The influence of stress and social defeat on neurobiological reinforcement mechanisms across reward to withdrawal in nicotine addiction
Abstract
Nicotine and cigarette/tobacco use continue to be a prevalent public health issue worldwide. The transition to nicotine addiction occurs through an allostatic cycle involving the stages of binging/intoxication, withdrawal/negative affective states, and preoccupation/anticipation. This review focuses on the psychological, neurobiological, and molecular mechanisms contributing to the negative affective state during withdrawal from nicotine with an emphasis on stress and how social defeat stress can affect these mechanisms. Psychologically, negative affect during withdrawal is thought to contribute to the transition from positive reinforcement of drug-taking to negative reinforcement of nicotine use. Nicotine binding to nicotinic acetylcholine receptors elicits a variety of neuronal signaling throughout the brain, over time producing within- and between-systems neuroadaptations across brain regions that govern reward, anxiety, pain, and stress responses. Continued nicotine use additionally dysregulates myriad molecular signaling pathways that directly affect nicotine intake/aversion and withdrawal-like symptoms. Throughout all of these mechanisms, non-pharmacological stress also plays an important role in mediating much of the negative affect associated with addiction. Social defeat stress increases a variety of neuropeptide signaling that consequently exacerbates drug taking and negative affective states. Understanding the mechanisms through which these stages manifest can better our understanding of addiction disease biology and provide novel avenues for therapeutic targets.
Article type: Review Article
Keywords: Withdrawal, CRF, VTA, Dopamine, NAChR, Stress
Affiliations: https://ror.org/0168r3w48grid.266100.30000 0001 2107 4242Department of Psychiatry, University of California San Diego School of Medicine, 3272 Skaggs Pharmaceutical Sciences Building, 9500 Gilman Dr, La Jolla, CA 92093 USA; https://ror.org/03m1g2s55grid.479509.60000 0001 0163 8573Sanford Burnham Prebys Medical Discovery Institute, La Jolla, CA 92037 USA; https://ror.org/043hw6336grid.462486.a0000 0004 4650 2882Institut de Neurosciences de la Timone, Aix-Marseille Université, 13005 Marseille, France
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s00213-025-06852-5 | PubMed: 40715842 | PMC: PMC12578779
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.8 MB)
Introduction
Despite increased policy and advocacy efforts over the decades, tobacco use remains the leading cause of preventable death and disease in the United States. Not only do almost half a million people die annually from tobacco use disorder (CDCTobaccoFree ref. 2021), but cigarette smoking is also a significant factor in deaths caused by other respiratory diseases like lung cancer and chronic obstructive pulmonary disease (Current Cigarette Smoking Among Adults — United States ref. 2005). Furthermore, cigarette smoking and tobacco use disorder cost the country $289 billion per year in associated healthcare costs and productivity loss (Key Substance Use and Mental Health Indicators in the United States: Results from the 2022 National Survey on Drug Use and Health ref. 2022). In an effort to curb many of the negative health outcomes associated with tobacco use, electronic cigarette use has grown rapidly in recent years. While adolescents and young adults often view these as safer alternatives due to the lack of carcinogens like tar that are produced by traditional cigarettes, electronic cigarettes still pose significant risks to lung and cardiovascular health (Farsalinos and Polosa ref. 2014; Hua and Talbot ref. 2016) in the non-smoking population. E-cigarettes are also more popular among adolescents compared to traditional cigarettes, and both human and rodent studies have shown that initiation of e-cigarette use in adolescence can potentiate cigarette/tobacco use in adulthood (Barrington-Trimis et al. ref. 2016; Kallupi et al. ref. 2019).
However, the largest outcome of using these products is nicotine addiction. Nicotine is the primary psychoactive component of tobacco and is the motivating factor for people to use both traditional tobacco products and e-cigarettes (Foll et al. ref. 2022; Markou ref. 2008). Nicotine addiction can be defined as “a chronic, relapsing disorder that has been characterized by a compulsion to seek and take nicotine, loss of control over nicotine intake, and emergence of a negative emotional state (e.g., dysphoria, anxiety, and/or irritability) that defines a motivational withdrawal syndrome when access to nicotine is prevented” (Koob and Moal ref. 2008). This is summarized as the now established three-stage cycle, comprising phases of binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation (Koob ref. 2011). However, this cycle is allostatic – rather than maintaining homeostasis in brain circuitry, over time this cycle worsens; the brain must constantly work to operate at lower hedonic affective set points (Koob and Moal ref. 2001; Koob and Schulkin ref. 2019). This allostasis also represents the transition from drug taking as positive reinforcement to drug taking as negative reinforcement to avoid or mitigate the negative affective state. Factors outside of nicotine intake can also serve as positive and negative reinforcers that contribute to the emergence of the negative affective state. The social aspects of smoking like social bonding or imitating loved ones, for example, can serve as a positive reinforcer of nicotine use (Urbán ref. 2010; Vink et al. ref. 2003; NIDA Research Monograph ref. 1976). Non-pharmacological stressors, including social defeat, stressful situations, or environmental stimuli, can serve as powerful negative reinforcement of smoking behavior (Ansell et al. ref. 2012; Sinha ref. 2008).
A critical goal in studying nicotine addiction is to identify new avenues to aid nicotine cessation in patients. Less than 30% of smokers who quit use FDA-approved medication, highlighting the need for increased awareness and access to these medications. Current therapeutics for nicotine cessation are effective, with varenicline being the most effective FDA-approved cessation therapy, doubling or tripling the odds of quitting after a year (Shang et al. ref. 2023; Ebbert et al. ref. 2015). However, long-term abstinence remains a challenge, and only 25% of smokers remain abstinent after 1 year, highlighting the need to improve current treatment strategies. Understanding the factors that drive nicotine withdrawal and negative affect can allow for more efficacious therapeutic development. The primary goal of this review is to examine psychological, neurobiological, and molecular contributions to the negative affective state during nicotine addiction. This review will also touch upon the role of non-pharmacological stress in initiating and potentiating the affective states of nicotine addiction.
Transition from positive to negative reinforcement
Reinforcement plays a key role in nicotine addiction as many behaviors are due to responses to specific stimuli, or reinforcers, that increase the probability of these behaviors – in this case, drug taking. Positive reinforcement is defined as the probability of a response or behavior increasing following presentation of a stimulus; conversely, negative reinforcement occurs when the probability of a response is increased through removal of an aversive stimulus (Koob et al. ref. 2014). Nicotine serves as the positive reinforcing stimulus, and the negative affective state during nicotine withdrawal serves as the negative reinforcing stimulus (Watkins et al. ref. 2000; George and Koob ref. 2017).
Nicotine, compared to other drugs of abuse, has a much narrower range of doses that are considered appetitive. Even moderate doses can be aversive or punishing, especially in nondependent subjects (Fowler and Kenny ref. 2014; Koffarnus and Winger ref. 2015). Acute nicotine intake produces increased anxiogenic-like behaviors, increases in the circulating stress biomarkers adrenocorticotropin-releasing hormone and cortisol, and unpleasant physiological responses like cough or nausea (Casarrubea et al. ref. 2015; Newhouse et al. ref. 1990). The severity of these aversive reactions has been shown to affect the development of chronic nicotine use, with those experiencing more aversive effects after first use less rapidly transitioning to regular use (Sartor et al. ref. 2010). In preclinical models of self-administration, nicotine doses are carefully titrated to prevent nicotine from serving as an aversive stimulus (Donny et al. ref. 1998). Through developing tolerance to the noxious effects of nicotine, habitual use can be established (Fowler and Kenny ref. 2014; Russell ref. 1979).
Nicotine taking as positive reinforcement
While acute nicotine initially produces aversive responses, it also produces acutely pleasurable effects on mood and cognition in humans, including mild euphoria, increased energy and focus, and increased arousal (Pomerleau and Pomerleau ref. 1992a; Stolerman and Jarvis ref. 1995; Benowitz ref. 1996). These positive effects can increase the likelihood of users repeatedly consuming nicotine. In fact, it was shown in a trial of smokers that subjects displayed greater positive reinforcement learning following cigarette consumption (Baker et al. ref. 2020). The acute positive reinforcing effects of nicotine are important in establishing self-administration behavior. Smoking is also used for relaxation in response to stress (Benowitz ref. 1988). Evidence from human and non-human animal studies points to various stressors throughout early life, adolescence, and adulthood as contributors to increased drug abuse either through earlier initiation of use, faster transition to increased use and/or binge patterns, and increased positive reinforcement of substance use (Torres and O’Dell ref. 2016; Kirsch and Lippard ref. 2022; Ahmed et al. ref. 2020).
Rodent models of intravenous nicotine self-administration have been reliably used for decades as they elicit positive reinforcement that consequently produces increased intake over time (Corrigall ref. 1992; Donny et al. ref. 1995). This robust increase in intake is seen especially in models that use intermittent extended access to nicotine self-administration, leading to increased motivation for drug-taking (Cohen et al. ref. 2012). It has been shown in both human and rodent tests that increased nicotine reinforcement is strongly positively correlated with cue-induced craving (humans) and cue-induced reinstatement (rodents) (Butler et al. ref. 2021).
The social defeat stress model pioneered by Dr. Klaus Miczek has become another valuable tool to study the contribution of stress on drug-taking and positive reinforcement, serving as a method of “behavioral sensitization” to stimulants or other drugs of abuse (Covington and Miczek ref. 2001). Miczek and colleagues’ seminal studies in this field have shown that intermittent and chronic stress exposure through this model has persistent effects on binge intake of the psychostimulant cocaine, shortens the latency to acquire drug, and increases motivation for drug (Covington and Miczek ref. 2001; Covington et al. ref. 2005; Miczek et al. ref. 2004). This model of social defeat stress has been adapted to investigate nicotine use, and interestingly, studies using this model have found that nicotine administered after exposure to social defeat stress may actually confer some resilience to stress in rodents (Parise et al. ref. 2020; Zou et al. ref. 2014). Throughout this review, we will incorporate the role of social defeat stress and external stressors in perpetuating the negative affective state and related behaviors.
Nicotine taking as negative reinforcement
Nicotine may be exerting its addictive effects through the transition from positive to negative reinforcement, as well as the shift from impulsive to compulsive behavior (Fig. 1). Impulsivity is defined as “a predisposition toward rapid, unplanned reactions to internal and external stimuli without regard for the negative consequences of these reactions to themselves or others” (Moeller et al. ref. 2001). Impulsivity is present at the early stages of addiction, and it is thought to be produced by positive reinforcement mechanisms. Feelings of intense arousal and excitement prior to the stimulus, as well as feelings of euphoria, reward, and pleasure after the behavior is performed, are the primary drivers of positive reinforcement (Perkins et al. ref. 2003; Pomerleau and Pomerleau ref. 1992b). Smokers have been shown to exhibit increased impulsive intake of nicotine compared to nonsmokers (Mitchell ref. 2004; Field et al. ref. 2006). However, there are often feelings of guilt or regret following the behavior, as drugs are normally taken for their initial pleasurable effects but potential negative consequences are not taken into account (Yang et al. ref. 2019). Over time, behavioral and neural adaptations shift drug taking to a negative reinforcement mechanism. Anxiety, stress, and pain prior to the behavior promote negative reinforcement, and once the behavior is performed, individuals experience relief from the negative emotional state (Pang et al. ref. 2014). Compulsivity, defined as “perseverative, repetitive actions that are excessive and inappropriate,” takes over from impulsivity in the later stages of addiction (Berlin and Hollander ref. 2014). This compulsivity has been historically thought to drive escalation of drug intake despite adverse consequences, although compulsivity as a measure is far more complex and cannot provide the whole picture of negative reinforcement in addiction (George et al. ref. 2022).
