Recommendations for recognizing and diagnosing Acute Hepatic Porphyria in atypical patient populations
Abstract
Background:
Acute Hepatic Porphyria is a group of four rare genetic but treatable diseases that often go undiagnosed due to its non-specific symptoms, under-recognition of the condition by clinicians, and the lack of access to specialists and appropriate testing. This case-control study investigates the phenotypic and demographic patterns in Acute Hepatic Porphyria (AHP) patients at a tertiary care center (University of California Los Angeles) to update recommendations for recognition and diagnosis of this disease in our community.
Method:
A retrospective chart analysis was conducted on 45 patients who were evaluated for AHP, Electronic Medical Record (EMR) data was collected and analyzed to investigate clinical differences and correlations.
Results:
27 patients tested positive for AHP through urinary metabolites and confirmatory genetic testing and 18 patients tested negative; of those, 16 patients received a definite alternative diagnosis. Hashimoto’s, T1DM (Type 1 Diabetes Mellitus), Fibromyalgia and cannabinoid use with cyclic vomiting syndrome were negatively correlated with AHP, while psychiatric disorders and OBGYN disorders were positively correlated with AHP. The highest rate of diagnosis resulted from a combination of genetic and biochemical testing. Testing outside of an acute attack was not associated with a positive diagnosis.
Conclusions:
Patients with a history of OBGYN disorders and psychiatric disorders may be at increased risk of having AHP, yet there is a lack of involvement of these specialties in the diagnosis and care of AHP, in addition to a lack of studies investigating AHP in non-white populations potentially leading to reduced recognition of AHP.
Article type: Research Article
Keywords: Acute Hepatic Porphyria, Underdiagnosis, Rare disease diagnosis, clinical manifestations, Diagnostic delays
Affiliations: University of California Los Angeles David Geffen School of Medicine; University of California San Francisco; University of California Los Angeles
License: CC BY 4.0 This work is licensed under a Creative Commons Attribution 4.0 International License, which allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use.
Article links: DOI: 10.21203/rs.3.rs-4244361/v1 | PubMed: 39711536 | PMC: PMC11661304
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.6 MB)
Introduction
Acute hepatic porphyria (AHP) is a group of four distinct metabolic diseases caused by genetic defects in the heme synthesis pathway (ref. 1): The most common subtype, Acute Intermittent Porphyria (AIP), caused by a mutation in the HMBS gene, followed by Variegate Porphyria (VP), resulting from a PPOX gene mutation and Hereditary Coproporphyria (HCP), caused by a mutation in the CPOX gene. The rarest subtype is ALA dehydratase deficiency porphyria (ADP), caused by a mutation in the ALAD gene (ref. 2). They present similarly as episodic, severe neurovisceral abdominal pain that builds up over a few days and can be difficult to manage, frequently requiring high doses of narcotic pain medications in addition to hospitalization and supportive care. It is thought that attacks are directly related to an accumulation of the heme precursors porphobilinogen (PBG) and/or delta-aminolaevulinic acid (delta-ALA or d-ALA), which appear to have neurotoxic properties, leading to long-term end-organ damage and comorbidities such as kidney and liver disease (ref. 3, ref. 4) as well as higher rates of miscarriage, low birth weight and infertility (ref. 5, ref. 6) if the disease is left untreated. There are currently two approved therapies for AHP: Panhematin (Hemin), an abortive, and a recent FDA approved RNA silencing therapy for prevention, Givosiran (Givlaari, Alnylam Pharmaceuticals, Cambridge MA, USA) (ref. 7). Financial assistance programs are available to reduce or eliminate the cost for those who are under or uninsured or otherwise socioeconomically disadvantaged.
In particular, the subtype Acute Intermittent Porphyria, with a currently diagnosed symptomatic population prevalence of 1:20,000 (ref. 2, ref. 8)shows a significant lack of studies in demographics other than European White (Caucasian) females. This despite a carrier prevalence as high as 1:1,500 (ref. 2) and being easily mistaken for any other cause of acute abdominal pain, including cholecystitis, appendicitis, pancreatitis, urinary tract disease (ref. 6, ref. 9) and OBGYN related pathology (ref. 6, ref. 10). This leads to AIP patients frequently being misdiagnosed, especially in non-Whites and perhaps even males, who may present atypically. While it is commonly accepted that AIP appears more frequent in both carrier status and symptomatic prevalence in patients of Caucasian background (ref. 2), this assumption may be due to most AIP studies being conducted on Caucasian-background patients, against which future studies were then evaluated. Naturally, the common genetic mutations found in the predominantly Nordic European populations of the largest contributor studies are not commonly found in other ethnicities, adding to the assumption that AIP affects predominantly patients of Caucasian heritage and patients of female sex, despite the AIP causing genetic mutations being inherited equally among the sexes (ref. 2, ref. 7). There are multiple studies from other countries highlighting surprising symptomatic prevalence rates of AIP as high as 1.16% among other ethnicities, such as Northern India (ref. 10), where patients present primarily with Acute Flaccid Paralysis and Neuropathies, and studies from Mexico including one showing a 0.67% symptomatic prevalence of AIP among the tested psychiatric populations (ref. 11).
Correct identification of the 4-disease group takes, on average, 10–15 years and often requires access to tertiary care, provider familiarity with disease and testing (ref. 12), which typically takes a week or longer to result, often after the patient is discharged and lost to follow up. Hence, AHP disproportionately affects the un- and underinsured socioeconomically disadvantaged patient populations, who are unable to return to clinic appointments due to lack of paid sick time, transportation, health insurance coverage, placing them at greatest risk for remaining undiagnosed and developing long term irreversible comorbidities due to lack of treatment.
During our most recent UCLA-UCSF joint study (ref. 13) aimed at reducing diagnostic delays in AHP using machine learning algorithms, we discovered that the current number of patients diagnosed with AHP within the UCLA Health patient population is 0.0034%, much lower than the symptomatic population prevalence estimate across the United States of 0.005% (ref. 3, ref. 4), yielding an under-diagnosis rate of 32% in comparison to the general US population prevalence. Considering that UCLA Health is a tertiary care healthcare system, we should see a diagnosis rate that exceeds the estimated population symptomatic prevalence. Our study aims to suggest areas of targeted intervention to identify undiagnosed patient cohorts: patients who may present atypically, such as males and patients of other ethnicities, who often may go undiagnosed due to structural barriers to specialist healthcare access and/or logistical testing difficulties.
This study aims to evaluate a cohort of patients who have recently been evaluated for AHP (2019–2022) to investigate phenotypic differences and correlations in demographics and clinical presentations by AHP subtype that may assist providers in clinical decision making to increase rates of AHP diagnosis specifically in non-White and atypically presenting patients in the future.
Materials and Methods
Study Population
This is a cohort study of a total of 45 patients at the University of Los Angeles California (UCLA) Health System who have undergone evaluation for AHP between 2019 and 2022. An initial data inquiry resulted in 153 patients with an International Statistical Classification of Diseases and Related Health Problems (ICD) code of any of the AHP subtypes. Manual chart review was then conducted to further classify patients into the AHP + cohort, the AHP− cohort or “other”, which was a cohort consisting of patients not utilized for this study due to either pending diagnostic testing or insufficient data in the chart.
Inclusion criteria consisted of patients over 18 years of age with a true AHP diagnosis code for the AHP + cohort (specified as an ICD code for any of the AHP subtypes + either a confirmatory genetic test and/or confirmatory biochemistry) and a tentative AHP diagnosis code for the AHP− cohort (specified as patients over 18 years of age with an ICD code for any of the AHP subtypes + subsequent negative genetic testing and/or negative biochemistry when symptomatic and/or a satisfactory alternative diagnosis was found. 27 patients ultimately were diagnosed with AHP (AHP + cases) and 18 were ruled out for the disease (AHP− controls).
Electronic Medical Record (EMR) data was collected from both groups for comparison and correlation analysis, including quantitative and qualitative information: demographics, clinical notes including past medical history, laboratory results including genetic mutations if available, referral and diagnosis sources, comorbid conditions, family history, and medication lists. Through manual chart review, symptoms, comorbidities, clinical histories, lab results and health system touch points for diagnosis and referral journey were collected for each individual patient and prepared for comparisons in frequency within the AHP + and AHP− cohort as well as between the cohorts. Clinical data was included in this study if it was presented more than once among a cohort or subtype cohort.
AHP + patients were then grouped into AHP subtype cohorts: Acute Intermittent Porphyria (AIP/HMBS mutation), Variegate Porphyria (VP/PPOX mutation) and Hereditary Coproporphyria (HCP/CPOX mutation) to investigate phenotypic differences and commonalities in genotypes. There were no patients with the ALA Dehydratase Deficiency (ADP/ALAD mutation) subtype in this cohort.
Data Analysis
Both descriptive and inferential statistical analysis of the data was carried out. Fisher’s Exact Test was used to find the significant difference in different parameters and values. Statistical significance was established at a p-value of 0.05 or less with a 95% Confidence Interval. All statistical calculations were performed using SPSS (IBM Corp., Armonk, NY) version 29.0.0.
