Burning mouth syndrome: updates on pathogenesis and diagnostic algorithms
Abstract
Burning Mouth Syndrome (BMS) is a complex, chronic neuropathic orofacial pain disorder characterized by a persistent burning or dysesthetic sensation in the oral cavity without an identifiable organic cause. Accurate diagnosis and effective management of BMS pose significant challenges to clinicians, necessitating a comprehensive and multidisciplinary approach. This review delves into BMS’s pathogenesis and diagnostic algorithms, highlighting the latest advancements in understanding the underlying mechanisms and diagnostic strategies. Utilizing specific diagnostic algorithms assists clinicians in assessing and selecting appropriate treatment strategies, thereby minimizing diagnostic delays. These algorithms are crucial for excluding other causes of oral burning by focusing on symptomatology, patient history, and clinical examination. They involve ruling out oral infections, nutritional deficiencies, hormonal imbalances, autoimmune disorders, and medication side effects as potential causative factors. Supporting the diagnostic process, additional tests such as blood tests (including a thrombophilic panel and Neuron-Specific Enolase), neurosensory assessments, neuroradiological examinations, and evaluations of psychological profiles and cognitive function may be employed. Neurosensory assessments and neuroradiological examinations can provide insights into possible neuropathic causes, while psychological and cognitive assessments can identify any psychological factors and the extent to which cognitive decline may contribute to the condition. A comprehensive diagnostic approach not only aids in the accurate identification of BMS but also helps differentiate it from other oral conditions with similar presentations. This thorough evaluation is essential for developing a tailored treatment plan that addresses each patient’s specific needs, ultimately improving clinical outcomes and enhancing the quality of life for individuals suffering from BMS.
Article type: Review Article
Keywords: Burning mouth syndrome, Dysesthetic sensation, Xerostomia, Anxiety, Depression, White matter change
Affiliations: Department of Neurosciences, Reproductive Sciences and Odontostomatology, University of Naples “Federico II”, 80131 Naples, Italy; Department of Health Sciences, School of Dentistry, University Magna Graecia of Catanzaro, 88100 Catanzaro, Italy; Department of Medicine, Surgery and Health Sciences, University of Trieste, 34129 Trieste, Italy; Therapeutic Dentistry Department, Institute for Dentistry, Sechenov University, 119991 Moscow, Russia; Department of Life Science, Health and Health Professions, Link Campus University, 00165 Rome, Italy
License: Copyright© 2025 The Author(s). Published by MRE Press. CC BY 4.0 This is an open access article under the CC BY 4.0 license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.22514/jofph.2025.064 | PubMed: 41436103 | PMC: PMC12738272
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.3 MB)
1. Introduction
Burning mouth syndrome (BMS), is a multifaceted idiopathic orofacial disorder characterized by persistent, chronic, and spontaneous pain. Patients often describe this discomfort as an oral burning sensation, lasting for over three months, without any identifiable local or systemic pathological changes. Although the tongue is frequently affected, any intraoral site may experience these distressing sensations [ref. 1]. Historically, this condition has been referred to by various terms, such as stomatodynia, glossodynia, burning tongue, oral dysesthesia, and complex oral sensitivity disorder—reflecting the broad spectrum of symptoms experienced by patients [ref. 2, ref. 3, ref. 4]. Moreover, individuals with BMS generally present with a range of additional intra-oral and extra-oral symptoms, further complicating and delaying the diagnostic process [ref. 5].
In recent years, a significant conceptual shift has emerged regarding the classification of BMS. While traditionally labeled as a “syndrome” due to its heterogeneous symptomatology—including burning sensations, xerostomia, dysgeusia, and other additional oral symptoms—this terminology has been increasingly questioned. The term “syndrome” implies a consistent cluster of clinical features that recur across most patients, yet in BMS, only the burning sensation appears to be reliably present, while other associated symptoms vary widely in intensity and presence. This inconsistency has prompted several experts in the field to advocate for a redefinition of BMS as a “disorder” rather than a “syndrome”. A recent Delphi consensus among international orofacial pain specialists supported the adoption of the term Burning Mouth Disorder (BMD) to more accurately reflect the clinical and pathophysiological complexity of the condition [ref. 6]. This terminology emphasizes the chronic neuropathic nature of the pain and the exclusion of other identifiable causes, aligning with the latest International Classification of Diseases (ICD-11) classification which categorizes BMS under “chronic primary pain” conditions [ref. 7].
Concurrently, an expanded diagnostic framework has been proposed to encompass patients presenting with dysaesthetic symptoms (such as tingling, taste disturbance, xerostomia, numbness, and itching) and perceptual symptoms (such as intraoral foreign body sensation, altered perception of tongue size or color, and sialorrhea), but without burning sensations, yet sharing key clinical features with BMS. The term Oral Dysaesthetic and Perceptual Disorder (ODPD) has been introduced to define this under-recognized subgroup, underscoring the need to broaden diagnostic criteria to better capture and manage such phenotypes [ref. 8].
In this review, we retain the term BMS for consistency. However, we fully acknowledge the evolving nosology that favors the more precise designation of BMD and the recognition of emerging categories such as ODPD. These developments have important implications for improving diagnostic accuracy, guiding future research, and optimizing clinical management.
Despite ongoing research, the pathogenesis of BMS remains elusive. Current evidence suggests the involvement of central and peripheral neuropathy [ref. 9, ref. 10], along with potential contributions from endocrinological and psychosocial factors [ref. 11, ref. 12, ref. 13], in the development and perpetuation of this enigmatic condition. Recognizing the intricate interplay of these factors is essential in developing effective strategies for diagnosis, treatment, and management.
This review aims to comprehensively present the multifaceted nature of BMS, exploring its clinical manifestations, potential underlying mechanisms, and the challenges encountered in both diagnosis and treatment. A pivotal focus of this review is on proposing and discussing specific diagnostic approaches to assist clinicians in differential diagnosis, and in the formulation personalized treatment plans for BMS patients. By synthesizing existing knowledge and presenting a structured approach to understanding and addressing BMS, this review endeavors to deepen the understanding of this complex affection and enhance clinical practices.
2. Epidemiology: prevalence, age, and sex distribution
The worldwide prevalence of BMS is estimated at 1.7% in the general population, rising to 7.7% among dental patients in clinical settings. However, precise prevalence data can vary significantly due to differences in populations, geographic regions, and diagnostic criteria [ref. 14]. Additionally, a population-based study by Kohorst et al. [ref. 15] reported a prevalence of 0.11% for BMS, with a higher incidence observed in women over the age of 60. In the general population, BMS prevalence is higher in Europe (5.5%) and North America (1.1%), and lower in Asia (1%). Conversely, in clinical settings, the prevalence is higher in Asia (8.9%) compared to South America (6.1%) and Europe (6.5%). These variations highlight the impact of geographic location on BMS prevalence [ref. 14].
Gender-specific analysis revealed a prevalence of 1.1% in females, significantly higher than the 0.38% observed in males in the general population. Consequently, the female-to-male ratio is approximately 3:1 in population-based studies, whereas in clinical settings it may reach 6:1 to 9:1, highlighting a notable gender disparity in BMS prevalence. This disparity may be related to physiological and behavioral differences [ref. 14].
Interestingly, recent large cohort studies suggest a shift in the onset of BMS towards older ages, with the disease manifesting predominantly around 65 years, emphasizing the evolving landscape of BMS epidemiology in the context of an aging population [ref. 14]. An age-specific analysis indicated a higher incidence of BMS among individuals aged 50 and above (3.31%) compared to those under 50 (1.92%). This trend may be attributed to various factors that differ across the countries, including hormonal changes, nutritional deficiencies, differences in socioeconomic and medical conditions or the cumulative effects of environmental factors over time (Table 1, Ref. [ref. 14]). This also highlights important methodological differences across studies.
Table 1.: Prevalence of burning mouth syndrome (BMS) according to Wu et al. [ref. 14].
| Prevalence category | ||
| General prevalence | 1.73 | |
| Clinical prevalence | 7.72 | |
| Country-specific general prevalence | ||
| Europe | 5.5 | |
| North-America | 1.1 | |
| Asia | 1.05 | |
| Country-specific clinical prevalence | ||
| Asia | 8.96 | |
| Europe | 6.46 | |
| South America | 6.05 | |
| Gender-specific general prevalence | ||
| Female | 1.15 | |
| Male | 0.38 | |
| Age-specific prevalence (yr) | ||
| <50 | 3.31 | |
| >50 | 1.92 | |
3. Aetiopathogenesis
3.1 Neuropathological insight
The pathogenesis of BMS remains an enigma and is not fully understood. Nevertheless, there is a widespread consensus that the etiology of BMS is intricate and multifactorial, with different patients exhibiting various combinations of triggers and perpetuating factors [ref. 16, ref. 17]. Neurological, psychological, and hormonal factors are extensively recognized as potential contributors to the development of this condition [ref. 12, ref. 18, ref. 19]. Additionally, local factors, systemic conditions, and certain medications have been implicated in provoking secondary burning sensations in the oral cavity [ref. 20, ref. 21]. A comprehensive understanding of these diverse factors, their distinct characteristics, and interactions is imperative for accurate diagnosis and, subsequently, for developing more effective treatment strategies.
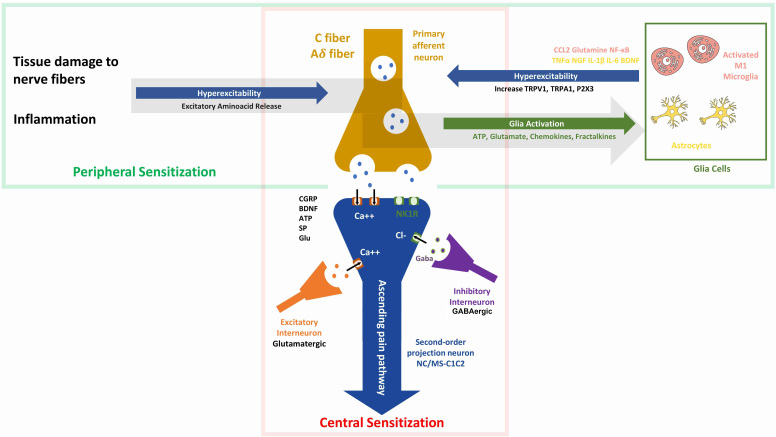
The trigeminal nerve transmits nociceptive sensory information to various regions of the central nervous system, which process both the sensory-discriminative and affective-emotional aspects of orofacial pain [ref. 22] (Fig. 1). Recent neuroimaging advancements have significantly enhanced our comprehension of the pathophysiological mechanisms underlying BMS [ref. 23, ref. 24]. These studies have provided consistent evidence that BMS is a neuropathic disorder involving both the peripheral (PNS) and central nervous systems (CNS), highlighting the role of the trigeminal nerve in transmitting abnormal pain signals in BMS patients [ref. 25, ref. 26]. In this scenario, both peripheral and central sensitization, characterized by an increased responsiveness of peripheral neurons and an enhanced excitability of central neurons may further contribute to persistent pain [ref. 27] (Fig. 2).
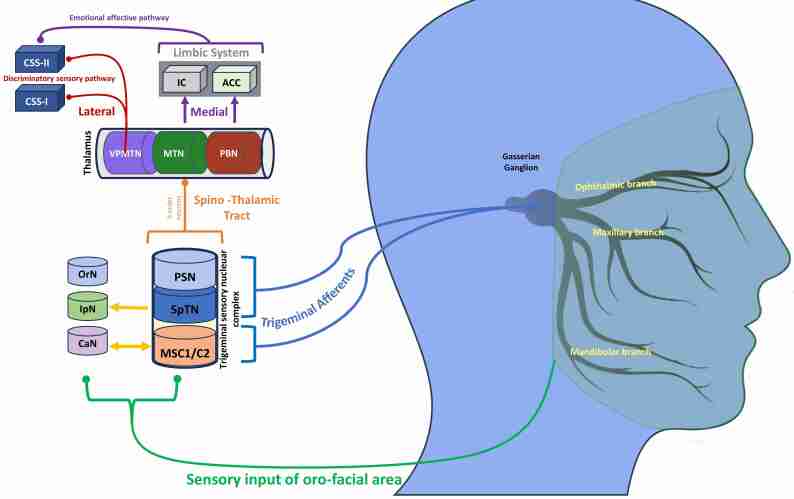
Understanding the involvement of the trigeminal nerve and its pathways in the CNS is crucial for developing targeted treatments for the sensory and emotional dimensions of BMS. BMS can be defined as a nociplastic pain because it is characterized by altered nociception in the absence of clear evidence of actual or threatened tissue damage that would activate peripheral nociceptors or indicate a disease or lesion of the somatosensory system causing the pain. This classification underlines the absence of visible signs or detectable disorders that typically accompany other types of pain, thereby situating BMS within the realm of conditions where the pain arises primarily from dysfunctions in the pain processing systems. Studies have shown that BMS is associated with dysfunction of the pain modulatory system, particularly the descending pain inhibition, where altered brain function can be demonstrated through neurophysiological tests and imaging, revealing an atypical response to painful stimuli without the usual physical causes [ref. 28, ref. 29].
3.2 Central neuropathy
Central neuropathy in BMS involves several key alterations in the CNS, including structural and functional changes that indicate brain hypoactivity, impacting pain modulation and sensory processing [ref. 23, ref. 24, ref. 26, ref. 30].
3.2.1 Striatal dopamine, substantia Nigra and midbrain raphe alterations
Striatal dopamine dysfunction, particularly depletion in the putamen, impairs pain inhibition in the trigeminal brainstem complex. Transcranial sonography reveals unique abnormalities, such as hypoechogenicity in the substantia nigra and midbrain raphe, suggesting nigrostriatal dopaminergic hypofunction. Indeed, a hypofunction in the nigrostriatal dopaminergic pathway, particularly in the basal ganglia and sensory cortex, has been suggested as a cause of reduction of endogenous pain inhibitory control. Specifically, the increased availability of dopamine D2 receptors, reflecting dopamine depletion, may contribute to chronic neuropathic pain within the trigeminal distribution [ref. 31, ref. 32].
3.2.2 Brain activation and connectivity
Advanced neuroimaging techniques, such as functional Magnetic Resonance Imaging (fMRI), have revealed a spectrum of changes affecting both structural and connectivity aspects of the brain’s pain matrix in BMS patients. These patients exhibit decreased activation, especially in the thalamus, alongside an expansion in hippocampal grey matter volume (GMV) and a reduction in the medial prefrontal cortex [ref. 33]. Further supporting this, studies have established a correlation between pain intensity and reductions in grey matter concentration (GMC) and GMV in BMS patients compared to control groups. Specifically, these reductions are observed in the thalamus, cingulate gyrus, cerebellar lobules, insula/frontal operculum, inferior temporal area, primary motor cortex, and medial and dorsolateral prefrontal cortex [ref. 24, ref. 34]. Similar reductions in GMV in the prefrontal cortex have been documented in various chronic pain conditions, such as back pain, trigeminal neuralgia, temporomandibular disorder, and functional dyspepsia, as well as in depression (D) and anxiety (A) [ref. 35].
Moreover, BMS patients show distinct brain activation patterns, including increased connectivity in the left insula, right amygdala, and right lateral orbitofrontal cortex, and decreased connectivity between the bilateral medial prefrontal cortex and the amygdala. These changes, which correlate with BMS duration, along with reduced thalamic activity, underscore the thalamus’s critical role in pain signal transmission and BMS pathogenesis [ref. 30]. Furthermore, reductions in cerebral blood flow in the middle temporal gyrus and insula have been documented, highlighting the role of altered neurovascular dynamics in BMS [ref. 35].
3.2.3 White matter hyperintensities
Recent research has identified an elevated incidence of White Matter Hyperintensities (WMHs) in specific brain regions of BMS patients, particularly in the frontal, parieto-occipital, and temporal areas. These WMHs, visible on MRI through T2-weighted or Fluid Attenuated Inversion Recovery (FLAIR) sequences, are early indicators of brain vulnerability and may signify accelerated brain aging (Fig. 3).
