Renal vascular patterns assessment before robot-assisted partial nephrectomy: a video-based correlation with hilum dissection
Abstract
Purpose:
Surgical video review (SVR) is an emerging tool for assessing patient outcomes, especially in complex surgeries such as robot-assisted partial nephrectomy (RAPN). Adhesive probability and morphological scores are used to evaluate fat management and warm ischemia, respectively; however, the factors influencing renal hilum control (RHC) during RAPN have not yet been assessed. The aim of this study is to use SVR to identify the renal vascular patterns and factors that influence RHC.
Methods:
We evaluated 60 surgical video recordings of patients undergoing RAPN in 2023–2024, and measured the time to hilum control (THC) and total operation time (TOT) using a stopwatch. Patient and surgical factors were recorded and SPSS software was used to identify the correlation of these factors and vascular patterns with THC.
Results:
We observed a median THC of 22.7 min representing 15.1% of TOT. A significant correlation was found between previous renal surgery (p = 0.033), complex vascular anatomy on the right side (artery bifurcation behind IVC) or left side (more than one artery) (p = 0.02) and a longer THC. No significant difference was found between surgeons (p = 0.753) or surgical approach (transperitoneal vs. retroperitoneal, p = 0.87).
Conclusion:
THC represents a relatively short part of the total RAPN time. Previous renal surgery and a complex vascular pattern with artery bifurcation behind IVC on the right side and more than one main renal artery on the left side, can lead to longer THC. A detailed understanding of renal vascular patterns can provide a patient-specific surgical planning and optimise strategies for RAPN.
Article type: Research Article
Keywords: Partial nephrectomy, Vascular pattern, Renal hilum dissection, Surgical video
Affiliations: https://ror.org/05yn9cj95grid.417290.90000 0004 0627 3712Department of Urology, Sorlandet Hospital, Kristiansand, Norway; https://ror.org/00pk1yr39grid.414311.20000 0004 0414 4503Department of Urology, Sorlandet Hospital, Arendal, Norway
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s00345-025-05701-6 | PubMed: 40448715 | PMC: PMC12126307
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (895 KB)
Introduction
Minimally invasive partial nephrectomy (PN), whenever feasible, is the standard of care for patients diagnosed with T1a and T1b kidney cancer [ref. 1]. However, this approach is technically challenging and requires a detailed understanding of renal and tumour surgical anatomy to optimise oncological, functional and perioperative outcomes [ref. 2].
Contrast-enhanced computer tomography (CT) of the abdomen is the reference standard for primary imaging of renal tumours and the vascular system. Study the renal arterial anatomy via CT scan has been previously used for arterial tree representation [ref. 3]. In approximately 75% of cases, a single renal artery and vein are present, but a high percentage (15–20%) of patients also show anatomic variations such as duplication or early branching [ref. 2]. Disposition of the renal hilar structures and their numbers are significantly more variable than the classical pattern given in standard textbooks of anatomy, and a precise knowledge of both normal and variant anatomies of renal hilum is essential in the era of robotic surgery [ref. 4].
Robotic surgery platforms enable high-quality video recordings, providing greater magnification and closer views of anatomical details. Surgical video review (SVR) is an emerging tool for assessing patient outcomes [ref. 5], especially in complex surgeries such as robot-assisted PN (RAPN). Three key surgical phases are critical during RAPN: renal hilum control (RHC), fat management and warm ischemia time (WIT). Adhesive probability and morphological scores are respectively used to evaluate fat management [ref. 6] and warm ischemia [ref. 7], along with SVR; however, the factors influencing RHC during RAPN have not yet been assessed.
The aim of this study is to use SVR to identify the factors that influence RHC according to the renal vascular patterns identified via renal CT scan.