Results
Demographics (Table 1)
Table 1: Sociodemographic Features on the Basis of Presence or Absence of AHP
| AHP+, (n = 27) n, (percentage) | AHP- (n = 18) n, (percentage) | p-value | ||
|---|---|---|---|---|
| Gender | Male | 5 (19) | 5 (28) | 0.489 |
| Female | 22 (81) | 13 (72) | ||
| Age | Minimum | 24 | 24 | 0.791 |
| Maximum | 70 | 79 | ||
| Mean (SD) | 47 (16) | 49.33 (19) | ||
| Ethnicity | White | 21 (78) | 14 (78) | 0.148 |
| Non-Hispanic | 0 | 1 (6) | ||
| Asian | 0 | 1 (6) | ||
| decline/unknown | 0 | 1 (6) | ||
| Hispanic or Latino | 4 (15) | 0 (0) | ||
| other/Non-Hispanic | 2 (7) | 1 (6) | ||
| Family History | Yes | 13 (48) | 2 (11) | <0.001* |
| AHP subtype | AIP | 18 (67) | ||
| VP | 6 (22) | |||
| HCP | 3 (11) |
AHP: Acute Hepatic Porphyria, AIP: Acute Intermittent Porphyria, HCP: Hereditary Coproporphyria, VP: Variegate Porphyria,, n = total number of patients
* p < 0.05 is statistically significant
Age and gender
In the AHP + cohort, 82% were female, and 19% were male, among the AHP− cohort, 72% were female, while 28% were male. The average patient age was 49.7 years. There was no significant difference found in gender or age distribution between the positive and negative groups.
Ethnicity
The majority (78%) of patients across both the positive and negative cohort were White, followed by 9% of non-White Hispanic or Latino, 4% non-Hispanic/black, 2% Asian and 2% were of unknown ethnic origin. While there were no statistically significant differences in ethnicity distribution between the positive and negative groups, all patients identifying as Hispanic or Latino were AHP+, whereas all AHP− patients identified as Asians.
Family history
In the AHP-positive group, 48% reported having a family history of AHP, while 11% in the AHP− group reported a positive family history, leading to a statistically significant difference (p < 0.001).
AHP subtypes
The most common AHP subtype was Acute Intermittent Porphyria (AIP, 67%) for both females and males, followed by Variegate Porphyria (VP, 22%), which was more common in females than males although not statistically significant due to small sample size, and lastly, Hereditary Coproporphyria (HCP, 11%) with an equal distribution among females and males. There were no patients with the ALA Dehydratase Deficiency (ALAD/ADP) Porphyria subtype in this study.
Referral and Diagnostic Journey
Table 2 indicates that 22% of patients came to UCLA based on self-referral for evaluation of Porphyria, followed by 15% of patients being referred from their primary care provider (PCP) and 7% by Hematology/Oncology. The rest of the patients were referred by Cardiology, Internal Medicine/Hospitalist, Dermatology, Medical Genetics, Neurology, Pediatrics, out of state (OOS) Porphyria specialists, and Outside Hospital (OSH) Gastroenterology in equal proportion. Interestingly, one patient was referred by their local veterinarian and ultimately tested positive for AHP.
Table 2: Source of Referral
| Referred by | percentage | n |
|---|---|---|
| Self | 22% | 6 |
| PCP to subspecialty: Rheumatology/Gastroenterology/Neurology | 15% | 4 |
| Internal medicine/Hospitalist | 11% | 3 |
| Genetics to Porphyria specialist | 4% | 1 |
| Rheumatology to Genetics | 4% | 1 |
| Pediatrics | 4% | 1 |
| Hematology/Oncology to Porphyria Specialist | 4% | 1 |
| General Dermatology to Specialist Dermatology | 4% | 1 |
| Veterinarian to human Neurology | 4% | 1 |
| Military cardiology to Hematology/Oncology | 4% | 1 |
| Neurology to Porphyria Specialist | 4% | 1 |
PCP = Primary Care Provider, n = total number of patients
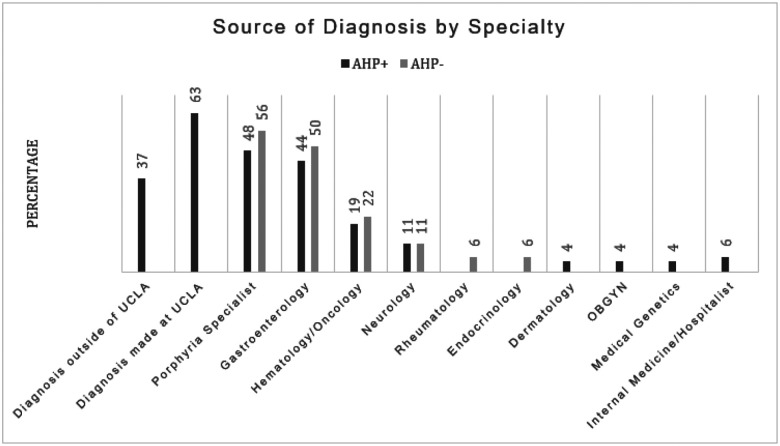
Figure 1 compares sources of diagnosis for AHP + and AHP− patients; 37% of AHP + patients had a prior diagnosis of AHP before coming to UCLA for further care, and 63% of patients were diagnosed at UCLA. The most common physician specialty to diagnose AHP was Gastroenterology (44%) followed by Hematology/Oncology (19%) and Neurology (11%). 1 patient was diagnosed through preconception genetic testing at their Obstetrician Gynecologist (OBGYN). Additionally, 48% of AHP + patients had been evaluated by a Porphyria specialist, which can consist of any medical specialty. Evaluation of AHP− patients showed a similar pattern, with the majority being assessed by Gastroenterology, Hematology/Oncology and Neurology. 56% of AHP− patients were formally assessed by a Porphyria specialist.
Method of Diagnosis
Table 3 provides an overview of all testing conducted in both AHP + and AHP− groups.
Table 3: Diagnostic Methods
| Ordered laboratory testing and symptomatic circumstances | AHP+ | AHP- | P Value* | ||
|---|---|---|---|---|---|
| percentage | Total (n = 27) | percentage | Total (n = 18) | ||
| Genetic Testing for AHP (HMBS, CPOX, PPOX) | 74% | 20 | 39% | 7 | |
| Positive genetic test result for AHP (HMBS, CPOX, PPOX) of those tested | 100% | 20 | 0% | 0 | <0.001* |
| Negative genetic test result for AHP for those tested | 0% | 0 | 100% | 7 | |
| Total Whole Blood Porphyrins taken | 63% | 17 | 44% | 8 | |
| WB porphyrins elevated:>80 mcg/dL | 35% | 6 | 50% | 4 | 0.667 |
| WB Porphyrins normal < 80 mcg/dL | 65% | 11 | 50% | 4 | |
| Total number of urinalysis (UA) taken | 89% | 24 | 89% | 16 | |
| Number of positive UA of those tested | 71% | 17 | 6%** | 1** | <0.001* |
| Urinalysis taken when symptomatic | 50% | 12 | 69% | 11 | |
| Urinalysis taken when not symptomatic | 50% | 12 | 31% | 5 | |
| Positive UA when symptomatic | 58% | 7 | 0% | 0 | |
| Negative UA when symptomatic | 42% | 5 | 100% | 11 | |
| Positive UA when not symptomatic | 50% | 6 | 0% | 0 | |
| Negative UA when not symptomatic | 50% | 6 | 100% | 5 | |
| Urinalysis breakdown by compound | |||||
| Number of UAs with both ALA and PBG available | 63% | 15 | 63% | 10 | |
| Fraction of positive UAs with either ALA or PBG or both if available elevated | 53% | 9 | 0% | 0 | |
| Fraction of positive UAs with both ALA and PBG negative but + for other compounds | 24% | 4 | 6%** | 1** | |
| Of positive UAs with both ALA and PBG available, PBG- but ALA+ | 20% | 3 | 0% | 0 | |
| Of positive UAS with both ALA and PBG available, PBG + but ALA- | 7% | 1 | 0% | 0 | |
| Fraction of positive UAs where ALA and PBG were not done or negative but other values were positive | 24% | 4 | 6%** | 1** | |
| Positive UAs with Uroporphyrins and Coproporphyrins only, no ALA/PBG | 6% | 1 | 6%** | 1** | |
| Positive UAs with no ALA/PBG but total Porphyrins | 12% | 2 | 0% | 0 | |
| Positive UAs with total Porphyrins, Uroporphyrins and Coproporphyrins but no ALA/PBG | 6% | 1 | 0% | 0 | |
AHP = Acute Hepatic Porphyria, + = positive, − = negative, HMBS = hydroxymethylbilane synthase, CPOX = Coproporphyrinogen Oxidase, PPOX = protoporphyrinogen oxidase, WB = Whole Blood, UA = Urinalysis, ALA = Aminolevulinic acid dehydratase, PBG = Porphobilinogen
* p < 0.05 is statistically significant
** 1 patient in the AHP− group had elevated Coproporphyrin I values with negative ALA and PBG, later found to have a FECH mutation and therefore was considered negative for the AHP subtypes included in this study (AIP, VP, HCP, ADP only).
