Adverse events associated with the use of cannabis-based products in people living with cancer: a systematic scoping review
Abstract
Purpose:
To summarise the extent and type of evidence in relation to adverse events (AEs) associated with the use of cannabis-based products (CBP) in people living with cancer.
Methods:
The Joanna Briggs Institute (JBI) methodology for scoping reviews was applied. A search was performed in MEDLINE (Ovid), Embase (Ovid), CINAHL (EBSCOhost), Scopus, Web of Science Core Collections and AMED (Ovid) from their inception to 7 May 2023. Primary studies reporting AEs associated with any form of natural or synthetic CBP use in any cancer care setting and location were included.
Results:
One hundred fifty-two studies were included, with the most prevalent being randomised controlled trials (RCTs) (n = 61), followed by non-randomised controlled trials (n = 26) and case reports (n = 23). CBP was mainly used in gastrointestinal, liver, or peritoneal cancer (n = 98) and haematological or lymphoid cancer (n = 92), primarily to manage nausea and vomiting (n = 78) and cancer pain (n = 37). The most common CBP ingredients were combinations of THC and CBD (n = 69), synthetic THC (n = 47), single compounds of THC (n = 42) and CBD (n = 16) with diverse forms, administration routes and doses. The primary methods of administration were oral (n = 94) and inhalation (n = 54). A broad range of AEs were reported; the most common were related to the nervous system (n = 118), psychiatric (n = 101) and gastrointestinal system (n = 81). Diverse patient characteristics, significant under-reporting and low-quality reporting were observed in many studies.
Conclusions:
More rigorous research designs that prioritise comprehensive, standardised reporting of AEs and CBP use are required to fully elucidate the safety profile of CBP use in cancer care.
Supplementary Information:
The online version contains supplementary material available at 10.1007/s00520-024-09087-w.
Article type: Review Article
Keywords: Adverse events, Cannabis, Randomised controlled trial
Affiliations: https://ror.org/0384j8v12grid.1013.30000 0004 1936 834XSchool of Pharmacy, Faculty of Medicine and Health, University of Sydney, Sydney, Australia; Health Research Group, Sydney, Australia
License: © The Author(s) 2024 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s00520-024-09087-w | PubMed: 39694905 | PMC: PMC11655613
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (666 KB)
Introduction
Cannabis-based products (CBP) are used by people living with cancer to manage cancer symptoms and side effects of conventional therapy, improve quality of life and promote general health [ref. 1]. The prevalence of CBP use in this population is estimated to be 20–48%, with increased use reported in the last 10 years [ref. 2–ref. 11].
Cannabis is a flowering plant belonging to the Cannabaceae family. Cannabis sativa, Cannabis indica, Cannabis ruderalis and hybrids of these species are the most commonly used for medicinal purposes. The plant contains a broad range of chemical compounds including cannabinoids, terpenes and flavonoids, which are attributed to therapeutic benefits and side effects [ref. 12].
Key constituents in CBP, namely cannabinoids, mimic the effects of endogenous cannabinoids by binding to CB1 and CB2 receptors distributed throughout the body and activating the cannabinoid system. CB1 is found predominantly in the central nervous system and CB2 in immune cells. Two notable cannabinoids found in cannabis are delta 9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD). Δ9-THC is the main psychoactive component causing euphoria, relaxation, anxiety or hallucinations. CBD is non-psychoactive and studied for its anti-inflammatory, analgesic, anxiolytic and neuroprotective properties [ref. 1, ref. 12]. In addition, various synthetic analogues are available.
CBP-containing cannabinoids are available in various forms. The dried parts of the plant (leaves, flowers, buds), extracts (oil, tincture), edibles (food, beverages), topical applications (creams, gels, oil) and synthetic forms (capsules, tablets, solutions, suppositories) are administered by inhalation (by smoking or vaporising), oral ingestion, sublingual, topical or rectal application [ref. 13].
Access to CBP is influenced by the legal frameworks of a given country or jurisdiction. An increasing number of jurisdictions are now allowing legal access to medicinal cannabis for use in cancer care [ref. 14, ref. 15]. This reflects shifting societal attitudes and healthcare approaches and an emerging evidence-base to support its use in cancer care [ref. 16–ref. 19]. The process of such legalisation requires the establishment of regulatory frameworks that oversee the production, sale and consumption of CBP.
Clinical concerns about the safety of CBP use by people living with cancer are warranted as they are potentially a population at increased risk of adverse effects. Knowledge about adverse events (AEs) is crucial for making informed decisions and delivering safe, evidence-based care.
To date, no review has comprehensively reported on AEs associated with the use of any type of CBP in the cancer care context. Therefore, the aim of this review was to summarise the literature reporting AEs associated with CBP use by people living with cancer.
Methods
The Joanna Briggs Institute (JBI) methodology for scoping reviews [ref. 20] was employed for this study, and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) [ref. 21] and relevant items from the updated PRISMA 2020 statement [ref. 22] (S1). The review protocol was submitted for publication prior to study screening and selection [ref. 23].
Search strategy
The search strategy was developed for MEDLINE (Ovid) (S2) in consultation with an academic librarian and adapted for Embase (Ovid), CINAHL (EBSCOhost), Scopus, Web of Science Core Collections and AMED (Ovid). Literature published from inception to 7 May 2023, with no language restrictions, was searched using three concepts and related search terms: cancer diagnoses, adverse effects and CBP (S3). Due to the breadth of papers identified, no hand searches were conducted for additional articles.
Eligibility criteria
The JBI framework for scoping review eligibility criteria of Population, Concept and Context (PCC) was applied [ref. 20].
Population
Included were people of any age, gender or ethnicity, living with any cancer type and stage and comorbidities and using CBPs with or without concurrent treatments or recreational use of tobacco/alcohol/drugs. Mixed populations, including palliative care where only some participants have cancer, were only included when data was reported for the cancer population subgroup. Studies reporting or evaluating the risk of developing cancer associated with CBP use in other populations were excluded.
Concept
The concept of interest was AEs associated with any form of CBP use in cancer. Here, CBP refers to all forms of cannabis including natural and synthetic products and recreational and medicinal products (registered or unregistered).
We adopted The International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use definition of an AE as being “any untoward medical occurrence in a patient administered a medicinal product and which does not necessarily have to have a causal relationship with this treatment. An AE can therefore be any unfavourable and unintended sign (for example, an abnormal laboratory finding), symptom, or disease temporally associated with the use of a medicinal product, whether or not considered related to this medicinal product” and a serious AE as “any untoward medical occurrence that at any dose results in death, is life-threatening, requires inpatient hospitalisation or prolongation of existing hospitalisation, results in persistent or significant disability/incapacity, is a congenital anomaly/birth defect, or is a medically important event or reaction” [ref. 24].
Context
AEs that occurred in any setting such as home, primary care, secondary care or palliative care across any geographical location were included. There were no limitations on the individuals identifying or reporting the AE, nor on the reasons for using CBP within the cancer setting, including recreational use.
Types of studies
Primary data sources including randomised controlled trials, non-randomised controlled trials, observational studies, case–control studies, case series, case reports, cross-sectional surveys and retrospective chart reviews were included. Preclinical studies and secondary data sources such as systematic reviews, meta-analyses and clinical guidelines were excluded. Only articles published in peer review journals were included. Non-English articles; grey literature such as newspaper reports, white papers and university theses; and data from adverse events reporting systems or clinical trial databases were excluded. A post hoc decision was made to exclude conference meeting abstracts reporting the findings of potentially eligible studies or case studies with very low-quality non-specific AE reporting.
Source of evidence selection
All citations retrieved in the search were managed using Covidence [ref. 25] for automatic and manual identification of duplicate records, screening and selecting studies and data extraction.
A calibration exercise was conducted by two authors using the first 100 articles, to refine the full-text exclusion categories. Titles, abstracts and full-text articles were independently screened by two reviewers. Any disagreements that arose during the screening and selection processes were resolved through discussions with the other reviewers.
Data extraction and analysis
After piloting and refining the data extraction template in Covidence, data were recorded by a single reviewer and verified by another. The study design, country/region, participant characteristics, reason for CBP use, CBP intervention, use of a comparator, concomitant interventions, recreational tobacco/alcohol/drug use, AE characteristics and author’s conclusion or comments were recorded. Authors of the included studies were not contacted to request for missing or additional data. The data extraction template is provided in S4.
To optimise clarity and consistency in reporting across the included studies, each AE was then categorised according to the Common Terminology Criteria (CTCAE) System Organ Class (SOC) [ref. 26]. Overarching categories were also created to summarise the different ingredients and administration routes of the CBP. Due to high heterogeneity in the data, categories for dose, duration and other exposures were not created and not reported using descriptive statistics. One author re-coded this data and a second verified the coding.
During data extraction and coding, any disagreements were resolved through discussions with the other reviewers. As the aim was to map and characterise the available evidence, a critical appraisal and risk of bias assessments were not conducted, as per scoping review methodology [ref. 20]. Microsoft Excel [ref. 27] was used to generate charts and figures. Other findings were summarised and narrated.
Results
Study selection
An initial 14,134 records were identified across the databases searched. Following the removal of duplicate articles (n = 6570) and those not meeting the inclusion criteria for title and abstract (n = 7229), 335 full-text articles were subject to full-text review. Of these, an additional 183 studies were excluded. These are reported in S5 along with the reasons for exclusion. A total of 152 articles were included [ref. 28–ref. 179]. Figure 1 presents the study selection process by stages [ref. 22].
