South African Propolis: Anti‐Helicobacter pylori Activity, Chemistry, and Toxicity
Abstract
Propolis, a resin‐like substance produced by bees, has previously shown antimicrobial activity against the ulcer‐causing gut pathogen Helicobacter pylori. South African propolis, however, was yet to be investigated. This study aimed to investigate a comprehensive range of South African propolis for its antimicrobial activity against H. pylori and to investigate toxicity. A total of 51 samples were collected from around South Africa and comparatively analysed with three Brazilian samples. The antimicrobial broth microdilution assay was used to determine the minimum inhibitory concentration (MIC) of ethanolic propolis extracts against three clinical H. pylori strains. A total of 27 South African propolis extracts presented antimicrobial activity better than that of the Brazilian samples (MIC ≤ 0.51 mg/mL). Samples with the best anti‐H. pylori activity were selected for chemical analysis using ultra‐performance liquid chromatography–mass spectrometry. The compounds pinocembrin, 3‐O‐acetylpinobanksin, and pinobanksin were found to be the most abundant. All propolis extracts investigated in this study were considered non‐toxic (mortality < 50%) when investigated using the brine shrimp lethality assay. This study demonstrates the in vitro potential of utilizing propolis for treating H. pylori infections and highlights the possible compounds responsible for the activity observed.
Article type: Research Article
Keywords: biological activity, liquid chromatography, propolis, toxicity
Affiliations: Department of Pharmacy and Pharmacology, Faculty of Health Sciences University of the Witwatersrand Johannesburg South Africa; Department of Pharmaceutical Sciences, Faculty of Sciences Tshwane University of Technology Pretoria South Africa; SAMRC Herbal Drugs Research Unit, Department of Pharmaceutical Sciences Tshwane University of Technology Pretoria South Africa
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202403200 | PubMed: 40098592 | PMC: PMC12351434
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (905 KB)
Introduction
Helicobacter pylori colonizes the human gastric mucosa and has been found to infect over 50% of the global population, with prevalence in South Africa ranging between 50% and 84% [ref. 1]. It is implicated in a variety of gastric ailments, most notably causing gastric ulcers [ref. 2]. Treatment options for H. pylori infections currently include proton pump inhibitors, bismuth and multiple antibiotics, such as amoxicillin and clarithromycin [ref. 3]. As H. pylori rapidly gains resistance to antibiotics, treatment options are increasingly losing their efficacy, with increasing resistance rates to clarithromycin reported in South Africa [ref. 4]. Thus, natural products serve as an attractive alternative treatment for H. pylori infections and related ailments.
Propolis, a resin‐like substance, is produced by the Apis mellifera bee and used to repair and reinforce the hive. It also acts as an antiseptic, preventing infections within the hive [ref. 5]. The medicinal use of propolis has been recorded for thousands of years and has been used historically in embalming and wound healing [ref. 6, ref. 7]. Propolis has since been shown to have numerous medicinal properties, including anti‐ulcer, anti‐inflammatory, anti‐tumour and antimicrobial activity [ref. 7, ref. 8, ref. 9, ref. 10]. Propolis has been noted as a valuable anti‐H. pylori agent, with studies investigating anti‐ulcer activity, enzyme inhibition and anti‐oxidant activity, as well as antimicrobial activity [ref. 11, ref. 12].
Several studies have investigated the anti‐ulcer and antimicrobial activity against H. pylori of propolis that were collected from different regions around the world, including Spain, Korea and Chile [ref. 11, ref. 12]. Brazilian propolis, commercially highly regarded, is noted for a wide range of medicinal and health benefits and was also shown to have anti‐H. pylori activity [ref. 12, ref. 13]. African propolis, however, has been poorly explored for its anti‐H. pylori activity. Only two studies were carried out in the past 10 years, both investigating Nigerian propolis, with the results indicating good antimicrobial activity [ref. 14, ref. 15, ref. 16]. Southern African propolis, however, is yet to be explored in this context. South African propolis was studied for its antimicrobial activity against a range of pathogens, with various propolis samples from South Africa and Brazil shown to have substantial antimicrobial activity against Staphylococcus aureus [ref. 7]. A South African propolis sample, along with three associated propolis compounds, pinocembrin, galangin and chrysin, were investigated against a variety of bacterial and fungal pathogens, where combinations of the compounds showed the best antimicrobial activity [ref. 17]. The anti‐H. pylori potential of South African propolis, however, was yet to be investigated.
The pharmacological activity of propolis is influenced by the chemical composition, which varies depending on the geographic location, season of collection, bee species and plant sources [ref. 11]. Over 300 compounds have been identified from propolis samples, including polyphenols, terpenoids, steroids, and amino acids. Flavonoids, such as pinocembrin, acacetin, and chrysin, were found to be the most abundant [ref. 10]. Antimicrobially active South African propolis was reported to be primarily comprised of pinocembrin, galangin, and chrysin. The chemical profiles noted for South African propolis were similar to that of temperate region propolis, and were distinct from Brazilian propolis, of the tropical regions [ref. 7].
