Neutral CB1 Receptor Antagonists as Pharmacotherapies for Substance Use Disorders: Rationale, Evidence, and Challenge
Abstract
Cannabinoid receptor 1 (CB1R) has been one of the major targets in medication development for treating substance use disorders (SUDs). Early studies indicated that rimonabant, a selective CB1R antagonist with an inverse agonist profile, was highly promising as a therapeutic for SUDs. However, its adverse side effects, such as depression and suicidality, led to its withdrawal from clinical trials worldwide in 2008. Consequently, much research interest shifted to developing neutral CB1R antagonists based on the recognition that rimonabant’s side effects may be related to its inverse agonist profile. In this article, we first review rimonabant’s research background as a potential pharmacotherapy for SUDs. Then, we discuss the possible mechanisms underlying its therapeutic anti-addictive effects versus its adverse effects. Lastly, we discuss the rationale for developing neutral CB1R antagonists as potential treatments for SUDs, the supporting evidence in recent research, and the challenges of this strategy. We conclude that developing neutral CB1R antagonists without inverse agonist profile may represent attractive strategies for the treatment of SUDs.
Article type: Review Article
Keywords: cannabinoid, CB1 receptor, Δ, rimonabant, PIMSR, AM4113, neutral antagonist, inverse agonist, substance use disorders
License: © 2022 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/cells11203262 | PubMed: 36291128 | PMC: PMC9600259
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.0 MB)
1. Introduction
Substance use disorder (SUD), defined as the uncontrollable and persistent use of drugs (including alcohol) despite substantial harm and adverse consequences, is still a severe social and health problem worldwide. SUD-related costs, including those in crimes, loss of productivity and healthcare, exceed $740 billion per year in the Unites States [ref. 1]. In recent years, opioid overdose and SUD-related diseases have increased dramatically with the fatal incidents up to ~50,000 in 2017 in the USA [ref. 2]. Although the United States Food and Drug Administration (FDA) approved several medications such as methadone, buprenorphine, and varenicline for the treatment of opioid or nicotine use disorders [ref. 3,ref. 4,ref. 5], the rate of relapse remains extremely high. Moreover, there is no FDA-approved medication for the treatment of psychostimulant use disorders [ref. 6]. Over the past decades, the cannabinoid receptor 1 (CB1R) has been given much attention as a promising target in medication development for treating SUDs [ref. 7,ref. 8,ref. 9]. The reason for such attention is because of convincing evidence indicating that rimonabant, a selective CB1R antagonist with an inverse agonist profile, is highly effective in reducing drug taking and drug-seeking behavior in experimental animals [ref. 7,ref. 8,ref. 10]. However, the severe adverse effects of rimonabant, such as nausea, emesis, depression, and suicidal tendencies observed in humans have led to its withdrawal from clinical trials worldwide [ref. 11]. Consequently, the US FDA decided not to approve CB1R ligands until better safety and efficacy data become available. In this mini-review article, we first review the rationale and supporting evidence for developing rimonabant and its analogs as promising pharmacotherapies for SUDs, and then discuss the possible mechanisms underlying rimonabant’s therapeutic benefits and unwanted side-effects. Lastly, we discuss the recent research progress and the challenges in developing neutral CB1 receptor antagonists as new pharmacotherapies for SUDs.
2. Mesocorticolimbic Dopamine System
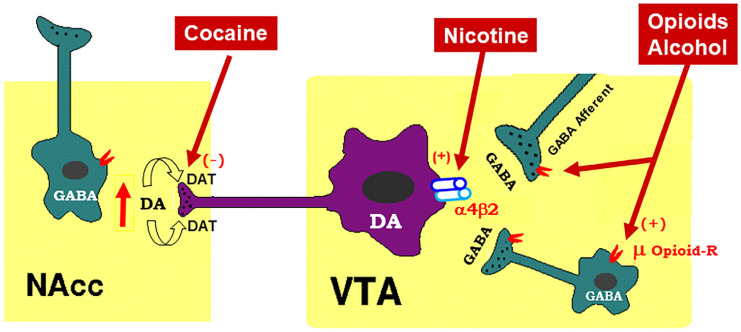
To better understand how cannabinoid CB1R antagonists produce anti-addictive effects, it is necessary to briefly review the current working hypothesis underlying drug reward and addiction. Addiction includes three stages—binge/intoxication, a stage at which an individual consumes an intoxicating substance and experiences its rewarding effects; withdrawal/negative affect, a stage at which an individual experiences a negative emotional state in the absence of the substance; and preoccupation/anticipation, a stage at which subject seeks substances again after a period of abstinence [ref. 12]. Although our understanding of the neural mechanisms underlying each stage of addiction is still not fully understood, a well-accepted view is that the rewarding effects of drugs of abuse are mediated mainly by activation of the mesocorticolimbic dopamine (DA) system. This system originates in DA neurons in the ventral tegmental area (VTA) and substantia nigra pars compacta (SNc) of the midbrain and projects to the prefrontal cortex (PFC), nucleus accumbens (NAc), and the dorsal striatum (SD) [ref. 6,ref. 13]. Different drugs of abuse activate this pathway by distinct receptor and cellular mechanisms [ref. 14,ref. 15,ref. 16] (Figure 1). For example, the psychostimulant cocaine activates this system mainly by blocking the DA transporter (DAT), while nicotine activates VTA DA neurons by stimulating nicotinic receptors located on DA neurons or glutamate neurons that project to DA neurons in the VTA and NAc [ref. 16,ref. 17,ref. 18]. Alcohol’s reinforcement has been associated with processes involving multiple molecular targets, including mu opioid receptors and NMDA receptors [ref. 12,ref. 19,ref. 20,ref. 21]. On the other hand, opioids activate midbrain DA neurons mainly by stimulation of opioid receptors located on GABAergic neurons in the rostromedial tegmentum (RMTg) and substantia nigra pars reticulata (SNr) that project to the VTA and SNc, respectively, causing increases in DA neurons firing and striatal DA release via GABA-mediated disinhibition [ref. 5,ref. 17,ref. 22]. Therefore, both the RMTg-VTA-NAc and SNr-SNc-DS DA pathways play a central role in drug reward and addiction [ref. 14,ref. 22], making the DA system a crucial target in medication development for the treatment of SUDs.
3. Endocannabinoid System
To better understand how CB1R antagonists produce therapeutic effects against drug abuse and addiction and how CB1R inverse agonists produce unwanted side-effects, let us briefly review the endocannabinoid (eCB) system and recent research on how cannabinoids modulate the mesocorticolimbic DA system, a critical action site for drugs of abuse.
The endocannabinoid (eCB) system consists of cannabinoid receptors (CB1Rs, CB2Rs, and others), endocannabinoids [anandamide (AEA) and 2-arachidonoylglycerol (2-AG)], enzymes for endocannabinoid synthesis [N-arachidonoyl phosphatidylethanolamine-phospholipase D (NAPE-PLD), diacylglycerol-lipase (DAG-lipase)] and degradation [fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MGL)], and their putative transport systems [ref. 23,ref. 24]. AEA was the first endocannabinoid discovered by Raphael Mechoulam and his colleagues in 1992 [ref. 25]. AEA is an endogenous CB1R agonist (Ki = 87.7 nM for rCB1; Ki = 239.2 nM for hCB1) and a weak CB2R agonist (Ki = 267.8 nM for rCB2; Ki = 439.5 nM for hCB2) [ref. 26]. The effects of AEA are mediated mainly by activation of CB1Rs and CB2Rs in the brain and periphery. However, AEA levels in the brain are very low in healthy subjects, and it has a very short half-life (~2 min) due to its fast degradation by fatty acid amide hydroxylase (FAAH) [ref. 27]. Therefore, the functional significance of AEA in the brain is largely unclear.
2-AG was the second eCB discovered in the brain [ref. 28]. It is an endogenous agonist of the CB1Rs (Ki = 1180 nM for rCB1; Ki = 3423 nM for hCB1) and CB2Rs (Ki = 1900 nM for rCB2; Ki = 1193 nM for hCB2) [ref. 26]. Unlike AEA, 2-AG is present at relatively high levels in the central nervous system (CNS). Therefore, it is thought to be a major eCB modulating brain function.
There are at least two types of cannabinoid receptors (CB1Rs and CB2Rs) identified in the brain [ref. 23]. The phytocannabinoids (Δ9-THC), synthetic cannabinoids (WIN55,212-2, CP55,940, HU-210), and the endocannabinoids (AEA, 2-AG) all have high binding affinities at both the CB1Rs and CB2Rs [ref. 23]. Cannabinoids may also bind to other putative cannabinoid receptors, such as G protein-coupled receptor 55 (GPR55), transient receptor potential vanilloid 1 (TRPV1) channel, and peroxisome proliferator-activated nuclear receptors (PPARs) [ref. 23]. Accumulative evidence indicates that cannabinoid action is mediated mainly by activation of CB1Rs and CB2Rs [ref. 23].
4. Cannabinoid Reward versus Aversion
Cannabis is the most commonly used substance worldwide as many people find it pleasurable [ref. 29]. However, the findings regarding the rewarding properties of cannabinoids in both humans and experimental animals are conflicting [ref. 23]. Indeed, cannabis use has often been associated with its psychoactive, rewarding effects [ref. 30,ref. 31]. The psychoactive effects of cannabis, combined with the ongoing cannabis legalization in the United States, may well explain why cannabis use is rising in the USA. For instance, from 2002 to 2019, the percentage of adults who reported using cannabis in the past year increased from 7.0 to 15.2% [ref. 32].
However, cannabis enjoyment is not universal, and some individuals report dysphoria, anxiety, and depression after cannabis use [ref. 33,ref. 34]. The increase in cannabis use also raises concerns about possible adverse effects of cannabis use, such as developing the amotivational syndrome [ref. 35,ref. 36], which is defined as “a reduction in the motivation to initiate or persist in goal-directed behavior” [ref. 37]. A series of human functional magnetic resonance imaging (fMRI) studies support these cannabis amotivational effects by evidence that Δ9-THC produces a significant reduction in reward-related brain activity or neural response to reward in healthy adults [ref. 38,ref. 39,ref. 40]. In congruent with these findings, other reports showed that Δ9-THC reduced the likelihood or motivation of reward-related learning and decision-making [ref. 41], dampened neural responses to music [ref. 42], and reduced striatal DA response to reward [ref. 43].
Similar paradoxical effects of cannabinoids have been discovered in non-human primates, as squirrel monkeys self-administer Δ9-THC or endocannabinoids [ref. 44,ref. 45], while other primate species (rhesus, baboon, cynomolgus) fail to demonstrate this behavior [ref. 46,ref. 47,ref. 48]. In rodents, Δ9-THC alone is not self-administered [ref. 49,ref. 50], although the mixture of Δ9-THC and cannabidiol was recently reported to be self-administered by rats [ref. 51,ref. 52]. In conditioned place preference (CPP) test, Δ9-THC typically produces conditioned place aversion [ref. 53,ref. 54], although place preferences have also been reported [ref. 55,ref. 56]. In electrical intracranial self-stimulation (ICSS) experiments, Δ9-THC was initially reported to facilitate electrical ICSS in rats [ref. 56,ref. 57,ref. 58], while other studies found suppression of ICSS in rats and mice [ref. 59,ref. 60,ref. 61,ref. 62,ref. 63]. In optogenetic ICSS (oICSS) maintained by optical stimulation of midbrain DA neurons or glutamate neurons, cannabinoids always produce a reduction in brain-stimulation reward (BSR) in mice, suggesting a reward-attenuating or aversive effect [ref. 64,ref. 65].
Similarly, the findings of cannabinoid action on DA transmission are also conflicting. There are reports indicating that activation of the CB1Rs increases DA neuronal firing in the VTA [ref. 66,ref. 67] and DA release in the NAc in rats [ref. 68,ref. 69,ref. 70,ref. 71]. However, in vitro voltammetry experiments in striatal brain slices demonstrate that the cannabinoids WIN55,212-2 or CP55,940 fail to alter [ref. 72,ref. 73] or produce a reduction in electrical stimulation-induced DA release in the dorsal striatum in guinea pigs, rats and mice [ref. 74,ref. 75]. In vivo microdialysis experiments in freely moving animals indicate that Δ9-THC produces a dose-dependent reduction in NAc DA in mice [ref. 76]. The neural mechanisms underlying such opposite affective and neurochemical effects of cannabinoids are not fully understood.
