Antioxidant Activity and Potential Cholesterol Modulating Effect of Punica granatum L. Peel Hydroethanolic Extract
Abstract
Lipoprotein metabolism is regulated by several key proteins, notably proprotein convertase subtilisin/kexin type 9 (PCSK9) and the low‐density lipoprotein receptor (LDLR). Punica granatum peel extract has been reported to exhibit lipid‐lowering properties; however, the underlying mechanisms of its action on lipid metabolism remain insufficiently characterized. In this study, the hydroethanolic extract of P. granatum peel (PPE) was found to be rich in phenolic compounds and exhibited strong antioxidant activity. Characterization by liquid chromatography coupled with diode array and multiple‐stage mass spectrometry (LC‐DAD‐MSⁿ) identified punicalin, pedunculagin I, and galloyl‐HHDP‐hexose as the main constituents. Similar to statin treatment, PPE exposure significantly upregulated PCSK9 and LDLR protein expression in cultured cells compared to untreated controls, though the effect was less pronounced than that observed with statins. These findings suggest that phenolic‐rich PPE may serve as a promising natural source for the development of therapeutic agents aimed at preventing cardiovascular diseases.
Article type: Research Article
Keywords: low‐density lipoprotein (LDL) receptor, oxidation, phenolic compounds, PCSK9, pomegranate peels
Affiliations: LR12ES05, “Nutrition‐Functional Foods & Health” (NAFS), Faculty of Medicine University of Monastir Monastir Tunisia; Higher School of Health Sciences and Technologies University of Sousse Sousse Tunisia; Department of Medicine University of Padova Padova Italy; Faculty of Pharmacy University of Monastir Monastir Tunisia; Department of Pharmaceutical and Pharmacological Sciences University of Padova Padova Italy
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202501668 | PubMed: 41205209 | PMC: PMC12761359
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (687 KB)
Introduction
Punica granatum L., commonly known as pomegranate and belonging to the Lythraceae family, is among the oldest fruit trees cultivated by humans. Pomegranate is a species native to Western Asia, but it has been cultivated and naturalized since ancient times in the regions of the Mediterranean basin. Pomegranate is an ancient and a popular fruit in Tunisia. The pomegranate tree is cultivated throughout the country, except in high‐altitude areas. Because of the excellent flavor, nutritional value, and medicinal properties of pomegranate, its cultivation has extended from a family character, with plantations in allotments, gardens, and/or scattered plantations, to public orchards, ensuring fruit distinction on a national scale. Pomegranate is a large round berry with a hard‐leathery bark, containing numerous arils in compartments delimited by thick partitions. Only the arils constitute the edible part of the pomegranate, representing approximately half of the fruit. The fruit is consumed fresh, and it is also used for the manufacture of juices, jellies, and jam. The by‐products, such as the fruit peels and seeds, are traditionally used in folk medicine and dyeing. They constitute a source of various bioactive molecules, such as phenolic compounds and flavonoids [ref. 1]. Indeed, the peels have potentially beneficial effects in diabetes, Alzheimer’s disease, cancer, metabolic syndromes, and cardiovascular diseases (CVDs) [ref. 2, ref. 3, ref. 4, ref. 5].
CVDs are the major cause of death in the world. They are the result of a disturbance in many mechanisms, such as inflammation, oxidation, lipid metabolism, hypertriglyceridemia, high levels of low‐density lipoprotein‐cholesterol (LDL‐C), and decreased levels of high‐density lipoprotein‐cholesterol (HDL‐C), that are usually associated with a high risk of CVDs. Currently, there are many therapeutic options for preventing CVDs, including the inhibitors of cholesterol synthesis (statins) or absorption (ezetimibe), the peroxisome proliferator activated receptors (PPAR)‐alpha receptor agonists (fibrates), and monoclonal antibodies against proprotein convertase subtilisin/kexin type 9 (PCSK9) [ref. 6].
Although these drugs offer an effective therapeutic option for controlling hypercholesterolemia, natural products, thanks to their extensive pharmacological and biological activities, which include lipid‐lowering, antioxidant, anti‐inflammatory, anti‐thrombotic, and immunomodulatory effects [ref. 7, ref. 8], are considered valid alternatives for preventing CVDs, especially in primary prevention in patients at low risk. The phytoconstituents of natural products cover a diverse range of chemical entities, such as polyphenols, flavonoids, steroidal saponins, organosulfur compounds, vitamins, and polysaccharides [ref. 9, ref. 10, ref. 11]. Many studies were conducted, and they have reported that polyphenols are used to ameliorate dyslipidemia, especially to decrease LDL‐C level and oxidation [ref. 12]. Polyphenols act on cholesterol synthesis, metabolism, and uptake by inhibiting the hydroxy‐methyl glutaryl coenzyme A (HMG‐CoA) reductase, activating the cholesterol ester transfer protein (CETP), and upregulating low‐density lipoprotein receptor (LDLR) and downregulating PCSK9 expressions, respectively [ref. 12, ref. 13].
