Canadian Network for Mood and Anxiety Treatments 2024 Clinical Practice Guideline for the Management of Perinatal Mood, Anxiety, and Related Disorders: Guide de pratique 2024 du Canadian Network for Mood and Anxiety Treatments pour le traitement des troubles de l’humeur, des troubles anxieux et des troubles connexes périnatals
Abstract
Background:
The Canadian Network for Mood and Anxiety Treatments (CANMAT) publishes clinical practice guidelines for mood and anxiety disorders. This CANMAT guideline aims to provide comprehensive clinical guidance for the pregnancy and postpartum (perinatal) management of mood, anxiety and related disorders.
Methods:
CANMAT convened a core editorial group of interdisciplinary academic clinicians and persons with lived experience (PWLE), and 3 advisory panels of PWLE and perinatal health and perinatal mental health clinicians. We searched for systematic reviews of prevention and treatment interventions for perinatal depressive, bipolar, anxiety, obsessive–compulsive and post-traumatic stress disorders (January 2013–October 2023). We prioritized evidence from reviews of randomized controlled trials (RCTs), except for the perinatal safety of medications where reviews of large high-quality observational studies were prioritized due to the absence of RCT data. Targeted searches for individual studies were conducted when systematic reviews were limited or absent. Recommendations were organized by lines of treatment based on CANMAT-defined levels of evidence quality, supplemented by editorial group consensus to balance efficacy, safety, tolerability and feasibility considerations.
Results:
The guideline covers 10 clinical sections in a question-and-answer format that maps onto the patient care journey: case identification; organization and delivery of care; non-pharmacological (lifestyle, psychosocial, psychological), pharmacological, neuromodulation and complementary and alternative medicine interventions; high-risk clinical situations; and mental health of the father or co-parent. Equity, diversity and inclusion considerations are provided.
Conclusions:
This guideline’s detailed evidence-based recommendations provide clinicians with key information to promote the delivery of effective and safe perinatal mental healthcare. It is hoped that the guideline will serve as a valuable tool for clinicians in Canada and around the world to help optimize clinical outcomes in the area of perinatal mental health.
Plain Language Summary Title:
The Canadian Network for Mood and Anxiety Treatments 2024 Guideline for Helping People with Mood, Anxiety and Related Disorders During Pregnancy and Postpartum
Article type: Review Article
Keywords: clinical practice guidelines, depression, postpartum depression, mood disorder, anxiety disorder, obsessive–compulsive disorder, post-traumatic stress disorder, bipolar disorder, perinatal, postpartum psychosis, pregnancy, postpartum period
Affiliations: Department of Psychiatry and Women’s College Research and Innovation Institute, Women’s College Hospital, Toronto, ON, Canada; Department of Psychiatry, 12366Temerty Faculty of Medicine, 7938University of Toronto, Toronto, ON, Canada; Institute for Health Policy, Management and Evaluation, 274071Dalla Lana School of Public Health, 7938University of Toronto, Toronto, ON, Canada; Department of Psychiatry and Behavioural Neurosciences, 3710McMaster University, Hamilton ON, Canada; Women’s Health Concerns Clinic, 25479St. Joseph’s Healthcare Hamilton, Hamilton, ON, Canada; Department of Psychiatry, Sunnybrook Health Sciences Centre, Toronto, ON, Canada; Department of Health and Society, 33530University of Toronto, Scarborough, ON, Canada; Waypoint Centre for Mental Health Care, Penetanguishene, ON, Canada; Lawrence Bloomberg Faculty of Nursing, 7938University of Toronto, Toronto, ON, Canada; Lunenfeld-Tanenbaum Research Institute, Mount Sinai Hospital, Toronto, ON, Canada; Department of Family Practice, 8166University of British Columbia, Vancouver, BC, Canada; Michael Smith Foundation for Health Research, Vancouver, BC, Canada; Faculty of Nursing and Cumming School of Medicine, 8166University of Calgary; Department of Pediatrics, 120479University of British Columbia, Vancouver, BC, Canada; School of Population and Public Health, 8166University of British Columbia, Vancouver, BC, Canada; BC Children’s Hospital Research Institute, 8166University of British Columbia, Vancouver, BC, Canada; Department of Psychiatry, 6221Western University, London, ON, Canada; Department of Obstetrics and Gynecology, 6221Western University, London, ON, Canada; Campbell Family Mental Health Research Institute, 7978Centre for Addiction and Mental Health, Toronto, ON, Canada; Toronto General Hospital Research Institute, 7989Centre for Mental Health, University Health Network, Toronto, ON, Canada; Canadian Perinatal Mental Health Collaborative, Barrie, ON, Canada; Department of Educational and Counselling Psychology, and Special Education, 8166University of British Columbia, Vancouver, BC, Canada; Department of Medicine, 5620McGill University, Montreal, QC, Canada; Centre for Outcomes Research and Evaluation, 507266Research Institute of the McGill University Health Centre, Montreal, QC, Canada; Department of Psychiatry, 5620McGill University, Montreal, QC, Canada; Department of Psychiatry, 4257Queen’s University and Providence Care Hospital, Kingston, ON, Canada; Department of Psychiatry, 8166University of British Columbia, Vancouver, BC, Canada; Department of Psychiatry, 1259University of Michigan, Ann Arbour, MI, USA; Department of Psychiatry, 2129University of Calgary, Calgary, AB, Canada; Department of Psychiatry, 26612Montreal Institute of Mental Health, Université de Montréal, Montréal, QC, Canada
License: © The Author(s) 2025 CC BY 4.0 This article is distributed under the terms of the Creative Commons Attribution 4.0 License (https://creativecommons.org/licenses/by/4.0/) which permits any use, reproduction and distribution of the work without further permission provided the original work is attributed as specified on the SAGE and Open Access page (https://us.sagepub.com/en-us/nam/open-access-at-sage).
Article links: DOI: 10.1177/07067437241303031 | PubMed: 39936923 | PMC: PMC11985483
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (2.6 MB)
Introduction
Mood, anxiety and related disorders are some of the most common conditions that arise in pregnancy and in the year after childbirth.1bibr2-07067437241303031–3 When untreated or undertreated, perinatal mood, anxiety and related disorders (PMADs) can negatively impact the well-being and quality of life of affected persons, and the health and development of their children and families.3,4 This makes the identification and management of PMADs a public health priority.
Clinical practice guidelines can synthesize best practices to help guide the delivery of timely, effective, high-quality care.5 The Canadian Network for Mood and Anxiety Treatments (CANMAT) is a not-for-profit scientific and educational organization that produces clinical guidelines outlining the latest research and treatment options for managing mood, anxiety and related disorders (www.canmat.org). Previous CANMAT guidelines for major depressive disorder (MDD) and bipolar disorder (BD) included sections on perinatal populations.6,7 This PMAD-specific CANMAT guideline aims to provide a more comprehensive guidance on best practices in care and engage a target audience beyond psychiatrists to include all clinicians (e.g., family physicians, midwives, nurses, obstetricians, pediatricians, psychotherapists, social workers and others) who may require this knowledge for case identification, referral and/or management.
This guideline summarizes the evidence for interventions to prevent and treat PMADs, including specifically MDD, anxiety disorders, obsessive–compulsive disorders (OCD), post-traumatic stress disorder (PTSD) and BD in pregnancy and up to 1 year postpartum. Postpartum psychosis, a severe postpartum clinical presentation that is strongly associated with bipolar disorder, is also included. The guideline also covers clinical presentations of mood, anxiety and related disorders that are unique to the perinatal period such as fear of childbirth and symptoms of PTSD that arise in relation to experiences of trauma around the time of birth, sometimes termed childbirth-related PTSD (CB-PTSD). While other types of mental disorders also affect people in pregnancy and/or postpartum—on their own or comorbid with PMADs (e.g., autism, attention-deficit-hyperactivity disorder, eating disorders, personality disorders, substance and alcohol use disorders and others)—they were considered to be outside the scope of this initial guideline.
Throughout the guideline, we strive to use language inclusive of women and gender-diverse childbearing people and their partners, including those who are transgender, non-binary and two-spirit.8,9 We aimed to include evidence related to equity, diversity and inclusion in each section of the guideline, focusing on specific considerations related to age, sexual and gender identity, race, ethnicity, culture and disability.
Key Caveats
There are some key caveats to consider when reading this guideline. First, while the first year postpartum is a conventional cut-off point in the literature, many PMADs continue beyond this period. The evidence and recommendations in this guideline may still be applicable after the first year postpartum, especially when considering issues related to lactation and interventions adapted to focus on parenting or the maternal-child dyad. Second, in the PMAD literature, many studies utilize symptom scales, and not diagnostic interviews for study inclusion. While symptom scales correlate highly with diagnostic interviews, it is difficult to differentiate in some cases whether the results of such research would apply to those with diagnosed PMADs specifically.10 Many individuals with PMAD symptoms—even when they do not meet diagnostic criteria for a mental disorder per se—may benefit from treatment, but throughout the guideline we aim to distinguish between interventions evaluated in populations with elevated symptoms versus those with diagnosed disorders when possible. Third, while the guideline is meant to assist clinicians in managing PMADs using an evidence-based approach, it is not meant to replace clinical judgement nor is it a legal or policy care standard. Clinicians and decision-makers will want to consider the recommendations in the context of their own settings (e.g., availability of specific psychotherapies, medication and/or other treatments) and their local regulations of clinical practice. Fourth, the guideline evidence review is updated to October 2023, so clinicians are encouraged to keep up to date with the latest research so as to provide their patients with the best possible care. Finally, the guideline is written in technical, academic language, with the target audience being healthcare professionals. This is not necessarily easily accessible for patients or families who are trying to decipher the guidelines for their own personal educational needs, so there will be an accompanying Patient and Family Guide.
Methods
The development of the guideline followed the structured methodology of previous CANMAT guidelines.7,11 The project was led by a core editorial group of 15 PMAD experts spanning psychiatry, psychology, pediatrics, nursing and public health, and 2 persons with lived experience (PWLE). Meetings of the core group were biweekly by video-conferencing for the duration of the project (April 2023 to October 2024), supplemented by email communication. The project received input from the CANMAT Board and 3 advisory panels comprising 27 individuals with diverse experiences and backgrounds from across Canada: (a) PWLE (n = 10), (b) non-mental health clinical experts, including midwifery, obstetrics and gynecology, nursing, pediatrics and primary care (n = 8) and (c) perinatal mental health clinicians (n = 9) (Supplement 1. Advisory Panels). Advisory panel members were engaged for input and feedback through structured surveys, email communication, focus groups and 1:1 discussions with the co-leads and other members of the project team.
Scope and Structure
The current guideline follows a question-and-answer format, with a focus on clinical questions relevant to the management of PMADs (Table 1). The questions were initially drafted by the core editorial group and CANMAT Board, then revised based on feedback from the advisory panels. Advisory panel members were asked to provide input on the nature and importance of the questions, how they were worded, and the rank order in which questions should be presented. They were also asked their perspective on whether there were any key clinical questions missing and the extent to which equity, diversity and inclusion were addressed. Feedback was reviewed and incorporated by the core editorial group, who approved the final set of questions by consensus agreement. The final questions were then shared back to the advisory panel members.
Table 1.: Guideline Sections and Questions.
| Epidemiology and Impact |
| Foundations of Clinical Management |
| Initial Diagnostic Assessment |
| Diagnostic Considerations |
| General Treatment Considerations |
| What are the recommendations for the: |
| 1. Identification of perinatal mood, anxiety and related disorders? |
| 2. Organization and delivery of perinatal mental healthcare services? |
| 3. Lifestyle interventions for the prevention and treatment of PMADs? |
| 4. Use of psychosocial interventions in the prevention and treatment of PMADs? |
| 5. Use of psychological interventions in the prevention and treatment of PMADs? |
| 6. Use of pharmacological interventions in pregnancy and lactation? |
| 7. Use of neuromodulation interventions in the prevention and treatment of PMADs? |
| 8. Use of complementary and alternative treatments for the prevention and treatment of PMADs? |
| 9. Management of high-risk clinical situations? |
| 10. Identification, prevention and treatment of PMADs in fathers and co-parents? |
Note. PMADs = perinatal mood, anxiety and related disorders.
Evidence Review
PubMed, EMBASE, PsycINFO, MEDLINE, CINAHL, Cochrane and Web of Science databases were searched for English- and French-language systematic reviews and meta-analyses published from 1 January 2013 to 29 October 2023. A list of key terms and subject headings were developed by the team, and refined by a medical librarian, according to 2 concepts: (a) mood, anxiety and related disorders and (b) the perinatal life stage. Large well-conducted randomized controlled trials (RCTs) are the highest standard of evidence, so we prioritized reviews that synthesized the results of RCTs in the current guidelines. In the absence of systematic reviews of RCTs (e.g., for medication safety in pregnancy), systematic reviews of large observational studies were included. Targeted searches for single RCTs and other studies were conducted when systematic review data were limited or absent (Supplement 2. Search Strategy).
After removing duplicates, 12,718 articles were identified for title and abstract screening, 733 underwent full-text review and 470 systematic reviews of interventions (RCTs, or observational studies in the setting of medications in pregnancy only) were identified (Supplement 2). Due to a paucity of systematic reviews relevant to BD, a targeted search on case identification and pharmacotherapy was conducted, resulting in an additional 89 studies. During study selection, articles on epidemiology and key foundations of management not explicitly covered in the 10 guideline questions and other clinical practice guidelines were flagged as potential reference documents, but not included for data extraction or quality appraisal (N = 942). Study selection was completed by 2 independent reviewers, with a third reviewer to adjudicate conflicts.
Data extraction and quality appraisal were conducted in 2 stages. First, to prioritize the most contemporary evidence, data from included studies published in the 5 years prior to the end of the search date (2018–2023) were extracted and appraised. If the reviews published in the 5 years prior to the search date did not provide adequate data for recommendations to be made, data were extracted and appraised from the remaining reviews. In the end, this was done only for Question 9. In total, data were extracted from 298 of the studies included in the search using a standardized data extraction form based on the Template for Intervention Description and Replication checklist. Quality appraisal was based on the Grading of Recommendations, Assessment, Development and Evaluations framework.12,13
After data extraction and quality appraisal, the authors assigned a “Level of Evidence” to each type of prevention or treatment intervention based on study design, size and quality for the totality of the evidence, as defined in previous CANMAT guidelines (Table 2). The section leads of the given question/section (3 to 5 authors/section) reviewed the quality appraisal of the articles pertaining to their specific sections and assigned the Level of Evidence based on the criteria in Table 2. Any discordance was resolved through input of the co-leads and additional authors when necessary to reach a consensus decision. In this guideline, green circles are used to indicate that there is positive empirical evidence that the intervention is effective, and red squares are used when there is empirical evidence that the intervention is not effective. Blue circles are used to represent the strength and quality of the evidence on safety of the intervention in pregnancy and/or lactation. The blue circles do not indicate whether the safety profile is favourable or not, only the Level of Evidence available.
Table 2.: CANMAT Criteria for Level of Evidence.
| Level of evidence | Symbol | Criteria | ||
|---|---|---|---|---|
| Efficacy | Perinatal safety | |||
| Positive | Negative | |||
| 1 | High-quality meta-analyses with narrow confidence intervals or replicated RCTs with adequate sample size | |||
| 2 | Lower-quality meta-analyses with wide confidence intervals and/or one or more RCTs with adequate sample size | |||
| 3 | At least 1 small-sample RCT or high-quality, controlled observational studies | |||
| 4 | Pilot studies, uncontrolled trials, anecdotal reports or expert consensus opinion | |||
Note. For Level 1 and Level 2, RCTs are required (adequate sample size is at least n ≥ 30 per treatment arm). Recommendations from epidemiological data primarily arise from observational studies, where the highest level of evidence is usually Level 3. Higher order recommendations (e.g., principles of care) reflect higher level judgement of strength of evidence from various data sources and therefore are primarily Level 3 or 4 evidence. RCT = randomized controlled trial.
Recommendation Development Process
Each core editorial group member and CANMAT board member co-author was assigned to one or more of the guideline questions. Recommendations were developed based on the level of evidence available for efficacy and clinical support for the intervention, namely the balance between efficacy, safety, tolerability and the feasibility of applying the intervention perinatally (Table 3). For example, a treatment with Level 1 evidence for efficacy could be recommended as a second- or third-line treatment option (or not recommended) due to concern about its safety, side effect profile or feasibility of its use. In addition, as there are no RCTs of medications in pregnancy (and few in the postpartum), Level 3 and Level 4 perinatal-specific evidence on efficacy, safety and tolerability of medications was combined with efficacy and tolerability evidence from non-perinatal populations to inform the recommendations. For example, a medication with Level 1 evidence for efficacy non-perinatally for which there is also high-quality reassuring Level 3 evidence for safety in perinatal populations could be considered a first-line treatment even in the absence of perinatal-specific efficacy data. In contrast, a medication with Level 1 evidence for efficacy in non-perinatal populations but with high-quality Level 3 evidence that there are safety concerns in perinatal populations could be downgraded to second-, third-line or even “not recommended” depending on the degree/severity of the safety concern.
Table 3.: CANMAT Criteria for Line of Treatment.
| Line of treatment | Criteriaa |
|---|---|
| First-line | Level 1 or Level 2 evidence plus clinical support |
| Second-line | Level 3 evidence or higher plus clinical support |
| Third-line | Level 4 evidence or higher plus clinical support |
| Not recommended | Level 1 or 2 evidence for lack of efficacy or safety concerns, plus clinical support |
Note. CANMAT = Canadian Network for Mood and Anxiety Treatments.
Clinical support reflects the authors’ expert opinion/consensus on the relevance of the evidence on safety, efficacy and feasibility of the intervention. For pharmacological treatment recommendations, due to the paucity of perinatal-specific randomized controlled trials, Level 3 or 4 evidence on efficacy and safety in perinatal-specific populations might be considered sufficient for a first-line or second-line recommendation in the presence of Level 1 or 2 evidence for efficacy in non-perinatal populations.
Preliminary recommendations were presented to the entire authorship group in a full-day meeting in March 2024 for initial feedback (in-person, with video-conferencing options for those unable to travel to the meeting), followed by ongoing discussion and consensus meetings within Question groups, along with the drafting of text to accompany the recommendations. The 2 project co-leads (SV, BF) then reviewed and revised each section. Draft recommendations and a full draft of the guideline were completed in June 2024. The advisory panels were then re-engaged to provide feedback on the recommendations and their explanations as well as the tone, language and completeness of the guideline. A final draft of the guideline was approved by the authorship group for submission for peer review on September 13, 2024.
Epidemiology and Impact
The prevalence of depression at any point in the perinatal period ranges from 6.5 to 12.9%, with a lower rate when major depression is confirmed by structured clinical interviews.14 There is a particularly high prevalence of depression in the postpartum period, estimated at up to 19% for clinically significant depressive symptoms, and 7% for a major depressive episode (MDE).14,15 The risk for postpartum depression (PPD) is usually highest within the first 6 months of childbirth. This is inconsistent with the more narrow “peripartum onset” specifier for depressive disorders in the Diagnostic and Statistical Manual of Mental Disorders 5th Edition Text Revision (DSM-5-TR) that is limited to symptom onset in pregnancy and within the first 4 weeks postpartum.15,16
Anxiety is even more common than depression in the perinatal period, with rates of clinically significant anxiety symptoms estimated at up to 20%.2,17 The prevalence of the related clinical presentation of fear of childbirth may be as high as 14%.18 Pooled prevalence of OCD is about 2% perinatally, although transient obsessions and/or compulsions may be more common, and there have been reports of transient symptom worsening perinatally possibly in as many as 70% of those affected by the illness.19bibr20-07067437241303031–21 PTSD has an estimated pooled prevalence of about 3% in pregnancy and about 4% after birth.22
The lifetime prevalence of BD is about 2–3% in non-pregnant populations, and diagnostic interviews in pregnancy and postpartum suggest the same.23 BD has the highest relapse rate for any mental disorder in the postpartum, in the range of 30–50%; the relapse rate may be as high as 66% among those who are medication-free during pregnancy.24 Most relapses are non-psychotic MDEs but as many as 1 in 6 (17%) are episodes of psychosis, mania or MDEs severe enough to require hospitalization. Postpartum psychosis occurs after approximately 1–2 in every 1000 births overall,25 but risk is increased in those with a pre-existing diagnosis of BD, and those who have previously experienced postpartum psychosis.26,27
While a comprehensive description of risk factors for each specific PMAD is outside the scope of this guideline, Table 4 lists common risk factors and correlates.3,28bibr29-07067437241303031bibr30-07067437241303031bibr31-07067437241303031bibr32-07067437241303031bibr33-07067437241303031bibr34-07067437241303031bibr35-07067437241303031bibr36-07067437241303031bibr37-07067437241303031–38 The strongest risk factor for any PMAD is a past history of the given disorder, and untreated or undertreated symptoms prior to or during pregnancy.39 Lack of social support and stressful life events are major risk factors for perinatal depression and anxiety.3 Perinatal depression appears to be more commonly experienced by some populations, including adolescents, immigrants,40 those who live in lower and middle-income countries,41,42 are racialized,43 Indigenous,44,45 have disabilities46,47 and are gender and sexual minorities.48 Anxiety disorders also appear to be more common in low and middle-income countries41and underserved populations.17,41,49 PTSD is more common in those who have experienced pregnancy complications, stillbirth and adverse childhood experiences.22 Nulliparity and lower socioeconomic status are associated with increased risk for fear of childbirth.50,51
Table 4.: Common PMAD Risk Factors.
| Genetic, mental and physical health factors |
|---|
| Reproductive, perinatal and postpartum health factors |
| Social and environmental factors |
Note. PMAD = perinatal mood, anxiety and related disorder.
The effective treatment of PMADs is of high priority for the short- and long-term health of the affected person, their child and family. Untreated or inadequately treated perinatal depression is linked to an increased risk of future depressive episodes, substance use, partner relationship problems and suicide.52bibr53-07067437241303031bibr54-07067437241303031–55 Depression in pregnancy is associated with perinatal and newborn health complications (e.g., gestational hypertension,56 preterm birth,57,58 low birth weight58 and neonatal intensive care unit (NICU) admission in infants59). Depression in both pregnancy and postpartum are associated with lower rates of breastfeeding,60 less optimal mother-infant interactions61 and higher rates of infant and child physical illness,62 hospitalizations,63 maltreatment64 and cognitive, emotional and behavioural problems.65,66 The effects of untreated or undertreated anxiety and related disorders on the health and well-being of childbearing individuals and their offspring are similar in nature and magnitude to those of depression.3,66bibr67-07067437241303031–68 Fear of childbirth specifically is associated with higher rates of labour interventions and complications, such as emergency caesarean birth.50,51,69,70 BD is also associated with increased risks for adverse perinatal and newborn health complications and an increased risk for PPD and psychosis.71,72
Foundations of Clinical Management
Initial Diagnostic Assessment
Regardless of the manner in which a potential PMAD is identified (see Question 1), the first step in clinical management is a comprehensive diagnostic assessment. It is important that this assessment is conducted in a supportive, non-judgmental and inclusive environment. Accessible, culturally safe and trauma-informed care are all essential to a comprehensive assessment and management plan. Clinicians are encouraged to educate themselves about how to safely care for diverse populations, including those who are racialized, a gender or sexual minority, or who have a disability and others. It is important for providers to consider the systemic ways in which some populations have been historically underserved, mistreated, excluded and/or stigmatized (including in health care encounters and systems) and how the experiences, outcomes and needs of certain populations may differ as a result.
The initial diagnostic assessment includes all elements of a general psychiatric interview, including the patient’s goals and expectations with the assessment, their sociodemographic characteristics, current and lifetime mental health and substance use disorders and their severity (including hospitalizations and suicidal behaviours), general medical history, current and past treatment response to psychological, pharmacological and other interventions (as well as what happened when prior interventions were discontinued), and developmental and family history. It is important to evaluate past and current risk factors for PMADs, family and social support, the roles and responsibilities of the patient with respect to caregiving (including for other children), and financial or work obligations. It is also important to inquire about health-related factors that can influence mental health and may be targeted through lifestyle interventions, such as exercise routine, diet and sleep patterns. Clinicians will want to assess for potential impact of the co-parent’s mental health (see Question 10) and/or whether there are concerns about intimate partner violence. Mental health issues in relation to previous pregnancies and other times of hormonal change (premenstrual disorders, mood worsening with the use of hormonal contraceptives, for example) are associated with elevated risk for subsequent PMADs.36 History of pregnancy, labour and delivery and/or postpartum complications, including pregnancy loss, can also inform the diagnostic and management plan.
Clinical Pearl—Trauma-Informed care
Questions about past trauma are important to explore with sensitivity.73,74 Traumatic experiences can have long-lasting, pervasive impact on one’s mental and physical health, but many individuals do not spontaneously disclose their past traumatic experiences due to fear, shame, feelings of blame and/or lack of trust. It is not uncommon for patients who have previously experienced trauma to present as guarded, sensitive and prone to emotional dysregulation. In the perinatal period, triggers of prior traumatic experiences can specifically include physical examinations, labour pain, difficult childbirth or other clinical scenarios where the patient does not experience adequate control, choice or autonomy. Trauma-informed care—different than intervention focused on treatment of the sequelae of trauma itself—refers to when healthcare clinicians and care systems are able to acknowledge the broad impacts of trauma, recognize clinical symptoms or other evidence of trauma in the clinical setting and integrate this knowledge into how care is delivered including by creating a safe environment, promoting collaborative treatment relationships and decisions and taking practical steps to reduce the likelihood that the treatment environment will mirror common characteristics of past traumatic experiences.73,74 Given the high prevalence of trauma, trauma-informed care is important at every stage of the assessment and treatment process for all persons with PMADs.
It is important to explore how and when a patient’s symptoms may have started or changed in pregnancy and/or in the postpartum, to identify potentially addressable factors that may have led to the development or worsening of illness. This may help identify whether there has been an increase in stressful life events, whether medication discontinuation may have precipitated relapse or whether there is a need for medication adjustment due to the physiological changes of pregnancy and/or the postpartum period. In the postpartum, exploring infant/newborn health can similarly inform diagnosis and treatment (i.e., preterm, difficult labour/delivery, medication withdrawal, feeding, sleeping, irritability, colic and sibling relationships).
The initial diagnostic assessment should also include a safety risk assessment, including thoughts of self-harm or harm to the infant/others (see Question 9). When possible and appropriate to obtain, collateral information from family members, friends and/or acquaintances can be helpful in clarifying diagnosis and risk. Thyroid-stimulating hormone (TSH) and complete blood count (CBC) or ferritin measurement can assist in ruling out common conditions that occur in pregnancy and postpartum such as thyroid disease or iron deficiency/anaemia, respectively. Other laboratory tests may be required on a case-by-case basis (e.g., Vitamin D levels with suspicion of Vitamin D deficiency). Additional testing and/or the use of electroencephalography (EEG) or neuroimaging are generally only indicated when there is clinical suspicion of epilepsy/seizures, traumatic brain injury or other serious medical conditions that can cause neuropsychiatric symptoms (e.g., anti-N-methyl-D-aspartate, anti-NMDA, receptor encephalitis or posterior reversible encephalopathy syndrome).
Diagnostic Considerations
Not all mood and anxiety symptoms experienced perinatally necessarily represent a psychiatric disorder. The “baby blues,” reported by 40% to 80% of individuals shortly after childbirth, includes symptoms such as mild mood swings, sadness, crying more easily, anxiety, and difficulties with sleep and concentration.75 Symptoms associated with the “baby blues” are neither persistent nor severe and usually improve significantly or disappear within a few weeks without intervention. Although less well-studied than the “baby blues,” the concept of “baby pinks” which appears to include mild symptoms of mood lability, irritability, feeling euphoric/elated, sleeplessness, increased energy and over-talkativeness, has also been reported.76 Like the “baby blues,” symptoms of the “baby pinks” also appear to start within the first 2 weeks postpartum, are mild, do not negatively impact daily functioning and improve without treatment. Longitudinal studies show that most cases of “baby blues” and “baby pinks” spontaneously remit and do not require medical intervention. However, some studies suggest a higher risk of PPD at 6–8 weeks postpartum in individuals who experience them.75,76 Therefore, CANMAT recommends monitoring those who experience “baby blues/pinks” carefully until resolution of symptoms (Level 4 ). In cases of pregnancy or child health complications, unexpected occurrences during labour and delivery that are experienced as traumatic, difficulty with breastfeeding or other major stressors, a DSM-5-TR diagnosis of adjustment disorder may be more appropriate than one of a PMAD, if criteria for another mood, anxiety or related disorder are not met.
In the DSM-5-TR, PMADs do not have their own diagnostic criteria, chapter or category.16 There is a specifier of “peripartum onset” for MDD, BD I and BD II that is used when the onset of these disorders occurs in pregnancy or within the first 4 weeks postpartum. In practice, those with onset of mental illness prior to pregnancy will also require treatment perinatally. Further, as mental illness can also arise after the first 4 weeks postpartum, both clinicians and researchers usually consider disorders to be “perinatal” when they present in individuals up to at least 1 year postpartum.77 Despite their high prevalence, there are no peripartum onset specifier for anxiety disorders, OCD or PTSD at this time.
Although there are no specific DSM-5-TR symptom criteria for perinatal populations, there are some perinatal-specific aspects of the clinical presentations. In depression, highly negative and persistent ruminations about one’s capacity as a parent and guilt about actions having a negative impact on the child are common. Anxiety—whether as part of a depressive, anxiety or related disorder—often focuses on worries related to the health of babies and the ability to parent.78 The intrusive nature of excessive worrying perinatally in depression and anxiety disorders may sometimes be difficult to distinguish from that of OCD (“obsessive worrying”), which is characterized by intrusive thoughts and/or compulsive behaviour. In any of these conditions, intrusive worries in the perinatal population can present as unwanted, intrusive thoughts or images of harm coming to the infant, often accompanied by reassurance seeking, checking behaviour and avoidance (e.g., avoiding letting others look after the baby).19 Fear of childbirth (also referred to as tokophobia) is an increasingly recognized clinical presentation of anxiety that encompasses many different fears related to pregnancy and childbirth, ranging from fear of pain, medical interventions and injury, to fear of loss of control, body change and staff misconduct, to fear of maternal and/or infant death.18,51 CB-PTSD, where PTSD symptoms focus on the birth and reminders of the birth in the context of a traumatic birthing experience, is now increasingly being described.79 It is also increasingly being recognized that childbirth can be experienced as traumatic and lead to poor mental well-being even when there were no serious obstetrical or neonatal complications per se.
Although this guideline is specifically focused on the management of PMADs, it is critical for clinicians to consider that mood and anxiety symptoms may be associated with other psychiatric conditions, including personality disorders (e.g., borderline personality disorder), eating disorders (e.g., anorexia, bulimia, binge eating) and/or alcohol and substance use disorders. In fact, in some cases, these psychiatric disorders other than PMADs may be the primary source of mood and anxiety symptoms. Whether psychiatric disorders other than PMADs are primary or comorbid with PMADs, they need to be fully assessed and managed. Finally, in Canada, clinicians have the duty to report to child protection services or the police (when urgent) in situations where there are concerns about child abuse and/or neglect. CANMAT recommends all clinicians to be aware of the laws of their local authorities.
General Treatment Considerations
It is ideal for mental illness to be stabilized prior to conception. In preconception planning, it is important to provide patients with updated information to help them balance the potential risks of their illness (or illness recurrence) with various treatment options when making decisions about plans for their treatment before, during and after pregnancy. In pregnancy and postpartum, the main goals of treatment are to achieve symptom remission, prevent recurrence or worsening, reduce or eliminate symptoms of comorbid disorders, including medical, psychiatric and substance or alcohol use disorders, and minimize any risks to the developing infant or child. Healthcare providers are encouraged to provide support for the management of psychosocial issues, which may include connecting patients to social services early in treatment planning. This may include connecting the patient with resources to assist with food insecurity, financial and/or housing instability, language classes for those who do not speak the local language, and/or supporting patients experiencing intimate partner violence. For individuals presenting with more than one psychiatric disorder, it is important to prioritize which disorder(s) need(s) to be addressed first. Factors to consider include the type and severity of disorders, symptom profile (e.g., presence of psychotic symptoms), impact on maternal distress and functioning, and safety concerns. Collaboration between multidisciplinary team members is encouraged in preparation for labour, delivery and the early postpartum period, especially in complex cases.
