Landscape of gene mutation in Chinese thyroid cancer patients: Construction and validation of lymph node metastasis prediction model based on clinical features and gene mutation marker
Abstract
Objectives:
Reporting the clinicopathological information of thyroid cancer (TC) patients from a central medical center of east China, and constructing the nomogram predicting lymph node metastasis (LNM).
Methods:
We collected the patients who underwent thyroid cancer surgery in our institute from July 1, 2019 to July 31, 2021, a total of 253 subjects were enrolled. We used HiPure FFPE DNA Kit to extract DNA and RNApure FFPE Kit to extract RNA from the paraffin sections of tumor tissue, the extracted DNA samples and RNA samples were used for NGS sequencing. The clinical and pathological information of TCGA‐THCA cohort was obtained as the validation cohort. Multivariate logistic regression analysis was performed to identify the independent prognostic factor, and the nomogram was subsequently constructed by “rms” R package.
Results:
Secondary cases contained more mutation of BRAF (90.48% vs. 62.07%) and TERT (33.0% vs. 3.0%), as compared with primary cases. Primary patients with positive lymph node were younger (40.9 ± 10.8 vs. 45.3 ± 11.8, p = 0.0031) and contained advanced TI‐RADS levels (4c: 22.8% vs. 8.3%, 5: 6.5% vs. 0/0%, p = 1.878e‐03), as well as more RET genetic alteration (16.3% vs. 2.7%, p = 2.566e‐03). We chose age, tumor diameter, RET fusion, and gender to construct the LNM predicting nomogram. Calibration plot, DCA curve, and the clinical impact plot verified the preferable prognostic value of the nomogram, with an AUC value of 0.724 (0.656–0.792). We successfully validated the prognostic value of the nomogram in TCGA‐THCA cohort. RET fusion might impact the process of protein digestion and absorption, cytokine‐cytokine receptor interaction, ECM‐receptor interaction, focal adhesion.
Conclusion:
We provide a novel nomogram to predict the LNM for TC patients, including the features of patient’s age, gender, tumor diameter, and RET alteration. Further studies from multiple medical centers are essential to validate the nomogram.
Article type: Research Article
Keywords: lymph node metastasis, nomogram, RET genetic alteration, thyroid cancer
Affiliations: Department of General Surgery The First Affiliated Hospital of Anhui Medical University Hefei China
License: © 2023 The Authors. Cancer Medicine published by John Wiley & Sons Ltd. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cam4.5945 | PubMed: 37081757 | PMC: PMC10278465
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (4.0 MB)
INTRODUCTION
The incidence of thyroid cancer (TC), ranking ninth place for global incidence, has been rapidly ascending over the last few decades in most areas of the world.ref. cam45945-bib-0001, ref. cam45945-bib-0002 Specifically, in China, from 2008 to 2012, about 47,550 new TC cases were diagnosed. The incidence rate of TC, reaching 7.56/100,000, has ranked the seventh in overall malignant tumors.ref. cam45945-bib-0003 From 2005 to 2015, the incidence of TC showed upward trend, increasing from 3.21/100,000 in 2005 to 9.61/100,000 in 2015.ref. cam45945-bib-0004 Differentiated thyroid cancer (DTC) is the most common subtype of TC,ref. cam45945-bib-0005 which includes papillary thyroid cancer (PTC) and follicular thyroid cancer (FTC), accounts for more than 90% of thyroid malignancies in the world.ref. cam45945-bib-0006 Surgery followed by radioactive iodine therapy or chemoradiation are both effective treatments and targeted treatment of the cancer for example BRAF inhibitors MEK inhibitors has also been a prospective method to cure of TC.ref. cam45945-bib-0005 Although majority of patients have favorable prognoses, with the 5‐year‐survival rate of about 98.2% in PTC patients (PTCP) around the world and 84.3% in TC patients in China,ref. cam45945-bib-0007, ref. cam45945-bib-0008, ref. cam45945-bib-0009, ref. cam45945-bib-0010 lymph node metastasis (LNM) is still a risk factor which contributes to the poor prognosis of TC patients.ref. cam45945-bib-0011 Therefore, we aim to construct new models to predict LNM to optimize treatment of patients.
The detection rate of lymph node metastasis (LNM) is frequent with 13.4%–60% in PTCP.ref. cam45945-bib-0008 The study of Nie et al.ref. cam45945-bib-0012 also found that the rate of lateral LNM and central LNM were 74.9% and 70.5%, respectively, in PTC patients in China. Until now, univariate and multivariate analyses have both revealed LNM played a significant role in predicting disease recurrence.ref. cam45945-bib-0011 Presence of LNM can cause five times more likely to relapse than the absence of LNM.ref. cam45945-bib-0011, ref. cam45945-bib-0013 The study of Lee et al.ref. cam45945-bib-0014 found that the deaths can be up to 16 patients, who died of distant metastasis or locoregional failure, among 18 patients with recurrence, nearly 89%.ref. cam45945-bib-0014 Therefore, we could consider LNM as a poor prognostic factor. Nowadays, for more accurate prognosis, many scientists have focused on some molecular marks. BRAFV600E mutation and RET mutation are greatly common, with frequency ranging from 33.2% to 88% in PTC and 40% to 50% in patients with sporadic medullary TC, respectively.ref. cam45945-bib-0008, ref. cam45945-bib-0013 BRAFV600E mutation can lead to aberrant activation of the mitogen‐activated protein kinase, which will increase cell proliferation and differentiation, eventually causing the thyroid oncogenesis.ref. cam45945-bib-0008, ref. cam45945-bib-0015, ref. cam45945-bib-0016, ref. cam45945-bib-0017 Moreover, BRAFV600E mutation is correlated with the poor survival rate of PTCP and some poor clinicopathologic features, such as multifocality, tumor size, vascular invasion,ref. cam45945-bib-0015, ref. cam45945-bib-0017 revealing it can be an important candidate biomarker for the prognosis of TC patients. RET mutation can cause tumor cell survival and proliferation, induce c‐cell transformation, finally exerting their oncogenic effects on the thyroid.ref. cam45945-bib-0013 Until now, many types of RET/PTC rearrangements have also been studied. RET/PTC1 is associated with excellent prognosis while RET/PTC3 is related with poor prognosis.ref. cam45945-bib-0018 Considering LNM and some molecular mutations are both associated with the prognosis of patients, so it is of interest to study the relation between them.
