Long-term exposure to ambient air pollutants and female infertility risk: a population-based cohort study in Taiwan
Abstract
Introduction:
Infertility affects over 186 million people globally, with about 1 in 7 couples in developed nations experiencing it. Causes include age-related fertility decline and environmental factors. Air pollution is a potential factor, but large-scale evidence is still lacking. This study examines the impact of several air pollutants on infertility in females aged 15 to 60, hypothesizing that air pollution increases infertility risks.
Method:
We constructed a cohort in Taiwan between July 1, 2003, and December 31, 2013, using the National Health Insurance Research Database (NHIRD) of females aged 15 to 60. Concentrations of SO2, CO, O3, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4 were estimated based on insurance registration. We calculated the HRs of exposure at a standard deviation increment for 10 years to determine the dose–response effect between air pollutants and infertility.
Result:
Long-term exposure to SO2, CO, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4 was associated with increased infertility in women of reproductive age. Each standard deviation increase in exposure to these pollutants was associated with a higher incidence of infertility by 13%, 116%, 35%, 77%, 80%, 66%, 76%, 116%, 52%, and 181%, respectively. Conversely, ozone exposure was associated with a 52% reduction in infertility risk.
Conclusion:
This study demonstrates the significant impact of air pollution on female infertility, showing a clear dose–response relationship between exposure to various pollutants and infertility rates. These findings highlight the need for efforts to reduce air pollution and its effects on reproductive health. Further research is needed to understand the mechanisms and inform public health policies.
Supplementary Information:
The online version contains supplementary material available at 10.1186/s12889-025-24213-x.
Article type: Research Article
Keywords: Female infertility, Air pollution, Reproductive health
Affiliations: Department of Orthopedics, Taichung Veterans General University, Taichung, Taiwan; https://ror.org/05031qk94grid.412896.00000 0000 9337 0481Department of Obstetrics and Gynecology, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan; https://ror.org/05d9dtr71grid.413814.b0000 0004 0572 7372Division of General Practice, Department of Medical Education, Changhua Christian Hospital, Changhua, Taiwan; https://ror.org/05031qk94grid.412896.00000 0000 9337 0481Department of Orthopedics, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan; https://ror.org/05031qk94grid.412896.00000 0000 9337 0481Department of Orthopedics, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan; https://ror.org/05d9dtr71grid.413814.b0000 0004 0572 7372Department of Ophthalmology, Changhua Christian Hospital, Changhua, Taiwan; https://ror.org/05vn3ca78grid.260542.70000 0004 0532 3749Department of Post-Baccalaureate Medicine, College of Medicine, National Chung Hsing University, Taichung, Taiwan
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s12889-025-24213-x | PubMed: 41029231 | PMC: PMC12487008
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.8 MB)
Introduction
Female infertility refers to the incapacity to achieve a clinical pregnancy, impacting more than 186 million individuals globally [ref. 1]. It is estimated to affect approximately 1 in 7 couples in developed nations and about 8–12% of reproductive-aged couples worldwide [ref. 1]. The World Health Organization defines infertility as the inability to achieve a clinical pregnancy after 12 months or more of regular unprotected sexual intercourse [ref. 2]. Numerous factors that could affect the inherent fertility of couples have been acknowledged. These encompass the duration of undesired non-conception, the decline in female fertility associated with age, infertility attributed to medical conditions, sperm quality, the influence of endocrine-disrupting chemicals, and environmental origins [ref. 3].
Air pollution emerges as a significant health concern, correlating with heightened rates of both mortality and morbidity [ref. 4]. For example, it has been linked to elevated cancer risk, as well as cardiovascular and respiratory disorders in both adults and children [ref. 5]. Furthermore, it’s associated with negative perinatal outcomes and associated with increased mortality following hip fracture in older adults [ref. 6]. Several lines of evidence suggest that prenatal exposure to environmental pollutants may influence reproductive and endocrine health, with emerging research linking maternal exposure from passive smoking and traffic-related air pollution to alterations in neonatal hormonal profiles [ref. 7]. Two systematic reviews indicate a strong link between female infertility and air pollution [ref. 8]. Conforti et al. found that in the IVF population, nitrogen dioxide and ozone were associated with reduced live birth rates, while in the general population, particulate matter (PM2.5 and PM10), sulfur dioxide, carbon monoxide, and nitrogen dioxide were linked to reduced fecundability, miscarriage, and stillbirths due to air pollution exposure [ref. 8]. Another systematic review analyzed the impact of air pollution on fertility rates, observing a statistically significant reduction in fertility rates associated with higher traffic-related air pollution, particularly in relation to the coarse fraction of particulate matter (PM) [ref. 9]. Apart from the aforementioned study, animal research has explored the impact of particulate urban air pollution (PM 2.5) on reproductive health. These studies found that exposure to PM 2.5 is associated with a reduced number of viable fetuses, higher incidence of implantation failures, low birth weight, low placenta weight, changes in estrous cyclicity, decreased ovarian follicles count, and decreased fertility indices [ref. 10].
Given the paucity of clinical evidence regarding the impact of air pollutants on female infertility, we embarked on this retrospective study to investigate the potential influence of decade-long exposure to various air pollutants on the likelihood of infertility among Taiwanese women aged 15 to 60. Our hypothesis postulated that extended exposure to air pollution might affect the incidence of female infertility. Specifically, we examined the effects of 11 air pollution compounds: sulfur dioxide (SO2), carbon monoxide (CO), ozone (O3), particulate matter with a diameter of less than 10 μm (PM10), particulate matter with a diameter of less than 2.5 μm (PM2.5), nitrogen oxides (NOX), nitrogen monoxide (NO), nitrogen dioxide (NO2), total hydrocarbons (THC), nonmethane hydrocarbons (NMHC), and methane (CH4). Through the utilization of a population-based approach and comprehensive air pollutant exposure data, this study endeavors to bridge these significant knowledge gaps, offering insights into the intricate relationship between air pollution and the risk of female infertility.
