Diagnostic accuracy of cerebral [18F]FDG PET in atypical parkinsonism
Abstract
Background:
Atypical parkinsonism (AP) often presents with Parkinson’s symptoms but has a much worse long-term prognosis. The diagnosis is presently based on clinical criteria, but a cerebral positron emission tomography (PET) scan with [18F]fluoro-2-deoxy-2-d-glucose ([18F]FDG) may assist in the diagnosis of AP such as multiple system atrophy (MSA), progressive supranuclear palsy (PSP), corticobasal degeneration (CBD), and Lewy body dementia (DLB). Only few studies have evaluated the sensitivity and specificity of [18F]FDG PET for separating the diseases in a mixed patient population, which we aim to assess in a retrospective material.
Results:
We identified 156 patients referred for a cerebral [18F]FDG PET for suspicion of AP during 2017–2019. The [18F]FDG PET was analysed by a nuclear medicine specialist blinded to clinical information but with access to dopamine transporter imaging. The reference standard was the follow-up clinical diagnosis (follow-up: 6–72 months). The overall accuracy for correct classification was 74%. Classification sensitivity (95% confidence interval, CI) and specificity (95% CI) for MSA (n = 20) were 1.00 (0.83–1.00) and 0.91 (0.85–0.95), for DLB/Parkinson with dementia (PDD) (n = 26) were 0.81 (0.61–0.93) and 0.97 (0.92–0.99) and for CBD/PSP (n = 68) were 0.62 (0.49–0.73) and 0.97 (0.90–0.99).
Conclusions:
Our results support the additional use of [18F]FDG PET for the clinical diagnosis of AP with moderate to high sensitivity and specificity. Use of [18F]FDG PET may be beneficial for prognosis and supportive treatment of the patients and useful for future clinical treatment trials.
Article type: Research Article
Keywords: Positron emission tomography, Neurodegenerative disorders, Progressive supranuclear palsy, Lewy body dementia, Multiple system atrophy, Corticobasal degeneration, Sensitivity, Specificity
Affiliations: https://ror.org/05bpbnx46grid.4973.90000 0004 0646 7373Department of Clinical Physiology and Nuclear Medicine, Copenhagen University Hospital Bispebjerg, Bispebjerg Bakke 23, Copenhagen, Denmark; https://ror.org/035b05819grid.5254.60000 0001 0674 042XFaculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark; https://ror.org/05bpbnx46grid.4973.90000 0004 0646 7373Department of Neurology, Copenhagen University Hospital Bispebjerg, Copenhagen, Denmark
License: © The Author(s) 2023 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s13550-023-01025-x | PubMed: 37572162 | PMC: PMC10423182
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (903 KB)
Background
Parkinson’s disease affects approximately 1% of individuals over the age of 65. Among them 75% have idiopathic Parkinson’s disease (IPD), while the remaining 25% have atypical syndromes, including symptomatic parkinsonism [ref. 1]. Some patients initially exhibit symptoms of Parkinson’s disease but quickly progress to more severe and potentially life-threatening symptoms. These are classified under atypical parkinsonism (AP), which encompasses: Dementia with Lewy Bodies (DLB); Progressive Supranuclear Palsy (PSP), Multiple System Atrophy (MSA) and Corticobasal Degeneration (CBD). The incidence rate of AP is 2.5–5.9 per 100.000 person-years and it increases with age. Patients with AP have a mean survival of 1.8–9.5 years from diagnosis [ref. 2–ref. 4].
Precise AP diagnosis is crucial for accurate prognosis and effective therapy, but the disease is often difficult to diagnose in early stages based on clinical criteria alone [ref. 5]. Some studies have employed cerebral [18F]FDG PET for improved differential diagnosis of Parkinson’s disease, as it can identify regional metabolism patterns related to each AP subtype [ref. 6–ref. 8]. However, the sensitivity and specificity of cerebral [18F]FDG PET in diagnosing AP remain under-examined, and evidence supporting its routine use is limited [ref. 8, ref. 9].
Although some studies report high sensitivity and specificity (> 85%) in diagnosing AP subtypes, these studies primarily focus on selected populations with diagnoses of either AP or IPD [ref. 10, ref. 11]. A prospective study on a mixed population found high sensitivity and specificity in distinguishing AP from DLB/PDD [ref. 12]. Nevertheless, no studies following the Standards for Reporting Diagnostic accuracy studies (STARD) guidelines have investigated the sensitivity and specificity of cerebral [18F]FDG PET in diagnosing AP in a mixed population. This is especially important when considering a diverse patient population reflective of daily clinical routine.
