Pollenkitt From Narcissus tazetta: Evaluation of Its UV‐Protective Potential as a Plant‐Based Biomaterial Source
Abstract
Pollenkitt, the lipid‐rich coating of pollen grains, is a promising natural ultraviolet (UV)‐protective source rich in phenolic and flavonoid compounds. This study assessed the UV protection capacity of pollenkitt extracted from Narcissus tazetta using six solvents (dH2O, acetone, diethyl ether, ethanol, chloroform, and methanol). Methanol extracts showed the highest UV absorbance and sun protection factor (SPF) (17.8), despite 35% lower phenolics than ethanol, suggesting specific compounds like rutin and ferulic acid may drive UV protection. Surprisingly, although the dH2O extracts yielded less pollenkitt, they exhibited strong UV absorption SPF (19.1) and antioxidant activity from water‐soluble compounds such as gentisic acid. Absorption, distribution, metabolism, excretion, and toxicity (ADMET) analysis predicted favorable bioavailability and low toxicity for most phenolics, with rosmarinic acid being predicted to possess properties associated with anticarcinogenic potential. However, rutin and naringenin were predicted to exhibit a skin sensitization risk. These findings underscore the potential of pollenkitt, specifically methanol and ethanol extracts, as a safe, natural UV shield for cosmetic, packaging, and coating applications.
Article type: Research Article
Keywords: antioxidants, natural products, pollen biotechnology, pollenkitt, ultraviolet (UV) protection
Affiliations: Department of Biology Marmara University Kadıköy, Istanbul Turkey; Institute of Pure and Applied Sciences Marmara University Istanbul Turkey; Department of Life Sciences University of Siena Siena Italy
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202502122 | PubMed: 41021701 | PMC: PMC12715996
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.1 MB)
Introduction
Ultraviolet (UV) radiation had increased globally in the past due to the progressive depletion of the ozone layer [ref. 1]. Although recent assessments indicate that the ozone layer is gradually recovering following the Montreal Protocol, prolonged and high levels of UV radiation (as caused by more clear skies following fewer precipitation events) still pose a serious risk to living organisms [ref. 2]. Therefore, defense strategies to protect living organisms against the harmful effects of UV rays have been widely investigated [ref. 3]. In recent years, research on potential defense and protection strategies using plant‐based materials (known to be versatile, natural, and environmentally friendly) has emerged as promising candidates in various sectors such as cosmetics, coatings, textiles, and food packaging [ref. 4]. To this end, the UV‐protective effects of plant extracts from various plant tissues, such as roots, leaves, fruits, and flowers, have been investigated [ref. 5], but alternative plant structures as new sources remain largely unexplored.
Pollenkitt (also known as pollen coat or tryphine) is an extracellular layer of pollen grains [ref. 6] that is derived from the tapetum and only rarely from different anther components such as the endothecium and microspores. After tapetal cell breakdown, the pollenkitt is deposited on the outermost surface of the pollen grain and fills the spaces and cavities of the exine [ref. 7, ref. 8]. Pollenkitt is composed of lipids, carotenoids, phenolics, flavonoids, proteins, and carbohydrates, the proportions of which vary among species [ref. 9]. One of the important functions of pollenkitt is to protect pollen grains from UV radiation [ref. 9]. Mori et al. [ref. 10] found that UV had less effect on the viability of pollen grains with pollenkitt. In addition, Rejón et al. [ref. 11] found that UV‐absorbing compounds such as phenolics and flavonoids in pollenkitt prevent UV from reaching the underlying exine, thus protecting the genetic material of pollen grains from UV. On the basis of these studies, we hypothesized that pollenkitt could be a valuable plant component to produce plant‐based UV‐protective materials.
Pollenkitt must be successfully isolated before it can be used as a plant‐based UV protector. The pollenkitt is a hydrophobic layer because it is mostly composed of lipids [ref. 9, ref. 12] and can be easily removed by treatment with organic solvents such as cyclohexane, diethyl ether, chloroform, carbon tetrachloride, hexane, heptane, cyclohexane, benzene, diethyl ether, methanol, and ethanol [ref. 13, ref. 14, ref. 15]. In addition, pollenkitt isolation frequently involves the use of multiple solvents [ref. 16]. Because the amount of raw material used in the production of plant‐based preservatives is important, the amount of pollenkitt isolated is just as important as its successful isolation. Isolated pollenkitt also has a critical UV protection capacity. Antioxidant activity and UV protection capacity are all closely related [ref. 17], as antioxidants reduce UV‐induced excessive production of reactive oxygen species. In addition, there is considerable evidence that plant polyphenols such as phenolics and flavonoids, which have antioxidant properties, are the most effective UV‐protective compounds because they act as radical scavengers [ref. 18]. The UV protection capacity of the isolated pollenkitt can then be determined by measuring antioxidant activity, phenolic, and flavonoid content. In addition, the profile of phenolic and flavonoid compounds can provide more detailed information on UV protection capacity. Various researchers have emphasized the importance of different chemicals in UV protection, such as caffeic acid and ferulic acid [ref. 19], quercetin, luteolin, and catechins [ref. 20], resveratrol [ref. 21], chlorogenic acid [ref. 22], and rutin and quercetin [ref. 23]. In addition, information on UV protection capacity can be obtained by measuring UV absorption values as well as the sun protection factor (SPF), which is the most important measurement for determining the efficacy of sunscreen materials [ref. 24, ref. 25]. As a result, the UV absorbance and SPF values of isolated pollenkitt can be useful in determining the UV protection capacity of pollenkitt.
The compounds in the plant‐based UV‐protective material are usually evaluated for their physicochemical, medicinal chemistry, excretion, and toxicological properties to assess their potential use as UV‐protective materials. Due to the time and cost constraints of in vitro experiments and the ethical concerns of toxicity testing, computational analyses are often preferred [ref. 26]. Low molecular weight (MW) compounds have high absorbency, making them very versatile [ref. 27, ref. 28]. Density affects the physical properties, processability, and performance that are critical to the coating and packaging industries. The log S and log P values define the hydrophilic and lipophilic behavior, which is key to evaluating the effectiveness of UV‐protective materials [ref. 29]. pK a values provide insight into acidity, improving formulation stability and efficiency [ref. 30]. The synthetic accessibility score (SAscore) evaluates the ease of compound synthesis, whereas Lipinski’s rule of five predicts bioavailability potential [ref. 31, ref. 32]. The natural product score (NPscore) suggests that naturally derived compounds generally have a lower toxicity risk [ref. 33]. Plasma clearance (CLplasma) and half‐life (T1/2) are critical in determining the duration of action of a compound and the frequency of dosing [ref. 34, ref. 35]. From a toxicological perspective, the Ames test assesses potential genotoxic risks, whereas the skin sensitization rule assesses the potential for allergic reactions [ref. 36]. The carcinogenicity rule evaluates cancer risk through genotoxic and non‐genotoxic mechanisms. The signal responsive antioxidant response element (SR‐ARE) examines resistance to oxidative stress. The Food and Drug Administration (FDA) Maximum Daily Dose (FDAMDD) establishes toxic dose limits [ref. 37]. Nonbiodegradable properties are addressed for environmental persistence and sustainability [ref. 38]. Together, these parameters provide critical insight into the design of UV‐protective biomaterials to ensure efficacy, stability, safety, and environmental compatibility. Understanding these properties is essential for tailoring plant‐based UV‐protective materials to meet the needs of various sectors, including cosmetics, textiles, food packaging, and coatings, while promoting sustainability and reducing environmental impact.
This study aims to investigate the potential of pollenkitt, the outermost layer of pollen grains, as a plant‐based UV‐protective material by evaluating its isolation process, UV‐protective capacity, and the physicochemical and toxicological properties of its compounds. We hypothesize that through comparative solvent extraction, including an aqueous extraction, we can identify specific pollenkitt extracts, particularly from methanol and dH2O, with superior UV protection and desirable biochemical profiles, thereby validating and optimizing pollenkitt as a sustainable and environmentally friendly source of industrially applicable UV‐protective biomaterials, such as in cosmetics, textiles, food packaging, and coatings.