Initial treatment selection depends on the nature and severity of the illness, previous response to treatment and patient preference. Severity (mild, moderate or severe) can be assessed based on the number of symptoms that a patient is presenting with, the severity of those symptoms and the social and occupational functional impact (Figure 1).16 For depression and anxiety symptom severity specifically, scores on brief patient-oriented outcome measures commonly and feasibly utilized in clinical practice such as the Edinburgh Postnatal Depression Scale (EPDS), Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder 7-item (GAD-7) may be used to aid in the severity assessment.80 The cut-off scores on these measures for the various severity levels in Figure 1 are meant only to be used to guide the severity assessment, and should be considered in the context of other aspects of the clinical picture (e.g., history, duration of symptoms, other comorbidities) to aid in decision-making about treatment.
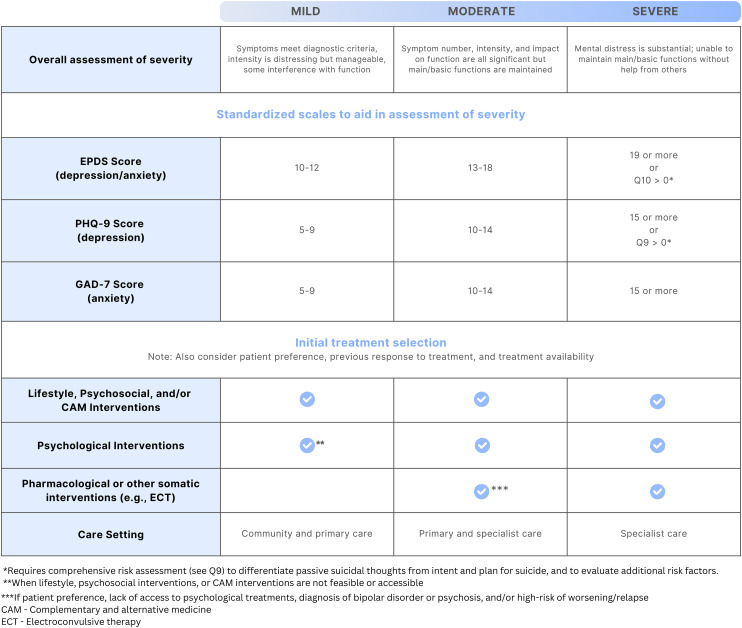
As shown in Figure 1, depression, anxiety and related disorders of mild or moderate severity can often be successfully managed with non-pharmacological options alone, including with lifestyle, psychosocial, psychological and complementary and alternative medicine (CAM) interventions. Medications and other somatic treatments may then be used in addition when symptoms do not respond to non-pharmacological treatment alone. As also shown in Figure 1, medications are typically needed to successfully manage depression, anxiety and related disorders when symptoms are of moderate-severity or severe, and in the treatment of BD. They are also used to prevent relapse or recurrence of symptoms when an individual was taking medication for maintenance treatment prior to pregnancy. In these cases, non-pharmacological interventions may also be implemented to complement the pharmacological treatment (e.g., peer support programs or psychological therapy may still be beneficial alongside a medication treatment). Other somatic treatments, such as non-invasive neuromodulation or electroconvulsive therapy (ECT) may be needed when medication options have not led to adequate remission, or for ECT when there is very severe illness and a rapid response to treatment is required (e.g., acute psychosis or catatonia).
When discussing treatment options, healthcare providers should provide ample time for discussion, including possible risks, side effects and expected benefits. Patient preference should also be taken into account. Health professionals should also encourage patients to discuss the treatment options with their partners/close support and/or to bring their partners/close support to an appointment so that treatment options can be discussed jointly, as appropriate.
Once treatment is initiated, CANMAT recommends regular monitoring. The routine use of standardized tools measuring the severity of symptoms such as the EPDS, PHQ-9 and the GAD-7 is highly encouraged to systematically monitor response to treatment and potential symptom worsening or relapse. Monitoring should occur more frequently while the patient remains symptomatic (e.g., every 1–3 weeks) and the frequency can be reduced once the symptoms have fully remitted (e.g., every 6–8 weeks) (Level 4 ).
Question 1. What Are the Recommendations for Identification of PMADs?
The first step in the pathway to effective diagnosis and treatment is case identification. Some women and pregnant or birthing people of other genders may recognize and report symptoms themselves, while others will not. Friends or family may identify symptoms and express concerns. Clinicians may also find themselves uncertain about the level of symptomatology that suggests a diagnosable disorder to be present. As such, while case identification can be made through sensitive clinical inquiry about mental health, the use of standardized clinical questionnaires to help identify cases has been investigated.81
The EPDS is a well-validated tool for the detection of perinatal depression that has been translated into 60 languages.82,83 While multiple cut-off scores have been suggested in the literature, evidence from a large systematic review suggests that a cut-off score of 11 or higher on the EPDS maximizes the ability to accurately identify depression, when compared to semi-structured diagnostic interviews in pregnant and postpartum populations (Level 3 ).84,85 While we recommend the EPDS preferentially, the PHQ-9, one of the most widely used depression self-report tools in primary care,86 has comparable test characteristics at a score of ≥10, so can be used if more practical to implement (Level 3 ).87 The PHQ-2 and Whooley Questionnaire have more moderate specificity (Level 3 ),88 but may be useful when the longer questionnaires are not feasible despite the trade-off of generating more false-positive cases.
Data are still emerging on the validity of standardized tools to identify cases of anxiety and related disorders in perinatal populations.89bibr90-07067437241303031bibr91-07067437241303031bibr92-07067437241303031–93 For generalized anxiety disorder (GAD), the GAD-7 scale (widely used in GAD screening non-perinatally), there is one study in pregnancy that suggests that a cut-off score of 7 may have better sensitivity and internal consistency than the score of 10 that is usually used for clinically meaningful symptomatology outside of the perinatal period.94 There is limited data about optimal scales and cut-off scores for the detection of other anxiety disorders, OCD, or PTSD in the perinatal period, although studies in this area are also emerging.95,96 Given the importance of case identification, if there is clinical suspicion, scales validated outside of the perinatal period—or those with some validation within the perinatal period—can be used with caution (Level 4 ).92
When a patient presents with symptoms of depression, anxiety or related disorders, it is also important to determine whether there is a current or past history of mania or hypomania because that may significantly alter the proposed treatment plan. The Mood Disorder Questionnaire (MDQ), a widely used instrument in non-perinatal populations, has evidence in the identification of BD in perinatal populations when a cut-off score of 7 or above is used and the supplementary questions are excluded (Level 3 ).97bibr98-07067437241303031bibr99-07067437241303031–100 The MDQ is excellent in ruling out the diagnosis of BD (>90% negative predictive value). However, positive cases need to be followed by a diagnostic assessment since only 50% of those scoring positive in the MDQ have a diagnosis of BD. The Highs, although less studied than the MDQ in non-perinatal populations, is the only instrument developed specifically for the detection of acute hypomania and mania symptoms in the postpartum period. The Highs is a 7-item questionnaire, where a score of 8 or higher can be used to detect BD, particularly when used at 1 and 6 weeks postpartum.101 The MDQ would be best utilized for assessment of past and acute symptoms whereas the Highs is best utilized to measure the severity of acute symptoms.
When using a standardized questionnaire for case identification, typically a cut-off score above a certain number represents a high likelihood of diagnosis. However, diagnoses cannot be made through questionnaires alone. When there is a concern for PMADs due to clinical suspicion and/or score on a questionnaire, the patient should receive further diagnostic assessment and treatment. It is also possible for an individual with a score below the threshold to still require care. As such, it is critical that when standardized clinical scales are used for case detection, that there is also a mechanism or pathway for proper diagnostic assessment and treatment.
One area of discussion has been whether standardized clinical questionnaires to identify PMADs should be used routinely with all perinatal patients. For perinatal depression, there is evidence that the use of a standardized questionnaire for case identification can improve identification rates, treatment rates and clinical outcomes (Level 2 ).102,103 Given the high prevalence of perinatal depression, the fact that perinatal people may not disclose their mental health concerns when not asked about them, the well-established risks of untreated illness and the validity of various standardized scales in identifying perinatal depression, the US Preventive Services Task Force has positioned themselves in favour of this strategy, as have organizations from other countries.102,104 In contrast, the Canadian Task Force on Preventive Health Care recommended against the routine use of standardized questionnaires with concern that the evidence in favour of the strategy was not strong enough to outweigh the potential economic costs and stress to patients of false-positive cases.105 Although there is high acceptability of the use of standardized questionnaires for case identification in perinatal populations,93,106 there was concern that use of a cut-off score on a standardized questionnaire on its own could lead to misdiagnosis or overdiagnosis, increased mental health stigma and unnecessary treatment.107
CANMAT believes that it is essential to ensure that cases of depression are identified as quickly and early as possible to promote timely and effective treatment, such that the evidence to date –which includes the good validity and acceptability of questionnaires in case identification and the Level 2 evidence for improvement of clinical outcomes—is sufficient to support the use of validated questionnaires to help in case identification. While standardized questionnaires may be helpful in identifying cases of PMADs other than depression as indicated previously, there is not yet evidence for how applying such tools routinely would impact on clinical outcomes. Further research on the feasibility and effectiveness of systematic or routine use of questionnaires to detect other PMADs is needed.
While there is no empirical evidence on the optimal timing for case identification and the feasibility of integrating questionnaires into routine care may be challenging in some settings, CANMAT recommends that clinicians providing antenatal, postnatal and/or pediatric care implement case identification of PMADs into their clinical practice throughout the perinatal period (e.g., once per trimester pregnancy, at postpartum obstetrical follow-up, in family practice and pediatric care up to 12 months postpartum) (Level 4 ).102,108,109
Considerations in Diverse Populations
While some well-validated tools have been translated into many languages, some populations may not recognize or experience mental health symptoms in a manner that is accurately captured by case identification tools standardized in a different population.110,111 Further, it has been found that local language versions of tools like the EPDS may been less likely to identify true cases of PMADs compared to the original English version.112 It may be beneficial to use tailored cut-off scores on questionnaires in specific populations, as evidenced in a recent scoping review showing that lower cut-off scores may be more appropriate in South and/or East Asian populations.113 There is limited evidence for optimal scales and cut-off scores in adoptive parents,82 and for those who are Indigenous or racialized.113bibr114-07067437241303031–115
Some racial and ethnic minority and migrant populations may be less likely to disclose mental health concerns due to the stigma surrounding mental illness and expectations of motherhood within their family or culture.110,116bibr117-07067437241303031bibr118-07067437241303031–119 There may also be a reluctance to disclose mental health challenges perinatally among those with disabilities due to fears of mistreatment or child apprehension.120 For people with intellectual and developmental disabilities, tools like the EPDS may not be as useful given this population’s challenges interpreting and recalling symptoms.121 There is also evidence that populations who are marginalized by racism and socioeconomic disadvantage are less likely to be assessed for PMADs.108 Further efforts to create culturally appropriate and accessible case identification tools and to examine and address inequities in assessment are needed.
Question 2. What Is Recommended for the Organization and Delivery of Healthcare Services?
Barriers to Care
Initial identification of PMADs may occur in antenatal or postpartum care, including in midwifery, public health nursing, primary care (e.g., family physicians, nurse practitioners), pediatrics or obstetrical settings. Ideally, healthcare services will be organized such that once PMADs are identified, there are clear pathways to assessment and treatment. Therefore, it is important to consider barriers to care and how to address them at the individual, healthcare provider and health system level, to improve treatment access and outcomes.
Multiple barriers to care for PMADs have been described.122 Key barriers include stigma, feelings of shame, guilt, embarrassment and lack of self-efficacy (i.e., a lack of confidence that there is anything that is under a person’s control that could help them to feel better). Sometimes patients and clinicians may lack knowledge about the symptoms of PMADs and that there are effective treatment options, or have specific perceptions about treatment that lead them not to want to seek care (e.g., concern that a medication may be the only option recommended when they prefer psychotherapy). Many patients have reported feeling too overwhelmed to seek care and some report fears of disclosing their mental health symptoms due to concerns about losing parental rights to their children. Further, interventions are effective only if they are accessible. While some can be implemented within one’s home, or with little to no cost, many interventions require funding, qualified personnel and other resources (e.g., transportation, childcare). Practical barriers to care include inadequate health insurance or financial issues, lack of childcare, time, transportation and/or internet connections for virtual services, as well as problems with care availability (i.e., there is variable access to specialized services in most jurisdictions with fewer treatment options especially in rural and remote regions).
Some populations, including those who are adolescents, disabled, gender-diverse, immigrants and refugees, Indigenous, racialized and sexual minorities, disproportionally experience barriers to care.48,123bibr124-07067437241303031bibr125-07067437241303031–126 Specifically, socioeconomic disadvantage and the structural impacts of colonization, racism, homo/bi/transphobia, ableism, and migration and country, community integration or settling experiences, impact access to care, provider-patient interactions, and in turn can negatively impact perinatal mental health outcomes.127 These structural factors and their legacies, including (intergenerational) trauma, must be considered and integrated into the training of care providers to ensure that all childbearing people receive safe, accessible and equitable perinatal mental health care.128
There is evidence that some racial and ethnic minority populations have lower rates of treatment initiation and engagement and higher dropout rates of psychological interventions despite very strong preferences for psychological over pharmacological interventions.116,129 Some barriers to care discussed above may partly explain race- and other identity-based differences in treatment preference modality, initiation and adherence. Culturally sensitive care and cultural adaptations of psychosocial and psychological interventions can reduce inequities in treatment adherence, as well as improve clinical outcomes among racial and ethnic minority groups.117,130 Clinicians are advised to provide all the information necessary for patients to make an informed decision, preferably in ways that are most accessible or applicable to that person (e.g., recommendations around web-based or social media resources, providing visual and printed materials, in various languages, etc., as opposed to use of medical or technical language). Clinicians are also advised to consider patients’ specific life circumstances, notably barriers to and/or availability of financial, social and other resources to support specific intervention initiation and adherence.
Models of Care
Strategies that have been suggested to decrease barriers and facilitate the implementation of perinatal mental health policy and practice are summarized into:119 (a) Design of care: Patients should have a choice in the care they receive; easy access; care should be delivered clearly, openly and honestly; flexible times of the appointments; practical supports such as assistance with childcare, transport and links with social work; access to interpreters if needed; and options for both in-person and virtual care; (b) Characteristics of healthcare providers: Healthcare providers should be open, non-judgmental, willing to listen, motivated, sensitive to verbal cues, interested and well-trained in perinatal mental health issues, dedicated to act as patients’ advocates, culturally sensitive, knowledgeable and confident, good communication skills and feel positively about the care they provide; (c) Organizational factors: There should be clear workflow procedures; collaborative working; personnel dedicated to perinatal mental health; and supervision; (d) Political factors: There should be free care or easy access through insurance policies (in Canada, many psychological services are not covered by universal healthcare insurance; in other settings there can be variability in access to many different types of services based on insurance status); services adequately funded with proper services and staff; clear and easy pathways of referral; and (e) Societal factors: There should be patient, family and public education to improve health literacy and support, and decrease stigma.
Unfortunately, most healthcare jurisdictions have not implemented systematic management pathways for PMADs. Instead, health care providers such as family physicians, midwives, nurses, obstetricians and pediatricians who identify possible cases of PMADs often rely on ad hoc referrals to local therapists or general mental health services, with referrals to more specialized perinatal mental health services only in regions where these are available. Similar to issues with mental healthcare in non-perinatal populations, it is thought that the lack of a systematic approach to perinatal mental health care delivery contributes to low treatment rates. Because of this, various models of care have been developed and evaluated in an effort to better organize and deliver care (Table 5).
Table 5.: Perinatal Mental Health Care Delivery Models.
| Line | Model of care | Level of evidence |
|---|---|---|
| First-line | Collaborative care | |
| Second-line | Stepped care | |
| Third-line | Staged care |
Collaborative care, also known as integrated care, is a mental healthcare model that uses interdisciplinary contributions by 2 or more health professionals to systematize case identification, assessment, triage and referral, initiation of treatment appropriate to the level of patient symptoms and risk, and ongoing/continuous follow-up symptom monitoring and management for entire patient populations.131 These models are typically based in primary or obstetrical care settings, with a mental-health trained behavioural health coordinator (e.g., RN) to assess, triage and monitor care and psychiatrists to advise primary care clinicians (i.e., family physicians and nurse practitioners) or obstetricians and oversee treatment programs. In this model, patients with mild and even moderate severity of symptoms can have their concerns initially addressed via front-line providers. For example, patients can be directly supported with social services, connections to peer support and other interventions in the primary care or obstetrical setting. Then, psychiatric support can be available when these initial interventions are insufficient or in more severe cases. Integrating mental healthcare into general healthcare settings (e.g., primary care and obstetrical or midwifery care) achieves greater access to treatment and continuity of care, which can provide benefits for patient mental health and obstetric outcomes.132 Collaborative care models are associated with high treatment satisfaction, and have been shown to be effective for the treatment of depressive and anxiety disorders in perinatal settings in some although not all trials (Level 2 ).133bibr134-07067437241303031–135 There is emerging evidence for perinatal psychiatric access programs, an approach that incorporates elements of collaborative care, but with less resource intensity, where health care providers can reach out to a specialized centre for resource and referrals, telephone provider consultation and direct patient consultation with expert perinatal psychiatrists.136
Stepped care models involve providing patients with treatments of increasing intensity depending on the severity of illness.137 Patients are typically advised to begin with the least intensive treatment shown to be effective for their symptom severity and then to ‘step up’ into more intensive interventions depending on their treatment response. These models are currently in use to deliver perinatal mental healthcare in certain national health systems (e.g., in the UK), but have not been extensively studied for PMADs (Level 3 ).138 In staged care models, patients are matched through a range of methods, including self-report questionnaires, to treatments according to their symptom severity.139 Staged care models have not been rigorously tested in perinatal populations. Some studies describe program development and there are non-randomized studies focusing on PPD and comorbid anxiety that show some promise for this type of intervention (Level 4 ).140bibr141-07067437241303031bibr142-07067437241303031–143
Stepped and staged care models differ from collaborative care models in that they do not necessarily encompass the population-based assessment and monitoring components. However, in health care systems where more comprehensive integrated care models have yet to be implemented, these approaches may be helpful. For example, in some Canadian jurisdictions, clinical care pathways have been developed to support antenatal, postnatal and pediatric care providers in assessing the severity of a patient’s PMAD symptoms and directing the patient to a specific step in the pathway based on this (e.g., peer support for mild symptoms, psychological therapy for symptoms of moderate-severity or medication for more severe symptoms).80,144
Studies of mental health care models for perinatal mental health have largely focused on interventions for depressive and anxiety disorders. Further research is needed beyond perinatal depression and anxiety, including in BD, PTSD, OCD and other perinatal mental disorders.145,146 Further research is also needed to determine the effectiveness of mental health care models in under-served populations, such as disabled, sexual minority, Black and Indigenous populations.147,148
Treatment Providers and Settings
Psychosocial interventions may be delivered in the community by peers, nurses and other community healthcare providers. Psychological interventions are usually delivered by trained mental health care professionals. However, task-sharing—the “rational distribution of tasks to less specialized personnel”149—can be leveraged to improve access to screening, self-care options and frontline psychological treatments.150,151 For example, brief psychological treatments to prevent and treat PMADs can be delivered by non (mental health) specialist providers.152bibr153-07067437241303031–154 Medications are usually prescribed by primary care clinicians (i.e., family physicians, nurse practitioners) and sometimes obstetricians. Psychiatrist input and follow-up is most important in complex or more severe cases, such as when there are comorbid conditions, inadequate response to initial treatments and in high-risk situations. Virtual delivery of care such as by telephone or video-conferencing can improve access to care by eliminating barriers to care, such as arranging childcare, transportation and geographical barriers, among others, with comparable effectiveness to in-person care for perinatal depression and anxiety (Level 2 ).155bibr156-07067437241303031bibr157-07067437241303031bibr158-07067437241303031bibr159-07067437241303031bibr160-07067437241303031bibr161-07067437241303031bibr162-07067437241303031bibr163-07067437241303031bibr164-07067437241303031–165
For individuals with more severe illness, intensive psychiatric day programs (also called “day hospitals”) have been developed, although these have only been evaluated in non-randomized studies (Level 4 ).166,167 For severe postpartum mental illness, mother–baby units (MBUs, not currently available in Canada) have been proposed as a means of providing specialized mental health treatment options for postpartum people with severe forms of mental illness without mother–infant separations. MBUs are meant to support the mother–infant relationship, while stabilizing maternal mental health. Evidence from non-randomized, uncontrolled studies shows positive effects on maternal mental health, mother–infant relationships and child development (Level 4 ).168,169 As there would be significant practical challenges to conducting randomized trials to provide additional evidence comparing MBUs to the standard of care, it may be important for jurisdictions currently without MBUs to consider that they may be particularly helpful in certain situations, especially for patients who may require prolonged hospitalizations (and thus long separations from their infants).
The range of different providers and settings in which perinatal mental health care can be accessed in Canada and other countries can result in a treatment landscape that is difficult to navigate.170 Patient navigation aims to facilitate timely access to mental healthcare by helping patients understand what services may be helpful for them, and connect them to the right service option (e.g., by identifying service options, coordinating with services to assist with referrals, and helping to remove access barriers).171 It has shown some promise in terms of clinical outcomes (e.g., reduced symptoms of depression and anxiety), patient satisfaction and service use (Level 4 ).171 Patient navigation’s effectiveness on its own without the actual connection to services that would occur in a collaborative, stepped or staged care model is not known.
Question 3. What Are the Recommendations for Lifestyle Interventions?
CANMAT considers lifestyle interventions as those that serve to intervene on health-related behaviours, including as this relates to diet, exercise and/or sleep. Prior CANMAT guidelines have also included light therapy in this category of interventions. A summary of lifestyle interventions for the prevention and treatment of PMADs is in Table 6.
Table 6.: Lifestyle Interventions for the Prevention and Treatment of PMADs.
| Line of treatment | Depressive symptoms | Anxiety symptoms | ||
|---|---|---|---|---|
| Intervention | Level of evidence | Intervention | Level of evidence | |
| Prevention | ||||
| First-line | Low to moderate-intensity exercise | — | ||
| Second-line | — | Low to moderate-intensity exercise | ||
| Third-line | aSleep protection especially in at-risk groups | — | ||
| Not recommended | Sleep education in non-clinical populations does not appear to prevent PPD | — | ||
| Infant behavioural sleep interventions in non-clinical populations may help with child sleep and maternal sleep quality but do not appear to prevent PPD | ||||
| Treatment | ||||
| First-line | Low to moderate-intensity exercise | — | ||
| Bright light therapy (postpartum) | ||||
| Second-line | — | Low to moderate-intensity exercise | ||
| Third-line | aSleep protection | aSleep protection | ||
| Sleep protection interventions are highly recommended for the prevention of relapse in perinatal bipolar disorder, including to help prevent postpartum psychosis and are also essential in the treatment of these conditions (Level 4 ) (see Question 9 for additional details) | ||||
Note. Exercise should be conducted under supervision to ensure clinical appropriateness. At-risk groups = those with a past history of, or current evidence of PMADs, and especially bipolar disorder and/or postpartum psychosis; CBT = cognitive–behavioural therapy; PMAD = perinatal mood, anxiety and related disorders; PPD = postpartum depression.
Sleep protection interventions may range from minimizing the time a mother has to be awake at night (e.g., bottle-feeding by another adult with pumped breastmilk or formula) to evidence-based interventions to treat insomnia when it is present. CBT for insomnia, CBT-I, is a highly effective treatment for insomnia where there is specifically effectiveness demonstrated for improving sleep quality, insomnia severity, depression and anxiety symptoms in pregnancy (Level 1 ), and open-label evidence for its use for insomnia in postpartum depression specifically (Level 4 ).
The evidence supports exercise interventions as helpful in preventing depressive symptoms from developing in pregnancy and postpartum in non-clinical populations (Level 2 ).172,173 Exercise of at least moderate intensity and greater than 150 min per week appears to most reduce the risk (Level 2 ).173 In terms of treatment, aerobic exercise has been associated with small to medium reductions in depression symptom severity, although available RCTs are of low-moderate quality (Level 2 ).174bibr175-07067437241303031bibr176-07067437241303031bibr177-07067437241303031–178 This applies to low-intensity activity (including walking), although moderate-intensity activity may lead to greater symptom reduction.175,176,179 Exercise interventions have also been shown to prevent anxiety symptoms and reduce their severity in pregnancy (Level 3 ).180 The extent to which these findings related to perinatal depressive and anxiety symptoms would generalize to those who meet diagnostic criteria for depressive, anxiety, anxiety-related or BDs is unknown. Regardless, exercise recommendations should be tailored to individual circumstances (e.g., finances, childcare support, physical limitations, other barriers), preferences, values and possible health risks (e.g., eating disorders, injuries) and adapted for the context of a person’s pregnancy and post-delivery recovery circumstances.
Sleep disruption is common in pregnancy (e.g., due to general discomfort, need for frequent urination and/or medical conditions such as sleep apnea) and when caring for an infant, and is linked to increased risk for and worsening symptoms of PMADs.181bibr182-07067437241303031bibr183-07067437241303031–184 Neither universal sleep education interventions nor behavioural sleep interventions for the infant (e.g., settling methods, such as “cry it out” or “fading,” or bedtime routines) appear to effectively prevent PPD in non-clinical populations (Level 2, negative).185,186 However, behavioural sleep interventions can help with child sleep and maternal sleep quality. Also, sleep disruption increases relapse risk for those with pre-existing mental illness,187 and especially BD in the postpartum period.188 So, initiatives to protect sleep perinatally are recommended in the prevention and treatment of PMAD symptoms (Level 4 ). Sleep protection interventions may range from minimizing the time a mother has to be awake at night (e.g., bottle-feeding by another adult with pumped breastmilk or formula for a night-time feed to allow a longer sleep time for the mother) to evidence-based interventions to treat insomnia when it is present so that the mother can sleep when the baby is sleeping. Cognitive–behavioural therapy (CBT) for insomnia, CBT-I, is a highly effective treatment for insomnia where there is specifically effectiveness demonstrated for improving sleep quality, insomnia severity, depression and anxiety symptoms in pregnancy (Level 1 ), and open-label evidence for its use for insomnia in PPD specifically (Level 4 ).164,189,190 Pharmacological management of insomnia in perinatal populations in addressed in Question 6.
A systematic review of 8 trials (n = 231) found bright light therapy to be superior to placebo with a small-to-moderate effect size in the treatment of depression in pregnancy and postpartum, although quality of the included studies was variable, statistical heterogeneity was high and in subgroup analysis, the effect was significant only in the postpartum (5 trials) but not in pregnancy (3 trials) (Level 2 ).191 Available studies focused on maternal diet and perinatal depressive symptoms were of low or very low quality, precluding recommendations therein; specific dietary supplements are discussed in Question 8.192
Question 4. What Are the Recommendations for the Use of Psychosocial Interventions?
An individual’s perception of insufficient support is a strong risk factor for PMADs.193bibr194-07067437241303031–195 Thus, CANMAT recommends optimizing informal supports such as those provided through community or embedded social networks of family members and friends (Level 4 ). Psychosocial interventions are designed specifically to enhance perceptions of support by providing informational (i.e., psychoeducational), emotional and instrumental support.196,197 Psychosocial interventions that have been evaluated perinatally include home-visiting support, peer support and psychoeducational programs (summarized in Table 7). Most research is focused on perinatal depression and anxiety. Given the paucity of empirical evidence in BD, PTSD and OCD, clinicians may need to consider evidence from non-perinatal populations in the management of these disorders.
Table 7.: Psychosocial Interventions for the Prevention and Treatment of PMADs.
| Line of treatment | Depressive symptoms | Anxiety symptoms | ||
|---|---|---|---|---|
| Intervention | Level of evidence | Intervention | Level of evidence | |
| Prevention | ||||
| First-line | Trained peer supporta | — | ||
| Second-line | Co-parenting (postpartum) | — | ||
| Third-line | — | Co-parenting (postpartum) | ||
| Not recommended | Home visits | Home visits | ||
| Psychoeducational programs | Psychoeducational programs | |||
| Treatment | ||||
| First-line | Trained peer support | — | ||
| Second-line | Listening visits (postpartum) | Trained peer support | ||
| Not recommended | Home visits | Home visits | ||
| Psychoeducational programs | Psychoeducational programs | |||
| In BD, trained peer support may be helpful as adjunctive to medication treatment in the prevention of symptom recurrence, including mania and depressive symptoms (Level 4 ). There is insufficient evidence for the role of structured psychoeducational programs in this population. | ||||
Note. BD = bipolar disorder; PMADs = perinatal mood, anxiety and related disorders.
If risk factors for and/or subsyndromal symptoms of PMADs are present.
There is strong evidence for peer support (i.e., support by trained peers) in reducing depressive symptoms in affected individuals during pregnancy and postpartum (Level 1 ).194,198 Peer support is effective whether delivered face-to-face, by telephone or online (virtually), in individual or group sessions. Efficacy has also been demonstrated in specific perinatal sub-populations, such as adolescents and parents of preterm infants.198,199 The optimal frequency for peer support appears to be at least once per week, with a length of less than 3 months being as effective as a length of 3–36 months. Peer support can also prevent PPD in at-risk populations such as those with risk factors for PPD or those with depressive symptoms that are subsyndromal (i.e., not severe enough to meet criteria for an MDE but have the potential to progress to an MDE if not addressed) (Level 2 ).200Peer support may further be effective for perinatal anxiety symptoms, though additional research is warranted (Level 3 ).201bibr202-07067437241303031–203 While there are no perinatal studies of trained peer support in BD, there is Level 2 evidence outside the perinatal period for adjunctive peer support (i.e., in addition to medication) in the prevention of symptom recurrence. Therefore, while empirical data in the perinatal population is awaited, adjunctive peer support from a peer with BD who had experienced pregnancy would be reasonable in this population (Level 4 ).7
Educating patients and families about pregnancy, parenting, PMAD risks, symptoms and treatments is important and part of best practices in healthcare. However, there is an overall lack of support for specific psychoeducational programs, including group-based prenatal care, that have been evaluated for the prevention and treatment of perinatal depression, anxiety and PTSD symptoms (Level 2 negative).134,174,204bibr205-07067437241303031bibr206-07067437241303031bibr207-07067437241303031bibr208-07067437241303031bibr209-07067437241303031bibr210-07067437241303031–211 That being said, one type of psychoeducational program, a co-parenting intervention where the focus is on educating parents and/or parental figures about how to share roles and effectively interact with each other while parenting a child together, does appear to be beneficial for the prevention of postpartum depressive (Level 2 ) and anxiety (Level 3 ) symptoms.212 Several structured psychoeducational programs have shown promise in relapse prevention in non-perinatal BD as adjunctive to medication treatment. However, to our knowledge no programs designed specifically for perinatal populations with BD have been rigorously evaluated.7
Home visiting by a nurse or other provider who provided unstructured support was generally not effective in preventing or treating perinatal depressive or anxiety symptoms (Level 2 negative for depression; Level 3 negative for anxiety ).198,213,214 However, one type of home visiting program, structured “listening visits,” which typically involves a nurse or other home visitor (∼4–6 sessions) trained in the use of empathic listening and problem-solving, has been shown to reduce depressive symptoms in the postpartum period (Level 2 ).215 This suggests that if home-visiting programs are implemented, these should be based on specific intervention models that have been evaluated and shown to be effective.
Question 5. What Are the Recommendations for the Use of Psychological Interventions?
Psychological treatment is recommended as a first-line option for treating moderate-severity presentations of depression, anxiety, OCD and PTSD or in mild-severity illness when lifestyle and psychosocial interventions alone are not fully effective, or are not accessible. Severe forms of these illnesses will not generally respond adequately to psychological treatments alone, and pharmacotherapy or other somatic interventions (e.g., neuromodulation) may be required to attain remission. While medications are foundational for the successful treatment of BD, adjunctive psychological interventions (i.e., in addition to medication) can be helpful in the treatment of depressive episodes, in relapse prevention and to improve quality of life.
