Genes Co-Expressed with ESR2 Influence Clinical Outcomes in Cancer Patients: TCGA Data Analysis
Abstract
ERβ has been assigned a tumor suppressor role in many cancer types. However, as conflicting findings emerge, ERβ’s tissue-specific expression and functional role have remained elusive. There remains a notable gap in compact and comprehensive analyses of ESR2 mRNA expression levels across diverse tumor types coupled with an exploration of its potential gene network. In this study, we aim to address these gaps by presenting a comprehensive analysis of ESR2 transcriptomic data. We distinguished cancer types with significant changes in ESR2 expression levels compared to corresponding healthy tissue and concluded that ESR2 influences patient survival. Gene Set Enrichment Analysis (GSEA) distinguished molecular pathways affected by ESR2, including oxidative phosphorylation and epithelial–mesenchymal transition. Finally, we investigated genes displaying similar expression patterns as ESR2 in tumor tissues, identifying potential co-expressed genes that may exert a synergistic effect on clinical outcomes, with significant results, including the expression of ACIN1, SYNE2, TNFRSF13C, and MDM4. Collectively, our results highlight the significant influence of ESR2 mRNA expression on the transcriptomic landscape and the overall metabolism of cancerous cells across various tumor types.
Article type: Research Article
Keywords: cancer, ESR2, bioinformatic, gene expression, transcriptomic, survival
Affiliations: Department of Histology and Embryology, Doctoral School, Poznan University of Medical Sciences, Święcickiego 6 Street, 60-781 Poznań, Poland; julia.lipowicz@student.ump.edu.pl; Department of Histology and Embryology, Poznan University of Medical Sciences, Święcickiego 6 Street, 60-781 Poznań, Poland
License: © 2024 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/ijms25168707 | PubMed: 39201394 | PMC: PMC11354723
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (5.8 MB)
1. Introduction
Estrogen receptor β (ERβ) is one of the three predominant 17β-estradiol (E2) receptors in cells, along with estrogen receptor α (ERα) and G-protein-coupled estrogen receptor 1 (GPER1) [ref. 1]. ERβ and ERα are members of the nuclear receptor superfamily, while GPER1 is present on cell membranes [ref. 2,ref. 3]. ERβ is encoded by the gene ESR2, located on chromosome 14, containing eight exons. As ESR2 is susceptible to alternative splicing, it can be transcribed into six isoforms, with ERβ1 characterized as the wild-type, full-length (530-amino-acids long, 59 kDa) protein [ref. 3,ref. 4,ref. 5]. For a full review and comparison of different ERs and their structure, we recommend perusing studies by Božović et al. [ref. 3] and Jia et al. [ref. 1].
ERβ displays diverse functions through genomic (nuclear, otherwise called classical) and non-genomic (rapid, extranuclear) pathways [ref. 6]. The classical pathways are initiated by the ligand-bound ERβ-E2 complex. This complex translocates into the nucleus, where it orchestrates the transcriptional regulation of target genes. The recognition of target genes can be mediated through the estrogen response element (ERE) canonical binding site (5′-GGTCAnnnTGACC-3′), a genomic target for ERβ, or via interaction with other transcription factors, including AP-1 and Sp-1 [ref. 6,ref. 7]. On the other hand, non-genomic pathways are carried out through rapid cellular mechanisms, including the protein–kinase signaling cascade or the cross-activation of growth factor signaling, possibly even in the absence of ERβ ligands [ref. 2,ref. 6,ref. 8].
ERβ is primarily expressed in the colon, kidney, lung, male reproductive tissues, and central nervous system [ref. 1]. While its precise physiological role remains incompletely understood, studies have suggested its role in immune response, the cardiovascular system, and prostate function [ref. 8,ref. 9]. In cancer, ERβ has been assigned a tumor suppressor role in many tumors, although exceptions exist, notably in lung cancer [ref. 6,ref. 8,ref. 10,ref. 11]. Nonetheless, the literature presents numerous inconsistencies. In breast cancer, ERβ expression levels were almost entirely lost compared to healthy tissue [ref. 12], and its re-expression inhibited breast cancer cell proliferation and upregulated apoptosis [ref. 13]. However, conflicting findings suggest that ERβ may enhance cell proliferation in breast cancer, with its expression correlating with poorer prognosis in some cases [ref. 14,ref. 15,ref. 16]. In colon cancer, most authors pointed to the protective role of ERβ. Animal models demonstrated that ERβ knock-down in mice leads to an increased number and size of intestinal adenomas, whereas treatment with ERβ-selective agonists presented the opposite effect [ref. 17,ref. 18]. Prostate cancer research further underscores the complexities surrounding ERβ. Multiple studies have identified it as a tumor suppressor or oncogene [ref. 19,ref. 20].
As described above, since ERβ discovery in 1996 [ref. 21], its tissue-specific expression and functional role have remained contentious due to conflicting findings across various studies. Andersson et al. [ref. 22] and Nelson et al. [ref. 23] investigated one of the reasons behind the inconsistencies in research, namely, antibody specificity. Their analysis revealed that only a fraction of commercially available antibodies are suitable for ERβ-targeted experiments, with PPZ0506 being the most specific [ref. 22,ref. 23]. Therefore, the validity of numerous publications may be compromised by the use of non-specific antibodies, particularly in studies based solely on proteomic data. Discrepancies between transcriptomic and proteomic research further underscore this challenge [ref. 7,ref. 22,ref. 23], likely partly due to non-specific protein detection. Further complications come from isoforms of ERβ, which diverge only through the domain encoded by the eighth exon [ref. 3,ref. 4,ref. 5]. Additionally, ERβ’s ability to localize within various cellular compartments, driven by both genomic and non-genomic activities [ref. 6], adds another layer of complexity [ref. 24,ref. 25].
In the literature, while there are studies that provide insight into ESR2/ERβ expression profiles across different tissues and cancer types, there remains a notable gap in compact and comprehensive analyses of ESR2 mRNA expression levels across diverse tumor types coupled with an exploration of its potential functional implications. Furthermore, as a transcription factor, ERβ plays a considerable role in regulating cellular metabolism through different target genes, of which not all have been fully elucidated.
In this study, we aim to address these gaps by presenting a comprehensive analysis of ESR2 transcriptomic data. We performed a series of bioinformatic analyses to specify ERβ’s role in various tumor types classified by The Cancer Genome Atlas (TCGA). We examined ESR2 expression levels in tumors and corresponding healthy tissues to identify tumors potentially influenced by ERβ. Moreover, we carried out correlations between ESR2 expression and tumor stage and grade. Furthermore, in selected tumor types, we extended our analysis to investigate overall survival (OS) and disease-free survival (DFS) in the context of ESR2 expression. We also employed bioinformatic tools like Gene Set Enrichment Analysis (GSEA, software version 4.3.3.) to distinguish molecular pathways affected by this receptor. Finally, we investigated genes displaying similar expression patterns to ESR2 in tumor tissues, identifying potential co-expressed genes that may synergistically affect OS and DFS outcomes. Collectively, our results highlight the significant influence of ESR2 mRNA expression on the transcriptomic landscape and overall metabolism of cancerous cells across various tumor types.
2. Results
2.1. Cancer Tissue and Corresponding Healthy Tissue Present Different ESR2 Expression Levels in Selected Cancer Types
To establish whether ESR2 expression varies between cancerous and normal tissue, we analyzed transcriptomic data from the TIMER2.0 database. Figure 1a illustrates the differential expression of ESR2 across selected tumor types compared to their corresponding normal tissue (tumor name abbreviations can be found in Abbreviations). Analysis of 22 tumor types based on TIMER2.0 data revealed 14 TCGA tumor types with significant differences (p-value < 0.05) with regard to ESR2 expression between tumor and corresponding normal tissue (Figure 1a). Of these tumor types, eight exhibited higher mRNA expression levels in normal tissue, including breast invasive carcinoma (BRCA) (p-value < 0.001), colon adenocarcinoma (COAD) (p-value < 0.001), and kidney chromophobe (KICH) (p-value < 0.001) (Figure 1a). Conversely, five tumor types displayed higher expression in cancerous tissue, including cholangio carcinoma (CHOL) (p-value < 0.001) and head and neck squamous cell carcinoma (HNSC) (p-value < 0.001) (Figure 1a). Additionally, a significant difference was found between human papilloma virus (HPV)-positive and HPV-negative HNSC samples (p-value < 0.001), with the former presenting higher ESR2 expression levels (Figure 1a).
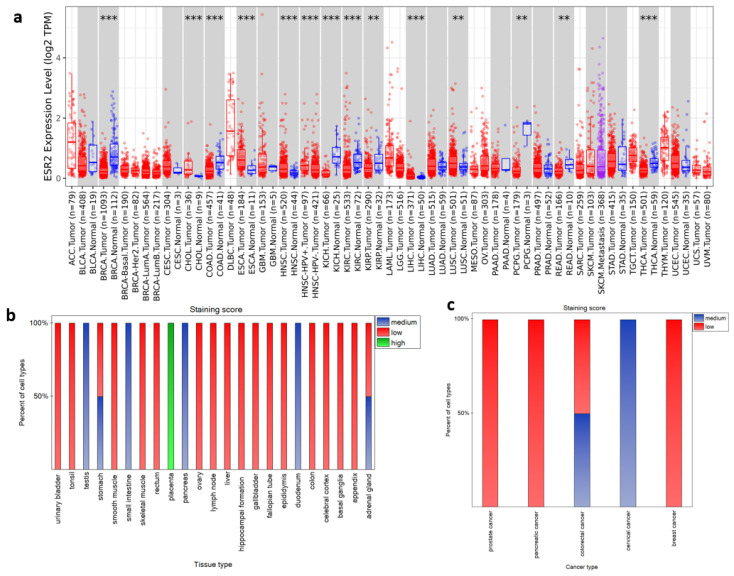
To further describe our gene of interest, we explored the Human Protein Atlas database. Proteomic data from HPA’s immunostaining (antibody CAB079300) (Figure 1b,c) included 49 normal tissue samples, with detailed information regarding cell types. Data for 9 tissue types were not available and 17 tissue types did not exhibit positive staining in any cell type, including those from breast, endometrium, kidney, and lung (summary of tissue samples presented in HPA ENSG00000140009-ESR2/tissue). Still, in available proteomic data, we found high staining scores for the placenta and medium staining scores for the duodenum, pancreas, small intestine, and testis (Figure 1b). Adrenal glands and stomach tissue presented medium and low staining scores, depending on cell types, as shown in Figure 1b. For specific cell types in different tissue samples and their staining scores, we present Supplementary Figure S1. Furthermore, the HPA database provides immunostaining data on pathological tissues (summary of pathological tissue samples presented in HPA ENSG00000140009-ESR2/pathology). Out of 19 cancerous tissue samples, 14 had negative ERβ antibody staining, including lung cancer, melanoma, renal cancer, and testis cancer (Figure 1c). Furthermore, cervical cancer was classified as a medium staining score, whereas colorectal cancer had varying degrees of staining, ranging from low to medium across different samples (Figure 1c).
Unfortunately, samples used in Human Protein Atlas proteomic data for immunochemistry do not correspond to samples in the TCGA datasets. RNA-seq data from pathological tissues available in the Human Protein Atlas are likewise harnessed from the TCGA repository. Therefore, they do not correspond to proteomic data. Human Protein Atlas does provide HPA RNA-seq results regarding normal tissue samples. However, the number of samples per tissue is not sufficient to calculate the correlation. Therefore, further analyses were carried out based on available TCGA transcriptomic data.
2.2. ESR2 Expression Correlates with the Grade and Stage of Several Tumor Types
To determine whether ESR2 expression level could be described as a potential biomarker in cancer, we searched the TISIDB database for correlations with the grade and stage of tumors.
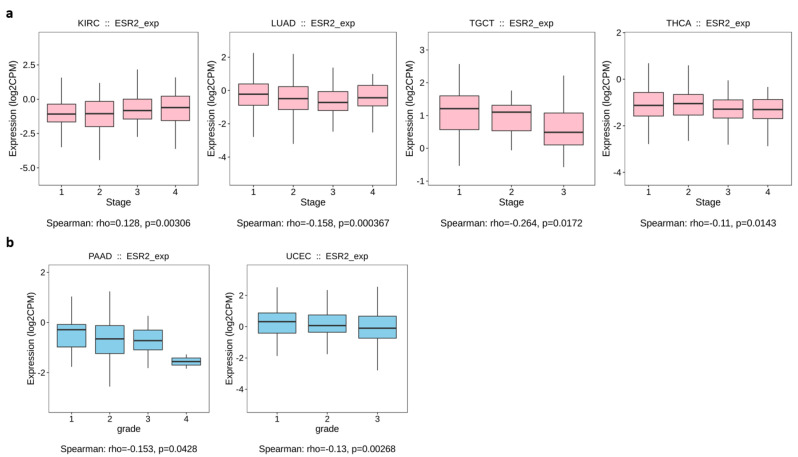
Out of 24 cancer types for which data were available on TISIDB, only 4 yielded significant results (p-value < 0.05) for the correlation between ESR2 expression and tumor stage (Figure 2). We found a positive correlation for kidney renal clear cell carcinoma (KIRC) (p-value = 0.00306), in which a higher ESR2 expression level correlated with advanced tumor stage, as shown in Figure 2a. A negative correlation was found for lung adenocarcinoma (LUAD) (p-value = 0.000367), testicular germ cell tumor (TGCT) (p-value = 0.0172), and thyroid carcinoma (THCA) (p-value = 0.0143), showing lower ESR2 expression levels correlated with tumor stage (Figure 2a). Furthermore, we observed a significant correlation between ESR2 expression and tumor grade in two out of eight cancer types. Specifically, pancreatic adenocarcinoma (PAAD) (p-value = 0.0428) and uterine corpus endometrial carcinoma (UCEC) (p-value = 0.00268) presented negative correlations; hence, lower ESR2 expression correlated with tumor grade, as shown in Figure 2b. The TISIDB database did not yield other significant results for correlations of ESR2 expression with the grade or stage of tumors.
2.3. ESR2 Expression Level Affects OS and DFS in Various Cancer Types
To review if ESR2 may be considered a prognostic factor, we examined the influence of ESR2 mRNA expression level on overall survival (OS) and disease-free survival (DFS) across various cancer types within the TCGA database (Figure 3). We noticed that ESR2 presented both positive and negative prognostic capacity, in some cases with regard to patient sex. Tumors that presented a reliance on ESR2 expression level in terms of OS and DFS were described in the literature as having a sex bias in occurrence and clinical outcomes [ref. 26,ref. 27,ref. 28,ref. 29,ref. 30,ref. 31]. To delineate patients’ subgroups based on ESR2 expression levels, we employed the Evaluate Cutpoints R application algorithm [ref. 32], facilitating the classification of individuals into “high ESR2 expression level” and “low ESR2 expression level” groups. Subsequently, we conducted OS and DFS analyses based on this division. Interestingly, 20 out of 29 cancer types presented significant results (p-value < 0.05) when we analyzed ESR2’s influence on OS. Additionally, 12 cancer types yielded significant results with regard to ESR2’s influence on DFS. Representative data are shown (Figure 3); for every significant result plotted, we present Supplementary Figures S2 and S3.
