Chemical Variability of the Leaf Essential Oil of Two Subspecies of Juniperus oxycedrus L. From Northwestern Algeria
Abstract
Thirty‐seven essential oil samples have been isolated from leaves of individual plants of Juniperus oxycedrus (subspecies oxycedrus and macrocarpa) growing wild in Tlemcen and Aïn Témouchent provinces (Northwestern Algeria). Analysis of eight selected oil samples by GC(RI), GC/MS and 13C NMR, allowed the identification of 88 components that accounted for 84.4–99.5 % of the whole compositions. The 37 compositions were subjected to statistical analysis which suggested the existence of groups and sub‐groups I, IIA and IIB. Group I, the most important group with 24 oil samples out of 37, differed from sub‐groups IIA and IIB, essentially by the high content of α‐pinene (mean value, 69.2 %; SD 6.24). Sub‐groups IIA and IIB were differentiated by their content of α‐pinene (Mean values, 49.7 % and 27.3 %, respectively) as well as δ‐3‐carene (10.3 %, sub‐group IIA), sabinene and (Z)‐6‐pentadecen‐2‐one (8.7 % and 4.9 %, respectively, sub‐group IIB).
Article type: Research Article
Keywords: Northwestern Algeria, Leaf essential oil, Chemical composition, Statistical analysis
Affiliations: Faculté des Sciences et Technologies Université Ahmed Zabana Cité Bourmadia 48000 Relizane Algérie; Laboratoire des Produits Naturels, Département de Biologie Université Abou Bekr Belkaïd, Imama 13000 Tlemcen Algérie; Laboratoire d’Ecologie et Gestion des Ecosystèmes Naturels, Département d’Ecologie et Environnement Université Abou Bekr Belkaïd, Imama 13000 Tlemcen Algérie.; Université de Corse-CNRS, UMR 6134 SPE Route des Sanguinaires 20000 Ajaccio France
License: © 2024 The Author(s). Chemistry & Biodiversity published by Wiley-VHCA AG CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202401584 | PubMed: 39373014 | PMC: PMC11826294
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (467 KB)
Introduction
Juniperus oxycedrus L. (Cupressaceae family) chiefly a Mediterranean species, is an evergreen dioecious shrub. In the flora of Algeria, it is recognized three subspecies, including: subsp. badia (H. Gay) Deb., subsp. macrocarpa (S. & Sm.) Ball. and subsp. oxycedrus [Syn. subsp. rufescens (Link) Deb.].[ref. cbdv202401584-bib-0001, ref. cbdv202401584-bib-0002] J. oxycedrus commonly known in Algeria as “Taga”,ref. cbdv202401584-bib-0001 grows from North Africa (Morocco, Algeria and Tunisia), into Portugal, Spain, France, Italy, Greece, the Balkans, Turkey and eastward into Iran.ref. cbdv202401584-bib-0003
Cade oil (empyreumatic oil) prepared by the destructive distillation of dry wood of J. oxycedrus, was largely employed as an antiparasitic and antiseptic for skin diseases, psoriasis and chronic eczema.ref. cbdv202401584-bib-0004 This plant is widely used as traditional medicine. It is recommended against diabetes, diarrhea, dermatitis, rheumatism and eye infections.[ref. cbdv202401584-bib-0005, ref. cbdv202401584-bib-0006, ref. cbdv202401584-bib-0007]
Oils of J. oxycedrus have many biological activities such as antibacterial,[ref. cbdv202401584-bib-0008, ref. cbdv202401584-bib-0009, ref. cbdv202401584-bib-0010, ref. cbdv202401584-bib-0011] antifungal,ref. cbdv202401584-bib-0008 antibiofilm,ref. cbdv202401584-bib-0011 anti‐quorum sensing,ref. cbdv202401584-bib-0012 antioxidant,[ref. cbdv202401584-bib-0012, ref. cbdv202401584-bib-0013, ref. cbdv202401584-bib-0014] anti‐tyrosinase and anti‐cholinesterase.ref. cbdv202401584-bib-0013
The composition of essential oil isolated from berries (fruits) of Algerian J. oxycedrus has been investigated. Most studies reported on the composition of a unique oil sample and concerned the subsp. oxycedrus.[ref. cbdv202401584-bib-0012, ref. cbdv202401584-bib-0015, ref. cbdv202401584-bib-0016, ref. cbdv202401584-bib-0017] α‐Pinene (56.4–88.4 %) was the major component in three out of four reported compositions; followed by myrcene (6.7–10.4 %) and germacrene D (14.2 %) or α‐amorphene (9.0 %). The chemical composition of the fourth sample was dominated by germacrene D (38.3 %) and α‐pinene (17.7 %). More informative was the work recently published by our groups. Indeed, the chemical composition of 33 oil samples isolated from the berries of individual trees of two subspecies of J. oxycedrus L., collected in Northwestern Algeria, was investigated and submitted to statistical analysis. Groups and sub‐groups were distinguished with respect to their contents of α‐pinene, germacrene D, myrcene and manoyl oxide.ref. cbdv202401584-bib-0018
In contrast, the composition of essential oil isolated from aerial parts (generally leaves and eventually stems) of Algerian J. oxycedrus subsp. oxycedrus and/or subsp. macrocarpa has been widely investigated although every report concerned only one oil sample.[ref. cbdv202401584-bib-0009, ref. cbdv202401584-bib-0011, ref. cbdv202401584-bib-0012, ref. cbdv202401584-bib-0013, ref. cbdv202401584-bib-0014, ref. cbdv202401584-bib-0019, ref. cbdv202401584-bib-0020, ref. cbdv202401584-bib-0021, ref. cbdv202401584-bib-0022, ref. cbdv202401584-bib-0023, ref. cbdv202401584-bib-0024, ref. cbdv202401584-bib-0025, ref. cbdv202401584-bib-0026]
In six oil samples out of nine belonging to the oxycedrus subspecies, α‐pinene was the major component although its content varied drastically from 23.6 % to 56.1 %. Other components present at appreciable content were very different from sample to sample: β‐phellandrene,ref. cbdv202401584-bib-0011 sabinene and terpinen‐4‐ol,ref. cbdv202401584-bib-0014 δ‐3‐carene,ref. cbdv202401584-bib-0019 germacrene D,ref. cbdv202401584-bib-0020 bulnesol,ref. cbdv202401584-bib-0013 epi‐manoyl oxide and (E,E)‐farnesol,ref. cbdv202401584-bib-0014 abietadiene.[ref. cbdv202401584-bib-0012, ref. cbdv202401584-bib-0013] The last three oil samples of the oxycedrus subspecies displayed very different compositions. The monoterpene‐rich oil sample from Mostaganem contained mainly terpinen‐4‐ol (17.48 %), α‐pinene (14.85 %) and sabinene (14.93 %).ref. cbdv202401584-bib-0021 One oil sample from Tiaret Forest displayed germacrene D (27.41 %) δ‐3‐carene (24.73 %) and myrcene (13.85 %) as major components.ref. cbdv202401584-bib-0022 In an oil sample from Tébessa various oxygenated pinane derivatives were detected, trans‐pinocarveol (7.0 %), cis‐verbenol (6.3 %), pinocarvone (5.1 %).ref. cbdv202401584-bib-0023
Otherwise, α‐pinene (29.1 %), β‐pinene (17.6 %) and β‐copaene (19.3 %) were the major components of an essential oil isolated from leaves and fruits of J. oxycedrus ssp. oxycedrus harvested in Tiaret Forest.ref. cbdv202401584-bib-0024 In contrast, another oil sample isolated from leaves, stems and fruits harvested in the Aures contained mainly manoyl oxide (23.4 %) beside (Z)‐pentadec‐6‐en‐2‐one (12.6 %).ref. cbdv202401584-bib-0025
The composition of two oil samples of the subspecies macrocarpa has also been investigated. The composition of the former from El Kala) was dominated by germacrene D (21.3 %), followed by (Z,Z)‐farnesol (10.9 %).ref. cbdv202401584-bib-0026 The major component of the sample from Oum El Bouaghi was the uncommon 5‐(Z)‐tetradecen‐1‐yl acetate (12.9 %).ref. cbdv202401584-bib-0009
