preliminary report. Schizophr Bull, 1989. [PubMed]
- A Raine. The SPQ: a scale for the assessment of schizotypal personality based on DSM-III-R criteria. Schizophr Bull, 1991. [PubMed]
- TH Holmes, RH Rahe. The Social Readjustment Rating Scale. J Psychosom Res, 1967. [PubMed]
- D Wechsler. Weschsler Adult Intelligence Scale-III, 1997
- FB Dickerson, JJ Boronow, C Stallings, AE Origoni, S Cole, F Leister, B Krivogorsky, RH Yolken. The catechol O-methyltransferase Val158Met polymorphism and herpes simplex virus type 1 infection are risk factors for cognitive impairment in bipolar disorder: additive gene-environmental effects in a complex human psychiatric disorder. Bipolar Disord, 2006. [PubMed]
- M De Pradier, P Gorwood, B Beaufils, J Ades, C Dubertret. Influence of the serotonin transporter gene polymorphism, cannabis and childhood sexual abuse on phenotype of bipolar disorder: a preliminary study. Eur Psychiatry, 2010. [PubMed]
- GM Hosang, R Uher, R Keers, S Cohen-Woods, I Craig, A Korszun, J Perry, F Tozzi, P Muglia, P McGuffin, AE Farmer. Stressful life events and the brain-derived neurotrophic factor gene in bipolar disorder. J Affect Disord, 2010. [PubMed]
- J Savitz, L van der Merwe, TK Newman, DJ Stein, R Ramesar. Catechol-o-methyltransferase genotype and childhood trauma may interact to impact schizotypal personality traits. Behav Genet, 2010. [PubMed]
- M Debnath, M Busson, S Jamain, B Etain, N Hamdani, J Oliveira, W Boukouaci, K Amokrane, H Moins-Teisserenc, M Lajnef, D Bengoufa, A Malafosse, F Bellivier, C Henry, JP Kahn, R Krishnamoorthy, D Charron, M Leboyer, R Tamouza. The HLA-G low expressor genotype is associated with protection against bipolar disorder. Hum Immunol, 2013. [PubMed]
- S Miller, J Hallmayer, PW Wang, SJ Hill, SL Johnson, TA Ketter. Brain-derived neurotrophic factor val66met genotype and early life stress effects upon bipolar course. J Psychiatr Res, 2013. [PubMed]
- CC Bortolasci, HO Vargas, A Souza-Nogueira, DS Barbosa, EG Moreira, SO Nunes, M Berk, S Dodd, M Maes. Lowered plasma paraoxonase (PON)1 activity is a trait marker of major depression and PON1 Q192R gene polymorphism-smoking interactions differentially predict the odds of major depression and bipolar disorder. J Affect Disord, 2014. [PubMed]
- ME Breen, F Seifuddin, PP Zandi, JB Potash, VL Willour. Investigating the role of early childhood abuse and HPA axis genes in suicide attempters with bipolar disorder. Psychiatr Genet, 2015. [PubMed]
- J Oliveira, B Etain, M Lajnef, N Hamdani, M Bennabi, D Bengoufa, A Sundaresh, AB Chaabane, F Bellivier, C Henry, JP Kahn, D Charron, R Krishnamoorthy, M Leboyer, R Tamouza. Combined effect of TLR2 gene polymorphism and early life stress on the age at onset of bipolar disorders. PLoS One, 2015. [PubMed]
- CP Zeni, B Mwangi, B Cao, KM Hasan, C Walss-Bass, G Zunta-Soares, JC Soares. Interaction between BDNF rs6265 Met allele and low family cohesion is associated with smaller left hippocampal volume in pediatric bipolar disorder. J Affect Disord, 2016. [PubMed]
- J Oliveira, R Kazma, E Le Floch, M Bennabi, N Hamdani, D Bengoufa, M Dahoun, C Manier, F Bellivier, R Krishnamoorthy, JF Deleuze, R Yolken, M Leboyer, R Tamouza. Toxoplasma gondii exposure may modulate the influence of TLR2 genetic variation on bipolar disorder: a gene-environment interaction study. Int J Bipolar Disord, 2016. [PubMed]
- AH Buss, A Durkee. An inventory for assessing different kinds of hostility. J Consult Psychol, 1957. [PubMed]
- GL Brown, FK Goodwin, JC Ballenger, PF Goyer, LF Major. Aggression in humans correlates with cerebrospinal fluid amine metabolites. Psychiatry Res, 1979. [PubMed]