PCSK9, discovered 20 years ago [ref. 14], is expressed particularly in the liver, intestinal epithelium, kidney, and the neurons. It plays a crucial role in LDL‐C assimilation by the liver. In the absence of PCSK9, circulating LDL particles are recognized by the LDLR, and the LDL–LDLR complex is then internalized by endocytosis. In the endosomes, the LDLs are dissociated from the receptors that will be recycled to the surface of the cells [ref. 15]. In the presence of PCSK9, recycling of LDLR is disrupted, leading to an increased LDL in the plasma. Other studies have demonstrated that PCSK9 can act on the hepatic production of triglycerides and on the metabolism of HDL [ref. 16, ref. 17]. So, PCSK9 inhibitors are used to prevent or treat dyslipidemia. Many biotechnological strategies have been developed to reach this target [ref. 18]. Monoclonal antibodies and small interfering RNA (siRNA) are now available, and macrocyclic peptides against PCSK9 are under development [ref. 19]. However, many studies have focused on natural products [ref. 20, ref. 21, ref. 22, ref. 23, ref. 24] that can affect PCSK9 expression to identify new orally absorbed small molecules [ref. 13]. Polyphenols are among these natural products. It is for these reasons that foods rich in phenolic compounds, like P. granatum [ref. 1], could be of great benefit in preventing dyslipidemia and CVDs.
Many parts of pomegranate have been used, namely, the flowers, leaves, juice, and peels. Pomegranate peels have many beneficial health effects. Indeed, they are known for their anti‐inflammatory, antiapoptotic, and antioxidant effects [ref. 24]. These effects are the result of the pomegranate’s richness in phenolic compounds, mainly ellagitannins [25, 26]. Peel extracts are known also to have lipid‐lowering effects by decreasing total lipids, total cholesterol, and LDL‐C and increasing HDL‐C [ref. 27]. It is for this reason that Sadeghipour et al. suggested that further studies are needed to explore the effect of pomegranate peels in dyslipidemia [ref. 28]. According to our knowledge, the mechanisms by which pomegranate peels intervene in lipid metabolism are not yet well described.
The objective of the present study was to elucidate the effect of pomegranate peel extract (PPE) on the expression of PCSK9 and LDLR in hepatic carcinoma cells (Huh7) to better understand its influence on lipid metabolism. Additionally, the chemical composition (using spectrophotometric methods and LC‐DAD‐MSn) and antioxidant activities (radical scavenging and reducing power) of the extract were characterized to support its potential valorization.
Results and Discussion
Extract Characterization
Total phenolic compound and flavonoid concentrations in PPE are summarized in Table 1. The extract was found to be very rich in phenolic compounds (202.62 ± 4.75 mg gallic acid equivalent/g extract) and in flavonoids (79.52 ± 0.90). It also had a powerful antioxidant activity (Table 1). The antioxidant activity was evaluated by free radical scavenging assay and reducing power. The concentrations providing 50% of radical scavenging activity and reducing power inhibition were very low, demonstrating a high antioxidant activity for PPE. With regard to 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) essay, EC50 of PPE was only 0.304 mg/mL. EC50 obtained from reducing power essay was 0.385 mg/mL.
TABLE 1: Total phenolic compounds, total flavonoids, and EC50 (mg/mL) values obtained from 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) antioxidant assay and reducing power of Punica granatum peel extract.
| Pomegranate peel extract | |
|---|---|
| Total phenolic compounds (mg gallic acid equivalent/g extract) | 202.62 ± 4.75 |
| Total flavonoids (mg catechin equivalent/g extract) | 79.52 ± 0.90 |
| EC50 (mg/mL) DPPH | 0.304 ± 0.008 |
| EC50 (mg/mL) reducing power | 0.385 ± 0.039 |
Phenolic compounds have been largely employed to enhance the treatment of CVDs [ref. 29]. Peel extracts derived from pomegranate fruits are notably abundant in both total phenolic compounds and flavonoids [ref. 1]. However, compared with many studies, our extract exhibited a higher proportion of flavonoids, which may be attributed to the extraction solvent employed. Methanol, aqueous [ref. 26], and hydro‐methanol [ref. 30] peel extract showed less flavonoid proportion than hydro‐ethanolic extract [ref. 31]. In the present study, ethanol/water extraction was used due to the higher concentration of these compounds in this phase. Javani‐Seraji et al. showed that the ethanolic extract (50%) exhibits the greatest total phenolic content compared to methanol, acetone, and water extracts [ref. 32].
In the present study, PPE had high phenolic compounds and flavonoid concentrations. It also had a high antioxidant activity (measured by DPPH free radical scavenging activity and reducing power essay methods). Indeed, the antioxidant activity and the phenolic content are usually strongly correlated [ref. 33]. DPPH free radical scavenging activity informs us about the presence of molecules that neutralize free radicals (DPPH). Reducing power essay gives us an idea about the presence of reductones that break the free radical chain or the peroxide formation [ref. 34].
LC‐DAD‐MSn
The analysis revealed that the hydroethanolic extract presented several constituents that could be ascribed to pomegranate phytochemicals. For instance, the chromatogram recorded with diode array detector (DAD) at 254 nm presented several peaks with UV spectra resuming tannins correlated to punicalagin and ellagic acid derivatives and the presence of flavonoid. The MS fragmentation obtained in negative ion mode and the comparison with reference compounds allowed the identification of punicalagin, rutin, and ellagic acid that were also confirmed by standard injection. Furthermore, other constituents were tentatively identified through MS data. The LC‐DAD‐MSn analysis revealed a complex profile with a predominance of hydrolyzable tannins, including punicalin, punicalagin α and β, pedunculagin I, punigluconin, galloyl‐HHDP‐hexose, and granatin B, along with the flavonoid rutin and phenolic derivatives such as ellagic acid and its glycosylated forms (hexoside and deoxyhexoside) (Table 2). Punicalin (10.74 ± 0.91 mg/100 mg) was the most abundant constituent, followed by pedunculagin I (5.26 ± 0.23 mg/100 mg) and galloyl‐HHDP‐hexose (4.13 ± 0.43 mg/100 mg).