Depression and Anxiety
Both CBT and interpersonal psychotherapy (IPT) are effective in the prevention of perinatal depression in those with a prior history of depression, subsyndromal depressive symptoms or those with psychosocial risk factors for perinatal depression such as low socioeconomic status, single parenthood and young maternal age (Level 1 ).200,205
Psychological treatments are also effective in treating MDEs in pregnancy and postpartum, with and without comorbid anxiety symptoms (Level 1 ).200,205,216bibr217-07067437241303031bibr218-07067437241303031bibr219-07067437241303031bibr220-07067437241303031bibr221-07067437241303031–222 The modalities for which there is the most evidence for efficacy are the manualized therapies, such as CBT,200,205,216,217,220bibr221-07067437241303031–222 IPT,200,205,217,218 mindfulness-based therapies (e.g., mindfulness-based stress reduction and mindfulness-based cognitive therapy),216,219,222 and behavioural activation (BA)223 (Level 1 ). CBT and mindfulness-based treatments are also effective when specifically targeting perinatal anxiety symptoms (Level 2 ).216,219,221,222 This includes emerging evidence for several forms of psychological treatment, primarily those containing elements of CBT, in the specific treatment of fear of childbirth symptoms (Level 2 ).218
Psychological treatments for depression and anxiety are typically delivered weekly, frequently ranging from 4–16 sessions, although there is also evidence for 1-day CBT workshops in the reduction of postpartum depressive and anxiety symptoms (Level 1 ).224bibr225-07067437241303031–226 They are effective when delivered individually or in group format, in-person or in virtual care (i.e., by telephone or video-conference) (Level 1 ).153,155,158,160,200,205,216bibr217-07067437241303031bibr218-07067437241303031bibr219-07067437241303031bibr220-07067437241303031bibr221-07067437241303031–222,227bibr228-07067437241303031bibr229-07067437241303031bibr230-07067437241303031bibr231-07067437241303031bibr232-07067437241303031bibr233-07067437241303031bibr234-07067437241303031bibr235-07067437241303031–236 There is specific evidence for virtual care in ethnically and racially diverse populations (Level 1 ).227,234,235 Non-specialist providers (e.g., nurses, midwives and peers)—individuals with no formal or prior experience in mental healthcare—can be trained to effectively deliver BA, CBT and IPT for perinatal depressive and anxiety symptoms(Level 1 ).153
Psychological interventions can also be delivered through self-help, where the patient works through a program of therapy on their own—usually using cognitive behaviour therapy techniques—via print workbooks or online web-based modules or mobile applications. These self-help interventions are considered guided when they occur along with online or telephone support from a trained coach to encourage completion, review progress and outcomes. Self-help psychological interventions (workbook and internet-based) reduce depressive symptoms, with benefits mostly observed for guided (Level 1 for PPD, and Level 2 for depression in pregnancy) rather than self-guided treatments (which is thought to be at least partially because the completion rates for self-guided treatments are very low).202,203,230,237bibr238-07067437241303031bibr239-07067437241303031bibr240-07067437241303031bibr241-07067437241303031bibr242-07067437241303031–243 Guided self-help interventions can also reduce anxiety (Level 2 ).155,158,160,216,227,231,232 with some evidence specifically for guided iCBT in pregnancy in fear of childbirth (Level 4 ).160 Guided self-help (internet- and non-internet based) interventions can also prevent prenatal depression and anxiety in at-risk populations such as those with risk factors for PMADs and/or subsyndromal levels of symptoms (Level 3 ).238bibr239-07067437241303031bibr240-07067437241303031bibr241-07067437241303031bibr242-07067437241303031–243
OCD
Less is known about psychological treatments for PMADs other than depression and anxiety. CBT with exposure and response prevention is recommended for OCD given its well established efficacy in non-perinatal OCD,244 despite the fact that the volume of evidence for CBT in perinatal OCD is not extensive (Level 3 ).245
PTSD
There has been interest in the secondary prevention of PTSD after a traumatic childbirth. Psychological debriefing interventions shortly after the event where the postpartum person is encouraged to process their emotional response to the traumatic event are not effective in the prevention of PTSD or the treatment of emergent symptoms (Level 1 negative).246 This is consistent with the evidence in non-perinatal populations.247 However, the lack of evidence for debriefing should not deter clinicians from providing clarification to patients around the events of a birth and answering questions that patients may have. In RCTs of mothers of preterm infants with elevated PTSD symptoms, there is some positive evidence for interventions with trauma-focused CBT components for the secondary prevention of PTSD (Level 2 ).246 RCTs of other interventions to prevent PTSD after traumatic childbirth that were included in systematic reviews are very heterogeneous and generally of low quality, precluding the ability to make a specific recommendation about them.246
There are multiple psychological treatments that are effective for PTSD non-perinatally (e.g., cognitive processing therapy, prolonged exposure therapy and written exposure therapy).248,249 However, there is limited evidence about which treatments would be most effective for a primary target of perinatal PTSD or posttraumatic stress symptoms, particularly for symptoms of PTSD related to events that occurred in childbirth.146,211,250 A recent pilot study using written exposure therapy for PTSD symptoms in pregnant individuals with PTSD and substance use disorders reported promising results (Level 4 ).251
Bipolar Disorder
Although psychological interventions for BD non-perinatally have not been shown to control symptoms on their own (i.e., in the absence of medication treatments) and have rarely been studied in the perinatal population, limited data on CBT, family-focused therapy and interpersonal and social rhythms therapy for BD in non-perinatal patients suggests some depressive symptom reduction, functional improvement and prevention of symptom recurrence when added as an adjunct to pharmacotherapy (Level 2 ). These could be considered as adjunctive interventions in pregnant and postpartum populations where feasible (Level 4 ).7
A summary of psychological interventions for the prevention and treatment of PMADs are in Tables 8 and 9, respectively.
Table 8.: Psychological Interventions for the Prevention of PMADs.
| Line of treatment | Intervention | Level of evidence | |
|---|---|---|---|
| Depression | First-line | CBTa | |
| IPTa | |||
| Second-line | Guided self-help psychological interventionsa | ||
| Anxiety | First-line | — | |
| Second-line | Guided self-help psychological interventionsa | ||
| OCD | n.d | ||
| PTSD | First-line | Interventions with trauma-focused CBT componentsb | |
| Not recommended | Psychological debriefing after traumatic childbirth | ||
| Bipolar disorder | First-line | — | |
| Second-line | — | ||
| Third-line | Adjunctive CBT (relapse prevention) | ||
| Adjunctive family-focused therapy (relapse prevention) | |||
| Adjunctive interpersonal and social rhythms therapy (relapse prevention) |
Note. CBT = cognitive–behaviour therapy; IPT = interpersonal therapy; OCD = obsessive–compulsive disorders; PMADs = perinatal mood, anxiety and related disorders; PTSD = post-traumatic stress disorder.
If risk factors for and/or subsyndromal symptoms of PMADs are present; bAfter preterm birth adjunctive = adjunctive to medication treatment; n/d = no data
Table 9.: Psychological Interventions for the Treatment of PMADs.
| PMAD | Line of treatment | Intervention | Level of evidence |
|---|---|---|---|
| Depression | First-line | CBTa | |
| IPTa | |||
| Mindfulness-based therapiesa | |||
| BAa | |||
| Guided internet-based self-help including iCBT and iBA in pregnancy | |||
| Second-line | Guided internet-based self-help interventions including iCBT and iBA postpartum | ||
| Anxiety | First-line | CBT for anxiety symptoms | |
| Mindfulness-based therapies for anxiety symptoms | |||
| Psychological therapies with CBT elements for symptoms of fear of childbirth | |||
| Second-line | Guided internet-based self-help for anxiety symptoms | ||
| Third-line | Guided iCBT for fear of childbirth | ||
| OCD | Second-line | CBT with exposure and response prevention elements | |
| PTSD | Third-line | Trauma-focused psychological treatments with evidence in non-perinatal populations | |
| Not recommended | Psychological debriefing after traumatic childbirth | ||
| Bipolar disorder | Third-line | Adjunctive CBT for MDE and quality of life | |
| Adjunctive family-focused therapy for MDE and quality of life | |||
| Adjunctive interpersonal and social rhythms therapy for MDE and quality of life |
Note. BA = behavioural activation; CBT = cognitive–behaviour therapy; IPT = interpersonal therapy; MDE = major depressive episode; OCD = obsessive–compulsive disorders; PMAD = perinatal mood, anxiety and related disorders; PTSD = post-traumatic stress disorder.
For MDE and for those with elevated depressive symptoms (with or without MDE diagnosis).
Question 6. What Are the Recommendations for the Use of Pharmacological Interventions?
Medications are most often used in depressive disorders, anxiety disorders, OCD and PTSD when non-pharmacological therapies are ineffective, and as first-line treatment when the initial symptoms are moderate-to-severe or severe. They may also be used as initial treatments in patients with milder symptoms who are unable to access psychological interventions, or who prefer medication over psychotherapy. Medication is the mainstay of treatment in BD and postpartum psychosis, regardless of symptom severity. While most medications used for PMADs are not perinatal-specific, brexanolone which is delivered via intravenous infusion, and its oral counterpart zuranolone, are novel medications that have recently been approved specifically for the treatment of PPD in the U.S. Brexanolone and zuranolone are neurosteroid derivatives of the progesterone metabolite allopregnanolone, and modulate Gamma-Aminobutyric Acid A (GABA-A) receptors, which are known to be involved in the pathophysiology of depression.252,253
Decisions about medication use during pregnancy and lactation will always involve balancing the benefits to the patient and foetus or developing infant (especially given the negative impacts of untreated or undertreated illness) against the potential risks of medication exposure in pregnancy or lactation (safety and tolerability). Some individuals will be taking medication prior to the pregnancy, so decisions centre around risk for relapse. Others will be faced with decisions about whether to initiate a medication when new symptoms arise. It is critical that clinicians have up-to-date knowledge about benefits and risks so that they can provide patients and significant others with clear, balanced and unbiased information. Decision-making can be optimized by: (a) advising patients about all of their treatment options (including not being treated), (b) helping them clarify their values and preferences about these options and (c) ensuring ample time for decision-making.254,255 Best practice supports describing estimates of efficacy and safety in absolute terms when such estimates are possible to provide (e.g., 1 in 1000), and acknowledging when there is uncertainty.256
Medication Safety in Pregnancy and Lactation
With some exceptions, most commonly used psychotropic medications are considered fairly low risk in terms of their impact on pregnancy outcomes, including for congenital malformations (first trimester exposure), and for fetal growth, pregnancy complications, neonatal health and latent child developmental effects (e.g., motor, language and socioemotional development). When evaluating medication safety in pregnancy, it is sometimes difficult to separate the potential impacts of a medication from those of other medications or exposures (including for example pre-pregnancy exposures such as fertility treatments), the underlying illness and other factors associated with the indication for the medication (e.g., genetic risk, smoking, high body mass index and others). Because RCTs of psychotropic medications have not been conducted in pregnant people, the best available evidence is usually from large observational (e.g., case-control, cohort) studies. Unlike RCTs, observational designs can be subject to bias (e.g., confounded by the illness itself or other associated factors). Multiple methods have been used to attempt to control for confounding variables (e.g., comparing siblings born to the same parents where only one sibling was exposed to medication in-utero, comparisons to unmedicated pregnant people with the same underlying illness, etc.), so this guideline uses data from these higher-quality observational studies to inform recommendations. As observational studies cannot fully control for confounders, some degree of uncertainty about risk of medications prescribed in pregnancy will always remain.
Similarly, most psychotropic medications appear to be low-risk in lactation. In lactation, available safety evidence derives mostly from case-series and small observational studies, and there is often less information on safety available than in pregnancy. However, the majority of psychotropic medications have a relative infant dose (RID, the dose received via breastmilk relative to the mother’s dose) of less than 10%, which is generally considered to be of minimal risk.257 A drug with an RID > 10% may still be appropriate to prescribe if it does not appear to confer risks. Caution should be exercised with preterm infants and those with renal, hepatic, neurological or other significant health conditions, particularly with agents with a long half-life or active metabolites.258 There is no convincing evidence that attempts to alter feed time or dump breastmilk to reduce infant exposure will reduce risks to the infant.
Therapeutic Drug Monitoring
The dosing and monitoring protocols for most psychotropic medications are generally similar to those of the non-perinatal period. There are pregnancy-related physiological changes that can impact the metabolism of some psychotropic medications, reducing their blood levels.259 In most cases, clinicians should monitor patients for possible symptom worsening and adjust medication dosage as clinically appropriate should worsening of symptoms occur. However, lamotrigine and lithium elimination both increase in pregnancy with reduction to pre-pregnancy levels fairly quickly postpartum. As such, perinatal-specific monitoring protocols are advised (see Antiepileptics and Lithium below).
Antidepressants
Antidepressants are first-line medications in depressive disorders, anxiety disorders, OCD and PTSD and are sometimes used in bipolar depression as an add-on medication. Most studies evaluating safety in pregnancy have focused on selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs) and other antidepressants as a whole or as a class (e.g., SSRIs), with fewer focusing on specific medications within classes. This means that, with some exceptions, it is difficult to distinguish between safety within medication classes. Data (summarized in Box 1) show that links between most antidepressants and most adverse pregnancy outcomes are reduced signficantly when confounding by indication is well-managed.260,261 Some experts advise tapering antidepressants near delivery to reduce the risk of poor neonatal adaptation syndrome (PNAS). While this approach is supported by evidence from a recent non-randomized case series, earlier evidence suggested that there was no difference in risk for neonatal respiratory distress between pregnancies where the antidepressant was and was not discontinued 14 days prior to delivery after accounting for maternal illness severity.262,263 While CANMAT acknowledges this is a developing area, the benefits of tapering to reduce the risk of PNAS likely do not outweigh the risk of worsening maternal illness severity; hence reducing or discontinuing antidepressants near delivery is not recommended (Level 4 ).
Box 1.
Summary of evidence on the safety of antidepressant use in pregnancy
Level 3 evidenceunless otherwise specified
Pregnancy complications: The risk of spontaneous abortion (SA) is not thought to be increased with SSRIs after accounting for maternal depression and other confounders, though there is less certainty about risk with venlafaxine, duloxetine and mirtazapine.261,264,265 Risks for gestational diabetes mellitus and gestational hypertension do not appear to be meaningfully increased in those taking SSRIs, SNRIs, TCAs, or monoamine oxidase inhibitors (MAOIs) during pregnancy.261,265,266 For postpartum haemorrhage (a concern that has been investigated due to an increased risk for bleeding with serotonergic antidepressants in non-pregnant populations), findings are mixed but the studies that have adjusted for psychiatric disorder suggest that the increase in blood loss with SSRIs and SNRIs is of small magnitude (i.e., ∼300 mL of excess blood loss) with no evidence of severe complications or mortality to the pregnant person or infant.265,267
Congenital malformations: SSRIs, SNRIs and TCAs as classes do not appear to increase risk for major congenital malformations.261,268 There may be an increased risk for cardiovascular malformations specifically during first-trimester exposure with SSRIs, SNRIs and bupropion, however this risk is thought to be very small in absolute terms (if not nil after accounting for confounders); some studies have suggested slightly greater cause for concern with paroxetine than for others in these classes.269bibr270-07067437241303031bibr271-07067437241303031–272 TCAs do not appear to be associated with meaningfully increased risk.269 For the MAOI tranylcypromine there are only case-series data, but severe facial dysmorphisms, cardiac malformations and fetal death have been reported (Level 4 ).273
Fetal growth: While the risk of growth restriction and shortened gestational age may be increased with antidepressants generally, the magnitude of these reductions is not likely to be clinically significant (i.e., <100 grams and <1 week gestational age in most cases).265,274bibr275-07067437241303031–276
Neonatal complications: Persistent pulmonary hypertension of the newborn is a rare but serious condition affecting 2/1000 infants. In foetuses exposed to SSRIs and SNRIs in the third trimester, the absolute risk may be increased to ∼3/1000 overall. For SSRIs, the elevation in risk may be lower for sertraline and escitalopram than for fluoxetine, but this is not definitive.277 Up to 30% of newborns exposed to SSRIs and SNRIs in pregnancy will display transient symptoms of PNAS, a constellation of symptoms that include jitteriness, respiratory distress, irritability, sleep disturbance, tachycardia and feeding problems.278 PNAS is self-limited in most cases, is rarely associated with severe complications, and usually resolves within 2–3 days with supportive care. This risk is increased with all SSRIs/SNRIs, but may be highest with paroxetine, venlafaxine and fluoxetine.261
Longer-term child health: There are no consistent associations between fetal exposure to SSRIs and later physical health (including asthma, cancer, epilepsy), neurodevelopmental (cognitive or motor problems, autism or attention-deficit hyperactivity disorder) or psychiatric problems such as anxiety or depression in adolescence.279 It is hypothesized that most observed associations with neurodevelopmental or psychiatric disorders in children may reflect that SSRIs are merely a “proxy exposure” for the shared genetics that place both parent and child at increased risk of these conditions (as well as the exposure to the environmental risk of parental mental illnesses).
In general, antidepressants do not have significant adverse effects on infants exposed via lactation, nor do they substantially impact breastmilk production.280 Fluoxetine has a longer half-life than many SSRIs and so other medications with shorter half-lives are preferred. There have also been case reports of infant seizure associated with bupropion, although these may have been due to other factors and not the medication per se. The only medication not recommended in lactation is the TCA doxepin, due to concern about infant sedation.281
Antiepileptics
Antiepileptic medications are used in the treatment of BD and certain anxiety disorders. Carbamazepine, lamotrigine and valproic acid are the only antiepileptics recommended as monotherapies in the CANMAT BD guidelines, so are reviewed here individually to the extent that it is possible to separate out specific medications from class effects. Valproic acid is associated with a substantially increased risk for infant congenital malformations (mainly neural tube and cardiac defects) after exposure in the first trimester of pregnancy and increased risk for child neurodevelopmental delay after second and third trimester exposure (Box 2).282,283 As such, valproic acid is not recommended in pregnancy. In fact, due to its known teratogenicity, it is not recommended for reproductive-aged persons of female sex generally due to the high prevalence of unplanned pregnancy. If an individual arrives in pregnancy or to a pre-pregnancy planning consultation while taking valproic acid, serious consideration should be given to whether it is possible to change the medication without substantial risk to maternal psychiatric stability. In cases where other medications have not been effective or tolerated (or if the pregnancy is nearing term and the risk for destabilization would be high with minimal reduction in risk from discontinuing at that point), clinicians and patients should fully discuss the risks and the potential benefits of continuing to maintain this medication. Although a systematic review of risks to the offspring associated with medication use in fathers and non-birthing parents was outside the scope of our guideline, it is important to mention recent literature investigating potential congenital malformation and neurodevelopmental risks associated with the use of valproate in fathers. While the European Medicines Agency’s Pharmacovigilance Risk Assessment Committee reported a study showing that paternal use of valproate exposure was associated with increased risk of neurodevelopmental disorders but not congenital malformations in the offspring,284 2 independent population-based studies that adjusted for important confounders did not find any association between valproate use in fathers and congenital malformations or neurodevelopmental outcomes in the offspring.285,286
Carbamazepine is also associated with an increased risk for congenital malformations (likely in a dose-dependent manner with greater risk as the dose increases). However, as the risk is less than with valproic acid and there is minimal concern about an association with adverse infant neurodevelopment or obstetric outcomes, it can be used with more confidence after the first trimester.282,283 Exposure to carbamazepine in pregnancy may increase the risk of infant vitamin K deficiency, so vitamin K should be administered in late pregnancy to reduce the risk for newborn bleeding.287 Carbamazepine concentrations are not significantly affected by pregnancy so dose adjustments are not typically required.281
Lamotrigine is the antiepileptic with the most favourable reproductive risk profile in pregnancy and lactation.288 However, its elimination increases as early as 5 weeks gestation and by >200% during pregnancy in ∼80% of patients so it can be difficult to attain therapeutic concentrations in a timely manner in pregnancy.289,290 For those already taking lamotrigine, measurement of a pre-pregnancy lamotrigine concentration can help determine the baseline therapeutic concentration. Having a target therapeutic concentration can be helpful in guiding treatment if symptoms worsen, and could be used for proactive dose adjustments in an effort to reduce risk for relapse.291 Lamotrigine levels return to pre-pregnancy levels within the first 2 weeks postpartum, so the dose can be tapered to the original dose associated with symptom remission prior to pregnancy.289 This is particularly important if the patient was taking a high dose (i.e., greater than 200 mg) prior to pregnancy, to prevent adverse side effects or toxicity.
Folic acid supplementation of at least 0.4 mg/day is recommended when antiepileptics are prescribed in pregnancy. The evidence for whether a higher dose of folic acid supplementation (i.e., 4 to 5 mg) definitely reduces the risk of neural tube and cardiac defects remains inconclusive.292 There has been some concern raised about whether folic acid could reduce the effectiveness of lamotrigine in non-perinatal populations with BD, so patients using lamotrigine with folic acid supplementation should be carefully monitored for potential clinical symptom re-emergence.293
Box 2.
Summary of evidence on the safety of antiepileptic use in pregnancy (for antiepileptics indicated in the treatment of psychiatric disorders)
All are Level 3 evidenceunless otherwise specified.
Pregnancy complications: Antiepileptics as a group are associated with a slightly increased risk for postpartum haemorrhage and induction of labour.294 Because these complications are also associated with epilepsy (the illness exposure primarily studied for antiepileptic exposure) independent of pharmacotherapy, it is difficult to estimate the risk for the non-epileptic pregnant patient with a psychiatric disorder. Analysis of data on antiepileptic exposure across studies suggests that these medications do not appear to increase the risk of gestational hypertension, bleeding in pregnancy, or caesarean section (Level 4 ).295
Congenital malformations: Risk for major congenital malformation is elevated with valproic acid exposure. This includes an increased risk of cardiac malformations (e.g., atrial septal defects), hypospadias, neural tube defects (e.g., spina bifida, craniosynostosis) and oral clefts.282,296,297 Risk of congenital malformations with valproic acid may be up to 3-fold higher or more than the general population. Carbamazepine is also associated with a slightly elevated risk for congenital malformations including neural tube defects, cardiac malformations, urinary tract abnormalities and oral cleft/palate defects, but to a much lesser degree (e.g., pooled OR 1.37).282,296,298 Risk for congenital malformations with lamotrigine is not significantly greater than that of the general population.282
Fetal growth: Antiepileptic medications in general have been associated with small absolute increased risks for small-for-gestational age and low birthweight, and with a small reduction in birthweight (∼119 g), but overall not birth height or head circumference.299 These risks may be increased with polytherapy of antiepileptic agents.299 Reduced fetal head growth may be associated with carbamazepine exposure.300
Neonatal complications: Antiepileptic exposure in general may increase the risk of neonatal intensive care unit admission and may be associated with increased risk for preterm birth but data are mostly in populations with epilepsy (which is associated with its own reproductive risks that may not be applicable in psychiatric populations).294 When lamotrigine exposure has been studied in isolation, it does not appear to be associated with neonatal complications (Level 4 ).301
Longer-term child health: Risk for global language deficits and for intellectual and developmental disorders are increased up to 4–5 fold with valproic acid exposure and there appears to be a dose–response relationship. There may be an elevated risk for neurodevelopmental disorders with exposure to pregabalin (discussed herein as it is a monotherapy recommended in the treatment of GAD).283,302,303 Neither carbamazepine nor lamotrigine have been consistently associated with neurodevelopmental risks including intelligence, or mental and behavioural disorders; data are limited for other antiepileptics.283,302,304,305
There is ample data suggesting that there are very few adverse effects from lamotrigine exposure in lactation.306 Carbamazepine levels are detectable in breastfed infants although only minor adverse effects have been reported.307 While few adverse effects of valproic acid in lactation are reported, it is not recommended to start valproic acid in lactation due to the risk for a subsequent pregnancy unless there is a reasonable clinical rationale, patient preference and use of a method of contraception that ensures that an unplanned pregnancy would be highly unlikely (e.g., birth control implant, intrauterine device) (Level 4 ).308 If a patient was taking valproic acid throughout the pregnancy, then it can be continued during lactation if the patient chooses to breastfeed.
Antipsychotics
Second-generation antipsychotics (e.g., olanzapine, quetiapine, risperidone) are a mainstay of medication treatment in BD (acute mania, depression and postpartum psychosis) and are also used in the treatment of MDD, some anxiety disorders, OCD and PTSD, mostly as adjunctive agents. Older, first-generation antipsychotics (e.g., haloperidol or loxapine) may be used in mania, severe acute agitation or postpartum psychosis (see Question 9). In general, antipsychotic dosing is similar inside and outside of the perinatal period, although the increased metabolism of pregnancy may lead to a need for dosage adjustments if symptoms re-emerge.309
Antipsychotics have typically been studied more as a class than individually with respect to their perinatal safety, with a few exceptions. Most data pertain to first-generation or older second-generation antipsychotic medications (e.g., olanzapine, quetiapine, risperidone) (Box 3). There is very limited information about long-acting injectable antipsychotic medications in the perinatal period, with the available evidence primarily from case reports and case series.310,311 Antipsychotics do not appear to be associated with an increased risk for congenital malformations, although risperidone has been linked in some, but not all analyses, with a small increased risk for cardiac malformations.312,313 Second-generation antipsychotics (particularly olanzapine and to a lesser extent quetiapine) may be associated with some increased risk for metabolic complications such as gestational diabetes and large-for-gestational age infants.314,315 Antipsychotic exposure in pregnancy may also be associated with transient delays in neurodevelopment in infancy, but does not at this time appear to be associated with longer-term child health or developmental problems.316
Box 3.
Summary of evidence for the safety of antipsychotic use in pregnancy
All are Level 3evidence unless otherwise specified.
Pregnancy complications: Second-generation antipsychotics with a high-risk for metabolic effects are associated with an increased risk of gestational diabetes that appears to be most significant for olanzapine (RR ∼ 1.6) and for quetiapine (RR ∼ 1.3) than for other second-generation antipsychotics.315 Risk for SA does not appear to be elevated for those taking first- or second- generation antipsychotics.317
Congenital malformations: Neither first- nor second-generation antipsychotics appear to increase the risk for major congenital malformations, although some data suggest the possibility of a small increased risk for cardiac malformations with risperidone (RR ∼ 1.3).312,318 Accumulating data for ziprasidone and lurasidone suggest a favourable safety profile without increased risk but more data are needed for lurasidone.309,312,319 Data for newer antipsychotics, such as cariprazine and asenapine, are too sparse to draw clinical conclusions.
Fetal growth: Second- but not first-generation antipsychotics are linked to large for gestational age (RR ∼ 1.6),314 and findings are mixed for small for gestational age and growth restriction with both types of agents.314,320
Neonatal complications: There does not appear to be an elevated risk for preterm birth320 or stillbirth.317 Infants exposed to first- or second-generation antipsychotics may experience symptoms in the time shortly after delivery such as respiratory distress, fussiness and gastrointestinal issues which may require neonatal intensive care admission, although the magnitude of elevated risk for these outcomes (or whether the risk for this is confounded by other factors) is unclear.309
Longer-term child health: Antipsychotic exposure in-utero has been relatively consistently associated with transient delays in motor development in the first few months of life that appear to resolve in the first 1–2 years (RR ∼ 1.36).316 Antipsychotics have not been consistently associated with long-term adverse effects on neurodevelopmental outcomes (including behavioural disorders, autism spectrum disorder, attention deficit hyperactivity disorder and intelligence quotient) or psychomotor development.320
All lactation safety evidence on antipsychotics stems from uncontrolled case series or observational studies (Level 4 ). Olanzapine and quetiapine are the agents with the most reported cases in lactation and appear to have a favourable risk profile.321,322 Although fewer cases have been reported, risperidone and aripiprazole also appear to have low transfer into breast milk, although risperidone has been linked to more reports of adverse neonatal reactions than the aforementioned medications.323,324 There have been several case reports of reduced breastmilk production with aripiprazole, but data are limited. Minimal data on lactation safety are available for ziprasidone, and even less for other second-generation antipsychotic medications. Adverse effects in infants are more commonly reported for first-generation than for second-generation antipsychotics in lactation, thus more caution is recommended when using first-generation antipsychotics in lactation.325,326
Lithium
Lithium is indicated in the treatment of BD and as an adjunctive agent in the treatment of MDD. For a significant number of individuals with a history of severe bipolar I episodes, lithium may be particularly effective and so reducing or stopping it in pregnancy must be weighed against risk of severe relapse. That being said, lithium use in the first trimester is associated with a small increased risk for malformations (including the rare Ebstein’s anomaly).327 It has also been linked to a small increased risk for some neonatal complications, including preterm birth, infant hypoglycemia, abnormal thyroid and kidney function lab values, and decreased muscle tone; data suggesting lack of longer-term neurodevelopmental impact in children are reasonably reassuring (Box 4).
Box 4.
Summary of evidence for the safety of lithium use in pregnancy
All are Level 3evidence unless otherwise specified.
Pregnancy complications: Spontaneous abortion, pre-eclampsia and postpartum haemorrhage do not appear to be associated with lithium exposure.328bibr329-07067437241303031–330
Congenital malformations: During the first trimester, there is an increased risk (Relative Risk, RR ∼ 1.7) for cardiovascular malformations including Ebstein’s anomaly that appears to be dose-dependent, but the absolute risk is low (i.e., an RR of 1.7 would increase the risk from 1–5 to 2–8 per 200,000 births).331 No associations with congenital malformations other than cardiac malformations have been clearly identified.329 A recent meta-analysis reported an increased risk for congenital anomalies overall, but this did not separate out the risk of cardiac malformations from the overall risk for malformations.328
Fetal growth: Low birth weight has not been associated with lithium exposure but some data suggest an increased risk for large for gestational age at birth (RR ∼ 2.6).328bibr329-07067437241303031–330
Neonatal complications: Data from well-controlled studies do not support a risk for preterm birth.328,330 There may be a slightly elevated risk of hypoglycemia, thyroid abnormalities, nephrogenic diabetes insipidus and hypotonia (Level 4 )332 and a higher rate of 28-day neonatal readmission to special care baby units (OR ∼ 1.6).330
Longer-term child health: Lithium has not been associated with adverse neurodevelopmental outcomes.316
Pregnancy-related physiological changes such as increased cardiac output and glomerular filtration rate increase the elimination rate of lithium during pregnancy which can lead to lithium concentrations that are lower than the therapeutic range and/or the patient’s pre-pregnancy therapeutic level, sometimes requiring medication adjustment.333,334 There are no standard guidelines for monitoring lithium levels in pregnancy. At first assessment or in early pregnancy, obtaining a baseline level is advised. Clinicians can then monitor lithium concentration once each trimester or if symptoms or side effects emerge and adjust as needed.24,333,335 Dose adjustments should be conservative (balancing the need for mood stabilization) to minimize any risk for adverse effects. Twice daily dosing may be necessary to prevent peak concentration side effects for the patient. Lithium should be held for any patient that develops preeclampsia due to impaired kidney function as this condition significantly increases the risk for maternal/fetal toxicity. If holding lithium for an extended period is required, an alternate mood stabilizer may need to be considered for relapse prevention. Patients with gestational hypertension, which also impairs kidney function, should be closely monitored to prevent toxicity. Because of anticipated maternal volume contraction post-delivery, the pre-pregnancy dose (or 150 to 300 mg less than the dose in pregnancy if the medication was started in pregnancy) can then be resumed immediately post-birth. Monitor the lithium level 5–7 days later, and adjust the dose as needed for symptom control and to reach therapeutic concentration. There is some controversy on whether to hold the lithium dose for 24–48 h at the onset of labour to both prevent maternal toxicity and to reduce lithium exposure levels through the umbilical cord at delivery (Box 5).
Box 5.
Lithium and holding the dose at the onset of labour: reconciling conflicting evidence
Some experts advocate for holding the dose to reduce the risks for maternal toxicity (due to maternal volume contraction post-delivery) and adverse neonatal effects (due to high levels of lithium exposure via the umbilical cord).336 Others have expressed concerns that holding the lithium dose could increase the risk for postpartum maternal relapse, with minimal benefit of reduced exposure to the infant.337 While maternal volume contraction does occur post-delivery, it is unlikely that there would be immediate maternal toxicity if a dose is not held. That being said, based on the half-life of lithium in the cerebrospinal fluid, holding the dose for 24–48 h should also not significantly increase the risk of episode recurrence.338,339 Existing studies with infants exposed to very high concentrations of lithium (mean 1.68 mmol/L, range up to 2.6 mmol/L) showed an increased risk for adverse effects in the neonate (Level 4 ).336 In contrast, a cohort study of 29 neonates exposed to lithium during birth did not show a significant association with adverse effects with a mean lithium level of 0.61 (SD = 0.31) mmol/L (Level 4 ).337 Therefore, in the absence of more definitive evidence, clinicians can consider holding the lithium dose at the onset of labour or induction (or for 24 h prior to a scheduled caesarean birth) when lithium concentration is 0.7 mmol/L or higher as this may reduce the amount of lithium in the umbilical cord without substantively increasing the risk for maternal relapse. An informed decision-making process should take place with the patient, considering both their clinical severity and risk for relapse, as well as the health of the pregnancy to date. Holding the lithium dose may not be appropriate in patients who are at very high risk for postpartum mania or psychosis, or other high-risk clinical situations such as suicide or infanticide (see Question 9).
