Revealing the therapeutic potential of synthetic cannabinoids: a systematic review of cannabinoid receptor binding dynamics and their implications for cancer therapy
Abstract
Background:
Cancer remains a major global health issue, prompting the need for innovative treatment approaches that extend beyond conventional methods such as chemotherapy and radiation. The endocannabinoid system (ECS), primarily the cannabinoid receptors CB1R and CB2R, presents a promising opportunity for cancer therapy by selectively targeting cell signaling pathways. This systematic review intends to explore the mode of action of synthetic cannabinoids as potential anticancer agents and their impact on tumor growth in various cancer cell lines.
Methods:
Of the 287 articles identified between January 1990 and July 2024, 27 studies met strict criteria focusing on their anticancer effects. Data extraction and quality assessment were conducted using GRADE criteria and the Cochrane Risk of Bias tool, ensuring robust evaluation of the studies’ reliability.
Results:
Various pharmacological actions of synthetic cannabinoids function as agonists, antagonists, and inverse agonists at the CB1R and CB2R receptors. Key findings indicate that CB2R agonists significantly reduce cancer cell proliferation through diverse mechanisms, with selective CB2R agonists effectively inhibiting cancer cell growth and survival. Studies involving CB1R antagonists, particularly in conjunction with CB2R agonists, highlight their role in blocking CB1R to either validate or enhance the efficacy of CB2R agonists in mitigating tumor growth. Inverse agonists targeting CB2R have shown moderate success in inducing cancer cell death by disrupting survival pathways. Notably, synthetic cannabinoid agonists display significant potential in targeting CB1 and CB2 receptors to inhibit tumor proliferation and promote apoptosis across various cancer types.
Conclusion:
The systematic review concludes that CB2R agonists can effectively inhibit tumor growth while inducing apoptosis in various cancers. Although CB1R agonists show potential in modulating cancer pathways, there is a notable lack of research on CB1 inverse agonists, emphasizing the need for further investigation. Additionally, the study advocates for greater exploration of mixed receptor agonist and receptor mode of action to validate these promising therapeutic approaches.
Graphical Abstract:
Article type: Review Article
Keywords: CB1 Receptor, CB2 Receptor, Synthetic Cannabinoid, Cancer, Agonist, Antagonist
Affiliations: https://ror.org/0149jvn88grid.412149.b0000 0004 0608 0662Department of Pharmaceutical Sciences, College of Pharmacy, King Saud Bin Abdulaziz University for Health Sciences, 11451 Riyadh, Saudi Arabia; https://ror.org/009p8zv69grid.452607.20000 0004 0580 0891Medical Research Core Facility and Platforms, King Abdullah International Medical Research Center (KAIMRC), Ministry of National Guards Health Affairs, Riyadh, Saudi Arabia; https://ror.org/02pecpe58grid.416641.00000 0004 0607 2419King Abdulaziz Medical City, National Guard Health Affairs (NGHA), Riyadh, Saudi Arabia; https://ror.org/0149jvn88grid.412149.b0000 0004 0608 0662Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, King Saud Bin Abdulaziz University for Health Sciences, Riyadh, Kingdom of Saudi Arabia; https://ror.org/05b0cyh02grid.449346.80000 0004 0501 7602Department of Biology, Faculty of Science, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia; https://ror.org/05b0cyh02grid.449346.80000 0004 0501 7602Microbiology and Immunology Unit, Natural and Health Sciences Research Center, Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s42238-025-00289-5 | PubMed: 40483537 | PMC: PMC12144815
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.5 MB)
Introduction
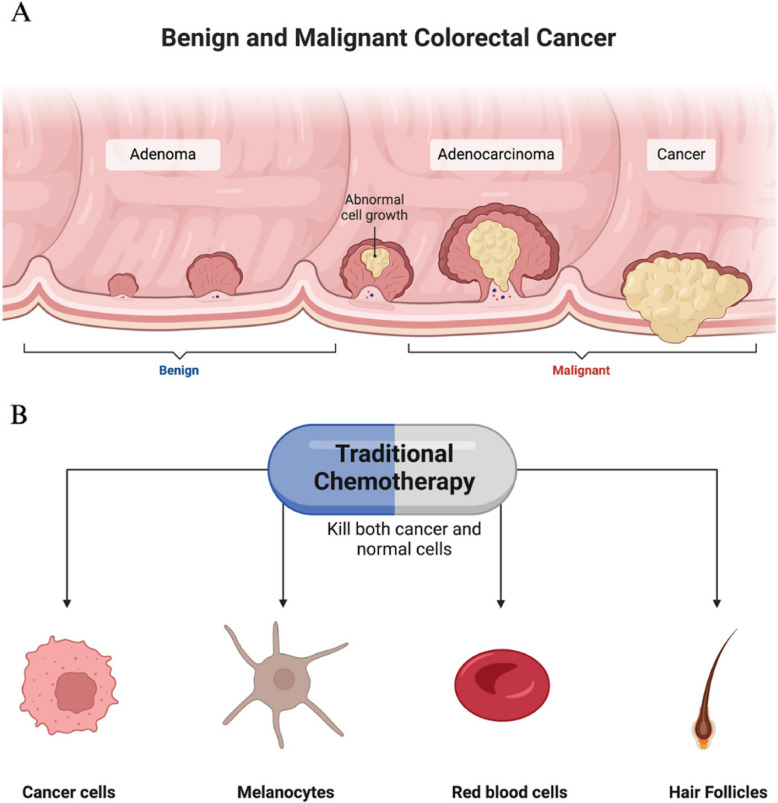
Cancer is a complex disease characterized by the abnormal growth of different types of cells in the body. There are over a hundred distinct types of cancer, each with different behaviors and responses to treatment. It is important to distinguish between benign and malignant tumors in cancer pathology (Jones ref. 2023), as illustrated in Fig. 1. In the World Health Organization (WHO) Eastern Mediterranean Region alone, over 788 000 individuals were diagnosed with cancer in ref. 2022. This number is anticipated to treble to reach 1.57 million cases by 2045, due to population increase and, more crucially, the high prevalence of cancer risk factors in the Region—such as tobacco use, obesity/overweight, physical inactivity, poor diets, and air pollution (Cancer, ref. 2025; Cancer Tomorrow, ref. 2025). In Saudi Arabia, the incidence of various cancers has tripled in recent years, likely due to lifestyle changes towards a Western model, limited cancer awareness, and inadequate screening programs. Key risk factors include obesity, genetics, sedentary habits, tobacco use, viral infections, and deficiencies in iodine and vitamin D (Ahmed et al. ref. 2024). The selective targeting of cancer cells remains a significant challenge in oncology, hindering the effectiveness of current therapeutic strategies. Therefore, there has been a growing emphasis on alternative treatments targeting molecular pathways involved in cancer formation and progression. A promising avenue for anticancer research lies in the recent discovery of cannabinoid receptor ligands (Walsh, ref. 2020).
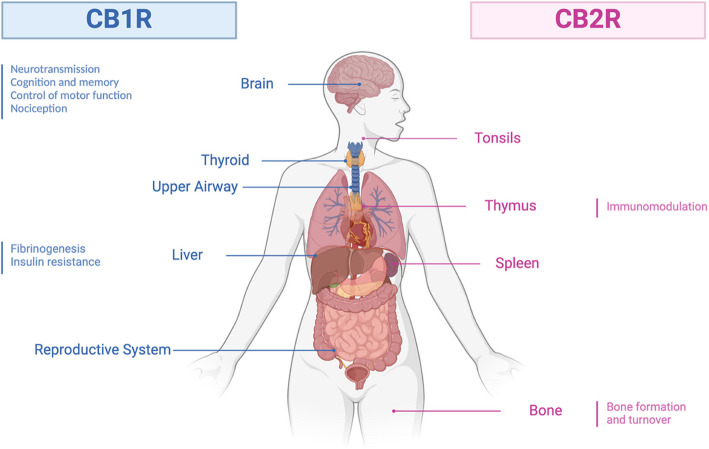
The endocannabinoid system (ECS) is a complex biological network composed of endogenous lipid-based retrograde neurotransmitters called endocannabinoids, cannabinoid receptors (CBRs), and the metabolic enzymes responsible for synthesizing and degrading endocannabinoids (Lu & Mackie ref. 2021). N-arachidonoyl-ethanolamine (AEA) and 2-arachidonoylglycerol (2-AG) are a major endocannabinoids in vivo system, as illustrated in Fig. 2 (Lu & Mackie ref. 2016). Cannabinoid receptor 1 (CB1R) and cannabinoid receptor 2 (CB2R), components of ECS, are G protein-coupled receptors primarily coupling to inhibitory G proteins (Moreno et al., ref. 2019). CB1R is primarily found in the central nervous system, while CB2R is predominantly found in the peripheral system, particularly in immune cells (Bie et al. ref. 2018), as shown in Fig. 3. CB2R plays a crucial role in inflammation, immunity, and cancer progression (Hashiesh et al. ref. 2021). This comprehensive system helps maintain homeostasis and regulates a wide range of physiological and pathological processes, including tumorigenesis and tumor suppression. Although the therapeutic potential of this intricate ECS network is promising, the direct anticancer properties of ligands targeting CBRs remain largely unexplored. Further investigation is needed to understand their potential as direct anticancer agents rather than as modulators of associated physiological processes.
