The impact of using cannabis during pregnancy on the infant and mother: An overview of systematic reviews, evidence map, targeted updates, and de novo synthesis
Abstract
Background:
Cannabis use during pregnancy is becoming more prevalent. While numerous studies have explored the relationship of cannabis use during pregnancy and outcomes for mothers and infants, uncertainty remains regarding the impact of cannabis use on pregnancy complications and later‐life outcomes for offspring.
Aims:
To produce a summary of the short and long‐term effects of prenatal cannabis exposure on fetal growth and development, neonatal conditions, later‐life, and maternal outcomes.
Materials and Methods:
An overview of systematic reviews, an evidence and gap map, targeted updates of previous reviews, and de novo evidence synthesis was conducted. The databases searched include PubMed (National Center for Biotechnology Information); MEDLINE (Ovid); Embase (Ovid) and CINAHL with Full Text (EBSCO). Assessment of risk of bias was conducted in duplicate for all studies. Relevant studies were coded and are presented as an evidence and gap map. Where possible, meta‐analyses were conducted with a narrative synthesis of the results. Primary studies and systematic reviews examining the relationship between cannabis consumption in pregnancy and the effect on fetal/child development, antenatal, and obstetric outcomes during pregnancy were eligible for inclusion.
Results:
There were 89 studies/reviews eligible for inclusion in this review. There was a potentially harmful impact of prenatal cannabis exposure on all fetal growth and development outcomes, some neonatal outcomes, some later‐life outcomes, and some maternal outcomes. The evidence regarding other neonatal conditions, later‐life, and maternal outcomes was mixed.
Conclusions:
The evidence suggests cannabis should be avoided during pregnancy.
Article type: Review Article
Keywords: female, fetal development, hallucinogens, humans, infant, newborn, systematic reviews as topic
Affiliations: Health Evidence Synthesis, Recommendations and Impact (HESRI), School of Public Health University of Adelaide Adelaide Australia; JBI, School of Public Health University of Adelaide Adelaide Australia; The Robinson Research Institute University of Adelaide Adelaide Australia; Albert S. Cook Library Towson University Maryland USA
License: © 2024 The Author(s). Australian and New Zealand Journal of Obstetrics and Gynaecology published by John Wiley & Sons Australia, Ltd on behalf of Royal Australian and New Zealand College of Obstetricians and Gynaecologists. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/ajo.13916 | PubMed: 39699257 | PMC: PMC12282043
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.9 MB)
INTRODUCTION
The prevalence of cannabis (marijuana) use has become widespread among the general population for medical and recreational purposes, with an estimated prevalence of 2 to 5% in Australia, Canada and USA.ref. ajo13916-bib-0001, ref. ajo13916-bib-0002, ref. ajo13916-bib-0003 Recent changes in legislation surrounding cannabis consumption has brought about significant shifts in usage trends and perceptions of associated risks,ref. ajo13916-bib-0001, ref. ajo13916-bib-0004 leading to a decreased perception of cannabis as harmful and a simultaneous increase in its use.ref. ajo13916-bib-0005 This evolving landscape introduces a challenge in understanding the risks and consequences of cannabis use, particularly among pregnant women, potentially influencing their decisions about using cannabis during pregnancy.ref. ajo13916-bib-0002, ref. ajo13916-bib-0005, ref. ajo13916-bib-0006, ref. ajo13916-bib-0007, ref. ajo13916-bib-0008
It has been found that some pregnant women turn to cannabisref. ajo13916-bib-0001, ref. ajo13916-bib-0009 for relief from various conditions, including pre‐existing health concerns and challenges related to pregnancy. A notable motivation for this choice is the belief that cannabis can alleviate nausea during early pregnancy, as well as manage appetite and mood.ref. ajo13916-bib-0010 This belief rests on the assumption that the associated risks for both the expectant mother and the developing fetus are minimal or negligible.ref. ajo13916-bib-0011, ref. ajo13916-bib-0012, ref. ajo13916-bib-0013 However, the scientific community remains divided on this issue, with ongoing research needed to provide clearer guidance.
Numerous studies of varying quality have explored the relationship between cannabis use during pregnancy and its effects on both the mother and the infant. Tetrahydrocannabinol (THC), the psychoactive component of cannabis, is believed to be transferred from the mother to the fetus through the placenta during pregnancy.ref. ajo13916-bib-0008, ref. ajo13916-bib-0014, ref. ajo13916-bib-0015, ref. ajo13916-bib-0016 This transfer raises concerns about potential developmental disruptions to the fetus as previous studies on humans and animals have found harmful effects.ref. ajo13916-bib-0003 Furthermore, research has found harmful effects of cannabis on the person ingesting the drug.ref. ajo13916-bib-0017 Despite these research efforts, uncertainty remains regarding the impact of maternal cannabis use on pregnancy complications and later‐life outcomes for offspring.ref. ajo13916-bib-0001, ref. ajo13916-bib-0007, ref. ajo13916-bib-0009 Unlike the well‐established understanding of the effects of other substances like tobacco and alcohol, the effects of maternal and in utero cannabis use has not been appropriately collated and effectively communicated to policy makers, highlighting the need for concentrated efforts in this area.ref. ajo13916-bib-0001
As such, clear and straightforward communication that accurately conveys the body of evidence related to the potential consequences of prenatal cannabis exposure on the mother, the fetus, and the child as they develop is crucial.ref. ajo13916-bib-0013 Bridging this knowledge gap and ensuring effective communication to all relevant parties is essential for informed decision‐making by expectant mothers, healthcare providers, and policymakers. Our goal is to contribute to this effort by providing a comprehensive overview of the impact of cannabis use during pregnancy across a range of outcomes.