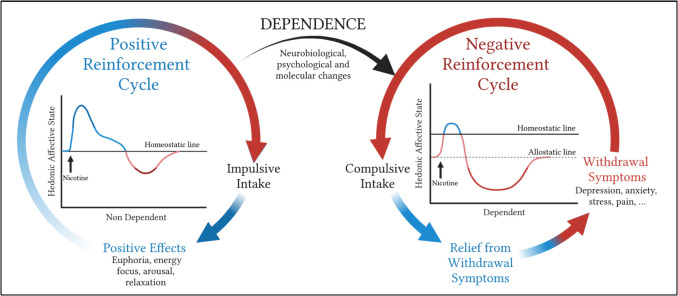
Chronic nicotine use produces robust withdrawal or negative affective behaviors in both humans and nonhuman animal models (Chellian et al. ref. 2021; Hamilton et al. ref. 2009; Hughes et al. ref. 1994; Malin and Goyarzu ref. 2009; Stoker et al. ref. 2008; O’dell et al. ref. 2004). Reported in the DSM-V, acute symptoms in humans include depression, anxiety, irritability, dysphoria, stress, as well as somatic symptoms like bradycardia and increased appetite; in protracted abstinence, users can experience longer-lasting symptoms of depressed mood, irritability, increased stress (McLaughlin et al. ref. 2015; Diagnostic and Statistical Manual of Mental Disorders ref. n.d.). In rodent models, physical symptoms of nicotine withdrawal are characterized by somatic signs like body shakes, ptosis, teeth chattering, etc (Ponzoni et al. ref. 2015; Damaj et al. ref. 2003; Grabus et al. ref. 2005). Social defeat stress can facilitate these symptoms, including nicotine-induced locomotor suppression (Domingues et al. ref. 2019). Withdrawal also induces changes in motivation as measured by behavioral tests like intracranial self-stimulation and conditioned place aversion, and importantly changes in affect as seen using open-field tests that measure anxiety-like behaviors, depression-like behaviors, and anhedonia-like behaviors (Epping-Jordan et al. ref. 1998; Kim and Im ref. 2024; Torres et al. ref. 2013; Wang et al. ref. 2018; Jackson et al. ref. 2013; Bruijnzeel and Markou ref. 2004). These behaviors are most often observed in models of extended access and/or chronic use of passive or self-administered nicotine vs. acute intake (Cohen and George ref. 2013). We have observed that within a novel rodent model of nicotine vapor self-administration, 3 weeks of 1 h self-administration sessions was sufficient to produce increased somatic signs of withdrawal and hyperalgesia precipitated by mecamylamine-induced withdrawal, as well as anxiety-like behaviors following protracted abstinence, similar to effects seen in both human smokers and intravenously self-administering rodents (Smith et al. ref. 2020). In these models, an abstinence period of just 2–3 days is sufficient to trigger withdrawal-like behaviors in rodents, and consequently increase self-administration of nicotine when access to drug is restored (Cohen et al. ref. 2012; O’dell et al. ref. 2007). This finding mimics human behavior, as after an abstinence period, people smoke more (Isaac and Rand ref. 1972; Nil et al. ref. 1987; Rusted et al. ref. 1998).
It is believed that withdrawal symptoms drive the shift to negative reinforcement – the motivation for nicotine intake changes to avoid these negative affective behaviors, rather than to seek out pleasurable effects. In fact, it has been shown that negative affective symptoms of anxiety-like behavior and hyperalgesia induced by nicotine abstinence predicted subsequent nicotine intake when access was restored (Cohen et al. ref. 2015). In human studies, there is a similar link between negative affect and smoking, as assessed by a meta-analysis of clinical data (Akbari et al. ref. 2020). This data shows that severity of these behaviors is a key factor in dependent animals’ excessive nicotine intake. While we have described the transition from positive to negative reinforcement from a psychological lens, negative affect can additionally be explained by neurobiological and molecular changes in the mechanisms of motivation.
Neurobiological mechanisms of negative affect
Nicotine’s mechanism of action comes from binding to neuronal nicotinic acetylcholine receptors (nAChRs), which are present ubiquitously in various conformations throughout the brain. Specifically, neuronal nAChRs are heteropentameric proteins consisting of various conformations of alpha- and beta-subunits (α2-α10 and β2-β4) (Changeux et al. ref. 1998; Vidal and Changeux ref. 1996), all of which produce different pharmacological properties in the brain. These receptors act as ligand-gated ion channels which primarily regulate endogenous acetylcholine signaling but also have affinity for exogenous nicotine (Changeux ref. 2010). When nicotine binds to these receptors, there is increased permeability of ions which serve to depolarize the cell and lead to neuronal firing.. nAChRs, and specific subunits, play critical roles in all components of addiction, from positive reinforcement to negative affect, and nAChR subunits have even been shown to mediate both positive and negative effects of nicotine. The α4, α5, α6, and β2 subunit-containing receptors are critical for mediating the positive reinforcing effects of nicotine (Changeux ref. 2010; Jackson et al. ref. 2008; Picciotto et al. ref. 1998; Wigestrand et al. ref. 2011; Brunzell et al. ref. 2015). Continued nicotine use also leads to upregulation of nAChRs due to receptor desensitization by nicotine (Henderson and Lester ref. 2015).
The mesocorticolimbic system as a positive reinforcer of nicotine intake
The reinforcing effects of nicotine are primarily driven by the mesocorticolimbic system, comprising the basal ganglia (including the nucleus accumbens (NAc) and striatum) and ventral tegmental area (VTA). Within the VTA, nAChRs are found on dopamine-producing neurons; when stimulated by nicotine, these cells release dopamine to downstream sites including the NAc, striatum, and prefrontal cortex (Xiao et al. ref. 2020; Faure et al. ref. 2014). Dopamine release is an important mechanism that enhances and sustains the motivation for and reinforcement of drug-taking behavior (Volkow et al. ref. 2017). With continued nicotine use, dopaminergic neurons become desensitized and have reduced firing during withdrawal, which contributes to the negative affective state (Wills et al. ref. 2022). Stress has been shown to activate dopaminergic signaling in the VTA and lead to increased drug-taking in rodent models of multiple drugs of abuse, including nicotine (Holly and Miczek ref. 2016; Morel et al. ref. 2018; Ortiz et al. ref. 2022; Adams et al. ref. 2021). Furthermore, they showed that social defeat stress alters mesocorticolimbic dopamine release that activates a VTA-accumbens-prefrontal cortex-amygdala neural circuit (Miczek et al. ref. 2008; Yap and Miczek ref. 2008). Both acute stressors and repeated stress exposure can induce persistent neuroadaptations in VTA dopaminergic signaling that can consequently alter addiction-like behaviors (Holly and Miczek ref. 2016; Tidey and Miczek ref. 1996).
nAChR signaling within withdrawal and negative affect
nAChRs mediate the physical and affective components of nicotine withdrawal differently. The β2 subunit is necessary for the anxiogenic and aversive effects of nicotine withdrawal, as demonstrated by β2 knockout mice exhibiting losses of anxiety-like behaviors, conditioned place aversion, and withdrawal-induced deficits in contextual fear conditioning (Jackson et al. ref. 2008; Jackson et al. ref. 2009a; Portugal et al. ref. 2008). The α6 subunit also plays a role in the anxiety-like symptoms of nicotine withdrawal (Jackson et al. ref. 2009b). The α3β4 receptor and α5 subunit independently mediate physical signs of withdrawal, as shown by a decrease in somatic signs of withdrawal and hyperalgesia following selective blockade of these receptors (Jackson et al. ref. 2013). The α7 receptor, which exhibits more rapid desensitization upon nicotine binding, is involved in producing somatic signs of withdrawal. Knockout of α7 subunits has been shown to delay self-stimulation (Stoker et al. ref. 2008; Salas et al. ref. 2007), potentially also giving it a role in the anhedonic behaviors associated with negative affect during withdrawal. α7 increases sensitivity to social defeat stress in female rats, which may increase vulnerability to addiction (Ortiz et al. ref. 2022). Additional information about the role of nAChR subtypes in mediating different aspects of nicotine addiction can be found in a comprehensive review by Picciotto and Kenny (ref. 2021).
Given the wide prevalence of nAChRs across most brain regions, other neurotransmitter systems are also involved in contributing to positive reinforcement and/or negative affective behaviors. Acetylcholine is the primary endogenous ligand for neuronal nAChRs. Acetylcholine as a neuromodulator is known to increase release of additional neurotransmitters, promote or suppress neuronal firing, and contribute to synaptic plasticity (Picciotto et al. ref. 2012). Endogenous acetylcholine signaling has been shown to activate the dopaminergic signaling system, similar to exogenous nicotine (Maskos ref. 2010; Clarke et al. ref. 1987; Zhou et al. ref. 2001). It was shown that upon administration of a positive allosteric modulator for the α7 nAChR subunit, endogenous acetylcholine potentiation was sufficient to attenuate precipitated nicotine withdrawal as measured by somatic signs of withdrawal in mice (Jackson et al. ref. 2018).
The extended amygdala
With continued nicotine use, within-system neuroadaptations occur which produce a decrease in dopamine signaling within the mesocorticolimbic pathway, leading to the overall reduction in brain reward function that may contribute to the negative affect stage of the addiction cycle. Along with within-system neuroadaptations, between-system neuroadaptations play an important role in the development of the negative affective state. The primary circuit involved in withdrawal and negative affect is the extended amygdala, consisting of the central nucleus of the amygdala (CeA), bed nucleus of the stria terminalis (BNST), and a portion of the NAc shell (Koob and Volkow ref. 2010) (Fig. 2). These brain regions are known to play a key role in emotional processing and stress-related behaviors. Mineur et al. showed that within the amygdala, both α7 and β2 subunits were important for different stress-mediated behaviors, with α7 knockout mice displaying reduced anxiety-like and depressive-like behaviors, and β2 knockout mice exhibiting decreased resistance to social defeat stress (Mineur et al. ref. 2016). Chronic nicotine exposure has been shown to facilitate long-term potentiation within the amygdala, and this requires α7 and β2 subunits (Huang et al. ref. 2008).