4.1. GABAergic CB1R Hypothesis of Cannabis Reward
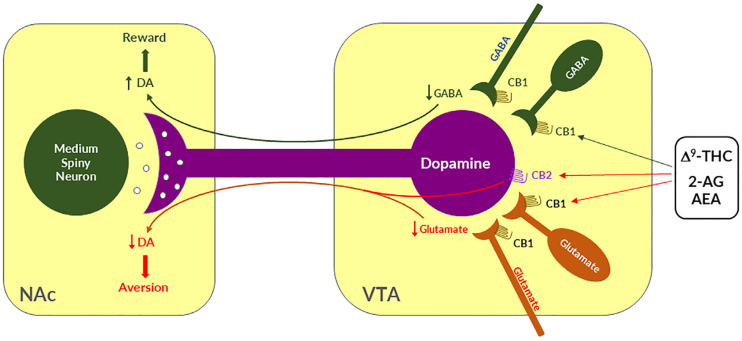
Given that midbrain DA neurons receive both inhibitory GABAergic and excitatory glutamatergic inputs, we proposed that differential CB1R expression on GABAergic neurons versus glutamatergic neurons may underlie cannabinoid reward versus aversion, respectively [ref. 7,ref. 23,ref. 64] (Figure 2). The GABAergic CB1R hypothesis is supported by electrophysiological findings in brain slices where stimulation of CB1Rs on VTA GABAergic neurons causes an increase in VTA DA neuron firing via GABA-mediated disinhibition [ref. 77,ref. 78,ref. 79,ref. 80]. However, so far, there is a lack of behavioral evidence in vivo supporting this GABA-CB1R hypothesis possibly due to the absence of reliable behavioral models of cannabinoid reward in rodents.
4.2. Glutamatergic CB1 Hypothesis of Cannabinoid Aversion
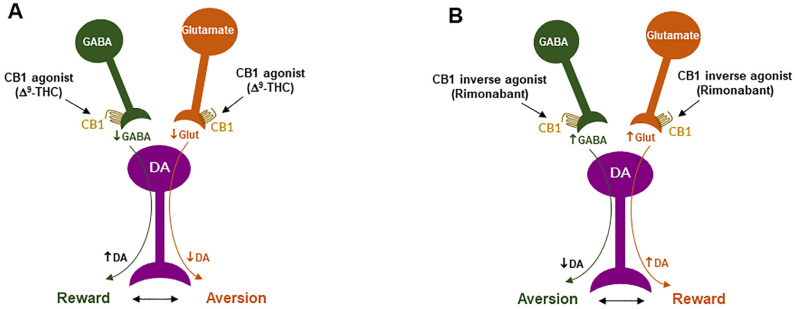
Clearly, the above GABAergic disinhibition hypothesis cannot explain how cannabinoids produce the aversive effects observed in rodents. To address this question, we have recently used advanced RNAscope in situ hybridization (ISH) assays to examine the cellular distributions of CB1Rs. We found that CB1Rs are expressed not only in VTA GABAergic neurons but also in VTA glutamatergic neurons [ref. 23,ref. 64,ref. 65]. Strikingly, optogenetic activation of VTA glutamatergic neurons produced potent rewarding effects, as assessed by CPP and optical ICSS (oICSS) [ref. 64,ref. 81]. Systemic administration of multiple cannabinoids (such as Δ9-THC, WIN55,212-2, ACEA, AM-2201) dose-dependently inhibited glutamate-mediated oICSS only in VgluT2-Cre control mice, but not in glutamate-CB1-knockout mice in which CB1Rs are selectively deleted from subcortical VgluT2-expressing glutamate neurons [ref. 64]. These findings suggest that activation of CB1Rs on glutamate neurons produces reward-attenuation or aversive effects by decreasing glutamatergic inputs onto VTA DA neurons (Figure 2).
The above findings also suggest that activation of brain CB1Rs is not always rewarding but it could be aversive, depending upon the cellular distribution of CB1R expression in the brain. Therefore, we propose that the hedonic effects of cannabis might depend on the balance of two opposing actions of cannabinoids on both GABAergic neurons and glutamatergic neurons (Figure 2). If more CB1Rs are expressed in VTA or VTA-projecting GABAergic neurons, cannabis will be rewarding as GABAergic disinhibition of VTA DA neurons is dominant. In contrast, if more CB1Rs are expressed in VTA or VTA-projecting glutamatergic neurons, cannabis will be aversive as CB1R mediated reduction in glutamatergic inputs onto VTA DA neurons is dominant. Congruently, if CB1R levels are equivalent on both types of neurons, cannabis should have no net effect on the brain reward function (Figure 2). This hypothesis appears to well explain why Δ9-THC or cannabinoids are rewarding in some human subjects and non-human primates (squirrel monkeys) in which more CB1Rs might be expressed in VTA-projecting GABA neurons but are ineffective or aversive in other human subjects and species (such as rats, mice) in which more CB1Rs might be expressed in VTA or VTA-projecting glutamate neurons (Figure 2).
4.3. Dopaminergic CB2 Hypothesis of Cannabinoid Aversion
In addition to the above glutamate-CB1R hypothesis, recent research indicates that CB2Rs are also expressed in VTA DA neurons and contribute to the aversive effects of cannabinoids [ref. 3,ref. 82]. Activation of CB2Rs by JWH133 inhibits VTA DA neuron activity and decreases NAc DA release in wildtype mice, but not in CB2-KO mice [ref. 83,ref. 84,ref. 85,ref. 86]. Activation of CB2Rs or overexpression of brain CB2Rs also inhibits cocaine self-administration, cocaine-induced CPP and hyperactivity in mice [ref. 85,ref. 87,ref. 88,ref. 89]. In rats, Δ9-THC and WIN55, 212-2 produce biphasic effects on electrical brain-stimulation reward (BSR)—reward-enhancing at lower doses and reward-attenuating (or aversive) at higher doses [ref. 63]. CB1R antagonism (by AM251) reduced the low dose-enhanced BSR, while CB2R antagonism (AM630) decreased the high dose-attenuated BSR. Congruently, selective CB1R and CB2R agonists produced significant BSR enhancement and inhibition, respectively [ref. 63]. Together, these findings suggest that DA-CB2R mechanisms, at least in part, underlie cannabinoid-induced aversion (Figure 2) [ref. 3,ref. 23]. Thus, the subjective effects of cannabinoids would depend on the balance of multiple cell type-specific receptor mechanisms. This cell type-specific cannabinoid receptor mechanism appears to well explain why cannabis or cannabinoids could be rewarding, aversive, or ineffective since the cellular distributions of CB1Rs and CB2Rs may be different in different subjects or species.
5. Rimonabant: The First CB1R Antagonist Approved for the Treatment of Obesity
Rimonabant discovery: Extensive studies in the past decades indicate that endocannabinoids are overactive in obese humans [ref. 90,ref. 91,ref. 92] and obese animals in both genetic and diet-induced obesity [ref. 93,ref. 94], which inspired research to develop CB1R antagonists for the treatment of obesity. The initial effort failed in developing a selective CB1R antagonist from modifications of the structure of Δ9-THC until 1994, when Sanofi Pharmaceutical Inc. in France developed rimonabant (also called SR141716A, trade names Acomplia, Zimulti) as the first CB1R antagonist used for the treatment of obesity [ref. 95].
In June 2006, the European Commission approved the sale of rimonabant in the then-25-member European Union as a prescription drug for use in conjunction with diet and exercise for overweight patients with a body mass index (BMI) > 30 kg/m2 or patients with a BMI > 27 kg/m2 with associated risk factors, such as type 2 diabetes or dyslipidemia [ref. 96]. It was the world’s first approved anti-obesity drug in this class. By 2008, rimonabant was available in 56 countries. Data from clinical trials showed that rimonabant had severe adverse effects—causing depressive disorders or mood alterations in up to 10% of subjects and suicidal ideation in around 1%. Other common adverse effects included nausea, vomiting, and upper respiratory tract infections in more than 10% of patients [ref. 97,ref. 98,ref. 99]. Post-marketing surveillance data found that the risk of psychiatric disorders in people taking rimonabant was doubled [ref. 100]. Rimonabant was submitted to the U.S. FDA for approval in 2005. In 2007, the FDA concluded that Sanofi-Aventis failed to demonstrate the safety of rimonabant and voted against recommending the anti-obesity treatment for approval [ref. 101]. Two weeks after FDA’s decision, the company withdrew the application. In October 2008, the European Medicines Agency recommended suspension of clinical use of rimonabant after concluding that its risks outweighed its benefits, and its approval was withdrawn by the European Commission in January 2009 [ref. 100]). As a consequence, clinical trials with rimonabant and almost all other CB1R antagonists with similar inverse agonist profiles such as the longer acting second generation surinabant (SR147778, Sanofi-Aventis), ibipinabant (SLV319, Solvay Pharmaceutical), taranabant (MK-0364, Merck), and otenabant (CP-945,598, Pfizer) [ref. 102] were terminated worldwide in 2008 [ref. 97,ref. 99].
Rimonabant’s failure is a sad and disappointing story in medication development for the treatment of obesity as clinical trial data indicate that rimonabant had significant beneficial effects in controlling body weight and obesity [ref. 103] and significant improvement in glycemic control and lipid profile in type 2 diabetic patients [ref. 104,ref. 105]. Unfortunately, rimonabant’s psychiatric side-effects, such as anxiety, depression, and suicidality [ref. 98], led to its withdrawal from clinical trials and the drug market. However, we should point out that some confounders could be implicated, such as the inclusion of obese patients with a previous history of depression, a fact that could increase the possibility of detecting depressive symptomatology such as suicidal ideation or suicide in some patients treated with rimonabant [ref. 106,ref. 107]. In addition, when we consider rimonabant’s beneficial and adverse effects, it is also important to ask—what are the neural mechanisms underlying its therapeutic anti-obesity effects versus its psychiatric adverse effects?
Pharmacology of rimonabant: Structurally, rimonabant is an aminoalkylindole [ref. 108]. This compound shows high affinity for the CB1R (Ki = 2 nM) but low affinity for the CB2R (Ki > 1000 nM). In vitro, rimonabant antagonizes the inhibitory effects of cannabinoid receptor agonists on both mouse vas deferens contractions and DA-stimulated adenylyl cyclase activities in rat brain membranes. After oral administration, rimonabant inhibited [3H]-CP55,940 binding to cerebral membranes with a median effective dose (ED50) value of 3.5 mg/kg [ref. 108]. Systemic administration (i.v.) of rimonabant inhibited the classical pharmacological effects of Δ9-THC such as hypoactivity, hypothermia, and antinociception in mice [ref. 109]. These data suggest that rimonabant is a selective CB1R antagonist both in vitro and in vivo.
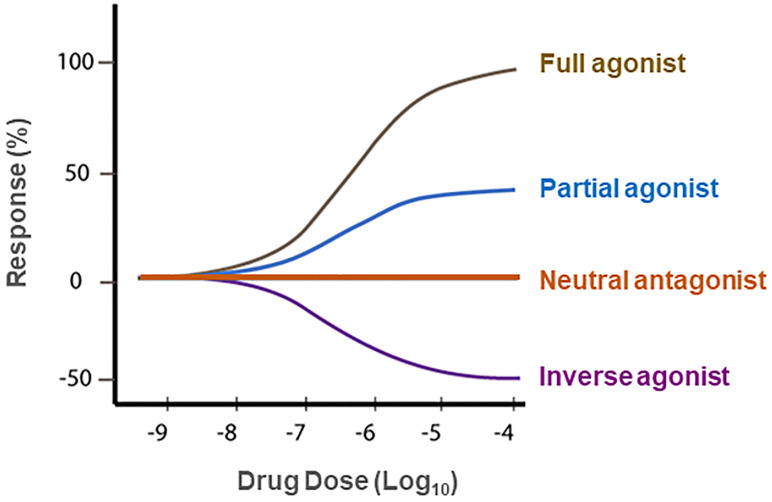
Unexpectedly, it was also reported that rimonabant alone, at high dose (>3 mg/kg), produced increased locomotor activity in mice [ref. 109], an effect that is opposite to that produced by the cannabinoid receptor agonist Δ9-THC [ref. 95]. This finding was supported by observation that intrathecal injection of rimonabant also evoked a significant thermal hyperalgesia in mice, an effect also opposite to cannabinoid agonist-induced analgesia [ref. 110]. Congruently, [35S]GTPγS binding assays in the cell membranes isolated from human CB1R-transfected Chinese hamster ovary (CHO) cells indicate that the cannabinoid agonist WIN 55,212-2 stimulated [35S]GTPγS binding by ~80% above basal levels while rimonabant produced a >20% decrease in basal [35S]GTPγS binding, suggesting that WIN 55,212-2 is a full CB1R agonist while rimonabant is an inverse agonist at CB1Rs [ref. 111] (Figure 3). These data indicate that rimonabant has dual actions—as a CB1R antagonist that blocks the activity of cannabinoid agonists on CB1Rs and as an inverse CB1R agonist by itself, producing an effect opposite to CB1R agonists in the absence of cannabinoids (Figure 3). This unique pharmacological action of rimonabant raised a fundamental question—whether CB1R antagonism or inverse agonism underlies the therapeutic anti-obesity versus the adverse effects of rimonabant. Clearly, understanding the receptor mechanisms underlying the therapeutic effects versus unwanted effects of rimonabant is critically important for successfully developing new generations of CB1R antagonists without significant adverse effects for the treatment of obesity and SUDs.
