Cannabinoid interventions for improving cachexia outcomes in cancer: a systematic review and meta‐analysis
Abstract
Cancer‐associated cachexia (CAC) is a wasting syndrome characterized by involuntary weight loss and anorexia. Clear definition and diagnostic criteria for CAC are lacking, which makes it difficult to estimate its prevalence, to interpret research and to compare studies. There is no standard treatment to manage CAC, but previous studies support the use of cannabinoids for cachexia in other chronic diseases including HIV and multiple sclerosis. However, only a few randomized controlled trials (RCTs) and one meta‐analysis of this intervention in cancer populations are available. Non‐randomized studies of interventions (NRSIs) are often excluded from reviews due to variable methodology and potential for biases. This review aimed to consider NRSIs alongside RCTs to provide a complete summary of the available evidence that clinical decision makers could use in future investigations. Literature searches were conducted using three databases for relevant RCTs or NRSIs according to Cochrane methodology. Abstract and full texts of retrieved manuscripts were selected and retrieved by two investigators based on the PRISMA‐A guidelines, and risk of bias and quality of evidence assessments were performed. Outcome data on weight, appetite, quality of life, performance status, adverse effects, and mortality were combined by narrative synthesis and meta‐analysis where possible. Ten studies were included, four of which were RCTs and six NRSIs matching the eligibility criteria. Very low‐quality evidence from meta‐analysis suggested no significant benefits of cannabinoids for appetite compared with control (standardized mean difference: −0.02; 95% confidence interval: −0.51, 0.46; P = 0.93). Patient‐reported observations from NRSIs suggested improvements in appetite. Another meta‐analysis of moderate quality evidence showed that cannabinoids were significantly less efficient than active or inactive control on quality of life (standardized mean difference: −0.25; 95% confidence interval: −0.43, −0.07; P = 0.007). The effectiveness of cannabinoids alone to improve outcomes of CAC remains unclear. Low‐quality evidence from both RCTs and NRSIs shows no significant benefits of cannabinoids for weight gain, appetite stimulation, and better quality of life, three important outcomes of cachexia. Higher quality research integrating cannabinoids into multi‐modal therapies may offer better opportunities for developing CAC‐specific treatments. This review also highlights that findings from non‐randomized studies of interventions (NRSIs) can provide evidence of the effects of an intervention and advocate for the feasibility of larger RCTs.
Article type: Review Article
Keywords: Cancer, Cachexia, Cannabinoids, Appetite, Weight, Quality of life
Affiliations: Division of Medicine University College London London UK; Department of Nutritional Sciences King’s College London London UK; Institute of Cardiovascular Sciences University College London London UK
License: © 2021 The Authors. Journal of Cachexia, Sarcopenia and Muscle published by John Wiley & Sons Ltd on behalf of Society on Sarcopenia, Cachexia and Wasting Disorders. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/jcsm.12861 | PubMed: 34881518 | PMC: PMC8818598
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.1 MB)
Introduction
Cachexia is a multifactorial wasting syndrome characterized by involuntary weight loss through the ongoing loss of muscle mass, with or without loss of adipose tissue.ref. jcsm12861-bib-0001, ref. jcsm12861-bib-0002, ref. jcsm12861-bib-0003 It is a life‐threatening aspect of advanced chronic disease, including cancer. Cancer‐associated cachexia (CAC) is driven by tumour‐host interactions resulting in progressive physical deterioration and functional impairment.ref. jcsm12861-bib-0004, ref. jcsm12861-bib-0005, ref. jcsm12861-bib-0006 It is associated with decreased quality of life (QoL) and tolerance to treatment and increased morbidity and mortality.ref. jcsm12861-bib-0002, ref. jcsm12861-bib-0007, ref. jcsm12861-bib-0008, ref. jcsm12861-bib-0009 Attempts to define cachexia are relatively recent; therefore, estimates of prevalence vary considerably,ref. jcsm12861-bib-0010 particularly in cancer patients depending on tumour type and stage.ref. jcsm12861-bib-0011 A lack of consensus on definition and diagnostic criteria also makes it difficult to interpret research on the effectiveness of interventions and to compare studies.ref. jcsm12861-bib-0002, ref. jcsm12861-bib-0003, ref. jcsm12861-bib-0012
There are no standard treatments or guidelines to manage CAC,ref. jcsm12861-bib-0008 but an effective strategy should aim to reduce or prevent wasting to favour survival in advanced cancer patients.ref. jcsm12861-bib-0013, ref. jcsm12861-bib-0014 Numerous therapeutic approaches have been developed to target wasting, weight loss and anorexia, three hallmarks of cachexia, including anti‐cytokine therapies and metabolic mediators to counter wasting (e.g. glucocorticoids, anabolic steroids, progestogens, and beta‐adrenoreceptor agonists); caloric or nutrient supplementation to prevent weight loss and promote muscle and weight gain; and using appetite stimulants like megestrol acetate and cannabinoids to manage anorexia.ref. jcsm12861-bib-0015, ref. jcsm12861-bib-0016, ref. jcsm12861-bib-0017 The benefits of megestrol acetate for appetite, caloric intake, nutritional status, QoL, and reduced muscle wasting in cachexia are well documented,ref. jcsm12861-bib-0015, ref. jcsm12861-bib-0018, ref. jcsm12861-bib-0019 but the weight gain associated with this drug often reflects fat deposition with little or no muscle growth.ref. jcsm12861-bib-0017, ref. jcsm12861-bib-0020, ref. jcsm12861-bib-0021 The potential of cannabinoids to relieve symptom burden in chronic diseases is recognized,ref. jcsm12861-bib-0022, ref. jcsm12861-bib-0023 but their effectiveness in CAC is unclear.
Cannabinoids mimic the effect of human endocannabinoids on metabolism and appetite by interacting with their receptors, CB1, and CB2,ref. jcsm12861-bib-0024 and may have therapeutic benefits for body weight and appetite. The most commonly studied cannabinoids are delta‐9‐tetrahydrocannabinol (THC) and cannabidiol (CBD). Currently, only three cannabinoid‐containing drugs are commercially available for clinical use. Both Marinol and Cesamet, also known as dronabinol and nabilone respectively, are synthetic analogs of THC indicated for chemotherapy‐induced nausea and vomiting in the USA and Canada.ref. jcsm12861-bib-0023 Dronabinol is sometimes also prescribed for HIV/AIDS‐associated wasting syndrome. Sativex is a cannabis extract buccal spray containing a mixed ratio of THC and CBD adjunctively indicated for neuropathic and cancer pain.ref. jcsm12861-bib-0025 None of the above are currently indicated for CAC.
Previous randomized controlled trials (RCTs) report that cannabinoids can induce improvements in body weight, appetite, physical functioning and QoL in cachectic patients with other chronic diseases including HIV infection and multiple sclerosis.ref. jcsm12861-bib-0026, ref. jcsm12861-bib-0027, ref. jcsm12861-bib-0028 However, this is not well studied in cancer patients. Most reports suggesting the benefits of cannabinoids for appetite in CAC are anecdotal or lack methodological homogeneity,ref. jcsm12861-bib-0029 and few RCTs and one meta‐analysis are available.ref. jcsm12861-bib-0016 The latter suggested cannabinoids were associated with improvements to appetite but not to QoL and more adverse events compared with placebo. However, the authors included few studies, which were small and likely underpowered, provided a poor description of their methodology, and carried out no supplementary searching suggesting that studies might have been missed.
Non‐randomized studies of interventions (NRSIs) on the effect of cannabinoids on outcomes of CAC are available. To our knowledge, NRSIs have not yet been considered in a systematic review and are often excluded due to their methodological variability and the potential for biases. However, the relatively small number of RCTs is likely to give an incomplete picture and result in missing outcomes.ref. jcsm12861-bib-0030, ref. jcsm12861-bib-0031 Findings from NRSIs could provide additional evidence on these outcomes and encourage the feasibility of larger, higher quality RCTs.ref. jcsm12861-bib-0032
Given the difficulty for clinical practitioners to manage cachexia and its severe health implications for patients, it is important to evaluate all the existing evidence relevant to developing efficient therapies. This review aimed to consider NRSIs alongside RCTs for a comprehensive approach to the available evidence on cannabinoid interventions in CAC, in order to inform clinical decisions and future investigations.
