Exploring the Chemical Fingerprint and In Vitro Biocompatibility of Wild‐Growing and Ex Situ Cultivated Stachys cretica subsp. cretica in the Same Region (Crete, Greece)
Abstract
The genus Stachys (Lamiaceae) comprises numerous species recognized for their ethnopharmacological importance and rich chemical profiles. In this study, we investigated the chemical composition and biocompatibility of both wild‐growing Cretan samples and asexually propagated samples of Stachys cretica L. subsp. cretica that were ex situ cultivated in the same region (Crete). The methanol (MeOH) extracts and infusions were analyzed using high‐performance liquid chromatography‐photodiode array detection coupled with mass spectrometry (HPLC‐PDA‐MS) and proton nuclear magnetic resonance (1H‐NMR) spectroscopy. A total of 23 compounds were identified in the analyzed samples by HPLC‐PDA‐MS, classified as flavonoids, phenylethanoid glycosides, and phenolic acids. The NMR screening confirmed the presence of chlorogenic acid, acteoside, lavandulifolioside, and leucosceptoside A in the MeOH extract of wild‐growing S. cretica subsp. cretica. Methanol extracts exhibited higher chemical diversity compared to infusions, particularly in flavonoids. Moreover, biocompatibility is crucial for most biomedical and pharmaceutical applications, ensuring that drugs, drug delivery systems, and cosmetics avoid possible side effects or cytotoxicity. Thus, normal human embryonic kidney cells (HEK293) were cultivated and cell viability (%) was estimated. This confirmed that S. cretica subsp. cretica extracts are biocompatible. The limited phytochemical variation observed between wild‐growing and cultivated samples suggests that cultivation within the species’ native range may preserve its chemical composition.
Article type: Research Article
Keywords: cytotoxicity, HPLC‐PDA‐MS, Lamiaceae, natural products, NMR
Affiliations: Department of Pharmacy Section of Pharmacognosy and Chemistry of Natural Products School of Health Sciences National and Kapodistrian University of Athens Athens Greece; Laboratory of Pharmacognosy School of Pharmacy Aristotle University of Thessaloniki University Campus Thessaloniki Greece; Department of Pharmacy University of Pisa Pisa Italy; Institute of Plant Breeding and Genetic Resources Hellenic Agricultural Organization Demeter (ELGO‐Dimitra) Thermi Greece; Department of Viticulture Floriculture & Plant Protection Institute of Olive Tree Subtropical Crops and Viticulture Hellenic Agricultural Organization‐Demeter (ELGO‐Dimitra) Athens Greece; Department of Basic Medical Sciences Laboratory of Biology Medical School National and Kapodistrian University of Athens Greece; Biomedical Research Foundation Academy of Athens Athens Greece
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley-VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202503054 | PubMed: 41494992 | PMC: PMC12774863
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.2 MB)
Introduction
The genus Stachys L. includes approximately 370 species (or 435 species and subspecies) and is recognized as one of the largest genera in the Lamiaceae family [ref. 1], predominantly found in the world’s warm temperate zones, the Mediterranean, and Southwest Asia. Its distribution also extends to secondary regions in North and South America, along with southern Africa [ref. 2]. Most Stachys species thrive in diverse habitats, including forests, rocky areas, and limestone environments, and they can be classified as annual or perennial herbs, as well as small shrubs [ref. 3]. In Greece, there are 54 Stachys taxa (species and subspecies), with a notable proportion being local endemics confined to the region [ref. 4]. According to Bhattacharjee’s classification (1980) [ref. 5], the genus Stachys is divided into two subgenera, Stachys and Betonica, with the former consisting of 19 sections and the latter containing two. In this classification, Stachys cretica L. subsp. cretica is placed in the section Eriostomum. This subspecies is a Steno‐Mediterranean entity primarily found in the eastern Mediterranean region, spanning from Krym (Transcaucasus) and Turkey‐in‐Europe to the Balkan Peninsula (including mainland and insular Greece, Bulgaria, and Albania), as well as Cyprus [ref. 1].
Different preparations from various Lamiaceae plants, including those of the genus Stachys, have been used in traditional medicine worldwide to treat infections, gastrointestinal disorders, inflammation, skin diseases, and respiratory conditions such as asthma [ref. 6, ref. 7]. Moreover, phytochemical studies have revealed that Stachys species contain a variety of bioactive compounds, including terpenoids, iridoids, phytosterols, and polyphenols (such as flavonoids), among others [ref. 8, ref. 9, ref. 10]. These compounds are associated with numerous pharmacological effects, including antioxidant, anti‐inflammatory, anti‐diabetic, antimicrobial, and cytotoxic activities [ref. 8, ref. 9].
Growing attention has been given to the antioxidant and anti‐inflammatory properties of Stachys species, particularly for their relevance in aging‐related processes [ref. 11, ref. 12]. Studies on Stachys extracts have shown protective effects against cellular damage, supporting their potential role in promoting healthy aging [ref. 12, ref. 13]. In continuation of our research on Stachys spp. [ref. 14, ref. 15, ref. 16, ref. 17], the present study aimed to investigate the biocompatibility of S. cretica subsp. cretica, avoiding any possible renal toxicity. Biocompatibility is considered very crucial for the design and development of a great variety of biomedical and pharmaceutical applications, such as drug delivery systems or cosmetics [ref. 18]. To evaluate cytotoxicity, several methods are feasible. In this study, normal human embryonic kidney cells (HEK293) were cultivated to be used for the estimation of cell viability (%), employing MTT (3‐[4,5‐dimethylthiazol‐2‐yl]‐2,5 diphenyl tetrazolium bromide) colorimetric assay and thus ensuring that S. cretica subsp. cretica extracts are biocompatible without any effect on cell proliferation or viability.
