Therapeutic approach to fibromyalgia: a consensus statement on pharmacological and non-pharmacological treatment from the neuropathic pain special interest group of the Italian neurological society
Abstract
Background:
Although fibromyalgia is a disabling disease, there is no targeted therapy for specific neurotransmitters or inflammatory mediators. Our aim was to provide neurologists with practical guidance for the management of these difficult patients based on a critical, narrative and non-systematic review of randomized controlled trials (RCTs) from the last 10 years.
Methods:
The members of the Special Interest Group Neuropathic Pain of the Italian Neurological Society evaluated the randomized controlled trials (RCTs) of the last 10 years and answered questions that allow a consensus on the main pharmacological and non-pharmacological approaches.
Results:
The neuropathic pain working group agreed on prescribing antiepileptic drugs or antidepressants in the case of comorbidities with anxiety and depression. As a second choice, experts have agreed on the association of antiepileptics and antidepressants, while they disagree with the use of opioids. Medical cannabis and nutraceuticals are promising new treatment options, although more data is needed to prove their efficacy. The neurologists agreed in suggesting physical activity at the first visit, particularly aerobic and strength training. As a second choice, they considered a cognitive behavioral therapy approach to be useful.
Conclusions:
Pharmacologic treatment with antiepileptic drugs and antidepressants in patients with co-occurring anxiety and depression, as well as an early nonpharmacologic approach based primarily on physical activity, may be a useful indication in contemporary neurology clinical practice. Non-pharmacological options, such as cognitive behavioral therapy and non-invasive brain stimulation NIBS, could improve evidence of efficacy and lead to relevant improvement in FM-related disability.
Article type: Research Article
Keywords: Fibromyalgia, Treatments, Recommendations, Pharmacological therapy, Non-pharmacological therapy
Affiliations: Fondazione IRCCS Carlo Besta, Milan, Italy; https://ror.org/02be6w209grid.7841.aDepartment of Human Neuroscience, Sapienza University, Rome, Italy; https://ror.org/05fz2yc38grid.414405.00000 0004 1784 5501Clinica Neurologica Bellaria Hospital, Bologna, Italy; https://ror.org/02p77k626grid.6530.00000 0001 2300 0941Child Neurology and Psychiatric Unit, Tor Vergata University, Rome, Italy; https://ror.org/039bp8j42grid.5611.30000 0004 1763 1124Department of Neurosciences, Biomedicine and Movement Sciences, University of Verona, Verona, Italy; https://ror.org/00mc77d93grid.511455.1Skin Biopsy Laboratory, Department of Neurology, Istituti Clinici Scientifici Maugeri IRCCS, Telese Terme, Italy; https://ror.org/05290cv24grid.4691.a0000 0001 0790 385XDepartment of Neurosciences, Reproductive Sciences and Odontostomatology, University Federico II of Naples, 80100 Naples, Italy; https://ror.org/027ynra39grid.7644.10000 0001 0120 3326DiBrain Department, Bari Aldo Moro University, Bari, Italy
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s10072-025-08048-3 | PubMed: 39982626 | PMC: PMC12003471
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.1 MB)
Introduction
Fibromyalgia (FM) is a disabling disease characterized by chronic widespread pain, fatigue, sleep disturbances, cognitive failure, and headaches. While the diagnostic criteria are well established [ref. 1], the pathophysiology is still unknown. The central and peripheral nervous systems have been found to be affected, with concomitant central sensitization and small fiber pathology [ref. 2], justifying the clinical interest of neurologists [ref. 3]. Unfortunately, pharmacological therapies of FM are often unsuccessful, while non-pharmacological treatments are the first to be recommended [ref. 4]. The heterogeneity of the clinical pictures of FM may suggest that different pathophysiological mechanisms, including immune and inflammatory factors, genetic predisposition to small fiber impairments, and mechanisms of central sensitization phenomena, together with psychiatric comorbidities, contribute to the clinical picture of FM [ref. 5].
Currently, there is no targeted therapy for specific neurotransmitters or inflammatory mediators, and evidence is limited as there are few randomized controlled trials (RCTs) and these often include small populations and sample sizes. In addition, there is little evidence for the effectiveness of non-pharmacological treatments [ref. 6].
In this scenario, pending the best possible clarification of the understanding of the different FM phenotypes based on specific pathophysiologic features, we sought to reach a consensus among neurologists on the most appropriate treatment for FM based on a review of recent RCTs in the field of pharmacologic and non-pharmacologic treatments. Our aim was to provide clinicians with practical guidance for the treatment of these difficult patients based on a critical and narrative review of RCTs from the last 10 years.
Methods
This was a consensus based on a narrative-systematic review of RCTs on pharmacological and non-pharmacological approaches to FM.
To this end, the active members of the Neuropathic Pain Special Interest Group of the Italian Neurological Society (NPG), who recently contributed to the diagnostic guideline for fibromyalgia [ref. 3], reviewed the most recent RCTs and answered questions that yielded a consensus on the main pharmacologic and non-pharmacologic approaches. The group of neurologists was also supported on certain arguments, such as physical activity, by external experts who were not involved in the final consensus.
The members of the NPG analyzed the RCTs of the last 10 years. For pharmacological therapies, the NPG members included antidepressants, antiepileptics and opioids and also included a chapter on cannabinoids and dietary supplements. For non-pharmacological therapies, they included acupuncture, cognitive behavioral therapy, physical activity and non-invasive brain stimulation.
NPG members searched the Pubmed database from 2014 to 2024 using specific keywords for selected pharmacological and non-pharmacological treatments (Table 1) with the following filters: Clinical trial, Phase I, II, III, Randomized controlled trial. For pharmacologic treatments, antidepressants, antiepileptics, opioids, cannabinoids, supplements, and nutrients were entered into Pubmed along with the term fibromyalgia. They selected only original studies and excluded reviews. NPG members included studies with placebos and/or comparisons with different active treatments.
Table 1: Search strategies for pharmacological and non-pharmacological treatments
| Type of treatment | Search string | Retrieved records |
|---|---|---|
| Pharmacological treatments | ||
| Antidepressants | (fibromyalgia) AND ("Antidepressive Agents" [Pharmacological Action] OR "Antidepressive Agents, Second-Generation" [Pharmacological Action] OR "Antidepressive Agents, Tricyclic" [Pharmacological Action]) | 40 |
| Antiepileptic drugs | (fibromyalgia) AND ("Anticonvulsants"[Mesh] OR "Anticonvulsants" [Pharmacological Action] OR “antiepileptic drugs”)* | 33 |
| Cannabinoids | (fibromyalgia) AND (cannabis OR cannabinol OR cannabinoid* OR cannabidiol OR bhang OR hashish OR hemp OR marihuana OR marijuana OR nabilone OR cesamet OR tetrahydrocannabinol OR THC OR dronabinol OR levonantradol OR nabiximols OR palmidrol OR tetrahydrocannabinolic OR sativex OR endocannabinoid*) | 10 |
| Muscle relaxants | (fibromyalgia) AND (muscle relaxants or cyclobenzaprine or tizanidine) | 0 |
| Nutraceutics | (fibromyalgia) AND (nutraceutics) | 7 |
| Opioids | fibromyalgia) AND (opioids or tramadol or tapentadol or naltrexone or morphine or naloxone) | 2 |
| Non-pharmacological treatments | ||
| Physical activity | (fibromyalgia) AND (exercise OR “physical activity”) | 129 |
| Acupuncture | (fibromyalgia) AND (acupuncture OR electroacupuncture OR acupuncture therapy) | 23 |
| Cognitive Behavioral Therapy | (fibromyalgia) AND (cognitive behavioral therapy OR CBT OR “cognitive behavioural therapy” OR acceptance and commitment therapy OR ACT OR mindfulness OR cognitive restructuring) | 77 |
| Non-Invasive Brain Stimulation | (fibromyalgia) AND ("non invasive brain stimulation" OR "non-invasive brain stimulation" OR NIBS OR transcranial magnetic stimulation OR TMS OR "repetitive transcranial magnetic stimulation" OR rTMS OR "theta burst stimulation" OR TBS OR transcranial direct current stimulation OR tDCS OR transcranial alternating current stimulation OR tACS OR transcranial random noise stimulation OR tRNS OR "deep transcranial magnetic stimulation" OR "deep TMS" OR neuromodulation) NOT (neurotransmitter agents OR deep brain stimulation OR DBS) | 50 |
| Diet | (fibromyalgia) AND (diet) | 10 |
The search was run on PubMed; filters: Clinical Study, Clinical Trial, Clinical Trial, Phase II, Clinical Trial, Phase III, Clinical Trial, Phase IV, Randomized Controlled Trial, from 2014—2024
For non-pharmacological therapies, NPG members included acupuncture, cognitive behavioral therapy, physical activity, diet, and non-invasive brain stimulation (NIBS), using the same criteria as the search for pharmacological RCTs.
The main outcomes considered were pain and disability scores and the effects on other features of FM, such as sleep and fatigue.
The NPG reviewed the studies and considered those it considered most relevant. In addition to evidence of efficacy, the group also considered safety. The questions were formulated based on the reported review and a realistic consideration of the options within the Italian healthcare system and readily available services. Among the non-pharmacological approaches, the procedures that require specialized personnel and equipment but are not reimbursed within the national healthcare system were indicated as the second choice.
The neurologists then voted on a scale from 0, ‘strongly disagree’, to 10, ‘strongly agree’, on their level of agreement with the recommendation.
Scores from 7 to 10 were considered agreement, 5–6 for an indeterminate opinion, 0–4 for disagreement.
We applied the chi-square test to assess the distribution of opinions, and the significant prevalence of a choice was scored as definite agreement or disagreement. JMP software version 17 was used.
Results
Table 1 contains the results of the search strategy. The following section summarises the relevant studies (Tables 1, 2 and 3).
Table 2: Randomized controlled trials testing the effect of pharmacological and nutraceutical treatments in fibromyalgia (FM)
| Reference | Active treatment | Comparator | Sample size | Outcome measures | Findings | Adverse events (active treatment) |
|---|---|---|---|---|---|---|
| Antidepressants | ||||||
| Upadhyaya et al., 2019 [ref. 7] | Duloxetine 30/60 mg | Placebo | 184 (juvenile FM) | BPI average pain severity | Negative | Nausea 25.3%, vomiting 15.4%, headache 14.3% |
| Bidari et al., 2019 [ref. 8] | Duloxetine 30–60 mg | Pregabalin 75–150 mg | 99 | WPI, BDI-2 | Positive | Nausea 34.3%, constipation 31.4%, headache 22.9%, drowsiness 20%, dry mouth 17.1%, dizziness 17.1%, insomnia: 17.1% |
| Pickering et al., 2018 [ref. 9] | Milnacipran 100 mg | Placebo | 54 | Status of CPM | Negative | Gastrointestinal disorders 28.4%, nervous system symptoms 14.7% |
| Ahmed et al., 2016 [ref. 10] | Milnacipran 100 mg | Placebo | 19 | Polysomnographic measures, BPI, FIQ | Positive for pain | Nausea/vomiting 22.2%, headache 16.7%, abdominal pain 11.1%, constipation 11.1%, sinusitis 11.8%, hot flush 11.8% |
| Miki et al., 2016 [ref. 11] | Mirtazapine 30 mg | Placebo | 422 | NRS | Positive | Somnolence 32.1%, weight gain 17.7%, increased appetite 11.6% |
| Murakami et al., 2015 [ref. 12] | Duloxetine 60 mg | Placebo | 393 | BPI, average pain score | Negative | Somnolence 26.3%, nausea 21.6%, constipation 14.9%, dizziness 5.7%, liver injury in 1 patient |
| Staud et al., 2015 [ref. 13] | Milnacipran 100 mg | Placebo | 46 | VAS, mechanical and heat pain sensitivity | Negative | Gastrointestinal disorders 10.9% |
| Leombruni et al., 2015 [ref. 14] | Duloxetine 60 mg | acetyl L-carnitine 1500 mg | 65 | VAS, MADRS, HADS-D | Positive in both arms | Nausea, anxiety, insomnia, and diarrhea in 8 patients |
| Combination of antidepressants + anticonvulsants | ||||||
| Abdel Fattah et al., 2020 [ref. 15] | Milnacipran 100 mg + pregabalin 300 mg | Pregabalin 300 mg | 58 | FIQ, VAS, Leeds Sleep Evaluation Questionnaire | Negative (combination treatment not superior to pregabalin) | Disturbed sleep pattern 26.9%, dizziness and drowsiness 19.2%, gastrointestinal disorders 15.4% |