Results
Effectiveness of Pollenkitt Isolation and Determination of the Amount of Isolated Pollenkitt
First, fresh pollen grains were tested for the presence of pollenkitt using a glycerin–gelatin solution containing safranin. According to the observations, the pollenkitt appeared as droplets on the surface of the pollen grains (Figure 1a). In addition, many pollen grains were clumped together with the pollenkitt (Figure 1b). Pollen grains were treated with dH2O, acetone, diethyl ether, ethanol, chloroform, and methanol to determine if the pollenkitt was removed. After dH2O treatment, the pollenkitt remained visible as yellow in the pollen grains (Figure 1c). However, after treatments with acetone, diethyl ether, ethanol, chloroform, and methanol, the pollenkitt was removed and no lipid droplets were found on the surface of the pollen grains (Figure 1d). According to the amount of pollenkitt on isolates, the most pollenkitt was isolated after acetone, ethanol, chloroform, and methanol treatment with no discernible difference, and the least after diethyl ether and dH2O treatment with no significant difference (Figure 1e).
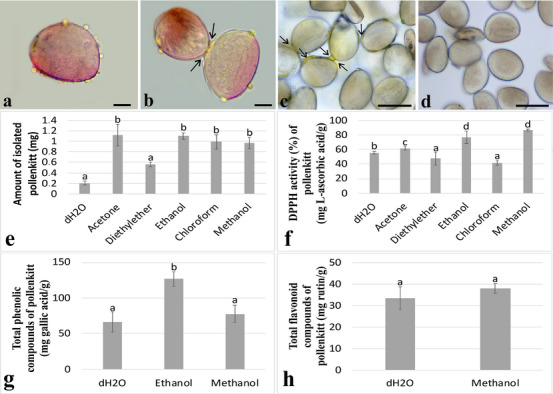
Determination of Total Antioxidant Activity, Total Phenolic, and Flavonoid Contents
The antioxidant activity, total phenolic content, and flavonoid content of isolated pollenkitt were investigated as these are closely related to the UV‐protective capacity of pollenkitt. Antioxidant activity was expressed as percentage inhibition of 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) radicals, whereas total phenolic and flavonoid contents were calculated on the basis of calibration curves and expressed as mg gallic acid equivalents/g and mg rutin equivalents/g, respectively. Pollenkitt demonstrated the highest DPPH% activity following ethanol (77.29 ± 0.84) and methanol (87.38 ± 0.12) treatments, with no statistically significant difference between the two. Acetone treatment also resulted in relatively high DPPH% activity (61.95 ± 2.79), although it was lower compared to ethanol and methanol. Furthermore, diethyl ether (47.78 ± 9.02) and chloroform (41.85 + 3.39) treatments resulted in the lowest DPPH% activity of pollenkitt, with no statistical difference. After treatments with diethyl ether and chloroform, pollenkitt showed the lowest DPPH% activity of pollenkitt after dH2O treatment (Figure 1f). Furthermore, the highest total phenolic content was detected in the ethanol extract (126.76 ± 10.04), followed by methanol (77.84 ± 12.18) and dH2O (66.24 ± 14.09). No phenolic content was detected in acetone, diethyl ether, and chloroform extracts. No total phenolic content was detected after treatments with acetone, diethyl ether, and chloroform (Figure 1g). Furthermore, total flavonoid content was determined only in methanol (38.08 ± 2.26) and dH2O (33.35 ± 0.52) extracts, with no statistically significant difference between these two groups. Acetone, diethyl ether, ethanol, and chloroform treatments did not result in any detectable total flavonoid content (Figure 1h).
Liquid Chromatography–Tandem Mass Spectrometry (LC–MS/MS) Analysis of Phenolic and Flavonoid Contents
The profiles of phenolic and flavonoid compounds were further analyzed using LC–MS/MS. According to the data, 21 chemicals were found in the pollenkitt extracted with different solvents, of which 12 were phenolic acids and 9 were flavonoids (Table 1). No gallic acid or chlorogenic acid was detected after any solvent treatment. The dH2O isolate contained the most gentisic acid, 4‐OH‐benzoic acid, rosmarinic acid, myricetin, luteolin, apigenin, galangin, and chrysin. However, the highest concentrations after dH2O isolation were found in the ethanol isolate for gentisic acid and apigenin and in the methanol isolate for 4‐OH‐benzoic acid, rosmarinic acid, myricetin, luteolin, galangin, and chrysin. The dH2O isolate did not contain caffeic acid or trans‐cinnamic acid. The acetone isolate contained the highest levels of caffeic acid and p‐coumaric acid, but no 4‐OH‐benzoic acid and galangin. The diethyl ether isolate contained the most trans‐cinnamic acid while having the lowest levels of protocatechualdehyde, ferulic acid, rosmarinic acid, sinapic acid, rutin, and luteolin. In addition, 4‐OH‐benzoic acid, gentisic acid, and caffeic acid were not detected in diethyl ether isolate. Protocatechuic acid, protocatechualdehyde, sinapic acid, resveratrol, naringenin, quercetin, and kaempferol were the most abundant in the ethanol extract, whereas 4‐OH‐benzoic acid was not detected. Of all the isolates tested, the chloroform extract did not have the highest concentration of any phenolic acid or flavonoid compound. However, the chloroform isolate had the lowest levels of protocatechuic acid, salicylic acid, p‐coumaric acid, resveratrol, myricetin, naringenin, quercetin, kaempferol, apigenin, and chrysin. In addition, 4‐OH‐benzoic acid and gentisic acid were not detected in the chloroform extract. The methanol isolate contained the highest levels of salicylic acid, ferulic acid, and rutin.
TABLE 1: Liquid chromatography–tandem mass spectrometry (LC–MS/MS) analytical results of phenolic and flavonoid compounds in pollenkitt isolated by different solvents (µg/L).
| Compounds | dH2O | Acetone | Diethyl ether | Ethanol | Chloroform | Methanol | |
|---|---|---|---|---|---|---|---|
| Phenolic compounds | Gallic acid | — | — | — | — | — | — |
| Gentisic acid | 36.63 | 18.74 | — | 30.23 | — | 30.11 | |
| Protocatechuic acid | 67.91 | 60.99 | 19.23 | 83.60 | 14.45 | 69.86 | |
| Protocatechualdehyde | 7.57 | 6.90 | 5.13 | 12.86 | 5.81 | 8.04 | |
| 4‐OH‐Benzoic acid | 47.35 | — | — | — | — | 30.40 | |
| Salicylic acid | 379.15 | 365.9 | 313.51 | 481.44 | 22.09 | 523.90 | |
| Chlorogenic acid | — | — | — | — | — | — | |
| Caffeic acid | — | 24.46 | — | 19.76 | 11.11 | 21.36 | |
| p‐Coumaric acid | 236.98 | 806.33 | 184.68 | 315.88 | 20.67 | 212.01 | |
| Ferulic acid | 33.34 | 61.90 | 4.61 | 69.73 | 6.39 | 129.87 | |
| Rosmarinic acid | 589.00 | 288.33 | 135.76 | 384.69 | 195.84 | 491.88 | |
| Sinapic acid | 50.26 | 44.66 | 25.98 | 76.26 | 44.59 | 52.52 | |
| Trans‐cinnamic acid | — | 33.37 | 47.27 | 21.97 | 23.21 | 43.73 | |
| Resveratrol | 327.85 | 477.27 | 103.43 | 554.62 | 58.28 | 374.34 | |
| Flavonoids | Rutin | 141.47 | 70.08 | 11.92 | 143.55 | 23.75 | 324.97 |
| Myricetin | 1547.57 | 381.74 | 175.49 | 1071.76 | 140.71 | 1306.39 | |
| Naringenin | 10.63 | 9.99 | 11.26 | 12.16 | 7.51 | 11.08 | |
| Quercetin | 618.67 | 601.19 | 374.46 | 1125.76 | 147.28 | 939.98 | |
| Luteolin | 345.05 | 50.25 | 47.82 | 96.70 | 58.57 | 154.66 | |
| Kaempferol | 7189.14 | 38 705.91 | 23 884.56 | 48 236.59 | 5562.05 | 36 949.38 | |
| Apigenin | 71.11 | 25.09 | 16.60 | 28.11 | 15.08 | 27.44 | |
| Galangin | 255.50 | — | 2.85 | 9.22 | 5.31 | 34.50 | |
| Chrysin | 70.61 | 3.27 | 4.30 | 7.30 | 1.28 | 10.92 |
Determination of UV‐A/UV‐B Absorbance and SPF Value
UV absorptions of pollenkitt isolated with different solvents were measured at wavelengths ranging from 280 to 400 nm. UV‐B and UV‐A absorbance values were determined in the following descending order: dH2O, methanol, ethanol, chloroform, diethyl ether, and acetone (Figure 2a,b). In order to determine the UV protection capacities, the SPF values of pollenkitt isolated with different solvents were calculated. According to the calculations, the dH2O isolate had the highest SPF value and the acetone isolate had the lowest SPF value. After dH2O, the methanol and ethanol isolates showed the highest values. In addition, diethyl ether and chloroform isolates had the lowest values after acetone (Figure 2c).
