Research Review: What we have learned about the endocannabinoid system in developmental psychopathology
Abstract
Background:
The endocannabinoid (eCB) system, the primary target of cannabis, has gained significant attention as a potential novel therapeutic approach for treating a range of psychiatric disorders characterized by dysregulation of stress, emotion, and social behavior. The use of cannabis itself as a pharmacotherapeutic in children and adolescents is limited due to various constraints, including legal status, stigma, and real or perceived negative side effects. Thus, compounds that target the eCB system without the notable unwanted effects of cannabis may offer a more viable approach for developing populations.
Methods:
In this narrative review, we provide an overview of the eCB system, summarizing its function throughout development and its potential contribution to psychopathology in children and adolescents. We highlight evidence of its behavioral role and the dysregulation of this system in various psychiatric disorders. Finally, we summarize current investigations into pharmacological and nonpharmacological therapeutic interventions designed to target the eCB system.
Conclusions:
The eCB system may offer an innovative target for treatments of various psychiatric disorders in child and adolescent populations. However, more research is needed to understand the nuanced developmental trajectory of this system and to determine whether existing compounds are safe and effective for use in these populations.
Article type: Review Article
Keywords: Endocannabinoid, 2‐arachidonoylglycerol, anandamide, psychopathology, development
Affiliations: Department of Psychiatry Mathison Centre for Mental Health Research and Education, and Hotchkiss Brain Institute, University of Calgary Calgary AB Canada; Department of Child and Adolescent Psychiatry, Psychosomatics and Psychotherapy Faculty of Medicine and University Hospital Cologne, University of Cologne Cologne Germany; Institute of Experimental and Clinical Pharmacology, Toxicology and Pharmacology of Natural Products Ulm University Medical Center Ulm Germany; Department for Child and Adolescent Psychiatry/Psychotherapy University of Ulm Ulm Germany; Department of Psychiatry and Behavioral Neurosciences Wayne State University School of Medicine Detroit MI USA
License: © 2025 The Author(s). Journal of Child Psychology and Psychiatry published by John Wiley & Sons Ltd on behalf of Association for Child and Adolescent Mental Health. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/jcpp.70006 | PubMed: 40639419 | PMC: PMC12626183
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (520 KB)
Introduction
Cannabis and cannabis‐based therapeutics, including the main cannabinoids Δ9‐tetrahydrocannabinol (THC) and cannabidiol (CBD), have long been of interest in psychiatry. While there have been promising advances in cannabinoid‐based treatments for adults, their use in children and adolescents is limited, in part due to the reported negative impacts on neurodevelopment (Albaugh et al., ref. 2021). This underscores the pressing need to explore alternative interventions that safely mimic the therapeutic effects of cannabis‐based treatments in younger populations.
Cannabinoids interact with the endocannabinoid (eCB) system, which regulates a wide range of homeostatic processes, including stress, emotion, pain, sleep, and feeding (Ligresti, De Petrocellis, & Di Marzo, ref. 2016). THC, which is responsible for the intoxicating effects of the drug (Ligresti et al., ref. 2016), is a high‐affinity partial agonist at the cannabinoid receptors CB1 and CB2, which were first isolated in the late 1980s (Devane, Dysarz, Johnson, Melvin, & Howlett, ref. 1988; Munro, Thomas, & Abu‐Shaar, ref. 1993). The discovery of cannabinoid receptors encouraged the search for their endogenous ligands. Subsequently, N‐arachidonoylethanolamine, more commonly referred to as anandamide (AEA), and 2‐arachidonoylglycerol (2‐AG) were discovered in the early 1990s (Devane et al., ref. 1992; Mechoulam et al., ref. 1995). AEA is a partial agonist at CB1 receptors, while 2‐AG is a full agonist. 2‐AG is more abundant in both the central nervous system and peripheral organs than AEA, with concentrations ranging from 100 to 1,000 times higher (Gonsiorek et al., ref. 2000). The elucidation of this system opened avenues for the development of pharmaceuticals that could provide the benefits of cannabis without intoxication.
In this review, we provide a comprehensive overview of the eCB system, its role and intersection with psychopathology during development, and current evidence for its involvement in behavior in individuals with and without psychiatric disorders. While several clinically relevant psychopathologies in childhood and adolescence have been linked to alterations in the eCB system, other prevalent psychiatric disorders in pediatric populations have not demonstrated such associations and are therefore excluded from this review. Given that the majority of psychiatric disorders have an age of onset during the first two decades of life (McGrath et al., ref. 2023), research investigating the role of the eCB system during this period could lead to treatments tailored to the developing brain and prevention strategies to stem the etiology of these disorders.
Overview of the eCB system
The eCB system has several features that differentiate it from classic neurotransmitter systems, as summarized in Figure 1. eCB signaling operates in a homeostatic retrograde fashion (Castillo, Younts, Chávez, & Hashimotodani, ref. 2012). In response to repeated activation, the postsynaptic neuron releases eCB ligands, which travel across the synaptic cleft, bind to CB1 receptors on the pre‐synaptic cell, and trigger an overall inhibition of synaptic activity. The net synaptic effect of eCB signaling is dependent on the pre‐synaptic neuron type (Scheyer, Yasmin, Naskar, & Patel, ref. 2023): for excitatory neurons, CB1 activation results in an overall decrease of transmission (Kreitzer & Regehr, ref. 2001); while in inhibitory neurons, it results in an overall increase of transmission (Wilson & Nicoll, ref. 2001). Unlike other neurotransmitters, which are typically stored in vesicles for future release, the eCBs are synthesized ‘on demand’ (Blankman & Cravatt, ref. 2013). The enzymes fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL) are responsible for the degradation of AEA and 2‐AG, respectively (Blankman & Cravatt, ref. 2013; Deutsch & Chin, ref. 1993). Navigating the intricate pathways of eCB metabolism presents challenges due to the complex synthesis and degradation processes of AEA and 2‐AG.