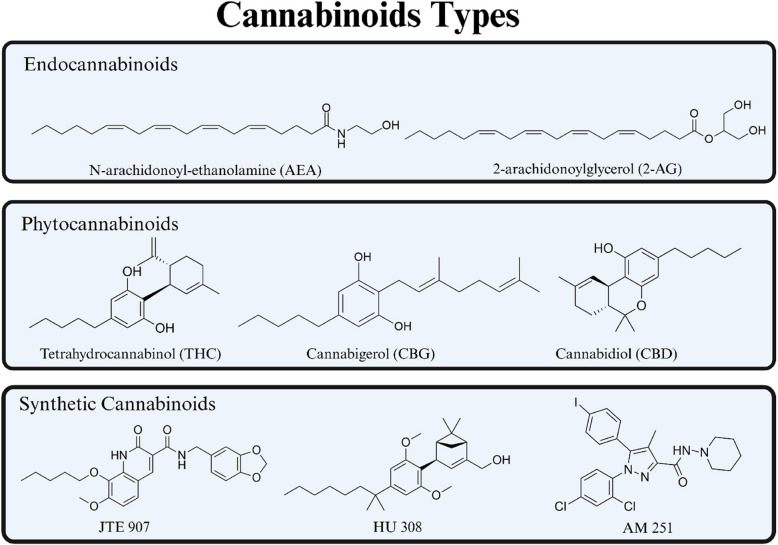
Phytocannabinoids, which are the natural cannabinoids found in Cannabis sativa, have been extensively studied for their potential anticancer effects (Koltai & Shalev ref. 2022). These compounds act as agonists for cannabinoid receptor 1 and cannabinoid receptor 2, facilitating their therapeutic applications through the activation of these CBRs (Gülck & Møller ref. 2020). By activating CB1R and CB2R, phytocannabinoids produce various therapeutic effects, including anti-nociception, anti-inflammation, anticonvulsant, and anti-emetic properties (Zou & Kumar ref. 2018). However, the recreational use of these compounds has predominantly limited their medical applications. An example of a phytocannabinoid is tetrahydrocannabinol (THC), as shown in Fig. 2, which acts as an agonist at CBRs and exhibits similar pharmacological effects to endocannabinoids (Maccarrone & Finazzi-Agró, ref. 2003).
Many newly synthesized cannabinoid analogs demonstrate high specificity toward CB2R. HU308 is a synthetic compound with agonistic behavior and selectivity for CB2R (Hanuš et al. ref. 1999). Furthermore, HU308 causes extracellular-regulated kinase (ERK1/2) and p38 to be down-regulated, and it also reduces the production of IL-6 and matrix metalloproteinase, specifically MMP-3 and MMP-13, in IL1β-activated fibroblast-like synoviocytes that are generated from synovial tissue from rheumatoid arthritis (Gui et al. ref. 2014). Nevertheless, research on HU308’s impact on anti-neoplastic activity and the mechanisms regulating it remained lacking.
Similarly, a recent investigation was conducted on the use of AM630 as a CB1R/CB2R antagonist in non-small cell lung cancer (NSCLC). According to the study, the agonist-mediated suppression of in vitro chemotaxis and chemoinvasion was reduced when a CB1R/CB2R-specific antagonist was administered (Preet et al., ref. 2011). On the other hand, JTE 907 is an extremely specific inverse agonist of CB2R. It has anti-inflammatory properties in vivo and binds to CB2 receptors in rats, mice, and humans with high-affinity Ki values of 0.38, 1.55, and 35.9 nM, respectively (Iwamura H et al. ref. 2001).
Despite the clinical use of synthetic cannabinoids for several years, their precise molecular mechanism of action remains poorly understood (Hinz & Ramer ref. 2022). Most research to date has focused on investigating the hypothesis that these compounds possess anti-tumor properties (Voicu et al. ref. 2023). In this comprehensive systematic review, we aim to address this research gap by methodically examining the mode of action of synthetic cannabinoid ligands as agonists, antagonists, and inverse agonists and their modulation of the CB1R and CB2R. Ultimately, we will investigate the impact of the ligand mode of action of synthetic cannabinoids on tumor growth across various cancer cell lines. This analysis will provide critical insights into the potential anticancer effects of these compounds and the underlying signaling pathways involved.
Methodology
Data sources and search strategy
A comprehensive literature search was conducted across PubMed, Science Direct, Google Scholar, and the Cochrane Database to identify relevant studies published from January 1990 to July ref. 2024. The search strategy employed a combination of Medical Subject Headings (MeSH) terms and keywords related to synthetic cannabinoids and cancer. The search terms included"cancer,""anticancer,""neoplasm,""antineoplasm,""tumor,""antitumor,""synthetic molecules,""agonist,""antagonist,""inverse agonist,""mixed agonist/antagonist,""cannabinoid receptor 1,"and"cannabinoid receptor 2."Boolean operators"AND"and"OR"were used to combine terms effectively (e.g.,"synthetic cannabinoids"AND"anticancer"AND"cannabinoid receptor 1"). Reference lists of pertinent articles were also reviewed to ensure the inclusion of all relevant studies.
Study selection and eligibility criteria
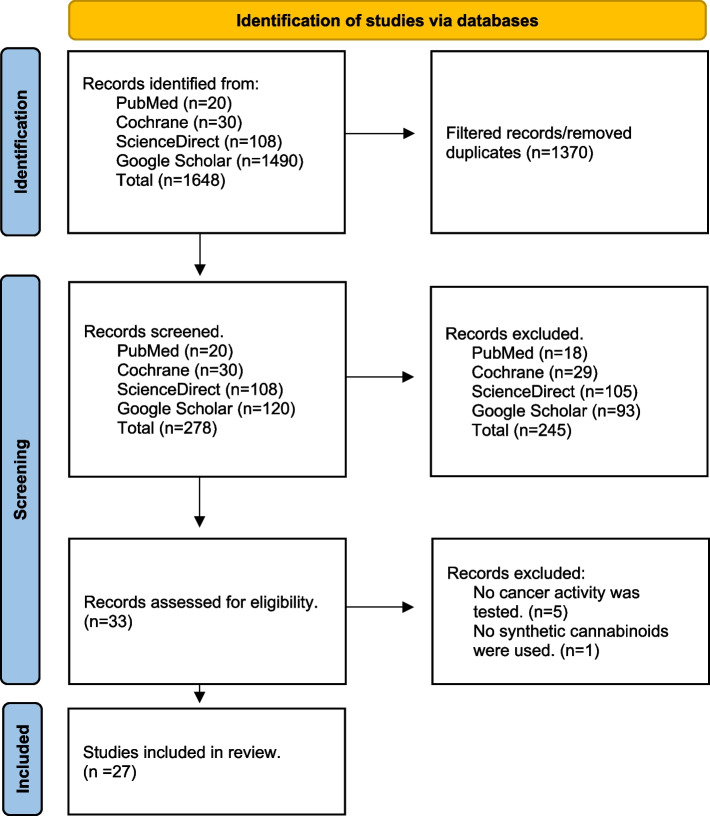
The initial search yielded 287 articles, screened by two independent reviewers using the predefined inclusion and exclusion criteria. To be eligible for inclusion, studies needed to report on the anticancer effects of synthetic cannabinoid receptor ligands, be conducted as in vitro or in vivo experiments, be published between January 1990 and July ref. 2024, be available in full text and published in English, and focus specifically on the mode of action of synthetic cannabinoid ligands and their anticancer activity. Studies were excluded if they were review articles, systematic reviews, unpublished articles, dissertations, commentaries, conference proceedings, case reports, book reviews, opinion articles, or editorials. Additionally, articles not accessible in full text, published in languages other than English, not specifically focused on cancer, or not involving synthetic cannabinoids as defined in the study parameters were excluded. Discrepancies between the reviewers were resolved through discussion or consultation with a third reviewer when necessary. After applying the inclusion and exclusion criteria, four studies were selected for detailed analysis.
Data extraction and quality assessment
Three reviewers independently extracted data using a standardized form. The information extracted included the authorship, publication year, study design, type of synthetic cannabinoid receptor ligand, type of cancer investigated, experimental models used (in vitro and in vivo), key findings on anticancer effects, and details regarding each ligand’s mode of action at cannabinoid receptors.
The quality of the included studies was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) criteria, adapted for preclinical research. Each study was evaluated for risk of bias, directness of evidence, consistency of results, precision, and publication bias. The overall quality of evidence was categorized as high, moderate, low, or very low. Any disagreements in data extraction or quality assessment were reconciled through consensus or consultation with a third reviewer to ensure accuracy and reliability. All studies were evaluated for potential bias by two independent researchers. The Cochrane Risk of Bias (ROB) assessment tool was used to assess the overall risk of bias in the randomized controlled trials included in the review.
Results
After a comprehensive literature search, 287 articles were screened, but only 27 were included in this systematic review, as shown in Fig. 4. These studies primarily investigated the impact of synthetic cannabinoids on tumor progression and examined how their mode of action at cannabinoid receptors contributes to the inhibition of tumor growth.
Synthetic cannabinoids can act as agonists, antagonists, or inverse agonists at CB1 and CB2 receptors, leading to diverse pharmacological effects across cancer cell lines. This section explores the anticancer activity of these compounds, with emphasis on their mode of action at cannabinoid receptors.
Synthetic cannabinoids 1 receptor
CB1R antagonists/inverse agonist
Our comprehensive review of CB1R antagonists and inverse agonists revealed diverse effects across various cancer types. As the CB1R inverse agonists showed their ability to reduce CB1R basal activity and mimic the antagonism mode of action, this section combined these two modes of action acting on CB1R. The findings are organized by cancer type, highlighting the key outcomes and mechanisms observed in each case illustrated in Table 1.