In a recent study, we evidenced a variability in the chemical composition of essential oils isolated from berries of J. oxycedrus (oxycedrus and macrocarpa subspecies) growing wild in provinces of Tlemcen and Aïn Témouchent, Northwestern Algeria.ref. cbdv202401584-bib-0018 Statistical analyses suggested the existence of two groups which were distinguished on the basis of α‐pinene content. According to the amounts of α‐pinene, germacrene D and myrcene, as well as manoyl oxide, group II was divided into two sub‐groups. The chemical composition of the essential oils belonging to subsp. oxycedrus was dominated by monoterpenes. The main constituents were α‐pinene (23.7–63.2 %) with appreciable contents of germacrene D (4.5–25.4 %). In contrast, germacrene D (4.4–30.0 %) was the major component in oil samples of subsp. macrocarpa, accompanied by a lower content of α‐pinene (10.0–24.3 %) and a significant amount of manoyl oxide (4.3–9.9 %).ref. cbdv202401584-bib-0018
The chemical variability of J. oxycedrus essential oil has been evidenced in a few countries and in a Mediterranean island:
- For instance, regarding essential oils isolated from 60 individual trees growing in Tunisia, the content of the major components varied substantially from sample to sample: α‐pinene (24.32–58.03 %), geranyl acetone (1.96–8.80 %), 13‐epi‐manoyl oxide (1.35–6.95 %). Hierarchical clustering and principal component analysis (PCA) allowed to establish four groups; one group being divided into two sub‐groups. The oils of the population from the continental site were clearly distinguished from those of the littoral localitiesref. cbdv202401584-bib-0027:
- The essential oil samples of J. oxycedrus collected among 20 populations across Bulgaria contained mainly α‐pinene, limonene, α‐curcumene, γ‐cadinene, δ‐cadinene, germacrene D, β‐caryophyllene, α‐caryophyllene, caryophyllene oxide and manoyl oxide, and they were grouped in eight chemotypes using advanced statistical methodsref. cbdv202401584-bib-0028;
- The composition of 54 samples of leaf oil of Juniperus oxycedrus ssp. oxycedrus from Corsica was investigated. The main constituents were α‐pinene, β‐phellandrene and δ‐3‐carene. Principal component analysis, allowed the distinction of two compositions differentiated by the contents of α‐pinene, β‐phellandrene and δ‐3‐carene. In parallel, the chemical compositions of 18 leaf oil samples of J. oxycedrus ssp. macrocarpa have been investigated. The contents of the main components varied drastically from sample to sample: α‐pinene (28.7–76.4 %), δ‐3‐carene (up to 17.3 %), β‐phellandrene (up to 12.3 %), manoyl oxide (up to 8.1 %). Principal component analysis, as well as k‐means partition suggested a unique group, with few atypical samples. In the main group, α‐pinene (43.3–63.8 %) was accompanied by β‐phellandrene (up to 11.9 %), δ‐3‐carene (up to 7.5 %) and manoyl oxide (up to 4.5 %).ref. cbdv202401584-bib-0030
Obviously, it appears from literature data that essential oil samples isolated from leaves (or aerial parts) of Juniperus oxycedrus displayed substantially different compositions. So, the aim of this study was to characterize the yield and the chemical composition of the essential oil from the leaves of Juniperus oxycedrus subsp. oxycedrus and subsp. macrocarpa, growing wild in Tlemcen and Aïn Témouchent provinces, Northwestern Algeria.
Firstly, we will report on the detailed analysis of eight essential oil samples, by combination of chromatographic and spectroscopic techniques. Secondly, the compositions of 37 leaf oil samples isolated from individual plants, were submitted to statistical analysis in order to evidence homogeneity or an eventual chemical variability.
Results and Discussion
Leaves of 37 individual plants from Juniperus oxycedrus have been harvested in two provinces of Northwestern Algeria, Tlemcen (three locations, Ouled Mimoun, Terny and Aïn Fezza) and Aïn Témouchent (one location, Béni Saf) (Figure 1); two subspecies, subsp. oxycedrus (28 individuals) and subsp. macrocarpa (9 individuals). Leaves of individual plants have been hydrodistilled using a Clevenger‐type apparatus, yielding colorless essential oil samples. We will report first on the yield of essential oil depending of the subspecies and the location of harvest, then, on the detailed analysis of eight oil samples, selected on the basis of their chromatographic profiles, by GC in combination with retention indices on two chromatographic columns of different polarity, by GC/MS and by 13C NMR, without isolation of the individual components;[ref. cbdv202401584-bib-0031, ref. cbdv202401584-bib-0032] lastly, on the statistical analysis of the 37 compositions.
Essential Oil Yield
The yields of J. oxycedrus leaf essential oil (LEO) (w/w vs. dry material) varied substantially from sample to sample, even within subspecies, ranging between 0.01 to 0.20 % for the oxycedrus subspecies (28 samples) and from 0.02 % to 0.16 % for the macrocarpa subspecies (9 samples) (Table 1). The highest yields (0.11±0.07 %) were observed for the plants collected in Terny and the lowest (0.06±0.03 %) were observed for the plants harvested in Aïn Fezza, while similar yield mean values (0.09 % and 0.08 %) have been calculated for the plants harvested in Ouled Mimoun (oxycedrus subspecies) and Béni Saf (macrocarpa subspecies), respectively.
Table 1: Yields (%) of the essential oils isolated from leaves of two subspecies of Juniperus oxycedrus.
| Locations of harvest | samples | Mean±SD[a] (%) | Min[b] | Max[c] | Subspecies |
|---|---|---|---|---|---|
| Ouled Mimoun | 9 | 0.09±0.05 | 0.04 | 0.19 | oxycedrus |
| Terny | 9 | 0.11±0.07 | 0.01 | 0.20 | oxycedrus |
| Aïn Fezza | 10 | 0.06±0.03 | 0.03 | 0.12 | oxycedrus |
| Béni Saf | 9 | 0.08±0.04 | 0.02 | 0.16 | macrocarpa |
[a] Standard deviation (SD); [b] Minimum (Min); [c] Maximum (Max)
Chemical Composition of Selected Oil Samples
Eight oil samples, six of the subspecies oxycedrus and two of the subspecies macrocarpa, selected on the basis of their chromatographic profile, were submitted to gas chromatography (GC), in combination with retention indices (RIs) on two columns of different polarity, gas chromatography mass spectroscopy (GC/MS) and 13C nuclear magnetic resonance (NMR), following a computerized method developed at University of Corsica.[ref. cbdv202401584-bib-0031, ref. cbdv202401584-bib-0032] The detailed analyses of those eight samples were reported in Table 2. In total, 88 compounds were identified, accounting for 84.4–99.5 % of the whole compositions, which were largely dominated by monoterpene hydrocarbons (68.6–83.9 %), sample S15 excepted (36.2 %). In seven oil samples, α‐pinene (43.7–72.1 %) was by far the major component followed by δ‐3‐carene (up to 12.8 %) and β‐phellandrene (up to 11.0 %). Myrcene (1.4–7.8 %), β‐pinene (0.9–4.1 %), sabinene (0.1–3.4 %) and p‐cymene (0.2–3.1 %) were found to be the other monoterpene hydrocarbons present at appreciable contents. Germacrene D (0.9–7.2 %) was the only sesquiterpene hydrocarbon present in a significant amount, beside γ‐cadinene (0.3–1.8 %), (E)‐β‐caryophyllene (0.2–1.1 %), δ‐cadinene (0.1–1.0 %) and α‐humulene (0.0–1.0 %). The occurrence of (Z)‐6‐pentadecen‐2‐one (0.1–1.8 %) could be highlighted. Conversely, the main components of sample S15 were α‐pinene (28.1 %) and germacrene D (15.7 %), followed by oxygenated sesquiterpenes, namely: 2,3‐dihydrofarnesol (3.6 % vs. 0.0–1.1 %), (2E,6E)‐farnesol (2.5 % vs. 0.3–2.2 %) and diterpenes, namely abietadiene (5.4 % vs. 0.2–1.8 %) and manoyl oxide (3.1 % vs. 0.0–0.9 %).