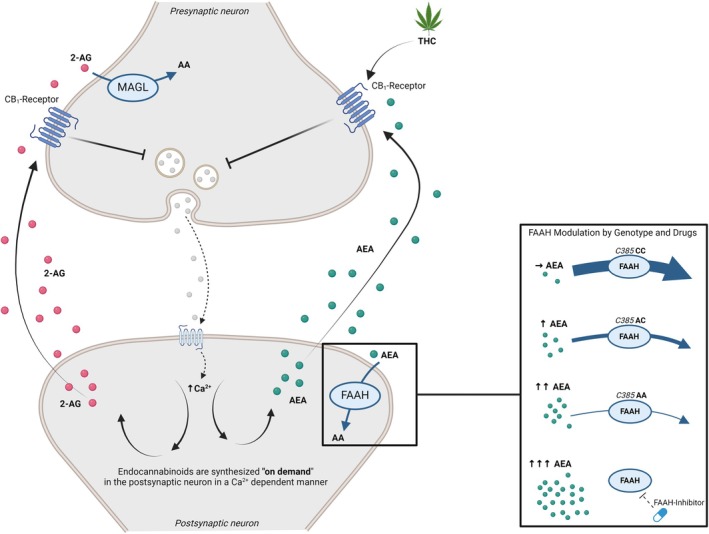
While interventions targeting the biosynthesis pathways of AEA and 2‐AG remain elusive, the identification of compounds that inhibit FAAH and MAGL offers promising prospects. FAAH and MAGL inhibitors can increase brain levels of AEA and 2‐AG, respectively (Ahn et al., ref. 2011; Long et al., ref. 2009), thus amplifying the physiologically relevant, endogenous eCB signal at specific neuronal targets. This contrasts with the administration of exogenous CB1 agonists, such as THC, which bind to CB1 receptors across the brain indiscriminately. As such, facilitating eCB function through enzyme manipulation may provide therapeutic opportunities that are not otherwise feasible with exogenous CB1 agonist administration.
Importantly, dysregulated eCB function has been implicated in multiple psychiatric disorders (Gowatch et al., ref. 2024), including posttraumatic stress disorder (PTSD; Mayo, Rabinak, Hill, & Heilig, ref. 2022), schizophrenia (Zamberletti & Rubino, ref. 2021), anxiety (Lutz, Marsicano, Maldonado, & Hillard, ref. 2015; Petrie, Nastase, Aukema, & Hill, ref. 2021), depression (Gorzalka & Hill, ref. 2011), and autism (De Pol & Kolla, ref. 2021). Consequently, pharmacological alteration of eCB signaling may present a novel therapeutic option for a variety of psychiatric concerns.
Evidence for the role of eCBs in human behavior has been drawn from studies measuring natural variation in their levels, as well as pharmacological studies that target eCB signaling. Genetic variants in the eCB system appear to play a role in a number of neurological and psychiatric symptoms (Smith, Stanley, Foss, Boles, & McKernan, ref. 2017). One source of variation in AEA levels is a common loss‐of‐function single‐nucleotide polymorphism (SNP; rs324420 FAAH 385C→A), of which ~37% of the population carry at least one allele copy, and ~5% two copies, though this varies based on race (Sherry, Ward, & Sirotkin, ref. 1999). This mutation results in reduced enzymatic activity and lower cellular expression levels (Chiang, Gerber, Sipe, & Cravatt, ref. 2004), and consequently higher AEA (Mayo et al., ref. 2020). AA homozygote individuals have the highest measurable levels of AEA, whereas AC heterozygotes have levels greater than wild‐type but less than AA homozygotes (see Figure 1). This variation offers a valuable means to study the eCB system in developing populations without requiring pharmacological interventions or invasive blood sampling.
A complementary yet distinct approach in adults involves pharmacological manipulation to acutely raise eCB levels. AEA levels can be increased through the administration of a FAAH inhibitor, which results in a 10‐fold increase in peripheral AEA levels, compared with the ~ 30% increase seen with the FAAH 385C→A (rs324420) mutation (Mayo et al., ref. 2020; Mayo, Asratian, Lindé, Holm, et al., ref. 2020). Similarly, 2‐AG levels can be pharmacologically increased through the administration of a MAGL inhibitor, although clinical research into the effects of this class of compounds is more limited. These methodologies have enabled the study of the effects of eCBs on stress, fear, and social behavior in adult human subjects.
Much of the literature investigating eCBs and behavioral phenotypes is mixed. It is important to note that eCB function is very sensitive to perturbations by a number of in and ex vivo factors, including food intake, time of day, and room temperature (Hillard, ref. 2018). Differences in these methodological considerations could serve as an important source of variation between findings and highlight the importance of conducting more rigorously controlled research in human populations.
Development of the eCB system
Childhood and adolescence are key periods of neurodevelopmental susceptibility wherein alterations can have lasting impacts on outcomes later in life (Otto et al., ref. 2021). Adolescents are particularly vulnerable to the negative effects of stress, which can have repercussions for future mental health (Andersen & Teicher, ref. 2008). During adolescence, important structural characteristics of the brain are in flux, including the functional connections between areas such as the amygdala, prefrontal cortex, hippocampus, and nucleus accumbens (Andersen & Teicher, ref. 2008). The rapid rate of change during this period makes adolescents particularly sensitive to perturbations from environmental exposures such as early life adversity, which is very common (Lopez et al., ref. 2021). This contributes to an elevated risk of various mental health issues throughout the lifespan (Fujiwara, ref. 2022). In this section, we outline what is currently known about the development of the eCB system and its contributions to mental health in youth.
The developmental trajectories of the eCB system appear to follow complex and fluctuating patterns. The interplay between eCB signaling and stress is bidirectional: while eCBs play a role in regulating the stress response, the eCB system itself can be adversely affected by stressful experiences (Morena, Patel, Bains, & Hill, ref. 2016). While animal models have provided detailed insights into eCB circuitry maturation, there is a paucity of literature translating these findings into humans (for a detailed overview of eCB fluctuations across development in preclinical models, see Meyer, Lee, & Gee, ref. 2018).
The eCB system appears to play a functional role in critical developmental periods. Current evidence suggests that CB1 receptor expression is observable in the human brain as early as 14 weeks gestation (Mato, Del Olmo, & Pazos, ref. 2003; Wang, Dow‐Edwards, Keller, & Hurd, ref. 2003). However, rodent studies have detected CB1 expression as early as gestational day 11, equivalent to weeks 5–6 in human embryos (Buckley, Hansson, Harta, & Mezey, ref. 1997; Wu, Jew, & Lu, ref. 2011), suggesting that CB1 receptor expression may emerge even earlier than originally thought. The expression of the CNR1 gene, which encodes the CB1 receptor, peaks in the human fetal prefrontal cortex during the second trimester and declines after birth (Tao et al., ref. 2020). Further, CB1 receptors have been shown to activate signal transduction cascades from early developmental stages (gestational days 14–21 in rat) and are transiently expressed in structures where they are absent in the adult brain, suggesting a functional role in fetal brain development (Fernández‐Ruiz, Berrendero, Hernández, & Ramos, ref. 2000).
CB1 receptor expression also fluctuates during the first two decades of postnatal life. In postnatal animal models, peak CB1 receptor expression in the prefrontal cortex and limbic areas occurs in early adolescence (Heng, Beverley, Steiner, & Tseng, ref. 2011; Meyer et al., ref. 2018), followed by a decrease into adulthood. However, there may not be a straightforward translation between the developmental trajectories observed in animals and humans. A recent study highlighted the number of cross‐species differences in eCB expression in the periphery across mice, rats, and nonhuman primates (Rosado‐Franco et al., ref. 2024), emphasizing the potential pitfalls of overgeneralizing preclinical findings into humans. Postmortem studies of human tissue have found that CB1 receptor expression in the prefrontal cortex peaks much earlier in life, around infancy and toddlerhood (Long, Lind, Webster, & Weickert, ref. 2012). Similarly, CB1 receptor expression was higher in infancy compared with adulthood in the human midbrain, an area implicated in pain processing (Kwok et al., ref. 2017). This preliminary evidence suggests that while both animal and human research points to the eCB system being a key player in brain development, the developmental trajectories observed in model species may not uniformly apply to humans, emphasizing the importance of further research in humans.