AHP was considered positive if the patient either had a positive genetic test for a known pathogenic mutation in the genes HMBS, CPOX, PPOX and/or positive biochemistry Urinalysis (UA) in the absence of a genetic test (asymptomatic carriers were included in this study). Qualifying biochemistry included elevations of any one or combination of random urinary delta- aminolaevulinic acid (d-ALA) > 5.4 mg/g creatinine, urinary random Porphobilinogen (PBG) > 2 mg/g creatinine, 24-hour Total Porphyrins > 120 μg/L (144 nmol/L). Uroporphyrins > 2 mcg/dL (< 2.4 nmol/L), Coproporphyrins > 2 mcg/dL (< 30 nmol/L) are listed but did not determine AHP status alone without a confirmatory genetic test.
Whole Blood (WB) Porphyrins are mentioned with a normal cut-off value of < 80 mcg/dL but did not affect a patients’ positive or negative status for AHP for the purposes of this study: In the AHP + group, 63% had WB porphyrins taken, while in the APH− group 44% had WB porphyrins taken. Among those who had WB porphyrins taken, 35% in the AHP + group and 50% in the AHP− group had elevated levels of porphyrins (> 80 mcg/dL,), indicating no significant difference (p = 0.667).
Genetic Testing
Genetic test results were available for 74% in the AHP + group and for 38.89% of patients in the AHP− group. Within the AHP + group that had genetic testing done, 48% had HMBS mutations, 19% had PPOX mutations and 7% had CPOX mutations. All available mutations were heterozygous, with 2 patients having the same HMBS mutation (1 male and 1 female: heterozygosity C517C > T, which is a known pathogenic association with autosomal dominant AIP).
Biochemistry
In both the AHP + and AHP− groups, 89% in both the AHP + and AHP− group had Urinalysis (UA) results available for biochemistry evaluation. Among the AHP + group with available UA, a total of 71% samples were positive, containing at least one elevated value of qualifying biochemistry (random urinary d-ALA, PBG, total Porphyrins, Uroporphyrins, Coproporphyrins with the latter two categories necessitating a positive genetic test to be considered positive for AHP). None of the individuals in the AHP− group had positive UA results except for 1 patient who had elevated Coproporphyria I values with negative ALA and PBG, later found to have a FECH (ferrochelatase) mutation and therefore was considered negative for the AHP subtypes included in this study.
63% of UAs taken from both the AHP + and AHP− group contained both ALA and PBG values. Of the AHP + positive UAs, 53% had either ALA or PBG or both (if both were available) elevated, 7% had PBG elevated but ALA negative, 20% had negative results for PBG but positive results for ALA. Further, 17% of UA samples that were negative for both ALA and PBG were positive for either Total Porphyrins, Coproporphyrins and/or Uroporphyrin (but had a positive genetic test).
Only 50% of the AHP + UAs were taken during a symptomatic attack and of those, 58% were positive. Of the 50% of AHP + UAs taken outside of an acute attack, 50% were positive also.
In the AHP + group, 81% of patients were symptomatic at the time of evaluation (Table 4 shows a list of symptoms considered for the purposes of this study). 74% had a positive genetic test for any of the AHP subtypes, 56% of patients had a positive biochemistry. 30% of all AHP + patients were symptomatic with a positive genetic test but had negative or not available biochemistry. 41% of AHP + patients had both a positive genetic test as well as positive biochemistry. One male patient had a positive genetic test with positive biochemistry but presented atypically with a Neurodevelopmental Disorder.
Table 4: Comparison of frequently presenting symptoms between AHP− and AHP + patients
| Presenting Symptoms | AHP+, (n = 27), % | AHP-, (n = 18), % |
|---|---|---|
| Visual and vestibular disturbances | 2 (7) | 2 (11) |
| Urinary symptoms* | 10 (37) | 2 (11) |
| Skin blisters or rashes | 10 (37) | 4 (22) |
| Abdominal pain | 20 (74) | 11 (61) |
| Bloating | 0 (0) | 5 (28) |
| Diarrhea | 0 (0) | 5 (28) |
| Paresthesia | 10 (37) | 6 (33) |
| Muscle weakness or spasms | 14 (52) | 5 (28) |
| Dysphagia | 0 (0) | 2 (11) |
| Nausea/Vomiting | 6 (22) | 3 (17) |
| Constipation | 6 (22) | 5 (28) |
| Tachycardia/palpitations | 3 (11) | 1 (11) |
| Body aches and pains | 0 (0) | 4 (22) |
| fatigue/easy fatigability | 0 (0) | 4 (22) |
| Back pain | 4 (15) | 2 (11) |
| Stabbing foot/leg pain | 7 (26) | 0 (0) |
| OBGYN disorders and symptoms** | 14 (52) | 2 (11) |
| Non-AHP medication brings relief (including Amitriptyline, Antihistamine, Steroids and Proton Pump Inhibitors) | 0 (0) | 6 (33) |
| Multiple allergies (> 5) listed in chart | 0 (0) | 4 (22) |
| Symptoms triggered by food | 0 (0) | 5 (28) |
AHP + = Acute Hepatic Porphyria positive, AHP− = Acute Hepatic Porphyria negative
* Urinary symptoms include discolored urine, urinary urgency, increased urinary frequency, burning/discomfort with urination without presence of UTI, cystitis without presence of infection
** OBGYN disorders and symptoms = Obstetrics and Gynecology, symptoms for this study included: vaginal itching/discomfort unspecified, spotting, irregular periods, ovarian cysts, dyspareunia, infertility, endometriosis, history of hysterectomy, miscarriage, cholestasis of pregnancy, premature delivery, fibroids, dysmenorrhea.
Differential diagnoses in AHP− patients
39% of AHP− patients had a negative genetic test with only 1 patient being ruled out for AHP based on the negative genetic test alone. 33% of patients were ruled out for AHP based on a negative UA alone, and 28% had both a negative genetic test and negative UA.
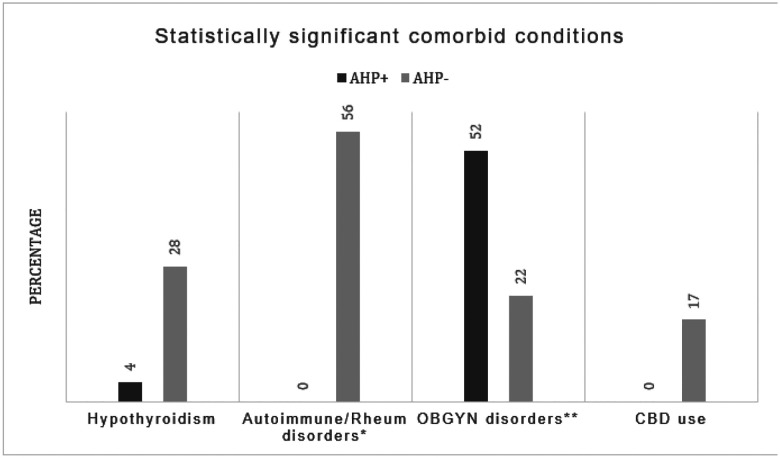
Table 5 provides an overview of the final diagnoses given to patients in the AHP− cohort: In 61% of AHP− patients an satisfactorily explanatory alternative diagnosis was ultimately found; of those, 28% were diagnosed with an autoimmune/rheumatological disorder (mast cell activation syndrome in 2 patients, Dermatomyositis in 1 patient, undifferentiated Connective Tissue disorder in 1 patient, Hypothyroidism in 1 patient), 22% of AHP− patients ultimately were diagnosed with a malignancy (sigmoid, pancreas, parathyroid, Chronic Myeloid Leukemia CML), 17% with Cyclic Vomiting Syndrome due to Cannabidiol (CBD) use, 11% patients were newly diagnosed with Irritable Bowel Syndrome (IBS) or functional Gastrointestinal (GI) disorder, 11% patients were diagnosed with Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), 6% had Shy Drager Syndrome, 6% Unspecified seizure disorder, 6% Endometriosis, 6% Medication reaction, and only 2 patients are still undiagnosed and have been entered into the undiagnosed rare diseases network for further workup, one of them notably having a history of bariatric surgery (see comorbidities section).