Results and Discussion
High‐Performance Liquid Chromatography‐Photodiode Array Detection Coupled With Mass Spectrometry and Nuclear Magnetic Resonance Analyses
The liquid chromatography‐mass spectrometry (LC‐MS) profiling of wild‐growing and cultivated S. cretica subsp. cretica samples (Table 1) revealed notable chemical diversity between the infusions and the methanol (MeOH) extracts, which can be attributed to the different extraction methods and solvents used [ref. 19]. Specifically, the MeOH extracts (wild plants SCWM; cultivated plants SCCM) were richer in chemical compounds, particularly phenylethanoid glycosides and flavonoids, compared to the infusion samples (wild plants SCWI; cultivated plants SCCI). Similarly, a phytochemical analysis of the aqueous and MeOH extracts of S. cretica subsp. anatolica Rech.f. showed distinct differences in their chemical profiles, with the MeOH extract displaying a notably richer composition than the aqueous extract [ref. 20]. This may be explained by the fact that MeOH, as a solvent, can extract less polar constituents more effectively than water. As LC‐MS is aimed mainly at the identification of compounds, quantitative comparison between wild and cultivated samples cannot be reliably made.
TABLE 1: Fragmentation data (in negative mode) for the compounds detected in infusions and methanol extracts of wild‐growing (SCWI; SCWM) and cultivated (SCCI; SCCM) plant samples of Stachys cretica subsp. cretica.
| No. | Rt (min) | UV (nm) | Negative mode, m/z (rel. intensity 100%) | Identification | Infusions | Methanol extracts | ||
|---|---|---|---|---|---|---|---|---|
| SCWI | SCCI | SCWM | SCCM | |||||
| 1 | 6.27 | 296, 325 | 191.1 [quinic acid‐H]‐, 353.1 [M‐H]‐ | Chlorogenic acid | + | + | + | + |
| 2 | 14.03 | 289, 331 | 137.1, 593.2 [M‐caffeoyl‐H]‐, 755.1 [M‐H]‐ | Lavandulifolioside | + | + | + | + |
| 3 | 14.85 | 289, 330 | 160.9 [caffeoyl group‐H]‐, 461.1 [M‐caffeoyl‐H]‐, 623.1 [M‐H]‐ | Acteoside | + | + | + | + |
| 4 | 15.99 | 252, 267, 346 | 285 [A‐H]‐, 447 [M‐H]‐ | Luteolin‐7‐O‐glucoside | + | — | + | + |
| 5 | 16.53 | 288, 327 | 137.1, 160.9 [caffeoyl group‐H]‐, 461.1 [M‐caffeoyl‐H]‐, 623.2 [M‐H]‐ | Isoacteoside | — | — | + | — |
| 6 | 17.48 | 251, 268, 336 | 299.1, 461.1, 623.1[M‐H]‐ | Methoxy apigenin‐dihexoside | — | — | + | — |
| 7 | 18.48 | 286, 328 | 769 [M‐H]‐ | Alyssonoside | + | + | + | + |
| 8 | 20.56 | 295, 331 | 637 [M‐H]‐ | Leucosceptoside Α | + | + | + | + |
| 9 | 23.00 | 252, 267, 348 | 299, 461 | Chrysoeriol‐hexoside | + | — | — | — |
| 10 | 21.26 | 268, 330 | 268.1 [A‐H, homolytic]‐, 431.1 [M‐H]‐ | Apigenin‐glucoside | — | — | + | — |
| 11 | 28.68 | 252, 267, 341 | 299, 665 | Chrysoeriol‐acetyl‐dihexoside | — | — | + | + |
| 12 | 29.37 | 275, 307, 314 | 285, [A‐H]‐, 651.1 [M‐H]‐ | Isoscutellarein‐acetyl‐dihexoside | — | — | + | + |
| 13 | 29.40 | 286, 333 | 175.1, 783.3 | Stachysoside C, tentatively or Leucosceptoside B | — | — | + | — |
| 14 | 29.84 | 253, 276, 300, 339 | 315, [A‐H]‐, 458.9 [M‐hexose‐acetyl‐H2O‐H]‐, 681.1 [M‐H]‐ | 3′‐Methylether‐hypolaetin‐acetyl‐dihexoside | — | — | + | + |
| 15 | 30.52 | 253, 278, 302, 333 | 315, [A‐H]‐, 681.1 [M‐H]‐ | 3′‐Methylether‐hypolaetin‐acetyl‐dihexoside‐ isomer | — | — | — | + |
| 16 | 31.07 | 288, 322 | 651.1 [M‐H]‐ | Martynoside | — | — | + | + |
| 17 | 39.31 | 269, 318 | 269, [A‐H]‐, 577.1 [M‐H]‐ | Apigenin‐7‐coumaroyl‐hexoside | — | — | + | + |
| 18 | 39.87 | 274, 315 | 269, [A‐H]‐, 577.1 [M‐H]‐ | Apigenin‐coumaroyl‐hexoside isomer | — | — | + | + |
| 19 | 40.55 | 285 ↑, 313 | 271, [A‐H]‐, 579.1 [M‐H]‐ | Naringenin‐coumaroyl‐hexoside | — | — | + | + |
| 20 | 40.98 | 284 ↑↑, 313 | 271, [A‐H]‐, 579.1 [M‐H]‐ | Naringenin‐coumaroyl‐hexoside isomer | — | — | + | + |
| 21 | 42.35 | 297, 307 | 342, 462, 582 | Acylated spermine | — | — | + | + |
| 22 | 43.04 | 270, 316 | 269, [A‐H]‐, 577.1 [M‐H]‐ | Apigenin‐coumaroyl‐hexoside isomer | — | — | + | + |
| 23 | 51.74 | 270, 315 | 269, [A‐H]‐, 593 [M‐coumaroyl residue‐H]‐, 723.1 [M‐H]‐ | Apigenin‐dicoumaroyl‐hexoside isomer | — | — | + | + |
The identified compounds are consistent with the phytochemical fingerprints typically reported for the genus Stachys [ref. 16]. In total, 23 compounds were detected across the four samples, with chlorogenic acid, lavandulifolioside, acteoside, leucosceptoside A, and alyssonoside being present in all.