Methods
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis (PRISMA)ref. jcsm12861-bib-0033 and Cochrane Handbook for Systematic Reviews of Interventionsref. jcsm12861-bib-0034 guidelines. A protocol was developed prior to initiating the review, but not published. A research question was formulated using the PICO approach. The inclusion and exclusion criteria, outcomes, and search were planned to capture as many studies as possible. Prior to the start of the investigation, it was also agreed by the investigators that meta‐analyses would be performed where possible and narrative syntheses generated for all other outcomes.
Eligibility criteria
Participants
Although the existing criteria for recognition of CAC enables more efficient diagnosis,ref. jcsm12861-bib-0002, ref. jcsm12861-bib-0003 it is under‐recognized in clinical practice. To reflect diagnostic oversights, adult (>18 years) cancer patients, whose baseline characteristics were judged to describe cachexia, were eligible, including individuals of any gender, ethnicity, disease stage in any care setting, and undergoing chemotherapy or radiotherapy. Individuals with an eating disorder, undergoing treatment for appetite and weight loss, or with a history or current habit of marijuana use were excluded.
Intervention
Cannabinoid‐based interventions included any smoked or ingested medical marijuana, plant‐based cannabinoids (THC and CBD) and synthetic cannabinoids (dronabinol, nabilone, or any other pharmaceutical form).
Comparison
No restrictions on the comparisons were applied to allow inclusion of qualitative evidence. Treatment comparisons were any active or inactive control. Active control included nutritional interventions administered orally (food fortification, snacks, and nutrient/caloric supplementation), while pharmacological interventions and co‐interventions involved the use of active drugs (appetite stimulants, anticytokines, and metabolic mediators), and other forms of cannabis. Inactive control included placebo, standard care or no treatment.
Outcome measures
Primary outcomes included changes in weight and appetite and secondary outcomes included performance status (PS), quality of life (QoL), adverse events (AEs), treatment‐related side effects, and mortality. Outcomes could be patient‐reported or clinician‐reported, using continuous or discreet methods, including validated scales, questionnaires, and interviews. The rationale for selecting the above outcomes was guided by previous workref. jcsm12861-bib-0016, ref. jcsm12861-bib-0027 and these were selected to reflect how patients perceive the symptoms of cachexia.
Studies
No restrictions on study design were applied to permit a comprehensive evaluation of the outcomes in a population of advanced cancer patients, in which ethical concerns complicate methodological implementation, such as randomization or blinding. All RCTs and NRSIs were included (refer to Quality of studies and risk of bias).
Search strategy
Electronic searches
To account for the lack of a standard definition of cachexia, the search strategy was designed to incorporate any terms associated with CAC, including wasting syndrome and weight loss, and cannabis‐based interventions. The electronic databases Ovid MEDLINE, Ovid Embase, and PubMed were searched from inception to May 2020, combining keyword terms with medical subject headings (MeSH), or equivalent, where possible. The full search strategies are shown in Figure S1. No restrictions on language or publication date and status (i.e. published, unpublished, conference abstracts, awaiting assessment, and in progress) were applied to account for the expected scarcity of evidence in this field.
Supplementary searching
Databases of registered and ongoing studies and reviews were searched, including PROSPERO, the ISRCTN registry, and ClinicalTrials.gov. Additional studies were identified by reference checking and citation tracking from studies identified as eligible for inclusion. Clarification was sought from corresponding authors where necessary.
Data collection and analysis
Selection of studies
All references were imported into and duplicates were removed using EndNote. One investigator independently conducted the first screening to identify eligible titles and abstracts. Studies were excluded where interventions did not involve cannabis, target CAC, or report on any given outcome of interest. Any study that did not meet the inclusion criteria was excluded, while in a second screening the full texts of potentially eligible studies for inclusion were reviewed and selected, including articles in English, French, and Spanish. Any uncertainties were discussed and resolved with at least one more co‐investigator. The full text of some studies that met the eligibility criteria were unavailable online and these were not included as no response was received upon request for access and/or contacting the author. Because these may have been of value in the analysis, the description of these studies is available in Table S1.
Quality of evidence and risk of bias
Two different scales, both recommended Cochrane tools, were used to assess methodological quality and risk of bias regarding study participation and attrition, measurement of the prognostic factor and outcomes, confounding, statistical analysis and reporting. ‘Risk of bias’ (Rob2) was used to assess RCTs and ‘Risk of bias in non‐randomized Studies‐of Interventions’ (ROBINS‐I) to assess NRSIs. Uncertainties were discussed and resolved and each aspect was rated. The results of both assessments were combined and summarized in a table where +, −, or ? indicated the level of risk as low, high, or unclear, respectively.
The overall quality of evidence for each outcome was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. The quality of evidence was downgraded for any significant study limitations (risk of bias), indirectness, important inconsistency or heterogeneity, imprecision, or potential publication bias, or upgraded for large magnitude or confounding effects, and dose–response gradient. Data from NRSIs started at low quality. The body of evidence for each outcome was judged as very low, low, moderate, or high and summarized narratively (Table S2). Reasons for down‐grading or up‐grading evidence are referred in the table’s footnotes.
Data synthesis and statistical analysis
Data extraction was carried out using a data collection form designed on Microsoft Office Excel (Microsoft Corporation) for this review, and data input was reviewed by two investigators. The form included extracted data on study design, participants, intervention, publication details, and outcomes of interest from the eligible studies using a template designed for this review. Where numerical data were not addressed in the full text or supplementary material and was only reported in graphs, and when no response was received from corresponding authors, it was extracted using a ruler on a magnified version of the figure.
Studies were grouped according to their design (RCTs or NRSIs). Outcome data and trends were described in terms of the number of studies, relevant effects, and statistical significance (P < 0.05) reported on the outcome. Results were combined narratively or by meta‐analysis where possible.
Studies only reported sufficient data to conduct meta‐analyses for QoL and appetite, which were pooled using Review Manager (RevMan version 5.4; The Nordic Cochrane Center) using a continuous, inverse variance, random effects analysis. A random effects model was used because of variability in both study design and participants, and interventions. Mean change and standard deviations (SDs) were available for most studies. Otherwise baseline and end‐of‐study means, and P values, were used to calculate mean change and compute SDs. In one RCT comprising of two treatment arms, data from each arm was compared with half the number in the control group to avoid duplicate reporting.ref. jcsm12861-bib-0035 The standardized mean difference (SMD) was used to account for differences in tools or methods of data collection for similar outcomes.ref. jcsm12861-bib-0036, ref. jcsm12861-bib-0037 The inconsistency (I 2) statistic was used to assess heterogeneity, which was subsequently classified as I 2 < 40%—low; 30 to 60%—moderate; 50 to 90%—substantial and >75%—considerable.ref. jcsm12861-bib-0038
Results
Eight hundred and seventy‐five studies were identified from searches with 716 titles and abstracts screened for eligibility following deduplication. We obtained and scrutinized 64 full‐text papers, of which 10 were included in this review (Figure 1). Studies were excluded if they: were unrelated or irrelevant, did not meet the inclusion criteria, were awaiting completion, or were a registered trial of an included study. Two records were trials registered on ClinicalTrials.gov. Oneref. jcsm12861-bib-0039 was an ongoing study that begun in July 2020 and is awaiting completion in October 2021 (Table S3). Oneref. jcsm12861-bib-0040 was the ClinicalTrials.gov record reported by an included study. Six studies (Table S1) were excluded because the full text could not be obtained online or retrieved physically.