REVIEW QUESTIONS
Question 1: For infants exposed prenatally to cannabis, what are the effects of cannabis on fetal development, neonatal withdrawal, birth outcomes, infant development, and child development (up to age 16 years)?
Question 2: For women who use cannabis in pregnancy, what are the effects of cannabis on antenatal/obstetric outcomes?
MATERIALS AND METHODS
The protocol was registered with PROSPERO (CRD42023390292). An ethics statement is not applicable as this is a review of studies. To conduct this review in a rigorous and timely manner, an approach similar to ‘GRADE Adolopment’ref. ajo13916-bib-0018 for adapting, adopting or developing new guidelines where none exist was applied. Initially, an overview of reviews approach was followed to identify the ‘best estimate of the effect’, which is defined as the association between maternal cannabis exposure and the prioritised outcomes of interest. This estimate was used for a particular outcome when a credibleref. ajo13916-bib-0019 up‐to‐date systematic review (from 2018 onwards, in an attempt to include only recent evidence) reported the outcome. Certainty in these results was established using the Grading of Recommendations Assessment Development and Evaluation (GRADE) approach.ref. ajo13916-bib-0018
Where multiple systematic reviews existed, the estimate that was deemed the most credible was used as the basis for a targeted update. Where multiple reviews existed assessing the same outcome and they were of similar credibility, the congruency of the results was assessed and, where needed, an updated or new meta‐analysis was developed.
Where new studies that reported an association between the exposure and the outcome of interest were identified, the best estimate from the systematic review was updated. Where studies report prioritised outcomes and no systematic reviews existed, we conducted our own synthesis of these results on studies post‐2018.
Furthermore, an evidence map of all studies from 2018 that met the inclusion criteria was developed. The following items were used to categorise the studies in EPPI‐Reviewerref. ajo13916-bib-0020 (Site Licence; Institute of Education, University of London): outcome domain, outcome measured, date of study, study location, study design.
Inclusion criteria
Population and exposure
This review focuses on pregnant women and infants exposed to cannabis. It targeted studies clearly linking cannabis consumption (as the primary drug in cases of poly‐drug use) to fetal and child development. The scope included papers that reviewed poly‐drug consumption and concurrent tobacco smoking, in cases where the paper was able to comment on the quality of the evidence concerning cannabis despite concurrent use of other drugs or tobacco.
Studies exploring cannabis’s effect on antenatal/obstetric outcomes during pregnancy were included. Cannabis may have been taken through any route of administration (eg smoked or ingested).
Comparator
No cannabis use during pregnancy.
Outcomes
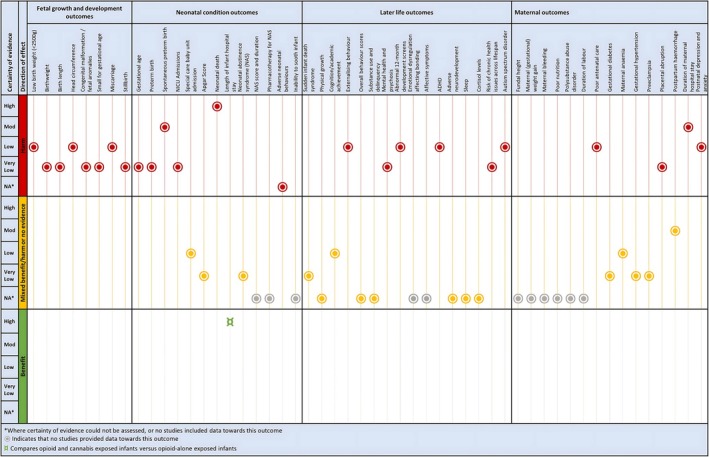
Figure 3 lists all outcomes that contributed data organised into four outcome domains. A full list of prioritised outcomes is presented in an online repository (osf.io/6f8w3). These were informed by the National Academy of Sciences review into prenatal cannabis exposureref. ajo13916-bib-0001 and supplemented by the author team and other literature.
Types of studies
Peer‐reviewed studies from The Organisation for Economic Cooperation and Development (OECD) countries published in the English language that specified an effect of cannabis exposure and its impact on fetal or child development or maternal antenatal and obstetric outcomes were included. Credibleref. ajo13916-bib-0018, ref. ajo13916-bib-0021 systematic reviews/meta‐analyses from 2018 were included. The evidence map included all relevant studies from 2018 and informed targeted updates or de novo synthesis.
The following criteria was used to determine credible reviews:
- published in the past five years (2018 onwards)
- included a comprehensive search strategy of two or more databases
- included formal critical appraisal/risk of bias assessment of included studies
- where multiple credible reviews existed, those that had performed a meta‐analysis and those that had applied GRADEref. ajo13916-bib-0022 were preferenced.
