Association of preconception cannabis use frequency with cannabis use during early pregnancy
Abstract
Background:
Cannabis use during pregnancy is increasingly common and is associated with adverse health outcomes for pregnant individuals and their offspring. Identifying preconception factors that are associated with prenatal cannabis use is critical to inform early prevention and intervention. This study tested whether frequency of preconception cannabis use was associated with cannabis use during early pregnancy using data from a large healthcare system with universal screening for cannabis use.
Methods:
This observational study included pregnant individuals in Kaiser Permanente Northern California (KPNC) who self-reported any cannabis use (daily, weekly, or monthly or less) during the year before pregnancy from 2011 to 2022 (excluding 2020). Prenatal cannabis use was based on self-report and/or a positive toxicology test during the first trimester of pregnancy at entrance to prenatal care (at ~ 8 weeks gestation). Modified Poisson models were fit to assess associations between frequency of preconception cannabis use and prenatal cannabis use, adjusting for covariates.
Results:
The sample of 40,806 pregnancies from 36,622 unique individuals who self-reported any preconception cannabis use was 65.7% non-White; 27.6% were aged < 25 years. Nearly half (45.1%) screened positive for prenatal use, including 23.7% by self-report and 36.6% by positive toxicology test. Compared to monthly or less preconception cannabis use, daily use (adjusted prevalence ratio [aPR] = 2.66, 95% CI 2.59–2.73) or weekly use (aPR = 1.99, 95% CI 1.93–2.05) was associated with greater risk of prenatal cannabis use. Results were similar when prenatal cannabis use was based on self-report only or on toxicology testing only.
Conclusions:
Greater frequency of preconception cannabis use was associated with substantially increased risk of prenatal cannabis use. Findings reinforce the need for early harm prevention efforts focused on reducing the frequency of cannabis use among women of reproductive age, including screening, education, and early linkage to intervention.
Supplementary Information:
The online version contains supplementary material available at 10.1186/s12884-025-08190-y.
Article type: Research Article
Keywords: Cannabis, Marijuana, Pregnancy, Preconception, Frequency
Affiliations: https://ror.org/00t60zh31grid.280062.e0000 0000 9957 7758Division of Research, Kaiser Permanente Northern California, 4480 Hacienda Drive, Building B, Pleasanton, CA 94588 USA; https://ror.org/043mz5j54grid.266102.10000 0001 2297 6811Department of Psychiatry and Behavioral Sciences, University of California, San Francisco, CA USA; https://ror.org/0190ak572grid.137628.90000 0004 1936 8753Department of Population Health, New York University, New York, NY USA; https://ror.org/00t60zh31grid.280062.e0000 0000 9957 7758Regional Offices, Kaiser Permanente Northern California, Pleasanton, CA USA; https://ror.org/0027frf26grid.488833.c0000 0004 0615 7519Health Research Institute, Kaiser Permanente Washington, Seattle, WA USA; https://ror.org/00cvxb145grid.34477.330000 0001 2298 6657Department of Health Systems and Population Health, University of Washington, Seattle, WA USA
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s12884-025-08190-y | PMC: PMC12506406
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (952 KB)
Introduction
Cannabis use and cannabis use disorder are becoming increasingly prevalent among pregnant women in the US [ref. 1, ref. 2]. Prenatal cannabis use is associated with significant health risks for pregnant women and their offspring, including preeclampsia, gestational hypertension, placental abruption, preterm birth, small-for-gestational-age infant, and admission to the neonatal intensive care unit [ref. 3–ref. 10]. As the strength of cannabis products increases in the US [ref. 11], there are growing concerns about the health risks of prenatal cannabis use, and national medical organizations recommend abstinence from cannabis during pregnancy [ref. 10, ref. 12].
