Neuropharmacology of Neuropathic Pain: A Systematic Review
Abstract
Neuropathic pain, a debilitating condition, remains challenging to manage effectively. An insight into neuropharmacological mechanisms is critical for optimizing treatment strategies. This systematic review aims to evaluate the role of neuropharmacological agents based on their efficacy, involved neurotransmitters, and receptors. A manual literature search was undertaken in PubMed including Medline, Cochrane Library, Google Scholar, Plos One, Science Direct, and clinicaltrials.gov from 2013 until 2023. Out of the 13 included studies, seven evaluated the role of gabapentinoids. Two main drugs from this group, gabapentin and pregabalin, function by binding voltage-gated calcium channels, lowering neuronal hyperexcitability and pain signal transmission, thereby relieving neuropathic pain. Four of the pooled studies reported the use of tricyclic antidepressants (TCAs) including amitriptyline and nortriptyline which work by blocking the reuptake of norepinephrine and serotonin, their increased concentration is thought to be central to their analgesic effect. Three articles assessed the use of serotonin-norepinephrine reuptake inhibitors (SNRIs) and reported them as effective as the TCAs in managing neuropathic pain. They work by augmenting serotonin and norepinephrine. Three studies focused on the use of selective serotonin reuptake inhibitors (SSRIs), modulating their effect by increasing serotonin levels; however, they were reported as not a highly effective treatment option for neuropathic pain. One of the studies outlined the use of cannabinoids for neuropathic pain by binding to cannabinoid receptors with only mild adverse effects. It is concluded that gabapentinoids, TCAs, and SNRIs were reported as the most effective therapy for neuropathic pain; however, for trigeminal neuralgia, anticonvulsants like carbamazepine were considered the most effective. Opioids were considered second-line drugs for neuropathic pain as they come with adverse effects and a risk of dependence. Ongoing research is exploring novel drugs like ion channels and agents modulating pain pathways for neuropathic pain management. Our review hopes to inspire further research into patient stratification by their physiology, aiding quicker and more accurate management of neuropathic pain while minimizing inadvertent side effects.
Article type: Review Article
Keywords: analgesics, nerve pain, neuropathic pain, neuropharmacology, neurotransmitters, pain modulation, peripheral neuropathy, pharmacological treatments, prisma, receptors
License: Copyright © 2024, Mian et al. CC BY 4.0 This is an open access article distributed under the terms of the Creative Commons Attribution License CC-BY 4.0., which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Article links: DOI: 10.7759/cureus.69028 | PubMed: 39385859 | PMC: PMC11464095
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.3 MB)
Introduction and background
Pathology affecting the nervous system can present long-term consequences in the form of pain, termed neuralgia, neuropathy, or neuropathic pain. Neuropathic pain was defined by the 2011 International Association for the Study of Pain as "pain caused by a lesion or disease of the somatosensory system" [ref. 1]. Neuropathic pain originates from a dysfunctional and maladaptive response of the nervous system to any form of “damage.” Nerve injury triggers a sequence of changes that lead to the degeneration and regeneration of damaged nerve endings, followed by sensitization phenomena occurring either at peripheral or central sites [ref. 2]. Not all neuropathic pains are alike. Neuropathic pain can vary in symptoms and its subsequent underlying mechanisms, even when caused by the same condition. Patient stratification, according to the underlying pathophysiology, can greatly enhance specific treatment modifications, which will ultimately lead to better patient outcomes [ref. 3]. Neuropathic pain often involves a combination of sensory loss (numbness) and sensory gain (allodynia), with the specific manifestations differing among individuals and conditions. These variations may indicate distinct pain mechanisms at play within each person, potentially offering insights into their treatment responses [ref. 4].
Neuropathic pain is typically classified based on the underlying cause of nerve injury. While there are numerous potential causes, some of the more common ones include diabetes, leading to painful diabetic neuropathy (PDN), shingles resulting in post-herpetic neuralgia (PHN), amputation causing stump and phantom limb pain, as well as neuropathic pain following surgery or trauma, stroke, spinal cord injury, trigeminal neuralgia, and HIV infection. In some cases, the cause remains unidentified [ref. 2].
People suffering from neuropathic pain often experience significant disability, especially after enduring moderate to severe pain for many years [ref. 5]. This leads to a significant decline in quality of life, loss of employment, and higher healthcare expenses [ref. 6]. Neuropathic pain poses significant treatment challenges, as only a small percentage of individuals experience meaningful benefits from any single intervention. A multidisciplinary approach is now recommended, integrating pharmacological treatments with physical or cognitive therapies or both [ref. 2].
This study aims to understand the pharmacological agents that improve neuropathic pain and presents a systematic review of literature on a variety of drugs, including antidepressants, anticonvulsants, opioids, cannabis, and emerging treatments targeting specific receptors and neurotransmitters. It will try to give an updated answer to the question: “Which pharmacological agents play a role in improving neuropathic pain, and which neurotransmitters and receptors are involved?” We hope to encourage treatment particularization for the different patient strata by reviewing recent research data.
Review
Methods
This systematic review follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to guarantee strict methodology.
Registration
This systematic review has been registered on PROSPERO, registration number CRD42024578582.
Databases and Search Strategy
A manual literature search was conducted in PubMed including Medline, Cochrane Library, Google Scholar, Plos One, Science Direct, and clinicaltrials.gov from 2013 until 2023. The last search date was 31 December 2023. The complete search strategy and selection of studies are depicted in the PRISMA flowchart in Figure 1.