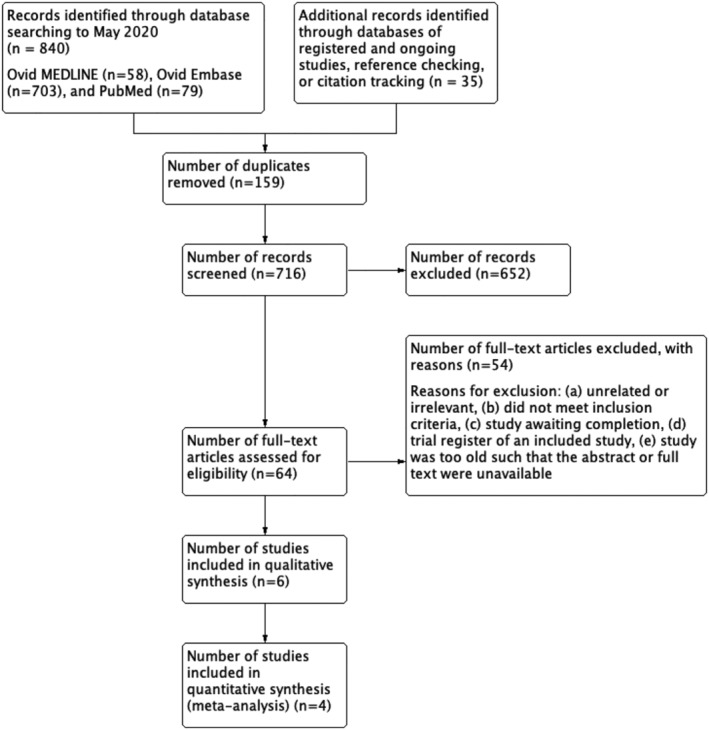
Characteristics of included studies
Ten studies were included in this reviewref. jcsm12861-bib-0023, ref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0042, ref. jcsm12861-bib-0043, ref. jcsm12861-bib-0044, ref. jcsm12861-bib-0045, ref. jcsm12861-bib-0046, ref. jcsm12861-bib-0047, ref. jcsm12861-bib-0048 (Table 1). All participants were adult cancer patients, mean age ranging from 47.3 to 67 years. The presence of cachexia was confirmed as previously described, using guidance from existing criteria. Study duration varied from 18 days to 6 months, including both intervention and follow up. Two studiesref. jcsm12861-bib-0042, ref. jcsm12861-bib-0048 were open‐label continuation studies. Methods of outcome reporting varied and comprised a range of both validated scales and procedures as described in Tables S5–S8.
Table 1: Characteristics of included studies
| Summary of studies | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| RCTs | |||||||||
| Study author, year | Study design | Duration and follow up | Participant characteristics | Sample size | Intervention | Route | Comparator | Outcome of interest | Additional outcomes |
| Brisbois et al., 2011ref. jcsm12861-bib-0041 | RCT, Canada | 18 days; F/U: 30 days | 12 male, 9 female; mean age (SD): Intervention group: 67(10.9) yearsComparator group: 65.5(8.0) years Advanced cancer with decreased food intake | I: 24C: 22 | 2.5 mg THC: | Oral | 2.5 mg placebo: | ||
| Jatoi et al., 2002ref. jcsm12861-bib-0042 | RCT, United Kingdom | open‐label continuation; ‘patient continued on treatment for as long as they and their healthcare providers thought it beneficial or until toxic side effects prompted study withdrawal’ | Intervention group: 65% male, 35% female; mean age (SD): 65(11) yearsComparator group: 66% male, 34% female; mean age (SD): 67(10)Advanced cancer with self‐reported weight loss >5 lbs (2.3 kg) in last 2 months, loss of appetite, <20 kcal/kg intake per day, and 0–2 PS score | I: 152C: 159 | 2.5 mg dronabinol capsules twice daily plus liquid placebo | Oral | 800 mg megestrol acetate liquid suspension daily plus capsule placebos | ||
| Strasser et al., 2006ref. jcsm12861-bib-0035 | RCT, Germany | 6 weeks; F/U at week 2, week 4 and week 6 | 54% men, 46% women; mean age: 61 yearsAdvanced cancer with involuntary weight loss >5% in last 6 months, anorexia, and <2 PS score | I: 100 (THC); 95 (cannabis extract, CE)C: 48 | THC: 2.5 mg THC capsulesCE: 2.5 mg: 1 mg THC:CBD capsulesThree times 2‐weeks supply taken twice daily (1 hour before lunch and dinner, or at bedtime), preferably with milk | Oral | Placebo capsules containing a standardization mediumThree times 2‐weeks supply taken twice daily (1 hour before lunch and dinner, or at | Other symptomsFunctional domains of QoL | |
| Turcott et al., 2018ref. jcsm12861-bib-0043 | RCT, Mexico | 8 weeks; F/U at week 2, 4 and 8 | Intervention group: 3 male (21.4%), 11 female (78.6%); mean age (SD): 61.1(10.6) years; 6 moderately malnourished, 8 severely malnourishedComparator group: 4 male (21.1%), 15 female (78.9%); mean age (SD): 52.6(11.8) years mean age; 6 moderately malnourished, 13 severely malnourishedConfirmed NSCLC with anorexia and <2 PS score | 47 (33 included in analysis) | 0.5 mg nabilone (CESAMET) for 2 weeksIncreased to 1 mg for next 6 weeks | Oral | 0.5 mg placebo for 2 weeksIncreased to 1 mg for next 6 weeks | ||
| Non‐RCTs | |||||||||
| Bar‐Sela et al., 2019ref. jcsm12861-bib-0044 | Pilot study | 6 months | 62.5% male, 38.5% female; median age: 66; median weight: 65.5 kgAdvanced cancer with weight loss of >5% in last 2 months, loss of appetite and <3 PS score | 11 | 10 mg THC:CBD (9.5:0.5)or5 mg THC:CBD (4.75:0.25) cannabis capsulesOnce daily for 2 weeks, then twice daily (first in the morning, then after 8 hours) | Oral | None | ||
| Kasvis et al., 2019ref. jcsm12861-bib-0045 | Retrospective observational study | 120 days; F/U at 30–75 days and 75–120 days (clinic visits) | Cancer patients referred to the Cannabis Pilot Project from McGill University Health CentreMean age (SD): 61 (11) years; 49% male, 51% female; 43% anorexia | 37 | medical cannabis treatment based on individual assessment by multidisciplinary team | Not specified | None | ||
| Kasvis et al., 2019ref. jcsm12861-bib-0046 | Retrospective chart review | 3 months | Mean age (SD): 47.3(16.1) years; 34 male (63%), 20 female (37%); 23 cancer (42.6%), 31 non‐cancer (57.4%); | 54 (51 included in analysis) | cannabinoid therapy (number of participants):THC/CBD (1:1): 6 total participantswith SC: 2 participantsno SC: 4 participants THC‐rich: 17 total participantswith SC: 8 participantsno SC:9 participants CBD‐rich: 0 participants combined therapies:THC/CBD and THC‐rich: 17 total participantswith SC: 7 participantsno SC: 10 participants THC/CBD and CBD‐rich: 7 total participantswith SC:3 participants no SC: 4 participants THC‐ and CBD‐rich: 17 total participantswith SC: 8 participantsno SC: 9 participants THC/CBD, THC‐rich, CBD‐rich: 1 total participantwith SC: 0 participantsno SC: 1 participant | 20.4% oral, 25.9% inhaled, 53.7% combined oral and inhaled | None | ||
| Nelson et al., 1994ref. jcsm12861-bib-0047 | Phase II trial | 28 days: F/U at week 2 and 4 | 13 male, 6 female; mean age: 65.12 years, median (range) age 64(52–81) years; median (range) PS: 2(0–3); median (range) mini‐mental status exam score: 29 (13–30);Advanced cancer patients | 10 | THC 2.5 mg p.o. t.i.d. one hour after meals2.5 mg b.i.d. for 3 days if >65 years | Oral | None | Unclear | |
| Plasse et al., 1991ref. jcsm12861-bib-0023 | Non‐RCT | 3 and 6 weeks | 33 male, 9 female; Karfnofsky performance status (median, range): 80 (60–100); previous THC exposure: 4Cancer patients | 42 | I: dronabinol treatment, 4 groups; group 1: 2.5 mg q.d.group 2: 2.5 mg b.i.d.group 3: 5 mg q.d.group 4: 5 mg b.i.d. study 1: group 3 received dose before breakfast;study 2: group 3 received dose before dinner; | Oral | None | ||
| Walsh et al., 2005ref. jcsm12861-bib-0048 | Case series | Open continuation; F/U biweekly or at every outpatient clinic visit until considered stable, then per routine clinical practice | Patients treated chronically with escalating dronabinol doses for cancer‐related anorexia | 6 | dronabinol was titrated from 7.5 to 15 mg daily in 5 patients, 1 patient remained on initial dose; | Oral | None | ||
Abbreviations: b.i.d., twice daily; C, comparison; CBD, cannabidiol; CE, cannabis extract;; F/U, follow up; HRQL, health‐related quality of life; I, intervention; NSCLC, non‐small cell lung cancer; p.o., oral; PS, performance status; q.d., daily; QoL, quality of life; RCT, randomized controlled trial; SC, synthetic cannabinoid; SD, standard deviation; THC, tetrahydrocannabinol; t.i.d., three times daily.