| Ramzy et al., 2017 [ref. 16] | Paroxetine 25 mg + pregabalin 75 mg | Pregabalin 75 mg + amitriptyline 25 mgPregabalin 75 mg + venlafaxine 75 mg | 75 | SSS-8, CESDS | Positive | Dry mouth 7.7%, abnormal taste 7.7%, weight gain 11.5% |
| Gilron et al., 2016 [ref. 17] | Duloxetine 120 mg + pregabalin 450 mg | PlaceboPregabalin 450 mgDuloxetine 120 mg | 41 | NRS | Positive for active vs. placebo and pregabalin/duloxetine | Fatigue 29.4%, drowsiness 26.5%, dry mouth 23.5%, constipation 11.8%, insomnia 11.8%, headache 11.8% |
| Anticonvulsants (gabapentinoids) | ||||||
| Karamanlioglu et al., 2021 [ref. 18] | Pregabalin + exercise | Exercise | 40 | pain VAS; PPT; DN4; SF36 | Positive with both treatments. Pregabaline + exercise is more effective than exercise alone | Pregabalin: dizziness (82.4%), somnolence (82.4%), foot edema(17.6%), weight gain (5.9%), and constipation (5.9%) |
| Arnold et al., 2019 [ref. 19] | Mirogabalin (15–30 mg) | Pregabalin 300 mgPlacebo | 3864 | ADPS; PGIC; FIQ | Negative for mirogabalin (both doses) vs placebo and pregabalinPositive for pregabalin vs placebo | no unexpected adverse events |
| Arnold et al., 2016 [ref. 20] | Pregabalin 75–450 mg/day | Placebo | 107(adolescents) | Pain NRS (primary); PGIC, ADPS, pain score change at week 15; 30% or 50% improvementin mean pain score; sleep quality NRS; FIQ-C | Negative for primary outcome measure, positive for secondary outcome measures | Dizziness (29.6%); Nausea (22.2%); Headache (18.5%); Weight increase (16.7%); Fatigue (14.8%) |
| Arnold et al., 2015 [ref. 21] | Pregabalin (300- 450 mg) | Placebo | 197 | Pain NRS (primary outcome measure); measures of anxiety, depression, patient function, and sleep | Positive | Dizziness (28.2%); Somnolence (19.9%); Constipation (10.5%); Nausea (9.4%) |
| Arnold et al., 2014 [ref. 22] | Pregabalin (165- 495 mg) | Placebo | 121 | LTR (primary outcome measure); pain NRS, global assessment, functional status, tiredness/fatigue, and sleep | Positive for primary endpoint | PGB (placebo)Dizziness 20.6% (20.7%)Somnolence 9.5% (10.3%)Headache 4.8% (3.4%)Fatigue 7.9% (1.7%)Nausea 7.9% (1.7%)Peripheral edema 17.5% (8.6%)Weight increase 9.5% (6.9%)Vision blurred 6.3% (0.0%)Dry mouth 4.8% (10.3%)Insomnia 11.1% (1.7%) |
| Nasser K et al., 2014 [ref. 23] | Pregabalin 300 mg once daily | Pregabalin 150 mg twice daily | 177 | Daily pain NRS (primary outcome measure). Weekly VASfor pain, fatigue,trouble sleeping, and waking unrefreshed. MSS, FIQ-R, PGIC | no significant difference between either treatment option | No differences in adverse events incidence or severity |
| Sawaddiruk et al., 2019 [ref. 24] | Pregabalin 150 mg + CoQ10 300 mg / day | Pregabalin 150 mg / day + placebo | 11 | PPT, Pain VAS, FIQ, HAMDT-A | Improvement of pain and anxiety with the association | Not reported |
| Cannabinoids | ||||||
| van de Donk et al., 2020 [ref. 25] | Different combinations of CBD and THC (Bedrocan, Bediol, Bedrolite) | Placebo | 20 | Pain scores, PPT, EPT, CBD and THC plasma concentration | Bediol: 30% decrease in pain scores vs. placebo | Cough |
| Chaves et al., 2020 [ref. 26] | THC-rich cannabis oil | Placebo | 17 | FIQ | Positive | Somnolence 87.5%, dizziness 25%, mouth dryness 25%, |
| Opioids | ||||||
| Bested et al., 2023 [ref. 27] | Low-dose naltrexone (4.5 mg) | Placebo | 58 | FIQR, SPIR, BPI-SF, DSIS, HADS, PD-Q, PCS, QST, pharmacokinetics | Negative | Headache, fatigue, nausea, dizziness |
| Nutraceutics | ||||||
| Leombruni et al., 2015 [ref. 14] | ALC 1500 mg | DLX 30/60 mg | 65 | MADRS, HADS, VAS, DT, SF36 and CGI-I | Positive with both treatments, except for HADS anxiety subscale | Only in the DLX group (8 pts) nausea, anxiety, insomnia, diarrhoea |
| Salaffi et al., 2023 [ref. 28] | DLX 60 mg + PGB 150 mg + PEA 1200 mg + ALC 1000 mg | DLX 60 mg + PGB 150 mg | 142 | WPI, FIQR, FASmod | Positive, with greater efficacy in the add-on therapy with PEA and ALC | Not indicated |
| Gilron et al., 2023 [ref. 29] | ALA from 300 mg/day + PGB from75 mg/day, titrated to MTD | PGB (starting at a dose of 75 mg once daily and titrated to individual maximally tolerated dose) | 54 | 0–10 PPI, FIQ, SF-36, MOS-SS, BPI, BDI-II, BAI, SF-MPQ. acetaminophen consumption | Absent additive benefit of combining ALA with PGB | Not frequent with combination therapy |
| Esposito et al., 2021 [ref. 30] | ALA 400 mg-800 mg | Placebo | 210 patients, only 12 with FM | 0–10 NRS and VAS, Glycemia, CRE, SGPT, SGOT | Positive for pain, no effects on laboratory tests | No AEs |
| Gilron et al., 2021 [ref. 31] | ALA 300 mg-1800 mg; titrated to MTD- on average 1663 mg/day | Placebo | 27 | 0–10 NRS,, FIQ, MOS-SS, PGICS, BPI, BPI-II, SF-36, acetaminophen consumption | Negative | No statistically significant differences between placebo and ALADuring titration: nausea (16%), headache (12%); at MTD: depression (4%), muscle spasms (4%). During taper/washout: headache (8%) |
| Schweiger et al., 2020 [ref. 32] | Migratens (ALA, CoQ10, vitamin D, Mg, TRY) | Acupunture | 60 | 0–10 VAS, FIQ-R, FSS | Positive | Migratens: gastrointestinal side effects (15%) |
| Martínez-Rodríguez et al. 2020 [ref. 33] | TRY and Mg-enriched Mediterranean die | Standard Mediterranean diet | 22 | psychological variablesPittsburgh SleepQuality Index4 months | Improvement of sleep duration and efficiency in TRY Mg group, anxiety and depression | None |
FIQ: Fibromyalgia Impact Questionnaire; VAS: Visual Analogue Scale; NRS: Numerical Rating Scale; BPI: Brief Pain Inventory; WPI: Widespread Pain Index; BDI-2: Beck Depression Inventory-2; CPM: conditioned pain modulation; SSS-8: Somatic Symptoms Scale-8; CESDS: Center for Epidemiological Studies Depression Scale; MARDS: Montgomery Asberg Depression Rating Scale; HADS-D: Depression subscale of the Hospital Anxiety and Depression Scale; PPT: Pressure Pain Threshold; DN4: Questionnaire Douleur neuropathique 4; SF-36: Short Form Health Survey 36; ADPS: Average Daily Pain Score; PGIC: Patient Global Impression of Change; EPT: electrical pain threshold; THC: tetrahydrocannabinol; CBD: cannabidiol; FIQR: Fibromyalgia Impact Questionnaire Revised; SPIR: Summed Pain Intensity Ratings; SOWS: Subjective Opiate Withdrawal Scale; BPI-SF: Brief Pain Inventory-Short Form; DSIS: Daily Sleep Interference Scale; HADS: Hospital Anxiety and Depression Scale; PD-Q: PainDETECT Questionnaire; PCS: Pain Catastrophizing Scale
ALC: Acetyl-L-carnitine; PEA: Palmitoylethanolamide; DLX: duloxetine; PGB: Pregabalin; ALA: alpha-lipoic acid; MADRS: Montgomery Asberg Depression Rating Scale; HADS: Hospital Anxiety and Depression Scale; VAS: Visual Analogue Scale; DT: Distress Thermometer; SF36: 36-item Short-Form Health Survey; FASmod: Modified Fibromyalgia Assessment Status; PPI: Present Pain Intensity (0–10); FIQ: Fibromyalgia Impact Questionnaire; MOS-SS: Medical Outcome Study Sleep Scale; CGI-I: Clinical Global Impression-Improvement; WPI: Wispread Pain Index; FIQR: revised Fibromyalgia Impact Questionnaire; FIQ: Fibromyalgia Impact Questionnaire; MTD: Maximal Tolerated Dose BPI: Brief Pain Inventory; BDI-II: Beck Depression Inventory-II, BAI: Beck Anxiety Inventory; SF-MPQ: Revised Short McGill Pain
Questionnaire Version; AE: advers effects; PGIC-S: Patients Global Impression of Change Scale; TRY: tryptophan; Mg: magnesium; CRE: creatinine level; SGPT: serum glutamic pyruvic transaminase; SGOT: serum glutamic-oxaloacetic transaminase; CoQ10: Coenzyme Q10, DASS: Depression Anxiety Stress Scales; 11-NRS: 11-point pain numerical rating scale; PSQI: Pittsburgh Sleep Quality Index; SF-12: 12-item Short Form Survey; FSS: Fatigue Severity Scale; PCS: Pain Catastrophizing Scale; EPICES: French questionnaire Evaluation de la Précarité et des Inégalités de Santé dans les Centres d’Examens de Santé
Table 3: Randomized controlled trials testing the effect of non-pharmacological treatments in fibromyalgia (FM)
| Reference | Active treatment | Comparator | Sample size | Outcome measures | Findings | Adverse events (active treatment) |
|---|---|---|---|---|---|---|
| Cognitive Behavioral Therapy | ||||||
| Luciano et al., 2014 [ref. 34] | ACT | RPT, WL | 156 | FIQ | Positive (compared to both control arms) | – |
| Simister et al., 2018 [ref. 35] | ACT + TAU | TAU | 66 | FIQ-R | Positive | – |
| Laura Andres-Rodriguez et al., 2019 [ref. 36] | MBSR + TAU | TAU | 70 | FIQ-R | Positive | – |
| Perez-Aranda et al., 2019 [ref. 37] | MBSR + TAU | FibroQoL + TAU, TAU | 225 | FIQ-R | Positive (compared to both control arms) | – |
| Acupuncture | ||||||
| Casanueva et al., 2014 [ref. 38] | Manual/needles as the TCM (6 sessions, 1 h/session over 6 weeks) | TAU | 100 | Myalgic score | Positive | Increased pain, physical discomfort |
| Vas et al., 2016 [ref. 39] | Manual/needles as the TCM (10 sessions, 20 min/session over 10 weeks) | Non-penetrating needles | 162 | VAS (pain) | Positive | Aggravation of FM symptoms (2.6%), headache (0.5%), post-acupuncture pain (1.4%)/bruising (2.6%), post-acupuncture vagal symptoms (0.7%) |
| Uğurlu et al., 2017 [ref. 40] | Manual/needles as the TCM (12 sessions, 30 min/session gradually decreasing over time) | Park sham device* | 50 | VAS (pain) | Positive (at FU) | NR |
| Zucker et al., 2017 [ref. 41] | Manual/needles as the TCM (8 sessions, 20 min/session gradually increasing over time) | Needles on non-traditional acupuncture points | 73 | VAS (pain) | Positive (only for patients with higher PPTs) | NR |
| Karatay et al., 2018 [ref. 42] | Manual/needles as the TCM (8 sessions, 30 min/session over 4 weeks) | Sham/simulated acupuncture§ | 75 | VAS (pain) | Positive | Discomfort/bruising at the sites of needle insertion |
| Mist & Jones, 2018 [ref. 43] | Manual/needles as the TCM (20 sessions, 45 min/session over 10 weeks) | Education | 30 | FIQ-R | Positive | Bruising, transient dizziness |
| Garrido-Ardila et al., 2020 [ref. 44] | Manual/needles as the TCM (10 sessions, 20 min/session over 5 weeks) | FKT (active control), no trt | 103 | 10-MWT | Negative | None |
| Schweiger et al., 2020 [ref. 32] | Manual/needles as the TCM (10 sessions, 30 min/session) | Nutritional supplement (Migratens® treatment) | 60 | VAS (pain) | Positive | None |
| Mawla et al., 2021 [ref. 45] | Electric acupuncture (8 sessions, 25 min/session over 4 weeks) | Mock laser acupuncture | 72 | BPI | Positive | NR |
| Physical activity | ||||||
| Larsson et al., 2015 [ref. 46] | Resistance exercise program (60 min sessions over 15 weeks) | Active control group | 130 | Isometric knee-extension force | Positive | – |
| Collado-Mateo et al., 2017 [ref. 47] | Exergame (postural, coordination, aerobic, strength, mobility; 60 min sessions over 8 weeks) | Non-exercise group | 83 | FIQ | Positive | – |
| Wang et al., 2018 [ref. 48] | Four Tai Chi groups (60 min sessions over 12–24 weeks) | Aerobic group | 226 | FIQ-R | Positive (at FU) | – |
| Andrade et al., 2019 [ref. 49] | Aquatic physical training (60 min sessions over 16 weeks) | Non-exercise group | 54 | FIQ | Positive | – |
| Izquierdo-Alventosa et al., 2020 [ref. 50] | Low-intensity exercise (endurance training, coordination; 60 min sessions over 8 weeks) | Non-exercise group | 32 | PCS | Positive | – |
| Serrat et al., 2021 [ref. 51] | Multicomponent treatment (pain neuroscience education, therapeutic exercise, CBT, mindfulness + pharmacological trt; 60 min sessions over 12 weeks) | Pharmacological trt | 272 | FIQ-R | Positive | – |
| Gentile et al., 2023 [ref. 52] | Supervised home-based multicomponent PA intervention focused on aerobic and resistance training | Non-supervised aerobic exercise | 34 | Fibromyalgia-linked invalidity questionnaireSkin biopsy | Positive | – |
| Diet | ||||||
| Vellisca & Latorre, 2014 [ref. 53] | Dietary elimination of MSG and aspartame | WL (no diet) | 72 | Pain intensity (seven-point NRS) | Negative | None |
| Slim et al., 2017 [ref. 54] | Gluten free diet | Hypocaloric Diet | 75 | Gluten sensitivity symptoms,FIQ,anxiety,depression, impression of improvement | Positive for both diets | None |
| Roman et al., 2018 [ref. 55] | Multispecies probiotics | Placebo | 40 | VAS,FIQ,anxiety, depression, cognitive evaluation | Improvement of s improved impulsivity and decision-making i | None |
| Mauro Martin et al., 2019 [ref. 56] | Olive tree-based supplement + IGUBAC diet | No diet | 31 | CGPS, PCS, FSS, FIS, anthropometric measures, blood biochemical analyses | Positive (CGPS within active arm; PCS, FSS for both arms) | None |