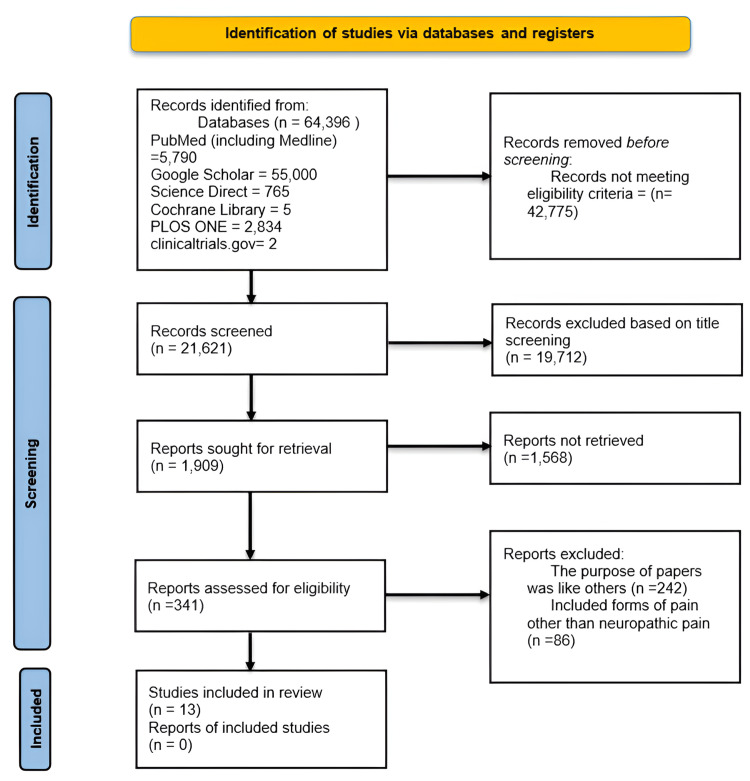
The databases and search methods are presented in Table 1 as follows:
Table 1: Search strategies of the databases searched along with the number of results before and after application of filters
| Database | Search Strategy | Results before Filters | Filters Applied | Results after Filters |
| PubMed (Including MEDLINE) | (((pharmacology[MeSH Subheading]) OR (neuropathic pain[MeSH Subheading])) AND (("2013/11/20"[Date – MeSH] : "3000"[Date – MeSH]))) AND (neuropathic pain[Title/Abstract]) | 5790 (2023/11/20) | Human Studies; Free full text; ARTICLE TYPE: •Clinical Trial •Meta-Analysis •Randomized Controlled Trial •Systematic Review ; PUBLICATION DATE: 2013/05/02 – 2023/11/20 ; SPECIES: Humans ; ARTICLE LANGUAGE: English ; SEX: • Female • Male ; AGE: Adult: >18 years ; OTHER: MEDLINE | 134 |
| Google Scholar | Neuropharmacology of Neuropathic Pain | 55,000 (2023/11/29) | From 2013 to 2023 | 19,200 Analyzed first 20 pages only. |
| Cochrane Central Register for Controlled Trials (Including Embase) | Neuropharmacology of Neuropathic Pain | 5 (2023/11/29) | From 2013 to 2023 English only | 3 |
| Plos One | Neuropharmacology of Neuropathic Pain | 2,834 (2023/12/31) | 2013/05/02 – 2023/11/20 | 2,203 |
| Science Direct | Neuropharmacology of Neuropathic Pain | 765 (2023/12/15) | Open Access & Open Archives 2013 to 2023 | 79 |
| Clinical Trials.gov | Condition: Neuropathic Pain Other terms: Chronic Pain Intervention: Neuropharmacology | 2 (2023/12/15) | 2013/05/02 – 2023/11/20 | 2 |
Eligibility Criteria
Studies were screened based on their respective types: randomized controlled trials (RCTs), systematic reviews, meta-analyses, and observational studies. The inclusion criteria included studies conducted on human adults, published in the last 10 years (2013 to 2023), and written in English. In contrast, non-English studies, studies with unavailable full texts or incomplete outcome data, studies involving pediatric populations, and studies published before 2013 were excluded. A detailed description of the inclusion and exclusion criteria is given in Table 2.
Table 2: Complete inclusion and exclusion criteria
| Inclusion Criteria | Exclusion Criteria | |
| a) | Human Studies | Animal Studies |
| b) | From 2013 to 2023 | Before 2013 |
| c) | English Texts | Non-English Texts |
| d) | Gender: All | |
| e) | Age: >18 years old | Age: ≤ 18 years old |
| f) | Free Papers | Papers that needed to be purchased |
Quality Appraisal
We used standardized quality assessment techniques to rigorously evaluate the quality of 13 selected studies. The Cochrane Risk of Bias tool for randomized controlled trials (RCTs), the SANRA (Scale for the Assessment of Non-Systematic Review Articles) tool for observational studies, and the AMSTAR (A Measurement Tool to Assess Systematic Reviews) checklist for systematic reviews were used to evaluate the quality of the studies as elucidated in Table 3 below. Three reviewers independently conducted the quality assessment. Any and all disputes were resolved through mutual discussion. A fourth reviewer was consulted if the need arose.
Table 3: AMSTAR: A Measurement Tool to Assess Systematic Reviews; SANRA: Scale for the Assessment of Narrative Review Articles.
| Quality Assessment Tool | Study Type |
| Cochrane Risk of Bias Assessment Tool [ref. 8] | Randomized Controlled Trials |
| AMSTAR Checklist [ref. 9] | Systematic Reviews and Meta-Analysis |
| SANRA Checklist [ref. 10] | Any other without a Clear methods section |
Figure 2, Table 4, and Table 5 present the specific overall scores for risks of bias for every study that was chosen.
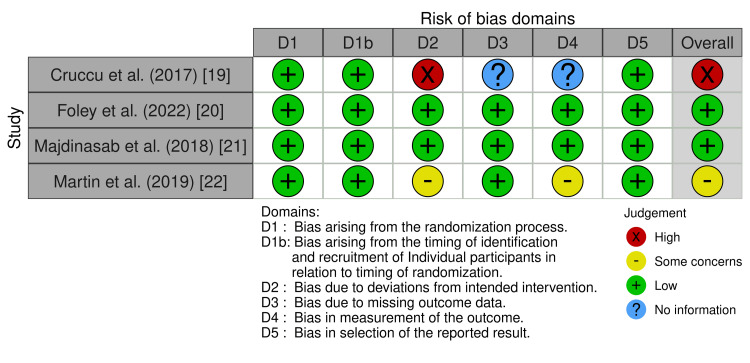
Table 4: PICO: Population/Patient Intervention Comparison Outcome; AMSTAR: A Measurement Tool to Assess Systematic Reviews [ref. 9].
| AMSTAR | ||||
| Wiffen et al. (2013) [ref. 16] | Chen et al. (2022) [ref. 17] | Wiffen et al. (2016) [ref. 2] | Bennici et al. (2021) [ref. 18] | |
| Did the research questions and inclusion criteria for the review include the components of PICO? | Yes | Yes | No | Yes |
| Did the report of the review contain an explicit statement that the review methods were established prior to the conduct of the review and did the report justify any significant deviations from the protocol? | Partial Yes | Yes | Partial Yes | Partial Yes |
| Did the review authors explain their selection of the study designs for inclusion in the review? | Yes | Yes | Yes | Yes |
| Did the review authors use a comprehensive literature search strategy? | Yes | Yes | Partial Yes | Partial Yes |
| Did the review authors perform study selection in duplicate? | Yes | Yes | Yes | Yes |
| Did the review authors perform data extraction in duplicate? | Yes | Yes | No | No |
| Did the review authors provide a list of excluded studies and justify the exclusions? | No | No | Yes | Yes |
| Did the review authors describe the included studies in adequate detail? | Yes | No | No | Yes |
| Did the review authors use a satisfactory technique for assessing the risk of bias (RoB) in individual studies that were included in the review? | Yes | Yes | No | Yes |
| Did the review authors report on the sources of funding for the studies included in the review? | No | No | Yes | No |
| If meta-analysis was performed did the review authors use appropriate methods for statistical combination of results? | No | Yes | No | No |
| If meta-analysis was performed, did the review authors assess the potential impact of RoB in individual studies on the results of the meta-analysis or other evidence synthesis? | No | Yes | No | No |
| Did the review authors account for RoB in individual studies when interpreting/ discussing the results of the review? | Yes | Yes | Yes | Yes |
| Did the review authors provide a satisfactory explanation for, and discussion of, any heterogeneity observed in the results of the review? | Yes | Yes | No | Yes |
| If they performed quantitative synthesis did the review authors carry out an adequate investigation of publication bias (small study bias) and discuss its likely impact on the results of the review? | Yes | Yes | No | No |
| Did the review authors report any potential sources of conflict of interest, including any funding they received for conducting the review? | Yes | Yes | Yes | No |
Table 5: SANRA= Scale for the Assessment of Non-Systematic Review Articles [ref. 10].