Materials and methods
Data source
In this study, we retrieved data from the National Health Insurance Research Database (NHIRD) of Taiwan, which was launched by the Taiwan government in 1995 and collected comprehensive health care for 98.29% of the population [ref. 11]. The database provided nationwide medical information including gender, date of birth, employment, inpatient and outpatient diagnoses, medical procedures, drug usage, treatment duration, and medical costs of approximately 23 million insured individuals in Taiwan [ref. 12]. We identified baseline information by using the Longitudinal Health Insurance Database 2000, a subset of the NHIRD containing 1 million randomly enrolled patients. The Research Ethics Committee approved this study protocol (IRB: 240310). This study was conducted in accordance with the principles of the Declaration of Helsinki and relevant regulations. Due to the anonymity of patient data in NHIRD, patient-informed consent was not required for this study.
Study population
We only included patients with complete available data, while those with missing, inconsistent, or unknown records of baseline information such as gender and birth year were therefore precluded. The enrollment population was defined as those who were female gender between July 1, 2003, and December 31, 2013. The further criteria for exclusion were detailed as follows, 1) subjects aged under 15 or over 60 at the beginning of the study, 2) those diagnosed with infertility, history of prior surgery of the genital organs, or history of radiotherapy before the beginning of the study, 3) subjects whose survival data were before the beginning of the study, and 4) subjects’ follow-up were under 5 years. To ensure adequate accumulation of long-term exposure and reliable outcome ascertainment, only participants with at least 5 years of follow-up were included. This threshold improves data stability and reduces the likelihood of misclassification.
Exposure data collection
We obtained the cumulative daily average concentration of eleven air pollutants, including sulfur dioxide (SO2), carbon monoxide (CO), ozone (O3), particulate matter < 10 μm in size (PM10), particulate matter < 2.5 μm in size (PM2.5), nitrogen oxides (NOX), nitrogen monoxide (NO), nitrogen dioxide (NO2), total hydrocarbons (THC), nonmethane hydrocarbons (NMHC), and methane (CH4), from the measurements recorded by the 76 monitoring stations maintained by the Taiwan Environmental Protection Administration (EPA), Executive Yuan. Data were collected for the period from July 1, 1993, to December 31, 2013. The cumulative daily average concentration of each pollutant was calculated from 10 years prior to the end of follow-up. Daily concentrations were extracted from the nearest monitoring station linked to each residence, and a time-weighted average was calculated across all registered addresses during this period. To account for residential mobility during the study period, we linked the air pollutant concentrations to each individual’s residential postal codes based on their insurance registration records. For participants who changed residence, exposure levels were updated accordingly, and the cumulative exposure was calculated as a time-weighted average based on the duration spent at each registered location.
Outcomes and confounders
The development of infertility was the major outcome of interest. Infertility diagnosis was based on ICD-9-CM codes 628.x, confirmed by ≥ 2 outpatient visits or ≥ 1 inpatient claim. We followed every patient until the primary end-point defined by either the development of infertility (withdrawal from the NHI program) or end of the study period (December 31, 2013), with survival time measured in months. Several confounders were introduced and adjusted in this study, including age, urbanization level, insurance amount, CCI score, ambient temperature, season, and lag0-2. Above them, patient data such as age, insurance amount, and CCI score were obtained from the NHIRD database. Specifically, the insurance amount was measured as the average value during the period of air pollutant exposure assessment, while the comorbidities were defined before the survival date. The meteorological factor of ambient temperature was collected from the EPA. The urbanization level was recorded according to the patient’s residence at the beginning of the follow-up period, while the season was defined by date. We also adopted the 3-day moving average of current day and two preceding days concentrations of air pollutants before the primary end-point (lag0-2) for adjusted confounders, based on the largest effect estimate found in previous literature [ref. 13]. Confounders were selected based on literature linking them to infertility and availability in the NHIRD. Lifestyle-related variables not directly available (e.g., smoking, BMI) were partially captured through insurance income, urbanization, and comorbidity proxies.
Statistical analysis
To demonstrate patient characteristics associated with air pollution, patients were divided by the exposure concentration of each pollutant into 3 tertiles and were compared using the chi-square chi-squared test or one-way analysis of variance among tertiles, post hoc tests were performed to explore differences when significance was indicated in the one-way analysis of variance. We plotted crude cumulative incidence curves of infertility for participants in three tertiles, with the difference between tertiles assessed by log-rank tests. We conducted Cox regression models to explore the dose–response effect between air pollutants and infertility risk by calculating the HRs of exposure at a standard deviation increment for 10 years. The regression models were adjusted for the abovementioned confounding factors. To address potential confounding from multiple coexisting pollutants, we included additional pollutants with low correlation (r < 0.3) to the primary exposure in the adjusted models. This reduces confounding without introducing multicollinearity. We also constructed two-pollutant models to test the robustness of the observed associations and assess the independent effect of each pollutant. All tests were 2-sided, and statistically, significance was considered when p < 0.05. All analyses were performed using the MetaTrial Research Platform. The software was developed by Biomedica Corporation, Taiwan.
Results
Study population
After the inclusion of patients with complete available data (n = 882391), those who were female gender were identified (n = 417251). The following exclusion criteria were then applied: (1) age under 15 or over 60 years at study initiation (n = 132,930); (2) diagnosis of infertility, history of prior genital organ surgery, or history of radiotherapy before study initiation (n = 1,645); (3) participants who died before the study start date (n = 35,825); and (4) follow-up duration under 5 years (n = 32,823). Consequently, 232,125 participants were included in the final analysis, with the selection process illustrated in Fig. 1.