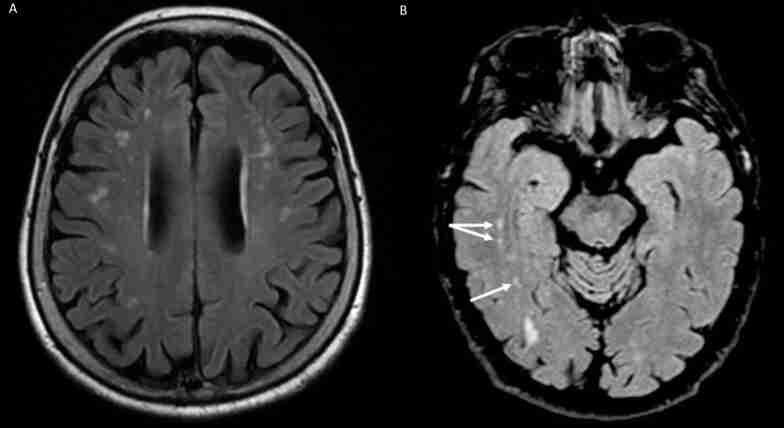
They are associated with cerebral small vessel arteriolosclerosis and potential links to vascular dementia and neurodegenerative diseases like Alzheimer’s. The connection between chronic orofacial pain, including BMS, and WMHs remains debated, with uncertainty about whether WMHs contribute to the onset of chronic pain or exacerbate BMS symptoms [ref. 36]. A recent study by Kato et al. [ref. 23] used Diffusion Tensor Imaging (DTI) and Neurite Orientation Dispersion and Density Imaging (NODDI) to evaluate white and gray matter abnormality in BMS. The study included 14 BMS patients and 11 healthy controls, analyzing various metrics such as fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD), radial diffusivity (RD), intracellular volume fraction (ICVF), and isotropic volume fraction (ISO). Results showed that BMS patients exhibited higher FA and ICVF and lower MD and RD in widespread white matter areas, as well as higher ISO and lower MD and RD predominantly in the amygdala. These findings suggest microstructural changes in both white and grey matter, indicating alterations in myelination, astrocytic hypertrophy, and neuroinflammation, reflecting the complex pathology of BMS [ref. 23].
3.3 Peripheral neuropathy
3.3.1 Peripheral nerve fiber alterations
BMS is linked to dysfunction of peripheral trigeminal fibers and their connectivity with the brainstem in about 20–30% of the patients [ref. 37]. Multiple studies have identified alterations in the nerve fibers of the tongue’s mucosal lining in BMS patients [ref. 25, ref. 38]. These studies have reported a decreased density of small intraepithelial and subpapillary nerve fibers, while larger subepithelial fibers remain intact. This significant reduction in intraepithelial nerve fiber density suggests the involvement of trigeminal small-fiber sensory neuropathy or atrophy of the oral mucosa. However, current histological evidence does not conclusively attribute these reductions specifically to Aδ or C fibers. Some studies hypothesize that degeneration of Aδ fibers may lead to disinhibition of C fibers, contributing to the persistent burning pain characteristic of BMS [ref. 37, ref. 39]. Additionally, sensory alterations in BMS correlate with functional deficits in other cranial nerves, such as facial and olfactory nerves. This dysfunction is supported by quantitative sensory testing (QST) and neurophysiological recordings, indicating abnormal responses to thermal and pain-evoked potentials, supporting the role of peripheral neuropathy in BMS. Trigeminal tactile Aβ fiber hypofunction has also been identified through electrical thresholds analysis following blink reflex stimulation in BMS patients [ref. 37, ref. 38]. Taste perception alterations in BMS patients have been a subject of investigation, with studies yielding varying results. Some research suggests that BMS patients experience disturbances in taste perception, potentially linked to dysfunction of the chorda tympani nerve, which plays a crucial role in taste sensation. These disturbances may manifest as reduced sensitivity to sweet, sour, salty, and bitter tastes, implicating the involvement of small afferent nerve fibers in the pathophysiology of BMS [ref. 40]. However, recent well-controlled studies have challenged this association. For instance, Kolkka et al. [ref. 41] conducted a study comparing BMS patients to age- and gender-matched control subjects and found no significant differences in taste perception or salivary composition between the groups. This suggests that taste alterations may not be a consistent feature of BMS and highlights the need for further research to clarify these findings.
3.3.2 Receptor dysregulation
Receptor dysregulation has been implicated in the pathophysiology of BMS. Immunohistochemical studies have demonstrated increased expression of transient receptor potential vanilloid 1 (TRPV1) and cannabinoid receptor 1 (CB1), along with decreased expression of cannabinoid receptor 2 (CB2), in the oral mucosa of BMS patients [ref. 42]. These receptors are involved in nociceptive signaling and may contribute to peripheral sensitization. Voltage-gated sodium channels (VGSCs), particularly Nav1.7 and Nav1.9, have also been investigated, although studies have not found statistically significant differences between BMS patients and healthy controls [ref. 43]. Purinergic receptors (P2X3) are known to be expressed in gustatory fibers of the chorda tympani rather than in trigeminal sensory fibers, suggesting it may be more relevant to taste signaling than to nociception in BMS [ref. 44]. Additionally, non-neuronal cells such as fibroblasts, Schwann cells, and astroglia can contribute to pain sensitization by releasing nerve growth factor (NGF), which increases during inflammation and enhances nociceptor responsiveness [ref. 45].
3.3.3 Recent advance in peripheral neuropathy
Recently, a new hypothesis for the pathogenesis of BMS has been proposed. This hypothesis revolves around the uncontrolled activation of specific calcium-permeable transmembrane channels, found within the intraoral mucosal nerve fibers, potentially triggered by an increase in reactive oxygen species (ROS) or impairments in anti-apoptotic pathways, leading to oxidative stress-mediated apoptosis signaling. This cascade of events likely results in the depolarization of nerve endings, generating action potentials that may be interpreted centrally as pain [ref. 46]. Neuron-specific enolase (NSE), a glycolytic enzyme predominantly found in neurons and neuroendocrine cells, has been investigated as a potential biomarker of neuronal damage or dysfunction in various neuropathic conditions. In the context of BMS, elevated levels of NSE have been reported in some studies, suggesting an underlying neuropathic component in a subset of patients [ref. 47]. Increased NSE expression may reflect peripheral or central sensitization processes or subtle neuronal injury within the trigeminal system [ref. 48]. Although current evidence remains limited and preliminary, NSE could contribute to the identification of neurobiological alterations in BMS and may, in the future, aid in the stratification of patients and the development of targeted therapeutic approaches. Further studies are warranted to validate its clinical utility in this setting.
4. Stress and neuromodulation
Stress plays a significant role in the pathogenesis and symptom modulation of BMS. Beyond immediate adaptive responses via catecholamines and cortisol, chronic stress induces long-term neuroendocrine, immune, and epigenetic changes that affect pain perception. These include the release of pro-inflammatory cytokines, altered neural plasticity, and modulation of gene expression through mechanisms like DNA methylation and microRNAs, potentially facilitating central sensitization and hyperalgesia. Experimental studies have shown that prolonged stress can suppress dopaminergic activity in the nucleus accumbens (NAc) and alter tyrosine hydroxylase expression in the ventral tegmental area (VTA), impairing dopamine synthesis and signaling in regions involved in orofacial pain, such as the NAc, dorsal striatum, and amygdala [ref. 49]. This highlights the bidirectional relationship between stress and pain processing in BMS. Moreover, BMS patients appear particularly vulnerable to stress-induced hyperalgesia. During the COVID-19 pandemic, they exhibited increased symptom severity and distress compared to controls, including higher levels of intrusive cognitions and emotional dysregulation. Chronic stress also affects neuroendocrine biomarkers: elevated salivary cortisol and α-amylase levels have been observed in BMS patients, with reductions correlating with symptom improvement. These markers may serve as objective indicators of stress-related exacerbation in BMS.
5. Interplay of mood disorders and burning mouth syndrome
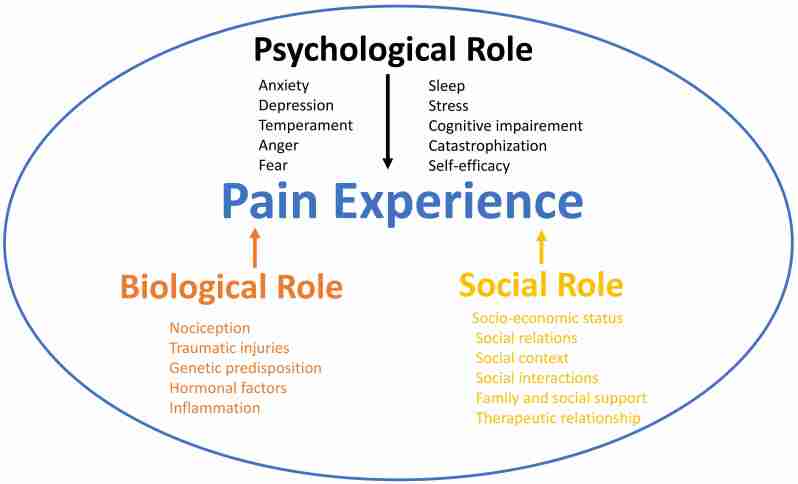
It is widely acknowledged that a dynamic interplay of psychological and neurological mechanisms plays a crucial role in the syndrome’s onset and progression, as suggested by the biopsychosocial model (Fig. 4).
Mood disorders (MDs), such as A and D, and certain aberrant personality traits are often linked with conditions like fibromyalgia, irritable bowel syndrome, chronic fatigue, and notably, BMS. A pivotal study by Taiminen et al. [ref. 50] revealed that over half of the patients with BMS had experienced at least one Axis I psychiatric disorder during their lifetime, with major D being the most prevalent. Notably, a significant proportion of these psychiatric conditions preceded the onset of BMS symptoms, suggesting a potential contributory role in the pathogenesis of the syndrome. Additionally, a considerable number of patients exhibited Axis II personality disorders, particularly those within cluster C, characterized by anxious and fearful behaviors. These findings underscore the importance of comprehensive psychiatric evaluation in patients presenting with BMS symptoms [ref. 50].
5.1 The chicken or the egg: causality dilemmas
A significant challenge in understanding BMS lies in determining whether MDs are a cause or a consequence. Until now, it is controversial whether A and D act as driving forces in the development of BMS or, conversely, if the onset of BMS triggers a decline in mental health, potentially leading to these mood disorders over time [ref. 51]. Recent evidence indicates that in approximately 80% of cases, MDs often precedes the onset of BMS, suggesting that psychological impairments appear to be potential contributing factors to BMS’s development. Specifically, patients with A and D are at a higher risk of developing BMS compared to those without such conditions. Interestingly, this correlation does not extend to bipolar disorder, which does not show a significant association with BMS [ref. 13]. A noteworthy finding is that A seems to predispose individuals to BMS at an earlier stage than D, suggesting a heightened vulnerability in anxious individuals. This leads to a hypothesis that A might induce secondary D, which in turn could contribute to the manifestation of pain, reinforcing the idea that BMS might be a somatic expression of depressive disorders [ref. 13].
Interestingly, a comprehensive nationwide cohort study by Kim et al. [ref. 18] found that patients with BMS were at an increased risk of developing D and A compared to individuals without BMS. This association persisted even after adjusting for sociodemographic characteristics and comorbid conditions, suggesting a potential predisposition to D and A in BMS patients [ref. 18]. Although the exact nature of this relationship remains unresolved, analysis of the studies indicates that the relationship between mood and pain is inherently bidirectional, with each factor influencing the other. Chronic pain and discomfort associated with BMS may exacerbate psychological distress, creating a challenging cycle that complicates both diagnosis and treatment. Conversely, the persistence of mood disorders can amplify the perception of pain, further entrenching this complex interplay between mood and pain [ref. 52].
5.2 Sleep disturbances and BMS: a bidirectional relationship?
MDs are closely linked with sleep disturbances (SD), which can be both symptoms and contributing factors. Common sleep issues like insomnia, difficulties in falling or staying asleep, and hypersomnia (excessive sleepiness) are pivotal in diagnosing MD. Poor sleep can increase irritability and exacerbate feelings of sadness or hopelessness, potentially worsening mood disorders. In some cases, SD may precede and trigger mood disorders in susceptible individuals. Long-term sleep disruption can alter brain chemistry and function, predisposing individuals to mood dysregulation. The co-occurrence of SD, with or without MD, may further exacerbate conditions like BMS.
In a case-control multicenter study, 78.8% of BMS patients experienced poor sleep, showing also an overall poorer sleep quality, higher levels of daytime sleepiness, and increased D and A compared to healthy controls. This highlights a significant correlation between BMS, poor sleep quality, A, and D. Possibly, poor sleep, by exacerbating negative moods, may create a feedback loop with the pain experienced by BMS patients [ref. 53, ref. 54].
5.3 Neurobiological insights among BMS, mood disorders and sleep disturbances
MDs, SD, chronic orofacial pain and BMS share overlapping neurophysiological mechanisms. These include dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis, altered monoaminergic neurotransmission (such as serotonin and dopamine), and dysfunctions in brain regions like the anterior cingulate cortex, insula, and prefrontal cortex [ref. 30, ref. 55, ref. 56]. Functional MRI studies further support this overlap, revealing structural and connectivity changes common to BMS, D and SD [ref. 24, ref. 30].
5.3.1 Brain changes and connectivity
Key brain areas, including the prefrontal cortex, amygdala, and hippocampus, exhibit notable changes in size and activity in individuals with mood disorders and chronic orofacial pain. These changes impact how we regulate emotions and perceive pain. For instance, increased activity in the default mode network (a brain network active during rest) is observed in BMS and MD. This could reflect a heightened focus on internal thoughts and pain sensations. Furthermore, there is a notable dysregulation in how emotions are processed, suggested by increased connectivity between the amygdala and other limbic regions. The insula, another critical area for emotional and internal bodily sensation processing, also shows increased connectivity. This change is believed to alter emotional and bodily awareness. On the other hand, a decrease in connectivity between the prefrontal cortex and regions processing pain signals has been observed. This suggests a disruption in both emotion regulation and cognitive processing in both conditions. Other brain areas, like the thalamus, somatosensory cortex, and hippocampus, also show altered connectivity. These changes in brain connectivity and function could lead to an intensified perception of pain and its prolonged presence, potentially affecting memory processes and the regulation of stress responses [ref. 30].
5.3.2 Neurotransmitters imbalance and genetic factors
Recent studies have highlighted a compelling link between neurotransmitter imbalances and the co-occurrence of MD and BMS, finding that BMS patients exhibit dysregulated levels of key neurotransmitters, including serotonin, dopamine, and noradrenaline, which are crucial for pain perception and modulation. This neurochemical basis for BMS is supported by the varied success of antidepressants in alleviating BMS symptoms [ref. 57, ref. 58]. Research into the role of dopamine, its receptors, and genetic polymorphisms has shown significant overlap in the dopaminergic system’s involvement in both pain and mood regulation. Dopamine, essential for the brain’s reward system, influences pleasure, mood regulation, and pain perception through direct and indirect pathways. Alterations in dopamine receptor functionality and levels might underlie the sensory issues characteristic of BMS [ref. 16]. Genetic studies have further explored these connections, especially the associations between dopamine receptor gene polymorphisms 2 and 4 (DRD2 and DRD4) and major depressive disorder. These polymorphisms can affect dopamine receptor expression and functionality, impacting mood regulation [ref. 59]. In BMS patients, dysregulation of these receptors, especially in pain-related brain areas, has been documented. Kolkka et al. [ref. 26] investigated the 957C>T polymorphism of the DRD2 gene in BMS patients, finding that the 957TT genotype is linked to higher pain thresholds, greater pain interference in daily activities, and more intense suffering. These findings collectively suggest a potential genetic predisposition in certain BMS and mood disorder patients related to dopamine pathway functionality.