| SANRA | |||||
| Fornasari et al. (2017) [ref. 11] | Kersten et al. (2015) [ref. 12] | Obata et al. (2017) [ref. 13] | Alles et al. (2018) [ref. 14] | Gambeta et al. (2020) [ref. 15] | |
| Justification of the article’s importance for the readership | 2 | 2 | 2 | 2 | 2 |
| Statement of concrete aims or formulation of questions | 2 | 2 | 2 | 2 | 2 |
| Description of the literature search | 0 | 0 | 2 | 0 | 0 |
| Referencing | 2 | 2 | 2 | 2 | 2 |
| Scientific reasoning | 2 | 1 | 2 | 1 | 2 |
| Appropriate presentation of data | 1 | 2 | 2 | 2 | 2 |
| Total Score | 9 | 9 | 12 | 9 | 10 |
Results
Three authors worked on the data extraction and compilation of subsequent results. We extracted data from the 13 studies included in our article. The results are presented in Table 6 and discussed below.
Table 6: Key findings and characteristics of all included studies
| Study Title | Study Design | Pharmacological Agents | Neurotransmitters | Receptors | Participants Included | Key Findings |
| Alles and Smith (2018) [ref. 14] | Narrative Review | Gabapentinoids, Opioids, TCAs, SNRIs, Sodium channel blockers, Potassium channel openers, Calcium channel blockers, Hyperpolarization-Activated Cyclic Nucleotide–Gated Channels Modulators, Transient Receptor Potential (TRP) Channel Modulators, Adenosine A3 Receptor Agonists, Colony-Stimulating Factor 1 Inhibitors, Purinergic Ionotropic 2X4 (P2X4) Receptor Antagonists, Brain-Derived Neurotrophic Factor (BDNF) Pathway Modulators | Norepinephrine, Serotonin, Glutamate, Substance P | α2δ-1 subunit of voltage-gated Ca2+ channels, Kv7.2 and Kv7.3 subunits, TRPV, Adenosine A31, P2X4 | Not Specified | Gabapentinoids are first-line treatments; antidepressants and opioids are also used but with varying efficacy. Research is exploring new drug targets including specific ion channels, receptors (TRPV1, adenosine A3, P2X4), and pathways involved in pain modulation. |
| Chen and Li (2022) [ref. 17] | Network Meta-Analysis and Systematic Review | Gabapentin, pregabalin, fluoxetine, lamotrigine, duloxetine, sertraline, amitriptyline, carbamazepine, vitamin B | Adrenaline, 5-hydroxytryptamine (5-HT), gamma-aminobutyric acid (GABA), glutamate | Not specified | 1040 | Gabapentin and pregabalin showed the most significant effects in relieving central poststroke pain. [Gabapentin > pregabalin > fluoxetine > lamotrigine > duloxetine > serqulin > amitriptyline > carbamazepine > vitamin B] However, study limitations, including small sample size and lack of proper RCTs, warrant further investigation. |
| Cruccu et al. (2018) [ref. 19] | Randomized Controlled Trial | Capsaicin 8% patch, pregabalin | Not specified | Low-threshold, mechano-sensitive C fibers (C tactile fibers). Capsaicin is a potent, highly selective vanilloid receptor subtype 1 (TRPV1) agonist | 488 | Capsaicin 8% patch was superior to pregabalin in relieving dynamic mechanical allodynia and associated with fewer adverse events. |
| Foley et al. (2022) [ref. 20] | Randomized Controlled Trial | Fluoxetine, Riluzole, Amiloride | Glutamatergic neurotransmission depression | Central receptors: Riluzole: Na- Channel inactivator Amiloride: Blocks acid-sensing ion channels (a mechanism of pain) in CNS (Central Nervous System) + PNS (Peripheral Nervous System) | 445 | No statistically significant benefit in neuropathic pain symptoms was identified in participants receiving fluoxetine, riluzole, or amiloride compared to placebo. |
| Fornasari D (2017) [ref. 11] | Narrative Review | TCAs, SNRIs, gabapentinoids, lidocaine, opioids | Serotonin, norepinephrine (NE) | Alpha-1 adrenergic receptors, NMDA receptors, calcium channels, Na-Channels, seven transmembrane G protein-coupled receptors, which are divided into three classes: mu, delta, and kappa receptors. Coupling of opioid receptors to calcium and potassium channels is thought to be a central mechanism of analgesia. | Not specified | Antidepressants and anticonvulsants, particularly TCAs and gabapentinoids, are effective first-line treatments for neuropathic pain. Opioids are effective but carry risks of abuse and addiction. Tapentadol is a new class of dual opioid analgesics, which inhibits noradrenaline uptake as well as acts as an agonist at mu opioid receptors. |
| Gambeta et al. (2020) [ref. 15] | Narrative Review | Carbamazepine, Oxcarbazepine, Gabapentin, Pregabalin, Lamotrigine, Phenytoin, Baclofen, Botulinum toxin type A, Eslicarbazepine | Glutamate, | Voltage-gated sodium channels | Not Specified | The first-line treatment for trigeminal neuralgia is pharmacological, with carbamazepine and oxcarbazepine being the most commonly prescribed drugs. These medications work by modulating voltage-gated sodium channels and reducing neuronal excitability. Other pharmacological agents, such as gabapentin, pregabalin, and botulinum toxin type A, can also be used. Surgical options are available for patients who do not respond to medication or experience intolerable side effects. |
| Kersten et al. (2015) [ref. 12] | Case Series | Cetuximab | Not specified | EGFR, MAPK, human epidermal growth factor receptor (HER), Na-Channels | 20 | EGFR inhibition with cetuximab effectively relieved neuropathic pain with minimal adverse events. |
| Majdinasab et al. (2019) [ref. 21] | Randomized Controlled Trial | Duloxetine, gabapentin | Serotonin, norepinephrine, GABA | Not specified | 104 | Both duloxetine and gabapentin significantly improved pain scores over time, with duloxetine associated with fewer side effects. |
| Martin et al. (2019) [ref. 22] | Randomized Controlled Trial | Ketamine, Dextromethorphan, memantine | Not specified | NMDA receptors | 60 | Oral dextromethorphan temporarily extended ketamine pain relief and improved cognitive-affective dimensions in neuropathic pain patients. |
| Bennici et al. (2021) [ref. 18] | Systematic Review | Tetrahydrocannabinol (THC), Cannabidiol (CBD) | Dopamine | CB1 & CB2 (Cannabinoid receptor 1 & 2) | 1080 | Cannabis (mainly a combination of THC and CBD) caused reduction in pain intensity and spasticity in patients with neuropathic pain due to fibromyalgia, radiculopathy, chronic non-cancerous pain, spinal cord cancer pain, diabetic neuropathy (DN), complex regional pain syndrome, pelvic neuropathic pain, spinal cord injury pain, multiple sclerosis, vertebral artery occlusion, arachnoid cysts, syringomyelia, and HIV-associated distal sensory predominant polyneuropathy.. Dizziness, headache, nausea, mild neurocognitive impairment, sedation, and “drug high” are common, while severe adverse events are rare. |