Given the potential differences in eCB development between species, it is important to identify where findings from disparate research modalities converge. A consistent theme emerging from both animal and human studies is that AEA and 2‐AG exhibit divergent developmental dynamics. In the human dorsolateral prefrontal cortex (dlPFC), both FAAH, which degrades AEA, and N‐acylphosphatidylethanolamine‐specific phospholipase D (NAPE‐PLD), which synthesizes AEA, increase in expression from infancy to adulthood, potentially peaking in early adulthood (Long et al., ref. 2012). In contrast, the enzymes regulating 2‐AG expression show more complex trajectories. MAGL, the primary 2‐AG degradation enzyme, peaks in the dlPFC during infancy and then declines into adulthood. Conversely, diacylglycerol lipase (DAGL), one of the 2‐AG synthesis enzymes, does not peak until adolescence (Long et al., ref. 2012). This suggests that 2‐AG levels in the human prefrontal cortex may reach their highest point during adolescence. Further, recent evidence from animal models indicates that biological sex may influence the trajectories of AEA and 2‐AG signaling (Bernabeu et al., ref. 2023; Lee, Hill, & Lee, ref. 2016). Therefore, it is important to characterize the developmental trajectories of AEA and 2‐AG in youth, considering biological factors such as sex.
Links between eCB development and psychopathology
Several key studies have demonstrated an important relationship between eCB system development and behavioral phenotypes linked to psychiatric risk. Using data from the Adolescent Brain Cognitive Development Study® (Casey et al., ref. 2018; Jernigan, Brown, & Dowling, ref. 2018), we previously investigated the associations between the FAAH C385A polymorphism, anxiety symptoms, and frontolimbic white matter pathways (structural connections) in children aged 9–11 years (Marusak, Evanski, Desai, & Rabinak, ref. 2022). Fractional anisotropy (FA), a common metric reflecting various microstructural characteristics of white matter, was used to examine white matter integrity (Kochunov et al., ref. 2012). Compared with C‐allele homozygotes, children with the FAAH 385A allele – associated with higher AEA levels – exhibited lower FA of the left fornix and parahippocampal cingulum, which are frontolimbic pathways critically involved in stress and anxiety regulation (Bubb, Metzler‐Baddeley, & Aggleton, ref. 2018; Senova, Fomenko, Gondard, & Lozano, ref. 2020). Notably, FAAH did not directly predict anxiety symptoms in this preadolescent sample. Instead, left fornix FA mediated the genotype–anxiety relationship, with higher FA associated with greater anxiety. These findings align with prior research suggesting that direct associations between FAAH and anxiety symptoms emerge later, during adolescence. For instance, in adolescents (12+ years), FAAH C385A was linked to lower anxiety and increased FA in the uncinate fasciculus (Gee et al., ref. 2016) – a frontolimbic pathway implicated in memory, cognitive control, and reward processing (Olson, Heide, Alm, & Vyas, ref. 2015). The FAAH C385A variant may influence frontolimbic white matter integrity in youth, shaping psychiatric risk over development. However, the direction of associations between FAAH, FA, and anxiety symptoms may vary by both tract and developmental stage.
More recent reports using the ABCD Study® data provide further insights into the relationship between the FAAH C385A polymorphism, brain function, and mental health. We again found no significant associations between FAAH genotype and anxiety symptoms, or FAAH C385A and functional activation of the nucleus accumbens or amygdala (key regions associated with reward and emotion‐related processing, respectively) during an emotional n‐back task (Desai et al., ref. 2024). However, children and adolescents homozygous for the A allele had significantly lower depressive symptoms than the CC homozygotes and AC heterozygotes. In another study, ABCD Study® data were used to identify a large, spatially distributed network that differed based on FAAH genotype (Sisk et al., ref. 2022). CC homozygotes whose functional connectome fingerprints more greatly resembled A allele carriers across this distributed network had lower anxiety, suggesting that the functional connectivity profile of A allele carriers may represent a resilience factor against anxiety. Together, these findings support the idea that genetic variation in FAAH during development has widespread, complex impacts across both structure and function of the brain, which may be relevant to mental health.
Behavioral and psychiatric correlates of eCB function
Behavioral studies in nonclinical controls
Dysregulation of fear‐ and stress‐related mechanisms is thought to underlie numerous psychiatric conditions (Shin & Liberzon, ref. 2010). Preclinical evidence for the role of eCBs in regulating stress and fear behaviors has been replicated in human studies; however, behavioral studies detailing the effects of eCBs on stress and fear in child and adolescent populations are more scarce. In nonclinical adults, individuals homozygous for the FAAH 385A allele demonstrate enhanced fear extinction and protection against stress‐induced negative affect (Mayo, Asratian, Lindé, Holm, et al., ref. 2020; Ney et al., ref. 2023; Spohrs et al., ref. 2022), as well as lower levels of anxiety than CC homozygotes (Dincheva et al., ref. 2015; Spohrs, Ulrich, Grön, Plener, & Abler, ref. 2022). Other studies have examined the effects of FAAH C385A on amygdala and ventromedial prefrontal cortex (vmPFC) activation – regions thought to play an important role in the regulation of fear‐related learning and memory, particularly fear extinction (Bukalo et al., ref. 2015). In studies of nonclinical adult samples, FAAH 385A carriers show lower activation of the amygdala in response to threat cues (Hariri et al., ref. 2009) and during early extinction recall (Zabik et al., ref. 2022), as well as enhanced functional connectivity between the amygdala and vmPFC at rest (Dincheva et al., ref. 2015). Research using both fMRI and a positron emission tomography (PET) radiotracer for FAAH found that lower FAAH levels in the amygdala were associated with lower amygdala response to a threatening social cue, as well as higher resting‐state functional connectivity between the amygdala and vmPFC (Green et al., ref. 2021, ref. 2022). Together, these findings suggest that FAAH may regulate frontolimbic brain circuits involved in fear and emotion‐related processing.
These observational neuroimaging studies appear to be replicated in pharmacological studies. We previously demonstrated that pharmacological FAAH inhibition resulted in greater recall of fear extinction memory, attenuation of subjective and autonomic stress reactivity, and protection against stress‐induced negative affect (Mayo, Asratian, Lindé, Morena, et al., ref. 2020). Neuroimaging data also showed that FAAH inhibition reduced activation in the anterior cingulate cortex, insula, and amygdala in response to emotional faces (Paulus et al., ref. 2021). Enhancing AEA signaling through FAAH inhibition may help protect against the negative effects of stress‐ and fear‐related behaviors.