There is also some debate around lithium use in lactation. Lithium is excreted by the kidney, so there is potential for immature infant renal function to lead to high infant lithium levels with resultant adverse effects. While some early data were concerning, it was unclear whether infant adverse effects were due to exposure in lactation, or from gestational exposure.340 More recent data suggest that adverse effects are limited to mild cases of hypotonia that resolve spontaneously and to transient or reversible lab abnormalities (e.g., blood urea nitrogen, creatinine, TSH).340 For those maintained on lithium therapy, the decision about whether to breastfeed (vs. formula feed or use donor milk) must be personalized to the risk of relapse when considering discontinuation or alternative treatment, the infant’s health and the patient’s preference.341 CANMAT does not recommend breastmilk exposure in premature infants, infants with kidney malformations, or with illnesses that may cause dehydration (Level 4 ). Postnatal lithium exposure via lactation may result in irritability, reduced muscle tone, or feeding difficulties. Therefore, CANMAT also recommends that infants be monitored for any change in behaviour and muscle tone and in the case of these or any other clinical concerns, lithium levels, cardiac (e.g., via electrocardiogram), thyroid and kidney function should be assessed (Level 4 ).
Sedative-Hypnotic Medications
Benzodiazepines are sometimes used to manage acute anxiety and sedative-hypnotic medications (i.e., benzodiazepines and the hypnotic benzodiazepine receptor agonists such as zopiclone or zolpidem) are also considered a treatment option in more severe cases of insomnia. These are intended for short-term use only due to concern about developing tolerance and dependency. Safety in pregnancy and lactation must be considered with respect to their use. While not consistently linked with fetal malformations, regular use of benzodiazepines up to delivery are associated with risk of respiratory problems, somnolence and NICU admission in infants (Level 3 ).342,343 There are generally fewer concerns with the use of occasional (“as needed”) benzodiazepines. Hypnotic benzodiazepine receptor agonist exposure in pregnancy has not been associated with risk for congenital malformations, but may be linked to a slightly increased risk for preterm birth, low birthweight and small-for-gestational age infants (Level 3 ).344 Whether there would be lower risk for these outcomes with periodic versus prolonged use is not known.
Treatment Recommendations
To decide Line of Treatment recommendations, the CANMAT guideline committee started with relevant non-perinatal treatment guidelines, where medications are ordered by efficacy and tolerability. We used the most recently published CANMAT guidelines for MDD, OCD and BD, the World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for anxiety disorders and the U.S. Department of Veterans Affairs and U.S. Department of Defense for PTSD since the most recent Canadian guidelines for anxiety disorders and PTSD were published in 2014.345bibr346-07067437241303031–347 We ranked the medication options for monotherapies from these guidelines only, as there is minimal safety evidence for specific combinations of medications in the perinatal period. Based on perinatal-specific considerations, we adjusted medications up or down in Lines of Treatment for the current guideline by consensus recommendation (Box 6). In pregnancy, we considered data on: (a) congenital malformation risk (usually a concern for first trimester exposures only), pregnancy and obstetric complications, fetal growth, neonatal complications and latent developmental effects and (b) the safety of the medications in lactation since it is preferable not to have to change a medication in the early postpartum period given that this is the highest-risk time for a relapse. In the postpartum recommendations, we considered safety in lactation, and the possibility of a future pregnancy, as many parents will be planning for another child soon and unplanned pregnancies are relatively common. We also considered other aspects of maternal tolerability that are important in the postpartum. For example, if a medication causes substantial maternal sedation, it may not be ideal due to night-time infant awakenings and daytime caregiving needs unless there is additional caregiver support.
Line of Treatment recommendations are for patients initiating a new medication or those who require a medication switch because the current medication is ineffective, poorly tolerated or contra-indicated perinatally. For patients on maintenance pharmacotherapy, risk for relapse if the medication were to be discontinued or switched (and its potential implications) must be weighed against medication safety concerns.348 Generally, the more severe the illness (i.e., number of episodes, severity of symptoms), and the longer the time to treatment response after initiation of medication in the past, the greater the risk of discontinuing or switching medication. Importantly (see Box 6), when symptoms are well-controlled, it is not usually advisable for a medication to be changed to another one based on its placement in the Line of Treatment tables, unless it is listed as “not recommended” due to serious issues about perinatal safety. For patients taking medications where there is insufficient evidence about safety in pregnancy or lactation to be included as a first-, second- or third-line treatment, the clinician will need to consider treatment history (i.e., were other medications previously ineffective for that patient?), and severity of illness (i.e., what would be the potential risk for relapse with medication discontinuation or switch and the potential consequences of such a relapse?) to help the patient make an informed choice aligned with their values and preferences. For patients who are not taking medication at the time of presentation, but who have responded to a specific medication in the past, the best agent may be the one they have responded to previously, even if it is in a “lower” line of treatment, unless it is specifically contraindicated.
Box 6.
Explanation of PMAD pharmacotherapy recommendation tables
- Options are classified into first-line, second-line and third-line treatment recommendations for each PMAD.
- Lines of treatment were created by initially considering the placement of individual agents in non-perinatal treatment guidelines, and then by adding, removing, or moving agents up or down in lines of treatment based on evidence for their safety and efficacy in pregnancy and in lactation. For example, if 2 medications were similarly effective and tolerated in non-perinatal populations, but safety data in pregnancy and lactation were more reassuring for one medication, that agent would be placed in a higher line of treatment.
- Within lines of treatment, agents are listed alphabetically. A justification for placement column contains the rationale for placing the medications within that line of treatment. Clinicians and patients can use this information to help select which agent(s) to try first within each line of treatment. Such decisions may be based on prior history or response/tolerability with previous treatments and/or how patients value the potential benefits and risks associated with specific agents.
- Although CANMAT recommends that agents listed in a higher line of treatment be tried first, there may be specific reasons for choosing an agent in a lower treatment line such as patient preference, prior treatment response or non-response, or other clinical features. Specifically:
Considerations of Monotherapy versus Combination Medication Treatment
As the combined use of 2 or more agents during pregnancy is associated with a slightly increased risk for adverse pregnancy and infant outcomes compared with monotherapy,349 monotherapy treatment is preferred. Clinicians are encouraged to consider a higher dosage of a single medication (or augmenting with psychotherapy when available) before prescribing multiple medications. For those presenting on 2 or more agents, it is recommended that clinicians take a careful history of the indication for each medication and its effectiveness, to determine whether one or more medications can be safely discontinued. Caution is recommended not to expose, or maintain exposure, to medications that are not clearly effective. However, lowering the dose or minimizing the number medication exposures should not be at the expense of inadequate treatment response. In this guideline, given the minimal available safety evidence about medication combinations, Lines of Treatment are presented for monotherapies only. Because combination treatment is often needed (especially in severe depression, OCD, BD and/or postpartum psychosis), clinicians may need to use the available safety evidence for monotherapies or medication classes to inform the best approach for a specific patient.
MDD
Pharmacotherapy in pregnancy is reserved for individuals who have a severe MDE, those at high risk for relapse, those who are unable to access or have an inadequate response to psychotherapy, or those who prefer to use medication. For those already taking maintenance antidepressant medication, there should be counselling around the risk for relapse if medication were to be discontinued for pregnancy. Discontinuation of antidepressants in pregnancy has been associated with an elevated risk for relapse among those with a more severe depression history (i.e., multiple previous episodes, a greater severity of depression), but population-level studies suggest that those with mild-to-moderate depression severity may be not at significantly increased risk.350 Patients should also be counselled on risk for antidepressant discontinuation syndrome, which could be particularly unpleasant if the patient is already experiencing nausea or other physical symptoms in pregnancy. The threshold for pharmacotherapy is lower in the postpartum because the main issue is transmission to infants via breast milk, and individuals could choose not to breastfeed if they have concerns about infant medication exposure.
Pregnancy. Antidepressant medications are the mainstay of pharmacotherapy treatment for MDD in pregnancy (Table 10). The first-line medications—citalopram, escitalopram and sertraline—are first-line medications outside of the perinatal period, generally well-tolerated and have the most reassuring safety data in pregnancy. The second-line medications are all first-line medications outside the perinatal period, but are downgraded here as initial selections because either there are some (albeit minimal) safety concerns, there is less certainty about their safety, or tolerability is not as good as that of the first-line medications. Selection from within second-line medications can be made by clinicians and patients based on what patients prefer, and how they value the various risks, side effects and benefits presented. Paroxetine is listed as a third-line agent in pregnancy because of its links, albeit somewhat inconsistent, to a higher risk for cardiac malformations. However, it is reasonable to prescribe paroxetine if patients have had a good response to it and prefer it over other available agents. In this case, Level II ultrasonography in pregnancy is recommended to evaluate for cardiac malformations . Several medications are listed as having insufficient safety data, including certain newer agents and the MAOI tranylcypromine for which the case reports that exist are of concern (Level 4 ).273 This does not mean automatic discontinuation in people who have had a good clinical response, but patients should be advised accordingly. Ketamine—increasingly being used in treatment-resistant MDD outside the perinatal period—may be particularly problematic, and likely patients should not use it during pregnancy until adequate safety data are available (Level 4 ).351
Table 10.: Medications for Major Depressive Disorder in Pregnancy.
| Line of treatment | Medication | Level of evidence efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Citalopram | Most reassuring safety data of all antidepressants | ||
| Escitalopram | ||||
| Sertraline | ||||
| Second-line | Bupropion | Less safety data than first-line agents, and rare case reports of infant seizure if continued in lactation | ||
| Desvenlafaxine | Less safety data than first-line agents | |||
| Duloxetine | Some uncertainty about spontaneous abortion risk | |||
| Fluoxetine | Possible higher PPHN and PNAS risk and longer half-life/higher passage into breastmilk if continued in lactation | |||
| Fluvoxamine | Maternal tolerability concerns at higher doses | |||
| Mirtazapine | Less safety data than first-line agents and some uncertainty about spontaneous abortion risk | |||
| Venlafaxine XR | Possible higher PNAS riskSome uncertainty about spontaneous abortion risk | |||
| Third-line | Paroxetine | Possible higher risk for CV malformations and PNAS | ||
| Quetiapine | Maternal and fetal metabolic impacts, sedation | |||
| Trazodone | Maternal tolerability concerns at high doses | |||
| Tricyclicsa | Maternal tolerability concerns | |||
| Insufficient data | Agomelatine, dextromethorphan/bupropion, ketamine, levomilnacipran, mianserin, milnacipran moclobemide, phenelzine, reboxetine, tranylcypromine (case reports of malformations and of fetal death), vilazodone, vortioxetine | |||
Note. CV = cardiovascular; insufficient data = insufficient data on safety in pregnancy/lactation; PNAS = poor neonatal adaptation syndrome; PPHN = persistent pulmonary hypertension of the newborn; SSRIs = selective serotonin reuptake inhibitors; SNRIs = serotonin-norepinephrine reuptake inhibitors.
Tricyclics (amitriptyline, clomipramine, desipramine, doxepin, imipramine, nortriptyline, protriptyline, trimipramine) are not listed individually herein as it is difficult to separate their safety within the class of medications and their tolerability issues render them as lower in the treatment line of agents (after SSRIs, SNRIs, and others) non-perinatally in any case.
Postpartum. Antidepressants are also the mainstay of pharmacotherapy postpartum. It is expected that the efficacy of antidepressants would be similar in the postpartum as in non-perinatal periods, but there is also some RCT evidence for the efficacy of SSRIs over placebo in treating PPD specifically (Level 2 ).352 For those who are breastfeeding, there is reassuring data for several antidepressants in terms of their safety in lactation to guide recommendations about lines of treatment (Table 11). The only antidepressant not recommended in lactation is the TCA doxepin given its potential to cause excessive somnolence in exposed infants.281 Insufficient safety data exist for most newer agents. However, if a patient and clinician feel that the benefit of continuing that medication outweighs its drawbacks, they may select it, and close infant monitoring is recommended.
Table 11.: Medications for Major Depressive Disorder in Lactation.
| Line of treatment | Medications | Level of evidence (efficacy vs. placebo) | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Citalopram | |||
| Escitalopram | Most reassuring safety data of antidepressants | |||
| Sertraline | ||||
| Second-line | Bupropion | Rare case reports of infant seizure | ||
| Desvenlafaxine | Less safety data than first-line agents | |||
| Duloxetine | Less safety data than first-line agents | |||
| Fluoxetine | Longer half-life and higher RID than first- and second-line agents, more side effects reported, no severe concerns | |||
| Fluvoxamine | Less tolerability at higher doses | |||
| Mirtazapine | Less safety data than first-line agents and potential for maternal sedation | |||
| Venlafaxine | Less tolerability than first-line agents | |||
| Third-line | Paroxetine | Downgraded due to possible higher risk for CV malformations in a future pregnancy | ||
| Quetiapine | Maternal metabolic impacts, sedation | |||
| Trazodone | Maternal tolerability concerns at high doses | |||
| Tricyclics (except doxepin) | a | Maternal tolerability concerns | ||
| Not recommended | Doxepin | n/a | n/a | Concern about excessive infant sedation |
| Insufficient data | Agomelatine, brexanolone, dextromethorphan/bupropion, ketamine, levominacipran, mianserin, milnacipran, moclobemide, phenelzine, reboxetine, tranylcypromine, vilazodone, vortioxetine, zuranolone | |||
Note. Insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in lactation; n/a = not applicable as safety concerns outweigh the value of efficacy evidence.
One trial identified in the systematic review of antidepressant efficacy for PPD (n = 58) found problem-solving therapy was superior to amitriptyline in the treatment of postpartum depression in Africa (Level 2 ).353
For those who are not breastfeeding, concerns about exposure to the infant do not apply, so general guidelines for treatment of MDD can be followed, also incorporating the possibility of postpartum-depression specific pharmacotherapies. Brexanolone (IV) and zuranolone (PO) (not yet available in Canada) are synthetic analogs of allopregnanolone that have been shown in RCTs to be effective for treating PPD in those who developed depression in the 3rd trimester of pregnancy or in the first 4 weeks postpartum specifically (Level 1 ).252,253 The onset of symptom reduction appears to be within hours for brexanolone and within days for zuranolone. However, common side effects of these medications include headache, dizziness and drowsiness or sedation, and brexanolone requires about 72 h of continuous monitoring in an inpatient setting for the intravenous infusion due to the potential for excessive sedation and sudden loss of consciousness. There is as yet an insufficient amount of safety data on these agents in lactation, so until data in lactation are available, they can be considered outside of lactation where available, feasible to administer and not cost-prohibitive.
There have been 2 small RCTs that studied psychotropic medications vs. placebo (sertraline n = 22, nortriptyline, n = 51) for preventing PPD in euthymic individuals with a history of PPD not taking medication. Sertraline was protective against PPD (Level 3 ) but nortriptyline was not (Level 2 ).354 Given the uncertainty of the available evidence, clinicians are advised to consider risk for patient relapse on an individualized basis when making recommendations about whether to start (or restart) antidepressants in euthymic patients with a history of PPD in the postpartum period.
Anxiety Disorders
While 7 anxiety disorders exist in the DSM-5, there is currently no formally recognized, distinct perinatal anxiety disorder. For individuals presenting in pregnancy or in pre-pregnancy planning on maintenance medication therapy, the principles outlined for decision-making in MDD (see above) can be applied for anxiety disorders, as there are no data specifically on risk for relapse with discontinuation of medication in pregnancy. The potential implications of relapse should be considered based on the nature of the person’s anxiety disorder, and the risks of impact of relapse or symptom recurrence on daily function.
There are no RCTs evaluating pharmacological treatments for anxiety disorders perinatally. In this guideline, formal treatment recommendations for initial medication selection are made for Generalized Anxiety Disorder (GAD), the most common anxiety disorder seen perinatally, although the principles can be applied for other anxiety disorders by consulting treatment guidelines for the other anxiety disorders along with the safety information in pregnancy and lactation presented herein. In perinatal GAD, antidepressants are also considered to be the main pharmacotherapeutic treatments, although medications from other classes are recommended in non-perinatal treatment guidelines. For GAD in pregnancy (Table 12), valproic acid is not recommended due to its teratogenicity (Box 3), nor is regular/daily benzodiazepine use (see sedative and hypnotics above). While no agents used to treat anxiety disorder in the postpartum period are absolutely contraindicated, there are inadequate data to recommend most newer agents in lactation and it is not recommend to start valproic acid due to risk for future pregnancy (Table 13).
Table 12.: Medications for Generalized Anxiety Disorder in Pregnancy.
| Line of treatment | Medication | Level of evidence efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Escitalopram | Most reassuring safety data of antidepressants | ||
| Sertraline | ||||
| Second-line | Citalopram | Less efficacy data than first-line agents, but reassuring safety data | ||
| Duloxetine | Less safety data than first-line agents, some uncertainty about spontaneous abortion risk | |||
| Fluoxetine | Not as much efficacy data and possible higher PPHN and PNAS risk, longer half-life and passage into breastmilk if continued in lactation | |||
| Venlafaxine | Higher PNAS risk than first-line agents, some uncertainty about spontaneous abortion risk | |||
| Third-line | Bupropion XL | Less efficacy data than higher-ranked agents, rare case reports of infant seizure if continued in lactation | ||
| Imipramine | Maternal tolerability concerns | |||
| Mirtazapine | Less efficacy and safety data than higher-ranked agents, uncertainty about spontaneous abortion risk | |||
| Paroxetine | Possible higher risk for CV malformations and PNAS than higher-ranked agents | |||
| Pregabalin | Concern for neurodevelopmental impacts | |||
| Quetiapine | Maternal sedation and metabolic effects | |||
| Trazodone | Maternal tolerability concerns at higher doses | |||
| Not recommended | Daily benzo-diazepines | n/a | n/a | Respiratory problems, somnolence and NICU admission in infants; inconsistent associations with congenital malformations; maternal dependence risk |
| Valproic acid | n/a | n/a | Substantially elevated risk for congenital malformations and child developmental delay | |
| Insufficient data | Agomelatine, buspirone, hydroxyzine, vilazodone | |||
Note. CV = cardiovascular; insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence; NICU = neonatal intensive care unit; PNAS = poor neonatal adaptation syndrome; PPHN = persistent pulmonary hypertension of the newborn.
Table 13.: Medications for Generalized Anxiety Disorder in Lactation.
| Line of treatment | Medications | Level of evidence for efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Escitalopram | Most reassuring safety data of antidepressants | ||
| Sertraline | ||||
| Second-line | Citalopram | Less efficacy data than first-line agents, but reassuring safety data | ||
| Duloxetine | Less safety data than first-line agents | |||
| Fluoxetine | Longer half-life and higher RID than first- and second-line agents, more side effects reported in infants | |||
| Venlafaxine | Less tolerability than first-line agents | |||
| Third-line | Bupropion XL | Less efficacy data, case reports of infant seizure | ||
| Imipramine | Maternal tolerability concerns | |||
| Lorazepam | Caution for infant sedation | |||
| Mirtazapine | Less efficacy data than second-line agents and possible maternal sedation | |||
| Paroxetine | Downgraded due to possible higher risk for CV malformations in a future pregnancy | |||
| Pregabalin | Few data in lactation, caution if future pregnancy | |||
| Quetiapine | Maternal sedation and metabolic effects | |||
| Trazodone | Maternal tolerability concerns at higher doses | |||
| Not recommended | Valproic acid | n/a | n/a | Concern re: starting this medication in lactation due to risk for future pregnancy. Can continue for lactation if was taking in pregnancy |
| Insufficient data | Agomelatine, buspirone, hydroxyzine, vilazodone | |||
Note. CV = cardiovascular; insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence; RID = relative infant dose.
OCD
There are no RCTs evaluating pharmacological treatments for OCD perinatally. The mainstays of medication treatment for OCD are antidepressants that inhibit serotonin reuptake (e.g., the SSRIs and the TCA clomipramine). In non-perinatal samples, higher doses tend to produce greater symptom improvement and remission rates than lower doses, and for some medications, dosages that exceed dose monograph recommendations may be needed.355 Medication recommendations for OCD are in Tables 14 and 15 for pregnancy and lactation, respectively. No first- or second-line pharmacotherapy treatments in the most recent CANMAT OCD guidelines fall into the not recommended or insufficient data category.356 Similar to anxiety disorders, there are no specific data on relapse risk if maintenance pharmacotherapy is discontinued, and the principles of depression management apply here as well.
Table 14.: Medications for Obsessive–Compulsive Disorder in Pregnancy.
| Line of treatment | Medications | Level of evidence for efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Citalopram | Most reassuring safety data, slightly less non-perinatal efficacy data | ||
| Escitalopram | Most reassuring safety data | |||
| Fluvoxamine | Reassuring safety data, maternal tolerability at higher doses may limit | |||
| Sertraline | Most reassuring safety data | |||
| Second-line | Clomipramine | Less well tolerated than first-line agents; however, safety data are reassuring | ||
| Fluoxetinea | Possible higher PPHN and PNAS risk than first-line agents and longer half-life and greater passage into breastmilk if continued into lactation | |||
| Third-line | Paroxetineb | Possible higher CV malformation and PNAS risk | ||
| Venlafaxinec | Less non-perinatal efficacy data, possible higher PNAS risk than first- and second-line agents | |||
Note. CV = cardiovascular; PNAS = poor neonatal adaptation syndrome; PPHN = persistent pulmonary hypertension of the newborn.
Dosages required may go up to 80 mg; bDosages required may go up to 60 mg; cDosages required may go up to 300 mg;
Table 15.: Medications for Obsessive–Compulsive Disorder in Lactation.
| Line of treatment | Level of evidence efficacy | Justification for placement | ||
|---|---|---|---|---|
| Medications | Non-perinatal | Postpartum | ||
| First-line | Citalopram | Most reassuring safety data, slightly less non-perinatal efficacy dataMost reassuring safety data | ||
| Escitalopram | ||||
| Fluvoxamine | Reassuring safety data, maternal tolerability at higher doses may limit | |||
| Sertraline | Most reassuring safety data | |||
| Second-line | Clomipramine | Tolerability concerns, and slightly less volume of safety data than first-line agents | ||
| Fluoxetinea | Longer half-life and higher RID than first and second-line agents, more side effects reported, no severe concerns | |||
| Third-line | Paroxetineb | Downgraded due to possible higher risk for CV malformations in a future pregnancy | ||
| Venlafaxinec | Less efficacy than first-line agents, and maternal tolerability | |||
Note. CV = cardiovascular; RID = relative infant dose.
Dosages required may go up to 80 mg; bDosages required may go up to 60 mg; cDosages required may go up to 300 mg.
PTSD
There are no RCTs evaluating pharmacological treatments for PTSD perinatally. As such, CANMAT recommends that the use of pharmacological treatment for PTSD perinatally follows the guidance from non-perinatal clinical practice guidelines for PTSD. Non-perinatal guidelines recommend psychological therapies as first-line treatments (i.e., before pharmacological therapies) unless these are not available, severity of illness is so high that patients are not able to engage in that type of therapy without stabilization with medication, or medication is preferred.347 When pharmacological treatments are used, recent U.S. clinical practice guidelines (updated 2023) recommend only the antidepressant medications sertraline, venlafaxine and paroxetine, indicating that the evidence for other medications is either of low quality or that evident benefits do not outweigh potential harms or tolerability issues.347 Fluoxetine was at one time also in the list in a previous version of this guideline, and other guidelines.347 However a recent systematic review has raised questions about its efficacy.357 Based on these recommendations, for pharmacological treatment of PTSD in perinatal populations, sertraline is considered first-line in pregnancy, with venlafaxine as second-line and paroxetine as third-line due to its risk profile (Table 16). In the postpartum, sertraline remains the first-line treatment recommendation. If another pregnancy is not planned (and birth control measures are in place), then paroxetine may become second-line together with venlafaxine (Table 17). Prazosin is a medication that has been inconsistently recommended in clinical practice guidelines for the treatment of nightmares specifically in PTSD. CANMAT does not recommend the use of prazosin perinatally as data during pregnancy are limited with some concerning fetal effects, and there are very minimal data available on its safety in lactation (Level 4 ).358
Table 16.: Medications for Post-traumatic Stress Disorder in Pregnancy.
| Line of treatment | Medications | Level of evidence for efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Sertraline | Most reassuring safety data | ||
| Second-line | Venlafaxine | Possible higher PNAS risk than first-line | ||
| Third-line | Paroxetine | Possible higher CV malformation and PNAS risk than first-line | ||
| Not recommended | Prazosin | n/a | n/a | Concern re: fetal effects |
Note. CV = cardiovascular; not recommended = not recommended due to concerns about safety in pregnancy; n/a = not applicable as safety concerns outweigh the value of efficacy evidence; PNAS = poor neonatal adaptation syndrome.
Table 17.: Medications for Post-traumatic Stress Disorder in Lactation.
| Line of treatment | Medications | Level of evidence for efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Sertraline | Most reassuring safety data | ||
| Second-line | Paroxetine | Less tolerability than first-line agent, and also downgraded due to possible higher risk for CV malformations in a future pregnancy | ||
| Venlafaxine | Less tolerability than first-line agent | |||
| Insufficient data | Prazosin | n/a | n/a | |
Note. CV = cardiovascular; insufficient data = insufficient data on safety in lactation; n/a = not applicable as safety concerns outweigh the value of efficacy evidence.
Bipolar Disorder
Although there are no RCTs for the pharmacotherapeutic treatment of BD perinatally, the mainstay of acute and maintenance treatment for BD in pregnancy and postpartum is medication, primarily certain antiepileptics, antipsychotics and lithium. The specific medications that are advised may differ depending on the type of illness (i.e., BD type I or type II), and phase of illness, namely acute depression, mania/mixed states and maintenance treatment. In the postpartum, if not considering lactation, treatment recommendations can be based on general CANMAT BD guidelines, except for postpartum psychosis (see Question 9). Clinical pearls for prescribing in perinatal bipolar disorder are in Box 7.
Box 7.
Clinical pearls for prescribing in perinatal BD
Maternal medical concerns may impact medication choices. For instance, quetiapine and olanzapine, due to concerns about metabolic impacts, may be downgraded in those who have risk factors for gestational diabetes (i.e., high body mass index, family history of type 2 diabetes), given diabetes increases the risk of pregnancy and neonatal complications. Similarly, lithium may be downgraded or not recommended in those with renal problems. For infants born preterm or with medical comorbidities, it is important to try to avoid medications with risks for adverse effects due to breastmilk exposure and those without data. If this is not an option, formula feeding is a recommended alternative to discontinuation of an effective agent. Discontinuation of effective treatment significantly increases the risk of episode recurrence in BD, particularly postpartum.
Maintenance
Prospective non-randomized studies in BD show that maintaining pharmacological treatment in pregnancy significantly decreases risk of symptom re-emergence (Level 3 ).359 Abruptly discontinuing mood stabilizers (i.e., antiepileptics, antipsychotics, lithium) in pregnancy leads to a much more rapid relapse (∼2 weeks after stopping the treatment) versus gradual discontinuation (∼22 weeks afterward) (Level 3 ).359 In most cases, the risk of discontinuation of medication will generally outweigh the potential safety concerns of maintenance treatment in pregnancy, even for medications that are likely to be associated with small elevations in risks for congenital malformations and other pregnancy complications (see Boxes 2boxed-text3-07067437241303031–4). Patients will need to be counselled about the various benefits and risks of their medications in the context of their clinical condition and relapse risk, but it is CANMAT’s opinion that most patients in remission who choose to stay on pharmacological treatment should be supported in continuing existing medication regimens in pregnancy. A notable exception to this recommendation is valproic acid. Serious consideration should be given to switching valproic acid to another medication even if it has been an effective treatment given the elevated risk for congenital malformations and neurodevelopmental problems in offspring (see Box 2).
Bipolar disorder has the highest relapse rate for any mental disorder in the postpartum period, at on average, 37%.24 Most relapses are non-psychotic depressive episodes, but as many as 1 in 6 (17%) will be episodes of psychosis, mania, or other severe presentations requiring psychiatric hospitalization.24 Risk of an acute mood episode postpartum is as high as 66% in those not taking vs. 23% in those taking medication.24 As such, continuation of pregnancy medications into the postpartum is strongly recommended and initiation of medication in the immediate postpartum is generally recommended to prevent relapse in those who did not take medication in pregnancy. The potential for adverse effects on the infant due to medication exposure in lactation (which will vary based on the medication) needs to be considered, as does the potential for maternal sleep deprivation. Sleep interruption is expected with a newborn but can be exacerbated when the patient is waking frequently at night to feed their infant. For this reason, plans for infant feeding must be tailored to the individual patient. Where possible, limiting the amount of patient waking overnight by pumping milk in advance or using formula or donor milk, and having a partner, family member, doula or night nurse do some or all of the nighttime infant feedings may optimize longer durations of consolidated sleep (Level 4 ).
Acute Treatment of Mania and Mixed Episodes
Monotherapies that are considered first-line treatment options for acute mania outside the perinatal period in the CANMAT/International Society for Bipolar Disorders (ISBD) bipolar disorder clinical practice guidelines are asenapine, aripiprazole, cariprazine, lithium, paliperidone, quetiapine, risperidone and valproic acid.7 In pregnancy, quetiapine (despite concerns about a small increased risk for metabolic and sedative side effects) and aripiprazole (despite fewer data being available than for quetiapine) remain first-line due to their relatively reassuring safety profiles (Table 18). Risperidone is downgraded to second-line due to a possible higher risk for congenital malformations than the previous agents. Lithium is also downgraded to second-line due to concerns about safety and complexity in its use in pregnancy, but should remain an important option to consider due to its effectiveness in preventing relapse after the acute phase. There are some data, although limited, on the safety of paliperidone in pregnancy, so it can be used but is downgraded to third-line in favour of the above-mentioned options for which there are more data available. Valproic acid is not recommended perinatally due to safety concerns. There are not enough safety data in pregnancy to reassuringly recommend other first-line medications (asenapine, cariprazine).
Table 18.: Medications for Acute Mania in Pregnancy.
| Line of treatment | Medications | Level of evidence for efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Aripiprazolea | Safety data mostly reassuring, although fewer safety data than quetiapine | ||
| Quetiapine | Safety data most reassuring, despite possible increased risk for gestational diabetes | |||
| Second-line | Haloperidol | Safety data reasonably reassuring, but maternal tolerability and relapse prevention lower than first-line | ||
| Lithium | Highly effective including for relapse prevention, but elevated risk for multiple adverse outcomes and complex to manage during pregnancy (see Lithium section) | |||
| Olanzapine | Gestational diabetes higher than quetiapine, should switch after lactation due to metabolic effects | |||
| Risperidoneb | Possible higher risk for congenital malformations, not as good as lithium in relapse prevention | |||
| Third-line | Carbamazepineb | Increased risk for congenital malformations and Vitamin K deficiency | ||
| Chlorpromazine | Less efficacy data than higher-ranked agents, few specific safety concerns other than class-effects | |||
| Clonazepam | Less efficacy data than higher-ranked agents and fetal safety with exposure across pregnancy a concern | |||
| Paliperidone | Safety data volume is small | |||
| Ziprasidone | Less efficacy data than higher-ranked agents and safety data volume are minimal | |||
| Not recommended | Tamoxifen | n/a | n/a | Teratogenic potential |
| Valproic acid | n/a | n/a | Substantially elevated risk for congenital malformations and child developmental delay | |
| Insufficient data | Asenapine, cariprazine, clozapine | |||
Note. insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in pregnancy/lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence.
Also first-line treatment for mixed states; bMain concern is for congenital malformations so could be considered higher in the line of treatment when used after first trimester.
Olanzapine, although an excellent anti-mania agent, is second-line outside the perinatal period due to concerns about metabolic impacts. Its safety data are relatively reassuring in pregnancy, despite a possible increased risk for gestational diabetes, but it can cause sedation, so remains a second-line agent herein. If used in pregnancy, a switch to a more metabolically friendly option should be considered to prevent relapse after lactation. Haloperidol, another second-line agent non-perinatally, also remains second-line given its reasonable reproductive safety profile, although as it does not have strong evidence for relapse prevention, it would also likely need to be switched to another medication after the acute phase. Carbamazepine and ziprasidone are second-line agents outside the perinatal period, but are downgraded to third-line perinatally due to the potential for malformations and vitamin K deficiency with carbamazepine, and the limited amount of safety data available for ziprasidone. Other non-perinatal third-line medication options remain third-line perinatally, except for tamoxifen which is not recommended due to safety concerns (Level 4 ).360 For recommendations on the management of acute agitation, see Question 9.