In this study, we gathered the clinicopathological information of TC patients from a central medical center of east China and identified the predict factors that can reflect the presence of LNM and finally constructed a nomogram that can predict LNM for TC patients.
METHODS AND MATERIALS
Collection of patients
We collected the patients who underwent thyroid cancer surgery in our institute from July 1, 2019 to July 31, 2021, a total of 253 subjects were enrolled. All the patients were diagnosed with unilateral or bilateral thyroid nodules of category 3 or higher by ultrasound, CT or MRI. The samples were further double checked with TC by intraoperative frozen section examination and postoperative pathological examination. We recorded the clinicopathological parameters of age, gender, tumor diameter, ultrasound TI‐RADS stage, with or without ultrasound abnormal node, with or without ultrasound abnormal calcification, tumor pathological type, tumor TNM stage, and with or without lymph node metastasis. The TNM staging was based on the staging definition criteria in the eighth edition of the American Joint Committee on Cancer (AJCC).
Inclusion and exclusion criteria
All the selected patients should meet the following criteria: (1) did not receive radionuclide iodine‐131 therapy or radiofrequency ablation before surgery; (2) between 18 and 75 years old; (3) without chronic diseases of hypertension, heart disease, diabetes, etc.; (4) cooperate with the follow‐up. Exclusion criteria: (1) without certain pathological diagnosis of TC; (2) with severe dysfunction of liver, kidney, heart, and lung; (3) not agree to receive NGS testing; (4) others who are considered inappropriate by the researchers. This study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University, and written informed consent was obtained from the participants and the data involved were anonymous without any identifiable private information.
Sequencing of tumor tissue
We used HiPure FFPE DNA Kit to extract DNA and RNApure FFPE Kit to extract RNA from the paraffin sections of tumor tissue, the extracted DNA samples and RNA samples were used for library construction, and then the Qsep100 automatic nucleic acid and protein analysis system was used to assess the quality of the constructed library. For NGS sequencing, the steps are as follows: (1) thawing the sequencing reagent; (2) cleaning the instrument before sequencing; (3) cleaning the Flowcell; (4) establishing a Sample Sheet; (5) dilution and mixing of the library; (6) adding the library; (7) run sequencing; (8) clean the instrument after sequencing. SNV and Indel mutations were filtered with the follow criteria: (1) mutation frequency > 0.01; (2) population frequency (refer to the East Asian population database of 1000G and ExAC) less than 0.01; (3) retain the sites of exons or alternatively spliced regions, and retain the sites of the target UTR region (such as the TERT promoter region); (4) remove the sites of synonymous mutation; (5) remove background mutations and low‐frequency mutations in repetitive regions.
Bioinformatic analysis
The clinical and pathological information of TCGA‐THCA cohort was obtained from UCSC Xena, as well as the gene expression profile and gene mutation matrix (https://xenabrowser.net/datapages/?cohort=GDC%20TCGA%20Thyroid%20Cancer%20(THCA)&removeHub=https%3A%2F%2Fxena.treehouse.gi.ucsc.edu%3A443). The heatmap for the clinical information and waterfall plot for the distribution of gene mutation was completed with “ComplexHeatmap” R package.ref. cam45945-bib-0019 Multivariate logistic regression analysis was performed to identify the independent prognostic factor, and the nomogram was subsequently constructed by “rms” R package. Calibration curve, decision curve and clinical impact curve analysis were all performed to assess the clinical predict function and accuracy of the nomogram via the “rms” and “rmda” packages. Differentially expressed genes (DEGs) was calculated by “limma” R package, and “clusterProfiler” packageref. cam45945-bib-0020 was employed to annotate the enriched biological process, cell components and molecular function of GO terms, as well as the KEGG pathways.
Statistics
Student’s t test was used to compare the distribution between two groups, and Fisher’s exact test was performed to distinguish the difference of categorical data. All statistical analyses were performed by R (Version: 4.1.2). A two‐tailed p value <0.05 was recognized statistically significant.
RESULTS
Basic information and differences between primary and secondary cases of AHMU‐TC cohort
With the pre‐set criteria, we collected a total of 253 TC patients from our institute, consist of 232 primary cases and 21 recurrent cases (also termed as secondary cases). The basic clinical pathological information of all the patients listed in Table 1 and Figure 1A. We first compared the different clinicopathological information between primary and secondary cases. We observed that the average age for secondary cases was higher than primary cases (51.5 ± 12.2 vs. 43 ± 11.4), and the tumor longest diameter in secondary group also showed a higher value (1.38 ± 1.2 vs. 0.958 ± 0.746). In addition, secondary group contained less ultrasound detected calcification (14.3% vs. 47.8%), but more lymph node metastasis (76.2% vs. 53.0%). We demonstrated the genetic alteration for all the 253 TC patients with the waterfall plot (Figure 1B), and several differences of genetic alteration was also displayed. Significantly, more secondary cases contained at least one mutant gene as compared with primary cases (90.5% vs. 88.0%, p = 5.048e‐06), and only one patient contained three types of mutant gene, BRAF, TERT, and PIK3CA, belonged to secondary group (Figure 1C). In addition, secondary cases contained more mutation of BRAF (90.48% vs. 62.07%) and TERT (33.0% vs. 3.0%, Figure 1C).