5.3.3 Neuroinflammation and HPA axis dysregulation
Research is increasingly focusing on neuroinflammation as a common pathway linking chronic pain conditions with psychiatric comorbidities [ref. 60]. The role of pro-inflammatory cytokines in altering neurotransmitter systems and neuronal activity is well-documented, influencing both emotional states and pain sensitivity. Specifically, cytokines, such as interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α), can modulate brain function and induce depressive symptoms, particularly through their effects on neurotransmitter metabolism and brain signaling pathways. Indeed, these cytokines may disrupt the synthesis and reuptake of neurotransmitters, impacting both pain perception and mood [ref. 61]. Although elevated salivary levels of IL-6 and TNF-α have been reported in BMS patients by Al-Maweri et al. [ref. 62], findings across studies remain inconsistent. A recent prospective case-control study by Moreau et al. [ref. 63] found no significant differences in salivary cytokines, steroid hormones, or neuroinflammatory markers between BMS patients and healthy controls, suggesting that systemic inflammation may not play a central role in BMS pathogenesis. This connection is attributed to the HPA axis’s role in mediating stress responses and its influence on neuroendocrine and immune functions. It suggests that HPA axis dysregulation might contribute to BMS pathogenesis, potentially through stress-induced neurogenic inflammation or altered pain perception [ref. 64]. Indeed, prolonged activation of the HPA axis can lead to cortisol overproduction, affecting neurotransmitter systems, notably serotonin and dopamine, crucial in both pain and mood regulation. Additionally, increased levels of pro-inflammatory cytokines, associated with both BMS and mood disorders, can further dysregulate the HPA axis, creating a feedback loop that may exacerbate these conditions [ref. 65].
5.3.4 Central sensitization
The alterations in CNS processing, commonly observed in MD and BMS, may lead to a state of central sensitization. This condition is typified by the heightened responsiveness of nociceptive neurons in the CNS to both normal and subthreshold afferent input, potentially resulting in amplified pain perception in BMS. Furthermore, central sensitization is also linked to MD, given its significant impact on neural networks that regulate emotions [ref. 26, ref. 66] (Fig. 2). It is important to note that the neurobiological mechanisms underpinning central sensitization involve both changes in synaptic efficacy and an increase in neuronal excitability within the CNS. These alterations can influence key neurotransmitter systems, such as serotonin and norepinephrine, which play vital roles in regulating both pain and mood [ref. 67].
6. Hormonal imbalance in BMS
Hormonal dysregulation, especially involving sex steroids, has been considered a possible contributing factor in BMS largely based on its higher prevalence in peri- and postmenopausal women. While early studies suggested that reduced estrogen may impact peripheral nerve sensitivity and salivary function, recent data remain inconclusive. For example, a recent prospective case-control study found no significant differences in salivary estrogen levels between BMS patients and healthy controls [ref. 63], while other investigations have reported reduced salivary estradiol in symptomatic postmenopausal women [ref. 68]. Experimental research has also proposed that menopause-related estrogen decline could increase pain sensitivity through upregulation of TRPV1 and NGF pathways, potentially sensitizing peripheral nerves [ref. 69]. However, such mechanisms remain largely theoretical and need further validation in clinical settings. Regarding adrenal steroids, data on dehydroepiandrosterone (DHEA) are inconsistent. Some studies suggest lower salivary DHEA levels in BMS patients, possibly reflecting chronic stress or adrenal fatigue, but other investigations have failed to confirm this association [ref. 70]. Overall, while hormonal fluctuations—particularly related to menopause—may contribute to BMS susceptibility, current evidence is mixed, and hormone replacement therapy has shown inconsistent results, suggesting that hormonal factors are likely part of a broader multifactorial etiology.
7. Association of systemic comorbidities and BMS
BMS patients frequently encounter a higher incidence of simultaneous medical comorbidities and consume more medications than controls. This scenario often results in an overall poorer health status, which can, in turn, intensify the symptoms and potentially accelerate the progression of BMS itself. MD, hypertension, hypercholesterolemia, hyperhomocysteinemia, hypothyroidism, and gastroesophageal reflux disease (GERD) are the most frequent comorbidities associated with BMS. Concerning co-occurring pain conditions, a systematic review by Moisset et al. [ref. 71] demonstrated that such comorbidities are relatively uncommon among individuals with BMS, thereby supporting the hypothesis that BMS may involve distinct pathophysiological mechanisms compared to other chronic pain disorders. Understanding the multifaceted nature of these comorbidities and their interactions with BMS is essential for developing a comprehensive and tailored treatment plan. This involves not only addressing the primary symptoms of BMS but also managing any underlying or concurrent conditions that may be contributing to the patient’s overall health challenges.
7.1 BMS and mood disorders
Several studies reported that MDs are the most frequently associated comorbidities in BMS [ref. 72]. Specifically, A, D, MDs, and medically unexplained extraoral physical symptoms (MUEPS) are the most commonly observed [ref. 53, ref. 72, ref. 73]. On one side, patients suffering from A and D are at an increased risk of developing BMS, with this risk being more pronounced in females and increasing with age. Notably, a prevalence of 80% of MDs has been observed among BMS patients, often preceding the development of BMS, suggesting that psychological factors might contribute to the pathogenesis of the disease [ref. 13]. On the other side, BMS patients may also be predisposed to A (odd ratio 2.64) and D (odd ratio 3.18) [ref. 72]. Furthermore, evidence suggests that individuals with BMS may be at increased risk for A and D, independent of sociodemographic characteristics and other comorbid conditions [ref. 17]. Considering this evidence, which supports the hypothesis of a bidirectional relationship between MDs and BMS, it is crucial to acknowledge that the co-occurrence of MDs and BMS tends to exacerbate both psychological symptoms and oral pain. BMS patients additionally show a high prevalence of SD, ranging between 78.8% and 90.2%. Interestingly, in almost half of the patients suffering from insomnia, the onset of this sleep disorder was observed on average 4 years before the BMS development [ref. 13, ref. 53]. Moreover, in recent research on 500 BMS patients, MUEPS have been observed in 169 (33.8%) patients with a mean of 2.8 ± 1.50 symptoms for each patient. In this study, irritable bowel syndrome (48; 9.6%), fibromyalgia (36; 7.2%), tinnitus (32; 6.4%), and vulvodynia (21; 4.2%) were the most common MUEPS reported [ref. 73]. Interestingly, Leuci et al. [ref. 74] reported that vulvodynia is often linked with lower sexual desire, though no direct correlation with A, D, or sleep disorders was found. This finding may suggest that BMS might independently affect sexual desire, potentially due to dysfunction in the brain’s reward system [ref. 74]. Additionally, BMS patients may experience other psychological comorbidities like social phobia, cancerphobia, hypochondria, and neuroticism. However, bipolar disorder does not show a significant link to BMS [ref. 13].
7.2 BMS and vascular comorbidities: hypertension, hypercholesterolemia and hyperhomocysteinemia
Several vascular risk factors have been investigated about BMS, including hypertension, hypercholesterolemia, and hyperhomocysteinemia. Although evidence remains limited and somewhat heterogeneous, these conditions have been reported with increased prevalence in BMS populations, suggesting potential shared mechanisms or comorbid vulnerability.
Several studies have explored the association between hypertension and BMS, although findings remain inconsistent. A case-control study by Jin et al. [ref. 75] found no significant difference in hypertension prevalence between BMS patients and controls. In contrast, other studies have reported a higher prevalence of hypertension among BMS patients, particularly in older women. For instance, one study indicated that 51.2% of postmenopausal BMS women had hypertension compared to 30.4% of age-matched controls [ref. 76]. This suggests that certain subgroups, such as older or socioeconomically disadvantaged women, may be more vulnerable to comorbid cardiovascular conditions alongside BMS. Factors such as lower education level and unemployment were also associated with increased hypertension rates in BMS patients, pointing to a potential biopsychosocial component in disease expression.
As for hypercholesterolemia, although specific studies investigating its pathophysiological role in BMS are lacking, data from Adamo et al. [ref. 5] reported a prevalence of 38.6% among BMS patients, suggesting that lipid imbalance may frequently coexist with BMS. These findings underline the importance of evaluating cardiovascular risk factors during clinical assessment and management of BMS patients.
Elevated homocysteine levels have been observed in some patients with BMS, although findings are also still limited. Hyperhomocysteinemia has been associated with a wide range of systemic effects, including vascular and neurological complications, and has been proposed as a potential risk factor for peripheral neuropathy. In BMS, a study by Adamo et al. [ref. 77] reported a high prevalence of hyperhomocysteinemia (up to 73%), which may be related to the presence of white matter hyperintensities (WMHs) and cerebral small vessel disease. While the exact mechanisms remain unclear, these findings suggest a possible role of vascular dysfunction in the pathophysiology of BMS. However, further research is needed to establish whether elevated homocysteine levels are causally linked to BMS or reflect broader comorbid conditions.
7.3 BMS and thyroid disorders
The association between thyroid disorders and BMS is a subject of ongoing debate. While some researchers believe that thyroid alterations have minimal impact on BMS, others argue that they may contribute to symptoms. Notably, thyroid disorders are not currently a criterion for excluding BMS diagnosis. The thyroid gland produces hormones such as triiodothyronine (T3) and thyroxine (T4), which are crucial for tissue development, metabolism, and various functions and processes of the nervous system. Additionally, these hormones are involved in the maturation and specialization of taste buds. Consequently, a deficiency in thyroid hormones may contribute to dysgeusia (distorted taste perception) [ref. 78]. Given the high prevalence of taste disturbances in BMS, several researchers have explored this connection. Specifically, the prevalence of hypothyroidism largely varies, with a study showing a prevalence of hypothyroidism of 5.2% [ref. 79] and another of 13.6% [ref. 5]. On the other side, even a lower prevalence of hyperthyroidism has been reported, between 1% and 3.2% [ref. 5, ref. 79]. Given that the majority of BMS patients are women and thyroid disorders predominantly affect women, it is challenging to definitively support the association between BMS and thyroid disorders based on current literature. However, a clinical study by Femiano et al. [ref. 80] reported improvement in BMS symptoms following correction of thyroid hormone dysfunction, supporting a possible therapeutic and diagnostic link between thyroid alterations and BMS [ref. 80].
7.4 BMS and gastroesophageal reflux disease (GERD)
The correlation between BMS and GERD is an area of growing interest in medical research. Until now, the classification systems for BMS have not ruled out gastrointestinal disorders, including GERD from the diagnostic criteria for BMS. Numerous studies suggest a potential link between GERD and BMS. However, a recent scoping review by Li et al. [ref. 81] indicates that the direct correlation and causal relationship between these two conditions still require clear demonstration. GERD is characterized by the backflow of stomach acid into the esophagus, often causing heartburn and can lead to oral burning sensations. This is presumably due to the irritation of nerve endings in the mouth by the refluxed acid, which may exacerbate BMS symptoms. Supporting this theory, observed lower oral pH values in GERD patients, suggest that gastric acid’s impact on the oral mucosa could be a contributing factor [ref. 82]. Additionally, many BMS patients have reported symptom improvement following treatment with proton pump inhibitors (PPIs), as noted in several studies [ref. 83, ref. 84, ref. 85]. This improvement further supports the hypothesis of a GERD-BMS connection. Furthermore, a high prevalence of oral pepsin saliva was found in BMS patients, which could lead to mucosal damage and heightened cytokine levels, triggering immune responses. However, despite gastric acid reflux and pepsin that may cause mucosal damage in the lower esophagus, the extension of tissue injury to oral mucosa has not been clearly demonstrated [ref. 84]. Nevertheless, it was found that GERD is one of the more common systemic comorbidities found in BMS [ref. 5, ref. 81, ref. 85]. These findings suggest a potential interplay between gastrointestinal symptoms and central nervous system manifestations in BMS patients. However, a comprehensive investigation into the mechanisms linking pain need to be further elucidated.
7.5 BMS and cognitive decline and neurological disorders
Recent studies have explored the possible connection between BMS and cognitive changes, although the evidence remains limited and not yet conclusive. For instance, research by Canfora et al. [ref. 86] reported impairments in specific cognitive domains, such as attention, working memory, and executive function, in patients with BMS, while constructive praxis and verbal memory remained preserved. The authors introduced the descriptive term “Burning Fog” to refer to this cognitive profile. Additionally, their study found increased Age-related White Matter Changes (ARWMC) scores in the temporal lobes of BMS patients, raising the hypothesis that microvascular brain alterations could contribute to the observed cognitive symptoms. However, a direct causal link between BMS and cerebral small vessel disease or dementia risk has not been established, and these findings should be interpreted with caution.
Another study by Dugan et al. [ref. 87] compared 120 individuals with BMS to 110 controls, noting significantly lower scores in verbal fluency tests among BMS patients, although no differences emerged in global cognitive function as measured by the Montreal Cognitive Assessment (MoCA). Moreover, patients with BMS and mild cognitive impairment (BMS-MCI) show greater emotional distress and poorer cognitive performance, especially in processing speed and executive function, compared to geriatric MCI patients, highlighting the role of mood and pain in cognitive decline [ref. 88].
The BMS Blueprint Persona study highlighted memory issues as a key challenge for patients, particularly impacting medication adherence. This cognitive aspect underscores the need for digital tools designed to support memory and improve treatment compliance in BMS care [ref. 89]. While there are case reports of BMS-like symptoms occurring in patients with neurodegenerative diseases, such as dementia with Lewy bodies or parkinsonism, these observations are anecdotal and do not establish a specific association between BMS and neurodegeneration. For example, a recent review by Guru et al. [ref. 90] found no direct evidence linking Parkinson’s disease and BMS.
An interesting, though still not fully understood, observation is the association between BMS and Restless Legs Syndrome (RLS), with both conditions potentially involving dopaminergic dysfunction. However, whether this reflects a common pathophysiological mechanism remains to be clarified, as genetic studies and mechanistic data are still inconclusive [ref. 91].
Overall, while some studies suggest that a subset of BMS patients may exhibit subtle cognitive or sensorimotor changes, these findings are not specific to BMS and should be interpreted within the broader context of chronic pain, comorbid mood disorders, and possible age-related factors. At present, no causal or diagnostic link between BMS and neurodegenerative disorders has been established. Further well-designed, longitudinal studies are needed to better understand these potential associations. Interdisciplinary collaboration between dentistry, neurology, and psychiatry remains essential in the comprehensive management of BMS patients.