While natural products are often assumed to be harmless and safe for consumption, several studies have shown that natural products can be hazardous and possibly lethal [ref. 18]. The toxicity of Southern African propolis is yet to be explored, with only one study investigating the toxicity of a single whole propolis sample and the compounds pinocembrin, galangin and chrysin [ref. 17]. Thus, this study aimed to investigate a range of South African propolis samples for their antimicrobial activity against H. pylori, investigate the toxicity, and provide a chemical profile of samples demonstrating good antimicrobial activity using ultra‐performance liquid chromatography–mass spectrometry (UPLC‐MS).
Results and Discussion
Antimicrobial Activity
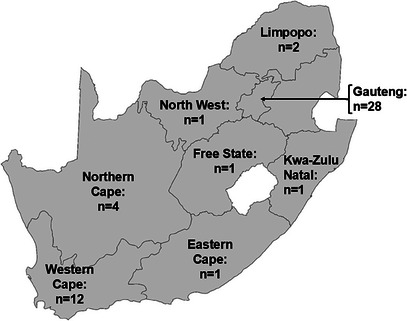
The minimum inhibitory concentration (MIC) values (n = 3) of 54 propolis ethanolic extracts tested against three clinical H. pylori strains are reported in Table 1. Good antimicrobial activity was recorded for the MIC values that were less than or equal to the mean MIC across all three Brazilian samples, 0.51 mg/mL. Good anti‐H. pylori activity was observed against at least one H. pylori strain for 27 South African samples, with notable mean MIC values across all three strains recorded for 19 propolis extracts. Good anti‐H. pylori activity was recorded for 15 samples from the Gauteng Province, six samples from the Western Cape, three samples from the Northern Cape and one sample each from Free State, Kwa‐Zulu Natal and Limpopo. The best activity was seen in samples 13 (Gauteng), 44 (Western Cape) and 37 (Northern Cape), with mean MIC values of 0.20, 0.20 and 0.23 mg/mL, respectively.
TABLE 1: The MIC values in mg/mL of South African propolis extracts against three clinical Helicobacter pylori strains represented as mean ± standard deviation.
| Propolis sample | Location | Province of origin | Clinical Strain 1 | Clinical Strain 2 | Clinical Strain 3 | Mean |
|---|---|---|---|---|---|---|
| 1 | Port Elizabeth—Baviaanskloof | Eastern Cape | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 2 | Bloemfontein | Free State | 0.39cbdv202403200-tbl1-note-0002 ± 0.00 | 0.59 ± 0.00 | 0.78 ± 0.00 | 0.59 ± 0.28 |
| 3 | Benoni—Bapsfontein | Gauteng | 0.78 ± 0.00 | 1.56 ± 0.00 | 0.39 ± 0.00 | 0.91 ± 0.43 |
| 4 | Bronkhorstspruit—Wilgerivier | Gauteng | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.29 ± 0.14 | 0.62 ± 0.19 |
| 5 | Edenvale | Gauteng | 0.20 ± 0.14 | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 0.23 |
| 6 | Edenvale | Gauteng | 0.29 ± 0.00 | 0.78 ± 0.00 | 0.20 ± 0.00 | 0.42 ± 0.19 |
| 7 | Johannesburg | Gauteng | 0.39 ± 0.00 | 0.78 ± 0.00 | 0.39 ± 0.00 | 0.52 ± 0.43 |
| 8 | Johannesburg | Gauteng | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.28 |
| 9 | Johannesburg | Gauteng | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 |
| 10 | Lakeside/Westlake | Gauteng | 0.78 ± 0.00 | 1.56 ± 0.00 | 0.39 ± 0.00 | 0.91 ± 0.47 |
| 11 | Midrand—Beaulieu | Gauteng | 0.10 ± 0.00 | 0.98 ± 0.00 | 0.10 ± 0.00 | 0.39 ± 0.37 |
| 12 | Midrand—President Park | Gauteng | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.18 |
| 13 | Pretoria | Gauteng | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.25 |
| 14 | Pretoria | Gauteng | 0.78 ± 0.00 | 3.13 ± 0.00 | 0.78 ± 0.00 | 1.56 ± 0.97 |
| 15 | Pretoria | Gauteng | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.10 |
| 16 | Pretoria East | Gauteng | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 17 | Pretoria—Groenkloof | Gauteng | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 18 | Pretoria—Lydiana Gardens | Gauteng | 0.78 ± 0.00 | 0.39 ± 0.00 | 0.59 ± 0.00 | 0.59 ± 0.26 |
