Chemical Profiling by High‐Performance Liquid Chromatography With Diode Array Detection and Gas Chromatography‐Mass Spectrometry Analysis and Antimicrobial Potential of Achillea santolina Plant Extracts against Extended‐Spectrum Beta‐Lactamase‐producing Escherichia coli and Fungal Pathogens
Abstract
The study examines the chemical composition and antimicrobial properties of petroleum ether and hydro‐methanolic extracts of Achillea santolina from Algeria. Chemical profiling was performed using high‐performance liquid chromatography with diode array detection for the hydro‐methanolic extract and gas chromatography‐mass spectrometry for the petroleum ether extract. Antibacterial and antifungal activities were evaluated using the disc diffusion method and broth dilution technique. Epicatechin (24.54 mg/g extract), and camphor (19.18%) were identified as main compounds in the hydro‐methanolic and petroleum ether extracts, respectively. Both extracts showed significant antibacterial effects against extended‐spectrum beta‐lactamase‐producing Escherichia coli strains, with inhibition diameters ranging from 10 to 13 mm, and minimum inhibitory concentration (MIC) values between 0.78 and 3.5 mg/mL. Anti‐fungal activity was also notable, particularly against Candida albicans, with an inhibition diameter of 14 mm, and MIC values between 0.39 and 1.56 mg/mL. The hydro‐methanolic extract showed up to 90% of growth inhibition against Aspergillus niger. These findings suggest that A. santolina could serve as a promising source of antimicrobial compounds to combat resistant pathogens.
Article type: Research Article
Keywords: antibacterial activity, antifungal activity, HPLC‐DAD, GC‐MS
Affiliations: Department of Biology Laboratory of Plant Biology and Environment “Medicinal plants” Axis Faculty of Sciences_ Badji Mokhtar University Annaba Algeria; Department of Nature and Life Sciences, Laboratory of Interactions, Biodiversity, Ecosystems and Biotechnology, Faculty of Sciences University 20 August 1955 Skikda Algeria; Department of Natural and Life Sciences Faculty of Sciences University of M’sila M’sila Algeria; Department of Chemistry and Technologies of Drug Sapienza University Rome Italy; Biology Department, College of Science Imam Mohammad Ibn Saud Islamic University (IMSIU) Riyadh Saudi Arabia
License: © 2024 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202403064 | PubMed: 39714424 | PMC: PMC12081026
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.0 MB)
Introduction
Long before the discovery of microbes, certain plants were recognized for their healing properties, now understood as antimicrobial compounds. Traditionally used for treating infections, many of these plants remain in use and are actively studied for their effectiveness against various pathogens [ref. 1].
The global rise in extended‐spectrum beta‐lactamase (ESBL)‐producing Escherichia coli strains, in African countries like Algeria, poses a significant problem due to their resistance to certain antibiotics, specifically in urinary tract infections [ref. 2]. E. coli, a Gram‐negative facultative anaerobe bacterium, is a major contributor to various infections and is associated with a wide range of gastrointestinal diseases, including conditions such as enteritis, bacteremia, diarrhea, and urinary tract infections [ref. 3]. The excessive use of antibiotics has significantly contributed to the growing drug resistance of E. coli, posing a serious threat to public health. Consequently, many researchers are exploring natural compounds from plant extracts as alternative approaches to combat ESBL‐producing E. coli [ref. 3].
Additionally, fungal infections are increasing, especially candidiasis, with Candida albicans as the most prevalent symbiotic fungus, commonly coexisting with humans and animals as a part of the normal microbial flora [ref. 4]. Infections caused by C. albicans typically arise when the immune system is weakened, leading to conditions such as oral, vaginal, skin, and nail candidiasis, as well as more severe systemic fungal infections [ref. 5]. If left untreated, C. albicans infections can progress to bloodstream infections, causing symptoms such as fever, weakness, appetite loss, anemia, and organ failure, posing a significant threat to the health and survival of affected individuals [ref. 6]. In recent years, several antibiotics have been used to combat infections caused by C. albicans, providing a broader range of antifungal activity effective against complex C. albicans infections. However, the use of antibiotics poses a number of challenges [ref. 7]. Similarly, the fungal growth of molds like Aspergillus spp., including Aspergillus niger, is an important cause of food spoilage and intoxication, with increasing resistance to standard treatments [ref. 9]. Therefore, there is a growing need for new, environmentally friendly methods that can either replace or reduce the reliance on antibiotics.
The growing demand for alternative treatments with herbal medicine has grown in recent years, renewing interest in plants that are safe, culturally acceptable, and effective [ref. 10]. Achillea santolina, our plant of interest, belongs to the genus Achillea and family Asteraceae, which includes approximately 115 species primarily found across North Africa, Europe, and Asia [ref. 11]. In Algeria, around five species of Achillea, including A. santolina, are widely distributed [ref. 12]. This plant has a longstanding role in traditional medicine for treating various ailments [ref. 13]. Many Achillea species are known for their antiseptic and infection‐fighting properties [ref. 14].
The bioactive properties of Achillea essential oils and extracts, such as analgesic, antioxidant, anti‐inflammatory, and antimicrobial activities [ref. 15], have been well documented.