Four studies were RCTs (n = 647) assessing the effect of cannabinoids on at least one outcome (appetite, weight, or QoL) in advanced cancer patients and tworef. jcsm12861-bib-0041, ref. jcsm12861-bib-0043 were pilot studies. Three multi‐centre trialsref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0042 used dronabinol and one studyref. jcsm12861-bib-0043 used nabilone. Three RCTsref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0043 used a placebo as the control. The remaining study, a large RCT,ref. jcsm12861-bib-0042 used megestrol acetate plus placebo as the standard treatment arm, dronabinol capsules plus liquid placebo as the intervention arm, and a combination of both dronabinol and megestrol acetate in a third intervention arm. Only the standard treatment and first intervention arm were included in this analysis, because they both included placebo and were most comparable. One studyref. jcsm12861-bib-0035 included a third intervention arm with cannabis extract (CE) to compare with THC, which were individually compared with the comparison arm (placebo). Two RCTsref. jcsm12861-bib-0035, ref. jcsm12861-bib-0042 prescribed 2.5 mg THC doses twice daily for the entire treatment and tworef. jcsm12861-bib-0041, ref. jcsm12861-bib-0043 prescribed increasing doses (from 0.5 mg to 20 mg per day maximum).
The remaining six studies were NRSI (n = 157) assessing the effects of cannabis or cannabinoid treatment on appetite and weight in cancer patients. Participants’ physical status and disease stage, and study design, varied. Where specified, treatment was administered orally. Two were retrospective studiesref. jcsm12861-bib-0045, ref. jcsm12861-bib-0046 one of which oneref. jcsm12861-bib-0045 was only reported as a conference abstract. The otherref. jcsm12861-bib-0046 was a retrospective chart review where one of the following cannabinoid therapies were either taken orally or inhaled: 1:1 THC/CBD, THC‐rich, CBD‐rich, 1:1 THC/CBD + THC‐rich, 1:1 THC/CBD + CBD‐rich, THC‐ + CBD‐rich, or all three. Two were single‐arm intervention studiesref. jcsm12861-bib-0023, ref. jcsm12861-bib-0044 one of which included two consecutive studies (lasting 3 and 6 weeks, respectively). In this study, patients were assigned to one of four treatment groups: 2.5 mg once or twice daily or 5 mg once or twice daily. Patients on 5 mg once daily received their dose before breakfast in the first 3 weeks, then before dinner until the end of the 6 weeks. One was a Phase‐II trialref. jcsm12861-bib-0047 and one was a case seriesref. jcsm12861-bib-0048 both of which specifically aimed to treat cancer‐related anorexia.
Risk of bias and quality assessment
Risk of bias was assessed for all outcomes of interest for which both objective and subjective measures were used in data collection methods (Figure 2). Risk of bias was determined as unclear by the author wherever information was lacking or vague. All four RCTs were at low risk of selection and reporting bias due to appropriate randomization, allocation, and analyses methods. Oneref. jcsm12861-bib-0035 was at high risk of performance bias due to major protocol violations by 84 participants, and one was unclearref. jcsm12861-bib-0043 because allocation was carried out by the protocol coordinator. Low or unclear risk of bias in the remaining three domains was determined primarily due to uncertainties concerning how missing data, and withdrawals were handled.
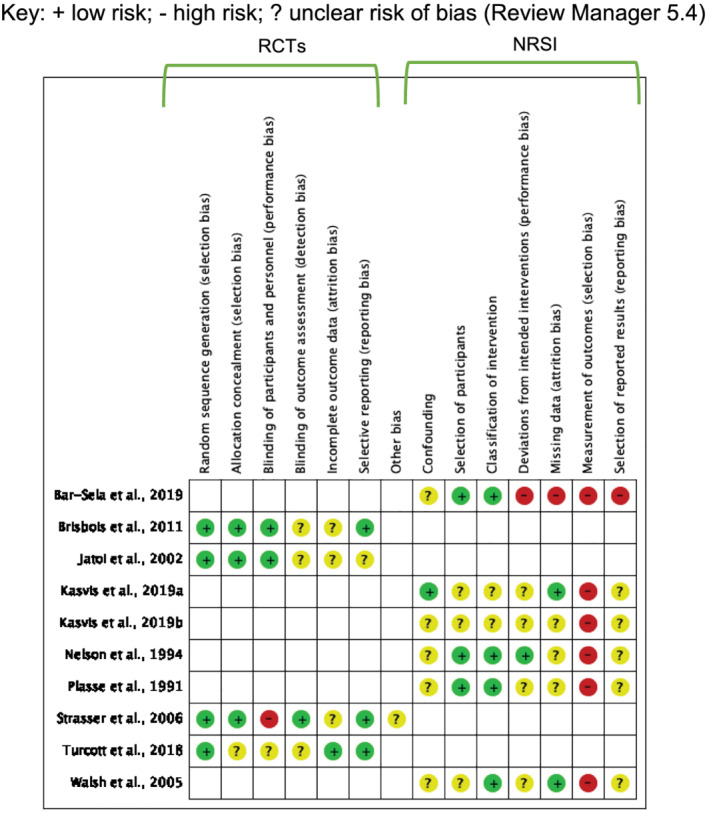
The remaining studies were at unclear risk of performance or reporting bias due to uncertainties. They also all had unclear risk of confounding for not controlling for pre‐conceptions associated with cannabis intake (i.e., cravings, relaxation and sleepiness related to the THC‐induced high, a bad trip or other associated side effects), recall bias, and no controls.
Risk of classification of intervention was judged to be lowref. jcsm12861-bib-0023, ref. jcsm12861-bib-0044, ref. jcsm12861-bib-0047, ref. jcsm12861-bib-0048 or unclearref. jcsm12861-bib-0045, ref. jcsm12861-bib-0046 on the basis that the nature of the intervention was reported in sufficient detail. There was lowref. jcsm12861-bib-0046, ref. jcsm12861-bib-0048 or unclearref. jcsm12861-bib-0023, ref. jcsm12861-bib-0045, ref. jcsm12861-bib-0047 risk of attrition bias due to unexplained missing outcome data. All studies were at high risk of selection bias because interventions were not blinded. One studyref. jcsm12861-bib-0044 was also at high risk of performance, attrition, and reporting bias due to significant withdrawals, no explanation or evidence of adjustments or sensitivity analyses, and insufficient information.
Primary outcomes
Weight
Eight of ten studies (80%), two RCTs and six NRSIs, reported data on aspects of this outcome (Table 2). The method of assessing weight varied and included self‐reported or physician‐reported, mean baseline and end‐of‐study body weight, body weight change (gain or loss) reported as a percentage, mean or median, median rate of weight loss, and as percentage of patients with >10% weight gain.
Table 2: Summary of findings from studies reporting on weight
| Outcome (units) | Study author, year | Intervention | Sample size | Results | ||
|---|---|---|---|---|---|---|
| Intervention | Control | p value | ||||
| RCTs | ||||||
| % patients with >10% increase above BL | Jatoi et al., 2002ref. jcsm12861-bib-0042 | 2.5 mg dronabinol capsules b.i.d. | I: 152C: 159 | Self‐reported 3, physician‐reported 5 | Self‐reported 11, physician‐reported 14 | 0.02, 0.009 |
| % patients with a max weight gain of 0%, 1–4%, 5–9% or >10% | 65, 23, 8, & 3 | 57, 23, 10, & 10 | 0.041 | |||
| Mean change in body weight, kg (SD) | Turcott et al., 2018ref. jcsm12861-bib-0043 | 0.5 mg nabilone for 2 weeks, then 1 mg for 6 weeks | I: 14C: 19 | −1.4 (1.6) | −1.09 (2.6) | 0.724 |
| NRSI | ||||||
| Percent % change, range | Bar‐Sela et al., 2019ref. jcsm12861-bib-0044 | Cannabis capsules10 mg THC:CBD (9.5:0.5) or5 mg THC:CBD (4.75:0.25)q.d. for 2 weeks then b.i.d. (morning, then +8 h) | 11 | 7.7–21.6 | None | Not reported |
| Mean BL and final weight, kg (SD)Mean weight change, kg | Kasvis et al., 2019ref. jcsm12861-bib-0045 | medical cannabis treatment based on individual assessment by multidisciplinary team | 37 | BL: 70.7 (19.3)Final: 66.1 (23.0)Calculated from mean BL and final weight: −4.6 | None | 0.509 |
| Mean BL and final weight, kg (SD)Mean weight change, kg | Kasvis et al., 2019ref. jcsm12861-bib-0046 | cannabinoid therapy (# of participants):THC/CBD (1:1) (6 total)THC‐rich (17 total)CBD‐rich (0 total)combined therapies (# of participants):THC/CBD and THC‐rich (17 total)THC/CBD and CBD‐rich (7 total)THC‐ and CBD‐rich (17 total)THC/CBD, THC‐rich, CBD‐rich (1 total) | 54 (51 included in analysis) | BL: 70.7 (14.6)Final: 71.0 (14.8)Calculated from mean BL and final weight: 0.3 | None | Not reported |
| Median weight change, kg (range) | Nelson et al., 1994ref. jcsm12861-bib-0047 | THC 2.5 mg p.o. t.i.d. 1 h post‐meals | 6 | 1.3 (1.0–2.7) | None | Not reported |
| Descriptive weight change, n | 2.5 mg b.i.d. for 3 days if >65 years | 3 gained weight2 maintained a stable weight1 lost weight | None | N/A | ||
| Median rate of weight loss before and after therapy, kg/months | Plasse et al., 1991ref. jcsm12861-bib-0023 | I: dronabinol treatment, 4 groups; group 1: 2.5 mg q.d.group 2: 2.5 mg b.i.d.group 3: 5 mg q.d.group 4: 5 mg b.i.d. Study 1: group 3 received dose before breakfast;Study 2: group 3 received dose before dinner; | 42 | Before I: group 1: −3.2group 2: −3.2group 3: −1.5group 4: −1.1 After I: group 1: −2.3group 2: −1.5group 3: −1.4group 4: 0.2 | None | Group 1 and group 3 only: <0.05 |
| Median weight gain, kg (range) | Walsh et al., 2005ref. jcsm12861-bib-0048 | 7.5 to 15 mg dronabinol q.d. in 5 patients, 1 patient remained on initial dose | 5 | 1 (0.5) | None | Not reported |
| Individual patient weight change, patient #, kg | 6 | 1: +12: 03: n/a (withdrew)4: +55: +16: +3 | None | Not reported | ||
Abbreviations: b.i.d., twice daily; C, comparison; CBD, cannabidiol; CE, cannabis extract; F/U, follow up; I, intervention; NRSI, non‐randomized study of intervention; q.d., daily; RCT, randomized controlled trial; SC, synthetic cannabinoid; SD, standard deviation; THC, tetrahydrocannabinol; t.i.d., three times daily.