| 19 | Pretoria—Lydiana Gardens | Gauteng | 0.20 ± 0.00 | 0.78 ± 0.00 | 0.20 ± 0.00 | 0.39 ± 0.29 |
| 20 | Pretoria—Northern | Gauteng | 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 21 | Sandton | Gauteng | 1.56 ± 0.00 | 3.13 ± 0.00 | 2.35 ± 0.00 | 2.35 ± 2.13 |
| 22 | Sandton | Gauteng | 0.78 ± 0.00 | 1.56 ± 0.00 | 1.56 ± 0.00 | 1.30 ± 0.51 |
| 23 | Sandton | Gauteng | 3.13 ± 0.00 | 3.13 ± 0.00 | 2.35 ± 0.00 | 2.87 ± 2.66 |
| 24 | Sandton—IDC Grayston Drive | Gauteng | 1.56 ± 0.00 | 3.13 ± 0.00 | 1.56 ± 0.00 | 2.08 ± 1.04 |
| 25 | Sandton—Paulshof | Gauteng | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.55 |
| 26 | Sandton—Riverclub | Gauteng | 6.25 ± 0.00 | 6.25 ± 0.00 | 6.25 ± 0.00 | 6.25 ± 2.74 |
| 27 | Sandton—Woodlands Eco Park | Gauteng | 1.56 ± 0.00 | 3.13 ± 0.00 | 3.13 ± 0.00 | 2.61 ± 0.82 |
| 28 | Sedibeng area—Devon | Gauteng | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.39 ± 0.00 | 0.65 ± 0.28 |
| 29 | Springs | Gauteng | 0.20 ± 0.00 | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 0.25 |
| 30 | Vereeniging—Walkerville | Gauteng | 0.20 ± 0.00 | 0.78 ± 0.00 | 0.20 ± 0.00 | 0.39 ± 0.30 |
| 31 | SANS | Kwa‐Zulu Natal | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.00 | 0.39 ± 0.31 |
| 32 | Amandelbult | Limpopo | 0.39 ± 0.00 | 0.78 ± 0.00 | 0.39 ± 1.11 | 0.52 ± 1.12 |
| 33 | Naboomspruit and Nylstroom | Limpopo | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 0.08 |
| 34 | Mooinooi | North West | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 35 | Douglas | Northern Cape | 0.39 ± 0.00 | 0.78 ± 0.00 | 0.59 ± 0.28 | 0.59 ± 0.18 |
| 36 | SANS | Northern Cape | 0.39 ± 0.00 | 0.78 ± 0.14 | 0.59 ± 0.00 | 0.59 ± 0.23 |
| 37 | SANS | Northern Cape | 0.20 ± 0.00 | 0.29 ± 0.00 | 0.20 ± 0.28 | 0.23 ± 1.45 |
| 38 | SANS | Northern Cape | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 |
| 39 | Orange River | Unknown | 1.56 ± 0.00 | 3.13 ± 0.00 | 2.35 ± 1.11 | 2.35 ± 0.86 |
| 40 | Beaufort West | Western Cape | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 |
| 41 | Botrivier | Western Cape | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.31 |
| 42 | Cape Town—Southern Suburbs | Western Cape | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 1.07 |
| 43 | Cape Town—Southern Suburbs | Western Cape | 0.10 ± 0.00 | 0.78 ± 0.00 | 0.10 ± 0.00 | 0.33 ± 0.25 |
| 44 | Cape Town—Southern Suburbs | Western Cape | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.35 |
| 45 | Graafwater | Western Cape | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.29 |
| 46 | Outeniqua Mountains—Oudtshoorn | Western Cape | 0.20 ± 0.00 | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 0.11 |
| 47 | SANS | Western Cape | 0.39 ± 0.00 | 0.78 ± 0.28 | 0.20 ± 0.00 | 0.46 ± 0.25 |
| 48 | SANS | Western Cape | 0.20 ± 0.00 | 0.39 ± 0.00 | 0.20 ± 0.00 | 0.26 ± 0.25 |
| 49 | Somerset West | Western Cape | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.28 |
| 50 | Stanford | Western Cape | 1.56 ± 0.00 | 1.56 ± 0.00 | 1.17 ± 0.55 | 1.43 ± 0.85 |
| 51 | Touwsrivier | Western Cape | 0.78 ± 0.00 | 1.56 ± 0.00 | 0.78 ± 0.00 | 1.04 ± 0.40 |
| B1 | Brazil | — | 0.39 ± 0.00 | 0.78 ± 0.00 | 0.39 ± 1.11 | 0.52 ± 0.77 |
| B2 | Brazil | — | 0.10 ± 0.00 | 0.10 ± 0.00 | 0.10 ± 0.00 | 0.10 ± 0.48 |
| B3 | Brazil | — | 1.17 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.55 | 0.91 ± 0.32 |
| Controls | ||||||
| Amoxicillin (µg/mL) | 0.31 ± 0.00 | 0.31 ± 0.00 | 0.16 ± 0.00 | 0.26 ± 0.08 | ||
| Acetone | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | > 6.25 ± 0.00 | ||
| Culture control | growth | growth | growth | growth |
Abbreviation: SANS, specific area not specified.