Understanding preconception factors that are associated with greater risk of using cannabis during pregnancy is critically important, as this knowledge could inform targeted prevention and intervention efforts before conception. Pregnant women often quit or cut down on their cannabis use over the course of pregnancy [ref. 13, ref. 14], and initial studies with smaller convenience samples suggest that women who use cannabis more frequently prior to pregnancy recognition are more likely to continue to use after pregnancy recognition [ref. 15]. While more frequent cannabis use before pregnancy may also be a risk factor for prenatal cannabis use, especially before pregnancy recognition [ref. 16], this relationship is understudied. If more frequent preconception cannabis use is strongly associated with prenatal use, efforts to implement screening for frequency of cannabis for reproductive aged women in women’s health settings more broadly may be warranted.
Using population-based data from a large health care system with universal screening for cannabis use both before and during pregnancy, we tested whether more frequent preconception cannabis use was associated with cannabis use during early pregnancy.
Methods
This cross-sectional observational study was conducted in Kaiser Permanente Northern California (KPNC) which universally screens for preconception and prenatal cannabis use at entry to prenatal care (at ~ 8 weeks gestation) through self-report and a urine toxicology test. The sample included pregnant individuals screened from 2011 to 2022 (excluding 2020 due to an electronic health record [EHR] system change that temporarily comprised data on self-reported prenatal substance use) who self-reported any cannabis use during the year before pregnancy (eFigure). This study followed STROBE guidelines and was approved by the KPNC IRB with a waiver of informed consent.
Preconception cannabis use was based on the frequency of self-reported cannabis use during the year before pregnancy, and prenatal cannabis use was based on any self-reported use since pregnancy at entry to prenatal care or a positive toxicology test during the first trimester (prenatal) (eMethods). Covariates were based on prior literature and availability in the electronic health record (EHR), including sociodemographic characteristics (age, race and ethnicity, neighborhood deprivation, Medicaid insurance coverage), parity, body mass index, and medical conditions diagnosed during the year before pregnancy onset, including psychiatric disorders (anxiety, depressive disorder and other), substance use disorders (cannabis use disorder, tobacco use disorder, and other alcohol and drug use disorders), sleep problems, common pain conditions (eMethods) and year of screen. Modified Poisson models with robust error variance were fit to assess associations between frequency of preconception cannabis use and prenatal cannabis use, adjusting for covariates and accounting for correlated observations of individuals with more than one pregnancy during the study period. In addition, we conducted two sets of sensitivity analyses. First, we refit the model excluding individuals who received a toxicology test before 30 days of gestation to minimize detection of cannabis use that occurred only during preconception, as heavy cannabis use may be detectable for up to 30 days. Second, we fit models with and without adjusting for cannabis use disorder to assess the impact of potential collinearity. Two-sided P values < 0.05 were considered statistically significant.
Results
Among 40,806 pregnancies from 36,622 unique individuals who self-reported any preconception cannabis use (23.4% daily, 21.0% weekly, 55.6% monthly or less), 65.7% were non-White and 27.6% were aged < 25 years. The self-reported prenatal cannabis questionnaire and toxicology testing occurred at a median (IQR) of 51.0 (40.0–61.0) days gestation and 58.0 (51.0–71.0) days gestation, respectively. Nearly half (45.1%) screened positive for prenatal use, including 23.7% by self-report and 36.6% by toxicology testing (Table 1). Greater frequency of preconception cannabis use was associated with younger age, non-White race, Medicaid insurance, obesity, and psychiatric, substance use and common pain conditions.