7.6 BMS in the COVID-19 pandemic
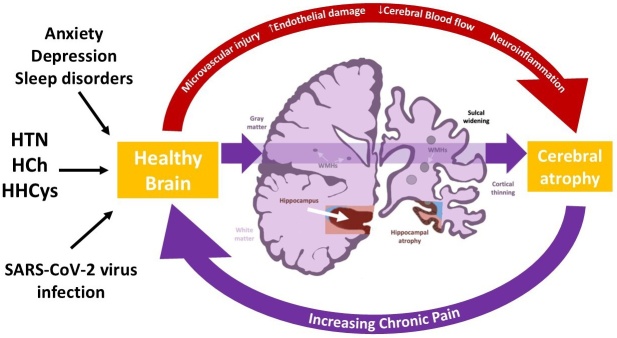
The emergence of the COVID-19 pandemic, caused by the SARS-CoV-2 virus, has introduced new complexities in understanding BMS. COVID-19 is known to affect various bodily systems and has the potential to lead to neurological symptoms, including those related to taste disturbance and burning sensation [ref. 92, ref. 93]. Although some COVID-19 patients have reported these symptoms, establishing a direct link between the virus and BMS continues to be a subject of ongoing research. Nevertheless, it is known that the COVID-19 pandemic has notably impacted the mental and overall health of individuals, leading to increased psychological stress and limited access to healthcare services [ref. 94]. This is particularly relevant for BMS, whose symptoms can intensify under psychological stress. The constrained access to healthcare during the pandemic may have resulted in delayed diagnosis and treatment of BMS, potentially exacerbating the condition. Indeed, Candela et al. [ref. 95] reported an increase in A, worsened sleep quality, and heightened pain intensity in BMS patients. Furthermore, a study by Ottaviani et al. [ref. 96] involving 100 BMS patients and 100 healthy controls from five Italian centers highlighted significant deep loneliness in BMS patients. This loneliness correlated with factors like age, higher education, stress, lower satisfaction in relationships, and perceived social support [ref. 96]. Moreover, during the pandemic, BMS patients exhibited increased post-traumatic stress symptoms, particularly intrusive thoughts, compared to healthy controls. These patients also showed lower levels of resilience and post-traumatic growth, indicating a reduced capacity to find positive meaning and personal growth following stressful life events [ref. 96]. This suggests that BMS patients may lack adaptive coping strategies and a positive mindset, making them more vulnerable to the negative effects of future traumas. Overall, comorbidities such as mood disorders (A, D, sleep disorders) and cardiovascular risk factors (hypertension, hypercholesterolemia, hyperhomocysteinemia) alongside the emerging impact of SARS-CoV-2 infection significantly contribute to brain aging in BMS patients. These factors, particularly cardiovascular risks, lead to microvascular injury, endothelial damage, and reduced cerebral blood flow, resulting in neuroinflammation, WMHs, and cerebral atrophy. These brain alterations may further increase chronic pain, creating a detrimental cycle. Chronic pain and WMHs serve as biomarkers of brain frailty, promoting progressive brain aging, reducing neuroplasticity, and exacerbating pain perception and mood disorders (Fig. 5).
8. Classification system of ICOP 2020; ICHD-3, ICD-11, and DSM-V
In the last years, the conceptualization and categorization of BMS have undergone significant changes shaped by its clinical manifestations and treatment outcomes. Multiple classification systems have been introduced to refine its definition and differentiate it from oral burning sensations linked to various local or systemic factors. These classification systems have been developed not only for accurate disease identification but also to enhance patient understanding and acceptance of their condition [ref. 77]. Establishing universally accepted diagnostic standards has been crucial for clinicians for precise disease identification and treatment strategies and for patients to benefit from a clear diagnosis framed within a globally recognized system, aiding in their understanding and acceptance of the disease.
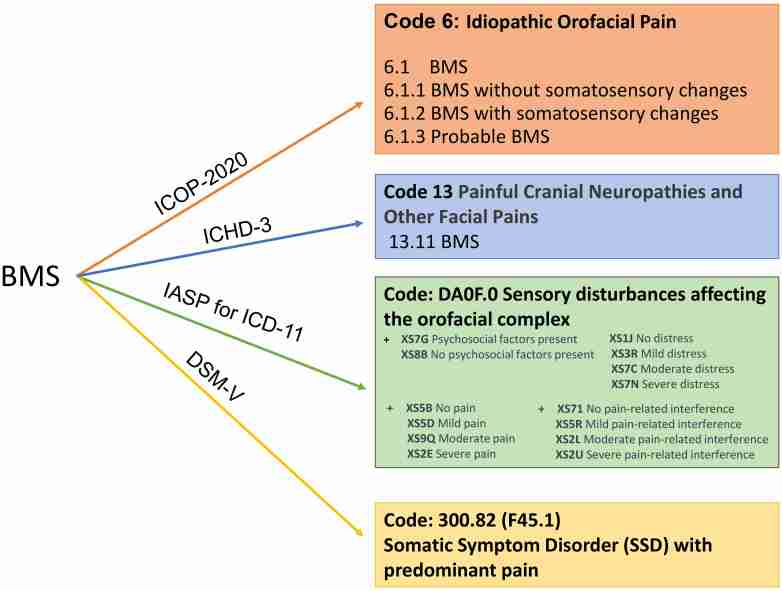
Although there is no consensus regarding the use of a universal and unique classification for BMS, the most used classifications are:
– The International Classification Headache Society (ICHD-3) [ref. 97];
– The International Classification of Orofacial Pain (ICOP-2020) [ref. 1];
– The International Association for the Study of Pain (IASP) for the International Classification of Diseases (ICD-11) [ref. 7] (Table 2, Fig. 6).
Table 2.: Classification system of ICOP 2020; ICHD-3 beta version, IASP for ICD-11.
| ICOP 2020 | Idiopathic orofacial pain | (6.1.1) BMS without somatosensory changes (6.1.2) BMS with somatosensory changes (6.1.3) Probable BMS | – Burning quality – Bilateral or unilateral – Poorly localized – Persistent – Felt superficially in the oral mucosa – Additional symptoms (xerostomia, taste disturbance) | Clinical neurological deficit – Other oral diseases – Systemic diseases (diabetes, nutritional deficiencies, thyroid disease) – medications | Quantitative somatosensory tests Blood tests (nutritional deficiencies, glycemic control, and thyroid function) oral cultures Imaging Psychometric tests |
| ICHD-3 beta version | Painful Cranial Neuropathies and Other Facial Pains | 13.11 | – Burning quality – Bilateral and diffuse – Felt superficially in the oral mucosa – Additional symptoms (xerostomia, taste disturbance) | – Clinical neurological deficit – Other oral diseases – Systemic diseases (diabetes, nutritional deficiencies, thyroid diseases) – medications | Not strictly reported Blood tests (nutritional deficiencies, glycemic control, and thyroid function) |
| IASP for ICD-11 | Sensory disturbances affecting the orofacial complex | DA0F.0 + XS7G: Psychosocial factors present + XS8B: No psychosocial factors present + XS5B: No pain XS5D: Mild pain XS9Q: Moderate pain XS2E: Severe pain + XS1J: No distress XS3R: Mild distress XS7C: Moderate distress XS7N: Severe distress + XS71: No pain-related interference XS5R: Mild pain-related interference XS2L: Moderate pain-related interference XS2U: Severe pain-related interference | -Burning quality Associated with – emotional distress (anxiety, anger/frustration or depressed mood) – interference with orofacial functions such as eating, yawning, speaking, etc. | – Clinical neurological deficit – Other oral diseases – Systemic diseases (diabetes, nutritional deficiencies, thyroid diseases) – medications | Not strictly reported Psychometric tests |
*A diagnosis of 6.1 Burning mouth syndrome implies that quantitative sensory testing has not been performed. 6.1.1 Burning mouth syndrome without somatosensory changes or 6.1.2 Burning mouth syndrome with somatosensory changes should be diagnosed only when quantitative sensory testing have been performed. A diagnosis of 6.1.3 is considered when pain is present for <3 months. Once 3 months have elapsed, the diagnosis becomes 6.1. ICOP: International Classification of Orofacial Pain; ICHD-3: International Classification Headache Society; IASP: International Association for the Study of Pain; ICD-11: International Classification of Diseases-11; BMS: Burning Mouth Syndrome.
Each offers distinct perspectives on categorizing BMS based on clinical evidence and research advancements. Moreover, the ongoing updates to these classifications, readily available online, permit clinicians to evaluate changes across time.
The ICHD-3 includes BMS under the classification of “Painful Cranial Neuropathies and Other Facial Pains”. This classification defines BMS as intraoral burning or dysaesthetic sensation, recurring daily for >2 hours/day for >3 months, without clinically evident causative lesions. The pain is generally burning in character and felt superficially in the oral mucosa but the mucosa appears normal at clinical examination, and sensory testing is normal. The pain is usually bilateral; the most common site is the tip of the tongue, and it may be associated with subjective dryness of the mouth, dysaesthesia, and altered taste. Laboratory investigations and brain imaging may indicate changes in central and peripheral nervous systems. Whether secondary BMS attributed to a local (candidiasis, lichen planus, hyposalivation) or systemic disorder (medication-induced, anemia, deficiencies of vitamin B12 or folic acid, Sjogren’s syndrome, diabetes) should be considered as an entity [ref. 98].
The ICOP 2020 includes BMS in idiopathic orofacial pain and offers a more detailed and inclusive classification system for orofacial pain, aimed at improving diagnosis, treatment, and research. Differently from ICHD-3, ICOP 2020 recommends conducting somatosensory assessments to further classify BMS into subgroups based on the presence or absence of somatosensory alterations. QST is often abnormal (differentiating the two subtypes), whereas clinical sensory examination very rarely reveals slight sensory deficits however, QST currently has no definitive diagnostic value in BMS. Similarly, to ICHD-3, BMS is defined as an intraoral burning or dysaesthetic sensation, recurring daily for >2 hours/day for >3 months, without evident causative lesions on clinical examination and investigation. The pain primarily affects the tongue but may also involve other areas of the oral mucosa, such as the lips, gums, and palatal regions. Although in most patients burning is usually bilateral, on rare occasions, it is unilateral differently from ICHD-3 where all patients who reported unilateral symptoms are included in atypical facial pain. Pain/burning is associated with additional oral symptoms in two-thirds of cases such as xerostomia, dysaesthesia and altered taste. BMS is diagnosed only when all local and systemic causes have been excluded (hence, previously, primary BMS). The diagnosis of BMS in the ICOP 2020 framework requires a thorough patient history, clinical examination, and the exclusion of other possible causes of the symptoms through appropriate investigations. These may include blood tests to check for nutritional deficiencies, glycemic control, and thyroid function, as well as oral cultures and possibly imaging studies. The ICOP 2020 may also consider the impact of the disease on the patient’s quality of life and functional status and the role of psychological comorbidities frequently associated to pain, differently from ICHD-3. Moreover, this classification underscores the importance of a multidisciplinary approach to diagnosing and managing a complex condition such as BMS, which may require contributions from various healthcare professionals, including dentists, oral medicine specialists, neurologists, and psychologists, to achieve optimal patient care [ref. 1].
The IASP has contributed to the development of pain-related diagnoses in the International Classification of Diseases, 11th Revision (ICD-11), which was adopted by the World Health Organization (WHO). The ICD-11 provides a modern standard for identifying health trends and statistics globally and contains significant updates to the classification and criteria of various conditions, including chronic pain conditions like BMS. One of the pivotal advancements in ICD-11 is its refined approach to chronic pain, now recognized both as a symptom and as a distinct disease entity, complete with a supportive coding framework. A groundbreaking feature of ICD-11 is the concept of “multiple parenting”, which allows diagnostic entities to be classified under more than one category. This multifaceted approach aligns more closely with the interdisciplinary nature of medical practice, bridging gaps between specialties such as oral medicine and neurology.
According to ICD-11, BMS is classified under secondary chronic headaches (MG30.62) and more specifically coded as DA0F.0, “sensory disturbances involving the orofacial region” (Foundation ID: 618998878 in the ICD-11 browser). In this classification, BMS is characterized by an intraoral burning or dysaesthetic sensation that recurs for more than two hours per day on 50% of the days over more than three months, without evident causative lesions on clinical investigation and examination. It is characterized by significant emotional distress (A, anger/frustration, or depressed mood) or interference with orofacial functions such as eating, yawning, speaking, etc. The diagnosis is appropriate independently of identified biological or psychological contributors unless another diagnosis would better account for the presenting symptoms. It is possible to include additional codes that specify the intensity of pain/burning; the presence of psychosocial factors and the interference of pain [ref. 7].
The analysis of ICOP 2020, ICHD-3, and ICD-11 underscores the critical role of psychosocial components. Consequently, many authors have advocated for the integration of these classifications with the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). This integration is particularly relevant for BMS within the framework of Somatic Symptom Disorder (SSD), as classified in the DSM-5 [ref. 99]. The DSM-5 code for SSD, 300.82 (F45.1), relates to conditions where individuals experience an intense focus on physical symptoms. These symptoms, such as the chronic burning pain characteristic of BMS, lead to substantial health-related A, disproportionate concerns about the symptoms, and significant distress or functional impairment. In the case of BMS, the emphasis on the subjective experience of distress and the extensive psychological impact, including health-related A and concern over symptoms, aligns with the SSD criteria. The persistent nature of BMS symptoms can perpetuate a cycle of distress and functional impairment, reflecting the SSD framework [ref. 99]. This alignment highlights the importance of a holistic approach to diagnosing and treating BMS. Such an approach should address not only the physical symptoms but also the psychological and social dimensions of the condition, emphasizing the integral role of psychosocial factors in the experience and management of BMS.
9. Diagnosis of BMS
The diagnosis of BMS presents a significant clinical challenge, as it requires the exclusion of other systemic or local disorders that could contribute to oral cavity pain. To accurately diagnose and treat this condition, clinicians should consider patients’ characteristics and pain characteristics at the same time [ref. 17].
Firstly, a detailed examination of the oral cavity, and extensive analysis of socio-demographic characteristics, habits, body mass index (BMI), and medical history including current medications, any history of mood disorders, systemic diseases, and familiarity with psychiatric and cognitive impairment is required for every patient (Table 3).
Table 3.: Characteristic assessment of BMS patients’ profile.
| Characteristics of the patient | Gender |
| Age | |
| Education level | |
| Marital status | |
| Job status | |
| BMI (with categories): | |
| – Underweight: BMI: <18.5 | |
| – Normal weight: BMI: 18.5–24.9 | |
| – Overweight: BMI: 25–29.9 | |
| – Obesity class I: BMI: 30–34.4 | |
| – Obesity class II: BMI: 35–39.9 | |
| – Obesity class III: BMI: 40 or greater | |
| Socio-demographic Characteristics | Smoking |
| Alcohol consumption | |
| Physical activity | |
| Medical Comorbidities | Hypertension |
| Hypercholesterolemia | |
| Previous Myocardial infarction | |
| Other Cardiovascular diseases | |
| Hyperhomocysteinemia | |
| Asthma | |
| Gastroesophageal reflux disease (GERD) | |
| Endocrine diseases | |
| Benign Prostatic Hypertrophy | |
| Hypothyroidism | |
| Hyperthyroidism | |
| HCV infection | |
| HBV infection | |
| Neoplastic disease | |
| Others | |
| Drugs Intake | ACE-inhibitors |
| Calcium Channel blockers | |
| Angiotensin II receptor antagonists (ARBs) | |
| Thiazide Diuretics | |
| Beta-blockers | |
| Statins | |
| Ezetimibe | |
| Antiplatelets | |
| Blood thinners | |
| Bisphosphonates or antiresorptive drugs | |
| Levothyroxine sodium | |
| Steroids | |
| Proton Pump inhibitors | |
| Others |
Recommended assessment for all BMS patients, to identify potential comorbidities and contributing factors (socio-demographic data, habits, medical conditions, and medications). BMI categories follow WHO criteria. BMI: body mass index; ACE: angiotensin-converting enzyme; HCV: Hepatitis C Virus; HBV: Hepatitis B Virus.
An extensive clinical examination should investigate signs of parafunctional habits, erythema, irritation, glossitis, atrophy of oral mucosa or tongue, mucosal ulceration or other abnormalities.
Medical history of the patients and laboratory investigation are also essential to exclude any possible local and systemic conditions causing oral burning [ref. 1]. For instance, there is a range of medications acting as potential triggers for BMS-like symptoms, such as antihypertensive drugs, particularly angiotensin-converting enzyme (ACE) inhibitors or antiretrovirals [ref. 100, ref. 101]. It is possible to perform challenge, dechallenge, and rechallenge to determine if a medication has caused the burning sensation. This procedure is useful for establishing a causal relationship between the intake of a specific medication and the onset of burning symptoms, providing a structured methodology for identifying pharmacological triggers. A thorough medical history is necessary to assess if the burning sensation started after the introduction of a new medication, and if so, to proceed with this methodology. A detailed analysis of the pain’s characteristics—such as its onset, duration, location, and any factors that may exacerbate or alleviate it—and the presence of extraoral symptoms (medically unexplained extraoral physical symptoms) is crucial. This analysis should employ both qualitative and quantitative pain assessment tools (Table 4).