Good antimicrobial activity is denoted in bold.
While the antimicrobial activity of South African propolis against H. pylori has not been previously investigated, South African propolis extracts have previously shown promising antimicrobial activity against other Gram‐negative bacteria, where MIC values of 0.13, 0.16 and 0.21 mg/mL were observed against Pseudomonas aeruginosa, Escherichia coli and Klebsiella pneumoniae, respectively [ref. 17].
Propolis from Nigeria was found to possess anti‐H. pylori activity when assessed using the MIC and disc‐well assays [ref. 14, ref. 15]. Similar inhibition was found for both clinical and reference strains, where MIC values recorded for propolis were eightfold higher than the positive control, amoxicillin [ref. 15]. In a separate study, Nigerian propolis was investigated against H. pylori isolated from gastric biopsy patients, and zones of inhibition of 30.00 and 23.00 mm were observed for propolis at concentrations of 400.00 and 200.00 µL/mL, respectively [ref. 14].
Propolis from northern Spain was investigated against H. pylori with ethanolic and propylene glycol extracts which were found to have MIC values ranging between 6.00 and 14.00 mg/mL [ref. 19]. These findings support the results of this study, indicating that propolis does possess antimicrobial activity against H. pylori. The results further confirm that variation in biological activity does exist from region to region, as the chemical distinction of propolis is region‐specific.
Chemical Analysis
Samples 13, 37 and 44 displayed the best antimicrobial activity, with mean MIC values of 0.20, 0.23 and 0.20 mg/mL, respectively, and were thus selected for chemical analysis using UPLC‐MS. Liquid chromatography coupled with MS, particularly in the negative mode, allows for fast identification and accurate quantification of flavonoids that are ubiquitous in propolis [ref. 10, ref. 20, ref. 21]. Compounds were identified by matching retention times (Rt), mass–charge values (m/z), and fragmentation patterns to data available in literature and databases (Table 2). The chromatographic profiles observed (Figure 1) correlate with that previously reported for South African propolis and showed a similar profile to that of common temperate propolis [ref. 22].
TABLE 2: Compounds identified in ethanolic extracts of propolis samples 13, 37 and 44 using UPLC‐MS.
| Peak # | Rt (min) | Elemental formula | [M−H]− calculated | [M−H]− theoretical | Mass error (mDa) | Fragments | Compound ID |
|---|---|---|---|---|---|---|---|
| 1 | 3.05 | C9H8O3 | 163.0395 | 163.0367 | 3.4 | — | p‐Coumaric acid |
| 2 | 5.30 | C16H14O5 | 285.0763 | 285.0713 | 4.5 | 267, 267, 252, 239, 139 | Pinobanksin‐methyl ether |
| 3 | 5.84 | C15H12O5 | 271.0606 | 271.0558 | −4.9 | 253 | Pinobanksin |
| 4 | 6.04 | C16H11O6 | 299.0556 | 299.0499 | −5.7 | 284 | Kaempferol‐3‐methyl ether |
| 5 | 6.27 | C17H14O7 | 329.0601 | 329.0761 | −6.0 | 314, 299, 271 | Dimethoxyquercetin |
| 6 | 6.72 | C16H12O5 | 283.0606 | 283.0554 | −5.2 | 268, 239, 211 | 5‐Methoxygalangin |
| 7 | 7.52 | C15H10O4 | 253.0407 | 253.0454 | 4.7 | 145 | Chrysin |
| 8 | 7.67 | C15H12O4 | 255.0657 | 255.0593 | −6.4 | 213, 151 | Pinocembrin |
| 9 | 7.76 | C15H10O5 | 269.0450 | 269.0409 | −4.1 | 227 | Galangin |
| 10 | 7.88 | C17H14O6 | 313.0712 | 313.0674 | −3.8 | 253 | 3‐O‐Acetylpinobanksin |
| 11 | 8.18 | C16H12O5 | 283.0606 | 283.0554 | −5.2 | 268, 239, 211 | 5‐Methoxygalangin |
| 12 | 8.52 | C15H10O4 | 253.0407 | 253.0454 | 4.7 | 145 | Chrysin |
| 13 | 8.79 | C18H16O6 | 327.0869 | 327.0817 | −5.2 | 271, 253 | Pinobanksin‐3‐O‐propionate |
| 14 | 9.46 | C18H16O3 | 279.1021 | 279.0953 | −6.8 | p‐Cinnamyl coumarate | |
| 15 | 9.62 | C19H18O6 | 341.1025 | 341.0973 | −5.2 | 271, 253 | Pinobanksin‐3‐O‐butyrate |