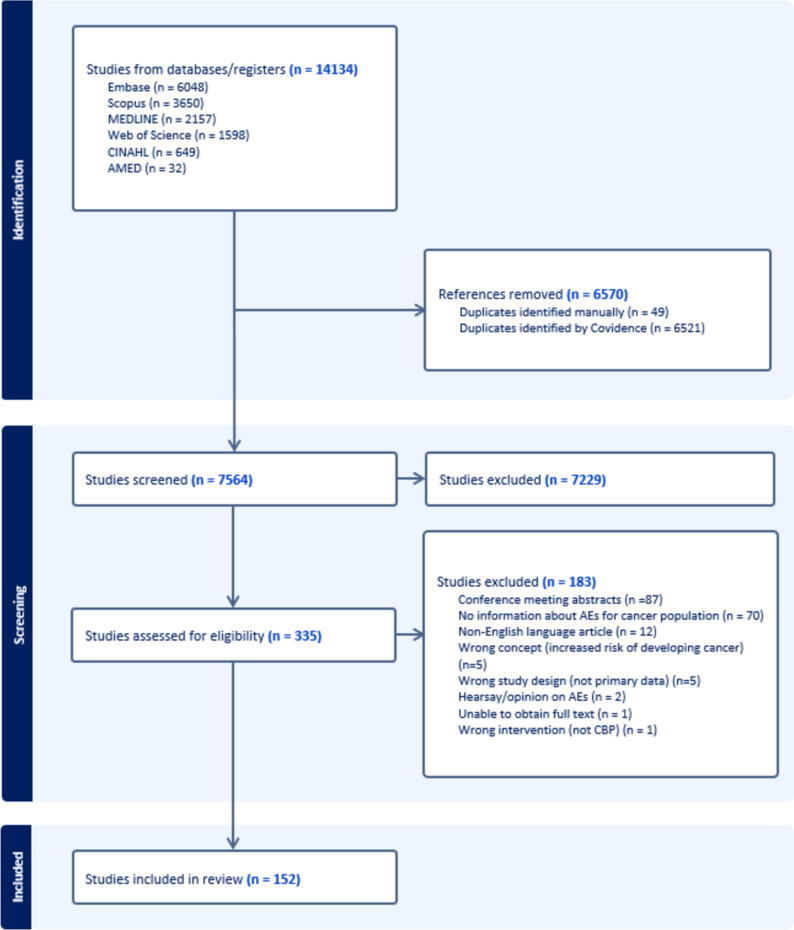
Study characteristics
Table 1 summarises key characteristics of the 152 included studies which were published between 1974 and 2023. Study designs included randomised controlled trials (RCTs) (n = 61), non-randomised controlled trials (n = 26), case reports (n = 23) and cross-sectional surveys (n = 17). Most studies (72.4%) included participants from both sexes. Information about ethnicity was under-reported in most studies.
Table 1: Study characteristics (n = 152)
*Six studies were conducted across multiple countries
†Switzerland, Romania, Belgium (n = 4); Spain, Hungary, Netherlands, Poland, Lithuania, Bulgaria (n = 3); Estonia, Finland, Czech Republic, Denmark, Latvia, India (n = 2); Sweden, Mexico, Taiwan, Europe, Austria, Italy, Hong Kong, Brazil, Thailand, France, South Africa, New Zealand, Latin America (n = 1)
‡Ninety-six studies included participants from multiple age groups
§One hundred four studies involved cancer diagnoses across multiple body systems
¶Included germ cell cancer (n = 2), adenocarcinoma (n = 2), solid tumours (n = 2), anaplastic carcinoma (n = 1), small cell cancer (n = 1), mesothelioma (n = 1)
CBP were used across a wide spectrum of cancer diagnoses, with gastrointestinal, liver or peritoneal cancer (64.5%) and haematological or lymphoid cancer (60.5%) being the most common. The most common exclusion criteria for participants across the included studies were psychiatric risk factors (50.0%), either a history of or current drug/alcohol/tobacco use (34.9%), cardiovascular risk factors (30.2%), hepatic risk factors (26.7%), renal risk factors (24.4%) and neurological risk factors (19.8%) risk factors.
Other characteristics of the included studies were the range of comparators and concomitant treatments being used. Depending on the reason for CBP use, the comparator interventions reported were placebo, prochlorperazine, thiethylperazine, levomepromazine, triflupromazine, metoclopramide, domperidone, dimenhydrinate, megestrol acetate, ondansetron, codeine and secobarbital. Reported concomitant treatments included biological therapy, chemotherapy, radiotherapy, immunotherapy, hormone therapy, opioids, other analgesics, corticosteroids, antiemetics, antiepileptics, psychotropics and cardiovascular therapy. However, a notable proportion of the studies (19.1%) did not report any information about concomitant interventions. Seventeen studies [ref. 46, ref. 61, ref. 64, ref. 66, ref. 77, ref. 92, ref. 131, ref. 135, ref. 136, ref. 143, ref. 146, ref. 158, ref. 160, ref. 163, ref. 168, ref. 169, ref. 174] reported participants’ recreational use of CBP.
Cannabis-based products (CBP) characteristics and context
Table 2 summarises the characteristics of CBP (ingredient, form and route of administration) and the context of use.
Table 2: CBP characteristics and context of use (n = 152)
*Thirteen studies involved multiple CBP ingredients
†Eighteen studies involved multiple routes of administration
‡Intramuscular n = 9, intravenous n = 1, intracranial n = 1
§Enteral n = 2, both ears n = 1, free form n = 1, intranasal n = 1
¶Thirty-three studies involved multiple CBP indications
#Appetite, arthralgia/myalgia, chemosensory perception, drowsiness, general discomfort, headache, itch, night sweats, other, palliative wound care, unclear purpose, quality of life, spasticity (vismodegib-related muscle cramps), strengthening body to combat cancer, toxicity, use as a supplement
CBP ingredient
The main CBP ingredients reported across the studies were combinations of THC and CBD (45.4%), followed by synthetic THC (30.9%) and single compounds of THC (27.6%) and CBD (10.5%), noting some studies reported use of more than one formulation. Sometimes, the exact ratio of THC to CBD was reported (e.g. 150:0, 15:0, 100:1, 20:1, 15:1, 2:1, 1:1, 1:2, 1:20, 1:50, 1:100), whilst other studies simply stated the CBP was balanced, high or low THC:CBD ratio, and for others, it was inferred (e.g. Cannabis sativa L., marijuana, mixed, dried cannabis, prescribed cannabis medication).
CBP form
A diverse range of forms of CBP were utilised across studies including capsules, oils, solutions, extracts, cigarettes, pastes, creams, ointments, sprays, infusions, lotions, suppositories, edibles, inflorescence, flowers and plants. There were substantial variations in the dosage and duration of administration.
CBP administration route
The administration routes of CBP varied widely. The predominant methods of administration were oral (61.8%), followed by inhalation (35.5%).
CBP source
The source of CBP interventions, whether obtained illicitly, self-prescribed or physician-prescribed, was inadequately documented across the studies, and is therefore not reported.
Context of CBP use
Reasons for CBP use also varied considerably, frequently involving multiple conditions within a single study. Nausea and vomiting (51.3%) were identified as the most common reason, followed by cancer pain (24.3%). Only 7.9% of the studies reported participants’ comorbidities.
Adverse event (AE) characteristics
Table 3 presents the distribution of AEs by CTCAE SOC involving CBP use. Nervous system (77.6%), psychiatric (66.4%) and gastrointestinal AEs (53.3%) were the most common. The majority of studies (80.9%) reported AEs belonging to multiple CTCAE SOCs. Further details are reported in S6.
Table 3: Distribution of AEs by CTCAE SOC (n = 152)
| CTCAE SOC | No. (%)* |
|---|---|
| Nervous system disorders | 118 (77.6%) |
| Psychiatric disorders | 101 (66.4%) |
| Gastrointestinal disorders | 81 (53.3%) |
| General disorders and administration site conditions | 62 (40.8%) |
| Vascular disorders | 43 (28.3%) |
| Metabolism and nutrition disorders | 34 (22.4%) |
| Eye disorders | 32 (21.1%) |
| Cardiac disorders | 31 (20.4%) |
| Respiratory, thoracic and mediastinal disorders | 21 (13.8%) |
| Death | 20 (13.2%) |
| Skin and subcutaneous tissue disorders | 17(11.2%) |
| Ear and labyrinth disorders | 12 (7.9%) |
| Infections and infestations | 12 (7.9%) |
| Musculoskeletal and connective tissue disorders | 12 (7.9%) |
| Investigations | 12 (7.9%) |
| Blood and lymphatic system disorders | 7 (4.6%) |
| Renal and urinary disorders | 7 (4.6%) |
| Neoplasms benign, malignant and unspecified (incl cysts and polyps) | 6 (3.9%) |
| Drug interactions | 4 (2.6%) |
| Hepatobiliary disorders | 3 (2%) |
| Endocrine disorders | 2 (1.3%) |
| Reproductive system and breast disorders | 2 (1.3%) |
| Congenital, familial and genetic disorders | 1 (0.7%) |
| Immune system disorders | 1 (0.7%) |
| Injury, poisoning and procedural complications | 1 (0.7%) |
*One hundred twenty-three studies reported AEs in multiple CTCAE SOCs
AEs were inconsistently described across the studies. Sixteen studies applied the CTCAE grading system for reporting severity of the AEs [ref. 30, ref. 36, ref. 38, ref. 39, ref. 71, ref. 72, ref. 76, ref. 90, ref. 116, ref. 119, ref. 121, ref. 132, ref. 147, ref. 155, ref. 165, ref. 166]. Other studies employed a combination of serious/non-serious and non-specific grading (e.g. mild/moderate/severe) with many AEs ungraded. Details such as timepoint of AE occurrence, frequency, length of follow-up, clinical outcome, impact on quality of life, AE risk measures and causality were not consistently reported.
AEs were reported by study participants, healthcare practitioners or research staff via questionnaires or surveys conducted through phone calls or in-person visits, health forums and social media platforms.
Blood and lymphatic system disorders
There were seven studies [ref. 72, ref. 73, ref. 87, ref. 92, ref. 134, ref. 155, ref. 166] that reported AEs impacting the blood and lymphatic system. These included anaemia, febrile neutropenia, risk of bleeding and haematological toxicity. Ten studies [ref. 29, ref. 40, ref. 72, ref. 86, ref. 90, ref. 92, ref. 103, ref. 134, ref. 151, ref. 166] reported abnormal haematology investigations including decreased neutrophil count, decreased platelet count, low blood count, pancytopenia, prolonged activated partial and thromboplastin time (APTT) associated with oral and inhaled formulations. In general, formulations containing CBD are more commonly implicated in haematological abnormalities [ref. 180].
Cardiac disorders
Thirty-one studies [ref. 57–ref. 61, ref. 70, ref. 71, ref. 78–ref. 80, ref. 89, ref. 90, ref. 95, ref. 99, ref. 100, ref. 115, ref. 120, ref. 126, ref. 132, ref. 150, ref. 159, ref. 163, ref. 168, ref. 174] reported cardiac AEs including secondary myocardial infarction, chest pain, tachycardia, palpitations, bradycardia, cardiovascular event, atrial fibrillation and irregular heartbeat identified with a prolonged electrocardiogram QTc interval. Whilst undefined formulations and those containing various combinations of CBD and THC were associated with cardiac disorders, THC is reported to be the main constituent associated with AEs affecting the cardiovascular system [ref. 181].