| Obata H (2017) [ref. 13] | Narrative Review | TCAs, SNRIs | Noradrenaline, serotonin (5-HT) | Alpha-2 adrenergic receptors | Not specified | Antidepressants inhibit neuropathic pain by increasing noradrenaline in the spinal cord and acting on the locus coeruleus. TCAs have stronger analgesic effects than drugs that selectively inhibit reuptake of only one neurotransmitter. |
| Wiffen et al. (2013) [ref. 16] | Systematic Review | Gabapentin, pregabalin, carbamazepine, lamotrigine, oxcarbazepine, topiramate, lacosamide, clonazepam, phenytoin, valproic acid | Not specified | Alpha-2 delta calcium channels, sodium channels, NMDA receptors | Not specified | Gabapentin and pregabalin showed reasonably good evidence for efficacy in painful diabetic neuropathy and postherpetic neuralgia, with a modest effect size. Other antiepileptic drugs had mixed or inconclusive evidence for neuropathic pain. |
| Wiffen et al. (2016) [ref. 2] | Systematic Review | Paracetamol/acetaminophen | Not specified | Central receptors | Not specified | No studies meeting inclusion criteria; evidence supporting the use of paracetamol for neuropathic pain is lacking. |
Gabapentinoids
Seven of the included studies evaluated the role of gabapentinoids in managing neuropathic pain [ref. 11,ref. 14–ref. 17,ref. 19,ref. 21]. They are considered the first-line treatments for various conditions causing neuropathic pain. Gabapentin and pregabalin, the two main drugs in this class, have shown efficacy in relieving central poststroke pain, painful diabetic neuropathy, and postherpetic neuralgia [ref. 17]. Their effects are modulated via binding of the α2δ-1 subunit of voltage-gated calcium channels, distributed throughout the nervous system. Their binding causes the release of excitatory neurotransmitters, including glutamate, substance P, and norepinephrine, reducing neuronal hyperexcitability and pain signal transmission [ref. 21].
Tricyclic Antidepressants (TCAs)
Four of the pooled studies examined the use of TCAs for treating neuropathic pain [ref. 11,ref. 13,ref. 14,ref. 17]. Of these studies, three identified TCAs as a first-line treatment [ref. 11,ref. 13,ref. 17], while one suggested they may come as a secondary choice given their potential side effects [ref. 14]. Among TCAs, amitriptyline and nortriptyline are the most extensively studied antidepressants for neuropathic pain. These medications have demonstrated efficacy in treating various neuropathic pain conditions, including peripheral neuropathy, postherpetic neuralgia, and pain following spinal cord injury. TCAs work primarily by blocking the reuptake of norepinephrine and serotonin in the central nervous system. The increase in noradrenaline, in particular, is thought to be central to their analgesic effects. Despite their effectiveness, TCAs are associated with potential side effects. Nortriptyline is often preferred over amitriptyline due to its milder side effect profile [ref. 13].
Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs)
Four articles reported the use of SNRIs for neuropathic pain [ref. 11,ref. 13,ref. 14,ref. 21]. Venlafaxine, duloxetine, and other SNRIs are antidepressants that have been used for neuropathic pain, and they perform the same as TCAs. Majdinasab et al. showed that they are effective by elevating the concentrations of serotonin and norepinephrine in the central nervous system, which are involved in pain regulation [ref. 21]. Alles and Smith have described SNRIs as being among the first-line management options in the same way as TCAs [ref. 14]. However, Obata H pointed out that even though SNRIs are effective, they might not be as effective as TCAs in the treatment of neuropathic pain, but they are better tolerated than the latter [ref. 13].
Opioids
Two studies report the effects of opioids on neuropathic pain [ref. 11,ref. 14]. Endogenous opioids work by using the mu, delta, and kappa receptors, which subsequently upregulate the inhibitor G protein, thus inhibiting the adenylate cyclase along with the cAMP. However, mechanisms common to both endogenous and exogenous opioids include their coupling to the calcium and potassium channels, contributing to their role as central analgesics. However, Fornasari et al. have tagged opioids as second and third-line agents when it comes to neuropathic pain. A major factor for this nomination is their poor side effect profile, including but not limited to addiction, diversion, and abuse potential [ref. 11]. Alles and Smith have categorized opioids as second-line agents in neuropathic pain due to the loss of u-opioid receptors in any kind of nerve injury [ref. 14]. Furthermore, they have extended the debate to include that any form of pain that is resistant to opioid use is likely neuropathic in nature due to the same mechanism [ref. 14].
Anticonvulsants
Besides gabapentinoids, other anticonvulsants like carbamazepine and oxcarbazepine play a crucial role in managing neuropathic pain, particularly trigeminal neuralgia [ref. 15]. Three of the included studies reported findings on anticonvulsants [ref. 15–ref. 17]. These medications primarily act by modulating voltage-gated sodium channels, reducing the excessive firing of neurons associated with this condition. By stabilizing neuronal membranes and inhibiting repetitive action potentials, they effectively alleviate pain. However, potential side effects include somnolence, dizziness, and drowsiness [ref. 16,ref. 17].
Selective Serotonin Reuptake Inhibitors (SSRIs)
Three articles focused on the application of SSRIs for neuropathic pain, among which fluoxetine was the most common [ref. 11,ref. 13,ref. 20]. These drugs are known to help by increasing serotonin levels in the brain. Foley et al. demonstrated that while serotonin is widely associated with mood regulation since it is a neurotransmitter, it also influences pain transmission by altering pain channels, possibly resulting in the relief of pain [ref. 20]. However, Obata et al. indicated that often, SSRIs are not regarded as a very effective modality in managing chronic neuropathic pain [ref. 13].