Among phenolic acids, chlorogenic acid (1) was identified. This compound has also been previously reported in S. recta [ref. 21], S. cretica subsp. smyrnaea Rech.f. [ref. 22], and S. iva Griseb. (currently known as S. horvaticii Micevski) [ref. 14]. Notably, a comprehensive study by Karioti et al. [ref. 23] reported chlorogenic acid as the predominant phenolic compound across various Balkan Stachys species. Moreover, its extensive occurrence throughout the genus has been thoroughly documented [ref. 9]. Chlorogenic acid is a well‐studied compound with multiple pharmacological properties, including anti‐inflammatory, anti‐apoptotic, antihypertensive, antiviral, antitumor, antibacterial, and antioxidant activities [ref. 24]. Phenylethanoid glycosides are main compounds found within the genus Stachys, exhibiting notable chemotaxonomic relevance [ref. 8, ref. 9]. Among them, acteoside (3) has been identified in several Stachys species, including S. lanata Crantz [ref. 25], S. byzantina K. Koch [ref. 26], and S. germanica L. subsp. salviifolia (Zen.) Gams [ref. 27]. Acteoside demonstrates a range of pharmacological activities, including strong anti‐inflammatory and antioxidant effects, neuroprotective properties, and the potential to inhibit cancer‐related pathways [ref. 28]. Additionally, leucosceptoside A (8) has been previously detected in other Greek native Stachys species such as S. tetragona Boiss. & Heldr. [ref. 29] and S. iva [ref. 14, ref. 17] and is reported to possess notable anti‐inflammatory and antioxidant activities [ref. 30]. Lavandulifolioside (2) was first isolated from S. lavandulifolia Vahl [ref. 31] and has been reported in some other species, such as S. macrantha (C. Koch.) Stearn [ref. 32], S. riederi Cham. [ref. 33], and S. iva [ref. 14]. It has also been detected in traces (<1%) or small amounts in S. recta subsp. recta (synonym of S. nitens Janka), S. beckeana Dörfl. & Hayek, S. zepcensis Formánek, S. alpina L. subsp. dinarica Murb., and S. plumosa Griseb. or in relatively higher amounts (3.97%) in S. atherocalyx K. Koch [ref. 23]. Lavandulifolioside has been reported to possess cardiovascular activity and notable anti‑arrhythmic effects by modulating cardiac electrophysiology [ref. 34].
The genus Stachys constitutes a rich source of flavonoids [ref. 9]. Flavones, mainly their 7‐O‐acetylallosylglucosides and 7‐O‐glucosides, have been widely found within the subgenus Stachys [ref. 8, ref. 9]. Among these, some derivatives of apigenin and chrysoeriol have previously been detected in wild‐growing Stachys taxa from Greece, such as apigenin and chrysoeriol 7‐O‐β‐D‐glucosides, as well as acylated forms such as 7‐(6″‐E‐p‐coumaroyl)‐D‐glucopyranosides and 7‐(3″‐E‐p‐coumaroyl)‐D‐glucopyranosides. In the case of apigenin, the corresponding Z‐isomers have also been identified to date. Additionally, 7‐O‐[6″‐O‐acetyl‐allosyl]‐(1→2)‐glucosides of both apigenin and chrysoeriol have been documented, as well as chrysoeriol 7‐O‐[6‴‐O‐acetyl‐allosyl]‐(1→2)‐glucoside [ref. 9]. In the present study, flavone derivatives were detected exclusively in MeOH extracts, with a similar flavonoid content observed in both the wild and cultivated samples. For example, luteolin‐7‐O‐glucoside (4), chrysoeriol‐hexosides (9), and isoscutellarein‐acetyl‐dihexoside (12) were identified in both MeOH samples, confirming their occurrence in members of this genus [ref. 9]. Notably, apigenin‐7‐glucoside (10) was detected only in the MeOH extract from the wild‐growing S. cretica subsp. cretica. Moreover, apigenin and naringenin p‐coumaroyl‐hexosides (17 and 19), along with their isomers (18 and 20), were also detected in both MeOH extracts of wild‐growing and the cultivated plant samples, aligning with previous reports on acylated flavone glucosides in Stachys spp. [ref. 9]. It is noteworthy that this is the first report of acylated spermine (21) in members of the genus Stachys; it is a polyamine, which activates defense responses in plants against both biotic and abiotic factors [ref. 35].