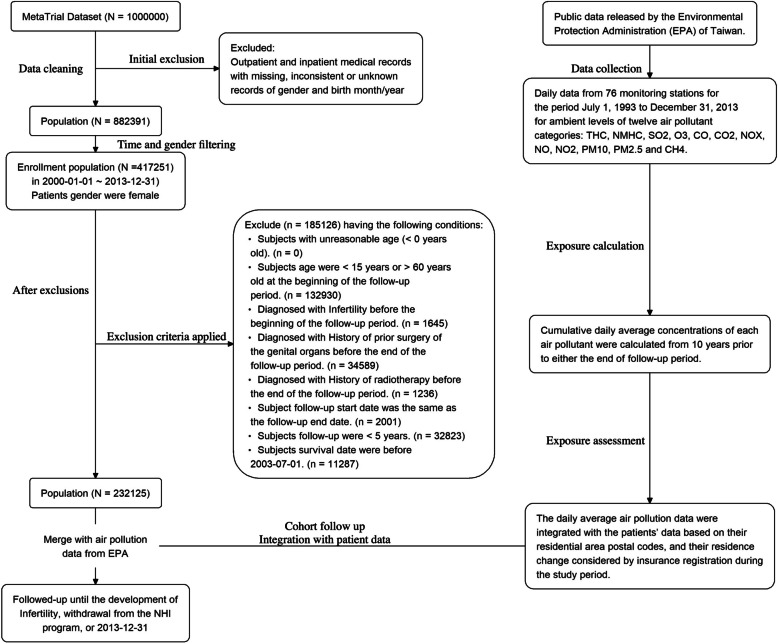
Characteristics and descriptive results
Table 1 presents the characteristics of the included cohort (n = 232,125), among whom 929 individuals (12.51%) were diagnosed with infertility during the follow-up period. The distribution of comorbidities is also shown. Characteristics stratified by tertiles of pollutant exposure are detailed in Supplementary Table 1, while mean concentrations and distributions of 10-year pollutant exposures are presented in Table 2.
Table 1: Baseline characteristics
| Characteristic | N (%) |
|---|---|
| Infertility | 4981 (2.15) |
| Age, years | |
| 15–35 | 121732 (52.44) |
| 36–60 | 110393 (47.56) |
| Mean ± SD | 35.08 ± 12.45 |
| Urbanization level | |
| 1 (highest) | 138881 (59.83) |
| 2 | 71156 (30.65) |
| 3 | 14280 (6.15) |
| 4 (lowest) | 1826 (0.79) |
| unknown | 5982 (2.58) |
| Insurance amount, NT$ | |
| financially dependent | 1343 (0.58) |
| 1–19999 | 79868 (34.41) |
| 20000–39999 | 102875 (44.32) |
| > = 40000 | 41027 (17.67) |
| unknown | 7012 (3.02) |
| CCI score | |
| Mean ± SD | 1.10 ± 1.68 |
| Comorbidity | |
| Inflammatory disease of the ovary, fallopian tube, pelvic cellular tissue, and peritoneum | 35926 (15.48) |
| Inflammatory disease of the uterus | 12241 (5.27) |
| Inflammatory disease of the cervix, vagina, and vulva | 107803 (46.44) |
| Endometriosis | 8828 (3.80) |
| Hypertension | 38765 (16.70) |
| Diabetes mellitus | 27325 (11.77) |
| Hypertriglyceridemia | 286 (0.12) |
| Hypercholesterolemia | 29970 (12.91) |
| Coronary artery disease | 21789 (9.39) |
| Disorders of eating | 302 (0.13) |
SD standard deviation, CCI score, Charlson Comorbidity Index score
Table 2: Mean and distribution of air pollutants over the exposure period
| SO2 (ppb) | CO (ppm) | O3 (ppb) | PM10 (μg/m3) | PM2.5 (μg/m3) | NOX (ppb) | NO (ppb) | NO2 (ppb) | THC (ppm) | NMHC (ppm) | CH4 (ppm) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 4.14 | 0.53 | 28.67 | 54.19 | 32.43 | 25.72 | 7.54 | 18.18 | 2.24 | 0.29 | 1.95 |
| SD | 1.35 | 0.11 | 1.73 | 8.93 | 6.05 | 6.8 | 3.68 | 3.48 | 0.12 | 0.08 | 0.07 |
| T1 | 3.59 | 0.48 | 27.77 | 47.66 | 27.67 | 21.44 | 4.93 | 16.55 | 2.17 | 0.25 | 1.93 |
| T2 | 4.04 | 0.59 | 29.11 | 56.18 | 34.95 | 29.6 | 8.51 | 20.49 | 2.31 | 0.33 | 1.97 |
SD standard deviation, T1 33.33 percentile, T2 66.66 percentile, ppb parts per billion; ppm, parts per million, μg/m3 microgram/cubic meter, SO2 sulfur dioxide, CO carbon monoxide, O3 ozone, PM10 particulate matter < 10 μm in size, PM2.5 particulate matter < 2.5 μm in size, NOX nitrogen oxides, NO nitrogen monoxide, NO2 nitrogen dioxide, THC total hydrocarbons, NMHC nonmethane hydrocarbons, CH4 methane
Cumulative incidence of infertility
For the outcome of interest, the incidence of infertility, we exhibited the results among tertiles in Table 3, with the p-value of one-way analysis of variance and post-hoc analysis. The cumulative incidence curves (Fig. 2) illustrate variation in infertility occurrence across different exposure levels, suggesting pollutant-specific patterns.