- ES Barratt. Factor analysis of some psychometric measures of impulsiveness and anxiety. Psychol Rep, 1965. [PubMed]
- MK Spearing, RM Post, GS Leverich, D Brandt, W Nolen. Modification of the Clinical Global Impressions (CGI) Scale for use in bipolar illness (BP): the CGI-BP. Psychiatry Res, 1997. [PubMed]
- JI Nurnberger, MC Blehar, CA Kaufmann, C York-Cooler, SG Simpson, J Harkavy-Friedman, JB Severe, D Malaspina, T Reich. Diagnostic interview for genetic studies. Rationale, unique features, and training. NIMH Genet Initiat Arch Gen Psychiat, 1994
- CP Boyd, E Gullone, GL Needleman, T Burt. The Family Environment Scale: reliability and normative data for an adolescent sample. Fam Process, 1997. [PubMed]
- M Hamilton. A rating scale for depression. J Neurol Neurosurg Psychiatry, 1960. [PubMed]
- T Brugha, P Bebbington, C Tennant, J Hurry. The list of threatening experiences: a subset of 12 life event categories with considerable long-term contextual threat. Psychol Med, 1985. [PubMed]
- G Parker. Parental characteristics in relation to depressive disorders. Br J Psychiatry, 1979. [PubMed]
- JK Wing, T Babor, T Brugha, J Burke, JE Cooper, R Giel, A Jablenski, D Regier, N Sartorius. SCAN. Schedules for Clinical Assessment in Neuropsychiatry. Arch Gen Psychiatry, 1990. [PubMed]
- G Claridge, P Broks. Schizotypy and hemisphere function—I. Personal Individ Differ, 1984
- LM Hooper, P Stockton, JL Krupnick, BL Green. Development, Use, and Psychometric Properties of the Trauma History Questionnaire. J Loss Trauma, 2011
- 123.Alemany S, Moya J, Ibanez MI, Villa H, Mezquita L, Ortet G, Gasto C, Fananas L et al (2015) Research letter: childhood trauma and the rs1360780 SNP of FKBP5 gene in psychosis: a replication in two general population samples. Psychol Med:1–3. doi:10.1017/S0033291715001695
- NC Stefanis, M Dragovic, BD Power, A Jablensky, D Castle, VA Morgan. Age at initiation of cannabis use predicts age at onset of psychosis: the 7- to 8-year trend. Schizophr Bull, 2013. [PubMed]
- 125.Miller BJ, Culpepper N, Rapaport MH (2013) C-reactive protein levels in schizophrenia. Clin Schizophr Relat Psychoses:1–22. doi:10.3371/CSRP.MICU.020813
- EL Kinnally, YY Huang, R Haverly, AK Burke, H Galfalvy, DP Brent, MA Oquendo, JJ Mann. Parental care moderates the influence of MAOA-uVNTR genotype and childhood stressors on trait impulsivity and aggression in adult women. Psychiatr Genet, 2009. [PubMed]
- J van Os, M Marcelis. The ecogenetics of schizophrenia: a review. Schizophr Res, 1998. [PubMed]
- J Chen, BK Lipska, N Halim, QD Ma, M Matsumoto, S Melhem, BS Kolachana, TM Hyde, MM Herman, J Apud, MF Egan, JE Kleinman, DR Weinberger. Functional analysis of genetic variation in catechol-O-methyltransferase (COMT): effects on mRNA, protein, and enzyme activity in postmortem human brain. Am J Hum Genet, 2004. [PubMed]
- OD Howes, AJ Montgomery, MC Asselin, RM Murray, I Valli, P Tabraham, E Bramon-Bosch, L Valmaggia, L Johns, M Broome, PK McGuire, PM Grasby. Elevated striatal dopamine function linked to prodromal signs of schizophrenia. Arch Gen Psychiatry, 2009. [PubMed]
- P Casadio, C Fernandes, RM Murray, M Di Forti. Cannabis use in young people: the risk for schizophrenia. Neurosci Biobehav Rev, 2011. [PubMed]
- T Okimura, S Tanaka, T Maeda, M Kato, M Mimura. Simulation of the capacity and precision of working memory in the hypodopaminergic state: relevance to schizophrenia. Neuroscience, 2015. [PubMed]
- D Thomas. Gene–environment-wide association studies: emerging approaches. Nat Rev Genet, 2010. [PubMed]