Table 1: Summary of studies investigating CB1R ligands in cancer cell apoptosis, proliferation, and metastasis
| Study | Ligand Used | Cancer Type | Cell Line (s) | Mechanism of Action | Key Outcome |
|---|---|---|---|---|---|
| CB1R Antagonists | |||||
| (Aviello et al. ref. 2012) | SR141716 (Rimonabant) and AM251 | Human colon cancer cell lines | HCT116 and Caco-2 cell lines | Combination between CB1R antagonist and cannabidiol | Antiproliferative effects in vitro via CB1R activation |
| (Preet et al., ref. 2011) | AM251 | Non-small cell lung cancer | A549 and SW-1573 cell lines | Tumour growth and metastasis | Inhibited non-small cell lung cancer cell proliferation, migration, and invasion both in vitro and in vivo |
| (Qamri et al., ref. 2009) | AM 251 | Primary human breast cancer | MDA-MB231, MDA-MB231-luc, and MDA-MB468 cell lines | Validation in vivo for many mouse model systems by reversed the CB1and 2 agonists’ effects to ensure that the cancer reduction is mediated by the effect of CB1 and 2 receptor agonists only | Validation that CB1 and CB2 agonists are responsible of 40% to 50% reduction in tumour development and a 65% to 80% reduction in lung metastasis |
| (Raup-Konsavage et al. ref. 2018) | PTI-1, PTI-2, and NPB-22 | Colorectal cancer | SW480, SW620, HT29, DLD-1, HCT115, LS174, and RKO cell lines | PTI-1and PTI-2: decreased the vitality of all cell lines examined, including HEK293 and CCD841 CoTr (normal cells)NPB-22: No known mechanisms | ↓cancer progression |
| (Fogli et al. ref. 2006) | AM251and SR141716 A | Human pancreatic cancerous cells | MIA PaCa-2 cancer cell line | SR141716 A: No significant impact on cell viabilityAM251: alters the transcription of genes in janus kinase/signal transducers and activators of the transcription signalling network, and interacts synergistically with 5-fluorouracil, a pyrimidine analogue | SR141716 A: No effect on cancer cell linesAM251: trigger apoptosis |
| (Widmer et al. ref. 2008) | AM251 | Glioma cancer cells | U373MG cell linem | Prevent cannabinoid-induced cell death in normal cell lines | No cancer effect on cancerous cell by itself, but it has protective role for normal cell lines |
| (Sarnataro et al. ref. 2006) | SR141716(Rimonabant) | Human breast cancer cell | MDA-MB-231 cells | Antiproliferative activity of SR141716 is mediated by the cannabinoid CB1R activation | ↓the growth of breast cancer and hinder metastatic diffusion |
| (Blázquez et al. ref. 2006) | SR141716 (Rimonabant) | Melanoma cell lines | B16 and A375 | CB1R inhibitions | No significant effect of SR141716 (Rimonabant) on melanoma |
| (K. Gustafsson et al. ref. 2009) | SR141716 A | Mantle cell lymphoma | They study did not specify the cell line | Blockage the CB1-mediated signalling pathway that leads to the activation of the de novo ceramide production pathway, which includes the enzymes CerS3 and CerS6 | Cancerous cell death |
| (Malfitano et al. ref. 2012) | SR141716(Rimonabant) | Fibrosarcoma cell line | Methylcholanthrene-induced fibrosarcoma (Meth-A) cell line | Reduced COX2 expression and induction of the CB1R expression | ↓cancer spread via inducing apoptosis |
| CB1R Agonist | |||||
| (Gurley et al., ref. 2012) | KM-233 | Glioblastoma multiforme | U87MG | modulating protein phosphorylation | Cellular signalling pathways alteration |
Colon cancer
An in vitro study on human colon cancer cell lines (HCT116 and Caco-2) showed that CB1R antagonists, particularly Rimonabant (SR141716) and AM251, may contribute to anticancer properties. The mechanism involves indirect activation of CB1 receptors, resulting in antiproliferative effects (Aviello et al. ref. 2012).
Another study evaluated a synthetic cannabinoid library on seven colorectal cancer (CRC) cell lines: SW480, SW620, HT29, DLD-1, HCT115, LS174, and RKO. The PTI family of compounds decreased the vitality of all cell lines examined, including normal cells (HEK293 and CCD841 CoTr). NPB-22 showed efficacy in only two cell lines (HCT116 and SW480) through an unknown mechanism (Raup-Konsavage et al. ref. 2018).
AM251, a selective CB1R antagonist, demonstrated a protective effect on human colorectal carcinoma Caco-2 cells in vitro. Both CB1 and CB2R antagonists reduced the cytotoxic effects of HU 210 and AEA (S. B. Gustafsson et al., ref. 2008).
Lung cancer
Research on non-small cell lung cancer (A549 and SW-1573 cells) demonstrated that synthetic cannabinoid agents, including CB1/CB2 agonists WIN 55,212–2 and JWH-015, significantly decreased the focal adhesion complex crucial for cancer cell migration. Pretreatment with AM251 (CB1 antagonist) and AM630 (CB2 antagonist) confirmed that the cancer-reducing effect was primarily mediated by cannabinoid receptor agonists. The main outcome was inhibition of cell proliferation, migration, and invasion both in vitro and in vivo (Preet et al., ref. 2011).
Breast cancer
Studies on primary human breast cancers and breast cancer cell lines (MDA-MB231, MDA-MB231-luc, and MDA-MB468) revealed expression of CB1 and CB2 receptors. Synthetic CB2 agonists and CB1/2 agonists reduced cell growth and migration in vitro. In vivo studies in mouse models showed a 40–50% reduction in tumor development and a 65–80% reduction in lung metastasis with CB1 and CB2R agonist treatment. The CB1 antagonist AM251 reversed these effects, confirming the role of CB1 receptors in cancer development reduction (Qamri et al., ref. 2009).
An in vivo study on MDA-MB-231 cells (more invasive than T47D and MCF-7 cells) demonstrated that the CB1R mediates SR141716’s antiproliferative activity. The effect resulted from a G1/S-phase cell cycle arrest rather than apoptosis or necrosis (Sarnataro et al. ref. 2006).
Pancreatic cancer
An in vitro study on human pancreatic cancer cells (MIA PaCa-2) using AM251, SR141716 A, and 5-Fluorouracil showed that AM251 triggered apoptosis and altered gene transcription in the Janus kinase/signal transducers and activators of transcription (JAK/STAT) signaling pathway. AM251 also demonstrated synergistic effects with 5-fluorouracil. However, WIN-55,212–2 (non-selective cannabinoid receptor agonist) and SR141716 A did not significantly impact cell viability at 10 μM over 24–72 h (Fogli et al. ref. 2006).
Glioma
Investigations on U373MG glioma cells revealed that AM251 almost completely inhibited cannabinoid-induced cell death in normal cell lines, suggesting a crucial role for CB1 receptors in cannabinoid-triggered apoptosis (Widmer et al. ref. 2008).
Another study on U87, U373, U118, and A172 human glioma cells showed no significant effect of SR141716a on cancer spread when used as a pretreatment (Recht et al. ref. 2001).
Research on C6 glioma cells demonstrated that blocking both CB1 (SR141716 A) and CB2 (SR144528) receptors was necessary to protect cells from the antiproliferative effects of Δ9-THC (Vaccani et al. ref. 2005).
Melanoma
In vitro studies on melanoma cell lines (B16 and A375) concluded that SR141716 (Rimonabant) had no significant effect on melanoma. The antagonist was primarily used to investigate the impact of cannabinoid receptors on melanoma cell lines (Blázquez et al. ref. 2006).
Lymphoma
Research on Mantle Cell Lymphoma showed that SR141716 A blocks the CB1-mediated signaling pathway, leading to the activation of the de novo ceramide production pathway (involving enzymes CerS3 and CerS6) and resulting in cell death (K. Gustafsson et al. ref. 2009).
Fibrosarcoma
An in vitro study on methylcholanthrene-induced fibrosarcoma (Meth-A) cell line revealed that Rimonabant (SR141716) reduces cancer spread by inducing apoptosis. The mechanism involves modulating cell cycle progression through regulation of p21waf, cyclins E, D1, and NF-κB (Malfitano et al. ref. 2012).
In conclusion, CB1R antagonists demonstrate varied effects across different cancer types, ranging from antiproliferative and pro-apoptotic activities to protective effects in some cases. These findings underscore the complex role of the endocannabinoid system in cancer biology and highlight the need for further research to fully elucidate the potential of CB1R antagonists in cancer treatment.
CB1R agonist
A single synthetic cannabinoid agent that is categorized under CB 1 receptor agonists was studied on human glioblastoma multiforme (GBM) cell line U87MG via in vivo and in vitro settings. Cellular signalling pathways alteration was mediated by KM-233 modulating protein phosphorylation in a time-dependent manner, including MEK, ERK1/2, Akt, BAD, STAT3, and p70S6 K (Gurley et al., ref. 2012). No further studies examined the effect of synthetic cannabinoid agonists on CB1 receptors in cancer development. Table 1 outlines the effects of synthetic cannabinoids from CB1R agonists on cancer cells, involving apoptosis, proliferation, and metastasis.
Synthetic cannabinoids 2 receptor
CB2R antagonist
Breast Cancer
SR144528, a CB2R-specific antagonist, has been extensively used in studies to clarify the role of CB2 receptors in mediating the antiproliferative and antimetastatic effects of synthetic cannabinoids on cancer cells. Another study evaluated the synthetic CB2R agonist JWH-133 in breast cancer. In the study by (Qamri et al. ref. 2009), JWH-133 inhibited the proliferation and migration of breast cancer cell lines. These effects were reversed when SR144528 was applied, confirming the CB2R-dependent nature of the response. In vivo experiments further demonstrated that JWH-133’s reduction in tumour growth and metastasis was negated by SR144528, underscoring its role in blocking CB2R-mediated antitumor effects.
Similarly, (Blázquez et al. ref. 2006) study reinforced SR144528’s ability to block CB2R-mediated actions. It reversed the anti-proliferative and anti-migratory effects of JWH-133 on breast cancer cells, which prominently expressed CB2Rs, such as MDA-MB231 and MDA-MB468. Moreover, MDA-MB231 cells treated with JWH-133 exhibited a 46% reduction in tumor growth, but this was reversed by SR144528. These findings highlight the significance of CB2Rs in regulating the therapeutic effects of cannabinoids and suggest that blocking CB2R can diminish these effects.
Glioma
In (Recht et al. ref. 2001) study on the antitumor effects of ajulemic acid (AJA), another synthetic cannabinoid, CB2R antagonism was crucial in understanding its mechanism of action. AJA demonstrated significant antitumor activity, primarily mediated through CB2Rs. The CB2R antagonist SR144528 reversed these effects, particularly in C6 glioma cells, where co-incubation led to a 63% reduction in AJA’s inhibitory action on tumour growth. This contrasted with THC, which was blocked by both CB1R and CB2R antagonists, further confirming that AJA’s antitumor efficacy is predominantly reliant on CB2 receptors.
Additionally, the study by (Jacobsson et al., 2001) examined the role of CB2R in cancer cell proliferation using JWH-015, a selective CB2R agonist. In C6 glioma cells, JWH-015 significantly inhibited proliferation, an effect that was reversed by SR144528. This reinforced the idea that CB2R activation is crucial for the antiproliferative effects of synthetic cannabinoids and that SR144528 serves as a reliable tool in verifying CB2R dependency in these responses.
Colorectal cancer
In contrast, (Aviello et al. ref. 2012) study on the chemopreventive effects of cannabidiol (CBD) in colorectal cancer cells revealed that CB2R activation was not a primary pathway. In human colorectal carcinoma cell lines Caco-2 and HCT116, the CB2R antagonists SR144528 and AM630 did not significantly alter the antiproliferative effects of CBD. However, the effects were reversed by CB1R antagonists, suggesting that the CB1R, rather than CB2R, plays a more critical role in mediating CBD’s anticancer effects. seen in Table 2.