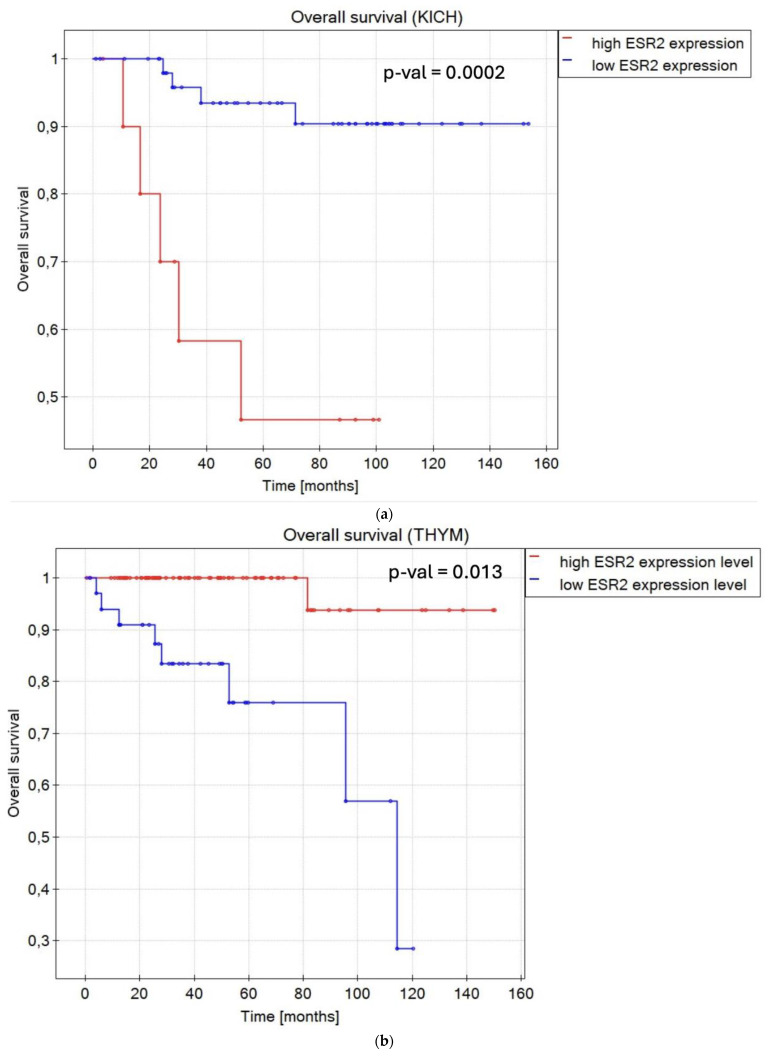
In terms of OS, in renal cancers, KICH (p-value = 0.0002) (Figure 3a), and KIRC (p-value = 0.000001) (Figure S2), a low ESR2 expression level was associated with longer OS compared to a high ESR2 expression level for both sexes. Likewise, low ESR2 expression was favorable in brain lower-grade glioma (LGG) (p-value = 0.000001) (Figure S2). In the case of patients with lymphoid neoplasm diffuse large B-cell lymphoma (DLBC) (p-value = 0.001) (Figure S2) and thymoma (THYM) (p-value = 0.013) (Figure 3b), a high ESR2 expression level was a positive prognostic factor. Notably, sex was determined to be a significant factor with regard to OS in cases of adrenocortical carcinoma (ACC) (p-value = 0.012) (Figure S2), bladder urothelial carcinoma (BLCA) (p-value = 0.01) (Figure S2), BRCA (p-value = 0.007) (Figure 3c), esophageal carcinoma (ESCA) (p-value = 0.029) (Figure S2), HNSC (p-value = 0.005) (Figure S2), kidney renal papillary cell carcinoma (KIRP) (p-value = 0.043) (Figure S2), acute myeloid leukemia (LAML) (p-value = 0.021) (Figure S2), LUAD (p-value = 0.023) (Figure S2), PAAD (p-value = 0.03) (Figure S2), stomach adenocarcinoma (STAD) (p-value = 0.004) (Figure S2), and THCA (p-value = 0.045) (Figure S2). Female patients with high ESR2 expression levels diagnosed with ACC, BLCA, BRCA, and ESCA presented longer OS compared to those with low ESR2 expression levels. When it came to male patients, high ESR2 expression levels in HNSC, LUAD, and PAAD and low ESR2 expression levels in KIRP, LAML, mesothelioma (MESO), STAD, and THCA were attributed to longer OS. Patient groups with only one sex, cervical squamous cell carcinoma and endocervical adenocarcinoma (CESC) (p-value = 0.004) (Figure S2), and ovarian serous cystadenocarcinoma (OV) (p-value = 0.002) (Figure S2) (only female samples) presented high ESR2 expression levels as a favorable factor with regard to OS.
Regarding DFS, ESR2 expression level was considered a prognostic factor in 14 tumor types. In KICH (p-value < 0.000001) (Figure S3), KIRC (p-value = 0.00002) (Figure 3d), and glioblastoma multiforme (GBM) (p-value = 0.003) (Figure S3), a low ESR2 expression level was associated with longer DFS, regardless of sex. Sex was determined as a significant factor in six tumor types considering DFS, not counting datasets with only one sex (TGCT and prostate adenocarcinoma (PRAD)). In ACC (p-value = 0.007) (Figure S3), female patients presented high ESR2 expression levels as a favorable factor in terms of DFS. In male patients with DLBC (p-value = 0.03) (Figure S3), LGG (p-value = 0.005) (Figure 3e), and STAD (p-value = 0.003) (Figure S3), low ESR2 expression was considered a favorable factor in terms of DFS. Similarly, a low ESR2 expression level was considered a favorable prognostic factor in PRAD (only male samples) (p-value = 0.024) (Figure S3), while a high ESR2 expression level was significantly correlated with longer DFS in TGCT (p-value = 0.004) (Figure S3) (only male samples).
However, no significant results were obtained in calculations of remaining TCGA tumor types regarding either overall survival or disease-free survival with ESR2 expression level as a prognostic factor. Therefore, we categorized tumor types based on the prognostic role of ESR2 expression (Figure 3f).
2.4. ESR2 Affects Various Molecular Pathways in Cancer
To better characterize ESR2’s role in various cancer types, we carried out Gene Set Enrichment Analysis (GSEA). We performed an analysis to compare ESR2-associated transcriptomic profiles with pre-defined lists of gene signatures. We selected human molecular signatures from different MSigDB collections, namely, Hallmark Gene Sets Collection and C2 Curated Gene Sets Collection. Across 30 cancer types, significant results (p-value < 0.05 and FDR < 0.05) were obtained for 18 types in the Hallmark collection analysis and 7 cancer types in the C2 collection.
Within Hallmark gene sets, ESR2 influence was most abundant in BLCA, BRCA, LUAD, MESO, PRAD, and TGCT (Figure 4a). The most enriched sets in the Hallmark collection were the G2M checkpoint gene set, epithelial–mesenchymal transition gene set, hypoxia gene set, and TNFα signaling via NFκβ gene set (Figure 4a).
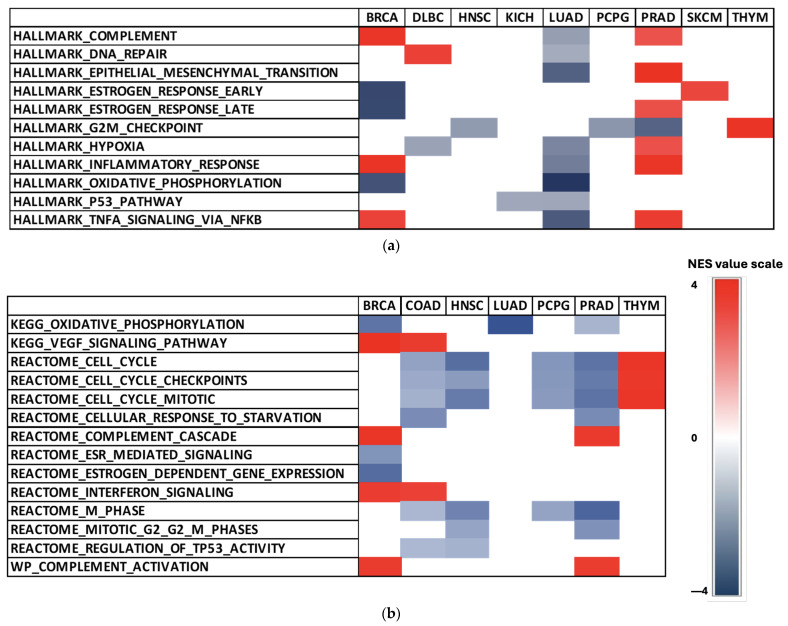
We found significant enrichment in the estrogen response gene set (Hallmark collection) in BRCA (FDR = 0.000), PRAD (FDR = 0.027), and skin cutaneous melanoma (SKCM) (FDR = 0.032) (Figure 4a). However, it is important to note that the estrogen response gene set included both ERα and ERβ. Therefore, those results should be interpreted with caution. Consistently negative NES values in 13 selected Hallmark gene sets were observed in BLCA, LUAD, MESO, and TGCT (Figure 4a). High NES values of Hallmark sets were achieved in PRAD (Figure 4a), with the epithelial–mesenchymal transition gene set demonstrating the highest score (FDR = 0.000).
We also found significant results for the C2 collection, particularly in gene sets associated with cell cycle and TP53 activity (Figure 4b). The estrogen-dependent gene expression and ESR-mediated signaling gene sets from the Reactome sub-collection in C2 yielded significant NES in BRCA (FDR = 0.000; FDR = 0.001) (Figure 4b). Additionally, low NES values were observed in LUAD (Figure 4b), with the oxidative phosphorylation gene set (KEGG sub-collection) reaching the lowest NES of the analysis (FDR = 0.000).
2.5. Various Genes Present Expression Patterns Similar to That of ESR2 in Cancer
As mentioned above, ERβ displays its functions through genomic and non-genomic pathways [ref. 6]. Signaling cascades controlled by ERβ include sets of known cofactors, including Akt, AP1, and SP1 [ref. 6,ref. 7]. However, the entire network of factors cooperating with ERβ is not known. ERβ has been shown to play a significant role in carcinogenesis, glucose homeostasis, and other important pathways [ref. 1]; thus, presenting which genes might be associated with this receptor would be valuable. Hence, we decided to look for genes that showed mRNA expression patterns similar to that of ESR2 in TCGA cancer types.
Employing transcriptomic data from cBioPortal, we established lists of thousands of co-expressed genes for each tumor type. The top co-expressed genes, considering both positive and negative correlations, were selected using Spearman’s coefficient. Subsequently, we conducted a comprehensive search to identify genes that appeared most frequently in these lists, pinpointing potential targets. Our analyses were centered on elucidating the joint impact of ESR2 and the selected co-expressed genes on overall survival (OS) and disease-free survival (DFS). From the initial pool of tens of thousands of genes, we designated 24 genes as the most promising targets, as listed in Table 1.
Table 1: Characteristics of genes of interest.
| Gene | Description | Source |
|---|---|---|
| ACIN1[14q11.2] | Apoptotic chromatin condensation inducer 1 regulates chromatin condensation after activation by caspase-3 during apoptosis. Additionally, it may be involved in mRNA regulation after splicing. | [ref. 33] |
| ARPC2[2q35] | Actin-related protein 2/3 complex subunit 2 mediates actin polymerization in the nucleus and therefore regulates transcription and homolog recombination in response to DNA damage. | [ref. 34,ref. 35] |
| CELF6[15q23] | CUGBP elav-like family member 6 regulates pre-mRNA alternative splicing and may be involved in mRNA editing and translation. | [ref. 36] |
| CFL1[11q13.1] | Cofilin 1 carries out F-actin depolymerization, thus regulating cell morphology through cytoskeletal organization in epithelial cells. | [ref. 37,ref. 38] |
| COL4A3[2q36.3] | The collagen α-3(IV) chain is one of the structural components of glomerular basement membranes. Tumstatin within this domain presents anti-tumor activity. | [ref. 39,ref. 40] |
| FAM30A[14q32.33] | Family with sequence similarity to 30 member A and may be a non-coding RNA. | [ref. 41] |
| FCRL5[1q23.1] | Fc receptor-like 5 is most likely involved in B-cell differentiation and may present an immunoregulatory role in marginal-zone B-cells. | [ref. 42] |
| FNBP4[11p11.2] | Formin-binding protein 4 may be involved in the regulation of cytoskeletal dynamics during cell division and migration. | [ref. 43] |
| LENG8[19q13.42] | Leukocyte receptor cluster member 8 encodes leukocyte-expressed receptors of the immunoglobulin superfamily. | [ref. 44] |
| MDM4[1q32.1] | MDM4 regulator of p53 inhibits p53- and p73-mediated cell cycle arrest and apoptosis and inhibits the degradation of MDM2. | [ref. 45,ref. 46] |
| NDUFB3[2q33.1] | NDAH–ubiquinone oxidoreductase subunit B3 is part of the electron transport chain of mitochondria on the inner membrane of the mitochondrion. | [ref. 47] |
| OCIAD2[4p11] | Ovarian carcinoma immunoreactive antigen domain containing 2 in one of the mitochondrial respiratory chain complex assembly factors. | [ref. 48] |
| PLIN3[19p13.3] | Perilipin 3 is a structural component of lipid droplets required for lipid storage in cells and is involved in mannose 6-phosphate receptor transport. | [ref. 49,ref. 50] |
| POU2AF1[11q23.1] | POU class 2 homeobox associating factor 1 is a transcriptional coactivator associated with POU2F1/OCT1 or POU2F2/OCT2 complexes; likewise, it is essential for B-cells’ ability to respond to antigens. | [ref. 51,ref. 52] |
| PPP1R3E[14q11.2] | Protein phosphatase 1 regulatory subunit 3E is predicted to be involved in the positive regulation of the glycogen biosynthetic process and to be a part of the protein phosphatase type 1 complex. | [ref. 53] |
| RAC1[7p22.1] | Rac family small GTPase 1 is a plasma membrane-associated protein binding to effector proteins involved in secretion, phagocytosis, migration, and differentiation. | [ref. 54,ref. 55,ref. 56] |
| RAP1GDS1[4q23] | Rap1 GTPase-GDP dissociation stimulator 1 is a guanine nucleotide exchange factor in the GDP-GTP dissociation–binding sequence. | [ref. 57,ref. 58] |
| SYNE2[14q23.2] | Spectrin repeat containing nuclear envelope protein 2 (Nesprin-2) is a component of the LInker of Nucleoskeleton and Cytoskeleton (LINC) and regulates the spatial organization of intracellular components. | [ref. 59] |
| TMEM141[9q34.3] | Transmembrane protein 141 is predicted to be an integral component of the membrane. | [ref. 60] |
| TNFRSF13C[22q13.2] | Tumor necrosis factor receptor superfamily member 13C is a B-cell-specific receptor that promotes the survival of mature B-cells and the B-cell response. | [ref. 61,ref. 62] |
| TPTEP2[22q13.1] | TPTE pseudogene 2 is a transmembrane phosphoinositide 3-phosphatase and tensin homolog 2 pseudogene. | [ref. 63] |
| VAMP1[12p13.31] | Vesicle-associated membrane protein 1 is involved in the targeting and fusion of transport vesicles to their target membrane. | [ref. 64,ref. 65] |
| ZBTB25[14q23.3] | Zinc finger- and BTB domain-containing 25 is predicted to be involved in the regulation of transcription by RNA polymerase II. | [ref. 66] |
| ZFYVE26[14q24.1] | Zinc finger FYVE-type-containing 26 encodes protein targeted to membrane lipids through interaction with phospholipids in the membrane. | [ref. 67] |
Notably, our findings highlight seven genes (FAM30A, MDM4, POU2AF1, SYNE2, TNFRSF13C, TPTEP2, VAMP1) presenting positive correlations with ESR2 expression patterns in all tumor types with high ESR2 expression as a positive prognostic factor with regard to OS or DFS (Figure 5a). Conversely, one target gene, RAC1, showed a negative correlation with ESR2 expression patterns in those tumor types.