Following LC‐MS analysis, proton nuclear magnetic resonance (1H‐NMR) screening of the Stachys samples was carried out, and the chemical categories of their components were identified based on peaks in specific regions. The overlaid 1H‐NMR spectra of the infusions and MeOH extracts (Figure 1) revealed variations in their chemical profiles, reflecting differences in metabolite composition among the samples, which could be attributed to the different extraction solvents and different methods of preparation [ref. 36].
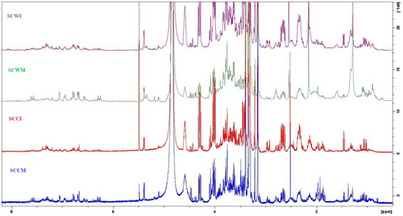
All spectra generally displayed signals in the aliphatic (δ 0.5–3.0 ppm), sugar and oxygenated methylene (δ 3.0–5.5 ppm), and aromatic (δ 6.0–8.1 ppm) regions, indicative of the presence of constituents which could belong to terpenoids, flavonoids, phenolic compounds, and carbohydrates. Notably, the MeOH extracts displayed richer chemical profiles than the infusions, particularly in terms of flavonoids and phenolic acid derivatives (especially phenylethanoid glycosides), which is consistent with the LC‐MS results. Flavonoids and phelylethanoid glycosides are widely found in Stachys spp. [ref. 8, ref. 9].
Regarding the comparison of 1H‐NMR spectra between cultivated and wild‐growing samples, the infusion from the cultivated sample (SCCI) exhibited a similar profile to that of the wild counterpart (SCWI). Likewise, the MeOH extract from the cultivated sample (SCCM) was close to the profile of the wild MeOH extract (SCWM).
Based on prior LC‐MS analysis indicating that the MeOH sample of wild‐growing material (SCWM sample) contained slightly more constituents, this extract was selected for further NMR investigation. Its 1H‐NMR spectrum was compared to those of previously isolated compounds reported in our own earlier studies on Stachys spp. [ref. 14, ref. 17]. Initially, chlorogenic acid was used to determine whether its signals could be identified in the 1H‐NMR spectrum of the SCWM sample, and its characteristic signals could be observed (Figure 2). This compound has been found in many Stachys spp. [ref. 16, ref. 22, ref. 23, ref. 37].
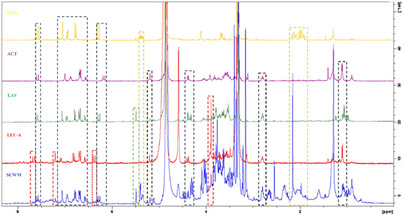
As a next step, to interpret the signals corresponding to phenylethanoid glycosides, we compared the spectra with those of compounds that have been commonly found in Stachys spp. [ref. 8, ref. 9] and have been previously isolated, namely acteoside, lavandulifolioside, and leucosceptoside A. The presence of these compounds was also supported by the afore‐mentioned LC‐MS results. The overlaid 1H‐NMR spectra of the SCWM extract and the reference compounds (chlorogenic acid, acteoside, lavandulifolioside, and leucosceptoside A) are shown in Figure 2, where key signals attributed to each compound are highlighted with differently coloured signals in the MeOH extract. It should be noted that alyssonoside was not included among the reference compounds; therefore, its presence could not be determined in the NMR analysis performed.
In addition, the 1H‐NMR spectrum of the SCWM was examined for the presence of flavonoids. Considering that isoscutellarein and hypolaetin derivatives are the main constituents in several Stachys taxa [ref. 8, ref. 9], a comparison was made with previously isolated reference compounds. However, it was not feasible to fully identify their derivatives due to signal overlapping and their low concentration.