Congenital, familial and genetic disorders
Newborn withdrawal syndrome in the first 24 h of life and neonatal peritonitis and intestinal invagination on the 2nd day of life were reported in one neonate whose mother used topical cannabis oil and 1–5 mL pure cannabis oil three times a day at 26 weeks of pregnancy [ref. 82].
Ear and labyrinth disorders
Twelve studies [ref. 29, ref. 33, ref. 34, ref. 69, ref. 88, ref. 95, ref. 116, ref. 123–ref. 126, ref. 155] reported AEs related to the ear and labyrinth, including vertigo, tinnitus, auditory disorders, ears buzzing, decreased hearing and noise sensitivity.
Endocrine disorders
Two studies [ref. 36, ref. 161] reported AEs affecting the thyroid gland, specifically hypothyroidism and thyroid disorders.
Eye disorders
Thirty-two studies [ref. 29, ref. 33, ref. 34, ref. 43, ref. 46, ref. 47, ref. 52, ref. 53, ref. 56, ref. 61, ref. 67, ref. 78, ref. 79, ref. 88, ref. 89, ref. 92, ref. 95, ref. 98–ref. 101, ref. 106, ref. 122, ref. 124–ref. 126, ref. 132, ref. 145–ref. 147, ref. 150, ref. 166] reported ophthalmic AEs. These included blurred vision, visual distortions, decreased/increased mean intraocular pressure, heavy eyed, ocular swelling and irritation, vision disturbance, amblyopia, visual scotoma, bilateral eye pain, photophobia, eye erythema, swollen eyelids, xerophalmia, dry eyes, vision alterations, itchy eyes, visual floaters and pupil dilation.
Gastrointestinal disorders
Eighty-one studies [ref. 29–ref. 31, ref. 33–ref. 35, ref. 40, ref. 46–ref. 48, ref. 50–ref. 53, ref. 56, ref. 58, ref. 59, ref. 61, ref. 65–ref. 67, ref. 71, ref. 72, ref. 74, ref. 76, ref. 78–ref. 80, ref. 82, ref. 85–ref. 90, ref. 95, ref. 97–ref. 100, ref. 103–ref. 107, ref. 111, ref. 114, ref. 116, ref. 119–ref. 125, ref. 127, ref. 130, ref. 132–ref. 135, ref. 138, ref. 139, ref. 147, ref. 149–ref. 151, ref. 154–ref. 156, ref. 159, ref. 161, ref. 163, ref. 166–ref. 168, ref. 172–ref. 174, ref. 176, ref. 178, ref. 179] reported AEs involving the gastrointestinal system. These included diarrhoea, dry mouth, nausea, vomiting, ascites, cannabinoid hyperemesis syndrome, abdominal pain/cramps/discomfort, sore mouth, GI irritation, constipation, epigastric distress, GERD, oral dysesthesia, persistent CINV, neutropenic colitis with perforation, obstipation, faecal incontinence, aerophagy, gastric ulcer haemorrhage, dysphagia, stomatitis, faecaloma, dyspepsia, mouth ulcers and thirst.
General disorders and administration site conditions
Sixty-two studies [ref. 29–ref. 36, ref. 39, ref. 40, ref. 46–ref. 48, ref. 52, ref. 57, ref. 61, ref. 66, ref. 72, ref. 75, ref. 76, ref. 78, ref. 81, ref. 83, ref. 85–ref. 90, ref. 95, ref. 97–ref. 99, ref. 102, ref. 103, ref. 106–ref. 108, ref. 114, ref. 122, ref. 127, ref. 132–ref. 134, ref. 142, ref. 143, ref. 146–ref. 148, ref. 150, ref. 151, ref. 155, ref. 156, ref. 161, ref. 165–ref. 169, ref. 171, ref. 172, ref. 178] reported general disorders and administration site AEs including fatigue, distal paresis of arm, pain (general, chest), altered general functioning, general deterioration, deteriorated clinical condition, declining performance status, worsened interference with activities of daily living, inactivity, postural dizziness, physiological side effects, hypothermia, weakness, fever, chills, asthenia, oedema, injection site reaction, local irritation, unsteady feet, unpleasant sensations (related to inhaling cannabis smoke) and gait disturbance.
Hepatobiliary disorders
Three studies [ref. 36, ref. 73, ref. 87] reported hepatobiliary AEs. A case–control study of immunotherapy in cancer [ref. 36] documented one case of hepatitis (CTCAE grade ≥ 2) in each group comprising of cannabis users (n = 1, 1.5%) and non-users (n = 1, 3%). The authors noted that they were classified as immune-related AEs even though relation to immunotherapy was not completely defined. An open-label extension study to investigate the long-term safety and tolerability of CBP in terminal cancer-related pain refractory to strong opioid analgesics [ref. 87] reported one case of hepatobiliary disorder in each group using balanced THC/CBD oromucosal spray (n = 1, 3%) and THC oromucosal spray (n = 1, 25%). There was no information on whether the events were serious, or treatment-related. Increased rates of hepatotoxicity leading to liver injury from CBD-drug interactions were observed in a cross-sectional survey [ref. 73] (see “Drug Interactions” section). Studies that reported abnormal liver investigations [ref. 86, ref. 90] are also listed in the “Investigations” section.
Immune system disorders
Immune-related AEs (CTCAE grade ≥ 2) were reported in a case–control study where CBP was used by its participants during immunotherapy [ref. 36]. Specific details of these AEs were not disclosed.
Infections and infestations
Twelve studies [ref. 32, ref. 40, ref. 42, ref. 72, ref. 86, ref. 87, ref. 90, ref. 92, ref. 136, ref. 158, ref. 160, ref. 166] reported infections and infestations that included thrush, UTI, pneumonia, invasive pulmonary aspergillosis, oral candidiasis, fungal infection (chest, disseminated), disseminated Fusarium infection, Campylobacter gastroenteritis and lower respiratory tract infection. CBD is the most common constituent observed to be associated with infections [ref. 180].
Injury, poisoning and procedural complications.
An open-label extension study to investigate the long-term safety and tolerability of balanced THC/CBD oromucosal spray and THC oromucosal spray in terminal cancer-related pain refractory to strong opioid analgesics reported AEs in this SOC [ref. 87]. Specific details regarding these AEs were not disclosed.
Investigations
Twelve studies [ref. 29, ref. 40, ref. 72, ref. 86, ref. 87, ref. 90, ref. 92, ref. 103, ref. 134, ref. 151, ref. 163, ref. 166] reported AEs identified from investigations, including decreased neutrophil count, decreased platelet count, low blood count, pancytopenia, prolonged activated partial thromboplastin time (APTT), increased gamma-glutamyl transferase (GGT), increased alanine aminotransferase (ALT), increased creatinine, prolonged electrocardiogram QTc interval and weight gain/loss.
Metabolism and nutrition disorders
Thirty-four studies [ref. 31, ref. 34, ref. 35, ref. 40, ref. 43, ref. 46, ref. 56, ref. 63, ref. 76, ref. 78, ref. 80, ref. 86, ref. 87, ref. 90, ref. 93, ref. 103, ref. 106, ref. 114, ref. 125, ref. 126, ref. 130, ref. 132, ref. 134, ref. 146–ref. 149, ref. 151, ref. 154, ref. 155, ref. 161, ref. 168, ref. 169, ref. 174] reported AEs related to metabolism and nutritional disorders including appetite increase/loss, anorexia, increased food intake, hunger, dehydration, hyperglycaemia, hypoalbuminemia, hypocalcaemia, hypokalaemia, hypomagnesemia, hyponatremia and hypophosphatemia.
Musculoskeletal and connective tissue disorders
Twelve studies [ref. 33, ref. 34, ref. 43, ref. 52, ref. 87, ref. 95, ref. 125, ref. 132, ref. 147, ref. 150, ref. 161, ref. 166] reported musculoskeletal and connective tissue AEs, including muscle twitching/pain/weakness, leg cramps, limb pain/weakness, arthralgia, back/bone/joint pain, jaw stiffness, decreased motor ability and tremor.
Neoplasms benign, malignant and unspecified (incl cysts and polyps)
Six studies [ref. 86, ref. 87, ref. 103, ref. 134, ref. 155, ref. 166] reported AEs related to neoplasms including tumour progression, tumour-related pain, tumour haemorrhage and metastases to the brain.
Nervous system disorders
AEs impacting the nervous system were reported in 118 studies [ref. 29–ref. 35, ref. 38–ref. 41, ref. 43, ref. 44, ref. 46–ref. 54, ref. 56–ref. 63, ref. 65–ref. 69, ref. 71–ref. 74, ref. 76, ref. 78–ref. 80, ref. 83, ref. 85–ref. 90, ref. 92–ref. 95, ref. 98–ref. 100, ref. 102–ref. 108, ref. 111, ref. 113, ref. 114, ref. 116, ref. 118–ref. 126, ref. 128, ref. 130, ref. 132–ref. 134, ref. 137–ref. 139, ref. 141–ref. 147, ref. 149–ref. 157, ref. 159, ref. 162–ref. 169, ref. 171, ref. 172, ref. 174–ref. 179]. Examples included drowsiness, somnolence, hypersomnia, sedation, lethargy, CNS depression, dizziness, slurred/impaired speech, neuralgia, headache, amnesia, dysgeusia, impaired motor coordination/balance, decreased concentration, paraesthesia, cognitive decline, seizures, syncope, falls, impaired awareness/thinking, cannabinoid-induced alteration of motor-evoked potentials, aphasia and dysarthria. Undefined and defined formulations administered via the oral and inhaled route were associated with AEs involving neurological symptoms. AEs involving the nervous system were mainly attributed to oral and inhaled formulations containing THC, noting some involved combinations of CBD and THC. THC is reported to be the most common constituent associated with AEs affecting the nervous system [ref. 181].
