Prevalence and Patterns of Skin Diseases among School Children in Egypt: A National Cross-sectional Study
Abstract
Background:
Pediatric dermatoses are common and impact the quality of life. This study aimed to estimate the prevalence and characteristics of pediatric dermatoses among 1ry and 2ry school students in Egypt.
Methods:
A Cross-sectional study was conducted on 23,203 1ry and 2ry school students of both sexes in eight governorates in Egypt between January 2023 and May 2023. A clinical examination for skin diseases was done, and a self-administered, author-designed questionnaire was given to children to complete with the assistance of their parents.
Results:
The average age of students was 12.00 ± 3.33 years. Prevalence of skin diseases was 37.9%. Positive family history of skin disease (aOR 3.482, 95% CI: 2.871–4.222, p < 0.001), sanitary water disposal (aOR 3.846, 95% CI: 3.001–4.930, p < 0.001), residence (aOR 1.760, 95% CI: 1.657–1.870, p < 0.001), father’s occupation and education (aOR 1.494, 95% CI: 1.334–1.672, p < 0.001 and aOR 1.349, 95% CI: 1.251–1.454, p < 0.001, respectively), and crowding index (aOR 1.469, 95% CI: 1.372–1.573, p < 0.001) were independent risk factors for exhibiting skin diseases.
Conclusion:
A high prevalence of pediatric dermatosis was established with associated sociodemographic risk factors, so healthcare and education programs and services should be directed toward children with continuous supervision and periodic examination.
Supplementary Information:
The online version contains supplementary material available at 10.1007/s44197-025-00440-8.
Article type: Research Article
Keywords: Acne, Dermatoses, Pityriasis Alba, Pityriasis Versicolor, Prevalence, Skin Diseases, Wart
Affiliations: https://ror.org/05sjrb944grid.411775.10000 0004 0621 4712Dermatology, Andrology and STDs Department, Faculty of Medicine, Menoufia University, Shebin El-Kom, Menoufia, Egypt; https://ror.org/05sjrb944grid.411775.10000 0004 0621 4712Public Health and Community Medicine Department, Faculty of Medicine, Menoufia University, Shebin El-Kom, Menoufia, Egypt; Faculty of Medicine, Menoufia National University, Tukh tanbisha, Menoufia, Egypt; Faculty of Applied Health Sciences Technology, Menoufia National University, Tukh Tanbisha, Menoufia, Egypt; https://ror.org/04f90ax67grid.415762.3Dermatology and Leprosy Clinic, Ministry of Health, Shebin El-Kom, Menoufia, Egypt; https://ror.org/04f90ax67grid.415762.3Benha Hospital for Mental Health, Ministry of Health, Qalyubia, Egypt; https://ror.org/04f90ax67grid.415762.3Birket Elsabe general hospital, Ministry of Health, Menoufia, Egypt; https://ror.org/04f90ax67grid.415762.3Elshorok Medical Area, Ministry of Health, Cairo, Egypt; https://ror.org/04f90ax67grid.415762.3Al Hadary Medical Center, Ministry of Health, Rod Al-Farag, Cairo, Egypt; https://ror.org/04f90ax67grid.415762.3Shoubra Public Hospital, Ministry of Health, Cairo, Egypt; https://ror.org/04f90ax67grid.415762.3Mansoura Health Insurance Organization, Ministry of Health, Mansoura, Egypt; https://ror.org/04f90ax67grid.415762.3Dermatology Department, May 15 Hospital, Ministry of Health, Cairo, Egypt
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s44197-025-00440-8 | PubMed: 40622428 | PMC: PMC12234915
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.2 MB)
Introduction
The prevalence of pediatric dermatoses varies among communities depending on multiple factors, such as genetic predisposition, social class, parents’ employment and education, hygienic practices, health consciousness, and standard of medical treatment. These variables explain the different records in various communities and provide each group with a distinct pattern of illness. Researching pediatric dermatosis prevalence aims to evaluate health literacy and service accessibility to develop child health care plans that address real community needs [ref. 1].
Skin diseases are a significant source of health problems, affecting a high proportion of the population and causing distress and disabilities. Although very common in many developing countries, skin diseases are not often significant health problems, even when serious illnesses may be heralded by skin alterations [ref. 2].
Children at school frequently have skin conditions. Nonetheless, there is relatively little epidemiologic research on the broader public. The following are some of the drawbacks of several earlier studies: The validity of the diagnosis is dubious in self-administered questionnaire studies or surveys performed by nondermatologists; a survey of the referred cases in hospital-based settings may not fully reflect the actual situation in the general community [ref. 3].
Even though they are not life-threatening, skin conditions like alopecia areata and acne can be very upsetting for students in school. Families impacted by some chronic refractory skin disorders, for example, vitiligo, psoriasis, and atopic dermatitis may have major concerns about the psychological effects, course, and consequences of the illness and the therapeutic choices about long-term safety [ref. 4].
The prevalence of skin disorders has been studied through several hospital and community-based investigations, with the sorts of problems varying according to geography, climate, and participant age. Nonetheless, there is a few population-based research on prevalence of skin diseases conducted in Egypt. However, population-based investigations are necessary to understand the disease implications and direct suitable medical care toward them. In contrast, data from hospital-based research is restricted to individuals who see the hospital’s doctors with serious skin conditions. However, the kind and severity of skin disorders and illnesses varied significantly across research conducted in communities and schools [ref. 5].