Material and methods
The storage of recorded surgical videos for a period of time is mandatory in Norway to ensure quality of healthcare services and provide medicolegal evidence. This requirement allowed us to retrospectively review surgical video recordings of 60 consecutive patients undergoing RAPN at our institution between January 2023 and June 2024. All procedures were performed on T1a tumours using Intuitive Xi robotic systems (Intuitive™, Sunnyvale, CA, USA). As part of the kidney cancer surgical team, one surgeon (O.B.) reviewed the video recordings and measured time to hilum control (THC) and total operation time (TOT) using a digital stopwatch and exact visual cues for starting and ending points. For THC, we defined the starting point as when the monopolar-curved scissors’ cut function is first used to dissect the renal hilum (gonadal vein on the left side, inferior vena cava (IVC) on the right side), right after colon decollation for the transperitoneal approach or the posterior opening of Gerota’s fascia, for the retroperitoneal approach. The ending point for THC was defined as when the Hem-o-lock clip is applied to the vessel loop surrounding the renal artery. In cases of more than one artery or the need to clamp the renal vein, the ending point was defined as when the Hem-o-lock clip is applied to the vessel loop surrounding the last vascular structure of the hilum. The TOT compromised the time from the first to the last use of the robotic instruments.
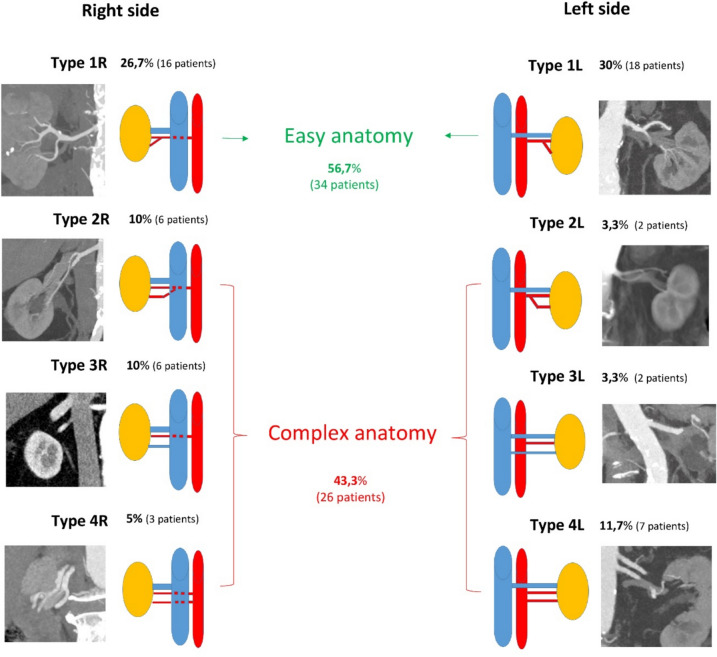
We assessed the renal vascular patterns using evaluation of the arterial and venous phase of the CT scans, performed preoperatively. Eight different vascular patterns (Fig. 1) were identified according to side, four on the right (Type 1R, 2R, 3R and 4R) and four on the left (Type 1L, 2L, 3L and 4L). Also the number of arteries or veins and proximity of the artery bifurcation to aorta were taken into consideration. We defined the anatomy of the renal hilum as easy if only one artery with a late bifurcation (lateral for the IVC on the right side, and more than 2 cm from the aorta on the left side) and one vein were present (Type 1L and 1R). Early bifurcation of the artery (behind IVC on the right side for Type 2R, and less than 2 cm from the aorta on the left side for type 2L) and more than one vein (Type 3R and 3L) or artery (Type 4R and 4L) was defined as a complex anatomy.
Patient factors such as age, BMI, previous renal surgery, inflammation (pyelonephritis or pancreatitis), radiation and immunotherapy were collected from patients’ medical records, as well as vascular anatomy and patterns observed in CT scans. Surgical factors such as intraoperative bleeding during RHC, surgeon, and approach were recorded and analysed using descriptive statistics.
We used SPSS software, including independent samples t-test, one-way ANOVA and Bonferroni multiple comparisons to identify the correlation of these factors and vascular patterns with THC. Additionally, we used stepwise multivariable regression models to assess the predictors, and statistical significance was set at p < 0.05.
Results
Population and surgical features are summarised in Table 1. The cohort comprised 72% males and had a median age of 67.5 years and an interquartile range (IQR) of 15. Most of the patients did not have previous kidney surgery, inflammation, radiotherapy or immunotherapy. The operations were distributed between three surgeons in a 1:1.8:1.5 manner and only 18.3% of RAPN were performed via a retroperitoneal approach.