Only randomised controlled trials, cohort studies, and case–control studies were eligible for the evidence map and targeted updates. Cross‐sectional studies, case reports, and case series were excluded due to their likelihood of only providing very low certainty evidence. Despite considering randomised controlled trials, none were found due to the nature of the questions. Other study designs excluded were editorials studies by anonymous authors, conference abstracts, commentaries, animal studies, studies of prevalence and qualitative research. Studies where the population was too narrow and that were not likely to be applicable (ie cannabis exposure in opioid‐dependent women only) were excluded.
Search strategy
The search strategy was developed with the input of a health librarian and peer‐reviewed according to the Peer Review of Electronic Search Strategies (PRESS) Statementref. ajo13916-bib-0023 by another information scientist. An initial limited search of PubMed to identify relevant articles on this topic was undertaken. The terminology contained in the titles and abstracts of relevant articles and the related subject headings and index terms used to describe the articles were used to develop a full search strategy for PubMed. The search strategy, including all identified keywords and index terms, was adapted for each included database and/or information source, using Polyglotref. ajo13916-bib-0024 and with the aid of a health librarian. The search was limited to publication dates from January 1, 2018 through the search date of January 4, 2023. The full search strategies for major databases are available in an online repository (osf.io/6f8w3).
The databases searched included PubMed (National Center for Biotechnology Information); MEDLINE (Ovid); Embase (Ovid); and CINAHL with Full Text (EBSCO). In the protocol for this review, it was originally planned to supplement the database search with Epistemonikos and Google Scholar specifically for systematic reviews using the key terms ‘marijuana or cannabis’ and ‘pregnancy’. However, due to time and resource constraints, this supplemental search was not conducted.
Study screening and selection
All identified citations were collated and uploaded into EndNote™ and duplicates removed. Studies were imported into the Deduplicatorref. ajo13916-bib-0025 tool for additional deduplication, and then imported into Covidenceref. ajo13916-bib-0026 for screening. Two or more independent reviewers screened titles and abstracts against the inclusion criteria. Potentially relevant studies were retrieved in full and assessed against the inclusion criteria by two or more independent reviewers in EPPI‐Reviewer. Disagreements that arose at any stage of the selection process were resolved through discussion or with an additional author. The results of the search and the study inclusion process is reported in full and presented in a Preferred Reporting Items for Systematic Reviews and Meta‐analyses (PRISMA 2020) flow diagramref. ajo13916-bib-0027 for the overview of reviews, evidence map and targeted updates.
MAPPING THE EVIDENCE
Studies included at the full‐text stage of screening in EPPI‐Reviewer were then subjected to mapping and categorisation. The following items were used to categorise the studies: outcome domain, outcome measured, date of study, study location, study design.
Assessment of methodological quality/critical appraisal
Studies were assessed for risk of bias using either the JBI Cohort or JBI Case–control tools.ref. ajo13916-bib-0021, ref. ajo13916-bib-0028 One review author assessed the risk of bias, and this was double‐checked by another member. Where a credible systematic review existed, the original risk of bias assessments of the individual studies were applied where possible. Any disagreements that arose between the reviewers were resolved through discussion, or with an additional reviewer/s. Risk of bias was undertaken at the study level and modified for GRADE risk of bias considerations at the outcome level if needed when a study reported multiple outcomes and this had an impact on critical appraisal judgements.
Data extraction
Extraction forms were tailored by the research team for systematic reviews and primary studies (see online repository; osf.io/6f8w3). The data extracted included specific details about the participants, concept, context, study methods and key findings relevant to the question/s. Data extraction forms were piloted by all members of the research team. One reviewer extracted data from the included evidence sources which was double‐checked by another reviewer. Disagreements in extractions were resolved through discussion, or with an additional reviewer.
Data synthesis and meta‐analysis
Meta‐analyses were sourced from credible reviews. Where updates or de novo synthesis was needed, studies were pooled, where possible, in a meta‐analysis where two or more studies reported results for the same outcome in a format conducive to meta‐analysis using Review Manager 5 (RevMan5, Cochrane Training, London, England). Where there was only one study contributing data to a particular outcome for a comparison, a forest plot is still presented for consistency purposes and to facilitate interpretation of the data.
For dichotomous data, effect sizes were calculated as odds ratios and presented with 95% confidence intervals (CIs). Incidence rate ratios were calculated where incidence rates were reported. Weighted (or standardised) mean differences and their 95% CIs were calculated for analysis for continuous outcomes or outcomes assessed using scales. Adjusted estimates were preferred but unadjusted estimates were used if no adjusted estimates were available. Effect sizes reported as correlation or regression coefficients are discussed narratively due to the difficulties and (often) inappropriateness of combining these effect estimates. The choice of model (random or fixed effects) and method for meta‐analysis were based on the guidance by Tufanaru et al.ref. ajo13916-bib-0029 To facilitate meta‐analysis, interquartile ranges were converted to standard deviations where needed and possible.ref. ajo13916-bib-0030
The credible systematic reviews underpinning a best estimate of an effect for an outcome may have included studies of lower evidence than cohort or case–control studies. These estimates were still used even if they used lower levels of evidence; however, they were only updated with cohort or case–control studies.