To the best of our knowledge, no studies have explored the chemical composition or evaluated the antimicrobial activity of A. santolina extracts in Algeria, while research on its essential oils has been conducted in regions such as Egypt [ref. 16], Jordan [ref. 17], and Iran [ref. 18]. This study is the first to evaluate the antimicrobial properties of A. santolina extracts, focusing on their effectiveness against resistant bacteria, particularly those producing ESBL. This work aims to explore the chemical composition of A. santolina extracts using gas chromatography‐mass spectrometry (GC‐MS) and high‐performance liquid chromatography with diode array detection (HPLC‐DAD) and to evaluate their anti‐bacterial and anti‐fungal activities, with a particular focus on their potential against resistant E. coli producing (ESBL) and other antifungal pathogens.
Results and Discussion
Chemical Composition by GC‐MS Analysis of the Petroleum Ether Extract of A. santolina
The GC‐MS analysis of the petroleum ether extract of A. santolina has identified 15 compounds, as shown in Table 1 and Figure 1. The primary constituents included camphor (19.2 %), palmitic acid (15.2 %), oleic acid (11.5 %), nonacosane (10.6 %), linoleic acid (7.7 %), and heptacosane (5.2 %). Camphor is commonly identified as a major compound in various Achillea species [ref. 19]. The GC/MS chromatogram is reported in Figure 2. The notable high camphor content in A. santolina, compared to other Achillea species, suggests that it belongs to a camphor‐dominant chemotype. The other constituents found in significant amounts were: dodecanoic acid, 3‐vanilpropanol, tetradecanoic acid, palmitelaidic acid, santonine, 3,7,11,15‐tetramethyl‐2‐hexadecene, stearic acid, pentacosane, and β‐sitosterol. The presence of these compounds or their interaction with other secondary metabolites may enhance the targeted elimination of microbial strains.
TABLE 1: Chemical composition of the petroleum ether extract of A. santolina from the Aflou region (Algeria) using the gas chromatography‐mass spectrometry (GC‐MS) technique.
| Peak No. | RT | Phenolic compounds | Molecular formula | RI | Petroleum ether extract (%) |
|---|---|---|---|---|---|
| 1 | 5.964 | Camphor | C₁₀H₁₆O | 1144 | 19.2 |
| 2 | 19.766 | Dodecanoic acid | C₁₂H₂₄O₂ | 1657 | tr |
| 3 | 23.74 | 3‐Vanilpropanol | C16H30O3Si2 | 1813 | tr |
| 4 | 24.225 | Tetradecanoic acid | C₁₄H₂₈O₂ | 1850 | tr |
| 5 | 27.894 | Palmitelaidic acid | C₁₆H₃₀O₂ | 2025 | tr |
| 6 | 28.322 | Palmitic acid | C₁₆H₃₂O₂ | 2045 | 15.2 |
| 7 | 30661 | Santonine | C15H18O3 | 2172 | tr |
| 9 | 30.886 | 3,7,11,15‐tetramethyl‐2‐hexadecene | C₂₀H₄₀ | 2180 | tr |
| 10 | 31.483 | Linoleic acid | C₁₈H₃₂O₂ | 2200 | 7.7 |
| 11 | 31.6 | Oleic acid | C₁₈H₃₄O₂ | 2208 | 11.5 |
| 12 | 32.091 | Stearic acid | C₁₈H₃₆O₂ | 2240 | tr |
| 13 | 36.59 | Pentacosane | C₂₅H₅₂ | 2500 | tr |
| 14 | 39.639 | Heptacosane | C₂₇H₅₆ | 2700 | 5.2 |
| 15 | 42.638 | Nonacosane | C₂₉H₆₀ | 2900 | 10.6 |
| 16 | 50.171 | β‐Sitosterol | C₂₉H₅₀O | 3380 | tr |
Abbreviations: RI, retention index; Rt, retention time; tr, percentage values ˂0.1%.