| Pagliai et al., 2020 [ref. 57] | Khorasan Wheat-Based Replacement | control wheat products | 20 | FIQ, WPI, SS | Positive for all scores | None |
| Martínez-Rodríguez et al., 2020 [ref. 33] | TRY + MG Mediterranean diet | Standard Mediterranean diet | 22 | PSQI, BSQ, STAI-TA, POMS-29, EAT-26 | Positive (STAI, BSQ, POMS-29, EAT-26) | None |
| Casini et al., 2024 [ref. 58] | Personalized Mediterranean diet | Generic personalized balanced diet | 84 | FIQ, fatigue, anxiety, depression, ten-point NRS | Positive (FIQ, fatigue, anxiety, depression within active arm) | None |
| NIBS – tDCS | ||||||
| Fagerlund et al., 2015 [ref. 59] | tDCS (5 sessions, 20 min/session, 2 mA on M1) | Sham | 48 | NRS (pain, stress) | Positive | Acute mood change (0.84%), skin redness (56.30%), sleepiness (55.46%), tingling (53.78%) |
| Silva et al., 2017 [ref. 60] | tDCS (20 min single session, 1 mA on DLPFC) | Sham | 40 | ANT | Positive | NR |
| Santos et al., 2018 [ref. 61] | tDCS (8 sessions, 20 min/session, DLPFC) | Sham | 40 | Memory (RAVLT) | Positive | NR |
| Brietzke et al. 2019 [ref. 62] | tDCS (60 sessions, 30 min/session, 2 mA on l-DLPFC) | Sham | 20 | VAS (pain) | Positive | NR |
| Castelo-Branco et al., 2019 [ref. 63] | tDCS (16 sessions) + aerobic exercise | Sham | 148 | Pain | Positive | |
| Matias et al., 2021 [ref. 64] | tDCS (5 sessions, 20 min/session over 5 days on M1) + functional exercise (over 8 weeks) | Sham | 31 | VAS (pain, anxiety), 6-MWT, BDI, FIQ | Negative | Headache, tingling, dizziness, nausea (42.85%) |
| Arroyo-Fernández et al., 2022 [ref. 65] | tDCS (5 sessions) + exercise | Sham + exercise, no intervention | 120 | Pain, FIQ, BDI | Positive | NR |
| Samartin et al., 2022 [ref. 66] | tDCS (15 sessions, 20 min/session on M1 or DLPFC or OIC) | Sham | 130 | SF-36, FIQ-R | Negative | NR |
| Samartin et al., 2022 [ref. 67] | tDCS (15 sessions, 20 min/session on M1 or DLPFC or OIC) | Sham | 130 | HADS | Positive | Tickling (42.9%), itching (46.4%), burning (28.6%) |
| Caumo et al., 2022 [ref. 68] | tDCS (20 sessions, 20 min/session on DLPFC) | Sham | 48 | PCS | Positive | NR |
| Loreti et al., 2023 [ref. 69] | tDCS (10 sessions, 26 min/session on M1) | Sham | 35 | VAS (pain) | Positive | Headache (6.47%), local tingling (5.29%), local redness (12.94%), somnolence (0.58%), local itch (2.35%), neck ache (1.76%) |
| Schein et al., 2023 [ref. 70] | tDCS (20 min single session, anodal l-DLPFC, cathodal r-DLPFC) | HAS | 18 | MEP | Positive | NR |
| NIBS – TMS | ||||||
| Boyer et al., 2014 [ref. 71] | HF rTMS (10 sessions on l-M1) | Sham | 38 | FIQ | Positive | None |
| Fitzgibbon et al., 2018 [ref. 72] | HF rTMS (20 sessions on l-DLPFC) | Sham | 26 | SF-MPQ, BPI, NRS (pain), MFI-20 | Positive (MFI-20) | Site discomfort (15.4%), headache (15.4%), nausea (3.8%), dizziness (3.8%), other (3.8%) |
| Altas et al., 2019 [ref. 73] | HF rTMS (15 sessions on l-M1 or l-DLPFC) | Sham | 30 | VAS, FIQ, FSS, SF-36, BDI | Positive | NR |
| Cheng et al., 2019 [ref. 74] | HF rTMS (10 sessions on l-DLPFC) | Sham | 20 | VAS (pain) | Positive | None |
| Tanwar et al., 2020 [ref. 75] | LF rTMS (20 sessions on r-DLPFC) | Sham | 90 | NRS (pain) | Positive | Headache (2%) |
| Bilir et al., 2021 [ref. 76] | HF rTMS (10 sessions on l-DLPFC) | Sham | 20 | VAS, FSS, HADS | Negative | None |
| Izquierdo- Alventosa et al., 2021 [ref. 77] | HF rTMS (10 sessions on l-DLPFC) | Sham, physical exercise | 49 | VAS (pain) | Positive | NR |
| Lacroix et al., 2021 [ref. 78] | HF rTMS (15 sessions on l-M1) | Sham | 78 | VAS, PGIC | Positive | None |
| Argaman et al., 2022 [ref. 79] | HF rTMS (10 sessions on M1) | Sham | 27 | BPI, MPQ, FIQ, SF-36, STAI, BDI | Positive | NR |
| Pareja et al., 2022 [ref. 80] | rTMS (8 sessions) + pharmacological trt | Pharmacological trt | 560 | FIQ, WPI, SSS | Positive | NR |
| NIBS – combined | ||||||
| Forogh et al., 2021 [ref. 77] | rTMS or tDCS (3 sessions, 20 min/session of rTMS or tDCS on DLPFC) | None | 30 | VAS (pain), FIQ-R, DASS-21 | Positive (rTMS, VAS) | Mild, transient headache (rTMS) |
ACT: Acceptance and Commitment Therapy; ANT: Attention Network Test; BDI: Beck Depression Inventory; BPI: Brief Pain Inventory; BSQ: Body Shape Questionnaire; C2: occipital nerve area; CGPS: Chronic Pain Grade Scale; ctrl: control; DASS-21: Depression Anxiety Stress Scale-21 Item; DLPFC: dorsolateral prefrontal cortex; EAT-26: Eating Attitudes Test-26; FibroQoL: multicomponent intervention for fibromyalgia; FIQ(-R): Fibromyalgia Impact Questionnaire (- Revised); FIS: Fatigue Impact Scale; FSS: Fatigue Severity Scale; FU: follow-up; HADS: Hospital Anxiety and Depression Scale; HAS: hypnotic analgesia session; IGUBAC: Inflammatory Gut-Brain Axis Control Diet; HF: high frequency; l: left; MBSR: Mindfulness-Based Stress Reduction; LF: low frequency; MEP: motor evoked potential; MFI-20: Multidimensional Fatigue Inventory; MFIS: Modified Fatigue Impact Scale; MG: magnesium-enriched; MSG: monosodium glutamate; M1: primary motor cortex; NIBS: non-invasive brain stimulation; NR: not reported; NRS: Numerical Rating Scale; OIC: operculo-insular cortex; PCS: Pain Catastrophizing Scale; PGIC: Patient Global Impression of Change; POMS-29: Profile of Mood States; PPTs: pain pressure thresholds; PSQI: Pittsburgh Sleep Quality Index; RAVLT: Rey Auditory Verbal Learning Test; r: right; RPT: recommended pharmacological treatment; rTMS: repetitive transcranial magnetic stimulation; SDBOLD: blood-oxygenation-level-dependent signal variability detected with functional magnetic resonance imaging; SF-MPQ: Short-Form McGill Pain Questionnaire; SF-36: Short Form 36 for health status survey; STAI-TA: State Trait Anxiety Inventory – trait anxiety; TAU: treatment as usual; TCM: traditional Chinese medicine; tDCS: transcranial direct current stimulation; 10-MWT: 10 m walking test; trt: treatment; TRY: tryptophan; VAS: Visual Analogue Scale; WL: waiting list
*Non-penetrating needle device with a blunt and retractable needle and a guide tube
§Needles inserted into points that are not recognized as acupoints or in meridians /simulated: needles into the bandage but not into the skin
Pharmacological therapy
Antidepressants. From the 40 clinical trials reviewed, we selected eleven randomised controlled trials (RCTs) that investigated the effect of antidepressants in patients with fibromyalgia (Table 2). Two placebo-controlled trials, involving a total of 577 patients, investigated the effect of duloxetine. [ref. 7, ref. 12]. Although the primary endpoint was not significantly improved by duloxetine compared to placebo, treatment with duloxetine was associated with an improvement in secondary endpoints, including analgesia and quality of life. An RCT comparing the effects of duloxetine and pregabalin showed an improvement in WPI scores with a statistically significant difference between the two treatment arms in favour of duloxetine [ref. 8]. An RCT compared the effect of duloxetine and acetyl-L-carnitine on pain, depression and anxiety [ref. 14]. Both drugs led to an overall clinical improvement, with positive effects on pain and depressive symptoms. Three RCTs with a total of 119 patients investigated the effect of milnacipran 100 mg [ref. 9, ref. 10, ref. 13]. Treatment with milnacipran showed no significant reactivation of conditioned pain modulatory function [ref. 9] and was not superior to placebo in reducing clinical pain and mechanical and thermal pain sensitivity in quantitative sensory tests [ref. 13]. Ahmed and colleagues investigated the effect of milnacipran on polysomnographic measures of sleep and pain. This drug did not significantly improve the polysomnographic parameters, but showed a significant improvement in daily pain perception and subjective sleep quality [ref. 10]. In an RCT involving 430 patients, the effect of mirtazapine on FMG was investigated [ref. 11]. The authors found a greater reduction in the mean NRS pain score from baseline to the end of treatment with mirtazapine (30 mg) compared to placebo [ref. 11]. Three RCTs investigated the efficacy of combination treatment with antidepressants and pregabalin [ref. 15–ref. 17]. The combination of pregabalin (450 mg) and duloxetine (120 mg) improved several clinical outcomes compared to pregabalin monotherapy [ref. 17]. Pregabalin (300 mg) in combination with milnacipran (100 mg) showed adequate efficacy in the treatment of patients with FMG, but no superiority over monotherapy [ref. 15]. The combined use of pregabalin (75 mg) plus paroxetine (25 mg) improved somatic and depressive symptoms compared to pregabalin plus amitriptyline (25 mg) or venlafaxine (75 mg) [ref. 16].
Recent studies have investigated the effect of dietary supplements as add-on therapy to gabapentinoids and antidepressants, as explained in the following paragraph (Table 2).
Although more RCTs are needed to draw convincing conclusions about efficacy in FM, antidepressants in monotherapy or in combination with a2d ligands are considered a valid treatment option for patients with FM. Minor side effects are common, but serious events are rare.
Antiepileptic drugs (gabapentinoids). According to the search strategy of this review, we screened 33 clinical trials on anticonvulsants in fibromyalgia and selected ten randomized controlled trials with a total of 4715 patients (Table 2).
Most studies investigated the efficacy of pregabalin alone or in combination with other medications compared to placebo. In particular, two RCTs showed the efficacy of PGB compared to placebo on pain and related symptoms in adults [ref. 21, ref. 22] and one study in adolescents with FM [ref. 20] failed to show a significant improvement in mean pain score. The association with CoQ10 showed an improvement in the efficacy of PGB [ref. 24]. The efficacy of PGB at a dose of 150 mg twice daily was found to be no better than a single daily dose of 300 mg [ref. 23]. The combination of PGB with exercise was more effective than exercise alone [ref. 18]. The combination with duloxetine showed greater efficacy than placebo and than the two monotherapies [ref. 17], while a study directly comparing the efficacy of PGB with that of duloxetine showed the superiority of duloxetine [ref. 8]. The combination with another antidepressant such as milnacipran did not show superiority compared to treatment with PGB alone [ref. 15]. Mirogabalin, a gabapentinoid that binds to the α2δ-subunit of the voltage-gated calcium channel and has significantly higher efficacy, was found to be safe but less effective than PGB and placebo at a dose of 15 mg once or twice daily [ref. 19]. Although gabapentinoids are generally effective, their use often causes side effects such as dizziness, drowsiness, headache and fatigue.
Medicinal cannabis there are numerous clinical studies on cannabis and D-9-tetrahydrocannabinol (THC) for chronic pain conditions. However, there is clinical evidence for the use of CBD in FM showing a wide-ranging therapeutic effect [ref. 81]. Unfortunately, there is only weak evidence of efficacy as CBD + THC therapy has not been thoroughly tested in large clinical trials.
A Cochrane review published in 2016 examined 2 randomized controlled trials of cannabinoids in FM involving 72 patients. Both studies investigated the effect of nabilone, a derivative of Delta 9-THC. In the study by Shrabek et al. nabilone was compared with placebo, in the other study with amitriptilyne. The authors described good results in terms of pain and sleep, but with a low level of evidence, short-term treatment and insufficient data on important outcome measures such as disability scores and psychopathological variables [ref. 25].
Two RCTs have been published in the last 10 years. Van de Donk et al. [ref. 25] tested 4 different cannabis strains: Bedrocan (22.4 mg THC, 0.1 mg CBD); Bediol (13.4 mg THC, 17.8 mg CBD); Bedrolite (18.4 mg CBD, 0.1 mg THC); and a placebo strain without THC or CBD, by inhalation). They evaluated the relief of experimental pressure pain, electrical pain and spontaneous pain (primary endpoints) as well as the subjective and psychotropic effects. The analgesic efficacy of the active treatment was limited, but strains containing THC plus CBD-bediol showed a 30% effect on all pain scores. The study by Chaves et al. [ref. 26, ref. 82] aimed to determine the benefits of a tetrahydrocannabinol (THC)-rich cannabis oil on the symptoms and quality of life of fibromyalgia patients. They used a THC-rich cannabis oil (24.44 mg/mL THC and 0.51 mg/mL9) compared to placebo in 17 women with fibromyalgia and observed a significant improvement in FIQ scores in the treated group.