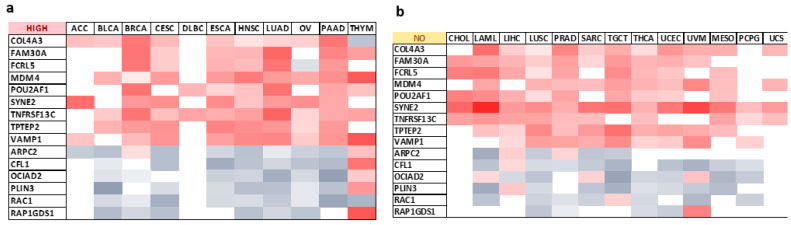
Furthermore, eight target genes (COL4A3, FAM30A, FCRL5, MDM4, SYNE2, TNFRSF13C, TPTEP2, VAMP1) were systematically positively correlated with ESR2 expression patterns in tumor types with low ESR2 expression as a prognostic factor concerning OS or DFS (Figure 5c). Negative correlations across those tumor types were observed in four target genes (ARPC2, CFL1, PLIN3, RAP1GDS1) (Figure 5c). The expression of selected genes in tumors without a significant influence of ESR2 on clinical outcomes is shown in Figure 5b.
To further examine the relationship between target genes and ESR2, we used UCSC Genome Browser. Within our pool of target genes, we looked for predicted transcription factor binding sites for ERβ (ERE canonical binding site 5′-GGTCAnnnTGACC-3′). As in computational modeling approaches, there is no clear difference between functional and non-functional transcription factor binding sites; we searched for EREs that co-occurred with epigenetic marks of active regulatory elements (H3K4me1 and H3K27ac) and promoters (H3K4me3). Furthermore, to strengthen the efficacy of our search, we added a predicted transcription factor binding site for HNF3α (FOXA1) (sequence 5′-[AC]A[AT]T[AG]TT[GT][AG][CT]T[CT]-3′) as a component of our research due to its known cooperation with ERβ in regulating gene activation [ref. 68]. Among the 24 target genes, 12 presented strong evidence of potential ERβ binding sites within their promoter or regulatory regions and were used as target genes in further analysis. Nine target genes (ACIN1 (Figure 5d and Figure S4), FNBP4 (Figure S4), MDM4 (Figure S4), NDUFB3 (Figure S4), OCIAD2 (Figure S4), PLIN3 (Figure S4), POU2AF1 (Figure S4), RAC1 (Figure S4), SYNE2 (Figure S4)) displayed multiple ERβ and HNF3α binding sites within regions with open chromatin epigenetic marks. Three target genes (CFL1 (Figure S4), TMEM141 (Figure S4), TNFRSF13C (Figure 5e)) presented ERβ binding sites without HNF3α binding sites present, though ERβ binding sites co-occurred with H3K4Me1, H3K4Me3, and H3K27Ac marks.
The remaining 12 target genes failed to exhibit substantial evidence to qualify as direct binding targets of ERβ. ARPC2 (Figure S5), COL4A3 (Figure S5), FCRL5 (Figure S5), LENG8 (Figure S5), RAP1GDS1 (Figure S5), VAMP1 (Figure S5), and ZFYVE26 (Figure S5) displayed multiple ERβ and HNF3α binding sites, notwithstanding epigenetic marks of regulatory elements or promoters. Hence, those target genes were not accounted for as possible hits. FAM30A (Figure S5) and PPP1R3E (Figure S5) showed multiple ERβ binding sites without simultaneous open chromatin marks present. Subsequently, they were not considered a significant result. TPTEP2 (Figure S5) and ZBTB25 (Figure S5) showed multiple HNF3α binding sites without ERβ binding sites present; thus, these genes were not recognized as possibly significant results. CELF6 (Figure S5) presented very little evidence of being regulated by ERβ due to scarce ERβ or HNF3α binding sites, all of which were not within open chromatin epigenetic marks; consequently, it was not analyzed further.
2.6. Selected Genes Present a Combined Effect with ESR2 on OS/DFS in Several Cancer Types
As shown in Figure 3f, ESR2 was not considered a sole prognostic factor in 9 cancer types in terms of OS and 17 cancer types in terms of DFS. Hence, we searched for a combined impact of ESR2 and 12 selected target genes on OS or DFS in those tumors. Before further analyzing the combined effects with ESR2, we examined the individual impact of target genes on the OS or DFS. Only genes that did not independently affect OS or DFS were considered (12 target genes: ACIN1, CFL1, FNBP4, MDM4, NDUFB3, OCIAD2, PLIN3, POU2AF1, RAC1, SYNE2, TMEM141, TNFRSF13C). Patients were divided into “high” and “low” expression groups (based simultaneously on ESR2 and target gene expression, with cut-off points calculated separately for each gene), including sex as a factor. Significant correlations (p-value < 0.05) were found for 17 cancer types, as shown in Figure 6 and Supplementary Figures S6 and S7. Lung squamous cell carcinoma (LUSC) and COAD demonstrated the greatest number of correlations, yielding nine and seven significant outcomes between ESR2 and target genes, respectively.
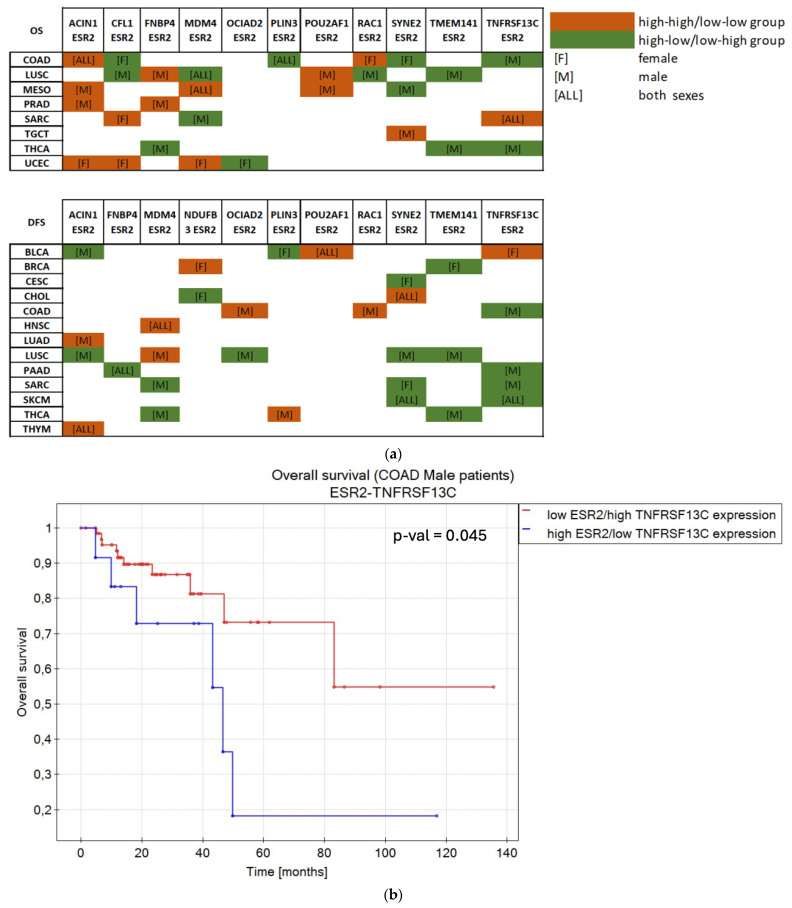
ACIN1 and SYNE2 showed a combined effect with ESR2 on either OS or DFS in 9 out of 17 cancer types, TNFRSF13C showed a combined effect with ESR2 in 8 cancer types, and MDM4 showed a combined effect with ESR2 in 7 cancer types. The remaining eight target genes (CFL1, FNBP4, NDUFB3, OCIAD2, PLIN3, POU2AF1, RAC1, TMEM141) presented a combined influence in five or fewer cancer types (Figure 6a).
BLCA patients of both sexes benefited from simultaneous high expression levels of POU2AF1 and ESR2 in terms of DFS (p-value = 0.028) (Figure 6d). Additionally, female patients with high levels of expression of TNFRSF13C and ESR2 showed longer DFS (p-value = 0.017) (Figure S7). Notably, low levels of expression of PLIN3 in female patients (p-value = 0.045) and high expression of ACIN1 in male patients (p-value = 0.007) correlated with high and low levels of ESR2 expression, respectively, resulting in extended DFS (Figure S7).
Moreover, BRCA female patients presented correlations regarding NDUFB3 (p-value = 0.039) and TMEM14 (p-value = 0.046) (Figure S7). Simultaneous high expression levels of NDUFB3 and ESR2 were associated with longer DFS, while low TMEM141–high ESR2 expression dynamics prolonged DFS in BRCA female patients.
CESC data yielded one significant correlation. Female patients with low SYNE2–high ESR2 expression benefited in terms of DFS (p-value = 0.008) (Figure S7).
Patients of both sexes with CHOL presented longer DFS when SYNE2 and ESR2 were simultaneously highly expressed (p-value = 0.049) (Figure S7). Within female CHOL patients, longer DFS was observed with NDUFB3 low–ESR2 high expression (p-value = 0.047) (Figure S7).
COAD data presented correlations between ESR2 and seven selected genes. Both sexes exhibited longer OS in high–high expression dynamics between ACIN1 and ESR2 (p-value = 0.033) and low PLIN3 and high ESR2 expression (p-value = 0.001) (Figure S6). Female patients with low ESR2–high CFL1 (p-value = 0.017) or SYNE2 (p-value = 0.033) expression or high expression of RAC1 and ESR2 (p-value = 0.02) were characterized by longer OS (Figure S6), while male patients benefited from simultaneous high expression levels of RAC1 and ESR2 in terms of DFS (p-value = 0.007) (Figure S7). High TNFRSF13C and low ESR2 expression positively affected OS (p-value = 0.045) (Figure 6b) and DFS (p-value < 0.000001) (Figure S7) in male patients. Moreover, male patients with COAD benefited from simultaneous low expression levels of OCIAD2 and ESR2 (p-value < 0.000001) (Figure S7).
HNSC data yielded one significant correlation, namely, high MDM4 and high ESR2 expression levels contributed to longer DFS in both female and male patients (p-value = 0.033) (Figure S7).
Male LUAD patients showed longer DFS when ACIN1 and ESR2 presented high expression levels (p-value = 0.013) (Figure S7).
LUSC data yielded the greatest number of correlations between ESR2 and target genes. The OS of patients of both sexes was prolonged with high MDM4 and low ESR2 expression (p-value = 0.036) (Figure S6). Simultaneously, high RAC1 and low ESR2 expression contributed to longer OS in LUSC patients of both sexes (p-value = 0.015) (Figure S6). Male LUSC patients showed longer DFS with high ESR2 and low ACIN1 (p-value = 0.009), OCIAD2 (p-value = 0.022), and SYNE2 (p-value = 0.007) or low ESR2 and high TMEM141 expression (p-value = 0.032) (Figure S7). DFS of male patients was extended with concurrent high expression levels of both MDM4 and ESR2 (p-value = 0.005) (Figure S7), while longer OS was also observed for high expression levels of ESR2 and FNBP4 (p-value = 0.036) or POU2AF1 (p-value = 0.013) (Figure S6). The high–low expression dynamic between ESR2 and CFL1 (p-value = 0.02) or TMEM141 (p-value = 0.046) and the low–high expression dynamic between ESR2 and RAC1 (p-value = 0.015) presented longer OS in LUSC male patients as well (Figure S6).
OS of MESO patients was affected by correlations between ESR2 and ACIN1, MDM4, POU2AF1, or SYNE2 expression levels. Simultaneous high expression levels of ESR2 and MDM4 correlated with longer OS in patients of both sexes (p-value = 0.037), while low levels of ACIN1 (p-value = 0.001) or POU2AF1 (p-value = 0.004) expression combined with high and low levels of ESR2 expression, respectively, correlated with longer OS of male patients (Figure S6). Male MESO patients benefited from longer OS with high SYNE2 and low ESR2 expression levels (p-value = 0.028) (Figure S6).
PAAD data presented significant prognostic outcomes for two target genes, FNBP4 and TNFRSF13C. Low FNBP4 and high ESR2 expression correlated with longer DFS in patients of both sexes (p-value = 0.013) (Figure S7). Concurrently, low TNFRSF13C and high ESR2 correlated with longer DFS in male patients with PAAD (p-value = 0.016) (Figure S7).
Male patients with PRAD showed longer OS when a low level of ESR2 expression was correlated with low expression levels of either ACIN1 (p-value = 0.048) or FNBP4 (p-value = 0.03) (Figure S6).
Sarcoma (SARC) patients with simultaneous low expression levels of TNFRSF13C and ESR2 presented longer OS (p-value = 0.034) (Figure S6). Similarly, female SARC patients with low expression levels of CFL1 and ESR2 showed longer OS (p-value = 0.017) (Figure 6c). TNFRSF13C and ESR2 high–low expression dynamics correlated with longer DFS in male SARC patients (p-value = 0.017) (Figure S7). Sex was also a factor in correlations of ESR2 with MDM4 and SYNE2. Male patients with SARC showed longer DFS (p-value = 0.044) (Figure S7) and OS (p-value = 0.015) (Figure S6) with high MDM4 and low ESR2 expression levels. Further, female patients with SARC with low ESR2 and high SYNE2 expression presented longer DFS (p-value = 0.008) (Figure S7).
In SKCM data, two noteworthy correlations were identified. Patients with low ESR2 and high SYNE2 (p-value = 0.033) or TNFRSF13C (p-value = 0.046) expression exhibited extended DFS, irrespective of sex (Figure S7).
A significant correlation emerged from the TGCT data as well. Simultaneous high expression levels of ESR2 and SYNE2 were associated with extended OS in male patients (p-value = 0.013) (Figure S6).
In male patients with THCA, notable correlations were observed between ESR2 and four target genes. Specifically, FNBP4 demonstrated an impact on both DFS (p-value = 0.028) (Figure S7) and OS (p-value = 0.00007) (Figure S6) when the expression levels of FNBP4 and ESR2 exhibited a high–low dynamic correlation. A similar relationship between high TMEM141 and low ESR2 influenced both DFS (p-value = 0.011) (Figure S7) and OS (p-value = 0.001) (Figure S6) in male patients with THCA. Moreover, male patients with concurrent low expression levels of PLIN3 and ESR2 exhibited extended DFS (p-value = 0.039) (Figure S7). Additionally, high TNFRSF13C and concomitant low ESR2 expression correlated with prolonged OS in male patients (p-value = 0.00005) (Figure S6).
In the THYM data, one significant result emerged, revealing that a simultaneous high expression level between ACIN1 and ESR2 was correlated with extended DFS in both sexes (p-value = 0.032) (Figure S7).
Within the UCEC data, correlations between ESR2 and four target genes were identified. Female UCEC patients who exhibited simultaneous low expression levels of ESR2 and ACIN1 (p-value = 0.018), CFL1 (p-value = 0.046), or MDM4 (p-value = 0.04) demonstrated prolonged OS (Figure S6). Furthermore, female patients with low ESR2 and high OCIAD2 expression also exhibited extended OS (p-value = 0.015) (Figure S6).
Analyses of ACC, DLBC, LAML, pheochromocytoma and paraganglioma (PCPG), uterine carcinosarcoma (UCS), and uveal melanoma (UVM) did not yield significant results.
3. Discussion
This study presents a comprehensive bioinformatics analysis of the potential role of ESR2 across various TCGA tumor types. We used tools and databases containing transcriptomic data to analyze whether ESR2 mRNA expression levels significantly influence overall survival and disease-free survival. Furthermore, we found genes co-expressed with ESR2 and evaluated their combined impact on patient survival.