6. CB1R Antagonists Are Promising for the Treatments of SUDs
Rationale: In a series of clinical trials known as the Studies with Rimonabant and Tobacco Use (STRATUS), it was found that rimonabant significantly increased abstinence rates and reduced smoking cessation-related weight gain [ref. 9,ref. 112,ref. 113,ref. 114,ref. 115], suggesting that the endocannabinoid system may also be involved in nicotine use disorder and CB1R antagonists or inverse agonists may be useful for the treatment of SUDs including nicotine use disorder.
Supporting evidence: The findings with either CB1R agonists or antagonists support this hypothesis of eCB involvement in SUDs. For example, Δ9-THC increases heroin self-administration in rats [ref. 116]. WIN55,212-2 increases motivation to nicotine self-administration and facilitates cue-induced reinstatement of nicotine seeking in rats [ref. 117]. WIN55,212-2 or CP55,940 facilitate alcohol self-administration, CPP, and binge-like behavior in rodents [ref. 118,ref. 119]. Accordingly, it was proposed that CB1R antagonists should also be effective in the treatment of SUDs [ref. 8,ref. 120,ref. 121].
Indeed, compelling preclinical evidence supports this hypothesis. For example, rimonabant reduced intravenous heroin self-administration under fixed-ratio and progressive-ratio reinforcement [ref. 122,ref. 123], nicotine cue-induced reinstatement of nicotine-seeking behavior in rats [ref. 124,ref. 125], and nicotine-enhanced DA release in the NAc [ref. 124]. Furthermore, rimonabant also prevents the development of morphine-induced CPP [ref. 126], and dose-dependently attenuates heroin- or heroin-associated cue-induced reinstatement of drug-seeking behavior [ref. 123]. In addition, rimonabant blocks acquisition of cocaine-induced CPP [ref. 127] and attenuates reinstatement of drug seeking caused by cocaine or cocaine-associated cues [ref. 128]). Congruently, rimonabant also blocks reinstatement of methamphetamine-seeking behavior [ref. 129] and reduces alcohol intake in rodents [ref. 130]. These exciting findings with rimonabant encouraged many pharmaceutical industries to develop other brain-penetrant CB1R antagonists such as the longer acting second generation surinabant (SR147778, Sanofi-Aventis), taranabant (MK-0364, Merck), otenabant (CP-945,598, Pfizer), and ibipinabant (SLV319, Solvay Pharmaceutical), for the treatment of obesity, smoking and drugs of abuse [ref. 102].
Receptor mechanisms: Unfortunately, all the above-mentioned ligands are not only CB1R antagonists but also inverse agonists. Thus, dissecting the role of CB1R antagonism versus inverse agonism in their therapeutic versus side-effects is critical for developing safer CB1R ligands for the treatment of SUDs.
CB1R antagonism may underlie the therapeutic effects of rimonabant: Given that rimonabant is a well-characterized CB1R antagonist, it is reasonable to hypothesize that the CB1R antagonism may underlie its therapeutic anti-obesity and anti-addictive effects. This is supported by several lines of evidence. First, cannabinoids and drugs of abuse may act in a common neural substrate—the mesocorticolimbic DA system via distinct cellular and receptor mechanisms (Figure 2). Assuming that CB1Rs are tonically activated by eCBs (2-AG, AEA), CB1R antagonism on GABA neurons or GABAergic terminals would produce a reduction in eCB-enhanced DA transmission (Figure 2), which may functionally counteract the DA-enhancing and the pro-addictive effects produced by drugs of abuse. Second, growing evidence indicates that drugs of abuse (such as cocaine, heroin, or nicotine) may increase eCB release in the VTA and/or NAc [ref. 131,ref. 132,ref. 133,ref. 134,ref. 135,ref. 136,ref. 137,ref. 138] (Figure 4). After releasing from post-synaptic neurons, such as VTA DA neurons (Wang et al., 2015), eCBs retrogradely diffuse back to activate presynaptic CB1Rs, producing a reduction in neurotransmitter (GABA, glutamate) release, which subsequently produces reward-enhancing and addictive effects (Figure 4). Accordingly, CB1R antagonism at presynaptic terminals would block the eCB-mediated effects, producing anti-addictive effects (Figure 4). Third, the neutral CB1R antagonists without CB1R inverse agonist profile (such as AM4113 and PIMSR) produce the similar anti-addictive effects as rimonabant in drug self-administration and reinstatement, but without rimonabant-like depressive effects [ref. 7,ref. 13,ref. 139]. Lastly, in theory, rimonabant’s anhedonic or aversive effects may also functionally counteract the rewarding effects of drug abuse; however, direct supporting evidence is missing due to the absence of selective CB1R inverse agonists.
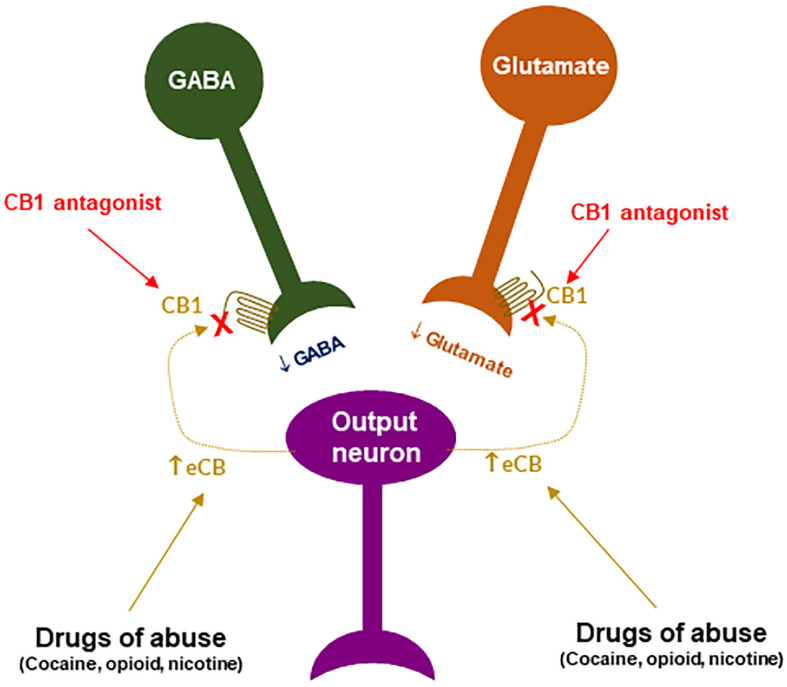
CB1R inverse agonism may underlie the adverse effects of rimonabant: As discussed above, CB1R agonism by Δ9-THC on GABA neurons or GABAergic terminals may produce an increase in NAc DA release and reward-enhancing effects via a disinhibition mechanism (Figure 2 and Figure 5A). Accordingly, CB1R inverse activation by rimonabant on the same receptor would produce opposite effects—enhanced GABA and reduced NAc DA release, which may translate into depressive-like subjective effects of rimonabant (Figure 5B).
Three animal models are often used to evaluate the rewarding versus aversive effects [ref. 140,ref. 141]. They are intracranial self-stimulation (ICSS) (also called brain-stimulation reward, BSR), NAc DA response to test drugs, and conditioned place preference/aversion (CPP/CPA). The findings with rimonabant from these models are mixed (Table 1). In the ICSS model, we [ref. 139] and others [ref. 142] previously reported that high-dose rimonabant inhibits electrical ICSS, while three other reports [ref. 143,ref. 144,ref. 145] indicate that rimonabant has no effect on ICSS. With respect to DA response to rimonabant, we have previously reported that, in mice, systemic administration of rimonabant produces a reduction in extracellular DA in the NAc [ref. 146]. However, in Long-Evan rats, we found that systemic rimonabant failed to alter extracellular NAc DA, while intra-NAc local perfusion of rimonabant at 100 µM (but not at 0.1, 1.0, or 10.0 µM) unexpectedly increased extracellular NAc DA [ref. 76]. Another report indicates that—in Wistar rats—rimonabant produced enhanced NAc DA response [ref. 147]. In the CPP/CPA model, most publications reported that rimonabant, at low doses, produced neither CPP nor CPA (Table 1). Thus, although some evidence supports that rimonabant could be aversive by itself, which could be related to its inverse agonist profile, conclusive supporting evidence is still lacking. The negative findings may be related to rimonabant doses tested in the above studies and/or the distinct cellular distributions of CB1R expression in different species (rats vs. mice) or subjects under different experimental conditions. As stated below, the important findings with PIMSR, a neutral CB1R antagonists without inverse agonist profile, in the same animal models provide valuable evidence supporting an assumption that the CB1R inverse agonism at least in part underlie the adverse psychiatric effects of rimonabant, as discussed above.
Table 1: The literature reports regarding the rewarding vs. aversive effects of SR141716 itself in rodents.
| Compound | Doses | Species | Results | References |
|---|---|---|---|---|
| Rimonabant | 20 mg/kg, i.p. | Rat | ↓ Electrical brain-stimulation reward | [ref. 142] |
| Rimonabant | 0.3, 1, 3, 10 mg/kg, i.p. | Rat | ↓ Electrical brain-stimulation reward | [ref. 139] |
| Rimonabant | 0.02, 0.3, 1.0 mg/kg, i.p. | Rat | No effect on electrical brain-stimulation reward | [ref. 144] |
| Rimonabant | 0.02 mg/kg, i.p. | Rat | No effect on electrical brain-stimulation reward | [ref. 145] |
| Rimonabant | 3, 10 mg/kg, i.p., | Mouse | No effect on electrical brain-stimulation reward | [ref. 143] |
| Rimonabant | 0.3, 1, 3 mg/kg, i.p. | Rat | Not produce CPP or CPA | [ref. 127] |
| Rimonabant | 0.1, 0.5, 3.0 mg/kg, i.p. | Rat | Not produces CPP or CPA | [ref. 148] |
| Rimonabant | 0.5, 1, 2 mg/kg, i.p. | Rat | Not produce CPP or CPA | [ref. 149] |
| Rimonabant | 3 mg/kg, i.p. | Rat | Not produce CPP or CPA | [ref. 89] |
| Rimonabant | 0.25, 0.5, 1 mg/kg, i.p. | Rat | Not produce CPP or CPA | [ref. 55] |
| Rimonabant | 0.1, 0.5, 3 mg/kg | Rat | Not produce CPP or CPA | [ref. 150] |
| Rimonabant | 0.3, 3 mg/kg, i.p. | Rat | Not produce CPP or CPA | [ref. 151] |
| Rimonabant | 0.25, 0.5, 2, 3 mg/kg | Rat | Produces CPP | [ref. 53] |
| Rimonabant | 3 mg/kg | Mouse | ↓ Accumbens DA | [ref. 146] |
| Rimonabant | 2, 10 mg/kg, i.p. | Rat | No effect on accumbens DA | [ref. 76] |
| Rimonabant | 1, 10, 30, 100 mM, intra-NAc | Rat | ↑ Accumbens DA | [ref. 76] |
| Rimonabant | 5, 10, 20 mg/kg, i.p. | Rat | ↑ Accumbens DA | [ref. 147] |
7. Neutral CB1R Antagonists as New Promising Therapies for SUDs
Although withdrawn from the market, rimonabant still remains a valuable tool in cannabinoid research and in developing newer generation of CB1R ligands with different profiles, such as neutral CB1R antagonists without inverse agonist profile, peripherally restricted CB1R ligands, and allosteric CB1R modulators [ref. 102]. Notably, the pyrazole skeleton of rimonabant has been widely used as the starting point with side chain modifications in developing newer generations of CB1R neutral antagonists, leading to a series of pyrazole-based tricyclic ligands (such as PIMSR, VCHSR, AM4113, AM6527, NESS06SN, etc.) that display neutral CB1R antagonist profiles [ref. 102]. In addition, many other non-pyrazole-based ligands with neutral CB1R antagonist profile (such as thioamide, THCV, O-2654, O-2050, amauromine, and 018-gluc) have also been reported [ref. 102]. Here, we briefly review a few novel CB1R neutral CB1R antagonists that have been tested in experimental animal models of drug abuse.