Cannabinoids
Bennici et al. mention the use of cannabis and cannabinoids for neuropathic pain relief, the review mentions that nabiximoles, products of cannabidiol and delta-9-tetrahydrocannabinol (THC), are approved for neuropathic pain and spasticity due to multiple sclerosis in certain regions [ref. 18]. Cannabinoids (CB) work by activating two receptors within the endocannabinoid system: cannabinoid receptor type 1 (CB1) and type 2 (CB2). The endocannabinoid system (eCS) is a biological network comprising endocannabinoids-endogenous lipid-based retrograde neurotransmitters that bind to cannabinoid receptors and cannabinoid receptor proteins distributed throughout the vertebrate central nervous system. Common side effects include dizziness, headache, nausea, mild neurocognitive impairment, sedation, and a "drug high," while severe adverse events are rare.
N-Methyl-D-Aspartate (NMDA) Receptor Blockers
Martin et al. reported that Ketamine is effective for postoperative pain, post-herpetic neuralgia, and phantom limb pain [ref. 22]. However, due to its poor side effect profile, including hepatic, cardiovascular, and psychodysleptic side effects, other NMDA receptor blockers like dextromethorphan and memantine can be used in its place to prolong ketamine’s analgesic effects [ref. 22]. Another benefit of these alternative options is that they can be administered orally, as opposed to the intravenous route of ketamine, improving medication compliance [ref. 22].
Paracetamol
Wiffen et al. have reported that paracetamol, with or without codeine or hydrocodone, is one of the first-line drugs used in postherpetic neuralgia, painful diabetic neuropathy, neuropathic low back pain, and phantom limb pain. The mechanism of paracetamol is still a point of debate, but it is said to be working by inhibiting the cyclooxygenase (COX) receptors in the brain, spleen, and lungs. However, there is no high-quality evidence supporting or contesting paracetamol’s efficacy in reducing neuropathic pain [ref. 2].
Emerging Therapies
Ongoing research is exploring novel drug targets for neuropathic pain management. These include specific ion channels, such as transient receptor potential vanilloid-1 (TRPV-1) and P2X purinoceptor 4 (P2X4), as well as receptors like adenosine A3. Additionally, pathways involved in pain modulation, such as glial cell activation and inflammatory processes, are being investigated as potential therapeutic targets [ref. 14]. Kersten et al. showed that any neurotransmitter that inhibits the EGFR (Epidermal Growth Factor Receptor) system blocks the MAPK (mitogen-activated protein kinases) pathway, eventually resulting in the inhibition of neuropathic pain. Another proposed mechanism of the EGFR-inhibitor drugs is their effect on the sodium channels, as demonstrated by improved outcomes when used alongside gabapentin and pregabalin in tumor cell lines [ref. 12].
Discussion
Neuropathic pain is challenging to manage and significantly impacts a patient’s quality of life [ref. 23]. Understanding the involvement of receptors and neurotransmitters can help tailor a regimen specific to the patient. Neuropathic pain can be central or peripheral, but regardless of the origin, gabapentinoids, TCAs, and SNRIs are the recommended first-line therapy [ref. 24]. However, conditions like trigeminal neuralgia, a type of peripheral neuropathic pain, do not respond well to these treatments and require carbamazepine or oxcarbazepine as a specific, effective therapy [ref. 25]. Similarly, opioids like tramadol and tapentadol, are the recommended second-line therapy [ref. 24]; however, the first line in intermittent exacerbation of neuropathic pain and cancer-related pain [ref. 26]. The National Institute for Health and Care (NICE) recommends that for managing neuropathic pain pharmacologically, patients should be offered a choice among amitriptyline, duloxetine, gabapentin, or pregabalin as initial treatment options (excluding trigeminal neuralgia). If the first, second, or third medications are ineffective or poorly tolerated, it is recommended to switch the medication [ref. 2]. This difference could be because of variations in underlying disease pathophysiology, differences in nerve involvement, and variability in pain receptors and neurotransmitters involved.
Antidepressants
Antidepressants exert their pharmacological effect by binding to noradrenaline and serotonin receptors, leading to increased levels of serotonin and norepinephrine in the synaptic cleft. According to a review by Obata et al., antidepressants that inhibit the reuptake of both neurotransmitters have stronger analgesic effects than a drug that selectively inhibits only one neurotransmitter. Additionally, norepinephrine plays a greater role than serotonin in the analgesic effect, and the number needed to treat (NNT) for SSRIs is higher than that for TCAs and SNRIs [ref. 13]. The exact mechanism of neuropathic analgesia of antidepressants is still unknown and requires further high-quality research. One proposed mechanism is that these drugs act by exerting their effects on the norepinephrine and serotonin neurotransmitters in the spinal pain pathway. Additional proposed mechanisms involve the modulation of histamine and sodium channels [ref. 27]. P2X4 is an adenosine 5’-triphosphate (ATP) cation channel receptor that plays a role in neuroinflammation. So far, duloxetine is one drug that has been shown to inhibit these receptors, thereby improving neuropathic pain. However, more in-depth research is yet to be seen [ref. 28].
Anticonvulsants
Anticonvulsants such as pregabalin and gabapentin bind to alpha-2-delta calcium channels. Gabapentinoids decrease the calcium channel currents by influencing the number of available calcium channels in the plasma membrane [ref. 29]. These drugs bind the pre-synaptic calcium channels that modulate the release of excitatory neurotransmitters by blocking calcium entry [ref. 30]. Gabapentin has been shown to reduce presynaptic gamma-aminobutyric acid (GABA) release in the locus coeruleus via the GABA-B receptors, which subsequently increases the glutamate neurotransmitter [ref. 31]. Between gabapentin and pregabalin, the latter has a bioavailability of more than 90% in contrast to the 30-60% of gabapentin. In addition, pregabalin has a linear dose-response relationship as it is absorbed in the small intestine as well as the proximal colon as compared to only small intestinal absorption of gabapentin. Admittedly, the absorption of pregabalin significantly decreases by food intake, but that of gabapentin does not [ref. 32]. This difference in pharmacokinetics could be of potential importance to patients with small intestinal absorptive or transit conditions. Active Crohn’s disease, radiation enteritis, hyperthyroidism, and neuroendocrine tumors decrease the small intestinal transit time, thereby potentially decreasing gabapentin absorption [ref. 33]. However, purely malabsorptive conditions like celiac disease do not alter pregabalin absorption [ref. 34]. Lamotrigine has shown efficacy as an adjunct therapy to TCAs, SNRIs, and gabapentinoids for spinal cord injury-induced neuropathic pain and intractable trigeminal neuralgia [ref. 35,ref. 36].