Table 5: Final Diagnosis of AHP− patients
| Final Diagnosis | n | percentage |
|---|---|---|
| Malignancy | 4 | 22% |
| New diagnosis of IBS/functional GI | 2 | 11% |
| Autoimmune disorders total* | 5 | 28% |
| Mast cell activation syndrome | 2 | 11% |
| Medication reaction | 1 | 6% |
| Endometriosis | 1 | 6% |
| cyclic vomiting syndrome | 3 | 17% |
| Undiagnosed** | 2 | 11% |
| CIDP | 2 | 11% |
| Shy Drager syndrome | 1 | 6% |
| EPP | 1 | 6% |
| Seizure disorder unspecified | 1 | 6% |
AHP− = Acute Hepatic Porphyria negative, n = total number of patients, IBS = irritable bowel syndrome, functional GI = functional gastrointestinal disorder, CIDP = Chronic Inflammatory Demyelinating Polyneuropathy, EPP = Erythropoietic Protoporphyria
* Autoimmune Disorders: Mast cell activation syndrome, CIDP, Shy Drager Syndrome
** Entered into the Unknown Rare Disease Network for further evaluation
Treatment of AHP + patients
52% of patients received Hemin/Panhematin, and 14% of those reported it not being effective for abdominal pain, as it only provided relief for neurological pain and parasthesias. 71% of patients on Hemin/Panhematin eventually discontinued the treatment due to complete cessation of relief. 26% of patients receive Givosiran [13], with 1 patient stating intolerance in form of intermittent nausea, headache and dizziness for 14 days post injection (Latino ethnicity, HMBS heterozygous C77G > A p. Arg26His) and one person reporting no relief (HMBS heterozygous, unknown exact mutation) until the dosage was increased based on body weight. 15% of patients received Dextrose solutions, 15% used opioid medications and in equal percentages of 4% patients utilized over the counter (OTC) pain relief, Intravenous Immunoglobulin (IVIG), Lupron, Chinese Herbs and Cannabinoid (CBD).
19% of patients were not undergoing any specific treatment for AHP at the time of the data collection phase (January 1, 2021 – December 31, 2022).
Clinical presentation of AHP + and AHP− patients
Symptoms began for 19% of AHP + patients regardless of genotype and gender with a seizure (unknown trigger and without pre-existing seizure related conditions per chart review), followed by skin blisters and/or rashes (11%), which were overall more common in the VP subtype (7%). For the HMBS subtype, 11% of females stated symptoms began with pregnancy, with 1 person terminating their pregnancy due to intolerable worsening symptoms and 1 person receiving twice weekly Panhematin infusions throughout her pregnancy, giving birth to a child with infantile pyknocytosis thought to be due to Panhematin use.
Other patients reported symptoms that began after an accident (4%) and surgery (4%).
45% of female patients reported symptom worsening with menses.
Table 4 and Fig. 2 show the presenting symptoms among AHP + and AHP− patients in our study, while Table 6 and Fig. 3 present the results of a correlation analysis aimed at predicting likelihood of having AHP based on existing comorbidities and symptoms alone.
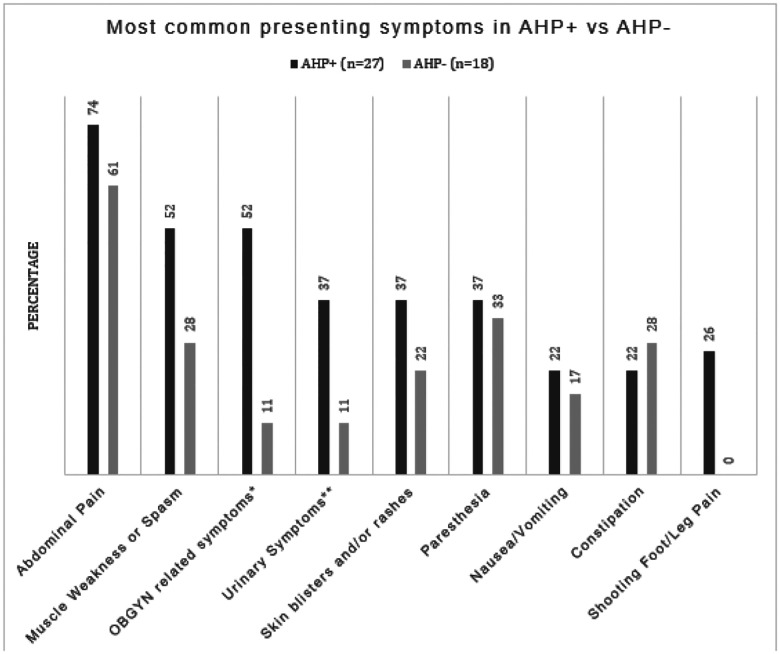
Table 6: Comorbid conditions and symptoms associated with presence or absence of AHP
| Significance value p* | ||
|---|---|---|
| Irritable Bowel Syndrome (IBS) | Correlation Coefficient | .036 |
| Sig. (2-tailed) | .813 | |
| N | 45 | |
| Hypothyroidism | Correlation Coefficient | −.347 |
| Sig. (2-tailed) | .020* | |
| N | 45 | |
| Autoimmune/Rheum Disorders** | Correlation Coefficient | −.580 |
| Sig. (2-tailed) | .000* | |
| N | 45 | |
| Psychiatric Disorders*** | Correlation Coefficient | .239 |
| Sig. (2-tailed) | .114 | |
| N | 45 | |
| Major Depressive Disorder (MDD) | Correlation Coefficient | −.095 |
| Sig. (2-tailed) | .535 | |
| N | 45 | |
| General Anxiety Disorder (GAD) | Correlation Coefficient | .109 |
| Sig. (2-tailed) | .476 | |
| N | 45 | |
| Neurodevelopmental Disorders including Autism | Correlation Coefficient | .176 |
| Sig. (2-tailed) | .247 | |
| N | 45 | |
| Schizophrenia/Psychosis | Correlation Coefficient | .100 |
| Sig. (2-tailed) | .513 | |
| N | 45 | |
| Neuropathy | Correlation Coefficient | −.025 |
| Sig. (2-tailed) | .870 | |
| N | 45 | |
| Structural Disorders **** | Correlation Coefficient | .024 |
| Sig. (2-tailed) | .877 | |
| N | 45 | |
| History of Guillain-Barr-Syndrome (GBS) | Correlation Coefficient | .036 |
| Sig. (2-tailed) | .813 | |
| N | 45 | |
| History of Seizures | Correlation Coefficient | .181 |
| Sig. (2-tailed) | .233 | |
| N | 45 | |
| Postural Orthostatic Tachycardia Syndrome (POTS) | Correlation Coefficient | −.145 |
| Sig. (2-tailed) | .340 | |
| N | 45 | |
| Hypertension (HTN) | Correlation Coefficient | .185 |
| Sig. (2-tailed) | .225 | |
| N | 45 | |
| Opioid Use Disorder | Correlation Coefficient | .176 |
| Sig. (2-tailed) | .247 | |
| N | 45 | |
| OBGYN Disorders***** | Correlation Coefficient | .296 |
| Sig. (2-tailed) | .048* | |
| N | 45 | |
| History of Miscarriage | Correlation Coefficient | −.064 |
| Sig. (2-tailed) | .677 | |
| N | 45 | |
| Endometriosis | Correlation Coefficient | −.064 |
| Sig. (2-tailed) | .677 | |
| N | 45 | |
| Irregular Menses including Spotting | Correlation Coefficient | .144 |
| Sig. (2-tailed) | .344 | |
| N | 45 | |
| Infertility | Correlation Coefficient | .176 |
| Sig. (2-tailed) | .247 | |
| N | 45 | |
| Ovarian Cysts | Correlation Coefficient | .255 |
| Sig. (2-tailed) | .091 | |
| N | 45 | |
| History of Gastroesophageal Reflux Disease (GERD) | Correlation Coefficient | .096 |
| Sig. (2-tailed) | .532 | |
| N | 45 | |
| Cannabidiol (CBD) use | Correlation Coefficient | −.327 |
| Sig. (2-tailed) | .028* | |
| N | 45 | |
| Type 1 Diabetes | Correlation Coefficient | −.185 |
| Sig. (2-tailed) | .225 | |
| N | 45 | |
| Attention Deficit and Hyperactivity Disorder (ADHD) | Correlation Coefficient | −.185 |
| Sig. (2-tailed) | .225 | |
| N | 45 |
AHP + = Acute Hepatic Porphyria positive patients, AHP− = Acute Hepatic Porphyria negative patients
CBD = Cannabidiol
* p < 0.05, Significant
** Autoimmune and rheumatological disorders include Hypothyroidism, Type 1 Diabetes, Fibromyalgia, Myasthenia Gravis
*** Psychiatric Disorders include Major Depressive Disorder, General Anxiety Disorder, Schizophrenia, Psychosis, Hallucinations, Attention Deficit and Hyperactivity Disorder
**** Structural Disorders include joint pains, replacements and surgeries, tendinopathy, Osteoarthritis, trigger finger
***** OBGYN = Obstetrics and Gynecology, symptoms and disorders for this study included: vaginal itching/discomfort unspecified, spotting, irregular periods, ovarian cysts, dyspareunia, infertility, endometriosis, history of hysterectomy, miscarriage, cholestasis of pregnancy, premature delivery, fibroids, dysmenorrhea.
Notably, abdominal pain was the most prevalent symptom in both the positive and negative group (74% and 61%), followed by muscle weakness and/or spasms (52% and 28%). Interestingly, shooting foot/leg pain was only present in the AHP + group, however the difference did not reach statistical significance due to small sample size.