TABLE 2: HPLC‐DAD‐MSn identification and quantification in mg/100 mg of Punica granatum peel extract.
| RT (min) | Compound | M − H | Fragments | mg/100 mg |
|---|---|---|---|---|
| 1.4 | Punicalin | 781 | 601 721 299 271 | 10.74 ± 0.91 |
| 3.29 | Punicalagin αcbdv70644-tbl2-note-0001 | 1083 | 601 781 299 271 | 0.02 ± 0.01 |
| 4.1 | Punicalagin βcbdv70644-tbl2-note-0001 | 1083 | 601 781 299 271 | 2.25 ± 0.10 |
| 4.5 | Pedunculagin I | 783 | 481 301 | 5.26 ± 0.23 |
| 4.5 | Punigluconin | 799 | 479 273 313 417 | 1.33 ± 0.14 |
| 5.2 | Galloyl‐HHDP‐hexose | 633 | 301 461 229 257 185 | 4.13 ± 0.43 |
| 5.5 | Ellagic acid hexoside | 463 | 301 229 257 185 201 | 3.12 ± 0.23 |
| 6 | Granatin B | 951 | 897 445 613 729 401 229 | 1.17 ± 0.11 |
| 6.7 | Rutincbdv70644-tbl2-note-0001 | 609 | 301 271 | 0.30 ± 0.05 |
| 6.9 | Ellagic acidcbdv70644-tbl2-note-0001 | 301 | 229 257 185 201 | 0.09 ± 0.02 |
| 6.9 | Ellagic acid hexoside | 463 | 301 229 257 185 201 | 0.05 ± 0.01 |
| 7.6 | Ellagic acid deoxyhexoside | 447 | 301 229 257 185 201 | 2.38 ± 0.12 |
aCompared with reference standard.
Hydrolyzable tannins are polyphenolic substances derived from gallic acid (3,4,5‐trihydroxybenzoic acid), including gallotannins and ellagitannins. Similarly, other studies have shown that ellagitannin is dominant in pomegranate peels [ref. 25, ref. 35]. The most abundant type of ellagitannin is punicalagin [ref. 36]. However, our results showed that punicalin was the most abundant one. Punicalagin is the most powerful antioxidant compared to other phenols [ref. 37]. The extract contains another hydrolysable tannin. It is galloyl‐HHDP‐hexose, which belongs to the gallotannin group. Both ellagitannin and gallotannin have gallic acid as molecule precursor. Collectively, the observed quantitative pattern indicates a distinctive phenolic fingerprint, characterized by high punicalin content, moderate punicalagin levels, and substantial diversity in hydrolyzable tannins, potentially contributing to the extract’s strong antioxidant and bioactive potential.
Cell Viability
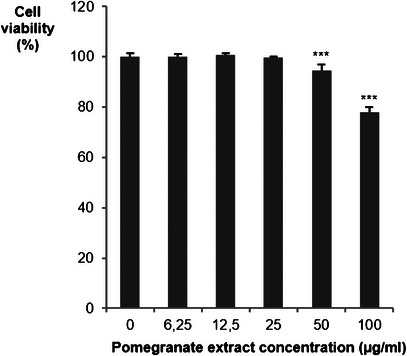
Before examining the effect of PPE on LDLR and PCSK9 gene expression, the maximum nontoxic concentration in Huh7 cells was determined. As shown in Figure 1, PPE begins to significantly reduce cell viability at 50 µg/mL, indicating a cytotoxic effect at higher doses. Therefore, subsequent experiments were performed at concentrations below this threshold (25 µg/mL) to avoid interference from cell death. PPE has demonstrated cytotoxic effects on hepatic carcinoma cells, notably the HepG2 cell line. The cytotoxicity is dose‐dependent. PPE has been shown to reduce cell viability, induce apoptosis, and cause cell cycle arrest in the G0/G1 and S phases [ref. 38].
Effect of PPE on PCSK9 and LDLR Expressions
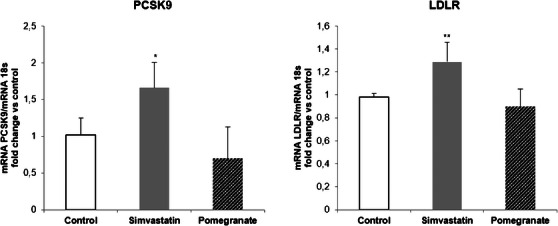
Huh7 cells were incubated with noncytotoxic concentrations of PPE (25 µg/mL) for 24 h. As positive control, simvastatin 20 µM (final concentration) was used. The results were compared to the untreated cells (control). The quantification of mRNA levels showed that both PCSK9 and LDLR expressions were induced by simvastatin, whereas PPE showed no effect (Figure 2).
Western blot analysis was used to assess the effect of simvastatin and PPE on protein expressions of LDLR and PCSK9. As expected, simvastatin significantly increased both PCSK9 and LDLR protein levels (Figure 3). A positive induction of both proteins was also observed after incubation with PPE.