This investigation aimed to determine the most common skin disorders among primary and secondary school students in Egypt, evaluate the sociodemographic determinants of their development, and raise awareness of the risk factors correlated with these conditions.
Methods
Study Design and Participants
A cross-sectional analytical investigation was carried out from January 2023 to May 2023, using a multistage random sampling procedure. Students aged 5 to 18 were recruited from 58 schools and selected randomly as part of the school health screening study. There was a balanced representation of all socioeconomic groups, considering the distribution of people who reside in rural and urban areas. The investigation’s sample was selected using data from the Egypt Demographic Health Survey conducted in 2015 [ref. 6].
Sampling
Egypt is a nation comprised of 27 governorates, making it an impressive country. The investigation was conducted using a multistage random sampling technique. Initially, a selection was made among the 27 governorates, with two major governorates from Upper Egypt and six central governorates from Lower Egypt selected. Subsequently, a selection was made of 20 districts and cities, picked randomly from the previously selected governorates. The study conducted a full investigation of both private and public elementary and secondary schools within the districts and cities. A total of 58 facilities were randomly selected from the eight governorates included in the study (Fig. 1). The investigation was performed three days a week. The days with the highest population density were selected to expedite and gather a large amount of data. During both educational courses and break time, students were randomly examined in a school selected from a list of schools. We met the participants in the morning and in private rooms immediately after starting the education day. Author-designed questionnaire was given to children to be completed with the assistance of their parents for sociodemographic data (Supplementary 1).
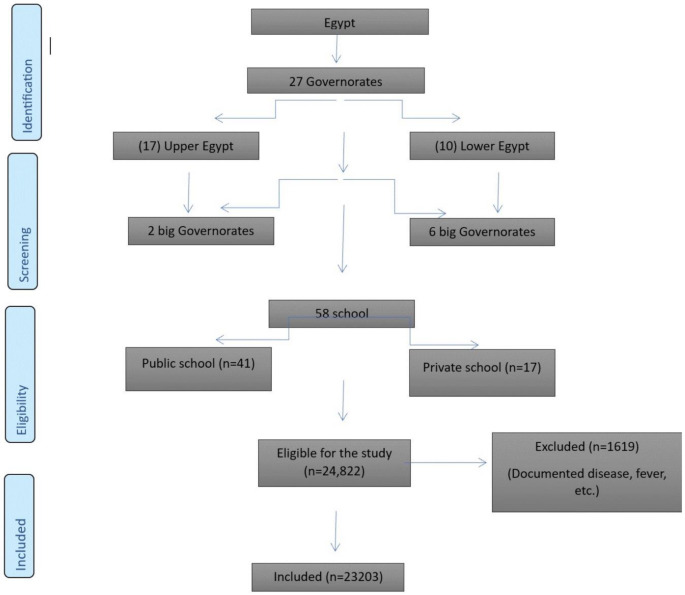
Between January 2023 and May 2023; 24,822 Egyptian students aged 5 to 18 were researched. To begin, we picked 23,423 adolescents and children who were eligible for the research, followed by 23,203 adolescents and children as a final total sample, excluding 220 children.
Inclusion and Exclusion Criteria
Participants eligible for inclusion in the study were children aged between 6 and 18 years who were enrolled in a formal educational institution and attended classes at least three days per week. Only those whose parents or legal guardians provided written informed consent were included. Additionally, participants were required to be able to complete the survey and undergo the physical examination without experiencing undue psychological distress or physical discomfort. Children were excluded from the study if their parents or legal guardians declined to provide informed consent.
Data Collection
All cases were exposed to the following.
- Full history taking, which included: (i) personal history: name, sex, age, and residence, school type, stage of education, frequency of bathing per week, family size, parents’ occupation, and crowding index; (ii) present history: course, onset, relation to diet, period of dermatoses, relation to sun exposure, relation to stress, previous treatment, and history of other skin illnesses; (iii) family history of any skin illnesses.
- Clinical examination, which included general examinations, bodyweight and height measurements, and a detailed dermatological examination of the entire body, including hair, nails, and mucous membranes. A clinical evaluation of skin illness was done to identify the distribution, clinical variants, and extent of the lesions. Wood’s light and dermoscopic examinations were done in case of doubtful diagnosis.
Statistical Analysis
SPSS (Statistical Package for the Social Sciences) version 26 was used to tabulate and analyze the data collected on an IBM-compatible computer. Quantitative normally distributed data were represented as mean and SD and range, while qualitative information was expressed as number and percentage (No. and %). The relationship between two qualitative variables was investigated using the Pearson Chi-squared test (χ2) while the relationship between two quantitative normally distributed variables was examined using the student’s t-test (t). Kolmogorov-Smirnov Test was used to test the normality of quantitative data. To identify the predictors of skin illnesses among the participants in the investigation, logistic regression was used. Statistical significance was established at a P-value of less than 0.05.
