Trypanocidal Effects of Polyprenylated Benzophenone Enriched Brazilian Red Propolis Extract and Its Isolated Constituent
Abstract
Chagas disease, a neglected tropical disease, affects millions of individuals in developing countries. Due to the limitations of current therapeutic options, the search for new bioactive compounds is crucial. Brazilian red propolis, a resinous bee product from northeastern Brazil, is recognized for its diverse chemical constituents and biological, including antiparasitic, activities. This study evaluated the in vitro activity of a polyprenylated benzophenone‐enriched Brazilian red propolis extract (SEBz) and its constituents, the mixture of isomers guttiferone E and xanthochymol (GUT/XAN), and oblongifolin B (OBL), against Trypanosoma cruzi (Y strain) trypomastigote and amastigote forms. Cytotoxicity and hemolysis assays assess safety. SEBz exhibited antiparasitic activity, with 50% effective concentration (EC50) values of 17.97 µg mL−1 (trypomastigotes) and 6.83 µg mL−1 (amastigotes) after 48 h, while demonstrating moderate cytotoxicity on the C2C12 myoblast cell line (50% cytotoxicity concentration [CC50] 42.80 µg mL−1 at 48 h). Both GUT/XAN and OBL showed trypanocidal effects (EC50 ≤ 5 µM against trypomastigotes; GUT/XAN EC50 7.91 µM on amastigotes), with moderate cytotoxicity (CC50 < 20 µM). Neither the extract nor the compounds induced significant hemolysis. Ultrastructural analysis of treated parasites revealed nuclear deformation and vacuolation. These findings support the potential of Brazilian red propolis benzophenones as promising leads for Chagas disease drug development.
Article type: Research Article
Keywords: Brazilian red propolis, guttiferone E/xanthochymol, oblongifolin B
Affiliations: Center For Research in Sciences and Technology University of Franca. Avenida Dr. Armando Salles of Oliveira Franca Brazil; School of Pharmaceutical Sciences of Ribeirão Preto University of São Paulo. Av. Do Café Ribeirão Preto Brazil
License: © 2026 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202502682 | PubMed: 41548073 | PMC: PMC12812226
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.3 MB)
Introduction
Trypanosoma cruzi, a hemoflagellate protozoan, is known for being the etiologic agent of Chagas disease (American trypanosomiasis), a neglected tropical disease throughout Latin America, with over 7 million infected individuals globally and around 30 000 new cases annually [ref. 1, ref. 2]. The chronic phase manifests with severe cardiac, digestive, and neurological complications, often resulting in fatal outcomes [ref. 1].
Current treatment strategies for Chagas disease rely primarily on two nitroheterocyclic drugs: benznidazole (BNZ) and nifurtimox (NFX). These agents are most effective during the acute phase of the disease, when parasite replication is high, yet their efficacy decreases in the chronic phase. Furthermore, both drugs are associated with significant adverse effects, leading to treatment discontinuation in a notable proportion of patients [ref. 3]. From this perspective, researchers are intensifying their efforts to develop different approaches to treat American trypanosomiasis, highlighting the studies with natural products, since they are substantial sources of new biologically active compounds [ref. 4, ref. 5, ref. 6, ref. 7].
Propolis, a resinous bee product, has been used in alternative medicine worldwide since ancient times as a natural antibiotic. It has recently become the focus of scientific research due to its broad range of properties and therapeutic actions, including antimicrobial, anticancer, anti‐inflammatory, antiviral, antioxidant, anesthetic, healing, hepatoprotective, and hematoprotective effects [ref. 8, ref. 9, ref. 10]. Honey bees (Apis mellifera) make propolis from a mixture of plant exudates, young botanical tissues and buds, pollen, wax, with their own saliva and enzymes; features that confer an intrinsic relation between botanical sources and chemical composition of propolis [ref. 11]. In honeycombs, propolis displays both structural and protective properties, being used to repair wall cracks, maintain appropriate humidity and heat levels, and also contributing to keeping a pathogen‐free environment, as well as keeping invaders away [ref. 10]. The wide biodiversity in Brazil is now responsible for 13 different types of propolis, differing in color and in their physicochemical properties, which are closely influenced by the geographic origin and seasonal variations of the plant sources involved in their production [ref. 12, ref. 13].
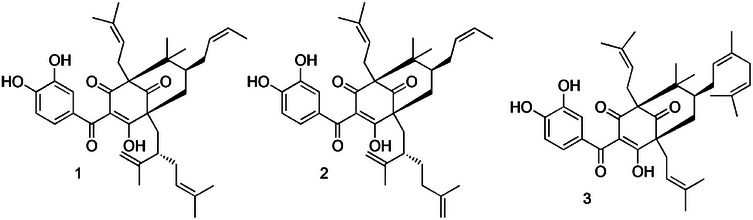
Red propolis, found on the northeastern side of Brazil, has as its botanical source two distinct trees, Dalbergia ecastaphyllum (L.) Taub. and Symphonia globulifera L.f. (Clusiaceae) [ref. 14]. While D. ecastophyllum is mainly responsible for the phenolic compounds, such as liquiritigenin, isoliquiritigenin, formononetin, vestitol, neovestitol, medicarpin, and 7‐O‐neovestitol; S. globulifera appears as the source of guttiferone E (GUT), xanthochymol (XAN), and oblongifolin B (OBL), both polyprenylated benzophenones [ref. 14, ref. 15]. Although red propolis extract demonstrates promising biological activities, including anti‐inflammatory [ref. 16], antimicrobial [ref. 17], and antiproliferative [ref. 18] properties, some studies carried out with Brazilian red propolis extracts showed antiprotozoal activity against Leishmania sp. [ref. 19, ref. 20, ref. 21, ref. 22], Trypanosoma sp. [ref. 19, ref. 23, ref. 24, ref. 25, ref. 26, ref. 27], and Trichomonas vaginalis [ref. 28]. Therefore, this work was focused on investigating the in vitro biological effects of a polyprenylated benzophenone‐enriched Brazilian red propolis extract (SEBz), as well as its compounds, the mixture of double bond isomers GUT/XAN and OBL (Figure 1) against trypomastigotes and amastigotes, T. cruzi (Y strain), assessing the ultrastructural damages induced in parasites, besides evaluating the cytotoxic and hemolytic properties.