Table 1: Patient characteristics among pregnant individuals who self-reported preconception cannabis use, 2011–2019, 2021–2022 (N = 40,806)
| Frequency of preconception cannabis use: | ||||||||
|---|---|---|---|---|---|---|---|---|
| All | Monthly(N = 22,683, 55.6%) | Weekly(N = 8,570, 21.0%) | Daily(N = 9,553, 23.4%) | |||||
| N | % | N | % | N | % | N | % | |
| Age group | ||||||||
| 14–17 | 757 | 1.9 | 356 | 1.6 | 189 | 2.2 | 212 | 2.2 |
| 18–24 | 10,472 | 25.7 | 4,691 | 20.7 | 2,333 | 27.2 | 3,448 | 36.1 |
| 25–34 | 22,876 | 56.1 | 13,355 | 58.9 | 4,659 | 54.4 | 4,862 | 50.9 |
| 35+ | 6,701 | 16.4 | 4,281 | 18.9 | 1,389 | 16.2 | 1,031 | 10.8 |
| Race and ethnicity | ||||||||
| Asian | 4,099 | 10.1 | 2,884 | 12.7 | 662 | 7.7 | 553 | 5.8 |
| Black | 5,723 | 14.0 | 2,446 | 10.8 | 1,239 | 14.5 | 2,038 | 21.3 |
| Hispanic | 10,775 | 26.4 | 5,861 | 25.8 | 2,213 | 25.8 | 2,701 | 28.3 |
| White | 18,063 | 44.3 | 10,347 | 45.6 | 3,995 | 46.6 | 3,721 | 39.0 |
| Other/Unknown | 2,146 | 5.3 | 1,145 | 5.1 | 461 | 5.4 | 540 | 5.7 |
| NDI quartile | ||||||||
| 1st | 6,923 | 17.0 | 4,082 | 18.0 | 1,435 | 16.7 | 1,406 | 14.7 |
| 2nd | 7,654 | 18.8 | 4,323 | 19.1 | 1,587 | 18.5 | 1,744 | 18.3 |
| 3rd | 10,991 | 26.9 | 6,180 | 27.3 | 2,299 | 26.8 | 2,512 | 26.3 |
| 4th | 15,238 | 37.3 | 8,098 | 35.7 | 3,249 | 37.9 | 3,891 | 40.7 |
| Parity | ||||||||
| 0 | 23,805 | 58.3 | 13,311 | 58.7 | 4,967 | 58.0 | 5,527 | 57.9 |
| 1 | 9,583 | 23.5 | 5,450 | 24.0 | 2,048 | 23.9 | 2,085 | 21.8 |
| 2+ | 4,654 | 11.4 | 2,530 | 11.2 | 962 | 11.2 | 1,162 | 12.2 |
| Unknown | 2,764 | 6.8 | 1,392 | 6.1 | 593 | 6.9 | 779 | 8.2 |
| Body mass index | ||||||||
| Underweight | 822 | 2.0 | 369 | 1.6 | 173 | 2.0 | 280 | 2.9 |
| Normal | 16,370 | 40.1 | 9,420 | 41.5 | 3,432 | 40.1 | 3,518 | 36.8 |
| Overweight | 11,222 | 27.5 | 6,357 | 28.0 | 2,331 | 27.2 | 2,534 | 26.5 |
| Obese | 12,223 | 30.0 | 6,440 | 28.4 | 2,593 | 30.3 | 3,190 | 33.4 |
| Unknown | 169 | 0.4 | 97 | 0.4 | 41 | 0.5 | 31 | 0.3 |
| Medicaid | 6,112 | 15.0 | 2,614 | 11.5 | 1,378 | 16.1 | 2,120 | 22.2 |
| Psychiatric diagnosis | ||||||||
| Anxiety or depressive disorder | 8480 | 20.8 | 4,265 | 18.8 | 1,877 | 21.9 | 2,338 | 24.5 |
| Other psychiatric disorders | 2,195 | 5.4 | 968 | 4.3 | 522 | 6.1 | 705 | 7.4 |
| SUD diagnosis | ||||||||
| Cannabis use disorder | 1,072 | 2.6 | 276 | 1.2 | 257 | 3.0 | 539 | 5.6 |
| Tobacco use disorder | 1,339 | 3.3 | 554 | 2.4 | 311 | 3.6 | 474 | 5.0 |
| Other alcohol or drug use disorders | 903 | 2.2 | 379 | 1.7 | 222 | 2.6 | 302 | 3.2 |
| Sleep problems | 997 | 2.4 | 552 | 2.4 | 218 | 2.5 | 227 | 2.4 |
| Common pain conditions | 18,090 | 44.3 | 9,647 | 42.5 | 3,810 | 44.5 | 4,633 | 48.5 |
| Prenatal cannabis use | ||||||||
| Self-report or urine toxicology test | 18,387 | 45.1 | 6001 | 26.5 | 4784 | 55.8 | 7602 | 79.6 |
| Self-report | 9,673 | 23.7 | 2855 | 12.6 | 2559 | 29.9 | 4259 | 44.6 |
| Urine toxicology test | 14,953 | 36.6 | 4110 | 18.1 | 3853 | 45.0 | 6990 | 73.2 |