Results
This research involved 23,203 students aged 6 to 18 years with a mean of 12.00 ± 3.33, 11,237 males (48.4%) and 11,966 females (51.6%). A total of 72.2% were at public school, 79.5% were at primary stage, and 61.6% of the students were of urban residence. A total of 78% of students had a bath ≤ 2 per week, and 72.2% had hair wash ≤ 2 per week. The family history of skin diseases was positive in 2.4%. Regarding the students’ mothers, 51.7% of them were not working, and 51.7% got a university education (Table 1).
Table 1: Sociodemographic and lifestyle characteristics of studied participants (n = 23,203)
| Variable | Mean ±SD | Range |
|---|---|---|
| Age (years) | 12.00 ± 3.33 | 18-Jun |
| Family size | 4.97 ± 1.11 | 11-Feb |
| House rooms | 2.82 ± 0.68 | 6-Jan |
| Crowding index | 1.84 ± 0.52 | 0.5-6 |
| Bathing per week | 1.83 ± 0.76 | 3-Jan |
| Hair wash per day | 1.96 ± 0.77 | 3-Jan |
| BMI (kg/m2) | 20.55 ± 3.85 | 9.72–39.84 |
| No. | % | |
| Sex | ||
| Male | 11237 | 48.4 |
| Female | 11966 | 51.6 |
| School type | ||
| Public | 16747 | 72.2 |
| Private | 6456 | 27.8 |
| Stage of education | ||
| Primary | 18451 | 79.5 |
| Secondary | 4752 | 20.5 |
| Residence | ||
| Urban | 14291 | 61.6 |
| Rural | 8912 | 38.4 |
| Frequency of bathing per week | ||
| ≤ 2 | 18104 | 78 |
| > 2 | 5099 | 22 |
| Frequency of hair wash per week | ||
| ≤ 2 | 16871 | 72.7 |
| > 2 | 6332 | 27.3 |
| Positive family history | 568 | 2.4 |
| Mother occupation | ||
| Working | 11209 | 48.3 |
| Not working | 11994 | 51.7 |
| Mother’s education | ||
| Illiterate | 1406 | 6.1 |
| Basic | 2118 | 9.1 |
| Secondary | 7024 | 30.3 |
| University | 11997 | 51.7 |
| Postgraduate | 658 | 2.8 |
| Father occupation | ||
| Working | 21026 | 90.6 |
| Not working | 2177 | 9.4 |
| Father’s education | ||
| Illiterate | 997 | 4.3 |
| Basic | 1968 | 8.5 |
| Secondary | 5116 | 22 |
| University | 14140 | 60.9 |
| Postgraduate | 982 | 4.2 |
| Water disposal | 20311 | 87.5 |
| Waste disposal | 20909 | 90.1 |
SD: standard deviation; BMI: body mass index
Skin diseases were observed in 8787 students (37.9%). A statistically significant difference was noticed between those with and without skin disease as skin diseases, which were higher in students with a mean age of 12.34 ± 3.40 years; those in public schools (74.1%); those in primary stage (77.2%) (Fig. 2); those who live in urban residence (53.8%); those having less frequency of bathing per week (76.3%); those with a positive family history of skin diseases (4.7%); those not having working mothers (57.5%); those having working fathers (93.3%); those having a mother (46.3%) and a father (55.2%) with university education; those with a family size of mean of 5.01 ± 1.18; those with fewer house rooms; and those with more crowding index and BMI (Table 2).
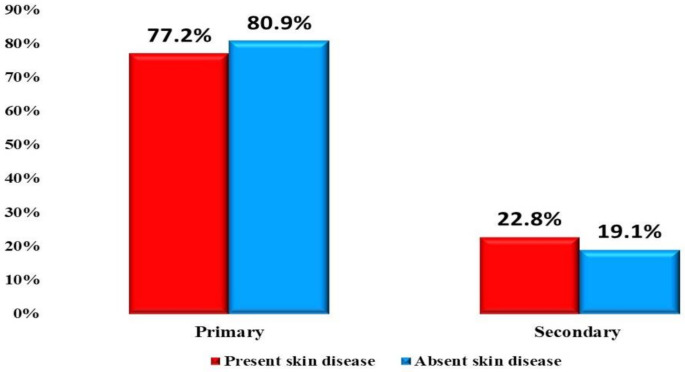
Table 2: Distribution of skin disease regarding sociodemographic data (n = 23,203)
| Characteristic | Skin disease occurrence | Test of sign | ||||
|---|---|---|---|---|---|---|
| Present (n = 8,787) | Absent (n = 14,416) | |||||
| No. | % | No. | % | χ2 | p-value | |
| Sex | 1.52 | 0.218 | ||||
| Male | 4301 | 48.9 | 6936 | 48.1 | ||
| Female | 4486 | 51.1 | 7480 | 51.9 | ||
| School type | 26.33 | < 0.001* | ||||
| Public | 6512 | 74.1 | 10235 | 71 | ||
| Private | 2275 | 25.9 | 4181 | 29 | ||
| Stage of education | 47.56 | < 0.001* | ||||
| Primary | 6782 | 77.2 | 11669 | 80.9 | ||
| Secondary | 2005 | 22.8 | 2747 | 19.1 | ||
| Residence | 362.29 | < 0.001* | ||||
| Urban | 4728 | 53.8 | 9563 | 66.3 | ||
| Rural | 4059 | 46.2 | 4853 | 33.7 | ||
| Frequency of bathing/week | 23.4 | < 0.001* | ||||
| ≤ 2 | 6708 | 76.3 | 11396 | 79.1 | ||
| > 2 | 2079 | 23.7 | 3020 | 20.9 | ||