- SB Manuck, JM McCaffery. Gene-environment interaction. Ann Rev Psychol, 2014. [PubMed]
- CY Liu, A Maity, X Lin, RO Wright, DC Christiani. Design and analysis issues in gene and environment studies. Environ Health, 2012. [PubMed]
- R Uher. Gene–environment interaction: overcoming methodological challenges. Genetic effects on environmental vulnerability to disease, 2008
- I Colman, M Kingsbury, Y Garad, Y Zeng, K Naicker, S Patten, PB Jones, TC Wild, AH Thompson. Consistency in adult reporting of adverse childhood experiences. Psychol Med, 2016. [PubMed]
- CM McKinney, TR Harris, R Caetano. Reliability of self-reported childhood physical abuse by adults and factors predictive of inconsistent reporting. Violence Vict, 2009. [PubMed]
- HL Fisher, TK Craig, P Fearon, K Morgan, P Dazzan, J Lappin, G Hutchinson, GA Doody, PB Jones, P McGuffin, RM Murray, J Leff, C Morgan. Reliability and comparability of psychosis patients’ retrospective reports of childhood abuse. Schizophr Bull, 2011. [PubMed]
- D Wolke, ST Lereya, HL Fisher, G Lewis, S Zammit. Bullying in elementary school and psychotic experiences at 18 years: a longitudinal, population-based cohort study. Psychol Med, 2014. [PubMed]
- MC Cutajar, PE Mullen, JR Ogloff, SD Thomas, DL Wells, J Spataro. Schizophrenia and other psychotic disorders in a cohort of sexually abused children. Arch Gen Psychiatry, 2010. [PubMed]
- L Arseneault, M Cannon, HL Fisher, G Polanczyk, TE Moffitt, A Caspi. Childhood trauma and children’s emerging psychotic symptoms: a genetically sensitive longitudinal cohort study. Am J Psychiatry, 2011. [PubMed]
- J Hardt, M Rutter. Validity of adult retrospective reports of adverse childhood experiences: review of the evidence. J Child Psychol Psychiatry, 2004. [PubMed]
- JP Selten, E van der Ven, BP Rutten, E Cantor-Graae. The social defeat hypothesis of schizophrenia: an update. Schizophr Bull, 2013. [PubMed]
- JE Houston, J Murphy, G Adamson, M Stringer, M Shevlin. Childhood sexual abuse, early cannabis use, and psychosis: testing an interaction model based on the National Comorbidity Survey. Schizophr Bull, 2008. [PubMed]
- J Murphy, JE Houston, M Shevlin, G Adamson. Childhood sexual trauma, cannabis use and psychosis: statistically controlling for pre-trauma psychosis and psychopathology. Soc Psychiatry Psychiatr Epidemiol, 2013. [PubMed]
- 146.Padmanabhan JL, Shah JL, Neeraj T, Keshavan MS (2017) The “polyenviromic risk score”: aggregating environmental risk factors predicts conversion to psychosis in familial high-risk subjects. Schizophr Res 181:17–22. doi:10.1016/j.schres.2016.10.014
- C Iyegbe, D Campbell, A Butler, O Ajnakina, P Sham. The emerging molecular architecture of schizophrenia, polygenic risk scores and the clinical implications for GxE research. Soc Psychiatry Psychiatr Epidemiol, 2014. [PubMed]
- B Misiak, D Frydecka, M Zawadzki, M Krefft, A Kiejna. Refining and integrating schizophrenia pathophysiology—relevance of the allostatic load concept. Neurosci Biobehav Rev, 2014
- O Marin. Developmental timing and critical windows for the treatment of psychiatric disorders. Nat Med, 2016. [PubMed]
- J Davis, S Moylan, BH Harvey, M Maes, M Berk. Neuroprogression in schizophrenia: pathways underpinning clinical staging and therapeutic corollaries. Aust N Z J Psychiatr, 2014
- AS Brown, J Hooton, CA Schaefer, H Zhang, E Petkova, V Babulas, M Perrin, JM Gorman, ES Susser. Elevated maternal interleukin-8 levels and risk of schizophrenia in adult offspring. Am J Psychiatry, 2004. [PubMed]