Table 3: Incidence of infertility among tertiles
| Pollutants | Tertiles of average daily exposure, n (%) | P value | Total, n (%) | ||
|---|---|---|---|---|---|
| T1 (lowest) | T2 | T3 (highest) | |||
| SO2 | 974/77141 (1.26) | 1799/75237 (2.39) | 2208/79747 (2.77) | < 0.001 | 4981/232125 (2.15) |
| CO | 443/77375 (0.57) | 1351/77375 (1.75) | 3187/77375 (4.12) | < 0.001 | 4981/232125 (2.15) |
| O3 | 3579/77372 (4.63) | 796/77365 (1.03) | 606/77388 (0.78) | < 0.001 | 4981/232125 (2.15) |
| PM10 | 1243/77360 (1.61) | 1681/72522 (2.32) | 2057/82243 (2.50) | < 0.001 | 4981/232125 (2.15) |
| PM2.5 | 832/77369 (1.08) | 2077/75510 (2.75) | 2064/79229 (2.61) | < 0.001 | 4973/232108 (2.14) |
| NOX | 603/77370 (0.78) | 1778/77380 (2.30) | 2600/77375 (3.36) | < 0.001 | 4981/232125 (2.15) |
| NO | 561/77375 (0.73) | 1839/77374 (2.38) | 2581/77376 (3.34) | < 0.001 | 4981/232125 (2.15) |
| NO2 | 675/77348 (0.87) | 1506/77402 (1.95) | 2800/77375 (3.62) | < 0.001 | 4981/232125 (2.15) |
| THC | 268/76816 (0.35) | 1084/76816 (1.41) | 3608/76817 (4.70) | < 0.001 | 4960/230449 (2.15) |
| NMHC | 541/76813 (0.70) | 1786/76819 (2.32) | 2633/76817 (3.43) | < 0.001 | 4960/230449 (2.15) |
| CH4 | 335/76816 (0.44) | 451/72513 (0.62) | 4174/81120 (5.15) | < 0.001 | 4960/230449 (2.15) |
SO2 sulfur dioxide, CO carbon monoxide, O3 ozone, PM10 particulate matter < 10 μm in size, PM2.5 particulate matter < 2.5 μm in size, NOX nitrogen oxides, NO nitrogen monoxide, NO2 nitrogen dioxide, THC total hydrocarbons, NMHC nonmethane hydrocarbons, CH4 methane
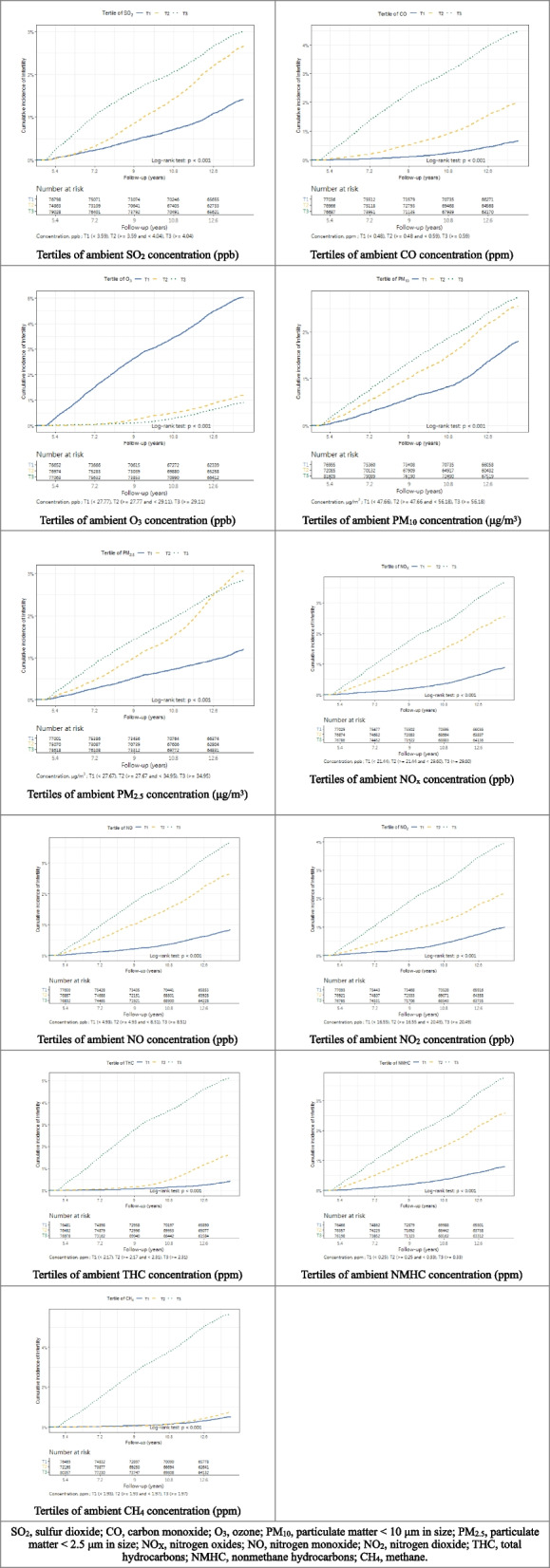
The dose–response effect between air pollutants and the incidence of infertility
To explore the dose–response effect between the concentrations of air pollutants and the incidence of infertility, we performed Cox regression models and calculated the HRs of exposure at a standard deviation increment for 10 years. The adjusted hazard ratios (HRs) are listed in Table 4. Each SD increase in the average exposure concentration of SO₂, CO, PM₁₀, PM₂.₅, NOX, NO, NO₂, THC, NMHC, and CH₄ was associated with a significantly elevated hazard of infertility by 13%, 116%, 35%, 77%, 80%, 66%, 76%, 116%, 52%, and 181%, respectively. In contrast, O₃ exposure was negatively associated with infertility, with a 52% lower hazard (HR: 0.48), indicating a statistically significant protective trend. To further illustrate the dose–response relationship, we categorized the exposure concentrations into tertiles and conducted Cox regression analyses using the lowest tertile (T1) as the reference. The adjusted hazard ratios for the second (T2) and third (T3) tertiles are presented in Table 5. A consistent trend of increasing hazard of infertility was observed across tertiles for most pollutants, particularly SO₂, CO, PM₁₀, PM₂.₅, NOX, NO, NO₂, THC, NMHC, and CH₄. Conversely, higher O₃ exposure tertiles were associated with a significantly reduced hazard, reinforcing the negative relationship seen in the standard deviation-based model.