This study therefore aims to assess the diagnostic accuracy of additional cerebral [18F]FDG PET in diagnosing AP and its subtypes, including MSA, DLB/PDD and 4-repeat tauopathies (4R-tauopathies), which encompass CBD and PSP, within a mixed population.
Material and methods
Ethical approvals
This study received approval from the Danish Patient Safety Authority (31-1521-255) and the Danish Data Protection Agency (P-2020-530).
Study population
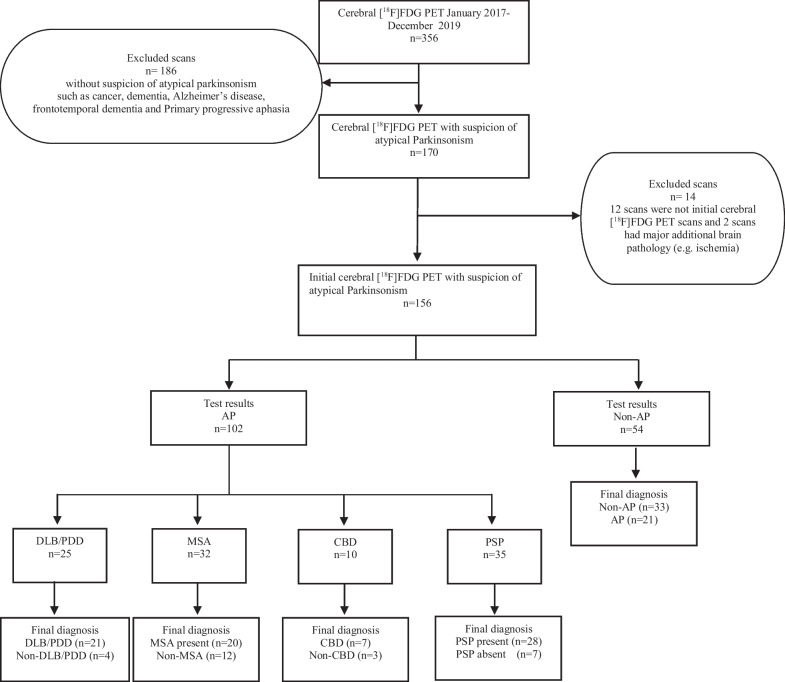
All cerebral [18F]FDG PET scans patient conducted from January 1st, 2017 to December 31st, 2019 at the Department of Clinical Physiology and Nuclear Medicine after referral from the Department of Neurology at Copenhagen University Hospital Bispebjerg were reviewed. The department is a tertiary movement disorder clinic evaluating approximately 2500 patients annually with diverse movement disorders. This study included patients with suspected Atypical Parkinsonism (AP) and excluded individuals showing initial symptoms or diagnoses of other diseases such as cancer, dementia, or Alzheimer’s disease. In cases where multiple cerebral [18F]FDG PET scans were available, the first referral indicating the possibility of AP was selected for analysis. Figure 1 provides the flowchart of inclusion.
A specialized physician with expertise in movement disorders clinically evaluated all participants. The Montreal Cognitive Assessment (MoCA) tool was used to measure global cognitive functions. Patients with MoCA scores above 22 underwent additional neuropsychological examinations.
Most patients underwent a Magnetic Resonance Imaging (MRI) scan (87%) and a DAT scan (84%). As part of the initial clinical assessment, 63% underwent a MoCA test and 70% of the patients underwent spinal fluid examinations, including routine tests as well as specific assessments for Alzheimer’s disease and Neurofilament Light Chain levels.
We adopted the diagnostic criteria for the subtypes of AP, including PSP [ref. 13], MSA [ref. 14], DLB/PDD [ref. 15] and CBD [ref. 16]. We decided to pool PSP and CBD as well as DLB and PDD due to the overlap of clinical presentation and similar neuropathology (Fig. 2).
We classified MSA based on features of parkinsonism (striatonigral degeneration, subgroup MSA-P) or cerebellar dysfunction (olivopontocerebellar atrophy, subgroup MSA-C).