Results and Discussion
The standardization of enriched extracts is a crucial step in natural product research, particularly to ensure the reproducibility of biological results, safety, and pharmacological efficacy. The chemical composition of extracts, such as propolis, can vary significantly depending on botanical and geographical origin, seasonality, extraction methods, and storage conditions [ref. 21, ref. 29, ref. 30]. Without standardization, results from different studies become difficult to compare, hindering progress in the development of phytopharmaceuticals or clinical formulations. Standardization allows for the identification and quantification of bioactive chemical markers, contributing to quality control and accurate therapeutic dosing [ref. 30]. In this study, experiments were conducted with SEBz, a polyprenylated benzophenone‐enriched extract, obtained previously [ref. 31], and which contains benzophenones GUT/XAN and OBL at 16.68% and 42.25% of the total extract content, respectively [ref. 31]. Also, in this work, the effects of isolated compounds, GUT/XAN and OBL, were evaluated.
For the screening of trypanocidal activity, the quantification of live T. cruzi trypomastigotes was performed using light microscopy and direct counting in a Neubauer chamber, considered the gold standard for phenotypic assays [ref. 32]. Although motility‐based evaluation may not fully capture viability, since some effects can be reversible, this method remains widely used in Trypanosoma spp. screening due to its simplicity and reliability [ref. 32]. Additionally, the use of C2C12 myoblasts as host cells for the amastigote assay can be justified by their robustness and susceptibility to T. cruzi infection [ref. 33]. Nevertheless, complementary approaches using human‐derived cell lines are warranted to validate these results and enhance translational relevance. Against trypomastigote forms, our results demonstrated significant trypanocidal activity of SEBz at all evaluated concentrations. SEBz exhibited a flagellar motility inhibition of 70.59% and 47.46% at concentrations of 50 and 25 µg mL−1, respectively, at 24 h (Figure 2A), and a flagellar motility inhibition of 68.71% and 49.09% at concentrations of 25 and 12.5 µg mL−1, respectively, at 48 h (Figure 2A). The 50% effective concentration (EC50) values for 24 and 48 h were 21.64 and 17.97 µg mL−1, respectively (Table 1). Against amastigote forms, SEBz exhibited significant activity at concentrations ranging from 12.5 to 3.12 µg mL−1, showing a percentage reduction of amastigote forms of 60.55% at the concentration of 12.5 µg mL−1 after 48 h of incubation (Figure 2B) and an EC50 value of 6.83 µg mL−1 (Table 1). The antiparasitic activity of extracts can be classified against protozoa as highly active (EC50 < 10 µg mL−1), active (EC50 between 10 and 50 µg mL−1), moderately active (EC50 between 50 and 100 µg mL−1), and non‐active (EC50 >100 µg mL−1) [ref. 34]. In this sense, SEBz was considered highly active against the amastigote forms of T. cruzi (Y strain).
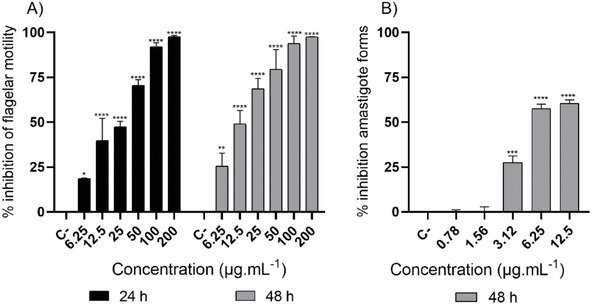
TABLE 1: In vitro cytotoxicity, hemolytic, and trypanosomal activities.
| CC50 | EC50 | SI | HC50 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C2C12 | Trypomastigote | Amastigote | Trypomastigote | Amastigote | |||||
| 24 h | 48 h | 24 h | 48 h | 48 h | 24 h | 48 h | 48 h | 3 h | |
| SEBz | 51.32(45.67–57.17) | 42.80(28.07–62.35) | 21.64(17.55–26.47) | 17.97(12.75–24.75) | 6.83(4.99–10.24) | 2.37 | 2.38 | 6.26 | >200 |
| GUT/XAN | 18.61(12.54–26.65) | 12.70(11.67–13.87) | 7.85(6.25–9.78) | 5.15(4.13–6.34) | 7.91(7.04–8.96) | 2.37 | 2.46 | 0.62 | >50 |
| OBL | 12.64(9.27–17.07) | 11.53(10.99–12.07) | 6.92(5.71–8.33) | 4.74(3.77–5.88) | 11.63(9.24–13.94) | 1.82 | 2.43 | 1.00 | >50 |
| BNZ | 59.36(41.69–127.40) | 47.76(29.49–115.80) | 8.33(6.05–11.41) | 3.59(2.94–4.30) | 5.49(4.53–6.47) | 7.12 | 13.30 | 8.70 | >50 |
CC50 – cytotoxic concentration to 50% of the cells. EC50 – effective concentration to 50% of the parasites. SI – selectivity index. HC50 – hemolytic concentration to 50% of red cells. C2C12 – myoblast cell line (ATCC: CRL‐1772). SEBz – polyprenylated benzophenone enriched Brazilian red propolis extract t (µg mL−1). GUT/XAN – guttiferone E/xanthochymol (µM); OBL – oblongifolin B (µM), and BNZ – benznidazole (µM). Concentration between parentheses represents a 95% confidence interval.