Monotherapies considered first-line treatments for the traditional DSM-IV definition of a mixed state in BD (i.e., full criteria for depression and mania are met at the same time) are asenapine and aripiprazole.361 As such, aripiprazole is the recommended first-line agent in pregnancy in this case. As yet, there are no medications that meet evidence levels to be first-line recommendations for the DSM-5 definitions of depression with mixed features or mania with mixed features.361
In lactation, quetiapine remains first-line for mania despite its metabolic effects and potential for sedation, as does aripiprazole although the volume of safety data is less than for quetiapine and it may reduce breastmilk. Data in lactation are also reasonably reassuring for risperidone so it can also be used as a first-line option. There is more potential for metabolic effects and sedation with olanzapine than the above-mentioned drugs, but its reasonable safety profile in lactation suggests that it can be considered a second-line option for acute mania. Switching from olanzapine to a more metabolically friendly option for relapse prevention in the longer-term would be advisable. Other potential medications are ranked accordingly based on what is known about their efficacy outside the perinatal period, and their safety in and impact on lactation (Table 19). As in pregnancy, aripiprazole is the first-line agent recommended for acute mixed states in lactation.
Table 19.: Medications for Acute Mania in Lactation.
| Line of treatment | Medications | Level of efficacy | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Aripiprazolea | Reassuring safety data in lactation although less data than for quetiapine and may reduce breastmilk production | ||
| Quetiapine | Reassuring safety data in lactation, although may cause maternal sedation | |||
| Risperidone | Some adverse effects in lactation (more than quetiapine) | |||
| Second-line | Carbamazepine | Less efficacy data than higher-ranked agents, some adverse effects reported in lactation | ||
| Haloperidol | Maternal tolerability lower, relapse prevention less than higher-ranked, is linked to some adverse lactation effects, can reduce breastmilk | |||
| Lithium | Highly effective, including for relapse prevention, but multiple concerns with lactation safety | |||
| Olanzapine | Reassuring safety data in lactation, although may cause maternal sedation (try lower doses) and will need to be switched longer-term due to metabolic effects | |||
| Paliperidone | Very little known but is active metabolite of risperidone, so may have similar impact | |||
| Third-line | Chlorpromazine | Less efficacy data than higher-ranked agents, may lead to infant drowsiness and neurodevelopmental effects, can reduce breastmilk | ||
| Clonazepam | Less efficacy data than higher-ranked agents, concern about infant sedation and other developmental problems, although hard to separate from pregnancy effects | |||
| Ziprasidone | Less efficacy data than higher-ranked agents and safety data volume are minimal | |||
| Not recommended | Tamoxifen | n/a | n/a | No safety data and can suppress lactation362 |
| Valproic acid | n/a | n/a | Concern re: starting this medication in lactation due to risk for future pregnancy. Can continue for lactation if was taking in pregnancy | |
| Insufficient data | Asenapine, cariprazine, clozapine | |||
Note. Insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in pregnancy/lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence.
Also first-line treatment for mixed states.
Bipolar I Depression
For acute BD I depression, quetiapine and lamotrigine are first-line recommendations in pregnancy, although it may be sometimes challenging to reach therapeutic levels with lamotrigine (and thus a therapeutic effect) in pregnancy due to the significant increase in metabolism (Table 20). Lithium, a first-line agent non-perinatally, is second-line in pregnancy due to its safety and use profile. As there are minimal safety data in pregnancy for lurasidone (another first-line agent non-perinatally) it is not included in the treatment recommendations here. Neither of the 2 non-perinatal second-line options (valproic acid and cariprazine) are recommend herein due to safety concerns and insufficient safety data, respectively. While olanzapine is efficacious, it is considered a third-line option outside of the perinatal period due to the potential for metabolic side effects. In pregnancy, however, due to its relatively favourable reproductive safety profile, it is a reasonable second-line medication in pregnancy, with a plan to change to a medication with a more favourable metabolic profile after lactation. Carbamazepine is considered a third-line option based on both its efficacy profile and the higher risk for congenital malformations than lamotrigine.
Table 20.: Medications for Acute Bipolar I Depression in Pregnancy.
| Line of treatment | Medications | Efficacy level of evidence | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Lamotrigine | Safety data reassuring; less efficacy data than quetiapine | ||
| Quetiapine | Safety data reassuring, despite possible increased risk for gestational diabetes | |||
| Second-line | Lithium | Elevated risk for multiple adverse outcomes and complex to manage during pregnancy (see Lithium section) | ||
| Olanzapine | Safety data in pregnancy reasonably reassuring despite risk for gestational diabetes higher than quetiapine, should switch long-term due to metabolic effects | |||
| Third-line | Carbamazepine | Less efficacy data than higher-ranked agents, increased risk for congenital malformations and Vitamin K deficiency | ||
| Not recommended | Valproic acid | n/a | n/a | Elevated risk for congenital malformations and child developmental delay |
| Insufficient data | Cariprazine, lurasidone | |||
Note. Not recommended = not recommended due to concerns about safety in pregnancy/lactation; insufficient data = insufficient data on safety in pregnancy/lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence.
In the postpartum, lamotrigine is a first-line medication given that its metabolism normalizes and it has a favourable reproductive safety profile in lactation (Table 21). Quetiapine follows as a first-line treatment, due to concern about maternal metabolic effects. The remainder of the monotherapies follow the same order as the recommendations in pregnancy, for similar reasons. There is some emergent safety data for lurasidone, which has Level 2 evidence for efficacy outside the perinatal period, however, the lactation safety data are not yet clear enough to support recommending this medication. Valproic acid is not recommended due to concern about future pregnancies in the reproductive-aged population, unless there is a reasonable clinical rationale, patient preference and use of a highly-effective method of contraception.
Table 21.: Medications for Acute Bipolar I Depression in Lactation.
| Line of treatment | Medications | Efficacy level of evidence | Justification | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Lamotrigine | Minimal cause for concern with lactation or maternal sedation | ||
| Quetiapine | Minimal cause for concern with lactation but some maternal sedation | |||
| Second-line | Lithium | Multiple concerns with lactation safety | ||
| Olanzapine | Reassuring safety data in lactation, although may cause maternal sedation (try lower doses) and may need to be switched longer-term due to metabolic effects | |||
| Third-line | Carbamazepine | Less efficacy data than higher-ranked agents, some adverse effects reported in lactation | ||
| Not recommended | Valproic acid | n/a | n/a | Concern re: starting this medication in lactation due to risk for future pregnancy. Can continue for lactation if was taking in pregnancy |
| Insufficient data | Cariprazine, lurasidone | |||
Note. Insufficient data = insufficient data on safety in pregnancy/lactation; not recommended = not recommended due to concerns about safety in pregnancy/lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence.
Bipolar II Depression
For acute BD II depression, recommendations based on non-perinatal efficacy suggest quetiapine should be first-line, and lamotrigine and lithium second-line due to their more limited efficacy. We also consider lamotrigine as a reasonable first-line agent in pregnancy due to its favourable safety profile (Table 22). Due to its potential adverse effects, lithium remains second-line. In the postpartum, as quetiapine is associated with sedation, lamotrigine might be preferred for some patients. As such, in lactation and in the postpartum generally, we recommend either quetiapine or lamotrigine as first-line options, with lithium remaining second-line as it is in non-perinatal populations (Table 23).
Table 22.: Medications for Acute Bipolar II Depression in Pregnancy.
| Line of treatment | Medications | Efficacy level of evidence | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Pregnancy | |||
| First-line | Lamotrigine | Safety data reassuring; less efficacy data than quetiapine | ||
| Quetiapine | Safety data reassuring, despite possible increased risk for gestational diabetes | |||
| Second-line | Lithium | Possible higher risk for cardiovascular malformations and other fetal impacts | ||
| Sertralinea | Safety data reassuring (caution for possible antidepressant-induced mania) | |||
| Third-line | Fluoxetinea | Safety data less reassuring than sertraline, possible higher PPHN and PNAS risk than sertraline (caution for possible antidepressant-induced mania) and longer half-life and greater passage into breastmilk if used in lactation | ||
| Venlafaxinea | Safety data less reassuring than sertraline, possible higher PPHN risk than sertraline (caution for possible antidepressant-induced mania) | |||
| Ziprasidoneb | Safety data volume are minimal | |||
| Not recommended | Paroxetine | n/a | n/a | |
| Valproic acid | n/a | n/a | Elevated risk for congenital malformations and child developmental delay | |
Note. Not recommended = not recommended due to concerns about safety in pregnancy/lactation; n/a = not applicable as safety concerns outweigh the efficacy evidence; PNAS = poor neonatal adaptation syndrome; PPHN = persistent pulmonary hypertension of the newborn.
Pure, non-mixed depression; bDepression and mixed hypomania.
Table 23.: Medications for Acute Bipolar II Depression in Lactation.
| Line of treatment | Medications | Efficacy level of evidence | Justification for placement | |
|---|---|---|---|---|
| Non-perinatal | Postpartum | |||
| First-line | Lamotrigine | Less efficacy data than quetiapine; minimal cause for concern with lactation or maternal sedation | ||
| Quetiapine | Effective, minimal cause for concern with lactation but some maternal sedation | |||
| Second-line | Lithium | Effective but multiple concerns with lactation safety | ||
| Sertralinea | Less effective than higher-ranked agents, safety data very reassuring in lactation (caution for possible antidepressant-induced mania) | |||
| Third-line | Fluoxetinea | Less effective than higher-ranked agents, longer half-life and higher RID than first and second-line agents, more side effects reported, no severe concerns (caution for possible antidepressant-induced mania) | ||
| Venlafaxinea | Less tolerability than higher-ranked agents (caution for possible antidepressant-induced mania) | |||
| Ziprasidoneb | Less effective than higher-ranked agents, safety data volume in lactation are minimal | |||
| Not recommended | Paroxetine | n/a | n/a | |
| Valproic acid | n/a | n/a | Concern re: starting this medication in lactation due to risk for future pregnancy. Can continue for lactation if was taking in pregnancy. | |
Pure, non-mixed depression only; bDepression and mixed hypomania. RID = relative infant dose.
Antidepressant monotherapy is sometimes carefully used with close monitoring in the case of depression without mixed features outside the perinatal period even though it confers some risk for antidepressant induced mania or mixed states. Due to the elevated risk for postpartum psychosis among those with BD (even BD II) this is not an ideal strategy perinatally, especially in the first 3 months postpartum and especially as monotherapy without a mood stabilizing medication (see Question 9). That being said, sertraline, given its relatively favourable reproductive safety profile in pregnancy and lactation, could be a second-line treatment in pregnancy and in lactation, although it should likely be used after the potentially more effective second-line options. Clinicians should provide education to patients (and partners when possible) about the possibility of emergent manic or mixed symptoms, and develop a plan, including how and when to seek urgent assistance.
Third-line treatments are the other antidepressants with some efficacy data in BD II depression (fluoxetine and venlafaxine), but that have a less favourable reproductive safety profile than sertraline (see antidepressants section and tables for MDD recommendations above). The same precautions about risk for postpartum psychosis would apply as were described above for sertraline. Of the third-line monotherapies in non-perinatal populations, ziprasidone has some reassuring safety data in pregnancy and lactation so is a possibility. Valproic acid is not recommended in either pregnancy or postpartum due to safety concerns.
Question 7. What Are the Recommendations for the Use of Neuromodulation?
Neuromodulation (or neurostimulation) interventions comprise a group of treatments that affect the central nervous system via electrical or magnetic stimulation. In non-perinatal populations, non-invasive interventions such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) are generally used in acute MDEs when initial treatments have not been successful.7,11 They are delivered daily or multiple times daily over a course of several weeks, which has limited their uptake for some patients. These non-invasive neuromodulation interventions have begun to receive some attention in the perinatal period.
Several RCTs of rTMS for PPD suggest efficacy with minimal maternal side effects, mostly when used as an adjunctive treatment in cases of incomplete response to antidepressant medication; however, the quality of the evidence is limited in most trials (Level 2 ).363bibr364-07067437241303031–365 Given the strong evidence for its use non-perinatally, adjunctive rTMS is recommended for PPD.11 Adjunctive tDCS is recommended as a third-line option for mild-to-moderate PPD, as further evidence to substantiate its use is required in the perinatal population.
There has been one small RCT of rTMS for unipolar depression in pregnancy (monotherapy and as an adjunct to antidepressant medication; n = 26); although 3 of eleven participants receiving rTMS experienced late preterm birth versus none in a sham-control group (Level 3 ).366,367 There has also been a small-sample RCT (n = 20) of tDCS as monotherapy for unipolar depression in pregnancy that generated promising results (Level 3 ).368bibr369-07067437241303031bibr370-07067437241303031–371 Since safety data on rTMS and tDCS in pregnancy remain limited, these are not recommended as routine treatments for depression in pregnancy at this time. No recommendations can be made at this time for the use of rTMS or tDCS perinatally for anxiety, OCD, PTSD or BD.
ECT, which is typically delivered 2–3 times a week, requires a general anaesthetic and is associated with more adverse effects than less invasive treatments, is also included in this category. In non-perinatal populations, ECT is generally used in severe depression, mania, psychosis and/or acute suicidality.7,11 Maintenance ECT is vital in some cases of refractory depression, and ECT may work particularly quickly in treating acute psychotic depression and in treating mania, so its use is clinically important in the perinatal period as well.
Studies on ECT safety in the perinatal period are limited to case studies or series, often with no comparison groups, and there are no RCTs.372,373 Reports of ECT use in pregnancy are limited to severe depression, psychosis and mania (with and without medications). ECT appears relatively safe even when administered during the first trimester of pregnancy. Adverse effects reported in pregnancy include fetal arrhythmia, fetal bradycardia, premature birth, abdominal pain, uterine contractions, vaginal bleeding, placental abruption, threatened abortion and fetal death, but it is difficult to quantify risk attributable to ECT in these high-risk cases (Level 3 ).370,372,373
Based on these considerations, ECT is an appropriate first-line intervention for acute depression with severe psychotic or catatonic features, acute suicidality or deteriorating physical condition in pregnancy. ECT is also recommended for severe presentations of depression, mania, mixed states or psychosis, when pharmacological treatments are not successful. ECT in pregnancy should occur in close collaboration with anaesthesia and obstetrical services. While most general anaesthetics and muscle relaxants used in ECT administration are considered compatible with pregnancy, several strategies to optimize safety of the patient and foetus have been recommended.374 These include administering anti-nausea agents prior to the procedure, adding sodium citrate (oral) to prevent aspiration, avoiding hyperventilation prior to induction (as pregnancy is already a state of mild hyperventilation), elevation of the right hip (especially after 20 weeks gestation) to avoid aortal-caval compression of placental blood flow and fetal monitoring before, during and after procedure (including continuous fetal monitoring throughout the procedure and for some time afterward once it is possible to do so based on the number of weeks gestation). Intubation may be required depending on the stage of pregnancy to avoid the risk of aspiration.
Safety concerns with ECT are less in the postpartum relative to pregnancy. It can be used for severe cases as in non-perinatal populations with consideration given to the perinatal context (e.g., needs for assistance with childcare). It is generally recommended that patients can resume breastfeeding immediately after anaesthesia as the passage of anaesthetic medications into the breastmilk is minimal.375
Question 8. What Is Recommended for the Use of Complementary and Alternative Treatments?
CAM treatments are increasingly being studied for depression and anxiety in perinatal populations.11 Research on CAMs perinatally is mostly in populations with mild-severity depressive episodes (PPD or MDE in pregnancy) or elevated depressive or anxiety symptoms based on standardized symptom scale cut-off scores. The methodological quality of the available literature is typically low, precluding the ability to make recommendations for any CAMs as first-line treatments for the perinatal context.376 Further, evidence on the use and effectiveness of some CAMs, such as yoga, acupuncture (a key component of traditional Chinese medicine), and consuming specific foods and supplements, has been culturally- or country-specific, so may not necessarily apply outside of the settings in which evaluation occurred. However, it is clinically reasonable to consider the use of CAMs that have promising evidence for efficacy and minimal evidence for harm as adjunctive treatments and/or in individuals who prefer the use of CAMs.
Yoga can prevent depressive symptoms in pregnancy, and can reduce depressive symptoms in pregnancy and in PPD (Level 2 ); the evidence is mixed for the treatment of individuals with a clinical diagnosis of MDE in pregnancy, and for prevention and treatment of anxiety in pregnancy.172,178,237,377bibr379-07067437241303031bibr378-07067437241303031bibr379-07067437241303031bibr380-07067437241303031bibr381-07067437241303031bibr382-07067437241303031bibr383-07067437241303031bibr384-07067437241303031bibr385-07067437241303031bibr386-07067437241303031bibr387-07067437241303031bibr388-07067437241303031bibr389-07067437241303031bibr390-07067437241303031bibr391-07067437241303031–391 Acupuncture as an adjunct to the standard of care has evidence in reducing depressive symptoms postpartum and in PPD specifically (Level 2 ).392 Massage therapy in pregnancy (in some studies as an adjunct to the standard of care) is effective in decreasing depressive symptoms in mild-severity MDE and in reducing anxiety symptoms (Level 2 ).237,385,386,393,394 Music therapy in the postpartum as an adjunct to the standard of care decreases depressive symptoms in individuals with mild-severity PPD (Level 2 ).395,396 Beneficial effects have also been found for reduction of perinatal depressive and anxiety symptoms (Level 2 ) but there is not enough evidence to recommend it for prevention of depression or anxiety symptoms (Level 4 ).209,237,384,385,395bibr396-07067437241303031bibr397-07067437241303031bibr398-07067437241303031bibr399-07067437241303031–400
Multiple RCTs have examined the effectiveness of macronutrients and micronutrients for treating perinatal depression and anxiety. In the single positive trial, 1000 IU of Vitamin D taken daily for 6 months postpartum led to improvement in depressive symptoms (Level 2 ).401 In mild-severity PPD, 8 weeks of saffron (15 mg po bid) led to depressive symptom improvement in one small-sample trial with a very high risk of bias (Level 3 ).402 Trials of Omega-3 fatty acids for the treatment of perinatal depression have had mixed results, so a recommendation cannot be definitively made about their use at this time.403
Although some of these nutrients may be used for other purposes in pregnancy or postpartum (e.g., for protection against neural tube defects in pregnancy with folic acids, or treatment of anaemia with iron supplements), CANMAT does not recommend the use of folic acids, high flavonoid foods, nor iron, selenium, calcium, zinc, magnesium or copper for the prevention or treatment of depression in pregnancy or PPD because these interventions have generally not be shown to be effective for these conditions (Level 2, negative).403 St. John’s Wort is also not recommended in pregnancy or in lactation based on non-controlled, case-series data suggesting a higher-than-usual risk for adverse outcomes (Level 4 ).404,405 There is insufficient evidence to provide recommendations for aromatherapy,237,387,400,406bibr407-07067437241303031bibr408-07067437241303031bibr409-07067437241303031–410 relaxation,237,378 hypnosis174 or probiotics (Level 4 negative for treatment of depression and anxiety in pregnancy for the last three interventions in this list ).411,412
A summary of recommendations for CAM treatments for depression and anxiety symptoms are in Table 24.
Table 24.: Complementary and Alternative Medicine Treatments for Depression and Anxiety Symptoms.
| Line of treatment | Depression symptoms | Anxiety symptoms | ||
|---|---|---|---|---|
| Intervention | Level of evidence | Intervention | Level of evidence | |
| Prevention | ||||
| First-line | — | — | ||
| Second-line | Yoga in pregnancy | — | ||
| Insufficient or mixed evidence | Aromatherapy postpartumMusic postpartum | Music postpartumYoga in pregnancy | ||
| Treatment | ||||
| First-line | — | — | ||
| Second-line | Adjunctive acupuncture postpartuma | Massage therapy in pregnancy | ||
| Massage in pregnancy | Adjunctive music in pregnancy and postpartum | |||
| Adjunctive music in pregnancy and postpartum(a for postpartum) | — | |||
| Vitamin D in postpartum | — | |||
| Yoga in pregnancy and postpartum(a for postpartum) | — | |||
| Third-line | Acupuncture in pregnancya | Massage therapy postpartum | ||
| Massage in pregnancya | Relaxation in pregnancy | |||
| Saffron postpartuma | — | |||
| Not recommended | Folic acids, high flavonoid foods, nor iron, selenium, calcium, zinc, magnesium, or copper | — | ||
| St. John’s Wort | Safety concerns | — | ||
| Insufficient or mixed evidence | Aromatherapy postpartumHypnosisOmega-3 fatty acidsProbiotics in pregnancyRelaxationYoga for MDE in pregnancy | Aromatherapy postpartumHypnosisProbiotics in pregnancyRelaxationYoga in pregnancy | ||
Note. The level of evidence for saffron postpartum was downgraded to Level 3 due to its significant high risk of bias. Given the low quality of the evidence and the difficulty in classifying it according to the standards described, all interventions started below first-line. Adjunctive = adjunctive to the standard of care; MDE = major depressive episode.
aAlso effective in mild MDE.
Question 9. What Are the Recommendations for Managing High-Risk Clinical Situations?
When providing care for PMADs, clinicians should always assess a patient’s risk to themself or others, including the foetus, infant or older children. Clinically high-risk symptom presentations may include acute psychotic episodes (e.g., postpartum psychosis), periods of increased acute risk of suicide, acute intoxication or agitation, and other situations where there are thoughts about or evidence of potential for harm to self or others, including child abuse or neglect.
High-risk clinical symptom presentations can occur regardless of past psychiatric history but individuals with a previous psychiatric history are at increased risk. In assessing safety, particular attention should be paid to the following: In any high-risk situation, referral to expert mental health providers is warranted. Emergency services may be required, which can be accessed by calling 9-1-1 or 9-8-8 (mental health crisis line) in Canada and the U.S. and/or sending a patient to the emergency department if there is a concern about imminent harm to the patient or others. Clinicians should follow their local professional practice guidelines, laws and policies with respect to involuntary hospitalizations and when to report to child protection services.
- Inquire about suicidal thoughts, intent and/or plan, including access to means (e.g., medications for intentional overdose, firearms, knives).
- Inquire about the presence of a safety plan and other strategies to mitigate risk.
- Inquire about thoughts about harming the infant or others, and distinguish intrusive, ego-dystonic thoughts about harm from intent to harm.
- Assess for psychotic symptoms, including delusions, hallucinations and/or disorganized thoughts or behaviours.
- Inquire about and observe for signs of inability of the patient to take care of themselves and/or others, including child neglect or abuse.
- Pursue collateral history, for instance from family members, to clarify the level of risk, and assess family/social support capacity to care for the patient and the infant as well as the availability and use of other supports.
Postpartum Psychosis
Postpartum psychosis occurs after about 1–2 in 1000 births and is a very high-risk perinatal mental health condition due to its association with risk of maternal suicide and infanticide.413 “Postpartum psychosis” is a term that is generally used to refer to a specific unique mix of mood and psychotic symptoms that are strongly associated with BD.414,415 Symptoms typically start within the first week after delivery, and almost always in the first 4 weeks postpartum, although they can also begin later in the postpartum period.416 They can include delusions, hallucinations, irritability, mood swings/emotional lability, catatonia, agitation and/or hyperactivity. Disorientation, depersonalization and derealization can occur, but are less common than the mood symptoms.416 There can be rapid fluctuation in symptoms, including changes in the level of consciousness in a delirium-like presentation. This can make diagnosis complicated as there may be fluctuation in and out of psychotic states. In some cases, patients may present signs of impaired attachment with their offspring.
When considering a diagnosis of postpartum psychosis, it is important to consider that psychotic symptoms such as delusions, hallucinations and disorganization of thoughts and behaviours can occur in several contexts. The DSM-5-TR allows the use of a “peripartum onset specifier” to characterize episodes of psychosis across various diagnoses (e.g., MDD with psychotic features, BD). Psychotic symptoms can also be seen in the context of a primary psychotic disorder such as schizophrenia or schizoaffective disorder, with acute intoxication, or more rarely due to a general medical condition. There is a need to consider this differential diagnosis. Treatment recommendations will differ if a better explanation for the presentation would be a major depression with psychotic features, acute intoxication or a psychotic episode due to a general medical condition, instead of a postpartum psychosis. There should be a full physical and neurological examination and basic laboratory testing including urine toxicology, to rule out any organic cause. Additional laboratory tests and/or the use of EEG or neuroimaging scans may be indicated when there is clinical suspicion of epilepsy/seizures, traumatic brain injury or other serious medical conditions, including those that can cause neuropsychiatric symptoms (e.g., anti-NMDA receptor encephalitis or posterior reversible encephalopathy syndrome).
Prevention. The strongest risk factors for postpartum psychosis are a personal and family history of BD, and a previous history of postpartum psychosis.24,416bibr417-07067437241303031–418 As such, individuals with a personal (or familial) history of BD or postpartum psychosis should be provided with psychoeducation about their risk during or even before pregnancy. Sleep deprivation may precipitate psychotic symptoms in this group, although a reduced need for sleep can also be an early sign of psychotic mania.419 A treatment plan aimed at the prevention of psychosis should be offered, including consideration of prophylactic medication treatment and strategies to protect sleep (Level 4 ).419 Lithium appears to have the best evidence in the prevention of postpartum psychosis (likely in cases of a past history of postpartum psychosis and bipolar I disorder) (Level 4 ).420 However, those with BD in remission on appropriate antimanic or antipsychotic treatment in pregnancy should continue their maintenance pharmacotherapy into the postpartum (Level 4 ). Regardless, close monitoring is required in the early postpartum to identify early emerging symptoms. A longer hospital stay for monitoring and implementation of sleep protection strategies may also be considered, especially if the patient displays initial signs of mental distress and/or does not have appropriate social supports.
Primiparity as well as pregnancy and delivery complications, including pre-eclampsia, have also been identified as factors associated with postpartum psychosis, although most primiparous people and those with obstetrical complications do not develop it, so no specific recommendations are made in this regard.24,416bibr417-07067437241303031–418
Management. Postpartum psychotic symptoms require urgent assessment and almost always require immediate treatment, in an in-patient treatment setting. It is essential to assess risk to self, the newborn, other children in the home and other family members. Given the acute risks associated with postpartum psychosis, it is not uncommon that breastfeeding will need to be paused or discontinued for the benefit of both the patient and the baby. Clinicians need to provide psychoeducation and ongoing support for patients, family and other members of the patient’s support system. Individuals with possible psychosis and thoughts of harming themselves and/or the baby should not be left alone, especially with the baby, until a comprehensive risk assessment is completed (Level 4 ). Safety planning and the creation of a plan of action with the patient and their support system including active ongoing involvement of mental health professionals should be formulated, closely monitored, and maintained until the psychotic symptoms fully subside (Level 4 ).213
Postpartum psychosis treatment should be individualized to address the primary disorder and possible contributing psychiatric comorbidities, and to prevent recurrence of psychosis. For acute treatment, the initial recommendation is to start antipsychotic medication with or without an intermittent benzodiazepine (such as lorazepam) to manage insomnia and/or to decrease agitation (Level 4 ).420 Lithium may be added to enhance treatment response and also due to evidence for its efficacy in relapse prevention, taking into account patient preference and considerations about lithium in lactation (Level 4 ) (see Question 6).420 ECT is an option if rapid treatment is required, if there is insufficient response to pharmacotherapy, or if there is a past history of response to ECT (see Question 7).420 In the context of known BD, treatment of acute postpartum psychosis must take into account prior response to medications. In this case, lines of treatment for acute mania in the BD section can then be followed (see Question 6).7
After the acute episode of postpartum psychosis is treated, patients will require careful ongoing monitoring and follow-up as outpatients, preferably in mental health specialty care for a minimum of 6 to 12 months, with the antimanic agent maintained for a minimum of 12 months for relapse prevention (Level 4 ).420 Optimizing social support and involving the partner, carer and/or support system in the post-acute phase are also important (Level 4 ).420 Peer support may be a key resource during recovery (Level 4 ).420
Acute Agitation
Acute agitation—that is, ranging from self-reported symptoms to observable impatience, restlessness and aggression—in PMADs would be expected to occur mainly in the context of mania or psychosis, but can also occur in acute interpersonal crises, or other etiologies that should be managed accordingly (e.g., a medical condition, substance intoxication or withdrawal).
There are no RCTs on the management of acute agitation in PMADs. A sequential approach to management is recommended from the least to the most restrictive (Level 4 ),421,422 as follows: The first step for cooperative persons is verbal de-escalation. The next step is offering oral medication. In this case, a benzodiazepine can be used, with lorazepam preferred over longer-acting benzodiazepines. Monotherapy is preferred but if the agitation is more severe, especially in the context of psychotic symptoms, an antipsychotic can be combined with a benzodiazepine. If an individual has already started on an antipsychotic, then the dose can be optimized. In some cases, an antipsychotic can be prescribed on an “as needed” basis. Olanzapine is supplied in a rapid dissolving form as well as intramuscular. Quetiapine is sedating but has a slower onset of action and is only available in oral form. Ziprasidone has also been suggested as an alternative for a quick response, but has less safety data perinatally.423 Typical antipsychotics are less favoured in general psychiatry but haloperidol (available in oral, intramuscular or intravenous forms) has minimal perinatal risk so it can be used; loxapine is also a reasonable option but less is known about its safety perinatally than for haloperidol.424 If there is imminent risk of violence, physical restraint may be necessary. In this case, special care must be taken with positioning, especially in the second or third trimesters of pregnancy, when using 4-point restraints, to avoid compression of the vena cava. Physical restraints should be used for the shortest amount of time possible, and with fetal monitoring if available (Level 4 ).421
Risk of Suicide
Suicidal thoughts are estimated to occur in as many as 8% of pregnant people and birthing parents.425 Most people who experience suicidal thoughts do not attempt suicide, and in many cases, being pregnant or having a child is a protective factor against suicide.426 However, although maternal mortality is relatively rare, suicide is a leading cause of death in the perinatal period according to data from several countries.427 The highest risk for suicide appears to be during acute episodes of severe depression, mania/mixed states, psychosis and/or substance or alcohol intoxication. Inquiry about suicidal thoughts should be part of mental health assessments and follow-up in all individuals with PMADs. Inquiring about or monitoring suicidal thoughts does not increase the risk for suicidal thoughts or actions. Some of the most commonly used standardized clinical questionnaires (e.g., EPDS, PHQ-9) contain an item focused on suicidal thoughts, so these can be used to aid in inquiry.
In the absence of perinatal-specific evidence, it is recommended that the assessment and management of suicide risk is similar to that in non-perinatal populations (Level 4 ).11 Assessment involves asking detailed questions about the presence, frequency and persistence of suicidal thoughts, whether there are specific plans for suicide, intent, or means to carry out a plan, and the potential for lethality. For instance, individuals with specific plans, intent and means, especially high-lethality means, would be considered much higher risk than those without these characteristics. Suicidal thoughts in the context of psychosis or acute intoxication would also be high-risk situations. It is important to always ask specifically about whether there is intent to end the life of their foetus or infant.
In the management of suicide risk, optimizing treatment for the underlying PMAD is key. For individuals at high or imminent risk for suicide, hospitalization is required (involuntarily if needed). For those not hospitalized, management requires careful vigilance, optimization of social supports and close monitoring by partners, family members and care providers. Prevention considerations include removing access to lethal means (e.g., removal of sharp objects, oversight of medications due to risk of misuse or overdose, etc…). Clinicians can work with patients and their support systems to identify warning signs and create a list of coping strategies and resources to access if suicidal thoughts arise (e.g., resources for 24/7 support, such as crisis lines including 9-8-8 in the U.S. and Canada, and the closest emergency services). Safety plans should be revisited in every follow-up appointment and modified as needed.
Thoughts of Infant-Related Harm
Thoughts of infant harm are common in the postpartum period.428 In most cases, thoughts are intrusive and unwanted and do not represent a safety concern. When care providers receive a disclosure of thoughts of harming one’s infant, the thoughts should be fully assessed before referral to child protection in case the latter is unnecessary. Recurrent unwanted intrusive thoughts or images of harming an infant on purpose (intentionally) that cause distress to the mother, with no actual intent or desire to harm the infant (i.e., “ego-dystonic” thoughts) may be best conceptualized as obsessional symptoms, with low risk of infant harm.428 These ego-dystonic thoughts are very distressing, and many patients—especially those who are from population groups that have a history of experiencing discrimination in health care—may conceal them from others due to shame, stigma and/or fear that their infant will be removed from their care.429 When inquiring about thoughts of infant harm, clinicians can provide education about their occurrence and contextualize risk to reduce barriers to disclosure. For example, clinicians can explain that thoughts about infant-related harm are common, but usually only dangerous in the context of postpartum psychosis or suicidality.
When the patient expresses intent to harm their infant, does not appear distressed by thoughts of harming the infant (i.e., “ego-syntonic” thoughts), or when the thoughts occur in individuals with psychosis or those at high risk for suicide, there is a greater risk of infant-related harm.430 For example, patients suffering from postpartum psychosis may believe that their infant is inhabited by an evil spirit or alien and can only be saved from this terrible situation if the parent kills the infant. A mother with severe depression and suicidal thoughts may believe their infant would suffer if the mother were to die. In this instance, the patient may genuinely believe it is better for them to include their infant in the suicide plans. When any of these situations are present, the affected mother or birthing parent should not be left alone with the infant or other children, and treatment should be sought emergently (Level 4 ). Infant-related harm can also occur spontaneously or impulsively out of extreme frustration, and clinicians should always be attentive to signs of possible child abuse and/or neglect.