Table 2: Summary of studies investigating CB2R ligands in cancer cell apoptosis, proliferation, and metastasis
| Study | Ligand Used | Cancer Type | Cell Line (s) | Mechanism of Action | Key Outcome |
|---|---|---|---|---|---|
| CB2R Antagonists | |||||
| (Qamri et al., ref. 2009) | SR144528 | Breast cancer | MDA-MB231, MDA-MB468 cell line | Inhibited CB2 receptor-mediated pathways, blocking antiproliferative and anti-migratory signals | Blocked antiproliferative and anti-migratory effects of JWH-133 |
| (Blázquez et al. ref. 2006) | SR144528 | Breast cancer | MDA-MB231 cell line | Antagonized CB2R signaling, reversing the tumor growth reduction induced by JWH-133 | Reversed the 46% reduction in tumor growth induced by JWH-133 |
| (Recht et al. ref. 2001) | SR144528 | Glioma | C6 Glioma cell line | Suppressed CB2 receptor-mediated antitumor pathways, reversing antitumor pathways of Ajulemic Acid (AJA) | Reversed 63% of Ajulemic Acid’s (AJA) antitumor effects |
| (Jacobsson et al., 2001) | SR144528 | Glioma | C6 Glioma cell line | Reversed CB2 receptor-mediated antiproliferative effects induced by JWH-015 | Reversed JWH-015’s antiproliferative effects |
| (Aviello et al. ref. 2012) | SR144528 | Colorectal Carcinoma | Caco-2 and HCT116 | CB2R plays a minimal role in CBD’s anticancer effects, with its blockade showing negligible impact | CB2R is less critical in CBD’s anticancer activity, limiting its therapeutic relevance |
CB2R agonist
Breast cancer
A study (Qamri et al., ref. 2009) reveals that JWH-133 inhibits cell proliferation and migration in breast cancer cell lines (MDA-MB231 and MDA-MB468) through CB2R-dependent pathways. Silencing CB2R using CB2R-specific siRNA reverses these effects, demonstrating the receptor’s critical role. In vivo, JWH-133 reduces tumor growth by 46% and lung metastasis by 76%. JWH-133 also suppresses key pathways linked to cancer progression, such as cyclooxygenase-2 (COX-2) and prostaglandin E2 (PGE2) production, while inhibiting Cdc42 and decreasing the nuclear expression of c-Fos and c-Jun. Furthermore, JWH-133 induces apoptosis by arresting the cell cycle in the G0/G1 phase, evidenced by increased apoptotic markers, and reduces cell proliferation and angiogenesis, as shown by Ki67 and CD31 staining in tumor samples from treated mice.
This in vitro study (Chutoe et al. ref. 2021) investigated the effect of cannabinoid receptor agonists on breast cancer, specifically focusing on the CB2R agonist GW405833. As described in Table 3, GW405833 significantly reduced the viability of MDA-MB-231 breast cancer cells in a concentration-dependent manner, with an IC50 of 23.46 µM. Additionally, GW405833 effectively inhibited cancer cell migration, with a concentration of 15 µM significantly decreasing migration after 24 h of treatment. The study also examined the interaction between breast cancer cells and bone cells using UMR-106 osteoblast-like cells. Pre-treatment of MDA-MB-231 cells with GW405833 before collecting conditioned media significantly improved UMR-106 cell viability by 18.5%, demonstrating the protective role of CB2R activation in reducing the harmful effects of breast cancer cells on bone cells.
Table 3: Summary of studies investigating CB2R ligands in cancer cell apoptosis, proliferation, and metastasis
| CB2R Agonists | |||||
|---|---|---|---|---|---|
| (Qamri et al. ref. 2009) | JWH-133 | Breast cancer | MDA-MB231, MDA-MB468 cell lines | Inhibited COX-2 and PGE2 production, induced apoptosis, G0/G1 cell arrest | Reduce tumor growth by 46% |
| (Chutoe et al. ref. 2021) | GW405833 | Breast Cancer, Bone Metastasis | MDA-MB231, UMR-106 cell lines | Reduced cell viability, induced apoptosis | Suppressed cell proliferation and migration, osteoblast viability by 18.5% |
| (Blázquez et al. ref. 2008) | JWH-133 | Glioblastoma | T98G, U87MG cell lines | Activated mitochondrial apoptotic and autophagic pathways | Inhibited proliferation, induced apoptosis |
| (Preet et al., ref. 2011) | JWH-015 | Non-small cell lung cancer | A549, SW-1573 cell lines | Suppressed AKT signaling, reduced MMP-9 activity | Inhibited migration, metastasis |
| (Fogli et al. ref. 2006) | JWH-015 | Pancreatic cancer | MIA PaCa-2 cell lines | Activated caspase 3/7, inhibited JAK/STAT, MAPK signaling | Induced apoptosis, proliferation |
| (Brandi et al. ref. 2013) | GW405833 | Pancreatic adenocarcinoma | Panc1 cell line | Disrupted Warburg effect, induced autophagy | Inhibited cell proliferation, induced cell death |
| (Blázquez et al. ref. 2006) | JWH-133 | Melanoma | B16, A375, MelJuso, melan-c, and Hermes 2b | Inhibited AKT phosphorylation, causing G1-S cell cycle arrest and apoptosis; reduced tumor vascularization | Significant in melanoma cell proliferation, migration, metastasis, tumor growth, and metastasis to lung and liver |
| (Duntsch et al. ref. 2006) | KM-233 | Glioma | U87MG | Potent cytotoxic effects with IC50 of 1.429 µM; penetrates the blood–brain barrier; minimal toxicity to healthy tissue | Suppressed tumor burden by 50% in SCID mice; superior efficacy compared to BCNU; promising glioma treatment |
| (Blázquez et al. ref. 2008) | JWH-133 | Glioma | C6.9, C6.4, U87 MG and primary glioma cells | Downregulated TIMP-1 expression via the ceramide pathway in cannabinoid-sensitive cells (C6.9, U87 MG, and primary glioma cells). No effect in C6.4 (rat glioma, cannabinoid-resistant)) | Reduce tumor growth, invasiveness, and migration in cannabinoid-sensitive cells; no significant effects in cannabinoid-resistant cells (C6.4) |
| (Jacobsson et al., 2001) | JWH-015 | Glioma | Rat C6 glioma cells | Inhibited cAMP levels, suppressed PKA signaling, induced G0/G1 cell cycle arrest; effects reversed by CB2R antagonist SR144528 | Significant inhibition of glioma cell proliferation (IC50 = 3.2 µM); confirmed CB2R-mediated antiproliferative effects |
| FG158a, | Neuroblastoma | SH-SY5Y, SK-N-BE | Selective CB2R activation (Ki = 21 nM); no CB1R binding; effect reversed by SR144528; downregulated ERK1/2 expression | Reduced cell viability in both cell lines; IC50 = 11.8 µM in SH-SY5Y cells | |
Glioblastoma
An in vitro experimental study (Blázquez et al. ref. 2008) investigates how the CB2R agonist JWH133 induces cell death in human glioblastoma cells by activating apoptotic and autophagic pathways. In CB2R-expressing glioblastoma cells T98G and U87MG, JWH133 triggers the mitochondrial apoptotic pathway, leading to mitochondrial membrane potential dissipation and the release of pro-apoptotic factors like cytochrome c, which activates caspases 9 and 3, driving apoptosis. Furthermore, JWH133 induces autophagy, as evidenced by the increased formation of acidic vesicular organelles (AVOs) and the conversion of LC3-I to LC3-II. Silencing autophagy-related genes Atg5 and Atg7 enhances apoptosis, suggesting that autophagy may act as a protective mechanism in CB2R-expressing glioblastoma cells. As highlighted in Table 3, these effects are receptor-specific, with CB2 deficient cells (LN229) showing no response and CB2R antagonists reversing the effects. This emphasizes the therapeutic potential of targeting CB2R in glioblastoma treatment.
Non-small cell lung cancer
Extending the focus to non-small cell lung cancer (NSCLC), (Preet et al., ref. 2011) explore CB2R activation using the agonists JWH-015 and JWH-133. In NSCLC cell lines A549 and SW-1573, JWH-015 inhibits cell migration and invasion primarily by suppressing AKT signalling and reducing AKT phosphorylation, critical for cancer cell survival and movement. Additionally, JWH-015 decreases matrix metalloproteinase 9 (MMP-9) expression and activity, a key enzyme involved in metastasis, as confirmed by ELISA and zymography assays. In vivo, JWH-133 reduces tumor growth by 70% and lung metastasis by 50% in SCID mice implanted with A549 tumors. These effects are reversed by the CB2R antagonist SR144528, confirming a CB2R-dependent mechanism. Moreover, CB2R activation disrupts focal adhesion formation, reducing vinculin and actin stress fibers, which are crucial for cell migration.
Pancreatic cancer
In pancreatic cancer, CB2R activation also shows promise. Research by (Fogli et al. ref. 2006) on MIA PaCa-2 pancreatic cancer cells highlights the cytotoxic effects of the CB2R agonist JWH-015, which induces apoptosis primarily through caspase 3/7 activation. JWH-015 inhibits cell proliferation in a time- and dose-dependent manner while downregulating the JAK/STAT and MAPK signaling pathways, which are crucial for cancer survival. As summarized in Table 3, JWH-015’s ability to reduce cytosolic histone-associated DNA fragments confirms apoptotic cell death, further emphasizing its therapeutic potential. Similarly, another study (Brandi et al. ref. 2013) explores the effects of the CB2R-specific agonist GW405833 (GW) on pancreatic adenocarcinoma cells. Treating Panc1 cells with GW significantly inhibits cell proliferation and induces autophagic cell death by disrupting key cancer-promoting pathways. GW downregulates proteins involved in energy metabolism, such as pyruvate kinase M2 (PKM2) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), thereby disrupting the Warburg effect. Additionally, GW upregulates mitochondrial matrix protein p32, promoting autophagic cell death by destabilizing mitochondrial membrane potential. GW also leads to the phosphorylation of keratin 18 (KRT18), contributing to cytoskeletal reorganization during cell death, underscoring CB2R activation as a potential therapy for pancreatic cancer.