Table 4.: Characteristic assessment of pain in BMS patients.
| Site | Diffuse across oral mucosa. |
| Bilateral or unilateral | |
| Localized on tongue or other oral mucosa sites | |
| Onset | Spontaneous or triggered burning/pain |
| Triggers | Dental treatment |
| Stressful life events | |
| New medications | |
| Illness | |
| COVID-19 or vaccines | |
| Character | Burning, Tender, Hot, Stinging, Scalding, Numbness, Discomfort, Raw, Unpleasant, Annoying |
| Superficial oral mucosa pain | |
| Intensity | Moderate, Severe |
| Time/Duration | Pain/burning >2 hours a day. |
| Occurs more than 3 months | |
| Pattern of Pain | Continuous, Intermittent |
| Same morning/afternoon/evening | |
| Worst in afternoon/evening | |
| Worst in morning | |
| Exacerbating Factors | Eating, chewing, or drinking |
| Relieving Factors | Eating, contact with certain foods or drinks |
| Additional Oral Symptoms | Xerostomia |
| Dysgeusia | |
| Intraoral Foreign Body Sensation or cenesthopathy | |
| Globus Pharyngeus | |
| Discerptions (color alterations: white tongue, red areas bullae and morphological alterations: swellings, dysmorphisms, teeth enlargements) | |
| Sialorrhea | |
| Itching | |
| Tingling sensation | |
| Occlusal Dysesthesia | |
| Oral dyskinesia | |
| Dysosmia | |
| Subjective Halitosis | |
| Psychological comorbidity and Feelings that amplify symptomatology | Anxiety |
| Depression | |
| Sleep disturbance (Trouble falling or staying asleep, or sleeping too much) | |
| Loneliness | |
| Stress | |
| Personality disorders | |
| Obsessive-compulsive disorder (OCD) | |
| Hypochondria (Worries about complaints) | |
| Not being able to stop or control worrying | |
| Lack of time/work-related stress/caring responsibilities/finances | |
| Lack of support/interpersonal conflicts | |
| Different opinions from caregivers/not been taken seriously | |
| Little interest or pleasure in doing things | |
| Feeling down, depressed, or hopeless | |
| Medically Unexplained Extraoral Symptoms | Irritable bowel syndrome |
| Fibromyalgia | |
| Chronic fatigue syndrome | |
| Tinnitus | |
| Ophthalmodynia | |
| Skin burning/itching | |
| Vulvodynia | |
| Functional dyspepsia | |
| Tensive Headache | |
| Dizziness | |
| Restless legs syndrome | |
| Ear itching | |
| Nasal itching/Burning | |
| Low Back pain | |
| Myofascial pain | |
| Anal itching | |
| Asthenia | |
| Premature Ejaculation |
This table summarizes key characteristics of pain and associated symptoms in Burning Mouth Syndrome (BMS) patients. It is intended to guide comprehensive clinical assessment, helping to document pain features, triggers, time course, exacerbating/relieving factors, additional oral and extraoral symptoms, and psychological comorbidities. A multidimensional evaluation of these aspects supports accurate diagnosis and personalized management.
The quality of pain is often described as burning, tender, tingling, hot, stinging, scalding, numbness, discomfort, raw, unpleasant, or annoying. The intensity of pain varies from mild to severe. BMS presents as a predominantly burning, neuropathic pain with a diurnal increase in intensity, and while initially challenging, it can be successfully managed in most cases through a collaborative, empathetic approach involving the other medical colleagues [ref. 29, ref. 102]. BMS patients generally report oral burning/pain that diffuse to the entire oral mucosa or restricted to the tongue, but it can also affect buccal mucosa, lip, floor of the mouth, gingiva, and palate or rarely burning may be unilateral. When pain is unilateral, it is essential to differentiate BMS from other orofacial pain conditions, such as persistent idiopathic facial pain (PIFP), persistent idiopathic dentoalveolar pain (PIDAP), and trigeminal neuralgia (TN) (Table 5, Ref. [ref. 1]).
Table 5.: Differential diagnosis of BMS from other chronic oral pain conditions based on ICOP 1st edition, 2020 [ref. 1].
| Burning Mouth Syndrome (BMS) | Burning, tender, tingling, hot, stinging, scalding, numbness, discomfort, raw, unpleasant, or annoying | Mild to severe | Diffuse to the entire oral mucosa or restricted to the tongue; can affect buccal mucosa, lip, floor of the mouth, gingiva, and palate; rarely unilateral | (A) Oral pain fulfilling criteria B and C (B) Recurring daily for >2 h per day for >3 mon (C) Pain has both of the following characteristics: (1) burning quality (2) felt superficially in the oral mucosa (D) Oral mucosa is of normal appearance, and local or systemic causes have been excluded |
| Trigeminal Neuralgia (TN) | Sharp, stabbing, electric shock-like, abrupt in onset and termination | Severe | Unilateral, usually affecting one side of the face, limited to the distribution of one or more divisions of the trigeminal nerve and triggered by innocuous stimuli | (A) Recurrent paroxysms of unilateral facial pain in the distribution(s) of one or more divisions of the trigeminal nerve, with no radiation beyond, and fulfilling criteria B and C (B) The pain has all the following characteristics: (1) lasting from a fraction of a second to 2 minutes (2) severe intensity (3) electric shock-like, shooting, stabbing or sharp in quality (C) Precipitated by innocuous stimuli within the affected trigeminal distribution |
| Persistent Idiopathic Facial Pain (PIFP) | Dull, aching, or burning | Mild to severe | Unilateral or bilateral, often vague and poorly localized | (A) Facial pain fulfilling criteria B and C: (B) Recurring daily for >2 h/d for >3 mon (C) Pain has both of the following characteristics: (1) poorly localized, and not following the distribution of a peripheral nerve (2) dull, aching or nagging quality (D) Clinical and radiographic examinations are normal, and local causes have been excluded |
| Persistent Dentoalveolar Pain (PIDAP) | Persistent, aching, sometimes throbbing | Mild to moderate | Localized to the teeth or alveolar processes | (A) Intraoral dentoalveolar pain fulfilling criteria B and C (B) Recurring daily for >2 h/d for >3 mon (C) Pain has both of the following characteristics: (1) localized to a dentoalveolar site (tooth or alveolar bone) (2) deep, dull, pressure-like quality (D) Clinical and radiographic examinations are normal, and local causes have been excluded |
The early onset of the disease is characterized by a less severe symptomatology in the morning, getting worse during the day, although generally alleviated by eating and drinking, but in a long-standing disease these criteria may be not respected [ref. 1]. Frequently, most patients report additional symptoms, such as xerostomia, alteration in taste perception, phantom taste, alteration in sensory perception, globus, and others that may differ between patients, resulting in a varying clinical presentation [ref. 5, ref. 10, ref. 36]. The characteristics of symptomatology are reported in Table 4. The additional symptoms frequently complicate the disease and contribute to diagnostic delay because many instrumental exams and evaluations by other specialists are required to exclude possible related local and systemic conditions with the same symptoms. Therefore, BMS may remain under-recognized and under-appreciated by both dental and medical professionals for a long time. It took an average of 30 months from the onset of symptoms until a definitive diagnosis of BMS was achieved and every patient consulted almost three specialists, receiving several misdiagnoses before a proper diagnosis [ref. 5]. Additionally, the somatosensory screening tests may be considered to assess sensory nerve dysfunction in BMS patients. A thorough assessment should also include the metabolic profile evaluation through hematological tests, to rule out the nutritional, hormonal, autoimmune, and thrombophilia conditions that may underlie or contribute to symptoms. The laboratory tests required in BMS patients are available in (Table 6).
Table 6.: Laboratory investigations in BMS patients.
| Complete Blood Count (CBC) | White Blood Count (WBC), Red Blood Count (RBC), Platelet Count (Plt), Hemoglobin, Hematocrit, Mean corpuscular volume (MCV) |
| Glucose Metabolism | Glucose, Glycated hemoglobin (Hemoglobin A1C) |
| Kidney Function | Blood urea, Creatinine |
| Lipid Profile | Total cholesterol, High-density lipoprotein (HDL), Triglycerides, Low-density lipoprotein (LDL) |
| Iron Studies | Serum Iron, Serum Ferritin, Transferrin |
| Electrolytes | Sodium, Potassium, Magnesium |
| Liver Function Tests | Aspartate Phosphatase (AST or SGOT), Alanine Aminotransferase (ALT or SGPT), Alkaline Phosphatase (ALP), Total Bilirubin, Direct Bilirubin, Indirect Bilirubin, Albumin |
| Protein Studies | Protein electrophoresis serum test |
| Thyroid Panel | Thyroid stimulating hormone (TSH), Free T3 and T4 thyroid levels |
| Immune Panel | Antinuclear Antibodies (ANA), Extractable Nuclear Antigen Antibodies (ENA; anti-SSA and anti-SSB), C-reactive protein (CRP) test |
| Hormonal Tests | Prolactin, oestradiol, testosterone |
| Nutritional Deficiency/Vitamin Levels | Vitamin B12, B1, B6 test, Vitamin D test, Zinc |
| Urine Test | Routine Urinalysis |
| Coagulation Profile | PT, PTT & INR, Fibrinogen |
| Other Specific Test | |
| Homocysteine, Folate | |
| NSE (Neuron Specific Enolase) | |
| Hypercoagulable Workup | Protein C activity, Protein S activity Plasma antithrombin III; Anti-β2-glycoprotein I antibodies (antiβ2GPI; IgM and IgG); Anticardiolipin antibodies (ACA; IgM and IgG); Lupus anticoagulants (Las) |
| Genetic tests to assess for thrombophilia | MTHFR C677T: To check for a mutation that might lead to hyperhomocysteinemia, which could increase the risk of clotting disorders. Fattore V Leiden (Factor V Leiden): To detect a specific gene mutation that leads to a higher chance of forming abnormal blood clots. Fattore II or Prothrombin G20210A: To identify a mutation in the prothrombin gene that can lead to an increased risk of thrombosis. MTHFR A1298C: Similar to the C677T mutation, to check for another common variant that may affect homocysteine levels and potentially lead to clotting issues. PAI 1 GG/5G: To identify polymorphisms in the plasminogen activator inhibitor-1 (PAI-1) gene that could be linked to thrombotic disorders. Factor V Y1702: This appears to be a less common genetic test and may refer to a specific mutation within the Factor V gene related to clotting risk, similar to Factor V Leiden. Factor V HR2 (H1299R): A test for another mutation in the Factor V gene, which can be associated with an increased risk of venous thromboembolism. |
AST or SGOT: Aspartate Aminotransferase or Serum Glutamic-Oxaloacetic Transaminase; ALT or SGPT: Alanine Aminotransferase or Serum Glutamic-Pyruvic Transaminase; anti-SSA: Anti-Sjögren’s-syndrome-related antigen A (Ro) antibody; anti-SSB: Anti-Sjögren’s-syndrome-related antigen B (La) antibody; PT: Prothrombin Time; PTT: Partial Thromboplastin Time; INR: International Normalized Ratio; ACA: Anticardiolipin Antibody; IgM: Immunoglobulin M; IgG: Immunoglobulin G; MTHFR: Methylenetetrahydrofolate Reductase.
Comprehensive diagnostic screening for nutritional deficiencies and anemia is imperative because deficits in ferritin, iron, vitamin B12, folate, and zinc are often present in iron-deficiency anemia and vitamin B12-deficiency anemia. These conditions can lead to a secondary form of oral burning. Therefore, when deficiencies are identified, targeted treatment to correct them is crucial. This typically involves supplementation and dietary modifications to replenish the deficient micronutrients [ref. 92]. Upon correcting these deficiencies, a careful reassessment of the patient’s symptoms is necessary to determine whether the oral burning has resolved or improved. This step is important as it can help differentiate between a secondary burning related to nutritional deficits and BMS, where the symptoms may not respond to nutritional supplementation.
Similarly, hyperhomocysteinemia is a condition frequently observed in patients with BMS. It is often associated with folate deficiency, as well as vitamins B6 and B12, all of which are necessary for homocysteine metabolism [ref. 103]. Management of hyperhomocysteinemia includes supplementation with these vitamins, and it’s critical to monitor the patient’s response to treatment because hyperhomocysteinemia is a risk factor for various neurodegenerative and vascular brain pathologies, including increased stroke risk, cognitive decline, dementia, and mood disorders through mechanisms such as oxidative stress, endothelial dysfunction, excitotoxicity, and impaired methylation processes [ref. 104, ref. 105, ref. 106, ref. 107]. Specifically, testing for mutations in the Methylenetetrahydrofolate Reductase (MTHFR) gene (C677T and A1298C) is advised in patients with hyperhomocysteinemia exhibiting suboptimal responses to vitamin B supplementation. Such mutations can affect the metabolism of homocysteine and, by extension, influence the patient’s response to treatment.
Additionally, when an MRI of the brain reveals a high burden of WMHs that cannot be accounted for by common risk factors, such as hypertension or aging, genetic testing may uncover a predisposition to vascular disorders that impair cerebral blood flow. This is particularly relevant in patients with a history of deep vein thrombosis, pulmonary embolism, pre-eclampsia, or recurrent miscarriages, where an underlying thrombophilic condition might exist.
In all cases, genetic tests should be considered as part of a comprehensive evaluation that includes a detailed clinical examination, patient’s medical history, and if necessary, genetic counseling. It is crucial to interpret the results within the overall clinical context of the patient and not in isolation. A Comprehensive Evaluation of Coagulation Parameters in the context of BMS is meticulously detailed in the Tables 6 and 7, with a particular focus on the modifications of test results in patients who are undergoing treatment with antiplatelet agents and blood thinners, which are commonly prescribed for individuals with BMS.
Table 7.: Comprehensive evaluation of coagulation parameters in the context of BMS: detailed insights and therapeutic considerations.
| Prothrombin Time (PT) | Indicates blood clotting is slower than normal, potential bleeding disorder or anticoagulant use | Indicates blood clotting is faster than normal, potential risk of thrombosis | No direct effect | Prolonged PT is expected with warfarin therapy |
| Activated Partial Thromboplastin Time (aPTT) | Indicates blood takes longer to clot, may suggest bleeding disorder, factor deficiency, or anticoagulant use | Not typically clinically significant | No direct effect | Prolonged aPTT may occur with heparin therapy |
| Fibrinogen | Indicates acute phase reactant response, may suggest inflammation, infection, or tissue injury | Possible risk of bleeding disorders or fibrinolytic states | No direct effect | No direct effect |
| Homocysteine | Increased risk of cardiovascular disease and thrombosis | Not typically clinically significant | No direct effect | No direct effect |
| Folate | Generally not clinically significant, but can indicate supplementation or dietary intake | Possible deficiency, risk of anemia, other hematological disorders, and associated with hyperhomocysteinemia | No direct effect | No direct effect |
| Protein C Activity | Increased risk of bleeding (rarely observed) | Increased risk of thrombosis | No direct effect | No direct effect |
| Protein S Activity | Increased risk of bleeding (rarely observed) | Increased risk of thrombosis | No direct effect | No direct effect |
| Antithrombin III | Possible non-specific increase, not typically clinically significant | Increased risk of thrombosis | No direct effect | No direct effect |
| Anti-β2-glycoprotein I Antibodies (IgM and IgG) | May indicate an autoimmune condition such as antiphospholipid syndrome | Negative or low values typically do not indicate an increased risk of thrombosis | No direct effect | No direct effect |
| Anticardiolipin Antibodies (IgM and IgG) | May indicate an autoimmune condition such as antiphospholipid syndrome | Negative or low values typically do not indicate an increased risk of thrombosis | No direct effect | No direct effect |
| Lupus Anticoagulants | May indicate an autoimmune condition such as antiphospholipid syndrome or other coagulation disorders | Negative or low values typically do not indicate an increased risk of autoimmune conditions | No direct effect | May interfere with test, especially in patients on warfarin or heparin |
This panel of coagulation tests is proposed to identify potential prothrombotic states or autoimmune abnormalities (e.g., antiphospholipid antibodies), which may contribute to microvascular alterations hypothesized in the pathogenesis of Burning Mouth Syndrome (BMS). Their use is intended to guide individualized diagnostic workup, particularly in patients with atypical clinical features or risk factors. IgM: Immunoglobulin M; IgG: Immunoglobulin G.