PIMSR: PIMSR is a pyrazole derivative, which was designed computationally to stabilize both the active and inactive states of CB1R to afford neutral antagonism [ref. 152]. In vitro radioligand binding assays indicate that PIMSR has as high affinity (Ki = 17–57 nM) for human CB1Rs expressed in cultured HEK cells as rimonabant does (Ki = 1.8–18 nM) [ref. 152]. Computational molecular modeling (CB1R docking) and Ca++ channel assays indicate that PIMSR blocked WIN55,212-2-induced inhibition of Ca++ influx [ref. 152]. Electrophysiological assays indicate that co-administration of PIMSR reversed the inhibitory effects of Δ9-THC or synthetic cannabinoids (AM2201, AM018) on excitatory glutamate transmission in the hippocampus [ref. 153]. However, unlike rimonabant that increases Ca++ influx, PIMSR itself has no effect on the Ca++ influx [ref. 152], suggesting that PIMSR is a neutral CB1R antagonist without inverse agonist profile [ref. 154].
Systemic administration of PIMSR (10 mg/kg/day for 28 days) significantly reduces body weight and food intake in high-fat diet-induced obese mice [ref. 154]. We have recently evaluated the therapeutic potential of PIMSR against cocaine use disorder in experimental animals (Table 2). We found that systemic administration of PIMSR dose-dependently inhibited cocaine self-administration under fixed-ratio (FR1, FR5) reinforcement, shifted the cocaine self-administration dose-response curve downward, decreased incentive motivation to seek cocaine under progressive-ratio reinforcement, and reduced cue-induced reinstatement of cocaine seeking [ref. 13]. In addition, PIMSR dose-dependently attenuated cocaine-enhanced ICSS maintained by electrical stimulation of the medial forebrain bundle in rats. Importantly, PIMSR itself failed to alter electrical ICSS, which is often interpreted as a lack of reward-attenuation or depression-like effects (Table 2). This is further supported by our finding that PIMSR alone produced neither rewarding nor aversive effects in the CPP/CPA test. We also examined the effects of PIMSR on food-taking behavior in mice. We found that PIMSR dose-dependently inhibited oral sucrose self-administration and reduced sucrose intake. This finding suggests that PIMSR also retains rimonabant’s therapeutic anti-obesity effects.
Table 2: Effects of CB1R neutral antagonists on drug-taking and drug-seeking behavior in experimental animals.
| Compound | Doses | Species | Results | References |
|---|---|---|---|---|
| PIMSR | 10, 30 mg/kg | Rat | ↓ Cocaine self-administration (FR2, FR5, PR) | [ref. 13] |
| PIMSR | 3, 10, 30 mg/kg, i.p. | Rat | ↓ Cocaine-cue-induced reinstatement | [ref. 13] |
| PIMSR | 3, 10, 30 mg/kg, i.p. | Rat | ↓ Cocaine-enhanced electrical brain-stimulation reward | [ref. 13] |
| PIMSR | 10, 30 mg/kg | Mouse | Not produce CPP or CPA by itself | [ref. 13] |
| PIMSR | 3, 10, 30 mg/kg, i.p. | Rat | Not alter electrical brain-stimulation reward by itself | [ref. 13] |
| AM4113 | 3, 10 mg/kg, i.p. | Rat | Not alter cocaine self-administration | [ref. 139] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | Not alter cocaine self-administration | [ref. 160] |
| AM4113 | 0.3, 1, 3, 10 mg/kg, i.p. | Rat | ↓ PR cocaine self-administration | [ref. 158] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Cue-induced cocaine seeking | [ref. 160] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Cocaine-primed drug seeking | [ref. 160] |
| AM4113 | 3, 10 mg/kg, i.p. | Rat | ↓ Methamphetamine self-administration | [ref. 139] |
| AM4113 | 0.3, 1, 3, 10 mg/kg, i.p. | Rat | ↓ Nicotine self-administration | [ref. 158] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Nicotine self-administration | [ref. 160] |
| AM4113 | 0.3, 1, 3, 10 mg/kg, i.p.; | Rat | ↓ Cue-induced nicotine seeking | [ref. 158] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Cue-induced nicotine seeking | [ref. 160] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Drug priming-induced nicotine seeking | [ref. 160] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Δ9-THC self-administration | [ref. 160] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Cue-induced Δ9-THC seeking | [ref. 160] |
| AM4113 | 0.3–3 mg/kg, i.m. | Monkey | ↓ Drug priming-induced Δ9-THC seeking | [ref. 160] |
| AM4113 | 3, 10 mg/kg, i.p. | Rat | ↓ Heroin self-administration | [ref. 139] |
| AM4113 | 1, 2.5 mg/kg, i.p. | Rat | ↓ Naloxone-precipitated CPA | [ref. 166] |
| AM4113 | 3, 10 mg/kg, i.p. | Rat | No effect on electrical brain-stimulation reward by itself | [ref. 139] |
| AM6527 | 1, 2.5 mg/kg, i.p. | Rat | ↓ Naloxone-precipitated CPA | [ref. 166] |
| Δ8-THCV | 10, 20 mg/kg, i.p. | Rat | ↓ Nicotine self-administration | [ref. 174] |
| Δ8-THCV | 0.03~3 mg/kg, i.p. | Mouse | ↓ Nicotine-induced CPP | [ref. 174] |
| Δ8-THCV | 10, 20 mg/kg, i.p. | Rat | ↓ Nicotine- or cue-induced nicotine seeking | [ref. 174] |
| Δ8-THCV | 0.3 mg/kg, i.p. | Mouse | ↓ Nicotine withdrawal-induced somatic signs | [ref. 174] |
AM4113: AM4113 is another novel CB1R neutral antagonist developed by Alexandros Makriyannis in 2007 [ref. 155]. Structurally, AM4113 is also a pyrazole-3-carboxamide analog of rimonabant [ref. 156]. In competitive [3H]-CP55,940 binding assays, AM4113 has a Ki value of 0.80 ± 0.44 nM and exhibits a 100-fold selectivity for CB1R over CB2R [ref. 157]. Unlike rimonabant, AM4113, at up to 10 µM concentration, has no effect on forskolin-stimulated cAMP accumulation in CB1R-transfected HEK-293 cells [ref. 157], suggesting a lack of an inverse agonist profile.
AM4113 also displayed positive therapeutic effects for SUDs in numerous behavioral tests (Table 2). Similar to rimonabant, AM4113 also inhibits nicotine self-administration, motivation for nicotine seeking, and nicotine priming-, nicotine-associated cue-, or stress-induced reinstatement of nicotine-seeking behavior in rats [ref. 158]. In consistency with these findings, we have recently reported that AM4113 dose-dependently inhibited heroin self-administration but was less effective at reducing cocaine or methamphetamine self-administration in rats under fixed-ratio reinforcement schedules [ref. 139]. In a similar way to AM251, pretreatment with AM4113 attenuated the aversive, affective effects of naloxone-precipitated morphine withdrawal in rats [ref. 159], suggesting an effect mediated by CB1R antagonism. In squirrel monkeys, both AM4113 and rimonabant attenuated nicotine- and Δ9-THC-seeking behaviors, as well as cue-induced reinstatement of cocaine seeking [ref. 160]. In addition, AM4113 reduced alcohol consumption and preference [ref. 161], and attenuated the discriminative effects of CB1R agonists [ref. 162]. Like rimonabant, AM4113 also reduced food intake and weight gain in rats, but not in CB1-KO mice [ref. 156,ref. 157,ref. 159] and precipitated cannabinoid withdrawal signs [ref. 163]. Together, these findings indicate that AM4113 retains rimonabant’s critical therapeutic effects for the treatment of SUDs and obesity (Table 2).
Importantly, AM4113 itself is devoid of many rimonabant-like untoward effects (Table 2). Unlike AM251, AM4113 did not potentiate vomiting in the ferret nor promote nausea [ref. 156,ref. 157]. Additionally, it did not produce malaise or anxiety-like effects [ref. 164]. AM4113 attenuated the depressive effects of WIN55,212–2 as assessed by behavioral profiles in open-field studies [ref. 164]. In the ICSS paradigm, rimonabant decreased electrical ICSS, while AM4114 did not [ref. 139], suggesting that AM4113 did not produce aversive or depression-like effects. All these data suggest that AM4113 has an improved safety profile over rimonabant. However, pharmacokinetic studies indicated that AM4113 had poor oral bioavailability [ref. 161]. In addition, AM4113 was reported to produce anxiety-like effects similar to that by rimonabant in an open field assay [ref. 164]. Thus, although AM4113 is still useful in the proof of concept study, the lack of oral bioavailability and the possible anxiety-like side-effects may limit its development as a clinical candidate [ref. 165].
AM6527: Unlike AM4113, AM6527 is an orally effective CB1R neutral antagonist [ref. 165]. AM6527 has high binding affinity to rat CB1R with ~100-fold selectivity for CB1R (Ki = 4.88 nM) over CB2Rs (Ki = 463) [ref. 165]. In experimental animals both AM6257 and AM4113 inhibited food-reinforced behavior under a FR5 reinforcement schedule with similar ED50 values (0.58 mg/kg vs. 0.78 mg/kg) after intraperitoneal administration. Notably, oral administration of AM6527 was also effective in attenuation of food intake with EC50 value of 1.49 mg/kg, while AM4113 was not effective at an oral dose up to 32 mg/kg [ref. 165]. Systemic administration of AM4113 or AM6527 prevented naloxone-precipitated morphine withdrawal in the CPA paradigm [ref. 166] (Table 2). In drug discriminative test, squirrel monkeys treated daily with the long-acting CB1R agonist AM2389 can effectively discriminate rimonabant from saline [ref. 167,ref. 168]. These discriminative-stimulus effects were both dose- and time-dependent and were stable for up to 48 days [ref. 167]. Importantly, antagonist substitution tests with the CB1R neutral antagonists AM4113 or AM6527 produced similar rimonabant-like discriminative effects, while agonist substitution tests with Δ9-THC (or nabilone, AM4054, JWH018, AM3506) reduced the discriminative effects of rimonabant [ref. 167], suggesting that both AM4113 and AM6527 retain rimonabant’s CB1R antagonist, but not inverse agonist profile. So far, the effects of AM6527 on drug self-administration and reinstatement of drug-seeking behavior have not been examined.
Tetrahydrocannabivarin (THCV): Δ9-Tetrahydrocannabivarin (Δ9-THCV), naturally found in Cannabis, is a homologue of Δ9-THC with a propyl side chain instead of a pentyl group. [35S]GTPγS binding studies indicated that Δ9-THCV acted as a CB1R neutral antagonist at low doses [ref. 169,ref. 170]. However, at higher doses, it may behave as a CB1 agonist, CB2R agonist or antagonist depending on the assays [ref. 170,ref. 171,ref. 172].
In experimental animals, Δ9-THCV produced hypophagic effects in both fasted and non-fasted mice [ref. 173]. Δ8-THCV is a synthetic, more stable, and easier-to-synthesize analogue of Δ9-THCV with a similar pharmacological receptor binding profile [ref. 171]. Systemic administration (i.p.) of Δ8-THCV significantly attenuated intravenous nicotine self-administration and cue-induced and nicotine-induced reinstatement of nicotine-seeking behavior in rats [ref. 174] (Table 2). Δ8-THCV also significantly attenuated nicotine-induced CPP and nicotine withdrawal symptoms in mice (Table 2), suggesting that Δ8-THCV or Δ9-THCV may have therapeutic potential for the treatment of SUDs [ref. 174].
In human clinical trials, Δ9-THCV treatment increased neural responding to rewarding and aversive stimuli [ref. 175]. Another human MRI study indicated that Δ9-THCV decreased resting state functional connectivity in the default mode network and increased connectivity in the cognitive control and dorsal visual stream networks and in the brain regions where functional connectivity is altered in obesity [ref. 176]. These findings suggest that Δ9-THCV deserve further research as a potential therapy for obesity and for SUDs.