Cannabis use in the year before pregnancy (preconception) was based on self-report, and cannabis use in the first trimester of pregnancy (prenatal) was based on positive self-report or positive urine toxicology test; all toxicology tests were confirmed with a confirmatory laboratory test. First trimester is defined as 90 days from last menstrual period. The 1 st NDI quartile indicates the lowest NDI (highest socioeconomic status), and the 4th quartile indicating the highest NDI (lowest socioeconomic status). Other psychiatric diagnoses include attention-deficit/hyperactivity disorder, bipolar disorder, personality disorder, post-traumatic stress disorder and psychotic disorder
NDI Neighborhood deprivation index, SUD Substance use disorder
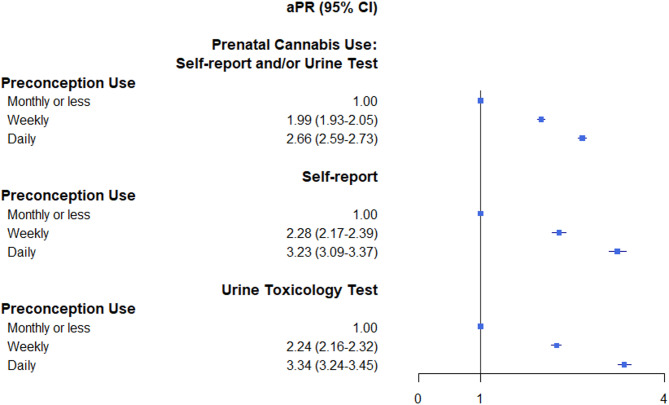
Adjusted models found a dose-response relationship between frequency of preconception use and odds of prenatal cannabis use. Compared to individuals who reported monthly or less preconception cannabis use, the risk of any prenatal cannabis use were greater among those who self-reported daily (adjusted prevalence ratio [aPR] = 2.66, 95% CI 2.59–2.73) or weekly preconception use (aPR = 1.99, 95% CI 1.93–2.05) (Fig. 1). Similar results were observed when prenatal cannabis use by self-report (aPR = 3.23, 95% CI 3.09–3.37 for daily and aPR = 2.28, 95% CI 2.17–2.39 for weekly vs. monthly or less, respectively) and toxicology tests (aPR = 3.34, 95% CI 3.24–3.45 for daily and aPR = 2.24, 95% CI 2.16–2.32 for weekly vs. monthly or less) were examined separately. Results from sensitivity analyses that excluded those whose toxicology tests were conducted prior to 30 days gestation (~ 1%) found similar results, with greater odds of prenatal cannabis use via toxicology testing among those who self-reported daily (aPR = 3.35, 95% CI 3.24–3.46) or weekly preconception use (aPR = 2.24, 95% CI 2.16–2.32) (eTable 1). In addition, results from models with and without adjusting for cannabis use disorder were almost identical, suggesting minimal concern of collinearity (eTable 2).
Discussion
Given the increasing prevalence of cannabis use among pregnant and reproductive-aged women and the expanding legalization of cannabis across the US, it is critical to understand preconception factors associated with prenatal cannabis use to inform early prevention and intervention. In this large study of > 40,000 pregnancies with self-reported preconception cannabis use, nearly half continued use during early pregnancy, with 80% of individuals who reported daily preconception use continuing to use into early pregnancy. These estimates of continued use during pregnancy are higher than those found by other studies [ref. 17, ref. 18], likely because other research has typically examined cannabis use after pregnancy recognition and relied solely on self-reported prenatal cannabis use, which can underestimate the true prevalence [ref. 19].