For assessment of heterogeneity and publication bias, the I 2 was interpreted according to the thresholds and guidance in the Cochrane Handbook for Systematic Reviews of Interventions.ref. ajo13916-bib-0031 For meta‐analyses with more than ten studies, funnel plots were developed to investigate the possibility of publication bias. All detailed meta‐analyses and forest plots are presented in the online repository. Narrative summaries of the results are presented in this report.
Subgroup analyses were performed where relevant data existed and presented potentially important differences in the population or exposure (eg concomitant tobacco or opioid exposure).
GRADE
The GRADE approachref. ajo13916-bib-0032 was followed for grading the certainty of evidence and GRADE evidence profiles were created using GRADEpro GDT for each comparison for prognostic factors.ref. ajo13916-bib-0033 Evidence from observational studies begins with a ‘high certainty’ rating. In the GRADE approach for establishing certainty in the evidence relating to the effect of an intervention, evidence from non‐randomised studies begins as low certainty. However, as a prognostic factor was being assessed, this evidence begins as high in line with guidance from the GRADE Working Group.ref. ajo13916-bib-0033 The evidence profile presents the following information where appropriate: absolute risks for the exposed and control, estimates of relative risk, and a rating of the certainty of the evidence based on the risk of bias, indirectness, heterogeneity, imprecision and risk of publication bias of the review results. Unless otherwise specified, baseline/comparative risks come from the control event rate or averages, or baseline characteristics of the sample. The outcomes reported in the evidence profiles have been prioritised by the funders of this review.
RESULTS
A summary of results is presented in Figure 3, along with a GRADE Certainty of the Evidence rating for each included outcome.
Study inclusion
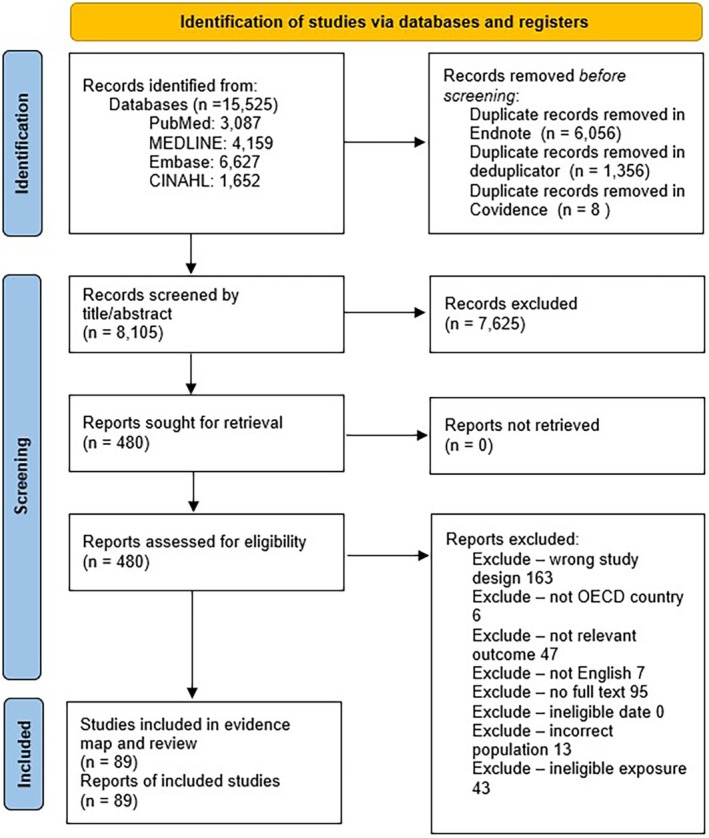
Following the exclusion of duplicate citations in Endnote™ and then in the Deduplicator tool, 8105 citations were identified for title and abstract screening in Covidence. There were 480 citations for full‐text review, with 391 studies excluded. A total of 89 reports were then included in this review (Fig. 1).
Methodological quality
The methodological quality of the studies substantially varied. In many studies, an issue identified was inadequate assessment of the exposure, as in many studies this was self‐reported as opposed to toxicology reports, urine tests or meconium testing. Another issue was not adequately addressing potential confounders in analyses. The full results of the study appraisals are available in the online repository.
Characteristics of included studies
This review included 89 studies overall. Of these, 58 were cohort studies, nine were case–control studies and 22 were systematic reviews. Most of the primary studies were from the US (n = 50).
The full details for the primary studies and credible reviews have been provided in the online repository. The full interactive evidence map is included in the online repository, with a static screenshot of the map provided in Figure 2.

Findings of the review
An overview of the results for each outcome that contributed data is presented in Figure 3.
Full analyses (including meta‐analysis and GRADE evidence profiles) are available in an online repository (osf.io/6f8w3).
FETAL GROWTH AND DEVELOPMENT OUTCOMES
For the domain of fetal growth and development, the evidence suggests a harmful impact on miscarriage, low birthweight, head circumference (low certainty), birthweight, birth length, congenital malformations, smallness for gestational age and stillbirth (very low certainty) (Table 1).