TABLE 1: Basic clinical features of enrolled patients.
| Primary (N = 232) | Secondary (N = 21) | Overall (N = 253) | |
|---|---|---|---|
| Gender | |||
| Female | 163 (70.3%) | 13 (61.9%) | 176 (69.6%) |
| Male | 69 (29.7%) | 8 (38.1%) | 77 (30.4%) |
| Age | |||
| Mean (SD) | 43.0 (11.4) | 51.5 (12.2) | 43.7 (11.7) |
| Median [Min, Max] | 43.0 [21.0, 71.0] | 54.0 [30.0, 74.0] | 44.0 [21.0, 74.0] |
| TI‐RADS | |||
| 3 | 4 (1.7%) | 0 (0%) | 4 (1.6%) |
| 4a | 46 (19.8%) | 4 (19.0%) | 50 (19.8%) |
| 4b | 129 (55.7%) | 0 (0%) | 128 (50.6%) |
| 4c | 37 (15.9%) | 1 (4.8%) | 38 (15.0%) |
| 5 | 8 (3.4%) | 1 (4.8%) | 9 (3.6%) |
| Missing | 8 (3.4%) | 15 (71.4%) | 23 (9.1%) |
| Ultrasound abnormal node | |||
| No | 61 (26.3%) | 3 (14.3%) | 64 (25.3%) |
| Yes | 171 (73.7%) | 18 (85.7%) | 189 (74.7%) |
| Ultrasound calcification | |||
| No | 121 (52.2%) | 18 (85.7%) | 139 (54.9%) |
| Yes | 111 (47.8%) | 3 (14.3%) | 114 (45.1%) |
| Histology | |||
| MTC | 4 (1.7%) | 0 (0%) | 4 (1.6%) |
| PTC | 228 (98.3%) | 21 (100%) | 249 (98.4%) |
| Lymph node | |||
| Negative | 109 (47.0%) | 5 (23.8%) | 114 (45.1%) |
| Positive | 123 (53.0%) | 16 (76.2%) | 139 (54.9%) |
| Diameter | |||
| Mean (SD) | 0.958 (0.746) | 1.38 (1.20) | 0.982 (0.782) |
| Median [Min, Max] | 0.800 [0.200, 5.50] | 1.10 [0.100, 4.50] | 0.800 [0.100, 5.50] |
| Missing | 17 (7.3%) | 8 (38.1%) | 25 (9.9%) |
| Stage | |||
| I | 215 (92.7%) | 4 (19.0%) | 219 (86.6%) |
| II | 14 (6.0%) | 11 (52.4%) | 25 (9.9%) |
| IVA | 3 (1.3%) | 2 (9.5%) | 5 (2.0%) |
| IVB | 0 (0%) | 4 (19.0%) | 4 (1.6%) |
| T stage | |||
| T1a | 179 (77.2%) | 6 (28.6%) | 185 (73.1%) |
| T1b | 39 (16.8%) | 4 (19.0%) | 43 (17.0%) |
| T2 | 13 (5.6%) | 3 (14.3%) | 16 (6.3%) |
| T3a | 1 (0.4%) | 1 (4.8%) | 2 (0.8%) |
| Tx | 0 (0%) | 7 (33.3%) | 7 (2.8%) |
| N stage | |||
| N0 | 110 (47.4%) | 4 (19.0%) | 114 (45.1%) |
| N1a | 74 (31.9%) | 0 (0%) | 74 (29.2%) |
| N1b | 48 (20.7%) | 17 (81.0%) | 65 (25.7%) |
| M stage | |||
| M0 | 232 (100%) | 5 (23.8%) | 237 (93.7%) |
| M1 | 0 (0%) | 16 (76.2%) | 16 (6.3%) |
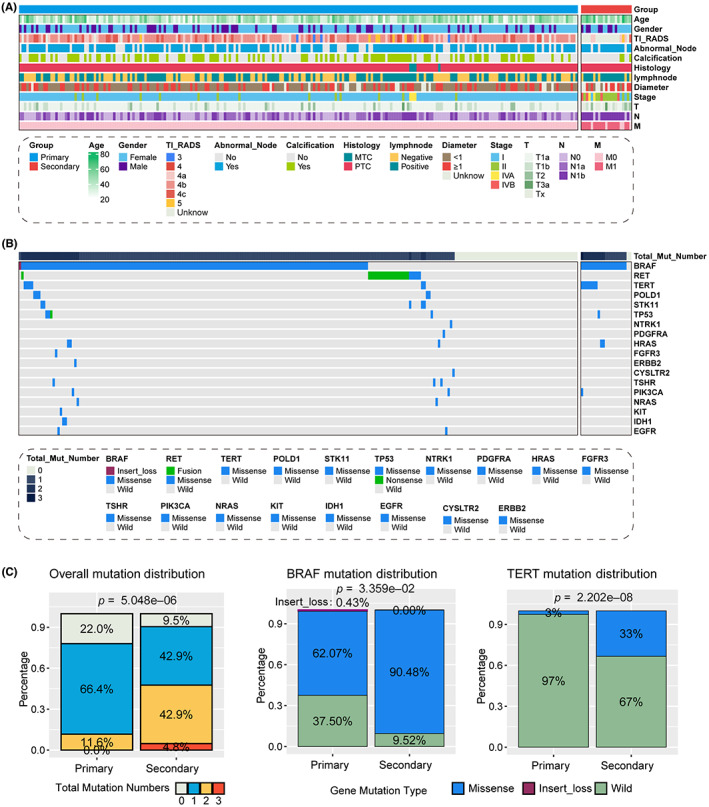
Differences between patients with and without lymph node metastasis of primary cases
As we talked in the introduction part, lymph node metastasis is tightly with the prognosis of TC, therefore it is essential to identify the novel factors to help the identification of lymph node metastasis in clinical work. We analyzed the diverse distribution of clinical and pathological features among patients with and without lymph node metastasis from AHMU‐TC cohort (Table 2; Figure 2). The distribution of patients’ sex is a little bit different, more than half enrolled male patients met lymph node metastasis (35.8% vs. 22.9%). Patients with positive lymph node were younger (40.9 ± 10.8 vs. 45.3 ± 11.8, p = 0.0031, Figure 2C) and contained advanced TI‐RADS levels (4c: 22.8% vs. 8.3%, 5: 6.5% vs. 0/0%, p = 1.878e‐03, Figure 2D). Moreover, we also found that patients with positive lymph node also had the larger tumor represented by the tumor diameter than those with negative lymph node ones (≥1 cm: 40.7% vs. 25.7%, p = 9.197e‐05, Figure 2F), as well as the presence of RET genetic alteration (16.3% vs. 2.7%, p = 2.566e‐03, Figure 2E).