Our analysis displayed that ESR2 mRNA expression was significantly elevated in normal tissue corresponding to eight tumor types, namely, BRCA, COAD, KICH, KIRC, KIRP, and THCA, aligning with the literature [ref. 69,ref. 70,ref. 71,ref. 72]. Furthermore, ESR2 mRNA expression was significantly higher in tumor tissue in CHOL, ESCA, HNSC, LIHC, and LUSC, which is consistent with prior research findings [ref. 73,ref. 74,ref. 75,ref. 76,ref. 77]. Next, we investigated a correlation between tumor grade or stage and ESR2 mRNA expression using the TISIDB database. Detected in our analysis, a negative correlation between ESR2 expression and grade in UCEC aligns with the literature [ref. 78,ref. 79]. Conversely, a negative correlation in PAAD contradicts previous findings, which notably relied on ERβ staining scores instead of transcriptomic data [ref. 80]. Moreover, ESR2 expression correlated with tumor stage in our study in KIRC, LUAD, TGCT, and THCA. The positive correlation found in KIRC and negative correlation found in LUAD and TGCT are consistent with the majority of previous research. However, some teams reported contradictory results. We attribute these discrepancies to the fact that analyses were conducted using immunohistochemical methods and various antibodies [ref. 81,ref. 82,ref. 83,ref. 84,ref. 85]. For instance, previous research by Dong et al. showed no association of ERβ expression with tumor stage in THCA, although they examined the protein expression of only isoform 2 of ERβ [ref. 86], not ESR2 mRNA levels.
We analyzed whether ESR2 mRNA expression influenced overall survival and disease-free survival across various TCGA tumor types. Our findings highlight high ESR2 expression as a significant positive prognostic factor in nine tumor types: BLCA, BRCA, CESC, ESCA, HNSC, LUAD, OV, PAAD, and THYM. Previous data are inconsistent regarding our analysis. For BLCA, discrepancies noted by Goto et al. underscore variations in immunohistochemical scoring systems and the use of unspecific antibodies [ref. 87]. In BRCA, while previous mRNA-based studies confirm our results [ref. 69,ref. 88], certain protein-based studies suggested that ERβ expression was correlated with poor prognosis [ref. 89,ref. 90]. Similarly, conflicting findings occur in CESC, with some reporting ERβ expression solely in invasive cervical tumors [ref. 91], while others find no ERβ expression in cervical cancer cells [ref. 92]. Despite the lack of a clear link between ERβ and CESC, data indicate its potential role in cervical carcinogenesis. For ESCA, Wang et al. showed that ERβ expression may predict a better outcome for patients [ref. 93], confirming our results. HNSC comprises different types of malignancies affecting many sites, including the oral cavity and larynx [ref. 94]. Studies indicate that ERβ-positive patients with oropharyngeal cancer tend to have longer 5-year survival than ERβ-negative patients [ref. 95]. Still, conflicting reports suggest ERβ’s role in cancer progression through enhanced proliferation and invasion in laryngeal carcinoma [ref. 96]. These inconsistencies may arise from the heterogeneity of HNSC itself and variations in methodologies, such as the use of unspecific antibodies, underscoring the need for further investigation in this area [ref. 22,ref. 23]. While our analysis suggests ERβ as a positive factor in lung cancer, the protein-based meta-analysis of Li et al. showed no significant correlations between receptor expression and clinicopathological features [ref. 97]. Nonetheless, ERβ is the main ER expressed in lung tumor specimens [ref. 82,ref. 98,ref. 99,ref. 100], suggesting its potential significance in estrogenic response in LUAD [ref. 82]. Additionally, the intracellular localization of ERβ emerged as a critical determinant of its activity in OV. Cytoplasmic ERβ was associated with poorer outcomes and chemoresistance in OV, whereas nuclear ERβ did not correlate with either OS or DFS [ref. 101]. However, it is worth noting that the mentioned IHC analysis was performed on a relatively small number of patient samples and required validation on a transcriptomic level [ref. 101]. In terms of PAAD, studies suggest that ERβ might play a significant role in the estrogen-dependent proliferation of pancreatic cancer cells in vitro [ref. 102]. Contrary to our findings, some studies present ERβ as a negative prognostic factor in pancreatic ductal adenocarcinoma (PDAC) [ref. 80]. It is important to note that these findings were based on protein data and conducted with antibodies of uncertain specificity, as highlighted by Andersson et al. [ref. 22] and Nelson et al. [ref. 23]. Moreover, estrogen’s impact on the thymus is well documented, including its role in the inhibition of postnatal thymocyte development [ref. 103,ref. 104]. While many studies have emphasized ERα-dependent estrogen signaling in the thymus [ref. 105,ref. 106,ref. 107], other studies support ERβ overexpression in THYM [ref. 108,ref. 109] and its role as a significant positive factor in terms of OS [ref. 103,ref. 110], which is in line with our analysis.
Moreover, we observed that low ESR2 expression is associated with longer OS in four tumor types: KIRP, LAML, MESO, and THCA. Notably, some studies indicated that ERβ may act as a tumor suppressor in kidney carcinomas, with others correlating higher expression of this receptor with worse survival outcomes [ref. 71,ref. 111,ref. 112]. Our results coincide with the latter. Further, a meta-analysis of gene signatures in acute myeloid leukemia did not establish a direct link between ESR2 expression and patient survival. However, it was noted that high ERβ/ERα ratios were necessary for anti-leukemic effects of ERβ signaling and that ERβ could suppress leukemogenesis [ref. 113]. Here, we present low ESR2 expression as a favorable factor. Therefore further research is required to evaluate whether this receptor affects OS in LAML. Regarding MESO, studies have linked high ERβ expression with better prognosis in OS [ref. 114,ref. 115]. Nevertheless, Pillai et al. [ref. 116] demonstrated that nuclear presence was a favorable factor in terms of OS in MESO, whereas the cytoplasmic fraction of this receptor was associated with poor survival [ref. 116]. In THCA, many studies emphasized the importance of the ERα/ERβ ratio on mRNA and protein levels and its influence on clinical outcomes. However, there is a lack of analyses regarding the specific effect of individual ESR2 expression [ref. 117].
Furthermore, we analyzed ESR2 mRNA expression in correlation with disease-free survival, yielding significant findings across six tumor types. High ESR2 expression emerged as a favorable prognostic indicator for DFS in CHOL, LUSC, and TGCT. To date, ERβ was found to mainly govern estrogen signaling in CHOL [ref. 118], manifesting anti-proliferative actions in extrahepatic CHOL. Other CHOL-related studies focused on the ERα/ERβ ratio’s impact on clinical outcomes [ref. 119,ref. 120]; therefore, the singular influence of ERβ has not been analyzed further so far. Moreover, many studies, summarized in [ref. 121], associated ERβ with a favorable prognosis, consistent with our findings for LUSC. However, few reports indicate ERβ’s involvement in chemoresistance and the promotion of invasion in LUSC [ref. 122]. To the best of our knowledge, the TGCT-related literature does not directly associate ESR2 mRNA expression with survival outcomes.
The remaining three DFS-related results from our analysis indicated a correlation between low ESR2 mRNA expression and extended DFS in GBM, LGG, and PRAD. However, contradictory previous studies have shown that loss of ERβ occurs during glial neoplasm progression [ref. 123,ref. 124], and low ERβ expression has been associated with poorer survival [ref. 125]. With regard to PRAD, Zellweger et al. showed that ERβ expression was associated with poor clinical outcomes in hormone-sensitive prostate cancer [ref. 126]. It is important to note that ERβ has also been presented as a protective factor in prostate cancer progression [ref. 127]. However, some studies show contradictory results [ref. 128], most likely due to the use of different antibodies and receptor isoforms. Lee et al. showed that ERβ’s isoforms also play an essential role in PRAD [ref. 24]: ERβ1 was assessed as an anti-proliferative factor, whereas ERβ2 and ERβ5 were continuously expressed in high-grade prostate tumors [ref. 129].
Moreover, our analyses revealed that both OS and DFS were influenced by ESR2 mRNA expression levels in ACC, DLBC, KICH, KIRC, and STAD. Previous studies have shown that ERβ predominates in adrenal glands, with lower expression in ACC [ref. 130]. In our analysis, we observed that elevated ESR2 mRNA expression in females with ACC was associated with longer OS and DFS, suggesting the tumor-suppressive role of ERβ [ref. 109,ref. 131]. Similarly, we found elevated ESR2 expression as a positive prognostic factor in DLBC, aligning with the existing literature associating ERβ activation with tumor growth inhibition [ref. 132]. In contrast, low ESR2 expression in KICH and KIRC correlated with better survival and prognosis [ref. 133], as in our study. Concerning STAD, low ESR2 expression in male patients was noted as a determinant of better OS and DFS. Xu et al. demonstrated that while ESR2 mRNA levels did not correlate with any clinicopathological parameters, the absence of ERβ at the protein level was associated with poor OS [ref. 134].
As a next step, we conducted GSEA of 30 tumor types to elucidate the role of ESR2 in molecular pathways. We found significant results in several key pathways, including epithelial–mesenchymal transition (EMT), hypoxia response, and cell cycle, among others. In PRAD, our findings align with previous research demonstrating EMT-repressing activity associated with ERβ1 [ref. 135]. Moreover, previous studies suggested that ERβ is implicated in the cellular response to hypoxic conditions, one of the hallmarks of solid tumors [ref. 136,ref. 137,ref. 138,ref. 139]. Our analysis corroborated these findings by revealing a significant enrichment of ESR2 expression in Hallmarks’ “Hypoxia” gene set, suggesting its importance in response to lowered oxygen levels in PRAD. We observed negative NES values in cell cycle-related sets that are potentially dependent on ESR2 expression, such as results for COAD. This aligns with previous research indicating ERβ as a tumor suppressor by arresting the cell cycle and promoting the apoptosis pathway in colorectal cancer [ref. 140].
To further investigate the role of ESR2 in tumors, we searched for co-expressed genes exhibiting expression patterns like ESR2 and possessing estrogen-related regulatory elements. We identified 12 potential genes associated with ESR2 expression in cancer tissue, previously not characterized in terms of ESR2. Subsequently, we assessed the combined influence of those genes and ESR2 on clinical outcomes in patients with TCGA tumor types as they did not present any impact on OS or DFS on their own. While our target genes have been described in the literature, they have primarily been discussed without explicit connections to ESR2 expression. In MESO and UCEC, concurrent low expression of ESR2 and MDM4 showed a combined positive effect on OS. This aligns with the established literature showing that high MDM4 expression tends to correlate with poorer prognosis in many cancer types as it is known to inhibit tumor suppressor activity of p53 in various cancer types [ref. 141,ref. 142,ref. 143]. In LUSC, MDM4 is typically highly expressed [ref. 144], as shown in our analysis. We show that low OCIAD2 and high ESR2 expression correlated with longer DFS in LUSC. OCIAD2 may indirectly exert tumor-promoting activities, and its downregulation led to the loss of mitochondrial structure and an overall decrease in proliferation and invasion in lung cancer [ref. 145,ref. 146]. However, there is a lack of studies showcasing a combined effect with ERβ.
Moreover, in BRCA, the simultaneous high expression of ESR2 and NDUFB3 correlated with longer DFS [ref. 69,ref. 88,ref. 89,ref. 90]. NUDFB3 expression has been shown to elevate mitochondrial ROS production, leading to apoptosis through the JNK signaling pathway and cycle arrest in HCC [ref. 147]. Similarly, in LUSC, it was shown that CFL1 mRNA expression increased with tumor growth, while the protein level of cofilin translated from CFL1 diminished, suggesting a posttranslational regulatory mechanism [ref. 148].
Conversely, in CHOL, we observed that combined high expression of ESR2 and low expression of NDUFB3 was associated with better DFS. In contrast, previous studies suggested that solely elevated ESR2 expression is a positive indicator of clinical outcomes [ref. 118]. The combined impact with NDUFB3 has not been previously reported. Furthermore, our analysis revealed a significant influence on OS in PRAD of the low-level co-expression of ESR2 and ACIN1. Notably, ACIN1 expression has been linked to bicalutamide resistance, a first-generation drug used in androgen deprivation therapy for prostate cancer [ref. 149]. In previous studies, elevated levels of CFL1 were correlated with an increased risk of lymph node metastasis and a deeper rate of local invasion in colon cancer [ref. 150]. Contrarily, we show that low ESR2 and high CFL1 expression were associated with better OS and DFS in COAD and improved OS in LUSC. Regarding DFS, we observed that high ESR2 and low FNBP4 expression correlated with positive outcomes in PAAD. While ERβ has been suggested as an adverse prognostic factor in PAAD [ref. 80], the role of FNBP4 remains relatively unexplored. To date, FNBP4 expression has been correlated with poor OS in hepatocellular carcinoma [ref. 151]. Moreover, ERβ’s exact role in LUSC and PRAD has not been unequivocally determined [ref. 80,ref. 126,ref. 127,ref. 128,ref. 152]. However, FNBP4 possesses in its promoter region the canonical sequence of ERE, which may suggest its direct regulation by ERβ. Our study also demonstrated another correlation: in UCEC, low ESR2 and high OCIAD2 expression were associated with longer OS. Since OCIAD2 expression was associated with adverse clinical outcomes in previous studies [ref. 153], our results linking it to ESR2 require further analysis to unravel potential mechanisms of interaction. Studies have shown that the downregulation of SYNE2 inhibits endothelial cell migration and may play a role in angiogenesis [ref. 154], while other reports indicate that SYNE2 depletion is significantly correlated with increased muscle cell proliferation [ref. 155]. Interestingly, in CHOL and TGCT, simultaneous high expression levels of ESR2 and SYNE2 improved patients’ clinical outcomes. At the same time, high ESR2 and low SYNE2 expression were significantly associated with longer DFS in CESC and LUSC, while an inverse relationship between those factors was noted in COAD, MESO, SARC, and SKCM. Therefore, the ESR2 regulation of SYNE2 expression could be tumor-dependent and requires further functional studies.
While previous studies have shown that ACIN1 may activate proapoptotic signaling pathways in colorectal cancer, we hypothesize that it may work synergistically together with ESR2, a known tumor suppressor in CRC [ref. 156,ref. 157]. Additionally, MDM4 and ESR2 have been reported to be upregulated in HNSC [ref. 75,ref. 158]. The precise role of ERβ in this context remains ambiguous, warranting a cautious interpretation of our results. In COAD, we observed a simultaneous low expression of ESR2 and OCIAD2 as a favorable factor in DFS. ERβ, a known tumor suppressor in COAD [ref. 140], may regulate OCIAD2 expression, potentially leading to mitochondria-related apoptosis [ref. 145]. Another identified connection relates to BLCA patients exhibiting simultaneous high ESR2 and low PLIN3 expression, which presented better clinical outcomes. Previous studies linked high PLIN3 expression with shorter survival time in other tumor types, such as lung adenocarcinoma [ref. 159]. Furthermore, POU2AF1 expression used to be considered lymphocyte-restricted. However, some studies showcase the expression of POU2AF1 in normal human airway epithelium and lung adenocarcinoma tissue [ref. 160,ref. 161]. In LUSC and BLCA, we presented that concomitant high expressions of ESR2 and POU2AF1 were associated with extended DFS. On the other hand, simultaneous low expression of ESR2 and POU2AF1 was associated with prolonged OS in MESO.
Furthermore, another ESR2-correlated gene was TMEM141, which encodes a transmembrane protein, likely involved in protein binding [ref. 162,ref. 163] and neural development [ref. 164]. However, data regarding TMEM141’s role in cancer are scarce. Additionally, in our analysis, the co-expression of TNFRSF13C and ESR2 was associated with OS or DFS in BLCA, COAD, PAAD, SARC, and SKCM. We hypothesize a regulatory link between those two factors, with a possible influence on tumor-infiltrating B cells, as TNFRSF13C has been demonstrated to prevent apoptosis by inhibiting Bim proteins and enhancing mitochondrial activity, thereby prolonging B-cells’ cellular life span [ref. 165,ref. 166,ref. 167,ref. 168]. The last interesting identified co-expressed gene was RAC1.
While previous studies have linked elevated RAC1 protein levels with high metastasis of lung tumor cells [ref. 169], we observed that low ESR2 and high RAC1 expression were associated with longer OS in LUSC. Conversely, in COAD, we demonstrated that the simultaneous low expression of ESR2 and RAC1 correlates with longer OS and DFS. Previous studies linked dysregulated RAC1 expression with tumor initiation, progression, and metastasis in cases of gastric, testicular, and breast cancers [ref. 170], establishing our results in COAD in line with the literature.