| 16 | 10.40 | C16H20O9 | 355.1029 | 355.1127 | 9.8 | 253 | Pinobanksin‐3‐O‐pentanoate |
| 17 | 11.11 | C21H22O6 | 369.1338 | 369.1277 | −6.1 | 271, 253 | Pinobanksin‐3‐O‐hexanoate |
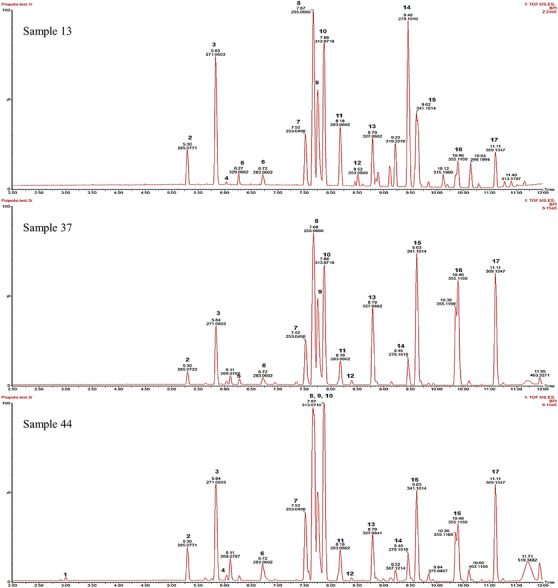
A total of 17 dominant compounds were identified from the samples, with pinocembrin, 3‐O‐acetylpinobanksin and pinobanksin most abundant. The compounds chrysin, p‐coumaric acid, pinobanksin‐methyl ether, pinobanksin‐3‐O‐propionate, pinobanksin‐3‐O‐butyrate, pinobanksin‐3‐O‐pentanoate, and pinobanksin‐3‐O‐hexanoate had previously been identified from South African propolis samples [ref. 22, ref. 23]. While the compounds kaempferol‐3‐methyl ether, dimethoxyquercetin, 5‐methoxygalangin, 3‐O‐acetylpinobanksin, and p‐cinnamyl coumarate were not previously identified in South African propolis, they were previously identified in South American, North American, Chinese and Mexican propolis, respectively [ref. 24, ref. 25, ref. 26, ref. 27, ref. 28].
It is plausible that the antimicrobial activity seen against H. pylori can be attributed mostly to pinocembrin, galangin and pinobanksin. These compounds have previously been noted for anti‐H. pylori activity [ref. 29]. The compounds chrysin, pinocembrin, galangin and caffeic acid phenyl ester were reported to show varying anti‐H. pylori activity, with caffeic acid phenyl ester being the most active, and synergy observed for the combination of chrysin with galangin [ref. 30]. Pinocembrin and galangin were previously reported to inhibit urease, an enzyme utilised by H. pylori during growth and colonization [ref. 2, ref. 31]. Inhibition of urease by these compounds could be attributed to the anti‐H. pylori activity displayed by propolis.
Toxicity
The toxicity of the investigated propolis extracts is summarized in Table 3. All propolis extracts investigated in this study were considered non‐toxic, with a percentage mortality of less than 50%. Brazilian propolis extracts were also non‐toxic, with an average mortality of 0.79% at 24 h and 3.73% at 48 h, for all three samples.
TABLE 3: The toxicity of propolis extracts shown as mean ± standard deviation percentage mortality at 24 and 48 h of exposure.
| Propolis sample | Mean percentage mortality | |
|---|---|---|
| 24 h | 48 h | |
| 1 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 2 | 0.00 ± 0.00 | 26.94 ± 1.36 |
| 3 | 0.72 ± 1.26 | 2.35 ± 2.44 |
| 4 | 1.08 ± 7.86 | 24.92 ± 2.74 |
| 5 | 7.20 ± 9.99 | 26.32 ± 1.55 |
| 6 | 6.58 ± 2.40 | 29.63 ± 4.07 |
| 7 | 0.52 ± 0.90 | 40.08 ± 0.95 |
| 8 | 0.79 ± 1.37 | 0.79 ± 1.37 |
| 9 | 1.12 ± 0.00 | 34.80 ± 2.68 |
| 10 | 1.55 ± 1.35 | 12.62 ± 7.87 |
| 11 | 0.00 ± 0.00 | 38.03 ± 4.10 |
| 12 | 14.30 ± 5.79 | 20.89 ± 2.71 |
| 13 | 0.00 ± 0.00 | 0.68 ± 1.18 |
| 14 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 15 | 9.98 ± 5.05 | 30.29 ± 1.17 |
| 16 | 0.00 ± 0.00 | 10.09 ± 1.09 |
| 17 | 0.76 ± 0.00 | 3.22 ± 2.06 |
| 18 | 0.90 ± 1.56 | 0.90 ± 1.56 |
| 19 | 0.00 ± 0.00 | 2.04 ± 1.79 |
| 20 | 0.00 ± 0.00 | 3.26 ± 3.61 |
| 21 | 0.81 ± 1.41 | 0.81 ± 1.41 |
| 22 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 23 | 0.00 ± 0.00 | 0.65 ± 1.13 |
| 24 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 25 | 0.00 ± 0.00 | 2.94 ± 3.22 |
| 26 | 0.00 ± 0.00 | 2.73 ± 2.37 |
| 27 | 0.00 ± 0.00 | 4.74 ± 2.45 |
| 28 | 33.09 ± 6.01 | 40.57 ± 1.88 |
| 29 | 0.53 ± 0.92 | 25.07 ± 5.24 |
| 30 | 0.00 ± 0.97 | 0.93 ± 0.87 |
| 31 | 0.00 ± 0.00 | 9.69 ± 1.45 |
| 32 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 33 | 0.00 ± 0.00 | 0.48 ± 0.99 |
| 34 | 0.00 ± 0.00 | 1.59 ± 2.75 |
| 35 | 3.96 ± 3.75 | 24.09 ± 4.62 |
| 36 | 0.00 ± 0.00 | 3.13 ± 1.48 |
| 37 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 38 | 0.00 ± 0.00 | 4.84 ± 1.76 |