TABLE 2: Different clinical features among patients with or without lymph node metastasis.
| Negative (N = 109) | Positive (N = 123) | p value | |
|---|---|---|---|
| Gender | |||
| Female | 84 (77.1%) | 79 (64.2%) | 0.044 |
| Male | 25 (22.9%) | 44 (35.8%) | |
| Age | |||
| Mean (SD) | 45.3 (11.8) | 40.9 (10.8) | 0.003 |
| Median [Min, Max] | 46.0 [24.0, 71.0] | 41.0 [21.0, 68.0] | |
| TI_RADS | |||
| 3 | 2 (1.8%) | 2 (1.6%) | <0.001 |
| 4a | 28 (25.7%) | 18 (14.6%) | |
| 4b | 67 (61.5%) | 61 (49.6%) | |
| 4c | 9 (8.3%) | 28 (22.8%) | |
| Unknown | 3 (2.8%) | 5 (4.1%) | |
| 4 | 0 (0%) | 1 (0.8%) | |
| 5 | 0 (0%) | 8 (6.5%) | |
| Abnormal Node | |||
| No | 35 (32.1%) | 26 (21.1%) | 0.073 |
| Yes | 74 (67.9%) | 97 (78.9%) | |
| Calcification | |||
| No | 62 (56.9%) | 59 (48.0%) | 0.19 |
| Yes | 47 (43.1%) | 64 (52.0%) | |
| Histology | |||
| MTC | 1 (0.9%) | 3 (2.4%) | 0.625 |
| PTC | 108 (99.1%) | 120 (97.6%) | |
| Diameter | |||
| <1 | 79 (72.5%) | 58 (47.2%) | <0.001 |
| ≥1 | 28 (25.7%) | 50 (40.7%) | |
| Unknown | 2 (1.8%) | 15 (12.2%) | |
| EGFR | |||
| Missense | 2 (1.8%) | 0 (0%) | 0.22 |
| Wild | 107 (98.2%) | 123 (100%) | |
| IDH1 | |||
| Missense | 1 (0.9%) | 1 (0.8%) | 1 |
| Wild | 108 (99.1%) | 122 (99.2%) | |
| KIT | |||
| Missense | 1 (0.9%) | 0 (0%) | 0.47 |
| Wild | 108 (99.1%) | 123 (100%) | |
| NRAS | |||
| Missense | 2 (1.8%) | 0 (0%) | 0.22 |
| Wild | 107 (98.2%) | 123 (100%) | |
| PIK3CA | |||
| Missense | 2 (1.8%) | 0 (0%) | 0.22 |
| Wild | 107 (98.2%) | 123 (100%) | |
| TSHR | |||
| Missense | 3 (2.8%) | 0 (0%) | 0.102 |
| Wild | 106 (97.2%) | 123 (100%) | |
| CYSLTR2 | |||
| Wild | 109 (100%) | 122 (99.2%) | 1 |
| Missense | 0 (0%) | 1 (0.8%) | |
| ERBB2 | |||
| Wild | 109 (100%) | 122 (99.2%) | 1 |
| Missense | 0 (0%) | 1 (0.8%) | |
| FGFR3 | |||
| Wild | 109 (100%) | 122 (99.2%) | 1 |
| Missense | 0 (0%) | 1 (0.8%) | |
| HRAS | |||
| Missense | 2 (1.8%) | 1 (0.8%) | 0.602 |
| Wild | 107 (98.2%) | 122 (99.2%) | |
| PDGFRA | |||
| Missense | 1 (0.9%) | 0 (0%) | 0.47 |
| Wild | 108 (99.1%) | 123 (100%) | |
| NTRK1 | |||
| Wild | 109 (100%) | 122 (99.2%) | 1 |
| Missense | 0 (0%) | 1 (0.8%) | |
| TP53 | |||
| Nonsense | 1 (0.9%) | 0 (0%) | 0.172 |
| Wild | 108 (99.1%) | 120 (97.6%) | |
| Missense | 0 (0%) | 3 (2.4%) | |
| STK11 | |||
| Missense | 2 (1.8%) | 3 (2.4%) | 1 |
| Wild | 107 (98.2%) | 120 (97.6%) | |
| POLD1 | |||
| Missense | 1 (0.9%) | 4 (3.3%) | 0.374 |
| Wild | 108 (99.1%) | 119 (96.7%) | |
| TERT | |||
| Missense | 5 (4.6%) | 1 (0.8%) | 0.102 |
| Wild | 104 (95.4%) | 122 (99.2%) | |
| RET | |||
| Fusion | 2 (1.8%) | 16 (13.0%) | 0.001 |
| Missense | 1 (0.9%) | 4 (3.3%) | |
| Wild | 106 (97.2%) | 103 (83.7%) | |
| BRAF | |||
| Missense | 72 (66.1%) | 72 (58.5%) | 0.31 |