Melanoma
CB2R activation has also shown efficacy in melanoma models. A study by (Blázquez et al. ref. 2006) focusing on the CB2R agonist JWH-133 reports that both human and mouse melanoma cells express CB1Rs and CB2Rs. JWH-133 significantly inhibits melanoma cell proliferation, migration, and metastasis by inhibiting AKT phosphorylation, leading to cell cycle arrest at the G1-S transition and inducing apoptosis. In vivo, JWH-133 reduces tumor growth and lung and liver metastasis in melanoma models, reversing the effects of CB2R antagonism and confirming a CB2R-dependent mechanism. The study also shows that JWH-133 reduces tumor vascularization, as evidenced by reduced CD31 staining and increased TUNEL staining.
Glioma
Further emphasizing the promise of CB2R-targeted therapies in glioma, an in vitro and in vivo study by (Duntsch et al. ref. 2006) investigates the novel CB2R agonist KM-233. As highlighted in Table 3, KM-233 exhibits potent cytotoxic effects in U87MG glioma cells, with an IC50 value of 1.429 µM, indicating strong efficacy at low concentrations. In vivo, KM-233 significantly reduces tumor burden by 50% in SCID mice, outperforming traditional chemotherapy agents like BCNU. KM-233 penetrates the blood–brain barrier effectively and shows minimal toxicity to healthy brain tissue, making it a strong candidate for glioma treatment.
Further investigation into glioma models reveals the potential for CB2R activation in treating gliomas. A study by (Blázquez et al. ref. 2008) demonstrates that JWH-133 reduces glioma tumor growth and downregulates tissue inhibitor of metalloproteinases-1 (TIMP-1), a protein linked to tumor invasiveness and poor prognosis. JWH-133 reduces TIMP-1 expression via the sphingolipid ceramide pathway, as co-treatment with the ceramide synthesis inhibitor fumonisin B1 reverses the effects. The downregulation of TIMP-1 reduces glioma cell migration, which is associated with decreased tumor proliferation and increased apoptosis.
Finally, in rat C6 glioma cells, a study by (Jacobsson et al., 2001) examines the antiproliferative effects of endogenous and synthetic cannabinoids. JWH-015 significantly inhibits glioma cell proliferation through CB2R activation, with an IC50 of 3.2 µM. This effect is reversed by the CB2R-specific antagonist SR144528, confirming the role of CB2R in mediating these effects. JWH-015 reduces cyclic AMP (cAMP) levels, suppressing protein kinase A (PKA) signaling, leading to G0/G1 cell cycle arrest, and preventing further cell proliferation. Other cannabinoids, such AEA and 2-AG, showed partial reliance on vanilloid receptors, as their effects were attenuated by the vanilloid receptor antagonist capsazepine.
Neuroblastoma
A study by (Gado et al. ref. 2022) investigated the anticancer effects of newly synthesized selective CB2R agonists against neuroblastoma cell lines. The research focused on the design and synthesis of compounds based on the 1,8-naphthyridin-2(1H)-one-3-carboxamide scaffold, introducing three ligands, FG158a, FG160a, and FG161a. Structural modifications included bromine, chlorine, or azide substitutions to examine their influence on receptor activity and selectivity. All three compounds exhibited high affinity for CB2R, with Ki values ranging from 16.5 to 45 nM, and no measurable affinity for CB1R (Ki > 10,000 nM), indicating CB2R selectivity.
In vitro, testing on SH-SY5Y and SK-N-BE neuroblastoma cell lines revealed a time and concentration-dependent decrease in cell viability for all compounds. FG158a showed an IC50 of 11.8 µM, FG160a 13.2 µM, and FG161a 27.53 µM in SH-SY5Y cells. Co-treatment with the CB2R antagonist SR144528 partially reversed the cytotoxic effects, supporting a CB2R-mediated mechanism. Additionally, FG158a was found to modulate ERK1/2 expression, suggesting possible involvement of the MAPK pathway in its activity. These findings support the relevance of CB2R activation in neuroblastoma and highlight the need for further investigation into these compounds’therapeutic potential.
CB2R inverse agonist
Pancreatic cancer
AM630 (iodopravadoline), a novel aminoalkylindole, is primarily recognized as an inverse agonist at CB2R in many systems, meaning it not only blocks the receptor but also reduces its basal activity, inducing the opposite effect of typical CB2R activation (Ross et al. ref. 1999). This classification was established (Ross et al. ref. 1999), who demonstrated that AM630 suppresses the constitutive activity of CB2R in certain experimental models. However, despite its inverse agonist properties, AM630 is frequently described as a selective CB2R antagonist in numerous studies, likely because it effectively blocks the actions of CB2R agonists in these settings.
This in vitro study (Fogli et al. ref. 2006) investigates the effects of cannabinoid derivatives on pancreatic cancer MIA PaCa-2 cells, focusing on AM630, a selective CB2R antagonist. AM630 demonstrated moderate cytotoxic effects, with an IC50 of 19.2 ± 1.7 µM after 72 h. Although AM251 was the primary focus, AM630 provided key insights into receptor-independent mechanisms of cannabinoid-induced cytotoxicity. The inhibition of cell growth by AM630 was independent of CB2R activation, as pancreatic cancer cells lacked CB2R expression, suggesting that its cytotoxicity may arise from broader cellular disruptions rather than CB2R antagonism.
Although this study did not fully detail specific pathways involving AM630, cannabinoid treatments were shown to influence critical signaling networks like JAK/STAT and MAPK, which regulate cell proliferation and survival. AM630’s impact on tumor growth may be attributed to interference with these pathways, which contribute to cell death through receptor-independent mechanisms.
Melanoma
In an in vitro and in vivo study (Blázquez et al. ref. 2006), summarized in Table 5, examined the role of cannabinoid receptor antagonists and agonists in melanoma treatment, emphasizing AM630. Here, AM630 effectively blocked the antiproliferative effects of cannabinoid receptor agonists, such as WIN-55,212–2, in melanoma cell lines B16 and A375. This confirmed that cannabinoid inhibition of melanoma cell growth was CB2R dependent. AM630 also reversed cannabinoid-induced apoptosis and angiogenesis suppression, demonstrating its ability to counteract the pathways through which cannabinoids slow melanoma progression. Furthermore, AM630 restored phosphorylation of Akt, a survival protein essential for cell cycle progression and melanoma cell viability, further emphasizing its modulation of CB2R-dependent mechanisms.
Table 4: Summary of studies investigating CB2R ligands in cancer cell apoptosis, proliferation, and metastasis
| CB2R Inverse Agonists | |||||
|---|---|---|---|---|---|
| (Fogli et al. ref. 2006) | AM630 | Pancreatic Cancer | MIA PaCa-2 cell line | Cytotoxic effects independent of CB2R activation | Moderate cytotoxicity (IC50 = 19.2 µM), suggesting receptor-independent mechanisms |
| (Blázquez et al. ref. 2006) | AM630 | Melanoma | B16, A375 cell lines | Blocked CB2R agonists’antiproliferative effects, restored Akt signaling | Reversed apoptosis and angiogenesis suppression, demonstrating CB2R-dependent mechanisms |
| (S. B. Gustafsson et al., ref. 2008) | AM630 | Colorectal Carcinoma | Caco-2 cell line | Did not block cannabinoid-induced cytotoxicity, suggesting CB2R independence | Cytotoxicity linked to oxidative stress pathways, further supporting receptor-independent mechanisms |
Colorectal cancer
A different trend emerged in an in vitro study (S. B. Gustafsson et al., ref. 2008) on the cytotoxic effects of cannabinoids on human colorectal carcinoma cells. Although a CB2R antagonist, AM630 did not mitigate the cytotoxic effects of cannabinoids like HU 210 and AEA in colorectal carcinoma Caco-2 cells. This suggests that the observed cytotoxicity was independent of CB2R activation, as AM630 failed to block cannabinoid-induced reductions in cell viability.
The study on colorectal carcinoma by (S. B. Gustafsson et al., ref. 2008) explored alternative mechanisms, revealing that oxidative stress played a key role in cannabinoid-induced cytotoxicity. Antioxidants like α-tocopherol and the nitric oxide synthase inhibitor L-NAME successfully attenuated the cytotoxic effects, confirming the involvement of oxidative stress pathways. AM630’s inability to block these effects further supported the idea that cannabinoids exert their cytotoxic actions through receptor-independent mechanisms, particularly involving oxidative stress shown in Table 5.
Synthetic cannabinoids mixed receptor
Cannabinoids mixed receptors (CB1/2R) agonist
Glioma
Research explored the anticancer actions of synthetic cannabinoids, specifically CP 55,940, WIN 55,212–2, and HU-210, which act as agonists on both CB1 and CB2 receptors. CP 55,940, a synthetic cannabinoid receptor agonist, potently affects CB1 and CB2 receptors. In glioma C6 cells from rats and glioblastoma U373 cells from humans, CP 55,940 activates these receptors, leading to apoptosis and significantly decreasing cell viability (Ortega et al. ref. 2015). CP 55,940 selectively induces cell death in tumor cells through apoptotic mechanisms, sparing primary astrocytes. Another study highlighted its antiproliferative effects in C6 glioma cells, with an IC50 value of 5.6 µM, linked to oxidative stress and ceramide accumulation (Jacobsson et al., 2001). Similarly, HU-210 and CP 55,940 induce cell death in human U373MG glioma cells through CB1R activation but only at high concentrations, raising concerns about their therapeutic potential (Widmer et al. ref. 2008).
WIN 55,212–2, a non-selective cannabinoid receptor agonist, also demonstrates efficacy in glioblastoma models. It induces caspase-independent apoptosis by modulating proteins such as p53 and cathepsin D while reducing HSP70 expression. WIN 55,212–2’s cytotoxic effects suggest its potential as a therapeutic agent against aggressive glioblastomas (Silva et al. ref. 2019).
Gastric Cancer
In human gastric adenocarcinoma cell lines (AGS), CP 55,940 induces significant morphological changes, reduces cell viability, and promotes apoptosis through key signaling pathways (Ortega et al. ref. 2016). WIN 55,212–2 also inhibits cell proliferation and invasion in gastric cancer through CB1R activation, inducing apoptosis and reducing phospho-AKT expression (Xian et al. ref. 2010).