Question 10. What Are the Recommendations for Managing the Mental Health of Fathers/Co-parents?
Most research on PMADs in the non-birthing parent focuses on depression in fathers, where the prevalence of clinically significant perinatal depressive symptoms is between 8–10%.431 Risk factors are similar to those of maternal depression, including a history of mental illness and psychosocial stressors such as relationship distress and financial instability.55,432 Unemployment is an independent risk factor in this group, whereas in the maternal depression literature this appears to be of less significance.55,432 There is a strong correlation between maternal and paternal depression, and maternal depression is a strong risk factor for paternal depression.55,432,433 Anxiety has also been studied in fathers, with wide-ranging prevalence rates but an overall estimate for clinically significant anxiety symptoms of about 11% across the perinatal period.434 There is some suggestion that rates may increase over the course of the antenatal period, peaking in the early postpartum before decreasing thereafter.432 Paternal depression, and to a lesser extent paternal anxiety, is associated with a small increased risk for emotional and behavioural problems in children including after accounting for maternal mental health.435bibr436-07067437241303031–437
Less is known about PMADs in non-birthing cisgender sexual minority (e.g., lesbian, bisexual, queer) or gender minority (e.g., transgender, non-binary) co-parents. There is some evidence to suggest a higher prevalence of perinatal depressive symptoms in these groups.48,438,439 These populations may encounter specific challenges during the transition to parenthood, including navigating heteronormative healthcare systems, minority stress and invisibility.438bibr439-07067437241303031–440 Little is known about how factors such as race, ethnicity, culture and language may impact the mental health of these groups.439 Similarly, little is known about the prevalence of PMADs other than depression and anxiety in fathers or co-parents, although theoretically the risks of relapse could be high at this time for other disorders (including BD), given the stress of sleep deprivation and other factors in the transition to parenthood.
It is recommended that clinicians, including those who are treating the birthing parent or the child, be attentive to the mental health of the co-parent (Level 4 ). Co-parents may be reluctant to disclose mental health concerns due to a lack of knowledge about paternal/partner/co-parent mental health, fear of being judged, stigma or prioritizing the birthing parent’s experience as more important.441 Standardized scales may be used to aid detection of depression and anxiety. The EPDS, at cut-off scores ranging from 7–10, has reasonable sensitivity/specificity (0.87/0.71 for a cut-off score of 7 to 0.71/0.86 for a cut-off score of 10) for probable depression in fathers, so can be used to aid case identification (Level 3 ), although how well this would generalize to other co-parents is not known.442
In systematic reviews, no RCTs focused on the treatment of depression in fathers or co-parents were identified.443 Interventions were generally targeted at non-clinical populations and were psychoeducational in nature, focusing on paternal well-being indirectly (i.e., via the mother or the child or the couple’s relationship), with equivocal results. Systematic reviews on perinatal anxiety in fathers also evaluated mostly psychoeducational interventions in non-clinical populations.444,445 In some RCTs evaluating paternal anxiety, intervention groups had slightly lower levels of anxiety symptoms post-intervention than controls, but whether the interventions prevented the development of diagnosable PMADs is not known.444 Data on treatment for sexual and gender minority co-parents are limited but known barriers to uptake of care include a lack of targeted mental health services and supports (including peer support) specifically for—or sensitive to—the unique experiences of sexual and gender minority co-parents.438
Discussion and Summary
This CANMAT guideline aims to provide clinicians with comprehensive and systematic guidance for the management of PMADs. The development of recommendations was complex at times, largely due to the gaps in available literature regarding the efficacy and safety of various treatments in the perinatal population overall and in specific subpopulations. There are also a lack of head-to-head comparisons within and across various treatment modalities (e.g., comparison of therapy and medication, or between types of therapies and types of medications). It is our hope that this guideline serves to encourage more research in the area. There is a need for better evidence in particular for conditions where there is controversy, lower-certainty and for diagnoses where there is a lack of perinatal-specific evidence such as OCD, PTSD and BD, and to a lesser extent anxiety disorders. Although there has been an attempt to improve the diagnostic criteria of certain perinatal mental health conditions, such as the “peripartum onset” specifier of the DSM-5, we encourage better acknowledgement of the unique perinatal aspects of other conditions such as anxiety, OCD, psychosis and the limitations inherent in restricting PPD onset to the first 4 weeks after childbirth. There is also a clear need for more research in specific sub-populations, including gender-diverse, sexually diverse and racialized people, and fathers and co-parents. This dynamic literature requires constant update, and it is our intent that this first CANMAT PMAD guideline can form the basis for future updates and guidance over time. We also hope that the current work will serve as a model for the development of clinical guidelines that can address other mental and behavioural disorders that were outside the scope of this guideline (e.g., autism, attention-deficit-hyperactivity disorder, eating disorders, personality disorders, substance and alcohol use disorders and others).
Supplementary Materials
- sj-docx-1-cpa-10.1177_07067437241303031 – Supplemental material for Canadian Network for Mood and Anxiety Treatments 2024 Clinical Practice Guideline for the Management of Perinatal Mood, Anxiety, and Related Disorders: Guide de pratique 2024 du Canadian Network for Mood and Anxiety Treatments pour le traitement des troubles de l’humeur, des troubles anxieux et des troubles connexes périnatals (DOCX)
- sj-docx-2-cpa-10.1177_07067437241303031 – Supplemental material for Canadian Network for Mood and Anxiety Treatments 2024 Clinical Practice Guideline for the Management of Perinatal Mood, Anxiety, and Related Disorders: Guide de pratique 2024 du Canadian Network for Mood and Anxiety Treatments pour le traitement des troubles de l’humeur, des troubles anxieux et des troubles connexes périnatals (DOCX)
References
- Maternal Mental Health in Canada, 2018/2019 (Released: 24 June 2019).
- EJ Fawcett, N Fairbrother, ML Cox, IR White, JM Fawcett. The prevalence of anxiety disorders during pregnancy and the postpartum period.. J Clin Psychiatry., 2019. [DOI]
- LM Howard, E Molyneaux, CL Dennis, T Rochat, A Stein, J Milgrom. Non-psychotic mental disorders in the perinatal period.. Lancet., 2014. [DOI | PubMed]
- A Stein, RM Pearson, SH Goodman. Effects of perinatal mental disorders on the fetus and child.. Lancet., 2014. [DOI | PubMed]
- R Graham, M Mancher, D Miller Wolman, S Greenfield, E Steinberg. Clinical practice guidelines we can trust., 2011
- SH Kennedy, RW Lam, RS McIntyre. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: Section 3. Pharmacological treatments.. Can J Psychiatry., 2016. [DOI | PubMed]
- LN Yatham, SH Kennedy, SV Parikh. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder.. Bipolar Disord., 2018. [DOI | PubMed]
- LA Brotto, LAM Galea. Gender inclusivity in women’s health research.. BJOG., 2022. [DOI | PubMed]
- 9Partnership for Women’s Health Research Canada (PWHR) and Women’s Health Research Institute (WHRI). Beyond the Binary in Canada Guide. Pathways toward equitable research with people who experience sex and gender-based health inequities. Partnership for Women’s Health Research Canada, 2024. https://pwhr.org/wp-content/uploads/2024/12/BTB_PWHR_Oct_29_2024-1.pdf.
- BP Yawn, W Pace, PC Wollan. Concordance of Edinburgh postnatal depression scale (EPDS) and patient health questionnaire (PHQ-9) to assess increased risk of depression among postpartum women.. J Am Board Fam Med., 2009. [DOI | PubMed]
- RW Lam, SH Kennedy, C Adams. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2023 Update on clinical guidelines for management of major depressive disorder in adults.. Can J Psychiatry., 2024. [PubMed]
- G Guyatt, AD Oxman, EA Akl. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables.. J Clin Epidemiol., 2011. [DOI | PubMed]
- GH Guyatt, AD Oxman, GE Vist. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations.. Br Med J., 2008. [DOI | PubMed]
- NI Gavin, BN Gaynes, KN Lohr, S Meltzer-Brody, G Gartlehner, T Swinson. Perinatal depression: a systematic review of prevalence and incidence.. Obstet Gynecol., 2005. [DOI | PubMed]
- Y Bai, Q Li, KK Cheng. Prevalence of postpartum depression based on diagnostic interviews: a systematic review and meta-analysis.. Depress Anxiety., 2023. [DOI | PubMed]
- 16American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2022.
- CL Dennis, K Falah-Hassani, R Shiri. Prevalence of antenatal and postnatal anxiety: systematic review and meta-analysis.. Br J Psychiatry., 2017. [DOI | PubMed]
- MA O’Connell, P Leahy-Warren, AS Khashan, LC Kenny, SM O’Neill. Worldwide prevalence of tocophobia in pregnant women: systematic review and meta-analysis.. Acta Obstet Gynecol Scand., 2017. [DOI | PubMed]
- N Fairbrother, F Collardeau, AYK Albert. High prevalence and incidence of obsessive-compulsive disorder among women across pregnancy and the postpartum.. J Clin Psychiatry., 2021. [DOI]
- N Hudepohl, JV MacLean, LM Osborne. Perinatal obsessive-compulsive disorder: epidemiology, phenomenology, etiology, and treatment.. Curr Psychiatry Rep., 2022. [DOI | PubMed]
- EJ Russell, JM Fawcett, D Mazmanian. Risk of obsessive-compulsive disorder in pregnant and postpartum women: a meta-analysis.. J Clin Psychiatry., 2013. [DOI | PubMed]
- PD Yildiz, S Ayers, L Phillips. The prevalence of posttraumatic stress disorder in pregnancy and after birth: a systematic review and meta-analysis.. J Affect Disord., 2017. [DOI | PubMed]
- GA Masters, J Hugunin, L Xu. Prevalence of bipolar disorder in perinatal women: a systematic review and meta-analysis.. J Clin Psychiatry., 2022. [DOI]
- R Wesseloo, AM Kamperman, T Munk-Olsen, VJ Pop, SA Kushner, V Bergink. Risk of postpartum relapse in bipolar disorder and postpartum psychosis: a systematic review and meta-analysis.. Am J Psychiatry., 2016. [DOI | PubMed]
- R VanderKruik, M Barreix, D Chou. The global prevalence of postpartum psychosis: a systematic review.. BMC Psychiatry., 2017. [DOI | PubMed]
- T Munk-Olsen, I Jones, TM Laursen. Birth order and postpartum psychiatric disorders.. Bipolar Disord., 2014. [DOI | PubMed]
- A Perry, K Gordon-Smith, L Jones, I Jones. Phenomenology, epidemiology and aetiology of postpartum psychosis: a review.. Brain Sci., 2021. [DOI]
- K Al-Abri, D Edge, CJ Armitage. Correction to: prevalence and correlates of perinatal depression.. Soc Psychiatry Psychiatr Epidemiol., 2023. [DOI | PubMed]
- K Al-Abri, D Edge, CJ Armitage. Prevalence and correlates of perinatal depression.. Soc Psychiatry Psychiatr Epidemiol., 2023. [DOI | PubMed]
- M Azami, G Badfar, A Soleymani, S Rahmati. The association between gestational diabetes and postpartum depression: a systematic review and meta-analysis.. Diabetes Res Clin Pract., 2019. [DOI | PubMed]
- H Bayrampour, A Vinturache, E Hetherington, DL Lorenzetti, S Tough. Risk factors for antenatal anxiety: a systematic review of the literature.. J Reprod Infant Psychol., 2018. [DOI | PubMed]
- HK Brown, A Qazilbash, N Rahim, CL Dennis, SN Vigod. Chronic medical conditions and peripartum mental illness: a systematic review and meta-analysis.. Am J Epidemiol., 2018. [DOI | PubMed]
- T Fu, C Wang, J Yan, Q Zeng, C Ma. Relationship between antenatal sleep quality and depression in perinatal women: a comprehensive meta-analysis of observational studies.. J Affect Disord., 2023. [DOI | PubMed]
- M Maghami, SP Shariatpanahi, D Habibi. Sleep disorders during pregnancy and postpartum depression: a systematic review and meta-analysis.. Int J Dev Neurosci., 2021. [DOI | PubMed]
- J Michalczyk, A Milosz, E Soroka. Postpartum psychosis: a review of risk factors, clinical picture, management, prevention, and psychosocial determinants.. Med Sci Monit., 2023. [DOI]
- D Pereira, AR Pessoa, N Madeira, A Macedo, AT Pereira. Association between premenstrual dysphoric disorder and perinatal depression: a systematic review.. Arch Womens Ment Health., 2022. [DOI | PubMed]
- TJ Reilly, E Roberts, VC Sagnay De La Bastida, P McGuire, P Dazzan, AE Cullen. Systematic review of the association between adverse life events and the onset and relapse of postpartum psychosis.. Front Psychiatry., 2023. [DOI | PubMed]
- MM Zacher Kjeldsen, A Bricca, X Liu, VG Frokjaer, KB Madsen, T Munk-Olsen. Family history of psychiatric disorders as a risk factor for maternal postpartum depression: a systematic review and meta-analysis.. JAMA Psychiatry., 2022. [DOI | PubMed]
- A Biaggi, S Conroy, S Pawlby, CM Pariante. Identifying the women at risk of antenatal anxiety and depression: a systematic review.. J Affect Disord., 2016. [DOI | PubMed]
- K Falah-Hassani, R Shiri, S Vigod, CL Dennis. Prevalence of postpartum depression among immigrant women: a systematic review and meta-analysis.. J Psychiatr Res., 2015. [DOI | PubMed]
- A Roddy Mitchell, H Gordon, J Atkinson. Prevalence of perinatal anxiety and related disorders in low- and middle-income countries: a systematic review and meta-analysis.. JAMA Netw Open., 2023. [DOI]
- S Shorey, CYI Chee, ED Ng, YH Chan, WWS Tam, YS Chong. Prevalence and incidence of postpartum depression among healthy mothers: a systematic review and meta-analysis.. J Psychiatr Res., 2018. [DOI | PubMed]
- S Mukherjee, MJ Trepka, D Pierre-Victor, R Bahelah, T Avent. Racial/ethnic disparities in antenatal depression in the United States: a systematic review.. Matern Child Health J., 2016. [DOI | PubMed]
- KA Black, I MacDonald, T Chambers, MB Ospina. Postpartum mental health disorders in indigenous women: a systematic review and meta-analysis.. J Obstet Gynaecol Can., 2019. [DOI | PubMed]
- S Owais, M Faltyn, AVD Johnson. The perinatal mental health of indigenous women: a systematic review and meta-analysis.. Can J Psychiatry., 2020. [DOI | PubMed]
- HK Brown, SN Vigod, K Fung. Perinatal mental illness among women with disabilities: a population-based cohort study.. Soc Psychiatry Psychiatr Epidemiol., 2022. [DOI | PubMed]
- M Mitra, LI Iezzoni, J Zhang, LM Long-Bellil, SC Smeltzer, BA Barton. Prevalence and risk factors for postpartum depression symptoms among women with disabilities.. Matern Child Health J., 2015. [DOI | PubMed]
- A Kirubarajan, LC Barker, S Leung. LGBTQ2S+ childbearing individuals and perinatal mental health: a systematic review.. BJOG., 2022. [DOI | PubMed]
- M Nielsen-Scott, G Fellmeth, C Opondo, F Alderdice. Prevalence of perinatal anxiety in low- and middle-income countries: a systematic review and meta-analysis.. J Affect Disord., 2022. [DOI | PubMed]
- N Fairbrother, A Albert, F Collardeau, C Keeney. The Childbirth Fear Questionnaire and the Wijma Delivery Expectancy Questionnaire as screening tools for specific phobia, fear of childbirth.. Int J Environ Res Public Health., 2022. [DOI]
- S Sanjari, R Chaman, S Salehin, S Goli, A Keramat. Update on the global prevalence of severe fear of childbirth in low-risk pregnant women: a systematic review and meta-analysis.. Int J Women Health Rep Sci., 2022
- LS Cohen, LL Altshuler, BL Harlow. Relapse of major depression during pregnancy in women who maintain or discontinue antidepressant treatment.. JAMA., 2006. [DOI | PubMed]
- H Khalifeh, IM Hunt, L Appleby, LM Howard. Suicide in perinatal and non-perinatal women in contact with psychiatric services: 15 year findings from a UK national inquiry.. Lancet Psychiatry., 2016. [DOI | PubMed]
- L Bonari, N Pinto, E Ahn, A Einarson, M Steiner, G Koren. Perinatal risks of untreated depression during pregnancy.. Can J Psychiatry., 2004. [DOI | PubMed]
- D Wang, YL Li, D Qiu, SY Xiao. Factors influencing paternal postpartum depression: a systematic review and meta-analysis.. J Affect Disord., 2021. [DOI | PubMed]
- M Shay, AL MacKinnon, A Metcalfe. Depressed mood and anxiety as risk factors for hypertensive disorders of pregnancy: a systematic review and meta-analysis.. Psychol Med., 2020. [DOI | PubMed]
- S Grigoriadis, EH VonderPorten, L Mamisashvili. The impact of maternal depression during pregnancy on perinatal outcomes: a systematic review and meta-analysis.. J Clin Psychiatry., 2013. [DOI]
- A Jarde, M Morais, D Kingston. Neonatal outcomes in women with untreated antenatal depression compared with women without depression: a systematic review and meta-analysis.. JAMA Psychiatry., 2016. [DOI | PubMed]
- S Gentile. Untreated depression during pregnancy: short- and long-term effects in offspring. A systematic review.. Neuroscience., 2017. [DOI | PubMed]
- S Kim, M Park, S Ahn. The impact of antepartum depression and postpartum depression on exclusive breastfeeding: a systematic review and meta-analysis.. Clin Nurs Res., 2022. [DOI | PubMed]
- RH Bind, A Biaggi, A Bairead. Mother-infant interaction in women with depression in pregnancy and in women with a history of depression: the psychiatry research and motherhood – depression (PRAM-D) study.. BJPsych Open., 2021. [DOI]
- AF Dadi, ER Miller, L Mwanri. Postnatal depression and its association with adverse infant health outcomes in low- and middle-income countries: a systematic review and meta-analysis.. BMC Pregnancy Childbirth., 2020. [DOI | PubMed]
- N Jacques, CL de Mola, G Joseph. Prenatal and postnatal maternal depression and infant hospitalization and mortality in the first year of life: a systematic review and meta-analysis.. J Affect Disord., 2019. [DOI | PubMed]
- S Ayers, R Bond, R Webb, P Miller, K Bateson. Perinatal mental health and risk of child maltreatment: a systematic review and meta-analysis.. Child Abuse Negl., 2019. [DOI | PubMed]
- H Baird, RA Harris, HP Santos. The effects of maternal perinatal depression on child IQ: a systematic review.. Matern Child Health J., 2023. [DOI | PubMed]
- A Rogers, S Obst, SJ Teague. Association between maternal perinatal depression and anxiety and child and adolescent development: a meta-analysis.. JAMA Pediatr., 2020. [DOI | PubMed]
- S Grigoriadis, L Graves, M Peer. Maternal anxiety during pregnancy and the association with adverse perinatal outcomes: systematic review and meta-analysis.. J Clin Psychiatry., 2018. [DOI]
- S Grigoriadis, L Graves, M Peer. A systematic review and meta-analysis of the effects of antenatal anxiety on postpartum outcomes.. Arch Womens Ment Health., 2019. [DOI | PubMed]
- M Khajehei, JA Swain, R Li. Fear of childbirth in nulliparous and multiparous women in Australia.. Br J Midwifery., 2023. [DOI]
- M Laursen, C Johansen, M Hedegaard. Fear of childbirth and risk for birth complications in nulliparous women in the Danish national birth cohort.. BJOG., 2009. [DOI | PubMed]
- MA Mohamed, A Elhelbawy, M Khalid, LA AbdAllatif, HE Lialy. Effects of bipolar disorder on maternal and fetal health during pregnancy: a systematic review.. BMC Pregnancy Childbirth., 2023. [DOI | PubMed]
- M Rusner, M Berg, C Begley. Bipolar disorder in pregnancy and childbirth: a systematic review of outcomes.. BMC Pregnancy Childbirth., 2016. [DOI | PubMed]
- J Sachdeva, S Nagle Yang, P Gopalan. Trauma informed care in the obstetric setting and role of the perinatal psychiatrist: a comprehensive review of the literature.. J Acad Consult Liaison Psychiatry., 2022. [DOI | PubMed]
- TIP 57 trauma-informed care in behavioural health services,. 2015
- MW O’Hara, JE McCabe. Postpartum depression: current status and future directions.. Annu Rev Clin Psychol., 2013. [DOI | PubMed]
- V Sharma, P Singh, C Baczynski, M Khan. A closer look at the nosological status of the highs (hypomanic symptoms) in the postpartum period.. Arch Womens Ment Health., 2021. [DOI | PubMed]
- JH Goodman. Postpartum depression beyond the early postpartum period.. J Obstet Gynecol Neonatal Nurs., 2004. [DOI]
- C Goldfinger, SM Green, M Furtado, RE McCabe. Characterizing the nature of worry in a sample of perinatal women with generalized anxiety disorder.. Clin Psychol Psychother., 2020. [DOI | PubMed]
- F Thiel, T Ein-Dor, G Dishy, A King, S Dekel. Examining symptom clusters of childbirth-related posttraumatic stress disorder.. Prim Care Companion CNS Disord., 2018. [DOI]
- Care pathway for the management of perinatal mental health.. 2024
- A Johnson, E Stevenson, L Moeller, J McMillian-Bohler. Systematic screening for perinatal mood and anxiety disorders to promote onsite mental health consultations: a quality improvement report.. J Midwifery Womens Health., 2021. [DOI | PubMed]
- J Cox. Thirty years with the Edinburgh postnatal depression scale: voices from the past and recommendations for the future.. Br J Psychiatry., 2019. [DOI | PubMed]
- SH Park, JI Kim. Predictive validity of the Edinburgh postnatal depression scale and other tools for screening depression in pregnant and postpartum women: a systematic review and meta-analysis.. Arch Gynecol Obstet., 2023. [DOI | PubMed]
- B Levis, Z Negeri, Y Sun, A Benedetti, BD Thombs. Accuracy of the Edinburgh postnatal depression scale (EPDS) for screening to detect major depression among pregnant and postpartum women: systematic review and meta-analysis of individual participant data.. Br Med J., 2020. [DOI]
- BD Thombs, B Levis, A Lyubenova. Overestimation of postpartum depression prevalence based on a 5-item version of the EPDS: systematic review and individual participant data meta-analysis.. Can J Psychiatry., 2020. [DOI | PubMed]
- RE Gliklich, MB Leavy, L Cosgrove. Harmonized outcome measures for use in depression patient registries and clinical practice.. Ann Intern Med., 2020. [DOI | PubMed]
- L Wang, K Kroenke, TE Stump, PO Monahan. Screening for perinatal depression with the patient health questionnaire depression scale (PHQ-9): a systematic review and meta-analysis.. Gen Hosp Psychiatry., 2021. [DOI | PubMed]
- RD Smith, JSY Shing, J Lin, K Bosanquet, DYT Fong, KYW Lok. Meta-analysis of diagnostic properties of the Whooley questions to identify depression in perinatal women.. J Affect Disord., 2022. [DOI | PubMed]
- S Ayers, R Coates, A Sinesi. Assessment of perinatal anxiety: diagnostic accuracy of five measures.. Br J Psychiatry., 2024. [DOI | PubMed]
- M Barkham, B Bewick, T Mullin. The CORE-10: a short measure of psychological distress for routine use in the psychological therapies.. Counsel Psychother Res., 2013. [DOI]
- R Coates, S Ayers, R de Visser, A Thornton. Evaluation of the CORE-10 to assess psychological distress in pregnancy.. J Reprod Infant Psychol., 2020. [DOI | PubMed]
- USPST Force, MJ Barry, WK Nicholson. Screening for anxiety disorders in adults: US preventive services task force recommendation statement.. JAMA., 2023. [DOI | PubMed]
- A Sinesi, H Cheyne, M Maxwell, R O’Carroll. The Stirling antenatal anxiety scale (SAAS): development and initial psychometric validation.. J Affect Disord Rep., 2022. [DOI]
- A Sinesi, M Maxwell, R O’Carroll, H Cheyne. Anxiety scales used in pregnancy: systematic review.. BJPsych Open., 2019. [DOI]
- N Fairbrother, A Albert, C Keeney, D Tchir, RB Cameron. Screening for perinatal OCD: a comparison of the DOCS and the EPDS.. Assessment., 2023. [DOI | PubMed]
- ME Williams, DM Strobino, CN Holliday. Measuring post-traumatic stress after childbirth: a review and critical appraisal of instruments.. J Reprod Infant Psychol., 2023. [DOI | PubMed]
- CT Clark, DK Sit, K Driscoll. Does screening with the MDQ and EPDS improve identification of bipolar disorder in an obstetrical sample?. Depress Anxiety., 2015. [DOI | PubMed]
- BN Frey, W Simpson, L Wright, M Steiner. Sensitivity and specificity of the mood disorder questionnaire as a screening tool for bipolar disorder during pregnancy and the postpartum period.. J Clin Psychiatry., 2012. [DOI | PubMed]
- L Merrill, L Mittal, J Nicoloro, C Caiozzo, PK Maciejewski, LJ Miller. Screening for bipolar disorder during pregnancy.. Arch Womens Ment Health., 2015. [DOI | PubMed]
- V Sharma, M Khan, C Corpse, P Sharma. Missed bipolarity and psychiatric comorbidity in women with postpartum depression.. Bipolar Disord., 2008. [DOI | PubMed]
- CA Chessick, S Dimidjian. Screening for bipolar disorder during pregnancy and the postpartum period.. Arch Womens Ment Health., 2010. [DOI | PubMed]
- MJ Barry, WK Nicholson. Screening for depression and suicide risk in adults: US preventive services task force recommendation statement.. JAMA., 2023. [DOI | PubMed]
- SS Leung, C Leung, TH Lam. Outcome of a postnatal depression screening programme using the Edinburgh postnatal depression scale: a randomized controlled trial.. J Public Health (Oxf)., 2011. [DOI | PubMed]
- S El-Den, L Pham, I Anderson. Perinatal depression screening: a systematic review of recommendations from member countries of the organisation for economic co-operation and development (OECD).. Arch Womens Ment Health., 2022. [DOI | PubMed]
- E Lang, H Colquhoun, JC LeBlanc. Recommendation on instrument-based screening for depression during pregnancy and the postpartum period.. CMAJ., 2022. [DOI]
- D Kingston, S McDonald, S Tough, MP Austin, K Hegadoren, G Lasiuk. Public views of acceptability of perinatal mental health screening and treatment preference: a population based survey.. BMC Pregnancy Childbirth., 2014. [DOI | PubMed]
- LJ Puryear, YH Nong, NP Correa, K Cox, CS Greeley. Outcomes of implementing routine screening and referrals for perinatal mood disorders in an integrated multi-site pediatric and obstetric setting.. Matern Child Health J., 2019. [DOI | PubMed]
- Screening and diagnosis of mental health conditions during pregnancy and postpartum: ACOG clinical practice guideline No. 4.. Obstet Gynecol., 2023. [DOI | PubMed]
- S Jia, M Zhi, J Liu, J Liu, L Jiang, Y Zhu. Evidence summary on the timing and frequency of screening for perinatal depression.. Ann Palliat Med., 2021. [DOI | PubMed]
- U Halbreich, S Karkun. Cross-cultural and social diversity of prevalence of postpartum depression and depressive symptoms.. J Affect Disord., 2006. [DOI | PubMed]
- M Skoog, IK Hallstrom, A Vilhelmsson. Health care professionals’ experiences of screening immigrant mothers for postpartum depression-a qualitative systematic review.. PLoS One., 2022. [DOI]
- SD Shrestha, R Pradhan, TD Tran, RC Gualano, JR Fisher. Reliability and validity of the Edinburgh postnatal depression scale (EPDS) for detecting perinatal common mental disorders (PCMDs) among women in low-and lower-middle-income countries: a systematic review.. BMC Pregnancy Childbirth., 2016. [DOI | PubMed]
- A Salehi, M Zhang, N Kithulegoda, S Vigod, N Ivers. Validation of the culturally adapted Edinburgh postpartum depression scale among east Asian, southeast Asian and south Asian populations: a scoping review.. Int J Ment Health Nurs., 2023. [DOI | PubMed]
- AW Chan, C Reid, P Skeffington, R Marriott. A systematic review of EPDS cultural suitability with indigenous mothers: a global perspective.. Arch Womens Ment Health., 2021. [DOI | PubMed]
- F Rahmani Ivary, S Fanaei, S Ghahremani. A systematic review of psychometric properties of the Edinburgh postnatal depression scale in Iranian population.. Int J Pediatr., 2019. [DOI]
- KB Kozhimannil, CM Trinacty, AB Busch, HA Huskamp, AS Adams. Racial and ethnic disparities in postpartum depression care among low-income women.. Psychiatr Serv., 2011. [DOI | PubMed]
- M Sambrook Smith, V Lawrence, E Sadler, A Easter. Barriers to accessing mental health services for women with perinatal mental illness: systematic review and meta-synthesis of qualitative studies in the UK.. BMJ Open., 2019. [DOI]
- V Schmied, E Black, N Naidoo, HG Dahlen, P Liamputtong. Migrant women’s experiences, meanings and ways of dealing with postnatal depression: a meta-ethnographic study.. PLoS One., 2017. [DOI]
- R Webb, N Uddin, E Ford. Barriers and facilitators to implementing perinatal mental health care in health and social care settings: a systematic review.. Lancet Psychiatry., 2021. [DOI | PubMed]
- LA Tarasoff, Y Lunsky, K Welsh. Unmet needs, limited access: a qualitative study of postpartum health care experiences of people with disabilities.. J Adv Nurs., 2023. [DOI | PubMed]
- K Gaskin, H James. Using the Edinburgh postnatal depression scale with learning disabled mothers.. Community Pract., 2006. [PubMed]
- A Jones. Help seeking in the perinatal period: a review of barriers and facilitators.. Soc Work Public Health., 2019. [DOI | PubMed]
- M Bacciaglia, HT Neufeld, E Neiterman, A Krishnan, S Johnston, K Wright. Indigenous maternal health and health services within Canada: a scoping review.. BMC Pregnancy Childbirth., 2023. [DOI | PubMed]
- M Greenfield, Z Darwin. Trans and non-binary pregnancy, traumatic birth, and perinatal mental health: a scoping review.. Int J Transgend Health., 2021. [DOI | PubMed]
- CL Tobin, P Di Napoli, CT Beck. Refugee and immigrant women’s experience of postpartum depression: a meta-synthesis.. J Transcult Nurs., 2018. [DOI | PubMed]
- A Wittkowski, S Patel, JR Fox. The experience of postnatal depression in immigrant mothers living in western countries: a meta-synthesis.. Clin Psychol Psychother., 2017. [DOI | PubMed]
- FM Anderson, SL Hatch, C Comacchio, LM Howard. Prevalence and risk of mental disorders in the perinatal period among migrant women: a systematic review and meta-analysis.. Arch Womens Ment Health., 2017. [DOI | PubMed]
- R Webb, N Uddin, G Constantinou. Meta-review of the barriers and facilitators to women accessing perinatal mental healthcare.. BMJ Open., 2023. [DOI]
- DE Jidong, N Husain, A Roche. Psychological interventions for maternal depression among women of African and Caribbean origin: a systematic review.. BMC Womens Health., 2021. [DOI | PubMed]
- C Ponting, NE Mahrer, H Zelcer, C Dunkel Schetter, DA Chavira. Psychological interventions for depression and anxiety in pregnant Latina and black women in the United States: a systematic review.. Clin Psychol Psychother., 2020. [DOI | PubMed]
- S Gilbody, P Bower, J Fletcher, D Richards, AJ Sutton. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes.. Arch Intern Med., 2006. [DOI | PubMed]
- N Byatt, W Xu, LL Levin, TA Moore Simas. Perinatal depression care pathway for obstetric settings.. Int Rev Psychiatry., 2019. [DOI | PubMed]
- CK Klatter, LM van Ravesteyn, J Stekelenburg. Is collaborative care a key component for treating pregnant women with psychiatric symptoms (and additional psychosocial problems)? A systematic review.. Arch Womens Ment Health., 2022. [DOI | PubMed]
- MC Prom, A Denduluri, LL Philpotts. A systematic review of interventions that integrate perinatal mental health care into routine maternal care in low- and middle-income countries.. Front Psychiatry., 2022. [DOI | PubMed]
- IJ Saldanha, GP Adam, G Kanaan. Delivery strategies for postpartum care: a systematic review and meta-analysis.. Obstet Gynecol., 2023. [DOI | PubMed]
- N Byatt, J Straus, A Stopa, K Biebel, L Mittal, TA Moore Simas. Massachusetts child psychiatry access program for moms: utilization and quality assessment.. Obstet Gynecol., 2018. [DOI | PubMed]
- A Rivero-Santana, L Perestelo-Perez, Y Alvarez-Perez. Stepped care for the treatment of depression: a systematic review and meta-analysis.. J Affect Disord., 2021. [DOI | PubMed]
- The perinatal mental health care pathways. National Health Service (NHS) England. Published May,. 2018
- V Sawrikar, E Stewart, HM LaMonica. Using staged care to provide “right care first time” to people with common affective disorders.. Psychiatr Serv., 2021. [DOI | PubMed]
- J Delgadillo, S Ali, K Fleck. Stratified care vs stepped care for depression: a cluster randomized clinical trial.. JAMA Psychiatry., 2022. [DOI | PubMed]
- CB DiSanza, PA Geller, A Bonacquisti, B Posmontier, JA Horowitz, LA Chiarello. A stepped care model of patient navigation to enhance engagement with perinatal mental health care.. Health Equity., 2020. [DOI | PubMed]
- P Kukreti, R Ransing, P Raghuveer. Stepped care model for developing pathways of screening, referral, and brief intervention for depression in pregnancy: a mixed-method study from development phase.. Indian J Soc Psychiatry., 2022. [PubMed]
- SS Olin, M McCord, REK Stein. Beyond screening: a stepped care pathway for managing postpartum depression in pediatric settings.. J Womens Health (Larchmt)., 2017. [DOI | PubMed]
- Care pathway for the management of perinatal mental health and addictions, July 2021.