Imaging methods
Subjects fasted 6 h prior to the PET study. The patients were injected with 199 ± 4.9 MBq of [18F]FDG intravenously, followed by a rest period of 60 min. Subsequently, patients underwent a 10 min [18F]FDG PET/CT scan in a digital Discovery MI PET/CT (GE Healthcare, Milwaukee, USA) in 3-dimensional mode. The ordered subset expectation maximization (OSEM) algorithm was used for image reconstruction and attenuation correction with CT. The [18F]FDG PET acquisition procedures complied with the European Association of Nuclear Medicine guidelines [ref. 17].
Imaging analysis
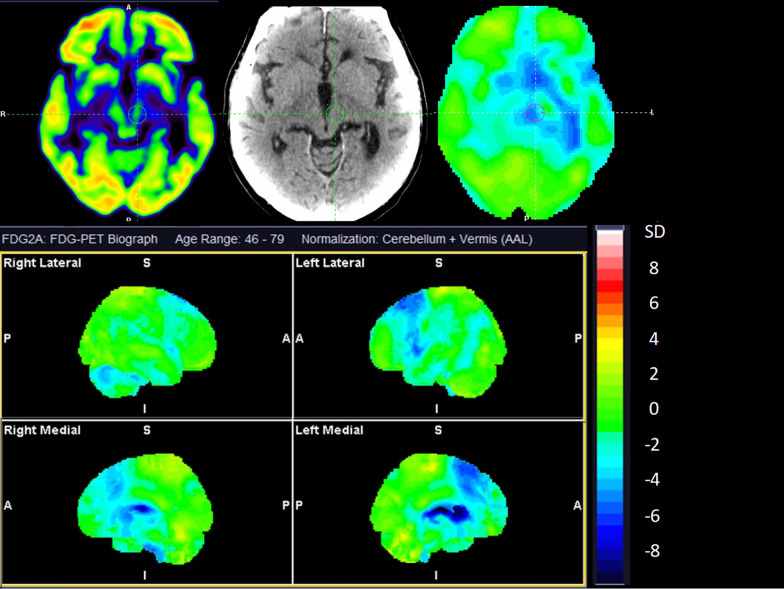
We used Scenium, SyngoVia (Siemens Healthineers, Erlangen, Germany) to analyse the images of the included patients. A nuclear medicine specialist with more than 10 years of experience in PET neuroimaging (LM) classified the scans as MSA-C, MSA-P, CBD, PSP, DLB, or non-AP, based on previously published disease-specific [18F]FDG features [ref. 18]. Briefly, a reduced [18F]FDG uptake in the cerebellum would suggest a potential diagnosis of MSA-C. MSA-P was considered when there was a symmetric or slightly asymmetric reduction in activity uptake in the posterior putamen. This suspicion was strengthened if both the posterior putamen and cerebellum showed decreased activity uptake with an absence of cortical involvement. CBD was considered if an asymmetrical reduction in frontoparietal activity was observed, alongside involvement of the ipsilateral basal ganglia and mesencephalon. For PSP, a more symmetrical reduction in activity uptake in the mesial and lateral frontal cortex, basal ganglia, and particularly the mesencephalon was indicative. DLB was typically characterized by a reduction in parietotemporooccipital activity uptake, along with the presence of a cingulate island sign. The reader was blinded to all clinical information during the classification process except for previous neuroimaging, if available. Thus, MRI scans and results from dopamine transporter imaging were visually interpreted without access to clinical data and used for reading of the [18F]FDG PET. In cases of uncertainty regarding the classification of scans, the nuclear medicine specialist would make a decision based on the best professional judgment to prevent any instances of missing data.
Reference standard
We used the clinical diagnosis as the reference standard, based on the international consensus criteria13−16, all clinical information, test results and initial results from the cerebral [18F]FDG. A medical student (NJH) reviewed the electronic patient journal and reviewed diagnoses, medications, treatment plans, and progress plans recorded at the Department of Neurology, Copenhagen University Hospital Bispebjerg, from the time of the initial cerebral [18F]FDG PET up to June 2021. If the diagnosis was unclear based on the medical record, a neurologist with more than 10 years of experience in movement disorders (AH) reviewed the journal for a final diagnosis. In cases of uncertainty regarding the final diagnosis, the neurologist would make a decision based on the best professional judgment.
Statistical analysis
We assessed the sensitivity, specificity, and both positive and negative predictive values (PPV and NPV) by comparing the classification using [18F]FDG PET with the reference standard. For robust results, the diseases were divided into four groups: MSA, 4R-tauopathies (CBD/PSP), DLB/PDD, and non-AP. Confidence intervals were calculated using R statistical software (R Core Team, 2017; R Foundation for Statistical Computing, Vienna, Austria; https://www.R-project.org). We followed the standards for reporting diagnostic accuracy studies (STARD).