In summary, we comprehensively analyzed ESR2 and selected co-expressed gene expression in TCGA tumor types. Despite the depth of the analysis, a notable limitation of our study was its reliance solely on transcriptomic data. However, the lack of this element was related to inconsistencies with regard to ERβ’s role in carcinogenesis in various tumor types, along with the method of detection of the protein. We intended to compare transcriptomic and proteomic data; therefore, we checked the Human Protein Atlas database. Notably, HPA offers two types of antibodies for Erβ: HPA068406 (Atlas Antibodies, Sigma-Aldrich, Milwaukee, WI, USA) and CAB079300 (R&D Systems). HPA068406 is a polyclonal rabbit antibody validated in the HPA database through immunocytochemistry, Western blot, and protein array, while CAB079300 is a monoclonal mouse antibody validated in HPA by immunohistochemical staining. Moreover, according to HPA, CAB079300 has shown inconsistencies, particularly in Western blot analysis, contradictory to Andersson et al. [ref. 22] and Nelson et al. [ref. 23]. Furthermore, HPA antibody HPA068406 fails to detect ERβ in healthy breast tissue samples, contradicting several studies [ref. 69,ref. 171]. Given the disparity between transcriptomic data from TIMER2.0 and immunostaining data from HPA, we could not perform a comparative analysis using protein data; hence, we focused on TCGA datasets. Additionally, posttranscriptional or posttranslational modifications that alter mRNA and protein levels of ESR2/ERβ could play a significant role. Therefore, the abundance of ESR2 mRNA might not be reflected in protein presence or activity. For this reason, we think a proteomic analysis is the next step to understanding ERβ’s activities and responsibilities in cancer, all while taking into consideration ERβ’s isoforms and the specificity of antibodies used in research, likewise mentioned before. Still, this study constitutes a compact and comprehensive analysis of ESR2 mRNA expression levels across diverse tumor types and an exploration of its potential functional implications.
4. Materials and Methods
4.1. Transcriptomic and Proteomic Data
All transcriptomic and proteomic data are available online, and the access is neither restricted nor requires patients’ consent. Transcriptomic data were downloaded from cBioPortal (https://www.cbioportal.org/, accessed on 1 February 2024) [ref. 172,ref. 173,ref. 174]. We used the Firehose Legacy datasets (previously known as Provisional datasets) divided by tumor type. The RNA sequencing-based mRNA expression data (RNASeq V2) were normalized using RSEM, resulting in mRNA expression z-scores, as described in [ref. 175]. Specific names of datasets and the numbers of patients in each dataset are available in Supplementary Table S1. Patients without full clinical data used in this study were excluded. Additional transcriptomic data were obtained from the Human Protein Atlas [ref. 176,ref. 177] (https://www.proteinatlas.org/ accessed on 1 February 2024) and TIMER2.0 [ref. 178,ref. 179,ref. 180] (http://timer.comp-genomics.org accessed on 1 February 2024).
Proteomic data were downloaded from the Human Protein Atlas (HPA), a database allowing for a genome-wide analysis of human proteins. Data were obtained through a phrase search (“ESR2”) and with the use of downloadable data from the “Data” tab, which was searched for genes of interest.
4.2. Clinical Data
All clinical data were downloaded from cBioPortal [ref. 172,ref. 173,ref. 174]. We used the OncoPrint tab to access clinical data, selecting characteristics from “Tracks” options, including overall survival status, overall survival time, disease-free survival status, disease-free survival time, and sex. Combined transcriptomic data and clinical features were used to plot Kapplan–Meier curves for overall survival and disease-free survival. Additional clinical data were acquired from TISIDB [ref. 181] (http://cis.hku.hk/TISIDB/ accessed on 1 February 2024).
4.3. Databases and Bioinformatic Tools
4.3.1. cBioPortal
We used the “Co-expression” tab in cBioPortal [ref. 172,ref. 173,ref. 174] to identify similarly co-expressed genes with regard to ESR2. Spearman’s correlations (rho) were calculated and results with a p-value < 0.05 and FDR < 0.05 were considered significant. Gene lists were downloaded and searched for the highest correlation coefficient. Target genes were selected based on the highest repeatability and the highest rho scores.
4.3.2. TIMER2.0
TIMER2.0 [ref. 178,ref. 179,ref. 180] was used to access transcriptomic data regarding tumors and corresponding normal tissue samples. Through the search bar (phrase “ESR2”) and “Cancer Exploration” features, we were able to generate plots presenting differential gene expression between tumor and normal tissue samples (“Gene_DE” tab). To validate correlations calculated based on cBioPortal data, we used the “Gene_Corr” tab to generate Spearman’s correlations between ESR2 and selected genes. Results with a p-value < 0.05 and FDR < 0.05 were considered significant.
4.3.3. Human Protein Atlas
The Human Protein Atlas (HPA) [ref. 176,ref. 177] database was searched with the phrase “ESR2” to acquire proteomic and transcriptomic data across tissue types. Protein and RNA expressions in tissue profiles were analyzed using the “Tissue” feature. The “Pathology” feature was used to search cancer-related data in HPA. Data from the “Downloadable data” tab were acquired and analyzed with regard to ESR2 and target genes.
Overviews of protein expression patterns in tissue samples in the HPA database; thus, the staining scores in immunohistochemistry assays mentioned in this manuscript, were provided by HPA. The guidelines for the classification of immunohistochemical results are described in the “Assays & Annotation” section of the HPA database (https://www.proteinatlas.org/about/assays+annotation#ihk accessed on 1 February 2024), including staining intensity (negative, weak, moderate, strong), fraction of stained cells (<25%, 25–75%, <75%), and subcellular localization (nucleus, cytoplasm, membrane). All the annotations in the HPA database are provided by a specialist, with verification by a second specialist.
4.3.4. TISIDB
In the TISIDB [ref. 181] database, we researched associations between ESR2 expression and clinical features across TCGA tumor types. Data were accessed through the search bar (phrase “ESR2”) and choosing appropriate tabs, including “Clinical”. We used TISIDB tools to generate survival curves and calculate Spearman’s correlations between ESR2 expression and stage or grade. Results with a p-value < 0.05 and FDR < 0.05 were considered significant.
The TISIDB database integrates multiple heterogeneous data types pertaining to tumor and immune system interactions. Genomics, transcriptomics, and clinical data of 30 non-hematologic cancer types were collected from TCGA to present associations between gene expression and clinical features. All classifications were likewise based on TCGA nomenclature, including the stage and grade classifications used in this study.
4.3.5. GSEA
We established Differentially Expressed Gene (DEG) lists based on cBioPortal [ref. 172,ref. 173,ref. 174] transcriptomic data. Cut-off points for p-value and FDR were both <0.05. The created DEG lists were used to run a preranked Gene Set Enrichment Analysis. We used the Gene Set Enrichment Analysis (GSEA) [ref. 182,ref. 183,ref. 184] tool to present whether predefined sets of genes showed statistically significant, concordant differences within provided samples. The predefined gene sets were provided by the Molecular Signatures Database (https://www.gsea-msigdb.org/gsea/index.jsp accessed on 1 February 2024). DEG lists were uploaded to the GSEA tool (GSEA software version 4.3.3.) and analyzed through GSEAPreRankedPage. The analysis was computed with default settings: probe sets were created with HUGO gene symbols, so the “No_collapse” option was chosen; the permutation number was set to 1000 and the permutation type “gene-sets” was selected. We used FDR < 0.05 for significant results due to the gene set permutation type of analysis and for gene set size correction.
4.3.6. UCSC Genome Browser
We used the University of Santa Cruz Genome Browser [ref. 185] (https://genome.ucsc.edu/index.html accessed on 1 February 2024) to visualize genomic data (based on GRCh38/hg38 [December 2013] human assembly). We searched for transcription sites for estrogen receptor β (ERβ, ESR2 gene) and hepatocyte nuclear factor 3-α (HNF3α, FOXA1 gene) in target genes through JASPAR CORE 2022 with default settings regarding score (minimum score = 400) (JASPAR CORE 2022 was the update available at the time of conducting this analysis). Moreover, we searched for co-occurring open chromatin marks near transcription sites for ERβ and HNF3α, namely, epigenetic marks of regulatory elements (H3K4Me1 and H3K27Ac) and promoters (H3K4Me3). We used ENCODE Regulation Layered H3K4Me1, Layered H3K4Me3, and Layered H3K27Ac tracks with default settings as they showed histone marks across the genome based on ChIP-seq data from seven selected cell lines (GM12878, H1-hESC, HSMM, HUVEC, K562, NHEK, NHLF). Each regulatory factor available in ENCODE Regulation tracks was assayed separately; consequently, complete data were attainable for a limited number of cell lines. Target genes with multiple ERβ and HNF3α binding sites co-occurring with open chromatin epigenetic marks were selected as possibly substantial hits for further analysis.
4.4. Statistical Analysis
Statical analyses we carried out using PQStat v.1.8.4 software (https://pqstat.pl/ accessed on 5 May 2024). Descriptive statistics of each dataset are demonstrated in Supplementary Table S1. The normality of the data was calculated with the Shapiro–Wilk test (α < 0.05). As all datasets proved to be not normally distributed, the correlations between two variables were calculated with two-tailed Spearman’s rank correlation coefficient (rho) (α < 0.05). In GSEA, statistical calculations of NES values were obtained within GSEA analysis software version 4.3.3. Overall survival (OS) and disease-free survival (DFS) analyses were estimated using the Mantel–Cox test (log-rank test) (α < 0.05). Patients were divided into “high” and “low” groups based on expression levels of ESR2 or target genes. Cut-off points for classification into “high” or “low” expression groups were based on the Evaluate Cutpoints R application algorithm [ref. 32], with the “survival” R package (RStudio software version 2024.01.0) as a selected method for stratification of the patient into two groups. Kapplan–Meier plots were graphed with PQStat v.1.8.4 software. In all statistical analyses, results with a p-value < 0.05 and FDR < 0.05 were considered significant.
5. Conclusions
- ESR2 mRNA expression differs between cancerous and normal tissue in various TCGA tumor types, including BRCA, COAD, KICH, CHOL, HNSC, and LUSC;
- ESR2 expression impacts patient survival in several TCGA tumor types, including BLCA, HNSC, THYM, KIRP, and THCA;
- GSEA analysis reveals ESR2 enrichment in gene sets related to epithelial–mesenchymal transition, hypoxia response, and cell cycle in cancers like PRAD and COAD;
- Twelve genes (ACIN1, CFL1, FNBP4, MDM4, NDUFB3, OCIAD2, PLIN3, POU2AF1, RAC1, SYNE2, TMEM141, TNFRSF13C) were identified as co-expressed with ESR2 and showing a combined effect with the receptor on patient survival in selected tumors, including BLCA, MESO, BRCA, COAD, and SKCM.
References
- M. Jia, K. Dahlman-Wright, J.Å. Gustafsson. Estrogen receptor alpha and beta in health and disease. Best Pract. Res. Clin. Endocrinol. Metab., 2015. [DOI | PubMed]
- P. Chen, B. Li, L. Ou-Yang. Role of estrogen receptors in health and disease. Front. Endocrinol., 2022. [DOI | PubMed]
- A. Božović, V. Mandušić, L. Todorović, M. Krajnović. Estrogen Receptor Beta: The Promising Biomarker and Potential Target in Metastases. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- S. Nilsson, S. Mäkelä, E. Treuter, M. Tujague, J. Thomsen, G. Andersson, E. Enmark, K. Pettersson, M. Warner, J.A. Gustafsson. Mechanisms of Estrogen Action. Physiol. Rev., 2001. [DOI | PubMed]
- R. Kumar, M.N. Zakharov, S.H. Khan, R. Miki, H. Jang, G. Toraldo, R. Singh, S. Bhasin, R. Jasuja. The dynamic structure of the estrogen receptor. J. Amino Acids, 2011. [DOI | PubMed]
- N.M. Hwang, L.P. Stabile. Estrogen Receptor ß in Cancer: To ß(e) or not to ß(e)?. Endocrinology, 2021. [DOI | PubMed]
- C.-Y. Lin, V.B. Vega, J.S. Thomsen, T. Zhang, S.L. Kong, M. Xie, K.P. Chiu, L. Lipovich, D.H. Barnett, F. Stossi. Whole-genome cartography of estrogen receptor α binding sites. PLoS Genet., 2007. [DOI | PubMed]
- N. Heldring, A. Pike, S. Andersson, J. Matthews, G. Cheng, J. Hartman, M. Tujague, A. Ström, E. Treuter, M. Warner. Estrogen receptors: How do they signal and what are their targets. Physiol. Rev., 2007. [DOI | PubMed]