Breast Cancer
WIN 55,212–2 inhibits cell proliferation and migration in breast cancer by modulating pathways related to cyclooxygenase-2 (COX-2) and prostaglandin E2 (PGE2). It reduces tumor growth and metastasis, with effects reversed by CB1 and CB2 antagonists, confirming the receptor’s role (Qamri et al. ref. 2009). HU-210, however, does not modulate estrogen signaling in breast cancer cells but shows potential therapeutic effects (Okazaki et al. ref. 2016).
Colorectal Cancer
HU-210 exerts potent antiproliferative and cytotoxic effects on human colorectal carcinoma Caco-2 cells. Its effects are receptor-independent, as CB1 and CB2 antagonists do not diminish its efficacy. Oxidative stress appears to mediate its cytotoxic action (Widmer et al. ref. 2008).
Non-Small Cell Lung Cancer (NSCLC)
WIN 55,212–2 inhibits proliferation and migration in NSCLC cell lines (A549 and SW-1573) by disrupting EGF-induced signaling pathways and attenuating AKT phosphorylation. It effectively reduces tumor growth and increases apoptosis in treated cells (Preet et al., ref. 2011).
Melanoma
WIN 55,212–2 disrupts the cell cycle at the G1-S transition, promoting apoptosis while sparing normal melanocytes. It inhibits Akt, a prosurvival protein, and reduces retinoblastoma protein phosphorylation, leading to apoptosis (Blázquez et al. ref. 2006).
Mantle Cell Lymphoma (MCL)
WIN 55,212–2 induces ceramide synthase expression (CerS3 and CerS6), enhancing ceramide accumulation through CB1R activation. This promotes apoptosis and reduces cell viability in MCL cell lines (K. Gustafsson et al. ref. 2009).
Prostate Cancer
WIN 55,212–2 exhibits cytotoxic effects in prostate cancer cell lines by modulating cannabinoid receptor pathways, emphasizing its therapeutic potential (Silva et al. ref. 2019).
Pancreatic Cancer
WIN 55,212–2 shows limited efficacy in MIA PaCa-2 pancreatic cancer cells at 10 µM over 24–72 h, suggesting receptor-independent mechanisms (Fogli et al. ref. 2006).
Ewing Sarcoma
WIN-55,212–2 has been identified as a potential target for anticancer drug development in Ewing sarcoma cell lines, specifically TC-71 and A-673. This compound confirmed the presence of CB1R and CB2R mRNA in TC-71 cells, revealing atypical properties of the cannabinoid receptors. Receptor interaction was detected exclusively with the non-selective CB1/CB2R radioligand WIN-55,212–2, not with CP-55,940. This interaction occurred at a single site with high affinity, indicating that the most effective cannabinoids also exhibited a strong affinity for these atypical receptors. Unique signaling profiles were noted, where only cannabinoids with high affinity reduced cell viability and induced cytotoxicity over time. pharmacological profiles and cytotoxic effects were observed in the A-673 Ewing sarcoma cell line (Shoeib et al. ref. 2021).
Multiple myeloma
WIN 55,212–2 selectively induces apoptosis in multiple myeloma cell lines via the CB2 receptor, activating caspases and altering Bcl-2 family protein expression, while normal cells remain unaffected. These findings underscore its potential as a therapeutic agent across different cancer types (Barbado et al. ref. 2017).
Overall, synthetic cannabinoids acting on CB1 and CB2 receptors demonstrate significant anticancer activity across various cancer types. Their effects, as summarized in Table 4, include reduced tumor growth, inhibition of proliferation and migration, and increased apoptosis. These findings highlight the therapeutic potential of mixed receptor agonists, though their efficacy and mechanisms vary depending on the cancer type and specific cannabinoids used.
Table 5: Summary of studies investigating CB1/CB2R Agonists in cancer cell apoptosis, proliferation, and metastasis
| Study | Agonist Used | Cancer Type | Cell Line | Mechanism of Action | Key Outcome |
|---|---|---|---|---|---|
| (Ortega et al. ref. 2015) | CP 55,940 | Glioma glioblastoma | C6 andU373 cell lines | Activation of Cannabinoid ReceptorsInduction of ApoptosisCell Morphological Changes | Cell Viability↓Anti-proliferative Effects↑Differential Sensitivity |
| (Widmer et al. ref. 2008) | CP 55,940 | Glioma | U373MG cell line | Activateapoptosis through the activation of downstream effectors, including caspases, which are crucial for programmed cell death | Induces cell death at high concentrations |
| (Widmer et al. ref. 2008) | HU-210 | Glioma | U373MG cell line | Induction of Apoptosisactivates the CB1R in U373MG human glioma cellsactivates ERK and c-Jun NH2-terminal kinase (JNK) | Apoptosis only at high concentrationsCell death, predominantly mediated by CB1R activation |
| (Silva et al. ref. 2019) | WIN 55,212–2 | Glioblastoma | GAMGU251WI-38NHAcell lines | Induction of ApoptosisRegulation of Key ProteinsInhibition of Cell MigrationAnti-Angiogenic Effects | Selective CytotoxicityTumor Growth ↓Inhibition of Matrix Metalloproteinases (MMPs)Clonogenicity ↓ |
| (Ortega et al. ref. 2016) | CP 55,940 | Gastric cancer | AGS cell line | Activates specific signaling pathways associated with apoptosis, involving the regulation of the endocannabinoid system and modulation of kinasesinduces DNA laddering | Changes in cell morphology ↓ cell viabilityApoptotic Induction |
| (Xian et al. ref. 2010) | WIN 55,212–2 | Gastric Cancer | AGSMKN-1 cell lines | Inhibition of ProliferationInduction of ApoptosisDown-Regulation of Phospho-AKTInhibition of Invasion | In percentages of surviving↓ cellsApoptotic Cell DeathIn the invasion index of↓ gastric cancer cells |
| (Qamri et al., ref. 2009) | WIN 55,212–2 | Breast cancer | MDA-MB231, MDA- MB231-luc, and MDA-MB468 cell lines | Inhibition of Cell ProliferationInhibition of MigrationModulation of Signaling Pathways | Tumor Growth ↓Metastasis Inhibition |
| (Okazaki et al. ref. 2016) | HU-210 | Breast cancer | MCF-7 cell line | Does not modulate estrogen receptor (ER) signaling in MCF-7 human breast cancer cellsaffects cell morphology and viability in a concentration-dependent manner | No Anti-Estrogenic ActivityLacks the endocrine-disrupting effects |
| (Widmer et al. ref. 2008) | HU-210 | Colorectal carcinoma | Caco-2 cell line | The cytotoxic effects were suggested to involve oxidative stresssynergistic cytotoxic effects when combined with the chemotherapeutic agent 5-fluorouracil | Cytotoxic EffectsOxidative stress |
| (Preet et al., ref. 2011) | WIN 55,212–2 | Non–small cell lung cancer | A549 and SW-1573 cell lines | Inhibition of Cell ProliferationReduced Migration and InvasionInhibition of AKT PhosphorylationDecreased MMP-9 Activity | Inhibition of Tumor GrowthIn Lung Metastasis↓Apoptosis↑ |
| (Blázquez et al. ref. 2006) | WIN 55,212–2 | Melanoma | B16A375 MelJuso | Inhibition of Cell GrowthCell Cycle ArrestInduction of Apoptosis | Tumor Growth↓Metastatic Spread ↓Apoptosis↑ |
| (K. Gustafsson et al. ref. 2009) | WIN 55,212–2 | Mantle cell lymphoma | Rec-1 mantle cell lymphoma cell lines | Activation of CB1 ReceptorsInduction of Ceramide SynthasesCeramide Accumulation | Apoptosis ↑Ceramide Species Induction |
| (Silva et al. ref. 2019) | WIN 55,212–2 | Prostate cancer cells | GAMGU251WI-38NHAcell lines | Induction of ApoptosisRegulation of Key ProteinsInhibition of Cell MigrationAnti-Angiogenic Effects | Selective CytotoxicityTumor Growth ↓Inhibition of Matrix Metalloproteinases (MMPs)Clonogenicity ↓ |
| (Fogli et al. ref. 2006) | WIN 55,212–2 | Pancreatic cancer | MIA PaCa-2 cell line | cannabinoid-mediated cytotoxic effects | Did not significantly affect cell viability |
| (Shoeib et al. ref. 2021) | WIN 55,212–2 | Ewing sarcoma | TC-71 andA-673 cell lines | G-Protein ActivationDistinct Signaling Profiles | CytotoxicityCell Viability↓The cannabinoid receptors expressed in EWS cells exhibit atypical properties compared to canonical cannabinoid receptors, resulting in unique functional profiles |
| (Shoeib et al. ref. 2021) | CP 55,940 | Ewing sarcoma | TC-71 andA-673 cell lines | inhibition of adenylyl cyclase activity by CB1R and CB2R | ↓cell viability and induce↓ cytotoxicity in a time-dependent manner |
| (Barbado et al. ref. 2017) | WIN 55,212–2 | Multiple myeloma | U266RPMI8226MM1.SU266-LR7RPMI8226-LR5MM1.R cell lines | Induction of ApoptosisCaspase ActivationAlteration of Bcl-2 Family ProteinsCeramide AccumulationModulation of Signaling Pathways | Tumor Growth↓Metastatic Spread↓Apoptosis↑ |
Cannabinoids mixed receptors (cb1/2r) inverse agonist and antagonist
There are currently no studies available that specifically examine agents functioning as mixed antagonists or inverse agonists for cannabinoid receptors CB1 and CB2.
Discussion
This systematic review examines the mode of action of synthetic cannabinoids and their impact on the cannabinoid receptors. Ultimately, discovering the mechanisms by which these synthetic agents could modulate the cancer progression or reduction. Cannabis receptors are presented in the cancerous cells in various percentages, enabling these receptors to be targeted as possible therapeutic agents (De Jesús et al., 2010) (X. Xu et al., 2006). Moreover, these receptors demonstrated noticeable cancerous cells modulating effect via multiple mechanisms such as enhancing apoptosis, reducing the angiogenesis, or lowering the cancer metastasis (Pennant & Hinton, 2023). All these proposed mechanisms modulating cancer cells using synthetic cannabinoid agents are evaluated in this systematic review, providing a comprehensive overview of the available synthetic cannabinoids and their potential therapeutic effects. Furthermore, investigate how the mode of action modulates the synthetic cannabinoids effects.