- MT Ashford, EK Olander, S Ayers. Computer- or web-based interventions for perinatal mental health: a systematic review.. J Affect Disord., 2016. [DOI | PubMed]
- YI Nillni, A Mehralizade, L Mayer, S Milanovic. Treatment of depression, anxiety, and trauma-related disorders during the perinatal period: a systematic review.. Clin Psychol Rev., 2018. [DOI | PubMed]
- C Meredith, C McKerchar, C Lacey. Indigenous approaches to perinatal mental health: a systematic review with critical interpretive synthesis.. Arch Womens Ment Health., 2023. [DOI | PubMed]
- K Robertson, R Wells. Black with ‘baby blues’: a systematic scoping review of programs to address postpartum depression in African American women.. Matern Child Health J., 2023. [DOI | PubMed]
- 149World Health Organization. World mental health report: transforming mental health for all. Geneva; 2022. Accessed 7 August 2024. https://www.who.int/publications/i/item/9789240049338
- K Clarke, M King, A Prost. Psychosocial interventions for perinatal common mental disorders delivered by providers who are not mental health specialists in low- and middle-income countries: a systematic review and meta-analysis.. PLoS Med., 2013. [DOI]
- A Rahman, J Fisher, P Bower. Interventions for common perinatal mental disorders in women in low- and middle-income countries: a systematic review and meta-analysis.. Bull World Health Organ., 2013. [DOI | PubMed]
- E Prina, C Ceccarelli, JO Abdulmalik. Task-sharing psychosocial interventions for the prevention of common mental disorders in the perinatal period in low- and middle-income countries: a systematic review and meta-analysis.. Int J Soc Psychiatry., 2023. [DOI | PubMed]
- DR Singla, A Lawson, BA Kohrt. Implementation and effectiveness of nonspecialist-delivered interventions for perinatal mental health in high-income countries: a systematic review and meta-analysis.. JAMA Psychiatry., 2021. [DOI | PubMed]
- TH Wang, LW Pai, YL Tzeng, TP Yeh, YK Teng. Effectiveness of nurses and midwives-led psychological interventions on reducing depression symptoms in the perinatal period: a systematic review and meta-analysis.. Nurs Open., 2021. [DOI | PubMed]
- H Bayrampour, J Trieu, T Tharmaratnam. Effectiveness of ehealth interventions to reduce perinatal anxiety: a systematic review and meta-analysis.. J Clin Psychiatry., 2019. [DOI]
- SM Canfield, KE Canada. Systematic review of online interventions to reduce perinatal mood and anxiety disorders in underserved populations.. J Perinat Neonatal Nurs., 2023. [DOI | PubMed]
- C Chen, X Wang, H Xu, Y Li. Effectiveness of digital psychological interventions in reducing perinatal depression: a systematic review of meta-analyses.. Arch Womens Ment Health., 2023. [DOI | PubMed]
- H Ching, JYX Chua, JS Chua, S Shorey. The effectiveness of technology-based cognitive behavioral therapy on perinatal depression and anxiety: a systematic review and meta-analysis.. Worldviews Evid Based Nurs., 2023. [DOI | PubMed]
- J Dol, B Richardson, GT Murphy, M Aston, D McMillan, M Campbell-Yeo. Impact of mobile health interventions during the perinatal period on maternal psychosocial outcomes: a systematic review.. JBI Evid Synth., 2020. [DOI | PubMed]
- K Evans, S Rennick-Egglestone, S Cox, Y Kuipers, H Spiby. Remotely delivered interventions to support women with symptoms of anxiety in pregnancy: mixed methods systematic review and meta-analysis.. J Med Internet Res., 2022. [DOI]
- N Hanach, N de Vries, H Radwan, N Bissani. The effectiveness of telemedicine interventions, delivered exclusively during the postnatal period, on postpartum depression in mothers without history or existing mental disorders: a systematic review and meta-analysis.. Midwifery., 2021. [DOI | PubMed]
- AK Lewkowitz, AR Whelan, NK Ayala. The effect of digital health interventions on postpartum depression or anxiety: a systematic review and meta-analysis of randomized controlled trials.. Am J Obstet Gynecol., 2024. [DOI | PubMed]
- X Liu, S Huang, Y Hu, G Wang. The effectiveness of telemedicine interventions on women with postpartum depression: a systematic review and meta-analysis.. Worldviews Evid Based Nurs., 2022. [DOI | PubMed]
- KA Silang, PR Sohal, KS Bright. Ehealth interventions for treatment and prevention of depression, anxiety, and insomnia during pregnancy: systematic review and meta-analysis.. JMIR Ment Health., 2022. [DOI]
- L Zhao, J Chen, L Lan. Effectiveness of telehealth interventions for women with postpartum depression: systematic review and meta-analysis.. JMIR Mhealth Uhealth., 2021. [DOI]
- CL Battle, MM Howard. A mother-baby psychiatric day hospital: history, rationale, and why perinatal mental health is important for obstetric medicine.. Obstet Med., 2014. [DOI | PubMed]
- K Connellan, C Bartholomaeus, C Due, DW Riggs. A systematic review of research on psychiatric mother-baby units.. Arch Womens Ment Health., 2017. [DOI | PubMed]
- PA Geller, B Posmontier, JA Horowitz, A Bonacquisti, LA Chiarello. Introducing mother baby connections: a model of intensive perinatal mental health outpatient programming.. J Behav Med., 2018. [DOI | PubMed]
- R Gillham, A Wittkowski. Outcomes for women admitted to a mother and baby unit: a systematic review.. Int J Womens Health., 2015. [DOI | PubMed]
- WQ Xue, KK Cheng, D Xu, X Jin, WJ Gong. Uptake of referrals for women with positive perinatal depression screening results and the effectiveness of interventions to increase uptake: a systematic review and meta-analysis.. Epidemiol Psychiatr Sci., 2020. [DOI]
- SA Harris, M Harrison, K Hazell-Raine, C Wade, V Eapen, J Kohlhoff. Patient navigation models for mental health of parents expecting or caring for an infant or young child: a systematic review.. Infant Ment Health J., 2023. [DOI | PubMed]
- S-y Mai, Y Liu, H-c He, X-y Yan. The effects of different exercise programs on the prevention of perinatal depression: a systematic review and meta-analysis.. CEOG., 2023. [DOI]
- M Yuan, H Chen, D Chen. Effect of physical activity on prevention of postpartum depression: a dose-response meta-analysis of 186,412 women.. Front Psychiatry., 2022. [DOI | PubMed]
- T Baskar, MN Mahajan, J Gaikwad. Interventions for postpartum depression: systematic review.. NeuroQuantology., 2022. [DOI]
- ID Morres, NA Tzouma, A Hatzigeorgiadis. Exercise for perinatal depressive symptoms: a systematic review and meta-analysis of randomized controlled trials in perinatal health services.. J Affect Disord., 2022. [DOI | PubMed]
- V Pentland, S Spilsbury, A Biswas, MF Mottola, S Paplinskie, MS Mitchell. Does walking reduce postpartum depressive symptoms? A systematic review and meta-analysis of randomized controlled trials.. J Women Health., 2022. [DOI]
- M Saligheh, D Hackett, P Boyce, S Cobley. Can exercise or physical activity help improve postnatal depression and weight loss? A systematic review.. Arch Womens Ment Health., 2017. [DOI | PubMed]
- J Wang, C Carru, S Sedda, PL Fiori, Z Li, Z Chen. Comparative impact of exercise-based interventions for postpartum depression: a Bayesian network meta-analysis.. Int J Gynaecol Obstet., 2024. [DOI | PubMed]
- X Liu, G Wang, Y Cao. Physical exercise interventions for perinatal depression symptoms in women: a systematic review and meta-analysis.. Front Psychol., 2022. [DOI | PubMed]
- M Sanchez-Polan, C Silva-Jose, E Franco. Prenatal anxiety and exercise. Systematic review and meta-analysis.. J Clin Med., 2021. [DOI]
- LP Hunter, JD Rychnovsky, SM Yount. A selective review of maternal sleep characteristics in the postpartum period.. J Obstet Gynecol Neonatal Nurs., 2009. [DOI]
- Q Lu, X Zhang, Y Wang. Sleep disturbances during pregnancy and adverse maternal and fetal outcomes: a systematic review and meta-analysis.. Sleep Med Rev., 2021. [DOI | PubMed]
- JA Mindell, RA Cook, J Nikolovski. Sleep patterns and sleep disturbances across pregnancy.. Sleep Med., 2015. [DOI | PubMed]
- HE Montgomery-Downs, R Stremler, SP Insana. Postpartum sleep in new mothers and fathers.. Open Sleep J., 2013. [DOI]
- J Park, SY Kim, K Lee. Effectiveness of behavioral sleep interventions on children’s and mothers’ sleep quality and maternal depression: a systematic review and meta-analysis.. Sci Rep., 2022. [DOI | PubMed]
- N Sasaki, N Yasuma, E Obikane. Psycho-educational interventions focused on maternal or infant sleep for pregnant women to prevent the onset of antenatal and postnatal depression: a systematic review.. Neuropsychopharmacol Rep., 2021. [DOI | PubMed]
- B Bei, J Milgrom, J Ericksen, J Trinder. Subjective perception of sleep, but not its objective quality, is associated with immediate postpartum mood disturbances in healthy women.. Sleep., 2010. [DOI | PubMed]
- A Lawson, KE Murphy, E Sloan, E Uleryk, A Dalfen. The relationship between sleep and postpartum mental disorders: a systematic review.. J Affect Disord., 2015. [DOI | PubMed]
- LM Swanson, H Flynn, JD Adams-Mundy, R Armitage, JT Arnedt. An open pilot of cognitive-behavioral therapy for insomnia in women with postpartum depression.. Behav Sleep Med., 2013. [DOI | PubMed]
- X Zheng, Z Zhu, J Chen. Efficacy of cognitive behavioural therapy for insomnia or sleep disturbance in pregnant women: a systematic review ad meta-analysis.. J Sleep Res., 2023. [DOI]
- X Li, L Fang, L Guan, J Zhang, M Zheng, D Zhu. The effects of light therapy on depression and sleep in women during pregnancy or the postpartum period: a systematic review and meta-analysis.. Brain Behav., 2023. [DOI]
- RS Opie, AC Uldrich, K Ball. Maternal postpartum diet and postpartum depression: a systematic review.. Matern Child Health J., 2020. [DOI | PubMed]
- A Bedaso, J Adams, W Peng, D Sibbritt. The relationship between social support and mental health problems during pregnancy: a systematic review and meta-analysis.. Reprod Health., 2021. [DOI | PubMed]
- R Huang, C Yan, Y Tian. Effectiveness of peer support intervention on perinatal depression: a systematic review and meta-analysis.. J Affect Disord., 2020. [DOI | PubMed]
- K Yang, J Wu, X Chen. Risk factors of perinatal depression in women: a systematic review and meta-analysis.. BMC Psychiatry., 2022. [DOI | PubMed]
- CL Dennis. Psychosocial and psychological interventions for prevention of postnatal depression: systematic review.. Br Med J., 2005. [DOI | PubMed]
- CL Dennis. Psychosocial interventions for the treatment of perinatal depression.. Best Pract Res Clin Obstet Gynaecol., 2014. [DOI | PubMed]
- NJ Highet. Mental health care in the perinatal period: Australian Clinical Practice Guideline., 2023
- Q Fang, L Lin, Q Chen. Effect of peer support intervention on perinatal depression: a meta-analysis.. Gen Hosp Psychiatry., 2022. [DOI | PubMed]
- E O’Connor, CA Senger, ML Henninger, E Coppola, BN Gaynes. Interventions to prevent perinatal depression: evidence report and systematic review for the US preventive services task force.. JAMA., 2019. [DOI | PubMed]
- R Carter, F Cust, E Boath. Effectiveness of a peer support intervention for antenatal depression: a feasibility study.. J Reprod Infant Psychol., 2020. [DOI | PubMed]
- S Shorey, CYI Chee, ED Ng, Y Lau, CL Dennis, YH Chan. Evaluation of a technology-based peer-support intervention program for preventing postnatal depression (part 1): randomized controlled trial.. J Med Internet Res., 2019. [DOI]
- F Sufredini, C Catling, J Zugai, S Chang. The effects of social support on depression and anxiety in the perinatal period: a mixed-methods systematic review.. J Affect Disord., 2022. [DOI | PubMed]
- M Buultjens, A Farouque, L Karimi, L Whitby, J Milgrom, B Erbas. The contribution of group prenatal care to maternal psychological health outcomes: a systematic review.. Women Birth., 2021. [DOI]
- USPST Force, SJ Curry, AH Krist. Interventions to prevent perinatal depression: US preventive services task force recommendation statement.. JAMA., 2019. [DOI | PubMed]
- L He, KL Soh, J Yu, A Chen, X Dong. Effect of family-centered interventions for perinatal depression: an overview of systematic reviews.. Front Psychiatry., 2023. [DOI | PubMed]
- K Hong, H Hwang, H Han. Perspectives on antenatal education associated with pregnancy outcomes: systematic review and meta-analysis.. Women Birth., 2021. [DOI | PubMed]
- H Iwata, E Mori, K Maehara, N Harada, A Saito. Effectiveness of parenting education for expectant primiparous women in Asia: a systematic review.. JBI Evid Synth., 2021. [DOI | PubMed]
- G Laccetta, M Di Chiara, MC De Nardo, G Terrin. Symptoms of post-traumatic stress disorder in parents of preterm newborns: a systematic review of interventions and prevention strategies.. Front Psychiatry., 2023. [DOI | PubMed]
- S Park, J Kim, J Oh, S Ahn. Effects of psychoeducation on the mental health and relationships of pregnant couples: a systemic review and meta-analysis.. Int J Nurs Stud., 2020. [DOI | PubMed]
- NR Stevens, ML Miller, AK Puetz, AC Padin, N Adams, DJ Meyer. Psychological intervention and treatment for posttraumatic stress disorder during pregnancy: a systematic review and call to action.. J Trauma Stress., 2021. [DOI | PubMed]
- X Xiao, AY Loke. The effects of co-parenting/intergenerational co-parenting interventions during the postpartum period: a systematic review.. Int J Nurs Stud., 2021. [DOI | PubMed]
- 213National Institute for Health and Care Excellence (NICE). Antenatal and postnatal mental health: clinical management and service guidance (CG192). UK: National Institute for Health and Care Excellence; Published December 2014, updated February 2020. Accessed 12 December 2024. https://www.nice.org.uk/guidance/cg192/resources/antenatal-and-postnatal-mental-health-clinical-management-and-service-guidance-pdf-35109869806789
- R Leonard, M Linden, A Grant. Effectiveness of family-focused home visiting for maternal mental illness: a systematic review and meta-analysis.. J Psychiatr Ment Health Nurs., 2021. [DOI | PubMed]
- JE McCabe, B Wickberg, J Deberg, RC Davila, LS Segre. Listening visits for maternal depression: a meta-analysis.. Arch Womens Ment Health., 2021. [DOI | PubMed]
- N Clinkscales, L Golds, K Berlouis, A MacBeth. The effectiveness of psychological interventions for anxiety in the perinatal period: a systematic review and meta-analysis.. Psychol Psychother., 2023. [DOI | PubMed]
- P Cuijpers, P Franco, M Ciharova. Psychological treatment of perinatal depression: a meta-analysis.. Psychol Med., 2023. [DOI | PubMed]
- CL Dennis, T Dowswell. Psychosocial and psychological interventions for preventing postpartum depression.. Cochrane Database Syst Rev., 2013. [DOI]
- LL Leng, XC Yin, SM Ng. Mindfulness-based intervention for clinical and subthreshold perinatal depression and anxiety: a systematic review and meta-analysis of randomized controlled trial.. Compr Psychiatry., 2023. [DOI | PubMed]
- LE Sockol. A systematic review of the efficacy of cognitive behavioral therapy for treating and preventing perinatal depression.. J Affect Disord., 2015. [DOI | PubMed]
- R Webb, R Bond, B Romero-Gonzalez, R Mycroft, S Ayers. Interventions to treat fear of childbirth in pregnancy: a systematic review and meta-analysis.. Psychol Med., 2021. [DOI]
- M Zimmermann, C Julce, P Sarkar. Can psychological interventions prevent or reduce risk for perinatal anxiety disorders? A systematic review and meta-analysis.. Gen Hosp Psychiatry., 2023. [DOI | PubMed]
- D Pettman, H O’Mahen, O Blomberg, AS Svanberg, L von Essen, J Woodford. Effectiveness of cognitive behavioural therapy-based interventions for maternal perinatal depression: a systematic review and meta-analysis.. BMC Psychiatry., 2023. [DOI | PubMed]
- Z Babiy, H Layton, CD Savoy. One-day peer-delivered cognitive behavioral therapy-based workshops for postpartum depression: a randomized controlled trial.. Psychother Psychosom., 2024. [DOI | PubMed]
- RJ Van Lieshout, H Layton, CD Savoy. Effect of online 1-day cognitive behavioral therapy-based workshops plus usual care vs usual care alone for postpartum depression: a randomized clinical trial.. JAMA Psychiatry., 2021. [DOI | PubMed]
- RJ Van Lieshout, H Layton, CD Savoy. In-person 1-day cognitive behavioral therapy-based workshops for postpartum depression: a randomized controlled trial.. Psychol Med., 2023. [DOI]
- S Lara-Cinisomo, A Ramirez Olarte, M Rosales, AZ Barrera. A systematic review of technology-based prevention and treatment interventions for perinatal depression and anxiety in Latina and African American women.. Matern Child Health J., 2021. [DOI | PubMed]
- X Li, DP Laplante, V Paquin, S Lafortune, G Elgbeili, S King. Effectiveness of cognitive behavioral therapy for perinatal maternal depression, anxiety and stress: a systematic review and meta-analysis of randomized controlled trials.. Clin Psychol Rev., 2022. [DOI | PubMed]
- U Nair, NR Armfield, MD Chatfield, S Edirippulige. The effectiveness of telemedicine interventions to address maternal depression: a systematic review and meta-analysis.. J Telemed Telecare., 2018. [DOI | PubMed]
- SA Loughnan, AE Joubert, A Grierson, G Andrews, JM Newby. Internet-delivered psychological interventions for clinical anxiety and depression in perinatal women: a systematic review and meta-analysis.. Arch Womens Ment Health., 2019. [DOI | PubMed]
- SA Loughnan, M Wallace, AE Joubert, H Haskelberg, G Andrews, JM Newby. A systematic review of psychological treatments for clinical anxiety during the perinatal period.. Arch Womens Ment Health., 2018. [DOI | PubMed]
- J Chae, HK Kim. Internet-based prenatal interventions for maternal health among pregnant women: a systematic review and meta-analysis.. Child Youth Serv Rev., 2021. [DOI]
- B Amani, D Merza, C Savoy. Peer-delivered cognitive-behavioral therapy for postpartum depression: a randomized controlled trial.. J Clin Psychiatry., 2021. [DOI]
- K Huh, H Layton, CD Savoy. Online public health nurse-delivered group cognitive behavioral therapy for postpartum depression: a randomized controlled trial during the COVID-19 pandemic.. J Clin Psychiatry., 2023. [DOI]
- D Merza, H Layton, C Savoy. Online peer-delivered 1-day cognitive behavioral therapy-based workshops for postpartum depression: a pilot study.. J Clin Psychiatry., 2023. [DOI]
- RJ Van Lieshout, H Layton, CD Savoy. Public health nurse-delivered group cognitive behavioural therapy for postpartum depression: a randomized controlled trial.. Can J Psychiatry., 2022. [DOI | PubMed]
- E Dominguez-Solis, M Lima-Serrano, JS Lima-Rodriguez. Non-pharmacological interventions to reduce anxiety in pregnancy, labour and postpartum: a systematic review.. Midwifery., 2021. [DOI | PubMed]
- O Gureje, BD Oladeji, AA Montgomery. High- versus low-intensity interventions for perinatal depression delivered by non-specialist primary maternal care providers in Nigeria: cluster randomised controlled trial (the EXPONATE trial).. Br J Psychiatry., 2019. [DOI | PubMed]
- J Milgrom, C Schembri, J Ericksen, J Ross, AW Gemmill. Towards parenthood: an antenatal intervention to reduce depression, anxiety and parenting difficulties.. J Affect Disord., 2011. [DOI | PubMed]
- H O’Mahen, JA Himle, G Fedock, E Henshaw, H Flynn. A pilot randomized controlled trial of cognitive behavioral therapy for perinatal depression adapted for women with low incomes.. Depress Anxiety., 2013. [DOI | PubMed]
- HA O’Mahen, DA Richards, J Woodford. Netmums: a phase II randomized controlled trial of a guided internet behavioural activation treatment for postpartum depression.. Psychol Med., 2014. [DOI | PubMed]
- K Trevillion, EG Ryan, A Pickles. An exploratory parallel-group randomised controlled trial of antenatal guided self-help (plus usual care) versus usual care alone for pregnant women with depression: DAWN trial.. J Affect Disord., 2020. [DOI | PubMed]
- F Vanobberghen, HA Weiss, DC Fuhr. Effectiveness of the thinking healthy programme for perinatal depression delivered through peers: pooled analysis of two randomized controlled trials in India and Pakistan.. J Affect Disord., 2020. [DOI | PubMed]
- JE Reid, KR Laws, L Drummond. Cognitive behavioural therapy with exposure and response prevention in the treatment of obsessive-compulsive disorder: a systematic review and meta-analysis of randomised controlled trials.. Compr Psychiatry., 2021. [DOI | PubMed]
- FL Challacombe, PM Salkovskis, M Woolgar, EL Wilkinson, J Read, R Acheson. A pilot randomized controlled trial of time-intensive cognitive-behaviour therapy for postpartum obsessive-compulsive disorder: effects on maternal symptoms, mother-infant interactions and attachment.. Psychol Med., 2017. [DOI | PubMed]
- LF de Graaff, A Honig, MG van Pampus, CAI Stramrood. Preventing post-traumatic stress disorder following childbirth and traumatic birth experiences: a systematic review.. Acta Obstet Gynecol Scand., 2018. [DOI | PubMed]
- HM Stileman, CA Jones. Revisiting the debriefing debate: does psychological debriefing reduce PTSD symptomology following work-related trauma? A meta-analysis.. Front Psychol., 2023. [DOI | PubMed]
- PA Coventry, N Meader, H Melton. Psychological and pharmacological interventions for posttraumatic stress disorder and comorbid mental health problems following complex traumatic events: systematic review and component network meta-analysis.. PLoS Med., 2020. [DOI]
- CR DeJesus, SL Trendel, DM Sloan. A systematic review of written exposure therapy for the treatment of posttraumatic stress symptoms.. Psychol Trauma., 2024. [DOI]
- KA Jones, I Freijah, SE Brennan. Interventions from pregnancy to two years after birth for parents experiencing complex post-traumatic stress disorder and/or with childhood experience of maltreatment.. Cochrane Database Syst Rev., 2023. [DOI]
- YI Nillni, TD Baul, E Paul, LB Godfrey, DM Sloan, SE Valentine. Written exposure therapy for treatment of perinatal PTSD among women with comorbid PTSD and SUD: a pilot study examining feasibility, acceptability, and preliminary effectiveness.. Gen Hosp Psychiatry., 2023. [DOI | PubMed]
- KM Deligiannidis, S Meltzer-Brody, H Gunduz-Bruce. Effect of zuranolone vs placebo in postpartum depression: a randomized clinical trial.. JAMA Psychiatry., 2021. [DOI | PubMed]
- S Meltzer-Brody, H Colquhoun, R Riesenberg. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials.. Lancet., 2018. [DOI | PubMed]
- SN Vigod, N Hussain-Shamsy, DE Stewart. A patient decision aid for antidepressant use in pregnancy: pilot randomized controlled trial.. J Affect Disord., 2019. [DOI | PubMed]
- LC Broughton, NJ Medlicott, AJ Smith. Effectiveness of patient decision aids in women considering psychotropic medication use during pregnancy: a literature review.. Arch Womens Ment Health., 2021. [DOI | PubMed]
- TC Hoffmann, M Bakhit, MA Durand, L Perestelo-Perez, C Saunders, JP Brito. Basing information on comprehensive, critically appraised, and up-to-date syntheses of the scientific evidence: an update from the international patient decision aid standards.. Med Decis Making., 2021. [DOI | PubMed]
- J Sprague, KL Wisner, DL Bogen. Pharmacotherapy for depression and bipolar disorder during lactation: a framework to aid decision making.. Semin Perinatol., 2020. [DOI | PubMed]
- N Kronenfeld, M Berlin, D Shaniv, M Berkovitch. Use of psychotropic medications in breastfeeding women.. Birth Defects Res., 2017. [DOI | PubMed]
- G Schoretsanitis, O Spigset, JC Stingl, KM Deligiannidis, M Paulzen, AA Westin. The impact of pregnancy on the pharmacokinetics of antidepressants: a systematic critical review and meta-analysis.. Expert Opin Drug Metab Toxicol., 2020. [DOI | PubMed]
- FMC Besag, MJ Vasey. Should antidepressants be avoided in pregnancy?. Drug Saf., 2023. [DOI | PubMed]
- G Eleftheriou, R Zandonella Callegher, R Butera. Consensus panel recommendations for the pharmacological management of pregnant women with depressive disorders.. Int J Environ Res Public Health., 2023. [DOI]
- TK Robakis, S Miyares, V Bergink. Risks and benefits of pre-delivery taper in pregnant women taking antidepressants.. Acta Psychiatr Scand., 2024. [DOI]
- W Warburton, C Hertzman, TF Oberlander. A register study of the impact of stopping third trimester selective serotonin reuptake inhibitor exposure on neonatal health.. Acta Psychiatr Scand., 2010. [DOI | PubMed]
- MI Kjaersgaard, ET Parner, M Vestergaard. Prenatal antidepressant exposure and risk of spontaneous abortion – a population-based study.. PLoS One., 2013. [DOI]
- P Desaunay, LG Eude, M Dreyfus. Benefits and risks of antidepressant drugs during pregnancy: a systematic review of meta-analyses.. Paediatr Drugs., 2023. [DOI | PubMed]
- SB Gumusoglu, BM Schickling, JA Vignato, DA Santillan, MK Santillan. Selective serotonin reuptake inhibitors and preeclampsia: a quality assessment and meta-analysis.. Pregnancy Hypertens., 2022. [DOI | PubMed]
- A Skalkidou, I Sundstrom-Poromaa, A Wikman, S Hesselman, AK Wikstrom, E Elenis. SSRI use during pregnancy and risk for postpartum haemorrhage: a national register-based cohort study in Sweden.. BJOG., 2020. [DOI | PubMed]
- SY Gao, QJ Wu, C Sun. Selective serotonin reuptake inhibitor use during early pregnancy and congenital malformations: a systematic review and meta-analysis of cohort studies of more than 9 million births.. BMC Med., 2018. [DOI | PubMed]
- C De Vries, S Gadzhanova, MJ Sykes, M Ward, E Roughead. A systematic review and meta-analysis considering the risk for congenital heart defects of antidepressant classes and individual antidepressants.. Drug Saf., 2021. [DOI | PubMed]
- S Grigoriadis, EH VonderPorten, L Mamisashvili. Antidepressant exposure during pregnancy and congenital malformations: is there an association? A systematic review and meta-analysis of the best evidence.. J Clin Psychiatry., 2013. [DOI]
- ZQ Lou, YY Zhou, X Zhang, HY Jiang. Exposure to selective noradrenalin reuptake inhibitors during the first trimester of pregnancy and risk of congenital malformations: a meta-analysis of cohort studies.. Psychiatry Res., 2022. [DOI | PubMed]
- E Turner, M Jones, LR Vaz, T Coleman. Systematic review and meta-analysis to assess the safety of bupropion and varenicline in pregnancy.. Nicotine Tob Res., 2019. [DOI | PubMed]
- D Kennedy, WS Webster, M Hill, HE Ritchie. Abnormal pregnancy outcome associated with high-dose maternal tranylcypromine therapy: case report and literature review.. Reprod Toxicol., 2017. [DOI | PubMed]
- A Biffi, A Cantarutti, F Rea, A Locatelli, R Zanini, G Corrao. Use of antidepressants during pregnancy and neonatal outcomes: an umbrella review of meta-analyses of observational studies.. J Psychiatr Res., 2020. [DOI | PubMed]
- R Vlenterie, M van Gelder, HR Anderson. Associations between maternal depression, antidepressant use during pregnancy, and adverse pregnancy outcomes: an individual participant data meta-analysis.. Obstet Gynecol., 2021. [DOI | PubMed]
- D Xing, R Wu, L Chen, T Wang. Maternal use of antidepressants during pregnancy and risks for adverse perinatal outcomes: a meta-analysis.. J Psychosom Res., 2020. [DOI | PubMed]
- R Masarwa, B Bar-Oz, E Gorelik, S Reif, A Perlman, I Matok. Prenatal exposure to selective serotonin reuptake inhibitors and serotonin norepinephrine reuptake inhibitors and risk for persistent pulmonary hypertension of the newborn: a systematic review, meta-analysis, and network meta-analysis.. Am J Obstet Gynecol., 2019. [DOI]
- J Wang, F Cosci. Neonatal withdrawal syndrome following late in utero exposure to selective serotonin reuptake inhibitors: a systematic review and meta-analysis of observational studies.. Psychother Psychosom., 2021. [DOI | PubMed]
- AS Rommel, V Bergink, X Liu, T Munk-Olsen, NM Molenaar. Long-term effects of intrauterine exposure to antidepressants on physical, neurodevelopmental, and psychiatric outcomes: a systematic review.. J Clin Psychiatry., 2020. [DOI]
- G Eleftheriou, R Zandonella Callegher, R Butera. Consensus panel recommendations for the pharmacological management of breastfeeding women with postpartum depression.. Int J Environ Res Public Health., 2024. [DOI]
- TWK Hale. K. Hale’s medications and mothers’ milk 2023: a manual of lactational pharmacology., 2023
- AA Veroniki, E Cogo, P Rios. Comparative safety of anti-epileptic drugs during pregnancy: a systematic review and network meta-analysis of congenital malformations and prenatal outcomes.. BMC Med., 2017. [DOI | PubMed]
- J Coste, PO Blotiere, S Miranda. Risk of early neurodevelopmental disorders associated with in utero exposure to valproate and other antiepileptic drugs: a nationwide cohort study in France.. Sci Rep., 2020. [DOI | PubMed]
- Potential risk of neurodevelopmental disorders in children born to men treated with valproate medicines: PRAC recommends precautionary measures.. 2024
- J Christensen, BB Trabjerg, JW Dreier. Valproate use during spermatogenesis and risk to offspring.. JAMA Netw Open., 2024. [DOI]