Although interest in the effects of propolis extracts on parasitic diseases has increased, studies evaluating the efficacy of Brazilian red propolis against T. cruzi, particularly the Y strain, remain limited [ref. 19, ref. 23, ref. 24, ref. 25, ref. 26]. Previous investigations have shown that red propolis extracts display antiparasitic activity against T. cruzi, although most focused on early parasite stages or used non‐enriched extracts [ref. 19, ref. 24]. Ethanolic and hydroethanolic red propolis extracts from different Brazilian regions exhibited moderate to high activity at concentrations ranging from 75 to 500 µg mL− 1 against epimastigote forms of T. cruzi, which correspond to the replicative stage in the invertebrate host [ref. 19, ref. 24]. Studies have also shown notable activity of red propolis extracts against Trypanosoma brucei, the causative agent of African trypanosomiasis [ref. 23, ref. 26, ref. 27]. Extracts from both Nigeria and Brazil, particularly those rich in phenolic compounds, displayed moderate to high activity, and the observed efficacy was correlated with total phenolic content [ref. 23, ref. 26, ref. 27]. However, the specific contribution of benzophenone derivatives was not assessed in those studies.
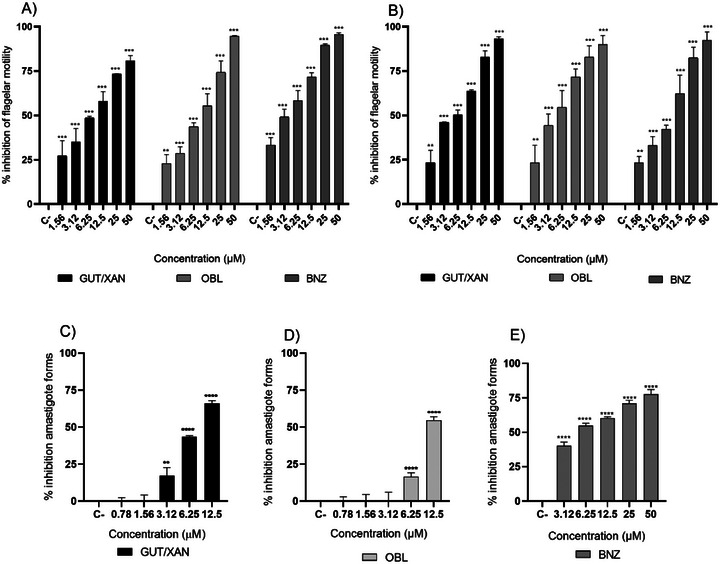
Given the variation in the chemical composition of propolis depending on its botanical and geographical origin, further phytochemical characterization is essential to identify the active constituents responsible for the observed activity [ref. 21, ref. 29]. In this context, the compounds GUT/XAN and OBL also exhibited significant trypanocidal activity against trypomastigote forms at all evaluated concentrations, similar to the standard drug BNZ. At 24 h, the inhibition of flagellar motility was 74.32%, 73.32%, and 82.43% at a concentration of 25 µM for GUT/XAN, OBL, and BNZ, respectively (Figure 2A). At 48 h, the inhibition of flagellar motility was 82.79% for GUT/XAN and OBL, and 89.49% for BNZ at a concentration of 25 µM (Figure 3B). The EC50 values at 24 h were 7.85, 6.92, and 8.33 µM (4.74, 4.18, and 2.17 µg mL−1) for GUT/XAN, OBL, and BNZ, respectively, and at 48 h, the EC50 values were 5.15, 4.74, and 3.59 µM (3.11, 2.86, and 0.93 µg mL−1), respectively (Table 1). Also, significant activity of GUT/XAN was observed, with a percentage reduction of amastigote forms of 66.07% at the concentration of 12.5 µM after 48 h of incubation (Figure 3C). OBL showed significant activity only at concentrations of 12.5 and 6.25 µM, with a percentage reduction of amastigote forms of 54.67% at the concentration of 12.5 µM, also after 48 h of incubation (Figure 3D). BNZ showed significant activity at all tested concentrations, with a percentage reduction of amastigote forms of 77.73% at the concentration of 50 µM (Figure 3E). The compound GUT/XAN exhibited the lowest EC50 value (7.91 µM; 4.78 µg mL− 1), which was comparable to that of BNZ (EC50 = 5.49 µM; 1.43 µg mL− 1). In contrast, OBL showed the highest EC50 value (11.63 µM; 7.01 µg mL− 1) (Table 1). The superior activity of SEBz and its isolated constituents (GUT/XAN and OBL) validates the strategy of targeting these specific compounds, which are the main active agents, for enhanced efficacy and standardization. While crude extracts are complex and variable, the enriched profile provides a more potent and reproducible antiparasitic effect, with the isolated benzophenones demonstrating high micromolar potency against the clinically relevant form, offering novel insights into the mechanism of action through observed ultrastructural damage [ref. 19, ref. 24, ref. 26].
The trypanocidal and leishmanicidal potential of prenylated benzophenones, particularly guttiferone A, has been widely documented [ref. 35, ref. 36, ref. 37]. Although these compounds share the same chemical backbone, subtle variations in the number and position of prenyl and hydroxyl groups attached to the benzophenone core can lead to distinct biological activities [ref. 38]. Guttiferone A has shown potent effects against protozoan parasites such as Plasmodium falciparum, likely through interference with mitochondrial redox balance [ref. 35]. Further studies revealed that guttiferone A and the related benzophenone nemorosone inhibit mitochondrial respiratory complexes II and III, disrupting parasite bioenergetics and redox homeostasis [ref. 36]. Efforts to enhance the bioactivity and selectivity of guttiferone A have resulted in several generations of semisynthetic analogues with improved antiparasitic potency against T. brucei, Leishmania donovani, and P. falciparum [ref. 39, ref. 40]. These findings reinforce that targeted structural modifications of the benzophenone scaffold can optimize both pharmacological and physicochemical properties. Overall, our findings are consistent with the literature, suggesting that the biological activity of SEBz is likely associated with the presence of benzophenone derivatives such as GUT/XAN and OBL.