| Wild | 37 (33.9%) | 50 (40.7%) | |
| Insert_loss | 0 (0%) | 1 (0.8%) |
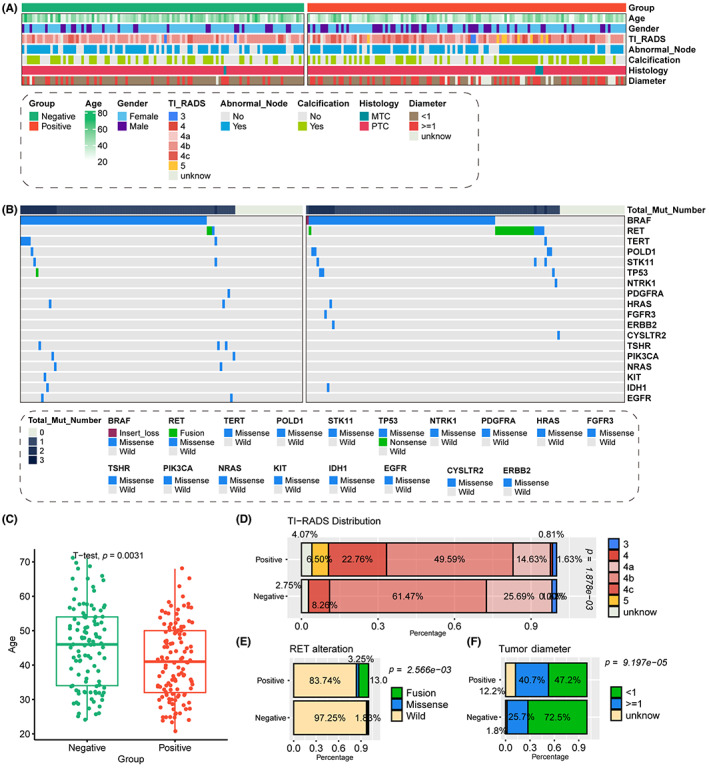
Identifying of the independent prognostic factor and constructing the nomogram
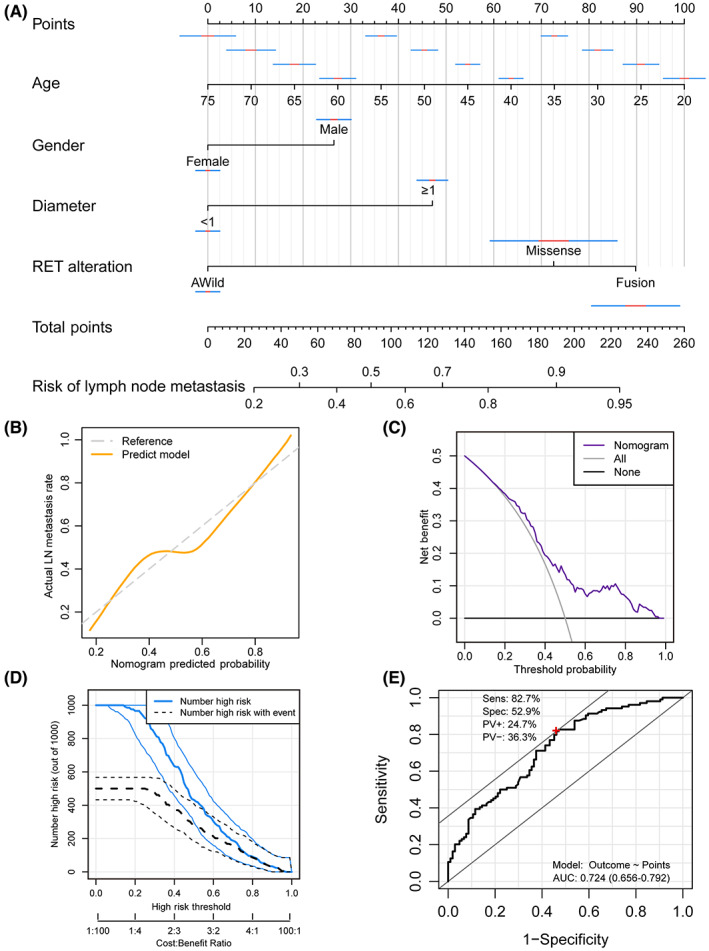
The factors that showed significantly difference between patients with/without lymph node metastasis, including age, gender, ultrasound defined TI‐RADS level, tumor diameter and RET genetic alteration, were enrolled for the multivariate logistic regression analysis (Table 3). We found that after adjusting the potential impact factors, patient age (p = 0.002), tumor diameter (p = 0.034), and RET fusion (p = 0.032) still as the prognostic factor to the presence of positive lymph node, while patient gender also showed a marginal prognostic value (p = 0.08). Therefore, we enrolled age, gender, tumor diameter, and RET alteration to construct the lymph node metastasis predict nomogram (Figure 3A). In the calibration plot, the nomogram predict result is tightly close to the refer line, which indicated that the prediction nomogram was an ideal predictive model (Figure 3B). DCA curve and the clinical impact curve was performed to demonstrate high clinical net benefit that almost over the entire threshold probability of the nomogram model (Figure 3C,D). To assess the predictive performance of the current model, we performed a ROC curve analysis, which exhibited an AUC value of 0.724 (0.656–0.792). The model’s sensitivity and specificity were 82.7% and 52.9%, respectively, serving as a supplementary indicator to signal the probability of lymph node metastasis for patients (Figure 3E).
TABLE 3: Multivariate logistic regression analysis for the prediction of lymph node metastasis.
| Characteristic | OR | 95% CI | β | SE | Wald value | p value |
|---|---|---|---|---|---|---|
| Age | 0.96 | 0.93, 0.98 | −0.044 | 0.014 | −3.062 | 0.002* |
| Gender | ||||||
| Female | — | — | — | — | — | |
| Male | 1.79 | 0.93, 3.49 | 0.584 | 0.335 | 1.742 | 0.081 |
| TI_RADS | ||||||
| 3 | — | — | — | — | — | |
| 4a | 0.69 | 0.07, 6.73 | −0.369 | 1.099 | −0.336 | 0.7 |
| 4b | 1.14 | 0.12, 10.6 | 0.134 | 1.068 | 0.125 | 0.9 |
| 4c + 5 | 3.66 | 0.35, 38.5 | 1.299 | 1.139 | 1.14 | 0.3 |
| Unknown | 1.09 | 0.08, 15.9 | 0.089 | 1.315 | 0.068 | >0.9 |
| Diameter | ||||||
| <1 | — | — | — | — | — | |
| ≥1 | 2.03 | 1.06, 3.95 | 0.710 | 0.335 | 2.121 | 0.034* |
| Unknown | 10.9 | 2.73, 73.4 | 2.388 | 0.799 | 2.987 | 0.003* |
| RET | ||||||
| Wild | — | — | — | — | — | |
| Fusion | 5.83 | 1.44, 39.4 | 1.762 | 0.804 | 2.193 | 0.028* |
| Missense | 4.43 | 0.55, 93.8 | 1.489 | 1.191 | 1.25 | 0.2 |
Abbreviations: CI, confidence interval; OR, odds ratio; SE, standard error for β.
* p < 0.05.