Table 1: Overview of findings for all outcomes
| Outcome | No. of systematic reviews (credible reviews) | No. of primary studies | Result (exposed vs unexposed) |
|---|---|---|---|
| Fetal growth and development | |||
| #Low birthweight (LBW) | 9 (3 credible) | 10 cohort studies | Significantly increased odds of LBW (odds ratio (OR) = 1.70, 95% CI: 1.44, 2.00) |
| #Birthweight (grams) | 8 (3 credible) | 13 cohort studies, and 2 case–control studies | Significantly decreased birthweight (mean deviation (MD) = −149.07 g, 95% CI: −197.19 g, −100.95 g) |
| #Birth length | None | 10 cohort studies | Significant reduction in birth length (MD = −0.86, 95% CI: −1.30, −0.41) |
| #Head circumference | 2 (1 credible) | 11 cohort studies, and one case–control study | Significantly decreased head circumference (MD = −0.59, 95% CI: −0.65, −0.53) |
| #Congenital malformation / fetal anomalies | 2 (1 credible umbrella review) | 6 cohort studies, and 1 case–control study | Risk ratio (RR): significant increase in the risk of congenital malformation / fetal anomaly (RR = 1.27, 95% CI: 1.17, 1.38)OR: significant increase in the odds of congenital malformation / fetal anomaly (OR = 2.02, 95% CI: 1.91, 2.14) |
| #Small for gestational age (SGA) | 5 (2 credible) | 7 cohort studies | Significant increase in the odds of SGA (OR = 1.60, 95% CI: 1.50, 1.70) |
| #Miscarriage | None | 1 cohort study | Significant increase in the risk of miscarriage/stillbirth (adjusted OR (aOR) = 12.1, 95% CI: 1.03, 141.8) |
| #Stillbirth | 6 (2 credible) | 7 cohort studies | RR: significant increase in the risk of stillbirth (RR = 1.63, 95% CI: 1.27, 2.09)OR: significant increase in the odds of stillbirth (OR = 1.29, 95% CI: 1.07, 1.56) |
| Neonatal conditions | |||
| #Gestational age | 6 (1 credible) | 15 cohort studies and 1 case–control study | Significant decrease in gestational age (MD = −0.20 weeks; 95% CI: −0.35, −0.05) |
| #Preterm birth and spontaneous preterm birth | 6 (4 credible) | 8 cohort studies | OR preterm birth: significant increase in the odds of preterm birth (OR = 1.36, 95% CI: 1.29, 1.44)OR spontaneous preterm birth: significant increase in the odds of spontaneous preterm birth (OR = 1.80, 95% CI: 1.68, 1.93)RR preterm birth: significant increase in the risk of preterm birth (RR = 1.26, 95% CI: 1.12, 1.42)RR spontaneous preterm birth: no association between prenatal cannabis exposure and risk of spontaneous preterm birth (RR = 1.21, 95% CI: 0.76, 1.93) |
| #Neonatal intensive care unit (NICU) admissions | 4 (4 credible) | 14 cohort studies and 1 case–control study | Significant increase in the odds of infants being admitted to the NICU (OR = 1.55, 95% CI: 1.25, 1.91) |
| #Special care baby unit admission | 1 (1 credible) | 1 cohort study | No association (aOR = 1.7, 95% CI: 0.7, 4.0) |
| #Apgar score | 3 (3 credible) | 11 cohort studies | Apgar score at 1 min: no association (MD = −0.23, 95% CI: 0.70, 0.23)Apgar score at 5 min: no association (MD = −0.04, 95% CI: −0.14, 0.05)Apgar score <7 at 5 min (RR): no association (RR = 1.21, 95% CI: 0.81, 1.80)Apgar score <7 at 5 min (OR): significant increase in the odds (OR = 1.50, 95% CI: 1.10, 2.05)Apgar score <7 at 1 min: no association (OR = 1.0, 95% CI: 0.8, 1.25)Apgar score <4 at 5 min: significant increase in the odds (RR = 1.95, 95% CI: 1.64, 2.32) |
| #Neonatal death | 1 (1 credible) | 4 cohort studies | Significantly associated with neonatal death (OR = 1.79, 95% CI: 1.42, 2.27) |
| #Length of infant hospital stay | None | 1 cohort study | The odds of a prolonged hospital stay were lower for prenatal cannabis exposure (OR = 0.68; 95% CI: 0.54, 0.86) |
| #Neonatal withdrawal / neonatal abstinence syndrome | 1 (1 credible) | 3 cohort studies | No significant association (OR = 0.83; 95% CI: 0.67, 1.02) |
| #Adverse neonatal behaviours | None | 1 case–control study | Self‐regulation: B = −0.185 (standard error (SE): 0.091, P = 0.043)Handling: B = 0.112 (SE: 0.061, P = 0.066)Attention: B = −0.185 (SE: 0.207, P = 0.371)Lethargy: B = 0.058 (SE: 0.051, P = 0.256) |
| Later‐life outcomes | |||
| #Sudden infant death syndrome (SIDS) | 1 (not credible) | 1 case‐cohort study | Full adjusted was an OR of 1.74 (95% CI: 0.29, 10.6; P > 0.500) |
| #Physical growth | None | 4 cohort studies | Not statistically significant |
| #Cognition/ academic achievement | 10 (1 credible) | 10 cohort studies. | Mixed results |
| #Behaviour (overall and externalising) | 9 (3 credible) | 11 cohort studies | Mixed results |
| #Mental health and psychosis | 5 (0 credible) | 2 cohort studies | Marijuana use after knowledge of pregnancy was associated with increased offspring psychosis proneness when adjusting for covariates (beta coefficient 1.41, 95% CI: 0.34, 2.48; P = 0.010)Psychotic like experiences in children aged 10: not statistically significant (1.37, 95% CI: 0.9, 2.08) |