The eCB system may also confer resilience to the negative consequences of early life adversity. We previously demonstrated that childhood maltreatment is associated with an increased risk for developing a substance use disorder (SUD) in adulthood (Capusan et al., ref. 2021), which fits with the broader literature. However, in another study, we found that adults with documented childhood trauma but no history of SUD had higher plasma AEA levels compared with nonclinical controls. These elevated AEA levels were associated with greater vmPFC activity during emotional conflict monitoring (Perini et al., ref. 2023), suggesting that circulating AEA concentrations may be linked to resilience, potentially through enhanced emotion regulation abilities. However, further experimental research is needed to confirm this hypothesis and to better characterize the developmental trajectory of these effects, particularly in adolescence.
In addition, 2‐AG has also been implicated in stress and fear processing. Preliminary evidence suggests that 2‐AG may play an important role in the attenuation of fear generalization, a phenomenon involving the transference of fear responses to neutral stimuli that is commonly observed in trauma‐related disorders (Bedse, Hill, & Patel, ref. 2020). Following stress induction, changes in peripheral 2‐AG concentrations, measured in both blood and saliva, have been observed in adults (Ney, Stone, et al., ref. 2021; Spohrs, Prost, et al., ref. 2022), but a recent meta‐analysis did not find consistent evidence supporting this trend (Gowatch et al., ref. 2024). To determine the therapeutic potential of eCBs in youth, it will be imperative to conduct well‐controlled, age‐appropriate behavioral studies on stress and fear processing.
Studies in psychiatric disorders
PTSD
While there is evidence that elevated AEA (occurring naturally or pharmacologically) is associated with protection against stress‐ and fear‐effects in nonclinical populations (Mayo, Asratian, Lindé, Holm, et al., ref. 2020; Mayo, Asratian, Lindé, Morena, et al., ref. 2020), the relationship in PTSD and trauma‐exposed individuals is more complex. In a study investigating fear conditioning in PTSD individuals, the FAAH C385A allele was associated with higher initial skin conductance responses to the conditioned stimulus (CS+), as well as increased physiological arousal to the conditioned stimulus that had not been paired with the aversive stimulus (CS‐), which suggests that there may have been fear generalization (Ney, Matthews, et al., ref. 2021). Another study measured blood eCB levels at hospital admission immediately following traumatic injury and followed up with adult participants 6–8 months later. The authors found that higher AEA concentrations immediately following the traumatic event were associated with a greater likelihood of PTSD diagnosis at follow‐up, and individuals homozygous for the FAAH A allele had significantly higher PTSD symptoms at follow‐up compared with CC and AC genotypes (deRoon‐Cassini et al., ref. 2022). Taken together, this suggests that FAAH and AEA may play different roles in stress and fear responses in those with PTSD compared with controls.
Race and ethnicity may also play a role in the interaction between AEA and trauma. Our recent study of urban youth (~60% racial minority) with high levels of trauma exposure (90% of participants) found that higher circulating AEA concentrations were associated with more severe PTSD symptoms, particularly hyperarousal (Marusak et al., ref. 2024). Additionally, youth carrying the FAAH A allele exhibited higher AEA concentrations and greater PTSD severity. This highlights the need for further research in trauma‐exposed youth and diverse populations to better understand these complex interactions, particularly given the high prevalence of trauma exposure among racially minoritized youth (Pumariega, Jo, Beck, & Rahmani, ref. 2022).
Social anxiety disorder (SAD)
eCBs have been implicated in social processing (Karhson, Hardan, & Parker, ref. 2016). There has been growing interest in whether the eCB system could serve as a useful therapeutic target for SAD. A PET imaging study found that adults with SAD had elevated levels of whole brain FAAH when compared with controls (Ahmed et al., ref. 2020), which may indicate lower AEA concentrations. Clinical trials have explored whether FAAH inhibitors are effective in adults with SAD, though suboptimal dosing potentially contributed to only marginal improvements (Schmidt et al., ref. 2021). Nonetheless, there may be a preliminary signal for the use of eCB‐based therapies in SAD which should be explored more thoroughly.
Autism spectrum disorder (ASD)
Unlike other psychiatric disorders, preliminary research into the eCB system in ASD has included studies in child and adolescent participants. A recent scoping review found alterations in peripheral eCB levels in children with ASD across several studies (De Pol & Kolla, ref. 2021). Peripheral AEA levels were consistently lower in ASD individuals compared with controls, while the findings for 2‐AG were mixed. This suggests that there may be eCB system alterations involved in ASD, though more research is required.
Depression
The eCB system has been investigated as a potential target for major depressive disorder (MDD) (Gorzalka & Hill, ref. 2011). One major catalyst of this area of research was the observation that the weight‐loss drug rimonabant, which blocks CB1 receptor action and downregulates eCB signaling, had the unintended effect of increasing depression and anxiety symptoms in a clinical trial for obesity (Nissen et al., ref. 2008). Since then, multiple studies have demonstrated that the eCB system is altered in individuals with MDD compared with controls, though the patterns of alteration have been variable (McWhirter, Bugarcic, Steel, & Schloss, ref. 2024). Early reports found that serum AEA and 2‐AG were lower in individuals with depression (Hill, Miller, Carrier, Gorzalka, & Hillard, ref. 2009; Hill, Miller, Ho, Gorzalka, & Hillard, ref. 2008). However, more recent studies found that both AEA and 2‐AG blood levels were higher in individuals with depression (Mazurka et al., ref. 2024; Obermanns et al., ref. 2023), and another found no significant differences in the serum concentrations of AEA or 2‐AG in patients with minor or MDD compared with controls (Ho, Hill, Miller, Gorzalka, & Hillard, ref. 2012).
Other alterations in eCB signaling pathways have also been implicated in depression. In one study, there was an increase of CB1 receptor density in the dlPFC of individuals with depression who died by suicide (Hungund et al., ref. 2004). Another study found reduced CB1 receptor‐positive glial cells in postmortem anterior cingulate cortex tissue of individuals with MDD (Koethe et al., ref. 2007). Thirty‐seven genetic mutations in the eCB signaling pathway were found to be more frequent in individuals with MDD compared with controls, though none were related to FAAH, MAGL, or CNR1 (Xu et al., ref. 2023). More research is needed to fully understand the complex relationships between eCB and the emergence of MDD.