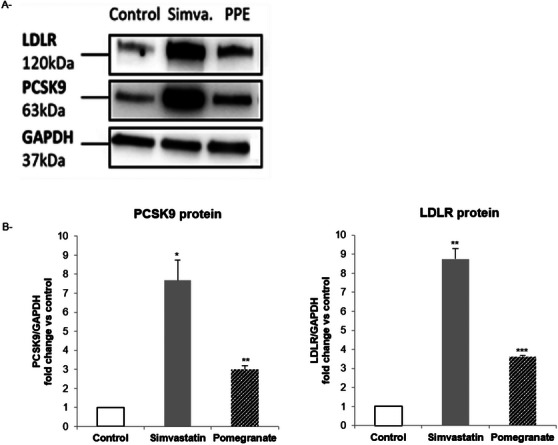
Phenolic compounds are known for their benefits in CVDs given their effects on several mechanisms, such as oxidation, signaling cascade, and endothelium dysfunction [ref. 39]. Moreover, polyphenols intervene by acting on lipid metabolism [ref. 40]. Many in vivo and in vitro investigations elucidated the pomegranate phenolic compounds effect on improving lipid profile [ref. 28, ref. 41]. In Huh7 cell, Khateeb et al. showed that pomegranate polyphenols upregulate the expression and activity of paraxonase1 and paraxonase2 that protect lipoproteins from oxidation [ref. 42]. This effect could also be due to an upregulation of ATP binding cassette subfamily A1 and liver X receptor α, which enhances cholesterol efflux in macrophages [ref. 43] or the activation of the PPARγ‐ABCA1/CYP7A1 cell signaling pathway in human hepatocyte cell line L‐02 [ref. 44]. In hamsters, ellagic acid promotes cholesterol removal by increasing fecal bile acid and upregulating the mRNA levels of the two pathways, LXR (/PPAR‐ABCA1) [ref. 45]. In western diet‐fed male mice, punicalagin modulates lipid intake and clearance in the liver [ref. 46]. However, few studies have explored the effect of pomegranate on PCSK9 and LDL receptors. Indeed, as PCSK9 and LDLR play a main role in cholesterol clearance, many studies have been conducted to explore the effect of different plant extracts on this pathway [ref. 22, ref. 23, ref. 47]. Dehghani et al. showed that PPE and punicalagin significantly increased LDLR protein levels and LDL cholesterol uptake and exhibited a dose‐dependent reduction in the expression of PCSK9 in HepG2 cells [ref. 48]. In our study, the incubation of Huh7 cells with statins was associated with increased concentrations of LDLR and PCSK9 mRNA. However, PPE has no significant effects on mRNA levels of these targets. In contrast, statins and PPE are associated with increased PCSK9 and LDLR protein levels. PCSK9 and LDLR have the same SRE (sterol regulatory element) motif that acts as promoter. Statins exert an upregulatory influence on both PCSK9 and LDLR expressions via sterol regulatory element binding proteins (SREBP). This effect is mediated by the ability of statins to enhance the binding of SREBP2 to SRE‐1, thus reducing plasma cholesterol levels [ref. 49]. Our results did not show any significant effect of PPE on mRNA of PCSK9 and LDLR genes. However, an important increase was demonstrated with regard to the protein level. The potential mechanism of action may involve either the activation of translation or the intracellular accumulation of both proteins (PCSK9 and LDLR). This can be explained by the fact that certain compounds in the extracts (flavonoids or polyphenols) may inhibit the proteasomal or lysosomal degradation of PCSK9 and LDLR by modulating the activity of proteolytic enzymes, thereby increasing their half‐life and accumulation. Additionally, some plant metabolites may directly bind to PCSK9 or LDLR, altering their conformation and protecting them from degradation. This can also affect their detection during analyses (e.g., improved antibody recognition in western blot). Furthermore, the extract may act on proteins that regulate the stability of PCSK9 or LDLR, such as IDOL (inducible degrader of the LDLR) or ubiquitin ligases, without altering the transcription of the PCSK9 and LDLR genes. Further studies are needed to better understand the mode of action of PPE. The upregulation of LDLR correlates with enhanced clearance of circulating LDL cholesterol, resulting in decreased plasma cholesterol levels and offering protection against CVDs. In contrast, increased PCSK9 expression promotes LDLR degradation and reduces receptor recycling, which elevates circulating LDL cholesterol levels. The potential protective role of elevated PCSK9 in CVD remains controversial. In the present study, the modest increase in PCSK9 suggests a homeostatic mechanism to prevent excessive LDL‐C clearance that could disrupt essential cellular functions. Moreover, emerging evidence indicates that PCSK9 possesses pleiotropic effects beyond LDLR regulation, including modulation of inflammatory pathways, vascular homeostasis, and immune responses [ref. 50]. Therefore, a moderate elevation of PCSK9 may contribute to cardiovascular protection by balancing these complex biological processes.
Conclusions
Due to their antioxidant properties and their effects on PCSK9 and LDLR, the phenolic‐rich extract of pomegranate (PPE) appears to represent valuable resources for the development of molecules to prevent CVDs. However, it is imperative that a comprehensive series of additional studies be conducted not only to identify and isolate the specific bioactive compounds within these extracts but also to gain a deeper understanding of the intricate mechanisms underlying their mode of action. Such investigations will contribute significantly to the advancement of knowledge regarding the potential therapeutic applications of PPE in the context of cardiovascular health.