To investigate the morphological effects of SEBz and its isolated constituents on T. cruzi, transmission electron microscopy (TEM) was performed on trypomastigote forms incubated for 48 h with the extract and isolated compounds at their respective EC50 concentrations. Control parasites exhibited preserved nuclear morphology and a kinetoplast with normal size and organization (Figure 4A). In contrast, parasites exposed to SEBz or its isolated compounds displayed evident morphological disruptions, including nuclear deformation and swelling of the kinetoplast. Notably, the presence of cytoplasmic vacuoles was more prominent in parasites incubated with GUT/XAN and OBL (Figure 4D,E). Alterations in the integrity of the plasma membrane were also observed in parasites incubated with GUT/XAN, OBL (Figure 4D,E), and BNZ (Figure 4B). Furthermore, all groups exposed to the extract and compounds, including those exposed to the reference drug BNZ, demonstrated a characteristic rounding of the parasite body, a feature absent in the control group. These findings suggest that both SEBz and its bioactive constituents exert marked ultrastructural effects on T. cruzi trypomastigotes, potentially compromising parasite viability.
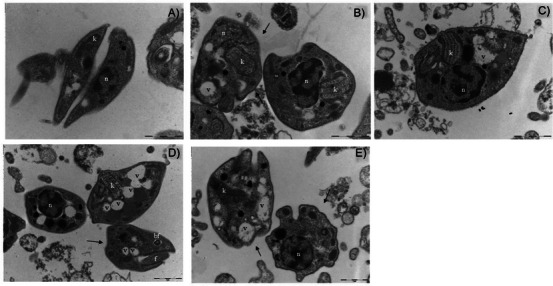
One of the most striking morphological alterations observed was the swelling of the kinetoplast in all experimental groups, with the most pronounced changes detected in parasites treated with BNZ and SEBz. The kinetoplast is a distinctive and essential mitochondrial DNA‐containing structure in kinetoplastid parasites, playing a key role in mitochondrial metabolism, cell division, and parasite infectivity [ref. 41]. Structural damage or swelling of the kinetoplast has been associated with mitochondrial membrane depolarization and impaired energy metabolism, ultimately leading to parasite death [ref. 42]. Polyprenylated benzophenone compounds are known for their pro‐oxidant activity at high concentrations and their ability to target mitochondrial structures [ref. 43]. Consistent with these prior observations, the effects observed may be related to redox imbalance and mitochondrial disruption induced by SEBz and its active constituents, contributing to the structural disintegration observed in trypomastigotes. Nevertheless, further studies integrating biochemical assays with transcriptomic and proteomic analyses will be essential to elucidate the precise molecular targets and pathways affected by red propolis‐derived compounds.
The superior activity of our polyprenylated benzophenone‐enriched extract (SEBz) and its isolated constituents (GUT/XAN and OBL) validates the strategy of targeting these specific compounds, which are the main active agents, for enhanced efficacy and standardization. While crude extracts are complex and variable, the enriched profile provides a more potent and reproducible antiparasitic effect, with the isolated benzophenones demonstrating high micromolar potency against the clinically relevant amastigote form, offering novel insights into the mechanism of action through observed ultrastructural damage.
The evaluation of toxicity represents a fundamental step in the screening of natural products for potential chemotherapeutic applications [ref. 19, ref. 31]. In the present study, the cytotoxic effects of SEBz and the compounds were assessed using the C2C12 mouse myoblast cell line. As shown in Table 1, SEBz exhibited lower cytotoxicity compared to the isolated compounds, with 50% cytotoxicity concentration (CC50) values of 51.32 µg mL−1 at 24 h and 42.80 µg mL−1 at 48 h. Despite this, the selectivity index (SI) indicated that SEBz was approximately 2‐fold more selective against the trypomastigote forms and 6‐fold more selective against the amastigote forms of the parasite, when compared to its effect on mammalian cells. In contrast, the isolated compounds GUT/XAN and OBL demonstrated higher cytotoxicity, with CC50 values of 18.61 and 12.64 µM (equivalent to 11.23 and 7.62 µg mL−1) at 24 h, and 12.70 and 11.53 µM (7.66 and 6.96 µg mL−1) at 48 h, respectively. These compounds were approximately 2.5 times more effective against trypomastigote forms; however, their selectivity against amastigotes was limited, with SI values ranging from 1.11 to 1.82, indicating a narrow therapeutic window (Table 1). For comparison, the reference drug BNZ presented CC50 values greater than 50 µM at 24 h, and demonstrated higher selectivity, with SI values of approximately 13 and 9 against trypomastigote and amastigote forms, respectively, at 48 h. Additionally, no hemolysis was observed under the experimental conditions evaluated, indicating a favorable safety profile regarding erythrocyte integrity.