- T Tomson, G Muraca, N Razaz. Paternal exposure to antiepileptic drugs and offspring outcomes: a nationwide population-based cohort study in Sweden.. J Neurol Neurosurg Psychiatry., 2020. [DOI | PubMed]
- A Kazmin, RC Wong, M Sermer, G Koren. Antiepileptic drugs in pregnancy and hemorrhagic disease of the newborn: an update.. Can Fam Physician., 2010. [PubMed]
- EA Pinheiro, CS Stika. Drugs in pregnancy: pharmacologic and physiologic changes that affect clinical care.. Semin Perinatol., 2020. [DOI | PubMed]
- A Karanam, PB Pennell, JA French. Lamotrigine clearance increases by 5 weeks gestational age: relationship to estradiol concentrations and gestational age.. Ann Neurol., 2018. [DOI | PubMed]
- AR Polepally, PB Pennell, RC Brundage. Model-based lamotrigine clearance changes during pregnancy: clinical implication.. Ann Clin Transl Neurol., 2014. [DOI | PubMed]
- CT Clark, KL Wisner. Treatment of peripartum bipolar disorder.. Obstet Gynecol Clin North Am., 2018. [DOI | PubMed]
- T Tomson, D Battino, R Bromley. Management of epilepsy in pregnancy: a report from the international league against epilepsy task force on women and pregnancy.. Epileptic Disord., 2019. [DOI | PubMed]
- JR Geddes, A Gardiner, J Rendell. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2(2 factorial randomised trial.. Lancet Psychiatry., 2016. [DOI | PubMed]
- L Viale, J Allotey, F Cheong-See. Epilepsy in pregnancy and reproductive outcomes: a systematic review and meta-analysis.. Lancet., 2015. [DOI | PubMed]
- E Gedzelman, KJ Meador. Antiepileptic drugs in women with epilepsy during pregnancy.. Ther Adv Drug Saf., 2012. [DOI | PubMed]
- T Tomson, D Battino, E Bonizzoni. Comparative risk of major congenital malformations with eight different antiepileptic drugs: a prospective cohort study of the EURAP registry.. Lancet Neurol., 2018. [DOI | PubMed]
- J Jentink, MA Loane, H Dolk. Valproic acid monotherapy in pregnancy and major congenital malformations.. N Engl J Med., 2010. [DOI | PubMed]
- YC Kaplan, O Demir. Use of phenytoin, phenobarbital carbamazepine, levetiracetam lamotrigine and valproate in pregnancy and breastfeeding: risk of major malformations, dose-dependency, monotherapy vs polytherapy, pharmacokinetics and clinical implications.. Curr Neuropharmacol., 2021. [DOI | PubMed]
- A Lavu, C Vaccaro, E Zusman. Antiseizure medication use during pregnancy and neonatal growth outcomes: a systematic review and meta-analysis.. Br J Clin Pharmacol., 2024. [DOI]
- AV Margulis, S Hernandez-Diaz, T McElrath. Relation of in-utero exposure to antiepileptic drugs to pregnancy duration and size at birth.. PLoS One., 2019. [DOI]
- M Cohen-Israel, I Berger, EY Martonovich, G Klinger, B Stahl, N Linder. Short- and long-term complications of in utero exposure to lamotrigine.. Br J Clin Pharmacol., 2018. [DOI | PubMed]
- C Haskey, M Galbally. Mood stabilizers in pregnancy and child developmental outcomes: a systematic review.. Aust N Z J Psychiatry., 2017. [DOI | PubMed]
- M Kellogg, KJ Meador. Neurodevelopmental effects of antiepileptic drugs.. Neurochem Res., 2017. [DOI | PubMed]
- J Adams, PA Janulewicz, EA Macklin. Neuropsychological effects in children exposed to anticonvulsant monotherapy during gestation: phenobarbital, carbamazepine, and phenytoin.. Epilepsy Behav., 2022. [DOI | PubMed]
- KJ Meador, MJ Cohen, DW Loring. Cognitive outcomes at age 3 years in children with fetal exposure to antiseizure medications (MONEAD study) in the USA: a prospective, observational cohort study.. Lancet Neurol., 2023. [DOI | PubMed]
- Lamotrigine.. 2023
- Carbamazepine.. 2022
- 308Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Valproic Acid. [Updated 2024 Sep 15]. Accessed 7 August 2024. https://www.ncbi.nlm.nih.gov/books/NBK501274/
- HK Betcher, C Montiel, CT Clark. Use of antipsychotic drugs during pregnancy.. Curr Treat Options Psychiatry., 2019. [DOI | PubMed]
- G Eleftheriou, R Butera, A Sangiovanni, C Palumbo, E Bondi. Long-acting injectable antipsychotic treatment during pregnancy: a case series.. Int J Environ Res Public Health., 2023. [DOI]
- SA Reinstein, J Cosgrove, T Malekshahi, KM Deligiannidis. Long-acting injectable antipsychotic use during pregnancy: a brief review and concise guide for clinicians.. J Clin Psychiatry., 2020. [DOI]
- KF Huybrechts, S Hernandez-Diaz, E Patorno. Antipsychotic use in pregnancy and the risk for congenital malformations.. JAMA Psychiatry., 2016. [DOI | PubMed]
- SN Vigod, T Gomes, AS Wilton, VH Taylor, JG Ray. Antipsychotic drug use in pregnancy: high dimensional, propensity matched, population based cohort study.. Br Med J., 2015. [DOI]
- E Heinonen, L Forsberg, U Norby, K Wide, K Kallen. Antipsychotic use during pregnancy and risk for gestational diabetes: a national register-based cohort study in Sweden.. CNS Drugs., 2022. [DOI | PubMed]
- Z Wang, ICK Wong, KKC Man, BH Alfageh, P Mongkhon, R Brauer. The use of antipsychotic agents during pregnancy and the risk of gestational diabetes mellitus: a systematic review and meta-analysis.. Psychol Med., 2021. [DOI | PubMed]
- EMP Poels, L Schrijver, AM Kamperman. Long-term neurodevelopmental consequences of intrauterine exposure to lithium and antipsychotics: a systematic review and meta-analysis.. Eur Child Adolesc Psychiatry., 2018. [DOI | PubMed]
- CG Coughlin, KA Blackwell, C Bartley, M Hay, KA Yonkers, MH Bloch. Obstetric and neonatal outcomes after antipsychotic medication exposure in pregnancy.. Obstet Gynecol., 2015. [DOI | PubMed]
- Z Wang, R Brauer, KKC Man, B Alfageh, P Mongkhon, ICK Wong. Prenatal exposure to antipsychotic agents and the risk of congenital malformations in children: a systematic review and meta-analysis.. Br J Clin Pharmacol., 2021. [DOI | PubMed]
- LS Cohen, TR Church, MP Freeman. Reproductive safety of lurasidone and quetiapine: update from the national pregnancy registry for psychiatric medications.. J Womens Health (Larchmt)., 2023. [DOI | PubMed]
- Z Wang, AYL Chan, D Coghill. Association between prenatal exposure to antipsychotics and attention-deficit/hyperactivity disorder, autism spectrum disorder, preterm birth, and small for gestational age.. JAMA Intern Med., 2021. [DOI | PubMed]
- I Pacchiarotti, J Leon-Caballero, A Murru. Mood stabilizers and antipsychotics during breastfeeding: focus on bipolar disorder.. Eur Neuropsychopharmacol., 2016. [DOI | PubMed]
- F Uguz. Second-generation antipsychotics during the lactation period: a comparative systematic review on infant safety.. J Clin Psychopharmacol., 2016. [DOI | PubMed]
- Risperidone. National Library of Medicine.. 2024
- Aripiprazole.. 2024
- Haloperidol.. 2023
- Chlorpromazine.. 2022
- J Galea, S Ellul, A Schembri, P Schembri-Wismayer, J Calleja-Agius. Ebstein anomaly: a review.. Neonatal Netw., 2014. [DOI | PubMed]
- M Fornaro, E Maritan, R Ferranti. Lithium exposure during pregnancy and the postpartum period: a systematic review and meta-analysis of safety and efficacy outcomes.. Am J Psychiatry., 2020. [DOI | PubMed]
- R Hastie, S Tong, R Hiscock. Maternal lithium use and the risk of adverse pregnancy and neonatal outcomes: a Swedish population-based cohort study.. BMC Med., 2021. [DOI | PubMed]
- T Munk-Olsen, X Liu, A Viktorin. Maternal and infant outcomes associated with lithium use in pregnancy: an international collaborative meta-analysis of six cohort studies.. Lancet Psychiatry., 2018. [DOI | PubMed]
- E Patorno, KF Huybrechts, BT Bateman. Lithium use in pregnancy and the risk of cardiac malformations.. N Engl J Med., 2017. [DOI | PubMed]
- EMP Poels, HH Bijma, M Galbally, V Bergink. Lithium during pregnancy and after delivery: a review.. Int J Bipolar Disord., 2018. [DOI | PubMed]
- CT Clark, RL Newmark, KL Wisner, C Stika, MJ Avram. Lithium pharmacokinetics in the perinatal patient with bipolar disorder.. J Clin Pharmacol., 2022. [DOI | PubMed]
- AA Westin, M Brekke, E Molden, E Skogvoll, M Aadal, O Spigset. Changes in drug disposition of lithium during pregnancy: a retrospective observational study of patient data from two routine therapeutic drug monitoring services in Norway.. BMJ Open., 2017. [DOI]
- KM Deligiannidis, N Byatt, MP Freeman. Pharmacotherapy for mood disorders in pregnancy: a review of pharmacokinetic changes and clinical recommendations for therapeutic drug monitoring.. J Clin Psychopharmacol., 2014. [DOI | PubMed]
- DJ Newport, AC Viguera, AJ Beach, JC Ritchie, LS Cohen, ZN Stowe. Lithium placental passage and obstetrical outcome: implications for clinical management during late pregnancy.. Am J Psychiatry., 2005. [DOI | PubMed]
- NM Molenaar, EMP Poels, T Robakis, R Wesseloo, V Bergink. Management of lithium dosing around delivery: an observational study.. Bipolar Disord., 2021. [DOI | PubMed]
- F Girard, T Suhara, T Sassa. 7Li 2d CSI of human brain on a clinical scanner.. MAGMA., 2001. [DOI | PubMed]
- PF Renshaw, S Wicklund. In vivo measurement of lithium in humans by nuclear magnetic resonance spectroscopy.. Biol Psychiatry., 1988. [DOI | PubMed]
- ML Imaz, M Torra, D Soy, L Garcia-Esteve, R Martin-Santos. Clinical lactation studies of lithium: a systematic review.. Front Pharmacol., 2019. [DOI | PubMed]
- M Galbally, V Bergink, SN Vigod. Breastfeeding and lithium: is breast always best?. Lancet Psychiatry., 2018. [DOI | PubMed]
- S Grigoriadis, L Graves, M Peer. Benzodiazepine use during pregnancy alone or in combination with an antidepressant and congenital malformations: systematic review and meta-analysis.. J Clin Psychiatry., 2019. [DOI]
- S Grigoriadis, L Graves, M Peer. Pregnancy and delivery outcomes following benzodiazepine exposure: a systematic review and meta-analysis.. Can J Psychiatry., 2020. [DOI | PubMed]
- S Grigoriadis, A Alibrahim, JK Mansfield, A Sullovey, GE Robinson. Hypnotic benzodiazepine receptor agonist exposure during pregnancy and the risk of congenital malformations and other adverse pregnancy outcomes: a systematic review and meta-analysis.. Acta Psychiatr Scand., 2022. [DOI | PubMed]
- B Bandelow, C Allgulander, DS Baldwin. World federation of societies of biological psychiatry (WFSBP) guidelines for treatment of anxiety, obsessive-compulsive and posttraumatic stress disorders – version 3. Part I: anxiety disorders.. World J Biol Psychiatry., 2023. [PubMed]
- MA Katzman, P Bleau, P Blier. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive-compulsive disorders.. BMC Psychiatry., 2014. [DOI]
- PP Schnurr, JL Hamblen, J Wolf. The management of posttraumatic stress disorder and acute stress disorder: synopsis of the 2023 U.S. Department of Veterans Affairs and U.S. Department of Defense clinical practice guideline.. Ann Intern Med., 2024. [DOI | PubMed]
- R Robiyanto, M Roos, JHJ Bos, E Hak, EP van Puijenbroek, CCM Schuiling-Veninga. Switching pattern and dose adjustment of antidepressants before and during pregnancy.. Arch Womens Ment Health., 2023. [DOI | PubMed]
- B Kallen, M Reis. Neonatal complications after maternal concomitant use of SSRI and other central nervous system active drugs during the second or third trimester of pregnancy.. J Clin Psychopharmacol., 2012. [DOI | PubMed]
- H Bayrampour, A Kapoor, M Bunka, D Ryan. The risk of relapse of depression during pregnancy after discontinuation of antidepressants: a systematic review and meta-analysis.. J Clin Psychiatry., 2020. [DOI]
- RM Pacilio, JF Lopez, SV Parikh, PD Patel, JA Geller. Safe ketamine use and pregnancy: a nationwide survey and retrospective review of informed consent, counseling, and testing practices.. J Clin Psychiatry., 2024. [DOI]
- JVE Brown, CA Wilson, K Ayre. Antidepressant treatment for postnatal depression.. Cochrane Database Syst Rev., 2021. [DOI]
- D Chibanda, AK Shetty, M Tshimanga, G Woelk, L Stranix-Chibanda, S Rusakaniko. Group problem-solving therapy for postnatal depression among HIV-positive and HIV-negative mothers in Zimbabwe.. J Int Assoc Provid AIDS Care., 2014. [DOI | PubMed]
- E Molyneaux, LA Telesia, C Henshaw, E Boath, E Bradley, LM Howard. Antidepressants for preventing postnatal depression.. Cochrane Database Syst Rev., 2018. [DOI]
- MH Bloch, J McGuire, A Landeros-Weisenberger, JF Leckman, C Pittenger. Meta-analysis of the dose-response relationship of SSRI in obsessive-compulsive disorder.. Mol Psychiatry., 2010. [DOI | PubMed]
- M Van Ameringen, N Fineberg, B Dell’Osso, E Hollander. New International Treatment Guidelines for Obsessive-Compulsive Disorder. In: 34th CINP World Congress of Neuropsychopharmacology, Montreal, QC, Canada, 7-10 May. 2023
- T Williams, NJ Phillips, DJ Stein, JC Ipser. Pharmacotherapy for post-traumatic stress disorder (PTSD).. Cochrane Database Syst Rev., 2022. [DOI]
- AD Davidson, A Bhat, F Chu, JN Rice, NA Nduom, DS Cowley. A systematic review of the use of prazosin in pregnancy and lactation.. Gen Hosp Psychiatry., 2021. [DOI | PubMed]
- AC Viguera, T Whitfield, RJ Baldessarini. Risk of recurrence in women with bipolar disorder during pregnancy: prospective study of mood stabilizer discontinuation.. Am J Psychiatry., 2007. [DOI | PubMed]
- B Buonomo, A Brunello, S Noli. Tamoxifen exposure during pregnancy: a systematic review and three more cases.. Breast Care (Basel)., 2020. [DOI | PubMed]
- LN Yatham, T Chakrabarty, DJ Bond. Canadian network for mood and anxiety treatments (CANMAT) and international society for bipolar disorders (ISBD) recommendations for the management of patients with bipolar disorder with mixed presentations.. Bipolar Disord., 2021. [DOI | PubMed]
- Drugs and Lactation Database (LactMed(R)).. 2006
- HJ Lee, SM Kim, JY Kwon. Repetitive transcranial magnetic stimulation treatment for peripartum depression: systematic review & meta-analysis.. BMC Pregnancy Childbirth., 2021. [DOI | PubMed]
- C Liu, W Pan, L Jia. Efficacy and safety of repetitive transcranial magnetic stimulation for peripartum depression: a meta-analysis of randomized controlled trials.. Psychiatry Res., 2020. [DOI | PubMed]
- L Peng, C Fu, F Xiong. Effects of repetitive transcranial magnetic stimulation on depression symptoms and cognitive function in treating patients with postpartum depression: a systematic review and meta-analysis of randomized controlled trials.. Psychiatry Res., 2020. [DOI | PubMed]
- DR Kim, E Wang, B McGeehan. Randomized controlled trial of transcranial magnetic stimulation in pregnant women with major depressive disorder.. Brain Stimul., 2019. [DOI | PubMed]
- A Miuli, M Pettorruso, G Stefanelli. Beyond the efficacy of transcranial magnetic stimulation in peripartum depression: a systematic review exploring perinatal safety for newborns.. Psychiatry Res., 2023. [DOI | PubMed]
- GN Konstantinou, SN Vigod, S Mehta, ZJ Daskalakis, DM Blumberger. A systematic review of non-invasive neurostimulation for the treatment of depression during pregnancy.. J Affect Disord., 2020. [DOI | PubMed]
- A Laurin, N Nard, M Dalmont. Efficacy and safety of transcranial electric stimulation during the perinatal period: a systematic literature review and three case reports.. J Clin Med., 2022. [DOI]
- F Pacheco, R Guiomar, AR Brunoni. Efficacy of non-invasive brain stimulation in decreasing depression symptoms during the peripartum period: a systematic review.. J Psychiatr Res., 2021. [DOI | PubMed]
- SN Vigod, KE Murphy, CL Dennis. Transcranial direct current stimulation (tDCS) for depression in pregnancy: a pilot randomized controlled trial.. Brain Stimul., 2019. [DOI | PubMed]
- K Calaway, S Coshal, K Jones, J Coverdale, R Livingston. A systematic review of the safety of electroconvulsive therapy use during the first trimester of pregnancy.. J ECT., 2016. [DOI | PubMed]
- S Coshal, K Jones, J Coverdale, R Livingston. An overview of reviews on the safety of electroconvulsive therapy administered during pregnancy.. J Psychiatr Pract., 2019. [DOI | PubMed]
- HB Ward, JA Fromson, JJ Cooper, G De Oliveira, M Almeida. Recommendations for the use of ECT in pregnancy: literature review and proposed clinical protocol.. Arch Womens Ment Health., 2018. [DOI | PubMed]
- Statement on resuming breastfeeding after anesthesia. American Society of Anesthesiologists,. 2019
- R Chow, E Huang, A Li. Appraisal of systematic reviews on interventions for postpartum depression: systematic review.. BMC Pregnancy Childbirth., 2021. [DOI | PubMed]
- L Corrigan, P Moran, N McGrath, J Eustace-Cook, D Daly. The characteristics and effectiveness of pregnancy yoga interventions: a systematic review and meta-analysis.. BMC Pregnancy Childbirth., 2022. [DOI | PubMed]
- MA Gargari, K Esmailpour, M Mirghafourvand, R Nourizadeh, E Mehrabi. Effects of psycho-education interventions on perceived childbirth fear and anxiety by pregnant women: A systematic review and meta-analysis.. Int J Women Health Rep Sci., 2021
- D Kolomanska, M Zarawski, A Mazur-Bialy. Physical activity and depressive disorders in pregnant women-a systematic review.. Medicina (B Aires)., 2019. [DOI]
- R Kwon, K Kasper, S London, DM Haas. A systematic review: the effects of yoga on pregnancy.. Eur J Obstet Gynecol Reprod Biol., 2020. [DOI | PubMed]
- IH Lin, CY Huang, SH Chou, CL Shih. Efficacy of prenatal yoga in the treatment of depression and anxiety during pregnancy: a systematic review and meta-analysis.. Int J Environ Res Public Health., 2022. [DOI]
- W Min, C Jiang, Z Li, Z Wang. The effect of mindfulness-based interventions during pregnancy on postpartum mental health: a meta-analysis.. J Affect Disord., 2023. [DOI | PubMed]
- G Wang, C Liang, G Sun. Yoga’s therapeutic effect on perinatal depression: a systematic review and meta-analysis.. Psychiatr Danub., 2022. [DOI | PubMed]
- X Yu, Y Liu, Y Huang, T Zeng. The effect of nonpharmacological interventions on the mental health of high-risk pregnant women: a systematic review.. Complement Ther Med., 2022. [DOI | PubMed]
- Y Zhu, R Wang, X Tang, Q Li, G Xu, A Zhang. The effect of music, massage, yoga and exercise on antenatal depression: a meta-analysis.. J Affect Disord., 2021. [DOI | PubMed]
- CA Smith, Z Shewamene, M Galbally, V Schmied, H Dahlen. The effect of complementary medicines and therapies on maternal anxiety and depression in pregnancy: a systematic review and meta-analysis.. J Affect Disord., 2019. [DOI | PubMed]
- RJ McCloskey, R Reno. Complementary health approaches for postpartum depression: a systematic review.. Soc Work Ment Health., 2019. [DOI]
- O Villar-Alises, P Martinez-Miranda, J Martinez-Calderon. Prenatal yoga-based interventions may improve mental health during pregnancy: an overview of systematic reviews with meta-analysis.. Int J Environ Res Public Health., 2023. [DOI]
- X Jiang, H Li, D Wang, L Shan, F Wang, Y Kang. Efficacy of nondrug interventions in perinatal depression: a meta-analysis.. Psychiatry Res., 2022. [DOI | PubMed]
- L Liu, C Liu, X Liu, Y Yang. Summary of the effect of an exercise intervention on antenatal depression and the optimal program: a systematic review and meta-analysis.. BMC Pregnancy Childbirth., 2023. [DOI | PubMed]
- NS Jarbou, KA Newell. Exercise and yoga during pregnancy and their impact on depression: a systematic literature review.. Arch Womens Ment Health., 2022. [DOI | PubMed]
- P Tong, LP Dong, Y Yang, YH Shi, T Sun, P Bo. Traditional Chinese acupuncture and postpartum depression: a systematic review and meta-analysis.. J Chin Med Assoc., 2019. [DOI | PubMed]
- HG Hall, R Cant, N Munk. The effectiveness of massage for reducing pregnant women’s anxiety and depression; systematic review and meta-analysis.. Midwifery., 2020. [DOI | PubMed]
- SM Mueller, M Grunwald. Effects, side effects and contraindications of relaxation massage during pregnancy: a systematic review of randomized controlled trials.. J Clin Med., 2021. [DOI]
- Q Wu, Z Liu, X Pang, L Cheng. Efficacy of five-element music interventions in perinatal mental health and labor pain: a meta-analysis.. Complement Ther Clin Pract., 2020. [DOI | PubMed]
- WJ Yang, YM Bai, L Qin. The effectiveness of music therapy for postpartum depression: a systematic review and meta-analysis.. Complement Ther Clin Pract., 2019. [DOI | PubMed]
- M Konsam, SRB D’Souza, SK Praharaj. Effectiveness of music on perinatal anxiety among pregnant women and newborn behaviors: a systematic review and narrative synthesis.. Indian J Psychol Med., 2023. [DOI | PubMed]
- CJ Lin, YC Chang, YH Chang. Music interventions for anxiety in pregnant women: a systematic review and meta-analysis of randomized controlled trials.. J Clin Med., 2019. [DOI]
- M Abera, C Hanlon, B Daniel. Effects of relaxation interventions during pregnancy on maternal mental health, and pregnancy and newborn outcomes: a systematic review and meta-analysis.. PLOS ONE., 2024. [DOI]
- R Widiasih, Y Rais, N Fithriyah, D Damayanti, NR Puspitasari. Intervention for preventing postpartum depression: a systematic review of studies in Indonesia.. Br J Midwifery., 2021. [DOI]
- M Rouhi, N Rouhi, S Mohamadpour, HP-R Tajrishi. Vitamin D reduces postpartum depression and fatigue among Iranian women.. Br J Midwifery., 2018. [DOI]
- J Tabeshpour, F Sobhani, SA Sadjadi. A double-blind, randomized, placebo-controlled trial of saffron stigma (Crocus sativus L.) in mothers suffering from mild-to-moderate postpartum depression.. Phytomedicine., 2017. [DOI | PubMed]
- Z Tsai, N Shah, U Tahir. Dietary interventions for perinatal depression and anxiety: a systematic review and meta-analysis of randomized controlled trials.. Am J Clin Nutr., 2023. [DOI | PubMed]
- A Lee, R Minhas, N Matsuda, M Lam, S Ito. The safety of St. John’s wort (Hypericum perforatum) during breastfeeding.. J Clin Psychiatry., 2003. [DOI | PubMed]
- W Schafer, N Wentzell, T Schink, U Haug. Characterization of pregnancies exposed to St. John’s wort and their outcomes: a claims data analysis.. Reprod Toxicol., 2021. [DOI | PubMed]
- M Abera, C Hanlon, B Daniel. Effects of relaxation interventions during pregnancy on maternal mental health, and pregnancy and newborn outcomes: a systematic review and meta-analysis.. PLoS One., 2024. [DOI]
- TM Hu, SH Lee, EW Loh. Effectiveness of aromatherapy for intrapartum and postpartum emotional problems among parturient women: a meta-analysis of randomized controlled trials.. Jpn J Nurs Sci., 2022. [DOI]
- K Rezaie-Keikhaie, M Hastings-Tolsma, S Bouya. Effect of aromatherapy on post-partum complications: a systematic review.. Complement Ther Clin Pract., 2019. [DOI | PubMed]
- Y Shamsunisha, A Arunesh, M Pandiaraja, V Venugopal, S Poonguzhali, M Kuppusamy. Aromatherapy for postpartum depression: a systematic review and meta-analysis.. J Family Reprod Health., 2023. [DOI | PubMed]
- SS Tsai, HH Wang, FH Chou. The effects of aromatherapy on postpartum women: a systematic review.. J Nurs Res., 2020. [DOI]
- V Desai, AL Kozyrskyj, S Lau. Effectiveness of probiotic, prebiotic, and synbiotic supplementation to improve perinatal mental health in mothers: a systematic review and meta-analysis.. Front Psychiatry., 2021. [DOI | PubMed]
- KC Trifkovic, D Micetic-Turk, S Kmetec. Efficacy of direct or indirect use of probiotics for the improvement of maternal depression during pregnancy and in the postnatal period: a systematic review and meta-analysis.. Healthcare., 2022. [DOI]
- RE Kendell, JC Chalmers, C Platz. Epidemiology of puerperal psychoses.. Br J Psychiatry., 1987. [DOI | PubMed]
- JH Kim, SS Choi, K Ha. A closer look at depression in mothers who kill their children: is it unipolar or bipolar depression?. J Clin Psychiatry., 2008. [DOI | PubMed]
- M Kimmel, H Thippeswamy, A Kamperman. Cross-continental collaboration for understanding postpartum major depression with psychotic features.. Front Glob Womens Health., 2022. [DOI | PubMed]
- V Sharma, D Mazmanian, L Palagini, A Bramante. Postpartum psychosis: revisiting the phenomenology, nosology, and treatment.. J Aff Disor Rep., 2022. [DOI]
- SM Albers, T Wen, C Monk. Postpartum psychosis during delivery hospitalizations and postpartum readmissions, 2016–2019.. Am J Obstet Gynecol MFM., 2023. [DOI | PubMed]
- V Sharma, KN Wood, B Weaver, D Mazmanian, M Thomson. Occurrence of postpartum manic or mixed episodes in women with bipolar I disorder: a systematic review and meta-analysis.. Bipolar Disord., 2024. [DOI | PubMed]
- C Carr, D Borges, K Lewis. Sleep and postpartum psychosis: a narrative review of the existing literature.. J Clin Med., 2023. [DOI]
- C Jairaj, G Seneviratne, V Bergink, IE Sommer, P Dazzan. Postpartum psychosis: a proposed treatment algorithm.. J Psychopharmacol., 2023. [DOI | PubMed]
- A Aftab, AA Shah. Behavioral emergencies: special considerations in the pregnant patient.. Psychiatr Clin North Am., 2017. [DOI | PubMed]
- MP Wilson, K Nordstrom, AA Shah, GM Vilke. Psychiatric emergencies in pregnant women.. Emerg Med Clin North Am., 2015. [DOI | PubMed]
- LM Prager, A Ivkovic. The Massachusetts general hospital comprehensive clinical psychiatry., 2016
- LL Altshuler, L Cohen, MP Szuba, VK Burt, M Gitlin, J Mintz. Pharmacologic management of psychiatric illness during pregnancy: dilemmas and guidelines.. Am J Psychiatry., 1996. [DOI | PubMed]
- M Xiao, Y Hu, S Huang, G Wang, J Zhao, J Lei. Prevalence of suicidal ideation in pregnancy and the postpartum: a systematic review and meta-analysis.. J Affect Disord., 2022. [DOI | PubMed]
- PCC Legazpi, MF Rodriguez-Munoz, ME Olivares-Crespo, N Izquierdo-Mendez. Review of suicidal ideation during pregnancy: risk factors, prevalence, assessment instruments and consequences.. Psicol Reflex Crit., 2022. [DOI | PubMed]
- LA Kobylski, J Keller, SD Molock, HN Le. Preventing perinatal suicide: an unmet public health need.. Lancet Public Health., 2023. [DOI]
- N Fairbrother, F Collardeau, SR Woody, DA Wolfe, JM Fawcett. Postpartum thoughts of infant-related harm and obsessive-compulsive disorder: relation to maternal physical aggression toward the infant.. J Clin Psychiatry., 2022. [DOI]
- FL Challacombe, AL Wroe. A hidden problem: consequences of the misdiagnosis of perinatal obsessive-compulsive disorder.. Br J Gen Pract., 2013. [DOI | PubMed]
- EC Brok, P Lok, DB Oosterbaan, AH Schene, I Tendolkar, PF van Eijndhoven. Infant-Related intrusive thoughts of harm in the postpartum period: a critical review.. J Clin Psychiatry., 2017. [DOI]
- WW Rao, XM Zhu, QQ Zong. Prevalence of prenatal and postpartum depression in fathers: a comprehensive meta-analysis of observational surveys.. J Affect Disord., 2020. [DOI | PubMed]
- NS Ansari, J Shah, CL Dennis, PS Shah. Risk factors for postpartum depressive symptoms among fathers: a systematic review and meta-analysis.. Acta Obstet Gynecol Scand., 2021. [DOI | PubMed]
- F Thiel, MM Pittelkow, HU Wittchen, S Garthus-Niegel. The relationship between paternal and maternal depression during the perinatal period: a systematic review and meta-analysis.. Front Psychiatry., 2020. [DOI | PubMed]
- JA Leiferman, CV Farewell, J Jewell. Anxiety among fathers during the prenatal and postpartum period: a meta-analysis.. J Psychosom Obstet Gynaecol., 2021. [DOI | PubMed]
- K Cheung, J Theule. Paternal depression and child externalizing behaviors: a meta-analysis.. J Fam Psychol., 2019. [DOI | PubMed]
- C Cui, M Li, Y Yang, C Liu, P Cao, L Wang. The effects of paternal perinatal depression on socioemotional and behavioral development of children: a meta-analysis of prospective studies.. Psychiatry Res., 2020. [DOI | PubMed]
- S Sweeney, C Wilson. Parental anxiety and offspring development: a systematic review.. J Affect Disord., 2023. [DOI | PubMed]
- KA Abelsohn, R Epstein, LE Ross. Celebrating the “other” parent: mental health and wellness of expecting lesbian, bisexual, and queer non-birth parents.. J Gay Lesbian Ment Health., 2013. [DOI]
- Z Darwin, J Domoney, J Iles, F Bristow, J Siew, V Sethna. Assessing the mental health of fathers, other co-parents, and partners in the perinatal period: mixed methods evidence synthesis.. Front Psychiatry., 2020. [DOI | PubMed]
- KRS Soled, PM Niles, E Mantell, M Dansky, W Bockting, M George. Childbearing at the margins: a systematic metasynthesis of sexual and gender diverse childbearing experiences.. Birth., 2023. [DOI | PubMed]
- C Davenport, J Lambie, C Owen, V Swami. Fathers’ experience of depression during the perinatal period: a qualitative systematic review.. JBI Evid Synth., 2022. [DOI | PubMed]
- AK Shafian, S Mohamed, NJ Nasution Raduan, AY Hway Ann. A systematic review and meta-analysis of studies validating Edinburgh postnatal depression scale in fathers.. Heliyon., 2022. [DOI]
- Z Goldstein, B Rosen, A Howlett, M Anderson, D Herman. Interventions for paternal perinatal depression: a systematic review.. J Affect Disord., 2020. [DOI | PubMed]
- ML Fisher, P Sutcliffe, C Southern, AL Grove, BK Tan. The effectiveness of interventions for the prevention or treatment of paternal perinatal anxiety: a systematic review.. J Clin Med., 2022. [DOI]
- SF Ghaffari, F Elyasi, SN Mousavinasab, Z Shahhosseini. A systematic review of clinical trials affecting anxiety, stress and fear of childbirth in expectant fathers.. Nurs Open., 2021. [DOI | PubMed]