| #Abnormal 12‐month development screens | 1 (0 credible) | 1 cohort study | Adjusted relative risk of 1.90 (95% CI: 0.92, 3.91) |
| #Attention‐deficit/hyperactivity disorder (ADHD) | 3 (0 credible) | 3 cohort studies | Hazards ratio (HR) of 1.14 (95% CI: 1.02, 1.28), indicating an association with prenatal cannabis exposure and ADHD |
| #Adverse neurodevelopment | 3 (0 credible) | 3 cohort studies | Developmental or behavioural diagnosis: children with progressive myoclonic epilepsy (PME) did not differ from those without PME in terms of the likelihood of having a developmental or behavioural diagnosis from a health professional (aOR: 0.95, 95% CI: 0.05, 17.21)Cerebral palsy (CP): exposure to cannabis and other intrauterine drugs provided a similar risk to developing CP (OR: 1.86, 95% CI: 0.65, 5.28).Atypical neurological exam at 6–9 months: unadjusted OR of 1.69 (95% CI: 0.54, 5.26)Atypical neurological exam at 12 months: unadjusted OR of 3.09 (95% CI: 0.76, 12.53) |
| #Autism spectrum disorders (ASD) | None | 2 cohort studies | Increased risk in the exposed group (HR 1.51, 95% CI: 1.17, 1.96) |
| Maternal outcomes | |||
| #Poor antenatal care | None | 1 cohort study | Pregnant people who consume cannabis are more likely to initiate care later (risk difference (RD) = 0.30, 95% CI: 1.15, 1.45) |
| Cannabis‐exposed pregnant people were less likely to receive adequate prenatal care (aOR = 0.59, 95% CI: 0.48, 0.73) | |||
| The cannabis‐exposed pregnant group had a lower likelihood of receiving a postpartum healthcare visit (OR = 0.65, 95% CI: 0.51, 0.82) | |||
| #Gestational diabetes | 2 (0 credible) | 6 cohort studies | No association between prenatal cannabis use and gestational diabetes (OR = 0.91, 95% CI: 0.66, 1.25) |
| #Maternal anaemia | None | 1 cohort study | Non‐significant finding of prenatal cannabis use and maternal anaemia (adjusted relative risk = 1.15, 95% CI: 0.93, 1.40) |
| #Gestational hypertension | None | 3 cohort studies | No significant association between prenatal cannabis use and an increased risk of gestational hypertension (OR = 1.06, 95% CI: 0.99, 1.14) |
| #Pre‐eclampsia | 1 (0 credible) | 7 cohort studies | No association between prenatal cannabis use and risk of pre‐eclampsia (OR = 0.93, 95% CI: 0.84, 1.04) |
| #Placental abruption | 2 (0 credible) | 5 cohort studies | A fixed effects meta‐analysis found an association between exposure to prenatal cannabis and increased risk of placental abruption (OR = 1.78, 95% CI: 1.62, 1.95) |
| #Postpartum haemorrhage | 1 (0 credible) | 2 cohort studies | No association between cannabis use within pregnancy and postpartum haemorrhage (OR = 1.04, 95% CI: 0.99, 1.09) |
| #Duration of hospital stay | None | 1 cohort study | Postnatal stay 3–6 days: Those exposed to cannabis prenatally were more likely to be discharged from hospital after 3–6 days (aOR = 0.97 95% CI: 0.95, 0.98) |
| Postnatal stay >7 days: Significant association | |||
| Pregnant people exposed to cannabis were more likely to stay longer (aOR = 1.17, 95% CI: 1.11, 1.23) | |||
| #Postpartum depression and anxiety | None | 1 cohort study | Anxiety: pregnant people who continued to use cannabis had a significantly higher risk of a higher generalised anxiety score than those who had never used cannabis (aOR = 2.55, 95% CI: 1.31, 4.96) |
| Depression: those who continued cannabis were more likely to have higher Edinburgh Postnatal Depression Scale scores compared with those who had never used (aOR = 2.75, 95% CI: 1.43, 5.29) | |||
NEONATAL OUTCOMES
For neonatal outcomes, a harmful impact of cannabis consumption was found on the following outcomes: neonatal death (high certainty), spontaneous preterm birth (moderate certainty), gestational age and neonatal intensive care unit (NICU) admissions (very low certainty evidence). There was mixed, equivocal or no evidence for special baby care unit admission, neonatal abstinence syndrome (NAS), Apgar scores, use of pharmacotherapy for NAS, and adverse neonatal behaviours (Table 1).
LATER‐LIFE OUTCOMES
For later‐life outcomes, the evidence suggests a harmful impact on externalising behaviour, abnormal 12‐month development screens, attention‐deficit/hyperactivity disorder and autism spectrum disorders (low certainty) and mental health and psychosis, and the risk of chronic health issues across the lifespan (very low certainty). There was mixed, equivocal or no evidence of sudden infant death syndrome, physical growth, cognitive/academic achievement, overall behaviour, substance use and delinquency, adverse neurodevelopment, sleep and cortisol levels (Table 1).