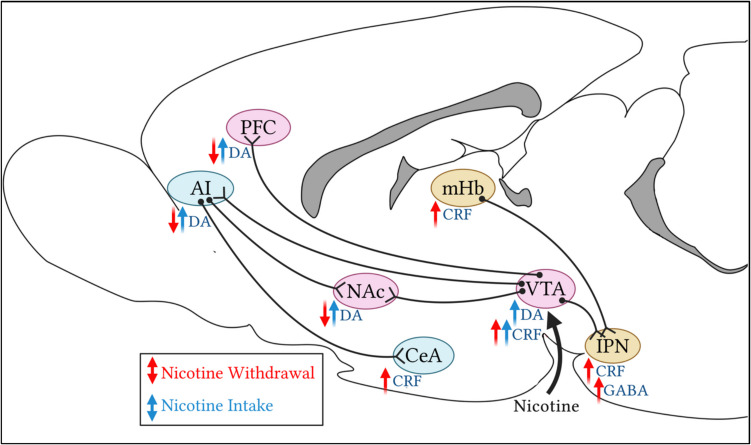
The anterior insular cortex
The anterior insular cortex, with afferents to the NAc and extended amygdala and dopaminergic terminals from the VTA, is another prime candidate influenced by nicotine addiction (Ibrahim ref. 2019). Nicotine has been shown to activate the insular cortex, and damage to this region has been shown to lead to reduced withdrawal symptoms, craving for nicotine, and even relapse (Naqvi et al. ref. 2007). The insula is believed to integrate sensory and cognitive signals as well as modulate the salience network. During acute abstinence from nicotine, studies have shown that the insula becomes flooded with craving signals, and signaling to downstream networks is enhanced, suggesting the insula’s role in producing the motivation to obtain the drug within the context of negative reinforcement (Naqvi and Bechara ref. 2009; Lerman et al. ref. 2014; Regner et al. ref. 2019). Imaging studies looking at the function of the insula during craving have reported increased activation in chronic nicotine users when presented with cues, and smoking consequently decreased connectivity of the insula to the anterior cingulate cortex (Goudriaan et al. ref. 2010; Faulkner et al. ref. 2019). While studies have not explored the direct role of CRF signaling within the insula in mediating negative affect in nicotine addiction, CRF signaling within the insula is known to mediate other types of addiction (Goudriaan et al. ref. 2010; Rotge et al. ref. 2017; Sánchez et al. ref. 1999; Martin-Fardon et al. ref. 2010; Cottone et al. ref. 2009).
The habenula-interpeduncular nucleus circuit
Another circuit highly susceptible to nAChR signaling is the habenular-interpeduncular nucleus axis. The habenula is divided into the lateral habenula and medial habenula, which exhibit different actions within nicotine addiction. The lateral habenula contains primarily α6 nAChR subunits, and it receives inputs from the VTA and sends outputs to dopaminergic neurons; activation of the lateral habenula decreases downstream dopaminergic signaling, and following chronic nicotine, this activation is no longer present, suggesting a role for the lateral habenula in regulating reward-related activity (Pierucci et al. ref. 2022; Matsumoto and Hikosaka ref. 2007; Paolini and Biasi ref. 2011; Zuo et al. ref. 2016). The medial habenula projects primarily to the interpeduncular nucleus (IPN). This circuit is densely populated with α3, α5, and β4 nAChR subunits that were shown above to be necessary for the physical symptoms of nicotine withdrawal, suggesting that this pathway may be mediating withdrawal-like behaviors (Salas et al. ref. 2009; Fowler et al. ref. 2011). The medial habenula has been shown to regulate aversion to nicotine, including through expression of the β4 subunit (Frahm et al. ref. 2011; Elayouby et al. ref. 2021; Ślimak ref. 2014). Medial habenular α5 nAChR subunits also regulate nicotine intake, as Fowler et. al found that α5 knockdown in mice in this region dose-dependently inhibited brain reward thresholds, and subsequent disruption of the IPN in these models led to increased nicotine intake (Fowler et al. ref. 2011). In the IPN, expression of the α2 subunit was shown to be important for fear conditioning, another affective symptom of withdrawal (Ishii et al. ref. 2005; Lotfipour et al. ref. 2013). Within the IPN, these withdrawal-like behaviors are mediated by GABAergic neuron activation specifically induced by nicotine withdrawal in dependent animals (Avelar and Gearge ref. 2022; Zhao-Shea et al. ref. 2013).
Between-systems neuroadaptations within negative affect
Corticotropin–releasing factor and stress signaling
Along with within-system neuroadaptations, between-system neuroadaptations play a large role in the shift to negative reinforcement. One of these between-system neuroadaptations is the extrahypothalamic stress system. Nicotine activates the hypothalamic–pituitary–adrenal (HPA) axis – the major stress signaling pathway in the brain and body – and causes the subsequent release of corticotropin-releasing factor (CRF) from the hypothalamus (Matta et al. ref. 1998; Okada et al. ref. 2003). Acute nicotine has been shown to be a potent activator of the HPA axis, causing the elevation of brain CRF and circulating adrenocorticotropic hormone and cortisol/corticosterone (Matta et al. ref. 1998; Steptoe and Ussher ref. 2006). Reciprocally, acute stress has been widely anecdotally cited and shown in laboratory settings to decrease resistance to smoking and cause more intense nicotine use in regular smokers (McKee et al. ref. 2011; Al’Absi ref. 2006). Like other drugs of abuse, nicotine activates CRF signaling in many regions of the brain, and this overactivation of CRF signaling may be responsible for many of the stress-related behaviors seen in withdrawal. Nicotine withdrawal produces increases in extracellular CRF levels in the CeA, and antagonism of the CRF receptors has been shown to block anxiety-like behavior and deficit in brain reward function seen with precipitated nicotine withdrawal in rodents (George et al. ref. 2007; Bruijnzeel et al. ref. 2007; Heinrichs ref. 2003; Heinrichs and Koob ref. 2004). Since then, many studies have confirmed the role of CRF in withdrawal-like behaviors through various manipulation studies within the extended amygdala (Cohen et al. ref. 2015; Bruijnzeel et al. ref. 2007; Bruijnzeel et al. ref. 2009; Qi et al. ref. 2014; Qi et al. ref. 2016).
The habenula-IPN pathway is also influenced by the CRF system (Grieder et al. ref. 2014; Zhao-Shea et al. ref. 2015). The IPN receives inputs from the VTA, and activation of this VTA-IPN circuit has been implicated in promoting anxiety-like behaviors through increased CRF signaling from the VTA (Zhao-Shea et al. ref. 2015). Grieder et al. found that dopaminergic neurons in the VTA that also express CRF have been shown to upregulate expression of CRF encoding-mRNA following chronic nicotine exposure. Virus-mediated downregulation of CRF-encoding mRNA in the VTA, as well as pharmacological antagonism of CRF1 receptors within the IPN, not only prevented withdrawal-induced negative affective state, but also prevented increased nicotine intake following an abstinence period. Building upon this work, Zhao-Shea et al. identified a meso-interpeduncular circuit in which VTA CRF neurons innervate both medial habenula glutamatergic cells and CRF1-expressing IPN neurons, leading to increased anxiety during nicotine withdrawal in rodents (Zhao-Shea et al. ref. 2015). We have further characterized the role of CRF in nicotine addiction in a previous review (Simpson et al. ref. 2020).
External stress can also influence CRF signaling in the VTA. Miczek and colleagues have shown that social defeat stress elevates CRF levels in the VTA, increasing activation of dopaminergic neurons in the VTA and leading to heightened dopamine signaling in the NAc (Holly et al. ref. 2015). This signaling cascade sensitizes the reward pathway, making rodents more prone to substance-seeking behavior (Holly et al. ref. 2016; Leonard et al. ref. 2017; Hwa et al. ref. 2016; Albrechet-Souza et al. ref. 2015). Antagonism of the CRF1 receptor selectively in the VTA blocked these CRF-induced effects and prevents the escalation of drug-seeking behavior seen after social stress (Han et al. ref. 2017; Burke et al. ref. 2016). CRF signaling in the VTA is a crucial mediator in the link between stress and vulnerability to substance use disorder, opening the door to target CRF signaling in the VTA as a potential therapeutic strategy.
Opioid receptor signaling
Another critical between-system neuroadaptation altered by nicotine use is endogenous opioid signaling. The mu opioid receptor (MOR) is implicated in both positive reinforcement and negative affective behaviors through its regulation of behavioral sensitization and analgesia. Repeated social defeat stress has been shown to increase MOR expression in the VTA, and these receptors are located on GABAergic neurons; the subsequent decreased opioid signaling reduces inhibition of VTA dopaminergic neurons (Nikulina et al. ref. 2008; Johnson and North ref. 1992). MOR agonism following social defeat stress produces increased locomotor activity, further implicating a disinhibition of dopaminergic cells (Nikulina et al. ref. 2005). MOR antagonism has been shown to elicit nicotine withdrawal symptoms in rodents (Biała et al. ref. 2005; Malin et al. ref. 1993), suggesting that this signaling system is needed for the reinforcing and rewarding effects of nicotine (Norman and D’Souza ref. 2017). Dynorphin, the primary endogenous activator for the kappa opioid receptor (KOR), has been shown to increase in the brain following acute nicotine administration (Hadjiconstantinou and Neff ref. 2011). This activation persists with chronic nicotine use and leads to increased anxiety-like behavior, decreased dopaminergic signaling, and increased receptor desensitization (Tejeda et al. ref. 2012; McCarthy et al. ref. 2010). KOR activation has also been shown to produce a stress-like state that may contribute to aversive affective behaviors and increase the propensity for reinstatement of drug-taking following abstinence (Wee and Koob ref. 2010).
Whole brain analysis to integrate signaling changes within nicotine withdrawal
Finally, while many of these canonical regions have been studied discretely or within limited circuits, the use of whole-brain analysis techniques allows for a more complete understanding of the brain-wide neurobiological shifts occurring during withdrawal. We have shown using iDISCO whole-brain clearing and FOS immunolabeling in mice that neural networks of healthy brains exhibit a high degree of modularity, but during nicotine withdrawal, brain connectivity increases to reduce the number of network modules to five key modules connecting almost every brain region. The largest modules are driven by cortical and extended amygdalar regions, which confirm that these regions drive the negative reinforcement and negative affect associated with withdrawal. We have further shown that these modularity changes correlate with increased expression of nAChR subunit genes and alteration of long-range cholinergic networks (Carrette et al. ref. 2023). Understanding the changes to whole brain functional connectivity can guide more detailed analyses of brain regions, as well as identify previously unexplored regions that may be contributing to nicotine withdrawal.
Molecular mechanisms of negative affect
Nicotine cessation strategies are variably effective, with approaches targeting the spectrum from nAChR desensitization to alleviating withdrawal symptoms. There are many behavioral treatments that try to target the rewarding effects of nicotine, either through decreasing the drug-related reward such as in cognitive behavioral therapy, or through increasing responses to natural reward such as in contingency management (Wardle et al. ref. 2024). Additionally, now outdated strategies like rapid-smoking have tried to improve cessation through increasing aversion to nicotine intake (Hajek and Stead ref. 2001; Tiffany et al. ref. 1986). Current FDA-approved strategies target nicotine withdrawal or craving, focusing on alleviation of negative affective symptoms (Cohen et al. ref. 2024). As there is a continued need to develop better therapeutics for nicotine cessation, many studies have shifted to obtaining a deeper understanding of molecular mechanisms governing the negative affective state within nicotine withdrawal. Identifying key contributors to the unpleasant behaviors experienced during withdrawal can provide insights into novel treatments for these symptoms and hopefully prevent or reduce relapse.