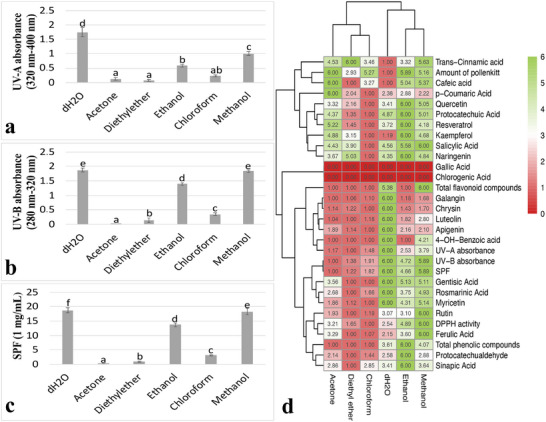
Heat‐Map Analysis
A heatmap was created to collectively evaluate the parameters of the solvents and determine the most appropriate solvent. The columns with the strongest green color represent the groups with the highest values, whereas the columns with the strongest red color represent the groups with the lowest rates of change. Methanol and ethanol were assigned to the same sub‐cluster, which then merged with dH2O in a higher level cluster. Similarly, chloroform and diethyl ether were assigned to the same sub‐cluster, which then merged with acetone to form a higher level cluster. In addition, a ranking was determined by calculating the total value of the cells in each column. The ranking showed that methanol, ethanol, and dH2O had the highest values, whereas chloroform, diethyl ether, and acetone had the lowest values (Figure 2d).
Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) Analysis
The ADMET scores, which include physicochemical properties, medicinal chemistry, and excretion characteristics, were used to evaluate the applicability of the phenolic and flavonoid compounds identified in the pollenkitt by LC–MS/MS analyses in relevant industries. The results are shown in Table 2. The MWs of the phenolic compounds ranged from 138.03 to 360.08, whereas the MWs of the flavonoid compounds ranged from 254.06 to 610.15. The density values of all molecules were determined to be between 0.9 and 1.1. All phenolic compounds tested had log S values between −4 and 0.5 log mol/L, which is considered optimal. However, among the flavonoid compounds, only chrysin, apigenin, luteolin, and naringenin were found to be outside of this optimal range. On the basis of log P values, gallic acid and protocatechuic acid among the phenolic compounds and rutin and myricetin among the flavonoid compounds were found to have a high tendency for lipophilicity. The pK a (acid) values of protocatechualdehyde and resveratrol among the phenolic substances were 7.27 and 9.62, respectively, whereas the other molecules were in the range of 3–5, and the pK a (acid) values of the flavonoid contents were in the range of 4–8. All of the pK a (basic) values were in the range of 1–5. The SAscore of all phenolic and flavonoid compounds was determined to be in the “easy” category. According to Lipinski’s rule evaluation, all phenolic compounds were classified as “accepted” as drug‐like substances. Rutin was the only flavonoid compound classified as “rejected,” with all other flavonoids classified as “accepted.” According to the NPscore analysis, the phenolic compounds with the highest scores were chlorogenic acid (2.246), caffeic acid (1.124), and rosmarinic acid (1.128). In the analysis of flavonoid compounds, rutin had the highest NPscore (2.015), whereas all other flavonoid compounds had NPscore values greater than 1. The CLplasma values showed that the phenolic compounds 4‐hydroxybenzoic acid, salicylic acid, and chlorogenic acid, as well as the flavonoid compounds rutin, galangin, and chrysin, had a low clearance. All other phenolic and flavonoid compounds showed moderate clearance. In addition, among the flavonoid compounds, chrysin had an ultra‐short half‐life with a T1/2 value of 0.967, whereas rutin had a moderate half‐life (T1/2: 4.616). The remaining flavonoid and phenolic compounds were found to have short half‐lives.
TABLE 2: Absorption, distribution, metabolism, excretion, and toxicity (ADMET) scores of phenolic and flavonoid compounds based on their physicochemical, medicinal, and excretion properties.
| Physicochemical property | Medicinal chemistry | Excretion | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compounds | MW | Density | Log S | Log P | pK a (acid) | pK a (base) | SAscore | Lipinski rule | NPscore | CLplasma | T1/2 | |
| Phenolic compounds | Gallic acid | 170.02 | 1.10 | −1.55 | 0.69 | 4.49 | 2.58 | Easy | + | 0.98 | 5.40 | 2.2 |
| Gentisic acid | 154.03 | 1.05 | −2.03 | 1.39 | 4.16 | 4.22 | Easy | + | 0.73 | 13.91 | 1.72 | |
| Protocatechuic acid | 154.03 | 1.05 | −1.69 | 1.00 | 4.15 | 2.02 | Easy | + | 0.73 | 4.94 | 2.25 | |
| Protocatechualdehyde | 152.05 | 0.98 | −1.77 | 1.12 | 7.27 | 4.14 | Easy | + | 0.85 | 10.28 | 1.52 | |
| 4‐OH‐Benzoic acid | 138.03 | 1.00 | −1.77 | 1.23 | 4.27 | 2.55 | Easy | + | 0.37 | 3.45 | 1.66 | |
| Salicylic acid | 138.03 | 1.00 | −1.99 | 2.26 | 3.81 | 2.59 | Easy | + | 0.13 | 2.66 | 1.31 | |
| Chlorogenic acid | 354.1 | 1.06 | −2.95 | 1.03 | 4.22 | 4.10 | Easy | + | 2.24 | 3.34 | 2.75 | |
| Caffeic acid | 180.04 | 1.01 | −1.83 | 1.19 | 4.47 | 2.80 | Easy | + | 1.12 | 14.35 | 2.07 | |
| p‐Coumaric acid | 164.05 | 0.97 | −2.11 | 1.44 | 4.57 | 2.98 | Easy | + | 0.84 | 7.54 | 1.57 | |
| Ferulic acid | 194.06 | 0.99 | −2.36 | 1.64 | 4.36 | 3.36 | Easy | + | 0.92 | 8.35 | 1.69 | |
| Rosmarinic acid | 360.08 | 1.03 | −3.03 | 2.00 | 5.38 | 3.13 | Easy | + | 1.12 | 13.23 | 1.90 | |
| Sinapic acid | 224.07 | 1.01 | −2.54 | 1.68 | 4.37 | 4.89 | Easy | + | 0.76 | 5.72 | 2.20 | |
| Trans‐cinnamic acid | 148.05 | 0.92 | −3.35 | 2.61 | 4.30 | 1.70 | Easy | + | 0.40 | 6.16 | 1.88 | |
| Resveratrol | 228.08 | 0.94 | −3.60 | 2.89 | 9.62 | 4.53 | Easy | + | 0.75 | 9.03 | 1.45 | |
| Flavonoid | Rutin | 610.15 | 1.10 | −2.39 | 0.98 | 4.57 | 5.32 | Easy | − | 2.01 | 1.61 | 4.61 |
| Myricetin | 318.04 | 1.09 | −3.44 | 1.11 | 6.26 | 1.43 | Easy | + | 1.69 | 6.77 | 1.62 | |
| Naringenin | 272.07 | 1.01 | −4.02 | 2.59 | 8.97 | 5.13 | Easy | + | 1.76 | 6.89 | 1.31 | |
| Quercetin | 302.04 | 1.06 | −3.72 | 1.44 | 6.40 | 2.08 | Easy | + | 1.70 | 8.28 | 1.58 | |
| Luteolin | 286.05 | 1.04 | −4.01 | 2.24 | 7.94 | 3.19 | Easy | + | 1.49 | 8.48 | 1.37 | |
| Kaempferol | 286.05 | 1.04 | −3.64 | 1.96 | 6.81 | 3.64 | Easy | + | 1.54 | 5.69 | 1.38 | |
| Apigenin | 270.05 | 1.01 | −4.21 | 2.98 | 8.40 | 3.66 | Easy | + | 1.35 | 5.93 | 1.20 | |
| Galangin | 270.05 | 1.01 | −3.19 | 2.22 | 5.84 | 3.60 | Easy | + | 1.42 | 4.63 | 1.05 | |
| Chrysin | 254.06 | 0.99 | −4.88 | 3.6 | 8.90 | 3.38 | Easy | + | 1.22 | 4.66 | 0.96 | |
Abbreviation: MW, molecular weight; NPscore, natural product score; SAscore, synthetic accessibility score.