Psychiatric disorders
Psychiatric AEs were reported in 101 studies [ref. 28, ref. 29, ref. 31, ref. 33–ref. 35, ref. 39, ref. 40, ref. 43–ref. 45, ref. 47–ref. 53, ref. 56–ref. 63, ref. 65–ref. 69, ref. 71, ref. 72, ref. 75, ref. 76, ref. 78–ref. 80, ref. 83, ref. 85–ref. 90, ref. 93–ref. 96, ref. 98–ref. 100, ref. 103–ref. 107, ref. 111, ref. 113, ref. 114, ref. 116, ref. 118–ref. 120, ref. 122–ref. 128, ref. 131–ref. 134, ref. 139, ref. 141, ref. 144–ref. 147, ref. 150–ref. 152, ref. 154, ref. 156, ref. 157, ref. 159, ref. 163, ref. 166–ref. 171, ref. 174–ref. 179]. Examples included insomnia, mood disorders (euphoria, feeling “high” or “drunk”, irritability, agitation, dysphoria), hallucination (auditory, visual), panic/fear, anxiety, depression, confusion/disorientation, intoxication, cannabinoid intoxication, psychic disturbance, bulimia, bad/wild/livid dreams, relaxed, depersonalisation/dissociation, distorted perception (time and space), delusions, apathy, paranoia, loss of motivation, psychomimetic effects, hyperventilation, tetanic symptoms, restlessness, personality/behavioural change, psychosis, libido change and hyperactivity. Undefined and defined formulations administered via the oral and inhaled route were associated with psychiatric disorders. However, THC is reported to be the common constituent associated with AEs affecting mental health [ref. 182].
Renal and urinary disorders
Seven studies [ref. 47, ref. 86, ref. 87, ref. 90, ref. 147, ref. 166, ref. 167] reported renal and urinary AEs, including urinary retention, cystitis, haematuria and increased nocturia. A phase 1 study of dexanabinol in brain cancer [ref. 90] reported treatment-related elevated creatinine (CTCAE grade 1) (n = 1, 3.8%) with 36 mg/kg dexanabinol (see the “Investigations” section).
Reproductive system and breast disorders
Two RCTs involving dronabinol reported AEs involving the reproductive system. One noted a case of vaginal discharge [ref. 40] whilst another documented cases of male impotence [ref. 85].
Respiratory, thoracic and mediastinal disorders
Twenty-one studies [ref. 33, ref. 35, ref. 40, ref. 48, ref. 61, ref. 73, ref. 76, ref. 87, ref. 92, ref. 98, ref. 99, ref. 116, ref. 117, ref. 127, ref. 147, ref. 154, ref. 155, ref. 163, ref. 166, ref. 171, ref. 178] reported respiratory AEs, most commonly dyspnoea. Others included cough, sore/burning throat, hoarseness, rhinorrhoea, rhinitis, nasal congestion, pneumonitis, respiratory depression and respiratory failure.
Skin and subcutaneous tissue disorders
Seventeen studies [ref. 36, ref. 40, ref. 55, ref. 56, ref. 71, ref. 87, ref. 90, ref. 95, ref. 125, ref. 132, ref. 147, ref. 149, ref. 150, ref. 156, ref. 161, ref. 163, ref. 174] reported AEs involving the skin, such as rash, dry skin, pruritus, urticaria, skin toxicity, hyperhidrosis and alopecia.
Vascular disorders
Forty-three studies [ref. 29, ref. 33, ref. 34, ref. 43, ref. 47, ref. 52, ref. 57–ref. 60, ref. 62, ref. 67, ref. 69, ref. 71, ref. 72, ref. 78–ref. 80, ref. 83, ref. 86–ref. 89, ref. 99, ref. 100, ref. 102–ref. 104, ref. 116, ref. 120, ref. 123–ref. 126, ref. 132, ref. 133, ref. 138, ref. 139, ref. 150, ref. 152, ref. 154, ref. 156, ref. 167] reported vascular AEs with the most common being hypotension and postural hypotension. Others included hypertension, hot flashes, flushing and a thromboembolic event (pulmonary embolism).
Drug interactions
Four studies reported CBP-drug interactions. A phase 1 study of dexanabinol in brain cancer [ref. 90] reported a case of treatment-related steroid myopathy (CTCAE grade 2) (n = 1, 3.8%) with 4 mg/kg dexanabinol. A case report noted the potential inhibition of CYP3A4 and/or CYP2D6 resulting in diminished metabolism of tamoxifen with a low dose CBD of 40 mg/day [ref. 129].
A cross-sectional survey by Guedon et al. [ref. 73] on CBD use across diverse cancer stages found increased CNS depression was observed in the concomitant use of CBD and morphine, metoclopramide, codeine, fentanyl, tramadol and altered vigilance with cetirizine, levocetirizine, hydroxyzine, alprazolam, quetiapine, zopiclone, clorazepate, sertraline and paroxetine. They also observed that the concomitant use of CBD with oxaliplatin, bortezomib, lenalidomide, dacarbazine, vincristine and methotrexate may lead to an increase in pre-existing hepatotoxicity. Higher rates of hepatotoxicity leading to liver injury were reported with the concomitant use of CBD and sulfamethoxazole-trimethoprim, paracetamol, pravastatin, amiodarone, ramipril or perindopril. Additionally, respiratory depression occurred with morphine, buprenorphine and dexchlorpheniramine whilst increased CBD exposure was noted with aprepitant, ketoconazole and omeprazole. CBD use enhanced exposure to morphine, sirolimus, apixaban and rivaroxaban leading to increased risk of bleeding with rivaroxaban and apixaban. The authors concluded that CBD use posed two primary clinical risks of CNS depression and hepatotoxicity due to drug interactions.
In another cross-sectional survey by Saadeh et al. [ref. 143] on medical marijuana use across varying cancer stages, drug interactions were reported with sympathomimetic agents, cytochrome P450 (CYP) isoform 1A2 substrates, CYP2C9 inhibitors and anticholinergic agents, with the highest occurrence noted with CNS depressants.
Death
Twenty studies [ref. 29, ref. 34, ref. 35, ref. 65, ref. 66, ref. 85–ref. 87, ref. 92, ref. 103, ref. 110, ref. 127, ref. 134, ref. 142, ref. 148, ref. 155, ref. 158, ref. 165, ref. 166, ref. 173] reported deaths among participants using a CBP. Of these, the authors of ten studies determined that causality was not attributed to CBP use and the remaining did not comment. There were no studies that reported death being directly attributed to CBP use. Further details are provided in S7.
Discussion
In this review, a wide range of AEs across all CTCAE categories were reported to be associated with a variety of CBP ingredients, forms/types, routes of administration, doses and duration of use, reason for use and patient characteristics. Low-quality reporting and substantial under-reporting of AEs were also identified in many studies. Challenges with synthesising the findings were further compounded by the heterogeneity in the included study designs, as well as the variations in formulations and dosages that often lacked detailed information.
Notwithstanding the limitations that prevented a complete summary of the AEs associated with CBP use, patterns were observed that warrant further research. For instance, many of the nervous system, psychiatric, gastrointestinal and cardiovascular AEs were predictable based on the known pharmacological effects of CBP and its constituents. Some of the AEs involving CNS depression, hepatotoxicity and cardiovascular events could pose serious health risks. These may be reduced with diligent clinical monitoring, careful dosage adjustments and other appropriate precautions [ref. 183].
The extensive range of synthetic and natural CBP used, including blends or pure forms of THC and CBD, was noteworthy. The different pharmacological actions of CBP constituents increase the risks of specific AEs. In general, THC is primarily associated with cardiovascular, neurological and psychiatric AEs [ref. 181, ref. 182], whilst CBD is implicated in drug interactions, hepatic injury, infections and haematological abnormalities [ref. 180]. The diversity in the forms and routes of CBP administration presents a distinct set of potential AEs. Smoking and inhalation can cause respiratory issues including bronchoconstriction and impairment of airway function. Oral ingestion may result in delayed onset of effects and AEs from overuse [ref. 184]. The wide variability in doses and duration of CBP use reported may collectively contribute to differences in the types and severity of AEs experienced [ref. 185, ref. 186]. Moreover, patients may self-administer CBP at disparate doses and durations based on individual preferences, severity of symptoms and perceived efficacy. Clinicians should consider these variables in the clinical management and monitoring of CBP use to help minimise patient harm. The standardisation of CBP formulations and protocols for use requires urgent attention.
The complex interplay of factors unique to each patient, such as reasons for CBP use, cancer type and stage, genetic variations, comorbidities and concomitant medications, may exert a significant impact on the manifestation of AEs. Patients living with different types of cancer may be susceptible to different AEs related to CBP use due to variations in tumour biology, treatment modalities and systemic effects [ref. 186]. The stage of cancer at which CBP are initiated can also influence the development of AEs. Patients with advanced cancer may experience more severe AEs or develop complications that interact with CBP. Genetic polymorphisms can affect the pharmacokinetics and pharmacodynamics of CBP, thereby influencing AE susceptibility and severity [ref. 187]. Individual tolerance and response to CBP may also be impacted by other pre-existing medical conditions. This necessitates vigilant monitoring and dose adjustments where required. In addition, the concurrent use of other medications may result in drug interactions with CBP which amplify AEs [ref. 188]. Therefore, a personalised approach that accounts for these unique patient characteristics is required.
Challenges with conducting this review included substantial under- or incomplete reporting of AEs. Indeed, 150 studies of CBP use in cancer populations were excluded for this reason. The observed under-reporting of AEs raises important questions about the reliability and completeness of existing data on the safety profile of CBP in cancer care. Improving AE reporting mechanisms is vital for advancing our understanding of CBP safety and promoting evidence-based decision-making in the oncology setting.
This review is not without limitations. Relevant studies may have been missed as grey literature, AE systems and clinical trial databases were omitted. Additionally, we did not contact the authors of potentially relevant excluded studies for further information. A risk of language bias was introduced by limiting to articles in English. An updated search was not conducted, nor did we conduct additional searches for any of the serious AEs reported due to the initial amount of data captured. Notwithstanding, given the large number and breadth of studies included, it is likely that most of the potential AEs associated with CBP use in this population were identified. The exception might be rare AEs that are yet to be reported in a case study or have only been reported in post-marketing surveillance.
Future recommendations include one or more systematic reviews to be undertaken to evaluate the risk of AEs associated with specific CBP and indications. Reviewers should employ critical methodologies to identify, analyse and synthesise existing evidence, providing detailed AE information associated with specific CBP formulations used within an oncology setting. Additionally, researchers should adhere to standardised protocols for reporting clinical studies that include detailed information about the intervention and outcomes.
Conclusion
The reports included in this review provide preliminary evidence to suggest that AEs are associated with CBP use in the context of cancer care. AEs associated with CBP use were reported across a broad subset of this population and involve a range of defined as well as poorly defined formulations and doses that can impact every body system. The overall inconsistent approaches and standards in the reporting, and under-reporting, of CBP-associated AEs suggest substantial gaps in our knowledge of these AEs that further complicates the assessment of the safety profile of CBP. By addressing these gaps, healthcare providers and policymakers will be better placed to make informed decisions regarding the use of CBP as part of cancer care.