Glucocorticoids
In mediating the stress response, the HPA axis relies on the production of CRF as well as circulating glucocorticoids (GCs), most notably cortisol in humans and corticosterone in rodents. As activation of the HPA axis is mediated by CRF, elevated CRF levels in the brain initiate a signaling cascade that ultimately leads to increased GC levels. Then, in a negative feedback loop, increased GC downregulates HPA axis signaling (Smith and Vale ref. 2006; Gjerstad et al. ref. 2018). In fact, GCs have been shown to regulate CRF expression (Tanimura and Watts ref. 1998; Imaki et al. ref. 1991). During chronic stress conditions, such as withdrawal following chronic nicotine use, circulating GC levels are decreased (Wong et al. ref. 2014; Frederick et al. ref. 1998). Conversely, increased GC administration can decrease some of the stress-related effects of sensitization to nicotine (Caggiula et al. ref. 1998). These changes to GC signaling may be implicated in negative affect during nicotine withdrawal. It was shown that nicotine withdrawal in rats produced lower corticosterone levels during restraint stress, and the authors suggested that this may implicate the precipitation of depressive-like behaviors during nicotine abstinence (Semba ref. 2004). GCs have also been shown to impact dopaminergic signaling: they can increase excitatory synaptic strength of dopaminergic synapses and promote desensitization of the reward system over time (De Jong ref. 2004; Marinelli and Piazza ref. 2002).
GCs are altered at both the extracellular and intracellular levels. Transcriptional profiling of the prefrontal cortex and NAc found that transcription factors associated with glucocorticoid receptors were significantly repressed in rats that underwent cocaine or oxycodone extended-access self-administration (Duttke et al. ref. 2022). Interestingly, single nucleotide polymorphisms (SNPs) at the FKBP5 gene have been shown to regulate severity of responses to nicotine withdrawal in humans (Jensen and Sofuoglu ref. 2016). The FKBP5 gene encodes a molecular co-chaperone of the GC receptor complex, so its activation is induced by GCs in order to terminate HPA axis signaling (Zannas et al. ref. 2016). The SNP rs3800373*C, associated with a common FKBP5 haplotype, yielded lower scores on the Minnesota Nicotine Withdrawal Scale in overnight abstinent smokers. Additional studies found similar results of this SNP in reducing alcohol withdrawal as well as a link between this SNP and heroin dependence (Huang et al. ref. 2014; Levran et al. ref. 2014). Altogether these studies show a link between the genetic regulation of glucocorticoids and the transition to substance abuse and negative affective behaviors. GC receptor antagonists are currently being investigated as treatments for alcohol use disorder, so there is potential for them to be investigated as treatments for nicotine use (McGinn et al. ref. 2021; Vendruscolo et al. ref. 2015).
5’ adenosine monophosphate kinase
Another molecular mechanism of interest has recently been shown to be the 5’ adenosine monophosphate kinase (AMPK) signaling pathway. AMPK is the key cellular regulator of energy metabolism; it assesses the ATP:AMP ratio and accordingly increases activity to generate ATP and ensure cells have the requisite amount of energy to function. AMPK is known to be necessary for synaptic activation, thus making it a crucial element of neuronal signaling (Marinangeli et al. ref. 2018). Chronic nicotine use has been shown to increase energy metabolism through both increased locomotor activity and increased thermogenesis, thus producing a negative energy balance in the system that is due to hypothalamic AMPK becoming inactivated (Martínez de Morentin et al. ref. 2012). Brynildsen et al. showed that activation of AMPK by AICAR (AMPK activator) and metformin (Type II diabetes therapeutic) abolished anxiety-like behaviors in mice during nicotine withdrawal. Furthermore, they saw that pretreating animals with systemic metformin prevented these anxiety-like behaviors while not impacting fed and fasted body weight, food intake, or glucose levels. Recently, similar results have been shown with overexpression of AMPK promoting antidepressant-like effects in mice undergoing withdrawal from methamphetamine, another psychostimulant (Hosseini et al. ref. 2022). Impairment of AMPK generates cellular stress, which over time can lead to dysregulated signaling, oxidative stress, and even cell death (Liu and Chern ref. 2015; Ronnett et al. ref. 2009). Additional studies examining other metabolic disorder therapeutics such as glucagon-like peptide 1 (GLP-1) agonists have found these to similarly play a role in nicotine intake and aversion (Tuesta et al. ref. 2017); we additionally have shown modulation of feeding-related hormone circulation following nicotine intake, suggesting a reciprocal relationship between nicotine and feeding regulation that could be explored for future therapeutic targets (Shankar et al. ref. 2024). Energy metabolism clearly plays a role in modulating negative affective behaviors, but much work needs to still be done in this field to properly assess the mechanisms surrounding these effects.
Brain-derived neurotrophic factor
Another protein of interest is brain-derived neurotrophic factor (BDNF), which is commonly known for its role in neuronal development and learning and memory. BDNF has been shown to play a role in nicotine dependence (Huang et al. ref. 2021). Nicotine withdrawal following chronic treatment has been shown to increase BDNF levels in the NAc and VTA (Kivinummi et al. ref. 2011). Increased BDNF expression has also been correlated with increased hyperalgesia during withdrawal (Shi et al. ref. 2015). Recently, Grieder et al. showed that BDNF is necessary for the transition to nicotine dependence (Grieder et al. ref. 2022). A single intra-VTA BDNF injection in drug-naïve mice produced a rewarding response to acute nicotine treatment and an aversive response to treatment 8 h after acute nicotine treatment similar to behavior seen in nicotine-dependent and nicotine-withdrawn mice, whereas control mice displayed opposite responses. The authors further showed that this behavior was due to a switch to dopamine receptor D2-mediated signaling, which they previously identified as occurring during nicotine withdrawal due to decreased tonic dopamine activity within the VTA. Rodent models of stress investigating the role of BDNF have similarly found that episodic stress increased BDNF levels in the VTA amplified dopaminergic signaling in the NAc, and these animals had increased psychostimulant intake (Miczek et al. ref. 2011). However, continuous social defeat stress led to reduced VTA BDNF levels and increased anhedonia. Interestingly, in a mouse model of nicotine exposure through water pipe tobacco smoke or injection, chronic nicotine administration prior to stress promoted resilience to stress as seen by reduced social avoidance in the chronic social defeat stress paradigm (Khalifeh et al. ref. 2020). BDNF signaling was found to confer this resilience as inhibition of the BDNF receptor tropomyosin receptor kinase B (TRKB) blocked the reduction in social avoidance after chronic stress. These studies suggest a critical role in BDNF signaling mediating the affective states of nicotine withdrawal.
Cannabinoid signaling
Cannabinoid (CB) receptors, along with their endogenous cannabinoid ligands, are a broad signaling system with involvement in many physiological and psychological functions. One of these functions is a molecular mediator of addiction. Studies have examined the role of CBs in the rewarding properties of addictive substances, particularly due to the abundance of CBs in dopaminergic VTA neurons (Mátyás et al. ref. 2008). In fact, CB receptor antagonism was shown to block the dopamine release within the NAc and the BNST (Cohen et al. ref. 2002). The cannabinoid system has specifically been shown to regulate nicotine intake; studies have shown that CB1 receptor activation increases motivation to self-administer nicotine (Gamaleddin et al. ref. 2015; Merritt et al. ref. 2008). Conversely, blockade of CB1 prevents nicotine-induced conditioned place preference and increases in locomotor activity, and CB1 antagonism was shown to reduce nicotine-self administration in a dose-dependent manner (Scherma et al. ref. 2016; Schindler et al. ref. 2016). Similar data has been shown following targeting of the endogenous cannabinoid ligands (Forget et al. ref. 2016). Stress and CB signaling have a reciprocal relationship, where exogenous CB receptor ligands increase HPA axis signaling, and antagonism of brain CRF signaling can block the increase in stress-like behaviors following CB1 receptor agonism (Puder et al. ref. 1982; De Fonseca et al. ref. 1996. As discussed above, the increase in stress signaling through CRF and GCs can heighten negative affective symptoms. Inhibition of the endocannabinoid fatty acid amide hydrolase exerts anxiolytic-like effects in mice (Griebel et al. ref. 2018). Furthermore, we recently showed that cannabidiol (CBD) treatment in rats made dependent via osmotic minipump prevented both somatic signs of withdrawal and hyperalgesia during nicotine withdrawal (Smith et al. ref. 2021). However, the mechanisms of this effect are still largely unknown as CBD is not a clear agonist of either CB1 or CB2, but may exhibit effects by binding to both receptors (Pertwee ref. 2008). Given that CBD is used clinically to alleviate stress and pain, these data further reinforce the importance of negative affect in driving addiction-like behaviors.
Social defeat stress and its effect on negative affect within nicotine addiction
Converging evidence shows that social defeat stress facilitates the transition to addiction through multiple mechanisms. Social defeat stress in rodents is a negative reinforcer that accelerates the acquisition of drug self-administration and triggers binge-like escalation of nicotine or psychostimulant intake (Ansell et al. ref. 2012; Sinha ref. 2008; Covington and Miczek ref. 2001; Covington et al. ref. 2005; Miczek et al. ref. 2004; Parise et al. ref. 2020; Zou et al. ref. 2014; Moeller et al. ref. 2001; Domingues et al. ref. 2019). Social defeat stress can facilitate these symptoms, including nicotine-induced locomotor suppression (Domingues et al. ref. 2019). Social defeat stress can also exacerbate somatic signs and anxiety-like withdrawal signs known to promote renewed consumption (Mineur et al. ref. 2016). At the circuit level, social defeat stress increases phasic and tonic dopamine release along a VTA → accumbens → prefrontal cortex → amygdala loop and elevates CRF tone within the VTA, making rodents more prone to substance-seeking behavior (Holly and Miczek ref. 2016; Morel et al. ref. 2018; Ortiz et al. ref. 2022; Adams et al. ref. 2021; Miczek et al. ref. 2008; Yap and Miczek ref. 2008; Holly et al. ref. 2016; Leonard et al. ref. 2017; Hwa et al. ref. 2016; Albrechet-Souza et al. ref. 2015). Intra-VTA CRF₁ antagonism normalizes these adaptations and limit self-administration (Han et al. ref. 2017; Burke et al. ref. 2016). Repeated social defeat stress upregulates MOR on VTA GABA interneurons, disinhibiting dopamine neurons and deepening the shift from positive to negative reinforcement (Nikulina et al. ref. 2008; Johnson and North ref. 1992; Nikulina et al. ref. 2005). Together, this dysregulation of CRF–dopamine–opioid transmissions contribute to the increased vulnerability to addition in individuals with a history of social defeat.