Experimental Section
Extract Preparation and Characterization
Extract Preparation
Pomegranate fruits were harvested in December 2023 from the Sousse region (35°49′32″ N, 10°38′28″ E) in central Tunisia. They were identified as P. granatum L. by Professor Mohamed Chaib from the Faculty of science, Sfax University, Tunisia, and a voucher specimen (Voucher No. L02‐2025) was deposited in the herbarium at the Faculty of science, Sfax University, The fruits were peeled off by hand and the peels were freeze dried (lyophilized) and blended. The powdered peels were extracted by hydroethanolic solutions [ethanol/water (40/60, v/v)] at a solvent‐to‐peel powder ratio of 10:1 (w/v) during one night with stirring. Then, the mixture was filtered under vacuum and the filtrate was subjected to rotary evaporation at 40°C.
Total Phenolic Content
The dosage of total phenolic compounds was carried out using the colorimetric Folin–Ciocâlteu method according to Montedoro et al. [ref. 51], with slight modifications [ref. 1]. Gallic acid was used to prepare standard range and to determine the calibration curve equation. The results were expressed as mg gallic acid equivalents per g of extract.
Total Flavonoid Content
The dosage of flavonoids was estimated by the colorimetric method of aluminum trichloride (AlCl3) at 510 nm [ref. 52]. The results were expressed as mg catechin equivalent per g of extract by referring to a calibration curve produced with catechin.
In Vitro Antioxidant Activity
The antioxidant activity of the two extracts was evaluated using two colorimetric methods: DPPH free radical scavenging activity and the reduction of ferric iron by the FRAP method (ferric reducing antioxidant power).
DPPH‐Free Radical Scavenging Activity
DPPH, a free radical of violet color in solution, was reduced to 2,2 diphenyl 1 picryl hydrazine of yellow color by a free radical sensor, and it showed a characteristic absorbance at a wavelength of 517 nm. DPPH free radical scavenging activity was calculated by following DPPH absorbance in the absence and in the presence of the extract. A volume of 1 mL of the DPPH solution was added to different quantities of extract. The mixtures were shaken and kept in the dark for 30 min. Finally, the optical density was measured at a wavelength of 517 nm using a spectrophotometer, with reference to a control sample without extract. The measured absorbance was then converted into percentage inhibition relative to the absorbance of the control solution. The relationship between percentage inhibition and total phenolic content was not linear but logarithmic. The kinetic analysis of this activity allowed the determination of concentrations corresponding to 50% inhibition (IC50). The result was expressed as the concentration of the extract responsible for 50% of the radical scavenging activity (EC50) [ref. 53].
Reducing Power
Measurement of reducing power by iron reduction was based on the increase in optical absorbance at 700 nm. To 250 µL of the extract solution, 625 µL of phosphate buffer solution (200 mM; pH 6.6) and 625 µL of 1% potassium ferricyanide [K3Fe(CN)6] were added. After incubation for 20 min at 50°C, 625 µL of 10% trichloroacetic acid (TCA) was added to the mixture, followed by centrifugation at 3000 rpm for 10 min. To 625 µL of the supernatant, 625 µL of distilled water and 125 µL of 0.1% ferric chloride (FeCl3) were added. The absorbance was measured at 700 nm, and the results were expressed based on the increase in optical density, which corresponds to an increase in the reducing power of the extracts.
The reducing power was calculated as a percentage of reduction. Then, the graph of reducing power percentage according the extract concentration was drawn. The result was expressed as the extract concentration responsible of 50% of reducing power (EC50) [ref. 54].
Liquid Chromatography Diode Array Multiple Stage Mass Spectrometry, LC‐DAD‐MSn
A 20 mg of the extract was carefully weighed and dissolved in 10 mL of methanol. The solution was sonicated for 15 min, followed by centrifugation for 10 min, and the supernatant was collected for analysis.
The chemical characterization of the extracts was performed using an Agilent 1260 system, coupled with a 1260 DAD and a Varian MS 500 ion trap mass spectrometer. An SBC18 column (4.6 × 50 mm, 1.8 µm) was utilized for separation, with the mobile phases consisting of water (1% formic acid) (A), acetonitrile (B), and methanol (C). The elution gradient followed these conditions: 95:5:0% (A/B/C) at 0 min; 50:35:15% (A/B/C) at 10 min; 5:85:10% (A/B/C) at 16 min; 0:85:15% (A/B/C) from 17.0 to 17.5 min, followed by a 5‐min re‐equilibration phase. The flow rate was set to 0.7 mL/min, with an injection volume of 10 µL. Chromatograms were obtained using the DAD within the 200–600 nm range, with traces recorded at 254, 280, 330, and 590 nm. UV spectra were acquired to identify each peak. Mass spectra were acquired using a mass spectrometer with an electrospray ionization source (EIS) in negative ion mode, covering a mass range of 105–2000 m/z. The ion trap acquired data in TDDS mode, enabling multiple reactions monitoring with multistage fragmentation. This approach allowed for the identification of secondary metabolites through comparison with reference standards and published literature. The mass spectrometer parameters were as follows: needle voltage set to 4100 V, nebulizer gas pressure at 30 psi, drying gas temperature at 270°C, drying gas pressure at 25 psi, spray chamber temperature at 50°C, capillary voltage at 75 V, and RF loading at 70%.
Chlorogenic acid, rutin, ellagic acid, and mulberroside A were used for quantification. Calibration curves were generated using standard solutions within the range of 1–50 µg/mL.