Supplementary Materials
References
- DI Abrams. Cannabis, Cannabinoids and cannabis-based medicines in cancer care. Integr Cancer Ther, 2022. [PubMed]
- K Martell, A Fairchild, B LeGerrier, R Sinha, S Baker, H Liu. Rates of cannabis use in patients with cancer. Curr Oncol, 2018. [PubMed]
- I Osaghae, OG Chido-Amajuoyi, BAA Khalifa, R Talluri, S Shete. Cannabis use among cancer survivors: use pattern, product type, and timing of use. Cancers (Basel), 2023. [PubMed]
- L Mills, N Lintzeris, M O’Malley, JC Arnold, IS McGregor. Prevalence and correlates of cannabis use disorder among Australians using cannabis products to treat a medical condition. Drug Alcohol Rev, 2022. [PubMed]
- YD Chang, JS Smith, R Oberoi-Jassal, V Desai, SL Winn, D Portman. Cannabis use in palliative care: the prevalence and clinical characteristics. Am Soc Clin Oncol, 2017
- KA Donovan, YD Chang, R Oberoi-Jassal, S Rajasekhara, J Smith, M Haas. Relationship of cannabis use to patient-reported symptoms in cancer patients seeking supportive/palliative care. J Palliat Med, 2019. [PubMed]
- K Tringale, Y Shi, J Hattangadi. Marijuana utilization in Cancer patients: a comprehensive analysis of National Health and nutrition examination survey data from 2005–2014. Int J Radiat Oncol Biol Phys, 2017
- M Horneber, G Bueschel, G Dennert, D Less, E Ritter, M Zwahlen. How many cancer patients use complementary and alternative medicine: a systematic review and metaanalysis. Integr Cancer Ther, 2011. [PubMed]
- 9.DISA. Marijuana Legality by State 2024 [cited 18 May 2024]. Available from: https://disa.com/marijuana-legality-by-state
- 10.Substance Abuse and Mental Health Services Administration. Key substance use and mental health indicators in the United States: results from the 2022 National Survey on Drug Use and Health. HHS publication no. PEP23–07–01–006 2023: Rockville, MD: Center for behavioral health statistics and quality, substance abuse and mental health services administration; [cited 18 May 2024]. Available from: https://www.samhsa.gov/data/
- 11.National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Committee on the Health Effects of Marijuana: An Evidence Review and Research Agenda. The health effects of cannabis and cannabinoids: the current state of evidence and recommendations for research. Washington (DC): National Academies Press (US) 2017 Jan 12 [Available from: 10.17226/24625
- I Turgeman, G Bar-Sela. Cannabis use in palliative oncology: a review of the evidence for popular indications. Isr Med Assoc J, 2017. [PubMed]
- D Gloss. An overview of products and bias in research. Neurotherapeutics, 2015. [PubMed]
- J Ablin, PA Ste-Marie, M Schäfer, W Häuser, MA Fitzcharles. Medical use of cannabis products: lessons to be learned from Israel and Canada. Schmerz, 2016. [PubMed]
- 15.National Conference of State Legislatures. State Medical Cannabis Laws [updated 22 June 2023, cited 07 March 2024]. Available from: https://www.ncsl.org/health/state-medical-cannabis-laws
- PP Bathula, MB Maciver. Cannabinoids in treating chemotherapy-induced nausea and vomiting, cancer-associated pain, and tumor growth. Int J Mol Sci, 2023. [PubMed]
- LA Smith, F Azariah, VT Lavender, NS Stoner, S Bettiol. Cannabinoids for nausea and vomiting in adults with cancer receiving chemotherapy. Cochrane Database Syst Rev, 2015. [PubMed]
- B Dariš, M Tancer Verboten, Ž Knez, P Ferk. Cannabinoids in cancer treatment: therapeutic potential and legislation. Bosn J Basic Med Sci, 2019. [PubMed]
- MP Davis. Cannabinoids for symptom management and cancer therapy: the evidence. J Natl Compr Canc Netw, 2016. [PubMed]
- 20.Peters MDJ, Godfrey CMP, Munn Z, Tricco AC, Khalil H, editors. (2020) Chapter 11: scoping reviews (2020 version). In: Aromataris E, Munn Z (Editors). JBI Manual for Evid Syn
- AC Tricco, E Lillie, W Zarin, KK O’Brien, H Colquhoun, D Levac. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med, 2018. [PubMed]
- 22.Equator Network. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews [updated 10 Nov 2022, cited 16 Feb 2023]. Available from: https://www.equator-network.org/reporting-guidelines/prisma/
- I Cheah, I Gelissen, J Hunter, J Harnett. Adverse events associated with the use of cannabis-based products in people living with cancer: a scoping review protocol. Eur J Integr Med, 2023
- 24.International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. ICH harmonised tripartite guideline. Post-approval safety data management: definitions and standards for expedited reporting E2D. Step 4 version. Dated 12 November 2003 [cited 16 Feb 2023]. Available from: https://database.ich.org/sites/default/files/E2D_Guideline.pdf
- 25.Covidence systematic review software [Internet]. Veritas Health Innovation, Melbourne, Australia. Available from: www.covidence.org
- 26.National Cancer Institute. CTEP cancer therapy evaluation program [updated 19 Apr 2021, cited 12 Mar 2023]. Available from: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_60
- 27.Microsoft Corporation. Microsoft Excel [Internet]. 2011. Available from: https://office.microsoft.com/excel
- A Abrahamov, R Mechoulam. An efficient new cannabinoid antiemetic in pediatric oncology. Life Sci, 1995. [PubMed]
- S Ahmedzai, DL Carlyle, IT Calder, F Moran. Anti-emetic efficacy and toxicity of nabilone, a synthetic cannabinoid, in lung cancer chemotherapy. Br J Cancer, 1983. [PubMed]
- D Allen. Dronabinol therapy: central nervous system adverse events in adults with primary brain tumors. Clin J Oncol Nurs, 2019. [PubMed]
- SP Anderson, DM Zylla, DM McGriff, TJ Arneson. Impact of medical cannabis on patient-reported symptoms for patients with cancer enrolled in Minnesota’s medical cannabis program. J Oncol Practice, 2019
- S Aprikian, P Kasvis, M Vigano, Y Hachem, M Canac-Marquis, A Vigano. Medical cannabis is effective for cancer-related pain: Quebec Cannabis Registry results. BMJ Supportive Palliat Care, 2023
- J Aviram, GM Lewitus, Y Vysotski, MA Amna, A Ouryvaev, S Procaccia. The effectiveness and safety of medical cannabis for treating cancer related symptoms in oncology patients. Front Pain Res (Lausanne, Switzerland), 2022
- J Aviram, GM Lewitus, Y Vysotski, A Uribayev, S Procaccia, I Cohen. Short-term medical cannabis treatment regimens produced beneficial effects among palliative cancer patients. Pharmaceuticals (Basel, Switzerland), 2020. [PubMed]
- L Bar-Lev Schleider, R Mechoulam, V Lederman, M Hilou, O Lencovsky, O Betzalel. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med, 2018. [PubMed]
- G Bar-Sela, I Cohen, S Campisi-Pinto, GM Lewitus, L Oz-Ari, A Jehassi. Cannabis consumption used by cancer patients during immunotherapy correlates with poor clinical outcome. Cancers, 2020. [PubMed]
- G Bar-Sela, D Tauber, I Mitnik, H Sheinman-Yuffe, T Bishara-Frolova, J Aharon-Peretz. Cannabis-related cognitive impairment: a prospective evaluation of possible influences on patients with cancer during chemotherapy treatment as a pilot study. Anticancer Drugs, 2018
- G Bar-Sela, M Vorobeichik, S Drawsheh, A Omer, V Goldberg, E Muller. The medical necessity for medicinal cannabis: prospective, observational study evaluating the treatment in cancer patients on supportive or palliative care. Evid-Based Complement Altern Med : eCAM, 2013
- G Bar-Sela, D Zalman, V Semenysty, E Ballan. The effects of dosage-controlled cannabis capsules on cancer-related cachexia and anorexia syndrome in advanced cancer patients: pilot study. Integr Cancer Ther, 2019. [PubMed]
- TD Brisbois, IH de Kock, SM Watanabe, M Mirhosseini, DC Lamoureux, M Chasen. Delta-9-tetrahydrocannabinol may palliate altered chemosensory perception in cancer patients: results of a randomized, double-blind, placebo-controlled pilot trial. Ann Oncol, 2011. [PubMed]
- C Carr, H Vertelney, J Fronk, S Trieu. Dronabinol for the treatment of paraneoplastic night sweats in cancer patients: a report of five cases. J Palliat Med, 2019. [PubMed]
- DW Cescon, AV Page, S Richardson, MJ Moore, S Boerner, WL Gold. Invasive pulmonary aspergillosis associated with marijuana use in a man with colorectal cancer. J Clin Oncol, 2008. [PubMed]
- HS Chan, JA Correia, SM MacLeod. Nabilone versus prochlorperazine for control of cancer chemotherapy-induced emesis in children: a double-blind, crossover trial. Pediatrics, 1987. [PubMed]
- AE Chang, DJ Shiling, RC Stillman. Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate. A prospective, randomized evaluation. Ann Internal Med, 1979. [PubMed]
- AE Chang, DJ Shiling, RC Stillman, NH Goldberg, CA Seipp, I Barofsky. A prospective evaluation of delta-9-tetrahydrocannabinol as an antiemetic in patients receiving adriamycin and cytoxan chemotherapy. Cancer, 1981. [PubMed]
- S Chapman, J Protudjer, C Bourne, LE Kelly, S Oberoi, MI Vanan. Medical cannabis in pediatric oncology: a survey of patients and caregivers. Support Care Cancer, 2021. [PubMed]
- ML Citron, TS Herman, F Vreeland, SH Krasnow, BE Fossieck, S Harwood. Antiemetic efficacy of levonantradol compared to delta-9-tetrahydrocannabinol for chemotherapy-induced nausea and vomiting. Cancer Treat Rep, 1985. [PubMed]