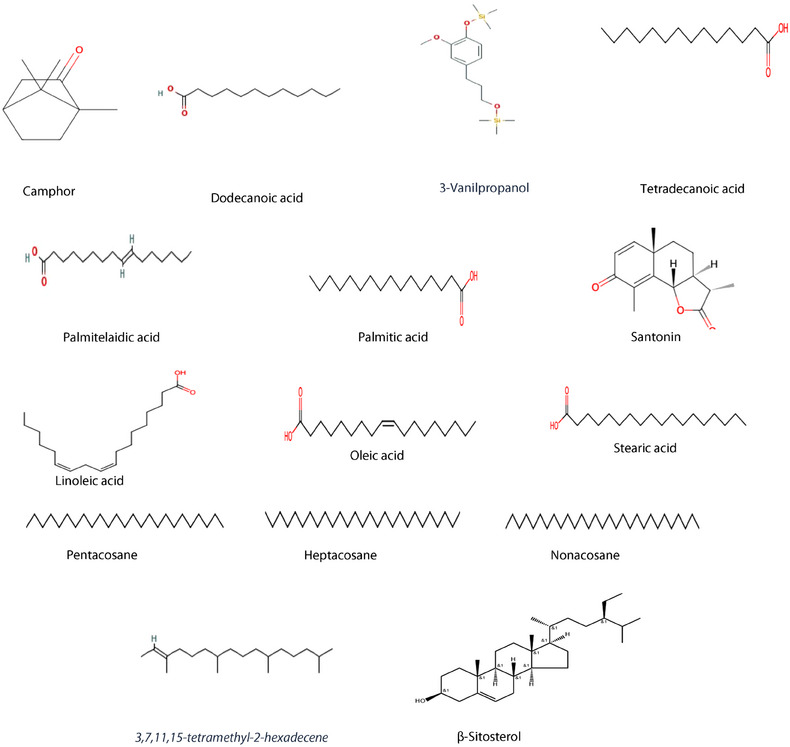
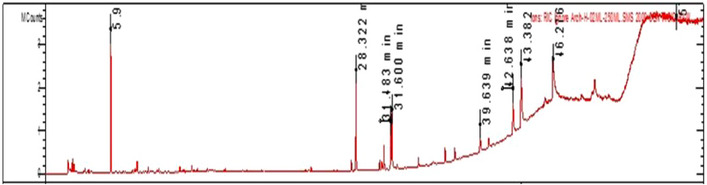
Berramdane et al. [ref. 20] reported that A. santolina essential oils from Algeria primarily contain camphor and 1,8‐cineole (see Figures 1, 2, 3, 4). In another Algerian region, A. santolina oils had cis‐sabinene hydrate and 1,8‐cineole as major components [ref. 21], while A. santolina grown in Egypt contains: 1,8‐cineole, camphor, bomeol, pinenes, artemisia ketone, and santolina alcohol as main compounds [ref. 22].
Quantitative Analysis of Phenolic Compounds by HPLC‐DAD
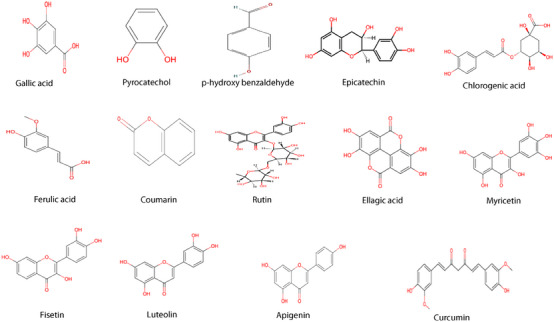
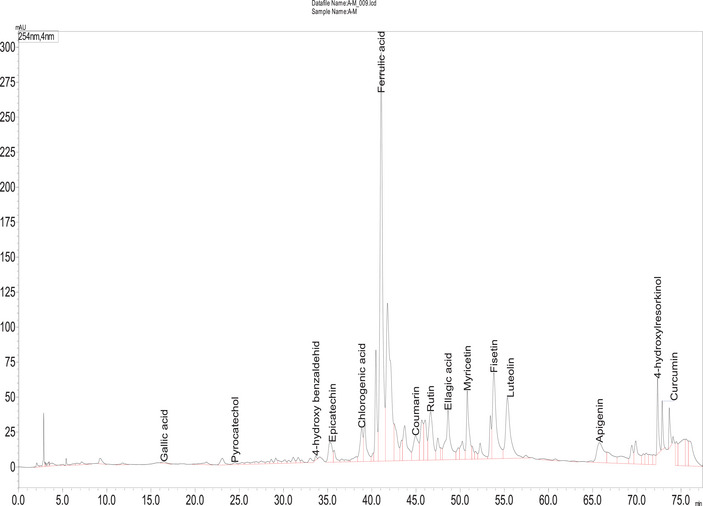
A reverse‐phase HPLC‐DAD method was used to analyze the phenolic compounds. For an optimal separation, various solvent combinations and flow rates were tested to acheive optimal separation. All chemicals used in this study were of HPLC grade. Compounds were identified by comparing their spectroscopic properties and retention times with reference standards (Table 2 and Figure 3). HPLC‐DAD chromatogram was reported in Figure 4.
TABLE 2: Phenolic composition of the extracts of the hydro‐methanol extract of A. santolina by high‐performance liquid chromatography with diode array detection (HPLC‐DAD) (mg/g extract).