Previous studies corroborate the moderate to high cytotoxic potential of red propolis and its constituents. In non‐tumorigenic breast epithelial cells (MCF‐10A), SEBz, GUT/XAN, and OBL showed CC50 values of 19.58 µg mL−1, 18.23 µM, and 31.46 µM, respectively [ref. 31]. Similarly, hydroalcoholic extracts of red propolis from different Brazilian regions showed CC50 values ranging from 65 to 85 µg mL−1 in murine embryo fibroblasts (BALB/c 3T3) [ref. 44], supporting the notion that, despite their biological activity, red propolis extracts may exert significant cytotoxic effects on non‐target cells. Although the low in vitro SI values observed for GUT/XAN and OBL indicate a narrow therapeutic window, this finding does not preclude their potential as antiparasitic leads. When compared with other natural trypanocidal agents, the SI values obtained here fall within the lower range of those reported, which often exhibit modest selectivity (SI ≈ 2–4) prior to chemical optimization [ref. 45, ref. 46]. In contrast, optimized derivatives or semisynthetic analogues generally achieve SI > 10, a threshold commonly regarded as indicative of a promising lead compound [ref. 46, ref. 47]. Thus, the current data suggest that SEBz and its benzophenone constituents represent preliminary scaffolds requiring further refinement to enhance safety and selectivity. Complementary strategies may help address these limitations. For example, structure–activity relationship (SAR) studies could guide targeted modifications of prenyl or hydroxyl groups to attenuate cytotoxicity while maintaining antiparasitic potency, and formulation or delivery approaches, including encapsulation or nanoparticle systems, could limit host exposure while improving pharmacokinetics, thereby expanding the therapeutic window. Additionally, further cytotoxicity profiling in mammalian cells, combined with mechanistic assays, will help determine whether host‐cell damage occurs.
Conclusions
Given the results obtained, it can be concluded that SEBz exhibits potent in vitro trypanocidal activity against the amastigote forms of Trypanosoma cruzi (Y strain), with moderate cytotoxicity. Among the compounds evaluated, GUT/XAN and OBL also demonstrated activity against both amastigote and trypomastigote forms of T. cruzi (Y strain), though with lower selectivity relative to mammalian cells. These findings pave the way for further investigations aimed at elucidating the mechanisms of action of these compounds and other propolis‐derived extracts. Additionally, in vivo studies would be highly valuable to better understand the antiparasitic, cytotoxic, and immunomodulatory properties of Brazilian red propolis extract and/or its active constituents. Moreover, these studies will also contribute to expanding the potential medicinal applications of this type of bee product.
Experimental
Preparation of Extract and Isolation of Compounds
Red propolis was purchased from Cooperativa de Apicultores de Canavieiras in the city of Canavieiras (Bahia state, Brazil) in April 2018. The polyprenylated benzophenone‐enriched extract and its constituents, the mixture of GUT/XAN and OBL, were obtained previously as described by Pires et al. [ref. 31]. Chromatographic analyses of SEBz were performed using reverse phase‐high‐performance liquid chromatography‐photo diode array (RP‐HPLC‐PDA) with a Shim‐pack VP‐ODS column (250 mm × 4.6 mm i.d., 5 µm; Shimadzu, Kyoto, Japan, Cat# 228‐34937‐92). The flow rate was set at 1 mL/min, and ultraviolet detection was conducted at 250 nm. A mobile phase gradient system was employed, consisting of 0.1% acetic acid in water (solvent A) (Sigma–Aldrich, St. Louis, USA, Cat#33209) and acetonitrile (solvent B) (Sigma‐Aldrich, Cat#34967). The gradient ranged from 85% to 100% of solvent B over 20 min. The column temperature was maintained at 40°C, and the injected volume was 20.0 µL. The analytical equation for GUT/XAN was y = 0.003x – 0.040 (r 2 = 0.998; limit of detection [LOD] = 0.462 µg mL−1 and quantification [LOQ] = 1.400 µg mL−1; the recovery mean was 92.422%). As for OBL, the analytical equation was y = 0.001x ‐ 0.015 (r2 = 0.996; LOD = 5.057 µg mL−1 and LOQ = 15.323 µg mL−1; the recovery mean was 96.888%) [ref. 31].
The purity of the isolated compound OBL and the mixture of double bond isomers GUT/XAN were estimated to be greater than 98% through both HPLC and nuclear magnetic resonance (NMR) analysis [ref. 31]. The chemical structures of the compounds were determined using one‐ and two‐dimensional 1H and 13C NMR experiments and compared with the existing literature data [ref. 14].
Maintenance of Parasites and Mammalian Cells
T. cruzi (Y strain) was kindly provided by Professor Dr. Sergio de Albuquerque (Faculty of Pharmaceutical Sciences of Ribeirão Preto, University of São Paulo, Ribeirão Preto, Brazil) and maintained in vivo in male BALB/c mice (Taconic Biosciences, RRID:IMSR_TAC:BALB), obtained at the Central Animal Facility of the Ribeirão Preto, through routine blood passages at the peak of parasitemia (7 days post‐infection). Bloodstream trypomastigotes were collected via cardiac puncture and subsequently used to infect cell cultures in vitro. All experimental procedures were approved by the Animal Ethics Committee of the University of Franca (Approval number: 2061190421) and conducted in accordance with Brazilian regulations and institutional guidelines for good animal care practices.
C2C12 cells (Mus musculus myoblast cell line, RRID:CVCL_0188) were obtained from the Cell Bank of Rio de Janeiro (HUCFF, UFRJ, RJ, Brazil) and cultured in Dulbecco’s Modified Eagle Medium (DMEM; Cultilab, Campinas, SP, Brazil, Cat# 460) supplemented with 5% heat‐inactivated fetal bovine serum (FBS; Cultilab, Campinas, Brazil, #Cat 63), penicillin (10 000 IU mL−1), and streptomycin (10 mg mL−1) (Sigma–Aldrich, Cat# 4333). Cultures were incubated at 37 °C in a humidified atmosphere containing 5% CO2 incubator (Sanyo, Osaka, Japan). For cell infection, C2C12 cells were exposed to bloodstream trypomastigotes at a ratio of 10 parasites per myoblast (10:1) in DMEM culture medium (Cultilab) and incubated following the standard operating procedures established in the laboratory for 7 days. Trypomastigotes were subsequently collected from the culture supernatant as described by Pagotti et al. (2021) [ref. 5].