In summary, there are few RCTs with weak evidence for the efficacy of CBD and THC in the treatment of FM, raising doubts about the best therapeutic formulation, route of administration and timing of treatment (Table 2). However, numerous observational studies appear to indicate good tolerability and therapeutic efficacy of medical cannabis in patients with FM [ref. 83], with a focus on cannabidiol [ref. 84, ref. 85]. Given the paucity of pharmacological options for FM, an RCT on the combination of THC and cannabidiol (bediol) could be of great benefit.
Opioids—There are few randomized controlled trials that have investigated the efficacy of opioids in fibromyalgia. Most of these studies were published more than 10 years ago.
More than 10 years ago, Sorensen et al. [ref. 82, ref. 84] reported on two different studies investigating the effect of morphine (0.3 mg/kg) on pain relief using different clinical scales. The conclusion from these studies was that morphine did not significantly improve pain, but few side effects such as nausea and/or vomiting were reported by patients. Later, tramadol (100 mg) was tested in fibromyalgia in a randomized controlled trial [ref. 85] in a small group of patients. This study found that tramadol led to a reduction in spontaneous pain, while no difference was found in objectively measured pain (pressure dolorimetry). A more reliable study was conducted by Russel et al. [ref. 86], which investigated tramadol (50–400 mg) in a large cohort of patients. The results of this study support the efficacy of tramadol over a 6-week period for the treatment of fibromyalgia pain, assessing the cumulative probability of discontinuation of the double-blind study due to inadequate pain relief. The combination of tramadol (37.5 mg) with paracetamol (325 mg) was then tested [ref. 87]. The main outcomes assessed were cumulative time to treatment discontinuation, pain relief, total number of tender points and specific questionnaires. The conclusion of this study was that this combination is effective in the treatment of fibromyalgia pain and has no serious adverse effects. Younger et al. [ref. 88] used naltrexone (50 mg/die) in a small group of fibromyalgia patients. Outcomes studied included changes in sensitivity to heat, cold, and mechanical pain, as well as measures of mood and opioid withdrawal symptoms. The authors concluded that naltrexone had no effect on pain and mood in fibromyalgia patients, suggesting that endogenous opioid activity is not involved in the pathophysiology of fibromyalgia. In addition, the effect of low-dose naltrexone (4.5 mg/day) on daily self-reported pain and overall life satisfaction, positive mood, sleep quality and fatigue was investigated in a small cohort of patients [ref. 89]. This study showed that low-dose naltrexone has a clinically beneficial effect on fibromyalgia pain without serious side effects. However, the conclusion of this study was not confirmed in a trial of low-dose naltrexone involving a larger cohort of fibromyalgia patients [ref. 27]. In fact, no clinically relevant analgesic efficacy of low-dose naltrexone treatment was observed in these patients when using the revised Fibromyalgia Impact Questionnaire scores and the sum of pain intensity ratings as primary outcomes.
To summarize, there are only a few RCTs, mostly from the last 10 years, investigating the efficacy of opioids and their antagonists. However, with the exception of tramadol, there is no clear evidence of the efficacy of these drugs for pain relief in fibromyalgia patients. Of the drugs studied, morphine and naltrexone showed no clear effect, while tramadol showed positive effects on pain relief, although objective evidence of its efficacy was lacking.
Given these contrasting results, further RCTs on these drugs, particularly tramadol, are desirable to establish objective measures.
Muscle relaxant drugs. In the last 10 years, research into RCTs to demonstrate the effect of muscle relaxant drugs has not yielded results. In a study conducted more than 10 years ago, Reynolds et al. conducted a double-blind cross-over study of cyclobenzaprine at an evening dose of 10 or 30 mg and found a positive effect on tender points, anxiety, stiffness, and fatigue at the 10-mg dose, with fewer side effects than at the other dose [ref. 90]. Cyclobenzapine is currently used in Italy and is also considered as therapeutic support in other recommendations [ref. 4], but the lack of recent studies does not allow to formulate an opinion on its possible efficacy.
Nutraceutical products. The effect of dietary supplements in fibromyalgia has recently been investigated in a few studies, but these had methodological limitations, such as small sample sizes and, in many cases, the concomitant use of other therapeutic strategies [ref. 28, ref. 29, ref. 31, ref. 32, ref. 89, ref. 91]. Acetyl-L-carnitine (ALC) has only been tested in one study of FM [ref. 92], but its usefulness in relieving neuropathic pain [ref. 93] and improving depressive symptoms [ref. 94, ref. 95] provides a rationale for its use. A recent RCT studied 65 female FM patients randomised to receive either duloxetine 60 mg/day or ALC 1500 mg/day p.o. and found a significant improvement in mood/depressive state, severity of illness and physical well-being. However, there was no significant reduction in VAS pain scores, and neither drug improved anxiety.
Palmitoylethanolamide (PEA) is an endogenous lipid mediator with neuroprotective, anti-inflammatory and analgesic effects. To date, only three studies have investigated the effect of PEA in fibromyalgia. Two of these were observational studies that showed a positive effect on pain and limited adverse effects [ref. 89, ref. 91]. Only one RCT aimed to evaluate the effects of PEA (600 mg b.i.d.) in combination with acetyl-carnitine in FM patients already receiving duloxetine and pregabalin.The addition of these supplements to conventional pharmacological therapies led to significant improvements in all outcomes assessed (WPI, FIQR, FASmod) [ref. 28].
Alpha-lipoic acid (ALA) is an antioxidant and immunomodulatory agent that plays an important role in various metabolic processes. In the CADENCE study, an ALA-pregabalin combination (with a randomized, double-blind, 3-phase crossover design) was investigated (over 6 weeks). This study provided no evidence of additional benefit of ALA compared to pregabalin in terms of daily pain and all secondary endpoints. The maximum tolerated doses of ALA and pregabalin were similar for both combination therapy and monotherapy, and adverse events (AEs) were rare [ref. 29].
The IMPALA study (a double-blind, randomized, placebo-controlled crossover study with a small sample size over 10 weeks) showed no beneficial effects of ALA compared to placebo, with no significant adverse events (good tolerability) [ref. 31]. Contrasting results (to the IMPALA study) were obtained by Esposito et al. [ref. 30] in a (monocentric, randomized, double-blind, placebo-controlled) clinical trial involving only 12 patients with fibromyalgia (who needed an alternative treatment to conventional painkillers because they were unable or unwilling to take painkillers). They reported benefits (with ALA 400–800 mg/day) in terms of pain intensity with no adverse effects (in particular, no hepatic toxicity and no effect on blood glucose levels).
A randomized, controlled study compared the effect of a nutrient combination containing coenzyme Q10, vitamin D, ALA, magnesium and tryptophan (Migratens) with the effect of acupuncture over a period of 3 months. This showed a reduction in pain and an improvement in quality of life (FIQ-R and FSS) with more limited effects for Migratens compared to acupuncture, despite a higher number of discontinuations due to side effects [ref. 32]. These authors recommend cyclical use to improve long-term compliance.
Magnesium (MG) supplementation has been suggested to alleviate the various symptoms associated with fibromyalgia, reduce certain types of pain and improve the central nervous system’s ability to withstand stress. In recent years, only one RCT examined the effects of MG on stress in 76 patients with fibromyalgia, including pain, sleep, quality of life, fatigue, catastrophizing, social vulnerability, and MGg blood concentrations [ref. 33, ref. 34, ref. 96]. The results show that magnesium improves mild/moderate, but not severe, stress and reduces pain perception in fibromyalgia patients.
Ultimately, the use of dietary supplements containing ALC, PEA and MG is promising for the treatment of FM in conjunction with pharmacologic therapies, but the data are inconclusive due to the lack of clinical trials with sufficient sample size on larger series.
Non pharmacological approach
Behavioral approach—Cognitive Behavioural Therapy (CBT) CBT enables patients to change their negative thoughts, feelings and expectations. This can improve mood, improve the ability to cope with pain and change the perception of pain. The pharmacological option can be complemented by CBT so that patients can become more aware of their condition.
Recent clinical experience confirms the benefits of CBT in the treatment of fibromyalgia [ref. 97, ref. 98]. A review published in 2021 by Heller [ref. 99] evaluated the latest literature and confirmed that CBT in combination with pharmacological treatment can help patients with fibromyalgia to improve pain and depression as well as catastrophizing attitudes. Some reports have shown an effect of CBT on brain connectivity [ref. 100, ref. 101].
Unfortunately, the clinical experience, although encouraging, is based on results obtained from small patient groups, short follow-up periods and studies without control conditions, so the results cannot be considered conclusive. The complexity of the clinical condition is considerable and it is likely that a single pharmacological option will not be sufficient: it is imperative that we develop multidisciplinary treatment programmes, including behavioural approaches such as CBT, to help patients with fibromyalgia.
Acceptance Commitment Therapy (ACT)—ACT belongs to the third wave of behavioral therapy approaches. It aims to improve patients’ functioning and quality of life by increasing psychological flexibility, defined as the ability to observe and accept aversive and disturbing thoughts, emotions and bodily sensations without reacting to them, and to promote behavior consistent with personal values [ref. 96].
The EFFIGACT study, a 6-month randomized controlled trial that enrolled and randomized 156 patients, showed that ACT was statistically superior to both the recommended pharmacological treatment (RPT: pregabalin + duloxetine) and the waiting list immediately after treatment, and improvements were maintained after 6 months [ref. 36, ref. 37]. It also showed that ACT appears to be a cost-effective treatment compared to RPT, as the ACT group incurred fewer direct costs compared to the two control arms during the 6-month study period due to lower costs for primary care visits and FM-related medications [ref. 34].
In addition, Varallo et al. have shown that acceptance interventions are effective in reducing kinesiophobia and improving performance-related physical functioning in individuals with comorbid obesity, as they can facilitate adherence to physical activity and promote weight loss in this highly comorbid clinical condition [ref. 102].
In addition, there is preliminary evidence that online delivery of ACT therapy is an effective, accessible and cost-effective treatment for people with FM and other chronic pain conditions [ref. 35].
Mindfulness-Based-Stress-Reduction (MBSR)—Mindfulness has gained attention among third-wave behavioral therapy interventions in the last year, and there are studies demonstrating the use of mindfulness for various forms of pain.
The Mindfulness-Based Stress Reduction (MBSR) program is a multi-component intervention that aims to alleviate suffering through the cultivation of mindfulness: Mindfulness is an attitude of “awareness that arises through conscious attention in the present moment and nonjudgmental observation of the unfolding experience” [ref. 100]: It is a helpful complementary treatment for patients with fibromyalgia, as it improves some of the main symptoms of fibromyalgia and reduces the subjective burden of the disease [ref. 101].
The EUDAIMON study, a 12-month randomized controlled trial, included 225 participants with fibromyalgia who were randomly assigned to 3 study arms: MBSR plus usual care (TAU), FibroQoL (multicomponent intervention for FM) plus TAU, and TAU alone. The results showed that functional outcomes and symptoms in fibromyalgia were moderately improved by MBSR plus usual care compared to usual care alone [ref. 37].
Other references confirm the benefits of MBSR in FM as an effective intervention to reduce the clinical severity of patients with FM. In addition, MBSR can modulate immune-inflammatory signaling pathways relevant to the pathophysiology of FM [ref. 36] (Table 3).
In summary, although the data are preliminary and based on studies without control conditions or long-term follow-up, it is reasonable to consider behavioural approaches to support patients suffering from FM during their difficult therapeutic strategies to provide them with tools that enable them to endure and manage pain and improve their quality of life and disability resulting from the disease.
Acupuncture. From the 23 screened clinical trials, nine randomized controlled trials (RCT) were selected to investigate the effect of acupuncture in patients with fibromyalgia (Table 3), involving 725 patients. Eight studies used the Traditional Chinese Medicine [TCM] needle acupuncture technique, while one study used electroacupuncture (EA) (i.e. AS Super 4 Digital Needle Stimulator) with low-frequency stimulation [ref. 45]. The predominant sham procedure was a non-penetrating needle procedure using a blunt and retractable needle and guide tube or placement on non-traditional acupuncture points [ref. 39, ref. 40], and better efficacy of true acupuncture compared to sham acupuncture was found. Interestingly, Uğurlu et al. [ref. 40, ref. 46] and Karatay et al. [ref. 42] described a significant improvement in pain intensity, functional impairment and emotional distress 2 and 3 months after treatment, respectively, although a consistent placebo effect after sham acupuncture was found only in the short term (1 month). Three studies did not use sham stimulation but alternative control conditions, namely a group discussion of a book on the current understanding of fibromyalgia [ref. 43], a dietary supplement [ref. 32], physiotherapy [ref. 44] or treatment as usual [ref. 38]. In three of these studies, the authors found clinically and statistically significant improvement in the acupuncture group compared to the control groups, not only in terms of pain intensity and functional outcome [ref. 43, ref. 44] but also in terms of other secondary measures such as pressure pain thresholds, emotional distress and fatigue, activity engagement, general health status and global subjective improvement [ref. 38], at post-treatment and follow-up assessments. Only in the RCT comparing acupuncture with physiotherapy was true acupuncture found to be as effective as the control treatment [ref. 44]. A notable result was found in the study by Zucker and colleagues [ref. 41], who investigated the effects of needle acupuncture taking into account patients’ baseline pressure pain threshold (PPT). The authors found that patients with a higher pressure pain threshold experienced more significant pain relief after active acupuncture. In contrast, participants with lower PPTs showed a stronger pain-relieving response to sham acupuncture [ref. 41]. Finally, only one selected RCT [ref. 45] examined the neural correlates of electrical acupuncture stimulation. Consistent with previous studies, the results reported that active acupuncture treatment reduced pain intensity more compared with sham treatment, although there was no difference in terms of acupuncture sensations and treatment credibility.