| 39 | 0.71 ± 1.23 | 3.20 ± 3.19 |
| 40 | 0.00 ± 0.00 | 20.74 ± 0.84 |
| 41 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 42 | 28.37 ± 1.59 | 40.45 ± 3.33 |
| 43 | 27.67 ± 7.99 | 41.91 ± 2.76 |
| 44 | 7.54 ± 3.43 | 22.98 ± 3.50 |
| 45 | 0.00 ± 0.00 | 1.45 ± 2.51 |
| 46 | 2.12 ± 2.35 | 2.68 ± 2.41 |
| 47 | 2.50 ± 0.93 | 1.73 ± 1.64 |
| 48 | 9.62 ± 3.22 | 31.60 ± 3.87 |
| 49 | 0.00 ± 0.00 | 1.71 ± 2.95 |
| 50 | 0.00 ± 0.00 | 0.74 ± 1.28 |
| 51 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| B1 | 0.85 ± 0.00 | 4.16 ± 1.96 |
| B2 | 0.00 ± 0.00 | 1.94 ± 3.08 |
| B3 | 1.51 ± 1.33 | 5.09 ± 3.06 |
| Controls | ||
| Saltwater | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Potassium dichromate | 100.00 ± 0.00 | 100.00 ± 0.00 |
While an assessment of a wide range of South African propolis was not previously investigated, the toxicity of propolis compounds and one South African propolis sample was previously investigated using the brine shrimp lethality assay [ref. 17]. The propolis compounds pinocembrin, galangin and chrysin, were investigated alone and in combination. The compounds were found to be non‐toxic, with toxicity lower for the combined compounds than that independently. The toxicity of the whole propolis South African sample was also non‐toxic, at 14.78% [ref. 17]. Propolis from other regions were also found to be non‐toxic. Iranian propolis was investigated for toxicity in Wistar rats and found no toxicological differences between the experimental and control groups after 48 h [ref. 32].
Conclusion
While propolis has been extensively investigated, very little attention has been paid to its antimicrobial properties against H. pylori, especially regarding South African propolis. This study determined the antimicrobial activity of a wide variety of South African propolis against H. pylori and found that the majority of ethanolic propolis extracts had good activity, comparable to that of the gold standard, Brazilian propolis. The antimicrobial results of this study show that the chemically distinct South African propolis exhibits better anti‐H. pylori activity than that of other global regions. The compounds pinocembrin and pinobanksin were identified in samples displaying the best anti‐H. pylori activity and could possibly be responsible for the activity seen. All propolis samples investigated held little to no toxicity.
This study provides novel evidence for the potential of utilizing South African propolis in apitherapy for H. pylori infections and related ailments and identifies compounds that could be responsible for the anti‐H. pylori activity that was observed. Further investigations could explore the use of propolis and the identified compounds in vivo for reducing H. pylori bacterial loads and gastric ulcer healing properties.
Materials and Methods
Propolis Sample Collection and Preparation
A total of 51 propolis samples (Figure 2) from various regions (dependent on beehive availability) around South Africa were donated by local beekeepers affiliated with the South African Bee Industry Organization (SABIO). Three propolis samples were obtained from Brazil (supplied by Kim Morgado) to serve as the ‘gold standard’ control. Propolis samples were stored away from light and at ambient temperature.
Before processing, samples were frozen at −20°C for 24 h. Extracts were prepared by submerging ground propolis in absolute ethanol. For antimicrobial and toxicity studies, 0.30 g/mL solutions were incubated at 37°C for 7 days with shaking at 50 rpm, followed by drying under a fume hood [ref. 7]. For chemical analysis, 1.00 mg/mL solutions were shaken for 24 h, sonicated for 10 min and filtered through a 0.22 µm syringe filter.