One RCTref. jcsm12861-bib-0042 found the standard treatment, megestrol acetate, resulted in greater weight gain (P = 0.02). The other RCT with placebo as a control reported no difference in mean (SD) change in weight between groups.ref. jcsm12861-bib-0043 Four of six NRSIsref. jcsm12861-bib-0023, ref. jcsm12861-bib-0044, ref. jcsm12861-bib-0046, ref. jcsm12861-bib-0047 reported small improvements to weight in groups receiving cannabinoids. Improvements ranged from 0.3 kg mean weight change,ref. jcsm12861-bib-0046 1.0–1.3 kg median weight gain,ref. jcsm12861-bib-0047, ref. jcsm12861-bib-0048 and 7.7–21.6% increase in weight.ref. jcsm12861-bib-0044
One of the two remaining NRSI reported a reduction in weight in groups receiving cannabinoids,ref. jcsm12861-bib-0045 and the other reported a smaller rate of weight loss in groups receiving higher doses of dronabinol (P < 0.05).ref. jcsm12861-bib-0023 The quality of evidence was very low.
Appetite
All studies (100%) reported on this outcome. Appetite was assessed using a range of different methods including validated scales,ref. jcsm12861-bib-0023, ref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0043, ref. jcsm12861-bib-0044, ref. jcsm12861-bib-0045, ref. jcsm12861-bib-0046 validated questionnairesref. jcsm12861-bib-0042, ref. jcsm12861-bib-0047 and self‐evaluations.ref. jcsm12861-bib-0048
Three RCTsref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0043 (n = 297) reported data in a format suitable for pooling in a meta‐analysis (Figure 3, Table S4). There was no difference in change in appetite in groups receiving cannabinoid treatment compared with groups receiving placebo, standard mean difference (SMD): −0.02 [95% confidence interval (CI): −0.51, 0.46; P = 0.93]. Heterogeneity was substantial (I 2 = 63%, P = 0.04). A sensitivity analysis revealed that when the study favouring interventionref. jcsm12861-bib-0041 was excluded, I 2 was reduced to 0% and there remained no difference between groups. This study had a small number of participants, used a different tool to evaluate appetite and reported much greater changes in score, drawing attention to the validity of the methods and subsequent results.
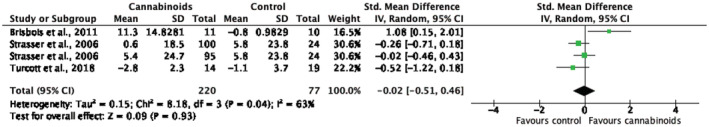
Data from the remaining seven NRSIs (Table 3) were not suitable for a meta‐analysis due to insufficient information and absence of comparison. In two of the RCTs,ref. jcsm12861-bib-0035, ref. jcsm12861-bib-0042 more patients in the control group reported improved appetite compared with the intervention group, with oneref. jcsm12861-bib-0042 reporting a significantly greater improvement in appetite in the group receiving megestrol acetate. All six NRSIs reported a positive effect on appetite post‐treatment, three of which reported a significant improvementref. jcsm12861-bib-0023, ref. jcsm12861-bib-0045, ref. jcsm12861-bib-0046 The remaining three observed fewer complaints about appetite lossref. jcsm12861-bib-0044 and better ratings of appetite, calorie count, and food intakeref. jcsm12861-bib-0047 from patients, although statistical significance was not reported. The quality of evidence was very low.
Table 3: Narrative summary of findings from studies reporting on appetite
| Study ID | Method of data collection and sample size | Outcomes |
|---|---|---|
| RCTs | ||
| Jatoi et al., 2002ref. jcsm12861-bib-0042 | Best follow‐up response (%) from validated questionnaires completed at BL and 1 monthI: 159C: 152 | More patients in the control group vs. the intervention group reported: |
| Strasser et al., 2006ref. jcsm12861-bib-0035 | Appetite Loss categoric scale in the EORTC QLQ‐C30jcsm12861-bib-0049 I: THC: 100CE: 95 C: 48 | |
| NRSI | ||
| Bar‐Sela et al., 2019ref. jcsm12861-bib-0044 | Appetite Loss subscale in the EORTC QLQ‐C30 before and at the end of the studyI: 6C: none | Significantly fewer complaints about appetite loss post‐treatment (P = 0.05)Increase in appetite 2 weeks post‐treatmentIndividual scores before and after treatment (n: before, after): 1: 0, 02: 32, 323: 78, 324: 78, 05: 78, 06: 100, 32 |
| Kasvis et al., 2019ref. jcsm12861-bib-0045 | Revised ESASjcsm12861-bib-0050 questionnaire completed at baseline, Visits 1 and 2I: 37C: none | |
| Kasvis et al., 2019ref. jcsm12861-bib-0046 | ESAS questionnaire, repeated at 3 months F/UI: 54C: none | |
| Nelson et al., 1994ref. jcsm12861-bib-0047 | 3‐Q interview, 1 appetite‐related Q: Since starting this drug, has your appetite shown no improvement, shown slight improvement, shown major improvement, or become completely normal? I: 18C: noneWeekly 1 day food diary recorded by the patientI: 19C: none | 13 patients reported improved appetite (Spearman’s rank correlation, r = 0.16) 10 reported ‘slight improvement’3 reported ‘major improvement’ Calorie count: Improved in 8 patientsDid not change in 1 patientNot reported in 10 patientsMedian (range) increase: 1032 (574–2436) kcal/day |
| Plasse et al., 1991ref. jcsm12861-bib-0023 | VAS completed before each meal with defining terms ‘extremely hungry’ and ‘not hungry at all’I: 42C: none | General increase in median appetite score (mm) from BL to end of study for each group (P < 0.05 between Groups 1 and 3 and 1 and 4): |
| Walsh et al., 2005ref. jcsm12861-bib-0048 | Self‐reported subjective evaluation5‐Q biweekly interview in outpatient clinicsI: 6C: none | ‘Rated their food intake as better or the same’‘Maintain stable appetite despite progressive disease’Question 1: How is your appetite? Better: 3 patientsSame: 3 patients Question 2: How is your food intake? Better: 2 patientsSame: 4 patients |
Abbreviations: BL, baseline; C, comparison; CE, cannabis extract; EORTC‐QLQ‐C30, European Organisation for Research and Treatment of Cancer—Quality of Life Questionnaire—Core 30; ESAS(‐r), Edmonton Symptom Assessment System (‐Revised); F/U, follow up; I, intervention; RCTs, randomized controlled trials; THC, tetrahydrocannabinol; VAS, visual analogue scale.