Borderline personality disorder (BPD) and nonsuicidal self‐injury (NSSI)
Research into the role of the eCB system in BPD has been mixed. In a pilot study, patients with BPD had reduced AEA concentrations in hair compared with controls (Wingenfeld et al., ref. 2018), and a PET imaging study has shown elevated FAAH levels in the PFC of BPD patients (Kolla et al., ref. 2020), suggesting lower concentrations of AEA in this region. In contrast, others have reported higher serum AEA concentrations in patients with BPD (Schaefer et al., ref. 2014). A more recent study observed elevated plasma AEA levels in patients diagnosed with BPD, but notably, the FAAH genotype appears to play a crucial role in this finding (Spohrs et al., ref. 2023). Unlike the control group, there was no significant difference in AEA levels for AA homozygotes compared with CC homozygotes in the BPD group, and the difference between groups was mainly driven by higher levels of AEA in the BPD CC group compared with the control CC group, suggesting that the relationship between AEA function and FAAH genotype may be altered in the disorder.
While not exclusive to BPD, recurrent nonsuicidal self‐injury (NSSI) is one of the clinical symptoms of BPD that can contribute to a diagnosis (American Psychiatric Association, ref. 2013). We recently demonstrated that female adolescents in a specialized outpatient clinic exhibiting nonsuicidal self‐injurious behavior (NSSI) had lower AEA levels compared with age‐matched peers not exhibiting NSSI, regardless of childhood maltreatment history (Ferger, Sigrist, Brodesser, Kaess, & Koenig, ref. 2024). Additionally, lower AEA levels were associated with a greater frequency of NSSI and a greater severity of childhood maltreatment. This research suggests that eCB processes, especially those related to AEA, may play a role in BPD and NSSI behaviors.
Obsessive‐compulsive disorder (OCD)
Given that exposure therapy – based on principles of fear extinction – is one of the established treatments for OCD (Berg, Webler, Klein, & Kushner, ref. 2024), exploring the impact of eCB signaling on extinction processes could be particularly relevant. Preclinical research supports the idea that increasing eCB signaling can enhance extinction learning (Gunduz‐Cinar et al., ref. 2023), which could potentially translate into human treatments for OCD. Human research investigating the eCB system in OCD remains scarce. However, a recent study found that levels of AEA and 2‐AG synthesizing enzymes were lower in adults with OCD compared with controls (Bellia et al., ref. 2024). This suggests that alterations in eCB synthesis may be associated with OCD pathology. Given these findings, further research into the eCB system’s role in OCD, particularly in children and adolescents, is warranted.
Schizophrenia
The relationship between adolescent cannabis use and the onset of psychosis in individuals with a predisposition to schizophrenia is well‐documented (Malone, Hill, & Rubino, ref. 2010), and implicates the eCB system in the disorder. Although schizophrenia often manifests in early adulthood, structural and functional neurodevelopmental alterations relevant to the disorder can be observed as early as the adolescent and even prenatal periods in individuals later diagnosed with schizophrenia (Zamberletti & Rubino, ref. 2021). A meta‐analysis found higher concentrations of AEA in the cerebrospinal fluid and in the blood of people with schizophrenia compared with healthy controls (Minichino et al., ref. 2019). Research on the role of eCB signaling and neurodevelopment in schizophrenia has predominantly been conducted in animal models. For instance, animal studies have demonstrated that disruptions in eCB signaling can impact neurodevelopmental processes associated with schizophrenia, such as synaptic plasticity and neural connectivity (Zamberletti & Rubino, ref. 2021). However, there is a notable scarcity of research focusing on the interactions between eCBs and schizophrenia during human development.
Clinical interventions targeting the eCB system in psychiatry
Pharmacological interventions
The development of FAAH inhibitors was stalled for a brief period of time following serious adverse events, including the death of one participant, during a Phase I multiple‐rising dose clinical trial involving the drug BIA‐10‐2,474 (Kerbrat et al., ref. 2016). Subsequent investigations found that the adverse events were due to off‐target effects of that specific molecule, and not attributable to a class effect of FAAH inhibitors (van Esbroeck et al., ref. 2017). FAAH inhibitor trials slowly recommenced; however, many trials that were paused during these investigations were ultimately left uncompleted.
In general, results from clinical trials in psychiatric conditions are mixed. Trials in PTSD (NCT05178316 and Eudra‐CT 2020‐001965‐36) and depression (NCT02498392 and EudraCT 2008‐001718‐26) failed to meet their primary endpoints. For cannabis use disorder, one of two trials showed that the FAAH inhibitor was able to reduce cannabis use 4 weeks after the commencement of the study (D’Souza et al., ref. 2019). In a large (n = 228), multisite RCT assessing FAAH inhibitor efficacy in cannabis use disorder (NCT03386487) that concluded in 2023, 8 weeks of therapy with a FAAH inhibitor did not show a reduction in self‐reported cannabis use compared with placebo, though the results have not yet been published. As previously mentioned, one clinical trial investigating FAAH inhibition in SAD did not meet its primary endpoint, but exploratory analyses showed that there may be a dose‐dependent effect of FAAH inhibition in the disorder that warrants further investigation (Schmidt et al., ref. 2021; NCT02432703). While almost all psychiatric FAAH inhibitor trials have been conducted in adults, there is one study that included adolescents that investigated the drug’s efficacy in ASD. While it failed to meet the primary end point, the treatment group showed better responses to secondary end points such as domain‐specific core symptoms, repetitive behaviors, and anxiety (Klein et al., ref. 2025).
Tourette syndrome has also received notable attention as a target for eCB‐based therapies. While a study with a FAAH inhibitor was terminated because no funding was available (NCT02134080), the results of an initial Phase 1b clinical trial with a MAGL inhibitor in adult patients demonstrated a significant reduction in premonitory urges, along with a significant effect on tic severity (Müller‐ Vahl et al., ref. 2022). These encouraging findings prompted the initiation of a phase 2 clinical trial. However, the trial failed to achieve its primary end point of a significant change in tic severity compared with placebo (Müller‐ Vahl et al., ref. 2021).
Nonpharmacological interventions
Given the potential limitations and risks associated with pharmacological interventions during sensitive developmental periods, nonpharmacological strategies that modulate the eCB system may represent a promising area of research. One such approach is acute exercise, which reliably elevates eCB levels in both humans and nonhuman animals (Desai et al., ref. 2022). Our recent systematic review and meta‐analysis revealed that acute exercise consistently increases both AEA and 2‐AG levels across various modalities (e.g., cycling, swimming) and species, including in individuals with physical and mental disorders. Environmental conditions may amplify these effects, as a strenuous hike at higher altitudes is associated with greater elevations in blood AEA concentrations compared with exercise at lower altitudes (Feuerecker et al., ref. 2012). This suggests that hypoxic stress may amplify exercise‐related increases in AEA, although the isolated effects of hypoxia on eCB signaling remain unclear.