- S Clarke, BE Butcher, AJ McLachlan, JD Henson, D Rutolo, S Hall. Pilot clinical and pharmacokinetic study of Δ9-tetrahydrocannabinol (THC)/cannabidiol (CBD) nanoparticle oro-buccal spray in patients with advanced cancer experiencing uncontrolled pain. PLoS ONE, 2022. [PubMed]
- BM Colls, DG Ferry, AJ Gray, VJ Harvey, EG McQueen. The antiemetic activity of tetrahydrocannabinol versus metoclopramide and thiethylperazine in patients undergoing cancer chemotherapy. N Z Med J, 1980. [PubMed]
- M Cote, M Trudel, C Wang, A Fortin. Improving quality of life with nabilone during radiotherapy treatments for head and neck cancers: a randomized double-blind placebo-controlled trial. Ann Otol Rhinol Laryngol, 2016. [PubMed]
- SM Crawford, R Buckman. Nabilone and metoclopramide in the treatment of nausea and vomiting due to cisplatinum: a double blind study. Med Oncol Tumor Pharmacother, 1986. [PubMed]
- CM Cronin, SE Sallan, R Gelber, VS Lucas, J Laszlo. Antiemetic effect of intramuscular levonantradol in patients receiving anticancer chemotherapy. J Clin Pharmacol, 1981. [PubMed]
- D Cunningham, CJ Bradley, GJ Forrest, AW Hutcheon, L Adams, M Sneddon. A randomized trial of oral nabilone and prochlorperazine compared to intravenous metoclopramide and dexamethasone in the treatment of nausea and vomiting induced by chemotherapy regimens containing cisplatin or cisplatin analogues. Eur J Cancer Clin Oncol, 1988. [PubMed]
- S D’Andre, S McAllister, J Nagi, KV Giridhar, E Ruiz-Macias, C Loprinzi. Topical cannabinoids for treating chemotherapy-induced neuropathy: a case series. Integr Cancer Ther, 2021. [PubMed]
- PB Dall’Stella, MFL Docema, MVC Maldaun, O Feher, CLP Lancellotti. Case report: clinical outcome and image response of two patients with secondary high-grade glioma treated with chemoradiation, PCV, and cannabidiol. Front Oncol, 2019. [PubMed]
- AM Dalzell, H Bartlett, JS Lilleyman. Nabilone: an alternative antiemetic for cancer chemotherapy. Arch Dis Child, 1986. [PubMed]
- BH Davies, RM Weatherstone, JDP Graham, RD Griffiths. A pilot study of orally administered Δ1-trans-tetrahydrocannabinol in the management of patients undergoing radiotherapy for carcinoma of the bronchus. Br J Clin Pharmacol, 1974. [PubMed]
- ML Devine, GJ Dow, BR Greenberg. Adverse reactions to delta-9-tetrahydrocannabinol given as an antiemetic in a multicenter study. Clin Pharm, 1987. [PubMed]
- RB Diasio, DS Ettinger, BE Satterwhite. Oral levonantradol in the treatment of chemotherapy-induced emesis: preliminary observations. J Clin Pharmacol, 1981. [PubMed]
- GJ Dow, FH Meyers, W Stanton, ML Devine. Serious reactions to oral delta-9-tetrahydrocannabinol in cancer chemotherapy patients. Clin Pharm, 1984. [PubMed]
- M Duran, E Perez, S Abanades, X Vidal, C Saura, M Majem. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br J Clin Pharmacol, 2010. [PubMed]
- LH Einhorn, C Nagy, B Furnas, SD Williams. Nabilone: an effective antiemetic in patients receiving cancer chemotherapy. J Clin Pharmacol, 1981. [PubMed]
- H Ekert, KD Waters, IH Jurk, J Mobilia, P Loughnan. Amelioration of cancer chemotherapy-induced nausea and vomiting by delta-9-tetrahydrocannabinol. Med J Aust, 1979. [PubMed]
- DA Elliott, N Nabavizadeh, JL Romer, Y Chen, JM Holland. Medical marijuana use in head and neck squamous cell carcinoma patients treated with radiotherapy. Support Care Cancer, 2016. [PubMed]
- MT Fallon, E Albert Lux, R McQuade, S Rossetti, R Sanchez, W Sun. Sativex oromucosal spray as adjunctive therapy in advanced cancer patients with chronic pain unalleviated by optimized opioid therapy: two double-blind, randomized, placebo-controlled phase 3 studies. Br J Pain, 2017. [PubMed]
- J Fehniger, AL Brodsky, A Kim, B Pothuri. Medical marijuana utilization in gynecologic cancer patients. Gynecol Oncol Rep, 2021. [PubMed]
- S Frytak, CG Moertel, JR O’Fallon, J Rubin, ET Creagan, MJ O’Connell. Delta-9-tetrahydrocannabinol as an antiemetic for patients receiving cancer chemotherapy. A comparison with prochlorperazine and a placebo. Ann Internal Med, 1979. [PubMed]
- S Frytak, CG Moertel, J Rubin. Metabolic studies of delta-9-tetrahydrocannabinol in cancer patients. Cancer Treat Rep, 1984. [PubMed]
- HH Gerhartz, T Binsack, E Hiller. Levonantradol for the treatment of chemotherapy-induced nausea and vomiting. Klin Wochenschr, 1983. [PubMed]
- CJ Gilbert, KV Ohly, G Rosner, WP Peters. Randomized, double-blind comparison of a prochlorperazine-based versus a metoclopramide-based antiemetic regimen in patients undergoing autologous bone marrow transplantation. Cancer, 1995. [PubMed]
- PD Good, RM Greer, GE Huggett, JR Hardy. An open-label pilot study testing the feasibility of assessing total symptom burden in trials of cannabinoid medications in palliative care. J Palliat Med, 2020. [PubMed]
- P Grimison, A Mersiades, A Kirby, N Lintzeris, R Morton, P Haber. Oral THC:CBD cannabis extract for refractory chemotherapy-induced nausea and vomiting: a randomised, placebo-controlled, phase II crossover trial. Ann Oncol, 2020. [PubMed]
- M Guedon, A Le Bozec, M Brugel, J Clarenne, C Carlier, M Perrier. Cannabidiol-drug interaction in cancer patients: a retrospective study in a real-life setting. British J Clin Pharmacol, 2023
- G Guy, J Gover, M Rogerson, B Atwell, J Dineen. Positive data in sativex phase IIb trial: support advancing into phase III development in cancer pain. Revista de la Sociedad Espanola del Dolor, 2010
- M Guzman, MJ Duarte, C Blazquez, J Ravina, MC Rosa, I Galve-Roperh. A pilot clinical study of Delta9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer, 2006. [PubMed]
- J Hardy, R Greer, G Huggett, A Kearney, T Gurgenci, P Good. Phase IIb randomized, placebo-controlled, dose-escalating, double-blind study of cannabidiol oil for the relief of symptoms in advanced cancer (MedCan1-CBD). J Clin Oncol, 2023. [PubMed]
- P Hawley, M Gobbo. Cannabis use in cancer: a survey of the current state at BC cancer before recreational legalization in Canada. Curr Oncol (Toronto, Ont), 2019
- ME Heim, W Romer, W Queisser. Clinical experience with levonantradol hydrochloride in the prevention of cancer chemotherapy-induced nausea and vomiting. J Clin Pharmacol, 1981. [PubMed]
- TS Herman, LH Einhorn, SE Jones, C Nagy, AB Chester, JC Dean. Superiority of nabilone over prochlorperazine as an antiemetic in patients receiving cancer chemotherapy. N Engl J Med, 1979. [PubMed]
- TS Herman, SE Jones, J Dean, S Leigh, R Dorr, TE Moon. Nabilone: a potent antiemetic cannabinol with minimal euphoria. Biomed / [publiee pour l’AAICIG], 1997
- K Hsu, E Whitham, G Kichenadasse. Potential role of cannabidiol for seizure control in a patient with recurrent glioma. J Clin Neurosci, 2020. [PubMed]
- A Huniadi, A Sorian, C Iuhas, A Bodog, MI Sandor. The effect of cannabis in the treatment of hodgkin’s lymphoma in a pregnant patient – extensive case report and literature review. J BUON, 2021. [PubMed]
- AW Hutcheon, JB Palmer, M Soukop, D Cunningham, C McArdle, J Welsh. A randomised multicentre single blind comparison of a cannabinoid anti-emetic (levonantradol) with chlorpromazine in patients receiving their first cytotoxic chemotherapy. Eur J Cancer Clin Oncol, 1983. [PubMed]
- S Jafri, E Hansen, R Fuenmayor, AA Case. Medical cannabis for insomnia in a patient with advanced breast cancer. J Pain Symptom Manage, 2023. [PubMed]
- A Jatoi, HE Windschitl, CL Loprinzi, JA Sloan, SR Dakhil, JA Mailliard. Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol, 2002. [PubMed]
- JR Johnson, M Burnell-Nugent, D Lossignol, ED Ganae-Motan, R Potts, MT Fallon. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manage, 2010. [PubMed]
- JR Johnson, D Lossignol, M Burnell-Nugent, MT Fallon. An open-label extension study to investigate the long-term safety and tolerability of THC/CBD oromucosal spray and oromucosal THC spray in patients with terminal cancer-related pain refractory to strong opioid analgesics. J Pain Symptom Manage, 2013. [PubMed]
- SE Jones, JR Durant, FA Greco, A Robertone. A multi-institutional phase III study of nabilone vs. placebo in chemotherapy-induced nausea and vomiting. Cancer Treatment Rev, 1982
- RA Joss, RL Galeazzi, A Bischoff, DD Do, A Goldhirsch, KW Brunner. Levonantradol, a new antiemetic with a high rate of side-effects for the prevention of nausea and vomiting in patients receiving cancer chemotherapy. Cancer Chemother Pharmacol, 1982. [PubMed]
- TM Juarez, D Piccioni, L Rose, A Nguyen, B Brown, S Kesari. Phase I dose-escalation, safety, and CNS pharmacokinetic study of dexanabinol in patients with brain cancer. Neuro-oncol Adv, 2021
- J Kenyon, W Liu, A Dalgleish. Report of objective clinical responses of cancer patients to pharmaceutical-grade synthetic cannabidiol. Anticancer Res, 2018. [PubMed]
- S Khwaja, A Yacoub, A Cheema, N Rihana, R Russo, AP Velez. Marijuana smoking in patients with leukemia. Cancer control, 2016. [PubMed]