Obstetrics and Gynecology (OBGYN) related symptoms and disorders (for this study, OBGYN disorders and symptoms included: vaginal itching/discomfort unspecified, spotting, irregular periods, ovarian cysts, dyspareunia, infertility, endometriosis, history of hysterectomy, history of miscarriage, cholestasis of pregnancy, premature delivery, fibroids, dysmenorrhea) were reported in 52% of AHP + patients compared to 22% of AHP− patients, showing a significant but weak positive correlation (correlation coefficient: 0.296, p = 0.048) with Acute Hepatic Porphyria. The presence of psychiatric disorders (for this study these included: Major Depressive Disorder, General Anxiety Disorder, Schizophrenia, Psychosis, Hallucinations, Attention Deficit and Hyperactivity Disorder) was also more prevalent in the AHP + group with 52% vs 28% in the AHP− group, showing a positive correlation with AH, however it did not reach statistical significance (p = 0.114)
In contrast, hypothyroidism exhibited a significant negative correlation with AHP (correlation coefficient: −0.347, p = 0.020, and the presence of autoimmune and rheumatological disorders (including Hypothyroidism, Type I Diabetes, Fibromyalgia and Myasthenia Gravis) in general showed a significant negative correlation with AHP (correlation coefficient: −0.580, p < 0.001). Cannabidiol use (CBD) with cyclic vomiting syndrome showed a statistically significant negative correlation with AHP (−0.327, p = 0.028).
Additionally, 22% of AHP− patients had a history of bariatric surgery whereas none of the AHP + patients did, however due to small sample size, the difference was not statistically significant.
The presence of the other symptoms and comorbidities listed did not exhibit any significant correlations with AHP + or − patients as indicated by non-significant p-values.
Genotype Phenotype correlations
Table 7 highlights the distribution of symptoms and comorbid conditions among AHP patients based on their genotypes.
Table 7:: Differences in symptoms or comorbid conditions of AHP+ patients based on genotypes
| Gene mutation | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Unknown | CPOX | HMBS | PPOX | P Value | ||||||
| Count | Column % | Count | Column % | Count | Column % | Count | Column % | |||
| Urinary symptoms** | No | 6 | 86% | 0 | 0% | 6 | 46% | 5 | 100% | 0.019* |
| Yes | 1 | 14% | 2 | 100% | 7 | 54% | 0 | 0% | ||
| Skin blisters and/or rashes | No | 6 | 86% | 1 | 50% | 10 | 77% | 1 | 20% | 0.076 |
| Yes | 1 | 14% | 1 | 50% | 3 | 23% | 4 | 80% | ||
| History of seizures | No | 6 | 86% | 1 | 50% | 10 | 77% | 3 | 60% | 0.560 |
| Yes | 1 | 14% | 1 | 50% | 3 | 23% | 2 | 40% | ||
| Symptoms worse with Menses | No | 2 | 29% | 1 | 50% | 6 | 46% | 3 | 60% | 0.959 |
| Yes | 3 | 43% | 1 | 50% | 5 | 39% | 1 | 20% | ||
| male | 2 | 29% | 0 | 0% | 2 | 15% | 1 | 20% | ||
| OBGYN-related symptoms and disorders*** | No | 1 | 14% | 0 | 0% | 4 | 31% | 3 | 60% | 0.603 |
| Yes | 4 | 57% | 2 | 100% | 7 | 54% | 1 | 20% | ||
| male | 2 | 29% | 0 | 0% | 2 | 15% | 1 | 20% | ||
| Psychiatric Disorders**** | No | 4 | 57% | 0 | 0% | 5 | 39% | 4 | 80% | 0.222 |
| Yes | 3 | 43% | 2 | 100% | 8 | 62% | 1 | 20% | ||
| Major Depressive Disorder (MDD) | No | 6 | 86% | 1 | 50% | 12 | 92% | 4 | 80% | 0.416 |
| Yes | 1 | 14% | 1 | 50% | 1 | 8% | 1 | 20% | ||
| General Anxiety Disorder (GAD) | No | 6 | 86% | 1 | 50% | 9 | 69% | 4 | 80% | 0.780 |
| Yes | 1 | 14% | 1 | 50% | 4 | 31% | 1 | 20% | ||
| Hallucinations | No | 5 | 71% | 2 | 100% | 12 | 92% | 5 | 100% | 0.427 |
| Yes | 2 | 29% | 0 | 0% | 1 | 8% | 0 | 0% | ||
| Schizophrenia | No | 7 | 100% | 2 | 100% | 12 | 92% | 5 | 100% | 1.000 |
| Yes | 0 | 0% | 0 | 0% | 1 | 8% | 0 | 0% | ||
| Neurodevelopmental Disorders including Autism | No | 7 | 100% | 2 | 100% | 11 | 85% | 5 | 100% | 0.741 |
| Yes | 0 | 0% | 0 | 0% | 2 | 15% | 0 | 0% | ||
| Psychosis | No | 6 | 86% | 1 | 50% | 13 | 100% | 5 | 100% | 0.100 |
| Yes | 1 | 14% | 1 | 50% | 0 | 0% | 0 | 0% | ||
| Post traumatic Stress Disorder (PTSD) | No | 7 | 100% | 2 | 100% | 11 | 85% | 5 | 100% | 0.741 |
| Yes | 0 | 0% | 0 | 0% | 2 | 15% | 0 | 0% | ||
| Hypothyroidism | No | 7 | 100% | 2 | 100% | 13 | 100% | 4 | 80% | 0.259 |
| Yes | 0 | 0% | 0 | 0% | 0 | 0% | 1 | 20% | ||
| Hypertension (HTN) | No | 3 | 43% | 2 | 100% | 9 | 69% | 4 | 80% | 0.514 |
| Yes | 4 | 57% | 0 | 0% | 4 | 31% | 1 | 20% | ||
| Guillen-Barr-Syndrome (GBS) | No | 7 | 100% | 2 | 100% | 11 | 85% | 4 | 80% | 0.658 |
| Yes | 0 | 0% | 0 | 0% | 2 | 15% | 1 | 20% | ||
| History of Gastroesophageal Disease (GERD) | No | 5 | 71% | 2 | 100% | 12 | 92% | 5 | 100% | 0.427 |
| Yes | 2 | 29% | 0 | 0% | 1 | 8% | 0 | 0% | ||
| CBD use | No | 6 | 86% | 2 | 100% | 10 | 77% | 4 | 80% | 1.000 |
| Yes | 1 | 14% | 0 | 0% | 3 | 23% | 1 | 20% | ||
| History of Abdominal Surgeries | No | 6 | 86% | 2 | 100% | 9 | 69% | 5 | 100% | 0.565 |
| Yes | 1 | 14% | 0 | 0% | 4 | 31% | 0 | 0% | ||
HMBS = hydroxymethylbilane synthase, CPOX = Coproporphyrinogen Oxidase, PPOX = protoporphyrinogen oxidase
AIP: Acute Intermittent Porphyria, HCP: Hereditary Coproporphyria, VP: Variegate Porphyria, N=total number of patients, CBD = Cannabidiol
* p<0.05 is statistically significant, determined via Fischer’s Exact Test
** Urinary symptoms include discolored urine, urinary urgency, increased urinary frequency, burning/discomfort with urination without presence of UTI, cystitis without presence of infection
*** OBGYN = Obstetrics and Gynecology, symptoms and disorders for this study included: vaginal itching/discomfort unspecified, spotting, irregular periods, ovarian cysts, dyspareunia, infertility, endometriosis, history of hysterectomy, miscarriage, cholestasis of pregnancy, premature delivery, fibroids, dysmenorrhea.
*** Psychiatric Disorders include Major Depressive Disorder, General Anxiety Disorder, Schizophrenia, Psychosis, Hallucinations, Attention Deficit and Hyperactivity Disorder
Patients with the CPOX genotype had the highest percentage (100%) of individuals experiencing urinary symptoms (for the purpose of this study defined as discolored urine, urinary urgency, increased urinary frequency, burning/discomfort with urination without presence of UTI, cystitis without presence of infection), followed by those with the HMBS genotype (54%) and none of the patients with PPOX mutations reported urinary symptoms, with a statistically significant difference of p = 0.019. The presence of skin blisters and rashes showed a similar trend, with the CPOX genotype exhibiting the highest percentage (50%), followed by the HMBS genotype (23%). However, these differences did not reach statistical significance (p = 0.076).
While psychiatric comorbidities were more prevalent in the HMBS genotype (62%) and the CPOX genotype (100%), the PPOX genotype had a markedly reduced prevalence of only 20%, however due to small sample size this difference was not statistically significant (p = 0.222).
Interestingly, there was a higher percentage of patients with the HMBS genotype (92%) who reported experiencing hallucinations compared to other genotypes, although once again due to the small sample size this finding did not reach statistical significance. Autoimmune and rheumatological disorders showed a significant negative correlation with the HMBS genotype (correlation coefficient = −0.369, p = 0.059). The remainder of symptoms and comorbid conditions listed did not significantly vary among the genotypes.