| Frequency of hair wash/week | 2.99 | 0.084 | ||||
| ≤ 2 | 6446 | 73.4 | 10425 | 72.3 | ||
| > 2 | 2341 | 26.6 | 3991 | 27.7 | ||
| Positive family history | 415 | 4.7 | 153 | 1.1 | 306.5 | < 0.001* |
| Mother occupation | 189.19 | < 0.001* | ||||
| Working | 3737 | 42.5 | 7472 | 51.8 | ||
| Not working | 5050 | 57.5 | 6944 | 48.2 | ||
| Mother’s education | 329.72 | < 0.001* | ||||
| Illiterate | 740 | 8.4 | 666 | 4.6 | ||
| Basic | 942 | 10.7 | 1176 | 8.2 | ||
| Secondary | 2674 | 30.4 | 4350 | 30.2 | ||
| University | 4070 | 46.3 | 7927 | 55 | ||
| Postgraduate | 361 | 4.1 | 297 | 2.1 | ||
| Father occupation | 116.39 | < 0.001* | ||||
| Working | 8195 | 93.3 | 12831 | 89 | ||
| Not working | 592 | 6.7 | 1585 | 11 | ||
| Father’s education | 466.4 | < 0.001* | ||||
| Illiterate | 620 | 7.1 | 377 | 2.6 | ||
| Basic | 805 | 9.2 | 1163 | 8.1 | ||
| Secondary | 1965 | 22.4 | 3151 | 21.9 | ||
| University | 4851 | 55.2 | 9289 | 64.4 | ||
| Postgraduate | 546 | 6.2 | 436 | 3 | ||
| Water disposal, yes | 8264 | 94 | 12047 | 83.6 | 549.69 | < 0.001* |
| Waste disposal, yes | 8342 | 94.9 | 12567 | 87.2 | 369.16 | < 0.001* |
| Mean ± SD | Mean ± SD | Test of sig. | p-value | |||
| Age (years) | 12.34 ± 3.40 | 11.80 ± 3.26 | t = 12.19 | < 0.001* | ||
| Family size | 5.01 ± 1.18 | 4.95 ± 1.06 | t = 4.12 | < 0.001* | ||
| House rooms | 2.70 ± 0.67 | 2.89 ± 0.67 | t = 21.30 | < 0.001* | ||
| Crowding index | 1.95 ± 0.58 | 1.78 ± 0.47 | t = 23.79 | < 0.001* | ||
| BMI (kg/m2) | 20.87 ± 4.15 | 20.36 ± 3.64 | t = 10.09 | < 0.001* | ||
*: statistically significant, χ2: chi-squared test, t: Student’s t-test, SD: standard deviation, BMI: body mass index
The most frequent skin illnesses were acne vulgaris, with a percentage of 5.6%, followed by pityriasis alba (4.3%), wart (4.2%), pityriasis versicolor (3.7%), and papular urticaria (3.5%) (Table 3).
Table 3: Distribution of skin diseases among the studied participants (n = 23,203)
| Skin disease | No. | % | |
|---|---|---|---|
| All | 8787 | 37.9 | |
| Infectious dermatosis | 4299 | 18.5 | |
| Viral | Wart | 976 | 4.2 |
| Herpes simplex | 652 | 2.8 | |
| Chicken pox | 84 | 0.4 | |
| Molluscum contagiosum | 18 | 0.1 | |
| Fungal | Pityriasis versicolor | 867 | 3.7 |
| White piedra | 82 | 0.4 | |
| Parasitic | Pediculosis | 490 | 2.1 |
| Scabies | 470 | 2 | |
| Bacterial | Folliculitis | 365 | 1.6 |
| Impetigo | 295 | 1.3 | |
| Non infectious | 8870 | ||
| Pilosebaceous | Acne | 1306 | 5.6 |
| Alopecia areata | 660 | 2.8 | |
| Traction alopecia | 423 | 1.8 | |
| Dandruff | 241 | 1 | |
| Telogen effluvium | 116 | 0.5 | |
| Dermatitis | Pityriasis alba | 1008 | 4.3 |
| Papular urticaria | 804 | 3.5 | |
| Seborrheic dermatitis | 647 | 2.8 | |
| Eczema | 645 | 2.8 | |
| Angular stomatitis | 633 | 2.7 | |
| Atopic dermatitis | 633 | 2.7 | |
| Contact dermatitis | 265 | 1.1 | |
| Berloque dermatitis | 52 | 0.2 | |
| Papulosquamous | Psoriasis | 171 | 0.7 |
| Lichen planus | 128 | 0.6 | |
| Pigmentary disorders | Vitiligo | 209 | 0.9 |
| Nevi | 133 | 0.6 | |
| Freckles | 102 | 0.4 | |
| Genodermatosis | Neurofibromatosis and others | 113 | 0.5 |
| Miscellaneous | Hemangioma | 148 | 0.6 |
| Acanthosis nigricans | 137 | 0.6 | |
| Nail dystrophy | 108 | 0.5 | |
| Milia | 106 | 0.5 | |
| Chilblain | 34 | 0.1 | |
| Lipoma | 25 | 0.1 | |
| Keloid | 23 | 0.1 | |
Multiplicities of dermatoses per child should be taken into consideration to avoid false perceptions of some discrepancies in frequencies and percentages
The prevalence of acne vulgaris was statistically significantly greater in women than that in men. In comparison, the prevalence of pityriasis alba, wart, and tinea versicolor was statistically significantly greater in men than that in women (p-value < 0.05) (Table 4).