Table 1: Population demographics and surgical features
| Variable | Overall(n = 60) | Variable | Overall(n = 60) |
|---|---|---|---|
| Age (years)Median [IQR] | 67.5 (15) | Vascular patternsType 1RType 2RType 3RType 4RType 1LType 2LType 3LType 4 L | 16 (26.7%)6 (10%)6 (10%)3 (5%)18 (30%)2 (3.3%)2 (3.3%)7 (11.7%) |
| Gender | |||
| Male | 43 (71.7%) | ||
| Female | 17 (28.3%) | ||
| Previous surgery | |||
| Yes | 4 (6.7%) | ||
| No | 56 (93.3%) | ||
| Previous Inflamation | Anatomy | ||
| Yes | 2(3.3%) | Easy | 34 (56.7%) |
| No | 58 (96.7%) | Complex | 26 (43.3%) |
| Previous radiotherapy | Operator | ||
| Yes | 1 (1.7%) | 1 | 14 (23.3%) |
| No | 59 (98.3) | 2 | 25 (41.7%) |
| 3 | 21 (35%) | ||
| Previous imunotherapy | Surgical approach | ||
| Yes | 2 (3.3%) | Transperitoneal | 49 (81.7%) |
| No | 58 (96.7%) | Retroperitoneal | 11 (18.3%) |
| BMIMedian [IQR] | 28.1 (6) | Time to hilum control(minutes) | |
| Side | Median [IQR] | 22.7 (17) | |
| Right | 31 (51.7%) | ||
| Left | 29 (48.3%) | ||
| Number of arteries | Total operative time | ||
| 1 | 50 (83.3%) | Median [IQR] | 133.6 (40) |
| 2 | 9 (15%) | ||
| 3 | 1 (1.7%) | ||
| Number of veins | Intraoperative bleeding | ||
| 1 | 53 (88.3%) | Yes | 4 (8.7%) |
| 2 | 7 (11.7%) | No | 56 (91.3%) |
More than 80% of the patients had only one artery or one vein and the eight vascular patterns are depicted in Fig. 1. Type 1R and 1L were characterised as easy anatomy and represented more than half of the patients (56.7%).
We observed a median THC of 22.7 min, representing 15.1% of TOT. A significant correlation was found between previous renal surgery and longer THC (p = 0.033), as presented in Table 2.
Table 2: Factors correlated with time to hilum control and the percent from total operative time
| Variable | N | THC | Percent THC/TOT | ||
|---|---|---|---|---|---|
| Mean(minutes) */Correlation** | p | Mean (%)*/Correlation. ** | p | ||
| Gender* | 0.265 | 0.281 | |||
| Male | 43 | 24.9 | 17.2 | ||
| Female | 17 | 21.2 | 17.7 | ||
| Previous surgery | 0.033 | 0.010 | |||
| Yes | 56 | 23.1 | 16.7 | ||
| No | 4 | 35.6 | 26.5 | ||
| Previous inflammation | |||||
| Yes | 58 | 23.5 | 0.129 | 17.1 | 0.061 |
| No | 2 | 36.1 | 27.1 | ||
| Previous radiation | |||||
| Yes | 59 | 23.9 | 0.981 | 17.4 | 0.588 |
| No | 1 | 24.2 | 13.1 | ||
| Previous immunotherapy | |||||
| Yes | 58 | 24.1 | 0.621 | 17.4 | 0.673 |
| No | 2 | 19.9 | 15.1 | ||
| Side* | 0.231 | 0.672 | |||
| Right | 31 | 22.2 | 16.9 | ||
| Left | 29 | 25.7 | 17.7 | ||
| Renal arteries* | |||||
| 1 | 50 | 21.9 | 0.007 | 16.3 | 0.051 |
| 2 | 9 | 34.7 | 22.9 | ||
| 3 | 1 | 25.4 | 17.8 | ||
| Renal veins* | 0.482 | 0.446 | |||
| 1 | 53 | 24.3 | 17.6 | ||
| 2 | 7 | 21.0 | 15.3 | ||
| Anatomy* | |||||