Table 2: Chemical composition of LEO of two subspecies of J. oxycedrus.
| Compounds[a] | RIa[b] | RIp[b] | RIaL[c] | RIpL[c] | S24 [d] | S19 [d] | S5[d] | S10[d] | S1[d] | S15[d] | S29 [e] | S33[e] | Identification | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Hexanal | 772 | 1081 | 777 | 1082 | 0.2 | 0.2 | 0.6 | 0.1 | 1.3 | 0.4 | 0.2 | ‐ | RI, MS, 13C NMR |
| 2 | (E)‐Hex‐2‐enal | 823 | 1221 | 827 | 1216 | 0.2 | 0.1 | 0.3 | 0.1 | 1.3 | 0.3 | 0.5 | 0.2 | RI, MS |
| 3 | Hexan‐1‐ol | 846 | 1347 | 855 | 1351 | Tr | ‐ | 0.1 | ‐ | 0.4 | ‐ | 0.2 | 0.3 | RI, MS |
| 4 | Tricyclene | 919 | nd | 922 | 1012 | 0.2 | 0.2 | 0.3 | 0.2 | 0.1 | 0.1 | 0.2 | 0.1 | RI, MS |
| 5 | α‐Thujene | 921 | 1014 | 926 | 1027 | 0.1 | Tr | 0.1 | Tr | Tr | 0.1 | 0.8 | 0.5 | RI, MS |
| 6 | α‐Pinene | 930 | 1014 | 935 | 1025 | 72.1 | 62.3 | 61.7 | 57.4 | 48.8 | 28.1 | 67.0 | 43.7 | RI, MS, 13C NMR |
| 7 | α‐Fenchene | 940 | 1050 | 945 | 1061 | 0.1 | Tr | 0.7 | Tr | Tr | 0.1 | 0.1 | 0.5 | RI, MS, 13C NMR |
| 8 | Camphene | 942 | 1062 | 947 | 1069 | 0.4 | 0.3 | 0.3 | 0.3 | 0.4 | 0.1 | 0.3 | 0.2 | RI, MS |
| 9 | Verbenene | 945 | 1124 | 946 | 1124 | 0.2 | 0.3 | 0.5 | 0.2 | 0.2 | 0.1 | 0.1 | 0.3 | RI, MS, 13C NMR |
| 10 | 1‐Octen‐3‐ol | 959 | 1444 | 966 | 1444 | 0.2 | 0.2 | 0.7 | 0.1 | 0.1 | 0.2 | 0.3 | 2.0 | RI, MS, 13C NMR |
| 11 | Sabinene | 963 | 1119 | 968 | 1122 | 0.3 | 0.2 | 0.2 | 0.2 | 0.1 | 0.3 | 2.7 | 3.4 | RI, MS, 13C NMR |
| 12 | β‐Pinene | 969 | 1109 | 973 | 1110 | 1.7 | 1.6 | 1.4 | 2.3 | 4.1 | 0.8 | 0.9 | 1.2 | RI, MS, 13C NMR |
| 13 | Myrcene | 979 | 1158 | 983 | 1161 | 1.5 | 1.4 | 1.4 | 2.3 | 3.7 | 0.7 | 7.8 | 2.1 | RI, MS, 13C NMR |
| 14 | 2‐Carene | 994 | 1128 | 998 | 1134 | 0.1 | 0.4 | 0.2 | 0.6 | 0.5 | 0.1 | 0.1 | 0.1 | RI, SM, 13C NMR |
| 15 | α‐Phellandrene | 996 | 1162 | 999 | 1168 | Tr | ‐ | 0.3 | 0.4 | 0.7 | 0.1 | ‐ | 0.1 | RI, MS, 13C NMR |
| 16 | δ‐3‐Carene | 1004 | 1145 | 1007 | 1147 | 0.1 | Tr | 8.2 | ‐ | Tr | 3.0 | 0.6 | 12.8 | RI, MS, 13C NMR |
| 17 | α‐Terpinene | 1008 | 1177 | 1011 | 1178 | Tr | ‐ | Tr | ‐ | ‐ | 0.1 | 0.2 | 0.3 | RI, MS |
| 18 | p‐Cymene | 1011 | 1268 | 1015 | 1270 | 0.6 | 0.2 | 1.3 | 1.6 | 3.1 | 0.9 | 0.8 | 1.5 | RI, MS, 13C NMR |
| 19 | Limonene* | 1020 | 1198 | 1024 | 1198 | 1.0 | 1.3 | 1.0 | 1.7 | 1.9 | 0.5 | 1.4 | 1.1 | RI, MS, 13C NMR |
| 20 | β‐Phellandrene* | 1020 | 1208 | 1021 | 1209 | 1.6 | 0.3 | 3.5 | 6.3 | 11.0 | 0.6 | 0.1 | 1.2 | RI, MS, 13C NMR |
| 21 | γ‐Terpinene | 1047 | 1242 | 1050 | 1245 | ‐ | ‐ | Tr | ‐ | ‐ | 0.1 | 0.4 | 0.5 | RI, MS |
| 22 | 1‐Octanol | 1051 | 1552 | 1057 | 1552 | 0.1 | 0.1 | Tr | 0.1 | Tr | 0.1 | 0.1 | 0.5 | RI, MS, 13C NMR |
| 23 | p‐Cymenene | 1071 | 1435 | 1074 | 1438 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | Tr | 0.1 | RI, MS |
| 24 | Terpinolene | 1077 | 1280 | 1079 | 1282 | ‐ | ‐ | 0.3 | 0.3 | 0.2 | 0.3 | 0.4 | 1.4 | RI, MS, 13C NMR |
| 25 | Linalool* | 1082 | 1542 | 1086 | 1543 | 0.5 | 0.3 | 0.2 | 0.1 | 0.3 | 0.3 | Tr | 0.2 | RI, MS |
| 26 | α‐Pinene oxide* | 1082 | 1374 | 1085 | 1364 | Tr | Tr | ‐ | Tr | Tr | Tr | 0.1 | ‐ | RI, MS |
| 27 | α‐Campholenal | 1103 | 1487 | 1107 | 1496 | 0.2 | 0.4 | 0.6 | 0.2 | 0.2 | 0.2 | 0.2 | 0.3 | RI, MS, 13C NMR |
| 28 | Fenchol* | 1103 | 1583 | 1101 | 1570 | 0.2 | 0.2 | 0.1 | 0.2 | 0.1 | 0.1 | 0.2 | 0.3 | RI, MS |
| 29 | Camphor | 1118 | 1512 | 1125 | 1515 | Tr | 0.1 | Tr | Tr | 0.1 | ‐ | ‐ | 0.1 | RI, MS |
| 30 | trans‐Pinocarveol | 1121 | 1650 | 1126 | 1661 | 0.5 | 1.3 | 1.2 | 0.4 | 0.5 | 0.3 | 0.3 | 0.7 | RI, MS, 13C NMR |
| 31 | cis‐Verbenol | 1123 | 1650 | 1126 | 1660 | ‐ | 0.3 | 0.1 | ‐ | ‐ | 0.1 | ‐ | 0.3 | RI, MS |
| 32 | trans‐Verbenol | 1127 | 1673 | 1134 | 1680 | 0.3 | 1.2 | 0.7 | 0.3 | 0.2 | 0.2 | 0.2 | 1.3 | RI, MS, 13C NMR |
| 33 | p‐Menth‐3‐en‐8‐ol | 1133 | 1597 | 1140f | 1600f | ‐ | ‐ | Tr | ‐ | ‐ | Tr | ‐ | ‐ | RI, MS |
| 34 | trans‐Pinocamphone* | 1137 | 1511 | 1144 | 1523 | 0.1 | 0.6 | 0.5 | Tr | 0.7 | 0.1 | 0.3 | 0.4 | RI, MS, 13C NMR |
| 35 | Pinocarvone* | 1137 | 1564 | 1140 | 1576 | 0.1 | 0.3 | 0.4 | 0.2 | 0.2 | 0.1 | Tr | Tr | RI, MS |
| 36 | p‐Mentha‐1,5‐dien‐8‐ol | 1144 | 1721 | 1149 | 1725 | 0.1 | 0.1 | 0.3 | 0.1 | 0.1 | 0.1 | 0.1 | 0.4 | RI, MS, |