Cell Viability
Numerous studies have highlighted the broad spectrum of pharmacological properties exhibited by several members of the genus Stachys, with particular emphasis on their antioxidant and anti‐inflammatory activities [ref. 12]. These bioactive effects are considered fundamental mechanisms through which Stachys spp. may exert anti‐aging benefits [ref. 11]. The members of this genus are notably rich in secondary metabolites, including polyphenols, terpenoids, flavonoids, iridoids, and phenolic acids, which play pivotal roles in mitigating oxidative stress, a key contributor to cellular aging and degenerative diseases [ref. 9, ref. 38]. Previous findings have demonstrated significant antioxidant capacity in both extracts and essential oils derived from various Stachys species, underscoring their potential in protecting biomolecular structures against oxidative damage associated with aging processes [ref. 12, ref. 13]. Moreover, chronic low‐grade inflammation, recognized as a central feature of aging and a precursor to numerous age‐related pathologies, may be attenuated by the anti‐inflammatory properties of Stachys members [ref. 12, ref. 13]. The anti‐inflammatory effects are primarily attributed to the modulation of pro‐inflammatory pathways by bioactive constituents such as flavonoids and iridoids, which have been shown to downregulate mediators of inflammation, thereby contributing to the prevention or delay of age‐associated disorders [ref. 9, ref. 13]. All these desirable properties are related to the general biocompatibility of Stachys members [ref. 39]. It is of crucial importance to investigate any possible toxicity of a plant material before starting to develop a system or a drug that is going to be used for biomedical or even research purposes [ref. 40]. For example, possible toxicity to the kidneys, being organs that produce urine for excretion, should be avoided in every attempt to design a successful pharmaceutical approach [ref. 41]. Actually, biocompatibility of several materials with HEK293 cells is commonly assessed to determine their suitability for biomedical applications, and in tissue engineering and drug delivery systems development [ref. 42]. HEK293 cells are considered an in vitro model since they present characteristics like those of epithelial cells, and due to their high transfection efficiency [ref. 43]. The cells’ morphology, viability, and proliferation rate are employed in well‐established methods investigating the biological effects of either artificial or natural materials [ref. 43]. If any undesirable effect is detected on this type of cell in the presence of a factor, consequently, this means that this factor is not suitable for further investigation if its biocompatibility is the preferred characteristic depending on the application [ref. 44, ref. 45].
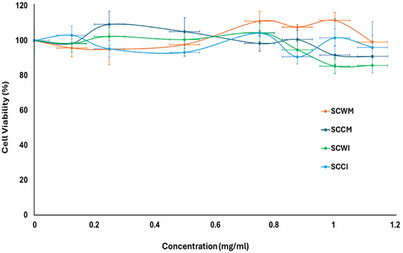
Thus, in the present study, S. cretica subsp. cretica MeOH extracts and infusions of wild and cultivated plant materials were employed in testing on HEK293 cells using the MTT colorimetric assay and cell viability (%) was evaluated compared to untreated cells. There was no significant effect (p‐value = 0.1564) of any of these four samples on the viability of HEK293 cells, as it is presented in Figure 3 (p < 0.05 was considered statistically significant), applying the Kruskal–Wallis non‐parametric test for statistical analysis since the criteria for parametric ANOVA were not met. The normality was checked based on the Shapiro‐Wilk Test (α = 0.05). When running the SW test, the p‐value is 0.1765. Thus, Kruskal–Wallis was selected. These findings indicated that the tested extracts and infusions are non‐toxic to kidney cells. Even for concentrations over 0.8 mg/mL, HEK293 cell viability remains very high (>90%), so the samples can be considered biocompatible in this type of cells, ensuring that their use is safe in this range of concentrations. Those concentrations are considered extremely high, compared to conventional therapeutic doses that are typically a few µg/mL or ng/mL, so the fact that biocompatibility was indicated on kidney cells even in extremely higher doses is a very promising result. There is no significant difference compared to untreated cells (negative control), considering p < 0.05 versus negative control, based on the Kruskal‐Wallis non‐parametric test. These results showed that MeOH extracts and infusions of both wild and cultivated material of S. cretica subsp. cretica could be a promising candidate for further analysis aimed at biomedical applications.
Conclusions
This study provided a comprehensive chemical characterization of wild‐growing and cultivated plant material of S. cretica subsp. cretica originating from or growing in the same region (Crete), examining in the same period (spring) and in a comparative fashion, their MeOH extracts and infusions using HPLC‐PDA‐MS and NMR analyses. Methanol extracts were significantly more abundant in flavonoids and phenylethanoid glycosides compared to aqueous infusions, as demonstrated by LC‐MS analysis. The phenylethanoid glycosides, namely acteoside, lavandulifolioside, leucosceptoside A, and chlorogenic acid, in the MeOH extract of wild‐growing S. cretica subsp. cretica were also confirmed by the NMR analysis. The differences between wild‐growing and cultivated samples were of minor importance, thus suggesting that cultivation in the same region of origin may retain the authentic phytochemical species profile. Also, to avoid any toxicity of the samples, the MTT colorimetric assay was employed to investigate their effect on the viability of human embryonic kidney cells (HEK293) in vitro. The results obtained indicated that both the MeOH extracts and infusions from wild (SCWM and SCWI) and cultivated (SCCM, SCCI) plant materials of S. cretica subsp. cretica are biocompatible (cell viability), thus suitable for biomedical and pharmaceutical applications. Ongoing studies from our research group have shown that the extracts and infusions of S. cretica subsp. cretica are also non‐toxic to skin fibroblasts. Further thorough studies will shed light on the possible biocompatibility to other cell types.