MATERNAL OUTCOMES
For maternal outcomes, the evidence identified a harmful impact on the duration of maternal hospital stay (moderate certainty), poor antenatal care, postnatal depression and anxiety (low certainty) and placental abruption (very low certainty). There was mixed, equivocal or no evidence for fundal height, maternal weight gain, bleeding, nutrition, polysubstance abuse disorder, duration of labour, gestational diabetes, maternal anaemia, gestational hypertension, pre‐eclampsia, antepartum or postpartum haemorrhage (Table 1).
DISCUSSION
This review included 89 studies that met the inclusion criteria across seven countries, investigating the short and long‐term effects of prenatal cannabis exposure on fetal growth and development, neonatal conditions, later‐life, and maternal outcomes. Summarising the main results of this review, the cumulative and overall weight of evidence indicates prenatal cannabis exposure has a harmful impact on most outcomes for both the mother and the offspring.
This review was able to address key questions regarding the impact of prenatal cannabis on a range of outcomes. Although studies to address most of the prioritised outcomes of interest were found, many outcomes were only informed from data from a small selection of these studies. Where statistically and clinically appropriate, results were combined across studies in a meta‐analysis, available in the online repository (osf.io/6f8w3). To enable this pooling, at times it was necessary to use data presented in the papers and convert these to a form suitable for pooling. Sometimes, the data were simply not accessible in a format that facilitated meta‐analysis. Where possible, the authors chose to combine studies in a meta‐analysis, despite the exposures and settings being somewhat different. Studies rarely distinguished different levels of cannabis intake and rarely discussed the timing of cannabis exposure. It was also difficult to determine (for some outcomes) the impact of co‐exposures such as tobacco use. Care must also be taken in the interpretation of the outcomes presented in this review, that report a significant effect, but have only been informed by the single study. The findings of this review have indicated that prenatal cannabis exposure is linked to increases in miscarriage, length of infant hospital stay, rates of poor antenatal care, and maternal anxiety and depression. However, these findings are only based on the results of a single cohort study, with no credible systematic reviews available. The impact of prenatal cannabis exposure on these outcomes requires further research effort, to better guide care and support.
The strengths of this review include its pragmatic approach and blend of relevant synthesis methods to ensure provision of the best available estimates of the impact of cannabis exposure. Modern evidence synthesis approaches were followed for establishing the certainty of the evidence by applying the GRADE approach for prognostic factors. Despite these strengths, this is a ‘restricted’ reviewref. ajo13916-bib-0034; therefore the authors made some design choices to expedite the review process. First, the review team did not search for unpublished studies, studies in languages other than English were excluded, and a search prior to 2018 was not undertaken. Where possible, the authors did assess for publication bias through visual inspection of funnel plots but this was only feasible for a small number of analyses given the lack of studies for the majority of outcomes. Where funnel plots were developed, publication bias was not detected. However, there remains the possibility that there are several studies potentially finding no association between cannabis and adverse outcomes that have not been published or were not identified during the search of this review.
In terms of the applicability of the results of this review, most studies were conducted in the US where there are potentially important differences in terms of legislation, cannabis use patterns and social demographics as compared to other countries. A further limitation of this review is that it does not include original studies not captured in credible reviews published earlier than 2018. However, this does suggest that the main conclusions are based on the most recent evidence and studies. This may increase the directness and applicability of this evidence given the changing patterns of cannabis consumption and the types and strength of cannabis available. Regardless of some of these applicability considerations, we believe there is a substantial body of evidence that warrants consideration, and these findings are applicable in many different contexts.
Mapping of the current state of the evidence helps to steer researchers in a productive direction and reduces redundant research efforts. The evidence and gap map conducted as part of this review identified many areas where there was mixed, equivocal or no evidence. Therefore, future research efforts in cannabis and pregnancy research could be focused on these areas identified in our review and discussed above, particularly maternal and later‐life outcomes.
CONCLUSION
This review has identified a substantial body of evidence supporting the negative impact of prenatal cannabis exposure. Although the evidence is not of high certainty across all outcomes, overwhelmingly the authors of included studies advised against cannabis intake during pregnancy where possible. The findings of this review support that recommendation.
FUNDING
This review is supported by a National Health and Medical Research Council (NHMRC) Ideas Grant (APP2011958).
This work is funded by the New South Wales (NSW) Ministry of Health Centre for Alcohol and Other Drugs and coordinated by the Sax Institute.
Zachary Munn is supported by a NHMRC Investigator Grant (1195676).
DISCLAIMER
This review was an Evidence Check rapid review brokered by the Sax Institute for the NSW Ministry of Health.
This Evidence Check review was produced using the Evidence Check methodology in response to specific questions from the commissioning agency.
It is not necessarily a comprehensive review of all literature relating to the topic area. It was current at the time of production (but not necessarily at the time of publication). It is reproduced for general information and third parties rely upon it at their own risk.
CONSTRAINTS ON PUBLISHING
None.