8. Summary
The complexities of the neurobiology of SUDs create enormous challenges in efforts to develop effective pharmacological treatments. The initial finding of the overactive eCB status in human subjects with obesity led to the discovery of rimonabant, the first available CB1R antagonist/inverse agonist, for the treatment of obesity in humans. Subsequently, an unexpected finding that rimonabant, when used for the treatment of obesity in humans, also precipitated smoking cessation, inspired huge research effort to explore rimonabant and other CB1R antagonists for the treatment of obesity and SUDs. Although the results from both clinical and preclinical studies are promising, the adverse effects of rimonabant tampered initial enthusiasm and led almost all CB1R ligands withdrawn from human use and clinical trials. This unsuccessful story with rimonabant, although extremely disappointing, also encouraged research to investigate the neural mechanisms underlying the therapeutic versus adverse effects of rimonabant and inspired us to develop and test newer generations of neutral CB1R antagonist for the treatment of SUDs. Extensive research in this area has led to the discovery of numerous neutral antagonists in the past decade. However, only a few of them have been tested in experimental animals. The results demonstrate that the neutral antagonists such as PIMSR and AM4113 retain the therapeutic anti-addictive effects of rimonabant but without significant rimonabant-like adverse effects, which provides the first proof-of-concept evidence supporting that a purer (neutral) CB1R antagonist with little inverse agonism may have a more favorable pharmacological profile in the treatment of obesity and SUDs.
However, there are many challenges in this research area. The studies reviewed above may represent only an early stage in this strategy since it is highly challenging to really “dissect” or “separate” the pharmacological effects produced by CB1R antagonism versus inverse agonism, as both actions may functionally counteract the action produced by drugs of abuse. As inverse agonism, CB1R antagonism, in theory, may also produce unwanted side-effects. Paradoxically, rimonabant itself is not always aversive in the currently used ICSS and CPP/CPA behavioral models. So far, there is only limited evidence indicating that the neutral antagonists (PIMSR, AM4113) are lack of unwanted psychiatric effects. In addition, although preclinical findings with PIMSR and AM4113 have provided encouraging results, the relatively poor blood–brain penetration ability of PIMSR and poor oral bioavailability of AM4113 may lower their translational potential for the use in humans for the treatment of SUDs. Clearly, more research effort is required to either improve the pharmacokinetic profiles of the available CB1R antagonists (such as PIMSR and AM4113) or develop newer generations of neutral antagonists for the treatment of obesity and SUDs.
References
- R.A. Rudd, N. Aleshire, J.E. Zibbell, R.M. Gladden. Increases in drug and opioid overdose deaths—United States, 2000–2014. MMWR Morb. Mortal Wkly. Rep., 2016. [DOI | PubMed]
- 2. CDC (Center for Disease Control and Prevention) Drug over Dose DeathsCDCAtlanta, GA, USA2017
- C.J. Jordan, Z.X. Xi. Progress in brain cannabinoid CB2 receptor research: From genes to behavior. Neurosci. Biobehav. Rev., 2019. [DOI | PubMed]
- C.J. Jordan, Z.X. Xi. Discovery and development of varenicline for smoking cessation. Expert Opin. Drug Discov., 2018. [DOI | PubMed]
- E. Galaj, A.H. Newman, Z.X. Xi. Dopamine D3 receptor-based medication development for the treatment of opioid use disorder: Rationale, progress, and challenges. Neurosci. Biobehav. Rev., 2020. [DOI | PubMed]
- G.F. Koob, N.D. Volkow. Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry, 2016. [DOI | PubMed]
- E. Galaj, Z.X. Xi. Potential of Cannabinoid Receptor Ligands as Treatment for Substance Use Disorders. CNS Drugs, 2019. [DOI | PubMed]
- B. Le Foll, S.R. Goldberg. Cannabinoid CB1 receptor antagonists as promising new medications for drug dependence. J. Pharmacol. Exp. Ther., 2005. [DOI | PubMed]
- M.E. Sloan, J.L. Gowin, V.A. Ramchandani, Y.L. Hurd, B. Le Foll. The endocannabinoid system as a target for addiction treatment: Trials and tribulations. Neuropharmacology, 2017. [DOI | PubMed]
- K. Butler, B. Le Foll. Novel therapeutic and drug development strategies for tobacco use disorder: Endocannabinoid modulation. Expert. Opin. Drug Discov., 2020. [DOI | PubMed]
- B. Le Foll, D.A. Gorelick, S.R. Goldberg. The future of endocannabinoid-oriented clinical research after CB1 antagonists. Psychopharmacology, 2009. [DOI | PubMed]
- W.M. Compton, E.M. Wargo, N.D. Volkow. Neuropsychiatric Model of Addiction Simplified. Psychiatr. Clin. N. Am., 2022. [DOI]
- E. Galaj, B. Hempel, A. Moore, B. Klein, G.H. Bi, E.L. Gardner, H.H. Seltzman, Z.X. Xi. Therapeutic potential of PIMSR, a novel CB1 receptor neutral antagonist, for cocaine use disorder: Evidence from preclinical research. Transl. Psychiatry, 2022. [DOI | PubMed]
- N.D. Volkow, R.A. Wise, R. Baler. The dopamine motive system: Implications for drug and food addiction. Nat. Rev. Neurosci., 2017. [DOI | PubMed]
- Z.X. Xi, E.L. Gardner. Hypothesis-driven medication discovery for the treatment of psychostimulant addiction. Curr. Drug Abuse Rev., 2008. [DOI | PubMed]
- Z.X. Xi, K. Spiller, E.L. Gardner. Mechanism-based medication development for the treatment of nicotine dependence. Acta Pharmacol. Sin., 2009. [DOI | PubMed]
- C.J. Jordan, J. Cao, A.H. Newman, Z.X. Xi. Progress in agonist therapy for substance use disorders: Lessons learned from methadone and buprenorphine. Neuropharmacology, 2019. [DOI | PubMed]
- R.A. Wise. Brain reward circuitry: Insights from unsensed incentives. Neuron, 2002. [DOI | PubMed]
- D. Ron, J. Wang. The NMDA Receptor and Alcohol Addiction. Biology of the NMDA Receptor, 2009
- W. Berrettini. Alcohol addiction and the mu-opioid receptor. Prog. Neuropsychopharmacol. Biol. Psychiatry, 2016. [DOI | PubMed]
- G. Di Chiara. Alcohol and dopamine. Alcohol Health Res. World, 1997. [PubMed]
- E. Galaj, Z.X. Xi. Progress in opioid reward research: From a canonical two-neuron hypothesis to two neural circuits. Pharmacol. Biochem. Behav., 2021. [DOI | PubMed]
- B. Hempel, Z.X. Xi. Receptor mechanisms underlying the CNS effects of cannabinoids: CB1 receptor and beyond. Adv. Pharmacol., 2022. [DOI | PubMed]
- K. Mackie. Distribution of cannabinoid receptors in the central and peripheral nervous system. Handb. Exp. Pharmacol., 2005. [DOI]
- W.A. Devane, L. Hanus, A. Breuer, R.G. Pertwee, L.A. Stevenson, G. Griffin, D. Gibson, A. Mandelbaum, A. Etinger, R. Mechoulam. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science, 1992. [DOI | PubMed]
- J.M. McPartland, M. Glass, R.G. Pertwee. Meta-analysis of cannabinoid ligand binding affinity and receptor distribution: Interspecies differences. Br. J. Pharmacol., 2007. [DOI | PubMed]
- C.J. Hillard, W.S. Edgemond, A. Jarrahian, W.B. Campbell. Accumulation of N-arachidonoylethanolamine (anandamide) into cerebellar granule cells occurs via facilitated diffusion. J. Neurochem., 1997. [DOI | PubMed]
- T. Sugiura, K. Waku. 2-Arachidonoylglycerol and the cannabinoid receptors. Chem. Phys. Lipids, 2000. [DOI | PubMed]
- P.J. Trangenstein, J.M. Whitehill, M.C. Jenkins, D.H. Jernigan, M.A. Moreno. Cannabis Marketing and Problematic Cannabis Use Among Adolescents. J. Stud. Alcohol Drugs, 2021. [DOI | PubMed]
- L. Fattore, P. Fadda, M.S. Spano, M. Pistis, W. Fratta. Neurobiological mechanisms of cannabinoid addiction. Mol. Cell Endocrinol., 2008. [DOI | PubMed]
- R. Maldonado, O. Valverde, F. Berrendero. Involvement of the endocannabinoid system in drug addiction. Trends Neurosci., 2006. [DOI | PubMed]
- National Survey on Drug Use and Health. 2019
- D.C. D’Souza, E. Perry, L. MacDougall, Y. Ammerman, T. Cooper, Y.T. Wu, G. Braley, R. Gueorguieva, J.H. Krystal. The psychotomimetic effects of intravenous delta-9-tetrahydrocannabinol in healthy individuals: Implications for psychosis. Neuropsychopharmacology, 2004. [DOI | PubMed]
- D. Raft, J. Gregg, J. Ghia, L. Harris. Effects of intravenous tetrahydrocannabinol on experimental and surgical pain. Psychological correlates of the analgesic response. Clin. Pharmacol. Ther., 1977. [DOI | PubMed]
- A. Lac, J.W. Luk. Testing the Amotivational Syndrome: Marijuana Use Longitudinally Predicts Lower Self-Efficacy Even After Controlling for Demographics, Personality, and Alcohol and Cigarette Use. Prev. Sci., 2018. [DOI | PubMed]
- A.S. Petrucci, E.M. LaFrance, C. Cuttler. A Comprehensive Examination of the Links between Cannabis Use and Motivation. Subst. Use Misuse, 2020. [DOI | PubMed]
- D.M. Barch, E.C. Dowd. Goal representations and motivational drive in schizophrenia: The role of prefrontal-striatal interactions. Schizophr. Bull., 2010. [DOI | PubMed]
- C.H. Murray, J.E. Glazer, R. Lee, R. Nusslock, H. de Wit. Delta9-THC reduces reward-related brain activity in healthy adults. Psychopharmacology, 2022. [DOI | PubMed]
- H.H. van Hell, G. Jager, M.G. Bossong, A. Brouwer, J.M. Jansma, L. Zuurman, J. van Gerven, R.S. Kahn, N.F. Ramsey. Involvement of the endocannabinoid system in reward processing in the human brain. Psychopharmacology, 2012. [DOI | PubMed]
- J.M. Jansma, H.H. van Hell, L.J. Vanderschuren, M.G. Bossong, G. Jager, R.S. Kahn, N.F. Ramsey. THC reduces the anticipatory nucleus accumbens response to reward in subjects with a nicotine addiction. Transl. Psychiatry, 2013. [DOI | PubMed]
- W. Lawn, T.P. Freeman, R.A. Pope, A. Joye, L. Harvey, C. Hindocha, C. Mokrysz, A. Moss, M.B. Wall, M.A. Bloomfield. Acute and chronic effects of cannabinoids on effort-related decision-making and reward learning: An evaluation of the cannabis ‘amotivational’ hypotheses. Psychopharmacology, 2016. [DOI | PubMed]