| 37 | Borneol | 1147 | 1695 | 1153 | 1700 | ‐ | ‐ | 0.1 | Tr | 0.1 | ‐ | 0.1 | 0.2 | RI, MS |
| 38 | Cryptone | 1154 | 1661 | 1157 | 1675 | 0.1 | ‐ | 0.4 | 0.4 | 0.5 | Tr | ‐ | 0.2 | RI, MS |
| 39 | p‐Cymene‐8‐ol | 1158 | 1844 | 1165 | 1848 | ‐ | ‐ | 0.4 | 0.2 | 0.1 | 0.1 | 0.1 | 0.4 | RI, MS |
| 40 | Terpinen‐4‐ol | 1160 | 1597 | 1165 | 1601 | 0.2 | 0.2 | 0.1 | 0.1 | Tr | 0.2 | 1.4 | 1.8 | RI, MS, 13C NMR |
| 41 | Myrtenal | 1168 | 1623 | 1171 | 1632 | 0.1 | 0.3 | 0.4 | 0.1 | 0.1 | 0.1 | 0.1 | 0.3 | RI, MS, |
| 42 | α‐Terpineol | 1170 | 1692 | 1176 | 1694 | 0.2 | 0.1 | 0.2 | 0.1 | 0.3 | 0.2 | 0.5 | 0.4 | RI, MS, 13C NMR |
| 43 | Myrtenol | 1177 | 1786 | 1182 | 1790 | 0.1 | 0.3 | 0.3 | 0.2 | 0.1 | 0.1 | 0.1 | 0.3 | RI, MS |
| 44 | Verbenone | 1183 | 1715 | 1184 | 1721 | Tr | ‐ | 0.1 | ‐ | 0.2 | ‐ | Tr | 0.1 | RI, MS |
| 45 | trans‐Carveol | 1195 | 1829 | 1201 | 1836 | 0.1 | 0.3 | 0.3 | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | RI, MS |
| 46 | cis‐Carveol | 1212 | 1865 | 1206 | 1854 | 0.1 | 0.1 | 0.1 | 0.3 | 0.2 | Tr | 0.1 | 0.1 | RI, MS |
| 47 | Bornyl acetate | 1267 | 1575 | 1270 | 1579 | ‐ | ‐ | Tr | ‐ | 0.3 | 0.3 | 0.1 | 0.3 | RI, MS |
| 48 | α‐Terpinyl acetate | 1329 | 1680 | 1333 | 1695 | 0.1 | 0.1 | 0.1 | 0.1 | 0.5 | 0.1 | 0.1 | 0.1 | RI, MS, 13C NMR |
| 49 | α‐Cubebene | 1345 | 1450 | 1352 | 1460 | 0.1 | ‐ | Tr | Tr | Tr | 0.1 | 0.1 | 0.1 | RI, MS |
| 50 | α‐Ylangene | 1367 | 1476 | 1370 | 1484 | ‐ | ‐ | ‐ | ‐ | Tr | 0.1 | Tr | ‐ | RI, MS |
| 51 | α‐Copaene | 1371 | 1484 | 1376 | 1491 | 0.1 | ‐ | ‐ | 0.1 | Tr | 0.1 | 0.1 | Tr | RI, MS |
| 52 | β‐Elemene | 1384 | 1583 | 1388 | 1591 | Tr | 0.1 | Tr | 0.2 | 0.1 | 0.2 | 0.1 | 0.2 | RI, MS |
| 53 | (E)‐β‐Caryophyllene | 1413 | 1589 | 1419 | 1599 | 0.6 | 0.9 | 0.2 | 1.1 | 0.6 | 1.7 | 0.4 | 0.2 | RI, MS, 13C NMR |
| 54 | β‐Coapene | 1422 | 1583 | 1427 | 1580 | 0.1 | 0.1 | Tr | 0.1 | 0.1 | 0.2 | 0.1 | 0.1 | RI, MS |
| 55 | (E)‐β‐Farnesene | 1443 | 1661 | 1449 | 1664 | ‐ | ‐ | 0.1 | 0.1 | 0.1 | ‐ | 0.1 | ‐ | RI, MS |
| 56 | α‐Humulene | 1446 | 1661 | 1449 | 1667 | 0.6 | 0.9 | ‐ | 1.0 | 0.4 | 1.4 | 0.4 | 0.1 | RI, MS, 13C NMR |
| 57 | γ‐Muurolene | 1467 | 1679 | 1473 | 1690 | 0.5 | 0.4 | 0.1 | 0.7 | 0.3 | 0.7 | 0.5 | 0.2 | RI, MS, 13C NMR |
| 58 | Germacrene D | 1472 | 1701 | 1476 | 1708 | 2.2 | 5.7 | 0.9 | 7.2 | 2.1 | 15.7 | 4.2 | 4.1 | RI, MS, 13C NMR |
| 59 | 2‐Tridecanone | 1471 | 1801 | 1479 | 1808 | 0.6 | 0.4 | 0.2 | 0.2 | 0.2 | ‐ | ‐ | 0.1 | RI, MS, 13C NMR |
| 60 | β‐Selinene | 1477 | 1710 | 1481 | 1717 | Tr | ‐ | ‐ | ‐ | Tr | Tr | Tr | Tr | RI, MS |
| 61 | α‐Muurolene | 1489 | 1715 | 1491 | 1723 | 0.2 | 0.3 | 0.1 | ‐ | 0.2 | 0.5 | 0.2 | 0.2 | RI, MS |
| 62 | β‐Bisabolene | 1496 | 1719 | 1500 | 1728 | Tr | ‐ | ‐ | Tr | 0.1 | 0.2 | ‐ | 0.1 | RI, MS |
| 63 | γ‐Cadinene | 1501 | 1749 | 1506 | 1763 | 0.3 | 0.5 | 0.5 | 1.8 | 1.1 | 0.6 | 0.3 | 0.3 | RI, MS, 13C NMR |
| 64 | Calamenene[g] | 1505 | 1824 | 1510 | 1835 | 0.1 | Tr | ‐ | 0.1 | Tr | 0.1 | Tr | Tr | RI, MS |
| 65 | δ‐Cadinene | 1510 | 1749 | 1514 | 1756 | 0.7 | 0.6 | 0.1 | 0.7 | 0.3 | 1.5 | 0.8 | 1.0 | RI, MS, 13C NMR |
| 66 | α‐Calacorene | 1523 | 1907 | 1530 | 1921 | ‐ | ‐ | ‐ | ‐ | Tr | Tr | Tr | 0.1 | RI, MS |
| 67 | α‐Cadinene | 1526 | 1783 | 1527 | 1769 | Tr | Tr | Tr | 0.1 | 0.1 | Tr | Tr | 0.1 | RI, MS |
| 68 | β‐Elemol | 1530 | 2071 | 1537 | 2088 | Tr | ‐ | ‐ | ‐ | ‐ | 0.1 | ‐ | 0.4 | RI, MS |
| 69 | Salviadienol | 1534 | 2103 | 1545[h] | 2110[h] | 0.3 | 0.6 | 0.3 | 0.3 | 0.3 | 0.5 | 0.3 | 0.2 | RI, MS |
| 70 | (E)‐Nerolidol | 1546 | 2035 | 1550 | 2036 | ‐ | ‐ | ‐ | ‐ | Tr | ‐ | ‐ | 0.2 | RI, MS |
| 71 | Caryophyllene oxide | 1566 | 1972 | 1570 | 1986 | 0.3 | 0.6 | 0.3 | 0.2 | 0.3 | 0.4 | 0.1 | Tr | RI, MS, 13C NMR |
| 72 | Salvial‐4(14)‐en‐1‐one | 1573 | 1997 | 1585 | 2036 | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | RI, MS |
| 73 | cis‐Guai‐6‐en‐10‐ol | 1590 | 1988 | 1575[i] | 2000[i] | 0.4 | 0.9 | 0.3 | 0.4 | 0.4 | 0.7 | 0.3 | 0.2 | RI, MS, 13C NMR |
| 74 | 1,10‐di‐epi‐Cubenol | 1610 | 2051 | 1606 | 2074 | 0.1 | 0.1 | 0.1 | 0.7 | 0.8 | 0.1 | Tr | 0.1 | RI, MS |