Secondary outcomes
Performance status
Two NRSIs (20%) reported on PS (Table 4). There were no changes in median PS pre‐ and post‐treatment in one study.ref. jcsm12861-bib-0047 Two participants in a case series discontinued treatment due to worsened PS.ref. jcsm12861-bib-0048 The quality of evidence was very low.
Table 4: Narrative summary of findings from studies that reported on PS and QoL
| Study ID | Method of data collection and sample size | Outcomes reported |
|---|---|---|
| NRSI reporting on PS | ||
| Nelson et al., 1994ref. jcsm12861-bib-0047 | ECOG PS scoreI: 19 | Median (range) at: |
| Walsh et al., 2005ref. jcsm12861-bib-0048 | Self‐reported subjective evaluation of PSI: 6 | 2 patients discontinued because of worsened PS |
| NRSI reporting on QoL | ||
| Bar‐Sela et al., 2019ref. jcsm12861-bib-0044 | EORTC QLQ‐C30 version 2 at Day 1I: 6C: none | Reported no difference before and after intervention |
| Walsh et al., 2005ref. jcsm12861-bib-0048 | Self‐reported subjective measure of well‐beingI: 5C: none5‐question interview at every outpatient clinic visit (biweekly), rated as B (better), W (worse), S (the same) or N (no)I: 6C: none | ‘Improved or remained stable’Question 3: How do you feel overall? 3 patients reported feeling better2 patients reported feeling the same1 patient reported feeling worse Question 4: How is your energy level? 3 patients reported better energy levels2 patients reported the same energy levels 1 patients reported worse energy levels |
Abbreviations: C, comparison; ECOG, Eastern Cooperative Oncology Group; EORTC‐QLQ‐C30, European Organisation for Research and Treatment of Cancer, Quality of Life Questionnaire, Core 3; ESAS, Edmonton Symptom Assessment System; I, intervention; NRSI, non‐randomized study of intervention; RCT, randomized controlled trial; SD, standard deviation.
Quality of life
Six of ten studies (60%), four RCTs and two NRSIs, reported on this outcome with QoL being assessed using both validated scalesref. jcsm12861-bib-0035, ref. jcsm12861-bib-0041, ref. jcsm12861-bib-0042, ref. jcsm12861-bib-0043, ref. jcsm12861-bib-0044 and self‐evaluations.ref. jcsm12861-bib-0048 Where QoL instruments were used, a higher score was indicative of a higher Global QoL, and a greater change in score was indicative of a greater improvement.ref. jcsm12861-bib-0051, ref. jcsm12861-bib-0052
There was sufficient data on Global QoL from four RCTs (n = 545) to allow a meta‐analysis (Figure 4, Table S5). There was a small and significantly greater improvement in Global QoL in groups receiving either active (megestrol acetate) or inactive (placebo) control compared with groups receiving cannabinoids, suggesting that cannabinoid treatment was less efficacious, SMD: −0.25 (95% CI: −0.43, −0.07); P = 0.007). There was no heterogeneity (I 2 = 0%, P = 0.58).
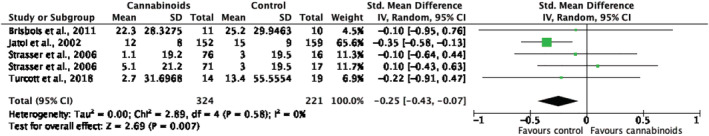
Data from the remaining two NRSIs (Table 4) were not suitable for a meta‐analysis due to insufficient data or the absence of a comparison group. Bar‐Sela et al.ref. jcsm12861-bib-0044 reported no difference between pre‐ and post‐intervention and Walsh et al.ref. jcsm12861-bib-0048 described relative improvements or stability in perceived well‐being and energy levels.
Data from one studyref. jcsm12861-bib-0046 reporting QoL in a wider patient population not treated with cannabinoids (Table S6) was excluded from the analysis because it did not match the inclusion criteria for intervention. The quality of evidence was moderate.
Adverse events
Nine of ten studies (90%), four RCT and five NRSI, reported on this outcome (Table S7). Assessment of AEs included number or percentage of patient‐reported symptoms or events, loss of follow up related to cancer, number of withdrawals or drop‐outs, and evaluation of side effects.
Two of the RCTsref. jcsm12861-bib-0041, ref. jcsm12861-bib-0042 showed no significant difference for the number or severity of AEs and serious AEs (SAEs), or the incidence of side effects, in the intervention compared with the control group. Oneref. jcsm12861-bib-0041 reported four AEs and one SAE were possibly related to treatment. The other two RCTsref. jcsm12861-bib-0035, ref. jcsm12861-bib-0043 showed no significant effect, although oneref. jcsm12861-bib-0035 reported more AEs in the intervention compared with the control group.
Twenty AEs were likely to be treatment‐related, although the intervention group was twice as numerous as the control group.
Of the five NRSI, oneref. jcsm12861-bib-0044 reported withdrawals due to treatment‐related effects. This same study also reported positive secondary effects of treatment on pain and fatigue reduction, and sleep and mood improvement after intervention. Most patients in four NRSI reported side effects,ref. jcsm12861-bib-0023, ref. jcsm12861-bib-0046, ref. jcsm12861-bib-0047, ref. jcsm12861-bib-0048 although it is unclear if those are related to the intervention. Only one studyref. jcsm12861-bib-0048 reported no new problems and treatment tolerability. The quality of evidence was very low.
Mortality
Three RCTs (30%) reported on mortalityref. jcsm12861-bib-0035, ref. jcsm12861-bib-0042, ref. jcsm12861-bib-0043 noting that more participants died in the intervention group compared with the control group. In one RCT,ref. jcsm12861-bib-0042 participants in the intervention group lived longer overall than participants in the control group (Table S8). The number of deaths in each study was small, and the quality of evidence for this outcome was very low.
Discussion
Cachexia is wasting condition, which seriously threatens patient prognosis. Currently, there is not a standardized treatment, and the lack of a robust definition hinders our ability to conduct high‐quality research. Various therapeutic approaches have been investigated, but their effectiveness in reducing and preventing weight loss and anorexia, two key indicators of cachexia, varies. This systematic review and meta‐analysis evaluated the effect of cannabinoid‐based interventions in CAC patients for changes in body weight, appetite, QoL, PS, AEs, and mortality. The aim was to compare evidence from NRSIs alongside RCTs to provide a thorough summary of the available evidence. To our knowledge, this is the first review to include NRSIs in its analysis.
Results from six NRSIs and four RCTs were comparable to a previously published systematic review of RCTs.ref. jcsm12861-bib-0016 In this review we showed it is unclear that cannabinoids alone can induce significant improvements in weight, appetite, quality of life, performance status, adverse effects, or mortality in CAC populations.
Weight and appetite loss caused by CAC are associated with a poorer prognosis,ref. jcsm12861-bib-0006 and have become common therapeutic targets in research. Previous studies report a beneficial effect of cannabinoids for weight and appetite in elderly and chronically ill patients.ref. jcsm12861-bib-0026, ref. jcsm12861-bib-0053, ref. jcsm12861-bib-0054 Very low quality evidence suggested no such benefits on weight in cancer patients, independently of the study design. This is consistent with a study showing no weight change for dronabinol compared with other appetite‐stimulating medicationsref. jcsm12861-bib-0055 and a systematic review reporting no significant weight change in cancer patients taking cannabis.ref. jcsm12861-bib-0027 One NRSIref. jcsm12861-bib-0023 found a significant reduction in rate of weight loss, suggesting that cannabinoids may be more useful in delaying weight loss rather than reversing it.
Other pharmacological interventions, such as megestrol acetate and ghrelin, focused on reduced appetite and food intake resulting from weight loss.ref. jcsm12861-bib-0015, ref. jcsm12861-bib-0019 Very low‐quality evidence suggests no significant improvements in appetite and a meta‐analysis showed that cannabinoids had no greater efficacy than a control treatment. There was high heterogeneity (I 2 = 63%, P = 0.04) and a small number of participants, so that further larger trials are needed. One of the included RCTs found that megestrol acetate led to greater weight gain and significant improvements in appetite,ref. jcsm12861-bib-0042 further discouraging any therapeutic advantage of cannabinoids without better‐quality studies. These findings contradict two previous systematic reviews that reported a small benefit.ref. jcsm12861-bib-0016, ref. jcsm12861-bib-0027 The inclusion of NRSIs in the analysis may explain this disparity. Also, previous work by Wang et al.ref. jcsm12861-bib-0016 concluded a significant effect of the intervention only after excluding the study that reported lower rate of increased appetite in the intervention group, without providing a clear rationale.