Topical Lidocaine
Topical lidocaine is considered a second-line agent and blocks voltage-gated sodium channels when administered as a patch [ref. 37], with mild local reactions as side effects [ref. 38]. Notedly, topical anesthetics should be distinguished from topical patches, as the latter have major systemic absorption as their primary mechanism, while the former only exert their effects close to the site of injection. Lidocaine can provide local anesthetic effects in the form of medicated plasters, gels, and sprays [ref. 39]. Topical lidocaine can be used as an adjunct in opioid-sparing pain management as it has been shown to be beneficial in post-surgical persistent pain (PSPP), PHN, diabetic peripheral neuropathy (DPN), carpal tunnel syndrome (CTS), chronic lower back pain (CLBP), and osteoarthritis (OA) [ref. 40].
Opioids
Strong opioids like morphine, oxycodone, and hydromorphone, as well as weaker opioids such as tramadol, have shown efficacy in treating neuropathic pain, comparable to antidepressants in terms of the NNT. Despite their effectiveness, opioids are typically considered second or even third-line treatments due to the risk of adverse reactions, potential for abuse, diversion, and addiction [ref. 41]. The analgesic effects of opioids arise from their actions in the brain, brainstem, spinal cord, and, under certain conditions, the peripheral terminals of primary afferent neurons. Endogenous opioid peptides, including β-endorphin, enkephalins, and dynorphins, bind to G protein-coupled receptors, classified into mu, delta, and kappa receptors. These receptors are linked to inhibitory G proteins, and their activation inhibits adenylate cyclase and intracellular cAMP production. A key mechanism of opioid-induced analgesia involves the coupling of these receptors to calcium and potassium channels [ref. 42].
Within the dorsal horn of the spinal cord, mu receptors predominate, with over 70% located pre-synaptically at the central terminals of nociceptors (C and A delta fibers). The remaining 30% are found post-synaptically on dendrites of second-order spinothalamic neurons and interneurons [ref. 42]. These interneurons are primarily responsible for releasing endogenous opioids like beta-enkephalin and endorphins, which act on mu receptors in the dorsal horn. Interneuron activation depends on descending pathway activity or direct stimulation by descending fibers. Activation of presynaptic mu receptors inhibits calcium ion channels, preventing neurotransmitter release, while activation of postsynaptic mu receptors opens potassium ion channels, causing potassium efflux and hyperpolarization of the projecting cell. Thus, stimulating mu-opioid receptors in the spinal cord effectively blocks synaptic transmission, reducing nociceptive stimuli reaching the thalamus and cortex, where pain is consciously perceived [ref. 42]. Despite their efficacy, the long-term use of opioids for nonmalignant pain remains controversial due to concerns about the potential development of tolerance to their analgesic effects, and the risk of addiction [ref. 42].
Riluzole and Amiloride
Riluzole is known to inactivate voltage-dependent sodium channels and depress glutamatergic neurotransmission, offering another potential avenue for pain management. Amiloride, which blocks acid-sensing ion channels implicated in both central and peripheral pain mechanisms, is used in the treatment of migraines. These insights into various drug mechanisms emphasize the need for continued research to enhance our understanding and treatment of neuropathic pain [ref. 20].
Riluzole was initially reported to be of little to no use for treating peripheral neuropathic pain [ref. 43]. However, recent studies have shown improved outcomes for patients experiencing cervical myelopathy who were treated with riluzole versus placebo [ref. 44]. Riluzole provides benefits to patients with Alzheimer’s disease by preserving cerebral glucose metabolism and to ALS patients by improving survival. It has also shown benefits to patients with spinal cord injury (SCI) in decreasing their pain [ref. 45,ref. 46].
Amiloride is a sodium channel-blocking diuretic that is commonly used for congestive heart failure and hypertension. One of its mechanisms of action is to inhibit the acid-sensing ion channels (ASIC). This effect has been shown to improve neuropathic pain by adopting the intrathecal injection approach for acute and chronic pains [ref. 47].
Vitamin B Complex
The Vitamin B complex including thiamine (B1), pyridoxine (B6), folic acid (B9), and cyanocobalamin (B12) have shown improvements in neuropathic pains of various etiologies, specifically diabetic neuropathy. These vitamins have the added advantage of being neuroprotectors of the entire nervous system, where B1 has a site-directed antioxidant property, B6 modulates nerve metabolism, and B12 stabilizes the myelin sheaths, blocks sensory nerve conduction, and decreases ectopic nerve firing. It is worth noting that B12 has a special role in nerve-regenerating activities as well. It has been shown to not only improve neuropathic pain symptoms but also completely regenerate and recover lost myelin sheaths and physiological nerve conduction velocities [ref. 48–ref. 51]. B12 is of particular importance for diabetic patients who are taking metformin, as this drug has been shown to decrease B12 absorption in the body, contributing to increased neuropathic pain [ref. 52].
Cannabinoids
Cannabinoids work by activating the cannabinoid receptors CB1 and CB2. The cannabinoid receptors work by blocking the voltage-gated calcium channels, inhibiting adenylate cyclase, and decreasing the cAMP levels [ref. 53]. Cannabinoids have been shown to have efficacy for improving neuropathic pain but only for the short term. This is due to their shorter efficacy window as well as a multitude of side effects, including neurological and psychiatric disorder development with long-term use [ref. 54]. Limitations to cannabis use for neuropathic pain also include a risk of dependence leading to cannabis use disorder. In addition to dependence, long-term use can also increase the risk of withdrawal symptoms once therapy is ceased, including but not limited to symptoms of anger, aggression, decreased appetite, irritability, restlessness, and insomnia [ref. 55].
Capsaicin
Capsaicin, a natural spice, has been historically used for its topical anesthetic effects. It exerts its effects through the activation of the transient receptor potential vanilloid-1 (TRPV1) on nerve fibers. Capsaicin use avoids the systemic side effects of systemic drugs and works by inducing a momentary discomfort at the application area, followed by a prolonged anesthetic response [ref. 56]. Capsaicin has primarily shown efficacy in treating postherpetic neuropathic pain and HIV neuropathy. The main adverse effects are transient pain and erythema over the application site [ref. 57].
Paracetamol
Paracetamol is one of the most commonly used analgesics and antipyretics. It is postulated to work by the inhibition of the central COX pathway, leading to an enhanced descending serotonergic effect. However, it is distinct from non-steroidal anti-inflammatory drugs (NSAIDs) as it does not have any peripheral anti-inflammatory effects [ref. 58]. Paracetamol is the first-line therapy for patients with OA [ref. 59]. OA has been shown to have a neuropathic pain component, as evidenced by a pathological innervation of the degenerative cartilage in the joint space [ref. 60]. Furthermore, the efficacy of paracetamol is being debated for OA, especially for long-term use. However, it is postulated that paracetamol has good synergistic actions when used as a combination therapy for neuropathic pain [ref. 61]. Paracetamol has shown considerable efficacy when used in combination with opioids for rheumatoid arthritis (RA), OA, and diabetic neuropathy [ref. 58].