In most studies, points were standardized among participants [ref. 38, ref. 40, ref. 42–ref. 45] considering FM tender points of fibromyalgia defined according to the 1990 ACR classification criteria [ref. 103] or predominant FM symptoms [ref. 38, ref. 41, ref. 42, ref. 45]. In the remaining three studies, the stimulation points were subjectively selected by experienced acupuncturists based on the individual patient [ref. 32, ref. 39, ref. 43]. The average duration of each stimulation was about 30 min and was repeated on average twice a week or once a week and then gradually increased/decreased to several times a week by the end of the treatment period. The longest treatment duration of the included studies was 10 weeks. Only two studies did not investigate follow-up measures. All but one study [ref. 44] reported long-term efficacy of acupuncture stimulation, and the longest follow-up period was 1 year [ref. 39] (Table 3). Further RCTs are needed to clearly demonstrate the efficacy of acupuncture for fibromyalgia. Mild side effects are frequently observed, but serious adverse events are rare. There are some differences between the various procedures in terms of acupuncture points, duration and number of stimulations.
Physical activity Exercise guidelines for fibromyalgia patients are based on a limited literature similar to that for healthy adults, but can be supplemented by recommendations from seven RCTs that examined various exercise interventions with 831 patients (Table 3). A manual selection of the most relevant RCTs was made based on sample size, methodological/statistical rigor applied, and the types of exercise protocols specified in the main generally accepted guidelines.
Larsson et al. [ref. 46] investigated resistance training and found an improvement in muscle strength and pain intensity. Collado-Mateo et al. [ref. 47] studied exergames and reported improvements in quality of life and pain. Wang et al. [ref. 48] compared Tai Chi with aerobic exercise and found similar improvements in fibromyalgia symptoms. Andrade et al. [ref. 49] investigated the effects of aquatic exercise and found a possible benefit in alleviating symptoms. Izquierdo-Alventosa et al. [ref. 50] studied low-intensity physical exercise and found a reduction in pain catastrophizing and improvements in psychological and physical aspects. Serrat et al. [ref. 51] investigated a multicomponent treatment that was shown to be effective in symptom control through neuroscience pain education, therapeutic exercise, cognitive behavioural therapy, and mindfulness. Finally, Gentile et al. [ref. 52] found that physical activity at home improved small fibre pathology and disease severity in a group of fibromyalgia patients (Table 3).
In accordance with the guidelines for healthy adults, some considerations are fundamental:
- Increasing the frequency of physical activity (PA) to 3 days per week is associated with a greater reduction in symptoms than 1–2 days per week.
- It is important to ensure adequate rest between exercises by alternating different body parts to optimize effectiveness.
- If an uninterrupted 30-min aerobic exercise session is not tolerated initially, breaking it up into shorter sessions and providing more support may improve adherence to the program [ref. 46, ref. 104].
The rate of progression within the FITT-VP (Frequency, Intensity, Time, Type-Volume Progression) framework depends on symptoms, so it is necessary to adjust the intensity or duration of PA during a flare-up. Minimizing the eccentric components of resistance exercise reduces muscle microtrauma during flare-ups [ref. 46, ref. 50, ref. 104].
Individuals suffering from fibromyalgia fall into physical inactivity due to symptoms. The prescription of PA should be based on pain tolerance and should be gradual while pain levels are monitored [ref. 50, ref. 104]. Structuring exercise programs to minimize barriers and accommodate individual preferences can improve adherence. This is best done in supervised or group sessions that encourage social support [ref. 51].
Functional activities with gradually increasing intensity, from light to moderate, are recommended despite the symptoms [ref. 50]. Proper demonstration of exercise biomechanics limits the risk of injury and PA in a controlled environment (temperature and humidity) can mitigate symptom exacerbation [ref. 46, ref. 104].
Both land-based and aquatic aerobic PA, together with exergames, are useful to improve physical function and reduce pain [ref. 47, ref. 49, ref. 104]. Complementary therapies such as Tai Chi and yoga also alleviate symptoms [ref. 48].
Overall, these studies highlight the importance of non-pharmacological interventions and in particular aerobic exercise and resistance training, possibly personalized and supervised, in the treatment of fibromyalgia and provide insights into the association with different approaches to alleviate symptoms and improve quality of life. However, further research is needed to investigate the long-term effects and optimal combinations of these interventions.
Diet—Among the causes unrelated to the specific causes of central sensitization, a fundamental mechanism in FM, is inflammation [ref. 105]. Inflammation consists of a dynamic sequence of phenomena manifested in an intense vascular response and, above all, in the release of endogenous substances: the chemical mediators of inflammation [ref. 106].
Diet can cause inflammation. Inflammatory conditions of the digestive system can trigger the release of cytokines, which can have effects on the central nervous system [ref. 107].
There is a general impression that a plant-based, Mediterranean, vegetarian and vegan diet can reduce musculoskeletal pain, especially in patients with rheumatoid arthritis [ref. 108].
However, there is only weak evidence for the beneficial effect of specific diets in FM, although studies suggest that weight control, a modified diet high in antioxidants and supplementation are beneficial in relieving symptoms [ref. 109].
Among the RCTs addressing the effects of diet on the symptoms of FM, seven described the effects of different dietary approaches on FM symptoms.
Mauro-Martín et al. [ref. 56] analyzed the effects of an olive tree-based supplement and a gluten-free, and low histamine diet (IGUBAC-Diet®), with antioxidant and anti-inflammatory characteristics, in women with fibromyalgia in a RCT trial with 31 women with FM They found a general improvement in pain, fatigue and quality of life.
Another RCT investigated the effects of avoiding monosodium glutamate (MSG) and aspartame on pain perception in fibromyalgia. However, eliminating MSG and aspartame from the diet did not lead to an improvement in fibromyalgia symptoms [ref. 53].
A recent RCT investigated the effects of a Mediterranean diet enriched with tryptophan (TRY) and magnesium (MG) on psychological variables (anxiety, mood, eating disorders, self-image) and sleep quality in women with fibromyalgia [ref. 33].
The intervention group received a Mediterranean diet enriched with high doses of TRY and MG (60 mg TRY and 60 mg MG), while the control group received the standard Mediterranean diet.
Patients receiving the supplement showed improvements in anxiety, depression and eating disorders. [ref. 33] The khorasan wheat replacement diet led to an improvement in key FMS indices such as WPI, SS and FIQ compared to the control wheat diet. The clinical improvement corresponded with the changes in the gut microbiota [ref. 57]. Roman et al. investigated the effect of supplementation with probiotics of different species on clinical symptoms and cognitive profile in patients with FM and found a positive effect on impulsivity and decision making [ref. 55, ref. 70, ref. 110]. Another study investigated the differential effects of a gluten-free diet compared to a hypocaloric diet in 75 patients with cystic fibrosis randomly assigned to the two diets. Both diets had positive effects on clinical symptoms and gluten sensitivity [ref. 54].
A recent RCT [ref. 58] studied 84 cystic fibrosis patients, 39 of whom followed a personalized Mediterranean diet (DIET group) and 45 a general balanced diet (NODIET group). Patients in the DIET group showed improvement in most fibromyalgia parameters, including disability score, fatigue and anxiety (Table 3).
In conclusion, further studies are needed to determine the best nutritional support for FM.
A hypocaloric Mediterranean-type diet with probiotics and magnesium and tryptophan supplementation appears to have a beneficial effect on FM.
Non invasive brain stimulation (NIBS) The therapeutic options for fibromyalgia (FM) are limited, and medications, which are often associated with side effects, play only a marginal supportive role [ref. 77]. Non-invasive brain stimulation (NIBS) techniques such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) are safe and well-tolerated options used for many pain conditions, including FM [ref. 111, ref. 112]. Here we summarize the most important randomized controlled trials (RCTs) investigating the efficacy of NIBS in the treatment of FM.
Transcranial direct current stimulation—tDCS. TDCS is a neuromodulation technique that can increase the excitability of corticospinal pathways [ref. 70]. It uses direct current applied to the scalp via electrodes to induce changes in cortical activity and excitability [ref. 110, ref. 113]. In FM patients, stimulation of target areas such as the primary motor cortex (M1) [ref. 59], dorsolateral prefrontal cortex (DLPFC) [ref. 62, ref. 68] and operculo insular cortex (OIC) [ref. 66, ref. 67] improves fatigue [ref. 69], quality of life (QoL) [ref. 69], pain catastrophizing and pain-related disability [ref. 68]. Transcranial direct current stimulation –tDCS- via the DLPFC improves specific cognitive functions related to episodic short- and long-term memory and executive functions [ref. 61], while no effects on alertness were found [ref. 60].Improvements in pain perception [ref. 62] have been demonstrated with lower visual analogue scale (VAS) scores and depressive symptoms [ref. 67] as assessed by the Fibromyalgia Impact Questionnaire (FIQ) and Beck’s Depression Inventory (BDI) [ref. 65].
Conflicting data are available on tDCS in combination with physical exercise [ref. 63–ref. 65].
Repetitive transcranial magnetic stimulation – rTMS. The effects of rTMS are mediated by an electromagnetic field over the scalp, which modulates the excitability of both deep brain and cortical areas [ref. 114]. In FM patients, rTMS of the DLPFC improves fatigue and pain [ref. 72, ref. 74, ref. 75]. When considering physical functioning, depression and perception of general health status as outcomes, rTMS of the left premotor cortex (PMC) is more effective than rTMS of the left DLPFC [ref. 73]. It should be mentioned that the effect of rTMS of the left DLPFC compared to sham stimulation on pain, stiffness, fatigue, quality of life, mood and cognitive state is controversial [ref. 76].
The rTMS-induced improvements in quality of life occur via the metabolism of the right limbic system, which may be the neural substrate of the rTMS effect on emotional dimensions [ref. 71]. It is noteworthy that studies show that the improvement in FM symptoms induced by rTMS lasts up to 6 months after the last stimulation [ref. 78, ref. 80, ref. 115]. Other studies [ref. 77, ref. 79] compared the effects of rTMS and tDCS on pain and quality of life in FM patients and showed a higher and longer lasting analgesic effect of rTMS (Table 3).
In conclusion, although more data are needed to support the benefits of NIBS in fibromyalgia, the available studies are encouraging and show that both tDCS and rTMS not only lead to pain reduction but could also have a positive effect on the complex galaxy of symptoms typical of the disease.
Consensus
After the presentation of the RCT revisions, the members of the NPG (N° 13) answered the following questions.
- Patients with fibromyalgia should take pharmacological treatment after the initial diagnosis
- Antidepressants should be used as a first approach (duloxetine 30/60 mg, * mirtazapine 30 mg/day)
- Antiepileptic drugs should be used as a first approach (pregabalin 150—300 mg/day) *
- The choice between antidepressants or antiepileptics depends on clinical assessment (comorbidity with depression and/or anxiety)
- Integrators and nutrients should be used as first choice
- Opioids should be suggested on first approach
- The combination of antiepileptics and antidepressants should be suggested in patients who do not respond to monotherapy
- Cannabis should be used in drug-resistant patients
- The following treatments would be worth further evidence: medical cannabis
- The following treatments would be worth further evidence: opioids (tramadol, tapentadol)
- The following treatments would be worth further investigation: nutraceutical products
- Physical activity (supervised multicomponent activity including aerobic and resistance training) should be suggested in the first approach
- A personalized Mediterranean diet should be prescribed at the first approach
- Cognitive behavioral therapy (acceptance and commitment and/or mindfulness) should be prescribed as a second-line non-pharmacological approach
- Motor or dorsolateral prefrontal cortex TMS or TDCS should be suggested as a * second-line non-pharmacological approach
- Traditional Chinese medicine acupuncture should be used as a second non-pharmacological option
In terms of pharmacological treatment, there was no clear consensus on the possibility of prescribing medication in the first approach: however, the group agreed on the choice between antiepileptics or antidepressants basing on clinical judgment about comorbidities for anxiety and depression (Fig. 1). The use of opioids as first choice is discouraged unless further studies are conducted, including other mild opioids such as tramadol and tapentadol (Fig. 2). The group does not advocate the prescription of nutraceuticals as first-line treatment, but recommends further systematic studies. (Fig. 2). As a second choice, experienced neurologists recommend the association between antiepileptic drugs and antidepressants, while they disagree with cannabis use, but consider it appropriate to design further more systematic studies (Figs. 1 and 2). Regarding the non-pharmacological approach, neurologists agree to suggest it in the first visit in the form of physical activity, especially aerobic exercise and strength training (Fig. 2). For the second option, they agree on the benefits of a cognitive behavioral approach but the most of them disagreed with the indication of acupuncture. (Fig. 2). For diet and NIBS, no consensus was reached on the potential benefits of a second-choice approach (Fig. 3).
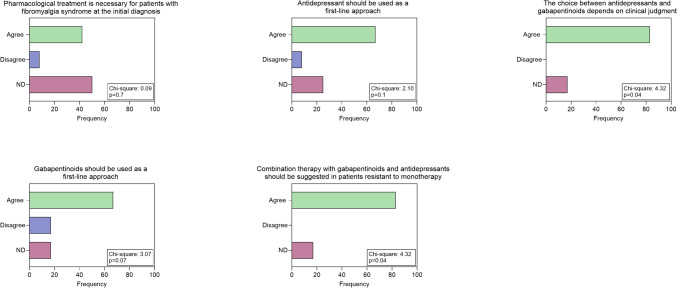
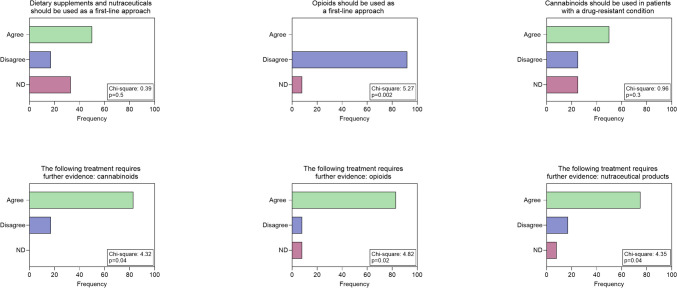
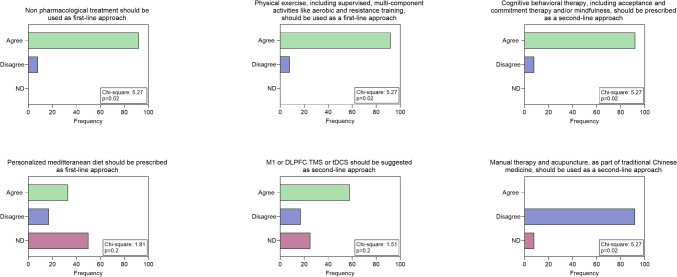
Figures 4 and 5 show the final recommendations.