| 75 | γ‐Eudesmol | 1617 | 2163 | 1617 | 2176 | Tr | 0.2 | 0.1 | Tr | 0.1 | 0.1 | 0.1 | Tr | RI, MS |
| 76 | τ‐Muurolol* | 1620 | 2177 | 1631 | 2186 | 0.1 | 0.2 | Tr | 0.2 | Tr | 0.3 | 0.1 | 0.2 | RI, MS |
| 77 | τ‐Cadinol* | 1620 | 2160 | 1626 | 2170 | Tr | 0.1 | 0.1 | Tr | 0.2 | 0.1 | 0.1 | 0.1 | RI, MS |
| 78 | β‐Eudesmol | 1631 | 2219 | 1634 | 2223 | ‐ | 0.4 | 0.1 | 0.1 | 0.1 | 0.6 | 0.2 | 0.1 | RI, MS |
| 79 | α‐Cadinol | 1633 | 2221 | 1640 | 2227 | 0.2 | 0.4 | Tr | Tr | 0.1 | 0.4 | 0.3 | 0.7 | RI, MS, 13C NMR |
| 80 | (Z)‐6‐Pentadecen‐2‐one | 1647 | 2027 | 1647[j] | 2030[j] | 1.7 | 1.8 | 0.9 | 1.6 | 1.3 | 0.1 | 0.1 | 0.5 | RI, MS, 13C NMR |
| 81 | Eudesma‐4(15),7‐dien‐1‐ol | 1664 | 2345 | 1676 | 2371 | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | 0.3 | 0.2 | 0.1 | RI, MS |
| 82 | 2,3‐Dihydrofarnesol | 1668 | 2260 | 1668[k] | 2262[l] | 0.5 | 1.1 | 0.3 | 0.3 | 0.6 | 3.6 | ‐ | 0.3 | RI, MS, 13C NMR |
| 83 | (2Z,6E)‐Farnesal | 1685 | 2310 | 1688[m] | 2219[m] | 0.1 | 0.2 | 0.1 | 0.2 | 0.1 | 0.6 | ‐ | 0.1 | RI, MS, 13C NMR |
| 84 | (2E,6E)‐Farnesol | 1697 | 2355 | 1710 | 2366 | 0.4 | 2.2 | 0.3 | 0.5 | 0.9 | 2.5 | 0.5 | 0.6 | RI, MS, 13C NMR |
| 85 | (2E,6E)‐Farnesal | 1711 | 2257 | 1715[n] | 2264[n] | 0.2 | 0.4 | 0.1 | 0.3 | 0.1 | 0.9 | ‐ | 0.2 | RI, MS, 13C NMR |
| 86 | Manoyl oxide | 1976 | 2329 | 1990 | 2376 | ‐ | 0.3 | 0.9 | 0.9 | 0.1 | 3.1 | 0.2 | 0.3 | RI, MS, 13C NMR |
| 87 | Abietatriene | 2028 | 2478 | 2033 | 2506 | 0.4 | 0.6 | 0.3 | 0.3 | 0.9 | 1.5 | 0.1 | 0.5 | RI, MS, 13C NMR |
| 88 | Abietadiene | 2064 | 2441 | 2062 | 2450 | 0.4 | 0.7 | 0.3 | 0.5 | 0.9 | 5.4 | 0.2 | 1.8 | RI, MS, 13C NMR |
| Monoterpene hydrocarbons | 80.1 | 68.6 | 81.5 | 73.9 | 74.9 | 36.2 | 83.9 | 71.1 | ||||||
| Oxygenated monoterpenes | 3.0 | 6.2 | 6.6 | 3.1 | 4.9 | 2.7 | 4.1 | 8.4 | ||||||
| Sesquiterpene hydrocarbons | 6.1 | 9.9 | 2.2 | 13.4 | 5.7 | 23.1 | 7.3 | 6.9 | ||||||
| Oxygenated sesquiterpenes | 2.8 | 7.7 | 2.3 | 3.4 | 4.3 | 11.3 | 2.3 | 3.6 | ||||||
| Diterpene hydrocarbons | 0.8 | 1.3 | 0.6 | 0.8 | 1.8 | 6.9 | 0.3 | 2.3 | ||||||
| Oxygenated diterpenes | 0.0 | 0.3 | 0.9 | 0.9 | 0.1 | 3.1 | 0.2 | 0.3 | ||||||
| Others | 2.4 | 2.4 | 2.6 | 2.0 | 4.4 | 1.1 | 1.4 | 3.5 | ||||||
| Total (%) | 95.2 | 96.4 | 96.7 | 97.5 | 96.1 | 84.4 | 99.5 | 96.1 |
[a] Components have been listed following their order of elution on apolar column (BP‐1); Percentages on apolar column, except those with an asterisk, % on polar column (BP‐20); [b] RIa, RIp: Retention indices on apolar and polar columns, respectively; [c] RIaL, RIpL: Retention indices from literature, on apolar and polar columns, respectively; Babushok et al., [33] otherwise stated; [d] Subsp. oxycedrus; [e] Subsp. macrocarpa; 13C NMR: Compound identified by 13C NMR, at least in one oil sample; nd: Not determined; Tr: Traces; [f] ref [34]; [g] Isomer not determined; [ h] ref [35]; [i] ref [36]; [j] ref [29], [k] ref [37]; [l] ref [38]; [m] ref [39]; [n] ref [40]
Chemical Variability
Thirty‐seven essential oil samples were obtained by hydrodistillation of leaves collected from individual trees of J. oxycedrus in four locations of Northwestern of Algeria: Ouled Mimoun, Terny and Aïn Fezza (Tlemcen province, oxycedrus subspecies, 28 samples) and Béni Saf (Aïn Témouchent province, macrocarpa subspecies, 9 samples) (Figure 1).
The 37 compositions were subjected to statistical analysis in order to distinguish clusters. The combination of hierarchical clustering dendrogram (HCA, Figure 2), and principal component analysis (PCA; Figure 3), for which the two axes (F1 and F3) accounted for 85.7 % of the total variance of the population, suggested the existence of two principal groups, one of these being subdivided into two sub groups IIA and IIB.
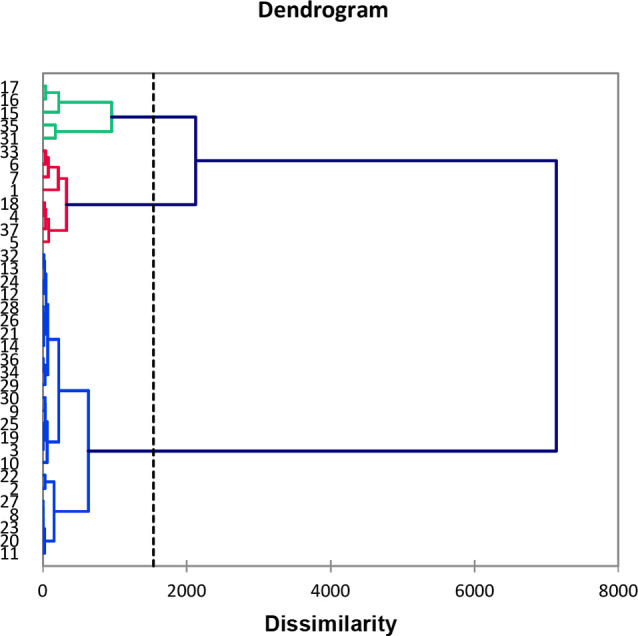
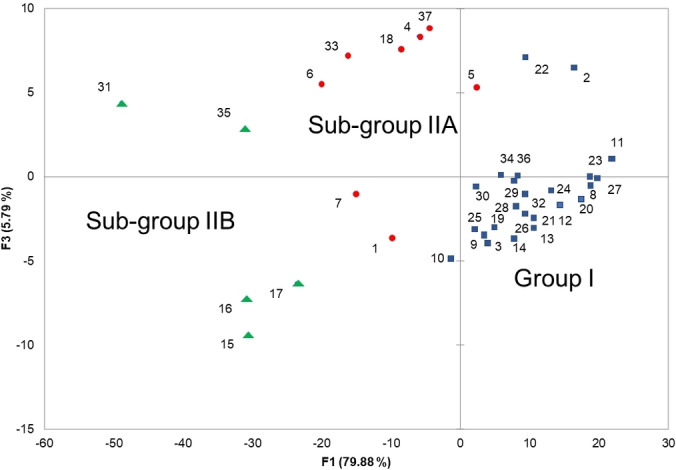
Group I, by far the most important group with 24 oil samples out of 37, differed from sub‐groups IIA and IIB, essentially by the content of α‐pinene (69.2 %, mean value, SD 6.24; vs. 49.7 %, SD 7.27 and 27.3 %, SD 7.89) (Table 3). Otherwise, regarding major components of group I, α‐pinene was followed by germacrene D (M=4.2 %, SD 2.39).