| Peak No. | Phenolic compounds | Molecular Formula | RT | Calib. eq. | R2 | Hydro‐methanolic extract (mg/g extract) |
|---|---|---|---|---|---|---|
| 1 | Gallic acid | C₇H₆O₅ | 15.225 | y = 45540x ‐84708 | 0.995 | 0.31 |
| 2 | Pyrocatechol | C₆H₆O₂ | 24.625 | y = 3772.8x + 23692 | 0.9999 | 0.32 |
| 3 | p‐Hydroxy benzaldehyde | C₇H₆O₃ | 33.367 | y = 34376x + 4239.6 | 0.9996 | 0.10 |
| 4 | Epicatechin | C₁₅H₁₄O₆ | 35.278 | y = 2097.6x + 7998.2 | 0.998 | 24.54 |
| 5 | Chlorogenic acid | C₁₆H₁₈O9 | 40.094 | y = 46920x ‐ 36953 | 0.9995 | 1.55 |
| 6 | Ferulic acid | C₁₀H₁₀O₄ | 42.564 | y = 42245x + 110701 | 0.9992 | 17.56 |
| 7 | Coumarin | C₉H₆O₂ | 45.178 | y = 81802x + 153471 | 0.9968 | 0.80 |
| 8 | Rutin | C₂₇H₃₀O₁₆ | 47.527 | y = 47899x + 56096 | 0.9997 | 2.77 |
| 9 | Ellagic acid | C₁₄H₆O₈ | 50.005 | y = 235073x ‐ 7E+06 | 0.9808 | 4.37 |
| 10 | Myricetin | C₁₅H₁₀O8 | 50.368 | y = 136859x + 71185 | 0.995 | 0.83 |
| 11 | Fisetin | C₁₅H₁₀O₆ | 51.243 | y = 100784x + 16688 | 0.9984 | 2.31 |
| 12 | Luteolin | C₁₅H₁₀O₅ | 57.872 | y = 89569x ‐ 62198 | 0.9995 | 2.38 |
| 13 | Apigenin | C₁₅H₁₀O₅ | 64.071 | y = 71990x + 62472 | 0.9996 | 1.24 |
| 14 | Curcumin | C₂₁H₂₀O₆ | 72.622 | y = 15850x + 140009 | 0.9932 | 1.44 |
Abbreviations: RT, retention time; R2, regression coefficient.
Fourteen compounds were detected in A. santolina, among which epicatechin was the predominant (24.54 mg/g extract), followed by ferulic acid (17.56 mg/g), ellagic acid (4.37 mg/g), rutin (2.77 mg/g), luteolin (2.38 mg/g), and fisetin (2.31 mg/g). Other constituents were chlorogenic acid (1.55 mg/g), curcumin (1.44 mg/g), apigenin (1.24 mg/g), myricetin (0.83 mg/g), coumarin (0.80 mg/g), pyrocatechol, gallic acid, and p‐hydroxy benzaldehyde in trace amounts. Chlorogenic acid, rutin, and apigenin are found in Achillea millefolium L. s.l. [ref. 23]. Epicatechin, a flavonoid commonly found in fruits and green tea, is obtained as a pale yellow powder and exhibits antibacterial activity by disrupting bacterial membranes and inhibiting toxin production [ref. 24]. Ferulic acid, typically isolated as a white crystalline powder, enhances the efficacy of quinolone antibiotics [ref. 25]. Plant‐derived compounds like ferulic, gallic acid (a white crystalline powder), and luteolin (a yellow crystalline powder) exhibit antimicrobial effects, disrupting membrane integrity [ref. 26]. Chlorogenic acid, widely present in foods, and obtained as a greenish‐brown crystalline powder, disrupts microbial cell membranes and metabolism, leading to cell death [ref. 27]. Gallic acid, a major polyphenol, combats viral and bacterial infections [ref. 28].
Antimicrobial Activity
The rise of drug‐resistant microbes like C. albicans, A. niger, and E. coli poses a real public health challenge, particularly due to the limited effectiveness of current antibiotics and antifungal treatments. E. coli is a leading cause of urinary tract infections and produces ESBL, while A. niger and C. albicans are involved in serious opportunistic infections [ref. 29]. This underscores the urgent need for new therapeutic agents with novel mechanisms of action.
The findings of the antimicrobial activity revealed that both hydro‐methanol and petroleum ether extracts of A. santolina exhibited significant and variable efficacy against different microbial strains, including E. coli, C. albicans, and A. niger (Table 3).
TABLE 3: Antimicrobial efficacy of A. santolina petroleum ether and hydro‐methanolic extracts.
| ExtractsMicrobial strains | Petroleum ether extract | Hydro‐methanolic extract | ||
|---|---|---|---|---|
| IZ (mm) | MIC (mg/mL) | IZ (mm) | MIC (mg/mL) | |
| E. coli ATCC 25922 | R | R | 10 ± 0.94a | 1.56 |
| E. coli ATCC 25933 | 13 ± 0.12a | 3.5 | R | R |
| E. coli 01 | R | R | 12 ± 0.54b | 1.56 |
| E. coli 02 | 13 ± 0.20a | 1.56 | 10 ± 0.61a | 1.56 |
| E. coli 03 | R | R | 11 ± 0.37c | 1.56 |
| E. coli 04 | 13 ± 1.02a | 0.78 | R | R |
| C. albicans ATCC 10231 | 10 ± 0.63b | 1.56 | R | R |
| C. albicans 01 | R | R | R | R |
| C. albicans 02 | 10 ± 0.22b | 0.39 | 10 ± 1.52a | 1.56 |
Note: R: Resistant; NA: No activity; ° :30 to 40%: low activity; °°: 50 to 60%: moderate activity; °°°: >70%: excellent activity. The values in the columns, indicated by unrelated characters (a, b, c), have differences significant (p < 0.05).