| Easy | 34 | 19.7 | 0.001 | 14.8 | 0.002 |
| Complex | 26 | 29.4 | 20.7 | ||
| Vascular pattern* | |||||
| Type 1RType 2RType 3RType 4RType 1LType 2LType 3LType 4L | 16 | 16.5 | 0.02 | 13.1 | 0.03 |
| 6 | 34.5 | 25.9 | |||
| 6 | 20.6 | 15.8 | |||
| 3 | 30.7 | 22.0 | |||
| 18 | 22.5 | 16.3 | |||
| 2 | 24.3 | 18.9 | |||
| 2 | 23.8 | 12.8 | |||
| 7 | 35.1 | 22.6 | |||
| Operator* | 0.753 | 0.220 | |||
| 1 | 14 | 25.9 | 19.5 | ||
| 2 | 25 | 23.4 | 17.9 | ||
| 3 | 21 | 23.1 | 15.2 | ||
| Surgical approach* | 0.387 | 0.062 | |||
| Transperitoneal | 49 | 23.3 | 16.5 | ||
| Retroperitoneal | 11 | 26.6 | 21.1 | ||
| Intraoperative bleeding* | 0.05 | 0.242 | |||
| Yes | 56 | 23.1 | 17.2 | ||
| No | 4 | 34.6 | 18.7 | ||
| Age** | 60 | .085 | 0.516 | 0.016 | 0.902 |
| BMI** | 60 | .089 | 0.499 | 0.086 | 0.514 |
Bold and bold Italic for "p" means p<0.05
THC time to hilum control, TOT total operative time, BMI body mass index
*ANOVA/t-test
**Pearson
Having a complex vascular anatomy (p = 0.001) with a particular vascular pattern (p = 0.02) and more than one kidney artery (p = 0.007) correlated with longer THC. The Bonferroni multiple comparisons of the vascular patterns demonstrated that type 2R (renal artery bifurcation behind the IVC) and 4L (more than one main artery on the left side) were significantly associated with longer THC compared to the other vascular patterns (Table 3).
Table 3: Bonferroni multiple comparisons having time to hilum control as the dependent variable
| (I) Vascular pattern | (J) Vascular pattern | Mean difference (I-J) | Std. error | Sig | 95% Confidence Interval | |
|---|---|---|---|---|---|---|
| Lower bound | Upper bound | |||||
| Type 1R | Type 2R | −18.0135 | 4.7379 | 0.011 | −33.616 | −2.411 |
| Type 3R | −4.0608 | 4.7379 | 1.000 | −19.664 | 11.542 | |
| Type 4R | −14.1913 | 6.2268 | 0.750 | −34.697 | 6.315 | |
| Type 1L | −5.9756 | 3.4006 | 1.000 | −17.174 | 5.223 | |
| Type 2L | −7.7469 | 7.4228 | 1.000 | −32.192 | 16.698 | |
| Type 3L | −7.3135 | 7.4228 | 1.000 | −31.758 | 17.131 | |
| Type 4L | −18.5278 | 4.4850 | 0.004 | −33.298 | −3.758 | |
| Type 2R | Type 1R | 18.0135 | 4.7379 | 0.011 | 2.411 | 33.616 |
| Type 3R | 13.9528 | 5.7141 | 0.505 | −4.865 | 32.771 | |
| Type 4R | 3.8222 | 6.9983 | 1.000 | −19.225 | 26.869 | |
| Type 1L | 12.0380 | 4.6655 | 0.357 | −3.327 | 27.403 | |
| Type 2L | 10.2667 | 8.0809 | 1.000 | −16.346 | 36.879 | |
| Type 3L | 10.7000 | 8.0809 | 1.000 | −15.912 | 37.312 | |
| Type 4L | −0.5143 | 5.5062 | 1.000 | −18.648 | 17.619 | |
| Type 3R | Type 1R | 4.0608 | 4.7379 | 1.000 | −11.542 | 19.664 |
| Type 2R | −13.9528 | 5.7141 | 0.505 | −32.771 | 4.865 | |
| Type 4R | −10.1306 | 6.9983 | 1.000 | −33.177 | 12.916 | |
| Type 1L | −1.9148 | 4.6655 | 1.000 | −17.279 | 13.450 | |