The toxicity parameters of the phenolic and flavonoid compounds found in the pollenkitt were determined using ADMET scoring, and the results are presented in Table 3. The flavonoid molecules had moderate mutagenic activity, as determined by the Ames toxicity parameters, with values ranging from 0.546 to 0.796. Among the phenolic compounds, resveratrol, rosmarinic acid, protocatechualdehyde, and gentisic acid were found to have moderate mutagenic activity, whereas the remaining phenolic compounds had low mutagenic activity, with values ranging from 0 to 0.3. Rosmarinic acid and resveratrol also exceeded the recommended FDAMDD value, with 0.924 and 0.882, respectively. In addition, all flavonoid compounds except rutin scored higher than the FDAMDD (scores above 0.7), indicating that they may be highly toxic. Furthermore, the phenolic acids 4‐hydroxybenzoic acid, salicylic acid, and sinapic acid as well as the flavonoids galangin and chrysin showed a moderate skin sensitization potential with values ranging from 0.4 to 0.5. All phenolic compounds were found to have low to moderate carcinogenic activity, with values ranging from 0 to 0.5. Among the flavonoids, kaempferol, apigenin, galangin, and chrysin had values greater than 0.7, indicating carcinogenic potential. According to the SR‐ARE parameter analysis, most phenolic compounds and only rutin among flavonoids showed excellent results, with values ranging from 0 to 0.1 (− −), 0.1 to 0.3 (−), and 0.3 to 0.5 (−). According to the Genotoxic Carcinogenicity Mutagenicity Rule, only chlorogenic acid and rosmarinic acid received a value of 1. In contrast, the non‐genotoxic carcinogenicity rule assigned a value of 1 to the following compounds: chlorogenic acid, caffeic acid, p‐coumaric acid, ferulic acid, rosmarinic acid, sinapic acid, and trans‐cinnamic acid. All compounds, except for 4‐hydroxybenzoic acid and salicylic acid (both with a value of 0), had moderate to high skin irritation potential, with values ranging from 2 to 8. All compounds had low nonbiodegradability potentials (values ranging from 0 to 2).
TABLE 3: Absorption, distribution, metabolism, excretion, and toxicity (ADMET) scores of phenolic and flavonoid compounds based on their toxicity parameters.
| Compounds | Ames toxicity | FDAMDD | Skin sensitization | Carcinogenicity | SR‐ARE | Genotoxic carcinogenicity mutagenicity rule | Non‐genotoxic carcinogenicity rule | Skin sensitization rule | Nonbiodegradable | |
|---|---|---|---|---|---|---|---|---|---|---|
| Phenolic compounds | Gallic acid | 0.48 | 0.20 | 0.99 | 0.21 | + + + | 0 | 0 | 7 | 1 |
| Gentisic acid | 0.40 | 0.19 | 0.64 | 0.3 | + + + | 0 | 0 | 4 | 1 | |
| Protocatechuic acid | 0.37 | 0.17 | 0.90 | 0.33 | + | 0 | 0 | 5 | 1 | |
| Protocatechualdehyde | 0.46 | 0.23 | 0.8 | 0.57 | − − | 0 | 0 | 6 | 0 | |
| 4‐OH‐Benzoic acid | 0.27 | 0.13 | 0.41 | 0.42 | − | 0 | 0 | 0 | 0 | |
| Salicylic acid | 0.31 | 0.11 | 0.43 | 0.28 | − | 0 | 0 | 0 | 0 | |
| Chlorogenic acid | 0.38 | 0.41 | 0.98 | 0.22 | − | 1 | 1 | 8 | 1 | |
| Caffeic acid | 0.27 | 0.30 | 0.97 | 0.17 | − | 0 | 1 | 7 | 1 | |
| p‐Coumaric acid | 0.21 | 0.27 | 0.78 | 0.18 | − | 0 | 1 | 2 | 0 | |
| Ferulic acid | 0.25 | 0.19 | 0.74 | 0.24 | − | 0 | 1 | 6 | 0 | |
| Rosmarinic acid | 0.54 | 0.92 | 1 | 0.07 | − | 1 | 1 | 8 | 1 | |
| Sinapic acid | 0.21 | 0.29 | 0.43 | 0.39 | − | 0 | 1 | 6 | 0 | |
| Trans‐cinnamic acid | 0.21 | 0.22 | 0.79 | 0.12 | − − | 0 | 1 | 2 | 0 | |
| Resveratrol | 0.60 | 0.82 | 0.94 | 0.43 | + + + | 0 | 0 | 5 | 1 | |
| Flavonoids | Rutin | 0.75 | 0.13 | 0.99 | 0.04 | − | 0 | 0 | 8 | 2 |
| Myricetin | 0.65 | 0.86 | 0.99 | 0.50 | + + | 0 | 0 | 8 | 1 | |
| Naringenin | 0.70 | 0.74 | 0.74 | 0.59 | + + + | 0 | 0 | 6 | 1 | |
| Quercetin | 0.58 | 0.78 | 0.89 | 0.6 | + + + | 0 | 0 | 8 | 1 | |
| Luteolin | 0.79 | 0.88 | 0.92 | 0.68 | + + + | 0 | 0 | 7 | 1 | |
| Kaempferol | 0.54 | 0.80 | 0.62 | 0.71 | + + + | 0 | 0 | 4 | 1 | |
| Apigenin | 0.61 | 0.88 | 0.64 | 0.79 | + + + | 0 | 0 | 3 | 1 | |
| Galangin | 0.56 | 0.75 | 0.57 | 0.70 | + + + | 0 | 0 | 4 | 1 | |
| Chrysin | 0.62 | 0.84 | 0.59 | 0.78 | + + + | 0 | 0 | 3 | 1 |
Abbreviations: FDAMDD, Food and Drug Administration Maximum Daily Dose; SR‐ARE, signal responsive antioxidant response element.