Results
After screening patient referrals, we excluded 200 [18F]FDG PET scans due to suspicions other than AP, such as cancer, dementia, or Alzheimer’s disease. Of these, 14 were follow-up cerebral [18F]FDG PET or had major MRI findings precluding a reliable AP diagnosis (Fig. 1) leaving a total of 156 patients referred for a cerebral [18F]FDG PET for suspicion of AP. Please refer to Table 1 for demographics of included patients. The patients had a median follow-up time of 23 months (range 6–72 months) after the cerebral [18F]FDG PET and 24 patients died in the study period. The follow-up diagnosis of the patients were MSA (n = 20, of which MSA-C (n = 5) and MSA-P (n = 15), 4R-tauopathies (n = 68), of which CBD (n = 23) and PSP (n = 45), DLB/PDD (n = 26), and non-AP (n = 42). Unfortunately, the patients did not have a histopathological diagnosis. [18F]FDG PET correctly classified a total of 116 (74%) patients into the four groups: MSA: 20, DLB/PDD: 21, 4R-tauopaties: 42, and non-AP: 33. Please refer to Tables 2 and 3 for overview of the classifications and subclassifications relative to follow-up. The sensitivity/specificity for AP was 0.82 (0.73–0.88)/0.79 (0.63–0.88), MSA was 1.00 (0.83–1.00)/0.91 (0.85–0.95), DLB/PDD was 0.81 (0.61–0.93)/0.97 (0.92–0.99), and 4R-tauopathies 0.62 (0.49–0.73)/0.97 (0.90–0.99) (Table 4).
Table 1: Subject demographics
| Follow up diagnosis | N | M:F | Mean age ± standard deviation | Median follow-up time (from PET to follow-up/ months (range) | Number with previous DAT imaging | Number with previous MRI | Number with previous MoCa test | Mean MoCa score ± standard deviation |
|---|---|---|---|---|---|---|---|---|
| MSA | 20 | 5:15 | 63.4 ± 3.8 | 21 (4–40) | 18 | 17 | 17 | 22.8 ± 4.6 |
| DLB/PDD | 26 | 16:10 | 71.7 ± 3.5 | 22 (7–42) | 18 | 20 | 12 | 21.9 ± 3.8 |
| CBD | 23 | 12:11 | 74.5 ± 2.0 | 24 (10–72) | 23 | 19 | 13 | 23.2 ± 3.7 |
| PSP | 45 | 25:20 | 72.2 ± 2.1 | 24 (6–50) | 41 | 41 | 29 | 21.7 ± 4.7 |
| Non-AP | 42 | 32:10 | 70.9 ± 2.8 | 23 (15–37) | 30 | 40 | 28 | 21.9 ± 5.7 |
M, males; F, females; MSA, multiple system atrophy; DLB/PDD, Dementia with Lewy bodies/Parkinson’s disease with dementia; CBS, corticobasal syndrome; PSP, progressive supranuclear palsy; AP, atypical parkinsonism
Table 2: Classifications
| Follow up diagnosis | Imaging test outcome N = 156 | Total | |||
|---|---|---|---|---|---|
| MSA | DLB/PDD | 4R-Tauopaties (CBD/PSP) | Non-AP | ||
| MSA | 20 | 0 | 0 | 0 | 20 |
| DLB/PDD | 0 | 21 | 0 | 5 | 26 |
| 4R-Tauopathies (CBD/PSP) | 8 | 2 | 42 | 16 | 68 |
| Non-AP | 4 | 2 | 3 | 33 | 42 |
| Total | 32 | 25 | 45 | 54 | 156 |
Rows with the follow-up diagnosis and columns with the corresponding imaging classification. MSA, multiple system atrophy; DLB/PDD, Dementia with Lewy bodies/Parkinson’s disease with dementia; CBD, corticobasal degeneration; PSP, progressive supranuclear palsy; AP, atypical parkinsonism
Table 3: Subgroup classifications