Table 4: The most common skin disease in relation to sex among studied participants (n = 23,203)
| Skin disease | Sex | |||
|---|---|---|---|---|
| Male (n = 11,237) | Female (n = 11,966) | |||
| No. | % | No. | % | |
| Acne vulgaris | 529 | 4.7 | 777 | 6.5 |
| Pityriasis alba | 659 | 5.9 | 349 | 2.9 |
| Wart | 536 | 4.8 | 440 | 3.7 |
| Tinea versicolor | 456 | 4.1 | 411 | 3.4 |
| Papular urticaria | 385 | 3.4 | 419 | 3.5 |
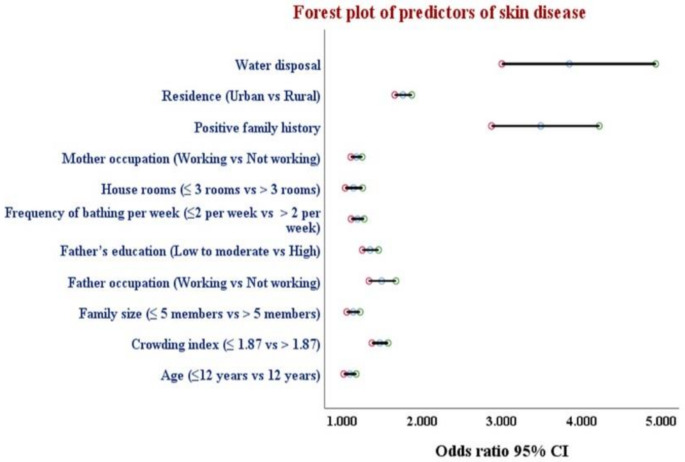
Binary logistic regression analysis was performed to confirm the effects of school type, educational stage, residence, frequency of bathing per week, positive family history of skin disease, mother occupation and education, father occupation and education, water and waste disposal, age, family size, number of house rooms, and crowding index and BMI on the likelihood that the student will exhibit a skin disease. As per univariable analysis, all predictor variables were statistically significant. Accordingly, all statistically significant predictor variables on univariable analysis were entered into a regression model. Residence (aOR 1.760, 95% CI: 1.657–1.870, p < 0.001), frequency of bathing per week (aOR 1.190, 95% CI: 1.111–1.274, p < 0.001), positive family history of skin disease (aOR 3.482, 95% CI: 2.871–4.222, p < 0.001), mother’s occupation (aOR 1.176, 95% CI: 1.107–1.249, p < 0.001), father’s occupation and education (aOR 1.494, 95% CI: 1.334–1.672, p < 0.001 and aOR 1.349, 95% CI: 1.251–1.454, p < 0.001, respectively), water disposal (aOR 3.846, 95% CI: 3.001–4.930, p < 0.001), age (aOR 1.093, 95% CI: 1.018–1.174, p = 0.014), family size (aOR 1.136, 95% CI: 1.056–1.223, p = 0.001), number of house rooms (aOR 1.140, 95% CI: 1.037–1.254, p = 0.007), and crowding index (aOR 1.469, 95% CI: 1.372–1.573, p < 0.001) were independent risk factors for exhibiting skin diseases (Table 5; Fig. 3).