| Type 2L | −3.6861 | 8.0809 | 1.000 | −30.298 | 22.926 | |
| Type 3L | −3.2528 | 8.0809 | 1.000 | −29.865 | 23.360 | |
| Type 4L | −14.4671 | 5.5062 | 0.316 | −32.600 | 3.666 | |
| Type 4R | Type 1R | 14.1913 | 6.2268 | 0.750 | −6.315 | 34.697 |
| Type 2R | −3.8222 | 6.9983 | 1.000 | −26.869 | 19.225 | |
| Type 3R | 10.1306 | 6.9983 | 1.000 | −12.916 | 33.177 | |
| Type 1L | 8.2157 | 6.1719 | 1.000 | −12.110 | 28.541 | |
| Type 2L | 6.4444 | 9.0348 | 1.000 | −23.309 | 36.198 | |
| Type 3L | 6.8778 | 9.0348 | 1.000 | −22.876 | 36.631 | |
| Type 4L | −4.3365 | 6.8296 | 1.000 | −26.828 | 18.155 | |
| Type 1L | Type 1R | 5.9756 | 3.4006 | 1.000 | −5.223 | 17.174 |
| Type 2R | −12.0380 | 4.6655 | 0.357 | −27.403 | 3.327 | |
| Type 3R | 1.9148 | 4.6655 | 1.000 | −13.450 | 17.279 | |
| Type 4R | −8.2157 | 6.1719 | 1.000 | −28.541 | 12.110 | |
| Type 2L | −1.7713 | 7.3768 | 1.000 | −26.065 | 22.522 | |
| Type 3L | −1.3380 | 7.3768 | 1.000 | −25.632 | 22.956 | |
| Type 4L | −12.5522 | 4.4085 | 0.176 | −27.070 | 1966 | |
| Type 2L | Type 1R | 7.7469 | 7.4228 | 1.000 | −16.698 | 32.192 |
| Type 2R | −10.2667 | 8.0809 | 1.000 | −36.879 | 16.346 | |
| Type 3R | 3.6861 | 8.0809 | 1.000 | −22.926 | 30.298 | |
| Type 4R | −6.4444 | 9.0348 | 1.000 | −36.198 | 23.309 | |
| Type 1L | 1.7713 | 7.3768 | 1.000 | −22.522 | 26.065 | |
| Type 3L | 0.4333 | 9.8971 | 1.000 | −32.160 | 33.027 | |
| Type 4L | −10.7810 | 7.9353 | 1.000 | −36.914 | 15.352 | |
| Type 3L | Type 1R | 7.3135 | 7.4228 | 1.000 | −17.131 | 31.758 |
| Type 2R | −10.7000 | 8.0809 | 1.000 | −37.312 | 15.912 | |
| Type 3R | 3.2528 | 8.0809 | 1.000 | −23.360 | 29.865 | |
| Type 4R | −6.8778 | 9.0348 | 1.000 | −36.631 | 22.876 | |
| Type 1L | 1.3380 | 7.3768 | 1.000 | −22.956 | 25.632 | |
| Type 2L | −0.4333 | 9.8971 | 1.000 | −33.027 | 32.160 | |
| Type 4L | −11.2143 | 7.9353 | 1.000 | −37.347 | 14.918 | |
| Type 4L | Type 1R | 18.5278 | 4.4850 | 0.004 | 3.758 | 33.298 |
| Type 2R | 0.5143 | 5.5062 | 1.000 | −17.619 | 18.648 | |
| Type 3R | 14.4671 | 5.5062 | 0.316 | −3.666 | 32.600 | |
| Type 4R | 4.3365 | 6.8296 | 1.000 | −18.155 | 26.828 | |
| Type 1L | 12.5522 | 4.4085 | 0.176 | −1.966 | 27.070 | |
| Type 2L | 10.7810 | 7.9353 | 1.000 | −15.352 | 36.914 | |
| Type 3L | 11.2143 | 7.9353 | 1.000 | −14.918 | 37.347 | |
Bold and bold Italic for "p" means p<0.05
Intraoperative bleeding during RHC was low (8.7%) and correlated with longer THC (p = 0.05), though no significant difference was found between surgeons (p = 0.753) or surgical approach (transperitoneal vs. retroperitoneal, p = 0.87).
After stepwise regression, the best model identified to predict THC included anatomical complexity and previous surgery (R-square = 0.281, p = 0.006), as shown in Table 4.