Table 3: Chemical variability of Juniperus oxycedrus LEO.
| Groups/samples | Group I/24 | Group II/13 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sub‐groups/samples | IIA/8 | IIB/5 | ||||||||||
| Components[a] | M[b] | SD[c] | Min[d] | Max[e] | M[b] | SD[c] | Min[d] | Max[e] | M[b] | SD[c] | Min[d] | Max[e] |
| α‐Thujene | 0.2 | 0.34 | Tr | 0.9 | 0.2 | 0.26 | Tr | 0.7 | 1.5 | 2.04 | 0.0 | 4.1 |
| α‐Pinene | 69.2 | 6.24 | 57.4 | 80.9 | 49.7 | 7.27 | 39.4 | 61.7 | 27.3 | 7.89 | 14.3 | 35.4 |
| Oct‐1‐en‐3‐ol | 0.2 | 0.14 | 0.0 | 0.5 | 0.8 | 0.59 | 0.1 | 2.0 | 0.3 | 0.17 | 0.2 | 0.6 |
| Sabinene | 0.9 | 1.40 | 0.2 | 5.3 | 1.0 | 1.52 | 0.1 | 3.5 | 8.7 | 11.94 | 0.1 | 25.2 |
| β‐Pinene | 1.6 | 0.42 | 0.9 | 2.4 | 1.7 | 0.98 | 1.2 | 4.1 | 0.9 | 0.10 | 0.8 | 1.1 |
| Myrcene | 1.9 | 1.33 | 0.7 | 7.8 | 1.9 | 0.83 | 1.1 | 3.7 | 1.3 | 0.74 | 0.7 | 2.4 |
| δ‐3‐Carene | 0.9 | 2.32 | 0.0 | 9.3 | 10.3 | 4.69 | Tr | 14.6 | 1.4 | 1.06 | 0.3 | 3.0 |
| α‐Terpinene | 0.1 | 0.18 | 0.0 | 0.7 | 0.1 | 0.11 | 0.0 | 0.3 | 0.7 | 0.99 | Tr | 2.2 |
| p‐Cymene | 0.5 | 0.41 | 0.1 | 1.6 | 1.5 | 1.00 | 0.1 | 3.1 | 2.8 | 2.82 | 0.6 | 4.6 |
| Limonene | 1.0 | 0.35 | Tr | 1.7 | 1.2 | 0.44 | 0.9 | 1.9 | 1.8 | 1.41 | 0.5 | 3.4 |
| β‐Phellandrene | 1.2 | 1.73 | 0.1 | 6.3 | 3.3 | 3.72 | 0.1 | 11.0 | 1.1 | 0.02 | 0.4 | 2.8 |
| γ‐Terpinene | 0.2 | 0.35 | 0.0 | 1.3 | 0.1 | 0.18 | 0.0 | 0.5 | 1.6 | 2.10 | 0.0 | 4.3 |
| Terpinen‐4‐ol | 0.5 | 0.78 | Tr | 2.8 | 0.6 | 1.00 | Tr | 2.6 | 4.2 | 5.42 | 0.2 | 11.6 |
| Germacrene D | 4.2 | 2.39 | Tr | 10.2 | 3.3 | 3.38 | 0.4 | 11.1 | 4.9 | 6.15 | 0.9 | 15.7 |
| 2‐Tridecanone | 0.2 | 0.18 | 0.0 | 0.6 | 0.3 | 0.20 | 0.0 | 0.6 | 1.4 | 1.58 | 0.0 | 3.7 |
| γ‐Cadinene | 0.6 | 0.46 | 0.1 | 1.8 | 0.8 | 0.62 | 0.2 | 1.9 | 0.9 | 0.97 | 0.1 | 2.4 |
| (E)‐Nerolidol | Tr | 0.01 | 0.0 | Tr | 0.5 | 1.30 | 0.0 | 3.7 | 0.0 | 0.01 | 0.0 | Tr |
| (Z)‐6‐Pentadecen‐2‐one | 1.1 | 0.76 | 0.1 | 2.8 | 1.7 | 0.93 | 0.5 | 2.9 | 4.9 | 6.09 | 0.1 | 12.0 |
| 2,3‐Dihydrofarnesol | 0.3 | 0.24 | 0.0 | 1.1 | 0.5 | 0.36 | 0.2 | 1.2 | 1.5 | 1.48 | 0.1 | 3.6 |
| (2E,6E)‐Farnesol | 0.8 | 0.66 | Tr | 2.5 | 0.7 | 0.46 | 0.2 | 1.7 | 1.9 | 0.73 | 1.1 | 2.7 |
| Manoyl oxide | 0.3 | 0.32 | 0.0 | 1.0 | 0.5 | 0.25 | 0.1 | 0.9 | 2.2 | 0.80 | 0.9 | 3.1 |
| Abietadiene | 0.5 | 0.43 | 0.0 | 1.9 | 1.0 | 0.81 | 0.1 | 2.5 | 2.4 | 2.06 | 0.7 | 5.4 |
[a] Percentages are given on apolar column (BP‐1); [b] Mean (M); [c] Standard deviation (SD); [d] Minimum content (Min); [e] Maximum content (Max); Tr: Traces.
Based on the contents of α‐pinene, sabinene, δ‐3‐carene, β‐phellandrene, terpinen‐4‐ol as well as 2‐tridecanone, (Z)‐6‐pentadecen‐2‐one, abietadiene and manoyl oxide, the samples of group II could be divided into two sub‐groups, IIA and IIB (Figure 3, Table 3).
Sub‐group IIA contained higher amount of α‐pinene (M=49.7 %, SD7.27) than sub‐group IIB (M=27.3 %, SD 7.89). Both sub‐groups differed mainly by their contents of sabinene (1.0 vs. 8.7 %), δ‐3‐carene (10.3 vs. 1.4 %) and (Z)‐6‐pentadecen‐2‐one (1.7 vs. 4.9 %).
It could be highlighted that all the samples from Aïn Fezza (S19‐S28, oxycedrus subspecies) belonged to group I. The nine samples from Ouled Mimoun (S1‐S9, oxycedrus subspecies) were equally reparteed between group I and sub‐group IIA. The nine samples from Terny (S10‐S18, oxycedrus subspecies) were reparteed between group I and sub‐group IIB, a last sample belonged to sub‐group IIA. Lastly, the nine samples from Béni Saf, macrocarpa subspecies, are mainly found in group I (5 samples) beside sub‐groups IIA (2 samples) and IIB (2 samples). The chemical variability appeared not related to the subspecies.
Comparing with previous results relative to the composition of J. oxycedrus leaf oil from Algeria, reported in the literature:
- an α‐pinene‐rich essential oil (92.2 %) was reported from National Park of Djurdjura, Bouira (Northern Central Algeria). This composition is clearly related to group I (highest content in α‐pinene)ref. cbdv202401584-bib-0017;
- an essential oil sample from Batna, (Northeastern Algeria) containing α‐pinene, 56.1 % may be associated to sub‐group IIA, although it differed by a high content of β‐phellandrene (17.9 %)ref. cbdv202401584-bib-0011;
- a composition from Tébessa, East Algeria with α‐pinene 42.2 % may be also associated to group IIA, although it differed by a high content of abietadiene (7.3 %)ref. cbdv202401584-bib-0012;
- an oil sample from Laghouat (Central North Algeria) displayed a content of α‐pinene (37.6 %) similar to those of some oil samples of sub‐group IIA, however with higher content of abietadiene (8.3 %) and bulnesol (7.2 %, scarcely reported in J. oxycedrus essential oils)ref. cbdv202401584-bib-0013;
- In parallel, a sample from Bouira (Central Algeria) contained α‐pinene (36.7 %) and it belongs to sub‐group IIA, taking into account the appreciable content of δ‐3‐carene (10.6 %)ref. cbdv202401584-bib-0019;
- Most in the South, at Founassa, near Aïn Sefra, an essential oil sample contained fair amount of α‐pinene (33.4 %). However, the high content of germacrene D (23.7 %) differentiated this sample from ours of sub‐group IIBref. cbdv202401584-bib-0020;
- A first sample from Mostaganem (Northwestern Algeria), that contained α‐pinene as major component (23.6 %), followed with sabinene (10.8 %) may be associated to sub‐group IIB, although it contained fair amounts of farnesol (8.5 %) and epi‐manoyl oxide (11.5 %), reported for the first time in J. oxycedrus essential oilsref. cbdv202401584-bib-0014;
- Another sample from Mostaganem, containing similar amounts of α‐pinene (14.9 %) and sabinene (14.9 %) could be also associated to sub‐group IIB, however, it differed by the content of terpinen‐4‐ol (17.5 %,)ref. cbdv202401584-bib-0021;
Lastly, the compositions of two other Algerian J. oxycedrus oil samples look atypical with respect to those observed in Tlemcen and Aïn Témouchent provinces, reported here and they cannot be introduced in any of the groups and sub‐groups. For instance, the composition of an oil sample from Tiaret Forest (Northwestern Algeria), was dominated by germacrene D (27.4 %), δ‐3‐carene (24.7 %), accompanied by myrcene (13.9 %) beside α‐pinene (4.1 %).ref. cbdv202401584-bib-0022 A sample from Tébessa (Eastern Algeria) contained mainly oxygenated pinane derivatives, trans‐pinocarveol (7.0 %), cis‐verbenol (6.3 %), pinocarvone (5.1 %), verbenone (3.6 %) while α‐pinene accounted for 0.2 % only.ref. cbdv202401584-bib-0023
Concerning leaf oils from the subspecies macrocarpa, they could not associate with our groups and sub‐groups. Indeed, the first one from El Kala, contained mainly germacrene D (21.3 %), (Z,Z)‐farnesol (10.9 %) and 8,13‐epoxy‐14,15‐dinorlabdane (8.8 %).ref. cbdv202401584-bib-0026 The second sample from Oum El Bouaghi, exhibited a very unusual composition dominated by (Z)‐5‐tetradecen‐1‐yl acetate (12.9 %) and γ‐muurolene (9.1 %).ref. cbdv202401584-bib-0009
Comparing the present results with previous studies that included statistical analysis, the compositions of essential oil samples from group I (α‐pinene M=69.2 %, SD 6.24) and sub‐group IIA (α‐pinene M=49.7 %, SD 7.27) resembled to those of essential oils from Corsica (subspecies oxycedrus) with α‐pinene M=73.3 %, SD 5.8 (group I) and M=56.1 %, SD 5.7 (group II).ref. cbdv202401584-bib-0029 Otherwise, we noticed that the nine samples from Béni Saf, macrocarpa subspecies, are present in the three groups and sub‐groups. Similarly, the chemical compositions of 18 leaf oil samples of J. oxycedrus ssp. macrocarpa from Corsica, varied substantially from sample to sample.ref. cbdv202401584-bib-0030