- T.P. Freeman, R.A. Pope, M.B. Wall, J.A. Bisby, M. Luijten, C. Hindocha, C. Mokrysz, W. Lawn, A. Moss, M.A.P. Bloomfield. Cannabis Dampens the Effects of Music in Brain Regions Sensitive to Reward and Emotion. Int. J. Neuropsychopharmacol., 2018. [DOI | PubMed]
- M.A.P. Bloomfield, C. Hindocha, S.F. Green, M.B. Wall, R. Lees, K. Petrilli, H. Costello, M.O. Ogunbiyi, M.G. Bossong, T.P. Freeman. The neuropsychopharmacology of cannabis: A review of human imaging studies. Pharmacol. Ther., 2019. [DOI | PubMed]
- Z. Justinova, G. Tanda, G.H. Redhi, S.R. Goldberg. Self-administration of delta9-tetrahydrocannabinol (THC) by drug naive squirrel monkeys. Psychopharmacology, 2003. [DOI | PubMed]
- G. Tanda, P. Munzar, S.R. Goldberg. Self-administration behavior is maintained by the psychoactive ingredient of marijuana in squirrel monkeys. Nat. Neurosci., 2000. [DOI | PubMed]
- R.T. Harris, W. Waters, D. McLendon. Evaluation of reinforcing capability of delta-9-tetrahydrocannabinol in rhesus monkeys. Psychopharmacologia, 1974. [DOI | PubMed]
- W.S. John, T.J. Martin, M.A. Nader. Behavioral Determinants of Cannabinoid Self-Administration in Old World Monkeys. Neuropsychopharmacology, 2017. [DOI | PubMed]
- R.S. Mansbach, K.L. Nicholson, B.R. Martin, R.L. Balster. Failure of Delta(9)-tetrahydrocannabinol and CP 55,940 to maintain intravenous self-administration under a fixed-interval schedule in rhesus monkeys. Behav. Pharmacol., 1994. [DOI | PubMed]
- G. Panagis, S. Vlachou, G.G. Nomikos. Behavioral pharmacology of cannabinoids with a focus on preclinical models for studying reinforcing and dependence-producing properties. Curr. Drug Abuse Rev., 2008. [DOI | PubMed]
- S. Vlachou, G. Panagis. Regulation of brain reward by the endocannabinoid system: A critical review of behavioral studies in animals. Curr. Pharm. Des., 2014. [DOI | PubMed]
- S. Spencer, D. Neuhofer, V.C. Chioma, C. Garcia-Keller, D.J. Schwartz, N. Allen, M.D. Scofield, T. Ortiz-Ithier, P.W. Kalivas. A Model of Delta(9)-Tetrahydrocannabinol Self-administration and Reinstatement That Alters Synaptic Plasticity in Nucleus Accumbens. Biol. Psychiatry, 2018. [DOI | PubMed]
- D. Neuhofer, S.M. Spencer, V.C. Chioma, L.N. Beloate, D. Schwartz, P.W. Kalivas. The loss of NMDAR-dependent LTD following cannabinoid self-administration is restored by positive allosteric modulation of CB1 receptors. Addict. Biol., 2020. [DOI | PubMed]
- J.F. Cheer, D.A. Kendall, C.A. Marsden. Cannabinoid receptors and reward in the rat: A conditioned place preference study. Psychopharmacology, 2000. [DOI | PubMed]
- M.V. DeVuono, K.L. Wills, D.V. MacPherson, K.M. Hrelja, L.A. Parker. Effect of footshock stress on place conditioning produced by Delta(9)-tetrahydrocannabinol and the fatty acid amide hydrolase (FAAH) inhibitor, URB597, in Sprague-Dawley rats. Psychopharmacology, 2017. [DOI | PubMed]
- D. Braida, S. Iosue, S. Pegorini, M. Sala. Delta9-tetrahydrocannabinol-induced conditioned place preference and intracerebroventricular self-administration in rats. Eur. J. Pharmacol., 2004. [DOI | PubMed]
- M. Lepore, X. Liu, V. Savage, D. Matalon, E.L. Gardner. Genetic differences in delta 9-tetrahydrocannabinol-induced facilitation of brain stimulation reward as measured by a rate-frequency curve-shift electrical brain stimulation paradigm in three different rat strains. Life Sci., 1996. [DOI | PubMed]
- E.L. Gardner, W. Paredes, D. Smith, A. Donner, C. Milling, D. Cohen, D. Morrison. Facilitation of brain stimulation reward by delta 9-tetrahydrocannabinol. Psychopharmacology, 1988. [DOI | PubMed]
- V. Katsidoni, A. Kastellakis, G. Panagis. Biphasic effects of Delta9-tetrahydrocannabinol on brain stimulation reward and motor activity. Int. J. Neuropsychopharmacol., 2013. [DOI | PubMed]
- A.J. Kwilasz, S.S. Negus. Dissociable effects of the cannabinoid receptor agonists Delta9-tetrahydrocannabinol and CP55940 on pain-stimulated versus pain-depressed behavior in rats. J. Pharmacol. Exp. Ther., 2012. [DOI | PubMed]
- S.S. Negus, L.L. Miller. Intracranial self-stimulation to evaluate abuse potential of drugs. Pharmacol. Rev., 2014. [DOI | PubMed]
- S. Vlachou, G.G. Nomikos, D.N. Stephens, G. Panagis. Lack of evidence for appetitive effects of Delta 9-tetrahydrocannabinol in the intracranial self-stimulation and conditioned place preference procedures in rodents. Behav. Pharmacol., 2007. [DOI | PubMed]
- J.M. Wiebelhaus, T.W. Grim, R.A. Owens, M.F. Lazenka, L.J. Sim-Selley, R.A. Abdullah, M.J. Niphakis, R.E. Vann, B.F. Cravatt, J.L. Wiley. Delta9-tetrahydrocannabinol and endocannabinoid degradative enzyme inhibitors attenuate intracranial self-stimulation in mice. J. Pharmacol. Exp. Ther., 2015. [DOI | PubMed]
- K.J. Spiller, G.H. Bi, Y. He, E. Galaj, E.L. Gardner, Z.X. Xi. Cannabinoid CB1 and CB2 receptor mechanisms underlie cannabis reward and aversion in rats. Br. J. Pharmacol., 2019. [DOI | PubMed]
- X. Han, Y. He, G.H. Bi, H.Y. Zhang, R. Song, Q.R. Liu, J.M. Egan, E.L. Gardner, J. Li, Z.X. Xi. CB1 Receptor Activation on VgluT2-Expressing Glutamatergic Neurons Underlies Delta(9)-Tetrahydrocannabinol (Delta(9)-THC)-Induced Aversive Effects in Mice. Sci. Rep., 2017. [DOI | PubMed]
- B.A. Humburg, C.J. Jordan, H.Y. Zhang, H. Shen, X. Han, G.H. Bi, B. Hempel, E. Galaj, M.H. Baumann, Z.X. Xi. Optogenetic brain-stimulation reward: A new procedure to re-evaluate the rewarding versus aversive effects of cannabinoids in dopamine transporter-Cre mice. Addict. Biol., 2021. [DOI | PubMed]
- J.F. Cheer, D.A. Kendall, R. Mason, C.A. Marsden. Differential cannabinoid-induced electrophysiological effects in rat ventral tegmentum. Neuropharmacology, 2003. [DOI | PubMed]
- J.F. Cheer, K.M. Wassum, M.L. Heien, P.E. Phillips, R.M. Wightman. Cannabinoids enhance subsecond dopamine release in the nucleus accumbens of awake rats. J. Neurosci., 2004. [DOI | PubMed]
- G. Tanda, F.E. Pontieri, G. Di Chiara. Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science, 1997. [DOI | PubMed]
- D.P. Covey, Y. Mateo, D. Sulzer, J.F. Cheer, D.M. Lovinger. Endocannabinoid modulation of dopamine neurotransmission. Neuropharmacology, 2017. [DOI | PubMed]
- M.L. Fitzgerald, E. Shobin, V.M. Pickel. Cannabinoid modulation of the dopaminergic circuitry: Implications for limbic and striatal output. Prog. Neuropsychopharmacol. Biol. Psychiatry, 2012. [DOI | PubMed]
- C.A.T. Paladini. Neurophysiology of substantia nigra dopamine neurons: Modulation by GABA and glutamate. Handbook of Basal Ganglia Structure and Function, 2017
- B. Szabo, T. Muller, H. Koch. Effects of cannabinoids on dopamine release in the corpus striatum and the nucleus accumbens in vitro. J. Neurochem., 1999. [DOI | PubMed]
- E.M. Castañeda, S.D. Oddie, Q. Whishaw. THC does not affect striatal dopamine release: Microdialysis in freely moving rats. Pharmacol. Biochem. Behav., 1991. [DOI | PubMed]
- G. Pillolla, M. Melis, S. Perra, A.L. Muntoni, G.L. Gessa, M. Pistis. Medial forebrain bundle stimulation evokes endocannabinoid-mediated modulation of ventral tegmental area dopamine neuron firing in vivo. Psychopharmacology, 2007. [DOI | PubMed]
- Z. Sidlo, P.H. Reggio, M.E. Rice. Inhibition of striatal dopamine release by CB1 receptor activation requires nonsynaptic communication involving GABA, H2O2, and KATP channels. Neurochem. Int., 2008. [DOI | PubMed]
- X. Li, B.J. Hempel, H.J. Yang, X. Han, G.H. Bi, E.L. Gardner, Z.X. Xi. Dissecting the role of CB1 and CB2 receptors in cannabinoid reward versus aversion using transgenic CB1- and CB2-knockout mice. Eur. Neuropsychopharmacol., 2021. [DOI | PubMed]
- C.R. Lupica, A.C. Riegel. Endocannabinoid release from midbrain dopamine neurons: A potential substrate for cannabinoid receptor antagonist treatment of addiction. Neuropharmacology, 2005. [DOI | PubMed]
- B. Szabo, S. Siemes, I. Wallmichrath. Inhibition of GABAergic neurotransmission in the ventral tegmental area by cannabinoids. Eur. J. Neurosci., 2002. [DOI | PubMed]
- M. Melis, M. Pistis. Endocannabinoid signaling in midbrain dopamine neurons: More than physiology?. Curr. Neuropharmacol., 2007. [DOI | PubMed]
- M. Melis, C. Sagheddu, M. De Felice, A. Casti, C. Madeddu, S. Spiga, A.L. Muntoni, K. Mackie, G. Marsicano, G. Colombo. Enhanced endocannabinoid-mediated modulation of rostromedial tegmental nucleus drive onto dopamine neurons in Sardinian alcohol-preferring rats. J. Neurosci., 2014. [DOI | PubMed]
- X.F. Wang, E. Galaj, G.H. Bi, C. Zhang, Y. He, J. Zhan, M.H. Bauman, E.L. Gardner, Z.X. Xi. Different receptor mechanisms underlying phytocannabinoid- versus synthetic cannabinoid-induced tetrad effects: Opposite roles of CB1 /CB2 versus GPR55 receptors. Br. J. Pharmacol., 2020. [DOI | PubMed]
- J. Manzanares, D. Cabanero, N. Puente, M.S. Garcia-Gutierrez, P. Grandes, R. Maldonado. Role of the endocannabinoid system in drug addiction. Biochem. Pharmacol., 2018. [DOI | PubMed]
- H.Y. Zhang, M. Gao, Q.R. Liu, G.H. Bi, X. Li, H.J. Yang, E.L. Gardner, J. Wu, Z.X. Xi. Cannabinoid CB2 receptors modulate midbrain dopamine neuronal activity and dopamine-related behavior in mice. Proc. Natl. Acad. Sci. USA, 2014. [DOI | PubMed]
- H.Y. Zhang, M. Gao, H. Shen, G.H. Bi, H.J. Yang, Q.R. Liu, J. Wu, E.L. Gardner, A. Bonci, Z.X. Xi. Expression of functional cannabinoid CB2 receptor in VTA dopamine neurons in rats. Addict. Biol., 2017. [DOI | PubMed]
- Z.X. Xi, X.Q. Peng, X. Li, R. Song, H.Y. Zhang, Q.R. Liu, H.J. Yang, G.H. Bi, J. Li, E.L. Gardner. Brain cannabinoid CB(2) receptors modulate cocaine’s actions in mice. Nat. Neurosci., 2011. [DOI | PubMed]
- D.J. Foster, J.M. Wilson, D.H. Remke, M.S. Mahmood, M.J. Uddin, J. Wess, S. Patel, L.J. Marnett, C.M. Niswender, C.K. Jones. Antipsychotic-like Effects of M4 Positive Allosteric Modulators Are Mediated by CB2 Receptor-Dependent Inhibition of Dopamine Release. Neuron, 2016. [DOI | PubMed]
- H.Y. Zhang, G.H. Bi, X. Li, J. Li, H. Qu, S.J. Zhang, C.Y. Li, E.S. Onaivi, E.L. Gardner, Z.X. Xi. Species differences in cannabinoid receptor 2 and receptor responses to cocaine self-administration in mice and rats. Neuropsychopharmacology, 2015. [DOI | PubMed]