Table 4: Hazard ratios for incidence of infertility of long-term exposure at an SD increment
| Pollutant | Adjusted HR (95% CI) | P value | SD |
|---|---|---|---|
| SO2 | 1.13 (1.09,1.18) | < 0.001 | 1.13 ppb |
| CO | 2.16 (2.09,2.22) | < 0.001 | 0.11 ppm |
| O3 | 0.48 (0.47,0.49) | < 0.001 | 1.73 ppb |
| PM10 | 1.35 (1.28,1.43) | < 0.001 | 8.93 μg/m3 |
| PM2.5 | 1.77 (1.67,1.88) | < 0.001 | 6.05 μg/m3 |
| NOX | 1.80 (1.73,1.86) | < 0.001 | 6.80 ppb |
| NO | 1.66 (1.61,1.72) | < 0.001 | 3.68 ppb |
| NO2 | 1.76 (1.69,1.83) | < 0.001 | 3.48 ppb |
| THC | 2.16 (2.09,2.22) | < 0.001 | 0.12 ppm |
| NMHC | 1.52 (1.47,1.56) | < 0.001 | 0.08 ppm |
| CH4 | 2.81 (2.72,2.90) | < 0.001 | 0.07 ppm |
Cox regression models were adjusted for age, urbanization level, insurance amount, CCI score, Inflammatory disease of the ovary, fallopian tube, pelvic cellular tissue, and peritoneum, Inflammatory disease of the uterus, Inflammatory disease of the cervix, vagina, and vulva, Endometriosis, Hypertension, Diabetes mellitus, Hypertriglyceridemia, Hypercholesterolemia, Coronary artery disease, ambient temperature, lag0-2, season, and controlled pollutants
HR hazard ratio, CI confidence interval, SD standard deviation, SO2 sulfur dioxide, CO carbon monoxide, O3 ozone, PM10 particulate matter < 10 μm in size, PM2.5 particulate matter < 2.5 μm in size, NOX nitrogen oxides, NO nitrogen monoxide, NO2 nitrogen dioxide, THC total hydrocarbons, NMHC nonmethane hydrocarbons, CH4 methane
Table 5: The dose–response association between air pollutants and infertility risk
| Pollutant category | Tertiles of average daily pollutant | Population | Infertility | PY | Incidence Rate | Adjusted HR (95% CI) | P value |
|---|---|---|---|---|---|---|---|
| SO2 | T1 (lowest) | Total (N = 232125) | 974 | 1017086 | 9.58 | 1.00 (reference) | – |
| T2 | 1799 | 985662 | 18.25 | 1.22 (1.13,1.32) | < 0.001 | ||
| T3 (highest) | 2208 | 1036732 | 21.30 | 1.71 (1.58,1.86) | < 0.001 | ||
| CO | T1 (lowest) | Total (N = 232125) | 443 | 1022715 | 4.33 | 1.00 (reference) | – |
| T2 | 1351 | 1014329 | 13.32 | 1.67 (1.50,1.87) | < 0.001 | ||
| T3 (highest) | 3187 | 1002435 | 31.79 | 3.47 (3.11,3.88) | < 0.001 | ||
| O3 | T1 (lowest) | Total (N = 232125) | 3579 | 998055 | 35.86 | 1.00 (reference) | – |
| T2 | 796 | 1017205 | 7.83 | 0.30 (0.28,0.33) | < 0.001 | ||
| T3 (highest) | 606 | 1024219 | 5.92 | 0.24 (0.21,0.26) | < 0.001 | ||
| PM10 | T1 (lowest) | Total (N = 232125) | 1243 | 1020533 | 12.18 | 1.00 (reference) | – |
| T2 | 1681 | 948868 | 17.72 | 1.36 (1.26,1.47) | < 0.001 | ||
| T3 (highest) | 2057 | 1070078 | 19.22 | 2.01 (1.82,2.22) | < 0.001 | ||
| PM2.5 | T1 (lowest) | Total (N = 232108) | 832 | 1021415 | 8.15 | 1.00 (reference) | – |
| T2 | 2077 | 988029 | 21.02 | 1.23 (1.13,1.33) | < 0.001 | ||
| T3 (highest) | 2064 | 1029946 | 20.04 | 2.25 (2.02,2.51) | < 0.001 | ||
| NOX | T1 (lowest) | Total (N = 232125) | 603 | 1021760 | 5.90 | 1.00 (reference) | – |
| T2 | 1778 | 1009107 | 17.62 | 1.69 (1.53,1.86) | < 0.001 | ||
| T3 (highest) | 2600 | 1008611 | 25.78 | 2.34 (2.11,2.60) | < 0.001 | ||
| NO | T1 (lowest) | Total (N = 232125) | 561 | 1021043 | 5.49 | 1.00 (reference) | – |
| T2 | 1839 | 1009660 | 18.21 | 1.72 (1.56,1.90) | < 0.001 | ||
| T3 (highest) | 2581 | 1008777 | 25.59 | 2.57 (2.30,2.86) | < 0.001 | ||
| NO2 | T1 (lowest) | Total (N = 232125) | 675 | 1021060 | 6.61 | 1.00 (reference) | – |
| T2 | 1506 | 1011807 | 14.88 | 1.44 (1.31,1.59) | < 0.001 | ||
| T3 (highest) | 2800 | 1006612 | 27.82 | 2.13 (1.93,2.34) | < 0.001 | ||
| THC | T1 (lowest) | Total (N = 230449) | 268 | 1015554 | 2.64 | 1.00 (reference) | – |
| T2 | 1084 | 1012641 | 10.70 | 1.12 (0.97,1.29) | 0.132 | ||
| T3 (highest) | 3608 | 989019 | 36.48 | 3.53 (3.07,4.06) | < 0.001 | ||
| NMHC | T1 (lowest) | Total (N = 230449) | 541 | 1013917 | 5.34 | 1.00 (reference) | – |
| T2 | 1786 | 1003607 | 17.80 | 1.30 (1.16,1.45) | < 0.001 | ||
| T3 (highest) | 2633 | 999690 | 26.34 | 1.85 (1.65,2.08) | < 0.001 | ||
| CH4 | T1 (lowest) | Total (N = 230449) | 335 | 1014880 | 3.30 | 1.00 (reference) | – |
| T2 | 451 | 960311 | 4.70 | 1.03 (0.89,1.20) | 0.670 | ||
| T3 (highest) | 4174 | 1042023 | 40.06 | 2.99 (2.65,3.38) | < 0.001 |
PY person years, HR hazard ratio, CI confidence interval, SO2 sulfur dioxide, CO carbon monoxide, O3 ozone, PM10 particulate matter < 10 μm in aerodynamic diameter, PM2.5 particulate matter < 2.5 μm in aerodynamic diameter, NOX nitrogen oxides, NO nitric oxide, NO2 nitrogen dioxide, THC total hydrocarbons, NMHC non-methane hydrocarbon, CH4 methane
The tertile values, in ppb (SO2, O3, NOX, NO, NO2); ppm (CO, THC, NMHC, CH4); and μg/m3 (PM10, PM2.5) were as follows:
SO2 (T1: < 3.59, T2: ≥ 3.59 and < 4.04, T3: ≥ 4.04); CO (T1: < 0.48, T2: ≥ 0.48 and < 0.59, T3: ≥ 0.59); O3 (T1: < 27.77, T2: ≥ 27.77 and < 29.11, T3: ≥ 29.11); PM10 (T1: < 47.66, T2: ≥ 47.66 and < 56.18, T3: ≥ 56.18); PM2.5 (T1: < 27.67, T2: ≥ 27.67 and < 34.95, T3: ≥ 34.95); NOX (T1: < 21.44, T2: ≥ 21.44 and < 29.60, T3: ≥ 29.60); NO (T1: < 4.93, T2: ≥ 4.93 and < 8.51, T3: ≥ 8.51); NO2 (T1: < 16.55, T2: ≥ 16.55 and < 20.49, T3: ≥ 20.49); THC (T1: < 2.17, T2: ≥ 2.17 and < 2.31, T3: ≥ 2.31); NMHC (T1: < 0.25, T2: ≥ 0.25 and < 0.33, T3: ≥ 0.33); CH4 (T1: < 1.93, T2: ≥ 1.93 and < 1.97, T3: ≥ 1.97)
per 10,000 person-years
To assess age-related confounding, a sensitivity analysis restricted to women aged 15–45 years was performed. Results remained consistent with the primary analysis. For example, PM₂.₅ exposure remained significantly associated with increased infertility hazard (HR: 1.74, 95% CI: 1.61–1.88, p < 0.001). These results are presented in Supplementary Tables S4 and S5.