AUTHOR CONTRIBUTIONS
Each author certifies that their contribution to this work meets the standards of the International Committee of Medical Journal Editors. ZM, DP, SH, TB, JS, THB: conceptualisation, data curation, formal analysis, investigation, methodology, project administration, resources, software, validation, visualisation, writing – original draft preparation, writing – review and editing. MS, CP, AG: conceptualisation, methodology, resources, supervision, validation, writing – review and editing.
References
- 1 National Academies of Sciences, Engineering, and Medicine . The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington (DC): National Academies Press (US), 2017.
- 2 Health AIo, Welfare . Alcohol, Tobacco & Other Drugs in Australia. Canberra: AIHW, 2024.
- The prevalence and significance of gestational cannabis use at an Australian tertiary hospital.. Aust N Z J Obstet Gynaecol, 2023. [PubMed]
- Trends and correlates of cannabis use in pregnancy: A population‐based study in Ontario, Canada from 2012 to 2017.. Can J Public Health, 2019. [PubMed]
- Association of State Recreational Marijuana Laws with Adolescent Marijuana use.. JAMA Pediatr, 2017. [PubMed]
- Trends in marijuana use among pregnant and nonpregnant reproductive‐aged women, 2002–2014.. JAMA, 2017. [PubMed]
- Intrauterine cannabis exposure affects fetal growth trajectories: The generation R study.. J Am Acad Child Adolesc Psychiatry, 2009. [PubMed]
- Birth outcomes associated with cannabis use before and during pregnancy.. Pediatr Res, 2012. [PubMed]
- The deleterious effects of cannabis during pregnancy on neonatal outcomes.. Med J Aust, 2020. [PubMed]
- Association between gestational cannabis exposure and maternal, perinatal, placental, and childhood outcomes.. J Dev Orig Health Dis, 2021. [PubMed]
- Women’s perspectives about cannabis use during pregnancy and the postpartum period: An integrative review.. Prev Med, 2019. [PubMed]
- Pregnant People’s perspectives on cannabis use during pregnancy: A systematic review and integrative mixed‐methods research synthesis.. J Midwifery Womens Health, 2022. [PubMed]
- Perceptions about cannabis use during pregnancy: A rapid best‐framework qualitative synthesis.. Can J Public Health, 2021. [PubMed]
- Effects of cannabis tetrahydrocannabinol on endocannabinoid homeostasis in human placenta.. Arch Toxicol, 2019. [PubMed]
- Cannabis use during pregnancy: Pharmacokinetics and effects on child development.. Pharmacol Ther, 2018. [PubMed]
- The role of the placenta in variability of fetal exposure to cocaine and cannabinoids: A twin study.. Can J Physiol Pharmacol, 2001. [PubMed]
- Cannabis (Marijuana) DrugFacts. National Institute on Drug Abuse.. 2019
- GRADE evidence to decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE‐ADOLOPMENT.. J Clin Epidemiol, 2017. [PubMed]
- Guidelines for the management of hypertensive disorders of pregnancy 2008.. Aust N Z J Obstet Gynaecol, 2009. [PubMed]
- EPPI‐reviewer 4.0: Software for Research Synthesis. London: Social Science Research Unit, Institute of Education, University of London.: EPPI Centre Software.. 2010
- Conducting systematic reviews of association (etiology): The Joanna Briggs Institute’s approach.. JBI Evid Implement, 2015
- GRADE: An emerging consensus on rating quality of evidence and strength of recommendations.. BMJ, 2008. [PubMed]
- PRESS peer review of electronic search strategies: 2015 guideline statement.. J Clin Epidemiol, 2016. [PubMed]
- Improving the translation of search strategies using the polyglot search translator: A randomized controlled trial.. J Med Libr Assoc, 2020. [PubMed]
- Deduplicator The Systematic Review Accelerator (SRA) Bond University.. 2023
- 26 Veritas Health Innovation . Covidence Systematic Review Software. Melbourne, Australia: Veritas Health Innovation, 2024.
- The PRISMA 2020 statement: An updated guideline for reporting systematic reviews.. BMJ, 2021. [PubMed]
- 28 Moola S , Munn Z , Tufanaru C et al. Systematic reviews of etiology and risk. In: Aromataris E , Munn Z , eds. JBI Manual for Evidence Synthesis. North Adelaide, Australia: JBI, 2020.
- Fixed or random effects meta‐analysis? Common methodological issues in systematic reviews of effectiveness.. Int J Evid Based Healthc, 2015. [PubMed]
- Estimating the sample mean and standard deviation from commonly reported quantiles in meta‐analysis.. Stat Methods Med Res, 2020. [PubMed]
- 31 Higgins JPTTJ , Chandler J , Cumpston M et al., eds. Cochrane Handbook for Systematic Reviews of Interventions, 2nd edn. Chichester (UK): John Wiley & Sons, 2019.
- GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables.. J Clin Epidemiol, 2011. [PubMed]
- GRADE guidelines 28: Use of GRADE for the assessment of evidence about prognostic factors: Rating certainty in identification of groups of patients with different absolute risks.. J Clin Epidemiol, 2020. [PubMed]
- The dark side of rapid reviews: A retreat from systematic approaches and the need for clear expectations and reporting.. Ann Intern Med, 2023. [PubMed]