- M. Warner, B. Huang, J.-A. Gustafsson. Estrogen Receptor β as a Pharmaceutical Target. Trends Pharmacol. Sci., 2017. [DOI | PubMed]
- R. Mal, A. Magner, J. David, J. Datta, M. Vallabhaneni, M. Kassem, J. Manouchehri, N. Willingham, D. Stover, J. Vandeusen. Estrogen Receptor Beta (ERβ): A Ligand Activated Tumor Suppressor. Front. Oncol., 2020. [DOI | PubMed]
- A.W. Nelson, W.D. Tilley, D.E. Neal, J.S. Carroll. Estrogen receptor beta in prostate cancer: Friend or foe?. Endocr.-Relat. Cancer, 2014. [DOI | PubMed]
- B. Huang, Y. Omoto, H. Iwase, H. Yamashita, T. Toyama, R.C. Coombes, A. Filipovic, M. Warner, J. Gustafsson. Differential expression of estrogen receptor α, β1, and β2 in lobular and ductal breast cancer. Proc. Natl. Acad. Sci. USA, 2014. [DOI | PubMed]
- O. Treeck, I. Juhasz-Boess, C. Lattrich, F. Horn, R. Goerse, O. Ortmann. Effects of exon-deleted estrogen receptor β transcript variants on growth, apoptosis and gene expression of human breast cancer cell lines. Breast Cancer Res. Treat., 2007. [DOI | PubMed]
- C. Zhao, E.W.-F. Lam, A. Sunters, E. Enmark, M.T. De Bella, R.C. Coombes, J. Gustafsson, K. Dahlman-Wright. Expression of estrogen receptor β isoforms in normal breast epithelial cells and breast cancer: Regulation by methylation. Oncogene, 2003. [DOI | PubMed]
- G.C. Markey, R. Cullen, P. Diggin, A.D.K. Hill, E.W. Mc Dermott, N.J. O’Higgins, M.J. Duffy. Estrogen receptor-β mRNA is associated with adverse outcome in patients with breast cancer. Tumor Biol., 2009. [DOI | PubMed]
- Increased Expression of Estrogen Receptor beta mRNA in Tamoxifen-Resistant Breast Cancer Patients
- V. Giroux, G. Bernatchez, J.C. Carrier. Chemopreventive effect of ERβ-Selective agonist on intestinal tumorigenesis in ApcMin/+ mice. Mol. Carcinog., 2010. [DOI | PubMed]
- V. Giroux, F. Lemay, G. Bernatchez, Y. Robitaille, J.C. Carrier. Estrogen receptor β deficiency enhances small intestinal tumorigenesis in ApcMin/+ mice. Int. J. Cancer, 2008. [DOI | PubMed]
- M. Yang, J. Wang, L. Wang, C. Shen, B. Su, M. Qi, J. Hu, W. Gao, W. Tan, B. Han. Estrogen induces androgen-repressed SOX4 expression to promote progression of prostate cancer cells. Prostate, 2015. [DOI | PubMed]
- T. Fixemer, K. Remberger, H. Bonkhoff. Differential expression of the estrogen receptor beta (ERβ) in human prostate tissue, premalignant changes, and in primary, metastatic, and recurrent prostatic adenocarcinoma. Prostate, 2002. [DOI]
- G.G. Kuiper, E. Enmark, M. Pelto-Huikko, S. Nilsson, J.A. Gustafsson. Cloning of a novel receptor expressed in rat prostate and ovary. Proc. Natl. Acad. Sci. USA, 1996. [DOI | PubMed]
- S. Andersson, M. Sundberg, N. Pristovsek, A. Ibrahim, P. Jonsson, B. Katona, C.-M. Clausson, A. Zieba, M. Ramström, O. Söderberg. Insufficient antibody validation challenges oestrogen receptor beta research. Nat. Commun., 2017. [DOI | PubMed]
- A.W. Nelson, A.J. Groen, J.L. Miller, A.Y. Warren, K.A. Holmes, G.A. Tarulli, W.D. Tilley, B.S. Katzenellenbogen, J.R. Hawse, V.J. Gnanapragasam. Comprehensive assessment of estrogen receptor beta antibodies in cancer cell line models and tissue reveals critical limitations in reagent specificity. Mol. Cell. Endocrinol., 2017. [DOI | PubMed]
- M.-T. Lee, B. Ouyang, S.-M. Ho, Y.-K. Leung. Differential expression of estrogen receptor beta isoforms in prostate cancer through interplay between transcriptional and translational regulation. Mol. Cell. Endocrinol., 2013. [DOI | PubMed]
- Y.-K. Leung, P. Mak, S. Hassan, S.-M. Ho. Estrogen receptor (ER)-β isoforms: A key to understanding ER-β signaling. Proc. Natl. Acad. Sci. USA, 2006. [DOI | PubMed]
- P. de Cremoux, D. Rosenberg, J. Goussard, C. Bremont-Weil, F. Tissier, C. Tran-Perennou, L. Groussin, X. Bertagna, J. Bertherat, M.-L. Raffin-Sanson. Expression of progesterone and estradiol receptors in normal adrenal cortex, adrenocortical tumors, and primary pigmented nodular adrenocortical disease. Endocr.-Relat. Cancer, 2008. [DOI | PubMed]
- N. Sato, K. Ise, S. Hata, S. Yamashita, A. Ito, H. Sasano, Y. Nakamura. Clinicopathological Significance of Estrogen Receptor β and Estrogen Synthesizing/Metabolizing Enzymes in Urothelial Carcinoma of Urinary Bladder. Pathol. Oncol. Res., 2021. [DOI | PubMed]
- D. Huang, M. Berglund, A. Damdimopoulos, P. Antonson, C. Lindskog, G. Enblad, R.-M. Amini, S. Okret. Sex- and Female Age-Dependent Differences in Gene Expression in Diffuse Large B-Cell Lymphoma—Possible Estrogen Effects. Cancers, 2023. [DOI | PubMed]
- Z.G. Gul, C.W. Liaw, R. Mehrazin. Gender Differences in Incidence, Diagnosis, Treatments, and Outcomes in Clinically Localized Bladder and Renal Cancer. Urology, 2020. [DOI | PubMed]
- Y. Wang, C. Wu, J. Zhou, H. Fang, J. Wang. Overexpression of estrogen receptor β inhibits cellular functions of human hepatic stellate cells and promotes the anti-fibrosis effect of calycosin via inhibiting STAT3 phosphorylation. BMC Pharmacol. Toxicol., 2022. [DOI | PubMed]
- M. Kim, S.-J. Kim, S.Y. Ha, Z. Xu, Y. Han, H.-G. Jee, S.W. Cho, Y.J. Park, K.E. Lee. BRAFV600E Mutation Enhances Estrogen-Induced Metastatic Potential of Thyroid Cancer by Regulating the Expression of Estrogen Receptors. Endocrinol. Metab., 2022. [DOI]
- M. Ogłuszka, M. Orzechowska, D. Jędroszka, P. Witas, A.K. Bednarek. Evaluate Cutpoints: Adaptable continuous data distribution system for determining survival in Kaplan-Meier estimator. Comput. Methods Programs Biomed., 2019. [DOI | PubMed]
- S. Sahara, M. Aoto, Y. Eguchi, N. Imamoto, Y. Yoneda, Y. Tsujimoto. Acinus is a caspase-3-activated protein required for apoptotic chromatin condensation. Nature, 1999. [DOI | PubMed]
- M.D. Welch, A.H. DePace, S. Verma, A. Iwamatsu, T.J. Mitchison. The human Arp2/3 complex is composed of evolutionarily conserved subunits and is localized to cellular regions of dynamic actin filament assembly. J. Cell Biol., 1997. [DOI | PubMed]
- B.R. Schrank, T. Aparicio, Y. Li, W. Chang, B.T. Chait, G.G. Gundersen, M.E. Gottesman, J. Gautier. Nuclear ARP2/3 drives DNA break clustering for homology-directed repair. Nature, 2018. [DOI | PubMed]
- P.J. Good, Q. Chen, S.J. Warner, D.C. Herring. A Family of human RNA-binding proteins related to the drosophila bruno translational regulator. J. Biol. Chem., 2000. [DOI | PubMed]
- S.W. Bai, M.T. Herrera-Abreu, J.L. Rohn, V. Racine, V. Tajadura, N. Suryavanshi, S. Bechtel, S. Wiemann, B. Baum, A.J. Ridley. Identification and characterization of a set of conserved and new regulators of cytoskeletal organization, cell morphology and migration. BMC Biol., 2011. [DOI | PubMed]
- Chfr Regulates a Mitotic Stress Pathway through Its RING-Finger Domain with Ubiquitin Ligase Activity
- Coexpression of the α 3(IV) and α 4(IV) Collagen Chains in Human Tissues
- Y. Maeshima, P.C. Colorado, A. Torre, K.A. Holthaus, J.A. Grunkemeyer, M.B. Ericksen, H. Hopfer, Y. Xiao, I.E. Stillman, R. Kalluri. Distinct antitumor properties of a type IV collagen domain derived from basement membrane. J. Biol. Chem., 2000. [DOI | PubMed]
- Y. Yang, Y. Zhao, N. Hu, J. Zhao, Y. Bai. lncRNA KIAA0125 functions as a tumor suppressor modulating growth and metastasis of colorectal cancer via Wnt/β-catenin pathway. Cell Biol. Int., 2019. [DOI | PubMed]
- Y. Nakayama, S.M. Weissman, A.L. Bothwell. BXMAS1 identifies a cluster of homologous genes differentially expressed in B cells. Biochem. Biophys. Res. Commun., 2001. [DOI | PubMed]
- T. Nagase, K.-I. Ishikawa, M. Suyama, R. Kikuno, M. Hirosawa, N. Miyajima, A. Tanaka, H. Kotani, N. Nomura, O. Ohara. Prediction of the coding sequences of unidentified human genes. XIII. The complete sequences of 100 new cDNA clones from brain which code for large proteins in vitro. DNA Res., 1999. [DOI | PubMed]
- H. Wende, A. Volz, A. Ziegler. Extensive gene duplications and a large inversion characterize the human leukocyte receptor cluster. Immunogenetics, 2000. [DOI | PubMed]
- E. Toufektchan, V. Lejour, R. Durand, N. Giri, I. Draskovic, B. Bardot, P. Laplante, S. Jaber, B.P. Alter, J.-A. Londono-Vallejo. Germline mutation of MDM4, a major p53 regulator, in a familial syndrome of defective telomere maintenance. Sci. Adv., 2020. [DOI | PubMed]
- Y. Jin, M.-S. Dai, S.Z. Lu, Y. Xu, Z. Luo, Y. Zhao, H. Lu. 14-3-3γ binds to MDMX that is phosphorylated by UV-activated Chk1, resulting in p53 activation. EMBO J., 2006. [DOI | PubMed]
- L. Jensen, M. Nielsen, J. Justesen, L. Hansen. Assignment1 of human NADH dehydrogenase (ubiquinone) 1 beta subcomplex 3 (NDUFB3) and of its four pseudogenes to human chromosomes 2q31.3, 1p13.3→p13.1, 9q32→q34.1, 14q22.3→q23.1 and 14q32.2 by radiation hybrid mapping. Cytogenet. Genome Res., 2001. [DOI | PubMed]
- K.J. Chojnacka, P. Elancheliyan, B.H.M. Mussulini, K. Mohanraj, S. Callegari, A. Gosk, T. Banach, T. Góral, K. Szczepanowska, P. Rehling. Ovarian carcinoma immunoreactive antigen–like protein 2 (OCIAD2) is a novel complex III–specific assembly factor in mitochondria. Mol. Biol. Cell, 2022. [DOI | PubMed]
- E. Díaz, S.R. Pfeffer. TIP47: A cargo selection device for mannose 6-phosphate receptor trafficking. Cell, 1998. [DOI | PubMed]
- R. Liu, J.-H. Lee, J. Li, R. Yu, L. Tan, Y. Xia, Y. Zheng, X.-L. Bian, P.L. Lorenzi, Q. Chen. Choline kinase alpha 2 acts as a protein kinase to promote lipolysis of lipid droplets. Mol. Cell, 2021. [DOI | PubMed]
- M. Strubin, J.W. Newell, P. Matthias. OBF-1, a novel B cell-specific coactivator that stimulates immunoglobulin promoter activity through association with octamer-binding proteins. Cell, 1995. [DOI | PubMed]
- Y. Luo, R.G. Roeder. Cloning, functional characterization, and mechanism of action of the B-cell-specific transcriptional coactivator OCA-B. Mol. Cell. Biol., 1995. [DOI | PubMed]
- S. Munro, H. Ceulemans, M. Bollen, J. Diplexcito, P.T. Cohen. A novel glycogen-targeting subunit of protein phosphatase 1 that is regulated by insulin and shows differential tissue distribution in humans and rodents. FEBS J., 2005. [DOI | PubMed]
- A.J. Ridley, H.F. Paterson, C.L. Johnston, D. Diekmann, A. Hall. The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell, 1992. [DOI | PubMed]
- M.R. Reijnders, N.M. Ansor, M. Kousi, W.W. Yue, P.L. Tan, K. Clarkson, J. Clayton-Smith, K. Corning, J.R. Jones, W.W. Lam. RAC1 Missense Mutations in Developmental Disorders with Diverse Phenotypes. Am. J. Hum. Genet., 2017. [DOI | PubMed]
- J. Zhao, R.K. Mialki, J. Wei, T.A. Coon, C. Zou, B.B. Chen, R.K. Mallampalli, Y. Zhao. SCF E3 ligase F-box protein complex SCFFBXL19regulates cell migration by mediating Rac1 ubiquitination and degradation. FASEB J., 2013. [DOI | PubMed]
- H. Shimizu, S. Toma-Fukai, S. Saijo, N. Shimizu, K. Kontani, T. Katada, T. Shimizu. Structure-based analysis of the guanine nucleotide exchange factor SmgGDS reveals armadillo-repeat motifs and key regions for activity and GTPase binding. J. Biol. Chem., 2017. [DOI | PubMed]
- H.G. Vikis, S. Stewart, K.-L. Guan. SmgGDS displays differential binding and exchange activity towards different Ras isoforms. Oncogene, 2002. [DOI | PubMed]
- S.S. Wallis, L.N. Ventimiglia, E. Otigbah, E. Infante, M.A. Cuesta-Geijo, G.R. Kidiyoor, M.A. Carbajal, R.A. Fleck, M. Foiani, S. Garcia-Manyes. The ESCRT machinery counteracts Nesprin-2G-mediated mechanical forces during nuclear envelope repair. Dev. Cell, 2021. [DOI | PubMed]
- S.J. Humphray, K. Oliver, A.R. Hunt, R.W. Plumb, J.E. Loveland, K.L. Howe, T.D. Andrews, S. Searle, S.E. Hunt, C.E. Scott. DNA sequence and analysis of human chromosome. Nature, 2004. [DOI | PubMed]
- N. Kayagaki, M. Yan, D. Seshasayee, H. Wang, W. Lee, D.M. French, I.S. Grewal, A.G. Cochran, N.C. Gordon, J. Yin. BAFF/BLyS receptor 3 binds the B cell survival factor BAFF ligand through a discrete surface loop and promotes processing of NF-κB. Immunity, 2002. [DOI | PubMed]
- M. Yan, J.R. Brady, B. Chan, W.P. Lee, B. Hsu, S. Harless, M. Cancro, I.S. Grewal, V.M. Dixit. Identification of a novel receptor for B lymphocyte stimulator that is mutated in a mouse strain with severe B cell deficiency. Curr. Biol., 2001. [DOI | PubMed]
- R. Brandenberger, H. Wei, S. Zhang, S. Lei, J. Murage, G.J. Fisk, Y. Li, C. Xu, R. Fang, K. Guegler. Transcriptome characterization elucidates signaling networks that control human ES cell growth and differentiation. Nat. Biotechnol., 2004. [DOI | PubMed]
- Structures and Chromosomal Localizations of Two Human Genes Encoding Synaptobrevins 1 and 2
- S. Isenmann, Y. Khew-Goodall, J. Gamble, M. Vadas, B.W. Wattenberg. A splice-isoform of vesicle-associated membrane protein-1 (VAMP-1) contains a mitochondrial targeting signal. Mol. Biol. Cell, 1998. [DOI | PubMed]
- S.-C. Chen, K.-S. Jeng, M.M.C. Lai. Zinc Finger-Containing Cellular Transcription Corepressor ZBTB25 Promotes Influenza Virus RNA Transcription and Is a Target for Zinc Ejector Drugs. J. Virol., 2017. [DOI | PubMed]