- SL Buka, MT Tsuang, EF Torrey, MA Klebanoff, RL Wagner, RH Yolken. Maternal cytokine levels during pregnancy and adult psychosis. Brain Behav Immun, 2001. [PubMed]
- G Scola, A Duong. Prenatal maternal immune activation and brain development with relevance to psychiatric disorders. Neuroscience, 2017. [PubMed]
- R Schmidt-Kastner, J van Os, G Esquivel, HW Steinbusch, BP Rutten. An environmental analysis of genes associated with schizophrenia: hypoxia and vascular factors as interacting elements in the neurodevelopmental model. Mol Psychiatry, 2012. [PubMed]
- TD Cannon, TG van Erp, IM Rosso, M Huttunen, J Lonnqvist, T Pirkola, O Salonen, L Valanne, VP Poutanen, CG Standertskjold-Nordenstam. Fetal hypoxia and structural brain abnormalities in schizophrenic patients, their siblings, and controls. Arch Gen Psychiatry, 2002. [PubMed]
- UK Haukvik, T McNeil, EH Lange, I Melle, AM Dale, OA Andreassen, I Agartz. Pre- and perinatal hypoxia associated with hippocampus/amygdala volume in bipolar disorder. Psychol Med, 2014. [PubMed]
- P Moran, J Stokes, J Marr, G Bock, L Desbonnet, J Waddington, C O’Tuathaigh. Gene x environment interactions in schizophrenia: evidence from genetic mouse models. Neural Plast, 2016. [PubMed]
- MD De Bellis, A Zisk. The biological effects of childhood trauma. Child Adolesc Psychiatr Clin N Am, 2014. [PubMed]
- B Misiak, M Krefft, T Bielawski, AA Moustafa, MM Sasiadek, D Frydecka. Toward a unified theory of childhood trauma and psychosis: a comprehensive review of epidemiological, clinical, neuropsychological and biological findings. Neurosci Biobehav Rev, 2017. [PubMed]
- 160.Moustafa AA, Salama M, Peak R, Tindle R, Salem A, Keri S, Misiak B, Frydecka D et al (2017) Interactions between cannabis and schizophrenia in humans and rodents. Rev Neurosci. doi:10.1515/revneuro-2016-0083
- J Davis, H Eyre, FN Jacka, S Dodd, O Dean, S McEwen, M Debnath, J McGrath, M Maes, P Amminger, PD McGorry, C Pantelis, M Berk. A review of vulnerability and risks for schizophrenia: beyond the two hit hypothesis. Neurosci Biobehav Rev, 2016. [PubMed]
- II Gottesman, TD Gould. The endophenotype concept in psychiatry: etymology and strategic intentions. Am J Psychiatry, 2003. [PubMed]
- CL Arfken, S Carney, NN Boutros. Translating biological parameters into clinically useful diagnostic tests. Curr Psychiatry Rep, 2009. [PubMed]
- F Thibaut, NN Boutros, M Jarema, B Oranje, A Hasan, ZJ Daskalakis, A Wichniak, A Schmitt, P Riederer, P Falkai. Consensus paper of the WFSBP Task Force on Biological Markers: Criteria for biomarkers and endophenotypes of schizophrenia part I: neurophysiology. World J Biol Psychiatry, 2015. [PubMed]
- OD Howes, RM Murray. Schizophrenia: an integrated sociodevelopmental-cognitive model. Lancet, 2014. [PubMed]
- E Barkus, P Morrison, M Di Forti, RM Murray. Are deficits in cognition associated with psychotic-like experiences after cannabis?. Hum Psychopharmacol, 2016. [PubMed]
- 167.Gawęda Ł, Prochwicz K, Adamczyk P, Frydecka D, Misiak B, Kotowicz K, Szczepanowski R, Florkowski M, Nelson B (2017) The role of self-disturbances and cognitive biases in the relationship between traumatic life events and psychosis proneness in a non-clinical sample. Schizophr Res. doi:10.1016/j.schres.2017.07.023
- M Bosia, A Pigoni, A Pirovano, C Lorenzi, M Spangaro, M Buonocore, M Bechi, F Cocchi, C Guglielmino, P Bramanti, E Smeraldi, R Cavallaro. COMT and STH polymorphisms interaction on cognition in schizophrenia. Neurol Sci, 2015. [PubMed]
- P Lopez-Garcia, L Young Espinoza, P Molero Santos, J Marin, F Ortuno Sanchez-Pedreno. Impact of COMT genotype on cognition in schizophrenia spectrum patients and their relatives. Psychiatry Res, 2013. [PubMed]