Exercise can also facilitate other eCB‐mediated processes. Aerobic exercise, when administered shortly after fear extinction, can enhance the consolidation of fear extinction learning (Crombie, Sartin‐Tarm, Sellnow, Ahrenholtz, Lee, Matalamaki, Adams, et al., ref. 2021a), which may be mediated through elevations in AEA levels (Crombie, Sartin‐Tarm, Sellnow, Ahrenholtz, Lee, Matalamaki, Almassi, et al., ref. 2021b). Further studies have also observed acute elevations in AEA following singing (Stone et al., ref. 2018) highlighting that activities beyond traditional exercise modalities like running or cycling may also impact circulating eCB levels. Despite these promising findings, a significant gap identified in our review is the lack of studies examining the effects of behavioral interventions, such as acute exercise, on circulating eCB levels in children or adolescents. Given the widespread use of physical activity interventions in youth and their associated mental health benefits (Recchia et al., ref. 2023), investigating eCBs as a biomarker could optimize these interventions.
Conclusion
The eCB system appears to be integral to numerous psychological processes, including stress, fear, emotion regulation, and social processing, which are critical to the emergence and progression of various psychiatric disorders. Preliminary evidence, particularly from animal models and adult studies, has provided valuable insights into eCB signaling. Clinical trials have yielded mixed results, highlighting a gap between preclinical discoveries and practical implications. This discrepancy underscores the need for a deeper understanding of the eCB system’s complex interactions with psychopathology and developmental processes. Additionally, emerging research shows significant developmental changes in the eCB system. These changes are crucial for understanding how eCB signaling influences psychiatric risk and can be targeted via interventions at different life stages. Future studies, particularly longitudinal studies, are needed to track circulating eCB concentrations and their impact on mental health across the life span. Studies should investigate how eCB concentrations fluctuate during sensitive developmental periods and explore nonpharmacological approaches that may modulate eCB concentrations and offer therapeutic benefits. Understanding the eCB system’s role in psychiatric risk during development may provide new intervention strategies, improve treatment outcomes, and reduce the burden of psychiatric disorders.
Key points
- The endocannabinoid system has been implicated in many mental health disorders.
- The endocannabinoid system is in flux during critical developmental periods throughout childhood and adolescence, and perturbations to this system from stress during these periods could impact future psychopathology.
- Pharmacological therapies targeting the endocannabinoid system, such as FAAH and MAGL inhibitors, can potentially provide the psychologically relevant benefits of cannabis without the psychoactive or putative neurodevelopmental effects.
- The development of the endocannabinoid system and its effects on child and adolescent psychopathology is a research area ripe for potential clinical translation.
References
- Investigating fatty acid amide hydrolase levels in social anxiety disorder: A positron emission tomography (PET) study using [C‐11]CURB.. Biological Psychiatry,, 2020
- Mechanistic and pharmacological characterization of PF‐04457845: A highly potent and selective fatty acid amide hydrolase inhibitor that reduces inflammatory and noninflammatory pain.. Journal of Pharmacology and Experimental Therapeutics,, 2011. [PubMed]
- Association of Cannabis use during Adolescence with Neurodevelopment.. JAMA Psychiatry,, 2021
- American Psychiatric Association . (2013). Diagnostic and statistical manual of mental disorders. Washington, DC: Author. 10.1176/appi.books.9780890425596
- Stress, sensitive periods and maturational events in adolescent depression.. Trends in Neurosciences,, 2008. [PubMed]
- 2‐Arachidonoylglycerol modulation of anxiety and stress adaptation: From grass roots to novel therapeutics.. Biological Psychiatry,, 2020. [PubMed]
- Selective alterations of endocannabinoid system genes expression in obsessive compulsive disorder.. Translational Psychiatry,, 2024. [PubMed]
- Extinction and beyond: An expanded framework for exposure and response prevention for obsessive‐compulsive disorder.. Frontiers in Psychology,, 2024. [DOI]
- Sexually dimorphic adolescent trajectories of prefrontal endocannabinoid synaptic plasticity equalize in adulthood, reflected by endocannabinoid system gene expression.. Cannabis and Cannabinoid Research,, 2023. [PubMed]
- Chemical probes of endocannabinoid metabolism.. Pharmacological Reviews,, 2013. [PubMed]
- The cingulum bundle: Anatomy, function, and dysfunction.. Neuroscience and Biobehavioral Reviews,, 2018. [PubMed]
- Expression of the CB1 and CB2 receptor messenger RNAs during embryonic development in the rat.. Neuroscience,, 1997
- Prefrontal inputs to the amygdala instruct fear extinction memory formation.. Science Advances,, 2015. [PubMed]
- Re‐examining the link between childhood maltreatment and substance use disorder: A prospective, genetically informative study.. Molecular Psychiatry,, 2021. [PubMed]
- The adolescent brain cognitive development (ABCD) study: Imaging acquisition across 21 sites.. Developmental Cognitive Neuroscience,, 2018. [PubMed]
- Endocannabinoid signaling and synaptic function.. Neuron,, 2012. [PubMed]
- Reduced cellular expression and activity of the P129T mutant of human fatty acid amide hydrolase: Evidence for a link between defects in the endocannabinoid system and problem drug use.. Human Molecular Genetics,, 2004. [PubMed]
- Aerobic exercise and consolidation of fear extinction learning among women with posttraumatic stress disorder.. Behaviour Research and Therapy,, 2021a. [PubMed]
- Exercise‐induced increases in anandamide and BDNF during extinction consolidation contribute to reduced threat following reinstatement: Preliminary evidence from a randomized controlled trial.. Psychoneuroendocrinology,, 2021b. [PubMed]
- Endocannabinoid markers in autism spectrum disorder: A scoping review of human studies.. Psychiatry Research,, 2021. [PubMed]
- Circulating endocannabinoids and genetic polymorphisms as predictors of posttraumatic stress disorder symptom severity: Heterogeneity in a community‐based cohort.. Translational Psychiatry,, 2022. [PubMed]
- A systematic review and meta‐analysis on the effects of exercise on the endocannabinoid system.. Cannabis and Cannabinoid Research,, 2022. [PubMed]
- Genetic variation in endocannabinoid signaling: Anxiety, depression, and threat‐ and reward‐related brain functioning during the transition into adolescence.. Behavioural Brain Research,, 2024. [PubMed]
- Enzymatic synthesis and degradation of anandamide, a cannabinoid receptor agonist.. Biochemical Pharmacology,, 1993. [PubMed]