| Follow up diagnosis | Imaging test outcome N = 42 | Follow up diagnosis | Imaging test outcome N = 20 | ||
|---|---|---|---|---|---|
| CBD | PSP | MSA-P | MSA-C | ||
| CBD | 7 | 4 | MSA-P | 12 | 3 |
| PSP | 3 | 28 | MSA-C | 0 | 5 |
Left: subgroup classification of the 4R-Tauopathies. Right: subgroup classification of MSA. Rows with follow-up diagnosis and columns with the corresponding imaging classification
CBD, corticobasal degeneration; PSP, progressive supranuclear palsy; MSA-P, multiple system atrophy with striatonigral degeneration; MSA-C, multiple system atrophy with cerebellar dysfunction
Table 4: Sensitivities and specificities
| Follow-up diagnosis | Sensitivity (95% CI) | Specificity (95% CI) | Positive predictive value (95% CI) | Negative predictive value (95% CI) |
|---|---|---|---|---|
| AP | 0.82 (0.73–0.88) | 0.79 (0.63–0.88) | 0.91 (0.85–0.95) | 0.61 (0.51–0.70) |
| MSA | 1.00 (0.83–1.00) | 0.91 (0.85–0.95) | 0.62 (0.49–0.74) | 1.00 (0.97–1.00) |
| DLB/PDD | 0.81 (0.61–0.93) | 0.97 (0.92–0.99) | 0.84 (0.66–0.93) | 0.96 (0.92–0.98) |
| 4R-tauopathies (CBD/PSP) | 0.62 (0.49–0.73) | 0.97 (0.90–0.99) | 0.93 (0.82–0.99) | 0.77 (0.71–0.82) |
| NON-AP | 0.79 (0.63–0.88) | 0.82 (0.73–0.88) | 0.61 (0.51–0.70) | 0.91 (0.85–0.95) |
AP, atypical parkinsonism; MSA, multiple system atrophy; DLB/PDD, Dementia with Lewy bodies/Parkinson’s disease with dementia; CBD, corticobasal degeneration; PSP, progressive supranuclear palsy
Discussion
Our study demonstrate that cerebral [18F]FDG PET provides a moderate to high diagnostic sensitivity and specificity for differentiation between subtypes of AP with significant sensitivity (> 80%) and specificity (> 90%). However, the sensitivity falls to approximately 62% for 4R-tauopathies, mirroring the known diagnostic challenges of these conditions. These findings support earlier studies that showed the efficacy of [18F]FDG PET in differentiating AP subtypes, which reported similar levels of sensitivity and specificity (> 85%) [ref. 10, ref. 11].
Challenges arise with CBD due to its overlapping metabolic pattern with PSP, which was identified in a prior prospective study that reported a low PPV (67%) for CBD [ref. 12]. This is likely because CBD and PSP share certain neuropathological features, such as basophilic inclusions and distinct cytoskeletal abnormalities, making them difficult to differentiate [ref. 19]. Our study’s moderate sensitivity in diagnosing 4R-tauopathies, particularly CBD, aligns with the general difficulties encountered in their accurate clinical diagnosis, as reported in previous studies [ref. 20, ref. 21].
The strengths of our study include the inclusion of a diverse patient population consecutively referred for cerebral [18F]FDG imaging due to suspected atypical parkinsonism, thereby reflecting real-world clinical practice. We also adhered to the standards for reporting diagnostic accuracy studies.