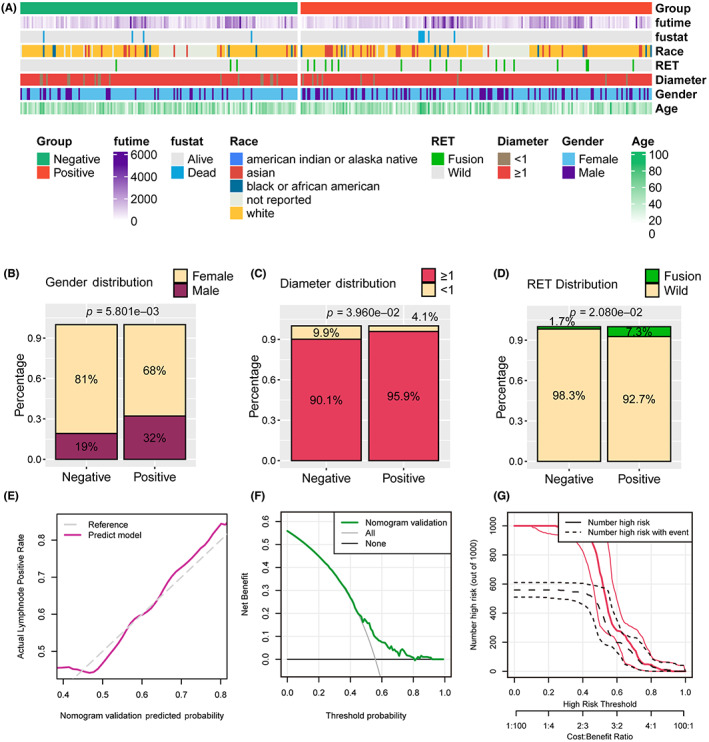
Validating the prognostic value of the nomogram in TCGA‐THCA cohort
We collected the clinicopathological features and gene mutation matrix of 390 TC patients from TCGA‐THCA cohort to validate the prognostic value of the nomogram, these features displayed in Figure 4A. We observed that more males met the positive lymph node after operation (32.0% vs. 19.0%, p = 0.0058, Figure 4B), which is consistent with our own cohort. Moreover, patients with positive lymph node also had the larger tumor represented by the tumor diameter than those with negative lymph node ones (≥1 cm: 95.9% vs. 90.1%, p = 0.0396, Figure 4C), as well as the presence of RET genetic alteration (7.3% vs. 1.7%, p = 0.0208, Figure 4D). In the calibration plot, the nomogram predict result is tightly close to the refer line, which indicated that the prediction nomogram was an ideal predictive model (Figure 4E). DCA curve and the clinical impact curves was performed to demonstrate high clinical net benefit that almost over the entire threshold probability of the nomogram model (Figure 4F,G).
Discover the potential mechanism of how RET fusion impact lymph node metastasis
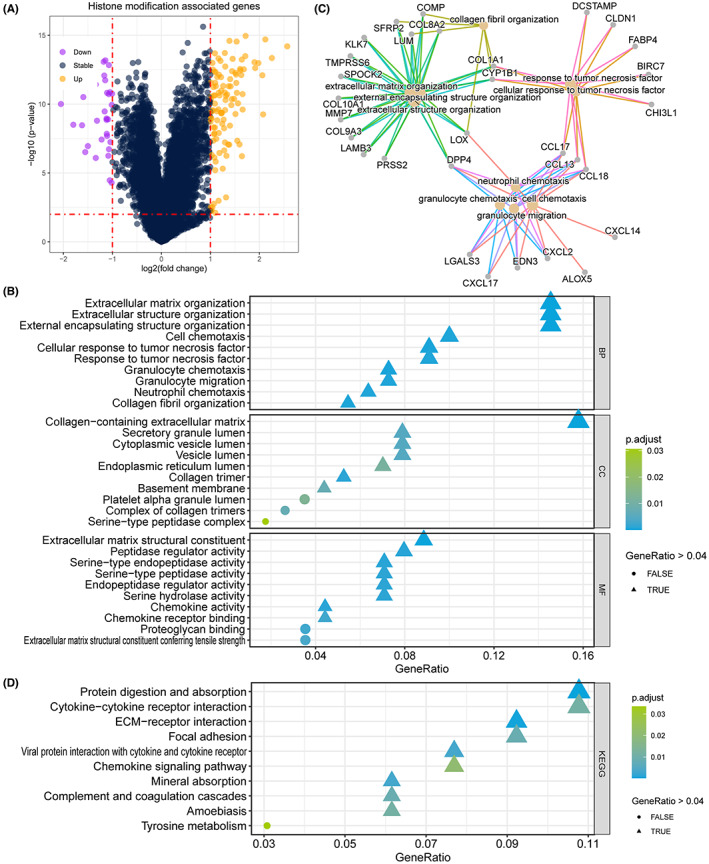
To reveal the potential correlation between RET fusion and TC patient lymph node metastasis, we collected the gene expression profile of the TCGA‐THCA cohort. We compared the different expression of genes, and found 102 unregulated gene in RET fusion samples, and 31 gene upregulated in samples with RET fusion (Figure 5A). And we subsequently annotated these 133 DEGs, and revealed that RET fusion mostly regulated the activation of extracellular matrix and structure organization, chemokine receptor binding (Figure 5B), and the network indicate that the key genes are LOX, DPP4, CCL17, CCL13, CCL18, CYP1B1, COL8A2 (Figure 5C). The KEGG pathway enrichment also revealed that RET fusion impact the process of protein digestion and absorption, cytokine‐cytokine receptor interaction, ECM‐receptor interaction, focal adhesion (Figure 5D).
A scheme of nomogram application
To enhance the practical applicability of the nomogram, we provide a specific example to guide its use in clinical predictions. For instance, in the case of a 53‐year‐old male thyroid cancer patient, we discovered that the tumor diameter exceeds 1 cm and presents a RET mutation. Consequently, according to the nomogram, we can identify the points corresponding to each parameter, which are 26, 40, 47, and 72. By summing these points, we obtain a total of 185 points, corresponding to an approximate probability of 0.88 for the occurrence of lymph node metastasis.