Discussion
The UV‐protective properties of pollenkitt, found in the pollen grains of many angiosperm species, are well known; however, its UV‐protective capacity has never been directly tested with concrete data in previous studies [ref. 9]. Pollenkitt, a hydrophobic layer, has been reported to be easily isolated from pollen grains of different plant species using different organic solvents for different purposes [ref. 12]. Dobson [ref. 39] used benzene to remove the pollenkitt layer and characterized the lipid compositions of pollenkitt from 69 angiosperm species. Cyclohexane has been used for pollen kit isolation in several studies, including the identification of pollenkitt‐specific oleosins in Brassica oleracea [ref. 40], the extraction of tapetal oleosin‐like fusion protein in Brassica carinata [ref. 41], the analysis of pollenkitt proteins in Cynodon dactylon [ref. 42], and the characterization of pollenkitt biomolecules in Helianthus annuus [ref. 15]. Additionally, diethyl ether and chloroform were used for pollenkitt extraction and fatty acid analysis in Oryza sativa [ref. 43]. Chichiriccò et al. [ref. 14] used carbon disulfide to study the lipid components of pollenkitt in Crocus vernus and Narcissus poeticus. Various solvent combinations have also been used extensively for pollenkitt isolation. For example, Lin et al. [ref. 16] used chloroform and methanol to extract pollenkitt from Taraxacum officinale and H. annuus pollen and determined the amount of pollen cement. However, most of these studies have focused on the analysis of the biomolecules within the isolated pollenkitt, with little emphasis on the volume of pollenkitt extracted by different solvents.
In this study, we hypothesized that the isolated pollenkitt may hold great promise for the development of plant‐based UV‐protective materials. Similar plant‐based strategies have recently been employed in textile applications, such as the incorporation of chitosan‐enriched milk thistle extract‐loaded liposomes onto nonwoven cotton fabrics [ref. 44], showing strong photoprotective and multifunctional properties. Since the amount of raw material is a critical factor in biotech and industrial product development processes, we first identified the solvent that yielded the highest amount of pollenkitt isolate. According to our results, acetone, ethanol, chloroform, and methanol yielded the highest amount of isolated pollenkitt, with no statistically significant differences among them. Although there was no statistically significant difference, dH2O and diethyl ether produced the least amount of pollenkitt. This variation between solvents is thought to be due to differences in their polarity values. It is well known that solvents with different polarities can extract lipid fractions from lipoidal layers, such as pollenkitt, in different ways.
In industrial biotechnology, raw material quantity and quality are critical parameters in the product development process. To evaluate the UV‐protective capacity of the isolated pollenkitt, we investigated its antioxidant activity, total phenolic content, and flavonoid content. In fact, researchers have suggested that the high antioxidant activity, phenolic, and flavonoid content of plant components used as UV filters contribute to UV filtration [ref. 45]. In addition, the pollenkitt layer is known to be rich in antioxidant activity, phenolics, and flavonoids [ref. 46]. Our results showed that the ethanol and methanol extracts had the highest antioxidant activity, with no statistically significant difference between them. This result is consistent with previous studies reporting that major antioxidant compounds are highly soluble in ethanol and methanol [ref. 47]. A comparison of ethanol and methanol extracts of Daemonorops acehensis resin showed that methanol extracts exhibited higher antioxidant activity than ethanol extracts [ref. 45]. It is interesting to note that the dH2O extract had significantly higher antioxidant activity than the diethyl ether and chloroform extracts. Similarly, Sari et al. [ref. 45] found that the dH2O extract of Gyrinops versteegii leaves had significantly high antioxidant activity, comparable to the 100% ethanol extract, which gave the best results.
Phenols and flavonoids, which contain double bonds or aromatic rings, can absorb UV radiation in the 200–400 nm range, making them ideal for sunscreen formulations [ref. 48]. Our study found that total phenolic content was present only in dH2O, ethanol, and methanol extracts, with methanol showing the highest content. The total flavonoid content was observed exclusively in dH2O and methanol extracts with no significant difference between them. This variation is likely due to factors such as plant species, tissue type, distribution of phytochemicals within the tissue, and solvent polarity. Studies have shown similar trends in the UV‐protective activity of isolates with different structures. For example, no phenolic content was detected in ethanol extracts of D. acehensis resin [ref. 45], whereas methanol extracts of Symphytum officinale seeds had significantly higher flavonoid content compared to ethanol extracts [ref. 49]. Consistent with the antioxidant activity results, phenolics and flavonoids were present in dH2O extracts but not in some other solvents, suggesting that low MW phenolic compounds or specific flavonoids with antioxidant properties that are more soluble in dH2O may play a critical role in UV protection.
Numerous studies have shown that various phenolic compounds, including protocatechuic acid, protocatechualdehyde, and sinapic acid, have anti‐UV potential due to their antioxidant and tyrosinase inhibitory activities [ref. 50]. In our case, these compounds were mainly found in ethanol extracts. Quercetin and kaempferol, known for their strong UV‐absorbing capacity, were found in the highest concentrations in ethanolic extracts by Emiliani et al. [ref. 51]. In addition, rutin, quercetin, and naringenin are often reported for their photoprotective properties, with rutin being most concentrated in methanol extracts and quercetin and naringenin in ethanol extracts [ref. 5]. These results emphasize the importance of solvent selection in the isolation process as it significantly affects the extraction of phenolic and flavonoid compounds.
Our results highlight the importance of aqueous solvents, with the highest UV‐A absorbance observed in H2O, followed by methanol and ethanol. For UV‐B absorbance, dH2O and methanol showed the highest absorbance with no significant difference, followed by ethanol. Similarly, the highest SPF value was found in the dH2O extract, followed by methanol and ethanol. Although dH2O did not provide a high amount of pollenkitt, it had strong UV absorption and a high SPF. This suggests that low MW phenolic compounds such as gentisic acid and antioxidant flavonoids such as luteolin and apigenin, which are soluble in dH2O, may contribute to UV protection. LC–MS/MS results further supported this hypothesis, showing the highest concentrations of gentisic acid, luteolin, and apigenin in dH2O extracts. However, we acknowledge that the low predicted aqueous solubility of luteolin and apigenin (log S ≈ −4) may restrict their individual contributions. Therefore, the strong UV absorbance and SPF values of the aqueous extract are more likely due to a combination of highly water‐soluble phenolics, such as gentisic acid, and possible synergistic effects within the pollenkitt matrix. In addition, it should be noted that residual proteins, glycoproteins, polysaccharides, or colloidal components within the pollenkitt matrix could also contribute to the apparent UV absorbance through light‐scattering effects. Although not directly assessed in the present study, this possibility is acknowledged as a limitation and may be addressed in future research.
Both ethanol and methanol extracts showed high antioxidant activity. Although the ethanol extract had a higher total phenolic content, it lacked total flavonoid content. LC–MS/MS analyses revealed the highest concentrations of various phenolic and flavonoid compounds in the ethanol extract. However, the methanol extract showed greater UV absorption and a higher SPF value than the ethanol extract. This suggests that higher concentrations of compounds such as rutin, ferulic acid, and salicylic acid in the methanol extract may have contributed to its superior UV protection. Various phenolic and flavonoid compounds have been found to have different absorption characteristics at different UV wavelengths [ref. 52]. The synergistic effects of the compounds in the methanol extract may have enhanced its UV‐absorbing performance.
According to FDA recommendations, a formulation can be classified as a sunscreen if it has an SPF greater than 2, but an SPF of 15 or higher is recommended for adequate protection. In our study, the SPF values of dH2O and methanol extracts were above 15, whereas the ethanol extract exceeded 10. These results suggest that the pollen layer has significant potential for the development of plant‐based UV‐protective materials. Notably, 30 mg of pollen‐derived pollenkitt showed SPF values ranging from 13 to 19 in various extracts, highlighting its promise compared to other plant extracts with SPF values ranging from 0.97 to 7.38 [ref. 53]. The dependence of SPF values on plant species, tissue type, solvent, and isolation protocol has been described in detail by Li et al. [ref. 4], but no information on pollen or pollen‐derived layers is included. This gap in the literature underscores the novelty and significance of our findings, which could advance plant‐based UV‐protective formulations.