- MJ Green, TY Chia, MJ Cairns, J Wu, PA Tooney, RJ Scott, VJ Carr. Catechol-O-methyltransferase (COMT) genotype moderates the effects of childhood trauma on cognition and symptoms in schizophrenia. J Psychiatr Res, 2014. [PubMed]
- R Larson, M Csikszentmihalyi. The experience sampling method. In: Csikszentmihalyi M (ed) Flow and the foundations of positive psychology, 2014
- U Reininghaus, MJ Kempton, L Valmaggia, TK Craig, P Garety, A Onyejiaka, C Gayer-Anderson, SH So, K Hubbard, S Beards, P Dazzan, C Pariante, V Mondelli, HL Fisher, JG Mills, W Viechtbauer, P McGuire, J van Os, RM Murray, T Wykes, I Myin-Germeys, C Morgan. Stress sensitivity, aberrant salience, and threat anticipation in early psychosis: an experience sampling study. Schizophr Bull, 2016. [PubMed]
- U Reininghaus, C Gayer-Anderson, L Valmaggia, MJ Kempton, M Calem, A Onyejiaka, K Hubbard, P Dazzan, S Beards, HL Fisher, JG Mills, P McGuire, TK Craig, P Garety, J van Os, RM Murray, T Wykes, I Myin-Germeys, C Morgan. Psychological processes underlying the association between childhood trauma and psychosis in daily life: an experience sampling study. Psychol Med, 2016. [PubMed]
- J van Os, BP Rutten, I Myin-Germeys, P Delespaul, W Viechtbauer, C van Zelst, R Bruggeman, U Reininghaus, C Morgan, RM Murray, M Di Forti, P McGuire, LR Valmaggia, MJ Kempton, C Gayer-Anderson, K Hubbard, S Beards, SA Stilo, A Onyejiaka, F Bourque, G Modinos, S Tognin, M Calem, MC O’Donovan, MJ Owen, P Holmans, N Williams, N Craddock, A Richards, I Humphreys, A Meyer-Lindenberg, FM Leweke, H Tost, C Akdeniz, C Rohleder, JM Bumb, E Schwarz, K Alptekin, A Ucok, MC Saka, EC Atbasoglu, S Guloksuz, G Gumus-Akay, B Cihan, H Karadag, H Soygur, ES Cankurtaran, S Ulusoy, B Akdede, T Binbay, A Ayer, H Noyan, G Karadayi, E Akturan, H Ulas, C Arango, M Parellada, M Bernardo, J Sanjuan, J Bobes, M Arrojo, JL Santos, P Cuadrado, JJ Rodriguez Solano, A Carracedo, E Garcia Bernardo, L Roldan, G Lopez, B Cabrera, S Cruz, EM Diaz Mesa, M Pouso, E Jimenez, T Sanchez, M Rapado, E Gonzalez, C Martinez, E Sanchez, MS Olmeda, L de Haan, E Velthorst, M van der Gaag, JP Selten, D van Dam, E van der Ven, F van der Meer, E Messchaert, T Kraan, N Burger, M Leboyer, A Szoke, F Schurhoff, PM Llorca, S Jamain, A Tortelli, F Frijda, J Vilain, AM Galliot, G Baudin, A Ferchiou, JR Richard, E Bulzacka, T Charpeaud, AM Tronche, M De Hert, R van Winkel, J Decoster, C Derom, E Thiery, NC Stefanis, G Sachs, H Aschauer, I Lasser, B Winklbaur, M Schlogelhofer, A Riecher-Rossler, S Borgwardt, A Walter, F Harrisberger, R Smieskova, C Rapp, S Ittig, F Soguel-dit-Piquard, E Studerus, J Klosterkotter, S Ruhrmann, J Paruch, D Julkowski, D Hilboll, PC Sham, SS Cherny, EY Chen, DD Campbell, M Li, CM Romeo-Casabona, A Emaldi Cirion, A Urruela Mora, P Jones, J Kirkbride, M Cannon, D Rujescu, I Tarricone, D Berardi, E Bonora, M Seri, T Marcacci, L Chiri, F Chierzi, V Storbini, M Braca, MG Minenna, I Donegani, A Fioritti, D La Barbera, CE La Cascia, A Mule, L Sideli, R Sartorio, L Ferraro, G Tripoli, F Seminerio, AM Marinaro, P McGorry, B Nelson, GP Amminger, C Pantelis, PR Menezes, CM Del-Ben, SH Gallo Tenan, R Shuhama, M Ruggeri, S Tosato, A Lasalvia, C Bonetto, E Ira, M Nordentoft, MO Krebs, N Barrantes-Vidal, P Cristobal, TR Kwapil, E Brietzke, RA Bressan, A Gadelha, NP Maric, S Andric, M Mihaljevic, T Mirjanic. Identifying gene-environment interactions in schizophrenia: contemporary challenges for integrated, large-scale investigations. Schizophr Bull, 2014. [PubMed]