Differences in AHP presentation based on gender
Table 8 presents a comparison of sociodemographic, clinical characteristics (including symptoms and comorbidities), and laboratory measurements between females and males with AHP.
Table 8: Clinical presentation and presenting symptoms of AHP + vs AHP− with gender breakdown
| Gender | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Female | Male | P Value* | |||||||||
| n | Column % | M | SD | n | Column % | M | SD | ||||
| AHP subtype | AIP | 15 | 68% | 3 | 60% | 0.773 | |||||
| HCP | 2 | 9% | 1 | 20% | |||||||
| VP | 5 | 23% | 1 | 20% | |||||||
| Gene mutation | Unknown | 5 | 23% | 2 | 40% | 0.876 | |||||
| CPOX | 2 | 9% | 0 | 0% | |||||||
| HMBS | 11 | 50% | 2 | 40% | |||||||
| PPOX | 4 | 18% | 1 | 20% | |||||||
| Age | 46.2 | 15.7 | 50.4 | 20.7 | 0.612 | ||||||
| Ethnicity | Egyptian | 1 | 5% | 0 | 0% | 1.000 | |||||
| Hispanic | 3 | 14% | 0 | 0% | |||||||
| Other, not Hispanic/Black | 1 | 5% | 0 | 0% | |||||||
| Peruvian | 1 | 5% | 0 | 0% | |||||||
| white | 16 | 73% | 5 | 100% | |||||||
| Symptoms and Comorbidities | Urinary symptoms** | No | 13 | 59% | 4 | 80% | 0.621 | ||||
| Yes | 9 | 41% | 1 | 20% | |||||||
| Skin blisters and rashes | No | 14 | 64% | 4 | 80% | 0.636 | |||||
| Yes | 8 | 36% | 1 | 20% | |||||||
| History of Seizures | No | 18 | 82% | 2 | 40% | 0.091 | |||||
| Yes | 4 | 18% | 3 | 60% | |||||||
| Psychiatric disorders*** | No | 11 | 50% | 2 | 40% | 1.000 | |||||
| Yes | 11 | 50% | 3 | 60% | |||||||
| Major Depressive Disorder | No | 18 | 82% | 5 | 100% | 0.561 | |||||
| Yes | 4 | 18% | 0 | 0% | |||||||
| Generalized Anxiety Disorder | No | 15 | 68% | 5 | 100% | 0.283 | |||||
| Yes | 7 | 32% | 0 | 0% | |||||||
| Hallucinations | No | 20 | 91% | 4 | 80% | 0.474 | |||||
| Yes | 2 | 9% | 1 | 20% | |||||||
| Schizophrenia | No | 21 | 96% | 5 | 100% | 1.000 | |||||
| Yes | 1 | 5% | 0 | 0% | |||||||
| Neurodevelopmental Disorders including Autism | No | 22 | 100% | 3 | 60% | 0.028* | |||||
| Yes | 0 | 0% | 2 | 40% | |||||||
| Psychosis | No | 20 | 91% | 5 | 100% | 1.000 | |||||
| Yes | 2 | 9% | 0 | 0% | |||||||
| Post Traumatic Stress Disorder | No | 20 | 91% | 5 | 100% | 1.000 | |||||
| Yes | 2 | 9.1% | 0 | 0% | |||||||
| Hypothyroidism | No | 21 | 96% | 5 | 100% | 1.000 | |||||
| Yes | 1 | 5% | 0 | 0% | |||||||
| Hypertension | No | 16 | 73% | 2 | 40% | 0.295 | |||||
| Yes | 6 | 27% | 3 | 60% | |||||||
| History of Guillain-Barr Syndrome | No | 19 | 86% | 5 | 100% | 0.605 | |||||
| Yes | 3 | 14% | 0 | 0% | |||||||
| History of GERD | No | 20 | 91% | 4 | 80% | 0.474 | |||||
| Yes | 2 | 9% | 1 | 20% | |||||||
| CBD use | No | 19 | 86% | 3 | 60% | 0.221 | |||||
| Yes | 3 | 14% | 2 | 40% | |||||||
| History of abdominal surgeries | No | 17 | 77% | 5 | 100% | 0.547 | |||||
| Yes | 5 | 23% | 0 | 0% | |||||||
AHP + = Acute Hepatic Porphyria positive patients, AHP− Acute Hepatic Porphyria negative patients
HMBS = hydroxymethylbilane synthase, CPOX = Coproporphyrinogen Oxidase, PPOX = protoporphyrinogen oxidase, AIP: Acute Intermittent Porphyria, HCP: Hereditary Coproporphyria, VP: Variegate Porphyria, n = total number of patients, GERD = Gastroesophageal Disease, CBD = Cannabidiol
* p < 0.05, Significant, determined via Fischer’s Exact Test and Independent Samples t-test
** OBGYN = Obstetrics and Gynecology, symptoms and disorders for this study included: vaginal itching/discomfort unspecified, spotting, irregular periods, ovarian cysts, dyspareunia, infertility, endometriosis, history of hysterectomy, miscarriage, cholestasis of pregnancy, premature delivery, fibroids, dysmenorrhea.
*** Urinary symptoms include discolored urine, urinary urgency, increased urinary frequency, burning/discomfort with urination without presence of UTI, cystitis without presence of infection
**** Psychiatric Disorders include Major Depressive Disorder, General Anxiety Disorder, Schizophrenia, Psychosis, Hallucinations, Attention Deficit and Hyperactivity Disorder
Across both AHP + and AHP− cohorts, females constituted 68%, while males accounted for 32%. The distribution of the subtypes of AHP (AIP, HCP, VP) did not significantly differ between females and males (p = 0.773). The most common gene mutations were HMBS for 50% of females and 40% of males. The mean age was 46.2 years (SD = 15.7) for females and 50.4 years (SD = 20.7) for males, without a statistically significant difference (p = 0.612). The majority of patients identified as White for both genders (73% of females, 100% of males). There were no statistically significant differences between females and males in clinical presentation (symptoms and/or comorbidities), except for the presence of neurodevelopmental disorders, which were present only in the male AHP + cohort (40% of males, p = 0.028). There was a trend towards abdominal pain being more common among females (77%) vs males (60%) however the difference did not reach statistical significance.
Discussion
The study aimed to expand upon existing clinical recommendations to reduce the diagnostic delay and improve rates of diagnosis of AHP in our diverse community. We investigated to what extent demographics, symptomatic presentation and comorbidities play a role in predicting AHP and what combination of testing could lead to the highest rates of diagnosis. Finally, we attempted to lay the groundwork for future investigations into potential genotype-phenotype correlations and phenotypic presentation differences based on ethnicity to further enable precision testing and treatment in the future.
Demographics – ethnicity and gender
Per the most recent population census conducted in California in 2022 (ref. 14), the largest ethnic group in Los Angeles County, the home of UCLA Health, is Hispanic or Latino (49%), followed by White (25%), Asian (16%) and Black or African American (9%) with 50% identifying as female and 34% identifying additionally as foreign persons/non-citizens or residents. There is a significant mismatch between the number of White patients (78%) evaluated for AHP at UCLA in comparison to any other ethnicity considering that Whites represent a minority of the population in Los Angeles County. Historically, it has always been assumed that AHP is more common in White/Caucasian and female patients (ref. 3, ref. 13, ref. 15), however there are several studies from other countries indicating a surprising symptomatic prevalence among non-White ethnicities (ref. 10, ref. 11) with atypical presentations (ref. 11, ref. 12) which is currently not reflected in the US based AHP evaluation guidelines.
Additionally, at least the total number of evaluated patients from Hispanic and male demographics should be higher to more closely reflect the local population which is comprised of nearly half Hispanic/Latinos. However, in our study, only ~ 9% of patients evaluated for AHP were Hispanic and all of them tested positive for AHP and only 22% of the entire patient cohort evaluated for AHP were male and half of them tested positive for AHP. While it is possible that AHP simply truly is not as common in non-White ethnicities and males, it is interesting to note the large percentage of positive patients of the non-White (100%) and male patients tested (50%), in comparison to the positive patient percentage of the evaluated White patients. Analysis of potential differences between AHP + males and females also suggested that men may present atypically with less abdominal pain than women (60% prevalence vs 77%) and statistically significantly more neurodevelopmental comorbidities than women regardless of the AHP subtype.
Ensuring increased testing of males and non-White patients may present an opportunity for intervention to increase diagnostic rates of AHP in our community and necessitates further studies in clinical and laboratory presentations of these specific patient subgroups to ensure updated presentation in our clinical AHP guidelines.
Whom to test – symptomatic presentation and comorbidities
Our study didn’t show any statistically significant differences in symptomatic presentation between the positive and negative cohort, partially due to small sample size but also due to these symptoms being seen in other much more common disease states as well; We could conclude that symptoms alone may not be the key factor in deciding which patients to test for AHP and that symptom onset especially following a seizure, pregnancy, severe illness, surgery or an accident, and comorbidities such as psychiatric disorders and OBGYN-related disorders should raise suspicion and lower threshold for AHP testing.