DISCUSSION
The increase in the incidence of thyroid cancer presents a “wave‐like” trend, its detection rate is increasing all over the world, and its diagnosis is getting younger and younger. The current mainstream view is that due to the high sensitivity and strong accuracy of high‐resolution ultrasound diagnostic technology, smaller and earlier thyroid nodules are continuously detected, and thyroid cancer accounts for about 5%–15% of them.ref. cam45945-bib-0021 At the same time, obesity, estrogen, radiation exposure, iodine intake, and other factors, have led to the high incidence of thyroid cancer in recent years.ref. cam45945-bib-0022, ref. cam45945-bib-0023, ref. cam45945-bib-0024
The occurrence and development of TC is a process involved multiple genetic alterations, including gene mutations, rearrangements, fusions, and methylation of oncogenes and tumor suppressor genes. PTC is the most common type of thyroid cancer, although a better outcome can be expected for PTC patients, 9.1%–38% of them will finally step into the status of lateral lymph node metastasis, which seriously affects the prognosis.ref. cam45945-bib-0025 Therefore, it is necessary to identify the risk factors of lymph node metastasis for TC patients, to distinguish patients with high risk of poor prognosis and give the aggressive treatment.
The product encoded by the BRAF gene is a protein kinase that regulates cell differentiation, reproduction, and apoptosis, and affects the carcinogenesis of the thyroid through the RAS‐RAF‐MEK‐ERK‐MAPK signaling pathway.ref. cam45945-bib-0026 According to the report of The Catalog of Somatic Mutations in Cancer database, the researchers collected nearly 80,000 cases of BRAF gene mutation, and found that BRAFV600E mutation ranks first among all types, accounting for more than 95%.ref. cam45945-bib-0027 In the current study, 65.21% patients were detected with BRAF mutations, 62.07% for primary cases, and 90.48% for secondary cases. BRAF mutation seems correlated with the recurrent of TC. Another study based on east China also reported that BRAF mutation is correlated with larger tumor size, higher probability of PTC recurrence and LNM.ref. cam45945-bib-0028 However, we failed to observe the correlation of BRAF mutation with LNM. A meta‐analysis reported that LNM is not associated with BRAF mutation of PTC patients.ref. cam45945-bib-0029
The RET proto‐oncogene encodes a transmembrane glycoprotein receptor with tyrosine kinase activity, of which participates in the processes of proliferation, differentiation and motility.ref. cam45945-bib-0030 The incidence of RET/PTC rearrangement is about 15%–20% in sporadic PTC, and the positive rate of RET gene mutation is nearly 50% in patients with medullary thyroid cancer. In people with clear radiation exposure, the positive rate of RET gene mutation can be as high as 65%. In the current study, we observed 23 cases contained the genetic alteration of RET, including 18 fusion and 5 missense mutation, all these genetic alterations presented in the primary cases, but no one in secondary cases. Furthermore, we also observed that RET fusion is the independent predictor for LNM, but not the missense mutation. Another clinical study from east China reported 10.4% RET fusions in 193 PTC patients,ref. cam45945-bib-0031 7.08% RET fusion reported in a study from 14 tertiary hospitals of China.ref. cam45945-bib-0032 Ullmann et al.ref. cam45945-bib-0033 also reported that RET‐driven tumors are more likely to have extrathyroidal extension, multifocal disease and distant metastases in American.
Drawing on the data from the 232 patients, we observed that age, gender, and tumor diameter exhibited predictive value for LNM, with early age, male gender, and a diameter greater than 1 cm appearing to be risk factors. Evidence from other studies supports our findings. A study from central China discovered that patients aged ≤18 years (OR = 4.41, p < 0.001) and 19–45 years (OR = 1.97, p = 0.002) had a higher risk of lateral LNM than patients aged >60 years.ref. cam45945-bib-0034 A systematic review and meta‐analysis encompassing 27,741 patients from 41 studies indicated that age < 45 years and male gender were risk factors for central lymph node metastasis.ref. cam45945-bib-0035 A 2010 study based on a Korean population found that tumor diameter >2 cm was significantly associated with lymph node metastasis,ref. cam45945-bib-0036 while a 2012 Japanese study supported this finding, stating that tumor size (>2 cm) was the strongest predictor of microscopic central and lateral node metastasis in multivariate logistic analysis. Moreover, tumor size most markedly impacted lymph node recurrence, but not distant recurrence.ref. cam45945-bib-0037 Considering the widespread use of Doppler ultrasound in the early diagnosis of TC, an increasing number of TC cases are being detected at an early stage. Consequently, current research should focus more on the impact of small tumor sizes on clinical prognosis, such as the <1 cm threshold established in this study.
Based on the identified prognostic factors, age, gender, tumor diameter, and RET alteration, we constructed the LNM prediction nomogram, validated by the calibration, DCA, and clinical impact curve, all the results indicated that the nomogram presenting a high accuracy. What’s more, we also validated the predict function of the nomogram based on the data from TCGA‐THCA cohort. For the potential underlying mechanism of how RET fusion impact LNM, we analyzed the DEGs and annotated, which pointed out that RET fusion might impact LNM through the regulation of cytokine‐cytokine receptor interaction, ECM‐receptor interaction and focal adhesion. We should focus on the advantages of the current study. First, we collected 253 TC patients and recorded the clinical information, pathological information and results of NGS. Second, we developed a LNM prediction nomogram based on the age, gender, tumor diameter, and RET alteration. Third, we successfully validated the prediction accuracy of the nomogram in external cohort. There are still several limitations of the current study. First of all, this study is a single center study, patients from multiple medical centers are necessary to collected to further validate the nomogram. In addition, the mechanism of how RET fusion promote LNM is limited, further experiment is needed in the future study, the current study is just a small step.
CONCLUSION
We provide a novel nomogram to predict the LNM for TC patients, concerns the features of patient’s age, gender, tumor diameter, and RET alteration. Further studies from multiple medical centers are essential to validate the predict stability and accuracy of the nomogram.