The petroleum ether extract demonstrated higher antibacterial activity against several strains of E. coli, particularly E. coli ATCC 25933, and two ESBL‐producing strains E. coli 04 and E. coli 02, with inhibition diameters of 13 ± 0.20 mm and MIC ranging from 0.78 to 3.5 mg/mL. In comparison, the hydro‐methanol extract also showed notable antibacterial activity, especially against E. coli ATCC 25922, E. coli 01, E. coli 03, and E. coli 02, with inhibition diameters ranging from 10 ± 0.61 to 12 ± 0.54 mm and MIC of 1.56 mg/mL. Our findings are consistent with those of Sharifi‐rad [ref. 30], who reported that the essential oil of Achillea wilhelmsii, showed a substantial antibacterial effect against ESBL‐producing E. coli isolates, with MIC ranging from 0.5 to 4 mg/mL [ref. 30].
These findings were compared with those from studies on other Achillea species and revealed that the antibacterial activity of A. santolina was lower than that observed in Achillea species from the Egyptian Sahara [ref. 31]. Similar results from Jordan showed that A. santolina essential oils demonstrated moderate antibacterial activity on E. coli, with inhibition zones ranging from 6.0 to 16.5 mm and MIC and minimum bactericidal concentration values between 60.0 and 480.0 µg/mL [ref. 32]. Such variations are due to the difference in chemical composition and compound abundance, which can be influenced by environmental factors, including location, season, climate, plant genetics, and extraction methods [ref. 33]. Therefore, the antibacterial characteristics of A. santolina are likely related to their high concentrations of 1, 8‐cineol, and camphor previously identified in the petroleum ether extract [ref. 20].
Regarding yeasts, A. santolina extracts were effective, particularly the petroleum ether extract, which inhibited C. albicans ATCC 10231 and C. albicans 02, with inhibition zones of 10 ± 0.63 and 10 ± 0.22 mm and MICs of 1.56 and 0.39 mg/mL, respectively. The hydro‐methanolic extract also showed antifungal potential against C. albicans 02 with an inhibition zone of 10 ± 1.52 mm. Given C. albicans‘ association with various infections [ref. 4], A. santolina extracts could offer a natural antifungal treatment.
For molds, both extracts displayed antifungal activity against A. niger. The petroleum ether extract achieved moderate inhibition of 75% and 60% for A. niger 01 and A. niger 03, while the hydro‐methanolic extract showed higher inhibition, reaching 90% against A. niger 01, although both A. niger 02 and A. niger 03 were resistant to it. The antimicrobial efficacy of A. santolina extracts can be attributed to their rich chemical composition (Tables 1 and 2).
HPLC analysis showed that A. santolina contains significant amounts of epicatechin, chlorogenic acid, gallic acid, and rutin, which likely contribute to its antibacterial properties. Epicatechin, found in plants like Camellia sinensis, has demonstrated antibacterial activity [ref. 34]. Similarly, gallic acid and chlorogenic acid are known for their antibacterial effects in plants such as Achillea millefolium and burdock leaves [ref. 28]. The interaction of phenolic and flavonoid compounds with cell membrane proteins may explain varying antibacterial activity among Achillea species [ref. 35].
GC‐MS analysis of the Petroleum ether extract of A. santolina showed identified several major compounds including camphor, palmitic acid, oleic acid, nonacosane, linoleic acid, heptacosane, and stearic acid. Studies on various Achillea species have demonstrated camphor’s strong antimicrobial activity [ref. 20], particularly against C. albicans [ref. 15]. Additionally, the antimicrobial properties of palmitic acid, heptacosane, and nonacosane have been well‐documented in the literature [ref. 36]. The presence of oleic and linoleic acids further contributes to the antibacterial effects of A. santolina, suggesting that these minor compounds may act synergistically with the primary bioactive constituents, thereby enhancing the overall therapeutic potential of the plant [ref. 37].
The differences in results may be attributed to variations in chemical composition and the abundance of compounds, which are influenced by several environmental factors such as location, season, climate, plant genetics, and extraction methods [ref. 33]. Additionally, the type of extract used can significantly affect the antimicrobial properties [ref. 11], as previous research has predominantly focused on essential oils, whereas our study focused on hydro‐methanolic and petroleum ether extracts. The observed antimicrobial activity could be attributed to the higher concentrations of flavonoids and phenolic compounds in the Achillea species examined [ref. 18]. Flavonoids, as noted by Salami, inhibit microbial growth through various mechanisms, including disruption of nucleic acid synthesis, plasma membrane function, and interference with energy metabolism [ref. 35]. The antimicrobial activity against the tested microorganisms could also result from synergistic interactions among the major compounds present in the extracts [ref. 37]. Given its effectiveness against a wide range of infections, A. santolina extracts may offer a promising natural treatment for both bacterial and fungal infections, emphasizing the potential of plant‐based alternatives in combating microbial diseases.