Neuropathic pain has multiple etiologies, and it stands to reason that the identification of each of these underlying pathologies and their mechanisms can aid in improved outcomes for this condition. However, drug side effects may also depend on the etiology, for example, drugs with CNS-related side effects may be less tolerated in patients with CNS lesions [ref. 41]. Majdinasab et al. presented a proposal for painful diabetic peripheral polyneuropathy (PDPP). The proposal suggested that other pain-controlling medications, such as TCAs or gabapentin, should be used in the first week. After one week, these medications should be discontinued, and only duloxetine should be used [ref. 21], considering that gabapentin works fast, and duloxetine works late but shows lesser side effects. Although this proposal needs more investigations and further studies, it hints at the importance of combined therapy.
Limitations
Our systematic literature review has limitations. We only included studies that provided free, full text, and paid or closed-access articles were excluded. Our study only includes English-based journals/articles, and this can be perceived as a major limitation as a lot of great research exists in the non-English literature and language, just like any other parameter should not be a barrier towards scientific advancement. Neuropathic pain has several manifestations, and though we tried to include the most common ones, there are still other forms and etiologies of it that were not discussed in this article. Similarly, we tried to include the most common drugs used for neuropathic pain, as well as novel and less common therapies, yet there may be several other therapies that were not discussed. Evidence of efficacy for drugs like paracetamol was lacking in any high-quality information, and evidence for the emerging therapies might still be in their early stages. Further high-quality research is awaited on many of this condition’s avenues.
Conclusions
Neuropathic pain can be caused by a number of underlying conditions. It can present in a variety of symptoms, like numbness, tingling, creeping sensations, sharp, shooting pains, lancinating pains, allodynia, hyperalgesia, hypersensitivity to touch, and burning sensations. Neuropathic pain is usually considered chronic in nature. A big part of this might be due to the fact that it is usually associated with chronic underlying conditions, like diabetes mellitus and HIV, and chronic central and peripheral nervous system diseases, like multiple sclerosis, spinal stenosis, spinal compression, and central post-stroke syndromes.
Regardless of the etiology, neuropathic pain, in general, can be managed on its own with specific treatment options. For decades, a ‘one-size-fits-all’ approach has been used for managing neuropathic pain. Advancements in science urge us to explore better avenues with greater precision to manage this chronic debilitating condition. Given the complex nature of neuropathic pain, future therapeutic strategies may benefit from drug combinations. While the development of new treatments for neuropathic pain faces significant challenges, a deeper understanding of the mechanisms involved offers hope for more effective and targeted therapies. This review is one step toward understanding the basic pathophysiology of each kind of neuropathic pain caused by different underlying etiologies, which may lead to highly customized and tailored regimens for each individual patient. We hope that this review paves the way for further research along these lines so that we can hope to minimize the detrimental effects of this condition on the quality of life of millions of patients.
References
- ED McNicol, MC Ferguson, R Schumann. Methadone for neuropathic pain in adults. Cochrane Database Syst Rev, 2017
- PJ Wiffen, R Knaggs, S Derry, P Cole, T Phillips, RA Moore. Paracetamol (acetaminophen) with or without codeine or dihydrocodeine for neuropathic pain in adults. Cochrane Database of Systematic Reviews, 2016
- SM Helfert, M Reimer, J Höper, R Baron. Individualized pharmacological treatment of neuropathic pain. Clin Pharmacol Ther, 2015. [PubMed]
- CA von Hehn, R Baron, CJ Woolf. Deconstructing the neuropathic pain phenotype to reveal neural mechanisms. Neuron, 2012. [PubMed]
- T Vos, AD Flaxman, M Naghavi. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet, 2012. [PubMed]
- R Andrew, S Derry, RS Taylor, S Straube, CJ Phillips. The costs and consequences of adequately managed chronic non-cancer pain and chronic neuropathic pain. Pain Pract, 2014. [PubMed]
- MJ Page, JE McKenzie, PM Bossuyt. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ, 2021
- JA Sterne, J Savović, MJ Page. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ, 2019
- BJ Shea, BC Reeves, G Wells. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ, 2017
- C Baethge, S Goldbeck-Wood, S Mertens. SANRA—a scale for the quality assessment of narrative review articles. Res Integr Peer Rev, 2019. [PubMed]
- D Fornasari. Pharmacotherapy for neuropathic pain: a review. Pain Ther, 2017. [PubMed]
- C Kersten, MG Cameron, B Laird, S Mjåland. Epidermal growth factor receptor-inhibition (EGFR-I) in the treatment of neuropathic pain. Br J Anaesth, 2015. [PubMed]
- H Obata. Analgesic mechanisms of antidepressants for neuropathic pain. Int J Mol Sci, 2017
- SR Alles, PA Smith. Etiology and pharmacology of neuropathic pain. Pharmacol Rev, 2018. [PubMed]
- E Gambeta, JG Chichorro, GW Zamponi. Trigeminal neuralgia: An overview from pathophysiology to pharmacological treatments. Mol Pain, 2020. [PubMed]
- PJ Wiffen, S Derry, RA Moore. Antiepileptic drugs for neuropathic pain and fibromyalgia – an overview of Cochrane reviews. Cochrane Database Syst Rev, 2013
- KY Chen, RY Li. Efficacy and safety of different antidepressants and anticonvulsants in central poststroke pain: a network meta-analysis and systematic review. PLoS One, 2022
- A Bennici, C Mannucci, F Calapai. Safety of medical cannabis in neuropathic chronic pain management. Molecules, 2021
- G Cruccu, TJ Nurmikko, E Ernault, FK Riaz, WT McBride, M Haanpää. Superiority of capsaicin 8% patch versus oral pregabalin on dynamic mechanical allodynia in patients with peripheral neuropathic pain. Eur J Pain, 2018. [PubMed]