Our heat‐map analysis suggested that methanol and ethanol extracts would produce better results. However, for commercial applications, ethanol extraction is preferred to methanol because it is less flammable, toxic, and odorous.
The use of isolated compounds in industries, such as cosmetics, coatings, textiles, and food packaging, may pose therapeutic and toxicological risks that affect their applicability. Given the time and cost challenges of in vitro experimentation, novel computational models can accelerate risk assessment through predictive analysis. This study used ADMET analysis to evaluate the physicochemical properties and toxicological risks of compounds identified via LC/MS–MS analysis. Low MW compounds are critical for absorption and distribution because they more easily penetrate biological membranes. All identified phenolic and flavonoid compounds, except rutin, had low MWs. Rutin (MW: 610.15) violates Lipinski’s rules due to its high MW, which was predicted to indicate low oral bioavailability [ref. 54].
According to the SAscore, compounds with a score below 6 are easier to synthesize, and all isolated phenolic and flavonoid compounds in our study had SAscore values below 6, suggesting a predicted ease of synthesis [ref. 55]. Most compounds were predicted to meet Lipinski’s rule of five for predicting oral bioavailability, except for rutin, which has a high MW (610.15) and was predicted to have low oral bioavailability [ref. 54]. The NPscore, which assesses how closely a molecule resembles natural products, indicated that all isolated molecules had predicted values within the expected range [ref. 33]. Additionally, most phenolic and flavonoid compounds showed predicted moderate plasma clearance and short half‐lives (T1/2 values ranging from 1.05 to 2.75 h). Although these systemic PK parameters, such as plasma clearance and half‐lives, are not directly relevant to cosmetic, packaging, and coating applications, they were considered complementary ADMET outputs to provide a broader view of the compounds’ safety and stability profiles and to support their potential applicability as biomaterials. For such applications, photostability, local retention, and formulation compatibility are more critical factors; these were not measured in this study but represent an important direction for future research.
The Ames toxicity test is critical for screening compounds for potential carcinogenicity. In our study, resveratrol (0.60), rutin (0.75), luteolin (0.79), and naringenin (0.70) had Ames toxicity scores based on in silico ADMET; these values indicate a model‐predicted mutagenicity risk and warrant experimental validation. Conversely, gallic acid (0.48), caffeic acid (0.27), and sinapic acid (0.21) had lower Ames toxicity scores, which are consistent with a lower mutagenicity risk in this model. The FDAMDD assesses the toxic dose threshold of chemicals. All phenolic compounds had acceptable FDAMDD values, except resveratrol and rosmarinic acid, whose values exceeded the model threshold, indicating a potential toxicity concern based on this prediction [ref. 37]. Most flavonoid compounds showed low safety profile scores in the model, suggesting a potential risk of exceeding safe daily intake levels. Skin sensitization, important in various industries, evaluates the potential for allergic skin reactions [ref. 26]. Except for 4‐OH‐benzoic acid and salicylic acid, all compounds were predicted to have a moderate to high skin sensitivity risk.
Carcinogenicity assessments are essential for estimating cancer risk from long‐term exposure to chemicals. ADMETLab 3.0 data classify carcinogenicity scores as follows: 0–0.3 (low toxicity), 0.3–0.7 (moderate toxicity), and 0.7–1.0 (high toxicity). In our study, phenolic compounds had carcinogenicity values in the low to moderate range, whereas flavonoid compounds had values in the moderate to high range according to the ADMETLab 3.0 model. Rosmarinic acid had the lowest carcinogenicity value (0.072), which is consistent with reports of its potential anticancer properties [ref. 56]. Most phenolic compounds, with the exception of gallic acid, gentisic acid, and resveratrol, showed model‐predicted SR‐ARE activation, which is associated with oxidative stress mitigation. Among flavonoids, only rutin exhibited model‐predicted SR‐ARE activation, which may be associated with oxidative stress regulation.
Toxiphor Rules predict the toxicity potential of molecules based on genotoxic and non‐genotoxic carcinogenicity, skin sensitization, and biodegradability. Our study found that only chlorogenic acid and rosmarinic acid showed model‐predicted genotoxic risks. According to the in silico assessment, seven phenolic compounds, including chlorogenic acid and rosmarinic acid, showed predicted non‐genotoxic carcinogenicity risks. The model did not predict genotoxic or non‐genotoxic carcinogenicity risks for any of the flavonoid compounds. All compounds, except 4‐OH‐benzoic acid and salicylic acid, were predicted to have a high risk of skin sensitization. According to the in silico assessment, only rutin showed low biodegradability, whereas the other compounds were classified as biodegradable. These findings highlight the diverse toxicity profiles of phenolic and flavonoid compounds, emphasizing the need for comprehensive safety assessments.
Conclusion
Our results show that the solvent used has a significant influence on both the amount and the biochemical composition of the isolated pollenkitt, which determines its UV‐protective capacity. Ethanol showed the highest isolation efficiency, whereas methanol extracts showed superior UV absorbance and SPF values, likely due to bioactive compounds such as rutin and ferulic acid. Surprisingly, although dH2O did not fully isolate pollenkitt, it still showed high UV absorbance and antioxidant activity, suggesting its potential for selective extraction of UV‐protective compounds. According to the in silico ADMET analysis, most phenolic compounds, particularly rosmarinic acid, showed predictions consistent with favorable bioavailability and low toxicity, whereas flavonoids exhibited higher skin sensitization potential, supporting the importance of comprehensive safety evaluation. These results support the use of methanol and ethanol for pollen extraction in plant‐based UV protection formulations. The unexpected efficiency of dH2O also suggests the potential for less invasive extraction methods. Further research should optimize extraction techniques and assess long‐term safety for industrial applications in pharmaceuticals, cosmetics, textiles, and food packaging.
Experimental Section
Pollen Material and Pollenkitt Isolation
This study used Narcissus tazetta pollen grains, which contain a high amount of pollenkitt [ref. 14]. Pollen was collected from plants in the Akçakoca/Düzce region (Turkey). They were dehydrated and stored at −20°C until use. Fresh pollen grains were mounted on glycerin–gelatin‐coated slides, supplemented with safranin, and examined under a light microscope to detect the presence of the pollenkitt.
Six different solvents were used to remove pollenkitt from pollen grains: dH2O, acetone, diethyl ether, ethanol, chloroform, and methanol in their absolute forms. We did not dilute the organic solvents to test their full effectiveness [ref. 14, ref. 15]. Following the pollen rehydration process, 30 mg of pollen grains were resuspended in 1 mL of solvent in a tube, vortexed for 1 min, and centrifuged at 5000 g for 5 min. Pellets were examined under a light microscope to determine if pollenkitt could be separated from pollen grains using different solvents. The supernatants were used as a pollenkitt source to determine the total antioxidant activity, total phenolic, and flavonoid content, for LC–MS/MS analysis, UV‐absorbing capacities, and SPF values.
Determination of the Amount of Isolated Pollenkitt
Before the isolation procedure, the weights of all tubes were precisely measured using an analytical balance and systematically documented. Following the pollenkitt isolation, the supernatants were kept at room temperature for 30 min in a vacuum incubator to evaporate the solvents and obtain pure pollenkitt. The weights of the tubes containing purified pollenkitt were determined using a precision balance and recorded. The amount of pollenkitt extracted from 30 mg of pollen using different solvents was calculated by subtracting the initial tube weight measurements from the final tube weight measurements.