Conclusions
This study demonstrates the significant antimicrobial potential of A. santolina from Algeria, which may be attributed to the presence of several key compounds. In the hydro‐methanolic extract, epicatechin (24.5 mg/g) and ferulic acid (17.56 mg/g) were found in notable concentrations, while the petroleum ether extract contains compounds such as camphor (19.2%), palmitic acid (15.2%), oleic acid (11.5%), and nonacosane (10.6%). Although the exact compound responsible for the antimicrobial activity remains unclear, these molecules collectively contribute to the plant’s efficacy against C. albicans and A. niger, as well as its significant antibacterial effect against ESBL‐producing E. coli isolates.
These findings support further exploration of A. santolina as a natural source for developing antimicrobial agents, especially for combating resistant pathogens. The study provides valuable insights into the chemical composition and antimicrobial properties of A. santolina, which could lead future research to the development of novel antimicrobial treatments. Future studies should focus on isolating and testing the active compounds in A. santolina to further evaluate their efficacy against a wider range of pathogens. Additionally, in vivo tests and clinical trials are important to assess the therapeutic potential of these compounds in real‐world applications. This research aims to develop plant‐based antimicrobial agents that could improve existing treatments and provide effective solutions to the growing issue of antimicrobial resistance.
Experimental
Plant Material
The aerial parts of A. santolina were collected in June 2021 from Aflou, Algeria, identified by Pr. K. Rebbas (voucher N° KR0030), washed, air‐dried, and powdered. Fifty grams were extracted by maceration with petroleum ether and 80% methanol for 24 h, then filtered, evaporated, and stored at 4°C for analysis.
HPLC Analyses (HPLC‐DAD)
The phenolic profile of the hydro‐methanolic extract of A. santolina was analyzed using a Shimadzu HPLC‐DAD (Shimadzu Corporation, Kyoto, Japan). The system included a solvent delivery unit (LC‐20AT) and a diode array detector (SPD‐M20A), the analysis was performed based on 27 standards [ref. 38] and controlled by LC‐solution software (CBM‐20A System Controller Shimadzu). Chromatographic separation was conducted on an Inertsil ODS‐3 column at 40°C. The sample (8 mg/mL in 80% methanol/water) was filtered and injected (20 µL).
The mobile phase consisted of 0.1% acetic acid in water (A) and methanol (B), with a 40‐minute gradient at a 1.5 mL/min flow rate. Detection occurred at 230–350 nm, with phenolic compounds identified by retention times and ultraviolet data, expressed as mg/g of dry extract.
Fatty Acids Analysis by GC–MS
In a 25 mL flask, 25 mg of A. santolina petroleum ether extract was combined with 2 mL of 0.5 N NaOH, sealed, and heated in a boiling water bath for 5 min. After cooling, 2 mL of BF₃–MeOH was added, and the mixture was reheated at 80°C for 3 min. Once cooled, 5 mL of saturated NaCl solution was added, followed by shaking and two n‐hexane extractions (20 mL each) [ref. 38]. For analysis, a Shimadzu GC17 AAF gas chromatograph with a DB‐1 non‐polar capillary column was used. Helium served as the carrier gas (1.2–1.3 mL/min), with injector and detector temperatures set at 250 and 270°C. The oven temperature started at 100°C for 5 min, ramped to 238°C at 3°C/min, and held for 9 min.
Fatty acid methyl esters (FAMEs) were analyzed by GC‐flame ionization detector and quantified via GC Solution software. GC‐MS analysis utilized a Varian Saturn 2100T ion trap analyzer at 70 eV, with detector, injector, and transfer line temperatures at 240, 220, and 290°C, respectively, identifying FAMEs with NIST/Wiley library matches and Supelco 37 standards [ref. 39].
Tested Microorganisms
The antimicrobial efficacy of the extracts was tested on four ESBL‐producing E. coli strains, five fungal strains (two C. albicans and three A. niger), and three reference strains from the Pasteur Institute in Algiers (E. coli ATCC 25922, E. coli ATCC 25933, and C. albicans ATCC 10231).
Antimicrobial Efficacy of A. santolina Extracts
Isolation and Characterization of ESBL‐producing E. coli and Fungal Strains
E. coli strains linked to urinary tract infections were obtained from a Skikda laboratory, representing patients of various ages: a newborn, a 5‐, a 35‐, and an 81‐year‐old. Three isolates were from females and one from a male. Strain identification and antibiograms were conducted using the Vitek 2 compact system. ESBL production was confirmed via double‐disc synergy and polymerase chain reaction (PCR) detection of the blaCTX‐M gene in all strains using standard PCR. The primer sequences and protocol were previously described [ref. 40]. All four multidrug‐resistant isolates resisted at least three antibiotic classes, including ampicillin, first‐ and third‐generation cephalosporins, ciprofloxacin, trimethoprim, and sulfamethoxazole; two also resisted amoxicillin/clavulanic acid.
Fungal Strains Isolation
Fungal strains were obtained from a private laboratory in Skikda and were isolated from various samples, including vaginal swabs, between toes, tongue, and nails. These samples were collected from two female and three male patients, aged between 30 and 60 years. The identification was conducted using both macroscopic and microscopic examinations, along with the germ tube test as described by Mackenzie [ref. 41], to confirm the presence of C. albicans.