Table 4: Stepwise regression analyses for warm ischemia time, excision time, reconstruction time and intermediate time
| Model summary | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Model | R | R square | Adjusted R square | Std. error of the estimate | Change statistics | ||||
| R square change | F change | df1 | df2 | Sig. F Change | |||||
| 1 | 0.423a | 0.179 | 0.165 | 628.490 | 0.179 | 12.673 | 1 | 58 | < 0.001 |
| 2 | 0.530b | 0.281 | 0.256 | 593.283 | 0.102 | 8.088 | 1 | 57 | 0.006 |
aPredictors: (Constant), Anatomy Complexity
bPredictors: (Constant), Anatomy Complexity, Previous Surgery
Discussion
Our present study identified, on CT scan, several patterns according to the disposition of the vascular structures in the renal hilum. The analyses revealed which patterns correlate to a more complex anatomy and longer THC.
To the best of our knowledge, no data are available on intraoperative video documentation review for RHC assessment during RAPN. Previous studies have assessed the importance of SVR. For example, De Backer et al. [ref. 5] considered 100 RAPNs and showed that surgical phase duration can be correlated with certain clinical outcomes. Kim et al. [ref. 6] tested SVR to assess the Mayo adhesive probability score and perirenal fat dissection time. Additionally, our group used SVR to evaluate WIT during RAPN and its impact on surgical margins and complication rate, in a previous study [ref. 7].
We found THC to be a relatively short portion (15%) of the TOT. For the present study, TOT depicts the total console surgery time as the time used to achieve pneumoperitoneum or docking the robot were not taken into account. De Backer et al. [ref. 5] also found a short time to hilum control with a median of 16 (±11.5) minutes, as compared to 22 min in our study. This could be due to including patients with redo RAPN and complex vascular anatomy in our cohort. Of the 100 patients in the cohort of De Backer et al., 77 procedures were performed offclamp that requires less dissection of all the hilar structures.
Previous renal surgery was registered in four of the patients who underwent a redo RAPN for tumour resection elsewhere in the kidney than previous resection bed. It correlated to longer THC as more fibrosis around the vascular structures in the hilum needed to be dissected in order to achieve RHC. No statistical correlation was found between longer THC and previous inflammation like pyelonephritis or pancreatitis, previous radiotherapy, or previous immunotherapy. This finding could be biased by the low number of patients (only one or two) who did not have such previous conditions. Though previous abdominal surgery poses surgical challenges for PN [ref. 8], several studies identified no impact on the outcomes of RAPN [ref. 9, ref. 10]. Redo RAPN is considered an effective approach [ref. 11], and evidence indicates good feasibility and safety, though it is a challenging procedure [ref. 12]. One challenge is RHC and it makes sense that more time is used to dissect the vascular structures, as more fibrosis around the hilar structures is present from previous RHC. Though RAPN remains the preferred option for treating small renal masses, percutaneous cryoablation is a valid alternative, particularly for challenging patients, as shown by Iossa et al. [ref. 13]. Two out of four patients who underwent redo RAPN were performed retroperitoneally. This could have biased our results together with a disproportioned percent of only 18% of the patients performed a retroperitoneally approach. This could also explain why we registered longer THC in the retroperitoneal group.
The anatomy of the renal vascular system and its application to PN was initially presented by Graves in 1954 with a special focus on segmental branching of the renal artery and its role in achieving selective ischemia [ref. 14]. Later, Trivedi et al. [ref. 15] identified six vascular branching patterns of segmental renal artery in an anatomical study and stated that knowledge of these patterns is essential to effective surgical planning in cases of PN and preventing complications. Clamping the segmental renal artery instead of the main renal artery during PN is a promising technique to decrease WIT, and understanding vascular branching patterns is essential for an appropriate hilar approach. However, selective or superselective clamping do not provide better renal function preservation than renal artery conventional clamping, as shown in systematic reviews [ref. 16] and randomized controlled trials [ref. 17]. This places doubt on the benefit of these techniques which additionally has higher risk of bleeding. To the best of our knowledge, no data are available on renal vascular anatomy with focus on conventional clamping, and our study identified vascular patterns that help for a more detailed understanding of renal vascular anatomy including cases of renal hilar masses, where RAPN is a promising technique [ref. 18].