- P Foley, RA Parker, F de Angelis. Efficacy of fluoxetine, riluzole and amiloride in treating neuropathic pain associated with secondary progressive multiple sclerosis. Pre-specified analysis of the MS-SMART double-blind randomised placebo-controlled trial. Mult Scler Relat Disord, 2022. [PubMed]
- N Majdinasab, H Kaveyani, M Azizi. A comparative double-blind randomized study on the effectiveness of duloxetine and gabapentin on painful diabetic peripheral polyneuropathy. Drug Des Devel Ther, 2019
- E Martin, M Sorel, V Morel. Dextromethorphan and memantine after ketamine analgesia: a randomized control trial. Drug Des Devel Ther, 2019
- HM Poole, P Murphy, TJ Nurmikko. Development and preliminary validation of the NePIQoL: a quality-of-life measure for neuropathic pain. J Pain Symptom Manage, 2009. [PubMed]
- M Sumitani, T Sakai, Y Matsuda, H Abe, S Yamaguchi, T Hosokawa, S Fukui. Executive summary of the clinical guidelines of pharmacotherapy for neuropathic pain: second edition by the Japanese society of pain clinicians. J Anesth, 2018. [PubMed]
- NS Kikkeri, S Nagalli. Trigeminal neuralgia. StatPearls. Published Online First: 3 March, 2024
- D Bates, BC Schultheis, MC Hanes. A comprehensive algorithm for management of neuropathic pain. Pain Med, 2019
- Jim Mcmorran, Damian Crowther C, Roger Henderson. Antidepressants for neuropathic pain (antidepressant therapy for neuropathic pain). 2024
- RA Sophocleous, L Ooi, R Sluyter. The P2X4 receptor: cellular and molecular characteristics of a promising neuroinflammatory target. Int J Mol Sci, 2022
- S Farrell, A Sargoy, J King, N Brecha, S Barnes. Acute administration of gabapentin reduces voltage-gated calcium channel current in mammalian retinal ganglion cells. Invest Ophthalmol Vis Sci, 2011
- R Yasaei, S Katta, P Patel, A Saadabadi. Gabapentin. Small Animal Critical Care Medicine, 2024
- T Suto, AL Severino, JC Eisenach, K Hayashida. Gabapentin increases extracellular glutamatergic level in the locus coeruleus via astroglial glutamate transporter-dependent mechanisms. Neuropharmacology, 2014. [PubMed]
- M Chincholkar. Gabapentinoids: pharmacokinetics, pharmacodynamics and considerations for clinical practice. Br J Pain, 2020. [PubMed]
- Z Vinarov, M Abdallah, JA Agundez. Impact of gastrointestinal tract variability on oral drug absorption and pharmacokinetics: an UNGAP review. Eur J Pharm Sci, 2021. [PubMed]
- DE Hanu-Cernat, AN Phipps, JH Raphael. Pregabalin assay in a patient with widespread neuropathic pain and late onset gluten intolerance. Pain Med, 2011. [PubMed]
- Lamotrigine for use in Spinal Cord Injury Induced Neuropathic Pain OR for Intractable Trigeminal Neuralgia. 2024
- N Agarwal, M Joshi. Effectiveness of amitriptyline and lamotrigine in traumatic spinal cord injury-induced neuropathic pain: a randomized longitudinal comparative study. Spinal Cord, 2017. [PubMed]
- E Cavalli, S Mammana, F Nicoletti, P Bramanti, E Mazzon. The neuropathic pain: an overview of the current treatment and future therapeutic approaches. Int J Immunopathol Pharmacol, 2019. [PubMed]
- A Liggieri, M Palladini. Lidocaine 5% patch in the treatment of localized neuropathic pain due to nerve compression. Case reports. SciElo Brazil, 2023
- S Derry, PJ Wiffen, RA Moore, J Quinlan. Topical lidocaine for neuropathic pain in adults. Cochrane Database Syst Rev, 2014
- M Voute, V Morel, G Pickering. Topical lidocaine for chronic pain treatment. Drug Des Devel Ther, 2021
- NB Finnerup, N Attal, S Haroutounian. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol, 2015. [PubMed]
- D Fornasari. Pain pharmacology: focus on opioids. Clin Cases Miner Bone Metab, 2014. [PubMed]
- BS Galer, LL Twilling, J Harle, RS Cluff, E Friedman, MC Rowbotham. Lack of efficacy of riluzole in the treatment of peripheral neuropathic pain conditions. Neurology, 2000. [PubMed]
- Z Ghogawala. Riluzole for cervical myelopathy. JAMA Netw Open, 2024
- DC Matthews, X Mao, K Dowd. Riluzole, a glutamate modulator, slows cerebral glucose metabolism decline in patients with Alzheimer’s disease. Brain, 2021. [PubMed]
- A Daverey, SK Agrawal. Neuroprotective effects of Riluzole and Curcumin in human astrocytes and spinal cord white matter hypoxia. Neurosci Lett, 2020. [PubMed]
- S Jeong, SH Lee, YO Kim, MH Yoon. Antinociceptive effects of amiloride and benzamil in neuropathic pain model rats. J Korean Med Sci, 2013. [PubMed]
- S Baltrusch. The role of neurotropic B vitamins in nerve regeneration. Biomed Res Int, 2021. [PubMed]
- KM Abdelrahman, KV Hackshaw. Nutritional supplements for the treatment of neuropathic pain. Biomedicines, 2021
- T Julian, R Syeed, N Glascow, E Angelopoulou, P Zis. B12 as a treatment for peripheral neuropathic pain: a systematic review. Nutrients, 2020
- S Farah, K Yammine. A systematic review on the efficacy of vitamin B supplementation on diabetic peripheral neuropathy. Nutr Rev, 2022. [PubMed]
- E Sayedali, AE Yalin, S Yalin. Association between metformin and vitamin B12 deficiency in patients with type 2 diabetes. World J Diabetes, 2023. [PubMed]
- RM Campos, AF Aguiar, Y Paes-Colli, PM Trindade, BK Ferreira, RA de Melo Reis, LS Sampaio. Cannabinoid therapeutics in chronic neuropathic pain: from animal research to human treatment. Front Physiol, 2021. [PubMed]
- F Petzke, T Tölle, MA Fitzcharles, W Häuser. Cannabis-based medicines and medical cannabis for chronic neuropathic pain. CNS Drugs, 2022. [PubMed]
- SP Ang, S Sidharthan, W Lai. Cannabinoids as a potential alternative to opioids in the management of various pain subtypes: benefits, limitations, and risks. Pain Ther, 2023. [PubMed]
- JF Peppin, M Pappagallo. Capsaicinoids in the treatment of neuropathic pain: a review. Ther Adv Neurol Disord, 2014. [PubMed]
- P Crawford, Y Xu. Topical capsaicin for treatment of chronic neuropathic pain in adults. Am Fam Physician, 2017
- H Hoshijima, M Hunt, H Nagasaka, T Yaksh. Systematic review of systemic and neuraxial effects of acetaminophen in preclinical models of nociceptive processing. J Pain Res, 2021. [PubMed]
- G Pickering, L Mezouar, H Kechemir, C Ebel-Bitoun. Paracetamol use in patients with osteoarthritis and lower back pain: infodemiology study and observational analysis of electronic medical record data. JMIR Public Health Surveill, 2022
- U Freo, C Ruocco, A Valerio, I Scagnol, E Nisoli. Paracetamol: a review of guideline recommendations. J Clin Med, 2021
- U Freo. Paracetamol for multimodal analgesia. Pain Manag, 2022. [PubMed]