Discussion
Principal results
This study found that long-term exposure to SO2, CO, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4 was associated with the incidence of infertility in age-reproductive women. In females, each increase of a standard deviation in average exposure concentration of SO2, CO, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4, indicated a higher incidence of infertility of 13%, 116%, 35%, 77%, 80%, 66%, 76%, 116%, 52%, and 181%, respectively. On the contrary, we found a significant reduction of 52% in the hazard ratio with ozone. Given that fertility typically declines after age 40, the inclusion of women aged 46–60 in our main analysis may raise concerns about age-related bias. To address this, we performed a sensitivity analysis focusing on women aged 15–45 years, consistent with the WHO-defined reproductive age range. The results were highly consistent with the primary findings, suggesting that our conclusions are robust across different age specifications. This supports the validity of our approach while preserving the advantages of using a broad population-based cohort.
Comparison with prior work
Our findings suggested that long-term exposure to SO2, CO, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4 acted consistently as a risk factor for higher incidence of infertility. Previous literature had discovered significant associations between increased levels of traffic-related air pollution, particularly the coarse fraction of particulate matter, and reduced fertility rates, with chronic exposure being more influential in infertility risk compared to short-term exposure [ref. 14]. The same trends were indicated in the infertility risk, with the pollutants of SO2, NO2. In a polluted area, Legro et al. found variable, cycle-dependent effects of declining air quality on reproductive outcomes after in vitro fertilization, with consistent associations between increased NO2 and lower live birth rates [ref. 15]. Furthermore, there is a suggestion that the likelihood of conception at the first unprotected menstrual cycle may decrease for couples exposed to average SO2 levels exceeding 40 μg/m3 during the second month before conception [ref. 16]. In comparison to Western countries, the PM₂.₅ concentrations observed in our study are relatively high. The average annual PM₂.₅ concentration in Taiwan was reported as 20.2 μg/m3 in 2023, which surpasses the WHO’s recommended limit of 5 μg/m3. In contrast, the average PM₂.₅ concentrations in 2015 were 13.9 μg/m3 in Europe and 7–12 μg/m3 in the United States [ref. 17]. These elevated exposure levels in Taiwan highlight the potential public health burden and underscore the global relevance of our findings, especially for regions facing similar air pollution challenges.
The ovarian reserve is a crucial measure of a woman’s reproductive potential, primarily determined by the quantity and quality of oocytes within the ovaries [ref. 18]. A noteworthy big-data approach by Santi et al. examined how environmental factors, particularly rising air pollution, impact anti-mullerian hormone (AMH) serum levels. While genetics play a major role in ovarian reserve at birth, this study suggests that environmental factors can also influence the decline in AMH and ovarian reserve during adulthood [ref. 19]. In contrast to our findings, Wu et al. [ref. 20] suggest that extended exposure to the air pollutant SO2 is linked to lower antral follicle count and a higher risk of poor ovarian response (< 4 oocytes retrieved), respectively, while NO2, PM10, PM2.5, CO, and O3 show no significant associations. These findings raise concerns about the potential adverse impact of atmospheric SO2 exposure on women’s ovarian reserve.
O3, a secondary air pollutant formed from the combination of hydrocarbons and nitrogen oxides in sunlight, exhibits potential non-linearities in this process [ref. 21]. Legro et al. discovered a connection between O3 concentration at a patient’s address and the likelihood of a live birth [ref. 15]. However, when accounting for NO2 and in vitro fertilization laboratory interactions, this association became insignificant. Moreover, Boulet et al. observed a weak positive correlation between O3 and implantation or live birth rates. Wu et al. identified higher O3 exposure in women with normal ovarian reserve (NOR) compared to poor responders [ref. 20]. However, multipollutant models showed no significant correlations.
Possible mechanism
While the precise mechanisms behind air pollution-related female infertility are not yet fully understood, we propose a potential mechanism: maternal exposure to contaminants during the pre- and peri-conceptional periods could lead to abnormalities in oocytes, embryos, and fetuses [ref. 22]. This could potentially hinder safe childbirth, impact overall health and mental well-being, and thereby increase vulnerability to and susceptibility to infertility. Air pollutants disrupt both animal and human gametogenesis, reducing reproductive efficacy [ref. 8]. These endocrine-disrupting properties could activate the Ras/Erk pathway via interactions with nuclear receptors like estrogen or androgen receptors and specific cytosolic targets, while diesel exhaust particles and PM2.5 impact ovarian function by disrupting the endocrine system, increasing oxidative stress and inflammation, and activating specific targets that trigger abnormal MAPK signaling [ref. 23]. Indeed, in a recent study of 777 men, increased air pollutant concentrations were significantly associated with both the hypomethylation of F3, ICAM-1, and TLR-2, and the hypermethylation of IFN-γ and IL-6 [ref. 24]. What’s most notable is that research has demonstrated that adding antioxidant factors to ovarian stimulation can enhance reproductive outcomes in women with polycystic ovarian syndrome [ref. 25].