There was a clear dose-response relationship between frequency of preconception cannabis use and greater odds of prenatal cannabis use, with those who used daily before pregnancy having about three times or higher risk of cannabis use during early pregnancy depending on prenatal cannabis screening method. Importantly, while results were strongest for prenatal cannabis use based on urine toxicology testing, findings were consistent across all measurement approaches (self-report, urine toxicology testing, or either). All models adjusted for key sociodemographic and behavioral covariates, highlighting preconception cannabis use as an independently important potential risk factor for prenatal cannabis use.
Our screening for prenatal cannabis use did not differentiate whether prenatal use only occurred before pregnancy recognition versus continued after. Women who use cannabis daily prior to conception may have greater odds of cannabis use during early pregnancy due to ongoing use prior to recognition of pregnancy, potentially due to the challenges of reducing or stopping frequent use [ref. 15, ref. 20]. While quitting cannabis use at any point during pregnancy is beneficial, even cannabis use that is limited only to early pregnancy, particularly during critical periods of fetal development, may carry risks [ref. 21]. More frequent preconception cannabis use could also indicate more severe addiction and vulnerability to cannabis use even after pregnancy recognition, especially for those who rely on cannabis to manage mental health and medical symptoms [ref. 22–ref. 26]. Women with more frequent preconception cannabis use are also more likely to use multiple modes of cannabis administration (e.g., smoking, edibles, vaping) [ref. 27], which could further influence the risk of continued use during pregnancy. Future research is needed to better understand how different aspects of preconception cannabis use, including mode, product strength, and reasons for use, influence risk and duration of prenatal cannabis use.
Research indicates that most pregnant women who use cannabis also used it before pregnancy [ref. 28], and our results demonstrate that women who use more frequently (i.e., weekly, daily) during the preconception period are far more likely to continue cannabis use into the prenatal period. These findings point to the value of preconception screening for identifying women who may benefit from prevention and cessation efforts even prior to pregnancy [ref. 29]. However, despite recommendations for routine cannabis screening for adults in medical settings [ref. 30] and the availability of valid, brief screens for the frequency of cannabis use [ref. 31], preconception cannabis screening is not common. Efforts to integrate screening and clinician education to better support women during the preconception period are essential to reducing prenatal cannabis use [ref. 29].
Notably, those using cannabis on a daily or near daily basis may have a cannabis use disorder. Screening for preconception use frequency provides an opportunity for clinicians who recognize a possible use disorder to refer patients for further evaluation in specialty addiction medicine treatment. Some patients will have a harder time with the withdrawal symptoms and nausea that is often experienced after stopping cannabis because of the coexisting nausea and vomiting of early pregnancy, therefore making ongoing cessation efforts less successful. While treatment in pregnancy is encouraged, treatment prior to pregnancy is ideal for harm prevention for both mother and child.
Finally, our study indicated that those with younger age, non-White race, Medicaid insurance, obesity, and psychiatric, substance use and common pain conditions had a greater frequency of preconception cannabis use. Public health messaging and education strategies that prioritize these at-risk groups may be most beneficial.
Strengths and limitations
Our study has important strengths, including a large, diverse sample of pregnant individuals universally screened for preconception and prenatal cannabis use. Our measure of prenatal cannabis use was based on self-report and urine toxicology testing, minimizing misclassification of our outcome. All analyses adjusted for key sociodemographic and health-related covariates available in the EHR, increasing the validity of our findings. The sample was large enough to examine differences by type of cannabis screening.
Our study also has several limitations. The sample comprised pregnant individuals in a large healthcare system in Northern California who screened positive for preconception cannabis use during standard prenatal care, potentially limiting generalizability to uninsured individuals, those who do not come in for prenatal care, or those outside of California. Preconception cannabis use was based on self-report at the time of prenatal screening which could underestimate use due to stigma or recall bias, and findings may not generalize to individuals who choose not to disclose their use. Further, we did not have information on preconception cannabis product strength or mode of administration. Future studies should examine these factors to better understand risk factors for cannabis use during pregnancy.