Antimicrobial Studies
Three clinically confirmed and identified isolates of H. pylori were obtained for this study (Dr. Ayodeji Idowu—African Institute of Digestive Diseases, Chris Hani Baragwanath Academic Hospital, Gauteng, South Africa; Ethical clearance number M210891, Human Research Ethics Committee, University of the Witwatersrand) and stored at −80°C in 20% glycerol in brain heart infusion (BHI) media.
Antimicrobial testing was carried out using the broth microdilution assay to determine the MIC, where BHI supplemented with Campylobacter Selective Supplement (Skirrow) and 7% foetal bovine serum was used. Propolis extracts were made up to 25.00 mg/mL using acetone, and a twofold serial dilution was created in 96‐well microtiter plates. Cultures of H. pylori made up to 0.50 McFarland turbidity standard were added to each well. The plates were sealed and incubated in a micro‐aerobic environment using an anaerobic tank and a microaerobic gas pack (CampyGen) for 24 h at 37°C. A positive control of 0.01 mg/mL amoxicillin, a negative control of acetone alone, and a culture control were included. A solution of 0.02 mg/mL p‐iodonitrotetrazolium violet was used to indicate the viability of cultures in each well. The MIC of each sample was taken as the lowest concentration that inhibited growth completely [ref. 33]. All assays were carried out in triplicate, and the mean and standard deviation were recorded.
Chemical Analysis
Samples that demonstrated the best antimicrobial activity were selected for chemical analysis using ultra‐performance liquid chromatography–quadrupole time of flight–mass spectrometry (UPLC‐QToF‐MS) (Department of Pharmaceutical Sciences, Tshwane University of Technology). Chromatographic analyses were performed on a Waters Acquity I Class Ultra‐Performance Liquid Chromatographic system coupled to a PDA detector (Waters, Milford, MA, USA). Chromatographic separation was achieved on an Acquity UPLC BEH C18 column (150 mm × 2.10 mm, i.d., 1.70 µm particle size; Waters) maintained at 40°C. The mobile phase consisted of 0.1% formic acid in water (Solvent A) and 0.1% formic acid in acetonitrile (Solvent B) at a flow rate of 0.35 mL/min. The gradient elution was as follows: 90% A: 10% B, changing to 60% A: 40% B in 2.5 min, changing to 55% A: 45% B in 8.5 min, changing to 20% A: 80% B in 1.5 min, keeping for 0.5 min, back to the initial ratio in 0.5 min and equilibrating the system for 1.5 min. The total running time was 15 min. The samples were injected in the mobile phase with an injection volume of 1.00 µL (full‐loop injection). Data were collected and processed by the chromatographic software MassLynx 4.2.
Mass spectrometry (G3 QTof, Waters) was operated in the negative ion electrospray mode. Nitrogen was used as the desolvation gas. The desolvation temperature was set to 350°C at a flow rate of 550 L/h and the source temperature was 100°C. The capillary and cone voltages were set to 2500 and 40 V, respectively. Data were collected between 100 and 1200 m/z. During acquisition, the LockSpray interface was used to ensure mass accuracy and reproducibility.
Toxicity Studies
The brine shrimp lethality assay was used to assess the toxicity, where Artemia franciscana (brine shrimp) eggs (Ocean Nutrition) were hatched in 32.00 g/L saltwater and exposed to light. Propolis extracts were made up to 1.00 mg/mL using 2% DMSO. Aliquots (400 µL) of brine shrimp in saltwater (40–60 shrimp) and propolis extracts were added in triplicate to the wells of a 48‐well micro‐titre plate. Dead shrimp were counted at 0, 24 and 48 h. Glacial acetic acid was added after 48 h, the total dead shrimp in each well were counted, and the percentage mortality was calculated. Samples with a mortality percentage greater than 50% are considered toxic. A positive control of 1.60 mg/mL potassium dichromate and a negative control of 32.00 g/L saltwater were included in each assay [ref. 34].