Limitations of our study include potential bias of the reference standard. The clinical diagnosis might have been influenced by neuroimaging results, challenging the independence of reference standards from the test results. A follow-up consultation would be necessary to assess the neurological status and clinical appearance without access to imaging data, which would significantly reduce the sample size of the most ill subjects, as 24 patients died in the study period, and a significant portion of the patient population, due to the severity of their illness, would be unable to participate. Another limitation involves the lack of post-mortem histopathological diagnosis, which raises questions about the follow-up diagnosis’s robustness, especially in uncertain clinical cases. However, histopathological diagnosis is rarely performed, and selective inclusion of patients with a histopathological diagnosis would not be possible with the present material. In a post-mortem sample of 25 patients with MSA symptomatology included at our Neurology department before 2015, 22 showed MSA pathology post-mortem, 2 showed other pathology, and one was inconclusive (unpublished data). Further limitations include the access to previous imaging in the diagnostic reading of the [18F]FDG PET, which limits the assessment of the independent information by [18F]FDG PET. However, clinical [18F]FDG PET reading without the access to MRI or other structural information is not relevant according to practise guidelines [ref. 11], and DAT scans are shown to be unable to distinguish between parkinsonian syndromes [ref. 14] but is likely to have improved the discrimination between parkinsonian syndromes from healthy ageing. Thus, we chose to include previous imaging to reflect daily clinical practice. Additional readers would have strengthen the results and provided measures of interrater variation. A high specificity of [18F]FDG PET is believed to be of diagnostic, prognostic, and survival benefit, as is also seen in other patient groups [ref. 22]. There are currently ongoing clinical treatment trials for AP subtypes [ref. 23], for which an early diagnosis is essential for correct stratification of the patients, ensuring more reliable and robust outcomes. Despite these limitations, our findings reinforce the utility of cerebral [18F]FDG PET. Specific tracers for proteinopathies could potentially enhance sensitivity and specificity, as seen with amyloid imaging in Alzheimer’s disease. Promising tracers such as the tau tracer [18F]APN-1607 and the novel [18F]PI-2620 are under study and could greatly aid in early diagnosis [ref. 24, ref. 25]. Unfortunately, an α-synuclein PET tracer is still missing [ref. 26]. The mitochondrial translocator protein (TSPO) binding [11C]PBR28 shows potential as a neuroinflammation tracer. Recent studies suggest its use as an imaging biomarker for MSA with high sensitivity and specificity [ref. 27]. However, the necessity for genotyping for TSPO single-nucleotide polymorphism to exclude potential false negatives remains a hurdle [ref. 28]. Future research to examine the role of tau and neuroinflammation PET tracers in diagnosing early-stage AP is greatly encouraged.
Conclusion
This study found a moderate to high diagnostic accuracy for [18F]FDG PET imaging in the diagnosis of AP in a mixed population resembling clinical routine. The results support the additional use of [18F]FDG PET for the clinical diagnosis of AP with high specificity. [18F]FDG PET may be beneficial for prognosis and supportive treatment of the patients and useful for future clinical treatment trials ensuring correct stratification of patients.
References
- RA Walsh, T Lynch, S Fahn. Parkinson’s disease. Neurodegenerative disorders, 2011
- R Savica, BR Grossardt, JH Bower, BF Boeve, JE Ahlskog, WA Rocca. Incidence of dementia with Lewy bodies and Parkinson disease dementia. JAMA Neurol, 2013. [DOI | PubMed]
- CD Stang, P Turcano, MM Mielke, KA Josephs, JH Bower, JE Ahlskog. Progressive supranuclear palsy and corticobasal syndrome: a population-based study. J Parkinsons Dis, 2020. [DOI | PubMed]
- A Foubert-Samier, A Pavy-Le Traon, F Guillet, M Le-Goff, C Helmer, F Tison. Disease progression and prognostic factors in multiple system atrophy: a prospective cohort study. Neurobiol Dis, 2020. [DOI | PubMed]
- S Baudrexel, C Seifried, B Penndorf, JC Klein, M Middendorp, H Steinmetz. The value of putaminal diffusion imaging versus 18-fluorodeoxyglucose positron emission tomography for the differential diagnosis of the Parkinson variant of multiple system atrophy. Mov Disord, 2014. [DOI | PubMed]