Recently, Kishore and collaborators have proposed using NSE as a novel biomarker for peripheral neuropathies. Found in neurons and neuroendocrine cells, NSE plays a crucial role in glycolysis. Its utility in clinical diagnostics lies in its ability to indicate neuronal damage, making it a valuable tool for assessing various neuropathic conditions. Incorporating NSE measurement into the screening process for peripheral neuropathies allows clinicians to detect early signs of neuronal damage, thereby facilitating timely intervention and management [ref. 47].
Clinicians should perform an electrocardiogram (ECG) before starting pharmacological treatment, especially when prescribing psychotropic drugs. The ECG helps to identify cardiovascular risks, such as prolonged QT interval, conduction disorders, or cardiac ischemia, which may affect treatment decisions. QTc prolongation (normal: <430 ms in men, <450 ms in women; borderline: 430–450 ms in men, 450–470 ms in women; prolonged: >450 ms in men, >470 ms in women) is often drug-induced and is a known risk factor for torsades de pointes, a potentially fatal arrhythmia [ref. 108].
Many psychiatric drugs, including some antipsychotics and antidepressants, are known to potentially cause QT interval prolongation. A baseline ECG can establish whether the patient has a prolonged QT interval before treatment initiation, allowing the physician to make informed decisions regarding the prescription. The incidence of this adverse event is unpredictable, but a common observation is that most patients have at least one identifiable risk factor in addition to antidepressants exposure, such as female gender, age older than 60 years, hypokalemia, and a previous diagnosis of cardiovascular disease [ref. 109]. Therefore, the Food and Drug Administration recommends evaluating periodic ECG monitoring in all patients who begin treatment with these drugs with monitoring of the electrolytes, especially potassium [ref. 110]. Following this initial assessment, imaging techniques, particularly MRI of the brain, the brain stem, and the maxillofacial area should be employed as supplementary tools. MRI of the brain and brain stem can help identify any central nervous system pathology, such as WMHs [ref. 36] or space-occupying lesions [ref. 111] which are vital for a comprehensive understanding of the patient’s condition.
For cases presenting with unilateral and localized burning sensations on the gingiva, orthopantomography (OPT) and Cone Beam Computed Tomography (CBCT) of the jaw are recommended. These imaging modalities are essential for ruling out dental causes of pain, including local infections of the jaw, which might mimic or exacerbate BMS symptoms. Further, in the assessment of BMS, particularly when patients report symptoms of dry mouth, it is crucial to include comprehensive immunological testing as part of the diagnostic workup. The assessment should encompass Antinuclear Antibodies (ANA), Extractable Nuclear Antigen Antibodies (ENA; anti-SSA: Anti-Sjögren’s-syndrome-related antigen A; anti-SSB: Anti-Sjögren’s-syndrome-related antigen B), and C-reactive protein (CRP) levels. Elevated levels of these markers can be instrumental in unveiling underlying immunological conditions, notably Sjogren’s Syndrome. Routine ANA testing is not recommended in BMS patients without xerostomia or other clinical signs suggestive of systemic autoimmune disease. Targeted ANA testing may be considered in selected patients with additional risk factors or symptoms [ref. 112]. The presence of these antibodies, alongside raised CRP levels, points to an autoimmune process that may be contributing to the patient’s symptoms, necessitating further evaluation. Imaging studies of the salivary glands, such as sialography, scintigraphy, salivary gland ultrasonography, or MRI of the salivary glands, become essential in this context. These imaging modalities offer valuable insights into glandular structure and functionality, helping to distinguish between objective xerostomia—where there is a quantifiable decrease in salivary flow—and subjective xerostomia, where patients perceive dryness without a measurable reduction in saliva production [ref. 113, ref. 114].
In patients that report globus or exhibit erythematous lesions of the soft palate or dental erosions, these may indicate an underlying GERD. In such cases, a consultation with a gastroenterologist is mandatory to address and manage the potential reflux contributing to BMS [ref. 81, ref. 85]. By adhering to this structured diagnostic approach, healthcare professionals can ensure a comprehensive evaluation of BMS, addressing both the primary symptoms and any underlying or contributing conditions. This meticulous attention to detail ensures that treatment strategies are well-informed and tailored to the individual needs of the patient, thereby improving outcomes and patient satisfaction.
10. Multidimensional clinical assessment of BMS: insights from IMMPACT
In the early 2000s, the international pain research community identified a critical gap: the lack of standardized outcome measures in chronic pain studies. This led to the establishment of the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) [ref. 115], which introduced a set of core outcome domains designed to capture the multidimensional nature of pain—including its intensity, as well as its physical, emotional, and social impacts. While IMMPACT recommendations were originally developed for clinical trials, they can also guide and enrich the clinical evaluation of complex pain conditions such as BMS, promoting a more comprehensive understanding of each patient’s experience and needs.
This is not intended as a recommendation to routinely administer all the listed questionnaires to every patient. Rather, it encourages clinicians to adopt a flexible, patient-centered approach, choosing appropriate tools based on clinical judgment and the patient’s presentation. Table 8 summarizes validated instruments used in research and selectively applicable in clinical settings to deepen the assessment of BMS-related impairment.
Table 8.: Integrated assessment tools for comprehensive evaluation of pain and psychological profile in BMS patients.
| Domain 1 | ||||
| VAS | Visual Analogue Scale | Pain Intensity | A 10 cm line representing pain intensity, with the left end indicating no pain and the right end indicating the worst possible pain. Patients mark their pain level on the line. Not suitable for patients with cognitive or physical impairments. | |
| NRS | Numeric Rating Scale | Pain Intensity | An 11-point scale for pain intensity, from 0 (no pain) to 10 (the most terrible pain imaginable). Patients verbally choose or circle the number representing their pain. Practical, simple, and requires no visuo-motor coordination. | |
| GCPS | Graded Chronic Pain Scale | Pain intensity | Consists of three scales that assess pain intensity from 0 to 10, estimating current pain, worst pain, and average pain intensity over the last 30 days. | |
| Domain 2 | ||||
| PCS | Pain Catastrophizing Scale | Impact of pain and quality | To assess thoughts and feelings that contribute to the amplification of pain perception. | |
| BPI | Brief Pain Inventory | Impact of Pain and quality | To evaluate how pain affects various aspects of daily life and its severity. | |
| SF-MPQ | Short Form of McGill Pain Questionnaire | Impact of Pain and quality | To assess different qualities of pain experience (e.g., throbbing, shooting, stabbing, etc.). | |
| PDQ | PainDetect Neuropathic Pain Questionnaire | Impact of Pain and quality | To identify neuropathic pain components in patients’ pain experience. | |
| DN4 | Screening Questionnaire for Neuropathic Pain | Impact of Pain and quality | To screen neuropathic pain through symptom checklist. | |
| PVAQ | Pain Vigilance and Awareness Questionnaire | Impact of Pain and quality | To evaluate individual’s attention to and awareness of pain and how the patient perceive and focus on their pain. | |
| Domain 3 | ||||
| HADS | Hospital Anxiety and Depression Scale | Emotional Impact of Pain | To evaluate levels of anxiety and depression that may accompany chronic pain conditions. | |
| SCL-90 R | Symptom Checklist-90-Revised | Emotional Impact of Pain | To evaluate a broad range of psychological problems and symptoms of psychopathology. | |
| POMS | Profile of Mood States | Emotional Impact of Pain | To assess transient, distinct mood states: Tension-Anxiety, Depression-Dejection, Anger-Hostility, Vigor-Activity, Fatigue-Inertia, and Confusion-Bewilderment. | |
| PHQ-9 | Patient Health Questionnaire-9 | Emotional Impact of Pain | To screen for depression severity, which can be impacted by chronic pain. | |
| PHQ-4 | Patient Health Questionnaire-4 | Emotional Impact of Pain | To screen for anxiety and depression. | |
| BAI-BDI | Beck Anxiety and Depression Inventories | Emotional Impact of Pain | To assess the severity of anxiety and depression. | |
| DEPS | Depression Scale | Emotional Impact of Pain | To screen for symptoms of depression. | |
| QIDS-SR16 | 16-item Quick Inventory of Depressive Symptomatology | Emotional Impact of Pain | To measure the severity of depressive symptoms. | |
| PASS | Pain Anxiety Symptom Scale | Emotional Impact of Pain | To assess anxiety specifically related to pain. | |
| POMS | Profile of Mood States | Emotional Impact of Pain | To assess transient, distinct mood states in patients with various medical conditions. | |
| HAM-D | Hamilton Depression Rating Scale | Emotional Impact of Pain | To assess the severity of depression. | |
| HAM-A | Hamilton Anxiety Rating Scale | Emotional Impact of Pain | To evaluate the severity of anxiety symptoms. | |
| Z-SRDS | Zung Self-Rating Depression Scale | Emotional Impact of Pain | To measure levels of depression. | |
| GAD-7 | General Anxiety Disorder-7 | Emotional Impact of Pain | To assess anxiety severity, often heightened in chronic pain conditions. | |
| ASI | Anxiety Sensitivity Index | Emotional Impact of Pain | To measure the extent to which individuals are concerned about anxiety symptoms. | |
| STAI-Y2eY2 | State-Trait Anxiety Inventory | Emotional Impact of Pain | Differentiates between the temporary condition of “state anxiety” and the more general and long-standing quality of “trait anxiety”. | |
| GHQ-12 | 12-Item General Health Questionnaire | Emotional Impact of Pain General mental Health status | To screen for psychiatric disorders and assesses overall mental health. | |
| DASS-21 | Depression Anxiety Stress Scales-21 | Emotional Impact of Pain | To measure the severity of depression, anxiety, and stress highlighting emotional distress in individuals. | |
| IES-R-6 | Impact of Event Scale-Revised | Emotional Impact of Pain Post-Traumatic Stress Symptoms | To assess symptoms of post-traumatic stress, focusing on specific aspects such as intrusive thoughts. | |
| PTGI-SF | Post Traumatic Growth Inventory Short Form | Emotional Impact of Pain Post-Traumatic Growth | To measure the degree of personal growth or positive psychological change following trauma. | |
| CDRS-10 | Connor-Davidson Resilience Scale | Emotional Impact of Pain Resilience | To evaluate levels of resilience, indicating an individual’s ability to cope with and bounce back from adversity. | |
| ULS-8 | Short-form UCLA Loneliness Scale-8 | Emotional impact of pain Loneliness | To measure feelings of loneliness and social isolation. | |
| MSPSS | Multidimensional Scale of Perceived Social Support | Emotional impact of pain Social Support | To assess the perception of social support from family, friends, and significant others. | |
| SIDAS | Suicidal Ideation Attributes Scale | Emotional impact of pain Suicidal Thoughts | To measure the frequency and intensity of suicidal ideation. | |
| NEO PI-R | NEO Personality Inventory-Revised | Emotional impact of pain Personality | To measure five dimensions of personality (Neuroticism, Extraversion, Openness, Agreeableness, Conscientiousness) and their facets. | |
| MMPI | Minnesota Multiphasic Personality Inventory | Emotional impact of pain Personality | Used to identify personality and psychosocial disorders, revealing characteristics in BMS patients similar to those in chronic pain patients. | |
| SCID-II | Structured Clinical Interview for DSM-IV Axis II Personality Disorders | Emotional impact of pain Personality | To evaluate personality disorders and split in Cluster A Cluster B. | |
| EPQ-RSC | Eysenck Personality Questionnaire-Revised Short Scale for Chinese | Emotional impact of pain Personality | To evaluate mental status, and level of anxiety and depression. | |
| MCMQ | Medical Coping Modes Questionnaire | Emotional impact of pain Coping Mechanisms | To measure 3 cognitive-behavioral, illness-related coping strategies | |
| TIPI | Ten-Item Personality Inventory | Emotional Impact of pain Personality | To explore the impact of personality on pain and related experiences, highlighting neuroticism’s significant association with pain symptoms. | |
| OCI | Obsessive-Compulsive Disorder | Emotional Impact of pain Personality | To evaluate the symptoms of obsessive-compulsive disorder. | |
| C-PAS | Compulsive Personality Assesment Scale | Emotional Impact of pain Personality | To evaluate traits associated with compulsive personality disorder. | |
| PSQI | Pittsburgh Sleep Quality Index | Emotional Impact of Pain Sleep | To Evaluate sleep quality and disturbances, which can be affected by pain. | |
| ISI | Insomnia Severity Index | Emotional Impact of Pain Sleep | To Assess the severity of insomnia symptoms related to pain conditions. | |
| ESS | Epworth Sleepiness Scale | Emotional Impact of pain Sleep | To assess the subjective daytime sleepiness. | |
| SLP-6 e 9 | Sleep Problem Index | Emotional Impact of pain Sleep | To assess common sleep disturbances such as trouble sleeping, nightmares and to evaluate sleep habits, such as sleep duration, difficulty waking up and daytime sleepiness. | |
| GOHAI | Global Oral Health Assessment Index | Impact on quality of life Oral Health Quality | To assess the impact of oral health on quality of life, focusing on functional, psychosocial, and pain/discomfort aspects. | |
| OHIP-14 | Oral Health Impact Profile | Impact on quality of life Oral Health Impact | To Measure the social and psychological effects of oral conditions on quality of life. | |
| OHR-QoL | Oral Health-Related Quality of Life | Impact on quality of life Oral Health Quality | To measure how oral health affects an individual’s quality of life. | |
| IPQ-R | Illness Perception Questionnaire | Impact on quality of life Illness Perceptions | To evaluate how patients perceive their illness, including beliefs about causes, consequences, and control/cure. | |
| HRQoL | Health-Related Quality of Life | Impact on quality of life General health status | To Assess how an individual’s health status affects their quality of life. | |
| GP-CORE | General Population-Clinical Outcomes in Routine Evaluation | Impact on quality of life General Health status | To monitor and evaluate a range of psychological issue, including well-being and life function in the general population. | |
| SF-36 | Short Form Health Survey 36 | Impact on quality of life General Health Status | A comprehensive measure of health status covering physical, emotional, and social well-being. | |
| EQ-5Q | EuroQol Questionnaire | Impact on quality of life General Health Status | To measure health-related quality of life. It consists of two parts: a descriptive system and the EQ visual analogue scale (EQ VAS). The descriptive system captures five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression, each with three levels of severity. | |
| GH-28 | General Health Questionnaire | Impact on quality of life General Mental health | To screen for psychiatric disorders and assesses overall mental health. | |
| PEG | 3-item Pain, Enjoyment, and General activity | Impact on quality of life Pain and General Well-being | To Assess pain intensity, interference with enjoyment of life, and interference with general activity. | |
| Domain 4 | ||||
| PGIC | Patient Global Impression of Change | Ratings of global improvement and satisfaction | To evaluate the overall change since the beginning of the treatment on a seven-point scale. | |
| CSQ | Client Satisfaction Questionnaire | Ratings of global improvement and satisfaction | To measure patient satisfaction with healthcare services. It typically consists of multiple items that explore the quality of care, the information provided, the provider’s behavior, and the overall healthcare experience. | |
| TSQM | The Treatment Satisfaction Questionnaire for Medication | Ratings of global improvement and satisfaction | To evaluate patients’ satisfaction with their medications exploring information about the patient’s experience with their treatment, focusing on several key aspects that affect their overall satisfaction and adherence to the prescribed regimen. | |
| CGI-S CGI-I CGI-E | Clinical global Severity of Illness Global Improvement; Efficacy Index | Ratings of global improvement and satisfaction | To evaluate the patient’s response to treatment and side effects based on a global overview of their condition. | |
| Domain 5 | ||||
| AEQ | Adverse Events Questionnaire | Symptoms and Adverse Events associated with medical treatments | To Collect patient-reported adverse events or side effects from treatment. | |
| UKU SERS | UKU Side Effect Rating Scale | Symptoms and Adverse Events associated with medical treatments | To assess side effects of psychotropic medicationsIt includes a clinician-rated version and a self-rating version for patients. | |
| ASEC | Antidepressant Side-Effect Checklist | Symptoms and Adverse Events associated with medical treatments | To assess side effects of psychotropic medications. | |
| PROMISE | Patient Reported Outcome Measure, Inquiry into Side-Effects | Symptoms and Adverse Events associated with medical treatments | To evaluate and monitor symptoms and adverse events associated with medical treatments. It provides a standardized set of measures that can assess physical, mental, and social health from the patient’s perspective. | |
| Domain 6 | ||||
| PMQ | The Pain Medication Questionnaire | Adherence of treatment and discontinuation | It specifically targets patients who are prescribed pain medications and explore patients’ beliefs about pain medication, their adherence to pain management regimens, their knowledge about their pain medication, and behaviors that might suggest misuse or abuse. | |
| TSQM | Treatment Satisfaction Questionnaire for Medication | Adherence of treatment and discontinuation | To understand the patient’s perspective on the medication they are receiving. This questionnaire can help to predict medication adherence, as greater satisfaction with medication is often linked to better adherence. | |
| BMQ | The Brief Medication Questionnaire | Adherence of treatment and discontinuation | To identify potential issues with medication adherence among patients. It’s designed to quickly gather information on a patient’s medication-taking behavior and barriers to adherence. | |
| MARS | Medication Adherence Report Scale | Adherence of treatment and discontinuation | To measure adherence to medication regimens. It contains a set of statements regarding medication-taking behaviors to which patients respond on a 4 or 5-point scale, with higher scores indicating better adherence. | |
This is a list of validated tools for the comprehensive assessment of pain and psychological profile in BMS patients. Tools are grouped by domain: pain intensity, pain impact and quality, emotional impact, global improvement and satisfaction, treatment-related adverse events, and treatment adherence.