While our study uniquely assessed any cannabis use during early pregnancy regardless of pregnancy recognition, urine toxicology tests may have detected residual frequent preconception cannabis use. However, this is unlikely to explain the observed associations given that urine toxicology tests typically detect cannabis use for up to 30 days in heavy users, and the median gestational age of urine toxicology testing was 58.0 days (IQR: 51.0–71.0). Further, sensitivity analyses that excluded those with toxicology tests before 30 days gestation found comparable results, suggesting that results are not driven by misclassification of prenatal cannabis use. Finally, this study was conducted among pregnant individuals with preconception cannabis use, and the sample size was not large enough to fit multivariable models examining frequency of prenatal cannabis use as the outcome.
Conclusions
In this large study of pregnant women with preconception cannabis use, nearly half screened positive for cannabis use during early pregnancy. Higher frequency of preconception use was strongly associated with increased odds of prenatal cannabis use. Findings highlight the potential importance of screening for and addressing frequent cannabis use among women of reproductive age even prior to pregnancy. Routine screening, patient resources, and early referrals during early pregnancy may help to reduce prenatal cannabis use and improve maternal and child health. Additional research is needed to examine how specific aspects of preconception cannabis use, include mode of administration, product strength, and reasons for use, influence risk of prenatal cannabis use to inform more tailored interventions.
Supplementary Materials
References
- ND Volkow, B Han, WM Compton, EF McCance-Katz. Self-reported medical and nonmedical cannabis use among pregnant women in the United States. JAMA, 2019. [DOI | PubMed]
- A Meinhofer, JM Hinde, KM Keyes, C Lugo-Candelas. Association of comorbid behavioral and medical conditions with cannabis use disorder in pregnancy. JAMA Psychiatr, 2022. [DOI]
- KC Young-Wolff, SR Adams, SE Alexeeff, Y Zhu, E Chojolan, NE Slama. Prenatal cannabis use and maternal pregnancy outcomes. JAMA Intern Med, 2024. [DOI | PubMed]
- I Baia, RMSM Domingues. The effects of cannabis use during pregnancy on low birth weight and preterm birth: a systematic review and meta-analysis. Am J Perinatol, 2022. [DOI | PubMed]
- SN Conner, V Bedell, K Lipsey, GA Macones, AG Cahill, MG Tuuli. Maternal marijuana use and adverse neonatal outcomes: a systematic review and meta-analysis. Obstet Gynecol, 2016. [DOI | PubMed]
- JK Gunn, CB Rosales, KE Center, A Nunez, SJ Gibson, C Christ. Prenatal exposure to cannabis and maternal and child health outcomes: a systematic review and meta-analysis. BMJ Open, 2016. [DOI | PubMed]
- JO Lo, B Shaw, S Robalino, CK Ayers, S Durbin, MC Rushkin. Cannabis use in pregnancy and neonatal outcomes: a systematic review and meta-analysis. Cannabis Cannabinoid Res, 2023. [DOI | PubMed]
- G Marchand, AT Masoud, M Govindan, K Ware, A King, S Ruther. Birth outcomes of neonates exposed to marijuana in utero: a systematic review and meta-analysis. JAMA Netw Open, 2022. [DOI | PubMed]
- KS Grant, E Conover, CD Chambers. Update on the developmental consequences of cannabis use during pregnancy and lactation. Birth Defects Res, 2020. [DOI | PubMed]
- SA Ryan, SD Ammerman, ME O’Connor. Marijuana use during pregnancy and breastfeeding: implications for neonatal and childhood outcomes. Pediatrics, 2018. [DOI | PubMed]
- MA ElSohly, CG Majumdar, S Chandra, MM Radwan. A 10-year trend in cannabis potency (2013–2022) in different geographical regions of the United States of America. Front Public Health, 2024. [DOI | PubMed]