Synthetic cannabinoids 1 receptor
The findings indicate that CB1R antagonists are used in conjunction with CB2R agonists to enhance the anticancer effects of CB2R or to highlight that the anticancer activity is primarily mediated by CB2R agonists. Research on CB1R agonists is quite limited, with only one study focusing on their effects on cancerous cells (Ellert-Miklaszewska, ref. 2021). This study investigated the human glioblastoma multiforme (GBM) cell line U87MG in both in vivo and in vitro settings, showing a reduction in tumor growth (Gurley et al., ref. 2012). Therefore, further research is needed to confirm the role of CB1R agonists in cancer therapy. Additionally, there is a lack of studies examining the effectiveness of CB1R inverse agonists in cancer modulation. As a result, more investigations are necessary to explore the role of these synthetic cannabinoids and their interactions with CB1R, including the mechanisms of agonists and inverse agonists, to enhance our understanding of their potential applications.
Synthetic Cannabinoids 2 Receptor
These findings suggest that CB2R agonists are the main modulators of cancer cell reduction among various cancerous cell lines with multiple proposed mechanisms. Furthermore, CB2R could be a promising target for treating many cancer types via agonistic activity. One example of this agonistic activity on CB2R is JWH-133, which is a selective agonist for CB2R. This agent inhibits the growth and survival of cancerous cells (S. Xu et al., ref. 2019). Meanwhile, CB1R antagonists are involved in various studies in combination with CB2R agonists either to validate the effect of CB2R agonists by blocking the CB1R or to potentiate the CB2R agonist function to reduce tumour growth. The role of the CR1R antagonist emphasizes the vital role of CB2R agonists.
For CB2R antagonists, studies focus on its role in blocking the cancer-fighting effects of cannabinoids that target the CB2R. For example, SR144528 was found to reverse the effects of JWH-133 in breast cancer research, which normally slows down cancer cell growth and migration. By applying SR144528, researchers could confirm that these effects depended on CB2 receptors. Other research papers studied the same concept with several CB2R antagonists. On the other hand, studies involving inverse agonist acts on CB2R showed a moderate ability to kill cancerous cells depending on the cancer type.
Furthermore, there are many mechanisms by which CB2R inverse agonists act on cancerous cells, including blocking pathways through which cancer cells can survive. Another mechanism unrelated to CB2R is shown in the results. All in all, antagonist acts on CB2R were mainly utilized to validate the impact of CB2R receptor cancer cell-mediated reduction. In contrast, the inverse agonist showed possible minor effects on cancer cell development and growth.
Synthetic cannabinoids mixed receptor
CBRs agonists, such as CP 55,940, WIN 55,212–2, and HU-210, demonstrate potential as anticancer agents by activating CB1 and CB2 receptors. CP 55,940 has shown strong efficacy in inducing apoptosis and decreasing cell viability in glioma and gastric cancer cells through the activation of these receptors and modulation of key signaling pathways. Consequently, it effectively alters cell density, highlighting its therapeutic potential for glial tumors. Further studies are necessary to elucidate the mechanisms of action for mixed CBR antagonists and inverse agonists. While some of these compounds show promising anticancer effects, more research is needed to fully understand their mechanisms of action.
Therapeutic implications
These findings highlight the potential of synthetic cannabinoids as promising anticancer agents, particularly due to their ability to target specific pathways selectively. This selectivity may allow for the development of treatments that avoid the adverse effects commonly associated with traditional chemotherapy by sparing healthy cells. Moreover, synthetic cannabinoids offer the potential to overcome resistance mechanisms seen in some cancer therapies by acting through unique pathways. These include inducing apoptosis specifically in cancer cells via CB2R activation, inhibiting cell proliferation through pathways like PI3 K/AKT and MAPK, reducing angiogenesis to limit tumor growth, modulating the tumor microenvironment to enhance immune detection, and lowering inflammation associated with cancer progression. As our understanding of cannabinoid receptor signaling grows, it opens opportunities for tailored treatments based on individual tumor characteristics, potentially leading to more personalized and effective cancer therapies.
Limitations and future directions
One significant limitation of this systematic review is the relatively small sample size and narrow focus of the included studies, which may impact the generalizability of these findings across various cancer types and patient populations. Additionally, inconsistencies in methodologies, such as variations in cannabinoid dosages, types, and treatment durations, pose challenges for direct comparisons and may undermine the reliability of the results. Furthermore, as most of the studies were conducted in vitro, there are inherent limitations in applying these findings to in vivo environments, where the responses of cell lines may not fully reflect the complex interactions present in living organisms. The lack of long-term studies also restricts our understanding of the sustained efficacy and potential adverse effects associated with synthetic cannabinoids.Moreover, many novel modes of action as allosteric modulation and bitopic ligands, have not been fully understood in the context of cancer, which limits the systematic review scope to the agonist, antagonist, and inverse agonist as modes of action. These limitations emphasize the need for more rigorously designed research across diverse models and populations to gain a clearer understanding of the therapeutic potential and safety of synthetic cannabinoids in cancer treatment.
Conclusion
The current systematic review introduces the anti-tumor mechanisms of synthetic cannabinoid receptors’ligands and their underlying mechanism of action. CB1R agonist illustrated a higher possibility of cellular signalling pathways alteration for cancer pathways, providing an optional cancer treatment. However, the CB1R antagonist and inverse agonist were utilized as confirmatory to CB2R agonistic activity.
Moreover, the research on CB2R antagonists and agonists highlighted their crucial roles in mediating cancer models’antiproliferative, antimetastatic, and cytotoxic effects. CB2R-specific antagonists reverse the anticancer effects of CB2R agonists, underscoring CB2R involvement in tumour growth inhibition and metastasis. Studies across various cancer types, including breast, glioblastoma, non-small cell lung, and pancreatic cancer, reveal that CB2R activation via agonists triggers apoptosis, autophagy, and significant reductions in cell proliferation, migration, and angiogenesis. In contrast, some studies that revealed the role of CB2R antagonists showed mixed results, often blocking the CB2R-mediated effects but also exhibiting cytotoxicity in a receptor-independent manner in certain cancers.
Synthetic cannabinoids show significant potential as therapeutic agents by targeting both CB1 and CB2 as receptor agonists to reduce tumour growth and promote apoptosis across various cancers. Their diverse mechanisms, including modulation of signalling pathways and oxidative stress, underline their promise in treatment. However, the absence of research on these receptors’ mixed antagonists or inverse agonists highlights a critical gap, suggesting a valuable direction for future studies to enhance our understanding of cannabinoid signalling in cancer therapy.
References
- Ahmed M, Alsuhaymi S, Abdulrhman O, Faqih G, Alalyani MA, Saeed M, Alkhodidi S, Alfahmi HH, Ali M, Munshi A, Gale T, Alsahly S, Alzahrani AS, Ali A, Fateh A, Alshareq A, Alfahmi H, Alazzmi M, Alshareq WA. Epidemiology of cancer in Saudi Arabia: a systematic review 2024. J Int Crisis Risk Commun Res. 2024;202:7:1058-1071. https://jicrcr.com/index.php/jicrcr/article/view/1519
- G Aviello, B Romano, F Borrelli, R Capasso, L Gallo, F Piscitelli, V Di Marzo, AA Izzo. Chemopreventive effect of the non-psychotropic phytocannabinoid cannabidiol on experimental colon cancer. J Mol Med, 2012. [DOI | PubMed]
- MV Barbado, M Medrano, T Caballero-Velázquez, I Álvarez-Laderas, LI Sánchez-Abarca, E García-Guerrero, J Martín-Sánchez, IV Rosado, JI Piruat, P Gonzalez-Naranjo, NE Campillo, JA Páez, JA Pérez-Simón. Cannabinoid derivatives exert a potent anti-myeloma activity both in vitro and in vivo. Int J Cancer, 2017. [DOI | PubMed]
- B Bie, J Wu, JF Foss, M Naguib. An overview of the cannabinoid type 2 receptor system and its therapeutic potential. Curr Opin Anaesthesiol, 2018. [DOI | PubMed]
- C Blázquez, A Carracedo, M Salazar, M Lorente, A Egia, L González-Feria, A Haro, G Velasco, M Guzmán. Down-regulation of tissue inhibitor of metalloproteinases-1 in gliomas: a new marker of cannabinoid antitumoral activity?. Neuropharmacology, 2008. [DOI | PubMed]
- C Blázquez, A Carracedo, L Barrado, P José Real, J Luis Fernández-Luna, G Velasco, M Malumbres, M Guzmán, C Blázquez, A Carracedo, L Barrado, P José Real, J Luis Fernández-Luna, G Velasco, M Malumbres, M Guzmán. Cannabinoid receptors as novel targets for the treatment of melanoma. Faseb J, 2006. [DOI | PubMed]
- J Brandi, I Dando, M Palmieri, M Donadelli, D Cecconi. Comparative proteomic and phosphoproteomic profiling of pancreatic adenocarcinoma cells treated with CB1 or CB2 agonists. Electrophoresis, 2013. [DOI | PubMed]
- Cancer. (2025). Retrieved January 6, 2025, from https://www.who.int/news-room/fact-sheets/detail/cancer
- Cancer Tomorrow. (2025). Retrieved January 6, 2025, from https://gco.iarc.fr/tomorrow/en
- Chutoe C, Khunluck T, Charoenphandhu N, Lertsuwan K. The effect of cannabinoid receptor agonists on breast cancer and its interaction with bone cells. http://www.scisoc.or.th/BMBThailand/BMB2021/. 2021.