To summarize, the use of antiepileptic drugs or antidepressants is not recommended as a first choice, while their use in case of comorbidity with anxiety and depression is based on medical judgment. Association is recommended when monotherapy fails.
Non-pharmacological treatments – aerobic exercise and resistance training – are recommended as first choice.
As a second-line treatment, neurologists suggest cognitive behavioral therapy.
Medicinal cannabis, tramadol or tapentadol, nutraceutics, are worth further controlled trials and could be used in individual patients under medical supervision. (Fig.4, Fig 5)
Discussion
With this study, the Neuropathic Pain Interest Group of the Italian Society of Neurology aimed to provide neurologists with practical guidelines for the treatment of fibromyalgia, a condition in which the involvement of the peripheral and central nervous system has been confirmed in several studies [ref. 2]. Previously, the same group had already published practical recommendations on the diagnostic procedures useful for the clinical assessment of fibromyalgia [ref. 3].
The group came to a consensus based on their clinical impression and supported by a review of the most recent randomized controlled trial, which was selected on the basis of a joint assessment of validity for clinical purposes.
The conclusions did not differ from those formulated by experienced rheumatologists [ref. 4] and previous guidelines [ref. 116]. The EULAR recommendations are based on a systematic review of the evidence since 2015, whereas we undertook a more flexible, narrative review of recent RCTs. As previously reported by other authors [ref. 117], the global approach in patients with fibromyalgia is particularly challenging, as the present study was able to confirm the lack of a specific pharmacological approach and the need for a multidisciplinary intervention, which is difficult to implement in current clinical practice. In relation to the EULAR recommendation, where no consensus was reached on the possibility of using CNS-acting medications in the treatment of comorbidities such as sleep disorders, neurologists agreed on the choice between antidepressants or anticonvulsants on medical judgment when there is a comorbidity for depression or anxiety. Given the prevalence of anxiety and depression in these patients, which ranges from 13 to 80% [ref. 118], antidepressants and antiepileptic drugs, which have been shown to be effective in the majority of patients, could also have a positive effect on pain symptoms and could be an indication for pharmacological treatment at the first approach. Furthermore, the group agreed on the efficacy of combination therapies with antiepileptics and antidepressants when monotherapy fails. Muscle relaxant drugs are commonly used in patients with FM, but the lack of studies in recent years and the lack of studies in the years before that does not even allow to formulate an opinion on their indication. In line with the rheumatologists’ guidelines [ref. 4], our group has also not recommended and even discouraged the habitual use of opioids and has not reached a positive consensus on medical cannabis and nutraceuticals as first- or second-line therapies. However, they are confident about good patient compliance with medical cannabis and nutraceuticals, which is an important point in patients with FM to avoid the common nocebo effect [126]. For these reasons, the group advocates further RCTs in a global scenario of paucity of target therapies.
Similar to the recommendations of the rheumatologists [ref. 4], our group of neurologists also recommended physical activity as the first measure. Following the recommendations of physical activity experts, the group recommended a personalized treatment based on supervised aerobic exercise and strength training with an individualized combination of several components. This is a practical proposal that can be implemented with the support of sports physicians as part of an adapted physical activity program. These programs could be performed in private gyms with additional costs for the patients, but they could also be performed at home after a preliminary examination [ref. 52].
The cognitive behavioral approach is also recognized as beneficial for patients with fibromyalgia, although it requires expert support that is very rarely available in our public health system. For these reasons, although the group reached a large consensus on its effectiveness, it was proposed as a second choice. However, the current consensus suggests that the non-pharmacological approach needs to be strengthened in the public health system in general. This also applies to NIBS, which, despite being supported by several studies in terms of efficacy and safety, is not accessible in public structures.
In contrast to the non-pharmacological interventions mentioned above, acupuncture and diet were not positively evaluated as first- or second-line interventions. The reason for this could be the use of different acupuncture procedures based on the individual experience of professionals, which our group could not rely on. Different diets were also investigated and the variability of the proposed dietary approach may currently discourage the recommendation of specific dietary indications. The Mediterranean diet is the easiest to follow in our country, so only limited agreement could be reached on its applicability in the clinical management of FM patients.
Study limitations The study was based on a non-systematic review of the current literature, which served to update the general knowledge of RCTs and to formulate practical guidelines. These are not based on evidence but on the subjective agreement of neurologists specializing in neuropathic pain.
Conclusions
There is still no specific therapeutic approach for FM based on causal mechanisms. In a general scenario of conflicting opinions about the pathophysiology and management of this complex disease, together with a general confusion about neurological expertise, the present consensus could be of help to confirm the role of the neurologist in the clinical management of patients with FM. Pharmacologic treatment with antiepileptics and antidepressants in patients with co-occurring anxiety and depression and an early non pharmacologic approach based primarily on physical exercise could be a useful indication in current neurology clinical practice. A better organized public health system, including more accessible non-pharmacological options such as cognitive behavioral therapy and NIBS, could improve evidence of efficacy and lead to relevant improvement in FM-related disability.
References
- F Wolfe, DJ Clauw, MA Fitzcharles, DL Goldenberg, W Häuser, RL Katz, PJ Mease, AS Russell, IJ Russell, B Walitt. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin Arthritis Rheum, 2016. [DOI | PubMed]
- M de Tommaso, E Vecchio, M Nolano. The puzzle of fibromyalgia between central sensitization syndrome and small fiber neuropathy: a narrative review on neurophysiological and morphological evidence. Neurol Sci, 2022. [DOI | PubMed]
- G Devigili, G Di Stefano, V Donadio, I Frattale, E Mantovani, M Nolano, G Occhipinti, V Provitera, S Quitadamo, S Tamburin, A Toscano, S Tozza, A Truini, M Valeriani, M de Tommaso. Clinical criteria and diagnostic assessment of fibromyalgia: position statement of the Italian Society of Neurology-Neuropathic Pain Study Group. Neurol Sci, 2023. [DOI | PubMed]
- GJ Macfarlane, C Kronisch, LE Dean, F Atzeni, W Häuser, E Fluß, E Choy, E Kosek, K Amris, J Branco, F Dincer, P Leino-Arjas, K Longley, GM McCarthy, S Makri, S Perrot, P Sarzi-Puttini, A Taylor, GT Jones. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis, 2017. [DOI | PubMed]
- B Ghavidel-Parsa, A Bidari. The crosstalk of the pathophysiologic models in fibromyalgia. Clin Rheumatol, 2023. [DOI | PubMed]
- J Bidonde, E Fisher, S Perrot, RA Moore, RF Bell, S Makri, W Häuser. Effectiveness of non-pharmacological interventions for fibromyalgia and quality of review methods: an overview of Cochrane Reviews. Semin Arthritis Rheum, 2023. [DOI | PubMed]
- HP Upadhyaya, LM Arnold, K Alaka, M Qiao, D Williams, R Mehta. Efficacy and safety of duloxetine versus placebo in adolescents with juvenile fibromyalgia: results from a randomized controlled trial. Pediatr Rheumatol Online J, 2019. [DOI | PubMed]
- A Bidari, E Moazen-Zadeh, B Ghavidel-Parsa, S Rahmani, S Hosseini, A Hassankhani. Comparing duloxetine and pregabalin for treatment of pain and depression in women with fibromyalgia: an open-label randomized clinical trial. DARU J Pharm Sci, 2019. [DOI]
- G Pickering, N Macian, N Delage, P Picard, JM Cardot, S Sickout-Arondo, F Giron, C Dualé, B Pereira, F Marcaillou. Milnacipran poorly modulates pain in patients suffering from fibromyalgia: a randomized double-blind controlled study. Drug Des Dev Ther, 2018. [DOI]
- M Ahmed, R Aamir, Z Jishi, MB Scharf. The Effects of Milnacipran on Sleep Disturbance in Fibromyalgia: A Randomized, Double-Blind, Placebo-Controlled, Two-Way Crossover Study. J Clin Sleep Med JCSM Off Publ Am Acad Sleep Med, 2016. [DOI]
- K Miki, M Murakami, H Oka, K Onozawa, S Yoshida, K Osada. Efficacy of mirtazapine for the treatment of fibromyalgia without concomitant depression: a randomized, double-blind, placebo-controlled phase IIa study in Japan. Pain, 2016. [DOI | PubMed]
- M Murakami, K Osada, H Mizuno, T Ochiai, L Alev, K Nishioka. A randomized, double-blind, placebo-controlled phase III trial of duloxetine in Japanese fibromyalgia patients. Arthritis Res Ther, 2015. [DOI | PubMed]
- R Staud, YE Lucas, DD Price, ME Robinson. Effects of milnacipran on clinical pain and hyperalgesia of patients with fibromyalgia: results of a 6-week randomized controlled trial. J Pain, 2015. [DOI | PubMed]
- P Leombruni, M Miniotti, F Colonna, C Sica, L Castelli, M Bruzzone, S Parisi, E Fusaro, P Sarzi-Puttini, F Atzeni, RG Torta. A randomised controlled trial comparing duloxetine and acetyl L-carnitine in fibromyalgic patients: preliminary data. Clin Exp Rheumatol, 2015. [PubMed]
- YH Abdel Fattah, R Elnemr. Efficacy of pregabalin as a monotherapy versus combined pregabalin and milnacipran in the management of fibromyalgia. Int J Rheum Dis, 2020. [DOI | PubMed]
- EA Ramzy. Comparative Efficacy of Newer Antidepressants in Combination with Pregabalin for Fibromyalgia Syndrome: A Controlled, Randomized Study. Pain Practice, 2017. [DOI | PubMed]
- I Gilron, LE Chaparro, D Tu, RR Holden, R Milev, T Towheed, D DuMerton-Shore, S Walker. Combination of pregabalin with duloxetine for fibromyalgia: a randomized controlled trial. Pain, 2016. [DOI | PubMed]
- DS Karamanlioglu, D Geler Kulcu, G Ozturk, P Akpinar, F Unlu Ozkan, I Aktas. Effectiveness of pregabalin treatment for trigger points in patients with comorbid myofascial pain syndrome and fibromyalgia syndrome: a randomized controlled trial. Somatosens Mot Res, 2021. [DOI | PubMed]
- LM Arnold, S Whitaker, C Hsu, D Jacobs, D Merante. Efficacy and safety of mirogabalin for the treatment of fibromyalgia: results from three 13-week randomized, double-blind, placebo- and active-controlled, parallel-group studies and a 52-week open-label extension study. Curr Med Res Opin, 2019. [DOI | PubMed]
- Safety and efficacy of pregabalin in adolescents with fibromyalgia: a randomized, double-blind, placebo-controlled trial and a 6-month open-label extension study. Pediatr Rheumatol Online J, 2016. [DOI | PubMed]
- LM Arnold, P Sarzi-Puttini, P Arsenault, T Khan, P Bhadra Brown, A Clair, JM Scavone, J Driscoll, J Landen, L Pauer. Efficacy and Safety of Pregabalin in Patients with Fibromyalgia and Comorbid Depression Taking Concurrent Antidepressant Medication: A Randomized Placebo-controlled Study. J Rheumatol, 2015. [DOI | PubMed]
- LM Arnold, P Arsenault, C Huffman, JL Patrick, M Messig, ML Chew, L Sanin, JM Scavone, L Pauer, AG Clair. Once daily controlled-release pregabalin in the treatment of patients with fibromyalgia: a phase III, double-blind, randomized withdrawal, placebo-controlled study. Curr Med Res Opin, 2014. [DOI | PubMed]