Conclusions and future directions
This review summarizes the mechanisms contributing to negative affect within nicotine addiction with an emphasis on the role of non-pharmacological stress, including social defeat stress (Fig. 3). The negative affective state emerges through the transition of nicotine taking as positive reinforcement to negative reinforcement. Negative affect and withdrawal are complex states mediated by a plethora of molecular, cellular, and circuit mechanisms to produce heightened aversive behaviors and emotional states. As nAChRs are ubiquitous within the brain, nicotine is pervasive in its reach, affecting many circuits that regulate negative affect within addiction. Importantly, stress and pain play a large role in these mechanisms. CRF is a key driver of the negative emotional states associated with nicotine withdrawal, and this is enhanced by changes in GC signaling that regulate CRF expression. Endogenous opioid signaling is also altered by nicotine and can contribute to the feelings of hyperalgesia associated with withdrawal. External stressors, particularly social defeat stress, also play a role in exacerbating drug-taking and subsequent negative affective states and withdrawal-like behaviors through altering signaling of dopamine, CRF, and opioids.
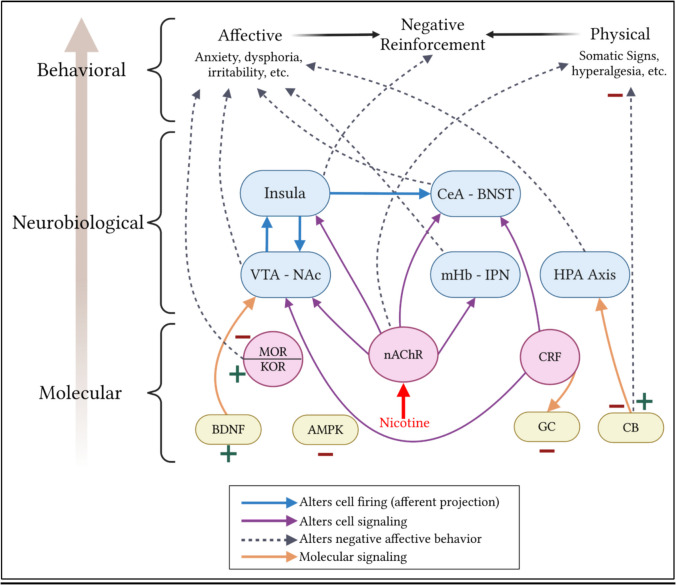
Because current therapeutic approaches for nicotine cessation are still largely ineffective (partial nAChR agonist varenicline, dopamine/norepinephrine reuptake inhibitor bupropion) (McDonough ref. 2015), understanding not only the neurobiological drivers but also the molecular mechanisms driving negative affect can provide insight into potential therapeutic avenues to mitigate withdrawal symptoms or prevent relapse. Some current therapeutics have been shown to reduce or prevent negative affective behaviors induced by nicotine dependence; these include metabolic disease treatments metformin and GLP-1 agonists, CBD, and degradation of nicotine by NicA2. While more studies are needed to fully clarify their mechanisms of action, it expands the understanding of nicotine’s pervasive reach in altering neuronal function and signaling to potentiate its addictive effects. Of the additional signaling molecules and neuropeptides covered in this review, many are effective at reducing nicotine intake or withdrawal-like behaviors at the preclinical stage but lack efficacy at the clinical stage. CRF receptor antagonists have been explored for multiple drugs of abuse (albeit not yet successfully) as a treatment option to counter the effects of substance use on the stress system (Spierling and Zorrilla ref. 2017), and targeting the opioid system poses its own risks in producing addiction-like behaviors. As we have shown the negative affective state to be important in the transition to negative reinforcement of nicotine use, current therapeutic strategies must take into account at least one of the following issues: reducing/preventing nicotine intake, mitigating withdrawal/negative affective symptoms, and reducing craving. Ultimately, the deeper our understanding of mechanisms driving negative affect, the greater the potential is for developing treatments to reduce or prevent nicotine addiction.
References
- D Adams, N Kaliss, A Missner, MM Valentine. The interplay of nicotine and social stress mediate dopaminergic neuron firing in the ventral tegmental area-nucleus accumbens pathway, contributing to stress and depressive mood disorders. Georgetown Sci Res J, 2021
- SH Ahmed, A Badiani, KA Miczek, CP Müller. Non-pharmacological factors that determine drug use and addiction. Neurosci Biobehav Rev, 2020. [PubMed]
- M Akbari, J Hasani, M Seydavi. Negative affect among daily smokers: A systematic review and meta-analysis. J Affect Disord, 2020. [PubMed]
- M Al’Absi. Hypothalamic–pituitary–adrenocortical responses to psychological stress and risk for smoking relapse. Int J Psychophysiol, 2006. [PubMed]
- L Albrechet-Souza. Corticotropin releasing factor binding protein and CRF 2 receptors in the ventral tegmental area: modulation of ethanol binge drinking in C 57 BL/6J mice. Alcohol: Clin Exp Res, 2015. [PubMed]
- EB Ansell, P Gu, K Tuit, R Sinha. Effects of cumulative stress and impulsivity on smoking status. Hum Psychopharmacol Clin Exp, 2012
- AJ Avelar, O George. How nicotine withdrawal symptoms fight each other: interpeduncular GABA neuron activity dynamically controls negative affect vs. coping behavior. Neuropsychopharmacology, 2022. [PubMed]
- TE Baker, Y Zeighami, A Dagher, CB Holroyd. Smoking decisions: altered reinforcement learning signals induced by nicotine state. Nicotine Tob Res, 2020. [PubMed]
- JL Barrington-Trimis. The e-cigarette social environment, e-cigarette use, and susceptibility to cigarette smoking. J Adolesc Health, 2016. [PubMed]
- NL Benowitz. Drug therapy. Pharmacologic aspects of cigarette smoking and nicotine addiction. N Engl J Med, 1988. [PubMed]
- NL Benowitz. Pharmacology of nicotine: addiction and therapeutics. Annu Rev Pharmacol Toxicol, 1996. [PubMed]
- GS Berlin, E Hollander. Compulsivity, impulsivity, and the DSM-5 process. CNS Spectr, 2014. [PubMed]
- G Biała, B Budzyńska, M Kruk. Naloxone precipitates nicotine abstinence syndrome and attenuates nicotine-induced antinociception in mice. Pharmacol Rep, 2005. [PubMed]
- AW Bruijnzeel, A Markou. Adaptations in cholinergic transmission in the ventral tegmental area associated with the affective signs of nicotine withdrawal in rats. Neuropharmacology, 2004. [PubMed]
- AW Bruijnzeel, G Zislis, C Wilson, MS Gold. Antagonism of CRF receptors prevents the deficit in brain reward function associated with precipitated nicotine withdrawal in rats. Neuropsychopharmacology, 2007. [PubMed]
- AW Bruijnzeel, M Prado, S Isaac. Corticotropin-releasing factor-1 receptor activation mediates nicotine withdrawal-induced deficit in brain reward function and stress-induced relapse. Biol Psychiat, 2009. [PubMed]
- DH Brunzell, AM Stafford, CI Dixon. Nicotinic receptor contributions to smoking: insights from human studies and animal models. Curr Addict Rep, 2015. [PubMed]
- AR Burke, JF DeBold, KA Miczek. CRF type 1 receptor antagonism in ventral tegmental area of adolescent rats during social defeat: prevention of escalated cocaine self-administration in adulthood and behavioral adaptations during adolescence. Psychopharmacology, 2016. [PubMed]
- K Butler, B Forget, SJ Heishman, B Le Foll. Significant association of nicotine reinforcement and cue reactivity: a translational study in humans and rats. Behav Pharmacol, 2021. [PubMed]
- AR Caggiula. The role of corticosteroids in nicotine’s physiological and behavioral effects. Psychoneuroendocrinology, 1998. [PubMed]
- Carrette LLG et al (2023) Hyperconnectivity of two separate long-range cholinergic systems contributes to the reorganization of the brain functional connectivity during nicotine withdrawal in male mice. eNeuro10:ENEURO.0019–23.2023. 10.1101/2023.03.29.534836
- M Casarrubea. Acute nicotine induces anxiety and disrupts temporal pattern organization of rat exploratory behavior in hole-board: a potential role for the lateral habenula. Front Cell Neurosci, 2015. [PubMed]
- CDCTobaccoFree (2021) Fast facts. Centers for disease control and prevention. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/fast_facts/index.htm. Accessed 15 June 2025
- J-P Changeux. Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat Rev Neurosci, 2010. [PubMed]
- JP Changeux. Brain nicotinic receptors: structure and regulation, role in learning and reinforcement. Brain Res Rev, 1998. [PubMed]
- R Chellian. Rodent models for nicotine withdrawal. J Psychopharmacol, 2021. [PubMed]
- PBS Clarke, DW Hommer, A Pert, LR Skirboll. Innervation of substantia nigra neurons by cholinergic afferents from pedunculopontine nucleus in the rat: neuroanatomical and electrophysiological evidence. Neuroscience, 1987. [PubMed]
- A Cohen, O George. Animal models of nicotine exposure: relevance to second-hand smoking, electronic cigarette use, and compulsive smoking. Front Psychiatr, 2013
- C Cohen, G Perrault, C Voltz, R Steinberg, P Soubrié. SR141716, a central cannabinoid (CB1) receptor antagonist, blocks the motivational and dopamine-releasing effects of nicotine in rats. Behav Pharmacol, 2002. [PubMed]
- A Cohen, GF Koob, O George. Robust escalation of nicotine intake with extended access to nicotine self-administration and intermittent periods of abstinence. Neuropsychopharmacology, 2012. [PubMed]
- A Cohen. Extended access to nicotine leads to a CRF1 receptor dependent increase in anxiety-like behavior and hyperalgesia in rats. Addict Biol, 2015. [PubMed]
- G Cohen, CM Bellanca, R Bernardini, JE Rose, R Polosa. Personalized and adaptive interventions for smoking cessation: Emerging trends and determinants of efficacy. iScience, 2024. [PubMed]
- Corrigall WA (1992) A rodent model for nicotine self-administration. In: Boulton AA, Baker GB, Wu PH (eds) Animal models of drug addiction. Humana Press, Totowa, NJ, pp. 315–344. 10.1385/0-89603-217-5:315
- P Cottone. CRF system recruitment mediates dark side of compulsive eating. Proc Natl Acad Sci, 2009. [DOI | PubMed]
- HE Covington, KA Miczek. Repeated social-defeat stress, cocaine or morphine. Psychopharmacology, 2001. [PubMed]
- HE Covington. Brief social defeat stress: long lasting effects on cocaine taking during a binge and Zif268 mRNA expression in the amygdala and prefrontal cortex. Neuropsychopharmacol, 2005
- Current Cigarette Smoking Among Adults — United States (2005–2012). https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6302a2.htm. Accessed 15 June 2025
- MI Damaj, W Kao, BR Martin. Characterization of spontaneous and precipitated nicotine withdrawal in the mouse. J Pharmacol Exp Ther, 2003. [PubMed]