IMMPACT recommends evaluating chronic pain patients across six core domains: pain intensity, physical functioning, emotional well-being, participant ratings of improvement and satisfaction with treatment, adverse events, and participant disposition (including adherence and reasons for treatment withdrawal). This framework is particularly relevant to BMS, where symptoms extend beyond pain intensity and may affect multiple aspects of life.
Suggested tools for each domain include:
• Pain Intensity (Domain 1): Numerical Rating Scale (NRS), Visual Analog Scale (VAS).
• Physical Functioning (Domain 2): Multidimensional Pain Inventory (MPI), Brief Pain Inventory (BPI), Pain Disability Index (PDI), which assess the impact of pain on activity, mood, relationships, sleep, and quality of life.
• Emotional Functioning (Domain 3): Hospital Anxiety and Depression Scale (HADS), Beck Depression Inventory (BDI), Profile of Mood States (POMS).
• Global Improvement and Satisfaction (Domain 4): Patient Global Impression of Change (PGIC).
• Symptoms and Adverse Events (Domain 5): Typically captured through clinical interviews and patient reports.
• Participant Disposition (Domain 6): Information on treatment adherence and reasons for discontinuation, gathered via structured interviews or questionnaires.
IMMPACT also highlights the importance of additional domains such as sleep disturbances, fatigue, and the emotional burden of chronic pain, which—although not part of the core domains—are highly relevant for BMS patients. Moreover, emerging evidence suggests a potential link between cognitive impairment and BMS, supporting the integration of cognitive screening into the clinical evaluation. Instruments such as the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) provide valuable screening for cognitive deficits and can guide the need for more comprehensive neuropsychological evaluation when indicated. Incorporating selected IMMPACT-based assessments and cognitive screening into routine BMS management fosters a holistic understanding of the condition, enabling more personalized treatment planning and improving quality of life.
11. Conclusions
BMS is a painful condition that necessitates careful evaluation for accurate diagnosis. It is essential to differentiate BMS from other orofacial pain conditions particularly when the pain is unilateral. Additionally, it is crucial to distinguish primary BMS from secondary forms of burning sensations. This distinction is vital as each of these conditions may require different therapeutic approaches. An accurate diagnosis involves considering a range of diagnostic tools including MRI of the brain, blood tests, and cognitive and psychological assessments. These evaluations help to provide a comprehensive understanding of the patient’s condition, leading to appropriate treatment, improved pain management, and enhanced quality of life.
References
- [1] International classification of orofacial pain, 1st edition (ICOP). Cephalalgia. 2020; 40: 129–221.
- [2] Fedele S, Fricchione G, Porter SR, Mignogna MD. Burning mouth syndrome (stomatodynia). QJM: An International Journal of Medicine. 2007; 100: 527–530.
- [3] Sullivan PD. The diagnosis and treatment of psychogenic glossodynia. Ear, Nose & Throat Journal. 1989; 68: 795–798.
- [4] Fortuna G, Di Lorenzo M, Pollio A. Complex oral sensitivity disorder: a reappraisal of current classification of burning mouth syndrome. Oral Diseases. 2013; 19: 730–732.
- [5] Adamo D, Calabria E, Canfora F, Coppola N, Pecoraro G, D’Aniello L, et al. Burning mouth syndrome: analysis of diagnostic delay in 500 patients. Oral Diseases. 2024; 30: 1543–1554.
- [6] Chmieliauskaite M, Stelson EA, Epstein JB, Klasser GD, Farag A, Carey B, et al. Consensus agreement to rename burning mouth syndrome and improve ICD-11 disease criteria: an international Delphi study. Pain. 2021; 162: 2548–2557.
- [7] The Lancet. ICD-11. The Lancet. 2019; 393: 2275.
- [8] Musella G, Canfora F, Caponio VCA, Vardas E, Kouri M, Nikitakis N, et al. Oral dysaesthetic and perceptual disorder, a distinct subset of chronic orofacial pain without burning symptoms: a case-control study. Journal of Oral Rehabilitation. 2025; 52: 651–666.
- [9] Shinoda M, Kubo A, Hayashi Y, Iwata K. Peripheral and central mechanisms of persistent orofacial pain. Frontiers in Neuroscience. 2019; 13: 1227.
- [10] Adamo D, Spagnuolo G. Burning mouth syndrome: an overview and future perspectives. International Journal of Environmental Research and Public Health. 2022; 20: 682.
- [11] Seol SH, Chung G. Estrogen-dependent regulation of transient receptor potential vanilloid 1 (TRPV1) and P2X purinoceptor 3 (P2X3): implication in burning mouth syndrome. Journal of Dental Sciences. 2022; 17: 8–13.
- [12] Egido-Moreno S, Valls-Roca-Umbert J, Perez-Sayans M, Blanco-Carrión A, Jane-Salas E, López-López J. Role of thyroid hormones in burning mouth syndrome. Systematic review. Medicina Oral, Patologia Oral, Cirugia Bucal. 2023; 28: e81–e86.
- [13] Lee SJ, Kim C, Yu H, Kim DK. Relationship of depression, anxiety, and bipolar disease with burning mouth syndrome: a nationwide cohort study. International Journal of Environmental Research and Public Health. 2023; 20: 3391.
- [14] Wu S, Zhang W, Yan J, Noma N, Young A, Yan Z. Worldwide prevalence estimates of burning mouth syndrome: a systematic review and meta-analysis. Oral Diseases. 2022; 28: 1431–1440.
- [15] Kohorst JJ, Bruce AJ, Torgerson RR, Schenck LA, Davis MDP. The prevalence of burning mouth syndrome: a population-based study. British Journal of Dermatology. 2015; 172: 1654–1656.
- [16] Klasser GD, Fischer DJ, Epstein JB. Burning mouth syndrome: recognition, understanding, and management. Oral and Maxillofacial Surgery Clinics of North America. 2008; 20: 255–271, vii.
- [17] Ni Riordain R, O’Dwyer S, McCreary C. Burning mouth syndrome-a diagnostic dilemma. Irish Journal of Medical Science. 2019; 188: 731–734.
- [18] Kim JY, Kim YS, Ko I, Kim DK. Association between burning mouth syndrome and the development of depression, anxiety, dementia, and Parkinson disease. JAMA Otolaryngology—Head & Neck Surgery. 2020; 146: 561–569.
- [19] Kang JH, Kim YY, Chang JY, Kho HS. Relationships between oral MUC1 expression and salivary hormones in burning mouth syndrome. Archives of Oral Biology. 2017; 78: 58–64.
- [20] Gu Y, Baldwin S, Canning C. Hypovitaminosis D, objective oral dryness, and fungal hyphae as three precipitating factors for a subset of secondary burning mouth syndrome. Heliyon. 2023; 9: e19954.
- [21] Salort-Llorca C, Mínguez-Serra MP, Silvestre FJ. Drug-induced burning mouth syndrome: a new etiological diagnosis. Medicina Oral, Patologia Oral, Cirugia Bucal. 2008; 13: E167–E170.
- [22] Iwata K, Takeda M, Oh SB, Shinoda M. Neurophysiology of orofacial pain. In CS Farah, R Balasubramaniam, MJ McCullough (eds.) Contemporary oral medicine (pp. 1–23). Springer: Cham. 2017.
- [23] Kato S, Kurokawa R, Suzuki F, Amemiya S, Shinozaki T, Takanezawa D, et al. White and gray matter abnormality in burning mouth syndrome evaluated with diffusion tensor imaging and neurite orientation dispersion and density imaging. Magnetic Resonance in Medical Sciences. 2024; 23: 204–213.
- [24] Sinding C, Gransjøen AM, Schlumberger G, Grushka M, Frasnelli J, Singh PB. Grey matter changes of the pain matrix in patients with burning mouth syndrome. European Journal of Neuroscience. 2016; 43: 997–1005.
- [25] Lauria G, Majorana A, Borgna M, Lombardi R, Penza P, Padovani A, et al. Trigeminal small-fiber sensory neuropathy causes burning mouth syndrome. Pain. 2005; 115: 332–337.
- [26] Kolkka M, Forssell H, Virtanen A, Puhakka A, Pesonen U, Jääskeläinen SK. Neurophysiology and genetics of burning mouth syndrome. European Journal of Pain. 2019; 23: 1153–1161.
- [27] Meacham K, Shepherd A, Mohapatra DP, Haroutounian S. Neuropathic pain: central vs. peripheral mechanisms. Current Pain and Headache Reports. 2017; 21: 28.
- [28] Imamura Y, Ozasa K, Nishihara C, Asano S, Takanezawa D, Ikeda M, et al. Burning mouth syndrome and nociplastic pain. Japanese Journal of Oral Diagnosis/Oral Medicine. 2020; 33: 145–152. (In Japanese)
- [29] Gremeau-Richard C, Pionchon P, Mulliez A, Dualé C, Dallel R. Enhanced pain facilitation rather than impaired pain inhibition in burning mouth syndrome female patients. The Journal of Headache and Pain. 2022; 23: 143.
- [30] Kurokawa R, Kamiya K, Inui S, Kato S, Suzuki F, Amemiya S, et al. Structural connectivity changes in the cerebral pain matrix in burning mouth syndrome: a multi-shell, multi-tissue-constrained spherical deconvolution model analysis. Neuroradiology. 2021; 63: 2005–2012.
- [31] Hagelberg N, Forssell H, Rinne JO, Scheinin H, Taiminen T, Aalto S, et al. Striatal dopamine D1 and D2 receptors in burning mouth syndrome. Pain. 2003; 101: 149–154.
- [32] Wood PB. Role of central dopamine in pain and analgesia. Expert Review of Neurotherapeutics. 2008; 8: 781–797.
- [33] Khan SA, Keaser ML, Meiller TF, Seminowicz DA. Altered structure and function in the hippocampus and medial prefrontal cortex in patients with burning mouth syndrome. Pain. 2014; 155: 1472–1480.
- [34] Lee YC, Jahng GH, Ryu CW, Byun JY. Change in gray matter volume and cerebral blood flow in patients with burning mouth syndrome. Journal of Oral Pathology & Medicine. 2019; 48: 335–342.
- [35] Ong WY, Stohler CS, Herr DR. Role of the prefrontal cortex in pain processing. Molecular Neurobiology. 2019; 56: 1137–1166.
- [36] Adamo D, Canfora F, Calabria E, Coppola N, Leuci S, Pecoraro G, et al. White matter hyperintensities in burning mouth syndrome assessed according to the age-related white matter changes scale. Frontiers in Aging Neuroscience. 2022; 14: 923720.
- [37] Jääskeläinen SK, Woda A. Burning mouth syndrome. Cephalalgia. 2017; 37: 627–647.
- [38] Puhakka A, Forssell H, Soinila S, Virtanen A, Röyttä M, Laine M, et al. Peripheral nervous system involvement in primary burning mouth syndrome—results of a pilot study. Oral Diseases. 2016; 22: 338–344.
- [39] Moura B de S, Ferreira NDR, DosSantos MF, Janini MER. Changes in the vibration sensitivity and pressure pain thresholds in patients with burning mouth syndrome. PLOS ONE. 2018; 13: e0197834.
- [40] Kolkka-Palomaa M, Jääskeläinen SK, Laine MA, Teerijoki-Oksa T, Sandell M, Forssell H. Pathophysiology of primary burning mouth syndrome with special focus on taste dysfunction: a review. Oral Diseases. 2015; 21: 937–948.
- [41] Kolkka M, Jääskeläinen S, Forssell H, Suominen A, Teerijoki-Oksa T, Loimaranta V, et al. Taste perception and saliva composition are not altered in burning mouth syndrome. Oral Diseases. 2025; 31: 1317–1332.
- [42] Yilmaz Z, Renton T, Yiangou Y, Zakrzewska J, Chessell IP, Bountra C, et al. Burning mouth syndrome as a trigeminal small fibre neuropathy: increased heat and capsaicin receptor TRPV1 in nerve fibres correlates with pain score. Journal of Clinical Neuroscience. 2007; 14: 864–871.
- [43] Beneng K, Yilmaz Z, Yiangou Y, McParland H, Anand P, Renton T. Sensory purinergic receptor P2X3 is elevated in burning mouth syndrome. International Journal of Oral and Maxillofacial Surgery. 2010; 39: 815–819.
- [44] Ishida Y, Ugawa S, Ueda T, Yamada T, Shibata Y, Hondoh A, et al. P2X2- and P2X3-positive fibers in fungiform papillae originate from the chorda tympani but not the trigeminal nerve in rats and mice. Journal of Comparative Neurology. 2009; 514: 131–144.
- [45] García-Domínguez M. NGF in neuropathic pain: understanding its role and therapeutic opportunities. Current Issues in Molecular Biology. 2025; 47: 93.
- [46] Heiliczer S, Yanko R, Sharav Y, Aframian DJ, Klutstein M, Wilensky A, et al. Oxidative stress-mediated proapoptosis signaling: a novel theory on the mechanism underlying the pathogenesis of burning mouth syndrome. The Journal of the American Dental Association. 2024; 155: 258–267.
- [47] Kishore J, Shaikh F, Zubairi AM, Mirza S, Alqutub MN, AlMubarak AM, et al. Evaluation of serum neuron specific enolase levels among patients with primary and secondary burning mouth syndrome. Cephalalgia. 2022; 42: 119–127.