- A. Aracil-Fernandez, J.M. Trigo, M.S. Garcia-Gutierrez, A. Ortega-Alvaro, A. Ternianov, D. Navarro, P. Robledo, P. Berbel, R. Maldonado, J. Manzanares. Decreased cocaine motor sensitization and self-administration in mice overexpressing cannabinoid CB(2) receptors. Neuropsychopharmacology, 2012. [DOI | PubMed]
- F. Delis, A. Polissidis, N. Poulia, Z. Justinova, G.G. Nomikos, S.R. Goldberg, K. Antoniou. Attenuation of Cocaine-Induced Conditioned Place Preference and Motor Activity via Cannabinoid CB2 Receptor Agonism and CB1 Receptor Antagonism in Rats. Int. J. Neuropsychopharmacol., 2017. [DOI | PubMed]
- S. Azar, S. Sherf-Dagan, A. Nemirovski, M. Webb, A. Raziel, A. Keidar, D. Goitein, N. Sakran, O. Shibolet, J. Tam. Circulating Endocannabinoids Are Reduced Following Bariatric Surgery and Associated with Improved Metabolic Homeostasis in Humans. Obes. Surg., 2019. [DOI | PubMed]
- A.M. Monteleone, F. Piscitelli, R. Dalle Grave, M. El Ghoch, V. Di Marzo, M. Maj, P. Monteleone. Peripheral Endocannabinoid Responses to Hedonic Eating in Binge-Eating Disorder. Nutrients, 2017. [DOI]
- H.J. van Eyk, L.D. van Schinkel, V. Kantae, C.E.A. Dronkers, J.J.M. Westenberg, A. de Roos, H.J. Lamb, J.W. Jukema, A.C. Harms, T. Hankemeier. Caloric restriction lowers endocannabinoid tonus and improves cardiac function in type 2 diabetes. Nutr. Diabetes, 2018. [DOI | PubMed]
- J.M. Perkins, S.N. Davis. Endocannabinoid system overactivity and the metabolic syndrome: Prospects for treatment. Curr. Diab. Rep., 2008. [DOI | PubMed]
- D. Duffy, D. Rader. Endocannabinoid antagonism: Blocking the excess in the treatment of high-risk abdominal obesity. Trends Cardiovasc. Med., 2007. [DOI | PubMed]
- M. Rinaldi-Carmona, F. Barth, M. Heaulme, D. Shire, B. Calandra, C. Congy, S. Martinez, J. Maruani, G. Neliat, D. Caput. SR141716A, a potent and selective antagonist of the brain cannabinoid receptor. FEBS Lett., 1994. [DOI | PubMed]
- 96. EMA European Public Assessment Report (EPAR) Acomplia: EPAR Summary for the PublicEMALondon, UK2007
- V. Di Marzo, J.P. Despres. CB1 antagonists for obesity—What lessons have we learned from rimonabant?. Nat. Rev. Endocrinol., 2009. [DOI | PubMed]
- R. Christensen, P.K. Kristensen, E.M. Bartels, H. Bliddal, A. Astrup. Efficacy and safety of the weight-loss drug rimonabant: A meta-analysis of randomised trials. Lancet, 2007. [DOI | PubMed]
- A.H. Sam, V. Salem, M.A. Ghatei. Rimonabant: From RIO to Ban. J. Obes., 2011. [DOI | PubMed]
- 100. EMA European Medicines Agency: Public Statement on Acomplia (Rimonabant) Withdrawal of the Marketing Authorisation in the European UnionEMALondon, UK2009
- S. Saul. F.D.A. Panel Rejects Drug for Obesity. The New York Times. 2007
- T. Nguyen, B.F. Thomas, Y. Zhang. Overcoming the Psychiatric Side Effects of the Cannabinoid CB1 Receptor Antagonists: Current Approaches for Therapeutics Development. Curr. Top. Med. Chem., 2019. [DOI | PubMed]
- J.P. Despres, R. Ross, G. Boka, N. Almeras, I. Lemieux, A.D.-L. Investigators. Effect of rimonabant on the high-triglyceride/low-HDL-cholesterol dyslipidemia, intraabdominal adiposity, and liver fat: The ADAGIO-Lipids trial. Arterioscler. Thromb. Vasc. Biol., 2009. [DOI | PubMed]
- J. Rosenstock, P. Hollander, S. Chevalier, A. Iranmanesh, S.S. Group. SERENADE: The Study Evaluating Rimonabant Efficacy in Drug-naive Diabetic Patients: Effects of monotherapy with rimonabant, the first selective CB1 receptor antagonist, on glycemic control, body weight, and lipid profile in drug-naive type 2 diabetes. Diabetes Care, 2008. [DOI | PubMed]
- F.X. Pi-Sunyer, L.J. Aronne, H.M. Heshmati, J. Devin, J. Rosenstock. Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients: RIO-North America: A randomized controlled trial. JAMA, 2006. [DOI | PubMed]
- M. Bluher. Efficacy and safety of the weight-loss drug rimonabant. Lancet, 2008. [DOI]
- J.P. Despres, L. Van Gaal, X. Pi-Sunyer, A. Scheen. Efficacy and safety of the weight-loss drug rimonabant. Lancet, 2008. [DOI]
- M. Rinaldi-Carmona, F. Barth, M. Heaulme, R. Alonso, D. Shire, C. Congy, P. Soubrie, J.C. Breliere, G. Le Fur. Biochemical and pharmacological characterisation of SR141716A, the first potent and selective brain cannabinoid receptor antagonist. Life Sci., 1995. [DOI | PubMed]
- D.R. Compton, M.D. Aceto, J. Lowe, B.R. Martin. In vivo characterization of a specific cannabinoid receptor antagonist (SR141716A): Inhibition of delta 9-tetrahydrocannabinol-induced responses and apparent agonist activity. J. Pharmacol. Exp. Ther., 1996. [PubMed]
- J.D. Richardson, L. Aanonsen, K.M. Hargreaves. SR 141716A, a cannabinoid receptor antagonist, produces hyperalgesia in untreated mice. Eur. J. Pharmacol., 1997. [DOI | PubMed]
- R.S. Landsman, T.H. Burkey, P. Consroe, W.R. Roeske, H.I. Yamamura. SR141716A is an inverse agonist at the human cannabinoid CB1 receptor. Eur. J. Pharmacol., 1997. [DOI | PubMed]
- K. Cahill, M.H. Ussher. Cannabinoid type 1 receptor antagonists for smoking cessation. Cochrane Database Syst. Rev., 2011. [DOI | PubMed]
- M.Y. Elrashidi, J.O. Ebbert. Emerging drugs for the treatment of tobacco dependence: 2014 update. Expert Opin. Emerg. Drugs, 2014. [DOI | PubMed]
- M.B. Steinberg, J. Foulds. Rimonabant for treating tobacco dependence. Vasc. Health Risk Manag., 2007. [PubMed]
- M.A. Huestis, S.J. Boyd, S.J. Heishman, K.L. Preston, D. Bonnet, G. Le Fur, D.A. Gorelick. Single and multiple doses of rimonabant antagonize acute effects of smoked cannabis in male cannabis users. Psychopharmacology, 2007. [DOI | PubMed]
- M. Solinas, L.V. Panlilio, S.R. Goldberg. Exposure to delta-9-tetrahydrocannabinol (THC) increases subsequent heroin taking but not heroin’s reinforcing efficacy: A self-administration study in rats. Neuropsychopharmacology, 2004. [DOI | PubMed]
- I. Gamaleddin, C. Wertheim, A.Z. Zhu, K.M. Coen, K. Vemuri, A. Makryannis, S.R. Goldberg, B. Le Foll. Cannabinoid receptor stimulation increases motivation for nicotine and nicotine seeking. Addict. Biol., 2012. [DOI | PubMed]
- G. Colombo, S. Serra, G. Brunetti, R. Gomez, S. Melis, G. Vacca, M.M. Carai, L. Gessa. Stimulation of voluntary ethanol intake by cannabinoid receptor agonists in ethanol-preferring sP rats. Psychopharmacology, 2002. [DOI | PubMed]
- D.N. Linsenbardt, S.L. Boehm. Agonism of the endocannabinoid system modulates binge-like alcohol intake in male C57BL/6J mice: Involvement of the posterior ventral tegmental area. Neuroscience, 2009. [DOI | PubMed]
- I.H. Gamaleddin, J.M. Trigo, A.B. Gueye, A. Zvonok, A. Makriyannis, S.R. Goldberg, B. Le Foll. Role of the endogenous cannabinoid system in nicotine addiction: Novel insights. Front. Psychiatry, 2015. [DOI | PubMed]
- P. Maccioni, G. Colombo, M.A. Carai. Blockade of the cannabinoid CB1 receptor and alcohol dependence: Preclinical evidence and preliminary clinical data. CNS Neurol. Disord. Drug Targets, 2010. [DOI | PubMed]
- M. Solinas, L.V. Panlilio, K. Antoniou, L.A. Pappas, S.R. Goldberg. The cannabinoid CB1 antagonist N-piperidinyl-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl) -4-methylpyrazole-3-carboxamide (SR-141716A) differentially alters the reinforcing effects of heroin under continuous reinforcement, fixed ratio, and progressive ratio schedules of drug self-administration in rats. J. Pharmacol. Exp. Ther., 2003. [DOI | PubMed]
- T.J. De Vries, J.R. Homberg, R. Binnekade, H. Raaso, A.N.M. Schoffelmeer. Cannabinoid modulation of the reinforcing and motivational properties of heroin and heroin-associated cues in rats. Psychopharmacology, 2003. [DOI | PubMed]
- C. Cohen, G. Perrault, C. Voltz, R. Steinberg, P. Soubrie. SR141716, a central cannabinoid (CB(1)) receptor antagonist, blocks the motivational and dopamine-releasing effects of nicotine in rats. Behav. Pharmacol., 2002. [DOI | PubMed]
- C. Cohen, G. Perrault, G. Griebel, P. Soubrie. Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: Reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716). Neuropsychopharmacology, 2005. [DOI | PubMed]
- B. Le Foll, S.R. Goldberg. Rimonabant, a CB1 antagonist, blocks nicotine-conditioned place preferences. Neuroreport., 2004. [DOI | PubMed]
- F. Chaperon, P. Soubrie, A.J. Puech, M.H. Thiebot. Involvement of central cannabinoid (CB1) receptors in the establishment of place conditioning in rats. Psychopharmacology, 1998. [DOI | PubMed]
- T.J. De Vries, Y. Shaham, J.R. Homberg, H. Crombag, K. Schuurman, J. Dieben, L.J. Vanderschuren, A.N. Schoffelmeer. A cannabinoid mechanism in relapse to cocaine seeking. Nat. Med., 2001. [DOI | PubMed]
- K. Anggadiredja, M. Nakamichi, T. Hiranita, H. Tanaka, Y. Shoyama, S. Watanabe, T. Yamamoto. Endocannabinoid system modulates relapse to methamphetamine seeking: Possible mediation by the arachidonic acid cascade. Neuropsychopharmacology, 2004. [DOI | PubMed]
- A.N. Henderson-Redmond, J. Guindon, D.J. Morgan. Roles for the endocannabinoid system in ethanol-motivated behavior. Prog. Neuropsychopharmacol. Biol. Psychiatry, 2016. [DOI | PubMed]
- S. Gonzalez, P.C. Schmid, J. Fernandez-Ruiz, R. Krebsbach, H.H. Schmid, J.A. Ramos. Region-dependent changes in endocannabinoid transmission in the brain of morphine-dependent rats. Addict. Biol., 2003. [DOI | PubMed]
- D. Vigano, M. Grazia Cascio, T. Rubino, F. Fezza, A. Vaccani, V. Di Marzo, D. Parolaro. Chronic morphine modulates the contents of the endocannabinoid, 2-arachidonoyl glycerol, in rat brain. Neuropsychopharmacology, 2003. [DOI | PubMed]
- D. Vigano, M. Valenti, M.G. Cascio, V. Di Marzo, D. Parolaro, T. Rubino. Changes in endocannabinoid levels in a rat model of behavioural sensitization to morphine. Eur. J. Neurosci., 2004. [DOI | PubMed]
- S. Gonzalez, M.G. Cascio, J. Fernandez-Ruiz, F. Fezza, V. Di Marzo, J.A. Ramos. Changes in endocannabinoid contents in the brain of rats chronically exposed to nicotine, ethanol or cocaine. Brain Res., 2002. [DOI | PubMed]
- H. Wang, T. Treadway, D.P. Covey, J.F. Cheer, C.R. Lupica. Cocaine-Induced Endocannabinoid Mobilization in the Ventral Tegmental Area. Cell Rep., 2015. [DOI | PubMed]