- FR De Fonseca, P Rubio, F Menzaghi, E Merlo-Pich, J Rivier, GF Koob, M Navarro. Corticotropin-releasing factor (CRF) antagonist [D-Phe12, Nle 21, 38, C alpha MeLeu37] CRF attenuates the acute actions of the highly potent cannabinoid receptor agonist HU-210 on defensive-withdrawal behavior in rats. J Pharmacol Exp Ther, 1996. [PubMed]
- IE de Jong, ER de Kloet. Glucocorticoids and vulnerability to psychostimulant drugs: toward substrate and mechanism. Ann N Y Acad Sci, 2004. [PubMed]
- Diagnostic and Statistical Manual of Mental Disorders (n.d.) DSM library. 10.1176/appi.books.9780890425787
- LP Domingues, BDB Antonio, MGMD Oliveira, IMHD Quadros. Consequence of two protocols of social defeat stress on nicotine-induced psychomotor effects in mice. BioMed Res Int, 2019. [PubMed]
- EC Donny, AR Caggiula, S Knopf, C Brown. Nicotine self-administration in rats. Psychopharmacology, 1995. [PubMed]
- EC Donny. Acquisition of nicotine self-administration in rats: the effects of dose, feeding schedule, and drug contingency. Psychopharmacology, 1998. [PubMed]
- SH Duttke. Glucocorticoid receptor-regulated enhancers play a central role in the gene regulatory networks underlying drug addiction. Front Neurosci, 2022. [PubMed]
- JO Ebbert. Effect of varenicline on smoking cessation through smoking reduction: a randomized clinical trial. JAMA, 2015. [PubMed]
- KS Elayouby. α3* Nicotinic acetylcholine receptors in the habenula-interpeduncular nucleus circuit regulate nicotine intake. J Neurosci, 2021. [PubMed]
- MP Epping-Jordan, SS Watkins, GF Koob, A Markou. Dramatic decreases in brain reward function during nicotine withdrawal. Nature, 1998. [PubMed]
- KE Farsalinos, R Polosa. Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarette substitutes: a systematic review. Ther Adv Drug Saf, 2014. [PubMed]
- P Faulkner. Neural basis of smoking-induced relief of craving and negative affect: contribution of nicotine. Addict Biol, 2019. [PubMed]
- P Faure, S Tolu, S Valverde, J Naudé. Role of nicotinic acetylcholine receptors in regulating dopamine neuron activity. Neuroscience, 2014. [PubMed]
- M Field, M Santarcangelo, H Sumnall, A Goudie, J Cole. Delay discounting and the behavioural economics of cigarette purchases in smokers: the effects of nicotine deprivation. Psychopharmacology, 2006. [PubMed]
- B Forget, M Guranda, I Gamaleddin, SR Goldberg, B Le Foll. Attenuation of cue-induced reinstatement of nicotine seeking by URB597 through cannabinoid CB1 receptor in rats. Psychopharmacology, 2016. [PubMed]
- CD Fowler, PJ Kenny. Nicotine aversion: Neurobiological mechanisms and relevance to tobacco dependence vulnerability. Neuropharmacology, 2014. [DOI | PubMed]
- CD Fowler, Q Lu, PM Johnson, MJ Marks, PJ Kenny. Habenular α5* nicotinic receptor signaling controls nicotine intake. Nature, 2011. [PubMed]
- S Frahm. Aversion to nicotine is regulated by the balanced activity of β4 and α5 nicotinic receptor subunits in the medial habenula. Neuron, 2011. [PubMed]
- SL Frederick. Cortisol and response to dexamethasone as predictors of withdrawal distress and abstinence success in smokers. Biol Psychiat, 1998. [PubMed]
- IH Gamaleddin. Role of the endogenous cannabinoid system in nicotine addiction: novel insights. Front Psychiatry, 2015. [PubMed]
- George O, Koob GF (2017) Chapter 1 – Overview of nicotine withdrawal and negative reinforcement (Preclinical). In: Hall FS, Young JW, Der-Avakian A (eds) Negative affective states and cognitive impairments in nicotine dependence. Academic Press, San Diego, pp. 1–20. 10.1016/B978-0-12-802574-1.00001-6
- O George. CRF–CRF1 system activation mediates withdrawal-induced increases in nicotine self-administration in nicotine-dependent rats. PNAS, 2007. [PubMed]
- O George, SH Ahmed, NW Gilpin. Are we compulsively chasing rainbows?. Neuropsychopharmacology, 2022. [PubMed]
- JK Gjerstad, SL Lightman, F Spiga. Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress, 2018. [PubMed]
- AE Goudriaan, MB De Ruiter, W Van Den Brink, J Oosterlaan, DJ Veltman. Brain activation patterns associated with cue reactivity and craving in abstinent problem gamblers, heavy smokers and healthy controls: an fMRI study. Addict Biol, 2010. [PubMed]
- SD Grabus. Nicotine physical dependence and tolerance in the mouse following chronic oral administration. Psychopharmacology, 2005. [PubMed]
- G Griebel. The selective reversible FAAH inhibitor, SSR411298, restores the development of maladaptive behaviors to acute and chronic stress in rodents. Sci Rep, 2018. [PubMed]
- TE Grieder. VTA CRF neurons mediate the aversive effects of nicotine withdrawal and promote intake escalation. Nat Neurosci, 2014. [PubMed]
- TE Grieder. Administration of BDNF in the ventral tegmental area produces a switch from a nicotine-non-dependent D1R-mediated motivational state to a nicotine-dependent-like D2R-mediated motivational state. Eur J Neurosci, 2022. [PubMed]
- M Hadjiconstantinou, NH Neff. Nicotine and endogenous opioids: neurochemical and pharmacological evidence. Neuropharmacology, 2011. [PubMed]
- Hajek P, Stead LF (2001) Aversive smoking for smoking cessation. Coch Database Syst Rev 2001(3):CD000546. 10.1002/14651858.CD000546.pub2
- KR Hamilton, SS Berger, ME Perry, NE Grunberg. Behavioral effects of nicotine withdrawal in adult male and female rats. Pharmacol Biochem Behav, 2009. [PubMed]
- X Han, JF DeBold, KA Miczek. Prevention and reversal of social stress-escalated cocaine self-administration in mice by intra-VTA CRFR1 antagonism. Psychopharmacology, 2017. [PubMed]
- SC Heinrichs. Modulation of social learning in rats by brain corticotropin-releasing factor. Brain Res, 2003. [PubMed]
- SC Heinrichs, GF Koob. Corticotropin-releasing factor in brain: a role in activation, arousal, and affect regulation. J Pharmacol Exp Ther, 2004. [PubMed]
- BJ Henderson, HA Lester. Inside-out neuropharmacology of nicotinic drugs. Neuropharmacology, 2015. [PubMed]
- EN Holly, KA Miczek. Ventral tegmental area dopamine revisited: effects of acute and repeated stress. Psychopharmacology, 2016. [PubMed]
- EN Holly, JF DeBold, KA Miczek. Increased mesocorticolimbic dopamine during acute and repeated social defeat stress: modulation by corticotropin releasing factor receptors in the ventral tegmental area. Psychopharmacology, 2015. [PubMed]
- EN Holly. Episodic social stress-escalated cocaine self-administration: role of phasic and tonic corticotropin releasing factor in the anterior and posterior ventral tegmental area. J Neurosci, 2016. [PubMed]
- MJ Hosseini, A Arabiyan, S Mobassem, H Ghavimi. Metformin attenuates depressive-like behaviour of methamphetamine withdrawal in mice: A mechanistic approach. World J Biol Psychiatr, 2022
- M Hua, P Talbot. Potential health effects of electronic cigarettes: a systematic review of case reports. Prev Med Rep, 2016. [PubMed]
- Y-Y Huang, ER Kandel, A Levine. Chronic nicotine exposure induces a long-lasting and pathway-specific facilitation of LTP in the amygdala. Learn Mem, 2008. [PubMed]
- M-C Huang. FKBP5 moderates alcohol withdrawal severity: human genetic association and functional validation in knockout mice. Neuropsychopharmacol, 2014
- Z Huang. BDNF and nicotine dependence: associations and potential mechanisms. Rev Neurosci, 2021
- JR Hughes, ST Higgins, WK Bickel. Nicotine withdrawal versus other drug withdrawal syndromes: similarities and dissimilarities. Addiction, 1994. [PubMed]
- LS Hwa, EN Holly, JF DeBold, KA Miczek. Social stress-escalated intermittent alcohol drinking: modulation by CRF-R1 in the ventral tegmental area and accumbal dopamine in mice. Psychopharmacology, 2016. [PubMed]
- C Ibrahim. The insula: a brain stimulation target for the treatment of addiction. Front Pharmacol, 2019. [PubMed]
- T Imaki, JL Nahan, C Rivier, PE Sawchenko, W Vale. Differential regulation of corticotropin-releasing factor mRNA in rat brain regions by glucocorticoids and stress. J Neurosci, 1991. [PubMed]
- PF Isaac, MJ Rand. Cigarette smoking and plasma levels of nicotine. Nature, 1972. [PubMed]
- K Ishii, JK Wong, K Sumikawa. A comparison of α 2 nicotinic acetylcholine receptor subunit mRNA expression in the central nervous system of rats and mice. J Comp Neurol, 2005. [PubMed]
- KJ Jackson, BR Martin, JP Changeux, MI Damaj. Differential role of nicotinic acetylcholine receptor subunits in physical and affective nicotine withdrawal signs. J Pharmacol Exp Ther, 2008. [PubMed]
- KJ Jackson, DH Kota, BR Martin, MI Damaj. The role of various nicotinic receptor subunits and factors influencing nicotine conditioned place aversion. Neuropharmacology, 2009. [DOI | PubMed]
- KJ Jackson, JM McIntosh, DH Brunzell, SS Sanjakdar, MI Damaj. The role of α6-containing nicotinic acetylcholine receptors in nicotine reward and withdrawal. J Pharmacol Exp Ther, 2009. [PubMed]
- KJ Jackson, SS Sanjakdar, PP Muldoon, JM McIntosh, MI Damaj. The α3β4* nicotinic acetylcholine receptor subtype mediates nicotine reward and physical nicotine withdrawal signs independently of the α5 subunit in the mouse. Neuropharmacology, 2013. [PubMed]
- A Jackson, RL Papke, MI Damaj. Pharmacological modulation of the α7 nicotinic acetylcholine receptor in a mouse model of mecamylamine-precipitated nicotine withdrawal. Psychopharmacology, 2018. [PubMed]
- KP Jensen, M Sofuoglu. Stress response genes and the severity of nicotine withdrawal. Pharmacogenomics, 2016. [PubMed]
- SW Johnson, RA North. Opioids excite dopamine neurons by hyperpolarization of local interneurons. J Neurosci, 1992. [PubMed]
- M Kallupi, G de Guglielmo, E Larrosa, O George. Exposure to nicotine vapor in male adolescent rats produces a withdrawal-like state and facilitates nicotine self-administration during adulthood. Eur Neuropsychopharmacol, 2019. [PubMed]