In Vitro Experiment
Cell Culture and Treatment
Huh7 cell lines were purchased from Tebubio SRL, Milan, Italy (code product 300156). All cell culture maintaining reagents and all the plastic supplies were obtained from EuroClone (Milan, Italy). The extracts were diluted in dimethyl sulfoxide (DMSO, Sigma‐Aldrich). A stock solution (50 mM, pH 7.2) of simvastatin (Merck, Sharp, and Dohme Research Laboratories, Kenilworth, NJ, USA) was prepared. Culture medium was MEM supplemented with 10% fetal bovine serum (FBS), 1% sodium pyruvate 100×, 1% l‐glutamine 200 mM, 1% penicillin/streptomycin solution, and 1% nonessential amino acids 100×. Culture conditions were 37°C, 5% CO2, and 95% air.
For the experiments, human hepatic cancer cells (Huh7) were incubated in MEM/10% FBS with the final concentration of DMSO did not exceed 0.5% v/v.
PCSK9 and LDLR Expression
Cells were seeded at a density of 80 000 cells, using MEM supplemented with 10% FBS. After 24 h, treatments were added. After 24 h, the cells were rinsed twice with PBS. Then, the iScript RT‐qPCR sample prep reagent (Bio‐Rad, Milan, Italy) was used to extract total RNA. Specific primers for 18S (FWD 5′‐CGGCTACCACATCCACGGAA‐3′, REV 5′‐CCTGAATTGTTATTTTTCGTCACTACC‐3′), PCSK9 (FWD 5′‐CCTGCGCGTGCTCAACT‐3′, REV 5′‐GCTGGCTTTTCCGAATAAACTC‐3′), and LDLR (FWD 5′‐TCTATGGAAGAACTGGCGGC‐3′ REV 5′‐ACCATCTGTC TCGAGGGGTA‐3′) were used for qPCR. The analyses were carried out using the following program: 10 min at 45°C, 5 min at 95°C, and 40 cycles of 5 s at 95°C, followed by 30 s at 60°C. The data were presented as Ct values and utilized for relative quantification of targets using ΔΔCt calculations. The ΔΔCt values were calculated by multiplying the ratio of the efficiency of specific primers to that of the housekeeping gene 18S. The efficiency was determined using the formula ((10(−1/slope)) − 1) × 100 [ref. 55].
Western Blot Analysis
Anti‐PCSK9, anti‐LDLR, and anti‐GAPDH primary antibodies (rabbit polyclonal antibody, GeneTex) were used. Anti‐rabbit secondary antibody was obtained from Jackson Immuno Research.
Cells were plated at a density of 300 000 cells per well in MEM/10% FBS. After 24 h, treatments were added. Seventy‐two hours later, cells were subjected to two washes with PBS (Sigma‐Aldrich) and were homogenized in lysis buffer (1% NP‐40, 50 mM Tris–HCl and 150 mM NaCl, pH 7.5). BCA assays (SERVA) were used to assess protein concentration. Then, 25 µg of total protein extract per sample were loaded onto a 4%–20% SDS–PAGE gel (Bio‐Rad) and separated under denaturing and reducing conditions. Following electrophoresis, the proteins were transferred to a nitrocellulose membrane. Blocking was carried out with 5% nonfat dried milk in tris‐buffered saline containing 0.2% Tween 20 (TBST20). Nitrocellulose membrane with the primary antibodies was incubated overnight at 4°C in agitation. Membrane and horseradish peroxidase conjugated secondary antibodies were incubated at room temperature for 1.5 h under agitation. UVITEC Alliance Q9 Advanced—manual system was used to measure luminescence signals and ImageJ software was used for quantitative densitometric analysis.
Statistical Analysis
It was conducted with SPSS 21.0 for Windows (SPSS, Chicago, IL, USA). Data are expressed as mean ± standard deviation. Comparison between means was performed using Student’s t‐test. A p value <0.05 was considered statistically significant.
Author Contributions
Raja Chaaba: conceptualization, laboratory analysis, writing original draft, review and editing of the final manuscript. Amel Nakbi: conceptualization. Ilaria Rossi: laboratory analysis, review and editing of the final manuscript. Hanen Jrah: writing original draft. Zahra Amri: laboratory analysis. Nicola Ferri: supervision, review and editing of the final manuscript. Sonia Hammami: project administration. Stefano Dall’Acqua: supervision, review and editing of the final manuscript. Mohamed Hammami: supervision. Sounira Mehri: data analysis.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
The authors received no specific funding for this work.