- S. Caille, L. Alvarez-Jaimes, I. Polis, D.G. Stouffer, L.H. Parsons. Specific alterations of extracellular endocannabinoid levels in the nucleus accumbens by ethanol, heroin, and cocaine self-administration. J. Neurosci., 2007. [DOI | PubMed]
- H. Zhang, A.A. Lipinski, E. Liktor-Busa, A.F. Smith, A. Moutal, R. Khanna, P.R. Langlais, T.M. Largent-Milnes, T.W. Vanderah. The Effects of Repeated Morphine Treatment on the Endogenous Cannabinoid System in the Ventral Tegmental Area. Front. Pharmacol., 2021. [DOI | PubMed]
- M.W. Buczynski, I.Y. Polis, L.H. Parsons. The volitional nature of nicotine exposure alters anandamide and oleoylethanolamide levels in the ventral tegmental area. Neuropsychopharmacology, 2013. [DOI | PubMed]
- X.H. He, C.J. Jordan, K. Vemuri, G.H. Bi, J. Zhan, E.L. Gardner, A. Makriyannis, Y.L. Wang, Z.X. Xi. Cannabinoid CB1 receptor neutral antagonist AM4113 inhibits heroin self-administration without depressive side effects in rats. Acta Pharmacol. Sin., 2019. [DOI | PubMed]
- H. Anisman, K. Matheson. Stress, depression, and anhedonia: Caveats concerning animal models. Neurosci. Biobehav. Rev., 2005. [DOI | PubMed]
- S. Scheggi, M.G. De Montis, C. Gambarana. Making Sense of Rodent Models of Anhedonia. Int. J. Neuropsychopharmacol., 2018. [DOI | PubMed]
- J.C. Arnold, G.E. Hunt, I.S. McGregor. Effects of the cannabinoid receptor agonist CP 55,940 and the cannabinoid receptor antagonist SR 141716 on intracranial self-stimulation in Lewis rats. Life Sci., 2001. [DOI | PubMed]
- T.W. Grim, J.M. Wiebelhaus, A.J. Morales, S.S. Negus, A.H. Lichtman. Effects of acute and repeated dosing of the synthetic cannabinoid CP55,940 on intracranial self-stimulation in mice. Drug Alcohol Depend., 2015. [DOI | PubMed]
- S. Vlachou, G.G. Nomikos, G. Panagis. Effects of endocannabinoid neurotransmission modulators on brain stimulation reward. Psychopharmacology, 2006. [DOI | PubMed]
- S. Vlachou, F. Stamatopoulou, G.G. Nomikos, G. Panagis. Enhancement of endocannabinoid neurotransmission through CB1 cannabinoid receptors counteracts the reinforcing and psychostimulant effects of cocaine. Int. J. Neuropsychopharmacol., 2008. [DOI | PubMed]
- X. Li, A.F. Hoffman, X.Q. Peng, C.R. Lupica, E.L. Gardner, Z.X. Xi. Attenuation of basal and cocaine-enhanced locomotion and nucleus accumbens dopamine in cannabinoid CB1-receptor-knockout mice. Psychopharmacology, 2009. [DOI | PubMed]
- E. Murillo-Rodriguez, S. Machado, N.B. Rocha, H. Budde, T.F. Yuan, O. Arias-Carrion. Revealing the role of the endocannabinoid system modulators, SR141716A, URB597 and VDM-11, in sleep homeostasis. Neuroscience, 2016. [DOI | PubMed]
- F. Li, Q. Fang, Y. Liu, M. Zhao, D. Li, J. Wang, L. Lu. Cannabinoid CB(1) receptor antagonist rimonabant attenuates reinstatement of ketamine conditioned place preference in rats. Eur. J. Pharmacol., 2008. [DOI | PubMed]
- G. Biala, B. Budzynska, N. Staniak. Effects of rimonabant on the reinstatement of nicotine-conditioned place preference by drug priming in rats. Behav. Brain. Res., 2009. [DOI | PubMed]
- M.E. Singh, A.N. Verty, I.S. McGregor, P.E. Mallet. A cannabinoid receptor antagonist attenuates conditioned place preference but not behavioural sensitization to morphine. Brain Res., 2004. [DOI | PubMed]
- Q. Fang, F.Q. Li, Y.Q. Li, Y.X. Xue, Y.Y. He, J.F. Liu, L. Lu, J.S. Wang. Cannabinoid CB1 receptor antagonist rimonabant disrupts nicotine reward-associated memory in rats. Pharmacol. Biochem. Behav., 2011. [DOI | PubMed]
- D. Hurst, U. Umejiego, D. Lynch, H. Seltzman, S. Hyatt, M. Roche, S. McAllister, D. Fleischer, A. Kapur, M. Abood. Biarylpyrazole inverse agonists at the cannabinoid CB1 receptor: Importance of the C-3 carboxamide oxygen/lysine3.28(192) interaction. J. Med. Chem., 2006. [DOI | PubMed]
- A.F. Hoffman, M.D. Lycas, J.R. Kaczmarzyk, C.E. Spivak, M.H. Baumann, C.R. Lupica. Disruption of hippocampal synaptic transmission and long-term potentiation by psychoactive synthetic cannabinoid ‘Spice’ compounds: Comparison with Delta(9) -tetrahydrocannabinol. Addict Biol., 2017. [DOI | PubMed]
- H.H. Seltzman, R. Maitra, K. Bortoff, J. Henson, P.H. Reggio, D. Wesley, J. Tam. Metabolic Profiling of CB1 Neutral Antagonists. Methods Enzymol., 2017. [DOI | PubMed]
- J.D. Salamone, P.J. McLaughlin, K. Sink, A. Makriyannis, L.A. Parker. Cannabinoid CB1 receptor inverse agonists and neutral antagonists: Effects on food intake, food-reinforced behavior and food aversions. Physiol. Behav., 2007. [DOI | PubMed]
- K.S. Sink, P.J. McLaughlin, J.A. Wood, C. Brown, P. Fan, V.K. Vemuri, Y. Peng, T. Olszewska, G.A. Thakur, A. Makriyannis. The novel cannabinoid CB1 receptor neutral antagonist AM4113 suppresses food intake and food-reinforced behavior but does not induce signs of nausea in rats. Neuropsychopharmacology, 2008. [DOI | PubMed]
- A.P. Chambers, V.K. Vemuri, Y. Peng, J.T. Wood, T. Olszewska, Q.J. Pittman, A. Makriyannis, K.A. Sharkey. A neutral CB1 receptor antagonist reduces weight gain in rat. Am. J. Physiol. Regul. Integr. Comp. Physiol., 2007. [DOI | PubMed]
- A.B. Gueye, Y. Pryslawsky, J.M. Trigo, N. Poulia, F. Delis, K. Antoniou, M. Loureiro, S.R. Laviolette, K. Vemuri, A. Makriyannis. The CB1 Neutral Antagonist AM4113 Retains the Therapeutic Efficacy of the Inverse Agonist Rimonabant for Nicotine Dependence and Weight Loss with Better Psychiatric Tolerability. Int. J. Neuropsychopharmacol., 2016. [DOI | PubMed]
- N.L. Cluny, V.K. Vemuri, A.P. Chambers, C.L. Limebeer, H. Bedard, J.T. Wood, B. Lutz, A. Zimmer, L.A. Parker, A. Makriyannis. A novel peripherally restricted cannabinoid receptor antagonist, AM6545, reduces food intake and body weight, but does not cause malaise, in rodents. Br. J. Pharmacol., 2010. [DOI | PubMed]
- C.W. Schindler, G.H. Redhi, K. Vemuri, A. Makriyannis, B. Le Foll, J. Bergman, S.R. Goldberg, Z. Justinova. Blockade of Nicotine and Cannabinoid Reinforcement and Relapse by a Cannabinoid CB1-Receptor Neutral Antagonist AM4113 and Inverse Agonist Rimonabant in Squirrel Monkeys. Neuropsychopharmacology, 2016. [DOI | PubMed]
- A. Balla, B. Dong, B.M. Shilpa, K. Vemuri, A. Makriyannis, S.C. Pandey, H. Sershen, R.F. Suckow, K.Y. Vinod. Cannabinoid-1 receptor neutral antagonist reduces binge-like alcohol consumption and alcohol-induced accumbal dopaminergic signaling. Neuropharmacology, 2018. [DOI | PubMed]
- B.D. Kangas, M.S. Delatte, V.K. Vemuri, G.A. Thakur, S.P. Nikas, K.V. Subramanian, V.G. Shukla, A. Makriyannis, J. Bergman. Cannabinoid discrimination and antagonism by CB(1) neutral and inverse agonist antagonists. J. Pharmacol. Exp. Ther., 2013. [DOI | PubMed]
- S. Tai, S.P. Nikas, V.G. Shukla, K. Vemuri, A. Makriyannis, T.U. Jarbe. Cannabinoid withdrawal in mice: Inverse agonist vs neutral antagonist. Psychopharmacology, 2015. [DOI | PubMed]
- T.U. Jarbe, B.J. LeMay, T. Olszewska, V.K. Vemuri, J.T. Wood, A. Makriyannis. Intrinsic effects of AM4113, a putative neutral CB1 receptor selective antagonist, on open-field behaviors in rats. Pharmacol. Biochem. Behav., 2008. [DOI | PubMed]
- K.S. Sink, V.K. Vemuri, J. Wood, A. Makriyannis, J.D. Salamone. Oral bioavailability of the novel cannabinoid CB1 antagonist AM6527: Effects on food-reinforced behavior and comparisons with AM4113. Pharmacol. Biochem. Behav., 2009. [DOI | PubMed]
- K.L. Wills, K. Vemuri, A. Kalmar, A. Lee, C.L. Limebeer, A. Makriyannis, L.A. Parker. CB1 antagonism: Interference with affective properties of acute naloxone-precipitated morphine withdrawal in rats. Psychopharmacology, 2014. [DOI | PubMed]
- B.D. Kangas, A.S. Zakarian, K. Vemuri, S.O. Alapafuja, S. Jiang, S.P. Nikas, A. Makriyannis, J. Bergman. Cannabinoid Antagonist Drug Discrimination in Nonhuman Primates. J. Pharmacol. Exp. Ther., 2020. [DOI | PubMed]
- J.L. Stewart, L.R. McMahon. Rimonabant-induced Delta9-tetrahydrocannabinol withdrawal in rhesus monkeys: Discriminative stimulus effects and other withdrawal signs. J. Pharmacol. Exp. Ther., 2010. [DOI | PubMed]
- R.G. Pertwee. Inverse agonism and neutral antagonism at cannabinoid CB1 receptors. Life Sci., 2005. [DOI | PubMed]
- R.G. Pertwee, A. Thomas, L.A. Stevenson, R.A. Ross, S.A. Varvel, A.H. Lichtman, B.R. Martin, R.K. Razdan. The psychoactive plant cannabinoid, Delta9-tetrahydrocannabinol, is antagonized by Delta8- and Delta9-tetrahydrocannabivarin in mice in vivo. Br. J. Pharmacol., 2007. [DOI | PubMed]
- S. Batkai, P. Mukhopadhyay, B. Horvath, M. Rajesh, R.Y. Gao, A. Mahadevan, M. Amere, N. Battista, A.H. Lichtman, L.A. Gauson. Delta8-Tetrahydrocannabivarin prevents hepatic ischaemia/reperfusion injury by decreasing oxidative stress and inflammatory responses through cannabinoid CB2 receptors. Br. J. Pharmacol., 2012. [DOI | PubMed]
- A. Thomas, L.A. Stevenson, K.N. Wease, M.R. Price, G. Baillie, R.A. Ross, R.G. Pertwee. Evidence that the plant cannabinoid Delta9-tetrahydrocannabivarin is a cannabinoid CB1 and CB2 receptor antagonist. Br. J. Pharmacol., 2005. [DOI | PubMed]
- G. Riedel, P. Fadda, S. McKillop-Smith, R.G. Pertwee, B. Platt, L. Robinson. Synthetic and plant-derived cannabinoid receptor antagonists show hypophagic properties in fasted and non-fasted mice. Br. J. Pharmacol., 2009. [DOI | PubMed]
- Z.X. Xi, P. Muldoon, X.F. Wang, G.H. Bi, M.I. Damaj, A.H. Lichtman, R.G. Pertwee, E.L. Gardner. Delta(8)—Tetrahydrocannabivarin has potent anti-nicotine effects in several rodent models of nicotine dependence. Br. J. Pharmacol., 2019. [DOI | PubMed]
- L. Tudge, C. Williams, P.J. Cowen, C. McCabe. Neural effects of cannabinoid CB1 neutral antagonist tetrahydrocannabivarin on food reward and aversion in healthy volunteers. Int. J. Neuropsychopharmacol., 2014. [DOI | PubMed]
- E. Rzepa, L. Tudge, C. McCabe. The CB1 Neutral Antagonist Tetrahydrocannabivarin Reduces Default Mode Network and Increases Executive Control Network Resting State Functional Connectivity in Healthy Volunteers. Int. J. Neuropsychopharmacol., 2015. [DOI | PubMed]