- S. Hanein, E. Martin, A. Boukhris, P. Byrne, C. Goizet, A. Hamri, A. Benomar, A. Lossos, P. Denora, J. Fernandez. Identification of the SPG15 gene, encoding spastizin, as a frequent cause of complicated autosomal-recessive spastic paraplegia, including Kjellin syndrome. Am. J. Hum. Genet., 2008. [DOI | PubMed]
- T. Drewes, A. Clairmont, L. Klein-Hitpass, G.U. Ryffel. Estrogen-inducible derivatives of hepatocyte nuclear factor-4, hepatocyte nuclear factor-3 and liver factor B1 are differently affected by pure and partial antiestrogens. Eur. J. Biochem., 1994. [DOI | PubMed]
- J. A Shaw, K. Udokang, J. Mosquera, H. Chauhan, J.L. Jones, R. A Walker. Oestrogen receptors alpha and beta differ in normal human breast and breast carcinomas. J. Pathol., 2002. [DOI | PubMed]
- J. Hartman, K. Edvardsson, K. Lindberg, C. Zhao, C. Williams, A. Ström, J.-A. Gustafsson. Tumor repressive functions of estrogen receptor β in SW480 colon cancer cells. Cancer Res., 2009. [DOI | PubMed]
- C.-P. Yu, J.-Y. Ho, Y.-T. Huang, T.-L. Cha, G.-H. Sun, D.-S. Yu, F.-W. Chang, S.-P. Chen, R.-J. Hsu. Estrogen inhibits renal cell carcinoma cell progression through estrogen Receptor-β activation. PLoS ONE, 2013. [DOI | PubMed]
- C.-K. Chou, S.-Y. Chi, Y.-Y. Hung, Y.-C. Yang, H.-C. Fu, J.-H. Wang, C.-C. Chen, H.-Y. Kang. Decreased Expression of Estrogen Receptors Is Associated with Tumorigenesis in Papillary Thyroid Carcinoma. Int. J. Mol. Sci., 2022. [DOI | PubMed]
- D. Alvaro, B. Barbaro, A. Franchitto, P. Onori, S.S. Glaser, G. Alpini, H. Francis, L. Marucci, P. Sterpetti, S. Ginanni-Corradini. Estrogens and insulin-like growth factor 1 modulate neoplastic cell growth in human Cholangiocarcinoma. Am. J. Pathol., 2006. [DOI | PubMed]
- W. Al-Khyatt, C. Tufarelli, R. Khan, S.Y. Iftikhar. Selective oestrogen receptor antagonists inhibit oesophageal cancer cell proliferation in vitro. BMC Cancer, 2018. [DOI | PubMed]
- E.G. Shatalova, A.J. Klein-Szanto, K. Devarajan, E. Cukierman, M.L. Clapper. Estrogen and cytochrome P450 1B1 contribute to both early- and late-stage head and neck carcinogenesis. Cancer Prev. Res., 2011. [DOI | PubMed]
- S. Huang, W. Wang, Y. Cheng, J. Lin, M. Wang. Clinicopathological and Prognostic Significance of Klotho and Estrogen Receptors Expression in Human Hepatocellular Carcinoma. Turk. J. Gastroenterol., 2021. [DOI | PubMed]
- The Expression of ERβ2, Bcl-xl and Bax in Non-Small Cell Lung Cancer and Associated with Prognosis
- W.J. van Weelden, C. Reijnen, H.V. Küsters-Vandevelde, J. Bulten, P. Bult, S. Leung, N.C. Visser, M. Santacana, P. Bronsert, M. Hirschfeld. The cutoff for estrogen and progesterone receptor expression in endometrial cancer revisited: A European Network for Individualized Treatment of Endometrial Cancer collaboration study. Hum. Pathol., 2020. [DOI | PubMed]
- J. Trovik, E. Wik, H.M. Werner, C. Krakstad, H. Helland, I. Vandenput, T.S. Njolstad, I.M. Stefansson, J. Marcickiewicz, S. Tingulstad. Hormone receptor loss in endometrial carcinoma curettage predicts lymph node metastasis and poor outcome in prospective multicentre trial. Eur. J. Cancer, 2013. [DOI | PubMed]
- H. Seeliger, I. Pozios, G. Assmann, Y. Zhao, M.H. Müller, T. Knösel, M.E. Kreis, C.J. Bruns. Expression of estrogen receptor beta correlates with adverse prognosis in resected pancreatic adenocarcinoma. BMC Cancer, 2018. [DOI | PubMed]
- W. Song, D. He, Y. Chen, C. Yeh, I. Hsu, Q. Huang, X. Zhang, L.S. Chang, L. Zuo, J. Chen. Targeting newly identified ERβ/TGF-β1/SMAD3 signals with the FDA-approved anti-estrogen Faslodex or an ERβ selective antagonist in renal cell carcinoma. Mol. Oncol., 2018. [DOI | PubMed]
- W. Chen, B. Xin, H. Pang, L. Han, W. Shen, Z. Zhao, L. Duan, P. Cao, L. Liu, H. Zhang. Downregulation of estrogen receptor β inhibits lung adenocarcinoma cell growth. Oncol. Rep., 2019. [DOI | PubMed]
- J.H. Lee, H.K. Kim, B.K. Shin. Expression of female sex hormone receptors and its relation to clinicopathological characteristics and prognosis of lung adenocarcinoma. J. Pathol. Transl. Med., 2020. [DOI | PubMed]
- S. Panza, M. Santoro, F. De Amicis, C. Morelli, V. Passarelli, P. D’aquila, F. Giordano, E. Cione, G. Passarino, D. Bellizzi. Estradiol via estrogen receptor beta influences ROS levels through the transcriptional regulation of SIRT3 in human seminoma TCam-2 cells. Tumor Biol., 2017. [DOI]
- Loss of Estrogen Receptor Beta Expression at Cancer Front Correlates with Tumor Progression and Poor Prognosis of Gallbladder Cancer
- W. Dong, J. Li, H. Zhang, Y. Huang, L. He, Z. Wang, Z. Shan, W. Teng. Altered Expression of Estrogen Receptor β2 is Associated with Different Biological Markers and Clinicopathological Factors in Papillary Thyroid Cancer
- T. Goto, H. Miyamoto. The Role of Estrogen Receptors in Urothelial Cancer. Front. Endocrinol., 2021. [DOI]
- H. Dalal, M. Dahlgren, S. Gladchuk, C. Brueffer, S.K. Gruvberger-Saal, L.H. Saal. Clinical associations of ESR2 (estrogen receptor beta) expression across thousands of primary breast tumors. Sci. Rep., 2022. [DOI | PubMed]
- E.V. Jensen, G. Cheng, C. Palmieri, S. Saji, S. Mäkelä, S. Van Noorden, T. Wahlström, M. Warner, R.C. Coombes, J. Gustafsson. Estrogen receptors and proliferation markers in primary and recurrent breast cancer. Proc. Natl. Acad. Sci. USA, 2001. [DOI | PubMed]
- G.P. Skliris, E. Leygue, L. Curtis-Snell, P.H. Watson, L.C. Murphy. Expression of oestrogen receptor-β in oestrogen receptor-α negative human breast tumours. Br. J. Cancer, 2006. [DOI | PubMed]
- The Cervical Malignant Cells Display a down Regulation of ER-α But Retain the ER-β Expression
- A. Fadiel, S.D. Choi, B. Park, T.-H. Kim, J. Buldo-Licciardi, M. Ahmadi, A. Arslan, K. Mittal, F. Naftolin. Expression of Ezrin and Estrogen Receptors During Cervical Carcinogenesis. Reprod. Sci., 2016. [DOI | PubMed]
- Q.-M. Wang, Y.-J. Qi, Q. Jiang, Y.-F. Ma, L.-D. Wang. Relevance of serum estradiol and estrogen receptor beta expression from a high-incidence area for esophageal squamous cell carcinoma in China. Med. Oncol., 2010. [DOI | PubMed]
- C.P.D.O. Neto, H.O. Brito, R.M. GIL DA Costa, L.M.O. Brito. Is There a Role for Sex Hormone Receptors in Head-and-neck Cancer? Links with HPV Infection and Prognosis. Anticancer Res., 2021. [DOI | PubMed]
- K. Grsic, I.L. Opacic, S. Sitic, M.M. Perisa, P. Suton, B. Sarcevic. The prognostic significance of estrogen receptor β in head and neck squamous cell carcinoma. Oncol. Lett., 2016. [DOI | PubMed]
- A. Atef, M.A. El-Rashidy, S. Elzayat, A.M. Kabel. The prognostic value of sex hormone receptors expression in laryngeal carcinoma. Tissue Cell, 2019. [DOI | PubMed]
- H. Li, H. Chen, J. Shi, Q. Fan, Z. Zhou, X. Tang, Y. Wang, Y. Liu. ERβ overexpression may not be a direct prognostic factor in patients with NSCLC: A meta-analysis. Int. J. Biol. Markers, 2022. [DOI | PubMed]
- H. Yu, D. Gu, P. Qian. Prognostic value of ESR2 expression on adjuvant chemotherapy in completely resected NSCLC. PLoS ONE, 2020. [DOI | PubMed]
- L.P. Stabile, S. Dacic, S.R. Land, D.E. Lenzner, R. Dhir, M. Acquafondata, R.J. Landreneau, J.R. Grandis, J.M. Siegfried. Combined Analysis of Estrogen Receptor β-1 and Progesterone Receptor Expression Identifies Lung Cancer Patients with Poor Outcome. Clin. Cancer Res., 2011. [DOI | PubMed]
- S. Liu, C. Hu, M. Li, J. An, W. Zhou, J. Guo, Y. Xiao. Estrogen receptor beta promotes lung cancer invasion via increasing CXCR4 expression. Cell Death Dis., 2022. [DOI | PubMed]
- I. De Stefano, G.F. Zannoni, M.G. Prisco, A. Fagotti, L. Tortorella, G. Vizzielli, L. Mencaglia, G. Scambia, D. Gallo. Cytoplasmic expression of estrogen receptor beta (ERβ) predicts poor clinical outcome in advanced serous ovarian cancer. Gynecol. Oncol., 2011. [DOI | PubMed]
- S. Konduri, R.E. Schwarz. Estrogen Receptor β/α ratio predicts response of pancreatic cancer cells to estrogens and phytoestrogens. J. Surg. Res., 2007. [DOI | PubMed]
- S.-Y. Li, Y.-X. Wang, L. Wang, Z.-B. Qian, M.-L. Ji. Cytoplasm estrogen receptor β5 as an improved prognostic factor in thymoma and thymic carcinoma progression. Oncol. Lett., 2015. [DOI | PubMed]
- H. Ishibashi, T. Suzuki, S. Suzuki, T. Moriya, C. Kaneko, T. Takizawa, M. Sunamori, M. Handa, T. Kondo, H. Sasano. Sex steroid hormone receptors in human thymoma. J. Clin. Endocrinol. Metab., 2003. [DOI | PubMed]
- M.D. Taves, J.D. Ashwell. Effects of sex steroids on thymic epithelium and thymocyte development. Front. Immunol., 2022. [DOI | PubMed]
- X. Wang, P. Lin, Y. Li, G. Chen, H. Yang, Y. He, Q. Li, R. Liu. Identification of potential agents for thymoma by integrated analyses of differentially expressed tumour-associated genes and molecular docking experiments. Exp. Ther. Med., 2019. [DOI | PubMed]
- H. Ishibashi, T. Suzuki, S. Suzuki, T. Moriya, C. Kaneko, T. Nakata, M. Sunamori, M. Handa, T. Kondo, H. Sasano. Estrogen inhibits cell proliferation through in situ production in human thymoma. Clin. Cancer Res., 2005. [DOI | PubMed]
- T. Mimae, K. Tsuta, F. Takahashi, A. Yoshida, T. Kondo, Y. Murakami, M. Okada, M. Takeuchi, H. Asamura, H. Tsuda. Steroid receptor expression in thymomas and thymic carcinomas. Cancer, 2011. [DOI | PubMed]
- F. Collins, S. MacPherson, P. Brown, V. Bombail, A.R. Williams, R. A Anderson, H.N. Jabbour, P.T. Saunders. Expression of oestrogen receptors, ERα, ERβ, and ERβ variants, in endometrial cancers and evidence that prostaglandin F may play a role in regulating expression of ERα. BMC Cancer, 2009. [DOI | PubMed]
- T.J. Lang. Estrogen as an immunomodulator. Clin. Immunol., 2004. [DOI | PubMed]
- Expression and Localization of Estrogen Receptors in Human Renal Cell Carcinoma and Their Clinical Significance
- J. Ding, C.-R. Yeh, Y. Sun, C. Lin, J. Chou, Z. Ou, C. Chang, J. Qi, S. Yeh. Estrogen receptor β promotes renal cell carcinoma progression via regulating LncRNA HOTAIR-miR-138/200c/204/217 associated CeRNA network. Oncogene, 2018. [DOI | PubMed]
- P. Modarres, F.M. Farsani, A.A. Nekouie, S. Vallian. Meta-analysis of gene signatures and key pathways indicates suppression of JNK pathway as a regulator of chemo-resistance in AML. Sci. Rep., 2021. [DOI | PubMed]
- J.R. Rodríguez-Cid, O. García-Acevedo, J. Benjamin-Contreras, D. Bonilla-Molina, R.R. Flores-Mariñelarena, L. Martínez-Barrera, J.A. Alatorre-Alexander, C.P. Sanchez-Ríos, M.d.R. Flores-Soto, P.J. Santillan-Doherty. Expression of estrogen receptor beta (ERβ) and its prognostic value in pleural mesothelioma. J. Thorac. Dis., 2019. [DOI | PubMed]
- G. Pinton, E. Brunelli, B. Murer, R. Puntoni, M. Puntoni, D.A. Fennell, G. Gaudino, L. Mutti, L. Moro. Estrogen receptor-β affects the prognosis of human malignant mesothelioma. Cancer Res., 2009. [DOI | PubMed]
- K. Pillai, M.H. Pourgholami, T.C. Chua, D.L. Morris. Oestrogen receptors are prognostic factors in malignant peritoneal mesothelioma. J. Cancer Res. Clin. Oncol., 2013. [DOI | PubMed]
- Y. Huang, W. Dong, J. Li, H. Zhang, Z. Shan, W. Teng. Differential expression patterns and clinical significance of estrogen receptor-α and β in papillary thyroid carcinoma. BMC Cancer, 2014. [DOI | PubMed]
- S.S. Jackson, R.M. Pfeiffer, C. Gabbi, L. Anderson, S.M. Gadalla, J. Koshiol. Menopausal hormone therapy and risk of biliary tract cancers. Hepatology, 2021. [DOI]
- E. Kilanczyk, D. Ruminkiewicz, J.M. Banales, P. Milkiewicz, M. Milkiewicz. DHEA Protects Human Cholangiocytes and Hepatocytes against Apoptosis and Oxidative Stress. Cells, 2022. [DOI | PubMed]
- D. Alvaro, G. Alpini, P. Onori, A. Franchitto, S. Glaser, G. Le Sage, F. Folli, A. Attili, E. Gaudio. Alfa and beta estrogen receptors and the biliary tree. Mol. Cell. Endocrinol., 2002. [DOI | PubMed]
- M.R. Castellanos, E. Fanous, R. Thaker, M.J. Flory, N. Seetharamu, M. Dhar, A. Starr, T.J. Strange. Expression patterns and clinical significance of estrogen receptor in non-small cell lung cancer. Pathol.-Res. Pr., 2023. [DOI | PubMed]
- Y. Hu, Y. Xu, T. Zhang, Q. Han, L. Li, M. Liu, N. Li, G. Shao. Cisplatin-activated ERβ/DCAF8 positive feedback loop induces chemoresistance in non-small cell lung cancer via PTEN/Akt axis. Drug Resist. Updat., 2023. [DOI | PubMed]
- G.R. Sareddy, B.C. Nair, V.K. Gonugunta, Q.-G. Zhang, A. Brenner, D.W. Brann, R.R. Tekmal, R.K. Vadlamudi. Therapeutic significance of estrogen receptor β agonists in gliomas. Mol. Cancer Ther., 2012. [DOI | PubMed]
- A. Batistatou, D. Stefanou, A. Goussia, E. Arkoumani, A.G. Papavassiliou, N.J. Agnantis. Estrogen receptor beta (ERβ) is expressed in brain astrocytic tumors and declines with dedifferentiation of the neoplasm. J. Cancer Res. Clin. Oncol., 2004. [DOI | PubMed]
- A. Batistatou, P. A Kyzas, A. Goussia, E. Arkoumani, S. Voulgaris, K. Polyzoidis, N.J. Agnantis, D. Stefanou. Estrogen receptor beta (ERβ) protein expression correlates with BAG-1 and prognosis in brain glial tumours. J. Neuro-Oncol., 2005. [DOI | PubMed]