Regarding the compositions of essential oils isolated from individual trees growing in Tunisia, α‐pinene was always the major component although its content varied substantially (24.32–58.03 %) as observed in the present study (14.3–80.9 %). α‐Pinene was accompanied by various compounds not present in our samples, β‐bourbonene (up to 14.28 %), 10‐epi‐α‐muurolol (up to 11.13 %), geranyl acetone (up to 8.80 %), 13‐epi‐manoyl oxide (up to 6.95 %). Four groups were distinguished, however, the mean value of every component in each group was not given.ref. cbdv202401584-bib-0027
The essential oil samples of J. oxycedrus collected among 20 populations across Bulgaria contained various components with low to medium content: α‐pinene (6.5–24.8 %, much lower content than in our samples), manoyl oxide (4.9–18.4 %), limonene (1.8–16.7 %), caryophyllene oxide (0.7–13.2 %), β‐caryophyllene (0.1–12.5 %), δ‐cadinene (0.4–11.9 %), germacrene D (0.0–11.7 %). Samples were grouped in eight chemotypes (however, the mean percentage of every compound with respect to the chemotype, was not given).ref. cbdv202401584-bib-0028
Conclusions
Thirty‐seven essential oil samples have been isolated from leaves of J. oxycedrus (subspecies oxycedrus and macrocarpa) growing wild in Tlemcen and Aïn Témouchent provinces (Northwestern Algeria). Analysis by combination of chromatographic and spectroscopic techniques of eight selected oil samples allowed the identification of 88 components that accounted for 84.4–99.5 % of the whole compositions. α‐Pinene was the major component although its percentage varied substantially. The 37 compositions were subjected to statistical analysis which suggested the existence of two principal groups I and II, one of these being subdivided into two sub‐groups IIA and IIB. Group I, the most important group with 24 oil samples out of 37, differed from sub‐groups IIA and IIB, essentially by the high content of α‐pinene (mean value, 69.2 %; SD 6.24). Sub‐groups IIA and IIB were differentiated by their content of α‐pinene (Mean values, 49.7 % and 27.3 %, respectively) as well as δ‐3‐carene (10.3 %, sub‐group IIA) and sabinene (8.7 %, sub group IIB). The fair content of (Z)‐6‐pentadecen‐2‐one in sub‐group IIB (Mean value 4.9 %) could be noticed. The compositions of J. oxycedrus essential oils from Algeria, previously reported, were discussed with respect to groups and sub‐groups of the present study.
Experimental Part
Plant Material
Leaves from 37 individual plants of two subspecies of Juniperus oxycedrus were collected during December 2018 to March 2019 in two localities of Northwestern of Algeria (Figure 1). Twenty‐eight samples of J. oxycedrus subsp. oxycedrus were sampled at Tlemcen province (Ouled Mimoun: S1‐S9; Terny: S10‐S18; Aïn Fezza: S19‐S28) and nine samples of J. oxycedrus subsp. macrocarpa were collected at Aïn Témouchent province (Béni Saf: S29‐S37).
Identification of the plants was performed by Dr. B. Babali and Prof. F. Hassani (Laboratory of Ecology and Management of Natural Ecosystems, University of Tlemcen, Algeria). A voucher specimen has been deposited at the Laboratory of Natural Products (Department of Biology, University of Tlemcen) under the accession N°C. 48.
Essential Oil Distillation
The leaves were dried for a week, in the shade, and then submitted (452–546 g) to hydrodistillation for 2 h using a Clevenger‐type apparatus. Yields have been calculated from dry material (w/w).
Analytical GC
GC analyses were performed on a Perkin‐Elmer Clarus 500 gas chromatograph (FID) equipped two fused silica capillary columns (50 m×0.22 mm, 0.25 μm film thickness), BP‐1 (polydimethyl siloxane) and BP‐20 (polyethylene glycol). The oven temperature was programmed from 60 °C to 220 °C at 2 °C/min and then held isothermal at 220 °C for 20 min, injector temperature: 250 °C; detector temperature: 250 °C; carrier gas: hydrogen (1.0 mL/min); split: 1/60.
The relative proportions of the oil constituents were expressed as percentages obtained by peak area normalization, without using correcting factors. Retention indices (RI) were determined relative to the retention times of a series of n‐alkanes with linear interpolation (“Target Compounds” software from Perkin‐Elmer).
GC/MS Analysis
The essential oils were analyzed with a Perkin‐Elmer TurboMass detector (quadrupole), directly coupled to a Perkin‐Elmer Autosystem XL, equipped with a fused‐silica capillary column (50 m×0.22 mm i.d., film thickness 0.25 μm), BP‐1 (dimethylpolysiloxane). Carrier gas, helium at 0.8 mL/min; split, 1/60; injection volume, 0.5 μL; injector temperature, 250 °C; oven temperature programmed from 60 °C to 220 °C at 2 °C/min and then held isothermal (20 min); Ion source temperature, 250 °C; energy ionization, 70 eV; electron ionization mass spectra were acquired over the mass range 40–400 Da.
13C NMR Analyses
13C NMR analysis was performed on a Bruker AVANCE 400 Fourier Transform spectrometer operating at 100.623 MHz for 13C, equipped with a 5 mm probe, in deuterated chloroform (CDCl3), with all shifts referred to internal tetramethylsilane (TMS). 13C NMR spectra were recorded with the following parameters: pulse width (PW), 4 μs (flip angle 45°); acquisition time, 2.73 s for 128 K data table with a spectral width (SW) of 220 000 Hz (220 ppm); CPD mode decoupling; digital resolution 0.183 Hz/pt. The number of accumulated scans ranged 2 000–3 000 for each sample (around 40 mg of oil in 0.5 mL of CDCl3). Exponential line broadening multiplication (1.0 Hz) of the free induction decay was applied before Fourier Transform.
Identification of Components
Identification of the components was based on:
- comparison of their GC retention indices (RI) on polar and apolar columns, determined relative to the retention times of a series of n‐alkanes with linear interpolation (Target Compounds software of Perkin‐Elmer), with those of authentic compounds and with reference dataref. cbdv202401584-bib-0033;
- computer matching against commercial mass spectral libraries[ref. cbdv202401584-bib-0041, ref. cbdv202401584-bib-0042, ref. cbdv202401584-bib-0043];
- comparison of the signals in the 13C NMR spectra of essential oils with those of reference spectra compiled in the laboratory spectral library, with the help of a laboratory‐made software.[ref. cbdv202401584-bib-0031, ref. cbdv202401584-bib-0032, ref. cbdv202401584-bib-0044] In the investigated samples individual components were identified by NMR at contents as low as 0.4–0.5 %.
Data Analysis
Principal Components Analysis (PCA) and Hierarchical clustering (Ward’s method) were performed by Xlstat (Adinsoft, France).ref. cbdv202401584-bib-0045
Author Contributions
Conceptualization: C.B., J.C. and F.T.; methodology: C.B. and F.T., sampling and extraction: C.E.W.M. and K.S. plant identification: B.B.; investigation: formal analysis, C.B. and M.P.; original draft preparation: C.B., J.C. and F.T., writing review: C.B. and J.C.
Conflict of Interests
The authors declare no conflict of interest.