Multiple authors have described the arrangement of renal hilar structures, through anatomical studies, identifying several patterns [ref. 4, ref. 19] which included the renal pelvis into their classifications. We used vascular structures in our classification as only the vein and artery are dissected during the RHC for PN. We identified eight patterns, four for each side, including both the normal structure and variations. A complete understanding of underlying normal and aberrant renal anatomy, coupled with patient- and tumour-specific anatomical characteristics, represents the foundation for proper preoperative surgical planning for PN [ref. 20]. We determined that having more than one renal artery requires longer THC as more time is expected to be used to dissect all the arteries to be clamped. In our classification, we did not include small accessory arteries that did not need to be clamped during WIT. An early bifurcation of the renal artery on both sides but especially on the right side, as the artery passes behind the IVC made the dissection of the main artery more difficult, and longer THC was observed.
We did not register a statistical significance between the three surgeons. Larcher et al. [ref. 21] indicated that RAPN outcomes might be affected by surgeon experience by shortening WIT and lowering the complication rate. The inhomogeneous surgical experience among the surgical team members and the small sample size caused us to evaluate the surgeons as an independent variable, instead of surgeon’s expertise, and that may have biased our results. Intraoperative bleeding rate was low during RHC and correlated with longer THC as more time was needed to control the bleeding before controlling the renal hilum.
Some patient characteristics and comorbidities were not recorded due to the retrospective design of this study. The study was not blinded because SVR requires a trained kidney surgeon to identify the specific steps of the procedure and no interobserver validation of pattern assignment was attempted. The vascular patterns identified in our study are valuable for cases when clamping of the main artery is chosen; for selective clamping, different intrarenal anatomical patterns should be considered. Despite these limitations, our results can be used to optimise preoperative surgical planning for RAPN and provide a basis for developing a hilum complexity score in the future.
Conclusion
THC represents a relatively short part of total console time during RAPN. Previous renal surgery and a complex vascular pattern with artery bifurcation behind IVC on the right side and more than one main renal artery on the left side can lead to longer THC. A detailed understanding of renal vascular patterns can provide patient-specific surgical planning and optimise surgical strategies for RAPN.
References
- B Ljungberg, L Albiges, Y Abu-Ghanem. European Association of Urology Guidelines on Renal Cell Carcinoma: the 2022 update. Eur Urol, 2022. [DOI | PubMed]
- T Klatte, V Ficarra, C Gratzke, J Kaouk, A Kutikov, V Macchi, A Mottrie, F Porpiglia, J Porter, CG Rogers, P Russo, RH Thompson, RG Uzzo, CG Wood, IS Gill. A literature review of renal surgical anatomy and surgical strategies for partial nephrectomy. Eur Urol, 2015. [DOI | PubMed]
- F Rocco, LA Cozzi, G Cozzi. Study of the renal segmental arterial anatomy with contrast-enhanced multi-detector computed tomography. Surg Radiol Anat, 2015. [DOI | PubMed]
- DSD Jadhav, DBR Zambare. Anatomical study of arrangement of renal hilar structures in indian adult human cadavers: anatomical study of arrangement of renal hilar structures. Natl J Integr Res Med, 2018. [DOI]
- P De Backer, M Peraire Lores, M Demuynck. Surgical phase duration in robot-assisted partial nephrectomy: a surgical data science exploration for clinical relevance. Diagnostics, 2023. [DOI | PubMed]