Evaluation of the Antimicrobial Activity
Antimicrobial activity against E. coli (ESBL) and C. albicans was evaluated using the Kirby‐Bauer disk diffusion method on Mueller‐Hinton agar [ref. 42], and MIC was determined by broth microdilution (CLSI, 2018), with interpretation according to Ponce et al. A. niger growth inhibition was evaluated via disk diffusion on chloramphenicol‐supplemented Sabouraud agar, calculated using Hajji [ref. 43] formula:
where dc represents the colony diameter in the control dish, and dt is the diameter in the treatment dish. Results were interpreted according to the scale proposed by Abd‐Ellatif et al. [ref. 44].
Statistical Analysis
The in vitro results were presented as mean ± SD of three measurements and the obtained data were evaluated using Tukey’s test and one‐way analysis of variance, using SPSS software. The differences are regarded as significant when p‐values < 0.05.
Author Contributions
Chahna Renda: data curation, formal analysis, writing original draft. Sadou Nina: data curation, writing–review and editing. Bouzana Amina: Investigation, writing–review and editing. Bougouizi Amina: writing–review and editing. Haouame Imane: data curation, formal analysis. Bendif Hamdi: writing–review and editing, validation, investigation. Stefania Garzoli: writing–review and editing and supervision. Rebbas Khellaf: resources.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Medicinal Plants and Antimicrobial Activity,”. Journal of Ethnopharmacology, 2005. [DOI | PubMed]
- High Level of Colonization With Third‐generation Cephalosporin‐resistant Enterobacterales in African Community Settings, Ghana,”. Ghana Diagnostic Microbiology and Infectious Disease, 2023. [DOI | PubMed]
- Antibacterial Activity and Mechanism of Luteolin Isolated From Lophatherum Gracile Brongn. Against Multidrug‐resistant Escherichia coli ,”. Frontiers in pharmacology, 2024. [DOI | PubMed]
- 4 G. Moran , D. Coleman , and D. Sullivan , “An Introduction to the Medically Important Candida Species,” in Candida Candidiasis, eds. R. A. Calderone and C. J. Clancy (Washington, DC: ASM Press, 2011), 9–25, 10.1128/9781555817176.ch2.
- Oral Candidiasis: A Disease of Opportunity,”. Journal of Fungi, 2020. [DOI | PubMed]
- Emerging Opportunistic Yeast Infections,”. Lancet Infectious Diseases, 2011. [DOI | PubMed]
- Essential Oils as Promising Treatments for Treating Candida albicans Infections: Research Progress, Mechanisms, and Clinical Applications,”. Frontiers in Pharmacology, 2024. [DOI | PubMed]
- Antifungal Activity of Lauric Acid Derivatives Against Aspergillus niger ,”. European Food Research and Technology, 2001
- Rare and Emerging Opportunistic Fungal Pathogens: Concern for Resistance Beyond Candida albicans and Aspergillus fumigatus ,”. Journal of Clinical Microbiology, 2004. [DOI | PubMed]
- Antioxidant Effects and Ex Vivo/in Vitro Mass Reduction of Calcium Oxalate Urinary Stones of Several Extracts From Paronychia Argentea L,”. Revista Agraria Academica, 2024. [DOI]
- Antioxidant and Antimicrobial Activity of the Essential Oil and Methanol Extracts of Achillea millefolium Subsp. Millefolium Afan. (Asteraceae). millefolium Afan. (Asteraceae),”. Journal of Ethnopharmacology, 2003. [DOI | PubMed]
- Nouvelle Flore De lʼAlgérie et Des Régions Désertiques Méridionales, Paris,”. Centre National De La Recherche Scientifique, 1963
- Antimicrobial, Antiviral Activity and GC‐MS Analysis of Essential Oil Extracted From Achillea Fragrantissima Plant Growing in Sinai Peninsula, Egypt,”. Journal of Microbial & Biochemical Technology, 2014. [DOI]
- Antibacterial Activity of Extracts Prepared From Tropical and Subtropical Plants on Methicillin‐resistant Staphylococcus aureus ,”. Journal of Health Science, 2002. [DOI]
- Antimicrobial Activity of Essential Oil and Methanol Extracts of Achillea sintenisii Hub. Mor. (Asteraceae),”. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives, 2003. [DOI]
- Chemical Composition and Insecticidal Potential of Essential Oils From Egyptian Plants against Sitophilus Oryzae (L.) (Coleoptera: Curculionidae) and Tribolium Castaneum (Herbst) (Coleoptera: Tenebrionidae),”. Applied Entomology and Zoology, 2008. [DOI]
- Chemical Composition of Essential Oils Hydrodistilled From Aerial Parts of Achillea Fragrantissima (Forssk.) Sch. Bip. And Achillea Santolina L. (Asteraceae) Growing in Jordan,”. Journal of Essential Oil Bearing Plants, 2020. [DOI]
- Variation in Polyphenolic Profiles, Antioxidant and Antimicrobial Activity of Different Achillea Species as Natural Sources of Antiglycative Compounds,”. Chemistry & Biodiversity, 2018. [DOI | PubMed]