Experimental
Plant Materials
The wild‐growing populations of S. cretica subsp. cretica in Crete (Figure 4) were accessed and marked for photography and research using the special collection permit issued by the Greek Ministry of Environment and Energy (YPEN/DPD/15539/845 of 24/2/2022, YPEN/DPD/38262 /2306 of 2/8/2023, and YPEN/DPD/80381/5557 of 9/8/2024). Cuttings were excised in early March 2022 from wild‐growing S. cretica subsp. cretica at Pateles (N 35.01796, E 25.41825, 435 m above sea level) of Ano Viannos, Crete (Greece) for ex situ propagation at the premises of the Institute of Breeding and Plant Genetic Resources (IPBGR), Hellenic Agricultural Organization Demeter (ELGO‐Dimitra) using 0.2% indole‐3‐butyric acid (IBA; Racidin, Fytorgan ABEE, Greece) and were placed in a bench‐top fog system with an average air temperature of 19–25°C and relative humidity at 70%–85% for 20 days to allow rooting. The rooted cuttings were then transplanted into 0.33 L pots containing common soil, peat, perlite, and soft stones at 1:3:1:2 in a non‐heated greenhouse over winter 2022–2023, and were consequently transplanted into bigger (2.5 L) pots with the same substrate for late winter transfer and consequent cultivation in Crete (Adele, Rethymno) in late February 2023, where they were watered only a couple of times for acclimatization (completely rain fed onward, supervised by the external collaborator Evangelos Papiomytoglou). The aerial parts of the cultivated sample of S. cretica subsp. cretica were collected in 20/4/2023 from Adele, Rethymno, for comparative phytochemical analysis with the wild‐growing samples collected in 21/4/2023 during full flowering in Pateles, Ano Viannos (same origin as the cultivated sample, Figure 4). The plant samples were taxonomically authenticated by Dr N. Krigas, and a voucher specimen was deposited in the herbarium of Balkan Botanic Garden of Kroussia under the International Plant Exchange Network accession number GR‐1‐BBGK‐23,022 (GR‐1‐IPBGR‐23,022‐SCW for the wild‐growing material and GR‐1‐IPBGR‐23,022‐SCC for the asexually propagated and cultivated material of the same genotype).

Plant Preparations
The dried aerial parts of the wild‐growing S. cretica subsp. cretica (94 g) and the cultivated material of S. cretica subsp. cretica (148 g) were finely ground and macerated sequentially with dichloromethane (DCM) and then with MeOH (3 × 24 h per solvent). The resulting mixtures were filtered, and the solvents were evaporated under reduced pressure at low temperature using a rotary evaporator to yield dry residues: 206 mg and 203.3 mg for DCM extracts, as well as 207.1 mg and 200.3 mg for MeOH extracts.
In addition, 4 g finely comminuted aerial parts from each plant sample were infused in 200 mL of boiling water for 5 min, separately. Since Stachys infusions have traditionally been prepared and consumed in Greece as a kind of ‘mountain tea’, a term commonly associated with various members of the genus Sideritis, the infusion preparation followed the guidelines outlined in the European Medicines Agency monograph on different species and subspecies of the genus Sideritis [ref. 46]. Then, the samples were filtered, and the solvents were evaporated under reduced pressure at low temperature using a rotary evaporator to yield 2.4 g and 2.5 g, respectively.
HPLC‐PDA‐MS and NMR Analyses
The HPLC‐PDA‐MS analysis was conducted using a Thermo Finnigan system (Palo Alto, CA, USA), which included an LC Pump Plus, an Autosampler, and a Surveyor PDA Plus Detector. The system was coupled to an ESI MSQ Plus mass spectrometer (Thermo Finnigan, San Jose, CA, USA), controlled by Xcalibur software (version 2.1). The mass spectrometer operated in both positive and negative ionization modes, scanning within an m/z range of 100 to 1000. The gas temperature was set at 350°C, with a nitrogen flow rate of 10 L/min and a capillary voltage of 3000 V. The cone voltage ranged between 60 and 110 V. Chromatographic separation was carried out on an SB‐Aq Zorbax RP‐C18 column (Agilent, Santa Clara, CA, USA) measuring 150 mm × 3.5 mm, with a particle size of 3.5 µm, maintained at 30°C. The mobile phase consisted of water adjusted to pH 2.8 with 0.05% (v/v) formic acid (eluent A) and acetonitrile (eluent B), at a flow rate of 0.4 mL/min. The gradient program was structured as follows: 0–7 min, 90%–85% A; 7–12 min, 85%–82% A; 12–25 min, 82% A; 25–27 min, 82%–75% A; 27–32 min, 75% A; 32–42 min, 75%–60% A; 42–49 min, 60% A; 49–53 min, 60%–90% A; and 53–60 min, 90% A. The sample injection volume was 5 µL. Ultraviolet‐visible spectra were recorded over a wavelength range of 220–600 nm, while chromatographic profiles were monitored at 280, 330, and 350 nm. The identification of the compounds was based on co‐elution with the isolated compounds.
NMR spectra were acquired in CD3OD (for infusions and MeOH extracts) using a Bruker DRX 400 spectrometer operating at 399.95 MHz for 1H‐NMR. Chemical shifts (δ) were reported in ppm and referenced to the solvent signal at 3.31 ppm for CD3OD. Αll samples were stored in activated desiccators containing P2O5, ensuring the complete removal of moisture before the NMR analysis.
Cell Culture
HEK293 from ATCC (Human embryonic kidney 293 cells; HEK293: HTB‐22TM) (LGC Standards GmbH, ATCC, Wesel, Germany) cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco BRL, Life Technologies, ThermoScientific, Paisley, UK), adding 10% fetal bovine serum (FBS) and 1% antibiotics (penicillin/streptomycin) (Gibco BRL, Life Technologies, Thermo Scientific, Paisley, UK) to the medium, and the cells were maintained at 37°C under 5% CO2 [ref. 47, ref. 48].