- Substance Abuse and Mental Health Services Administration (2023) Key substance use and mental health indicators in the United States: Results from the 2022 National Survey on Drug Use and Health (HHS Publication No. PEP23-07-01-006, NSDUH Series H-58). Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. https://www.samhsa.gov/data/report/2022-nsduh-annual-national-report
- M Khalifeh. Nicotine induces resilience to chronic social defeat stress in a mouse model of water pipe tobacco exposure by activating BDNF signaling. Behav Brain Res, 2020. [PubMed]
- B Kim, H-I Im. Behavioral characterization of early nicotine withdrawal in the mouse: a potential model of acute dependence. Behav Brain Funct, 2024. [PubMed]
- DE Kirsch, ETC Lippard. Early life stress and substance use disorders: The critical role of adolescent substance use. Pharmacol Biochem Behav, 2022. [PubMed]
- T Kivinummi, K Kaste, T Rantamäki, E Castrén, L Ahtee. Alterations in BDNF and phospho-CREB levels following chronic oral nicotine treatment and its withdrawal in dopaminergic brain areas of mice. Neurosci Lett, 2011. [PubMed]
- MN Koffarnus, G Winger. Individual differences in the reinforcing and punishing effects of nicotine in rhesus monkeys. Psychopharmacology, 2015. [PubMed]
- GF Koob. Neurobiology of addiction. FOC, 2011
- GF Koob, M Le Moal. Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology, 2001. [PubMed]
- GF Koob, M Le Moal. Addiction and the brain antireward system. Annu Rev Psychol, 2008. [PubMed]
- GF Koob, J Schulkin. Addiction and stress: an allostatic view. Neurosci Biobehav Rev, 2019. [PubMed]
- GF Koob, ND Volkow. Neurocircuitry of Addiction. Neuropsychopharmacology, 2010. [PubMed]
- GF Koob. Addiction as a stress surfeit disorder. Neuropharmacology, 2014. [DOI | PubMed]
- B Le Foll. Tobacco and nicotine use. Nat Rev Dis Primers, 2022. [PubMed]
- MZ Leonard, JF DeBold, KA Miczek. Escalated cocaine “binges” in rats: enduring effects of social defeat stress or intra-VTA CRF. Psychopharmacology, 2017. [PubMed]
- C Lerman. Large-scale brain network coupling predicts acute nicotine abstinence effects on craving and cognitive function. JAMA Psychiat, 2014
- O Levran. Stress-related genes and heroin addiction: a role for a functional FKBP5 haplotype. Psychoneuroendocrinology, 2014. [PubMed]
- Y-J Liu, Y Chern. AMPK-mediated regulation of neuronal metabolism and function in brain diseases. J Neurogenet, 2015. [PubMed]
- S Lotfipour. Targeted deletion of the mouse α2 nicotinic acetylcholine receptor subunit gene (Chrna2) potentiates nicotine-modulated behaviors. J Neurosci, 2013. [PubMed]
- Malin DH, Goyarzu P (2009) Rodent models of nicotine withdrawal syndrome. In: Henningfield JE, London ED, Pogun S (eds) Nicotine psychopharmacology. Springer, Berlin, Heidelberg, pp. 401–434. 10.1007/978-3-540-69248-5_14
- DH Malin, JR Lake, VA Carter, JS Cunningham, OB Wilson. Naloxone precipitates nicotine abstinence syndrome in the rat. Psychopharmacology, 1993. [PubMed]
- C Marinangeli. AMP-activated protein kinase is essential for the maintenance of energy levels during synaptic activation. iScience, 2018. [PubMed]
- M Marinelli, PV Piazza. Interaction between glucocorticoid hormones, stress and psychostimulant drugs*. Eur J Neurosci, 2002. [PubMed]
- A Markou. Neurobiology of nicotine dependence. Philos Trans R Soc Lond B Biol Sci, 2008. [PubMed]
- PB Martínez de Morentin. Nicotine induces negative energy balance through hypothalamic amp-activated protein kinase. Diabetes, 2012. [PubMed]
- R Martin-Fardon, EP Zorrilla, R Ciccocioppo, F Weiss. Role of innate and drug-induced dysregulation of brain stress and arousal systems in addiction: Focus on corticotropin-releasing factor, nociceptin/orphanin FQ, and orexin/hypocretin. Brain Res, 2010. [PubMed]
- U Maskos. Role of endogenous acetylcholine in the control of the dopaminergic system via nicotinic receptors. J Neurochem, 2010. [PubMed]
- M Matsumoto, O Hikosaka. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature, 2007. [PubMed]
- SG Matta, Y Fu, JD Valentine, BM Sharp. Response of the hypothalamo-pituitary-adrenal axis to nicotine. Psychoneuroendocrinology, 1998. [PubMed]
- F Mátyás. Identification of the sites of 2-arachidonoylglycerol synthesis and action imply retrograde endocannabinoid signaling at both GABAergic and glutamatergic synapses in the ventral tegmental area. Neuropharmacology, 2008. [PubMed]
- MJ McCarthy, H Zhang, NH Neff, M Hadjiconstantinou. Nicotine withdrawal and κ-opioid receptors. Psychopharmacology, 2010. [PubMed]
- M McDonough. Update on medicines for smoking cessation. Aust Prescr, 2015. [PubMed]
- MA McGinn. Glucocorticoid receptor modulators decrease alcohol self-administration in male rats. Neuropharmacology, 2021. [PubMed]
- SA McKee. Stress decreases the ability to resist smoking and potentiates smoking intensity and reward. J Psychopharmacol, 2011. [PubMed]
- I McLaughlin, JA Dani, M De Biasi. Nicotine Withdrawal. Curr Top Behav Neurosci, 2015. [PubMed]
- LL Merritt, BR Martin, C Walters, AH Lichtman, MI Damaj. The endogenous cannabinoid system modulates nicotine reward and dependence. J Pharmacol Exp Ther, 2008. [PubMed]
- KA Miczek, HE Covington, EM Nikulina, RP Hammer. Aggression and defeat: persistent effects on cocaine self-administration and gene expression in peptidergic and aminergic mesocorticolimbic circuits. Neurosci Biobehav Rev, 2004. [PubMed]
- KA Miczek, JJ Yap, HE Covington. Social stress, therapeutics and drug abuse: preclinical models of escalated and depressed intake. Pharmacol Ther, 2008. [PubMed]
- KA Miczek, EM Nikulina, A Shimamoto, HE Covington. Escalated or suppressed cocaine reward, tegmental bdnf, and accumbal dopamine caused by episodic versus continuous social stress in rats. J Neurosci, 2011. [PubMed]
- YS Mineur. Multiple nicotinic acetylcholine receptor subtypes in the mouse amygdala regulate affective behaviors and response to social stress. Neuropsychopharmacol, 2016
- SH Mitchell. Effects of short-term nicotine deprivation on decision-making: delay, uncertainty and effort discounting. Nicotine Tob Res, 2004. [PubMed]
- FG Moeller, ES Barratt, DM Dougherty, JM Schmitz, AC Swann. Psychiatric aspects of impulsivity. AJP, 2001
- C Morel. Nicotinic receptors mediate stress-nicotine detrimental interplay via dopamine cells’ activity. Mol Psychiatry, 2018. [PubMed]
- NH Naqvi, A Bechara. The hidden island of addiction: the insula. Trends Neurosci, 2009. [PubMed]
- NH Naqvi, D Rudrauf, H Damasio, A Bechara. Damage to the insula disrupts addiction to cigarette smoking. Science, 2007. [PubMed]
- PA Newhouse. Neuroendocrine, physiologic, and behavioral responses following intravenous nicotine in nonsmoking healthy volunteers and in patients with alzheimer’s disease. Psychoneuroendocrinology, 1990. [PubMed]
- Krasnegor NA (ed) (1979) The behavioral aspects of smoking: NIDA research monograph 26. US Department of Health, Education, and Welfare, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute on Drug Abuse, Division of Research
- EM Nikulina, KA Miczek, RP Hammer. Prolonged effects of repeated social defeat stress on mRNA expression and function of μ-opioid receptors in the ventral tegmental area of rats. Neuropsychopharmacol, 2005
- EM Nikulina, I Arrillaga-Romany, KA Miczek, RP Hammer. Long-lasting alteration in mesocorticolimbic structures after repeated social defeat stress in rats: time course of mu-opioid receptor mRNA and FosB/DeltaFosB immunoreactivity. Eur J Neurosci, 2008. [PubMed]
- R Nil, PP Woodson, K Bättig. Effects of smoking deprivation on smoking behavior and heart rate response in high and low CO absorbing smokers. Psychopharmacology, 1987. [PubMed]
- H Norman, MS D’Souza. Endogenous opioid system: a promising target for future smoking cessation medications. Psychopharmacology, 2017. [PubMed]
- LE O’dell, AW Bruijnzeel, S Ghozland, A Markou, GF Koob. Nicotine withdrawal in adolescent and adult rats. Ann N Y Acad Sci, 2004. [PubMed]
- LE O’Dell. Extended access to nicotine self-administration leads to dependence: circadian measures, withdrawal measures, and extinction behavior in rats. J Pharmacol Exp Ther, 2007. [PubMed]
- S Okada, T Shimizu, K Yokotani. Extrahypothalamic corticotropin-releasing hormone mediates (−)-nicotine-induced elevation of plasma corticosterone in rats. Eur J Pharmacol, 2003. [PubMed]
- V Ortiz, R Costa Campos, H Fofo, SP Fernandez, J Barik. Nicotinic receptors promote susceptibility to social stress in female mice linked with neuroadaptations within VTA dopamine neurons. Neuropsychopharmacology, 2022. [PubMed]
- RD Pang, R Khoddam, CR Guillot, AM Leventhal. Depression and anxiety symptoms moderate the relation between negative reinforcement smoking outcome expectancies and nicotine dependence. J Stud Alcohol Drugs, 2014. [PubMed]
- M Paolini, M De Biasi. Mechanistic insights into nicotine withdrawal. Biochem Pharmacol, 2011. [PubMed]
- LF Parise. Nicotine treatment buffers negative behavioral consequences induced by exposure to physical and emotional stress in adolescent male mice. Psychopharmacology, 2020. [PubMed]
- KA Perkins, C Jetton, J Keenan. Common factors across acute subjective effects of nicotine. Nicotine Tob Res, 2003. [PubMed]
- RG Pertwee. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin. Br J Pharmacol, 2008. [PubMed]
- MR Picciotto, PJ Kenny. Mechanisms of nicotine addiction. Cold Spring Harb Perspect Med, 2021. [PubMed]
- MR Picciotto. Acetylcholine receptors containing the β2 subunit are involved in the reinforcing properties of nicotine. Nature, 1998. [PubMed]
- MR Picciotto, MJ Higley, YS Mineur. Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron, 2012. [PubMed]