- S Kleinman, SA Weitzman, N Cassem, E Andrews. Double blind trial of delta-9-tetrahydrocannabinol (THC) versus placebo as an adjunct to prochlorperazine for chemotherapy-induced vomiting. Curr Therapeut Res- Clin Exp, 1983
- CM Klier, GP Amminger, OD Kothgassner, C Laczkovics, A Felnhofer. Letter to the editor: cannabidiol treatment – is there an effect on cognitive functioning, quality of life, and behavior? A Case Report. J Child Adolesc Psychopharmacol, 2021. [PubMed]
- JC Kluin-Neleman, FA Neleman, OJ Meuwissen, RA Maes. delta 9-Tetrahydrocannabinol (THC) as an antiemetic in patients treated with cancerchemotherapy; a double-blind cross-over trial against placebo. Vet Hum Toxicol, 1979. [PubMed]
- TS Kutiel, O Vornicova, G Bar-Sela. Cannabis for vismodegib-related muscle cramps in a patient with advanced basal cell carcinoma. J Pain Symptom Manage, 2018. [PubMed]
- CS Lam, K Zhou, HH-F Loong, VC-H Chung, C-K Ngan, YT Cheung. The use of traditional, complementary, and integrative medicine in cancer: data-mining study of 1 million web-based posts from health forums and social media platforms. J Med Int Res, 2023
- M Lane, FE Smith, RA Sullivan, TF Plasse. Dronabinol and prochlorperazine alone and in combination as antiemetic agents for cancer chemotherapy. Am J Clin Oncol, 1990. [PubMed]
- M Lane, CL Vogel, J Ferguson, S Krasnow, JL Saiers, J Hamm. Dronabinol and prochlorperazine in combination for treatment of cancer chemotherapy-induced nausea and vomiting. J Pain Symptom Manage, 1991. [PubMed]
- J Laszlo, VS Lucas, DC Hanson, CM Cronin, SE Sallan. Levonantradol for chemotherapy-induced emesis: phase I-II oral administration. J Clin Pharmacol, 1981. [PubMed]
- M Levitt, A Wilson, D Bowman, S Kemel, G Krepart, V Marks. Physiologic observations in a controlled clinical trial of the antiemetic effectiveness of 5, 10, and 15 mg of delta 9-tetrahydrocannabinol in cancer chemotherapy. Ophthalmologic implications. J Clin Pharmacol, 1981. [PubMed]
- AM Li, SR Rassekh. Hypotension associated with ingestion of cannabinoids in two children with cancer. CMAJ, 2016. [PubMed]
- AH Lichtman, EA Lux, R McQuade, S Rossetti, R Sanchez, W Sun. Results of a double-blind, randomized, placebo-controlled study of nabiximols oromucosal spray as an adjunctive therapy in advanced cancer patients with chronic uncontrolled pain. J Pain Symptom Manage, 2018. [PubMed]
- VS Lucas, J Laszlo. delta 9-Tetrahydrocannabinol for refractory vomiting induced by cancer chemotherapy. JAMA, 1980. [PubMed]
- HH Lucraft, MK Palmer. Randomised clinical trial of levonantradol and chlorpromazine in the prevention of radiotherapy-induced vomiting. Clin Radiol, 1982. [PubMed]
- ME Lynch, P Cesar-Rittenberg, AG Hohmann. A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manage, 2014. [PubMed]
- DM Macari, B Gbadamosi, I Jaiyesimi, S Gaikazian. Medical cannabis in cancer patients: a survey of a community hematology oncology population. Am J Clin Oncol, 2020. [PubMed]
- K Madden, K Tanco, E Bruera. Clinically significant drug-drug interaction between methadone and cannabidiol. Pediatrics, 2020. [PubMed]
- V Maida. Nabilone for the treatment of paraneoplastic night sweats: a report of four cases. J Palliat Med, 2008. [PubMed]
- V Maida. Medical cannabis in the palliation of malignant wounds-a case report. J Pain Symptom Manage, 2017. [PubMed]
- V Maida, M Ennis, S Irani, M Corbo, M Dolzhykov. Adjunctive nabilone in cancer pain and symptom management: a prospective observational study using propensity scoring. J Supportive Oncol, 2008
- 112.Marchese M, Zhu C, Charbonneau LF, Peragine C, De Angelis C. Description and disposition of patients with cancer accessing a novel, pharmacist-led cannabis consultation service. JCO Oncology Practice. 2022;313((Marchese, Charbonneau, Peragine, De Angelis) Department of Pharmacy, Sunnybrook Odette Cancer Centre, Toronto, Canada(Marchese, Zhu, De Angelis) Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, Canada(De Angelis) Sunnybrook Research Instit):00748
- M McCabe, FP Smith, JS Macdonald, PV Woolley, D Goldberg, PS Schein. Efficacy of tetrahydrocannabinol in patients refractory to standard antiemetic therapy. Invest New Drugs, 1988. [PubMed]
- E Meiri, H Jhangiani, JJ Vredenburgh, LM Barbato, FJ Carter, H-M Yang. Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr Med Res Opin, 2007. [PubMed]
- P Mekavuthikul, J Phuditshinnapatra, S Chomchai. Cannabis oil could provoke secondary myocardial infarction in a high-risk patient after dropping into both ear and sublingual administration: a case report. Clin Toxicol, 2020
- CM Melen, M Merrien, AM Wasik, G Panagiotidis, O Beck, K Sonnevi. Clinical effects of a single dose of cannabinoids to patients with chronic lymphocytic leukemia. Leuk Lymphoma, 2022. [PubMed]
- S Merkle, SS Tavernier. Cannabis use and bleomycin: an overview and case study of pulmonary toxicity. Clin J Oncol Nurs, 2018. [PubMed]
- JA Neidhart, MM Gagen, HE Wilson, DC Young. Comparative trial of the antiemetic effects of THC and haloperidol. J Clin Pharmacol, 1981. [PubMed]
- K Nelson, D Walsh, P Deeter, F Sheehan. A phase II study of delta-9-tetrahydrocannabinol for appetite stimulation in cancer-associated anorexia. J Palliat Care, 1994. [PubMed]
- N Niederle, J Schutte, CG Schmidt. Crossover comparison of the antiemetic efficacy of nabilone and alizapride in patients with nonseminomatous testicular cancer receiving cisplatin therapy. Klin Wochenschr, 1986. [PubMed]
- SW Nielsen, SD Hasselsteen, HSH Dominiak, D Labudovic, L Reiter, SO Dalton. Oral cannabidiol for prevention of acute and transient chemotherapy-induced peripheral neuropathy. Support Care Cancer, 2022. [PubMed]
- SW Nielsen, CH Ruhlmann, L Eckhoff, D Bronnum, J Herrstedt, SO Dalton. Cannabis use among Danish patients with cancer: a cross-sectional survey of sociodemographic traits, quality of life, and patient experiences. Supportive Care Cancer, 2022
- A Niiranen, K Mattson. A cross-over comparison of nabilone and prochlorperazine for emesis induced by cancer chemotherapy. Am J Clin Oncol, 1985. [PubMed]
- A Niiranen, K Mattson. Antiemetic efficacy of nabilone and dexamethasone: a randomized study of patients with lung cancer receiving chemotherapy. Am J Clin Oncol, 1987. [PubMed]
- R Noyes, SF Brunk, DA Avery, AC Canter. The analgesic properties of delta-9-tetrahydrocannabinol and codeine. Clin Pharmacol Ther, 1975. [PubMed]
- R Noyes, SF Brunk, DA Baram, A Canter. Analgesic effect of delta-9-tetrahydrocannabinol. J Clin Pharmacol, 1975. [PubMed]
- R Ofir, G Bar-Sela, M Weyl Ben-Arush, S Postovsky. Medical marijuana use for pediatric oncology patients: single institution experience. Pediatr Hematol Oncol, 2019. [PubMed]
- LE Orr, JF McKernan, B Bloome. Antiemetic effect of tetrahydrocannabinol. Compared with placebo and prochlorperazine in chemotherapy-associated nausea and emesis. Arch Internal Med, 1980. [PubMed]
- V Parihar, A Rogers, AM Blain, SRK Zacharias, LL Patterson, MA-M Siyam. Reduction in tamoxifen metabolites endoxifen and N-desmethyltamoxifen with chronic administration of low dose cannabidiol: a CYP3A4 and CYP2D6 drug interaction. J Pharm Pract, 2022. [PubMed]
- LM Philpot, P Ramar, A Jatoi, J Rosedahl, R Canning, JO Ebbert. Cannabis in cancer survivors who report high impact chronic pain: findings from a 1500+ patient survey. Am J Hospice Palliative Care, 2022
- H Poghosyan, EJ Noonan, P Badri, I Braun, GJ Young. Association between daily and non-daily cannabis use and depression among United States adult cancer survivors. Nurs Outlook, 2021. [PubMed]
- S Polito, T MacDonald, M Romanick, J Jupp, J Wiernikowski, A Vennettilli. Safety and efficacy of nabilone for acute chemotherapy-induced vomiting prophylaxis in pediatric patients: a multicenter, retrospective review. Pediatr Blood Cancer, 2018. [PubMed]
- M Pomeroy, JJ Fennelly, M Towers. Prospective randomized double-blind trial of nabilone versus domperidone in the treatment of cytotoxic-induced emesis. Cancer Chemother Pharmacol, 1986. [PubMed]
- RK Portenoy, ED Ganae-Motan, S Allende, R Yanagihara, L Shaiova, S Weinstein. Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain, 2012. [PubMed]
- D Portman, KA Donovan. Cannabinoid hyperemesis syndrome: a case report of a confounding entity in oncology care. J Oncol Practice, 2018
- D Portman, KA Donovan. Case of pneumonitis associated with cannabis vaping and cancer immunotherapy. JCO Oncol Practice, 2020
- E Pralong, R Maduri, RT Daniel, M Messerer. Cannabinoid-induced alteration of motor-evoked potentials (MEPs) prior to intradural spinal tumor removal: a nasty surprise. Child’s Nervous Syst, 2018
- TJ Priestman, SG Priestman. An initial evaluation of Nabilone in the control of radiotherapy-induced nausea and vomiting. Clin Radiol, 1984. [PubMed]
- NJ Raghunathan, J Brens, S Vemuri, QS Li, JJ Mao, D Korenstein. In the weeds: a retrospective study of patient interest in and experience with cannabis at a cancer center. Supportive Care Cancer, 2022
- M Reblin, S Sahebjam, NC Peeri, YC Martinez, Z Thompson, KM Egan. Medical cannabis use in glioma patients treated at a comprehensive cancer center in Florida. J Palliat Med, 2019. [PubMed]