- K Nasser, AJ Kivitz, MJ Maricic, DS Silver, SL Silverman. Twice daily versus once nightly dosing of pregabalin for fibromyalgia: a double-blind randomized clinical trial of efficacy and safety. Arthritis Care Res, 2014. [DOI]
- P Sawaddiruk, N Apaijai, S Paiboonworachat, T Kaewchur, N Kasitanon, T Jaiwongkam, S Kerdphoo, N Chattipakorn, SC Chattipakorn. Coenzyme Q10 supplementation alleviates pain in pregabalin-treated fibromyalgia patients via reducing brain activity and mitochondrial dysfunction. Free Radical Res, 2019. [DOI | PubMed]
- T van de Donk, M Niesters, MA Kowal, E Olofsen, A Dahan, M van Velzen. An experimental randomized study on the analgesic effects of pharmaceutical-grade cannabis in chronic pain patients with fibromyalgia. Pain, 2019. [DOI | PubMed]
- C Chaves, PCT Bittencourt, A Pelegrini. Ingestion of a THC-Rich Cannabis Oil in People with Fibromyalgia: A Randomized, Double-Blind Placebo-Controlled Clinical Trial. Pain Med (Malden, Mass), 2020. [DOI]
- K Bested, LM Jensen, T Andresen, G Tarp, L Skovbjerg, TSD Johansen, AV Schmedes, IK Storgaard, JS Madsen, MU Werner, A Bendiksen. Low-dose naltrexone for treatment of pain in patients with fibromyalgia: a randomized, double-blind, placebo-controlled, crossover study. Pain reports, 2023. [DOI | PubMed]
- F Salaffi, S Farah, P Sarzi-Puttini, M Di Carlo. Palmitoylethanolamide and acetyl-L-carnitine act synergistically with duloxetine and pregabalin in fibromyalgia: results of a randomised controlled study. Clin Exp Rheumatol, 2023. [DOI | PubMed]
- I Gilron, S Robb, D Tu, RR Holden, R Milev, T Towheed. Combination analgesic development for enhanced clinical efficacy (the CADENCE trial): a double-blind, controlled trial of an alpha-lipoic acid-pregabalin combination for fibromyalgia pain. Pain, 2023. [DOI | PubMed]
- C Esposito, E Ugo Garzarella, C Santarcangelo, A Di Minno, M Dacrema, R Sacchi, G Piccinocchi, R Piccinocchi, M Daglia. Safety and efficacy of alpha-lipoic acid oral supplementation in the reduction of pain with unknown etiology: A monocentric, randomized, double-blind, placebo-controlled clinical trial. Biomed Pharmacother Biomed Pharmacotherapie, 2021. [DOI]
- I Gilron, S Robb, D Tu, R Holden, T Towheed, D Ziegler, L Wang, R Milev, C Gray. Double-blind, randomized, placebo-controlled crossover trial of alpha-lipoic acid for the treatment of fibromyalgia pain: the IMPALA trial. Pain, 2021. [DOI | PubMed]
- V Schweiger, E Secchettin, C Castellani, A Martini, E Mazzocchi, A Picelli, E Polati, K Donadello, MT Valenti, L Dalle Carbonare. Comparison between Acupuncture and Nutraceutical Treatment with Migratens® in Patients with Fibromyalgia Syndrome: A Prospective Randomized Clinical Trial. Nutrients, 2020. [DOI | PubMed]
- A Martínez-Rodríguez, JÁ Rubio-Arias, DJ Ramos-Campo, C Reche-García, B Leyva-Vela, Y Nadal-Nicolás. Psychological and Sleep Effects of Tryptophan and Magnesium-Enriched Mediterranean Diet in Women with Fibromyalgia. Int J Environ Res Public Health, 2020. [DOI | PubMed]
- JV Luciano, F D’Amico, A Feliu-Soler, LM McCracken, J Aguado, MT Peñarrubia-María, M Knapp, A Serrano-Blanco, J García-Campayo. Cost-Utility of Group Acceptance and Commitment Therapy for Fibromyalgia Versus Recommended Drugs: An Economic Analysis Alongside a 6-Month Randomized Controlled Trial Conducted in Spain (EFFIGACT Study). J Pain, 2017. [DOI | PubMed]
- HD Simister, GA Tkachuk, BL Shay, N Vincent, JJ Pear, RQ Skrabek. Randomized Controlled Trial of Online Acceptance and Commitment Therapy for Fibromyalgia. J Pain, 2018. [DOI | PubMed]
- L Andrés-Rodríguez, X Borràs, A Feliu-Soler, A Pérez-Aranda, A Rozadilla-Sacanell, J Montero-Marin, M Maes, JV Luciano. Immune-inflammatory pathways and clinical changes in fibromyalgia patients treated with Mindfulness-Based Stress Reduction (MBSR): A randomized, controlled clinical trial. Brain Behav Immun, 2019. [DOI | PubMed]
- A Pérez-Aranda, A Feliu-Soler, J Montero-Marín, J García-Campayo, L Andrés-Rodríguez, X Borràs, A Rozadilla-Sacanell, MT Peñarrubia-Maria, N Angarita-Osorio, LM McCracken, JV Luciano. A randomized controlled efficacy trial of mindfulness-based stress reduction compared with an active control group and usual care for fibromyalgia: the EUDAIMON study. Pain, 2019. [DOI | PubMed]
- B Casanueva, P Rivas, B Rodero, C Quintial, J Llorca, MA González-Gay. Short-term improvement following dry needle stimulation of tender points in fibromyalgia. Rheumatol Int, 2014. [DOI | PubMed]
- J Vas, K Santos-Rey, R Navarro-Pablo, M Modesto, I Aguilar, MÁ Campos, JF Aguilar-Velasco, M Romero, P Párraga, V Hervás, O Santamaría, C Márquez-Zurita, F Rivas-Ruiz. Acupuncture for fibromyalgia in primary care: a randomised controlled trial. Acupunct Med, 2016. [DOI | PubMed]
- FG Uğurlu, N Sezer, L Aktekin, F Fidan, F Tok, S Akkuş. The effects of acupuncture versus sham acupuncture in the treatment of fibromyalgia: a randomized controlled clinical trial. The effects of acupuncture versus sham acupuncture in the treatment of fibromyalgia: a randomized controlled clinical trial. Acta Reumatol Portuguesa, 2017
- NA Zucker, A Tsodikov, SD Mist, S Cina, V Napadow, RE Harris. Evoked Pressure Pain Sensitivity Is Associated with Differential Analgesic Response to Verum and Sham Acupuncture in Fibromyalgia. Pain Med (Malden, Mass.), 2017. [DOI]
- S Karatay, SC Okur, H Uzkeser, K Yildirim, F Akcay. Effects of Acupuncture Treatment on Fibromyalgia Symptoms, Serotonin, and Substance P Levels: A Randomized Sham and Placebo-Controlled Clinical Trial. Pain Med (Malden, Mass.), 2018. [DOI]
- SD Mist, KD Jones. Randomized Controlled Trial of Acupuncture for Women with Fibromyalgia: Group Acupuncture with Traditional Chinese Medicine Diagnosis-Based Point Selection. Pain Med (Malden, Mass.), 2018. [DOI]
- EM Garrido-Ardila, MV González-López-Arza, M Jiménez-Palomares, A García-Nogales, J Rodríguez-Mansilla. Effectiveness of acupuncture vs. core stability training in balance and functional capacity of women with fibromyalgia: a randomized controlled trial. Clin Rehab, 2020. [DOI]
- I Mawla, E Ichesco, HJ Zöllner, RAE Edden, T Chenevert, H Buchtel, MD Bretz, H Sloan, CM Kaplan, SE Harte, GA Mashour, DJ Clauw, V Napadow, RE Harris. Greater Somatosensory Afference With Acupuncture Increases Primary Somatosensory Connectivity and Alleviates Fibromyalgia Pain via Insular γ-Aminobutyric Acid: A Randomized Neuroimaging Trial. Arth Rheumatol (Hoboken, N.J.), 2021. [DOI]
- A Larsson, A Palstam, M Löfgren, M Ernberg, J Bjersing, I Bileviciute-Ljungar, B Gerdle, E Kosek, K Mannerkorpi. Resistance exercise improves muscle strength, health status and pain intensity in fibromyalgia–a randomized controlled trial. Arthritis Res Ther, 2015. [DOI | PubMed]
- D Collado-Mateo, FJ Dominguez-Muñoz, JC Adsuar, MA Garcia-Gordillo, N Gusi. Effects of Exergames on Quality of Life, Pain, and Disease Effect in Women With Fibromyalgia: A Randomized Controlled Trial. Arch Phys Med Rehabil, 2017. [DOI | PubMed]
- C Wang, CH Schmid, RA Fielding, WF Harvey, KF Reid, LL Price, JB Driban, R Kalish, R Rones, T McAlindon. Effect of tai chi versus aerobic exercise for fibromyalgia: comparative effectiveness randomized controlled trial. BMJ (Clinical research ed), 2018. [DOI | PubMed]
- CP Andrade, AR Zamunér, M Forti, NY Tamburús, E Silva. Effects of aquatic training and detraining on women with fibromyalgia: controlled randomized clinical trial. Eur J Phys Rehab Med, 2019. [DOI]
- R Izquierdo-Alventosa, M Inglés, S Cortés-Amador, L Gimeno-Mallench, J Chirivella-Garrido, J Kropotov, P Serra-Añó. Low-Intensity Physical Exercise Improves Pain Catastrophizing and Other Psychological and Physical Aspects in Women with Fibromyalgia: A Randomized Controlled Trial. Int J Environ Res Public Health, 2020. [DOI | PubMed]
- M Serrat, JP Sanabria-Mazo, M Almirall, M Musté, A Feliu-Soler, JL Méndez-Ulrich, A Sanz, JV Luciano. Effectiveness of a Multicomponent Treatment Based on Pain Neuroscience Education, Therapeutic Exercise, Cognitive Behavioral Therapy, and Mindfulness in Patients With Fibromyalgia (FIBROWALK Study): A Randomized Controlled Trial. Phys Ther, 2021. [DOI | PubMed]
- 52.Gentile, E., Quitadamo, S. G., Clemente, L., Bonavolontà, V., Lombardi, R., Lauria, G., Greco, G., Fischetti, F., & De Tommaso, M. (2023). A multicomponent physical activity home-based intervention for fibromyalgia patients: effects on clinical and skin biopsy features. Clinical and experimental rheumatology, 10.55563/clinexprheumatol/iukp4c. Advance online publication. 10.55563/clinexprheumatol/iukp4c
- MY Vellisca, JI Latorre. Monosodium glutamate and aspartame in perceived pain in fibromyalgia. Rheumatol Int, 2014. [DOI | PubMed]
- M Slim, EP Calandre, JM Garcia-Leiva, F Rico-Villademoros, R Molina-Barea, CM Rodriguez-Lopez, P Morillas-Arques. The Effects of a Gluten-free Diet Versus a Hypocaloric Diet Among Patients With Fibromyalgia Experiencing Gluten Sensitivity-like Symptoms: A Pilot, Open-Label Randomized Clinical Trial. J Clin Gastroenterol, 2017. [DOI | PubMed]
- P Roman, AF Estévez, A Miras, N Sánchez-Labraca, F Cañadas, AB Vivas, D Cardona. A Pilot Randomized Controlled Trial to Explore Cognitive and Emotional Effects of Probiotics in Fibromyalgia. Sci Rep, 2018. [DOI | PubMed]
- IS Mauro-Martín, L Collado-Yurrita, S Sanz-Rojo. Short-time strategy for fibromyalgia treatment based on olive nutraceutical and Inflammatory Gut-Brain Axis Control Diet (IGUBAC) Diet. Curr Top Nutraceutical Res, 2019. [DOI]
- G Pagliai, B Colombini, M Dinu, A Whittaker, A Masoni, G Danza, A Amedei, G Ballerini, S Benedettelli, F Sofi. Effectiveness of a Khorasan Wheat-Based Replacement on Pain Symptoms and Quality of Life in Patients with Fibromyalgia. Pain Med (Malden, Mass.), 2020. [DOI]
- I Casini, V Ladisa, L Clemente, M Delussi, E Rostanzo, S Peparini, AM Aloisi, M De Tommaso. A personalized Mediterranean Diet improves pain and quality of life in patients with fibromyalgia. Pain Ther, 2024. [DOI | PubMed]
- AJ Fagerlund, OA Hansen, PM Aslaksen. Transcranial direct current stimulation as a treatment for patients with fibromyalgia: a randomized controlled trial. Pain, 2015. [DOI | PubMed]
- AF Silva, M Zortea, S Carvalho, J Leite, IL Torres, F Fregni, W Caumo. Anodal transcranial direct current stimulation over the left dorsolateral prefrontal cortex modulates attention and pain in fibromyalgia: randomized clinical trial. Sci Rep, 2017. [DOI | PubMed]
- VSDSD Santos, M Zortea, RL Alves, CCDS Naziazeno, JS Saldanha, SDCR Carvalho, AJDC Leite, ILDS Torres, A Souza, PÜ Calvetti, F Fregni, W Caumo. Cognitive effects of transcranial direct current stimulation combined with working memory training in fibromyalgia: a randomized clinical trial. Sci Rep, 2018. [DOI | PubMed]
- AP Brietzke, M Zortea, F Carvalho, PRS Sanches, DPJ Silva, ILDS Torres, F Fregni, W Caumo. Large Treatment Effect With Extended Home-Based Transcranial Direct Current Stimulation Over Dorsolateral Prefrontal Cortex in Fibromyalgia: A Proof of Concept Sham-Randomized Clinical Study. J Pain, 2020. [DOI | PubMed]
- L Castelo-Branco, E Uygur Kucukseymen, D Duarte, MM El-Hagrassy, C Bonin Pinto, ME Gunduz, A Cardenas-Rojas, K Pacheco-Barrios, Y Yang, P Gonzalez-Mego, A Estudillo-Guerra, L Candido-Santos, I Mesia-Toledo, H Rafferty, W Caumo, F Fregni. Optimised transcranial direct current stimulation (tDCS) for fibromyalgia-targeting the endogenous pain control system: a randomised, double-blind, factorial clinical trial protocol. BMJ Open, 2019. [DOI | PubMed]
- MGL Matias, D Germano Maciel, IM França, MS Cerqueira, TCLA Silva, AH Okano, R Pegado, WH Brito Vieira. Transcranial Direct Current Stimulation Associated With Functional Exercise Program for Treating Fibromyalgia: A Randomized Controlled Trial. Arch Phys Med Rehabil, 2022. [DOI | PubMed]
- R Arroyo-Fernández, J Avendaño-Coy, R Velasco-Velasco, R Palomo-Carrión, E Bravo-Esteban, A Ferri-Morales. Effectiveness of Transcranial Direct Current Stimulation Combined With Exercising in People With Fibromyalgia: A Randomized Sham-Controlled Clinical Trial. Arch Phys Med Rehabil, 2022. [DOI | PubMed]
- N Samartin-Veiga, AJ González-Villar, M Pidal-Miranda, A Vázquez-Millán, MT Carrillo-de-la-Peña. Active and sham transcranial direct current stimulation (tDCS) improved quality of life in female patients with fibromyalgia. Qual Life Res, 2022. [DOI | PubMed]