Regarding ovarian function, a study did find a significant negative association between SO2 exposure throughout the entire antral follicle development stage and AFC [ref. 26]. This suggests that during the follicular vascularization transition stage, SO2 might be expedited in reaching follicles and granulosa cells, potentially contributing to impaired follicle development, hastened follicle loss, or altered ovarian function. The ovarian toxicity of environmental contaminants, characterized by meiotic disruption in oocyte formation resulting from genetic or epigenetic aberrations, can affect dormant follicles within the primordial pool that undergo growth stimulation and have the potential to develop into the antral stage [ref. 27]. Subsequently, these antral candidates are typically chosen to progress to the pre-ovulatory stage, marked by the transition from primary to pre-antral, during which the oocyte within the follicle enlarges, granulosa cells proliferate, and a layer of theca cells forms outside the granulosa cells, serving as a conduit for transporting steroids and growth factors to the granulosa cells to promote follicle vascularization [ref. 28]. Additionally, this bloodstream enables toxins to reach follicles, where they can suppress granulosa cell proliferation and growth. Furthermore, SO2 toxicology in mammalian organ development involves cellular injuries caused by inflammation and oxidative stress, such as lipid peroxidation, DNA damage, protein oxidation, and interference with signal transduction [ref. 29].
While ozone (O₃) is traditionally considered a harmful air pollutant, emerging evidence suggests that under certain controlled clinical settings with specific dosing protocols, ozone therapy may exert anti-inflammatory and antioxidant effects. For instance, medical ozone therapy administered in controlled healthcare environments has shown potential benefits in conditions such as HIV/AIDS, fibromyalgia, and peripheral arterial disease [ref. 30–ref. 32]. Some studies have explored its use in reproductive medicine, including pelvic inflammatory disease and tubal infertility [ref. 31]. It is crucial to emphasize that these therapeutic effects are achieved through controlled medical administration and are not equivalent to ambient environmental ozone exposure, which remains harmful to human health. However, the negative association observed in our study between ozone exposure and infertility risk is counterintuitive and must be interpreted with caution. Ambient ozone levels are influenced by seasonal and geographic factors and may act as a proxy for lower traffic-related co-pollutants. Additionally, negative confounding or interaction with other pollutants cannot be ruled out. Further research is needed to clarify this unexpected finding. This negative trend may reflect negative confounding, as ozone concentrations are often higher in suburban areas with lower levels of traffic-related pollutants. It is also possible that ecological bias affected the results, given that area-level monitoring data may not precisely represent individual-level exposure. These findings highlight the need for future studies to incorporate more detailed exposure metrics and consider pollutant interactions to better understand the role of ozone in reproductive health.
This study possesses several notable strengths. Firstly, it is the inaugural investigation to reveal the association between long-term exposure to air pollution and female infertility. Secondly, our analysis encompassed a comprehensive range of pollutants and utilized a significantly large sample, bolstering the statistical robustness of the findings. Thirdly, we conducted stratified analyses considering potential confounding factors such as age, urbanization level, insurance amount, CCI score, ambient temperature, season, and lag0-2, along with a wide spectrum of demographic characteristics. Furthermore, in registry-based studies, a minimum follow-up threshold is often necessary to ensure valid exposure-outcome relationships. Our choice of a 5-year minimum follow-up reflects this principle and strengthens the internal validity of the analysis. While it may reduce inclusion of short-term registrants, the large sample size and nationwide coverage help preserve overall representativeness. Lastly, the population-based data used in this study is representative of the general population in Taiwan.
This study has several limitations. First, key reproductive and lifestyle factors such as smoking status, alcohol consumption, body mass index (BMI), marital status, and oral contraceptive use were not available in the NHIRD. Although proxy variables—such as comorbidities, insurance level, and urbanization—were included in the adjusted models, the potential for residual confounding remains. Second, clinical variables such as disease severity, laboratory data, and symptomatology were unavailable, limiting the granularity of outcome characterization. Third, both exposure and outcome misclassification are possible. Exposure was estimated based on insurance-registered residential postal codes, which may not reflect actual pollutant exposure due to mobility or address reporting errors. Although we incorporated updated residential records to reduce spatial misclassification, some inaccuracy may persist. Outcome misclassification may also occur, particularly in defining infertility, as the database does not capture fertility intention, menopausal status, male partner fertility status, or utilization of assisted reproductive technologies—all of which may influence diagnostic classification. Additionally, death was inferred from withdrawal from the National Health Insurance program, which may not be completely accurate. Fourth, the retrospective nature of the study limits causal inference, and unmeasured or unknown confounders cannot be excluded. Finally, while the use of a five-year follow-up period strengthens the exposure-outcome temporality, it may inadvertently exclude short-term insurance registrants, potentially introducing selection bias. Nonetheless, the large sample size and nationwide coverage enhance the generalizability of the findings. Despite these limitations, the results provide consistent epidemiologic evidence supporting the association between long-term exposure to SO₂, CO, PM₁₀, PM₂.₅, NOₓ, NO, NO₂, THC, NMHC, and CH₄ and increased risk of female infertility.
Conclusion
This study illustrates the considerable influence of air pollution on female infertility, revealing a distinct dose–response correlation between exposure to different pollutants and infertility rates. Prolonged exposure to SO2, CO, PM10, PM2.5, NOX, NO, NO2, THC, NMHC, and CH4 heightened infertility risks, while ozone exposure mitigated them. Our results underscore the importance of heightened awareness among clinicians and government stakeholders. In particular, targeted urban planning and environmental policies aimed at reducing NO₂ and PM exposure among women of reproductive age may serve as crucial public health interventions. They emphasize the necessity for extensive initiatives to curb air pollution and its impact on reproductive health. Further investigation is warranted to elucidate the underlying mechanisms and guide public health policies.