- H Kim, M Kim, SS Byun, SK Hong, S Lee. Clinical implication of adherent perinephric fat in robot-assisted partial nephrectomy: validation with video review. Front Surg, 2022. [DOI | PubMed]
- OS Barnoiu, HR Yazdani Arazi, AV Andersen. Minimising warm ischaemia time during robot-assisted partial nephrectomy. A video-based assessment of tumour excision, kidney reconstruction and intermediate time. Scand J Urol, 2024. [DOI | PubMed]
- N Raison, N Doeuk, T Malthouse, V Kasivisvanathan, W Lam, B Challacombe. Challenging situations in partial nephrectomy. Int J Surg, 2016. [DOI | PubMed]
- S Rintoul-Hoad, R Catterwell, B Challacombe. Robot-assisted partial nephrectomy: excellent outcomes can persist despite previous abdominal surgery. BJU Int, 2016. [DOI | PubMed]
- H Zargar, W Isac, R Autorino, A Khalifeh, O Nemer, O Akca, H Laydner, LF Brandao, RJ Stein, JH Kaouk. Robot-assisted laparoscopic partial nephrectomy in patients with previous abdominal surgery: single center experience. Int J Med Robot, 2015. [DOI | PubMed]
- AT Beksac, U Carbonara, M Abou Zeinab, M Meagher, S Hemal, A Tafuri, G Tuderti, A Antonelli, R Autorino, G Simone, IH Derweesh, J Kaouk. Redo robotic partial nephrectomy for recurrent renal tumors: a multi-institutional analysis. J Endourol, 2022. [DOI | PubMed]
- F Di Maida, AA Grosso, R Campi, L Lambertini, ML Gallo, A Cadenar, V Salamone, S Coco, D Paganelli, A Tuccio, L Masieri, A Minervini. Redo partial nephrectomy for local recurrence after previous nephron-sparing surgery. Surgical insights and oncologic results from a high-volume robotic center. Eur Urol Open Sci, 2023. [DOI | PubMed]
- V Iossa, SD Pandolfo, R Buonopane, A Di Girolamo, F Fiore, G Sessa, R Vitale, A Ferraro, EM Amodeo, P Porcaro, E Punzi, G Lombardi, V Imperatore. Robot-assisted partial nephrectomy vs. percutaneous cryoablation for T1a renal tumors: a single-center retrospective analysis of outcomes and costs. Int Urol Nephrol, 2025. [DOI | PubMed]
- FT Graves. The anatomy of the intrarenal arteries and its application to segmental resection of the kidney. Br J Surg, 1954. [DOI | PubMed]
- S Trivedi, U Sharma, M Rathore, MR John. Comprehensive study of arrangement of renal hilar structures and branching pattern of segmental renal arteries: an anatomical study. Cureus, 2023. [DOI | PubMed]
- L Zhou, X Wei, WJ Sun, Q Liu, ZY Jian, H Li, KJ Wang. Selective versus hilar clamping during minimally invasive partial nephrectomy: a systematic review and meta-analysis. J Endourol, 2015. [DOI | PubMed]
- JA Long, G Fiard, J Giai, Y Teyssier, A Fontanell, C Overs, D Poncet, JL Descotes, JJ Rambeaud, A Moreau-Gaudry, T Ittobane, A Bouzit, JL Bosson, C Lanchon. Superselective ischemia in robotic partial nephrectomy does not provide better long-term renal function than Renal Artery Clamping in a Randomized Controlled Trial (EMERALD): should we take the risk?. Eur Urol Focus, 2022. [DOI | PubMed]
- SD Pandolfo, Z Wu, R Campi, R Bertolo, D Amparore, A Mari, P Verze, C Manfredi, A Franco, F Ditonno, C Cerrato, M Ferro, F Lasorsa, R Contieri, L Napolitano, A Tufano, G Lucarelli, S Cilio, S Perdonà, S Siracusano, R Autorino, A Aveta. Outcomes and techniques of robotic-assisted partial Nephrectomy (RAPN) for renal hilar masses: a comprehensive systematic review. Cancers (Basel), 2024. [DOI | PubMed]
- C Divya, NS Ashwini, BV Swaroop Raj, KV Venkateshu. Study of arrangement of renal hilar structures in human cadavers. Int J Anat Res, 2018. [DOI]
- H Zargar, K Zargar-Shoshtari, H Laydner, R Autorino. Anatomy of contemporary partial nephrectomy: a dissection of the available evidence. Eur Urol, 2015. [DOI | PubMed]
- A Larcher, F Muttin, B Peyronnet, G De Naeyer, ZE Khene, P Dell’Oglio, C Ferreiro, P Schatteman, U Capitanio, F D’Hondt, F Montorsi, K Bensalah, A Mottrie. The learning curve for robot-assisted partial nephrectomy: impact of surgical experience on perioperative outcomes. Eur Urol, 2019. [DOI | PubMed]