- Composition of the Essential Oil of Achillea goniocephala Boiss. Et Bal. From Turkey,”. Journal of Essential Oil Research, 2001. [DOI]
- New Chemotype of Essential Oil of Achillea Santolina L. collected From Different Regions of Algeria,”. Journal of Food Measurement and Characterization, 2018. [DOI]
- Chemical Composition and Antioxidant Activity of Essential Oil of Achillea Santolina ,”. Indian Journal of Natural Scinces (, 2020
- Comparative Study of the Essential Oils and Extracts of Achillea fragrantissima (Forssk.) Sch. Bip. And Achillea Santolina L. (Asteraceae) From Egypt,”. Die Pharmazie‐An International Journal of Pharmaceutical Sciences, 2004
- Development of an RP‐HPLC Method for the Analysis of Phenolic Compounds in Achillea millefolium L,”. Journal of Liquid Chromatography & Related Technologies, 2008. [DOI]
- Evaluation of the Antioxidant Potential of Grape Seed and Bearberry Extracts in Raw and Cooked Pork,”. Meat Science, 2007. [DOI | PubMed]
- Ferulic Acid Potentiates the Antibacterial Activity of Quinolone‐based Antibiotics Against Acinetobacter baumannii ,”. Microbial Pathogenesis, 2019. [DOI | PubMed]
- The Antibacterial Activity and Mechanism of Action of Luteolin Against Trueperella pyogenes ,”. Infection and Drug Resistance, 2020. [DOI | PubMed]
- The Antibacterial Activity and Mechanism of Chlorogenic Acid Against Foodborne Pathogen Pseudomonas aeruginosa ,”. Foodborne Pathogens and Disease, 2019. [DOI | PubMed]
- An Update on the Potential Mechanism of Gallic Acid as an Antibacterial and Anticancer Agent,”. Food Science & Nutrition, 2023. [DOI | PubMed]
- Emerging Challenges in Antimicrobial Resistance: Implications for Pathogenic Microorganisms, Novel Antibiotics, and Their Impact on Sustainability,”. Frontiers in Microbiology, 2024. [DOI | PubMed]
- Antibacterial Activities of Essential Oils From Iranian Medicinal Plants on Extended‐spectrum β‐lactamase‐producing Escherichia coli ,”. Cellular and Molecular Biology, 2016
- Chemical Composition and Antibacterial Activity of Essential Oils and Major Fractions of Four Achillea Species and Their Nanoemulsions against Foodborne Bacteria,”. LWT—Food Science and Technology, 2016. [DOI]
- Effect of Ethnomedicinal Plants Used in Folklore Medicine in Jordan as Antibiotic Resistant Inhibitors on Escherichia coli ,”. BMC Complementary and Alternative Medicine, 2010. [DOI | PubMed]
- Phytochemical Analysis by LC MS/MS and in Vitro Antioxidant Activity of the Algerian Endemic Plant Dianthus sylvestris Subsp,”. Aristidis (Batt) Greuter & Burdet Global NEST Journal, 2023
- Investigation of the Antibacterial Activity of 3‐ O ‐octanoyl‐(‐)‐epicatechin,”. Journal of Applied Microbiology, 2008. [DOI | PubMed]
- Inhibitory Effect of Different Fennel (Foeniculum vulgare) Samples and Their Phenolic Ompounds on Formation of Advanced Glycation Products and Comparison of Antimicrobial and Antioxidant Activities,”. Food Chemistry, 2016. [DOI | PubMed]
- Chemical Composition of Essential Oil/Volatiles and Fatty Acids, and Antimicrobial Evaluation of Two Endemic Achillea (Asteraceae) Species: Achillea Schischkinii Sosn. And Achillea Teretifolia Willd,”. Journal of Research in Pharmacy, 2024. [DOI]
- Anti‐Candida albicans Activity of Some Iranian Plants Used in Traditional Medicine,”. Journal De Mycologie Médicale, 2009. [DOI]
- Rapid and Easy Method for Simultaneous Measurement of Widespread 27 Compounds in Natural Products and Foods,”. Journal of Chemical Metrology, 2020. [DOI]
- Comprehensive Identification and Quantification of Chlorogenic Acids in Sweet Cherry by Tandem Mass Spectrometry Techniques,”. Journal of Food Composition and Analysis, 2018. [DOI]
- Prevalence and Characterization of Carbapenem‐Resistant Enterobacterales Among Inpatients and Outpatients in Skikda, Algeria,”. The Journal of Infection in Developing Countries, 2024. [DOI | PubMed]
- Serum Tube Identification of Candida albicans ,”. Journal of Clinical Pathology, 1962. [DOI | PubMed]
- Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. 11th ed.,. 2018
- Evaluation in Vitro de L’activité Antifongique de Quatre Plantes Médicinales Marocaines sur Cinq Champignons Phytopathogènes,”. Revue Marocaine De Protection Des Plantes (, 2016
- Promising Antifungal Effect of Some Folkloric Medicinal Plants Collected From El‐Hammam Habitat, Egypt Against Dangerous Pathogenic and Toxinogenic Fungi,”. ARPN Journal of Agricultural and Biological Science, 2011