- N Samartin-Veiga, M Pidal-Miranda, AJ González-Villar, C Bradley, L Garcia-Larrea, AT O’Brien, MT Carrillo-de-la-Peña. Transcranial direct current stimulation of 3 cortical targets is no more effective than placebo as treatment for fibromyalgia: a double-blind sham-controlled clinical trial. Pain, 2022. [DOI | PubMed]
- W Caumo, RL Alves, P Vicuña, CFDS Alves, L Ramalho, PRS Sanches, DP Silva, IL da Silva Torres, F Fregni. Impact of Bifrontal Home-Based Transcranial Direct Current Stimulation in Pain Catastrophizing and Disability due to Pain in Fibromyalgia: A Randomized, Double-Blind Sham-Controlled Study. J Pain, 2022. [DOI | PubMed]
- EH Loreti, AM Freire, A Alexandre da Silva, E Kakuta, UR Martins Neto, EC Konkiewitz. Effects of Anodal Transcranial Direct Current Stimulation on the Primary Motor Cortex in Women With Fibromyalgia: A Randomized, Triple-Blind Clinical Trial. Neuromodulation J Int Neuromodulation Soc, 2023. [DOI]
- B Schein, G Beltran, BR França, PRS Sanches, DP Silva, IL Torres, F Fegni, W Caumo. Effects of Hypnotic Analgesia and Transcranial Direct Current Stimulation on Pain Tolerance and Corticospinal Excitability in Individuals with Fibromyalgia: A Cross-Over Randomized Clinical Trial. J Pain Res, 2023. [DOI | PubMed]
- L Boyer, A Dousset, P Roussel, N Dossetto, S Cammilleri, V Piano, S Khalfa, O Mundler, A Donnet, E Guedj. rTMS in fibromyalgia: a randomized trial evaluating QoL and its brain metabolic substrate. Neurology, 2014. [DOI | PubMed]
- BM Fitzgibbon, KE Hoy, LA Knox, EK Guymer, G Littlejohn, D Elliot, LE Wambeek, S McQueen, KA Elford, SJ Lee, PG Enticott, PB Fitzgerald. Evidence for the improvement of fatigue in fibromyalgia: A 4-week left dorsolateral prefrontal cortex repetitive transcranial magnetic stimulation randomized-controlled trial. Eur J Pain (London, England), 2018. [DOI]
- EU Altas, A Askin, L Beşiroğlu, A Tosun. Is high-frequency repetitive transcranial magnetic stimulation of the left primary motor cortex superior to the stimulation of the left dorsolateral prefrontal cortex in fibromyalgia syndrome?. Somatosens Mot Res, 2019. [DOI | PubMed]
- CM Cheng, SJ Wang, TP Su, MH Chen, JC Hsieh, ST Ho, YM Bai, NT Kao, WH Chang, CT Li. Analgesic effects of repetitive transcranial magnetic stimulation on modified 2010 criteria-diagnosed fibromyalgia: Pilot study. Psych Clin Neurosci, 2019. [DOI]
- S Tanwar, B Mattoo, U Kumar, R Bhatia. Repetitive transcranial magnetic stimulation of the prefrontal cortex for fibromyalgia syndrome: a randomised controlled trial with 6-months follow up. Adv Rheumatol (London, England), 2020. [DOI]
- I Bilir, A Askin, I Sengul, A Tosun. Effects of High-Frequency Neuronavigated Repetitive Transcranial Magnetic Stimulation in Fibromyalgia Syndrome: A Double-Blinded, Randomized Controlled Study. Am J Phys Med Rehabil, 2021. [DOI | PubMed]
- B Forogh, H Haqiqatshenas, T Ahadi, S Ebadi, V Alishahi, S Sajadi. Repetitive transcranial magnetic stimulation (rTMS) versus transcranial direct current stimulation (tDCS) in the management of patients with fibromyalgia: A randomized controlled trial. Neurophysiol Clin Clin Neurophysiol, 2021. [DOI]
- A Lacroix, P Vergne-Salle, JC Dumont, A Labrunie, P Balestrat, B Calvet, M Girard. Effectiveness of Repetitive Transcranial Magnetic Stimulation on Fibromyalgia Patients Responding to a First Repetitive Transcranial Magnetic Stimulation Induction Course After Six Months of Maintenance Treatment: A Randomized Pilot-Controlled Study. Neuromodulation J Int Neuromodulation Soc, 2022. [DOI]
- Y Argaman, Y Granovsky, E Sprecher, A Sinai, D Yarnitsky, I Weissman-Fogel. Clinical Effects of Repetitive Transcranial Magnetic Stimulation of the Motor Cortex Are Associated With Changes in Resting-State Functional Connectivity in Patients With Fibromyalgia Syndrome. J Pain, 2022. [DOI | PubMed]
- JL Pareja, O Cáceres, P Zambrano, F Martín, FJ Berral, M Blanco. Treatment with low-intensity transcranial magnetic stimulation in women with fibromyalgia improves diagnostic variables up to 6 months after treatment completion. Clin Exp Rheumatol, 2022. [DOI | PubMed]
- B Walitt, P Klose, MA Fitzcharles, T Phillips, W Häuser. Cannabinoids for fibromyalgia. Cochrane Database System Rev, 2016. [DOI | PubMed]
- J Sörensen, A Bengtsson, E Bäckman, KG Henriksson, M Bengtsson. Pain analysis in patients with fibromyalgia. Effects of intravenous morphine, lidocaine, and ketamine. Scandinavian J Rheumatol, 1995. [DOI]
- NH Strand, J Maloney, M Kraus, C Wie, M Turkiewicz, DA Gomez, O Adeleye, MW Harbell. Cannabis for the Treatment of Fibromyalgia: A Systematic Review. Biomedicines, 2023. [DOI | PubMed]
- J Sörensen, A Bengtsson, J Ahlner, KG Henriksson, L Ekselius, M Bengtsson. Fibromyalgia–are there different mechanisms in the processing of pain? A double blind crossover comparison of analgesic drugs. J Rheumatol, 1997. [PubMed]
- G Biasi, S Manca, S Manganelli, R Marcolongo. Tramadol in the fibromyalgia syndrome: a controlled clinical trial versus placebo. Int J Clin Pharmacol Res, 1998. [PubMed]
- IJ Russell, M Kamin, RM Bennett, TJ Schnitzer, JA Green, WA Katz. Efficacy of tramadol in treatment of pain in fibromyalgia. J Clin Rheumatol Pract Rep Rheum Musculoskeletal Dis, 2000. [DOI]
- RM Bennett, M Kamin, R Karim, N Rosenthal. Tramadol and acetaminophen combination tablets in the treatment of fibromyalgia pain: a double-blind, randomized, placebo-controlled study. Am J Med, 2003. [DOI | PubMed]
- JW Younger, AJ Zautra, ET Cummins. Effects of naltrexone on pain sensitivity and mood in fibromyalgia: no evidence for endogenous opioid pathophysiology. PLoS ONE, 2009. [DOI | PubMed]
- V Schweiger, A Martini, P Bellamoli, K Donadello, C Schievano, GD Balzo, P Sarzi-Puttini, M Parolini, E Polati. Ultramicronized Palmitoylethanolamide (um-PEA) as Add-on Treatment in Fibromyalgia Syndrome (FMS): Retrospective Observational Study on 407 Patients. CNS Neurol Disord: Drug Targets, 2019. [DOI | PubMed]
- WJ Reynolds, H Moldofsky, P Saskin, FA Lue. The effects of cyclobenzaprine on sleep physiology and symptoms in patients with fibromyalgia. J Rheumatol, 1991. [PubMed]
- R Del Giorno, S Skaper, A Paladini, G Varrassi, S Coaccioli. Palmitoylethanolamide in Fibromyalgia: Results from Prospective and Retrospective Observational Studies. Pain Ther, 2015. [DOI | PubMed]
- M Rossini, O Di Munno, G Valentini, G Bianchi, G Biasi, E Cacace, D Malesci, G La Montagna, O Viapiana, S Adami. Double-blind, multicenter trial comparing acetyl l-carnitine with placebo in the treatment of fibromyalgia patients. Clin Exp Rheumatol, 2007. [PubMed]
- S Chiechio, A Caricasole, E Barletta, M Storto, MV Catania, A Copani, M Vertechy, R Nicolai, M Calvani, D Melchiorri, F Nicoletti. L-Acetylcarnitine induces analgesia by selectively up-regulating mGlu2 metabotropic glutamate receptors. Mol Pharmacol, 2002. [DOI | PubMed]
- G Bersani, G Meco, A Denaro, D Liberati, C Colletti, R Nicolai, FS Bersani, A Koverech. L-Acetylcarnitine in dysthymic disorder in elderly patients: a double-blind, multicenter, controlled randomized study vs. fluoxetine European neuropsychopharmacology. J Eur College Neuropsychopharmacol, 2013. [DOI]
- R Zanardi, E Smeraldi. A double-blind, randomised, controlled clinical trial of acetyl-L-carnitine vs. amisulpride in the treatment of dysthymia European. neuropsychopharmacology. J Eur College Neuropsychopharmacol, 2006. [DOI]
- SC Hayes, JB Luoma, FW Bond, A Masuda, J Lillis. Acceptance and commitment therapy: model, processes and outcomes. Behav Res Ther, 2006. [DOI | PubMed]
- K Thieme, DC Turk, RH Gracely, H Flor. Differential psychophysiological effects of operant and cognitive behavioural treatments in women with fibromyalgia. Eur J Pain (London, England), 2016. [DOI]
- JV Luciano, R Neblett, C Peñacoba. The Contribution of the Psychologist in the Assessment and Treatment of Fibromyalgia. Curr Treat Options Rheum, 2023. [DOI]
- HL Heller, AR Borges, LOA Franco, JP De Oliveira Aucelio. Role of cognitive behavioral therapy in fibromyalgia: A systematic review. Open J Rheumatol Autoimmune Dis, 2021. [DOI]
- J Kabat-Zinn. Mindfulness-based interventions in context: Past, present, and future. Clin Psychol Sci Pract, 2003. [DOI]
- E Cash, P Salmon, I Weissbecker, WN Rebholz, R Bayley-Veloso, LA Zimmaro, A Floyd, E Dedert, SE Sephton. Mindfulness meditation alleviates fibromyalgia symptoms in women: results of a randomized clinical trial. Annal behav Med Public Soc Behav Med, 2015. [DOI]
- G Varallo, R Cattivelli, EM Giusti, G Landi, C Spatola, GM Ruggiero, C Franceschini, E Tossani, S Grandi, P Capodaglio, G Castelnuovo. The efficacy of a brief acceptance-based group intervention in a sample of female patients with fibromyalgia and comorbid obesity: a randomised controlled trial. Clin Exp Rheumatol, 2023. [DOI | PubMed]
- F Wolfe, HA Smythe, MB Yunus, RM Bennett, C Bombardier, DL Goldenberg, P Tugwell, SM Campbell, M Abeles, P Clark. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheumatism, 1990. [DOI | PubMed]
- J Bidonde, AJ Busch, B Bath, S Milosavljevic. Exercise for adults with fibromyalgia: an umbrella systematic review with synthesis of best evidence. Curr Rheumatol Rev, 2014. [DOI | PubMed]
- G Descalzi, D Ikegami, T Ushijima, EJ Nestler, V Zachariou, M Narita. Epigenetic mechanisms of chronic pain. Trends Neurosci, 2015. [DOI | PubMed]
- LA Abdulkhaleq, MA Assi, R Abdullah, M Zamri-Saad, YH Taufiq-Yap, MNM Hezmee. The crucial roles of inflammatory mediators in inflammation: A review. Vet World, 2018. [DOI | PubMed]
- AF Speciani, G Piuri. Measuring Biomarkers for an Innovative Personal Food Profile. J Am Coll Nutr, 2015. [DOI | PubMed]
- M Kurapatti, D Carreira. Diet Composition’s Effect on Chronic Musculoskeletal Pain: A Narrative Review. Pain Physician, 2023. [DOI | PubMed]
- FZ Kadayifci, MJ Bradley, AM Onat, HN Shi, S Zheng. Review of nutritional approaches to fibromyalgia. Nutr Rev, 2022. [DOI | PubMed]
- Evidence-Based Guidelines and Secondary Meta-Analysis for the Use of Transcranial Direct Current Stimulation in Neurological and Psychiatric Disorders. Int J Neuropsychopharmacol, 2021. [DOI | PubMed]
- JP Lefaucheur, A Antal, R Ahdab, D Ciampi de Andrade, F Fregni, EM Khedr, M Nitsche, W Paulus. The use of repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) to relieve pain. Brain Stimul, 2008. [DOI | PubMed]
- NE O’Connell, L Marston, S Spencer, LH DeSouza, BM Wand. Non-invasive brain stimulation techniques for chronic pain. Cochrane Database System Rev, 2018. [DOI | PubMed]
- M Zortea, L Ramalho, RL Alves, CFDS Alves, G Braulio, ILDS Torres, F Fregni, W Caumo. Transcranial Direct Current Stimulation to Improve the Dysfunction of Descending Pain Modulatory System Related to Opioids in Chronic Non-cancer Pain: An Integrative Review of Neurobiology and Meta-Analysis. Front Neurosci, 2019. [DOI | PubMed]
- 114.Lefaucheur, J. P., André-Obadia, N., Antal, A., Ayache, S. S., Baeken, C., Benninger, D. H., Cantello, R. M., Cincotta, M., de Carvalho, M., De Ridder, D., Devanne, H., Di Lazzaro, V., Filipović, S. R., Hummel, F. C., Jääskeläinen, S. K., Kimiskidis, V. K., Koch, G., Langguth, B., Nyffeler, T., Oliviero, A., … Garcia-Larrea, L. (2014). Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS). Clinical neurophysiology : official journal of the In
- R Izquierdo-Alventosa, M Inglés, S Cortés-Amador, L Gimeno-Mallench, N Sempere-Rubio, P Serra-Añó. Effectiveness of High-Frequency Transcranial Magnetic Stimulation and Physical Exercise in Women With Fibromyalgia: A Randomized Controlled Trial. Phys Ther, 2021. [DOI | PubMed]
- J Ablin, MA Fitzcharles, D Buskila, Y Shir, C Sommer, W Häuser. Treatment of fibromyalgia syndrome: recommendations of recent evidence-based interdisciplinary guidelines with special emphasis on complementary and alternative therapies. Evid-Based Complemen Alternat Med: eCAM, 2013. [DOI]
- LM Arnold, DJ Clauw. Challenges of implementing fibromyalgia treatment guidelines in current clinical practice. Postgrad Med, 2017. [DOI | PubMed]
- M Uçar, Ü Sarp, Ö Karaaslan, AI Gül, N Tanik, HO Arik. Health anxiety and depression in patients with fibromyalgia syndrome. J Int Med Res, 2015. [DOI | PubMed]