References
- Phytochemical Content, Fatty Acids Composition and Antioxidant Potential of Different Pomegranate Parts: Comparison Between Edible and Non Edible Varieties Grown in Tunisia,”. International Journal of Biological Macromolecules, 2017. [DOI | PubMed]
- Assessing the Multitargeted Antidiabetic Potential of Three Pomegranate Peel‐Specific Metabolites: An In Silico and Pharmacokinetics Study,”. Food Science & Nutrition, 2023. [DOI | PubMed]
- Pomegranate: A Source of Multifunctional Bioactive Compounds Potentially Beneficial in Alzheimer’s Disease,”. Pharmaceuticals (Basel), 2023. [PubMed]
- An Insight Into Anticancer Bioactives From Punica granatum (Pomegranate),”. Anti‐Cancer Agents in Medicinal Chemistry, 2022. [DOI | PubMed]
- Pomegranate (Punica granatum L.) Peel Extract Ameliorates Metabolic Syndrome Risk Factors in Patients With Non‐Alcoholic Fatty Liver Disease: A Randomized Double‐Blind Clinical Trial,”. Nutrition Journal, 2023. [DOI | PubMed]
- Lipid‐Lowering Therapy and Cardiovascular Prevention in Elderly,”. Drugs, 2025. [PubMed]
- The Potential of Natural Products in the Management of Cardiovascular Disease,”. Current Pharmaceutical Design, 2024. [DOI | PubMed]
- Traditional Uses, Phytochemistry, Pharmacology and Toxicology of Ruta graveolens L.: A Critical Review and Future Perspectives,”. Drug Design, Development and Therapy, 2024. [DOI | PubMed]
- Bioactive Compounds (BACs): A Novel Approach to Treat and Prevent Cardiovascular Diseases,”. Current Problems in Cardiology, 2023. [DOI | PubMed]
- The Therapeutic Potential of Plant Polysaccharides in Metabolic Diseases,”. Pharmaceuticals, 2022. [DOI | PubMed]
- Metabolomics Reveals the Effects of Hydroxysafflor Yellow A on Neurogenesis and Axon Regeneration After Experimental Traumatic Brain Injury,”. Pharmaceutical Biology, 2023. [DOI | PubMed]
- Bioactivity of Dietary Polyphenols: The Role in LDL‐C Lowering,”. Foods, 2021. [DOI | PubMed]
- Naturally Occurring PCSK9 Inhibitors,”. Nutrients, 2020. [DOI | PubMed]
- The Secretory Proprotein Convertase Neural Apoptosis‐Regulated Convertase 1 (NARC‐1): Liver Regeneration and Neuronal Differentiation,”. PNAS, 2003. [DOI | PubMed]
- Targeting the Proprotein Convertase Subtilisin/Kexin Type 9 for the Treatment of Dyslipidemia and Atherosclerosis,”. Journal of the American College of Cardiology, 2013. [DOI | PubMed]
- Proprotein Convertase Subtilisin Kexin Type 9 and High‐Density Lipoprotein Metabolism: Experimental Animal Models and Clinical Evidence,”. Translational Research, 2016. [DOI | PubMed]
- Beyond LDL: What Role for PCSK9 in Triglyceride‐Rich Lipoprotein Metabolism?,”. Trends in Endocrinology and Metabolism, 2018. [DOI | PubMed]
- A Novel Small‐Molecule PCSK9 Inhibitor E28362 Ameliorates Hyperlipidemia and Atherosclerosis,”. Acta Pharmacologica Sinica, 2024. [DOI | PubMed]
- PCSK9 Inhibitors in the Management of Cardiovascular Risk: A Practical Guidance,”. Vascular Health and Risk Management, 2022. [DOI | PubMed]
- The Cholesterol‐Lowering Effect of Capsella Bursa‐Pastoris Is Mediated via SREBP2 and HNF‐1α‐Regulated PCSK9 Inhibition in Obese Mice and HepG2 Cells,”. Foods, 2021. [DOI | PubMed]
- Sesquiterpenoids From the Aerial Parts of Salvia plebeia With Inhibitory Activities on Proprotein Convertase Subtilisin/Kexin Type 9 Expression,”. Journal of Natural Products, 2021. [DOI | PubMed]
- The Modulation of PCSK9 and LDLR by Supercritical CO2 Extracts of Mentha longifolia and Isolated Piperitone Oxide, an In Vitro Study,”. Molecules (Basel, Switzerland), 2021. [DOI | PubMed]
- Chemical Constituents From Morus alba With Proprotein Convertase Subtilisin/Kexin Type 9 Expression and Secretion Inhibitory Activity,”. Organic & Biomolecular Chemistry, 2023. [DOI | PubMed]
- The Anti‐Inflammatory, Anti‐Apoptotic, and Antioxidant Effects of a Pomegranate‐Peel Extract Against Acrylamide‐Induced Hepatotoxicity in Rats,”. Life (Basel), 2022. [PubMed]
- Antioxidant Properties and Phenolic Profile Characterization by LC–MS/MS of Selected Tunisian Pomegranate Peels,”. Journal of Food Science and Technology, 2017. [DOI | PubMed]
- HPLC Evaluation of Phenolic Profile, Nutritive Content, and Antioxidant Capacity of Extracts Obtained From Punica granatum Fruit Peel,”. Advances in Pharmacological and Pharmaceutical Sciences, 2013
- Phenolic Profiles, Antihyperglycemic, Antihyperlipidemic, and Antioxidant Properties of Pomegranate (Punica granatum) Peel Extract,”. Journal of Food Biochemistry, 2019. [DOI | PubMed]
- Lipid Lowering Effect of Punica granatum L. Peel in High Lipid Diet Fed Male Rats,”. Evidence‐Based Complementary and Alternative Medicine, 2014. [DOI | PubMed]
- Phenolic Compounds Exerting Lipid‐Regulatory, Anti‐Inflammatory and Epigenetic Effects as Complementary Treatments in Cardiovascular Diseases,”. Biomolecules, 2020. [DOI | PubMed]
- Phenolic Profiles, Antihyperglycemic, Antihyperlipidemic, and Antioxidant Properties of Pomegranate (Punica granatum) Peel Extract,”. Journal of Food Biochemistry, 2019. [DOI | PubMed]
- Potential Assessment of Pomegranate (Punica granatum L.) Fruit Peels as a Source of Natural Antioxidants,”. Pharmacophore, 2018
- Influence of Extraction Techniques on the Efficiency of Pomegranate (Punica granatum L.) Peel Extracts in Oxidative Stability of Edible Oils,”. Food Science & Nutrition, 2023. [DOI | PubMed]
- Polyphenols, Antioxidant, Antibacterial, and Biofilm Inhibitory Activities of Peel and Pulp of Citrus medica L., Citrus bergamia, and Citrus medica Cv. Salo Cultivated in Southern Italy,”. Molecules (Basel, Switzerland), 2019. [DOI | PubMed]
- 34 M. Gordon , “The Mechanism of Antioxidant Action In Vitro,” in Food Antioxidants (Springer, 1990), 10.1007/978-94-009-0753-9.