Author Contributions
Sarhana Dinat: writing – original draft, methodology, investigation, funding acquisition, formal analysis, data curation, conceptualization. Ané Orchard: writing – review and editing, supervision, funding acquisition, conceptualization. Efficient Ncube: writing – review and editing, methodology, investigation, formal analysis. Weiyang Chen: writing – review and editing, methodology, investigation, formal analysis. Alvaro Viljoen: writing – review and editing, resources. Sandy Van Vuuren: writing – review and editing, supervision, software, resources, project administration, funding acquisition, conceptualization.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- A South African Perspective on Helicobacter pylori: Prevalence, Epidemiology and Antimicrobial Chemotherapy,”. African Journal of Microbiology Research, 2013
- Pathogenesis of Helicobacter pylori Infection,”. Clinical Microbiology Reviews, 2006. [PubMed]
- Worldwide H. pylori Antibiotic Resistance: A Systematic Review,”. Journal of Gastrointestinal and Liver Diseases, 2010. [PubMed]
- Current Understanding and Management of Helicobacter pylori Infection: An Updated Appraisal,”. F1000Research, 2018
- Historical Aspects of Propolis Research in Modern Times,”. Evidence‐Based Complementary and Alternative Medicine, 2013
- Biological Activity of Bee Propolis in Health and Disease,”. Asian Pacific Journal of Cancer Prevention, 2006. [PubMed]
- Antimicrobial Activity and Chemometric Modelling of South African Propolis,”. Journal of Applied Microbiology, 2015. [PubMed]
- Antioxidative and Gastroprotective Activities of Anti‐Inflammatory Formulations Derived From Chestnut Honey in Rats,”. Nutrition Research, 2006
- Honey as an Apitherapic Product: Its Inhibitory Effect on Urease and Xanthine Oxidase,”. Journal of Enzyme Inhibition and Medicinal Chemistry, 2016. [PubMed]
- Propolis and Its Gastroprotective Effects on NSAID‐Induced Gastric Ulcer Disease: A Systematic Review,”. Nutrients, 2021
- Propolis and Its Potential to Treat Gastrointestinal Disorders,”. Evidence‐Based Complementary and Alternative Medicine, 2018. [PubMed]
- Propolis: The Future Therapy Against Helicobacter pylori‐Mediated Gastrointestinal Diseases,”. Journal of Applied Biomedicine, 2018
- Propolis Research and the Chemistry of Plant Products,”. Natural Product Reports, 2011. [PubMed]
- Anti‐Helicobacter pylori Activities of Nigerian Propolis (Bee Product),”. Planta Medica, 2013
- Nigerian Propolis: Chemical Composition, Antioxidant Activity and α‐Amylase and α‐Glucosidase Inhibition,”. Natural Product Research, 2019. [PubMed]
- A Scoping Review of African Natural Products against Gastric Ulcers and Helicobacter pylori ,”. Journal of Ethnopharmacology, 2022
- The New Buzz: Investigating the Antimicrobial Interactions Between Bioactive Compounds Found South African Propolis,”. Journal of Ethnopharmacology, 2019
- Assessing African Medicinal Plants for Efficacy and Safety: Pharmacological Screening and Toxicology,”. Journal of Ethnopharmacology, 2004. [PubMed]
- The Antimicrobial Effects of Propolis Collected in Different Regions in the Basque Country (Northern Spain),”. World Journal of Microbiology and Biotechnology, 2022
- Mass Spectrometry in the Structural Analysis of Flavonoids,”. Journal of Mass Spectrometry, 2004. [PubMed]
- Recent Advances and Trends in the Liquid‐Chromatography–Mass Spectrometry Analysis of Flavonoids,”. Journal of Chromatography A, 2016. [PubMed]
- Chemical Profiling and Chemometric Analysis of South African Propolis,”. Biochemical Systematics and Ecology, 2014
- Antioxidant Activity of Propolis of Various Geographic Origins,”. Food Chemistry, 2004
- Chemical Composition of North American Bee Propolis and Biological Activity Towards Larvae of Greater Wax Moth (Lepidoptera: Pyralidae),”. Journal of Chemical Ecology, 1994. [PubMed]
- Cytotoxicity of Constituents From Mexican Propolis Against a Panel of Six Different Cancer Cell Lines,”. Natural Products Communications, 2010
- The Strong Anti‐Kinetoplastid Properties of Bee Propolis: Composition and Identification of the Active Agents and Their Biochemical Targets,”. Molecules, 2020
- Bioactive Components and Mechanisms of Poplar Propolis in Inhibiting Proliferation of Human Hepatocellular Carcinoma HepG2 Cells,”. Biomedicine & Pharmacotherapy, 2021
- Phytochemical Constituents of Propolis Flavonoid, Immunological Enhancement, and Anti‐Porcine Parvovirus Activities Isolated From Propolis,”. Frontiers in Veterinary Science, 2022
- Revisiting Therapeutic Strategies for H. pylori Treatment in the Context of Antibiotic Resistance: Focus on Alternative and Complementary Therapies,”. Molecules, 2021
- Propolis Polyphenolic Compounds Affect the Viability and Structure of Helicobacter pylori In Vitro ,”. Revista Brasileira de Farmacognosia, 2019
- Effect of In Vitro Simulated Digestion on the Anti‐Helicobacter pylori Activity of Different Propolis Extracts,”. Journal of Enzyme Inhibition and Medicinal Chemistry, 2023
- Chemical Composition, Oral Toxicity and Antimicrobial Activity of Iranian Propolis,”. Food Chemistry, 2007
- Antimicrobial Activity of South African Medicinal Plants,”. Journal of Ethnopharmacology, 2008. [PubMed]
- Antimicrobial Activity and Toxicity Profile of Selected Southern African Medicinal Plants Against Neglected Gut Pathogens,”. South African Journal of Science, 2019