- Determination and characterization of a cannabinoid receptor in rat brain.. Molecular Pharmacology,, 1988. [PubMed]
- Isolation and structure of a brain constituent that binds to the cannabinoid receptor.. Science,, 1992. [PubMed]
- FAAH genetic variation enhances fronto‐amygdala function in mouse and human.. Nature Communications,, 2015
- Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF‐04457845) in the treatment of cannabis withdrawal and dependence in men: A double‐blind, placebo‐controlled, parallel group, phase 2a single‐site randomised controlled trial.. The Lancet Psychiatry,, 2019. [PubMed]
- Alterations of the endocannabinoid system in adolescents with non‐suicidal self‐injury as a function of childhood maltreatment.. Translational Psychiatry,, 2024. [PubMed]
- The endogenous cannabinoid system and brain development.. Trends in Neurosciences,, 2000. [PubMed]
- Effects of exercise stress on the endocannabinoid system in humans under field conditions.. European Journal of Applied Physiology,, 2012. [PubMed]
- Impact of adverse childhood experience on physical and mental health: A life‐course epidemiology perspective.. Psychiatry and Clinical Neurosciences,, 2022. [PubMed]
- Individual differences in frontolimbic circuitry and anxiety emerge with adolescent changes in endocannabinoid signaling across species.. Proceedings of the National Academy of Sciences of the United States of America,, 2016. [PubMed]
- Endocannabinoid 2‐Arachidonyl glycerol is a full agonist through human type 2 cannabinoid receptor: Antagonism by anandamide.. Molecular Pharmacology,, 2000. [PubMed]
- Putative role of endocannabinoid signaling in the etiology of depression and actions of antidepressants.. Progress in Neuro‐Psychopharmacology and Biological Psychiatry,, 2011. [PubMed]
- Endocannabinoids and stress‐related Neurospsychiatric disorders: A systematic review and meta‐analysis of basal concentrations and response to acute psychosocial stress.. Cannabis and Cannabinoid Research,, 2024. [PubMed]
- Fatty acid amide hydrolase binding is inversely correlated with amygdalar functional connectivity: A combined positron emission tomography and magnetic resonance imaging study in healthy individuals.. Journal of Psychiatry and Neuroscience,, 2021. [PubMed]
- Fatty acid amide hydrolase levels in brain linked with threat‐related amygdala activation.. Neuroimage. Reports,, 2022. [PubMed]
- A cortico‐amygdala neural substrate for endocannabinoid modulation of fear extinction.. Neuron,, 2023. [PubMed]
- Divergent effects of genetic variation in endocannabinoid signaling on human threat‐ and reward‐related brain function.. Biological Psychiatry,, 2009. [PubMed]
- Differential developmental trajectories for CB1 cannabinoid receptor expression in limbic/associative and sensorimotor cortical areas.. Synapse,, 2011. [PubMed]
- Circulating endocannabinoids and N‐acyl ethanolamines are differentially regulated in major depression and following exposure to social stress.. Psychoneuroendocrinology,, 2009. [PubMed]
- Serum endocannabinoid content is altered in females with depressive disorders: A preliminary report.. Pharmacopsychiatry,, 2008. [PubMed]
- Circulating endocannabinoids: From whence do they come and where are they going?. Neuropsychopharmacology,, 2018. [PubMed]
- Serum contents of endocannabinoids are correlated with blood pressure in depressed women.. Lipids in Health and Disease,, 2012. [PubMed]
- Upregulation of CB1 receptors and agonist‐stimulated [35S]GTPγS binding in the prefrontal cortex of depressed suicide victims.. Molecular Psychiatry,, 2004. [PubMed]
- The adolescent brain cognitive development study.. Journal of Research on Adolescence,, 2018. [PubMed]
- Endocannabinoid signaling in social functioning: An RDoC perspective.. Translational Psychiatry,, 2016. [PubMed]
- Acute neurologic disorder from an inhibitor of fatty acid amide hydrolase.. New England Journal of Medicine,, 2016. [PubMed]
- Efficacy and safety of JNJ‐42165279, a fatty acid amide hydrolase inhibitor, in adolescents and adults with autism spectrum disorder: A randomized, phase 2, placebo‐controlled study.. Neuropsychopharmacology,, 2025
- Fractional anisotropy of water diffusion in cerebral white matter across the lifespan.. Neurobiology of Aging,, 2012. [PubMed]
- Expression of CB1 cannabinoid receptor in the anterior cingulate cortex in schizophrenia, bipolar disorder, and major depression.. Journal of Neural Transmission,, 2007. [PubMed]
- Elevated fatty acid amide hydrolase in the prefrontal cortex of borderline personality disorder: A [11C]CURB positron emission tomography study.. Neuropsychopharmacology,, 2020. [PubMed]
- Retrograde inhibition of presynaptic calcium influx by endogenous cannabinoids at excitatory synapses onto Purkinje cells.. Neuron,, 2001. [PubMed]
- Age‐dependent plasticity in endocannabinoid modulation of pain processing through postnatal development.. Pain,, 2017. [PubMed]
- Developmental regulation of fear learning and anxiety behavior by endocannabinoids.. Genes, Brain and Behavior,, 2016. [PubMed]
- From Phytocannabinoids to cannabinoid receptors and endocannabinoids: Pleiotropic physiological and pathological roles through complex pharmacology.. Physiological Reviews,, 2016. [PubMed]
- Selective blockade of 2‐arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects.. Nature Chemical Biology,, 2009. [PubMed]
- Developmental trajectory of the endocannabinoid system in human dorsolateral prefrontal cortex.. BMC Neuroscience,, 2012. [PubMed]
- The social ecology of childhood and early life adversity.. Pediatric Research,, 2021. [PubMed]
- The endocannabinoid system in guarding against fear, anxiety and stress.. Nature Reviews Neuroscience,, 2015
- Adolescent cannabis use and psychosis: Epidemiology and neurodevelopmental models.. British Journal of Pharmacology,, 2010. [PubMed]
- Endocannabinoid dysregulation and PTSD in urban adolescents: Associations with anandamide concentrations and FAAH genotype.. Psychopharmacology., 2024. [DOI]
- Impact of childhood trauma exposure, genetic variation in endocannabinoid signaling, and anxiety on Frontolimbic pathways in children.. Cannabis and Cannabinoid Research,, 2022. [DOI | PubMed]
- Ontogenetic development of cannabinoid receptor expression and signal transduction functionality in the human brain.. European Journal of Neuroscience,, 2003. [PubMed]
- Protective effects of elevated anandamide on stress and fear‐related behaviors: Translational evidence from humans and mice.. Molecular Psychiatry,, 2020
- Elevated anandamide, enhanced recall of fear extinction, and attenuated stress responses following inhibition of fatty acid amide hydrolase: A randomized, controlled experimental medicine trial.. Biological Psychiatry,, 2020. [PubMed]
- Targeting the endocannabinoid system in the treatment of posttraumatic stress disorder: A promising case of preclinical‐clinical translation?. Biological Psychiatry,, 2022. [PubMed]