- T. Zellweger, S. Stürm, S. Rey, I. Zlobec, J.R. Gsponer, C. A Rentsch, L.M. Terracciano, A. Bachmann, L. Bubendorf, C. Ruiz. Estrogen receptor β expression and androgen receptor phosphorylation correlate with a poor clinical outcome in hormone-naïve prostate cancer and are elevated in castration-resistant disease. Endocr.-Relat. Cancer, 2013. [DOI | PubMed]
- H. Bonkhoff, R. Berges. The Evolving role of oestrogens and their receptors in the development and progression of prostate cancer. Eur. Urol., 2009. [DOI | PubMed]
- T. Grindstad, K. Skjefstad, S. Andersen, N. Ness, Y. Nordby, S. Al-Saad, S. Fismen, T. Donnem, M.R. Khanehkenari, L.-T. Busund. Estrogen receptors α and β and aromatase as independent predictors for prostate cancer outcome. Sci. Rep., 2016. [DOI | PubMed]
- Y.-K. Leung, H.-M. Lam, S. Wu, D. Song, L. Levin, L. Cheng, C.-L. Wu, S.-M. Ho. Estrogen receptor β2 and β5 are associated with poor prognosis in prostate cancer, and promote cancer cell migration and invasion. Endocr.-Relat. Cancer, 2010. [DOI | PubMed]
- L. Barzon, G. Masi, M. Pacenti, M. Trevisan, F. Fallo, A. Remo, G. Martignoni, D. Montanaro, V. Pezzi, G. Palù. Expression of aromatase and estrogen receptors in human adrenocortical tumors. Virchows Arch., 2007. [DOI | PubMed]
- K.M. Eyster. The Estrogen Receptors: An Overview from Different Perspectives. Methods Mol. Biol., 2016. [DOI | PubMed]
- K. Yakimchuk, M. Iravani, M.S. Hasni, P. Rhönnstad, S. Nilsson, M. Jondal, S. Okret. Effect of ligand-activated estrogen receptor β on lymphoma growth in vitro and in vivo. Leukemia, 2011. [DOI | PubMed]
- Z. Han, Y. Zhang, Y. Sun, J. Chen, C. Chang, X. Wang, S. Yeh. ERβ-Mediated Alteration of circATP2B1 and miR-204-3p Signaling Promotes Invasion of Clear Cell Renal Cell Carcinoma. Cancer Res., 2018. [DOI | PubMed]
- C.Y. Xu, J.L. Guo, Z.N. Jiang, S.D. Xie, J.Y. Shen, L.B. Wang. Prognostic role of estrogen receptor α and estrogen receptor β in gastric cancer. Ann. Surg. Oncol., 2010. [DOI | PubMed]
- P. Mak, I. Leav, B. Pursell, D. Bae, X. Yang, C.A. Taglienti, L.M. Gouvin, V.M. Sharma, A.M. Mercurio. ERβ impedes prostate cancer EMT by destabilizing HIF-1α and inhibiting VEGF-mediated snail nuclear localization: Implications for gleason grading. Cancer Cell, 2010. [DOI | PubMed]
- J. Fares, M.Y. Fares, H.H. Khachfe, H.A. Salhab, Y. Fares. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther., 2020. [DOI | PubMed]
- T. Bae, S.P. Hallis, M.-K. Kwak. Hypoxia, oxidative stress, and the interplay of HIFs and NRF2 signaling in cancer. Exp. Mol. Med., 2024. [DOI | PubMed]
- P. Dey, P. Jonsson, J. Hartman, C. Williams, A. Ström, J. Gustafsson. Estrogen receptors β1 and β2 have opposing roles in regulating proliferation and bone metastasis genes in the prostate cancer cell line PC3. Mol. Endocrinol., 2012. [DOI | PubMed]
- A.A. Rawłuszko-Wieczorek, J. Lipowicz, M. Nowacka, K. Ostrowska, P. Pietras, M. Blatkiewicz, M. Ruciński, P.P. Jagodziński, M. Nowicki. Estrogen receptor β affects hypoxia response in colorectal cancer cells. Biochim. Biophys. Acta-Mol. Basis Dis., 2024. [DOI | PubMed]
- B. Refaat, A. Aslam, S. Idris, A.H. Almalki, M.Y. Alkhaldi, H.A. Asiri, R.A. Almaimani, A. Mujalli, F. Minshawi, S.A. Alamri. Profiling estrogen, progesterone, and androgen receptors in colorectal cancer in relation to gender, menopausal status, clinical stage, and tumour sidedness. Front. Endocrinol., 2023. [DOI | PubMed]
- W. Lin, Y. Yan, Q. Huang, D. Zheng. MDMX in Cancer: A Partner of p53 and a p53-Independent Effector. Biol. Targets Ther., 2024. [DOI | PubMed]
- F. Toledo, G.M. Wahl. MDM2 and MDM4: p53 regulators as targets in anticancer therapy. Int. J. Biochem. Cell Biol., 2007. [DOI | PubMed]
- Q. Li, G. Lozano. Molecular Pathways: Targeting Mdm2 and Mdm4 in Cancer Therapy. Clin. Cancer Res., 2013. [DOI | PubMed]
- H. Zhao, Y.-Z. Xie, R. Xing, M. Sun, F. Chi, Y.-C. Zeng. MDMX is a prognostic factor for non-small cell lung cancer and regulates its sensitivity to cisplatin. Cell. Oncol., 2017. [DOI | PubMed]
- J. Hong, A. Shiba-Ishii, Y. Kim, M. Noguchi, N. Sakamoto. Ovarian carcinoma immunoreactive antigen domain 2 controls mitochondrial apoptosis in lung adenocarcinoma. Cancer Sci., 2021. [DOI | PubMed]
- T.-F. Chen, H.-F. Hao, Y. Zhang, X.-Y. Chen, H.-S. Zhao, R. Yang, P. Li, L.-X. Qiu, Y.-H. Sang, C. Xu. HBO1 induces histone acetylation and is important for non-small cell lung cancer cell growth. Int. J. Biol. Sci., 2022. [DOI | PubMed]
- Z. Zhang, Q. Zhao, Z. Wang, F. Xu, Y. Liu, Y. Guo, C. Li, T. Liu, Y. Zhao, X. Tang. Hepatocellular carcinoma cells downregulate NADH:Ubiquinone Oxidoreductase Subunit B3 to maintain reactive oxygen species homeostasis. Hepatol. Commun., 2024. [DOI | PubMed]
- Relationship of Intracellular Proteolysis with CAP1 and Cofilin1 in Non-Small-Cell Lung Cancer
- Y. Li, H. Wang, Y. Pan, S. Wang, Z. Zhang, H. Zhou, M. Xu, X. Liu. Identification of bicalutamide resistance-related genes and prognosis prediction in patients with prostate cancer. Front. Endocrinol., 2023. [DOI]
- A.C.M. Sousa-Squiavinato, R.I. Vasconcelos, A.S. Gehren, P.V. Fernandes, I.M. de Oliveira, M. Boroni, J.A. Morgado-Díaz. Cofilin-1, LIMK1 and SSH1 are differentially expressed in locally advanced colorectal cancer and according to consensus molecular subtypes. Cancer Cell Int., 2021. [DOI | PubMed]
- K.-W. Zheng, C.-H. Zhang, W. Wu, Z. Zhu, J.-P. Gong, C.-M. Li. FNBP4 is a Potential Biomarker Associated with Cuproptosis and Promotes Tumor Progression in Hepatocellular Carcinoma. Int. J. Gen. Med., 2023. [DOI | PubMed]
- C.-T. Wu, Y.-L. Chang, J.-Y. Shih, Y.-C. Lee. The significance of estrogen receptor β in 301 surgically treated non–small cell lung cancers. J. Thorac. Cardiovasc. Surg., 2005. [DOI | PubMed]
- M. Maki, H. JeongMin, T. Nakagawa, H. Kawai, N. Sakamoto, Y. Sato, M. Noguchi. Aberrant OCIAD2 demethylation in lung adenocarcinoma is associated with outcome. Pathol. Int., 2022. [DOI | PubMed]
- S.J. King, K. Nowak, N. Suryavanshi, I. Holt, C.M. Shanahan, A.J. Ridley. Nesprin-1 and nesprin-2 regulate endothelial cell shape and migration. Cytoskeleton, 2014. [DOI | PubMed]
- D.T. Warren, T. Tajsic, J.A. Mellad, R. Searles, Q. Zhang, C.M. Shanahan. Novel nuclear nesprin-2 variants tether active extracellular signal-regulated MAPK1 and MAPK2 at promyelocytic leukemia protein nuclear bodies and act to regulate smooth muscle cell proliferation. J. Biol. Chem., 2010. [DOI | PubMed]
- The ACIN1 Gene Is Hypermethylated in Early Stage Lung Adenocarcinoma
- Y.-S. Chen, C.-W. Liu, Y.-C. Lin, C.-Y. Tsai, C.-H. Yang, J.-C. Lin. The SRSF3-MBNL1-Acin1 circuit constitutes an emerging axis to lessen DNA fragmentation in colorectal cancer via an alternative splicing mechanism. Neoplasia, 2020. [DOI | PubMed]
- Y.A. Valentin-Vega, J.A. Barboza, G.P. Chau, A.K. El-Naggar, G. Lozano. High levels of the p53 inhibitor MDM4 in head and neck squamous carcinomas. Hum. Pathol., 2007. [DOI | PubMed]
- X. Men, W. Zhu. Silencing of Perilipin 3 Inhibits Lung Adenocarcinoma Cell Immune Resistance by Regulating the Transcription of PD-L1 Through c-Myc. Immunol. Investig., 2023. [DOI | PubMed]
- J. Zhang, Y. Li, Y. Yang, J. Huang, Y. Sun, X. Zhang, X. Kong. A novel iTreg-related signature for prognostic prediction in lung adenocarcinoma. Cancer Sci., 2023. [DOI | PubMed]
- H. Zhou, A. Brekman, W.-L. Zuo, X. Ou, R. Shaykhiev, F.J. Agosto-Perez, R. Wang, M.S. Walters, J. Salit, Y. Strulovici-Barel. POU2AF1 Functions in the Human Airway Epithelium To Regulate Expression of Host Defense Genes. J. Immunol., 2016. [DOI | PubMed]
- K. Luck, D.-K. Kim, L. Lambourne, K. Spirohn, B.E. Begg, W. Bian, R. Brignall, T. Cafarelli, F.J. Campos-Laborie, B. Charloteaux. A reference map of the human binary protein interactome. Nature, 2020. [DOI | PubMed]
- C. Haenig, N. Atias, A.K. Taylor, A. Mazza, M.H. Schaefer, J. Russ, S.-P. Riechers, S. Jain, M. Coughlin, J.-F. Fontaine. Interactome Mapping Provides a Network of Neurodegenerative Disease Proteins and Uncovers Widespread Protein Aggregation in Affected Brains. Cell Rep., 2020. [DOI | PubMed]
- L. Sun, X. Yang, A. Khan, X. Yu, H. Zhang, S. Han, X. Habulieti, Y. Sun, R. Wang, X. Zhang. Panoramic variation analysis of a family with neurodevelopmental disorders caused by biallelic loss-of-function variants in TMEM141, DDHD2, and LHFPL. Front. Med., 2023. [DOI | PubMed]
- R.T. Woodland, C.J. Fox, M.R. Schmidt, P.S. Hammerman, J.T. Opferman, S.J. Korsmeyer, D.M. Hilbert, C.B. Thompson. Multiple signaling pathways promote B lymphocyte stimulator–dependent B-cell growth and survival. Blood, 2008. [DOI | PubMed]
- J. Jellusova, A.V. Miletic, M.H. Cato, W.-W. Lin, Y. Hu, G.A. Bishop, M.J. Shlomchik, R.C. Rickert. Context-specific BAFF-R signaling by the NF-κB and PI3K pathways. Cell Rep., 2013. [DOI | PubMed]
- F.M. Wensveen, E. Slinger, M.H. van Attekum, R. Brink, E. Eldering. Antigen-affinity controls pre-germinal center B cell selection by promoting Mcl-1 induction through BAFF receptor signaling. Sci. Rep., 2016. [DOI | PubMed]
- Q. Zeng, S. Qin, H. Zhang, B. Liu, J. Qin, X. Wang, R. Zhang, C. Liu, X. Dong, S. Zhang. Rapamycin attenuates BAFF-extended proliferation and survival via disruption of mTORC1/2 signaling in normal and neoplastic B-lymphoid cells. J. Cell. Physiol., 2017. [DOI | PubMed]
- H. Peng, Z. Huang, P. Li, Z. Sun, X. Hou, Z. Li, R. Sang, Z. Guo, S. Wu, Y. Cao. Investigating the efficacy and mechanisms of Jinfu’ an decoction in treating non-small cell lung cancer using network pharmacology and in vitro and in vivo experiments. J. Ethnopharmacol., 2024. [DOI | PubMed]
- H. Marei, A. Malliri. Rac1 in human diseases: The therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases, 2016. [DOI | PubMed]
- B.-W. Park, K.-S. Kim, M.-K. Heo, W.-I. Yang, S.I. Kim, J.-H. Kim, G.E. Kim, K.S. Lee. The changes of estrogen receptor-β variants expression in breast carcinogenesis: Decrease of estrogen receptor-β2 expression is the key event in breast cancer development. J. Surg. Oncol., 2006. [DOI | PubMed]
- E. Cerami, J. Gao, U. Dogrusoz, B.E. Gross, S.O. Sumer, B.A. Aksoy, A. Jacobsen, C.J. Byrne, M.L. Heuer, E. Larsson. The cBio cancer genomics portal: An open platform for exploring multidimensional cancer genomics data. Cancer Discov., 2012. [DOI | PubMed]
- J. Gao, B.A. Aksoy, U. Dogrusoz, G. Dresdner, B.E. Gross, S.O. Sumer, Y. Sun, A. Jacobsen, R. Sinha, E. Larsson. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal., 2013. [DOI | PubMed]
- I. de Bruijn, R. Kundra, B. Mastrogiacomo, T.N. Tran, L. Sikina, T. Mazor, X. Li, A. Ochoa, G. Zhao, B. Lai. Analysis and Visualization of Longitudinal Genomic and Clinical Data from the AACR Project GENIE Biopharma Collaborative in cBioPortal. Cancer Res., 2023. [DOI | PubMed]
- B. Li, C.N. Dewey. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform., 2011. [DOI | PubMed]
- M. Uhlén, L. Fagerberg, B.M. Hallström, C. Lindskog, P. Oksvold, A. Mardinoglu, Å. Sivertsson, C. Kampf, E. Sjöstedt, A. Asplund. Proteomics. Tissue-Based Map of the Human Proteome. Science, 2015. [DOI | PubMed]
- M. Uhlén, E. Björling, C. Agaton, C.A.-K. Szigyarto, B. Amini, E. Andersen, A.-C. Andersson, P. Angelidou, A. Asplund, C. Asplund. A human protein atlas for normal and cancer tissues based on antibody proteomics. Mol. Cell. Proteom. MCP, 2005. [DOI | PubMed]
- T. Li, J. Fu, Z. Zeng, D. Cohen, J. Li, Q. Chen, B. Li, X.S. Liu. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res., 2020. [DOI | PubMed]
- T. Li, J. Fan, B. Wang, N. Traugh, Q. Chen, J.S. Liu, B. Li, X.S. Liu. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res., 2017. [DOI | PubMed]
- R.D. Lasley, R.M. Mentzer. Preconditioning and its potential role in myocardial protection during cardiac surgery. J. Card. Surg., 1995. [DOI | PubMed]
- B. Ru, C.N. Wong, Y. Tong, J.Y. Zhong, S.S.W. Zhong, W.C. Wu, K.C. Chu, C.Y. Wong, C.Y. Lau, I. Chen. TISIDB: An integrated repository portal for tumor–immune system interactions. Bioinformatics, 2019. [DOI | PubMed]
- A. Subramanian, P. Tamayo, V.K. Mootha, S. Mukherjee, B.L. Ebert, M.A. Gillette, A. Paulovich, S.L. Pomeroy, T.R. Golub, E.S. Lander. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA, 2005. [DOI | PubMed]
- V.K. Mootha, C.M. Lindgren, K.-F. Eriksson, A. Subramanian, S. Sihag, J. Lehar, P. Puigserver, E. Carlsson, M. Ridderstråle, E. Laurila. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet., 2003. [DOI | PubMed]
- M. Mobashir, S.P. Turunen, M.A. Izhari, I.M. Ashankyty, T. Helleday, K. Lehti. An Approach for Systems-Level Understanding of Prostate Cancer from High-Throughput Data Integration to Pathway Modeling and Simulation. Cells, 2022. [DOI | PubMed]
- L.R. Nassar, G.P. Barber, A. Benet-Pagès, J. Casper, H. Clawson, M. Diekhans, C. Fischer, J.N. Gonzalez, A.S. Hinrichs, B.T. Lee. The UCSC Genome Browser database: 2023 update. Nucleic Acids Res., 2022. [DOI | PubMed]