AUTHOR CONTRIBUTIONS
Meng Wei: Conceptualization (equal); investigation (equal); visualization (equal); writing – original draft (equal). Rui Wang: Data curation (equal); formal analysis (equal). Wanxue Zhang: Resources (equal); software (equal); visualization (equal). Jing Zhang: Investigation (equal); validation (equal); visualization (equal). Qiang Fang: Resources (equal); supervision (equal); validation (equal); writing – original draft (equal). zheng fang: Investigation (equal); methodology (equal). Bin Liu: Investigation (equal); supervision (equal); writing – review and editing (equal). Yongxiang Li: Conceptualization (equal); data curation (equal); writing – review and editing (equal).
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The research contents and research programs were reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University (Quick‐PJ‐2022‐13‐44).
Supplementary Materials
References
- Thyroid cancer mortality and incidence: a global overview.. Int J Cancer., 2015. [PubMed]
- Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries.. CA Cancer J Clin., 2021. [PubMed]
- Incidence and mortality of thyroid cancer in China, 2008‐2012.. Chin J Cancer Res., 2019. [PubMed]
- Thyroid cancer: incidence and mortality trends in China, 2005‐2015.. Endocrine., 2020. [PubMed]
- Thyroid cancer.. Lancet., 2016. [PubMed]
- Thyroid carcinoma.. Lancet., 2003. [PubMed]
- Cancer statistics, 2019.. CA Cancer J Clin., 2019. [PubMed]
- Predicting factors of central lymph node metastasis and BRAF(V600E) mutation in Chinese population with papillary thyroid carcinoma.. World J Surg Oncol., 2021. [PubMed]
- 2015 American Thyroid Association Management Guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines task force on thyroid nodules and differentiated thyroid cancer.. Thyroid., 2016. [PubMed]
- American Association of Clinical Endocrinologists, American College of Endocrinology, and Associazione Medici Endocrinologi Medical Guidelines for clinical practice for the diagnosis and management of thyroid nodules—2016 update.. Endocr Pract., 2016. [PubMed]
- RET gene mutation analysis and long‐term clinical outcomes of medullary thyroid cancer patients.. Nucl Med Commun., 2020. [PubMed]
- Skip metastasis in papillary thyroid carcinoma is difficult to predict in clinical practice.. BMC Cancer., 2017. [PubMed]
- Molecular genetics and phenomics of RET mutations: impact on prognosis of MTC.. Mol Cell Endocrinol., 2010. [PubMed]
- Recurrence and survival after gross total removal of resectable undifferentiated or poorly differentiated thyroid carcinoma.. Thyroid., 2016. [PubMed]
- Clinical impact of BRAF mutation on the diagnosis and prognosis of papillary thyroid carcinoma: a systematic review and meta‐analysis.. Eur J Clin Invest., 2016. [PubMed]
- The BRAF(V600E) mutation in papillary thyroid microcarcinoma: does the mutation have an impact on clinical outcome?. Clin Endocrinol (Oxf)., 2014. [PubMed]
- Association between BRAF (V600E) mutation and clinicopathological features of papillary thyroid carcinoma: a Brazilian single‐Centre case series.. Arch Endocrinol Metab., 2019. [PubMed]
- Update on fundamental mechanisms of thyroid cancer.. Front Endocrinol (Lausanne)., 2020. [PubMed]
- Complex heatmaps reveal patterns and correlations in multidimensional genomic data.. Bioinformatics., 2016. [PubMed]
- clusterProfiler 4.0: a universal enrichment tool for interpreting omics data.. Innovation (Camb)., 2021. [PubMed]
- Global patterns and trends in incidence and mortality of thyroid cancer in children and adolescents: a population‐based study.. Lancet Diabetes Endocrinol., 2021. [PubMed]
- Risk factors for thyroid cancer: what do we know so far?. Acta Clin Croat., 2020. [PubMed]
- The association of obesity with thyroid carcinoma risk.. Cancer Med., 2022. [PubMed]
- Diet as a possible influencing factor in thyroid cancer incidence: the point of view of the nutritionist.. Panminerva Med., 2021. [PubMed]
- Risk factors for central and lateral lymph node metastases in patients with papillary thyroid micro‐carcinoma: retrospective analysis on 484 cases.. Front Endocrinol (Lausanne)., 2021. [PubMed]
- BRAF gene: from human cancers to developmental syndromes.. Saudi J Biol Sci., 2015. [PubMed]
- The COSMIC (catalogue of somatic mutations in cancer) database and website.. Br J Cancer., 2004. [PubMed]
- The prognostic implication of the BRAF V600E mutation in papillary thyroid cancer in a Chinese population.. Int J Endocrinol., 2022. [PubMed]
- Risk and prognostic factors for BRAF(V600E) mutations in papillary thyroid carcinoma.. Biomed Res Int., 2022. [PubMed]
- RET as a diagnostic and therapeutic target in sporadic and hereditary endocrine tumors.. Endocr Rev., 2006. [PubMed]
- Diagnostic performance of next‐generation sequencing and genetic profiling in thyroid nodules from a single center in China.. Eur Thyroid J., 2022. [PubMed]
- Genetic and clinicopathologic characteristics of papillary thyroid carcinoma in the Chinese population: high BRAF mutation allele frequency, multiple driver gene mutations, and RET fusion may indicate more advanced TN stage.. Onco Targets Ther., 2022. [PubMed]
- RET fusion‐positive papillary thyroid cancers are associated with a more aggressive phenotype.. Ann Surg Oncol., 2022
- Patient’s age with papillary thyroid cancer: is it a key factor for cervical lymph node metastasis?. Eur J Surg Oncol., 2023
- The risk of central nodal metastasis based on prognostic factors of the differentiated thyroid carcinoma: a systematic review and meta‐analysis study.. Eur Arch Otorhinolaryngol., 2023
- Clinical risk factors associated with cervical lymph node recurrence in papillary thyroid carcinoma.. Thyroid., 2010. [PubMed]
- Tumor size is the strongest predictor of microscopic lymph node metastasis and lymph node recurrence of N0 papillary thyroid carcinoma.. Endocr J., 2013. [PubMed]