In Vitro Determination of Total Antioxidant Activity, Total Phenolic, Total Flavonoid Contents
After the solvents had evaporated, pure pollenkitt was mixed with the solvents to a final concentration of 1 mg/mL. To determine the DPPH radical scavenging activity, 50 µL pollenkitt solution was mixed with 50 µL DPPH solution and incubated in the dark for 30 min. The absorbances of pollenkitt solutions were spectrophotometrically measured at 517 nm [ref. 57]. Each group’s solvent served as a blank, whereas l‐ascorbic acid was used as a positive control. The DPPH% radical scavenging activity was calculated using the formula proposed by [ref. 58], as given in the following formula:
To determine the total phenolic content, 50 µL pollenkitt solutions were mixed with 50 µL of Folin–Ciocalteu reagent and 50 µL of 7.5% Na2CO3. The absorbances of the pollenkitt solutions were spectrophotometrically measured at 750 nm after 1 h incubation in the dark [ref. 58]. Each group’s own solvent served as a blank for pollenkitt. Gallic acid at a stock concentration of 1 mg/mL was used to make the calibration curve. The total flavonoid content was determined by mixing 50 µL pollenkitt solutions with 50 µL of 2% AlCl3 and 50 µL of sodium citrate solutions. After 15 min incubation in the dark, the absorbances of the pollenkitt solutions were spectrophotometrically measured at 435 nm [ref. 58]. Each group’s own solvent was used as a blank. Rutin was used to create calibration curves.
LC–MS/MS Analysis of Phenolic and Flavonoid Contents
The pure pollenkitt was combined with 100% ethanol to achieve a final concentration of 1 mg/mL for LC–MS/MS analysis. A 50 µL aliquot of the pollenkit sample was combined with the internal standard solution and then treated with the extraction reagent. After centrifugation at 3600 g for 5 min, the supernatant was used for chromatography. To ensure accurate detection in each analysis, the mass spectrometric parameters of the standard and sample ions were optimized.
In Vitro Determination of UV‐A and UV‐B Absorbance and SPF Value
Following the pollenkitt isolation procedure, supernatants were kept at room temperature in a vacuum incubator for 30 min to evaporate the solvents and obtain pure pollenkitt. The pollenkitt was combined with its solvents to achieve a final concentration of 1 mg/mL. Prior to analysis, extracts were centrifuged at 5000 g for 10 min and filtered through 0.22 µm syringe filters to remove particulates, thereby minimizing turbidity. Solvent‐specific blanks were also recorded and used for baseline correction, minimizing the impact of potential scattering artifacts. The absorbances of pollenkitt solutions were measured every 5 nm between 280 and 400 nm with a UV spectrophotometer. The solvents from each group were used as controls. SPF values were calculated spectrophotometrically according to Mansur’s equation [ref. 59] using absorbance values between 290 and 320 nm, as given in the following formula:
In the formula, CF is the correction factor (=10), EE(λ) is the erythemal effect, I(λ) is the solar intensity, and Abs(λ) is the sample absorbance at wavelength λ. The EE × I constants were obtained from Sayre et al. [ref. 60] and are shown in Table 4. Absorbance values between 320 and 400 nm were measured only for evaluating UV‐A protective capacity.
TABLE 4: Normalized product function used in the calculation of sun protection factor (SPF) [ref. 60].
| λ | 290 | 295 | 300 | 305 | 310 | 315 | 320 | Total = 1 |
| EE × I | 0.0150 | 0.0817 | 0.2874 | 0.3278 | 0.1864 | 0.0839 | 0.0180 |
Statistical and Computational Analysis
Statistical analysis was performed using SPSS 16.0 software, and data were subjected to one‐way ANOVA with a p value threshold of 0.05. To analyze and visualize the data, a heatmap was generated using the “pheatmap” function in the R programming language and the RStudio integrated development environment. The ADMET analysis was performed using ADMETlab3.0 (https://admetlab3.scbdd.com/) to evaluate physicochemical properties, medicinal chemistry, excretion, and toxicity using the canonical SMILES formulas of phenolic and flavonoid compounds isolated from pollenkit (Supporting Information).
Author Contributions
Aslıhan Çetinbaş‐Genç: conceptualization, investigation, interpretation, writing original draft. Orçun Toksöz: conceptualization, investigation, interpretation, writing original draft. Özkan Kilin: data curation, investigation. Nüzhet Cenk Sesal: data curation, funding acquisition. Giampiero Cai: conceptualization, interpretation, supervision, writing original draft, editing. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Current Insights and Future Perspectives of Ultraviolet Radiation (UV) Exposure: Friends and Foes to the Skin and Beyond the Skin,”. Environment International, 2024. [DOI | PubMed]
- Photochemical Processes of Cell DNA Damage by UV Radiation of Various Wavelengths: Biological Consequences,”. Molecular Biology, 2024. [DOI]
- Emerging Strategies to Protect the Skin From Ultraviolet Rays Using Plant‐Derived Materials,”. Antioxidants, 2020. [DOI | PubMed]
- Antibacterial Mechanism of Lignin and Lignin‐Based Antimicrobial Materials in Different Fields,”. International Journal of Biological Macromolecules, 2023. [DOI | PubMed]
- In Vitro Antioxidant Activity and Solar Protection Factor of Blackberry and Raspberry Extracts in Topical Formulation,”. Journal of Cosmetic Dermatology, 2019. [DOI | PubMed]
- A Dye Combination for the Staining of Pollenkitt and Pollen Wall,”. Plant Reproduction, 2021. [DOI | PubMed]
- Pollen Wall Development in Flowering Plants,”. New Phytologist, 2007. [DOI | PubMed]
- Anther Endothecium‐Derived Very‐Long‐Chain Fatty Acids Facilitate Pollen Hydration in Arabidopsis,”. Molecular Plant, 2018. [DOI | PubMed]
- Pollenkitt – Its Composition, Forms and Functions,”. Flora—Morphology, Distribution, Functional Ecology of Plants, 2005. [DOI]
- The Unusual Conformational Preference of N1, N5, N10‐Tri‐p‐Coumaroylspermidine EZ Isomers From the Japanese Apricot Tree, Prunus mume, for the (ZZZ)‐Form,”. Phytochemistry Letters, 2019. [DOI]
- The Pollen Coat Proteome: At the Cutting Edge of Plant Reproduction,”. Proteomes, 2016. [DOI | PubMed]
- Cloud Condensation Nuclei Activity of Six Pollenkitts and the Influence of Their Surface Activity,”. Atmospheric Chemistry and Physics, 2019. [DOI]
- Diversity and Function of Maize Pollen Coat Proteins: From Biochemistry to Proteomics,”. Frontiers in Plant Science, 2015. [DOI | PubMed]
- Pollenkitt of Some Monocotyledons: Lipid Composition and Implications for Pollen Germination,”. Plant Biology, 2019. [DOI | PubMed]
- Stigma Receptivity With Pollen in Sunflower Accompanies Novel Histochemical and Biochemical Changes in Both Male and Female Reproductive Structures,”. Vegetos, 2020. [DOI]
- Pollenkitt Wetting Mechanism Enables Species‐Specific Tunable Pollen Adhesion,”. Langmuir, 2013. [DOI | PubMed]
- Recent Advances on the Roles of Flavonoids as Plant Protective Molecules After UV and High Light Exposure,”. Physiologia Plantarum, 2021. [DOI | PubMed]