- Endocannabinoid concentrations in major depression: Effects of childhood maltreatment and relation to hippocampal volume.. Translational Psychiatry,, 2024. [PubMed]
- Age of onset and cumulative risk of mental disorders: A cross‐national analysis of population surveys from 29 countries.. The Lancet Psychiatry,, 2023. [PubMed]
- Endocannabinoid levels in female‐sexed individuals with diagnosed depression: A systematic review.. BMC Women’s Health,, 2024. [PubMed]
- Identification of an endogenous 2‐monoglyceride, present in canine gut, that binds to cannabinoid receptors.. Biochemical Pharmacology,, 1995. [PubMed]
- The role of the endocannabinoid system and genetic variation in adolescent brain development.. Neuropsychopharmacology,, 2018. [PubMed]
- Measuring disturbance of the endocannabinoid system in psychosis: A systematic review and meta‐analysis.. JAMA Psychiatry,, 2019. [PubMed]
- Neurobiological interactions between stress and the endocannabinoid system.. Neuropsychopharmacology,, 2016. [PubMed]
- Monoacylglycerol lipase inhibition in Tourette syndrome: A 12‐week, randomized, controlled study.. Movement Disorders,, 2021. [PubMed]
- Endocannabinoid modulation using monoacylglycerol lipase inhibition in Tourette syndrome: A phase 1 randomized, placebo‐controlled study.. Pharmacopsychiatry,, 2022. [PubMed]
- Molecular characterization of a peripheral receptor for cannabinoids.. Nature,, 1993
- Endocannabinoid reactivity to acute stress: Investigation of the relationship between salivary and plasma levels.. Biological Psychology,, 2021. [PubMed]
- Hair endocannabinoids predict physiological fear conditioning and salivary endocannabinoids predict subjective stress reactivity in humans.. Psychoneuroendocrinology,, 2023. [PubMed]
- Cannabinoid polymorphisms interact with plasma endocannabinoid levels to predict fear extinction learning.. Depression and Anxiety,, 2021. [PubMed]
- Effect of rimonabant on progression of atherosclerosis in patients with abdominal obesity and coronary artery disease: The STRADIVARIUS randomized controlled trial.. JAMA,, 2008. [PubMed]
- Genetic variation of the 5‐HT1A rs6295, 5‐HT2A rs6311, and CNR1 rs1049353 and an altered endocannabinoid system in depressed patients.. Brain and Behavior: A Cognitive Neuroscience Perspective,, 2023
- Development of the uncinate fasciculus: Implications for theory and developmental disorders.. Developmental Cognitive Neuroscience,, 2015. [PubMed]
- Mental health and well‐being from childhood to adulthood: Design, methods and results of the 11‐year follow‐up of the BELLA study.. European Child & Adolescent Psychiatry,, 2021. [PubMed]
- The effects of FAAH inhibition on the neural basis of anxiety‐related processing in healthy male subjects: A randomized clinical trial.. Neuropsychopharmacology,, 2021. [PubMed]
- Resilience to substance use disorder following childhood maltreatment: Association with peripheral biomarkers of endocannabinoid function and neural indices of emotion regulation.. Molecular Psychiatry,, 2023. [PubMed]
- Endocannabinoids, cannabinoids and the regulation of anxiety.. Neuropharmacology,, 2021. [PubMed]
- Trauma and US minority children and youth.. Current Psychiatry Reports,, 2022. [PubMed]
- Physical activity interventions to alleviate depressive symptoms in children and adolescents: A systematic review and meta‐analysis.. JAMA Pediatrics,, 2023. [PubMed]
- Roadmap for the expression of canonical and extended endocannabinoid system receptors and metabolic enzymes in peripheral organs of preclinical animal models.. Physiological Reports,, 2024. [PubMed]
- Fatty acid ethanolamide levels are altered in borderline personality and complex posttraumatic stress disorders.. European Archives of Psychiatry and Clinical Neuroscience,, 2014. [PubMed]
- Endocannabinoids at the synapse and beyond: Implications for neuropsychiatric disease pathophysiology and treatment.. Neuropsychopharmacology,, 2023. [PubMed]
- The effects of inhibition of fatty acid amide hydrolase (FAAH) by JNJ‐42165279 in social anxiety disorder: A double‐blind, randomized, placebo‐controlled proof‐of‐concept study.. Neuropsychopharmacology,, 2021
- Anatomy and function of the fornix in the context of its potential as a therapeutic target.. Journal of Neurology, Neurosurgery & Psychiatry,, 2020. [PubMed]
- dbSNP‐database for single nucleotide polymorphisms and other classes of minor genetic variation.. Genome Research,, 1999. [PubMed]
- The neurocircuitry of fear, stress, and anxiety disorders.. Neuropsychopharmacology,, 2010. [PubMed]
- Genetic variation in endocannabinoid signaling is associated with differential network‐level functional connectivity in youth.. Journal of Neuroscience Research,, 2022. [PubMed]
- Rare genetic variants in the endocannabinoid system genes CNR1 and DAGLA are associated with neurological phenotypes in humans.. PLoS One,, 2017. [PubMed]
- Plasma endocannabinoid levels in patients with borderline personality disorder and healthy controls.. International Journal of Molecular Sciences,, 2023
- Endocannabinoid system reactivity during stress processing in healthy humans.. Biological Psychology,, 2022. [PubMed]
- FAAH polymorphism (rs324420) modulates extinction recall in healthy humans: An fMRI study.. European Archives of Psychiatry and Clinical Neuroscience,, 2022. [PubMed]
- An analysis of endocannabinoid concentrations and mood following singing and exercise in healthy volunteers.. Frontiers in Behavioral Neuroscience,, 2018. [DOI | PubMed]
- Cannabinoid receptor CNR1 expression and DNA methylation in human prefrontal cortex, hippocampus and caudate in brain development and schizophrenia.. Translational Psychiatry,, 2020. [PubMed]
- Activity‐based protein profiling reveals off‐target proteins of the FAAH inhibitor BIA 10‐2474.. Science,, 2017. [PubMed]
- Preferential limbic expression of the cannabinoid receptor mRNA in the human fetal brain.. Neuroscience,, 2003. [PubMed]
- Endogenous cannabinoids mediate retrograde signalling at hippocampal synapses.. Nature,, 2001. [PubMed]
- Reduced levels of the endocannabinoid arachidonylethanolamide (AEA) in hair in patients with borderline personality disorder – A pilot study.. Stress,, 2018. [PubMed]
- Lasting impacts of prenatal cannabis exposure and the role of endogenous cannabinoids in the developing brain.. Future Neurology,, 2011. [PubMed]
- Genetic variations in the retrograde endocannabinoid signaling pathway in Chinese patients with major depressive disorder.. Frontiers in Neurology,, 2023. [DOI]
- A common genetic variant in fatty acid amide hydrolase is linked to alterations in fear extinction neural circuitry in a racially diverse, nonclinical sample of adults.. Journal of Neuroscience Research,, 2022. [PubMed]
- Impact of endocannabinoid system manipulation on neurodevelopmental processes relevant to schizophrenia.. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging,, 2021. [PubMed]