Other publications support the benefits of cannabinoids for food intake and appetite in healthy populations and HIV patients.ref. jcsm12861-bib-0027, ref. jcsm12861-bib-0056, ref. jcsm12861-bib-0057 Future research should invest how the pathophysiology of CAC may inhibit this effect in cancer patients. For example, chronic cannabis use was associated with decreased food intake and a lower prevalence of obesity in two representative US surveys.ref. jcsm12861-bib-0058 Because obesity and CAC share a pathophysiology characterized by systemic inflammation and metabolic disturbances, it could be interesting to investigate if and how these mechanisms interfere in the normal stimulatory effect of cannabinoids. In particular, if they act on the human CB1 and CB2 receptors, known to act on regulatory pathways of appetite and metabolism.
Although the majority of studies used validated methods, the body of evidence was rated as very low due to methodological limitations including unmasked allocation and outcome assessment. For example, the known effects of cannabis, such as the ‘munchies’ (sudden and strong desire for food) could have influenced any patient‐perceived improvements in appetite, food intake, calorie count, and/or taste of food. Further studies on this outcome are warranted.
The benefits of cannabinoids on QoL are elusive because both across RCTs and NRSIs, the evidence is anecdotal or self‐reported. The only systematic review evaluating this outcome concluded contradictory results, where cannabinoids both have a potent and counterproductive effect on QoL.ref. jcsm12861-bib-0016 Other findings, including those in the present review, remain inconclusive.ref. jcsm12861-bib-0059 Evidence of moderate quality found no significant benefits of cannabinoids on QoL, while a meta‐analysis favoured an effect in the control group. Unlike for appetite, data from NRSIs also concluded no effect. Our analysis aligns with other publications that cannabinoids may be less efficient to improve QoL.ref. jcsm12861-bib-0027, ref. jcsm12861-bib-0060 This effect was independent of study design, supporting the informative value NRSI may bring to RCT data.ref. jcsm12861-bib-0030, ref. jcsm12861-bib-0061
The importance of QoL in cancer care is reported elsewhere in the literature.ref. jcsm12861-bib-0062, ref. jcsm12861-bib-0063, ref. jcsm12861-bib-0064 A qualitative study analysing patient perception of cannabis use emphasized that marijuana and its derivatives were increasingly popular among cancer patients to alleviate treatment‐related side effects.ref. jcsm12861-bib-0065 Any therapy for advanced chronic disease should consider the patients’ lived experience of that treatment together with its efficacy. Higher quality studies using objective measurements of QoL are needed to determine the benefits of cannabinoids and provide optimal support to patients.
Other outcomes of interest included PS, AEs, and mortality. Very low‐quality evidence did not allow a conclusion on the effects of cannabinoids for PS or mortality, but suggested that the incidence of AEs was unlikely to be related to cannabinoid treatment. It was concerning that only three studies reported on mortality, despite a study finding a significant correlation between severe weight loss and time from cancer diagnosis to death.ref. jcsm12861-bib-0066
Nutritional intake was reported in some studies, but excluded from this review to avoid wrongfully equating it to appetite.ref. jcsm12861-bib-0067 However a cross‐sectional observational study finding a significant association between malnutrition and QoL in cancer patientsref. jcsm12861-bib-0064 suggested that it may be a more robust measure of appetite. Other measures include endocrine markers of appetite such as insulin and ghrelin, which were shown to be influenced by cannabis in healthy cannabis users.ref. jcsm12861-bib-0068 Together with nutritional intake, they may offer a more objective alternative to measure the orexigenic potential of cannabinoids in CAC.
Strengths
A thorough search strategy was implemented across three databases without any limits on study design, date, language, or publication status. Compared with previous published work, this review used the most recent evidence available from both RCTs and NRSIs across more outcomes. A meta‐analysis was conducted which challenged or confirmed previous findings from other meta‐analyses.ref. jcsm12861-bib-0016, ref. jcsm12861-bib-0027 An assessment of the quality of evidence was performed to distinguish the importance of outcomes and minimize bias towards positive results from low quality evidence.
Limitations
The Cochrane Library was not searched because previous work by Wang et al.,ref. jcsm12861-bib-0016 which this review only aimed to extend, had already examined the collection. As the largest collection of medical literatures, this remains a limitation which could add value to the present study.
The identification of studies was limited by the absence of a clear definition of cachexia and lack of standardized treatment, so that cachexia was not always clearly recognized in the study populations. In the context of this review, a research project submitted to University College London, which did not allow for a double screening, only one investigator conducted the first screening. This is a major limitation implying that some relevant studies may have been excluded or missed.
The majority of the available evidence consisted of observational reports lacking a comparison group or a robust methodology, most likely due to the measurement of subjective outcomes. The lack of methodological homogeneity, expected from the inclusion of NRSI, could only be minimized where a meta‐analysis was possible. More than half the studies included were unmasked and at high risk of selection of bias, which was reflected in very low‐quality ratings of the evidence overall. The low number of studies did not permit an evaluation of publication bias via a funnel plot.
Other limitations included short study duration and small sample size undermine the informative value of this review for clinical decision makers.
Conclusion
With no high‐quality evidence, no recommendations can be made to support the use of cannabinoids alone to improve symptoms and outcomes in CAC patients. Multi‐modal therapies integrating cannabinoids alongside other treatment strategies may have a greater potential. Without better data, identifying and investigating other nutritional and pharmacological interventions may provide more conclusive findings.
To our knowledge, our systematic review is the first to include NRSI in its method and analysis. With respect to study design, this review uniquely shows that lower quality of evidence exists in both randomized and non‐randomized studies, and that data from non‐randomized studies bring valuable information to that of RCTs. This method proved particularly useful to address a complex, challenging aspect of cancer like cachexia. Future research adapting data from both RCT and NRSI will be essential to identify interventions that address both the pathophysiological and patient‐centred aspects of cachexia, including nutritional status, appetite, and quality of life.
Conflict of interest
The authors declare no potential conflicts of interest.
Funding
The submission charges were funded by UCL Library.
Ethics statement
The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia and Muscle.ref. jcsm12861-bib-0069 This systematic review is based on published data and does not contain sensitive clinical study or patient data.
References
- Cachexia as a common characteristic in multiple chronic disease.. J Cachexia Sarcopenia Muscle, 2019
- Cachexia: a new definition.. Clin Nutr, 2008. [PubMed]
- Definition and classification of cancer cachexia: an international consensus.. Lancet Oncol, 2011. [PubMed]
- Cachexia: new advances in the management of wasting diseases.. J Am Med Dir Assoc, 2008. [PubMed]
- Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort.. J Gerontol A Biol Sci Med Sci, 2006. [PubMed]
- The effectiveness of nutritional interventions in malnutrition and cachexia.. Proc Nutr Soc, 2015. [PubMed]
- Prevalence and clinical impact of cachexia in chronic illness in Europe, USA, and Japan: facts and numbers update 2016: editorial.. J Cachexia Sarcopenia Muscle, 2016. [PubMed]
- 8 Rogers JB , Minteer JF . Cachexia. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2020.