- Milk Thistle Extracts Could Enhance the UV‐Protection Efficiency and Stability of Mineral Filters in Sunscreen Formulations,”. Journal of Photochemistry and Photobiology A: Chemistry, 2024. [DOI]
- In Vitro and In Vivo Evaluation of Caffeic and Ferulic Acids as Topical Photoprotective Agents,”. International Journal of Pharmaceutics, 2000. [DOI | PubMed]
- Natural Phenolics in the Prevention of UV‐Induced Skin Damage. A Review,”. Biomedical Papers of the Medical Faculty of the University Palacky, Olomouc, Czech Republic, 2003
- Effect of Resveratrol Incorporated in Liposomes on Proliferation and UV‐B Protection of Cells,”. International Journal of Pharmaceutics, 2008. [DOI | PubMed]
- Simultaneous Separation and Purification of Total Polyphenols, Chlorogenic Acid and Phlorizin From Thinned Young Apples,”. Food Chemistry, 2013. [DOI | PubMed]
- Biological Activities of Zn (II) and Cu (II) Complexes With Quercetin and Rutin: Antioxidant Properties and UV‐Protection Capacity,”. Revista de Chimie, 2014
- Reliable and Simple Spectrophotometric Determination of Sun Protection Factor: A Case Study Using Organic UV Filter‐Based Sunscreen Products,”. Journal of Cosmetic Dermatology, 2018. [DOI | PubMed]
- Screening of In Vitro Sun Protection Factor of Some Medicinal Plant Extracts by Ultraviolet Spectroscopy Method,”. Journal of Applied Biology and Biotechnology, 2020. [DOI]
- In Silico Prediction of Skin Sensitization for Compounds via Flexible Evidence Combination Based on Machine Learning and Dempster–Shafer Theory,”. Chemical Research in Toxicology, 2024. [DOI | PubMed]
- Generation of a Set of Simple, Interpretable ADMET Rules of Thumb,”. Journal of Medicinal Chemistry, 2008. [DOI | PubMed]
- Defining Optimum Lipophilicity and Molecular Weight Ranges for Drug Candidates—Molecular Weight Dependent Lower Log D Limits Based on Permeability,”. Bioorganic & Medicinal Chemistry Letters, 2009. [DOI | PubMed]
- Drug Delivery Strategies for Avobenzone: A Case Study of Photostabilization,”. Pharmaceutics, 2023. [DOI | PubMed]
- Development of Methods for the Determination of pKa Values,”. Analytical Chemistry Insights, 2013. [DOI]
- Critical Assessment of Synthetic Accessibility Scores in Computer‐Assisted Synthesis Planning,”. Journal of Cheminformatics, 2023. [DOI | PubMed]
- Experimental and Computational Approaches to Estimate Solubility and Permeability in Drug Discovery and Development Settings,”. Advanced Drug Delivery Reviews, 2012. [DOI]
- NaPLeS: A Natural Products Likeness Scorer—Web Application and Database,”. Journal of Cheminformatics, 2019. [DOI | PubMed]
- Pharmacokinetics and Toxicokinetics: Fundamentals and Applications in Toxicology,”. Veterinary Toxicology: Basic and Clinical Principals, 2007
- An Evolved Transformer Model for ADME/Tox Prediction,”. Electronics, 2024. [DOI]
- Alternative Toxicity Testing: Analyses on Skin Sensitization, Toxcast Phases I and II, and Carcinogenicity Provide Indications on How to Model Mechanisms Linked to Adverse Outcome Pathways,”. Journal of Environmental Science and Health, Part C, 2015. [DOI]
- Estimating the Safe Starting Dose in Phase I Clinical Trials and no Observed Effect Level Based on QSAR Modeling of the Human Maximum Recommended Daily Dose,”. Regulatory Toxicology and Pharmacology, 2004. [DOI | PubMed]
- Physicochemical, Medicinal Chemistry, and ADMET Characteristics of Bee Antimicrobial Peptides as Natural Bio‐Preservatives to Extend Food Shelf Life: A Roadmap for Food Safety Regulation,”. Journal of Biomolecular Structure and Dynamics (, 2024. [DOI]
- Survey of Pollen and Pollenkitt Lipids–Chemical Cues to Flower Visitors?”. American Journal of Botany, 1988. [DOI]
- Characterization of Oleosins in the Pollenkitt of Brassica oleracea ,”. Plant Cell, 1997. [DOI | PubMed]
- Modifying the Pollen Coat Protein Composition in Brassica,”. Plant Journal, 2002. [DOI]
- Dual Function of Novel Pollen Coat (Surface) Proteins: IgE‐Binding Capacity and Proteolytic Activity Disrupting the Airway Epithelial Barrier,”. PLoS ONE, 2013. [DOI | PubMed]
- Deficiency of a Triterpene Pathway Results in Humidity‐Sensitive Genic Male Sterility in Rice,”. Nature Communications, 2018. [DOI]
- Chitosan‐Enriched Milk Thistle Extract‐Loaded Liposomes Anchored on Nonwoven Cotton Fabric With Antioxidant, Anti‐Aging and UV Protective Effects,”. International Journal of Biological Macromolecules, 2025. [DOI | PubMed]
- Effect of Ethanol Polarity on Extraction Yield, Antioxidant, and Sunscreen Activities of Phytochemicals From Gyrinops versteegii Leaves,”. IOP Conference Series: Materials Science and Engineering, 2020. [DOI]
- Fossil‐Like Pollen Grains for Construction of UV‐Responsive Photochromic and Fluorogenic Dual‐Functional Film,”. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2021. [DOI]
- Composition and Antioxidant Activity of Raisin Extracts Obtained From Various Solvents,”. Food Chemistry, 2008. [DOI | PubMed]
- Do the Polyphenolic Compounds From Natural Products Can Protect the Skin From Ultraviolet Rays?”. Results in Chemistry, 2022. [DOI]
- Ultraviolet Light Assisted Extraction of Flavonoids and Allantoin From Aqueous and Alcoholic Extracts of Symphytum officinale ,”. Journal of Intercultural Ethnopharmacology, 2017. [DOI | PubMed]
- Antioxidant, Photoprotective and Inhibitory Activity of Tyrosinase in Extracts of Dalbergia ecastaphyllum ,”. PLoS ONE, 2018. [DOI | PubMed]
- Flavonols Protect Arabidopsis Plants Against UV‐B Deleterious Effects,”. Molecular Plant, 2013. [DOI | PubMed]
- Application of Time‐Dependent Density Functional and Natural Bond Orbital Theories to the UV–Vis Absorption Spectra of Some Phenolic Compounds,”. Journal of Physical Chemistry A, 2015. [DOI | PubMed]
- Determination of Sun Protection Factor (SPF) Number of Some Aqueous Herbal Extracts,”. Journal of Applied Pharmaceutical Science, 2013. [DOI]
- Effect of Date Palm (Phoenix dactylifera) Phytochemicals on Aβ1–40 Amyloid Formation: An In‐Silico Analysis,”. Frontiers in Neuroscience, 2022. [DOI | PubMed]
- Computational Pharmacology and Computational Chemistry of 4‐Hydroxyisoleucine: Physicochemical, Pharmacokinetic, and DFT‐Based Approaches,”. Frontiers in Chemistry, 2023. [DOI | PubMed]
- Rosmarinic Acid and Its Derivatives: Current Insights on Anticancer Potential and Other Biomedical Applications,”. Biomedicine & Pharmacotherapy, 2023. [DOI | PubMed]
- Phytochemical Screening and Free Radical Scavenging Activities of Orange Baby Carrot and Carrot (Daucus carota Linn.) Root Crude Extracts,”. Journal of Chemical and Pharmaceutical Research, 2013
- High Correlation of 2, 2‐Diphenyl‐1‐Picrylhydrazyl (DPPH) Radical Scavenging, Ferric Reducing Activity Potential and Total Phenolics Content Indicates Redundancy in Use of All Three Assays to Screen for Antioxidant Activity of Extracts of Plants From the Malaysian Rainforest,”. Antioxidants, 2013. [DOI | PubMed]
- Determinaçäo do Fator de Proteçäo Solar por Espectrofotometria,”. Anais Brasileiros De Dermatologia, 1986
- A Comparison of In Vivo and In Vitro Testing of Sunscreening Formulas,”. Photochemistry and Photobiology, 1979. [PubMed]