- M Pierucci. Nicotine modulation of the lateral habenula/ventral tegmental area circuit dynamics: an electrophysiological study in rats. Neuropharmacology, 2022. [PubMed]
- CS Pomerleau, OF Pomerleau. Euphoriant effects of nicotine in smokers. Psychopharmacology, 1992. [DOI | PubMed]
- CS Pomerleau, OF Pomerleau. Euphoriant effects of nicotine in smokers. Psychopharmacology, 1992. [PubMed]
- L Ponzoni. Different physiological and behavioural effects of e-cigarette vapour and cigarette smoke in mice. Eur Neuropsychopharmacol, 2015. [PubMed]
- GS Portugal, JW Kenney, TJ Gould. β2 subunit containing acetylcholine receptors mediate nicotine withdrawal deficits in the acquisition of contextual fear conditioning. Neurobiol Learn Mem, 2008. [PubMed]
- M Puder. Corticotrophin and corticosterone secretion following Δ1-tetrahydrocannabinol, in intact and in hypothalamic deafferentated male rats. Exp Brain Res, 1982. [PubMed]
- X Qi. Sustained AAV-mediated overexpression of CRF in the central amygdala diminishes the depressive-like state associated with nicotine withdrawal. Transl Psychiatry, 2014. [PubMed]
- X Qi. Overexpression of CRF in the BNST diminishes dysphoria but not anxiety-like behavior in nicotine withdrawing rats. Eur Neuropsychopharmacol, 2016. [PubMed]
- MF Regner. The insula in nicotine use disorder: Functional neuroimaging and implications for neuromodulation. Neurosci Biobehav Rev, 2019. [PubMed]
- GV Ronnett, S Ramamurthy, AM Kleman, LE Landree, S Aja. AMPK in the brain: its roles in energy balance and neuroprotection. J Neurochem, 2009. [PubMed]
- J-Y Rotge. Bidirectional regulation over the development and expression of loss of control over cocaine intake by the anterior insula. Psychopharmacology, 2017. [PubMed]
- MA Russell. Tobacco dependence: Is nicotine rewarding or aversive?. NIDA Res Monogr, 1979
- JM Rusted, A Mackee, R Williams, P Willner. Deprivation state but not nicotine content of the cigarette affects responding by smokers on a progressive ratio task. Psychopharmacology, 1998. [PubMed]
- R Salas, A Main, D Gangitano, M De Biasi. Decreased withdrawal symptoms but normal tolerance to nicotine in mice null for the α7 nicotinic acetylcholine receptor subunit. Neuropharmacology, 2007. [PubMed]
- R Salas, R Sturm, J Boulter, MD Biasi. Nicotinic receptors in the habenulo-interpeduncular system are necessary for nicotine withdrawal in mice. J Neurosci, 2009. [PubMed]
- MM Sánchez, LJ Young, PM Plotsky, TR Insel. Autoradiographic and in situ hybridization localization of corticotropin-releasing factor 1 and 2 receptors in nonhuman primate brain. Journal of Comparative Neurology, 1999. [PubMed]
- CE Sartor. Initial response to cigarettes predicts rate of progression to regular smoking: findings from an offspring-of-twins design. Addict Behav, 2010. [PubMed]
- M Scherma. Interactions between the endocannabinoid and nicotinic cholinergic systems: preclinical evidence and therapeutic perspectives. Psychopharmacology, 2016. [PubMed]
- CW Schindler. Blockade of nicotine and cannabinoid reinforcement and relapse by a cannabinoid CB1-receptor neutral antagonist AM4113 and inverse agonist rimonabant in squirrel monkeys. Neuropsychopharmacol, 2016
- J Semba. Nicotine withdrawal induces subsensitivity of hypothalamic–pituitary–adrenal axis to stress in rats: implications for precipitation of depression during smoking cessation. Psychoneuroendocrinology, 2004. [PubMed]
- X Shang. Effectiveness and safety of varenicline for smoking cessation: an overview and meta-analysis. J Addict Med, 2023. [PubMed]
- K Shankar, J Ramborger, S Bonnet-Zahedi, LLG Carrette, O George. Acute nicotine intake increases feeding behavior through decreasing glucagon signaling in dependent male and female rats. Horm Behav, 2024. [PubMed]
- W Shi. BDNF/TRK/KCC2 pathway in nicotine withdrawal-induced hyperalgesia. Transl Neurosci, 2015. [PubMed]
- S Simpson, K Shankar, A Kimbrough, O George. Role of corticotropin-releasing factor in alcohol and nicotine addiction. Brain Res, 2020. [PubMed]
- R Sinha. Chronic stress, drug use, and vulnerability to addiction. Ann N Y Acad Sci, 2008. [PubMed]
- MA Ślimak. Habenular expression of rare missense variants of the β4 nicotinic receptor subunit alters nicotine consumption. Front Human Neurosci, 2014
- SM Smith, WW Vale. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci, 2006. [PubMed]
- LC Smith. Validation of a nicotine vapor self-administration model in rats with relevance to electronic cigarette use. Neuropsychopharmacology, 2020. [PubMed]
- LC Smith. Cannabidiol reduces withdrawal symptoms in nicotine-dependent rats. Psychopharmacology, 2021. [PubMed]
- SR Spierling, EP Zorrilla. Don’t stress about CRF: Assessing the translational failures of CRF1 antagonists. Psychopharmacology, 2017. [PubMed]
- A Steptoe, M Ussher. Smoking, cortisol and nicotine. Int J Psychophysiol, 2006. [PubMed]
- AK Stoker, S Semenova, A Markou. Affective and somatic aspects of spontaneous and precipitated nicotine withdrawal in C57BL/6J and BALB/cByJ mice. Neuropharmacology, 2008. [PubMed]
- IP Stolerman, MJ Jarvis. The scientific case that nicotine is addictive. Psychopharmacology, 1995. [PubMed]
- SM Tanimura, AG Watts. Corticosterone can facilitate as well as inhibit corticotropin-releasing hormone gene expression in the rat hypothalamic paraventricular nucleus*. Endocrinology, 1998. [PubMed]
- HA Tejeda, LA Natividad, JE Orfila, OV Torres, LE O’Dell. Dysregulation of kappa-opioid receptor systems by chronic nicotine modulate the nicotine withdrawal syndrome in an age-dependent manner. Psychopharmacology, 2012. [PubMed]
- JW Tidey, KA Miczek. Social defeat stress selectively alters mesocorticolimbic dopamine release: an in vivo microdialysis study. Brain Res, 1996. [PubMed]
- ST Tiffany, EM Martin, TB Baker. Treatments for cigarette smoking: an evaluation of the contributions of aversion and counseling procedures. Behav Res Ther, 1986. [PubMed]
- OV Torres, LE O’Dell. Stress is a principal factor that promotes tobacco use in females. Prog Neuropsychopharmacol Biol Psychiatry, 2016. [PubMed]
- OV Torres, LG Gentil, LA Natividad, LM Carcoba, LE O’Dell. Behavioral, biochemical, and molecular indices of stress are enhanced in female versus male rats experiencing nicotine withdrawal. Front Psychiatr, 2013
- LM Tuesta. GLP-1 acts on habenular avoidance circuits to control nicotine intake. Nat Neurosci, 2017. [PubMed]
- R Urbán. Smoking outcome expectancies mediate the association between sensation seeking, peer smoking, and smoking among young adolescents. Nicotine Tob Res, 2010. [PubMed]
- LF Vendruscolo. Glucocorticoid receptor antagonism decreases alcohol seeking in alcohol-dependent individuals. J Clin Investig, 2015. [PubMed]
- C Vidal, J-P Changeux. Neuronal nicotinic acetylcholine receptors in the brain. Physiology, 1996
- JM Vink, G Willemsen, DI Boomsma. The association of current smoking behavior with the smoking behavior of parents, siblings, friends and spouses. Addiction, 2003. [PubMed]
- ND Volkow, RA Wise, R Baler. The dopamine motive system: implications for drug and food addiction. Nat Rev Neurosci, 2017. [PubMed]
- T Wang. Social and anxiety-like behaviors contribute to nicotine self-administration in adolescent outbred rats. Sci Rep, 2018. [PubMed]
- MC Wardle. Behavioral therapies targeting reward mechanisms in substance use disorders. Pharmacol Biochem Behav, 2024. [PubMed]
- SS Watkins, GF Koob, A Markou. Neural mechanisms underlying nicotine addiction: acute positive reinforcement and withdrawal. Nicotine Tob Res, 2000. [PubMed]
- S Wee, GF Koob. The role of the dynorphin–κ opioid system in the reinforcing effects of drugs of abuse. Psychopharmacology, 2010. [PubMed]
- MB Wigestrand. Decreased α4β2 nicotinic receptor number in the absence of mRNA changes suggests post-transcriptional regulation in the spontaneously hypertensive rat model of ADHD: α4β2 nicotinic receptor disturbances in SHRs. J Neurochem, 2011. [PubMed]
- L Wills. Neurobiological mechanisms of nicotine reward and aversion. Pharmacol Rev, 2022. [PubMed]
- JA Wong, WB Pickworth, AJ Waters, M Al’bsi, AM Leventhal. Cortisol levels decrease after acute tobacco abstinence in regular smokers. Human Psychopharmacol: Clin Exp, 2014
- C Xiao, C-Y Zhou, J-H Jiang, C Yin. Neural circuits and nicotinic acetylcholine receptors mediate the cholinergic regulation of midbrain dopaminergic neurons and nicotine dependence. Acta Pharmacol Sin, 2020. [PubMed]
- B Yang, J Liu, L Popova. feeling hopeful motivates change: emotional responses to messages communicating comparative risk of electronic cigarettes and combusted cigarettes. Health Educ Behav, 2019. [PubMed]
- JJ Yap, KA Miczek. Stress and rodent models of drug addiction: role of VTA-Accumbens-PFC-Amygdala circuit. Drug Discov Today Dis Model, 2008
- AS Zannas, T Wiechmann, NC Gassen, EB Binder. Gene–stress–epigenetic regulation of fkbp5: clinical and translational implications. Neuropsychopharmacol, 2016
- R Zhao-Shea, L Liu, X Pang, PD Gardner, AR Tapper. Activation of GABAergic neurons in the interpeduncular nucleus triggers physical nicotine withdrawal symptoms. Curr Biol, 2013. [DOI | PubMed]
- R Zhao-Shea. Increased CRF signalling in a ventral tegmental area-interpeduncular nucleus-medial habenula circuit induces anxiety during nicotine withdrawal. Nat Commun, 2015. [PubMed]
- F-M Zhou, Y Liang, JA Dani. Endogenous nicotinic cholinergic activity regulates dopamine release in the striatum. Nat Neurosci, 2001. [PubMed]
- S Zou, D Funk, MJ Shram, AD Lê. Effects of stressors on the reinforcing efficacy of nicotine in adolescent and adult rats. Psychopharmacology, 2014. [PubMed]
- W Zuo. Nicotine regulates activity of lateral habenula neurons via presynaptic and postsynaptic mechanisms. Sci Rep, 2016. [PubMed]