- Evolving classification systems for cancer cachexia: ready for clinical practice?. Support Care Cancer, 2010. [PubMed]
- Orphan disease status of cancer cachexia in the USA and in the European Union: a systematic review.. J Cachexia Sarcopenia Muscle, 2019. [PubMed]
- Determining the prevalence and severity of cancer cachexia in advanced non‐small cell lung cancer and its relationship with chemotherapy outcomes.. Support Care Cancer, 2020. [PubMed]
- Defining and classifying cancer cachexia: a proposal by the SCRINIO Working Group.. JPEN J Parenter Enteral Nutr, 2009. [PubMed]
- Three cachexia phenotypes and the impact of fat‐only loss on survival in FOLFIRINOX therapy for pancreatic cancer.. J Cachexia Sarcopenia Muscle, 2018. [PubMed]
- Advances in cancer cachexia: intersection between affected organs, mediators, and pharmacological interventions.. Biochim Biophys Acta Rev Cancer, 2020. [PubMed]
- Therapeutic strategies against cancer cachexia.. Eur J Transl Myol, 2019. [PubMed]
- Medical cannabinoids for cancer cachexia: a systematic review and meta‐analysis.. Biomed Res Int, 2019
- A new look at an old drug for the treatment of cancer cachexia: megestrol acetate.. Clin Nutr, 2013. [PubMed]
- Megestrol acetate for the treatment of anorexia‐cachexia syndrome.. Cochrane Database Syst Rev, 2005. [PubMed]
- Systematic review of megestrol acetate in the treatment of anorexia‐cachexia syndrome.. J Pain Symptom Manage, 2004. [PubMed]
- Body‐composition changes in patients who gain weight while receiving megestrol acetate.. J Clin Oncol, 1993. [PubMed]
- Megestrol acetate in patients with AIDS‐related cachexia.. Ann Intern Med, 1994. [PubMed]
- Cannabis in cancer care.. Clin Pharmacol Ther, 2015. [PubMed]
- Recent clinical experience with dronabinol.. Pharmacol Biochem Behav, 1991. [PubMed]
- Cannabinoids in health and disease.. Dialogues Clin Neurosci, 2007. [PubMed]
- Is there a legitimate role for the therapeutic use of cannabinoids for symptom management in chronic kidney disease?. J Pain Symptom Manage, 2011. [PubMed]
- Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS.. J Pain Symptom Manage, 1995. [PubMed]
- Systematic review and meta‐analysis of cannabinoids in palliative medicine.. J Cachexia Sarcopenia Muscle, 2018. [PubMed]
- Cannabinoids and cystic fibrosis: A novel approach to etiology and therapy.. J Cannabis Therapeut, 2002
- Cancer Cachexia and Cannabinoids.. Complement Med Res, 1999
- Which clinical studies provide the best evidence? The best RCT still trumps the best observational study.. BMJ (Clinical research ed), 2000
- 31 Schünemann HJ , Higgins JPT , Vist GE , Glasziou P , Akl EA , Skoetz N , et al. Chapter 14: Completing ‘Summary of findings’ tables and grading the certainty of the evidence. In Cochrane Handbook for Systematic Reviews, Vol. 6. Cochrane; 2019.
- 32 Reeves BC , Deeks JJ , Higgins JPT , Shea B , Tugwell P , Wells GA . Chapter 24: Including non‐randomized studies on intervention effects. In Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al., eds. Cochrane Handbook for Systematic Reviews of Interventions version 60. Cochrane; 2019.
- Preferred Reporting Items for Systematic Reviews and Meta‐Analyses: The PRISMA Statement.. PLoS Med, 2009. [PubMed]
- 34 Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al. In Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al., eds. Cochrane Handbook for Systematic Reviews of Interventions, 6th ed. Cochrane; 2019.
- Comparison of Orally Administered Cannabis Extract and Delta‐9‐Tetrahydrocannabinol in Treating Patients With Cancer‐Related Anorexia‐Cachexia Syndrome: A Multicenter, Phase III, Randomized, Double‐Blind, Placebo‐Controlled Clinical Trial From the Cannabis‐In‐Cachexia‐Study‐Group.. J Clin Oncol, 2006. [PubMed]
- 36 Schünemann HJ , Vist GE , Higgins JPT , Santesso N , Deeks JJ , Glasziou P , et al. Chapter 15: Interpreting results and drawing conclusions. In Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al., eds. Cochrane Handbook for Systematic Reviews of Interventions version 60. Cochrane; 2019.
- Interpreting estimates of treatment effects: implications for managed care.. Pharm Therapeut, 2008
- 38 Deeks JJ , Higgins JPT , Altman DG . Chapter 10: Analysing data and undertaking meta‐analyses. In: Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al., eds. Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). Cochrane; 2021. Available from www.training.cochrane.org/handbook
- Safety and Efficacy of Inhaled Synthetic THC/CBD for Improving Physical Functioning and for Modulating Cachexia Progression in Patients With Advanced Cancer and Associated Cachexia 2020. Available from:
- Nabilone Effect on the Attenuation of Anorexia, Nutritional Status and Quality of Life in Lung Cancer Patients 2017. Available from:
- Delta‐9‐tetrahydrocannabinol may palliate altered chemosensory perception in cancer patients: results of a randomized, double‐blind, placebo‐controlled pilot trial.. Ann Oncol, 2011. [PubMed]
- Dronabinol versus megestrol acetate versus combination therapy for cancer‐associated anorexia: a North Central Cancer Treatment Group study.. J Clin Oncol, 2002. [PubMed]
- The effect of nabilone on appetite, nutritional status, and quality of life in lung cancer patients: a randomized, double‐blind clinical trial.. Support Care Cancer, 2018. [PubMed]
- The Effects of Dosage‐Controlled Cannabis Capsules on Cancer‐Related Cachexia and Anorexia Syndrome in Advanced Cancer Patients: Pilot Study.. Integr Cancer Ther, 2019. [PubMed]
- Medical cannabis improves appetite and stabilizes weight in cancer patients.. Support Care Cancer, 2019
- Health‐related quality of life across cancer cachexia stages.. Ann Palliat Med, 2019. [PubMed]
- A phase II study of delta‐9‐tetrahydrocannabinol for appetite stimulation in cancer‐associated anorexia.. J Palliat Care, 1994
- The Efficacy and Tolerability of Long‐Term Use of Dronabinol in Cancer‐Related Anorexia: A Case Series.. J Pain Symptom Manage, 2005. [PubMed]
- The European Organization for Research and Treatment of Cancer QLQ‐C30: a quality‐of‐life instrument for use in international clinical trials in oncology.. J Natl Cancer Inst, 1993. [PubMed]
- The Edmonton Symptom Assessment System 25 years later: past, present, and future developments.. J Pain Symptom Manage, 2017. [PubMed]
- 51 Ballatori E , Roila F , Ruggeri B , Bruno A , Tiberti S , Orio FD , editors. The Uniscale Assessment of Quality of Life: Applications to Oncology. USA: Springer Science+Business Media LLC; 2010.
- Re‐validation and shortening of the Functional Assessment of Anorexia/Cachexia Therapy (FAACT) questionnaire.. Qual Life Res, 2000. [PubMed]
- Long‐term efficacy and safety of dronabinol for acquired immunodeficiency syndrome‐associated anorexia.. J Pain Symptom Manage, 1997. [PubMed]
- Anorexia of aging in long term care: is dronabinol an effective appetite stimulant?‐‐a pilot study.. J Nutr Health Aging, 2007. [PubMed]
- Efficacy and Safety of Appetite‐Stimulating Medications in the Inpatient Setting.. Ann Pharmacother, 2019. [PubMed]
- Endogenous cannabinoid system as a modulator of food intake.. Int J Obes Relat Metab Disord, 2003. [PubMed]
- Cannabinoids and appetite: food craving and food pleasure.. Int Rev Psychiatry, 2009. [PubMed]
- Obesity and cannabis use: results from 2 representative national surveys.. Am J Epidemiol, 2011. [PubMed]
- The impact of cannabis and cannabinoids for medical conditions on health‐related quality of life: A systematic review and meta‐analysis.. Drug Alcohol Depend, 2017. [PubMed]
- Improving Quality of Life With Nabilone During Radiotherapy Treatments for Head and Neck Cancers: A Randomized Double‐Blind Placebo‐Controlled Trial.. Ann Otol Rhinol Laryngol, 2016. [PubMed]
- A comparison of observational studies and randomized, controlled trials.. N Engl J Med, 2000. [PubMed]
- Use of cannabinoids in cancer care: palliative care.. Curr Oncol, 2016. [PubMed]
- The medical necessity for medicinal cannabis: prospective, observational study evaluating the treatment in cancer patients on supportive or palliative care.. Evid Based Complement Alternat Med, 2013. [PubMed]
- Relationship between nutritional status and quality of life in patients with cancer.. Eur J Cancer, 2008. [PubMed]
- Voice of cancer patients (VoCP): Patient perceptions regarding use of marijuana and its derivatives in cancer.. J Clin Oncol, 2018
- Evaluation of nutritional status in advanced metastatic cancer.. Support Care Cancer, 2003. [PubMed]
- Cancer cachexia: a systematic literature review of items and domains associated with involuntary weight loss in cancer.. Crit Rev Oncol Hematol, 2011. [PubMed]
- Effects of oral, smoked, and vaporized cannabis on endocrine pathways related to appetite and metabolism: a randomized, double‐blind, placebo‐controlled, human laboratory study.. Transl Psychiatry, 2020. [PubMed]
- Ethical guidelines for publishing in the Journal of Cachexia, Sarcopenia and Muscle: update 2019.. J Cachexia Sarcopenia Muscle, 2019. [PubMed]