Supplementary Materials
References
- MC Inhorn, P Patrizio. Infertility around the globe: new thinking on gender, reproductive technologies and global movements in the 21st century. Hum Reprod Update, 2015. [DOI | PubMed]
- F Zegers-Hochschild, GD Adamson, S Dyer. The international glossary on infertility and fertility care, 2017. Fertil Steril, 2017. [DOI | PubMed]
- C Alviggi, R Guadagni, A Conforti, G Coppola, S Picarelli, P De Rosa. Association between intrafollicular concentration of benzene and outcome of controlled ovarian stimulation in IVF/ICSI cycles: a pilot study. J Ovar Res, 2014
- AJ Cohen, M Brauer, R Burnett, HR Anderson, J Frostad, K Estep. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet, 2017. [DOI | PubMed]
- M Richardson, M Guo, B Fauser, N Macklon. Environmental and developmental origins of ovarian reserve. Hum Reprod Update, 2014. [DOI | PubMed]
- SH Chuang, YJ Kuo, SW Huang, HW Zhang, HC Peng, YP Chen. Association between long-term exposure to air pollution and the rate of mortality after hip fracture surgery in patients older than 60 years: nationwide cohort study in Taiwan. JMIR Public Health Surveill, 2024. [DOI | PubMed]
- S Dehghani, M Fararouei, A Rafiee, L Hoepner, V Oskoei, M Hoseini. Prenatal exposure to polycyclic aromatic hydrocarbons and effects on neonatal anthropometric indices and thyroid-stimulating hormone in a Middle Eastern population. Chemosphere, 2022. [DOI | PubMed]
- A Conforti, M Mascia, G Cioffi, C De Angelis, G Coppola, P De Rosa. Air pollution and female fertility: a systematic review of literature. Reprod Biol Endocrinol, 2018. [DOI | PubMed]
- MAC Vizcaíno, M Gonzalez-Comadran, B Jacquemin. Outdoor air pollution and human infertility: a systematic review. Fertil Sterility, 2016. [DOI]
- MM Veras, NR Damaceno-Rodrigues, RMG Silva, JN Scoriza, PHN Saldiva, EG Caldini. Chronic exposure to fine particulate matter emitted by traffic affects reproductive and fetal outcomes in mice. Environ Res, 2009. [DOI | PubMed]
- CL Cheng, YHY Kao, SJ Lin, CH Lee, ML Lai. Validation of the National Health Insurance Research Database with ischemic stroke cases in Taiwan. Pharmacoepidemiol Drug Saf, 2011. [DOI | PubMed]
- AW Hsing, JP Ioannidis. Nationwide population science: lessons from the Taiwan national health insurance research database. JAMA Intern Med, 2015. [DOI | PubMed]
- R Chen, P Yin, X Meng, C Liu, L Wang, X Xu. Fine particulate air pollution and daily mortality. A nationwide analysis in 272 Chinese cities. Am J Respir Crit Care Med, 2017. [DOI | PubMed]
- S Mahalingaiah, J Hart, F Laden, L Farland, M Hewlett, J Chavarro. Adult air pollution exposure and risk of infertility in the Nurses’ Health Study II. Hum Reprod, 2016. [DOI | PubMed]
- RS Legro, MV Sauer, GL Mottla, KS Richter, X Li, WC Dodson. Effect of air quality on assisted human reproduction. Hum Reprod, 2010. [DOI | PubMed]
- J Dejmek, R Jelínek, I Solansky’, I Benes, RJ Srám. Fecundability and parental exposure to ambient sulfur dioxide. Environ Health Perspect, 2000. [DOI | PubMed]
- N Li, R Friedrich, C Schieberle. Exposure of individuals in Europe to air pollution and related health effects. Front Public Health, 2022. [DOI | PubMed]
- 18.Medicine PCotASfR. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertility and sterility. 2015;103(3):e9-e17.
- 19.Santi D, La Marca A, Michelangeli M, Casonati A, Grassi R, Baraldi E, et al., editors. Ovarian reserve and exposure to environmental pollutants (ORExPo study). Endocrine Abstracts; 2019: Bioscientifica.
- S Wu, G Hao, Y Zhang, X Chen, H Ren, Y Fan. Poor ovarian response is associated with air pollutants: a multicentre study in China. EBioMedicine, 2022. [DOI | PubMed]
- S Zhou, Y Xi, Y Chen, Z Zhang, C Wu, W Yan. Ovarian dysfunction induced by chronic whole-body PM2.5 exposure. Small, 2020. [DOI]
- R Canipari, L De Santis, S Cecconi. Female fertility and environmental pollution. Int J Environ Res Public Health, 2020. [DOI | PubMed]
- MG Palmerini, G Zhurabekova, A Balmagambetova, SA Nottola, S Miglietta, M Belli. The pesticide Lindane induces dose-dependent damage to granulosa cells in an in vitro culture. Reprod Biol, 2017. [DOI | PubMed]
- M-A Bind, J Lepeule, A Zanobetti, A Gasparrini, AA Baccarelli, BA Coull. Air pollution and gene-specific methylation in the normative aging study: association, effect modification, and mediation analysis. Epigenetics, 2014. [DOI | PubMed]
- C Alviggi, F Cariati, A Conforti, P De Rosa, R Vallone, I Strina. The effect of FT500 Plus® on ovarian stimulation in PCOS women. Reprod Toxicol, 2016. [DOI | PubMed]
- X Feng, J Luo, X Wang, W Xie, J Jiao, X Wu. Association of exposure to ambient air pollution with ovarian reserve among women in Shanxi province of north China. Environ Pollut, 2021. [DOI | PubMed]
- FJ Broekmans, D de Ziegler, CM Howles, A Gougeon, G Trew, F Olivennes. The antral follicle count: practical recommendations for better standardization. Fertil Steril, 2010. [DOI | PubMed]
- 28.Rimon-Dahari N, Yerushalmi-Heinemann L, Alyagor L, Dekel N. Ovarian folliculogenesis. Molecular mechanisms of cell differentiation in gonad development. 2016:167–90.
- S Petruzzi, B Musi, G Bignami. Acute and chronic sulphur dioxide (SO~2) exposure: an overview of its effects on humans and laboratory animals. Annali-Istituto Superiore Di Sanita, 1994
- H Steinhart, S Schulz, R Mutters. Evaluation of ozonated oxygen in an experimental animal model of osteomyelitis as a further treatment option for skull-base osteomyelitis. Eur Arch Otorhinolaryngol, 1999. [DOI | PubMed]
- MK Aslan, Ö Boybeyi, MF Şenyücel, Ş Ayva, Ü Kısa, N Aksoy. Protective effect of intraperitoneal ozone application in experimental ovarian ischemia/reperfusion injury. J Pediatr Surg, 2012. [DOI | PubMed]
- R Giunta, A Coppola, C Luongo, A Sammartino, S Guastafierro, A Grassia. Ozonized autohemotransfusion improves hemorheological parameters and oxygen delivery to tissues in patients with peripheral occlusive arterial disease. Ann Hematol, 2001. [DOI | PubMed]