MTT Assay
The cytotoxicity of the S. cretica subsp. cretica extracts was estimated, applying MTT colorimetric assay (Thiazolyl Blue Tetrazolium Bromide M5655, Sigma‐Aldrich, Darmstadt, Germany). A spectrophotometer was utilized to quantify the cell viability by measuring the optical density of each of the tested samples. In total, 8000–10 000 cells/well were seeded in 96‐well plates and treated in increasing concentrations of each extract, ranging from 0 to 0.125 mg/mL. At 24 h post‐treatment, the medium was removed from each sample, and 10 µL of MTT solution (concentration 5 mg/mL in phosphate‐buffered saline (Gibco BRL, Life Technologies, ThermoScientific, Paisley, UK) was added. After 2 h of incubation, the supernatant was removed, and 100 µL of dimethyl sulfoxide was added to each well. The plates were left on a shaker for 30 min at room temperature, and right after, the optical density was measured at 570 nm (background normalization was achieved through one more measurement at 650 nm). Cell viability (%) was estimated compared to untreated cells. Applying the Kruskal–Wallis non‐parametric test for statistical analysis, p < 0.05 was considered statistically significant.
Author Contributions
Ekaterina‐Michaela Tomou: methodology, validation, formal analysis, investigation, writing – original draft preparation, writing – review and editing, and visualization. Maria Anagnostou: validation, investigation, writing – original draft preparation, writing – review and editing, and visualization. Francesco Cesare Battisti: investigation. Anastasia Karioti: HPLC‐PDA‐MS analysis. Nikos Krigas: plant materials and writing – review and editing. Nefeli Lagopati: conceptualization, methodology, formal analysis, resources, writing – original draft preparation, writing – review and editing, visualization, and supervision. Helen Skaltsa: conceptualization, methodology, formal analysis, resources, writing – review and editing, and supervision. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This research received no external funding.
References
- A New Subspecies of Stachys cretica (section Eriostomum, Lamiaceae) From Turkey,”. Turkish Journal of Botany, 2010. [DOI]
- Taxonomical Notes on Stachys sect. Eriostomum (Lamiaceae) in Turkey,”. Turkish Journal of Botany, 2012. [DOI]
- Nomenclatural and Taxonomic Notes on Some Stachys taxa (Lamiaceae),”. Turkish Journal of Botany, 2021. [DOI]
- Vascular Plants of Greece: An Annotated Checklist,”. Englera, 2013. [DOI]
- Taxonomic Studies in Stachys II: A New Infrageneric Classification of Stachys L.,”. Royal Botanic Garden Edinburgh, 1980. [DOI]
- Ethnobotanical Use of Stachys L. (Lamiaceae) Taxa in Turkey,”. International Journal of Nature and Life Sciences, 2020. [DOI]
- The Health Benefits of Three Hedgenettle Herbal Teas (Stachys byzantina, Stachys inflata, and Stachys lavandulifolia)—Profiling Phenolic and Antioxidant Activities,”. European Journal of Integrative Medicine, 2020. [DOI]
- Phytochemical and Biological Studies of Stachys species in Relation to Chemotaxonomy: A Review,”. Phytochemistry, 2014. [DOI | PubMed]
- Genus Stachys: A Review of Traditional Uses, Phytochemistry and Bioactivity,”. Medicines, 2020. [DOI | PubMed]
- Stachys Species: Comparative Evaluation of Phenolic Profile and Antimicrobial and Antioxidant Potential,”. Antibiotics, 2023. [DOI | PubMed]
- The Free Radical Theory of Aging Matures,”. Physiological Reviews, 1998. [DOI | PubMed]
- Genus Stachys—Phytochemistry, Traditional Medicinal Uses, and Future Perspectives,”. Molecules, 2024. [DOI | PubMed]
- Pharmaceutical and Biological Properties of Stachys Species: A Review,”. Brazilian Journal of Pharmaceutical Sciences, 2022. [DOI]
- Valorisation of Stachysetin From Cultivated Stachys iva Griseb. as Anti‐diabetic Agent: A Multi‐spectroscopic and Molecular Docking Approach,”. Journal of Biomolecular Structure & Dynamics, 2021. [DOI | PubMed]
- Phytochemical Study of Stachys candida Bory & Chaubard (Lamiaceae),”. Biochemical Systematics and Ecology, 2021. [DOI]
- Metabolic Characterization of Four Members of the Genus Stachys L. (Lamiaceae),”. Agronomy, 2023. [DOI]
- Phytochemical Study of Stachys iva Griseb. and In Vitro Evaluation of AhR Transcriptional Activity,”. Chemistry and Biodiversity, 2024. [DOI | PubMed]
- Biomaterials for Drug Delivery and Human Applications,”. Materials, 2024. [DOI | PubMed]
- Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties,”. Molecules, 2010. [DOI | PubMed]