Supplementary Materials
References
- 1 P. Quézel , F. Médail , ‘Nouvelle flore de l’Algérie et des régions désertiques méridionales ’, Edition CNRS, Tome II, Paris, France, 1962, 1169.
- 2 A. Dobignard , C. Chatelain , ‘Index synonymique de la flore d’Afrique du nord: Pteridophyta, Gymnospermae, Monocotyledoneae’, éditions des conservatoire et jardinbotaniques, Vol. 1, Genève, Suisse, 2010, 455.
- 3 A. Farjon , ‘A Handbook of the World’s Conifers’, Koninklijke Brill NV, Leiden, Vol. 2, Boston, The Netherlands, 2010.
- 4IUCN, ‘A guide to medicinal plants in North Africa’, IUCN Centre for Mediterranean cooperation Editor, Malaga, Spain, 2005, 256 p.
- 5J. Bellakhdar, ‘La pharmacopée marocaine traditionnelle: Médecine arabe ancienne et savoirs populaires’, Ibis Press, Eds. Le Fennec, Paris, France, 1997, 764.
- 6L. El Rhaffari, A. Zaid, ‘Pratique de la phytothérapie dans le sud-est du Maroc (Tafilalet) : Un savoir empirique pour une pharmacopée rénovée’, Proceedings of the 4th European Congress on Ethnopharmocology, „From the sources of knowledge to the medicines of the future“, Metz (France), 2000/05/11-13, IRD Editions, 2002, 468 p. ISBN 2-7099-1504-9.
- 7A. Boudjelal, C. Henchiri, M. Sari, D. Sarri, N. Hendel, A. Benkhaled, G. Ruberto, J. Ethnopharmacol. 2013, 148(2), 395–402
- 8 S. Menad , D. Achour , M. Brada , Int. J. Biosci. 2018, 13, 98–103.
- 9 N. Basa , H. Djebaili , M. Mokrani , M. M. Senoussi , S. Boulehbal , A. Zellagui , J. Pharm. Res. Int. 2022, 34, 1–7.
- 10 A. H. A. Boukhaloua , M. Berrayah , F. Bennabi , A. Ayache , F. Abdeldjebar , Ukr. J. Ecol. 2022, 12, 31–39.
- 11 A. Zatout , R. Djibaoui , G. Flamini , R. Ascrizzi , C. Benbrahim , H. E. Mazari , F. Benkredda , S. Mechaala , A. Kassah-Laouar , Afr. J. Clin. Exper. Microbiol. 2022, 23, 278–289.
- 12 S. Boudiba , A. Ngenge Tamfu , B. Berka , K. Hanini , S. Hioun , K. Allaf , L. Boudiba Nat. Prod. Commun. 2021, 16, 1–11.
- 13 K. Cheraif , B. Bakchiche , A. Gherib , S. K. Bardaweel , M. Çol Ayvaz , G. Flamini , R. Ascrizzi , M. A. Ghareeb , Molecules 2020, 25, 1710.32276465
- 14 S. Menad , D. Achour , M. Brada , South Asian J. Exp. Biol. 2022, 12, 492–498.
- 15 C. Zahaf , D. Achour , M. Mokhtar , M. Brada , South Asian J. Exp. Biol. 2017, 7, 74–83.
- 16 F. Bachiri , F. Benouaklil , F. Hamaidi-Chergui , F. Saidi , J. Essent. Oil-Bear. Plants 2022, 25, 1–10.
- 17 Y. Foudil-Cherif , N. Yassaa , Food Chem. 2012, 135, 1796–1800.22953925
- 18M. Boussaïd, C. Bekhechi, A. Bouayed, F. Hassani, J. Casanova, M. Paoli, F. Tomi, Chem. Biodiversity, 2024, 21, e202400862 Doi: 10.1002/cbdv.202400862..
- 19 D. Dahmane , T. Dob , C. Chelghoum , J. Mater. Environ. Sci. 2015, 6, 3159–3167.
- 20A. Bouyahyaoui, ‘Contribution à la valorisation des substances naturelles: Etude des huiles essentielles des Cupressacées de la région de l’Atlas algérien’, Thèse de doctorat en Sciences, Filière: Biologie, Option: Microbiologie, 2017, Université Abdelhamid Ibn Badis de Mostaganem, Algeria.
- 21 S. Menad , D. Achour , M. Brada , Int. J. Biosci. 2018, 13, 98–103.
- 22 S. Guerroudj , M. Maatoug , K. Naceur , R. Chaibi , M. Khene , A. Boualem , Ukr. J. Ecol. 2021, 11, 105–111.
- 23 T. Dob , D. Dahmane , C. Chelghoum , Pharm. Biol. 2006, 44, 1–6.
- 24 A. H. A. Boukhaloua , M. Berrayah , F. Bennabi , A. Ayache , F. Abdeldjebar , Ukr. J. Ecol. 2022, 12, 31–39.
- 25H. Fadel, F. Benayache, J. C. Chalchat, G. Figueredo, P. Chalard, H. Hazmoune, S. Benayache, Nat Prod. Res. 2021, 35, 2616-2620 10.1080/14786419.2019.1687473.
- 26 H. Djebaili , A. Zellagui , N. Gherraf , J. Nat. Prod. Plant Resour. 2013, 3, 40–44.
- 27 H. Medini , A. Elaissi , M. L. Khouja , I. Chraief , F. Farhat , M. Hammami , R. Chemli , F. Harzallah-Skhiri , Chem. Biodiversity 2010, 7, 1250–1266.
- 28 I. B. Semerjieva , V. D. Zheljazkov , I. Dincheva , T. Astatkie , M. Kačániová , Ind. Crops Prod. 2020, 158, 113005.
- 29 J. B. Boti , A. Bighelli , C. Cavaleiro , L. Salgueiro , J. Casanova , Flavour Fragr. J. 2006, 21, 268–273.
- 30 J. Ottavioli , A. Bighelli , J. Casanova , F. Tomi , Nat. Prod. Commun. 2018, 13, 213–216.
- 31 F. Tomi , P. Bradesi , A. Bighelli , J. Casanova , J. Magn. Reson. Anal. 1995, 1, 25–34.
- 32 Z. A. Ouattara , J. B. Boti , A. C. Ahibo , S. Sutour , J. Casanova , F. Tomi , A. Bighelli , Flavour Fragr. J. 2014, 29, 371–379.
- 33 V. I. Babushok , P. J. Linstrom , I. G. Zenkevich , J. Phys. Chem. Ref. Data. 2011, 40(043101), 1–47.
- 34 P. Salehi , A. Sonboli , F. Eftekhar , S. Nejad-Ebrahimi , M. Yousef Zadi , Biol. Pharm. Bull 2005, 28,1892–1896.16204941
- 35 K. H. C. Baser , G. Özek , T. Özek , A. Duran , H. Duman , Flavour Fragr. J. 2006, 21, 650–655.
- 36 P. Weyerstahl , H. Marchall , U. Splittgerber , D. Wolf , Flavour Fragr. J. 2000, 15, 153–173.
- 37 J. A. Pino , R. Marbot , J. Agric. Food Chem. 2001, 49(12), 5880–5882.11743778
- 38 J. Ledauphin , . J. F. Saint-Clair , O. Lablanquie , H. Guichard , N. Founier , E. Guichard , D. Barillier , J. Agric. Food Chem. 2004, 52, 5124–5134.15291485
- 39 M. Gonny , P. Bradesi , J. Casanova , Flavour Fragr. J. 2004, 19, 424–433.
- 40 M. Gonny , C. Cavaleiro , L. Salgueiro , J. Casanova , Flavour Fragr. J. 2006, 21, 99–106.
- 41 W. A. Konig , D. H. Hochmuth , D. Joulain , ‘Terpenoids and Related Constituents of Essential Oils’, Library of Mass Finder 2.1, Institute of Organic Chemistry, Hamburg, 2001.
- 42R. P. Adams, ‘Identification of Essential Oils Components by Gas Chromatography/Mass Spectroscopy’,, 2017, ed. 4.1, 30 Jan. 2017, ISBN 978-1-932633-21-4.
- 43 National Institute of Standards and Technology, PC Version of the NIST/EPA/NIH Mass Spectral Library; NIST: Gaithersburg, MD, USA, 2008.
- 44O. Bazzali, T. H. Thai, T. M. Hoi, N. S. Khang, N. T. Hien, J. Casanova, A. Bighelli, F. Tomi, Molecules, 2016, 21, 840–851
- 45P. Legendre, L. Legendre, ‘Numerical Ecology’, 2nd Edn, Elsevier Science, Amsterdam, 1998.