- Profiling Phenolic Composition in Pomegranate Peel From Nine Selected Cultivars Using UHPLC‐QTOF‐MS and UPLC‐QQQ‐MS,”. Frontiers in Nutrition, 2021. [DOI | PubMed]
- Pomegranate Variety and Pomegranate Plant Part, Relevance From Bioactive Point of View: A Review,”. Bioresources and Bioprocessing, 2021. [PubMed]
- Identification and Quantification of Phenolic Compounds From Pomegranate (Punica granatum L.) Peel, Mesocarp, Aril and Differently Produced Juices by HPLC‐DAD–ESI/MSn,”. Food Chemistry, 2011. [DOI | PubMed]
- Pomegranate Seeds and Peel Ethanolic Extracts Anticancer Potentials and Related Genetic, Histological, Immunohistochemical, Apoptotic and Oxidative Stress Profiles: In Vitro Study,”. Journal of Experimental Pharmacology, 2023. [DOI | PubMed]
- Polyphenols, Inflammation, and Cardiovascular Disease,”. Current Atherosclerosis Reports, 2013. [DOI | PubMed]
- Polyphenols Effect on Circulating Lipids and Lipoproteins: From Biochemistry to Clinical Evidence,”. Current Pharmaceutical Design, 2018. [DOI | PubMed]
- Beneficial Effects of Pomegranate Peel Extract on Plasma Lipid Profile, Fatty Acids Levels and Blood Pressure in Patients With Diabetes Mellitus Type‐2: A Randomized, Double‐Blind, Placebo‐Controlled Study,”. Journal of Functional Foods, 2020
- Paraoxonase 1 (PON1) Expression in Hepatocytes Is Upregulated by Pomegranate Polyphenols: A Role for PPAR‐Gamma Pathway,”. Atherosclerosis, 2010. [DOI | PubMed]
- Pomegranate Peel Polyphenols Inhibit Lipid Accumulation and Enhance Cholesterol Efflux in raw264.7 Macrophages,”. Food and Function, 2016. [DOI | PubMed]
- Effects of Pomegranate Peel Polyphenols on Lipid Accumulation and Cholesterol Metabolic Transformation in L‐02 Human Hepatic Cells via the PPARgamma‐ABCA1/CYP7A1 Pathway,”. Food and Function, 2016. [DOI | PubMed]
- Effects of Ellagic Acid‐Rich Extract of Pomegranates Peel on Regulation of Cholesterol Metabolism and Its Molecular Mechanism in Hamsters,”. Food and Function, 2015. [DOI | PubMed]
- Punicalagin Prevents Hepatic Steatosis Through Improving Lipid Homeostasis and Inflammation in Liver and Adipose Tissue and Modulating Gut Microbiota in Western Diet‐Fed Mice,”. Molecular Nutrition & Food Research, 2021
- Bergamot (Citrus bergamia) Peel Extract as New Hypocholesterolemic Agent Modulating PCSK9 Expression,”. Journal of Functional Foods, 2023
- Exploring the Potential of Pomegranate (Punica granatum) Peel Extract and Punicalagin as Novel Anti‐Hypercholesterolemic Agents,”. Functional Foods in Health and Disease, 2024. [DOI]
- Hepatocyte Nuclear Factor 1alpha Plays a Critical Role in PCSK9 Gene Transcription and Regulation by the Natural Hypocholesterolemic Compound Berberine,”. Journal of Biological Chemistry, 2009. [DOI | PubMed]
- The Role of PCSK9 in Heart Failure and Other Cardiovascular Diseases—Mechanisms of Action Beyond Its Effect on LDL Cholesterol,”. Heart Failure Reviews, 2024. [DOI | PubMed]
- Simple and Hydrolyzable Phenolic Compounds in Virgin Olive Oil. 1. Their Extraction, Separation, and Quantitative and Semiquantitative Evaluation by HPLC,”. Journal of Agricultural and Food Chemistry, 1992. [DOI]
- The Determination of Flavonoid Contents in Mulberry and Their Scavenging Effects on Superoxide Radicals,”. Food Chemistry, 1999. [DOI]
- Antioxidant Principles From Bauhinia tarapotensis ,”. Journal of Natural Products, 2001. [DOI | PubMed]
- Studies on Products of Browning Reaction Antioxidative Activities of Products of Browning Reaction Prepared From Glucosamine,”. Japanese Journal of Nutrition and Dietetics, 1986. [DOI]
- Analysis of Relative Gene Expression Data Using Real‐Time Quantitative PCR and the 2(‐Delta Delta C(T)) Method,”. Methods (San Diego, Calif), 2001. [DOI | PubMed]