Table 5: Predictors of the likelihood of occurrence of skin disease
| Predictor | Univariable | Multivariable | ||||
|---|---|---|---|---|---|---|
| cOR | 95% CI | p-value | aOR | 95% CI | p-value | |
| School type (private) | 1.169 | 1.101–1.241 | < 0.001* | 1.001 | 0.937–1.069 | 0.983 |
| Stage of education (primary) | 1.256 | 1.177–1.340 | < 0.001* | 1.037 | 0.957–1.124 | 0.378 |
| Residence (urban) | 1.692 | 1.602–1.786 | < 0.001* | 1.760 | 1.657–1.870 | < 0.001* |
| Frequency of bathing/week (≤ 2) | 1.170 | 1.098–1.246 | < 0.001* | 1.190 | 1.111–1.274 | < 0.001* |
| Positive family history (no) | 4.621 | 3.832–5.573 | < 0.001* | 3.482 | 2.871–4.222 | < 0.001* |
| Mother occupation (working) | 1.454 | 1.378–1.534 | < 0.001* | 1.176 | 1.107–1.249 | < 0.001* |
| Mother’s education (high) | 1.306 | 1.238–1.377 | < 0.001* | 1.002 | 0.932–1.077 | 0.959 |
| Father occupation (not working) | 1.710 | 1.550–1.887 | < 0.001* | 1.494 | 1.334–1.672 | < 0.001* |
| Father’s education (high) | 1.302 | 1.232–1.376 | < 0.001* | 1.349 | 1.251–1.454 | < 0.001* |
| Water disposal (no) | 3.107 | 2.815–3.430 | < 0.001* | 3.846 | 3.001–4.930 | < 0.001* |
| Waste disposal, yes | 2.758 | 2.478–3.070 | < 0.001* | 0.827 | 0.629–1.086 | 0.172 |
| Age (≤ 12 years) | 1.316 | 1.248–1.388 | < 0.001* | 1.093 | 1.018–1.174 | 0.014* |
| Family size (≤ 5 members) | 1.378 | 1.298–1.462 | < 0.001* | 1.136 | 1.056–1.223 | 0.001* |
| House rooms (> 3 rooms) | 1.646 | 1.520–1.783 | < 0.001* | 1.140 | 1.037–1.254 | 0.007* |
| Crowding index (≤ 1.87) | 1.903 | 1.804–2.008 | < 0.001* | 1.469 | 1.372–1.573 | < 0.001* |
| BMI (kg/m2) (≤ 19.82) | 1.216 | 1.153–1.283 | < 0.001* | 1.048 | 0.983–1.117 | 0.150 |
cOR = crude odds ratio. aOR = adjusted odds ratio. CI = confidence interval. () = reference category
Discussion
Pediatric dermatoses are common among school children, especially in developing countries. It can cause psychological distress, loss of self-confidence, and school abstinence, especially with affection of apparent parts of the skin [ref. 7].
In Egypt, few school-based investigations have been done by dermatologists to identify the prevalence of skin diseases, and all investigations were conducted in small numbers and in only one governorate [ref. 7–ref. 9]. The current study was conducted on a relatively larger number of children in eight governorates.
This study involved students aged 6 to 18 years, including 11,237 male and 11,966 female students. A skin condition was detected in 8,787 students, accounting for 37.9% of the total population. This finding aligns with earlier research conducted in African countries, where the prevalence of skin diseases ranged from 39.6 to 40.2% [ref. 10, ref. 11]. However, subsequent investigations demonstrated a greater incidence of skin problems, precisely 76% and 60%, respectively [ref. 7, ref. 12]. Other studies revealed a lower prevalence of skin diseases, 23.8% and 26.7% [ref. 8, ref. 13]. These variabilities may be due to different climatic, social, and hygienic factors.
In this study, the prevalence of skin illnesses was significantly higher in primary school students, as in the study conducted in Turkey [ref. 14]. This may be related to the large number of children included in the research who were at the primary stage, as primary education in Egypt is obligatory and includes a larger number of children than the secondary stage, which is more representative.
In a previous study in Iraq, 53.8% of students had skin diseases in urban areas similar to this study [ref. 15]. However, other studies demonstrated a higher prevalence in rural areas due to lower socioeconomic standards and poor sanitation [ref. 7, ref. 9].
In this study, we demonstrated a statistically significant variation between those with and without skin disease regarding the type of school, as it was higher in public schools. In line with this, Ewurum et al. discovered a greater prevalence of skin illnesses documented among children in public schools compared to private schools [ref. 11]. Moreover, Mohammed and colleagues in Sudan documented a prevalence of 45.5 and 25% for public and private schools, respectively [ref. 16]. Similarly, Laczynski and Cestari in Brazil documented prevalence figures of 93% and 83%, respectively, for public and private schools [ref. 17]. In this study, most public-school pupils were of low socioeconomic class, which may explain why skin diseases were more common among them. Risk factors for skin illnesses, such as poor hygiene and overcrowding, have been associated with low socioeconomic status.
In this study, noninfectious dermatoses were 38.2%, like reports in Iraq 33.7% [ref. 18] and Tanzania 35.4% [ref. 19], and infectious dermatoses were 18.9%, like the report in Jaipur 19.8% [ref. 12]. Infectious dermatoses may be underestimated in cross-sectional studies due to seasonal variation in disease occurrence, home isolation of diagnosed cases, and home care in febrile conditions. Moreover, the lack of seeking medical advice for most noninfectious dermatoses, as lesions are asymptomatic or due to ignorance, led to an increase in their prevalence.
The most common skin illnesses observed in this research were acne vulgaris, with a percentage of 5.6%, followed by pityriasis alba 4.3%, wart 4.2%, pityriasis versicolor 3.7%, and papular urticaria 3.5%.
The most common skin disease recorded in this study was acne at 5.6%, which was significantly higher in females; these discoveries were in line with research conducted in Turkey that revealed an acne prevalence of 6% and more in females [ref. 14]. Furthermore, another study in Hong Kong showed that acne was the most frequent skin illness among school kids 9.8% [ref. 20]. This could be explained by including students in the adolescent period up to 18 years. In other investigations, the prevalence figures were 2.7% in Brazil and Ethiopia [ref. 17, ref. 21]. This distinction can be explained by the racial variances, genetic susceptibility, hormonal profile, and dietary habits that play a significant role in acne vulgaris occurrence.