In Vitro Trypanocidal Assay
For the experiment against the trypomastigote forms, the SEBz and GUT/XAN, OBL, and BNZ (LAFEPE—Pernambuco, BR) were previously dissolved in dimethyl sulfoxide (DMSO, Synth‐ Diadema, Brazil, Cat# 01D1011.01.BJ) and added to the wells of 96‐well plates (Kasvi—São José dos Pinhais, Brazil, Cat# K26‐096 V‐N) by serial dilution at concentrations ranging from 6.25 to 200 µg mL−1 for the extract and from 1.56 to 50 µM for the compounds. Next, the trypomastigote form was adjusted to a final concentration of 1 x 106 parasites/well in supplemented DMEM medium (Cultilab) and added to each well. The plates were incubated at 37 °C in a humidified atmosphere containing 5% CO2 (Sanyo) for 24 and 48 h. After incubation, trypanocidal activity was evaluated by quantifying the number of viable (motile and morphologically intact) parasites using a Neubauer chamber (Kasvi, Cat# K5‐0011) under an optical microscope (Nikon, New York, USA) [ref. 5].
To evaluate the activity against the intracellular amastigote forms, C2C12 cells were seeded at a density of 5 × 105 cells/mL in 24‐well plates (Kasvi, Cat# K12‐024), each containing a 13 mm round coverslip (Kasvi, Cat# K5‐0013), and incubated for 24 h at 37 °C in a humidified atmosphere with 5% CO2 (Sanyo). After incubation, non‐adherent cells were removed by washing with fresh culture medium, and the adherent cells were infected with bloodstream trypomastigotes at a ratio of 10 parasites per myoblast (10:1) for 48 h under the same incubation conditions [ref. 33]. Following infection, non‐internalized trypomastigotes were removed by washing the wells with fresh culture medium. SEBz and the compounds GUT/XAN, OBL, and BNZ (LAFEPE) were then added at concentrations previously determined to maintain at least 60% cell viability, as assessed by the 3‐(4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyl tetrazolium bromide (MTT) assay. The extract and compounds were applied for 48 h at concentrations ranging from 0.78 to 12.5 µg mL−1 for the extract and 0.78 to 12.5 µM for the compounds, except for BNZ, which was tested at concentrations ranging from 1.56 to 50 µM. After the period, cells were washed with phosphate‐buffered saline (PBS, 1X), fixed with methanol (Synth, Cat# 162728247) for 5 min, and stained with Giemsa (Synth, Cat# 00G1003.08.AB) for 30 min. Coverslips were mounted on slides and examined under transmitted light microscopy (Nikon) at 100× magnification. The number of intracellular amastigotes was determined by randomly counting 200 cells per sample. Negative control groups (trypomastigote and intracellular amastigote forms) were maintained under the same conditions and cultured in DMEM containing 0.1% DMSO (Synth) [ref. 5].
Ultrastructural Damage Analysis
To assess ultrastructural changes in the parasites, TEM was employed. Trypomastigote forms (1 × 106 cells/mL) were exposed to SEBz and the compounds GUT/XAN, OBL, and BNZ (LAFEPE) at their respective EC50 concentrations for 24 h. After incubation, the parasites were centrifuged and fixed in a solution of 3% glutaraldehyde (Sigma‐Aldrich, Cat# G7651) prepared in 0.1 M phosphate buffer (pH 7.2) for 24 h. Following fixation, the samples were washed twice with PBS and prepared for TEM, according to the methodology described by Candido et al. [ref. 49]. Microscopic analysis was performed using a JEOL JEM‐100CXII transmission electron microscope (JEOL). Parasites cultured in DMEM medium containing 0.1% DMSO served as the negative control.
Cytotoxic Activity
C2C12 myoblast cells were counted using a Neubauer chamber (Kasvi) and adjusted to a concentration of 2 × 105 cells/well. Subsequently, they were seeded in a 96‐well culture plate with supplemented DMEM medium (Cultilab). The extract and compounds previously dissolved in DMSO (Synth) were added at concentrations ranging from 6.25 to 200 µg mL−1 for the extract and from 1.56 to 50 µM for the compounds and incubated at 37°C in the presence of 5% CO2 (Sanyo) for 24 and 48 h. Afterward, the cells were washed, and 20 µL of a solution containing 7 mg of MTT (Sigma–Aldrich, Cat# 475989) in PBS was added to each well. The plates were incubated at 37°C and in a 5% CO2 atmosphere for 4 h, and the formazan precipitate was solubilized with 100 µL of isopropyl alcohol (Sigma‐Aldrich, Cat# 563935). The absorbance was read at 550 nm with a spectrophotometer (Libra S12; Biochrom Corp, Miami, USA) [ref. 50]. Cells were incubated with DMEM containing 0.1% DMSO as the negative control, and with 25% DMSO as the positive control for cytotoxicity [ref. 51].
Hemolytic Activity
The hemolytic activity assay was conducted as described by Lazcano‐Pérez [ref. 52] with adaptations. Approximately 5 mL of a 3% suspension of defibrinated sheep erythrocytes (Laborclin‐Pinhais, Brazil, Cat# 520246) was prepared in 0.9% saline solution (Synth, Cat# 01C1060.01.AG). Approximately 50 µL of diluted erythrocytes was added to each well of a sterile 96‐well plate (Kasvi), and the samples previously diluted in DMSO were added at concentrations ranging from 6.25 to 200 µg mL−1 for the extracts and from 1.56 to 50 µM. The plates were then incubated at room temperature for 3 h, and the hemolysis was determined by hemoglobin release, quantified by the absorbance of the supernatants at 415 nm, read using a spectrophotometer (Libra S12 – Biochrom). The negative control consisted of erythrocytes in a 0.9% NaCl solution containing 0.1% DMSO, while the positive control consisted of erythrocytes in water.