In contrast, Hypothyroidism and Type 1 Diabetes were strongly negatively correlated with AHP, perhaps reducing the probability that the patient would test positive for AHP. There was also a trend of patients with a history of bariatric surgery presenting with AHP like symptoms, however none of them tested positive for AHP, but this warrants awareness. Lastly, CBD use with cyclic vomiting syndrome was also associated with the AHP negative group, lowering the suspicion for AHP in those patients. Differential diagnoses to evaluate against in AHP-presenting patients may be malignancies of the GI system and the endocrine system, autoimmune and rheumatological conditions such as Systemic Mastocytosis, Connective Tissue Disorders, Dermatomyositis, and other neurological conditions such as CIDP, justifying the commonly seen involvement of Neurology and Rheumatology in the evaluation of AHP patients.
The role of OBGYN in AHP
Considering the statistically significant increased prevalence of certain OBGYN-related disorders in the AHP + group in comparison to the AHP− group in our study, and a number of female AHP + patients reporting symptom onset or worsening with pregnancy to the extent of elective termination due to symptom intolerability and adverse outcomes for their newborn if they did carry to term, it is surprising that not more patients are being referred from and/or evaluated by OBGYN (only 1 patient) and Psychiatry (0 patients). While a significant increase in especially pregnancy related complications and comorbidities in AIP subtype patients particularly is well documented in other countries, there are no such studies in the US: Sweden has studied the correlations between infertility, miscarriage and AIP for decades (ref. 4), equally, studies from Greece (ref. 5), China (ref. 8), Bosnia (ref. 16) and India (ref. 17) have documented the increased prevalence of spontaneous abortions (ref. 5, ref. 8), symptom worsening with pregnancy to the point of elective terminations (ref. 17), significant adverse maternal and infant outcomes in pregnancy of AIP patients such as maternal-infant mortality rates of up to 42%, low infant birth weight and maternal hypertension (ref. 5) and AIP mimicry of pre-eclampsia and eclampsia in patients with AIP (ref. 8). As AHP is the great mimicker of so many other disorders, it is likely simply misdiagnosed within OBGYN, however considering the significantly increased adverse outcomes that are well documented, we might want to consider testing for AHP as part of the standard prenatal genetic screens and routine monthly urinalysis conducted at OBGYN offices throughout the course of pregnancy, at least for the duration of a study to assess prevalence.
The role of Psychiatry in AHP
Similar to OBGYN, Psychiatry is an underrepresented field of referral and diagnosis source for AHP; in fact, not a single patient was referred to further care or diagnosed by a psychiatrist in our study, despite the high prevalence of psychiatric disorders in our study. The increased prevalence of psychiatric disorders and the mimicry of specifically AIP causing misdiagnosis of Schizophrenia, Psychosis, Hallucinations, Bipolar I, Major Depressive Disorder and General Anxiety Disorder has already been studied and is well documented (ref. 18), even in the US (ref. 19), where previous studies confirm our results of a disproportionate prevalence of Hallucinations (especially in the HMBS/AIP subtype), Schizophrenia, Psychosis, MDD and GAD in our AHP + patient population.
Extending research and clinical involvement of AHP into the field of Psychiatry appears to potentially be beneficial in yielding higher rates of AHP diagnosis and reducing misdiagnosis.
Laboratory testing recommendations
The 2021 study by Karl Anderson et al (ref. 15) discussed the current testing recommendations for AHP, preferring urinary PBG over ALA as the gold standard in significance, however it was suggested to review these recommendations every 2–3 years. In our patient cohort, the most common first line of testing was also urinary ALA and/or PBG, while Whole Blood (WB) Porphyrin testing is second in frequency, without showing any actual diagnostic ability to distinguish between AHP + and AHP− patients per our data from our patient cohort. Across both genders, if we only consider ALA and/or PBG then only 56% of patients would have tested positive for AHP; overlooking the 17% of AHP + patients who tested negative for ALA/PBG but were positive for genetic testing. Furthermore, using only one value (ALA or PBG) would have only identified 17% of positive patients. Adding Total Urinary Porphyrins, Coproporphyrins and Uroporphyrins to the biochemistry testing increased our positive patient cohort from 56–74%, increasing the chance of diagnosis. The likelihood of an AHP patient testing positive based on urinary metabolites alone seems to be 50% regardless of whether they are tested during an acute attack; In our study half of all AHP + patients who were tested during an attack tested negative for urinary metabolites while half of patients tested outside of an attack also tested positive for urinary metabolites.
If we had only determined AHP status by available genetic testing, we would have captured 74% of all AHP + patients, higher than standard biochemistry (urinary ALA/PBG, 56%) alone. However, genetic testing still carries a stigma with many populations (ref. 6) and due to the relatively low clinical penetrance of around 10% (01, Bissel et al), expanding genetic tests to wider screens would potentially yield plenty of false positives.
Variants of Unknown Significance (VUS) may result in a patient not receiving the correct diagnosis (ref. 20), which plagues the diagnosis of many rare diseases, including AHP, and with the increasing ease and access that patients have to ‘at home’ genetic tests, we are likely to see more and more self-referred patients, many of may not actually have a Porphyria, or alternatively, may have subtypes or similar diseases we have not yet discovered. One of the most challenging things in clinical practice is working a patient who presents with a genetic test result in hand but who does not have a typical clinical or biochemical metabolite trail to cement the diagnosis. Even among patients who have a known pathogenic mutation in HMBS, the gene associated with AIP, not all of them experience an acute pain attack, these are known as latent porphyrics and their natural history remains poorly studied. One potential advantage of adding a targeted genetic screen for patients who present for a Porphyria evaluation may be to identify such latent porphyrics and could add substantially to our understanding of the natural history of Porphyrias. However, it is advisable to undertake this only in the setting of an academic research protocol. Clinicians who work with Porphyria know that when discussing a lower potential of having a diagnosis of AHP (e.g. when there is either just a VUS in an AHP gene or when there are no typical symptoms or elevated urinary metabolites) is more of an art than a science. There are clearly environmental and physiologic factors–dehydration, starvation, anemia, certain medications—that impact whether a patient with a bona de mutation in an AHP gene will develop an acute attack (ref. 1). Given that clinical penetrance of individual genetic mutations can be quite variable (ref. 1), it is highly likely that there are other, unidentified modifier genes and pathways. In our experience at UCLA, only about 1 in 30 patients who come for a Porphyria evaluation actually end up getting a Porphyria diagnosis. This is quite often disappointing, frustrating, and confusing for them as many patients have already been to other doctors, as well as the internet, and are pre-convinced that they have a Porphyria. They may have been told by other practitioners that they ‘most likely’ or ‘definitely’ have a Porphyria, yet the only evidence is that they have mild, non-specific elevations in the urinary metabolites or a VUS in an AHP related gene.
Focusing on both provider and patient education about AHP should help sharpen diagnostic accuracy, improve clinical care and alleviate patient anxieties. Medical providers simply need to be aware of AHP as part of the differential diagnosis of recurrent acute pain attacks (especially in women of childbearing age) and know how to screen for AHP in the appropriate clinical context. Our cohort suggests that outreach to practitioners in the fields of mental health and obstetrics/gynecology may bear the most fruit and give them a resource of where to turn if they think about AHP but do not know what to do next. Providers also need to have a basic understanding of how to interpret ‘positive’ urine metabolite results. AHP is ruled in when urine metabolites that are elevated 10–100 times during a time of acute symptoms, whereas non-specific elevations of only two to three-fold are likely the result of dietary, processing, or other environmental factors. Normal or trivially elevated urine metabolites during an acute pain attack rules out AHP and alternative diagnoses for the acute pain should be sought. On the other hand, if urine metabolites are normal or trivially elevated during an asymptomatic period this does not rule out AHP. So, it is very important that we ‘leave the door’ open for this last subset of patients and avoid dismissing or minimizing patients’ concerns and knowing that there are some patients out there who are latent porphyrics. We typically give these patients a prescription for appropriate urine tests to keep in their purse or wallet so that, in the future, if they present to a healthcare facility with symptoms suggestive of AHP, they can give the provider the correct tests to order. We also encourage the patients to invite their acute care provider to contact us to discuss the case, further testing, and supportive care.
Treatment recommendations
The majority of patients are still being treated with Panhematin (ref. 6, ref. 15), even in our cohort, however among our patients this treatment appears to be effective primarily for acute relief of neurological symptoms and improvement in biochemistry, with limited or no effect on abdominal pain. Further, it appears to lose its efficacy over time altogether.
A surprising number of patients (15%) are still being treated with Dextrose, which has led to significant self-reported weight gain in the dextrose-treated patients in our cohort.
Only about a quarter of our patients are treated with Givosiran, most likely due to difficulties in providers being aware of the treatment, the prior authorization process required by insurance, and the lack of knowledge that financial assistance programs are available for patients who are under or uninsured. It is also not yet known if Givosiran will work long term without losing efficacy, and if it is equally effective for all genotypes and ethnicities; further studies are needed. In our patient cohort, only 1 patient was unable to tolerate Givosiran (HMBS, Latino).