- A Braillon, S Bewley. Committee opinion 722: marijuana use during pregnancy and lactation. Obstet Gynecol, 2018. [DOI | PubMed]
- JY Ko, SL Farr, VT Tong, AA Creanga, WM Callaghan. Prevalence and patterns of marijuana use among pregnant and nonpregnant women of reproductive age. Am J Obstet Gynecol, 2015. [DOI]
- S Singh, KB Filion, HA Abenhaim, MJ Eisenberg. Prevalence and outcomes of prenatal recreational cannabis use in high-income countries: a scoping review. BJOG, 2020. [DOI | PubMed]
- CK Pike, MJ Sofis, AJ Budney. Correlates of continued cannabis use during pregnancy. Drug Alcohol Depend, 2021. [DOI | PubMed]
- KC Thomson, CJ Greenwood, P Letcher, EA Spry, JA Macdonald, HM McAnally. Continuities in maternal substance use from early adolescence to parenthood: findings from the intergenerational cohort consortium. Psychol Med, 2023. [DOI | PubMed]
- AM Allen, AM Jung, AC Alexander, SS Allen, KD Ward, M al’Absi. Cannabis use and stressful life events during the perinatal period: cross-sectional results from pregnancy risk assessment monitoring system (PRAMS) data, 2016. Addiction, 2020. [DOI | PubMed]
- JY Ko, KC Coy, SC Haight, TM Haegerich, L Williams, S Cox. Characteristics of marijuana use during pregnancy – eight states, pregnancy risk assessment monitoring system, 2017. MMWR Morb Mortal Wkly Rep, 2020. [DOI | PubMed]
- KC Young-Wolff, V Sarovar, LY Tucker, N Goler, A Conway, C Weisner. Validity of self-reported cannabis use among pregnant females in Northern California. J Addict Med, 2020. [DOI | PubMed]
- JR Hughes, S Naud, AJ Budney, JR Fingar, PW Callas. Attempts to stop or reduce daily cannabis use: an intensive natural history study. Psychol Addict Behav, 2016. [DOI | PubMed]
- ND Volkow, WM Compton, EM Wargo. The risks of marijuana use during pregnancy. JAMA, 2017. [DOI | PubMed]
- LB McKenzie, SA Keim, MA Klebanoff. Risk perceptions about cannabis use and receipt of health-related information during pregnancy. Am J Health Promot, 2022. [DOI]
- JC Chang, JA Tarr, CL Holland, NM De Genna, GA Richardson, KL Rodriguez. Beliefs and attitudes regarding prenatal marijuana use: perspectives of pregnant women who report use. Drug Alcohol Depend, 2019. [DOI | PubMed]
- KR Skelton, AA Hecht, SE Benjamin-Neelon. Women’s cannabis use before, during, and after pregnancy in New Hampshire. Prev Med Rep, 2020. [DOI | PubMed]
- M Vanstone, S Taneja, A Popoola, J Panday, D Greyson, R Lennox. Reasons for cannabis use during pregnancy and lactation: a qualitative study. CMAJ, 2021. [DOI | PubMed]
- C Barbosa-Leiker, E Burduli, CL Smith, O Brooks, M Orr, M Gartstein. Daily cannabis use during pregnancy and postpartum in a state with legalized recreational cannabis. J Addict Med, 2020. [DOI | PubMed]
- KC Young-Wolff, CA Cortez, JR Nugent, AA Padon, JJ Prochaska, SR Adams. Sociodemographic differences in modes of cannabis use among pregnant individuals in Northern California. Drug Alcohol Depend, 2025. [DOI | PubMed]
- KC Young-Wolff, V Sarovar, LY Tucker, A Conway, S Alexeeff, C Weisner. Self-reported daily, weekly, and monthly cannabis use among women before and during pregnancy. JAMA Netw Open, 2019. [DOI | PubMed]
- KR Skelton, KC Young-Wolff. Preconception cannabis use: an important but overlooked public health issue. Womens Health, 2022. [DOI]
- CD Patnode, LA Perdue, M Rushkin, EA O’Connor. Screening for unhealthy drug use in primary care in adolescents and adults, including pregnant persons: updated systematic review for the U.S. Preventive services task force, 2020
- TE Matson, GT Lapham, JF Bobb, M Oliver, KA Hallgren, EC Williams. Validity of the single-item screen-cannabis (SIS-C) for cannabis use disorder screening in routine care. JAMA Netw Open, 2022. [DOI | PubMed]