Statistical Analysis
In vitro experiments were performed in triplicate and repeated three times. The EC50, CC50, and HC50 values were calculated as the mean obtained from each individual experiment using nonlinear regression of dose–response curves. The SI, which indicates parasite toxicity as compared to the host, was calculated as the ratio between CC50 and EC50. Data were statistically analyzed by two‐way analysis of variance followed by Dunnett’s comparison. The statistical analyses were performed by using GraphPad Prism 5 (GraphPad Software, San Diego, California, USA, RRID:SCR_002798).
Author Contributions
Nicoli D. Oliveira, Mariana C. Pagotti, Lucas A. de L. Paula, Mariana Z. Paixão, and Daiane A. dos Santos performed the biological assays, analyzed the data, and wrote the manuscript. Rodrigo C. S. Veneziani and Renata Faleiros de Freitas performed the phytochemical procedures. Sérgio R. Ambrósio, Jairo K. Bastos, and Lizandra G. Magalhães designed the experiments, project administration, and funding acquisition.
Funding
This study was supported by the National Council for Scientific and Technological Development, Brazil–CNPq (Fellowships: 308903/2021‐8, 307778/2021‐5, 304425/2025‐7, 307707/2021‐0, and 304423/2025‐4) Coordination for the Improvement of Higher Education Personnel, Brazil‐CAPES (Finance code 001, process number 88887.605060/2021‐00) and São Paulo Research Foundation, Brazil‐FAPESP (2017/04138‐8 and 2018/05080‐6). The Article Processing Charge for the publication of this research was funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior ‐ Brasil (CAPES) (ROR identifier: 00x0ma614).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Chagas Disease (American trypanosomiasis) Fact Sheet. Available at Chagas disease (also known as American trypanosomiasis) (Accessed November 14,. 2025
- Chagas Disease: Symptoms, Transmission, and Current Treatments. Available at:. 2025
- Further Investigations of Nitroheterocyclic Compounds as Potential Antikinetoplastid Drug Candidates,”. Biomolecules, 2023. [DOI | PubMed]
- Natural Products as a Source for Treating Neglected Parasitic Diseases,”. International Journal of Molecular Sciences, 2013. [DOI | PubMed]
- Trypanocidal Activity of Dysphania ambrosioides, Lippia alba, and Tetradenia riparia Essential Oils Against Trypanosoma Cruzi,”. Chemistry and Biodiversity, 2021. [DOI | PubMed]
- Exploring the Potential of Natural Products as Antiparasitic Agents for Neglected Tropical Diseases,”. Current Topics in Medicinal Chemistry, 2024. [DOI | PubMed]
- Anti‐Trypanosoma, anti‐Leishmania and cytotoxic activities of natural products from Psidium brownianum Mart. ex DC. and Psidium guajava var. Pomifera analysed by LC–MS,”. Acta Tropica, 2017. [PubMed]
- The Immunomodulatory and Anticancer Properties of Propolis,”. Clinical Reviews in Allergy & Immunology, 2013. [DOI | PubMed]
- Comparação entre a composição química e capacidade antioxidante de diferentes extratos de própolis verde,”. 2016. [DOI]
- Propolis: A Wonder Bees Product and Its Pharmacological Potentials,”. Advances in Pharmacological Sciences, 2013. [DOI]
- Antiparasitic Properties of Propolis Extracts and Their Compounds,”. Chemistry and Biodiversity, 2021. [DOI | PubMed]
- Propolis Research and the Chemistry of Plant Products,”. Natural Product Reports, 2011. [DOI | PubMed]
- Origin and Chemical Variation of Brazilian Propolis,”. Evidence‐Based Complementary and Alternative Medicine, 2005. [DOI | PubMed]
- Dalbergia ecastaphyllum (L.) Taub. And Symphonia globulifera L.f.: The Botanical Sources of Isoflavonoids and Benzophenones in Brazilian Red Propolis,”. Molecules, 2020. [DOI | PubMed]
- Baccharis dracunculifolia and Dalbergia ecastophyllum, Main Plant Sources for Bioactive Properties in Green and Red Brazilian Propolis,”. Plants, 2020. [DOI | PubMed]
- Neovestitol, an Isoflavonoid Isolated From Brazilian Red Propolis, Reduces Acute and Chronic Inflammation: Involvement of Nitric Oxide and IL‐6,”. Scientific Reports, 2016. [DOI | PubMed]
- The Antibacterial Potential of Brazilian Red Propolis Against the Formation and Eradication of Biofilm of Helicobacter pylori ,”. Antibiotics, 2024. [DOI | PubMed]
- Antiproliferative Flavanoid Dimers Isolated From Brazilian Red Propolis,”. Journal of Natural Products, 2020. [DOI | PubMed]
- Antitrypanosomal, Antileishmanial and Cytotoxic Activities of Brazilian Red Propolis and Plant Resin of Dalbergia ecastaphyllum (L) Taub,”. Food and Chemical Toxicology, 2018. [DOI | PubMed]
- Effects of Brazilian Propolis on Leishmania amazonensis ,”. Memorias Do Instituto Oswaldo Cruz, 2007. [DOI | PubMed]
- Parasite Killing of Leishmania (V) Braziliensis by Standardized Propolis Extracts,”. Evidence‐Based Complementary and Alternative Medicine, 2017. [DOI | PubMed]