There also remains the question whether to treat asymptomatic AHP + patients. It is unknown at this time to what extent, if at all, clinical presentation is correlated with developing subsequent comorbidities known to affect AHP + patients, such as liver cancer. Long term studies following asymptomatic carriers’ health outcomes are needed.
Strengths and Weaknesses
Patients were excluded from this study if insufficient EMR data was present leading to an inability to determine the true diagnosis of the patient; this could potentially mean that patients were left out of the study who in fact did have AHP or have been ruled out at another health center. Similarly, information was collected from both the standardized EMR documentation fields as well as the qualitative free note sections in the EMR, which is subject to the individual interpretation of the original recording physician, which played a role in determining whether a patient was suffering from an acute AHP attack at the time of the urinalysis. However, conversely, this in-depth manual chart review included not only the standardized EMR fields that can be computationally queried, but also the subjective qualitative data including attached pdfs from other health institutions enriched the details of this study, giving us a greater in depth look into Acute Hepatic Porphyria with potential to find new patterns that we otherwise would have not discovered.
Despite the small sample size, and therefore increased likelihood of Type II error, we hope this study can lay the groundwork for future more detailed investigations of phenotype-genotype correlations as well as clinical presentations in non-white and non-female patients to overall improve the rate of diagnosis of this disease, leading to earlier treatment and reducing unnecessary suffering and collateral comorbidities caused by AHP, especially in traditionally underserved patient populations.
Summary of Recommendations for Clinical Practice
Whom to test?
- AHP may present differently across age groups, sexes and ethnicities.
- Family history is still a strong predictor of AHP.
- Hispanic patients are equally as likely to suffer from AHP as White patients based on our study.
- Male patients may present atypically without classic abdominal pain, but with greater frequency of neurological and neurodevelopmental disorders including Autism.
- Patients with the following conditions may be less likely to have AHP.
- T1DM
- History of bariatric surgery
- History of CBD use with cyclic vomiting syndrome
How to test
- For the highest probability of a true positive test, include ideally both genetic screening as well as biochemical testing normalized to creatinine consisting of urinary ALA, PBG, Total Porphyrins, Coproporphyrins and Uroporphyrins regardless of the subtype of AHP suspected and irrespective of symptomatic presentation of the patient at the time of sample collection.
- Whole Blood Porphyria testing has shown no ability to distinguish between our negative and positive patients.
How to treat
- Avoidance of environmental and / or physiologic triggers (dehydration, starvation, anemia, and certain potentially Porphyria-triggering medications).
- IV dextrose to abort acute mild attacks can be very effective in the emergent care settings.
- Panhematin/Hemin is a commonly used acute abortive treatment, however its limited availability in non-tertiary care settings and intricacies in its administration adds access difficulty. Whether tachyphylaxis to hemin might occur for some AHP patients in the long-term needs to be further investigated.
- Givosiran (Givlaari) is a new siRNA-based treatment that effectively keeps AHP patients from experiencing further attacks of abdominal pain and avoids the need for hospitalization. Both Givosiran and hemin are very expensive and should only be used in consultation with a practitioner who has experience; financial assistance is available through the manufacturer Alnylam for patients who are unable to obtain coverage for the medication.
- The bene t of treating asymptomatic AHP + patients is unknown but considering the increased prevalence of certain comorbidities in the AHP + group such as OBGYN and psychiatric disorders, further studies are warranted to investigate this topic. The safety pro les for current treatment options are well established and this may warrant a low threshold to initiate treatment based on provider discretion after risk-bene t analysis.
Conclusions
This study underscores the critical need for improved diagnostic approaches and increased awareness of Acute Hepatic Porphyria (AHP) among diverse patient populations. The significant underdiagnosis in non-White and male patients suggests that current clinical guidelines and diagnostic practices may be insufficiently inclusive of these groups. Our findings advocate for broader and more inclusive testing criteria, incorporating genetic and biochemical tests beyond traditional urinary metabolites to enhance diagnostic accuracy. Additionally, increased education and outreach efforts targeted at healthcare providers in fields such as psychiatry and obstetrics/gynecology will further improve diagnostic rates. Future research should focus on genotype-phenotype correlations and the development of tailored treatment strategies to address the unique needs of diverse patient demographics. By addressing these gaps, we can move towards more equitable healthcare outcomes and improve the quality of life for all individuals affected by AHP.
References
- HL Bonkovsky, P Siao, Z Roig, ET Hedley-Whyte, TJ Flotte. Case records of the Massachusetts General Hospital. Case 20–2008. A 57-year-old woman with abdominal pain and weakness after gastric bypass surgery.. N Engl J Med., 2008. [PubMed]
- DA Jaramillo-Calle. Porphyria.. N Engl J Med., 2017
- M Balwani, B Wang, KE Anderson, JR Bloomer, DM Bissell, HL Bonkovsky. Acute hepatic porphyrias: Recommendations for evaluation and long-term management.. Hepatology., 2017. [PubMed]
- C Andersson, E Innala, T Backstrom. Acute intermittent porphyria in women: clinical expression, use and experience of exogenous sex hormones. A population-based study in northern Sweden.. J Intern Med., 2003. [PubMed]
- A Farfaras, F Zagouri, G Zografos, A Kostopoulou, TN Sergentanis, S Antoniou. Acute intermittent porphyria in pregnancy: a common misdiagnosis.. Clin Exp Obstet Gynecol., 2010. [PubMed]
- M Marcacci, A Ricci, C Cuoghi, S Marchini, A Pietrangelo, P Ventura. Challenges in diagnosis and management of acute hepatic porphyrias: from an uncommon pediatric onset to innovative treatments and perspectives.. Orphanet J Rare Dis., 2022. [PubMed]
- H Lenglet, C Schmitt, T Grange, H Manceau, N Karboul, F Bouchet-Crivat. From a dominant to an oligogenic model of inheritance with environmental modifiers in acute intermittent porphyria.. Hum Mol Genet., 2018. [PubMed]
- J Zhang, Y Hu, J Zheng, J Gao, H Hou, N Liu, Y Wang. Treatment of acute intermittent porphyria during pregnancy and posterior reversible encephalopathy syndrome after delivery: A case report.. Exp Ther Med., 2017. [PubMed]
- R Sachdev, KR Haldiya, AK Dixit. Acute Intermittent Porphyria in a Kumhar community of Western Rajasthan.. J Assoc Physicians India., 2005. [PubMed]
- S Kumar, A Bhalla, N Sharma, DP Dhibar, S Kumari, S Varma. Clinical, Biochemical Characteristics and Hospital Outcome of Acute Intermittent Porphyria Patients: A Descriptive Study from North India.. Ann Indian Acad Neurol., 2017. [PubMed]
- A Jara-Prado, P Yescas, FJ Sanchez, C Rios, R Garnica, E Alonso. Prevalence of acute intermittent porphyria in a Mexican psychiatric population.. Arch Med Res., 2000. [PubMed]
- HL Bonkovsky, VC Maddukuri, C Yazici, KE Anderson, DM Bissell, JR Bloomer. Acute porphyrias in the USA: features of 108 subjects from porphyrias consortium.. Am J Med., 2014. [PubMed]
- B Bhasuran, K Schmolly, Y Kapoor, NL Jayakumar, R Doan, J Amin. Reducing diagnostic delays in Acute Hepatic Porphyria using electronic health records data and machine learning: a multicenter development and validation study.. medRxiv., 2023
- 14.<OMII-Medicare-Databook-February-2022.pdf> .
- KE Anderson, R Lobo, D Salazar, M Schloetter, G Spitzer, AL White. Biochemical Diagnosis of Acute Hepatic Porphyria: Updated Expert Recommendations for Primary Care Physicians.. Am J Med Sci., 2021. [PubMed]
- A Cerovac, A Brigic, D Softic, A Barakovic, S Adzajlic. Uncontrolled Acute Intermittent Porphyria as a Cause of Spontaneous Abortion.. Med Arch., 2020. [PubMed]
- MT Sahu, S Rajaram, AK Saxena, N Goel, S Ghumman. Medical termination of pregnancy in acute intermittent porphyria.. Gynecol Obstet Invest., 2006. [PubMed]
- L Duque-Serrano, L Patarroyo-Rodriguez, D Gotlib, JC Molano-Eslava. Psychiatric Aspects of Acute Porphyria: a Comprehensive Review.. Curr Psychiatry Rep., 2018. [PubMed]
- K Burgovne, R Swartz, J Ananth. Porphyria: reexamination of psychiatric implications.. Psychother Psychosom., 1995. [PubMed]
- M Alfadhel, N Saleh, H Alenazi, H Baffoe-Bonnie. Acute intermittent porphyria caused by novel mutation in HMBS gene, misdiagnosed as cholecystitis.. Neuropsychiatr Dis Treat., 2014. [PubMed]