- C Duntsch, MK Divi, T Jones, Q Zhou, M Krishnamurthy, P Boehm, G Wood, A Sills, BM More. Safety and efficacy of a novel cannabinoid chemotherapeutic, KM-233, for the treatment of high-grade glioma. J Neurooncol, 2006. [DOI | PubMed]
- A Ellert-Miklaszewska, IA Ciechomska, B Kaminska. Synthetic Cannabinoids induce autophagy and mitochondrial apoptotic pathways in human glioblastoma cells independently of deficiency in TP53 or PTEN Tumor Suppressors. Cancers, 2021. [DOI]
- Filipiuc LE, Creangă-Murariu I, Tamba BI, Ababei DC, Rusu RN, Stanciu GD, Ștefanescu R, Ciorpac M, Szilagyi A, Gogu R, Filipiuc SI, Tudorancea IM, Solcan C, Alexa-Stratulat T, Cumpăt MC, Cojocaru DC, Bild V. JWH-182: a safe and effective synthetic cannabinoid for chemotherapy-induced neuropathic pain in preclinical models. Scientific Reports, . 2024;14(1). 10.1038/s41598-024-67154-y
- S Fogli, P Nieri, A Chicca, B Adinolfi, V Mariotti, P Iacopetti, MC Breschi, S Pellegrini. Cannabinoid derivatives induce cell death in pancreatic MIA PaCa-2 cells via a receptor-independent mechanism. FEBS Lett, 2006. [DOI | PubMed]
- F Gado, R Ferrisi, S Di Somma, F Napolitano, KA Mohamed, LA Stevenson, S Rapposelli, G Saccomanni, G Portella, RG Pertwee, RB Laprairie, AM Malfitano, C Manera. Synthesis and In Vitro characterization of selective cannabinoid CB2 receptor agonists: biological evaluation against neuroblastoma cancer cells. Molecules, 2022. [DOI | PubMed]
- H Gui, X Liu, Z-W Wang, D-Y He, D-F Su, S-M Dai. Expression of cannabinoid receptor 2 and its inhibitory effects on synovial fibroblasts in rheumatoid arthritis. Rheumatology, 2014. [DOI | PubMed]
- SN Gurley, AH Abidi, P Allison, P Guan, C Duntsch, JH Robertson, SD Kosanke, ST Keir, DD Bigner, AJ Elberger, BM Moore. Mechanism of anti-glioma activity and in vivo efficacy of the cannabinoid ligand KM-233. J Neurooncol, 2012. [DOI | PubMed]
- Gustafsson SB, Lindgren T, Jonsson M, Jacob SO. (2008). Cannabinoid receptor-independent cytotoxic effects of cannabinoids in human colorectal carcinoma cells: synergism with 5-fluorouracil. Cancer Chemotherapy and Pharmacology 2008;63(4):691-701. 10.1007/S00280-008-0788-5
- T Gülck, BL Møller. Phytocannabinoids: Origins and Biosynthesis. Trends Plant Sci, 2020. [DOI | PubMed]
- K Gustafsson, B Sander, J Bielawski, YA Hannun, J Flygare. Potentiation of cannabinoid-induced cytotoxicity in mantle cell lymphoma through modulation of ceramide metabolism. Mol Cancer Res, 2009. [DOI | PubMed]
- L Hanuš, A Breuer, S Tchilibon, S Shiloah, D Goldenberg, M Horowitz, RG Pertwee, RA Ross, R Mechoulam, E Fride. HU-308: A specific agonist for CB 2, a peripheral cannabinoid receptor. Proc Natl Acad Sci, 1999. [DOI | PubMed]
- HM Hashiesh, C Sharma, SN Goyal, NK Jha, S Ojha. Pharmacological properties, therapeutic potential and molecular mechanisms of JWH133, a CB2 receptor-selective agonist. Front Pharmacol, 2021. [DOI | PubMed]
- B Hinz, R Ramer. Cannabinoids as anticancer drugs: current status of preclinical research. Br J Cancer, 2022. [DOI | PubMed]
- H Iwamura, H Suzuki, Y Ueda, T Kaya, T Inaba. In vitro and in vivo pharmacological characterization of JTE-907, a novel selective ligand for cannabinoid CB2 receptor. J Pharmacol Exp Ther, 2001. [DOI | PubMed]
- Jones A. Longdom Publishing SL: Open access journals. Longdom. https://www.longdom.org/open-access/pathology-of-cancer-causes-pathophysiology-diagnosis-prevention-and-treatment-102140.html#ai. 2023.
- H Koltai, N Shalev. Anti-Cancer Activity of Cannabis sativa Phytocannabinoids: Molecular Mechanisms and Potential in the Fight against Ovarian Cancer and Stem Cells. Cancers, 2022. [DOI | PubMed]
- H-C Lu, K Mackie. An introduction to the endogenous cannabinoid system. Biol Psychiat, 2016. [DOI | PubMed]
- H-C Lu, K Mackie. Review of the Endocannabinoid System. Biol Psychiatry, 2021. [DOI]
- M Maccarrone, A Finazzi-Agró. The endocannabinoid system, anandamide and the regulation of mammalian cell apoptosis. Cell Death Differ, 2003. [DOI | PubMed]
- AM Malfitano, C Laezza, M Galgani, G Matarese, A D’Alessandro, P Gazzerro, M Bifulco. The CB1 receptor antagonist rimonabant controls cell viability and ascitic tumour growth in mice. Pharmacol Res, 2012. [DOI | PubMed]
- Moreno E, Cavic M, Krivokuca A, Casadó V, Canela E. (2019). The Endocannabinoid System as a Target in Cancer Diseases: Are We There Yet? Front Pharmacol, 10. 10.3389/fphar.2019.00339
- A Ortega, E Rangel-López, A Hidalgo-Miranda, A Morales, E Ruiz-García, A Meneses-García, A Herrera-Gómez, JL Aguilar-Ponce, IG González-Herrera, P Guevara-Salazar, O Prospero-García, SA Del Angel. On the effects of CP 55–940 and other cannabinoid receptor agonists in C6 and U373 cell lines. Toxicol in Vitro, 2015. [DOI | PubMed]
- H Okazaki, S Takeda, H Ishii, S Matsuo, E Furuta, K Watanabe, H Aramaki. HU-210, a synthetic analog of ∆9-THC, is not a modifier of estrogen signaling in MCF-7 human breast cancer cells. Fundamental Toxicological Sciences, 2016. [DOI]
- A Ortega, VM García-Hernández, E Ruiz-García, A Meneses-García, A Herrera-Gómez, JL Aguilar-Ponce, E Montes-Servín, O Prospero-García, SA Del Angel. Comparing the effects of endogenous and synthetic cannabinoid receptor agonists on survival of gastric cancer cells. Life Sci, 2016. [DOI | PubMed]
- A Preet, Z Qamri, MW Nasser, A Prasad, K Shilo, X Zou, JE Groopman, RK Ganju. Cannabinoid receptors, CB1 and CB2, as novel targets for inhibition of non-small cell lung cancer growth and metastasis. Cancer Prev Res (Philadelphia, Pa.), 2011. [DOI]
- Z Qamri, A Preet, MW Nasser, CE Bass, G Leone, SH Barsky, RK Ganju. Synthetic cannabinoid receptor agonists inhibit tumor growth and metastasis of breast cancer. Mol Cancer Ther, 2009. [DOI | PubMed]
- WM Raup-Konsavage, M Johnson, CA Legare, GS Yochum, DJ Morgan, KE Vrana. Synthetic cannabinoid activity against colorectal cancer cells. Cannabis and Cannabinoid Research, 2018. [DOI | PubMed]
- LD Recht, R Salmonsen, R Rosetti, T Jang, G Pipia, T Kubiatowski, P Karim, AH Ross, R Zurier, N Scott Litofsky, S Burstein. Antitumor effects of ajulemic acid (CT3), a synthetic non-psychoactive cannabinoid. Biochem Pharmacol, 2001. [DOI | PubMed]
- RA Ross, HC Brockie, LA Stevenson, VL Murphy, F Templeton, A Makriyannis, RG Pertwee. Agonist-inverse agonist characterization at CB1 and CB2 cannabinoid receptors of L759633, L759656 and AM630. Br J Pharmacol, 1999. [DOI | PubMed]
- D Sarnataro, S Pisanti, A Santoro, P Gazzerro, AM Malfitano, C Laezza, M Bifulco. The Cannabinoid CB1 Receptor Antagonist Rimonabant (SR141716) Inhibits Human Breast Cancer Cell Proliferation through a Lipid Raft-Mediated Mechanism. Mol Pharmacol, 2006. [DOI | PubMed]
- AM Shoeib, AL Yarbrough, BM Ford, LN Franks, A Urbaniak, LL Hensley, LN Benson, S Mu, A Radominska-Pandya, PL Prather. Characterization of cannabinoid receptors expressed in Ewing sarcoma TC-71 and A-673 cells as potential targets for anti-cancer drug development. Life Sci, 2021. [DOI | PubMed]
- AG Silva, CFB Lopes, CG Carvalho Júnior, RG Thomé, HB dos Santos, R Reis, RIM de Ribeiro, A.. WIN 55,212–2 induces caspase-independent apoptosis on human glioblastoma cells by regulating HSP70, p53 and Cathepsin D. Toxicol in Vitro, 2019. [DOI | PubMed]
- A Vaccani, P Massi, A Colombo, T Rubino, D Parolaro. Cannabidiol inhibits human glioma cell migration through a cannabinoid receptor-independent mechanism. Br J Pharmacol, 2005. [DOI | PubMed]
- V Voicu, F-M Brehar, C Toader, R-A Covache-Busuioc, AD Corlatescu, A Bordeianu, HP Costin, B-G Bratu, L-A Glavan, AV Ciurea. Cannabinoids in medicine: a multifaceted exploration of types, therapeutic applications, and emerging opportunities in neurodegenerative diseases and cancer therapy. Biomolecules, 2023. [DOI | PubMed]
- KB Walsh, HK Andersen. Molecular pharmacology of synthetic cannabinoids: Delineating cb1 receptor-mediated cell signaling. In Int J Mol Sci, 2020. [DOI]
- M Widmer, CO Hanemann, J Zajicek. High concentrations of cannabinoids activate apoptosis in human U373MG glioma cells. J Neurosci Res, 2008. [DOI | PubMed]
- XS Xian, H Park, YK Cho, IS Lee, SW Kim, MG Choi, IS Chung, KH Han, JM Park. Effect of a synthetic cannabinoid agonist on the proliferation and invasion of gastric cancer cells. J Cell Biochem, 2010. [DOI | PubMed]
- Yazbeck V, Alesi E, Myers J, Hackney MH, Cuttino L, Gewirtz DA. An overview of chemotoxicity and radiation toxicity in cancer therapy . 2022:1–27. 10.1016/bs.acr.2022.03.007
- S Zou, U Kumar. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int J Mol Sci, 2018. [DOI | PubMed]