Acne was present in students’ age ranging from 15 to 18 and represents 22% of students of the same age. Another study in secondary school in Egypt revealed a prevalence of clinically confirmed acne of 24.4%, with greater rates among women that may be due to hormonal changes throughout menstruation or greater levels of stress among women [ref. 22]. Acne is a multifaceted illness, and the study only assessed acne cases for three months during the winter (January to May). Notably, the production of skin surface lipids declines in the winter, and there is a negative association between lipids and acne incidence. Patients with acne were found to have psychological, social, and emotional problems that were as marked as those described by cases with chronic disabling asthma, diabetes, epilepsy, arthritis, and back pain. Similar community studies in secondary school children reported a higher level of psychiatric problems [ref. 23, ref. 24].
The second most frequent illness was pityriasis alba, with a prevalence of 4.3%, which was significantly higher in males. In a previous study in Ismailia City, Egypt, 10.3% of primary school students had pityriasis alba [ref. 25]. This may be due to skin dryness due to sun exposure, going to school on foot, and being more prevalent in males as they are more exposed to sunlight during outdoor play and helping parents at work.
The prevalence of warts in this research was 4.2%, similar to the prevalence of 4.6% reported by Mengist Dessie et al. in Northern Ethiopia [ref. 26]. This may be due to sharing shoes and clothes and barefoot activities. Lower prevalence of 3.3% and 2.6% were documented in Saudi Arabia [ref. 27] and Turkey, respectively [ref. 14].
The prevalence of pityriasis versicolor in this research was 3.7%, comparable with that reported by Mengist Dessie et al. [ref. 26] in Northern Ethiopia at 6.3% and by Fung and Lo [ref. 20] in Hong Kong at 4.5%. Pityriasis versicolor may be explained by the inclusion of older students up to 18 years of age in this study. The stimulation of sex hormones, such as androgen stimulation, that occurs in adolescence results in greater development of the sebaceous gland with more secretion of sebum and skin lipids, which favors the growth of Malassezia, the etiologic agent of pityriasis versicolor. Moreover, the moist and humid environment is commonly encountered throughout physical activity by the adolescent group [ref. 28].
Noninfective skin lesions, such as papular urticaria, were in the fifth rank, with a prevalence of 3.5%, similar to that reported in Jaipur [ref. 12]. Papular urticaria results from an exaggerated response to insect bites and stings. It may be worsened by poor environmental sanitation and inadequate drainage prevalent in developing countries like Egypt [ref. 29].
In this study, skin diseases were significantly associated with risk factors, such as frequency of bathing per week, family size, house rooms, and crowding index like the study by Alkalash et al. [ref. 7]. Additionally, Ewurum et al. discovered that using a multivariate logistic regression and identified the number of individuals per room, the frequency of bathing per week, and the source of water as important risk variables for skin illnesses [ref. 11]. The study conducted by Dessie et al. found a strong association between skin illnesses and factors such as history of skin illnesses and personal hygiene [ref. 26].
Skin diseases were significantly associated with parents’ occupations. In line with our study findings, in Ethiopia, Lulu et al. [ref. 21] and Alkalash et al. [ref. 7] also discovered a significant correlation between skin illness and parents’ occupations.
Study Strengths and Limitations
This research was performed on a large sample size covering the entire country. However, this study had a one-point inspection of the skin for illnesses, which may have overlooked the students with infectious skin disorders for a short period, underestimating the real burden of skin illnesses in these kids.
Conclusion
Skin disorders were prevalent among schoolchildren. Living conditions and sociodemographic factors are significant risk factors for skin illnesses. The high prevalence of skin illnesses in our environment can be reduced by implementing standard hygienic practices by school students and their parents/caregivers. Consequently, we advocated for implementing a preventive health education program that would educate school students at various levels, as well as their families and teachers, regarding skin diseases.
Supplementary Materials
References
- M Balai, AK Khare, LK Gupta, A Mittal, CM Kuldeep. Pattern of pediatric dermatoses in a tertiary care centre of South West Rajasthan. Indian J Dermatol, 2012. [DOI | PubMed]
- H Cortés, M Rojas-Márquez, ML Del Prado‐Audelo, OD Reyes‐Hernández, M González‐Del Carmen, G Leyva‐Gómez. Alterations in mental health and quality of life in patients with skin disorders: a narrative review. Int J Dermatol, 2022. [DOI | PubMed]
- 3.Paller AS, Mancini AJ. Paller and Mancini-Hurwitz clinical pediatric dermatology: A textbook of skin disorders of childhood and adolescence. 6th ed. Elsevier Health Sciences; 2020.