- [48] Toyama M, Kudo C, Mukai C, Inoue M, Oyamaguchi A, Hanamoto H, et al. Trigeminal nervous system sensitization by infraorbital nerve injury enhances responses in a migraine model. Cephalalgia. 2017; 37: 1317–1328.
- [49] Dieb W, Ouachikh O, Durif F, Hafidi A. Nigrostriatal dopaminergic depletion produces orofacial static mechanical allodynia. European Journal of Pain. 2016; 20: 196–205.
- [50] Taiminen T, Kuusalo L, Lehtinen L, Forssell H, Hagelberg N, Tenovuo O, et al. Psychiatric (axis I) and personality (axis II) disorders in patients with burning mouth syndrome or atypical facial pain. Scandinavian Journal of Pain. 2011; 2: 155–160.
- [51] Schiavone V, Adamo D, Ventrella G, Morlino M, De Notaris EB, Ravel MG, et al. Anxiety, depression, and pain in burning mouth syndrome: first chicken or egg? Headache. 2012; 52: 1019–1025.
- [52] Canfora F, Ottaviani G, Calabria E, Pecoraro G, Leuci S, Coppola N, et al. Advancements in understanding and classifying chronic orofacial pain: key insights from biopsychosocial models and international classifications (ICHD-3, ICD-11, ICOP). Biomedicines. 2023; 11: 3266.
- [53] Adamo D, Sardella A, Varoni E, Lajolo C, Biasotto M, Ottaviani G, et al. The association between burning mouth syndrome and sleep disturbance: a case-control multicentre study. Oral Diseases. 2018; 24: 638–649.
- [54] Alhendi F, Ko E, Graham L, Corby P. The association of sleep disturbances with burning mouth syndrome: an overlooked relationship—a qualitative systematic review. Oral Diseases. 2023; 29: 6–20.
- [55] Tan Y, Wu X, Chen J, Kong L, Qian Z. Structural and functional connectivity between the amygdala and orbital frontal cortex in burning mouth syndrome: an fMRI study. Frontiers in Psychology. 2019; 10: 1700.
- [56] Mbiydzenyuy NE, Qulu LA. Stress, hypothalamic-pituitary-adrenal axis, hypothalamic-pituitary-gonadal axis, and aggression. Metabolic Brain Disease. 2024; 39: 1613–1636.
- [57] Adamo D, Pecoraro G, Coppola N, Calabria E, Aria M, Mignogna M. Vortioxetine versus other antidepressants in the treatment of burning mouth syndrome: an open-label randomized trial. Oral Diseases. 2021; 27: 1022–1041.
- [58] Adamo D, Pecoraro G, Aria M, Favia G, Mignogna MD. Vortioxetine in the treatment of mood disorders associated with burning mouth syndrome: results of an open-label, flexible-dose pilot study. Pain Medicine. 2020; 21: 185–194.
- [59] Taheri N, Pirboveiri R, Sayyah M, Bijanzadeh M, Ghandil P. Association of DRD2, DRD4 and COMT genes variants and their gene-gene interactions with antipsychotic treatment response in patients with schizophrenia. BMC Psychiatry. 2023; 23: 781.
- [60] Echeverria-Villalobos M, Tortorici V, Brito BE, Ryskamp D, Uribe A, Weaver T. The role of neuroinflammation in the transition of acute to chronic pain and the opioid-induced hyperalgesia and tolerance. Frontiers in Pharmacology. 2023; 14: 1297931.
- [61] Jin H, Li M, Jeong E, Castro-Martinez F, Zuker CS. A body-brain circuit that regulates body inflammatory responses. Nature. 2024; 630: 695–703.
- [62] Al-Maweri SA, Javed F, Kalakonda B, AlAizari NA, Al-Soneidar W, Al-Akwa A. Efficacy of low level laser therapy in the treatment of burning mouth syndrome: a systematic review. Photodiagnosis and Photodynamic Therapy. 2017; 17: 188–193.
- [63] Moreau C, El Habnouni C, Lecron JC, Morel F, Delwail A, Le Gall-Ianotto C, et al. Salivary metabolome indicates a shift in tyrosine metabolism in patients with burning mouth syndrome: a prospective case-control study. Pain. 2023; 164: e144–e156.
- [64] Geerlings MI, Gerritsen L. Late-life depression, hippocampal volumes, and hypothalamic-pituitary-adrenal axis regulation: a systematic review and meta-analysis. Biological Psychiatry. 2017; 82: 339–350.
- [65] Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychological Bulletin. 2014; 140: 774–815.
- [66] Takeuchi T, Hashimoto K, Koyama A, Asakura K, Hashizume M. The association of central sensitisation with depression, anxiety, and somatic symptoms: a cross-sectional study of a mental health outpatient clinic in Japan. Life. 2024; 14: 612.
- [67] Latremoliere A, Woolf CJ. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. The Journal of Pain. 2009; 10: 895–926.
- [68] Ozasa K, Noma N, Young A, Korczeniewska OA, Eliav E, Imamura Y. Potential differences in somatosensory function during premenopause and early and late postmenopause in patients with burning mouth syndrome: an observational case-control study. The Journal of Dental Sciences. 2022; 17: 399–406.
- [69] Gao N, Li M, Wang W, Liu Z, Guo Y. The dual role of TRPV1 in peripheral neuropathic pain: pain switches caused by its sensitization or desensitization. Frontiers in Molecular Neuroscience. 2024; 17: 1400118.
- [70] Fernández-Agra M, González-Serrano J, de Pedro M, Virto L, Caponio VCA, Ibáñez-Prieto E, et al. Salivary biomarkers in burning mouth syndrome: a systematic review and meta-analysis. Oral Diseases. 2023; 29: 2600–2613.
- [71] Moisset X, Calbacho V, Torres P, Gremeau-Richard C, Dallel R. Co-occurrence of pain symptoms and somatosensory sensitivity in burning mouth syndrome: a systematic review. PLOS ONE. 2016; 11: e0163449.
- [72] Galli F, Lodi G, Sardella A, Vegni E. Role of psychological factors in burning mouth syndrome: a systematic review and meta-analysis. Cephalalgia. 2017; 37: 265–277.
- [73] Mignogna MD, Pollio A, Fortuna G, Leuci S, Ruoppo E, Adamo D, et al. Unexplained somatic comorbidities in patients with burning mouth syndrome: a controlled clinical study. Journal of Orofacial Pain. 2011; 25: 131–140.
- [74] Leuci S, Coppola N, Adamo D, Crocetto F, Barone B, Baldares S, et al. Sexual desire, mood disorders and sleep disturbances in female BMS patients: a controlled study. Journal of Oral Pathology & Medicine. 2023; 52: 276–282.
- [75] Jin JQ, Cui HM, Han Y, Su S, Liu HW. Multifactor analysis of patients with oral sensory complaints in a case-control study. Chinese Medical Journal. 2020; 133: 2822–2828.
- [76] Canfora F, Calabria E, Pecoraro G, Leuci S, Coppola N, Mazzaccara C, et al. Prevalence of hypertension and correlation with mental health in women with burning mouth syndrome: a case-control study. Frontiers in Cardiovascular Medicine. 2022; 9: 969148.
- [77] Calabria E, Canfora F, Adamo D. Chronic orofacial pain and white matter hyperintensities. Aging (Albany NY). 2023; 15: 1708–1710.
- [78] LeVasseur E, Fogel M, Khanna D. A study on the effects of hypothyroidism on the senses: a comprehensive narrative review. Cureus. 2024; 16: e65684.
- [79] Morr Verenzuela CS, Davis MDP, Bruce AJ, Torgerson RR. Burning mouth syndrome: results of screening tests for vitamin and mineral deficiencies, thyroid hormone, and glucose levels-experience at Mayo Clinic over a decade. International Journal of Dermatology. 2017; 56: 952–956.
- [80] Femiano F, Lanza A, Buonaiuto C, Gombos F, Nunziata M, Cuccurullo L, et al. Burning mouth syndrome and burning mouth in hypothyroidism: proposal for a diagnostic and therapeutic protocol. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics. 2008; 105: e22–e27.
- [81] Li L, Wu S, Noma N, Young A, Wang X, Yan Z. Relationship between burning mouth disorder and gastroesophageal reflux disease: a scoping review. Oral Diseases. 2024; 30: 3600–3609.
- [82] Aframian DJ, Ofir M, Benoliel R. Comparison of oral mucosal pH values in bulimia nervosa, GERD, BMS patients and healthy population. Oral Diseases. 2010; 16: 807–811.
- [83] Becker S, Schmidt C, Berghaus A, Tschiesner U, Olzowy B, Reichel O. Does laryngopharyngeal reflux cause intraoral burning sensations? A preliminary study. European Archives of Oto-Rhino-Laryngology. 2011; 268: 1375–1381.
- [84] Lechien JR, Hans S, De Marrez LG, Dequanter D, Rodriguez A, Muls V, et al. Prevalence and features of laryngopharyngeal reflux in patients with primary burning mouth syndrome. Laryngoscope. 2021; 131: E2627–E2633.
- [85] Russo M, Crafa P, Franceschi M, Rodriguez-Castro KI, Franzoni L, Guglielmetti S, et al. Burning mouth syndrome and reflux disease: relationship and clinical implications. Acta Biomedica. 2022; 93: e2022329.
- [86] Canfora F, Calabria E, Cuocolo R, Ugga L, Buono G, Marenzi G, et al. Burning fog: cognitive impairment in burning mouth syndrome. Frontiers in Aging Neuroscience. 2021; 13: 727417.
- [87] Dugan C, Parlatescu I, Dobre M, Pîrvu RE, Milanesi E. Insights on brain functions in burning mouth syndrome. Frontiers in Systems Neuroscience. 2022; 16: 975126.
- [88] Femminella GD, Canfora F, Musella G, Di Tella GS, Ugga L, Pecoraro G, et al. Cognitive profile in burning mouth syndrome versus mild cognitive impairment: a comparative study. Oral Diseases. 2025; 31: 611–632.
- [89] Canfora F, Cataldi M, Mignogna MD, Ottaviani G, Leuci S, Coppola N, et al. Blueprint persona and information and communication technology interventions: addressing unmet needs in burning mouth syndrome care. Journal of Evidence-Based Dental Practice. 2025; 25: 102047.
- [90] O G, Balasubramaniam R, Klasser GD. Burning mouth disorder and Parkinson’s disease: a scoping review of the literature. Journal of Oral Rehabilitation. 2023; 50: 488–500.
- [91] Prakash S, Ahuja S, Rathod C. Dopa responsive burning mouth syndrome: restless mouth syndrome or oral variant of restless legs syndrome? Journal of the Neurological Sciences. 2012; 320: 156–160.
- [92] Leng A, Shah M, Ahmad SA, Premraj L, Wildi K, Li Bassi G, et al. Pathogenesis underlying neurological manifestations of long COVID syndrome and potential therapeutics. Cells. 2023; 12: 816.
- [93] Mastrangelo A, Bonato M, Cinque P. Smell and taste disorders in COVID-19: from pathogenesis to clinical features and outcomes. Neuroscience Letters. 2021; 748: 135694.
- [94] The Lancet Psychiatry. COVID-19 and mental health. The Lancet Psychiatry. 2021; 8: 87.
- [95] Candela CF, Pia LJ, Pons-Fuster E, Tvarijonaviciute A. Impact of the COVID-19 pandemic upon patients with burning mouth syndrome. Journal of Stomatology Oral and Maxillofacial Surgery. 2022; 123: 101–104.
- [96] Ottaviani G, Canfora F, Leuci S, Coppola N, Pecoraro G, Rupel K, et al. COVID-19 impact on post-traumatic stress symptoms in burning mouth syndrome: a multicentric study. Oral Diseases. 2024; 30: 4653–4667.
- [97] Gobel H. The international classification of headache disorders 3rd edition. 2018. Available at: https://ichd-3.org/ (Accessed: 09 February 2022).
- [98] Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia. 2013; 33: 629–808.
- [99] Regier DA, Kuhl EA, Kupfer DJ. The DSM-5: classification and criteria changes. World Psychiatry. 2013; 12: 92–98.
- [100] Obara T, Naito H, Nojima T, Koga H, Nakao A. Burning mouth syndrome induced by angiotensin-converting enzyme inhibitors. Cureus. 2020; 12: e11376.
- [101] Borrás-Blasco J, Belda A, Rosique-Robles JD, Castera ME, Abad FJ. Burning mouth syndrome due to efavirenz therapy. Annals of Pharmacotherapy. 2006; 40: 1471–1472.
- [102] Yang G, Jin J, Wang K, Baad-Hansen L, Liu H, Cao Y, et al. Conditioned pain modulation differences in central and peripheral burning mouth syndrome (BMS) patients. Journal of Oral Rehabilitation. 2025; 52: 443–452.
- [103] Wu YH, Jin YT, Wu YC, Yu-Fong Chang J, Chiang CP, Sun A. Anemia, hematinic deficiencies, and hyperhomocysteinemia in male and female burning mouth syndrome patients. The Journal of Dental Sciences. 2022; 17: 935–941.
- [104] Ganguly P, Alam SF. Role of homocysteine in the development of cardiovascular disease. Nutrition Journal. 2015; 14: 6.
- [105] Gaifullina AS, Lazniewska J, Gerasimova EV, Burkhanova GF, Rzhepetskyy Y, Tomin A, et al. A potential role for T-type calcium channels in homocysteinemia-induced peripheral neuropathy. Pain. 2019; 160: 2798–2810.
- [106] Gerasimova E, Burkhanova G, Chernova K, Zakharov A, Enikeev D, Khaertdinov N, et al. Hyperhomocysteinemia increases susceptibility to cortical spreading depression associated with photophobia, mechanical allodynia, and anxiety in rats. Behavioural Brain Research. 2021; 409: 113324.
- [107] Hainsworth AH, Yeo NE, Weekman EM, Wilcock DM. Homocysteine, hyperhomocysteinemia and vascular contributions to cognitive impairment and dementia (VCID). Biochimica et Biophysica Acta. 2016; 1862: 1008–1017.
- [108] Page A, Aktas MK, Soyata T, Zareba W, Couderc JP. “QT clock” to improve detection of QT prolongation in long QT syndrome patients. Heart Rhythm. 2016; 13: 190–198.
- [109] Nagamine T, Watanabe T, Toyofuku A. QTc shortening on electrocardiogram with amitriptyline may indicate no effect on pain relief in burning mouth syndrome. Clinical Neuropharmacology. 2024; 47: 33–36.
- [110] Chen CC, Lin C, Lee DJ, Lin CS, Chen SJ, Sung CC, et al. Monitoring serum potassium concentration in patients with severe hyperkalemia: the role of bloodless artificial intelligence-enabled electrocardiography. Clinical Kidney Journal. 2025; 18: sfaf092.
- [111] Suga T, Takenoshita M, Tu TTH, Sugawara T, Kirimura S, Toyofuku A. A case of vestibular schwannoma mimicking burning mouth syndrome. BioPsychoSocial Medicine. 2021; 15: 7.
- [112] Liu YF, Kim Y, Yoo T, Han P, Inman JC. Burning mouth syndrome: a systematic review of treatments. Oral Diseases. 2018; 24: 325–334.
- [113] Veenbergen S, Kozmar A, van Daele PLA, Schreurs MWJ. Autoantibodies in Sjögren’s syndrome and its classification criteria. Journal of Translational Autoimmunity. 2022; 5: 100138.
- [114] Shiboski CH, Shiboski SC, Seror R, Criswell LA, Labetoulle M, Lietman TM, et al. 2016 American college of rheumatology/European league against rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis & Rheumatology. 2017; 69: 35–45.
- [115] Farag AM, Albuquerque R, Ariyawardana A, Chmieliauskaite M, Forssell H, Nasri-Heir C, et al. World workshop in oral medicine VII: reporting of IMMPACT-recommended outcome domains in randomized controlled trials of burning mouth syndrome: a systematic review. Oral Diseases. 2019; 25: 122–140.