- Antileishmanial Activity and Chemical Composition From Brazilian geopropolis Produced by Stingless Bee Melipona Fasciculata,”. Revista Brasileira de Farmacognosia, 2019. [DOI]
- Chemical Characterisation of Nigerian Red Propolis and Its Biological Activity Against Trypanosoma brucei ,”. Phytochemical Analysis, 2016. [DOI | PubMed]
- Antioxidant, Antimicrobial, Antiparasitic, and Cytotoxic Properties of Various Brazilian Propolis Extracts,”. PLoS ONE, 2017. [DOI | PubMed]
- Anti‐Trypanosoma cruzi Potential of Vestitol Isolated From Lyophilized Red Propolis,”. Molecules, 2023. [DOI | PubMed]
- The Activity of Red Nigerian Propolis and Some of Its Components Against Trypanosoma brucei and Trypanosoma congolense ,”. Molecules, 2023. [DOI | PubMed]
- Evaluation of the Antitrypanosomal Activity, Cytotoxicity and Phytochemistry of Red Brazilian Propolis,”. PLoS ONE, 2024. [DOI | PubMed]
- Chemical Composition, Immunostimulatory, Cytotoxic and Antiparasitic Activities of the Essential Oil From Brazilian Red Propolis,”. PLoS ONE, 2018. [DOI | PubMed]
- Propolis Volatile Compounds: Chemical Diversity and Biological Activity: A Review,”. Chemistry Central Journal, 2014. [DOI | PubMed]
- Best Practice in Research: Consensus Statement on Ethnopharmacological Field Studies—ConSEFS,”. Journal of Ethnopharmacology, 2018. [DOI | PubMed]
- Polyprenylated Benzophenones From Brazilian Red Propolis: Analytical Characterization and Anticancer Activity,”. Chemistry and Biodiversity, 2024. [DOI | PubMed]
- Experimental Models in Chagas Disease: A Review of the Methodologies Applied for Screening Compounds Against Trypanosoma cruzi ,”. Parasitology Research, 2018. [DOI | PubMed]
- Synthesis and Biological Evaluation of New Chalcogen Semicarbazone (S,Se) and Their Azole Derivatives Against Chagas Disease,”. Journal of Medicinal Chemistry, 2024. [DOI | PubMed]
- Antiprotozoal and Cytotoxic Activities in Vitro of Colombian Annonaceae,”. Journal of Ethnopharmacology, 2007. [DOI | PubMed]
- Antimicrobial Evaluation of the Polyisoprenylated Benzophenones Nemorosone and Guttiferone A,”. Phytotherapy Research, 2011. [DOI | PubMed]
- Role of Mitochondria in the Leishmanicidal Effects and Toxicity of Acyl Phloroglucinol Derivatives: Nemorosone and Guttiferone A,”. Parasitology, 2015. [DOI | PubMed]
- Leishmanicidal Activity of Benzophenones and Extracts From Garcinia brasiliensis Mart. Fruits,”. Phytomedicine, 2010. [DOI | PubMed]
- Structural Diversity and Bioactivities of Natural Benzophenones,”. Natural Product Reports, 2014. [DOI | PubMed]
- Third‐Generation Analogues of Guttiferone A,”. Bioorganic & Medicinal Chemistry Letters, 2025. [DOI | PubMed]
- Synthesis of Novel Guttiferone A Derivatives: In‐vitro Evaluation Toward Plasmodium falciparum, Trypanosoma brucei and Leishmania donovani ,”. European Journal of Medicinal Chemistry, 2013. [DOI | PubMed]
- Mitochondrion of Protozoan Parasite Emerges as Potent Therapeutic Target: Exciting Drugs Are on the Horizon,”. Current Pharmaceutical Design, 2008. [DOI | PubMed]
- Kinetoplastid Species Maintained by a Small Mammal Community in the Pantanal Biome,”. Pathogens, 2022. [DOI | PubMed]
- A Photoalkylative Fluorogenic Probe of Guttiferone A for Live Cell Imaging and Proteome Labeling in Plasmodium falciparum ,”. Molecules, 2020. [DOI | PubMed]
- Antimicrobial and Cytotoxic Activity of Red Propolis: An Alert for Its Safe Use,”. Journal of Applied Microbiology, 2015. [DOI | PubMed]
- In Vitro Phenotypic Activity and In Silico Analysis of Natural Products From Brazilian Biodiversity on Trypanosoma Cruzi,”. Molecules, 2021. [DOI | PubMed]
- Antiprotozoal and Cytotoxic Screening of 45 Plant Extracts From Democratic Republic of Congo,”. Journal of Ethnopharmacology, 2008. [DOI | PubMed]
- Natural‐Product‐Inspired Design and Synthesis of Two Series of Compounds Active Against Trypanosoma Cruzi: Insights Into Structure–Activity Relationship, Toxicity, and Mechanism of Action,”. Bioorganic Chemistry, 2022. [DOI | PubMed]
- Improving the Drug‐Likeness of Inspiring Natural Products—Evaluation of the Antiparasitic Activity Against Trypanosoma cruzi Through Semi‐Synthetic and Simplified Analogues of Licarin A,”. Scientific Reports, 2020. [DOI | PubMed]
- Efficacy of Diterpene Polyalthic Acid Combined With Amphotericin B Against Leishmania Amazonensis in Vitro,”. Pharmaceuticals, 2024. [DOI | PubMed]
- MTT Colorimetric Assay for Testing Macrophage Cytotoxic Activity in Vitro,”. Journal of Immunological Methods, 1990. [DOI | PubMed]
- Unexpected Low‐Dose Toxicity of the Universal Solvent DMSO,”. FASEB Journal, 2014. [DOI | PubMed]
- Hemolytic, Anticancer and Antigiardial Activity of Palythoa caribaeorum Venom,”. Journal of Venomous Animals and Toxins Including Tropical Diseases, 2018. [DOI | PubMed]