- C Flohr, R Hay. Putting the burden of skin diseases on the global map. Br J Dermatol, 2021. [DOI | PubMed]
- M Davila, LJ Christenson, RD Sontheimer. Epidemiology and outcomes of dermatology in-patient consultations in a Midwestern US university hospital. Dermatol Online J, 2010. [DOI | PubMed]
- 6.El-Zanaty F, Way A. Egypt health issue survey. Ministry of Health and Population, Cairo, Egypt. 2015. https://dhsprogram.com/pubs/pdf/FR313/FR313.pdf
- SH Alkalash, MA Gaber, AA Kamal. Prevalence of skin diseases among primary school children in Benha city, Kalubia governorate, Egypt. Menoufia Med J, 2023. [DOI]
- EA El-Khateeb, RA Lotfi, KM Abdel‐Aziz, SE El‐Shiekh. Prevalences of skin diseases among primary schoolchildren in damietta, Egypt. Int J Dermatol, 2014. [DOI | PubMed]
- RE El-Dawela, AN Fatehy, AA Abd Elmoneim. Prevalence of skin diseases among school children: a survey in the Sohag Governorate. J Egypt Women Dermatol Soc, 2012. [DOI]
- OE Amoran, OO Runsewe-Abiodun, AO Mautin, IO Amoran. Determinants of dermatological disorders among school children in sagamu, Nigeria. Educ Res, 2011
- O Ewurum, CA Ibeneme, TO Nnaji, AN Ikefuna. Spectrum of skin disorders among primary school children in umuahia, South-East Nigeria. Niger J Clin Pract, 2022. [DOI | PubMed]
- M Nijhawan, M Bagri, S Nijhawan, S Bishnoi, S Agarwal, S Nijhawan. Pattern of common skin diseases among school going children in Semi-Urban area of jaipur: A cross-sectional study. Indian J Paediatr Dermatol, 2020. [DOI]
- A Hogewoning, A Amoah, JN Bavinck, D Boakye, M Yazdanbakhsh, A Adegnika. Skin diseases among schoolchildren in ghana, gabon, and Rwanda. Int J Dermatol, 2013. [DOI | PubMed]
- A Uludağ, SO Kılıc, S Isık, YH Ertekin, M Tekin, S Cevizci. Prevalence of skin disorders in primary and secondary school age children in canakkale, turkey: a community-based survey. Adv Dermatology Allergology/Postępy Dermatol Alergol, 2016. [DOI]
- BA Algharbawi, SJ Baraznchi, JK Al-Mnehil. Prevalence and pattern of skin disorder among primaryschool children in Wasit governorate, iraq, 2021. Tex J Med Sci, 2023. [DOI]
- SA Mohammed, MM Elhassan, K Hussein. The pattern of paediatric dermatoses among primary school children in Khartoum north, 2007. Sudan J Public Health, 2010
- CM Laczynski, SD Cestari. Prevalence of dermatosis in scholars in the region of ABC Paulista. Bras Dermatol, 2011. [DOI]
- KA Khalifa, TS Al-Hadithi, FH Al-Lami, JK Al-Diwan. Prevalence of skin disorders among primary-school children in Baghdad governorate, Iraq. East Mediterr Health J, 2010. [DOI | PubMed]
- M Amiri, FF Furia, M Bakari. Skin disorders among children living in orphanage centres in Dar Es salaam, Tanzania. Trop Med Health, 2020. [DOI | PubMed]
- WK Fung, KK Lo. Prevalence of skin disease among school children and adolescents in a student health service center in Hong Kong. Pediatr Dermatol, 2000. [DOI | PubMed]
- Y Lulu, G Tolesa, J Cris. Prevalence and associated factors of skin diseases among primary school children in illuababorzone, oromia regional state, South West Ethiopia. Indo Am J Pharm Res, 2017. [DOI]
- K Tayel, M Attia, N Agamia, N Fadl. Acne vulgaris: prevalence, severity, and impact on quality of life and self-esteem among Egyptian adolescents. J Egypt Public Health Assoc, 2020. [DOI | PubMed]
- M Kilkenny, V Stathakis, ME Hibbert, G Patton, J Caust, G Bowes. Acne in Victorian adolescents: associations with age, gender, puberty and psychiatric symptoms. J Paediatr Child Health, 1997. [DOI | PubMed]
- A Smithard, C Glazebrook, HC Williams. Acne prevalence, knowledge about acne and psychological morbidity in mid-adolescence: a community‐based study. Br J Dermatol, 2001. [DOI | PubMed]
- WS Elshafey, LA Fiala, RW Mohamed, NA Ismael. The distribution and determinants of pityriasis Alba among elementary school students in Ismailia City. J Am Sci, 2012
- A Mengist Dessie, S Fenta Feleke, S Getaye Workie, T Getinet Abebe, Y Mossu Chanie, A Kassa Yalew. Prevalence of skin disease and its associated factors among primary schoolchildren: A Cross-Sectional study from a Northern Ethiopian town. Clin Cosmet Investig Dermatol, 2022. [DOI]
- TT Amin, A Ali, F Kaliyadan. Skin disorders among male primary school children in al hassa, Saudi arabia: prevalence and socio-demographic correlates-a comparison of urban and rural populations. Rural Remote Health, 2011. [DOI]
- SM He, WD Du, S Yang, SM Zhou, W Li, J Wang. The genetic epidemiology of Tinea versicolor in China. Mycoses, 2008. [DOI | PubMed]
- K Bahamdan, A Mahfouz, T Tallab, IA Badawi, OM Al-Amari. Skin diseases among adolescent male boys in abha, Saudi Arabia. Int J Dermatol, 1996. [DOI | PubMed]