- AG de Volder, J Francart, C Laterre, G Dooms, A Bol, C Michel. Decreased glucose utilization in the striatum and frontal lobe in probable striatonigral degeneration. Ann Neurol, 1989. [DOI | PubMed]
- J Blin, M-J Vidailhet, B Pillon, B Dubois, J-R Feve, Y Agid. Corticobasal degeneration: decreased and asymmetrical glucose consumption as studied with PET. Mov Disord, 1992. [DOI | PubMed]
- Z Walker, F Gandolfo, S Orini, V Garibotto, F Agosta, J Arbizu. Clinical utility of FDG PET in Parkinson’s disease and atypical parkinsonism associated with dementia. Eur J Nucl Med Mol Imaging, 2018. [DOI | PubMed]
- F Nobili, J Arbizu, F Bouwman, A Drzezga, F Agosta, P Nestor. European Association of Nuclear Medicine and European Academy of Neurology recommendations for the use of brain 18 F-fluorodeoxyglucose positron emission tomography in neurodegenerative cognitive impairment and dementia: Delphi consensus. Eur J Neurol, 2018. [DOI | PubMed]
- T Eckert, A Barnes, V Dhawan, S Frucht, MF Gordon, AS Feigin. FDG PET in the differential diagnosis of parkinsonian disorders. Neuroimage, 2005. [DOI | PubMed]
- CC Tang, KL Poston, T Eckert, A Feigin, S Frucht, M Gudesblatt. Differential diagnosis of parkinsonism: a metabolic imaging study using pattern analysis. Lancet Neurol, 2010. [DOI | PubMed]
- S Hellwig, F Amtage, A Kreft, R Buchert, OH Winz, W Vach. [18F]FDG-PET is superior to [123I]IBZM-SPECT for the differential diagnosis of parkinsonism. Neurology, 2012. [DOI | PubMed]
- GU Höglinger, G Respondek, M Stamelou, C Kurz, KA Josephs, AE Lang. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord, 2017. [DOI | PubMed]
- S Gilman, GK Wenning, PA Low, DJ Brooks, CJ Mathias, JQ Trojanowski. Second consensus statement on the diagnosis of multiple system atrophy. J Auton Nerv Syst, 2008
- IG McKeith, BF Boeve, DW Dickson, G Halliday, J-P Taylor. Diagnosis and management of dementia with Lewy bodies. Neurology, 2017. [DOI | PubMed]
- MJ Armstrong, I Litvan, AE Lang, TH Bak. Criteria for the diagnosis of corticobasal degeneration. Neurology, 2013. [DOI | PubMed]
- E Guedj, A Varrone, R Boellaard, NL Albert, H Barthel, B van Berckel. EANM procedure guidelines for brain PET imaging using [18F]FDG, version 3. Eur J Nucl Med Mol Imaging, 2022. [DOI | PubMed]
- LK Teune, AL Bartels, BM De Jong, ATM Willemsen, SA Eshuis, JJ De Vries. Typical cerebral metabolic patterns in neurodegenerative brain diseases. Mov Disord, 2010. [DOI | PubMed]
- MB Feany, LA Mattiace, DW Dickson. Neuropathologic overlap of progressive supranuclear palsy, Pick’s disease and corticobasal degeneration. J Neuropathol Exp Neurol, 1996. [DOI | PubMed]
- S Morbelli, G Esposito, J Arbizu, H Barthel, R Boellaard, NI Bohnen. EANM practice guideline/SNMMI procedure standard for dopaminergic imaging in Parkinsonian syndromes 1.0. Eur J Nucl Med Mol Imaging, 2020. [DOI | PubMed]
- R Murray, M Neumann, MS Forman, J Farmer, L Massimo, A Rice. Cognitive and motor assessment in autopsy-proven corticobasal degeneration. Neurology, 2007. [DOI | PubMed]
- T Mkhitarjan, A Areškevičiūtė, EL Lund, L Marner, AM Hejl. Sporadic fatal insomnia presenting with initial symptoms of parkinsonism and abnormal dopamine transporter imaging. Mov Disord Clin Pract, 2022. [DOI | PubMed]
- E Jabbari, N Holland, V Chelban, PS Jones, R Lamb, C Rawlinson. Diagnosis across the spectrum of progressive supranuclear palsy and corticobasal syndrome. JAMA Neurol, 2020. [DOI | PubMed]
- L Li, FT Liu, M Li, JY Lu, YM Sun, X Liang. Clinical utility of 18F-APN-1607 Tau PET imaging in patients with progressive supranuclear palsy. Mov Disord, 2021. [DOI | PubMed]
- M Song, L Beyer, L Kaiser, H Barthel, T van Eimeren, K Marek. Binding characteristics of [18F]PI-2620 distinguish the clinically predicted tau isoform in different tauopathies by PET. J Cereb Blood Flow Metab, 2021. [DOI | PubMed]
- Š Korat, NSR Bidesi, F Bonanno, A Di Nanni, ANN Hoàng, K Herfert. Alpha-synuclein PET tracer development-an overview about current efforts. Pharmaceuticals, 2021. [DOI | PubMed]
- A Jucaite, Z Cselényi, WC Kreisl, EA Rabiner, A Varrone, RE Carson. Glia imaging differentiates multiple system atrophy from Parkinson’s disease: a positron emission tomography study with [11C]PBR28 and machine learning analysis. Mov Disord, 2022. [DOI | PubMed]
- DR Owen, AJ Yeo, RN Gunn, K Song, G Wadsworth, A Lewis. An 18-kDa translocator protein (TSPO) polymorphism explains differences in binding affinity of the PET radioligand PBR28. J Cereb Blood Flow Metab, 2012. [DOI | PubMed]