- LC‐MS/MS Profiles and in Vitro Biological Activities of Extracts of an Endemic Species From Turkey: Stachys cretica ssp. anatolica ,”. Plants, 2021. [DOI | PubMed]
- Analysis of the Constituents of Aqueous Preparations of Stachys recta by HPLC–DAD and HPLC–ESI‐MS,”. Journal of Pharmaceutical and Biomedical Analysis, 2010. [DOI | PubMed]
- Phenolic Ingredients and Therapeutic Potential of Stachys cretica subsp. smyrnaea for the Management of Oxidative Stress, Alzheimer’s Disease, Hyperglycemia, and Melasma,”. Industrial Crops and Products, 2019. [DOI]
- Chemical Profiling of Six Stachys Taxa From Balkan Peninsula,”. Biochemical Systematics and Ecology, 2022. [DOI]
- Chlorogenic Acid: A Review on Its Mechanisms of Anti‐Inflammation, Disease Treatment, and Related Delivery Systems,”. Frontiers in Pharmacology, 2023. [DOI | PubMed]
- Iridoid Glycoside Constituents of Stachys lanata ,”. Journal of Natural Products, 2008. [DOI | PubMed]
- Chemical Composition, Free‐Radical‐Scavenging and Insecticidal Activities of the Aerial Parts of Stachys byzantina ,”. Arch Biol Sci, 2010. [DOI]
- Phytochemical composition of polar fraction of Stachys germanica L. subsp. salviifolia (Ten.) Gams, a typical plant of Majella National Park,”. Natural Product Research, 2013. [DOI | PubMed]
- Unraveling the Dual Anti‐Inflammatory and Antioxidant Mechanisms of Acteoside: Computational Insights and Experimental Validation,”. Chemistry and Biodiversity, 2025. [DOI | PubMed]
- Secondary Metabolites From Stachys tetragona Boiss. & Heldr. ex Boiss. and Their Chemotaxonomic Significance,”. Biochemical Systematics and Ecology, 2018. [DOI]
- Leucosceptosides A and B: Two Phenyl‐Ethanoid Glycosides With Important Occurrence and Biological Activities,”. Biomolecules, 2022. [DOI | PubMed]
- Lavandulifolioside: A New Phenylpropanoid Glycoside From Stachys lavandulifolia ,”. Helvetica Chimica Acta, 1988. [DOI]
- Iridoid and Phenylpropanoid Glycosides From Stachys macrantha ,”. Phytochemistry, 1992. [DOI]
- Phenylethanoid Glycosides From Stachys riederi ,”. Nature Medicine, 1994
- The Antiarrhythmic Effects of Lavandulifolioside and Ferulic Acid From Leonurus cardiaca Extracts on Cardiac Electrophysiology,”. Planta Medica, 2013. [DOI]
- Spermine Differentially Refines Plant Defense Responses Against Biotic and Abiotic Stresses,”. Frontiers in Plant Science, 2019. [DOI | PubMed]
- Ultrasound‐Assisted Extraction of Stachys chasmosericea: Phytochemical Profile, Antioxidant Potentials via RACI Analysis, and Enzyme Inhibitory Activities,”. Preparative Biochemistry & Biotechnology (, 2025. [DOI]
- Antioxidant Potential, Enzyme Inhibition Activity, and Phenolic Profile of Extracts From Stachys cretica subsp. vacillans ,”. Industrial Crops and Products, 2019. [DOI]
- Anthelminthic and Antimicrobial Effects of Hedge Woundwort (Stachys sylvatica L.) Growing in Southern Kazakhstan,”. Frontiers in Pharmacology, 2024. [DOI | PubMed]
- Phytochemical Analysis and Biological Activity of Three Stachys Species (Lamiaceae) From Romania,”. Plants, 2021. [DOI | PubMed]
- The Evolving Role of Investigative Toxicology in the Pharmaceutical Industry,”. Nature Reviews Drug Discovery, 2023. [DOI | PubMed]
- Assessment of the Toxicity of Biocompatible Materials Supporting Bone Regeneration: Impact of the Type of Assay and Used Controls,”. Toxics, 2022. [DOI | PubMed]
- Biomaterials for Tissue Engineering Applications and Current Updates in the Field: A Comprehensive Review,”. AAPS PharmSciTech, 2022. [DOI | PubMed]
- Affecting HEK293 Cell Growth and Production Performance by Modifying the Expression of Specific Genes,”. Cells, 2021. [DOI | PubMed]
- Mechanism of Action and Cellular Responses of HEK293 Cells on Challenge With Zwitterionic Carbon Dots,”. Colloids and Surfaces B: Biointerfaces, 2021. [DOI | PubMed]
- Design of Double Functionalized Carbon Nanotube for Amphotericin B and Genetic Material Delivery,”. Scientific Reports, 2022. [DOI | PubMed]
- European Union Herbal Monograph on Sideritis scardica Griseb., Sideritis clandestina (Bory & Chaub.) Hayek, Sideritis raeseri Boiss. & Heldr., Sideritis syriaca L., Herba. (. 2016
- Biological Effect of Silver‐Modified Nanostructured Titanium Dioxide in Cancer,”. Cancer Genomics and Proteomics, 2021. [DOI | PubMed]
- Pulsed Electromagnetic Fields (PEMFs) Trigger Cell Death and Senescence in Cancer Cells,”. International Journal of Molecular Sciences, 2024. [DOI | PubMed]