- RA Roffman. Stress inoculation training in the control of THC toxicities. Int J Addict, 1986. [PubMed]
- S Russmann, A Winkler, KO Lovblad, Z Stanga, C Bassetti. Lethal ischemic stroke after cisplatin-based chemotherapy for testicular carcinoma and cannabis inhalation. Eur Neurol, 2002. [PubMed]
- CE Saadeh, DR Rustem. Medical marijuana use in a community cancer center. J Oncol Practice, 2018
- SE Sallan, C Cronin, M Zelen, NE Zinberg. Antiemetics in patients receiving chemotherapy for cancer: a randomized comparison of delta-9-tetrahydrocannabinol and prochlorperazine. N Engl J Med, 1980. [PubMed]
- SE Sallan, NE Zinberg, E Frei. Antiemetic effect of delta-9-tetrahydrocannabinol in patients receiving cancer chemotherapy. N Engl J Med, 1975. [PubMed]
- N Sarid, M Zada, S Lev-Ran, E Yashphe, I Givon, M Barzilai. Medical cannabis use by Hodgkin lymphoma patients: experience of a single center. Acta Haematol, 2018. [PubMed]
- J Schloss, J Lacey, J Sinclair, A Steel, M Sughrue, D Sibbritt. A phase 2 randomised clinical trial assessing the tolerability of two different ratios of medicinal cannabis in patients with high grade gliomas. Front Oncol, 2021. [PubMed]
- G Schmidt-Wolf, P Cremer-Schaeffer. Interim results of the survey accompanying the prescription of cannabis-based medicines in Germany regarding dronabinol. Deutsches Arzteblatt Int, 2021
- H Senderovich, S Waicus. A case report on cannabinoid hyperemesis syndrome in palliative care: how good intentions can go wrong. Oncol Res Treat, 2022. [PubMed]
- VR Sheidler, DS Ettinger, RB Diasio, JP Enterline, MD Brown. Double-blind multiple-dose crossover study of the antiemetic effect of intramuscular levonantradol compared to prochlorperazine. J Clin Pharmacol, 1984. [PubMed]
- Y Singh, C Bali. Cannabis extract treatment for terminal acute lymphoblastic leukemia with a Philadelphia chromosome mutation. Case Rep Oncol, 2013. [PubMed]
- JE Stambaugh, J McAdams, F Vreeland. Dose ranging evaluation of the antiemetic efficacy and toxicity of intramuscular levonantradol in cancer subjects with chemotherapy-induced emesis. J Clin Pharmacol, 1984. [PubMed]
- M Staquet, C Gantt, D Machin. Effect of a nitrogen analog of tetrahydrocannabinol on cancer pain. Clin Pharmacol Ther, 1978. [PubMed]
- N Steele, RJ Gralla, DW Braun, CW Young. Double-blind comparison of the antiemetic effects of nabilone and prochlorperazine on chemotherapy-induced emesis. Cancer Treat Rep, 1980. [PubMed]
- F Strasser, D Luftner, K Possinger, G Ernst, T Ruhstaller, W Meissner. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabi. J Clin Oncol, 2006. [PubMed]
- RC Stuart-Harris, CA Mooney, IE Smith. Levonantradol: a synthetic cannabinoid in the treatment of severe chemotherapy-induced nausea and vomiting resistant to conventional anti-emetic therapy. Clin Oncol, 1983. [PubMed]
- KT Sura, L Kohman, D Huang, SV Pasniciuc. Experience with medical marijuana for cancer patients in the palliative setting. Cureus, 2022. [PubMed]
- S Sutton, BL Lum, FM Torti. Possible risk of invasive pulmonary aspergillosis with marijuana use during chemotherapy for small cell lung cancer. Drug Intell Clin Pharm, 1986. [PubMed]
- DL Sweet, NJ Miller, W Weddington, E Senay, L Sushelsky. delta 9-Tetrahydrocannabinol as an antiemetic for patients receiving cancer chemotherapy. A pilot study. J Clin Pharmacol, 1981. [PubMed]
- M Szyper-Kravitz, R Lang, Y Manor, M Lahav. Early invasive pulmonary aspergillosis in a leukemia patient linked to aspergillus contaminated marijuana smoking. Leuk Lymphoma, 2001. [PubMed]
- T Taha, D Meiri, S Talhamy, M Wollner, A Peer, G Bar-Sela. Cannabis impacts tumor response rate to nivolumab in patients with advanced malignancies. Oncologist, 2019. [PubMed]
- SD Tavhare, R Acharya, RG Reddy, KS Dhiman. Management of chronic pain with Jalaprakshalana (water-wash) Shodhita (processed) Bhanga (Cannabis sativa L.) in cancer patients with deprived quality of life: an open-label single arm clinical trial. Ayu., 2019. [PubMed]
- C Tofthagen, A Perlman, P Advani, B Ernst, J Kaur, W Tan. Medical marijuana use for cancer-related symptoms among Floridians: a descriptive study. J Palliat Med, 2022. [PubMed]
- C Tschoe, L Johnson, A Giugliano, A Sarwal. Serotonin syndrome with exposure from tetrahydrocannabinol: a case report to highlight the side effects of increasing use of cbd products. Neurology, 2020
- JG Turcott, M del Rocio Guillen Nunez, D Flores-Estrada, LF Onate-Ocana, ZL Zatarain-Barron, F Barron. The effect of nabilone on appetite, nutritional status, and quality of life in lung cancer patients: a randomized, double-blind clinical trial. Support Care Cancer, 2018. [PubMed]
- C Twelves, M Sabel, D Checketts, S Miller, B Tayo, M Jove. A phase 1b randomised, placebo-controlled trial of nabiximols cannabinoid oromucosal spray with temozolomide in patients with recurrent glioblastoma. Br J Cancer, 2021. [PubMed]
- LB Tyson, RJ Gralla, RA Clark, MG Kris, LA Bordin, GJ Bosl. Phase 1 trial of levonantradol in chemotherapy-induced emesis. Am J Clin Oncol, 1985. [PubMed]
- JT Ungerleider, T Andrysiak, L Fairbanks, J Goodnight, G Sarna, K Jamison. Cannabis and cancer chemotherapy: a comparison of oral delta-9-THC and prochlorperazine. Cancer, 1982. [PubMed]
- JT Ungerleider, G Sarna, LA Fairbanks, J Goodnight, T Andrysiak, K Jamison. THC or Compazine for the cancer chemotherapy patient–the UCLA study Part II: Patient drug preference. Am J Clin Oncol, 1985. [PubMed]
- JGC van Hasselt, MM van den Heuvel, JHM Schellens, JH Beijnen, D Brandsma, ADR Huitema. Severe cannabinoid intoxication in a patient with non-small-cell lung cancer. J Palliat Care, 2012. [PubMed]
- B Waissengrin, D Urban, Y Leshem, M Garty, I Wolf. Patterns of use of medical cannabis among Israeli cancer patients: a single institution experience. J Pain Symptom Manage, 2015. [PubMed]
- D Walsh, J Kirkova, MP Davis. The efficacy and tolerability of long-term use of dronabinol in cancer-related anorexia: a case series. J Pain Symptom Manage, 2005. [PubMed]
- PP Warren, EM Bebin, LB Nabors, JP Szaflarski. The use of cannabidiol for seizure management in patients with brain tumor-related epilepsy. Neurocase, 2017. [PubMed]
- EM Webster, GS Yadav, S Gysler, B McNamara, J Black, J Tymon-Rosario. Prescribed medical cannabis in women with gynecologic malignancies: a single-institution survey-based study. Gynecol Oncol Reports, 2020
- J Welsh, F Stuart, G Sangster, R Milstead, S Kaye, H Cash. Oral levonantradol in the control of cancer chemotherapy-induced emesis. Cancer Chemother Pharmacol, 1983. [PubMed]
- P Zaki, A Blake, A Wolt, S Chan, L Zhang, A Wan. The use of medical cannabis in cancer patients. J Pain Manag, 2017
- 177.Zhou ES, Nayak MM, Chai PR, Braun IM. Cancer patient’s attitudes of using medicinal cannabis for sleep. Journal of psychosocial oncology. 2021((Zhou, Nayak, Chai, Braun) Department of Psychosocial Oncology and Palliative Care, Dana-Farber Cancer Institute, MA, Boston, United States(Zhou) Division of Sleep Medicine, Harvard Medical School, MA, Boston, United States(Chai) Division of Medical Toxic):1–7
- Y Zolotov, L Eshet, O Morag. Preliminary assessment of medical cannabis consumption by cancer survivors. Complement Therapies Med, 2021
- DM Zylla, J Eklund, G Gilmore, A Gavenda, J Guggisberg, G VazquezBenitez. A randomized trial of medical cannabis in patients with stage IV cancers to assess feasibility, dose requirements, impact on pain and opioid use, safety, and overall patient satisfaction. Supportive Care Cancer, 2021
- JD Brown, AG Winterstein. Potential adverse drug events and drug-drug interactions with medical and consumer cannabidiol (CBD) use. J Clin Med, 2019. [PubMed]
- JD Brown. Potential adverse drug events with tetrahydrocannabinol (THC) due to drug-drug interactions. J Clin Med, 2020. [PubMed]
- K Johnson, AJ Weldon, MA Burmeister. Differential effects of cannabis constituents on schizophrenia-related psychosis: a rationale for incorporating cannabidiol into a schizophrenia therapeutic regimen. Front Psychiatry, 2024. [PubMed]
- Z Latif, N Garg. The impact of marijuana on the cardiovascular system: a review of the most common cardiovascular events associated with marijuana use. J Clin Med, 2020. [PubMed]
- S Kitdumrongthum, D Trachootham. An individuality of response to cannabinoids: challenges in safety and efficacy of cannabis products. Molecules, 2023. [PubMed]
- RT Smith, SA Gruber. Contemplating cannabis? The complex relationship between cannabinoids and hepatic metabolism resulting in the potential for drug-drug interactions. Front Psychiatry, 2022. [PubMed]
- MA Alsherbiny, CG Li. Medicinal cannabis-potential drug interactions. Medicines (Basel), 2018. [PubMed]
- M Babayeva, ZG Loewy. Cannabis pharmacogenomics: a path to personalized medicine. Curr Issues Mol Biol, 2023. [PubMed]
- V Lopera, A Rodríguez, P Amariles. Clinical relevance of drug interactions with cannabis: a systematic review. J Clin Med, 2022. [PubMed]
